Filed by Shire plc
pursuant to Rule 425 under the Securities Act
of 1933 and deemed filed pursuant to
Rule 14a-12 of the Securities Exchange
Act of 1934
Subject Company: Baxalta Incorporated
Commission File No. 001-36782
JP Morgan Healthcare Conference Creating the Global Leader in Rare Diseases Flemming Ørnskov, MD, MPH Chief Executive Officer January 12, 2016

Forward Looking Statements Forward-Looking Statements Statements included herein that are not historical facts, including without limitation statements concerning our proposed business combination with Baxalta Incorporated (“Baxalta”) and the timing and financial and strategic benefits thereof, our 20x20 ambition that targets $20 billion in combined product sales by 2020, as well as other targets for future financial results, capital structure, performance and sustainability of the combined company, the combined company’s future strategy, plans, objectives, expectations and intentions, the anticipated timing of clinical trials and approvals for, and the commercial potential of, inline or pipeline products are forward-looking statements. Such forward-looking statements involve a number of risks and uncertainties and are subject to change at any time. In the event such risks or uncertainties materialize, Shire’s results could be materially adversely affected. The risks and uncertainties include, but are not limited to, the following: • • • • the proposed combination with Baxalta may not be completed due to a failure to satisfy certain closing conditions, including any shareholder or regulatory approvals or the receipt of applicable tax opinions; the businesses may not be integrated successfully, such integration may be more difficult, time-consuming or costly than expected, or the expected benefits of the transaction may not be realized; disruption from the proposed transaction may make it more difficult to conduct business as usual or maintain relationships with patients, physicians, employees or suppliers; the combined company may not achieve some or all of the anticipated benefits of Baxalta’s spin-off from Baxter International, Inc. (“Baxter”) and the proposed transaction may have an adverse impact on Baxalta’s existing arrangements with Baxter, including those related to transition, manufacturing and supply services and tax matters; the failure to achieve the strategic objectives with respect to the proposed combination with Baxalta may adversely affect the combined company’s financial condition and results of operations; Shire may not complete its proposed acquisition of Dyax Corp. (“Dyax”) due to the occurrence of an event, change or other circumstances that gives rise to the termination of the relevant merger agreement or the failure to satisfy certain closing conditions, including the Dyax shareholder approval; products and product candidates may not achieve commercial success; product sales from ADDERALL XR and INTUNIV are subject to generic competition; the failure to obtain and maintain reimbursement, or an adequate level of reimbursement, by third-party payers in a timely manner for the combined company’s products may affect future revenues, financial condition and results of operations, particularly if there is pressure on pricing of products to treat rare diseases; supply chain or manufacturing disruptions may result in declines in revenue for affected products and commercial traction from competitors; regulatory actions associated with product approvals or changes to manufacturing sites, ingredients or manufacturing processes could lead to significant delays, an increase in operating costs, lost product sales, an interruption of research activities or the delay of new product launches; the successful development of products in various stages of research and development is highly uncertain and requires significant expenditures and time, and there is no guarantee that these products will receive regulatory