As filed with the Securities and Exchange Commission on June 26, 2019
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
Castle Biosciences, Inc.
(Exact Name of Registrant as Specified in Its Charter)
|
Delaware
|
8071
|
77-0701774
|
|
(State or Other Jurisdiction of
Incorporation or Organization) |
(Primary Standard Industrial
Classification Code Number) |
(I.R.S. Employer
Identification Number) |
820 S. Friendswood Drive, Suite 201
Friendswood, Texas 77546
(866) 788-9007
(Address, Including Zip Code, and Telephone Number, Including Area Code, of Registrant’s Principal Executive Offices)
Derek Maetzold
President and Chief Executive Officer
Castle Biosciences, Inc.
820 S. Friendswood Drive, Suite 201
Friendswood, Texas 77546
(866) 788-9007
(Name, Address, Including Zip Code, and Telephone Number, Including Area Code, of Agent for Service)
Copies to:
|
Thomas A. Coll, Esq.
Divakar Gupta, Esq. Karen E. Anderson, Esq. Cooley LLP 4401 Eastgate Mall San Diego, California 92121 (858) 550-6000 |
Peter N. Handrinos, Esq.
Anthony Gostanian, Esq. Latham & Watkins LLP 200 Clarendon Street Boston, Massachusetts 02116 (617) 880-4500 |
Approximate date of commencement of proposed sale to the public:
As soon as practicable after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933 check the following box: o
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer
|
o
|
Accelerated filer
|
o
|
|
Non-accelerated filer
|
☒
|
Smaller reporting company
|
☒
|
|
|
|
Emerging growth company
|
☒
|
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. o
CALCULATION OF REGISTRATION FEE
Title of each class of securities to be registered |
Proposed maximum aggregate offering price(1) |
Amount of registration fee(2) |
||||
Common Stock, $0.001 par value per share |
$ | 57,500,000 | $ | 6,969 | ||
| (1) | Estimated solely for the purpose of calculating the amount of the registration fee in accordance with Rule 457(o) under the Securities Act of 1933, as amended. Includes the offering price of shares that the underwriters have the option to purchase. |
| (2) | Calculated pursuant to Rule 457(o) based on an estimate of the proposed maximum aggregate offering price. |
The Registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment that specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the registration statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
The information in this preliminary prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and we are not soliciting offers to buy these securities in any jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION, DATED JUNE 26, 2019
PRELIMINARY PROSPECTUS
Shares

Common Stock
This is the initial public offering of Castle Biosciences, Inc. We are offering shares of our common stock. Prior to this offering, there has been no public market for our common stock. We estimate that the initial public offering price of our common stock will be between $ and $ per share.
We have applied to list our common stock on The Nasdaq Global Select Market under the symbol “CSTL.”
We are an “emerging growth company” as that term is used in the Jumpstart Our Business Startups Act of 2012 and, as such, we have elected to comply with certain reduced public company reporting requirements for this prospectus and future filings.
Investing in our common stock involves a high degree of risk. See “Risk Factors” beginning on page 11.
Per Share |
Total |
|||||
Initial public offering price |
$ | $ | ||||
Underwriting discounts and commissions(1) |
$ | $ | ||||
Proceeds to us (before expenses) |
$ | $ | ||||
| (1) | We have agreed to reimburse the underwriters for certain expenses. See “Underwriting.” |
We have granted the underwriters a 30-day option to purchase up to a total of additional shares of common stock from us at the initial public offering price less the underwriting discounts and commissions.
The underwriters expect to deliver the shares of common stock to purchasers on or about , 2019 through the book-entry facilities of The Depository Trust Company.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
Joint Book-Running Managers
|
SVB Leerink
|
Baird
|
Co-Managers
|
Canaccord Genuity
|
BTIG
|
The date of this prospectus is , 2019
TABLE OF CONTENTS
Page |
|||
We have not, and the underwriters have not, authorized anyone to provide any information or to make any representations other than those contained in this prospectus or in any free writing prospectuses prepared by or on behalf of us or to which we have referred you. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. This prospectus is an offer to sell only the shares offered hereby, but only under circumstances and in jurisdictions where it is lawful to do so. The information contained in this prospectus or in any applicable free writing prospectus is current only as of its date, regardless of its time of delivery or any sale of shares of our common stock. Our business, financial condition, results of operations and prospects may have changed since that date.
Through and including , 2019 (25 days after the commencement of this offering), all dealers that buy, sell or trade shares of our common stock, whether or not participating in this offering, may be required to deliver a prospectus. This delivery requirement is in addition to the dealers’ obligation to deliver a prospectus when acting as underwriters and with respect to their unsold allotments or subscriptions.
This prospectus includes statistical and other industry and market data that we obtained from industry publications and research, surveys and studies conducted by third parties. Industry publications and third-party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information.
For investors outside the United States: we have not, and the underwriters have not, done anything that would permit this offering or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. Persons outside the United States who come into possession of this prospectus must inform themselves about, and observe any restrictions relating to, the offering of the shares of common stock and the distribution of this prospectus outside the United States.
i
This summary highlights information contained in other parts of this prospectus. Because it is only a summary, it does not contain all of the information that you should consider before investing in shares of our common stock and it is qualified in its entirety by, and should be read in conjunction with, the more detailed information appearing elsewhere in this prospectus. You should read the entire prospectus carefully, especially “Risk Factors” and our financial statements and the related notes, before deciding to buy shares of our common stock. Unless the context requires otherwise, references in this prospectus to “Castle Biosciences,” “we,” “us” and “our” refer to Castle Biosciences, Inc.
Overview
We are a commercial-stage dermatological cancer company focused on providing physicians and their patients with personalized, clinically actionable genomic information to make more accurate treatment decisions. We believe that the traditional approach to developing a treatment plan for certain cancers using clinical and pathology factors alone is inadequate and can be improved by incorporating personalized genomic information. Our non-invasive products utilize proprietary algorithms to provide an assessment of a patient’s specific risk of metastasis or recurrence of their cancer, allowing physicians to identify patients who are likely to benefit from an escalation of care as well as those who may avoid unnecessary medical and surgical interventions. Our lead product, DecisionDx-Melanoma, is a proprietary multi-gene expression profile, or GEP, test that predicts the risk of metastasis or recurrence for patients diagnosed with invasive cutaneous melanoma, a deadly skin cancer. We also market DecisionDx-UM, which is a proprietary GEP test that predicts the risk of metastasis for patients with uveal melanoma, a rare eye cancer. Based on the substantial clinical evidence that we have developed, we have received Medicare coverage for both of our products, which represents approximately 50% of our addressable patient population. We also have two late-stage proprietary products in development that address cutaneous squamous cell carcinoma, or SCC, and suspicious pigmented lesions which are indications with high clinical need in dermatological cancer.
Our focus on dermatologic cancers has provided us with unique insights that have enabled us to drive adoption of DecisionDx-Melanoma, as well as to identify opportunities for additional products to address unmet clinical needs in dermatologic cancer. We have processed over 40,000 clinical samples since commercial launch, with total proprietary GEP report volume increasing from less than 4,000 in 2015 to more than 13,400 in 2018. Our annual revenue increased from $13.8 million in 2017 to $22.8 million in 2018.
Skin cancer is the most commonly diagnosed cancer in the United States. There are more than 5.5 million new cases of skin cancer diagnosed annually, compared with 1.6 million new cases for all other cancers combined. DecisionDx-Melanoma targets more than an estimated 100,000 patients diagnosed with invasive cutaneous melanoma each year, which we believe is underreported. In addition, our two late-stage proprietary products target approximately 200,000 patients diagnosed with SCC with high-risk features and approximately 300,000 patients with suspicious pigmented lesions without a definitive diagnosis of skin cancer. We estimate that the total addressable U.S. market for these three indications is approximately $1.8 billion.
Healthcare providers, predominately dermatologists and surgeons who treat melanoma patients, make nearly all treatment decisions for patients diagnosed with skin cancers based upon their expected risk of metastasis or recurrence. Historically these treatment decisions have been based solely on clinical and pathology factors, such as tumor depth or width and evidence of metastasis to the sentinel lymph node, or SLN. Physicians use these factors to group, or stage, patients into stage-related populations. The average risk of metastasis within a population then guides treatment decisions for all patients within a respective population. However, an individual patient’s risk of metastasis can be significantly different from these stage-related population averages, thereby resulting in some patients receiving unnecessary medical and surgical interventions and some patients being undertreated. This treatment paradigm has led to suboptimal patient care and unnecessary costs to the healthcare system.
We believe that incorporating the genomics of each individual patient’s tumor biology to inform on their specific risk of metastasis can aid the decision-making process for their treatment plan, help optimize health outcomes and reduce healthcare costs. The genomics of cutaneous melanoma and other skin cancers are highly complex because, unlike some other types of cancer, the presence or absence of a single gene or a limited number of genes has not been shown to accurately predict the risk of metastasis or recurrence. Rather, we believe that risk of metastasis or recurrence of skin cancer requires the analysis of gene expression profiles occurring at the RNA
1
level through the application of artificial intelligence, deep learning and proprietary techniques to identify clinically relevant genomic patterns. Once identified, we then undertake extensive clinical validation and clinical utility studies to develop products that address key unmet medical needs for patients and physicians.
We developed our proprietary, non-invasive DecisionDx-Melanoma product for cutaneous melanoma to utilize gene expression to provide more complete information. The product interrogates the biology of a patient’s tumor by analyzing the gene expression profile of 31 genes, a process made possible by our proprietary algorithm, developed using machine learning techniques. We have published 18 studies to support the two current clinically actionable uses of DecisionDx-Melanoma, analyzing more than 3,700 patient samples. We also market DecisionDx-UM, a genomic test for use in identifying patients diagnosed with uveal melanoma who are at a low risk of metastasis. Similar to DecisionDx-Melanoma, this product also uses a proprietary algorithm developed using machine learning techniques to interrogate the biology of a patient’s tumor by analyzing the gene expression profile of 15 genes of a patient’s tumor.
We are developing additional products targeting the challenges faced by physicians in treating their patients’ skin cancer, with two products in late-stage development. DecisionDx-SCC is a proprietary GEP test designed to predict the risk of metastasis in patients diagnosed with SCC. We are also developing a proprietary GEP test designed to assist physicians in the diagnosis of suspicious pigmented lesions. We intend to commercially launch both of these products in the second half of 2020.
Our Competitive Advantages
We are focused on providing actionable genomic information to physicians and their patients. We believe our key competitive advantages are due in part to the following factors:
| • | Development of our products requires our machine learning expertise and our proprietary algorithm, which are complex and difficult to replicate. |
| • | We have demonstrated the ability to provide clinically actionable information despite the complex genomics of skin cancer. |
| • | Our vast and growing database of tumor samples and associated long-term outcomes data enables us to improve our current products and accelerate development of new products. |
| • | We have generated, and will continue to generate, robust clinical validity and utility data supporting the use of our products. |
| • | We have established relationships with physicians that allow us to optimize our interactions, increase adoption of our current products and identify areas of unmet clinical need to efficiently launch additional products. |
| • | We have experience in navigating the reimbursement landscape. |
Dermatologic Cancer Market Overview
Skin cancer is the uncontrolled growth of abnormal skin cells. There are six types of pre-cancers and skin cancers that result in a total annual incidence of 5.5 million patients, three of which are highlighted in the figure below. The three most common forms of skin cancers are basal cell carcinomas, SCC and cutaneous melanoma. Pre-cancers include suspicious pigmented lesions, which are unusual-looking lesions that may be melanoma.
2

Our Solution and Products
Nearly all treatment plan decisions for dermatological cancer are based upon an estimate of a patient’s prognosis — whether their cancer is more or less likely to metastasize. We use the gene expression profile of an individual patient’s tumor biology to inform specific prognosis of metastasis or recurrence and aid the treatment plan decision-making process of the treating physician and their patient to help optimize health outcomes and reduce healthcare costs. Due to the biological complexity of skin cancers, developing accurate products takes scientific diligence, stringent clinical protocols, machine learning expertise, proprietary algorithms and significant investments of time and capital. In addition, the underlying tissue samples and associated outcomes data required to develop and validate these products are difficult to obtain. Once successfully developed and validated, commercial success requires the generation of clinical use documentation to support appropriate physician adoption, reimbursement success and guideline inclusion.
The table below summarizes our progress with our two commercial, proprietary products as well as our two near-term product candidates:

3
Our Strategy
We intend to build upon our position as a leading provider of genomic information for dermatological cancers. To realize this objective we plan to:
| • | Expand adoption of our currently marketed products and educate physicians and their patients on the need for our products to make a more informed treatment plan decision. We believe that cancer treatment plans will be most effective if decisions are personalized for each patient based on the biology of their specific tumor, instead of a one-size-fits-all approach. We will continue to educate physicians and their patients on the diagnostic discordance that leads to over- and under-treatment. |
| • | Continue to generate evidence supporting the clinical utility and validity of our products. We have conducted extensive clinical utility and validity studies to support the adoption of, and reimbursement for, our products. In order to maintain our competitive advantage and increase sales of our products, we will continue to generate additional clinical data to support the use of our products. |
| • | Execute planned expansion of our commercial channel. We plan to increase sales of our products by adding new physicians to our customer base as well as increasing orders by physicians already using our products. We recently increased the number of sales and medical affairs representatives and are planning additional increases through 2019 in order to provide more frequent physician interactions and support the launch of additional products. |
| • | Expand coverage and reimbursement for our products. We plan to have increasing dialogue with third-party payors to highlight our clinical utility and patient outcomes data. We believe these data will validate the benefit of our products for patients and will persuade more third-party payors to provide coverage and reimbursement. Additionally, we will continue to emphasize our ability to reduce overall cost to the healthcare system by appropriately classifying high-risk patients and removing the need for unnecessary invasive procedures for low-risk patients. |
| • | Utilize our development expertise and commercial channel insight to provide additional solutions. We are continuing to develop products that address the challenges facing physicians, including genomic tests for patients with SCC with high-risk factors and suspicious pigmented lesions, addressing an aggregate of approximately 500,000 additional potential patients. |
Risks Associated with This Offering
Our business and our ability to implement our business strategy are subject to numerous risks, as more fully described in the section entitled “Risk Factors” immediately following this prospectus summary. You should read these risks before you invest in our common stock. We may be unable, for many reasons, including those that are beyond our control, to implement our business strategy. In particular, risks associated with our business include:
| • | Our reliance upon a small number of third-party payors for a significant portion of our revenue may materially adversely affect our financial condition and results of operations. |
| • | Due to how we recognize revenue, our quarterly revenues may not reflect our underlying business. |
| • | We have incurred significant losses since inception, and we may never achieve or sustain profitability. |
| • | Our revenue currently depends primarily on sales of DecisionDx-Melanoma, and we will need to generate sufficient revenue from this and other products to grow our business. |
| • | Billing for our products is complex and requires substantial time and resources to collect payment. |
| • | New product development involves a lengthy and complex process, and we may be unable to develop and commercialize, or receive reimbursement for, on a timely basis, or at all, new products. |
| • | We currently have limited reimbursement coverage for our lead product, DecisionDx-Melanoma, and if third-party payors, including government and commercial payors, do not provide sufficient coverage of, or adequate reimbursement for, our products, our commercial success will be negatively affected. |
| • | Interim, topline and preliminary data from our clinical studies that we announce or publish from time to time may change as more data become available and are subject to audit and verification procedures that could result in material changes in the final data. |
4
| • | We are subject to numerous federal and state healthcare statutes and regulations, and complying with laws pertaining to our business is an expensive and time-consuming process. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties and a material adverse effect to our business and operations. |
| • | Our products are currently marketed as laboratory developed tests, and any changes in regulations or the U.S. Food and Drug Administration’s enforcement discretion for laboratory developed tests, or violations of regulations by us, could adversely affect our business, prospects, results of operations or financial condition. |
| • | If we are unable to obtain and maintain sufficient intellectual property protection for our technology, or if the scope of the intellectual property protection obtained is not sufficiently broad, our competitors could develop and commercialize diagnostic tests similar or identical to ours, and our ability to successfully commercialize our products may be impaired. |
Corporate and Other Information
We were incorporated in Delaware in September 2007. Our principal executive offices are located at 820 S. Friendswood Drive, Suite 201, Friendswood, Texas 77546, and our telephone number is (866) 788-9007. Our corporate website address is www.CastleBiosciences.com. Information contained on or accessible through our website is not a part of this prospectus, and the inclusion of our website address in this prospectus is an inactive textual reference only.
This prospectus contains references to our trademarks and to trademarks belonging to other entities. Solely for convenience, trademarks and trade names referred to in this prospectus, including logos, artwork and other visual displays, may appear without the ® or TM symbols, but such references are not intended to indicate, in any way, that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. We do not intend our use or display of other companies’ trade names or trademarks to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
Implications of Being an Emerging Growth Company and a Smaller Reporting Company
We qualify as an emerging growth company as defined in the Jumpstart Our Business Startups Act, as amended, or the JOBS Act, enacted in April 2012. An emerging growth company may take advantage of reduced reporting requirements that are otherwise applicable to public companies. These provisions include, but are not limited to:
| • | being permitted to present only two years of audited financial statements and only two years of related Management’s Discussion and Analysis of Financial Condition and Results of Operations in this prospectus; |
| • | not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, as amended, or the Sarbanes-Oxley Act; |
| • | reduced disclosure obligations regarding executive compensation in our periodic reports, proxy statements and registration statements; and |
| • | exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and any golden parachute payments not previously approved. |
We may take advantage of these reporting exemptions until we are no longer an emerging growth company. We will remain an emerging growth company until the earlier of (1) the last day of the fiscal year (a) following the fifth anniversary of the completion of this offering, (b) in which we have total annual gross revenue of at least $1.07 billion or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates exceeds $700 million as of the prior June 30th and (2) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period.
We have elected to take advantage of certain of the reduced disclosure obligations in the registration statement of which this prospectus is a part and may elect to take advantage of other reduced reporting requirements in future filings. As a result, the information that we provide to our stockholders may be different than you might receive from other public reporting companies in which you hold equity interests.
The JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with the adoption of new or revised accounting standards. We have elected to avail ourselves of
5
this exemption. Therefore, we may not be subject to the same implementation timing for new or revised accounting standards as other public companies that are not emerging growth companies, which may make comparison of our financial statements to those of other public companies more difficult.
We are also a smaller reporting company as defined in the Securities Exchange Act of 1934, as amended, or the Exchange Act. We may continue to be a smaller reporting company even after we are no longer an emerging growth company. We may take advantage of certain of the scaled disclosures available to smaller reporting companies and will be able to take advantage of these scaled disclosures for so long as our voting and non-voting common stock held by non-affiliates is less than $250.0 million measured on the last business day of our second fiscal quarter, or our annual revenue is less than $100.0 million during the most recently completed fiscal year and our voting and non-voting common stock held by non-affiliates is less than $700.0 million measured on the last business day of our second fiscal quarter.
6
The Offering
| • | approximately $17 million for selling and marketing activities, including expansion of our sales force to support the ongoing commercialization of our current products and future products; |
| • | approximately $17 million for research and development, related to the continued support of our current products as well as the development of our product pipeline; and |
| • | the remainder for working capital and other general corporate purposes, including the additional costs associated with being a public company. |
See “Use of Proceeds” for additional information.
The number of shares of our common stock to be outstanding after this offering is based on 12,296,294 shares of common stock outstanding as of March 31, 2019, after giving effect to the conversion of our outstanding shares of convertible preferred stock into 9,959,831 shares of common stock, and excludes:
| • | 2,575,158 shares of common stock issuable upon the exercise of outstanding stock options as of March 31, 2019, at a weighted-average exercise price of $1.88 per share; |
| • | shares of common stock reserved for future issuance under the 2019 equity incentive plan, or the 2019 Plan, which will become effective upon the execution and delivery of the underwriting agreement for this offering (with such shares include new shares plus the number of shares (not to exceed shares) (i) that remain available for the issuance of awards under the 2018 equity incentive plan, or the 2018 Plan, at the time the 2019 Plan becomes effective, and (ii) any shares underlying outstanding stock awards granted under the 2018 Plan and the 2008 stock plan, or the 2008 Plan, that expire or are repurchased, forfeited, cancelled or withheld, as more fully described in the section titled “Executive and Director Compensation – Equity Incentive Plans”), as well as any automatic increases in the number of our common stock reserved for future issuance under the 2019 Plan; |
| • | shares of common stock reserved for future issuance under the 2019 employee stock purchase plan, or the ESPP, as well as any automatic increases in the number of our common stock reserved for future issuance under the ESPP, which will become effective upon the execution and delivery of the underwriting agreement for this offering; and |
| • | 137,935 shares of our common stock issuable upon the exercise of certain warrants outstanding as of March 31, 2019, at a weighted-average exercise price of $5.47 per share. |
7
Unless otherwise indicated, all information contained in this prospectus assumes or gives effect to:
| • | the conversion of all our outstanding shares of convertible preferred stock as of March 31, 2019, into an aggregate of 9,959,831 shares of common stock in connection with the completion of this offering; |
| • | the issuance by us of convertible promissory notes in January 2019 and February 2019 and the conversion of approximately $11.8 million of aggregate principal amount, plus accrued interest thereon, of such convertible promissory notes which will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019; |
| • | the net exercise of certain outstanding warrants to purchase shares of our Series F redeemable convertible preferred stock for an aggregate of shares of common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) in connection with the completion of this offering; |
| • | the adjustment of outstanding warrants to purchase shares of our convertible preferred stock into warrants to purchase shares of our common stock in connection with the completion of this offering; |
| • | no exercise by the underwriters of their option to purchase up to a total of additional shares of our common stock to cover overallotments; |
| • | no exercise of the outstanding options described above; |
| • | the filing of our amended and restated certificate of incorporation and the adoption of our amended and restated bylaws immediately prior to the completion of this offering; and |
| • | a one-for- reverse stock split of our common stock to be effected prior to the completion of this offering. |
A $1.00 increase in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would decrease the number of shares of our common stock issued on conversion of our convertible promissory notes (and therefore the number of shares to be outstanding after this offering) by shares. A $1.00 decrease in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would increase the number of shares of our common stock issued on conversion of our convertible promissory notes (and therefore the number of shares to be outstanding after this offering) by shares.
8
Summary Financial Data
The following tables set forth a summary of our financial data as of, and for the periods ended on, the dates indicated. We have derived the following summary of our statements of operations and comprehensive loss data for the years ended December 31, 2017 and 2018 and our balance sheet data as of December 31, 2018 from our audited financial statements appearing elsewhere in this prospectus. We have derived the following summary of our statements of operations and comprehensive loss data for the three months ended March 31, 2018 and 2019 and our balance sheet data as of March 31, 2019 from our unaudited interim condensed financial statements appearing elsewhere in this prospectus.You should read these data together with our financial statements and related notes appearing elsewhere in this prospectus and the information in “Selected Financial Data” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” Our historical results are not necessarily indicative of the results to be expected in the future, and our operating results for the interim periods are not necessarily indicative of the results that may be expected for any other interim periods or any future year.
Years Ended December 31, |
Three Months Ended March 31, |
|||||||||||
2017 |
2018 |
2018 |
2019 |
|||||||||
(unaudited) |
||||||||||||
(in thousands, except share and per share data) |
||||||||||||
Statements of Operations and Comprehensive Loss Data: |
||||||||||||
Net revenues |
$ | 13,754 | $ | 22,786 | $ | 3,659 | $ | 8,717 | ||||
Cost of sales |
4,922 | 5,297 | 1,253 | 1,598 | ||||||||
Gross margin |
8,832 | 17,489 | 2,406 | 7,119 | ||||||||
Operating expenses: |
||||||||||||
Research and development |
4,473 | 4,854 | 1,263 | 1,394 | ||||||||
Selling, general and administrative |
15,259 | 16,470 | 4,228 | 6,047 | ||||||||
Total operating expenses |
19,732 | 21,324 | 5,491 | 7,441 | ||||||||
Operating loss |
(10,900 | ) |
(3,835 | ) |
(3,085 | ) |
(322 | ) |
||||
Interest income |
26 | 24 | 5 | 21 | ||||||||
Interest expense |
(1,649 | ) |
(2,275 | ) |
(529 | ) |
(1,024 | ) |
||||
Other income (expense), net |
163 | (272 | ) |
(21 | ) |
(33 | ) |
|||||
Loss before income taxes |
(12,360 | ) |
(6,358 | ) |
(3,630 | ) |
(1,358 | ) |
||||
Income tax expense |
10 | 9 | — | — | ||||||||
Net loss and comprehensive loss |
(12,370 | ) |
(6,367 | ) |
(3,630 | ) |
(1,358 | ) |
||||
Convertible preferred stock cumulative dividends |
2,897 | 3,577 | 810 | 928 | ||||||||
Accretion of redeemable convertible preferred stock to redemption value |
41 | 219 | 49 | 56 | ||||||||
Net loss and comprehensive loss attributable to common stockholders |
$ | (15,308 | ) |
$ | (10,163 | ) |
$ | (4,489 | ) |
$ | (2,342 | ) |
Loss per share attributable to common stockholders, basic and diluted(1) |
$ | (6.62 | ) |
$ | (4.37 | ) |
$ | (1.94 | ) |
$ | (1.00 | ) |
Weighted-average shares outstanding, basic and diluted(1) |
2,310,640 | 2,323,197 | 2,312,515 | 2,336,236 | ||||||||
Pro forma loss per share, basic and diluted (unaudited)(1)(2) |
$ | $ | ||||||||||
Pro forma weighted-average shares outstanding, basic and diluted (unaudited)(1)(2) |
||||||||||||
9
| (1) | See Note 3 to our financial statements appearing elsewhere in this prospectus for an explanation of the method used to calculate the basic and diluted loss per share and the number of shares used in the computation of the per share amounts. |
| (2) | The calculations for the unaudited pro forma loss per share, basic and diluted, assume the conversion of all our outstanding shares of convertible preferred stock into shares of our common stock and the net exercise of certain outstanding warrants to purchase shares of our convertible preferred stock for common stock as if the conversion or net exercise had occurred at the beginning of the period presented or the issuance date, if later, upon completion of this offering. For the three months ended March 31, 2019, the calculations further assume the conversion into shares of common stock all outstanding principal and accrued interest related to the convertible promissory notes as if the conversion had occurred at the respective issuance dates of the convertible promissory notes. Because the convertible promissory notes were not issued until after December 31, 2018, the assumed conversion of such notes into shares of common stock had no impact on the calculations for the year ended December 31, 2018. |
As of March 31, 2019 |
|||||||||
Actual |
Pro forma(1) |
Pro forma as adjusted(2)(3) |
|||||||
(unaudited, in thousands) |
|||||||||
Balance Sheet Data: |
|||||||||
Cash and cash equivalents |
$ | 16,231 | $ | $ | |||||
Working capital(4) |
$ | 18,035 | |||||||
Total assets |
$ | 30,966 | |||||||
Convertible promissory notes(5) |
$ | 3,568 | |||||||
Long-term debt |
$ | 23,632 | |||||||
Preferred stock warrant liability |
$ | 1,181 | |||||||
Convertible preferred stock |
$ | 1,501 | |||||||
Redeemable convertible preferred stock |
$ | 45,051 | |||||||
Total stockholders’ equity (deficit) |
$ | (49,432 | ) |
||||||
| (1) | Pro forma amounts reflect (i) the filing and effectiveness of our amended and restated certificate of incorporation immediately prior to the completion of this offering, (ii) the conversion of all our outstanding shares of convertible preferred stock into an aggregate of 9,959,831 shares of our common stock and the resulting reclassification of the carrying value of the convertible preferred stock to permanent equity, in connection with the completion of this offering, (iii) the net exercise of certain outstanding warrants to purchase shares of our Series F redeemable convertible preferred stock for an aggregate of shares of common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), in connection with the completion of this offering, (iv) the adjustment of outstanding warrants to purchase shares of our redeemable convertible preferred stock into warrants to purchase shares of our common stock and the resulting reclassification of our preferred stock warrant liability to additional paid-in-capital, a component of stockholders’ deficit, in connection with the completion of this offering, and (v) the conversion of approximately $11.8 million of aggregate principal amount, plus accrued interest thereon, of convertible promissory notes which will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019. |
| (2) | Pro forma as adjusted amounts reflect the pro forma conversion adjustment described in footnote (1) above, as well as the sale of shares of our common stock in this offering at an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of the prospectus), and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
| (3) | A $1.00 increase (decrease) in the assumed initial public offering price would increase (decrease) each of cash and cash equivalents, working capital, total assets and total stockholders’ equity by $ , assuming the number of shares offered by us as stated on the cover page of this prospectus remain unchanged and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. Similarly, a one million share increase (decrease) in the number of shares offered by us, as set forth on the cover page of this prospectus, would increase (decrease) each of cash and cash equivalents, working capital, total assets and total stockholders’ equity by $ , assuming the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) remains the same, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
| (4) | We define working capital as current assets minus current liabilities. See our financial statements and related notes included elsewhere in this prospectus for further details regarding our current assets and liabilities. |
| (5) | Principal amount, less unamortized discounts and issuance costs, plus embedded derivative liability. See Note 7 to our unaudited interim condensed financial statements included elsewhere in this prospectus for additional information on the accounting treatment of the convertible promissory notes. |
10
Investing in our common stock is speculative and involves a high degree of risk. Before investing in our common stock, you should consider carefully the risks described below, together with the other information contained in this prospectus, including our financial statements and the related notes appearing at the end of this prospectus. If any of the following risks occur, our business, financial condition, results of operations and future growth prospects could be materially and adversely affected. In these circumstances, the market price of our common stock could decline, and you may lose all or part of your investment. This prospectus also contains forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those anticipated in the forward-looking statements as a result of a number of factors, including the risks described below. See “Special Note Regarding Forward-Looking Statements.”
Risks Related to Our Financial Condition
Our reliance upon a small number of third-party payors for a significant portion of our revenue may materially adversely affect our financial condition and results of operations.
We receive a substantial portion of our revenue from a small number of third-party payors, primarily Medicare and United Healthcare. Our revenue for our test reports provided for patients covered by Medicare and United Healthcare as a percentage of total revenue, was 9% and 11%, respectively, for the year ended December 31, 2017, and 36% and 12%, respectively, for the year ended December 31, 2018. In addition, our current accounts receivable balances for Medicare and United Healthcare, as a percentage of our total current accounts receivable, were 0% and 10%, respectively, as of December 31, 2017, and 54% and 7%, respectively, as of December 31, 2018. Our long-term accounts receivable balances for Medicare and United Healthcare, as a percentage of our total long-term accounts receivable, were 0% and 15%, respectively, as of December 31, 2017, and 0% and 15%, respectively, as of December 31, 2018. If our largest current payors were to significantly reduce, or cease to pay, the amount they reimburse for our products, or if they do not reach favorable coverage and reimbursement decisions for our products, or attempt to recover amounts they had already paid, it could have a material adverse effect on our business, financial condition and results of operations and cause significant fluctuations in our results of operations.
Due to how we recognize revenue, our quarterly revenues may not reflect our underlying business.
We have concluded that our contracts include variable consideration because the amounts paid by Medicare or commercial health insurance carriers may be paid at less than our standard rates or not paid at all, with such differences considered implicit price concessions. Variable consideration attributable to these price concessions is measured at the expected value using the “most likely amount” method under Accounting Standards Update No. 2014-09, Revenue from Contracts with Customers (Topic 606), or ASC 606. The amounts are determined by historical average collection rates by test type and payor category taking into consideration the range of possible outcomes, the predictive value of our past experiences, the time period of when uncertainties expect to be resolved and the amount of consideration that is susceptible to factors outside of our influence, such as the judgment and actions of third parties. Such variable consideration is included in the transaction price only to the extent it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainties with respect to the amount are resolved. Variable consideration may be constrained and excluded from the transaction price in situations where there is no contractually agreed upon reimbursement coverage or in the absence of a predictable pattern and history of collectability with a payor. Variable consideration for Medicare claims is deemed to be fully constrained when the payment of such claims is subject to approval by an Administrative Law Judge, or ALJ, at an appeal hearing, due to factors outside our influence (i.e., judgment or actions of third parties) and the uncertainty of the amount to be received is not expected to be resolved for a long period of time. Variable consideration is evaluated each reporting period and adjustments are recorded as increases or decreases in revenues. As a result of the timing and amount of adjustments for variable consideration, our operating results and comparisons of such results on a period-to-period basis may be difficult to understand and may not be meaningful. In addition, these fluctuations in revenue may make it difficult for us, for research analysts and for investors to accurately forecast our revenue and operating results. If our revenue or operating results fall below expectations, the price of our common stock would likely decline.
We have incurred significant losses since inception, and we may never achieve or sustain profitability.
Since our inception, we have had a history of net losses. As of March 31, 2019, we had a cash balance of approximately $16.2 million and an accumulated deficit of approximately $58.8 million. We cannot predict if we
11
will achieve sustained profitability in the near future or at all. We expect that our losses will continue for the foreseeable future as we plan to invest significant additional funds toward the expansion of our commercial organization, the conduct of additional clinical utility and validity studies to support adoption of our products and the development or acquisition of additional products. Our auditors have issued a going concern opinion on our financial statements as of and for the years ended December 31, 2017 and 2018, expressing substantial doubt about our ability to continue as a going concern. As a public company, we will also incur significant legal, accounting and other expenses that we did not incur as a private company. These increased expenses will make it harder for us to achieve and sustain future profitability. We may also incur significant losses in the future for a number of reasons, many of which are beyond our control, including the other risks described in this prospectus, adoption of our products, coverage of and reimbursement rates for our products from third-party payors, and future research and development activities. Our failure to achieve and sustain profitability in the future could cause the market price of our common stock to decline.
We are an early, commercial-stage company and have a limited operating history, which may make it difficult to evaluate our current business and predict our future performance.
We are an early commercial-stage company and have a limited operating history. Our limited operating history may make it difficult to evaluate our current business and this makes predictions about our future success or viability subject to significant uncertainty. In particular, we intend to use a portion of the net proceeds from this offering to increase our headcount, including through the expansion of our sales and marketing and research and development teams, which will increase our operating costs in a manner not historically reflected in our financial statements. In combination with our other anticipated increased operating expenses in connection with becoming a public company, these anticipated changes in our operating expenses may make it difficult to evaluate our current business, assess our future performance relative to prior performance and accurately predict our future performance.
We will continue to encounter risks and difficulties frequently experienced by early commercial-stage companies, including those associated with increasing the size of our organization and the prioritization of our commercial, research and business development activities. If we do not address these risks successfully, our business could suffer.
The terms of our credit facility place restrictions on our operating and financial flexibility, and failure to comply with covenants or to satisfy certain conditions of the agreement governing the credit facility may result in acceleration of our repayment obligations and foreclosure on our pledged assets, which could significantly harm our liquidity, financial condition, operating results, business and prospects and cause the price of our securities to decline.
Our November 2018 loan and security agreement, which we amended in June 2019, or the 2018 LSA, with Oxford Finance LLC, or Oxford, and Silicon Valley Bank, or SVB, is secured by a lien covering substantially all of our assets, excluding intellectual property. The 2018 LSA provides for a five-year $25.0 million term-loan facility, of which $25.0 million has been disbursed to us as of the date of this prospectus.
The 2018 LSA requires us to achieve certain revenue levels tested monthly on a trailing three-month basis. Although we were in compliance with this covenant as of the most recently tested month prior to the date of this prospectus, there can be no assurance of our ability to maintain compliance with the revenue covenant as of any future date. The 2018 LSA also requires us to comply with a number of other covenants (affirmative and negative), including restrictive covenants that limit our ability to: incur additional indebtedness; encumber the collateral securing the loan; acquire, own or make investments; repurchase or redeem any class of stock or other equity interest; declare or pay any cash dividend or make a cash distribution on any class of stock or other equity interest; transfer a material portion of our assets; acquire other businesses; and merge or consolidate with or into any other organization or otherwise suffer a change in control, in each case subject to exceptions.
In addition to other specified events of default, and subject to limited exceptions, the lenders could declare an event of default upon the occurrence of any event that they interpret as having a material impairment in their lien on the collateral under the agreement, a material adverse change in our business, operations or condition (financial or otherwise) or a material impairment in the prospect of repayment of our obligations under the agreement. If we default under the credit facility, the lenders may accelerate all of our repayment obligations and, if we are unable to access funds to meet those obligations or to renegotiate our agreement, the lenders could take control of our pledged assets and we would have to immediately cease operations. During the continuance
12
of an event of default, the then-applicable interest rate on the then-outstanding principal balance will increase by 5.0%. Upon an event of default, the lenders could also require us to repay the loan immediately, together with a prepayment charge of up to 2.5% of the then-outstanding principal balance, together with other fees. If we were to renegotiate the agreement under such circumstances, the terms may be significantly less favorable to us. If we were liquidated, the lenders’ right to repayment would be senior to the rights of our stockholders to receive any proceeds from the liquidation. Any declaration by the lenders of an event of default could significantly harm our liquidity, financial condition, operating results, business, and prospects and cause the price of our securities to decline.
We may incur additional indebtedness in the future. The debt instruments governing such indebtedness may contain provisions that are as, or more, restrictive than the provisions governing our existing indebtedness. If we are unable to repay, refinance or restructure our indebtedness when payment is due, the lenders could proceed against the collateral or force us into bankruptcy or liquidation.
The audit report of our independent registered public accounting firm expresses substantial doubt about our ability to continue as a going concern.
The audit report from our independent registered public accounting firm expresses substantial doubt that we can continue as an ongoing business due to uncertainties about our ability to comply with certain debt covenants under our long-term debt that is required to finance operations and our future financial statements may include a similar qualification about our ability to continue as a going concern. Our audited financial statements were prepared assuming that we will continue as a going concern and do not include any adjustments that may result from the outcome of this uncertainty.
If we are unable to meet the applicable debt covenants, the lenders could accelerate all of our repayment obligations under the 2018 LSA and we would need to seek additional or alternate financing or modify our operational plans. If we seek additional financing to fund our business activities in the future and there remains substantial doubt about our ability to continue as a going concern, investors or other financing sources may be unwilling to provide additional funding to us on commercially reasonable terms or at all.
We have identified material weaknesses in our internal control over financial reporting. If our internal control over financial reporting is not effective, we may not be able to accurately report our financial results or file our periodic reports in a timely manner, which may cause adverse effects on our business and may cause investors to lose confidence in our reported financial information and may lead to a decline in our stock price.
Effective internal control over financial reporting is necessary for us to provide reliable financial reports in a timely manner. In connection with the audits of our financial statements for the years ended December 31, 2017 and 2018, we concluded that there were material weaknesses in our internal control over financial reporting. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the annual or interim financial statements will not be prevented or detected on a timely basis.
These material weaknesses related to a lack of (i) appropriately designed and implemented controls over the review and approval of manual journal entries and the related supporting journal entry calculations, (ii) personnel with appropriate knowledge, experience and training commensurate with accounting and reporting requirements and (iii) appropriately designed and implemented controls to evaluate variable consideration and the related constraint in accordance with ASC 606, and resulted in certain material corrections to the financial statements.
In an attempt to remediate these weaknesses, we have hired an SEC compliance and technical accounting director and plan to hire additional finance and accounting personnel to augment our accounting staff and to provide more resources for complex accounting matters and financial reporting. However, we cannot assure you that these efforts will remediate our material weaknesses in a timely manner, or at all.
If we are unable to successfully remediate our material weaknesses or identify any future significant deficiencies or material weaknesses, the accuracy and timing of our financial reporting may be adversely affected, a material misstatement in our financial statements could occur, we may be unable to maintain compliance with securities law requirements regarding timely filing of periodic reports, which may adversely affect our business and our stock price may decline as a result.
13
In addition, even if we remediate our material weaknesses, following the completion of this offering, we will be required to expend significant time and resources to further improve our internal controls over financial reporting, including by further expanding our finance and accounting staff to meet the demands that will be placed upon us as a public company, including the requirements of the Sarbanes-Oxley Act. If we fail to adequately staff our accounting and finance function to remediate our material weaknesses, or fail to maintain adequate internal control over financial reporting, any new or recurring material weaknesses could prevent our management from concluding our internal control over financial reporting is effective and impair our ability to prevent material misstatements in our financial statements, which could cause our business to suffer.
Changes in financial accounting standards or practices may cause adverse, unexpected financial reporting fluctuations and affect our reported operating results.
U.S. GAAP is subject to interpretation by the Financial Accounting Standards Board, the Securities and Exchange Commission, or the SEC, and various bodies formed to promulgate and interpret appropriate accounting principles. A change in accounting standards or practices can have a significant effect on our reported results and may even affect our reporting of transactions completed before the change is effective. For example, as described in “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Revenue Recognition” and in Note 2 to our audited financial statements, we recently adopted the revenue recognition standard under ASC 606 which superseded previous revenue recognition guidance applicable to us. New accounting pronouncements and varying interpretations of accounting pronouncements have occurred and may occur in the future. Changes to existing rules or the questioning of current practices may adversely affect our reported financial results or the way we conduct our business.
Our quarterly and annual operating results and cash flows may fluctuate in the future, which could cause the market price of our stock to decline substantially.
Numerous factors, many of which are outside our control, may cause or contribute to significant fluctuations in our quarterly and annual operating results. These fluctuations may make financial planning and forecasting uncertain. In addition, these fluctuations may result in unanticipated decreases in our available cash, which could negatively affect our business and prospects. In addition, one or more of such factors may cause our revenue or operating expenses in one period to be disproportionately higher or lower relative to the others. As a result, comparing our operating results on a period-to-period basis may be difficult to understand and may not be meaningful. You should not rely on our past results as indicative of our future performance.
In addition, a significant portion of our operating expense is relatively fixed in nature, and planned expenditures are based in part on expectations regarding future revenue. Accordingly, unexpected revenue shortfalls could decrease our gross margins and cause significant changes in our operating results from quarter to quarter. If this occurs, the trading price of our stock could fall substantially.
This variability and unpredictability caused by factors such as those described above could also result in our failing to meet the expectations of industry or financial analysts or investors for any period. If our revenue or operating results fall below the expectations of analysts or investors or below any guidance we may provide, or if the guidance we provide is below the expectations of analysts or investors, the price of our common stock could decline substantially. Such a stock price decline could occur even when we have met any previously publicly stated guidance we may provide.
We may need to raise additional capital to fund our existing operations, commercialize new products or expand our operations.
We believe the net proceeds from this offering, together with our existing cash and cash equivalents and anticipated cash generated from sales of our products, will be sufficient to fund our operating expenses through at least the next 24 months. If our available cash balances, net proceeds from this offering and anticipated cash generated from sales of our products are insufficient to satisfy our liquidity requirements including because of lower demand for our products, lower than currently expected rates of reimbursement from third-party payors or other risks described in this prospectus, we may finance our cash needs through a combination of equity offerings, debt financings, collaborations, strategic alliances and marketing, distribution or licensing arrangements. We do not currently have any committed external source of funds. In addition, we may seek additional capital due to favorable market conditions or strategic considerations, even if we believe we have sufficient funds for our current or future operating plans.
14
We may consider raising additional capital in the future to expand our business, to pursue strategic investments, to take advantage of financing opportunities or for other reasons, including to:
| • | increase our sales and marketing efforts for DecisionDx-Melanoma and address competitive developments; |
| • | fund ongoing development of our pipeline products, including for SCC and suspicious pigmented lesions, in addition to other programs in development; |
| • | expand our laboratory testing facility and related testing capacity; |
| • | expand our technologies into other types of skin cancer management and detection products; |
| • | acquire, license or invest in technologies; |
| • | acquire or invest in complementary businesses or assets; and |
| • | finance capital expenditures and general and administrative expenses. |
Our present and future funding requirements will depend on many factors, including:
| • | our ability to achieve revenue growth; |
| • | our rate of progress in establishing payor coverage and reimbursement arrangements with third-party payors; |
| • | our rate of progress in, and cost of the sales, marketing, coverage and reimbursement activities associated with, establishing adoption of DecisionDx-Melanoma, among our other products; |
| • | the cost of expanding our laboratory operations and offerings, including our sales, marketing, coverage and reimbursement efforts; |
| • | our rate of progress in, and cost of research and development activities associated with, diagnostic products in research and early development; |
| • | the potential cost of, and delays in, the development of new products as a result of changes in regulatory oversight applicable to our products; and |
| • | the effect of competing technological and market developments. |
To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect your rights as a common stockholder. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making acquisitions or capital expenditures or declaring dividends.
If we raise additional funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or products, or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings or other arrangements when needed, we may be required to delay, limit, reduce or terminate our commercialization, research and development efforts or grant rights to third parties to market and/or develop products that we would otherwise prefer to market and develop ourselves.
Risks Related to Our Business
Our revenue currently depends primarily on sales of DecisionDx-Melanoma, and we will need to generate sufficient revenue from this and other products to grow our business.
Most of our revenue in 2017 and 2018 was derived from the sale of our lead product, DecisionDx-Melanoma. While we also derive revenue from DecisionDx-UM, we expect that the majority of our revenue for the foreseeable future will be derived from sales of DecisionDx-Melanoma. Further, we believe that our long-term commercial success will depend on our ability to develop and market additional products, such as our pipeline products for SCC and suspicious pigmented lesions. Our ability to derive revenue from DecisionDx-Melanoma, DecisionDx-UM and any future products that we commercialize is uncertain and depends on favorable coverage and reimbursement policies from government payors, like Medicare, and from private payors, like insurance
15
companies. Without positive coverage policies, our products may not be reimbursed and we may not be able to recognize revenue. If we are unable to increase sales and expand coverage and reimbursement for DecisionDx-Melanoma, develop and commercialize other products, and successfully obtain coverage and adequate reimbursement for such products, our revenue and our ability to achieve and sustain profitability would be impaired, and the market price of our stock could decline substantially.
Billing for our products is complex and requires substantial time and resources to collect payment.
Billing for clinical laboratory testing services is complex, time-consuming and expensive. Depending on the billing arrangement and applicable law, we bill various payors, including Medicare, Medicaid, private insurance companies, private healthcare institutions, and patients, all of which have different billing requirements. We generally bill third-party payors for products and pursue reimbursement on a case-by-case basis where pricing contracts are not in place. To the extent laws or contracts require us to bill patient co-payments or co-insurance, we must also comply with these requirements. We may also face increased risk in our collection efforts, including potential write-offs of accounts receivable and long collection cycles, which could adversely affect our business, results of operations and financial condition.
Several factors make the billing process complex, including:
| • | differences between the billing rates and reimbursement rates for our products; |
| • | compliance with complex federal and state regulations related to billing government healthcare programs, including Medicare, Medicaid and TRICARE; |
| • | risk of government audits related to billing; |
| • | disputes among payors as to which party is responsible for payment; |
| • | differences in coverage and information and billing requirements among payors, including the need for prior authorization and/or advanced notification; |
| • | the effect of patient co-payments or co-insurance and our ability to collect such payments from patients; |
| • | changes to billing codes used for our products; |
| • | changes to requirements related to our current or future clinical trials, including our registry studies, which can affect eligibility for payment; |
| • | ongoing monitoring provisions of LCDs for our products, which can affect the circumstances under which a claim would be considered medically necessary; |
| • | incorrect or missing billing information; and |
| • | the resources required to manage the billing and claims appeals process. |
We use standard industry billing codes, known as CPT codes, to bill for our products. If these codes were to change, there is a risk of an error being made in the claim adjudication process. Such errors can occur with claims submission, third-party transmission or in the processing of the claim by the payor. Claim adjudication errors may result in a delay in payment processing or a reduction in the amount of the payment we receive.
As we introduce new products, we may need to add new codes to our billing process as well as our financial reporting systems. Failure or delays in effecting these changes in external billing and internal systems and processes could negatively affect our collection rates, revenue and cost of collecting.
Additionally, our billing activities require us to implement compliance procedures and oversight, train and monitor our employees, and undertake internal audits to evaluate compliance with applicable laws and regulations as well as internal compliance policies and procedures. When payors deny our claims, we may challenge the reason, low payment amount or payment denials. Payors also conduct external audits to evaluate payments, which add further complexity to the billing process. If the payor makes an overpayment determination, there is a risk that we may be required to return all or some portion of prior payments we have received. Additionally, the Patient Protection and Affordable Care Act requires providers and suppliers to report and return any overpayments received from government payors under the Medicare and Medicaid programs within 60 days of identification. Failure to identify and return such overpayments exposes the provider or supplier to liability
16
under federal false claims laws. These billing complexities, and the related uncertainty in obtaining payment for our products, could negatively affect our revenue and cash flow, our ability to achieve profitability, and the consistency and comparability of our results of operations.
We rely on third parties for tumor sample collection, preparation and delivery. Any defects in sample collection or preparation by such third parties and any delays in delivery of such samples could cause errors in our test reports and delay our ability to deliver test reports in a timely manner, which could significantly harm our business.
The tumor tissue samples that we test are biopsied, preserved, prepared and delivered to us by third parties, including dermatopathologists and laboratory facilities. As such, we rely on these third parties to prepare, label and deliver the tissue samples that we test in compliance with applicable laws and guidelines, and in a timely manner. Therefore, the accuracy and correctness of the test reports that we deliver are dependent on proper chain of custody and appropriate methods of sample collection or preparation utilized by these third parties, and our ability to timely deliver reports is dependent upon the ability of these third parties to provide these samples to us in a timely manner. Any errors in any part of the sample collection or preparation process could cause us to deliver incorrect test reports, potentially resulting in harm to patients whose physicians implement a change in treatment decisions based upon our test report. If we are unable to timely deliver test reports, physicians may be less likely to recommend and order our products. The occurrence of any of the foregoing could significantly harm our reputation and our results of operations, causing significant harm to our business.
We rely on our database of tumor samples for the development and improvement of our products. Depletion or loss of our tumor samples could significantly harm our business.
The development and validation of accurate products is a complex process that requires access to tumor tissue specimens and long-term outcomes data. Our research and development efforts to improve our existing products and develop new products may require the depletion of our existing database of tumor samples. If our tumor samples are lost or destroyed, or substantially depleted before we are able to generate meaningful data, we may be unable to improve our existing products, continue the development of pipeline products or validate product candidates. While we have historically been able to create and maintain a large sample bank to expand the clinical use of our products and develop new products, we may be unable to do so in the future. If we were unable to maintain or replenish our sample bank, we may be unable to improve our products or develop new products.
If our sole laboratory facility becomes damaged or inoperable or we are required to vacate our existing facility, our ability to conduct our laboratory analysis and pursue our research and development efforts may be jeopardized.
We currently perform all of our testing and store our database of tumor samples at a single laboratory facility in Phoenix, Arizona. Our facility and equipment could be harmed or rendered inoperable by natural or man-made disasters, including war, fire, earthquake, power loss, communications failure, terrorism, burglary or other events, which may make it difficult or impossible for us to perform our testing services for some period of time or to receive and store samples. The inability to perform tests or to reduce the backlog of sample analysis that could develop if our facility becomes inoperable, for even a short period of time, may result in the loss of revenue, loss of customers or harm to our reputation, and we may be unable to regain that revenue, those customers or repair our reputation in the future. Furthermore, integral parties in our supply chain are operating from single sites, increasing their vulnerability to natural disasters or other sudden, unforeseen and severe adverse events.
In addition, the loss of our tumor samples due to such events could limit or prevent our ability to conduct research and development analysis on existing tests as well as tests in active pipeline development.
While we have a business continuity plan in place, our facility and the equipment we use to perform our testing and research and development could be unavailable or costly and time-consuming to repair or replace. It would be difficult, time-consuming and expensive to rebuild our facility, to locate and qualify a new facility, replace certain pieces of equipment or license or transfer our proprietary technology to a third-party, particularly in light of licensure and accreditation requirements. Even in the unlikely event that we are able to find a third party with such qualifications to enable us to resume our operations, we may be unable to negotiate commercially reasonable terms.
17
We carry insurance for damage to our property and the disruption of our business, but this insurance may not cover all of the risks associated with damage or disruption to our business, may not provide coverage in amounts sufficient to cover our potential losses and may not continue to be available to us on acceptable terms, if at all.
Our current or future products may not achieve or maintain significant commercial market acceptance.
We believe our commercial success is dependent upon our ability to continue to successfully market and sell our products, to continue to expand our current relationships and develop new relationships with healthcare providers, to expand and maintain coverage for our products, and to develop and commercialize new products. Our ability to achieve and maintain commercial market acceptance of our existing and future products will depend on a number of factors, including:
| • | our ability to increase awareness of our products through successful clinical utility and validity studies; |
| • | the rate of adoption of our products by physicians and other healthcare providers; |
| • | our ability to achieve guideline inclusion for our products; |
| • | the timeliness with which we can provide our clinical reports to the ordering physician; |
| • | the timing and scope of any regulatory approval for our products, if such approvals become required, and maintaining ongoing compliance with regulatory requirements; |
| • | our ability to obtain and maintain positive coverage decisions for our products from government and commercial payors; |
| • | our ability to obtain and maintain adequate reimbursement from third-party payors, including Medicare, Medicare Advantage plans, United Healthcare and BlueCross BlueShield plans, which accounted for an aggregate of approximately 73% and 83% of our total revenue for the years ended December 31, 2017 and 2018, respectively; |
| • | the impact of our investments in research and development and commercial growth; |
| • | negative publicity regarding our or our competitors’ products resulting from scientific publications, or defects or errors in the products; and |
| • | our ability to further validate our products through clinical research and accompanying publications. |
We cannot assure you that we will be successful in addressing each of these factors or other factors that might affect the market acceptance of our products. If we are unsuccessful in achieving and maintaining market acceptance of our products, our business and results of operations will suffer.
New product development involves a lengthy and complex process, and we may be unable to develop and commercialize, or receive reimbursement for, on a timely basis, or at all, new products.
We continually seek to develop new product offerings, which requires us to devote considerable resources to research and development. For example, before we can commercialize our pipeline products for SCC and suspicious pigmented lesions, we will need to expend significant resources in order to conduct substantial research and development, including clinical utility and validity studies, and further develop and scale our laboratory processes and infrastructure to accommodate additional products.
Our product development process takes time and involves a high degree of risk, and such development efforts may fail for many reasons, including failure of the product to perform as expected, failure to successfully complete analytic and clinical validation, or failure to demonstrate the clinical utility of the product.
As we develop new products, we will have to make significant investments in research and development, marketing, selling, coverage and reimbursement activities. Typically, few research and development projects result in a commercialized product, and there can be no assurance that we will be able to successfully develop new products that can be commercialized. At any point, we may abandon development of a product or we may be required to expend considerable resources conducting research, which would adversely affect the timing for generating potential revenue from a new product and our ability to invest in other products in our pipeline. If a clinical validation study fails to demonstrate the prospectively defined endpoints of the study or if we fail to sufficiently demonstrate analytical validity or clinical utility, we might choose to abandon the development of the product, which could harm our business. In addition, competitors may develop and commercialize competing products or technologies faster than us or at a lower cost.
18
We may experience limits on our revenue if we are unable to increase and support adoption of our products by physicians and other healthcare providers.
Physicians and other healthcare providers may be unwilling to adopt our products due to their reliance on existing traditional clinical and pathology staging criteria and our ability to generate revenue from our products would be significantly impaired if we were unable to educate physicians, healthcare providers, patients and third-party payors about the benefits and advantages of our products. We will need to continue to educate physicians and pathologists about the benefits and cost-effectiveness of our products through published papers, presentations at scientific conferences, one-on-one marketing efforts by our sales force and one-on-one education by our medical affairs team. However, physicians and other healthcare providers may be reluctant to adopt our products in circumstances where our products are not incorporated into the current standard of care or practice guidelines. For example, while clinical utility of DecisionDx-Melanoma has been demonstrated in peer reviewed publications, the SLNB surgery is the most widely used staging tool for determining a cutaneous melanoma patient’s metastatic risk. Whether healthcare providers adopt DecisionDx-Melanoma as a complementary or triage diagnostic method relative to the SLNB surgery will depend on our ability to increase awareness of DecisionDx-Melanoma and its clinical validation.
In addition, our testing services are performed by our certified laboratory located in Phoenix, Arizona, under the Clinical Laboratory Improvement Amendments of 1988, or CLIA, rather than by local laboratory or pathology practices. Accordingly, it may be difficult for us to collect samples from pathologists, and pathologists may be reluctant to support our testing services.
We rely on limited or sole suppliers for some of the reagents, equipment, chips and other materials used by our products, and we may not be able to find replacements or transition to alternative suppliers.
We rely on limited or sole suppliers for certain reagents and other materials and components that we use for our products. Some of these items are unique to these suppliers and vendors. While we have developed alternate sourcing strategies for these materials and vendors, we cannot be certain whether these strategies will be effective or the alternative sources will be available when we need them. If these suppliers can no longer provide us with the materials we need, if the materials do not meet our quality specifications or are otherwise unusable, if we cannot obtain acceptable substitute materials, or if we elect to change suppliers, an interruption in laboratory operations could occur, we may not be able to deliver patient reports on a timely basis, or at all, and we may incur higher one-time switching costs. Any such interruption may significantly affect our future revenue, cause us to incur higher costs, and harm our customer relationships and reputation. In addition, in order to mitigate these risks, we maintain inventories of these supplies at higher levels than would be the case if multiple sources of supply were available. If our testing volume decreases or we switch suppliers, we may hold excess supplies with expiration dates that occur before use which would adversely affect our losses and cash flow position. As we introduce any new products, we may experience supply issues as we ramp test volume. If we should encounter delays or difficulties in securing, reconfiguring or revalidating the equipment, reagents or other materials we require for our products, our business, financial condition, results of operations and reputation could be adversely affected.
If our products do not meet the expectations of physicians and patients, our operating results, reputation and business could suffer.
Our success depends on physician and patient confidence that we can provide reliable, high-quality information that will improve treatment outcomes, lower healthcare costs and enable better patient care. We believe that patients, physicians and other healthcare providers are likely to be particularly sensitive to defects and errors in our products, including if our products fail to accurately predict risk of metastasis with high accuracy from samples, and there can be no guarantee that our products will meet their expectations. As a result, the failure of our products to perform as expected could significantly impair our operating results and our reputation, including if we become subject to legal claims arising from any defects or errors in our products or reports.
If we are unable to compete successfully, our business will suffer and we may be unable to increase or sustain our revenue or achieve profitability.
We face competition from companies and academic institutions that have either developed or may seek to develop products intended to compete with our products. Potential competitors within the broader genomics
19
profiling space based on tissue sample collection include laboratory companies such as Laboratory Corporation of America and Myriad Genetics, and other companies which have strong infrastructures capable of supporting the commercialization of diagnostic services.
In addition, competitors may develop their own versions of our solutions in countries where we do not have patents or where our intellectual property rights are not recognized and compete with us in those countries, including encouraging the use of their solutions by physicians in other countries.
Some potential competitors may have longer operating histories, larger customer bases, greater brand recognition and market penetration, substantially greater financial, technological and research and development resources and selling and marketing capabilities, and more experience dealing with third-party payors. As a result, they may be able to respond more quickly to changes in customer requirements, devote greater resources to the development, promotion and sale of their products than we do or sell their products at prices designed to win significant levels of market share. We may not be able to compete effectively against these organizations. Increased competition and cost-saving initiatives on the part of governmental entities and other third-party payors are likely to result in pricing pressures, which could harm our sales, profitability or ability to gain market share. In addition, competitors may be acquired by, receive investments from or enter into other commercial relationships with larger, well-established and well-financed companies. Certain potential competitors may be able to secure key inputs from vendors on more favorable terms, devote greater resources to marketing and promotional campaigns, adopt more aggressive pricing policies and devote substantially more resources to test development than we can. In addition, companies or governments that control access to testing through umbrella contracts or regional preferences could promote our competitors or prevent us from performing certain services. If we are unable to compete successfully against current and future competitors, our business will suffer and we may be unable to increase market acceptance and sales of our products, which could prevent us from increasing our revenue or achieving profitability and could cause our stock price to decline. As we add new tests and services, we will face many of these same competitive risks for these new tests.
The sizes of the markets for our current and future products have not been established with precision, and may be smaller than we estimate.
Our estimates of the total addressable markets for DecisionDx-Melanoma, DecisionDx-UM and our products in development are based on a number of internal and third-party estimates, including, without limitation, the annual rate of patients with the applicable form of skin cancer, the list price of our products relative to the reimbursement we expect to receive from third-party payors and the assumed prices at which we can sell our products in markets that have not been established. For example, we estimate that the total addressable market for DecisionDx-Melanoma is approximately $540 million, which is based, in part, on our review of multiple recent publications which show that diagnosis of melanoma is underreported by 30% to 40%. While we believe our assumptions and the data underlying our estimates are reasonable, these assumptions and estimates may not be correct and the conditions supporting our assumptions or estimates may change at any time, thereby reducing the predictive accuracy of these underlying factors. As a result, our estimates of the annual total addressable market for our current or future products may prove to be incorrect. If the actual number of patients who would benefit from our products, the price at which we can sell future products, or the annual total addressable market for our products is smaller than we have estimated, it may impair our sales growth and have an adverse impact on our business.
The diagnostic testing industry is subject to rapid change, which could make our current or future products obsolete.
Our industry is characterized by rapid changes, including technological and scientific breakthroughs, frequent new product introductions and enhancements and evolving industry standards, all of which could make our current products and the other products we are developing obsolete. Our future success will depend on our ability to keep pace with the evolving needs of physicians and patients on a timely and cost-effective basis and to pursue new market opportunities that develop as a result of scientific and technological advances. In recent years, there have been numerous advances in technologies relating to the diagnosis and treatment of cancer. There have also been advances in methods used to analyze very large amounts of molecular information. We must continuously enhance our existing products and develop new products to keep pace with evolving standards of care. If we do not update our products to reflect new scientific knowledge about cancer biology, information about new cancer therapies or relevant clinical trials, our products could become obsolete and sales of our current products and any new products we develop could decline or fail to grow as expected.
20
Risks Related to Government Regulation and Reimbursement
We currently have limited reimbursement coverage for our lead product, DecisionDx-Melanoma, and if third-party payors, including government and commercial payors, do not provide sufficient coverage of, or adequate reimbursement for, our products, our commercial success will be negatively affected.
Our revenue depends on achieving broad coverage and adequate reimbursement for our products from third-party payors, including both government and commercial third-party payors. If third-party payors do not provide coverage of, or do not provide adequate reimbursement for, a substantial portion of the list price of our products, we may need to seek additional payment from the patient beyond any co-payments and deductibles, which may adversely affect demand for our products. Coverage determinations by a third-party payor may depend on a number of factors, including, but not limited to, a third-party payor’s determination of whether our products are appropriate, medically necessary or cost-effective. If we are unable to provide third-party payors with sufficient evidence of the clinical utility and validity of our products, they may not provide coverage, or may provide limited coverage, which will adversely affect our revenues and our ability to succeed. To the extent that more competitors enter our markets, the availability of coverage and the reimbursement rate for our products may decrease as we encounter pricing pressure from these competitors.
Since each third-party payor makes its own decision as to whether to establish a policy to cover our products, enter into a contract with us and set the amount it will reimburse for a product, these negotiations are a time-consuming and costly process, and they do not guarantee that the third-party payor will provide coverage or adequate reimbursement for our products. In addition, the determinations by a third-party payor whether to cover our products and the amount it will reimburse for them are often made on an indication-by-indication basis. In cases where there is no coverage policy or we do not have a contracted rate for reimbursement as a participating provider, the patient is typically responsible for a greater share of the cost of the product, which may result in further delay of our revenue, increase our collection costs or decrease the likelihood of collection.
Our claims for reimbursement from third-party payors may be denied upon submission, and we may need to take additional steps to receive payment, such as appealing the denials. Such appeals and other processes are time-consuming and expensive, and may not result in payment. Third-party payors may perform audits of historically paid claims and attempt to recoup funds years after the funds were initially distributed if the third-party payors believe the funds were paid in error or determine that our products were medically unnecessary. If a third-party payor audits our claims and issues a negative audit finding, and we are not able to overturn the audit findings through appeal, the recoupment may result in a material adverse effect on our revenue. Additionally, in some cases commercial third-party payors for whom we are not a participating provider may elect at any time to review claims previously paid and determine the amount they paid was too much. In these situations, the third-party payor will typically notify us of their decision and then offset whatever amount they determine they overpaid against amounts they owe us on current claims. We cannot predict when, or how often, a third-party payor might engage in these reviews and we may not be able to dispute these retroactive adjustments. We adopted the new revenue recognition guidance under ASC 606 on January 1, 2018 using the full retrospective method and adjusted the comparative reporting period for the year ended December 31, 2017. Under ASC 606, we recognize revenue at the amount we expect to be entitled, subject to a constraint for variable consideration, in the period in which our tests are delivered to the treating physician. We have determined that our contracts contain variable consideration under ASC 606 because the amounts paid by third-party payors may be paid at less than our standard rates or not paid at all, with such differences considered implicit price concessions. Variable consideration is recognized only to the extent it is probable that a significant reversal of revenue will not occur in future periods when the uncertainties are resolved. We consider variable consideration to be fully constrained (and therefore not recognized) for Medicare claims when the payment of such claims is subject to approval by an ALJ at an appeal hearing, due to the level of uncertainty and timing of the outcome. Variable consideration is evaluated each reporting period and adjustments are recorded as increases or decreases in revenues. Due to the outcome of ALJ hearings, potential future changes in insurance coverage policies, contractual rates and other trends in the reimbursement of our tests, our revenues may fluctuate significantly from period to period.
Although we are an in-network participating provider with some commercial third-party payors, including several Blue Cross Blue Shield plans, and certain large, national commercial third-party payors, including Aetna, other commercial third-party payors have issued non-coverage policies that currently categorize DecisionDx-UM and
21
DecisionDx-Melanoma as experimental or investigational. If we are not successful in obtaining coverage from third-party payors, in reversing existing non-coverage policies, or if other third-party payors issue similar non-coverage policies, this could have a material adverse effect on our business and operations.
Palmetto GBA, or Palmetto, the Medicine Administrative Contractor, or MAC, responsible for administering MolDX, the program that assesses molecular diagnostic technologies, issued a final LCD for DecisionDx-Melanoma, which became effective on December 3, 2018. This LCD provides for coverage of DecisionDx-Melanoma for certain sentinel lymph node biopsy, or SLNB, eligible patients with cutaneous melanoma tumors with clinically negative sentinel node basins who are being considered for SLNB to determine eligibility for adjuvant therapy. Similarly, Palmetto issued a final LCD for DecisionDx-UM effective July 10, 2017. This LCD provides for coverage of DecisionDx-UM to determine metastatic risk in connection with the management of a patient’s newly diagnosed uveal melanoma and to guide surveillance and referral to medical oncology for those patients. We worked with Palmetto to obtain these positive coverage decisions through the submission of a detailed dossier of analytical and clinical data to substantiate that the tests meet Medicare’s medical necessity requirements. Per their joint operating agreement, Noridian Healthcare Solutions LLC, or Noridian, the MAC responsible for administering claims for laboratory services performed in Arizona, has adopted the same coverage policy as Palmetto for DecisionDx-UM and DecisionDx-Melanoma. This coverage process is lengthy, time-consuming, has changed over time, may change in the future and requires significant dedication of resources, and as we develop new products, we may be unsuccessful in receiving LCD determinations for those products or in maintaining our current LCDs. On a periodic basis, the Centers for Medicare & Medicaid Services, or CMS, requests bids for its MAC services, and MAC jurisdictions have changed in the past. A change in our MAC, or future changes in the MolDx program, the elimination of the program, or a change in the administrator of that program, may affect our ability to obtain Medicare coverage and reimbursement for products for which we have coverage, for products for which we do not yet have coverage, or for any products we may launch in the future, or delay payments for our tests.
Under Medicare, payment for products like ours is generally made under the Clinical Laboratory Fee Schedule, or CLFS, with payment amounts assigned to specific procedure billing codes. In April 2014, Congress passed the Protecting Access to Medicare Act of 2014, or PAMA, which included substantial changes to the way in which clinical laboratory services are paid under Medicare. Under PAMA, certain laboratories were required to report to CMS, beginning in 2017 and every three years thereafter (or annually for advanced diagnostic laboratory tests, or ADLTs), commercial third-party payor payment rates and volumes for each test they perform. CMS uses this data to calculate a weighted median payment rate for each test, which will be used to establish revised Medicare CLFS reimbursement rates for the test. Laboratories that fail to report the required payment information may be subject to substantial civil monetary penalties. We bill Medicare for our products, and therefore we are subject to reporting requirements under PAMA.
On May 17, 2019, CMS determined that DecisionDx-UM meets the criteria for “existing ADLT” status. This means that beginning in 2021, the DecisionDx-UM Medicare rate will be set annually based upon the median private payor rate for the first half of the preceding calendar year. Specifically, the median private payor rate from January 1 to June 30, 2019 will be used to set the Medicare rate for the calendar year 2021. From May 17, 2019 through December 31, 2020, our rate will be set by Noridian, our local MAC. Also, on May 17, 2019, CMS determined that DecisionDx-Melanoma meets the criteria for “new ADLT” status. This means that from July 1, 2019 through March 31, 2020 the Medicare reimbursement rate will equal the initial list price of $7,193.00. The rate for April 1, 2020 through December 31, 2020 will be calculated based upon the median private payor rate from July 1, 2019 to November 30, 2019. Accordingly, beginning in 2021, the rate will be set annually based upon the median private payor rate for the first half of the preceding calendar year. If CMS determines the list charge amount for DecisionDx-Melanoma was greater than 130% of the weighted median of private payor rates, CMS will recoup from us the difference between the actual list charge and 130% of the weighted median. If we are unable to obtain and maintain adequate reimbursement rates from commercial third-party payors, this may adversely affect our Medicare rate. It is unclear what impact new pricing structures, such as those adopted under PAMA, may have on our business, financial condition, results of operations or cash flows.
The U.S. federal government continues to show significant interest in pursuing health care reform and reducing health care costs. Similarly, commercial third-party payors may seek to reduce costs by limiting coverage or
22
reducing reimbursement for our products. Any government-adopted reform measures or changes to commercial third-party payor coverage and reimbursement policies could cause significant pressure on the pricing of, and reimbursement for, health care products and services, including our products, which could decrease demand for our products, and adversely affect our sales and revenue.
In addition, some third-party payors have implemented, or are in the process of implementing, laboratory benefit management programs, often using third-party benefit managers to manage these programs. The stated goals of these programs are to help improve the quality of outpatient laboratory services, support evidence-based guidelines for patient care and lower costs. The impact on laboratories, such as ours, of active laboratory benefit management by third parties is unclear, and we expect that it could have a negative impact on our revenue in the short term. It is possible that third-party payors will resist reimbursement for the products that we offer, in favor of less expensive products, may require pre-approval for our products or may impose additional pricing pressure on and substantial administrative burden for reimbursement for our products.
We expect to continue to focus substantial resources on increasing coverage and reimbursement for our current products and any future products we may develop. We believe it may take several years to achieve broad coverage and adequate contracted reimbursement with a majority of third-party payors for our products. However, we cannot predict whether, under what circumstances, or at what payment levels third-party payors will cover and reimburse our products. If we fail to establish and maintain broad adoption of, and coverage and reimbursement for, our products, our ability to generate revenue could be harmed and our future prospects and our business could suffer.
Our products are currently marketed as laboratory developed tests, and any changes in regulations or the U.S. Food and Drug Administration’s enforcement discretion for laboratory developed tests, or violations of regulations by us, could adversely affect our business, prospects, results of operations or financial condition.
The diagnostics industry is highly regulated, and we cannot assure you that the regulatory environment in which we operate will not change significantly and adversely in the future. In many instances, there are no significant regulatory or judicial interpretations of these laws and regulations. Although the U.S. Food and Drug Administration, or FDA has statutory authority to assure that medical devices are safe and effective for their intended uses, the FDA has generally exercised its enforcement discretion and not enforced applicable regulations with respect to in vitro diagnostics that are designed, manufactured and used within a single laboratory. These tests are referred to as laboratory developed tests, or LDTs. We currently market our products as LDTs.
The FDA has adopted a policy of enforcement discretion with respect to LDTs whereby the FDA does not actively require premarket review of LDTs or otherwise impose its requirements applicable to other medical devices on LDTs. However, the FDA has stated its intention to modify its enforcement discretion policy with respect to LDTs. The FDA could ultimately modify its current approach to LDTs in a way that would subject our products marketed as LDTs to the enforcement of additional regulatory requirements. Moreover, legislative measures have recently been proposed in Congress that, if ultimately enacted, could provide the FDA with additional authority to require premarket review of and regulate LDTs. If and when such changes to the regulatory framework occur, we could for the first time be subject to enforcement of regulatory requirements as a device manufacturer such as registration and listing requirements, medical device reporting requirements and the requirements of the FDA’s Quality System Regulation. We may be required to conduct additional clinical trials prior to continuing to sell our existing products or launching any other products we may develop. This may increase the cost of conducting, or otherwise harm, our business.
Moreover, even if the FDA does not modify its policy of enforcement discretion, the FDA may disagree that we are marketing our LDTs within the scope of its policy of enforcement discretion and may impose significant regulatory requirements. While we believe that we are currently in material compliance with applicable laws and regulations as historically enforced by the FDA, we cannot assure you that the FDA will agree with our determination. A determination that we have violated these laws and regulations, or a public announcement that we are being investigated for possible violations, could adversely affect our business, prospects, results of operations or financial condition.
If the FDA begins to actively regulate our diagnostic products, we may be required to obtain premarket clearance under Section 510(k) of the U.S. Federal Food, Drug and Cosmetic Act, or FDCA, or a premarket approval, or PMA. The process for submitting a 510(k) premarket notification and receiving FDA clearance usually takes from three to 12 months, but it can take significantly longer and clearance is never guaranteed. The process for
23
submitting and obtaining FDA approval of a PMA is much more costly, lengthy and uncertain. It generally takes from one to three years or even longer, and approval is not guaranteed. PMA approval typically requires extensive clinical data and can be significantly longer, more expensive and more uncertain than the 510(k) clearance process. Despite the time, effort and expense expended, there can be no assurance that a particular device ultimately will be cleared or approved by the FDA through either the 510(k) clearance process or the PMA process on a timely basis, or at all. Moreover, there can be no assurance that any cleared or approved labeling claims will be consistent with our current claims or adequate to support continued adoption of and reimbursement for our products. If premarket review is required for some or all of our products, the FDA may require that we stop selling our products pending clearance or approval, which would negatively impact our business. Even if our products are allowed to remain on the market prior to clearance or approval, demand or reimbursement for our products may decline if there is uncertainty about our products, if we are required to label our products as investigational by the FDA, or if the FDA limits the labeling claims we are permitted to make for our products. As a result, we could experience significantly increased development costs and a delay in generating additional revenue from our products, or from other products now in development.
If the FDA imposes significant changes to the regulation of LDTs it could reduce our revenues or increase our costs and adversely affect our business, prospects, results of operations or financial condition.
We conduct business in a heavily regulated industry, and failure to comply with federal, state and foreign laboratory licensing requirements and the applicable requirements of the FDA or any other regulatory authority, could cause us to lose the ability to perform our tests, experience disruptions to our business, or become subject to administrative or judicial sanctions.
The diagnostics industry is highly regulated, and the laws and regulations governing the marketing of diagnostic tests are extremely complex. Areas of the regulatory environment that may affect our ability to conduct business include, without limitation:
| • | federal and state laws applicable to test ordering, documentation of tests ordered, billing practices and claims payment and/or regulatory agencies enforcing those laws and regulations; |
| • | federal and state fraud and abuse laws; |
| • | federal and state laboratory anti-mark-up laws; |
| • | coverage and reimbursement levels by Medicare, Medicaid, other governmental payors and private insurers; |
| • | restrictions on coverage of and reimbursement for tests; |
| • | federal and state laws governing laboratory testing, including CLIA, and state licensing laws; |
| • | federal and state laws and enforcement policies governing the development, use and distribution of diagnostic medical devices, including LDTs; |
| • | federal, state and local laws governing the handling and disposal of medical and hazardous waste; |
| • | federal and state Occupational Safety and Health Administration rules and regulations; and |
| • | the Health Insurance Portability and Accountability Act of 1996, or HIPAA, and similar state data privacy laws. |
In particular, the FDCA defines a medical device to include any instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including a component, part, or accessory, intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals. Our products are considered by the FDA to be subject to regulation as medical devices, and marketed under FDA’s policy of enforcement discretion for LDTs. Among other things, pursuant to the FDCA and its implementing regulations, the FDA regulates the research, testing, manufacturing, safety, labeling, storage, recordkeeping, premarket clearance or approval, marketing and promotion, and sales and distribution of medical devices in the United States to ensure that medical products distributed domestically are safe and effective for their intended uses. In addition, the FDA regulates the import and export of medical devices manufactured between the United States and international markets.
24
We are also subject to CLIA, a federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for the diagnosis, prevention or treatment of disease. CLIA regulations establish specific standards with respect to personnel qualifications, facility administration, proficiency testing, quality control, quality assurance and inspections. Any testing subject to CLIA regulation must be performed in a CLIA certified or accredited lab. CLIA certification or accreditation is also required in order for us to be eligible to bill state and federal healthcare programs, as well as commercial third-party payors, for our products. We have a current CLIA accreditation under the College of American Pathologists, or CAP, program to conduct our tests at our clinical reference laboratory in Phoenix, Arizona. To maintain our CLIA accreditation, we have elected to be subject to survey and inspection every two years by CAP. Moreover, CLIA inspectors may make random inspections of our laboratory from time to time.
In addition, certain states require our laboratory to be licensed in such states in order to test specimens from those states. Accordingly, our laboratory is also licensed by California, Maryland, New York, Pennsylvania and Rhode Island. Other states may have similar requirements or may adopt similar requirements in the future. Although we have obtained licenses from states where we believe we are required to be licensed, we may become aware of other states that require out-of-state laboratories to obtain licensure in order to accept specimens from the state, and it is possible that other states currently have such requirements or will have such requirements in the future.
In order to test specimens from New York, LDTs must be approved by the New York State Department of Health, or NYSDOH, on a test-by-test basis before they are offered. Our laboratory director must also be separately qualified to be a laboratory director in New York. DecisionDx-UM, DecisionDx-PRAME and DecisionDx-Melanoma have each been approved and our laboratory director has been qualified by NYSDOH. We are subject to periodic inspection by the NYSDOH and are required to demonstrate ongoing compliance with NYSDOH regulations and standards. To the extent NYSDOH identified any non-compliance and we are unable to remedy such non-compliance, the State of New York could withdraw approval for our products. We will need to seek NYSDOH approval of any future LDTs we develop and want to offer for clinical testing to New York residents, and there can be no assurance that we will be able to obtain such approval.
We may also be subject to regulation in foreign jurisdictions as we seek to expand international utilization of our products or such jurisdictions adopt new licensure requirements, which may require review of our products in order to offer them or may have other limitations such as restrictions on the transport of human tissue samples necessary for us to perform our tests that may limit our ability to make our products available outside of the United States. Complying with licensure requirements in new jurisdictions may be expensive, time-consuming and subject us to significant and unanticipated delays.
CAP maintains a clinical laboratory accreditation program. While not required for the operation of a CLIA-certified laboratory, many private insurers require CAP accreditation as a condition to contracting with clinical laboratories to cover their tests. In addition, some countries outside the United States require CAP accreditation as a condition to permitting clinical laboratories to test samples taken from their citizens. CAP accredited laboratories are surveyed for compliance with CAP standards every two years in order to maintain accreditation. Failure to maintain CAP accreditation could have a material adverse effect on the sales of our products and the results of our operations. Our most recent CAP inspection occurred in the fourth quarter of 2018 and our CLIA accreditation certificate expires on December 20, 2020.
Failure to comply with applicable clinical laboratory licensure requirements may result in a range of enforcement actions, including suspension, limitation or revocation of our CLIA accreditation and/or state licenses, imposition of a directed plan of action, onsite monitoring, civil monetary penalties, criminal sanctions and revocation of the laboratory’s approval to receive Medicare and Medicaid payment for its services, as well as significant adverse publicity. Any sanction imposed under CLIA, its implementing regulations, or state or foreign laws or regulations governing clinical laboratory licensure or our failure to renew our CLIA accreditation, or a state or foreign license, could have a material adverse effect on our business, financial condition and results of operations. Even if we were able to bring our laboratory back into compliance, we could incur significant expenses and potentially lose revenue in doing so.
25
The FDA may modify its enforcement discretion policy with respect to LDTs in a risk-based manner, and we may become subject to extensive regulatory requirements and may be required to conduct additional clinical trials prior to continuing to sell our existing tests or launching any other tests we may develop, which may increase the cost of conducting, or otherwise harm, our business.
If the FDA changes or ends its policy of enforcement discretion with respect to LDTs, and our products become subject to the FDA’s requirements for premarket review of medical devices, we may be required to cease commercial sales of our products and conduct clinical trials prior to making submissions to the FDA to obtain premarket clearance or approval. If we are required to conduct such clinical trials, delays in the commencement or completion of clinical trials could significantly increase our product development costs and delay commercialization of any currently marketed testing that we may be required to cease selling or the commercialization of any future tests that we may develop. Many of the factors that may cause or lead to a delay in the commencement or completion of clinical trials may also ultimately lead to delay or denial of regulatory clearance or approval. The commencement of clinical trials may be delayed due to insufficient patient enrollment, which is a function of many factors, including the size of the patient population, the nature of the protocol, the proximity of patients to clinical sites and the eligibility criteria for the clinical trial.
The FDA requires medical device manufacturers to comply with, among other things, current good manufacturing practices for medical devices, known as the Quality System Regulation, which requires manufacturers to follow elaborate design, testing, control, documentation and other quality assurance procedures during the manufacturing process; the medical device reporting regulation, which requires that manufacturers report to the FDA if their device may have caused or contributed to a death or serious injury or malfunctioned in a way that would likely cause or contribute to a death or serious injury if it were to recur; labeling regulations, including the FDA’s general prohibition against promoting products for unapproved or “off-label” uses; and the reports of corrections and removals regulation, which requires manufacturers to report to the FDA if a device correction or removal was initiated to reduce a risk to health posed by the device or to remedy a violation of the FDCA caused by the device which may present a risk to health.
Even if we were able to obtain FDA clearance or approval for one or more of our products, if required, a diagnostic test may be subject to limitations on the indications for which it may be marketed or to other regulatory conditions. In addition, such clearance or approval may contain requirements for costly post-market testing and surveillance to monitor the safety or efficacy of the test.
In addition, the FDA’s and other regulatory authorities’ policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approvals. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing authorization that we may have obtained and we may not achieve or sustain profitability, which would adversely affect our business, prospects, financial condition and results of operations.
Interim, topline and preliminary data from our clinical studies that we announce or publish from time to time may change as more data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publicly disclose preliminary or topline or data from our clinical studies, which is based on a preliminary analysis of then-available data, and the results and related findings and conclusions are subject to change following a more comprehensive review of the data related to the particular study. We also make assumptions, estimations, calculations and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully and carefully evaluate all data. As a result, the topline results that we report may differ from future results of the same studies, or different conclusions or considerations may qualify such results, once additional data have been received and fully evaluated. Topline data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, topline data should be viewed with caution until the final data are available. From time to time, we may also disclose interim data from our clinical studies. Interim data from clinical studies that we may complete are subject to the risk that one or more of the clinical outcomes may materially change as more patient data become available. Adverse differences between preliminary or interim data and final data could significantly harm our reputation and marketing efforts.
Further, others, including healthcare providers or payors, may not accept or agree with our assumptions, estimates, calculations, conclusions or analyses or may interpret or weigh the importance of data differently,
26
which could impact the value of the particular program, the approvability or commercialization of the particular product candidate or product and our company in general. In addition, the information we choose to publicly disclose regarding a particular study is based on what is typically extensive information, and you or others may not agree with what we determine is the material or otherwise appropriate information to include in our disclosure, and any information we determine not to disclose may ultimately be deemed significant with respect to future decisions, conclusions, views, activities or otherwise regarding our business. If the topline or interim data that we report differ from actual results, or if others, including healthcare providers or payors, disagree with the conclusions reached, our ability to commercialize, our product candidates may be harmed, which could harm our business, operating results, prospects or financial condition.
Changes in health care policy could increase our costs, decrease our revenues and impact sales of and reimbursement for our products.
In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, or the ACA, became law. This law substantially changed the way health care is financed by both government and commercial third-party payors, and significantly impacted our industry. The ACA contains a number of provisions that are expected to impact our business and operations, some of which in ways we cannot currently predict, including those governing enrollment in state and federal health care programs, reimbursement changes and fraud and abuse, which impact existing state and federal health care programs and will result in the development of new programs. Among other things, the ACA requires medical device manufacturers to pay a sales tax equal to 2.3% of the price for which such manufacturer sells its medical devices, and began to apply to sales of taxable medical devices after December 31, 2012. FDA officials have indicated that a laboratory will not have to pay the tax under the proposed “notification” procedure in the Notification draft guidance. However, the laboratory would have to pay the tax at the time that it lists a test with FDA. In FDA’s Notification draft guidance, listing occurs at the time a laboratory submits either a PMA or 510(k) for the test. While it is possible that this tax will apply to some or all of our products or products that are in development, for the time being, Congress has enacted a two-year moratorium on the medical device tax until January 1, 2020.
Since 2016 there have been efforts to repeal all or part of the ACA, and the current administration and the U.S. Congress have taken action to roll back certain provisions of the ACA. The current administration and the U.S. Congress may take further action regarding the ACA, including, but not limited to, repeal or replacement. Additionally, on December 14, 2018, a Texas U.S. District Court Judge ruled that the ACA is unconstitutional in its entirety because the tax penalty on certain individuals who fail to maintain qualifying health coverage for all or part of a year, commonly referred to as the “individual mandate,” was repealed by Congress as part of the Tax Cuts and Jobs Act of 2017, or the TCJA. While the Texas District Court Judge, as well as the current administration and CMS, have stated that the ruling will have no immediate effect pending appeal of the decision, it is unclear how this decision, subsequent appeals, and other efforts to repeal and replace the ACA will impact the ACA and our business.
On August 2, 2011, the Budget Control Act of 2011 was signed into law, which, among other things, reduced Medicare payments to providers by 2% per fiscal year, effective on April 1, 2013 and, due to subsequent legislative amendments to the statute, will remain in effect through 2027, unless additional Congressional action is taken.
We anticipate there will continue to be proposals by legislators at both the federal and state levels, regulators and commercial third-party payors to reduce costs while expanding individual healthcare benefits. Certain of these changes could impose additional limitations on the prices we will be able to charge for our products, the coverage of or the amounts of reimbursement available for our products from third-party payors, including government and commercial payors.
