UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 20-F
INFORMATION FOR SHAREHOLDERS (Mark One)
|
◻ |
REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 |
or
|
☒ |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2018
or
|
◻ |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
or
|
◻ |
SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission file number 1-14978
Smith & Nephew plc
(Exact name of Registrant as specified in its charter)
England and Wales
(Jurisdiction of incorporation or organization)
15 Adam Street, London WC2N 6LA
(Address of principal executive offices)
Securities registered or to be registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class |
|
Name on each exchange on which registered |
|
American Depositary Shares Ordinary Shares of 20¢ each |
|
New York Stock Exchange New York Stock Exchange* |
*Not for trading, but only in connection with the registration of American Depositary Shares, pursuant to the requirements of the Securities and Exchange Commission.
Securities registered or to be registered pursuant to Section 12(g) of the Act: None.
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act: None.
Indicate the number of outstanding shares of each of the issuer’s class of capital or common stock as of the close of the period covered by the annual report: 887,952,258 Ordinary Shares of 20¢ each
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act Yes ☒ No ◻
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Yes ◻ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days: Yes ☒ No ◻
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ☒ No ◻
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or an emerging growth company.
|
Large Accelerated Filer ☒ |
Accelerated Filer ◻ |
Non-accelerated filer ◻ |
|
Emerging growth company ◻ |
|
|
If an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided pursuant to Section 13(a) of the Exchange Act. Yes ◻ No ◻
† The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012.
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing.
|
◻ U.S. GAAP |
|
☒ International Financial Reporting Standards as issued by the International Accounting Standards Board |
|
|
|
◻ Other |
If “Other” has been checked in response to the previous question indicate by check mark which financial statement item the registrant has elected to follow: Item 17 ◻ Item 18 ◻
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ◻ No ☒
Indicate by check mark whether the registrant has filed all documents and reports required to be filed by Sections 12, 13 or 15(d) of the Securities Exchange Act of 1934 subsequent to the distribution of securities under a plan confirmed by a court. Yes ◻ No ◻

|
|
|
|
|
|
|
Strategic report |
Smith & Nephew Annual Report 2018 |
|
CONTENTS
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Strategic report |
|
|
|
Governance report |
|
|
|
Accounts |
|
|
|
Other information |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2 |
|
|
53 |
|
|
116 |
|
|
187 |
||||
|
4 |
|
|
54 |
|
|
117 |
|
|
OTHER FINANCIAL INFORMATION |
|
|||
|
6 |
|
|
71 |
|
|
124 |
|
|
201 |
||||
|
8 |
|
|
74 |
|
|
125 |
|
|
GO ONLINE To learn more about Smith & Nephew, please visit www.smith-nephew.com
|
|
|||
|
9 |
|
|
76 |
|
|
125 |
|
|
|
||||
|
10 |
|
|
84 |
|
|
126 |
|
|
|
||||
|
12 |
|
|
|
|
|
|
127 |
|
|
|
|||
|
14 |
|
|
|
|
|
|
128 |
|
|
|
|||
|
24 |
|
|
|
|
|
|
129 |
|
|
|
|||
|
32 |
|
|
|
|
|
|
178 |
|
|
|
|||
|
36 |
|
|
|
|
|
|
180 |
|
|
|
|||
|
38 |
|
|
|
|
|
|
|
|
|
|
|
||
|
40 |
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|

|
|
Smith & Nephew Annual Report 2018 |
|
1 |
Our purpose
Physical health is never just about our body. It’s our mind, feelings and ambitions. When something holds it back, it’s our whole life on hold.
We’re here to change that, to use technology to take the limits off living, and help other medical professionals do the same.
So that farmworkers, rugby players, grandmas and their grandkids stare down fear, see that anything’s possible, then go on stronger. Inspired by a simple promise. Two words that bring together all we do…
Life Unlimited

|
2 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
Smith & Nephew is a leading portfolio medical technology company
|
OUR PURPOSE |
|
|
HIGHLIGHTS |
|
|
|
|
Life Unlimited |
|
|
REVENUE |
|
REVENUE BY GEOGRAPHY |
|
|
Smith & Nephew exists to restore people’s bodies, and their self-belief. |
|
|
$4,904m |
|
|
|
|
|
|
|
|
|
||
|
OUR CULTURE PILLARS |
|
|
EMPLOYEES |
|
||
|
These guide our behaviours and build a winning spirit. Care A culture of empathy and understanding for each other, our customers and patients Collaboration A culture of teamwork based on mutual trust and respect Courage A culture of continuous learning, innovation and accountability
OUR STRATEGIC IMPERATIVES |
|
|
16,000+ |
|
||
|
|
|
YEARS |
|
|
|
|
|
|
|
160+ |
|
United States |
$2,354m |
|
|
|
|
|
Other Established Markets |
$1,693m |
||
|
|
|
|
Emerging Markets |
$857m |
||
|
|
|
|
|
|
|
|
|
|
|
COUNTRIES SUPPORTING |
|
|
|
|
|
|
|
HEALTHCARE PROFESSIONALS |
|
|
|
|
|
|
|
100+ |
|
|
|
|
|
|
|
MANUFACTURING & QUALITY |
|
|||
|
|
|
Smith & Nephew takes great pride in its expertise in manufacturing products to the highest quality and ensuring they reach our customers in a timely manner. |
|
|||
|
Five new strategic imperatives form our value creation plan for the medium term. 1 Achieve the full potential of our portfolio 2 Transform the business through enabling technologies 3 Expand in high-growth segments 4 Strengthen talent and capabilities 5 Become the best owner
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
Smith & Nephew Annual Report 2018 |
|
3 |
|
OUR GLOBAL FRANCHISE AREAS |
||
|
ORTHOPAEDICS |
SPORTS MEDICINE & ENT |
ADVANCED WOUND MANAGEMENT |
|
Orthopaedics includes an innovative range of Hip and Knee Implants used to replace diseased, damaged or worn joints and Trauma products used to stabilise severe fractures and correct bone deformities.
|
Our Sports Medicine and Ear, Nose and Throat (ENT) businesses offer advanced products and instruments used to repair or remove soft tissue. They operate in growing markets where unmet clinical needs provide opportunities for procedural and technological innovation.
|
Our Advanced Wound Management portfolio provides a comprehensive set of products to meet broad and complex clinical needs, to help healthcare professionals get CLOSER TO ZERO human and economic consequences of wounds.
|
|
|
|
|
|
|
|
|
OUR NEW COMMERCIAL STRUCTURE
In 2018, we initiated substantial changes to our commercial organisation to move to a franchise-led model from January 2019. Under this, a president leads each of our three specialised global marketing franchises – Orthopaedics, Sports Medicine & ENT and Advanced Wound Management. Aligned with and supporting the franchises are presidents and regional commercial organisations for Europe, Middle East, and Africa (EMEA), and Asia Pacific (APAC). The franchise presidents also have commercial responsibility for the US.
|
INNOVATION |
|
||
|
Smith & Nephew delivers innovation that aims to improve quality of life. New products and business models enable healthcare professionals to offer patients improved outcomes. We develop technology through our global R&D programme, and additionally acquire exciting products where we can add value through technical or commercial acumen. |
|
|
|
|
10% MORE INVESTED IN R&D IN 2018 |
|
|
|
|
$246m |
|
||
|
|
|
||
|
4 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
Financial highlights
|
REVENUE |
|
|
|
|
|
|
|
|
|
|
|
REVENUE BY MARKET |
|
KPI |
|
$4,904m |
|
|
|
Established Markets |
Emerging Markets |
|
|
Reported |
Underlying1 |
|
|
$4,047m |
$857m |
|
|
+3% |
+2% |
|
|
Reported |
Reported |
|
|
|
|
|
|
+2% |
+7% |
|
|
Group revenue was up 3% on a reported basis (including 1% from foreign exchange tailwind) and 2% on an underlying basis. |
|
Underlying1 |
Underlying1 |
|||
|
|
|
|
|
+1% |
+8% |
|
|
|
|
|
|
|
|
|
|
DIVIDEND PER SHARE |
|
|
|
OPERATING PROFIT |
|
KPI |
|
36.0¢ |
|
+3% |
|
$863m |
8% | |
|
|
|
|
|
|
|
|
|
The 3% year-on-year increase reflects the growth in adjusted earnings and is in-line with our progressive policy. |
|
Operating profit margin of 17.6% is down 200bps year-on-year due to impact of restructuring charges. |
||||
|
|
|
|
|
|
|
|
|
EARNINGS PER SHARE (EPS) |
|
|
TRADING PROFIT1 |
|
KPI |
|
|
76.0¢ |
|
13% |
|
$1,123m |
+7% |
|
|
|
|
|
|
|
|
|
|
The decrease reflects the impact of the restructuring |
|
Trading profit margin1 was 22.9%, up 90bps year-on-year reflecting both improved trading performance and cost control and a 50bps one-off legal settlement benefit. |
||||
|
|
|
|
|
|
|
|
|
ADJUSTED EARNINGS PER SHARE1 (EPSA) |
|
R&D EXPENDITURE |
|
KPI |
||
|
100.9¢ |
|
+7% |
|
$246m |
+10% |
|
|
|
|
|
|
|
|
|
|
The increase reflects improved trading performance and lower tax rate on trading.1 |
|
R&D expenditure was up 10% reflecting our increased investment in new products and clinical evidence. |
||||
|
|
|
|
|
|
|
|
|
RETURN ON INVESTED CAPITAL1 (ROIC) |
|
|
|
|||
|
12.5% |
|
‑180bps |
|
|
|
|
|
The decrease reflects primarily the fall in operating profit. |
|
|
|
|||
1 These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198.
|
|
A period of significant transformation; in leadership, structure, culture and strategy |
The phrase ‘step change’ is used too often, but today I believe that Smith & Nephew stands at the start of such a transformation. Roberto Quarta Chair |
DEAR SHAREHOLDER
2018 was a busy year for Smith & Nephew. Performance improved across the year, whilst the Company underwent a period of significant transformation; in leadership, structure, culture and strategy.
CHIEF EXECUTIVE OFFICER
In 2017, Olivier Bohuon told us he intended to retire after more than seven years as Chief Executive Officer. Under his leadership, Smith & Nephew experienced important and necessary change and he significantly strengthened the foundations of our Company. I would like to take this opportunity to thank him for his service and wish him a long and healthy retirement.
In May 2018, Namal Nawana joined Smith & Nephew as Chief Executive Officer and was appointed to the Board as an Executive Director.
Namal is a global industry insider, an innovator, and proven leader. Most recently, he was Chief Executive Officer, President and a member of the Board of Directors of medical diagnostics company Alere, Inc. Here he led the successful turnaround of this global business before its acquisition by Abbott Laboratories in 2017.
Before joining Alere, Namal spent more than 15 years at Johnson & Johnson, in roles of increasing responsibility in Europe, Asia and North America, culminating in Worldwide President of DePuy Synthes Spine. We were delighted when he agreed to join Smith & Nephew.
|
|
Smith & Nephew Annual Report 2018 |
|
5 |
LEADERSHIP & CULTURE
Since May, Namal has worked closely with the Board. We have reviewed and endorsed his actions to restructure the Company. He has rapidly built a highly experienced new leadership team, bringing in strong external leaders as well as promoting from within Smith & Nephew. Members of this team meet regularly with the Board, and we have seen for ourselves the clear focus and strong collaboration across this team.
The Board has long held culture as an important indicator of the underlying health of the Company. We have welcomed the importance Namal has placed on this, and his forensic approach to defining the Company’s purpose and the behaviours all employees must display to deliver the strategy. This was not an academic exercise, or conducted by just the senior management team, but rather a case study in how to engage employees, with 6,000 employees contributing to the process. The Board believes that the new purpose – Life Unlimited – and Culture Pillars of Care, Collaboration and Courage – are both authentic and inspiring.
STRATEGY
In December, the Board approved the new strategic imperatives that will drive value creation in the medium term. This was a culmination of a collaborative process between the Board and the Chief Executive Officer and senior leadership team over a number of months. During this process we tested their insight of, and vision for, the medical technology industry and found their analysis of the opportunities the Company faces was detailed and compelling.
The five strategic imperatives are similarly robust. The Board welcomes their wide-ranging scope – to accelerate growth, both organically and through acquisitions, strengthen people and capabilities, and improve the operations of our business globally.
2018 PERFORMANCE
The Board closely monitors the performance of the business through regular updates from the Chief Executive Officer and Chief Financial Officer and other members of the senior leadership team.
2018 performance was solid, with an improved dynamic in the second half. The Board noted how well the new team delivered this acceleration whilst also undertaking important work to restructure the Group. Whilst there is still much work to be done, the new Group structure is now in place. The Board endorses the guidance for further progress in 2019.
The Board is pleased that shareholders will benefit from strong growth in adjusted earnings per share, which is reflected in the 3% increase in the full year dividend to 36.0 cents per share. The performance of our shares is also noteworthy, increasing 13% from when Namal joined up to the end of 2018, strongly outperforming the FTSE 100.
BOARD CHANGES
During 2018, we welcomed Roland Diggelmann as a Non-Executive Director. Roland was, until recently, Chief Executive Officer of Roche Diagnostics and a member of the Corporate Executive Committee of F. Hoffmann-La Roche Ltd. He brings direct experience in orthopaedics from previous senior roles at Zimmer.
Ian Barlow will step down from the Board at the Annual General Meeting in April 2019, having completed a nine-year term. Ian has served Smith & Nephew with great distinction as our Senior Independent Director, and previously as Chair of the Audit Committee. I have been grateful for his counsel and thank him for his significant contribution over the years.
Michael Friedman, Chair of our Compliance & Culture Committee, will also be retiring at that time after six years’ service, and I thank him for his leadership in this crucial area. Robin Freestone will replace Ian as Senior Independent Director and Marc Owen will replace Michael as Chair of the extended Compliance & Culture Committee.
Smith & Nephew values diversity, and I am pleased that this is reflected in our Board, which, following these changes will be 30% female and include six nationalities. We continue to look for opportunities to widen our outlook and expertise with an expanded mandate.
The phrase ‘step change’ is used too often, but today I believe that Smith & Nephew stands at the start of such a transformation. Whilst there is still much work to be done, the Board is excited by the prospects and looks forward to supporting the new management team as they realise Smith & Nephew’s full potential.
Yours sincerely,

Roberto Quarta
Chair
|
6 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
|
Chief Executive Officer’s review At Smith & Nephew, we aspire to be amongst the highest-performing portfolio medical technology companies |
DEAR SHAREHOLDER
Everyone has health issues at some stage in their life. At Smith & Nephew, we have the opportunity to help patients get back to their lives as quickly as possible, and as well as possible. Whether it be in Orthopaedics, Sports Medicine or Wound Management we recognise this opportunity and it inspires and motivates our work each day around the world.
CREATING A PURPOSE-DRIVEN CULTURE
I believe that a successful and sustainable business has a foundation that is built on a purpose-driven culture. When I joined, we asked our employees which elements of our culture they liked and that we should retain, as well as what we needed to improve at Smith & Nephew. 6,000 employees responded.
It was clear that our colleagues cared deeply about the work that we do. It was also clear that they recognised that we could do better. The opportunity was to find an authentic and inspiring purpose that combines this caring spirit with a greater focus on working more effectively and instilling a strong accountability to deliver consistently on our commitments.
Life Unlimited captures the essence of Smith & Nephew and our purpose to address meaningfully the health issues that hinder people from living their lives to their fullest.
To support this brand purpose we have developed three culture pillars: Care, Collaboration and Courage – which we launched with employees at the end of 2018. Grounded in the service of patients and practitioners, these simple tenets guide us in our work together and couple the idea of continuous learning and improvement with the aspiration to lead in all our endeavours.
OUR BUSINESS AND STRATEGIC IMPERATIVES
Smith & Nephew is a portfolio medical technology company with a broad and deep range of high quality products. We have examples of market-leading technology in almost every area of our business. We also operate in large and attractive global markets, with solid long-term growth prospects supported by favourable demographics and lifestyle trends.
At the end of 2018, we launched five new strategic imperatives that recognise the specific business and markets we operate in, and form the basis of our value creation plan for the medium-term.
1 Achieve the full potential of our portfolio
2 Transform the business through enabling technologies
3 Expand in high-growth segments
4 Strengthen talent and capabilities
5 Become the best owner
These highlight the key multi-year initiatives in which the Company is now engaged. They also detail the specific plans and metrics for the upcoming calendar year from which all employees build their own individual annual objectives.
INCREASING CUSTOMER CENTRICITY
One of the most significant changes we are making is implementing a new commercial model. In line with industry best practice for global medical technology businesses, we are moving from a regional selling model to a global franchise structure. We have put dedicated presidents of Orthopaedics, Sports Medicine & ENT, and Advanced Wound Management in place.
Each president has global upstream marketing responsibility, as well as full commercial responsibility for the franchise in the US. Outside the US, we will have two regions, Europe, Middle East and Africa, and Asia Pacific. Both regions are now represented on the Smith & Nephew Executive Committee ensuring continued focus on commercial execution. As specialists, the presidents bring great insight into our customers’ current and future needs, wherever they are in the world and will be able to direct the full resources of their franchises to meet these.
I am delighted with the quality of leaders we have attracted. The focus is now on unlocking the potential of Smith & Nephew, with five members of my executive team directly responsible for driving growth in their franchises and regions.
|
|
Smith & Nephew Annual Report 2018 |
|
7 |
|
|
I am delighted with the quality of talent we have attracted. The focus is now on unlocking the potential of Smith & Nephew, with five members of my executive team directly responsible for driving organic growth in their franchises and regions. Namal Nawana Chief Executive Officer |
ADDING VALUE THROUGH ACQUISITIONS
Smith & Nephew completed 2018 with a net debt1 to adjusted EBITDA ratio2 of 0.8x and, with strong cash flows and cash conversion, we will look to appropriately deploy capital to M&A initiatives more significantly than in recent years as part of our business model for success.
Technology acquisitions such as Rotation Medical have proven to be a great success. From its REGENETEN™ Bioinductive Implant for rotator cuff repair, we have driven performance well-ahead of our deal model, with more than 130% growth in 2018. We believe there is still much more to come from this product as we add manufacturing capacity and launch in new international markets in 2019.
In December, we announced the acquisition of Ceterix Orthopaedics, the developer of the NovoStitch™ Pro Meniscal Repair System. This product is highly complementary to our portfolio and will significantly expand our opportunity in the underserved meniscal repair segment.
I expect us to continue to enhance our position in high-growth, high-innovation markets over time and capitalise on our platform as a global medical device portfolio company.
2018 RESULTS
In 2018, revenue growth was 3% on a reported basis and 2% underlying, and we delivered a meaningful improvement in trading profit margin.
Geographically, we continued to build upon our strong position in the Emerging Markets, which now account for 17% of Group revenue. A solid performance in the US, our largest market, was somewhat offset by continued weakness in some European markets.
At a franchise level, highlights included the market beating growth from Knee Implants, the strong return to growth delivered in Hip Implants, and the increased adoption of our NAVIO robotics platform. Growth from our Advanced Wound Devices franchise also stood out, driven by our PICO single-use Negative Pressure Wound Therapy system. Actions are underway to improve weaker performances from Arthroscopic Enabling Technology and Advanced Wound Bioactives.
After a slow start to 2018, it’s pleasing that our team accelerated performance as the year progressed, whilst at the same time making the important changes to how we run the Company.
FOCUSED ON DELIVERY
At Smith & Nephew, we aspire to be amongst the highest-performing portfolio medical technology companies.
We start 2019 with a new executive leadership team and operating structure in place. We have clarified our brand purpose with Life Unlimited and have introduced new culture pillars and strategic imperatives to support it.
Together, we are confident that we are building the right foundation for sustainable success and an ability to grow consistently with our markets in the future. This confidence is reflected in our financial guidance for further improvements in both revenue and margin performance in 2019, explained in detail by our Chief Financial Officer, Graham Baker, on page 37 of this Annual Report.
There is much to do to achieve our goals and aspirations but we are grateful for the opportunity to positively affect the patients, practitioners and health systems that we serve globally. I look forward to updating you on our progress.
Yours sincerely,

Namal Nawana
Chief Executive Officer
1 Net debt is reconciled in Note 15 to the Group accounts.
2 These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198.
|
8 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
|
A successful business needs to have a purpose-driven culture |
Life Unlimited captures the essence of Smith & Nephew and our purpose to address meaningfully the health issues that hinder people from living their lives to their fullest. Namal Nawana Chief Executive Officer |
|
|
Life Unlimited |
|
|
|
||
|
|
||
|
CARE |
COLLABORATION |
COURAGE |
|
A culture of empathy and understanding for each other, our customers and patients |
A culture of teamwork, |
A culture of continuous |
|
– We step into our customers’ shoes, anticipate their needs and deliver the highest levels of innovation and service – We strive to have the best understanding of the patients whom we ultimately serve, and we develop our products with them in mind – Our passion for what we do drives us to continuously improve and expand the positive impact that we have on the world |
– We are stronger, and achieve more, as a team. By joining forces we are both unstoppable and efficient – Through transparent and respectful communication, we are motivated by a shared purpose and understand the impact of our individual contributions on our collective goals – By encouraging different perspectives and leveraging our global experiences, we achieve the best outcomes |
– By staying curious, thinking big and having the humility to challenge our conventional ways of thinking, we push the boundaries of our industry – Fostering an entrepreneurial, can-do attitude we look for solutions and achieve them through talent and force of will – With a growth mindset, we have the capability and confidence to win, and we do so with integrity and the highest ethical standards |
|
|
Smith & Nephew Annual Report 2018 |
|
9 |
|
Five new strategic imperatives form our value creation plan for the medium term |
|
GROW |
|
MEASURING PERFORMANCE Behind our strategic imperatives sits a detailed dashboard of key performance indicators that we use to track and evaluate our performance. These cover Commercial, Operations, R&D, People, our SG&A and cost base, as well as return on investment and cash. Whilst many of these are commercially sensitive, and hence will not be published, they all support our objective to deliver on our financial guidance for 2019. This is detailed in the CFO review on page 37. Other published metrics include our work to simplify the organisation and processes through our restructuring programme APEX, and our focus on turning profit into cash. These are also described in more detail in the CFO review. 2018 KPIs were set against previous strategic priorities. These measured revenue growth in our Established and Emerging Markets, operating and trading profit margin and R&D investment. These measures are reported on page 4.
|
||
|
1 |
|
Achieve the full potential of our portfolio Improving execution to accelerate organic performance with a focus on (i) platform-specific plans, (ii) Ambulatory Surgery Centres and (iii) Emerging Markets, especially China and Latin America. |
|
|
|
|
|
|
|
|
|
2 |
|
Transform the business through enabling technologies By acquiring and developing leading enabling technologies to transform procedures, including robotics, imaging and augmented reality. |
|
|
|
|
|
|
|
|
|
3 |
|
Expand in high-growth segments By accelerating portfolio growth, strengthening or establishing leadership positions, and driving meaningful synergies. |
|
|
|
|
|
|
|
|
|
TOGETHER |
|
|||
|
4 |
|
Strengthen talent and capabilities By developing a winning culture to improve retention and attract talent. |
|
|
|
|
|
|
|
|
|
EFFECTIVELY |
|
|||
|
5 |
Become the best owner To drive meaningful margin expansion through operational transformation and organisation simplification. |
|||
|
10 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
Competing in large, attractive markets
The healthcare sector is a growing market driven by long‑term trends. Global healthcare spend amounted to $7.7 trillion, or 10.4% of global GDP in 2017, and is projected to increase at an annual rate of 5.4% over the five year period to 2022.*
The medical devices and supplies segment of healthcare is today worth more than $400 billion per annum. Within that, Smith & Nephew’s product segments are worth approximately $38 billion, growing at around 4% annually.
The main drivers for healthcare demand include demographic shift towards ageing populations and an increase in lifestyle-related ailments such as diabetes and obesity. The World Health Organisation (WHO), for example, states that obesity has nearly tripled since 1975 worldwide – a major risk factor for diseases such as diabetes and musculoskeletal disorders.
Faster growing emerging markets with an emerging middle class also drive demand. The Brookings Institute estimates that 65% of the global population will be middle class by 2030. Access to middle class comforts encourages sedentary lifestyles that may lead to greater incidence of diabetes, obesity and other health conditions. Wealthier patients also try to exert more choice over healthcare and have greater expectation of quality of life.
The number of people aged 60 years and older will outnumber children younger than five years by 2020, according to WHO. This change in dynamic puts healthcare providers and governments increasingly under economic pressure. Politicians seek ways to reduce overall healthcare expenditure whilst maintaining the quality of care and treatment provided.
|
COMPLIANCE Interactions between medical device companies and healthcare professionals or government officials are subject to strict control. These include laws and industry codes, including the AdvaMed Code of Ethics and the Med Tech Europe Code of Business Practice. Legislation covering corruption and bribery, such as the UK Bribery Act and the US Foreign Corrupt Practices Act, also applies to Smith & Nephew world-wide. There is also a strong focus on compliance and cost control in emerging markets such as China. We are committed to ensuring regulatory compliance globally, at all times, and to execute business with integrity. |
GEO-POLITICAL FACTORS
Some uncertainty continues around the UK’s exit from the European Union and its regulatory impact. The European Union is the UK’s biggest export market for medical devices. Around $2 billion worth of products are sent to European countries each year.
Smith & Nephew has taken steps to prepare for the various Brexit scenarios, including moving certain of its product certifications from BSI UK to BSI Netherlands, ensuring these remain with a Notified Body domiciled in the European Union. There is also uncertainty around US-China trade relations, which has resulted in tariffs on some medical devices being exported between the two countries.
A HIGHLY REGULATED INDUSTRY
The medical device sector is highly regulated. This is vital in determining whether products are both safe and effective.
National regulatory authorities administer and enforce a complex series of laws and regulations that govern the design, development, approval, manufacture, labelling, marketing and sale of healthcare products. They also review data supporting the safety and efficacy of such products. In many countries, there is a requirement for products to be authorised or registered prior to entering the market, and such authorisation or registration needs to be subsequently maintained. For example, the US Food and Drug Administration (FDA) continues to enforce an increase in the amounts of testing and documentation required for FDA approval of new drugs and medical devices.
In Europe, the European Union Medical Device Regulations came into force in 2017 and will apply from May 2020. This will also impose tougher requirements of market entry and post-market surveillance of medical devices. Although healthcare systems are less costly in Europe than in the US, strained government budgets and demographic challenges are driving an increased focus on value-based healthcare to demonstrate the value of innovation through evidence.
The major regulatory agencies for Smith & Nephew’s products include the US FDA, the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK, the Ministry of Health, Labour and Welfare in Japan, the National Medical Products Administration (NMPA) in China, formerly the China Food and Drug Administration, and the Australian Therapeutic Goods Administration.
We are subject to regular inspections and audits by regulatory agencies and notified bodies, and in some cases remediation activities have been required and will continue to require significant financial and resource investment.
* Source: Deloitte 2019 Global Health Care Outlook.
|
|
Smith & Nephew Annual Report 2018 |
|
11 |
EVOLVING MODELS OF HEALTHCARE
The traditional approach to healthcare provision has been symptom and volume (fee‑for-service) oriented which – in combination with current demographic trends – has put upward pressure on healthcare costs. In response, stakeholders are increasingly seeking to shift the focus from ‘break-fix’ to a more holistic and value-based approach focused on disease prevention and treatment results (fee-for-outcome).
Healthcare practitioners are no longer the only decision-makers, but are part of larger multi-stakeholder purchasing processes. Economic stakeholders have increasing influence on the purchase process for medical devices. New payment models, such as bundled procedure payments, risk sharing, or quality incentives/penalties, are shifting the focus from clinical utility and safety alone to clinical outcomes and health economic performance.
There are a number of emerging trends which will shape our marketplace in the medium term.
There is an emerging trend for greater use of outpatient surgery. This is leading to a shift in where total joint procedures take place. Historically these have been inpatient procedures requiring an overnight stay in a hospital. With improvements in technology, more minimally invasive techniques and better pain management, total joint procedures can take place in the outpatient setting for the right patients. For example in the US there are ambulatory surgery centers (ASCs), smaller clinical units with no overnight beds. Costs are lower when no overnight stay is required, important in the context of pressure on health budgets around the world.
Other emerging trends include digital health, with connected devices monitoring patients to prevent conditions, support rehabilitation and measure outcomes. Robotics is also becoming increasingly present in the operating room, offering surgeons greater precision and consistency.
|
SEASONALITY Some seasonality is evident in medical devices. Orthopaedic reconstruction and sports medicine procedures tend to be higher in the winter months when accidents and sports-related injuries are highest. Elective procedures tend to slow down in the summer months due to holidays. Due to the nature of our product range, there is little seasonal impact on our Advanced Wound Management franchises. The majority of our business is in the Northern Hemisphere, including approximately 50% in the US and 25% in Europe. In the US, out-of-pocket costs for health insurance plans are tied to medical expenses in a calendar year. As a result, households who have reached their deductible (or out-of‑pocket) cap may find that accessing care later in the year comes at a lower cost, which may encourage some to schedule any required treatments or procedures in the final months of any given year. COMPETITION Smith & Nephew’s franchises have several competitors which differ with respect to product focus, geographic reach and overall scale. For example, our main surgical competitors are larger in scale and tend to be more exposed to the US, whereas our key wound competitors are generally not US centric. In Orthopaedics, as one of four leading players, we compete against US-based companies Stryker, Zimmer Biomet and Depuy Synthes (a Johnson & Johnson company). In Sports Medicine, we hold a leading position behind Arthrex (US), and also compete against Stryker and Depuy Synthes. We are the second largest global Advanced Wound Management business. We lead the somewhat fragmented Advanced Wound Care sub-segment alongside Mölnlycke (Sweden) and ConvaTec (UK). In Advanced Wound Devices, we are the primary challenger to Negative Pressure Wound Therapy incumbent Acelity (US). In our Advanced Wound Bioactives franchise, our key products lead their respective categories. |
||
|
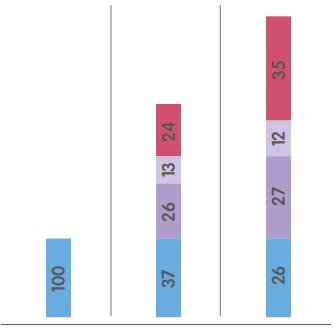
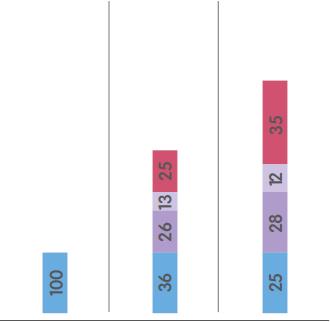
MARKET SIZE1 |
|
|
|
|
|
|
|
|
|
||||||||||||
|
ORTHOPAEDICS
|
|
|
|
|
|
SPORTS MEDICINE2
|
|
|
ADVANCED WOUND MANAGEMENT
|
||||||||||||
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
Hip & Knee Implants |
|
|
Trauma & Extremities |
|
|
|
|
|
|
||||||||||||
|
$14.5bn |
+2% |
|
|
$6.0bn |
+4% |
|
|
$5.0bn |
+5% |
|
|
$9.0bn |
+5% |
||||||||
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
A |
SMITH & NEPHEW |
12% |
|
|
A |
SMITH & NEPHEW |
8% |
|
|
A |
SMITH & NEPHEW |
26% |
|
|
A |
SMITH & NEPHEW |
14% |
||||
|
B |
ZIMMER BIOMET |
33% |
|
|
B |
DEPUY SYNTHES3 |
43% |
|
|
B |
ARTHREX |
31% |
|
|
B |
ACELITY |
16% |
||||
|
C |
STRYKER |
21% |
|
|
C |
STRYKER |
26% |
|
|
C |
STRYKER |
15% |
|
|
C |
MOLNLYCKE |
10% |
||||
|
D |
DEPUY SYNTHES3 |
20% |
|
|
D |
ZIMMER BIOMET |
11% |
|
|
D |
DEPUY SYNTHES3 |
12% |
|
|
D |
CONVATEC |
7% |
||||
|
E |
OTHERS |
14% |
|
|
E |
OTHERS |
12% |
|
|
E |
OTHERS |
16% |
|
|
E |
OTHERS |
53% |
||||
1Data used in 2018 estimates generated by Smith & Nephew is based on publicly available sources and internal analysis and represents an indication of market shares and sizes.
2Representing repair products and arthroscopic enabling technologies, and excluding ENT.
3A division of Johnson & Johnson.
|
12 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
Value creation is driven by our new brand purpose, culture pillars and strategic imperatives
|
OUR RESOURCES |
CREATING VALUE THROUGH |
||||||
|
|
|
||||||
|
Our people & culture Attracting, developing and retaining the best employees is important. We strive to build a purpose-driven culture based on strong and authentic values. Ethics & compliance Committed to doing business the right way, applying strict principles to the way we work. Sales & marketing Supporting customers through highly specialised sales teams with in-depth technical knowledge that surgeons and nurses greatly value. Manufacturing & quality Operating global manufacturing efficiently, to the highest standards, to ensure quality and competitiveness. Medical education Supporting the safe and effective use of our products through medical education. Research & development Innovation is part of our culture and we are increasing the amount we invest in new products. Sustainability We focus on three aspects of sustainability; economic prosperity, social responsibility and environmental stewardship. |
Purpose-driven culture We believe in Life Unlimited, and have three culture pillars that guide our behaviours and build a winning team spirit: Care, Collaboration and Courage. |
Strategic imperatives Our five new strategic imperatives reflect our ambition to maximise commercial advantage from our marketplace. They will form our value creation plan for the medium term. |
|||||
|
Life Unlimited |
|||||||
|
Strong product portfolio
We have market-leading technology across our broad range of products. We deploy our capital to drive continued |
Customer centricity Serving our customers is at the heart of our model. We have a global franchise model led by management who are specialists in their markets. This keeps us close to our customers, ensuring we can anticipate and meet their needs. |
||||||
|
|
|
|
|
|
|
||
|
|
OUR RESOURCES PAGES 23‑34 |
OUR CULTURE PILLARS PAGE 8 |
|
|
OUR STRATEGIC IMPERATIVES PAGE 9 |
||
|
|
Smith & Nephew Annual Report 2018 |
|
13 |
|
VALUE DELIVERED IN 2018 |
VALUE SHARED |
|||
|
REVENUE |
Shareholders We have a progressive dividend policy and in respect of 2018 our shareholders benefited from a 3% increase in dividend. In addition, our shares rose 14% over the course of 2018. Patients Patients in more than 100 countries were treated with our products in 2018. We continued to widen access to our products, with 17% of revenue now coming from sales to the emerging markets. Customers We continued to expand treatment options available through R&D and acquisitions, published more than 200 pieces of clinical or economic evidence, and provided extensive professional development training. Employees 6,000 employees engaged in the development of our new purpose and culture pillars which are guiding revised evaluation, diversity and development programmes. Communities We work in a sustainable, ethical and responsible manner, making $8m in cash and product donations in 2018. |
|||
|
$4,904m |
||||
|
OPERATING PROFIT |
TRADING PROFIT1 |
|||
|
$863m |
$1,123m |
|||
|
DIVIDEND |
JOBS |
|||
|
$321m |
16,000+ |
|||
|
EFFICIENCY SAVINGS |
PUBLISHED CLINICAL EVIDENCE |
|||
|
$60m |
200+ |
|||
|
PRACTITIONER TRAINING INSTANCES |
PHILANTHROPIC DONATIONS |
|||
|
50,000+ |
$8m |
|||
|
1 These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198. |
||||
|
OUR FRANCHISES |
OUR RESULTS |
OUR RESOURCES |
||
|
14 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
From 1 January 2019, we will serve our customers through three franchises

|
|
Orthopaedics |
|
Sports Medicine & ENT |
|
Advanced Wound Management |
|
|
|
Orthopaedics includes an innovative range of Hip and Knee Implants used to replace diseased, damaged or worn joints and Trauma products used to stabilise severe fractures and correct bone deformities. |
|
Our Sports Medicine and Ear, Nose and Throat (ENT) businesses offer advanced products and instruments used to repair or remove soft tissue. They operate in growing markets where unmet clinical needs provide opportunities for procedural and technological innovation. |
|
Our Advanced Wound Management portfolio provides a comprehensive set of products to meet broad and complex clinical needs, to help healthcare professionals get CLOSER TO ZERO human and economic consequences of wounds. |
|
|
|
Smith & Nephew Annual Report 2018 |
|
15 |
|
Orthopaedics Proven products to enhance quality of life Smith & Nephew’s Orthopaedics franchise includes an innovative range of Hip and Knee Implants used to replace diseased, damaged or worn joints, and Trauma products used to stabilise severe fractures, correct bone deformities, treat arthritis and heal soft tissue complications. |
With best-in-class products utilising unique materials and backed by strong data, our Orthopaedics franchise is well-positioned for further growth. Skip Kiil President of Orthopaedics |
|||||
KNEE IMPLANTS
Every year more than two million patients receive total, partial or revision knee replacements worldwide.1 Smith & Nephew’s range of products for specialised knee replacement procedures include leading products for total, partial and patellofemoral joint resurfacing procedures. Customers and patients benefit from our unique technologies including our proprietary advanced bearing surface, VERILAST™, our robotics-assisted platform, NAVIO™ Surgical System, and our customised VISIONAIRE™ Patient-Matched Instrumentation.
Smith & Nephew’s JOURNEY™ II Total Knee Arthroplasty system is designed and demonstrated to replicate normal knee positions, shapes, and motions.2‑8 The range includes bi-cruciate stabilised and cruciate retaining options, and the JOURNEY II XR, an innovative bi-cruciate retaining knee implant launched in 2018, which is designed to retain the anterior and posterior cruciate ligaments (ACL/PCL) and deliver normal perception of movement and muscle control.9
The LEGION™/GENESIS™ II Total Knee System is a comprehensive system designed to allow surgeons to address a wide range of knee procedures. It includes the LEGION Revision Knee System, designed to offer surgeons improved options to deal with the complexities associated with revision knee arthroplasty.
These systems feature VERILAST Technology, our advanced bearing surface of OXINIUM™ Oxidized Zirconium with highly cross-linked polyethylene. The LEGION Primary Knee with VERILAST Technology has been laboratory-tested for 45 million cycles of wear simulation, approximating 30 years of activity. While lab testing is not the same as clinical performance, the tests showed significant reduction in wear compared to conventional technologies.10*
Our ANTHEM™ Total Knee System and ORTHOMATCH™ Universal Instrumentation Platform, launched in 2017, are designed to provide wider market access to affordable knee treatment. ANTHEM is tailored to meet the anatomical needs of patients from Asia, the Middle East, Africa and Latin America and the ORTHOMATCH instrumentation platform reduces weight, footprint and unnecessary cost without compromising on quality.11
The NAVIO Surgical System provides accuracy,12‑20 flexibility and confidence utilising real-time imaging (without the need for a preoperative CT scan), hand-held robotics, a portable cart, and multiple partial and total knee implant options in an economically sound platform.21 NAVIO offers both partial and total knee options that include the first and only robotics-assisted bi-cruciate retaining knee procedure commercially available today.
Additionally, our knee systems can utilise our VISIONAIRE Patient-Matched Instrumentation, whereby an MRI and X-Rays are used to create customised cutting guides designed to allow the surgeon to achieve optimal alignment of the new implant.22
HIP IMPLANTS
Smith & Nephew’s Hip Implants franchise offers a range of specialist products for reconstruction of the hip joint. This may be necessary due to conditions such as arthritis causing persistent pain and/or as a result of hip fracture. Every year more than two million patients worldwide undergo total, resurfacing and revision hip replacement procedures.1
Smith & Nephew has developed a range of primary hip systems. Core systems include the ANTHOLOGY™ Hip System, SYNERGY™ Hip System and the POLAR3™ Total Hip Solution. This diversity exemplifies our commitment to providing surgeons with implant and instrumentation options that meet the specific demands of their patients and preferred surgical approach, most notably the direct anterior or posterolateral approach. We also market the BIRMINGHAM HIP™ Resurfacing (BHR) System, an important option for surgeons treating suitable patients.
|
16 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
OUR FRANCHISES continued
Smith & Nephew’s portfolio also includes the REDAPT™ Revision Hip System. The need to perform a revision can occur for a variety of reasons including infection, dislocation, or failure of the implants to achieve biologic fixation. REDAPT is designed to turn such complex hip revisions into efficient, reproducible surgeries, allowing surgeons to effectively recreate a patient’s unique functionality, while quickly and easily addressing issues such as poor bone quality.23
The REDAPT Fully Porous Acetabular Cup with CONCELOC™ Technology is designed to allow ingrowth through an additive, or 3D printing, manufacturing process which produces a porous implant that mimics the structure of cancellous bone. The 3D printing method allows for complex design geometries that would be difficult, expensive or impossible to achieve with traditional manufacturing methods.24
TRAUMA
In Trauma, the TRIGEN™ INTERTAN™ hip fracture system allows patients to experience lower risk of implant failure and re-operation, faster time to fracture union, and a high return to pre-fracture status.25
The EVOS™ Plate and Screw System is a stainless steel, highly versatile system with a multitude of plate geometries and longer screw lengths than standard mini fragment systems. The EVOS Small Fragment system for lower extremity fractures and general trauma utilisation features more points of fixation and greater breadth of plate options.
For extremities and limb restoration, our range includes the TAYLOR SPATIAL FRAME™ External Fixator as well as plates, screws, arthroscopes, instrumentation, resection and suture anchor products for foot and ankle and hand and wrist repair as well as INVISIKNOT™, a unique syndesmotic fixation device for the ankle.
OUR PERFORMANCE IN 2018
|
|
Revenue |
Reported |
Underlying |
|
Knee Implants |
$1,017m |
3% | 3% |
|
Hip Implants |
$613m |
2% | 2% |
|
Trauma |
$493m |
0% | 1% |
In Knee Implants we delivered a market beating growth in 2018. This was driven by double-digit underlying growth across our JOURNEY II Total Knee System, LEGION Revision Knee System and ANTHEM Knee System for the Emerging Markets.
In Hip Implants, performance improved markedly in the second half of the year, with improved execution driving demand for the POLAR3 total hip solution, with its class-leading survivorship data, and the continued roll-out of the REDAPT Revision System.
In Trauma we delivered good growth from INTERTAN Nails and drove an increasing contribution from the new EVOS SMALL plating system, offset by reduced tender activity in the Middle East.
References
1 2018 Smith & Nephew Market Model.
2 Noble PC, et al. Clin Orthop Relat Res. 2005;431:157‑165.
3 Mayman DJ, et al. Poster presented at: ISPOR Symposium; May 2018; USA.
4 Nodzo SR, et al. Tech Orthop. 2018;33:37‑41.
5 Takubo A, et al. J Knee Surg. 2017;30:725–729.
6 Kosse NM, et al. Poster 99 presented at: 2nd World Arthroplasty Congress; April 2018; Italy.
7 Kaneko T, et al. J Orthop. 2017;14(1):201‑206.
8 Grieco TF, et al. J Arthroplasty. 2017;33(2):565‑571.
9 Smith & Nephew 00225 V3 JOURNEY II BCS and CR Design Rationale 0118.
10 Papannagari R, et al. Poster 1141, ORS Annual Meeting, 2011.
11 Smith & Nephew 07147 V2 ANTHEM Total Knee System Design Rationale 10.18.
12 Herry Y, et al. Int Orthop. 2017;41:2265‑2271.
13 Batailler C, et al. Poaster presented at: ESSKA; May 2018; UK.
14 Gregori A, et al. Paper presented at: International Society for Computer Assisted Orthopaedic Surgery; June 2015; Canada.
15 Gregori A, et al. Abstract presented at: 15th EFORT Congress; June 2014; UK.
16 Smith JR, et al. Poster presented at: Congress of the International Society of Biomechanics; August 2013; Brazil.
17 Jaramaz B, et al. Paper presented at: International Society for Computer Assisted Orthopaedic Surgery; June 2015; Canada.
18 Mitra R. Poster presented at: World Arthroplasty Congress; April 2018; Italy.
19 Jaramaz B, et al. EPiC Series in Health Sciences. 2018;2:98‑101.
20 Jaramaz B, et al. Poster presented at: 19th EFORT Annual Congress; May/June 2018; Spain.
21 Sg2 Healthcare Intelligence. Technology Guide: Orthopedic Surgical Robotics. 2014.
22 Smith & Nephew 2012 VISIONAIRE Design Rationale 7128‑1567 Rev. A10/12.
23 Smith & Nephew 10864 V1 REDAPT Revision Acetabular Augment Design Rationale 0718.
24 Smith & Nephew 03955 V2 CONCELOC Material Specifications 0317.
25 Smith & Nephew 05036 V2 TRIGEN INTERTAN Claims Brochure 0817.
26 National Joint Registry for England, Wales and Northern Ireland: 15th Annual Report. 2018.
* The LEGION Primary CR Knee System completed 45 million cycles of in vitro simulated wear testing, which is an estimate of 30 years of activity. Other LEGION VERILAST Primary Knee Systems underwent similar lab testing comparable to industry standards. The results of in vitro wear simulation testing have not been proven to quantitatively predict clinical wear performance. Also, a reduction in total polyethylene wear volume or wear rate alone may not result in improved clinical outcomes as wear particle size and morphology are also critical factors in the evaluation of the potential for wear mediated osteolysis and associated aseptic implant loosening. Particle size and morphology were not evaluated as part of the testing.
** These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194‑198.
|
|
Smith & Nephew Annual Report 2018 |
|
17 |
|
|
|||
|
POLAR3 TOTAL HIP SOLUTION |
|||
|
The POLARSTEM and R3 Total Hip Solution has the best survivorship figures of any total hip construct at seven years according to the world’s largest national joint registry.26 |
|
|
|
|
18 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
|
OUR FRANCHISES continued Sports Medicine & ENT Technology to improve healthcare |
|
We have a leading portfolio of instruments and implants for soft tissue repair, and a proud history of successfully addressing unmet clinical needs. Brad Cannon President of Sports Medicine & ENT
|
||
|
|
|
|
||
|
Smith & Nephew’s Sports Medicine and Ear, Nose and Throat (‘ENT’) franchise operates in growing markets where unmet clinical needs provide opportunities for procedural and technological innovation. SPORTS MEDICINE JOINT REPAIR In Sports Medicine Joint Repair, our technologies, instruments and implants enable surgeons to perform minimally invasive surgery of the joints, including the repair of soft tissue injuries and degenerative conditions of the shoulder, knee, hip and small joints.
For shoulder repair, we market products primarily for Rotator Cuff Repair (RCR) and instability repair, two of the most commonly performed sports medicine procedures. Our key shoulder repair products include a variety of suture anchors, such as HEALICOIL™ Suture Anchors featuring open-architecture design and SUTUREFIX™ and Q-FIX™ All-Suture Anchors, suture passers such as FIRSTPASS™ ST, and ULTRABRAID™ and ULTRATAPE Sutures. All these products can be used together or in conjunction with other existing products from the Smith & Nephew portfolio in a single procedure, significantly expanding the breadth of our comprehensive solutions for shoulder repair.
Enhancing our RCR portfolio, the REGENETEN™ Bioinductive Implant, acquired in 2017, is a breakthrough technology and technique that balances biomechanics and biology to enhance the body’s natural healing response, helping tendons heal by
|
|
|||
|
REGENETEN BIOINDUCTIVE IMPLANT |
||||
|
Rotator cuff disease is a significant and costly problem3,16,17 that causes ongoing pain and limits patients’ mobility.18 The REGENETEN Bioinductive Implant stimulates the body’s natural healing response to support new tendon growth and disrupt disease progression.3,19 |
|
|||
|
|
Smith & Nephew Annual Report 2018 |
|
19 |
The REGENETEN implant is highly complementary to our Sports Medicine portfolio, especially for enhancing a broad spectrum of rotator cuff repairs, serving an unmet clinical need and providing a compelling new treatment option for our customers.
In knee repair, the FAST-FIX™ family of Meniscal Repair Systems, FIRSTPASS MINI Suture Passer, and the ACUFEX™ Meniscal Root Repair System increase the number of meniscal injuries we can help surgeons address. For ligaments, the ENDOBUTTON™ and ULTRABUTTON™ fixed and adjustable loop devices, BIOSURE™ interference screws, and the new ACUFEX™ EXTRA-ARTICULAR Reconstruction Guide System give surgeons multiple tools for performing single and complex ligament repairs. Outside the United States, the CARGEL™ Bioscaffold can be used in conjunction with microfracture to repair articular cartilage. With these products, we provide a unique package of solutions used by surgeons to help them restore knee function for their patients.
In December 2018, we announced the acquisition of Ceterix Orthopaedics, Inc., the developer of the NovoStitch Pro Meniscal Repair System. This unique device addresses complex meniscal tear patterns not adequately served by other repair systems and is highly complementary to Smith & Nephew’s leading FAST-FIX 360 Meniscal Repair System. The acquisition completed on 22 January 2019.
The Smith & Nephew joint repair portfolio includes implants made from a variety of biocompatible materials, including next-generation anchors made of soft, all-suture material and REGENESORB™, an advanced biocomposite. For example, the Q-FIX All-Suture Anchor is ideal for a variety of arthroscopic shoulder and hip repairs, offering fixation performance superior to commonly used all-suture anchors and traditional anchors.4 The SUTUREFIX ULTRA All-Suture Anchor is an attractive option for procedures in which anatomic space is very limited5, while still delivering high fixation strength.6 Implants made from REGENESORB, including versions of the HEALICOIL™ Suture Anchor for shoulder repair and BIOSURE™ Interference Screw for knee repair, have been shown to be absorbed and replaced by bone within 24 months in pre-clinical studies.7 *
ARTHROSCOPIC ENABLING TECHNOLOGIES (AET)
AET products are often used in conjunction with products from Sports Medicine Joint Repair. AET includes high definition imaging solutions, industry leading energy-based and mechanical resection platforms, and fluid management and access technologies. Our platforms work in concert to facilitate access to various joint spaces, visualise the patient’s anatomy, resect degenerated or damaged tissue and prepare the joint for a soft tissue repair.
The WEREWOLF™ and QUANTUM 2™ COBLATION™ Controllers, which are used with a wide range of high performance COBLATION radio frequency (RF) wands, enable surgeons to remove soft tissue precisely8 and control bleeding in a variety of arthroscopic procedures. WEREWOLF, our latest advance in COBLATION™ Technology, and the FLOW 50™ Wand have demonstrated faster patient recovery9 and better long-term patient outcomes10,11 and safety12,13 in knee procedures.* The WEREWOLF and QUANTUM 2 Controllers and their associated wands carry broad indications across Sports Medicine.
The LENS™ Integrated Visualisation System provides outstanding image quality and functionality in a simple three-in-one console (CCU, LED Light Source and Image Management System), camera head and iPad application. Our DYONICS™ shaver blades provide superior resection due to their sharpness and reduced clogging with their debris evacuation capabilities.14 GoFLO™ and Double® Pump Fluid Management Systems facilitate surgical access by expanding the joint space, providing haemostasis, and maintaining the saline environment necessary to perform arthroscopic procedures.
EAR, NOSE & THROAT (ENT)
In ENT, our COBLATION Technology has been used to remove tonsils and adenoids for over 15 years and is preferred by surgeons and patients for its ability to remove tissue at low temperatures with minimal damage to surrounding tissue.15 COBLATION Technology is also marketed for use in turbinate and laryngeal procedures.
Our RAPID RHINO™ Carboxymethylcellulose (CMC) Technology is featured in both dissolvable and removable nasal and sinus dressings and epistaxis treatment products. When mixed with water, CMC forms a cushioning gel that naturally drains from the body after several days and supports healing by maintaining a moist physical environment.
OUR PERFORMANCE IN 2018
|
Revenue |
Reported |
Underlying |
|
|
Sports Medicine Joint Repair |
$697m |
11% | 8% |
|
AET |
$600m |
2% | 3% |
|
Other Surgical Business*** |
$209m |
10% | 10% |
In 2018, strong growth in Sports Medicine Joint Repair franchise was driven by our shoulder repair portfolio. Within this, the recently acquired REGENETEN™ Bioinductive Implant for rotator cuff repair delivered more than 130% growth, performing ahead of expectations.
AET performance was held back by continued softness in mechanical and legacy radio-frequency resection. We expect the launch of the FLOW 90™ COBLATION™ wand for shoulder in the first half of 2019 to support better growth.
Other Surgical Businesses double-digit growth reflects strong demand for our robotic NAVIO Surgical System from both the Established and Emerging markets.
References
1 Bokor DJ, et al. MLTJ. 2015;5(3):144‑150.
2 Arnoczky SP, et al. Arthroscopy. 2017;33(2):278‑283.
3 Bokor DJ, et al. MLTJ. 2016;6(1):16‑25.
4 Douglass NP, et al. Arthroscopy. 2017;33(5):977‑985.e5.
5 Data on file Smith & Nephew. Report 15002117. 2013.
6 Data on file Smith & Nephew. Report 15002059. 2013.
7 Data on file Smith & Nephew. Report 15000897. 2010.
8 Amiel D, et al. Arthroscopy. 2004;20(5):503‑510.
9 Spahn G, et al. Knee Surg Sports Traumatol Arthrosc. 2008;16(6):565–573.
10 Spahn, G, et al. Arthroscopy. 2010;26(Suppl 9):S73‑80.
11 Spahn G, et al. Knee Surg Sports Traumatol Arthrosc. 2016;24(5):1560‑1568.
12 Gharaibeh M, et al. Cartilage. 2018;9(3):241‑247.
13 Voloshin I, et al. Am J Sports Med. 2007;35(10):1702‑1707.
14 Data on file Smith & Nephew. Report 15005165. 2016.
15 Woloszko J, et al. Proc of SPIE. 2003;4949:341‑352.
16 Washburn III R, et al. Arthrosc Tech. 2017:6(2);e297‑e301.
17 Mather RC, et al. J Bone Joint Surg Am. 2013;95:1993‑2000.
18 Lin JC, et al. J Am Med Dir Assoc. 2008;9(9):626‑632.
19 Schlegel TF, et al. J Shoulder Elbow Surg. 2018;27(2):242‑251.
* FDA cleared for use in the knee on all soft tissue types.
** These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198.
*** Includes ENT and NAVIO robotics system.
|
20 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
|
OUR FRANCHISES continued Advanced Wound Management Reducing the burden of wounds
Our Advanced Wound Management portfolio provides a comprehensive set of products to meet broad and complex clinical needs, to help healthcare professionals get CLOSER TO ZERO human and economic consequences of wounds. |
|
Our customers choose the breadth and depth of our portfolio, innovation, and expertise in order to achieve their ‘CLOSER TO ZERO’ goals. Simon Fraser President of Advanced Wound Management
|
|
ADVANCED WOUND MANAGEMENT
Because of the breadth and depth of our portfolio we are uniquely positioned to support customers who follow best practice guidelines, including managing wounds with T.I.M.E.
T.I.M.E. stands for Tissue non-viable, Infection and/or Inflammation, Moisture imbalance, and Edge of wound non-advancing. These represent critical barriers of wound healing. T.I.M.E. was first established as a concept for best practice wound management in 20031 by a panel of world leading experts, and has since been widely adopted around the world, becoming a staple reference framework for routine clinical practice.2
We use T.I.M.E. to help our customers navigate the complexity of product choices they face based on which clinical goal they may have, and we also use it to guide our own new product and programme development, as well as life cycle management, to ensure we remain relevant to the evolving clinical needs.
Having supported T.I.M.E. since its inception, at Smith & Nephew we are uniquely positioned to provide customers with differentiated and effective products via our Advanced Wound Care (AWC), Advanced Wound Devices (AWD) and Advanced Wound Bioactives (AWB) portfolio across each T.I.M.E.-based clinical need.
ADVANCED WOUND CARE (AWC)
Our AWC range covers several segments aimed at helping improve outcomes in the Infection and Moisture balance clinical goals of T.I.M.E.
In infection management, our silver-based dressings (ACTICOAT™, DURAFIBER Ag™ and ALLEVYN™ Ag) provide clinicians with a range of solutions to address individual patient needs in managing wound infection. ACTICOAT, for instance, is a fast-acting, highly effective antimicrobial3 for serious infection on a wide range of wounds. Our Cadexomer iodine based IODOSORB™ dressing is indicated outside the US to deliver best in class efficacy effective against biofilms across numerous clinically relevant in vitro tests,4 animal biofilm models4 and in clinical practice.5
In exudate (or moisture) management our products are designed to respond to varying levels of wound exudate providing appropriate wound fluid absorption, lock in and evaporation properties to promote an optimal wound healing environment. This helps patients get on with their lives as well as lowering costs for materials and nursing time by reducing unnecessary dressing changes. Our key growth brand in this space is the ALLEVYN range with two focus variants, ALLEVYN Gentle Border dressing (versatile and adaptable, so suitable for a wide variety of chronic and acute wounds6,7) and ALLEVYN LIFE dressing (our most advanced dressing, uniquely differentiated by its distinct quadrilobe shape which lasts for up to two times longer than any other dressing8). The ALLEVYN range was extended in 2018 through the launch of ALLEVYN LIFE Non‑Bordered to ensure the portfolio continues to meet broad needs.
The rest of our AWC range includes our film and post-operative dressings, skincare products and gels. Leading brands include OPSITE™ dressings, IV3000™, PROSHIELD™ and SECURA™.
ADVANCED WOUND BIOACTIVES (AWB)
Our AWB portfolio covers key product segments aimed at helping improve outcomes in the Tissue viability and wound Edge advancement clinical goals of T.I.M.E.
In this part of our business we focus on the commercialisation of topical biologics and a skin substitute that provide a unique approach to debridement, dermal repair, and tissue regeneration.
Our portfolio includes Collagenase SANTYL™ Ointment, OASIS® Wound Matrix and OASIS ULTRA Tri-Layer Matrix (a naturally-derived, extracellular matrix replacement product indicated for the management of both chronic and traumatic wounds) and REGRANEX™ (becaplermin) Gel 0.01%.
Our most significant product by sales in this segment is SANTYL Ointment9, the only FDA-approved biologic enzymatic debriding agent for chronic dermal ulcers and severe burns. SANTYL plays an integral role in debriding chronic dermal ulcers and severely burned areas.
|
|
Smith & Nephew Annual Report 2018 |
|
21 |
PICO 7
The UK’s National Institute for Health and Care Excellence (NICE) issued a Medtech innovation briefing26 on the prophylactic use of PICO which highlighted its potential to be more effective at preventing surgical site infections than standard surgical dressings. This is the only such briefing published by NICE on an NPWT device for preventing such complications.


|
22 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
OUR FRANCHISES continued
ADVANCED WOUND DEVICES (AWD)
Our AWD portfolio covers key product segments aimed at helping improve outcomes in the Tissue viability, Moisture balance, and wound Edge advancement clinical goals of T.I.M.E.
In the NPWT segment, the PICO™ Single Use Negative Pressure Wound Therapy System (sNPWT) brings the effectiveness of traditional NPWT in a modern, small portable system.10 It is designed for both open wounds and closed incisions, and leverages our proprietary AIRLOCK™ dressing technology.
During the year, we extended the PICO range with the introduction of two new models.
PICO 7 delivers a more efficient vacuum and superior leak management than the previous version11, includes an industry-first dressing-full indicator, which is intended to help reduce unnecessary dressing changes and wastage, and is over 25% quieter than the previous version.12
PICO 7Y, launched in Europe in 2018, is the first sNPWT system to include an innovative integrated Y connector enabling the utilisation of two dressings concurrently from one pump, in practice allowing for two wounds to be addressed at the same time, thereby potentially reducing cost.
The PICO evidence base continued to grow, validating the patient and provider benefits of the technology, with the publication of key studies in multiple indications including orthopaedics, vascular, plastics, OBGYN, breast reconstruction and chronic wounds.13‑24
With RENASYS™ NPWT system, our strategy is to simplify the delivery of NPWT, combining the advantages of PICO with the simplicity and power of RENASYS TOUCH, an intuitive touchscreen traditional NPWT device delivering advanced features to manage large, highly exuding wounds.23,24
This franchise also includes the VERSAJET™ Hydrosurgery system, a surgical debridement device used by surgeons to excise and evacuate non-viable tissue, bacteria and contaminants from wounds, burns and soft tissue injuries.25
OUR PERFORMANCE IN 2018
|
|
Revenue |
Reported |
Underlying |
|
Advanced Wound Care |
$740m |
3% | 1% |
|
Advanced Wound Bioactives |
$320m |
6% | 6% |
|
Advanced Wound Devices |
$215m |
10% | 9% |
In 2018, performance in Advanced Wound Care included good growth in the US, led by ALLEVYN LIFE and our pressure ulcer prevention strategy, offset by softness in some European countries.
In Advanced Wound Bioactives, performance from SANTYL, our largest product, was weaker than the previous year as volumes came under pressure. Following review of two large safety studies, the FDA approved the removal of the boxed warning from REGRANEX, and we will relaunch this product in early 2019.
Advanced Wound Devices delivered strong growth led by demand for our PICO sNPWT, which benefited from the launch of two new models in 2018.
References
1 Schultz et al., Wound Rep Reg 2003;11:1‑28.
2 Leaper et al. Int Wound J 2012;9 Suppl.2):1–19.
3 Wright et al. Am.Jnl.Inf.Contrl 1998; 26(6) 572‑577.
4 Fitzgerald et al. Wound Repair Regen. 2017;25,13–24.
5 Malone et al. J. Antimicrob. Chemother. 2017; 72,2093–2101.
6 Smith&Nephew DOF DS/14/318/R.2015.
7 Smith&Nephew DOF DS/09/013/R2.2009.
8 Joy et al. Jnl Wound Care 2015;24(7):312,314‑7.
9 SANTYL is indicated for chronic dermal ulcers and severely burned areas. Occasionally, slight transient erythema has been noted in surrounding tissue when applied outside the wound. One case of systemic hypersensitivity has been reported after 1 year of treatment with collagenase and cortisone. Use of SANTYL Ointment should be terminated when debridement is complete and granulation tissue is well-established. See full prescribing information for more details.
10 Hurd et al. Ostomy Wound Mngt. March 2014; Vol.60:3.
11 Smith&Nephew DOF DS.17/666/R2.
12 Smith&Nephew DOF DS/17/701/R.
13 Delhougne et al. Ostomy Wound Manage 2018; 64(1):26‑33.
14 Dingemans et al. Int. Orthopaedics 2018; 42(4): 747‑753.doi:10.1007/s00264‑018‑3781‑6.
15 Fleming, et al. Journal of Hospital Infection 2018; 99(1): 75‑80. doi:10.1016/j.jhin.2017.10.022.
16 Galiano et al. Plastic & Reconstructive Surgery Global. 2018;6(1):e1560.
17 Hyldig et al. Bjog 2018;doi: 10.1111/1471‑0528.15413.
18 Svensson-Björk et al, Wound Repair & Regeneration 2018;26(1):77‑86.
19 Yamaguchi et al. Jnl Dermatology 2018; 45(4): 483‑486. doi:10.1111/1346‑8138.14180.
20 Innocenti et al. J Reconstr Microsurg. 2018;Aug 15.
21 Edwards et al. Wounds UK. 2018;14:56–62.
22 Giannini et al, Jnl. Wound Care, 2018; 27(8):520‑525.
23 Forlee, et al. WUWHS, 2016;Florence.
24 Forlee, et al. EWMA; 2018;Poland.
25 Mosti, et al. Wounds. 2006;18(8):227‑237.
26 NICE Medtech Innovation briefing (MIB149) June 2018.
* These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198.
|
|
Smith & Nephew Annual Report 2018 |
|
23 |
The resources we need to deliver our products and serve our customers
| 24 | |
| 26 | |
| 27 | |
| 28 | |
| 29 | |
| 30 | |
| 32 |

|
24 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
A unifying purpose and culture of care, collaboration and courage to win
Smith & Nephew has a proud history of more than 160 years of improving health around the world. Whilst we have grown significantly from our beginnings as a small family pharmacy in Hull, England, our caring spirit has remained the same.
In 2018, led by our new management team, Smith & Nephew began the work to create a culture that, whilst rooted in caring, was also clearly aligned on a unifying purpose and culture of collaboration and courage to win.
Throughout the year, in addition to gauging our progress against our framework of Great Place to Work (GPTW), we engaged employees in a review of our existing culture and future aspirations. GPTW Pulse surveys were conducted in four of our major markets – US, China, UK and Australia/New Zealand – with an overall response rate of 75% and China again receiving country-level recognition. GPTW recognition was also received during the year in Austria, Ireland, Poland and the UAE.
In addition, we conducted a voluntary feedback survey and subsequent focus groups with participation from nearly 40% of the workforce to review our company culture. Together, this input formed the basis for a new purpose, Life Unlimited (see page 8), and our culture pillars of Care, Collaboration and Courage. These pillars represent the best of Smith & Nephew today, as well as what we aspire to be in the future.
CARE
Our culture pillar of Care means that we show empathy and understanding for each other, our customers and patients. We step into our customers’ shoes, anticipate their needs and deliver the highest levels of innovation and service.
We strive to have the best understanding of the patients whom we ultimately serve, and we develop our products with them in mind. And, our passion for what we do drives us to continuously improve and expand the positive impact that we have on the world.
In 2018, our people displayed this culture in numerous ways, including charitable donations and sponsorships of more than 50 organisations in the communities where we live and work. This extends to support of our own employees in times of need.
For example, in 2018, US employees who were displaced from their homes due to a gas pipeline explosion were provided lodging or heaters until their power returned.
We encourage all our employees to volunteer their time and talents by providing paid time for volunteer efforts. Many functions structured their team building activities around group volunteering opportunities such as Make a Wish Foundation events and Habitat for Humanity.
By continuously improving our own performance, we can increase our positive influence on the world. To encourage this, we offer advancement and development opportunities for our employees. Employee advancement is merit-based, reflecting performance as well as demonstration of our newly created Winning Behaviours, which underpin our culture pillars and replace our previous core competencies.
Each year Smith & Nephew conducts a comprehensive global development and capability review process to identify high potential employees and ensure they have well defined career development plans.
Employees are provided with opportunities to develop their skills and career through new assignments and on the job experiences. In addition, succession plans are in place for key executive roles and other critical positions across our business.
COLLABORATION
Our culture pillar of Collaboration means we work together as a team, based on mutual trust and respect. Through transparent and respectful communication, we are motivated by a shared purpose and understand the impact of our individual contributions on our collective goals. And, by encouraging different perspectives and leveraging our global experiences we achieve the best outcomes.
In 2018, we broadened our quarterly business performance communications to include live global webcasts featuring Namal Nawana and members of his executive team. These included an open question and answer dialogue with employees around the world in real-time. The feedback has been tremendously positive, increasing transparency and supporting our shared purpose.
We provide peer-to-peer recognition to celebrate achievements or just say ‘thank you’ via our Going the Extra Mile (GEM) programme. Awards range from simple notes to appreciation through substantial monetary awards.
We are committed to employment practices based on equal opportunities, regardless of colour, creed, race, national origin, sex, age, marital status, sexual orientation, or physical or intellectual disability. We believe a person’s ability to perform essential functions of a job is the only relevant criteria.
|
|
Smith & Nephew Annual Report 2018 |
|
25 |
In 2018, an internal evaluation showed that those teams with greater diversity achieved better results. The work also revealed that our people understand why valuing difference is important and our teams benefit from high levels of trust and respect.
We have raised awareness of preventing unconscious biases through our management and Human Resources training globally, carrying out a Talent Acquisition Diversity and Inclusion Masterclass. We also conducted inclusion workshops at the 2018 Managing Directors’ Meeting and numerous regional leadership business meetings.
We stepped-up our efforts to accelerate the development of women in our business. We have extended our Elevate women’s leadership development programme, including nearly 300 participants in 2018. We also attended the 2018 Conference of the Society of Women Engineers, to generate further awareness and recruit female talent in the science and engineering fields. In addition, in 2018, we added another female leader to our executive team.
COURAGE
Our culture pillar of Courage is about continuous learning, innovation and accountability. By staying curious, thinking big and having the humility to challenge our conventional ways of thinking, we push the boundaries of our industry. Fostering an entrepreneurial, can-do attitude we look for solutions and achieve them through talent and force of will. And, with a growth mindset, we have the capability and confidence to win, and we do so with integrity and the highest ethical standards. We start each year by setting clear and measurable objectives with a clear strategy communicated Company-wide.
The strategic imperatives and annual targets form the basis of individual objectives of every employee in the Company according to his or her role. Through this process, each employee can clearly see how their efforts contribute to the overall success of the business, which drives execution, accountability and engagement.
We continued to provide opportunities for all levels of the organisation to strengthen their skills through development programmes including Pioneer, Edge and Continuous Learning Journey. These programmes consistently received positive feedback from participants.
Smith & Nephew’s compensation also supports high-performance and accountability. Employees are compensated based on sustained performance that helps deliver timely and tangible results to drive the business forward and support our culture.
Having a robust compensation framework is vital as we seek to recruit high calibre people. By following this philosophy we have found that we not only attract, retain, and motivate talent, but it also helps drive better business results and provides an equitable work environment. We are Living Wage Accredited in the UK, voluntarily paying above the government required minimum as we believe employees should receive fair compensation for the work they do.
|
REDEFINING OUR CULTURE |
SUPPORTING WOMEN |
|
|
6,000 employees participated in our programme to define our new purpose and culture pillars. We used feedback surveys and ran workshops at our sites across the world, including in Japan. |
We encourage women to follow careers in STEM. Our Society of Women Engineers (SWE) chapter is thriving, with more than 110 members. We had a major presence at the 2018 SWE Conference, recruiting for talented new graduate engineers. |
For more information about how we are putting people first, download our Sustainability Report from our website.
|
|
|
|
|
|
|
|
|
|
NUMBER OF EMPLOYEES1 2018 |
|||||
|
Total employees |
Senior managers2 and above |
Board of Directors |
|||
|
16,377 |
788 |
12 |
|||
|
Male |
Female |
Male |
Female |
Male |
Female |
|
58% |
42% |
73% |
27% |
75% |
25% |
|
1 Number of employees at 31 December 2018 including part time employees and employees on leave of absence. 2 Senior managers and above include all employees classed as Directors, Senior Directors, Vice Presidents, Executive Officers and includes all statutory directors and Directors of our subsidiary companies.
|
|||||
|
26 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
OUR RESOURCES continued
Smith & Nephew has a strong reputation for integrity and ethical behaviour
CODE OF CONDUCT AND BUSINESS PRINCIPLES
Smith & Nephew earns trust with customers, healthcare professionals, government authorities, patients and the public by acting in an honest and fair manner in all aspects of its operations.
We expect the same from those with whom we do business, including vendors who provide us with products and services and distributors and independent agents that sell our products. Our Code of Conduct and Business Principles governs the way we operate to achieve these objectives.
Smith & Nephew takes into account ethical, social, environmental, legal and financial considerations as part of its operating methods. We have a robust whistle-blowing system in all jurisdictions in which we operate which is benchmarked against industry metrics. We are committed to upholding the promise we make in our Code of Conduct to not retaliate against anyone who makes a report in good faith.
GLOBAL COMPLIANCE PROGRAMME
Smith & Nephew has implemented what we believe to be a world-class Global Compliance Programme that helps our businesses comply with laws and regulations.
This includes: Board and executive oversight committees; global policies and procedures; on-boarding and annual training for employees and managers; training for distributors and agents and higher-risk vendors; monitoring and auditing processes; reporting channels and employee-recognition for demonstrating our values in their everyday work.
We provide resources and tools to guide employees to make decisions that comply with the law, local industry codes and our Code of Conduct. We review and approve significant interactions with healthcare professionals or government officials in advance. We regularly assess existing and emerging risks in the countries in which we operate.
We assess the compliance controls in Smith & Nephew’s businesses. We conduct audits, supported by data analytics, with central and local monitoring. We review the issues our testing generates to identify patterns.
New distributors and other higher-risk third parties are subject to screening and are contractually obligated to comply with applicable laws and our Code of Conduct. Compliance training and certifications are included in this process, including guidelines for those who need to enter the operating room when acting on our behalf.
Senior leaders, including all Vice Presidents and above, are required to complete an annual certification to the Chief Executive Officer to confirm the implementation of required policies. Managers and employees make an annual compliance certification and conflict of interest disclosure.
We constantly seek new ways to enhance our Compliance Programme. New measures in 2018 included expanding the compliance ambassador process where selected sales staff serve as compliance contact for their peers for some training and questions, successfully completing a pilot for an enhanced root cause analysis methodology for recurring issues and implementing additional processes and training on data privacy.
AN ETHICAL EMPLOYER
We recruit, employ and promote employees on the sole basis of the qualifications and abilities needed for the work to be performed. We do not tolerate discrimination on any grounds and provide equal opportunity based on merit.
We do not use any form of forced, compulsory or child labour. We support the Universal Declaration of Human Rights of the United Nations. This means we respect the human rights, dignity and privacy of the individual and the right of employees to freedom of association, freedom of expression and the right to be heard.
As a global medical technology business, Smith & Nephew recognises that we have a responsibility to take a robust approach to preventing slavery and human trafficking. Smith & Nephew is committed to preventing slavery and human trafficking in its corporate activities, and its supply chains. Our full policy on preventing slavery is available on our website.
|
|
Smith & Nephew Annual Report 2018 |
|
27 |
|
We put customers at the heart of our commercial model |
|
Our customers are the providers of medical and surgical treatments and services in over 100 countries worldwide, ranging from orthopaedic surgeons to wound care nurses, general practitioners and other clinicians, but increasingly also economic stakeholders. |
|
These include purchasing professionals in hospitals, healthcare insurers, materials managers and others. We serve these customers through our sales force and other channels. Our sales representatives are highly trained and skilled individuals. Becoming a sales representative requires intense training, including passing a strict certification programme. Depending on their area of specialism, representatives in our surgical businesses must be able to demonstrate a detailed knowledge of all the surgical instruments used to implant a device, or have specific understanding of the various surgical techniques a customer might use. In our Advanced Wound Management business, sales representatives must have a detailed understanding of how patients live with wounds and how clinicians seek to prevent and treat them, as well as deep knowledge of the clinical and economic benefits of using our products within treatment protocols. Once a sales representative is certified, they typically spend the majority of their time working directly with and supporting customers, or identifying and contacting new customers. They help to provide in-hospital support to aid in the safe and effective use of our range of advanced medical technologies and techniques. In 2018, we began the implementation of a global franchise structure with dedicated presidents of Orthopaedics, Sports Medicine & ENT and Advanced Wound Management to direct and support our customer facing activities. This new structure will replace our regional selling model in 2019. Under the new structure, each president will lead their franchise with global upstream marketing responsibility and full commercial responsibility for their franchise in the US. They will also lead one or more shared global commercial support teams, in the critical areas of professional education, sales training and healthcare economics. In addition, the president of Sports Medicine & ENT has commercial responsibility for Latin America and Canada. Outside of the Americas, our commercial activities will be run through two regions – Europe, Middle East and Africa, and Asia Pacific – each under a president. Under these presidents will be country clusters, a group of countries, based on geographic proximity, critical mass of revenue, and similar go-to-market strategies. They will be led by a single managing director and have business unit leads for each franchise. This structure will reduce complexity and take out administrative costs, and importantly will bring us closer to our customers. In 2018, we began to make these changes while keeping stability in our selling and customer-facing organisations. |
|
28 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
OUR RESOURCES continued
Efficiently delivering products of the highest quality
Smith & Nephew takes great pride in its expertise in manufacturing products to the highest quality and ensuring they reach our customers in a timely manner.
We operate manufacturing facilities in nine countries across the globe and have central distribution facilities in the US, Europe and Asia. Products are shipped to individual country locations to meet customer requirements.
Manufacturing is a dynamic process and our Global Operations leadership team is focused on successfully supporting delivery of the Group’s strategic priorities by ensuring our footprint and expertise is ready to respond to geographical growth, new product development, greater external regulatory scrutiny and the commercial pressure to be ever more efficient.
Products for our Orthopaedics franchise are made in sites in Memphis (Tennessee, US), Aarau (Switzerland), Tuttlingen (Germany), Beijing (China), Warwick (UK), Puschino (Russia) and Devrukh (India). Memphis is our largest location and is home to the design and manufacturing process of the OXINIUM Oxidised Zirconium, a patented metal alloy available for many of our knee and hip implant systems as part of our VERILAST technology.
In Sports Medicine Joint Repair, products are manufactured at our Mansfield (Massachusetts, US) and Alajuela (Costa Rica) facilities.
The majority of our Advanced Wound Management products are manufactured at our facilities in Hull (UK), Suzhou (China) and Curaçao. We have also invested in a new facility in Fort Worth, Texas, to support our Advanced Wound Bioactives franchise. Our Oklahoma City facility in the US produces and services electro/mechanical capital equipment as well as single-use sterile devices and also assembles some of our NPWT devices using components from third parties.
We procure raw materials, components, finished products and packaging materials from suppliers in various countries. These include metal forgings and castings for orthopaedic products, optical and electronic sub-components for Sports Medicine Joint Repair products, active ingredients and semi-finished goods for Advanced Wound Management as well as packaging materials across all product ranges. Suppliers are selected, and standardised contracts negotiated, by a centralised procurement team wherever possible, with a view to ensuring value for money based on the total spend across the Group. On an ongoing basis, we work closely with our key suppliers to ensure high quality, delivery performance and continuity of supply.
We outsource certain parts of our manufacturing processes where necessary to obtain specialised expertise or to lower cost without undue risk to our intellectual property. Suppliers of outsourced products and services are selected based on their ability to deliver products and services to our specification, and adhere to and maintain an appropriate quality system. Our specialist teams work with and monitor suppliers through on-site assessments and performance audits to ensure the required levels of quality, service and delivery.
Our Global Supply Chain team ensures that our products reach our internal and external customers where and when they are needed, in a compliant and efficient manner. We operate main holding warehouses for surgical products, one in each of Memphis (TN, US), Columbus (OH, US), Baar (Switzerland) and Singapore. These facilities consolidate and ship to local country and distributor facilities. Our distribution hubs for Advanced Wound Management products are located in Neunkirchen (Germany), Derby (UK) and Lawrenceville (Georgia, US).
QUALITY AND REGULATORY AFFAIRS
Quality is of paramount importance to Smith & Nephew. In 2018, we restructured the global Quality and Regulatory Affairs function to ensure consistent high standards across the Group. This function is led by the Chief Quality and Regulatory Affairs Officer, a new role reporting directly to the Chief Executive Officer.
Requirements of global regulatory agencies have become more stringent in recent years and we expect them to continue to do so. The team is leading a major Group-wide programme to prepare for implementation of the European Union (EU) Medical Devices Regulation (MDR), which came into force in May 2017, with a three-year transition period until May 2020. The regulation includes new requirements for the manufacture, supply and sale of all CE marked products sold in Europe and requires the re-registration of all medical devices, regardless of where they are manufactured.
Quality and Regulatory Affairs has also provided leadership in preparing the Group for Brexit, which has required the management of changes to our European Authorised Representative strategy and Notified Body relationships. Finally, the team continued to support the expansion of our portfolio globally through the registration of new products and existing products in new markets.
|
|
Smith & Nephew Annual Report 2018 |
|
29 |
|
Supporting the safe and effective use of our products Smith & Nephew is dedicated to helping healthcare professionals improve the quality of care for patients. We are proud to support the development of surgeons and nurses by providing skills training and education on our products and techniques. |
||
|
In 2018, we provided more than 30,000 instances of training to surgeons through our Smith & Nephew training centres in the US, UK and China, as well as running many courses at third party centres around the world. In 2018, we opened a new surgical training centre in Phoenix (Arizona) to bring professional development and skills training to customers, primarily in the West and Southwest of the United States, complementing Smith & Nephew’s existing US facilities in Memphis (Tennessee), Andover (Massachusetts), Austin (Texas) and Plymouth (Minnesota). Working under expert guidance, attendees learn new techniques and refine skills, to ensure the safe and effective use of our products. These courses are attended by residents, fellows and practising surgeons who work together to review, discuss and train on current and forward-looking surgical techniques in their areas of clinical expertise. Our courses help up-and-coming surgeons develop trust and gain the experience and confidence necessary to become experts in their field. |
Thousands of nurses receive face-to-face training from Smith & Nephew representatives every year, including attending courses at our centres, and through our representatives visiting them at their place of work. In 2018, more than 20,000 clinicians benefited from our specialist wound care education training courses. In addition, we provide healthcare professionals our online resources such as the Global Wound Academy, The Wound Institute and, for surgeons, our Education and Evidence website. Recently we began utilising innovative, digital technologies to accelerate the learning experience of surgeons. In 2018, we provided digital training on Smith & Nephew products and techniques to 225,000 healthcare professionals, a 25% increase over the prior year. ECC ENDORSED BY ROYAL COLLEGE OF SURGEONS In 2018, Smith & Nephew’s Expert Connect Centre (ECC) in Watford, UK became the first commercial surgical training facility in Europe to be accredited by the Royal College of Surgeons. The recognition enables delegates to receive Continuing Professional Development (CPD) points when attending Smith & Nephew sessions, demonstrating their commitment to developing their surgical skills. |
|
|
3D PRINTING OF REDAPTCUPS |
||
|
Additive manufacturing (AM), commonly referred to as 3D printing, is a novel manufacturing method that involves the use of a laser or electron beam, for example, to sinter polymer or metal powders into a solid part that is built layer‑by-layer. The REDAPT Revision Femoral System includes a Fully Porous Acetabular Cup featuring Smith & Nephew’s unique CONCELOC Advanced Porous Titanium. This mimics the structure of cancellous bone, allowing ingrowth for improved fixation and stability. |
||
|
30 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
|
OUR RESOURCES continued We are increasing our investment in new products Smith & Nephew’s global Research & Development (R&D) function supports the Group’s strategic imperatives by delivering innovative system solutions that aim to improve clinical and healthcare economic outcomes. We do this in partnership with our customers, executing new product development and clinical programmes across the enterprise. |
|
Innovation is the lifeblood of our Company. We strive to partner with our customers to create meaningful solutions for unmet needs, with the goal of improving outcomes. Vasant Padmanabhan President of Research & Development
|
In 2018, we invested $246 million in R&D, equivalent to 5% of Group revenue. Over time, we are committed to increasing this investment, driven by the needs of the business to support sustainable growth. In 2018, we launched a number of major new products and publications with evidence of clinical and economic value. Our major new product launches included the full commercial release of the bi-cruciate retaining JOURNEY II XR total knee arthroplasty (TKA) system, updates and extensions to our REDAPT Revision Hip System, the EVOS SMALL Plating System in Trauma, a suite of all-suture anchor shoulder repair systems in sports medicine and two new versions of our leading PICO Single Use Negative Pressure Wound Therapy System (sNPWT).
We published more than 200 different abstracts and publications in peer-reviewed journals; a significant increase compared to previous years, and the result of increased investment in clinical studies. Highlights included 14 abstracts accepted at the World Arthroplasty Congress in Rome in April and the completion of one of the largest multi-centre retrospective patient cohorts ever studied with the JOURNEY II BCS TKA system. A number of studies highlighted how PICO can help manage scarring and surgical site infections. We were also successful in securing the NICE Medtech Innovation Briefing for PICO described on page 21.
OUR ENTERPRISE R&D OPERATING MODEL
Our enterprise R&D model provides governance and simple processes for new product development, starting with front end innovation and research, moving through new product development and launch, and ending with support of released products.
Project selection is critical; we focus on projects that will make a meaningful difference to our customers and their patients. This includes investing in incremental innovation to improve existing products. It also involves driving greater efficiency through innovation, potentially reducing our costs of goods. Finally, we aim to transform our business using disruptive technologies, services and business models.
Following project selection, the team challenges itself to execute flawlessly. This means developing the right product at the right cost and quality, and supported by clinical evidence. Our R&D experts in the UK, US, Europe, Singapore, China and India have extensive customer and sector knowledge, which is augmented by interaction with our marketing teams. Strict criteria are applied to ensure new products fulfil an unmet clinical need, have a strong commercial rationale, and are technologically feasible.
R&D works closely with the marketing, clinical, regulatory affairs, manufacturing and supply chain management teams to ensure we can produce new products to clinical, cost and time specifications.
We also continue to invest in scouting for new technologies, identifying complementary opportunities in our core and adjacent segments. In addition, we invest in small companies developing compelling technologies in our franchise areas through our incubation fund, and provide our expertise to help the development process, including supporting clinical studies, and typically secure preferred access to technology as it nears market readiness.
We work in partnership with academia. As an example, with the University of Hull we have created one of the world’s largest Wound Care Research Clusters and with Imperial College London we are developing enhanced surgical techniques relating to ligament function, biomechanics and soft tissue injuries of the knee.
We look to support our innovations with compelling evidence of clinical and economic value. The global R&D function includes our Clinical, Medical and Scientific Affairs (CMSA) teams, led by the Chief Medical Officer. This team ensures that, from conception, plans are developed to support product launches with the evidence increasingly required by clinicians, payers and regulators. Our products undergo clinical and health economic assessments both during their development and post-launch.
For 2019, we have a strong pipeline, with a number of important launches planned, and also expect to maintain the high cadence of clinical and economic evidence.
|
|
Smith & Nephew Annual Report 2018 |
|
31 |
|
|
|
TREATING DIFFICULT REVISIONS |
|
|
|
The REDAPT Revision Femoral System includes an additive, or 3D printed Fully Porous Acetabular Cup. With a number of new REDAPT Augments, to be used in conjunction with the porous shell, we are enabling surgeons to treat more difficult acetabular revisions.
|
||
|
|
WIDENING OUR PORTFOLIO |
||
|
|
The Q-FIX™ CURVED, Q-FIX MINI and SUTUREFIX CURVED All-Suture Anchor systems are important additions to Smith & Nephew’s sports medicine portfolio. For use in procedures where space is limited and the anatomy can be difficult to access, they are designed to aid in optimal suture anchor placement during drilling and insertion.
|
||
|
WORLD FIRST |
|||
|
The new PICO 7Y Single Use Negative Pressure Wound Therapy System (sNPWT) with AIRLOCK Technology is the first sNPWT system to include an innovative integrated Y extension. This enables the utilisation of two dressings concurrently from one pump, in practice allowing for two wounds to be addressed at the same time, thereby potentially reducing cost. |
|||
|
EXPANSIVE, USER FRIENDLY SYSTEM |
|
||
|
The EVOS SMALL Plating System is indicated for fixation of small and long bone fractures in adults and children. It is an expansive, user friendly system with multiple fixation options including non-locking, locking, variable-angle locking, optimised plate contours and screw trajectories as well as a low profile construct. |
|
|
|
|
32 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
Sustainability is better business
SUSTAINABILITY IS AT THE CORE OF THE BUSINESS
In 2016, we launched our Group Sustainability Strategy, setting out our aspirational goals and targets. The strategy is integrated with our Group Business Strategy. This ensures that the three main aspects of sustainability – economic prosperity, social responsibility and environmental stewardship – are tackled together.
This is a summary report of our sustainability activities and progress in 2018. Our annual Sustainability Report, published at the same time as this Annual Report, describes the Group Sustainability Strategy and its associated goals in more detail. It also specifies targets to move our performance towards these goals, and provides detailed information regarding the progress made during 2018. It is available on our website.
GROUP SUSTAINABILITY STRATEGY
Smith & Nephew has been and remains committed to working in a sustainable, ethical and responsible manner everywhere we do business. We are proud of our achievements over many years, as witnessed by our recurring inclusion in leading indices such as FTSE4Good and the Dow Jones Sustainability Index.
At the heart of the Group Sustainability Strategy are 10 long-term aspirational goals. These encompass all aspects of our business, and inform and drive our business strategy.
The Board has endorsed these and executive management is behind them. These goals are set out on the next page.
The Board has evaluated the social and environmental risks as part of their ongoing risk management duties and has concluded that none of these risks are material in the context of the Group as a whole.
We have set medium-term targets to 2020 which support our longer-term goals. These are discussed in more detail in our 2018 Sustainability Report.
2018 was a year in which we accelerated progress toward the achievement of our 2020 targets. We once again delivered improvements across our traditional areas of focus: employee health and safety, carbon emissions and water consumption. In addition, we deepened and broadened our understanding of our impacts in the areas of material efficiency, life cycle environmental impacts, and labour practices. We deployed the social responsibility strategy developed in the previous year, positively contributing to employee engagement and supporting the communities in which we operate.
|
SUSTAINABILITY VISION AND MISSION We envision a world in which healthcare professionals have access to the solutions they need to help patients restore their health, engage in society, enhance the environment and improve their wellbeing. Our sustainability strategy aims to achieve this vision. It outlines the steps we take with a view to leading our industry in the development and use of products and services that: Satisfy unmet health needs and promote greater access to treatment; Offer easier, better, faster and more effective treatment, enabling productive engagement in society; Prioritise materials that are reused, remanufactured or recycled; Are manufactured using raw materials sourced from an environmentally and socially sound supply chain; Use natural resources efficiently; Are manufactured by processes that are not hazardous to people or the environment; and Implement the most sustainable product options. |
|
|
Smith & Nephew Annual Report 2018 |
|
33 |
OUR PERFORMANCE
Our 10 long-term aspirational goals
|
2020 target |
Performance to 31 December 2018 |
|
ZERO WORK-RELATED INJURIES AND ILLNESSES ACROSS THE VALUE CHAIN |
|
|
10% reduction in Total Injury Rate (TIR) from 2016 actual. |
13% reduction achieved (2016 TIR = 0.52, 2018 TIR = 0.45). |
|
WATER: Total water impacts of products and solutions balanced with local human and ecosystem needs. |
|
|
Water footprint available for products accounting for 75% of revenue and considerations embedded in new product development process. Total potable water consumption no higher than 2016 actual. |
Water reduction of 21% achieved since 2016. Products and tools identified as per target. Life cycle assessment for a representative product underway with completion expected early 2019. |
|
WASTE: All materials are either shipped as part of product or returned for beneficial use. |
|
|
Total material efficiency estimated for products accounting for 75% of revenue and 80% or more of waste generated reused, recycled or recovered. |
79% of our total waste reused, recycled or energy recovered from, up from 74% in 2016. Products and tools identified as per target. Life cycle assessment underway. |
|
CARBON: 80% absolute reduction in total life cycle greenhouse gas emissions by 2050. |
|
|
Estimate total life cycle greenhouse gas emissions of products accounting for 75% of revenue. Total Scope 1 & 2 greenhouse gas emissions reduced by 10% from 2016 actual. |
Products and tools identified as per target. Life cycle assessment underway. 16% reduction in emissions since 2016. |
|
ETHICAL BUSINESS PRACTICES: All activities conducted in compliance with applicable International Labour Organization (ILO) conventions, involve no environmental degradation, and are free from corruption. |
|
|
Labour practices throughout the supply chain associated with products accounting for 75% of revenue compliant with applicable ILO conventions. |
Products identified and assessment to applicable ILO conventions completed for internal operations. Engagement with upstream suppliers and downstream distributors and agents initiated. |
|
ZERO PRODUCT-RELATED AND SERVICE-RELATED PATIENT INJURIES |
|
|
Robust system in place to detect, record, investigate and eliminate root cause of product and service-related patient injuries. |
Systems in place. Data being used to craft models to identify at-risk attributes. |
|
ROBUST SOCIAL RESPONSIBILITY PROGRAMMES that contribute to the attraction and retention of top talent. |
|
|
Social responsibility strategy which aligns philanthropy, employee volunteering and wellness to the business strategy. |
Social responsibility strategy in place. Alignment to new strategic imperatives under way. |
|
PRODUCTS AND SERVICES are aligned to market economic, social and environmental expectations and anticipate future market conditions. |
|
|
Sustainability attributes described for products accounting for 75% of revenue. Robust emphasis on sustainability attributes of new products/services in place. |
Products identified and sustainability attributes agreed. New product development (NPD) sustainability focus planning under way. |
|
STRATEGIC RISKS AND OPPORTUNITIES are understood and business activities are aligned to risk appetite. |
|
|
Enterprise risk management arrangements are embedded in the routine business decision-making process. |
Risk register reinvigorated. ‘Deep dive’ programme instituted with focus on both assurance that relevant risks have been identified and effectiveness of mitigating actions is accurately assessed. Actions to further embed into the business decision-making process are planned for 2019. |
|
ENVIRONMENTAL, SOCIAL, AND ECONOMIC impacts of business activities fully understood and appropriately balanced. |
|
|
Formal programmes in place to measure/assess the economic, social and environmental impacts of (1) potential acquisitions, (2) technologies to be extended to Emerging Markets, (3) innovative business models, (4) cost-of-quality reduction initiatives, and (5) manufacturing siting, functional optimisation and site utilisation alternatives. |
Launched Enterprise Risk Management Policy and Manual and provided training in risk identification and mitigation. |
|
34 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
SUSTAINABILITY continued
Our plan focuses on both the foundational and competitive advantage elements required to deliver our value proposition sustainably. We employ a continuous improvement approach based upon the implementation of forward-looking solutions (such as investing in new materials and processes that provide significant benefits with respect to human rights, safety, energy, waste and/or communities) and bridging technologies to secure future game-changing performance.
EMPLOYEE SAFETY, WELLNESS AND VOLUNTEERING
A healthy and safe working environment is fundamental to the way we work at Smith & Nephew. We must ensure that the safety of our employees and those who work with us is given the highest priority when we perform our daily activities in our offices around the world, when we visit customers and in our manufacturing environment.
Engagement with the communities in which we operate continues to broaden and deepen through the active attention of site leadership, empowerment of local camaraderie councils as well as broader application of company-paid volunteering allowance and company matching of employee donations to charity. We continue to strengthen and deepen employee wellness programmes with a focus on enabling healthy lifestyle choices.
SOCIAL RESPONSIBILITY STRATEGY IMPLEMENTATION
We further improved our understanding of performance against relevant labour standards in both our operations and in our supply chain. We undertook measures to improve performance across the entire business, taking significant steps toward the implementation of the Social Responsibility Strategy which was adopted in 2017.
Our Social Responsibility Strategy aims to improve the alignment of our charitable donations, volunteering, wellness and professional development with both our Group Business Strategy and the needs and desires of our employees. Our goal is to impact positively both employee engagement and the quality of life in communities in which we operate.
In 2018, we improved our understanding of product and service attributes which are important to customers and our employees’ view of the role of the organisation in society.
In 2019, we will further drive labour practice improvements in our supply chain and turn our attention more fully to identifying and delivering the socially responsible attributes which help drive quality of life in the communities in which we operate.
CO2e REPORTING METHODOLOGY, MATERIALITY AND SCOPE
We report the carbon footprint of our Scope 1 and 2 greenhouse gas (GHG) emissions in tonnes of CO2 equivalent from our business operations for the calendar year ended 31 December 2018. Our focus is on the areas of largest environmental impact including manufacturing sites, warehouses, R&D sites and offices. Smaller locations representing less than 2% of our overall emissions are not included. Acquisitions completed before 2018 are included in the data.
Our GHG emissions reporting represents our core business operations and facilities which fall within the scope of our consolidated financial statements. Primary data from energy suppliers has been used wherever possible.
| 2018 | 2017 | 2016 | |
|
CO2e emissions (tonnes) from: |
|||
|
Direct emissions |
9,956 | 9,451 | 9,822 |
|
Indirect emissions |
67,886 | 76,107 | 82,415 |
|
Total |
77,842 | 85,558 | 92,237 |
|
Intensity ratio |
|||
|
CO2e (t) per $m sales revenue |
15.9 | 17.8 | 19.6 |
|
CO2e (t) per full-time employee |
4.7 | 5.2 | 5.9 |
Revenue: 2018: $4.9bn; 2017: $4.8bn; 2016: $4.7bn. Full-time employee data: 2018: 16,681; 2017: 16,333; 2016: 15,584.
We report our emissions in two ‘scopes’.
Scope 1 figures include: Direct sources of emissions which mainly comprise the fuels we use on-site, such as gas and heating oil and fugitive emissions arising mainly from the losses of refrigerant gases.
Scope 2 figures include: Indirect sources of emissions such as purchased electricity and steam we use at our sites.
Location-based emissions are calculated in compliance with the WRI/WBCSD GHG Protocol Corporate Accounting and Reporting Standard and have been calculated using carbon conversion factors published by BEIS/DEFRA for 2018. We have applied the emission factors most relevant to the source data, including DEFRA 2018 (for UK locations), IEA 2016 (for overseas locations) and for the US we have used the most recently available US EPA ‘Emissions & Generation Resource Integrated Database’ (eGRID) for the regions in which we operate. All other emission factors for gas, oil, steam and fugitive emissions are taken from DEFRA 2018.
|
|
Smith & Nephew Annual Report 2018 |
|
35 |
We improved performance, delivered meaningful efficiency savings, and generated good cash flow

