Exhibit 99.1

Contacts: | ||
Jody Cain | ||
LHA Investor Relations | ||
310-691-7100 | ||
jcain@lhai.com | ||
PDL BioPharma Reports 2020 First Quarter Financial Results
Provides Update to Asset Monetization Plan
Discusses Impact of COVID-19 on Operations and Monetization Plan
- Conference Call with Slides Begins at 4:30 p.m. Eastern Time Today -
INCLINE VILLAGE, Nev. (May 7, 2020) - PDL BioPharma, Inc. (“PDL” or “the Company”) (Nasdaq: PDLI) reports financial results for the three months ended March 31, 2020 and provides a business update:
In March 2020, the Company announced that its Board of Directors (the “Board”) approved a Plan of Complete Liquidation and passed a resolution to seek stockholder approval at its next Annual Meeting of Stockholders to dissolve the Company under Delaware state law in the event the Board concludes that the whole Company sale process is unlikely to maximize the value that can be returned to the stockholders. The Company has not set a definitive timeline to file for dissolution and intends to pursue its monetization strategy in a disciplined and cost-effective manner seeking to maximize returns to stockholders. The Company recognizes, however, that accelerating the timeline, while continuing to seek to optimize asset value, could increase returns to stockholders due to reduced general and administrative (“G&A”) expenses as well as potentially providing faster returns to stockholders. While the Company cannot provide a definitive timeline for the liquidation process, it has been targeting the end of 2020 for completing the monetization of its key assets. However, the Company recognizes that the duration and extent of the public health issues related to the COVID-19 pandemic make it possible, and perhaps probable, that the timing may be delayed. As announced previously, the Company has engaged financial advisors and initiated processes either to sell these assets separately or to transact the Company as a whole.
“We continue to execute on the strategy of monetizing our assets to unlock the full value of the company for our stockholders,” said Dominique Monnet, president and CEO of PDL. “Our plan is to follow a disciplined approach with a focus on maximizing net proceeds. We remain confident in the high quality of our assets, and we believe that they are attractive acquisition targets.
“Earlier this week we announced Board of Director approval for a distribution of all of PDL’s shares of Evofem Biosciences common stock via a special one-time dividend to PDL stockholders as our first distribution under the Plan of Complete Liquidation,” he added “We previously stated the ambitious goal of completing the monetization of our key assets by the end of 2020. While we are encouraged by our progress, we recognize that the impact of the COVID-19 pandemic on our assets and the businesses of potential buyers of those assets could cause some delays, which makes it possible, and perhaps probable, that the timing of the sale or sales may be delayed. Again, our intent is to pursue monetization in a disciplined and cost-effective manner and to distribute the net proceeds to stockholders in a tax-efficient manner in the form of share repurchases and dividends, or by other means.”
Discontinued Operations Classified as Assets Held for Sale
As a result of these decisions and the actions put in place in the first quarter of 2020, at March 31, 2020 the assets held for sale and discontinued operations criteria were met for the Company’s royalty assets and for Noden Pharma, its pharmaceutical segment. The royalty assets are a component of the Income Generating Assets segment.
1
During the period in which a component meets the assets held for sale and discontinued operations criteria, an entity must present the assets and liabilities of the discontinued operation separately in the asset and liability sections of the balance sheet for the current and comparative reporting periods. The prior period balance sheet is reclassified for the held for sale items. For statements of operations, the current and prior periods report the results of operations of the component in discontinued operations. While the current period and prior period are presented herein on a comparative basis in accordance with U.S. Generally Accepted Accounting Principles (“GAAP”), the presentation has changed from the reporting of GAAP financial results in our fourth quarter 2019 earnings release.