27
We are subject to numerous federal and state healthcare statutes and regulations, and complying with laws pertaining to our business is an expensive and time-consuming process. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties and a material adverse effect to our business and operations.
Physicians, other healthcare providers and third-party payors play a primary role in the recommendation of our products. Our arrangements with healthcare providers, third-party payors and customers may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that affect the business and financial arrangements and relationships through which we market and sell our products. The laws that affect our ability to operate include, but are not limited to:
| • | the federal Anti-Kickback Statute, or the AKS, which prohibits, among other things, any person or entity from knowingly and willfully soliciting, receiving, offering or paying any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchase, order or recommendation of an item or service reimbursable, in whole or in part, under a federal healthcare program, such as the Medicare and Medicaid programs. The term “remuneration” has been broadly interpreted to include anything of value, such as specimen collection materials or test kits. There are a number of statutory exceptions and regulatory safe harbors protecting some common activities from prosecution, however these are drawn narrowly. Additionally, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. Violations are subject to civil and criminal fines and monetary penalties of up to $100,000 for each violation, plus up to three times the remuneration involved, imprisonment of up to ten years and exclusion from government healthcare programs. In addition, the ACA codified case law that a claim including items or services resulting from a violation of the AKS constitutes a false or fraudulent claim for purposes of the federal False Claims Act, or the FCA; |
| • | the Stark Law, which prohibits a physician from making a referral for certain designated health services covered by the Medicare or Medicaid program, including laboratory and pathology services, if the physician or an immediate family member of the physician has a financial relationship with the entity providing the designated health services and prohibits that entity from billing, presenting or causing to be presented a claim for the designated health services furnished pursuant to the prohibited referral, unless an exception applies. Sanctions for violating the Stark Law include denial of payment, civil monetary penalties and exclusion from the federal health care programs. Failure to refund amounts received as a result of a prohibited referral on a timely basis may constitute a false or fraudulent claim and may result in civil penalties and additional penalties under the FCA; |
| • | federal civil and criminal false claims laws and civil monetary penalty laws, such as the FCA, which can be enforced by private citizens through civil qui tam actions, prohibits individuals or entities from, among other things, knowingly presenting, or causing to be presented through distribution of template medical necessity language or other coverage and reimbursement information, false, fictitious or fraudulent claims for payment or approval by the federal government, including federal health care programs, such as Medicare and Medicaid, and knowingly making, using or causing to be made or used a false record or statement material to a false or fraudulent claim, or knowingly making a false statement to improperly avoid, decrease or conceal an obligation to pay money to the federal government. In addition, a claim including items or services resulting from a violation of the AKS constitutes a false or fraudulent claim for purposes of the FCA. Private individuals can bring False Claims Act “qui tam” actions, on behalf of the government and such individuals, commonly known as “whistleblowers,” may share in amounts paid by the entity to the government in fines or settlement. When an entity is determined to have violated the federal civil False Claims Act, the government may impose civil fines and penalties, plus treble damages, and exclude the entity from participation in Medicare, Medicaid and other federal healthcare programs; |
| • | HIPAA, which, among other things, imposes criminal liability for executing or attempting to execute a scheme to defraud any healthcare benefit program, including private third-party payors, knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense, and knowingly and willfully falsifying, concealing or covering up |
28
a material fact or making any materially false, fictitious or fraudulent statement or representation, in connection with the delivery of or payment for healthcare benefits, items or services. Like the AKS, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation;
| • | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and their implementing regulations, which imposes privacy, security and breach reporting obligations with respect to individually identifiable health information upon entities subject to the law, such as health plans, healthcare clearinghouses and certain healthcare providers, known as covered entities, and their respective business associates, individuals or entities that perform services for them that involve individually identifiable health information. Failure to comply with the HIPAA privacy and security standards can result in civil monetary penalties, and, in certain circumstances, criminal penalties. HITECH also created new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority to file civil actions for damages or injunctions in U.S. federal courts to enforce HIPAA and seek attorneys’ fees and costs associated with pursuing federal civil actions; |
| • | state laws that prohibit other specified practices, such as billing physicians for tests that they order or providing tests at no or discounted cost to induce physician or patient adoption; insurance fraud laws; waiving coinsurance, copayments, deductibles, and other amounts owed by patients; billing a state Medicaid program at a price that is higher than what is charged to one or more other third-party payors employing, exercising control over or splitting professional fees with licensed professionals in violation of state laws prohibiting fee splitting or the corporate practice of medicine and other professions; and |
| • | federal and state consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm consumers; |
| • | the federal transparency requirements under the Physician Payments Sunshine Act, created under the ACA, which requires, among other things, certain manufacturers of drugs, devices, biologics and medical supplies reimbursed under Medicare, Medicaid, or the Children’s Health Insurance Program to annually report to CMS information related to payments and other transfers of value provided to physicians, certain other healthcare professionals, and teaching hospitals and physician ownership and investment interests, including such ownership and investment interests held by a physician’s immediate family members. Failure to submit required information may result in civil monetary penalties for all payments, transfers of value or ownership or investment interests that are not timely, accurately, and completely reported in an annual submission, and may result in liability under other federal laws or regulations. We believe that we are exempt from these reporting requirements. We cannot assure you, however, that our regulators, principally the federal government, will agree with our determination, and a determination that we have violated these laws and regulations, or a public announcement that we are being investigated for possible violations, could adversely affect our business; |
| • | the prohibition on reassignment of Medicare claims, which, subject to certain exceptions, precludes the reassignment of Medicare claims to any other part; |
| • | state and foreign law equivalents of each of the above federal laws, such as anti-kickback and false claims laws, that may impose similar or more prohibitive restrictions, and may apply to items or services reimbursed by any non-governmental third-party payors, including private insurers; and |
| • | federal, state and foreign laws that govern the privacy and security of health information or personally identifiable information in certain circumstances, including state health information privacy and data breach notification laws which govern the collection, use, disclosure, and protection of health-related and other personal information, many of which differ from each other in significant ways and often are not pre-empted by HIPAA, thus complicating compliance efforts. |
As a clinical laboratory, our business practices may face additional scrutiny from government regulatory agencies such as the Department of Justice, the U.S. Department of Health and Human Services Office of Inspector General, or OIG, and CMS. Certain arrangements between clinical laboratories and referring physicians have been identified in fraud alerts issued by the OIG as implicating the AKS. The OIG has stated that it is particularly concerned about these types of arrangements because the choice of laboratory, as well as the decision
29
to order laboratory tests, typically are made or strongly influenced by the physician, with little or no input from patients. Moreover, the provision of payments or other items of value by a clinical laboratory to a referral source could be prohibited under the Stark Law unless the arrangement meets all criteria of an applicable exception. The government has been active in enforcement of these laws as they apply to clinical laboratories.
We have entered into consulting and scientific advisory board arrangements, speaking arrangements and clinical research agreements with physicians and other healthcare providers, including some who could influence the use of our products. Because of the complex and far-reaching nature of these laws, regulatory agencies may view these transactions as prohibited arrangements that must be restructured, or discontinued, or for which we could be subject to other significant penalties. We could be adversely affected if regulatory agencies interpret our financial relationships with providers who may influence the ordering of and use of our products to be in violation of applicable laws.
The scope and enforcement of each of these laws is uncertain and subject to rapid change in the current environment of healthcare reform. Federal and state enforcement bodies have recently increased their scrutiny of interactions between healthcare companies, healthcare providers and other third parties, including charitable foundations, which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry. It is possible that governmental authorities may conclude that our business practices, including our consulting arrangements with physicians, as well as our financial assistance programs, do not comply with current or future statutes, regulations, agency guidance or case law involving applicable healthcare laws. Responding to investigations can be time and resource-consuming and can divert management’s attention from the business. Any such investigation or settlement could increase our costs or otherwise have an adverse effect on our business.
Ensuring that our business arrangements with third parties comply with applicable healthcare laws and regulations is costly. If our operations are found to be in violation of any of these laws or any other current or future governmental laws and regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, disgorgement, individual imprisonment, exclusion from government funded healthcare programs, such as Medicare and Medicaid, contractual damages, reputational harm, diminished profits and future earnings, additional reporting obligations and oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws, and the curtailment or restructuring of our operations, any of which could substantially disrupt our operations. If any of the physicians or other healthcare providers or entities with whom we do business is found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs.
We are subject to certain U.S. anti-corruption, anti-money laundering, export control, sanctions, and other trade laws and regulations and may become subject to their similar foreign equivalents. We can face serious consequences for violations.
U.S. and foreign anti-corruption, anti-money laundering, export control, sanctions, and other trade laws and regulations, or collectively, Trade Laws, prohibit, among other things, companies and their employees, agents, legal counsel, accountants, consultants, contractors, and other partners from authorizing, promising, offering, providing, soliciting, or receiving, directly or indirectly, corrupt or improper payments or anything else of value to or from recipients in the public or private sector. Violations of Trade Laws can result in substantial criminal fines and civil penalties, imprisonment, the loss of trade privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm, and other consequences. We have direct or indirect interactions with officials and employees of government agencies or government-affiliated hospitals, universities, and other organizations. We also expect that we may engage in non-U.S. activities over time. We expect to rely on third-party suppliers and/or third parties to obtain necessary permits, licenses, and patent registrations. We can be held liable for the corrupt or other illegal activities of our personnel, agents, or partners, even if we do not explicitly authorize or have prior knowledge of such activities. Any violations of the laws and regulations described above may result in substantial civil and criminal fines and penalties, imprisonment, the loss of export or import privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm and other consequences.
30
Our collection, use and disclosure of individually identifiable information, including health and/or employee information, is subject to state, federal and foreign privacy and security regulations, and our failure to comply with those regulations or to adequately secure the information we hold could result in significant liability or reputational harm.
We and any potential collaborators are subject to federal, state, and foreign data protection laws and regulations (i.e., laws and regulations that address privacy and data security). In the United States, numerous federal and state laws and regulations, including federal health information privacy laws, state data breach notification laws, state health information privacy laws, and federal and state consumer protection laws (e.g., Section 5 of the Federal Trade Commission Act), that govern the collection, use, disclosure, and protection of health-related and other personal information could apply to our operations or the operations of our collaborators.
In the ordinary course of our business, we collect and store sensitive data, including protected health information, or PHI, personally identifiable information, or PII, credit card and other financial information, intellectual property and proprietary business information owned or controlled by ourselves or our customers, payors and other parties. We manage and maintain our applications and data utilizing a combination of on-site systems, managed data centers, and cloud-based data centers. We utilize external security and infrastructure vendors to manage parts of our data centers.
The secure processing, storage, maintenance and transmission of this critical information is vital to our operations and business strategy, and we devote significant resources to protecting such information. Although we take measures to protect sensitive data from unauthorized access, use or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers or viruses or breached due to employee error, malfeasance, or other malicious or inadvertent disruptions. Any such breach or interruption could compromise our networks and the information stored there could be accessed by unauthorized parties, publicly disclosed, lost, or stolen. Any such access, breach, or other loss of information could result in legal claims or proceedings, and liability under federal or state laws that protect the privacy of personal information, such as HIPAA, as amended by HITECH, and regulatory penalties. Notice of breaches must be made to affected individuals, the Secretary of the Department of Health and Human Services, and for extensive breaches, notice may need to be made to the media or State Attorneys General. Such a notice could harm our reputation and our ability to compete. Although we have implemented security measures and a formal, dedicated enterprise security program to prevent unauthorized access to patient data, such data is currently accessible through multiple channels, and there is no guarantee we can protect our data from breach. Unauthorized access, loss or dissemination could also disrupt our operations (including our ability to conduct our analyses, provide test results, bill payers or patients, process claims and appeals, provide customer assistance, conduct research and development activities, collect, process, and prepare company financial information, provide information about our products and other patient and physician education and outreach efforts through our website, and manage the administrative aspects of our business) and damage our reputation, any of which could adversely affect our business. In addition, we may obtain health information from third parties that are also subject to privacy and security requirements under HIPAA, as amended by HITECH.
Further, various states, such as California and Massachusetts, have implemented similar privacy laws and regulations, such as the California Confidentiality of Medical Information Act, that impose restrictive requirements regulating the use and disclosure of health information and other personally identifiable information. These laws and regulations are not necessarily preempted by HIPAA, particularly if a state affords greater protection to individuals than HIPAA. Where state laws are more protective, we have to comply with the stricter provisions. In addition to fines and penalties imposed upon violators, some of these state laws also afford private rights of action to individuals who believe their personal information has been misused. California’s patient privacy laws, for example, provide for penalties of up to $250,000 and permit injured parties to sue for damages. The interplay of federal and state laws may be subject to varying interpretations by courts and government agencies, creating complex compliance issues for us and our clients and potentially exposing us to additional expense, adverse publicity and liability. Further, as regulatory focus on privacy issues continues to increase and laws and regulations concerning the protection of personal information expand and become more complex, these potential risks to our business could intensify. Changes in laws or regulations associated with the enhanced protection of certain types of sensitive data, such PHI or PII along with increased customer demands for enhanced data security infrastructure, could greatly increase our cost of providing our services, decrease demand for our services, reduce our revenue and/or subject us to additional liabilities.
31
Compliance with U.S. and international data protection laws and regulations could require us to take on more onerous obligations in our contracts, restrict our ability to collect, use and disclose data, or in some cases, impact our ability to operate in certain jurisdictions. Failure to comply with U.S. and international data protection laws and regulations could result in government enforcement actions (which could include civil, criminal, and administrative penalties), private litigation, and/or adverse publicity and could negatively affect our operating results and business. Claims that we have violated individuals’ privacy rights, failed to comply with data protection laws, or breached our contractual obligations, even if we are not found liable, could be expensive and time-consuming to defend and could result in adverse publicity that could harm our business.
Ethical, legal and social concerns related to the use of genetic information could reduce demand for our products.
Genetic testing has raised ethical, legal, and social issues regarding privacy and the appropriate uses of the resulting information. Governmental authorities have, through the Genetic Information Nondisclosure Act of 2008, and could further, for social or other purposes, limit or regulate the use of genetic information or genetic testing or prohibit testing for genetic predisposition to certain conditions, particularly for those that have no known cure. Ethical and social concerns may also influence governmental authorities to deny or delay the issuance of patents for technology relevant to our business. While we do not currently perform genetic tests for genetic predisposition to certain conditions, these concerns may lead patients to refuse to use, or clinicians to be reluctant to order, our genomic tests or genetic tests for somatic mutations even if permissible. These and other ethical, legal and social concerns may limit market acceptance of our products or reduce the potential markets for our products, either of which could have an adverse effect on our business, financial condition, or results of operations.
Risks Related to Intellectual Property
If we are unable to obtain and maintain sufficient intellectual property protection for our technology, or if the scope of the intellectual property protection obtained is not sufficiently broad, our competitors could develop and commercialize diagnostic tests similar or identical to ours, and our ability to successfully commercialize our products may be impaired.
We rely on patent protection as well as trademark, copyright, trade secret and other intellectual property rights protection as well as nondisclosure, confidentiality and other contractual restrictions to protect our brands and proprietary tests and technologies, all of which provide limited protection and may not adequately protect our rights or permit us to gain or keep any competitive advantage. If we fail to protect our intellectual property, third parties may be able to compete more effectively against us. In addition, we may incur substantial litigation costs in our attempts to recover or restrict use of our intellectual property.
As is the case with other life science companies, our success depends in large part on our ability to obtain and maintain protection of the intellectual property we may own solely or jointly with others or in-license from others, particularly patents, in the United States and other countries with respect to our products and technologies. We apply for patents covering our products and technologies and uses thereof, as we deem appropriate. However, obtaining and enforcing life sciences patents is costly, time-consuming and complex, and we may fail to apply for patents on important tests, services and technologies in a timely fashion or at all, or we may fail to apply for patents in potentially relevant jurisdictions. We may not be able to file and prosecute all necessary or desirable patent applications, or maintain, enforce and license any patents that may issue from such patent applications, at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. We may not have the right to control the preparation, filing and prosecution of patent applications, or to maintain the rights to patents licensed from or to third parties. Therefore, these patents and applications may not be prosecuted and enforced in a manner consistent with the best interests of our business.
As of the date of this prospectus, we had four licensed U.S. patents and two pending U.S. patent applications, with foreign counterparts. It is possible that none of our pending patent applications will result in issued patents in a timely fashion or at all, and even if patents are granted, they may not provide a basis for intellectual property protection of commercially viable tests or services, may not provide us with any competitive advantages, or may be challenged and invalidated by third parties. It is possible that others will design around our future patented technologies. We may not be successful in defending any such challenges made against our patents or patent applications. Any successful third-party challenge to our patents could result in the
32
unenforceability or invalidity of such patents and increased competition to our business. Even if our patents are held valid and enforceable, they may still be found insufficient to provide protection against competing products and services sufficient to achieve our business objectives. We may have to challenge the patents or patent applications of third parties, such as to counter infringement or unauthorized use. In addition, in an infringement proceeding, a court may decide that a patent of ours is invalid or unenforceable, or may refuse to enjoin the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. Even if we prevail against an infringer in a U.S. district court or foreign trial-level court, there is always the risk that the infringer will file an appeal and the initial court judgment will be overturned at the appeals court and/or that an adverse decision will be issued by the appeals court relating to the validity or enforceability of our patents. The outcome of patent litigation or other proceeding can be uncertain, and any attempt by us to enforce our patent rights against others or to challenge the patent rights of others may not be successful, or, if successful, may take substantial time and result in substantial cost, and may divert our efforts and attention from other aspects of our business.
The patent positions of life sciences companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. No consistent policy regarding the breadth of claims allowed in such companies’ patents has emerged to date in the United States or elsewhere. Courts frequently render opinions in the life sciences field that may affect the patentability of certain inventions or discoveries, including opinions that may affect the patentability of methods for analyzing or comparing DNA sequences.
In particular, the patent positions of companies engaged in the development and commercialization of genomic diagnostic tests are particularly uncertain. Various courts, including the U.S. Supreme Court, have rendered decisions that affect the scope of patentability of certain inventions or discoveries relating to certain diagnostic tests and related methods. These decisions state, among other things, that a patent claim that recites an abstract idea, natural phenomenon or law of nature (for example, the relationship between particular genetic variants and cancer) are not themselves patentable. Precisely what constitutes a law of nature is uncertain, and it is possible that certain aspects of genetic diagnostics tests would be considered natural laws. Accordingly, the evolving case law in the United States may adversely affect our ability to obtain patents and may facilitate third-party challenges to any owned or licensed patents. The laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the United States, and we may encounter difficulties in protecting and defending such rights in foreign jurisdictions. The legal systems of many other countries do not favor the enforcement of patents and other intellectual property protection, particularly those relating to life science technologies, which could make it difficult for us to stop the infringement of our patents in such countries. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business.
To the extent our intellectual property offers inadequate protection, or is found to be invalid or unenforceable, we would be exposed to a greater risk of direct competition, and our competitive position could be adversely affected, as could our business. Both the patent application process and the process of managing patent disputes can be time-consuming and expensive. Moreover, if a third party has intellectual property rights that cover the practice of our technology, we may not be able to fully exercise or extract value from our intellectual property rights. The following examples are illustrative:
| • | others may be able to develop and/or practice technology that is similar to our technology or aspects of our technology, but that are not covered by the claims of the patents that we own or control, assuming such patents have issued or do issue; |
| • | we or our licensors or any future strategic partners might not have been the first to conceive or reduce to practice the inventions covered by the issued patents or pending patent applications that we own or have exclusively licensed; |
| • | we or our licensors or any future strategic partners might not have been the first to file patent applications covering certain of our inventions; |
| • | others may independently develop similar or alternative technologies or duplicate any of our technologies without infringing our intellectual property rights; |
| • | it is possible that our pending patent applications will not lead to issued patents; |
33
| • | issued patents that we own or have exclusively licensed may not provide us with any competitive advantage, or may be held invalid or unenforceable, as a result of legal challenges by our competitors; |
| • | our competitors might conduct research and development activities in countries where we do not have patent rights and then use the information learned from such activities to develop competitive tests for sale in our major commercial markets; |
| • | third parties performing manufacturing or testing for us using our products or technologies could use the intellectual property of others without obtaining a proper license; |
| • | parties may assert an ownership interest in our intellectual property and, if successful, such disputes may preclude us from exercising exclusive rights over that intellectual property; |
| • | we may not develop or in-license additional proprietary technologies that are patentable; |
| • | we may not be able to obtain and maintain necessary licenses on commercially reasonable terms, or at all; and |
| • | the patents of others may have an adverse effect on our business. |
Should any of these events occur, they could significantly harm our business and results of operations.
Changes in patent law in the United States and other jurisdictions could diminish the value of patents in general, thereby impairing our ability to protect our products.
As is the case with other life sciences companies, our success is heavily dependent on intellectual property, particularly patents relating to our research programs and products. Obtaining and enforcing patents in the life sciences industry involves both technological and legal complexity and is therefore costly, time consuming and inherently uncertain. Changes in either the patent laws or interpretation of the patent laws in the United States or the United States Patent and Trademark Office, or the USPTO, rules and regulations could increase these uncertainties and costs. Recent patent reform legislation in the United States and other countries, including the Leahy-Smith America Invents Act, or the AIA, signed into law on September 16, 2011, could increase those uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. The AIA includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted, redefine prior art and provide more efficient and cost-effective avenues for competitors to challenge the validity of patents. These include allowing third-party submission of prior art to the USPTO during patent prosecution and additional procedures to attack the validity of a patent in USPTO administered post-grant proceedings, including post-grant review, inter partes review, and derivation proceedings. For applications filed after March 15, 2013 that do not claim the benefit of applications filed before that date, the AIA transitioned the United States from a first to invent system to a first-inventor-to-file system in which, assuming that the other statutory requirements are met, the first inventor to file a patent application will be entitled to the patent on an invention regardless of whether a third party was the first to invent the claimed invention. The AIA and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications, our ability to obtain future patents, and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business, financial condition, results of operations and prospects.
The U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. Depending on future actions by the U.S. Congress, the U.S. courts, the USPTO and the relevant law-making bodies in other countries, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and patents that we might obtain in the future.
Our in-licensed intellectual property has been discovered through government funded programs and thus may be subject to federal regulations such as “march-in” rights, certain reporting requirements and a preference for U.S.-based companies, and compliance with such regulations may limit our exclusive rights, and limit our ability to contract with non-U.S. manufacturers.
Intellectual property rights that have been in-licensed pursuant to our license agreement, or the License Agreement, with The Washington University in St. Louis, Missouri, or WUSTL, have been generated through the use of U.S. government funding, and are therefore subject to certain federal regulations. As a result, the United
34
States federal government may retain certain rights to intellectual property embodied in our current or future product candidates under the Bayh-Dole Act. These federal government rights include a “nonexclusive, nontransferable, irrevocable, paid-up license” to use inventions for any governmental purpose. The Bayh-Dole Act also provides federal agencies with “march-in rights.” March-in rights allow the government, in specified circumstances, to require the contractor or successors in title to the patent to grant a “nonexclusive, partially exclusive, or exclusive license” to a “responsible applicant or applicants” if it determines that (1) adequate steps have not been taken to commercialize the invention, (2) government action is necessary to meet public health or safety needs or (3) government action is necessary to meet requirements for public use under federal regulations. If the patent owner refuses to do so, the government may grant the license itself.
The U.S. government also has the right to take title to these inventions if the licensor fails to disclose the invention to the government or fails to file an application to register the intellectual property within specified time limits. Intellectual property generated under a government funded program is also subject to certain reporting requirements, compliance with which may require us to expend substantial resources. In addition, the U.S. government requires that any products embodying any of these inventions or produced through the use of any of these inventions be manufactured substantially in the United States, and the License Agreement requires that we comply with this requirement. This preference for U.S. industry may be waived by the federal agency that provided the funding if the owner or assignee of the intellectual property can show that reasonable but unsuccessful efforts have been made to grant licenses on similar terms to potential licensees that would be likely to manufacture substantially in the United States or that under the circumstances domestic manufacture is not commercially feasible. This preference for U.S. industry may limit our ability to contract with non-U.S. product manufacturers for products covered by such intellectual property. To the extent any of our owned or future in-licensed intellectual property is also generated through the use of U.S. government funding, the provisions of the Bayh-Dole Act may similarly apply.
Issued patents covering our products and related technologies could be found invalid or unenforceable if challenged.
The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability. Some of our patents or patent applications (including licensed patents) have been, are being or may be challenged at a future point in time in an opposition, nullification, derivation, reexamination, inter partes review, post-grant review or interference action in court or before patent offices or similar proceedings for a given period after allowance or grant, during which time third parties can raise objections against such grant. In the course of such proceedings, which may continue for a protracted period of time, the patent owner may be compelled to limit the scope of the allowed or granted claims thus attacked, or may lose the allowed or granted claims altogether. Any successful third-party challenge to our patents in this or any other proceeding could result in the unenforceability or invalidity of such patents, which may lead to increased competition to our business, which could harm our business. In addition, if the breadth or strength of protection provided by our patents and patent applications is threatened, regardless of the outcome, it could dissuade companies from collaborating with us to license, develop or commercialize current or future diagnostic tests.
We may not be aware of all third-party intellectual property rights potentially relating to our products. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until approximately 18 months after filing or, in some cases (e.g., U.S. applications for which a request not to publish has been filed), not until such patent applications issue as patents. We might not have been the first to make the inventions covered by each of our pending patent applications and we might not have been the first to file patent applications for these inventions. To determine the priority of these inventions, we have and may have to participate in interference proceedings, derivation proceedings or other post-grant proceedings declared by the USPTO that could result in substantial cost to us. The outcome of such proceedings is uncertain. We can give no assurance that all of the potentially relevant art relating to our patents and patent applications has been found; overlooked prior art could be used by a third party to challenge the validity, enforceability and scope of our patents or prevent a patent from issuing from a pending patent application. As a result, we may not be able to obtain or maintain protection for certain inventions. No assurance can be given that other patent applications will not have priority over our patent applications. In addition, changes to the patent laws of the United States allow for various post-grant opposition proceedings that have not been extensively tested, and their outcome is therefore uncertain. Therefore, the validity, enforceability and scope of our patents in the United States and other countries cannot be predicted with certainty and, as a result, any patents that we own or license may not provide
35
sufficient protection against our competitors. Furthermore, if third parties bring these proceedings against our patents, we could experience significant costs and management distraction.
Our commercial success depends significantly on our ability to operate without infringing upon the intellectual property rights of third parties.
The life sciences industry is subject to rapid technological change and substantial litigation regarding patent and other intellectual property rights. Our potential competitors in both the United States and abroad, may have substantially greater resources and are likely to make substantial investments in patent portfolios and competing technologies, and may apply for or obtain patents that could prevent, limit or otherwise interfere with our ability to make, use and sell our products. Numerous third-party patents exist in fields relating to our products and technologies, and it is difficult for industry participants, including us, to identify all third-party patent rights relevant to our products and technologies. Moreover, because some patent applications are maintained as confidential for a certain period of time, we cannot be certain that third parties have not filed patent applications that cover our products and technologies.
Patents could be issued to third parties that we may ultimately be found to infringe. Third parties may have or obtain valid and enforceable patents or proprietary rights that could block us from using our technology. Our failure to obtain or maintain a license to any technology that we require may materially harm our business, financial condition and results of operations. Furthermore, we would be exposed to a threat of litigation.
From time to time, we may be party to, or threatened with, litigation or other proceedings with third parties, including non-practicing entities, who allege that our products, components of our products, and/or proprietary technologies infringe, misappropriate or otherwise violate their intellectual property rights. The types of situations in which we may become a party to such litigation or proceedings include:
| • | we may initiate litigation or other proceedings against third parties seeking to invalidate the patents held by those third parties or to obtain a judgment that our products or technologies do not infringe those third parties’ patents; |
| • | we may participate at substantial cost in International Trade Commission proceedings to abate importation of products that would compete unfairly with our products or technologies; |
| • | if a competitor files patent applications that claim technology also claimed by us or our licensors, we or our licensors may be required to participate in interference, derivation or opposition proceedings to determine the priority of invention, which could jeopardize our patent rights and potentially provide a third party with a dominant patent position; |
| • | if third parties initiate litigation claiming that our products or technologies infringe their patent or other intellectual property rights, we will need to defend against such proceedings; |
| • | if third parties initiate litigation or other proceedings seeking to invalidate patents owned by or licensed to us or to obtain a declaratory judgment that their products, services, or technologies do not infringe our patents or patents licensed to us, we will need to defend against such proceedings; |
| • | we may be subject to ownership disputes relating to intellectual property, including disputes arising from conflicting obligations of consultants or others who are involved in developing our products and technologies; and |
| • | if a license to necessary technology is terminated, the licensor may initiate litigation claiming that our products or technologies infringe or misappropriate its patent or other intellectual property rights and/or that we breached our obligations under the license agreement, and we would need to defend against such proceedings. |
These lawsuits and proceedings, regardless of merit, are time-consuming and expensive to initiate, maintain, defend or settle, and could divert the time and attention of managerial and technical personnel, which could materially adversely affect our business. Any such claim could also force us to do one or more of the following:
| • | incur substantial monetary liability for infringement or other violations of intellectual property rights, which we may have to pay if a court decides that the diagnostic test or technology at issue infringes or violates the third party’s rights, and if the court finds that the infringement was willful, we could be ordered to pay treble damages and the third party’s attorneys’ fees; |
36
| • | stop manufacturing, offering for sale, selling, using, importing, exporting or licensing the diagnostic test or technology incorporating the allegedly infringing technology or stop incorporating the allegedly infringing technology into such test or technology; |
| • | obtain from the owner of the infringed intellectual property right a license, which may require us to pay substantial upfront fees or royalties to sell or use the relevant technology and which may not be available on commercially reasonable terms, or at all; |
| • | redesign our products and technologies so they do not infringe or violate the third party’s intellectual property rights, which may not be possible or may require substantial monetary expenditures and time; |
| • | enter into cross-licenses with applicable third party, which could weaken our overall intellectual property position; |
| • | lose the opportunity to license our technology to others or to collect royalty payments based upon successful protection and assertion of our intellectual property against others; |
| • | find alternative suppliers for non-infringing technologies, which could be costly and create significant delay; or |
| • | relinquish rights associated with one or more of our patent claims, if our claims are held invalid or otherwise unenforceable. |
Third parties may be able to sustain the costs of complex intellectual property litigation more effectively than we can because they have substantially greater resources. In addition, intellectual property litigation, regardless of its outcome, may cause negative publicity, adversely impact our business, cause delays, or prohibit us from marketing or otherwise commercializing our products and technologies. Any uncertainties resulting from the initiation and continuation of any litigation could have a material adverse effect on our ability to raise additional funds or otherwise have a material adverse effect on our business, results of operation, financial condition or cash flows.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments, which could have a material adverse effect on the price of our common stock. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our common stock. The occurrence of any of these events may have a material adverse effect on our business, results of operation, financial condition or cash flows.
We depend on information technology systems that we license from third parties. Any failure of such systems or loss of licenses to the software that comprises an essential element of such systems could significantly harm our business.
We depend on information technology systems for significant elements of our operations, such as our laboratory information management systems, including test validation, specimen tracking and quality control, our bioinformatics analytical software systems and our test report generating systems. Essential elements of these systems depend on software that we license from third parties. If we are unable to maintain the licenses to this software or our software providers discontinue or alter the programs on which we rely, it could render our test reports unreliable or hinder our ability to generate accurate test reports, among other things. Any of the foregoing could have a material adverse effect on our competitive position, business, financial conditions, results of operations and prospects.
37
We rely on licenses from third parties, and if we lose these licenses or are not able to obtain licenses to third-party technology on reasonable grounds or at all, then we may not be able to continue to commercialize existing diagnostic tests, be subjected to future litigation and may not be able to commercialize new diagnostic tests in the future.
We are party to certain royalty-bearing license agreements that grant us rights to use certain intellectual property, including patents and patent applications, in certain specified fields of use. Although we intend to develop products and technologies through our own internal research, we may need to obtain additional licenses from others to advance our research, development and commercialization activities. Our license agreements impose, and we expect that future license agreements will impose, various development, diligence, commercialization and other obligations on us.
In the future, we may identify third-party technology we may need, including to develop or commercialize new diagnostic tests or services. In return for the use of a third party’s technology, we may agree to pay the licensor royalties based on sales of our solutions. Royalties are a component of the cost of our products or services and affect our margins. We may also need to negotiate licenses to patents or patent applications before or after introducing a commercialized test. The in-licensing and acquisition of third-party intellectual property rights is a competitive area, and a number of more established companies are also pursuing strategies to in-license or acquire third-party intellectual property rights for technologies that we may consider attractive or necessary. These established companies may have a competitive advantage over us due to their size, cash resources and greater clinical development and commercialization capabilities. Furthermore, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. In addition, we expect that competition for the in-licensing or acquisition of third-party intellectual property rights for technologies that are attractive to us may increase in the future, which may mean fewer suitable opportunities for us as well as higher acquisition or licensing costs. We may not be able to obtain necessary or strategic licenses to patents or patent applications, and our business may suffer if we are unable to enter into these licenses on acceptable terms or at all, if any necessary licenses are subsequently terminated, if the licensors fail to abide by the terms of the licenses or fail to prevent infringement by third parties, or if the licensed patents or other rights are found to be invalid or unenforceable.
In spite of our efforts, our licensors might conclude that we have materially breached our obligations under such license agreements and might therefore terminate the license agreements, thereby removing or limiting our ability to develop and commercialize tests and technology covered by these license agreements. If these in-licenses are terminated, or if the underlying patents fail to provide the intended exclusivity, competitors or other third parties might have the freedom to seek regulatory approval of, and to market, tests identical to ours and we may be required to cease our development and commercialization activities. For example, we license certain intellectual property from WUSTL that is incorporated into DecisionDx-UM. In 2018, we provided more than 1,400 test reports for DecisionDx-UM. If this license agreement were terminated, we would be unable to continue to issue test reports and thus sales of DecisionDx-UM. Any of the foregoing could have a material adverse effect on our competitive position, business, financial conditions, results of operations and prospects.
Moreover, disputes may arise with respect to any one of our licensing agreements, including:
| • | the scope of rights granted under the license agreement and other interpretation-related issues; |
| • | the extent to which our products, technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement; |
| • | the sublicensing of patent and other rights under our collaborative development relationships; |
| • | our diligence obligations under the license agreement and what activities satisfy those diligence obligations; |
| • | the inventorship and ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our partners; and |
| • | the priority of invention of patented technology. |
If we do not prevail in such disputes, we may lose any of such license agreements.
In addition, the agreements under which we currently license intellectual property or technology from third parties are complex, and certain provisions in such agreements may be susceptible to multiple interpretations.
38
The resolution of any contract interpretation disagreement that may arise could narrow what we believe to be the scope of our rights to the relevant intellectual property or technology, or increase what we believe to be our financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, financial condition, results of operations and prospects. Moreover, if disputes over intellectual property that we have licensed prevent or impair our ability to maintain our current licensing arrangements on commercially acceptable terms, we may be unable to successfully develop and commercialize the affected diagnostic tests, which could have a material adverse effect on our business, financial conditions, results of operations and prospects.
Our failure to maintain such licenses could have a material adverse effect on our business, financial condition and results of operations. Any of these licenses could be terminated, such as if either party fails to abide by the terms of the license, or if the licensor fails to prevent infringement by third parties or if the licensed patents or other rights are found to be invalid or unenforceable. Absent the license agreements, we may infringe patents subject to those agreements, and if the license agreements are terminated, we may be subject to litigation by the licensor. Litigation could result in substantial costs and be a distraction to management. If we do not prevail, we may be required to pay damages, including treble damages, attorneys’ fees, costs and expenses, royalties or, be enjoined from selling our products or services, including DecisionDx-UM and DecisionDx-Melanoma, which could adversely affect our ability to offer our products or services, our ability to continue operations and our financial condition.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on our products in all countries throughout the world would be prohibitively expensive. The requirements for patentability may differ in certain countries, particularly developing countries, and the breadth of patent claims allowed can be inconsistent. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the United States, and we may encounter difficulties in protecting and defending such rights in foreign jurisdictions. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own tests or products and may also export infringing tests or products to territories where we have patent protection, but enforcement is not as strong as in the United States. These products may compete with our products. Our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of many other countries do not favor the enforcement of patents and other intellectual property protection, particularly those relating to life science technologies, which could make it difficult for us to stop the infringement of our patents in such countries. We do not have patent rights in certain foreign countries in which a market may exist. Moreover, in foreign jurisdictions where we do have patent rights, proceedings to enforce our patent rights could result in substantial cost and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license. We may not be able to stop a competitor from marketing and selling in foreign countries tests, products and services that are the same as or similar to our products and technologies, in which case our competitive position in the international market would be harmed.
If we are unable to protect the confidentiality of our trade secrets, the value of our technology could be materially adversely affected and our business could be harmed.
In addition to pursuing patents on our technology, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. We take steps to protect our trade secrets, in part, by entering into agreements, including confidentiality agreements, non-disclosure agreements and intellectual property assignment agreements, with our employees, consultants, academic institutions, corporate partners and, when needed, our advisers. However, we cannot be certain that such
39
agreements have been entered into with all relevant parties, and we cannot be certain that our trade secrets and other confidential proprietary information will not be disclosed or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. For example, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and, once disclosed, we are likely to lose trade secret protection and may not be able to obtain adequate remedies for such breaches. Such agreements may not be enforceable or may not provide meaningful protection for our trade secrets or other proprietary information in the event of unauthorized use or disclosure or other breaches of the agreements, and we may not be able to prevent such unauthorized disclosure. If we are required to assert our rights against such party, it could result in significant cost and distraction.
Monitoring unauthorized disclosure is difficult, and we do not know whether the steps we have taken to prevent such disclosure are, or will be, adequate. If we were to enforce a claim that a third party had illegally obtained and was using our trade secrets, it would be expensive and time-consuming, and the outcome would be unpredictable. In addition, courts outside the United States may be less willing to protect trade secrets.
We also seek to preserve the integrity and confidentiality of our confidential proprietary information by maintaining physical security of our premises and physical and electronic security of our information technology systems, but it is possible that these security measures could be breached. If any of our confidential proprietary information were to be lawfully obtained or independently developed by a competitor, absent patent protection, we would have no right to prevent such competitor from using that technology or information to compete with us, which could harm our competitive position.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties.
We do and may employ individuals who previously worked with universities or other companies, including potential competitors. We could in the future be subject to claims that we or our employees, consultants, or independent contractors have inadvertently or otherwise used or disclosed alleged trade secrets or other confidential information of current or former employers or competitors. Although we try to ensure that our employees, consultants and independent contractors do not use the intellectual property, proprietary information, know-how or trade secrets of others in their work for us, we may become subject to claims that we caused an individual to breach the terms of his or her non-competition or non-solicitation agreement, or that we or these individuals have, inadvertently or otherwise, used or disclosed the alleged trade secrets or other proprietary information of a current or former employer or competitor. Although, we are currently not subject to any such claims.
While we may litigate to defend ourselves against these claims, even if we are successful, litigation could result in substantial costs and could be a distraction to management and other employees. If our defenses to these claims fail, in addition to requiring us to pay monetary damages, a court could prohibit us from using technologies or features that are essential to our products, if such technologies or features are found to incorporate or be derived from the trade secrets or other proprietary information of the current or former employers. Therefore, we could be required to obtain a license from such third-party employer to commercialize our products or technology. Such a license may not be available on commercially reasonable terms or at all. Moreover, any such litigation or the threat thereof may adversely affect our reputation, our ability to form strategic alliances or sublicense our rights to collaborators, engage with scientific advisors or hire employees or consultants, each of which would have an adverse effect on our business, results of operations and financial condition.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
We have not yet registered certain of our trademarks in all of our potential markets, although we have registered DecisionDx, DecisionDx-UM and DecisionDx-Melanoma in the United States. Our current or future registered or unregistered trademarks or trade names may be challenged, infringed, circumvented or declared generic or descriptive determined to be infringing on other marks. We may not be able to protect our rights to these trademarks and trade names or may be forced to stop using these names, which we need for name recognition by potential partners or customers in our markets of interest. During trademark registration proceedings, we may receive rejections. Although we would be given an opportunity to respond to those rejections, we may be unable
40
to overcome such rejections. In addition, in the USPTO and in comparable agencies in many foreign jurisdictions, third parties are given an opportunity to oppose pending trademark applications and to seek to cancel registered trademarks. Opposition or cancellation proceedings may be filed against our trademarks, and our trademarks may not survive such proceedings. In addition, third parties have used trademarks similar and identical to our trademarks in foreign jurisdictions, and have filed or may in the future file for registration of such trademarks. If they succeed in registering or developing common law rights in such trademarks, and if we are not successful in challenging such third-party rights, we may not be able to use these trademarks to market our products in those countries. If we are unable to establish name recognition based on our trademarks and trade names, we may not be able to compete effectively and our business may be adversely affected. We may license our trademarks and trade names to third parties, such as distributors. Although these license agreements may provide guidelines for how our trademarks and trade names may be used, a breach of these agreements or misuse of our trademarks and tradenames by our licensees may jeopardize our rights in or diminish the goodwill associated with our trademarks and trade names.
We may be subject to claims challenging the inventorship of our patents and other intellectual property.
We or our licensors may be subject to claims that former employees, collaborators or other third parties have an interest in our owned or in-licensed patents, trade secrets or other intellectual property as an inventor or co-inventor. For example, we or our licensors may have inventorship disputes arise from conflicting obligations of employees, consultants or others who are involved in developing our products. Litigation may be necessary to defend against these and other claims challenging inventorship or our or our licensors’ ownership of our owned or in-licensed patents, trade secrets or other intellectual property. If we or our licensors fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights, such as exclusive ownership of, right to use, or right to exclude others from using, intellectual property that is important to our products. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations and prospects.
In addition, while it is our policy to require our employees and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own. Our assignment agreements may not be self-executing or may be breached, and we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property.
Obtaining and maintaining our patent protection depends on compliance with various required procedures, document submissions, fee payments and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance fees, renewal fees, annuity fees and various other governmental fees on patents and/or applications must be paid to the USPTO and various governmental patent agencies outside of the United States at several stages over the lifetime of the patents and/or applications. We have systems in place to remind us to pay these fees, and we employ an outside firm and rely on our outside counsel to pay these fees due to non-U.S. patent agencies. The USPTO and various non-U.S. governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. We employ reputable law firms and other professionals to help us comply, and in many cases, an inadvertent lapse can be cured by payment of a late fee or by other means in accordance with the applicable rules. However, there are situations in which non-compliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction, such as failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we, or our licensors, fail to maintain the patents and patent applications covering our products and technologies, potential competitors may be able to enter the market without infringing our patents and this circumstance would have a material adverse effect on our business.
Patent terms may be inadequate to protect our competitive position on our products for an adequate amount of time.
Patents have a limited lifespan, and the protection patents afford is limited. In the United States, if all maintenance fees are timely paid, the natural expiration of a patent is generally 20 years from its earliest U.S.
41
non-provisional filing date. Various extensions may be available, but the term of a patent, and the protection it affords, is limited. Even if patents covering our products are obtained, once the patent term has expired, we may be open to competition from competitive tests or products. Given the amount of time required for the development, testing and regulatory review of potential new tests or products, patents protecting such tests or products might expire before or shortly after such tests or products are commercialized. As a result, our owned and licensed patent portfolio may not provide us with sufficient rights to exclude others from commercializing tests or other products similar or identical to ours.
Risks Related to Employee Matters and Managing Growth and Other Risks Related to Our Business
We are highly dependent on the services of our key personnel.
We are highly dependent on the services of our key personnel, including Derek J. Maetzold, our President and Chief Executive Officer. Although we have entered into agreements with them regarding their employment, they are not for a specific term and each of may terminate their employment with us at any time, though we are not aware of any present intention of any of these individuals to leave us.
Our research and development programs and laboratory operations depend on our ability to attract and retain highly skilled scientists and technicians. We may not be able to attract or retain qualified scientists and technicians in the future due to the competition for qualified personnel among life science businesses, particularly near our sole laboratory facility located in Phoenix, Arizona. We also face competition from universities and public and private research institutions in recruiting and retaining highly qualified scientific personnel. We may have difficulties locating, recruiting or retaining qualified sales people. Recruiting and retention difficulties can limit our ability to support our research and development and sales programs. All of our employees are at-will, which means that either we or the employee may terminate their employment at any time.
Our employees, clinical investigators, consultants, speakers, vendors and any current or potential commercial partners may engage in misconduct or other improper activities, including non-compliance with regulatory standards and requirements and insider trading.
We are exposed to the risk of fraud or other misconduct by our employees, clinical study investigators, consultants, speakers, vendors and any potential commercial partners. Misconduct by these parties could include intentional, reckless and/or negligent conduct or disclosure of unauthorized activities to us that violates: federal laws and regulations or those of comparable foreign regulatory authorities, including those laws that require the reporting of true, complete and accurate information; manufacturing standards; federal and state health and data privacy, security, fraud and abuse, government price reporting, transparency reporting requirements, and other healthcare laws and regulations in the United States and abroad; sexual harassment and other workplace misconduct; or laws that require the true, complete and accurate reporting of financial information or data. Such misconduct could also involve the improper use of information obtained in the course of clinical studies, which could result in regulatory sanctions and cause serious harm to our reputation. We intend to adopt a code of conduct applicable to all of our employees prior to completion of this offering, as well as a disclosure program and other applicable policies and procedures, but it is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant civil, criminal and administrative penalties, damages, fines, disgorgement, individual imprisonment, exclusion from government funded healthcare programs, such as Medicare, Medicaid and other federal healthcare programs, contractual damages, reputational harm, diminished profits and future earnings, additional integrity reporting and oversight obligations, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
We may be unable to manage our future growth effectively, which could make it difficult to execute our business strategy.
We have experienced significant revenue growth in a short period of time. We may not achieve similar growth rates in future periods. You should not rely on our operating results for any prior periods as an indication of our future operating performance. To effectively manage our anticipated future growth, we must continue to maintain
42
and enhance our financial, accounting, laboratory operations, customer support and sales administration systems, processes and controls. Failure to effectively manage our anticipated growth could lead us to over-invest or under-invest in development, operational and administrative infrastructure, result in weaknesses in our infrastructure, systems, or internal controls, give rise to operational mistakes, losses, loss of customers, productivity or business opportunities, and result in loss of employees and reduced productivity of remaining employees.
We also anticipate further growth in our business operations. This future growth could create strain on our organizational, administrative and operational infrastructure, including laboratory operations, quality control, customer service and sales organization management. We expect to increase headcount and to hire more specialized personnel in the future as we grow our business. We will need to continue to hire, train and manage additional qualified scientists, laboratory personnel, client and account services personnel, and sales and marketing staff and improve and maintain our technology to properly manage our growth. If our new hires perform poorly, if we are unsuccessful in hiring, training, managing and integrating these new employees or if we are not successful in retaining our existing employees, our business may be harmed.
In addition, our anticipated growth could require significant capital expenditures and might divert financial resources from other projects such as the development of new diagnostic tests and services. As we commercialize additional diagnostic tests, we may need to incorporate new equipment, implement new technology systems, or hire new personnel with different qualifications. Failure to manage this growth or transition could result in turnaround time delays, higher costs, declining quality, deteriorating customer service, and slower responses to competitive challenges. A failure in any one of these areas could make it difficult for us to meet market expectations for our products, and could damage our reputation and the prospects for our business.
We may not be able to maintain the quality or expected turnaround times of our products, or satisfy customer demand as it grows. Our ability to manage our growth properly will require us to continue to improve our operational, financial and management controls, as well as our reporting systems and procedures. The time and resources required to implement these new systems and procedures is uncertain, and failure to complete this in a timely and efficient manner could adversely affect our operations. If our management is unable to effectively manage our anticipated growth, our expenses may increase more than expected, our revenue could decline or grow more slowly than expected and we may be unable to implement our business strategy. The quality of our products and services may suffer, which could negatively affect our reputation and harm our ability to retain and attract customers.
U.S. federal income tax reform could adversely affect us.
On December 22, 2017, the U.S. government enacted the TCJA that significantly revises the Internal Revenue Code of 1986, as amended, or the Code. The TCJA, among other things, contains significant changes to corporate taxation, including reduction of the corporate tax rate from a top marginal rate of 35% to a flat rate of 21%, limitation of the tax deduction for interest expense to 30% of adjusted earnings (except for certain small businesses), limitation of the deduction for net operating losses generated after December 31, 2017 to 80% of current year taxable income and elimination of net operating loss carrybacks, one time taxation of offshore earnings at reduced rates regardless of whether they are repatriated, elimination of U.S. tax on foreign earnings (subject to certain important exceptions), immediate deductions for certain new investments instead of deductions for depreciation expense over time, and modifying or repealing many business deductions and credits. We do not expect the TCJA to have a material impact to our current projection of minimal cash taxes for the near future. However, we continue to examine the impact that the TCJA may have on our business in the longer term. Accordingly, notwithstanding the reduction in the corporate income tax rate, the overall impact of the TCJA is uncertain and our business and financial condition could be adversely affected. In addition, it is uncertain if and to what extent various states will conform to the TCJA. The impact of the TCJA on holders of our common stock is also uncertain and could be adverse. We urge prospective investors to consult with their legal and tax advisors with respect to the TCJA and the potential tax consequences of investing in or holding our common stock.
Our ability to use net operating losses to offset future taxable income may be subject to limitations.
As of December 31, 2018, we had federal net operating loss carryforwards of approximately $62.2 million. These net operating loss carryforwards could expire unused and be unavailable to offset future income tax liabilities. Under the TCJA, federal net operating losses incurred in 2018 and in future years may be carried forward
43
indefinitely, but the deductibility of such federal net operating losses is limited. It is uncertain if and to what extent various states will conform to the TCJA. In addition, under Sections 382 and 383 of the Code, and corresponding provisions of state law, if a corporation undergoes an “ownership change” (which is generally defined as a greater than 50% change (by value) in its equity ownership over a three-year period), the corporation’s ability to use its pre-change net operating loss carryforwards and other pre-change tax attributes to offset its post-change income or taxes may be limited. We have experienced an ownership change in the past and we may also experience additional ownership changes in the future as a result of subsequent shifts in our stock ownership, some of which may be outside of our control. If an ownership change occurs and our ability to use our net operating loss carryforwards is materially limited, it would harm our future operating results by effectively increasing our future tax obligations.
Our business could be negatively impacted by cyber security threats.
We are increasingly dependent upon information technology systems, infrastructure and data to operate our business. In the ordinary course of business, we collect, store and transmit confidential information (including but not limited to intellectual property, proprietary business information and personal information). It is critical that we do so in a secure manner to maintain the confidentiality and integrity of such confidential information. We also have outsourced elements of our operations to third parties, and as a result we manage a number of third-party contractors who have access to our confidential information.
Despite the implementation of security measures, given their size and complexity and the increasing amounts of confidential information that they maintain, our internal information technology systems and those of our contractors and consultants are potentially vulnerable to breakdown or other damage or interruption from service interruptions, system malfunction, natural disasters, terrorism, war and telecommunication and electrical failures, as well as security breaches from inadvertent or intentional actions by our employees, contractors, consultants, business partners, and/or other third parties, or from cyber-attacks by malicious third parties (including the deployment of harmful malware, ransomware, denial-of-service attacks, social engineering and other means to affect service reliability and threaten the confidentiality, integrity and availability of information), which may compromise our system infrastructure or lead to data leakage. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and reputational damage and the further development and commercialization of our products could be delayed.
While we have not experienced any such system failure, accident or security breach to date, we cannot assure you that our data protection efforts and our investment in information technology will prevent significant breakdowns, data leakages, breaches in our systems or other cyber incidents that could have a material adverse effect upon our reputation, business, operations or financial condition. For example, we maintain a tumor specimen database comprised of over 38,000 samples some of which were used to develop and validate DecisionDx-Melanoma, some of which are currently being used to improve on the test and some of which will be used in the future. If we were to lose this database, our ability to further validate, improve and therefore maintain and grow sales of DecisionDx-Melanoma could be significant impaired.
Furthermore, significant disruptions of our internal information technology systems or security breaches could result in the loss, misappropriation, and/or unauthorized access, use, or disclosure of, or the prevention of access to, confidential information (including trade secrets or other intellectual property, proprietary business information, and personal information), which could result in financial, legal, business, and reputational harm to us. For example, any such event that leads to unauthorized access, use, or disclosure of personal information, including personal information related to our patient samples or employees, could harm our reputation directly, compel us to comply with federal and/or state breach notification laws and foreign law equivalents, subject us to mandatory corrective action, and otherwise subject us to liability under laws and regulations that protect the privacy and security of personal information, which could result in significant legal and financial exposure and reputational damages that could potentially have an adverse effect on our business.
Product or professional liability lawsuits against us could cause us to incur substantial liabilities and could limit our commercialization of our products.
We face an inherent risk of product and professional liability exposure related to our products. The marketing, sale and use of our products could lead to the filing of product liability claims were someone to allege that our
44
products identified or reported inaccurate or incomplete information, or otherwise failed to perform as designed. We may also be subject to liability for errors in, a misunderstanding of or inappropriate reliance upon, the information we provide in the ordinary course of our business activities.
If we cannot successfully defend ourselves against claims that our products caused injury or otherwise failed to function properly, we could incur substantial liabilities. Regardless of merit or eventual outcome, product liability claims may result in:
| • | decreased demand for our current tests any tests that we may develop, and the inability to commercialize such tests; |
| • | injury to our reputation and significant negative media attention; |
| • | reluctance of experts willing to conduct our clinical studies; |
| • | initiation of investigations by regulators; |
| • | significant costs to defend the related litigation and diversion of management’s time and our resources; |
| • | substantial monetary awards to study subjects or patients; |
| • | product recalls, withdrawals or labeling, or marketing or promotional restrictions; and |
| • | loss of revenue. |
We currently carry product liability insurance. However, the amount of this insurance may not be adequate to cover all liabilities that we may incur. Insurance coverage is increasingly expensive. We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise.
International expansion of our business exposes us to business, regulatory, political, operational, financial, and economic risks associated with doing business outside of the United States.
We currently do not accept orders from customers outside of the United States, but our long term business strategy incorporates potential international expansion. Doing business internationally involves a number of risks, including:
| • | multiple, conflicting and changing laws and regulations such as privacy regulations, tax laws, export and import restrictions, economic sanctions and embargoes, employment laws, regulatory requirements and other governmental approvals, permits and licenses; |
| • | limits in our ability to penetrate international markets if we are not able to perform tests locally; |
| • | logistics and regulations associated with shipping tissue samples, including infrastructure conditions and transportation delays; |
| • | difficulties in staffing and managing foreign operations; |
| • | failure to obtain regulatory approvals for the commercialization of our products in various countries; |
| • | complexities and difficulties in obtaining intellectual property protection and enforcing our intellectual property; |
| • | complexities associated with managing multiple payor reimbursement regimes, government payors, or patient self-pay systems; |
| • | financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the impact of local and regional financial crises on demand and payment for our products and exposure to foreign currency exchange rate fluctuations; |
| • | natural disasters, political and economic instability, including wars, terrorism, and political unrest, outbreak of disease, boycotts, curtailment of trade and other business restrictions; and |
| • | regulatory and compliance risks that relate to maintaining accurate information and control over sales and distributors’ activities that may fall within the purview of the U.S. Foreign Corrupt Practices Act, or FCPA, its books and records provisions, or its anti-bribery provisions. |
Any of these factors could significantly harm our future international expansion and operations and, consequently, our revenue and results of operations.
45
Requirements associated with being a public company will increase our costs significantly, as well as divert significant company resources and management attention.
After the completion of this offering, we will be subject to the reporting requirements of the Securities Exchange Act of 1934, as amended, or the Exchange Act, or the other rules and regulations of the SEC or any securities exchange relating to public companies. Sarbanes-Oxley, as well as rules subsequently adopted by the SEC, and The Nasdaq Stock Market, or Nasdaq, to implement provisions of Sarbanes-Oxley, impose significant requirements on public companies, including requiring establishment and maintenance of effective disclosure and financial controls and changes in corporate governance practices. Further, pursuant to the Dodd-Frank Wall Street Reform and Consumer Protection Act of 2010, the SEC has adopted additional rules and regulations in these areas, such as mandatory “say on pay” voting requirements that will apply to us when we cease to be an emerging growth company. Stockholder activism, the current political environment and the current high level of government intervention and regulatory reform may lead to substantial new regulations and disclosure obligations, which may lead to additional compliance costs and impact the manner in which we operate our business in ways we cannot currently anticipate. Compliance with the various reporting and other requirements applicable to public companies requires considerable time and attention of management. We cannot assure you that we will satisfy our obligations as a public company on a timely basis.
We expect the rules and regulations applicable to public companies to substantially increase our legal and financial compliance costs and to make some activities more time-consuming and costly. If these requirements divert the attention of our management and personnel from other business concerns, they could have a material adverse effect on our business, financial condition and results of operations. The increased costs will decrease our net income or increase our net loss, and may require us to reduce costs in other areas of our business or increase the prices of our products. In addition, as a public company, it may be more difficult or more costly for us to obtain certain types of insurance, including directors’ and officers’ liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. The impact of these events could also make it more difficult for us to attract and retain qualified personnel to serve on our board of directors, our board committees or as executive officers.
If we fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have a material adverse effect on the success of our business.
We, and the third parties with whom we share our facilities, are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Each of our operations involve the use of hazardous and flammable materials, including chemicals and biological and radioactive materials. Our operations also produce hazardous waste. We generally contract with third parties for the disposal of these materials and wastes. We cannot eliminate the risk of contamination or injury from these materials. We could be held liable for any resulting damages in the event of contamination or injury resulting from the use of hazardous materials by us or the third parties with whom we share our facilities, and any liability could exceed our resources. We also could incur significant costs associated with civil or criminal fines and penalties.
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials.
In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research and development. Failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions.
Risks Related to this Offering and Ownership of Our Common Stock
There has been no prior public market for our common stock, the stock price of our common stock may be volatile or may decline regardless of our operating performance, and you may not be able to resell your shares at or above the initial public offering price.
There has been no public market for our common stock prior to this offering. The initial public offering price for our common stock will be determined through negotiations between the underwriters and us and may vary from
46
the market price of our common stock following this offering. If you purchase shares of our common stock in this offering, you may not be able to resell those shares at or above the initial public offering price. An active or liquid market in our common stock may not develop upon the completion of this offering or, if it does develop, it may not be sustainable.
The market price of our common stock may fluctuate significantly in response to numerous factors, many of which are beyond our control, including:
| • | our operating performance and the performance of other similar companies; |
| • | our success in marketing and selling our products; |
| • | reimbursement determinations by third-party payors and reimbursement rates for our products; |
| • | changes in our projected operating results that we provide to the public, our failure to meet these projections or changes in recommendations by securities analysts that elect to follow our common stock; |
| • | regulatory or legal developments in the United States and other countries; |
| • | the level of expenses related to product development and clinical studies for our products; |
| • | our ability to achieve product development goals in the timeframe we announce; |
| • | announcements of clinical study results, regulatory developments, acquisitions, strategic alliances or significant agreements by us or by our competitors; |
| • | the success or failure of our efforts to acquire, license or develop additional tests; |
| • | recruitment or departure of key personnel; |
| • | the economy as a whole and market conditions in our industry; |
| • | trading activity by a limited number of stockholders who together beneficially own a significant percentage of our outstanding common stock; |
| • | the expiration of market standoff or contractual lock-up agreements; |
| • | the size of our market float; and |
| • | any other factors discussed in this prospectus. |
In addition, the stock market in general, and diagnostic and life sciences companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our securities, regardless of our actual operating performance. If the market price of our securities after this offering does not exceed the initial public offering price, you may not realize any return on your investment in us and may lose some or all of your investment. In the past, stockholders have filed securities class action litigation following periods of market volatility. If we were to become involved in securities litigation, it could subject us to substantial costs, divert resources and the attention of management from our business and adversely affect our business.
Substantial amounts of our outstanding shares may be sold into the market when lock-up or market standoff periods end. If there are substantial sales of shares of our common stock, the price of our common stock could decline.
The price of our common stock could decline if there are substantial sales of our common stock, particularly sales by our directors, executive officers and significant stockholders, or if there is a large number of shares of our common stock available for sale and the market perceives that sales will occur. After this offering, we will have outstanding shares of our common stock, based on the number of shares outstanding as of March 31, 2019. All of the shares of common stock sold in this offering will be available for sale in the public market. All of our outstanding shares of common stock are currently restricted from resale as a result of market
47
standoff and lock-up agreements, as more fully described in “Shares Eligible for Future Sale.” These shares will become available to be sold 181 days after the date of this prospectus. Shares held by directors, executive officers and other affiliates will be subject to volume limitations under Rule 144 under the Securities Act of 1933, as amended, or the Securities Act.
After our initial public offering, certain of our stockholders will have rights, subject to some conditions, to require us to file registration statements covering their shares or to include their shares in registration statements that we may file for ourselves or our stockholders, subject to market standoff and lockup agreements. We also intend to register shares of common stock that we have issued and may issue under our employee equity incentive plans. Once we register these shares, they will be able to be sold freely in the public market upon issuance, subject to existing market standoff or lock-up agreements. SVB Leerink LLC and Robert W. Baird & Co. Incorporated may, in their discretion, permit our stockholders to sell shares prior to the expiration of the restrictive provisions contained in those lock-up agreements.
The market price of the shares of our common stock could decline as a result of the sale of a substantial number of our shares of common stock in the public market or the perception in the market that the holders of a large number of shares intend to sell their shares.
If you purchase shares of our common stock in this offering, you will experience substantial and immediate dilution.
If you purchase shares of our common stock in this offering, you will experience substantial and immediate dilution of $ per share, based on the difference between the initial public offering price of $ per share and the pro forma net tangible book value per share of the common stock you acquire, because the price that you pay will be substantially greater than the pro forma net tangible book value per share of the common stock that you acquire. This dilution is due in large part to the fact that our earlier investors paid substantially less than the initial public offering price when they purchased their shares of our capital stock. You will experience additional dilution upon exercise of options to purchase common stock under our equity incentive plans, upon vesting of options to purchase common stock under our equity incentive plans, if we issue restricted stock to our employees under our equity incentive plans or if we otherwise issue additional shares of our common stock.
We will have broad discretion in the use of the net proceeds of this offering and may not use them effectively or in ways that increase the value of our share price.
We will have broad discretion in the application of the net proceeds from this offering, including for any purposes described in the section titled “Use of Proceeds,” and you and other stockholders may disagree with how we spend or invest these proceeds. The failure by our management to apply these funds effectively could adversely affect our business and financial condition. Pending their use, we may invest the net proceeds from this offering in a manner that does not produce income or that loses value. These investments may not yield a favorable return to our investors.
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
The trading market for our common stock will depend in part on the research and reports that securities or industry analysts publish about us or our business. Securities and industry analysts do not currently, and may never, publish research on our company. If no or only very few securities analysts commence coverage of us, or if industry analysts cease coverage of us, the trading price for our common stock would be negatively affected. If one or more of the analysts who cover us downgrade our common stock or publish inaccurate or unfavorable research about our business, our common stock price would likely decline. If one or more of these analysts cease coverage of us or fail to publish reports on us regularly, demand for our common stock could decrease, which might cause our common stock price and trading volume to decline.
Our disclosure controls and procedures may not prevent or detect all errors or acts of fraud.
Upon the completion of this offering, we will become subject to the periodic reporting requirements of the Exchange Act. We designed our disclosure controls and procedures to reasonably assure that information we must disclose in reports we file or submit under the Exchange Act is accumulated and communicated to management,
48
and recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC. We believe that any disclosure controls and procedures or internal controls and procedures, no matter how well-conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met.
These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. For example, our directors or executive officers could inadvertently fail to disclose a new relationship or arrangement causing us to fail to make any related party transaction disclosures. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by an unauthorized override of the controls. Accordingly, because of the inherent limitations in our control system, misstatements due to error or fraud may occur and not be detected. In addition, we do not have a risk management program or processes or procedures for identifying and addressing risks to our business in other areas.
We are an emerging growth company and a smaller reporting company and we cannot be certain if the reduced reporting requirements applicable to emerging growth companies will make our common stock less attractive to investors.
We are an emerging growth company as defined in the JOBS ACT, and we intend to take advantage of some of the exemptions from reporting requirements that are applicable to other public companies that are not emerging growth companies, including:
| • | being permitted to provide only two years of audited financial statements, in addition to any required unaudited interim financial statements, with correspondingly reduced “Management’s Discussion and Analysis of Financial Condition and Results of Operations” disclosure; |
| • | not being required to comply with the auditor attestation requirements in the assessment of our internal control over financial reporting; |
| • | not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements; |
| • | reduced disclosure obligations regarding executive compensation; and |
| • | not being required to hold a non-binding advisory vote on executive compensation or obtain stockholder approval of any golden parachute payments not previously approved. |
In addition, as an emerging growth company the JOBS Act allows us to delay adoption of new or revised accounting pronouncements applicable to public companies until such pronouncements are made applicable to private companies. We have elected to use this extended transition period under the JOBS Act. We may take advantage of these reporting exemptions until we are no longer an emerging growth company. We will remain an emerging growth company until the earlier of (1) the last day of the fiscal year (a) following the fifth anniversary of the completion of this offering, (b) in which we have total annual gross revenue of at least $1.07 billion or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates exceeds $700 million as of the prior June 30th and (2) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period.
We are also a smaller reporting company as defined in the Exchange Act. We may continue to be a smaller reporting company even after we are no longer an emerging growth company. We may take advantage of certain of the scaled disclosures available to smaller reporting companies and will be able to take advantage of these scaled disclosures for so long as our voting and non-voting common stock held by non-affiliates is less than $250.0 million measured on the last business day of our second fiscal quarter, or our annual revenue is less than $100.0 million during the most recently completed fiscal year and our voting and non-voting common stock held by non-affiliates is less than $700.0 million measured on the last business day of our second fiscal quarter.
We cannot predict if investors will find our common stock less attractive because we will rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
49
We do not intend to pay dividends for the foreseeable future.
We have never declared nor paid cash dividends on our capital stock. We currently intend to retain any future earnings to finance the operation and expansion of our business, and we do not expect to declare or pay any dividends in the foreseeable future. In addition, the terms of the 2018 LSA precludes us from paying dividends without prior consent. Consequently, stockholders must rely on sales of their common stock after price appreciation, which may never occur, as the only way to realize any future gains on their investment.
The concentration of our stock ownership will likely limit your ability to influence corporate matters, including the ability to influence the outcome of director elections and other matters requiring stockholder approval.
Based upon shares outstanding as of June 14, 2019, our executive officers, directors and the holders of more than 5% of our outstanding common stock, in the aggregate, beneficially owned approximately % of our common stock, and upon the completion of this offering, that same group, in the aggregate, will beneficially own approximately % of our common stock. As a result, these stockholders, acting together, will have significant influence over all matters that require approval by our stockholders, including the election of directors and approval of significant corporate transactions. Corporate actions might be taken even if other stockholders, including those who purchase shares in this offering, oppose them. This concentration of ownership might also have the effect of delaying or preventing a change of control of our company that other stockholders may view as beneficial.
Delaware law and provisions in our amended and restated certificate of incorporation and amended and restated bylaws that will be in effect at the completion of this offering could make a merger, tender offer or proxy contest difficult, thereby depressing the trading price of our common stock.
Provisions of our amended and restated certificate of incorporation and amended and restated bylaws, which will become effective immediately prior to and upon the completion of this offering, respectively, may delay or discourage transactions involving an actual or potential change in our control or change in our management, including transactions in which stockholders might otherwise receive a premium for their shares or transactions that our stockholders might otherwise deem to be in their best interests. Therefore, these provisions could adversely affect the price of our common stock. Among other things, our amended and restated certificate of incorporation and amended and restated bylaws:
| • | permit our board of directors to issue up to 10,000,000 shares of preferred stock, with any rights, preferences and privileges as they may designate (including the right to approve an acquisition or other change in our control); |
| • | provide that the authorized number of directors may be changed only by resolution of the board of directors; |
| • | provide that the board of directors or any individual director may only be removed with cause and the affirmative vote of the holders of at least 66-2/3% of the voting power of all of our then outstanding common stock; |
| • | provide that all vacancies, including newly created directorships, may, except as otherwise required by law, be filled by the affirmative vote of a majority of directors then in office, even if less than a quorum; |
| • | divide our board of directors into three classes; |
| • | require that any action to be taken by our stockholders must be effected at a duly called annual or special meeting of stockholders and not be taken by written consent; |
| • | provide that stockholders seeking to present proposals before a meeting of stockholders or to nominate candidates for election as directors at a meeting of stockholders must provide notice in writing in a timely manner and also specify requirements as to the form and content of a stockholder’s notice; |
| • | do not provide for cumulative voting rights (therefore allowing the holders of a majority of the shares of common stock entitled to vote in any election of directors to elect all of the directors standing for election, if they should so choose); |
| • | provide that special meetings of our stockholders may be called only by the chairman of the board, our Chief Executive Officer or by the board of directors pursuant to a resolution adopted by a majority of the total number of authorized directors; and |
50
| • | provide that the Court of Chancery of the State of Delaware will be the sole and exclusive forum for the following types of actions or proceedings under Delaware statutory or common law: (i) any derivative action or proceeding brought on our behalf; (ii) any action or proceeding asserting a claim of breach of a fiduciary duty owed by any of our current or former directors, officers or other employees to us or our stockholders; (iii) any action or proceeding asserting a claim against us or any of our current or former directors, officers or other employees, arising out of or pursuant to any provision of the Delaware General Corporation Law, our certificate of incorporation or our bylaws; (iv) any action or proceeding to interpret, apply, enforce or determine the validity of our certificate of incorporation or our bylaws; (v) any action or proceeding as to which the Delaware General Corporation Law confers jurisdiction to the Court of Chancery of the State of Delaware; and (vi) any action asserting a claim against us or any of our directors, officers or other employees governed by the internal affairs doctrine, in all cases to the fullest extent permitted by law and subject to the court’s having personal jurisdiction over the indispensable parties named as defendants; provided these provisions of our amended and restated certificate of incorporation and amended and restated bylaws will apply to suits brought to enforce a duty or liability created by the Securities Act, but stockholders will not be deemed to have waived our compliance with the federal securities laws and the regulations thereunder; and provided these provisions of our amended and restated certificate of incorporation and amended and restated bylaws will not apply to suits brought to enforce a duty or liability created by the Exchange Act or any other claim for which the federal courts have exclusive jurisdiction. |
The amendment of any of these provisions, with the exception of the ability of our board of directors to issue shares of preferred stock and designate any rights, preferences and privileges thereto, would require approval by the holders of at least 66-2/3% of our then-outstanding common stock.
In addition, as a Delaware corporation, we are subject to Section 203 of the Delaware General Corporation Law. These provisions may prohibit large stockholders, in particular those owning 15% or more of our outstanding voting stock, from merging or combining with us for a certain period of time. A Delaware corporation may opt out of this provision by express provision in its original certificate of incorporation or by amendment to its certificate of incorporation or bylaws approved by its stockholders. However, we have not opted out of this provision.
These and other provisions in our amended and restated certificate of incorporation, amended and restated bylaws and Delaware law could make it more difficult for stockholders or potential acquirors to obtain control of our board of directors or initiate actions that are opposed by our then-current board of directors, including delay or impede a merger, tender offer or proxy contest involving our company. The existence of these provisions could negatively affect the price of our common stock and limit opportunities for you to realize value in a corporate transaction.
For information regarding these and other provisions, see “Description of Capital Stock.”
Our amended and restated certificate of incorporation will provide that the Court of Chancery of the State of Delaware is the exclusive forum for certain disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees.
Our amended and restated certificate of incorporation will provide that, to the fullest extent permitted by law and subject to the court’s having personal jurisdiction over the indispensable parties named as defendants, the Court of Chancery of the State of Delaware is the exclusive forum for the following types of actions or proceedings under Delaware statutory or common law: (i) any derivative action or proceeding brought on our behalf; (ii) any action or proceeding asserting a breach of fiduciary duty owed by any of our current or former directors, officers or other employees to us or our stockholders; (iii) any action or proceeding asserting a claim against us or any of our current or former directors, officers or other employees arising out of or pursuant to any provision of the Delaware General Corporation Law, our amended and restated certificate of incorporation or amended and restated bylaws; (iv) any action or proceeding to interpret, apply, enforce or determine the validity of our certificate of amended and restated incorporation or our amended and restated bylaws; (v) any action or proceeding as to which the Delaware General Corporation Law confers jurisdiction to the Court of Chancery of the State of Delaware; and (vi) any action asserting a claim against us or any of our directors, officers or other employees that is governed by the internal affairs doctrine; provided these provisions of our amended and restated certificate of incorporation and amended and restated bylaws will apply to suits brought to enforce a
51
duty or liability created by the Securities Act, but stockholders will not be deemed to have waived our compliance with the federal securities laws and the regulations thereunder; and provided, that, this provision would not apply to suits brought to enforce a duty or liability created by the Exchange Act, or any other claim for which the federal courts have exclusive jurisdiction. These choice of forum provisions may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees and may discourage these types of lawsuits. Furthermore, the enforceability of similar choice of forum provisions in other companies’ certificates of incorporation has been challenged in legal proceedings, and it is possible that a court could find these types of provisions to be inapplicable or unenforceable. If a court were to find the choice of forum provisions contained in our amended and restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions.
52
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements. The forward-looking statements are contained principally in the sections entitled “Prospectus Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business.” These statements relate to future events or to our future financial performance and involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. Forward-looking statements include, but are not limited to, statements about:
| • | estimates of our addressable market, future revenue, expenses, capital requirements and our needs for additional financing; |
| • | expectations with respect to reimbursement for our products, including third-party payor reimbursement and coverage decisions; |
| • | anticipated cost, timing and success of our products in development, and our plans to research, develop and commercialize new tests; |
| • | our ability to obtain funding for our operations, including funding necessary to complete the expansion of our operations and development of our product candidates; |
| • | the implementation of our business model and strategic plans for our products, technologies and businesses; |
| • | our ability to manage and grow our business by expanding our sales to existing customers or introducing our products to new customers; |
| • | our ability to develop and maintain sales and marketing capabilities; |
| • | regulatory developments in the United States and foreign countries; |
| • | the performance of our third-party suppliers; |
| • | the success of competing diagnostic products that are or become available; |
| • | our ability to attract and retain key personnel; |
| • | our expectations regarding the period during which we qualify as an emerging growth company under the JOBS Act; |
| • | our use of the proceeds from this offering; and |
| • | our expectations regarding our ability to obtain and maintain intellectual property protection for our products and our ability to operate our business without infringing on the intellectual property rights of others. |
In some cases, you can identify these statements by terms such as “anticipate,” “believe,” “could,” “estimate,” “expects,” “intend,” “may,” “plan,” “potential,” “project,” “should,” “will,” “would” or the negative of those terms, and similar expressions that convey uncertainty of future events or outcomes. These forward-looking statements reflect our management’s beliefs and views with respect to future events and are based on estimates and assumptions as of the date of this prospectus and are subject to risks and uncertainties. In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this prospectus, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements. We discuss many of the risks associated with the forward-looking statements in this prospectus in greater detail under the heading “Risk Factors.” Moreover, we operate in a very competitive and rapidly changing environment. New risks emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. Given these uncertainties, you should not place undue reliance on these forward-looking statements.
53
You should carefully read this prospectus and the documents that we reference in this prospectus and have filed as exhibits to the registration statement, of which this prospectus is a part, completely and with the understanding that our actual future results may be materially different from what we expect. We qualify all of the forward-looking statements in this prospectus by these cautionary statements.
Except as required by law, we assume no obligation to update these forward-looking statements publicly, or to update the reasons actual results could differ materially from those anticipated in any forward-looking statements, whether as a result of new information, future events or otherwise.
54
We estimate that we will receive net proceeds of approximately $ million (or approximately $ million if the underwriters’ option to purchase additional shares is exercised in full) from the sale of the shares of common stock offered by us in this offering, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
A $1.00 increase (decrease) in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would increase (decrease) the net proceeds to us from this offering by approximately $ million, assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. Similarly, a one million share increase (decrease) in the number of shares offered by us, as set forth on the cover page of this prospectus, would increase (decrease) the net proceeds to us by $ , assuming the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) remains the same, and after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us.
The principal purposes of this offering are to obtain additional capital to support our operations, to create a public market for our common stock and to facilitate our future access to the public equity markets. We anticipate that we will use the net proceeds of this offering as follows:
| • | approximately $17 million for selling and marketing activities, including expansion of our sales force to support the ongoing commercialization of our current products and future products; |
| • | approximately $17 million for research and development related to the continued support of our current products as well as the development of our product pipeline; and |
| • | the remainder for working capital and other general corporate purposes, including the additional costs associated with being a public company. |
We may also use a portion of the net proceeds from this offering to in-license, acquire, or invest in complementary businesses, technologies, products or assets. However, we have no current plans, commitments or obligations to do so.
We believe that the net proceeds from this offering together with our existing cash and cash equivalents and anticipated cash generated from sales of our products will be sufficient to fund our operating expenses through at least the next 24 months, although there can be no assurance in that regard.
Our expected use of net proceeds from this offering represents our current intentions based upon our present plans and business condition. As of the date of this prospectus, we cannot predict with certainty all of the particular uses for the net proceeds to be received upon the completion of this offering, or the amounts that we will actually spend on the uses set forth above. The amounts and timing of our actual use of the net proceeds will vary depending on numerous factors, including the progress, cost and results of development programs, our ability to obtain additional financing, and other factors described under “Risk Factors” in this prospectus, many of which are outside our control and are difficult to anticipate, as well as the amount of cash used in our operations. Therefore, our actual expenditures may differ materially from the estimates described above. Our management will have broad discretion in the application of the net proceeds, and investors will be relying on our judgment regarding the application of the net proceeds from this offering between the uses set forth above.
Pending their use, we may invest the net proceeds from this offering in short- and intermediate-term, interest-bearing obligations, investment-grade instruments, certificates of deposit or direct or guaranteed obligations of the U.S. government.
55
We have never declared or paid any cash dividends on our capital stock. We currently intend to retain all available funds and any future earnings to support our operations and finance the growth and development of our business. We do not intend to pay cash dividends on our common stock for the foreseeable future. Any future determination related to our dividend policy will be made at the discretion of our board of directors and will depend upon, among other factors, our results of operations, financial condition, capital requirements, contractual restrictions, business prospects and other factors our board of directors may deem relevant. In addition, the terms of the 2018 LSA with SVB and Oxford prohibit us from paying dividends on our common stock without prior consent.
56
The following table sets forth our cash and cash equivalents and our capitalization as of March 31, 2019 as follows:
| • | on an actual basis; |
| • | on a pro forma basis to reflect (1) the filing and effectiveness of our amended and restated certificate of incorporation immediately prior to the completion of this offering, (2) the conversion of all outstanding shares of our convertible preferred stock as of March 31, 2019 into 9,959,831 shares of our common stock and the resulting reclassification of the carrying value of the convertible preferred stock to permanent equity in connection with the completion of this offering, (3) the net exercise of certain outstanding warrants to purchase shares of our Series F redeemable convertible preferred stock for an aggregate of shares of common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) in connection with the completion of this offering, (4) the adjustment of outstanding warrants to purchase shares of our convertible preferred stock into warrants to purchase shares of our common stock and the resulting reclassification of our preferred stock warrant liability to additional paid-in-capital, a component of stockholders’ deficit, in connection with the completion of this offering and (5) the conversion of approximately $11.8 million of aggregate principal amount, plus accrued interest thereon, of convertible promissory notes which will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019; and |
| • | on a pro forma as adjusted basis to give further effect to our issuance and sale of shares of common stock in this offering at an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
The pro forma and pro forma as adjusted information below is illustrative only, and our cash and cash equivalents and capitalization following the completion of this offering will be adjusted based on the actual initial public offering price and other terms of this offering determined at pricing. You should read this information in conjunction with our financial statements and the related notes included in this prospectus and the “Management’s Discussion and Analysis of Financial Condition and Results of Operations” section and other financial information contained in this prospectus.
As of March 31, 2019 |
|||||||||
Actual |
Pro Forma |
Pro Forma as Adjusted(1) |
|||||||
(unaudited, in thousands, except share and per share data) |
|||||||||
Cash and cash equivalents |
$ | 16,231 | $ | $ | |||||
Convertible promissory notes(2) |
$ | 3,568 | $ | $ | |||||
Long-term debt |
23,632 | ||||||||
Preferred stock warrant liability |
1,181 | ||||||||
Convertible preferred stock Series C, $0.001 par value; 503,056 shares authorized, 503,056 shares issued and outstanding, actual; shares authorized and no shares issued and outstanding, pro forma and pro forma adjusted |
1,501 | ||||||||
Redeemable convertible preferred stock Series A, B, D, E-1, E-2, E-2A, E-3 and F, $0.001 par value; 11,846,877 shares authorized, 9,456,775 shares issued and outstanding, actual; shares authorized and no shares issued and outstanding, pro forma and pro forma as adjusted |
45,051 | ||||||||
Stockholders’ deficit: |
|||||||||
Common stock, $0.001 par value; 17,308,384 shares authorized, 2,336,463 shares issued and outstanding, actual; shares authorized, shares issued and outstanding, pro forma; shares authorized, shares issued and outstanding, pro forma as adjusted |
2 | ||||||||
Additional paid-in capital |
9,412 | ||||||||
Accumulated deficit |
(58,846 | ) |
|||||||
Total stockholders’ equity (deficit) |
(49,432 | ) |
|||||||
Total capitalization |
$ | 25,501 | $ | $ | |||||
57
| (1) | Each $1.00 increase (decrease) in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would increase (decrease) the pro forma as adjusted amount of each of cash and cash equivalents, additional paid-in capital, total capitalization and total stockholders’ equity by approximately $ , assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. Similarly, each increase (decrease) of one million shares in the number of shares offered by us at the assumed initial public offering price per share of $ (the midpoint of the price range set forth on the cover page of this prospectus) would increase (decrease) each of cash and cash equivalents, additional paid-in capital, total capitalization and total stockholders’ equity by approximately $ , after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
| (2) | Principal amount, less unamortized discounts and issuance costs, plus embedded derivative liability. See Note 7 to our unaudited interim condensed financial statements included elsewhere in this prospectus for additional information on the accounting treatment of the convertible promissory notes. |
A $1.00 increase in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would decrease the number of shares of our common stock issued on conversion of our convertible promissory notes (and therefore the number of shares to be outstanding after this offering) by shares. A $1.00 decrease in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would increase the number of shares of our common stock issued on conversion of our convertible promissory notes (and therefore the number of shares to be outstanding after this offering) by shares.
The number of shares of common stock shown as issued and outstanding after this offering is based on the number of shares of our common stock outstanding as of March 31, 2019, and excludes:
| • | 2,575,158 shares of common stock issuable upon the exercise of outstanding stock options as of March 31, 2019, at a weighted-average exercise price of $1.88 per share; |
| • | shares of common stock reserved for future issuance under the 2019 Plan, which will become effective upon the execution and delivery of the underwriting agreement for this offering (with such shares include new shares plus the number of shares (not to exceed shares) (i) that remain available for the issuance of awards under the 2018 Plan at the time the 2019 Plan becomes effective, and (ii) any shares underlying outstanding stock awards granted under the 2018 Plan and the 2008 Plan, that expire or are repurchased, forfeited, cancelled or withheld, as more fully described in the section titled “Executive and Director Compensation – Equity Incentive Plans”), as well as any automatic increases in the number of our common stock reserved for future issuance under the 2019 Plan; |
| • | shares of common stock reserved for future issuance under the ESPP, as well as any automatic increases in the number of our common stock reserved for future issuance under the ESPP, which will become effective upon the execution and delivery of the underwriting agreement for this offering; and |
| • | 137,935 shares of our common stock issuable upon the exercise of certain warrants outstanding as of March 31, 2019, at a weighted-average exercise price of $5.47 per share. |
58
If you invest in our common stock in this offering, your ownership interest will be immediately diluted to the extent of the difference between the initial public offering price per share of our common stock and the pro forma as adjusted net tangible book value per share of our common stock after this offering.
As of March 31, 2019, we had a historical net tangible book value (deficit) of $(50.0) million, or $(21.39) per share of common stock. Our historical net tangible book value (deficit) per share represents the amount of our total tangible assets less total liabilities and preferred stock, which is not included in our stockholders deficit, divided by the total number of shares of common stock outstanding at March 31, 2019.
After giving effect to: (i) the automatic conversion of all outstanding shares of our convertible preferred stock into 9,959,831 shares of our common stock and the resulting reclassification of the carrying value of the convertible preferred stock to permanent equity, in connection with the completion of this offering; (ii) the net exercise of certain outstanding warrants to purchase shares of our Series F redeemable convertible preferred stock for an aggregate of shares of common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) in connection with the completion of this offering; (iii) the adjustment of outstanding warrants to purchase shares of our convertible preferred stock into warrants to purchase shares of our common stock and the resulting reclassification of our preferred stock warrant liability to additional paid-in-capital, a component of stockholders’ deficit, in connection with the completion of this offering; and (iv) the conversion of approximately $11.8 million of aggregate principal amount, plus accrued interest thereon, of convertible promissory notes which will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019; our pro forma net tangible book value (deficit) as of March 31, 2019 is $ million, or $ per share of our common stock.
Net tangible book value dilution per share to new investors represents the difference between the amount per share paid by purchasers of shares of common stock in this offering and the pro forma as adjusted net tangible book value per share of common stock immediately after completion of this offering. After giving further effect to the sale of shares of common stock that we are offering at the initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, our pro forma as adjusted net tangible book value as of March 31, 2019 is $ million, or approximately $ per share. This amount represents an immediate increase in pro forma net tangible book value of $ per share to our existing stockholders and an immediate dilution in pro forma net tangible book value of approximately $ per share to new investors participating in this offering.
Dilution per share to new investors is determined by subtracting pro forma as adjusted net tangible book value per share after this offering from the assumed initial public offering price per share paid by new investors. The following table illustrates this dilution:
Assumed initial public offering price per share |
$ | |||||
Historical net tangible book value (deficit) per share as of March 31, 2019, before giving effect to this offering |
$ | (21.39 | ) |
|||
Pro forma increase in historical net tangible book value (deficit) per share as of March 31, 2019 attributable to conversion of all outstanding shares of convertible preferred stock, net exercise of certain warrants and conversion of convertible promissory notes as described in the preceding paragraph |
||||||
Pro forma net tangible book value per share as of March 31, 2019, before giving effect to this offering |
$ | |||||
Increase in pro forma net tangible book value per share attributable to investors participating in this offering |
||||||
Pro forma as adjusted net tangible book value per share after this offering |
||||||
Dilution per share to new investors participating in this offering |
$ |
59
Each $1.00 increase (decrease) in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would increase (decrease) the pro forma as adjusted net tangible book value per share after this offering by approximately $ , and dilution in pro forma net tangible book value per share to new investors by approximately $ , assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and the estimated offering expenses payable by us.