|
36 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
|
Chief Financial Officer’s Review Our performance accelerated across the year and we take good momentum into 2019 |
DEAR SHAREHOLDER
Smith & Nephew delivered a solid financial performance in 2018. We improved performance, delivered meaningful efficiency savings, sustained investments in R&D for growth and continued to generate strong cash flow. This was achieved while also making important changes to our leadership, structures and culture which all position us well for further progress in 2019 and beyond.
2018 PERFORMANCE
Group revenue in 2018 was $4,904 million, an increase of 3% on a reported basis and 2% on an underlying basis.1 Our performance accelerated across the year, with 3% revenue growth on an underlying basis1 in the second half, and we take good momentum into 2019.
The reported operating profit for 2018 was $863 million, an 8% reduction from the previous year primarily reflecting the costs of the Accelerating Performance and Execution (APEX) restructuring programme as described below.
Trading profit1 for the year was $1,123 million and the trading profit margin was up 90bps to 22.9%. This reflects both the 50bps benefit of a one-off legal settlement and improved trading performance and cost control.
The reported tax rate was 15.1% (2017: 12.7%). The tax rate on trading results1 for the year to 31 December 2018 was 16.1% (2017: 17.1%). This was lower than the guided rate of between 20‑21% mainly due to a one-off benefit from a tax provision release following expiry of statute of limitations and a beneficial geographical mix of profits.
The reported tax rate was lower than the tax rate on trading results as a higher proportion of non-trading items were in higher tax jurisdictions, notably the US.
Basic earnings per share (‘EPS’) was down 13% to 76.0¢, also primarily due to APEX restructuring charges. Adjusted earnings per share1 (‘EPSA’) was up 7% at 100.9¢, reflecting the legal settlement gain, improved trading performance and the lower tax rate on trading results.
I’m pleased to report that trading cash flow1 was $951 million, up from $940 million in 2017, and we had another year of strong cash conversion (as defined on page 195) at 85% (2017: 90%). Return On Invested Capital (ROIC1 – as defined on page 198) was 12.5% (2017: 14.3%), reflecting the reduction in operating profit over the prior year noted above.
CAPITAL RETURNS
The appropriate use of capital on behalf of shareholders is important to Smith & Nephew. The Board believes in maintaining an efficient, but prudent, capital structure, while retaining the flexibility to make value enhancing acquisitions. This approach is set out in our Capital Allocation Framework which we used to prioritise the use of cash and ensure an appropriate capital structure.
Our commitment, in order of priority, is to:
|
1. |
Continue to invest in the business to drive organic growth; |
|
2. |
Maintain our progressive dividend policy; |
|
3. |
Realise acquisitions in-line with strategy; and |
|
4. |
Return any excess capital to shareholders. |
Net debt2 was $1,104 million at year-end, a decrease of $177 million from $1,281 million at 31 December 2017. As part of our strategy to expand in higher growth markets, we expect to participate actively in value-enhancing M&A opportunities, and I am pleased with progress of our REGENETEN business, acquired from Rotation Medical in Q4 2017, which exceeded expectations throughout 2018. We have a strong balance sheet and substantial capacity for acquisitions. Appropriate financing for larger acquisitions would be considered on a case-by-case basis, as opportunities arise.
CETERIX ACQUISITION
In January 2019 we completed the acquisition of Ceterix Orthopaedics, Inc., the developer of the NovoStitch Pro Meniscal Repair System. The acquisition supports the Company’s strategy to invest in innovative technologies that address unmet clinical needs. The cost of the acquisition was $45 million upon completion, $5 million deferred and up to a further $55 million over the next five years, contingent on financial performance.
EFFICIENCY
Our new Strategic Imperatives include Become the Best Owner. One of the ways we will do this is through simplifying the organisation and its processes, including through the APEX programme, initiated at the end of 2017.
We are on track across all three workstreams of 1) Manufacturing, Warehousing and Distribution, 2) General and Administrative (G&A) Expenses, and 3) Commercial Effectiveness. APEX is expected to drive an annualised benefit of $160 million by 2022 for a one-off cost of $240 million.
|
|
Smith & Nephew Annual Report 2018 |
|
37 |
|
In 2018, APEX incurred restructuring costs of $120 million with actions undertaken that resulted in benefits of approximately $60 million in the year. Graham Baker Chief Financial Officer |
We continue to keep the programme under review to ensure the delivery of benefits and assess any further incremental opportunities that may arise.
In 2018, APEX incurred restructuring costs of $120 million with actions undertaken that resulted in benefits of approximately $60 million in the year.
During the year, I was pleased to take on responsibility for our Global Business Services (GBS) and IT functions. Both functions have important roles to play both in supporting delivery of the business strategy and delivering efficiencies as part of the APEX programme.
Expert people, improved systems and effective processes are core to those functions and both functions provide high quality service to the business from a mixture of locations, both near to our business and in cost effective regional centres.
We now have fully functioning GBS centres in Costa Rica, Malaysia, India and Poland. Our employees in these locations are integral members of the Smith & Nephew team and we expect all the centres to expand to take on more work in the coming year.
UK’S WITHDRAWAL FROM THE EU
The Group does not believe that the UK’s decision to leave the EU will have a significant impact on our long-term ability to conduct business into and out of the EU or UK. We are making good progress with our preparations for the various scenarios. Early in 2019, our preparations were assessed by external advisors on behalf of the Internal Audit function, with the findings reviewed by the Audit Committee.
OUTLOOK
Our 2019 guidance for further improvement in underlying performance at the top and bottom line is an important step in realising our medium-term ambition to outgrow our markets. In terms of revenue, we expect our underlying growth to be in the range of 2.5% to 3.5% in 2019. On a reported basis this equates to a range of around 1.8% to 2.8% at exchange rates prevailing on 1 February 2019 and including the effect of the Ceterix acquisition.
We expect 2019 trading profit margin to be in the range of 22.8% to 23.2%, a further 40‑80bps improvement over 2018, excluding the one-off 50bps legal settlement benefit. The tax rate on trading results for 2019 is expected to be in the range 19% to 21%, subject to any material changes to tax law, or other one-off items.
Following the implementation of the new franchise-based organisational structure from January 2019, we have concluded that we will have reportable segments for our three main franchises; Orthopaedics, Sports Medicine & ENT and Advanced Wound Management, from 2019 onwards. I hope that investors and others will benefit from the additional information this will provide.
Yours sincerely,

Graham Baker
Chief Financial Officer
|
Revenue $4,904m Reported Underlying1 +3% +2% |
|
|
Earnings per share EPS 76.0¢ |
‑13% |
|
Adjusted earnings per share (EPSA) 100.9¢
|
+7% |
|
1 These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198. 2 Net debt is reconciled in Note 15 to the Group accounts. |
|
|
38 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
We have a strong balance sheet and substantial capacity for acquisitions
GROUP PERFORMANCE
|
2018 $ million |
2017 $ million |
Change $ million |
|
|
Consolidated income statement |
|||
|
Revenue |
4,904 | 4,765 | 139 |
|
Operating profit |
863 | 934 | (71) |
|
Trading profit1 |
1,123 | 1,048 | 75 |
|
Profit before tax |
781 | 879 | (98) |
|
Attributable profit |
663 | 767 | (104) |
|
EPS |
76.0¢ |
87.8¢ |
(11.8¢) |
|
EPSA1 |
100.9¢ |
94.5¢ |
6.4¢ |
NON-IFRS MEASURES
The underlying increase in revenues by market reconciles to reported growth, the most directly comparable financial measure calculated in accordance with International Financial Reporting Standards (IFRS), as follows:
|
2018 |
2017 |
Reported
growth |
Underlying growth |
Reconciling items |
||
|
Acquisitions/ |
Currency |
|||||
|
US |
2,354 | 2,306 | 2% | 1% | 1% | 0% |
|
Other Established Markets2 |
1,693 | 1,658 | 2% | 0% | 0% | 2% |
|
Emerging Markets2 |
857 | 801 | 7% | 8% | 0% | (1%) |
|
Total |
4,904 | 4,765 | 3% | 2% | 0% | 1% |
|
|
|
|
|
|
|
|
Trading profit reconciles to operating profit, the most directly comparable financial measure calculated in accordance with IFRS, as follows:
|
2018 |
2018% |
2017 |
2017% | |
|
Operating profit |
863 | 17.6% | 934 | 19.6% |
|
Acquisition and disposal related items |
(7) | (0.1%) | (10) | (0.2%) |
|
Restructuring and rationalisation costs |
120 | 2.4% |
– |
– |
|
Amortisation and impairment of acquisition intangibles |
113 | 2.3% | 140 | 2.9% |
|
Legal and other |
34 | 0.7% | (16) | (0.3%) |
|
Trading profit |
1,123 | 22.9% | 1,048 | 22.0% |
DIVIDENDS
The 2017 final dividend of 22.7 US cents per ordinary share totalling $198 million was paid on 9 May 2018. The 2018 interim dividend of 14.0 US cents per ordinary share totalling $123 million was paid on 31 October 2018.
RETURN ON INVESTED CAPITAL
Return On Invested Capital1 (ROIC) is a measure of the return generated on capital invested by the Group. It provides a metric for long-term value creation and encourages compounding reinvestment within the business and discipline around acquisitions with low returns and long payback. ROIC decreased from 14.3% in 2017 to 12.5% in 2018 as a result of the reduction in operating profit.
ROIC is defined as:
|
Net Operating Profit less Adjusted Taxes |
|
|
|
(Opening Net Operating Assets + |
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
39 |
BALANCE SHEET
|
|
|
|
|
|
|
2018 $ million |
2017 $ million |
Change $ million |
|
Consolidated balance sheet |
|
|
|
|
Goodwill and intangible assets |
3,547 | 3,742 | (195) |
|
Other non-current assets |
1,435 | 1,393 | 42 |
|
Current assets |
3,077 | 2,731 | 346 |
|
Total assets |
8,059 | 7,866 | 193 |
|
Total equity |
4,874 | 4,644 | 230 |
|
Non-current liabilities |
1,720 | 1,876 | (156) |
|
Current liabilities |
1,465 | 1,346 | 119 |
|
Total liabilities |
3,185 | 3,222 | (37) |
|
Total liabilities and equity |
8,059 | 7,866 | 193 |
|
Net debt3 |
1,104 | 1,281 | (177) |
|
|
|
|
|
Goodwill decreased by $34 million primarily as a result of foreign currency movements. Intangible assets decreased by $161 million with amortisation and impairment of $179 million and foreign currency movements of $14 million partially offset by net additions of $32 million.
Other non-current assets increased by $42 million primarily due to an increase of $30 million in the retirement benefit assets for our UK and US pension schemes. Trade investments also increased by $13 million as a result of additions of $4 million and fair value remeasurements of $9 million. Current assets increased by $346 million with trade and other receivables increasing $59 million as a result of the timing of collections, inventories increasing $91 million primarily due to sales growth and new product build, and cash increasing $196 million due to insurance recoveries and stronger underlying profits.
Non-current liabilities decreased by $156 million primarily due to the reclassification of certain payables and borrowings to current liabilities, as they now fall due within one year. Current liabilities increased by $119 million as a result of the aforementioned reclassifications from non-current to current liabilities and an increase of $18 million in bank overdrafts.
CASH FLOW STATEMENT
|
2018 $ million |
2017 $ million |
Change $ million |
|
|
Consolidated cash flow statement |
|||
|
Cash generated from operations |
1,108 | 1,273 | (165) |
|
Trading cash flow1 |
951 | 940 | 11 |
|
Free cash flow1 |
584 | 714 | (130) |
Cash generated from operations of $1,108 million is after paying out $3 million of acquisition and disposal related items, $83 million of restructuring and rationalisation expenses and $104 million relating to legal and other costs.
Trading cash flow1 increased by $11 million driven by higher trading profit and lower capital expenditure. These movements were partly offset by a working capital net outflow which included an increase in inventory as described above. Free cash flow1 decreased by $130 million primarily related to higher cash outflows for restructuring and rationalisation expenses and legal and other costs.
During the year ended 31 December 2018, the Group purchased a total of 2.7 million (2017: 3.2 million) ordinary shares at a cost of $48 million (2017: $52 million) as part of the ongoing programme to buy back an equivalent number of shares to those vesting as part of the employee share plans.
LIQUIDITY AND CAPITAL RESOURCES
The Group’s policy is to ensure that it has sufficient funding and facilities in place to meet foreseeable borrowing requirements.
The Group’s net debt3 decreased from $1,281 million at the beginning of 2018 to $1,104 million at the end of 2018, representing an overall decrease of $177 million.
At 31 December 2018, the Group held $333 million (2017: $155 million) in cash net of bank overdrafts. The Group had committed facilities available of $2,429 million at 31 December 2018 of which $1,429 million was drawn. Smith & Nephew intends to repay the $125 million of facilities due within one year by using available cash and drawing down on the longer-term facilities.
The principal variations in the Group’s borrowing requirements result from the timing of dividend payments, acquisitions and disposals of businesses, timing of capital expenditure and working capital fluctuations. Smith & Nephew believes that its capital expenditure needs and its working capital funding for 2019, as well as its other known or expected commitments or liabilities, can be met from its existing resources and facilities.
The Group’s planned future contributions are considered adequate to cover the current underfunded position in the Group’s defined benefit plans.
1These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198.
2Included within the 2017 analysis is a reclassification of $20 million of revenue formerly included in Other Established Markets which has now been included in Emerging Markets in order to present consistent analysis to the 2018 results.
3Net debt is reconciled in Note 15 to the Group accounts.
|
40 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
Our risk management process
Our approach to risk
Like all businesses, we face a number of risks and uncertainties.
Some come from outside our organisation, others from within. Some we can’t control, some we can. Many of our risks are similar to those experienced by similar businesses and for 2018 we are undoubtedly aligned with other businesses as risks around Brexit uncertainty and political unrest globally have heightened.
Successful management of existing and emerging risks is critical to the long‑term success of our business and to the achievement of our strategic objectives. In order to seize market opportunities and leverage the potential for success, risk must be accepted to a reasonable degree within our tolerances. Risk management is therefore an integral component of the Group’s Corporate Governance.
As in previous years our Enterprise Risk Management process is based on a holistic approach to risk management, leveraging risk identification and risk treatments already in place throughout our Business Areas whilst incorporating the same risk processes into the strategic planning process. Our belief is that the strategic and operational benefits of managing risk are achieved when Enterprise Risk Management is aligned with the strategic and operational goals of the organisation, and our process and governance structure firmly aligns to this approach.
The current financial year has seen further maturity of the risk management framework with further testing of key controls through ‘deep dives’ by the Group Risk Team and a number of different risk topics presented to the Board and its Committees. We have also designed our Enterprise Risk Management Framework to be fully integrated into our business processes. This will be built on further throughout 2019.
Our risk governance framework is set out below. At the very top of our structure is our Board, setting our risk appetite and monitoring the application of our risk framework through strategy, execution and practically through the outputs of regular risk ‘deep dives’ and reviews by the business and Group Risk Team. The Board cascades our risk appetite throughout our organisation through the Executive Committee, risk owner community and our management group with a formal ‘bottom up’ process ensuring that risks are escalated back through the process to our Board and form our Principal Risks as appropriate. Providing rigour and independence across this process is our Executive Committee and the Group Risk Team.
At the third line of defence is our Internal Audit Function, providing an annual opinion on the effectiveness of our Risk Management process to the Executive Committee, chaired by the Chief Executive Officer, and then to the Board and its Committees.
BOARD OF DIRECTORS AND BOARD COMMITTEES
- Responsible for regular oversight of risk management and for our annual strategic risk review
- Monitors risks through Board processes (Strategy Review, Disclosures, M&A, Investments, Disposals) and Committees (Audit and Compliance & Culture), management reviews and ‘deep dives’ of selected risk areas
- The Audit Committee is responsible for ensuring oversight of the process by which risks relating to the Company and its operations are managed and for reviewing the operating effectiveness of the Group’s Risk Management process
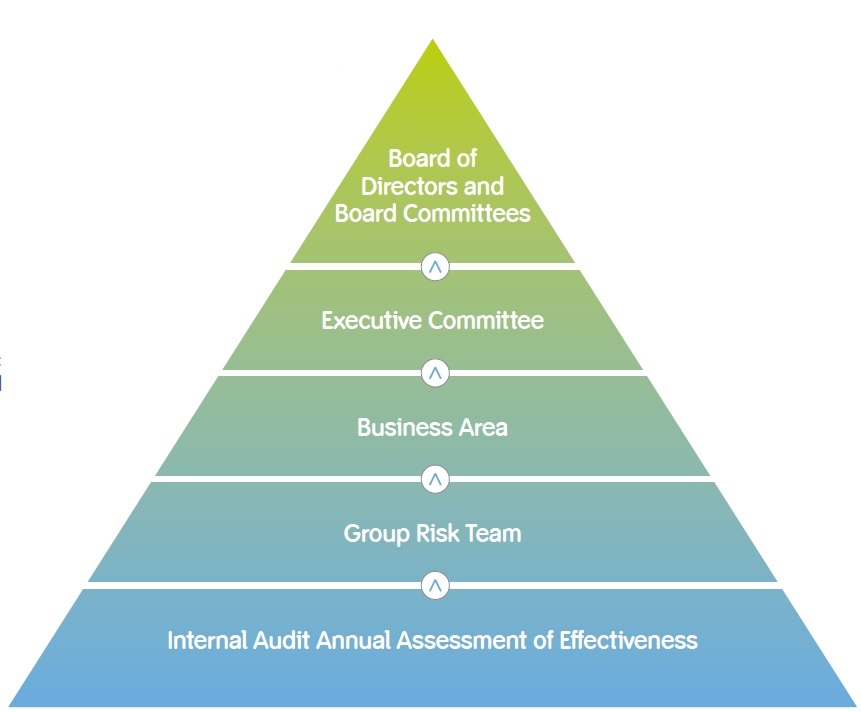
|
|
Smith & Nephew Annual Report 2018 |
|
41 |
EXECUTIVE COMMITTEE SITTING AS GROUP RISK COMMITTEE
Reviews external/internal environment for emerging risks
Reviews risk register updates from Business Area Risk Groups
Identifies significant risks and assesses effectiveness of mitigating actions
BUSINESS AREA
Business Area Risk Champions provide support to ensure a framework is designed and implemented for alignment to the requirements of the Enterprise Risk Management Framework
Carry out day-to-day risk management activities
Identify and assess risk
Implement strategy and mitigating actions to treat risk within Business Area
Business Area Risk Champions lead regular risk register updates
GROUP RISK TEAM
Manages implementation of all aspects of the Group’s approach to Enterprise Risk Management including implementation of processes, tools and systems to identify, assess, measure, manage, monitor and report risks
Facilitates implementation and coordination through Business Area Risk Champions
Provides resources and training to support process
Regular risk reporting to the Executive Committee
Prepares Board and Group Risk Committee reports
ANNUAL ASSESSMENT OF EFFECTIVENESS – INTERNAL AUDIT AND CONTROL FUNCTIONS
Provides independent assurance to the Board and Audit Committee on the effectiveness of the Group’s Risk Management process
|
Risk management life cycle |
|
|
Our risk management life cycle was fully refreshed in 2017 and was updated in 2018 to align with our new strategic imperatives which we launched in December. |
|
|
Our Risk Management Policy, sponsored by our Chief Executive Officer, is supported by an Enterprise Risk Management Manual and the risk team provide regular training to all risk champions throughout the year. As in 2018 risks continue to be managed through a ‘bottom up’ and ‘top down’ process, with regular oversight from the Executive Committee and quarterly reports to the Board Committees. An overview of our risk management life cycle can be found below: |
|
|
1 Risk identification IDENTIFYING risks associated with the achievement of our objectives by function at the Group level. 2 Gross (inherent) risk assessment ASSESSING the level of inherent (gross) risk. 3 Current control identification IDENTIFYING existing controls to mitigate risks 4 Net (residual) risk ASSESSING the level of residual (net) risk after mitigation so that risk levels are managed within defined tolerance thresholds without being over-controlled or foregoing desirable opportunities. 5 Risk response planning IDENTIFYING additional actions required to meet our expected risk tolerance level and ASSIGNING risk owners, timeframes and actions for ongoing management and reporting. 6 Risk reporting REPORTING the status of our most significant risks through the ‘bottom up’ business area processes and the ‘top down’ Executive Committee and Board process. 7 Monitoring and review MONITORING of risks and actions by management, the accountable Executive and Board. |
|
|
42 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
RISK REPORT continued
2018 principal risks
We assess our Principal Risks in terms of their potential impact on our ability to deliver our Strategic Imperatives
These links are highlighted across the following pages and further information on the Strategic Imperatives is found on page 9.
|
BUSINESS CONTINUITY AND BUSINESS CHANGE |
|
|
|||||||
|
|
|||||||||
|
Operating with a global remit, increased outsourcing, more sophisticated materials and the speed of technological change in an already complex manufacturing process leads to greater potential for disruptive events. Ensuring our ability to continually execute and operate key sites and facilities in order to develop, manufacture and sell our products within this environment is a key strategic imperative of the organisation. In addition, the pace and scope of our business ‘change’ initiatives increases the execution risk that benefits may not be fully realised, costs of these changes may increase, or that our business as usual activities may not perform in-line with our plans. |
|
|
Examples of risks Failure or significant performance issues experienced at critical/single source facilities. Disruption to manufacturing at single or sole source facility (lack of manufacturing redundancy). Supplier failure impacts ability to meet customer demand (single source suppliers). Natural disaster impacts ability to meet customer demand. Significant ‘change’ prevents our projects and programmes such as APEX achieving the intended benefits and disrupts existing business activities. |
|
|
Risk Tolerance In operating our business, executing our change programmes and in managing our suppliers and facilities we have a low to moderate tolerance for this risk. |
|||
|
|
Change from 2017 No change. |
|
|||||||
|
|
Actions taken by management Comprehensive product quality processes and controls are in place from design to customer supply. Emergency and incident management and business recovery plans are in place at major facilities and for key products and key suppliers. Undertaking risk based review programmes for critical suppliers. Project management governance and toolkits and project steering committee oversight to support successful execution of programme and projects. Executive Committee and Audit Committee oversight of Risks to change programmes. Brexit Steering Group regularly monitors the evolving impact of Brexit and oversees our response. |
|
|
Link to strategy Our Strategic Imperative to ‘Become the best owner’ requires us to ensure we remain sustainable into the future and through periods of business change. |
|||||
|
|
Oversight Board |
||||||||
|
|
Smith & Nephew Annual Report 2018 |
|
43 |
|
CYBER SECURITY |
|
|
|||||||
|
|
|||||||||
|
High profile incidents coupled with increasing government focus has resulted in raised awareness of the extent and potential impact of cyber security breaches. Our increasing business dependence on networked systems and the internet, the design of new products, connectable products and embedded software and the rapidly evolving cyber security threat landscape provides a level of risk exposure not experienced in prior years. In response to this we have undertaken an exercise to understand our threats and vulnerabilities to target cyber security investment in the right places. |
|
|
Examples of risks Loss of intellectual property/major data privacy breach or significant impact on business operations from Malware or Ransomware outbreak. Cyber security is not considered in the design of new products with more products being connectable/having embedded software. |
|
|
Risk Tolerance In managing our cyber risk and the possible disruption and reputational impact we have a low to moderate tolerance for Cyber Security Risk. |
|||
|
Change from 2017 No change. |
|||||||||
|
|
Actions taken by management Security information and event management (SIEM) in place provides real-time analysis of security alerts generated by applications and network hardware. Annual penetration testing and quarterly vulnerability testing. Endpoint protection and Intrusion detection/prevention. The adoption of additional authentication tools to reduce the likelihood of remote attacks. Annual mandatory training and continuous awareness training for end-users. Security governance structure in place including a Cyber Security Steering Committee. Further strengthening governance including a programme to monitor cyber security capabilities and controls, technical and governance matters. |
|
|
Link to strategy Our Strategic Imperative to ‘Transform the business through enabling technologies’ requires us to deliver technology solutions in compliance with laws and regulations and in a way that protects any vulnerability to Cyber Risk. |
|||||
|
|
Oversight Audit Committee |
||||||||
|
QUALITY AND REGULATORY |
|
|
|||||||
|
|
|||||||||
|
Global regulatory bodies continue to increase their expectations of manufacturers and distributors of medical devices. Our products are used in the human body and therefore patient safety is of paramount importance. The European Medical Device Regulations, launch of ISO13485‑2016, the Medical Device Single Audit Programme and the tightening of the Chinese YY standards have increased the focus on clinical and technical evidence, supplier controls and continual product risk reduction. 2018 has also brought uncertainty in terms of Britain’s exit from the EU as well as future trade and regulatory relations between the EU and UK. Like many other companies we are planning for the impact of a range of eventualities, particularly in continuity assessment and how our products will continue to be appropriately registered for trade around the EU. |
|
|
Examples of risks Defects in design or manufacturing of products supplied to, and sold by, the Group could lead to product recalls or product removal or result in loss of life or major injury. Significant non-compliance with policy, regulations or standards governing products and operations regarding registration, manufacturing, distribution, sales or marketing. Failure to obtain proper approvals for new or changed technologies, products or processes. CE certificates issued by UK notified bodies prior to the withdrawal date may be rendered void post Brexit. |
|
|
Risk Tolerance Our response to this risk continues to be critical and our ability to align the standards required to ensure safe and compliant products is the key driver for our extremely low tolerance for risk in this area. |
|||
|
Change from 2017 No change. |
|||||||||
|
Actions taken by management Comprehensive and documented product quality processes and controls from design to customer distribution are in place. Standardised monitoring and compliance with quality management practices through our Global Quality Assurance and Regulatory Affairs organisation. Incident management teams in place to respond immediately in the event of an incident relating to patient safety. Governance framework in place for reporting, investigating and responding to instances of product safety and complaints. Brexit working group is in place considering various deal/no deal scenarios. |
Link to strategy Our Strategic Imperative to ‘Become the best owner’ requires us to operate effectively and efficiently and to produce compliant products of the highest quality to provide safe and effective solutions to our customers. |
||||||||
|
|
Oversight Board Compliance & Culture Committee |
||||||||
|
44 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
RISK REPORT continued
|
NEW PRODUCT INNOVATION, DESIGN & DEVELOPMENT INCLUDING INTELLECTUAL PROPERTY |
|||||||
|
|
|||||||
|
Our product portfolio is becoming increasingly complex, especially as we move to more innovative connected product technologies. Our success relies on investing in safe products and platforms, aligned internal and external design, and development innovation in order to compete effectively. The need to be considered in our approach to protecting our products, process and intellectual property is essential. |
|
|
Examples of risks Failure to develop an appropriate pipeline of commercially successful products to meet and anticipate the needs of our customers ahead of the competition. Insufficient long-term planning to respond to competitor disruptive entries into marketspace. Inadequate innovation due to low Research & Development (R&D) investment, R&D skills gap or poor product development execution. Lower value business segment investment, such as product maintenance and line extension projects. Competitors may assert patents or other intellectual property rights against the Company, or fail to respect the Company’s intellectual property rights. |
|
|
Risk Tolerance In pursuit of our strategy to be innovative in our product offering we have a moderate to high tolerance for risk. |
|
|
Change from 2017 Reduced risk. |
|||||||
|
|
Actions taken by management Global R&D organisation and governance framework providing strategic direction for allocation of R&D investments across all businesses. Clear stage-gate process to continually evaluate R&D investments decisions and development of new products. Enhanced relationship with Commercial team to focus on developing products that customers need. Strengthened Clinical Affairs programme integrated with Global Marketing. Cross functional New Product Design and R&D processes focused on identifying new products and potentially disruptive technologies and solutions. Monitoring of external market trends and collation of customer insights to develop product strategies. Careful attention to intellectual property considerations. |
|
|
Link to strategy Our Strategic Imperative to ‘Transform the business through enabling technologies’ depends heavily on our ability to continue to develop new innovative products and bring them to market. |
|||
|
|
Oversight Board |
||||||
|
TALENT MANAGEMENT |
|||||||
|
|
|||||||
|
We recognise that people management, effective succession planning and the ability to attract and retain talent is of great importance to the success of our Company. In the current economic environment of strong competition and reduced spending, retention of top talent is a critical risk which requires a strong process in relation to retention and engagement. Failure to do so can result in risks in our ability to execute Company strategy and achieve business objectives in relevant functions and to be effective in the chosen market/discipline and leadership of newer workforce which may impact the Company’s future success. |
|
|
Examples of risks Loss of key talent, high attrition and lack of appropriate succession planning in context of required skillsets for future business needs. Loss of competitive advantage due to an inability to attract and retain Top Talent. Loss of intellectual capital due to poor retention of talent. |
|
|
Risk Tolerance We have a moderate tolerance for this risk. |
|
|
|
Change from 2017 No change. |
|
|||||
|
Actions taken by management Talent planning and people development processes are well established across the Group. Talent and succession planning is discussed annually by the Board and regularly by the Executive Committee and Nomination & Governance Committee. Identification of high performing individuals and practices to plan for the succession of key roles. Consistent and robust performance management process. Development of strategic skills resourcing plan by functional areas. |
Link to strategy All our strategic imperatives rely on ensuring we have the right talent within our organisation to deliver maximum efficiency in everything we do and to build strong leaders for the future. |
||||||
|
|
Oversight Board |
||||||
|
|
Smith & Nephew Annual Report 2018 |
|
45 |
|
PRICING AND REIMBURSEMENT |
|||||||
|
|
|||||||
|
Our success depends on our ability to sell our products profitably in spite of increasing pricing pressures from customers, and governments providing adequate funding to meet increasing demands arising from demographic trends. The prices we charge are therefore impacted by budgetary constraints and our ability to persuade customers and governments of the economic value of our products, based on clinical data, cost, patient outcomes and comparative effectiveness. We further face challenging market dynamics, such as consolidation of customers into buying groups, increasing professionalisation of procurement departments and the commoditisation of entire product groups, which continue to challenge prices. |
|
|
Examples of risks Reduced reimbursement levels and increasing pricing pressures. Systemic challenge on number of elective procedures. Lack of compelling health economics data to support reimbursement requests. Risk of adverse trading margins due to fluctuating foreign currency exchange rates across our main manufacturing countries (US, UK, Costa Rica and China) and where our products are sold. |
|
|
Risk Tolerance In implementing innovative pricing strategies, we have a moderate to high tolerance for risk and are willing to accept certain risks in pursuit of new business opportunities. |
|
|
|
Change from 2017 No change. |
|
|||||
|
Actions taken by management Development of innovative economic product and service solutions for both Established and Emerging Markets. Appropriate breadth of portfolio and geographic spread to mitigate exposure to localised risks. Incorporating health economic components into the design and development of new products. Emphasising value propositions tailored to specific stakeholders and geographies through strategic investment and marketing programmes. |
Link to strategy Our Strategic Imperative to ‘Achieve the full potential of our portfolio’ depends on our ability to sell our products profitably in spite of increased pricing pressures from payers. |
||||||
|
|
Oversight Board |
||||||
|
MERGERS AND ACQUISITIONS |
|||||||
|
|
|||||||
|
As the Company grows to meet the needs of our customers and patients, we recognise that we are not able to develop all the products and services required using internal resources and therefore need to undertake mergers and acquisitions in order to expand our offering and to complement our existing business. In other areas, we may divest businesses which are no longer core to our activities. It is crucial for our long-term success that we make the right choices around acquisitions and divestments. We have a well‑defined cross-functional process for managing risks associated with mergers and acquisitions that is subject to scrutiny from executive management and the Board of Directors. |
|
|
Examples of risks Failure to identify appropriate acquisitions or to conduct effective acquisition due diligence. Failure to integrate newly acquired businesses effectively, including integration with Company standards, policies and financial controls. |
|
|
Risk Tolerance In acquiring new businesses and business models, we have a moderate to high tolerance for commercial risk and are willing to accept certain risks in pursuit of new business. |
|
|
|
Change from 2017 No change. |
|
|||||
|
Actions taken by management Acquisition activity is aligned with corporate strategy and prioritised towards products, franchises and markets identified to have the greatest long-term potential. Clearly defined investment appraisal process based on return on capital, in accordance with Capital Allocation Framework and comprehensive post-acquisition review programme. Undertaking detailed and comprehensive cross-functional due diligence prior to acquisitions. Compliance risks included as part of due diligence reviews, integration plans and reporting for acquisitions. |
Link to strategy Our Strategic Imperatives to ‘Expand in high-growth segments’ and ‘Transform the business through enabling technologies’ depend on our ability to identify the right acquisitions, to conduct thorough due diligence and to integrate acquisitions effectively. |
||||||
|
|
Oversight Board |
||||||
|
46 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
RISK REPORT continued
|
LEGAL AND COMPLIANCE RISKS |
|||||||
|
|
|||||||
|
Our global remit results in heavy regulation across multiple jurisdictions. There is increasing public scrutiny of ethics in business and ‘doing the right thing’ is part of our licence to operate. National regulatory authorities enforce a complex pattern of laws and regulations that govern the design, development, approval, manufacture, labelling, marketing and sale of healthcare products. Operating across this increasingly complex and dynamic legal and compliance environment, including regulations on bribery and corruption, with poor legal and compliance practices can lead to fines, penalties, reputational risk and competitive disadvantage. We have adopted a proactive, holistic approach, which guides the Company towards a culture of compliance and turns the resolution of legal and compliance issues into a source of competitive advantage. |
|
|
Examples of risks Failure to act in an ethical manner consistent with our Code of Conduct. Violation of anti-corruption or healthcare laws, breach by employee or third party representative. Misuse or loss of personal information of patients, employees, research subjects, consumers or customers results in violations of data privacy laws, including General Data Protection Regulations. |
|
|
Risk Tolerance In complying with legal and compliance requirements, we have an extremely low tolerance. |
|
|
|
Change from 2017 No change. |
|
|||||
|
Actions taken by management Ethics & Compliance Committee oversees our ethical and compliance practices. Global compliance programme, policies and procedures. All employees are required to undertake annual training and to certify compliance on an annual basis with our Code of Conduct and Business Principles. Group monitoring and auditing programmes in place. Confidential independent reporting channels for employees and third parties to report concerns. |
Link to strategy Our Strategic Imperative to ‘Become the best owner’ requires us to comply with applicable laws and regulations and do the right thing as part of our licence to operate. |
||||||
|
|
Oversight Board Compliance & Culture Committee |
||||||
|
COMMERCIAL EXECUTION |
|||||||
|
|
|||||||
|
We continue to make good and strong progress delivering our priorities and are proud of the pace with which our strategic and operational decisions are quickly translated into actions. Effective communication and engagement with our customers are critical to the long-term success of our business. We are confident that we have the right priorities, structures and capabilities across the Group and we acknowledge that only strong and continued execution will keep us ahead of our competitors and best placed to serve our customers. Failure to execute our priorities will impact our ability to continue to grow our business and serve our customers. |
|
|
Examples of risks Failure to execute our strategy adequately from high‑level ambition to specific actions to make the ambition a reality. Inability to keep pace with significant product innovation and technical advances to develop commercially viable products. Failure to adapt our priorities and execution appropriately when conditions change meaning that transformational programmes do not deliver the expected outcomes. Failure to engage effectively with our key stakeholders to meet their evolving needs leading to loss of customers. |
|
|
Risk Tolerance We have a low to moderate tolerance level for commercial execution risk. |
|
|
|
Change from 2017 No change. |
|
|||||
|
Actions taken by management Global R&D organisation and supporting governance framework. Improved market development and launch execution – commitment to win profitably in our target markets. Strategic planning process clearly linked to business and Group Risk. Global transformational programmes in place providing agile opportunities for efficiencies, growth and a strengthened competitive position. |
Link to strategy Our Strategic Imperatives to ‘Achieve the full potential of our portfolio’, ‘Transform the business through enabling technologies’ and ‘Expand in high-growth segments’ requires excellent commercial execution. |
||||||
|
|
Oversight Board |
||||||
|
|
Smith & Nephew Annual Report 2018 |
|
47 |
|
POLITICAL AND ECONOMIC |
|||||||
|
|
|||||||
|
Across our business we are exposed to the effects of political and economic risks from the impact of Brexit to changes in the regulatory and competitive landscape to the impact of the US Administration’s changed approach to Trade Policy. Turning to Brexit, there remain heightened levels of political and regulatory uncertainty in the UK following the referendum vote to leave the EU in June 2016, the triggering of Article 50 in March 2017 and the general election in June 2017. This uncertainty is expected to continue for the foreseeable future until EU exit negotiations have been completed and alternative trade deals have been put in place. This situation may adversely impact trading performance across the sector. Regulatory risk forms the most significant risk presently; the ability for us to continue to manufacture and register our products in a compliant manner for global distribution is key. |
|
|
Examples of risks Macro-economic uncertainty or downturn in the UK economy as a result of Brexit. Regulatory risk whereby certificates issued by UK Notified Bodies are no longer recognised in EU following March 2019. Global political uncertainty – regarding trade policy. Implementation of healthcare reforms and/or protectionist measures and regulation or legislation in local markets. Exchange rate volatility. The availability of markets and market access rights. Impact on strategy and operations. Increases in import and labour costs. Retention of skilled labour and recruitment concerns. Increases in tariffs and restrictions on global trade. |
|
|
Risk Tolerance In preparing for Brexit and managing risk arising from changes to our political economical environment, we have a low to moderate tolerance. |
|
|
|
Change from 2017
New risk previously incorporated into
|
|
|||||
|
Actions taken by management Continued engagement with governments, administrations and regulatory bodies. The Group has a Brexit committee which meets regularly and is split into a number different work streams namely: Regulatory – the most complex work stream considering product registration. Supply chain – considering the impacts on import and export requirements in UK and Europe; supply routes, managing inventory levels in the UK and Europe to minimise disruption from border clearance and managing labelling changes required as a result of the regulatory changes. UK and Ireland (‘UKI’) market – considering issues expected to impact the UKI specifically the Irish border issue – including whether a changed route to market is required in Ireland. HR – considering issues such as impact on EU workers currently employed in UK and future mobility considerations. Finance – considering issues such as impact on tax including EU trading arrangements and the VAT implications of the changes and the impact on financial reporting, eg EU/UK endorsement of IFRS. Improved engagement and monitoring/lobbying on localisation initiatives. |
Link to strategy Our Strategic Imperative to ‘Become the best owner’ requires us to operate effectively within different global political situations, which change constantly. |
||||||
|
|
Oversight Board |
||||||
|
48 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
RISK REPORT continued
‘Deep Dives’ and reviews completed in the year
During the year, the risks identified through the ‘bottom up’ and ‘top down’ processes were mapped against each other with the most significant risks forming our Principal Risks.
These risks and our tolerance levels were discussed with each member of the Executive Committee separately and collectively in September and were presented to the Board during the Strategy Review in December 2018. In December, the Accountable Executives were further required to validate that the risk profile had not significantly changed since the initial exercise in June. No changes were required to our risk profile as a result of this exercise, which was also formally validated by each Accountable Executive.
Throughout 2018, the Board assessed and monitored risk in a number of areas and specific ‘Deep Dive’ reviews were also completed by the Group Risk team. In addition, in early 2019, the Board reviewed the results of a deep dive on Brexit risk. The 2018 risk reviews and deep dives included:
|
|
BOARD AND AUDIT COMMITTEE ‘DEEP DIVES’ AND REVIEWS |
|||||
|
|
LEGAL, COMPLIANCE |
|
RESEARCH AND |
|
FUNCTIONAL OVERSIGHT |
|
|
|
During the year, the Ethics & Compliance |
|
During the year, the Board received a report from the R&D function which included a focus on the risks associated with the R&D programme and strategies to manage these risks. |
|
The Board and Audit Committee receive regular updates throughout the year from functions such as IT, Tax, Treasury and Financial Operations. The Audit Committee also receives an update three times a year on the progress of the risk management programme. |
|
|
|
MANUFACTURING |
|
HR |
|
M&A |
|
|
|
During the year, the Board has received a number of presentations from the global operations team considering the risks in particular associated with the manufacturing footprint and the supply chain and proposals to mitigate these risks. |
|
The Board has reviewed and approved all Executive Officer changes during the year within the context of the overall strategy and have been kept updated on management actions to implement the new culture pillars. |
|
Each Board meeting considers Corporate Development within the context of the Group Strategy and also reviews specific acquisitions. During the year, the Board also undertook retrospective reviews of previous acquisitions compared to expectations in the original deal models. |
|
|
|
IT/CYBER |
|
|
|||
|
|
The Audit Committee received reports on IT and cyber security including an assessment of the existing risks and benchmarking against industry standards. |
|
|
|||
|
|
|
|
|
|
|
|
|
|
GROUP RISK TEAM ’DEEP DIVES’ |
|||||
|
|
|
|
|
|
|
|
|
|
A series of planned ‘Deep Dives’ have been completed in the year across our Business Risk Areas, including Global Manufacturing, Strategic Sourcing, Supply Chain, Global Quality and Regulatory, Global Business Services and IT. These reviews were introduced in 2017 to supplement reports provided to the Board and primarily cover an ‘independent’ assessment of compliance to the expected Risk Management Framework and in particular the adequacy of stated mitigating activities. The results are reported through the Risk Champions and Accountable Executives to the Audit Committee and are tracked and monitored to resolution by the Group Risk Team. |
|||||
|
|
|
|||||
|
|
Smith & Nephew Annual Report 2018 |
|
49 |
|
2019 Risk Management Plan |
||||||||||
|
|
|
|
||||||||
|
Our work will continue to evolve in 2019 and we will strengthen our approach to managing risks across the organisation, including our business areas and product groups. |
|
We will continue to ensure a truly collaborative approach to risk management with risk accountability sitting squarely with management and a proactive Group Risk function influencing decision making through effective challenge and timely consultation. |
||||||||
|
|
|
|
||||||||
|
2019 RISK MANAGEMENT TIMELINE |
||||||||||
|
Q1 2019 |
|
Q2 2019 |
|
Q3 2019 |
|
Q4 2019 |
|
Q1 2020 |
||
|
INTERNAL AUDIT |
|
|
|
|
|
|||||
|
Risk Management Effectiveness Review report to Audit Committee |
|
|
|
2020 Risk Based Internal Audit Plan Preparation |
|
|
|
Risk Management Effectiveness Review report to Audit Committee |
||
|
|
|
|
|
|
|
|
|
|
||
|
GROUP RISK TEAM |
||||||||||
|
Refresh Enterprise Risk Management Policy and Manual |
Risk training Report to Audit Committee Facilitate ‘top down’ review process |
Prepare 2020 Enterprise Risk Management Strategy Prepare Review of Principal Risks Report to Audit Committee |
Refresh Enterprise Risk Management Policy and process |
|||||||
|
|
|
|
|
|
|
|
|
|
||
|
BUSINESS RISK AREAS |
||||||||||
|
Quarterly Risk Review by leadership teams |
|
Quarterly Risk Review by leadership teams Review Principal Risks and map to Strategic Imperatives |
|
Quarterly Risk Review by leadership teams Risk Register refresh and submission to Group Risk Team annual certification |
|
Quarterly Risk Review by leadership teams |
|
Quarterly Risk Review by leadership teams |
||
|
|
|
|
|
|
|
|
|
|
||
|
EXECUTIVE COMMITTEE |
||||||||||
|
Review reports from Group Risk Team |
|
Review reports from Group Risk Team |
|
‘Top Down’ review of Principal Risks |
|
Approve Principal Risks |
|
Review reports from Group Risk Team |
||
|
|
|
|
|
|
|
|
|
|
||
|
AUDIT COMMITTEE |
||||||||||
|
Review and approval of the Group’s 2018 Risk Management Process and Viability Statement |
|
Receive report from the Group Risk Team and review Enterprise Risk Management process |
|
Receive report from the Group Risk Team and review Enterprise Risk Management process |
|
Review and approve Principal Risks |
|
Review and approval of the Group’s 2019 Risk Management Process and Viability Statement |
||
|
|
|
|
|
|
|
|
|
|
||
|
BOARD |
||||||||||
|
Approve Principal Risks |
|
|
|
Review of Principal Risks |
|
|
|
Approve Principal Risks |
||
|
50 |
Strategic report |
Smith & Nephew Annual Report 2018 |
|
RISK REPORT continued
HOW WE ASSESS OUR PROSPECTS
During the year, the Board has carried out a robust assessment of the Principal Risks affecting the Company, particularly those which could threaten the business model. These risks and the actions being taken to manage or mitigate them are explained in detail on pages 41–49 of this Annual Report.
In reaching our Viability Statement conclusion, we have undertaken the following process:
– The Audit Committee reviewed the Risk Management process at their meetings in February, July, September and December, receiving presentations from the Group Risk function, explaining the processes followed by management in identifying and managing risk throughout the business.
– In October, a series of detailed one-to-one discussions were held with each member of the Executive Committee and the Group Risk Team. In these discussions, the Executives were asked to consider the significant risks which they believed could seriously impact the profitability and future prospects of the Company and the principal risks that would threaten its business model, future performance, solvency or liquidity.
– As part of the strategy business updates in September, the Board considered and discussed the Principal Risks which could impact the business model over the next three years and discussed with the management team how these risks were being managed and mitigated.
– Throughout the year, a number of ‘deep dives’ and reviews into different risks were conducted by the Board, the Audit Committee and the Ethics & Compliance Committee looking into the nature of the risks and how they were mitigated, as detailed on page 48 of this Annual Report.
– Throughout the year, a number of ‘deep dives’ into specific risk areas were conducted by the Group Risk Team, the results of which were presented to and discussed by the Audit Committee and are detailed on page 48 of this Annual Report.
ASSESSMENT PERIOD
The Board have determined that the three-year period to December 2021 is an appropriate period over which to provide its Viability Statement. This period is aligned to the Group’s Strategic Planning process and reflects the Board’s best estimate of the future viability of the business.
SCENARIO TESTING
For the purpose of testing the viability of the Company, we have undertaken a robust scenario assessment of the principal risks and some other risks, which could threaten the viability or existence of the Company. These have been modelled as follows:
– In carrying out scenario modelling of the principal and significant risks on the following page we have also evaluated the impact of a severe but plausible combination of these risks actually occurring over the three-year period. We have considered and discussed a report setting out the terms of our current financing arrangements and potential capacity for additional financing should this be required in the event of one of the scenarios modelled occurring.
– We are satisfied that we have robust mitigating actions in place as detailed on pages 42–47 of this Annual Report. We recognise, however, that the long-term viability of the Company could also be impacted by other, as yet unforeseen, risks or that the mitigating actions we have put in place could turn out to be less effective than intended.
|
|
Smith & Nephew Annual Report 2018 |
|
51 |
2018 SCENARIOS MODELLED
|
SCENARIO 1 – PRICING AND REIMBURSEMENT PRESSURES |
|
|
Pricing and reimbursement pressures or currency exchange volatility (Principal Risk) – leading to a major loss of revenues and profits. |
Action taken: We have modelled a 1% annual price erosion from 2019. |
|
Link to strategy – Achieve the full potential of our portfolio |
Link to Principal Risks – Pricing and Reimbursement |
|
|
|
|
SCENARIO 2 – OPERATIONAL RISK |
|
|
Execution risk – our inability to launch new products losing significant market share to the competition. |
Action taken: We have modelled a universal 1% reduction of annual volume growth from 2019. |
|
Product liability claims – giving rise to significant claims and legal fees. |
Action taken: We have modelled a one-off significant product liability claim in 2020, without any insurance coverage. |
|
Temporary loss of key production capability – resulting in our inability to manufacture a key product for a period of time. |
Action taken: We have modelled the loss of a factory, resulting in the loss of production and sales of a key product for two years from 2020. |
|
Link to strategy – Become the best owner – Transform the business through enabling technologies – Achieve the full potential of our portfolio |
Link to Principal Risks – New Product Innovation, Design & Development Including Intellectual Property – Commercial Execution – Business Continuity and Business Change |
|
|
|
|
SCENARIO 3 – LEGAL REGULATORY AND COMPLIANCE RISKS |
|
|
Regulatory measures – impacting our ability to continue to sell key products. |
Action taken: We have modelled the complete loss of revenue from a key product for each year from 2019. |
|
Bribery and corruption claims – giving rise to significant fines. |
Action taken: We have assumed a one-off significant fine in 2020. |
|
Link to strategy – Become the best owner |
Link to Principal Risks – Legal and Compliance – Quality and Regulatory |
|
|
|
|
SCENARIO 4 – CYBER SECURITY |
|
|
Inability to issue invoices or collect money for a period of time. |
Action taken: We have modelled one of our key regions being unable to invoice for a month in 2020 due to an IT disruption. |
|
Link to strategy – Transform the business through enabling technologies |
Link to Principal Risks – Cyber Security |
|
|
|
|
OTHER |
|
|
Political and economic risk – for example, political upheaval, which could cause us to withdraw from a major market for a period of time. |
Action taken: We have modelled a major disruption due to a hard Brexit having a regulatory impact, and also causing severe delays with imports and exports for three months. |
|
Link to strategy – Become the best owner |
Link to Principal Risks – Political and Economic |
VIABILITY STATEMENT
Having assessed the principal risks, the Board has determined that we have a reasonable expectation that the Company will be able to continue in operation and meet its liabilities as they fall due over a period of three years from 1 January 2019. In our long-term planning we consider horizons of both five and ten years. However, as most of our efforts are focused on the coming three years, we have chosen this period when considering our viability.
Our conclusion is based on our current Strategic Plan approved by the Board in February 2019, having regard to longer-term strategic intentions, yet to be formulated in detail. However, we operate in a changing marketplace, which might cause us to adapt our Strategic Plan. In responding to changing external conditions, we will continue to evaluate any additional risks involved which might impact the business model.
By order of the Board, on 21 February 2019.

Susan Swabey
Company Secretary
|
52 |
Governance |
Smith & Nephew Annual Report 2018 |
|
|
Governance |
|
| 54 | |
| 54 | |
| 58 | |
| 71 | |
| 74 | |
| 76 | |
| 117 |

The Board is committed to the highest standards of corporate governance and we comply with all the provisions of the UK Corporate Governance Code 2016 (the Code). The Company’s American Depositary Shares are listed on the New York Stock Exchange (NYSE) and we are therefore subject to the rules of the NYSE as well as to the US securities laws and the rules of the Securities Exchange Commission (SEC) applicable to foreign private issuers. We comply with the requirements of the NYSE and SEC and have no significant differences to report between the UK and US corporate governance standards. We shall explain in this Corporate Governance Statement and in the reports on the Audit Committee, the Nomination & Governance Committee, the Compliance & Culture Committee and the Remuneration Committee, how we have applied the provisions and principles of the Financial Conduct Authority’s (FCA) Listing Rules, Disclosure & Transparency Rules (DTRs) and the Code throughout the year. The Code can be found at
www.frc.org.uk/getattachment/ca7e94c4‑b9a9‑49e2‑a824‑ad76a322873c/UK-Corporate-Governance-Code-April‑2016.pdf.
In addition, we are making progress in complying with certain aspects of the 2018 Corporate Governance Code early.
The Directors’ Report comprises pages 2, 10‑11, 24‑25,32‑39, 40‑83, 116, 154‑156, 173, 187‑209 of the Annual Report.
|
|
Smith & Nephew Annual Report 2018 |
|
53 |
|
Chair’s introduction We are making progress implementing the 2018 UK Corporate Governance Code. |
|
DEAR SHAREHOLDER,
I am pleased to present the governance section of our Annual Report, which includes details about the Board and an explanation of our individual roles and responsibilities. We also summarise the activities of the Board during the year and the Chair of each Board Committee discusses the activities of that Committee during the past year.
The publication by the Financial Reporting Council in 2018 of the new UK Corporate Governance Code (the ‘Code’) and the Guidance on Board Effectiveness has caused us to reconsider some aspects of how we will operate as a Board going forward. We are in the process of reorganising the composition and the responsibilities of some of the Board Committees to reflect the new emphasis in the Code on listening to the employee voice and having regard to the views of all our key stakeholders. We explain our intended approach in the reports from the Nomination & Governance Committee and the Ethics & Compliance Committee, which will become the expanded Compliance & Culture Committee. The scope of the Remuneration Committee has also been broadened and this is discussed in the Remuneration Report. These changes are reflected in revised terms of reference, which are available on our website and in revised Committee memberships, explained further in the individual Committee reports.
We have also included a section explaining how the Board has fulfilled its duties towards our key stakeholders – our employees, our customers and suppliers, our investors and governments and regulators – in 2018, and how we plan to build on this in 2019.
In particular, we have considered how we can listen better to the employee voice. We feel that as a unitary Board, this is very much the responsibility of the Board as a whole and that this responsibility should not fall to any one designated director or to an employee representative. We are a global company operating out of multiple locations, with employees coming from many different perspectives. Elsewhere in this report, you will have read about the work Namal and the executive team have done in the past year, developing the new purpose, strategic imperatives and culture pillars. This work included engagement with a large number of our employees across the world at all levels of the organisation. The Board intends to build on these now established means of engagement, so that we too can listen effectively and respond to the employee voice. This programme will be developed for us by the expanded Compliance & Culture Committee and will be in addition to the engagement we currently have with employees when we undertake site visits as part of our Board programme.
CHANGES TO THE BOARD IN 2018
During the year to 31 December 2018, there were the following changes to the Board:
|
1. |
Namal Nawana joined the Board as Chief Executive Officer on 7 May 2018. |
|
2. |
Olivier Bohuon retired from the Board as Chief Executive Officer on 7 May 2018. |
|
3. |
Roland Diggelmann joined the Board as a Non-Executive Director and Member of the Audit Committee on 1 March 2018. |
|
4. |
Joseph Papa retired from the Board on 12 April 2018. |
At the Annual General Meeting to be held on 11 April 2019 there will be some additional Board changes. Ian Barlow, our Senior Independent Director and former Chair of the Audit Committee will be retiring after nine years’ service to the Company. Michael Friedman, Chair of our Compliance & Culture Committee will also be retiring after six years’ service.
We would like to thank both Ian and Michael for their outstanding contribution to the Company through periods of change. Michael has given us deeper insight into regulatory matters and Ian has been a valuable support to me on many matters, most recently in finding Namal Nawana, our new Chief Executive Officer.
Robin Freestone will succeed Ian as Senior Independent Director and Marc Owen will succeed Michael as Chair of the expanded Compliance & Culture Committee. Over the course of the coming months, we shall be undertaking a search for a new Non-Executive Director with recent and relevant financial experience to be our financial expert and in time take over from Robin as Chair of the Audit Committee.