approval; the actions of certain customers could affect the combined company’s ability to sell or market products profitably, and fluctuations in buying or distribution patterns by such customers can adversely affect the combined company’s revenues, financial condition or results of operations; investigations or enforcement action by regulatory authorities or law enforcement agencies relating to the combined company’s activities in the highly regulated markets in which it operates may result in significant legal costs and the payment of substantial compensation or fines; adverse outcomes in legal matters and other disputes, including the combined company’s ability to enforce and defend patents and other intellectual property rights required for its business, could have a material adverse effect on the combined company’s revenues, financial condition or results of operations; Shire is undergoing a corporate reorganization and was the subject of an unsuccessful acquisition proposal and the consequent uncertainty could adversely affect the combined company’s ability to attract and/or retain the highly skilled personnel needed to meet its strategic objectives; failure to achieve the strategic objectives with respect to Shire’s acquisition of NPS Pharmaceuticals Inc. or Dyax may adversely affect the combined company’s financial condition and results of operations; the combined company will be dependent on information technology and its systems and infrastructure face certain risks, including from service disruptions, the loss of sensitive or confidential information, cyber-attacks and other security breaches or data leakages that could have a material adverse effect on the combined company’s revenues, financial condition or results of operations; the combined company may be unable to retain and hire key personnel and/or maintain its relationships with customers, suppliers and other business partners; difficulties in integrating Dyax or Baxalta into Shire may lead to the combined company not being able to realize the expected operating efficiencies, cost savings, revenue enhancements, synergies or other benefits at the time anticipated or at all; and • • • • • • • • • • • • • • • other risks and uncertainties detailed from time to time in Shire’s, Dyax’s or Baxalta’s filings with the Securities and Exchange Commission (“SEC”), including those risks outlined in Baxalta’ current Registration Statement on Form S-1, as amended, and in “Item 1A: Risk Factors” in Shire’s Annual Report on Form 10-K for the year ended December 31, 2014. All forward-looking statements attributable to us or any person acting on our behalf are expressly qualified in their entirety by this cautionary statement. Readers are cautioned not to place undue reliance on these forward-looking statements that speak only as of the date hereof. Except to the extent otherwise required by applicable law, we do not undertake any obligation to republish revised forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. 2

Forward Looking Statements Additional Information This communication does not constitute an offer to buy or solicitation of any offer to sell securities or a solicitation of any vote or approval. It does not constitute a prospectus or prospectus equivalent document. This communication relates to the proposed business combination between Shire and Baxalta. The proposed combination will be submitted to Shire’s and Baxalta’s shareholders for their consideration and approval. In connection with the proposed combination, Shire and Baxalta will file relevant materials with (i) the SEC, including a Shire registration statement on Form S-4 that will include a proxy statement of Baxalta and a prospectus of Shire, and (ii) the Financial Conduct Authority (FCA) in the UK, including a prospectus relating to Shire ordinary shares to be issued in connection with the proposed combination and a circular to the shareholders of