An increase of one million shares in the number of shares offered by us, as set forth on the cover page of this prospectus, would increase our pro forma as adjusted net tangible book value per share after this offering by approximately $ and decrease the dilution to investors participating in this offering by approximately $ per share, assuming that the assumed initial public offering price remains the same, and after deducting the estimated underwriting discounts and commissions and the estimated offering expenses payable by us. Similarly, a decrease of one million shares in the number of shares offered by us, as set forth on the cover page of this prospectus, would decrease the pro forma as adjusted net tangible book value per share after this offering by approximately $ and increase the dilution to investors participating in this offering by approximately $ per share, assuming that the assumed initial public offering price remains the same, and after deducting the estimated underwriting discounts and commissions and the estimated offering expenses payable by us.
If the underwriters exercise their option to purchase additional shares of our common stock in full in this offering, the pro forma as adjusted net tangible book value after the offering would be $ per share, the increase in pro forma as adjusted net tangible book value per share to existing stockholders would be $ per share and the dilution per share to new investors would be $ per share, in each case assuming an initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus).
To the extent that outstanding options with an exercise price per share that is less than the pro forma as adjusted net tangible book value per share are exercised, new investors will experience further dilution. In addition, we may choose to raise additional capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the issuance of these securities could result in further dilution to our stockholders.
The following table summarizes on a pro forma as adjusted basis as of March 31, 2019, the number of shares of common stock purchased or to be purchased from us, the total consideration paid or to be paid to us in cash and the average price per share paid by existing stockholders for shares issued prior to this offering and the price to be paid by new investors in this offering. The calculation below is based on the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), before deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. As the table below shows, investors participating in this offering will pay an average price per share substantially higher than our existing stockholders paid.
Shares Purchased |
Total Consideration |
Average Price Per Share |
|||||||||||||
Number |
Percent |
Amount |
Percent |
||||||||||||
Existing stockholders |
$ | $ | |||||||||||||
Investors participating in this offering |
|||||||||||||||
Total |
100.0 | % |
100.0 | % |
|||||||||||
Each $1.00 increase (decrease) in the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) would increase (decrease) total consideration paid by new investors, total consideration paid by all stockholders and the average price per share paid by all stockholders by $ million, $ million and $ , respectively, assuming the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
Similarly, a one million share increase (decrease) in the number of shares offered by us, as set forth on the cover page of this prospectus, would increase (decrease) the total consideration paid by investors participating in this offering, total consideration paid by all stockholders and the average price per share paid by all stockholders by
60
approximately $ million, $ million and $ , respectively, assuming the assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) remains the same, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
The foregoing tables and calculations exclude:
| • | 2,575,158 shares of common stock issuable upon the exercise of outstanding stock options as of March 31, 2019, at a weighted-average exercise price of $1.88 per share; |
| • | shares of common stock reserved for future issuance under the 2019 Plan, which will become effective upon the execution and delivery of the underwriting agreement for this offering (with such shares include new shares plus the number of shares (not to exceed shares) (i) that remain available for the issuance of awards under the 2018 Plan at the time the 2019 Plan becomes effective, and (ii) any shares underlying outstanding stock awards granted under the 2018 Plan and the 2008 Plan that expire or are repurchased, forfeited, cancelled or withheld, as more fully described in the section titled “Executive and Director Compensation – Equity Incentive Plans”), as well as any automatic increases in the number of our common stock reserved for future issuance under the 2019 Plan; |
| • | shares of common stock reserved for future issuance under the ESPP, as well as any automatic increases in the number of our common stock reserved for future issuance under the ESPP, which will become effective upon the execution and delivery of the underwriting agreement for this offering; and |
| • | 137,935 shares of our common stock issuable upon the exercise of certain warrants outstanding as of March 31, 2019, at a weighted-average exercise price of $5.47 per share. |
We may choose to raise additional capital through the sale of equity or convertible debt securities due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. To the extent we issue additional shares of common stock or other equity or convertible debt securities in the future, there will be further dilution to investors participating in this offering.
61
The following tables set forth our selected financial data for the periods indicated. The following selected statements of operations and comprehensive loss data for the years ended December 31, 2017 and 2018 and the balance sheet data as of December 31, 2017 and 2018 are derived from our audited financial statements appearing elsewhere in this prospectus. The following selected statements of operations and comprehensive loss data for the three months ended March 31, 2018 and 2019 and the balance sheet data as of March 31, 2019 are derived from our unaudited interim condensed financial statements appearing elsewhere in this prospectus. This selected financial data should be read together with our financial statements and related notes and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” appearing elsewhere in this prospectus. This selected financial data in this section are not intended to replace our financial statements and related notes. Our historical results are not necessarily indicative of our future results and our operating results for the interim periods are not necessarily indicative of the results that may be expected for any other interim periods or any future year.
Years Ended December 31, |
Three Months Ended March 31, |
|||||||||||
2017 |
2018 |
2018 |
2019 |
|||||||||
(unaudited) |
||||||||||||
(in thousands, except share and per share data) |
||||||||||||
Statements of Operations and Comprehensive Loss Data: |
||||||||||||
Net revenues |
$ | 13,754 | $ | 22,786 | $ | 3,659 | $ | 8,717 | ||||
Cost of sales |
4,922 | 5,297 | 1,253 | 1,598 | ||||||||
Gross margin |
8,832 | 17,489 | 2,406 | 7,119 | ||||||||
Operating expenses: |
||||||||||||
Research and development |
4,473 | 4,854 | 1,263 | 1,394 | ||||||||
Selling, general and administrative |
15,259 | 16,470 | 4,228 | 6,047 | ||||||||
Total operating expenses |
19,732 | 21,324 | 5,491 | 7,441 | ||||||||
Operating loss |
(10,900 | ) |
(3,835 | ) |
(3,085 | ) |
(322 | ) |
||||
Interest income |
26 | 24 | 5 | 21 | ||||||||
Interest expense |
(1,649 | ) |
(2,275 | ) |
(529 | ) |
(1,024 | ) |
||||
Other income (expense), net |
163 | (272 | ) |
(21 | ) |
(33 | ) |
|||||
Loss before income taxes |
(12,360 | ) |
(6,358 | ) |
(3,630 | ) |
(1,358 | ) |
||||
Income tax expense |
10 | 9 | — | — | ||||||||
Net loss and comprehensive loss |
(12,370 | ) |
(6,367 | ) |
(3,630 | ) |
(1,358 | ) |
||||
Convertible preferred stock cumulative dividends |
2,897 | 3,577 | 810 | 928 | ||||||||
Accretion of redeemable convertible preferred stock to redemption value |
41 | 219 | 49 | 56 | ||||||||
Net loss and comprehensive loss attributable to common stockholders |
$ | (15,308 | ) |
$ | (10,163 | ) |
$ | (4,489 | ) |
$ | (2,342 | ) |
Loss per share attributable to common stockholders, basic and diluted(1) |
$ | (6.62 | ) |
$ | (4.37 | ) |
$ | (1.94 | ) |
$ | (1.00 | ) |
Weighted-average shares outstanding, basic and diluted(1) |
2,310,640 | 2,323,197 | 2,312,515 | 2,336,236 | ||||||||
Pro forma loss per share, basic and diluted (unaudited)(1)(2) |
$ | $ | ||||||||||
Pro forma weighted-average shares outstanding, basic and diluted (unaudited)(1)(2) |
||||||||||||
62
| (1) | See Note 3 to our financial statements appearing elsewhere in this prospectus for an explanation of the method used to calculate the basic and diluted loss per share and the number of shares used in the computation of the per share amounts. |
| (2) | The calculations for the unaudited pro forma loss per share, basic and diluted, assume the conversion of all our outstanding shares of convertible preferred stock into shares of our common stock and the net exercise of certain outstanding warrants to purchase shares of our convertible preferred stock for common stock as if the conversion or net exercise had occurred at the beginning of the period presented or the issuance date, if later, upon completion of this offering. For the three months ended March 31, 2019, the calculations further assume the conversion into shares of common stock all outstanding principal and accrued interest related to the convertible promissory notes as if the conversion had occurred at the respective issuance dates of the convertible promissory notes. Because the convertible promissory notes were not issued until after December 31, 2018, the assumed conversion of such notes into shares of common stock had no impact on the calculations for the year ended December 31, 2018. |
December 31, |
March 31. |
||||||||
2017 |
2018 |
2019 |
|||||||
(unaudited) |
|||||||||
(in thousands) |
|||||||||
Balance Sheet Data: |
|||||||||
Cash and cash equivalents |
$ | 1,212 | $ | 4,479 | $ | 16,231 | |||
Working capital(1) |
$ | 1,464 | $ | 11,389 | $ | 18,035 | |||
Total assets |
$ | 9,820 | $ | 22,405 | $ | 30,966 | |||
Convertible promissory notes(2) |
$ | — | $ | — | $ | 3,568 | |||
Long-term debt |
$ | 17,603 | $ | 24,500 | $ | 23,632 | |||
Preferred stock warrant liability |
$ | 525 | $ | 1,194 | $ | 1,181 | |||
Convertible preferred stock |
$ | 1,501 | $ | 1,501 | $ | 1,501 | |||
Redeemable convertible preferred stock |
$ | 34,538 | $ | 44,995 | $ | 45,051 | |||
Total stockholders’ deficit |
$ | (50,311 | ) |
$ | (56,565 | ) |
$ | (49,432 | ) |
| (1) | We define working capital as current assets minus current liabilities. See our financial statements and related notes included elsewhere in this prospectus for further details regarding our current assets and liabilities. |
| (2) | Principal amount, less unamortized discounts and issuance costs, plus embedded derivative liability. See Note 7 to our unaudited interim condensed financial statements included elsewhere in this prospectus for additional information on the accounting treatment of the convertible promissory notes. |
63
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
You should read the following discussion and analysis of financial condition and results of operations together with the section titled “Selected Financial Data” and our financial statements and related notes included elsewhere in this prospectus. This discussion and other parts of this prospectus contain forward-looking statements that involve risk and uncertainties, such as statements of our plans, objectives, expectations and intentions. Our actual results could differ materially from those discussed in these forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to, those discussed in the section titled “Risk Factors.”
Overview
We are a commercial-stage dermatological cancer company focused on providing physicians and their patients with personalized, clinically actionable genomic information to make more accurate treatment decisions. We believe that the traditional approach to developing a treatment plan for certain cancers using clinical and pathology factors alone is inadequate and can be improved by incorporating personalized genomic information. Our non-invasive products utilize proprietary algorithms to provide an assessment of a patient’s specific risk of metastasis or recurrence of their cancer, allowing physicians to identify patients who are likely to benefit from an escalation of care as well as those who may avoid unnecessary medical and surgical interventions. Our lead product, DecisionDx-Melanoma, is a proprietary GEP test that predicts the risk of metastasis or recurrence for patients diagnosed with invasive cutaneous melanoma, a deadly skin cancer. We estimate more than 100,000 patients are diagnosed with invasive cutaneous melanoma each year in the United States. We launched DecisionDx-Melanoma in May 2013. We also market DecisionDx-UM, which is a proprietary GEP test that predicts the risk of metastasis for patients with uveal melanoma, a rare eye cancer. We launched DecisionDx-UM in January 2010. Based on the substantial clinical evidence that we have developed, we have received Medicare coverage for both of our products, which represents approximately 50% of our addressable patient population. We also have two late-stage proprietary products in development that address SCC and suspicious pigmented lesions which are indications with high clinical need in dermatological cancer. These indications are areas of high clinical need in dermatological cancer and, together, represent an additional addressable market of approximately 500,000 patients in the United States.
We have processed over 40,000 clinical samples since commercial launch and our annual revenue increased from $13.8 million in 2017 to $22.8 million in 2018. New ordering clinicians (first time in ordering a test) grew by 9% for the three months ended March 31, 2019 compared to the three months ended March 31, 2018.
The numbers of DecisionDx-Melanoma and DecisionDx-UM test reports delivered by us since 2017 are presented in the table below:
DecisionDx- Melanoma |
DecisionDx-UM |
Total |
|||||||
Q1 2017 |
2,012 | 318 | 2,330 | ||||||
Q2 2017 |
2,193 | 328 | 2,521 | ||||||
Q3 2017 |
2,561 | 305 | 2,866 | ||||||
Q4 2017 |
2,534 | 363 | 2,897 | ||||||
Year Ended December 31, 2017 |
9,300 | 1,314 | 10,614 | ||||||
Q1 2018 |
2,727 | 322 | 3,049 | ||||||
Q2 2018 |
2,899 | 382 | 3,281 | ||||||
Q3 2018 |
3,136 | 324 | 3,460 | ||||||
Q4 2018 |
3,270 | 385 | 3,655 | ||||||
Year Ended December 31, 2018 |
12,032 | 1,413 | 13,445 | ||||||
Q1 2019 |
3,232 | 360 | 3,592 | ||||||
64
Since our inception in 2008, we have devoted substantially all of our resources to organizing and staffing our company, business planning, raising capital, discovering product candidates, conducting clinical study activities to generate evidence demonstrating the clinical validity, clinical utility, economic benefits, and patient outcomes of our products, and commercialization activities for those products. We currently market two proprietary GEP products and generate substantially all of our revenue from those activities. To date, we have financed our operations primarily through private placements of preferred stock, revenue generated from sales of our products, bank debt and convertible notes.
The principal focus of our current commercial efforts is to distribute our molecular diagnostic testing products through our direct sales force. The number of test reports we generate is a key indicator that we use to assess our business. A test report is generated when we receive a sample in our laboratory, the relevant information relating to that test is entered into our Laboratory Information Management System, the genomic profile of the sample is performed and a report providing the results of that profile is sent to the physician who ordered the test.
We bill third-party payors and patients for the tests we perform. The majority of our revenue collections is paid by third-party insurers, including Medicare. We have received LCDs, which provide coverage for our tests that meet certain criteria for Medicare and Medicare Advantage beneficiaries, representing approximately 60 million covered lives. As it relates to DecisionDx-UM, we have contracts or have received positive medical policy decisions from additional payors representing approximately 83 million covered lives. A “covered life” means a subscriber, or a dependent of a subscriber, who is insured under an insurance policy for such an insurance carrier.
On May 17, 2019, CMS determined that DecisionDx-UM meets the criteria for “existing ADLT” status. This means that beginning in 2021, the DecisionDx-UM Medicare rate will be set annually based upon the median private payor rate for the first half of the preceding calendar year. Specifically, the median private payor rate from January 1 to June 30, 2019 will be used to set the Medicare rate for the calendar year 2021. From May 17, 2019 through December 31, 2020, our rate will be set by Noridian, our local MAC. Also, on May 17, 2019, CMS determined that DecisionDx-Melanoma meets the criteria for “new ADLT” status. This means that from July 1, 2019 through March 31, 2020 the Medicare reimbursement rate will equal the initial list price of $7,193.00. The rate for April 1, 2020 through December 31, 2020 will be calculated based upon the median private payor rate from July 1, 2019 to November 30, 2019. Accordingly, beginning in 2021, the rate will be set annually based upon the median private payor rate for the first half of the preceding calendar year. If CMS determines the list charge amount for DecisionDx-Melanoma was greater than 130% of the weighted median of private payor rates, CMS will recoup from us the difference between the actual list charge and 130% of the weighted median.
Since our inception, we have incurred significant operating losses and negative cash flows. For the year ended December 31, 2018, our net loss was $6.4 million, our net cash used in operating activities was $12.3 million and we had an accumulated deficit of $57.5 million as of December 31, 2018. For the three months ended March 31, 2019, our net loss was $1.4 million, our net cash provided by operating activities was $1.3 million and we had an accumulated deficit of $58.8 million as of March 31, 2019. We also have substantial indebtedness, the terms of which require us to meet a monthly three-month trailing revenue covenant. Although we were in compliance with this covenant as of December 31, 2018 and the most recently tested month prior to the date of this prospectus, management’s projections, including consideration of certain revenue recognition policies, previously indicated potential non-compliance with the revenue covenant during the next 12 months from the date of issuance of our annual financial statements. For these reasons, the report of our independent registered public accounting firm on our financial statements as of December 31, 2017 and 2018 and for each of the years then ended includes an explanatory paragraph indicating that there is substantial doubt about our ability to continue as a going concern. Subsequently, in June 2019, we entered into an amendment to the 2018 LSA which, among other changes, modified the revenue covenant. As a result, management now expects to be in compliance with the amended covenant for at least the next 12 months.
Our ability to generate revenue sufficient to achieve profitability will depend heavily on the successful commercialization of our currently marketed products and the products we plan to launch in the future. We anticipate that a substantial portion of our capital resources and efforts in the foreseeable future will be focused on the commercialization of our existing products, the development of our future product candidates, and the potential commercialization of our product candidates.
65
We expect to continue to incur significant expenses and increasing operating losses for at least the next two years. We believe that the net proceeds from this offering, together with our existing cash and cash equivalents and anticipated cash generated from sales of our products, will be sufficient to fund our operating expenses through at least the next 24 months. We have based these estimates on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we expect. Additionally, our estimates and assumptions could be adversely impacted by the uncertainty associated with our ability to continue as a going concern.
Our net losses may fluctuate significantly from period to period, depending on the timing of our planned development activities, the growth of our sales and marketing activities and the timing of revenue recognition under ASC 606. We expect our expenses will increase substantially over time as we:
| • | execute clinical studies to generate evidence supporting our current and future product candidates; |
| • | execute our commercialization strategy for our current and future products; |
| • | continue our ongoing and planned development of new products; |
| • | seek to discover and develop additional product candidates; |
| • | hire additional scientific and research and development staff; and |
| • | add additional operational, financial and management information systems and personnel. |
Furthermore, following the closing of this offering, we expect to incur additional costs associated with operating as a public company, including significant legal, accounting, investor relations and other expenses that we did not incur as a private company.
Factors affecting our performance
We believe there are several important factors that have impacted and that we expect will impact our operating performance and results of operations, including:
| • | Report volume. We believe that the number of reports we deliver to physicians is an important indicator of growth of adoption among the healthcare provider community. Our revenue and costs are affected by the volume of testing and mix of customers. Our performance depends on our ability to retain and broaden adoption with existing prescribing physicians, as well as attract new physicians. |
| • | Reimbursement. We believe that expanding reimbursement is an important indicator of the value of our products. Payors require extensive evidence of clinical utility, clinical validity, patient outcomes and health economic benefits in order to provide reimbursement for diagnostics products. Our revenue depends on our ability to demonstrate the value of our products to these payors. |
| • | Gross margin. We believe that our gross margin is an important indicator of the operating performance of our business. Higher gross margins reflect the average selling price of our tests, as well as the operating efficiency of our laboratory operations. |
| • | New product development. A significant aspect of our business is our investment in research and development activities, including activities related to the development of new products. In addition to the development of new product candidates, we believe these studies are critical to gaining physician adoption of new products and driving favorable coverage decisions by payors for such products. |
Components of the Results of Operations
Net Revenues
We generate revenues from the sale of our products, primarily from the sale of DecisionDx-Melanoma and DecisionDx-UM. We bill third-party payors and patients for the tests we perform.
We adopted the new revenue recognition guidance, ASC 606, on January 1, 2018 using the full retrospective method and adjusted the comparative reporting period for the year ended December 31, 2017. Under ASC 606, we recognize revenue at the amount we expect to be entitled, subject to a constraint for variable consideration, in the period in which our tests are delivered to the treating physicians. We have determined that our contracts contain variable consideration under ASC 606 because the amounts paid by third-party payors may be paid at
66
less than our standard rates or not paid at all, with such differences considered implicit price concessions. Variable consideration is recognized only to the extent it is probable that a significant reversal of revenue will not occur in future periods when the uncertainties are resolved. We consider variable consideration to be fully constrained (and therefore not recognized) for Medicare claims when the payment of such claims is subject to approval by an ALJ at an appeal hearing, due to the level of uncertainty and timing of the outcome. Variable consideration is evaluated each reporting period and adjustments are recorded as increases or decreases in revenues. Importantly, we expect to recognize any revenue adjudicated by the ALJ in the reporting period in which we are notified of the ALJ hearing outcome. Due to ALJ hearings, potential future changes in insurance coverage policies, contractual rates, and other trends in the reimbursement of our tests, our revenues may fluctuate significantly from period to period.
Our ability to increase our revenues will depend on our ability to further penetrate our target market, and, in particular, generate sales through our direct sales force, develop and commercialize additional tests, obtain reimbursement from additional third-party payors and increase our reimbursement rate for tests performed. In the near term, our financial performance will be highly dependent on reimbursement for DecisionDx-Melanoma. The use of DecisionDx-Melanoma is not yet broadly covered under positive coverage policies, although many third-party payors have begun to reimburse for this test.
Cost of Sales
The components of our cost of sales are material and service costs, personnel costs, which includes stock-based compensation expense, equipment and infrastructure expenses associated with testing samples, electronic medical records, order and delivery systems, shipping charges to transport samples, third-party test fees, and allocated overhead including rent, information technology costs, equipment depreciation and utilities. Costs associated with performing tests are recorded when the test is accessioned regardless of whether and when revenues are recognized with respect to that test. As a result, our cost of sales as a percentage of revenues may vary significantly from period to period because we do not recognize all revenues in the period in which the associated costs are incurred. We expect cost of sales in absolute dollars to increase as the number of tests we perform increases.
Gross margin, calculated as net revenue minus cost of sales, is a key indicator we use to assess our business. For example, in 2016, we transitioned the performance of our genomic tests to a proprietary dedicated clinical lab from a third-party contract lab. This transition increased the gross margin from our genomic tests while also increasing our level of control of the performance of our tests and laboratory quality metrics.
Research and Development
Research and development expenses include costs incurred to develop our genomic tests, collect clinical samples and conduct clinical studies to develop and support our products. These costs consist of personnel costs, including stock-based compensation expense, prototype materials, laboratory supplies, consulting costs, regulatory costs, electronic medical records set up costs, costs associated with setting up and conducting clinical studies and allocated overhead, including rent, information technology, equipment depreciation and utilities. We expense all research and development costs in the periods in which they are incurred. We expect our research and development expenses to increase in absolute dollars as we continue to invest in research and development activities related to developing enhanced and new products.
Selling, General and Administrative
Selling, general and administrative, or SG&A, expenses include executive, selling and marketing, legal, finance and accounting, human resources, billing and client services. These expenses consist of personnel costs, including stock-based compensation expense, direct marketing expenses, audit and legal expenses, consulting costs, training and medical education activities, payor outreach programs and allocated overhead, including rent, information technology, equipment depreciation, and utilities. In the near term, we expect increases in SG&A expenses related to compliance with the rules and regulations of the SEC and Nasdaq, investor relations activities, and additional insurance expenses. Other administrative and professional services expenses within SG&A are expected to increase with scale of our business, but selling and marketing-related expenses are expected to increase significantly, consistent with our growth strategy.
67
Interest Expense
Interest expense is attributable to borrowings under our term debt, revolving line of credit and convertible promissory notes, and also includes the amortization of debt discount and issuance costs.
Income Tax Expense
Our financial statements do not reflect any federal or state income tax benefits attributable to the net losses we have incurred, due to the uncertainty of realizing a benefit from those items. As of December 31, 2018, we had federal net operating loss carryforwards of $62.2 million, of which $48.3 million will begin to expire in 2028 if not utilized to offset taxable income, and $13.9 million may be carried forward indefinitely. Also, as of December 31, 2018, we had state net operating loss carryforwards of $60.8 million, which begin to expire in 2028 if not utilized to offset state taxable income.
Results of Operations
Comparison of the three months ended March 31, 2018 and 2019
The following table summarizes our results of operations for the periods indicated (in thousands, except percentages):
Three Months Ended March 31, |
Increase (Decrease) |
|||||||||||
2018 |
2019 |
|||||||||||
(unaudited) |
||||||||||||
Net revenue |
$ | 3,659 | $ | 8,717 | $ | 5,058 | 138.2 | % |
||||
Cost of sales |
1,253 | 1,598 | 345 | 27.5 | % |
|||||||
Gross margin |
2,406 | 7,119 | 4,713 | 195.9 | % |
|||||||
Operating expenses: |
||||||||||||
Research and development |
1,263 | 1,394 | 131 | 10.4 | % |
|||||||
Selling, general and administrative |
4,228 | 6,047 | 1,819 | 43.0 | % |
|||||||
Total operating expenses |
5,491 | 7,441 | 1,950 | 35.5 | % |
|||||||
Operating loss |
(3,085 | ) |
(322 | ) |
2,763 | 89.6 | % |
|||||
Interest income |
5 | 21 | 16 | 320.0 | % |
|||||||
Interest expense |
(529 | ) |
(1,024 | ) |
(495 | ) |
(93.6 | )% |
||||
Other income (expense), net |
(21 | ) |
(33 | ) |
(12 | ) |
(57.1 | )% |
||||
Loss before income taxes |
(3,630 | ) |
(1,358 | ) |
2,272 | 62.6 | % |
|||||
Income tax expense |
— | — | — | — | ||||||||
Net loss |
$ | (3,630 | ) |
$ | (1,358 | ) |
$ | 2,272 | 62.6 | % |
||
Net Revenues
Net revenues increased by $5.1 million, or 138.2%, due to a combination of increased test volume and higher per-unit revenues. Approximately 88% of the increase is attributable to DecisionDx-Melanoma test revenues with the remainder primarily attributable to DecisionDx-UM. For the three months ended March 31, 2019, we experienced an increase in overall test volumes of 17.8% and an increase in the average revenue per test of 102.2%, compared to the three months ended March 31, 2018. The increase in average revenue per test is primarily associated with the effect of the issuance of the final LCD for DecisionDx-Melanoma, issued by Palmetto and effective December 3, 2018. Coincident with the issuance of the LCD, a Medicare reimbursement rate for the test was established along with an agreement for payment of covered claims submitted for reimbursement beginning in February 2018. Note that as the Medicare LCD was not effective and the reimbursement rate was not known until the fourth quarter of 2018, the associated revenues were not recognized until the fourth quarter of 2018, which is when the criteria for revenue recognition was met. Medicare revenues for DecisionDx-Melanoma associated with test reports delivered in the first quarter of 2018, but not recognized in revenue until the fourth quarter of 2018, were $0.8 million. For each of the three months ended March 31, 2018 and 2019, we recorded positive revenue adjustments of $0.6 million related to tests delivered in previous periods. Additionally, for the three months ended March 31, 2019, revenues included $0.2 million for tests associated with contract research activities.
68
Cost of Sales
Cost of sales for the three months ended March 31, 2019 increased by $0.3 million compared to the three months ended March 31, 2018, primarily due to higher personnel costs due to additional headcount in our laboratory testing operations. As a percentage of net revenues, cost of sales was 34.2% for the three months ended March 31, 2018, compared to 18.3% for 2019, with the improvement principally a result of the increased operating leverage on the higher revenues. Due to the nature of our business, a significant portion of our cost of sales expenses represent fixed costs associated with our testing operations. Accordingly, our cost of sales expense will not necessarily increase or decrease commensurately with the change in net revenues from period to period.
Research and Development
Research and development expenses increased by $0.1 million, or 10.4%, for the three months ended March 31, 2019, compared to the three months ended March 31, 2018, primarily associated with increases in personnel costs, legal fees and other costs, partially offset by lower clinical trials expense.
Selling, General and Administrative
SG&A expense increased by $1.8 million, or 43.0%, for the three months ended March 31, 2019 compared to the three months ended March 31, 2018. Approximately 57% of the increase is attributable to higher personnel costs, particularly within our sales function due to headcount expansion. The remainder of the increase was primarily associated with higher professional fees and increased travel costs. The higher professional fees were primarily attributable to accounting and reimbursement services.
Interest Expense
Interest expense increased by $0.5 million, or 93.6%, for the three months ended March 31, 2019 compared to the three months ended March 31, 2018. The effect of the issuance of the convertible promissory notes in January and February 2019 added $0.3 million in interest expense for the three months ended March 31, 2019 and consisted of the accrual of the contractual 8% interest plus the amortization of issuance costs and debt discount. The remainder of the increase is due to a combination of higher average outstanding balances and higher interest rates on our variable-rate term debt and revolving line of credit under our banking credit facility. Average outstanding bank debt balances were approximately $5.6 million higher during the three months ended March 31, 2019 compared to the three months ended March 31, 2018 and average interest rates increased by approximately 1.0% from the same period in 2018.
Other Income (Expense), Net
Other income (expense), net, consists of the change in fair value of our liability for convertible preferred stock warrants and, beginning in 2019, the change in fair value of the embedded derivative liability associated with the convertible promissory notes. These liabilities are adjusted to their current fair values each period.
69
Comparison of the years ended December 31, 2017 and 2018
The following table summarizes our results of operations for the periods indicated (in thousands, except percentages):
Years Ended December 31, |
Increase (Decrease) |
|||||||||||
2017 |
2018 |
|||||||||||
Net revenue |
$ | 13,754 | $ | 22,786 | $ | 9,032 | 65.7 | % |
||||
Cost of sales |
4,922 | 5,297 | 375 | 7.6 | % |
|||||||
Gross margin |
8,832 | 17,489 | 8,657 | 98.0 | % |
|||||||
Operating expenses: |
||||||||||||
Research and development |
4,473 | 4,854 | 381 | 8.5 | % |
|||||||
Selling, general and administrative |
15,259 | 16,470 | 1,211 | 7.9 | % |
|||||||
Total operating expenses |
19,732 | 21,324 | 1,592 | 8.1 | % |
|||||||
Operating loss |
(10,900 | ) |
(3,835 | ) |
7,065 | 64.8 | % |
|||||
Interest income |
26 | 24 | (2 | ) |
(7.7 | )% |
||||||
Interest expense |
(1,649 | ) |
(2,275 | ) |
(626 | ) |
(38.0 | )% |
||||
Other income (expense), net |
163 | (272 | ) |
(435 | ) |
(266.9 | )% |
|||||
Loss before income taxes |
(12,360 | ) |
(6,358 | ) |
6,002 | 48.6 | % |
|||||
Income tax expense |
10 | 9 | (1 | ) |
(10.0 | )% |
||||||
Net loss |
$ | (12,370 | ) |
$ | (6,367 | ) |
$ | 6,003 | 48.5 | % |
||
Net Revenues
Net revenues increased $9.0 million, or 65.7%, due to a combination of increased test volume and higher per-unit revenues. Essentially all of the increase is attributable to DecisionDx-Melanoma. In 2018, we experienced an increase in overall test volumes of 26.7% and an increase in the average revenue per test of 30.8%, compared to 2017. The increase in average revenue per test is primarily related to the final LCD for DecisionDx-Melanoma, issued by Palmetto and effective December 3, 2018. Subsequent to the issuance of the LCD, a reimbursement rate for the test was established and an agreement to pay all covered claims submitted for reimbursement beginning in February 2018. Included in net revenues for the year ended December 31, 2018 were adjustments for variable consideration that unfavorably impacted net revenues by $0.9 million. There was no such amount for the year ended December 31, 2017 due to our election of a transition practical expedient in connection with the adoption of ASC 606 that permitted us to use the transaction price at the date the transaction was completed, rather than estimating variable consideration amounts, in the 2017 comparative reporting period.
Cost of Sales
Cost of sales for the year ended December 31, 2018 increased by $0.4 million compared to the year ended December 31, 2017, primarily due to higher personnel costs due to additional headcount in our laboratory testing operations. As a percentage of net revenues, cost of sales was 35.8% for the year ended December 31, 2017, compared to 23.2% for 2018, with the improvement principally a result of the increased leverage on the higher revenues. Due to the nature of our business, a significant portion of our cost of sales expenses represent fixed costs associated with our testing operations. Accordingly, our cost of sales expense will not necessarily increase or decrease commensurately with the change in net revenues from period to period.
Research and Development
Research and development expenses increased by $0.4 million, or 8.5%, for the year ended December 31, 2018, compared to the year ended December 31, 2017, primarily associated with increases in clinical trial activities and related expenses.
Selling, General and Administrative
SG&A expense increased by $1.2 million, or 7.9%, for the year ended December 31, 2018 compared to the year ended December 31, 2017. Approximately 50% of the increase is attributable to higher consulting and professional fees. The remainder of the increase was primarily associated with higher personnel costs, with the
70
balance attributable to several other minor variations. The higher consulting and professional fees were primarily attributable to accounting and reimbursement services. The higher personnel costs reflect increased headcount in administrative support personnel, partially offset by the effect of temporary headcount fluctuations in our sales and marketing function during the year.
Interest Expense
Interest expense increased by $0.6 million, or 38.0%, for the year ended December 31, 2018 compared to the year ended December 31, 2017. The increase is due to a combination of higher average outstanding debt balances and higher interest rates on our variable-rate term debt and revolving line of credit. Average outstanding debt balances were approximately $4.5 million higher in 2018 compared to 2017 and average interest rates increased by approximately 1.0%.
Other Income (Expense), Net
Other income (expense), net, in both periods consists of the change in fair value of our convertible preferred stock warrants, which are recorded as a liability on the balance sheet. The liability is adjusted to its current fair value each period.
Income Tax Expense
We recorded minimal income tax expense in both the years ended December 31, 2017 and 2018, because the income tax benefit of net loss in both periods was fully offset by changes in the valuation allowance on net deferred tax assets, as we have determined that it is more likely than not that these benefits will not be realized.
Liquidity and Capital Resources
Since our inception, we have incurred significant operating losses and negative cash flows. For the year ended December 31, 2018, our net loss was $6.4 million, our net cash used in operating activities was $12.3 million and we had an accumulated deficit of $57.5 million as of December 31, 2018. For the three months ended March 31, 2019, our net loss was $1.4 million, our net cash provided by operating activities was $1.3 million and we had an accumulated deficit of $58.8 million as of March 31, 2019. We also have substantial indebtedness, the terms of which require us to meet a monthly three-month trailing revenue covenant. Although we were in compliance with this covenant as of December 31, 2018 and the most recently tested month prior to the date of this prospectus, management’s projections, including consideration of certain revenue recognition policies, previously indicated potential non-compliance with the revenue covenant during the 12 months following the date of issuance of our annual financial statements. For these reasons, the report of our independent registered public accounting firm on our financial statements as of December 31, 2017 and 2018 and for each of the years then ended includes an explanatory paragraph indicating that there is substantial doubt about our ability to continue as a going concern. Subsequently, in June 2019, we entered into an amendment to our 2018 LSA which, among other changes, modified the revenue covenant. As a result, management now expects to be in compliance with the amended covenant for at least the next 12 months.
Our continuation as a going concern is dependent on our ability to obtain additional financing until we can generate sufficient cash flows from operations to meet our obligations. We intend to continue to seek additional debt or equity financing to continue our operations. There is no assurance that we will attain profitability or that additional debt or equity financing will be available to us.
As of March 31, 2019, we have raised aggregate cash proceeds of $46.6 million from the sale of our convertible preferred stock, in various private placements beginning in 2008, which we have used to fund our operations. In addition, we have obtained financing through term debt, a revolving line of credit and convertible promissory notes, which are discussed further below.
Funding Requirements
Our primary uses of capital are, and we expect will continue to be, compensation and related expenses, clinical research and development services, laboratory and related supplies, legal and other regulatory expenses and general administrative costs. We anticipate that a substantial portion of our capital resources and efforts in the foreseeable future will be focused on the commercialization of our existing products, the development of our future product candidates the potential commercialization of our product candidates, should their development be successful, and general administrative costs.
71
We have two product candidates in the late stage development that we plan to launch commercially in the second half of 2020. The successful development of other product candidates is highly uncertain. At this time, we cannot reasonably estimate or know the nature, timing and estimated costs of the efforts that will be necessary to complete the clinical development of all our product candidates. We are also unable to predict when, if ever, revenue will commence from sales of our product candidates. This is due to the numerous risks and uncertainties associated with developing genomic tests, including, among others, the uncertainty of:
| • | successful commencement and completion of clinical trials protocols; |
| • | successful identification and acquisition of tissue samples; |
| • | the development and validation of genomic classifiers; and |
| • | acceptance of new genomic tests by physicians, patients and third-party payors. |
A change in the outcome of any of these variables with respect to the development of any of our product candidates would significantly change the costs and timing associated with the development of that product candidate.
We will also incur costs as a public company that we have not previously incurred or have previously incurred at lower rates, including increased costs and expenses for fees to members of our board of directors, increased personnel costs, increased director and officer insurance premiums, audit and legal fees, investor relations fees and expenses for compliance with public-company reporting requirements under the Exchange Act and rules implemented by the SEC and Nasdaq.
As of December 31, 2018 and March 31, 2019, we had cash and cash equivalents of $4.5 million and $16.2 million, respectively. In the first quarter of 2019, we received proceeds of $11.8 million from the sale of convertible promissory notes. We believe that the net proceeds from this offering, together with our existing cash and cash equivalents and anticipated cash generated from sales of our products, will be sufficient to fund our operating expenses through at least the next 24 months.
We have based these estimates on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we expect. Because of the numerous risks and uncertainties associated with research, development and commercialization of product candidates, we are unable to estimate the exact amount of our working capital requirements. Our future funding requirements will depend on and could increase significantly as a result of many factors, including those listed above.
Until such time, if ever, as we can generate revenue sufficient to achieve profitability, we expect to finance our operations through a combination of equity and debt financings, which may not be available to us on the timing needed or on terms that we deem to be favorable. We do not currently have any committed external source of funds. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interest of our stockholders will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of common stockholders. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making acquisitions or capital expenditures or declaring dividends. If we are unable to raise additional funds through equity or debt financings or other arrangements when needed, we may be required to delay, limit, reduce or terminate our product discovery and development activities or future commercialization efforts.
72
Long-Term Debt
Our long-term debt consists of term debt and a revolving line of credit and are presented in the table below (in thousands):
As of December 31, |
As of March 31, |
||||||||
2017 |
2018 |
2019 |
|||||||
(unaudited) |
|||||||||
Term debt |
$ | 16,013 | $ | 21,350 | $ | 21,350 | |||
Revolving line of credit |
4,000 | 5,000 | 4,011 | ||||||
Total principal amount |
20,013 | 26,350 | 25,361 | ||||||
Unamortized discount and issuance costs |
(1,410 | ) |
(1,850 | ) |
(1,729 | ) |
|||
Total long-term debt |
18,603 | 24,500 | 23,632 | ||||||
Less: Current portion of long-term debt |
1,000 | — | — | ||||||
Long-term debt, less current portion |
$ | 17,603 | $ | 24,500 | $ | 23,632 | |||
The table above excludes our convertible promissory notes, which are classified as current liabilities in the balance sheet. See the “Convertible Promissory Notes” section below for details.
Term Debt
2017 Loan and Security Agreement
On March 31, 2017, we executed a loan and security agreement, or the 2017 LSA, with Oxford, as collateral agent, and SVB with Oxford and SVB as equal syndicated lenders, or the Lenders. The 2017 LSA provides a term loan of $15.0 million, or the 2017 Term Loan, and a credit line of up to $5.0 million (discussed in the “Revolving Line of Credit” section below) and granted to the Lenders a priority first lien security interest in substantially all of our assets. The 2017 Term Loan of $15.0 million was made on the effective date of the agreement and was used to repay all outstanding obligations with respect to our previous lending arrangement. The maturity date of the 2017 Term Loan was to be April 1, 2021, or the 2017 Term Loan Maturity Date.
The 2017 Term Loan bore interest at a floating per annum rate equal to the greater of 7.25% and 6.48% above the U.S. LIBOR rate and required the payment of interest only each month for the one-year period beginning with the date of initial funding of the loan. The applicable annual interest rate of the 2017 LSA on the effective date, and as of December 31, 2017, was 7.46% and 8.04%, respectively. Repayment of principal was to be made in 36 monthly installments of principal and interest beginning in the month following the one-year interest-only period; provided, that if we achieved a stipulated revenue target for the trailing 12-month period ending January 31, 2018, 30 fixed monthly installments, beginning in the month following an 18-month interest-only period. Additionally, the 2017 LSA also included an obligation to make an additional final payment of 6.25% (6.75% in the case the stipulated revenue target was met) of the aggregate original principal amount of the 2017 Term Loan upon any prepayment or on the 2017 Term Loan Maturity Date. As we achieved the stipulated revenue target, repayment of principal was to be made in 30 fixed monthly installments beginning in the month following the 18-month interest-only period (i.e., November 1, 2018) and the additional final payment amount was to be 6.75% of the original principal amount, or $1,012,500. The final payment amount was being amortized as additional interest expense using the effective interest method over the term of the debt.
As stipulated in the 2017 LSA and concurrent with the funding of the 2017 Term Loan, we issued Series F preferred stock warrants to the Lenders which had an aggregate initial fair value of $382,597. In accordance with ASC 480-10, Distinguishing Liabilities from Equity, or ASC 480-10, the warrants are liability-classified as the underlying to the warrant is a puttable security. The initial recognition of the warrant liability created a discount to the debt, which is being amortized over the debt term using the effective interest method. With respect to Oxford, the 2017 LSA was accounted for as a new borrowing. With respect to SVB, the 2017 LSA was accounted for as a modification of our previous lending arrangement with SVB, and therefore no extinguishment gain or loss was recognized.
2018 Loan and Security Agreement
On November 30, 2018, we entered into the 2018 LSA with Oxford, as collateral agent, and Oxford and SVB as equal syndicated lenders. The 2018 LSA replaced the 2017 LSA and provided for a $20.0 million term loan, or
73
the 2018 Term Loan, and a credit line of up to $5.0 million (discussed in the “Revolving Line of Credit” section below), prior to amendment of the 2018 LSA on June 13, 2019, as discussed below. Our obligations under the 2018 LSA are secured by substantially all of our assets, excluding intellectual property and subject to certain other exceptions and limitations. We have the right to prepay the 2018 Term Loan in whole or in part at any time, subject to a prepayment fee of 2.50% if prepaid on or prior to November 30, 2019, 1.50% if prepaid after November 30, 2019 and on or prior to November 30, 2020, and 0.75% thereafter. Upon prepayment, we are also obligated to pay a non-refundable early termination fee of $496,785. Amounts prepaid or repaid under the 2018 Term Loan may not be reborrowed. The 2018 LSA contained a financial covenant that requires us to achieve a monthly trailing six-month revenue target each month throughout the term of the agreement, which was recently amended and changed to a monthly trailing three-month revenue target, as discussed further below. As of December 31, 2018 and March 31, 2019, we were in compliance with this covenant. In addition, the 2018 LSA contains customary conditions of borrowing, events of default and covenants, including covenants that restrict our ability to dispose of assets, merge with or acquire other entities, incur indebtedness and make distributions to holders of our capital stock. Should an event of default occur, including the occurrence of a material adverse change, we could be liable for immediate repayment of all obligations under the 2018 LSA.
On June 13, 2019, we entered into an amendment to the 2018 LSA, or the Amendment, which, among other things, requires that we achieve certain revenue levels tested monthly on a trailing three-month basis. For each month through December 31, 2019, the trailing three-month revenue requirements are calculated as a percentage of our previously approved applicable monthly revenue targets. For monthly periods ending after December 31, 2019, the trailing three-months revenue requirements will be determined by the Lenders upon receipt and review of our monthly financial projections for the year, subject to certain specified criteria regarding minimum requirements. Revenues, if any, that we recognize as a result of an ALJ appeal process from consolidated claims initiatives for DecisionDx-Melanoma do not count toward the minimum revenue requirements. We were in compliance with this covenant as the the most recently tested month prior to the date of this prospectus. Additionally, the Amendment eliminated the $5.0 million revolving line and increased the 2018 Term Loan by $5.0 million.
The 2018 Term Loan bears interest at a floating rate equal to the greater of 8.55% and the 30-day U.S. LIBOR rate as reported in The Wall Street Journal on the last business day of the month that precedes the month in which the interest will accrue, plus 6.48%. The applicable interest rate on the 2018 Term Loan was 8.98% as of December 31, 2018 and 8.97% as of March 31, 2019, respectively. Interest on the 2018 Term Loan is payable monthly in arrears. We are permitted to make interest-only payments on the 2018 Term Loan through May 31, 2020. The principal is required to be repaid in 30 equal monthly installments beginning on June 1, 2020. All unpaid principal and accrued and unpaid interest is due on November 1, 2022, or the 2018 Term Loan Maturity Date. We are also obligated to make an additional final payment of 6.75% of the aggregate original principal amount, or $1,350,000 as of March 31, 2019, upon any prepayment or on the 2018 Term Loan Maturity Date. The final payment amount is being amortized as additional interest expense using the effective interest method of the term of the debt. The final payment amount increased to $1,687,500 in connection with the $5.0 million increase to the 2018 Term Loan under the Amendment.
The 2018 Term Loan was fully funded on November 30, 2018 and the 2018 Revolving Line (defined and discussed below) was fully drawn upon on November 30, 2018. Proceeds from the 2018 LSA were used to repay all outstanding obligations under the 2017 LSA and to provide working capital. As a condition of the loan, we issued Series F preferred stock warrants to the Lenders with an aggregate initial fair value of $158,000. In accordance with ASC 480-10, the warrants are liability-classified as the underlying to the warrant is a puttable security. The initial recognition of the warrant liability created a discount to the debt, which is being amortized over the debt term using the effective interest method. The 2018 LSA was accounted for as a modification of the previous lending arrangement with SVB and Oxford, and therefore no extinguishment gain or loss was recognized.
Revolving Line of Credit
In connection with the issuance of the 2017 Term Loan, we repaid $2.0 million that was drawn on the revolving line under our previous lending arrangement. In the 2017 LSA, the Lenders agreed to provide availability to us of up to $5.0 million of credit, or the 2017 Revolving Line, based on the application of an advance rate against either specified accounts receivable, or accounts receivable in the aggregate, contingent on our satisfaction of borrowing base eligibility requirements. Any outstanding credit advanced against the 2017 Revolving Line was to
74
be due in full no later than March 31, 2019. We drew $4.0 million on the 2017 Revolving Line in June 2017 and drew the remaining $1.0 million balance in May 2018. The 2017 Revolving Line bore interest at a floating per annum rate equal to the greater of 6.25% and 5.48% above the U.S. LIBOR rate. The applicable interest rate on the 2017 Revolving Line at December 31, 2017 was 7.04%.
In the 2018 LSA, the Lenders continued to provide credit availability to us of up to $5.0 million, or the 2018 Revolving Line, with similar terms as the 2017 Revolving Line, except that the 2018 Revolving Line was to be due in full no later than November 30, 2020. The 2018 Revolving Line was eliminated in connection with the Amendment. The applicable interest rate on the 2018 Revolving Line was 7.98% and 7.97% as of December 31, 2018 and March 31, 2019, respectively.
Convertible Promissory Notes
In January and February 2019, we issued $11,770,375 principal amount of unsecured convertible promissory notes, or the Notes, of which $4,755,882 was with related parties (executive officers, members of our board of directors or entities affiliated with them). The Notes bear simple interest at a rate of 8% per annum with a maturity date of January 31, 2020.
On or before the maturity date, the entire outstanding principal amount of and accrued interest on the Notes, or the Conversion Amount, is automatically convertible into shares of our equity securities issued and sold in a single or series of related transactions, with the principal purpose of raising capital, in which we sell shares of such equity securities for aggregate gross proceeds of at least $10.0 million, or the Next Equity Financing. The number of shares of such equity securities issuable in the Next Equity Financing is equal to the quotient of the Conversion Amount as of the closing date of the Next Equity Financing divided by a per share price that is equal to 80% of the lowest per share purchase price of the equity securities sold in the Next Equity Financing. If the Notes have not been repaid or converted prior to the maturity date, then, at the request of the holders of a majority of the then-outstanding principal amount of and accrued interest on the Notes, then the Conversion Amount as of the maturity date will convert into shares of our Series F redeemable convertible preferred stock, or any senior equity security issued by us after the first issuance of the Notes, in each case at a conversion price equal to the price at which such security was last sold (currently $5.8208 for shares of our Series F redeemable convertible preferred stock). If we undergo a change of control while the Notes are outstanding, we are required to repurchase each Note from each holder at a repurchase price equal to two times the principal amount of such Note, plus any accrued and unpaid interest on such Note as of the date of such repurchase.
As discussed further in Note 7 to the unaudited interim condensed financial statements appearing elsewhere in this prospectus, the Notes contain a beneficial conversion feature and an embedded derivative, both of which created a significant debt discount that is being amortized over the life of the debt (that is, through January 31, 2020) using the effective interest method, which will result in significant increases in non-cash interest expense in each succeeding reporting period through the date of maturity or through such earlier date the Notes are otherwise settled or converted.
75
The following table summarizes the aggregate values recorded for the Notes at issuance and as of March 31, 2019 (in thousands):
At Issuance1 |
March 31, 2019 |
|||||
(unaudited) |
||||||
Liability component: |
||||||
Principal |
$ | 11,770 | $ | 11,770 | ||
Unamortized issuance costs |
(74 | ) |
(73 | ) |
||
Unamortized discount from beneficial conversion feature |
(8,378 | ) |
(8,226 | ) |
||
Unamortized discount from embedded derivative |
(2,816 | ) |
(2,765 | ) |
||
Net carrying amount of the liability component |
502 | 706 | ||||
Embedded derivative liability |
2,816 | 2,862 | ||||
Total |
$ | 3,318 | $ | 3,568 | ||
Equity component: |
||||||
Carrying value of beneficial conversion feature recorded in additional paid-in capital |
$ | 8,378 | $ | 8,378 | ||
| 1 | The Notes were issued on January 31, 2019, February 12, 2019 and February 27, 2019. |
The Notes are classified as current liabilities on the balance sheet due to their contractual maturity date of January 31, 2020.
Cash Flows
The following table summarizes our sources and uses of cash for each of the periods presented (in thousands):
Years Ended December 31, |
Three Months Ended March 31, |
|||||||||||
2017 |
2018 |
2018 |
2019 |
|||||||||
(unaudited) |
||||||||||||
Net cash (used in) provided by operating activities |
$ | (12,234 | ) |
$ | (12,295 | ) |
$ | (2,509 | ) |
$ | 1,288 | |
Net cash used in investing activities |
(495 | ) |
(277 | ) |
(29 | ) |
(244 | ) |
||||
Net cash provided by financing activities |
8,867 | 15,838 | 5,348 | 10,707 | ||||||||
Net (decrease) increase in cash and cash equivalents |
$ | (3,862 | ) |
$ | 3,266 | $ | 2,810 | $ | 11,751 | |||
Operating Activities
Net cash used in operating activities of $12.2 million for the year ended December 31, 2017 and was primarily attributable to the net loss of $12.4 million, increases in accounts receivable of $0.4 million and decreases in accrued compensation of $0.2 million, partially offset by net non-cash charges of $0.8 million, primarily consisting of $0.5 million in amortization of debt discount and issuance costs and depreciation expense of $0.3 million. Net cash used in operating activities of $12.3 million for the three months ended March 31, 2019 was primarily attributable to increases in accounts receivable of $8.4 million, the net loss of $6.4 million and increases in inventory of $0.6 million, partially offset by increases in accrued compensation and other accrued liabilities of $1.5 million and net non-cash charges of $1.4 million. Non-cash charges primarily consisted of $0.6 million in amortization of debt discount and issuance costs, stock compensation expense of $0.3 million, changes in the fair value of our preferred stock warrants of $0.3 million and depreciation expense of $0.3 million.
Net cash used in operating activities was $2.5 million for the three months ended March 31, 2018 and was primarily attributable to the net loss of $3.6 million, partially offset by increases in accrued compensation of $1.0 million and non-cash charges of $0.3 million. Non-cash charges primarily consisted of $0.1 million each of amortization of debt discount and issuance costs, stock compensation expense and depreciation expense. Net cash provided by operating activities of $1.3 million for the three months ended March 31, 2019 was primarily
76
attributable to decreases in accounts receivable of $3.8 million and net non-cash charges of $0.6 million, partially offset by decreases in accrued compensation of $1.8 million and the net loss of $1.4 million. Non-cash charges primarily consisted of $0.3 million in amortization of debt discount and issuance costs, stock compensation expense of $0.2 million and depreciation expense of $0.1 million.
Investing Activities
Net cash used in investing activities for the years ended December 31, 2017 and 2018 and for the three months ended March 31, 2018 and 2019 consisted entirely of purchases of property and equipment.
Financing Activities
Net cash provided by financing activities was $8.9 million for the year ended December 31, 2017 and consisted primarily of $6.6 million of net proceeds from the issuance of term debt and preferred stock warrants in connection with the 2017 LSA, $4.0 million in proceeds from our line of credit and $0.3 million in proceeds from the issuance of preferred stock, partially offset by a $2.0 million in repayment on our line of credit. Net cash provided by financing activities was $15.8 million for the year ended December 31, 2018, and consisted primarily of $10.4 million in proceeds from the issuance of preferred stock and preferred stock warrants, $4.4 million from the issuance of term debt and preferred stock warrants in connection with the 2018 LSA and $1.0 million in proceeds from our line of credit.
Net cash provided by financing activities was $5.3 million for the three months ended March 31, 2018, essentially all of which was attributable to proceeds received from the issuance of preferred stock. Net cash provided by financing activities was $10.7 million for the three months ended March 31, 2019 and consisted primarily of $11.7 million in net proceeds from the issuance of the Notes, partially offset by $1.0 million in repayments on our line of credit.
Contractual Obligations and Commitments
The following table summarizes our contractual obligations as of December 31, 2018 (in thousands):
Payments Due by Period |
|||||||||||||||
Less than 1 year |
1-3 years |
3-5 years |
More than 5 years |
Total |
|||||||||||
Principal payments on long-term debt |
$ | — | $ | 17,667 | $ | 8,683 | $ | — | $ | 26,350 | |||||
Interest payments on long-term debt(1) |
2,195 | 3,138 | 329 | — | 5,662 | ||||||||||
Operating lease obligations(2) |
294 | 553 | 415 | — | 1,262 | ||||||||||
Total(3) |
$ | 2,489 | $ | 21,358 | $ | 9,427 | $ | — | $ | 33,274 | |||||
| (1) | Our long-term debt bears interest at a variable rate and therefore our actual interest payments are subject to changes in market interest rates. Estimated interest payments included in the table above are based on interest rates in effect as of December 31, 2018 applied to the principal balances outstanding as of December 31, 2018 and considering the contractual repayment schedule. |
| (2) | Primarily associated with facility leases for our Phoenix, Arizona laboratory and our Friendswood, Texas corporate headquarters. |
| (3) | The table above does not include the obligations associated with the convertible promissory notes issued in 2019. See the discussion above under “Convertible Promissory Notes” for information about these obligations. |
Off-Balance Sheet Arrangements
We do not currently have, nor did we have any off-balance sheet arrangements, as defined in the rules and regulations of the SEC, during the periods presented.
Critical Accounting Policies and Significant Judgments and Estimates
Our financial statements are prepared in accordance with U.S. GAAP. The preparation of our financial statements and related disclosures requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, costs and expenses, and the disclosure of contingent assets and liabilities in our financial statements. We base our estimates on historical experience, known trends and events and various other factors that we
77
believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. We evaluate our estimates and assumptions on an ongoing basis. Our actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are described in more detail in Note 2 to our audited financial statements appearing elsewhere in this prospectus, we believe that the following accounting policies are those most critical to the judgments and estimates used in the preparation of our financial statements.
Revenue Recognition
We recognize revenue in accordance with ASC 606. We adopted ASC 606 with an initial application date of January 1, 2018 using the full retrospective method. In accordance with ASC 606, we follow a five-step process to recognize revenues: (1) identify the contract with the customer; (2) identify the performance obligations; (3) determine the transaction price; (4) allocate the transaction price to the performance obligation; and (5) recognize revenues when the performance obligations are satisfied.
All of our revenues from contracts with customers are associated with the provision of diagnostic and prognostic cancer test reports. Our revenues are primarily derived from DecisionDx-Melanoma, for cutaneous melanoma, and we also have revenues attributable to DecisionDx-UM, for uveal melanoma.
We have determined that we have a contract with the patient when the treating physician orders the test. Our contracts generally contain a single performance obligation, which is the delivery of the test report, and we satisfy our performance obligation at the point in time when we deliver the test report to the treating physician, at which point we can bill for the report. The amount of revenue recognized reflects the amount of consideration we expect to receive, or the transaction price, and considers the effects of variable consideration, which is discussed further below.
Once we satisfy our performance obligations and bill for the test report, the timing of the collection of payments may vary based on the payment practices of the third-party payor and the existence of contractually established reimbursement rates. Most of the payments for our test reports are made by third-party payors, including Medicare and commercial health insurance carriers. Certain contracts contain a contractual commitment of a reimbursement rate that differs from our list prices. However, absent a contractually committed reimbursement rate with a commercial carrier or governmental program, our diagnostic tests may or may not be covered by these entities’ existing reimbursement policies. In addition, patients do not enter into direct agreements with us that commit them to pay any portion of the cost of the tests in the event that their insurance provider declines to reimburse us. We may pursue, on a case-by-case basis, reimbursement from such patients in the form of co-payments and co-insurance, in accordance with the contractual obligations that we have with the insurance carrier or health plan. These situations may result in a delay in the collection of payments.
The Medicare claims that are covered by policy under an LCD are generally paid at the established rate by our Medicare contractor within 30 days from receipt. As the LCD currently only covers a portion of the DecisionDx-Melanoma test reports, we provide for patients covered by Medicare, we intend to attempt to expand this LCD coverage policy in the future. Medicare claims that were either submitted to Medicare prior to the LCD’s effective date or that are not otherwise covered by the terms of the LCD, but that may meet the definition of being reasonable and necessary pursuant to Section 1862(a)(1)(A) of the Social Security Act, are generally appealed by us. If our appeal is successful at the first level (termed “redetermination”), second level (termed “reconsideration”) or third level of appeal (de novo hearing with an ALJ), payment may be made for all or a portion of the claim.
We have concluded that our contracts include variable consideration because the amounts paid by Medicare or commercial health insurance carriers may be paid at less than our standard rates or not paid at all, with such differences considered implicit price concessions. Variable consideration attributable to these price concessions is measured at the expected value using the “most likely amount” method under ASC 606. The amounts are determined by historical average collection rates by test type and payor category taking into consideration the range of possible outcomes, the predictive value of our past experiences, the time period of when uncertainties expect to be resolved and the amount of consideration that is susceptible to factors outside of our influence, such as the judgment and actions of third parties. Such variable consideration is included in the transaction price only to the extent it is probable that a significant reversal in the amount of cumulative revenue recognized will not
78
occur when the uncertainties with respect to the amount are resolved. Variable consideration may be constrained and excluded from the transaction price in situations where there is no contractually agreed upon reimbursement coverage or in the absence of a predictable pattern and history of collectability with a payor. Variable consideration for Medicare claims is deemed to be fully constrained when the payment of such claims is subject to approval by an ALJ at an appeal hearing, due to factors outside our influence (i.e., judgment or actions of third parties) and the uncertainty of the amount to be received is not expected to be resolved for a long period of time. Variable consideration is evaluated each reporting period and adjustments are recorded as increases or decreases in revenues. Included in revenues for the years ended December 31, 2017 and 2018 and for the three months ended March 31, 2018 and 2019 were $0, $(0.9) million, $0.6 million and $0.6 million, respectively, of revenue increases (decreases) associated with changes in estimated variable consideration related to performance obligations satisfied in previous periods. The amount was zero for the year ended December 31, 2017 due to the application of a transition practical expedient in connection with the adoption of ASC 606.
Because our contracts with customers have an expected duration of one year or less, we have elected the practical expedient in ASC 606 to not disclose information about our remaining performance obligations. Any incremental costs to obtain contracts are recorded as SG&A expense as incurred due to the short duration of our contracts. Contract balances consisted solely of accounts receivable (both current and noncurrent) as of December 31, 2017 and 2018 and March 31, 2019.
DecisionDx-Melanoma Claims Consolidation
In June 2017, we submitted to the Office of Medicare Hearings and Appeals, or OMHA, a formal request to participate in a program to consolidate and adjudicate large volumes of claim disputes at an accelerated rate. The program consolidates outstanding claim appeals pending at the ALJ level and uses a statistical-sampling approach where multiple ALJs will determine reimbursement results for a sample of claims which are then extrapolated to the universe of claims. Our consolidated appeal includes 2,698 DecisionDx-Melanoma claims dating from 2013 through spring 2017. The judges who will review the sample sets have been identified and the hearings were held in April 2019 with a supplemental hearing in May 2019. No formal ruling has been issued to date. In accordance with ASC 606, we have not recognized (fully constrained the variable consideration) any revenues attributable to these claims in our financial statements pending the outcome of this matter. We expect to recognize any revenue adjudicated by the ALJ in the reporting period in which we are notified of the ALJ hearing outcome.
Stock-Based Compensation
Stock-based compensation expense for equity instruments issued to employees is measured based on the grant-date fair value of the awards. The fair value of each employee stock option is estimated on the date of grant using the Black-Scholes option-pricing valuation model. We recognize compensation costs on a straight-line basis for all employee stock-based compensation awards over the requisite service period of the awards, which is generally the awards’ vesting period (typically four years). Forfeitures are accounted for as they occur.
Following is a description of the significant assumptions used in the option pricing model:
| • | Expected term. The expected term is the period of time that granted options are expected to be outstanding. We have set the expected term using the simplified method based on the weighted average of both the period to vesting and the period to maturity for each option as we have concluded that our stock option exercise history does not provide a reasonable basis upon which to estimate the expected term. |
| • | Expected volatility. Because our stock is not traded in an active market, we calculate volatility by using the historical stock prices of a group of similar companies looking back over the estimated life of the option and averaging the volatilities of these companies. |
| • | Risk-free interest rate. We base the risk-free interest rate used in the Black-Scholes option valuation model on the market yield in effect at the time of option grant provided from the Federal Reserve Board’s Statistical Releases and historical publications from the Treasury constant maturities rates for the equivalent remaining terms. |
| • | Dividend yield. We have not paid, and do not have plans to pay, cash dividends. Therefore, we use an expected dividend yield of zero in the Black-Scholes option valuation model. |
79
The following table sets forth the assumptions used to determine the fair value of stock options:
|
|
Years Ended December 31,
|
Three Months
Ended March 31, |
|
|
|
2017
|
2018
|
2019
|
|
Average expected term
|
6 years
|
6 years
|
6 years
|
|
Expected stock price volatility
|
58.64% - 60.23%
|
57.77% - 58.29%
|
58.22% - 58.84%
|
|
Risk-free interest rate
|
1.59% - 2.13%
|
2.77% - 3.13%
|
2.18% - 2.47%
|
|
Dividend yield
|
0%
|
0%
|
0%
|
Common Stock Valuations
As there has been no public market for our common stock to date, the estimated fair value of the common stock underlying our stock options was determined by our board of directors, with input from management, considering our most recently available third-party valuations of common stock and our board of directors’ assessment of additional objective and subjective factors that it believed were relevant, and factors that may have changed from the date of the most recent valuation through the date of the grant, which intended all options granted to be exercisable at a price per share not less than the per share fair value of our common stock underlying those options on the date of grant. Prior to our initial public offering, given the absence of a public trading market for our common stock, the valuations of our common stock were determined in accordance with the guidelines outlined in the American Institute of Certified Public Accountants Practice Aid, Valuation of Privately-Held-Company Equity Securities Issued as Compensation. The assumptions we use in the valuation model are based on future expectations combined with management judgment. In the absence of a public trading market, our board of directors with input from management exercised significant judgment and considered numerous objective and subjective factors to determine the fair value of our common stock as of the date of each option grant, including the following factors:
| • | contemporaneous independent valuations performed at periodic intervals by an independent third-party valuation firm; |
| • | the prices, rights, preferences and privileges of our preferred stock relative to the common stock; |
| • | our operating and financial performance and forecast and capital resources; |
| • | current business conditions; |
| • | the hiring of key personnel; |
| • | our stage of commercialization; |
| • | the status of research and development efforts; |
| • | the likelihood of achieving a liquidity event for the shares of common stock underlying these stock options, such as an initial public offering or sale of our company, given prevailing market conditions; |
| • | any adjustment necessary to recognize a lack of marketability for our common stock; |
| • | trends and developments in our industry; |
| • | the market performance of comparable publicly traded technology companies; and |
| • | the U.S. and global economic and capital market conditions. |
In valuing our common stock, we utilized the market approach (including the Guideline Public Company method and recent company transactions method considering our Series F redeemable convertible preferred stock financing), which are considered highly complex and subjective valuation methodologies. The Guideline Public Company method estimates value based on a comparison to comparable public companies that are similar to us in line of business, size, stage of life cycle, and financial leverage. From the comparable companies, a representative market value multiple is determined which is applied to our operating results to estimate an equity value, or Equity Value. The company transaction method estimates the Equity Value based on an assessment of recent transactions in our common stock as well as an assessment of recent preferred stock financing transactions.
80
The Equity Value was allocated among the various classes of our equity securities to derive a per share value of our common stock. We performed this allocation using the option pricing method, or OPM, which treats the securities comprising our capital structure as call options with exercise prices based on the liquidation preferences of our various series of preferred stock and the exercise prices of our options and warrants, and a probability-weighted expected return method, or PWERM, which involves the estimation of multiple future potential outcomes for us, and estimates of the probability of each potential outcome. The per share value of our common stock determined using the PWERM approach is ultimately based upon probability-weighted per share values resulting from the various future scenarios, which include an initial public offering, merger or sale or continued operation as a private company.
After the Equity Value is determined and allocated to the various classes of shares, a discount for lack of marketability, or DLOM, is applied to arrive at the fair value of the common stock. A DLOM is meant to account for the lack of marketability of a stock that is not traded on public exchanges. For financial reporting purposes, we considered the amount of time between the valuation date and the grant date of our stock options to determine whether to use the latest common stock valuation or a straight-line interpolation between the two valuation dates. This determination included an evaluation of whether the subsequent valuation indicated that any significant change in valuation had occurred between the previous valuation and the grant date.
For valuations after the consummation of this offering, our board of directors will determine the fair value of each share of underlying common stock based on the closing price of our common stock on the date of grant or other relevant determination date, as reported on the Nasdaq Global Select Market.
Recent Accounting Pronouncements
Refer to Note 2, “Summary of Significant Accounting Policies,” in the accompanying notes to our financial statements appearing elsewhere in this prospectus for a discussion of recent accounting pronouncements.
Quantitative and Qualitative Disclosures about Market Risk
Interest Rate Risk
Our primary market risk exposure relates to interest rates on our outstanding long-term debt, which bears interest at variable market rates. Based on bank borrowings outstanding as of December 31, 2018 and March 31, 2019, if market interest rates were to increase by 1.0%, our interest expense and operating cash flows would be adversely impacted by approximately $250,000 on an annual basis.
Effects of Inflation
Inflation generally affects us by increasing our cost of labor and research and development costs. We do not believe inflation has had a material effect on our results of operations during the periods presented.
JOBS Act Accounting Election
We are an emerging growth company within the meaning of the JOBS Act. Section 107(b) of the JOBS Act provides that an emerging growth company can leverage the extended transition period, provided in Section 102(b) of the JOBS Act, for complying with new or revised accounting standards. Thus, an emerging growth company can delay the adoption of new or revised accounting standards that have different effective dates for public and private companies until those standards apply to private companies. We have elected to use this extended transition period and, as a result, our financial statements may not be comparable to companies that comply with public company effective dates. We also intend to rely on other exemptions provided by the JOBS Act, including without limitation, not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act.
We will remain an emerging growth company until the earliest of (1) the last day of the fiscal year following the fifth anniversary of the consummation of this offering, (2) the last day of the fiscal year in which we have total annual gross revenue of at least $1.07 billion, (3) the last day of the fiscal year in which we are deemed to be a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock held by non-affiliates exceeded $700.0 million as of the last business day of the second fiscal quarter of such year or (4) the date on which we have issued more than $1.0 billion in non-convertible debt securities during the prior three-year period.
81
Overview
We are a commercial-stage dermatological cancer company focused on providing physicians and their patients with personalized, clinically actionable genomic information to make more accurate treatment decisions. We believe that the traditional approach to developing a treatment plan for certain cancers using clinical and pathology factors alone is inadequate and can be improved by incorporating personalized genomic information. Our non-invasive products utilize proprietary algorithms to provide an assessment of a patient’s specific risk of metastasis or recurrence of their cancer, allowing physicians to identify patients who are likely to benefit from an escalation of care as well as those who may avoid unnecessary medical and surgical interventions. Our lead product, DecisionDx-Melanoma, is a proprietary multi-gene expression profile, or GEP, test that predicts the risk of metastasis or recurrence for patients diagnosed with invasive cutaneous melanoma, a deadly skin cancer. We also market DecisionDx-UM, which is a proprietary GEP test that predicts the risk of metastasis for patients with uveal melanoma, a rare eye cancer. Based on the substantial clinical evidence that we have developed, we have received Medicare coverage for both of our products, which represents approximately 50% of our addressable patient population. We also have two late-stage proprietary products in development that address cutaneous squamous cell carcinoma, or SCC, and suspicious pigmented lesions, which are indications with high clinical need in dermatological cancer.
Skin cancer is the most commonly diagnosed cancer in the United States. There are more than 5.5 million new cases of skin cancer diagnosed annually, compared with 1.6 million new cases for all other cancers combined. DecisionDx-Melanoma targets more than an estimated 100,000 patients diagnosed with invasive cutaneous melanoma each year, which we believe is underreported. In addition, our two late-stage proprietary products target approximately 200,000 patients diagnosed with SCC with high-risk features and approximately 300,000 patients with suspicious pigmented lesions without a definitive diagnosis of skin cancer. We estimate that the total addressable U.S. market for these three indications is approximately $1.8 billion.
Healthcare providers, predominately dermatologists and surgeons who treat melanoma patients, make nearly all treatment decisions for patients diagnosed with skin cancers based upon their expected risk of metastasis or recurrence. Historically these treatment decisions have been based solely on clinical and pathology factors, such as tumor depth or width, ulceration status, nerve invasion and evidence of metastasis to the sentinel lymph node, or SLN. Physicians use these factors to group, or stage, patients into stage-related populations. The average risk of metastasis within a population then guides treatment decisions for all patients within a respective population. However, an individual patient’s risk of metastasis can be significantly different from these stage-related population averages, thereby resulting in some patients receiving unnecessary medical and surgical interventions and some patients being undertreated. This treatment paradigm has led to suboptimal patient care and unnecessary costs to the healthcare system.
We believe that incorporating the genomics of each individual patient’s tumor biology to inform on their specific risk of metastasis can aid the decision-making process for their treatment plan, help optimize patient outcomes and reduce healthcare costs. The genomics of cutaneous melanoma and other skin cancers are highly complex because, unlike some other types of cancer, the presence or absence of a single gene or a limited number of genes has not been shown to accurately predict the risk of metastasis or recurrence. Rather, we believe that risk of metastasis or recurrence of skin cancer requires the analysis of gene expression profiles occurring at the RNA level through the application of artificial intelligence, deep learning and proprietary techniques to identify clinically relevant genomic patterns. Once identified, we then undertake extensive clinical validation and clinical utility studies to develop products that address key unmet medical needs for patients and physicians.
In cutaneous melanoma, nearly every treatment plan decision is based upon a patient’s risk of metastasis and the traditional clinical and pathology factors that are used to estimate population-based rates of metastasis can be inaccurate predictors of an individual patient’s risk. A primary tumor diagnostic biopsy is used to identify most of these factors, such as tumor thickness and ulceration status. These factors then assist physicians in their decision of whether to discuss or recommend a second procedure, the invasive sentinel lymph node biopsy, or SLNB, surgery which can provide additional prognostic information. However, the clinical and pathology factors that lead to a recommendation to perform the SLNB surgery have limitations, in that they are not very predictive when it comes to the likelihood that the patient will have an SLN-positive biopsy result, meaning that melanoma cells are found in their lymph nodes. In fact, approximately 88% of patients who undergo the SLNB surgery are found to have an SLN-negative biopsy result, and these patients remain either as Stage I, the lowest risk group,
82
or as Stage II, the next lowest risk group. Furthermore, despite being classified as low-risk, two out of three patients who develop metastatic disease and die from the primary melanoma tumor received an SLN-negative biopsy result and were classified as Stage I or II following SLNB surgery. The significant diagnostic discordance that exists between the clinical and pathologic factors and likelihood of both an SLN-positive biopsy result and the risk of metastasis or recurrence has led to an opportunity to improve care by adding personalized genomic information.
We developed our proprietary, non-invasive DecisionDx-Melanoma product to address this diagnostic discordance in patients with Stage I-III cutaneous melanoma. The product interrogates the biology of a patient’s tumor by analyzing the gene expression profile of 31 genes, a process made possible by our proprietary algorithm, developed using machine learning techniques. DecisionDx-Melanoma reports the risk of metastasis or recurrence for a patient’s melanoma into two classes and two subclasses, ranging from Class 1A, the lowest risk group, through Class 2B, the highest risk group, based on the genomics of a patient’s tumor. Physicians and patients use this additional tumor-specific genomic information, along with traditional staging criteria, to make better-informed decisions about how to manage the disease.
We have published 18 studies to support the two current clinically actionable uses of DecisionDx-Melanoma. The first use immediately following diagnosis is based upon a patient’s likelihood of having an SLN-negative biopsy result so that physicians and their patients can discuss the risk and benefit of undergoing the SLNB surgery. Data from our 1,421 patient prospective study showed that the 1,065 patients with a melanoma less than or equal to 2.0 mm thick, defined as T1-T2 melanoma, and who received a DecisionDx-Melanoma Class 1A test result, had only a 4.6% likelihood of an SLN-positive biopsy result. This is clinically relevant because current guidelines do not recommend offering the SLNB surgery if the likelihood of an SLN-positive biopsy result is 5% or less. Therefore, all patients with a T1-T2 melanoma and a DecisionDx-Melanoma Class 1A test result, which represented approximately 70% of all T1-T2 patients in our study, could avoid the SLNB surgery based on current guidelines. The second use of our product is to inform the appropriate treatment plan, regardless of the decision to undergo or avoid the SLNB surgery. The aggregate data from all of our published long-term archival population showed a 99.6% negative predictive value, or NPV, for melanoma-specific survival at five years for patients who received a DecisionDx-Melanoma Class 1A test result; meaning that at the five-year mark, 99.6% of patients with a Class 1A test result did not die from their melanoma. We have also demonstrated, in all four of our clinical impact studies, that physicians changed their treatment plans approximately 50% of the time after receiving our test results, showing that DecisionDx-Melanoma can change the way physicians treat their patients.
We also market DecisionDx-UM, a genomic test for use in identifying patients diagnosed with uveal melanoma who are at a low risk of metastasis. Uveal melanoma is a rare but deadly disease with approximately 1,600 patients diagnosed in the United States annually. Similar to DecisionDx-Melanoma, this product also uses a proprietary algorithm developed using machine learning techniques to interrogate the biology of a patient’s tumor by analyzing the gene expression profile of 15 genes of a patient’s tumor. Because approximately 30% of uveal melanoma goes on to metastasize within three years, prior to the availability of DecisionDx-UM in 2010, once the primary eye tumor was treated, nearly all patients were managed under an aggressive metastatic surveillance treatment plan. We have an expansive, peer-reviewed publication dataset with 14 studies documenting the validity and utility of DecisionDx-UM. The first prospective, multi-center study reported a 98% NPV at five years for metastatic-free survival for patients who received a Class 1A test result. Based upon this and additional clinical validity data, two clinical impact studies we conducted reported that over 90% of post-diagnostic management decisions align with the DecisionDx-UM results.
We are developing additional products targeting the challenges faced by physicians in treating their patients’ skin cancer, with two products in late stage development. One of our product candidates, DecisionDx-SCC, is a proprietary GEP test designed to predict the risk of metastasis in patients diagnosed with SCC. Approximately one million patients are diagnosed with SCC in the United States each year. We estimate that approximately 200,000 of those patients have one or more high-risk factors associated with their SCC. The available clinical and pathology staging systems result in many patients receiving unnecessary adjuvant interventions or, for those who may benefit from adjuvant interventions, being placed in “watchful waiting” regimens. Our most recent development study analysis suggests that significant improvements relative to available staging systems could be possible. Based on the timing and results of our planned and ongoing clinical validation studies, we intend to commercially launch this product in the second half of 2020.
83
We are also developing a proprietary GEP test designed to assist physicians in the diagnosis of suspicious pigmented lesions. Of the approximately two million pigmented lesion skin biopsies performed for the diagnosis of melanoma annually in the United States, we estimate that approximately 300,000 cannot be confidently confirmed as melanoma or benign lesion through the use of histopathology alone. Based on the timing and results of our planned and ongoing clinical studies, we intend to commercially launch this product in the second half of 2020.
We have built a commercial organization that focuses on providing solutions to dermatologists, dermatological pathologists and surgeons who care for patients with skin cancer. Our focus on dermatologic cancers has provided us with unique insights into the challenges faced by these physicians that have enabled us to drive adoption of DecisionDx-Melanoma, as well as to identify opportunities for additional products to address unmet clinical needs in dermatologic cancer. We have processed over 40,000 clinical samples since commercial launch, with total proprietary GEP report volume increasing from less than 4,000 in 2015 to more than 13,400 in 2018. Our annual revenue increased from $13.8 million in 2017 to $22.8 million in 2018. We will continue to develop clinical data to substantiate the value of our marketed products, which we believe is important for physicians as they adopt our products for multiple uses. Since the end of 2018, we have also expanded our sales organization from 22 to 35 individuals, as well as expanded our medical affairs group, to further educate physicians, which we believe drives adoption of our products.

Our Competitive Advantages
We are focused on providing actionable genomic information to physicians and their patients. We believe our key competitive advantages are due in part to the following factors:
| • | Development of our products required our machine learning expertise and our proprietary algorithm, which are complex and difficult to replicate. We develop our products using our machine-learning expertise to analyze clinical specimens with associated long-term outcomes data to identify genomic patterns in tumor biology that we believe will accurately predict the risk of metastasis and recurrence. We then validate these genomic patterns, by refining and locking down algorithms to enable additional studies to validate the accuracy of our tests and subsequently document the clinically actionable changes made by physicians when they incorporate our test results into their treatment plan decisions. |
| • | We have demonstrated the ability to provide clinically actionable information despite the complex genomics of skin cancer. In the diagnosis and prognosis of cancer, there is significant current interest in DNA driver mutations as being a predictor of the behavior of cancers. We believe that while the behavior of some cancers may be elucidated by DNA analysis and the response to certain targeted therapies, the majority of skin cancer behavior will best be understood at the gene expression level. Specifically, while DNA mutations of a specific gene are important for tumor behavior, the impact of |
84
other genes, epigenetic changes to the gene and the non-tumor environment which cannot be discerned by the mutation that a gene carries are critical to the understanding of tumor behavior. We believe that focusing on the expression of the gene or how the gene behaves will be more accurate than the mutation of the gene itself.
| • | Our vast and growing database of tumor samples and associated long-term outcomes data enables us to improve our current products and accelerate development of new products. The development and validation of accurate tests is a complex process that requires access to tumor tissue specimens and long-term outcomes data. Such data is not readily available for skin cancer, which creates a barrier to rapid test development and validation. However, over the past ten years we created a sample bank comprised of over 38,000 samples, including 5,900 well-annotated samples that we have used in our clinical studies to date. We have been able to use this sample bank to expand the clinical use of our products, evaluate improvements in new proprietary genomic algorithm approaches and develop new products. |
| • | We have generated, and will continue to generate, robust clinical validity and utility data supporting the use of our products. For example, DecisionDx-Melanoma has been the focus of 18 clinical studies involving more than 3,700 patients published since 2015. We also are making significant investments in further clinical studies to continue to support DecisionDx-Melanoma, DecisionDx-UM and our pipeline products. This growing set of data is significant in educating physicians and patients about the value of our products and supporting reimbursement of our products by third-party payors. |
| • | We have established relationships with physicians that allow us to optimize our interactions, increase adoption of our current products and identify areas of unmet clinical need to efficiently launch additional products. We have published rigorous clinical data, which allows our sales and medical affairs representatives to have substantive, in depth dialogues with physicians. Through these established relationships we have been able to integrate our products into physicians’ work flows and identify further educational programs, which we believe drives adoption of our products. We can also leverage these relationships to identify areas of significant unmet medical need and efficiently launch additional skin cancer products. |
| • | We have experience in navigating the reimbursement landscape. In the molecular diagnostics industry securing reimbursement for new tests is a long, complex and uncertain process. We have developed significant expertise in securing reimbursement for our products. |
Our Strategy
We intend to build upon our position as a leading provider of genomic information for dermatological cancers. To realize this objective we plan to:
| • | Expand adoption of our currently marketed products and educate physicians and their patients on the need for our products to make a more informed treatment plan decision. We believe that cancer treatment plans will be most effective if decisions are personalized for each patient based on the biology of their specific tumor, instead of a one-size-fits-all approach. We will continue to educate physicians and their patients on the diagnostic discordance that leads to over- and under-treatment. |
| • | Continue to generate evidence supporting the clinical utility and validity of our products. We have conducted extensive clinical utility and validity studies to support the adoption of, and reimbursement for, our products. In order to maintain our competitive advantage and increase sales of our products, we will continue to generate additional clinical data to support the use of our products. |
| • | Execute planned expansion of our commercial channel. We plan to increase sales of our products by adding new physicians to our customer base as well as increasing orders by physicians already using our products. We recently increased the number of sales and medical affairs representatives and are planning additional increases through 2019 in order to provide more frequent physician interactions and support the launch of additional products. |
| • | Expand coverage and reimbursement for our products. We plan to have increasing dialogue with third-party payors to highlight our clinical utility and patient outcomes data. We believe these data will |
85
validate the benefit of our products for patients and will persuade more third-party payors to provide coverage and reimbursement. Additionally, we will continue to emphasize our ability to reduce overall cost to the healthcare system by appropriately classifying high-risk patients and removing the need for unnecessary invasive procedures for low-risk patients.
| • | Utilize our development expertise and commercial channel insight to provide additional solutions. We are continuing to develop products that address the challenges facing physicians, including genomic tests for patients with SCC with high-risk factors and suspicious pigmented lesions, addressing an aggregate of approximately 500,000 additional potential patients. |
Dermatologic Cancer Market Overview
Skin cancer is the uncontrolled growth of abnormal skin cells. There are six types of pre-cancers and skin cancers that result in a total annual incidence of 5.5 million patients, three of which are highlighted in the figure below.
The three most common forms of skin cancers are basal cell carcinomas, SCC and cutaneous melanoma. Basal cell carcinomas are abnormal, uncontrolled growths or lesions that arise in the skin’s basal cells, which line the deepest layer of the epidermis. Basal cell carcinomas rarely metastasize beyond the original tumor site. Cutaneous squamous cell carcinoma, or SCC, the second most common form of skin cancer, is an uncontrolled growth of abnormal cells arising from the squamous cells in the epidermis, the skin’s outermost layer. Melanoma, an aggressive form of skin cancer, originates in the pigment-producing melanocytes in the basal layer of the epidermis.
Pre-cancers include suspicious pigmented lesions, which are unusual-looking lesions that may be melanoma, and actinic keratosis, also known as a solar keratosis.

Cutaneous Melanoma
Melanoma tumors originate in the pigment-producing melanocytes in the basal layer of the epidermis. Approximately half of all melanomas are diagnosed prior to expanding into the dermis and are classified as in situ or non-invasive melanomas and DecisionDx-Melanoma is not used in this population. Worldwide statistics suggest that there were nearly 300,000 new cases of melanoma diagnosed worldwide with the U.S. Surveillance, Epidemiology, and End Results, or SEER, database estimating that approximately 96,000 invasive cutaneous melanomas will be diagnosed in the United States in 2019, resulting in approximately 9,300 patient deaths each year. However, multiple recent publications show that diagnosis of melanoma is underreported by between 30% and 40%, meaning that actual diagnoses may be between 125,000 and 135,000 new cases, representing an estimated U.S. total addressable market, or TAM, of $540 million. According to these publications, underreporting reflects the fact that the majority of diagnoses are made by community-based dermatologists and dermatopathologists rather than institutional based specialists who more typically have tumor registry support.
86
Additionally, the incidence of melanoma has steadily increased annually over the last several decades, with an estimated 53% growth in the United States in the last ten years.
After a diagnosis of invasive cutaneous melanoma, healthcare providers have traditionally used clinical and pathology factors from the initial biopsy to estimate the patient’s risk of metastasis. This estimation process, or staging, is used to determine nearly all treatment decisions. Invasive melanoma is staged as Stage I through Stage IV. Tumors characterized as Stage I have invaded the dermis but are thin, with less than or equal to 2.0 mm invasion into the dermis if not ulcerated or less than or equal to 1.0 mm invasion into the dermis if ulcerated. Stage I tumors have the lowest population risk of metastasis and death from melanoma. Stage II tumors, though localized, are thicker than 2.0 mm if not ulcerated or greater than 1.0 mm if ulcerated. Stage III tumors have evidence of regional metastasis, such as palpable metastasis at the regional lymph node basin, in-transit or satellite disease, or melanoma cell(s) in the SLN but without evidence of distant metastatic spread. Stage IV tumors are those in which distant metastasis, such as to the lung or brain, has been detected.
All patients who are diagnosed with an invasive cutaneous melanoma will undergo a wide local excision procedure with the surgical margins determined by the depth of the tumor. The invasive SLNB surgery is recommended to be considered for patients with melanomas greater than or equal to 0.8 mm thick or with the presence of an adverse pathologic feature such as the presence of ulceration, high mitotic rate, and transected base. It is believed that tumors with these anatomic based features have a likelihood of an SLN-positive biopsy result 5% or more of the time. If the SLNB surgery is performed, then the wide local excision is performed at that time. An SLN-positive biopsy result, meaning that at least one melanoma cell was seen in the SLN tissue, leads to re-staging the patient as Stage III. Guideline committees have selected this 5% threshold due to a combination of some studies showing a false negative rate of the SLNB surgery at 5% (meaning that 5% of the time, or more, the guideline committees expect a patient with an SLN-negative biopsy result will subsequently develop lymph node metastasis) as well as matching to the reported surgical complication rate. However, the published literature documents a higher surgical complication and false negative rate.