Roberto Quarta
Chair
|
54 |
Governance |
Smith & Nephew Annual Report 2018 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
ROBERTO QUARTA (69) Chair Joined the Board in December 2013 and appointed Chair following election by shareholders at the 2014 Annual General Meeting. He was also appointed Chair of the Nomination & Governance Committee and a Member of the Remuneration Committee on that day. Career and experience Roberto is a graduate and a former Trustee of the College of the Holy Cross, Worcester (MA), US. He started his career as a manager trainee at David Gessner Ltd, before moving on to Worcester Controls Corporation and then BTR plc, where he was a divisional Chief Executive. Between 1985 and 1989 he was Executive VP of Hitchiner Manufacturing Co., Inc. He returned to BTR plc in 1989 as Divisional Chief Executive, where he was appointed to the main board. From here he moved to BBA Aviation plc, as CEO and then as Chair, until 2007. He has held several board positions, including Non-Executive Director of Powergen plc, Equant N.V., BAE Systems plc and Foster Wheeler AG. His previous Chairmanships include Italtel SpA, Rexel S.A., IMI plc and SPIE SA. He is currently Chair of WPP plc. He is a partner at Clayton Dubilier & Rice and a former member of the Investment Committee of Fondo Strategico Italiano S.p.A. Skills and competencies Roberto’s career in private equity brings valuable experience to Smith & Nephew, particularly when evaluating acquisitions and new business opportunities. He has an in-depth understanding of differing global governance requirements having served as a director and chairman of a number of UK and international companies. Since his appointment as Chair in April 2014, he has conducted a comprehensive review into the composition of the Board and its Committees, and conducted the search for new Non-Executive Directors, resulting in the appointment of Vinita Bali in 2014, Erik Engstrom and Robin Freestone in 2015, Angie Risley and Marc Owen during 2017, and Roland Diggelmann in 2018. Roberto also conducted the search resulting in the appointment of Namal Nawana as our CEO in 2018. Nationality
|
NAMAL NAWANA (48) Chief Executive Officer Joined the Board and was appointed Chief Executive Officer on 7 May 2018. He will stand for election by shareholders at the AGM on 11 April 2019. He is based in Andover, US. Career and experience Prior to Smith & Nephew Namal was Chief Executive Officer and a member of the Board of Medical Diagnostics Company Alere Inc, until its $8 billion acquisition by Abbott in 2017. Before joining Alere, Namal spent more than 15 years at Johnson & Johnson in progressively senior leadership roles globally including the role of Worldwide President of Johnson & Johnson’s multi-billion dollar spine franchise, DePuy Synthes Spine. In addition to his role as CEO of Smith & Nephew, Namal is also a member of the Board of Directors of Hologic, Inc. a Nasdaq listed company and Advamed (Advanced Medical Technology Association). Skills and competencies Namal holds an undergraduate degree in Mechanical Engineering and a Masters degree in Medical Science from the University of Adelaide, South Australia, as well as an MBA from Henley Management College. He is a global leader with broad experience in healthcare and medical technology. Nationality
|
GRAHAM BAKER (50) Chief Financial Officer Joined the Board as Chief Financial Officer in March 2017. He is based in London, UK. Career and experience Graham holds an MA degree in Economics from Cambridge University and qualified as a Chartered Accountant and Chartered Tax Adviser with Arthur Andersen. In 1995, he joined AstraZeneca PLC where he worked for 20 years, holding multiple senior roles, including Vice President Finance & Chief Financial Officer, North America (2008‑2010), Vice President, Global Financial Services (2010‑2013) and Vice President, Finance, International (2013‑2015) with responsibility for all emerging markets. Most recently, Graham was Chief Financial Officer of generic pharmaceuticals company Alvogen. Skills and competencies Graham has deep sector knowledge and has had extensive exposure to established and emerging markets which is extremely relevant to his role at Smith & Nephew. He has a strong track record of delivering operational excellence and has relevant experience across major finance roles and geographic markets, leading large teams responsible for significant budgets. Nationality
|
|
|
Smith & Nephew Annual Report 2018 |
|
55 |
|
|
|
|
|
|
|
|
|
|
|
|
|
VINITA BALI (63) Independent Non-Executive Director Appointed Independent Non-Executive Director in December 2014 and Member of the Remuneration Committee and Compliance & Culture Committee. Career and experience Vinita holds an MBA from the Jamnalal Bajaj Institute of Management Studies, University of Bombay and a BA in Economics from the University of Delhi. She commenced her career in India with a Tata Group Company, and then joined Cadbury India, subsequently working with Cadbury Schweppes plc in the UK, Nigeria and South Africa. She has held a number of senior global positions in marketing and general management at The Coca-Cola Company based in the US and South America, becoming President of the Andean Division in 1999 and VP, Corporate Strategy in 2001. In 2003, she joined Zyman Group, LLC, a US-based consultancy, as Managing Principal. Vinita was MD and CEO of Britannia Industries Limited, a leading Indian publicly listed food company from 2005 to 2014. Currently, Vinita is NED of Syngene International Limited, Bunge Limited and CRISIL India (a Standard & Poor Company). She is also a member of the Advisory Board of PwC India. Skills and competencies Vinita has an impressive track record of achievement with blue-chip global corporations in multiple geographies including India, Africa, South America, US and UK, all key markets for Smith & Nephew. Her strong appreciation of customer service and marketing brings deep insight as Smith & Nephew continues to develop innovative ways to serve our markets and grow our business. Nationality
|
THE RT. HON BARONESS VIRGINIA BOTTOMLEY OF NETTLESTONE DL (70) Independent Non-Executive Director Appointed Independent Non-Executive Director in April 2012 and Member of the Remuneration Committee and Nomination & Governance Committee in April 2014 and will join the Compliance & Culture Committee in April 2019. Career and experience Virginia gained her MSc in Social Administration from the London School of Economics following her first degree. She was appointed a Life Peer in 2005 following her career as a Member of Parliament between 1984 and 2005. She served successively as Secretary of State for Health and then Culture, Media and Sport. Virginia was formerly a Director of Bupa and AkzoNobel NV. She is currently a Director of International Resources Group Limited, where she is Chair of Board & CEO Practice at Odgers Berndtson. She is a member of the International Advisory Council of Chugai Pharmaceutical Co., Chancellor of University of Hull and Sheriff of Kingston upon Hull. She is a Trustee of The Economist Newspaper. Skills and competencies Virginia’s extensive experience within Government, particularly as Secretary of State for Health, brings a unique insight into the healthcare system both in the UK and globally, whilst her experience on the board of Bupa brings an understanding of the private healthcare sector and an insight into the needs of our customers. Her experience running the board practice at a search firm gives her a valuable skillset as a member of the Nomination & Governance Committee and Remuneration Committee. Her long association with Hull, the home of many of our UK employees, also brings an added perspective. Nationality
|
ROLAND DIGGELMANN (51) Independent Non-Executive Director Appointed Independent Non-Executive Director and Member of the Audit Committee on 1 March 2018. He was elected by shareholders at the AGM on 12 April 2018. He will join the Compliance & Culture Committee in April 2019. Career and experience Roland studied Business Administration at the University of Berne. In 1995, he joined Sulzer AG as Manager Strategic Planning and progressed into further senior roles over the years until his appointment as Executive Vice President, Sales Europe and Asia Pacific from 2002 to 2004 for Sulzer Medica (later known as Centerpulse). Roland joined Zimmer Group in 2004, in the role of Managing Director of Zimmer Japan and then later in 2006 as Senior Vice President, EMEA until 2008. Roland joined Roche Diagnostics in 2008 starting as president of Asia Pacific before assuming the role of Chief Executive Officer of the Diagnostics Division of F. Hoffmann-La Roche Ltd from 2012 until September 2018. Skills and competencies Having spent his whole career in medical devices, with 12 years at Sulzer and Zimmer, Roland brings an in-depth knowledge of the medical device industry and healthcare environment which is of great value to Smith & Nephew. Nationality
|
|
56 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Leadership
Our Board of Directors continued
|
|
|
|
|
|
|
|
|
|
|
|
|
ERIK ENGSTROM (55) Independent Non-Executive Director Appointed Independent Non-Executive Director in January 2015 and Member of the Audit Committee. He will join the Nomination & Governance Committee in April 2019. Career and experience Erik is a graduate of the Stockholm School of Economics (BSc) and of the Royal Institute of Technology in Stockholm (MSc). In 1988, he graduated with an MBA from Harvard Business School as a Fulbright Scholar. Erik commenced his career at McKinsey & Company and then worked in publishing, latterly as President and COO of Random House Inc. and as President and CEO of Bantam Doubleday Dell, North America. In 2001, he moved on to be a partner at General Atlantic Partners, a private equity investment firm. Between 2004 and 2009, he was CEO of Elsevier, the division specialising in scientific and medical information and then from 2009 CEO of RELX Group. Skills and competencies Erik has successfully reshaped RELX Group’s business in terms of portfolio and geographies. He brings a deep understanding of how technology can be used to transform a business and insight into the development of new commercial models that deliver attractive economics. His experience as a CEO of a global company gives him valuable insights as a member of our Audit and Nomination & Governance Committees. Nationality
|
ROBIN FREESTONE (60) Independent Non-Executive Director Appointed Independent Non-Executive Director and Member of the Audit Committee and the Remuneration Committee in September 2015 and Chair of the Audit Committee in April 2017. Robin will succeed Ian Barlow as Senior Independent Director following the AGM on 11 April 2019 and join the Nomination & Governance Committee. Career and experience Robin graduated with a BA in Economics from The University of Manchester and later qualified and commenced his career as a Chartered Accountant at Deloitte. He has held a number of senior financial positions throughout his career, including at ICI plc, Henkel Ltd and at Amersham plc. Robin was the Deputy CFO and then later the CFO of Pearson plc between 2006 and August 2015, where he was heavily involved with the transformation and diversification of Pearson. He was previously NED at eChem Ltd, Chair of the 100 Group and Senior Independent Director and Chair of the Audit Committee of Cable & Wireless Communications plc. Robin is NED and Chair of the Audit Committee at Capri Holdings Ltd, (formerly Michael Kors Holdings Ltd). Robin became Chair of the ICAEW Corporate Governance Committee in 2017 and is currently a NED and Chair of the Audit Committee at MoneySupermarket.com plc. Robin will be appointed as Chair of their Board with effect from the conclusion of its Annual General Meeting on 9 May 2019. Skills and competencies Robin has been a well-regarded FTSE 100 CFO who has not only been heavily involved with transformation and diversification, but also the healthcare industry at Amersham, where his acquisition experience is of value to Smith & Nephew as it continues to grow globally and in different markets. He brings financial expertise and insight as Chair of the Audit Committee and an understanding of how to attract and retain talent in a global business as a member of the Remuneration Committee. Nationality
|
MARC OWEN (59) Independent Non-Executive Director Appointed Independent Non-Executive Director and Member of the Audit Committee in October 2017 and Member of the Compliance & Culture Committee in March 2018. He will be appointed chair of the Compliance & Culture Committee in April 2019. Career and experience Marc graduated from Oxford University with a BA and BCL in Law. In 1984 he was called to the Bar, following four years at Corpus Christi College Cambridge as a fellow and director of studies in law. He decided upon a corporate career and undertook an MBA at Stanford University. Marc commenced his healthcare and technology career at McKinsey & Company where he progressed to senior partner and eventually a founding partner of McKinsey’s Business Technology Office. In September 2001, Marc joined McKesson Corporation and served as Executive Vice President and member of the Executive Committee. He delivered strategic objectives and led over 40 acquisitions and divestments over a 10‑year period. In late 2011 he headed Mckesson Speciality Health, which operates over 130 cancer centres across the US and provides services including market intelligence, supply chain services, patient access to therapy, provider and patient engagement and clinical trial support. His final executive role came in 2014 where he was appointed Chair of the European Management Board at Celesio AG. He retired in March 2017 once he had improved operations, set the strategy and recruited his successor. Skills and competencies Marc is a proven leader with an astute, strategic vision, capable of building significant international healthcare businesses. He has strong commercial healthcare expertise which the Board values deeply and makes him ideally placed to Chair the Compliance & Culture Committee. Nationality
|
|
|
Smith & Nephew Annual Report 2018 |
|
57 |
|
|
|
|
|
|
DIRECTORS WHO SERVED DURING 2018 RETIRING AT ANNUAL GENERAL MEETING |
|
|
|
|
|
|
ANGIE RISLEY (60) Independent Non-Executive Director Appointed Independent Non-Executive Director in September 2017 and appointed Chair of the Remuneration Committee on 12 April 2018. Career and experience After graduating from Exeter University, and completing a 1‑year personnel management programme, Angie joined the United Biscuits graduate scheme. After working in various different HR roles she joined Pizza Hut (UK) Ltd as Human Resources Director, a joint venture between PepsiCo and Whitbread plc. After five years she joined Whitbread, becoming Executive Director on the plc board responsible for HR and Corporate Social Responsibility in 2004. Between 2007–2013 she was the Group HR Director for Lloyds Banking Group, joining J Sainsbury plc as Group HRD in January 2013. Over the years, Angie has been a member of the Low Pay Commission and has held a number of Non-Executive Directorships with Biffa plc, Arriva and Serco Group plc, and now Smith & Nephew. At Serco she was the Chair of the Remuneration Committee. Previously she has attended Remuneration Committees of Whitbread, Lloyds Banking Group, Arriva and attends Sainsbury’s today. She is also a Non-Executive Director on the Sainsbury’s Bank Board. Skills and competencies Angie is a well-regarded FTSE 100 Human Resources Director, proven Non-Executive Director and Remuneration Committee Chair. She has gained experience in a wide range of sectors, including a regulated environment. This diversity of experience is welcomed by the Board and the Remuneration Committee. Angie is also an additional resource and sounding board for Smith & Nephew’s own internal Human Resources function. Nationality
|
IAN BARLOW (67) Independent Non-Executive Director Appointed Independent Non-Executive Director in March 2010, Chairman (now Member) of the Audit Committee in May 2010, Member of the Compliance & Culture Committee in October 2014 and Senior Independent Director and Member of the Nomination & Governance Committee on 6 April 2017. Ian will retire from the Board at the Annual General Meeting on 11 April 2019 and will not stand for re-election. Career and experience Ian is a Chartered Accountant with considerable financial experience both internationally and in the UK. He was a Partner at KPMG, latterly Senior Partner, London, until 2008. At KPMG, he was Head of UK tax and legal operations. Previously he was Chairman of WSP Group plc, and is currently NED and Chairman of the Audit Committees of The Brunner Investment Trust PLC, Foxtons Group plc and Urban&Civic plc. MICHAEL FRIEDMAN (75) Independent Non-Executive Director Appointed Independent Non-Executive Director in April 2013 and Chairman of the Compliance & Culture Committee in August 2014. Michael will be retiring from the Board at the Annual General Meeting on 11 April 2019 and will not stand for re-election. Career and experience Michael is medically trained, specialising in Internal Medicine and Medical Oncology. He was formerly CEO of City of Hope in California, and also served as Director of the institution’s cancer centre. He was formerly Senior VP of research, medical and public policy for Pharmacia Corporation and also Deputy Commissioner and Acting Commissioner at the US Food and Drug Administration (FDA). He has served on a number of boards in a non-executive capacity and is currently a NED of Celgene Corporation, MannKind Corporation and Intuitive Surgical, Inc. |
SUSAN SWABEY (57) Company Secretary Joined Smith & Nephew in May 2009 as Company Secretary with responsibility for Board support and corporate governance, employee and executive share plans and subsidiary governance. In 2016, she also assumed responsibility for leading the group’s risk management programme. She is based in Watford. Career and experience Susan has over 30 years’ experience as a Company Secretary in a wide range of companies including Prudential plc, Amersham plc and RMC Group plc. Her work has covered board support, corporate governance, remuneration, corporate transactions, group risk management, share registration, listing obligations, corporate social responsibility, pensions, insurance and employee and executive share plans. Susan holds an MA from Corpus Christi College Oxford in Literae Humaniores and is a Fellow of the Institution of Chartered Secretaries: The Governance Institute. She is also Chair of ShareGift, the share donation charity, a member of the Financial Reporting Council Lab Steering Group and a frequent speaker on corporate governance and related matters. |
|
58 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Leadership
|
|
|
|
|
Namal Nawana is supported in the day-to-day management of the Group by Graham Baker, Chief Financial Officer, and a strong team of Executive Officers. |
|
|
|
|
|
|
|
RODRIGO BIANCHI (58) Interim President, Asia Pacific Joined Smith & Nephew in July 2013 with responsibility for Greater China, India, Russia, Asia, Middle East and Africa, focusing on continuing our strong momentum in these regions. With effect from 1 January 2016, Rodrigo also became responsible for the Latin American, Australian, New Zealand and Japanese markets. His role was further expanded in May 2017, when he became responsible for oversight of the markets in Europe and Canada. He is now President of Asia Pacific. He is based in Dubai, UAE. Skills and experience Rodrigo’s experience in the healthcare industry includes 26 years with Johnson & Johnson in progressively senior roles. Most recently, he was Regional Vice President for the Medical Devices and Diagnostics division in the Mediterranean region and prior to that President of Mitek and Ethicon, Inc. He started his career at Procter & Gamble, Italy. Nationality
|
BRAD CANNON (50) President, Sports Medicine & ENT Joined Smith & Nephew in 2012 and has since been the President of Smith & Nephew’s Europe and Canada business, the Company’s Chief Marketing Officer, and now serves as the President of the Global Sports Medicine and Ear, Nose and Throat business. He is based in Andover, US. Skills and experience Brad was most recently the Chief Marketing Officer and prior to that the President of Europe and Canada, where he successfully led the commercial business in those regions. He has also served as the President of Global Orthopaedic Franchises, leading Smith & Nephew’s Reconstruction, Endoscopy, Trauma and Extremities businesses. Prior to Smith & Nephew, Brad worked in Medtronic’s Spine and Biologics division. From 2009, he was responsible for Medtronic’s Spine International division and held positions heading US sales and global commercial operations. Brad is a graduate of Washington and Lee University, and the Wharton School of Business at the University of Pennsylvania. Nationality
|
|
NEW COMMERCIAL MODEL: CREATION OF GLOBAL FRANCHISES |
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
59 |
|
|
|
|
|
|
|
|
|
|
|
|
|
MASSIMILIANO COLELLA (50) President, Europe, Middle East and Africa Joined Smith & Nephew in 2013 as Senior Vice President for Mid-Tier Business based in Dubai, Massimiliano (Max to his colleagues and friends), afterwards took over the role of SVP Smith & Nephew AsiaPac in Singapore. Promoted to President Europe and Canada, he moved back to Europe in 2017. He is now President of Europe, Middle East and Africa. Massimiliano is based in Baar, Switzerland. Skills and experience Over his 26 years spent in the medical device industry, Massimiliano has held a number of national and international roles in Europe, Middle‑East and Asia. Before joining Smith & Nephew, Max worked for 21 years in Johnson & Johnson, leading a number of different businesses in Ethicon and DePuy Synthes franchises. Nationality
|
PHIL COWDY (51) Executive Vice President, Business Development & Corporate Affairs Joined Smith & Nephew in 2008 as Director of Investor Relations. From 2010 his responsibility expanded as Head of Corporate Affairs, including media, investor relations, global brand and government affairs, together with Strategic Planning. Between 2015 and 2018 he was also responsible for IT. In 2018 he took on additional responsibility for Business Development. He is based in London, UK. Skills and experience Prior to joining Smith & Nephew, Phil served as a senior Director at Deutsche Bank for 13 years, providing corporate finance and equity capital markets advice to a variety of UK‑based companies. He qualified as a chartered accountant with EY. Nationality
|
SIMON FRASER (51) President of Advanced Wound Management Joined Smith & Nephew in January 2019 with commercial leadership responsibility for Advanced Wound Management in the US and global upstream marketing for the Advanced Wound Management Franchise. As part of his executive responsibilities Simon will also provide leadership for Healthcare Systems. Simon is based in Fort Worth, US. Skills and Experience Simon brings to this role more than 25 years of experience across medical devices, pharmaceuticals and diagnostics, including wound management. Importantly, he is a purpose-driven and accomplished business leader who has successfully managed large, global commercial organisations with full P&L responsibility while growing business and earning market share. Prior to joining Smith & Nephew Simon was Group Vice President of Dentsply-Sirona’s Dental Implant Global Business Unit. Prior to this Simon was Vice President, US Commercial Infectious Diseases including corporate accounts at Abbott Laboratories. Simon joined Abbott following the acquisition of Alere where he had three successful years as the President of Latin America. Prior to these roles, Simon had a 15‑year career with Johnson & Johnson, where he held increasingly senior commercial roles spanning surgical devices, wound management, implants and pharmaceuticals including both global strategic marketing and P&L responsibilities. Nationality
|
|
60 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Leadership
Executive team continued
|
|
|
|
|
|
|
|
|
|
|
|
|
MARK GLADWELL (43) President of Global Operations Joined Smith & Nephew in August 2018 with responsibility for Global Manufacturing, Global Supply Chain, Global Procurement, Global Engineering and Global Operational Excellence, and all Operational Strategy and programmes including APEX projects related to Global Operations. Mark is based in Watford, UK. Skills and experience Mark joined Smith & Nephew from QIAGEN, a provider of sample and assay technologies for molecular diagnostics, applied testing, academic and pharmaceutical research. There he was Senior Vice President of Global Operations responsible for global manufacturing, supply chain, quality assurance and control, regulatory affairs, and global customer service. Mark is a seasoned operational leader bringing more than 20 years of experience in progressively senior operations roles across global organisations including DuPont, AGFA Medical Imaging, Johnson & Johnson, and Alere Inc. Mark has experience of working and living in Europe and the US and operating global manufacturing and supply chain organisations with a significant focus and track record in delivering operational excellence transformation programmes. Nationality
|
MELISSA GUERDAN (44) Chief Quality and Regulatory Affairs Officer Joined Smith & Nephew in July 2018 with responsibility for Quality and Regulatory Affairs and is based in Andover, US. Skills and Experience Melissa brings more than 20 years of leadership experience in Quality and Regulatory Affairs spanning the pharmaceutical, medical device and biologics industries. Melissa has deep compliance and operations knowledge and has progressed through senior leadership roles in global organisations including Pfizer, Baxter, Covidien and Alere. Most recently, Melissa was Senior Vice President, Quality and Regulatory for Alere where she had executive responsibility for establishing enterprise vision, strategy and direction for all aspects of quality, compliance and regulatory affairs. Melissa is adept at inspiring diverse global organisations to achieve common goals and has consistently delivered material value at the enterprise level through transformational quality and regulatory improvement programmes. Melissa holds a BA degree in Biology and Psychology, and holds an MBA from DePaul University. Nationality
|
SKIP KIIL (44) President of Orthopaedics Joined Smith & Nephew in November 2018 with global responsibility for the Orthopaedics franchise, which includes Reconstruction, Trauma, Extremities and Robotics. Skip is based in Memphis, US. Skills and Experience Skip is a seasoned leader who brings a wealth of global experience from diverse medical technology companies, and importantly, significant global experience in Orthopaedics markets over an extended period. Prior to joining Smith & Nephew, Skip was most recently responsible for all Global Commercial Operations at NuVasive and member of the senior executive leadership team. Prior to this, Skip spent three years with Alcon, a division of Novartis Corporation, based in Geneva Switzerland where he served as Surgical Head, Europe, Middle East, Africa and Russia. While at Alcon, Skip led the successful commercial transformation of its $1.1bn surgical business across both developed and emerging growth markets. Before joining Alcon, Skip had a successful 12‑year career with Stryker Corporation, beginning in sales and holding progressively senior positions in commercial leadership in the US as well as in global marketing. Skip also had general management experience in Japan, as well as group leadership responsibilities in Europe where he held the role of Vice President and General Manager of its Medical Surgical Group. Nationality
|
|
|
Smith & Nephew Annual Report 2018 |
|
61 |
|
|
|
|
|
|
|
|
|
|
|
|
|
ELGA LOHLER (51) Chief Human Resources Officer Joined Smith & Nephew in January 2002 as Director of HR and has since held progressively senior positions in Wound Management, Operations, Corporate Functions and Group. Elga became Chief Human Resources Officer in December 2015 and leads the Global Human Resources, Internal Communication and Sustainability Functions. Elga is based in Fort Worth, US. Skills and Experience Elga has more than 25 years’ Human Resources experience. Prior to joining Smith & Nephew, Elga held Human Resources roles at Transnet, Sensormatic (now Tyco) and Advanced Tissue Sciences, which was acquired by Smith & Nephew in 2002. Through these roles, Elga has developed deep expertise in strategy planning and development, organisational design and effectiveness, restructuring and integration and transformational change in support of business objectives. In her current roles, Elga is responsible for driving Smith & Nephew’s human capital strategy across the enterprise in support of the company’s overall business plan and strategic direction. Elga holds an undergraduate degree in Psychology and a Master’s degree in Organizational Psychology, both from the University of Witwatersrand in South Africa. Nationality
|
CATHY O’ROURKE (46) Chief Legal and Compliance Officer Cathy joined Smith & Nephew in February 2013 and became Chief Legal Officer in May 2017 and Chief Legal and Compliance Officer in July 2018. Cathy heads up the Global Legal and Compliance functions and is based in Andover, US. Skills and Experience Prior to being appointed Chief Legal Officer, Cathy had various responsibilities within Legal as Assistant General Counsel – Litigation and Investigations. Prior to joining Smith & Nephew, Cathy spent 11 years of her career with Davis Polk & Wardwell LLP. Cathy earned her Juris Doctorate in Law from Harvard University. Nationality
|
VASANT PADMANABHAN (52) President of Research & Development Vasant joined Smith & Nephew in August 2016 and is responsible for Research and Innovation, New Product Development, Safety Affairs, Clinical Affairs, Medical device/Pharmacovigilance and Clinical Operations. He is based in Andover, US. Skills and Experience Vasant brings extensive experience in R&D and technology. Prior to Smith & Nephew, Vasant was Senior Vice President of Technical Operations at Thoratec Corporation, a leader in mechanical circulatory support solutions for the treatment of heart failure. In this role, he provided leadership to a 600 member team, with responsibility for global R&D, Programme Management, Operations and Quality. Prior to Thoratec, Vasant had an 18‑year career at Medtronic, starting as a Staff Scientist and, progressing through more senior roles, ultimately becoming Vice President of Product Development for the Implantable Defibrillator Business. Vasant holds a Ph.D degree in Biomedical Engineering from Rutgers University, USA and an MBA degree from the Carlson School of Management, Minnesota. Nationality
|
|
62 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Leadership
Roles and composition
Whilst we all share collective responsibility for the activities of the Board, some of our roles have been defined in greater detail below.
|
CHAIR - Building a well-balanced Board. - Chairing Board meetings and setting Board agendas. - Ensuring effectiveness of the Board and enabling the annual review of effectiveness. - Encouraging constructive challenge and facilitating effective communication between Board members. - Promoting effective Board relationships. - Ensuring effective two-way communication and debate with shareholders and stakeholders. - Promoting high standards of corporate governance. - Maintaining appropriate balance between stakeholders. |
|
CHIEF FINANCIAL OFFICER - Supporting the Chief Executive Officer in developing and implementing the Group strategy. - Leading the global finance function, developing key finance talent and planning for succession. - Ensuring effective financial reporting, processes and controls are in place. - Recommending the annual budget and long-term strategic and financial plan. - Maintaining relationships with shareholders. |
|
CHIEF EXECUTIVE OFFICER - Developing and implementing Group strategy. - Recommending the annual budget and three-year strategic and financial plan. - Ensuring coherent leadership of the Group. - Managing the Group’s risk profile and establishing effective internal controls. - Regularly reviewing organisational structure, developing executive team and planning for succession. - Ensuring the Chair and Board are kept advised and updated regarding key matters. - Maintaining relationships with shareholders and advising the Board accordingly. - Setting the tone at the top with regard to compliance and sustainability matters. - Day-to-day running of the business.
|
|
SENIOR INDEPENDENT DIRECTOR - Chairing meetings in the absence of the Chair. - Acting as a sounding board for the Chair on Board-related matters. - Acting as an intermediary for the other Directors where necessary. - Available to shareholders and stakeholders on matters which cannot otherwise be resolved. - Leading the annual evaluation into the Board’s effectiveness. - Leading the search for a new Chair, if necessary. |
|
COMPANY SECRETARY - Advising the Board on matters of corporate governance. - Supporting the Chair and Non-Executive Directors. - Point of contact for investors on matters of corporate governance. - Ensuring good governance practices at Board level and throughout the Group. |
|
|
Smith & Nephew Annual Report 2018 |
|
63 |
Corporate governance framework
The Board is responsible to shareholders for approving the strategy of the Group, for overseeing the performance of the Group and evaluating and monitoring the management of risk. Each member of the Board has access, collectively and individually, to the Company Secretary and is also entitled to obtain independent professional advice at the Company’s expense, should they decide it is necessary in order to fulfil their responsibilities as Directors.
The Board delegates certain matters, as follows, to Board Committees, consisting of members of the Board:
|
BOARD |
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Audit Committee Provides independent assessment of the financial affairs of the Company, reviews financial statements and controls oversight of the risk management process and key risks, such as cyber security. Manages use of internal and external auditors. |
|
Remuneration Committee Determines Remuneration Policy and packages for Executive Directors and Executive Officers, having regard to pay across the Group. |
|
Nomination & Reviews size and composition of the Board, succession planning, diversity and governance matters. |
|
Compliance & Reviews and monitors ethics and compliance, quality and regulatory matters across the Group. Role to be expanded in 2019 to include oversight of culture, sustainability and stakeholder relationships. |
|
Ad hoc Ad hoc committees may be established to review and approve specific matters or projects. |
|||||
|
|
|
|
|
|
|
|
|
|
|||||
The Board delegates the day-to-day running of the business to Namal Nawana, Chief Executive Officer, who is assisted in his role by the Executive Committee comprising the Executive team shown on pages 58‑61. The governance framework below outlines the Executive Committee arrangements as follows:
|
EXECUTIVE COMMITTEE |
|
The Executive Committee meets regularly and makes decisions collectively. It recommends and implements strategy, recommends budget and three-year plan to the Board for approval, ensures liaison between commercial and corporate functions, receives regular reports from sub-committees, reviews major investments, divestment and capital expenditure proposals and approves business development projects. |
|
|
|
|
|
|
|
|
|
|
|
|
|
Monthly Operating Review Wider group of senior commercial and financial leaders reviews monthly commercial and marketing and operating results against budget, identifying gaps and agreeing remedial actions. |
|
Franchise, Functional and Regional Leadership Meetings Senior management meetings to drive performance across each franchise, function and region. |
|
Portfolio Innovation Board Defines portfolio allocation principles, reviewing and challenging current shape of portfolio, identifying gaps and opportunities and re-prioritising segments and geographies. |
|
Group Ethics & Compliance Committee Reviews compliance matters and country business unit or function compliance reports. |
||||
|
|
|
|
|
|
|
|
||||
|
Finance & Banking Committee Approves banking and treasury matters, guarantees, Group structure changes relating to mergers, acquisitions and disposals. |
|
Disclosures Committee Approves release of communications to investors and Stock Exchanges. |
|
Mergers & Acquisitions Investment Committee Oversees Corporate Development Strategy, monitors status of transactions and approves various stages in merger, acquisition and disposal process. |
|
IT Governance Board Oversees IT and cyber security. |
||||
|
|
|
|
|
|
|
|
||||
|
Diversity & Inclusion Council Implements strategies to promote diversity and inclusion. |
|
Global Benefits Committee Oversees all policies and processes relating to pensions and employee benefit plans. |
|
Health, Safety & Oversees health, safety and environmental matters. |
|
|
||||
|
64 |
Governance |
Smith & Nephew Annual Report 2018 |
|
|
LEADERSHIP |
|
|
Responsibilities of the Board |
|
|
|
|
January |
Early February |
Late February |
April |
May |
|
|
|
|
2017 Preliminary Results |
2017 Financial Statements |
Annual General Meeting |
Q1 2018 |
|
STRATEGY |
|
|
|
|
|
|
|
– Approving the Group strategy including major changes to corporate and management structure. – Approving acquisitions, mergers, disposals, capital transactions in excess of $50 million. – Setting priorities for capital investment across the Group. – Approving annual budget, financial plan, three-year business plan. – Approving major borrowings and finance and banking arrangements. – Approving changes to the size and structure of the Board and the appointment and removal of Directors and the Company Secretary. – Approving Group policies relating to sustainability, health and safety, Code of Conduct and Code of Share Dealing and other matters. – Approving the appointment and removal of key professional advisers. |
|
Approved Strategic Plan for 2018‑2020 Approved 2018 Budget |
Reviewed capital allocation policies Reviewed report on post acquisitions reviews |
|
|
|
|
PERFORMANCE |
|
|
|
|
|
|
|
– Reviewing performance against strategy, budgets and financial and business plans. – Overseeing Group operations and maintaining a sound system of internal control. – Determining the dividend policy and dividend recommendations. – Approving the appointment and removal of the external auditor on the recommendation of the Audit Committee. – Approving significant changes to accounting policies or practices. – Overseeing succession planning at Board and Executive Officer level. – Approving the use of the Company’s shares in relation to employee and executive share incentive plans on the recommendation of the Remuneration Committee. |
|
|
Reviewed financial performance Received update on European business |
|
Reviewed financial performance Received updates on global operations |
|
|
SHAREHOLDER COMMUNICATIONS |
|
|
|
|
|
|
|
– Approving preliminary announcement of annual results, the publication of the Annual Report, the half-yearly report, the quarterly Trading Reports, the release of price sensitive announcements and any listing particulars, circulars or prospectuses. – Approving the Sustainability Report. – Maintaining relationships and continued engagement with shareholders. |
|
|
Approved Preliminary Announcement 2017 Considered payment of final dividend |
Approved the: Annual Report for 2017
Notice of the Annual General Sustainability Report 2017 |
Prepared for the Annual General Meeting to be held later that day |
Approved Q1 2018 Trading Report |
|
RISK |
|
|
|
|
|
|
|
– Overseeing the Group’s risk management programme. – Regularly reviewing the risk register. – Overseeing risk management processes (see pages 41‑49 for further details). |
|
|
Approved Annual Risk Management Report |
|
Reviewed update on Brexit |
|
|
PROVIDING ADVICE |
|
|
|
|
|
|
|
– Using experience gained within other companies and organisations to advise management both within and between Board meetings on an ad hoc basis. |
||||||
|
OTHER MATTERS |
|
|
|
|
|
|
|
|
|
Reviewed Non‑Executive Director Fees |
|
|
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
65 |
|
The Schedule of Matters Reserved to the Board describes the role and responsibilities of the Board more fully and can be found on our website at www.smith-nephew.com. |
|
|
|
|
June |
July |
September |
October |
December |
|
|
|
|
H1 2018 |
Site visit to Berlin |
Q3 Trading Report |
Strategic Planning |
|
Strategy |
|
|
|
|
|
|
|
– Approving the Group strategy including major changes to corporate and management structure. – Approving acquisitions, mergers, disposals, capital transactions in excess of $50 million. – Setting priorities for capital investment across the Group. – Approving annual budget, financial plan, three-year business plan. – Approving major borrowings and finance and banking arrangements. – Approving changes to the size and structure of the Board and the appointment and removal of Directors and the Company Secretary. – Approving Group policies relating to sustainability, health and safety, Code of Conduct and Code of Share Dealing and other matters. – Approving the appointment and removal of key professional advisers. |
|
Approved re-financing of the revolving credit facility |
Considered the Company’s organisational design Considered the Company’s strategic focus |
Update on organisational change |
|
Approved the strategic plan for 2019-2021 Approved the budget for 2019 |
|
Performance |
|
|
|
|
|
|
|
– Reviewing performance against strategy, budgets and financial and business plans. – Overseeing Group operations and maintaining a sound system of internal control. – Determining the dividend policy and dividend recommendations. – Approving the appointment and removal of the external auditor on the recommendation of the Audit Committee. – Approving significant changes to accounting policies or practices. – Overseeing succession planning at Board and Executive Officer level. – Approving the use of the Company’s shares in relation to employee and executive share incentive plans on the recommendation of the Remuneration Committee. |
|
Received report from new Chief Executive Officer on first impressions |
Reviewed financial performance, operating review and scorecards |
Reviewed financial performance and global operations |
Reviewed financial performance Received update on Medical Devices global market |
|
|
Shareholder communications |
|
|
|
|
|
|
|
– Approving preliminary announcement of annual results, the publication of the Annual Report, the half-yearly report, the quarterly Trading Reports, the release of price sensitive announcements and any listing particulars, circulars or prospectuses. – Approving the Sustainability Report. – Maintaining relationships and continued engagement with shareholders. |
|
|
Approved H1 2018 Results Announcement |
Considered report on investor and media perspectives |
Approved Q3 2018 Trading Report |
|
|
Risk |
|
|
|
|
|
|
|
– Overseeing the Group’s risk management programme. – Regularly reviewing the risk register. – Overseeing risk management processes (see pages 41‑49 for further details). |
|
|
|
|
|
Reviewed risk management programme as part of strategy review |
|
Providing Advice |
|
|
|
|
|
|
|
– Using experience gained within other companies and organisations to advise management both within and between Board meetings on an ad hoc basis. |
||||||
|
OTHER MATTERS |
|
|
|
|
|
|
|
|
|
|
|
Approved the terms of the Directors’ and Officers’ Liability Insurance |
|
Reviewed results of external board effectiveness review and agreed follow up actions |
|
66 |
Governance |
Smith & Nephew Annual Report 2018 |
|
|
LEADERSHIP |
|
|
Our approach to stakeholders |
|
|
EMPLOYEES |
The Board regularly takes the opportunity to meet with staff at all levels in the organisation when making site visits across our operations. Regular staff surveys are undertaken, which the Board reviews and follows up on outcomes. The Compliance & Culture Committee reviews certain workplace policies and whistle-blowing incidents, ensuring that appropriate follow up is implemented as necessary. We ensure that when making strategic decisions the impact upon our employees is fully considered.
Actions during 2018
– The Board reviewed and discussed the results of the Culture Survey and focus forums initiated by Namal Nawana, Chief Executive Officer on joining the Company. See page 24‑25.
– We actively debated and approved the culture pillars of Care, Collaboration and Courage, which will underpin our purpose and corporate strategy.
– We met with key employees on our site visit to Berlin in September and some of us have undertaken additional site visits and spent time on the road with our sales representatives.
– The Ethics & Compliance Committee reviewed our policies on sexual harassment.
– In making decisions regarding organisational change and the APEX programme, we have considered the impact on employees, ensuring that all are treated fairly and with respect.
– The Chief Executive Officer and senior management hold quarterly webcasts for the workforce, which encourage employee engagement and dialogue.
|
Actions planned for 2019 – The Board will continue to consider the impact on employees when making strategic decisions. – We will continue to take opportunities to meet staff at all levels across the organisation when conducting site visits. We plan to visit our Memphis facility in 2019. – The Compliance & Culture Committee (formerly Ethics & Compliance Committee) will assume responsibility for overseeing our corporate culture and workplace policies and will report back regularly to the Board. – In listening to the employee voice, we have decided not to appoint an employee director or to nominate a single designated Non-Executive Director. Instead, we have designated our Compliance & Culture Committee as a whole with the responsibility for engaging with employees. This programme of employee engagement will be developed and implemented during 2019. – The Board will continue to monitor the outcomes from employee surveys and forums. |
|
CUSTOMERS AND SUPPLIERS |
The Board receives regular updates at Board meetings from the management team on relationships with our key customers and suppliers and how these relationships are evolving as we respond to different market conditions and environments. We also take the opportunity to meet with key customers during our site visits.
Actions during 2018
– As part of our country visit to Germany, we visited La Charité Hospital in Berlin, which is the largest University hospital in Germany. We met with surgeons who used our products and also the Chief Executive who outlined some of the challenges in the German market.
– A number of us accompanied sales representatives in the UK, US and Switzerland as they met with customers and surgeons who used our products.
– During the course of the year, we received management updates on different areas of the business. These included presentations on the management of our supply chain, and the changing market environment and expectations from our customers.
|
Actions planned for 2019 – The Board will continue to receive management updates on our customers and suppliers. – We will meet with customers and surgeons when we visit our Memphis facility. – The Compliance & Culture Committee will assume responsibility for overseeing relationships with our key stakeholders including customers and suppliers. |
|
|
Smith & Nephew Annual Report 2018 |
|
67 |
|
INVESTORS |
The Board meets with retail investors at the Annual General Meeting and responds to letters and emails from shareholders throughout the year. Members of the Board are always happy to engage with investors, if they have matters they wish to raise with the non-executive team. The Chair and other Board members report back to the Board following their meetings with investors. A short report on our major shareholders and any significant changes in their holdings since the previous meeting is reviewed at each Board meeting. Copies of the analyst reports on the Company and its peers are also circulated to Directors.
Actions during 2018
– The Executive Directors held 124 meetings with investors representing 43.8% of the Company’s share capital. They discussed a range of topics including strategy, performance and organisational structure.
– The Chair, Roberto Quarta, held 7 meetings and telephone calls with investors holding approximately 12.6% of the Company’s share capital. They discussed a range of topics including the performance of the Company, the appointment of Namal Nawana as our new Chief Executive Officer, our Strategy, the structure of the Board and succession planning at Board and Executive level.
– The Chair of the Remuneration Committee, Angie Risley reached out to our top 20 shareholders holding 44% of the company’s shares and received responses from 9 shareholders collectively holding 17.1% of the company’s share capital in connection with Graham Baker’s pay following his increased responsibility for IT and Global Business Services. See page 85 for further details.
|
Actions planned for 2019 – The Board will continue to receive regular updates at Board meetings on management and Chair meetings with investors and will review regular analyst reports. – The management team and the Chair and Non-Executive Directors will continue to engage with shareholders. If you have matters to raise with the non-executive team, please contact the Company Secretary. |
|
GOVERNMENTS AND REGULATORS |
In many countries, our principal customers are governments, who purchase our products for their national health systems. It is important that we maintain good relationships with governments so that we continue to develop cost efficient solutions to their national healthcare issues. We operate in a heavily regulated industry and our businesses across the world are regulated by many different authorities in different jurisdictions. Our Compliance & Culture Committee has for a number of years overseen our relationships with our key regulators, particularly in areas of ethics, compliance and quality.
Actions during 2018
– The Compliance & Culture Committee received regular reports from the Quality and Regulatory function on regulatory activities and the results of regulatory inspections.
– The Compliance & Culture Committee received regular reports from the Legal & Compliance function on the activities of key agencies relating to ethics and compliance matters.
– The Board received regular updates from the business areas on the pricing challenges faced by the business when dealing with governments operating with limited financial resources.
– The Board and the Audit Committee received regular updates relating to the progress towards Brexit and management plans to manage the transition as smoothly as possible.
|
Actions planned for 2019 – The Compliance & Culture Committee will continue to oversee relationships between the Company and our regulators. – The Board and the Audit Committee will continue to monitor management preparations for Brexit. |
|
68 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Leadership
Responsibilities of the Board
BOARD AND COMMITTEE ATTENDANCE
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Board |
|
Committee |
|||
|
|
|
|
|
|
Audit |
Remuneration |
Nominations |
Compliance & |
|
Total meetings |
|
|
10 |
|
8 | 5 |
4 |
4 |
|
|
Appointed |
|
|
|
|
|
|
|
|
Roberto Quarta¹ |
December 2013 |
|
10/10 |
|
– |
5/5 |
3/4 |
– |
|
Olivier Bohuon² |
April 2011 |
|
5/5 |
|
– |
– |
– |
– |
|
Graham Baker |
March 2017 |
|
10/10 |
|
– |
– |
– |
– |
|
Vinita Bali |
December 2014 |
|
10/10 |
|
– |
5/5 |
– |
4/4 |
|
Ian Barlow³ |
March 2010 |
|
10/10 |
|
7/8 |
– |
4/4 |
4/4 |
|
Virginia Bottomley |
April 2012 |
|
10/10 |
|
– |
5/5 |
4/4 |
– |
|
Roland Diggelmann4 |
1 March 2018 |
|
7/7 |
|
5/6 |
– |
– |
– |
|
Erik Engstrom |
January 2015 |
|
10/10 |
|
8/8 |
– |
– |
– |
|
Robin Freestone |
September 2015 |
|
10/10 |
|
8/8 |
5/5 |
– |
2/2 |
|
Michael Friedman |
April 2013 |
|
10/10 |
|
– |
– |
– |
4/4 |
|
Namal Nawana5 |
7 May 2018 |
|
5/5 |
|
– |
– |
– |
– |
|
Marc Owen |
October 2017 |
|
10/10 |
|
8/8 |
– |
– |
3/3 |
|
Joseph Papa6 |
August 2008 |
|
4/4 |
|
3/3 |
2/2 |
– |
2/2 |
|
Angie Risley |
September 2017 |
|
10/10 |
|
– |
5/5 |
– |
– |
1Roberto Quarta missed one Nomination & Governance Committee meeting on 11 April 2018 because of another commitment.
2Olivier Bohuon retired from the Board on 7 May 2018.
3Ian Barlow missed one Audit Committee meeting on 11 April 2018 in order to attend a funeral.
4Roland Diggelmann was appointed on 1 March 2018. He missed one Audit Committee meeting on 24 July 2018 because of another commitment scheduled before his appointment.
5Namal Nawana was appointed on 7 May 2018 and attended all his scheduled meetings to 31 December 2018.
6Joseph Papa retired from the Board at the Annual General Meeting on 12 April 2018.
INDEPENDENCE OF DIRECTORS
We require our Non-Executive Directors to remain independent from management so that they are able to exercise independent oversight and effectively challenge management. We therefore continually assess the independence of each of our Non-Executive Directors. The Executive Directors have determined that all our Non-Executive Directors are independent in accordance with both UK and US requirements. None of our Non-Executive Directors or their immediate families has ever had a material relationship with the Group. None of them receives additional remuneration apart from Directors’ fees, nor do they participate in the Group’s share plans or pension schemes. None of them serve as directors of any companies or affiliates in which any other Director is a director.
MANAGEMENT OF CONFLICTS OF INTEREST
None of our Directors or their connected persons, has any family relationship with any other Director or Officer, nor has a material interest in any contract to which the Company or any of its subsidiaries are, or were, a party during the year or up to 15 February 2019.
Each Director has a duty under the Companies Act 2006 to avoid a situation in which they have or may have a direct or indirect interest that conflicts or might conflict with the interests of the Company. This duty is in addition to the existing duty owed to the Company to disclose to the Board any interest in a transaction or arrangement under consideration by the Company.
If any Director becomes aware of any situation which might give rise to a conflict of interest, they must, and do, inform the rest of the Board immediately and the Board is then permitted under the Company’s Articles of Association to authorise such conflict. This information is then recorded in the Company’s Register of Conflicts, together with the date on which authorisation was given. In addition, each Director certifies on an annual basis that the information contained in the Register of Conflicts is correct.
When the Board decides whether or not to authorise a conflict, only the Directors who have no interest in the matter are permitted to participate in the discussion and a conflict is only authorised if the Board believes that it would not have an impact on the Board’s ability to promote the success of the Company in the long term. Additionally, the Board may determine that certain limits or conditions must be imposed when giving authorisation. No actual conflicts have been identified, which have required approval by the Board. However, eight situations have been identified which could potentially give rise to a conflict of interest and these have been duly authorised by the Board and are reviewed on an annual basis.
OUTSIDE DIRECTORSHIPS
We encourage our Executive Directors to serve as Non-Executive Directors of external companies. We believe that the work they do as Non-Executive Directors of other companies has benefits for their executive roles with the Company, giving them a fresh insight into the role of a Non‑Executive Director. Namal Nawana is an independent Director of Hologic Inc. Namal discussed his external role with the Chair prior to his appointment and the Chair was satisfied that he had the capacity for the time commitment required. Suitable arrangements were put in place when reaching a settlement with Hologic related to historical intellectual property litigation to ensure that Namal was not party to any of the negotiations or discussions, which could have given rise to an actual conflict.
RE-APPOINTMENT OF DIRECTORS
In accordance with the Code, all Directors offer themselves to shareholders for re-election annually, except those who are retiring immediately after the Annual General Meeting. Each Director may be removed at any time by the Board or the shareholders.
DIRECTOR INDEMNITY ARRANGEMENTS
Each Director is covered by appropriate directors’ and officers’ liability insurance and there are also Deeds of Indemnity in place between the Company and each Director. These Deeds of Indemnity mean that the Company indemnifies Directors in respect of any proceedings brought by third parties against them personally in their capacity as Directors of the Company. The Company would also fund ongoing costs in defending a legal action as they are incurred rather than after judgement has been given. In the event of an unsuccessful defence in an action against them, individual Directors would be liable to repay the Company for any damages and to repay defence costs to the extent funded by the Company.
PURCHASE OF ORDINARY SHARES
In order to avoid shareholder dilution, shares allotted to employees through employee share schemes are bought back on a quarterly basis and subsequently cancelled as stated in Note 19.2 of the accounts on page 172.
|
|
Smith & Nephew Annual Report 2018 |
|
69 |
Board effectiveness review
The Board effectiveness review in 2018 was externally facilitated by Dr Tracy Long of Boardroom Review. Dr Long interviewed each member of the Board and the Company Secretary, reviewed minutes, Board papers and other Board documents, and attended and observed the Board and Committee meetings held in October 2018. She then prepared a report summarising her findings, which she presented to the Board for discussion in December.
FINDINGS
Overall she observed that the Board had many strengths, was effective with a diversity of perspectives and was an open supportive environment. It was led by a strong Chair, the Chief Executive Officer and Chief Financial Officer were both highly regarded and recent appointments had strengthened the Board’s domain knowledge. Finance, risk and governance controls were working well and corporate culture was openly discussed at Board level.
RECOMMENDATIONS
She did note however that there were challenges ahead, given the number of initiatives underway across the Company to lift performance, the recent appointment of a new Chief Executive Officer, a refreshed management team and the development of new Strategic Imperatives, Purpose and Culture Pillars. There would be additional work for the Board in coming months to get behind the new strategy and develop a shared perspective on the future strategy and appetite for risk.
The Board accepted the recommendations and an action plan is being developed to address them.
|
RECOMMENDATION 1 |
|
– The Board will need to ensure that it continually reviews and ensures alignment of its appetite for risk against the changing landscape and revised Strategic Imperatives, particularly as the Company continued to evolve. This will require continued monitoring of Board composition and succession planning. |
|
RECOMMENDATION 2 |
|
– Performance management will need to evolve to monitor alignment with the new strategy, with an increased emphasis on a globally consistent culture and purpose. |
|
RECOMMENDATION 3 |
|
– The workload, composition and support for the Board Committees will be reviewed to ensure a more even balance of workload and greater diversity on each Committee. |
|
RECOMMENDATION 4 |
|
– The Chair and Chief Executive Officer will agree shared priorities for Board site visits at the beginning of each year, so that individual and group site visits could be arranged within these agreed parameters rather than on an ad hoc basis. |
The areas for attention identified in the 2017 review have been addressed as follows:
|
ACTIONS IDENTIFIED |
ACTION TAKEN |
|
Changes could be made to the Board calendar to spread our work more efficiently and effectively throughout the year, with an even greater focus on people issues, R&D and commercial execution. |
During the year, the Board and Committee calendar for the year has been reviewed and updated and a revised rolling calendar has been prepared for 2019, aligned to the new Group strategy and the updated Strategic Imperatives. |
|
We would like to spend more time on our site visits meeting the local teams, their staff, our customers and local hospitals to give us a deeper understanding of our markets, our customers and competition, and to assist in assessing bench strength further down the Company. |
The Board site visit to Berlin in September 2018 incorporated meetings with the German management team and a visit to La Charite, the largest university hospital in Germany to meet with surgeons and hospital staff. This helped us gain a deeper understanding of the views and needs of our customers. |
|
Further improvements could be made to how we monitor performance against our strategic objectives, tracking development and implementation of our strategy, and lessons learned from our successes and shortfalls. |
During the year, we have evolved the way reports on performance are presented to the Board. Going forward, performance reports will be aligned with the new Strategy and Strategic Imperatives. |
The reviews in 2019 and 2020 will be facilitated internally and led by the Senior Independent Director and the Company Secretary. The 2021 review will also be facilitated externally.
|
70 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Leadership
Board development
BOARD DEVELOPMENT PROGRAMME
Our Board Development Programme is directed to the specific needs and interests of our Directors. We focus the development sessions on facilitating a greater awareness and understanding of our business rather than formal training in what it is to be a Director. We value our visits to the different Smith & Nephew sites around the world, where we meet with the local managers of our businesses and see the daily operations in action. Meeting our local managers helps us to understand the challenges they face and their plans to meet those challenges. We also take these opportunities to look at our products and in particular the new products being developed by our R&D teams. This direct contact with the business in the locations in which we operate around the world helps us to make effective investment and strategic decisions. Meeting our local managers also helps us when making succession planning decisions below Board level.
All Non-Executive Directors are encouraged to visit our overseas businesses, if they happen to be travelling for other purposes. Our local management teams enjoy welcoming Non-Executive Directors to their business and it emphasises the interest the Board takes in all our operations. The Chair regularly reviews the development needs of individual Directors and the Board as a whole.
The following development sessions covering the Smith & Nephew business and wider market issues were held during the year:
|
June |
September |
October |
December |
|
|
|
|
|
|
2018 PROGRAMME |
|||
|
The Board received an overview from the new Chief Executive Officer on his first impressions of the Company and his proposals for a new organisational structure. |
Visit to Germany to meet with our local management team and to visit La Charité, the largest university hospital in Germany and one of our significant customers. The Board received presentations from local surgeons and members of the hospital’s executive team about the challenges faced in the German healthcare market and role played by Smith & Nephew. The Board received updates from new members of the Executive team on their first impressions on joining the Company and their plans for the EMEA business, Global Operations and Quality and Regulatory Assurance. The Board received updates from the Executive team on Investor and Media coverage, and the results of the culture survey and focus groups carried out earlier in the summer. |
Presentation from Deloitte LLP to the Remuneration Committee on the changes to be made to the role of the Committee in light of the new UK Corporate Governance Code. Presentation by the Company Secretary to the Audit Committee/Nomination & Governance Committee on the changes required by the UK Corporate Governance Code to the roles and structure of the Board and its Committees. Presentation to the Board by McKinsey on the current environment and trends in the Medical Devices industry.
|
Presentations from the entire Executive team as part of the Board’s annual Strategy Review, covering the whole business.
|
INDUCTION PROGRAMME FOR NEW DIRECTORS
– During 2018, Roland Diggelmann joined the Board. He and Angie Risley and Marc Owen continued to receive tailored induction programmes relevant to their skills and experiences and their roles on the Board. These induction programmes included:
– One-to-one meetings with senior executives to understand the roles played by our senior employees and specifically how we do things at Smith & Nephew;
– Visits to our sites local to the Director in UK, US and Switzerland to get a feel of how our research and manufacturing operations are run;
– Opportunities to accompany our sales representatives in the US on the road to better understand the daily challenges they face; and
– Meetings with our external advisers for example Freshfields, our Corporate lawyers, KPMG, our Auditor, and Deloitte LLP, our Remuneration Committee adviser to explain the legal and regulatory background to their role on our Board and how these issues are approached at Smith & Nephew.
|
|
Smith & Nephew Annual Report 2018 |
|
71 |
|
Nomination & Governance Committee report |
|
|
|
|
Ensuring the Board evolves to align with the new Strategic Imperatives and with the developing external regulatory environment. |
|
|
|
|
|
Membership
|
|
|
Member from |
|
Meetings attended |
|
Roberto Quarta (Chair)1 |
|
April 2014 |
|
3/4 |
|
Ian Barlow2 |
|
April 2014 |
|
4/4 |
|
Erik Engstrom3 |
|
April 2019 |
|
N/A |
|
Robin Freestone3 |
|
April 2019 |
|
N/A |
|
Virginia Bottomley |
|
April 2014 |
|
4/4 |
1Roberto Quarta missed the meeting in April because of a conflicting commitment.
2Ian Barlow will be retiring from the Board and the Committee at the Annual General Meeting to be held on 11 April 2019, having completed nine years’ service.
3Erik Engstrom and Robin Freestone will join the Nomination & Governance Committee on 11 April 2019.
The terms of reference for the Nominations & Governance Committee describe the role and responsibilities of this Committee more fully and can be found on our website at
www.smith-nephew.com.
|
|
Early February |
March |
April |
October |
|
|
|
|
|
|
|
BOARD COMPOSITION |
||||
|
– Reviewing the size and composition of the Board. – Overseeing Board succession plans. – Recommending the appointment of Directors. – Monitoring Board diversity. |
Approved the re-appointment of Directors who had completed three or six years’ service and the annual appointment of Directors serving in excess of six years. Discussed progress on search for a new Chief Executive Officer. |
Considered candidates for the role of Chief Executive Officer and agreed to recommend to the Board the appointment of Namal Nawana. |
|
Reviewed the composition of the Board and agreed to appoint Robin Freestone as Senior Independent Director to replace Ian Barlow who would be retiring from the Board at the Annual General Meeting in 2019. |
|
CORPORATE GOVERNANCE |
||||
|
– Overseeing governance aspects of the Board and its Committees. – Overseeing the review into the effectiveness of the Board. – Considering and updating the Schedule of Matters Reserved to the Board and the terms of reference of the Board Committees. – Monitoring external corporate governance activities and keeping the Board updated. – Overseeing the Board Development Programme and the induction process for new Directors. |
Reviewed and approved the Schedule of Matters Reserved to the Board and the terms of reference of the Board Committees. |
|
Reviewed current governance trends in the UK and particularly the changes likely to be proposed in the UK Corporate Governance Code. |
Reviewed the annual cadence of Board and committee meetings. Considered the implications of the UK Corporate Governance Code 2018 and in particular the expanded role of the Board relating to corporate culture and relationships with stakeholders and agreed that the remit of the Ethics & Compliance Committee be expanded to undertake some of these responsibilities on behalf of the Board. |
|
72 |
Governance |
Smith & Nephew Annual Report 2018 |
|
NOMINATION & GOVERNANCE COMMITTEE REPORT continued
In 2018, we held four meetings. In addition to members of the Committee, the Company Secretary, Chief Executive Officer and Chief Human Resources Officer also attended all or some of the meetings. Other Non-Executive Directors were invited to join the meetings to discuss the search for a new Chief Executive Officer.
Further matters were resolved by written resolution including the appointment of Roland Diggelmann.
Since the year end, we have also discussed the future structure of the Board and its Committees and completed our year end governance processes.
APPOINTMENT OF CHIEF EXECUTIVE OFFICER
During the early part of the year, the Committee continued its search for a new Chief Executive Officer to replace Olivier Bohuon who had announced his intention to retire by the end of 2018. The Committee recommended the appointment of Namal Nawana, who joined the Company as Chief executive Officer in May 2018. Namal brings a wealth of experience of the international medical devices industry to his role. The Nomination & Governance Committee was advised by Russell Reynolds on this appointment. Russell Reynolds also advises the Company on other executive recruitment and appointments.
NON-EXECUTIVE DIRECTORS
The Committee has reviewed the composition of the Board and its Committees to ensure that it evolves to align with the new Strategic Imperatives and with the developing external regulatory environment.
The Committee also recommended the appointment of Roland Diggelmann as an additional Non-Executive Director who joined the Board in March 2018. Roland has many years’ experience in the Medical Devices industry. Russell Reynolds advised the Committee on this appointment.
Ian Barlow will be retiring from the Board and from his position as Senior Independent Director at the Annual General Meeting to be held on 11 April 2019, following completion of nine years’ service as Non-Executive Director and until 2017 as Chairman of the Audit Committee and then latterly as Senior Independent Director. The Committee recommended to the Board that Robin Freestone be appointed Senior Independent Director in Ian’s place at the conclusion of the Annual General Meeting, subject to his re-appointment by shareholders.
Robin will continue to Chair the Audit Committee, but the Committee will undertake a search for an additional Non-Executive Director with recent and relevant financial experience who could in time succeed Robin as Chair of the Audit Committee. When considering candidates for this role, the Committee will be mindful of building a diverse Board and will aim to ensure an appropriate balance of genders, ethnicity, backgrounds, skills and experience.
Michael Friedman will also be retiring from the Board and from his position as Chair of the Compliance & Culture Committee at the Annual General Meeting, following the completion of six years’ service. He will be succeeded as Chair of the expanded Compliance & Culture Committee by Marc Owen.
Erik Engstrom and Robin Freestone will be joining the Nomination & Governance Committee as additional members in April 2019 after the Annual General Meeting and Virginia Bottomley and Roland Diggelmann will be joining the Compliance & Culture Committee at the same time.
DIVERSITY
We aim to have a Board which represents a wide range of backgrounds, skills and experiences. We also value a diversity of outlook, approach and style in our Board members. We believe that a balanced Board is better equipped to consider matters from a broader perspective, understanding the views of our stakeholders as well as our shareholders and therefore come to decisions that have considered a wider range of issues and perspectives than would be the case in a more homogeneous Board. Diversity is not simply a matter of gender, ethnicity or other easily measurable characteristics. Diversity of outlook and approach is harder to measure than gender or ethnicity but is equally important. A Board needs a range of skills from technical adherence to governance or regulatory matters to understanding the business in which we operate. It needs some members with a long corporate memory and others who bring new insights from other fields.
There needs to be both support and challenge on the Board as well as a balance of gender and commercial and international experience. When selecting new members for the Board, we take these considerations into account, as well as professional background. A new Board member needs to fit in with their fellow Board members, but also needs to provide a new way of looking at things.
During 2018, 25% of our Board were female. With the retirement of Ian Barlow and Michael Friedman in April 2019, this percentage rises to 30%. Looking forward, we are working towards a Board with 33% female representation in line with the Hampton-Alexander Review. From 7 May 2018, 17% of our Board were of non-white ethnicity. This rises to 20% after the Annual General Meeting in April 2019.
We will also look to increase ethnic diversity on the Board following the Parker Review as appropriate. We will continue to appoint our Directors on merit, valuing the unique contribution that they will bring to the Board, regardless of gender, ethnicity or any other diversity measure.
SUCCESSION PLANNING
Since the appointment of Namal Nawana as Chief Executive Officer in May 2018, we have initiated substantial changes to our structure to move to a franchise-led model, as described on page 14. Throughout the year, the Board and Nomination & Governance Committee have monitored the consequent changes to the organisational structure and approved appointments to key leadership positions. Individual Directors have acted as a sounding board for the executive team when considering succession plans in key areas. Given the level of change during 2018, this will be a continued focus of the Nomination & Governance Committee during 2019.
|
|
Smith & Nephew Annual Report 2018 |
|
73 |
We believe the Board’s composition gives us the necessary balance of diversity, skills experience, independence and knowledge to ensure we continue to run the business effectively and deliver sustainable growth. In order to ensure that our Board remains diverse, we analyse the skills and experiences we require against the skills and experiences on our Board using the following matrix. We review this matrix regularly to ensure that it is refreshed to meet the changing needs of the Company.
|
CEO |
|
Financial |
|
International |
|
Healthcare/ |
|
Emerging market |
|
|
|
|
|
|
|
|
|
|
|
Four members of the Board are either current or recent CEOs |
|
Two members of the Board have recent and relevant financial experience |
|
Seven members of the Board have international experience |
|
Six members of the Board have different levels of experience within the Healthcare industry |
|
Two members of the Board have Emerging Market experience |
|
|
|
|
|
|
|
|
|
|
|
UK Governance |
|
Remuneration |
|
Gender |
|
Ethnic |
|
Other |
|
|
|
|
|
|
|
|
|
|
|
Six members of the Board have considerable experience of working in a UK listed environment and four members of the Board have experience of the US listed environment |
|
Five members of the Board have Remuneration Committee experience within a UK listed context |
|
Nine members of the Board are male and three are female |
|
Ten members of the Board are white and two are of Asian ethnicity |
|
Various Board members bring experiences in a variety of fields including customer focus, investment markets, government affairs, digital and corporate social responsibility |
GOVERNANCE
During the year, the Nomination & Governance Committee also addressed a number of governance matters. We received updates from the Company Secretary on new developments in corporate governance and reporting in the UK. We reviewed the independence of our Non-Executive Directors, considered potential conflicts of interest and the diversity of the Board and made recommendations concerning these matters to the Board.
We also took the opportunity of reviewing whether the annual cadence of our Board and Committee meetings was appropriate for the work we need to undertake. As a result of this review, we have changed the physical October meeting to approve the third quarter trading figures to a half day Board telephone call. We have also introduced additional monthly Board update calls for those months when we do not meet physically and will be introducing separate site visits for Non-Executive Directors to meet employees and customers.
We also considered how we would implement the requirements of the 2018 UK Corporate Governance Code and agreed that the Remuneration Committee would retain responsibility for provisions relating to executive pay and pay matters generally. This is discussed further on pages 84-85. We also agreed that the remit of the new Compliance & Culture Committee would be expanded to cover responsibility for overseeing corporate culture and relationships with stakeholders on behalf of the Board. The revised remit of the Compliance & Culture Committee is discussed on pages 74–75 and our approach to listening to the voice of our employees and other stakeholders is discussed on pages 66–67. We have noted the three options set out in the 2018 UK Corporate Governance Code for listening to the Employee Voice and have determined that this will be the responsibility of the Compliance & Culture Committee as this is too important an issue to fall to one Non-Executive Director.
LOOKING FORWARD
During 2019 our focus will include:
|
> |
Search for an additional Non-Executive Director with recent and relevant financial experience to serve on the Audit Committee. |
|
|
|
|
> |
Monitoring the implementation of the revised Board and Committee structure to ensure that the Company complies with the UK Corporate Governance Code or explains why not. |

Roberto Quarta
Chairman of the Nomination & Governance Committee
|
74 |
Governance |
Smith & Nephew Annual Report 2018 |
|
|
Compliance & Culture Committee report (formerly Ethics & Compliance Committee) |
|
|
|
|
|
Broadening our scope to oversee culture in addition to our focus on compliance and quality. |
|
|
|
|
|
|
|
Membership
|
|
|
Member from |
|
Meetings attended |
|
Michael Friedman (Chair)1 |
|
August 2014 |
|
4/4 |
|
Vinita Bali |
|
April 2015 |
|
4/4 |
|
Virginia Bottomley2 |
|
April 2019 |
|
N/A |
|
Ian Barlow3 |
|
October 2014 |
|
4/4 |
|
Roland Diggelmann4 |
|
April 2019 |
|
N/A |
|
Marc Owen5 |
|
1 March 2018 |
|
3/3 |
|
Joseph Papa6 |
|
August 2008 |
|
2/2 |
|
Robin Freestone |
|
24 July 2018 |
|
2/2 |
1Michael Friedman will retire from the Board and the Committee at the Annual General Meeting on 11 April 2019.
2Virginia Bottomley will join the Committee on 11 April 2019.
3Ian Barlow will retire from the Board and the Committee on 11 April 2019.
4Roland Diggelmann will join the Committee on 11 April 2019.
5Marc Owen joined the Committee on 1 March 2018.
6Joseph Papa retired from the Board and the Committee on 12 April 2018.
|
|
February |
April |
July |
October |
|
|
|
|
|
|
|
ETHICS & COMPLIANCE |
||||
|
– Overseeing ethics and compliance programmes, strategies and plans. – Monitoring ethics and compliance process improvements and enhancements. – Assessing compliance performance based on monitoring, auditing and internal and external investigations data. – Reviewing allegations of significant potential compliance issues. – Receiving reports from the Group’s Ethics & Compliance Committee meetings and from the Chief Legal and Compliance Officer. |
Noted the progress made on the Global Compliance Programme Plan for 2017 and noted the plan of action for 2018. |
Reviewed the ethics and compliance training programmes in place across the Group. Reviewed significant findings from monitoring, auditing and progress on Corrective and Preventative Actions. |
Noted progress on the 2018 Global Compliance Plan of Action. Reviewed significant findings from monitoring auditing, and progress on Corrective and Preventative Actions. |
Received Ethics & Compliance update in new format and noted insights from various aspects of Global Compliance Programme. |
|
QUALITY ASSURANCE AND REGULATORY AFFAIRS (QARA) |
||||
|
– Overseeing the processes by which regulatory and quality risks relating to the Company and its operations are identified and managed. – Receiving and assessing regular functional reports and presentations from the Chief Quality and Regulatory Affairs Officer, or the SVP Quality Assurance. |
Reviewed various quality metrics. Noted the plans to address the potential impact on the Group of both the EU Medical Device Regulations (EU MDR) and Brexit. |
Reviewed various quality metrics and approved the Global Quality Plan for 2018, noting the additional work to be done to implement the EU MDR. |
Reviewed various quality metrics including the results of inspections by the FDA and Notified Bodies, progress on handling complaints and in preparing for the EU MDR. |
Received Quality and Regulatory report in updated format from newly appointed Chief Quality and Regulatory Affairs Officer, noting status of various Quality and Regulatory metrics and initiatives. |
|
OTHER MATTERS |
February |
|
|
|
|
|
Reviewed the sexual harassment policies and procedures in place across the Group. |
As a follow up to the February meeting, reviewed the circumstances and actions taken to address the sexual harassment allegations made in the year. |
||
The Terms of Reference of the Compliance & Culture Committee describe the role and responsibilities more fully and can be found on our website at www.smith-nephew.com.
|
|
Smith & Nephew Annual Report 2018 |
|
75 |
In 2018, we held four physical meetings. Each meeting was attended by all members of the Committee. The Company Secretary, the Chief Legal and Compliance Officer and the Chief Quality and Regulatory Affairs Officer or SVP, Quality Assurance also attended all or part of the meetings by invitation.
At each meeting we noted and considered the activities of compliance and enforcement agencies and investigation of possible improprieties. At every meeting a report on the Quality Assurance Regulatory Assurance (QARA) function was provided along with updates of product complaint trends. We also reviewed a report on the activities of the Group’s Ethics & Compliance Committee and reviewed the progress of the Global Compliance Programme.
OVERSIGHT OF QUALITY & REGULATORY
Product safety is at the heart of our business. Regulatory authorities across the world enforce a complex series of laws and regulations that govern the design, development, approval, manufacture, labelling, marketing and sale of healthcare products. During the year, we oversaw the quality and regulatory activities of our business. At each meeting, we received a report on quality and regulatory matters from the Chief Quality and Regulatory Affairs Officer, or the SVP Quality Assurance.
We reviewed the results of inspections carried out by the FDA and other regulators and monitored the progress of improvements following some of these inspections. We also monitored the work being undertaken to help our manufacturing sites to prepare for future inspections.
We reviewed the results of quality audits undertaken during the year, noted follow up actions and monitored progress made to address these actions.
OVERSIGHT OF ETHICS & COMPLIANCE
‘Doing the right thing’ is part of our licence to operate. During the year, we oversaw the ethics and compliance activities of our business. At each meeting we received a report on ethics and compliance matters from the Chief Legal and Compliance Officer.
We regularly review our compliance programme as it relates to healthcare professionals and third party sellers (such as distributors and sales agents), particularly in higher risk markets. For healthcare professionals, this includes policies, training and certification for employees and sales agents, as well as pre-approval of consulting services and grants and fellowships. For third parties, our programme includes due diligence, contracts with compliance terms, compliance training and certification, and site assessments to check compliance controls and monitoring visits to review books and records.
We ensure that comprehensive due diligence is carried out prior to an acquisition and we ensure that following acquisitions new businesses are integrated rapidly into the Smith & Nephew compliance programme.
We oversee the employee compliance training programme, ensuring that all new employees are trained on our Code of Conduct, which sets out our basic legal and ethical principles for conducting business. We are updated on significant calls made to our whistle-blower line, which enables employees and members of the public to contact us anonymously through an independent provider (where allowed by local law) and are updated on allegations of potentially significant improprieties and the Company’s response.
During the year, we expanded our remit and reviewed the policies and procedures we have in place to handle claims of sexual harassment.
LOOKING FORWARD
The Board and the Committee have considered the UK Corporate Governance Code 2018 and have decided to expand the role of the Committee to cover a broader focus. Whilst oversight of our Ethics & Compliance programme will continue to remain a key focus for us and we will monitor and assess quality and regulatory issues, we will also oversee culture across the organisation and relationships with our key stakeholders. We will develop the mechanism for ensuring that the Board listens to the employee voice and will oversee the Sustainability Programme.
Our focus for 2019 will include:
|
> |
Develop a programme to enable the Board to monitor and assess the corporate culture. |
|
|
|
|
> |
Develop a programme to enable the Board to engage with employees, building on existing processes within the Company. |
|
|
|
|
> |
Ensure oversight of the Company’s Sustainability programme and relationships with key stakeholders. |
This will be my final report as Chair of this Committee, as I shall be retiring from the Board at the Annual General Meeting. I should like to thank my colleagues on the Committee and particularly Marc Owen who will be succeeding me as Chair of the Committee and overseeing its expanded focus on culture, the employee voice and stakeholders.

Michael Friedman
Chair of the Compliance & Culture Committee
|
76 |
Governance |
Smith & Nephew Annual Report 2018 |
|
|
|
|
||
|
|
2018 was my first full year as Chair of the Audit Committee. I have overseen a busy schedule – with the Committee meeting eight times during the year. |
|
|
|
|
|
|
|
Membership
|
|
|
Member from |
|
Meetings attended |
|
Robin Freestone (Chair)1 |
|
September 2015 |
|
8/8 |
|
Ian Barlow1,2 |
|
May 2010 |
|
7/8 |
|
Roland Diggelmann3,4 |
|
March 2018 |
|
5/6 |
|
Erik Engstrom |
|
January 2015 |
|
8/8 |
|
Mark Owen |
|
October 2017 |
|
8/8 |
|
Joseph Papa5 |
|
February 2011 |
|
3/3 |
1Designated financial experts under the SEC Regulations or recent and relevant financial experience under the UK Corporate Governance Code.
2Ian Barlow missed one Audit Committee meeting on 11 April 2018.
3Roland Diggelmann joined the Audit Committee on 1 March 2018.
4Roland Diggelmann missed one Audit Committee meeting on 24 July 2018.
5Joseph Papa retired from the Board and Audit Committee at the Annual General Meeting on 12 April 2018.
In addition to discharging its role in accordance with its terms of reference, the Committee has met its commitments to provide assurance in respect of various non-routine matters. Areas of scrutiny for the Committee in 2018 have included: the progression of NAPO, (our SAP Enterprise Resource Planning migration in North America); top risks identified in the 2017 Annual Report such as Cyber Security; Brexit readiness; and the Accelerating Performance and Execution (APEX) programme. In 2018 the Committee oversaw the adoption of two accounting standards, IFRS 15 Revenue from contracts with customers and IFRS 9 Financial Instruments. A detailed impact assessment of the application of IFRS 15 was undertaken with the conclusion being that there was no significant impact on the timing and recognition of revenue. The main impact on adoption of IFRS 9 has been the implementation of the expected credit loss methodology for the calculation of the loss allowance for trade receivables. This resulted in an additional loss allowance of $14 million being recognised on 1 January 2018. With regard to IFRS 16 Leases, the Committee considered the application of exemptions, estimated impact, transition preparations (including the implementation of a new lease accounting software solution) and transition readiness. IFRS 16 will be adopted on 1 January 2019 with no restatement of comparatives.
In May 2018 we welcomed our new Chief Executive Officer, Namal Nawana. Under Namal’s leadership, Smith & Nephew has established its new strategy and organisational structure. That will provide the context for our focus of activity for 2019 – particularly as it will form the backdrop for our risk assessment. Roland Diggelmann joined the Audit Committee in March 2018 where his experience and expertise enables him to provide appropriate challenge to information presented at meetings by the Executive. Ian Barlow will be standing down as a member of the Board after the AGM on 11 April. He has served as member of the Committee since 2010 (including seven years as Chair) and has been the Senior Independent Director since 2017. I should like to take this opportunity to thank Ian for his valuable contribution and wise counsel during his tenure.
KPMG have now completed their fourth year’s audit and continue to provide robust challenge. We have negotiated fees that will continue to be reviewed for good market practice. We have also agreed arrangements for rotation of the senior partner in accordance with recommendations set out in the Financial Reporting Council’s (FRC) Guidance for Audit Committees 2016 and as required by the Securities Exchange Commission (SEC). Finally, we note the positive comments received from the FRC following its review of KPMG’s audit of Smith & Nephew’s financial statements for 2017.
Our focus for 2019 will include:
|
> |
The next phase of the APEX programme. |
|
|
|
|
> |
Ensuring compliance with new leasing standard IFRS 16. |
|
|
|
|
> |
Risk management process – aligned to the new strategy and organisational structure. |
|
> |
Continuing to provide assurance on the effectiveness of managing the risks associated with Cyber Security. |
|
|
|
|
> |
Ensuring that we are compliant with additional governance and reporting requirements coming into effect for 2019 – such as the revised UK Corporate Governance Code. |