Shire. Baxalta will mail the proxy statement/prospectus to its shareholders and Shire will mail the circular to its shareholders. This communication is not a substitute for the registration statement, proxy statement/prospectus, UK prospectus, circular or other document(s) that Shire and/or Baxalta may file with the SEC or the FCA in connection with the proposed transaction. INVESTORS AND SECURITY HOLDERS OF SHIRE AND BAXALTA ARE URGED TO READ CAREFULLY THE REGISTRATION STATEMENT, PROXY STATEMENT/PROSPECTUS AND OTHER DOCUMENTS FILED WITH THE SEC AND THE UK PROSPECTUS AND CIRCULAR WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT SHIRE, BAXALTA AND THE PROPOSED TRANSACTION. Investors and security holders may obtain free copies of these documents (when they are available) and other related documents filed with the SEC at the SEC’s web site at www.sec.gov. Investors may request copies of the documents filed with the SEC by Shire by directing a request to Shire’s Investor Relations department at Shire plc, Attention: Investor Relations, 300 Shire Way, Lexington, MA 02421 or to Shire’s Investor Relations department at +1 484 595 2220 in the U.S. and +44 1256 894157 in the UK or by email to investorrelations@shire.com. Investors may request copies of the documents filed with the SEC by Baxalta by directing a request to Mary Kay Ladone at mary.kayklandone@baxalta.com or (224) 948-3371. The statements in this presentation are Shire’s statements and not those of Baxalta or any third party. Certain Information Regarding Participants Shire, Baxalta and their respective directors and executive officers may be deemed participants in the solicitation of proxies in connection with the proposed transaction. You can find information about Shire’s directors and executive officers in Shire’s Annual Report on Form 10-K for the year ended December 31, 2014, which was filed with the SEC on February 24, 2015. You can find information about Baxalta’s directors and executive officers in Baxalta’s registration statement on Form S-1, which was filed with the SEC on September 1, 2015. Additional information regarding the special interests of these directors and executive officers in the proposed transaction will be included in the registration statement, proxy statement/prospectus or other documents filed with the SEC if any when they become available. You may obtain these documents (when they become available) free of charge at the SEC’s web site at www.sec.gov and from Investor Relations at Shire or Baxalta as described above. This communication shall not constitute an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sale of securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction. No offering of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the U.S. Securities Act of 1933, as amended. Trademarks Shire owns or has rights to use the trademarks, service marks and trade names that it uses in conjunction with the operation of its business. Some of the trademarks that Shire owns or has the rights to use that are referenced in this communication include: ADDERALL XR, CINRYZE, ELAPRASE, FIRAZYR, GATTEX/REVESTIVE, INTUNIV, LIALDA, NATPARA, REPLAGAL, PENTASA, VPRIV, VYVANSE and XAGRID. Baxalta states that it owns or has the right to use certain trademarks referenced in this communication, including: ADVATE, ADYNOVATE, ARALAST, FEIBA, FLEXBUMIN, GAMMAGARD, GAMMAGARD LIQUID, GLASSIA, HYQVIA, OBIZUR, ONCASPAR, ONIVYDE, RECOMBINATE, RIXUBIS and SUBCUVIA, which may be registered or used in the United States and other jurisdictions. Basis of Forecasts The Shire forecasts included herein are derived from Shire’s Long Range Plan (the "LRP") and Shire papers subsequently produced as part of the business planning process. Shire produces a long range plan annually. The LRP was updated in March 2015, as part of Shire’s annual planning cycle, and was reviewed by the Board in April 2015. This LRP was