Cutaneous Squamous Cell Carcinoma
Cutaneous squamous cell carcinoma, the second most common form of skin cancer, is an uncontrolled growth of abnormal cells arising from the squamous cells in the epidermis, the skin’s outermost layer. Approximately one million patients are diagnosed with SCC each year in the United States and incidence has doubled over the last three decades. Worldwide data on SCC is inconsistently reported but the incidence outside the United States is estimated to be greater than two million diagnoses annually.
Until recently, SCC was considered a benign skin cancer. However, due to the rate of increased incidence, more patients die annually from SCC in the United States (approximately 15,000 patients) than from cutaneous melanoma. Similar to melanoma, SCC staging systems generally rely upon tumor dimensions and presence or absence of other adverse features.
Suspicious Pigmented Lesions
Suspicious pigmented lesions are unusual-looking lesions that may be melanoma. There are approximately two million skin biopsies performed specifically for the diagnosis of melanoma in the United States. Approximately 15% of these biopsies are classified as indeterminate, in which case a pathologist cannot make a definitive diagnosis as to whether the biopsy is benign or malignant.
Uveal Melanoma Market Overview
The incidence of uveal melanoma has remained relatively constant over time with approximately 1,600 patients diagnosed per year in the United States, representing an estimated U.S. TAM of $7.0 million. Uveal melanomas arise from the three tissues comprising the uveal tract and vary by location with approximately 90% occurring in choroid, 5% in the ciliary body and 5% in the iris. Uveal melanoma may also be referred to as ocular melanoma.
Significant Limitations of Current Clinical and Pathology Staging Systems for Skin Cancer
The dermatologic skin cancer market has significant unmet clinical needs, as clinical and pathology staging systems have traditionally applied a population-wide approach and have not incorporated the genomics of each patient’s tumor biology. This is unlike the diagnostic process applied to other solid tumors, such as breast and
87
prostate cancer, where the broader use of genomics to understand tumor biology has led to individualized patient treatment plans. Not incorporating tumor biology leads to a discordance between the estimated and actual risk of metastasis, which results in over- and under-treatment as well as increased healthcare costs.
Cutaneous Melanoma
The clinical and pathology staging system for invasive cutaneous melanoma is based upon the anatomic findings of the melanoma; that is what the pathologist can see under the microscope from an initial tumor biopsy and what the physician can feel or see during a clinical exam or upon imaging. While this staging system provides population-based risk of metastasis estimates it does not evaluate nor incorporate the biology of the patient’s primary tumor.
Importantly, while it was formerly believed that the SLNB surgery improved melanoma specific survival, the landmark, prospective, randomized multi-center study conducted by the National Cancer Institute, the MSLT-I study, showed that death from melanoma was the same in patients who were randomized to the SLNB surgery or observation, indicating that the SLNB surgery is prognostic, and not therapeutic, as it relates to the risk of death from melanoma. On average, 12% of patients undergoing the SLNB surgery will have an SLN-positive biopsy result and 88% will not. The invasive SLNB surgery carries significant healthcare burden. For instance, the overall complication rate of SLNB was shown to be 11.3% in a systematic review of 21 articles representing 9,047 patients. A separate review reported that the false negative rate of the SLNB surgery ranged from 5% to 21%, with a median rate of 18%.
Both the complication and false negative rates are above the recommended 5% threshold proposed by guideline committees. Further, the SLNB surgery requires the use of general anesthesia and nearly half of the surgeries are performed in an in-patient setting, leading to an average reimbursed cost of $20,000 to $24,000. Finally, the 88% SLN-negative rate carries significant patient and healthcare system implications. This means that 88% of the time, patients undergoing an SLNB will have no change in their treatment plan and no change in their staging, but are still exposed to the complications from the surgery, including general anesthesia risks, cost and a median false negative rate of 18%. Thus, while it is true that patients who are SLN positive, or re-staged to Stage III, have a higher population-based rate of metastasis and death from melanoma, the SLNB surgery does not improve melanoma-specific survival, carries risk of complications, a high false negative rate and significant costs.
In addition to the significant clinical issues involved in only using the traditional clinical and pathology factors to determine SLNB eligibility, a discordance exists between an individual’s stage and their risk of metastasis or death from melanoma. Based on data from SEER and the American Joint Committee on Cancer, or AJCC, of the patients diagnosed with Stage I, II or III cutaneous melanoma, 80% of melanomas are classified as the lowest risk, Stage I, and 12% are classified as next lowest risk, Stage II. However, these data show that patients with melanomas that are initially diagnosed as Stage I or II represent 60% of all deaths in patients initially diagnosed as Stage I, II or III. Furthermore, while patients with Stage III melanoma are at a higher population risk of metastasis and death from melanoma, the five-year melanoma-specific survival rate these patients is 77%. The limitations of the current staging system not only result in unnecessary SLNB surgeries for certain low-risk patients, but we believe also leads to overtreatment with adjuvant immune-oncology and targeted therapies for certain patients with Stage III melanoma.
The risk of metastasis determines the treatment plans in newly diagnosed patients, including the recommendation for the SLNB surgery, decisions around the initiation of advanced imaging for active surveillance, frequency and specialty for clinical follow-up, initiation of adjuvant therapy and discussion of clinical trial enrollment opportunities. The graphic below illustrates the limitations of the SLNB surgery and staging systems as a risk of metastasis indicator.
88
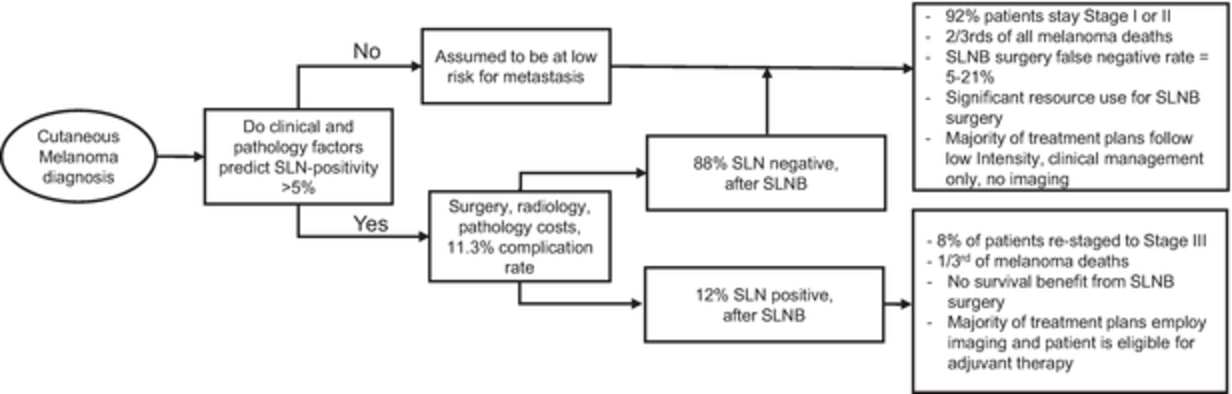
Cutaneous Squamous Cell Carcinoma
The clinical staging for SCC also presents challenges for physicians. Unlike in cutaneous melanoma, where longitudinal databases were developed in an attempt to align population-based risk of metastasis with clinical and pathology factors, the same level of effort has not been given to SCC. As recently as three years ago, there were as many as three SCC staging systems in the United States, each with widely divergent classifications for high- and low-risk patients. For example, a 2014 study compared the AJCC and National Comprehensive Cancer Network, or NCCN, systems to assess concordance between the AJCC and NCCN systems. The AJCC system classified 82% as low risk while the NCCN system classified 13% as low risk. As such, this level of discordance results in the staging systems minimally impacting treatment plans, with patients frequently being over- and under-treated.
Today, there are two principal staging systems for SCC: the AJCC version and the Brigham Women’s Hospital, or BWH, version. Both systems rely upon clinical and pathology factors to stage or classify risk of metastasis, and have good clinical NPV but have low sensitivity and positive predictive value, or PPV. For example, analysis of the first 217 patients analyzed from our initial clinical validation study found NPV for regional or distant metastasis of 96% and 95% for AJCC and BWH, respectively, meaning that of 100 patients classified as low risk, 96 and 95 of them would not have gone on to metastasize. However, sensitivity was 46% and 23%, respectively, which means that out of 100 patients who metastasized, only 46 and 23 were correctly identified as high risk. In addition, the PPV was 12% and 13%, respectively, meaning that of 100 patients classified as high risk, only 12 and 13 actually metastasized. From a PPV perspective, this means that developing an adjuvant treatment plan that includes radiation, or chemotherapy or complete lymph surgical dissection, or a combination of these, may be appropriate for the one out of eight patients who will metastasize but not for the remaining seven patients who would not have metastasized. Furthermore, the low sensitivity means that half to three-quarters of the patients who will go on to metastasize are not even considered for adjuvant treatment. These accuracy metrics have created significant discordance in the approach to managing patients with high-risk features, including intervention for all high-risk patients and “watch and wait” for all high-risk patients. The end result is an unacceptable clinical discordance in the approach to treatment plans and significant over- and under-treatment for a diagnosis that leads to the most skin cancer deaths in the United States.
Suspicious Pigmented Lesions
A pigmented lesion biopsy that is difficult to diagnose may lead to an indeterminate diagnosis, in which case the treating physician generally leans towards making a conservative decision and assume that the lesion is melanoma. A definitive diagnosis of invasive cutaneous melanoma results in a treatment plan that involves wider margins for the definitive wide local excision surgery, consideration of the SLNB surgery and post-diagnosis management plans, including frequent, high intensity surveillance using advanced imaging, frequent clinical visits and encouragement to enroll in clinical studies. If the indeterminate lesion was benign, then the recommendation in the majority of cases would be no additional intervention. Thus, the tendency of physicians to treat an indeterminate diagnosis as melanoma leads to significant over-treatment decisions, complications and increased healthcare costs.
89
Uveal Melanoma
Approximately 97% of patients with uveal melanoma have no evidence of metastatic disease at the time of diagnosis and the success rate for definitive treatment of the primary tumor surpasses 90%. However, within three years, approximately 30% of all patients will experience metastases. Prior to commercial availability of DecisionDx-UM, other clinical staging and molecular diagnostic tests existed for uveal melanoma, but the lack of prospective studies, coupled with low accuracy, resulted in these tests primarily being used for research purposes rather than for clinical management of patients in the United States. As a result, nearly all U.S. centers grouped patients into a single, high-risk treatment plan that included frequent, high intensity surveillance using advanced imaging, frequent clinical visits and encouragement to enroll in clinical studies.
Our Solution
We use the gene expression profile of an individual patient’s tumor biology to inform specific prognosis of metastasis or recurrence and aid the decision-making process of the treating physician and their patient to help optimize health outcomes and reduce healthcare costs. Due to the biological complexity of skin cancers, developing accurate products takes scientific diligence, stringent clinical protocols, machine learning expertise, proprietary algorithms and significant investments of time and capital. In addition, the underlying tissue samples and associated outcomes data required to develop and validate these products are difficult to obtain. Once successfully developed and validated, commercial success requires the generation of clinical use documentation to support appropriate physician adoption, reimbursement success and guideline inclusion.
We have commercially launched DecisionDx-Melanoma, a proprietary GEP test designed to identify the risk of metastasis or recurrence in patients diagnosed with cutaneous melanoma, and DecisionDx-UM, a proprietary GEP test designed to identify the risk of metastasis in patients diagnosed with uveal melanoma. Multiple studies for both products have been published since completion of the initial clinical validation studies and have confirmed the accuracy of our products. Also, multiple clinical impact studies have demonstrated a significant impact on physician decisions to alter their treatment plan when the results of our test are considered in concert with the traditional clinical and pathology factors. Both of our currently marketed products may be reimbursed by Medicare under positive coverage policies. In addition, we have received widespread positive private payor coverage and positive guideline inclusion for DecisionDx-UM, our first melanoma test. Since commercial launch, we have processed more than 40,000 clinical patient samples.

Our products are designed to provide the following benefits:
| • | Better Information for Physicians. We provide physicians and their patients with a report that contains clinically actionable information to inform the treatment plan for each individual patient. Our reports are updated as new clinical data is generated that may enable additional clinical decisions to be made. Based on four studies that we have conducted on clinical actionability, based on our test reports, physicians changed a patient’s treatment in more than 50% of cases, indicating physician confidence in the evidence underlying our reports. |
90
| • | Better Patient Care. The clinical evidence shows that our products are accurate predictors of a patient’s specific risk of metastasis or recurrence of their cancer based upon the gene expression profile of their tumor, independent of available clinical and pathology factors. Physicians use this information to identify patients who are likely to benefit from an escalation of care as well as those who may avoid unnecessary treatments, such as medical and surgical interventions. |
| • | Reduced Healthcare Costs for Payors. We believe our products have the potential to reduce overall healthcare costs by enabling physicians and their patients to avoid unnecessary medical and surgical interventions, including the SLNB surgery. As an example, without DecisionDx-Melanoma, 88% of patients who receive the SLNB surgery, which has an average in-patient reimbursed cost of $20,000 to $24,000, are found to be SLN-negative and remain classified as low risk. If all patients eligible for the SLNB surgery were tested and their test results were acted upon, we estimate the potential savings to the U.S. healthcare system could be up to $250 million, after considering the cost of DecisionDx-Melanoma. |
Our Products
We currently market two proprietary products, DecisionDx-Melanoma and DecisionDx-UM, and have two active proprietary products in development, which we believe support an estimated total addressable market of $1.8 billion in the United States. We have received positive local coverage determinations, or LCDs, providing Medicare coverage for both of our commercial products. These LCDs facilitate reimbursement from Medicare, which represents approximately 50% of the addressable patient population. We also have third-party payor coverage for over 100 million lives for DecisionDx-UM and over 14 million lives for DecisionDx-Melanoma.
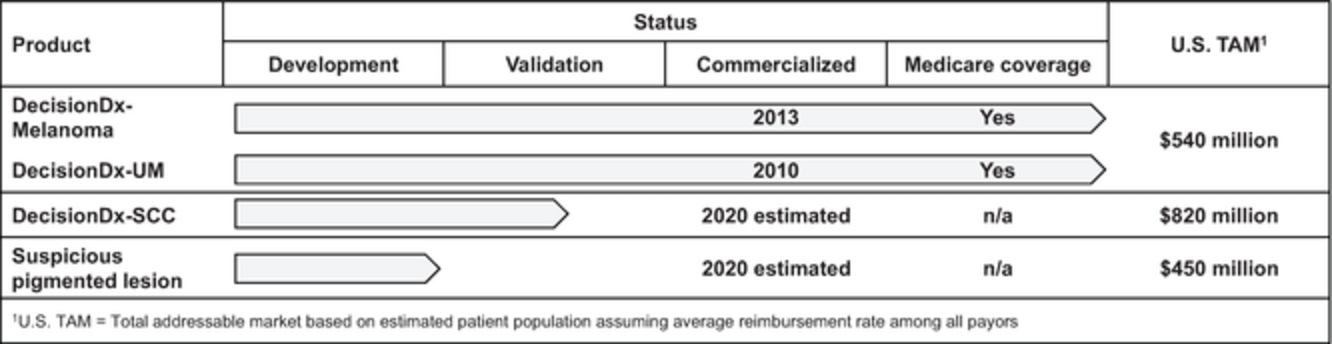
DecisionDx-Melanoma

Overview
We developed and market DecisionDx-Melanoma to healthcare providers for use with patients diagnosed with invasive cutaneous melanoma. Without the use of DecisionDx-Melanoma, these patients are classified in low- and high-risk categories based on population-wide clinical and pathology features, which impact a physician’s treatment plan recommendations, including whether or not to offer the invasive SLNB surgery, frequency and use of clinical imaging and follow-up frequency, adjuvant therapy and clinical trial enrollment. Unfortunately, these clinical and pathology features do not take incorporate the genomics of an individual patient’s tumor biology, which often leads to a misclassification of a patient’s risk of metastasis or recurrence.
To address this need for a more accurate predictor of metastatic risk, we discovered, developed and completed validation for DecisionDx-Melanoma. This product is designed to help physicians identify high-risk patients with Stage I and II melanomas based on biological information, or expression, from 31 genes within their tumor tissue. DecisionDx-Melanoma does not change a physician’s standard diagnostic workflow for suspicious pigmented lesions, which includes performing the initial biopsy procedure and placing the biopsied tissue in formalin. The dermatopathologist then embeds the specimen in a paraffin block, cuts sections that are stained for viewing under a microscope and makes a diagnosis of invasive melanoma. We then extract and purify RNA from
91
sections of the remaining specimen to run our test. We report test results in two classes and two subclasses. Class 1A represents the lowest risk group, Class 1B represents a low risk group, Class 2A represents an increased risk group and Class 2B represents the highest risk group.
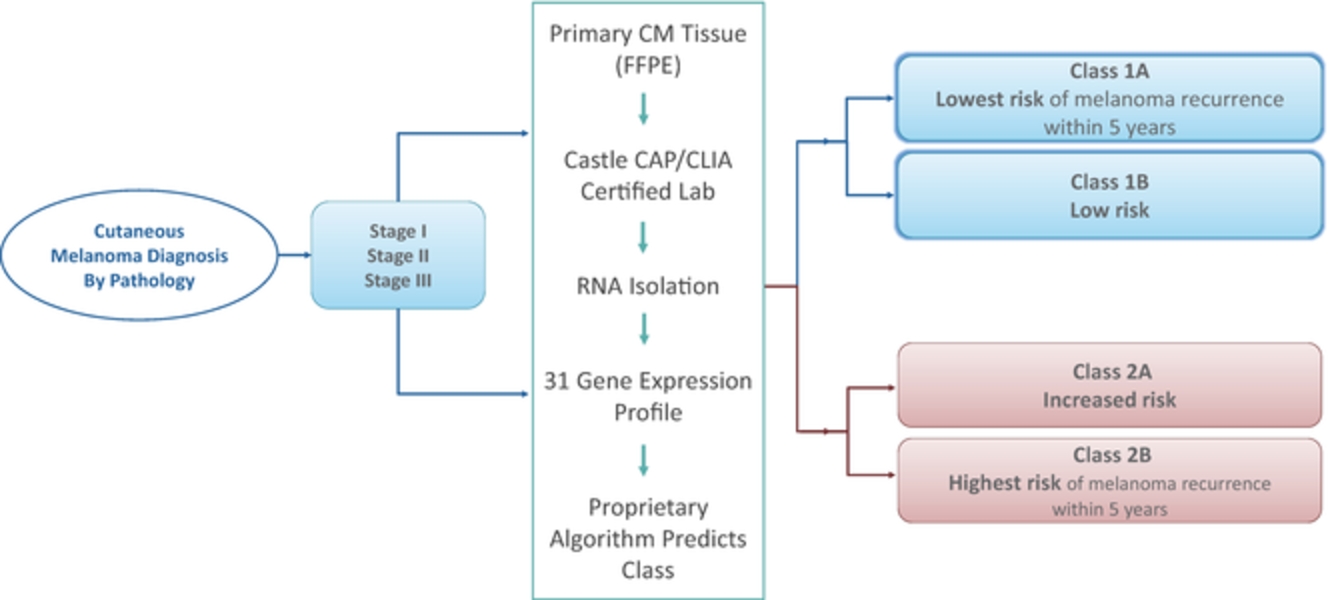
Clinical Validation
We have published 11 clinical validation studies of DecisionDx-Melanoma, which we believe is the largest clinical validation program of the metastatic risk of cutaneous melanoma ever conducted. Based on our published data, we have shown that DecisionDx-Melanoma is an accurate independent predictor of the risk of metastasis or recurrence, showing an aggregate melanoma-specific five-year survival rate of 99% for patients who receive the Class 1A test result.
Our first study, published in January 2015, analyzed 104 patients with Stage I, II and III melanoma from an independent cohort with long term outcomes data. This study reported a five-year disease free survival rate of 98% for patients with Stage I and II melanoma who received a Class 1 test result. In addition, the study also reported that only 2% of patients with a Class 1 test result were SLN-positive.
Our January 2019 study published in the Journal of the American Academy of Dermatology reviewed data on 690 patients with Stage I, II and III melanoma from all three previously published long-term archival publications, and enabled analysis of clinically important subgroups. Overall, the study reported a five-year melanoma specific survival rate of 99% for patients with Stage I, II or III melanoma who received a Class 1A test result.
Our long-term outcomes study data shows that we can provide a more specific individual risk of metastasis and death from melanoma that is distinct from the AJCC stage approach that limits prediction to clinical and pathology factors. The only endpoint reported by the AJCC is death from melanoma. For patients diagnosed with Stage I melanoma, DecisionDx-Melanoma predominantly identifies patients with a risk of death from melanoma that is similar to a patient with Stage IIIA melanoma, with the remaining group having a 99.6% likelihood of being alive from melanoma at five years. For patients with an intermediate risk Stage II melanoma, DecisionDx-Melanoma can distinguish between patients who have a very low risk of death from melanoma (>99% likelihood of being alive at 5-years) from those who have a higher risk of death from melanoma that is similar to a patient with Stage IIIA/IIIB melanoma. For patients with a Stage III melanoma, DecisionDx-Melanoma can identify patients who have a likelihood of death from melanoma similar to a patient with Stage IIA melanoma, with the remainder having a risk similar to a patient with Stage IIIC melanoma.
The ability of DecisionDx-Melanoma to improve the risk of recurrence or risk of death from melanoma accuracy of these patient populations is clinically significant as NCCN guidelines recommend that the duration and frequency of follow-up and intensity of cross-sectional imaging be based on a patient’s individual conditional probability of recurrence. The NCCN guideline cut-point for these decisions is between Stage I-IIA versus Stage IIB-III. For example, the chart below demonstrates that a patient with a Stage I melanoma but a DecisionDx-Melanoma Class 2B test result has a melanoma specific survival rate of 89.5%, which is a higher
92
risk than if the patient had a Stage IIIA melanoma. Today, patients with a Stage III melanoma are recommended to have an increased follow-up schedule, undergo routine cross-sectional imaging, consider initiation of adjuvant therapy, such as an anti-PD1 inhibitor, and consider enrollment in a clinical trial. None of these options would be considered in a patient with a Stage I melanoma in the absence of a DecisionDx-Melanoma test result.

Our first prospective, multi-center study of 322 patients with Stage I, II and III melanoma was published in August 2017. This interim analysis reported a recurrence free survival rate of 97% and overall survival rate of 99% for patients with Stage I, II and III melanoma who received a Class 1 test result.
The most recent, independent, prospective, multi-center study of 86 patients with Stage IB and II melanoma was published in February 2019 in the Journal of The European Academy of Dermatology and Vernereology. This study reported a recurrence free survival rate of 100% for patients with Stage IB and II melanoma who received a Class 1 test result.
93
When evaluating DecisionDx-Melanoma, one of the most important criteria is whether the test adds new information that is independent of the traditional clinical and pathology factors. The formal statistical method used to evaluate independence is the Cox multivariate analysis. Outputs of the Cox multivariate analysis include statistical significance, measured by p-value, as well as the power of the result, measured by Hazard Ratio, or HR. A p-value of less than 0.05 indicates statistical significance and thus independence. If statistical significance is reached, then the HR indicates the power of the result, with a higher HR indicating greater outcome prediction. For example, an HR of nine means that patients with a high-risk test result are nine times more likely to experience metastasis or death than a low-risk test result. The table below shows the Cox multivariate analysis of the disease-free survival, melanoma-specific survival and recurrence free survival from the four performance studies noted above.
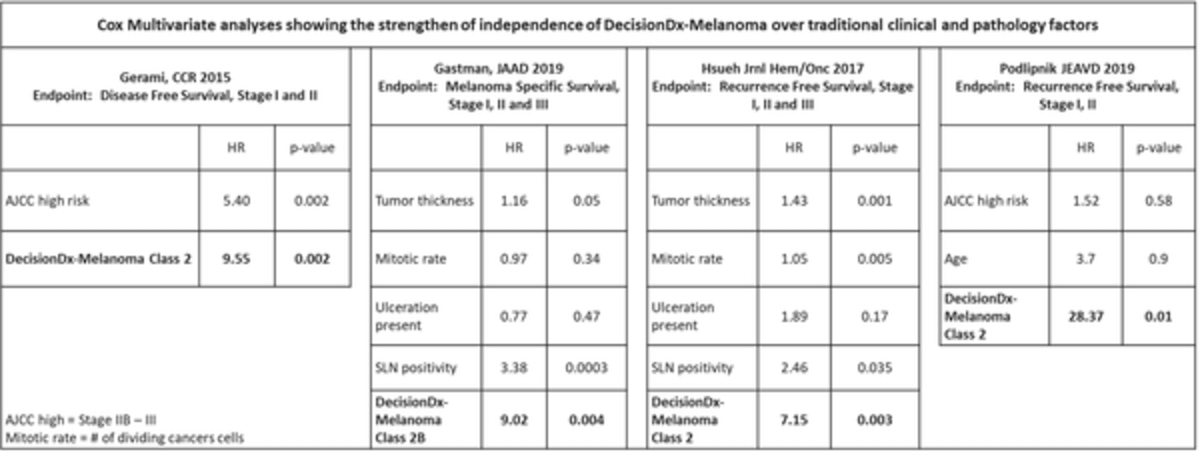
94
The American Academy of Dermatology and other organizations use the Strength of Recommendation Taxonomy, or SORT, system to evaluate prognostic tests such as DecisionDx-Melanoma. The SORT system ranks evidence of clinical validity as levels 1, 2 or 3, and assigns a strength of recommendation as levels A, B or C. A SORT level 1A is the highest level and 3C is the lowest. For SORT ranking, “a systematic review or meta-analysis of good quality studies” or “a prospective study with good follow-up” represents a level 1 for good quality evidence of clinical validity. For SORT strength of recommendation, “consistent, good quality evidence” represents a level A recommendation. A meta-analysis was recently completed and presented at the 2019 Annual Meeting of the American Academy of Dermatology. This meta-analysis reviewed multiple peer-reviewed published clinical validation studies of DecisionDx-Melanoma, including prospective studies. The meta-analysis and the prospective studies satisfied the level 1 ranking of good quality studies and the consistency of DecisionDx-Melanoma data across these studies satisfied the level A strength of recommendation. Thus, the authors concluded that DecisionDx-Melanoma achieved a 1A level of evidence of clinical validity and strength of recommendation under the SORT system. Furthermore, as shown below, the multi-variate analysis for recurrence-free survival found DecisionDx-Melanoma to be the strongest predictor of risk of recurrence compared to the evaluable clinical and pathology factors.

In addition, we conducted a prospective, multi-center study of 1,421 patients, which was published in Future Oncology in January 2019 which focused on the performance of DecisionDx-Melanoma to predict metastasis to the SLN. This study found that patients with a Class 1A test result with melanomas less than or equal to 2.0 mm thick, which represents 86% of all melanomas, have a 95% probability for an SLN-negative biopsy result. Analyzing this data by age shows that patients 65 years of age or older have a 98% NPV, those between 64 and 55 years of age have a 95% NPV and patients under 55 years of age have a 92% NPV. For physicians and patients evaluating whether to use DecisionDx-Melanoma to guide decision-making on the SLNB surgery, the impact on melanoma specific survival is an important consideration if the SLN status is not known. To address this, we analyzed the long-term outcome data from our Gastman 2019 publication and showed that patients of all ages with a melanoma less than or equal to 2.0 mm thick and a Class 1A test result have a five-year melanoma specific survival rate of 99.6%, while similar patients 55 years or older had a melanoma specific survival rate of 99.3%. This study shows that use of DecisionDx-Melanoma for patients with melanomas of less than or equal to 2.0 mm thick could potentially result in 74% less SLNB surgeries.
95
Clinical Utility
We have published four studies documenting how DecisionDx-Melanoma impacts treatment plan decisions. Across all four studies, based on the results of our test reports, physicians changed their treatment plan recommendations approximately 50% of the time. This change in the management of patient treatment plan recommendations compares favorably to leading molecular diagnostic tests as well as to the SLNB surgery, which only changes clinical decision-making approximately 12% of the time.
Study |
Design |
# of Patients |
% Change in Management |
||
Berger et al. CMRO 2016 |
Prospectively tested cohort, multi-center. Retrospective pre test / post test management. |
156 | 53% |
||
Dillon et al. SKIN J Cutan Med 2018 |
Prospective, multi-center: pre test / post test management. |
247 | 49% |
||
Farberg et al. J Drugs Derm 2017 |
169 physician impact study: patient vignettes with pre test / post test management. |
n/a | 47-50% |
||
Schuitevoerder et al. J Drugs Derm 2018 |
Prospectively tested cohort, single center. Retrospective pre test / post test management; and modeling of prospective cohort. |
91 | 52% |
||
These studies illustrate how physicians use DecisionDx-Melanoma to inform the treatment pathway for patients who have been diagnosed with invasive cutaneous melanoma. The initial use of the test is to determine whether to offer and recommend the SLNB surgery to patients with melanomas less than or equal to 2.0 mm thick. Following this decision, physicians use DecisionDx-Melanoma to guide the appropriate post-SLNB surgery treatment plan for their patients, including decision-making regarding advanced imaging, frequency of clinical visits, referral to medical oncology, adjuvant therapy, clinical trial enrollment, and watchful waiting.

Health Economics
We believe that the use of DecisionDx-Melanoma can reduce the number of SLNB surgeries, which has an average in-person reimbursed cost of between $20,000 and $24,000, thereby reducing overall cost of patient treatment for invasive cutaneous melanoma. If all patients eligible for the SLNB surgery were tested and their test results were acted upon, we estimate the potential savings to the U.S. healthcare system could be up to $250 million, after considering the cost of DecisionDx-Melanoma.
96
In addition, DecisionDx-Melanoma can be used to make more informed decisions on advanced imaging, frequency of clinical visits, referral to medical oncology, adjuvant therapy initiation and clinical trial enrollment. In some cases, the DecisionDx-Melanoma test result may guide an appropriate reduction in these decisions based upon a low risk of metastasis and in others it will guide an appropriate increase with the end result being improved use of healthcare resources.
Summary of Our DecisionDx-Melanoma Studies
The table below summarizes the DecisionDx-Melanoma clinical studies that have been published to date:
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Clinical Validity
|
|
|
|
|
Guidance of sentinel lymph node biopsy decisions in patients with T1-T2 melanoma using gene expression profiling
|
Future Oncology, January 2019
(SLNB rate: multicenter; prospective; n=1421 Survival analysis: retrospective; n=690) |
•
|
Patients with T1/T2 melanomas and a Class 1A result had a SLN positive rate <5% while Class 2B patients had a rate above 10%. This is clinically significant as national guidelines do not recommend a SLNB if the risk is <5% and do recommend it if the risk is >10%.
|
|
|
•
|
Melanoma-specific survival (MSS) was 99.6% for patients with Class 1A, T1/T2 tumors who would avoid a SLNB.
|
|
|
|
|
|
|
|
Prospective validation of the prognostic 31-gene expression profiling test in primary cutaneous melanoma
|
Cancer Medicine, March 2019 (singlecenter; prospective; n=159 stage I-III melanomas)
|
•
|
Median follow-up of 45 months for recurrence-free cases.
|
|
•
|
DecisionDx-Melanoma Class 1 was an independent predictor of recurrence (p=0.0001) and the most significant predictor of recurrence with a hazard ratio of 9.2. DecisionDx-Melanoma Class 1 was also an independent predictor of recurrence (p=0.009) and the most significant predictor of distant metastasis with a hazard ratio of 19.0.
|
||
|
|
•
|
NPV for Class 1 for distant metastasis free survival was 99%.
|
|
|
|
•
|
Of 29 recurrences, 10 (34%) occurred in SLN positive cases while 23 (79%) occurred in Class 2. Of the 10 recurrences in SLN positive cases, 9 were Class 2.
|
|
|
|
|
|
|
|
Early outcome of a 31-GEP test in 86 AJCC stage IB-II melanoma patients. A prospective mutlicentre cohort study
|
Journal of the European Academy of Dermatology and Venereology, February 2019 (Multicenter; prospective; n=86 stage IB-II melanomas)
|
•
|
DecisionDx-Melanoma Class 1 was an independent predictor of recurrence (p=0.01) and the most significant predictor of recurrence with a hazard ratio of 18.82. AJCC stage and age were not independent of DecisionDx-Melanoma Class 1.
|
|
•
|
NPV for Class 1 for RFS was 100%.
|
||
|
•
|
All recurrences occurred in Class 2 patients.
|
||
97
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Identification of patients at risk for metastasis using a prognostic 31-gene expression profile in subpopulations of melanoma patients with favorable outcomes by standard criteria
|
Journal of the American Academy of Dermatology, January, 2019
(Multicenter; archival; n=690 stage I-III melanomas) |
•
|
DecisionDx-Melanoma Class 1A was an independent predictor of RFS, DMFS and MSS in the entire cohort and the most significant predictor with hazard ratios of 2.92, 2.89 and 9.02 for RFS, DMFS and MSS, respectively.
|
|
•
|
Subpopulation analysis of patients with Stage I-IIA melanoma showed that DecisionDx-Melanoma Class 1A was the only independent predictor of RFS, DMFS and MSS for all three endpoints compared to tumor thickness, ulceration status, and mitotic rate. Tumor thickness was an independent predictor for RFS but DecisionDx-Melanoma Class 1A was 499% greater than tumor thickness for this endpoint.
|
||
|
•
|
Subpopulation analysis of patients with melanomas <1.0mm showed that DecisionDx-Melanoma Class 1A was an independent predictor or RFS and the most significant predictor with a hazard ratio of 9.34 which was over 200% greater than SLNB status.
|
||
|
•
|
NPV for Class 1A for MSS was 99%.
|
||
|
|
|
|
|
|
Estimation of Prognosis in Invasive Cutaneous Melanoma: An Independent Study of the Accuracy of a GEP Profile Test
|
Dermatologic Surgery,
December, 2018 (Independent; single center; prospective; n=256 stage I/II melanomas) |
•
|
Patients with a DecisionDx-Melanoma Class 2 result were 22 times more likely to metastasize compared to a Class 1 result. Multi-variate statistical analysis for independence was not reported.
|
|
•
|
NPV for Class 1 for recurrence was 99%.
|
||
|
|
|
|
|
|
Interim analysis of survival in a prospective, multi-center registry cohort of cutaneous melanoma patients tested with a prognostic 31-GEP test
|
Journal of Hematology and Oncology, August, 2017 (Multicenter; prospective; n=322 stage I-III melanomas)
|
•
|
DecisionDx-Melanoma Class 1 was an independent predictor of RFS and the most significant predictor with a hazard ratio of 7.15 and 290% greater than the nearest predictor, SLNB.
|
|
•
|
NPV for Class 1 was 98-99% for RFS, DMFS and OS.
|
||
|
•
|
Of 12 distant metastatic events, 10 occurred in the Class 2 group compared to 6 in the SLN positive group.
|
||
|
|
|
|
|
98
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Development of a prognostic genetic signature to predict the metastatic risk associated with cutaneous melanoma
|
Clinical Cancer Research, January, 2015
(Multicenter; archival; n=268 stage I-IV melanomas) |
•
|
DecisionDx-Melanoma Class 1 was an independent predictor of disease free survival, DFS, and the most significant predictor with a hazard ratio of 9.55 compared to 5.40 for AJCC stage.
|
|
•
|
5-year DFS rate for Class 1 (97%) was significantly better than for Class 2 (31%; p<0.0001) and 98% for patients with Stage I or II melanomas.
|
||
|
•
|
SLN positivity rate was 2% in patients with a Class 1 result.
|
||
|
|
|
|
|
|
Identification of high-risk cutaneous melanoma tumors is improved when combining the online American Joint Committee on Cancer Individualized Melanoma Patient Outcome Prediction Tool with a 31-GEP based classification
|
Journal of the American Academy of Dermatology, May, 2016
(Multicenter; archival; n=205 stage I-II melanomas) |
•
|
DecisionDx-Melanoma Class 1 was an independent predictor of RFS, DMFS and OS compared to the AJCC Individualized Melanoma Patient Outcome Prediction Tool and was 163% or greater than AJCC for all outcomes.
|
|
•
|
Adding the DecisionDx-Melanoma Class 1 result to AJCC staging improved sensitivity for identifying recurrence, distant metastasis or death by up to 22% compared to AJCC staging alone.
|
||
|
•
|
21% of cases had discordant risk prediction from DecisionDx-Melanoma Class 1 and AJCC tools, with the DecisionDx-Melanoma providing the more accurate prognosis for the majority of cases.
|
||
|
|
|
|
|
|
Gene expression profiling for molecular staging of cutaneous melanoma in patients undergoing sentinel lymph node biopsy
|
Journal of the American Academy of Dermatology, May, 2015
(Multicenter; archival; n=217 stage I-III melanomas all of whom underwent SLNB) |
•
|
DecisionDx-Melanoma Class 1 was an independent predictor of RFS, DMFS and OS with hazard ratios of 4.9, 3.9 and 4.7, respectively, and was 185% - 392% greater than SLNB.
|
|
•
|
DecisionDx-Melanoma Class 1 NPV for distant metastasis was 82% compared to 67% for SLNB.
|
||
|
•
|
DecisionDx-Melanoma Class 1 sensitivity was 85%, 84% and 85% compared to SLNB sensitivity of 35%, 38% and 29% for the endpoints of DFS, DMFS and OS.
|
||
|
|
|
|
|
|
Performance of a prognostic 31-GEP in an independent cohort of 523 cutaneous melanoma patients
|
BMC Cancer, February, 2018
(Multicenter; archival; n=523 stage I-III melanomas) |
•
|
DecisionDx-Melanoma Class 1A was an independent predictor of RFS and DMFS with hazard ratios of 3.8 and 5.3, respectively, and was between 146% and 408% greater than SLNB and tumor thickness.
|
|
•
|
NPV for MSS for Class 1A patients was 100% for Stage I, 100% for Stage II, 94% for Stage IIIA and 91% for Stage IIIB-C patients.
|
||
99
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Performance of a 31-GEP test in cutaneous melanomas of the head and neck
|
Head & Neck, January, 2019
(Multicenter; archival; n=157 melanomas of the head and neck region) |
•
|
DecisionDx-Melanoma Class 1 was an independent predictor of RFS, DMFS, OS and MSS with hazard ratios of 2.8, 2.8, 4.1 and 6.8, respectively compared to AJCC stage.
|
|
•
|
NPV for Class 1A for MSS was 98%.
|
||
|
|
|
|
|
|
Clinical Utility
|
|
|
|
|
Prospective, Multicenter Clinical Impact Evaluation of a 31-GEP Test for Management of Melanoma Patients
|
SKIN: The Journal of Cutaneous Medicine, March, 2018
(Multicenter, prospective; pre-test post-test methodology; n=247 patients) |
•
|
Post-test management plans changed by 49% compared to pre-test plans; 85% of Class 2 patients and 36% of Class 1 patients having a change in management (p<0.001).
|
|
•
|
Significant changes for imaging (p<0.001), request for laboratory work (p=0.04) and frequency of office visits (p<0.001).
|
||
|
|
|
|
|
|
Clinical impact of a 31-GEP test for cutaneous melanoma in 156 prospectively and consecutively tested patients
|
Current Medical Research and Opinion, September, 2016 (Multicenter; retrospective chart review, pre-test post-test methodology; n=156 patients)
|
•
|
Post-test management plans were recorded in 53% of patients, with 77% of Class 2 patients and 37% of Class 1 patients (p<0.0001) having a change in management.
|
|
•
|
94% of patient management changes were concordant with the risk indicated by the GEP test result (p<0.0001).
|
||
|
|
|
|
|
|
Impact of a 31-gene Expression Profiling Test for Cutaneous Melanoma on Dermatologists’ Clinical Management Decisions
|
Journal of Drugs in Dermatology, May, 2017 (Intended use pre-test post-test vignette methodology; n=170 participating dermatologists)
|
•
|
Risk appropriate management recommendations for implementing SLNB and imaging were more likely to be made following incorporation of DecisionDx-Melanoma test results (p<0.05).
|
|
•
|
Dermatologists changed their tumor thickness inflection point for implementing SLNB, oncology referral and imaging from 1.0mm to 0.7mm following a Class 2 DecisionDx-Melanoma test result.
|
||
|
|
|
|
|
|
Impact of Genetic Expression Profile on Decision-Making in Clinically Node Negative Melanoma Patients After Surgical staging
|
Journal of Drugs in Dermatology, February, 2018 (Single center, prospective study at Oregon Health and Science Center; n=91 patients)
|
•
|
DecisionDx-Melanoma test results were significantly associated with management of patients with Stage I or II melanoma by Dermatology (most often Class 1) or Surgical Oncology (most often Class 2) (p<0.05).
|
|
•
|
Decision-tree model derived from the treatment and clinical data found that DecisionDx-Melanoma class result accounted for 52% of management changes whereas AJCC stage accounted for 48%.
|
||
100
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Factors affecting dermatologists’ use of a 31-gene expression profiling test as an adjunct for predicting metastatic risk in cutaneous melanoma
|
Journal of Drugs in Dermatology, May, 2018
(Intended use pre-test post-test vignette methodology; n=181 participating dermatologists) |
•
|
The DecisionDx-Melanoma result had a significant impact on the likelihood that SLNB would be recommended for a patient with a T1b tumor.
|
|
•
|
The presence of ulceration increased the proportion of respondents who would recommend the test (p<0.001).
|
||
|
|
|
|
|
|
Management decisions made by physician assistants and nurse practitioners in cutaneous malignant melanoma patients: impact of a 31-GEP test
|
Journal of Drugs in Dermatology, November, 2018
(Intended use pre-test post-test vignette methodology; n=164 participating nurse practitioners and physician assistants) |
•
|
DecisionDx-Melanoma Class 1 results led to a significant decrease in the proportion of PA/NPs who would recommend SLNB, imaging or quarterly follow-up intervals, while Class 2 results led to significant increases in each (p<0.01 for 5 of 6 patient vignettes included in clinical impact survey).
|
|
|
|
|
|
|
Analytical Validity
|
|
|
|
|
Analytic validity of DecisionDx-Melanoma, a proprietary GEP test, for determining metastatic risk in melanoma patients
|
Diagnostic Pathology, February, 2018
(Inter-assay, inter-instrument, and inter-observer analysis; technical reliability in clinically tested melanoma specimens) |
•
|
Inter-assay concordance on 168 specimens was 99% and matched probability scores were significantly correlated (R2 = 0.96); inter-instrument concordance was 95% and matched probability scores were correlated (R2=0.99; p < 0.001).
|
|
•
|
A technical success rate of 98% was achieved for the test in 7,023 clinical cases.
|
||
DecisionDx-UM

Overview
At the time of diagnosis nearly all patients with uveal melanoma have no evidence of metastasis yet approximately 30% of uveal melanoma goes on to metastasize within three years and nearly 50% of uveal melanoma goes on to metastasize, overall. Traditional clinical staging and molecular diagnostic tests exist for uveal melanoma, but the lack of prospective studies of these tests, coupled with low accuracy, has resulted in these tests primarily being used for research purposes rather than for clinical management of patients in the United States. As a result, nearly all U.S. centers group patients into a single, high-risk treatment plan that included frequent, high intensity surveillance using advanced imaging, frequent clinical visits and encouragement to enroll in clinical studies.
DecisionDx-UM is a proprietary GEP test that helps healthcare providers predict the risk of metastasis in patients with uveal melanoma. We licensed the intellectual property for DecisionDx-UM from The Washington University in St. Louis, Missouri, or WUSTL, completed analytical validation and began marketing DecisionDx-UM in late 2009 for use in patients diagnosed with uveal melanoma without evidence of metastatic disease. DecisionDx-UM identifies which patients are at low risk for progression of their disease so that their physicians can appropriately de-escalate the level of care provided. In 2018, DecisionDx-UM was delivered to more than 1,400 patients, representing approximately 87% of the patients diagnosed in the United States annually.
Fourteen peer-reviewed publications supporting the clinical validity and utility of DecisionDx-UM have been conducted.
101
The Kaplan-Meier plot from the initial prospective, multi-center Collaborative Ocular Oncology Group, or COOG, study found a 97% NPV for risk of metastasis. This study also compared DecisionDx-UM to the traditional clinical and pathology factors as well as chromosome 3 status, which is an alternative molecular test to predict the risk of metastasis in uveal melanoma. As is shown in the Cox multivariate analysis, the only statistically significant factor in predicting a likelihood of metastasis was DecisionDx-UM.
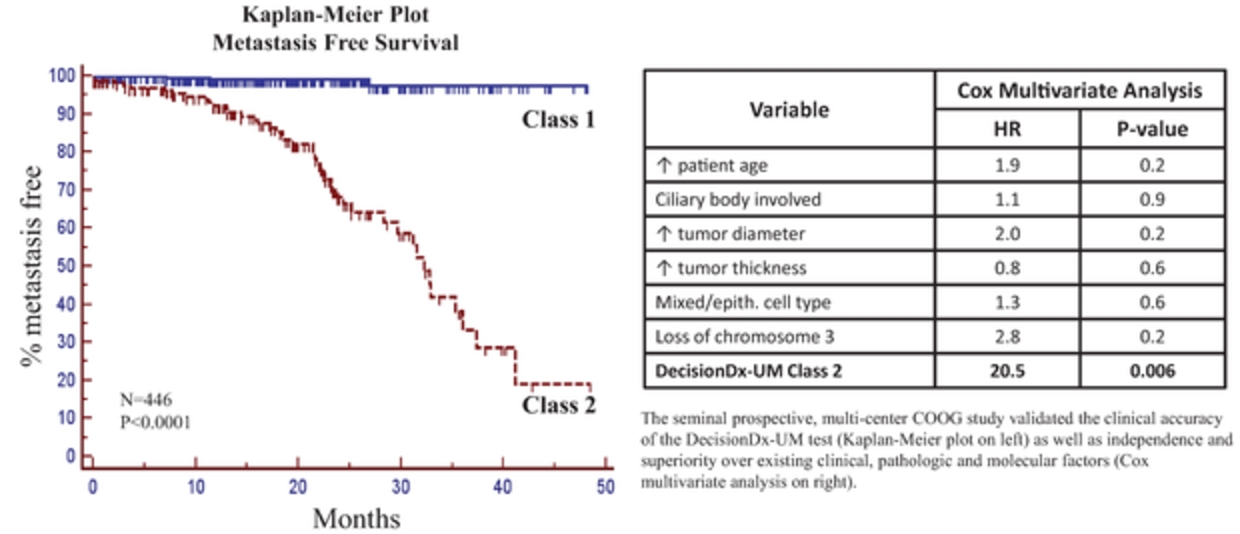
The data from the COOG study, as well as the consistency shown from the additional clinical validity studies, has supported wide spread adoption of DecisionDx-UM with more than 90% of the ocular oncology institutions in the United States ordering this test.
DecisionDx-UM has been used to guide treatment plan decisions regarding the intensity of a patient’s surveillance and management plan as well as clinical trial enrollment.
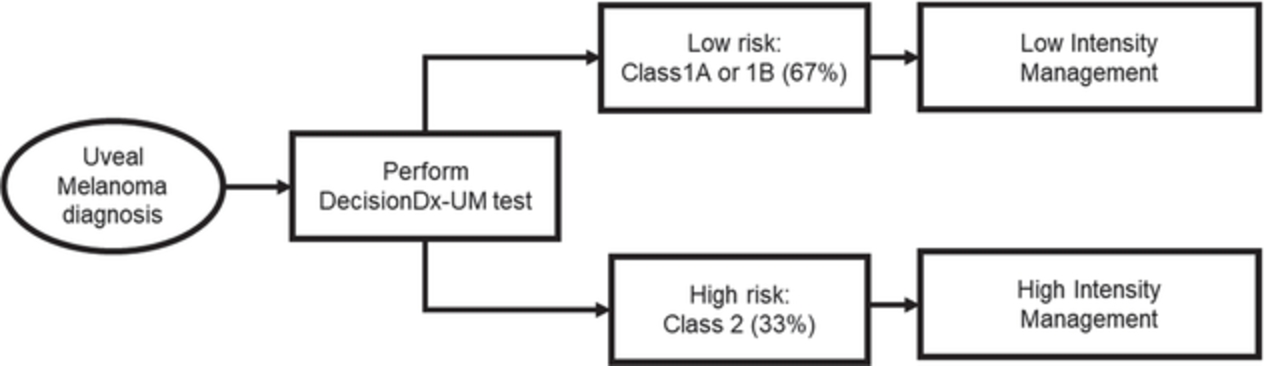
102
Summary of Our DecisionDx-UM Studies
The table below summarizes the DecisionDx-UM clinical studies that have been published to date:
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Clinical Validity
|
|
|
|
|
Collaborative Ocular Oncology Group report number 1: prospective validation of a multi-gene prognostic assay in uveal melanoma
|
Ophthalmology, August, 2012 (Multicenter, prospective, n=446)
|
•
|
DecisionDx-UM Class 1 was the only significant predictor of MFS (p<0.0001) in multivariate analysis, compared to patient age, ciliary body involvement, tumor thickness, tumor diameter, cell type, and chromosome 3 status.
|
|
•
|
NPV for Class 1 for MFS was 99%
|
||
|
•
|
MFS rates as 50-months were 97% for Class 1 and 20% for Class 2 patients (p<0.0001).
|
||
|
|
|
|
|
|
Do largest basal tumor diameter and the American Joint Committee on Cancer’s cancer staging influence prognostication by gene expression profiling in choroidal melanoma
|
American Journal of Ophthalmology, November, 2018
(Two-centers, retrospective, consecutively tested patients; n=293) |
•
|
NPV for Class 1A for MFS was 99%.
|
|
•
|
Three-year MFS rates were 99% for Class 1A, 90% for Class 1B, and 60% for Class 2 patients.
|
||
|
•
|
DecisionDx-UM was a significant predictor of metastasis in multivariate analysis (p=0.001) with increasing largest basal diameter.
|
||
|
|
|
|
|
|
Clinical performance and management outcomes with DecisionDx-UM in a prospective multi-center study
|
Journal of Oncology, June, 2016
(Multicenter, prospective; n=70) |
•
|
NPV for Class 1 for MFS was 95%
|
|
•
|
Three year MFS rates were 100% for Class 1 and 63% for Class 2 patients (p=0.003).
|
||
|
•
|
DecisionDx-UM was the most significant predictor of metastasis in multivariate analysis (p=0.016) compared to age, largest basal diameter, ciliary body involvement, and tumor thickness.
|
||
|
|
|||
|
Uveal melanoma: molecular pattern, clinical features, and radiation response
|
American Journal of Ophthalmology, August, 2012
(Single-center, retrospective; n=197) |
•
|
5-year disease specific survival, or DFS, rates were 93% for Class 1 patients and 38% for Class 2 patients (p<0.0001).
|
|
•
|
DecisionDx-UM Class 1 was the only significant predictor of DFS (p<0.0001) in multivariate analysis compared with cell type, age of patient at radiation, and pretreatment ultrasound measurement.
|
||
|
•
|
DecisionDx-UM Class 1 was the only significant predictor disease-specific mortality (p<0.0001) in multivariate analysis with cell type, age of patient at radiation, and pretreatment ultrasound measurement.
|
||
|
|
|
|
|
|
An accurate, clinically feasible multi-gene expression assay for predicting metastasis in uveal melanoma
|
Journal of Molecular Diagnostics, July, 2010
(Single-center, prospective (outcomes); n=172) |
•
|
MFS rates were significantly different between Class 1 and Class 2 patients (p<0.0001).
|
103
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Uveal melanoma: from lesion size and cell type to molecular class
|
Canadian Journal of Ophthalmology, June, 2012
(Independent, meta-analysis) |
•
|
DecisionDx-UM had the strongest predictive value for UM metastasis and mortality.
|
|
Gene expression profiling and PRAME status versus tumor-node-metastasis staging for prognostication in uveal melanoma
|
American Journal of Ophthalmology, November, 2018
(Single-center, retrospective consecutively tested patients; n=240) |
•
|
DecisionDx-UM was the most significant predictor of metastasis in multivariate analysis (p<0.0001) with PRAME status, TNM stage, and gender.
|
|
•
|
DecisionDx-UM was the most significant predictor of metastasis in multivariate analysis (p<0.0001) with PRAME status, largest basal diameter, tumor thickness, ciliary body involvement, and gender.
|
||
|
|
|
|
|
|
Prognostic implications of tumor diameter in association with gene expression profile for uveal melanoma
|
JAMA Ophthalmology, July, 2016
(Independent, retrospective, two centers, n=580 patients) |
•
|
NPV for Class 1 of MSS was 96%.
|
|
•
|
DecisionDx-UM was the most significant predictor of metastasis (p<0.001) in multivariate analysis compared to age, sex, tumor thickness, largest basal diameter, ciliary body involvement, and cell type.
|
||
|
|
|
|
|
|
Driver mutations in uveal melanoma: Associations with gene expression profile and patient outcomes
|
JAMA Ophthalmology, July, 2016
(Independent, retrospective, single center, n=81 patients) |
•
|
DecisionDx-UM was the most significant predictor of metastasis (p<0.001) in multivariate analysis compared to age, sex, ciliary body involvement, cell type, extraocular extension, largest basal diameter, tumor thickness, and mutational status.
|
|
•
|
DecisionDx-UM was the most significant predictor of melanoma-specific death (p<0.001) compared to age, sex, ciliary body involvement, cell type, extraocular extension, largest basal diameter, tumor thickness, and mutational status.
|
||
|
|
|
|
|
|
Independent prognostic significance of gene expression profile class and largest basal diameter of posterior uveal melanomas
|
American Journal of Ophthalmology, February, 2016
(Single-center, prospective; n=299) |
•
|
DecisionDx-UM was the most significant prognostic factor for UM mortality (p=0.0019) in multivariate analysis.
|
|
|
|
|
|
|
Sufficiency of FNAB aspirates of posterior uveal melanoma for cytologic versus GEP classification in 159 patients, and relative prognostic significance of these classifications
|
Graefes Archives in Clinical Experimental Ophthalmology, January, 2014
(Prospective; n=159) |
•
|
NPV for Class 1 for DFS was 95%.
|
|
•
|
Five-year disease-specific survival rates were 92% for Class 1 patients and 55% for Class patients (p=0.005).
|
||
|
|
|
|
|
104
|
Study
|
Peer-Reviewed Publications
(Methods) |
Main Findings
|
|
|
Clinical Utility
|
|
|
|
|
Clinical performance and management outcomes with the DecisionDx-UM GEP test in a prospective multi-center study
|
Journal of Oncology, June, 2016
(Multi-center, prospective; n=70) |
•
|
81% of Class 1 patients were prescribed low-intensity surveillance schedules (annual metastatic screening) compared to 0% of Class 2 patients.
|
|
•
|
100% of Class 2 patients were prescribed high intensity surveillance schedules (quarterly-biannual metastatic screening) compared to 19% of Class 1 patients (p<0.001).
|
||
|
|
|
|
|
|
Current clinical practice: differential Management of Uveal Melanoma in the Era of Molecular Tumor Analyses
|
Clinical Ophthalmology, December, 2014
(Multi-center, chart review, pre-test post-test methodology; n=88) |
•
|
100% of Medicare-eligible Class 1 patients had low-intensity surveillance schedules.
|
|
•
|
100% of Medicare-eligible Class 2 patients had high-intensity surveillance schedules (p<0.0001).
|
||
|
|
|
|
|
|
Analytical Validity
|
|
|
|
|
Gene Expression Profiling in Uveal Melanoma: Technical Reliability and Correlation and Correlation of Molecular Class with Pathologic Characteristics. Diagnostic Pathology
|
Diagnostic Pathology, August 2017
(Technical reliability studies and technical success in clinical testing (n=5,516)) |
•
|
Intra-assay, inter-assay (short-term and long-term), and inter-laboratory concordance experiments demonstrated 100% Class concordance and a high degree of correlation between discriminant scores.
|
|
•
|
Inter-instrument/operator concordance was 96% between Class 1 and 2.
|
||
|
•
|
96.3% technical success was achieved on fine-needle aspiration biopsy (FNAB) and formalin-fixed paraffin-embedded (FFPE) specimens in clinical testing.
|
||
|
|
|
|
|
|
An Accurate, Clinically Feasible Multi-Gene Expression Assay for Predicting Metastasis in Uveal Melanoma
|
Journal of Molecular Diagnostics, July, 2010
(Technical reliability studies) |
•
|
Technical success was 95% in 609 samples.
|
|
•
|
DecisionDx-UM was successful in 50/51 FNABs that had insufficient quantity for cytology.
|
||
|
•
|
100% Class concordance in tissue from FNABs and matched fresh-frozen tumor slices.
|
||
|
•
|
31/32 (97%) Class concordance in multiple tumor sections from 7 tumors.
|
||
Additional Ancillary Products
We also offer two next generation sequencing panels to complement our proprietary cutaneous and uveal melanoma products. When the U.S. Food and Drug Administration, or FDA, approved adjuvant BRAF/MEK inhibitor therapy for patients diagnosed with Stage III cutaneous melanoma, either at the time of diagnosis or subsequent progression, physicians ordering our tests requested tests to provide information on these specific genes. In order to provide a more complete product offering to physicians, we offer a 3-gene test to confirm BRAF, NRAS and kit status to inform drug therapy decisions. This 3-gene panel can be ordered at the same time as, or after, DecisionDx-Melanoma. For uveal melanoma, we offer a 7-gene panel that can also be ordered at the same time or subsequent to DecisionDx-UM. We also offer a proprietary GEP test for PRAME status of patients diagnosed with uveal melanoma, DecisionDx-PRAME.
105
Development Engine and Pipeline
We identify areas of significant unmet medical needs in the treatment of skin cancer and leverage our expertise in cancer genomics and data analytics to develop clinically actionable products. We focus first on previously unanswered medical questions, which we often identify as the result of the in-depth conversations our sales and medical affairs representatives have with physicians. We leverage our deep understanding of skin cancer clinical treatment pathways to identify areas of unmet medical need where better information can improve, and potentially transform, patient outcomes. Next, our team of scientists and laboratory technologists use the latest genomic methods to discover gene expression profiles, RNA, which are likely to impact treatment pathway decisions. Our bioinformatics team works collaboratively with our scientists to build proprietary algorithms based on machine-learning techniques to predict the risk of metastasis or recurrence based on the genomics of each individual patient’s tumor biology.
Once we have generated a product candidate that we believe will address an unmet medical need, we work to validate the product candidate through extensive testing of patient tissue samples combined with clinical outcomes data. We use both our extensive data bank of patient tumor samples and clinical outcomes data to run development and validation studies and conduct clinical studies to collect new samples in additional diseases. We publish our data regularly to drive adoption, reimbursement and guideline inclusion.
We have gained expertise in developing proprietary algorithms, conducting clinical studies and using the necessary instrumentation required for efficiently developing our pipeline products. We have used these development principles to explore multiple potential solutions, and have identified two principal areas to expand our pipeline: tests for patients with SCC and patients with suspicious pigmented lesions.
DecisionDx-SCC
The current treatment pathway for patients identified as having high-risk SCC suffers from a low PPV for risk of metastasis or recurrence as well as low sensitivity. As a result, many patients categorized as high risk received adjuvant therapy and other unnecessary medical and surgical interventions even though they would not have gone on to metastasize. Additionally, the low sensitivity means that many patients who will go on to metastasize are not identified as high risk and thus are not offered the benefits of adjuvant therapy and other interventions. To address this clinical need in SCC, we are developing DecisionDx-SCC, a proprietary GEP test designed to identify high-risk patients by demonstrating better PPV and sensitivity compared to traditional staging systems, while maintaining similar NPV. We believe this product candidate will enable more informed clinical decisions regarding adjuvant intervention and other management decisions. We have ongoing multi-center studies involving more than 46 U.S. centers. Our development analysis has identified several gene expression profile algorithms that exhibit significant differential expression between non-recurrent and recurrent cases. We believe that one of these algorithms may demonstrate statistically higher sensitivity and PPV as compared to traditional staging methods.
Based on the timing and results of our clinical validation studies, we intend to commercially launch DecisionDx-SCC in the second half of 2020.
106

DecisionDx for Suspicious Pigmented Lesions
A biopsy of a pigmented lesion may lead to an indeterminate diagnosis, in which case the treating physician generally leans towards making a conservative decision and assumes that the lesion is melanoma. To address this clinical need, we are developing a proprietary GEP test designed to be used as an adjunct to histopathology when the distinction between a benign lesion and melanoma is uncertain. Our analysis has identified several gene expression profile algorithms that we believe may be used to confirm whether a specimen is melanoma or benign lesion.
Based on the timing and results of our planned and ongoing clinical studies, we intend to commercially launch this product in the second half of 2020.
Our Commercial Channel
Sales and Marketing
Our sales and marketing efforts are currently focused on the United States skin cancer market. We employ a direct sales and marketing strategy to educate dermatologists, surgeons and other physicians on the clinical and economic benefits of our products. Our sales approach is highly technical, and our team is trained to articulate the scientific and clinical evidence behind our products and how they influence the clinical care pathway and ultimately improve patient outcomes. Our sales force is focused on educating and informing the entire patient care team, which consists of treating clinicians, nurses, laboratory and pathology personnel, and finance administrators, on the appropriate use and value of our test.
As of March 31, 2019, our sales and marketing team totaled 35 employees. Our sales team is focused on leading high clinical impact discussions with the treating clinician’s areas of interest, including clinical utility, patient outcomes, and supporting evidence. Our previous expansion to approximately 25 sales and marketing employees occurred in mid-2015 and showed significant promotion responsiveness over the past four years.
We increased our outside sales territories from 14 to 23 in the first quarter of 2019 and also added supporting inside sales associates and marketing staff. We believe that this increase in sales and marketing personnel will increase the adoption of our products as we educate physicians on the clinical benefits of our products as well as the expanded use of our products in guiding SLNB surgery recommendations. We may, in late 2019 or in 2020, add additional sales representatives and the associated sales support personnel in preparation for launch of our pipeline products. Based upon current knowledge, we believe that an optimally efficient dermatology salesforce to support these products ranges between 35 and 45 individuals.
DecisionDx-UM addresses a small cancer market, and patients are managed by a small group of ocular oncology surgeons, generally ophthalmology or retina trained specialists. We serve these patients and their physicians by providing highly technical interactions that focus on optimizing the appropriate use of our proprietary and ancillary products.
107
Medical Affairs
We also deploy an experienced medical affairs group to assist education of treating physicians and key opinion leaders, to identify and engage sites for our sponsored clinical studies and to evaluate collaborative study opportunities. Our medical affairs strategy complements our sales and marketing and clinical research operations efforts.
Our Laboratory Report
We design our test reports with input from physicians to provide an easy to read risk classification and present specific actionable information to enable improved treatment decisions between patients and their physicians. We update our test reports as new data become available that may impact physicians’ treatment decisions based on the use of our tests. For example, the most recent update to our DecisionDx-Melanoma report incorporates data from our prospective multicenter 1,421 patient study showing how our tests can predict the likelihood of a patient experiencing an SLN-positive result from the SLNB surgery.
The image below is an example of a current clinical report for DecisionDx-Melanoma:

Reimbursement
The primary source of revenue for our products is reimbursement from third-party payors, which includes government payors, such as Medicare and Medicaid, and commercial payors, such as insurance companies. Achieving broad coverage and reimbursement of our current products by third-party payors and continued Medicare coverage are key components of our financial success. De novo coverage by government and third-party payors for our pipeline tests will be important over time.
Government Payors
Medicare coverage is limited to items and services that are within the scope of a Medicare benefit category and that are reasonable and necessary for the diagnosis or treatment of an illness or injury. LCDs are made through an evidence-based process by Medicare Administrative Contractors, or MACs, with opportunities for public participation.
108
Palmetto issued a final LCD for DecisionDx-Melanoma, which became effective on December 3, 2018. Noridian adopted the same coverage policy as Palmetto on that date, and subsequently issued a final LCD on February 10, 2019. This LCD provides for coverage for patients who are eligible for the SLNB surgery with cutaneous melanoma tumors of 2.0 mm in thickness or less, and patients with clinically negative SLN basins who are being considered for the SLNB surgery to determine eligibility for adjuvant therapy. Melanomas less than or equal to 2.0 mm thick represent 86% of all melanomas. The median age at diagnosis is 63 years old, therefore the Medicare eligible population represents close to 50% of the addressable market.
Separately, Palmetto issued a final LCD for DecisionDx-UM, which became effective in July 2017, and Noridian issued a similar LCD that became effective in September 2017. The Noridian LCD provides for coverage to determine metastatic risk in connection with the management of a patient’s newly diagnosed uveal melanoma and to guide surveillance and referral to medical oncology for those patients. Similar to cutaneous melanoma the median age at diagnosis for uveal melanoma is estimated at 58-62 years old, therefore the Medicare eligible population represents close to 45% of the addressable market.
On May 17, 2019, the Centers for Medicare & Medicaid Services, or CMS, determined that DecisionDx-UM meets the criteria for existing advanced diagnostic laboratory test, or ADLT, status, also referred to as “existing ADLT” status. This means that beginning in 2021, the DecisionDx-UM Medicare rate will be set annually based upon the median private payor rate for the first half of the preceding calendar year. Specifically, the median private payor rate from January 1 to June 30, 2019 will be used to set the Medicare rate for the calendar year 2021. From May 17, 2019 through December 31, 2020, our rate will be set by Noridian, our local MAC. Also, on May 17, 2019, CMS determined that DecisionDx-Melanoma meets the criteria for “new ADLT” status. This means that from July 1, 2019 through March 31, 2020 the Medicare reimbursement rate will equal the initial list price of $7,193.00. The rate for April 1, 2020 through December 31, 2020 will be calculated based upon the median private payor rate from July 1, 2019 to November 30, 2019. Accordingly, beginning in 2021, the rate will be set annually based upon the median private payor rate for the first half of the preceding calendar year. If CMS determines the list charge amount for DecisionDx-Melanoma was greater than 130% of the weighted median of private payor rates, CMS will recoup from us the difference between the actual list charge and 130% of the weighted median.
Commercial Third-Party Payors
We are actively engaged in efforts to achieve broad coverage and reimbursement for our current and future products, followed by contracting with commercial payors. Achieving positive coverage reduces the need for appeals and reduces failures to collect from the patient’s commercial payor. Even with positive coverage decisions, we still experience delays in time to payment. Achieving in-network contracts with third-party payors can shorten the time required to receive payments. Implementing our strategy includes our managed care and medical affairs teams educating third-party payors regarding our strong clinical utility and outcomes data, which we believe validates the value of our products and will persuade more third-party payors to provide value-based reimbursement.
We have broad positive policy coverage for our DecisionDx-UM test, have executed contracts with certain commercial payors and anticipate increases in contracting in 2019 and 2020. We also have positive policy recommendations from many third-party technical assessment review groups.
We are beginning our coverage discussions for DecisionDx-Melanoma and anticipate increases in contracting and coverage in 2019 and 2020.
Dependence on Third-Party Payors
We receive a substantial portion of our revenue from a small number of third-party payors, primarily Medicare, BlueCross BlueShield affiliates and Medicare Advantage plans. Our revenue from patients covered by Medicare, Medicare Advantage plans, United Healthcare and BlueCross BlueShield plans, as a percentage of total revenue, was 36%, 17%, 12% and 18%, respectively, for the year ended December 31, 2018. BlueCross BlueShield plans and Medicare Advantage plans represent an aggregation of multiple payors making independent reimbursement decisions; however, these plans often base reimbursement decisions on common guidelines which can influence multiple plans simultaneously.
109
Competition
We are focused on providing high value diagnostic and prognostic solutions for dermatological cancers. We believe that there is limited existing competition for our products that provide evidence-based genomic solutions to physicians and their patients.
We believe the principal competitive factors in our target markets include:
| • | Proprietary, disciplined approach to genomic analysis including the use of proprietary deep learning, machine learning and other techniques to identify and optimize gene selection and algorithmic approaches to answer the clinically important questions with accuracy tests. This involves the ability to design and efficiently conduct the right clinical studies at the right time; |
| • | Research and development investments to document the quality, quantity, consistency and strength of the clinical validity data, the impact our products have on clinical use, and demonstration of net health outcome improvement that reduce health system costs; |
| • | Maintaining a strong reputation with the treating physician by providing consistent, transparent, clinically relevant information that will improve the appropriate management of their patients; |
| • | Ease of use in accessing our products, reimbursement support for the physician and their patient and laboratory reports that clearly communicate the clinically relevant data points; |
| • | Demonstrated ability to work with, and secure coverage and reimbursement from, governmental and commercial payors; |
| • | Ability to efficiently commercialize pipeline products to the same customer base as our current products. |
We believe we compete favorably on the factors described above.
Our principal competition for DecisionDx-Melanoma is existing traditional clinical and pathology staging criteria. While some clinical and pathology criteria have changed over time, this approach has been the standard of care in the United States for many years, and physicians may be unwilling to accept the validity of the published data adopt our test until it has become incorporated into national guidelines. In addition, we may, in the near future, face competition from a limited number of companies who are working in this disease space such as Neracare and Skylinedx.
DecisionDx-UM competes with a subsidiary of LabCorp and several academic laboratories all of which have had tests available for several years. To date, our data has demonstrated that DecisionDx-UM is clinically and statistically superior to these products.
We are unaware of late-stage work being performed to develop and validate a product that would compete with our pipeline product for SCC. We believe that the primary competitor for our SCC product, if we are successful in validating a clinically useful product, will initially be existing traditional clinical and pathology staging criteria.
We are aware of competitors that would likely compete with our pipeline product for suspicious pigmented lesions such as Myriad Genetics. If we are successful in validating a clinically useful product, then we expect to compete favorably with them.
Laboratory Operations
The Laboratory Developed Tests, or LDTs, that we commercialize are conducted in our CAP accredited, CLIA-certified facility in Phoenix, Arizona. Within this 12,000 square foot facility, we perform all laboratory procedures involved in our tests from receiving a requisition form to issuance of the final test result. Our most recent capacity plan analysis indicates that we can process up to 60,000 proprietary GEP test orders annually for our proprietary tests prior to expanding the wet lab facility although we would require additional space to increase sample intake, clinical research operations, IT and administration. We performed more than 12,800 proprietary GEP tests in 2018. We moved into this facility in April 2016. We are able to provide our proprietary DecisionDx-Melanoma and DecisionDx-UM products for patients in all fifty states including those that require specific, additional, out-of-state lab licenses or qualifications such as New York, California, Pennsylvania, Maryland, and Rhode Island. Upon receipt, orders and samples are processed through an automated laboratory
110
information management system which, in addition to tracking sample chain of custody, initiates and tracks accessioning, sample eligibility, technical data generation, algorithmic analysis and results reporting. The majority of samples are received as paraffin embedded sections from a formalin fixed tissue specimen.
The laboratory facility houses all functions related to quality control and assurance, licensing, accreditation and regulatory compliance. Our quality management program ensures the quality of our laboratory services and products through continuous monitoring of a broad range of key performance indicators including technical, customer service and cybersecurity metrics. Through this program, we promote a philosophy of continuous improvement based upon adherence to validated standards.
Raw Materials and Suppliers
We procure reagents, equipment, chips/cards and other materials used to perform our tests from sole suppliers such as Thermo, Inc., and Qiagen, Inc. Some of these items are unique to these suppliers and vendors. While we have developed alternate sourcing strategies for these materials and vendors and while we have experienced no business interruption due to an inability to source these materials, we cannot be certain whether these strategies will be effective or whether alternative sources will be available when we need them. If these suppliers can no longer provide us with the materials we need to perform our test services, they do not meet our quality specifications, or we cannot obtain acceptable substitute materials, our business would be negatively affected.
License Agreement with The Washington University
In November 2009, we entered into a license agreement, or the License Agreement, with WUSTL to license certain patent rights and technical information from WUSTL for the development of melanoma products, or the Products, and services, or the Services. The rights licensed under this agreement are used in DecisionDx-UM only.
Under the License Agreement, we obtain an exclusive, worldwide, royalty-bearing license to certain patent rights owned by WUSTL, or the Patent Rights, and a non-exclusive, worldwide license to certain technical information and research property owned by WUSTL, with the right to grant sublicenses under certain conditions, to develop the Products and Services. WUSTL retains the right to use the Patent Rights for research purposes.
The Patent Rights that we license pursuant to the License Agreement have been generated through the use of U.S. government funding and are therefore subject to certain federal regulations. See “Risk Factors—Risks Related to Intellectual Property—Our in-licensed intellectual property has been discovered through government funded programs and thus may be subject to federal regulations such as “march-in” rights, certain reporting requirements and a preference for U.S.-based companies, and compliance with such regulations may limit our exclusive rights, and limit our ability to contract with non-U.S. manufacturers.”
Under the License Agreement, we are required to use best efforts to carry out the activities under an agreed-upon development plan, or the Development Plan, and meet any and all milestones set forth in the Development Plan. We are required to make milestone payments to WUSTL upon successful completion of development and commercialization milestones as set forth in the Development Plan. For each Product or Service that receives FDA approval, premarket approval or premarket notification, we are obligated to make a milestone payment to WUSTL in the mid-four digits. For the issuance of the first U.S. patent and the first foreign patent, we are obligated to make aggregate milestone payments to WUSTL in the low-five digits.
Under the License Agreement, we were obligated to pay WUSTL an initial license issue fee in the low-five digits. We are also obligated to make royalty payments to WUSTL equal to (i) a percentage in the mid-single digits of our and any of our affiliates’ or sub-licensees’ net sales of Products and (ii) a percentage in the low-single digits of our and any of our affiliates’ or sub-licensees’ revenue from Services. We are also obligated to make royalty payments to WUSTL in the low-to-mid single digit percentage of net sales, with minimum royalty payments to WUSTL every six-month period following the first commercial sale.
The term of the License Agreement will continue for ten years following the last-to-expire valid claim relating to the Patent Rights, unless terminated earlier. WUSTL may terminate the License Agreement upon written notice in the event of (i) our material breach if such breach remains uncured for 90 days, (ii) the exercise of certain rights by us with respect to the Patent Rights and/or the licensed technical information outside the scope of the License Agreement, or (iii) for certain insolvency-related events. We may terminate the License Agreement without cause upon written notice to WUSTL and payment of any amount due to WUSTL under the License Agreement.
111
Intellectual Property
Our core technology for our products is related to methods and devices for analysis of genetic expression. Using this technology, we are able to provide a more accurate prediction of a patient’s metastatic risk as compared to other methods. We have secured and continue to pursue intellectual property rights globally, including through patent protection covering analysis of metastasis in cutaneous melanoma, as well as treatment of cutaneous squamous cell carcinoma. We also rely on trademarks, trade secrets, know-how, continuing technological innovation and potential in-licensing opportunities to develop and maintain our proprietary position. For more information, please see “Risk Factors—Risks Related to Intellectual Property.”
Patents and Patent Applications
We have developed a global patent portfolio that as of December 31, 2018, comprised of six issued patents, including four issued U.S. patents, and 13 pending patent applications, including three U.S. applications. Our patent portfolio consists of two owned patent families and an exclusively in-licensed portfolio from WUSTL, which includes seven pending and issued patents across two families. This global patent portfolio has filing dates ranging from 2009 to 2018, and therefore are projected to expire between 2029 and 2038, subject to any patent term extension or patent term adjustment that might be available in a particular jurisdiction. The owned and licensed families contain issued patents and pending applications that relate to devices, systems, and methods for macromolecular analysis, and reflect our active and ongoing research programs. The commercial foci of these patent families are discussed below.
|
Commercial Focus
|
Number of Issued Patents
and Pending Patent Applications |
|
Methods for predicting risk of metastasis in cutaneous melanoma
|
10
|
|
Compositions and methods for detecting cancer metastasis
|
5
|
|
Methods of diagnosing and treating patients with cutaneous squamous cell carcinoma
|
1
|
|
Method for predicting risk of metastasis
|
2
|
|
Method of predicting risk for recurrence for soft tissue sarcoma
|
1
|
Individual patents extend for varying periods depending on the date of filing of the patent application or the date of patent issuance and the legal term of patents in the countries in which they are obtained. Generally, patents issued for regularly filed applications in the United States are granted a term of 20 years from the earliest effective non-provisional filing date. In addition, in certain instances, a patent term can be extended to recapture a period due to delay by the United States Patent and Trademark Office, or USPTO in issuing the patent as well as a portion of the term effectively lost as a result of the FDA regulatory review period. However, as to the FDA component, the restoration period cannot be longer than five years and the total patent term including the restoration period must not exceed 14 years following FDA approval. The duration of foreign patents varies in accordance with provisions of applicable local law, but typically is also 20 years from the earliest effective non-provisional filing date. However, the actual protection afforded by a patent varies on a product-by-product basis, from country to country, and depends upon many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patent.
Trademarks and Trade Secrets
As of December 31, 2018, our U.S. trademark portfolio contained four trademark registrations.
We rely upon trade secrets, know-how, continuing technological innovation and potential in-licensing opportunities to develop and maintain our competitive position. We seek to protect our intellectual property and proprietary technology, in part, by entering into confidentiality agreements and intellectual property assignment agreements with our employees, consultants, corporate partners and, as applicable, our advisors. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with an employee or a third party. These agreements may be breached, and we may not have adequate remedies for any breach. We additionally seek to preserve the integrity and confidentiality of our data and trade secrets, such as our proprietary algorithms, by maintaining the physical security of our premises and physical and electronic security
112
of our information technology systems. While we have confidence in these individuals, organizations and systems, agreements or security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our commercial partners, collaborators, employees and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
Government Regulation and Product Approval
Regulations
Clinical Laboratory Improvement Amendments of 1988
As a clinical reference laboratory, we are required to hold certain federal, state and local licenses, certifications and permits to conduct our business. Under CLIA, we are required to hold a certificate applicable to the type of laboratory tests we perform and to comply with standards applicable to our operations, including test processes, personnel, facilities administration, equipment maintenance, recordkeeping, quality systems and proficiency testing. We must maintain CLIA compliance and certification to be eligible to bill for diagnostic services provided to Medicare beneficiaries.
To renew our CLIA certificate, we are subject to survey and inspection every two years to assess compliance with program standards. Because we are a College of American Pathologists, or CAP, accredited laboratory, CMS does not perform this survey and inspection and relies on our CAP survey and inspection. We may also be subject to additional unannounced inspections. The regulatory and compliance standards applicable to the testing we perform may change over time, and any such changes could have a material effect on our business.
Penalties for non-compliance with CLIA requirements include suspension, limitation or revocation of the laboratory’s CLIA certificate, as well as directed plan of correction, state on-site monitoring, civil money penalties, civil injunctive suit or criminal penalties.
State Laboratory Licensing
In addition to federal certification requirements of laboratories under CLIA, CLIA provides that states may adopt laboratory regulations and licensure requirements that are more stringent than those under federal law. Such laws, among other things, establish standards for the day-to-day operation of a clinical reference laboratory, including the training and skills required of personnel and quality control. We currently provide laboratory services in all 50 states and maintain out-of-state laboratory licenses in New York, California, Maryland, Pennsylvania and Rhode Island.
Multiple states require the licensure of out-of-state laboratories that accept specimens from those states. Because we receive specimens from New York, our clinical reference laboratory is required to be licensed by New York, under New York laws and regulations. New York law also mandates proficiency testing for laboratories licensed under New York state law, regardless of whether such laboratories are located in New York. If a laboratory is out of compliance with New York statutory or regulatory standards, the New York Department of Health, or NYDOH, may suspend, limit, revoke or annul the laboratory’s New York license, censure the holder of the license, or assess civil money penalties. We have received written approval from NYDOH to offer our proprietary DecisionDx-Melanoma DecisionDx-UM and DecisionDx-PRAME products in New York. If we were to be found out of compliance with New York laboratory requirements, we could be subject to such sanctions, which could harm our business.
Federal Oversight of Laboratory Developed Tests
The laws and regulations governing the marketing of diagnostic products are evolving, extremely complex, and in many instances, there are no significant regulatory or judicial interpretations of these laws and regulations. Clinical laboratory tests are regulated under CLIA, as administered by CMS, as well as by applicable state laws. In addition, the Federal Food, Drug and Cosmetic Act, or FDCA, defines a medical device to include any instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including a component part, or accessory, intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals. Our in vitro testing products are considered by the FDA to be subject to regulation as medical devices. Among other things, pursuant
113
to the FDCA and its implementing regulations, the FDA regulates the research, testing, manufacturing, safety, labeling, storage, recordkeeping, pre-market clearance or approval, marketing and promotion, and sales and distribution of medical devices in the United States to ensure that medical products distributed domestically are safe and effective for their intended uses. In addition, the FDA regulates the export of medical devices manufactured in the United States to international markets.
Although the FDA has statutory authority to assure that medical devices are safe and effective for their intended uses, the FDA has generally exercised its enforcement discretion and not enforced applicable regulations with respect to in vitro diagnostics that are designed, manufactured, and used within a single laboratory for use only in that laboratory. These tests are referred to as LDTs. As a result, we believe our diagnostic services are currently subject to the FDA’s enforcement discretion and are not subject to the FDA’s oversight. However, reagents, instruments, software or components provided by third parties and used to perform LDTs may be subject to regulation.
In recent years, FDA has stated its intention to modify its enforcement discretion policy with respect to LDTs. For example, on July 31, 2014, the FDA notified Congress of its intent to modify, in a risk-based manner, its policy of enforcement discretion with respect to LDTs. On October 3, 2014, the FDA issued two draft guidance documents entitled “Framework for Regulatory Oversight of Laboratory Developed Tests (LDTs),” or the Framework Guidance, and “FDA Notification and Medical Device Reporting for Laboratory Developed Tests (LDTs),” or the Reporting Guidance. The Framework Guidance states that FDA intends to modify its policy of enforcement discretion with respect to LDTs in a risk-based manner consistent with the classification of medical devices generally in Classes I through III. The Reporting Guidance would further enable FDA to collect information regarding the LDTs currently being offered for clinical use through a notification process, as well as to enforce its regulations for reporting safety issues and collecting information on any known or suspected adverse events related to the use of an LDT.
Although the FDA halted finalization of the guidance in November 2016 to allow for further public discussion on an appropriate oversight approach to LDTs and to give congressional authorizing committees the opportunity to develop a legislative solution, the FDA could ultimately modify its current approach to LDTs in a way that would subject our products marketed as LDTs to the enforcement of regulatory requirements.
Medical Device Regulatory Framework
Although we currently market our proprietary testing products as LDTs, which are currently subject to enforcement discretion, we could be subject to more onerous FDA compliance obligations in the future. Specifically, if the FDA begins to actively regulate LDTs, then, unless an exemption applies, each new or significantly modified medical device we seek to commercially distribute in the United States will require either a premarket notification to the FDA requesting permission for commercial distribution under Section 510(k) of the FDCA, also referred to as a 510(k) clearance, or approval from the FDA of a premarket approval, or PMA, application. Both the 510(k) clearance and PMA processes can be resource intensive, expensive, and lengthy, and require payment of significant user fees.
Device Classification
Under the FDCA, medical devices are classified into one of three classes-Class I, Class II or Class III depending on the degree of risk associated with each medical device and the extent of control needed to provide reasonable assurances with respect to safety and effectiveness.
Class I includes devices with the lowest risk to the patient and are those for which safety and effectiveness can be reasonably assured by adherence to a set of FDA regulations, referred to as the General Controls for Medical Devices, which require compliance with the applicable portions of the FDA’s Quality System Regulation, facility registration and product listing, reporting of adverse events and malfunctions, and appropriate, truthful and non-misleading labeling and promotional materials. Some Class I devices also require premarket clearance by the FDA through the 510(k) premarket notification process described below. Most Class I products are exempt from the premarket notification requirements.
Class II devices are those that are subject to the General Controls, and Special Controls as deemed necessary by the FDA to ensure the safety and effectiveness of the device. These Special Controls can include performance standards, patient registries, FDA guidance documents and post-market surveillance. Most Class II devices are
114
subject to premarket review and clearance by the FDA. Premarket review and clearance by the FDA for Class II devices is accomplished through the 510(k) premarket notification process.
Class III devices include devices deemed by the FDA to pose the greatest risk such as life-supporting or life-sustaining devices, or implantable devices, in addition to those deemed novel and not substantially equivalent following the 510(k) process. The safety and effectiveness of Class III devices cannot be reasonably assured solely by the General Controls and Special Controls described above. Therefore, these devices are subject to the PMA application process, which is generally more costly and time-consuming than the 510(k) process. Through the PMA application process, the applicant must submit data and information demonstrating reasonable assurance of the safety and effectiveness of the device for its intended use to the FDA’s satisfaction. Accordingly, a PMA typically includes, but is not limited to, extensive technical information regarding device design and development, pre-clinical and clinical trial data, manufacturing information, labeling and financial disclosure information for the clinical investigators in device studies. The PMA application must provide valid scientific evidence that demonstrates to the FDA’s satisfaction a reasonable assurance of the safety and effectiveness of the device for its intended use.
The Investigational Device Process
In the United States, absent certain limited exceptions, human clinical trials intended to support medical device clearance or approval require an investigational device exemption, or IDE, application. Some types of studies deemed to present “non-significant risk” are deemed to have an approved IDE once certain requirements are addressed and Institutional Review Board, or IRB, approval is obtained. If the device presents a “significant risk” to human health, as defined by the FDA, the sponsor must submit an IDE application to the FDA and obtain IDE approval prior to commencing the human clinical trials. The IDE application must be supported by appropriate data, such as animal and laboratory testing results, showing that it is safe to test the device in humans and that the testing protocol is scientifically sound. Generally, clinical trials for a significant risk device may begin once the IDE application is approved by the FDA and the study protocol and informed consent are approved by appropriate IRBs at the clinical trial sites. Submission of an IDE will note necessarily result in the ability to commence clinical trials, and although the FDA’s approval of an IDE allows clinical testing to go forward for a specified number of subjects, it does not bind the FDA to accept the results of the trial as sufficient to prove the product’s safety and efficacy, even if the trial meets its intended success criteria.
All clinical trials must be conducted in accordance with the FDA’s IDE regulations that govern investigational device labeling, prohibit promotion and specify an array of recordkeeping, reporting and monitoring responsibilities of study sponsors and study investigators. Clinical trials must further comply with the FDA’s good clinical practice regulations for IRB approval and for informed consent and other human subject protections. Required records and reports are subject to inspection by the FDA. The results of clinical testing may be unfavorable, or, even if the intended safety and efficacy success criteria are achieved, may not be considered sufficient for the FDA to grant marketing approval or clearance of a product. The commencement or completion of any clinical trial may be delayed or halted, or be inadequate to support approval of a PMA application, for numerous reasons.