Robin Freestone
Chair of the Audit Committee
The Terms of Reference of the Audit Committee describe the role and responsibilities more fully and can be found on our website at www.smith-nephew.com.
|
|
Smith & Nephew Annual Report 2018 |
|
77 |
SIGNIFICANT MATTERS RELATED TO THE FINANCIAL STATEMENTS
We considered the following key areas of judgement in relation to the 2018 accounts and at each half-year and quarterly trading report, which we discussed in all cases with management and the external auditor:
Valuation of inventories
A feature of the Orthopaedic Reconstruction and Trauma & Extremities franchises (whose inventory makes up approximately 60% of the Group total inventory) is the high level of product inventory required, some of which is located at customer premises and is available for customers’ immediate use.
Complete sets of products, including large and small sizes, have to be made available in this way. These sizes are used less frequently than standard sizes and towards the end of the product life cycle are inevitably in excess of requirements. Adjustments to carrying value are therefore required to be made to orthopaedic inventory to anticipate this situation. These adjustments are calculated in accordance with a formula based on levels of inventory compared with historical usage. This formula is applied on an individual product line basis and is first applied when a product group has been on the market for two years. This method of calculation is considered appropriate based on experience, but it does involve management estimation of customer demand, effectiveness of inventory deployment, length of product lives, phase-out of old products and efficiency of manufacturing planning systems.
Our action
At each quarter end, we received reports from, and discussed with, management the level of provisioning and material areas at risk. The provisioning level was 18% at 31 December 2018 (19% as at 31 December 2017). We challenged the basis of the provisions and concluded that the proposed levels were appropriate and have been consistently estimated.
Liability provisioning
The recognition of provisions for legal disputes is subject to a significant degree of estimation. Provision is made for loss contingencies when it is considered probable that an adverse outcome will occur and the amount of the loss can be reasonably estimated. In making its estimates, management takes into account the advice of internal and external legal counsel and uses third party actuarial modelling where appropriate. Provisions are reviewed regularly and amounts updated where necessary to reflect developments in the disputes. The ultimate liability may differ from the amount provided depending on the outcome of court proceedings and settlement negotiations or if investigations bring to light new facts.
Our action
As members of the Board, we receive regular updates from the Chief Legal and Compliance Officer. These updates form the basis for the level of provisioning.
The Group carries a provision relating to potential liabilities arising on its portfolio of metal-on-metal hip products of $192 million as of 31 December 2018. We received detailed reports from management on this position, including the actuarial model used to estimate the provision, and challenged the key assumptions, including the number of claimants and projected value of each claim. The provisions for legal matters have increased by $27 million during the year, primarily due to an increase in the metal-on-metal provision. There have been some smaller movements from other cases having been resolved. We have determined that the proposed levels of provisioning at year end of $217 million included within ‘provisions’ in Note 17.1 in 2018 ($190 million in 2017) were appropriate in the circumstances.
Impairment
In carrying out impairment reviews of acquisition intangible assets a number of significant assumptions have to be made when preparing cash flow projections. These include the future rate of market growth, discount rates, the market demand for the products acquired, the future profitability of acquired businesses or products, levels of reimbursement and success in obtaining regulatory approvals. If actual results should differ or changes in expectations arise, impairment charges may be required, which would adversely impact operating results.
Our action
We reviewed management’s reports on the key assumptions with respect to acquisition intangible assets – particularly the forecast future cash flows and discount rates used to make these calculations. We concluded that the carrying value of these assets is appropriately supported by the cash flow projections. We have also considered the disclosure surrounding these reviews, and concluded that the review and disclosure were appropriate.
Taxation
The Group operates in numerous tax jurisdictions around the world and it is Group policy to submit its tax returns to the relevant tax authorities as promptly as possible. At any given time, the Group is involved in disputes and tax audits and will have a number of tax returns potentially subject to audit. Significant issues may take several years to resolve. In estimating the probability and amount of any tax charge, management takes into account the views of internal and external advisers and updates the amount of tax provision whenever necessary. The ultimate tax liability may differ from the amount provided depending on factors including interpretations of tax law, settlement negotiations or changes in legislation.
Our action
We annually review our processes and approve the principles for management of tax risks. We review quarterly reports from management evaluating existing risks and tax provisions. Based on a thorough report from management of tax liabilities and our challenge of the basis of any tax provisions recorded, we concluded that the levels of provisions and disclosures were appropriate.
|
78 |
Governance |
Smith & Nephew Annual Report 2018 |
|
AUDIT COMMITTEE REPORT continued
|
|
|
|
Early February |
Late February |
April |
May |
|
|
|
Preliminary Announcement |
Financial Statements |
|
2018 Q1 Trading Report |
|
|
FINANCIAL ACCOUNTING AND REPORTING |
|
|
|
|
|
|
|
– Reviewing significant financial reporting judgements and accounting policies and compliance with accounting standards. – Ensuring the integrity of the financial statements and their compliance with UK and US statutory requirements. – Ensuring the Annual Report and Accounts are fair, balanced and understandable and recommending their adoption by the Board. – Monitoring announcements relating to the Group’s financial performance. |
|
Endorsed 2017 results and Preliminary announcement Report from KPMG on 2017 results Reviewed draft 2017 Annual Report including report of the Audit Committee Assessed compliance with UK and US governance requirements |
Approved the Annual Report and Accounts for 2017 including report of the Audit Committee – confirming fair, balanced and understandable Report from KPMG on 2017 statements – Unqualified Opinion Approved letter of representation for 2017 Reviewed S302 and S906 certifications Confirmed Going Concern and Viability Statement |
Debrief of Annual Report process and timetable for 2018 Approved Senior Finance Officers Code of Ethics Reviewed summary of Company audits |
Reviewed and endorsed 2018 Q1 Trading Report and announcement Approved the Company’s policy and report on Conflict Minerals for submission to NYSE |
|
|
INTERNAL AUDIT |
|
|
|
|
|
|
|
– Agreeing Internal Audit plans and reviewing reports of Internal Audit work. – Monitoring the effectiveness of the Internal Audit function. – Reviewing the control observations made by the Internal Auditor, the adequacy of management’s response to recommendations and the status of any unremediated actions. |
|
Reviewed year end Report |
Reviewed effectiveness of Internal Audit |
Reviewed progress |
|
|
|
RISK MANAGEMENT |
|
|
|
|
|
|
|
– On behalf of the Board, reviewing and ensuring oversight of the processes by which risks are managed, through regular functional reports and presentations, and reporting any issues arising out of such reviews to the Board. – Reviewing the process undertaken and deep-dive work required to complete the Viability Statement and recommending its adoption to the Board. – Reviewing the impact of risk management and internal controls and working closely with the Ethics & Compliance Committee. – Overseeing risk management processes (see pages 40–49 for further details). – Regularly reviewing the risk register. |
|
Risk Management Update |
Confirmed effective system of risk management in place and approved the Viability Statement |
|
|
|
|
INTERNAL CONTROLS |
|
|
|
|
|
|
|
– Monitoring the effectiveness of internal controls and compliance with the UK Corporate Governance Code 2016 and the Sarbanes-Oxley Act, specifically sections 302 and 404. – Reviewing the operation of the Group’s risk mitigation processes and the control environment over financial risk. |
|
Considered Sarbanes-Oxley (Sox) and MAPs Update |
Reviewed effectiveness of Internal Controls over financial reporting |
Considered Sarbanes-Oxley (Sox) and MAPs Planning 2018 |
|
|
|
FRAUD & WHISTLE-BLOWING |
|
|
|
|
|
|
|
– Receiving reports on the processes in place to prevent fraud and to enable whistle-blowing. – If significant, receive and review reports of potential fraud or whistle-blowing incidents. – Reviewed Internal Audit Report on Fraud. |
|
Reviewed Internal Audit Report on Fraud and Whistle-blowing |
|
|
|
|
|
EXTERNAL AUDITOR |
|
|
|
|
|
|
|
– Overseeing the Board’s relationship with the external auditor. – Monitoring and reviewing the independence and performance of the external auditor and evaluating their effectiveness. – Making recommendations to the Board for the appointment or re-appointment of the external auditor. – Monitoring and approving the external auditor’s fees.
|
|
Confirmed independence of KPMG |
Reviewed effectiveness and independence and concluded their effectiveness Approved external auditor fees for 2017 |
Endorsed External Audit Plan |
Approved 2017 fee overruns |
|
|
OTHER MATTERS |
|
|
|
|
|
|
|
|
|
Endorsed NAPO processing and scheduling plan Approved Audit Committee’s TOR |
Approved consultancy fees to external advisors |
Treasury Update Cyber Security Project reports including Apex China Channel Management |
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
79 |
|
|
|
July |
September |
October |
December |
|
|
|
2018 H1 |
|
2018 Q3 |
|
|
FINANCIAL ACCOUNTING AND REPORTING |
|
|
|
|
|
|
– Reviewing significant financial reporting judgements and accounting policies and compliance with accounting standards. – Ensuring the integrity of the financial statements and their compliance with UK and US statutory requirements. – Ensuring the Annual Report and Accounts are fair, balanced and understandable and recommending their adoption by the Board. – Monitoring announcements relating to the Group’s financial performance. |
|
Reviewed and endorsed H1 results and announcement Report from KPMG on H1 results Approved letter of representation for H1 2018 |
|
Reviewed and endorsed Q3 Trading Report and announcement Clarified MDR policy New reporting and regulatory requirements update |
Reviewed accounting matters for 2018 Considered and approved critical accounting policies and judgements in advance of 2018 year end Reviewed preparation work for adoption of IFRS 16 Approved plans for delivery of the Annual Report 2018 |
|
INTERNAL AUDIT |
|
|
|
|
|
|
– Agreeing Internal Audit plans and reviewing reports of Internal Audit work. – Monitoring the effectiveness of the Internal Audit function. – Reviewing the control observations made by the Internal Auditor, the adequacy of management’s response to recommendations and the status of any unremediated actions. |
|
|
Reviewed progress |
|
Reviewed Progress and approved Charter and 2019 Plan |
|
RISK MANAGEMENT |
|
|
|
|
|
|
– On behalf of the Board, reviewing and ensuring oversight of the processes by which risks are managed, through regular functional reports and presentations, and reporting any issues arising out of such reviews to the Board. – Reviewing the process undertaken and deep-dive work required to complete the Viability Statement and recommending its adoption to the Board. – Reviewing the impact of risk management and internal controls and working closely with the Ethics & Compliance Committee – Overseeing risk management processes (see pages 40–49 for further details). – Regularly reviewing the risk register. |
|
Risk Management Update |
|
Risk Management Update |
Risk Management Update |
|
INTERNAL CONTROLS |
|
|
|
|
|
|
– Monitoring the effectiveness of internal controls and compliance with the UK Corporate Governance Code 2016 and the Sarbanes-Oxley Act, specifically sections 302 and 404. – Reviewing the operation of the Group’s risk mitigation processes and the control environment over financial risk. |
|
|
Considered Sarbanes-Oxley (Sox) and MAPs progress |
|
Considered Sarbanes-Oxley (Sox) and MAPs progress |
|
FRAUD & WHISTLE-BLOWING |
|
|
|
|
|
|
– Receiving reports on the processes in place to prevent fraud and to enable whistle-blowing. – If significant, receive and review reports of potential fraud or whistle-blowing incidents. – Reviewed Internal Audit Report on Fraud |
|
Reviewed Internal Audit Report on Fraud |
|
|
Reviewed Internal Audit Report on Fraud and Whistle-blowing |
|
EXTERNAL AUDITOR |
|
|
|
|
|
|
– Overseeing the Board’s relationship with the external auditor. – Monitoring and reviewing the independence and performance of the external auditor and evaluating their effectiveness. – Making recommendations to the Board for the appointment or re-appointment of the external auditor. – Monitoring and approving the external auditor’s fees.
|
|
Approved Engagement letter for 2018 Noted partner rotation arrangements |
KPMG Fee |
Results of FRC Review of KPMG |
|
|
OTHER MATTERS |
|
|
|
|
|
|
|
|
Cyber Security Update |
Update on NAPO Project reports including APEX Tax Update/Strategy Cyber Security |
|
Tax Update Cyber Update APEX Update
|
|
80 |
Governance |
Smith & Nephew Annual Report 2018 |
|
AUDIT COMMITTEE REPORT continued
OTHER MATTERS RELATED TO THE FINANCIAL STATEMENTS
Regular private meetings have taken place between the Audit Committee and the External auditor (KPMG) and the Audit Committee and the Group Head of Internal Audit.
As well as the identified significant matters, other matters that the Audit Committee considered during 2018 were:
Business combinations
During 2018, we considered and concurred with management that there had been no changes to the provisional fair values recognised in the 2017 acquisition of Rotation Medical, Inc.
Post Retirement Benefits
The Group has post retirement defined benefit pension schemes, which require estimation in setting the assumptions. We received a report from management setting out their proposed assumptions for the UK and US schemes and concurred with management that these assumptions were appropriate.
Since the year end
Since the year end we have also reviewed the results for the full year 2018, the preliminary announcement, Annual Report and Accounts, for 2018 and have concluded that they are fair, balanced and understandable. In coming to this conclusion, we have considered the description of the Group’s strategy and key risks, the key elements of the business model, which is set out on pages 12–13, risks and the key performance indicators and their link to the strategy.
EXTERNAL AUDITOR
Independence of external auditor
Following a competitive tender in 2014, KPMG was appointed external auditor of the Company in 2015. We are satisfied that KPMG are fully independent from the Company’s management and free from conflicts of interest. Our Auditor Independence Policy, which ensures that this independence is maintained, is available on the Company’s website.
We believe that the implementation of this policy helps ensure that auditor objectivity and independence is safeguarded. The policy also governs our approach when we require our external auditor to carry out non-audit services, and all such services are strictly governed by this policy.
The Auditor Independence Policy also governs the policy regarding audit partner rotation with the expectation that the audit partner will rotate at least every five years. Stephen Oxley has been in tenure for four years as our Audit Partner. The Audit Committee confirms it has complied with the provision of the Competition and Markets Authority Order.
Effectiveness of external auditor(s)
We conducted a review into the effectiveness of the external audit as part of the 2018 year end process, in line with previous years. We sought the views of key members of the finance management team, considered the feedback from this process and shared it with management.
During the year, we also considered the inspection reports from the Audit Oversight Board in the UK and determined that we were satisfied with the audit quality provided by KPMG.
The Audit Committee regularly receives feedback from KPMG, including at each meeting where management present their summary of critical accounting estimates as at each quarter end.
Overall therefore, we concluded that KPMG had carried out their audit for 2018 effectively.
The Audit Committee continues to review not only the effectiveness of the external auditor, KPMG, but also its market competitiveness.
Appointment of external auditor at Annual General Meeting
Resolutions will be put to the Annual General Meeting to be held on 11 April 2019 proposing the re-appointment of KPMG as the Company’s auditor and authorising the Board to determine its remuneration, on the recommendation of the Audit Committee in accordance with the Competition and Markets Authority (CMA) Order 2014.
Disclosure of Information to the auditor
In accordance with Section 418 of the Companies Act 2006, the Directors serving at the time of approving the Directors’ Report confirm that, to the best of their knowledge and belief, there is no relevant audit information of which the Auditor, KPMG, is unaware and the Directors also confirm that they have taken reasonable steps to be aware of any relevant audit information and, accordingly, to establish that the Auditor is aware of such information.
Non-Audit Fees Paid to the auditor
Non-audit fees are subject to approval in line with the Auditor Independence Policy which is reviewed annually and forms part of the terms of reference of the Audit Committee.
The Audit Committee recognises the importance of the independence of the external auditor and ensures that the Auditor’s independence should not be breached. The Audit Committee ensures that the Auditor does not receive a fee from the Company or its subsidiaries that would be deemed large enough to impact its independence or be deemed a contingent fee. The total fees for permitted non-audit services shall be no more than 70% of the average of the fees paid in the last three consecutive financial years for the statutory audits of the Company and its subsidiaries.
|
|
Smith & Nephew Annual Report 2018 |
|
81 |
Any pre-approved aggregate, individual amounts up to $25,000 may be authorised by the Group Treasurer and Senior Vice-President Group Finance respectively and amounts up to $50,000 by the Chief Financial Officer. Any individual amount over $50,000 must be pre-approved by the Chairman of the Audit Committee. If unforeseen additional permitted services are required, or any which exceed the amounts approved, again pre-approval by the Chairman of the Audit Committee is required.
The following reflects the non-audit fees incurred with KPMG in 2018, which were approved by the Chairman of the Audit Committee:
| 2018 | 2017 | ||
|
|
|
$ million |
$ million |
|
Tax fees and compliance services |
Assistance with tax compliance in Singapore only |
– |
0.1 |
Tax compliance services conducted by KPMG in 2017 only took place in countries where it is required by law for the auditor to conduct these services.
The ratio of non-audit fees to audit fees for the year ended 31 December 2018 is 0.00. The ratio of non-audit fees to audit fees for the year ended 31 December 2017 was 0.02.
Full details are shown in Note 3.2 of the Notes to the Group accounts.
Audit Fees paid to the auditor
Fees for professional services provided by KPMG, the Group’s independent auditor in each of the last two fiscal years, in each of the following categories were:
|
|
2018 | 2017 |
|
|
$ million |
$ million |
|
Audit fees |
6.0 | 4.4 |
|
Audit-related fees |
– |
– |
|
Total |
6.0 | 4.4 |
INTERNAL AUDIT
The Internal Audit team, which reports functionally to the Audit Committee, carries out risk-based reviews across the Group. These reviews examine the management of risks and controls over financial, operational, IT and transformation programme activities.
The audit team, led by the Group Head of Internal Audit, consists of appropriately qualified and experienced employees. Third parties may be engaged to support audit work as appropriate.
The Group Head of Internal Audit has direct access to, and has regular meetings with, the Audit Committee Chair and prepares formal reports for Audit Committee meetings on the activities and key findings of the function, together with the status of management’s implementation of recommendations. The Audit Committee has unrestricted access to all internal audit reports, should it wish to review them.
During the year, the team completed 53 risk based audits and reviews across the Group. These included Financial and Operational Market based reviews covering the EMEA, APAC, US and Latam Regions; Global Operations, including manufacturing and supply chain; IT and various Programme Assurance reviews ranging from the implementation of SAP across the North American Business to MDR preparedness. Key issues noted during reviews included the need to better control user access to some systems, also the need to improve internal controls in a number of smaller markets. Management has taken swift action to implement Internal Audit’s recommendations.
A periodic review of the Internal Audit function is undertaken by an independent external consultant, in accordance with the guidelines of the Institute of Internal Auditors. In 2018 KPMG completed an ISA 610 review of the function and this concluded satisfactorily. Finally, the performance of the function is assessed using a structured questionnaire, allowing Non Executive and Executive and senior management, plus the external auditor, to comment on key aspects of the function’s performance. The Audit Committee, which re-approved the function’s charter in December 2018, has satisfied itself that adequate, objective internal audit standards and procedures exist within the Group and that the Internal Audit function is effective.
RISK MANAGEMENT PROGRAMME
Whilst the Board is responsible for ensuring oversight of strategic risks relating to the Company, determining an appropriate level of risk appetite, and monitoring risks through a range of Board and Board Committee processes, the Audit Committee is responsible for ensuring oversight of the processes by which operational risks, relating to the Company and its operations are managed and for reviewing financial risks and the operating effectiveness of the Group’s Risk Management process.
During the year, we reviewed our Risk Management processes and progress was discussed at our meetings in February, July, September and December. We approved the Risk Management Programme for 2018 and monitored performance against that plan specifically reviewing the work undertaken by the risk champions across the Group, identifying the risks which could impact their areas of our business.
The Risk Management programme in 2018 followed the new risk management policy and manual rolled out across the Company in 2017. This programme combines a “bottom up” approach (whereby risks are identified within business areas by local risk champions working with their leadership teams), with a top-down approach (when the Executive Committee meets as the Risk Committee to consider the risks facing the Group at an enterprise level).
Throughout the year, the Audit Committee maintained oversight of this programme. We reviewed the principal risks identified and the heat maps prepared by management showing how these risks were being managed. We considered those risks where the risk profile was changing particularly political and economic risks as a consequence of Brexit. We also reviewed the deep dives undertaken by the Group Risk team, which looked more closely at the risks impacting certain business areas.
|
82 |
Governance |
Smith & Nephew Annual Report 2018 |
|
AUDIT COMMITTEE REPORT continued
Since the year end, we have reviewed a report from the Group Head of Internal Audit into the effectiveness of the Risk Management Programme throughout the year. We considered the principal risks, the actions taken by management to review those risks and the Board risk appetite in respect of each risk.
We concluded that the Risk Management process during 2018 and up to the date of approval of this Annual Report was effective. Work will continue in 2019 and beyond to continue to enhance the process.
See pages 40–49 for further information on our Risk Management Process.
VIABILITY STATEMENT
We also reviewed management’s work in conducting a robust assessment of those risks which would threaten our business model and the future performance or liquidity of the Company, including its resilience to the threats of viability posed by those risks in severe but plausible scenarios. This assessment included stress and sensitivity analyses of these risks to enable us to evaluate the impact of a severe but plausible combination of risks. We then considered whether additional financing would be required in such eventualities. Based on this analysis, we recommended to the Board that it could approve and make the Viability Statement on pages 50‑51.
GOING CONCERN
The Group’s business activities, together with the factors likely to affect its future development, performance and position are set out in the ‘Financial review and principal risks’ section on pages 42‑47.
The financial position of the Group, its cash flows, liquidity position and borrowing facilities are described on page 38‑39.
In addition, the Notes to the Group accounts include the Group’s objectives, policies and processes for managing its capital; its financial risk management objectives; details of its financial instruments and hedging activities; and its exposure to credit risk and liquidity risk.
The Group has considerable financial resources and its customers and suppliers are diversified across different geographic areas. As a consequence, the Directors believe that the Group is well placed to manage its business risk successfully despite the ongoing uncertain economic outlook.
The Directors have a reasonable expectation that the Group has adequate resources to continue in operational existence for the foreseeable future. Thus they continue to adopt the going concern basis for accounting in preparing the annual financial statements.
Management also believes that the Group has sufficient working capital for its present requirements.
EVALUATION OF INTERNAL CONTROLS
Management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rule 13a–15(f) and 15d–15(f) under the US Securities Exchange Act of 1934.
There is an established system of internal control throughout the Group and our country business units. The main elements of the internal control framework are:
|
- |
The management of each country and group function is responsible for the establishment, maintenance and review of effective financial controls within their business unit or function. |
|
- |
The Group’s IT organisation is responsible for the establishment of effective IT controls within the core financial systems and underlying IT infrastructure. |
|
- |
The Financial Controls & Compliance Group has responsibility for the review of the effectiveness of controls operating in the countries, functions and IT organisation, either by performing testing directly or reviewing testing performed in-country. |
|
- |
The Group Finance Manual sets out financial and accounting policies. The Group’s Minimum Acceptable Practices (MAPs) have been further enhanced during 2018 by simplifying and clarifying the requirements as well as broadening their scope and incorporating the core financial controls. The business is required to self-assess their level of compliance with the MAPs twice a year and remediate any gaps. MAPs compliance is validated through spot-checks conducted by the Financial Controls & Compliance Group and during both Internal Audit and external audit visits. |
|
- |
There are clearly defined lines of accountability and delegations of authority. |
|
- |
During the year, there has been further progress in standardising our core financial controls globally and merging with the MAPs. In 2019, there will be a focus on implementing a technology solution to facilitate the operation and testing of controls. |
|
- |
The Internal Audit function executes a risk-based annual work plan, as approved by the Audit Committee. |
|
- |
The Audit Committee reviews reports from Internal Audit on their findings on internal financial controls, including compliance with MAPs and from the SVP Group Finance and the heads of the Financial Controls & Compliance, Taxation and Treasury functions. |
|
- |
The Audit Committee reviews regular reports from the Financial Controls & Compliance Group with regard to compliance with the Sarbanes-Oxley Act including the scope and results of management’s testing and progress regarding any remediation, as well as the aggregated results of MAPs self-assessments performed by the business. |
|
- |
Business continuity planning, including preventative and contingency measures, back-up capabilities and the purchase of insurance. |
|
- |
Risk management policies and procedures including segregation of duties, transaction authorisation, monitoring, financial and managerial review and comprehensive reporting and analysis against approved standards and budgets. |
|
|
Smith & Nephew Annual Report 2018 |
|
83 |
|
- |
A treasury operating framework and Group treasury team, accountable for all treasury activities, which establishes policies and manages liquidity and financial risks, including foreign exchange, interest rate and counterparty exposures. Treasury policies, risk limits and monitoring procedures are reviewed regularly by the Audit Committee, or the Finance and Banking Committee, on behalf of the Board. |
|
- |
Our published Group tax strategy which details our approach to tax risk management and governance, tax compliance, tax planning, the level of tax risk we are prepared to accept and how we deal with tax authorities, which is reviewed by the Audit Committee on behalf of the Board. |
|
- |
The Audit Committee reviews the Group whistle-blower procedures to ensure they are effective. |
|
- |
The Audit Committee received and reviewed a report on the progress of the Finance Transformation during 2018 and the mitigation of the associated risks. |
This system of internal control has been designed to manage rather than eliminate material risks to the achievement of our strategic and business objectives and can provide only reasonable, and not absolute, assurance against material misstatement or loss. Because of inherent limitation, our internal controls over financial reporting may not prevent or detect all misstatements. In addition, our projections of any evaluation of effectiveness in future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. Entities where the Company does not hold a controlling interest have their own processes of internal controls similar to those of the Company.
We have reviewed the system of internal financial control and satisfied ourselves that we are meeting the required standards both for the year ended 31 December 2018 and up to the date of approval of this Annual Report. No concerns were raised with us in 2018 regarding possible improprieties in matters of financial reporting.
This process complies with the Financial Reporting Council’s ‘Guidance on Risk Management, Internal Control and Related Financial and Business Reporting’ on the UK Corporate Governance Code and additionally contributes to our compliance with the obligations under the Sarbanes-Oxley Act and other internal assurance activities. There has been no change during the period covered by this Annual Report that has materially affected, or is reasonably likely to materially affect, the Group’s internal control over financial reporting.
The Board is responsible overall for reviewing and approving the adequacy and effectiveness of the risk management framework and the system of internal controls over financial, operational (including quality management and ethical compliance) processes operated by the Group. The Board has delegated responsibility for this review to the Audit Committee. The Audit Committee, through its Internal Audit function, reviews the adequacy and effectiveness of internal control procedures and identifies any significant weaknesses and ensures these are remediated within agreed timelines. The latest review covered the financial year to 31 December 2018 and included the period up to the approval of this Annual Report. The main elements of this review are as follows:
|
- |
The Chief Executive Officer and the Chief Financial Officer evaluated the effectiveness of the design and operation of the Group’s disclosure controls and procedures as at 31 December 2018. Based upon this evaluation, the Chief Executive Officer and Chief Financial Officer concluded on 21 February 2019 that the disclosure controls and procedures were effective as at 31 December 2018. |
|
- |
Management is responsible for establishing and maintaining adequate internal control over financial reporting. Management assessed the effectiveness of the Group’s internal control over financial reporting as at 31 December 2018 in accordance with the requirements in the US under section 404 of the Sarbanes-Oxley Act. In making that assessment, they used the criteria set forth by the Committee of Sponsoring Organisations of the Treadway Commission in Internal Control-Integrated Framework (2013). Based on their assessment, management concluded and reported that, as at 31 December 2018, the Group’s internal control over financial reporting was effective based on those criteria. Having received the report from management, the Audit Committee reports to the Board on the effectiveness of controls. KPMG, an independent registered public accounting firm issued an audit report on the Group’s internal control over financial reporting as at 31 December 2018. |
CODE OF ETHICS FOR SENIOR FINANCIAL OFFICERS
We have adopted a Code of Ethics for Senior Financial Officers, which applies to the Chief Executive Officer, the Chief Financial Officer, the SVP Group Finance and the Group’s senior financial officers. There have been no waivers to any of the Code’s provisions nor have there been any substantive amendments to the Code during 2018 or up until 15 February 2019. A copy of the Code of Ethics for Senior Financial Officers can be found on our website at www.smith-nephew.com.
In addition, every individual in the finance function certifies to the Chief Financial Officer that they have complied with the Finance Code of Conduct.
EVALUATION OF COMPOSITION, PERFORMANCE AND EFFECTIVENESS OF THE AUDIT COMMITTEE
The composition, performance and effectiveness of the Audit Committee was evaluated this year in accordance with the EU Audit Reform. Its effectiveness is also reviewed in conjunction with the annual Board evaluation, conducted by Boardroom Review.
The review by the Audit Committee found the following and the below action will be taken during 2019:
|
Composition |
Following retirement of Ian Barlow an additional member with recent and relevant financial experience will be required |
|
Performance & Effectiveness |
The Audit Committee is well chaired, with a clear role, an efficient use of time and high quality information |
|
84 |
Governance |
Smith & Nephew Annual Report 2018 |
|
|
Directors’ Remuneration Report Remuneration Committee report |
|||
|
Our focus in 2019 will be on aligning our remuneration arrangements to the new Strategic Imperatives of the Company |
|||
|
|
|
|
|
|
MEMBERSHIP |
|
|
|
|
Member from |
Meetings attended |
|
Angie Risley1 (Chair) |
September 2017 |
5/5 |
|
Vinita Bali |
April 2015 |
5/5 |
|
Virginia Bottomley |
April 2014 |
5/5 |
|
Robin Freestone |
September 2015 |
5/5 |
|
Joseph Papa1 |
April 2011 |
2/2 |
|
Roberto Quarta |
April 2014 |
5/5 |
|
1 Angie Risley was appointed Chair of the Committee on 12 April 2018 replacing Joseph Papa, on his retirement from the Board. |
||
DEAR SHAREHOLDER,
It is a great pleasure to be writing to you for the first time as Chair of the Remuneration Committee. I should like to take the opportunity of thanking my predecessor, Joseph Papa, who was a great support to me during our handover process. The recruitment of Namal Nawana as our new Chief Executive Officer and the consequent changes to his leadership team have given the Committee a full programme during the year. We have also considered the expansion of the role of the Remuneration Committee in the light of revised remuneration regulations and corporate governance reforms. At all times, we have sought to ensure that our pay arrangements support and drive delivery of the strategic aims set out by the Company, while making careful decisions to align pay outcomes with the performance delivered during the period.
Review of 2018 Performance
During the year, the Group delivered underlying revenue growth of 2% and a 90bps improvement in trading profit margin, in line with guidance. Performance improved over the course of the year, with 1% underlying revenue growth in the first half and 3% in the second half. Highlights included our continued strength in the Emerging Markets, with China delivering double-digit growth. At a franchise level, our Knee Implants franchise continued to deliver market-beating growth and Hip Implants improved markedly in the second half. In Sports Medicine REGENETEN for shoulder repair, acquired in December 2017, had an outstanding year as we more than doubled sales and the acquisition of Ceterix, completed in early 2019, offers exciting opportunities in knee repair. Our performance in Advanced Wound Care and Advanced Wound Devices in the US also stood out across 2018. The team delivered these successes whilst controlling costs. This contributed to the margin growth, which also included a gain from a one-off legal settlement. Most impressively, the significant change in structure and leadership in the second half did not detract from delivery and the stronger finish to the year.
Pay for performance is important to us and therefore members of the Audit Committee joined us to consider our results and determine the extent to which performance against the targets in our incentive plans were met. Taking into account this financial performance along with consideration of how our Executive Directors performed against their business performance objectives, the Remuneration Committee determined that Namal Nawana would receive a cash incentive payment of 67.7% of his full year’s salary and an Equity Incentive award of 35.9% of his full year’s salary (prorated to reflect his joining date in May 2018) and that Graham Baker would receive a cash incentive payment of 95.5% of his salary and an Equity Incentive award of 50% of his salary (pro-rated to reflect his increase in salary from July 2018 to reflect increased responsibilities). The Committee also determined that Olivier Bohuon would receive a cash incentive payment of 94% of his salary in respect of the period worked as Chief Executive Officer to May 2018, when he retired from the Board. Reviewing the performance of the Company, the Committee was confident that these outcomes appropriately reflected our underlying performance over the year as a whole.
The Committee also reviewed performance over the past three years and determined that 93% of the target Performance Share Plan awards made in 2016 to Olivier Bohuon would vest reflecting the performance of the Company as a whole. Namal Nawana and Graham Baker were not employed by the Company at the time these awards were made.
The total remuneration paid to Namal Nawana, Olivier Bohuon and Graham Baker in 2018 is detailed further on page 89.
Looking forward – remuneration arrangements for 2019
As explained on pages 8–11, the Board approved a new strategy during 2018 and Namal articulated his vision for the business externally earlier in 2019. During the course of 2019 our focus will be on developing remuneration arrangements to align with this strategy and to drive the performance and behaviours to deliver that strategy. We shall engage with shareholders during the course of 2019 to ensure firstly that shareholders understand our business strategy and secondly how we intend to align our remuneration
|
|
Smith & Nephew Annual Report 2018 |
|
85 |
arrangements to that strategy. We shall ensure that shareholder views are appropriately reflected in the Remuneration Policy we submit at the 2020 Annual General Meeting.
Looking forward – UK corporate governance code
We are mindful of the corporate governance changes which have come into effect at the beginning of 2019. During 2018, we discussed how we would be addressing these changes. We have updated our Terms of Reference to expand our remit to consider pay and benefits across the Company in more depth and breadth than before. The review of our Remuneration Policy in 2019 will also consider the new provisions of the UK Corporate Governance Code.
Although we are not required to report on the CEO pay ratio until next year, we have compared the pay of our outgoing Chief Executive Officer, Olivier Bohuon and our new Chief Executive Officer, Namal Nawana to the median pay in the UK and determined that our CEO pay ratio is 95:1. Further details are given on page 102.
Appointment of Namal Nawana
We determined the remuneration arrangements for Namal Nawana, our incoming Chief Executive Officer, having regard both to the remuneration policy approved by shareholders in 2017 and also to the competitive market environment for Medical Devices Chief Executives of Namal’s calibre and experience, most of whom are based in the US. We are very grateful for the support and guidance we received from the shareholders we spoke to when we were considering appropriate remuneration arrangements.
His remuneration arrangements are described more fully in the following pages. Namal is employed on a standard US executive contract. He receives a base salary of $1,540,000 and participates in the Annual Incentive Plan (cash and equity) and the Performance Share Plan. He also participates in the retirement plans available to our US Executives: Executive Plus Plan, 401k and 401k plus. The Company contribution to these plans is: 20% of base salary to the Executive Plus Plan, standard company match for 401k and standard 401k plus contribution up to the IRS maximum. He will be required to give six months’ notice and the notice period from the Company is 12 months. He received no sign-on or buy-out award on joining the Company.
Executive officer remuneration
We also considered and approved remuneration arrangements for a number of Executive Officers who have moved into new roles or joined the Company as part of Namal’s review of the leadership team and organisational structure. In particular, we consulted shareholders over the summer with regard to increasing our Chief Financial Officer, Graham Baker’s base salary by 5% to reflect his increased responsibilities for our IT and Global Business Services functions. We are grateful to those shareholders who responded to this consultation and were overwhelmingly supportive. We reached out to the holders of over 40% of our share capital and received responses from nearly half of these shareholders, all of whom were supportive.
Retirement of Olivier Bohuon
We also determined the retirement arrangements for Olivier Bohuon, our former Chief Executive Officer, in line with the Remuneration Policy approved by our shareholders in 2017.
Olivier stepped down from the Board on Namal’s appointment in May 2018 and remained employed in an advisory capacity supporting the transition for a period of six months, during which time he continued to receive the same salary and benefits as in 2017. He participated in the Annual Incentive Plan for the period worked as Chief Executive Officer in 2018, but did not receive a 2018 award under the Performance Share Plan. As a good leaver, his Equity Incentive Awards vested on his retirement date on 7 November 2018, and his Performance Share Awards have been pro-rated for length of time served since the date of award and will vest subject to the original performance conditions on their original vesting dates in 2019 and 2020. Additionally, his 2017 award remains subject to a two-year post vesting holding period.
2018 Annual general meeting
We were pleased that our Remuneration Report received over 97% of votes in favour at the 2018 Smith & Nephew plc AGM. This demonstrates the strong support from our shareholders for our remuneration arrangements.
I should like to thank the shareholders who have engaged with us and supported us during the year and particularly those I met as part of my induction programme.
I should also like to thank my fellow Committee members for their support during the year and to welcome Deloitte as our new remuneration advisors.
LOOKING FORWARD
In summary, our focus for 2019 includes:
|
> |
Developing a new Remuneration Policy to align with the new strategy to deliver the performance and behaviours required to deliver the results. |
|
|
|
|
> |
Engaging with key shareholders whilst developing a new Remuneration Policy to ensure alignment of views. |
|
|
|
|
> |
Continuing to develop our understanding and oversight of pay arrangements across the Group focusing particularly on the CEO pay ratio and the gender pay ratio. |
Angie Risley
Chair of the Remuneration Committee
The Terms of Reference of the Remuneration Committee describe the role and responsibilities more fully and can be found on our website at www.smith-nephew.com.
|
86 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration Committee report continued
MEASURES IN OUR VARIABLE PAY PLANS
|
FINANCIAL MEASURES IN ANNUAL INCENTIVE PLAN |
|
|
Revenue (35%) |
Revenue is a key driver of profit growth. |
|
Trading Profit Margin (25%) |
Trading profit margin is a critical measure both for the business and our shareholders and delivering margin improvements is a core commitment under our strategy. |
|
Trading Cash Flow (15%) |
Cash flow from our Established Markets is necessary in order to fund growth in Emerging Markets, innovation, organic growth and acquisitions. |
|
BUSINESS OBJECTIVES IN ANNUAL INCENTIVE PLAN |
|
|
|
|
|
Growth (8.3%) |
Revenue growth through achieving the full potential of our portfolio transforming our business through enabling technologies and expanding in high growth segments is fundamental to our future success. |
|
Business Process (8.3%) |
We need to release resources from the businesses through improved structures, efficiencies and business processes in order to re-invest in our higher growth areas, including Emerging Markets, innovation, organic growth and acquisitions. |
|
People (8.3%) |
We need to attract and retain the right people to achieve our strategy through improving our operating model and drive the right behaviours for all of our people globally. |
|
PERFORMANCE MEASURES IN OUR PERFORMANCE SHARE PLAN |
|
|
|
|
|
Relative TSR (25%) |
If we execute our strategy successfully, this will lead to an increased return for our shareholders, whether you invest in the healthcare sector or in the FTSE. |
|
Cumulative Free Cash Flow (25%) |
Cash flow from our Established Markets is necessary in order to fund growth in Emerging Markets, innovation, organic growth and acquisitions. |
|
Sales Growth (25%) |
Sales growth is a key driver of profit growth. |
|
Return on Invested Capital (25%) |
Return on invested capital is a high priority for our shareholders which will drive better financial discipline and enhanced operating performance. |
|
COMPLIANCE STATEMENT We have prepared this Directors’ Remuneration Report (the Report) in accordance with The Enterprise and Regulatory Reform Act 2012‑2013 (clauses 81‑84) and The Large and Medium-Sized Companies and Groups (Accounts and Reports) (Amendment) Regulations 2013 (the Regulations). The Report also meets the relevant requirements of the Financial Conduct Authority (FCA) Listing Rules. The first part of the Report (pages 89 to 105) is the annual report on remuneration (the Implementation Report). The Implementation Report will be put to shareholders for approval as an advisory vote at the Annual General Meeting on 11 April 2019. The Implementation Report explains how the Remuneration Policy was implemented during 2018 and also how it is currently being implemented in 2019. The second part of the Report (pages 106 to 114) is the Directors’ Remuneration Policy Report (the Policy Report) which was approved by shareholders at the Annual General Meeting held in April 2017. The Policy Report describes our Remuneration Policy as it relates to the Directors of the Company. All payments we make to any Director of the Company will be in accordance with this Remuneration Policy. This Policy remains unchanged in 2019 and it is intended that it will next be put to shareholder vote at the Annual General Meeting to be held in 2020. |
|
|
Smith & Nephew Annual Report 2018 |
|
87 |
The Remuneration Committee presents the Annual Report on remuneration (the Implementation Report), which will be put to shareholders for an advisory vote at the Annual General Meeting to be held on 11 April 2019.
The terms of reference of the Remuneration Committee describe our role and responsibilities more fully and can be found on our website: www.smith-nephew.com.
WORK OF THE REMUNERATION COMMITTEE IN 2018
In 2018, we held five meetings and determined ten matters by written resolution, mainly relating to Executive Officer Remuneration and termination packages and the appointment of Deloitte LLP as new Remuneration Committee advisors. Each meeting was attended by all members of the Committee. The Chief Executive Officer, the Chief Human Resources Officer and the SVP Global Reward, key members of the finance function and the Company Secretary also attended all or part of some of the meetings, except when their own remuneration was being discussed. We also met with the independent Remuneration Consultants, Willis Towers Watson, in the first part of the year and Deloitte in the second half of the year without management present. Our programme of work in 2018 can be found in the table on the next page.
Since year end, we have also reviewed the financial results for 2018 against the targets under the short-term and long-term incentive arrangements jointly with the Audit Committee, and have agreed the targets for the short-term and long-term incentive plans for 2019. We have also approved increases to the salaries of Executive Directors and Executive Officers and determined cash payments under the Annual Incentive Plan, awards under the Equity Incentive Programme and the Performance Share Programme, and the vesting of awards under the Performance Share Programme granted in 2016. Finally, we approved the wording of this Directors’ Remuneration Report.
INDEPENDENT REMUNERATION COMMITTEE ADVISORS
During the year, the Remuneration Committee received information and advice from both Willis Towers Watson and Deloitte LLP. Both firms are independent executive remuneration consultancy firms appointed by the Remuneration Committee following a full tender process in 2011 and 2018 respectively. Deloitte LLP replaced Willis Towers Watson as our primary Remuneration advisors in July 2018, although Willis Towers Watson continue to provide market benchmark data on compensation design and levels for our Executive Director and Executive Officer positions. During the year, both firms provided advice on market trends and remuneration issues in general, attended Remuneration Committee meetings, assisted in the review of the Directors’ Remuneration Report, undertook calculations relating to the TSR performance conditions, advised on Executive Director and Officer pay and investor views and engagement.
The fees paid to Deloitte LLP for Remuneration Committee advice during 2018, charged on a time and expense basis, were £68,200 and the fees paid to Willis Towers Watson were £91,000. Deloitte LLP also provided other tax and consultancy services to the Company. Willis Towers Watson also provided other human resources and compensation advice to the Company for the level below the Board. Both Deloitte LLP and Willis Towers Watson comply with the Code of Conduct in relation to Executive Remuneration Consulting in the United Kingdom and the Remuneration Committee is satisfied that their advice is objective and independent.
|
88 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
|
|
Late January |
Early February |
July |
September |
October |
|
|
Approval of salaries, awards and payouts in 2018 |
Approval of targets and Remuneration strategy for 2018 |
Mid-year Review of Remuneration |
Review of Graham Baker’s base pay |
Review of |
|
DETERMINATION OF REMUNERATION POLICY AND PACKAGES |
|||||
|
Determination of Remuneration Policy for Executive Directors and senior executives. Approval of individual remuneration packages for Executive Directors and Executive Officers, at least annually, and any major changes to individual packages throughout the year. Consideration of remuneration policies and practices across the Group in particular relating to CEO Pay ratio and Gender Pay. Approval of appropriate performance measures for short-term and long-term incentive plans for Executive Directors and senior executives. Determination of pay-outs under short-term and long-term incentive plans for Executive Directors and senior executives.
|
Approved quantum of cash payments and awards to Executive Directors and Officers under the Annual Incentive Plan, the Equity Incentive Programme and Performance Share Programme, (in the context of 2017 financial performance). Reviewed the fees for the Chair, Executive Directors and Officers. |
Agreed the targets for the short-term and long-term incentive plans for 2018. Approved the remuneration strategy for 2018 against the proposed business plan. Considered appropriate remuneration package for new Chief Executive Officer. Reviewed Chair’s pay. |
Report from Deloitte on current market trends in remuneration matters and an analysis of how the Company’s remuneration arrangements aligned with current practices elsewhere. Reviewed the performance of long-term awards granted in 2016, 2017 and 2018. |
Approved 5% base salary increase for Graham Baker to reflect his increased responsibilities for the IT and Global Business Services functions following consultation programme with the holders of 40% of the Company’s shares. |
Agreed to update the Company’s Remuneration Policy to align with the new corporate strategy during 2019 to put to shareholders for approval in 2020. Agreed to make minor adjustments to certain targets and measures in 2019 in line with the existing remuneration Policy. Reviewed benchmarking data for the Executive Directors and Executive officers prepared in accordance with agreed methodology. Considered reports on Gender Pay Gap. |
|
OVERSIGHT OF ALL COMPANY SHARE PLANS |
|||||
|
Determination of the use of long-term incentive plans and overseeing the use of shares in executive and all‑employee plans. |
|
|
Reviewed adherence to shareholding guidelines for Directors and Senior Management. Monitored dilution limits and the number of shares available for use in respect of Executive and all-employee share plans. Approved amendments to various share plan rules to reflect regulatory changes. |
|
|
|
REPORTING AND ENGAGEMENT WITH SHAREHOLDERS ON REMUNERATION MATTERS |
|||||
|
Approval of the Directors’ Remuneration Report ensuring compliance with related governance provisions. Continuation of constructive engagement on remuneration matters with shareholders. |
|
Approved the |
Reviewed the shareholder response to the Remuneration Report at the AGM and noted feedback. |
|
Reviewed the shareholder response to the Remuneration Report at the AGM and noted feedback. |
|
OTHER MATTERS |
|||||
|
|
Audit Committee in attendance to answer questions related to audited numbers and provide assurance. |
|
Confirmed the appointment of Deloitte LLP as the new independent advisors to the Committee. |
|
Discussed recent corporate governance changes and the impact they would have on the Remuneration Committee. |
|
|
Smith & Nephew Annual Report 2018 |
|
89 |
Remuneration implementation report
SINGLE TOTAL FIGURE ON REMUNERATION
The amounts for 2018 have been converted into US$ for ease of comparability using the exchange rates of £ to US$1.334 and € to US$1.180 (2017: £ to US$1.2877 and € to US$1.1279).
|
|
|
Namal Nawana Appointed 7 May 2018 |
|
Olivier Bohuon
Appointed 1 April 2011 |
|
|
Graham Baker Appointed 1 March 2017 |
|
Julie Brown
Appointed 4 February 2013 |
|||||||||||||||
|
|
|
|
2018 |
|
|
2017 |
|
|
2018 |
|
|
2017 |
|
|
2018 |
|
|
2017 |
|
|
2018 |
|
|
2017 |
|
FIXED PAY |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Base salary |
|
$ |
1,006,923 |
|
|
– |
|
$ |
490,615 |
|
$ |
1,330,347 |
|
$ |
707,628 |
|
$ |
547,273 |
|
|
– |
|
$ |
21,606 |
|
Pension payments |
|
$ |
222,010 |
|
|
– |
|
$ |
147,184 |
|
$ |
399,104 |
|
$ |
212,302 |
|
$ |
164,182 |
|
|
– |
|
$ |
6,482 |
|
Taxable benefits |
|
$ |
59,754 |
|
|
– |
|
$ |
44,322 |
|
$ |
177,433 |
|
$ |
26,758 |
|
$ |
22,308 |
|
|
– |
|
$ |
637 |
|
ANNUAL VARIABLE PAY |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
Annual Incentive Plan – cash |
|
$ |
1,042,655 |
|
|
– |
|
$ |
455,345 |
|
$ |
1,208,911 |
|
$ |
676,025 |
|
$ |
683,797 |
|
|
– |
|
|
– |
|
HYBRID |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Annual Incentive Plan – equity |
|
$ |
552,290 |
|
|
– |
|
|
– |
|
$ |
665,173 |
|
$ |
353,817 |
|
$ |
361,200 |
|
|
– |
|
|
– |
|
LONG-TERM VARIABLE PAY |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
Performance Share Plan |
|
|
|
|
|
– |
|
$ |
1,193,678 |
|
$ |
1,335,721 |
|
|
– |
|
|
– |
|
|
– |
|
|
– |
|
Total |
|
$ |
2,883,632 |
|
|
|
|
$ |
2,331,144 |
|
$ |
5,116,689 |
|
$ |
1,976,530 |
|
$ |
1,778,760 |
|
|
|
|
$ |
28,725 |
|
Base salary |
the actual salary receivable for the year. |
|
|
Pension payments |
the value of the salary supplement in lieu of pension or contribution to any pension scheme made by the Company. |
|
|
Taxable benefits |
the gross value of all taxable benefits (or benefits that would be taxable in the UK) received in the year. |
|
|
Annual Incentive Plan – cash |
the value of the cash incentive payable for performance in respect of the relevant financial year. |
|
|
Annual Incentive Plan – equity |
the value of the equity element awarded in respect of performance in the relevant financial year, but subject to an ongoing performance test as described on pages 94–95 of this report. |
|
|
Performance Share Plan |
the value of shares vesting that were subject to performance over the three-year period ending on 31 December in the relevant financial year. For awards vesting in early 2019 this is based on an estimated share price of 1,380.98p per share, which was the average price of a share over the last quarter of 2018. The value of the 2015 share awards that vested in 2018 have now been restated with the share price on the date of actual vesting being 1,325.65p per share on 9 March 2018. |
|
|
Total |
the sum of the above elements. |
All data is presented in our reporting currency of US$. Amounts for Olivier Bohuon have been converted from EURO and amounts for Julie Brown and Graham Baker from GBP using average exchange rates. Given currency volatility in 2018, this may give the impression of changes that are misleading. Data is presented in local currency in the subsequent sections in the interests of full transparency.
|
90 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
FIXED PAY
Base salary
In February 2018, it was agreed that with effect from 1 April 2018, Executive Directors would be paid the following base salaries per annum.
|
|
|
2018 |
|
2017 |
|
Olivier Bohuon (retired from Board on 7 May 2018 and from Company on 7 November 2018) |
|
€1,179,490 |
|
€1,179,490 |
|
Graham Baker |
|
£520,000 |
|
£510,000 |
Namal Nawana was appointed Chief Executive Officer on 7 May 2018 and paid a base salary of $1,540,000 per annum.
After a period of consultation with shareholders, Graham Baker’s salary was increased by 5% to £546,210 to take effect from 1 July 2018, when he assumed additional responsibilities for the IT and Global Business Services functions.
In February 2019, we reviewed the base salaries of the Executive Directors, having considered general economic conditions and average salary increases across the rest of the Group, which have averaged at 2.9% in the UK and 3% in the US. The Remuneration Committee has agreed that Namal Nawana’s salary will increase by 2.5% and Graham Baker’s salary will increase by 2% to $1,578,500 and £557,134 respectively with effect from 1 April 2019.
Pension Payments
In 2018, Graham Baker and Olivier Bohuon, until his retirement from the Company on 7 November 2018, received a salary supplement of 30% of their basic salary to apply towards their retirement savings, in lieu of membership of one of the Company’s pension schemes.
Namal Nawana participates in the retirement plans available to our US Executives: Executive Plus Plan, 401k and 401k plus. During 2018, total Company Pension and 401k contributions for Mr Nawana amounted to $222,010 which on an annualised basis is equivalent to 21.34% of salary. Due to the fact that Mr Nawana reached the annual cap on 401k contributions in the period from joining on 7 May 2018, the actual percentage for 2018 equated to 22.05%. For 2019, the combined pension and 401k Company contribution is expected to be less than 21.47% of his base salary.
Benefits
In 2018, our UK based Executive Directors (Olivier Bohuon and Graham Baker) received death in service cover of seven-times basic salary, of which four-times salary is payable as a lump sum, with the balance used to provide for any spouse and dependent persons. Namal Nawana participated in the US Life Assurance Program, which in total is capped at $2 million. They also received health cover for themselves and their families, a car allowance and financial consultancy advice. Olivier Bohuon also received assistance with travel costs between London and Paris. The same arrangements will apply in 2019 for Namal Nawana and for Graham Baker. The following table summarises the value of benefits in respect of 2017 and 2018. Olivier Bohuon and Julie Brown received these benefits until they retired from the Board on 7 May 2018 and 11 January 2017 respectively.
|
|
Namal Nawana |
|
Olivier Bohuon |
|
Graham Baker |
|
Julie Brown |
||||||||
|
|
2018 |
|
2017 |
|
2018 |
|
2017 |
|
2018 |
|
2017 |
|
2018 |
|
2017 |
|
Health cover |
$ 6,635
|
|
– |
|
£2,915 |
|
£17,807 |
|
£1,369 |
|
£1,217 |
|
– |
|
£44 |
|
Car and fuel allowance |
$ 8,467
|
|
– |
|
£5,288 |
|
£15,000 |
|
£17,676 |
|
£14,182 |
|
– |
|
£451 |
|
Financial consultancy advice |
£33,485 |
|
– |
|
£15,733 |
|
£34,204 |
|
£1,020 |
|
£1,925 |
|
– |
|
– |
|
|
– |
|
– |
|
– |
|
€37,736 |
|
– |
|
– |
|
– |
|
– |
|
Travel costs |
– |
|
– |
|
£7,056 |
|
£33,703 |
|
– |
|
– |
|
– |
|
– |
|
Subscriptions |
– |
|
– |
|
£2,245 |
|
£4,023 |
|
– |
|
– |
|
– |
|
– |
|
|
Smith & Nephew Annual Report 2018 |
|
91 |
ANNUAL VARIABLE PAY
Annual Incentive Plan 2018 – cash element
The performance measures and weightings which apply to the cash element of the Annual Incentive Plan are set out in the Remuneration Policy approved by shareholders in 2017 and detailed on page 108.
The weightings of the performance measures and the figures for threshold, target and maximum relating to the financial objectives of the cash element can be summarised as follows:
|
Weighting |
Threshold |
Target |
Maximum |
Actual |
|
|
Revenue |
35% |
$4,887m |
$5,039m |
$5,190m |
$4,960m1 |
|
Trading profit margin |
25% | 22.0% | 22.5% | 22.9% |
22.7%1 |
|
Trading cash flow |
15% |
$886m |
$985m |
$1,083m |
$951m |
1 At constant exchange rates. See page 131.
The Committee determined that this performance fairly reflected the overall performance of the Company during 2018 and therefore resulted in a bonus achievement of 71% of salary in respect of the financial objectives.
|
|
|
Weight |
|
Achieved % of target |
|
Award % of salary |
|
|
Revenue |
|
35 |
% |
74 |
% |
25.9 |
% |
|
Trading profit margin |
|
25 |
% |
129 |
% |
32.3 |
% |
|
Trading cash flow |
|
15 |
% |
83 |
% |
12.5 |
% |
Accordingly, the following amounts have been earned by Namal Nawana and Graham Baker under the cash element of the Annual Incentive Plan in respect of their financial objectives.
|
Namal Nawana (pro–rated to reflect date of joining Company 7 May 2018) |
$ 708,268
|
|
Graham Baker (pro-rated to reflect change to salary from 1 July 2018) |
£374,287 |
The same measures and weightings will apply to the financial measurements of the cash element of the Annual Incentive Plan 2019. For reasons of commercial sensitivity, we are unable to disclose the precise targets now, but they will be disclosed in full in the 2019 Remuneration Report at the time of vesting.
Business Objectives
When setting business objectives for the upcoming year, the Board looks not only at the expected financial performance for the year, but also at the actions it expects the Executive Director to carry out in the year to build a solid foundation for financial performance over the longer term. In reviewing performance against these objectives at the end of the year, the Board is mindful that there is not always a necessary correlation between financial performance and the achievement of business objectives.
The table on page 92 overleaf sets out how Namal Nawana and Graham Baker have performed against the business objectives of People, Business Process and Customers.
|
92 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
|
|
Namal Nawana |
Graham Baker |
|
|
|
People |
|
|
|
|
– Assessed and took action at the senior leadership level designing a new operating model and attracting four new Executive Committee leaders to Smith & Nephew as well as numerous vice presidents. Improved diversity and aligned team in support of the new purpose, culture pillars and strategic imperatives. – Successfully launched new brand purpose – Life Unlimited – culture pillars and winning behaviours. Engaged organisation with more than 40% of employees actively participating in the process. – Implemented an employee engagement platform through the introduction of Company-wide live, interactive broadcast meetings and extended face-to-face engagement opportunities with >20% of global employee base through face-to-face Town Halls, factory and other site meetings. |
– Key accomplishments in 2018 included strengthening Finance leadership team and improving diversity, as well as successfully integrating Global Business Services and IT following transfer of additional responsibilities mid-year. Supported successful CEO transition including assessment of the overall Company and design of the new organisational operating model. |
|
|
|
Business Process |
|
|
|
|
– Completed a robust market and internal organisational assessment as well as external benchmarking to inform the development of a clear strategy resulting in a newly designed operating model including a flatter, franchise-led organisational design with global supporting functions. – Introduced new Company strategy with five imperatives to drive medium and long-term value creation for shareholders with detailed and robust KPIs put in place internally for continuous measurement throughout the relevant period. – Engaged with R&D organisations in all business areas and reprioritised programmes to align with strategy. Personally led robotic programme acceleration and the team exceeded overall target of 80% key product launches delivered to plan. |
– Strong delivery of APEX restructuring programme, at both Group level, with around $60 million of benefits realised and tight control of operating expenditure, and in Finance function with successful insourcing of transaction processing and IT upgrades. Delivered a significantly improved full year trading margin and built robust plans for further multi-year expansion. Additionally, delivered meaningful reduction in trading tax rate¹ for 2018, down to 16.1%. |
|
|
|
Customers |
|
|
|
|
– Demonstrated strong customer focus meeting with hundreds of customers in aggregate including through hospital visits and attending medical education events and industry conferences. – Engaged regularly with key shareholders and investors through face-to-face meetings built around the financial calendar and key investor conferences in London and the United States. – Delivered significant shareholder returns through strong stock out-performance of FTSE 100 index and improved dividend distribution. |
– Regular engagement with shareholders, supporting financial calendar reporting and key investor conferences, as well as management of debt provider and other key financial stakeholders. Delivered significant shareholder returns through strong stock out-performance of FTSE 100 index and improved dividend distribution. |
|
This resulted in a bonus achievement of 33.2% of salary in respect of the business objectives.
|
Weight |
Achieved % of |
Award % of |
|
|
People |
8.33% | 133% | 11.1% |
|
Business Process |
8.33% | 116% | 9.7% |
|
Customers |
8.33% | 150% | 12.5% |
Accordingly, the following amount has been earned by Namal Nawana under the cash element of the Annual Incentive Plan in respect of his business objectives.
|
Namal Nawana (pro-rated to reflect date of joining Company 7 May 2018) |
$ 334,387
|
This resulted in a bonus achievement of 25% of salary in respect of the business objectives.
|
Weight |
Achieved % of |
Award % of |
|
|
People |
8.33% | 100% | 8.33% |
|
Business Process |
8.33% | 100% | 8.33% |
|
Customers |
8.33% | 100% | 8.33% |
Accordingly, the following amount has been earned by Graham Baker under the cash element of the Annual Incentive Plan in respect of his business objectives.
|
Graham Baker (pro-rated to reflect 5% salary increase with effect from 1 July 2018) |
£132,664 |
|
|
Smith & Nephew Annual Report 2018 |
|
93 |
As well as considering the monetary outcome of the formulaic calculation of these awards, the Committee considered whether discretion should be applied to override these formulaic outcomes and concluded that the monetary outcomes were aligned with the financial performance of the Company, individual performance during 2018 and the intention of the Remuneration Policy.
For 2019, the business objectives for the Executive Directors will be; Growth, People, and Business Processes to align with the new Strategic Imperatives. These business objectives will be equally weighted.
The Committee also considered the level of Cash Incentive Payment to be made to Olivier Bohuon who retired as Chief Executive Officer on 7 May 2018. 75% of his Cash Incentive Payment was based on the Financial Objectives, which resulted in a payout of 94% of target as described on page 91. 25% of his Cash Incentive Payment was based on his performance against his Business Objectives: People, Business Process and Customers. In 2018, these objectives were all aligned to ensuring an orderly transition to the new Chief Executive Officer. The Committee considered his performance as Chief Executive Officer against these business objectives for the period up to 7 May 2018 and concluded that he had broadly met these objectives and that therefore the payout in respect of his Business Objectives should be in line with the payout in respect of the overall financial objectives at 94% as follows:
|
Amount |
Award % |
|
|
Financial Objectives (75% of award) |
€289,463 |
94 |
|
Business Objectives (25% of award) |
€96,488 |
94 |
|
Total Cash Incentive Payment |
€385,951 |
1 These non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194–198.
|
94 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
Annual Incentive Plan – equity element
The individual performance of all employees in the Group is assessed on two bases. The first looks at what has been achieved, namely the extent to which the employee has performed against the financial and business objectives set at the beginning of the year. The second looks at how this performance has been achieved, reflecting the right culture and values in accordance with our critical enablers. Against each, the employee is rated as having performed below, in-line or above expectations.
|
Assessment of how Executive Directors have achieved |
||||
|
Below expectations |
In-line with expectations |
Above expectations |
||
|
Assessment of what has been achieved |
Below expectations |
No Award |
No Award |
No Award |
|
In-line with expectations |
No Award |
Award of 50% of Salary |
Award of 55% of Salary |
|
|
Above expectations |
No Award |
Award of 55% of Salary |
Award of 65% of Salary |
|
The Remuneration Committee has considered the performance of Namal Nawana and Graham Baker in exactly the same way as other employees in the Group when determining the level of Equity Incentive Award to be made to them. In assessing their performance against the same financial and business objectives used to determine the level of their cash award, the Remuneration Committee has determined that on the first criterion (assessing what they have achieved) Namal Nawana and Graham Baker have both performed in line with expectations throughout the year. On the second criterion (assessing how they have achieved), the Remuneration Committee has determined that Namal Nawana has exceeded expectations and Graham Baker has performed in line with expectations. These ratings result in an Equity Incentive Award of 55% of salary (pro-rated to 35.9% to reflect his appointment on 7 May 2018) for Namal Nawana and 50% of salary for Graham Baker. In summary, as a result of the financial performance described on page 91 and the individual performance described in the table on page 92, the Remuneration Committee determined that the following awards be made under the Annual Incentive Plan in respect of performance in 2018:
|
Executive Director |
Cash Component |
Equity Component |
||
|
% of salary |
Amount |
% of salary |
Amount |
|
|
Namal Nawana |
67.7% | $ 1,042,655
|
35.9% | $ 552,290
|
|
Graham Baker |
95.5% |
£506,951 |
50.0% |
£265,328 |
These figures are converted into dollars and included under Annual Incentive Plan (cash) and (equity) in the single figure table on page 89.
The precise awards granted in 2019 to Namal Nawana and Graham Baker in respect of service in 2018 will be announced when the awards are made and will be disclosed in the 2019 Annual Report. The Committee also determined that no Equity Incentive Award would be made to Olivier Bohuon who had retired as Chief Executive Officer during the year. As a result of the 2018 performance assessment for Graham Baker, the first tranche of the Equity Incentive Award made in 2018 will vest. Both the grant and vesting of these awards are subject to Graham’s performance discussed on page 92. Namal Nawana was not employed during 2017 and therefore received no Equity Incentive award in 2018.
|
Director |
Date of Grant |
Number of shares |
Number of shares to vest from |
|
Graham Baker |
7 March 2018 |
7,242 | 14,485 |
EQUITY INCENTIVE AWARD FROM PRIOR YEARS
The following Equity Incentive awards held by Olivier Bohuon vested in their entirety on his retirement from the Company on 7 November 2018 in accordance with the plan rules:
|
Director |
Date of Grant |
Number of shares under award |
|
Olivier Bohuon |
7 March 2018 |
41,587 |
|
7 March 2017 |
28,787 | |
|
7 March 2016 |
17,608 |
EQUITY INCENTIVE AWARDS IN 2019
The Equity Incentive Award element will operate in 2019 in exactly the same way as in 2018 and previous years. The Remuneration Committee will assess what has been achieved by the Executive Directors against the same financial and business objectives used to determine the level of their cash awards. The Remuneration Committee will assess how the Executive Directors have achieved their objectives by considering the role played by the Executive Directors in establishing an appropriate culture and set of values throughout the organisation. The level of Equity Incentive Award to be made will be determined according to the matrix above.
|
|
Smith & Nephew Annual Report 2018 |
|
95 |
LONG-TERM VARIABLE PAY
Performance Share Plan
Performance Share Programme – 2018 grants
Performance share awards granted in 2018 were made to Graham Baker and to Namal Nawana on his appointment under the Global Share Plan 2010 to a maximum value of 190% of salary (95% for target performance). As Olivier Bohuon had already indicated his intention to retire at the time the awards were made in 2018, no award was made to him. The four equally weighted performance measures are relative TSR, return on invested capital, sales growth and cumulative free cash flow. These measures are aligned with our financial priorities and strategies. Performance will be measured over the three financial years from 1 January 2018 and awards will vest subject to performance and continued employment in 2021. Sufficient shares will be sold to cover taxation obligations and the Executive Directors will be required to hold the net shares for a further period of two years.
The two equally weighted peer groups against which the Company’s TSR performance will be measured are defined at the start of each performance period based on constituents of the following:
– A sector-based peer group based on those companies classified as the S&P 1200 Global Healthcare subset comprising Medical Devices, equipment and supplies companies (official industry classifications of ‘Health Care Equipment and Supplies, Life Sciences Tools & Services and Health Care Technology’). This is a broader sector-based peer group than in previous years, so that we maintain a focus on outperforming our broad sector without being impacted by the volatility of a smaller group.
– FTSE 100 constituents excluding financial services and commodities companies. This is in response to shareholders who assess our performance not based on sector, but instead based on the index we operate in.
The Group’s TSR performance and its performance relative to the comparator groups is independently monitored and reported to the Remuneration Committee by Deloitte LLP.
Total Shareholder Return (TSR) performance is relative to two separate indices as follows:
|
Award vesting as % of salary at date of grant |
||
|
Relative TSR ranking |
Sector Based Peer Group |
FTSE100 Peer Group |
|
Below median |
Nil |
Nil |
|
Median |
5.9375% |
5.9375% |
|
Upper quartile or above |
23.75% |
23.75% |
Awards will vest on a straight-line basis between these points. If the Company’s TSR performance is below median against both indices, none of this part of the award will vest.
|
96 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
Return on invested capital (ROIC), adds focus on enhancing operating performance and reducing the under-performing asset base. 25% of the award will vest subject to ROIC:
ROIC will be defined as:
|
Net Operating Profit1 less Adjusted Taxes2 |
|
(Opening Net Operating Assets + Closing Net Operating Assets)3 ÷ 2 |
ROIC will be measured each year of the three-year performance period and a simple average of the three years will be compared to the targets below (precise numbers will be included in the Remuneration Report prospectively). The Remuneration Committee will have the discretion to adjust ROIC targets in the case of significant events such as material mergers, acquisitions and disposals and that such adjustment will be consistent with the deal model and approved by the Board at the time of the transaction.
1 Operating profit is as disclosed in the Group income statement in the Annual Report.
2 Adjusted taxes represents our taxation charge per the Group income statement adjusted for the impact of tax on items not included in Operating Profit notably interest income and expense, other finance costs and share of results of associates.
3 Net Operating Assets comprises net assets from the Group balance sheet (Total assets less Total liabilities) excluding the following items: Investments, Investments in associates, Retirement benefit assets and liabilities, Long-term borrowings, Bank overdrafts, borrowings and loans, and Cash at bank.
The awards subject to ROIC will vest as follows:
|
Return on Invested Capital |
Award vesting as % of salary |
|
Below Threshold 11.6% |
Nil |
|
Threshold 11.6% (‑1.25% of target) |
11.875% |
|
Target 12.9% (as derived from the Strategic Plan) |
23.75% |
|
Maximum or above 14.1% (+1.25% of target) |
47.5% |
Awards will vest on a straight-line basis between these points.
Sales growth focuses on growth in both Established Markets and Emerging Markets. 25% of the award will be subject to sales growth and will vest as follows:
|
Sales growth over three-year period commencing 1 January 2018 |
Award vesting as % of salary |
|
Below Threshold |
Nil |
|
Threshold (‑2.7% of target) |
11.875% |
|
Target |
23.75% |
|
Maximum or above (+2.7% of target) |
47.5% |
It is not possible to disclose precise targets for sales growth as this will give commercially sensitive information to our competitors concerning our growth plans and is potentially price sensitive information. This target however will be disclosed in the 2020 Annual Report, when the Committee will discuss performance against the target.
Cumulative free cash flow is defined as net cash inflow from operating activities, less capital expenditure, less the cash flow input of certain adjusted items. Free cash flow is the most appropriate measure of cash flow performance because it relates to cash generated to finance additional investments in business opportunities, debt repayments and distribution to shareholders. This measure includes significant elements of operational financial performance and helps to align Executive Director awards with shareholder value creation.
It is important as it is derived from increased revenues and healthy trading profits. Having a healthy cash flow will enable us to continue to grow and invest. 25% of the award will be subject to cumulative free cash flow performance and will vest as follows:
|
Cumulative free cash flow |
Award vesting as % of salary |
|
Below $1,575m |
Nil |
|
$1,575m (‑13% of target) |
11.875% |
|
$1,810m |
23.75% |
|
$2,046m or more (+13% of target) |
47.5% |
|
|
Smith & Nephew Annual Report 2018 |
|
97 |
Performance Share Programme 2019
Performance share awards will be made in 2019 to the Executive Directors under the Global Share Plan 2010 to a maximum value of 190% of salary (95% for target performance). Performance will be measured over the three financial years commencing 1 January 2019 against the same four equally weighted performance measures as in 2018: relative TSR, return on invested capital, sales growth and cumulative free cash flow. On vesting, sufficient shares will be sold to cover taxation obligations and the Executive Directors will be required to hold the net shares for a further period of two years.
TSR performance will be measured in the same way as in 2018 as described on page 95 against the same two peer groups.
Return on invested capital (ROIC) will be measured in the same way as in 2018, as described on page 96.
The targets will be as follows:
|
Return on Invested Capital |
Award vesting as % of salary |
|
Below Threshold 11.8% |
Nil |
|
Threshold 11.8% (‑1.25% of target) |
11.875% |
|
Target 13.1% (as derived from the Strategic Plan) |
23.75% |
|
Maximum or above 14.3% (+1.25% of target) |
47.5% |
Awards will vest on a straight-line basis between these points.
Sales growth will be measured in the same way as in 2018, as described on page 96. The targets will be as follows:
|
Sales growth over three-year period commencing 1 January 2019 |
Award vesting as % of salary |
|
Below Threshold |
Nil |
|
Threshold (‑2.7% of target) |
11.875% |
|
Target |
23.75% |
|
Maximum or above (+2.7% of target) |
47.5% |
It is not possible to disclose precise targets for sales growth as this will give commercially sensitive information to our competitors concerning our growth plans and is potentially price sensitive information. This target however will be disclosed in the 2021 Annual Report, when the Committee will discuss performance against the target.
Cumulative free cash flow will be measured in the same way as in 2018, as described on page 96. The targets will be as follows:
|
Cumulative free cash flow |
Award vesting as % of salary |
|
Below $1,923m |
Nil |
|
$1,923m (‑13% of target) |
11.875% |
|
$2,210m |
23.75% |
|
$2,497m or more (+13% of target) |
47.5% |
|
98 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
Performance Share Programme 2016
Since the end of the year, the Remuneration Committee has reviewed the vesting of conditional awards made to Executive Directors under the Global Share Plan 2010 in 2016. Vesting of the conditional awards made in 2016 was subject to performance conditions based on TSR, revenue in Emerging Markets and cumulative free cash flow measured over a three-year period commencing 1 January 2016.
25% of the award was based on the Company’s TSR relative to a bespoke group of 12 Medical Devices companies. This group comprised of the following companies: Baxter, Becton Dickinson, Boston Scientific, Coloplast, Conmed, Edwards Life Sciences, Medtronic, NuVasive, Orthofix, Stryker, Wright Medical and Zimmer. The following companies delisted during the period and were therefore removed: Covidien, C R Bard, Nobel Biocare and St Jude Medical. Against this peer group, the Company’s TSR performance ranked below median meaning that this part of the award therefore vested at 0%.
25% of the award was based on revenues in Emerging Markets. The threshold set in 2016 was $2,316 million with a target of $2,725 million. Over the three-year period, the adjusted revenues in Emerging Markets were $2,560 million. These adjustments include translational foreign exchange. This part of the award therefore vested at 20% out of the 25% target.
50% of the award was based on cumulative free cash flow performance. Over the three-year period, the adjusted cumulative free cash flow was $1,929 million which is between target and maximum. These adjustments include items such as Board-approved M&A, including the acquisition of Rotation Medical and the disposal of the Gynaecological business and Board-approved Business Plans such as the APEX programme, the commercial restructuring programme and exceptional expenditure to comply with the EU Medical Devices Regulations. This part of the award therefore vested at 73%.
|
Threshold |
Target |
Maximum |
Actual |
Percentage |
|
|
TSR |
Median |
– |
Upper Quartile |
Below Median |
0% |
|
Emerging Markets Sales |
$2,316m |
$2,725m |
$3,133m |
$2,560m |
20% |
|
Cumulative Free Cash Flow |
$1,585m |
$1,822m |
$2,059m |
$1,929m |
73% |
Overall therefore, the conditional awards made in 2016 will vest at 93% of target (46.5% of maximum) on 7 March 2019 as follows:
|
Director |
Date of grant |
Number of shares under award at maximum |
Number vesting |
|
Olivier Bohuon |
7 March 2016 |
139,396
Pro-rated for length of time held prior to |
64,819 |
As well as considering the monetary outcome of the formulaic calculation of these awards, the Committee considered whether discretion should be applied to override these formulaic outcomes and concluded that the monetary outcomes were aligned with the financial performance of the Company during the performance period and the intention of the Remuneration Policy.
Neither Namal Nawana nor Graham Baker were employed by the Company in 2016 and therefore have no Performance Share Awards to vest on 7 March 2019.
DETAILS OF OUTSTANDING AWARDS MADE UNDER THE PERFORMANCE SHARE PROGRAMME
Details of conditional awards over shares granted to Executive Directors subject to performance conditions are shown below. These awards were granted under the Global Share Plan 2010. The performance conditions and performance periods applying to these awards are detailed on page 95–97.
|
|
Date granted |
Number of ordinary shares under award at maximum |
Date of vesting |
|
Olivier Bohuon |
7 March 2016 |
139,3961,2 |
7 March 2019 |
|
|
7 March 2017 |
97,7441,2 |
7 March 2020 |
|
Graham Baker |
7 March 2017 |
79,166 |
7 March 2020 |
|
|
7 March 2018 |
75,058 |
9 March 2021 |
|
Namal Nawana |
9 May 2018 |
108,800 |
9 May 2021 |
1 Pro-rated to reflect Olivier Bohuon’s retirement from the Company on 7 November 2018.
2 On 5 February 2019, 53.5% of the award granted at maximum to Olivier Bohuon lapsed following completion of the performance period.
|
|
Smith & Nephew Annual Report 2018 |
|
99 |
SUMMARY OF SCHEME INTERESTS AWARDED DURING THE FINANCIAL YEAR
|
|
Namal Nawana1 |
Olivier Bohuon2 |
Graham Baker1 |
|||
|
|
Number of shares |
Face value |
Number of shares |
Face value |
Number of shares |
Face value |
|
Annual Equity Incentive Award (see page 94) |
– |
– |
40,801 |
€589,745 |
21,727 |
£280,500 |
|
Performance Share Award at maximum (see page 98) |
108,800 |
£1,419,311 |
– |
– |
75,058 |
£969,000 |
1 Annual Equity Incentive Awards for 2018 were based on performance for 2017, hence Namal Nawana received no award.
2 Olivier Bohuon did not receive a Performance Share Award in 2018, as he had announced his intention to retire.
Please see Policy Table on pages 108 and 109 for details of how the above plans operate. The number of shares is calculated using the closing share price on the day before the grant, which for the awards granted on 7 March 2018 was 1291p, and for the awards granted on 9 May 2018 was 1304.5p.
SINGLE TOTAL FIGURE ON REMUNERATION
Chair and Non-Executive Directors
|
|
|
Basic annual fee1 |
|
Committee Chairman /Senior |
|
Intercontinental travel fee |
|
|
|
Total |
||||||||||||||
|
Director |
|
2018 |
|
2017 |
|
2018 |
|
2017 |
|
2018 |
|
2017 |
|
2018 |
|
2017 | ||||||||
|
Roberto Quarta |
|
£ |
418,695 |
|
£ |
412,000 |
|
|
– |
|
|
– |
|
|
– |
|
£ |
7,000 |
|
£ |
418,695 |
|
£ |
419,000 |
|
Vinita Bali2 |
|
|
– |
|
£ |
36,750 |
|
|
– |
|
|
– |
|
|
– |
|
£ |
7,000 |
|
|
– |
|
£ |
43,750 |
|
|
|
$ |
129,780 |
|
$ |
59,780 |
|
|
– |
|
|
– |
|
$ |
42,000 |
|
$ |
21,000 |
|
$ |
171,780 |
|
$ |
80,780 |
|
Ian Barlow |
|
£ |
69,500 |
|
£ |
68,135 |
|
£ |
20,000 |
|
£ |
20,000 |
|
|
– |
|
£ |
7,000 |
|
£ |
89,500 |
|
£ |
95,135 |
|
Virginia Bottomley |
|
£ |
69,500 |
|
£ |
68,135 |
|
|
– |
|
|
– |
|
|
– |
|
£ |
7,000 |
|
£ |
69,500 |
|
£ |
75,135 |
|
Roland Diggelmann3 |
|
£ |
59,000 |
|
|
– |
|
|
– |
|
|
– |
|
|
– |
|
|
– |
|
£ |
59,000 |
|
|
– |
|
Erik Engstrom |
|
£ |
69,500 |
|
£ |
68,135 |
|
|
– |
|
|
– |
|
|
– |
|
£ |
7,000 |
|
£ |
69,500 |
|
£ |
75,135 |
|
Robin Freestone |
|
£ |
69,500 |
|
£ |
68,135 |
|
£ |
20,000 |
|
£ |
16,667 |
|
|
– |
|
£ |
7,000 |
|
£ |
89,500 |
|
£ |
91,802 |
|
Michael Friedman |
|
$ |
129,780 |
|
$ |
129,780 |
|
$ |
$ 35,000
|
|
$ |
35,000 |
|
$ |
42,000 |
|
$ |
42,000 |
|
$ |
206,780 |
|
$ |
206,780 |
|
Brian Larcombe4 |
|
|
– |
|
£ |
20,750 |
|
|
– |
|
£ |
1,277 |
|
|
– |
|
|
– |
|
|
– |
|
£ |
22,027 |
|
Marc Owen5 |
|
$ |
129,780 |
|
$ |
30,000 |
|
|
– |
|
|
– |
|
$ |
$ 42,000
|
|
$ |
14,000 |
|
$ |
$ 171,780
|
|
$ |
44,000 |
|
Joseph Papa6 |
|
$ |
44,115 |
|
$ |
129,780 |
|
|
– |
|
$ |
35,000 |
|
$ |
14,000 |
|
$ |
35,000 |
|
$ |
58,115 |
|
$ |
199,780 |
|
Angie Risley7 |
|
£ |
69,500 |
|
£ |
18,173 |
|
£ |
14,172 |
|
|
– |
|
|
– |
|
£ |
7,000 |
|
£ |
83,672 |
|
£ |
25,173 |
|
1 |
The basic annual fee includes shares purchased for the Chairman and Non-Executive Directors in lieu of part of the annual fee, details of which can be found on the table on page 100. |
|
2 |
Vinita Bali elected to receive the payment of her fee in US$ in August 2017 having previously been in GBP. |
|
3 |
Roland Diggelmann was appointed to the Board on 1 March 2018. |
|
4 |
Brian Larcombe retired from the Board with effect from 6 April 2017. |
|
5 |
Marc Owen was appointed to the Board with effect from 1 October 2017. |
|
6 |
Joseph Papa retired from the Board with effect from 12 April 2018. |
|
7 |
Angie Risley was appointed to the Board with effect from 18 September 2017. |
|
100 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
Chair and Non-Executive Director Fees
In February 2019, the Remuneration Committee reviewed the fees paid to the Chairman and determined that with effect from 1 April 2019 the fees paid would remain unchanged. The Board reviewed the fees paid to the Non-Executive Directors and determined that with effect from 1 April 2019, the fees would remain unchanged as follows:
|
Annual fee paid to the Chair |
£420,240 of which £105,060 paid in shares |
|
Annual fee paid to Non-Executive Directors |
£69,500 of which £6,500 paid in shares |
|
Intercontinental travel fee (per meeting) |
£3,500 or $7,000 |
|
Fee for Senior Independent Director and Committee Chairman |
£20,000 or $35,000 |
Payments made to past Directors
Olivier Bohuon retired as Chief Executive Officer of the Board on 7 May 2018 and as an employee of the Company on 7 November 2018 and was paid in accordance with the Remuneration Policy approved by shareholders in 2017 as an employee and the terms of his service agreement. In respect of the transition period, (from 7 May to 7 November 2018) he received salary (€612,427), benefits (£39,472) and a payment in lieu of pension (€176,924). In accordance with the Plan Rules, on his retirement from the Company all unvested Equity Incentive Awards vested in their entirety and the outstanding Performance Share Plan awards were pro-rated for service to 7 November 2018 and will, subject to the performance conditions being satisfactorily met at the end of the three-year performance period, vest on the original vesting dates on the third anniversary of the respective dates of grant. He will be required to retain any vested shares, net of tax, in relation to the 2017 award for a further two-year period after the vesting date. In light of his anticipated retirement, no Performance Share Plan award was made in 2018.
No other payments were made to former Directors in the year.
Payments for loss of office
No payments were made in respect of a Director’s loss of office in 2018.
Service contracts
Executive Directors are employed on rolling service contracts with notice periods of up to 12 months from the Company and six months from the Executive Director. Further information can be found on page 111 of the Policy Report.
Outside directorships
Olivier Bohuon was a Non-Executive Director of Virbac SA and received £7,269 in respect of this appointment up to 7 May 2018. He was also a Non-Executive Director of Shire Plc and received £48,348 in respect of this appointment up to 7 May 2018.
Namal Nawana is a Non-Executive Director of Hologic, Inc. and received $56,231 in respect of this appointment from 7 May 2018 to 31 December 2018.
Directors’ interests in ordinary shares
Beneficial interests of the Executive Directors in the ordinary shares of the Company are as follows:
|
|
Namal Nawana |
|
Graham Baker |
|
Olivier Bohuon |
|||||
|
|
7 May 20181 |
31 December |
15 February 20192 |
|
1 January 2018 |
31 December |
15 February 20192 |
|
1 January 2018 |
7 May 20183 |
|
Ordinary shares |
– |
224,214 | 224,2144 |
|
– |
10,076 | 10,0764 |
|
467,811 | 531,470 |
|
Share options |
– |
– |
– |
|
– |
2,734 | 2,734 |
|
– |
– |
|
Performance share awards5 |
– |
108,800 |
108,800 |
|
79,166 |
154,224 | 154,224 |
|
423,680 | 304,948 |
|
Equity Incentive awards |
– |
– |
– |
|
– |
21,727 | 21,727 |
|
87,956 | 85,305 |
1 Namal Nawana was appointed to the Board on 7 May 2018.
2 The latest practicable date for this Annual Report.
3 Olivier Bohuon retired from the Board on 7 May 2018.
4 The ordinary shares held by Namal Nawana on 15 February 2019 represent 276.41% of his base annual salary and for Graham Baker 28.67% of his base salary.
5 These share awards are subject to further performance conditions before they may vest, as detailed on pages 95 to 97.
The beneficial interest of each Executive Director is less than 1% of the ordinary share capital of the Company.
|
|
Smith & Nephew Annual Report 2018 |
|
101 |
Beneficial interests of the Chair and Non-Executive Directors in the ordinary shares of the Company are as follows:
|
Director |
1 January 2018 (or date of |
31 December 2018 (or date of retirement if earlier) |
15 February 20191 |
Shareholding as % |
|
Roberto Quarta |
28,261 | 32,449 | 32,449 | 112.04 |
|
Vinita Bali4 |
6,836 | 7,154 | 7,154 | 152.12 |
|
Ian Barlow |
19,009 | 19,291 | 19,291 | 402.75 |
|
Virginia Bottomley |
18,714 | 19,024 | 19,024 | 397.18 |
|
Roland Diggelmann5 |
– |
4,867 | 4,867 | 101.61 |
|
Erik Engstrom |
15,547 | 15,796 | 15,796 | 329.78 |
|
Robin Freestone |
15,525 | 15,774 | 15,774 | 329.32 |
|
Michael Friedman4 |
9,910 | 10,212 | 10,212 | 149.26 |
|
Marc Owen |
– |
7,290 | 7,290 | 213.29 |
|
Joseph Papa6 |
13,860 | 13,860 |
N/A |
N/A |
|
Angie Risley |
– |
1,960 | 1,960 | 40.92 |
1 The latest practicable date for this Annual Report.
2 Calculated using the closing share price of 1,451p per ordinary share and $37.97 per ADS on 15 February 2019, and an exchange rate of £1/$1.284687.
3 All Non-Executive Directors in office since 1 January 2018 held the required shareholding during the year except Angie Risley.
4 Vinita Bali, Michael Friedman and Marc Owen hold some of their shares in the form of ADS.
5 Roland Diggelmann was appointed to the Board on 1 March 2018.
6 Joseph Papa retired from the Board on 12 April 2018.
The beneficial interest of each Non-Executive Director is less than 1% of the ordinary share capital of the Company.
Chief Executive Officer remuneration compared to employees generally
The percentage change in the remuneration of the Chief Executive Officer between 2017 and 2018 compared to that of employees generally was as follows:
|
Base salary % change 2018 |
Benefits |
Annual cash bonus |
|
|
Chief Executive Officer1 |
12.6% | 41.3% | 23.9% |
|
Average for all employees |
2.6% |
N/A |
N/A |
1 Amounts paid to Olivier Bohuon up to his retirement on 7 May 2018 and to Namal Nawana after his appointment on the same date.
The average cost of wages and salaries for employees generally increased by 6% in 2018 (see Note 3.1 to the Group accounts). Figures for annual cash bonuses are included in the numbers.
When considering remuneration arrangements for our Executive Directors, the Remuneration Committee takes into account pay across the Group in the following ways:
Salary levels and increases for all employees including Executive Directors take account of the scope and responsibility of position, the skills, experience and performance of the individual and general economic conditions within the relevant geographical market. When considering increases to Executive Director base salaries, the Committee considers the average pay increases in the market where the Executive Director is based.
All employees including the Executive Directors have performance objectives determined at the beginning of the year which cascade down from the Strategic Imperatives for the Group. The level of variable pay determined for all employees, whether in the form of shares or cash is dependent on performance against these imperatives, both financially and personally.
Executive Directors participate in benefits plans and arrangements comparable to benefits paid to other senior executives in the relevant geography.
The Remuneration Committee is keeping under review the level of pension benefits and cash payments in lieu of pensions paid to our Executive Directors, which for historical reasons are currently higher than paid to most employees.
Executive Directors participate in the same senior executive incentive plans (currently the Annual Incentive Programme and the Performance Share Programme) as other Executive Officers and senior executives. The level of award reflect the differing seniority of participants but the same performance conditions apply for all.
|
102 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
Chief Executive Officer Pay Ratio
The Committee has chosen to provide our CEO pay ratio data for 2018, despite the requirement not coming into force until 2019, as we consider that it is important to take a lead in this area.
Our calculations are based on actual pay data for 2018, (in accordance with Option A as set out in the Companies (Miscellaneous Reporting) Regulations 2018) using a combined figure for CEO pay comprising: pay for Olivier Bohuon (Chief Executive Officer until 7 May 2018); and pay for Namal Nawana (Chief Executive Officer from 7 May 2018).
Comparisons have been made with employees at median (P50), lower (P25) and upper (P75) quartiles. The Committee is satisfied that the individuals identified in the employee comparison group appropriately reflect the employee pay profile at those quartiles, and that the overall picture presented by the ratios is consistent with our pay, reward and progression policies for UK employees.
The table below sets out the ratio at the median, lower and upper quartiles:
|
Year |
P25 (lower quartile) |
P50 (median) |
P75 (upper quartile) |
|
2018 |
142:1 |
95:1 |
59:1 |
The table below provides the total pay figure used for each quartile employee, and the salary component within this.
|
Component |
CEO (combined) |
P25 (lower quartile) |
P50 (median) |
P75 (upper quartile) |
|
Salary |
$ 1,497,538
|
$ 32,976
|
$ 51,434
|
$ 83,011
|
|
Total pay |
$ 5,214,776
|
$ 36,597
|
$ 54,923
|
$ 87,956
|
In assessing our pay ratio, the Committee would like to highlight that 2018 reflects a year of change for the CEO role at Smith & Nephew, with the transition from Olivier Bohuon to Namal Nawana in May. Next year we expect the figures to relate solely to remuneration for Namal Nawana.
Relative importance of spend on pay
When considering remuneration arrangements for our Executive Directors and employees as a whole, the Remuneration Committee also takes into account the overall profitability of the Company and the amounts spent elsewhere, particularly in returning profits to shareholders in the form of dividends and share buy backs.
The following table sets out the total amounts spent in 2018 and 2017 on remuneration, the attributable profit for each year and the dividends declared and paid in each year.
|
|
For the year to 31 December 2018 |
For the year to 31 December 2017 |
% change |
|
Attributable profit for the year |
$663m |
$767m |
14% |
|
Dividends paid during the year |
$321m |
$269m |
19% |
|
Share buyback |
$48m1 |
$52m1 |
8% |
|
Total Group spend on remuneration |
$1,330m |
$1,231m |
8% |
1 Shares are bought in the market in respect of shares issued as part of the executive and employee share plans.
|
|
Smith & Nephew Annual Report 2018 |
|
103 |
Total Shareholder Return
A graph of the Company’s TSR performance compared to that of the FTSE 100 index is shown below in accordance with Schedule 8 to the Regulations.

However, as we compare the Company’s performance to a tailored sector peer group of medical devices companies (see page 95), when considering TSR performance in the context of the Global Share Plan 2010, we feel that the following graph showing the TSR performance of this peer group is also of interest.

|
104 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
Remuneration implementation report continued
Table of historic data
The following table details information about the pay of the Chief Executive Officer in the previous ten years:
|
|
Long-term incentive vesting rates against maximum opportunity |
|||||
|
Year |
Chief Executive Officer |
Single figure of total |
|
Annual Cash Incentive |
Performance shares % |
Options % |
|
2018 |
Namal Nawana1 |
$2,883,632 |
|
697 |
N/A |
– |
|
2018 |
Olivier Bohuon2 |
$2,331,144 |
|
637 |
46.5 |
– |
|
2017 |
Olivier Bohuon |
$5,116,6896 |
|
61 | 54 |
– |
|
2016 |
Olivier Bohuon |
$3,332,850 |
|
30 | 8 |
– |
|
2015 |
Olivier Bohuon |
$5,342,377 |
|
75 | 33.5 |
– |
|
2014 |
Olivier Bohuon |
$6,785,121 |
|
43 | 57 |
– |
|
2013 |
Olivier Bohuon |
$4,692,858 |
|
84 |
– |
– |
|
2012 |
Olivier Bohuon |
$4,956,771 |
|
84 |
N/A |
– |
|
2011 |
Olivier Bohuon3,4 |
$7,442,191 |
|
68 |
N/A |
– |
|
2011 |
David Illingworth5 |
$3,595,787 |
|
37 | 27 |
27 |
|
2010 |
David Illingworth |
$4,060,707 |
|
57 | 70 |
61 |
1Appointed Chief Executive Officer on 7 May 2018.
2Retired as Chief Executive Officer on 7 May 2018.
3Appointed Chief Executive Officer on 1 April 2011.
4Includes recruitment award of €1,400,000 cash and a share award over 200,000 ordinary shares with a value of €1,410,000 on grant.
5Resigned as Chief Executive Officer on 1 April 2011.
6Prior years are restated to reflect amounts not known at the date of signing the previous Annual Report.
7Calculated as 103.8% for Namal Nawana and 94% for Olivier Bohuon, (disclosed on pages 92-93), divided by the maximum potential payout of 150%.
Gender Pay Ratio
In 2018, the Remuneration Committee reviewed our UK Gender Pay ratio. It was noted that the average pay gap had increased from 29% (in 2017) to 31% (in 2018) and the median pay gap from 15% to 21% for the same period. We recognised that the reasons for this increase were a higher level of female attrition with more males being promoted or recruited during the year into senior positions, as well as the move of one senior female executive from UK to US. We shall continue to review these figures and the actions being taken by management to address these gaps across our global business.
A number of initiatives were established in 2018 including: Developing our female leaders – though a programme we call Elevate (280 female professionals participated in the programme); Training our managers and Human Resource professionals in areas such as increasing awareness of unconscious biases; Conducting a masterclass for the Talent Acquisition Team – to further drive diversity and inclusion in our approach to recruitment; and Procuring Talent – widening recruitment channels to improve diversity and inclusion in our candidate pipeline.
From 2019 a programme of activity is planned, under the sponsorship of the Chief Executive Officer and Executive Committee and in line with our new purpose and culture pillars. It includes plans to: leverage a new spirit of inclusion at the Executive level to drive change at a global level; sponsor female talent and leveraging role models to increase diversity in senior roles; pro-actively map the market for female talent, launch a female acceleration programme to drive development of talented women; and train our leaders and managers on how to lead inclusively beyond recruitment.
Shareholding Requirements
The Chief Executive Officer is required to hold three times his salary in the form of shares and the Chief Financial Officer is required to hold two times his salary. Our current remuneration arrangements also require Executive Directors to retain any shares received in respect of Performance Share Awards made in or after 2017 for a period of two years after vesting. The Remuneration Committee will be considering these requirements further as part of our review of Remuneration Policy in 2019 and in particular, we will be looking to introduce some form of post-cessation shareholding requirement for our Executive Directors.
|
|
Smith & Nephew Annual Report 2018 |
|
105 |
Statement of voting at Annual General Meeting held in 2018
At the Annual General Meeting held on 12 April 2018, votes cast by proxy and at the meeting and votes withheld in respect of the votes on the Directors’ Remuneration Report were as follows:
|
Resolution |
Votes for |
% for |
Votes against |
% against |
Total votes |
Votes withheld |
|
Approval of the Directors’ Remuneration Report (excluding policy) |
581,091,881 | 97.29 | 16,160,313 | 2.71 | 597,252,194 | 407,092 |
Senior management remuneration
The Group’s administrative, supervisory and management body (senior management) is comprised for US reporting purposes, of Executive Directors and Executive Officers. Details of the current Executive Directors and Executive Officers are given on pages 54 and 58–61.
Compensation paid to senior management in respect of 2016, 2017 and 2018 was as follows:
| 2018 | 2017 | 2016 | ||
|
Total compensation (excluding pension emoluments, but including cash payments under the performance-related incentive plans) |
$ 15,935,000
|
$ 13,573,000
|
$ 12,874,000
|
|
|
Total compensation for loss of office |
$ 433,000
|
$2,711,000 |
– |
|
|
Aggregate increase in accrued pension scheme benefits |
– |
– |
– |
|
|
Aggregate amounts provided for under supplementary schemes |
$ 1,570,000
|
$ 872,000
|
$ 1,112,000
|
|
As at 15 February 2019, senior management owned 306,666 shares and 112,107 ADSs, constituting less than 0.1% of the share capital of the Company. For this purpose, the Group is defined as the Executive Directors, members of the Executive Committee, including the Company Secretary and their Persons Closely Associated. Details of share awards granted during the year and held as at 15 February 2019 by members of senior management are as follows:
|
Share awards granted |
Total share awards |
|
|
Equity Incentive awards |
218,794 | 257,530 |
|
Performance Share awards at maximum |
510,358 | 987,432 |
|
Conditional share awards under the Global Share Plan 2010 |
95,890 | 244,056 |
|
Options under Employee ShareSave plans |
6,504 | 9,041 |
Dilution headroom
The Remuneration Committee ensures that at all times the number of new shares which may be issued under any share-based plans, including all-employee plans, does not exceed 10% of the Company’s issued share capital over any rolling 10‑year period (of which up to 5% may be issued to satisfy awards under the Company’s discretionary plans). The Company monitors headroom closely when granting awards over shares taking into account the number of options or shares that might be expected to lapse or be forfeited before vesting or exercise. In the event that insufficient new shares are available, there are processes in place to purchase shares in the market to satisfy vesting awards and to net-settle option exercises.
Over the previous 10 years (2009 to 2018), the number of new shares issued under our share plans has been as follows:
|
All-employee share plans 2019 |
7,567,286 (0.86% of issued share capital as at 15 February 2019) |
|
Discretionary share plans |
31,010,812 (3.54% of issued share capital as at 15 February 2019) |
By order of the Board, on 21 February 2019
Angie Risley
Chair of the Remuneration Committee
|
106 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
The Policy Report
FUTURE POLICY TABLE – EXECUTIVE DIRECTORS
The following table and accompanying notes explain the different elements of remuneration we pay to our Executive Directors. It was approved by shareholders at the 2017 Annual General Meeting on 6 April 2017.
|
Base salary |
||
|
We are a FTSE 50 listed company, operating in over 100 countries around the world. Our strategy to generate cash from Established Markets in order to invest for growth in higher growth geographies and franchises means that we are competing for international talent and our base salaries therefore need to reflect what our Executive Directors would receive if they were to work in another international company of a similar size, complexity and geographical scope. |
||
|
How the component operates |
Maximum levels of payment |
Framework in which performance is assessed |
|
Salaries are normally reviewed annually, with any increase applying from 1 April. – Salary levels and increases take account of: – Market movements within a peer group of similarly sized UK listed companies; – Scope and responsibility of the position; – Skill/experience and performance of the individual Director; – General economic conditions in the relevant geographic market; and – Average increases awarded across the Company, with particular regard to increases in the market in which the Executive is based. |
The base salary of the Executive Directors with effect from 1 April 2017 will be as follows: – Olivier Bohuon €1,179,490. – Graham Baker £510,000. The factors noted in the previous column will be taken into consideration when making increases to base salary and when appointing a new Director. In normal circumstances, base salary increases for Executive Directors will relate to the geographic market and peer group. In addition, the average increases for employees across the Group will be taken into account. The Remuneration Committee retains the right to approve higher increases when there is a substantial change in the scope of the Executive Director’s role. A full explanation will be provided in the Implementation Report should higher increases be approved in exceptional cases. |
Performance in the prior year is one of the factors taken into account and poor performance is likely to lead to a zero salary increase.
|
|
Payment in lieu of pension |
||
|
In order to attract and retain Executive Directors with the capability of driving our corporate strategy, we need to provide market-competitive retirement benefits similar to the benefits they would receive if they were to work for one of our competitors. At the same time, we seek to avoid exposing the Company to defined benefit pension risks, and where possible will make payments in lieu of providing a pension. |
||
|
How the component operates |
Maximum levels of payment |
Framework in which performance is assessed |
|
Current Executive Directors receive an allowance in lieu of membership of a Company-run pension scheme. Base salary is the only component of remuneration which is pensionable. |
Up to 30% of base salary. |
The level of payment in lieu of a pension paid to Executive Directors is not dependent on performance. |
|
|
Smith & Nephew Annual Report 2018 |
|
107 |
|
Benefits |
||
|
In order to attract and retain Executive Directors with the capability of driving our corporate strategy, we need to provide a range of market‑competitive benefits similar to the benefits they would receive if they were to work for one of our competitors. It is important that our Executive Directors are free to focus on the Company’s business without being diverted by concerns about medical provision, risk benefit cover or, if required, relocation issues. |
||
|
How the component operates |
Maximum levels of payment |
Framework in which performance is assessed |
|
A wide range of benefits may be provided depending on the benefits provided for comparable roles in the location in which the Executive Director is based. These benefits will include, as a minimum, healthcare cover, life assurance, long-term disability, annual medical examinations, company car or car allowance. The Committee retains the discretion to provide additional benefits where necessary or relevant in the context of the Executive’s location. Where applicable, relocation costs may be provided in-line with the Company’s relocation policy for employees, which may include removal costs, assistance with accommodation, living expenses for self and family and financial consultancy advice. In some cases such payments may be grossed up.
|
The policy is framed by the nature of the benefits that the Remuneration Committee is willing to provide to Executive Directors. The maximum amount payable will depend on the cost of providing such benefits to an employee in the location at which the Executive Director is based. Shareholders should note that the cost of providing comparable benefits in different jurisdictions may vary widely. As an indication, the cost of such benefits provided in 2016 was as follows: – Olivier Bohuon €150,511. – Julie Brown £22,244. The maximum amount payable in benefits to an Executive Director, in normal circumstances, will not be significantly more than amounts paid in 2016 (or equivalent in local currency). The Remuneration Committee retains the right to pay more than this should the cost of providing the same underlying benefits increase or in the event of a relocation. A full explanation will be provided in the Implementation Report should the cost of benefits provided be significantly higher. |
The level and cost of benefits provided to Executive Directors is not dependent on performance but on the package of benefits provided to comparable roles within the relevant location.
|
ALL-EMPLOYEE ARRANGEMENTS
|
All-employee share plans |
||
|
To enable Executive Directors to participate in all-employee share plans on the same basis as other employees. |
||
|
How the component operates |
Maximum levels of payment |
Framework in which performance is assessed |
|
ShareSave Plans are operated in the UK and 31 other countries internationally. In the US, an Employee Stock Purchase Plan is operated. These plans enable employees to save on a regular basis and then buy shares in the Company. Executive Directors are able to participate in such plans on a similar basis to other employees, depending on where they are located. |
Executive Directors may currently invest up to £500 per month in the UK ShareSave Plan. The Remuneration Committee may exercise its discretion to increase this amount up to the maximum permitted by the HM Revenue & Customs. Similar limits will apply in different locations. |
The potential gains from all-employee plans are not based on performance but are linked to growth in the share price.
|
|
108 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
The Policy Report continued
ANNUAL INCENTIVES
|
|
|
|
|
Annual Incentive Plan – cash incentive |
||
|
To motivate and reward the achievement of specific annual financial and business objectives related to the Company’s strategy and sustained through a clawback mechanism explained more fully in the notes. The objectives which determine the payment of the annual cash incentive and the level of the annual equity award are linked closely to the Group strategy. The financial measures of Revenue, Trading Profit Margin and Trading Cash Flow underline our strategy for growth. The business objectives are also linked to the Group strategy. These change from year to year to reflect the evolving strategy, but will typically be linked to the Strategic Priorities set out in this Annual Report. The Implementation Report each year will explain how each objective is linked to a specific strategic priority. |
||
|
How the component operates |
Maximum levels of payment |
Framework in which performance is assessed |
|
The Annual Incentive Plan comprises a cash and an equity component, both based on the achievement of financial and business objectives set at the start of the year. The cash component is paid in full after the end of the performance year. At the end of the year, the Remuneration Committee determines the extent to which performance against these has been achieved and sets the award level. |
The total maximum payable under the Annual Incentive Plan is 215% of base salary (150% Cash Incentive and 65% Equity Incentive). In respect of the Cash Incentive: – 150% salary awarded for maximum performance. – 100% salary awarded for target performance. – 50% salary awarded for threshold performance. – Performance assessed against individual objectives and Group financial targets. |
The cash and share awards are subject to malus and clawback as detailed in the notes following this table. 75% of the cash component is based on financial performance measures, which currently include Revenue (35%), Trading Profit Margin (25%) and Trading Cash Flow (15%). 25% of the cash component is based on other business goals linked to the Company’s strategy, which could include financial and non‑financial measures. The Remuneration Committee retains the discretion to adjust the relative weightings of the financial and business components, and to adopt any performance measure that is relevant to the Company. |
|
Annual Incentive Plan – equity incentive |
||
|
To drive share ownership and encourage sustained high standards through the application of a ‘malus’ provision over three years, explained more fully in the notes. |
||
|
How the component operates |
Maximum levels of payment |
Framework in which performance is assessed |
|
The equity award component comprises conditional share awards (made at the time of the cash award), with vesting phased over the following three years. The equity component vests 1/3, 1/3, 1/3 on successive award anniversaries, only if performance remains satisfactory over each of these three years; otherwise the award will lapse. Participants will receive an additional number of shares equivalent to the amount of dividend payable per vested share during the relevant performance period. |
In respect of the Equity Incentive: – Performance is assessed against individual performance, which includes an element of Group financial targets. – 65% of salary awarded for maximum performance. – 50% of salary awarded for target performance. – 0% of salary awarded for performance assessed to be below target.
|
The Remuneration Committee will use its judgement of the individual’s performance based both on what has been achieved during the year and how it has been achieved in determining the level of equity award that may be awarded within the range of 0% to 65% of salary. The equity component will vest in three equal tranches over a three-year period, provided that satisfactory performance is sustained.
|
|
|
Smith & Nephew Annual Report 2018 |
|
109 |
LONG-TERM INCENTIVES (AWARDS ACTIVELY BEING MADE)
|
Performance Share Programme |
||
|
To motivate and reward longer-term performance linked to the long-term strategy and share price of the Company. The performance measures which determine the level of vesting of the Performance Share Awards are linked to our corporate strategy. |
||
|
How the component operates |
Maximum levels of payment |
Framework in which performance is assessed |
|
The Performance Share Programme comprises conditional share awards which vest after three years, subject to the achievement of stretching performance targets linked to the Company’s strategy. Awards may be subject to clawback in the event of material financial misstatement or misconduct. Participants will receive an additional number of shares equivalent to the amount of dividend payable per vested share during the relevant performance period. On vesting, a number of shares are sold to cover the tax liability. The remaining shares are required to be held by the Executive Director for a further two-year holding period. |
Annual awards: – 190% of salary for maximum performance. – 95% of salary for target performance. – 47.5% of salary for threshold performance. |
Currently: – 25% of the award vests on achievement of a three‑year cumulative free cash flow target. – 25% of the award vests subject to three-year Total Shareholder Return (TSR) at median performance relative to Global Healthcare companies and to FTSE 100 companies. – 25% of the award vests subject to the achievement of return on invested capital targets. – 25% of the award vests subject to total sales growth. – These measures, the targets and performance against them are described more fully in the Implementation Report. – The Performance Share Award will vest on the third anniversary of the date of grant, depending on the extent to which the performance conditions are met over the three-year period commencing in the year the award was made. – The Remuneration Committee retains the discretion to change the measures and their respective weightings to ensure continuing alignment with the Company’s strategy. – The cash and share awards are subject to malus and clawback as detailed in the notes following this table. – Awards made prior to 2017 were subject to TSR against a sector peer group, cash flow and revenue in Emerging Markets targets. |
|
110 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
The Policy Report continued
ILLUSTRATIONS OF THE APPLICATION OF THE REMUNERATION POLICY 2017
The following charts show the potential split between the different elements of the Executive Directors’ remuneration under three different performance scenarios.
|
Figures as at salary levels in 2017, when the Policy Report was approved by shareholders |
|||
|
CHIEF EXECUTIVE OFFICER |
CHIEF FINANCIAL OFFICER |
||
|
|
|
||
|
|
|||
|
|
|||
|
Total Remuneration by Performance Scenario for 2017 Financial Year (percentage split) |
|||
|
CHIEF EXECUTIVE OFFICER |
CHIEF FINANCIAL OFFICER |
||
|
|
|
||
MALUS AND CLAWBACK
The Remuneration Committee may determine that an unvested award or part of an award may not vest (regardless of whether or not the performance conditions have been met) or may determine that any cash bonus, vested shares, or their equivalent value in cash be returned to the Company in the event that any of the following matters is discovered:
– A material misstatement of the Company’s financial results; or
– A material error in determining the extent to which any performance condition has been satisfied; or
– A significant adverse change in the financial performance of the Company, or a significant loss at a general level or at the country business unit or function in which a participant worked; or
– Inappropriate conduct (for example reputational issues), capability or performance by a participant, or within a team business area or profit centre.
These provisions apply to share awards under the Global Share Plan 2010 and cash amounts under the Annual Cash Incentive Plan.
POLICY ON RECRUITMENT ARRANGEMENTS
Our policy on the recruitment of Executive Directors is to pay a fair remuneration package for the role being undertaken and the experience of the Executive Director appointed. In terms of base salary, we will seek to pay a salary comparable, in the opinion of the Committee, to that which would be paid for an equivalent position elsewhere. The Remuneration Committee will determine a base salary in-line with the policy and having regard to the parameters set out on in the future policy table. Incoming Executive Directors will be entitled to pension, benefit and incentive arrangements which are the same as provided to existing Executive Directors. On that basis, incentive awards would not exceed 405% of base salary.
We recognise that in the event that we require a new Executive Director to relocate to take up a position with the Company, we will also pay relocation and related costs as described in the future policy table, which is in-line with the relocation arrangements we operate across the Group.
We also recognise that in many cases, an external appointee may forfeit sizeable cash bonuses and share awards if they choose to leave their former employer and join us. The Remuneration Committee therefore believes that we need the ability to compensate new hires for incentive awards they give up on joining us. The Committee will use its judgement in determining any such compensation, which will be decided on a case-by-case basis. We will only provide compensation which is no more beneficial than that given up by the new appointee and we will seek evidence from the previous employer to confirm the full details of bonus or share awards being forfeited. As far as possible, we will seek to replicate forfeited share awards using Smith & Nephew incentive plans or through reliance on Rule 9.4.2 in the Listing Rules, whilst at the same time aiming for simplicity.
|
|
Smith & Nephew Annual Report 2018 |
|
111 |
If we appoint an existing employee as an Executive Director of the Company, pre-existing obligations with respect to remuneration, such as pension, benefits and legacy share awards, will be honoured. Should these differ materially from current arrangements, these will be disclosed in the next Implementation Report.
We will supply details via an announcement to the London Stock Exchange of an incoming Executive Director’s remuneration arrangements at the time of their appointment.
SERVICE CONTRACTS
We employ Executive Directors on rolling service contracts with notice periods of up to 12 months from the Company and six months from the Executive Director. On termination of the contract, we may require the Executive Director not to work their notice period and pay them an amount equivalent to the base salary and payment in lieu of pension and benefits they would have received if they had been required to work their notice period.
Under the terms of the Executive Director’s service contract, Executive Directors are restricted for a period of 12 months after leaving the employment of the Company from working for a competitor, soliciting orders from customers and offering employment to employees of Smith & Nephew. The Company retains the right to waive these provisions in certain circumstances. In the event that these provisions are waived or the former Executive Director commences employment earlier than at the end of the notice period, no further payments shall be made in respect of the portion of notice period not worked. Directors’ service contracts are available for inspection at the Company’s registered office: 15 Adam Street, London WC2N 6LA.
POLICY ON PAYMENT FOR LOSS OF OFFICE
Our policy regarding termination payments to departing Executive Directors is to limit severance payments to pre-established contractual arrangements. In the event that the employment of an Executive Director is terminated, any compensation payable will be determined in accordance with the terms of the service contract between the Company and the Executive Director, as well as the rules of any incentive plans.
Under normal circumstances (excluding termination for gross misconduct) all leavers are entitled to receive termination payments in lieu of notice equal to base salary, payment in lieu of pension, and benefits. In some circumstances additional benefits may become payable to cover reimbursement of untaken holiday leave, repatriation and outplacement fees, legal and financial advice.
In addition, we may also in exceptional circumstances exercise our discretion to pay the Executive Director a proportion of the annual cash incentive they would have received had they been required to work their notice period. Any entitlement or discretionary payment may be reduced in-line with the Executive Director’s duty to mitigate losses, subject to applying our non-compete clause.
We will supply details via an announcement to the London Stock Exchange of a departing Executive Director’s termination arrangements at the time of departure.
In the case of a change of control which results in the termination of an Executive Director or a material alteration to their responsibilities or duties, within 12 months of the event, the Executive Director would be entitled to receive 12 months’ base salary plus payment in lieu of pension and benefits. In addition, the Remuneration Committee has discretion to pay an Executive Director in these circumstances an annual cash incentive. For Directors appointed prior to 1 November 2012, an automatic annual cash incentive is payable at target.
In the event that an Executive Director leaves for reasons of ill-health, death, redundancy or retirement in agreement with the Company, then the vesting of any outstanding annual cash incentive and equity incentive awards will generally depend on the Remuneration Committee’s assessment of performance to date. Performance share awards will be pro-rated for the time worked during the relevant performance period, and will remain subject to performance over the full performance period.
For all other leavers, the annual cash incentive will generally be forfeited and outstanding equity incentive awards and performance share awards will lapse.
One-off awards granted on appointment will normally lapse on leaving except in cases of death, retirement, redundancy, or ill-health. The Remuneration Committee has discretion to permit such awards to vest in other circumstances and will be subject to satisfactorily meeting performance conditions if applicable.
The Remuneration Committee retains discretion to alter these provisions on a case-by-case basis following a review of circumstances and to ensure fairness for both shareholders and Executive Directors.
We will supply details via an announcement to the London Stock Exchange of an out-going Executive Director’s remuneration arrangements around the time of leaving.
|
112 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
The Policy Report continued
CHANGES TO POLICY
The 2017 Remuneration Policy makes the following changes to the 2014 Remuneration policy:
– Introduction of a two-year holding period for vested Performance shares;
– Flexibility to change measures;
– Increased emphasis on financial objectives in the Annual Incentive Plan, increases from 70% to 75%; and
– Increased shareholding requirement to 300% of salary for the Chief Executive Officer.
Further details can be found in the letter from the Chairman of the Remuneration Committee on page 79 of the 2017 Annual Report.
POLICY ON SHAREHOLDING REQUIREMENTS
The Remuneration Committee believes that one of the best ways our Executive Directors can have a greater alignment with shareholders is for them to hold a significant number of shares in the Company. The Chief Executive Officer is therefore expected to build up a holding of Smith & Nephew shares worth three times their base salary and the Chief Financial Officer is expected to build up a holding of two times their basic salary. In order to reinforce this expectation, we require them to retain 50% of the shares (after tax) vesting under the equity incentive programmes until this holding has been met, recognising that differing international tax regimes affect the pace at which an Executive Director may fulfil the shareholding requirement. When calculating whether or not this requirement has been met, we will include ordinary shares or ADRs held by the Executive Director and their immediate family. Ordinarily, we would expect this required shareholding to have been built up within a period of five years from the date of appointment.
Furthermore, from awards made in 2017, we require our Executive Directors to retain all the shares (after tax) vesting under the Performance Share Programme for a period of two years after vesting.
STATEMENT OF CONSIDERATION OF EMPLOYMENT CONDITIONS ELSEWHERE IN THE COMPANY AND DIFFERENCES TO THE EXECUTIVE DIRECTOR POLICY
All employees across the Group have performance-based pay linked to objectives derived from the strategic priorities, which underpin the performance metrics in the Executive Director Incentive Plans.
Executive Director base salaries will generally increase at a rate in-line with the average salary increases awarded across the Company. Given the diverse geographic markets within which the Company operates, the Committee will generally be informed by the average salary increase in both the market local to the Executive and the UK, recognising the Company’s place of listing, and will also consider market data periodically.
A range of different pension arrangements operate across the Group depending on location and/or length of service. Executive Directors and Executive Officers either participate in the legacy pension arrangements relevant to their local market or receive a cash payment of 30% of salary in lieu of a pension. Senior executives who do not participate in a local Company pension plan receive a cash payment of 20% of salary in lieu of pension. Differing amounts apply for lower levels within the Company.
The Company has established a benefits framework under which the nature of benefits varies by geography. Executive Directors participate in benefit arrangements similar to those applied for employees within the applicable location.
All employees are set objectives at the beginning of each year, which link through to the objectives set for the Executive Directors. Annual cash incentives payable to employees across the Company depend on the satisfactory completion of these objectives as well as performance against relevant Group and country business unit financial targets relating to revenue, trading profit and trading cash, similar to the financial targets set for the Executive Directors.
Executive Officers and senior executives (61 as at 2017) participate in the annual Equity Incentive Programme and the Performance Share Programme. The maximum amounts payable are lower, but the performance conditions are the same as those that apply to the Executive Directors.
No specific consultation with employees has been undertaken relating to Director remuneration. However, regular employee surveys are conducted across the Group, which cover a wide range of issues relating to local employment conditions and an understanding of Group-wide strategic matters. As at 2017, around 5,000 employees in 63 countries participate in one or more of our global share plans.
|
|
Smith & Nephew Annual Report 2018 |
|
113 |
FUTURE POLICY TABLE – CHAIRMAN AND NON-EXECUTIVE DIRECTORS
The following table and accompanying notes explain the different elements of remuneration we pay to our Chairman and Non-Executive Directors. No element of their remuneration is subject to performance. All payments made to the Chairman are determined by the Remuneration Committee, whilst payments made to the Non-Executive Directors are determined by the Directors who are not themselves Non-Executive Directors, currently the Chairman, the Chief Executive Officer and the Chief Financial Officer.
|
Annual fees |
|
|
Basic annual fee |
|
|
To attract and retain Directors by setting fees at rates comparable to what would be paid in an equivalent position elsewhere. A proportion of the fees are paid in shares in the third quarter of each year in order to align Non-Executive Directors’ fees with the interests of shareholders. |
|
|
How the component operates |
Maximum levels of payment |
|
Fees will be reviewed periodically. In future, any increase will be paid in shares until 25% of the total fee is paid in shares. Fees are set in-line with market practice for fees paid by similarly sized UK listed companies. Annual fees are set and paid in UK Sterling or US Dollars depending on the location of the Non-Executive Director. If appropriate, fees may be set and paid in alternative currencies. |
Annual fees are currently as follows: – £63,000 in cash plus £5,135 in shares; or – $120,000 in cash plus $9,780 in shares. Chairman fee: – £309,000 plus £103,000 in shares. Whilst it is not expected to increase the fees paid to the Non-Executive Directors and the Chairman by more than the increases paid to employees generally, in exceptional circumstances higher fees might become payable. The total maximum aggregate fees payable to the Non-Executive Directors will not exceed £1.5 million as set out in the Company’s Articles of Association. |
|
Fee for Senior Independent Director and Committee Chairmen |
|
|
To compensate Non-Executive Directors for the additional time spent as Committee Chairmen or as the Senior Independent Director. |
|
|
How the component operates |
Maximum levels of payment |
|
A fixed fee is paid, which is reviewed periodically. |
– £20,000 in cash; or – $35,000 in cash. Whilst it is not expected that the fees paid to the Senior Independent Director or Committee Chairmen will exceed the increases paid to employees generally, in exceptional circumstances, higher fees might become payable. |
|
Intercontinental travel |
|
|
To compensate Non-Executive Directors for the time spent travelling to attend meetings in another continent. |
|
|
How the component operates |
Maximum levels of payment |
|
A fixed fee is paid, which is reviewed periodically. |
– £3,500 in cash; or – $7,000 in cash. Whilst it is not expected to increase these fees by more than the increases paid to employees generally, in exceptional circumstances, higher fees might become payable. |
|
114 |
Governance |
Smith & Nephew Annual Report 2018 |
|
Directors’ Remuneration Report
The Policy Report continued
NOTES TO FUTURE POLICY TABLE – NON-EXECUTIVE DIRECTORS
Changes to Remuneration Policy
There have been no changes to our Remuneration Policy as it applies to Non-Executive Directors, since the Policy was initially approved by shareholders in April 2014.
Additional duties undertaken by Non-Executive Directors
In the event that the Chairman or a Non-Executive Director is required to undertake significant additional executive duties in order to support the Executive Directors during a period of absence due to illness or a gap prior to the appointment of a permanent Executive Director, the Remuneration Committee is authorised to determine an appropriate level of fees which shall be payable. These fees will not exceed the amounts which would normally be paid to a permanent Executive Director undertaking such duties and shall not include participation in short- or long-term incentive arrangements or benefit plans.
Policy on recruitment arrangements
Any new Non-Executive Director shall be paid in accordance with the current fee levels on appointment, in line with the Policy set out above. With respect to the appointment of a new Chairman, fee levels will take into account market rates, the individual’s profile and experience, the time required to undertake the role and general business conditions. In addition, the Remuneration Committee retains the right to authorise the payment of relocation assistance or an accommodation allowance in the event of the appointment of a Chairman not based within the UK.
Letters of appointment
The Chairman and Non-Executive Directors have letters of appointment which set out the terms under which they provide their services to the Company and are available for inspection at the Company’s registered office: 15 Adam Street, London WC2N 6LA. The appointment of Non-Executive Directors is not subject to a notice period, nor is there any compensation payable on loss of office, for example, should they not be re-elected at an Annual General Meeting. The appointment of the Chairman is subject to a notice period of six months.
The Chairman and Non-Executive Directors are required to acquire a shareholding in the Company equivalent in value to one times their basic fee within two years of their appointment to the Board.
STATEMENT OF CONSIDERATION OF SHAREHOLDER VIEWS
The broad outline of our remuneration arrangements have remained largely unchanged since 2012. As our strategy has evolved, we have altered some of the measures we use in our short- and long-term incentive plans, but the overall structure of our remuneration arrangements has remained the same. Shareholders formally approved the Remuneration Policy in respect of our Executive Directors at the Annual General Meeting in 2014. Joseph Papa, Chairman of the Remuneration Committee, has met with shareholders before the policy was approved and every year since, in order to ascertain shareholder views on our remuneration arrangements.
Ahead of the Annual General Meeting in 2016, Mr Papa held meetings and calls with 28 shareholders holding approximately 33% of the Company’s Share Capital. Although the holders of 53% of our shares voted against the Remuneration Report in 2016, our engagement ahead of the 2016 Annual General Meeting had shown us that shareholders were broadly supportive of our Remuneration Policy and those who opposed the Remuneration Report were primarily voting against the use of discretion rather than any aspect of the Remuneration Policy.
During 2016, following the Annual General Meeting, Mr Papa continued to engage extensively with shareholders. In Autumn 2016, he met with or held telephone calls with 28 shareholders holding around 41% of the Company’s shares. The shareholders he met ranged from some of our top 20 shareholders down to smaller active and engaged shareholders holding fewer than one million shares. He discussed our proposals to continue with the same overall remuneration arrangements, whilst altering the performance measures used in the short- and long-term incentive plans. We found the discussions with shareholders at this time useful in helping to understand the measures and targets which were important to our shareholders, and those which shareholders did not support. As a result of these discussions, some updated performance measures have been incorporated into our incentive plans for 2017 and a two-year holding period will now apply on the vesting of performance shares for our Executive Directors.
|
|
Smith & Nephew Annual Report 2018 |
|
115 |
Accounts
| 116 | |
| 117 | |
| 124 | |
| 125 | |
| 129 | |
| 178 | |
| 180 |