subsequently adjusted to reflect revised expectations for SHP625 following trial results in the second quarter of 2015, the Dyax acquisition and other updates for 2015 actual performance. The forecast product sales in this announcement are consistent with the LRP, which is at constant exchange rates, and reflects net sales for each product and key line extensions currently identified as in Phase III, Phase II and those in Phase I included in the LRP as launching before the end of 2020. The forecast product sales included in the LRP are risk-adjusted to reflect Shire’s assessment of the individual probability of launch of products in development, and the probability of success in further life cycle management trials. Estimates for these probabilities are based on industry wide data for relevant clinical trials in the pharmaceutical industry at a similar stage of development. For each pharmaceutical product, there is a range of possible outcomes from clinical development, driven by a number of variables, including safety, efficacy and product labelling. In addition, if a product is approved, the effect of commercial factors including the patient population, the competitive environment, pricing and reimbursement is also uncertain. As a result, the actual net sales achieved by a product over its commercial life will be different, perhaps materially so, from the risk adjusted net sales figures in this announcement and should be considered in this light. The forecast product sales for Baxalta included in this communication have been stated on a constant currency and risk adjusted basis. 3

Global Leader in Rare Diseases with Compelling Strategic/Financial Rationale $1B+ 65% >50 Multiple $1B+ durable, best-in-class franchises Revenue from rare diseases Rare disease pipeline programs >$500M + 16-17% Operating cost synergies alone Additional revenue synergies Attractive non-GAAP effective tax rate Baxalta spinoff from Baxter 8 Companies successfully integrated in past 3 years Highly experienced management team Extensive diligence performed on tax liability 4 Proven Track Record of Integration Compelling Financial Profile and Value Creation Strategic Fit: Global Leader In Rare Diseases •Double-digit top-line growth • Expected accretion to Non-GAAP diluted EPS in 2017 and beyond • Expect combined annual operating cash flow of approximately $6B beginning in 2018 • Attractive ROIC; projected to exceed Shire cost of capital in 2020 • $18 per Baxalta share • Will not jeopardize the tax-free status of the • $18B fully funded bank facility; • Shire plans to de-lever rapidly post-close • Structured to maintain an investment grade credit rating for combined entity Financing Cash consideration ROIC Operating cash flow Earnings accretion Growth profile
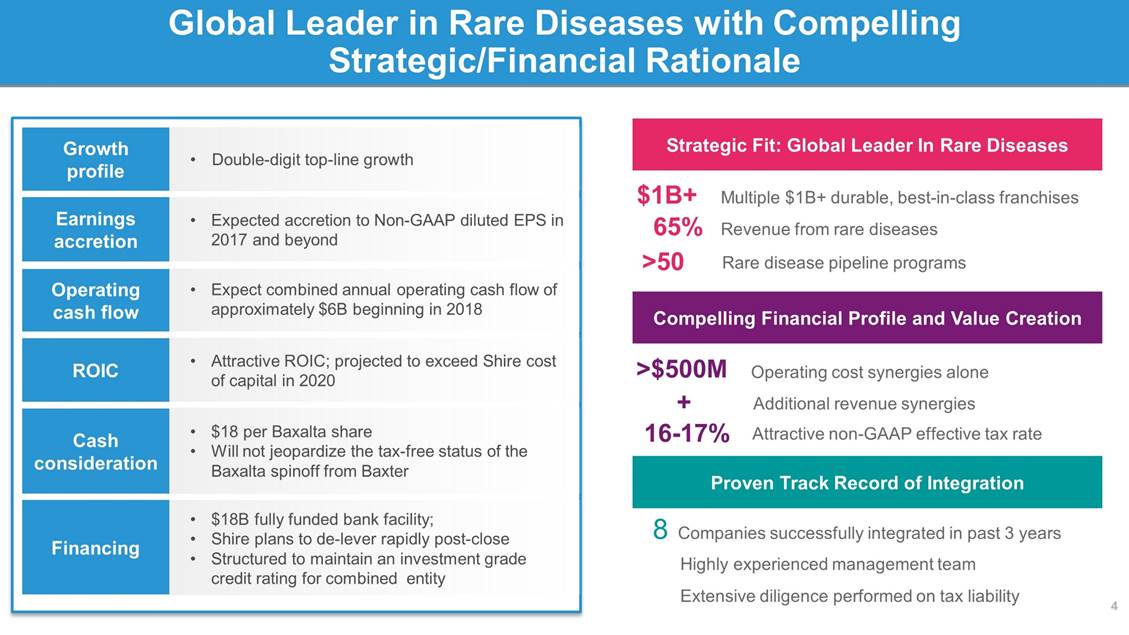
Proposed Transaction with Baxalta Would Add 3 New Areas of Category Leadership Shire Baxalta 5 (1) Pending completion of Dyax acquisition (2) Subject to regulatory approvals. Source: Analyst consensus and Shire management projections. Category and 2015E net salesKey brands Oncology Immunology (~$2.5B) Hematology (~$4B) Category and 2015E product salesKey brands DX-29301 Lifitegrast / GI / Endocrine (~$1.5B) Ophthalmics Neuroscience (~$2B) LSDs (~$1.5B) HAE (~$1B)

Double-digit Top-line Growth and Increasing Focus on Rare Diseases of revenue from rare diseases >$20B Rare diseases Shire Baxalta 2015E(1) Combined 2020E(2) Shire Combined 2015E(1) 6 Baxalta 2015E(1) (1) Based on Shire and Baxalta company guidance. (2) Based on Shire management projections. ~11% CAGR ~$6B ~$6B + 90% 65% ~40% Joint portfolio increases Shire’s proportion Strong, synergistic revenue growth
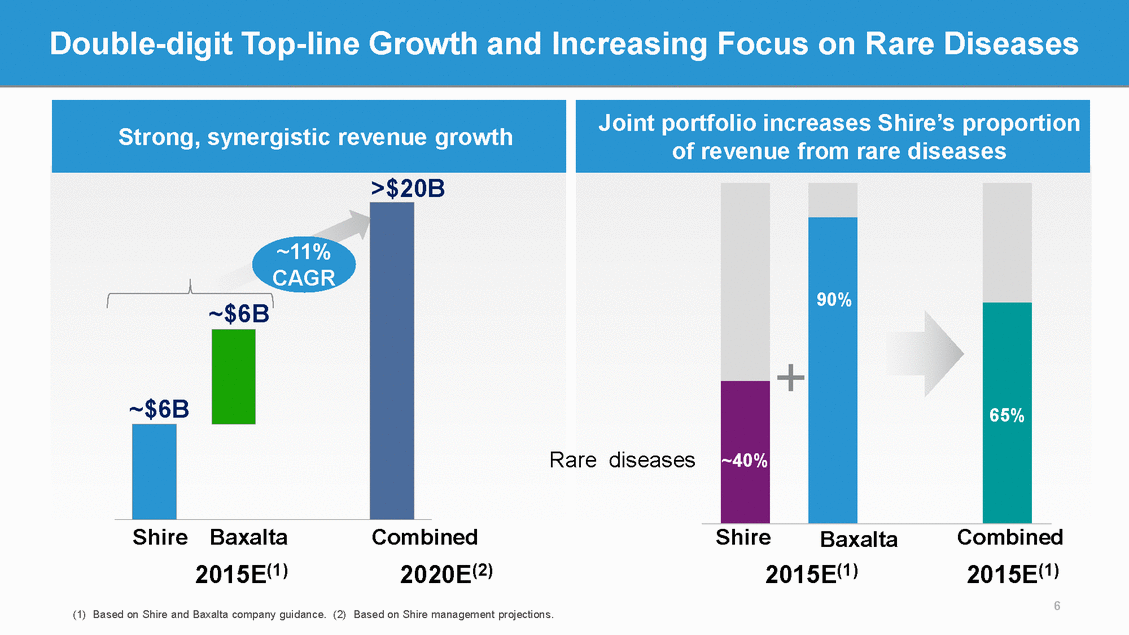