First Quarter Financial Highlights
• | Total revenues were $6.0 million, consisting primarily of LENSAR product revenue. |
• | LENSAR revenues were $6.0 million, a decrease of 11% over the prior-year period, with procedure volume declining 6%. |
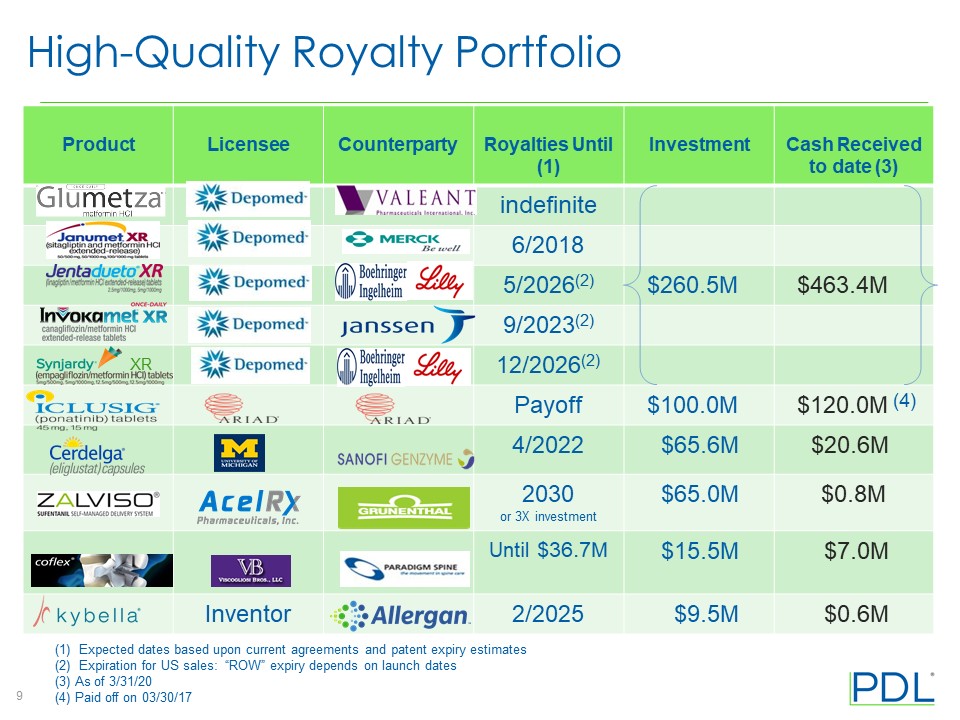
• | Net cash from all royalty rights was $13.6 million, up 8% from $12.6 million for the prior-year period. |
• | U.S. market share for branded Tekturna® and the authorized generic of Tekturna of approximately 68% at March 31, 2020 declined from 73% as of December 31, 2019. |
• | GAAP net loss was $31.7 million. Non-GAAP net loss was $6.7 million. A reconciliation of GAAP to non-GAAP financial results can be found in Table 4 at the end of this news release. |
Revenue Highlights
• | Total revenues for the first quarter of 2020 were $6.0 million and consisted primarily of LENSAR product revenue. |
◦ | Product revenue from LENSAR was $6.0 million, an 11% decrease from the first quarter of 2019. LENSAR procedure volume for the first quarter of 2020 declined 6% from the prior-year period, primarily due to lower system sales and procedures driven by the negative impact of the COVID-19 pandemic and the associated deferral of elective medical procedures, primarily in South Korea and China. While LENSAR U.S. operating results for the first quarter of 2020 were not impacted as significantly by the COVID-19 pandemic, beginning in late March and into the second quarter of 2020 the pandemic resulted in the cancellation of practically all elective cataract surgeries. LENSAR operating results are expected to improve as elective medical procedures gradually open throughout the remainder of 2020. |
Operating Expense Highlights
• | Operating expenses from continuing operations of the Company include G&A expenses for corporate overhead as these costs have historically not been allocated to individual segments. |
• | Operating expenses for the first quarter of 2020 were $37.9 million, a $23.0 million increase from the first quarter of 2019. The increase was primarily a result of an acceleration of equity awards and the accrual for cash severance and retention payments under our wind-down retention plan totaling $18.7 million, and for increased professional service costs. The vesting of equity awards was accelerated when the Board approved a Plan of Complete Liquidation in February 2020 as this action constituted a change in control. |
• | There were decreases in cost of product revenue and sales and marketing expenses in our Medical Devices segment due to a decline in revenue, while G&A and research and development expenses reflected modest increases. |
• | Net loss from continuing operations for the first quarter of 2020 was $31.8 million, a $23.3 million increase from the first quarter of 2019. |
Discontinued Operations Highlights
• | Discontinued operations consist of the following items: |
◦ | Net royalty revenues from acquired royalty rights, which include cash royalties received and a change in fair value of the royalty rights assets, were $9.4 million compared with $12.3 million in the prior-year period. The decrease is primarily related to the anticipated decrease in fair value of the royalty rights for the Type 2 diabetes products acquired from Assertio Therapeutics. PDL received $13.6 million in net cash from all its royalty rights in the first quarter of 2020, up from $12.6 million in the prior-year period. See Table 3 for a rollforward of royalty assets for the first quarter of 2020 compared with the comparable period in 2019. |