|
116 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
STATEMENT OF DIRECTORS’ RESPONSIBILITIES IN RESPECT OF THE ANNUAL REPORT AND THE FINANCIAL STATEMENTS
The Directors are responsible for preparing this Annual Report and Form 20‑F Information and the Group and Parent Company Financial Statements, in accordance with applicable law and regulations.
Company law requires the Directors to prepare Group and Parent Company financial statements for each financial year. Under that law they are required to prepare the Group financial statements in accordance with International Financial Reporting Standards (IFRSs) as issued by the International Accounting Standards Board (IASB) and as adopted by the European Union (IFRSs as adopted by the EU) and applicable law and have elected to prepare the Parent Company financial statements in accordance with UK accounting standards, including FRS 101 Reduced Disclosure Framework.
Under company law the Directors must not approve the financial statements unless they are satisfied that they give a true and fair view of the state of affairs of the Group and Parent Company and of their profit or loss for that period. In preparing each of the Group and Parent Company Financial Statements, the Directors are required to:
select suitable accounting policies and then apply them consistently;
make judgements and estimates that are reasonable, relevant, reliable and prudent;
for the Group financial statements, state whether they have been prepared in accordance with IFRSs, as issued by the IASB and adopted by the EU;
for the Parent Company financial statements, state whether applicable UK Accounting Standards have been followed, subject to any material departures disclosed and explained in the Parent Company Financial Statements;
assess the Group and Parent Company’s ability to continue as a going concern, disclosing, as applicable, matters related to going concern; and
use the going concern basis of accounting unless they either intend to liquidate the Group or the Parent Company or to cease operations, or have no realistic alternative but to do so.
The Directors are responsible for keeping adequate accounting records that are sufficient to show and explain the Parent Company’s transactions and disclose with reasonable accuracy at any time the financial position of the Parent Company and enable them to ensure that its financial statements comply with the Companies Act 2006. They are responsible for such internal control as they determine is necessary to enable the preparation of financial statements that are free from material misstatement, whether due to fraud or error, and have general responsibility for taking such steps as are reasonably open to them to safeguard the assets of the Group and to prevent and detect fraud and other irregularities.
Under applicable law and regulations, the Directors are also responsible for preparing a Strategic Report, Directors’ Report, Directors’ Remuneration Report and Corporate Governance Statement that complies with that law and those regulations.
The Directors are responsible for the maintenance and integrity of the corporate and financial information included on the Company’s website. Legislation in the UK governing the preparation and dissemination of financial statements may differ from legislation in other jurisdictions.
RESPONSIBILITY STATEMENT OF THE DIRECTORS IN RESPECT OF THE ANNUAL REPORT
We confirm that to the best of our knowledge:
the financial statements, prepared in accordance with the applicable set of accounting standards, give a true and fair view of the assets, liabilities, financial position and profit or loss of the Company and the undertakings included in the consolidation taken as a whole; and
the Strategic Report and Directors’ Report include a fair review of the development and performance of the business and the position of the issuer and the undertakings included in the consolidation taken as a whole, together with a description of the principal risks and uncertainties that they face.
The Strategic Report, which has been prepared in accordance with the requirements of the Companies Act 2006, comprises pages 2–51.
The Directors’ Report has also been prepared in accordance with the Companies Act 2006 and The Small Companies and Groups (Accounts and Directors’ Report) Regulations 2008 comprising of pages 2, 10–11, 24–25, 32–39, 40–83, 116, 154–156, 173 and pages 187–209 of the Annual Report, and has been approved and signed on behalf of the Board.
We consider the Annual Report and financial statements, taken as a whole, are fair, balanced and understandable and provide the information necessary for shareholders to assess the Group’s position and performance, business model and strategy.
By order of the Board, on 21 February 2019
Susan Swabey
Company Secretary
|
|
Smith & Nephew Annual Report 2018 |
|
117 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the shareholders and board of directors
Smith & Nephew plc:
Opinions on the Consolidated Financial Statements and Internal Control Over Financial Reporting
We have audited the accompanying Group balance sheets of Smith & Nephew plc and subsidiaries (the “Group”) as of 31 December 2018 and 2017, the related Group income statements, Group statements of comprehensive income, Group cash flow statements and Group statements of changes in equity for each of the years in the three-year period ended 31 December 2018, and the related notes (collectively, the “consolidated financial statements”). We also have audited the Group’s internal control over financial reporting as of 31 December 2018, based on criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Group as of 31 December 2018 and 2017, and the results of its operations and its cash flows for each of the years in the three-year period ended 31 December 2018, in conformity with International Financial Reporting Standards (“IFRS”) as issued by the International Accounting Standards Board and in conformity with IFRS as adopted by the European Union. Also in our opinion, the Group maintained, in all material respects, effective internal control over financial reporting as of 31 December 2018, based on criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission.
Basis for Opinions
The Group’s management is responsible for these consolidated financial statements, for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Evaluation of Internal Controls. Our responsibility is to express an opinion on the Group’s consolidated financial statements and an opinion on the Group’s internal control over financial reporting based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Group in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud, and whether effective internal control over financial reporting was maintained in all material respects.
Our audits of the consolidated financial statements included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
KPMG LLP
We have served as the Group’s auditor since 2015.
London, United Kingdom
21 February 2019
|
118 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
GROUP FINANCIAL STATEMENTS
THIS PAGE LEFT INTENTIONALLY BLANK
|
|
Smith & Nephew Annual Report 2018 |
|
119 |
THIS PAGE LEFT INTENTIONALLY BLANK
|
120 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
GROUP FINANCIAL STATEMENTS
THIS PAGE LEFT INTENTIONALLY BLANK
|
|
Smith & Nephew Annual Report 2018 |
|
121 |
THIS PAGE LEFT INTENTIONALLY BLANK
|
122 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
GROUP FINANCIAL STATEMENTS
THIS PAGE LEFT INTENTIONALLY BLANK
|
|
Smith & Nephew Annual Report 2018 |
|
123 |
THIS PAGE LEFT INTENTIONALLY BLANK
|
124 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
GROUP FINANCIAL STATEMENTS
Critical judgements and estimates
The Group prepares its consolidated financial statements in accordance with IFRS as issued by the IASB and IFRS as adopted by the EU, the application of which often requires judgements and estimates to be made by management when formulating the Group’s financial position and results. Under IFRS, the Directors are required to adopt those accounting policies most appropriate to the Group’s circumstances for the purpose of presenting fairly the Group’s financial position, financial performance and cash flows.
The Group’s accounting policies do not include any critical judgements. The Group’s accounting policies are set out in Notes 1-24 of the Notes to the Group accounts. Of those, the policies which require the most use of management’s estimation are as follows:
VALUATION OF INVENTORIES
A feature of the Orthopaedic Reconstruction and Trauma & Extremities franchises (whose inventory make up approximately 60% of the Group’s total inventory) is the high level of product inventory required, some of which is located at customer premises and is available for customers’ immediate use. Complete sets of products, including large and small sizes, have to be made available in this way. These sizes are used less frequently than standard sizes and towards the end of the product life cycle are inevitably in excess of requirements. Adjustments to carrying value are therefore required to be made to orthopaedic inventory to anticipate this situation. These adjustments are calculated in accordance with a formula based on levels of inventory compared with historical usage. This formula is applied on an individual product line basis and is first applied when a product group has been on the market for two years. This method of calculation is considered appropriate based on experience, but it does require management estimate in respect of customer demand, effectiveness of inventory deployment, length of product lives, phase-out of old products and efficiency of manufacturing planning systems.
IMPAIRMENT
In carrying out impairment reviews of intangible assets, a number of significant assumptions have to be made when preparing cash flow projections. These include the future rate of market growth, discount rates, the market demand for the products acquired, the future profitability of acquired businesses or products, levels of reimbursement and success in obtaining regulatory approvals. If actual results should differ or changes in expectations arise, impairment charges may be required which would adversely impact operating results.
LIABILITY PROVISIONING
The recognition of provisions for legal disputes is subject to a significant degree of estimation. Provision is made for loss contingencies when it is considered probable that an adverse outcome will occur and the amount of the loss can be reasonably estimated. In making its estimates, management takes into account the advice of internal and external legal counsel. Provisions are reviewed regularly and amounts updated where necessary to reflect developments in the disputes. The ultimate liability may differ from the amount provided depending on the outcome of court proceedings and settlement negotiations or if investigations bring to light new facts.
TAXATION
The Group operates in numerous tax jurisdictions around the world and it is Group policy to submit its tax returns to the relevant tax authorities as promptly as possible. At any given time, the Group is involved in disputes and tax audits and will have a number of tax returns potentially subject to audit. Significant issues may take several years to resolve. In estimating the probability and amount of any tax charge, management takes into account the views of internal and external advisers and updates the amount of tax provision whenever necessary. The ultimate tax liability may differ from the amount provided depending on factors including interpretations of tax law, settlement negotiations or changes in legislation.
|
|
Smith & Nephew Annual Report 2018 |
|
125 |
|
|
|
|
|
Year ended |
|
Year ended |
|
Year ended |
|
|
|
|
|
|
31 December |
|
31 December |
|
31 December |
|
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
$ million |
|
|
Revenue |
|
2 |
|
4,904 |
|
4,765 |
|
4,669 |
|
|
Cost of goods sold |
|
|
|
(1,298) |
|
(1,248) |
|
(1,272) |
|
|
Gross profit |
|
|
|
3,606 |
|
3,517 |
|
3,397 |
|
|
Selling, general and administrative expenses |
|
3 |
|
(2,497) |
|
(2,360) |
|
(2,366) |
|
|
Research and development expenses |
|
3 |
|
(246) |
|
(223) |
|
(230) |
|
|
Operating profit |
|
2 & 3 |
|
863 |
|
934 |
|
801 |
|
|
Interest income |
|
4 |
|
8 |
|
6 |
|
6 |
|
|
Interest expense |
|
4 |
|
(59) |
|
(57) |
|
(52) |
|
|
Other finance costs |
|
4 |
|
(20) |
|
(10) |
|
(16) |
|
|
Share of results of associates |
|
11 |
|
(11) |
|
6 |
|
(3) |
|
|
Profit on disposal of business |
|
21 |
|
– |
|
– |
|
326 |
|
|
Profit before taxation |
|
|
|
781 |
|
879 |
|
1,062 |
|
|
Taxation |
|
5 |
|
(118) |
|
(112) |
|
(278) |
|
|
Attributable profit for the year1 |
|
|
|
663 |
|
767 |
|
784 |
|
|
Earnings per ordinary share1 |
|
6 |
|
|
|
|
|
|
|
|
Basic |
|
|
|
76.0¢ |
|
87.8¢ |
|
88.1¢ |
|
|
Diluted |
|
|
|
75.7¢ |
|
87.7¢ |
|
87.8¢ |
|
Group statement of comprehensive income
|
|
|
|
|
Year ended |
|
Year ended |
|
Year ended |
|
|
|
|
|
|
31 December |
|
31 December |
|
31 December |
|
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
$ million |
|
|
Attributable profit for the year1 |
|
|
|
663 |
|
767 |
|
784 |
|
|
Other comprehensive income: |
|
|
|
|
|
|
|
|
|
|
Items that will not be reclassified to income statement |
|
|
|
|
|
|
|
|
|
|
Re-measurement of net retirement benefit obligations |
|
18 |
|
11 |
|
64 |
|
(81) |
|
|
Taxation on other comprehensive income |
|
5 |
|
(1) |
|
(9) |
|
10 |
|
|
Total items that will not be reclassified to income statement |
|
|
|
10 |
|
55 |
|
(71) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Items that may be reclassified subsequently to income statement |
|
|
|
|
|
|
|
|
|
|
Cash flow hedges – forward foreign exchange contracts |
|
|
|
|
|
|
|
|
|
|
Gains/(losses) arising in the year |
|
|
|
21 |
|
(45) |
|
(15) |
|
|
Losses transferred to inventories for the year |
|
|
|
2 |
|
21 |
|
20 |
|
|
Fair value remeasurement of available for sale asset |
|
|
|
– |
|
(10) |
|
10 |
|
|
Exchange differences on translation of foreign operations |
|
|
|
(132) |
|
181 |
|
(134) |
|
|
Taxation on other comprehensive income |
|
5 |
|
(3) |
|
– |
|
– |
|
|
Total items that may be reclassified subsequently to income statement |
|
|
|
(112) |
|
147 |
|
(119) |
|
|
Other comprehensive (loss)/income for the year, net of taxation |
|
|
|
(102) |
|
202 |
|
(190) |
|
|
Total comprehensive income for the year1 |
|
|
|
561 |
|
969 |
|
594 |
|
|
1 |
Attributable to equity holders of the Company and wholly derived from continuing operations. |
 The Notes on pages 129-177 are an integral part of these accounts.
The Notes on pages 129-177 are an integral part of these accounts.
|
126 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
GROUP FINANCIAL STATEMENTS
|
|
|
|
|
At |
|
At |
|
|
|
|
|
|
31 December |
|
31 December |
|
|
|
|
|
|
2018 |
|
2017 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
|
Assets |
|
|
|
|
|
|
|
|
Non-current assets |
|
|
|
|
|
|
|
|
Property, plant and equipment |
|
7 |
|
1,062 |
|
1,049 |
|
|
Goodwill |
|
8 |
|
2,337 |
|
2,371 |
|
|
Intangible assets |
|
9 |
|
1,210 |
|
1,371 |
|
|
Investments |
|
10 |
|
34 |
|
21 |
|
|
Investments in associates |
|
11 |
|
105 |
|
118 |
|
|
Other non-current assets |
|
13 |
|
16 |
|
16 |
|
|
Retirement benefit assets |
|
18 |
|
92 |
|
62 |
|
|
Deferred tax assets |
|
5 |
|
126 |
|
127 |
|
|
|
|
|
|
4,982 |
|
5,135 |
|
|
Current assets |
|
|
|
|
|
|
|
|
Inventories |
|
12 |
|
1,395 |
|
1,304 |
|
|
Trade and other receivables |
|
13 |
|
1,317 |
|
1,258 |
|
|
Cash at bank |
|
15 |
|
365 |
|
169 |
|
|
|
|
|
|
3,077 |
|
2,731 |
|
|
Total assets |
|
|
|
8,059 |
|
7,866 |
|
|
|
|
|
|
|
|
|
|
|
Equity and liabilities |
|
|
|
|
|
|
|
|
Equity attributable to owners of the Company |
|
|
|
|
|
|
|
|
Share capital |
|
19 |
|
177 |
|
178 |
|
|
Share premium |
|
|
|
608 |
|
605 |
|
|
Capital redemption reserve |
|
|
|
18 |
|
17 |
|
|
Treasury shares |
|
19 |
|
(214) |
|
(257) |
|
|
Other reserves |
|
|
|
(340) |
|
(228) |
|
|
Retained earnings |
|
|
|
4,625 |
|
4,329 |
|
|
Total equity |
|
|
|
4,874 |
|
4,644 |
|
|
Non-current liabilities |
|
|
|
|
|
|
|
|
Long-term borrowings |
|
15 |
|
1,301 |
|
1,423 |
|
|
Retirement benefit obligations |
|
18 |
|
114 |
|
131 |
|
|
Other payables |
|
14 |
|
53 |
|
128 |
|
|
Provisions |
|
17 |
|
153 |
|
97 |
|
|
Deferred tax liabilities |
|
5 |
|
99 |
|
97 |
|
|
|
|
|
|
1,720 |
|
1,876 |
|
|
Current liabilities |
|
|
|
|
|
|
|
|
Bank overdrafts, borrowings and loans |
|
15 |
|
164 |
|
27 |
|
|
Trade and other payables |
|
14 |
|
957 |
|
957 |
|
|
Provisions |
|
17 |
|
121 |
|
129 |
|
|
Current tax payable |
|
5 |
|
223 |
|
233 |
|
|
|
|
|
|
1,465 |
|
1,346 |
|
|
Total liabilities |
|
|
|
3,185 |
|
3,222 |
|
|
Total equity and liabilities |
|
|
|
8,059 |
|
7,866 |
|
The accounts were approved by the Board and authorised for issue on 21 February 2019 and are signed on its behalf by:
|
Roberto Quarta |
Namal Nawana |
Graham Baker |
|
Chairman |
Chief Executive Officer |
Chief Financial Officer |
 The Notes on pages 129-177 are an integral part of these accounts.
The Notes on pages 129-177 are an integral part of these accounts.
|
|
Smith & Nephew Annual Report 2018 |
|
127 |
|
|
|
|
|
Year ended |
|
Year ended |
|
Year ended |
|
|
|
|
|
|
31 December |
|
31 December |
|
31 December |
|
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
$ million |
|
|
Cash flows from operating activities |
|
|
|
|
|
|
|
|
|
|
Profit before taxation |
|
|
|
781 |
|
879 |
|
1,062 |
|
|
Net interest expense |
|
4 |
|
51 |
|
51 |
|
46 |
|
|
Depreciation, amortisation and impairment |
|
|
|
435 |
|
447 |
|
463 |
|
|
Loss on disposal of property, plant and equipment and software |
|
|
|
19 |
|
13 |
|
15 |
|
|
Share-based payments expense (equity settled) |
|
23 |
|
35 |
|
31 |
|
27 |
|
|
Share of results of associates |
|
11 |
|
11 |
|
(6) |
|
3 |
|
|
Profit on disposal of business |
|
21 |
|
– |
|
– |
|
(326) |
|
|
Net movement in post-retirement benefit obligations |
|
|
|
(35) |
|
(40) |
|
(85) |
|
|
Increase in inventories |
|
|
|
(152) |
|
(17) |
|
(47) |
|
|
Increase in trade and other receivables |
|
|
|
(108) |
|
(40) |
|
(74) |
|
|
Increase/(decrease) in trade and other payables and provisions |
|
|
|
71 |
|
(45) |
|
(49) |
|
|
Cash generated from operations1 |
|
|
|
1,108 |
|
1,273 |
|
1,035 |
|
|
Interest received |
|
|
|
2 |
|
2 |
|
3 |
|
|
Interest paid |
|
|
|
(54) |
|
(50) |
|
(48) |
|
|
Income taxes paid |
|
|
|
(125) |
|
(135) |
|
(141) |
|
|
Net cash inflow from operating activities |
|
|
|
931 |
|
1,090 |
|
849 |
|
|
Cash flows from investing activities |
|
|
|
|
|
|
|
|
|
|
Acquisitions, net of cash acquired |
|
21 |
|
(29) |
|
(159) |
|
(214) |
|
|
Capital expenditure |
|
2 |
|
(347) |
|
(376) |
|
(392) |
|
|
Purchase of investments |
|
10 |
|
(4) |
|
(8) |
|
(2) |
|
|
Distribution from associate |
|
11 |
|
2 |
|
– |
|
– |
|
|
Proceeds on disposal of business |
|
21 |
|
– |
|
– |
|
343 |
|
|
Tax on disposal of business |
|
|
|
– |
|
– |
|
(118) |
|
|
Net cash used in investing activities |
|
|
|
(378) |
|
(543) |
|
(383) |
|
|
Cash flows from financing activities |
|
|
|
|
|
|
|
|
|
|
Proceeds from issue of ordinary share capital |
|
|
|
3 |
|
5 |
|
10 |
|
|
Purchase of own shares |
|
|
|
(48) |
|
(52) |
|
(368) |
|
|
Proceeds from borrowings due within one year |
|
20 |
|
24 |
|
53 |
|
34 |
|
|
Settlement of borrowings due within one year |
|
20 |
|
(30) |
|
(64) |
|
(38) |
|
|
Proceeds from borrowings due after one year |
|
20 |
|
370 |
|
570 |
|
890 |
|
|
Settlement of borrowings due after one year |
|
20 |
|
(371) |
|
(706) |
|
(759) |
|
|
Proceeds from own shares |
|
|
|
10 |
|
5 |
|
6 |
|
|
Settlement of currency swaps |
|
20 |
|
(8) |
|
24 |
|
(25) |
|
|
Equity dividends paid |
|
19 |
|
(321) |
|
(269) |
|
(279) |
|
|
Net cash used in financing activities |
|
|
|
(371) |
|
(434) |
|
(529) |
|
|
Net increase/(decrease) in cash and cash equivalents |
|
|
|
182 |
|
113 |
|
(63) |
|
|
Cash and cash equivalents at beginning of year |
|
20 |
|
155 |
|
38 |
|
102 |
|
|
Exchange adjustments |
|
20 |
|
(4) |
|
4 |
|
(1) |
|
|
Cash and cash equivalents at end of year2 |
|
|
|
333 |
|
155 |
|
38 |
|
|
1 |
Includes $83m (2017: $15m, 2016: $62m) of outgoings on restructuring and rationalisation expenses, $3m (2017: $3m, 2016: $24m) of acquisition-related costs and $104m (2017: $25m, 2016: $36m) of legal and other costs. |
|
2 |
Cash and cash equivalents is net of bank overdrafts of $32m (2017: $14m, 2016: $62m). |
 The Notes on pages 129-177 are an integral part of these accounts.
The Notes on pages 129-177 are an integral part of these accounts.
|
128 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
GROUP FINANCIAL STATEMENTS
Group statement of changes in equity
|
|
|
|
|
|
|
Capital |
|
|
|
|
|
|
|
|
|
|
|
|
Share |
|
Share |
|
redemption |
|
Treasury |
|
Other |
|
Retained |
|
Total |
|
|
|
|
capital |
|
premium |
|
reserve |
|
shares2 |
|
reserves3 |
|
earnings |
|
equity |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 31 December 2015 |
|
183 |
|
590 |
|
12 |
|
(294) |
|
(256) |
|
3,731 |
|
3,966 |
|
|
Attributable profit for the year1 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
784 |
|
784 |
|
|
Other comprehensive expense |
|
– |
|
– |
|
– |
|
– |
|
(119) |
|
(71) |
|
(190) |
|
|
Equity dividends declared and paid |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(279) |
|
(279) |
|
|
Share-based payments recognised |
|
– |
|
– |
|
– |
|
– |
|
– |
|
27 |
|
27 |
|
|
Taxation on share-based payments |
|
– |
|
– |
|
– |
|
– |
|
– |
|
2 |
|
2 |
|
|
Purchase of own shares |
|
– |
|
– |
|
– |
|
(368) |
|
– |
|
– |
|
(368) |
|
|
Cost of shares transferred to beneficiaries |
|
– |
|
– |
|
– |
|
40 |
|
– |
|
(34) |
|
6 |
|
|
Cancellation of treasury shares |
|
(3) |
|
– |
|
3 |
|
190 |
|
– |
|
(190) |
|
– |
|
|
Issue of ordinary share capital4 |
|
– |
|
10 |
|
– |
|
– |
|
– |
|
– |
|
10 |
|
|
At 31 December 2016 |
|
180 |
|
600 |
|
15 |
|
(432) |
|
(375) |
|
3,970 |
|
3,958 |
|
|
Attributable profit for the year1 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
767 |
|
767 |
|
|
Other comprehensive income |
|
– |
|
– |
|
– |
|
– |
|
147 |
|
55 |
|
202 |
|
|
Equity dividends declared and paid |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(269) |
|
(269) |
|
|
Share-based payments recognised |
|
– |
|
– |
|
– |
|
– |
|
– |
|
31 |
|
31 |
|
|
Taxation on share-based payments |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(3) |
|
(3) |
|
|
Purchase of own shares |
|
– |
|
– |
|
– |
|
(52) |
|
– |
|
– |
|
(52) |
|
|
Cost of shares transferred to beneficiaries |
|
– |
|
– |
|
– |
|
26 |
|
– |
|
(21) |
|
5 |
|
|
Cancellation of treasury shares |
|
(2) |
|
– |
|
2 |
|
201 |
|
– |
|
(201) |
|
– |
|
|
Issue of ordinary share capital4 |
|
– |
|
5 |
|
– |
|
– |
|
– |
|
– |
|
5 |
|
|
At 31 December 2017 |
|
178 |
|
605 |
|
17 |
|
(257) |
|
(228) |
|
4,329 |
|
4,644 |
|
|
Adjustment on initial application of IFRS 9 (net of tax) |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(11) |
|
(11) |
|
|
Adjusted balance as at 1 January 2018 |
|
178 |
|
605 |
|
17 |
|
(257) |
|
(228) |
|
4,318 |
|
4,633 |
|
|
Attributable profit for the year1 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
663 |
|
663 |
|
|
Other comprehensive income |
|
– |
|
– |
|
– |
|
– |
|
(112) |
|
10 |
|
(102) |
|
|
Equity dividends declared and paid |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(321) |
|
(321) |
|
|
Share-based payments recognised |
|
– |
|
– |
|
– |
|
– |
|
– |
|
35 |
|
35 |
|
|
Taxation on share-based payments |
|
– |
|
– |
|
– |
|
– |
|
– |
|
1 |
|
1 |
|
|
Purchase of own shares |
|
– |
|
– |
|
– |
|
(48) |
|
– |
|
– |
|
(48) |
|
|
Cost of shares transferred to beneficiaries |
|
– |
|
– |
|
– |
|
40 |
|
– |
|
(30) |
|
10 |
|
|
Cancellation of treasury shares |
|
(1) |
|
– |
|
1 |
|
51 |
|
– |
|
(51) |
|
– |
|
|
Issue of ordinary share capital4 |
|
– |
|
3 |
|
– |
|
– |
|
– |
|
– |
|
3 |
|
|
At 31 December 2018 |
|
177 |
|
608 |
|
18 |
|
(214) |
|
(340) |
|
4,625 |
|
4,874 |
|
|
1 |
Attributable to equity holders of the Company and wholly derived from continuing operations. |
|
2 |
Refer to Note 19.2 for further information. |
|
3 |
Other reserves comprises gains and losses on cash flow hedges, foreign exchange differences on translation of foreign operations and net changes on fair value of trading investments. The cumulative translation loss within other reserves at 31 December 2018 was $339m (2017: $207m loss, 2016: $388m loss). |
|
4 |
Issue of ordinary share capital in connection with the Group’s share incentive plans. |
 The Notes on pages 129-177 are an integral part of these accounts.
The Notes on pages 129-177 are an integral part of these accounts.
|
|
Smith & Nephew Annual Report 2018 |
|
129 |
Smith & Nephew plc (the Company) is a public limited company incorporated in England and Wales. In these accounts, the ‘Group’ means the Company and all its subsidiaries. The principal activities of the Group are to develop, manufacture, market and sell medical devices and services.
As required by the European Union’s IAS Regulation and the Companies Act 2006, the Group has prepared its accounts in accordance with International Financial Reporting Standards (IFRS) as adopted by the European Union (EU) effective as at 31 December 2018. The Group has also prepared its accounts in accordance with IFRS as issued by the International Accounting Standards Board (IASB) effective as at 31 December 2018. IFRSs as adopted by the EU differs in certain respects from IFRS as issued by the IASB. However, the differences have no impact for the periods presented.
The preparation of accounts in conformity with IFRS requires management to use estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the accounts and the reported amounts of revenues and expenses during the year. The accounting policies requiring management to use significant estimates and assumptions are: inventories, impairment, taxation and liability provisions. These are discussed on page 124. Although these estimates are based on management’s best knowledge of current events and actions, actual results ultimately may differ from those estimates. Estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to estimates are recognised prospectively.
The Directors continue to adopt the going concern basis for accounting in preparing the annual financial statements. The Directors have a reasonable expectation that the Group has adequate resources to continue in operational existence for the foreseeable future. Further information regarding the Group’s business activities, together with the factors likely to affect its future development, performance and position, are set out on pages 10–22.
As described in Note 15, the Group meets its funding requirements through a mixture of shareholders’ funds, bank borrowings and private placement notes. At 31 December 2018 the Group had committed borrowing facilities of $2.4bn and total liquidity of $1.3bn, including net cash and cash equivalents of $333m and undrawn committed borrowing facilities of $1bn. The earliest expiry date of the Group’s committed borrowing facilities is in respect of $125m of Senior Notes due to expire in November 2019. In addition, Note 16 includes the Group’s objectives, policies and processes for managing its capital; our financial risk management objectives; details of our financial instruments and hedging activities; and our exposures to foreign exchange, interest rate and credit risk.
The Group’s forecasts and projections, taking into account reasonably possible changes in trading performance, show that the Group has sufficient financial resources. The Directors have reasonable expectation that the Company and the Group are well placed to manage their business risks and to continue in operational existence for a period of at least three years from the date of the approval of the financial statements. Accordingly, the Directors continue to adopt the going concern basis (in accordance with the guidance ‘Guidance on Risk Management, Internal Control and Related Financial and Business Reporting’ issued by the FRC) in preparing the consolidated financial statements.
New accounting standards effective 2018
IFRS 15 Revenue from contracts with customers
On 1 January 2018, the Group adopted IFRS 15 Revenue from contracts with customers using the modified retrospective method for contracts which were not completed as of that date. The Group applied the practical expedients in relation to contracts with variable consideration and contracts that were completed at the beginning of the earliest period presented and/or modified before the beginning of the earliest period presented.
Under IFRS 15, revenue is recognised as the performance obligations to deliver products or services are satisfied and revenue is recorded based on the amount of consideration expected to be received in exchange for satisfying the performance obligations. The Group undertook a detailed impact assessment applying IFRS 15 to all the existing ways in which the Group delivers products or services to customers to identify divergence with previous accounting practice governed by IAS 18 Revenue and concluded that IFRS 15 does not have a significant impact on the timing and recognition of revenue. Accordingly, there was no adjustment required on transition to IFRS 15.
IFRS 9 Financial Instruments
On 1 January 2018, the Group adopted IFRS 9 Financial Instruments. The Group has not restated comparative information for prior periods with respect to classification and measurement (including loss allowance) requirements.
The amendments to IFRS 9 mainly relate to the classification and measurement of financial instruments. IFRS 9 largely retains the existing requirements in IAS 39 Financial Instruments: Recognition and Measurement for the classification and measurement of financial liabilities; however, it eliminates the previous IAS 39 categories for financial assets of held to maturity, loans and receivables and available for sale. The Group elected, from 1 January 2018, to present changes in the fair value of trade investments in the income statement. The Group also elected to continue to apply the hedge accounting guidance in IAS 39 Financial Instruments: Recognition and Measurement.
|
130 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
With respect to loss allowances for trade receivables, IFRS 9 replaces the ‘incurred loss’ model in IAS 39 with an ‘expected credit loss’ (ECL) model. The Group, from 1 January 2018, has measured loss allowances for trade receivables at an amount equal to lifetime expected credit losses. In determining credit risk, the Group considers reasonable and supportable information that is relevant and available without undue cost or effort. This includes both quantitative and qualitative information and analysis based on the Group’s historical experience, and forward-looking information. The Group considers the model and some of the assumptions used in calculating these ECLs as sources of estimation uncertainty. The Group performed the calculation of ECL rates separately for customer groups which were segmented based on common risk characteristics such as credit risk grade and type of customer (for example government and non-government). While not material, the Group has determined that the application of IFRS 9 at 1 January 2018 results in an additional loss allowance for trade receivables of $14m. This transition adjustment gives rise to a deferred tax credit of $3m.
The following table and the accompanying notes below explain the original measurement categories under IAS 39 and the new measurement categories under IFRS 9 for each class of the Group’s financial assets and financial liabilities as at 1 January 2018.
The effect of adopting IFRS 9 on the carrying amounts of financial assets at 1 January 2018 relates solely to the loss allowance for trade receivables as described above.
|
|
|
|
|
|
|
Original carrying |
|
New carrying |
|
|
|
|
|
|
|
amount under IAS 39 |
|
amount under IFRS 9 |
|
|
|
Original classification under IAS 39 |
|
New classification under IFRS 9 |
|
$ million |
|
$ million |
|
Financial assets |
|
|
|
|
|
|
|
|
|
Forward foreign exchange contacts |
|
Fair value - hedging instrument |
|
Fair value - hedging instrument |
|
25 |
|
25 |
|
Investments |
|
Available for sale |
|
Fair value through profit or loss |
|
21 |
|
21 |
|
Currency swaps |
|
Fair value through profit or loss |
|
Fair value through profit or loss |
|
3 |
|
3 |
|
Trade and other receivables |
|
Loans & receivables |
|
Amortised cost |
|
1,148 |
|
1,134 |
|
Cash at bank |
|
Loans & receivables |
|
Amortised cost |
|
169 |
|
169 |
|
Total financial assets |
|
|
|
|
|
1,366 |
|
1,352 |
|
Financial liabilities |
|
|
|
|
|
|
|
|
|
Acquisition consideration |
|
Fair value through profit or loss |
|
Fair value through profit or loss |
|
(160) |
|
(160) |
|
Forward foreign exchange contracts |
|
Fair value - hedging instrument |
|
Fair value - hedging instrument |
|
(45) |
|
(45) |
|
Currency swaps |
|
Fair value through profit or loss |
|
Fair value through profit or loss |
|
(1) |
|
(1) |
|
Interest rate swaps |
|
Fair value - hedging instrument |
|
Fair value - hedging instrument |
|
(2) |
|
(2) |
|
Private placement debt |
|
Other financial liabilities |
|
Other financial liabilities |
|
(1,123) |
|
(1,123) |
|
Bank overdrafts |
|
Other financial liabilities |
|
Other financial liabilities |
|
(14) |
|
(14) |
|
Bank loans |
|
Other financial liabilities |
|
Other financial liabilities |
|
(313) |
|
(313) |
|
Trade and other payables |
|
Other financial liabilities |
|
Other financial liabilities |
|
(877) |
|
(877) |
|
Total financial liabilities |
|
|
|
|
|
(2,535) |
|
(2,535) |
A number of other new amendments to standards are effective from 1 January 2018 but they do not have a material effect on the Group’s financial statements.
Accounting standards issued but not yet effective
A number of new standards and amendments to standards are effective for annual periods beginning after 1 January 2019 and earlier application is permitted; however, the Group has not early adopted them in preparing these consolidated financial statements. These are not expected to have a significant impact on adoption, apart from IFRS 16 Leases which is described below.
The Group will adopt IFRS 16 using the modified retrospective approach and the right of use asset on transition will equal the lease liability. The cumulative effect of initially adopting the IFRS 16 will be recognised as an adjustment to retained earnings at 1 January 2019 with no restatement of comparative information. The Group plans to apply the practical expedient to grandfather the definition of a lease on transition. This means that it will apply IFRS 16 to all contracts entered into before 1 January 2019 and identified as leases in accordance with IAS 17 and IFRIC 4.
The Group intends to avail itself of the exemptions for short-term leases and leases of low-value items. The Group will recognise new assets and liabilities, primarily with regard to its operating leases of property and motor vehicles. In addition the Group will no longer recognise accruals relating to the straight-lining of rent expense for leases which include a rent-free period.
|
|
Smith & Nephew Annual Report 2018 |
|
131 |
The Group has designed a new lease accounting process and has implemented a new lease accounting software solution. The Group has assessed the estimated impact that initial application of IFRS 16 will have on its consolidated financial statements, as described below. The actual impacts of adopting the standard on 1 January 2019 may change because the Group has not finalised the testing and assessment of controls over its new lease accounting process, and the new accounting policies are subject to change until the Group presents its first financial statements that include the initial application of the standard.
Based on the information currently available, the Group estimates that it will recognise additional lease assets and liabilities of $145m to $165m as at 1 January 2019. The Group does not expect the adoption of IFRS 16 to have a material impact on the income statement.
1.1 Consolidation
The Group accounts include the accounts of Smith & Nephew plc and its subsidiaries for the periods during which they were members of the Group.
Subsidiaries are entities controlled by the Group. The Group controls an entity when it is exposed to, or has rights to, variable returns from its involvement with the entity and has the ability to affect those returns through its power over the entity. Subsidiaries are consolidated in the Group accounts from the date that the Group obtains control, and continue to be consolidated until the date that such control ceases. Intra-group balances and transactions, and any unrealised income and expenses arising from intra-group transactions, are eliminated on consolidation. All subsidiaries have year ends which are co-terminus with the Group’s, with the exception of jurisdictions whereby a different year end is required by local legislation.
When the Group loses control over a subsidiary, it derecognises the assets and liabilities of the subsidiary and any related components of equity. Any resulting gain or loss is recognised in profit or loss. Any retained interest in the former subsidiary is measured at fair value.
1.2 Foreign currencies
Functional and presentation currency
The Group accounts are presented in US Dollars. The Company’s functional currency is US Dollars.
Foreign currency transactions
Transactions in foreign currencies are translated to the respective functional currencies of Group companies at exchange rates at the dates of the transactions. Monetary assets and liabilities denominated in foreign currencies are retranslated to the functional currency as at the exchange rate at the reporting date. Non-monetary items are not retranslated.
Foreign operations
Balance sheet items of foreign operations, including goodwill and fair value adjustments arising on acquisition are translated into US Dollars on consolidation at the exchange rates at the reporting date. Income statement items and the cash flows of foreign operations are translated at average rates as an approximation to actual transaction rates, with actual transaction rates used for large one off transactions.
Foreign currency differences are recognised in ‘Other comprehensive income’ and accumulated in ‘Other reserves’ within equity. These include: exchange differences on the translation at closing rates of exchange of non-US Dollar opening net assets; the differences arising between the translation of profits into US Dollars at actual (or average, as an approximation) and closing exchange rates; to the extent that the hedging relationship is effective, the difference on translation of foreign currency borrowings or swaps that are used to finance or hedge the Group’s net investments in foreign operations; and the movement in the fair value of forward foreign exchange contracts used to hedge forecast foreign exchange cash flows.
The exchange rates used for the translation of currencies into US Dollars that have the most significant impact on the Group results were:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
Average rates |
|
|
|
|
|
|
|
|
Sterling |
|
1.33 |
|
1.29 |
|
1.35 |
|
|
Euro |
|
1.18 |
|
1.13 |
|
1.11 |
|
|
Swiss Franc |
|
1.02 |
|
1.02 |
|
1.02 |
|
|
Year end rates |
|
|
|
|
|
|
|
|
Sterling |
|
1.28 |
|
1.35 |
|
1.23 |
|
|
Euro |
|
1.14 |
|
1.20 |
|
1.05 |
|
|
Swiss Franc |
|
1.02 |
|
1.02 |
|
0.98 |
|
|
132 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
2 BUSINESS SEGMENT INFORMATION
The Group was engaged in a single business activity, being the development, manufacture and sales of medical technology products and services.
Development, manufacturing, supply chain and central functions are managed globally for the Group as a whole. Sales were managed through two geographical selling regions, US and International, with a president for each who was responsible for the commercial review of that region. The Executive Committee (‘ExCo’), comprises geographical presidents and certain heads of function and is chaired by the Chief Executive Officer (‘CEO’). ExCo is the body through which the CEO uses the authority delegated to him by the Board of Directors to manage the operations and performance of the Group. All significant operating decisions regarding the allocation and prioritisation of the Group’s resources and assessment of the Group’s performance were made by ExCo, and whilst the members have individual responsibility for the implementation of decisions within their respective areas, it was at the ExCo level that these decisions are made. Accordingly, ExCo was considered to be the Group’s chief operating decision maker as defined by IFRS 8 Operating Segments.
In making decisions about the prioritisation and allocation of the Group’s resources, ExCo reviews financial information on an integrated basis for the Group as a whole and determines the best allocation of resources to Group-wide projects. This information is prepared substantially on the same basis as the Group’s IFRS financial statements aside from the adjustments described in Note 2.2. In assessing performance, ExCo also considers financial information presented on a geographical selling region and product franchise basis for revenue. Financial information for corporate and functional costs is presented on a Group-wide basis.
The types of products and services offered by the Group’s global business segment are as follows:
|
- |
Knee Implants, which offers an innovative range of products for specialised knee replacement procedures; |
|
- |
Hip Implants, which offers a range of specialist products for reconstruction of the hip joint; |
|
- |
Trauma & Extremities, consisting of internal and external devices used in the stabilisation of severe fractures and deformity correction procedures; |
|
- |
Sports Medicine Joint Repair, which offers surgeons a broad array of instruments, technologies and implants necessary to perform minimally invasive surgery of the joints; |
|
- |
Arthroscopic Enabling Technologies, which offers healthcare providers a variety of technologies such as fluid management equipment for surgical access, high definition cameras, digital image capture, scopes, light sources and monitors to assist with visualisation inside the joints, radio frequency, electromechanical and mechanical tissue resection devices, and hand instruments for removing damaged tissue; |
|
- |
Other Surgical Businesses, which includes robotics-assisted surgery, various products and technologies to assist in surgical treatment of the ear, nose and throat, and gynaecological instrumentation, until the Gynaecology business disposal in August 2016; |
|
- |
Advanced Wound Care, which includes products for the treatment and prevention of acute and chronic wounds, including leg, diabetic and pressure ulcers, burns and post-operative wounds; |
|
- |
Advanced Wound Bioactives, which includes biologics and other bioactive technologies that provide unique approaches to debridement and dermal repair/regeneration; and |
|
- |
Advanced Wound Devices, which consists of traditional and single-use Negative Pressure Wound Therapy and hydrosurgery systems. |
The segment information is prepared in conformity with the accounting policies of the Group and the accounting standard IFRS 8 Operating Segments.
The segment profit measure reported to the Chief Executive Officer and the ExCo for the purposes of resource allocation and assessment is trading profit and excludes the effects of non-recurring, infrequent, non-cash and other items that management does not otherwise believe are indicative of the underlying performance of the consolidated Group as discussed in Note 2.2. Group financing (including interest receivable and payable) is managed on a net basis outside the business segment.
During 2018, the Group began its transition to a global franchise structure with three dedicated franchises for Orthopaedics, Sports Medicine & ENT, and Advanced Wound Management, each with their own president. From 2019 onwards, with the Group’s operating structure organised around three franchises, the chief operating decision maker will be able to monitor performance, make operating decisions and allocate resources on a global franchise basis in contrast with 2018 and prior, where these were done on a Group-wide basis. The franchise presidents will be responsible for implementing the operating decisions for their respective franchise in the US. Regional presidents in EMEA and APAC will be responsible for this implementation in their respective regions. Based on the aforementioned changes from January 2019, the Group has concluded that there will be three reportable segments from this date. The Group will not restate comparative information in 2019, other than revenue, as the Group was managed as one operating segment in 2018 and 2017, and historical financial information is not available on a franchise basis. The Group does have sufficient historical financial information of revenue by franchise to restate the comparative information of revenue by operating segment.
|
|
Smith & Nephew Annual Report 2018 |
|
133 |
2.1 Revenue by business segment and geography
|
Revenue is recognised as the performance obligations to deliver products or services are satisfied and is recorded based on the amount of consideration expected to be received in exchange for satisfying the performance obligations. Revenue is recognised primarily when control is transferred to the customer, which is generally when the goods are shipped or delivered in accordance with the contract terms, with some transfer of services taking place over time. Substantially all performance obligations are performed within one year. There is no significant revenue associated with the provision of discrete services. Payment terms to our customers are based on commercially reasonable terms for the respective markets while also considering a customer’s credit rating. Appropriate provisions for returns, trade discounts and rebates are deducted from revenue. Rebates primarily comprise chargebacks and other discounts granted to certain customers. Chargebacks are discounts that occur when a contracted customer purchases directly through an intermediary wholesaler. The contracted customer generally purchases product at its contracted price plus a mark-up from the wholesaler. The wholesaler in turn charges the Group for the difference between the price initially paid by the wholesaler and the contract price paid to the wholesaler by the customer. The provision for chargebacks is based on expected sell-through levels by the Group’s wholesale customers to contracted customers, as well as estimated wholesaler inventory levels. The revenue accounting policy for the year ending 31 December 2017 was consistent with the requirements of IAS 18. Revenue was recognised once the significant risks and rewards of ownership had been transferred to the customer, rather than the satisfaction of the performance obligations to deliver products or services. Reconstruction, Sports Medicine, Trauma & Other Reconstruction, Sports Medicine, Trauma & Other consists of the following franchises: Knee Implants and Hip Implants, Sports Medicine Joint Repair, Arthroscopic Enabling Technologies, Trauma & Extremities and Other Surgical Business. Sales of inventory located at customer premises and available for customers’ immediate use are recognised when notification is received that the product has been implanted or used. Substantially all other revenue is recognised when control is transferred to the customer, which is generally when the goods are shipped or delivered in accordance with the contract terms. Revenue is recognised for the amount of consideration expected to be received in exchange for transferring the products or services. In general our Reconstruction, Sports Medicine, Trauma & Other business in Established Markets is direct to hospitals and ambulatory surgery centres whereas in the Emerging Markets we generally sell through distributors. Advanced Wound Management Advanced Wound Management consists of the following product franchises: Advanced Wound Care, Advanced Wound Bioactives and Advanced Wound Devices. Substantially all revenue is recognised when control is transferred to the customer, which is generally when the goods are shipped or delivered in accordance with the contract terms. Revenue is recognised for the amount of consideration expected to be received in exchange for transferring the products or services. Appropriate provisions for returns, trade discounts and rebates are deducted from revenue, as explained above. The majority of our Advanced Wound Management business, and in particular products used in community and homecare facilities, is through wholesalers and distributors. The proportion of sales direct to hospitals is higher in our Advanced Wound Devices business in Established Markets. |
Segment revenue reconciles to statutory revenues from continuing operations as follows:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Reportable segment revenue |
|
|
|
|
|
|
|
|
Revenue from external customers |
|
4,904 |
|
4,765 |
|
4,669 |
|
|
134 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
Disaggregation of revenue:
The following table shows the disaggregation of Group revenue by product franchise:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Revenue by product from continuing operations |
|
|
|
|
|
|
|
|
Knee Implants |
|
1,017 |
|
984 |
|
932 |
|
|
Hip Implants |
|
613 |
|
599 |
|
597 |
|
|
Trauma & Extremities |
|
493 |
|
495 |
|
475 |
|
|
Sports Medicine Joint Repair |
|
697 |
|
627 |
|
587 |
|
|
Arthroscopic Enabling Technologies |
|
600 |
|
615 |
|
631 |
|
|
Other Surgical Businesses |
|
209 |
|
189 |
|
214 |
|
|
Advanced Wound Care |
|
740 |
|
720 |
|
719 |
|
|
Advanced Wound Bioactives |
|
320 |
|
342 |
|
342 |
|
|
Advanced Wound Devices |
|
215 |
|
194 |
|
172 |
|
|
Consolidated revenue from continuing operations |
|
4,904 |
|
4,765 |
|
4,669 |
|
The following table shows the disaggregation of Group revenue by geographic market and product category. The disaggregation of revenue into the two product categories below reflects that in general the products in the Advanced Wound Management franchises are sold to wholesalers and intermediaries, while products in the other franchises are sold directly to hospitals, ambulatory surgery centres and distributors. The further disaggregation of revenue by Established Markets and Emerging Markets reflects that in general our products are sold through distributors and intermediaries in the Emerging Markets while in the Established Markets, with the exception of the Advanced Wound Care and Bioactives franchises, products are in general sold direct to hospitals and ambulatory surgery centres. The disaggregation by Established Markets and Emerging Markets also reflects their differing economic factors including volatility in growth and outlook.
|
|
|
|
|
|
|
2018 |
|
|
|
|
|
2017 |
|
|
|
|
|
2016 |
|
|
|
Established Markets1 |
|
Emerging |
|
Total |
|
Established Markets1 |
|
Emerging |
|
Total |
|
Established Markets1 |
|
Emerging |
|
Total |
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
Reconstruction, Sports Medicine, Trauma & Other Surgical Businesses |
|
2,944 |
|
685 |
|
3,629 |
|
2,867 |
|
642 |
|
3,509 |
|
2,868 |
|
568 |
|
3,436 |
|
Advanced Wound Management |
|
1,103 |
|
172 |
|
1,275 |
|
1,097 |
|
159 |
|
1,256 |
|
1,092 |
|
141 |
|
1,233 |
|
Total |
|
4,047 |
|
857 |
|
4,904 |
|
3,964 |
|
801 |
|
4,765 |
|
3,960 |
|
709 |
|
4,669 |
|
1 |
Established Markets comprises the US, Australia, Canada, Europe, Japan and New Zealand. |
In 2018, the Group has presented sales attributed to the country of destination rather than based on the location of the Group’s businesses. Comparatives have been presented on a consistent basis with 2018. US revenue for 2018 was $2,354m (2017: $2,306m, 2016: $2,299m) and UK revenue for 2018 was $211m (2017: $222m, 2016: $266m).
Contract assets and liabilities
The nature of our products and services do not generally give rise to contract assets as we do not typically incur costs to fulfil a contract before a product or service is provided to the customer. The Group generally satisfies performance obligations within one year from the contract inception date. There was no material revenue recognised in the current reporting period that related to carried-forward contract liabilities (deferred income) or performance obligations satisfied in the previous year. There is no material revenue that is likely to arise in future periods from unsatisfied performance obligations at the balance sheet date. Therefore, there are no associated significant accrued income and deferred income balances at 31 December 2018. As of 31 December 2018, contract assets principally comprised trade receivables and contract liabilities principally comprise rebates (as described in the accounting policy above). The accrual for rebates at 31 December 2018 was $65m (2017: $56m) with $334m being recognised in revenue in 2018.
Major customers
No single customer generates revenue greater than 10% of the consolidated revenue.
|
|
Smith & Nephew Annual Report 2018 |
|
135 |
2.2 Trading and operating profit by business segment
Trading profit is a trend measure which presents the long-term profitability of the Group excluding the impact of specific transactions that management considers affect the Group’s short-term profitability. The Group presents this measure to assist investors in their understanding of trends. The Group has identified the following items, where material, as those to be excluded from operating profit when arriving at trading profit: acquisition and disposal-related items; amortisation and impairment of acquisition intangibles; significant restructuring programmes; gains and losses arising from legal disputes; and other significant items. Further detail is provided in Notes 2.3, 2.4 and 2.5. Operating profit reconciles to trading profit as follows:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Operating profit of the business segment |
|
863 |
|
934 |
|
801 |
|
|
Acquisition and disposal related items |
|
(7) |
|
(10) |
|
9 |
|
|
Restructuring and rationalisation costs |
|
120 |
|
– |
|
62 |
|
|
Amortisation and impairment of acquisition intangibles |
|
113 |
|
140 |
|
178 |
|
|
Legal and other |
|
34 |
|
(16) |
|
(30) |
|
|
Trading profit of the business segment |
|
1,123 |
|
1,048 |
|
1,020 |
|
2.3 Acquisition and disposal related items
For the year to 31 December 2018 the credit relates to a remeasurement of contingent consideration for a prior year acquisition and adjustments to provisions on disposal of a business, partially offset by costs associated with the acquisition of Rotation Medical, Inc.
For the year to 31 December 2017 the credit relates to a remeasurement of contingent consideration for a prior year acquisition partially offset by costs associated with the acquisition of Rotation Medical, Inc. For the year to 31 December 2016, these costs relate to the costs associated with the integration of Blue Belt Technologies and other acquisitions.
2.4 Restructuring and rationalisation costs
Restructuring and rationalisation costs of $120m were incurred in 2018 (2017: $nil, 2016: $62m) primarily relating to the implementation of the Accelerating Performance and Execution (APEX) programme that was announced in February 2018. In 2016, these costs primarily related to the Group Optimisation Plan.
2.5 Legal and other
For the year ended 31 December 2018 charges primarily relate to legal expenses for ongoing metal-on-metal hip claims and an increase of $72m in the provision that reflects the present value of the estimated costs to resolve all other known and anticipated metal-on-metal hip claims globally. The year to 31 December 2018 also includes costs for implementing the requirements of the EU Medical Device Regulations that will apply from May 2020. These charges in the year to 31 December 2018 were partially offset by a credit of $84m relating to settlement agreements with insurers related to product liability claims involving macrotextured components withdrawn from the market in 2003.
For the year ended 31 December 2017 charges primarily relate to legal expenses for patent litigation with Arthrex and ongoing metal-on-metal hip claims and an increase of $10m in the provision that reflects the present value of the estimated costs to resolve all other known and anticipated metal-on-metal hip claims globally. These charges were offset by a $54m credit following a settlement payment received from Arthrex relating to patent litigation. For the year ended 31 December 2016, the net credit of $30m primarily relates to a $44m curtailment credit on post-retirement benefits in the UK pension scheme partially offset by legal expenses incurred for patent litigation with Arthrex. Also included in a net $1m credit in respect of insurance recoveries of $24m and legal expenses of $23m, relating to the ongoing metal-on-metal hip claims globally.
2.6 Assets and liabilities by business segment and geography
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Reconciliation of assets of the business segment to the consolidated Group |
|
|
|
|
|
|
|
|
Assets of the business segment |
|
7,476 |
|
7,508 |
|
7,147 |
|
|
Unallocated corporate assets: |
|
|
|
|
|
|
|
|
Deferred tax assets |
|
126 |
|
127 |
|
97 |
|
|
Retirement benefit assets |
|
92 |
|
62 |
|
– |
|
|
Cash at bank |
|
365 |
|
169 |
|
100 |
|
|
Total assets of the consolidated Group |
|
8,059 |
|
7,866 |
|
7,344 |
|
|
136 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
In presenting information on the basis of geographical segments, non-current segment assets are based on their location:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Geographical segment assets |
|
|
|
|
|
|
|
|
United Kingdom |
|
354 |
|
364 |
|
335 |
|
|
United States of America |
|
3,186 |
|
3,295 |
|
3,145 |
|
|
Other |
|
1,224 |
|
1,287 |
|
1,238 |
|
|
Total non-current assets of the consolidated Group1 |
|
4,764 |
|
4,946 |
|
4,718 |
|
|
1 |
Non-current assets excludes retirement benefit assets and deferred tax assets. |
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Reconciliation of liabilities of the business segment to the consolidated Group |
|
|
|
|
|
|
|
|
Liabilities of the business segment |
|
1,284 |
|
1,311 |
|
1,247 |
|
|
Unallocated corporate liabilities: |
|
|
|
|
|
|
|
|
Long-term borrowings |
|
1,301 |
|
1,423 |
|
1,564 |
|
|
Retirement benefit obligations |
|
114 |
|
131 |
|
164 |
|
|
Deferred tax liabilities |
|
99 |
|
97 |
|
94 |
|
|
Bank overdrafts, borrowings and loans - current |
|
164 |
|
27 |
|
86 |
|
|
Current tax payable |
|
223 |
|
233 |
|
231 |
|
|
Total liabilities of the consolidated Group |
|
3,185 |
|
3,222 |
|
3,386 |
|
|
|
|
|
|
|
|
|
|
|
Depreciation, amortisation and impairment of the business segment |
|
|
|
|
|
|
|
|
Depreciation of property, plant and equipment |
|
251 |
|
243 |
|
224 |
|
|
Amortisation of acquired intangibles |
|
113 |
|
130 |
|
130 |
|
|
Amortisation of other intangible assets |
|
63 |
|
62 |
|
61 |
|
|
Total depreciation and amortisation |
|
427 |
|
435 |
|
415 |
|
|
Impairment losses on property, plant and equipment1 |
|
5 |
|
– |
|
– |
|
|
Impairment losses on acquisition intangibles1 |
|
– |
|
10 |
|
48 |
|
|
Impairment losses on other intangible assets1 |
|
3 |
|
– |
|
– |
|
|
Impairment losses on trade investments1 |
|
– |
|
2 |
|
– |
|
|
Total depreciation, amortisation and impairment |
|
435 |
|
447 |
|
463 |
|
|
1 |
Impairments recognised in operating profit, within the administrative expenses line. |
Segment acquisition of property, plant and equipment and intangibles reconciles to that of the consolidated group, and comprises the following:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Additions to property, plant and equipment |
|
332 |
|
308 |
|
320 |
|
|
Additions to intangibles |
|
15 |
|
68 |
|
72 |
|
|
Capital expenditure (excluding business combinations) |
|
347 |
|
376 |
|
392 |
|
|
Trade investments |
|
4 |
|
8 |
|
2 |
|
|
Acquisitions – Goodwill |
|
– |
|
132 |
|
211 |
|
|
Acquisitions – Intangible assets |
|
– |
|
61 |
|
85 |
|
|
Acquisitions – Property, plant and equipment |
|
– |
|
1 |
|
2 |
|
|
Capital and acquisition expenditure |
|
351 |
|
578 |
|
692 |
|
|
|
Smith & Nephew Annual Report 2018 |
|
137 |
|
Accounting policies Research expenditure is expensed as incurred. Internal development expenditure is only capitalised if the recognition criteria in IAS 38 Intangible Assets have been satisfied. The Group considers that the regulatory, technical and market uncertainties inherent in the development of new products mean that in most cases development costs should not be capitalised as intangible assets until products receive approval from the appropriate regulatory body. Payments to third parties for research and development projects are accounted for based on the substance of the arrangement. If the arrangement represents outsourced research and development activities the payments are generally expensed except in limited circumstances where the respective development expenditure would be capitalised under the principles established in IAS 38. By contrast, the payments are capitalised if the arrangement represents consideration for the acquisition of intellectual property developed at the risk of the third party. Capitalised development expenditures are amortised on a straight-line basis over their useful economic lives from product launch. Advertising costs Advertising costs are expensed as incurred. |
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Revenue |
|
4,904 |
|
4,765 |
|
4,669 |
|
|
Cost of goods sold1 |
|
(1,298) |
|
(1,248) |
|
(1,272) |
|
|
Gross profit |
|
3,606 |
|
3,517 |
|
3,397 |
|
|
Research and development expenses2 |
|
(246) |
|
(223) |
|
(230) |
|
|
Selling, general and administrative expenses: |
|
|
|
|
|
|
|
|
Marketing, selling and distribution expenses |
|
(1,820) |
|
(1,781) |
|
(1,712) |
|
|
Administrative expenses3,4,5,6 |
|
(677) |
|
(579) |
|
(654) |
|
|
|
|
(2,497) |
|
(2,360) |
|
(2,366) |
|
|
Operating profit |
|
863 |
|
934 |
|
801 |
|
|
1 |
2018 includes $4m charge relating to legal and other items (2017: $nil, 2016: $nil). |
|
2 |
2018 includes $9m charge relating to legal and other items (2017: $nil, 2016: $nil). |
|
3 |
2018 includes $63m of amortisation of software and other intangible assets (2017: $62m, 2016: $61m). |
|
4 |
2018 includes $113m of amortisation and impairment of acquisition intangibles and $120m of restructuring and rationalisation expenses (2017: $140m of amortisation and impairment of acquisition intangibles, 2016: $62m of restructuring and rationalisation expenses and $178m of amortisation and impairment of acquisition intangibles). |
|
5 |
2018 includes $21m charge relating to legal and other items (2017: $16m credit, 2016: $30m credit). |
|
6 |
2018 includes $7m credit of acquisition and disposal related items (2017: $10m credit, 2016: $9m charge). |
Note that items detailed in 1,2,4,5 and 6 are excluded from the calculation of trading profit, the segment profit measure.
Operating profit is stated after charging/(crediting) the following items:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Other operating income |
|
(107) |
|
(66) |
|
(25) |
|
|
Amortisation of intangibles |
|
176 |
|
192 |
|
191 |
|
|
Impairment of intangible assets |
|
3 |
|
10 |
|
48 |
|
|
Impairment of property, plant and equipment |
|
5 |
|
– |
|
– |
|
|
Depreciation of property, plant and equipment |
|
251 |
|
243 |
|
224 |
|
|
Loss on disposal of property, plant and equipment and intangible assets |
|
19 |
|
13 |
|
15 |
|
|
Operating lease payments for land and buildings |
|
32 |
|
36 |
|
39 |
|
|
Operating lease payments for other assets |
|
25 |
|
21 |
|
19 |
|
|
Advertising costs |
|
88 |
|
102 |
|
88 |
|
In 2018 other operating income primarily comprises an insurance recovery relating to product liability claims involving macrotextured components voluntarily withdrawn from the market in 2003 and a gain relating to patent litigation (2017: gain relating to patent litigation, 2016: insurance recovery relating to metal-on-metal claims). In 2018, $84m (2017: $54m, 2016: $24m) of other operating income was included with legal and other items, as explained in Note 2.5, and does not form part of the segment trading profit.
|
138 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
3 OPERATING PROFIT continued
3.1 Staff costs and employee numbers
Staff costs during the year amounted to:
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
$ million |
|
|
Wages and salaries1 |
|
|
|
1,330 |
|
1,231 |
|
1,227 |
|
|
Social security costs |
|
|
|
176 |
|
178 |
|
129 |
|
|
Pension costs (including retirement healthcare)2 |
|
18 |
|
65 |
|
64 |
|
23 |
|
|
Share-based payments |
|
23 |
|
35 |
|
31 |
|
27 |
|
|
|
|
|
|
1,606 |
|
1,504 |
|
1,406 |
|
|
1 |
The 2017 wages and salaries cost has been amended from $1,157m to $1,231m. This amendment has no impact on the Group Income Statement, Group Statement of Comprehensive Income, Group Balance Sheet, Group Cash Flow Statement and Group Statement of Changes in Equity for any period presented. |
|
2 |
In 2016, pension costs include the past service cost credit of $49m arising primarily from the closure of the UK defined benefit scheme to future accrual. |
During the year ended 31 December 2018, the average number of employees was 16,681 (2017: 16,333, 2016: 15,584).
3.2 Audit Fees – information about the nature and cost of services provided by the auditor
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Audit services: |
|
|
|
|
|
|
|
|
Group accounts |
|
2.6 |
|
2.4 |
|
2.0 |
|
|
Local statutory audit pursuant to legislation |
|
3.4 |
|
2.0 |
|
2.0 |
|
|
Other services: |
|
|
|
|
|
|
|
|
Non-audit services |
|
– |
|
0.1 |
|
0.5 |
|
|
Taxation services: |
|
|
|
|
|
|
|
|
Compliance services |
|
– |
|
– |
|
0.1 |
|
|
Advisory services |
|
– |
|
– |
|
– |
|
|
Total auditor’s remuneration |
|
6.0 |
|
4.5 |
|
4.6 |
|
|
Arising: |
|
|
|
|
|
|
|
|
In the UK |
|
2.4 |
|
2.5 |
|
2.5 |
|
|
Outside the UK |
|
3.6 |
|
2.0 |
|
2.1 |
|
|
|
|
6.0 |
|
4.5 |
|
4.6 |
|
4 INTEREST AND OTHER FINANCE COSTS
4.1 Interest income/(expense)
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Interest income |
|
8 |
|
6 |
|
6 |
|
|
Interest expense: |
|
|
|
|
|
|
|
|
Bank borrowings |
|
(11) |
|
(11) |
|
(9) |
|
|
Private placement notes |
|
(38) |
|
(38) |
|
(37) |
|
|
Other |
|
(10) |
|
(8) |
|
(6) |
|
|
|
|
(59) |
|
(57) |
|
(52) |
|
|
Net interest expense |
|
(51) |
|
(51) |
|
(46) |
|
|
|
Smith & Nephew Annual Report 2018 |
|
139 |
4.2 Other finance costs
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
$ million |
|
|
Retirement benefit net interest expense |
|
18 |
|
(3) |
|
(5) |
|
(7) |
|
|
Unwinding of discount |
|
|
|
(9) |
|
(5) |
|
(9) |
|
|
Other |
|
|
|
(8) |
|
— |
|
– |
|
|
Other finance costs |
|
|
|
(20) |
|
(10) |
|
(16) |
|
Foreign exchange gains or losses arose primarily on the translation of intercompany and third party borrowings and amounted to a net $11m gain in 2018 (2017: net $25m loss, 2016: net $22m gain). These amounts were matched by the fair value gains or losses on currency swaps held to manage this currency risk.
|
Accounting policy The charge for current taxation is based on the results for the year as adjusted for items which are non-assessable or disallowed. It is calculated using tax rates that have been enacted or substantively enacted by the balance sheet date. The Group operates in numerous tax jurisdictions around the world and it is Group policy to submit its tax returns to the relevant tax authorities as promptly as possible. At any given time, the Group is involved in disputes and tax audits and will have a number of tax returns potentially subject to audit. Significant issues may take several years to resolve. In estimating the probability and amount of any tax charge, management takes into account the views of internal and external advisers and updates the amount of tax provision whenever necessary. The ultimate tax liability may differ from the amount provided depending on factors including interpretations of tax law, settlement negotiations or changes in legislation. Deferred tax is recognised in respect of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for taxation purposes. Deferred tax is not recognised: for temporary differences related to investments in subsidiaries and associates where the Group is able to control the timing of the reversal of the temporary difference and it is probable that this will not reverse in the foreseeable future; on the initial recognition of non-deductible goodwill; and on the initial recognition of an asset or liability in a transaction that is not a business combination and that, at the time of the transaction, does not affect the accounting or taxable profit. Deferred tax assets are recognised to the extent that it is probable that future taxable profits will be available against which they can be used. Deferred tax assets are reviewed at each reporting date. Deferred tax is measured on an undiscounted basis, and at the tax rates that have been enacted or substantively enacted by the reporting date that are expected to apply in the periods in which the asset or liability is settled. It is recognised in the income statement except when it relates to items credited or charged directly to other comprehensive income or equity, in which case the deferred tax is also recognised within other comprehensive income or equity respectively. Deferred tax assets and liabilities are offset when they relate to income taxes levied by the same taxation authority, when the Group intends to settle its current tax assets and liabilities on a net basis and that authority permits the Group to make a single net payment. |
|
140 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
5.1 Taxation charge attributable to the Group
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Current taxation: |
|
|
|
|
|
|
|
|
UK corporation tax |
|
27 |
|
23 |
|
23 |
|
|
Overseas tax |
|
131 |
|
177 |
|
261 |
|
|
Current income tax charge |
|
158 |
|
200 |
|
284 |
|
|
Adjustments in respect of prior periods |
|
(33) |
|
(60) |
|
(53) |
|
|
Total current taxation |
|
125 |
|
140 |
|
231 |
|
|
Deferred taxation: |
|
|
|
|
|
|
|
|
Origination and reversal of temporary differences |
|
(3) |
|
32 |
|
24 |
|
|
Changes in tax rates |
|
1 |
|
(49) |
|
– |
|
|
Adjustments to estimated amounts arising in prior periods |
|
(5) |
|
(11) |
|
23 |
|
|
Total deferred taxation |
|
(7) |
|
(28) |
|
47 |
|
|
Total taxation as per the income statement |
|
118 |
|
112 |
|
278 |
|
|
Taxation in other comprehensive income |
|
(4) |
|
9 |
|
(10) |
|
|
Taxation in equity |
|
(1) |
|
3 |
|
(2) |
|
|
Taxation attributable to the Group |
|
113 |
|
124 |
|
266 |
|
The 2018 net prior period adjustment benefits of $38m mainly relate to the expiry of statute of limitations and tax accrual to tax return adjustments, partially offset by an increase in certain other tax provisions. The 2017 and 2016 net prior period adjustment benefits of $71m and $30m respectively mainly relate to provision releases following agreement reached with the IRS on US tax matters after the conclusion of US tax audits in 2017 and 2016, provision releases on the expiry of statute of limitations, and tax accrual to tax return adjustments, partially offset by an increase in certain other tax provisions.
Included in the total 2017 tax charge is a $32m net benefit as a result of the US tax reform legislation enacted in December 2017, which comprises a benefit from a revaluation of deferred tax balances included within changes in tax rates, partially offset by a current tax charge relating to the deemed repatriation of foreign profits not previously taxed in the US.
Total taxation as per the income statement of $118m includes a $51m net credit (2017: $58m net credit, 2016: $48m net charge) as a consequence of restructuring and rationalisation costs, acquisitions and disposals, amortisation and impairment of acquisition intangibles, and legal and other items. In 2017, the net credit included a net benefit from US tax reform.
Factors affecting future tax charges
The Group operates in numerous tax jurisdictions around the world and is subject to factors that may affect future tax charges including the review by the European Commission into whether the UK CFC financing exemption rules constitute illegal State Aid (see below), transfer pricing, tax rate changes, tax legislation changes, tax authority interpretation, expiry of statute of limitations and resolution of tax audits and disputes.
At any given time the Group has unagreed years outstanding in various countries and is involved in tax audits and disputes, some of which may take several years to resolve. Provisions are based on best estimates and management’s judgements concerning the likely ultimate outcome of the audit or dispute. Management consider the specific circumstances of each tax position and takes external advice, where appropriate, to assess the range of potential outcomes and estimate additional tax that may be due. Current tax payable of $223m (2017: $233m) includes $178m (2017: $201m) of provisions, penalties and interest for uncertain tax positions which relate to multiple issues across the jurisdictions in which the Group operates. Other creditors includes $8m of other interest on these provisions. The anticipated impact of IFRIC 23 is not expected to give rise to a material difference to the tax risk provisions.
|
|
Smith & Nephew Annual Report 2018 |
|
141 |
The Group believes that it has made adequate provision in respect of additional tax liabilities that may arise from unagreed years and tax audits and disputes, the majority of which relate to transfer pricing matters as would be expected for a Group operating internationally. However, the actual liability for any particular issue may be higher or lower than the amount provided, resulting in a negative or positive effect on the tax charge in any given year. A reduction in the tax charge may also arise for other reasons such as an expiry of the relevant statute of limitations. Whilst the impact can vary from year-to-year, we believe the possibility of a material adverse impact on the tax charge for 2019 is remote. Depending on the final outcome of certain tax audits which are currently in progress, possible statute of limitations expiry and other factors, a credit to the tax charge at or around similar levels in recent years could arise. In respect of the risks provided for at 31 December 2018, we do not envisage circumstances that would give rise to a release of more than approximately half of the provisions held.
As referenced in our 2017 Annual Report, one of the factors that may affect our future tax charge is the review by the European Commission (EC) into whether the UK CFC financing exemption rules between 2013 and 2018 constituted illegal State Aid. The EC issued its preliminary view in October 2017 that the financing exemption did constitute State Aid and their final decision, following their investigation, is expected in 2019. Depending on the outcome, this may then be subject to a further legal process. The financing exemption was introduced into UK legislation by the British government in 2013. In line with many other UK-based international groups which have followed this legislation, we may be affected by the final outcome. If the preliminary findings of the EC investigation were ultimately to be upheld, we calculate our maximum potential liability as at 31 December 2018 to be approximately $147m. We do not consider at present that any provision is required in respect of this amount based on our current assessment of the issue.
In December 2016, the Group appealed to the First Tier Tribunal in the UK against a decision by HM Revenue and Customs (HMRC) relating to the tax deductibility of historical foreign exchange losses. The decision of the Tribunal was released on 8 February 2017 and it upheld the Group’s appeal. HMRC appealed against this decision and their appeal was heard by the Upper Tribunal in June 2018. The decision was released on 29 November 2018 and it upheld the decision of the First Tier Tribunal. In the event that HMRC either is not granted leave to appeal or is not successful in their appeal, with the result that the Group is ultimately successful in the Courts, the Group’s tax charge would be reduced in the year of success. Whether HMRC will be granted leave to appeal in the Court of Appeal is uncertain at the present date. No tax benefit for these losses has been taken to date. Should the case become final in 2019, we estimate that we would receive a tax refund of approximately $100m. In addition, remaining losses would be potentially available to offset future profits, the benefit of which will depend on future facts and circumstances.
In 2016, the UK Government enacted legislation to reduce the main rate of UK statutory corporation tax to 19.0% from 1 April 2017 and 17.0% from 1 April 2020.
The UK standard rate of corporation tax for 2018 is 19.0% (2017: 19.3%, 2016: 20.0%). Overseas taxation is calculated at the rates prevailing in the respective jurisdictions. The table below reconciles the expected tax charge at the UK statutory rate with the actual tax charge:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Profit before taxation |
|
781 |
|
879 |
|
1,062 |
|
|
Expected taxation at UK statutory rate of 19.0% (2017: 19.3%, 2016: 20.0%) |
|
148 |
|
169 |
|
212 |
|
|
Differences in overseas taxation rates1 |
|
(5) |
|
48 |
|
34 |
|
|
Disposal of the Gynaecology business (mainly at the US tax rate) |
|
– |
|
– |
|
56 |
|
|
Benefit of US Manufacturing deduction |
|
– |
|
(9) |
|
(7) |
|
|
R&D tax credits |
|
(6) |
|
(3) |
|
(3) |
|
|
Tax losses not recognised |
|
4 |
|
10 |
|
1 |
|
|
Utilisation of previously unrecognised tax losses |
|
(1) |
|
(6) |
|
(9) |
|
|
Impact of US tax reform |
|
– |
|
(32) |
|
– |
|
|
Expenses not deductible for tax purposes |
|
15 |
|
11 |
|
23 |
|
|
Change in tax rates |
|
1 |
|
(5) |
|
1 |
|
|
Adjustments in respect of prior years2 |
|
(38) |
|
(71) |
|
(30) |
|
|
Total taxation as per the income statement |
|
118 |
|
112 |
|
278 |
|
|
1 |
Reflects the geographical mix of profits offset by the impact of intra-group loans provided to finance US acquisitions and business operations. |
|
2 |
The adjustments in respect of prior years are explained on the previous page. |
|
142 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
5.2 Deferred taxation
Movements in the main components of deferred tax assets and liabilities were as follows:
|
|
|
|
|
|
|
|
|
|
|
Inventory, |
|
|
|
|
|
|
Accelerated |
|
|
|
Retirement |
|
|
|
provisions, |
|
|
|
|
|
|
tax |
|
|
|
benefit |
|
Macrotexture |
|
losses and other |
|
|
|
|
|
|
depreciation |
|
Intangibles |
|
obligations |
|
claims |
|
differences |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 31 December 2016 |
|
(73) |
|
(209) |
|
28 |
|
52 |
|
205 |
|
3 |
|
|
Exchange adjustment |
|
1 |
|
(2) |
|
2 |
|
– |
|
13 |
|
14 |
|
|
Movement in income statement – current year |
|
(9) |
|
15 |
|
(6) |
|
(1) |
|
(31) |
|
(32) |
|
|
Movement in income statement – prior years |
|
2 |
|
4 |
|
– |
|
– |
|
5 |
|
11 |
|
|
Movement in other comprehensive income |
|
– |
|
– |
|
(17) |
|
– |
|
4 |
|
(13) |
|
|
Movement in equity |
|
– |
|
– |
|
– |
|
– |
|
(3) |
|
(3) |
|
|
Changes in tax rate |
|
29 |
|
71 |
|
– |
|
(18) |
|
(33) |
|
49 |
|
|
Acquisitions |
|
– |
|
(22) |
|
– |
|
– |
|
23 |
|
1 |
|
|
At 31 December 2017 |
|
(50) |
|
(143) |
|
7 |
|
33 |
|
183 |
|
30 |
|
|
Adjustment on initial application of IFRS 9 |
|
– |
|
– |
|
– |
|
– |
|
3 |
|
3 |
|
|
Adjusted balance at 1 January 2018 |
|
(50) |
|
(143) |
|
7 |
|
33 |
|
186 |
|
33 |
|
|
Exchange adjustment |
|
– |
|
2 |
|
– |
|
– |
|
(7) |
|
(5) |
|
|
Movement in income statement – current year |
|
11 |
|
14 |
|
– |
|
(33) |
|
11 |
|
3 |
|
|
Movement in income statement – prior years |
|
(12) |
|
1 |
|
4 |
|
– |
|
12 |
|
5 |
|
|
Movement in other comprehensive income |
|
– |
|
– |
|
(6) |
|
– |
|
(3) |
|
(9) |
|
|
Movement in equity |
|
– |
|
– |
|
– |
|
– |
|
1 |
|
1 |
|
|
Changes in tax rate |
|
(1) |
|
– |
|
– |
|
– |
|
– |
|
(1) |
|
|
At 31 December 2018 |
|
(52) |
|
(126) |
|
5 |
|
– |
|
200 |
|
27 |
|
Represented by:
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Deferred tax assets |
|
126 |
|
127 |
|
|
Deferred tax liabilities |
|
(99) |
|
(97) |
|
|
Net position at 31 December |
|
27 |
|
30 |
|
The Group has gross unused trading and non-trading tax losses of $149m (2017: $271m) and gross unused capital losses of $102m (2017: $113m) available for offset against future profits of which $14m of trading losses will expire within 3-8 years from the balance sheet date if not utilised. A deferred tax asset of $28m (2017: $38m) has been recognised in respect of $74m (2017: $132m) of the trading and non-trading tax losses. No deferred tax asset has been recognised on the remaining unused tax losses as they are not expected to be realised in the foreseeable future. There are no temporary differences in respect of investments in subsidiaries and associates for which deferred tax liabilities have not been recognised (2017: $nil).
|
|
Smith & Nephew Annual Report 2018 |
|
143 |
|
Accounting policies Earnings per share Basic earnings per share is calculated by dividing the profit attributable to equity holders by the weighted average number of ordinary shares in issue during the year, excluding shares held by the Company in the Employees’ Share Trust or as treasury shares. Diluted earnings per share Diluted earnings per share is calculated by adjusting the basic earnings per share for the effect of conversion to ordinary shares associated with dilutive potential ordinary shares, which comprise share options and awards granted to employees. Adjusted earnings per share Adjusted earnings per share is a trend measure, which presents the long-term profitability of the Group excluding the impact of specific transactions that management considers affects the Group’s short-term profitability. The Group presents this measure to assist investors in their understanding of trends. Adjusted attributable profit is the numerator used for this measure. The Group has identified the following items as those to be excluded when arriving at adjusted attributable profit: acquisitions and disposals related items including amortisation and impairment of acquisition intangible assets; significant restructuring programmes; significant gains and losses arising from legal disputes and other significant items (including US tax reform) and taxation thereon. |
The calculations of the basic, diluted and adjusted earnings per ordinary share are based on the following attributable profit and numbers of shares:
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Earnings |
|
|
|
|
|
|
|
|
|
|
Attributable profit for the year |
|
|
|
663 |
|
767 |
|
784 |
|
|
Adjusted attributable profit (see below) |
|
|
|
881 |
|
826 |
|
735 |
|
Attributable profit is reconciled to adjusted attributable profit as follows:
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
$ million |
|
|
Attributable profit for the year |
|
|
|
663 |
|
767 |
|
784 |
|
|
Acquisition and disposal related items |
|
|
|
(7) |
|
(10) |
|
9 |
|
|
Restructuring and rationalisation costs |
|
3 |
|
120 |
|
– |
|
62 |
|
|
Amortisation and impairment of acquisition intangibles1 |
|
9 |
|
118 |
|
140 |
|
178 |
|
|
Legal and other2 |
|
|
|
38 |
|
(13) |
|
(20) |
|
|
Profit on disposal of business |
|
21 |
|
– |
|
– |
|
(326) |
|
|
US tax reform |
|
5 |
|
– |
|
(32) |
|
– |
|
|
Taxation on excluded items |
|
5 |
|
(51) |
|
(26) |
|
48 |
|
|
Adjusted attributable profit |
|
|
|
881 |
|
826 |
|
735 |
|
|
1 |
Amortisation and impairment of acquisition intangibles includes a $113m charge within operating profit and a $5m charge within share of result of associates. |
|
2 |
Legal and other charge in 2018 includes $34m (2017: $16m credit, 2016: $30m credit) within operating profit (refer to Note 2.5) and a $4m charge (2017: $3m charge, 2016: $5m charge) within other finance costs for unwinding of the discount on the provision for known, anticipated and settled metal-on-metal hip claims globally. In 2016, legal and other credit also includes a $5m charge within share of results of associates for expenses incurred by Bioventus for an aborted initial public offering of shares. |
|
144 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
The numerators used for basic and diluted earnings per ordinary share are the same. The denominators used for all categories of earnings per ordinary share are as follows:
|
|
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
Number of shares (millions) |
|
|
|
|
|
|
|
|
|
|
Basic weighted number of shares |
|
|
|
873 |
|
874 |
|
890 |
|
|
Dilutive impact of share incentive schemes outstanding |
|
|
|
3 |
|
1 |
|
3 |
|
|
Diluted weighted average number of shares |
|
|
|
876 |
|
875 |
|
893 |
|
|
Earnings per ordinary share |
|
|
|
|
|
|
|
|
|
|
Basic |
|
|
|
76.0¢ |
|
87.8¢ |
|
88.1¢ |
|
|
Diluted |
|
|
|
75.7¢ |
|
87.7¢ |
|
87.8¢ |
|
|
Adjusted3 |
|
|
|
100.9¢ |
|
94.5¢ |
|
82.6¢ |
|
|
3 |
Adjusted earnings per share is calculated using the basic weighted number of shares. |
7 PROPERTY, PLANT AND EQUIPMENT
|
Accounting policies Property, plant and equipment Items of property, plant and equipment are stated at cost less accumulated depreciation and any accumulated impairment losses. Depreciation is calculated to write off the cost of items of property, plant and equipment less their estimated residual values using the straight‑line method over their estimated useful lives, and is ultimately recognised in profit or loss. Leased assets are depreciated over the shorter of the lease term and their useful lives unless it is reasonably certain that the Group will obtain ownership by the end of the lease term. Freehold land is not depreciated. The estimated useful lives of items of property, plant and equipment is s 3–20 years and for buildings is 20–50 years. Assets in course of construction are not depreciated until they are available for use. Depreciation methods, useful lives and residual values are reviewed at each reporting date and adjusted if appropriate. Finance costs relating to the purchase or construction of property, plant and equipment and intangible assets that take longer than one year to complete are capitalised based on the Group weighted average borrowing costs. All other finance costs are expensed as incurred. Impairment of assets The carrying values of property, plant and equipment are reviewed for impairment when events or changes in circumstances indicate the carrying value may be impaired. If any such indication exists, the recoverable amount of the asset is estimated in order to determine the extent of impairment loss. Where it is not possible to estimate the recoverable amount of an individual asset, the Group estimates the recoverable amount of the cash-generating unit to which it belongs. An asset’s recoverable amount is the higher of an asset’s or cash-generating unit’s fair value less costs to sell and its value-in-use. In assessing value‑in-use, its estimated future cash flow is discounted to its present value using a pre-tax discount rate that reflects the current market assessment of the time value of money and the risks specific to the asset. |
|
|
Smith & Nephew Annual Report 2018 |
|
145 |
|
|
|
|
|
Land and buildings |
|
Plant and equipment |
|
Assets in |
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
course of |
|
|
|
|
|
|
|
|
Freehold |
|
Leasehold |
|
Instruments |
|
Other |
|
construction |
|
Total |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Cost |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At 1 January 2017 |
|
|
|
165 |
|
119 |
|
1,116 |
|
1,023 |
|
115 |
|
2,538 |
|
|
Exchange adjustment |
|
|
|
6 |
|
1 |
|
63 |
|
33 |
|
3 |
|
106 |
|
|
Acquisitions |
|
21 |
|
– |
|
– |
|
– |
|
1 |
|
– |
|
1 |
|
|
Additions |
|
|
|
1 |
|
– |
|
176 |
|
28 |
|
103 |
|
308 |
|
|
Disposals |
|
|
|
– |
|
(27) |
|
(73) |
|
(79) |
|
(12) |
|
(191) |
|
|
Transfers |
|
|
|
56 |
|
(20) |
|
2 |
|
56 |
|
(115) |
|
(21) |
|
|
At 31 December 2017 |
|
|
|
228 |
|
73 |
|
1,284 |
|
1,062 |
|
94 |
|
2,741 |
|
|
Exchange adjustment |
|
|
|
(5) |
|
(1) |
|
(45) |
|
(20) |
|
(1) |
|
(72) |
|
|
Additions |
|
|
|
3 |
|
3 |
|
179 |
|
21 |
|
126 |
|
332 |
|
|
Disposals |
|
|
|
– |
|
– |
|
(73) |
|
(24) |
|
(1) |
|
(98) |
|
|
Impairment |
|
|
|
– |
|
– |
|
– |
|
– |
|
(1) |
|
(1) |
|
|
Transfers |
|
|
|
33 |
|
1 |
|
43 |
|
(7) |
|
(89) |
|
(19) |
|
|
At 31 December 2018 |
|
|
|
259 |
|
76 |
|
1,388 |
|
1,032 |
|
128 |
|
2,883 |
|
|
Depreciation and impairment |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At 1 January 2017 |
|
|
|
50 |
|
42 |
|
781 |
|
672 |
|
11 |
|
1,556 |
|
|
Exchange adjustment |
|
|
|
3 |
|
– |
|
45 |
|
24 |
|
– |
|
72 |
|
|
Charge for the year |
|
|
|
6 |
|
7 |
|
146 |
|
84 |
|
– |
|
243 |
|
|
Disposals |
|
|
|
– |
|
(22) |
|
(67) |
|
(74) |
|
(11) |
|
(174) |
|
|
Transfers |
|
|
|
2 |
|
3 |
|
(1) |
|
(9) |
|
– |
|
(5) |
|
|
At 31 December 2017 |
|
|
|
61 |
|
30 |
|
904 |
|
697 |
|
– |
|
1,692 |
|
|
Exchange adjustment |
|
|
|
(2) |
|
– |
|
(30) |
|
(14) |
|
– |
|
(46) |
|
|
Charge for the year |
|
|
|
8 |
|
6 |
|
163 |
|
74 |
|
– |
|
251 |
|
|
Impairment |
|
|
|
– |
|
1 |
|
– |
|
3 |
|
– |
|
4 |
|
|
Disposals |
|
|
|
– |
|
– |
|
(59) |
|
(21) |
|
– |
|
(80) |
|
|
Transfers |
|
|
|
5 |
|
– |
|
23 |
|
(28) |
|
– |
|
– |
|
|
At 31 December 2018 |
|
|
|
72 |
|
37 |
|
1,001 |
|
711 |
|
– |
|
1,821 |
|
|
Net book amounts |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At 31 December 2018 |
|
|
|
187 |
|
39 |
|
387 |
|
321 |
|
128 |
|
1,062 |
|
|
At 31 December 2017 |
|
|
|
167 |
|
43 |
|
380 |
|
365 |
|
94 |
|
1,049 |
|
Land and buildings includes land with a cost of $21m (2017: $21m) that is not subject to depreciation. There were no assets held under finance leases at 31 December 2018 and 2017.
Transfers from assets in course of construction includes $19m (2017: $4m) of software and $nil (2017: $12m) net book value of other non-current assets.
Group capital expenditure relating to property, plant and equipment contracted but not provided for amounted to $16m (2017: $26m).
The amount of borrowing costs capitalised in 2018 and 2017 was minimal.
|
146 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
|
Accounting policy Goodwill is not amortised but is reviewed for impairment annually. Goodwill is allocated to the cash-generating unit (CGU) that is expected to benefit from the acquisition. The goodwill is tested annually for impairment by comparing the recoverable amount to the carrying value of the CGUs. The CGUs identified by management are at the aggregated product franchise levels of Orthopaedics, Other Surgical Devices and Advanced Wound Management, in the way the core assets are used to generate cash flows. If the recoverable amount of the CGU is less than its carrying amount then an impairment loss is determined to have occurred. Any impairment losses that arise are recognised immediately in the income statement and are allocated first to reduce the carrying amount of goodwill and then to the carrying amounts of the other assets of the CGU. In carrying out impairment reviews of goodwill a number of significant assumptions have to be made when preparing cash flow projections. These include the future rate of market growth, discount rates, the market demand for the products acquired, the future profitability of acquired businesses or products, levels of reimbursement and success in obtaining regulatory approvals. If actual results should differ, or changes in expectations arise, impairment charges may be required which would adversely impact operating results. |
|
|
|
|
|
2018 |
|
2017 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
|
Cost |
|
|
|
|
|
|
|
|
At 1 January |
|
|
|
2,371 |
|
2,188 |
|
|
Exchange adjustment |
|
|
|
(34) |
|
51 |
|
|
Acquisitions |
|
21 |
|
– |
|
132 |
|
|
At 31 December |
|
|
|
2,337 |
|
2,371 |
|
|
Impairment |
|
|
|
|
|
|
|
|
At 1 January and 31 December |
|
|
|
– |
|
– |
|
|
Net book amounts |
|
|
|
2,337 |
|
2,371 |
|
Management has identified four CGUs in applying the provisions of IAS 36 Impairment of Assets: Orthopaedics, Other Surgical Devices, Advanced Wound Care & Devices and Bioactives.