Combined Robust and Innovative Rare Disease Pipeline Preclinical Phase 2 Phase 3(3) SHP620(1) (Maribavir) CMV in transplant patients Registration Phase 1 SHP607 Prevention of ROP Firazyr (Japan) HAE NATPAR (EU) Hypoparathyroidism SHP606 (Lifitegrast) Dry eye disease SHP 621 (Meritage) EoE SHP610 Sanfilippo A SHP609 Hunter IT(ph 2/3) SHP625 Alagille syndrome SHP555 (US) Chronic constipation SHP640 FST-100(1) Infectious conjunctivitis SHP616 (Cinryze) (Japan) HAE prophylaxis SHP625 Primary sclerosing cholangitis DX2930 (1)(6) HAE prophylaxis SHP625 Primary biliary cirrhosis SHP625 Progressive familial intrahepatic cholestasis LDX (Japan) ADHD INTUNIV (Japan) ADHD SHP616 (Cinryze) Acute AMR SHP616 (Cinryze SC) HAE prophylaxis SHP616 (Cinryze) NMO SHP465 ADHD Rare diseases programs Glaucoma Primary immunodeficiency rFVIIa cancer post-gemcitabine Myelofibrosis ALL biosimilar including Dyax. (3) Including Phase 2/3 registrational diligence findings as of December 2015. (5) Ex-US and Based on Shire management projections; including Obizur; (8) currently on hold Baxalta(4) Shire(2) 27 Research programs SHP611 MLD (Ph 1/2) SHP623 (rC1-INH) HAE prophylaxis SHP636 MRT for CF SHP637 MRT for UCD TH / GCH1 Gene pod Parkinson’s subset SHP6088 DEB SHP639 SHP627 Focal segmental glomerulosclerosis SHP630 adRP SHP631 Hunter CNS (Armagen) SHP622 Friedreich’s ataxia SHP616 (Cinryze) Acute neuromyelitis optica SHP626 Non-alcoholic steatohepatitis Anti-TFPI Hemophilia BAX 826 EHL rFVIII PSA BAX 888 FVIII gene therapy SM301 Anti-FcyRIIb IgG Pacritinib AML Pacritinib CLL / PV / myelodysplastic syndrome SM201 / SM211 Anti-FcyRIIb IgG BAX 930 TTP (rADAMTS13) BAX 069 (Imalumab anti-MIF) malignant ascites BAX 335 FIX gene therapy MM-398 (nal-IRI) Pancreatic 1st Line Oncaspar Acute myeloid leukemia SM101 (FcƴRIIb) SLE SM101 (FcƴRIIb) IgA N (Berger) BAX 069 (Imalumab Anti-MIF) colorectal Oncaspar BAX 817Lyophilized ALL ADYNOVATE / BAX 855Rigosertib (Europe)Myelodysplastic syndrome EHL rFVIII PEG BAX 2001 (Rigosertib)BAX 923 High-risk MDSAdalimumab biosimilar Calaspargase pegolBAX 2200 ALLEtanercept (Enbrel) HYQVIA CIDP MM-398 (nal-IRI) Gastric 1st Line 20% IGSC (EU) MM-398 (nal-IRI)(5) Pancreatic Pacritinib Oncaspar (EU) (1) Phase 3 ready. (2) Shire pipeline as of Q3 2015, trials. (4) From Baxalta investor presentations and ex-Taiwan. (6) Pending completion of Dyax acquisition. (7) recently launched products Adynovate, Vonvendi and 7 Key metrics on combined portfolio >60Combined development programs in the pipeline >50Pipeline programs in rare diseases >30New product launches planned with ~$5B sales potential by 2020(7)
Developing and Sustaining Category Leadership

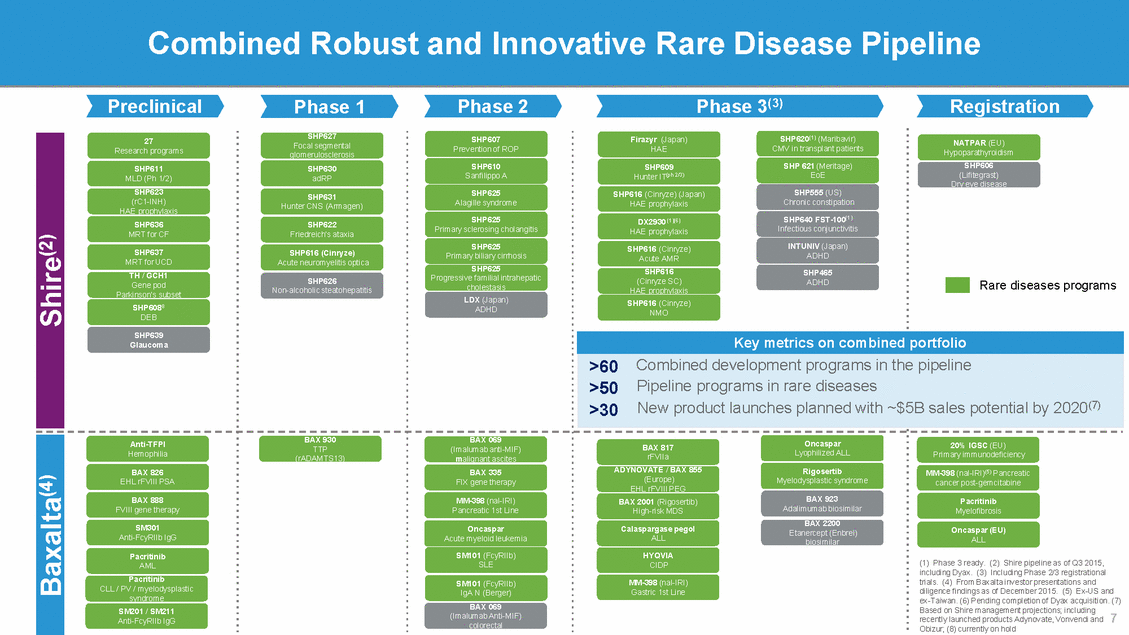
Shire: Industry-leading Rare Disease Capabilities with an Unparalleled Potential for Growth Cutting-edge flexible manufacturing Specialized research and development expertise Targeted, high-touch go-to-market model Category leadership in rare and specialty conditions Global commercial infrastructure High-talent, agile organization Broad channel management capabilities Focused and disciplined BD capability Lean G&A model 9
Shire’s Current 5 Areas of Category Leadership 10 1 Pending completion of Dyax transaction Major marketed products Selected late-stage pipeline programs DX-29301 (HAE prophylaxis) SHP609 (Hunter intrathecal) SHP465 (ADHD) Lifitegrast (DED)SHP640 (Conjunctivitis) SHP607 (ROP) SHP621 (EOE) SHP625 (PFIC) GI / Endocrine Ophthalmics Neuroscience LSD HAE

Category Leadership: Ophthalmics Preclinical Phase 1 Phase 2 Phase 3 Registration Dry Eye Disease (lifitegrast) Bacterial and Viral Infectious Conjunctivitis (SHP640) Retinopathy of Prematurity (SHP607) adRP1 (SHP630) Glaucoma (SHP639) 11 1. Autosomal dominant retinitis pigmentosa
Dry-Eye Disease (DED): A Complex Ocular Surface Condition • A chronic and progressive multifactorial disease of the tears and ocular surface • Associated with inflammation that may eventually lead to damage to the surface of the eye • Often diagnosed by patient reported symptoms (stinging, burning, blurred vision) and/or objective signs (Schirmer test, staining, etc.) 12