2
▪ | The asset held for sale classification requires the Company to record the estimated cost to sell the asset as a deduction to the carrying value of the asset. In the first quarter of 2020, the Company recorded $6.0 million as the estimated cost to sell the royalty assets. |
◦ | Product revenue from Noden was $15.0 million compared with $20.0 million in the prior-year period. Revenues for the U.S. and the rest of the world were $3.9 million and $11.1 million, respectively, compared with $12.2 million and $7.8 million, respectively, in the prior-year period. The decline in U.S. revenue is primarily a result of the launch of an authorized generic of Tekturna as well as the launch of a third-party generic form of aliskiren in March 2019. U.S. market share for branded Tekturna and authorized generic of Tekturna of approximately 68% declined from the market share of 73% as of December 31, 2019. |
▪ | In the first quarter of 2020, the Company recorded $1.9 million as the estimated cost to sell Noden. |
• | Net loss from discontinued operations for the first quarter of 2020 was $0.2 million, a $15.3 million decrease from the first quarter of 2019. The decrease was primarily due to the estimated cost to sell the assets classified as held for sale of $7.9 million and the write down of Noden to reflect fair value upon its reclassification as an asset held for sale. |
Other Financial Highlights
• | As of March 31, 2020, the Company’s investment in Evofem had a market value of $82.6 million, a decrease of $13.8 million from December 31, 2019. The Company acquired its investment in Evofem in two tranches in the second quarter of 2019, for a total of $60.0 million. |
• | On a GAAP basis, the net loss attributable to PDL’s stockholders for the first quarter of 2020 was $31.7 million, or $0.26 per share, compared with GAAP net income attributable to PDL’s stockholders of $6.7 million, or $0.05 per diluted share, for the prior-year period. Non-GAAP net loss attributable to PDL’s stockholders was $6.7 million for the first quarter of 2020, compared with non-GAAP net income of $11.9 million for the first quarter of 2019. |
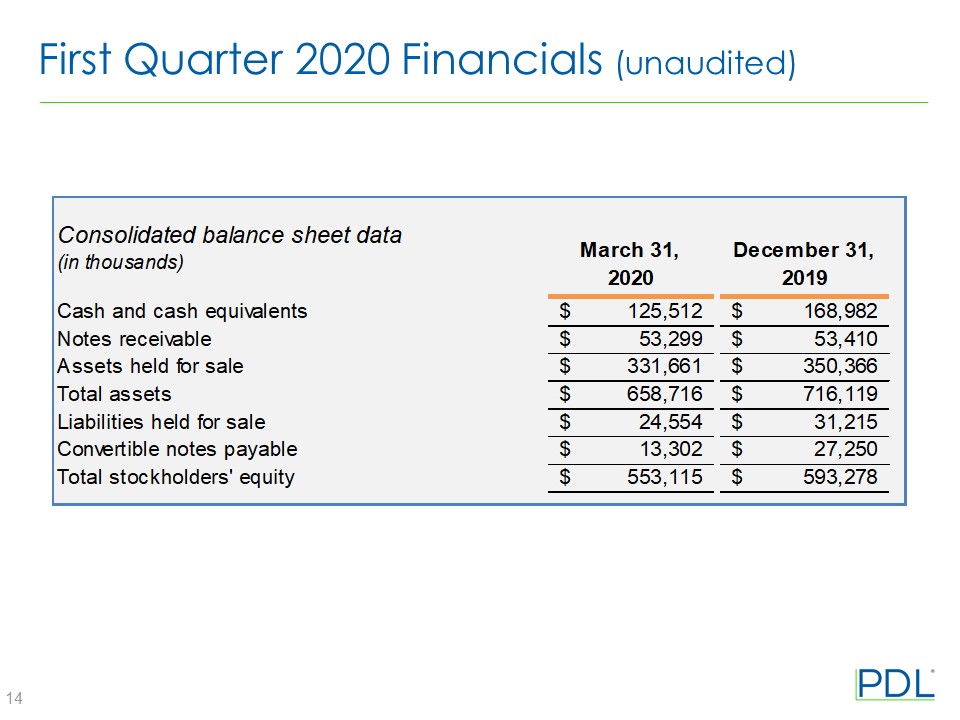
• | PDL had cash and cash equivalents from continuing operations of $125.5 million as of March 31, 2020, compared with $169.0 million as of December 31, 2019. |
◦ | The $43.5 million reduction was primarily the result of common stock repurchases of $19.2 million, the net cash used for the repurchase of convertible debt of $18.0 million and net cash used in operations of $14.6 million. This reduction was partially offset by the proceeds from royalty rights of $13.6 million. |
Stock Repurchase Programs
• | In January 2020, PDL began repurchasing shares of its common stock in the open market pursuant to the |
10b5-1 program entered into in December 2019. In the first quarter of 2020, the Company acquired 6.3 million shares for $20.3 million, at an average cost of $3.20 per share, including commissions.