For the purpose of goodwill impairment testing, the Advanced Wound Care & Devices and Bioactives CGUs have been aggregated (Advanced Wound Management), as this is the level at which goodwill is monitored and level at which the economic benefits relating to the goodwill within these CGUs is realised.
Goodwill is allocated to the Group’s CGUs as follows:
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Orthopaedics |
|
727 |
|
566 |
|
|
Other Surgical Devices |
|
1,313 |
|
1,501 |
|
|
Advanced Wound Management |
|
297 |
|
304 |
|
|
|
|
2,337 |
|
2,371 |
|
Impairment reviews were performed as of September 2018 and September 2017 by comparing the recoverable amount of each CGU with its carrying amount, including goodwill. These were updated during December, taking into account any significant events that occurred between September and December.
For each CGU, the recoverable amounts are based on value-in-use which is calculated from pre-tax cash flow projections for three years using data from the Group’s budget and strategic planning process, the results of which are reviewed and approved by the Board. These projections exclude any estimated future cash inflows or outflows expected to arise from future restructurings. The three-year period is in line with the Group’s strategic planning process. In determining the growth rates used in the calculations of the value-in-use, management considered annual revenue growth. Projections are based on anticipated volume and value growth in the markets served by the Group and assumptions as to market share movements. Each year the projections for the previous year are compared to actual results and variances are factored into the assumptions used in the current year.
The discount rates used in the value-in-use calculations reflect management’s assessment of risks specific to the assets of each CGU.
|
|
Smith & Nephew Annual Report 2018 |
|
147 |
8.1 Orthopaedics CGU
The sales growth and trading profit margin used in the value-in-use calculation for the Orthopaedics CGU, which includes the Reconstruction and Trauma businesses, reflects management’s distinctive orthopaedic reconstruction strategy, which combines cutting edge innovation, disruptive business models and a strong Emerging Markets platform to drive our performance.
Revenue growth rates for the three-year period ranged from 3.6% to 4.5% for the various components of the Orthopaedics CGU. The average growth rate used to extrapolate the cash flows beyond the three-year period in calculating the terminal value is 2.0%. The pre-tax discount rate used in the Orthopaedics CGU value-in-use calculation reflects the geographical mix and is 9.6%.
8.2 Other Surgical Devices CGU
The value-in-use calculation for the Other Surgical Devices CGU reflects growth rates and trading profit margins consistent with management’s strategy to rebalance Smith & Nephew towards higher growth areas such as, for example, Sports Medicine.
Revenue growth rates for the three-year period ranged from 2.6% to 4.7% for the various components of the Other Surgical Devices CGU. The weighted average growth rate used to extrapolate the cash flows beyond the three-year period in calculating the terminal value is 2.0%. The pre-tax discount rate used in the Other Surgical Devices CGU value-in-use calculation reflects the geographical mix of the revenues and is 9.6%.
8.3 Advanced Wound Management CGU
The aggregated Advanced Wound Management CGU comprises the Advanced Wound Care & Devices and Bioactives CGUs.
In performing the value-in-use calculation for this combined CGU, management considered the Group’s focus across the wound product franchises, focusing on widening access to the customer, the higher added value sectors of healing chronic wounds and tissue repair using bioactives, and by continuing to improve efficiency.
Revenue growth rates for the three-year period ranged from 2.8% to 4.2% for the various components of the Advanced Wound Management CGU. The weighted average growth rate used to extrapolate the cash flows beyond the three-year period in calculating the terminal value is 2.0%. The pre-tax discount rate used in the Advanced Wound Management CGU value-in-use calculation reflects the geographical mix and industry sector and is 9.6%.
8.4 Sensitivity to changes in assumptions used in value-in-use calculations
The calculations of value-in-use for the identified CGUs are most sensitive to changes in discount and growth rates. Management’s consideration of these sensitivities is set out below:
Growth of market and market share – management has considered the impact of a variance in market growth and market share. The value‑in‑use calculations shows that if the assumed long-term growth rates were reduced to nil, the recoverable amount of each CGU would still be greater than its carrying value.
Discount rate – management has considered the impact of an increase in the discount rate applied to the value-in-use calculations. This sensitivity analysis shows that for the recoverable amount of each CGU to be less than its carrying value, the discount rate would have to be increased to 21.5% for the Orthopaedics CGU, 13.5% for the Other Surgical Devices CGU and 20.8% for the Advanced Wound Management CGU.
|
148 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
|
Accounting policies Intangible assets Intangible assets acquired separately from a business combination (including purchased patents, know-how, trademarks, licences and distribution rights) are initially measured at cost. The cost of intangible assets acquired in a material business combination (referred to as acquisition intangibles) is the fair value as at the date of acquisition. Following initial recognition, intangible assets are carried at cost less any accumulated amortisation and any accumulated impairment losses. All intangible assets are amortised on a straight-line basis over their estimated useful economic lives. The estimated useful economic life of an intangible asset ranges between 3–20 years depending on its nature. Internally-generated intangible assets are expensed in the income statement as incurred. Purchased computer software and certain costs of information technology projects are capitalised as intangible assets. Software that is integral to computer hardware is capitalised as plant and equipment. Impairment of intangible assets The carrying values of intangible assets are reviewed for impairment when events or changes in circumstances indicate the carrying value may be impaired. If any such indication exists, the recoverable amount of the asset is estimated in order to determine the extent of impairment loss. Where it is not possible to estimate the recoverable amount of an individual asset, the Group estimates the recoverable amount of the CGU to which it belongs. An asset’s recoverable amount is the higher of an asset’s or CGU’s fair value less costs to sell and its value-in-use. In assessing value-in-use, its estimated future cash flow is discounted to its present value using a pre-tax discount rate that reflects the current market assessments of the time value of money and the risks specific to the asset. In carrying out impairment reviews of intangible assets a number of significant assumptions have to be made when preparing cash flow projections. These include the future rate of market growth, discount rates, the market demand for the products acquired, the future profitability of acquired businesses or products, levels of reimbursement and success in obtaining regulatory approvals. If actual results should differ, or changes in expectations should arise, impairment charges may be required which would adversely impact operating results. |
|
|
Smith & Nephew Annual Report 2018 |
|
149 |
|
|
|
|
|
|
|
Customer and |
|
|
|
Assets |
|
|
|
|
|
|
|
|
Product- |
|
distribution |
|
|
|
in course of |
|
|
|
|
|
|
Technology |
|
related |
|
related |
|
Software |
|
construction |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Cost |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At 1 January 2017 |
|
301 |
|
1,849 |
|
121 |
|
329 |
|
– |
|
2,600 |
|
|
Exchange adjustment |
|
10 |
|
38 |
|
1 |
|
12 |
|
– |
|
61 |
|
|
Acquisitions1 |
|
59 |
|
2 |
|
– |
|
– |
|
– |
|
61 |
|
|
Additions |
|
– |
|
2 |
|
3 |
|
63 |
|
– |
|
68 |
|
|
Disposals |
|
(6) |
|
(43) |
|
(5) |
|
(5) |
|
– |
|
(59) |
|
|
Transfers |
|
(6) |
|
6 |
|
– |
|
4 |
|
– |
|
4 |
|
|
At 31 December 2017 |
|
358 |
|
1,854 |
|
120 |
|
403 |
|
– |
|
2,735 |
|
|
Exchange adjustment |
|
(4) |
|
(18) |
|
(8) |
|
(6) |
|
– |
|
(36) |
|
|
Additions |
|
– |
|
1 |
|
– |
|
8 |
|
6 |
|
15 |
|
|
Disposals |
|
– |
|
(1) |
|
– |
|
(4) |
|
– |
|
(5) |
|
|
Transfers |
|
– |
|
– |
|
– |
|
12 |
|
7 |
|
19 |
|
|
At 31 December 2018 |
|
354 |
|
1,836 |
|
112 |
|
413 |
|
13 |
|
2,728 |
|
|
Amortisation and impairment |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At 1 January 2017 |
|
36 |
|
886 |
|
80 |
|
187 |
|
– |
|
1,189 |
|
|
Exchange adjustment |
|
2 |
|
21 |
|
1 |
|
6 |
|
– |
|
30 |
|
|
Charge for the year: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amortisation |
|
6 |
|
133 |
|
15 |
|
38 |
|
– |
|
192 |
|
|
Impairment |
|
– |
|
10 |
|
– |
|
– |
|
– |
|
10 |
|
|
Disposals |
|
11 |
|
(61) |
|
(3) |
|
(4) |
|
– |
|
(57) |
|
|
Transfers |
|
(4) |
|
4 |
|
– |
|
– |
|
– |
|
– |
|
|
At 31 December 2017 |
|
51 |
|
993 |
|
93 |
|
227 |
|
– |
|
1,364 |
|
|
Exchange adjustment |
|
(1) |
|
(14) |
|
(4) |
|
(3) |
|
– |
|
(22) |
|
|
Charge for the year: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amortisation |
|
24 |
|
103 |
|
6 |
|
43 |
|
– |
|
176 |
|
|
Impairment |
|
– |
|
– |
|
– |
|
3 |
|
– |
|
3 |
|
|
Disposals |
|
– |
|
(1) |
|
– |
|
(2) |
|
– |
|
(3) |
|
|
At 31 December 2018 |
|
74 |
|
1,081 |
|
95 |
|
268 |
|
– |
|
1,518 |
|
|
Net book amounts |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At 31 December 2018 |
|
280 |
|
755 |
|
17 |
|
145 |
|
13 |
|
1,210 |
|
|
At 31 December 2017 |
|
307 |
|
861 |
|
27 |
|
176 |
|
– |
|
1,371 |
|
|
1 |
In 2017 this relates to technology and product-related intangibles acquired with the purchase of Rotation Medical, Inc. |
Transfers into software and assets in course of construction includes $19m (2017: $4m) of software transferred from property, plant and equipment.
|
150 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
Amortisation and impairment of acquisition intangibles is set out below:
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Technology |
|
24 |
|
6 |
|
|
Product-related |
|
86 |
|
124 |
|
|
Customer and distribution related |
|
3 |
|
10 |
|
|
Total |
|
113 |
|
140 |
|
Group capital expenditure relating to software contracted but not provided for amounted to $nil (2017: $nil). There was no impairment charge in 2018. In 2017, a product-related intangible asset was determined to have a value in use below its carrying value, resulting in an impairment charge of $10m being recognised.
|
Accounting policy Investments, other than those related to associates, are initially recorded at fair value plus any directly attributable transaction costs on the trade date. The Group has investments in unquoted entities and an entity that holds mainly unquoted equity securities, which by their nature have no fixed maturity date or coupon rate. These investments are classed as fair value through profit or loss. The fair value of these investments is based on the underlying fair value of the equity securities: marketable securities are valued by reference to closing prices in the market; and non-marketable securities are estimated considering factors including the purchase price; prices of recent significant private placements of securities of the same issuer and estimates of liquidation value. Changes in fair value based on externally observable valuation events are recognised in profit or loss. The investments accounting policy for the year ending 31 December 2017 was consistent with the requirements of IAS 39. Changes in fair value were recorded through other comprehensive income, rather than through profit or loss. |
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
At 1 January |
|
21 |
|
25 |
|
|
Additions |
|
4 |
|
8 |
|
|
Fair value remeasurement1 |
|
9 |
|
(10) |
|
|
Impairment |
|
– |
|
(2) |
|
|
At 31 December |
|
34 |
|
21 |
|
|
1 |
The Group adopted IFRS 9 on 1 January 2018 and elected to present changes in the fair value of trade investments in the income statement from that date. The fair value remeasurement of trade investments in 2017 was recognised in other comprehensive income. |
|
Accounting policy Investments in associates, being those entities over which the Group has a significant influence and which is neither a subsidiary nor a joint venture, are accounted for using the equity method, with the Group recording its share of the associates’ profit and loss and other comprehensive income. The Group’s share of associates’ profit or loss is included in one separate income statement line and is calculated after deduction of their respective taxes. |
At 31 December 2018 and 31 December 2017, the Group holds 49% of Bioventus LLC (Bioventus). Bioventus is a limited liability company operating as a partnership. The company’s headquarters is located in Durham, North Carolina, US. Bioventus focuses its medical product development around its core competencies of orthobiologic therapies and orthopaedic diagnostics from which it develops and markets clinically proven orthopaedic therapies and diagnostic tools, including osteoarthritis pain treatments, bone growth stimulators and ultrasound devices. Bioventus sells bone healing stimulation devices and is a provider of osteoarthritis injection therapies. The Group’s ability to recover the value of its investment is dependent upon the ongoing clinical and commercial success of these products. The loss after taxation recognised in the income statement relating to Bioventus was $11m (2017: $6m profit).
The carrying amount of this investment was reviewed for impairment as at the balance sheet date. For the purposes of impairment testing the recoverable amount of this investment was based on its fair value less cost to sell, estimated using discounted cash flows.
|
|
Smith & Nephew Annual Report 2018 |
|
151 |
The amounts recognised in the balance sheet and income statement for associates are as follows:
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Balance sheet |
|
105 |
|
118 |
|
|
Income statement (loss)/profit |
|
(11) |
|
6 |
|
Summarised financial information for significant associates
Set out below is the summarised financial information for Bioventus, adjusted for differences with Group accounting policies:
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Summarised statement of comprehensive income |
|
|
|
|
|
|
Revenue |
|
320 |
|
301 |
|
|
Attributable (loss)/profit for the year |
|
(19) |
|
1 |
|
|
Group adjustments1 |
|
(3) |
|
11 |
|
|
Total comprehensive (loss)/profit |
|
(22) |
|
12 |
|
|
Group share of (loss)/profit for the year at 49% |
|
(11) |
|
6 |
|
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Summarised balance sheet |
|
|
|
|
|
|
Non-current assets |
|
296 |
|
332 |
|
|
Current assets |
|
149 |
|
122 |
|
|
Non-current liabilities |
|
(234) |
|
(246) |
|
|
Current liabilities |
|
(56) |
|
(47) |
|
|
Net assets |
|
155 |
|
161 |
|
|
Group’s share of net assets at 49% |
|
76 |
|
79 |
|
|
Group adjustments1 |
|
26 |
|
35 |
|
|
Group’s carrying amount of investment at 49% |
|
102 |
|
114 |
|
|
1 |
Group adjustments include an adjustment to align the useful life of intangible assets with Group policy. |
During the year the Group received a $2m (2017: $nil) cash distribution from Bioventus.
At December 2018, the Group held an equity investment in one other associate (2017: one) with a carrying value of $3m (2017: $3m).
|
Accounting policy Finished goods and work-in-progress are valued at factory cost, including appropriate overheads, on a first-in first-out basis. Raw materials and bought-in finished goods are valued at purchase price. All inventories are reduced to net realisable value where lower than cost. Inventory acquired as part of a business acquisition is valued at selling price less costs to sell and a profit allowance for selling efforts. Orthopaedic instruments are generally not sold but provided to customers and distributors for use in surgery. They are recorded as inventory until they are deployed at which point they are transferred to plant and equipment and depreciated over their useful economic lives of between three and seven years. A feature of the orthopaedic business is the high level of product inventory required, some of which is located at customer premises and is available for customers’ immediate use (referred to as consignment inventory). Complete sets of product, including large and small sizes, have to be made available in this way. These outer sizes are used less frequently than standard sizes and towards the end of the product life cycle are inevitably in excess of requirements. Adjustments to carrying value are therefore required to be made to orthopaedic inventory to anticipate this situation. These adjustments are calculated in accordance with a formula based on levels of inventory compared with historical or forecast usage. This formula is applied on an individual product line basis and is first applied when a product group has been on the market for two years. This method of calculation is considered appropriate based on experience but it involves management judgements on effectiveness of inventory deployment, length of product lives, phase-out of old products and efficiency of manufacturing planning systems. |
|
152 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Raw materials and consumables |
|
219 |
|
207 |
|
213 |
|
|
Work-in-progress |
|
88 |
|
69 |
|
55 |
|
|
Finished goods and goods for resale |
|
1,088 |
|
1,028 |
|
976 |
|
|
|
|
1,395 |
|
1,304 |
|
1,244 |
|
Reserves for excess and obsolete inventories were $305m (2017: $296m, 2016: $303m). The increase in reserves of $9m in the year comprised a $19m increase in the reserve relating to the write-off of inventory which was partially offset by foreign exchange movements of $10m.
The determination of the estimate of excess and obsolete inventory is a critical accounting estimate and includes assumptions on the future usage of all different items of finished goods. This estimate is not considered to have a range of potential outcomes that is significantly different to the $305m held at 31 December 2018.
The cost of inventories recognised as an expense and included in cost of goods sold amounted to $1,126m (2017: $1,013m, 2016: $1,131m). In addition, $94m was recognised as an expense within cost of goods sold resulting from inventory write-offs (2017: $68m, 2016: $85m).
Notwithstanding inventory acquired within acquisitions, no inventory is carried at fair value less costs to sell in any year.
13 TRADE AND OTHER RECEIVABLES
|
Accounting policy Trade and other receivables are carried at amortised cost, less any allowances for uncollectible amounts. They are included in current assets, except for maturities greater than 12 months after the balance sheet date. These are classified as non-current assets. The trade and other receivables accounting policy for the year ending 31 December 2017 was consistent with the requirements of IAS 39. Provisions against trade receivables were based on incurred losses, rather than the expected credit loss allowance. The Group manages credit risk through credit limits which require authorisation commensurate with the size of the limit and which are regularly reviewed. Credit limit decisions are made based on available financial information and the business case. Significant receivables are regularly reviewed and monitored at Group level. The Group has no significant concentration of credit risk, with exposure spread over a large number of customers and geographies. Furthermore, the Group’s principal customers are backed by government and public or private medical insurance funding, which historically represent a lower risk of default. The maximum exposure to credit risk at the reporting date is the fair value of each class of receivable. The Group does not hold any collateral as security. Allowance losses are calculated by reviewing lifetime expected credit losses using historic and forward-looking data on credit risk. The Group performed the calculation of expected credit loss rates separately for customer groups which were segmented based on common risk characteristics such as credit risk grade and type of customer (such as government and non-government). |
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Trade and other receivables due within one year |
|
|
|
|
|
|
|
|
Trade receivables |
|
1,166 |
|
1,125 |
|
1,042 |
|
|
Less: loss allowance |
|
(62) |
|
(69) |
|
(54) |
|
|
Trade receivables – net |
|
1,104 |
|
1,056 |
|
988 |
|
|
Derivatives – forward foreign exchange, currency swaps and interest rate contracts |
|
37 |
|
28 |
|
48 |
|
|
Other receivables |
|
107 |
|
92 |
|
76 |
|
|
Prepayments |
|
69 |
|
82 |
|
73 |
|
|
|
|
1,317 |
|
1,258 |
|
1,185 |
|
|
Due after more than one year |
|
|
|
|
|
|
|
|
Other non-current assets |
|
16 |
|
16 |
|
– |
|
|
|
|
1,333 |
|
1,274 |
|
1,185 |
|
Trade receivables are classified as loans and receivables. Management considers that the carrying amount of trade and other receivables approximates to the fair value. Allowance losses are calculated by reviewing lifetime expected credit losses using historic and forward-looking data on credit risk. The loss allowance expense for the year was $14m (2017: $17m expense, 2016: $7m expense). Other non-current assets primarily relate to long-term prepayments.
|
|
Smith & Nephew Annual Report 2018 |
|
153 |
The following table provides information about the ageing of and expected credit losses for trade receivables:
|
|
2018 Weighted average loss rate |
|
2018 Loss allowance |
|
2018 Gross carrying amount |
|
2017 Gross carrying amount |
|
2016 Gross carrying amount |
|
|
|
% |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Not past due |
0.3% |
|
(2) |
|
647 |
|
664 |
|
725 |
|
|
Past due not more than 3 months |
0.4% |
|
(1) |
|
271 |
|
225 |
|
142 |
|
|
Past due not more than 3–6 months |
2.6% |
|
(2) |
|
78 |
|
65 |
|
51 |
|
|
Past due more than 6 months |
33.5% |
|
(57) |
|
170 |
|
171 |
|
124 |
|
|
|
|
|
(62) |
|
1,166 |
|
1,125 |
|
1,042 |
|
|
Loss allowance |
|
|
|
|
(62) |
|
(69) |
|
(54) |
|
|
Trade receivables – net |
|
|
|
|
1,104 |
|
1,056 |
|
988 |
|
The Group’s expected credit loss accounting policy includes guidance on how the expected credit loss percentages should be determined; it does not include preset limits as the customer groups and risk profiles are not consistent across all of our markets. Each market determines their own percentages based on their historic experience and future expectations, and in line with the general guidance in the Group’s policy.
Movements in the loss allowance were as follows:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
At 31 December |
|
69 |
|
54 |
|
64 |
|
|
Adjustment on initial application of IFRS 9 |
|
14 |
|
|
|
|
|
|
Adjusted balance at 1 January |
|
83 |
|
|
|
|
|
|
Exchange adjustment |
|
(3) |
|
3 |
|
(3) |
|
|
Reclassification1 |
|
(8) |
|
– |
|
– |
|
|
Acquisitions |
|
– |
|
1 |
|
– |
|
|
Net receivables provided during the year |
|
14 |
|
17 |
|
7 |
|
|
Utilisation of provision |
|
(24) |
|
(6) |
|
(14) |
|
|
At 31 December |
|
62 |
|
69 |
|
54 |
|
1 On transition to IFRS 9, the Group reclassified a credit note provision from the loss allowance to gross trade receivables.
Trade receivables include amounts denominated in the following major currencies:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
US Dollar |
|
527 |
|
418 |
|
416 |
|
|
Sterling |
|
45 |
|
54 |
|
57 |
|
|
Euro |
|
201 |
|
212 |
|
193 |
|
|
Other |
|
331 |
|
372 |
|
322 |
|
|
Trade receivables – net |
|
1,104 |
|
1,056 |
|
988 |
|
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Trade and other payables due within one year |
|
|
|
|
|
|
Trade and other payables |
|
854 |
|
873 |
|
|
Derivatives – forward foreign exchange, currency swaps and interest rate contracts |
|
25 |
|
48 |
|
|
Acquisition consideration |
|
78 |
|
36 |
|
|
|
|
957 |
|
957 |
|
|
Other payables due after one year |
|
|
|
|
|
|
Acquisition consideration |
|
49 |
|
124 |
|
|
Other payables |
|
4 |
|
4 |
|
|
|
|
53 |
|
128 |
|
|
154 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
The acquisition consideration includes $99m (2017: $104m) contingent upon future events.
The acquisition consideration due after more than one year is expected to be payable as follows: $21m in 2020, $23m in 2021, $1m in 2022, $1m in 2023 and $3m due in over five years (2017: $50m in 2019, $24m in 2020, $43m in 2021, $2m in 2022, and $5m due in over five years).
15.1 Net debt
Net debt comprises borrowings and credit balances on currency swaps less cash at bank.
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Bank overdrafts, borrowings and loans due within one year |
|
164 |
|
27 |
|
|
Long-term bank borrowings |
|
304 |
|
300 |
|
|
Private placement notes |
|
997 |
|
1,123 |
|
|
Borrowings |
|
1,465 |
|
1,450 |
|
|
Cash at bank |
|
(365) |
|
(169) |
|
|
Credit/(debit) balance on derivatives – currency swaps |
|
1 |
|
(2) |
|
|
Credit balance on derivatives – interest rate swaps |
|
3 |
|
2 |
|
|
Net debt |
|
1,104 |
|
1,281 |
|
Borrowings are repayable as follows:
|
|
|
Within |
|
Between |
|
Between |
|
Between |
|
Between |
|
|
|
|
|
|
|
|
one year or |
|
one and |
|
two and |
|
three and |
|
four and |
|
After |
|
|
|
|
|
|
on demand |
|
two years |
|
three years |
|
four years |
|
five years |
|
five years |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 31 December 2018 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Bank loans |
|
7 |
|
– |
|
– |
|
304 |
|
– |
|
– |
|
311 |
|
|
Bank overdrafts |
|
32 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
32 |
|
|
Private placement notes |
|
125 |
|
– |
|
262 |
|
125 |
|
105 |
|
505 |
|
1,122 |
|
|
|
|
164 |
|
– |
|
262 |
|
429 |
|
105 |
|
505 |
|
1,465 |
|
|
At 31 December 2017 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Bank loans |
|
13 |
|
– |
|
300 |
|
– |
|
– |
|
– |
|
313 |
|
|
Bank overdrafts |
|
14 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
14 |
|
|
Private placement notes |
|
– |
|
124 |
|
– |
|
264 |
|
125 |
|
610 |
|
1,123 |
|
|
|
|
27 |
|
124 |
|
300 |
|
264 |
|
125 |
|
610 |
|
1,450 |
|
15.2 Liquidity risk exposures
The Board has established a set of policies to manage funding and currency risks. The Group uses derivative financial instruments only to manage the financial risks associated with underlying business activities and their financing.
Liquidity risk is the risk that the Group is not able to settle or meet its obligations on time or at a reasonable price. The Group’s policy is to ensure that there is sufficient funding and facilities in place to meet foreseeable borrowing requirements. The Group manages and monitors liquidity risk through regular reporting of current cash and borrowing balances and periodic preparation and review of short and medium-term cash forecasts, having regard to the maturities of investments and borrowing facilities.
The Group has available committed facilities of $2.4bn (2017: $2.4bn). The interest payable on borrowings under committed facilities is either at fixed or floating rates. Floating rates are typically based on the LIBOR (or other reference rate) relevant to the term and currency concerned.
|
|
Smith & Nephew Annual Report 2018 |
|
155 |
The Company is subject to restrictive covenants under its principal facility agreements. These financial covenants are tested at the end of each half year for the 12 months ending on the last day of the testing period. As of 31 December 2018, the Company was in compliance with these covenants. The facilities are also subject to customary events of default, none of which are currently anticipated to occur.
The Group’s committed facilities are:
|
Facility |
|
Date due |
|
|
$80 million 2.47% Senior Notes |
|
November 2019 |
|
|
$45 million Floating Rate Senior Notes |
|
November 2019 |
|
|
$75 million 3.23% Senior Notes |
|
January 2021 |
|
|
$190 million 2.97% Senior Notes |
|
November 2021 |
|
|
$75 million 3.46% Senior Notes |
|
January 2022 |
|
|
€265 million bilateral, term loan facility |
|
April 2022 |
|
|
$50 million 3.15% Senior Notes |
|
November 2022 |
|
|
$1.0 billion syndicated, revolving credit facility |
|
June 2023 |
|
|
$105 million 3.26% Senior Notes |
|
November 2023 |
|
|
$100 million 3.89% Senior Notes |
|
January 2024 |
|
|
$305 million 3.36% Senior Notes |
|
November 2024 |
|
|
$25 million Floating Rate Senior Notes |
|
November 2024 |
|
|
$75 million 3.99% Senior Notes |
|
January 2026 |
|
15.3 Year end financial liabilities by contractual maturity
The table below analyses the Group’s year end financial liabilities by contractual maturity date, including contractual interest payments and excluding the impact of netting arrangements:
|
|
|
Within one |
|
Between |
|
Between |
|
|
|
|
|
|
|
|
year or on |
|
one and |
|
two and |
|
After |
|
|
|
|
|
|
demand |
|
two years |
|
five years |
|
five years |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 31 December 2018 |
|
|
|
|
|
|
|
|
|
|
|
|
Non-derivative financial liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
Bank overdrafts and loans |
|
39 |
|
– |
|
304 |
|
– |
|
343 |
|
|
Trade and other payables |
|
854 |
|
1 |
|
1 |
|
2 |
|
858 |
|
|
Private placement notes |
|
164 |
|
35 |
|
571 |
|
522 |
|
1,292 |
|
|
Acquisition consideration |
|
78 |
|
21 |
|
25 |
|
3 |
|
127 |
|
|
Derivative financial liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
Currency swaps/forward foreign exchange contracts – outflow |
|
2,394 |
|
– |
|
– |
|
– |
|
2,394 |
|
|
Currency swaps/forward foreign exchange contracts – inflow |
|
(2,393) |
|
– |
|
– |
|
– |
|
(2,393) |
|
|
|
|
1,136 |
|
57 |
|
901 |
|
527 |
|
2,621 |
|
|
At 31 December 2017 |
|
|
|
|
|
|
|
|
|
|
|
|
Non-derivative financial liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
Bank overdrafts and loans |
|
27 |
|
– |
|
300 |
|
– |
|
327 |
|
|
Trade and other payables |
|
873 |
|
1 |
|
1 |
|
2 |
|
877 |
|
|
Private placement notes |
|
36 |
|
161 |
|
476 |
|
647 |
|
1,320 |
|
|
Acquisition consideration |
|
36 |
|
50 |
|
69 |
|
5 |
|
160 |
|
|
Derivative financial liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
Currency swaps/forward foreign exchange contracts – outflow |
|
2,737 |
|
– |
|
– |
|
– |
|
2,737 |
|
|
Currency swaps/forward foreign exchange contracts – inflow |
|
(2,739) |
|
– |
|
– |
|
– |
|
(2,739) |
|
|
|
|
970 |
|
212 |
|
846 |
|
654 |
|
2,682 |
|
The amounts in the tables above are undiscounted cash flows, which differ from the amounts included in the balance sheet where the underlying cash flows have been discounted.
|
156 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
15.4 Liquidity and capital resources
The Group’s policy is to ensure that it has sufficient funding and facilities to meet foreseeable borrowing requirements.
At 31 December 2018, the Group held $333m (2017: $155m, 2016: $38m) in cash net of bank overdrafts. The Group had committed facilities available of $2,429m at 31 December 2018 of which $1,429m was drawn. Smith & Nephew intends to repay the amounts due within one year using available cash and drawing down on the longer-term facilities.
The principal variations in the Group’s borrowing requirements result from the timing of dividend payments, acquisitions and disposals of businesses, timing of capital expenditure and working capital fluctuations. Smith & Nephew believes that its capital expenditure needs and its working capital funding for 2019, as well as its other known or expected commitments or liabilities, can be met from its existing resources and facilities. The Group’s net debt decreased from $1,281m at the beginning of 2018 to $1,104m at the end of 2018, representing an overall decrease of $177m.
The Group’s planned future contributions are considered adequate to cover the current underfunded position in the Group’s defined benefit plans.
16 FINANCIAL INSTRUMENTS AND RISK MANAGEMENT
|
Accounting policy Derivative financial instruments Derivative financial instruments are initially recognised at fair value on the date a derivative contract is entered into and are subsequently remeasured at their fair value at subsequent balance sheet dates. Changes in the fair value of derivative financial instruments that are designated and effective as cash flow hedges of forecast third party transactions are recognised in other comprehensive income until the associated asset or liability is recognised. Amounts taken to other comprehensive income are transferred to the income statement in the period in which the hedged transaction affects profit and loss. Where the hedged item is the cost of a non-financial asset, the amounts taken to other comprehensive income are transferred to the initial carrying value of the asset. Changes in the fair values of hedging instruments that are designated and effective as net investment hedges are matched in other comprehensive income against changes in value of the related net assets. Interest rate derivatives transacted to fix interest rates on floating rate borrowings are accounted for as cash flow hedges and changes in the fair values resulting from changes in market interest rates are recognised in other comprehensive income. Amounts taken to other comprehensive income are transferred to the income statement when the hedged transaction affects profit and loss. Interest rate derivatives transacted to convert fixed rate borrowings into floating rate borrowings are accounted for as fair value hedges and changes in the fair values resulting from changes in market interest rates are recognised in the income statement. Any ineffectiveness on hedging instruments and changes in the fair value of derivative financial instruments that do not qualify for hedge accounting are recognised in the income statement within other finance costs as they arise. Hedge accounting is discontinued when the hedging instrument expires or is sold, terminated or exercised, or no longer qualifies for hedge accounting. At that point in time, any cumulative gain or loss on the hedging instrument recognised in other comprehensive income is retained there until the forecast transaction occurs. If a hedged transaction is no longer expected to occur, the net cumulative gain or loss recognised in other comprehensive income is transferred. |
16.1 Foreign exchange risk management
The Group operates in many countries and as a consequence has transactional and translational foreign exchange exposure. It is Group policy for operating units not to hold material unhedged monetary assets or liabilities other than in their functional currencies.
Foreign exchange variations affect trading results in two ways. Firstly, on translation of overseas sales and profits into US Dollars and secondly, transactional exposures arising where some, or all of the costs of sale are incurred in a different currency from the sale. The principal transactional exposures arise as the proportion of costs in US Dollars, Sterling and Swiss Francs exceed the proportion of sales in each of these currencies and correspondingly the proportion of sales in Euros exceeds the proportion of costs in Euros.
|
|
Smith & Nephew Annual Report 2018 |
|
157 |
The impact of currency movements on the cost of purchases is partly mitigated by the use of forward foreign exchange contracts. The Group uses forward foreign exchange contracts, designated as cash flow hedges, to hedge forecast third party trading cash flows up to one year. When a commitment is entered into, forward foreign exchange contracts are normally used to increase the hedge to 100% of the exposure. Cash flows relating to cash flow hedges are expected to occur within 12 months of inception and profits and losses on hedges are expected to enter into the determination of profit (within cost of goods sold) within a further 12‑month period. The principal currencies hedged by forward foreign exchange contracts are US Dollars, Euros, Sterling and Singapore Dollars. At 31 December 2018, the Group had contracted to exchange within one year the equivalent of $2.1bn (2017: $2.3bn). Based on the Group’s net borrowings as at 31 December 2018, if the US Dollar were to weaken against all currencies by 10%, the Group’s net borrowings would increase by $25m (2017: decrease by $3m) principally due to the €265m term loan.
If the US Dollar were to weaken by 10% against all other currencies, then the fair value of the forward foreign exchange contracts as at 31 December 2018 would have been $38m lower (2017: $53m lower). Similarly, if the Euro were to weaken by 10% against all other currencies, then the fair value of the forward foreign exchange contracts as at 31 December 2018 would have been $15m higher (2017: $12m higher). Movements in the fair value of forward foreign exchange contracts would be recognised in other comprehensive income and accumulated in the hedging reserve.
A 10% strengthening of the US Dollar or Euro against all other currencies at 31 December 2018 would have had the equal but opposite effect to the amounts shown above, on the basis that all other variables remain constant.
The Group’s policy is to hedge all actual foreign exchange exposures and the Group’s forward foreign exchange contracts are designated as cash flow hedges. The net impact of transaction related foreign exchange on the income statement from a movement in exchange rates on the value of forward foreign exchange contracts is not significant. In addition, the movements in the fair value of other financial instruments used for hedging such as currency swaps for which hedge accounting is not applied, offset movements in the values of assets and liabilities and are recognised through the income statement. Hedge ineffectiveness is caused by actual cash flows in foreign currencies varying from forecast cash flows.
16.2 Interest rate risk management
The Group is exposed to interest rate risk on cash, borrowings and certain currency and interest rate swaps which are at floating rates. When required the Group uses interest rate derivatives to meet its objective of protecting borrowing costs within parameters set by the Board. These interest rate derivatives are accounted for as cash flow hedges and, as such, changes in fair value resulting from changes in market interest rates are recognised in other comprehensive income and accumulated in the hedging reserve, with the fair value of the interest rate derivatives recorded in the balance sheet.
Additionally, the Group uses interest rate swaps to reduce the overall level of fixed rate debt, within parameters set by the Board. When used in this way, interest rate derivatives are accounted for as fair value hedges. The fair value movement of the derivative is offset in the income statement against the fair value movement in the underlying fixed rate debt.
Based on the Group’s gross borrowings and cash as at 31 December 2018, if interest rates were to increase by 100 basis points in all currencies then the annual net interest charge would increase by $3m (2017: $6m). A decrease in interest rates by 100 basis points in all currencies would have an equal but opposite effect to the amounts shown above.
16.3 Credit risk management
The Group limits exposure to credit risk on counterparties used for financial instruments through a system of internal credit limits. The financial exposure of a counterparty is determined as the total of cash and deposits, plus the risk on derivative instruments, assessed as the fair value of the instrument plus a risk element based on the nominal value and the historic volatility of the market value of the instrument. The Group does not anticipate non-performance of counterparties and believes it is not subject to material concentration of credit risk as the Group operates within a policy of counterparty limits designed to reduce exposure to any single counterparty.
The maximum credit risk exposure on derivatives at 31 December 2018 was $37m (2017: $28m), being the total debit fair values on forward foreign exchange contracts and currency swaps. The maximum credit risk exposure on cash at bank at 31 December 2018 was $365m (2017: $169m). The Group’s exposure to credit risk is not material as the amounts are held in a wide number of banks in a number of different countries.
Credit risk on trade receivables is detailed in Note 13.
|
158 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
The amounts relating to items designated as hedging instruments were as follows:
|
|
|
|
|
Carrying |
|
Carrying |
|
Changes in |
|
Hedge |
|
Amounts reclassified |
|
|
|
|
|
Nominal |
|
Amount |
|
Amount |
|
fair value |
|
ineffectiveness |
|
from hedging reserve |
|
|
|
|
|
amount |
|
Assets |
|
Liabilities |
|
in OCI |
|
in profit or loss |
|
to profit or loss |
|
Line item in |
|
|
|
$ million |
|
$ million |
|
$million |
|
$ million |
|
$ million |
|
$ million |
|
profit or loss |
|
At 31 December 2018 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Foreign currency risk |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Forward exchange contracts1 |
|
2,394 |
|
37 |
|
(22) |
|
23 |
|
– |
|
2 |
|
Cash flow hedges |
|
Interest rate risk |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest rate swaps2 |
|
(200) |
|
– |
|
(3) |
|
– |
|
– |
|
– |
|
N/A |
|
At 31 December 2017 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Foreign currency risk |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Forward exchange contracts1 |
|
2,737 |
|
25 |
|
(45) |
|
(24) |
|
– |
|
21 |
|
Cash flow hedges |
|
Interest rate risk |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest rate swaps2 |
|
(200) |
|
– |
|
(2) |
|
– |
|
– |
|
– |
|
N/A |
|
1 |
Presented in Trade and other receivables and Trade and other payables on the Balance Sheet. |
|
2 |
Presented in Trade and other payables on the Balance Sheet. |
16.4 Net investment hedge
Part of the Group’s net investment in its Euro subsidiaries is hedged by a new €265m Euro-denominated bank loan which mitigates the foreign currency risk arising from the subsidiaries’ net assets. The loan is designated as a hedging instrument for the changes in the value of the net investment that is attributable to changes in the EUR/USD spot rate, and is 100% hedged.
To assess hedge effectiveness, the Group determines the economic relationship between the hedging instrument and the hedged item by comparing changes in the carrying amount of the debt that is attributable to a change in the spot rate with changes in the investment in the foreign operation due to movements in the spot rate (the offset method). The Group’s policy is to hedge the net investment only to the extent of the debt principal. Hedge ineffectiveness occurs if the value of the Euro-denominated bank loan exceeds the value of the Euro subsidiaries.
16.5 Currency and interest rate profile of interest bearing liabilities and assets
Short-term debtors and creditors are excluded from the following disclosures.
Currency and interest rate profile of interest bearing liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fixed rate liabilities |
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
average |
|
|
|
|
|
|
|
|
Interest |
|
|
|
|
|
|
|
Weighted |
|
time |
|
|
|
|
Gross |
|
Currency |
|
rate |
|
Total |
|
Floating |
|
Fixed rate |
|
average |
|
for which |
|
|
|
|
borrowings |
|
swaps |
|
swaps |
|
liabilities |
|
rate liabilities |
|
liabilities |
|
interest rate |
|
rate is fixed |
|
|
|
|
$ million |
|
$ million |
|
$million |
|
$ million |
|
$ million |
|
$ million |
|
% |
|
Years |
|
|
At 31 December 2018 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
US Dollar |
|
(1,142) |
|
(193) |
|
(3) |
|
(1,338) |
|
(483) |
|
(855) |
|
3.4 |
|
4.8 |
|
|
Other |
|
(323) |
|
(61) |
|
– |
|
(384) |
|
(384) |
|
– |
|
– |
|
– |
|
|
Total interest bearing liabilities |
|
(1,465) |
|
(254) |
|
(3) |
|
(1,722) |
|
(867) |
|
(855) |
|
|
|
|
|
|
At 31 December 2017 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
US Dollar |
|
(1,428) |
|
(291) |
|
(2) |
|
(1,721) |
|
(866) |
|
(855) |
|
3.4 |
|
5.8 |
|
|
Other |
|
(22) |
|
(95) |
|
– |
|
(117) |
|
(117) |
|
– |
|
– |
|
– |
|
|
Total interest bearing liabilities |
|
(1,450) |
|
(386) |
|
(2) |
|
(1,838) |
|
(983) |
|
(855) |
|
|
|
|
|
In 2018, the Group also had liabilities due for deferred and contingent acquisition consideration (denominated in US Dollars, Euros and Russian Rubles) totalling $127m (2017: $160m, 2016: $120m) on which no interest was payable (see Note 14). There were no other significant interest bearing or non-interest bearing financial liabilities. Floating rates on liabilities are typically based on the one, three or six-month LIBOR (or other reference rate) relevant to the currency concerned. The weighted average interest rate on floating rate borrowings as at 31 December 2018 was 4% (2017: 3%).
|
|
Smith & Nephew Annual Report 2018 |
|
159 |
Currency and interest rate profile of interest bearing assets:
|
|
|
Interest |
|
Cash |
|
Currency |
|
|
|
Floating |
|
Fixed |
|
|
|
|
rate swaps |
|
at bank |
|
swaps |
|
Total assets |
|
rate assets |
|
rate assets |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 31 December 2018 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
US Dollars |
|
– |
|
289 |
|
60 |
|
349 |
|
349 |
|
– |
|
|
Other |
|
– |
|
76 |
|
193 |
|
269 |
|
269 |
|
– |
|
|
Total interest bearing assets |
|
– |
|
365 |
|
253 |
|
618 |
|
618 |
|
– |
|
|
At 31 December 2017 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
US Dollars |
|
– |
|
110 |
|
94 |
|
204 |
|
204 |
|
– |
|
|
Other |
|
– |
|
59 |
|
294 |
|
353 |
|
353 |
|
– |
|
|
Total interest bearing assets |
|
– |
|
169 |
|
388 |
|
557 |
|
557 |
|
– |
|
Floating rates on assets are typically based on the short-term deposit rates relevant to the currency concerned.
16.6 Fair value of financial assets and liabilities
|
Accounting policy Measurement of fair values A number of the Group’s accounting policies and disclosures require the measurement of fair values, for both financial assets and liabilities and non-financial assets acquired in a business combination (see Note 21). When measuring the fair value of an asset or liability, the Group uses market observable data as far as possible. Fair values are categorised into different levels in the fair value hierarchy based on the inputs used in the valuation techniques as follows: Level 1: quoted prices (unadjusted) in active markets for identical assets or liabilities; Level 2: inputs other than quoted prices included in Level 1 that are observable for the asset or liability, either directly (ie as prices) or indirectly (ie derived from prices); and Level 3: inputs for the asset or liability that are not based on observable data (unobservable inputs). The Group recognises transfers between the levels of the fair value hierarchy at the end of the reporting period during which the change has occurred. |
There has been no change in the classification of financial assets and liabilities, the method and assumptions used in determining fair value and the categorisation of financial assets and liabilities within the fair value hierarchy from those disclosed in the Annual Report for the year ended 31 December 2017 other than as disclosed in Note 1.
The Group enters into derivative financial instruments with financial institutions with investment grade credit ratings. The fair value of forward foreign exchange contracts is calculated by reference to quoted market forward exchange rates for contracts with similar maturity profiles. The fair value of currency swaps is determined by reference to quoted market spot rates. As a result, foreign forward exchange contracts and currency swaps are classified as Level 2 within the fair value hierarchy. The changes in counterparty credit risk had no material effect on the hedge effectiveness for derivatives designated in hedge relationships and other financial instruments recognised at fair value. The fair value of investments is based upon third party pricing models for share issues. As a result, investments are considered Level 3 in the fair value hierarchy. There were no transfers between Levels 1, 2 and 3 during 2018 and 2017. For cash and cash equivalents, short-term loans and receivables, overdrafts and other short-term liabilities which have a maturity of less than three months, the book values approximate the fair values because of their short-term nature.
Long-term borrowings are measured in the balance sheet at amortised cost. As the Group’s long-term borrowings are not quoted publicly and as market prices are not available, their fair values are estimated by discounting future contractual cash flows to net present values at the current market interest rates available to the Group for similar financial instruments as at the year end. The fair value of the private placement notes is determined using a discounted cash flow model based on prevailing market rates.
|
160 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
The following table shows the carrying amounts and fair values of financial assets and financial liabilities, including their levels in the fair value hierarchy. It does not include fair value information for financial assets and financial liabilities not measured at fair value if the carrying amount is a reasonable approximation of fair value.
|
|
|
|
|
|
|
|
|
|
|
|
|
Carrying |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
amount |
|
|
|
|
|
Fair value |
|
|
|
|
Fair value – |
|
|
|
|
|
Fair value |
|
Other |
|
|
|
|
|
|
|
|
|
|
|
|
hedging |
|
Amortised |
|
Fair value |
|
through profit |
|
financial |
|
|
|
|
|
|
|
|
|
|
|
|
instruments |
|
cost |
|
through OCI |
|
or loss |
|
liabilities |
|
Total |
|
Level 2 |
|
Level 3 |
|
Total |
|
|
At 31 December 2018 |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Financial assets measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Forward foreign exchange contracts |
|
36 |
|
– |
|
– |
|
– |
|
– |
|
36 |
|
36 |
|
– |
|
36 |
|
|
Investments |
|
– |
|
– |
|
– |
|
34 |
|
– |
|
34 |
|
– |
|
34 |
|
34 |
|
|
Currency swaps |
|
– |
|
– |
|
1 |
|
– |
|
– |
|
1 |
|
1 |
|
– |
|
1 |
|
|
|
|
36 |
|
– |
|
1 |
|
34 |
|
– |
|
71 |
|
|
|
|
|
|
|
|
Financial liabilities measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition consideration |
|
– |
|
– |
|
– |
|
(99) |
|
– |
|
(99) |
|
– |
|
(99) |
|
(99) |
|
|
Forward foreign exchange contracts |
|
(20) |
|
– |
|
– |
|
– |
|
– |
|
(20) |
|
(20) |
|
– |
|
(20) |
|
|
Currency swaps |
|
– |
|
– |
|
(2) |
|
– |
|
– |
|
(2) |
|
(2) |
|
– |
|
(2) |
|
|
Interest rate swaps |
|
(3) |
|
– |
|
– |
|
– |
|
– |
|
(3) |
|
(3) |
|
– |
|
(3) |
|
|
|
|
(23) |
|
– |
|
(2) |
|
(99) |
|
– |
|
(124) |
|
|
|
|
|
|
|
|
Financial assets not measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade and other receivables |
|
– |
|
1,211 |
|
– |
|
– |
|
– |
|
1,211 |
|
|
|
|
|
|
|
|
Cash at bank |
|
– |
|
365 |
|
– |
|
– |
|
– |
|
365 |
|
|
|
|
|
|
|
|
|
|
– |
|
1,576 |
|
– |
|
– |
|
– |
|
1,576 |
|
|
|
|
|
|
|
|
Financial liabilities not measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition consideration |
|
– |
|
– |
|
– |
|
(28) |
|
– |
|
(28) |
|
|
|
|
|
|
|
|
Bank overdrafts |
|
– |
|
– |
|
– |
|
– |
|
(32) |
|
(32) |
|
|
|
|
|
|
|
|
Bank loans |
|
– |
|
– |
|
– |
|
– |
|
(311) |
|
(311) |
|
|
|
|
|
|
|
|
Private placement debt in a hedge relationship |
|
– |
|
– |
|
– |
|
– |
|
(197) |
|
(197) |
|
|
|
|
|
|
|
|
Private placement debt not in a hedge relationship |
|
– |
|
– |
|
– |
|
– |
|
(925) |
|
(925) |
|
|
|
|
|
|
|
|
Trade and other payables |
|
– |
|
– |
|
– |
|
– |
|
(858) |
|
(858) |
|
|
|
|
|
|
|
|
|
|
– |
|
– |
|
– |
|
(28) |
|
(2,323) |
|
(2,351) |
|
|
|
|
|
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
161 |
During the year ended 31 December 2018, acquisition consideration decreased by $33m due to $29m of payments for acquisitions made in prior years and $4m of remeasurements. The fair value of contingent consideration is estimated using a discounted cash flow model. The valuation model considers the present value of expected payment, discounted using a risk-adjusted discount rate. The expected payment is determined by considering the possible scenarios, which relate to the achievement of established milestones and targets, the amount to be paid under each scenario and the probability of each scenario. As a result, contingent consideration is classified as Level 3 within the fair value hierarchy.
|
|
|
|
|
|
|
|
|
|
|
|
|
Carrying |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
amount |
|
|
|
|
|
Fair value |
|
|
|
|
Fair value – |
|
|
|
|
|
Fair value |
|
Other |
|
|
|
|
|
|
|
|
|
|
|
|
hedging |
|
Amortised |
|
Fair value |
|
through profit |
|
financial |
|
|
|
|
|
|
|
|
|
|
|
|
instruments |
|
cost |
|
through OCI |
|
or loss |
|
liabilities |
|
Total |
|
Level 2 |
|
Level 3 |
|
Total |
|
|
At 31 December 2017 |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Financial assets measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Forward foreign exchange contracts |
|
25 |
|
– |
|
– |
|
– |
|
– |
|
25 |
|
25 |
|
– |
|
25 |
|
|
Investments |
|
– |
|
– |
|
– |
|
21 |
|
– |
|
21 |
|
– |
|
21 |
|
21 |
|
|
Currency swaps |
|
– |
|
– |
|
3 |
|
– |
|
– |
|
3 |
|
3 |
|
– |
|
3 |
|
|
|
|
25 |
|
– |
|
3 |
|
21 |
|
– |
|
49 |
|
|
|
|
|
|
|
|
Financial liabilities measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition consideration |
|
– |
|
– |
|
– |
|
(104) |
|
– |
|
(104) |
|
– |
|
(104) |
|
(104) |
|
|
Forward foreign exchange contracts |
|
(45) |
|
– |
|
– |
|
– |
|
– |
|
(45) |
|
(45) |
|
– |
|
(45) |
|
|
Currency swaps |
|
– |
|
– |
|
(1) |
|
– |
|
– |
|
(1) |
|
(1) |
|
– |
|
(1) |
|
|
Interest rate swaps |
|
(2) |
|
– |
|
– |
|
– |
|
– |
|
(2) |
|
(2) |
|
– |
|
(2) |
|
|
|
|
(47) |
|
– |
|
(1) |
|
(104) |
|
– |
|
(152) |
|
|
|
|
|
|
|
|
Financial assets not measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade and other receivables |
|
– |
|
1,148 |
|
– |
|
– |
|
– |
|
1,148 |
|
|
|
|
|
|
|
|
Cash at bank |
|
– |
|
169 |
|
– |
|
– |
|
– |
|
169 |
|
|
|
|
|
|
|
|
|
|
– |
|
1,317 |
|
– |
|
– |
|
– |
|
1,317 |
|
|
|
|
|
|
|
|
Financial liabilities not measured at fair value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition consideration |
|
– |
|
– |
|
– |
|
(56) |
|
– |
|
(56) |
|
|
|
|
|
|
|
|
Bank overdrafts |
|
– |
|
– |
|
– |
|
– |
|
(14) |
|
(14) |
|
|
|
|
|
|
|
|
Bank loans |
|
– |
|
– |
|
– |
|
– |
|
(313) |
|
(313) |
|
|
|
|
|
|
|
|
Private placement debt in a hedge relationship |
|
– |
|
– |
|
– |
|
– |
|
(198) |
|
(198) |
|
|
|
|
|
|
|
|
Private placement debt not in a hedge relationship |
|
– |
|
– |
|
– |
|
– |
|
(925) |
|
(925) |
|
|
|
|
|
|
|
|
Trade and other payables |
|
– |
|
– |
|
– |
|
– |
|
(877) |
|
(877) |
|
|
|
|
|
|
|
|
|
|
– |
|
– |
|
– |
|
(56) |
|
(2,327) |
|
(2,383) |
|
|
|
|
|
|
|
|
162 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
17 PROVISIONS AND CONTINGENCIES
|
Accounting policy In the normal course of business the Group is involved in various legal disputes. Provisions are made for loss contingencies when it is deemed probable that an adverse outcome will occur and the amount of the losses can be reasonably estimated. Where the Group is the plaintiff in pursuing claims against third parties, legal and associated expenses are charged to the income statement as incurred. The recognition of provisions for legal disputes is subject to a significant degree of estimation. In making its estimates management takes into account the advice of internal and external legal counsel. Provisions are reviewed regularly and amounts updated where necessary to reflect developments in the disputes. The ultimate liability may differ from the amount provided depending on the outcome of court proceedings or settlement negotiations or as new facts emerge. A provision for onerous contracts is recognised when the expected benefits to be derived by the Group from a contract are lower than the unavoidable cost of meeting its obligations under the contract. For the purpose of calculating any onerous lease provision, the Group takes the discounted future lease payments (if any), net of expected rental income. Before a provision is established, the Group recognises any impairment loss on the assets associated with that contract. A provision for rationalisation is recognised when the Group has approved a detailed and formal restructuring plan, and the restructuring either has commenced or has been announced publicly. Future operating losses are not provided for. |
17.1 Provisions
|
|
|
Rationalisation |
|
|
|
Legal and other |
|
|
|
|
|
|
provisions |
|
Metal-on-metal |
|
provisions |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 1 January 2017 |
|
20 |
|
163 |
|
98 |
|
281 |
|
|
Net charge to income statement |
|
– |
|
10 |
|
2 |
|
12 |
|
|
Unwinding of discount |
|
– |
|
3 |
|
– |
|
3 |
|
|
Utilised |
|
(15) |
|
(19) |
|
(28) |
|
(62) |
|
|
Transfers |
|
– |
|
– |
|
(9) |
|
(9) |
|
|
Exchange adjustment |
|
1 |
|
– |
|
– |
|
1 |
|
|
At 31 December 2017 |
|
6 |
|
157 |
|
63 |
|
226 |
|
|
Net charge to income statement |
|
120 |
|
72 |
|
(2) |
|
190 |
|
|
Unwinding of discount |
|
– |
|
4 |
|
– |
|
4 |
|
|
Utilised |
|
(90) |
|
(41) |
|
(14) |
|
(145) |
|
|
Exchange adjustment |
|
(1) |
|
– |
|
– |
|
(1) |
|
|
At 31 December 2018 |
|
35 |
|
192 |
|
47 |
|
274 |
|
|
Provisions – due within one year |
|
35 |
|
50 |
|
36 |
|
121 |
|
|
Provisions – due after one year |
|
– |
|
142 |
|
11 |
|
153 |
|
|
At 31 December 2018 |
|
35 |
|
192 |
|
47 |
|
274 |
|
|
Provisions – due within one year |
|
6 |
|
73 |
|
50 |
|
129 |
|
|
Provisions – due after one year |
|
– |
|
84 |
|
13 |
|
97 |
|
|
At 31 December 2017 |
|
6 |
|
157 |
|
63 |
|
226 |
|
The principal elements within rationalisation provisions relate to the implementation of the Accelerating Performance and Execution (APEX) programme that was announced in February 2018.
Following the settlement of a group of the US metal-on-metal hip claims (discussed below) the Group has estimated a provision of $192m (2017: $157m) relating to the present value at 31 December 2018 of the estimated costs to resolve all other known and anticipated metal-on-metal hip claims globally. The estimated value of the provision has been determined using an actuarial model. Given the inherent uncertainty in assumptions relating to factors such as the number of claims and outcomes, the actual costs may differ significantly from this estimate. A range of expected outcomes between the 5th and 95th percentile generated by the actuarial model would not give rise to a significantly different outcome in 2019. Based on the actuarial model the likelihood of a charge similar to that incurred in 2018 being incurred in 2019 is remote. The provision does not include any possible insurance recoveries on these claims or legal fees associated with defending claims. The Group carries considerable product liability insurance, and will continue to defend claims vigorously.
The legal and other provisions mainly relate to various other product liability and intellectual property litigation matters.
All provisions are expected to be substantially utilised within five years of 31 December 2018 and none are treated as financial instruments.
|
|
Smith & Nephew Annual Report 2018 |
|
163 |
17.2 Contingencies
The Company and its subsidiaries are party to various legal proceedings, some of which include claims for substantial damages. The outcome of these proceedings cannot readily be foreseen, but except as described herein management believes none of them is likely to result in a material adverse effect on the financial position of the Group. The Group provides for outcomes that are deemed to be probable and can be reliably estimated. There is no assurance that losses will not exceed provisions or will not have a significant impact on the Group’s results of operations in the period in which they are realised.
In August 2003, the Group withdrew voluntarily from all markets the macrotextured versions of its OXINIUM femoral knee components. A charge of $154m was recorded in 2004 for anticipated expenses in connection with macrotexture claims. Most of that amount has since been applied to settlements of such claims, and all claims have now been resolved. The aggregate cost at 31 December 2018 related to this matter is approximately $205m. The Group has sought recovery from its primary and excess insurers for costs of resolving the claims. The primary insurance carrier has paid $60m in full settlement of its policy liability. However, the excess carriers denied coverage, citing defences relating to the wording of the insurance policies and other matters. In December 2004, the Group brought suit against them in the US district court for the Western District of Tennessee. An additional $22m was received during 2007 from a successful settlement with a third party. In 2018, the Group agreed to settle the suit against the insurers for a total of approximately $84m which has been recognised in the Group’s 2018 operating profit.
17.3 Legal proceedings
Product liability claims
The Group faces claims from time to time for alleged defects in its products and has on occasion recalled or withdrawn products from the market. Such claims are endemic to the medical device industry. The Group maintains product liability insurance subject to limits and deductibles that management believes are reasonable. All policies contain exclusions and limitations, however, and there can be no assurance that insurance will be available or adequate to cover all claims.
In recent years, there has been heightened concern about possible adverse effects of hip implant products with metal-on-metal bearing surfaces, and the Group has incurred, and will continue to incur expenses to defend claims in this area. As of February 2019, and giving effect to the US settlements described below, approximately 1,023 such claims were pending with the Group around the world. Most claims relate to the Group’s Birmingham Hip Resurfacing (BHR) product and its two modular metal-on-metal components: the Birmingham Hip Modular Head (BHMH) and the optional metal liner component of the R3 Acetabular System (R3ML). The BHMH and R3ML are no longer on the market: the R3ML was withdrawn in 2012 and the BHMH was phased out in 2014. In 2015, the Group ceased offering smaller sizes of the BHR and restricted instructions for BHR use in female patients. These actions were taken to ensure that the BHR is only used in those patient groups where it continues to demonstrate strong performance.
In 2015 and 2016, the Group’s US subsidiary settled a large part of the majority of its US metal-on-metal hip lawsuits in two group settlements, without admitting liability. Insurance receipts covered most of the amounts paid, with the net cash cost being $25m. In November 2017, the Group’s US subsidiary entered into a memorandum of understanding to settle a third group of claims, without admitting liability. The third settlement was finalised in 2018. These cases principally related to the Group’s modular metal-on-metal hip components, which are no longer on the market. On 5 April 2017, the Judicial Panel on Multidistrict Litigation (MDL) ordered Smith & Nephew BHR cases pending or later filed in US federal court to be consolidated for pre-trial proceedings and transferred to the federal court in Baltimore, Maryland. As of February 2019, there were approximately 571 cases pending in the MDL in the United States. In England and Wales, the Group’s UK subsidiary entered into a group settlement in 2017 to settle 150 claims principally related to the Group’s modular metal-on-metal hip component, which are no longer on the market. Metal-on-metal hip implant claims against various companies in England and Wales were consolidated for trials under group litigation orders in the High Court in London. As of February 2019, the majority of the BHR claims pending against the Group in England and Wales have been discontinued.
The Group has requested indemnity from its product liability insurers for most of these metal-on-metal hip implant claims. Each insurer makes its own decision as to coverage issues, and the liability of some insurers depends on exhaustion of lower levels of coverage. Insurers of the lower layers of the Group’s insurances have indemnified the Group in respect of these claims up to the limits of those insurances. The Group has commenced arbitration proceedings against another insurer in respect of that insurer’s share of the claims and associated defence costs in the amount of $50m.
Litigation outcomes are difficult to predict and defence costs can be significant. The Group takes care to monitor the clinical evidence relating to its metal hip implant products and ensure that its product offerings are designed to serve patients’ interests.
Intellectual property disputes
The Group is engaged, as both plaintiff and defendant, in litigation with various competitors and others over claims of patent infringement and other intellectual property matters. These disputes are being heard in courts in the US and other jurisdictions and also before agencies that examine patents. Outcomes are rarely certain and costs are often significant.
|
164 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
The Group prosecuted and defended a series of patent infringement suits against Arthrex in US federal courts in Oregon and Texas starting in 2004, principally relating to suture anchors for use in shoulder surgery. Arthrex paid $99m in June 2015 in connection with the Oregon litigation, and most of that award (net of various expenses) was recognised in the Group’s operating profit at that time. The Group asserted the same patent against additional Arthrex products in a follow-up suit that was scheduled for trial in February 2017 in the Oregon court. Arthrex asserted its own suture anchor patents against Smith & Nephew in 2014 and 2015 in the US District Court for the Eastern District of Texas. In December 2016, the jury in that case decided that two of the Group’s US subsidiaries infringed two asserted Arthrex patents and awarded Arthrex $17.4m. In February 2017, the parties reached a settlement resulting in the dismissal of all patent litigation in Oregon and Texas. Smith & Nephew agreed to pay Arthrex $8m, and each party agreed to additional payments contingent on the outcome of patent validity proceedings currently pending at the US Patent & Trademark Office relating to the asserted patents. In November 2017, the US Patent & Trademark Office issued a Reexamination Certificate confirming validity of certain claims of US Patent No. 5,601,557 asserted by Smith & Nephew against Arthrex in the Oregon litigation. The issuing of the Reexamination Certificate triggered a payment of $80m which was received by Smith & Nephew in December 2017, and $54m (net of various expenses) is recognised in the Group’s 2017 operating profit. The Group has fully provided for any possible additional payment relating to its historical sales.
In February 2016, ConforMIS, Inc. filed suit against the Group’s US subsidiary in the Eastern Division of the US District Court for the District of Massachusetts, alleging that a number of its patents (generally directed to patient specific instrumentation associated with knee arthroplasty) are infringed by Smith & Nephew’s VISIONAIRE cutting guides and associated knee implants. The suit requested damages and an injunction. Smith & Nephew sought to invalidate the asserted patents at the US Patent & Trademark Office and has also filed counterclaims for infringement by ConforMis of the Group’s US patents. In September 2018, the Group entered into a settlement with ConforMis whereby Smith & Nephew paid $10.5m to settle the dispute and obtain a non-exclusive license under ConforMis patents pertaining to patient-specific instruments.
Smith & Nephew brought suit against Hologic in the US District Court for Massachusetts (Boston) in June 2010 for infringement of two patents. A trial was held in September 2012. The jury returned a verdict in Smith & Nephew’s favour, finding both asserted patents valid and infringed. Smith & Nephew’s motion for permanent injunction was granted, but stayed pending the result of re-examination proceedings instituted by Hologic in the United States Patent & Trademark Office (USPTO). In August 2016, Smith & Nephew divested its Gynaecology business to Medtronic, but retained rights to assert these patents against Hologic. On 25 October 2016, the USPTO upheld validity of one of the patents and Hologic appealed this decision. On 14 March 2018, the Federal Circuit affirmed the USPTO and again upheld validity of the patent. In October 2018, the parties reached a settlement and Hologic agreed to pay an amount approximating $35m. After deductions of payments to interested third parties, the Group recognised approximately $23m from this payment in the Group’s 2018 operating profit.
17.4 Tax Matters
At any given time the Group has unagreed years outstanding in various countries and is involved in tax audits and disputes, some of which may take several years to resolve. The Group believes that it has made adequate provision in respect of related additional tax liabilities that may arise. See Note 5 for further details.
|
|
Smith & Nephew Annual Report 2018 |
|
165 |
18 RETIREMENT BENEFIT OBLIGATIONS
|
Accounting policy The Group sponsors defined benefit plans in a number of countries. A defined benefit pension plan defines an amount of pension benefit that an employee will receive on retirement or a minimum guaranteed return on contributions, which is dependent on various factors such as age, years of service and final salary. The Group’s obligation is calculated separately for each plan by discounting the estimated future benefit that employees have earned in return for their service in the current and prior periods. The fair value of any plan assets is deducted to arrive at the net liability. The calculation of the defined benefit obligation is performed annually by external actuaries using the projected unit credit method. Remeasurements arising from defined benefit plans comprise actuarial gains and losses and the return on the plan assets net of the costs of managing the plan assets. The Group recognises these immediately in other comprehensive income (OCI) and all other expenses, such as service cost, net interest cost, administration costs and taxes, are recognised in the income statement. A number of key assumptions are made when calculating the fair value of the Group’s defined benefit pension plans. These assumptions impact the balance sheet asset and liabilities, operating profit, finance income/costs and other comprehensive income. The most critical assumptions are the discount rate, the rate of inflation and mortality assumptions to be applied to future pension plan liabilities. The discount rate is based on the yield at the reporting date on bonds that have a credit rating of AA, denominated in the currency in which the benefits are expected to be paid and have a maturity profile approximately the same as the Group’s obligations. In determining these assumptions management take into account the advice of professional external actuaries and benchmarks its assumptions against external data. The Group determines the net interest expense/income on the net defined benefit liability/asset for the period by applying the discount rate used to measure the defined benefit obligation at the beginning of the annual period to the net defined benefit liability/asset. The Group also operates a number of defined contribution plans. A defined contribution plan is a pension plan under which the Group and employees pay fixed contributions to a third party financial provider. The Group has no further payment obligations once the contributions have been paid. Contributions are recognised as an employee benefit expense when they are due. |
18.1 Retirement benefit net assets/(obligations)
The Group’s retirement benefit assets/(obligations) comprise:
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Funded plans: |
|
|
|
|
|
|
UK Plan |
|
77 |
|
53 |
|
|
US Plan |
|
13 |
|
9 |
|
|
Other plans |
|
(34) |
|
(46) |
|
|
|
|
56 |
|
16 |
|
|
Unfunded plans: |
|
|
|
|
|
|
Other plans |
|
(60) |
|
(60) |
|
|
Retirement healthcare |
|
(18) |
|
(25) |
|
|
|
|
(22) |
|
(69) |
|
|
Amount recognised on the balance sheet – liability |
|
(114) |
|
(131) |
|
|
Amount recognised on the balance sheet – asset |
|
92 |
|
62 |
|
The Group sponsors defined benefit pension plans for its employees or former employees in 14 countries and these are established under the laws of the relevant country. Funded plans are funded by the payment of contributions and the assets are held by separate trust funds or insurance companies. The provision of retirement and related benefits across the Group is kept under regular review. Employees’ retirement benefits are the subject of regular management review. The Group’s defined benefit plans provide employees with an entitlement to retirement benefits varying between 1.3% and 66.7% of final salary on attainment of retirement age. The level of entitlement is dependent on the years of service of the employee.
The Group’s two major defined benefit pension plans are in the UK and US. Both these plans were closed to new employees in 2003 and defined contribution plans are offered to new joiners. The US and UK Plans were closed to future accrual in March 2014 and December 2016 respectively.
The UK Plan operates under trust law and responsibility for its governance lies with a Board of Trustees. This Board is composed of representatives of the Group, plan participants and an independent trustee, who act on behalf of members in accordance with the terms of the Trust Deed and Rules and relevant legislation. The UK Plan’s assets are held by the trust. Annual increases on benefits in payment are dependent on inflation.
|
166 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
The 2018 court case in relation to Guaranteed Minimum Pensions does not impact the UK Plan as members were not contracted out of the State Earnings Related Pension (Serps) between 1990 and 1997.
There is no legislative minimum funding requirement in the UK, however the Group has agreed with the Board of Trustees to pay a schedule of supplementary payments (see Note 18.8). The Trust Deed of the UK Plan and the Plan Document of the US Plan provide the Group with a right to a refund of surplus assets assuming the full settlement of plan liabilities in the event of a plan wind-up. Furthermore, in the ordinary course of business the UK trustee and US committee have no rights to unilaterally wind up, or otherwise augment the benefits due to members of the plans. Based on these rights, any net surplus in the UK and US Plans is recognised in full.
The US Plan is governed by a US Pension Committee which is comprised of both plan participants and representatives of the Group. In the US, the Pension Protection Act (2006) established both a minimum required contribution and a maximum deductible contribution. Failure to contribute at least the minimum required amount will subject the Company to significant penalties, and contributions in excess of the maximum deductible have negative tax consequences. The minimum funding requirement is intended to fully fund the present value of accrued benefits over seven years.
18.2 Reconciliation of benefit obligations and pension assets
The movement in the Group’s pension benefit obligation and pension assets is as follows:
|
|
|
|
|
|
|
2018 |
|
|
|
|
|
2017 |
|
|
|
|
Obligation |
|
Asset |
|
Total |
|
Obligation |
|
Asset |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Amounts recognised on the balance sheet at beginning of the period |
|
(1,625) |
|
1,556 |
|
(69) |
|
(1,577) |
|
1,413 |
|
(164) |
|
|
Income statement expense: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current service cost |
|
(12) |
|
– |
|
(12) |
|
(12) |
|
– |
|
(12) |
|
|
Past service credit |
|
7 |
|
– |
|
7 |
|
4 |
|
– |
|
4 |
|
|
Settlements |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
|
Interest (expense)/income |
|
(40) |
|
40 |
|
– |
|
(44) |
|
42 |
|
(2) |
|
|
Administration costs and taxes |
|
(3) |
|
– |
|
(3) |
|
(3) |
|
– |
|
(3) |
|
|
Costs recognised in income statement |
|
(48) |
|
40 |
|
(8) |
|
(55) |
|
42 |
|
(13) |
|
|
Re-measurements: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Actuarial gain due to liability experience |
|
6 |
|
– |
|
6 |
|
1 |
|
– |
|
1 |
|
|
Actuarial gain/(loss) due to financial assumptions change |
|
97 |
|
– |
|
97 |
|
(38) |
|
– |
|
(38) |
|
|
Actuarial gain due to demographic assumptions |
|
11 |
|
– |
|
11 |
|
42 |
|
– |
|
42 |
|
|
Return on plan assets (less than)/more than discount rate |
|
– |
|
(103) |
|
(103) |
|
– |
|
59 |
|
59 |
|
|
Re-measurements recognised in OCI |
|
114 |
|
(103) |
|
11 |
|
5 |
|
59 |
|
64 |
|
|
Cash: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Employer contributions |
|
– |
|
44 |
|
44 |
|
– |
|
53 |
|
53 |
|
|
Employee contributions |
|
(4) |
|
4 |
|
– |
|
(4) |
|
4 |
|
– |
|
|
Benefits paid directly by the Group |
|
3 |
|
(3) |
|
– |
|
2 |
|
(2) |
|
– |
|
|
Benefits paid, taxes and administration costs paid from scheme assets |
|
100 |
|
(100) |
|
– |
|
102 |
|
(102) |
|
– |
|
|
Net cash |
|
99 |
|
(55) |
|
44 |
|
100 |
|
(47) |
|
53 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Exchange rates |
|
50 |
|
(50) |
|
– |
|
(98) |
|
89 |
|
(9) |
|
|
Amount recognised on the balance sheet |
|
(1,410) |
|
1,388 |
|
(22) |
|
(1,625) |
|
1,556 |
|
(69) |
|
|
Amount recognised on the balance sheet – liability |
|
(245) |
|
131 |
|
(114) |
|
(290) |
|
159 |
|
(131) |
|
|
Amount recognised on the balance sheet – asset |
|
(1,165) |
|
1,257 |
|
92 |
|
(1,335) |
|
1,397 |
|
62 |
|
|
|
Smith & Nephew Annual Report 2018 |
|
167 |
Represented by:
|
|
|
|
|
|
|
2018 |
|
|
|
|
|
2017 |
|
|
|
|
Obligation |
|
Asset |
|
Total |
|
Obligation |
|
Asset |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
UK Plan |
|
(718) |
|
795 |
|
77 |
|
(854) |
|
907 |
|
53 |
|
|
US Plan |
|
(424) |
|
437 |
|
13 |
|
(481) |
|
490 |
|
9 |
|
|
Other Plans |
|
(268) |
|
156 |
|
(112) |
|
(290) |
|
159 |
|
(131) |
|
|
Total |
|
(1,410) |
|
1,388 |
|
(22) |
|
(1,625) |
|
1,556 |
|
(69) |
|
All benefits are vested at the end of each reporting period. The weighted average duration of the defined benefit obligation at the end of the reporting period is 19 years and 11 years for the UK and US Plans respectively.
18.3 Plan assets
The market value of the US, UK and Other Plans assets are as follows:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
UK Plan: |
|
|
|
|
|
|
|
|
Assets with a quoted market price: |
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
2 |
|
8 |
|
6 |
|
|
Equity securities |
|
127 |
|
235 |
|
213 |
|
|
Other bonds |
|
41 |
|
43 |
|
38 |
|
|
Liability driven investments |
|
246 |
|
192 |
|
239 |
|
|
Diversified growth funds |
|
138 |
|
152 |
|
130 |
|
|
|
|
554 |
|
630 |
|
626 |
|
|
Other assets: |
|
|
|
|
|
|
|
|
Insurance contract |
|
241 |
|
277 |
|
214 |
|
|
Market value of assets |
|
795 |
|
907 |
|
840 |
|
|
US Plan: |
|
|
|
|
|
|
|
|
Assets with a quoted market price: |
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
– |
|
– |
|
– |
|
|
Equity securities |
|
79 |
|
88 |
|
178 |
|
|
Government bonds – fixed interest |
|
91 |
|
201 |
|
128 |
|
|
Corporate bonds |
|
267 |
|
201 |
|
128 |
|
|
Market value of assets |
|
437 |
|
490 |
|
434 |
|
|
Other Plans: |
|
|
|
|
|
|
|
|
Assets with a quoted market price: |
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
2 |
|
4 |
|
4 |
|
|
Equity securities |
|
42 |
|
43 |
|
35 |
|
|
Government bonds – fixed interest |
|
3 |
|
4 |
|
3 |
|
|
Government bonds – index linked |
|
3 |
|
3 |
|
3 |
|
|
Corporate and other bonds |
|
13 |
|
11 |
|
11 |
|
|
Insurance contracts |
|
34 |
|
36 |
|
34 |
|
|
Property |
|
20 |
|
19 |
|
12 |
|
|
Other quoted securities |
|
4 |
|
2 |
|
2 |
|
|
|
|
121 |
|
122 |
|
104 |
|
|
Other assets: |
|
|
|
|
|
|
|
|
Insurance contracts |
|
35 |
|
37 |
|
35 |
|
|
Market value of assets |
|
156 |
|
159 |
|
139 |
|
|
Total market value of assets |
|
1,388 |
|
1,556 |
|
1,413 |
|
|
168 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
No plans invest directly in property occupied by the Group or in financial securities issued by the Group.
Both the UK and US Plans hold a mixture of growth assets and matching assets. The growth assets of the UK and US Plans are invested in a diversified range of industries across a broad range of geographies. The UK Plan matching assets include liability matching assets and annuity policies purchased by the trustees, which aim to match the benefits to be paid to certain members from the plan and therefore remove the investment, inflation and demographic risks in relation to those liabilities. The terms of the policy define that the contract value exactly matches the amount and timing of the pensioner obligations covered by the contract. In accordance with IAS 19R Employee Benefits, the fair value of the insurance contract is deemed to be the present value of the related obligations which is discounted at the AA corporate bond rate. In December 2014, the low risk asset portfolio held by the UK Plan was transferred into liability driven investments (LDI) which invests in a mixture of gilts and swaps.
18.4 Expenses recognised in the income statement
The total expense relating to retirement benefits recognised for the year is $65m (2017: $64m, 2016: $23m). Of this cost recognised for the year, $57m (2017: $51m, 2016: $48m) relates to defined contribution plans and $8m (2017: $13m net credit, 2016: $25m net credit) relates to defined benefit plans.
The cost charged in respect of the Group’s defined contribution plans represents contributions payable to these plans by the Group at rates specified in the rules of the plans. These were charged to operating profit in selling, general and administrative expenses. There were $nil outstanding payments as at 31 December 2018 due to be paid over to the plans (2017: $nil, 2016: $nil).
In 2016, the $25m net credit for the year includes a $44m curtailment gain arising from the closure of the UK Plan to future accrual and $5m past service credit relating to redundancies.
Defined benefit plan costs comprise service cost which is charged to operating profit in selling, general and administrative expenses and net interest cost and administration costs and taxes which are reported as other finance costs.
The defined benefit pension costs charged for the UK and US Plans are:
|
|
|
2018 |
|
2017 |
|
2016 |
|
||||||
|
|
|
UK Plan |
|
US Plan |
|
UK Plan |
|
US Plan |
|
UK Plan |
|
US Plan |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Service cost |
|
– |
|
– |
|
– |
|
– |
|
7 |
|
– |
|
|
Past service credit |
|
– |
|
– |
|
– |
|
– |
|
(49) |
|
– |
|
|
Settlement loss |
|
– |
|
– |
|
– |
|
– |
|
1 |
|
– |
|
|
Net interest cost, administration and taxes |
|
– |
|
– |
|
1 |
|
2 |
|
– |
|
3 |
|
|
|
|
– |
|
– |
|
1 |
|
2 |
|
(41) |
|
3 |
|
18.5 Principal actuarial assumptions
The following are the principal financial actuarial assumptions used at the reporting date to determine the UK and US defined benefit obligations and expense.
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
% per annum |
|
% per annum |
|
% per annum |
|
|
UK Plan: |
|
|
|
|
|
|
|
|
Discount rate |
|
2.7 |
|
2.4 |
|
2.6 |
|
|
Future salary increases |
|
n/a |
|
n/a |
|
3.8 |
|
|
Future pension increases |
|
3.2 |
|
3.2 |
|
3.3 |
|
|
Inflation (RPI) |
|
3.2 |
|
3.2 |
|
3.3 |
|
|
Inflation (CPI) |
|
2.2 |
|
2.2 |
|
2.3 |
|
|
US Plan: |
|
|
|
|
|
|
|
|
Discount rate |
|
4.2 |
|
3.5 |
|
4.0 |
|
|
Future salary increases |
|
n/a |
|
n/a |
|
n/a |
|
|
Inflation |
|
n/a |
|
n/a |
|
n/a |
|
|
|
Smith & Nephew Annual Report 2018 |
|
169 |
Actuarial assumptions regarding future mortality are based on mortality tables. The UK uses the S2NA with projections in line with the CMI 2016 table and the US uses the RP2014 table with MP2018 scale. The current longevities underlying the values of the obligations in the defined benefit plans are as follows:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
years |
|
years |
|
years |
|
|
Life expectancy at age 60 |
|
|
|
|
|
|
|
|
UK Plan: |
|
|
|
|
|
|
|
|
Males |
|
28.9 |
|
28.8 |
|
29.7 |
|
|
Females |
|
30.4 |
|
30.3 |
|
31.1 |
|
|
US Plan: |
|
|
|
|
|
|
|
|
Males |
|
24.9 |
|
25.2 |
|
25.1 |
|
|
Females |
|
27.1 |
|
27.4 |
|
27.4 |
|
|
Life expectancy at age 60 in 20 years’ time |
|
|
|
|
|
|
|
|
UK Plan: |
|
|
|
|
|
|
|
|
Males |
|
31.1 |
|
31.0 |
|
32.5 |
|
|
Females |
|
31.9 |
|
31.8 |
|
33.0 |
|
|
US Plan: |
|
|
|
|
|
|
|
|
Males |
|
25.1 |
|
25.5 |
|
25.4 |
|
|
Females |
|
27.7 |
|
28.0 |
|
27.9 |
|
18.6 Sensitivity analysis
The calculation of the defined benefit obligation is sensitive to the assumptions used. The following table summarises the increase/decrease on the UK and US defined benefit obligation and pension costs as a result of reasonably possible changes in some of the assumptions while holding all other assumptions consistent. The sensitivity to the inflation assumption change includes corresponding changes to the future salary increases and future pension increase assumptions. The analysis does not take into account the full distribution of cash flows expected under the plan.
Changes to the inflation assumption will not have any effect on the US Pension Plan as it was closed to future accrual in 2014 and it has no other inflation-linked assumptions.
|
|
|
Increase in pension obligation |
|
Increase in pension cost |
|
||||
|
$ million |
|
+50bps/+1yr |
|
‑50bps/‑1yr |
|
+50bps/+1 yr |
|
‑50bps/‑1yr |
|
|
UK Plan: |
|
|
|
|
|
|
|
|
|
|
Discount rate |
|
-67.2 |
|
+73.0 |
|
-2 |
|
+2 |
|
|
Inflation |
|
+66.6 |
|
-60.2 |
|
+1 |
|
-1 |
|
|
Mortality |
|
+29.0 |
|
-28.8 |
|
+1 |
|
– |
|
|
US Plan: |
|
|
|
|
|
|
|
|
|
|
Discount rate |
|
-21.0 |
|
+23.0 |
|
-1 |
|
+1 |
|
|
Inflation |
|
n/a |
|
n/a |
|
n/a |
|
n/a |
|
|
Mortality |
|
+10.0 |
|
-10.2 |
|
– |
|
– |
|
|
170 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
18.7 Risk
The pension plans expose the Group to the following risks:
|
Interest rate risk |
Volatility in financial markets can change the calculations of the obligation significantly as the calculation of the obligation is linked to yields on AA-rated corporate bonds. A decrease in the bond yield will increase the measure of plan liabilities, although this will be partially offset by increases in the value of matching plan assets such as bonds and insurance contracts. In the UK, the liability matching portfolio held in conventional and index-linked gilts was transferred into liability driven investments in order to reduce interest rate risk. |
|
Inflation risk |
The UK Plan is linked to inflation. A high rate of inflation will lead to a higher liability. This risk is managed by holding inflation-linked bonds and an inflation-linked insurance contract in respect of some of the obligation. In the UK, the liability matching portfolio held in conventional and index-linked gilts was transferred into liability driven investments in order to reduce inflation risk. The UK Plan is closed to future accrual which reduces the exposure to this risk. The US Plan is also closed to future accrual and has no other inflation-linkage thus eliminating the exposure to this risk. |
|
Investment risk |
If the return on plan assets is below the discount rate, all else being equal, there will be an increase in the plan deficit. In the UK, this risk is partially managed by a portfolio of liability matching assets and a bulk annuity, together with a dynamic de-risking policy to switch growth assets into liability matching assets over time. The US Plan has a dynamic de-risking policy to shift plan assets from return-seeking (growth) assets to liability matching assets over time. The US Pension Plan has an established glide path with two remaining funding level triggers that are designed to stabilize funding status by reducing the Plan’s exposure to return-seeking assets. |
|
Longevity risk |
The present value of the plans defined benefit liability is calculated by reference to the best estimate of the mortality of the plan participants both during and after their employment. An increase in the life expectancy of plan participants above that assumed will increase the benefit obligation. The UK Plan, in order to minimise longevity risk, has entered into an insurance contract which covers a portion of pensioner obligations. |
18.8 Funding
A full valuation is performed by actuaries for the Trustees of each plan to determine the level of funding required. Employer contributions rates, based on these full valuations, are agreed between the Trustees of each plan and the Group. The assumptions used in the actuarial valuations used for funding purposes may differ from those assumptions above.
UK Plan
The most recent full actuarial valuation of the UK Plan was undertaken as at 30 September 2015. The next full actuarial valuation as at 30 September 2018 has commenced. Contributions to the UK Plan in 2018 were $25m (2017: $24m, 2016: $32m). This included supplementary payments of $25m (2017: $24m, 2016: $26m).
The Group has currently agreed to pay annual supplementary payments of $25m until 2021. These supplementary payments will be reviewed when the 30 September 2018 valuation has been completed.
US Plan
The most recent full actuarial valuation of the US Plan was undertaken as at 1 January 2018. The next full actuarial valuation will take place as at 1 January 2019. Future accruals to the US Plan ceased as at 31 March 2014. Contributions to the US Plan were $10m (2017: $20m, 2016: $20m) which represented supplementary payments of $10m (2017: $20m, 2016: $20m).
The planned supplementary contribution for 2019 is being kept under review given the funding status.
|
|
Smith & Nephew Annual Report 2018 |
|
171 |
|
Accounting policy Incremental costs directly attributable to the issue of ordinary shares, net of any tax effects, are recognised as a deduction from equity. When shares recognised as equity are repurchased, the amount of the consideration paid, which includes directly attributable costs, net of any tax effects, is recognised as a deduction from equity. Repurchased shares are classified as treasury shares and are presented in the treasury share reserve. When treasury shares are sold or reissued subsequently, the amount received is recognised as an increase in equity and the resulting surplus or deficit on the transaction is presented within share premium. |
19.1 Share capital
|
|
|
Ordinary shares (20¢) |
|
Deferred shares (£1.00) |
|
Total |
|
||||
|
|
|
Thousand |
|
$ million |
|
Thousand |
|
$ million |
|
$ million |
|
|
Authorised |
|
|
|
|
|
|
|
|
|
|
|
|
At 31 December 2016 |
|
1,223,591 |
|
245 |
|
50 |
|
– |
|
245 |
|
|
At 31 December 2017 |
|
1,223,591 |
|
245 |
|
50 |
|
– |
|
245 |
|
|
At 31 December 2018 |
|
1,223,591 |
|
245 |
|
50 |
|
– |
|
245 |
|
|
Allotted, issued and fully paid |
|
|
|
|
|
|
|
|
|
|
|
|
At 1 January 2016 |
|
915,447 |
|
183 |
|
50 |
|
– |
|
183 |
|
|
Share options |
|
1,283 |
|
– |
|
– |
|
– |
|
– |
|
|
Shares cancelled |
|
(13,007) |
|
(3) |
|
– |
|
– |
|
(3) |
|
|
At 31 December 2016 |
|
903,723 |
|
180 |
|
50 |
|
– |
|
180 |
|
|
Share options |
|
655 |
|
– |
|
– |
|
– |
|
– |
|
|
Shares cancelled |
|
(13,523) |
|
(2) |
|
– |
|
– |
|
(2) |
|
|
At 31 December 2017 |
|
890,855 |
|
178 |
|
50 |
|
– |
|
178 |
|
|
Share options |
|
418 |
|
– |
|
– |
|
– |
|
– |
|
|
Shares cancelled |
|
(3,321) |
|
(1) |
|
– |
|
– |
|
(1) |
|
|
At 31 December 2018 |
|
887,952 |
|
177 |
|
50 |
|
– |
|
177 |
|
The deferred shares were issued in 2006 in order to comply with English Company law. They are not listed on any stock exchange and have extremely limited rights and effectively have no value. These rights are summarised as follows:
|
- |
The holder shall not be entitled to participate in the profits of the Company; |
|
- |
The holder shall not have any right to participate in any distribution of the Company’s assets on a winding up or other distribution except that after the return of the nominal amount paid up on each share in the capital of the Company of any class other than the deferred shares and the distribution of a further $1,000 in respect of each such share there shall be distributed to a holder of a deferred share (for each deferred share held) an amount equal to the nominal value of the deferred share; |