DED: One of the Most Common Eye Diseases in the U.S.1 ~29 million affected Incidence may be growing: aging population, menopause, use of other irritating medications, improved awareness/diagnosis 14 million diagnosed2 ~1/3 of diabetics may have DED3 7-8 million moderate/ patients2 severe Rx treatments: ~$1B4 Existing 1) The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007). - Ocul Surf. 2007 Apr;5(2):93-107. Review. Internal Shire market research No treatment indicated for signs and symptoms Possibility of ocular burning5 May need several months before onset of effect5 2) 3) 4) 5) Cafrey BE, et al, Adv Exp Med Biol 1 13 998 Allergan 2014 10-k Restasis Prescribing Information

Lifitegrast: Specifically Designed to Address DED Inflammation Small-molecule selected from 1500 candidate molecules Built specifically to block LFA-1 / ICAM-1 interaction ICAM-1 has been shown to be expressed at higher levels in patients with dry eye Acts as an ICAM-1 decoy and selectively binds with LFA-1 Thought to inhibit the release of inflammatory cytokines, T-cell migration, T-cell proliferation, and T-cell adhesion Lifitegrast is designed to break the self-perpetuating cycle of cytokine release and T-cell activation/mobilization that results in chronic inflammation and ultimately ocular surface damage T-cell activation blocked through blockade of ICAM-1/LFA-1 Interaction T-cell Proliferation and Cytokine Release T-cell Activation Note: Ref.:USAN/INN (Jan 2012) ; Zhong et al., Med Chem Lett (2012) 14 Lifitegrast

Lifitegrast: Potential to be 1st Treatment Indicated for Signs AND Symptoms1 Comprehensive clinical program conducted in ~2,000 patients OPUS-3 also met secondary endpoint of statistically significant impact on Symptoms after 14 days of treatment 1 2 Subject to approval by regulatory authorities OPUS-1 met primary endpoint against signs but not symptoms; OPUS-2 met primary endpoint against symptoms but not signs 15 Phase Study Number Indication Subjects Endpoints 2 Phase 2 Dry Eye 230 Signs 3 OPUS-1 Dry Eye 588 Signs/Symptoms2 3 OPUS-2 Dry Eye 718 Signs/Symptoms2 3 OPUS-3 Dry Eye 711 Symptoms 3 SONATA Dry Eye 332 Safety
Lifitegrast: Next Steps in 2016 Q3: 2016 • Potential approval and launch Q1: 2016 • Resubmission in response to FDA’s complete letter response Build-out commercial infrastructure Implement disease awareness campaign Continue engagement with international regulatory authorities Note: Subject to approval by regulatory authorities 16
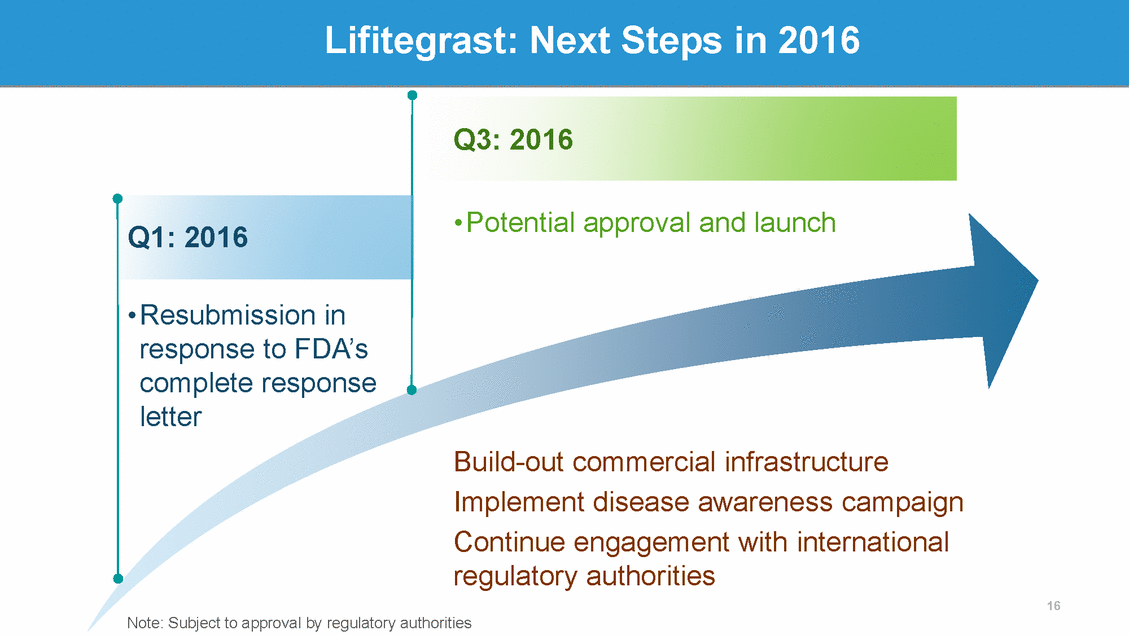
SHP607: Retinopathy of Prematurity (ROP) Mature Healthy Retina2 (Birth at 40 Weeks) • ROP is caused by abnormal blood vessel growth in the retina that can lead to retinal detachment and blindness ROP primarily affects premature babies, is one of the most common causes of visual loss in childhood, and can lead to lifelong vision impairment and blindness1 60k babies born prematurely each year in the U.S./EU are at risk of developing ROP • Premature Retina with ROP2 (Birth at 28 Weeks GA) • 1 https://nei.nih.gov/health/rop/rop 2 Examination at 34 weeks post-conceptual age 17
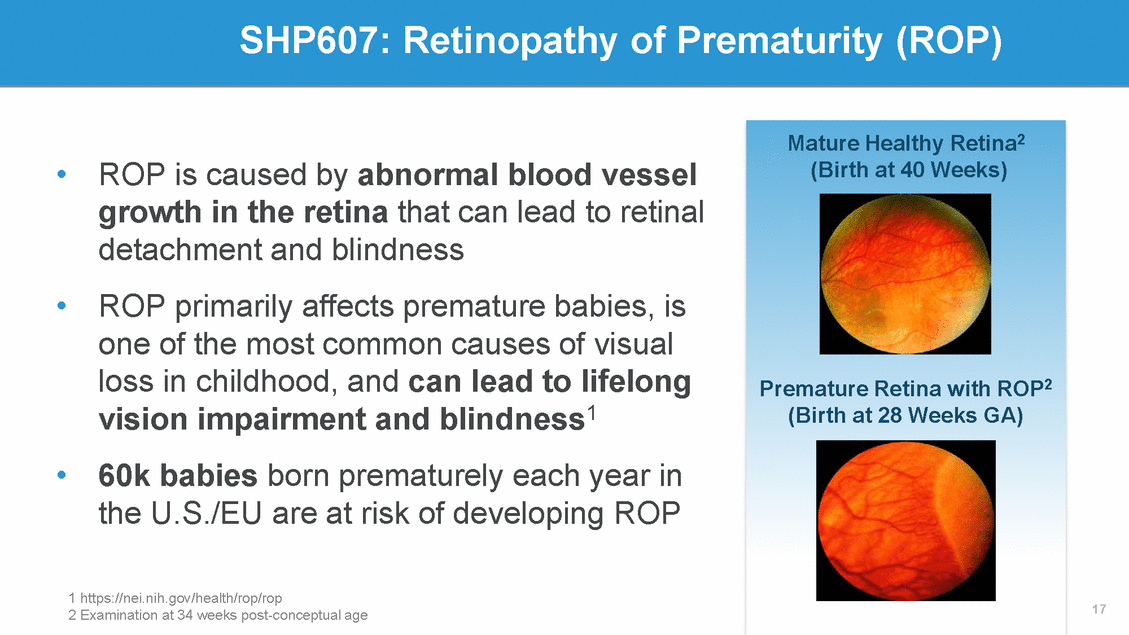
SHP607 (IGF-1/IGF-1 BP) Replaces Low Levels of Intra-Uterine IGF-1 • Low levels of intra-uterine Insulin Like Growth Factor-1 (IGF-1) correlate with higher rates of ROP • IV infusions of SHP607 beginning w/in the first 24-48 hours of life replaces low levels of IGF-1 until endogenous production of IGF-1 begins at ~30 weeks of gestational age. 18
SHP607: Phase 2 Top-Line Data Expected in Q2 2016 Q2: 2016 Phase 2 top-line results Q1: 2016 Phase 2 fully enrolled Primary Endpoint: Prevention Secondary Endpoints: of ROP • • • • Incidence of Bronchopulmonary Dysplasia (BPD) Time to NICU discharge Brain volume 19 Growth