• | Under this same program, in the first quarter of 2020, the Company also repurchased $15.9 million par value of convertible notes. |
• | As of April 30, 2020, the Company had approximately 116.5 million shares of common stock outstanding. |
Conference Call and Webcast
PDL will hold a conference call to discuss financial results and provide a business update at 4:30 p.m. Eastern time today. Slides to accompany the conference call will be available in the Investor Relations section of https://www.pdl.com/.
To access the live conference call via phone, please dial 844-535-4071 from the U.S. and Canada or 706-679-2458 internationally. The conference ID is 7238226. A telephone replay will be available for one week beginning approximately one hour after the completion of the call and can be accessed by dialing 855-859-2056 from the U.S. and Canada or 404-537-3406 internationally. The replay passcode is 7238226.
To access the live and subsequently archived webcast of the conference call, go to the Investor Relations section of https://www.pdl.com/ and select “Events & Presentations.”
About PDL BioPharma, Inc.
Throughout its history, PDL's mission has been to improve the lives of patients by aiding in the successful development of innovative therapeutics and healthcare technologies. PDL BioPharma was founded in 1986 as Protein Design Labs, Inc. when it pioneered the humanization of monoclonal antibodies, enabling the discovery of a new generation of targeted treatments that
3
have had a profound impact on patients living with different cancers as well as a variety of other debilitating diseases. In 2006, the Company changed its name to PDL BioPharma, Inc.
As of December 2019, PDL ceased making additional strategic transactions and investments and is pursuing a formal process to unlock the value of its portfolio by monetizing its assets and ultimately distributing net proceeds to stockholders.
For more information please visit https://www.pdl.com/
NOTE: PDL, PDL BioPharma, the PDL logo and associated logos and the PDL BioPharma logo are trademarks or registered trademarks of, and are proprietary to, PDL BioPharma, Inc. which reserves all rights therein. Noden, Noden Pharma, Tekturna, Tekturna HCT, Rasilez and Rasilez HCT and associated logos are trademarks or registered trademarks of, and are proprietary to, Noden Pharma DAC, which reserves all right therein. LENSAR and associated logos are trademarks or registered trademarks of, and are proprietary to, LENSAR, Inc., which reserves all rights therein.
Forward-looking Statements
This press release contains “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, including as it relates to the Company’s proposed Evofem stock distribution and plan of liquidation. Each of these forward-looking statements involves risks and uncertainties. Actual results may differ materially from those, express or implied, in these forward-looking statements. Important factors that could impair the value of the Company’s assets and business, including the implementation or success of the Company’s monetization strategy/plan of complete liquidation, are disclosed in the risk factors contained in the Company’s Annual Report on Form 10-K, filed with the Securities and Exchange Commission (the “SEC”) on March 11, 2020, and subsequent filings. All forward-looking statements are expressly qualified in their entirety by such factors. We do not undertake any duty to update any forward-looking statement except as required by law.
Important Additional Information and Where to Find It
The Company plans to file a proxy statement (the “2020 Proxy Statement”) with the SEC in connection with the solicitation of proxies for the 2020 Annual Meeting, together with a WHITE proxy card. STOCKHOLDERS ARE URGED TO READ THE 2020 PROXY STATEMENT (INCLUDING ANY AMENDMENTS OR SUPPLEMENTS THERETO) AND ANY OTHER RELEVANT DOCUMENTS THAT THE COMPANY WILL FILE WITH THE SEC CAREFULLY IN THEIR ENTIRETY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION.