The 510(k) Clearance Process
Under the 510(k) clearance process, the manufacturer must submit to the FDA a premarket notification, demonstrating that the device is “substantially equivalent” to a legally marketed predicate device. A predicate device is a legally marketed device that is not subject to a PMA, i.e., a device that was legally marketed prior to May 28, 1976 (pre-amendments device) and for which a PMA is not required, a device that has been reclassified from Class III to Class II or I, or a device that was previously found substantially equivalent through the 510(k) process. To be “substantially equivalent,” the proposed device must have the same intended use as the predicate device, and either have the same technological characteristics as the predicate device or have different technological characteristics and not raise different questions of safety or effectiveness than the predicate device. Clinical data is sometimes required to support substantial equivalence.
After a 510(k) premarket notification is submitted, the FDA determines whether to accept it for substantive review. If it lacks necessary information for substantive review, the FDA will refuse to accept the 510(k) notification. If it is accepted for filing, the FDA begins a substantive review. By statute, the FDA is required to complete its review of a 510(k) notification within 90 days of receiving the 510(k) notification. As a practical
115
matter, clearance often takes longer, and clearance is never assured. Although many 510(k) premarket notifications are cleared without clinical data, the FDA may require further information, including clinical data, to make a determination regarding substantial equivalence, which may significantly prolong the review process. If the FDA agrees that the device is substantially equivalent, it will grant clearance to commercially market the device.
If the FDA determines that the device is not “substantially equivalent” to a predicate device, or if the device is automatically classified into Class III, the device sponsor must then fulfill the much more rigorous premarketing requirements of the PMA approval process, or seek reclassification of the device through the de novo process. The de novo classification process is an alternate pathway to classify medical devices that are automatically classified into Class III but which are low to moderate risk. A manufacturer can submit a petition for direct de novo review if the manufacturer is unable to identify an appropriate predicate device and the new device or new use of the device presents a moderate or low risk. De novo classification may also be available after receipt of a “not substantially equivalent” letter following submission of a 510(k) to FDA.
After a device receives 510(k) clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a new or major change in its intended use, will require a new 510(k) clearance or, depending on the modification, could require a PMA application. The FDA requires each manufacturer to determine whether the proposed change requires a new submission in the first instance, but the FDA can review any such decision and disagree with a manufacturer’s determination. Many minor modifications are accomplished by a letter-to-file in which the manufacture documents the change in an internal letter-to-file. The letter-to-file is in lieu of submitting a new 510(k) to obtain clearance for such change. The FDA can always review these letters to file in an inspection. If the FDA disagrees with a manufacturer’s determination regarding whether a new premarket submission is required for the modification of an existing 510(k)-cleared device, the FDA can require the manufacturer to cease marketing and/or recall the modified device until 510(k) clearance or approval of a PMA application is obtained. In addition, in these circumstances, the FDA can impose significant regulatory fines or penalties for failure to submit the requisite application(s).
The PMA Approval Process
Following receipt of a PMA application, the FDA conducts an administrative review to determine whether the application is sufficiently complete to permit a substantive review. If it is not, the agency will refuse to file the PMA. If it is, the FDA will accept the application for filing and begin the review. The FDA has 180 days to review a filed PMA application, although the review of an application more often occurs over a significantly longer period of time. During this review period, the FDA may request additional information or clarification of information already provided, and the FDA may issue a major deficiency letter to the applicant, requesting the applicant’s response to deficiencies communicated by the FDA.
Before approving or denying a PMA, an FDA advisory committee may review the PMA at a public meeting and provide the FDA with the committee’s recommendation on whether the FDA should approve the submission, approve it with specific conditions, or not approve it. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
Prior to approval of a PMA, the FDA may conduct inspections of the clinical trial data and clinical trial sites, as well as inspections of the manufacturing facility and processes. Overall, the FDA review of a PMA application generally takes between one and three years, but may take significantly longer. The FDA can delay, limit or deny approval of a PMA application for many reasons, including:
| • | the device may not be shown safe or effective to the FDA’s satisfaction; |
| • | the data from pre-clinical studies and/or clinical trials may be found unreliable or insufficient to support approval; |
| • | the manufacturing process or facilities may not meet applicable requirements; and |
| • | changes in FDA approval policies or adoption of new regulations may require additional data. |
If the FDA evaluation of a PMA is favorable, the FDA will issue either an approval letter, or an approvable letter, the latter of which usually contains a number of conditions that must be met in order to secure final approval of the PMA. When and if those conditions have been fulfilled to the satisfaction of the FDA, the agency will issue a PMA approval letter authorizing commercial marketing of the device, subject to the
116
conditions of approval and the limitations established in the approval letter. If the FDA’s evaluation of a PMA application or manufacturing facilities is not favorable, the FDA will deny approval of the PMA or issue a not approvable letter. The FDA also may determine that additional tests or clinical trials are necessary, in which case the PMA approval may be delayed for several months or years while the trials are conducted and data is submitted in an amendment to the PMA, or the PMA is withdrawn and resubmitted when the data are available. The PMA process can be expensive, uncertain and lengthy and a number of devices for which the FDA approval has been sought by other companies have never been approved by the FDA for marketing.
New PMA applications or PMA supplements are required for modification to the manufacturing process, equipment or facility, quality control procedures, sterilization, packaging, expiration date, labeling, device specifications, ingredients, materials or design of a device that has been approved through the PMA process. PMA supplements often require submission of the same type of information as an initial PMA application, except that the supplement is limited to information needed to support any changes from the device covered by the approved PMA application and may or may not require as extensive technical or clinical data or the convening of an advisory panel, depending on the nature of the proposed change.
In approving a PMA application, as a condition of approval, the FDA may also require some form of post-approval study or post-market surveillance, whereby the applicant conducts a follow-up study or follows certain patient groups for a number of years and makes periodic reports to the FDA on the clinical status of those patients when necessary to protect the public health or to provide additional or longer term safety and effectiveness data for the device. The FDA may also approve a PMA application with other post-approval conditions intended to ensure the safety and effectiveness of the device, such as, among other things, restrictions on labeling, promotion, sale, distribution and use. New PMA applications or PMA supplements may also be required for modifications to any approved diagnostic tests, including modifications to our manufacturing processes, device labeling and device design, based on the findings of post-approval studies.
Federal and State Physician Self-Referral Prohibitions
We are subject to the federal physician self-referral prohibitions, commonly known as the Stark Law, and to comparable state laws. Together these restrictions generally prohibit us from billing a patient or any governmental or private payer for certain designated health services, including clinical laboratory services, when the physician ordering the service, or any member of such physician’s immediate family, has a financial interest, such as an ownership or investment interest in or compensation arrangement with us, unless the arrangement meets an exception to the prohibition.
Sanctions for a Stark Law violation include the following:
| • | denial of payment for the services provided in violation of the prohibition; |
| • | refunds of amounts collected by an entity in violation of the Stark Law; |
| • | a civil penalty of up to $24,748 for each bill or claim for a service arising out of the prohibited referral; |
| • | the imposition of up to three times the amounts for each item or service wrongfully claimed; |
| • | possible exclusion from federal healthcare programs, including Medicare and Medicaid; and |
| • | a civil penalty of up to $164,992 for each arrangement or scheme that the parties know (or should know) has the principal purpose of circumventing the Stark Law’s prohibition. |
These prohibitions apply regardless of any intent by the parties to induce or reward referrals or the reasons for the financial relationship and the referral. In addition, knowing violations of the Stark Law may also serve as the basis for liability under the federal False Claims Act, or the FCA, which can result in additional civil and criminal penalties.
Federal and State Anti-Kickback Laws
The federal Anti-Kickback Statute, or the AKS, makes it a felony for a person or entity, including a clinical laboratory, to knowingly and willfully offer, pay, solicit or receive any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, in order to induce business that is reimbursable under any federal health care program. A violation of the AKS may result in imprisonment for up to ten years and fines of up to $100,000
117
for each violation and administrative civil money penalties of $100,000 plus up to three times the amount of the remuneration paid. Convictions under the AKS result in mandatory exclusion from federal health care programs for a minimum of five years. In addition, The U.S. Department of Health and Human Services, or HHS, has the authority to impose civil assessments and fines and to exclude health care providers and others engaged in prohibited activities from Medicare, Medicaid and other federal health care programs. In addition, the government may assert that a claim that includes items or services resulting from a violation of the AKS constitutes a false or fraudulent claim under the FCA, which is discussed in greater detail below. Additionally, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
Although the AKS applies only to items and services reimbursable under any federal health care program, a number of states have passed statutes substantially similar to the AKS that apply to all payors. Penalties of such state laws include imprisonment and significant monetary fines.
Federal and state law enforcement authorities scrutinize arrangements between health care providers and potential referral sources to ensure that the arrangements are not designed as a mechanism to induce patient care referrals or induce the purchase or prescribing of particular products or services. Generally, courts have taken a broad interpretation of the scope of the AKS, holding that the statute may be violated if merely one purpose of a payment arrangement is to induce referrals or purchases.
In addition to statutory exceptions to the AKS, regulations provide for a number of safe harbors. If an arrangement meets the provisions of a safe harbor, it is deemed not to violate the AKS. An arrangement must fully comply with each element of an applicable safe harbor in order to qualify for protection.
Failure to meet the requirements of the safe harbor, however, does not render an arrangement illegal. Rather, the government may evaluate such arrangements on a case-by-case basis, taking into account all facts and circumstances.
Other Federal and State Health Care Laws
In addition to the requirements discussed above, several other health care fraud and abuse laws could have an effect on our business. For example, provisions of the Social Security Act permit Medicare and Medicaid to exclude an entity that charges the federal health care programs substantially in excess of its usual charges for its services. The terms “usual charge” and “substantially in excess” are subject to varying interpretations.
The FCA prohibits, among other things, a person from knowingly presenting, or causing to be presented, a false or fraudulent claim for payment or approval and from, making, using, or causing to be made or used, a false record or statement material to a false or fraudulent claim in order to secure payment or retaining an overpayment by the federal government. In addition to actions initiated by the government itself, the statute authorizes actions to be brought on behalf of the federal government by a private party having knowledge of the alleged fraud. Because the complaint is initially filed under seal, the action may be pending for some time before the defendant is even aware of the action. If the government intervenes and is ultimately successful in obtaining redress in the matter or if the plaintiff succeeds in obtaining redress without the government’s involvement, then the plaintiff will receive a percentage of the recovery. Finally, the Social Security Act includes its own provisions that prohibit the filing of false claims or submitting false statements in order to obtain payment. Several states have enacted comparable false claims laws which may be broader in scope and apply regardless of payor.
The civil monetary penalties statute imposes penalties against any person or entity that, among other things, is determined to have presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent. A person who offers or provides to a Medicare or Medicaid beneficiary any remuneration, including waivers of co-payments and deductible amounts (or any part thereof), that the person knows or should know is likely to influence the beneficiary’s selection of a particular provider, practitioner or supplier of Medicare or Medicaid payable items or services may be liable under the civil monetary penalties statute. Moreover, in certain cases, providers who routinely waive copayments and deductibles for Medicare and Medicaid beneficiaries, for example, in connection with patient assistance programs, can also be held liable under the AKS and False Claims Act. One of the statutory exceptions to the prohibition is non-routine, unadvertised waivers of copayments or deductible amounts based on individualized determinations of financial need or exhaustion of reasonable collection efforts. The Office of Inspector General of HHS, or OIG, emphasizes, however, that this exception
118
should only be used occasionally to address special financial needs of a particular patient. Although this prohibition applies only to federal healthcare program beneficiaries, applicable state laws related to, among other things, unlawful schemes to defraud, excessive fees for services, tortious interference with patient contracts and statutory or common law fraud, may also be implicated for similar practices offered to patients covered by commercial payor.
The federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, created new federal criminal statutes that prohibit, among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors, and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. Like the AKS, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
The Physician Payments Sunshine Act, enacted as part of the ACA, as amended among other things, also imposed annual reporting requirements on manufacturers of certain devices, drugs and biologics for certain payments and transfers of value by them and in some cases their distributors to physicians and teaching hospitals, and certain other healthcare professionals as of January 1, 2022; as well as ownership and investment interests held by physicians and their immediate family members. Any failure to comply with these reporting requirements could result in significant fines and penalties. Because we manufacture our own LDTs solely for use by or within our own laboratory, we believe that we are exempt from these reporting requirements. We cannot assure you, however, that the government will agree with our determination, and a determination that we have violated these laws and regulations, or a public announcement that we are being investigated for possible violations, could adversely affect our business, prospects, results of operations or financial condition.
If our operations are found to be in violation of any of the fraud and abuse laws described above or any other laws that apply to us, we may be subject to penalties, including potentially significant criminal, civil and administrative penalties, damages, fines, disgorgement, imprisonment, exclusion from participation in government healthcare programs, contractual damages, reputational harm, integrity oversight and reporting obligations, diminished profits and future earnings, and the curtailment or restructuring of our operations.
International Regulations
Many countries in which we may offer any of our testing products in the future have anti-kickback regulations prohibiting providers from offering, paying, soliciting or receiving remuneration, directly or indirectly, in order to induce business that is reimbursable under any national health care program. In situations involving physicians employed by state-funded institutions or national health care agencies, violation of the local anti-kickback law may also constitute a violation of the U.S. Foreign Corrupt Practices Act, or FCPA.
The FCPA prohibits any U.S. individual, business entity or employee of a U.S. business entity to offer or provide, directly or through a third party, including any potential distributors we may rely on in certain markets, anything of value to a foreign government official with corrupt intent to influence an award or continuation of business or to gain an unfair advantage, whether or not such conduct violates local laws. In addition, it is illegal for a company that reports to the SEC to have false or inaccurate books or records or to fail to maintain a system of internal accounting controls. We will also be required to maintain accurate information and control over sales and distributors’ activities that may fall within the purview of the FCPA, its books and records provisions and its anti-bribery provisions.
The standard of intent and knowledge in the Anti-Bribery cases is minimal-intent and knowledge are usually inferred from that fact that bribery took place. The accounting provisions do not require intent. Violations of the FCPA’s anti-bribery provisions for corporations and other business entities are subject to a fine of up to $2 million and officers, directors, stockholders, employees, and agents are subject to a fine of up to $100,000 and imprisonment for up to five years. Other countries, including the United Kingdom and other OECD Anti-Bribery Convention members, have similar anti-corruption regulations, such as the United Kingdom Anti-Bribery Act.
When marketing our testing products outside of the United States, we may be subject to foreign regulatory requirements governing human clinical testing, prohibitions on the import of tissue necessary for us to perform our testing products or restrictions on the export of tissue imposed by countries outside of the United States or
119
the import of tissue into the United States, and marketing approval. These requirements vary by jurisdiction, differ from those in the United States and may in some cases require us to perform additional pre-clinical or clinical testing. In many countries outside of the United States, coverage, pricing and reimbursement approvals are also required.
Privacy and Security Laws
Health Insurance Portability and Accountability Act
Under HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, HHS has issued regulations to protect the privacy and provide for the security of protected health information, or PHI, used or disclosed by certain entities including health care providers, such as us. HIPAA also regulates standardization of data content, codes and formats used in certain health care transactions and standardization of identifiers for health plans and providers. Penalties for violations of HIPAA and HITECH laws and regulations include civil and criminal penalties.
Three standards have been promulgated under HIPAA’s and HITECH’s regulations: the Standards for Privacy of Individually Identifiable Health Information, which restrict the use and disclosure of certain individually identifiable health information, the Standards for Electronic Transactions, which establish standards for common healthcare transactions, such as claims information, plan eligibility, payment information and the use of electronic signatures, and the Security Standards for the Protection of Electronic Protected Health Information, which require covered entities and business associates to implement and maintain certain security measures to safeguard certain electronic health information, including the adoption of administrative, physical and technical safeguards to protect such information.
The HIPAA privacy regulations cover the use and disclosure of PHI by covered entities as well as business associates, which are defined to include subcontractors that create, receive, maintain, or transmit PHI on behalf of a business associate. They also set forth certain rights that an individual has with respect to his or her PHI maintained by a covered entity, including the right to access or amend certain records containing PHI, or to request restrictions on the use or disclosure of PHI. The HIPAA security regulations establish requirements for safeguarding the confidentiality, integrity, and availability of PHI that is electronically transmitted or electronically stored. HITECH, among other things, established certain health information security breach notification requirements. A covered entity must notify any individual whose PHI is breached according to the specifications set forth in the breach notification rule. The HIPAA privacy and security regulations establish a uniform federal “floor” and do not preempt state laws that are more stringent or provide individuals with greater rights with respect to the privacy or security of, and access to, their records containing PHI or insofar as such state laws apply to personal information that is broader in scope than PHI.
Individuals (or their personal representatives, as applicable) have the right to access test reports directly from laboratories and to direct that copies of those reports be transmitted to persons or entities designated by the individual.
HIPAA authorizes state attorneys general to file suit on behalf of their residents for violations. Courts are able to award damages, costs and attorneys’ fees related to violations of HIPAA in such cases. While HIPAA does not create a private right of action allowing individuals to file suit against us in civil court for violations of HIPAA, its standards have been used as the basis for duty of care cases in state civil suits such as those for negligence or recklessness in the misuse or breach of PHI. In addition, HIPAA mandates that the Secretary of HHS conduct periodic compliance audits of HIPAA covered entities, such as us, and their business associates for compliance with the HIPAA privacy and security standards. It also tasks HHS with establishing a methodology whereby harmed individuals who were the victims of breaches of unsecured PHI may receive a percentage of the civil monetary penalty paid by the violator.
As a covered entity with downstream vendors and subcontractors and, in certain instances, as a business associate of other covered entities with whom we have entered into a business associate agreement, we have certain obligations under HIPAA regarding the use and disclosure of any PHI that may be provided to us. HIPAA and HITECH impose civil and criminal penalties against covered entities and business associates for noncompliance with privacy and security requirements. Further, various states, such as California and Massachusetts, have implemented similar privacy laws and regulations that impose restrictive requirements regulating the use and disclosure of health information and other personally identifiable information.
120
Numerous other federal, state and foreign laws, including consumer protection laws and regulations, govern the collection, dissemination, use, access to, confidentiality and security of patient health information. We intend to continue to comprehensively protect all personal information and to comply with all applicable laws regarding the protection of such information.
Reimbursement for Clinical Laboratory Services
We generate revenue on our products from several sources, including third-party payors, laboratory services intermediaries, and self-paying individuals. Depending on the billing arrangement and applicable law, we must bill various third-party payors, such as insurance companies, Medicare, Medicaid, and patients, all of which have different billing requirements. Compliance with applicable laws and regulations as well as internal compliance policies and procedures adds further complexity to the billing process. CMS establishes new procedures and continuously evaluates and implements changes to the reimbursement process for billing the Medicare program.
To receive reimbursement from third-party payors, we bill our tests using a variety of Current Procedural Terminology, or CPT, codes, as defined by the American Medical Association. For many of the genetic tests we conduct, there may not be an appropriate CPT code for some of the genes in a panel, in which case the test may be billed under a miscellaneous code for an unlisted molecular pathology procedure. Because these miscellaneous codes do not describe a specific service, the third-party payor claim may need to be examined to determine the service that was provided, whether the service was appropriate and medically necessary and whether payment should be rendered. This process can require a letter of medical necessity from the ordering physician and it can result in a delay in processing the claim, a lower reimbursement amount, or denial of the claim.
With the evolution of genetic testing, we have seen individual third-party payors’ medical coverage policies around the CPT codes we bill and their associated payment rates change over time, resulting in changes to our reimbursement revenues. We believe all of our products provide significant clinical value and reduction in downstream healthcare spend, as evidenced in research studies and clinical publications, which we believe will continue to support and drive third-party payor reimbursement.
Under Medicare, payment for our laboratory tests are generally made under the CLFS with payment amounts assigned to specific procedure billing codes. In April 2014, Congress passed PAMA, which included substantial changes to the way in which clinical laboratory services will be paid under Medicare. Under PAMA, laboratories that receive the majority of their Medicare revenue from payments made under the CLFS or the Physician Fee Schedule are required to report to CMS, beginning in 2017 and every three years thereafter (or annually for ADLTs), private payor payment rates and volumes for their tests. Laboratories that fail to report the required payment information may be subject to substantial civil monetary penalties. As required under PAMA, CMS will use the rates and volumes reported by laboratories to develop Medicare payment rates for laboratory tests equal to the volume-weighted median of the private payor payment rates for the tests. On June 23, 2016, CMS published the final rule implementing the reporting and rate-setting requirements under PAMA.
As set forth under PAMA, for tests furnished on or after January 1, 2018, Medicare payments for clinical diagnostic laboratory tests will be paid based upon these reported private payor rates. For clinical diagnostic laboratory tests that are assigned a new or substantially revised CPT code, initial payment rates will be assigned by the gap-fill methodology, as under prior law. Initial payment rates for new ADLTs will be based on the actual list charge for the laboratory test.
The payment rates calculated under PAMA became effective on January 1, 2018. Any reductions to payment rates resulting from the new methodology are limited to 10% per test per year in each of the years 2018 through 2020 and to 15% per test per year in each of the years 2021 through 2023.
On May 17, 2019, CMS determined that DecisionDx-UM meets the criteria for “existing ADLT” status. This means that beginning in 2021, the DecisionDx-UM Medicare rate will be set annually based upon the median private payor rate for the first half of the preceding calendar year. Specifically, the median private payor rate from January 1 to June 30, 2019 will be used to set the Medicare rate for the calendar year 2021. From May 17, 2019 through December 31, 2020, our rate will be set by Noridian, our local MAC. Also, on May 17, 2019, CMS determined that DecisionDx-Melanoma meets the criteria for “new ADLT” status. This means that from July 1, 2019 through March 31, 2020 the Medicare reimbursement rate will equal the initial list price of $7,193.00. The rate for April 1, 2020 through December 31, 2020 will be calculated based upon the median private payor rate from July 1, 2019 to November 30, 2019. Accordingly, beginning in 2021, the rate will be set
121
annually based upon the median private payor rate for the first half of the preceding calendar year. If CMS determines the list charge amount for DecisionDx-Melanoma was greater than 130% of the weighted median of private payor rates, CMS will recoup from us the difference between the actual list charge and 130% of the weighted median.
PAMA also authorizes the adoption of new, temporary billing codes and/or unique test identifiers for FDA-cleared or approved tests as well as ADLTs. The AMA’s CPT Editorial Panel has approved a proposal to create a new section of billing codes to facilitate implementation of this section of PAMA. These proprietary laboratory analyses codes may be requested by a clinical laboratory or manufacturer to specifically identify their test. If approved, the codes are issued by the AMA on a quarterly basis.
Healthcare Reform
In March 2010, the Patient Protection and Affordable Care Act of 2010, as amended by the Health Care and Education Reconciliation Act of 2010, collectively the ACA, was enacted in the U.S. The Affordable Care Act made a number of substantial changes to the way healthcare is financed both by governmental and private insurers. For example, the ACA requires each medical device manufacturer to pay a sales tax equal to 2.3% of the price for which such manufacturer sells its medical devices. The medical device tax has been suspended until December 31, 2019, but is scheduled to return beginning in 2020. It is unclear at this time when, or if, the provision of our LDTs will trigger the medical device tax if the FDA ends its policy of general enforcement discretion and regulates certain LDTs as medical devices, and it is possible that this tax will apply to some or all of our existing testing products or testing products we may develop in the future. The ACA also contains a number of other provisions, including provisions governing enrollment in federal and state healthcare programs, reimbursement matters and fraud and abuse, which we expect will impact our industry and our operations in ways that we cannot currently predict.
On December 14, 2018, a U.S. District Court Judge in the Northern District of Texas, or Texas District Court Judge, ruled that the entire Affordable Care Act is invalid based primarily on the fact that the Tax Cuts and Jobs Act of 2017, or the TCJA, repealed the tax-based shared responsibility payment imposed by the ACA, on certain individuals who fail to maintain qualifying health coverage for all or part of a year, which is commonly referred to as the “individual mandate”. While the Texas District Court Judge, as well as the current presidential administration and CMS, have stated that this ruling will have no immediate effect, it is unclear how this decision, subsequent appeals, and other efforts to repeal and replace the ACA will impact the ACA.
Legal Proceedings
From time to time, we may become involved in litigation or other legal proceedings. We are not currently a party to any litigation or legal proceedings that, in the opinion of our management, are likely to have a material adverse effect on our business. Regardless of outcome, litigation can have an adverse impact on us because of defense and settlement costs, diversion of management resources and other factors.
Facilities
We lease approximately 4,195 square feet of office space in Friendswood, Texas under an agreement that expires June 30, 2019, which we have the option to renew for successive one year periods beginning on July 1 of each subsequent year with the last such one-year extension commencing on July 1, 2020. We also lease approximately 12,000 square feet of laboratory space in Phoenix, Arizona under an agreement that expires July 31, 2023. We believe that our existing facilities will be sufficient for our needs for the foreseeable future.
Employees
As of March 31, 2019, we had 96 employees, of whom 94 were full-time employees. Our employees are not represented by labor unions or covered by collective bargaining agreements. We consider our relationship with our employees to be good.
122
The following table sets forth information about our executive officers, directors and key employees as of June 14, 2019.
|
Name
|
Age
|
Position(s)
|
|
Executive Officers
|
|
|
|
Derek J. Maetzold
|
57
|
President, Chief Executive Officer and Director
|
|
Federico A. Monzon, M.D.
|
51
|
Chief Medical Officer
|
|
Bernhard E. Spiess
|
59
|
Chief Operating Officer
|
|
Frank Stokes
|
49
|
Chief Financial Officer
|
|
|
|
|
|
Non-Employee Directors
|
|
|
|
Daniel M. Bradbury
|
58
|
Chairman of the Board of Directors
|
|
Bonnie H. Anderson(2)(3)
|
61
|
Director
|
|
Mara G. Aspinall(1)(3)
|
56
|
Director
|
|
G. Bradley Cole(1)(2)
|
63
|
Director
|
|
Joseph C. Cook III(1)
|
48
|
Director
|
|
David Kabakoff, Ph.D.(2)(3)
|
71
|
Director
|
|
|
|
|
|
Key Employees
|
|
|
|
Kristen Oelschlager, R.N.
|
51
|
Senior Vice President, Clinical Operations
|
|
Toby Juvenal
|
59
|
Senior Vice President, Sales
|
|
Robert W. Cook, Ph.D.
|
47
|
Vice President, Medical Affairs and Research and Development
|
| (1) | Member of the audit committee. |
| (2) | Member of the compensation committee. |
| (3) | Member of the nominating and corporate governance committee. |
Executive Officers
Derek J. Maetzold founded Castle Biosciences in September 2007 and has served as our President and Chief Executive Officer and as a member of our board of directors since inception. Previously, Mr. Maetzold held leadership roles at Encysive Pharmaceuticals, Schering-Plough Corporation (now Merck), Integrated Communications, Amylin Pharmaceuticals and Sandoz Pharmaceuticals (now a division of Novartis). Mr. Maetzold currently serves as a director of the Ocular Melanoma Foundation. He has contributed to the discovery, development and commercialization of five diagnostic and prognostic tests in cancers, has co-authored multiple scientific publications and is a co-inventor of a number of technologies at Castle Biosciences and Encysive Pharmaceuticals. Mr. Maetzold holds a B.S. degree in Biology from George Mason University and completed additional coursework at the University of Calgary Health Sciences Center and the MBA program at the University of California, Riverside.
Our board of directors believes that Mr. Maetzold’s experience as our founder, director and as our President and Chief Executive Officer, as well as his expertise in the development and commercialization of diagnostic tests, qualify him to serve on our board of directors.
Federico A. Monzon, M.D. has served as our Chief Medical Officer since November 2015. From September 2013 to October 2015, Dr. Monzon served as Medical Director of Oncology and Medical Director for Latin America at Invitae Corporation, a provider of genetic diagnostics for hereditary disorders. Since October 2011, Dr. Monzon held the position of Director of Molecular Pathology at the Cancer Genetics Laboratory from Baylor College of Medicine, where he also had an academic appointment as Associate Professor in the Department of Pathology & Immunology. Since September 2013, Dr. Monzon has held an academic appointment as Clinical Associate Professor in the Department of Pathology and Immunology. Dr. Monzon holds an M.D. degree from the Universidad Nacional Autónoma de México, and is board certified in Anatomic and Clinical Pathology and Molecular Genetic Pathology by the American Board of Pathology. He completed his Pathology residency at Thomas Jefferson University and his Molecular Pathology fellowship at the University of Pittsburgh.
123
Bernhard E. Spiess has served as our Chief Operating Officer since May 2016. From April 1997 to April 2016, Mr. Spiess held various positions with Beckman Coulter, Inc., a manufacturer of biomedical testing instrument systems, tests and supplies, including as Vice President, Strategic Marketing, Blood Virus & Infectious Diseases from February 2015 to April 2016, and as Vice President, Marketing, Molecular Diagnostics from April 2008 to February 2015. Throughout his career, Mr. Spiess has held positions of increasing responsibility in laboratory testing and management, scientific support, sales, strategic and tactical marketing, and product and business management. Mr. Spiess holds a B.App.Sc. degree in Medical Technology from the Royal Melbourne Institute of Technology.
Frank Stokes has served as our Chief Financial Officer since December 2017. From January 2017 to December 2017, Mr. Stokes served as Chief Financial Officer of Hammock Pharmaceuticals, a specialty pharmaceutical company focused on the development and commercialization of women’s health and urology products. From May 2011 to December 2016, Mr. Stokes served as a Managing Director of Leerink Swann (now SVB Leerink). Mr. Stokes also held positions as a Managing Director at Robert W. Baird & Co. Incorporated and Wachovia Securities, LLC. While at SVB Leerink and Robert W. Baird & Co., Mr. Stokes led life sciences, tools and diagnostics sector investment banking efforts, and managed financings and mergers and acquisitions transactions. Mr. Stokes holds a B.S. degree in Business Administration and J.D. and MBA degrees from the University of North Carolina at Chapel Hill.
Non-Employee Directors
Daniel M. Bradbury has served as a member our board of directors since September 2012, and as chairman of our board of directors since September 2014. Since June 2018, Mr. Bradbury has served as Chief Executive Officer of Equillium, Inc., a publicly traded biotechnology company, where he also served as its President from March 2017 until June 2018. Mr. Bradbury is the founder of, and has served as the managing member of, BioBrit, LLC, a life sciences consulting and investment firm, since September 2012. Mr. Bradbury served as President, Chief Executive Officer and a director of Amylin Pharmaceuticals, Inc., from March 2007 until Amylin’s acquisition by Bristol-Myers Squibb Company in August 2012. Prior to Amylin, Mr. Bradbury worked in marketing and sales for ten years at SmithKline Beecham Pharmaceuticals. Mr. Bradbury serves on the board of directors of numerous private companies and two publicly traded companies, Equillium, Inc. and Intercept Pharmaceuticals, Inc. Mr. Bradbury previously served on the board of directors of the following publicly traded companies: Corcept Therapeutics, Inc., from 2012 to 2019; Geron Corporation, from 2012 to 2019; and Illumina, Inc. from 2004 to 2017. Mr. Bradbury holds a Bachelor of Pharmacy from Nottingham University and a Diploma in Management Studies from Harrow and Ealing Colleges of Higher Education in the United Kingdom.
Our board of directors believes that Mr. Bradbury’s extensive experience as a life sciences executive and his other executive and board of directors experience qualify him to serve on our board of directors.
Bonnie H. Anderson has served as a member of our board of directors since June 2015. Since February 2008, Ms. Anderson has served as the Chief Executive Officer and as a member of the board of directors of Veracyte, Inc., a publicly traded genomic diagnostics company, and as the chairman of Veracyte’s board of directors, since December 2016. From August 2013 to February 2017, she also served as Veracyte’s President. Prior to Veracyte, Ms. Anderson was an independent strategic consultant from April 2006 to January 2008. Ms. Anderson was a Vice President at Beckman Coulter, Inc., a manufacturer of biomedical testing instrument systems, tests and supplies, from September 2000 to March 2006. In addition to her positions with Veracyte, Ms. Anderson currently serves as a trustee emeritus of the Keck Graduate Institute of Applied Life Sciences. Ms. Anderson holds a B.S. degree in Medical Technology from Indiana University of Pennsylvania.
Our board of directors believes that Ms. Anderson’s career in the diagnostics and testing industry qualify her to serve on our board of directors
Mara G. Aspinall has served as a member of our board of directors since February 2015. Ms. Aspinall has served as Managing Director of BlueStone Venture Partners, a life science-focused venture capital firm, since December 2017. Since June 2014, Ms. Aspinall has served as the President and Chief Executive Officer of Health Catalysts Group, a consulting firm that focuses on growth of early-stage life science and technology companies. From September 2011 until June 2014, Ms. Aspinall served as the President and Chief Executive Officer of the Ventana Medical Systems and the Global Head of Roche Tissue Diagnostics, a global leader in the development and commercialization of tissue-based cancer diagnostics. Prior to 2011, Ms. Aspinall served as President of Genzyme Pharmaceuticals and Genzyme Genetics. Ms. Aspinall currently serves as a director of
124
Allscripts Healthcare Solutions, Orasure Technologies and UK-based company Abcam plc., along with other privately-held healthcare technology and medical insurance companies. Ms. Aspinall holds a B.A. degree in International Relations from Tufts University and an MBA from Harvard Business School.
Our board of directors believes that Ms. Aspinall’s operational expertise, international experience and digital health knowledge qualify her to serve on our board of directors.
G. Bradley Cole has served as a member of our board of directors since December 2018. Since June 2014, Mr. Cole has served as Chief Financial Officer of Genomic Health, Inc., a publicly traded global provider of genomic-based diagnostic tests, where he also served as Chief Operating Officer from January 2009 until March 2018, and as Executive Vice President, Operations from January 2008 until January 2009. Mr. Cole previously held Chief Financial Officer positions at multiple publicly traded companies, including Applied Biosciences, Inc. Mr. Cole holds a B.S. degree in Accounting from Biola University and an MBA from San Jose State University.
Our board of directors believes that Mr. Cole’s extensive experience in finance and operations as a public company executive qualify him to serve on our board of directors.
Joseph C. Cook III has served as a member of our board of directors since August 2018. Since February 2003, Mr. Cook has served as a Managing Director at Mountain Group Partners, an investment firm with investments in the Life Sciences, AgTech and Technology sectors. From January 2001 until February 2003, Mr. Cook served as a Director, Private Placements in the Investment Banking Group of Robert W. Baird & Co. Incorporated. Mr. Cook also previously served as a Vice President in the Investment Banking Group at J.C. Bradford & Co. Mr. Cook serves on the board of directors of MiNDERA Corp., a non-invasive transcriptome testing company, and Cerebrotech Medical Systems, Inc., a machine learning cerebral diagnostic company. Mr. Cook holds a B.A. degree in Economics from Davidson College.
Our board of directors believes Mr. Cook’s investing expertise and experience in the life sciences and investment banking industries qualify him to serve on our board of directors.
David Kabakoff, Ph.D. has served as a member of our board of directors since September 2017. Since 2012, Dr. Kabakoff has served as a partner at HealthQuest Capital. Since 2007, Dr. Kabakoff has also served as an executive partner at Sofinnova Ventures. Dr. Kabakoff’s currently serves as chairman of the board of directors of NextCure, Inc., a publicly traded biopharmaceutical company. Dr. Kabakoff also serves on the board of directors of numerous private companies, including Antiva Biosciences, Inc., a biopharmaceutical company, Dauntless Pharmaceuticals, a biopharmaceutical company, Lineagen (chairman), a genetic testing company, bioTheranostics, a molecular diagnostics company, Rainier Therapeutics, Inc., a biopharmaceutical company and Neurana Pharmaceuticals, Inc., a biopharmaceutical company. From 2006 until October 2014, Dr. Kabakoff served as a director of InterMune Inc., a publicly traded biopharmaceutial company. In 2001 Dr. Kabakoff co-founded Salmedix, Inc., a developer of cancer drug treatments, served as its Chairman and Chief Executive Officer and led its acquisition in June 2005 by Cephalon, Inc., a biopharmaceutical company. From 1996 to 2000, Dr. Kabakoff held the positions of Executive Vice President of Dura Pharmaceuticals, Inc. and President and Chief Executive Officer of Spiros, both pharmaceutical companies. From 1989 to 1996, Dr. Kabakoff was employed as Chief Executive Officer of Corvas International, Inc., a developer of biotherapeutics, and from 1983 to 1989 held senior executive positions with Hybritech, a biotechnology company. Dr. Kabakoff holds a B.A. degree in Chemistry from Case Western Reserve University and a Ph.D. from Yale University.
Our board of directors believes that Dr. Kabakoff’s educational background, as well as financial understanding of the diagnostics industry gained from his investing experience, qualify him to serve on our board of directors.
Key Employees
Kristen Oelschlager, R.N. has served as our Senior Vice President of Clinical Operations since October 2008. From May 1996 to September 2008, Ms. Oelschlager served as Director of Clinical Research of Arizona Pulmonary Specialists, where she managed a multi-specialty clinical research department. Ms. Oelschlager has co-authored multiple scientific publications and is a co-inventor of a number of our technologies. Ms. Oelschlager completed her core nursing requirements at Purdue University and holds an A.S. degree in Nursing from Indiana Vocational Technical College.
125
Toby Juvenal has served as our Senior Vice President of Sales since October 2008. From December 2005 to June 2007, Mr. Juvenal served as Vice President of Sales of Encysive Pharmaceuticals. Mr. Juvenal has also held management positions at deCODE Genetics, Genzyme Pharmaceuticals and Genetics Institute. Mr. Juvenal holds a B.S. degree in Marketing from the University of Florida.
Robert W. Cook, Ph.D. has served as our Vice President of Medical Affairs and Research and Development since April 2018. Previously, he served as our Manager of Scientific Relations from February 2011 to June 2015 and our Executive Director of Research and Development from June 2015 to April 2018. Dr. Cook has held postdoctoral fellowships at Baylor College of Medicine and the University of California, San Diego. Dr. Cook holds a B.S. degree in Molecular Biology from Temple University and a Ph.D. from Northwestern University in Biochemistry, Molecular Biology and Cell Biology.
Board Composition
Our business and affairs are organized under the direction of our board of directors, which currently consists of seven members. The primary responsibilities of our board of directors are to provide oversight, strategic guidance, counseling and direction to our management. Our board of directors meets on a regular basis and on an ad hoc basis as required.
Our board of directors has determined that all of our directors other than Mr. Maetzold are independent directors, as defined by Rule 5605(a)(2) of The Nasdaq Listing Rules.
In accordance with the terms of our amended and restated certificate of incorporation and amended and restated bylaws, which will become effective immediately prior to and upon the completion of this offering, respectively, we will divide our board of directors into three classes, as follows:
| • | Class I, which will consist of Joseph C. Cook III and David Kabakoff, Ph.D., whose terms will expire at our annual meeting of stockholders to be held in 2020; |
| • | Class II, which will consist of Mara G. Aspinall and Daniel M. Bradbury, whose terms will expire at our annual meeting of stockholders to be held in 2021; and |
| • | Class III, which will consist of Bonnie H. Anderson, G. Bradley Cole and Derek J. Maetzold, whose terms will expire at our annual meeting of stockholders to be held in 2022. |
At each annual meeting of stockholders to be held after the initial classification, the successors to directors whose terms then expire will serve until the third annual meeting following their election and until their successors are duly elected and qualified. The authorized size of our board of directors is currently seven members. The authorized number of directors may be changed only by resolution of our board of directors. Any additional directorships resulting from an increase in the number of directors will be distributed between the three classes so that, as nearly as possible, each class will consist of one-third of the directors. This classification of our board of directors may have the effect of delaying or preventing changes in our control or management. Our directors may be removed for cause by the affirmative vote of the holders of at least % of our voting stock.
Board Leadership Structure
Our board of directors is currently chaired by Mr. Bradbury who has authority, among other things, to call and preside over board of directors meetings, to set meeting agendas and to determine materials to be distributed to the board of directors. Accordingly, the Chairman has substantial ability to shape the work of the board of directors. We believe that separation of the positions of Chairman and Chief Executive Officer reinforces the independence of the board of directors in its oversight of our business and affairs. In addition, we have a separate chair for each committee of our board of directors. The chair of each committee is expected to report annually to our board of directors on the activities of their committee in fulfilling their responsibilities as detailed in their respective charters or specify any shortcomings should that be the case.
Role of the Board in Risk Oversight
The audit committee of our board of directors is primarily responsible for overseeing our risk management processes on behalf of our board of directors. Going forward, we expect that the audit committee will receive reports from management periodically regarding our assessment of risks. In addition, the audit committee reports regularly to our board of directors, which also considers our risk profile. The audit committee and our board of
126
directors focus on the most significant risks we face and our general risk management strategies. While our board of directors oversees our risk management, management is responsible for day-to-day risk management processes. Our board of directors expects management to consider risk and risk management in each business decision, to proactively develop and monitor risk management strategies and processes for day-to-day activities and to effectively implement risk management strategies adopted by the audit committee and our board of directors. We believe this division of responsibilities is the most effective approach for addressing the risks we face and that our board of directors’ leadership structure, which also emphasizes the independence of our board of directors in its oversight of its business and affairs, supports this approach.
Board Committees
Our board of directors has established an audit committee, a compensation committee and a nominating and corporate governance committee.
Audit Committee
Our audit committee consists of G. Bradley Cole, Mara G. Aspinall and Joseph C. Cook III. Our board of directors has determined that each of the members of our audit committee satisfies the Nasdaq and SEC independence requirements. G. Bradley Cole serves as the chair of our audit committee. The functions of this committee include, among other things:
| • | evaluating the performance, independence and qualifications of our independent auditors and determining whether to retain our existing independent auditors or engage new independent auditors; |
| • | reviewing and approving the engagement of our independent auditors to perform audit services and any permissible non-audit services; |
| • | monitoring the rotation of partners of our independent auditors on our engagement team as required by law; |
| • | prior to engagement of any independent auditor, and at least annually thereafter, reviewing relationships that may reasonably be thought to bear on their independence, and assessing and otherwise taking the appropriate action to oversee the independence of our independent auditor; |
| • | reviewing our annual and quarterly financial statements and reports, including the disclosures contained under the caption “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” and discussing the statements and reports with our independent auditors and management; |
| • | reviewing, with our independent auditors and management, significant issues that arise regarding accounting principles and financial statement presentation and matters concerning the scope, adequacy and effectiveness of our financial controls; |
| • | reviewing with management and our independent auditors any earnings announcements and other public announcements regarding material developments; |
| • | establishing procedures for the receipt, retention and treatment of complaints received by us regarding financial controls, accounting or auditing matters and other matters; |
| • | preparing the report that the SEC requires in our annual proxy statement; |
| • | reviewing and providing oversight of any related-person transactions in accordance with our related person transaction policy and reviewing and monitoring compliance with legal and regulatory responsibilities, including our code of business conduct and ethics; |
| • | reviewing our major financial risk exposures, including the guidelines and policies to govern the process by which risk assessment and risk management are implemented; |
| • | reviewing on a periodic basis our investment policy; and |
| • | reviewing and evaluating on an annual basis the performance of the audit committee and the audit committee charter. |
127
Our board of directors has determined that G. Bradley Cole qualifies as an audit committee financial expert within the meaning of SEC regulations and meets the financial sophistication requirements of the Nasdaq Listing Rules. In making this determination, our board has considered prior experience, business acumen and independence. Both our independent registered public accounting firm and management periodically meet privately with our audit committee.
We believe that the composition and functioning of our audit committee complies with all applicable requirements of the Sarbanes-Oxley Act, and all applicable SEC and Nasdaq rules and regulations. We intend to comply with future requirements to the extent they become applicable to us.
Compensation Committee
Our compensation committee consists of David Kabakoff, Ph.D., Bonnie H. Anderson and G. Bradley Cole. David Kabakoff, Ph.D. serves as the chair of our compensation committee. Our board of directors has determined that each of the members of our compensation committee is a non-employee director, as defined in Rule 16b-3 promulgated under the Exchange Act, and satisfies the Nasdaq independence requirements. The functions of this committee include, among other things:
| • | reviewing, modifying and approving (or if it deems appropriate, making recommendations to the full board of directors regarding) our overall compensation strategy and policies; |
| • | reviewing and approving or, in the case of our chief executive officer's compensation, making recommendations to the full board of directors regarding the compensation and other terms of employment of our executive officers; |
| • | reviewing and approving (or if it deems it appropriate, making recommendations to the full board of directors regarding) performance goals and objectives relevant to the compensation of our executive officers and assessing their performance against these goals and objectives; |
| • | reviewing and approving (or if it deems it appropriate, making recommendations to the full board of directors regarding) the equity incentive plans, compensation plans and similar programs advisable for us, as well as modifying, amending or terminating existing plans and programs; |
| • | evaluating risks associated with our compensation policies and practices and assessing whether risks arising from our compensation policies and practices for our employees are reasonably likely to have a material adverse effect on us; |
| • | reviewing and making recommendations to the full board of directors regarding the type and amount of compensation to be paid or awarded to our non-employee board members; |
| • | establishing policies with respect to votes by our stockholders to approve executive compensation as required by Section 14A of the Exchange Act and determining our recommendations regarding the frequency of advisory votes on executive compensation, to the extent required by law; |
| • | reviewing and assessing the independence of compensation consultants, legal counsel and other advisors as required by Section 10C of the Exchange Act; |
| • | administering our equity incentive plans; |
| • | establishing policies with respect to equity compensation arrangements; |
| • | reviewing the competitiveness of our executive compensation programs and evaluating the effectiveness of our compensation policy and strategy in achieving expected benefits to us; |
| • | reviewing and making recommendations to the full board of directors regarding the terms of any employment agreements, severance arrangements, change in control protections and any other compensatory arrangements for our executive officers; |
| • | reviewing with management and approving our disclosures under the caption “Compensation Discussion and Analysis” in our periodic reports or proxy statements to be filed with the SEC, to the extent such caption is included in any such report or proxy statement; |
| • | preparing the report that the SEC requires in our annual proxy statement; and |
128
| • | reviewing and assessing on an annual basis the performance of the compensation committee and the compensation committee charter. |
We believe that the composition and functioning of our compensation committee complies with all applicable requirements of the Sarbanes-Oxley Act, and all applicable SEC and Nasdaq rules and regulations. We intend to comply with future requirements to the extent they become applicable to us.
Nominating and Corporate Governance Committee
Our nominating and corporate governance committee consists of Bonnie H. Anderson, Mara G. Aspinall and David Kabakoff, Ph.D. Our board of directors has determined that each of the members of this committee satisfies the Nasdaq independence requirements. Bonnie H. Anderson serves as the chair of our nominating and corporate governance committee. The functions of this committee include, among other things:
| • | identifying, reviewing and evaluating candidates to serve on our board of directors consistent with criteria approved by our board of directors; |
| • | determining the minimum qualifications for service on our board of directors; |
| • | evaluating director performance on the board and applicable committees of the board and determining whether continued service on our board is appropriate; |
| • | evaluating, nominating and recommending individuals for membership on our board of directors; |
| • | evaluating nominations by stockholders of candidates for election to our board of directors; |
| • | considering and assessing the independence of members of our board of directors; |
| • | developing a set of corporate governance policies and principles, including a code of business conduct and ethics, periodically reviewing and assessing these policies and principles and their application and recommending to our board of directors any changes to such policies and principles; |
| • | considering questions of possible conflicts of interest of directors as such questions arise; and |
| • | reviewing and assessing on an annual basis the performance of the nominating and corporate governance committee and the nominating and corporate governance committee charter. |
We believe that the composition and functioning of our nominating and corporate governance committee complies with all applicable requirements of the Sarbanes-Oxley Act, and all applicable SEC and Nasdaq rules and regulations. We intend to comply with future requirements to the extent they become applicable to us.
Compensation Committee Interlocks and Insider Participation
None of our current or former executive officers serve as a member of the compensation committee. None of our officers serve, or have served during the last completed fiscal year, on the board of directors or compensation committee, or other committee serving an equivalent function, of any other entity that has one or more of its executive officers serving as a member of our board of directors or our compensation committee. For a description of transactions between us and members of our compensation committee and affiliates of such members, please see “Certain Relationships and Related Party Transactions.”
Code of Business Conduct and Ethics
We have adopted a written code of business conduct and ethics that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or person performing similar functions. Following this offering, a current copy of the code will be available on the Corporate Governance section of our website, www.castlebiosciences.com.
Limitation of Liability and Indemnification
Our amended and restated certificate of incorporation, which will become effective immediately prior to the completion of this offering, limits the liability of directors to the maximum extent permitted by Delaware law. Delaware law allows a corporation to eliminate the personal liability of directors of a corporation to the corporation and its stockholders for monetary damages for breach of their fiduciary duties as directors, except for liability for any:
129
| • | breach of his or her duty of loyalty to the corporation or its stockholders; |
| • | act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; |
| • | unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the Delaware General Corporation Law; or |
| • | transaction from which the director derived an improper personal benefit. |
Our amended and restated certificate of incorporation, which will become effective immediately prior to the completion of this offering, does not eliminate a director’s duty of care and, in appropriate circumstances, equitable remedies, such as injunctive or other forms of non-monetary relief, will remain available under Delaware law. These limitations also do not affect a director’s responsibilities under any other laws, such as the federal securities laws or other state or federal laws. Our amended and restated bylaws, which will become effective upon the completion of this offering, provide that we will indemnify our directors and executive officers and may indemnify other officers, employees and other agents, to the fullest extent permitted by law. Our amended and restated bylaws, which will become effective upon the completion of this offering, also provide that we are obligated to advance expenses incurred by a director or officer in advance of the final disposition of any action or proceeding and also permit us to secure insurance on behalf of any officer, director, employee or other agent for any liability arising out of his or her actions in connection with their services to us, regardless of whether our amended and restated bylaws permit such indemnification. We have obtained a policy of directors’ and officers’ liability insurance.
We have entered, and intend to continue to enter, into separate indemnification agreements with our directors and executive officers, in addition to the indemnification provided for in our amended and restated bylaws. These agreements, among other things, will require us to indemnify our directors and executive officers for certain expenses, including attorneys’ fees, judgments, fines and settlement amounts incurred by a director or executive officer in any action or proceeding arising out of their services as one of our directors or executive officers or any other company or enterprise to which the person provides services at our request. We believe that these bylaw provisions and indemnification agreements are necessary to attract and retain qualified persons as directors and officers.
The limitation of liability and indemnification provisions in our amended and restated certificate of incorporation and amended and restated bylaws may discourage stockholders from bringing a lawsuit against directors for breach of their fiduciary duties. They may also reduce the likelihood of derivative litigation against directors and officers, even though an action, if successful, might benefit us and our stockholders. A stockholder’s investment may be harmed to the extent we pay the costs of settlement and damage awards against directors and officers pursuant to these indemnification provisions.
We believe that these provisions in our amended and restated certificate of incorporation and amended and restated bylaws and these indemnification agreements are necessary to attract and retain qualified persons as directors and officers.
Except as otherwise disclosed under the heading “Legal Proceedings” in the “Business” section of this prospectus, at present, there is no pending litigation or proceeding involving any of our directors or executive officers as to which indemnification is required or permitted, and we are not aware of any threatened litigation or proceeding that may result in a claim for indemnification.
130
EXECUTIVE AND DIRECTOR COMPENSATION
Our named executive officers for the year ended December 31, 2018, which include our principal executive officer and our two other most highly compensated executive officers, are:
| • | Derek J. Maetzold, our President and Chief Executive Officer |
| • | Frank Stokes, our Chief Financial Officer |
| • | Federico A. Monzon, M.D., our Chief Medical Officer |
Summary Compensation Table
Name and principal position |
Year |
Salary ($) |
Option awards ($)(1) |
Non-equity Incentive Plan Compensation ($)(2) |
All other compensation ($)(3) |
Total ($) |
||||||||||||
Derek J. Maetzold President and Chief Executive Officer |
2018 | 409,500 | 161,040 | 206,000 | 14,218 | 790,758 | ||||||||||||
Frank Stokes Chief Financial Officer |
2018 | 275,000 | 177,347 | 96,250 | — | 548,597 | ||||||||||||
Federico A. Monzon, M.D. Chief Medical Officer |
2018 | 317,075 | 33,387 | 112,400 | 11,000 | 473,862 | ||||||||||||
| (1) | This column reflects the aggregate grant date fair value of options without regard to forfeitures granted during the year measured pursuant to Financial Accounting Standards Board Accounting Standards Codification Topic 718 (ASC 718). The valuation assumptions we used in calculating the fair value of options are set forth in Note 13 to our financial statements appearing at the end of this prospectus. The amounts do not reflect the actual economic value that may be realized by the named executive officer. |
| (2) | Amounts reported represent performance-based cash bonuses earned in 2018 and paid in 2019, as further described below under “—Performance Bonus Compensation.” |
| (3) | Amounts reflect the following: for Mr. Maetzold, $14,218 in 401(k) matching contributions; and for Dr. Monzon, $11,000 in 401(k) matching contributions. |
Narrative Disclosure to Summary Compensation Table
Annual Base Salary
The compensation of our named executive officers is generally determined and approved by our board of directors, based on the recommendation of the compensation committee of our board of directors. The 2018 base salaries were $412,000, $275,000 and $319,000 for Mr. Maetzold, Mr. Stokes and Dr. Monzon, respectively.
Performance Bonus Compensation
In addition to base salaries, our named executive officers are eligible to receive annual performance-based cash bonuses, which are designed to provide appropriate incentives to achieve defined annual performance goals and to reward our executives for individual achievement towards these goals. The annual performance-based bonus each named executive officer is eligible to receive is generally based on the extent to which we achieve the corporate goals that our board of directors establishes each year and the individual’s contributions to such achievements. At the end of the year, our board of directors reviews each executive’s performance and determines the actual bonus payout to be awarded to each of our named executive officers.
For 2018, the target bonus for Mr. Maetzold was 50% of base salary. For 2018, the target bonus for each of Mr. Stokes and Dr. Monzon was 35% of base salary. Our corporate performance objectives for 2018, as established by our board of directors, included achievement of revenue, reimbursement and pipeline program goals. In March 2019, our board of directors approved a 95% overall achievement level of our corporate goals and awarded bonuses to our named executive officers based on Company achievements.
Equity-Based Incentive Awards
We believe that our ability to grant equity-based awards is a valuable and necessary compensation tool that aligns the long-term financial interests of our employees, consultants and directors with the financial interests of our stockholders. In addition, we believe that our ability to grant equity-based awards helps us to attract, retain
131
and motivate employees, consultants and directors, and encourages them to devote their best efforts to our business and financial success. Our board of directors is responsible for approving equity grants. Vesting of equity awards is generally tied to continuous service with us and serves as an additional retention measure. Our executives generally are awarded an initial new hire grant upon commencement of employment. Additional grants may occur periodically in order to specifically incentivize executives with respect to achieving certain corporate goals or to reward executives for exceptional performance.
Prior to this offering, we have granted all equity awards pursuant to the 2008 Plan and 2018 Plan, the terms of which are described below under “—Equity Benefit Plans.” All options are granted with a per share exercise price equal to no less than the fair market value of a share of our common stock on the date of the grant of such award. Generally, our stock option awards vest over a four-year period subject to the holder’s continuous service to us.
In May 2018, under the 2008 Plan, our board of directors granted Mr. Maetzold an option to purchase 120,000 shares of common stock, Mr. Stokes an option to purchase 162,295 shares of common stock and Dr. Monzon an option to purchase 15,000 shares of common stock, each with an exercise price per share of $1.96. In November 2018, under the 2018 Plan, our board of directors granted Mr. Maetzold an option to purchase 25,000 shares of common stock and Dr. Monzon an option to purchase 15,000 shares of common stock, each with an exercise price per share of $1.96. Each of the 2018 options vests as follows: 25% of the shares subject to the option vests on the first anniversary of the vesting commencement date, and the balance of the shares vest in 36 equal monthly installments thereafter, subject to the executive’s continuous service with us. In addition, Mr. Maetzold’s May 2018 option includes an early exercisability feature. For additional information, please see below under “—Outstanding Equity Awards at Fiscal Year-End.”
Agreements with Our Named Executive Officers
Below are descriptions of our employment agreements with our named executive officers. Each of our executive officers’ employment is at will and may be terminated by us at any time. Any potential payments and benefits due upon a qualifying termination of employment or a change in control are further described below under “— Potential Payments and Benefits upon Termination or Change in Control.”
Mr. Maetzold. We entered into an amended and restated employment agreement with Mr. Maetzold in September 2012, which was amended in February 2017 and in June 2019 and which governs the current terms of his employment with us. Under the terms of the employment agreement, as amended, Mr. Maetzold is entitled to an annual base salary of $445,000. In addition, Mr. Maetzold is eligible to receive an annual performance cash bonus with a target of 60% of his base salary based on corporate performance and his individual performance, as determined by our board of directors. Pursuant to his employment agreement, as amended, Mr. Maetzold is entitled to receive periodic grants of stock options, as determined by our board of directors.
Mr. Stokes. We entered into an employment agreement with Mr. Stokes in November 2017, which governs the current terms of his employment with us. Under the terms of the employment agreement, Mr. Stokes was entitled to an initial base salary of $275,000 (most recently increased to $316,250). In addition, he is eligible to receive an annual performance cash bonus with a target payout of 35% of his base salary based on corporate performance, as determined by our board of directors, and Mr. Stokes’s individual performance as determined by our chief executive officer. Pursuant to his employment agreement, in May 2018, we granted to Mr. Stokes an option to purchase 162,295 shares of common stock, as further described below under “—Outstanding Equity Awards at Fiscal Year-End.”
Dr. Monzon. We entered into an employment agreement with Dr. Monzon in October 2015, which governs the current terms of his employment with us. Under the terms of the employment agreement, Dr. Monzon was entitled to an initial annual base salary of $300,000 (most recently increased to $328,776) and a one-time signing bonus of $30,000. In addition, he is eligible to receive an annual performance cash bonus with a target payout of 35% of his base salary based on corporate performance, as determined by our board of directors, and Dr. Monzon’s individual performance as determined by our chief executive officer. Pursuant to his employment agreement, in January 2016, we granted to Dr. Monzon an option to purchase 100,000 shares of common stock, as further described below under “—Outstanding Equity Awards at Fiscal Year-End.”
132
Potential Payments and Benefits upon Termination or Change in Control
Regardless of the manner in which a named executive officer’s service terminates, each named executive officer is entitled to receive amounts earned during his term of service, including unpaid salary and unused vacation, as applicable. In addition, each of our named executive officers is eligible to receive certain benefits in connection with certain terminations pursuant to his employment agreement with us, as described below. For the definitions of “cause,” “good reason,” “disability,” and “change of control” referenced below, please refer to the individual employment agreements with each of our named executive officers.
Mr. Maetzold. Upon Mr. Maetzold’s resignation without good reason, Mr. Maetzold will be entitled to receive 12 months of continued base salary, to be paid in accordance with our normal payroll procedures and less any applicable withholdings. In the event Mr. Maetzold’s service is terminated without cause (excluding death or disability) or he resigns with good reason at any time prior to our undergoing a change of control, Mr. Maetzold will be eligible to receive (i) aggregate severance pay equal to 150% of his base salary, 50% of which is payable in a lump sum after the date of termination, less any applicable withholdings, and the remaining 100% of which is payable in equal regular installments over 12 months in accordance with our normal payroll procedures and less any applicable withholdings, (ii) 18 months’ acceleration on the vesting of any unvested portion of outstanding options, (iii) COBRA continuation at our expense for up to 18 months, and (iv) a cash bonus equal to 150% of the greater of the most recent annual performance bonus target or actual bonus earned, to be paid in equal regular installments over 12 months in accordance with our normal payroll procedures and less any applicable withholdings. In the event Mr. Maetzold’s service is terminated without cause (excluding death or disability) or he resigns with good reason at any time following a change of control, Mr. Maetzold will be eligible to receive (i) aggregate severance pay equal to 300% of his base salary, 200% of which is payable in a lump sum after the date of termination, less any applicable withholdings, and the remaining 100% of which is payable in equal regular installments over 12 months in accordance with our normal payroll procedures and less any applicable withholdings, (ii) immediate vesting of any unvested portion of options outstanding as of the date of termination, (iii) COBRA continuation at our expense for up to three years, and (iv) a cash bonus equal to 300% of the greater of the most recent annual performance bonus target or actual bonus earned, to be paid in a lump sum after the date of termination. In the event of termination due to death or disability, Mr. Maetzold will be eligible to receive immediate vesting of any unvested portion of options outstanding as of the date of termination.
Mr. Stokes and Dr. Monzon. Upon the executive’s termination without cause or resignation for good reason, the executive will be eligible to receive (i) continued base salary for six months for Mr. Stokes or four months for Dr. Monzon (net of any amounts earned through consulting arrangements with us following the executive’s termination), to be paid in accordance with our normal payroll procedures and less any applicable withholdings, (ii) continued COBRA benefits during such period, and (iii) 24 months’ accelerated vesting of the option granted pursuant to the executive’s employment agreement in the event of such termination prior to our undergoing a change of control. In addition, in the event the executive’s service is terminated without cause or the executive resigns for good reason within six months following a change of control, the executive will be eligible to receive (i) immediate vesting of any unvested portion of the option granted pursuant to executive’s employment agreement, (ii) 12 months of continued base salary (net of any amounts earned through consulting arrangements with us following the executive’s termination), to be paid in accordance with our normal payroll procedures and less any applicable withholdings, and (iii) continued COBRA benefits during such period.
All severance benefits described above, other than the continued COBRA benefits for Mr. Stokes and Dr. Monzon, are subject to (i) the execution and effectiveness of a release of claims in favor of us and (ii) compliance with the executive’s employment agreement and employee proprietary information agreement.
Each of our named executive officers holds stock options under the 2008 Plan (and for Mr. Maetzold and Dr. Monzon, also under the 2018 Plan) that were granted subject to the general terms of the applicable equity plan and the form of stock option agreement. A description of the termination and change in control provisions in the 2008 Plan, 2018 Plan and stock options granted thereunder is provided below under “—Equity Benefit Plans” and the specific vesting terms of each named executive officer’s stock options are described below under “—Outstanding Equity Awards at Fiscal Year-End.”
133
Outstanding Equity Awards at Fiscal Year-End
The following table presents information regarding outstanding equity awards held by our named executive officers as of December 31, 2018.
Option Awards(1) |
|||||||||||||||
Name and principal position |
Vesting Commencement Date |
Number of Securities Underlying Unexercised Options (#) Exercisable |
Number of Securities Underlying Unexercised Options (#) Unexercisable(2) |
Option Exercise Price ($)(3) |
Option Expiration Date |
||||||||||
Derek J. Maetzold |
6/1/2015 | 70,000 | 10,000 | 1.57 | (4) |
6/3/2020 | |||||||||
7/21/2017 | 7,083 | 12,917 | 1.93 | 7/20/2022 | |||||||||||
5/10/2018 | 120,000 | (5) |
— | 1.96 | 5/9/2028 | ||||||||||
11/12/2018 | — | 25,000 | 1.96 | 11/11/2028 | |||||||||||
Frank Stokes |
12/4/2017 | 162,295 | (5) |
— | 1.96 | 5/9/2028 | |||||||||
Federico A. Monzon, M.D. |
11/1/2015 | 77,083 | 22,917 | 1.57 | 1/17/2026 | ||||||||||
5/10/2018 | 15,000 | (5) |
— | 1.96 | 5/9/2028 | ||||||||||
11/12/2018 | — | 15,000 | 1.96 | 11/11/2028 | |||||||||||
| (1) | All of the option awards were granted under either the 2008 Plan or the 2018 Plan, the terms of which are described below under “—Equity Benefit Plans.” |
| (2) | Each option award vests as follows: 25% of the shares subject to the option vest on the first anniversary of the vesting commencement date, and the balance of the shares vest in equal monthly installments thereafter over the next 36 months, provided in each case that the holder is then providing services to us in accordance with the terms of the 2008 Plan or the 2018 Plan, as applicable. Certain option awards are subject to acceleration, as described above under “— Potential Payments and Benefits upon Termination or Change in Control.” |
| (3) | Unless otherwise indicated, all of the option awards were granted with a per share exercise price equal to the fair market value of one share of our common stock on the date of grant, as determined in good faith by our board of directors. |
| (4) | The fair market value of one share of our common stock on the date of grant, as determined in good faith by our board of directors, was $1.42. On the date of grant, Mr. Maetzold owned more than 10% of the voting power of all classes of our stock. Pursuant to the 2008 Plan, the exercise price per share for the incentive stock option award granted to Mr. Maetzold on 6/4/2015 is $1.57, which represents 110% of the fair market value of one share of our common stock on the date of grant. |
| (5) | The option is exercisable immediately, in whole or in part, conditioned upon the named executive officer’s entering into a restricted stock purchase agreement with respect to any unvested shares. The shares subject to the option vest and/or are released from the Company’s repurchase option, as to 1/4th of the shares subject to the option on the first anniversary of the vesting commencement date, and thereafter as to 1/48th of the shares subject to such option on each monthly anniversary of the vesting commencement date, such that all shares will be vested on the fourth anniversary of the vesting commencement date, subject to the holder continuing to provide services to the Company through such vesting date. |
We did not engage in any repricings or other modifications or cancellations to any of our named executive officers’ outstanding equity awards during the year ended December 31, 2018.
Perquisites, Health, Welfare and Retirement Benefits
Our named executive officers, during their employment with us, are eligible to participate in our employee benefit plans, including our medical, dental, vision, group term life, disability, employee assistance, and accidental death and dismemberment insurance plans, in each case on the same basis as all of our other employees. In addition, we provide a 401(k) plan to our employees, including our named executive officers, as discussed in the section below entitled “—401(k) Plan.”
We generally do not provide perquisites or personal benefits to our named executive officers, except in limited circumstances. We do, however, pay a portion of the premiums for medical, dental, vision, group term life, disability, employee assistance and accidental death and dismemberment insurance for all of our employees who work at least 30 hours per week, including our named executive officers. Our board of directors may elect to adopt qualified or nonqualified benefit plans in the future if it determines that doing so is in our best interests and in the best interests of our stockholders.
401(k) Plan
We maintain a defined contribution employee retirement plan, or 401(k) plan, for our employees. Our executive officers are eligible to participate in the 401(k) plan on the same basis as our other employees. The 401(k) plan
134
is intended to qualify as a tax-qualified plan under Section 401(k) of the Code. The 401(k) plan provides that each participant may contribute up to the lesser of 100% of his or her compensation or the statutory limit, which was $18,500 for calendar year 2018. Participants that are 50 years or older can also make “catch-up” contributions, which in calendar year 2018 was up to an additional $6,000 above the statutory limit. We currently make matching contributions into the 401(k) plan on behalf of participants. Participant contributions are held and invested, pursuant to the participant’s instructions, by the plan’s trustee.
Nonqualified Deferred Compensation
Our named executive officers did not participate in, or earn any benefits under, a nonqualified deferred compensation plan sponsored by us during the year ended December 31, 2018. Our board of directors may elect to provide our officers and other employees with nonqualified defined contribution or other nonqualified deferred compensation benefits in the future if it determines that doing so is in our best interests.
Equity Benefit Plans
The principal features of our equity plans are summarized below. These summaries are qualified in their entirety by reference to the actual text of the plans, which are filed as exhibits to the registration statement of which this prospectus is a part.
2019 Equity Incentive Plan
Our board of directors adopted, and our stockholders approved, the 2019 Plan in 2019. The 2019 Plan will become effective immediately prior to and contingent upon the date of the underwriting agreement related to this offering. The 2019 Plan is a successor to and continuation of the 2018 Plan. No stock awards may be granted under the 2019 Plan until the date of the underwriting agreement related to this offering. Once the 2019 Plan is effective, no further grants will be made under the 2018 Plan.
Stock Awards. The 2019 Plan provides for the grant of incentive stock options, or ISOs, within the meaning of Section 422 of the Code, to employees, including employees of any parent or subsidiary, and for the grant of nonstatutory stock options, or NSOs, stock appreciation rights, restricted stock awards, restricted stock unit awards, performance stock awards, performance cash awards and other forms of stock awards to employees, directors and consultants, including employees and consultants of our affiliates.
Authorized Shares. Initially, the maximum number of shares of our common stock that may be issued under the 2019 Plan after it becomes effective will be shares, which is the sum of (1) new shares, plus (2) the number of shares (not to exceed shares) (i) that remain available for the issuance of awards under the 2018 Plan at the time the 2019 Plan becomes effective, and (ii) any shares subject to outstanding stock options or other stock awards that were granted under the 2008 Plan and the 2018 Plan that terminate or expire prior to exercise or settlement; are forfeited because of the failure to vest or otherwise return to us; or are reacquired or withheld (or not issued) to satisfy a tax withholding obligation or the purchase or exercise price. In addition, the number of shares of our common stock reserved for issuance under the 2019 Plan will automatically increase on January 1 of each calendar year, starting on January 1, 2020 (assuming the 2019 Plan becomes effective in 2019) through January 1, 2029, in an amount equal to 5% of the total number of shares of our capital stock outstanding on the last day of the calendar year before the date of each automatic increase, or a lesser number of shares determined by our board of directors. The maximum number of shares of our common stock that may be issued on the exercise of ISOs under the 2019 Plan is .
Shares subject to stock awards granted under the 2019 Plan that expire or terminate without being exercised in full or that are paid out in cash rather than in shares do not reduce the number of shares available for issuance under the 2019 Plan. If any shares of common stock issued pursuant to a stock award are forfeited back to or repurchased or reacquired by us for any reason, the shares that are forfeited or repurchased or reacquired will revert to and again become available for issuance under the 2019 Plan. Any shares reacquired in satisfaction of tax withholding obligations or as consideration for the exercise or purchase price of a stock award will again become available for issuance under the 2019 Plan.
Plan Administration. Our board of directors, or a duly authorized committee of our board of directors, will administer the 2019 Plan and is referred to as the “plan administrator” herein. Our board of directors may also delegate to one or more of our officers the authority to (1) designate employees (other than officers) to receive
135
specified stock awards and (2) determine the number of shares subject to such stock awards. Under the 2019 Plan, our board of directors has the authority to determine award recipients, grant dates, the numbers and types of stock awards to be granted, the applicable fair market value, and the provisions of each stock award, including the period of exercisability and the vesting schedule applicable to a stock award.
Under the 2019 Plan, the board of directors also generally has the authority to effect, with the consent of any adversely affected participant, (A) the reduction of the exercise, purchase, or strike price of any outstanding award, (B) the cancellation of any outstanding award and the grant in substitution therefore of other awards, cash, or other consideration, or (C) any other action that is treated as a repricing under generally accepted accounting principles.
Stock Options. ISOs and NSOs are granted under stock option agreements adopted by the plan administrator. The plan administrator determines the exercise price for stock options, within the terms and conditions of the 2019 Plan, provided that the exercise price of a stock option generally cannot be less than 100% of the fair market value of our common stock on the date of grant. Options granted under the 2019 Plan vest at the rate specified in the stock option agreement as determined by the plan administrator.
The plan administrator determines the term of stock options granted under the 2019 Plan, up to a maximum of 10 years. Unless the terms of an optionholder’s stock option agreement provide otherwise, if an optionholder’s service relationship with us or any of our affiliates ceases for any reason other than disability, death, or cause, the optionholder may generally exercise any vested options for a period of three months following the cessation of service. This period may be extended in the event that exercise of the option is prohibited by applicable securities laws or our insider trading policy. If an optionholder’s service relationship with us or any of our affiliates ceases due to death, or an optionholder dies within a certain period following cessation of service, the optionholder or a beneficiary may generally exercise any vested options for a period of 18 months following the date of death. If an optionholder’s service relationship with us or any of our affiliates ceases due to disability, the optionholder may generally exercise any vested options for a period of 12 months following the cessation of service. In the event of a termination for cause, options generally terminate upon the termination date. In no event may an option be exercised beyond the expiration of its term.
Acceptable consideration for the purchase of common stock issued upon the exercise of a stock option will be determined by the plan administrator and may include (1) cash, check, bank draft or money order, (2) a broker- assisted cashless exercise, (3) the tender of shares of our common stock previously owned by the optionholder, (4) a net exercise of the option if it is an NSO, or (5) other legal consideration approved by the plan administrator.
Unless the plan administrator provides otherwise, options generally are not transferable except by will or the laws of descent and distribution. Subject to approval of the plan administrator or a duly authorized officer in each case, (i) an option may be transferred pursuant to a domestic relations order, official marital settlement agreement, or other divorce or separation instrument and (ii) an optionholder may designate a beneficiary who may exercise the option following the optionholder’s death.
Tax Limitations on ISOs. The aggregate fair market value, determined at the time of grant, of our common stock with respect to ISOs that are exercisable for the first time by an award holder during any calendar year under all of our stock plans may not exceed $100,000. Options or portions thereof that exceed such limit will generally be treated as NSOs. No ISO may be granted to any person who, at the time of the grant, owns or is deemed to own stock possessing more than 10% of our total combined voting power or that of any of our affiliates unless (1) the option exercise price is at least 110% of the fair market value of the stock subject to the option on the date of grant, and (2) the term of the ISO does not exceed five years from the date of grant.
Restricted Stock Unit Awards. Restricted stock unit awards are granted under restricted stock unit award agreements adopted by the plan administrator. Restricted stock unit awards may be granted in consideration for any form of legal consideration that may be acceptable to our board of directors and permissible under applicable law. A restricted stock unit award may be settled by cash, delivery of stock, a combination of cash and stock as deemed appropriate by the plan administrator, or in any other form of consideration set forth in the restricted stock unit award agreement. Additionally, dividend equivalents may be credited in respect of shares covered by a restricted stock unit award. Except as otherwise provided in the applicable award agreement, restricted stock unit awards that have not vested will be forfeited once the participant’s continuous service ends for any reason.
136
Restricted Stock Awards. Restricted stock awards are granted under restricted stock award agreements adopted by the plan administrator. A restricted stock award may be awarded in consideration for cash, check, bank draft or money order, past or future services to us, or any other form of legal consideration that may be acceptable to our board of directors and permissible under applicable law. The plan administrator determines the terms and conditions of restricted stock awards, including vesting and forfeiture terms. If a participant’s service relationship with us ends for any reason, we may receive any or all of the shares of common stock held by the participant that have not vested as of the date the participant terminates service with us through a forfeiture condition or a repurchase right.
Stock Appreciation Rights. Stock appreciation rights are granted under stock appreciation right agreements adopted by the plan administrator. The plan administrator determines the purchase price or strike price for a stock appreciation right, which generally cannot be less than 100% of the fair market value of our common stock on the date of grant. A stock appreciation right granted under the 2019 Plan vests at the rate specified in the stock appreciation right agreement as determined by the plan administrator.
The plan administrator determines the term of stock appreciation rights granted under the 2019 Plan, up to a maximum of ten years. If a participant’s service relationship with us or any of our affiliates ceases for any reason other than cause, disability, or death, the participant may generally exercise any vested stock appreciation right for a period of three months following the cessation of service. This period may be further extended in the event that exercise of the stock appreciation right following such a termination of service is prohibited by applicable securities laws or our insider trading policy. If a participant’s service relationship with us, or any of our affiliates, ceases due to disability or death, or a participant dies within a certain period following cessation of service, the participant or a beneficiary may generally exercise any vested stock appreciation right for a period of 12 months in the event of disability and 18 months in the event of death. In the event of a termination for cause, stock appreciation rights generally terminate immediately upon the occurrence of the event giving rise to the termination of the individual for cause. In no event may a stock appreciation right be exercised beyond the expiration of its term.
Performance Awards. The 2019 Plan permits the grant of performance-based stock and cash awards. Our compensation committee may structure awards so that the stock or cash will be issued or paid only following the achievement of certain pre-established performance goals during a designated performance period.
The performance goals that may be selected include one or more of the following: (i) sales; (ii) revenues; (iii) assets; (iv) expenses; (v) market penetration or expansion; (vi) earnings from operations; (vii) earnings before or after deduction for all or any portion of interest, taxes, depreciation, amortization, incentives, service fees or extraordinary or special items, whether or not on a continuing operations or an aggregate or per share basis; (viii) net income or net income per common share (basic or diluted); (ix) return on equity, investment, capital or assets; (x) one or more operating ratios; (xi) borrowing levels, leverage ratios or credit rating; (xii) market share; (xiii) capital expenditures; (xiv) cash flow, free cash flow, cash flow return on investment, or net cash provided by operations; (xv) stock price, dividends or total stockholder return; (xvi) development of new technologies or products; (xvii) sales of particular products or services; (xviii) economic value created or added; (xix) operating margin or profit margin; (xx) customer acquisition or retention; (xxi) raising or refinancing of capital; (xxii) successful hiring of key individuals; (xxiii) resolution of significant litigation; (xxiv) acquisitions and divestitures (in whole or in part); (xxv) joint ventures and strategic alliances; (xxvi) spin-offs, split-ups and the like; (xxvii) reorganizations; (xxviii) recapitalizations, restructurings, financings (issuance of debt or equity) or refinancings; (xxix) or strategic business criteria, consisting of one or more objectives based on the following goals: achievement of timely development, design management or enrollment, meeting specified market penetration or value added, payor acceptance, patient adherence, peer reviewed publications, issuance of new patents, establishment of or securing of licenses to intellectual property, product development or introduction (including, without limitation, any clinical trial accomplishments, regulatory or other filings, approvals or milestones, discovery of novel products, maintenance of multiple products in pipeline, product launch or other product development milestones), geographic business expansion, cost targets, cost reductions or savings, customer satisfaction, operating efficiency, acquisition or retention, employee satisfaction, information technology, corporate development (including, without limitation, licenses, innovation, research or establishment
137
of third-party collaborations), manufacturing or process development, legal compliance or risk reduction, patent application or issuance goals, or goals relating to acquisitions, divestitures or other business combinations (in whole or in part), joint ventures or strategic alliances; and (xxx) other measures of performance selected by the board of directors.
The performance goals may be based on company-wide performance or performance of one or more business units, divisions, affiliates, or business segments, and may be either absolute or relative to the performance of one or more comparable companies or the performance of one or more relevant indices. Our board of directors is authorized at any time in its sole discretion, to adjust or modify the calculation of a performance goal for such performance period in order to prevent the dilution or enlargement of the rights of participants, (a) in the event of, or in anticipation of, any unusual or extraordinary corporate item, transaction, event or development; (b) in recognition of, or in anticipation of, any other unusual or nonrecurring events affecting us, or our financial statements in response to, or in anticipation of, changes in applicable laws, regulations, accounting principles, or business conditions; or (c) in view of the board of director’s assessment of our business strategy, performance of comparable organizations, economic and business conditions, and any other circumstances deemed relevant. Specifically, the board of directors is authorized to make adjustment in the method of calculating attainment of performance goals and objectives for a performance period as follows: (i) to exclude the dilutive effects of acquisitions or joint ventures; (ii) to assume that any business divested by us achieved performance objectives at targeted levels during the balance of a performance period following such divestiture; and (iii) to exclude the effect of any change in the outstanding shares of our common stock by reason of any stock dividend or split, stock repurchase, reorganization, recapitalization, merger, consolidation, spin-off, combination or exchange of shares or other similar corporate change, or any distributions to common stockholders other than regular cash dividends. In addition, the board of directors is authorized to make adjustment in the method of calculating attainment of performance goals and objectives for a performance period as follows: (i) to exclude restructuring and/or other nonrecurring charges; (ii) to exclude exchange rate effects, as applicable, for non-U.S. dollar denominated net sales and operating earnings; (iii) to exclude the effects of changes to generally accepted accounting standards required by the Financial Accounting Standards Board; (iv) to exclude the effects of any items that are “unusual” in nature or occur “infrequently” as determined under generally accepted accounting principles; (v) to exclude the effects to any statutory adjustments to corporate tax rates; and (vi) to make other appropriate adjustments selected by the board of directors.
Other Stock Awards. The plan administrator may grant other awards based in whole or in part by reference to our common stock. The plan administrator will set the number of shares under the stock award and all other terms and conditions of such awards.
Changes to Capital Structure. In the event there is a specified type of change in our capital structure, such as a stock split, reverse stock split, or recapitalization, appropriate adjustments will be made to (1) the class and maximum number of shares reserved for issuance under the 2019 Plan, (2) the class and maximum number of shares by which the share reserve may increase automatically each year, (3) the class and maximum number of shares that may be issued on the exercise of ISOs and (4) the class and number of shares and exercise price, strike price, or purchase price, if applicable, of all outstanding stock awards.
Corporate Transactions. The 2019 Plan provides that in the event of certain specified significant corporate transactions (or a change in control, as defined below), unless otherwise provided in an award agreement or other written agreement between us and the award holder, the plan administrator may take one or more of the following actions with respect to such stock awards:
| • | arrange for the assumption, continuation, or substitution of a stock award by a successor corporation; |
| • | arrange for the assignment of any reacquisition or repurchase rights held by us to a successor corporation; |
| • | accelerate the vesting, in whole or in part, of the stock award and provide for its termination if not exercised (if applicable) at or before the effective time of the transaction; |
| • | arrange for the lapse, in whole or in part, of any reacquisition or repurchase rights held by us; |
| • | cancel or arrange for the cancellation of the stock award, to the extent not vested or not exercised before the effective time of the transaction, in exchange for a cash payment, if any; or |
138
| • | make a payment equal to the excess, if any, of (A) the value of the property the participant would have received on exercise of the award immediately before the effective time of the transaction, over (B) any exercise price payable by the participant in connection with the exercise. |
The plan administrator is not obligated to treat all stock awards or portions of stock awards in the same manner and is not obligated to take the same actions with respect to all participants.
Under the 2019 Plan, a corporate transaction is generally the consummation of: (1) a sale or other disposition of all or substantially all of our assets, (2) the sale or other disposition of more than 50% of our outstanding securities, (3) a merger or consolidation where we do not survive the transaction or (4) a merger or consolidation where we do survive the transaction but the shares of our common stock outstanding immediately before such transaction are converted or exchanged into other property by virtue of the transaction.
Change in Control. In the event of a change in control, the plan administrator may take any of the above-mentioned actions. Awards granted under the 2019 Plan may be subject to additional acceleration of vesting and exercisability upon or after a change in control as may be provided in the applicable stock award agreement or in any other written agreement between us or any affiliate and the participant, but in the absence of such provision, no such acceleration will automatically occur. Under the 2019 Plan, a change in control is generally (1) the acquisition by any person or company of more than 50% of the combined voting power of our then outstanding stock, (2) a merger, consolidation or similar transaction in which our stockholders immediately before the transaction do not own, directly or indirectly, more than 50% of the combined voting power of the surviving entity (or the parent of the surviving entity) in substantially the same proportions as their ownership immediately prior to such transaction, (3) a sale, lease, exclusive license or other disposition of all or substantially all of our assets other than to an entity more than 50% of the combined voting power of which is owned by our stockholders in substantially the same proportions as their ownership of our outstanding voting securities immediately prior to such transaction, (4) a complete dissolution or liquidation of us or (5) when a majority of our board of directors becomes comprised of individuals who were not serving on our board of directors on the date of the underwriting agreement related to this offering, or the incumbent board, or whose nomination, appointment, or election was not approved by a majority of the incumbent board still in office.
Plan Amendment or Termination. Our board of directors has the authority to amend, suspend, or terminate the 2019 Plan, provided that such action does not materially impair the existing rights of any participant without such participant’s written consent. Certain material amendments also require the approval of our stockholders. No ISOs may be granted after the tenth anniversary of the earlier of (A) the date our board of directors adopts the 2019 Plan or (B) the date the 2019 Plan is approved by our stockholders. No stock awards may be granted under the 2019 Plan while it is suspended or after it is terminated.
2018 Equity Incentive Plan
Our board of directors adopted the 2018 Plan in August 2018 and our stockholders approved the 2018 Plan in October 2018. As of March 31, 2019, 183,109 shares were available for the future grant of stock awards under the 2018 Plan and 730,500 shares were subject to outstanding stock awards that were granted under the 2018 Plan. We expect that any shares remaining available for issuance under the 2018 Plan will become available for issuance under the 2019 Plan in connection with this offering.
Stock Awards. The 2018 Plan provides for the grant of ISOs within the meaning of Section 422 of the Code to employees, including employees of any parent or subsidiary, and for the grant of NSOs, stock appreciation rights, restricted stock awards, restricted stock unit awards and other stock awards to employees, directors and consultants, including employees and consultants of our affiliates. To date, we have only granted stock options under the 2018 Plan.
Authorized Shares. Subject to certain capitalization adjustments, the aggregate number of shares of common stock that may be issued pursuant to stock awards under the 2018 Plan may not exceed a maximum of 676,607 shares, or the 2018 Plan’s share reserve, which number is the sum of (i) 500,000 new shares plus (ii) 176,607 shares subject to the 2008 Plan’s share reserve as of the effective date of the 2018 Plan. The 2018 Plan’s share reserve may be increased by up to 1,904,637 shares, which is equal to the number of shares that were subject to outstanding stock awards under the 2008 Plan as of the effective date of the 2018 Plan, that expire or terminate for any reason, are forfeited or otherwise return to us or are reacquired, withheld or not issued to satisfy a tax withholding obligation or to satisfy the exercise or purchase price of a stock award, as
139
such shares become available from time to time. The maximum number of shares of our common stock that may be issued pursuant to the exercise of ISOs under the 2018 Plan is 5,162,488 shares.
Shares subject to stock awards granted under the 2018 Plan that expire or terminate without all of the shares covered by such stock award having been issued or that are settled in cash rather than in shares do not reduce the number of shares available for issuance under the 2018 Plan. Additionally, if any shares issued pursuant to a stock award are forfeited back to or repurchased or reacquired by us for any reason, including because of the failure to meet a contingency or condition required to vest, then the shares that are forfeited, repurchased or reacquired will revert to and again become available for issuance under the 2018 Plan. This includes shares used to pay the exercise price of a stock award or to satisfy the tax withholding obligations related to a stock award.
Plan Administration. Our board of directors, or a duly authorized committee of our board of directors, will administer the 2018 Plan and is referred to as the “plan administrator” herein. The plan administrator may also delegate to one or more of our officers the authority to (1) designate employees (other than officers) to receive specified stock awards and (2) determine the number of shares subject to such stock awards. Under the 2018 Plan, the plan administrator has the authority to determine award recipients, dates of grant, the numbers and types of stock awards to be granted, the applicable fair market value and the provisions of each stock award, including the period of their exercisability and the vesting schedule applicable to a stock award.