Category Leadership: Hereditary Angioedema SHP616 (CINRYZE SC) HAE Prophylaxis DX-2930 DX-29301 Novel antibody therapy Less frequent subcutaneous administration Very high efficacy in early trials Phase 3 development Cinryze and Firazyr Ability to address both prophylactic (Cinryze) and acute (Firazyr) HAE needs Prophylaxis requires IV administration Cinryze SC Novel delivery method Subcutaneous formulation to allow simpler patient-administered prophylaxis Phase 3 trials underway • • • • • • • • • 1 Pending completion of proposed Dyax acquisition 20

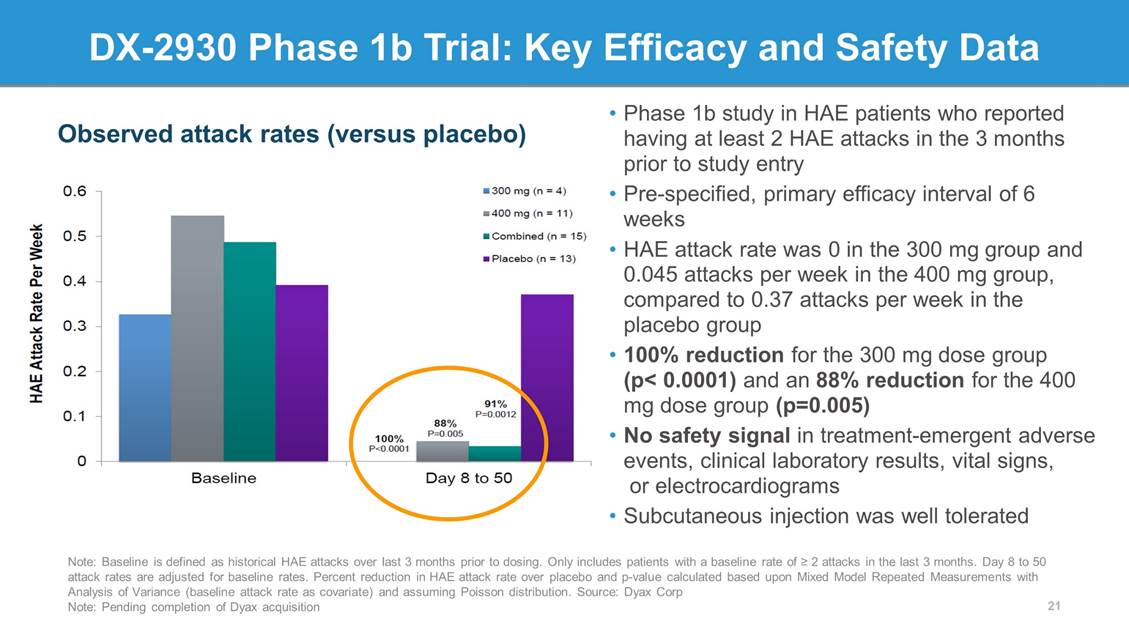
DX-2930 Phase 1b Trial: Key Efficacy and Safety Data • Phase 1b study in HAE patients who reported having at least 2 HAE attacks in the 3 months prior to study entry • Pre-specified, primary efficacy interval of 6 weeks • HAE attack rate was 0 in the 300 mg group and 0.045 attacks per week in the 400 mg group, compared to 0.37 attacks per week in the placebo group • 100% reduction for the 300 mg dose group (p< 0.0001) and an 88% reduction for the 400 mg dose group (p=0.005) • No safety signal in treatment-emergent adverse events, clinical laboratory results, vital signs, or electrocardiograms • Subcutaneous injection was well tolerated Observed attack rates (versus placebo) Note: Baseline is defined as historical HAE attacks over last 3 months prior to dosing. Only includes patients with a baseline rate of > 2 attacks in the last 3 months. Day 8 to 50 attack rates are adjusted for baseline rates. Percent reduction in HAE attack rate over placebo and p-value calculated based upon Mixed Model Repeated Measurements with Analysis of Variance (baseline attack rate as covariate) and assuming Poisson distribution. Source: Dyax Corp Note: Pending completion of Dyax acquisition 21
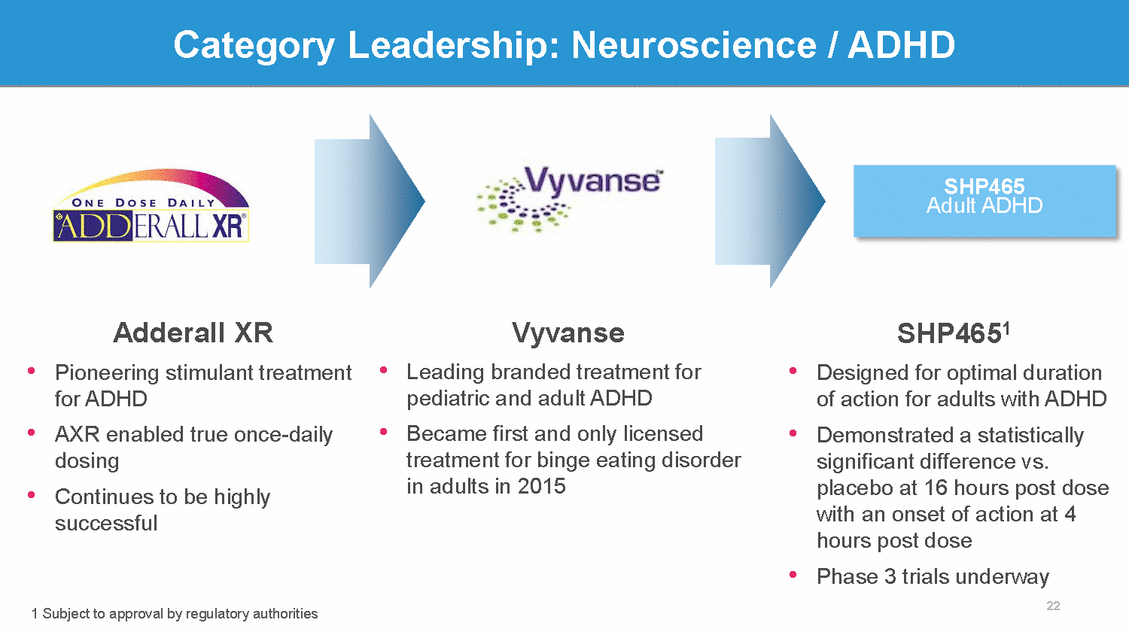
Category Leadership: Neuroscience / ADHD SHP465 Adult ADHD Adderall XR Pioneering stimulant treatment for ADHD AXR enabled true once-daily dosing Continues to be highly successful Vyvanse Leading branded treatment for pediatric and adult ADHD Became first and only licensed treatment for binge eating disorder in adults in 2015 SHP4651 Designed for optimal duration of action for adults with ADHD Demonstrated a statistically significant difference vs. placebo at 16 hours post dose with an onset of action at 4 hours post dose Phase 3 trials underway 22 • • • • • • • • 1 Subject to approval by regulatory authorities
Category Leadership: GI / Endocrine – SHP621 for EoE • • • • Eosinophilic Esophagitis (EoE): chronic immune-mediated condition with est. >150k patients in the US SHP621: Oral budenoside formulated in a viscous suspension 12-week, 93-patient Phase 2 trial1 met all endpoints Phase 3 trial underway 23 1 Randomized, double-blinded, placebo-controlled trial in 25-30 US EoE centers. 10ml/dose of SHP621 was given BID during study period and QD thereafter

2016 Outlook

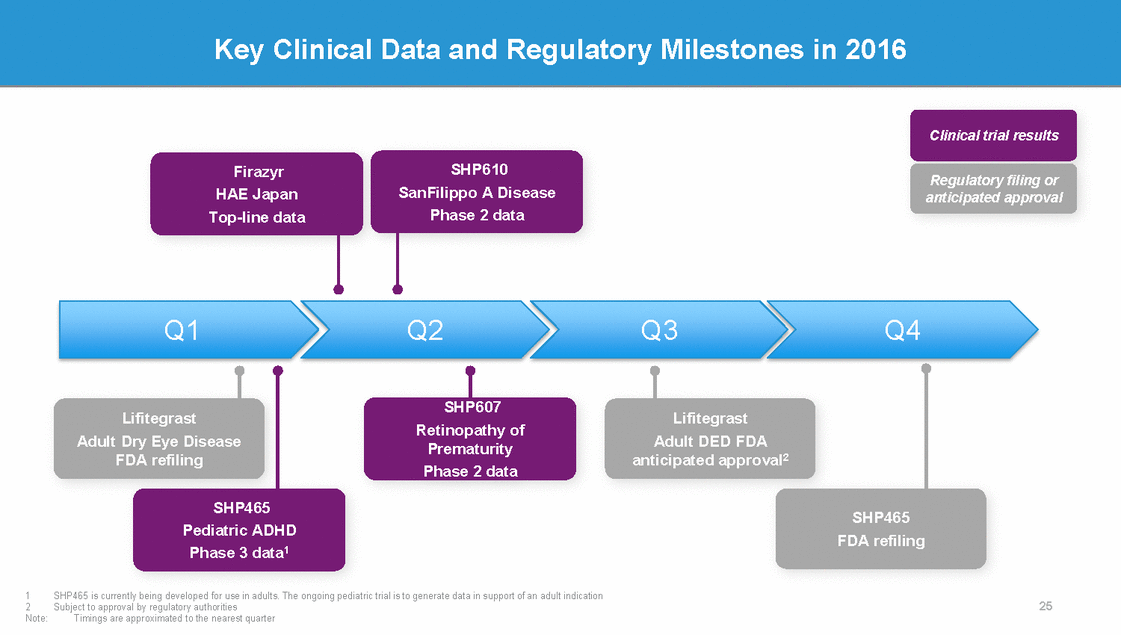
Key Clinical Data and Regulatory Milestones in 2016 Clinical trial results SHP610 Firazyr HAE Japan Top-line data Regulatory filing or anticipated approval SanFilippo A Disease Phase 2 data Q1 Q2 Q3 Q4 SHP607 Lifitegrast Lifitegrast Retinopathy of Prematurity Phase 2 data Adult Dry Eye Disease FDA refiling Adult DED FDA anticipated approval2 SHP465 SHP465 Pediatric ADHD FDA refiling Phase 3 data1 1 2 Note: SHP465 is currently being developed for use in adults. The ongoing pediatric trial is to generate data in support of an adult indication Subject to approval by regulatory authorities Timings are approximated to the nearest quarter 25
2016: A Year of Growth and Investment for Shire Expected return to double-digit topline growth rates Baxalta1 Integrate Advance SHP607 (ROP), DX-29303 (HAE) and SHP465 (ADHD) Expand and Launch lifitegrast2 Largest-ever number of Phase 3 trials (>12) strengthen manufacturing 1 Pending completion of Baxalta acquisition 2 Subject to approval by regulatory authorities 3 Pending completion of Dyax integration 26
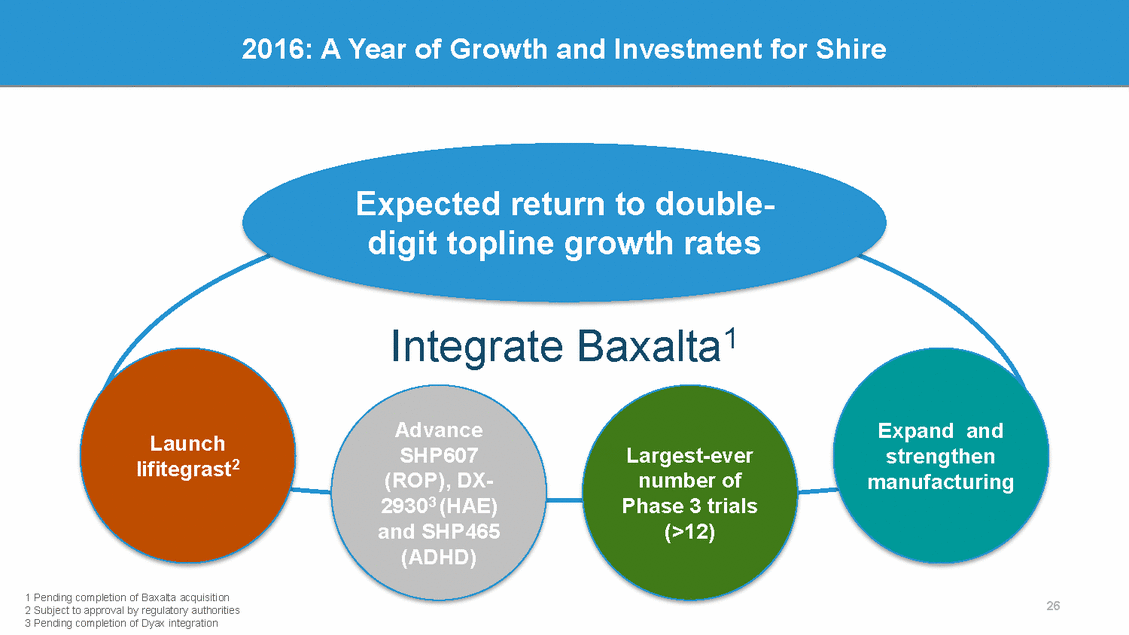
LShire Our purpose We enable people with life-altering conditions to lead better lives.