Stockholders will be able to obtain, free of charge, copies of the 2020 Proxy Statement, any amendments or supplements thereto and any other documents (including the WHITE proxy card) when filed by the Company with the SEC in connection with the 2020 Annual Meeting at the SEC’s website (http://www.sec.gov), at the Company’s website (http://investor.pdl.com/investor-relations/sec-filings) or by contacting Okapi Partners by phone (for stockholders, banks and brokers) at 877-259-6290 or (all others outside the U.S.) at 212-297-0720, by email at info@okapipartners.com or by mail at Okapi Partners LLC, 1212 Avenue of the Americas, 24th Floor, New York, NY 10036.
Participants in the Solicitation
The Company, its directors and certain of its executive officers and other employees may be deemed to be participants in the solicitation of proxies from stockholders in connection with the 2020 Annual Meeting. Additional information regarding the identity of these potential participants, none of whom owns in excess of one percent (1%) of the Company’s shares, and their direct or indirect interests, by security holdings or otherwise, will be set forth in the 2020 Proxy Statement and other materials to be filed with the SEC in connection with the 2020 Annual Meeting. Information relating to the foregoing can also be found in the Company’s definitive proxy statement for its 2019 annual meeting of stockholders (the “2019 Proxy Statement”), filed with the SEC on April 30, 2019. To the extent holdings of the Company’s securities by such potential participants (or the identity of such participants) have changed since the information printed in the 2019 Proxy Statement, such information has been or will be reflected on Statements of Change in Ownership on Forms 3 and 4 filed with the SEC. You may obtain free copies of these documents using the sources indicated above.
4
TABLE 1
PDL BIOPHARMA, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS DATA
(In thousands, except per share amounts)
Three Months Ended March 31, | ||||||||
2020 | 2019 | |||||||
Revenues | ||||||||
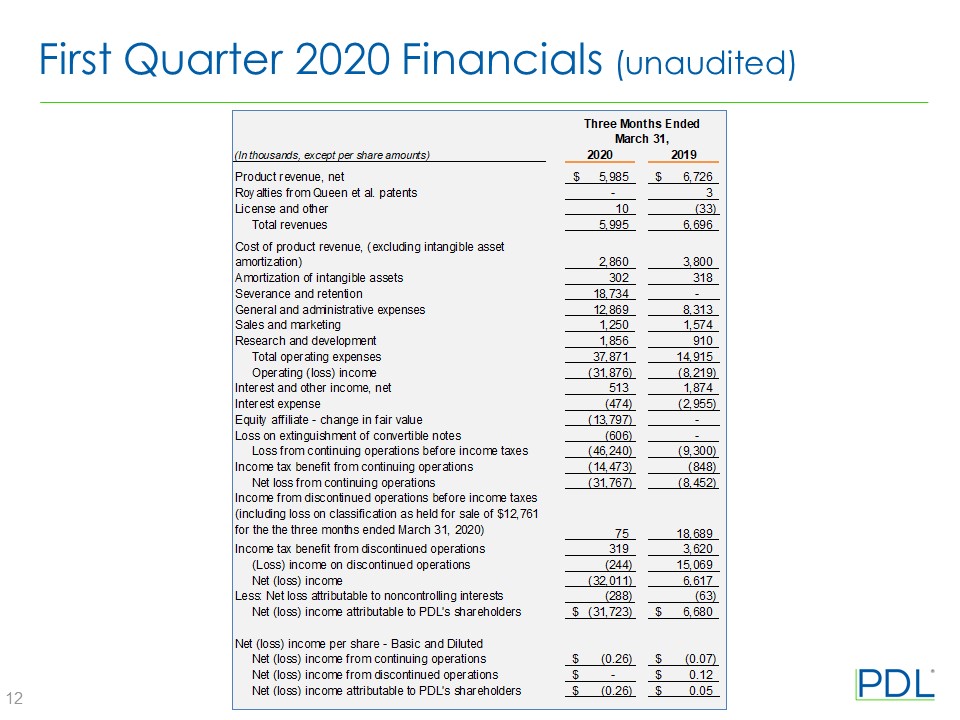
Product revenue, net | $ | 5,985 | $ | 6,726 | ||||
Royalties from Queen et al. patents | — | 3 | ||||||
License and other | 10 | (33 | ) | |||||
Total revenues | 5,995 | 6,696 | ||||||
Operating Expenses | ||||||||
Cost of product revenue (excluding intangible asset amortization) | 2,860 | 3,800 | ||||||
Amortization of intangible assets | 302 | 318 | ||||||
Severance and retention | 18,734 | — | ||||||
General and administrative | 12,869 | 8,313 | ||||||