|
- |
The holder shall not be entitled to receive notice, attend, speak or vote at any general meeting of the Company; and |
|
- |
The Company may create, allot and issue further shares or reduce or repay the whole or any part of its share capital or other capital reserves without obtaining the consent of the holders of the deferred shares. |
The Group’s objectives when managing capital are to ensure the Group has adequate funds to continue as a going concern and sufficient flexibility within the capital structure to fund the ongoing growth of the business and to take advantage of business development opportunities including acquisitions.
The Group determines the amount of capital taking into account changes in business risks and future cash requirements. The Group reviews its capital structure on an ongoing basis and uses share buy-backs, dividends and the issue of new shares to adjust the retained capital.
|
172 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
The Group considers the capital that it manages to be as follows:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Share capital |
|
177 |
|
178 |
|
180 |
|
|
Share premium |
|
608 |
|
605 |
|
600 |
|
|
Capital redemption reserve |
|
18 |
|
17 |
|
15 |
|
|
Treasury shares |
|
(214) |
|
(257) |
|
(432) |
|
|
Retained earnings and other reserves |
|
4,285 |
|
4,101 |
|
3,595 |
|
|
|
|
4,874 |
|
4,644 |
|
3,958 |
|
19.2 Treasury shares
Treasury shares represent the holding of the Company’s own shares in respect of the Smith & Nephew Employees’ Share Trust and shares bought back as part of the share buy-back programme. In 2018 the Group purchased a total of 2.7m shares for a cost of $48m as part of the ongoing programme to buy back an equivalent number of shares to those vesting as part of the employee share plans. In 2017 the Group purchased a total of 3.2m shares for a cost of $52m as part of the same programme.
The Smith & Nephew 2004 Employees’ Share Trust (Trust) was established to hold shares relating to the long-term incentive plans referred to in the ‘Directors’ Remuneration Report’. The Trust is administered by an independent professional trust company resident in Jersey and is funded by a loan from the Company. The cost of the Trust is charged to the income statement as it accrues. A dividend waiver is in place in respect of those shares held under the long-term incentive plans. The Trust only accepts dividends in respect of nil-cost options and deferred bonus plan shares. The waiver represents less than 1% of the total dividends paid.
The movements in Treasury shares and the Employees’ Share Trust are as follows:
|
|
|
|
|
Employees’ |
|
|
|
|
|
|
Treasury |
|
Share Trust |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
At 1 January 2017 |
|
411 |
|
21 |
|
432 |
|
|
Shares purchased |
|
52 |
|
– |
|
52 |
|
|
Shares transferred from treasury |
|
(19) |
|
19 |
|
– |
|
|
Shares transferred to Group beneficiaries |
|
(9) |
|
(17) |
|
(26) |
|
|
Shares cancelled |
|
(201) |
|
– |
|
(201) |
|
|
At 31 December 2017 |
|
234 |
|
23 |
|
257 |
|
|
Shares purchased |
|
48 |
|
– |
|
48 |
|
|
Shares transferred from treasury |
|
(29) |
|
29 |
|
– |
|
|
Shares transferred to Group beneficiaries |
|
(13) |
|
(27) |
|
(40) |
|
|
Shares cancelled |
|
(51) |
|
– |
|
(51) |
|
|
At 31 December 2018 |
|
189 |
|
25 |
|
214 |
|
|
|
|
Number |
|
Number |
|
Number |
|
|
|
|
of shares |
|
of shares |
|
of shares |
|
|
|
|
million |
|
million |
|
million |
|
|
At 1 January 2017 |
|
27.8 |
|
1.5 |
|
29.3 |
|
|
Shares purchased |
|
3.2 |
|
– |
|
3.2 |
|
|
Shares transferred from treasury |
|
(1.3) |
|
1.3 |
|
– |
|
|
Shares transferred to Group beneficiaries |
|
(0.6) |
|
(1.2) |
|
(1.8) |
|
|
Shares cancelled |
|
(13.5) |
|
– |
|
(13.5) |
|
|
At 31 December 2017 |
|
15.6 |
|
1.6 |
|
17.2 |
|
|
Shares purchased |
|
2.7 |
|
– |
|
2.7 |
|
|
Shares transferred from treasury |
|
(1.9) |
|
1.9 |
|
– |
|
|
Shares transferred to Group beneficiaries |
|
(0.9) |
|
(1.8) |
|
(2.7) |
|
|
Shares cancelled |
|
(3.3) |
|
– |
|
(3.3) |
|
|
At 31 December 2018 |
|
12.2 |
|
1.7 |
|
13.9 |
|
|
|
Smith & Nephew Annual Report 2018 |
|
173 |
19.3 Dividends
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
The following dividends were declared and paid in the year: |
|
|
|
|
|
|
|
|
Ordinary final of 22.7¢ for 2017 (2016: 18.5¢, 2015: 19.0¢) paid 9 May 2018 |
|
198 |
|
162 |
|
170 |
|
|
Ordinary interim of 14.0¢ for 2018 (2017: 12.3¢, 2016: 12.3¢) paid 31 October 2018 |
|
123 |
|
107 |
|
109 |
|
|
|
|
321 |
|
269 |
|
279 |
|
A final dividend for 2018 of 22.0¢ per ordinary share was proposed by the Board on 7 February 2019 and will be paid, subject to shareholder approval, on 8 May 2019 to shareholders on the Register of Members on 5 April 2019. The estimated amount of this dividend is $192m. The Group pursues a progressive dividend policy, with the aim of increasing the US Dollar value of ordinary dividends over time broadly based on the Group’s underlying growth in earnings, while taking into account capital requirements and cash flows. Future dividends will be dependent upon future earnings, the future financial condition of the Group and the Board’s dividend policy. The Board reviews the appropriate level of total annual dividend each year at the time of the full year results. The Board intends that the interim dividend will be set by a formula and will be equivalent to 40% of the total dividend for the previous year. Smith & Nephew plc, the Parent Company of the Group, is a non-trading investment holding company which derives its distributable reserves from dividends paid by subsidiary companies. The distributable reserves of the Parent Company approximate to the balance on the profit and loss account reserve, less treasury shares and exchange reserves, which at 31 December 2018 amounted to $2,274m.
|
Accounting policy In the Group cash flow statement, cash and cash equivalents includes cash at bank, other short-term liquid investments with original maturities of three months or less and bank overdrafts. In the Group balance sheet, bank overdrafts are shown within bank overdrafts and loans under current liabilities. |
Analysis of net debt
|
|
Borrowings |
|
|
|
|||||||||||
|
|
|
|
|
|
|
Due within |
|
Due after |
|
Net |
|
Net |
|
|
|
|
|
|
Cash |
|
Overdrafts |
|
one year |
|
one year |
|
currency swaps |
|
interest swaps |
|
Total |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 1 January 2016 |
|
120 |
|
(18) |
|
(28) |
|
(1,434) |
|
(2) |
|
1 |
|
(1,361) |
|
|
Net cash flow impact |
|
(18) |
|
(45) |
|
4 |
|
(129) |
|
25 |
|
(2) |
|
(165) |
|
|
Exchange adjustment |
|
(2) |
|
1 |
|
– |
|
(1) |
|
(22) |
|
– |
|
(24) |
|
|
At 31 December 2016 |
|
100 |
|
(62) |
|
(24) |
|
(1,564) |
|
1 |
|
(1) |
|
(1,550) |
|
|
Net cash flow impact |
|
64 |
|
49 |
|
9 |
|
139 |
|
(24) |
|
(1) |
|
236 |
|
|
Termination of finance lease |
|
– |
|
– |
|
2 |
|
3 |
|
– |
|
– |
|
5 |
|
|
Exchange adjustment |
|
5 |
|
(1) |
|
– |
|
(1) |
|
25 |
|
– |
|
28 |
|
|
At 31 December 2017 |
|
169 |
|
(14) |
|
(13) |
|
(1,423) |
|
2 |
|
(2) |
|
(1,281) |
|
|
Net cash flow/debt movement |
|
200 |
|
(18) |
|
(118) |
|
126 |
|
8 |
|
(1) |
|
197 |
|
|
Exchange adjustment |
|
(4) |
|
– |
|
(1) |
|
(4) |
|
(11) |
|
– |
|
(20) |
|
|
At 31 December 2018 |
|
365 |
|
(32) |
|
(132) |
|
(1,301) |
|
(1) |
|
(3) |
|
(1,104) |
|
|
174 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
Reconciliation of net cash flow to movement in net debt
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Net cash flow from cash net of overdrafts |
|
182 |
|
113 |
|
(63) |
|
|
Settlement of currency swaps |
|
8 |
|
(24) |
|
25 |
|
|
Net cash flow from borrowings |
|
7 |
|
147 |
|
(127) |
|
|
Change in net debt from net cash flow |
|
197 |
|
236 |
|
(165) |
|
|
Termination of finance lease |
|
– |
|
5 |
|
– |
|
|
Exchange adjustment |
|
(20) |
|
28 |
|
(24) |
|
|
Change in net debt in the year |
|
177 |
|
269 |
|
(189) |
|
|
Opening net debt |
|
(1,281) |
|
(1,550) |
|
(1,361) |
|
|
Closing net debt |
|
(1,104) |
|
(1,281) |
|
(1,550) |
|
Cash and cash equivalents
For the purposes of the Group cash flow statement cash and cash equivalents at 31 December 2018 comprise cash at bank net of bank overdrafts.
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Cash at bank |
|
365 |
|
169 |
|
100 |
|
|
Bank overdrafts |
|
(32) |
|
(14) |
|
(62) |
|
|
Cash and cash equivalents |
|
333 |
|
155 |
|
38 |
|
The Group operates in over 100 countries around the world, some of which impose restrictions over cash movement. These restrictions have only a minimal impact of the management of the Group’s cash.
Cash (inflows)/outflows arising from financing activities
|
|
|
Repayment |
|
Borrowing |
|
Cash |
|
|
|
|
|
Proceeds from own |
|
|
|
|
|
|
of bank |
|
of bank |
|
outflow |
|
|
|
Purchase of |
|
shares/issue of |
|
|
|
|
|
|
loans |
|
loans |
|
from other |
|
Dividends |
|
own shares |
|
ordinary shares |
|
Total |
|
|
2018 |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Debt |
|
401 |
|
(394) |
|
8 |
|
– |
|
– |
|
– |
|
15 |
|
|
Equity |
|
– |
|
– |
|
– |
|
321 |
|
48 |
|
(13) |
|
356 |
|
|
Total |
|
401 |
|
(394) |
|
8 |
|
321 |
|
48 |
|
(13) |
|
371 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2017 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Debt |
|
770 |
|
(623) |
|
(24) |
|
– |
|
– |
|
– |
|
123 |
|
|
Equity |
|
– |
|
– |
|
– |
|
269 |
|
52 |
|
(10) |
|
311 |
|
|
Total |
|
770 |
|
(623) |
|
(24) |
|
269 |
|
52 |
|
(10) |
|
434 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2016 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Debt |
|
797 |
|
(924) |
|
25 |
|
– |
|
– |
|
– |
|
(102) |
|
|
Equity |
|
– |
|
– |
|
– |
|
279 |
|
368 |
|
(16) |
|
631 |
|
|
Total |
|
797 |
|
(924) |
|
25 |
|
279 |
|
368 |
|
(16) |
|
529 |
|
|
|
Smith & Nephew Annual Report 2018 |
|
175 |
|
Accounting policy The Group accounts for business combinations using the acquisition method when control is transferred to the Group. The consideration transferred in the acquisition is measured at fair value, as are the identifiable net assets acquired. Any goodwill that arises is tested annually for impairment. Any gain on a bargain purchase is recognised in profit or loss immediately. Transaction costs are expensed as incurred, except if related to the issue of debt or equity securities. Any contingent consideration payable is measured at fair value at the acquisition date. If the contingent consideration is classified as equity, then it is not remeasured and settlement is accounted for within equity. Otherwise, subsequent changes in the fair value of the contingent consideration are recognised in profit or loss. |
21.1 Acquisitions
Year ended 31 December 2018
The Group made no acquisitions deemed to be business combinations within the scope of IFRS 3 in the year ended 31 December 2018. The cash outflow of $29m relates to acquisitions completed in prior years.
Year ended 31 December 2017
During the year ended 31 December 2017, the Group acquired one medical technology business deemed to be a business combination within the scope of IFRS 3 Business Combinations as follows. The acquisition accounting was completed in 2018 with no adjustments to the provisional fair value disclosed in the Group’s 2017 Annual Report.
On 5 December 2017, the Group completed the acquisition of 100% of the share capital of Rotation Medical, Inc., a developer of a novel tissue regeneration technology for shoulder rotator cuff repair. The acquisition furthers our strategy to invest in disruptive technologies that accelerate the transformation of Smith & Nephew to higher growth. The maximum consideration payable of $210m has a fair value of $196m and includes $17m of deferred and $72m of contingent consideration. The fair value of the contingent consideration is determined from the acquisition agreement, the Board-approved acquisition model and a risk-free discount rate of 2.5%. The maximum contingent consideration is $85m. The fair values of assets acquired and liabilities assumed are set out below:
|
|
|
$ million |
|
|
Intangible assets |
|
61 |
|
|
Property, plant & equipment and inventory |
|
3 |
|
|
Trade and other receivables |
|
2 |
|
|
Trade and other payables |
|
(3) |
|
|
Net deferred tax assets |
|
1 |
|
|
Net assets |
|
64 |
|
|
Goodwill |
|
132 |
|
|
Consideration (net of $nil cash acquired) |
|
196 |
|
The goodwill is attributable to the control premium, the acquired workforce and the synergies that can be expected from integrating Rotation Medical, Inc. into the Group’s existing business. The goodwill is not expected to be deductible for tax purposes.
During the year ended 31 December 2017, the contribution to revenue and attributable profit from this acquisition is immaterial. If the acquisition had occurred at the beginning of the year, its contribution to revenue and attributable profit would have also been immaterial.
Year ended 31 December 2016
During the year ended 31 December 2016, the Group acquired two medical technology businesses deemed to be business combinations within the scope of IFRS 3 Business Combinations. The acquisition accounting was completed during 2017 with no measurement adjustments made.
On 4 January 2016, the Group completed the acquisition of 100% of the share capital of Blue Belt Holdings Inc., a business specialising in robotic technologies. The acquisition secures a leading position in the fast growing area of Orthopaedic robotics-assisted surgery. The fair value of consideration is $265m and includes $51m deferred consideration. The fair values of assets acquired were:
|
176 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE GROUP ACCOUNTS continued
|
|
|
$ million |
|
|
Aggregate identifiable assets acquired and liabilities assumed |
|
|
|
|
Intangible assets |
|
70 |
|
|
Property, plant & equipment and inventory |
|
13 |
|
|
Trade and other payables |
|
(11) |
|
|
Provisions |
|
(10) |
|
|
Deferred tax assets |
|
16 |
|
|
Net assets |
|
78 |
|
|
Goodwill |
|
184 |
|
|
Consideration (net of $3m cash acquired) |
|
262 |
|
The goodwill is attributable to the revenue synergies of providing a full robotic surgery offering and future applications of the technological expertise. The goodwill is not expected to be deductible for tax purposes.
On 8 January 2016 the Group completed the acquisition of BST-CarGel, a first-line cartilage repair product from Piramal Healthcare (Canada) Limited. The fair value of the consideration is $42m and included $37m of deferred and contingent consideration. The fair values of net assets acquired are: product intangible assets of $15m, inventory of $1m, and a deferred tax liability of $1m. The goodwill, which is expected to be deductible for tax purposes, arising on the acquisition is $27m, is attributable to the future penetration into new markets expected from the transaction. During the year ended 31 December 2016, the contribution to revenue and attributable profit from these acquisitions is immaterial. If the acquisitions had occurred at the beginning of the year, their contribution to revenue and attributable profit would have also been immaterial.
21.2 Disposal of business
During the year ended 31 December 2016, the Group disposed of its Gynaecology business for cash consideration of $350m. The net assets disposed included $6m plant and equipment, and $4m inventory. Disposal related costs of $7m and liabilities of $7m resulted in a pre-tax gain on disposal of $326m. Tax paid on the disposal was $118m. For the years ended 31 December 2017 and 31 December 2018, the Group did not dispose of any businesses.
|
Accounting policy Leases are classified as finance leases when the terms of the lease transfer substantially all the risks and rewards of ownership to the Group. All other leases are classified as operating leases. Payments under operating leases are expensed in the income statement on a straight-line basis over the term of the lease. Lease incentives received are recognised as an integral part of the total lease expense, over the term of the lease. |
Future minimum lease payments under non-cancellable operating leases fall due as follows:
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Land and buildings: |
|
|
|
|
|
|
Within one year |
|
37 |
|
40 |
|
|
After one and within two years |
|
30 |
|
35 |
|
|
After two and within three years |
|
27 |
|
27 |
|
|
After three and within four years |
|
22 |
|
23 |
|
|
After four and within five years |
|
16 |
|
19 |
|
|
After five years |
|
52 |
|
56 |
|
|
|
|
184 |
|
200 |
|
|
Other assets: |
|
|
|
|
|
|
Within one year |
|
17 |
|
17 |
|
|
After one and within two years |
|
11 |
|
11 |
|
|
After two and within three years |
|
4 |
|
5 |
|
|
After three and within four years |
|
2 |
|
1 |
|
|
|
|
34 |
|
34 |
|
|
|
Smith & Nephew Annual Report 2018 |
|
177 |
23 OTHER NOTES TO THE ACCOUNTS
|
Accounting policy The Group operates a number of equity-settled executive and employee share plans. For all grants of share options and awards, the fair value at the grant date is calculated using appropriate option pricing models. The grant date fair value is recognised over the vesting period as an expense, with a corresponding increase in retained earnings. |
The Group operates the following equity-settled executive and employee share plans: Smith & Nephew Global Share Plan 2010, Smith & Nephew ShareSave Plan (2012), Smith & Nephew International ShareSave Plan (2012) and the Smith & Nephew France ShareSave plan (2012). At 31 December 2018, 4,911,000 options (2017: 5,277,000, 2016: 5,780,000) were outstanding with a range of exercise prices from 538 to 1,097 pence.
At 31 December 2018, the maximum number of shares that could be awarded under the Group’s long-term incentive plans was 5,678,000 (2017: 5,854,000, 2016: 5,807,000). These include conditional share awards granted to senior employees and equity and performance share awards granted to senior executives under the Global Share Plan 2010.
The expense charged to the income statement for share-based payments for the year is $35m (2017: $31m, 2016: $27m).
23.2 Related party transactions
Trading transactions
In the course of normal operations, the Group traded with its associates detailed in Note 11. The aggregated transactions, which have not been disclosed elsewhere in the financial statements are $nil (2017: $nil, 2016: $nil).
Key management personnel
The remuneration of executive officers (including Non-Executive Directors) during the year is summarised below:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
|
Short-term employee benefits |
|
18 |
|
15 |
|
15 |
|
|
Share-based payments expense |
|
10 |
|
7 |
|
7 |
|
|
Pension and post-employment benefit entitlements |
|
2 |
|
1 |
|
1 |
|
|
Compensation for loss of office |
|
– |
|
3 |
|
– |
|
|
|
|
30 |
|
26 |
|
23 |
|
Directors’ remuneration disclosures are included on pages 84–105.
On 22 January 2019 the Group completed the acquisition of 100% of the share capital of Ceterix Orthopaedics, Inc., the developer of the NovoStitch™ Pro Meniscal Repair System. This unique device addresses complex tear patterns not adequately served by other repair systems and is highly complementary to the Group’s FAST-FIX™ 360 Meniscal Repair System.
This acquisition will be treated as a business combination under IFRS 3. The maximum consideration, all payable in cash, is $105m and the provisional fair value consideration is $96m and includes $5m of deferred consideration and $46m of contingent consideration which relates to the achievement of established milestones and targets. The fair value of contingent consideration is determined from the acquisition agreement, the Board approved acquisition model and a risk-free discount rate of 3.3%. Acquired net assets have a provisional value of $2m which is not expected to have material fair value adjustments. The remaining $94m will be allocated between identifiable intangible assets including technology, research and development in-progress and goodwill, with the majority expected to be goodwill. Goodwill represents the control premium, the acquired workforce and the synergies expected from integrating Ceterix Orthopaedics, Inc. into the Group’s existing business, and is not expected to be deductible for tax purposes. The contribution to revenue and attributable profit from this acquisition is expected to be immaterial for the year ending 31 December 2019.
|
178 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
|
|
|
|
|
At 31 December |
|
At 31 December |
|
|
|
|
|
|
2018 |
|
2017 |
|
|
|
|
Notes |
|
$ million |
|
$ million |
|
|
Fixed assets |
|
|
|
|
|
|
|
|
Investments |
|
2 |
|
7,092 |
|
7,092 |
|
|
Current assets |
|
|
|
|
|
|
|
|
Debtors |
|
3 |
|
1,697 |
|
1,084 |
|
|
Cash at bank |
|
5 |
|
277 |
|
88 |
|
|
|
|
|
|
1,974 |
|
1,172 |
|
|
Creditors: amounts falling due within one year |
|
|
|
|
|
|
|
|
Borrowings |
|
5 |
|
(145) |
|
(4) |
|
|
Other creditors |
|
4 |
|
(2,277) |
|
(1,202) |
|
|
|
|
|
|
(2,422) |
|
(1,206) |
|
|
Net current liabilities |
|
|
|
(448) |
|
(34) |
|
|
Total assets less current liabilities |
|
|
|
6,644 |
|
7,058 |
|
|
Creditors: amounts falling due after one year |
|
|
|
|
|
|
|
|
Borrowings |
|
5 |
|
(1,301) |
|
(1,423) |
|
|
Total assets less total liabilities |
|
|
|
5,343 |
|
5,635 |
|
|
|
|
|
|
|
|
|
|
|
Equity shareholders’ funds |
|
|
|
|
|
|
|
|
Called up equity share capital |
|
|
|
177 |
|
178 |
|
|
Share premium account |
|
|
|
608 |
|
605 |
|
|
Capital redemption reserve |
|
|
|
18 |
|
17 |
|
|
Capital reserve |
|
|
|
2,266 |
|
2,266 |
|
|
Treasury shares |
|
|
|
(214) |
|
(257) |
|
|
Exchange reserve |
|
|
|
(52) |
|
(52) |
|
|
Profit and loss account |
|
|
|
2,540 |
|
2,878 |
|
|
Shareholders’ funds |
|
|
|
5,343 |
|
5,635 |
|
The accounts were approved by the Board and authorised for issue on 21 February 2019 and signed on its behalf by:
|
Roberto Quarta |
Namal Nawana |
Graham Baker |
|
Chairman |
Chief Executive Officer |
Chief Financial Officer |
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
|
Smith & Nephew Annual Report 2018 |
|
179 |
Statement of changes in equity
|
|
|
|
|
|
|
Capital |
|
|
|
|
|
|
|
|
|
Total |
|
|
|
|
Share |
|
Share |
|
redemption |
|
Capital |
|
Treasury |
|
Exchange |
|
Profit and |
|
shareholders’ |
|
|
|
|
capital |
|
premium |
|
reserve |
|
reserves |
|
shares |
|
reserves |
|
loss account |
|
funds |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
At 1 January 2017 |
|
180 |
|
600 |
|
15 |
|
2,266 |
|
(432) |
|
(52) |
|
1,169 |
|
3,746 |
|
|
Attributable profit for the year |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
2,167 |
|
2,167 |
|
|
Net gain on cash flow hedges |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
1 |
|
1 |
|
|
Exchange adjustments |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
1 |
|
1 |
|
|
Equity dividends paid in the year |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(269) |
|
(269) |
|
|
Share-based payments recognised1 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
31 |
|
31 |
|
|
Cost of shares transferred to beneficiaries |
|
– |
|
– |
|
– |
|
– |
|
26 |
|
– |
|
(21) |
|
5 |
|
|
New shares issued on exercise of share options |
|
– |
|
5 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
5 |
|
|
Cancellation of treasury shares |
|
(2) |
|
– |
|
2 |
|
– |
|
201 |
|
– |
|
(201) |
|
– |
|
|
Treasury shares purchased |
|
– |
|
– |
|
– |
|
– |
|
(52) |
|
– |
|
– |
|
(52) |
|
|
At 31 December 2017 |
|
178 |
|
605 |
|
17 |
|
2,266 |
|
(257) |
|
(52) |
|
2,878 |
|
5,635 |
|
|
Attributable profit for the year |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
28 |
|
28 |
|
|
Net gain on cash flow hedges |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
1 |
|
1 |
|
|
Equity dividends paid in the year |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(321) |
|
(321) |
|
|
Share-based payments recognised1 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
– |
|
35 |
|
35 |
|
|
Cost of shares transferred to beneficiaries |
|
– |
|
– |
|
– |
|
– |
|
40 |
|
– |
|
(30) |
|
10 |
|
|
New shares issued on exercise of share options |
|
– |
|
3 |
|
– |
|
– |
|
– |
|
– |
|
– |
|
3 |
|
|
Cancellation of treasury shares |
|
(1) |
|
– |
|
1 |
|
– |
|
51 |
|
– |
|
(51) |
|
– |
|
|
Treasury shares purchased |
|
– |
|
– |
|
– |
|
– |
|
(48) |
|
– |
|
– |
|
(48) |
|
|
At 31 December 2018 |
|
177 |
|
608 |
|
18 |
|
2,266 |
|
(214) |
|
(52) |
|
2,540 |
|
5,343 |
|
|
1 |
The Company operates a number of equity-settled executive and employee share plans. For all grants of share options and awards, the fair value as at the date of grant is calculated using an appropriate option pricing model and the corresponding expense is recognised over the vesting period. Subsidiary companies are recharged for the fair value of share options that relate to their employees. The disclosure relating to the Company is detailed in Note 23.1 of the Notes to the Group accounts. |
Further information on the share capital of the Company can be found in Note 19.1 of the Notes to the Group accounts.
The total distributable reserves of the Company are $2,274m (2017: $2,569m). In accordance with the exemption permitted by Section 408 of the Companies Act 2006, the Company has not presented its own profit and loss account. The attributable profit for the year dealt with in the accounts of the Company is $28m (2017: $2,167m).
Fees paid to KPMG LLP for audit and non-audit services to the Company itself are not disclosed in the individual accounts because Group financial statements are prepared which are required to disclose such fees on a consolidated basis. The fees for the consolidated Group are disclosed in Note 3.2 of the Notes to the Group accounts.
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
180 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
Smith & Nephew plc (the Company) is a public limited company incorporated in England and Wales.
The separate accounts of the Company are presented as required by the Companies Act 2006. On 1 January 2015, the Company transitioned from previously extant UK Generally Accepted Accounting Practices to Financial Reporting Standard 101 Reduced Disclosure Framework (‘Reduced Disclosure Framework’). These financial statements and accompanying notes have been prepared in accordance with the Reduced Disclosure Framework for all periods presented. There were no transitional adjustments required on adoption of the new standard. The financial information for the Company has been prepared on the same basis as the consolidated financial statements, applying identical accounting policies as outlined throughout the Notes to the Group accounts. The Directors have determined that the preparation of the Company financial statements on a going concern basis is appropriate as the Company receives dividend cash receipts from its subsidiary undertakings which enable it to meet its liabilities as they fall due.
In applying these policies, management is required to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the accounts and the reported amounts of revenues and expenses during the reporting period. Although these estimates are based on management’s best knowledge of current events and actions, actual results ultimately may differ from those estimates.
In these financial statements, the Company has applied the exemptions available under FRS 101 in respect of the following disclosures:
A Cash Flow Statement and related notes;
Comparative period reconciliations for share capital and tangible fixed assets;
Disclosures in respect of transactions with wholly-owned subsidiaries;
Disclosures in respect of capital management;
The effects of new but not yet effective IFRSs; and
Disclosures in respect of the compensation of key management personnel.
As the consolidated financial statements include the equivalent disclosures, the Company has also taken the exemptions under FRS 101 available in respect of the following disclosures:
IFRS 2 Share Based Payments in respect of group settled share based payments; and
Certain disclosures required by IFRS 13 Fair Value Measurement and the disclosures required by IFRS 7 Financial Instrument Disclosures.
The Company proposes to continue to adopt the reduced disclosure framework of FRS 101 in its next financial statements.
2 INVESTMENTS
|
ACCOUNTING POLICY Investments in subsidiaries are stated at cost less provision for impairment. |
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
At 1 January |
|
7,092 |
|
5,322 |
|
|
Additions |
|
– |
|
1,770 |
|
|
At 31 December |
|
7,092 |
|
7,092 |
|
Investments represent holdings in subsidiary undertakings. In 2017, the Company increased its investment in Smith & Nephew (Overseas) Limited.
In accordance with Section 409 of the Companies Act 2006, a listing of all entities invested in by the consolidated Group is provided in Note 8.
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
|
Smith & Nephew Annual Report 2018 |
|
181 |
3 DEBTORS
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Amounts falling due within one year: |
|
|
|
|
|
|
Amounts owed by subsidiary undertakings |
|
1,635 |
|
1,007 |
|
|
Prepayments and accrued income |
|
3 |
|
3 |
|
|
Current asset derivatives – forward foreign exchange contracts |
|
36 |
|
25 |
|
|
Current asset derivatives – forward foreign exchange contracts – subsidiary undertakings |
|
20 |
|
45 |
|
|
Current asset derivatives – currency swaps |
|
1 |
|
3 |
|
|
Current taxation |
|
2 |
|
1 |
|
|
|
|
1,697 |
|
1,084 |
|
Allowance losses on amounts owed by subsidiary undertakings are calculated by reviewing 12-month expected credit losses using historic and forward-looking data on credit risk. The loss allowance expense for the year was $nil (2017: $nil).
4 OTHER CREDITORS
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Amounts falling due within one year: |
|
|
|
|
|
|
Amounts owed to subsidiary undertakings |
|
2,204 |
|
1,119 |
|
|
Other creditors |
|
12 |
|
10 |
|
|
Current liability derivatives – forward foreign exchange contracts |
|
20 |
|
45 |
|
|
Current liability derivatives – forward foreign exchange contracts – subsidiary undertakings |
|
36 |
|
25 |
|
|
Current liability derivatives – currency swaps |
|
2 |
|
1 |
|
|
Current liability derivatives – interest rate swaps |
|
3 |
|
2 |
|
|
|
|
2,277 |
|
1,202 |
|
5 CASH AND BORROWINGS
|
ACCOUNTING POLICY Financial instruments Currency swaps are used to match foreign currency assets with foreign currency liabilities. They are initially recorded at fair value and then for reporting purposes remeasured to fair value at exchange rates and interest rates at subsequent balance sheet dates. |
Changes in the fair value of derivative financial instruments are recognised in the profit and loss account as they arise.
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Bank loans, borrowing and overdrafts due within one year or on demand |
|
145 |
|
4 |
|
|
Borrowings due after one year |
|
1,301 |
|
1,423 |
|
|
Borrowings |
|
1,446 |
|
1,427 |
|
|
Cash at bank |
|
(277) |
|
(88) |
|
|
Credit/(debit) balance on derivatives – currency swaps |
|
1 |
|
(2) |
|
|
Credit balance on derivatives - interest rate swaps |
|
3 |
|
2 |
|
|
Net debt |
|
1,173 |
|
1,339 |
|
All currency swaps are stated at fair value. Gross US Dollar equivalents of $253m (2017: $388m) receivable and $254m (2017: $386m) payable have been netted. Currency swaps comprise foreign exchange swaps and were used in 2018 and 2017 to hedge intra-group loans.
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
182 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE COMPANY ACCOUNTS continued
6 CONTINGENCIES
|
|
|
2018 |
|
2017 |
|
|
|
|
$ million |
|
$ million |
|
|
Guarantees in respect of subsidiary undertakings |
|
– |
|
1 |
|
The Company gives guarantees to banks to support liabilities and cross guarantees to support overdrafts.
The Company operated defined benefit pension plans in 2004 but at the end of 2005 its pension plan obligations were transferred to Smith & Nephew UK Limited. The Company has provided guarantees to the trustees of the pension plans to support future amounts due from participating employers (see Note 18 of the Notes to the Group accounts).
7 DEFERRED TAXATION
The Company has gross unused capital losses of $80m (2017: $90m) available for offset against future chargeable gains. No deferred tax asset has been recognised on these unused losses as they are not expected to be realised in the foreseeable future.
8 GROUP COMPANIES
In accordance with Section 409 of the Companies Act 2006, a full list of subsidiaries, associates, joint arrangements, joint ventures and partnerships are listed below, including their country of incorporation. All companies are 100% owned, unless otherwise indicated. The share capital disclosed comprises ordinary shares which are indirectly held by Smith & Nephew plc, unless otherwise stated.
|
Company name |
|
Country of |
|
Registered |
|
Company name |
|
Country of |
|
Registered |
|
UK |
|
|
|
|
|
Smith & Nephew Nominee Company Limited2 |
|
England & Wales |
|
London |
|
Blue Belt Technologies UK Limited2 |
England & Wales |
London |
Smith & Nephew Nominee Services Limited2 |
England & Wales |
London |
|||||
|
Michelson Diagnostic Limited3 (7%) |
|
England & Wales |
|
Kent |
|
Smith & Nephew Orthopaedics Limited |
|
England & Wales |
|
London |
|
Neotherix Limited3 (24.9%) |
|
England & Wales |
|
York |
|
Smith & Nephew Pensions Nominees Limited2 |
|
England & Wales |
|
London |
|
Plus Orthopedics (UK) Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew Pharmaceuticals Limited2 |
|
England & Wales |
|
Hull |
|
Smith & Nephew (Overseas) Limited1,5 |
|
England & Wales |
|
London |
|
Smith & Nephew Raisegrade Limited1,2 |
|
England & Wales |
|
London |
|
Smith & Nephew ARTC Limited |
|
England & Wales |
|
London |
|
Smith & Nephew Rareletter Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew Beta Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew Trading Group Limited1 |
|
England & Wales |
|
London |
|
Smith & Nephew China Holdings UK Limited1 |
|
England & Wales |
|
London |
|
Smith & Nephew UK Executive Pension Scheme Trustee Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew Consumer Products Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew UK Limited1,5 |
|
England & Wales |
|
London |
|
Smith & Nephew Employees Trustees Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew UK Pension Fund Trustee Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew ESN Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew USD Limited1 |
|
England & Wales |
|
London |
|
Smith & Nephew Extruded Films Limited |
|
England & Wales |
|
Hull |
|
Smith & Nephew USD One Limited1 |
|
England & Wales |
|
London |
|
Smith & Nephew Finance2 |
|
England & Wales |
|
London |
|
T.J. Smith and Nephew, Limited |
|
England & Wales |
|
Hull |
|
Smith & Nephew Finance Oratec2 |
|
England & Wales |
|
London |
|
The Albion Soap Company Limited2 |
|
England & Wales |
|
London |
|
Smith & Nephew Healthcare Limited2 |
|
England & Wales |
|
Hull |
|
TP Limited1 |
|
Scotland |
|
Edinburgh |
|
Smith & Nephew Investment Holdings Limited1 |
|
England & Wales |
|
London |
|
|
|
|
|
|
|
Smith & Nephew Medical Fabrics Limited2 |
|
England & Wales |
|
London |
|
|
|
|
|
|
|
Smith & Nephew Medical Limited |
|
England & Wales |
|
Hull |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
|
Smith & Nephew Annual Report 2018 |
|
183 |
|
Company name |
|
Country of |
|
Registered |
|
Company name |
|
Country of |
|
Registered |
|
Rest of Europe |
|
|
|
|
|
|
|
|
|
|
|
Smith & Nephew GmbH |
|
Austria |
|
Vienna |
|
US |
|
|
|
|
|
ArthroCare Belgium SPRL2 |
|
Belgium |
|
Zaventem |
|
Arthrocare Corporation1 |
|
United States |
|
Wilmington |
|
Smith & Nephew S.A.-N.V |
|
Belgium |
|
Zaventem |
|
Bioventus LLC3 (49%) |
|
United States |
|
Wilmington |
|
Smith & Nephew A/S |
|
Denmark |
|
Hoersholm |
|
Blue Belt Holdings, Inc.1 |
|
United States |
|
Wilmington |
|
Smith & Nephew Oy |
|
Finland |
|
Helsinki |
|
Blue Belt Technologies, Inc.1 |
|
United States |
|
Philadelphia |
|
A2 Surgical2 |
|
France |
|
Neuilly-sur-Seine |
|
Charlie Merger Corp. |
|
United States |
|
Wilmington 19808 |
|
Smith & Nephew France SAS1 |
|
France |
|
Neuilly-sur-Seine |
|
Delphi Ventures V, L.P.3 (6.9%) |
|
United States |
|
Wilmington |
|
Smith & Nephew S.A.S. |
|
France |
|
Neuilly-sur-Seine |
|
Healicoil, Inc. |
|
United States |
|
Wilmington |
|
Smith & Nephew Business Services GmbH & Co. KG1 |
|
Germany |
|
Hamburg |
|
Hipco, Inc. |
|
United States |
|
Wilmington |
|
Smith & Nephew Business Services Verwaltungs GmbH |
|
Germany |
|
Hamburg |
|
Leaf Healthcare Inc.3 (11%) |
|
United States |
|
Wilmington |
|
Smith & Nephew Deutschland (Holding) GmbH1 |
|
Germany |
|
Hamburg |
|
Memphis Biomed Ventures I, LP3 (4.61%) |
|
United States |
|
Dover |
|
Smith & Nephew GmbH |
|
Germany |
|
Hamburg |
|
Miach Orthopaedics, Inc3 (8.3%) |
|
United States |
|
Sherborn |
|
Smith & Nephew Orthopaedics GmbH |
|
Germany |
|
Tuttlingen |
|
Oratec Interventions, Inc. |
|
United States |
|
Wilmington |
|
Plus Orthopedics Hellas S.A.4 |
|
Greece |
|
Athens |
|
Orthopaedic Biosystems Ltd., Inc. |
|
United States |
|
Phoenix |
|
Smith & Nephew Hellas S.A.4 |
|
Greece |
|
Athens |
|
OsteoBiologics, Inc. |
|
United States |
|
Wilmington |
|
Smith & Nephew (Ireland) Trading Limited |
|
Ireland |
|
Dublin 2 |
|
Plus Orthopedics LLC |
|
United States |
|
Wilmington |
|
Smith & Nephew Finance Ireland Limited |
|
Ireland |
|
Dublin 1 |
|
Rotation Medical, Inc. |
|
United States |
|
Wilmington |
|
Smith & Nephew S.r.l. |
|
Italy |
|
Milan |
|
Sinopsys Surgical, Inc.3 (12.4%) |
|
United States |
|
Wilmington |
|
ArthroCare Luxembourg S.a.r.l.1,2 |
|
Luxembourg |
|
Luxembourg |
|
Smith & Nephew Consolidated, Inc.1 |
|
United States |
|
Wilmington |
|
Smith & Nephew Finance S.a.r.l.1 |
|
Luxembourg |
|
Luxembourg |
|
Smith & Nephew OUS, Inc. |
|
United States |
|
Wilmington |
|
Smith & Nephew International S.A.1 |
|
Luxembourg |
|
Luxembourg |
|
Smith & Nephew, Inc.1 |
|
United States |
|
Wilmington |
|
Smith & Nephew (Europe) B.V.1 |
|
Netherlands |
|
Amsterdam |
|
Surgical Frontiers Series I, LLC3 (33.46%) |
|
United States |
|
Dover |
|
Smith & Nephew B.V. |
|
Netherlands |
|
Amsterdam |
|
Trice Medical Inc.3 (6%) |
|
United States |
|
Wilmington 19808 |
|
Smith & Nephew Management B.V.1 |
|
Netherlands |
|
Amsterdam |
|
|
|
|
|
|
|
Smith & Nephew Nederland CV |
|
Netherlands |
|
Amsterdam |
|
Africa, Asia, Australasia and Other America |
|
|
|
|
|
Smith & Nephew A/S |
|
Norway |
|
Oslo |
|
Smith & Nephew Argentina S.R.L.2 |
|
Argentina |
|
Buenos Aires |
|
Smith & Nephew sp. z.o.o. |
|
Poland |
|
Warsaw |
|
ArthroCare (Australasia) Pty Ltd4 |
|
Australia |
|
North Ryde |
|
Smith & Nephew Lda |
|
Portugal |
|
Lisbon |
|
Smith & Nephew Pty Limited |
|
Australia |
|
North Ryde |
|
DC LLC |
|
Russian Federation |
|
Puschino |
|
Smith & Nephew Surgical Holdings Pty Limited1,2 |
|
Australia |
|
North Ryde |
|
Smith & Nephew LLC |
|
Russian Federation |
|
Moscow |
|
Smith & Nephew Surgical Pty Limited1,2 |
|
Australia |
|
North Ryde |
|
Smith & Nephew S.A.U |
|
Spain |
|
Barcelona |
|
Smith & Nephew Comercio de Produtos Medicos LTDA |
|
Brazil |
|
São Paulo |
|
Smith & Nephew Aktiebolag |
|
Sweden |
|
Molndal |
|
Smith & Nephew (Alberta) Inc.2 |
|
Canada |
|
Calgary |
|
Lumina Adhesives AB3 (11%) |
|
Sweden |
|
Gothenburg |
|
Smith & Nephew Inc. |
|
Canada |
|
Toronto |
|
Plus Orthopedics Holding AG1 |
|
Switzerland |
|
Baar |
|
Tenet Medical Engineering, Inc. |
|
Canada |
|
Calgary |
|
Smith & Nephew Manufacturing AG |
|
Switzerland |
|
Aarau |
|
Smith & Nephew Finance Holdings Limited2,5 |
|
Cayman Islands |
|
South Church Street, George Town |
|
Smith & Nephew Orthopaedics AG1 |
|
Switzerland |
|
Baar |
|
ArthoCare Medical Devices (Beijing) Co. Limited4 |
|
China |
|
Chao Yang District, Beijing |
|
Smith & Nephew Schweiz AG |
|
Switzerland |
|
Baar |
|
Plus Orthopedics (Beijing) Co. Limited2 |
|
China |
|
Shunyi District, Beijing |
|
Smith & Nephew AG |
|
Switzerland |
|
Baar |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
184 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE COMPANY ACCOUNTS continued
|
Company name |
|
Country of |
|
Registered |
|
Company name |
|
Country of |
|
Registered |
|
|
|
|
|
|
|
|
|
|
|
|
|
Company name |
|
Country of |
|
Registered |
|
Registered Office addresses |
||||
|
Smith & Nephew Medical (Shanghai) Limited |
|
China |
|
Shanghai Free |
|
UK |
|
|
||
|
Smith & Nephew Medical (Suzhou) Limited |
|
China |
|
Suzhou City |
|
London |
|
15 Adam Street, London, WC2N 6LA |
||
|
Smith & Nephew Orthopaedics (Beijing) Co., Ltd |
|
China |
|
Beijing Economic and |
|
Kent |
|
Ground Floor, Eclipse House, Eclipse Park, Sittingbourne Road, Maidstone, Kent, ME14 3EN |
||
|
|
|
|
|
Technical |
|
York |
|
25 Carr Lane, York, YO26 5HT |
||
|
|
|
|
|
Development |
|
Hull |
|
101 Hessle Road, Hull, HU3 2BN |
||
|
|
|
|
|
Area |
|
Edinburgh |
|
4th Floor, 115 George Street, Edinburgh, EH2 4JN |
||
|
S&N Holdings SAS1 |
|
Colombia |
|
Bogota |
|
Rest of Europe |
||||
|
Smith & Nephew Colombia S.A.S |
|
Colombia |
|
Bogota |
|
Vienna |
|
Concorde Business Park, 1/C/3 2320, Schwechat, Austria |
||
|
ArthroCare Costa Rica Srl |
|
Costa Rica |
|
Costa Rica |
|
Zaventem |
|
Hector Heenneaulaan 366, 1930 Zaventem, Belgium |
||
|
Smith & Nephew Curaçao N.V. |
|
Curaçao |
|
Willemstad |
|
Hoersholm |
|
Slotsmarken 14, Hoersholm, DK‑2970, Denmark |
||
|
Smith & Nephew Beijing Holdings Limited1 |
|
Hong Kong |
|
Hong Kong |
|
Helsinki |
|
Ayritie 12 C, 01510, Vantaa, Finland |
||
|
Smith & Nephew Limited |
|
Hong Kong |
|
Hong Kong |
|
Neuilly-sur-Seine |
|
40, Boulevard du Parc, 92200 Neuilly-sur-Seine, France |
||
|
Smith & Nephew Suzhou Holdings Limited1 |
|
Hong Kong |
|
Hong Kong |
|
Hamburg |
|
Friesenweg 4, Haus 21, 22763, Hamburg, Germany |
||
|
Adler Mediequip Private Limited |
|
India |
|
Pune |
|
Tuttlingen |
|
Alemannenstrasse 14, 78532, Tuttlingen, Germany |
||
|
ArthroCare India Medical Device Private Limited2 |
|
India |
|
Mumbai |
|
Athens |
|
Protopappa Street 43, GR 16346, Ilioupoli, Athens, Greece |
||
|
Smith & Nephew Healthcare Private Limited |
|
India |
|
Mumbai‑59 |
|
Dublin 1 |
|
3rd Floor, Kilmore House, Park Lane, Spencer Dock, Dublin 1, Ireland |
||
|
Ortho-Space Ltd.3 (16.8%) |
|
Israel |
|
Caesarea |
|
Dublin 2 |
|
13-18 City Quay, Dublin 2, D02 ED70, Ireland |
||
|
Smith & Nephew KK |
|
Japan |
|
Tokyo |
|
Milan |
|
Via de Capitani 2A, 20864, Agrate Brianza (MI), Italy |
||
|
Smith & Nephew Chusik Hoesia |
|
Korea, Republic of |
|
Seoul |
|
Luxembourg |
|
163, Rue de Kiem, L‑8030 Strassen, Luxembourg |
||
|
Smith & Nephew Healthcare Sdn Berhad |
|
Malaysia |
|
Kuala Lumpur |
|
Amsterdam |
|
Bloemlaan 2, 2132NP, Hoofddorp, The Netherlands |
||
|
Smith & Nephew Services SDN. BHD. |
|
Malaysia |
|
Kuala Lumpur |
|
Oslo |
|
Nye Vakas vei 64, 1395, Hvalsted, Norway |
||
|
Smith & Nephew S.A. de C.V. |
|
Mexico |
|
Mexico City |
|
Warsaw |
|
Ul Osmanska 12, 02‑823, Warsaw, Poland |
||
|
Smith & Nephew Limited1 |
|
New Zealand |
|
Auckland |
|
Lisbon |
|
Estrada Nacional no 10 ao Km. 131, Parque Tejo – Bloco C, 2625‑445 Forte de Casa, Vila Franca de Xira, Portugal |
||
|
Smith & Nephew Superannuation Scheme Limited |
|
New Zealand |
|
Auckland |
|
Moscow |
|
2nd Syromyatnichesky lane, Moscow, 105120, Russian Federation |
||
|
Smith & Nephew, Inc. |
|
Puerto Rico |
|
San Juan |
|
Puschino |
|
8/1 Stroiteley Street, 142290, City of Puschino, Moscow Region, Russian Federation |
||
|
Smith & Nephew Pte Limited1 |
|
Singapore |
|
Singapore |
|
Barcelona |
|
Edificio Conata I, c/Fructuos Gelabert 2 y 4, San Joan Despi – 08970, Barcelona, Spain |
||
|
Smith & Nephew (Pty) Limited1 |
|
South Africa |
|
Westville |
|
Molndal |
|
PO Box 143, S‑431 22 Molndal, Sweden |
||
|
Smith & Nephew Pharmaceuticals (Proprietary) Limited |
|
South Africa |
|
Westville |
|
Gothenburg |
|
Varbergsgatan 2A / 412 65 Göteborg/Sweden |
||
|
Smith & Nephew Limited |
|
Thailand |
|
Huai Khwang District, Bangkok |
|
Baar |
|
Oberneuhofstr 10d, Baar, 6340 |
||
|
Sri Siam Medical Limited1,3 (48.99%) |
|
Thailand |
|
Lumpini Phatumwan, Bangkok |
|
Aarau |
|
Schachenallee 29, 5000, Aarau, Switzerland |
||
|
Smith ve Nephew Medikal Cihazlar Ticaret Limited Sirketi |
|
Turkey |
|
Sariyer, Istanbul |
|
|
|
|
||
|
Smith & Nephew FZE |
|
United Arab Emirates |
|
Jebel Ali, Dubai |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 Holding company. 2 Dormant company. |
|
|
|
|
|
|
|
|
|
|
|
3 Not 100% owned by Smith & Nephew Group. |
|
|
|
|
|
|
|
|
||
|
4 In liquidation. |
|
|
|
|
|
|
|
|
|
|
|
5 Directly owned by Smith & Nephew plc. |
|
|
|
|
|
|
|
|
|
|
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
|
Smith & Nephew Annual Report 2018 |
|
185 |
|
Registered Office addresses |
|
Registered Office addresses |
||||
|
US |
|
|
|
Shunyi District, Beijing |
|
22 Linhe Avenue, Linhe Economic Development Zone, Shunyi District, Beijing, 101300, China |
|
Wilmington |
|
CT Corporation, 1209 Orange Street, Wilmington, DE 19801, USA |
|
Shanghai Free Trade Test Zone |
|
Part B, 4th Floor, Tong Yong Building, |
|
Philadelphia |
|
CT Corporation 1515 Market Street, |
|
Suzhou City |
|
12, Wuxiang Road, West Area of Comprehensive Bonded Zone, Suzhou Industrial Park, Suzhou City, SIP, Jiangsu Province, China |
|
Dover |
|
160 Greentree Drive, Suite 101, Dover, Delaware, 19904, USA |
|
Beijing Economic and Technical Development Area |
|
No. 98 Kechuang Dongliujie, |
|
Phoenix |
|
CT Corporation System, 3800 North Central Avenue, Phoenix AZ 85012, USA |
|
Bogota |
|
Calle 100 No. 7 – 33 to 1 P3, Bogota D.C., Colombia |
|
Sherborn |
|
c/o Martha Murray 19 Saddlebrook Road |
|
Costa Rica |
|
Building B32, 50 meters South of Revisión Téchnica Vehicular, Province de Alajuela, Canton Alajuela, Coyol Free Zone, District San José, Costa Rica |
|
Wilmington 19808 |
|
251 Little Falls Drive, Wilmington DE 19808, USA |
|
Willemstad |
|
Pietermaai 15, PO Box 4905, Curaçao |
|
Africa, Asia, Australasia and Other America |
|
Hong Kong |
|
Unit 813 – 816, 8/F, Delta House, 3 On Yiu Street, |
||
|
Buenos Aires |
|
Maipu 1300, 13th Floor, City of Buenos Aires, Argentina |
|
Pune |
|
Sushrut House, Survey no.288, |
|
North Ryde |
|
85 Waterloo Road, North Ryde NSW 2113, Australia |
|
Mumbai |
|
5A, Bakhtawar, 5th Floor, behind The Oberoi, |
|
São Paulo |
|
Avenida do Cafe, 277, Centro Empresarial do Aco, Centro Empresarial do Aco, Torre B, 4 andar, conjuto, CEP 04311-000, São Paulo 403, Jabaquara, Brazil |
|
Mumbai‑59 |
|
501‑B – 509‑B Dynasty Business Park, Andheri Kurla Road, Andheri East, Mumbai‑59, Maharashtra, India |
|
Calgary |
|
3500‑855‑2 Street SW, Calgary AB AB T2P 4J8, Canada |
|
Caesarea |
|
7 Halamarish, Caesarea, 3088900, Israel |
|
Toronto |
|
199, Bay Street, 4000, Toronto, Ontario M5L 1A9, Canada |
|
Tokyo |
|
2‑4‑1, Shiba -Koen, Minato-Ku, |
|
South Church Street, Georgetown |
|
c/o Maples Corporate Services Limited, P.O. Box 309, Ugland House, Grand Cayman, KY1-1104, Cayman Islands |
|
Seoul |
|
13th Floor, ASEM Tower, Gangnam-gu 13th Floor, |
|
Chao Yang District, Beijing |
|
Room 17‑021, Internal B17 floor, B3‑24th floor, No 3 Xin Yuan South Rd, Chao Yang District, Beijing, China |
|
Kuala Lumpur |
|
Level 25, Menara Hong Leong, NO. 6 Jalan Damanlela Bukit Damansara Kuala Lumpur W.P. 50490 Kuala Lumpur, Malaysia |
|
|
|
|
|
Mexico City |
|
Av. Insurgentes Sur, numero 1602, Piso No.7, |
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
186 |
Accounts |
Smith & Nephew Annual Report 2018 |
|
NOTES TO THE COMPANY ACCOUNTS continued
|
Registered Office addresses |
|
Registered Office addresses |
||||
|
Auckland |
|
36a Hillside Road, Wairau Valley, Auckland, 0627 NZ, New Zealand |
|
Lumpini Phatumwan, Bangkok |
|
16th Floor, GPF Witthayu Tower A, 93/1 Wireless Road, |
|
San Juan |
|
Edificio Cesar Castillo, Calle Angel Buonomo |
|
Sariyer, Istanbul |
|
Bahcekoy Merkez Mah. Ergene Nehri SK No:8/4 Bahcekoy Sariyer Istanbul, Turkey |
|
Singapore |
|
50 Raffles Place, #32‑01 Singapore Land Tower, |
|
Jebel Ali, Dubai |
|
PO Box 16993 LB02016, Jebel Ali, |
|
Westville |
|
30 The Boulevard, Westway Office Park, |
|
|
|
|
|
Huai Khwang District, Bangkok |
|
16th Floor Building A, 9th Tower Grand Rama 9, |
|
|
|
|
9 SUBSIDIARY UNDERTAKINGS EXEMPT FROM AUDIT
The following UK subsidiaries will take advantage of the audit exemption set out within Section 479A of the Companies Act 2006 for the year ended 31 December 2018:
–– Smith & Nephew China Holdings UK Limited (Registration number: 9152387)
–– Smith & Nephew Investment Holdings Limited (Registration number: 384546)
–– Smith & Nephew Trading Group Limited (Registration number: 681256)
–– Smith & Nephew USD One Limited (Registration number: 10428326)
–– TP Limited (Registration number: SC005366)
 THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
THE PARENT COMPANY FINANCIAL STATEMENTS OF SMITH & NEPHEW PLC ON PAGES 178-186 DO NOT FORM PART OF SMITH & NEPHEW’S
ANNUAL REPORT ON FORM 20-F AS FILED WITH THE SEC.
|
|
Smith & Nephew Annual Report 2018 |
|
187 |
BUSINESS OVERVIEW AND GROUP HISTORY
In 2018, Smith & Nephew’s operations were organised into geographical selling regions and product franchises within the medical technology industry.
The Group has a history dating back 160 years to the family enterprise of Thomas James Smith who opened a small pharmacy in Hull, UK in 1856. Following his death in 1896, his nephew Horatio Nelson Smith took over the management of the business.
By the late 1990s, Smith & Nephew had expanded into being a diverse healthcare company with operations across the globe, producing various medical devices, personal care products and traditional and advanced wound care treatments. In 1998, Smith & Nephew announced a major restructuring to focus management attention and investment on three global business units – Advanced Wound Management, Endoscopy and Orthopaedics – which offered high growth and margin opportunities. In 2011, the Endoscopy and Orthopaedics businesses were brought together to create an Advanced Surgical Devices division. In 2015, the Advanced Wound Management and Advanced Surgical Devices divisions were brought together to form a global business across nine product franchises, managed as three geographical selling regions with global functions for operations, R&D and corporate support functions. In 2019, a new global franchise commercial model will be implemented with three dedicated franchises: Orthopaedics, Sports Medicine & ENT, and Advanced Wound Management, each with their own president. The franchise presidents will be responsible for implementing the operating decisions for their respective franchise in the US. Regional presidents in EMEA and APAC will be responsible for this implementation in their respective regions.
Smith & Nephew was incorporated and listed on the London Stock Exchange in 1937 and in 1999 the Group was also listed on the New York Stock Exchange. In 2001, Smith & Nephew became a constituent member of the FTSE 100 index in the UK. This means that Smith & Nephew is included in the top 100 companies traded on the London Stock Exchange measured in terms of market capitalisation.
Today, Smith & Nephew is a public limited company incorporated and headquartered in the UK and carries out business around the world.
PROPERTIES
The table below summarises the main properties which the Group uses and their approximate areas.
|
|
|
Approximate area |
|
|
|
|
(square feet 000’s) |
|
|
Group head office in London, UK |
|
13 |
|
|
UK office and surgical training facility in Watford, UK |
|
60 |
|
|
Manufacturing and office facilities in Memphis, Tennessee, US |
|
968 |
|
|
Wound management manufacturing, research and office facility in Hull, UK |
|
473 |
|
|
Manufacturing facility in Suzhou, China |
|
288 |
|
|
Manufacturing facility in Alajuela, Costa Rica |
|
265 |
|
|
Distribution facility in Memphis, Tennessee, US |
|
248 |
|
|
Manufacturing facility in Beijing, China |
|
192 |
|
|
Manufacturing facility in Oklahoma City, Oklahoma, US |
|
155 |
|
|
Regional headquarters in Andover, Massachusetts, US |
|
144 |
|
|
Bioactives headquarters and laboratory space in Fort Worth, Texas, US |
|
139 |
|
|
Research and office facility in Austin, Texas, US |
|
136 |
|
|
Manufacturing facility in Aarau, Switzerland |
|
121 |
|
|
Manufacturing facility in Mansfield, Massachusetts, US |
|
98 |
|
|
Manufacturing facility in Devrukh, India |
|
74 |
|
|
Regional headquarters and distribution facility in Baar, Switzerland |
|
71 |
|
|
Manufacturing facility in Tuttlingen, Germany |
|
50 |
|
The Group Global Operations strategy includes ongoing assessment of the optimal facility footprint. The Orthopaedics manufacturing facilities in Memphis, Tennessee are largely freehold, a portion of Tuttlingen and the Advanced Wound Management facilities in Hull are freehold while other principal locations are leasehold. The Group has freehold and leasehold interests in real estate in other countries throughout the world, but no other is individually significant to the Group. Where required, the appropriate governmental authorities have approved the facilities.
OFF-BALANCE SHEET ARRANGEMENTS
Management believes that the Group does not have any off-balance sheet arrangements, as defined by the SEC in item 5E of Form 20-F, that have or are reasonably likely to have a current or future effect on the Group’s financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that is material to investors.
|
188 |
Other information |
Smith & Nephew Annual Report 2018 |
|
GROUP INFORMATION continued
RELATED PARTY TRANSACTIONS
Except for transactions with associates (see Note 23.2 of Notes to the Group accounts), no other related party had material transactions or loans with Smith & Nephew over the last three financial years.
RISK FACTORS
There are known and unknown risks and uncertainties relating to Smith & Nephew’s business. The factors listed on pages 188–191 could cause the Group’s business, financial position and results of operations to differ materially and adversely from expected and historical levels. In addition, other factors not listed here that Smith & Nephew cannot presently identify or does not believe to be equally significant could also materially adversely affect Smith & Nephew’s business, financial position or results of operations.
Highly competitive markets
The Group competes across a diverse range of geographic and product markets. Each market in which the Group operates contains a number of different competitors, including specialised and international corporations. Significant product innovations, technical advances or the intensification of price competition by competitors could adversely affect the Group’s operating results. Some of these competitors may have greater financial, marketing and other resources than Smith & Nephew. These competitors may be able to initiate technological advances in the field, deliver products on more attractive terms, more aggressively market their products or invest larger amounts of capital and research and development (R&D) into their businesses. There is a possibility of further consolidation of competitors, which could adversely affect the Group’s ability to compete with larger companies due to insufficient financial resources. If any of the Group’s businesses were to lose market share or achieve lower than expected revenue growth, there could be a disproportionate adverse impact on the Group’s share price and its strategic options. Competition exists among healthcare providers to gain patients on the basis of quality, service and price. There has been some consolidation in the Group’s customer base and this trend is expected to continue. Some customers have joined group purchasing organisations or introduced other cost containment measures that could lead to downward pressure on prices or limit the number of suppliers in certain business areas, which could adversely affect Smith & Nephew’s results of operations and hinder its growth potential.
Continual development and introduction of new products
The medical devices industry has a rapid rate of new product introduction. In order to remain competitive, the Group must continue to develop innovative products that satisfy customer needs and preferences or provide cost or other advantages. Developing new products is a costly, lengthy and uncertain process. The Group may fail to innovate due to low R&D investment, a R&D skills gap or poor product development. A potential product may not be brought to market or not succeed in the market for any number of reasons, including failure to work optimally, failure to receive regulatory approval, failure to be cost-competitive, infringement of patents or other intellectual property rights and changes in consumer demand. The Group’s products and technologies are also subject to marketing attack by competitors. Furthermore, new products that are developed and marketed by the Group’s competitors may affect price levels in the various markets in which the Group operates. If the Group’s new products do not remain competitive with those of competitors, the Group’s revenue could decline.
The Group maintains reserves for excess and obsolete inventory resulting from the potential inability to sell its products at prices in excess of current carrying costs. Marketplace changes resulting from the introduction of new products or surgical procedures may cause some of the Group’s products to become obsolete. The Group makes estimates regarding the future recoverability of the costs of these products and records a provision for excess and obsolete inventories based on historical experience, expiration of sterilisation dates and expected future trends. If actual product life cycles, product demand or acceptance of new product introductions are less favourable than projected by management, additional inventory write-downs may be required.
Dependence on government and other funding
In most markets throughout the world, expenditure on medical devices is ultimately controlled to a large extent by governments. Funds may be made available or withdrawn from healthcare budgets depending on government policy. The Group is therefore largely dependent on future governments providing increased funds commensurate with the increased demand arising from demographic trends.
Pricing of the Group’s products is largely governed in most markets by governmental reimbursement authorities. Initiatives sponsored by government agencies, legislative bodies and the private sector to limit the growth of healthcare costs, including price regulation, excise taxes and competitive pricing, are ongoing in markets where the Group has operations. This control may be exercised by determining prices for an individual product or for an entire procedure. The Group is exposed to government policies favouring locally sourced products. The Group is also exposed to changes in reimbursement policy, tax policy and pricing which may have an adverse impact on revenue and operating profit. Provisions in US healthcare legislation which previously imposed significant taxes on medical device manufacturers have been suspended since 2016 but may be reinstated. There may be an increased risk of adverse changes to government funding policies arising from deterioration in macro-economic conditions from time to time in the Group’s markets.
The Group must adhere to the rules laid down by government agencies that fund or regulate healthcare, including extensive and complex rules in the US. Failure to do so could result in fines or loss of future funding.
|
|
Smith & Nephew Annual Report 2018 |
|
189 |
World economic conditions
Demand for the Group’s products is driven by demographic trends, including the ageing population and the incidence of osteoporosis and obesity. Supply of, use of and payment for the Group’s products are also influenced by world economic conditions which could place increased pressure on demand and pricing, adversely impacting the Group’s ability to deliver revenue and margin growth. The conditions could favour larger, better capitalised groups, with higher market shares and margins. As a consequence, the Group’s prosperity is linked to general economic conditions and there is a risk of deterioration of the Group’s performance and finances during adverse macro-economic conditions.
During 2018, economic conditions worldwide continued to create several challenges for the Group, including the US Administration’s changed approach to trade policy, deferrals of procedures, heightened pricing pressure, significant declines in capital equipment expenditures at hospitals and increased uncertainty over the collectability of government debt, particularly those in the Emerging Markets. These factors tempered the overall growth of the Group’s global markets and could have an increased impact on growth in the future.
Political uncertainties
The Group operates on a worldwide basis and has distribution channels, purchasing agents and buying entities in over 100 countries. Political upheaval in some of those countries or in surrounding regions may impact the Group’s results of operations. Political changes in a country could prevent the Group from receiving remittances of profit from a member of the Group located in that country or from selling its products or investments in that country. Furthermore, changes in government policy regarding preference for local suppliers, import quotas, taxation or other matters could adversely affect the Group’s revenue and operating profit. War, economic sanctions, terrorist activities or other conflict could also adversely impact the Group. These risks may be greater in Emerging Markets, which account for an increasing portion of the Group’s business.
There remain heightened levels of political and regulatory uncertainty in the UK following the result of the referendum in June 2016 to leave the European Union, the triggering of Article 50 in March 2017 and the general election in June 2017. As of the date of this report, there remains uncertainty as to the UK’s future relationship with the EU. This may adversely impact trading performance across the sector. Regulatory uncertainty forms the most significant risk presently; the ability for us to continue to manufacture and register our products in a compliant manner for global distribution is key. Smith & Nephew has taken steps to prepare for the various Brexit scenarios, including moving certain of its product certifications from UK-based notified bodies to notified bodies based in the EU. The UK accounts for approximately 5% of global Group revenue and the majority of our manufacturing takes place outside the UK and EU. There is also uncertainty around US-China trade relations, which has resulted in tariffs on some medical devices being exported between the two countries.
Currency fluctuations
Smith & Nephew’s results of operations are affected by transactional exchange rate movements in that they are subject to exposures arising from revenue in a currency different from the related costs and expenses. The Group’s manufacturing cost base is situated principally in the US, the UK, China, Costa Rica and Switzerland, from which finished products are exported to the Group’s selling operations worldwide. Thus, the Group is exposed to fluctuations in exchange rates between the US Dollar, Sterling and Swiss Franc and the currency of the Group’s selling operations, particularly the Euro, Australian Dollar and Japanese Yen. If the US Dollar, Sterling or Swiss Franc should strengthen against the Euro, Australian Dollar and the Japanese Yen, the Group’s trading margin could be adversely affected. The Group manages the impact of exchange rate movements on revenue and cost of goods sold by a policy of transacting forward foreign currency commitments when firm purchase orders are placed. In addition, the Group’s policy is for forecast transactions to be covered between 50% and 90% for up to one year. However, the Group is exposed to medium to long-term adverse movements in the strength of currencies compared to the US Dollar. The Group uses the US Dollar as its reporting currency. The US Dollar is the functional currency of Smith & Nephew plc. The Group’s revenues, profits and earnings are also affected by exchange rate movements on the translation of results of operations in foreign subsidiaries for financial reporting purposes. See ‘Liquidity and capital resources’ on page 39.
Manufacturing and supply
The Group’s manufacturing production is concentrated at main facilities in Memphis, Mansfield and Oklahoma City in the US, Hull and Warwick in the UK, Aarau in Switzerland, Tuttlingen in Germany, Devrukh in India, Suzhou and Beijing in China, Alajuela in Costa Rica, Puschino in Russia and Curaçao, in Dutch Caribbean. If major physical disruption took place at any of these sites, it could adversely affect the results of operations. Physical loss and consequential loss insurance is carried to cover such risks but is subject to limits and deductibles and may not be sufficient to cover catastrophic loss. Management of orthopaedic inventory is complex, particularly forecasting and production planning. There is a risk that failures in operational execution could lead to excess inventory or individual product shortages.
The Group is reliant on certain key suppliers of raw materials, components, finished products and packaging materials or in some cases on a single supplier. These suppliers must provide the materials and perform the activities to the Group’s standard of quality requirements. A supplier’s failure to meet expected quality standards could create liability for the Group and adversely affect sales of the Group’s related products. The Group may be forced to pay higher prices to obtain raw materials, which it may not be able to pass on to its customers in the form of increased prices for its finished products. In addition, some of the raw materials used may become unavailable, and there can be no assurance that the Group will be able to obtain suitable and cost effective substitutes. Any interruption of supply caused by these or other factors could negatively impact Smith & Nephew’s revenue and operating profit.
|
190 |
Other information |
Smith & Nephew Annual Report 2018 |
|
GROUP INFORMATION continued
The Group will, from time to time including as part of the APEX programme, outsource or insource the manufacture of components and finished products to third parties and will periodically relocate the manufacture of product and/or processes between existing and/or new facilities. While these are planned activities, with these transfers there is a risk of disruption to supply.
Requirements of global regulatory agencies have become more stringent in recent years and we expect them to continue to do so. The Group’s Quality and Regulatory Affairs team is leading a major Group-wide programme to prepare for implementation of the EU Medical Devices Regulation (MDR), which came into force in May 2017, with a three-year transition period until May 2020. The regulation includes new requirements for the manufacture, supply and sale of all CE marked products sold in Europe and requires the reregistration of all medical devices, regardless of where they are manufactured.
Attracting and retaining key personnel
The Group’s continued development depends on its ability to hire and retain highly-skilled personnel with particular expertise. This is critical, particularly in general management, research, new product development and in the sales forces. If Smith & Nephew is unable to retain key personnel in general management, research and new product development or if its largest sales forces suffer disruption or upheaval, its revenue and operating profit would be adversely affected. Additionally, if the Group is unable to recruit, hire, develop and retain a talented, competitive workforce, it may not be able to meet its strategic business objectives.
Proprietary rights and patents
Due to the technological nature of medical devices and the Group’s emphasis on serving its customers with innovative products, the Group has been subject to patent infringement claims and is subject to the potential for additional claims. Claims asserted by third parties regarding infringement of their intellectual property rights, if successful, could require the Group to expend time and significant resources to pay damages, develop non-infringing products or obtain licences to the products which are the subject of such litigation, thereby affecting the Group’s growth and profitability. Smith & Nephew attempts to protect its intellectual property and regularly opposes third party patents and trademarks where appropriate in those areas that might conflict with the Group’s business interests. If Smith & Nephew fails to protect and enforce its intellectual property rights successfully, its competitive position could suffer, which could harm its results of operations.
Product liability claims and loss of reputation
The development, manufacture and sale of medical devices entail risk of product liability claims or recalls. Design and manufacturing defects with respect to products sold by the Group or by companies it has acquired could damage, or impair the repair of, body functions. The Group may become subject to liability, which could be substantial, because of actual or alleged defects in its products. In addition, product defects could lead to the need to recall from the market existing products, which may be costly and harmful to the Group’s reputation. There can be no assurance that customers, particularly in the US, the Group’s largest geographical market, will not bring product liability or related claims that would have a material adverse effect on the Group’s financial position or results of operations in the future, or that the Group will be able to resolve such claims within insurance limits. As at 31 December 2018, a provision of $192m is recognised relating to the present value of the estimated costs to resolve all unsettled known and unknown anticipated metal-on-metal hip implant claims globally.
Regulatory standards and compliance in the healthcare industry
Business practices in the healthcare industry are subject to regulation and review by various government authorities. In general, the trend in many countries in which the Group does business is towards higher expectations and increased enforcement activity by governmental authorities. While the Group is committed to doing business with integrity and welcomes the trend to higher standards in the healthcare industry, the Group and other companies in the industry have been subject to investigations and other enforcement activity that have incurred and may continue to incur significant expense. Under certain circumstances, if the Group were found to have violated the law, its ability to sell its products to certain customers could be restricted.
International regulation
The Group operates across the world and is subject to extensive legislation, including anti-bribery and corruption and data protection, in each country in which the Group operates. Our international operations are governed by the UK Bribery Act and the US Foreign Corrupt Practices Act which prohibit us or our representatives from making or offering improper payments to government officials and other persons or accepting payments for the purpose of obtaining or maintaining business. Our international operations in the Emerging Markets which operate through distributors increase our Group exposure to these risks.
The Group is also required to comply with the requirements of the EU General Data Protection Regulation (GDPR), which imposes additional obligations on companies regarding the handling of personal data and provides certain individual privacy rights to persons whose data is stored and became effective on 25 May 2018. As privacy and data protection have become more sensitive issues for regulators and consumers, new privacy and data protection laws, such as the GDPR, continue to develop in ways we cannot predict. Ensuring compliance with evolving privacy and data protection laws and regulations on a global basis may require us to change or develop our current business models and practices and may
|
|
Smith & Nephew Annual Report 2018 |
|
191 |
increase our cost of doing business. Despite those efforts, there is a risk that we may be subject to fines and penalties, litigation, and reputational harm in connection with our European activities as enforcement of such legislation has increased in recent years on companies and individuals where breaches are found to have occurred. Failure to comply with the requirements of privacy and data protection laws, including GDPR, could adversely affect our business, financial condition or results of operations.
Regulatory approval
The international medical device industry is highly regulated. Regulatory requirements are a major factor in determining whether substances and materials can be developed into marketable products and the amount of time and expense that should be allotted to such development.
National regulatory authorities administer and enforce a complex series of laws and regulations that govern the design, development, approval, manufacture, labelling, marketing and sale of healthcare products. They also review data supporting the safety and efficacy of such products. Of particular importance is the requirement in many countries that products be authorised or registered prior to manufacture, marketing or sale and that such authorisation or registration be subsequently maintained. The major regulatory agencies for Smith & Nephew’s products include the Food and Drug Administration (FDA) in the US, the Medicines and Healthcare products Regulatory Agency in the UK, the Ministry of Health, Labour and Welfare in Japan, the China Food and Drug Administration and the Australian Therapeutic Goods Administration. At any time, the Group is awaiting a number of regulatory approvals which, if not received, could adversely affect results of operations. In 2017, the EU reached agreement on a new set of Medical Device Regulations which entered into force on 25 May 2017. These have a three-year transition period and therefore, will fully apply in EU Member States from 26 May 2020.
The trend is towards more stringent regulation and higher standards of technical appraisal. Such controls have become increasingly demanding to comply with and management believes that this trend will continue. Regulatory requirements may also entail inspections for compliance with appropriate standards, including those relating to Quality Management Systems or Good Manufacturing Practices regulations. All manufacturing and other significant facilities within the Group are subject to regular internal and external audit for compliance with national medical device regulation and Group policies. Payment for medical devices may be governed by reimbursement tariff agencies in a number of countries. Reimbursement rates may be set in response to perceived economic value of the devices, based on clinical and other data relating to cost, patient outcomes and comparative effectiveness. They may also be affected by overall government budgetary considerations. The Group believes that its emphasis on innovative products and services should contribute to success in this environment. Failure to comply with these regulatory requirements could have a number of adverse consequences, including withdrawal of approval to sell a product in a country, temporary closure of a manufacturing facility, fines and potential damage to Company reputation.
Failure to make successful acquisitions
A key element of the Group’s strategy for continued growth is to make acquisitions or alliances to complement its existing business. Failure to identify appropriate acquisition targets or failure to conduct adequate due diligence or to integrate them successfully would have an adverse impact on the Group’s competitive position and profitability. This could result from the diversion of management resources towards the acquisition or integration process, challenges of integrating organisations of different geographic, cultural and ethical backgrounds, as well as the prospect of taking on unexpected or unknown liabilities. In addition, the availability of global capital may make financing less attainable or more expensive and could result in the Group failing in its strategic aim of growth by acquisition or alliance.
Relationships with healthcare professionals
The Group seeks to maintain effective and ethical working relationships with physicians and medical personnel who assist in the research and development of new products or improvements to our existing product range or in product training and medical education. If we are unable to maintain these relationships our ability to meet the demands of our customers could be diminished and our revenue and profit could be materially adversely affected.
Reliance on sophisticated information technology
The Group uses a wide variety of information systems, programmes and technology to manage our business. The Group also develops and sells certain products that are or will be connected to networks and/or the internet. Our systems are vulnerable to a cyber attack, malicious intrusion, loss of data privacy or any other significant disruption. Our systems have been and will continue to be the target of such threats. We have systems in place to minimise the risk and disruption of these intrusions and to monitor our systems on an ongoing basis for current or potential threats. There can be no assurance that these measures will prove effective in protecting Smith & Nephew from future interruptions and as a result the performance of the Group could be materially adversely affected.
Other risk factors
Smith & Nephew is subject to a number of other risks, which are common to most global medical technology groups and are reviewed as part of the Group’s Risk Management process.
|
192 |
Other information |
Smith & Nephew Annual Report 2018 |
|
GROUP INFORMATION continued
FACTORS AFFECTING SMITH & NEPHEW’S RESULTS OF OPERATIONS
Government economic, fiscal, monetary and political policies are all factors that materially affect the Group’s operation or investments of shareholders. Other factors include sales trends, currency fluctuations and innovation. Each of these factors is discussed further in the ‘Our Markets’ on pages 10–11, ‘Financial review’ on pages 38–39 and ‘Taxation information for shareholders’ on pages 206–207.
|
|
|
2018 |
|
2017 |
|
2016 |
|
2015 |
|
2014 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Income statement |
|
|
|
|
|
|
|
|
|
|
|
|
Revenue |
|
4,904 |
|
4,765 |
|
4,669 |
|
4,634 |
|
4,617 |
|
|
Cost of goods sold |
|
(1,298) |
|
(1,248) |
|
(1,272) |
|
(1,143) |
|
(1,162) |
|
|
Gross profit |
|
3,606 |
|
3,517 |
|
3,397 |
|
3,491 |
|
3,455 |
|
|
Selling, general and administrative expenses |
|
(2,497) |
|
(2,360) |
|
(2,366) |
|
(2,641) |
|
(2,471) |
|
|
Research and development expenses |
|
(246) |
|
(223) |
|
(230) |
|
(222) |
|
(235) |
|
|
Operating profit1 |
|
863 |
|
934 |
|
801 |
|
628 |
|
749 |
|
|
Net interest payable |
|
(51) |
|
(51) |
|
(46) |
|
(38) |
|
(22) |
|
|
Other finance costs |
|
(20) |
|
(10) |
|
(16) |
|
(15) |
|
(11) |
|
|
Share of results of associates |
|
(11) |
|
6 |
|
(3) |
|
(16) |
|
(2) |
|
|
Profit on disposal of business |
|
– |
|
– |
|
326 |
|
– |
|
– |
|
|
Profit before taxation |
|
781 |
|
879 |
|
1,062 |
|
559 |
|
714 |
|
|
Taxation |
|
(118) |
|
(112) |
|
(278) |
|
(149) |
|
(213) |
|
|
Attributable profit for the year |
|
663 |
|
767 |
|
784 |
|
410 |
|
501 |
|
|
Earnings per ordinary share |
|
|
|
|
|
|
|
|
|
|
|
|
Basic earnings per share |
|
76.0¢ |
|
87.8¢ |
|
88.1¢ |
|
45.9¢ |
|
56.1¢ |
|
|
Diluted earnings per share |
|
75.7¢ |
|
87.7¢ |
|
87.8¢ |
|
45.6¢ |
|
55.7¢ |
|
|
Average number of shares used in basic earnings per share (millions) |
|
873 |
|
874 |
|
890 |
|
894 |
|
893 |
|
|
Average number of shares used in diluted earnings per share (millions) |
|
876 |
|
875 |
|
893 |
|
899 |
|
899 |
|
|
Adjusted attributable profit2 |
|
|
|
|
|
|
|
|
|
|
|
|
Attributable profit for the year |
|
663 |
|
767 |
|
784 |
|
410 |
|
501 |
|
|
Acquisition and disposal related items |
|
(7) |
|
(10) |
|
9 |
|
25 |
|
125 |
|
|
Restructuring and rationalisation costs |
|
120 |
|
– |
|
62 |
|
65 |
|
61 |
|
|
Legal and other |
|
38 |
|
(13) |
|
(20) |
|
187 |
|
(2) |
|
|
Amortisation and impairment of acquisition intangibles |
|
118 |
|
140 |
|
178 |
|
204 |
|
129 |
|
|
Profit on disposal of business |
|
– |
|
– |
|
(326) |
|
– |
|
– |
|
|
US tax reform |
|
– |
|
(32) |
|
– |
|
– |
|
– |
|
|
Taxation on excluded items |
|
(51) |
|
(26) |
|
48 |
|
(130) |
|
(71) |
|
|
Adjusted attributable profit |
|
881 |
|
826 |
|
735 |
|
761 |
|
743 |
|
|
Adjusted earnings per ordinary share (EPSA)3 |
|
100.9¢ |
|
94.5¢ |
|
82.6¢ |
|
85.1¢ |
|
83.2¢ |
|
1 Reconciliation of operating to trading profit is presented below.
2 Non-IFRS financial measures are explained and reconciled to the most directly comparable financial measure prepared in accordance with IFRS on pages 194-198.
3 Adjusted earnings per ordinary share is calculated by dividing adjusted attributable profit by the basic weighted number of shares.
Reconciliation of operating profit to trading profit
|
|
|
2018 |
|
2017 |
|
2016 |
|
2015 |
|
2014 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Operating profit |
|
863 |
|
934 |
|
801 |
|
628 |
|
749 |
|
|
Acquisition and disposal related items |
|
(7) |
|
(10) |
|
9 |
|
12 |
|
118 |
|
|
Restructuring and rationalisation costs |
|
120 |
|
– |
|
62 |
|
65 |
|
61 |
|
|
Amortisation and impairment of acquisition intangibles |
|
113 |
|
140 |
|
178 |
|
204 |
|
129 |
|
|
Legal and other |
|
34 |
|
(16) |
|
(30) |
|
190 |
|
(2) |
|
|
Trading profit |
|
1,123 |
|
1,048 |
|
1,020 |
|
1,099 |
|
1,055 |
|
|
|
Smith & Nephew Annual Report 2018 |
|
193 |
|
|
|
2018 |
|
2017 |
|
2016 |
|
2015 |
|
2014 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Group balance sheet |
|
|
|
|
|
|
|
|
|
|
|
|
Non-current assets |
|
4,982 |
|
5,135 |
|
4,815 |
|
4,692 |
|
4,866 |
|
|
Current assets |
|
3,077 |
|
2,731 |
|
2,529 |
|
2,475 |
|
2,440 |
|
|
Total assets |
|
8,059 |
|
7,866 |
|
7,344 |
|
7,167 |
|
7,306 |
|
|
Share capital |
|
177 |
|
178 |
|
180 |
|
183 |
|
184 |
|
|
Share premium |
|
608 |
|
605 |
|
600 |
|
590 |
|
574 |
|
|
Capital redemption reserve |
|
18 |
|
17 |
|
15 |
|
12 |
|
11 |
|
|
Treasury shares |
|
(214) |
|
(257) |
|
(432) |
|
(294) |
|
(315) |
|
|
Retained earnings and other reserves |
|
4,285 |
|
4,101 |
|
3,595 |
|
3,475 |
|
3,586 |
|
|
Total equity |
|
4,874 |
|
4,644 |
|
3,958 |
|
3,966 |
|
4,040 |
|
|
Non-current liabilities |
|
1,720 |
|
1,876 |
|
2,038 |
|
1,857 |
|
2,104 |
|
|
Current liabilities |
|
1,465 |
|
1,346 |
|
1,348 |
|
1,344 |
|
1,162 |
|
|
Total liabilities |
|
3,185 |
|
3,222 |
|
3,386 |
|
3,201 |
|
3,266 |
|
|
Total equity and liabilities |
|
8,059 |
|
7,866 |
|
7,344 |
|
7,167 |
|
7,306 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Group cash flow statement |
|
|
|
|
|
|
|
|
|
|
|
|
Cash generated from operations |
|
1,108 |
|
1,273 |
|
1,035 |
|
1,203 |
|
961 |
|
|
Net interest paid |
|
(52) |
|
(48) |
|
(45) |
|
(36) |
|
(33) |
|
|
Income taxes paid |
|
(125) |
|
(135) |
|
(141) |
|
(137) |
|
(245) |
|
|
Net cash inflow from operating activities |
|
931 |
|
1,090 |
|
849 |
|
1,030 |
|
683 |
|
|
Capital expenditure (including trade investments and net of disposals of property, plant and equipment) |
|
(351) |
|
(384) |
|
(394) |
|
(360) |
|
(379) |
|
|
Acquisitions and disposals |
|
(29) |
|
(159) |
|
(214) |
|
(44) |
|
(1,552) |
|
|
Proceeds on disposal of business (net of tax) |
|
– |
|
– |
|
225 |
|
– |
|
– |
|
|
Distrubution from/(investment in) associate |
|
2 |
|
– |
|
– |
|
(25) |
|
(2) |
|
|
Proceeds from associate loan redemption |
|
– |
|
– |
|
– |
|
– |
|
188 |
|
|
Proceeds from own shares |
|
10 |
|
5 |
|
6 |
|
5 |
|
4 |
|
|
Equity dividends paid |
|
(321) |
|
(269) |
|
(279) |
|
(272) |
|
(250) |
|
|
Issue of ordinary capital and treasury shares purchased |
|
(45) |
|
(47) |
|
(358) |
|
(61) |
|
(35) |
|
|
Net cash flow from operating, investing and financing activities |
|
197 |
|
236 |
|
(165) |
|
273 |
|
(1,343) |
|
|
Termination of finance lease |
|
– |
|
5 |
|
– |
|
– |
|
– |
|
|
Exchange adjustments |
|
(20) |
|
28 |
|
(24) |
|
(21) |
|
(17) |
|
|
Opening net debt |
|
(1,281) |
|
(1,550) |
|
(1,361) |
|
(1,613) |
|
(253) |
|
|
Closing net debt |
|
(1,104) |
|
(1,281) |
|
(1,550) |
|
(1,361) |
|
(1,613) |
|
|
Selected financial ratios |
|
|
|
|
|
|
|
|
|
|
|
|
Gearing (closing net debt as a percentage of total equity) |
|
22.7% |
|
27.6% |
|
39% |
|
34% |
|
40% |
|
|
Dividends per ordinary share |
|
36.0¢1 |
|
35.0¢ |
|
30.8¢ |
|
30.8¢ |
|
29.6¢ |
|
|
Research and development costs to revenue |
|
5.0% |
|
4.7% |
|
4.9% |
|
4.8% |
|
5.1% |
|
|
Capital expenditure (including intangibles but excluding goodwill and trade investments) to revenue |
|
7.1% |
|
7.9% |
|
8.4% |
|
7.7% |
|
8.1% |
|
1 The Board has proposed a final dividend of 22.0 US cents per share which together with the first interim dividend of 14.0 US cents makes a total for 2018 of 36.0 US cents.
|
194 |
Other information |
Smith & Nephew Annual Report 2018 |
|
GROUP INFORMATION continued
NON-IFRS FINANCIAL INFORMATION – ADJUSTED MEASURES
These Financial Statements include financial measures that are not prepared in accordance with International Financial Reporting Standards (IFRS).These measures, which include trading profit, trading profit margin, tax rate on trading results, EPSA, ROIC, trading cash flow, free cash flow, trading profit to trading cash conversion ratio, net debt to adjusted EBITDA, and underlying growth, exclude the effect of certain cash and non-cash items that Group management believes are not related to the underlying performance of the Group. These non-IFRS financial measures are also used by management to make operating decisions because they facilitate internal comparisons of performance to historical results.
Non-IFRS financial measures are presented in these Financial Statements as the Group’s management believe that they provide investors with a means of evaluating performance of the business segment and the consolidated Group on a consistent basis, similar to the way in which the Group’s management evaluates performance, that is not otherwise apparent on an IFRS basis, given that certain non-recurring, infrequent, non-cash and other items that management does not otherwise believe are indicative of the underlying performance of the consolidated Group may not be excluded when preparing financial measures under IFRS. These non-IFRS measures should not be considered in isolation from, as substitutes for, or superior to financial measures prepared in accordance with IFRS.
Underlying revenue growth
‘Underlying growth in revenue’ is used to compare the revenue in a given year to the previous year on a like-for-like basis. This is achieved by adjusting for the impact of sales of products acquired in material business combinations or disposed of and for movements in exchange rates.
Underlying growth in revenue is considered by the Group to be an important measure of performance in terms of local functional currency since it excludes those items considered to be outside the influence of local management. The Group’s management uses this non-IFRS measure in its internal financial reporting, budgeting and planning to assess performance on both a business and a consolidated Group basis. Revenue growth at constant currency is important in measuring business performance compared to competitors and compared to the growth of the market itself.
The Group considers that revenue from sales of products acquired in material business combinations results in a step-up in growth in revenue in the year of acquisition that cannot be wholly attributed to local management’s efforts with respect to the business in the year of acquisition. Depending on the timing of the acquisition, there will usually be a further step change in the following year. A measure of growth excluding the effects of business combinations also allows senior management to evaluate the performance and relative impact of growth from the existing business and growth from acquisitions. The process of making business acquisitions is directed, approved and funded from the Group corporate centre in line with strategic objectives.
The material limitation of the underlying growth in revenue measure is that it excludes certain factors, described above, which ultimately have a significant impact on total revenues. The Group compensates for this limitation by taking into account relative movements in exchange rates in its investment, strategic planning and resource allocation. In addition, as the evaluation and assessment of business acquisitions is not within the control of local management, performance of acquisitions is monitored centrally until the business is integrated.
The Group’s management considers that the non-IFRS measure of underlying growth in revenue and the IFRS measure of growth in revenue are complementary measures, neither of which management uses exclusively.
‘Underlying growth in revenue’ reconciles to growth in revenue reported, the most directly comparable financial measure calculated in accordance with IFRS by making two adjustments, the ‘constant currency exchange effect’ and the ‘acquisitions and disposals effect’, described below.
The ‘constant currency exchange effect’ is a measure of the increase/decrease in revenue resulting from currency movements on non-US Dollar sales and is measured as the difference between: 1) the increase/decrease in the current year revenue translated into US Dollars at the current year average exchange rate and the prior revenue translated at the prior year rate; and 2) the increase/decrease being measured by translating current and prior year revenues into US Dollars using the prior year closing rate.
The ‘acquisitions and disposals effect’ is the measure of the impact on revenue from newly acquired material business combinations and recent material business disposals. This is calculated by comparing the current year, constant currency actual revenue (which include acquisitions and exclude disposals from the relevant date of completion) with prior year, constant currency actual revenue, adjusted to include the results of acquisitions and exclude disposals for the commensurate period in the prior year. These sales are separately tracked in the Group’s internal reporting systems and are readily identifiable.
|
|
Smith & Nephew Annual Report 2018 |
|
195 |
Reported revenue growth, the most directly comparable financial measure calculated in accordance with IFRS, reconciles to underlying growth in revenue as follows:
|
|
|
|
|
|
|
Reconciling items |
|
||
|
2018 |
|
Reported growth |
|
Underlying growth |
|
Acquisitions/disposals |
|
Currency impact |
|
|
Consolidated revenue by franchise |
|
% |
|
% |
|
% |
|
% |
|
|
Sports Medicine, Trauma & Other |
|
4 |
|
2 |
|
1 |
|
1 |
|
|
Sports Medicine Joint Repair |
|
11 |
|
8 |
|
2 |
|
1 |
|
|
Arthroscopic Enabling Technologies |
|
(2) |
|
(3) |
|
– |
|
1 |
|
|
Trauma & Extremities |
|
– |
|
(1) |
|
– |
|
1 |
|
|
Other Surgical Businesses |
|
10 |
|
10 |
|
– |
|
– |
|
|
Reconstruction |
|
3 |
|
3 |
|
– |
|
– |
|
|
Knee Implants |
|
3 |
|
3 |
|
– |
|
– |
|
|
Hip Implants |
|
2 |
|
2 |
|
– |
|
– |
|
|
Advanced Wound Management |
|
2 |
|
– |
|
– |
|
2 |
|
|
Advanced Wound Care |
|
3 |
|
1 |
|
– |
|
2 |
|
|
Advanced Wound Bioactives |
|
(6) |
|
(6) |
|
– |
|
– |
|
|
Advanced Wound Devices |
|
10 |
|
9 |
|
– |
|
1 |
|
|
Total |
|
3 |
|
2 |
|
– |
|
1 |
|
|
|
|
|
|
|
|
Reconciling items |
|
||
|
2017 |
|
Reported growth |
|
Underlying growth |
|
Acquisitions/disposals |
|
Currency impact |
|
|
Consolidated revenue by franchise |
|
% |
|
% |
|
% |
|
% |
|
|
Sports Medicine, Trauma & Other |
|
1 |
|
3 |
|
(2) |
|
– |
|
|
Sports Medicine Joint Repair |
|
7 |
|
6 |
|
– |
|
1 |
|
|
Arthroscopic Enabling Technologies |
|
(3) |
|
(3) |
|
– |
|
– |
|
|
Trauma & Extremities |
|
4 |
|
4 |
|
– |
|
– |
|
|
Other Surgical Businesses |
|
(11) |
|
7 |
|
(19) |
|
1 |
|
|
Reconstruction |
|
4 |
|
3 |
|
– |
|
1 |
|
|
Knee Implants |
|
6 |
|
5 |
|
– |
|
1 |
|
|
Hip Implants |
|
– |
|
– |
|
– |
|
– |
|
|
Advanced Wound Management |
|
2 |
|
2 |
|
– |
|
– |
|
|
Advanced Wound Care |
|
– |
|
– |
|
– |
|
– |
|
|
Advanced Wound Bioactives |
|
– |
|
– |
|
– |
|
– |
|
|