Under the 2018 Plan, the plan administrator also generally has the authority to effect, with the consent of any adversely affected participant, (1) the reduction of the exercise, purchase, or strike price of any outstanding award; (2) the cancellation of any outstanding award and the grant in substitution therefore of other awards, cash, or other consideration; or (3) any other action that is treated as a repricing under generally accepted accounting principles.
Stock Options. ISOs and NSOs are granted under stock option agreements adopted by the plan administrator. The plan administrator determines the exercise price for stock options, within the terms and conditions of the 2018 Plan, provided that the exercise price of a stock option generally cannot be less than 100% of the fair market value of our common stock on the date of grant. Options granted under the 2018 Plan vest at the rate specified in the stock option agreement as determined by the plan administrator.
The plan administrator determines the term of stock options granted under the 2018 Plan, up to a maximum of 10 years. If an optionholder’s service relationship with us or any of our affiliates ceases for any reason other than disability, death or cause, the optionholder may generally exercise any vested options for a period of three months following the cessation of service. This period may be extended in the event that exercise of the option is prohibited by applicable securities laws or if the sale of common stock received upon exercise would violate our insider trading policy. If an optionholder’s service relationship with us or any of our affiliates ceases due to death, or an optionholder dies within a certain period following cessation of service, the optionholder or a beneficiary may generally exercise any vested options for a period of 18 months following the date of death. If an optionholder’s service relationship with us or any of our affiliates ceases due to disability, the optionholder may generally exercise any vested options for a period of 12 months following the cessation of service.
In the event of a termination for cause, options generally terminate upon the termination date. In no event may an option be exercised beyond the expiration of its term.
Acceptable consideration for the purchase of common stock issued upon the exercise of a stock option will be determined by the plan administrator and may include (1) cash, check, bank draft or money order, (2) a broker-assisted cashless exercise, (3) the tender of shares of our common stock previously owned by the optionholder, (4) a net exercise of the option if it is an NSO, (5) a deferred payment arrangement or (6) other legal consideration approved by the plan administrator.
Unless the plan administrator provides otherwise, options generally are not transferable except by will or the laws of descent and distribution. Subject to approval of the plan administrator or a duly authorized officer in each case, (1) an option may be transferred pursuant to a domestic relations order, official marital settlement agreement, or other divorce or separation instrument and (2) an optionholder may designate a beneficiary who may exercise the option following the optionholder’s death.
Tax Limitations on ISOs. The aggregate fair market value, determined at the time of grant, of our common stock with respect to ISOs that are exercisable for the first time by an optionholder during any calendar year under all of our stock plans may not exceed $100,000. Options or portions thereof that exceed such limit will generally be
140
treated as NSOs. No ISO may be granted to any person who, at the time of the grant, owns or is deemed to own stock possessing more than 10% of our total combined voting power or that of any of our affiliates unless (1) the option exercise price is at least 110% of the fair market value of the stock subject to the option on the date of grant and (2) the term of the ISO does not exceed five years from the date of grant.
Changes to Capital Structure. In the event there is a specified type of change in our capital structure, such as a stock split, reverse stock split, or recapitalization, appropriate and proportionate adjustments will be made to (1) the class and maximum number of shares reserved for issuance under the 2018 Plan, (2) the class and maximum number of shares that may be issued on the exercise of ISOs and (3) the class and number of shares and exercise price, strike price, or purchase price, if applicable, of all outstanding stock awards.
Transactions. The 2018 Plan provides that in the event of a corporate transaction or a change in control, each outstanding stock award shall be treated as the plan administrator determines, including, without limitation, that each stock award may be assumed, continued or a similar award substituted for such stock award by the surviving or acquiring corporation (or its parent). Notwithstanding the foregoing, in the event of a change in control in which the surviving or acquiring corporation (or its parent) does not assume, continue or substitute for such outstanding stock awards, then the vesting and exercisability of such stock awards shall accelerate in full. In addition, the plan administrator shall notify the participant that the stock award shall be fully vested and exercisable for a specified period of time, and any such award shall terminate upon the expiration of such period for no consideration, unless otherwise determined by the plan administrator.
The plan administrator is not obligated to treat all stock awards similarly in the transaction.
In addition, a stock award under the 2018 Plan may be subject to additional acceleration of vesting and exercisability upon or after a change in control as may be provided in the award agreement or other written agreement between us and the participant, but in the absence of such provision, no such acceleration will automatically occur, except as described above.
Under the 2018 Plan, a corporate transaction is generally the consummation of (1) a sale or other disposition of all or substantially all of our assets, (2) the sale or other disposition of more than 50% of our outstanding securities, (3) a merger, consolidation or similar transaction where we do not survive the transaction or (4) a merger, consolidation or similar transaction where we do survive the transaction but the shares of our common stock outstanding immediately before such transaction are converted or exchanged into other property by virtue of the transaction.
Under the 2018 Plan, a change in control is generally (1) the acquisition by any person or company of more than 50% of the combined voting power of our then outstanding stock, (2) a consummated merger, consolidation or similar transaction in which our stockholders immediately before the transaction do not own, directly or indirectly, more than 50% of the combined voting power of the surviving entity (or the parent of the surviving entity) in substantially the same proportions as their ownership immediately prior to such transaction, (3) a complete dissolution or liquidation of us, (4) a consummated sale, lease, exclusive license or other disposition of all or substantially all of our assets other than to an entity more than 50% of the combined voting power of which is owned by our stockholders in substantially the same proportions as their ownership of our outstanding voting securities immediately prior to such transaction or (5) when a majority of our board of directors becomes comprised of individuals who were not serving on our board of directors on the date the 2018 Plan was adopted, or the incumbent board, or whose nomination, appointment or election was not approved by a majority of the incumbent board still in office.
Plan Amendment or Termination. Our board of directors has the authority to amend, suspend, or terminate the 2018 Plan, provided that such action does not impair the existing rights of any participant without such participant’s written consent. Certain material amendments also require the approval of our stockholders. Unless terminated sooner, the 2018 Plan will automatically terminate on August 14, 2028. No stock awards may be granted under the 2018 Plan while it is suspended or after it is terminated.
2008 Stock Plan
Our board of directors adopted the 2008 Plan in September 2008 and our stockholders approved the 2008 Plan in September 2008. The 2008 Plan was further amended by our board of directors and stockholders, most recently in May 2017. No further awards were granted upon the effective date of the 2018 Plan. As of March 31, 2019, there were outstanding stock options covering a total of 1,844,658 shares that were granted under the 2008 Plan.
141
Stock Awards. The 2008 Plan provides for the grant of ISOs within the meaning of Section 422 of the Code to employees, including employees of any parent or subsidiary, and for the grant of NSOs and restricted stock awards to employees, directors and consultants, including employees and consultants of our affiliates.
Authorized Shares. Shares are no longer available for the grant of stock awards under the 2008 Plan. However, if a stock award granted under the 2008 Plan expires or terminates, is forfeited or otherwise returns to us or is reacquired, withheld or not issued to satisfy a tax withholding obligation or to satisfy the exercise or purchase price of a stock award, the shares of our common stock not acquired pursuant to the stock award again will become available for subsequent issuance under the 2019 Plan.
Plan Administration. Our board of directors, or a committee appointed by our board of directors, has the authority to administer the 2008 Plan and is referred to as the “plan administrator” herein. Under the 2008 Plan, the plan administrator has the authority to determine award recipients, dates of grant, the numbers and types of stock awards to be granted, the applicable fair market value and the provisions of each stock award, including the period of their exercisability and the vesting schedule applicable to a stock award.
Under the 2018 Plan, the plan administrator also generally has the authority to institute an exchange program under which (i) outstanding options are surrendered or cancelled in exchange for options and/or cash and/or (ii) the exercise price of an outstanding option is reduced.
Stock Options. ISOs and NSOs are granted under stock option agreements adopted by the plan administrator. The plan administrator determines the exercise price for stock options, within the terms and conditions of the 2008 Plan, provided that the exercise price of a stock option generally cannot be less than 100% of the fair market value of our common stock on the date of grant. Options granted under the 2008 Plan vest at the rate specified in the stock option agreement as determined by the plan administrator.
The plan administrator determines the term of stock options granted under the 2008 Plan, up to a maximum of ten years. If an optionholder’s service relationship with us or any of our affiliates ceases for any reason other than disability or death, the optionholder may generally exercise any vested options for a period of three months following the cessation of service. If an optionholder’s service relationship with us or any of our affiliates ceases due to death, the optionholder or a beneficiary may generally exercise any vested options for a period of 12 months following the date of death. If an optionholder’s service relationship with us or any of our affiliates ceases due to disability, the optionholder may generally exercise any vested options for a period of 12 months following the cessation of service. In no event may an option be exercised beyond the expiration of its term.
Acceptable consideration for the purchase of common stock issued upon the exercise of a stock option will be determined by the plan administrator and may include (1) cash, (2) check, (3) promissory note, (4) delivery of other shares, (5) a cashless exercise program, (6) such other consideration permitted by applicable laws or (7) any combination of the foregoing.
Unless the plan administrator provides otherwise, options generally are not transferable except by will or the laws of descent and distribution. Subject to approval of the plan administrator, an optionholder may designate a beneficiary who may exercise the option following the optionholder’s death.
Tax Limitations on ISOs. The aggregate fair market value, determined at the time of grant, of our common stock with respect to ISOs that are exercisable for the first time by an optionholder during any calendar year under all of our stock plans may not exceed $100,000. Options or portions thereof that exceed such limit will generally be treated as NSOs. No ISO may be granted to any person who, at the time of the grant, owns or is deemed to own stock possessing more than 10% of our total combined voting power or that of any of our affiliates unless (1) the option exercise price is at least 110% of the fair market value of the stock subject to the option on the date of grant and (2) the term of the ISO does not exceed five years from the date of grant.
Changes to Capital Structure. In the event there is a specified type of change in our capital structure, such as a stock split, reverse stock split, or recapitalization, adjustments will be made to the number and class of shares that may be delivered under the 2008 Plan and/or the number, class, and price of shares covered by each outstanding award.
Transactions. The 2008 Plan provides that in the event of a merger or a change in control, each outstanding stock award shall be treated as the plan administrator determines, including, without limitation, that each stock award may be assumed or a similar award substituted for such stock award by the successor corporation (or its
142
parent). Notwithstanding the foregoing, in the event of a change in control in which the successor corporation (or its parent) does not assume or substitute for such outstanding stock awards, then the vesting and exercisability of such stock awards shall accelerate in full. In addition, the plan administrator shall notify the participant that the stock award shall be fully vested and exercisable for a specified period of time, and any such award shall terminate upon the expiration of such period for no consideration, unless otherwise determined by the plan administrator. The plan administrator is not obligated to treat all stock awards similarly in the transaction.
Under the 2008 Plan, a change in control generally means the occurrence of any of the following: (1) the acquisition by any person or group of more than 50% of the total voting power of our stock; (2) a majority of members of the board of directors is replaced during any 12 month period by directors whose appointment or election is not endorsed by a majority of the members of the board of directors prior to the date of the appointment or election; or (3) any person acquires (or has acquired during the 12 month period ending on the date of the most recent acquisition by such person or persons) assets from us that have a total gross fair market value equal to or more than 50% of the total gross fair market value of all of our assets immediately prior to such acquisition or acquisitions.
2019 Employee Stock Purchase Plan
Our board of directors adopted, and our stockholders approved, the ESPP in 2019. The ESPP will become effective immediately prior to and contingent upon the date of the underwriting agreement related to this offering. The purpose of the ESPP is to secure the services of new employees, to retain the services of existing employees, and to provide incentives for such individuals to exert maximum efforts toward our success and that of our affiliates. The ESPP is intended to qualify as an “employee stock purchase plan” within the meaning of Section 423 of the Code for U.S. employees.
Share Reserve. Following this offering, the ESPP authorizes the issuance of shares of our common stock under purchase rights granted to our employees or to employees of any of our designated affiliates. The number of shares of our common stock reserved for issuance will automatically increase on January 1 of each calendar year, beginning on January 1, 2020 (assuming the ESPP becomes effective in 2019) through January 1, 2029, by the lesser of (1) 1% of the total number of shares of our common stock outstanding on the last day of the calendar year before the date of the automatic increase and (2) shares; provided that before the date of any such increase, our board of directors may determine that such increase will be less than the amount set forth in clauses (1) and (2). As of the date hereof, no shares of our common stock have been purchased under the ESPP.
Administration. Our board of directors, or a committee of our board of directors, will administer the ESPP. The ESPP is implemented through a series of offerings under which eligible employees are granted purchase rights to purchase shares of our common stock on specified dates during such offerings. Under the ESPP, we may specify offerings with durations of not more than 27 months, and may specify shorter purchase periods within each offering. Each offering will have one or more purchase dates on which shares of our common stock will be purchased for employees participating in the offering. An offering under the ESPP may be terminated under certain circumstances.
Payroll Deductions. Generally, all regular employees, including executive officers, employed by us or by any of our designated affiliates, may participate in the ESPP and may contribute, normally through payroll deductions, up to 15% of their earnings (as defined in the ESPP) for the purchase of our common stock under the ESPP. Unless otherwise determined by our board of directors, common stock will be purchased for the accounts of employees participating in the ESPP at a price per share that is at least the lesser of (1) 85% of the fair market value of a share of our common stock on the first date of an offering or (2) 85% of the fair market value of a share of our common stock on the date of purchase.
Limitations. Employees may have to satisfy one or more of the following service requirements before participating in the ESPP, as determined by our board of directors, including: (1) being customarily employed for more than 20 hours per week, (2) being customarily employed for more than five months per calendar year or (3) continuous employment with us or one of our affiliates for a period of time (not to exceed two years). No employee may purchase shares under the ESPP at a rate in excess of $25,000 worth of our common stock based on the fair market value per share of our common stock at the beginning of an offering for each calendar year
143
such a purchase right is outstanding. Finally, no employee will be eligible for the grant of any purchase rights under the ESPP if immediately after such rights are granted, such employee has voting power over 5% or more of our outstanding capital stock measured by vote or value under Section 424(d) of the Code.
Changes to Capital Structure. In the event that there occurs a change in our capital structure through such actions as a stock split, merger, consolidation, reorganization, recapitalization, reincorporation, stock dividend, dividend in property other than cash, large nonrecurring cash dividend, liquidating dividend, combination of shares, exchange of shares, change in corporate structure, or similar transaction, the board of directors will make appropriate adjustments to: (1) the class(es) and maximum number of shares reserved under the ESPP, (2) the class(es) and maximum number of shares by which the share reserve may increase automatically each year, (3) the class(es) and number of shares subject to and purchase price applicable to outstanding offerings and purchase rights and (4) the class(es) and number of shares that are subject to purchase limits under ongoing offerings.
Corporate Transactions. In the event of certain significant corporate transactions, any then-outstanding rights to purchase our stock under the ESPP may be assumed, continued, or substituted for by any surviving or acquiring entity (or its parent company). If the surviving or acquiring entity (or its parent company) elects not to assume, continue, or substitute for such purchase rights, then the participants’ accumulated payroll contributions will be used to purchase shares of our common stock within 10 business days before such corporate transaction, and such purchase rights will terminate immediately.
Under the ESPP, a corporate transaction is generally the consummation of: (1) a sale or other disposition of all or substantially all of our assets; (2) the sale or other disposition of more than 50% of our outstanding securities; (3) a merger or consolidation where we do not survive the transaction; and (4) a merger or consolidation where we do survive the transaction but the shares of our common stock outstanding immediately before such transaction are converted or exchanged into other property by virtue of the transaction.
ESPP Amendment or Termination. Our board of directors has the authority to amend or terminate the ESPP, provided that except in certain circumstances such amendment or termination may not materially impair any outstanding purchase rights without the holder’s consent. We will obtain stockholder approval of any amendment to the ESPP as required by applicable law or listing requirements.
Director Compensation
The following table sets forth in summary form information concerning the compensation that was earned by each of our non-employee directors during the year ended December 31, 2018:
Name(1) |
Fees Earned or Paid in Cash |
Total |
||||
Bonnie H. Anderson |
$ | 20,000 | (2) |
$ | 20,000 | |
Mara G. Aspinall |
$ | 46,000 | (2) |
$ | 46,000 | |
Daniel M. Bradbury |
— | — | ||||
G. Bradley Cole |
— | — | ||||
Joseph C. Cook III |
— | — | ||||
Joseph C. Cook, Jr . |
— | — | ||||
David Kabakoff, Ph.D. |
— | — | ||||
| (1) | As of December 31, 2018, the aggregate number of shares outstanding under all options to purchase our common stock held by our non-employee directors were: Ms. Anderson, 30,168 and Ms. Aspinall, 30,168. None of our other non-employee directors held outstanding options as of December 31, 2018. None of our non-employee directors held unvested stock awards (other than options) as of December 31, 2018. |
| (2) | Earned in connection with service on the board of directors and/or committees of our board of directors. The amount for Ms. Aspinall includes $26,000 earned for her service on a strategic committee of our board of directors, which service terminated following the second quarter of 2018. |
Mr. Maetzold, our President and Chief Executive Officer, is also a member of our board of directors but did not receive any additional compensation for his service as a director. See above for more information regarding the compensation earned by Mr. Maetzold.
We have entered into written agreements with each of Mss. Anderson and Aspinall and Mr. Cole in connection with their service on the board of directors.
144
The agreements with Mss. Anderson and Aspinall, who joined the board of directors in February 2015 and June 2015, respectively, provide for quarterly cash payments of $5,000 for their service on the board of directors. Pursuant to the agreements, each of Mss. Anderson and Aspinall was also granted an option to purchase 20,826 shares of the Company's common stock pursuant to the 2008 Plan, which vests as to 25% of the shares subject to the option on the first anniversary of the date they commenced service on the board of directors, and as to 1/36th of the remaining shares in equal monthly installments thereafter, subject to her continued service. The options will vest in full upon a change in control of the Company. We have also made certain other grants of stock options to Mss. Anderson and Aspinall for their service on the board.
The agreement with Mr. Cole, who joined the board of directors in December 2018, provides for an annual cash retainer of $20,000 for his service on the board of directors and an annual cash retainer of $10,000 as chair of the audit committee. Pursuant to the agreement, on March 5, 2019, Mr. Cole was also granted an option to purchase 30,168 shares of the Company's common stock pursuant to the 2018 Plan, which vests as to 25% of the shares subject to the option on the first anniversary of the date he commenced service on the board of directors, and as to 1/36th of the remaining shares in equal monthly installments thereafter, subject to his continued service. The option will vest in full upon a change in control of the Company.
We have reimbursed and will continue to reimburse all of our non-employee directors for their travel, lodging and other reasonable expenses incurred in attending meetings of our board of directors and committees of our board of directors.
Non-Employee Director Compensation Policy
Our board of directors adopted a non-employee director compensation policy in May 2019 that will become effective upon the execution and delivery of the underwriting agreement related to this offering. This compensation policy will be applicable to each member of our board of directors who is not also serving as an employee or consultant to us and will supersede any agreements we have with individual directors.
This compensation policy provides that each such non-employee director will receive the following compensation for service on our board of directors:
| • | an annual cash retainer of $36,000; |
| • | an additional annual cash retainer of $40,000 for service as chairman of the board of directors; |
| • | an additional annual cash retainer of $7,500, $5,000 and $4,000 for service as a member of the audit committee, compensation committee and the nominating and corporate governance committee, respectively; |
| • | an additional annual cash retainer of $7,500, $5,000 and $4,000 for service as chair of the audit committee, compensation committee and the nominating and corporate governance committee, respectively (in addition to the commitee member service retainer); |
| • | an initial option grant to purchase shares of our common stock on the date of each such non-employee director’s appointment to our board of directors, vesting in 36 monthly installments following the grant date; and |
| • | an annual option grant to purchase shares of our common stock on the date of each of our annual stockholder meetings, vesting one year following the grant date. |
Each of the option grants described above will be granted under the 2019 Plan, the terms of which are described in more detail below under “— Equity Benefit Plans — 2019 Equity Incentive Plan.” Each such option grant will vest and become exercisable subject to the director’s continuous service to us, provided that each option will vest in full upon a change in control (as defined in the 2019 Plan). The term of each option will be 10 years, subject to earlier termination as provided in the 2019 Plan, provided that upon a termination of service other than for death, disability or cause, the post-termination exercise period will be three months from the date of termination. An eligible director may decline all or any portion of his or her compensation by giving notice to us prior to the date cash may be paid or equity awards are to be granted, as the case may be.
145
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
The following includes a summary of transactions since January 1, 2016 to which we have been a party, in which the amount involved in the transaction exceeded $120,000, and in which any of our directors, executive officers or more than 5% of our capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest, other than equity and other compensation, termination, change in control and other arrangements, which are described under “Executive and Director Compensation.”
Series F Preferred Stock Financings
In January 2018 we completed a second Series F financing in which we sold and issued an aggregate of 940,605 shares of our Series F redeemable convertible preferred stock at a purchase price of $5.8208 per share, for aggregate gross proceeds of approximately $5.5 million, and issued warrants to purchase an aggregate of 67,233 shares of our Series F redeemable convertible preferred stock, at a purchase price of $0.01 per warrant share, for aggregate gross proceeds of approximately $672. The participants in this financing included the following executive officers and members of our board of directors, or entities affiliated with them:
Participants |
Shares of Series F Redeemable Convertible Preferred Stock |
Warrants to Purchase Shares of Series F Redeemable Convertible Preferred Stock |
Aggregate Consideration |
||||||
Sofinnova HealthQuest Partners, L.P.(1) |
167,230 | 14,107 | $ | 973,412 | |||||
MGC Venture Partners 2013, L.P.(2) |
154,988 | 13,077 | $ | 902,154 | |||||
Industry Ventures Healthcare, LLC |
140,832 | 11,880 | $ | 819,755 | |||||
BioBrit, LLC(3) |
98,145 | 8,279 | $ | 571,282 | |||||
Joseph C. Cook, Jr.(4) |
30,233 | 2,550 | $ | 175,980 | |||||
Joseph C. Cook, III(5) |
24,840 | 2,095 | $ | 144,589 | |||||
Mara G. Aspinall |
17,180 | — | $ | 100,001 | |||||
Bernhard E. Spiess |
6,872 | — | $ | 40,001 | |||||
Derek J. Maetzold |
1,000 | — | $ | 5,821 | |||||
| (1) | Sofinnova HealthQuest Partners, L.P. is affiliated with David Kabakoff, Ph.D., one of our non-employee directors. |
| (2) | MGC Venture Partners 2013, L.P. is affiliated with each of Joseph C. Cook, III, one of our non-employee directors, and Joseph C. Cook, Jr., a former non-employee director. |
| (3) | BioBrit, LLC is affiliated with Daniel M. Bradbury, one of our non-employee directors. |
| (4) | Joseph C. Cook, Jr., a former non-employee director, resigned from our board of directors in August 2018. |
| (5) | Joseph C. Cook, III, one of our non-employee directors, was elected to our board of directors in August 2018. |
In May 2018 we completed the initial closing of a third Series F financing in which we sold and issued an aggregate of 783,248 shares of our Series F redeemable convertible preferred stock at a purchase price of $5.8208 per share, for aggregate gross proceeds of approximately $4.6 million. In June 2018 we completed a second closing under this financing in which we sold and issued an aggregate of 85,711 shares of our Series F redeemable convertible preferred stock at a purchase price of $5.8208 per share, for aggregate gross proceeds of approximately $0.5 million. The participants in this financing included the following executive officers and members of our board of directors, or entities affiliated with them:
Participants |
Shares of Series F Redeemable Convertible Preferred Stock |
Aggregate Consideration |
||||
Sofinnova HealthQuest Partners, L.P.(1) |
429,494 | $ | 2,499,999 | |||
MGC Venture Partners 2013, L.P.(2) |
112,243 | $ | 653,344 | |||
Industry Ventures Healthcare, LLC |
85,227 | $ | 496,089 | |||
BioBrit, LLC(3) |
52,458 | $ | 305,348 | |||
Joseph C. Cook, Jr.(4) |
21,138 | $ | 123,040 | |||
Joseph C. Cook, III(5) |
13,784 | $ | 80,234 | |||
146
| (1) | Sofinnova HealthQuest Partners, L.P. is affiliated with David Kabakoff, Ph.D., one of our non-employee directors. |
| (2) | MGC Venture Partners 2013, L.P. is affiliated with each of Joseph C. Cook, III, one of our non-employee directors, and Joseph C. Cook, Jr., a former non-employee director. |
| (3) | BioBrit, LLC is affiliated with Daniel M. Bradbury, one of our non-employee directors. |
| (4) | Joseph C. Cook, Jr., a former non-employee director, resigned from our board of directors in August 2018. |
| (5) | Joseph C. Cook, III, one of our non-employee directors, was elected to our board of directors in August 2018. |
Convertible Promissory Note Financing
In January 2019 and February 2019, we issued and sold to investors convertible promissory notes in the aggregate principal amount of approximately $11.8 million. The convertible promissory notes carry an interest rate of 8% per annum. Upon the completion of this offering, all outstanding principal and accrued interest under the convertible promissory notes will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019. The participants in this convertible promissory note financing included the following executive officers and members of our board of directors, or entities affiliated with them:
Participants |
Aggregate Principal Amount |
||
Sofinnova HealthQuest Partners, L.P.(1) |
$ | 1,027,882 | |
MGC Venture Partners 2013, L.P.(2) |
$ | 1,200,000 | |
Industry Ventures Healthcare, LLC |
$ | 727,492 | |
BioBrit, LLC(3) |
$ | 874,537 | |
Joseph C. Cook, Jr.(4) |
$ | 600,000 | |
Joseph C. Cook, III(5) |
$ | 205,971 | |
Derek J. Maetzold |
$ | 100,000 | |
Bernhard E. Spiess |
$ | 20,000 | |
| (1) | Sofinnova HealthQuest Partners, L.P. is affiliated with David Kabakoff, Ph.D., one of our non-employee directors. |
| (2) | MGC Venture Partners 2013, L.P. is affiliated with each of Joseph C. Cook, III, one of our non-employee directors, and Joseph C. Cook, Jr., a former non-employee director. |
| (3) | BioBrit, LLC is affiliated with Daniel M. Bradbury, one of our non-employee directors. |
| (4) | Joseph C. Cook, Jr., a former non-employee director, resigned from our board of directors in August 2018. |
| (5) | Joseph C. Cook, III, one of our non-employee directors, was elected to our board of directors in August 2018. |
Investor Agreements
In connection with our preferred stock financings described above, we entered into amendments to our existing investor rights agreement, voting agreement and right of first refusal and co-sale agreement, which contain voting rights, information rights, rights of first refusal and co-sale and registration rights, among other things, with certain of our stockholders. These rights will terminate upon the closing of this offering, except for the registration rights as more fully described below in “Description of Capital Stock—Registration Rights.”
Stock Options Granted to Executive Officers and Directors
We have granted stock options to our executive officers and directors, as more fully described in the section titled “Executive and Director Compensation.”
Indemnification Agreements
We have entered, and intend to continue to enter, into separate indemnification agreements with each of our directors and executive officers, as described in “Management — Limitation of Liability and Indemnification.”
Policies and Procedures for Transactions with Related Persons
We have adopted a written related-person transactions policy that sets forth our policies and procedures regarding the identification, review, consideration and oversight of “related-person transactions.” For purposes of our policy only, a “related-person transaction” is a transaction, arrangement or relationship (or any series of similar
147
transactions, arrangements or relationships) in which we and any “related person” are participants involving an amount that exceeds $120,000. Transactions involving compensation for services provided to us as an employee, consultant or director are not considered related-person transactions under this policy. A related person is any executive officer, director, nominee to become a director or a holder of more than five percent of our common stock, including any of their immediate family members and affiliates, including entities owned or controlled by such persons.
Under the policy, where a transaction has been identified as a related-person transaction, management must present information regarding the proposed related-person transaction to our audit committee (or, where review by our audit committee would be inappropriate, to another independent body of our board of directors) for review. The presentation must include a description of, among other things, all of the parties thereto, the direct and indirect interests of the related persons, the purpose of the transaction, the material facts, the benefits of the transaction to us and whether any alternative transactions are available, an assessment of whether the terms are comparable to the terms available from unrelated third parties and management’s recommendation. To identify related-person transactions in advance, we rely on information supplied by our executive officers, directors and certain significant stockholders. In considering related-person transactions, our audit committee or another independent body of our board of directors takes into account the relevant available facts and circumstances including, but not limited to:
| • | the risks, costs and benefits to us; |
| • | the impact on a director’s independence in the event the related person is a director, immediate family member of a director or an entity with which a director is affiliated; |
| • | the terms of the transaction; |
| • | the availability of other sources for comparable services or products; and |
| • | the terms available to or from, as the case may be, unrelated third parties. |
In the event a director has an interest in the proposed transaction, the director must recuse himself or herself from the deliberations and approval.
148
The following table sets forth information regarding beneficial ownership of our capital stock by:
| • | each person, or group of affiliated persons, known by us to beneficially own more than 5% of our common stock; |
| • | each of our directors; |
| • | our named executive officer; and |
| • | all of our current executive officers and directors as a group. |
The percentage ownership information under the column entitled “Before Offering” is based on 12,596,294 shares of common stock outstanding as of June 14, 2019, assuming conversion of all outstanding shares of our convertible preferred stock into 9,959,831 shares of common stock.
The percentage ownership information under the column entitled “After Offering” is based on the sale of shares of common stock in this offering, assuming (i) no exercise of the underwriters’ option to purchase additional shares, (ii) the net exercise of certain outstanding warrants to purchase shares of our Series F redeemable convertible preferred stock for an aggregate of shares of common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) upon the completion of this offering, and (iii) the conversion of approximately $11.8 million of aggregate principal amount, plus accrued interest thereon, of convertible promissory notes which will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019, which will occur in connection with the closing of this offering.
Information with respect to beneficial ownership has been furnished by each director, officer or beneficial owner of more than 5% of our common stock. We have determined beneficial ownership in accordance with the rules of the SEC. These rules generally attribute beneficial ownership of securities to persons who possess sole or shared voting power or investment power with respect to those securities. In addition, the rules include shares of common stock issuable pursuant to the exercise of stock options that are either immediately exercisable or exercisable on or before August 13, 2019, which is 60 days after June 14, 2019. These shares are deemed to be outstanding and beneficially owned by the person holding those options for the purpose of computing the percentage ownership of that person, but they are not treated as outstanding for the purpose of computing the percentage ownership of any other person. Unless otherwise indicated, the persons or entities identified in this table have sole voting and investment power with respect to all shares shown as beneficially owned by them, subject to applicable community property laws.
Except as otherwise noted below, the address for each person or entity listed in the table is c/o Castle Biosciences, Inc., 820 S. Friendswood Drive, Suite 201, Friendswood, Texas 77546.
Number of Shares Beneficially Owned |
Percentage of Shares Beneficially Owned |
|||||||||||
Name and Address of Beneficial Owner |
Before Offering |
After Offering |
Before Offering |
After Offering |
||||||||
Greater than 5% Stockholders |
||||||||||||
Derek J. Maetzold(1) |
2,360,375 | 18.4 | % |
% |
||||||||
Sofinnova HealthQuest Partners, L.P.(2) |
2,057,226 | 16.3 | % |
% |
||||||||
MGC Venture Partners 2013, L.P.(3) |
1,629,715 | 12.9 | % |
% |
||||||||
Industry Ventures Healthcare, LLC(4) |
1,456,019 | 11.5 | % |
% |
||||||||
BioBrit, LLC(5) |
1,021,004 | 8.1 | % |
% |
||||||||
Directors and Named Executive Officers |
||||||||||||
Bonnie H. Anderson(6) |
46,180 | * |
% |
|||||||||
Mara G. Aspinall(7) |
46,180 | * |
% |
|||||||||
Daniel M. Bradbury(5) |
1,021,004 | 8.1 | % |
% |
||||||||
G. Bradley Cole |
— | * |
% |
|||||||||
Joseph C. Cook III(8) |
1,835,832 | 14.6 | % |
% |
||||||||
David Kabakoff, Ph.D.(2) |
2,057,226 | 16.3 | % |
% |
||||||||
Derek J. Maetzold(1) |
2,360,375 | 18.4 | % |
% |
||||||||
149
Number of Shares Beneficially Owned |
Percentage of Shares Beneficially Owned |
|||||||||||||
Name and Address of Beneficial Owner |
Before Offering |
After Offering |
Before Offering |
After Offering |
||||||||||
Frank Stokes(9) |
162,295 | 1.3 | % |
|||||||||||
Federico A. Monzon, M.D.(10) |
108,750 | * |
% |
|||||||||||
All current executive officers and directors as a group (10 persons)(11) |
7,827,685 | 58.6 | % |
% |
||||||||||
| * | Represents beneficial ownership of less than 1%. |
| (1) | The number of shares beneficially owned before the offering consists of (i) 1,906,375 shares of common stock held by Derek J. Maetzold, (ii) 200,000 shares of common stock held by DJM Grantor Retained Annuity Trust No. 1, or the Maetzold Trust, and (iii) 254,000 shares of common stock issuable upon the exercise of stock options. In addition to the foregoing shares, the number of shares beneficially owned after the offering includes shares of common stock issuable upon the conversion of a convertible promissory note held by Mr. Maetzold in the principal amount of $100,000.00 plus accrued interest upon the completion of this offering based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019. Mr. Maetzold is a trustee of the Maetzold Trust. |
| (2) | The number of shares beneficially owned before the offering consists of (i) 2,043,119 shares of common stock and (ii) 14,107 shares of common stock issuable upon the exercise of warrants. The number of shares beneficially owned after the offering consists of shares of common stock, which includes (i) shares of common stock issuable upon the net exercise of warrants to purchase 14,107 shares of common stock and (ii) shares of common stock issuable upon the conversion of a convertible promissory note in the principal amount of $1,027,882.00 plus accrued interest, in each case, upon the completion of this offering and based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of each such issuance on , 2019. HealthQuest Venture Management, L.L.C., or HealthQuest Management, is the managing member of Sofinnova HealthQuest Partners, L.P., or HealthQuest Partners. David Kabakoff, Ph.D., is a partner of HealthQuest Management. Garheng Kong is the managing member of HealthQuest Partners and has sole voting and investment power over the shares held by HealthQuest Partners. Each of HealthQuest Management, Mr. Kong and Dr. Kabakoff disclaims beneficial ownership over all shares held by HealthQuest Partners except to the extent of any pecuniary interest therein. The address of HealthQuest Management, HealthQuest Partners, Mr. Kong and Dr. Kabakoff is 1301 Shoreway Rodd, Suite 350, Belmont, CA 94002. |
| (3) | The number of shares beneficially owned before the offering consists of (i) 1,620,778 shares of common stock and (ii) 8,937 shares of common stock issuable upon the exercise of warrants. In addition to the foregoing shares, the number of shares beneficially owned after the offering includes shares of common stock issuable upon the conversion of a convertible promissory note in the principal amount of $1,200,000.00 plus accrued interest upon the completion of this offering based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019, or the MGC Note Shares. MGC Venture Partners 2013 GP, LLC, or MGC Partners GP, is the general partner of MGC Venture Partners 2013, L.P., or MGC Partners LP. Each of Joseph C. Cook, Jr., Joseph C. Cook III, Steven D. Singleton and Byron W. Smith are managing directors of MGC Partners GP and share voting and investment power over the shares held by MGC Partners LP. The address of MGC Partners LP, MGC Partners GP and each of the foregoing individuals is 3835 Cleghorn Avenue, Suite 300 Nashville, TN 37215. |
| (4) | Consists of (i) 1,444,139 shares of common stock and (ii) 11,880 shares of common stock issuable upon the exercise of warrants. The number of shares beneficially owned after the offering consists of shares of common stock, which includes (i) shares of common stock issuable upon the net exercise of warrants to purchase 11,880 shares of common stock and (ii) shares of common stock issuable upon the conversion of a convertible promissory note in the principal amount of $727,492.11 plus accrued interest, in each case, upon the completion of this offering and based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of each such issuance on , 2019. Industry Ventures Management VII, LLC, or Industry Management, is the sole manager of Industry Ventures Healthcare LLC, or Industry Healthcare. Johan Swildens is the sole managing member of Industry Management and has voting and investment power over the shares held by Industry Healthcare. The address of Industry Healthcare, Industry Management and Mr. Swildens is 30 Hotaling Place, San Francisco, CA 94111. |
| (5) | Daniel M. Bradbury is the managing member of BioBrit, LLC, or BioBrit, and has voting and investment power of the shares held by BioBrit. The number of shares beneficially owned after the offering includes shares of common stock issuable upon the conversion of a convertible promissory note in the principal amount of $874,537.48 plus accrued interest held by BioBrit upon the completion of this offering based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019. The address of BioBrit is 2223 Avenida de la Playa, Suite 108, La Jolla, CA 92037. |
| (6) | Consists of (i) 21,457 shares of common stock held by Bonnie H. Anderson, (ii) 17,180 shares of common stock held by the Bonnie H. Anderson Living Trust, or Anderson Trust, and (iii) 7,543 shares of common stock issuable upon the exercise of stock options. Bonnie H. Anderson is a trustee of the Anderson Trust. |
| (7) | Consists of (i) 17,180 shares of common stock and (ii) 29,000 shares of common stock issuable upon the exercise of stock options. |
| (8) | The number of shares beneficially owned before the offering consists of (i) 206,117 shares of common stock and (ii) the shares described in note 3 above. The number of shares beneficially owned after the offering consists of (i) the shares described in note 3 above and (ii) shares of common stock issuable upon the conversion of a convertible promissory note in the principal amount of $205,970.51 plus accrued interest held by Mr. Cook III upon the completion of this offering based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019. |
| (9) | Consists of 162,295 shares of common stock issuable upon the exercise of stock options. |
| (10) | Consists of 108,750 shares of common stock issuable upon the exercise of stock options. |
| (11) | Includes the shares described in notes (1), (2) and (5) through (10), and shares held or issuable upon the exercise of stock options by executive officers who are not named in the table above. |
150
Upon filing of our amended and restated certificate of incorporation and the completion of this offering, our authorized capital stock will consist of 200,000,000 shares of common stock, par value $0.001 per share, and 10,000,000 shares of preferred stock, par value $0.001 per share. All of our authorized preferred stock upon the completion of this offering will be undesignated. The following is a summary of the rights of our common and preferred stockholders and some of the provisions of our amended and restated certificate of incorporation and amended and restated bylaws, which will become effective immediately prior to and upon the completion of this offering, respectively, and of the Delaware General Corporation Law. This summary is not complete. For more detailed information, please see our amended and restated certificate of incorporation and amended and restated bylaws, which are filed as exhibits to the registration statement of which this prospectus is a part, as well as the relevant provisions of the Delaware General Corporation Law.
Common Stock
Outstanding Shares
As of March 31, 2019, after giving effect to the conversion of our outstanding shares of convertible preferred stock in connection with the completion of this offering, there were 12,296,294 shares of common stock issued and outstanding held of record by 77 stockholders. Based on the number of shares of common stock outstanding as of March 31, 2019, and assuming (1) the issuance by us of shares of common stock in this offering, (2) the net exercise of certain outstanding warrants to purchase shares of our Series F redeemable convertible preferred stock for an aggregate of shares of common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) upon the completion of this offering and (3) the conversion of approximately $11.8 million of aggregate principal amount, plus accrued interest thereon, of convertible promissory notes which will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019, there will be shares of common stock outstanding upon the completion of this offering.
As of March 31, 2019, there were 2,575,158 shares of common stock subject to outstanding options under our equity incentive plans.
Voting
Our common stock is entitled to one vote for each share held of record on all matters submitted to a vote of the stockholders, including the election of directors, and does not have cumulative voting rights. Accordingly, the holders of a majority of the shares of our common stock entitled to vote in any election of directors can elect all of the directors standing for election.
Dividends
Subject to preferences that may be applicable to any then-outstanding preferred stock, the holders of common stock are entitled to receive dividends, if any, as may be declared from time to time by our board of directors out of legally available funds.
Liquidation
In the event of our liquidation, dissolution or winding-up, holders of our common stock will be entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of all of our debts and other liabilities, subject to the satisfaction of any liquidation preference granted to the holders of any outstanding shares of preferred stock.
Rights and Preferences
Holders of our common stock have no preemptive, conversion or subscription rights, and there are no redemption or sinking fund provisions applicable to our common stock. The rights, preferences and privileges of the holders of our common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of our preferred stock that we may designate and issue in the future.
151
Fully Paid and Nonassessable
All of our outstanding shares of common stock are, and the shares of common stock to be issued in this offering will be, fully paid and nonassessable.
Convertible Preferred Stock
As of March 31, 2019, there were 9,959,831 shares of convertible preferred stock outstanding, held of record by 69 stockholders.
In connection with the closing of this offering, all outstanding shares of convertible preferred stock will be converted into shares of our common stock. Immediately prior to the closing of this offering, our certificate of incorporation will be amended and restated and all of our previously outstanding shares of convertible preferred stock will be converted into shares of common stock. Under the amended and restated certificate of incorporation, our board of directors will have the authority, without further action by the stockholders, to issue up to shares of convertible preferred stock in one or more series, to establish from time to time the number of shares to be included in each such series, to fix the rights, preferences and privileges of the shares of each wholly unissued series and any qualifications, limitations or restrictions thereon and to increase or decrease the number of shares of any such series, but not below the number of shares of such series then outstanding.
Our board of directors may authorize the issuance of preferred stock with voting or conversion rights that could adversely affect the voting power or other rights of the holders of the common stock. The issuance of preferred stock, while providing flexibility in connection with possible acquisitions and other corporate purposes, could, among other things, have the effect of delaying, deferring or preventing a change in our control that may otherwise benefit holders of our common stock and may adversely affect the market price of the common stock and the voting and other rights of the holders of common stock. We have no current plans to issue any shares of preferred stock.
Warrants
As of March 31, 2019, the following shares of our redeemable convertible preferred stock were issuable upon the exercise of outstanding warrants:
| • | Warrants to purchase an aggregate of 12,999 shares of our Series E-1 redeemable convertible preferred stock for an exercise price of $3.77 per share, which we issued to investors in connection with a financing transaction. |
| • | A warrant to purchase 15,292 shares of our Series E-2 redeemable convertible preferred stock for an exercise price of $4.58 per share, which we issued to SVB. |
| • | A warrant to purchase 6,554 shares of our Series E-3 redeemable convertible preferred stock for an exercise price of $5.34 per share, which we issued to SVB. |
| • | Warrants to purchase an aggregate of 103,090 shares of our Series F redeemable convertible preferred stock for an exercise price of $5.82 per share, which we issued to SVB and Oxford. |
| • | Warrants to purchase an aggregate of 34,282 shares of our Series F redeemable convertible preferred stock for an exercise price of $0.01 per share, which we issued to investors in connection with a financing transaction. |
Each of the foregoing warrants provide for the adjustment of the number of shares issuable upon the exercise thereof in the event of stock splits, recapitalizations, reclassifications and consolidations.
Stock Options
As of March 31, 2019, 2,575,158 shares of common stock were issuable upon the exercise of outstanding stock options, at a weighted-average exercise price of $1.88 per share. For information regarding the terms of our equity incentive plans, see “Executive and Director Compensation—Equity Benefit Plans.”
Registration Rights
After the closing of this offering, certain holders of shares of our common stock, including all of the current preferred stockholders, including certain holders of five percent of our capital stock and entities affiliated with
152
certain of our directors, will be entitled to certain rights with respect to registration of such shares under the Securities Act. These shares are referred to as registrable securities. The holders of these registrable securities possess registration rights pursuant to the terms of the investors’ rights agreement and are described in additional detail below.
The registration of shares of our common stock pursuant to the exercise of the registration rights described below would enable the holders to trade these shares without restriction under the Securities Act when the applicable registration statement is declared effective. We are required to pay all registration expenses, other than underwriting discounts, selling commissions and stock transfer taxes of the shares registered pursuant to the demand, piggyback and Form S-3 registrations described below.
Generally, in an underwritten offering, the managing underwriter, if any, has the right, subject to specified conditions, to limit the number of shares the holders may include. The demand, piggyback and Form S-3 registration rights described below will expire upon the earliest to occur of (1) five years after the closing of this offering and (2) the consummation of a “Liquidation Event,” as such term is defined in our seventh amended and restated certificate of incorporation, as amended.
Demand Registration Rights
The holders of the registrable securities will be entitled to certain demand registration rights. Subject to the terms of the lockup agreements described under “Underwriters,” at any time beginning 180 days after the date of this prospectus, the holders of at least 50% of the registrable securities then outstanding may make a written request that we register all or a portion of their shares, subject to certain specified exceptions. Such request for registration must cover securities that have an aggregate offering price that exceeds $10,000,000. We will not be required to effect more than two registrations pursuant to these demand registration rights.
Piggyback Registration Rights
In connection with this offering, the holders of registrable securities were entitled to, and the necessary percentage of holders waived, their rights to notice of this offering and to include their shares of registrable securities in this offering. If we propose to register for offer and sale any of our securities under the Securities Act in another offering, either for our own account or for the account of other security holders, the holders of registrable securities will be entitled to certain “piggyback” registration rights allowing them to include their shares in such registration, subject to certain marketing and other limitations. As a result, whenever we propose to file a registration statement under the Securities Act, including a registration statement on Form S-3 as discussed below, other than with respect to a demand registration or a registration statement on Forms S-4 or S-8, the holders of these shares are entitled to notice of the registration and have the right, subject to limitations that the underwriters may impose on the number of shares included in the registration, to include their shares in the registration.
Form S-3 Registration Rights
The holders of the registrable securities will be entitled to certain Form S-3 registration rights. Holders of at least 10% of the registrable securities may request that we register for offer and sale their shares on Form S-3 if we are qualified to file a registration statement on Form S-3, subject to certain specified exceptions. Such request for registration on Form S-3 must cover securities the aggregate offering price of which exceeds $1,000,000. We will not be required to effect more than one registration on Form S-3 within any six-month period.
153
Anti-Takeover Effects of Provisions of Our Amended and Restated Certificate of Incorporation, Our Amended and Restated Bylaws and Delaware Law
Delaware Anti-Takeover Law
We are subject to Section 203 of the Delaware General Corporation Law, or Section 203. Section 203 generally prohibits a public Delaware corporation from engaging in a “business combination” with an “interested stockholder” for a period of three years following the time that such stockholder became an interested stockholder, unless:
| • | prior to such time the board of directors of the corporation approved either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder; |
| • | upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding (but not the outstanding voting stock owned by the interested stockholder) those shares owned (1) by persons who are directors and also officers and (2) employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or |
| • | at or subsequent to such time, the business combination is approved by the board of directors and authorized at an annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66-2/3% of the outstanding voting stock which is not owned by the interested stockholder. |
Section 203 defines a business combination to include:
| • | any merger or consolidation involving the corporation and the interested stockholder; |
| • | any sale, transfer, pledge or other disposition involving the interested stockholder of 10% or more of the assets of the corporation; |
| • | subject to exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder; |
| • | subject to exceptions, any transaction involving the corporation that has the effect of increasing the proportionate share of the stock of any class or series of the corporation beneficially owned by the interested stockholder; and |
| • | the receipt by the interested stockholder of the benefit of any loans, advances, guarantees, pledges or other financial benefits provided by or through the corporation. |
In general, Section 203 defines an interested stockholder as any entity or person beneficially owning 15% or more of the outstanding voting stock of the corporation and any entity or person affiliated with or controlling or controlled by the entity or person.
Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws
Provisions of our amended and restated certificate of incorporation and amended and restated bylaws, which will become effective immediately prior to and upon the completion of this offering, respectively, may delay or discourage transactions involving an actual or potential change in our control or change in our management, including transactions in which stockholders might otherwise receive a premium for their shares or transactions that our stockholders might otherwise deem to be in their best interests. Therefore, these provisions could adversely affect the price of our common stock. Among other things, our amended and restated certificate of incorporation and amended and restated bylaws:
| • | permit our board of directors to issue up to 10,000,000 shares of preferred stock, with any rights, preferences and privileges as they may designate (including the right to approve an acquisition or other change in our control); |
| • | provide that the authorized number of directors may be changed only by resolution of the board of directors; |
154
| • | provide that the board of directors or any individual director may only be removed with cause and the affirmative vote of the holders of at least 662∕3% of the voting power of all of our then outstanding common stock; |
| • | provide that all vacancies, including newly created directorships, may, except as otherwise required by law, be filled by the affirmative vote of a majority of directors then in office, even if less than a quorum; |
| • | divide our board of directors into three classes; |
| • | require that any action to be taken by our stockholders must be effected at a duly called annual or special meeting of stockholders and not be taken by written consent; |
| • | provide that stockholders seeking to present proposals before a meeting of stockholders or to nominate candidates for election as directors at a meeting of stockholders must provide notice in writing in a timely manner and also specify requirements as to the form and content of a stockholder’s notice; |
| • | do not provide for cumulative voting rights (therefore allowing the holders of a majority of the shares of common stock entitled to vote in any election of directors to elect all of the directors standing for election, if they should so choose); |
| • | provide that special meetings of our stockholders may be called only by the chairman of the board, our Chief Executive Officer or by the board of directors pursuant to a resolution adopted by a majority of the total number of authorized directors; |
| • | provide that the Court of Chancery of the State of Delaware will be the sole and exclusive forum for the following types of actions or proceedings under Delaware statutory or common law: (i) any derivative action or proceeding brought on our behalf, (ii) any action or proceeding asserting a claim of breach of a fiduciary duty owed by any of our current or former directors, officers or other employees to us or our stockholders; (iii) any action or proceeding asserting a claim against us or any of our current or former directors, officers or other employees, arising out of or pursuant to any provision of the Delaware General Corporation Law, our certificate of incorporation or our bylaws; (iv) any action or proceeding to interpret, apply, enforce or determine the validity of our certificate of incorporation or our bylaws; (v) any action or proceeding as to which the Delaware General Corporation Law confers jurisdiction to the Court of Chancery of the State of Delaware; and (vi) any action asserting a claim against us or any of our directors, officers or other employees governed by the internal affairs doctrine, in all cases to the fullest extent permitted by law and subject to the court’s having personal jurisdiction over the indispensable parties named as defendants; provided these provisions of our amended and restated certificate of incorporation and amended and restated bylaws will not apply to suits brought to enforce a duty or liability created by the Exchange Act, or any other claim for which the federal courts have exclusive jurisdiction; and |
| • | provide that unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States of America shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act, subject to and contingent upon a final adjudication in the State of Delaware of the enforceability of such exclusive forum provision. |
The amendment of any of these provisions, with the exception of the ability of our board of directors to issue shares of preferred stock and designate any rights, preferences and privileges thereto, would require approval by the holders of at least 662∕3% of our then-outstanding common stock.
Exchange Listing
We have applied for listing of our common stock on The Nasdaq Global Select Market under the symbol “CSTL.”
Transfer Agent and Registrar
The transfer agent and registrar for our common stock is Broadridge Corporate Issuer Solutions, Inc.
155
SHARES ELIGIBLE FOR FUTURE SALE
Immediately prior to this offering, there has been no public market for our common stock. Future sales of substantial amounts of common stock in the public market could adversely affect prevailing market prices. Furthermore, since only a limited number of shares will be available for sale shortly after this offering because of contractual and legal restrictions on resale described below, sales of substantial amounts of common stock in the public market after the restrictions lapse could adversely affect the prevailing market price for our common stock as well as our ability to raise equity capital in the future.
Based on the number of shares of common stock outstanding as of March 31, 2019, upon the completion of this offering and assuming (1) the 1-for- reverse stock split of all outstanding shares of our capital stock, (2) the conversion of all of our outstanding shares of convertible preferred stock as of March 31, 2019 into an aggregate of 9,959,831 shares of common stock, (3) the net exercise of certain outstanding warrants to purchase shares of our Series F redeemable convertible preferred stock for an aggregate of shares of common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus) upon completion of this offering, (4) the conversion of approximately $11.8 million of aggregate principal amount, plus accrued interest thereon, of convertible promissory notes which will automatically convert upon the completion of this offering into an aggregate of shares of our common stock, based on an assumed initial public offering price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus), and assuming the occurrence of the conversion on , 2019, there will be shares of common stock outstanding upon the completion of this offering, (5) no exercise of the underwriters’ option to purchase additional shares of common stock, and (6) no exercise of outstanding options, an aggregate of shares of common stock will be outstanding. All of the shares sold in this offering will be freely tradable in the public market without restriction or further registration under the Securities Act, unless held by an affiliate of ours. Except as set forth below, the remaining shares of common stock outstanding after this offering will be restricted as a result of securities laws or lock-up agreements. In addition, any shares sold in this offering to entities affiliated with our existing stockholders and directors will be subject to lock-up agreements. These remaining shares will generally become available for sale in the public market as follows:
| • | no restricted shares will be eligible for immediate sale upon the completion of this offering; |
| • | up to restricted shares will be eligible for sale under Rule 144 or Rule 701 upon expiration of lock-up agreements 180 days after the date of this prospectus; and |
| • | the remainder of the restricted shares will be eligible for sale from time to time thereafter upon expiration of their respective holding periods under Rule 144, as described below, but could be sold earlier if the holders exercise any available registration rights. |
Rule 144
In general, under Rule 144 as currently in effect, beginning 90 days after the effective date of the registration statement of which this prospectus is a part, any person who is not an affiliate of ours and has held their shares for at least six months, including the holding period of any prior owner other than one of our affiliates, may sell shares without restriction, provided current public information about us is available. In addition, under Rule 144, any person who is not an affiliate of ours and has held their shares for at least one year, including the holding period of any prior owner other than one of our affiliates, would be entitled to sell an unlimited number of shares immediately upon the completion of this offering without regard to whether current public information about us is available. Beginning 90 days after the effective date of the registration statement of which this prospectus is a part, a person who is an affiliate of ours and who has beneficially owned restricted securities for at least six months, including the holding period of any prior owner other than one of our affiliates, is entitled to sell a number of restricted shares within any three-month period that does not exceed the greater of:
| • | 1% of the number of shares of our common stock then outstanding, which will equal approximately shares immediately after this offering; or |
| • | the average weekly trading volume of our common stock on Nasdaq during the four calendar weeks preceding the filing of a notice on Form 144 with respect to the sale. |
156
Sales of restricted shares under Rule 144 held by our affiliates are also subject to requirements regarding the manner of sale, notice and the availability of current public information about us. Rule 144 also provides that affiliates relying on Rule 144 to sell shares of our common stock that are not restricted shares must nonetheless comply with the same restrictions applicable to restricted shares, other than the holding period requirement.
Notwithstanding the availability of Rule 144, the holders of substantially all of our restricted shares have entered into lock-up agreements as described below and their restricted shares will become eligible for sale at the expiration of the restrictions set forth in those agreements.
Rule 701
Under Rule 701, shares of our common stock acquired upon the exercise of currently outstanding options or pursuant to other rights granted under our stock plans may be resold by:
| • | persons other than affiliates, beginning 90 days after the effective date of the registration statement of which this prospectus is a part, subject only to the manner-of-sale provisions of Rule 144; and |
| • | our affiliates, beginning 90 days after the effective date of the registration statement of which this prospectus is a part, subject to the manner-of-sale and volume limitations, current public information and filing requirements of Rule 144, in each case, without compliance with the six-month holding period requirement of Rule 144. |
As of March 31, 2019, options to purchase a total of 2,575,158 shares of common stock were outstanding, of which 1,219,548 were vested. Of the total number of shares of our common stock issuable under these options, substantially all are subject to contractual lock-up agreements with us or the underwriters described below under “Underwriting” and will become eligible for sale at the expiration of the restrictions set forth in those agreements.
Lock-Up Agreements
We, along with our directors, executive officers and substantially all of our other stockholders and optionholders, have agreed that, for a period of 180 days after the date of this prospectus, except with the prior written consent of each of SVB Leerink LLC and Robert W. Baird & Co. Incorporated, and subject to specified exceptions, we or they will not offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant for the sale of, or otherwise dispose of or transfer, any shares of common stock or any securities convertible into or exercisable or exchangeable for shares of common stock, or enter into any swap or any other agreement or any transaction that transfers to another, in whole or in part, directly or indirectly, any of the economic consequences of ownership of the common stock. Each of SVB Leerink LLC and Robert W. Baird & Co. Incorporated has advised us that it has no current intent or arrangement to release any of the shares subject to the lock-up agreements prior to the expiration of the lock-up agreements.
After this offering, certain of our employees, including our executive officers and/or directors, may enter into written trading plans that are intended to comply with Rule 10b5-1 under the Exchange Act. Sales under these trading plans would not be permitted until the expiration of the lock-up agreements relating to the offering described above.
Registration Rights
Upon the closing of this offering, the holders of an aggregate of shares of our common stock will have rights, subject to certain conditions, to require us to file registration statements covering their shares or to include their shares in registration statements that we may file for ourselves or other stockholders. After registration pursuant to these rights, these shares will become freely tradable without restriction under the Securities Act. See “Description of Capital Stock — Registration Rights” for additional information regarding these registration rights.
Equity Incentive Plans
We intend to file with the SEC a registration statement on Form S-8 under the Securities Act covering the shares of common stock reserved for issuance under the 2008 Plan, the 2018 Plan, the 2019 Plan and the ESPP. The registration statement is expected to be filed and become effective as soon as practicable after the completion of this offering. Accordingly, shares registered under the registration statement will be available for sale in the open market following its effective date, subject to Rule 144 volume limitations and the lock-up agreements described above, if applicable.
157
MATERIAL U.S. FEDERAL INCOME TAX CONSEQUENCES
TO NON-U.S. HOLDERS OF OUR COMMON STOCK
The following summary describes the material U.S. federal income tax consequences of the acquisition, ownership and disposition of our common stock acquired in this offering by Non-U.S. Holders (as defined below). This discussion does not address all aspects of U.S. federal income taxes that may be relevant to Non-U.S. Holders in light of their particular circumstances, including but not limited to alternative minimum tax consequences and the potential application of the Medicare contribution tax on net investment income. Nor does it address any state, local or non-U.S. tax consequences, or any U.S. federal tax consequences other than income taxes (such as U.S. federal estate or gift tax consequences). Rules different from those described below may apply to certain Non-U.S. Holders that are subject to special treatment under the Code, such as financial institutions, insurance companies, tax-exempt organizations, tax-qualified retirement plans, broker-dealers and traders in securities, commodities or currencies, government organizations, certain former citizens or long-term residents of the United States, “controlled foreign corporations,” “passive foreign investment companies,” corporations that accumulate earnings to avoid U.S. federal income tax, persons that hold our common stock as part of a “straddle,” “hedge,” “conversion transaction,” “synthetic security,” integrated investment or other risk reduction strategy, holders deemed to sell our common stock under the constructive sale provisions of the Code, persons who have a functional currency other than the U.S. dollar, and accrual method taxpayers subject to special tax accounting rules under Section 451(b) of the Code. Such Non-U.S. Holders are urged to consult their tax advisors to determine the U.S. federal, state, local and other tax consequences that may be relevant to them. Furthermore, the discussion below is based upon the provisions of the Code, and U.S. Treasury regulations, published administrative pronouncements, rulings and judicial decisions thereunder as of the date hereof. Such authorities may be repealed, revoked or modified, perhaps retroactively, so as to result in U.S. federal income tax consequences different from those discussed below. We have not requested a ruling from the U.S. Internal Revenue Service, or IRS, with respect to the statements made and the conclusions reached in the following summary, and there can be no assurance that the IRS will agree with such statements and conclusions. This discussion assumes that the Non-U.S. Holder holds our common stock as a “capital asset” within the meaning of Section 1221 of the Code (generally, property held for investment).
The following discussion is for general information only and is not tax advice for any Non-U.S. Holders under their particular circumstances. Persons considering the purchase of our common stock pursuant to this offering should consult their tax advisors concerning the U.S. federal income tax and other tax consequences of acquiring, owning and disposing of our common stock in light of their particular situations as well as any consequences arising under the laws of any other taxing jurisdiction, including any state, local and non-U.S. tax consequences and any U.S. federal non-income tax consequences.
For the purposes of this discussion, a “Non-U.S. Holder” is, for U.S. federal income tax purposes, a beneficial owner of common stock that is not a U.S. Holder. A “U.S. Holder” means a beneficial owner of our common stock that is for U.S. federal income tax purposes (a) an individual who is a citizen or resident of the United States, (b) a corporation or other entity treated as a corporation that is created or organized in or under the laws of the United States, any state thereof or the District of Columbia, (c) an estate the income of which is subject to U.S. federal income taxation regardless of its source or (d) a trust if it (1) is subject to the primary supervision of a court within the United States and one or more U.S. persons have the authority to control all substantial decisions of the trust or (2) has a valid election in effect under applicable U.S. Treasury regulations to be treated as a U.S. person. Also, partnerships and their partners, or other entities that are treated as partnerships for U.S. federal income tax purposes and their equity holders (regardless of their place of organization or formation) and entities that are treated as disregarded entities for U.S. federal income tax purposes (regardless of their place of organization or formation) are not addressed by this discussion and are, therefore, not considered to be Non-U.S. Holders for the purposes of this discussion.
Distributions on Our Common Stock
Subject to the discussion below regarding backup withholding and foreign accounts, distributions, if any, made on our common stock to a Non-U.S. Holder generally will constitute dividends for U.S. tax purposes to the extent made out of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles) and will be subject to withholding tax at a 30% rate or such lower rate as may be specified by an applicable income tax treaty. To obtain a reduced rate of withholding under a treaty, a Non-U.S. Holder generally will be required to provide us with a properly executed IRS Form W-8BEN (in the case of individuals) or
158
IRS Form W-8BEN-E (in the case of entities), or other appropriate form, certifying the Non-U.S. Holder’s entitlement to benefits under that treaty. This certification must be provided to us or our paying agent prior to the payment of dividends and must be updated periodically. In the case of a Non-U.S. Holder that is an entity, U.S. Treasury regulations and the relevant tax treaty provide rules to determine whether, for purposes of determining the applicability of a tax treaty, dividends will be treated as paid to the entity or to those holding an interest in that entity. If a Non-U.S. Holder holds stock through a financial institution or other agent acting on the holder’s behalf, the holder will be required to provide appropriate documentation to such agent. The holder’s agent will then be required to provide such certification to us or our paying agent, either directly or through other intermediaries. If you are eligible for a reduced rate of U.S. federal withholding tax under an income tax treaty, you should consult with your tax advisor to determine if you are able to obtain a refund or credit if any excess amount is withheld by timely filing an appropriate claim for a refund with the IRS.
We generally are not required to withhold tax on dividends paid to a Non-U.S. Holder that are effectively connected with the Non-U.S. Holder’s conduct of a trade or business within the United States (and, if required by an applicable income tax treaty, are attributable to a permanent establishment that such holder maintains in the United States) if a properly executed IRS Form W-8ECI, certifying that the dividends are so connected, is furnished to us (or, if stock is held through a financial institution or other agent, to such agent). In general, such effectively connected dividends will be subject to U.S. federal income tax, on a net-income basis at the regular graduated rates applicable to U.S. residents, unless a specific treaty exemption applies. A corporate Non-U.S. Holder receiving effectively connected dividends may also be subject to an additional “branch profits tax,” which is imposed, under certain circumstances, at a rate of 30% (or such lower rate as may be specified by an applicable treaty) on the corporate Non-U.S. Holder’s effectively connected earnings and profits, subject to certain adjustments. Non-U.S. Holders should consult their tax advisors regarding any applicable income tax treaties that may provide for different rules.
To the extent distributions on our common stock, if any, exceed our current and accumulated earnings and profits, they will first reduce the Non-U.S. Holder’s adjusted basis in our common stock as a non-taxable return of capital, but not below zero, and then any excess will be treated as gain and taxed in the same manner as gain realized from a sale or other disposition of common stock as described in the next section.
Gain on Disposition of Our Common Stock
Subject to the discussion below regarding backup withholding and foreign accounts, a Non-U.S. Holder generally should not be subject to U.S. federal income tax with respect to gain realized on a sale or other disposition of our common stock unless (a) the gain is effectively connected with a trade or business of such holder in the United States (and, if required by an applicable income tax treaty, is attributable to a permanent establishment that such holder maintains in the United States), (b) the Non-U.S. Holder is a nonresident alien individual and is present in the United States for 183 or more days in the taxable year of the disposition and certain other conditions are met, or (c) we are or have been a “United States real property holding corporation,” or a USRPHC, within the meaning of Code Section 897(c)(2) at any time within the shorter of the five-year period preceding such disposition or such holder’s holding period.
If you are a Non-U.S. Holder described in clause (a) above, you will be required to pay tax on the net gain derived from the sale at regular graduated U.S. federal income tax rates, unless a specific treaty exemption applies, and corporate Non-U.S. Holders described in clause (a) above may be subject to the additional branch profits tax at a 30% rate or such lower rate as may be specified by an applicable income tax treaty. If you are an individual Non-U.S. Holder described in clause (b) above, you will be required to pay a flat 30% tax on the gain derived from the sale, which gain may be offset by U.S. source capital losses (even though you are not considered a resident of the United States), provided you have timely filed U.S. federal income tax returns with respect to such losses.
With respect to clause (c) above, whether we are a USRPHC depends on the fair market value of our U.S. real property interests relative to the fair market value of our worldwide real property interests plus our trade and business assets. We believe that we are not, and do not anticipate becoming, a USRPHC. However, there can be no assurance that we will not become a USRPHC in the future. Even if we are treated as a USRPHC, gain realized by a Non-U.S. Holder on a disposition of our common stock will not be subject to U.S. federal income tax so long as (1) the Non-U.S. Holder owned, directly, indirectly and constructively, no more than 5% of our common stock at all times within the shorter of (a) the five-year period preceding the disposition or (b) the
159
holder’s holding period and (2) our common stock is regularly traded on an established securities market within the meaning of applicable U.S. Treasury regulations. There can be no assurance that our common stock will continue to qualify as regularly traded on an established securities market. If any gain on your disposition of our common stock is taxable because we are a USRPHC and either your ownership of our common stock exceeds 5% or our common stock is not regularly traded on an established securities market, you will be taxed on such disposition generally in the manner applicable to U.S. persons, and in addition a purchaser of your common stock may be required to withhold tax with respect to that obligation.
Information Reporting Requirements and Backup Withholding
Generally, we or certain financial middlemen must report information to the IRS with respect to any dividends we pay on our common stock (even if payments are not subject to withholding) including the amount of any such dividends, the name and address of the recipient, and the amount, if any, of tax withheld. A similar report is sent to the holder to whom any such dividends are paid. Pursuant to tax treaties or certain other agreements, the IRS may make its reports available to tax authorities in the recipient’s country of residence.
Dividends paid by us (or our paying agents) to a Non-U.S. Holder may also be subject to U.S. backup withholding. U.S. backup withholding generally will not apply to a Non-U.S. Holder who provides a properly executed IRS Form W-8BEN (in the case of individuals), IRS Form W-8BEN-E (in the case of entities) or IRS Form W-8ECI, or otherwise establishes an exemption. Notwithstanding the foregoing, backup withholding may apply if the payor has actual knowledge, or reason to know, that the holder is a U.S. person who is not an exempt recipient.
Under current U.S. federal income tax law, U.S. information reporting and backup withholding requirements generally will apply to the proceeds of a disposition of our common stock effected within the United States or through certain U.S.-related brokers, unless the holder provides a properly executed IRS Form W-8BEN (in the case of individuals), IRS Form W-8BEN-E (in the case of entities) or IRS Form W-8ECI, or otherwise establishes an exemption. Generally, U.S. information reporting and backup withholding requirements will not apply to a payment of disposition proceeds to a Non-U.S. Holder where the transaction is effected outside the United States through a non-U.S. office of a non-U.S. broker. Information reporting and backup withholding requirements may, however, apply to a payment of disposition proceeds if the broker has actual knowledge, or reason to know, that the holder is, in fact, a U.S. person. For information reporting purposes, certain brokers with substantial U.S. ownership or operations will generally be treated in a manner similar to U.S. brokers.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or credit against a Non-U.S. Holder’s U.S. federal income tax liability, provided the required information is timely furnished to the IRS. If backup withholding is applied to you, you should consult with your tax advisor to determine if you are able to obtain a tax refund or credit with respect to the amount withheld.
Foreign Accounts
A U.S. federal withholding tax of 30% may apply to dividends and, subject to the discussion of certain proposed U.S. Treasury regulations below, the gross proceeds of a disposition of our common stock paid to a foreign financial institution (as specifically defined by applicable rules), including when the foreign financial institution holds our common stock on behalf of a Non-U.S. Holder, unless such institution enters into an agreement with the U.S. government to withhold on certain payments and to collect and provide to the U.S. tax authorities substantial information regarding U.S. account holders of such institution (which may include certain equity holders of such institution, as well as certain account holders that are foreign entities with U.S. owners). Foreign financial institutions located in jurisdictions that have an intergovernmental agreement with the United States governing these withholding and reporting requirements may be subject to different rules. This U.S. federal withholding tax of 30% will also apply to dividends on and, subject to the discussion of certain proposed U.S. Treasury regulations below, the gross proceeds of a disposition of our common stock paid to a non-financial foreign entity unless such entity provides the withholding agent with either a certification that it does not have any substantial direct or indirect U.S. owners or provides information regarding direct and indirect U.S. owners of the entity. The withholding tax described above will not apply if the foreign financial institution or non-financial foreign entity otherwise qualifies for an exemption from these rules. Under certain circumstances, a Non-U.S. Holder might be eligible for refunds or credits of such taxes.
160
The U.S. Treasury recently released proposed regulations which, if finalized in their present form, would eliminate the federal withholding tax of 30% applicable to the gross proceeds of a sale or other disposition of our common stock. In its preamble to such proposed regulations, the U.S. Treasury stated that taxpayers may generally rely on the proposed regulations until final regulations are issued.
Prospective investors should consult their tax advisors regarding the possible impact of these rules on their investment in our common stock, and the possible impact of these rules on the entities through which they hold our common stock, including, without limitation, the process and deadlines for meeting the applicable requirements to prevent the imposition of this 30% withholding tax.
EACH PROSPECTIVE INVESTOR SHOULD CONSULT ITS TAX ADVISOR REGARDING THE TAX CONSEQUENCES OF PURCHASING, HOLDING AND DISPOSING OF OUR COMMON STOCK, INCLUDING THE CONSEQUENCES OF ANY RECENT AND PROPOSED CHANGE IN APPLICABLE LAW, AS WELL AS TAX CONSEQUENCES ARISING UNDER ANY STATE, LOCAL, NON-U.S. OR U.S. FEDERAL NON-INCOME TAX LAWS.
161
SVB Leerink LLC and Robert W. Baird & Co. Incorporated are acting as representatives of each of the underwriters named below and as joint book-running managers for this offering. Subject to the terms and conditions set forth in an underwriting agreement among us and the underwriters, we have agreed to sell to the underwriters, and each of the underwriters has agreed, severally and not jointly, to purchase from us, the number of shares of common stock set forth opposite its name below.
Underwriter |
Number of Shares |
||
SVB Leerink LLC |
|||
Robert W. Baird & Co. Incorporated |
|||
BTIG, LLC |
|||
Canaccord Genuity LLC |
|||
Total |
|||
Subject to the terms and conditions set forth in the underwriting agreement, the underwriters have agreed, severally and not jointly, to purchase all of the shares sold under the underwriting agreement if any of these shares are purchased. If an underwriter defaults, the underwriting agreement provides that the purchase commitments of the nondefaulting underwriters may be increased or the underwriting agreement may be terminated.
We have agreed to indemnify the underwriters against certain liabilities, including liabilities under the Securities Act, or to contribute to payments the underwriters may be required to make in respect of those liabilities.
The underwriters are offering the shares, subject to prior sale, when, as and if issued to and accepted by them, subject to approval of legal matters by their counsel, including the validity of the shares, and other conditions contained in the underwriting agreement, such as the receipt by the underwriters of officers’ certificates and legal opinions. The underwriters reserve the right to withdraw, cancel or modify offers to the public and to reject orders in whole or in part.
Commissions and Discounts
The representatives have advised us that the underwriters propose initially to offer the shares to the public at the public offering price set forth on the cover page of this prospectus and to dealers at that price less a concession not in excess of $ per share. After the initial offering of the shares, the public offering price, concession or any other term of the offering may be changed.
The following table shows the public offering price, underwriting discount and proceeds, before expenses, to us. The information assumes either no exercise or full exercise by the underwriters of their option to purchase additional shares.
|
|
|
Total
|
|
|
|
Per
Share |
Without
Option |
With
Option |
|
Public offering price
|
$
|
$
|
$
|
|
Underwriting discount
|
$
|
$
|
$
|
|
Proceeds, before expenses, to us
|
$
|
$
|
$
|
The expenses of the offering, not including the underwriting discount, are estimated at $ and are payable by us. We have also agreed to reimburse the underwriters for certain of their expenses, in an amount of up to $ , as set forth in the underwriting agreement.
Option to Purchase Additional Shares
We have granted an option to the underwriters, exercisable for 30 days after the date of this prospectus, to purchase up to additional shares at the public offering price, less the underwriting discount. If the underwriters exercise this option, each will be obligated, subject to conditions contained in the underwriting agreement, to purchase a number of additional shares proportionate to that underwriter’s initial amount reflected in the above table.
162
No Sales of Similar Securities
We, our executive officers and directors and our other existing security holders have agreed not to sell or transfer any shares of common stock or securities convertible into or exchangeable or exercisable for common stock, for 180 days after the date of this prospectus without first obtaining the written consent of SVB Leerink LLC and Robert W. Baird & Co. Incorporated on behalf of the underwriters. Specifically, we and these other persons have agreed, with certain limited exceptions, not to directly or indirectly:
| • | offer, pledge, sell or contract to sell any common stock; |
| • | sell any option or contract to purchase any common stock; |
| • | purchase any option or contract to sell any common stock; |
| • | grant any option, right or warrant for the sale of any common stock; |
| • | otherwise dispose of or transfer any common stock; |
| • | exercise any right with respect to the registration of any common stock, or file or cause to be filed any registration statement in connection therewith; or |
| • | enter into any swap or other agreement or any transaction that transfers, in whole or in part, directly or indirectly, the economic consequence of ownership of any common stock, whether any such swap, or transaction is to be settled by delivery of shares or other securities, in cash or otherwise. |
The lock-up provisions apply to common stock and to securities convertible into or exchangeable or exercisable for common stock. They also apply to common stock owned now or acquired later by the person executing the lock-up agreement or for which the person executing the lock-up agreement later acquires the power of disposition.
Nasdaq Global Select Market Listing
We have applied to list the shares of our common stock on the Nasdaq Global Select Market under the symbol “CSTL.”
Determination of Offering Price
Before this offering, there has been no public market for our common stock. The initial public offering price will be determined through negotiations between us and the representatives. In addition to prevailing market conditions, the factors to be considered in determining the initial public offering price are:
| • | the valuation multiples of publicly traded companies that the representatives believe to be comparable to us; |
| • | our financial information; |
| • | the history of, and the prospects for, our company and the industry in which we compete; |
| • | an assessment of our management, its past and present operations, and the prospects for, and timing of, our future revenues; |
| • | the present state of our development; and |
| • | the above factors in relation to market values and various valuation measures of other companies engaged in activities similar to ours. |
An active trading market for the shares may not develop. It is also possible that after this offering, our shares of common stock will not trade in the public market at or above the initial public offering price.
The underwriters do not expect to sell more than 5% of the shares in the aggregate to accounts over which they exercise discretionary authority.
Price Stabilization, Short Positions and Penalty Bids
Until the distribution of the shares is completed, SEC rules may limit underwriters and selling group members from bidding for and purchasing our common stock. However, the representatives may engage in transactions that stabilize the price of the common stock, such as bids or purchases to peg, fix or maintain that price.
163
In connection with the offering, the underwriters may purchase and sell our common stock in the open market. These transactions may include short sales, purchases on the open market to cover positions created by short sales and stabilizing transactions. Short sales involve the sale by the underwriters of a greater number of shares than they are required to purchase in the offering. “Covered” short sales are sales made in an amount not greater than the underwriters’ option to purchase additional shares described above. The underwriters may close out any covered short position by either exercising their option to purchase additional shares or purchasing shares in the open market. In determining the source of shares to close out the covered short position, the underwriters will consider, among other things, the price of shares available for purchase in the open market as compared to the price at which they may purchase shares through the option granted to them under the underwriting agreement described above. “Naked” short sales are sales in excess of such option. The underwriters must close out any naked short position by purchasing shares in the open market. A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of our common stock in the open market after pricing that could adversely affect investors who purchase in the offering. Stabilizing transactions consist of various bids for or purchases of shares of common stock made by the underwriters in the open market prior to the completion of the offering.
The underwriters may also impose a penalty bid. This occurs when a particular underwriter repays to the underwriters a portion of the underwriting discount received by it because the representatives have repurchased shares sold by or for the account of such underwriter in stabilizing or short covering transactions.
Similar to other purchase transactions, the underwriters’ purchases to cover the syndicate short sales may have the effect of raising or maintaining the market price of our common stock or preventing or retarding a decline in the market price of our common stock. As a result, the price of our common stock may be higher than the price that might otherwise exist in the open market. The underwriters may conduct these transactions on the Nasdaq Global Market, in the over-the-counter market or otherwise.
Neither we nor any of the underwriters make any representation or prediction as to the direction or magnitude of any effect that the transactions described above may have on the price of our common stock. In addition, neither we nor any of the underwriters make any representation that the representatives will engage in these transactions or that these transactions, once commenced, will not be discontinued without notice.
Electronic Distribution
In connection with the offering, certain of the underwriters or securities dealers may distribute prospectuses by electronic means, such as e-mail.
Other Relationships
The underwriters and their affiliates are full service financial institutions engaged in various activities, which may include sales and trading, commercial and investment banking, advisory, investment management, investment research, principal investment, hedging, market making, brokerage and other financial and non-financial activities and services. Some of the underwriters and their affiliates have engaged in, and may in the future engage in, investment banking and other commercial dealings in the ordinary course of business with us or our affiliates. They have received, or may in the future receive, customary fees and commissions for these transactions. Silicon Valley Bank, a lender under our loan agreement, is an affiliate of SVB Leerink LLC, one of the underwriters in this offering.
In addition, in the ordinary course of their business activities, the underwriters and their affiliates may make or hold a broad array of investments and actively trade debt and equity securities (or related derivative securities) and financial instruments (including bank loans) for their own account and for the accounts of their customers. Such investments and securities activities may involve securities and/or instruments of ours or our affiliates. The underwriters and their affiliates may also make investment recommendations and/or publish or express independent research views in respect of such securities or financial instruments and may hold, or recommend to clients that they acquire, long and/or short positions in such securities and instruments.
164
Notice to Prospective Investors in the European Economic Area
In relation to each Member State of the European Economic Area which has implemented the Prospectus Directive (each, a “Relevant Member State”), with effect from and including the date on which the Prospectus Directive is implemented in that Relevant Member State, no offer of shares that are the subject of the offering contemplated by this prospectus may be made to the public in that Relevant Member State other than:
| A. | to any legal entity which is a “qualified investor” as defined in the Prospectus Directive; |
| B. | to fewer than 150 natural or legal persons (other than “qualified investors” as defined in the Prospectus Directive), per Relevant Member State, subject to obtaining the prior consent of the representatives of the underwriters; or |
| C. | in any other circumstances falling within Article 3(2) of the Prospectus Directive, |
provided that no such offer of shares shall result in a requirement for us or any underwriter to publish a prospectus pursuant to Article 3 of the Prospectus Directive or a supplemental prospectus pursuant to Article 16 of the Prospectus Directive.
Each person in a Relevant Member State who initially acquires any shares or to whom any offer is made will be deemed to have represented, warranted and agreed to and with the representatives of the underwriters and us that it is a “qualified investor” within the meaning of the law in that Relevant Member State implementing Article 2(1)(e) of the Prospectus Directive. In the case of any shares being offered to a financial intermediary as that term is used in Article 3(2) of the Prospectus Directive, each such financial intermediary will also be deemed to have represented, warranted and agreed that the shares acquired by it in this offering have not been acquired on a non-discretionary basis on behalf of, nor have they been acquired with a view to offering those shares to the public, other than their offer or resale in a Relevant Member State to “qualified investors” as so defined or in circumstances in which the prior consent of the representatives of the underwriters has been obtained to each such proposed offer or resale.
We, the underwriters and their affiliates will rely upon the truth and accuracy of the foregoing representations, warranties and agreements.
This prospectus has been prepared on the basis that any offer of shares in any Relevant Member State will be made pursuant to an exemption under the Prospectus Directive from the requirement to publish a prospectus for offers of shares. Accordingly, any person making or intending to make an offer in that Relevant Member State of shares which are the subject of the offering contemplated in this prospectus may only do so in circumstances in which no obligation arises for us or any of the underwriters to publish a prospectus pursuant to Article 3 of the Prospectus Directive in relation to such offer. Neither we nor the underwriters have authorized, nor do they authorize, the making of any offer of shares in circumstances in which an obligation arises for us or the underwriters to publish a prospectus for such offer.
For purposes of the above provisions, the expression “an offer of shares to the public” in relation to any shares in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and the shares to be offered so as to enable an investor to decide to purchase or subscribe for the shares, as the same may be varied in that Relevant Member State by any measure implementing the Prospectus Directive in that Relevant Member State. The expression “Prospectus Directive” means Directive 2003/71/EC (as amended, including by Directive 2010/73/EU), and includes any relevant implementing measure in the Relevant Member State.
Notice to Prospective Investors in the United Kingdom
In addition, in the United Kingdom, this document is being distributed only to, and is directed only at, and any offer subsequently made may only be directed at persons who are “qualified investors” (as defined in the Prospectus Directive) (i) who have professional experience in matters relating to investments falling within Article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005, as amended, or the Order, and/or (ii) who are high net worth companies (or persons to whom it may otherwise be lawfully communicated) falling within Article 49(2)(a) to (d) of the Order (all such persons together being referred to as “relevant persons”). This document must not be acted on or relied on in the United Kingdom.
165
Any person in the United Kingdom that is not a relevant person should not act or rely on the information included in this document or use it as basis for taking any action. In the United Kingdom, any investment or investment activity that this document relates to may be made or taken exclusively by relevant persons.
Notice to Prospective Investors in Switzerland
The shares may not be publicly offered in Switzerland and will not be listed on the SIX Swiss Exchange, or SIX, or on any other stock exchange or regulated trading facility in Switzerland. This document has been prepared without regard to the disclosure standards for issuance prospectuses under art. 652a or art. 1156 of the Swiss Code of Obligations or the disclosure standards for listing prospectuses under art. 27 ff. of the SIX Listing Rules or the listing rules of any other stock exchange or regulated trading facility in Switzerland. Neither this document nor any other offering or marketing material relating to the shares or the offering may be publicly distributed or otherwise made publicly available in Switzerland.
Neither this document nor any other offering or marketing material relating to the offering, us, or the shares have been or will be filed with or approved by any Swiss regulatory authority. In particular, this document will not be filed with, and the offer of shares will not be supervised by, the Swiss Financial Market Supervisory Authority, and the offer of shares has not been and will not be authorized under the Swiss Federal Act on Collective Investment Schemes, or CISA. The investor protection afforded to acquirers of interests in collective investment schemes under the CISA does not extend to acquirers of shares.
Notice to Prospective Investors in the Dubai International Financial Centre
This prospectus relates to an Exempt Offer in accordance with the Offered Securities Rules of the Dubai Financial Services Authority, or DFSA. This prospectus is intended for distribution only to persons of a type specified in the Offered Securities Rules of the DFSA. It must not be delivered to, or relied on by, any other person. The DFSA has no responsibility for reviewing or verifying any documents in connection with Exempt Offers. The DFSA has not approved this prospectus nor taken steps to verify the information set forth herein and has no responsibility for the prospectus. The shares to which this prospectus relates may be illiquid and/or subject to restrictions on their resale. Prospective purchasers of the shares offered should conduct their own due diligence on the shares. If you do not understand the contents of this prospectus you should consult an authorized financial advisor.
Notice to Prospective Investors in Australia
No placement document, prospectus, product disclosure statement or other disclosure document has been lodged with the Australian Securities and Investments Commission, or ASIC, in relation to the offering. This prospectus does not constitute a prospectus, product disclosure statement or other disclosure document under the Corporations Act 2001, or the Corporations Act, and does not purport to include the information required for a prospectus, product disclosure statement or other disclosure document under the Corporations Act.
Any offer in Australia of the shares may only be made to persons, or the Exempt Investors, who are “sophisticated investors” (within the meaning of section 708(8) of the Corporations Act), “professional investors” (within the meaning of section 708(11) of the Corporations Act) or otherwise pursuant to one or more exemptions contained in section 708 of the Corporations Act so that it is lawful to offer the shares without disclosure to investors under Chapter 6D of the Corporations Act.
The shares applied for by Exempt Investors in Australia must not be offered for sale in Australia in the period of 12 months after the date of allotment under the offering, except in circumstances where disclosure to investors under Chapter 6D of the Corporations Act would not be required pursuant to an exemption under section 708 of the Corporations Act or otherwise or where the offer is pursuant to a disclosure document which complies with Chapter 6D of the Corporations Act. Any person acquiring shares must observe such Australian on-sale restrictions.
This prospectus contains general information only and does not take account of the investment objectives, financial situation or particular needs of any particular person. It does not contain any securities recommendations or financial product advice. Before making an investment decision, investors need to consider whether the information in this prospectus is appropriate to their needs, objectives and circumstances, and, if necessary, seek expert advice on those matters.
166
Notice to Prospective Investors in Hong Kong
The securities have not been offered or sold and will not be offered or sold in Hong Kong, by means of any document, other than (a) to “professional investors” as defined in the Securities and Futures Ordinance (Cap. 571) of Hong Kong and any rules made under that Ordinance; or (b) in other circumstances which do not result in the document being a “prospectus” as defined in the Companies Ordinance (Cap. 32) of Hong Kong or which do not constitute an offer to the public within the meaning of that Ordinance. No advertisement, invitation or document relating to the securities has been or may be issued or has been or may be in the possession of any person for the purposes of issue, whether in Hong Kong or elsewhere, which is directed at, or the contents of which are likely to be accessed or read by, the public of Hong Kong (except if permitted to do so under the securities laws of Hong Kong) other than with respect to securities which are or are intended to be disposed of only to persons outside Hong Kong or only to “professional investors” as defined in the Securities and Futures Ordinance and any rules made under that Ordinance.