Sales and marketing | 1,250 | 1,574 | ||||||
Research and development | 1,856 | 910 | ||||||
Total operating expenses | 37,871 | 14,915 | ||||||
Operating loss from continuing operations | (31,876 | ) | (8,219 | ) | ||||
Non-operating expense, net | ||||||||
Interest and other income, net | 513 | 1,874 | ||||||
Interest expense | (474 | ) | (2,955 | ) | ||||
Equity affiliate - change in fair value | (13,797 | ) | — | |||||
Loss on extinguishment of convertible notes | (606 | ) | — | |||||
Total non-operating expense, net | (14,364 | ) | (1,081 | ) | ||||
Loss from continuing operations before income taxes | (46,240 | ) | (9,300 | ) | ||||
Income tax benefit from continuing operations | (14,473 | ) | (848 | ) | ||||
Net loss from continuing operations | (31,767 | ) | (8,452 | ) | ||||
Income from discontinued operations before income taxes (including loss on classification as held for sale of $12,761 for the three months ended March 31, 2020) | 75 | 18,689 | ||||||
Income tax expense of discontinued operations | 319 | 3,620 | ||||||
(Loss) income on discontinued operations | (244 | ) | 15,069 | |||||
Net (loss) income | (32,011 | ) | 6,617 | |||||
Less: Net loss attributable to noncontrolling interests | (288 | ) | (63 | ) | ||||
Net (loss) income attributable to PDL's stockholders | $ | (31,723 | ) | $ | 6,680 | |||
Net (loss) income per share - basic | ||||||||
Net (loss) income from continuing operations | $ | (0.26 | ) | $ | (0.07 | ) | ||
Net (loss) income from discontinued operations | $ | 0.00 | $ | 0.12 | ||||
Net (loss) income attributable to PDL’s shareholders | $ | (0.26 | ) | $ | 0.05 | |||
Net (loss) income per share - diluted | ||||||||
Net (loss) income from continuing operations | $ | (0.26 | ) | $ | (0.07 | ) | ||
Net (loss) income from discontinued operations | $ | 0.00 | $ | 0.12 | ||||
Net (loss) income attributable to PDL’s shareholders | $ | (0.26 | ) | $ | 0.05 | |||
Weighted-average shares outstanding | ||||||||
Basic | 122,896 | 128,799 | ||||||
Diluted | 122,896 | 128,799 | ||||||
TABLE 2
PDL BIOPHARMA, INC.
CONDENSED CONSOLIDATED BALANCE SHEET DATA
(Unaudited)
(In thousands)
March 31, | December 31, | |||||||
2020 | 2019 | |||||||
Cash and cash equivalents | $ | 125,512 | $ | 168,982 | ||||
Notes receivable | $ | 53,299 | $ | 53,410 | ||||
Assets held for sale | $ | 331,661 | $ | 350,366 | ||||
Total assets | $ | 658,716 | $ | 716,119 | ||||
Liabilities held for sale | $ | 24,554 | $ | 31,215 | ||||
Total convertible notes payable | $ | 13,302 | $ | 27,250 | ||||
Total stockholders’ equity | $ | 553,115 | $ | 593,278 | ||||
TABLE 3
PDL BIOPHARMA, INC.