Appendix

The Shire/Baxalta Combination Will Not Cause the Spin-off of Baxalta by Baxter to be Taxable • • The spin-off of Baxalta stock by Baxter (“Spin-off”) was designed to be tax-free Shire has been advised by Cravath, Swaine & Moore LLP that the Shire/Baxalta combination (“Combination”) will not cause the Spin-off to be taxable as a “device” under IRC § 355(a)(1)(B) or a change of control under IRC § 355(e) if: • • • it was motivated by a strong business purpose; at the time of the Spin-off neither Baxter nor Baxalta intended or expected that Baxalta would be acquired; and the Spin-off and the Combination are not parts of a common plan • Shire took a measured approach in first presenting an acquisition proposal to Baxalta, before due diligence was possible Cravath subsequently conducted extensive due diligence on the background of the Spin-off, addressing whether the three conditions listed above are satisfied, including: • • • • • review of internal Baxter and Baxalta documents discussions with Baxter and Baxalta management Officer’s Certificates from Baxter, Baxalta and Shire determination that Shire had no engagement with Baxter or Baxalta about a combination at any time prior to the Spin-off • Based on this diligence, Cravath has provided Shire a “will” level tax opinion to the effect that the Combination will not cause the Spin-off to be taxable • A “will” level opinion of counsel is the strongest form of tax opinion: Based on the facts and circumstances and current US law there are no reasonable, compelling technical arguments that would lead to a different conclusion • No ruling from the IRS is available for this type of Combination • • the Combination falls within the IRS’s “no rulings” policy Companies ordinarily proceed on the basis of tax opinions concerning issues within the IRS’s “no rulings” policy

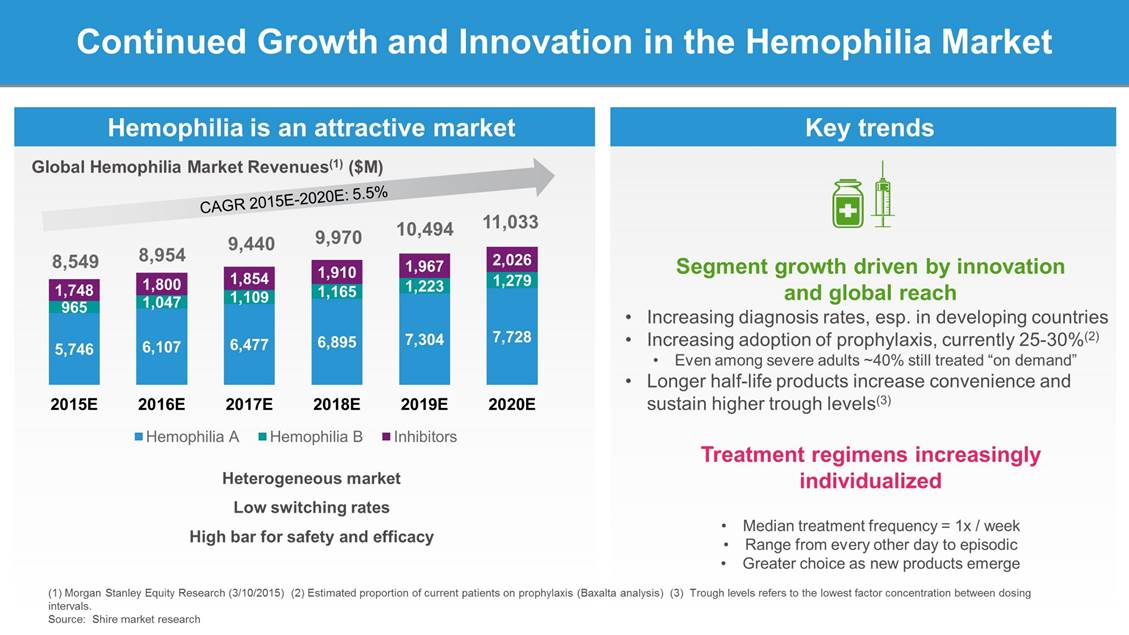
Continued Growth and Innovation in the Hemophilia Market Global Hemophilia Market Revenues(1) ($M) 11,033 10,494 9,970 9,440 8,954 8,549 Segment growth driven by innovation and global reach Increasing diagnosis rates, esp. in developing countries Increasing adoption of prophylaxis, currently 25-30%(2) • Even among severe adults ~40% still treated “on demand” Longer half-life products increase convenience and sustain higher trough levels(3) • • • 2015E 2016E 2017E 2018E 2019E Inhibitors 2020E Hemophilia A Hemophilia B Treatment regimens increasingly individualized Heterogeneous market Low switching rates High bar for safety and efficacy • • • Median treatment frequency = 1x / week Range from every other day to episodic Greater choice as new products emerge (1) Morgan Stanley Equity Research (3/10/2015) (2) Estimated proportion of current patients on prophylaxis (Baxalta analysis) (3) Trough levels refers to the lowest factor concentration between dosing intervals. Source: Shire market research 1,748 965 5,746 1,800 1,047 6,107 1,854 1,109 6,477 1,910 1,165 6,895 1,967 1,223 7,304 2,026 1,279 7,728 Key trends Hemophilia is an attractive market
Breadth, Established Position, and Innovation Propel Immunoglobulins (IG) Business Baxalta’s Broad IG Portfolio The Only Once-A-Month IGSC Future Flagship Brand Expanding Baxalta’s IGSC Platform With 20% Concentration Baxalta’s Cornerstone IG brand The Only IgA < 1μg/ml Baxalta’s Platform for Growth in IG • • • • Broadest IG portfolio in the industry - Durable assets supported by specialized competitive requirements Expected 9%(1) annual sales growth in 2015 with over 10 years of on-market experience - Capacity expansion unlocks potential growth Patient demand for IG products remains robust and expected to continue for the foreseeable future Best-in-class product as the only once-monthly SubQ IG brand Strong initial U.S. uptake following Q4 2014 launch Continued sales growth expected from: • Increasing share of SubQ IG market Converting current IVIG patients to SubQ Additional future indications (e.g., neurology) - - - - • Geographic expansion (1) Growth rate compares to 2014 pro forma sales as guided by Baxalta in its Q3 2015 Performance Update Source: Baxalta investor presentations, Shire management projections and analysis. HyQvia launch driving growth Leading market position