Notice to Prospective Investors in Japan
The securities have not been and will not be registered under the Financial Instruments and Exchange Law of Japan (Law No. 25 of 1948, as amended) and, accordingly, will not be offered or sold, directly or indirectly, in Japan, or for the benefit of any Japanese Person or to others for re-offering or resale, directly or indirectly, in Japan or to any Japanese Person, except in compliance with all applicable laws, regulations and ministerial guidelines promulgated by relevant Japanese governmental or regulatory authorities in effect at the relevant time. For the purposes of this paragraph, “Japanese Person” shall mean any person resident in Japan, including any corporation or other entity organized under the laws of Japan.
Notice to Prospective Investors in Singapore
This prospectus has not been registered as a prospectus with the Monetary Authority of Singapore. Accordingly, this prospectus and any other document or material in connection with the offer or sale, or invitation for subscription or purchase, of Non-CIS Securities may not be circulated or distributed, nor may the Non-CIS Securities be offered or sold, or be made the subject of an invitation for subscription or purchase, whether directly or indirectly, to persons in Singapore other than (i) to an institutional investor under Section 274 of the Securities and Futures Act, Chapter 289 of Singapore, or the SFA, (ii) to a relevant person pursuant to Section 275(1), or any person pursuant to Section 275(1A), and in accordance with the conditions specified in Section 275, of the SFA, or (iii) otherwise pursuant to, and in accordance with the conditions of, any other applicable provision of the SFA.
Where the Non-CIS Securities are subscribed or purchased under Section 275 of the SFA by a relevant person which is:
| (a) | a corporation (which is not an accredited investor (as defined in Section 4A of the SFA)) the sole business of which is to hold investments and the entire share capital of which is owned by one or more individuals, each of whom is an accredited investor; or |
| (b) | a trust (where the trustee is not an accredited investor) whose sole purpose is to hold investments and each beneficiary of the trust is an individual who is an accredited investor, |
securities (as defined in Section 239(1) of the SFA) of that corporation or the beneficiaries’ rights and interest (howsoever described) in that trust shall not be transferred within six months after that corporation or that trust has acquired the Non-CIS Securities pursuant to an offer made under Section 275 of the SFA except:
| (a) | to an institutional investor or to a relevant person defined in Section 275(2) of the SFA, or to any person arising from an offer referred to in Section 275(1A) or Section 276(4)(i)(B) of the SFA; |
| (b) | where no consideration is or will be given for the transfer; |
| (c) | where the transfer is by operation of law; |
| (d) | as specified in Section 276(7) of the SFA; or |
| (e) | as specified in Regulation 32 of the Securities and Futures (Offers of Investments) (Shares and Debentures) Regulations 2005 of Singapore. |
167
Notice to Prospective Investors in Canada
The shares may be sold only to purchasers purchasing, or deemed to be purchasing, as principal that are accredited investors, as defined in National Instrument 45-106 Prospectus Exemptions or subsection 73.3(1) of the Securities Act (Ontario), and are permitted clients, as defined in National Instrument 31-103 Registration Requirements, Exemptions and Ongoing Registrant Obligations. Any resale of the shares must be made in accordance with an exemption from, or in a transaction not subject to, the prospectus requirements of applicable securities laws.
Securities legislation in certain provinces or territories of Canada may provide a purchaser with remedies for rescission or damages if this prospectus (including any amendment thereto) contains a misrepresentation, provided that the remedies for rescission or damages are exercised by the purchaser within the time limit prescribed by the securities legislation of the purchaser’s province or territory. The purchaser should refer to any applicable provisions of the securities legislation of the purchaser’s province or territory for particulars of these rights or consult with a legal advisor.
Pursuant to section 3A.3 (or, in the case of securities issued or guaranteed by the government of a non-Canadian jurisdiction, section 3A.4) of National Instrument 33-105 Underwriting Conflicts (NI 33-105), the underwriters are not required to comply with the disclosure requirements of NI 33-105 regarding underwriter conflicts of interest in connection with this offering.
168
The validity of the shares of common stock being offered by this prospectus will be passed upon for us by Cooley LLP. The underwriters are being represented by Latham & Watkins LLP.
The financial statements of Castle Biosciences, Inc. as of December 31, 2017 and 2018, and for each of the years in the two-year period ended December 31, 2018, have been included herein and in the registration statement in reliance upon the report of KPMG LLP, independent registered public accounting firm, appearing elsewhere herein, and upon the authority of said firm as experts in accounting and auditing. The audit report covering the December 31, 2017 and 2018 financial statements contains an explanatory paragraph that states there are uncertainties about the Company's ability to comply with certain debt covenants under its long-term debt that is required to finance operations, which raises substantial doubt about the entity's ability to continue as a going concern. The financial statements do not include any adjustments that might result from the outcome of that uncertainty. The audit report covering the December 31, 2017 and 2018 financial statements refers to a change to the method of accounting for revenue from contracts with customers in 2018 due to the adoption of Accounting Standards Update 2014-09, Revenue from Contracts with Customers (Topic 606), as amended.
WHERE YOU CAN FIND ADDITIONAL INFORMATION
We have filed with the SEC a registration statement on Form S-1 under the Securities Act, with respect to the shares of common stock being offered by this prospectus. This prospectus does not contain all of the information in the registration statement and its exhibits. For further information with respect to us and the common stock offered by this prospectus, we refer you to the registration statement and its exhibits. Statements contained in this prospectus as to the contents of any contract or any other document referred to are not necessarily complete, and in each instance, we refer you to the copy of the contract or other document filed as an exhibit to the registration statement. Each of these statements is qualified in all respects by this reference.
You can read our SEC filings, including the registration statement, over the Internet at the SEC’s website at www.sec.gov. You may also read and copy any document we file with the SEC at its public reference facilities at 100 F Street NE, Washington, D.C. 20549. You may also obtain copies of these documents at prescribed rates by writing to the Public Reference Section of the SEC at 100 F Street N.E., Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference facilities. You may also request a copy of these filings, at no cost, by writing us at 820 S. Friendswood Drive, Suite 201, Friendswood, Texas 77546, or calling us at (832) 974-1550.
Upon the completion of this offering, we will be subject to the information reporting requirements of the Exchange Act, and we will file reports, proxy statements and other information with the SEC. These reports, proxy statements and other information will be available for inspection and copying at the public reference room and website of the SEC referred to above. We also maintain a website at www.castlebiosciences.com, at which, following the completion of this offering, you may access these materials free of charge as soon as reasonably practicable after they are electronically filed with, or furnished to, the SEC. The information contained in, or that can be accessed through, our website is not incorporated by reference in, and is not part of, this prospectus.
169
CASTLE BIOSCIENCES, INC.
INDEX TO FINANCIAL STATEMENTS
Page |
|||
Audited Financial Statements: |
|||
Interim Condensed Financial Statements (Unaudited): |
|||
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Stockholders and Board of Directors
Castle Biosciences, Inc.:
Opinion on the Financial Statements
We have audited the accompanying balance sheets of Castle Biosciences, Inc. (the Company) as of December 31, 2017 and 2018, the related statements of operations and comprehensive loss, convertible preferred stock and stockholders’ deficit, and cash flows for each of the years in the two-year period ended December 31, 2018, and the related notes (collectively, the financial statements). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2017 and 2018, and the results of its operations and its cash flows for each of the years in the two-year period ended December 31, 2018, in conformity with U.S. generally accepted accounting principles.
Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in note 1 to the financial statements, there are uncertainties about the Company's ability to comply with certain debt covenants under its long-term debt that is required to finance operations, which raises substantial doubt about its ability to continue as a going concern. Management's plans in regard to these matters are also described in note 1. The financial statements do not include any adjustments that might result from the outcome of these uncertainties.
Change in Accounting Principle
As discussed in note 2 to the financial statements, the Company has elected to change its method of accounting for revenue from contracts with customers in 2018 due to the adoption of Accounting Standards Update No. 2014-09, Revenue from Contracts with Customers (Topic 606), as amended. The Company adopted Topic 606 using the full retrospective method.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ KPMG LLP
We have served as the Company’s auditor since 2018.
San Diego, California
May 13, 2019
F-2
CASTLE BIOSCIENCES, INC.
BALANCE SHEETS
December 31, |
||||||
2017 |
2018 |
|||||
ASSETS |
||||||
Current Assets |
||||||
Cash and cash equivalents |
$ | 1,212,063 | $ | 4,478,512 | ||
Accounts receivable, net |
4,652,317 | 12,089,719 | ||||
Inventory |
304,576 | 882,233 | ||||
Prepaid expenses and other current assets |
515,510 | 675,562 | ||||
Total current assets |
6,684,466 | 18,126,026 | ||||
Long-term accounts receivable, net |
1,561,786 | 2,532,011 | ||||
Property and equipment, net |
1,469,651 | 1,528,996 | ||||
Intangible assets, net |
40,119 | 4,167 | ||||
Other assets – long-term |
64,300 | 213,735 | ||||
Total assets |
$ | 9,820,322 | $ | 22,404,935 | ||
LIABILITIES, CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ DEFICIT |
||||||
Current Liabilities |
||||||
Accounts payable |
$ | 1,116,725 | $ | 1,450,766 | ||
Accrued compensation |
2,496,279 | 4,571,011 | ||||
Other accrued liabilities |
607,845 | 715,244 | ||||
Current portion of long-term debt |
1,000,000 | — | ||||
Total current liabilities |
5,220,849 | 6,737,021 | ||||
Long-term debt |
17,602,686 | 24,499,752 | ||||
Preferred stock warrant liability |
524,694 | 1,193,726 | ||||
Accrued compensation − long-term |
727,558 | — | ||||
Deferred rent liability |
15,791 | 43,587 | ||||
Total liabilities |
24,091,578 | 32,474,086 | ||||
Commitments and Contingencies (Note 10) |
||||||
Convertible Preferred Stock |
||||||
Convertible preferred stock Series C, $0.001 par value, 503,056 shares authorized as of December 31, 2017 and 2018; 503,056 shares issued and outstanding as of December 31, 2017 and 2018; $2,277,144 and $2,417,195 aggregate liquidation preference as of December 31, 2017 and 2018, respectively |
1,500,994 | 1,500,994 | ||||
Redeemable convertible preferred stock Series A, B, D, E-1, E-2, E-2A, E-3 and F, $0.001 par value, 8,738,465 and 9,640,493 shares authorized as of December 31, 2017 and 2018, respectively; 7,611,010 and 9,456,775 shares issued and outstanding as of December 31, 2017 and 2018, respectively; $43,396,181 and $57,570,036 aggregate liquidation preference as of December 31, 2017 and 2018, respectively |
34,538,255 | 44,995,157 | ||||
Stockholders’ Deficit |
||||||
Common stock, $0.001 par value; 15,000,000 and 15,102,000 shares authorized as of December 31, 2017 and 2018, respectively; 2,311,798 and 2,335,880 shares issued and outstanding as of December 31, 2017 and 2018, respectively |
2,312 | 2,336 | ||||
Additional paid-in capital |
808,767 | 920,940 | ||||
Accumulated deficit |
(51,121,584 | ) |
(57,488,578 | ) |
||
Total stockholders’ deficit |
(50,310,505 | ) |
(56,565,302 | ) |
||
Total liabilities, convertible preferred stock and stockholders’ deficit |
$ | 9,820,322 | $ | 22,404,935 | ||
The accompanying notes are an integral part of these financial statements.
F-3
CASTLE BIOSCIENCES, INC.
STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
Years Ended December 31, |
||||||
2017 |
2018 |
|||||
NET REVENUES |
$ | 13,753,912 | $ | 22,785,734 | ||
COST OF SALES |
4,921,854 | 5,296,849 | ||||
Gross margin |
8,832,058 | 17,488,885 | ||||
OPERATING EXPENSES |
||||||
Research and development |
4,473,136 | 4,854,232 | ||||
Selling, general and administrative |
15,258,598 | 16,470,296 | ||||
Total operating expenses |
19,731,734 | 21,324,528 | ||||
Operating loss |
(10,899,676 | ) |
(3,835,643 | ) |
||
Interest income |
26,184 | 23,514 | ||||
Interest expense |
(1,649,594 | ) |
(2,273,842 | ) |
||
Other income (expense), net |
163,288 | (272,417 | ) |
|||
Loss before income taxes |
(12,359,798 | ) |
(6,358,388 | ) |
||
Income tax expense |
9,731 | 8,606 | ||||
Net loss and comprehensive loss |
(12,369,529 | ) |
(6,366,994 | ) |
||
Convertible preferred stock cumulative dividends |
2,896,966 | 3,576,743 | ||||
Accretion of redeemable convertible preferred stock to redemption value |
40,932 | 219,138 | ||||
Net loss and comprehensive loss attributable to common stockholders |
$ | (15,307,427 | ) |
$ | (10,162,875 | ) |
Loss per share attributable to common stockholders, basic and diluted |
$ | (6.62 | ) |
$ | (4.37 | ) |
Weighted-average shares outstanding, basic and diluted |
2,310,640 | 2,323,197 | ||||
Pro forma loss per share, basic and diluted (unaudited) |
$ | |||||
Pro forma weighted-average shares outstanding, basic and diluted (unaudited) |
||||||
The accompanying notes are an integral part of these financial statements.
F-4
Convertible Preferred Stock Series C |
Redeemable Convertible Preferred Stock Series A, B, D, E-1, E-2, E-2A, E-3 and F |
Common Stock |
Additional Paid-in Capital |
Accumulated Deficit |
Total Stockholders’ Deficit |
||||||||||||||||||||||
Shares |
Amount |
Shares |
Amount |
Shares |
Amount |
||||||||||||||||||||||
BALANCE, JANUARY 1, 2017 |
503,056 | $ | 1,500,994 | 7,524,343 | $ | 34,208,160 | 2,309,298 | $ | 2,309 | $ | 737,455 | $ | (38,752,055 | ) |
$ | (38,012,291 | ) |
||||||||||
Stock compensation expense |
— | — | — | — | — | — | 108,696 | — | 108,696 | ||||||||||||||||||
Exercise of common stock options |
— | — | — | — | 2,500 | 3 | 3,548 | — | 3,551 | ||||||||||||||||||
Accretion of redeemable convertible preferred stock to redemption value: |
|||||||||||||||||||||||||||
Series E-1 |
— | — | — | 4,174 | — | — | (4,174 | ) |
— | (4,174 | ) |
||||||||||||||||
Series E-2A |
— | — | — | 383 | — | — | (383 | ) |
— | (383 | ) |
||||||||||||||||
Series E-3 |
— | — | — | 11,565 | — | — | (11,565 | ) |
— | (11,565 | ) |
||||||||||||||||
Series F |
— | — | — | 24,810 | — | — | (24,810 | ) |
— | (24,810 | ) |
||||||||||||||||
Exercise of Series B redeemable convertible preferred stock warrants |
— | — | 86,667 | 289,163 | — | — | — | — | — | ||||||||||||||||||
Net loss |
— | — | — | — | — | — | — | (12,369,529 | ) |
(12,369,529 | ) |
||||||||||||||||
BALANCE, DECEMBER 31, 2017 |
503,056 | 1,500,994 | 7,611,010 | 34,538,255 | 2,311,798 | 2,312 | 808,767 | (51,121,584 | ) |
(50,310,505 | ) |
||||||||||||||||
Stock compensation expense |
— | — | — | — | — | — | 293,639 | — | 293,639 | ||||||||||||||||||
Exercise of common stock options |
— | — | — | — | 24,082 | 24 | 37,672 | — | 37,696 | ||||||||||||||||||
Issuance of Series F redeemable convertible preferred stock |
— | — | 1,809,564 | 9,990,482 | — | — | — | — | — | ||||||||||||||||||
Accretion of redeemable convertible preferred stock to redemption value: |
|||||||||||||||||||||||||||
Series E-1 |
— | — | — | 4,180 | — | — | (4,180 | ) |
— | (4,180 | ) |
||||||||||||||||
Series E-2A |
— | — | — | 384 | — | — | (384 | ) |
— | (384 | ) |
||||||||||||||||
Series E-3 |
— | − | — | 11,595 | — | — | (11,595 | ) |
— | (11,595 | ) |
||||||||||||||||
Series F |
— | — | — | 202,979 | — | — | (202,979 | ) |
— | (202,979 | ) |
||||||||||||||||
Exercise of redeemable convertible preferred stock warrants: |
|||||||||||||||||||||||||||
Series E-1 |
— | — | 3,250 | 14,983 | — | — | — | — | — | ||||||||||||||||||
Series F |
— | — | 32,951 | 232,299 | — | — | — | — | — | ||||||||||||||||||
Net loss |
— | — | — | — | — | — | — | (6,366,994 | ) |
(6,366,994 | ) |
||||||||||||||||
BALANCE, DECEMBER 31, 2018 |
503,056 | $ | 1,500,994 | 9,456,775 | $ | 44,995,157 | 2,335,880 | $ | 2,336 | $ | 920,940 | $ | (57,488,578 | ) |
$ | (56,565,302 | ) |
||||||||||
The accompanying notes are an integral part of these financial statements.
F-5
CASTLE BIOSCIENCES, INC.
STATEMENTS OF CASH FLOWS
Years Ended December 31, |
||||||
2017 |
2018 |
|||||
OPERATING ACTIVITIES |
||||||
Net loss |
$ | (12,369,529 | ) |
$ | (6,366,994 | ) |
Adjustments to reconcile net loss to net cash used in operating activities: |
||||||
Depreciation |
270,308 | 287,064 | ||||
Stock compensation expense |
108,696 | 293,639 | ||||
Amortization of intangibles |
40,512 | 35,952 | ||||
Amortization of debt discount and issuance costs |
520,228 | 566,260 | ||||
Change in fair value of preferred stock warrant liability |
(163,288 | ) |
272,417 | |||
Other |
— | (23,887 | ) |
|||
Change in operating assets and liabilities: |
||||||
Accounts receivable |
(428,211 | ) |
(8,407,626 | ) |
||
Prepaid expenses and other current assets |
(134,241 | ) |
(160,052 | ) |
||
Inventory |
(115,556 | ) |
(577,657 | ) |
||
Other assets |
(31,080 | ) |
12,884 | |||
Accounts payable |
370,902 | 197,248 | ||||
Accrued compensation |
(184,529 | ) |
1,347,174 | |||
Other accrued liabilities |
(122,120 | ) |
201,271 | |||
Deferred rent liability |
4,321 | 27,796 | ||||
Net cash used in operating activities |
(12,233,587 | ) |
(12,294,511 | ) |
||
INVESTING ACTIVITES |
||||||
Purchases of property and equipment |
(494,854 | ) |
(277,036 | ) |
||
Net cash used in investing activities |
(494,854 | ) |
(277,036 | ) |
||
FINANCING ACTIVITIES |
||||||
Proceeds from issuance of preferred stock and preferred stock warrants (including exercised warrants) |
289,163 | 10,382,507 | ||||
Proceeds from issuance of term debt and preferred stock warrants, net of issuance costs |
6,573,729 | 4,417,793 | ||||
Proceeds from line of credit |
4,000,000 | 1,000,000 | ||||
Repayment of line of credit |
(2,000,000 | ) |
— | |||
Proceeds from exercise of common stock options |
3,551 | 37,696 | ||||
Net cash provided by financing activities |
8,866,443 | 15,837,996 | ||||
NET CHANGE IN CASH AND CASH EQUIVALENTS |
(3,861,998 | ) |
3,266,449 | |||
Beginning of year |
5,074,061 | 1,212,063 | ||||
End of year |
$ | 1,212,063 | $ | 4,478,512 | ||
SUPPLEMENTAL DISCLOSURE OF CASH PAID FOR: |
||||||
Interest |
$ | 1,107,633 | $ | 1,635,438 | ||
Income taxes |
$ | — | $ | 159,732 | ||
DISCLOSURE OF NON-CASH INVESTING AND FINANCING ACTIVITIES: |
||||||
Accrued capital expenditures |
$ | 12,913 | $ | 58,398 | ||
Deferred offering costs incurred but not paid |
$ | — | $ | 91,307 | ||
The accompanying notes are an integral part of these financial statements.
F-6
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS
1. Organization and Description of Business
Castle Biosciences, Inc. (the “Company”) was incorporated in the state of Delaware on September 12, 2007. The Company is a commercial-stage dermatological cancer company focused on providing physicians and their patients with personalized, clinically actionable genomic information to make more accurate treatment decisions. The Company is based in Friendswood, Texas (a suburb of Houston, Texas) and laboratory operations are conducted in the Company’s facilities located in Phoenix, Arizona.
Going Concern, Liquidity and Capital Resources
The Company has incurred significant operating losses since its inception. As of December 31, 2017 and 2018, the Company had an accumulated deficit of $51.1 million and $57.5 million, respectively, cash and cash equivalents totaling $1.2 million and $4.5 million respectively, and working capital of $1.5 million and $11.4 million, respectively. The Company also has substantial indebtedness, the terms of which require it to meet a monthly six-month trailing revenue covenant. Although the Company is in compliance with this covenant as of December 31, 2018, management’s projections indicate that the Company may be unable to maintain compliance with the revenue covenant during the next 12 months. The Company is currently in discussion with its lenders to amend the covenant. There can be no assurance that the Company will be successful in obtaining an amendment or a waiver. If the Company fails to maintain compliance with the covenant, the Company’s lenders could accelerate the repayment of the debt. In such event, there is no assurance the Company would be able to refinance the debt on terms acceptable to the Company, if at all. These factors raise substantial doubt about the Company’s ability to continue as a going concern. These financial statements do not include any adjustments to reflect the possible future effects of the recoverability and classification of assets or the amounts and classification of liabilities that might be necessary should the Company be unable to continue as a going concern.
The Company has financed its operations to date primarily through private placements of its preferred stock, the sale of its prognostic testing services, and bank debt borrowings. The ability of the Company to fund operations according to plan is largely dependent on its ability to improve the timing and amount of collections from the sale of tests; however, as the attainment of managed care coverage policy decisions is ordinarily a protracted process, funding from other sources will likely be necessary.
While the Company intends to substantially fund planned operations for the next 12 months using cash on hand and collections from test sales, management expects that a net usage of cash will continue to occur through 2019 and, consequently, additional capital from other sources will be required. As discussed in Note 15, “Subsequent Events,” the Company secured $11.8 million in financing through the sale of unsecured convertible notes. Additional equity and debt capital are believed by management to be available and funding arrangements are actively being sought as of the date of this report. In addition to the procurement of capital to fund operations, management has identified discretionary spending included in the Company’s operating plan that can be deferred until prudently effectuated.
2. Summary of Significant Accounting Policies
Basis of Presentation
The Company’s financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”). The Company has no subsidiaries and all operations are conducted by the Company.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities as of the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Significant items subject to such estimates include revenue recognition, the determination of fair value of the Company’s preferred stock warrants, the valuation of stock options, assessing future tax exposure and the realization of deferred tax assets, the useful lives and recoverability of property and equipment, and contingent liabilities. The Company bases these estimates on historical and anticipated results, trends, and various
F-7
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
other assumptions that the Company believes are reasonable under the circumstances, including assumptions as to future events. These estimates form the basis for making judgments about the carrying values of assets and liabilities and recorded revenues and expenses that are not readily apparent from other sources. Actual results could differ from those estimates and assumptions.
Operating Segments
Operating segments are identified as components of an enterprise about which separate discrete financial information is available for evaluation by the chief operating decision maker, or decision-making group, in making decisions on how to allocate resources and assess performance. The Company views its operations and manages its business as one operating segment. All revenues are attributable to U.S.-based operations and all assets are held in the United States.
Cash and Cash Equivalents including Concentrations of Credit Risk
Cash equivalents consist of short-term, highly-liquid investments with original maturities of three months or less. Cash equivalents consist primarily of amounts invested in money market accounts. A majority of the Company’s cash and cash equivalents are deposited with a single financial institution. Deposits in this institution may exceed the amount of insurance provided on such deposits by the Federal Deposit Insurance Corporation for U.S. institutions. The Company has not experienced any losses on its deposits of cash and cash equivalents. Management believes that the Company is not exposed to significant credit risk due to the financial position of the depository institutions in which those deposits are held.
Revenue Recognition
Revenue is recognized in accordance with Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 606, Revenue from Contracts with Customers (“ASC 606”). The Company adopted ASC 606 with an initial application date of January 1, 2018 using the full retrospective method, as discussed under Recently Adopted Accounting Principles below. In accordance with ASC 606, the Company follows a five-step process to recognize revenues: 1) identify the contract with the customer, 2) identify the performance obligations, 3) determine the transaction price, 4) allocate the transaction price to the performance obligations and 5) recognize revenues when the performance obligations are satisfied.
All of the Company’s revenues from contracts with customers are associated with the provision of diagnostic and prognostic cancer testing services. Most of the Company’s revenues are attributable to DecisionDx-Melanoma for cutaneous melanoma. The Company also provides a test for uveal melanoma, DecisionDx-UM. Information on the disaggregation of revenues by the Company’s significant third-party payors is included under Payor Concentration below. The Company has determined that it has a contract with the patient when the treating clinician orders the test. The Company’s contracts generally contain a single performance obligation, which is the delivery of the test report, and the Company satisfies its performance obligation at a point in time upon the delivery of the test report to the treating physician, which then triggers the billing for the service. The amount of revenue recognized reflects the amount of consideration the Company expects to be entitled (the “transaction price”) and considers the effects of variable consideration, which is discussed further below.
Once the Company satisfies its performance obligations and bills for the service, the timing of the collection of payments may vary based on the payment practices of the third-party payor and the existence of contractually established reimbursement rates. Most of the payments for the Company’s services are made by third-party payors, including Medicare and commercial health insurance carriers. Certain contracts contain a contractual commitment of a reimbursement rate that differs from the Company’s list prices. However, absent a contractually committed reimbursement rate with a commercial carrier or governmental program, the Company’s diagnostic tests may or may not be covered by these entities’ existing reimbursement policies. In addition, patients do not enter into direct agreements with the Company that commit them to pay any portion of the cost of the tests in the event that their insurance provider declines to reimburse the Company. The Company may pursue, on a case-by-case basis, reimbursement from such patients in the form of co-payments and co-insurance, in accordance with the contractual obligations that the Company has with the insurance carrier or health plan. These situations may result in a delay in the collection of payments.
F-8
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
The Medicare claims that are covered by policy under a Local Coverage Determination (“LCD”) are generally paid at the established rate by the Company’s Medicare contractor within 30 days from receipt. Medicare claims that were either submitted to Medicare prior to the LCD coverage commencement date or are not covered by the terms of the LCD but meet the definition of being medically reasonable and necessary pursuant to the controlling 42 C.F.R §410.32(a) regulation are ultimately reviewed and payment is determined by an Administrative Law Judge (“ALJ”), at an appeal hearing.
The Company has concluded that its contracts include variable consideration because the amounts paid by Medicare or commercial health insurance carriers may be paid at less than the Company’s standard rates or not paid at all, with such differences considered implicit price concessions. Variable consideration attributable to these price concessions is measured at the expected value using the “most likely amount” method under ASC 606. The amounts are determined by historical average collection rates by test type and payor category taking into consideration the range of possible outcomes, the predictive value of the Company’s past experiences, the time period of when uncertainties expect to be resolved and the amount of consideration that is susceptible to factors outside of the Company’s influence, such as the judgment and actions of third parties. Such variable consideration is included in the transaction price only to the extent it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainties with respect to the amount are resolved. Variable consideration may be constrained and excluded from the transaction price in situations where there is no contractually agreed upon reimbursement coverage or in the absence of a predictable pattern and history of collectability with a payor. Variable consideration for Medicare claims is deemed to be fully constrained when the payment of such claims is subject to approval by an ALJ at an appeal hearing, due to factors outside the Company’s influence (i.e., judgment or actions of third parties) and the uncertainty of the amount to be received is not expected to be resolved for a long period of time. Variable consideration is evaluated each reporting period and adjustments are recorded as increases or decreases in revenues. Included in revenues for the years ended December 31, 2017 and 2018 was $0 and $867,157, respectively, of revenue decreases associated with changes in estimated variable consideration related to performance obligations satisfied in previous periods. The amount was zero for the year ended December 31, 2017 due to the application of a transition practical expedient in connection with the adoption of ASC 606, as discussed below under Recently Adopted Accounting Pronouncements.
Because the Company’s contracts have an expected duration of one year or less, the Company has elected the practical expedient in ASC 606 to not disclose information about its remaining performance obligations. Any incremental costs to obtain contracts are recorded as selling, general and administrative expense as incurred due to the short duration of the Company’s contracts. Contract balances consisted solely of accounts receivable (both current and noncurrent) as of December 31, 2017 and 2018.
DecisionDx-Melanoma Claims Consolidation
In June 2017, the Company submitted to the Office of Medicare Hearings and Appeals (“OMHA”) a formal request to participate in a program that OMHA developed with the intent of providing appellants a means to have large volumes of claim disputes adjudicated at an accelerated rate. The program consolidates outstanding claims at the ALJ level and uses a statistical-sampling approach where five ALJ’s will determine reimbursement results for a sample of claims which are then extrapolated to the universe of claims. The consolidation includes 2,698 DecisionDx-Melanoma claims dating from 2013 through spring 2017. The judges who will review the sample sets have been identified and the hearings were held in April 2019 and no formal ruling has been issued to date. In accordance with ASC 606, the Company has not recognized (fully constrained the variable consideration) any revenues attributable to these claims in its financial statements pending the outcome of this matter. The Company expects to recognize any revenue adjudicated by the ALJ in the periodic reporting period in which the Company is notified of the ALJ hearing outcome.
Payor Concentration
The Company relies upon reimbursements from third-party government payors (primarily Medicare) and private-payor insurance companies to collect accounts receivable related to sales of its diagnostic tests.
F-9
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
The Company’s significant third-party payors and their related revenues as a percentage of total revenues and accounts receivable balances are as follows:
Percentage of Revenues |
Percentage of Accounts Receivable (current) |
Percentage of Accounts Receivable (non-current) |
||||||||||||||||
Years Ended December 31, |
As of December 31, |
As of December 31, |
||||||||||||||||
2017 |
2018 |
2017 |
2018 |
2017 |
2018 |
|||||||||||||
Medicare |
9 | % |
36 | % |
— | % |
54 | % |
— | % |
— | % |
||||||
Medicare Advantage plans |
23 | % |
17 | % |
22 | % |
9 | % |
16 | % |
18 | % |
||||||
United Healthcare |
11 | % |
12 | % |
10 | % |
7 | % |
15 | % |
15 | % |
||||||
BlueCross BlueShield plans |
30 | % |
18 | % |
41 | % |
18 | % |
45 | % |
41 | % |
||||||
Accounts Receivable and Allowance for Doubtful Accounts
The Company classifies accounts receivable balances that are expected to be paid more than one year from the balance sheet date as non-current assets. The estimated timing of payment utilized as a basis for classification as non-current is determined by analyses of historical payor-specific payment experience, adjusted for known factors that are expected to change the timing of future payments.
The Company accrues an allowance for doubtful accounts against its accounts receivable when it is probable that an account is not collectible, based on write off history, credit risk of specific accounts, aging analysis and other information available on specific accounts. The Company generally does not perform evaluations of customers’ financial condition and generally does not require collateral. Accounts receivable are written off when all efforts to collect the balance have been exhausted. Historically, the Company’s bad debt expense has not been significant. The allowance for doubtful accounts was zero as of December 31, 2017 and 2018. Adjustments for implicit price concessions attributable to variable consideration, as discussed above, are incorporated into the measurement of the accounts receivable balances and are not part of the allowance for doubtful accounts.
Inventory
The Company carries an inventory of test supplies in the Phoenix, Arizona laboratory. The inventory is carried at the lower of weighted average cost and net realizable value and expensed through cost of sales as the supplies are used.
Property and Equipment
Property and equipment are stated at cost less accumulated depreciation and amortization. Depreciation is computed using the straight-line method over the estimated useful lives of the assets, generally between five and ten years. Leasehold improvements are amortized using the straight-line method over the shorter of the estimated useful life of the asset or the term of the lease. The Company’s leasehold improvements primarily relate to its office and laboratory in Phoenix, Arizona, and are generally being amortized through the end of the lease term in July 2023. Maintenance and repairs are charged to expense as incurred, and material improvements are capitalized. When assets are retired or otherwise disposed of, the cost and accumulated depreciation are removed from the balance sheet and any resulting gain or loss is reflected in the statements of operations and comprehensive loss in the period realized.
Patent Rights Licenses
The Company capitalizes the purchase of material licenses that grant interest in and to certain patent rights owned primarily by research and education institutions. These costs are amortized using the straight-line method over the shorter of the period of expected commercial benefit or the life of the underlying patent. Amortization of patent license costs is charged to research and development expense.
Long-Lived Assets
The Company reviews long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of the assets may not be recoverable. An impairment loss is recognized when the total of estimated future undiscounted cash flows, expected to result from the use of the asset and its eventual
F-10
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
disposition, are less than the carrying amount. Impairment, if any, would be calculated based on the excess of the carrying amount of the long-lived asset over the long-lived asset’s fair value. There were no impairment charges recognized for the years ended December 31, 2017 and 2018.
Fair Value of Financial Instruments
The carrying amount of the Company’s long-term debt approximates fair value due to its variable market interest rate and management’s opinion that current rates and terms that would be available to the Company with the same maturity and security structure would be essentially equivalent to that of the Company’s long-term debt. Refer to Note 8 for additional information on fair value measurements.
Deferred Rent
The Company has negotiated certain landlord/tenant incentives, rent holidays and escalations in the base price of rent payments under operating leases. The Company recognizes these incentives, rent holidays and rent escalations on a straight-line basis over the lease term. Deferred rent balances are classified as current or non-current in the accompanying balance sheets based upon the period when reversal of the liability is expected to occur.
Cost of Sales
Cost of sales is expensed as incurred and includes direct labor costs, equipment, supplies, materials and infrastructure expenses associated with testing tissue samples, third-party lab processing and service costs, third-party collection costs, and shipping charges to transport samples.
Research and Development
Research and development costs are charged to operations as incurred. Advance payments for goods and services that will be used in future research and development activities are expensed when the activity has been performed or when the goods have been received rather than when the payment is made. Upfront and milestone payments due to third parties that perform research and development services on behalf of the Company will be expensed as services are rendered or when the milestone is achieved.
Research and development costs include, but are not limited to, payroll and personnel-related expenses, stock-based compensation expense, materials, laboratory supplies, and consulting costs.
Selling, General and Administrative Expenses
Selling, general and administrative (“SG&A”) expenses are attributable to sales, marketing, executive, finance and accounting, legal and human resources functions. These expenses consist of personnel costs (including salaries, employee benefit costs, bonuses and equity-based compensation expenses), customer services expenses, direct marketing expenses, educational and promotional expenses, market research, audit and legal expenses, and consulting. The Company expenses all SG&A costs as incurred.
Accrued Compensation
The Company accrues for liabilities under discretionary employee and executive bonus plans. These estimated compensation liabilities are based on progress against corporate objectives approved by the Board of Directors, compensation levels of eligible individuals, and target bonus percentage levels. The Board of Directors reviews and evaluates the performance against these objectives and ultimately determines what discretionary payments are made. As of December 31, 2017, and 2018, the Company accrued approximately and $2,148,128 and $3,197,234, respectively, for liabilities associated with these employee and executive bonus plans. These amounts are classified as current or noncurrent accrued liabilities in the balance sheets based on the expected timing of payment.
Retirement Plan
The Company has an Internal Revenue Code (“IRC”) Section 401(k) profit sharing plan (the “Plan”) for eligible employees. The Plan is funded by employee contributions and provides for discretionary contributions in the form of matching and/or profit-sharing contributions. For the years ended December 31, 2017 and 2018, the Company provided a discretionary matching contribution of $362,187 and $337,632, respectively.
F-11
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
Income Taxes
The Company recognizes deferred tax assets and liabilities for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using statutory tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in the period that includes the statutory enactment date. Valuation allowances are established to reduce deferred tax assets when it is more likely than not that some portion or all of the deferred tax assets will not be realized.
Tax benefits are recognized only for tax positions that are more likely than not to be sustained upon examination by tax authorities. The amount recognized is measured as the largest amount of benefit that is greater than 50% likely to be realized upon settlement. A liability for unrecognized tax benefits is recorded for any tax benefits claimed in the Company’s tax returns that do not meet these recognition and measurement standards.
The Company’s policy for recording interest and penalties associated with uncertain tax positions is to record such items as a component of tax expense. No material amounts of tax-related interest or penalties were recorded during the years ended December 31, 2017 and 2018.
Stock-Based Compensation
Stock-based compensation expense for equity instruments issued to employees is measured based on the grant-date fair value of the awards. The fair value of each employee stock option is estimated on the date of grant using the Black-Scholes option-pricing valuation model. The Company recognizes compensation costs on a straight-line basis for all employee stock-based compensation awards over the requisite service period of the awards, which is generally the awards’ vesting period (typically four years). Forfeitures are accounted for as they occur.
Deferred Offering Costs
Deferred offering costs consist primarily of legal fees, which are direct and incremental fees related to the Company’s planned initial public offering (the “IPO”). The deferred offering costs will be offset against the IPO proceeds upon the consummation of the IPO. In the event the offering is aborted, the deferred offering costs will be expensed. There were no deferred offering costs as of December 31, 2017. As of December 31, 2018, the Company had incurred $91,307 in deferred offering costs, which are reported as other assets - long term on the balance sheet.
Comprehensive Loss
Comprehensive loss is defined as a change in equity during a period from transactions and other events and circumstances from non-owner sources. The Company’s comprehensive loss was the same as its reported net loss for all periods presented.
Unaudited Pro Forma Information
Unaudited pro forma basic and diluted loss per share attributable to common stockholders for the year ended December 31, 2018 has been computed to give effect to the assumed automatic conversion of all the Company’s outstanding shares of convertible preferred stock into shares of common stock and the net exercise of certain preferred stock warrants into shares of common stock as if such conversion and net exercise had occurred at the beginning of 2018, or the date of issuance, if later. The assumed net exercise of certain preferred stock warrants is associated with warrants to purchase shares of the Company’s Series F preferred stock for $0.01 per share that will become exercisable into shares of the Company’s common stock immediately prior to the closing of the IPO and which will otherwise expire upon the closing of the IPO if not exercised. The assumed net exercise of these preferred stock warrants is based on an IPO price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus). Shares of common stock to be issued in the IPO and shares issued upon conversion of the outstanding principal plus accrued interest related to the convertible promissory notes are excluded from the pro forma basic and diluted loss per share information. See Note 15, “Subsequent Events,” for information on the issuance of the convertible promissory notes in January and February 2019.
F-12
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
Recently Adopted Accounting Pronouncements
In May 2014, the FASB issued Accounting Standards Update (“ASU”) 2014-09, Revenues from Contracts with Customers (Topic 606). This guidance applies to any entity that either enters into contracts with customers to transfer goods or services or enters into contracts for the transfer of nonfinancial assets unless those contracts are within the scope of other standards. The core principle of this guidance is that an entity should recognize revenue to depict the transfer of promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. This guidance supersedes existing revenue recognition guidance, including most industry-specific guidance, as well as certain related guidance on accounting for contract costs. The Company early adopted ASC 606 on January 1, 2018 using the full retrospective method and adjusted the comparative reporting period for the year ended December 31, 2017. The cumulative impact on periods prior to January 1, 2017, which was not material, was recognized as a cumulative effect adjustment to the opening balance of accumulated deficit on January 1, 2017. In connection with the adoption of ASC 606, the Company elected the transition practical expedient that permitted the Company to use the transaction price at the date the transaction was completed, rather than estimating variable consideration amounts, in the 2017 comparative reporting period. For additional information and disclosures about the Company’s revenue recognition under ASC 606, see the Revenue Recognition section included above.
In August 2018, the FASB issued ASU 2018-13, Fair Value Measurement (Topic 820), – Disclosure Framework – Changes to the Disclosure Requirements for Fair Value Measurement (“ASU-2018-13”), which removes, modifies and adds certain disclosure requirements for fair value measurements. The Company’s early adoption of ASU 2018-13 on January 1, 2018 had no impact on the Company’s financial statements. Refer to Note 8 for additional information on fair value measurements.
Accounting Pronouncements Yet to be Adopted
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842), which supersedes FASB ASC Topic 840, Leases (Topic 840), and provides principles for the recognition, measurement, presentation and disclosure of leases for both lessees and lessors. The new standard requires lessees to apply a dual approach, classifying leases as either finance or operating leases based on the principle of whether or not the lease is effectively a financed purchase by the lessee. This classification will determine whether lease expense is recognized based on an effective interest method for finance leases or on a straight-line basis over the term of the lease for operating leases. A lessee is also required to record a right-of-use asset and a lease liability for all leases with a term of greater than 12 months regardless of classification. Leases with a term of 12 months or less will be accounted for similar to existing guidance for operating leases. For companies that are not emerging growth companies (“EGCs”), the ASU is effective for fiscal years beginning after December 15, 2018. For EGCs, the ASU is effective for fiscal years beginning after December 15, 2019. The Company will adopt the new standard using the modified retrospective method, under which the Company will apply Topic 842 to existing and new leases as of January 1, 2020, but prior periods will not be restated and will continue to be reported under Topic 840 guidance in effect during those periods. The Company anticipates that the adoption will not have a material impact on its statements of operations and comprehensive loss or its statements of cash flows but expects to recognize right-of-use assets and liabilities for lease obligations associated with its operating leases. The Company’s operating lease arrangements are discussed in Note 9.
In June 2016, the FASB issued ASU No. 2016-13, Financial Instruments–Credit Losses, which requires the measurement of expected credit losses for financial instruments carried at amortized cost, such as accounts receivable, held at the reporting date based on historical experience, current conditions and reasonable forecasts. The main objective of this ASU is to provide financial statement users with more decision-useful information about the expected credit losses on financial instruments and other commitments to extend credit held by a reporting entity at each reporting date. In November 2018, the FASB issued ASU No. 2018-19, Codification Improvements to Topic 326, Financing Instruments–Credit Losses, which included an amendment of the effective date for nonpublic entities. For non-EGCs, the ASU is effective for fiscal years beginning after December 15, 2019. For EGCs, the standard is effective for fiscal years beginning after December 15, 2021. The Company does not believe the adoption of this standard will have a significant impact on its financial statements, given its history of minimal bad debt expense relating to trade accounts receivable.
F-13
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
3. Loss per share
Loss per share is computed by dividing net loss attributable to common stockholders for the period by the weighted-average number of common shares outstanding during the period. Diluted loss per share reflects the additional dilution from potential issuances of common stock, such as stock issuable pursuant to the exercise of stock options, as well as from the possible conversion of the Company’s convertible preferred stock and exercise of outstanding warrants. The treasury stock and if-converted methods are used to calculate the potential dilutive effect of these common stock equivalents. However, potentially dilutive shares are excluded from the computation of diluted loss per share when their effect is antidilutive. Due to the Company reporting a net loss attributable to common stockholders for all periods presented, all potentially dilutive securities were antidilutive and have been excluded from the computation of diluted loss per share.
The table below provides potentially dilutive securities not included in the Company’s calculation of diluted loss per share because to do so would be antidilutive:
As of December 31, |
||||||
2017 |
2018 |
|||||
Shares issuable upon conversion of Series A convertible preferred stock |
533,711 | 533,711 | ||||
Shares issuable upon conversion of Series B convertible preferred stock |
816,654 | 816,654 | ||||
Shares issuable upon conversion of Series C convertible preferred stock |
503,056 | 503,056 | ||||
Shares issuable upon conversion of Series D convertible preferred stock |
756,416 | 756,416 | ||||
Shares issuable upon conversion of Series E-1 convertible preferred stock |
826,392 | 829,642 | ||||
Shares issuable upon conversion of Series E-2 convertible preferred stock |
934,433 | 934,433 | ||||
Shares issuable upon conversion of Series E-2A convertible preferred stock |
27,306 | 27,306 | ||||
Shares issuable upon conversion of Series E-3 convertible preferred stock |
824,000 | 824,000 | ||||
Shares issuable upon conversion of Series F convertible preferred stock |
2,892,098 | 4,734,613 | ||||
Shares issuable upon exercise of stock options |
1,433,612 | 2,023,158 | ||||
Shares issuable upon exercise of preferred stock warrants |
119,707 | 172,217 | ||||
Total |
9,667,385 | 12,155,206 | ||||
Unaudited Pro Forma Loss Per Share Attributable to Common Stockholders
The calculation of unaudited pro forma basic and diluted loss per share attributable to common stockholders for the year ended December 31, 2018 is set forth in the table below:
Pro Forma |
|||
Year Ended December 31, 2018 |
|||
(unaudited) |
|||
Basic and Diluted Loss per Share |
|||
Numerator |
|||
Net loss attributable to common stockholders, as reported |
$ | (10,162,875 | ) |
Pro forma adjustment to reflect assumed conversion of convertible preferred stock, cumulative dividends |
3,576,743 | ||
Pro forma adjustment to reflect assumed conversion of redeemable convertible preferred stock, accretion to redemption value |
219,138 | ||
Pro forma adjustment to remove change in fair value of preferred stock warrants, net of tax |
272,417 | ||
Pro forma net loss attributable to common stockholders |
$ | (6,094,577 | ) |
F-14
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
Pro Forma |
|||
Year Ended December 31, 2018 |
|||
(unaudited) |
|||
Denominator |
|||
Weighted-average shares outstanding, basic and diluted |
2,323,197 | ||
Pro forma adjustment to reflect assumed conversion of convertible preferred stock |
9,562,627 | ||
Pro forma adjustment to reflect assumed net exercise of certain preferred stock warrants |
|||
Shares used to compute pro forma loss per share, basic and diluted |
|||
Pro forma loss per share, basic and diluted |
$ | ||
Pro forma diluted loss per share does not include outstanding stock options and preferred stock warrants (except for certain preferred stock warrants described in Note 2 under Unaudited Pro Forma Information) since the effect would be antidilutive due to the pro forma net loss attributable to common stockholders for the period.
4. Property and Equipment, Net
Property and equipment, net consisted of the following:
As of December 31, |
||||||
2017 |
2018 |
|||||
Lab equipment |
$ | 980,589 | $ | 1,153,210 | ||
Computer equipment |
510,976 | 629,437 | ||||
Leasehold improvements |
545,401 | 553,921 | ||||
Furniture and fixtures |
71,151 | 101,813 | ||||
Total |
2,108,117 | 2,438,381 | ||||
Less accumulated depreciation |
(638,466 | ) |
(909,385 | ) |
||
Property and equipment, net |
$ | 1,469,651 | $ | 1,528,996 | ||
Depreciation expense was recorded in the statements of operations and comprehensive loss as follows:
Years Ended December 31, |
||||||
2017 |
2018 |
|||||
Cost of sales |
$ | 189,073 | $ | 163,995 | ||
Research and development |
2,427 | 2,847 | ||||
Selling, general and administrative |
78,808 | 120,222 | ||||
Total |
$ | 270,308 | $ | 287,064 | ||
5. Intangible Assets, Net
Intangible assets consist of capitalized license costs as follows:
As of December 31, |
||||||||||||
2017 |
2018 |
|||||||||||
Value |
Weighted- Average Remaining Life (Years) |
Value |
Weighted- Average Remaining Life (Years) |
|||||||||
Licenses |
$ | 274,534 | 1 | $ | 274,534 | 0 | ||||||
Accumulated amortization |
(234,415 | ) |
(270,367 | ) |
||||||||
Intangible assets, net |
$ | 40,119 | $ | 4,167 | ||||||||
F-15
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
Amortization expense was $40,512 and $35,952, respectively, for the years ended December 31, 2017 and 2018, respectively.
6. Other Accrued Liabilities
Other accrued liabilities consisted of the following:
As of December 31, |
||||||
2017 |
2018 |
|||||
Accrued state income taxes |
$ | 9,731 | $ | 8,606 | ||
Accrued interest |
113,436 | 185,580 | ||||
Accrued royalties |
186,734 | 239,751 | ||||
Accrued service fees |
200,195 | 281,307 | ||||
Accrued lease payments |
3,877 | — | ||||
Other |
93,872 | — | ||||
$ | 607,845 | $ | 715,244 | |||
7. Long-Term Debt
The Company’s long-term debt consists of term debt and a revolving line of credit and are presented in the table below:
As of December 31, |
||||||
2017 |
2018 |
|||||
Term debt |
$ | 16,012,500 | $ | 21,350,000 | ||
Revolving line of credit |
4,000,000 | 5,000,000 | ||||
Total principal amount |
20,012,500 | 26,350,000 | ||||
Unamortized discount and issuance costs |
(1,409,814 | ) |
(1,850,248 | ) |
||
Total long-term debt |
18,602,686 | 24,499,752 | ||||
Less: Current portion of long-term debt |
1,000,000 | — | ||||
Long-term debt, less current portion |
$ | 17,602,686 | $ | 24,499,752 | ||
Future maturities of principal amounts on long-term debt as of December 31, 2018 are as follows:
Term Debt |
Revolving Line of Credit |
Total |
|||||||
Years Ending December 31, |
|||||||||
2019 |
$ | — | $ | — | $ | — | |||
2020 |
4,666,667 | 5,000,000 | 9,666,667 | ||||||
2021 |
8,000,000 | — | 8,000,000 | ||||||
2022 |
8,683,333 | — | 8,683,333 | ||||||
2023 |
— | — | — | ||||||
Thereafter |
— | — | — | ||||||
Total |
$ | 21,350,000 | $ | 5,000,000 | $ | 26,350,000 | |||
Term Debt
2017 Loan and Security Agreement
On March 31, 2017, the Company executed a Loan and Security Agreement (“2017 LSA”) with Oxford Finance LLC (“Oxford”), as collateral agent, and Silicon Valley Bank (“SVB”) with Oxford and SVB as equal syndicated lenders (collectively, the “Lenders”). The 2017 LSA provides a term loan of $15.0 million (the
F-16
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
“2017 Term Loan”) and a credit line of up to $5.0 million (discussed in the “Revolving Line of Credit” section below) and granted to the Lenders a priority first lien security interest in substantially all of the Company’s assets. The 2017 Term Loan of $15.0 million was made on the effective date of the agreement and was used to repay all outstanding obligations with respect to the Company’s previous lending arrangement. The maturity date of the 2017 Term Loan was to be April 1, 2021 (the “2017 Term Loan Maturity Date”).
The 2017 Term Loan bore interest at a floating per annum rate equal to the greater of 1) 7.25% and 2) 6.48% above the U.S. LIBOR rate and required the payment of interest only each month for the one-year period beginning with the date of initial funding of the loan. The applicable annual interest rate of the 2017 LSA on the effective date, and as of December 31, 2017, was 7.46% and 8.04%, respectively. Repayment of principal was to be made in 36 monthly installments of principal and interest beginning in the month following the one-year interest-only period. Or, provided that the Company achieved a stipulated revenue target for the trailing 12-month period ending January 31, 2018, 30 fixed monthly installments, beginning in the month following an 18-month interest-only period. Additionally, the 2017 LSA also included an obligation to make an additional final payment of 6.25% (6.75% in the case the stipulated revenue target was met) of the aggregate original principal amount of the 2017 Term Loan upon any prepayment or on the 2017 Term Loan Maturity Date. As the Company achieved the stipulated revenue target, repayment of principal was to be made in 30 fixed monthly installments beginning in the month following the 18-month interest-only period (i.e., November 1, 2018) and the additional final payment amount was to be 6.75% of the original principal amount, or $1,012,500. The final payment amount was being amortized as additional interest expense using the effective interest method over the term of the debt.
As stipulated in the 2017 LSA and concurrent with the funding of the 2017 Term Loan, the Company issued Series F preferred stock warrants to the Lenders which had an aggregate initial fair value of $382,597. In accordance with ASC 480-10, Distinguishing Liabilities from Equity (“ASC 480-10”), the warrants are liability-classified as the underlying to the warrant is a puttable security. The initial recognition of the warrant liability created a discount to the debt, which is being amortized over the debt term using the effective interest method. With respect to Oxford, the 2017 LSA was accounted for as a new borrowing. With respect to SVB, the 2017 LSA was accounted for as a modification of the Company’s previous lending arrangement with SVB, and therefore no extinguishment gain or loss was recognized.
2018 Loan and Security Agreement
On November 30, 2018 (the “Closing Date”), the Company entered into a new Loan and Security Agreement (the “2018 LSA”) with Oxford, as collateral agent, and Oxford and SVB as equal syndicated lenders. The 2018 LSA replaced the 2017 LSA and provides for a $20.0 million secured term loan credit facility (the “2018 Term Loan”) and a credit line of up to $5.0 million (discussed in the “Revolving Line of Credit” section below). The Company’s obligations under the 2018 LSA are secured by substantially all of its assets, excluding intellectual property and subject to certain other exceptions and limitations. The Company has the right to prepay the 2018 Term Loan in whole or in part at any time, subject to a prepayment fee of 2.50% if prepaid on or prior to November 30, 2019, 1.50% if prepaid after November 30, 2019 and on or prior to November 30, 2020, and 0.75% thereafter. Upon prepayment, the Company is also obligated to pay a non-refundable early termination fee of $496,785. Amounts prepaid or repaid under the 2018 Term Loan may not be reborrowed. The 2018 LSA contains a financial covenant that requires the Company to achieve a trailing six-month revenue target each month throughout the term of the agreement. As of December 31, 2018, the Company was in compliance with this covenant. In addition, the 2018 LSA contains customary conditions of borrowing, events of default and covenants, including covenants that restrict the Company’s ability to dispose of assets, merge with or acquire other entities, incur indebtedness and make distributions to holders of the Company’s capital stock. Should an event of default occur, including the occurrence of a material adverse change, the Company could be liable for immediate repayment of all obligations under the 2018 LSA.
The 2018 Term Loan bears interest at a floating rate equal to the greater of 1) 8.55% and 2) the 30-day U.S. LIBOR rate as reported in The Wall Street Journal on the last business day of the month that precedes the month in which the interest will accrue, plus 6.48%. The applicable interest rate on the 2018 Term Loan was 8.98% at December 31, 2018. Interest on the 2018 Term Loan is payable monthly in arrears. The Company is permitted to make interest-only payments on the 2018 Term Loan for the 18 months following the Closing Date. The principal is required to be repaid in 30 equal monthly installments beginning on June 1, 2020. All unpaid
F-17
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
principal and accrued and unpaid interest is due on November 1, 2022 (the “2018 Term Loan Maturity Date”). The Company is also obligated to make an additional final payment of 6.75% of the aggregate original principal amount, or $1,350,000, upon any prepayment or on the 2018 Term Loan Maturity Date. The final payment amount is being amortized as additional interest expense using the effective interest method over the term of the debt.
The 2018 Term Loan was fully funded on the Closing Date and the 2018 Revolving Line (defined and discussed below) was fully drawn upon on the Closing Date. Proceeds from the 2018 LSA were used to repay all outstanding obligations under the 2017 LSA and to provide working capital. As a condition of the loan, the Company issued Series F preferred stock warrants to the Lenders with an aggregate initial fair value of $158,000. In accordance with ASC 480-10, the warrants are liability-classified as the underlying to the warrant is a puttable security. The initial recognition of the warrant liability created a discount to the debt, which is being amortized over the debt term using the effective interest method. The 2018 LSA was accounted for as a modification of the previous lending arrangement with SVB and Oxford, and therefore no extinguishment gain or loss was recognized.
Revolving Line of Credit
In connection with the issuance of the 2017 Term Loan, the Company repaid $2.0 million that was drawn on the revolving line under the Company’s previous lending arrangement. In the 2017 LSA, the Lenders agreed to provide availability to the Company of up to $5.0 million of credit (the “2017 Revolving Line”), based on the application of an advance rate against either specified accounts receivable, or accounts receivable in the aggregate, contingent on the Company’s satisfaction of borrowing base eligibility requirements. Any outstanding credit advanced against the 2017 Revolving Line was to be due in full no later than March 31, 2019. The Company drew $4.0 million on the 2017 Revolving Line in June 2017 and drew the remaining $1.0 million balance in May 2018. The 2017 Revolving Line bore interest at a floating per annum rate equal to the greater of 1) 6.25% and 2) 5.48% above the U.S. LIBOR rate. The applicable interest rate on the 2017 Revolving Line at December 31, 2017 was 7.04%.
In the 2018 LSA, the Lenders continued to provide credit availability to the Company of up to $5.0 million (the “2018 Revolving Line”), with similar terms as the 2017 Revolving Line, except that 2018 Revolving Line is due in full no later than November 30, 2020. The applicable interest rate on the 2018 Revolving Line was 7.98% at December 31, 2018.
8. Fair Value Measurements
Fair value is defined as the price that would be received to sell an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market in an orderly transaction between market participants at the measurement date. The fair value hierarchy prioritizes the inputs to valuation techniques used in measuring fair value. There are three levels to the fair value hierarchy based on the reliability of inputs, as follows:
Level 1 – Observable inputs that reflect quoted prices (unadjusted) for identical assets or liabilities in active markets.
Level 2 – Inputs other than quoted prices included in Level 1 that are observable for the asset or liability, either directly or indirectly.
Level 3 – Unobservable inputs in which little or no market data exists, therefore requiring the Company to develop its own assumptions.
Financial instruments measured at fair value are classified in their entirety based on the lowest level of input that is significant to the fair value measurement. The Company’s assessment of the significance of a particular input to the fair value measurement in its entirety requires management to make judgments and consider factors specific to the asset or liability. The use of different assumptions and/or estimation methodologies may have a material effect on estimated fair values. Accordingly, the fair value estimates disclosed, or amounts recorded may not be indicative of the amount that the Company or holders of the instruments could realize in a current market exchange.
F-18
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
The table below provides information, by level within the fair value hierarchy, of the Company’s financial assets and liabilities that are accounted for at fair value on a recurring basis as of December 31, 2017 and 2018:
As of December 31, 2017 |
||||||||||||
Quoted Prices in Active Markets for Identical Items (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
Total |
|||||||||
Liabilities: |
||||||||||||
Preferred stock warrants |
$ | — | $ | — | $ | 524,694 | $ | 524,694 | ||||
As of December 31, 2018 |
||||||||||||
Quoted Prices in Active Markets for Identical Items (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
Total |
|||||||||
Liabilities: |
||||||||||||
Preferred stock warrants |
$ | — | $ | — | $ | 1,193,726 | $ | 1,193,726 | ||||
The following table discloses the summary of changes in the fair value of the Level 3 preferred stock warrant liability:
Balance, January 1, 2017 |
$ | 402,001 | |
Issuance of warrants |
382,597 | ||
Change in fair value included in net loss |
(163,288 | ) |
|
Exercised warrants |
(96,616 | ) |
|
Balance, December 31, 2017 |
524,694 | ||
Issuance of warrants |
631,320 | ||
Change in fair value included in net loss |
272,417 | ||
Exercised warrants |
(234,705 | ) |
|
Balance, December 31, 2018 |
$ | 1,193,726 |
The change in fair value of the preferred stock warrant liability is recorded as other income (expense), net on the statements of operations and comprehensive loss.
The fair value of the warrants for shares of Series A, Series B, Series E-1, Series E-2, Series E-2A, Series E-3, and Series F redeemable convertible preferred stock was estimated by management using the Black-Scholes option pricing model with the following assumptions:
|
|
As of December 31,
|
|
|
|
2017
|
2018
|
|
Average expected life (years)
|
8
|
9
|
|
Expected stock price volatility
|
59.0% - 63.0%
|
64%
|
|
Risk-free interest rate
|
1.03% - 2.40%
|
2.51% - 2.69%
|
|
Dividend yield
|
0%
|
0%
|
9. Operating Leases
The Company leases its office headquarters in Friendswood, Texas (the “Friendswood Lease”). In December 2017, the Company entered into an amendment to the Friendswood Lease to rent additional office space within the facility. As of December 31, 2018, the Company’s commitment under the lease extends through June 30, 2019 and the Company has two one-year renewal options thereafter. If these options are exercised, the term of the lease would extend through June 30, 2021.
F-19
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
In April 2016, the Company took occupancy of a newly-constructed office and laboratory facility located in Phoenix, Arizona (the “Phoenix Lease”), replacing its previous facility. The term of the Phoenix Lease is through July 31, 2023.
Other leasing arrangements include office copiers and a corporate apartment.
All leases have been classified as operating leases. Rent expense is recognized on a straight-line basis over the term of the leases. Future minimum lease payments under all non-cancellable operating leases are as follows:
Years Ending December 31, |
|||
2019 |
$ | 294,230 | |
2020 |
277,647 | ||
2021 |
275,085 | ||
2022 |
266,943 | ||
2023 |
148,453 | ||
Thereafter |
— | ||
$ | 1,262,358 | ||
Rent expense was $229,040 and $314,868 for the years ended December 31, 2017 and 2018, respectively.
10. Commitments and Contingencies
From time to time, the Company may be involved in legal proceedings arising in the ordinary course of business. The Company believes there is no threatened litigation or litigation pending that could have, individually or in the aggregate, a material adverse effect on the Company’s financial position, results of operations or cash flows.
11. Stockholders’ Equity
Common Stock
The Company’s Amended and Restated Certificate of Incorporation, as amended on February 27, 2019, authorizes the Company to issue 17,308,384 shares of common stock with a par value of $0.001 per share. The holder of each share of common stock shall have one vote for each share of stock. The common stockholders are also entitled to receive dividends whenever funds and assets are legally available and when declared by the Board of Directors, subject to the prior rights of holders of all series of redeemable convertible preferred stock outstanding. No dividends were declared or paid during the years ended December 31, 2017 or 2018. The terms of the 2018 LSA restrict the Company’s ability to pay dividends on common stock with prior consent of the Lenders.
The Company amended its Amended and Restated Certificate of Incorporation on January 4, 2018 to reduce the number of shares of common stock that the Company is authorized to issue to 13,700,000. On May 31, 2018, the Company amended its Amended and Restated Certificate of Incorporation to increase the number of shares of common stock that the Company is authorized to issue to 14,602,000. On October 3, 2018, the Company amended its Amended and Restated Certificate of Incorporation to increase the number of shares of common stock that the Company is authorized to issue to 15,102,000. On January 31, 2019, the Company amended its Amended and Restated Certificate of Incorporation to increase the number of shares of common stock that the Company is authorized to issue to 16,957,416. On February 27, 2019, the Company amended its Amended and Restated Certificate of Incorporation to increase the number of shares of common stock that the Company is authorized to issue to 17,308,384. Certain of these amendments also included changes to the number of authorized preferred shares, as discussed in Note 12.
12. Convertible Preferred Stock and Preferred Stock Warrants
Convertible Preferred Stock
The Company’s convertible preferred stock is classified outside of stockholders’ equity in accordance with authoritative guidance for the classification and measurement of potentially redeemable securities. The preferred stock is contingently redeemable upon events that are outside of the Company’s control including liquidation,
F-20
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
sale, or transfer of control of the Company. The Company has determined not to adjust the carrying values of the convertible preferred stock to liquidation preferences of such shares because of the uncertainty of whether or when such events would occur. Further, except for Series C preferred stock, the preferred stock is redeemable upon the majority vote of specified holders of the preferred stock. Information on liquidation and redemption terms are discussed further under Liquidation Provisions and Redemption Rights, respectively, below.
The Company amended its Amended and Restated Certificate of Incorporation on January 4, 2018 to increase the authorized number shares of Series F redeemable convertible preferred stock to 3,981,548. On May 31, 2018, the Company amended its Amended and Restated Certificate of Incorporation to increase the authorized number of shares of Series F redeemable convertible preferred stock to 4,883,486. On January 31, 2019, the Company amended its Amended and Restated Certificate of Incorporation to increase the authorized number of shares of Series F redeemable convertible preferred stock to 6,738,902. On February 27, 2019, the Company amended its Amended and Restated Certificate of Incorporation to increase the authorized number of shares of Series F redeemable convertible preferred stock to 7,089,870.
On January 12, 2018, the Company issued 940,605 shares of Series F redeemable convertible preferred stock in exchange for $5.475 million in cash. In two subsequent closings on May 31, 2018 and June 8, 2018, the Company issued 783,248 and 85,711 additional shares, respectively, in exchange for $4,599,000 and $499,000, respectively, in cash. The designations, rights and preferences associated with the Series F redeemable convertible preferred stock are provided below. Proceeds of $93,872 from the sale of 16,127 of these shares were received prior to issuance at December 31, 2017 and these proceeds were reported as a liability on the December 31, 2017 balance sheet. In 2018, these proceeds were reclassified to preferred stock when the shares were ultimately issued. In conjunction with the January 12, 2018 issuance of the Series F redeemable convertible preferred shares, the Company also issued warrants to purchase 67,233 additional shares of redeemable convertible preferred stock at $0.01 per share with an initial fair value of $473,320.
Convertible preferred stock consists of the following:
Convertible preferred stock
As of December 31, 2017 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series C |
503,056 | 503,056 | $ | 3.4800 | $ | 1,750,635 | $ | 526,509 | $ | 2,277,144 | $ | 1,500,994 | |||||||||
Redeemable convertible preferred stock
As of December 31, 2017 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series A |
533,711 | 533,711 | $ | 2.1400 | $ | 1,142,142 | $ | 622,127 | $ | 1,764,269 | $ | 1,142,142 | |||||||||
Series B |
816,654 | 816,654 | $ | 2.2500 | 1,837,472 | 835,842 | 2,673,314 | 1,931,634 | |||||||||||||
Series D |
756,416 | 756,416 | $ | 3.7700 | 2,851,688 | 1,111,654 | 3,963,342 | 2,851,688 | |||||||||||||
Series E-1 |
842,641 | 826,392 | $ | 3.7700 | 3,115,498 | 845,367 | 3,960,865 | 3,079,416 | |||||||||||||
Series E-2 |
949,725 | 934,433 | $ | 4.5776 | 4,277,461 | 1,160,631 | 5,438,092 | 4,277,461 | |||||||||||||
Series E-2A |
27,306 | 27,306 | $ | 4.5776 | 124,996 | 28,738 | 153,734 | 124,377 | |||||||||||||
Series E-3 |
830,554 | 824,000 | $ | 5.3405 | 4,400,572 | 1,011,675 | 5,412,247 | 4,381,914 | |||||||||||||
Series F |
3,981,458 | 2,892,098 | $ | 5.8208 | 16,834,324 | 3,195,994 | 20,030,318 | 16,749,623 | |||||||||||||
Total |
8,738,465 | 7,611,010 | $ | 34,584,153 | $ | 8,812,028 | $ | 43,396,181 | $ | 34,538,255 | |||||||||||
F-21
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
Convertible preferred stock
As of December 31, 2018 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series C |
503,056 | 503,056 | $ | 3.4800 | $ | 1,750,635 | $ | 666,560 | $ | 2,417,195 | $ | 1,500,994 | |||||||||
Redeemable convertible preferred stock
As of December 31, 2018 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series A |
533,711 | 533,711 | $ | 2.1400 | $ | 1,142,142 | $ | 713,498 | $ | 1,855,640 | $ | 1,142,142 | |||||||||
Series B |
816,654 | 816,654 | $ | 2.2500 | 1,837,472 | 982,840 | 2,820,312 | 1,931,634 | |||||||||||||
Series D |
756,416 | 756,416 | $ | 3.7700 | 2,851,688 | 1,339,789 | 4,191,477 | 2,851,688 | |||||||||||||
Series E-1 |
842,641 | 829,642 | $ | 3.7700 | 3,127,750 | 1,095,281 | 4,223,031 | 3,098,578 | |||||||||||||
Series E-2 |
949,725 | 934,433 | $ | 4.5776 | 4,277,461 | 1,502,821 | 5,780,282 | 4,277,461 | |||||||||||||
Series E-2A |
27,306 | 27,306 | $ | 4.5776 | 124,996 | 38,738 | 163,734 | 124,762 | |||||||||||||
Series E-3 |
830,554 | 824,000 | $ | 5.3405 | 4,400,572 | 1,363,688 | 5,764,260 | 4,393,509 | |||||||||||||
Series F |
4,883,486 | 4,734,613 | $ | 5.8208 | 27,559,235 | 5,212,065 | 32,771,300 | 27,175,383 | |||||||||||||
Total |
9,640,493 | 9,456,775 | $ | 45,321,316 | $ | 12,248,720 | $ | 57,570,036 | $ | 44,995,157 | |||||||||||
Any discount to the original issue price is accreted with a charge to additional paid-in capital over the period through the earliest date the redeemable convertible preferred stock could become redeemable. See the Redemption Rights section below for additional information. Unpaid cumulative accruing dividends are included in calculating the aggregate liquidation preference, but such dividends are not recorded in the financial statements unless and until a liquidation event or a deemed liquidation event becomes probable of occurring. See the Liquidation Provisions section below for additional information.
The rights, preferences and privileges of the Series A, Series B, Series C, Series D, Series E-1, Series E-2, Series E-2A, Series E-3, and Series F convertible preferred stock are as follows:
Dividends
The holders of the outstanding shares of Series A, Series B, Series C, Series D, Series E-1, Series E-2, Series E-2A, Series E-3, and Series F convertible preferred stock are entitled to receive, when and if declared by the Board of Directors, a cash dividend in the following order of preference and at the rates per annum noted:
Convertible Preferred Stock Class |
Annual Dividend Rate |
||
Series A |
$ | 0.17120 | |
Series B |
$ | 0.18000 | |
Series C |
$ | 0.27840 | |
Series D |
$ | 0.30160 | |
Series E-1 |
$ | 0.30160 | |
Series E-2 |
$ | 0.36620 | |
Series E-2A |
$ | 0.36620 | |
Series E-3 |
$ | 0.42720 | |
Series F |
$ | 0.46570 | |
The right to receive dividends on shares of preferred stock shall not be cumulative, and no right to such dividends shall accrue to holders of preferred stock by reason of the fact that dividends on said shares are not
F-22
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
declared or paid in any calendar year. However, refer to Liquidation Provisions below for information regarding cumulative dividends that are payable upon a liquidation event or a deemed liquidation event.
Conversion Rights
Each share of Series A, Series B, Series C, Series D, Series E-1, Series E-2, Series E-2A, Series E-3, and Series F convertible preferred stock is, at the option of the holder, convertible at any time into that number of fully paid and non-assessable shares of common stock determined by dividing the original issue price by the conversion price. The conversion price for each series preferred stock shall initially be the original issue price of such series of preferred stock and shall be adjusted in accordance with the conversion provisions contained in the Company’s Amended and Restated Certificate of Incorporation. Each share of convertible preferred stock will automatically be converted into shares of common stock based on the then effective conversion price (i) upon the affirmative election of the holders of at least a majority of the outstanding shares of Series E-1, Series E-2, Series E-2A, Series E-3, and Series F convertible preferred stock or (ii) immediately upon the closing of a firmly underwritten public offering filed under the Securities Act of 1933, as amended, covering the offer and sale of common stock for the account of the Company in which the gross cash proceeds to the Company are at least $30 million.
Liquidation Provisions
Upon a liquidation event (any liquidation, dissolution or winding up on the Company, whether voluntary or involuntary) or a deemed liquidation event (generally, a change of control of the Company or the sale of substantially all the assets or intellectual property of the Company), the holders of Series A, Series B, Series C, Series D, Series E-1, Series E-2, Series E-2A, Series E-3, and Series F convertible preferred stock are entitled to distributions based on the liquidation value of the stock, which is initially set equal to the original issue price for the series of preferred stock, plus any unpaid cumulative accruing dividends, at the annual dividend rates provided above, accruing from the date of issuance of the shares of preferred stock, plus any other declared and unpaid dividends.
Regarding liquidation preferences, the holders of each successive series of preferred stock shall be entitled to receive on a pari passu basis and prior and in preference to any distribution of any of the assets or surplus funds of the Company to the holders of any prior-issued preferred stock, or common stock, the liquidation preference specified for each share of preferred stock.
Voting Rights
Each holder of preferred stock is entitled to the number of votes equal to the number of shares of common stock into which the shares of preferred stock could be converted. Holders of the preferred stock are entitled to vote on all matters on which the holders of common stock shall be entitled to vote.
Redemption Rights
The holders of at least a majority (voting together as a single class and not as a separate series, and on an as-converted basis) of the then outstanding shares of Series A preferred stock, Series B preferred stock, Series D preferred stock, Series E-1 preferred stock, Series E-2 preferred stock, Series E-2A preferred stock, Series E-3 preferred stock and Series F preferred stock (the “Redemption Stock”), and the holders of at least a majority (voting together as a single class and not as a separate series, and on an as-converted basis) of the then outstanding shares of Series E-1 preferred stock, Series E-2 preferred stock, Series E-2A preferred stock, Series E-3 preferred stock and Series F preferred stock may request, in writing, and any time after five years from the date of first issuance of Series F preferred stock (i.e., five years from July 15, 2015), the redemption of all outstanding shares of the Redemption Stock. The Company shall, upon such written request, redeem all outstanding shares in three equal annual installments beginning on the date specified in the redemption request, which date may not be less than 90 days after the Company’s receipt of such request. The redemption price shall be the original issue price of the preferred stock plus an amount for all declared and unpaid dividends thereon. As of December 31, 2017 and 2018, there were no declared and unpaid dividends; therefore the redemption value would have been equal to the original issue value if the preferred stock had been redeemable as of those dates. The foregoing redemption rights do not apply to Series C preferred stock.
F-23
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
Preferred Stock Warrant Liabilities
The Company has issued warrants to purchase shares of its convertible preferred stock. The warrant provides the holder the option to purchase a specified number of shares of a particular series of the Company’s convertible preferred stock for a specified price. The holder may exercise the warrant in cash or exercise pursuant to a cashless exercise whereby a calculated number of shares are withheld upon exercise to satisfy the exercise price. The warrants do not provide the holder any voting rights until the warrants are exercised. In the event of conversion of the Company’s convertible preferred stock to common shares, the warrants become exercisable into common shares of the Company’s stock, subject to certain adjustments.
In accordance with ASC 480-10, the Company accounts for preferred stock warrants outstanding as a liability at fair value and adjusts the instruments to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until exercised, and any change in fair value is recognized in the Company’s statements of operations and comprehensive loss as other income (expense), net. The fair value of the warrants issued by the Company has been estimated using a Black-Scholes model at each measurement date. Refer to Note 8 for additional information on the fair value measurement assumptions and changes in the fair value of the preferred stock warrants recorded on the Company’s balance sheets.
Warrants to purchase redeemable convertible preferred shares consist of the following:
As of December 31, 2017 |
|||||||||||||||||||
Warrants |
Year of First Issuance |
Shares |
Exercise Price |
Year of Expiration |
Exercised |
Expired |
Outstanding |
||||||||||||
Series A Preferred Stock |
2008 | 108,057 | $ | 2.1400 | 2015 |
(96,375 | ) |
(11,682 | ) |
— | |||||||||
Series B Preferred Stock |
2010 | 86,667 | $ | 2.2500 | 2017 |
(86,667 | ) |
— | — | ||||||||||
Series E-1 Preferred Stock |
2014 | 16,249 | $ | 3.7700 | 2024 |
— | — | 16,249 | |||||||||||
Series E-2 Preferred Stock |
2014 | 15,292 | $ | 4.5776 | 2024 |
— | — | 15,292 | |||||||||||
Series E-3 Preferred Stock |
2015 | 6,554 | $ | 5.3405 | 2024 |
— | — | 6,554 | |||||||||||
Series F Preferred Stock |
2016 | 81,612 | $ | 5.8208 | 2026-2028 |
— | — | 81,612 | |||||||||||
Total |
314,431 | (183,042 | ) |
(11,682 | ) |
119,707 | |||||||||||||
As of December 31, 2018 |
|||||||||||||||||||
Warrants |
Year of First Issuance |
Shares |
Exercise Price |
Year of Expiration |
Exercised |
Expired |
Outstanding |
||||||||||||
Series A Preferred Stock |
2008 | 108,057 | $ | 2.1400 | 2015 |
(96,375 | ) |
(11,682 | ) |
— | |||||||||
Series B Preferred Stock |
2010 | 86,667 | $ | 2.2500 | 2017 |
(86,667 | ) |
— | — | ||||||||||
Series E-1 Preferred Stock |
2014 | 16,249 | $ | 3.7700 | 2024 |
(3,250 | ) |
— | 12,999 | ||||||||||
Series E-2 Preferred Stock |
2014 | 15,292 | $ | 4.5776 | 2024 |
— | — | 15,292 | |||||||||||
Series E-3 Preferred Stock |
2015 | 6,554 | $ | 5.3405 | 2024 |
— | — | 6,554 | |||||||||||
Series F Preferred Stock |
2016 | 103,090 | $ | 5.8208 | 2026-2028 |
— | 103,090 | ||||||||||||
Series F Preferred Stock |
2018 | 67,233 | $ | 0.0100 | 2023 |
(32,951 | ) |
— | 34,282 | ||||||||||
Total |
403,142 | (219,243 | ) |
(11,682 | ) |
172,217 | |||||||||||||
13. Stock Incentive Plan and Stock Based Compensation
Stock Incentive Plan
On September 6, 2008, the Company adopted the 2008 Stock Plan (the “2008 Plan”). The 2008 Plan provides for the granting of options to purchase common stock to employees, directors and consultants of the Company. The Company may grant incentive stock options (“ISOs”), non-statutory stock options (“NSOs”) or restricted stock awards (“RSAs”) under the 2008 Plan. ISOs may only be granted to Company employees (including directors who are also considered employees). NSOs and RSAs may be granted to Company employees, directors and consultants. Options under the 2008 Plan may be granted for terms of up to ten years from the date of grant, as determined by the Board of Directors; provided, however, that with respect to an ISO granted to a person who owns stock representing more than 10% of the voting power of all classes of stock of the Company, the term
F-24
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
shall be for no more than five years from the date of grant. The exercise price of options granted under the 2008 Plan must be at a price no less than 100% of the estimated fair value of the shares on the date of grant, as determined by the Board of Directors, provided however, that with respect to an ISO granted to an employee who at the time of grant of such option owns stock representing more than 10% of the voting power of all classes of stock of the Company, the exercise price shall not be less than 110% of the estimated fair value of the shares on the date of grant.
Options granted under the 2008 Plan generally vest over four years (generally 25% after one year and monthly thereafter).
On August 15, 2018, the Company adopted the 2018 Stock Plan (the “2018 Plan”). Following the adoption of the 2018 Plan, no additional stock awards will be granted under the 2008 Plan; however, all stock awards granted under the 2008 Plan are subject to the terms of the 2008 Plan. Further, any shares that would have otherwise remained available for future grants under the 2008 Plan ceased to be available on the adoption date of the 2018 Plan and, instead, the number of shares of common stock equal to the 2008 Plan’s available reserve were added to the available reserve of the 2018 Plan, as will any shares issued under the 2008 Plan which expire, are forfeited or terminate prior to exercise or settlement.
Under the 2018 Plan, the Company may grant ISOs, NSOs or RSAs. ISOs may only be granted to Company employees (including directors who are also considered employees). NSOs and RSAs may be granted to Company employees, directors and consultants. Options under the 2018 Plan may be granted for terms of up to ten years from the date of grant, as determined by the Board of Directors, provided however, that with respect to an ISO granted to a person who owns stock representing more than 10% of the voting power of all classes of stock of the Company, the term shall be for no more than five years from the date of grant. The exercise price of options granted under the 2018 Plan must be at a price no less than 100% of the estimated fair value of the shares on the date of grant, as determined by the Board of Directors, provided however, that with respect to an ISO granted to an employee who at the time of grant of such option owns stock representing more than 10% of the voting power of all classes of stock of the Company, the exercise price shall not be less than 110% of the estimated fair value of the shares on the date of grant.
Activity under the Company’s 2008 and 2018 Plans for the years ended December 31, 2017 and 2018 is set forth below:
Weighted-Average |
|||||||||||||||
Shares Available for Grant |
Stock Options Outstanding |
Exercise Price |
Remaining Contractual Term (Years) |
Aggregate Intrinsic Value |
|||||||||||
Balance as of January 1, 2017 |
129,505 | 1,479,321 | $ | 1.42 | |||||||||||
Additional options authorized |
500,000 | — | |||||||||||||
Granted |
(130,750 | ) |
130,750 | $ | 1.93 | ||||||||||
Exercised |
— | (2,500 | ) |
$ | 1.42 | ||||||||||
Forfeited/Cancelled |
173,959 | (173,959 | ) |
$ | 1.51 | ||||||||||
Balance as of December 31, 2017 |
672,714 | 1,433,612 | $ | 1.46 | |||||||||||
Additional options authorized |
676,607 | — | |||||||||||||
Granted |
(707,545 | ) |
707,545 | $ | 1.96 | ||||||||||
Exercised |
— | (24,082 | ) |
$ | 1.57 | ||||||||||
Forfeited/Cancelled |
93,916 | (93,917 | ) |
$ | 1.49 | ||||||||||
Balance as of December 31, 2018 |
735,692 | 2,023,158 | $ | 1.63 | 6.54 | $ | 2,301,631 | ||||||||
Exercisable at December 31, 2018 |
1,120,515 | $ | 1.42 | 4.61 | $ | 1,508,822 | |||||||||
F-25
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
Determining Fair Value of Stock Options - Summary of Assumptions
The Company uses the Black-Scholes option pricing model to estimate the fair value of each option grant on the date of grant or any other measurement date. Following is a description of the significant assumptions used in the option pricing model:
| • | Expected term - The expected term is the period of time that granted options are expected to be outstanding. The Company has set the expected term using the simplified method based on the weighted average of both the period to vesting and the period to maturity for each option as the Company has concluded that its stock option exercise history does not provide a reasonable basis upon which to estimate the expected term. |
| • | Expected volatility - Because the Company’s stock is not traded in an active market, the Company calculates volatility by using the historical stock prices of a group of similar companies looking back over the estimated life of the option and averaging the volatilities of these companies. |
| • | Risk-free interest rate - The Company bases the risk-free interest rate used in the Black-Scholes option valuation model on the market yield in effect at the time of option grant provided from the Federal Reserve Board’s Statistical Releases and historical publications from the Treasury constant maturities rates for the equivalent remaining terms. |
| • | Dividend yield - The Company has not paid and does not have plans to pay cash dividends in the future. Therefore, the Company uses an expected dividend yield of zero in the Black-Scholes option valuation model. |
The following table sets forth the assumptions used to determine the fair value of stock options:
|
|
Years Ended December 31,
|
|
|
|
2017
|
2018
|
|
Average expected term
|
6 years
|
6 years
|
|
Expected stock price volatility
|
58.64% - 60.23%
|
57.77% - 58.29%
|
|
Risk-free interest rate
|
1.59% - 2.13%
|
2.77% - 3.13%
|
|
Dividend yield
|
0%
|
0%
|
Stock-based compensation expense related to stock options is included in the statements of operations and comprehensive loss as follows:
Years Ended December 31, |
||||||
2017 |
2018 |
|||||
Cost of sales |
$ | 18,277 | $ | 23,721 | ||
Research and development |
13,767 | 55,975 | ||||
Selling, general and administrative |
76,652 | 213,943 | ||||
Total stock-based compensation expense |
$ | 108,696 | $ | 293,639 | ||
No tax benefits related to stock-based compensation were recorded in the statements of operations and comprehensive loss during the years ended December 31, 2017 and 2018 due to the valuation allowance on net deferred tax assets. The weighted-average grant date fair value of options granted during the years ended December 31, 2017 and 2018 was $1.04 and $1.11 per option, respectively. As of December 31, 2018, there was $878,095 of total unrecognized compensation cost, which is expected to be recognized on a straight-line basis over a weighted-average period of 1.8 years. The total unrecognized compensation costs will be adjusted for forfeitures in future periods as they occur. The intrinsic value of stock options exercised and the related tax benefit associated with the tax deduction was not material for either of the years ended December 31, 2017 and 2018.
F-26
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
14. Income Taxes
The components of income tax expense are as follows:
Years Ended December 31, |
||||||
2017 |
2018 |
|||||
Current tax expense |
||||||
U.S. Federal |
$ | — | $ | — | ||
State and local |
9,731 | 8,606 | ||||
Total current |
9,731 | 8,606 | ||||
Deferred tax expense |
||||||
U.S. Federal |
— | — | ||||
State and local |
— | — | ||||
Total deferred |
— | — | ||||
Total income tax expense |
$ | 9,731 | $ | 8,606 | ||
The differences between income taxes expected at the U.S. federal statutory rate (34% for 2017, 21% for 2018) and the reported income tax expense are summarized as follows:
Years Ended December 31, |
||||||
2017 |
2018 |
|||||
Pre-tax loss |
$ | (12,359,798 | ) |
$ | (6,358,388 | ) |
U.S. federal taxes at statutory rate |
(4,202,332 | ) |
(1,335,261 | ) |
||
State income taxes |
(253,522 | ) |
(1,174,207 | ) |
||
Permanent differences |
160,042 | 235,979 | ||||
Change in corporate tax rate |
6,186,483 | — | ||||
Research and development (“R&D”) tax credit |
(215,281 | ) |
— | |||
Change in valuation allowance |
(1,670,809 | ) |
2,278,081 | |||
Other |
5,150 | 4,014 | ||||
Total income tax expense |
$ | 9,731 | $ | 8,606 | ||
Significant components of deferred tax assets and liabilities are as follows:
As of December 31, |
||||||
2017 |
2018 |
|||||
Deferred tax assets: |
||||||
Net operating loss (“NOL”) carryforwards |
$ | 11,564,168 | $ | 16,184,285 | ||
Accounts payable |
— | 389,822 | ||||
Accrued liabilities |
831,974 | 1,432,067 | ||||
R&D tax credit |
718,907 | 714,907 | ||||
Intangible assets |
55,386 | 60,723 | ||||
Stock-based compensation |
15,033 | 12,359 | ||||
Charitable contributions |
3,938 | 3,938 | ||||
Total deferred tax assets |
13,189,406 | 18,798,101 | ||||
Less valuation allowance |
(12,097,388 | ) |
(14,375,469 | ) |
||
Deferred tax assets, net |
1,092,018 | 4,422,632 | ||||
Deferred tax liabilities: |
||||||
Accounts receivable |
— | (3,248,520 | ) |
|||
Prepaid expenses |
— | (378,276 | ) |
|||
Property and equipment |
(138,680 | ) |
(130,905 | ) |
||
Section 481(a) adjustment (cash to accrual) |
(953,338 | ) |
(664,931 | ) |
||
Total deferred tax liabilities |
(1,092,018 | ) |
(4,422,632 | ) |
||
Net deferred tax asset (liability) |
$ | — | $ | — | ||
F-27
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
At December 31, 2017 and 2018, the Company had NOL carryforwards for federal income tax purposes of approximately $48,308,000 and $62,201,000, respectively, of which $48,308,000 will begin to expire in 2028 if not utilized to offset taxable income, and $13,893,000 may be carried forward indefinitely. Future changes in ownership, as defined by Section 382 of the IRC, could limit the amount of NOL carryforwards used in any one year. Also, as of December 31, 2017 and 2018, the Company had state net operating loss carryforwards of approximately $41,586,000 and $60,815,000, respectively, which begin to expire in 2028 if not utilized to offset state taxable income.