CONDENSED ROYALTY ASSET DATA
(Unaudited)
(In thousands)
Three Months Ended | ||||||||||||||||||||||||
March 31, 2020 | March 31, 2019 | |||||||||||||||||||||||
(in thousands) | Cash Royalties | Change In Fair Value | Total | Cash Royalties | Change In Fair Value | Total | ||||||||||||||||||
Assertio | $ | 11,177 | $ | (3,161 | ) | $ | 8,016 | $ | 10,968 | $ | (552 | ) | $ | 10,416 | ||||||||||
VB | 266 | 206 | 472 | 267 | 128 | 395 | ||||||||||||||||||
U-M | 2,005 | (1,391 | ) | 614 | 1,267 | (536 | ) | 731 | ||||||||||||||||
AcelRx | 79 | 200 | 279 | 68 | 2,088 | 2,156 | ||||||||||||||||||
KYBELLA | 42 | (29 | ) | 13 | 50 | (1,491 | ) | (1,441 | ) | |||||||||||||||
$ | 13,569 | $ | (4,175 | ) | $ | 9,394 | $ | 12,620 | $ | (363 | ) | $ | 12,257 | |||||||||||
Fair Value as of | Royalty Rights - | Fair Value as of | ||||||||||
(in thousands) | December 31, 2019 | Change in Fair Value | March 31, 2020 (1) | |||||||||
Assertio | $ | 218,672 | $ | (3,161 | ) | $ | 215,511 | |||||
VB | 13,590 | 206 | 13,796 | |||||||||
U-M | 20,398 | (1,391 | ) | 19,007 | ||||||||
AcelRx | 12,952 | 200 | 13,152 | |||||||||
KYBELLA | 584 | (29 | ) | 555 | ||||||||
$ | 266,196 | $ | (4,175 | ) | $ | 262,021 | ||||||
_____________
(1) Excludes the aggregate estimated remaining costs to sell of $5.8 million.
TABLE 4
PDL BIOPHARMA, INC.
GAAP to NON-GAAP RECONCILIATION:
NET (LOSS) INCOME
(Unaudited)
(In thousands)
A reconciliation between net (loss) income on a GAAP basis and on a non-GAAP basis is as follows: | ||||||||
Three Months Ended | ||||||||
March 31, | ||||||||
2020 | 2019 | |||||||
GAAP net (loss) income attributed to PDL’s stockholders as reported | $ | (31,723 | ) | $ | 6,680 | |||
Adjustments to Non-GAAP net income (as detailed below) | 25,012 | 5,175 | ||||||
Non-GAAP net income attributed to PDL’s stockholders | $ | (6,711 | ) | $ | 11,855 | |||
An itemized reconciliation between net (loss) income on a GAAP basis and on a non-GAAP basis is as follows: | ||||||||
Three Months Ended | ||||||||
March 31, | ||||||||
2020 | 2019 | |||||||
GAAP net (loss) income attributed to PDL’s stockholders, as reported | $ | (31,723 | ) | $ | 6,680 | |||
Adjustments: | ||||||||
Mark-to-market adjustment to fair value - royalty assets | 4,175 | 363 | ||||||
Mark-to-market adjustment to equity affiliate | 11,334 | — | ||||||
Non-cash stock-based compensation expense | 18,274 | 1,169 | ||||||
Non-cash debt offering costs | 280 | 1,923 | ||||||
Non-cash depreciation and amortization expense | 757 | 1,128 | ||||||
Mark-to-market adjustment on warrants held | 2,453 | 33 | ||||||
Non-cash amortization of intangible assets | 691 | 1,572 | ||||||
Income tax effect related to above items | (12,952 | ) | (1,013 | ) | ||||
Total adjustments | 25,012 | 5,175 | ||||||
Non-GAAP net (loss) income | $ | (6,711 | ) | $ | 11,855 | |||
Use of Non-GAAP Financial Measures
We supplement our consolidated financial statements presented on a GAAP basis by providing an additional measure which may be considered a “non-GAAP” financial measure under applicable rules of the Securities and Exchange Commission. We believe that the disclosure of this non-GAAP financial measure provides our investors with additional information that reflects the amounts and financial basis upon which our management assesses and operates our business. These non-GAAP financial measures are not in accordance with generally accepted accounting principles and should not be viewed in isolation or as a substitute for reported, or GAAP, net income, and is not a substitute for, or superior to, measures of financial performance performed in conformity with GAAP.
“Non-GAAP net income” is not based on any standardized methodology prescribed by GAAP and represents GAAP net income adjusted to exclude (1) mark-to-market adjustments related to the fair value election for our investments in royalty rights presented in our earnings, which include the fair value re-measurement of future discounted cash flows for each of the royalty rights assets we have acquired, (2) market-to-mark adjustment to our equity affiliate, (3) non-cash stock-based compensation expense, (4) non-cash interest expense related to PDL debt offering costs, (5) mark-to-market adjustments related to warrants held, (6) non-cash amortization of intangible assets, (7) non-cash depreciation and amortization expense and (8) the
related tax effect of all reconciling items within our reconciliation. Non-GAAP financial measures used by PDL may be calculated differently from, and therefore may not be comparable to, non-GAAP measures used by other companies.
# # #