In general, under Section 382 and 383 of the IRC, a corporation that undergoes an “ownership change” is subject to limitations on its ability to utilize its pre-change NOLs and certain tax credits, to offset future taxable income and tax. In general, an ownership change occurs if the aggregate stock ownership of certain stockholders changes by more than 50 percentage points over such stockholders’ lowest percentage of ownership during the testing period (generally three years). The Company performed a Section 382 analysis from inception through the year ended December 31, 2017 and concluding the Company had experienced an ownership change in 2011 and 2014. These changes in ownership did not result in the expiration of any NOLs or R&D credits. However, future changes in ownership may further limit the ability of the Company to utilize its NOL carryforwards and R&D tax credit carryforwards.
At December 31, 2017 and 2018, the Company placed a valuation allowance of $12,097,388 and $14,375,469, respectively, against the entirety of its net deferred tax asset balance, as the Company has not determined that it is more likely than not to be realized.
The Company assessed whether it had any significant uncertain tax positions related to open tax years and concluded there were none. Accordingly, no reserve for uncertain tax positions has been recorded as of December 31, 2017 and 2018. The Company is generally no longer subject to tax examinations for U.S. federal income tax purposes for fiscal years prior to 2015 and fiscal years prior to 2014 for multiple state jurisdictions. However, since the Company has been in an NOL position since 2008, the Company’s 2008 to 2013 tax returns are potentially subject to examination adjustments to the extent of those NOL carryforwards.
On December 22, 2017, the 2017 Tax Cuts and Jobs Act (the “TCJA”) was signed into law. The TCJA significantly changes U.S. tax law by, among other things, lowering the U.S. corporate income tax rate from a maximum of 35% to a flat 21% rate, effective January 1, 2018. As a result of the decrease in the corporate income tax rate, the Company revalued its ending net deferred tax assets at December 31, 2017 but did not recognize any incremental income tax expense (benefit) in 2017 as the impacts of the change were fully offset by changes in the Company’s valuation allowance on deferred tax assets.
On December 22, 2017, the Securities and Exchange Commission issued Staff Accounting Bulletin No. 118 (“SAB 118”) to address the application of GAAP in situations when a registrant does not have the necessary information available, prepared or analyzed (including computations) in reasonable detail to complete the accounting for certain income tax effects of the TCJA. SAB 118 provides registrants up to one year to after the enactment date of the TCJA to finalize the recording of the related tax impacts. The Company’s accounting for the impacts of the TCJA are complete as of December 31, 2018. No adjustments under SAB 118 were recorded during the year ended December 31, 2018.
15. Subsequent Events
On January 13, 2019, the Company’s Board of Directors approved the issuance of unsecured convertible promissory notes (the “Notes”) in a financing transaction. The initial closing of the issuance of the Notes occurred in January 2019 and the Company issued notes to 54 purchasers for an aggregate of approximately $8.1 million in gross proceeds. In February 2019, the Company issued notes to an additional 11 purchasers for an aggregate of approximately $3.7 million in gross proceeds. Of the aggregate proceeds of $11.8 million, $4.8 million was received from related parties. The Notes bear simple interest at a rate of 8% per annum with a maturity date of January 31, 2020. On or before the maturity date, the entire outstanding principal amount of and accrued interest on the Notes (the “Conversion Amount”), is automatically convertible into shares of the Company’s equity securities issued and sold in a single or series of related transactions, with the principal purpose of raising capital, in which the Company sells shares of such equity securities for aggregate gross proceeds of at least $10.0 million (the “Next Equity Financing”). The number of shares of such equity securities
F-28
CASTLE BIOSCIENCES, INC.
NOTES TO FINANCIAL STATEMENTS (CONTINUED)
issuable in the Next Equity Financing is equal to the quotient of the Conversion Amount as of the closing date of the Next Equity Financing divided by a per share price that is equal to 80% of the lowest per share purchase price of the equity securities sold in the Next Equity Financing. If the Notes have not been repaid or converted prior to the maturity date, then, at the request of the holders of a majority of the then-outstanding principal amount of and accrued interest on the Notes, then the Conversion Amount as of the maturity date will convert into shares of the Company’s Series F redeemable convertible preferred stock, or any senior equity security issued by the Company after the first issuance of the Notes, in each case at a conversion price equal to the price at which such security was last sold (currently $5.8208 for shares of the Company’s Series F redeemable convertible preferred stock). If a change of control of the Company occurs while the Notes are outstanding, the Company is required to repurchase each Note from each holder at a repurchase price equal to two times the principal amount of such Note, plus any accrued and unpaid interest on such Note as of the date of such repurchase.
On January 31, 2019 and February 27, 2019, the Company amended its Amended and Restated Certificate of Incorporation to increase the number of authorized shares of common stock and preferred stock, as discussed in Notes 11 and 12.
On March 13, 2019, the Company’s Board of Directors approved a grant of 510,000 options to purchase shares of common stock of the Company as ISOs. The per share exercise price is $2.77 per share, which the Board believes is equal to the fair market value of one share of common stock as of that date. The options shall have a term beginning on the grant date and ending on the date immediately preceding the ten-year anniversary of the grant date. The options are initially subject to vesting. On the first anniversary of the vesting commencement date, 1/4th of the shares subject to the option shall vest; thereafter 1/48th of the shares subject to the option shall vest at the end of each one-month of anniversary of vesting commencement date, provided in each case that the optionee is then providing services to the Company in accordance with the terms of the 2018 Plan.
For the purposes of the financial statements as of December 31, 2018 and the year then ended, the Company has evaluated the subsequent events through May 13, 2019, the date the audited annual financial statements were issued.
F-29
CASTLE BIOSCIENCES, INC.
CONDENSED BALANCE SHEETS
December 31, 2018 |
March 31, 2019 |
Pro Forma As of March 31, 2019 |
|||||||
(unaudited) |
(unaudited) |
||||||||
ASSETS |
|||||||||
Current Assets |
|||||||||
Cash and cash equivalents |
$ | 4,478,512 | $ | 16,230,647 | |||||
Accounts receivable, net |
12,089,719 | 9,229,423 | |||||||
Inventory |
882,233 | 809,297 | |||||||
Prepaid expenses and other current assets |
675,562 | 741,423 | |||||||
Total current assets |
18,126,026 | 27,010,790 | |||||||
Long-term accounts receivable, net |
2,532,011 | 1,551,034 | |||||||
Property and equipment, net |
1,528,996 | 1,719,557 | |||||||
Intangible assets, net |
4,167 | 1,667 | |||||||
Other assets – long-term |
213,735 | 682,585 | |||||||
Total assets |
$ | 22,404,935 | $ | 30,965,633 | |||||
LIABILITIES, CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ DEFICIT |
|||||||||
Current Liabilities |
|||||||||
Accounts payable |
$ | 1,450,766 | $ | 1,609,458 | |||||
Accrued compensation |
4,571,011 | 2,761,765 | |||||||
Other accrued liabilities |
715,244 | 1,036,222 | |||||||
Convertible promissory notes (including $4,755,882 principal amount with related parties as of March 31, 2019) (Note 7) |
— | 3,568,136 | |||||||
Total current liabilities |
6,737,021 | 8,975,581 | |||||||
Long-term debt |
24,499,752 | 23,632,316 | |||||||
Preferred stock warrant liability |
1,193,726 | 1,180,569 | |||||||
Deferred rent liability |
43,587 | 57,372 | |||||||
Total liabilities |
32,474,086 | 33,845,838 | |||||||
Commitments and Contingencies (Note 10) |
|||||||||
Convertible Preferred Stock |
|||||||||
Convertible preferred stock Series C, $0.001 par value, 503,056 shares authorized as of December 31, 2018 and March 31, 2019; 503,056 shares issued and outstanding as of December 31, 2018 and March 31, 2019; $2,417,195 and $2,451,728 aggregate liquidation preference as of December 31, 2018 and March 31, 2019, respectively; no shares issued and outstanding as of March 31, 2019, pro forma |
1,500,994 | 1,500,994 | |||||||
Redeemable convertible preferred stock Series A, B, D, E-1, E-2, E-2A, E-3 and F, $0.001 par value, 9,640,493 and 11,846,877 shares authorized as of December 31, 2018 and March 31, 2019, respectively; 9,456,775 shares issued and outstanding as of December 31, 2018 and March 31, 2019; $57,570,036 and $58,464,077 aggregate liquidation preference as of December 31, 2018 and March 31, 2019, respectively; no shares issued and outstanding as of March 31, 2019, pro forma |
44,995,157 | 45,050,955 | |||||||
Stockholders’ Deficit |
|||||||||
Common stock, $0.001 par value; 15,102,000 and 17,308,384 shares authorized as of December 31, 2018 and March 31, 2019, respectively; 2,335,880 and 2,336,463 shares issued and outstanding as of December 31, 2018 and March 31, 2019, respectively; shares issued and outstanding as of March 31, 2019, pro forma |
2,336 | 2,337 | |||||||
Additional paid-in capital |
920,940 | 9,412,070 | |||||||
Accumulated deficit |
(57,488,578 | ) |
(58,846,561 | ) |
|||||
Total stockholders’ deficit |
(56,565,302 | ) |
(49,432,154 | ) |
|||||
Total liabilities, convertible preferred stock and stockholders’ deficit |
$ | 22,404,935 | $ | 30,965,633 | |||||
The accompanying notes are an integral part of these unaudited condensed financial statements.
F-30
CASTLE BIOSCIENCES, INC.
CONDENSED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(UNAUDITED)
Three Months Ended March 31, |
||||||
2018 |
2019 |
|||||
NET REVENUES |
$ | 3,658,565 | $ | 8,716,987 | ||
COST OF SALES |
1,252,983 | 1,597,958 | ||||
Gross margin |
2,405,582 | 7,119,029 | ||||
OPERATING EXPENSES |
||||||
Research and development |
1,262,690 | 1,393,851 | ||||
Selling, general and administrative |
4,227,791 | 6,046,622 | ||||
Total operating expenses |
5,490,481 | 7,440,473 | ||||
Operating loss |
(3,084,899 | ) |
(321,444 | ) |
||
Interest income |
5,383 | 20,789 | ||||
Interest expense |
(530,225 | ) |
(1,024,400 | ) |
||
Other income (expense), net |
(20,698 | ) |
(32,928 | ) |
||
Loss before income taxes |
(3,630,439 | ) |
(1,357,983 | ) |
||
Income tax expense |
— | — | ||||
Net loss and comprehensive loss |
(3,630,439 | ) |
(1,357,983 | ) |
||
Convertible preferred stock cumulative dividends |
809,322 | 928,575 | ||||
Accretion of redeemable convertible preferred stock to redemption value |
49,225 | 55,798 | ||||
Net loss and comprehensive loss attributable to common stockholders |
$ | (4,488,986 | ) |
$ | (2,342,356 | ) |
Loss per share attributable to common stockholders, basic and diluted |
$ | (1.94 | ) |
$ | (1.00 | ) |
Weighted-average shares outstanding, basic and diluted |
2,312,515 | 2,336,236 | ||||
Pro forma loss per share, basic and diluted |
$ | |||||
Pro forma weighted-average shares outstanding, basic and diluted |
||||||
The accompanying notes are an integral part of these unaudited condensed financial statements.
F-31
CASTLE BIOSCIENCES, INC.
CONDENSED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND STOCKHOLDERS’ DEFICIT
(UNAUDITED)
Convertible Preferred Stock Series C |
Redeemable Convertible Preferred Stock Series A, B, D, E-1, E-2, E-2A, E-3 and F |
Common Stock |
Additional Paid-in Capital |
Accumulated Deficit |
Total Stockholders’ Deficit |
||||||||||||||||||||||
Shares |
Amount |
Shares |
Amount |
Shares |
Amount |
||||||||||||||||||||||
BALANCE, JANUARY 1, 2018 |
503,056 | $ | 1,500,994 | 7,611,010 | $ | 34,538,255 | 2,311,798 | $ | 2,312 | $ | 808,767 | $ | (51,121,584 | ) |
$ | (50,310,505 | ) |
||||||||||
Stock compensation expense |
— | — | — | — | — | — | 61,707 | — | 61,707 | ||||||||||||||||||
Exercise of common stock options |
— | — | — | — | 750 | 1 | 1,064 | — | 1,065 | ||||||||||||||||||
Issuance of Series F redeemable convertible preferred stock |
— | — | 940,605 | 4,967,890 | — | — | — | — | — | ||||||||||||||||||
Accretion of redeemable convertible preferred stock to redemption value: |
|||||||||||||||||||||||||||
Series E-1 |
— | — | — | 1,030 | — | — | (1,030 | ) |
— | (1,030 | ) |
||||||||||||||||
Series E-2A |
— | — | — | 95 | — | — | (95 | ) |
— | (95 | ) |
||||||||||||||||
Series E-3 |
— | − | — | 2,855 | — | — | (2,855 | ) |
— | (2,855 | ) |
||||||||||||||||
Series F |
— | — | — | 45,245 | — | — | (45,245 | ) |
— | (45,245 | ) |
||||||||||||||||
Exercise of Series F redeemable convertible preferred stock warrants |
— | — | 8,279 | 58,367 | — | — | — | — | — | ||||||||||||||||||
Net loss |
— | — | — | — | — | — | — | (3,630,439 | ) |
(3,630,439 | ) |
||||||||||||||||
BALANCE, MARCH 31, 2018 |
503,056 | $ | 1,500,994 | 8,559,894 | $ | 39,613,737 | 2,312,548 | $ | 2,313 | $ | 822,313 | $ | (54,752,023 | ) |
$ | (53,927,397 | ) |
||||||||||
BALANCE, JANUARY 1, 2019 |
503,056 | $ | 1,500,994 | 9,456,775 | $ | 44,995,157 | 2,335,880 | $ | 2,336 | $ | 920,940 | $ | (57,488,578 | ) |
$ | (56,565,302 | ) |
||||||||||
Stock compensation expense |
— | — | — | — | — | — | 168,421 | — | 168,421 | ||||||||||||||||||
Exercise of common stock options |
— | — | — | — | 583 | 1 | 915 | — | 916 | ||||||||||||||||||
Accretion of redeemable convertible preferred stock to redemption value: |
|||||||||||||||||||||||||||
Series E-1 |
— | — | — | 1,031 | — | — | (1,031 | ) |
— | (1,031 | ) |
||||||||||||||||
Series E-2A |
— | — | — | 95 | — | — | (95 | ) |
— | (95 | ) |
||||||||||||||||
Series E-3 |
— | − | — | 2,863 | — | — | (2,863 | ) |
— | (2,863 | ) |
||||||||||||||||
Series F |
— | — | — | 51,809 | — | — | (51,809 | ) |
— | (51,809 | ) |
||||||||||||||||
Recognition of beneficial conversion feature on convertible promissory notes |
— | — | — | — | — | — | 8,377,592 | — | 8,377,592 | ||||||||||||||||||
Net loss |
— | — | — | — | — | — | — | (1,357,983 | ) |
(1,357,983 | ) |
||||||||||||||||
BALANCE, MARCH 31, 2019 |
503,056 | $ | 1,500,994 | 9,456,775 | $ | 45,050,955 | 2,336,463 | $ | 2,337 | $ | 9,412,070 | $ | (58,846,561 | ) |
$ | (49,432,154 | ) |
||||||||||
The accompanying notes are an integral part of these unaudited condensed financial statements.
F-32
CASTLE BIOSCIENCES, INC.
CONDENSED STATEMENTS OF CASH FLOWS
(UNAUDITED)
Three Months Ended March 31, |
||||||
2018 |
2019 |
|||||
OPERATING ACTIVITIES |
||||||
Net loss |
$ | (3,630,439 | ) |
$ | (1,357,983 | ) |
Adjustments to reconcile net loss to net cash (used in) provided by operating activities: |
||||||
Depreciation |
70,413 | 77,420 | ||||
Stock compensation expense |
61,707 | 168,421 | ||||
Amortization of intangibles |
9,337 | 2,500 | ||||
Amortization of debt discounts and issuance costs |
145,197 | 334,327 | ||||
Change in fair value of preferred stock warrant liability |
20,698 | (13,157 | ) |
|||
Change in fair value of embedded derivative |
— | 46,085 | ||||
Change in operating assets and liabilities: |
||||||
Accounts receivable |
(37,990 | ) |
3,841,273 | |||
Prepaid expenses and other current assets |
(98,951 | ) |
(65,861 | ) |
||
Inventory |
97,035 | 72,936 | ||||
Other assets |
35,224 | (14,181 | ) |
|||
Accounts payable |
(217,193 | ) |
(328,835 | ) |
||
Accrued compensation |
1,014,079 | (1,809,246 | ) |
|||
Other accrued liabilities |
(6,410 | ) |
320,978 | |||
Deferred rent liability |
28,475 | 13,785 | ||||
Net cash (used in) provided by operating activities |
(2,508,818 | ) |
1,288,462 | |||
INVESTING ACTIVITES |
||||||
Purchases of property and equipment |
(28,584 | ) |
(243,623 | ) |
||
Net cash used in investing activities |
(28,584 | ) |
(243,623 | ) |
||
FINANCING ACTIVITIES |
||||||
Proceeds from issuance of preferred stock and preferred stock warrants (including exercised warrants) |
5,347,421 | — | ||||
Proceeds from issuance from convertible promissory notes (including $4,755,882 from related parties), net of issuance costs |
— | 11,695,495 | ||||
Repayments on line of credit |
— | (989,115 | ) |
|||
Proceeds from exercise of common stock options |
1,065 | 916 | ||||
Net cash provided by financing activities |
5,348,486 | 10,707,296 | ||||
NET CHANGE IN CASH AND CASH EQUIVALENTS |
2,811,084 | 11,752,135 | ||||
Beginning of year |
1,212,063 | 4,478,512 | ||||
End of year |
$ | 4,023,147 | $ | 16,230,647 | ||
SUPPLEMENTAL DISCLOSURE OF CASH PAID (REFUNDED) FOR: |
||||||
Interest |
$ | 453,492 | $ | 572,449 | ||
Income taxes |
$ | — | $ | (150,000 | ) |
|
DISCLOSURE OF NON-CASH INVESTING AND FINANCING ACTIVITIES: |
||||||
Accrued capital expenditures |
$ | 14,356 | $ | 82,756 | ||
Deferred offering costs incurred but not paid |
$ | — | $ | 463,170 | ||
The accompanying notes are an integral part of these unaudited condensed financial statements.
F-33
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS
(UNAUDITED)
1. Organization and Description of Business
Castle Biosciences, Inc. (the “Company”) was incorporated in the state of Delaware on September 12, 2007. The Company is a commercial stage dermatological cancer company focused on providing physicians and their patients with personalized, clinically actionable genomic information to make more accurate treatment decisions. The Company is based in Friendswood, Texas (a suburb of Houston, Texas) and laboratory operations are conducted in the Company’s facilities located in Phoenix, Arizona.
Going Concern, Liquidity and Capital Resources
The Company has incurred significant operating losses since its inception. As of December 31, 2018 and March 31, 2019, the Company had an accumulated deficit of $57.5 million and $58.8 million, respectively, cash and cash equivalents totaling $4.5 million and $16.2 million, respectively, and working capital of $11.4 million and $18.0 million, respectively. The Company also has substantial indebtedness, the terms of which required it to meet a monthly six-month trailing revenue covenant. At the time of issuance of the Company’s financial statements as of December 31, 2018 and 2017 and for the years then ended, the Company disclosed that substantial doubt was raised about the Company ability to continue as a going concern, because its projections indicated potential non-compliance with this covenant during the next 12 months. As discussed in Note 8, “Long-Term Debt,” on June 13, 2019, the Company entered into an amendment to its bank debt agreement, which among other changes, modified the revenue covenant from a trailing six-month calculation to a trailing three-month calculation with revised revenue targets. As a result, management now expects to be in compliance with the amended covenant during the next 12 months. The Company has financed its operations to date primarily through private placements of its preferred stock, the sale of its prognostic testing services, bank debt borrowings and the issuance of convertible promissory notes. The ability of the Company to fund operations according to plan is largely dependent on its ability to improve the timing and amount of collections from the sale of tests; however, as the attainment of managed care coverage policy decisions is ordinarily a protracted process, funding from other sources will likely be necessary.
While the Company intends to substantially fund planned operations for the next 12 months using cash on hand and collections from test sales, management expects that a net usage of cash will continue to occur through 2019 and, consequently, additional capital from other sources will be required. As discussed in Note 7, “Convertible Promissory Notes,” the Company secured $11.8 million in financing through the sale of unsecured convertible notes. Additional equity and debt capital are believed by management to be available and funding arrangements are actively being sought as of the date of these financial statements. In addition to the procurement of capital to fund operations, management has identified discretionary spending included in the Company’s operating plan that can be deferred until prudently effectuated.
2. Summary of Significant Accounting Policies
Basis of Presentation
The Company’s financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”). The Company has no subsidiaries and all operations are conducted by the Company.
Unaudited Interim Financial Information
The accompanying balance sheet as of March 31, 2019, the statements of operations and comprehensive loss, the statements of convertible preferred stock and stockholders’ deficit and statements of cash flows for the three months ended March 31, 2018 and 2019 are unaudited. The unaudited interim financial statements have been prepared on the same basis as the audited annual financial statements and, in the opinion of management, reflect all adjustments, which include only normal recurring adjustments, necessary for the fair statement of the Company’s financial position as of March 31, 2019 and the results of its operations and its cash flows for the three months ended March 31, 2018 and 2019. The financial data and other information disclosed in these notes related to the three months ended March 31, 2018 and 2019 are also unaudited. The results for the three months
F-34
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
ended March 31, 2019 are not necessarily indicative of results to be expected for the year ending December 31, 2019, any other interim periods, or any future year or period. The balance sheet as of December 31, 2018 included herein was derived from the audited financial statements as of that date. Certain disclosures have been condensed or omitted from the interim financial statements. These financial statements should be read in conjunction with the Company’s audited financial statements included elsewhere in this prospectus.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities as of the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Significant items subject to such estimates include revenue recognition, the determination of fair value of the Company’s preferred stock warrants, the valuation of stock options, assessing future tax exposure and the realization of deferred tax assets, the useful lives and recoverability of property and equipment, and contingent liabilities. The Company bases these estimates on historical and anticipated results, trends, and various other assumptions that the Company believes are reasonable under the circumstances, including assumptions as to future events. These estimates form the basis for making judgments about the carrying values of assets and liabilities and recorded revenues and expenses that are not readily apparent from other sources. Actual results could differ from those estimates and assumptions.
Operating Segments
Operating segments are identified as components of an enterprise about which separate discrete financial information is available for evaluation by the chief operating decision maker, or decision-making group, in making decisions on how to allocate resources and assess performance. The Company views its operations and manages its business as one operating segment. All revenues are attributable to U.S.-based operations and all assets are held in the United States.
Cash and Cash Equivalents including Concentrations of Credit Risk
Cash equivalents consist of short-term, highly liquid investments with original maturities of three months or less. Cash equivalents consist primarily of amounts invested in money market accounts. A majority of the Company’s cash and cash equivalents are deposited with a single financial institution. Deposits in this institution may exceed the amount of insurance provided on such deposits by the Federal Deposit Insurance Corporation for U.S. institutions. The Company has not experienced any losses on its deposits of cash and cash equivalents. Management believes that the Company is not exposed to significant credit risk due to the financial position of the depository institutions in which those deposits are held.
Revenue Recognition
Revenue is recognized in accordance with Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 606, Revenue from Contracts with Customers (“ASC 606”). In accordance with ASC 606, the Company follows a five-step process to recognize revenues: 1) identify the contract with the customer, 2) identify the performance obligations, 3) determine the transaction price, 4) allocate the transaction price to the performance obligations and 5) recognize revenues when the performance obligations are satisfied.
All of the Company’s revenues from contracts with customers are associated with the provision of diagnostic and prognostic cancer testing services. Most of the Company’s revenues are attributable to DecisionDx-Melanoma for cutaneous melanoma. The Company also provides a test for uveal melanoma, DecisionDx-UM. Information on the disaggregation of revenues by the Company’s significant third-party payors is included under Payor Concentration below. The Company has determined that it has a contract with the patient when the treating clinician orders the test. The Company’s contracts generally contain a single performance obligation, which is the delivery of the test report, and the Company satisfies its performance obligation at a point in time upon the
F-35
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
delivery of the test report to the treating physician, at which point the Company can bill for the report. The amount of revenue recognized reflects the amount of consideration the Company expects to be entitled (the “transaction price”) and considers the effects of variable consideration, which is discussed further below.
Once the Company satisfies its performance obligations and bills for the service, the timing of the collection of payments may vary based on the payment practices of the third-party payor and the existence of contractually established reimbursement rates. Most of the payments for the Company’s services are made by third-party payors, including Medicare and commercial health insurance carriers. Certain contracts contain a contractual commitment of a reimbursement rate that differs from the Company’s list prices. However, absent a contractually committed reimbursement rate with a commercial carrier or governmental program, the Company’s diagnostic tests may or may not be covered by these entities’ existing reimbursement policies. In addition, patients do not enter into direct agreements with the Company that commit them to pay any portion of the cost of the tests in the event that their insurance provider declines to reimburse the Company. The Company may pursue, on a case-by-case basis, reimbursement from such patients in the form of co-payments and co-insurance, in accordance with the contractual obligations that the Company has with the insurance carrier or health plan. These situations may result in a delay in the collection of payments.
The Medicare claims that are covered by policy under a Local Coverage Determination (“LCD”) are generally paid at the established rate by the Company’s Medicare contractor within 30 days from receipt. Medicare claims that were either submitted to Medicare prior to the LCD coverage commencement date or are not covered by the terms of the LCD but meet the definition of being medically reasonable and necessary pursuant to the controlling Section 1862(a)(1)(A) of the Social Security Act are generally appealed and may ultimately be paid at the first (termed “redetermination”), second (termed “reconsideration”) or third level of appeal (de nova hearing with an ALJ). A successful appeal at any of these levels results in payment.
The Company has concluded that its contracts include variable consideration because the amounts paid by Medicare or commercial health insurance carriers may be paid at less than the Company’s standard rates or not paid at all, with such differences considered implicit price concessions. Variable consideration attributable to these price concessions is measured at the expected value using the “most likely amount” method under ASC 606. The amounts are determined by historical average collection rates by test type and payor category taking into consideration the range of possible outcomes, the predictive value of the Company’s past experiences, the time period of when uncertainties expect to be resolved and the amount of consideration that is susceptible to factors outside of the Company’s influence, such as the judgment and actions of third parties. Such variable consideration is included in the transaction price only to the extent it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainties with respect to the amount are resolved. Variable consideration may be constrained and excluded from the transaction price in situations where there is no contractually agreed upon reimbursement coverage or in the absence of a predictable pattern and history of collectability with a payor. Variable consideration for Medicare claims is deemed to be fully constrained when the payment of such claims is subject to approval by an ALJ at an appeal hearing, due to factors outside the Company’s influence (i.e., judgment or actions of third parties) and the uncertainty of the amount to be received is not expected to be resolved for a long period of time. Variable consideration is evaluated each reporting period and adjustments are recorded as increases or decreases in revenues. Included in revenues for the three months ended March 31, 2018 and 2019 was $631,822 and $648,192, respectively, of revenue increases associated with changes in estimated variable consideration related to performance obligations satisfied in previous periods.
Because the Company’s contracts with customers have an expected duration of one year or less, the Company has elected the practical expedient in ASC 606 to not disclose information about its remaining performance obligations. Any incremental costs to obtain contracts are recorded as selling, general and administrative expense as incurred due to the short duration of the Company’s contracts. Contract balances consisted solely of accounts receivable (both current and noncurrent) as of December 31, 2018 and March 31, 2019.
F-36
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
DecisionDx-Melanoma Claims Consolidation
In June 2017, the Company submitted to the Office of Medicare Hearings and Appeals (“OMHA”) a formal request to participate in a program that OMHA developed with the intent of providing appellants a means to have large volumes of claim disputes adjudicated at an accelerated rate. The program consolidates outstanding claims at the ALJ level and uses a statistical-sampling approach where five ALJ’s will determine reimbursement results for a sample of claims which are then extrapolated to the universe of claims. The consolidation includes 2,698 DecisionDx-Melanoma claims dating from 2013 through spring 2017. The judges who will review the sample sets have been identified and the hearings were held in April 2019 with a supplemental hearing in May 2019. No formal ruling has been issued to date. In accordance with ASC 606, the Company has not recognized (fully constrained the variable consideration) any revenues attributable to these claims in its financial statements pending the outcome of this matter. The Company expects to recognize any revenue adjudicated by the ALJ in the periodic reporting period in which the Company is notified of the ALJ hearing outcome.
Payor Concentration
The Company relies upon reimbursements from third-party government payors (primarily Medicare) and private-payor insurance companies to collect accounts receivable related to sales of its diagnostic tests.
The Company’s significant third-party payors and their related revenues as a percentage of total revenues and accounts receivable balances are as follows:
Percentage of Revenues |
||||||||||||||||||
Three Months Ended March 31, |
Percentage of Accounts Receivable (current) |
Percentage of Accounts Receivable (non-current) |
||||||||||||||||
2018 |
2019 |
Dec. 31, 2018 |
March 31, 2019 |
Dec. 31, 2018 |
March 31, 2019 |
|||||||||||||
Medicare |
10 | % |
19 | % |
54 | % |
15 | % |
— | % |
— | % |
||||||
Medicare Advantage plans |
25 | % |
25 | % |
9 | % |
26 | % |
18 | % |
17 | % |
||||||
United Healthcare |
20 | % |
9 | % |
7 | % |
10 | % |
15 | % |
5 | % |
||||||
BlueCross BlueShield plans |
17 | % |
9 | % |
18 | % |
29 | % |
41 | % |
46 | % |
||||||
Accounts Receivable and Allowance for Doubtful Accounts
The Company classifies accounts receivable balances that are expected to be paid more than one year from the balance sheet date as non-current assets. The estimated timing of payment utilized as a basis for classification as non-current is determined by analyses of historical payor-specific payment experience, adjusted for known factors that are expected to change the timing of future payments.
The Company accrues an allowance for doubtful accounts against its accounts receivable when it is probable that an account is not collectible, based on write off history, credit risk of specific accounts, aging analysis and other information available on specific accounts. The Company generally does not perform evaluations of customers’ financial condition and generally does not require collateral. Accounts receivable are written off when all efforts to collect the balance have been exhausted. Historically, the Company’s bad debt expense has not been significant. The allowance for doubtful accounts was zero as of December 31, 2017 and 2018 and as of March 31, 2019. Adjustments for implicit price concessions attributable to variable consideration, as discussed above, are incorporated into the measurement of the accounts receivable balances and are not part of the allowance for doubtful accounts.
Fair Value of Financial Instruments
The carrying amount of the Company’s long-term debt approximates fair value due to its variable market interest rate and management’s opinion that current rates and terms that would be available to the Company with the same maturity and security structure would be essentially equivalent to that of the Company’s long-term debt. The estimated fair value of the Company’s convertible promissory notes at March 31, 2019 was $15,300,633. These estimated fair values are “Level 3” fair value measurements, as defined in Note 9.
F-37
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
Accrued Compensation
The Company accrues for liabilities under discretionary employee and executive bonus plans. These estimated compensation liabilities are based on progress against corporate objectives approved by the Board of Directors, compensation levels of eligible individuals, and target bonus percentage levels. The Board of Directors reviews and evaluates the performance against these objectives and ultimately determines what discretionary payments are made. As of December 31, 2018, and March 31, 2019, the Company accrued approximately $3,197,234 and $1,145,865, respectively, for liabilities associated with these employee and executive bonus plans. These amounts are classified as current or noncurrent accrued liabilities in the balance sheets based on the expected timing of payment.
Deferred Offering Costs
Deferred offering costs consist primarily of legal fees, which are direct and incremental fees related to the Company’s planned initial public offering (the “IPO”). The deferred offering costs will be offset against the IPO proceeds upon the consummation of the IPO. In the event the offering is aborted, the deferred offering costs will be expensed. As of December 31, 2018 and March 31, 2019, the Company had incurred $91,307 and $554,477 in deferred offering costs, which are reported as other assets - long-term on the balance sheet.
Comprehensive Loss
Comprehensive loss is defined as a change in equity during a period from transactions and other events and circumstances from non-owner sources. The Company’s comprehensive loss was the same as its reported net loss for all periods presented.
Unaudited Pro Forma Information
The accompanying unaudited pro forma balance sheet information as of March 31, 2019 has been prepared assuming the automatic conversion of all outstanding shares of convertible preferred stock into 9,959,831 shares of common stock, the net exercise of certain preferred stock warrants into shares of common stock and the conversion of the outstanding principal plus accrued interest of the convertible promissory notes into shares of common stock, immediately prior to completion of the Company’s planned IPO. The assumed net exercise of certain preferred stock warrants is associated with warrants to purchase shares of the Company’s Series F preferred stock for $0.01 per share that will become exercisable into shares of the Company’s common stock immediately prior to the closing of the IPO and which will otherwise expire upon the closing of the IPO if not exercised. The assumed net exercise of these preferred stock warrants and the assumed conversion of the outstanding principal plus accrued interest of the convertible promissory notes are based on an IPO price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus). The pro forma balance sheet was prepared as though the completion of the proposed offering, reclassification of the carrying value of the convertible preferred stock to permanent equity, net exercise of certain preferred stock warrants and conversion of the outstanding principal plus accrued interest of the convertible promissory notes had occurred on March 31, 2019. Additionally, the pro forma balance sheet information assumes that the Company’s preferred stock warrant liability associated with the unexercised warrants will be transferred to additional paid-in capital as a result of the IPO because they will become common stock warrants not subject to remeasurement. Shares of common stock to be issued in the IPO and any related net proceeds are excluded from the pro forma balance sheet information.
Unaudited pro forma basic and diluted loss per common share attributable to common stockholders for the three months ended March 31, 2019 has been computed to give effect to the assumed automatic conversion of all the Company’s outstanding shares of convertible preferred stock into shares of common stock, the net exercise of certain preferred stock warrants (as described above) into shares of common stock and the conversion of the outstanding principal plus accrued interest of the convertible promissory notes into shares of common stock as if such conversions and net exercise had occurred at the beginning of 2019, or the date of issuance, if later. The assumed net exercise of these preferred stock warrants and the assumed conversion of the outstanding principal
F-38
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
plus accrued interest of the convertible promissory notes are based on an IPO price of $ per share (the midpoint of the price range set forth on the cover page of this prospectus). Shares of common stock to be issued in the IPO are excluded from the pro forma basic and diluted loss per share information.
Accounting Pronouncements Yet to be Adopted
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842), which supersedes FASB ASC Topic 840, Leases (Topic 840), and provides principles for the recognition, measurement, presentation and disclosure of leases for both lessees and lessors. The new standard requires lessees to apply a dual approach, classifying leases as either finance or operating leases based on the principle of whether or not the lease is effectively a financed purchase by the lessee. This classification will determine whether lease expense is recognized based on an effective interest method for finance leases or on a straight-line basis over the term of the lease for operating leases. A lessee is also required to record a right-of-use asset and a lease liability for all leases with a term of greater than 12 months regardless of classification. Leases with a term of 12 months or less will be accounted for similar to existing guidance for operating leases. For companies that are not emerging growth companies (“EGCs”), the ASU is effective for fiscal years beginning after December 15, 2018. For EGCs, the ASU is effective for fiscal years beginning after December 15, 2019. The Company will adopt the new standard using the modified retrospective method, under which the Company will apply Topic 842 to existing and new leases as of January 1, 2020, but prior periods will not be restated and will continue to be reported under Topic 840 guidance in effect during those periods. The Company anticipates that the adoption will not have a material impact on its statements of operations and comprehensive loss or its statements of cash flows but expects to recognize right-of-use assets and liabilities for lease obligations associated with its operating leases.
In June 2016, the FASB issued ASU No. 2016-13, Financial Instruments—Credit Losses, which requires the measurement of expected credit losses for financial instruments carried at amortized cost, such as accounts receivable, held at the reporting date based on historical experience, current conditions and reasonable forecasts. The main objective of this ASU is to provide financial statement users with more decision-useful information about the expected credit losses on financial instruments and other commitments to extend credit held by a reporting entity at each reporting date. In November 2018, the FASB issued ASU No. 2018-19, Codification Improvements to Topic 326, Financing Instruments—Credit Losses, which included an amendment of the effective date for nonpublic entities. For non-EGCs, the ASU is effective for fiscal years beginning after December 15, 2019. For EGCs, the standard is effective for fiscal years beginning after December 15, 2021. The Company does not believe the adoption of this standard will have a significant impact on its financial statements, given its history of minimal bad debt expense relating to trade accounts receivable.
3. Loss per share
Loss per share is computed by dividing net loss attributable to common stockholders for the period by the weighted-average number of common shares outstanding during the period. Diluted loss per share reflects the additional dilution from potential issuances of common stock, such as stock issuable pursuant to the exercise of stock options, as well as from the possible conversion of the Company’s convertible preferred stock, convertible promissory notes and exercise of outstanding warrants. The treasury stock and if-converted methods are used to calculate the potential dilutive effect of these common stock equivalents. However, potentially dilutive shares are excluded from the computation of diluted loss per share when their effect is antidilutive. Due to the Company reporting a net loss attributable to common stockholders for all periods presented, all potentially dilutive securities were antidilutive and have been excluded from the computation of diluted loss per share.
The table below provides potentially dilutive securities not included in the Company’s calculation of diluted loss per share because to do so would be antidilutive:
March 31, 2018 |
March 31, 2019 |
|||||
Shares issuable upon conversion of Series A convertible preferred stock |
533,711 | 533,711 | ||||
Shares issuable upon conversion of Series B convertible preferred stock |
816,654 | 816,654 | ||||
Shares issuable upon conversion of Series C convertible preferred stock |
503,056 | 503,056 | ||||
F-39
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
March 31, 2018 |
March 31, 2019 |
|||||
Shares issuable upon conversion of Series D convertible preferred stock |
756,416 | 756,416 | ||||
Shares issuable upon conversion of Series E-1 convertible preferred stock |
826,392 | 829,642 | ||||
Shares issuable upon conversion of Series E-2 convertible preferred stock |
934,433 | 934,433 | ||||
Shares issuable upon conversion of Series E-2A convertible preferred stock |
27,306 | 27,306 | ||||
Shares issuable upon conversion of Series E-3 convertible preferred stock |
824,000 | 824,000 | ||||
Shares issuable upon conversion of Series F convertible preferred stock |
3,840,982 | 4,734,613 | ||||
Shares issuable upon exercise of stock options |
1,422,882 | 2,575,158 | ||||
Shares issuable upon exercise of preferred stock warrants |
178,661 | 172,217 | ||||
Shares issuable upon conversion of convertible promissory notes(1) |
— | 2,180,529 | ||||
Total |
10,664,493 | 14,887,735 | ||||
| (1) | Based on conversion of the convertible promissory notes at maturity into Series F convertible preferred stock. In the event of the Next Equity Financing (as defined in Note 7), the convertible promissory notes convert into equity securities as discussed in Note 7. |
Unaudited Pro Forma Loss Per Share Attributable to Common Stockholders
The calculation of unaudited pro forma basic and diluted loss per share attributable to common stockholders for the three months ended March 31, 2019 is set forth in the table below:
Pro Forma |
|||
Three Months Ended March 31, 2019 |
|||
Basic and Diluted Loss per Share |
|||
Numerator |
|||
Net loss attributable to common stockholders, as reported |
$ | (2,342,356 | ) |
Pro forma adjustment to reflect assumed conversion of convertible preferred stock, cumulative dividends |
928,575 | ||
Pro forma adjustment to reflect assumed conversion of redeemable convertible preferred stock, accretion to redemption value |
55,798 | ||
Pro forma adjustment to remove change in fair value of preferred stock warrants, net of tax |
(13,157 | ) |
|
Pro forma adjustment to reflect assumed conversion of convertible promissory notes, interest expense and embedded derivative change in fair value, net of tax |
382,832 | ||
Pro forma net loss attributable to common stockholders |
$ | (988,308 | ) |
Denominator |
|||
Weighted-average shares outstanding, basic and diluted |
2,336,236 | ||
Pro forma adjustment to reflect assumed conversion of convertible preferred stock |
9,959,831 | ||
Pro forma adjustment to reflect assumed net exercise of certain preferred stock warrants |
|||
Pro forma adjustment to reflect assumed conversion of convertible promissory notes |
|||
Shares used to compute pro forma loss per share, basic and diluted |
|||
Pro forma loss per share, basic and diluted |
$ | ||
Pro forma diluted loss per share does not include outstanding stock options and preferred stock warrants (except for certain preferred stock warrants described in Note 2 under Unaudited Pro Forma Information) since the effect would be antidilutive due to the pro forma net loss attributable to common stockholders for the period.
F-40
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
4. Property and Equipment, Net
Property and equipment, net consisted of the following:
December 31, 2018 |
March 31, 2019 |
|||||
Lab equipment |
$ | 1,153,210 | $ | 1,280,479 | ||
Computer equipment |
629,437 | 759,294 | ||||
Leasehold improvements |
553,921 | 553,921 | ||||
Furniture and fixtures |
101,813 | 112,668 | ||||
Total |
2,438,381 | 2,706,362 | ||||
Less accumulated depreciation |
(909,385 | ) |
(986,805 | ) |
||
Property and equipment, net |
$ | 1,528,996 | $ | 1,719,557 | ||
Depreciation expense was recorded in the statements of operations and comprehensive loss as follows:
Three Months Ended March 31, |
||||||
2018 |
2019 |
|||||
Cost of sales |
$ | 38,617 | $ | 43,490 | ||
Research and development |
971 | 1,604 | ||||
Selling, general and administrative |
30,825 | 32,326 | ||||
Total |
$ | 70,413 | $ | 77,420 | ||
5. Intangible Assets, Net
Intangible assets consist of capitalized license costs as follows:
December 31, 2018 |
March 31, 2019 |
|||||||||||
Value |
Weighted- Average Remaining Life (Years) |
Value |
Weighted- Average Remaining Life (Years) |
|||||||||
Licenses |
$ | 274,534 | 0 | $ | 274,534 | 0 | ||||||
Accumulated amortization |
(270,367 | ) |
(272,867 | ) |
||||||||
Intangible assets, net |
$ | 4,167 | $ | 1,667 | ||||||||
Amortization expense was $9,337 and $2,500, for the three months ended March 31, 2018 and 2019, respectively.
6. Other Accrued Liabilities
Other accrued liabilities consisted of the following:
December 31, 2018 |
March 31, 2019 |
|||||
Accrued state income taxes |
$ | 8,606 | $ | 8,606 | ||
Accrued interest |
185,580 | 303,204 | ||||
Accrued royalties |
239,751 | 80,457 | ||||
Accrued service fees |
281,307 | 637,955 | ||||
Other |
— | 6,000 | ||||
Total |
$ | 715,244 | $ | 1,036,222 | ||
F-41
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
7. Convertible Promissory Notes
In January and February 2019, the Company issued $11,770,375 principal amount of unsecured convertible promissory notes (the “Notes”), of which $4,755,882 was with related parties (executive officers, members of the Company’s Board of Directors or entities affiliated with them). The Notes bear simple interest at a rate of 8% per annum with a maturity date of January 31, 2020.
On or before the maturity date, the entire outstanding principal amount of and accrued interest on the Notes (the “Conversion Amount”), is automatically convertible into shares of the Company’s equity securities issued and sold in a single or series of related transactions, with the principal purpose of raising capital, in which the Company sells shares of such equity securities for aggregate gross proceeds of at least $10.0 million (the “Next Equity Financing”). The number of shares of such equity securities issuable in the Next Equity Financing is equal to the quotient of the Conversion Amount as of the closing date of the Next Equity Financing divided by a per share price that is equal to 80% of the lowest per share purchase price of the equity securities sold in the Next Equity Financing. If the Notes have not been repaid or converted prior to the maturity date, then, at the request of the holders of a majority of the then-outstanding principal amount of and accrued interest on the Notes, then the Conversion Amount as of the maturity date will convert into shares of the Company’s Series F redeemable convertible preferred stock, or any senior equity security issued by the Company after the first issuance of the Notes, in each case at a conversion price equal to the price at which such security was last sold (currently $5.8208 for shares of the Company’s Series F redeemable convertible preferred stock). If a change of control of the Company occurs while the Notes are outstanding, the Company is required to repurchase each Note from each holder at a repurchase price equal to two times the principal amount of such Note, plus any accrued and unpaid interest on such Note as of the date of such repurchase.
The Company determined that the Notes contain embedded derivatives that require bifurcation and separate accounting under ASC 815-15, Embedded Derivatives. The Company determined that two such features in the Notes are not considered clearly and closely related to the host debt instrument and therefore require separate accounting: a) the automatic conversion feature in connection with the Next Equity Financing and b) the acceleration upon a change of control feature. Under ASC 815-15, these features are bundled together and accounting for as a single, compound embedded derivative. The Company determined the fair value of the embedded derivative liability at the issuance date, creating a discount to the carrying value of the Notes, which is amortized over the life of the debt using the effective interest method. The embedded derivative is recorded at fair value each reporting period, with changes in fair value recorded as “other income (expense), net” in the statements of operations and comprehensive loss. The embedded derivative liability is included in the same line as the convertible promissory notes on the balance sheet, as detailed in the table below. No hedge accounting treatment was applied. For details regarding the fair value measurement of the embedded derivative, see Note 9.
The Company also assessed the optional conversion feature into Series F redeemable convertible preferred stock at maturity and determined that this feature does not meet the definition of a derivative instrument because the settlement terms involve gross delivery of the underlying shares, which are not readily convertible to cash. The Company then assessed whether this feature caused the Notes to be subject to ASC 470-20, Debt with Conversion and Other Options and determined that the beneficial conversion feature guidance is applicable to the Notes. At issuance, the Company concluded that the Notes have a beneficial conversion feature because the fair value of the Series F preferred stock exceeded the conversion price of $5.8208 per share that would be applicable under the optional conversion at maturity, assuming no other equity securities senior to Series F preferred stock are sold. Under ASC 470-20, this beneficial conversion feature is measured at intrinsic value as of the issuance date of the Notes and is recognized as additional paid-in capital, creating a discount to the carrying value of the Notes that is amortized over the life of the debt using the effective interest method.
F-42
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
The following table summarizes the aggregate values recorded for the Notes at issuance and as of March 31, 2019:
At Issuance(1) |
March 31, 2019 |
|||||
Liability component: |
||||||
Principal (including $4,755,882 with related parties) |
$ | 11,770,375 | $ | 11,770,375 | ||
Unamortized issuance costs |
(74,880 | ) |
(73,523 | ) |
||
Unamortized discount from beneficial conversion feature |
(8,377,592 | ) |
(8,225,806 | ) |
||
Unamortized discount from embedded derivative |
(2,815,946 | ) |
(2,764,941 | ) |
||
Net carrying amount of the liability component |
501,957 | 706,105 | ||||
Embedded derivative liability |
2,815,946 | 2,862,031 | ||||
Total |
$ | 3,317,903 | $ | 3,568,136 | ||
Equity component: |
||||||
Carrying value of beneficial conversion feature recorded in additional paid-in capital |
$ | 8,377,592 | $ | 8,377,592 | ||
| (1) | The Notes were issued on January 31, 2019, February 12, 2019 and February 27, 2019. |
The Notes are classified as current liabilities on the balance sheet due to their contractual maturity date of January 31, 2020. Amortization of discounts and issuance costs on the Notes totaled $204,148 for the three months ended March 31, 2019 and were included in interest expense.
The amounts recognized in net loss for the three months ended March 31, 2019 for the embedded derivative liability are as follows:
Three Months Ended March 31, 2019 |
||||||
Statement of Operations and Comprehensive Loss Location |
Gain (Loss) Recognized in Net Loss |
|||||
Derivatives Not Classified as Hedging Instruments |
||||||
Embedded derivative in convertible promissory notes |
Other income (expense), net |
$ | (46,085 | ) |
||
The following table presents the fair value and balance sheet classification for the embedded derivative liability:
March 31, 2019 |
|||||||||
Classification |
Balance Sheet Location |
Fair Value |
|||||||
Derivatives Not Classified as Hedging Instruments |
|||||||||
Embedded derivative in convertible promissory notes |
Liability | Convertible promissory notes |
$ | 2,862,031 | |||||
8. Long-Term Debt
The Company’s long-term debt consists of term debt and a revolving line of credit and are presented in the table below:
December 31, 2018 |
March 31, 2019 |
|||||
Term debt |
$ | 21,350,000 | $ | 21,350,000 | ||
Revolving line of credit |
5,000,000 | 4,010,885 | ||||
Total principal amount |
26,350,000 | 25,360,885 | ||||
Unamortized discount and issuance costs |
(1,850,248 | ) |
(1,728,569 | ) |
||
Total long-term debt |
$ | 24,499,752 | $ | 23,632,316 | ||
F-43
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
Term Debt
2018 Loan and Security Agreement
On November 30, 2018 (the “Closing Date”), the Company entered into a new Loan and Security Agreement (the “2018 LSA”) with Oxford Finance LLC (“Oxford”), as collateral agent, and Oxford and Silicon Valley Bank (“SVB”) as equal syndicated lenders (collectively, the “Lenders”). The 2018 LSA replaced the Company’s previous lending arrangement and provided for a $20.0 million secured term loan credit facility (the “2018 Term Loan”) and a credit line of up to $5.0 million (discussed in the “Revolving Line of Credit” section below), prior to amendment of the 2018 LSA on June 13, 2019, as discussed below. The Company’s obligations under the 2018 LSA are secured by substantially all of its assets, excluding intellectual property and subject to certain other exceptions and limitations. The Company has the right to prepay the 2018 Term Loan in whole or in part at any time, subject to a prepayment fee of 2.50% if prepaid on or prior to November 30, 2019, 1.50% if prepaid after November 30, 2019 and on or prior to November 30, 2020, and 0.75% thereafter. Upon prepayment, the Company is also obligated to pay a non-refundable early termination fee of $496,785. Amounts prepaid or repaid under the 2018 Term Loan may not be reborrowed. The 2018 LSA contains a financial covenant that requires the Company to achieve a monthly trailing six-month revenue target each month throughout the term of the agreement, which was recently amended and changed to a monthly trailing three-month target, as discussed further below. As of March 31, 2019, the Company was in compliance with this covenant. In addition, the 2018 LSA contains customary conditions of borrowing, events of default and covenants, including covenants that restrict the Company’s ability to dispose of assets, merge with or acquire other entities, incur indebtedness and make distributions to holders of the Company’s capital stock. Should an event of default occur, including the occurrence of a material adverse change, the Company could be liable for immediate repayment of all obligations under the 2018 LSA.
On June 13, 2019, the Company entered into an amendment of the 2018 LSA (the “Amendment”) which, among other things, requires that the Company achieve certain revenue requirements tested monthly on a trailing three-month basis. For each month through December 31, 2019, the trailing three-month revenues are calculated as a percentage of the Company’s previously approved applicable monthly revenue targets. For monthly periods ending after December 31, 2019, the trailing three-months revenue requirements will be determined by the Lenders upon receipt and review of the Company’s monthly financial projections for the year, subject to certain specified criteria regarding minimum requirements. Revenues, if any, that the Company recognizes as a result of an ALJ appeal process from consolidated claims initiatives for DecisionDx-Melanoma do not count toward the minimum revenue requirements. Additionally, the Amendment eliminated the $5.0 million revolving line and increased the 2018 Term Loan amount by $5.0 million.
The 2018 Term Loan bears interest at a floating rate equal to the greater of 1) 8.55% and 2) the 30-day U.S. LIBOR rate as reported in The Wall Street Journal on the last business day of the month that precedes the month in which the interest will accrue, plus 6.48%. The applicable interest rate on the 2018 Term Loan was 8.98% and 8.97% at December 31, 2018 and March 31, 2019, respectively. Interest on the 2018 Term Loan is payable monthly in arrears. The Company is permitted to make interest-only payments on the 2018 Term Loan for the 18 months following the Closing Date. The principal is required to be repaid in 30 equal monthly installments beginning on June 1, 2020. All unpaid principal and accrued and unpaid interest is due on November 1, 2022 (the “2018 Term Loan Maturity Date”). The Company is also obligated to make an additional final payment of 6.75% of the aggregate original principal amount, or $1,350,000 as of March 31, 2019, upon any prepayment or on the 2018 Term Loan Maturity Date. The final payment amount is being amortized as additional interest expense using the effective interest method over the term of the debt. The final payment amount increased to $1,687,500 in connection with the $5.0 million increase to the 2018 Term Loan under the Amendment.
The 2018 Term Loan was fully funded on the Closing Date and the 2018 Revolving Line (defined and discussed below) was fully drawn upon on the Closing Date. Proceeds from the 2018 LSA were used to repay all outstanding obligations under the Company’s previous lending arrangement and to provide working capital. As a condition of the loan, the Company issued Series F preferred stock warrants to the Lenders with an aggregate initial fair value of $158,000. In accordance with ASC 480-10, the warrants are liability-classified as the
F-44
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
underlying to the warrant is a puttable security. The initial recognition of the warrant liability created a discount to the debt, which is being amortized over the debt term using the effective interest method. The 2018 LSA was accounted for as a modification of the previous lending arrangement with SVB and Oxford, and therefore no extinguishment gain or loss was recognized.
Revolving Line of Credit
Under the 2018 LSA, the Company had a $5.0 million revolving line of credit (the “2018 Revolving Line”), contingent on the Company’s satisfaction of borrowing base eligibility requirements. The 2018 Revolving Line bore interest at a floating per annum rate equal to the greater of 1) 6.25% and 2) 5.48% above the U.S. LIBOR rate. The applicable interest rate on the 2018 Revolving Line at December 31, 2018 and March 31, 2019 was 7.98% and 7.97%, respectively. The 2018 Revolving Line was to be due in full no later than November 30, 2020, but was eliminated in connection with the Amendment.
9. Fair Value Measurements
Fair value is defined as the price that would be received to sell an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market in an orderly transaction between market participants at the measurement date. The fair value hierarchy prioritizes the inputs to valuation techniques used in measuring fair value. There are three levels to the fair value hierarchy based on the reliability of inputs, as follows:
Level 1 – Observable inputs that reflect quoted prices (unadjusted) for identical assets or liabilities in active markets.
Level 2 – Inputs other than quoted prices included in Level 1 that are observable for the asset or liability, either directly or indirectly.
Level 3 – Unobservable inputs in which little or no market data exists, therefore requiring the Company to develop its own assumptions.
Financial instruments measured at fair value are classified in their entirety based on the lowest level of input that is significant to the fair value measurement. The Company’s assessment of the significance of a particular input to the fair value measurement in its entirety requires management to make judgments and consider factors specific to the asset or liability. The use of different assumptions and/or estimation methodologies may have a material effect on estimated fair values. Accordingly, the fair value estimates disclosed, or amounts recorded may not be indicative of the amount that the Company or holders of the instruments could realize in a current market exchange.
The table below provides information, by level within the fair value hierarchy, of the Company’s financial assets and liabilities that are accounted for at fair value on a recurring basis as of December 31, 2018 and March 31, 2019:
As of December 31, 2018 |
||||||||||||
Quoted Prices in Active Markets for Identical Items (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
Total |
|||||||||
Liabilities: |
||||||||||||
Preferred stock warrants |
$ | — | $ | — | $ | 1,193,726 | $ | 1,193,726 | ||||
F-45
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
As of March 31, 2019 |
||||||||||||
Quoted Prices in Active Markets for Identical Items (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
Total |
|||||||||
Liabilities: |
||||||||||||
Preferred stock warrants |
$ | — | $ | — | $ | 1,180,569 | $ | 1,180,569 | ||||
Embedded derivative in convertible promissory notes(1) |
$ | — | $ | — | $ | 2,862,031 | $ | 2,862,031 | ||||
| (1) | Included in the convertible promissory notes line on the balance sheet. |
The following table discloses the summary of changes in the fair value of Level 3 fair value measurements:
Three Months Ended March 31, 2019 |
||||||
Preferred stock warrants |
Embedded derivative in convertible promissory notes |
|||||
Balance, December 31, 2018 |
$ | 1,193,726 | $ | — | ||
Issuance of convertible promissory notes |
— | 2,815,946 | ||||
Change in fair value included in net loss |
(13,157 | ) |
46,085 | |||
Balance, March 31, 2019 |
$ | 1,180,569 | $ | 2,862,031 | ||
The changes in fair value of the preferred stock warrant liability and the embedded derivative liability are recorded as “other income (expense), net” in the statements of operations and comprehensive loss.
The fair value of the warrants for shares of Series A, Series B, Series E-1, Series E-2, Series E-2A, Series E-3, and Series F redeemable convertible preferred stock was estimated by management using the Black-Scholes option pricing model with the following assumptions:
|
|
December 31, 2018
|
March 31, 2019
|
|
Average expected life (years)
|
9
|
7
|
|
Expected stock price volatility
|
64%
|
64%
|
|
Risk-free interest rate
|
2.51% - 2.69%
|
2.22% - 2.39%
|
|
Dividend yield
|
0%
|
0%
|
Certain features of the Notes were determined to be an embedded derivative requiring bifurcation and separate accounting, as discussed in Note 7. The fair value of the embedded derivative was determined based on a probability-weighted income approach discounted at an interest rate that is consistent with the appropriate market interest rate (11.32% as of March 31, 2019) considering management’s estimates of the probability of the possible settlement outcomes. As of March 31, 2019, management assessed the probability of the occurrence of the Next Equity Financing at 90%.
10. Commitments and Contingencies
From time to time, the Company may be involved in legal proceedings arising in the ordinary course of business. The Company believes there is no threatened litigation or litigation pending that could have, individually or in the aggregate, a material adverse effect on the Company’s financial position, results of operations or cash flows.
F-46
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
11. Convertible Preferred Stock and Preferred Stock Warrants
Convertible Preferred Stock
Convertible preferred stock consists of the following:
Convertible preferred stock
As of December 31, 2018 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series C |
503,056 | 503,056 | $ | 3.4800 | $ | 1,750,635 | $ | 666,560 | $ | 2,417,195 | $ | 1,500,994 | |||||||||
Redeemable convertible preferred stock
As of December 31, 2018 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series A |
533,711 | 533,711 | $ | 2.1400 | $ | 1,142,142 | $ | 713,498 | $ | 1,855,640 | $ | 1,142,142 | |||||||||
Series B |
816,654 | 816,654 | $ | 2.2500 | 1,837,472 | 982,840 | 2,820,312 | 1,931,634 | |||||||||||||
Series D |
756,416 | 756,416 | $ | 3.7700 | 2,851,688 | 1,339,789 | 4,191,477 | 2,851,688 | |||||||||||||
Series E-1 |
842,641 | 829,642 | $ | 3.7700 | 3,127,750 | 1,095,281 | 4,223,031 | 3,098,578 | |||||||||||||
Series E-2 |
949,725 | 934,433 | $ | 4.5776 | 4,277,461 | 1,502,821 | 5,780,282 | 4,277,461 | |||||||||||||
Series E-2A |
27,306 | 27,306 | $ | 4.5776 | 124,996 | 38,738 | 163,734 | 124,762 | |||||||||||||
Series E-3 |
830,554 | 824,000 | $ | 5.3405 | 4,400,572 | 1,363,688 | 5,764,260 | 4,393,509 | |||||||||||||
Series F |
4,883,486 | 4,734,613 | $ | 5.8208 | 27,559,235 | 5,212,065 | 32,771,300 | 27,175,383 | |||||||||||||
Total |
9,640,493 | 9,456,775 | $ | 45,321,316 | $ | 12,248,720 | $ | 57,570,036 | $ | 44,995,157 | |||||||||||
Convertible preferred stock
As of March 31, 2019 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series C |
503,056 | 503,056 | $ | 3.4800 | $ | 1,750,635 | $ | 701,093 | $ | 2,451,728 | $ | 1,500,994 | |||||||||
Redeemable convertible preferred stock
As of March 31, 2019 |
|||||||||||||||||||||
Shares Authorized |
Shares Issued and Outstanding |
Original Issue Price per Share |
Original Issue Value |
Accumulated and Undeclared Dividends |
Aggregate Liquidation Preference |
Carrying Value |
|||||||||||||||
Series A |
533,711 | 533,711 | $ | 2.1400 | $ | 1,142,142 | $ | 736,028 | $ | 1,878,170 | $ | 1,142,142 | |||||||||
Series B |
816,654 | 816,654 | $ | 2.2500 | 1,837,472 | 1,019,086 | 2,856,558 | 1,931,635 | |||||||||||||
Series D |
756,416 | 756,416 | $ | 3.7700 | 2,851,688 | 1,396,041 | 4,247,729 | 2,851,688 | |||||||||||||
Series E-1 |
842,641 | 829,642 | $ | 3.7700 | 3,127,750 | 1,156,979 | 4,284,729 | 3,099,609 | |||||||||||||
Series E-2 |
949,725 | 934,433 | $ | 4.5776 | 4,277,461 | 1,587,196 | 5,864,657 | 4,277,461 | |||||||||||||
Series E-2A |
27,306 | 27,306 | $ | 4.5776 | 124,996 | 41,203 | 166,199 | 124,857 | |||||||||||||
Series E-3 |
830,554 | 824,000 | $ | 5.3405 | 4,400,572 | 1,450,486 | 5,851,058 | 4,396,372 | |||||||||||||
Series F |
7,089,870 | 4,734,613 | $ | 5.8208 | 27,559,235 | 5,755,742 | 33,314,977 | 27,227,191 | |||||||||||||
Total |
11,846,877 | 9,456,775 | $ | 45,321,316 | $ | 13,142,761 | $ | 58,464,077 | $ | 45,050,955 | |||||||||||
F-47
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
Preferred Stock Warrant Liabilities
Warrants to purchase redeemable convertible preferred shares consist of the following:
As of December 31, 2018 |
|||||||||||||||||||
Warrants |
Year of First Issuance |
Shares |
Exercise Price |
Year of Expiration |
Exercised |
Expired |
Outstanding |
||||||||||||
Series A Preferred Stock |
2008 | 108,057 | $ | 2.1400 | 2015 |
(96,375 | ) |
(11,682 | ) |
— | |||||||||
Series B Preferred Stock |
2010 | 86,667 | $ | 2.2500 | 2017 |
(86,667 | ) |
— | — | ||||||||||
Series E-1 Preferred Stock |
2014 | 16,249 | $ | 3.7700 | 2024 |
(3,250 | ) |
— | 12,999 | ||||||||||
Series E-2 Preferred Stock |
2014 | 15,292 | $ | 4.5776 | 2024 |
— | — | 15,292 | |||||||||||
Series E-3 Preferred Stock |
2015 | 6,554 | $ | 5.3405 | 2024 |
— | — | 6,554 | |||||||||||
Series F Preferred Stock |
2016 | 103,090 | $ | 5.8208 | 2026-2028 |
— | — | 103,090 | |||||||||||
Series F Preferred Stock |
2018 | 67,233 | $ | 0.0100 | 2023 |
(32,951 | ) |
— | 34,282 | ||||||||||
Total |
403,142 | (219,243 | ) |
(11,682 | ) |
172,217 | |||||||||||||
As of March 31, 2019 |
|||||||||||||||||||
Warrants |
Year of First Issuance |
Shares |
Exercise Price |
Year of Expiration |
Exercised |
Expired |
Outstanding |
||||||||||||
Series A Preferred Stock |
2008 | 108,057 | $ | 2.1400 | 2015 |
(96,375 | ) |
(11,682 | ) |
— | |||||||||
Series B Preferred Stock |
2010 | 86,667 | $ | 2.2500 | 2017 |
(86,667 | ) |
— | — | ||||||||||
Series E-1 Preferred Stock |
2014 | 16,249 | $ | 3.7700 | 2024 |
(3,250 | ) |
— | 12,999 | ||||||||||
Series E-2 Preferred Stock |
2014 | 15,292 | $ | 4.5776 | 2024 |
— | — | 15,292 | |||||||||||
Series E-3 Preferred Stock |
2015 | 6,554 | $ | 5.3405 | 2024 |
— | — | 6,554 | |||||||||||
Series F Preferred Stock |
2016 | 103,090 | $ | 5.8208 | 2026-2028 |
— | — | 103,090 | |||||||||||
Series F Preferred Stock |
2018 | 67,233 | $ | 0.0100 | 2023 |
(32,951 | ) |
— | 34,282 | ||||||||||
Total |
403,142 | (219,243 | ) |
(11,682 | ) |
172,217 | |||||||||||||
12. Stock Incentive Plan and Stock Based Compensation
Stock Incentive Plan
Activity under the Company’s 2008 Stock Plan and the 2018 Stock Plan for the three months ended March 31, 2019 is set forth below:
Weighted-Average |
|||||||||||||||
Shares Available for Grant |
Stock Options Outstanding |
Exercise Price |
Remaining Contractual Term (Years) |
Aggregate Intrinsic Value |
|||||||||||
Balance as of December 31, 2018 |
735,692 | 2,023,158 | $ | 1.63 | |||||||||||
Granted |
(554,000 | ) |
554,000 | $ | 2.77 | ||||||||||
Exercised |
— | (583 | ) |
$ | 1.57 | ||||||||||
Forfeited/Cancelled |
1,417 | (1,417 | ) |
$ | 1.85 | ||||||||||
Balance as of March 31, 2019 |
183,109 | 2,575,158 | $ | 1.88 | 6.91 | $ | 2,299,621 | ||||||||
Exercisable at March 31, 2019(1) |
1,219,548 | $ | 1.48 | 4.39 | $ | 1,212,753 | |||||||||
| (1) | Vested and exercisable options. Additionally, 321,578 outstanding unvested options with a weighted-average exercise price of $1.96 per share may be exercised prior to vesting as of March 31, 2019 under an early-exercise provision in the stock option award agreement. In the event of such exercise, the shares obtained upon exercise would be restricted and subject to forfeiture prior to vesting. No such early exercises have occurred to date. |
F-48
CASTLE BIOSCIENCES, INC.
NOTES TO CONDENSED FINANCIAL STATEMENTS (CONTINUED)
(UNAUDITED)
Determining Fair Value of Stock Options - Summary of Assumptions
The Company uses the Black-Scholes option pricing model to estimate the fair value of each option grant on the date of grant or any other measurement date. The following table sets forth the assumptions used to determine the fair value of stock options:
|
|
Three Months Ended
March 31, 2019 |
|
Average expected term
|
6 years
|
|
Expected stock price volatility
|
58.22% - 58.84%
|
|
Risk-free interest rate
|
2.18% - 2.47%
|
|
Dividend yield
|
0
|
Stock-based compensation expense related to stock options is included in the statements of operations and comprehensive loss as follows:
Three Months Ended March 31, |
||||||
2018 |
2019 |
|||||
Cost of sales |
$ | 4,774 | $ | 10,124 | ||
Research and development |
12,681 | 20,184 | ||||
Selling, general and administrative |
44,252 | 138,113 | ||||
Total stock-based compensation expense |
$ | 61,707 | $ | 168,421 | ||
No options were granted during the three months ended March 31, 2018. The weighted-average grant date fair value of options granted during the three months ended March 31, 2019 was $1.56 per option, respectively. As of March 31, 2019, there was $1,577,669 of total unrecognized compensation cost, which is expected to be recognized on a straight-line basis over a weighted-average period of 3.23 years. The total unrecognized compensation costs will be adjusted for forfeitures in future periods as they occur.
13. Income Taxes
No income tax expense or benefit was recorded for the three months ended March 31, 2018 and 2019 due to the full valuation allowance on the Company’s net deferred tax asset, based on the Company’s assessment that it is more likely than not that the benefits of those deferred tax assets will not be realized.
14. Subsequent Events
As discussed in Note 8, on June 13, 2019, the Company entered into an amendment of the 2018 LSA with Oxford and SVB. The Amendment modified the monthly trailing revenue covenant from a six-month calculation to a three-month calculation with revised revenue targets. Additionally, the Amendment eliminated the 2018 Revolving Line and increased the 2018 Term Loan amount by $5.0 million, with the same terms and conditions applicable to the existing term loan.
On June 24, 2019, the Company’s Board of Directors approved the grant of options to purchase 33,000 shares of Common Stock of the Company for an exercise price of $3.98 per share. The options were granted pursuant to the Company’s 2018 Equity Incentive Plan, have a ten-year term and vest over a four year period (with 25% vesting after one year from the vesting start date and the remainder vesting in monthly installments thereafter).
For the purposes of the interim financial statements as of March 31, 2019 and for the three months then ended, the Company has evaluated the subsequent events through June 26, 2019, the date the interim financial statements were issued.
F-49
Shares

Common Stock
PRELIMINARY PROSPECTUS
Joint Book-Running Managers
SVB Leerink
Baird
Co-Managers
Canaccord Genuity
BTIG
, 2019
Through and including , 2019 (the 25th day after the commencement of this offering), all dealers that buy, sell or trade shares of our common stock, whether or not participating in this offering, may be required to deliver a prospectus. This delivery requirement is in addition to the dealers’ obligation to deliver a prospectus when acting as underwriters and with respect to their unsold allotments or subscriptions.
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Unless otherwise indicated, all references to “Castle Biosciences,” the “company,” “we,” “our,” “us” or similar terms refer to Castle Biosciences, Inc.
| Item 13. | Other Expenses of Issuance and Distribution. |
The following table sets forth all expenses to be paid by us, other than underwriting discounts and commissions, in connection with this offering. All amounts shown are estimates except for the SEC registration fee, the FINRA filing fee and the exchange listing fee.
SEC registration fee |
$ | 6,969 | |
FINRA filing fee |
9,125 | ||
Exchange listing fee |
150,000 | ||
Printing and engraving expenses |
* |
||
Legal fees and expenses |
* |
||
Accounting fees and expenses |
* |
||
Transfer agent and registrar fees and expenses |
* |
||
Miscellaneous expenses |
* |
||
Total |
$ | * |
| * | To be provided by amendment. |
| Item 14. | Indemnification of Directors and Officers. |
We are incorporated under the laws of the State of Delaware. Section 145 of the Delaware General Corporation Law provides that a Delaware corporation may indemnify any persons who were, are, or are threatened to be made, parties to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative (other than an action by or in the right of such corporation), by reason of the fact that such person is or was an officer, director, employee or agent of such corporation, or is or was serving at the request of such corporation as an officer, director, employee or agent of another corporation or enterprise. The indemnity may include expenses (including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by such person in connection with such action, suit or proceeding, provided that such person acted in good faith and in a manner he or she reasonably believed to be in or not opposed to the corporation’s best interests and, with respect to any criminal action or proceeding, had no reasonable cause to believe that his or her conduct was illegal. A Delaware corporation may indemnify any persons who were, are, or are threatened to be made, a party to any threatened, pending or completed action or suit by or in the right of the corporation by reason of the fact that such person is or was a director, officer, employee or agent of such corporation, or is or was serving at the request of such corporation as a director, officer, employee or agent of another corporation or enterprise. The indemnity may include expenses (including attorneys’ fees) actually and reasonably incurred by such person in connection with the defense or settlement of such action or suit, provided such person acted in good faith and in a manner he or she reasonably believed to be in or not opposed to the corporation’s best interests, except that no indemnification is permitted without judicial approval if the officer or director is adjudged to be liable to the corporation. Where an officer or director is successful on the merits or otherwise in the defense of any action referred to above, the corporation must indemnify him or her against the expenses (including attorneys’ fees) actually and reasonably incurred.
Our amended and restated certificate of incorporation and amended and restated bylaws, which will become effective immediately prior to and upon the closing of this offering, respectively, provide for the indemnification of our directors and officers to the fullest extent permitted under the Delaware General Corporation Law. Section 102(b)(7) of the Delaware General Corporation Law permits a corporation to provide in its certificate of incorporation that a director of the corporation shall not be personally liable to the corporation or its stockholders for monetary damages for breach of fiduciary duties as a director, except for liability for any:
| • | transaction from which the director derives an improper personal benefit; |
| • | act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; |
II-1
| • | unlawful payment of dividends or redemption of shares; or |
| • | breach of a director’s duty of loyalty to the corporation or its stockholders. |
Our amended and restated certificate of incorporation, as currently in effect, includes such a provision, and our amended and restated certificate of incorporation that will become effective immediately prior to the completion of this offering will include such a provision. Expenses incurred by any officer or director in defending any such action, suit or proceeding in advance of its final disposition shall be paid by us upon delivery to it of an undertaking, by or on behalf of such director or officer, to repay all amounts so advanced if it shall ultimately be determined that such director or officer is not entitled to be indemnified by us.
Section 174 of the Delaware General Corporation Law provides, among other things, that a director who willfully or negligently approves of an unlawful payment of dividends or an unlawful stock purchase or redemption may be held liable for such actions. A director who was either absent when the unlawful actions were approved or dissented at the time may avoid liability by causing his or her dissent to such actions to be entered in the books containing minutes of the meetings of the board of directors at the time such action occurred or immediately after such absent director receives notice of the unlawful acts.
As permitted by the Delaware General Corporation Law, we have entered into indemnity agreements with each of our directors and executive officers, that require us to indemnify such persons against any and all costs and expenses (including attorneys’, witness or other professional fees) actually and reasonably incurred by such persons in connection with any action, suit or proceeding (including derivative actions), whether actual or threatened, to which any such person may be made a party by reason of the fact that such person is or was a director or officer or is or was acting or serving as an officer, director, employee or agent of us or any of our affiliated enterprises. Under these agreements, we are not required to provide indemnification for certain matters, including:
| • | indemnification beyond that permitted by the Delaware General Corporation Law; |
| • | indemnification for any proceeding with respect to the unlawful payment of remuneration to the director or officer; |
| • | indemnification for certain proceedings involving a final judgment that the director or officer is required to disgorge profits from the purchase or sale of our stock; |
| • | indemnification for proceedings involving a final judgment that the director’s or officer’s conduct was in bad faith, knowingly fraudulent or deliberately dishonest or constituted willful misconduct or a breach of his or her duty of loyalty, but only to the extent of such specific determination; |
| • | indemnification for proceedings or claims brought by an officer or director against us or any of our directors, officers, employees or agents, except for claims to establish a right of indemnification or proceedings or claims approved by our board of directors or required by law; |
| • | indemnification for settlements the director or officer enters into without our consent; or |
| • | indemnification in violation of any undertaking required by the Securities Act of 1933, as amended, or the Securities Act, or in any registration statement filed by us. |
The indemnification agreements also set forth certain procedures that will apply in the event of a claim for indemnification thereunder. Except as otherwise disclosed under the heading “Legal Proceedings” in the “Business” section of the prospectus included in this registration statement, there is at present no pending litigation or proceeding involving any of our directors or executive officers as to which indemnification is required or permitted, and we are not aware of any threatened litigation or proceeding that may result in a claim for indemnification.
We have an insurance policy in place that covers our officers and directors with respect to certain liabilities, including liabilities arising under the Securities Act, or otherwise.
We plan to enter into an underwriting agreement which provides that the underwriters are obligated, under some circumstances, to indemnify our directors, officers and controlling persons against specified liabilities, including liabilities under the Securities Act.
II-2
| Item 15. | Recent sales of unregistered securities. |
Set forth below is information regarding securities issued and options granted by us since January 1, 2016 that were not registered under the Securities Act. Also included is the consideration, if any, received by us, for such securities and options and information relating to the Securities Act, or rule of the SEC, under which exemption from registration was claimed.
| (1) | In February 2016, we issued a warrant to purchase an aggregate of 12,885 shares of our Series F redeemable convertible preferred stock to Silicon Valley Bank, or SVB, in connection with the entry into a loan and security agreement with SVB. |
| (2) | In December 2016, we issued a warrant to purchase an aggregate of 4,295 shares of our Series F redeemable convertible preferred stock to SVB in connection with the entry into an amendment to our loan and security agreement with SVB. |
| (3) | In March 2017, we issued a warrant to purchase an aggregate of 64,432 shares of our Series F redeemable convertible preferred stock to SVB and Oxford Finance LLC, or Oxford, in connection with the entry into a loan and security agreement with SVB and Oxford. |
| (4) | In January 2018, we sold and issued an aggregate of 940,605 shares of our Series F redeemable convertible preferred stock at a purchase price of $5.8208 per share, for aggregate gross proceeds of approximately $5.5 million, and issued warrants, at a purchase price of $0.01 per warrant share, to purchase an aggregate of 67,233 shares of our Series F redeemable convertible preferred stock, for aggregate gross proceeds of approximately $672, each to accredited investors. |
| (5) | In May 2018, we sold and issued an aggregate of 783,248 shares of our Series F redeemable convertible preferred stock at a purchase price of $5.8208 per share, for aggregate gross proceeds of approximately $4.6 million. |
| (6) | In June 2018, we sold and issued an aggregate of 85,711 shares of our Series F redeemable convertible preferred stock at a purchase price of $5.8208 per share, for aggregate gross proceeds of approximately $0.5 million. |
| (7) | In November 2018, we issued warrants to each of SVB and Oxford to purchase an aggregate of 21,478 shares of our Series F redeemable convertible preferred stock in connection with the entry into our current loan and security agreement with SVB and Oxford. |
| (8) | In January 2019 and February 2019, we issued convertible promissory notes in an aggregate principal amount of approximately $11.8 million to investors. |
| (9) | From January 1, 2016 through June 26, 2019, we granted stock options to purchase an aggregate of 1,973,789 shares of our common stock at a weighted-average exercise price of $2.11 per share, to certain of our employees, consultants and directors in connection with services provided to us by such persons. Through June 26, 2019, options to purchase 327,165 shares have been exercised for aggregate consideration of approximately $405 thousand and options to purchase 345,917 shares have been cancelled. |
The offers, sales and issuances of the securities described in paragraphs (1) through (8) were deemed to be exempt from registration under the Securities Act in reliance on Section 4(a)(2) (or Regulation D promulgated thereunder) in that the issuance of securities to the accredited investors did not involve a public offering. The recipients of securities in each of these transactions acquired the securities for investment only and not with a view to or for sale in connection with any distribution thereof and appropriate legends were affixed to the securities issued in these transactions. Each of the recipients of securities in these transactions was an accredited investor under Rule 501 of Regulation D. No underwriters were involved in these transactions.
The offers, sales and issuances of the securities described in paragraph (9) were deemed to be exempt from registration under the Securities Act in reliance on either Rule 701 in that the transactions were under compensatory benefit plans and contracts relating to compensation as provided under Rule 701 or Section 4(a)(2) in that the issuance of securities to the accredited investors did not involve a public offering. The recipients of such securities were our employees, directors or bona fide consultants and received the securities under the 2008 Stock Plan and the 2018 Equity Incentive Plan.
II-3
Appropriate legends were affixed to the securities issued in these transactions. Each of the recipients of securities in these transactions had adequate access, through employment, business or other relationships, to information about us.
| Item 16. | Exhibits and financial statement schedules. |
| (a) | Exhibits. |
EXHIBIT INDEX
|
Exhibit number
|
Description of document
|
|
1.1†
|
Form of Underwriting Agreement.
|
|
Seventh Amended and Restated Certificate of Incorporation, as amended.
|
|
|
Form of Amended and Restated Certificate of Incorporation to become effective immediately prior to the closing of the offering.
|
|
|
Bylaws.
|
|
|
Form of Amended and Restated Bylaws to become effective immediately prior to the closing of the offering.
|
|
|
4.1†
|
Form of Common Stock Certificate.
|
|
Fifth Amended and Restated Investors’ Rights Agreement, dated July 15, 2015, by and among the Registrant and certain of its stockholders, as amended.
|
|
|
Form of warrant to purchase Series E-1 redeemable convertible preferred stock issued to investors.
|
|
|
Form of warrant to purchase Series E-2 redeemable convertible preferred stock issued to Silicon Valley Bank on December 31, 2014.
|
|
|
Form of warrant to purchase Series F redeemable convertible preferred stock issued to Silicon Valley Bank on February 22, 2016.
|
|
|
Form of warrant to purchase Series F redeemable convertible preferred stock issued to Oxford Finance LLC and Silicon Valley Bank on March 31, 2017.
|
|
|
Form of warrant to purchase Series F redeemable convertible preferred stock issued to Oxford Finance LLC and Silicon Valley Bank on November 30, 2018.
|
|
|
Form of Warrant to Purchase Series F redeemable convertible preferred stock issued to investors.
|
|
|
5.1†
|
Opinion of Cooley LLP.
|
|
10.1+
|
Form of Indemnity Agreement by and between the Registrant and its directors and officers.
|
|
Castle Biosciences, Inc. 2008 Stock Plan
|
|
|
Forms of Stock Option Agreement, Exercise Notice and Investment Representation Statement under the 2008 Stock Plan.
|
|
|
Castle Biosciences, Inc. 2018 Equity Incentive Plan.
|
|
|
Forms of Stock Option Grant Notice, Option Agreement and Notice of Exercise under the 2018 Equity Incentive Plan.
|
|
|
Castle Biosciences, Inc. 2019 Equity Incentive Plan.
|
|
|
Forms of Stock Option Grant Notice, Option Agreement and Notice of Exercise under the 2019 Equity Incentive Plan.
|
|
|
Castle Biosciences, Inc. 2019 Employee Stock Purchase Plan.
|
|
|
Form of Director Agreement by and between the Registrant and certain of its directors.
|
|
|
Amended and Restated Executive Employment Agreement, dated September 20, 2012, as amended, by and between the Registrant and Derek J. Maetzold.
|
|
|
Offer Letter Agreement, dated October 9, 2015, by and between the Registrant and Federico Monzon, M.D.
|
|
|
Offer Letter Agreement, dated March 2, 2016, by and between the Registrant and Bernhard Spiess.
|
|
|
Offer Letter Agreement, dated November 9, 2017, by and between the Registrant and Frank Stokes.
|
|
|
Standard Office Lease, dated as of October 5, 2015, by and between the Registrant and Merced Restart Phoenix Investors II, LLC.
|
|
|
Office Building Lease, dated as of March 11, 2015, by and between the Registrant and RMG Leasing (a/k/a Cedarwood Professional Building), as amended.
|
II-4
|
Exhibit number
|
Description of document
|
|
Loan and Security Agreement, dated November 30, 2018, by and among the Registrant, Oxford Finance LLC and Silicon Valley Bank.
|
|
|
Exclusive License Agreement, dated as of November 14, 2009, by and between the Registrant and The Washington University.
|
|
|
Non-Employee Director Compensation Policy.
|
|
|
First Amendment to Loan and Security Agreement, dated June 13, 2019, by and among the Registrant, Oxford Finance LLC and Silicon Valley Bank.
|
|
|
Consent of Independent Registered Public Accounting Firm.
|
|
|
23.2†
|
Consent of Cooley LLP. Reference is made to Exhibit 5.1.
|
|
Power of Attorney. Reference is made to the signature page hereto.
|
| † | To be filed by amendment. |
| + | Indicates management contract or compensatory plan. |
| # | Certain portions of this exhibit (indicated by “[***]”) have been omitted as we have determined (i) the omitted information is not material and (ii) the omitted information would likely cause harm to us if publicly disclosed. |
| (b) | Financial statement schedules. |
No financial statement schedules are provided because the information called for is not required or is shown either in the financial statements or the notes thereto.
| Item 17. | Undertakings. |
The undersigned Registrant hereby undertakes to provide to the underwriters at the closing specified in the Underwriting Agreement certificates in such denominations and registered in such names as required by the underwriters to permit prompt delivery to each purchaser.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the Registrant pursuant to the foregoing provisions, or otherwise, the Registrant has been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the Registrant of expenses incurred or paid by a director, officer or controlling person of the Registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the Registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
The undersigned Registrant hereby undertakes that:
| (a) | For purposes of determining any liability under the Securities Act, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the Registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective. |
| (b) | For the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| (c) | That, for the purpose of determining liability under the Securities Act to any purchaser: |
| (i) | If the registrant is subject to Rule 430C, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, |
II-5
however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
| (d) | For the purpose of determining liability under the Securities Act to any purchaser in the initial distribution of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser: |
| (i) | Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424; |
| (ii) | Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant; |
| (iii) | The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and |
| (iv) | Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
II-6
SIGNATURES
Pursuant to the requirements of the Securities Act, the Registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Friendswood, State of Texas, on the 26th day of June, 2019.
|
|
CASTLE BIOSCIENCES, INC.
|
|
|
|
|
|
|
|
By:
|
/s/ Derek J. Maetzold
|
|
|
|
Derek J. Maetzold
President and Chief Executive Officer |
KNOW ALL BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Derek J. Maetzold and Frank Stokes, and each of them, as his or her true and lawful attorneys-in-fact and agents, each with the full power of substitution, for him or her and in his or her name, place or stead, in any and all capacities, to sign any and all amendments to this registration statement (including post-effective amendments), and to sign any registration statement for the same offering covered by this registration statement that is to be effective upon filing pursuant to Rule 462(b) promulgated under the Securities Act, and all post-effective amendments thereto, and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about the premises, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or their substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act, this registration statement has been signed by the following persons in the capacities and on the dates indicated.
|
Signature
|
Title
|
Date
|
|
|
|
|
|
/s/ Derek J. Maetzold
|
President, Chief Executive Officer and
Member of the Board of Directors (Principal Executive Officer) |
June 26, 2019
|
|
Derek J. Maetzold
|
||
|
|
|
|
|
/s/ Frank Stokes
|
Chief Financial Officer
(Principal Financial and Accounting Officer) |
June 26, 2019
|
|
Frank Stokes
|
||
|
|
|
|
|
/s/ Daniel M. Bradbury
|
Chairman of the Board of Directors
|
June 26, 2019
|
|
Daniel M. Bradbury
|
|
|
|
|
|
|
|
/s/ Bonnie H. Anderson
|
Member of the Board of Directors
|
June 26, 2019
|
|
Bonnie H. Anderson
|
|
|
|
|
|
|
|
/s/ Mara G. Aspinall
|
Member of the Board of Directors
|
June 26, 2019
|
|
Mara G. Aspinall
|
|
|
|
|
|
|
|
/s/ G. Bradley Cole
|
Member of the Board of Directors
|
June 26, 2019
|
|
G. Bradley Cole
|
|
|
|
|
|
|
|
/s/ Joseph C. Cook III
|
Member of the Board of Directors
|
June 26, 2019
|
|
Joseph C. Cook III
|
|
|
|
|
|
|
|
/s/ David Kabakoff, Ph.D.
|
Member of the Board of Directors
|
June 26, 2019
|
|
David Kabakoff, Ph.D.
|
|
|
II-7