Advanced Wound Devices |
|
13 |
|
13 |
|
– |
|
– |
|
|
Total |
|
2 |
|
3 |
|
(1) |
|
– |
|
Trading profit, trading profit margin, trading cash flow and trading profit to cash conversion ratio
Trading profit, trading profit margin (trading profit expressed as a percentage of revenue), trading cash flow and trading profit to cash conversion ratio (trading cash flow expressed as a percentage of trading profit) are trend measures, which present the long-term profitability of the Group. The adjustments made exclude the impact of specific transactions that management considers affect the Group’s short-term profitability and cash flows. The Group has identified the following items, where material, as those to be excluded from operating profit and cash generated from operations when arriving at trading profit and trading cash flow, respectively: acquisition and disposal related items arising in connection with business combinations, including amortisation of acquisition intangible assets, impairments and integration costs; restructuring events; gains and losses resulting from legal disputes and uninsured losses. In addition to these items, gains and losses that materially impact the Group’s profitability or cash flows on a short-term or one-off basis are excluded from operating profit and cash generated from operations when arriving at trading profit and trading cash flow. The cash contribution to fund defined benefit pension schemes that are closed to future accrual are also excluded from cash generated from operations when arriving at trading cash flow.
|
196 |
Other information |
Smith & Nephew Annual Report 2018 |
|
GROUP INFORMATION continued
Adjusted earnings per ordinary share (EPSA)
EPSA is a trend measure, which presents the long-term profitability of the Group excluding the post-tax impact of specific transactions that management considers affect the Group’s short-term profitability and the one-off impact of US tax reform. The Group presents this measure to assist investors in their understanding of trends. Adjusted attributable profit is the numerator used for this measure and is determined by adjusting attributable profit for the items that are excluded from operating profit when arriving at trading profit and items that are recognised below operating profit that affect the Group’s short-term profitability. The most directly comparable financial measure calculated in accordance with IFRS is basic earnings per ordinary share (‘EPS’).
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash generated |
|
|
|
|
|
|
|
|
Operating |
|
Profit before |
|
|
|
Attributable |
|
from operating |
|
Earnings |
|
|
|
|
Revenue |
|
profit1 |
|
tax2 |
|
Taxation3 |
|
profit4 |
|
activities5 |
|
per share6 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
¢ |
|
|
2018 Reported |
|
4,904 |
|
863 |
|
781 |
|
(118) |
|
663 |
|
1,108 |
|
76.0 |
|
|
Acquisition and disposal related items |
|
– |
|
(7) |
|
(7) |
|
1 |
|
(6) |
|
3 |
|
(0.7) |
|
|
Restructuring and rationalisation costs |
|
– |
|
120 |
|
120 |
|
(24) |
|
96 |
|
83 |
|
11.0 |
|
|
Amortisation and impairment of acquisition intangibles |
|
– |
|
113 |
|
118 |
|
(27) |
|
91 |
|
– |
|
10.3 |
|
|
Legal and other7 |
|
– |
|
34 |
|
38 |
|
(1) |
|
37 |
|
104 |
|
4.3 |
|
|
Capital expenditure |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(347) |
|
– |
|
|
2018 Adjusted |
|
4,904 |
|
1,123 |
|
1,050 |
|
(169) |
|
881 |
|
951 |
|
100.9 |
|
Acquisition and disposal related items: For the year to 31 December 2018 the credit relates to a remeasurement of contingent consideration for a prior year acquisition and adjustments to provisions on disposal of a business, partially offset by costs associated with the acquisition of Rotation Medical, Inc.
Restructuring and rationalisation costs: For the year to 31 December 2018, these costs relate to the implementation of the Accelerating Performance and Execution (APEX) programme that was announced in February 2018.
Amortisation and impairment of acquisition intangibles: For the year to 31 December 2018 the charge relates to the amortisation of intangible assets acquired in material business combinations.
Legal and other: For the year ended 31 December 2018 charges primarily relate to legal expenses for ongoing metal-on-metal hip claims and an increase of $72m in the provision that reflects the present value of the estimated costs to resolve all other known and anticipated metal-on-metal hip claims globally. The year to 31 December 2018 also includes costs for implementing the requirements of the EU Medical Device Regulations that will apply from May 2020. These charges in the year to 31 December 2018 were partially offset by a credit of $84m relating to settlements with insurers related to product liability claims involving macrotextured components withdrawn from the market in 2003. $35m of cash funding to closed defined benefit pension schemes is excluded from trading cash flow.
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash generated |
|
|
|
|
|
|
|
|
Operating |
|
Profit before |
|
|
|
Attributable |
|
from operating |
|
Earnings |
|
|
|
|
Revenue |
|
profit1 |
|
tax2 |
|
Taxation3 |
|
profit4 |
|
activities5 |
|
per share6 |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
¢ |
|
|
2017 Reported |
|
4,765 |
|
934 |
|
879 |
|
(112) |
|
767 |
|
1,273 |
|
87.8 |
|
|
Acquisition and disposal related items |
|
– |
|
(10) |
|
(10) |
|
2 |
|
(8) |
|
3 |
|
(0.9) |
|
|
Restructuring and rationalisation costs |
|
– |
|
– |
|
– |
|
– |
|
– |
|
15 |
|
– |
|
|
Amortisation and impairment of acquisition intangibles |
|
– |
|
140 |
|
140 |
|
(40) |
|
100 |
|
– |
|
11.4 |
|
|
Legal and other7 |
|
– |
|
(16) |
|
(13) |
|
12 |
|
(1) |
|
25 |
|
(0.1) |
|
|
US tax reform |
|
– |
|
– |
|
– |
|
(32) |
|
(32) |
|
– |
|
(3.7) |
|
|
Capital expenditure |
|
– |
|
– |
|
– |
|
– |
|
– |
|
(376) |
|
– |
|
|
2017 Adjusted |
|
4,765 |
|
1,048 |
|
996 |
|
(170) |
|
826 |
|
940 |
|
94.5 |
|
|
|
Smith & Nephew Annual Report 2018 |
|
197 |
Acquisition and disposal related items: For the year to 31 December 2017 the credit relates to a remeasurement of contingent consideration for a prior year acquisition partially offset by costs associated with the acquisition of Rotation Medical, Inc.
Restructuring and rationalisation costs: There were no restructuring and rationalisation costs in the year to 31 December 2017. The restructuring and rationalisation cash flows relate to the implementation of the Group Optimisation plan that was announced in May 2014 and completed at the end of 2016.
Amortisation and impairment of acquisition intangibles: For the year to 31 December 2017 the charge relates to the amortisation of intangible assets acquired in material business combinations and an impairment charge of $10m.
Legal and other: For the year ended 31 December 2017 charges primarily relate to legal expenses for patent litigation with Arthrex and ongoing metal-on-metal hip claims and an increase of $10m in the provision that reflects the present value of the estimated costs to resolve all other known and anticipated metal-on-metal hip claims globally. These charges were offset by a $54m credit following a settlement payment received from Arthrex relating to patent litigation. $44m of cash funding to closed defined benefit pension schemes is excluded from trading cash flow.
1 Represents a reconciliation of operating profit to trading profit.
2Represents a reconciliation of reported profit before tax to trading profit before tax.
3Represents a reconciliation of reported tax to trading tax.
4Represents a reconciliation of reported attributable profit to adjusted attributable profit.
5 Represents a reconciliation of cash generated from operations to trading cash flow.
6 Represents a reconciliation of basic earnings per ordinary share to adjusted earnings per ordinary share (EPSA).
7From 1 January 2017, the ongoing funding of defined benefit pension schemes is not included in management’s definition of trading cash flow as there is no defined benefit service cost for these schemes.
Free Cash Flow
Free cash flow is a measure of the cash generated for the Group to use after capital expenditure according to its Capital Allocation Framework, it is defined as the net cash flow from operating activities less: capital expenditure and cash flows from interest and income taxes. A reconciliation from net cash flow from operating activities, the most comparable IFRS measure, to free cash flow is set out below:
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
$ million |
|
$ million |
|
$ million |
|
Net cash flow from operating activities |
|
1,108 |
|
1,273 |
|
1,035 |
|
Capital expenditure |
|
(347) |
|
(376) |
|
(392) |
|
Interest received |
|
2 |
|
2 |
|
3 |
|
Interest paid |
|
(54) |
|
(50) |
|
(48) |
|
Income taxes paid |
|
(125) |
|
(135) |
|
(141) |
|
Free cash flow |
|
584 |
|
714 |
|
457 |
Net debt to adjusted EBITDA ratio
Net debt to adjusted EBITDA ratio is used by the Group to measure leverage. Net debt is reconciled in Note 15 to the Group accounts. Adjusted EBITDA is defined as trading profit before depreciation of property, plant and equipment and amortisation of other intangible assets.
The calculation of net debt to adjusted EBITDA ratio is set out below:
|
|
|
2018 |
|
|
|
$ million |
|
Net debt |
|
1,104 |
|
|
|
|
|
Trading profit |
|
1,123 |
|
Depreciation of property, plant and equipment |
|
251 |
|
Amortisation of other intangible assets |
|
63 |
|
Adjusted EBITDA |
|
1,437 |
|
Net debt to adjusted EBITDA ratio (x) |
|
0.8 |
|
198 |
Other information |
Smith & Nephew Annual Report 2018 |
|
GROUP INFORMATION continued
Return on invested capital (ROIC)
Return on invested capital (ROIC) is a measure of the return generated on capital invested by the Group. It provides a metric for long-term value creation and encourages compounding reinvestment within the business and discipline around acquisitions with low returns and long payback.
ROIC is defined as: Net Operating Profit less Adjusted Taxes/((Opening Net Operating Assets + Closing Net Operating Assets)/2).
|
|
|
2018 |
|
2017 |
|
2016 |
|
|
|
$ million |
|
$ million |
|
$ million |
|
Operating profit |
|
863 |
|
934 |
|
801 |
|
Taxation |
|
(118) |
|
(112) |
|
(278) |
|
Taxation adjustment1 |
|
(14) |
|
(10) |
|
107 |
|
Net operating profit less adjusted taxes |
|
731 |
|
812 |
|
630 |
|
|
|
|
|
|
|
|
|
Total equity |
|
4,874 |
|
4,644 |
|
3,958 |
|
Retirement benefit asset |
|
(92) |
|
(62) |
|
– |
|
Investments |
|
(34) |
|
(21) |
|
(25) |
|
Investments in associates |
|
(105) |
|
(118) |
|
(112) |
|
Cash at Bank |
|
(365) |
|
(169) |
|
(100) |
|
Long term borrowings |
|
1,301 |
|
1,423 |
|
1,564 |
|
Retirement benefit obligation |
|
114 |
|
131 |
|
164 |
|
Bank overdrafts and loans |
|
164 |
|
27 |
|
86 |
|
Net operating assets |
|
5,857 |
|
5,855 |
|
5,535 |
|
Average net operating assets |
|
5,856 |
|
5,695 |
|
5,452 |
|
Return on invested capital |
|
12.5% |
|
14.3% |
|
11.5% |
1 Being the taxation on interest income, interest expense, other finance costs, share of results of associates and profit on disposal of business.
CONTRACTUAL OBLIGATIONS
Contractual obligations at 31 December 2018 were as follows:
|
|
|
Payments due by period |
|
||||||
|
|
|
Less than |
|
One to |
|
Three to |
|
More than |
|
|
|
|
one year |
|
three years |
|
five years |
|
five years |
|
|
|
|
$ million |
|
$ million |
|
$ million |
|
$ million |
|
|
Debt obligations |
|
39 |
|
– |
|
304 |
|
– |
|
|
Private placement notes |
|
164 |
|
332 |
|
274 |
|
522 |
|
|
Operating lease obligations |
|
54 |
|
72 |
|
40 |
|
52 |
|
|
Retirement benefit obligation |
|
25 |
|
50 |
|
– |
|
– |
|
|
Purchase obligations |
|
359 |
|
12 |
|
– |
|
– |
|
|
Capital expenditure |
|
16 |
|
– |
|
– |
|
– |
|
|
Other |
|
98 |
|
44 |
|
2 |
|
3 |
|
|
|
|
755 |
|
510 |
|
620 |
|
577 |
|
Other contractual obligations represent $20m of derivative contracts and $127m of acquisition consideration. Provisions that do not relate to contractual obligations are not included in the above table.
The Group has currently agreed to pay annual supplementary payments of $25m until 2021 for the UK defined benefit plan. These supplementary payments will be reviewed when the 30 September 2018 valuation has been completed.
There are a number of agreements that take effect, alter or terminate upon a change in control of the Company or the Group following a takeover, such as bank loan agreements and Company share plans. None of these are deemed to be significant in terms of their potential impact on the business of the Group as a whole. In addition, there are service contracts between the Company and its Executive Directors which provide for the automatic payment of a bonus following loss of office or employment occurring because of a successful takeover bid. Further details are set out on page 111.
The Company does not have contracts or other arrangements which individually are essential to the business.
|
|
Smith & Nephew Annual Report 2018 |
|
199 |
COMMENTARY ON THE INCOME STATEMENT FOR THE YEAR ENDED 31 DECEMBER 2017
|
- |
Group revenue increased by $96m from $4,669m to $4,765m in 2017. |
|
- |
Cost of goods sold decreased by $24m from $1,272m to $1,248m in 2017 primarily due to underlying efficiencies. |
|
- |
Selling, general and administrative expenses decreased by $6m from $2,366m to $2,360m. In 2017, administrative expenses included amortisation of software and other intangible assets of $62m (2016: $61m), nil restructuring and rationalisation expenses (2016: $62m), an amount of $140m relating to amortisation and impairment of acquired intangibles (2016: $178m), $10m credit for acquisition and disposal related items (2016: $9m charge) and $16m net credit for legal and other items (2016: $30m credit). Excluding the above items, selling, general and administrative expenses were $2,184m in 2017, an increase of $98m from $2,086m in 2016. |
|
- |
Research and development expenditure as a percentage of revenue remained broadly consistent at 4.7% in 2017 (2016: 4.9%). Expenditure was $223m in 2017 compared to $230m in 2016. |
|
- |
Operating profit increased by $133m from $801m to $934m in 2017. The year-on-year increase in operating profit primarily reflects a gain of $54m from the settlement of an intellectual property matter, no restructuring charges and lower amortisation and impairment of acquisition intangibles in 2017. |
|
- |
The taxation charge decreased by $166m from $278m to $112m in 2017. The reported tax rate was lower at 12.7% (2016: 26.2%) due to the $32m net benefit in 2017 from US tax reform and the tax impact of the Gynaecology disposal in 2016. |
COMMENTARY ON THE GROUP BALANCE SHEET AS AT 31 DECEMBER 2017
Non-current assets increased by $320m from $4,815m to $5,135m in 2017. This is principally attributable to the following:
|
- |
Property, plant and equipment increased by $67m from $982m to $1,049m in 2017. There were $308m of additions, $1m acquired with the Rotation Medical acquisition and favourable currency movements of $34m. This was partially offset by depreciation of $243m and $33m of assets disposed. |
|
- |
Goodwill increased by $183m from $2,188m to $2,371m in 2017 due to additions of $132m from the acquisition of Rotation Medical and favourable currency movements of $51m. Intangible assets decreased by $40m from $1,411m to $1,371m in 2017. Amortisation and impairment during 2017 was $202m and this was partially offset by net additions of $70m, $61m from the acquisition of Rotation Medical and favourable currency movements of $31m. |
|
- |
Retirement benefit assets of $62m (2016: nil) for UK and US retirement schemes due to changes in actuarial assumptions and contributions made during 2017. |
|
- |
Deferred tax assets increased by $30m in the year from $97m in 2016 to $127m in 2017 due to changes primarily in deferred tax on inventory provisions. |
Current assets increased by $202m from $2,529m to $2,731m in 2017. This is principally attributable to the following:
|
- |
Inventories rose by $60m to $1,304m in 2017 from $1,244m in 2016 primarily due to favourable currency movements of $62m. |
|
- |
The level of trade and other receivables increased by $73m to $1,258m in 2016 from $1,185m in 2016. The movement primarily relates to increased net trade receivables of $68m (including favourable currency movements of $44m). |
|
- |
Cash at bank has increased by $69m from $100m in 2016 to $169m in 2017 due to the timing of a receipt for the settlement of patent litigation in 2017. |
Current liabilities decreased by $2m from $1,348m in 2016 to $1,346m in 2017 primarily due to a $59m decrease in bank overdrafts to $27m and a $18m decrease in provisions to $129m. These movements were partially offset by a $73m increase in trade and other payables to $957m primarily due to $47m currency movements and $28m timing difference on the payment of expenses associated with a patent litigation gain.
Non-current liabilities decreased by $162m from $2,038m to $1,876m in 2017. This is principally attributable to the following:
|
- |
Long term borrowings decreased by $141m from $1,564m in 2016 to $1,423m in 2017 due to repayments made in 2017. |
|
- |
Retirement benefit obligations decreased $33m from $164m in 2016 to $131m in 2017 due to changes in actuarial assumptions. |
|
- |
Trade and other payables were $46m higher at $128m in 2017 from $82m in 2016 primarily due to movements in deferred and contingent consideration for acquisitions. |
|
- |
Provisions decreased by $37m from $134m in 2016 to $97m in 2017 primarily due to changes in classification between provisions due within one year. |
|
200 |
Other information |
Smith & Nephew Annual Report 2018 |
|
GROUP INFORMATION continued
|
Annual General Meeting |
11 April 2019 |
|
First quarter Trading Report |
2 May 2019 |
|
Payment of 2018 final dividend |
8 May 2019 |
|
Half year results announced |
31 July 20191 |
|
Third quarter Trading Report |
31 October 2019 |
|
Payment of 2019 interim dividend |
November 2019 |
|
Full year results announced |
February 20201 |
|
Annual Report available |
February/March 2020 |
|
Annual General Meeting |
April 2020 |
|
1 |
Dividend declaration dates. |
Annual General Meeting
The Company’s Annual General Meeting (‘AGM’) will be held on Thursday, 11 April 2019 at 2pm at No.11 Cavendish Square, London W1G 0AN. Registered shareholders have been sent either a Notice of Annual General Meeting or notification of availability of the Notice of Annual General Meeting.
Corporate headquarters and registered office
The corporate headquarters is in the UK and the registered office address is: Smith & Nephew plc, 15 Adam Street, London W2N 6LA, UK. Registered in England and Wales No. 324357. Tel. +44 (0)20 7401 7646 website: www.smith-nephew.com.
|
|
Smith & Nephew Annual Report 2018 |
|
201 |
Registrar
All general enquiries concerning shareholdings, dividends, changes to shareholders’ personal details and the AGM should be addressed to:
Computershare Investor Services plc, The Pavilions, Bridgwater Road, Bristol, BS99 6ZZ.
Tel: 0370 703 0047
Tel: +44 (0) 117 378 5450 from outside the UK
Website: www.investorcentre.co.uk
* Lines are open from 8:30am to 5:30pm Monday to Friday, excluding public holidays in England and Wales.
We make quarterly financial announcements, which are made available through Stock Exchange announcements and on the Group’s website (www.smith-nephew.com). Copies of recent Annual Reports, press releases, institutional presentations and audio webcasts are also available on the website.
We send paper copies of the Notice of Annual General Meeting and Annual Report only to those shareholders and ADS holders who have elected to receive shareholder documentation by post. Electronic copies of the Annual Report and Notice of Annual General Meeting are available on the Group’s website at www.smith-nephew.com. Both ordinary shareholders and ADS holders can request paper copies of the Annual Report, which the Company provides free of charge. The Company will continue to send to ordinary shareholders by post the Form of Proxy notifying them of the availability of the Annual Report and Notice of Annual General Meeting on the Group’s website. If you elect to receive the Annual Report and Notice of Annual General Meeting electronically you are informed by e-mail of the documents’ availability on the Group’s website. ADS holders receive the Form of Proxy by post, but will not receive a paper copy of the Notice of Annual General Meeting.
INVESTOR COMMUNICATIONS
The Company maintains regular dialogue with individual institutional shareholders, together with results presentations. To ensure that all members of the Board develop an understanding of the views of major investors, the Executive Directors review significant issues raised by investors with the Board. Non-Executive Directors are sent copies of analysts’ and brokers’ briefings. There is an opportunity for individual shareholders to question the Directors at the Annual General Meeting and the Company regularly responds to letters from shareholders on a range of issues.
UK CAPITAL GAINS TAX
For the purposes of UK capital gains tax, the price of the Company’s ordinary shares on 31 March 1982 was 35.04p.
SMITH & NEPHEW SHARE PRICE
The Company’s ordinary shares are quoted on the London Stock Exchange under the symbol SN. The Company’s share price is available on the Smith & Nephew website www.smith-nephew.com and at www.londonstockexhchange.com where the live financial data is updated with a 15-minute delay.
AMERICAN DEPOSITARY SHARES (‘ADSs’) AND AMERICAN DEPOSITARY RECEIPTS (‘ADRs’)
In the US, the Company’s ordinary shares are traded in the form of ADSs, evidenced by ADRs, on the New York Stock Exchange under the symbol SNN. Each American Depositary Share represents two ordinary shares. Deutsche Bank is the authorised depositary bank for the Company’s ADR programme. This relationship is governed under a Depositary Agreement that contains a clause which the Company has been advised by different Depositary Banks was previously considered as standard. This clause supports what we understand to be the normal practice under the US voting system, whereby votes which have not been instructed are then deemed to have given a discretionary proxy (‘auto-proxy’). Whilst the wording of our Depositary Agreement does grant auto-proxy rights, the Company can confirm that we do not believe that it is appropriate, within a UK context, to utilise this clause. The Company has therefore always instructed our Depositary Bank to not exercise this right and to only vote those ADRs which have been specifically instructed. The Company will look to remove this clause when updating the Depositary Agreement clause during the course of 2019.
It is also the Company’s practice to always notify our ADR holders of upcoming Annual General Meetings and General Meetings and the availability of relevant documentation on the Company website as well as providing instructions on how to submit votes, where applicable. This is in accordance with US regulations, which require NYSE ADR listed issuers to solicit ADR votes regardless of the wording in their Depositary Agreement.
|
202 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
ADS ENQUIRIES
All enquiries regarding ADS holder accounts and payment of dividends should be addressed to:
Deutsche Bank Shareholder Services
American Stock Transfer and Trust Company
Operations Centre 6201 15th Avenue
Brooklyn, New York
NY 11219
Tel: +1 866 249 2593 (toll free)
E-mail: db@astfinancial.com
Website: www.adr.db.com
The Deutsche Bank Global Direct Investor Services Program is available for US residents, enabling investment directly in ADSs with reduced brokerage commissions and service costs. For further information on Global Direct contact Deutsche Bank Shareholder Services (as above) or visit www.adr.db.com.
SMITH & NEPHEW ADS PRICE
The Company’s ADS price can be obtained from the official New York Stock Exchange website at www.nyse.com and the Smith & Nephew website www.smith-nephew.com where the live financial data is updated with a 15-minute delay, and is quoted daily in the Wall Street Journal.
ADS PAYMENT INFORMATION
The Company hereby discloses ADS payment information for the year ended 31 December 2018 in accordance with the Securities and Exchange Commission rules 12.D.3 and 12.D.4 relating to Form 20-F filings by foreign private issuers. The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors, including payment of dividends by the Company by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deductions from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may generally refuse to provide fee-attracting services until its fee for those services are paid.
|
Persons depositing or withdrawing shares must pay |
For |
|
|
$5.00 (or less) per 100 ADSs (or portion of 100 ADSs) $0.05 (or less) per ADS |
Issuance of ADSs, including issuances resulting from a distribution of shares or rights or other property Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates Any cash distribution to ADS registered holders, including payment of dividend |
|
|
$0.05 (or less) per ADS per calendar year Registration or transfer fees |
Depositary services Transfer and registration of shares on our share register to or from the name of the depositary or its agent when shares are deposited or withdrawn |
|
|
Taxes and other governmental charges the depositary or the custodian have to pay on any ADS or share underlying an ADS, for example, stock transfer taxes, stamp duty or withholding taxes |
As necessary |
|
|
Any charges incurred by the depositary or its agents for servicing the deposited securities |
As necessary |
|
During 2018, a fee of one US cent per ADS was collected on the 2017 final dividend paid in May and a fee of one US cent per ADS was collected on the 2018 interim dividend paid in October. In the period 1 January 2018 to 15 February 2019, the total program payments made by Deutsche Bank Trust Company Americas were $829,350.
|
|
Smith & Nephew Annual Report 2018 |
|
203 |
DIVIDEND HISTORY
Smith & Nephew has paid dividends on its ordinary shares in every year since 1937. Following the capital restructuring and dividend reduction in 2000, the Group adopted a policy of increasing its dividend cover (the ratio of EPSA, as set out in the ‘Selected financial data’, to ordinary dividends declared for the year). This was intended to increase the financing capability of the Group for acquisitions and other investments. From 2000 to 2004, the dividend increased in line with inflation and, in 2004, dividend cover stood at 4.1 times. Having achieved this level of dividend cover the Board changed its policy, from that of increasing dividends in line with inflation, to that of increasing dividends for 2005 and after by 10%. Following the redenomination of the Company’s share capital into US Dollars, the Board reaffirmed its policy of increasing the dividend by 10% a year in US Dollar terms. On 2 August 2012, the Board announced its intention to pursue a progressive dividend policy, with the aim of increasing the US Dollar value of ordinary dividends over time broadly based on the Group’s underlying growth in earnings, while taking into account capital requirements and cash flows.
At the time of the full year results, the Board reviews the appropriate level of total annual dividend each year. The Board intends that the interim dividend will be set by a formula and will be equivalent to 40% of the total dividend for the previous year. Dividends will continue to be declared in US Dollars with an equivalent amount in Sterling payable to those shareholders whose registered address is in the UK, or who have validly elected to receive Sterling dividends.
An interim dividend in respect of each fiscal year is normally declared in July or August and paid in October or November. A final dividend will be recommended by the Board of Directors and paid subject to approval by shareholders at the Company’s Annual General Meeting.
Future dividends of Smith & Nephew will be dependent upon: future earnings; the future financial condition of the Group; the Board’s dividend policy; and the additional factors that might affect the business of the Group set out in ‘Special note regarding forward-looking statements’ and ‘Risk Factors’.
DIVIDENDS PER SHARE
The table below sets out the dividends per ordinary share in the last five years.
|
|
|
Year ended 31 December |
|
||||||||
|
|
|
2018 |
|
2017 |
|
2016 |
|
2015 |
|
2014 |
|
|
Pence per share: |
|
|
|
|
|
|
|
|
|
|
|
|
Interim |
|
10.670 |
|
9.340 |
|
10.080 |
|
7.680 |
|
6.820 |
|
|
Final |
|
17.120 |
1 |
16.240 |
|
14.420 |
|
13.000 |
|
12.340 |
|
|
Total |
|
27.790 |
|
25.580 |
|
24.500 |
|
20.680 |
|
19.160 |
|
|
US cents per share: |
|
|
|
|
|
|
|
|
|
|
|
|
Interim |
|
14.000 |
|
12.300 |
|
12.300 |
|
11.800 |
|
11.000 |
|
|
Final |
|
22.000 |
|
22.700 |
|
18.500 |
|
19.000 |
|
18.600 |
|
|
Total |
|
36.000 |
|
35.000 |
|
30.800 |
|
30.800 |
|
29.600 |
|
1 Translated at the Bank of England rate on 15 February 2019.
From 6 April 2018 dividends below £2,000 per tax year became tax free and dividends above £2,000 per tax year became subject to personal income tax at the rate of 7.5% for basic rate taxpayers, 32.5% for higher rate taxpayers and 38.1% for additional rate taxpayers. If you need to pay tax, how you pay depends on the amount of dividend income you receive in a year. If your dividend income is up to £10,000 you can request HMRC to change your tax code so that the tax will be taken from your wages or pension or you can complete a self-assessment tax return. If your dividend income is over £10,000 in the tax year, you will need to complete a self-assessment tax return. This will apply to both cash and dividend reinvestment plan (‘DRiP’) dividends, although dividends paid on shares held within pensions and ISAs will be unaffected, remaining tax free.
Between 6 April 2016 and 6 April 2018 dividends below £5,000 per tax year were tax free and dividends above £5,000 per tax year were subject to personal income tax at the rates referred to above.
Dividends shown in the table above, prior to 6 April 2016, include the associated UK tax credit of 10%, but exclude the deduction of withholding taxes.
Since the second interim dividend for 2005, all dividends have been declared in US cents per ordinary share.
In respect of the proposed final dividend for the year ended 31 December 2018 of 22.0 US cents per ordinary share, the record date will be 5 April 2019 and the payment date will be 8 May 2019. The Sterling equivalent per ordinary share will be set following the record date. Shareholders may elect to receive their dividend in either Sterling or US Dollars and the last day for election will be 17 April 2019. The ordinary shares will trade ex-dividend on both the London and New York Stock Exchanges from 4 April 2019.
The proposed final dividend of 22.0 US cents per ordinary share, which together with the interim dividend of 14.0 US cents, makes a total for 2018 of 36.0 US cents.
|
204 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
SHARE CAPITAL
The principal trading market for the ordinary shares is the London Stock Exchange. The ordinary shares were listed on the New York Stock Exchange on 16 November 1999, trading in the form of ADSs evidenced by ADRs. Each ADS represents two ordinary shares from 14 October 2014, before which time one ADS represented five ordinary shares. The ADS facility is sponsored by Deutsche Bank acting as depositary.
All the ordinary shares, including those held by Directors and Executive Officers, rank pari passu with each other. On 23 January 2006, the ordinary shares of 122/9p were redenominated as ordinary shares of US 20 cents (following approval by shareholders at the Extraordinary General Meeting in December 2005). The new US Dollar ordinary shares carry the same rights as the previous ordinary shares. The share price continues to be quoted in Sterling. In 2006, the Company issued £50,000 of shares in Sterling in order to comply with English law. These were issued as deferred shares, which are not listed on any stock exchange. They have extremely limited rights and therefore effectively have no value. These shares are held by the Company Secretary, although the Board reserves the right to transfer them to a member of the Board should it so wish.
Shareholdings
As at 15 February 2019, to the knowledge of the Group, there were 14,301 registered holders of ordinary shares, of whom 95 had registered addresses in the US and held a total of 185,400 ordinary shares (0.02% of the total issued). Because certain ordinary shares are registered in the names of nominees, the number of shareholders with registered addresses in the USA is not representative of the number of beneficial owners of ordinary shares resident in the US.
As at 15 February 2019, 41,419,215 ADSs equivalent to 82,838,430 ordinary shares or approximately 9.5% of the total ordinary shares in issue, were outstanding and were held by 85 registered ADS holders.
Major shareholders
As far as is known to Smith & Nephew, the Group is not directly or indirectly owned or controlled by another corporation or by any Government and the Group has not entered into arrangements, the operation of which may at a subsequent date result in a change in control of the Group.
As at 15 February 2019, the Company is not aware of any person who has a significant direct or indirect holding of securities in the Company, as defined in the Disclosure and Transparency Rules (DTRs) of the Financial Conduct Authority (FCA), other than as shown below, and is not aware of any persons holding securities which may control the Company. There are no securities in issue which have special rights as to the control of the Company.
The following table shows the last notification(s) received by the Company, in accordance with the FCA’s DTRs relating to notifiable interests in the voting rights in the company’s issued share capital:
|
|
|
|
As at 31 December |
|
|||||
|
|
|
15 February 2019 |
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
%* |
|
%* |
|
%* |
|
%* |
|
|
BlackRock, Inc. |
|
5.2 |
|
5.2 |
|
5.2 |
|
5.2 |
|
|
|
|
|
As at 31 December |
|
|||||
|
|
|
15 February 2019 |
|
2018 |
|
2017 |
|
2016 |
|
|
|
|
‘000 |
|
‘000 |
|
‘000 |
|
‘000 |
|
|
BlackRock, Inc. |
|
46,427 |
|
46,427 |
|
46,427 |
|
46,427 |
|
* Percentage of ordinary shares in issue, excluding Treasury shares.
|
|
Smith & Nephew Annual Report 2018 |
|
205 |
Purchase of ordinary shares on behalf of the Company
At the AGM, the Company will be seeking a renewal of its current permission from shareholders to purchase up to 10% of its own shares. In order to avoid shareholder dilution, shares allotted to employees through employee share schemes are bought back on a quarterly basis and subsequently cancelled by the Company.
From 1 January 2018 to 15 February 2019, in the months listed below, the Company has purchased 3,644,693 ordinary shares at a cost of $65,825,922.
|
|
|
Total shares |
|
Average price |
|
Approximate US$ value |
|
|
|
|
purchased |
|
paid per share |
|
of shares purchased |
|
|
|
|
000's |
|
pence |
|
under the plan |
|
|
14-15 February 2018 (Q4 2017) |
|
557 |
|
1,261.6308 |
|
$ 9,892,872
|
|
|
16-23 May 2018 (Q1 2018) |
|
1,226 |
|
1,320.3255 |
|
$ 21,758,003
|
|
|
31 July-1 August 2018 (Q2 2018) |
|
216 |
|
1,324.2552 |
|
$ 3,772,227
|
|
|
19-22 November 2018 (Q3 2018) |
|
670 |
|
1,395.2269 |
|
$ 12,015,283
|
|
|
11-15 February 2019 (Q4 2018) |
|
976 |
|
1,465.1940 |
|
$ 18,387,536
|
|
The shares were purchased in the open market by J.P. Morgan Securities plc on behalf of the Company.
Exchange controls and other limitations affecting security holders
There are no UK governmental laws, decrees or regulations that restrict the export or import of capital or that affect the payment of dividends, interest or other payments to non-resident holders of Smith & Nephew’s securities, except for certain restrictions imposed from time to time by Her Majesty’s Treasury of the United Kingdom pursuant to legislation, such as the United Nations Act 1946 and the Emergency Laws Act 1964, against the Government or residents of certain countries.
There are no limitations, either under the laws of the UK or under the Articles of Association of Smith & Nephew, restricting the right of non-UK residents to hold or to exercise voting rights in respect of ordinary shares, except that where any overseas shareholder has not provided to the Company a UK address for the service of notices, the Company is under no obligation to send any notice or other document to an overseas address. It is, however, the current practice of the Company to send every notice or other document to all shareholders regardless of the country recorded in the register of members, with the exception of details of the Company’s dividend reinvestment plan, which are not sent to shareholders with recorded addresses in the US and Canada.
|
206 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
TAXATION INFORMATION FOR SHAREHOLDERS
The comments below are of a general and summary nature and are based on the Group’s understanding of certain aspects of current UK and US federal income tax law and practice relevant to the ADSs and ordinary shares not in ADS form. The comments address the material US and UK tax consequences generally applicable to a person who is the beneficial owner of ADSs or ordinary shares and who, for US federal income tax purposes, is a citizen or resident of the US, a corporation (or other entity taxable as a corporation) created or organised in or under the laws of the US (or any State therein or the District of Columbia), or an estate or trust the income of which is included in gross income for US federal income tax purposes regardless of its source (each a US Holder). The comments set out below do not purport to address all tax consequences of the ownership of ADSs or ordinary shares that may be material to a particular holder and in particular do not deal with the position of shareholders who directly, indirectly or constructively own 10% or more of the Company’s issued ordinary shares. This discussion does not apply to (i) persons whose holding of ADSs or ordinary shares is effectively connected with or pertains to either a permanent establishment in the UK through which a US Holder carries on a business in the UK or a fixed base from which a US Holder performs independent personal services in the UK, or (ii) persons whose registered address is inside the UK. This discussion does not apply to certain investors subject to special rules, such as certain financial institutions, tax-exempt entities, insurance companies, broker-dealers and traders in securities that elect to use the mark-to-market method of tax accounting, partnerships or other entities treated as partnerships for US federal income tax purposes, US Holders holding ADSs or ordinary shares as part of a hedging, conversion or other integrated transaction or US Holders whose functional currency for US federal income tax purposes is other than the US Dollar. In addition, the comments below do not address the potential application of the provisions of the United States Internal Revenue Code known as the Medicare Contribution Tax, any alternative minimum tax consequences, any US federal tax other than income tax or any US state, local or non-US (other than UK) taxes. The summary deals only with US Holders who hold ADSs or ordinary shares as capital assets for tax purposes. The summary is based on current UK and US law and practice which is subject to change, possibly with retroactive effect. US Holders are recommended to consult their own tax advisers as to the particular tax consequences to them of the ownership of ADSs or ordinary shares. The Company believes, and this discussion assumes, that the Company was not a passive foreign investment company for its taxable year ended 31 December 2018.
This discussion is based in part on representations by the depositary and assumes that each obligation under the deposit agreement and any related agreement will be performed in accordance with its terms. For purposes of US federal income tax law, US Holders of ADSs will generally be treated as owners of the ordinary shares represented by the ADSs. However, the US Treasury has expressed concerns that parties to whom depositary shares are released before shares are delivered to the depositary (pre-released) may be taking actions that are inconsistent with the claiming of foreign tax credits by owners of depositary shares. Such actions would also be inconsistent with the claiming of the reduced rate of tax, described below, applicable to dividends received by certain non-corporate US Holders. Accordingly, the availability of the reduced tax rate for dividends received by certain non-corporate US Holders of ADSs could be affected by actions that may be taken by parties to whom ADSs are pre-released.
Taxation of distributions in the UK and the US
The UK does not currently impose a withholding tax on dividends paid by a UK corporation, such as the Company.
Distributions paid by the Company will generally be taxed as foreign source dividends to the extent paid out of the Company’s current or accumulated earnings and profits as determined for US federal income tax purposes. Because the Company does not maintain calculations of its earnings and profits under US federal income tax principles, it is expected that distributions generally will be reported to US Holders as dividends. Such dividends will not be eligible for the dividends-received deduction generally allowed to corporate US Holders.
Dividends paid to certain non-corporate US Holders of ordinary shares or ADSs may be subject to US federal income tax at lower rates than those applicable to other types of ordinary income if certain conditions are met. Non-corporate US Holders should consult their own tax advisers to determine whether they are subject to any special rules that limit their ability to be taxed at these favourable rates.
Taxation of capital gains
US Holders, who are not resident or ordinarily resident for tax purposes in the UK, will not generally be liable for UK capital gains tax on any capital gain realised upon the sale or other disposition of ADSs or ordinary shares unless the ADSs or ordinary shares are held in connection with a trade carried on in the UK through a permanent establishment (or in the case of individuals, through a branch or agency). Furthermore, UK resident individuals who acquire ADSs or ordinary shares before becoming temporarily non-UK residents may remain subject to UK taxation of capital gains on gains realised while non-resident.
For US federal income tax purposes, gains or losses realised upon a taxable sale or other disposition of ADSs or ordinary shares by US Holders generally will be US source capital gains or losses and will be long-term capital gains or losses if the ADSs or ordinary shares were held for more than one year. The amount of a US Holder’s gain or loss will be equal to the difference between the amount realised on the sale or other disposition and such holder’s tax basis in the ADSs, or ordinary shares, each determined in US Dollars.
|
|
Smith & Nephew Annual Report 2018 |
|
207 |
Inheritance and estate taxes
HM Revenue & Customs imposes inheritance tax on capital transfers which occur on death, and in the seven years preceding death. HM Revenue & Customs considers that the US/UK Double Taxation Convention on Estate and Gift Tax applies to inheritance tax. Consequently, a US citizen who is domiciled in the USA and is not a UK national or domiciled in the UK will not be subject to UK inheritance tax in respect of ADSs and ordinary shares. A UK national who is domiciled in the US will be subject to UK inheritance tax but will be entitled to a credit for any US federal estate tax charged in respect of ADSs and ordinary shares in computing the liability to UK inheritance tax. Special rules apply where ADSs and ordinary shares are business property of a permanent establishment of an enterprise situated in the UK.
US information reporting and backup withholding
Payments of dividends on, or proceeds from the sale of, ADSs or ordinary shares that are made within the US or through certain US-related financial intermediaries generally will be subject to US information reporting, and may be subject to backup withholding, unless a US Holder is an exempt recipient or, in the case of backup withholding, provides a correct US taxpayer identification number and certain other conditions are met.
Any backup withholding deducted may be credited against the US Holder’s US federal income tax liability, and, where the backup withholding exceeds the actual liability, the US Holder may obtain a refund by timely filing the appropriate refund claim with the US Internal Revenue Service.
US Holders who are individuals or certain specified entities may be required to report information relating to securities issued by a non-US person (or foreign accounts through which the securities are held), subject to certain exceptions (including an exception for securities held in accounts maintained by US financial institutions). US Holders should consult their tax advisers regarding their reporting obligations with respect to the ADSs or ordinary shares.
UK stamp duty and stamp duty reserve tax
UK stamp duty is charged on documents and in particular instruments for the transfer of registered ownership of ordinary shares. Transfers of ordinary shares in certificated form will generally be subject to UK stamp duty at the rate of ½% of the consideration given for the transfer with the duty rounded up to the nearest £5.
UK stamp duty reserve tax (SDRT) arises when there is an agreement to transfer shares in UK companies ‘for consideration in money or money’s worth’, and so an agreement to transfer ordinary shares for money or other consideration may give rise to a charge to SDRT at the rate of ½% (rounded up to the nearest penny). The charge of SDRT will be cancelled, and any SDRT already paid will be refunded, if within six years of the agreement an instrument of transfer is produced to HM Revenue & Customs and the appropriate stamp duty paid.
Transfers of ordinary shares into CREST (an electronic transfer system) are exempt from stamp duty so long as the transferee is a member of CREST who will hold the ordinary shares as a nominee for the transferor and the transfer is in a form that will ensure that the securities become held in uncertificated form within CREST. Paperless transfers of ordinary shares within CREST for consideration in money or money’s worth are liable to SDRT rather than stamp duty. SDRT on relevant transactions will be collected by CREST at ½%, and this will apply whether or not the transfer is effected in the UK and whether or not the parties to it are resident or situated in the UK.
A charge of stamp duty or SDRT at the rate of 1.5% of the consideration (or, in some circumstances, the value of the shares concerned) will arise on a transfer or issue of ordinary shares to the depositary or to certain persons providing a clearance service (or their nominees or agents) for the conversion into ADRs and will generally be payable by the depositary or person providing clearance service. In accordance with the terms of the Deposit Agreement, any tax or duty payable by the depositary on deposits of ordinary shares will be charged by the depositary to the party to whom ADRs are delivered against such deposits.
No liability for stamp duty or SDRT will arise on any transfer of, or agreement to transfer, an ADS or beneficial ownership of an ADS, provided that the ADS and any instrument of transfer or written agreement to transfer remains at all times outside the UK, and provided further that any instrument of transfer or written agreement to transfer is not executed in the UK and the transfer does not relate to any matter or thing done or to be done in the UK (the location of the custodian as a holder of ordinary shares not being relevant in this context). In any other case, any transfer of, or agreement to transfer, an ADS or beneficial ownership of an ADS could, depending on all the circumstances of the transfer, give rise to a charge to stamp duty or SDRT.
|
208 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
ARTICLES OF ASSOCIATION
The following summarises certain material rights of holders of the Company’s ordinary shares under the material provisions of the Company’s Articles of Association and English law. This summary is qualified in its entirety by reference to the Companies Act and the Company’s Articles of Association. In the following description, a ‘shareholder’ is the person registered in the Company’s register of members as the holder of an ordinary share.
The Company is incorporated under the name Smith & Nephew plc and is registered in England and Wales with registered number 324357.
The Company’s ordinary shares may be held in certificated or uncertificated form. No holder of the Company’s shares will be required to make additional contributions of capital in respect of the Company’s shares in the future. In accordance with English law, the Company’s ordinary shares rank equally.
Directors
Under the Company’s Articles of Association, a Director may not vote in respect of any contract, arrangement, transaction or proposal in which he, or any person connected with him, has any material interest other than by virtue of his interests in securities of, or otherwise in or through, the Company. This is subject to certain exceptions relating to proposals (a) indemnifying him in respect of obligations incurred on behalf of the Company, (b) indemnifying a third party in respect of obligations of the Company for which the Director has assumed responsibility under an indemnity or guarantee, (c) relating to an offer of securities in which he will be interested as an underwriter, (d) concerning another body corporate in which the Director is beneficially interested in less than 1% of the issued shares of any class of shares of such a body corporate, (e) relating to an employee benefit in which the Director will share equally with other employees and (f) relating to any insurance that the Company is empowered to purchase for the benefit of Directors of the Company in respect of actions undertaken as Directors (and/or officers) of the Company.
A Director shall not vote or be counted in any quorum present at a meeting in relation to a resolution on which he is not entitled to vote.
The Directors are empowered to exercise all the powers of the Company to borrow money, subject to the limitation that the aggregate amount of all monies borrowed after deducting cash and current asset investments by the Company and its subsidiaries shall not exceed the sum of $6,500,000,000.
Any Director who has been appointed by the Directors since the previous Annual General Meeting of shareholders, either to fill a casual vacancy or as an additional Director holds office only until the conclusion of the next Annual General Meeting and then shall be eligible for re-election by the shareholders. The Company’s Articles of Association provide that Directors should regularly be submitted for re-election at intervals of three years, however in accordance with the UK Corporate Governance Code, all Directors are subject to annual re-election. If not re-appointed, a Director retiring at a meeting shall retain office until the meeting appoints someone in his place, or if it does not do so, until the conclusion of the meeting. The Directors are subject to removal with or without cause by the Board or the shareholders. Directors are not required to hold any shares of the Company by way of qualification.
Under the Company’s Articles of Association and English law, a Director may be indemnified out of the assets of the Company against liabilities he may sustain or incur in the execution of his duties.
Rights attaching to ordinary shares
Under English law, dividends are payable on the Company’s ordinary shares only out of profits available for distribution, as determined in accordance with accounting principles generally accepted in the UK and by the Companies Act 2006. Holders of the Company’s ordinary shares are entitled to receive final dividends as may be declared by the Directors and approved by the shareholders in a general meeting, rateable according to the amounts paid up on such shares, provided that the dividend cannot exceed the amount recommended by the Directors.
The Company’s Board of Directors may declare such interim dividends as appear to them to be justified by the Company’s financial position. If authorised by an ordinary resolution of the shareholders, the Board may also direct payment of a dividend in whole or in part by the distribution of specific assets (and in particular of paid up shares or debentures of the Company).
Any dividend unclaimed after 12 years from the date the dividend was declared, or became due for payment, will be forfeited and will revert to the Company.
There were no material modifications to the rights of shareholders under the Articles during 2018.
Voting rights of ordinary shares
The Company’s Articles of Association provide that voting at any general meeting of shareholders is by a show of hands unless a poll, which is a written vote, is duly demanded and held. On a show of hands, every shareholder who is present in person at a general meeting has one vote regardless of the number of shares held. On a poll, every shareholder who is present in person or by proxy has one vote for each ordinary share held by that shareholder. A poll may be demanded by any of the following:
– The chair of the meeting;
– At least five shareholders present or by proxy entitled to vote on the resolution;
– Any shareholder or shareholders representing in the aggregate not less than one-tenth of the total voting rights of all shareholders entitled to vote on the resolution; or
– Any shareholder or shareholders holding shares conferring a right to vote on the resolution on which there have been paid-up sums in aggregate equal to not less than one-tenth of the total sum paid up on all the shares conferring that right.
A Form of Proxy will be treated as giving the proxy the authority to demand a poll, or to join others in demanding one, as above.
It is the Company’s usual practice to vote by poll at Annual General Meetings.
The necessary quorum for a general meeting is two shareholders present in person or by proxy carrying the right to vote upon the business to be transacted.
|
|
Smith & Nephew Annual Report 2018 |
|
209 |
Matters are transacted at general meetings of the Company by the processing and passing of resolutions of which there are two kinds; ordinary or special resolutions:
– Ordinary resolutions include resolutions for the re-election of Directors, the approval of financial statements, the declaration of dividends (other than interim dividends), the appointment and re-appointment of auditors or the grant of authority to allot shares. An ordinary resolution requires the affirmative vote of a majority of the votes of those persons voting at the meetings at which there is a quorum.
– Special resolutions include resolutions amending the Company’s Articles of Association, dis-applying statutory pre-emption rights or changing the Company’s name; modifying the rights of any class of the Company’s shares at a meeting of the holders of such class or relating to certain matters concerning the Company’s winding up. A special resolution requires the affirmative vote of not less than three-quarters of the votes of the persons voting at the meeting at which there is a quorum.
Annual General Meetings must be convened upon advance written notice of 21 days. Other general meetings must be convened upon advance written notice of at least 14 clear days. The days of delivery or receipt of notice are not included. The notice must specify the nature of the business to be transacted. Meetings are convened by the Board of Directors. Members with 5% of the ordinary share capital of the Company may requisition the Board to convene a meeting.
Variation of rights
If, at any time, the Company’s share capital is divided into different classes of shares, the rights attached to any class may be varied, subject to the provisions of the Companies Act, with the consent in writing of holders of three-quarters in nominal value of the issued shares of that class or upon the adoption of a special resolution passed at a separate meeting of the holders of the shares of that class. At every such separate meeting, all the provisions of the Articles of Association relating to proceedings at a general meeting apply, except that the quorum is to be the number of persons (which must be two or more) who hold or represent by proxy not less than one-third in nominal value of the issued shares of the class and at any such meeting a poll may be demanded in writing by any person or their proxy who hold shares of that class. Where a person is present by proxy or proxies, he is treated as holding only the shares in respect of which the proxies are authorised to exercise voting rights.
Rights in a winding up
Except as the Company’s shareholders have agreed or may otherwise agree, upon the Company’s winding up, the balance of assets available for distribution:
– After the payment of all creditors including certain preferential creditors, whether statutorily preferred creditors or normal creditors; and
– Subject to any special rights attaching to any other class of shares;
– Is to be distributed among the holders of ordinary shares according to the amounts paid-up on the shares held by them. This distribution is generally to be made in US Dollars. A liquidator may, however, upon the adoption of any extraordinary resolution of the shareholders and any other sanction required by law, divide among the shareholders the whole or any part of the Company’s assets in kind.
Limitations on voting and shareholding
There are no limitations imposed by English law or the Company’s Articles of Association on the right of non-residents or foreign persons to hold or vote the Company’s ordinary shares or ADSs, other than the limitations that would generally apply to all of the Company’s shareholders.
Transfers of shares
The Board may refuse to register the transfer of shares held in certificated form which:
– Are not fully paid (provided that it shall not exercise this discretion in such a way as to prevent stock market dealings in the shares of that class from taking place on an open and proper basis);
– Are not duly stamped or duly certified or otherwise shown to the satisfaction of the Board to be exempt from stamp duty, lodged at the Transfer Office or at such other place as the Board may appoint and (save in the case of a transfer by a person to whom no certificate was issued in respect of the shares in question) accompanied by the certificate for the shares to which it relates, and such other evidence as the Board may reasonably require to show the right of the transferor to make the transfer and, if the instrument of transfer is executed by some other person on his behalf, the authority of that person so to do;
– Are in respect of more than one class of shares; or
– Are in favour of more than four transferees.
Deferred shares
Following the re-denomination of share capital on 23 January 2006, the ordinary shares’ nominal value became 20 US cents each. There were no changes to the rights or obligations of the ordinary shares. In order to comply with the Companies Act 2006, a new class of Sterling shares was created, deferred shares, of which £50,000 were issued and allotted in 2006 as fully paid to the Chief Executive Officer. These shares were subsequently transferred and are now held by the Company Secretary, although the Board reserves the right to transfer them to a member of the Board should it so wish. These deferred shares have no voting or dividend rights and on winding up only are entitled to repayment at nominal value only if all ordinary shareholders have received the nominal value of their shares plus an additional US$1,000 each.
Amendments
The Company does not have any special rules about amendments to its Articles of Association beyond those imposed by law.
|
210 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
CROSS REFERENCE TO FORM 20‑F
This table provides a cross reference from the information included in this Annual Report to the requirements of Form 20-F.
|
Part I |
|
Page |
|
Item 1 |
Identity of Directors, Senior Management and Advisers |
n/a |
|
Item 2 |
Offer Statistics and Expected Timetable |
n/a |
|
Item 3 |
Key Information |
|
|
|
A – Selected Financial Data |
192–198 |
|
|
B – Capitalization and Indebtedness |
n/a |
|
|
C – Reason for the Offer and Use of Proceeds |
n/a |
|
|
D – Risk Factors |
188–191 |
|
Item 4 |
Information on the Company |
|
|
|
A – History and Development of the Company |
180, 187, 200, 216 |
|
|
B – Business Overview |
2–49, 133–134, 199 |
|
|
C – Organizational Structure |
150–151, 182–186 |
|
|
D – Property, Plant and Equipment |
144–145, 187 |
|
Item 4A |
Unresolved Staff Comments |
None |
|
Item 5 |
Operating and Financial Review and Prospects |
|
|
|
A – Operating Results |
2, 4, 36–39, 188–191, 199 |
|
|
B – Liquidity and Capital Resources |
39, 154–156, 173–174 |
|
|
C – Research and Development, Patents and Licences, etc. |
4, 30–31, 137 |
|
|
D – Trend Information |
10–11, 38–39, 188–191 |
|
|
E – Off Balance Sheet Arrangements |
187 |
|
|
F – Tabular Disclosure of Contractual Obligations |
198 |
|
|
G – Safe Harbor |
216 |
|
Item 6 |
Directors, Senior Management and Employees |
|
|
|
A – Directors and Senior Management |
53–61 |
|
|
B – Compensation |
84–114 |
|
|
C – Board Practices |
53–83, 87 |
|
|
D – Employees |
24–25, 138 |
|
|
E – Share Ownership |
99–100, 177 |
|
Item 7 |
Major Shareholders and Related Party Transactions |
|
|
|
A – Major Shareholders |
204 |
|
|
B – Related Party Transactions |
177, 188 |
|
|
C – Interests of Experts and Counsel |
n/a |
|
Item 8 |
Financial information |
|
|
|
A – Consolidated Statements and Other Financial Information |
116–177, 194–198 |
|
|
– Legal Proceedings |
163–164 |
|
|
– Dividends |
203 |
|
|
B – Significant Changes |
None |
|
Item 9 |
The Offer and Listing |
|
|
|
A – Offer and Listing Details |
201–202, 205 |
|
|
B – Plan of Distribution |
n/a |
|
|
C – Markets |
201–202 |
|
|
D – Selling Shareholders |
n/a |
|
|
E – Dilution |
n/a |
|
|
F – Expenses of the Issue |
n/a |
|
|
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
211 |
|
Part I |
|
Page |
|
Item 10 |
Additional Information |
|
|
|
A – Share Capital |
n/a |
|
|
B – Memorandum and Articles of Association |
208–209 |
|
|
C – Material Contracts |
None |
|
|
D – Exchange Controls |
205 |
|
|
E – Taxation |
206–207 |
|
|
F – Dividends and Paying Agents |
n/a |
|
|
G – Statement by Experts |
n/a |
|
|
H – Documents on Display |
216 |
|
|
I – Subsidiary Information |
182–186 |
|
Item 11 |
Quantitative and Qualitative Disclosure about Market Risk |
156–161, 188–191 |
|
Item 12 |
Description of Securities Other than Equity Securities |
|
|
|
A – Debt Securities |
n/a |
|
|
B – Warrants and Rights |
n/a |
|
|
C – Other Securities |
n/a |
|
|
D – American Depositary Shares |
201–202 |
|
Part II |
|
|
|
Item 13 |
Defaults, Dividend Arrearages and Delinquencies |
None |
|
Item 14 |
Material Modifications to the Rights of Security Holders and Use of Proceeds |
None |
|
Item 15 |
Controls and Procedures |
82–83 |
|
Item 16 |
(Reserved) |
|
|
|
A – Audit Committee Financial Expert |
76 |
|
|
B – Code of Ethics |
83 |
|
|
C – Principal Accountant Fees and Services |
80–81, 138 |
|
|
D – Exemptions from the Listing Standards for Audit Committees |
n/a |
|
|
E – Purchases of Equity Securities by the Issuer and Affiliated Purchasers |
172, 205 |
|
|
F – Change in Registrant’s Certifying Accountant |
n/a |
|
|
G – Corporate Governance |
52 |
|
|
H – Mine Safety Disclosure |
n/a |
|
Part III |
|
|
|
Item 17 |
Financial Statements |
n/a |
|
Item 18 |
Financial Statements |
116–177, 194–198 |
|
Item 19 |
Exhibits |
217–223 |
|
212 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
GLOSSARY OF TERMS
Unless the context indicates otherwise, the following terms have the meanings shown below:
|
Term |
Meaning |
|
ACL |
The anterior cruciate ligament (ACL) is one of the four major ligaments in the human knee. |
|
ADR |
In the US, the Company’s ordinary shares are traded in the form of American Depositary Shares evidenced by American Depositary Receipts (ADRs). |
|
ADS |
In the US, the Company’s ordinary shares are traded in the form of American Depositary Shares (ADSs). |
|
Arthroscopic Enabling Technologies |
A product group which includes a variety of technologies such as fluid management equipment for surgical access, high definition cameras, digital image capture, scopes, light sources and monitors to assist with visualisation inside the joints, radio frequency, electromechanical and mechanical tissue resection devices, and hand instruments for removing damaged tissue. |
|
Advanced Wound Bioactives |
A product group which includes biologics and other bioactive technologies that provide unique approaches to debridement and dermal repair/regeneration. |
|
Advanced Wound Care |
A product group which includes products for the treatment and prevention of acute and chronic wounds, including leg, diabetic and pressure ulcers, burns and post-operative wounds. |
|
Advanced Wound Devices |
A product group which includes traditional and single-use Negative Pressure Wound Therapy and hydrosurgery systems. |
|
AGM |
Annual General Meeting of the Company. |
|
Arthroscopy |
Endoscopy of the joints is termed ‘arthroscopy’, with the principal applications being the knee and shoulder. |
|
Basis Point |
One hundredth of one percentage point. |
|
Chronic wounds |
Chronic wounds are those with long or unknown healing times including leg ulcers, pressure sores and diabetic foot ulcers. |
|
Company |
Smith & Nephew plc or, where appropriate, the Company’s Board of Directors, unless the context otherwise requires. |
|
Companies Act |
Companies Act 2006, as amended, of England and Wales. |
|
Emerging Markets |
Emerging Markets include include Latin America, Asia (excluding Japan), Africa and Russia. |
|
EPSA |
EPSA (Adjusted earnings per ordinary share) is a trend measure, which presents the long-term profitability of the Group excluding the post-tax impact of specific transactions that management considers affects the Group’s short-term profitability. The Group presents this measure to assist investors in their understanding of trends. Adjusted attributable profit is the numerator used for this measure and is determined by adjusting attributable profit for the items that are excluded from operating profit when arriving at trading profit and items that are recognised below operating profit that affect the Group’s short-term profitability. |
|
Endoscopy |
Through a small incision, surgeons are able to see inside the body using a monitor and identify and repair defects. |
|
Established Markets |
Established Markets include United States of America, Europe, Australia, New Zealand, Canada and Japan. |
|
Euro or € |
References to the common currency used in the majority of the countries of the European Union. |
|
FDA |
US Food and Drug Administration. |
|
Financial statements |
Refers to the consolidated Group Accounts of Smith & Nephew plc. |
|
FTSE 100 |
Index of the largest 100 listed companies on the London Stock Exchange by market capitalisation. |
|
Group or Smith & Nephew |
Used for convenience to refer to the Company and its consolidated subsidiaries, unless the context otherwise requires. |
|
Health economics |
A branch of economics concerned with issues related to efficiency, effectiveness, value and behaviour in the production and consumption of health and healthcare. |
|
Hip Implants |
A product group which includes specialist products for reconstruction of the hip joint. |
|
IFRIC |
International Financial Reporting Interpretations as adopted by the EU and as issued by the International Accounting Standards Board. |
|
IFRS |
International Financial Reporting Standards as adopted by the EU and as issued by the International Accounting Standards Board. |
|
|
Smith & Nephew Annual Report 2018 |
|
213 |
|
Term |
Meaning |
|
Knee implants |
A product group which includes an innovative range of products for specialised knee replacement procedures. |
|
LSE |
London Stock Exchange. |
|
MHRA |
The Medicines and Healthcare products Regulatory Agency in the UK. |
|
Negative Pressure Wound Therapy |
A technology used to treat chronic wounds such as diabetic ulcers, pressure sores and post-operative wounds through the application of sub-atmospheric pressure to an open wound. |
|
NHS |
The UK National Health Service. |
|
NYSE |
New York Stock Exchange. |
|
Orthopaedic products |
Orthopaedic reconstruction products include joint replacement systems for knees, hips and shoulders and support products such as computer-assisted surgery and minimally invasive surgery techniques. Orthopaedic trauma devices are used in the treatment of bone fractures including rods, pins, screws, plates and external frames. |
|
Other Surgical Businesses |
A product group which includes robotics-assisted surgery, various products and technologies to assist in surgical treatment of the ear, nose and throat, and gynaecological instrumentation, until the Gynaecology business disposal in August 2016. |
|
OXINIUM |
OXINIUM material is an advanced load bearing technology. It is created through a proprietary manufacturing process that enables zirconium to absorb oxygen and transform to a ceramic on the surface, resulting in a material that incorporates the features of ceramic and metal. Management believes that OXINIUM material used in the production of components of knee and hip implants exhibits unique performance characteristics due to its hardness, low-friction and resistance to roughening and abrasion. |
|
Parent Company |
Smith & Nephew plc. |
|
Pound Sterling, Sterling, £, |
References to UK currency. 1p is equivalent to one hundredth of £1. |
|
SEC |
US Securities and Exchange Commission. |
|
Sports Medicine Joint Repair |
The Sports Medicine Joint Repair franchise includes instruments, technologies and implants necessary to perform minimally invasive surgery of joints. |
|
Trading results |
Trading profit, trading profit margin (trading profit expressed as a percentage of revenue), trading cash flow and trading profit to cash conversion ratio (trading cash flow expressed as a percentage of trading profit) are trend measures, which present the long-term profitability of the Group excluding the impact of specific transactions that management considers affect the Group’s short-term profitability and cash flows. The Group has identified the following items, where material, as those to be excluded from operating profit and cash generated from operations when arriving at trading profit and trading cash flow, respectively: acquisition and disposal related items arising in connection with business combinations, including amortisation of acquisition intangible assets, impairments and integration costs; restructuring events; gains and losses resulting from legal disputes and uninsured losses. In addition to these items, gains and losses that materially impact the Group’s profitability or cash flows on a short-term or one-off basis and the cash cost to fund defined benefit pension schemes that are closed to future accrual are excluded from operating profit and cash generated from operations when arriving at trading profit and trading cash flow, respectively. |
|
Trauma & Extremities |
A product group which includes internal and external devices used in the stabilisation of severe fractures and deformity correction procedures. |
|
UK |
United Kingdom of Great Britain and Northern Ireland. |
|
Underlying growth |
Growth after adjusting for the effects of currency translation and the inclusion of the comparative impact of acquisitions and exclusion of disposals. |
|
US |
United States of America. |
|
US Dollars, $ or cents or ¢ |
References to US currency. 1 cent is equivalent to one hundredth of US$1. |
|
214 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
INDEX
|
2017 Financial highlights |
199 |
|
Group statement of comprehensive income |
125 |
|
Accounting Policies |
124, 129–131 |
|
Report of Independent Registered Public Accounting Firm |
117 |
|
Accounts Presentation |
216 |
|
Information for shareholders |
201–216 |
|
Acquisitions |
7, 36, 175–176 |
|
Intangible assets |
148–150 |
|
Acquisition and disposal related items |
135, 196, 197 |
|
Intellectual property |
163–164 |
|
American Depositary Shares |
201–202 |
|
Interest and other finance costs |
138–139 |
|
Articles of Association |
208–209 |
|
Inventories |
151–152 |
|
Audit fees |
81, 138 |
|
Investments |
150 |
|
Board |
54–57 |
|
Investment in associates |
150–151 |
|
Business overview |
2–3, 187–191 |
|
Key Performance Indicators |
4 |
|
Business segment information |
14–22, 132–136 |
|
Leases |
137, 176 |
|
Cash and borrowings |
154–156 |
|
Legal and other |
135, 196, 197 |
|
Chairman’s statement |
4–5 |
|
Legal proceedings |
163–164 |
|
Chief Executive Officer’s review |
6–7 |
|
Liquidity and capital resources |
39, 156 |
|
Chief Financial Officer’s review |
36–37 |
|
Manufacturing and quality |
28–29 |
|
Company balance sheet |
178 |
|
Medical education |
29 |
|
Company notes to the accounts |
180–186 |
|
New accounting standards |
129–131 |
|
Contingencies |
162–164, 182 |
|
Off-balance sheet arrangements |
187 |
|
Contractual obligations |
198 |
|
Operating profit |
137–138 |
|
Corporate Governance Statement |
52 |
|
Other finance costs |
139 |
|
Critical judgements and estimates |
124 |
|
Our markets |
10–11 |
|
Cross Reference to Form 20-F |
210–211 |
|
Outlook and trend information |
10–11, 36–39, |
|
Currency fluctuations |
189 |
|
Parent Company accounts |
178–186 |
|
Currency translation |
131 |
|
People/Employees |
24–25 |
|
Deferred taxation |
142 |
|
Provisions |
162–164 |
|
Directors’ Remuneration Report |
84–114 |
|
Property, plant and equipment |
144–145 |
|
Directors’ responsibility statement |
116 |
|
Regulation |
10, 43 |
|
Dividends |
173, 203 |
|
Related party transactions |
177, 188 |
|
Earnings per share |
4, 37, 38, 143–144 |
|
Research and development |
30–31 |
|
Employees/People |
24–25 |
|
Restructuring and rationalisation expenses |
135, 196, 197 |
|
Employee share plans |
177 |
|
Retirement benefit obligation |
165–170 |
|
Ethics and compliance |
26, 74–75 |
|
Risk factors |
188–191 |
|
Executive Officers |
58–61 |
|
Risk report |
40–51 |
|
Factors affecting results of operations |
191 |
|
Sales and marketing |
27 |
|
Financial instruments |
156–161 |
|
Selected financial data |
192–193 |
|
Financial review |
38–39 |
|
Share based payments |
177 |
|
Glossary of terms |
212–213 |
|
Share capital |
171–172, 204–205 |
|
Goodwill |
146–147 |
|
Strategic imperatives |
9 |
|
Group balance sheet |
126 |
|
Sustainability |
32–34 |
|
Group cash flow statement |
127 |
|
Taxation |
139–142 |
|
Group companies |
182–186 |
|
Taxation information for shareholders |
206–207 |
|
Group history |
187 |
|
Total shareholder return |
103 |
|
Group income statement |
125 |
|
Trade and other payables |
153–154 |
|
Group notes to the accounts |
129–177 |
|
Trade and other receivables |
152–153 |
|
Group overview |
2–3, 187 |
|
Treasury shares |
172 |
|
Group statement of changes in equity |
128 |
|
|
|
|
|
|
|
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
215 |
IRAN NOTICE
Section 13(r) of the Exchange Act requires issuers to make specific disclosure in their annual reports of certain types of dealings with Iran, including transactions or dealings with Iranian government-owned entities, as well as dealings with entities sanctioned for activities related to terrorism or proliferation of weapons of mass destruction, even when those activities are not prohibited by US law and do not involve US persons.
The Group does not have a legal entity based in Iran, but in 2018 it exported certain medical devices to Iran, via sales by non- US entities, to a privately owned Iranian distributor for sale in Iran. Sales by the distributor were made to hospitals that we understand are owned or controlled by the Government of Iran.
The Group’s direct and indirect sales of US origin medical devices into Iran are permitted pursuant to section 560.530(a)(3)(i) of the Iranian Transactions and Sanctions Regulations, and its indirect sales of non-US origin medical devices into Iran are made in accordance with applicable law. The Group also provides training to its distributor(s) and surgeons in Iran as necessary and ordinarily incident to the safe and effective use of the medical devices, which is also permitted by applicable law.
In 2018, S&N’s gross revenues from sales to Iran were approximately US$3.4m and net losses were approximately US$1.1m.
The Group is reporting the entire gross revenues and net profits for the activities described above, which figures include sales of US origin medical devices. Although the Group is not required to disclose the sales of US origin medical devices because such sales to Iran are licensed under US law, the Group is including sales of these devices in its total gross revenue and net profit figures as it does not separately break out revenues and profits by country of origin.
ABOUT SMITH & NEPHEW
The Smith & Nephew Group (the Group) is a portfolio medical technology business with leadership positions in Orthopaedics, Advanced Wound Management and Sports Medicine, and revenue of approximately $4.9bn in 2018. Smith & Nephew plc (the Company) is the Parent Company of the Group. It is an English public limited company with its shares listed on the premium list of the UK Listing Authority and traded on the London Stock Exchange. Shares are also traded on the New York Stock Exchange in the form of American Depositary Shares (ADSs).
This is the Annual Report of Smith & Nephew plc for the year ended 31 December 2018. It comprises, in a single document, the Annual Report and Accounts of the Company in accordance with UK requirements and the Annual Report on Form 20-F in accordance with the regulations of the United States Securities and Exchange Commission (SEC).
Smith & Nephew operates on a worldwide basis and has distribution channels in over 100 countries. The Group is engaged in a single business activity, being the development, manufacture and sale of medical technology products and services. In 2018, the Group was structured as two geographical selling regions: US and International; with a president for each who is responsible for the commercial view of that region. Research & Development, Manufacturing, Supply Chain and Central functions are managed globally for the Group as a whole.
Smith & Nephew’s corporate website, www.smith-nephew.com, gives additional information on the Group, including an electronic version of this Annual Report. Information made available on this website, or other websites mentioned in this Annual Report, are not and should not be regarded as being part of, or incorporated into, this Annual Report.
For the convenience of the reader, a Glossary of technical and financial terms used in this document is included on pages 212–213. The product names referred to in this document are identified by use of capital letters and the TM (on first occurrence) and are trademarks owned by or licensed to members of the Group.
|
216 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
INFORMATION FOR SHAREHOLDERS continued
PRESENTATION
The Group’s fiscal year end is 31 December. References to a particular year in this Annual Report are to the fiscal year, unless otherwise indicated. Except as the context otherwise requires, ‘Ordinary Share’ or ‘share’ refer to the ordinary shares of Smith & Nephew plc of 20 US cents each.
The Group Accounts of Smith & Nephew in this Annual Report are presented in US Dollars. Solely for the convenience of the reader, certain parts of this Annual Report contain translations of amounts in US Dollars into Sterling at specified rates. These translations should not be construed as representations that the US Dollar amounts actually represent such Sterling amounts or could be converted into Sterling at the rate indicated.
Unless stated otherwise, the translation of US Dollars and cents to Sterling and pence in this Annual Report has been made at the Bank of England exchange rate on the date indicated. On 15 February 2019, the latest practicable date for this Annual Report, the Bank of England rate was US$1.28 per £1.00.
The results of the Group, as reported in US Dollars, are affected by movements in exchange rates between US Dollars and other currencies. The Group applied the average exchange rates prevailing during the year to translate the results of companies with functional currency other than US Dollars. The currencies which most influenced these translations in the years covered by this report were Sterling, Swiss Franc and the Euro.
The Accounts of the Group in this Annual Report are presented in millions (m) unless otherwise indicated.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
The Group’s reports filed with, or furnished to, the US Securities and Exchange Commission (SEC), including this document and written information released, or oral statements made, to the public in the future by or on behalf of the Group, contain ‘forward-looking statements’ within the meaning of the US Private Securities Litigation Reform Act of 1995, that may or may not prove accurate. For example, statements regarding expected revenue growth and trading profit margins discussed under ‘Outlook’ and ‘Strategic Priorities’, market trends and our product pipeline are forward-looking statements. Phrases such as ‘aim’, ‘plan’, ‘intend’, ‘anticipate’, ‘well-placed’, ‘believe’, ‘estimate’, ‘expect’, ‘target’, ‘consider’ and similar expressions are generally intended to identify forward-looking statements. Forward-looking statements involve known and unknown risks, uncertainties and other important factors that could cause actual results, to differ materially from what is expressed or implied by the statements.
For Smith & Nephew, these factors include: economic and financial conditions in the markets we serve, especially those affecting healthcare providers, payers and customers; price levels for established and innovative medical devices; developments in medical technology; regulatory approvals, reimbursement decisions or other government actions; product defects or recalls; litigation relating to patent or other claims; legal compliance risks and related investigative, remedial or enforcement actions; strategic actions, including acquisitions and dispositions and our success in performing due diligence, valuing and integrating acquired businesses; disruption that may result from transactions or other changes we make in our business plans or organisation to adapt to market developments and numerous other matters that affect us or our markets, including those of a political, economic, business, competitive or reputational nature; relationships with healthcare professionals; reliance on information technology. Specific risks faced by the Group are described under ‘Risk factors’ on pages 188–191 of this Annual Report. Any forward-looking statement is based on information available to Smith & Nephew as of the date of the statement. All written or oral forward-looking statements attributable to Smith & Nephew are qualified by this caution. Smith & Nephew does not undertake any obligation to update or revise any forward-looking statement to reflect any change in circumstances or in Smith & Nephew’s expectations.
PRODUCT DATA
Product data and product share estimates throughout this report are derived from a variety of sources including publicly available competitors’ information, internal management information and independent market research reports.
DOCUMENTS ON DISPLAY
It is possible to read and copy documents referred to in this Annual Report at the Registered Office of the Company. Documents referred to in this Annual Report that have been filed with the Securities and Exchange Commission in the US may be read and copied at the SEC’s public reference room located at 450 Fifth Street, NW, Washington DC 20549. Please call the SEC at 1-800-SEC-0330 for further information on the public reference rooms and their copy charges. The SEC also maintains a website at www.sec.gov that contains reports and other information regarding registrants that file electronically with the SEC. This Annual Report and some of the other information submitted by the Group to the SEC may be accessed through the SEC website.
|
|
Smith & Nephew Annual Report 2018 |
|
217 |
EXHIBIT INDEX
|
Exhibit No. |
Description of Document |
Incorporated Herein by Reference To |
Filed |
|
|
|
|
|
|
|
|
1 |
|
Form 20-F for the year ended December 31, 2011 filed on March 1, 2012 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
2 |
|
Smith & Nephew plc is not party to any single instrument relating to long-term debt pursuant to which a total amount of securities exceeding 10% of Smith & Nephew plc’s total assets (on a consolidated basis) is authorized to be issued. Smith & Nephew plc hereby agrees to furnish to the SEC, upon its request, a copy of any instrument defining the rights of holders of its long-term debt or the rights of holders of the long-term debt of any of its subsidiaries for which consolidated or unconsolidated financial statements are required to be filed with the SEC. |
|
|
|
|
|
|
|
|
|
4 |
(a) (i) |
Form 20-F for the year ended December 31, 2013 filed on March 6, 2014 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(ii) |
Form 20-F for the year ended December 31, 2013 filed on March 6, 2014 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(iii) |
Form 20-F for the year ended December 31, 2014 filed on March 5, 2015 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
218 |
|
Smith & Nephew Annual Report 2018 |
|
|
Exhibit No. |
Description of Document |
Incorporated Herein by Reference To |
Filed |
|
|
|
|
|
|
|
|
4 |
(a) (iv) |
Form 20-F for the year ended December 31, 2014 filed on March 5, 2015 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(v) |
|
X |
|
|
|
|
|
|
|
|
4 |
(c) (i) |
Form 20-F for the year ended December 31, 2010 filed on March 3, 2011 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(ii) |
Form 20-F for the year ended December 31, 2009 filed on March 26, 2010 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(iii) |
Letter of Appointment of The Rt. Hon Baroness Virginia Bottomley |
Form 20-F for the year ended December 31, 2012 filed on February 28, 2013 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
(lv) |
Form 20-F for the year ended December 31, 2012 filed on February 28, 2013 (File No.1-14978) |
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
219 |
|
Exhibit No. |
Description of Document |
Incorporated Herein by Reference To |
Filed |
|||||||
|
|
|
|
|
|
||||||
|
4 |
(c) (v) |
Form 20-F for the year ended December 31, 2016 filed on March 6, 2017 (File No.1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(vi) |
Form 20-F for the year ended December 31, 2013 filed on March 6, 2014 (File No.1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(vii) |
Form 20-F for the year ended December 31, 2014 filed on March 5, 2015 (File No.1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(viii) |
Form 20-F for the year ended December 31, 2014 filed on March 5, 2015 (File No.1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(ix) |
Form 20-F for the year ended December 31, 2015 filed on March 4, 2016 (File No.1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(x) |
Form 20-F for the year ended December 31, 2015 filed on March 4, 2016 (File No.1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(xi) |
Form 20-F for the year ended December 31, 2015 filed on March 4, 2016 (File No.1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(xii) |
Form 20-F for the year ended December 31, 2004 filed on March 16, 2005 (File No. 1-14978) |
|
|||||||
|
|
|
|
|
|
||||||
|
|
(xiii) |
Form 20-F for the year ended December 31, 2004 filed on March 16, 2005 (File No. 1-14978) |
|
|||||||
|
220 |
|
Smith & Nephew Annual Report 2018 |
|
|
Exhibit No. |
Description of Document |
Incorporated Herein by Reference To |
Filed |
|
|
|
|
|
|
|
|
4 |
(c) (xiv) |
The Smith & Nephew 2001 US Share Plan |
Registration Statement on Form S-8 No. 333-13694 filed on July 9, 2001 (File No. 1-14978) |
|
|
|
|
|
|
|
|
(xv) |
Form 20-F for the year ended December 31, 2002 filed on April 25, 2003 (File No. 1-14978) |
|||
|
|
(xvi) |
Form 20-F for the year ended December 31, 2004 filed on March 16, 2005 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xvii) |
Form 20-F for the year ended December 31, 2002 filed on April 25, 2003 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xviii) |
Form 20-F for the year ended December 31, 2002 filed in April 25, 2003 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xix) |
Form 20-F for the year ended December 31, 2002 filed on April 25, 2003 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xx) |
Form 20-F for the year ended December 31, 2002 filed on April 25, 2003 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxi) |
Form 20-F for the year ended December 31, 2003 filed on March 26, 2004 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxii) |
Registration statement on Form S-8 No. 333-122801 filed on February 14, 2005 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxiii) |
Registration statement on Form S-8 No. 333-122801 filed on February 14, 2005 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxiv) |
Registration statement on Form S-8 No. 333-122801 filed on February 14, 2005 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxv) |
Smith & Nephew U.S. Employee Stock Purchase Plan |
Registration statement on Form S-8 No. 333-12052 filed on May 30, 2000 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
(xxvi) |
Smith & Nephew Long Service Award Scheme |
Registration Statement on Form S-8 No. 33-39814 filed on April 5, 1991 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
(xxvii) |
Registration statement on Form S-8 No. 333-155172 filed on November 7, 2008 (File No. 1-14978) |
|
|
|
|
Smith & Nephew Annual Report 2018 |
|
221 |
|
Exhibit No. |
Description of Document |
Incorporated Herein by Reference To |
Filed |
|
|
4 |
(c)(xxviii) |
Registration statement on Form S-8 No. 333-155173 filed on November 7, 2008 (File No. 1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxix) |
Registration statement on Form S-8 No. 333-158239 filed on March 27, 2009 (File No. 1-14978) |
|
|
|
|
(xxx) |
Form 20-F for the year ended December 31, 2016 filed on March 6, 2017 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxxi) |
Form 20-F for the year ended December 31, 2012 filed on February 28, 2013 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxxii) |
Form 20-F for the year ended December 31, 2012 filed on February 28, 2013 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxxiii) |
Form 20-F for the year ended December 31, 2016 filed on March 6, 2017 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxxiv) |
Letter of Appointment of Robin Freestone as Audit Committee Chairman |
Form 20-F for the year ended December 31, 2016 filed on March 6, 2017 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
(xxxv) |
Letter of Appointment of Ian Barlow as Senior Independent Director |
Form 20-F for the year ended December 31, 2016 filed on March 6, 2017 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
(xxxvi) |
Form 20-F for the year ended December 31, 2016 filed on March 6, 2017 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxxvii) |
Form 20-F for the year ended December 31, 2017 filed on March 5, 2018 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxxviii) |
Form 20-F for the year ended December 31, 2017 filed on March 5, 2018 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xxxix) |
Form 20-F for the year ended December 31, 2017 filed on March 5, 2018 (File No.1-14978) |
|
|
|
|
|
|
|
|
|
|
(xl) |
Form 20-F for the year ended December 31, 2017 filed on March 5, 2018 (File No.1-14978) |
|
|
|
222 |
|
Smith & Nephew Annual Report 2018 |
|
|
|
|
|
|
|
|
Exhibit No. |
Description of Document |
Incorporated Herein by Reference To |
Filed |
|
|
|
(xli) |
Form 20-F for the year ended December 31, 2017 filed on March 5, 2018 (File No.1-14978) |
|
|
|
4 |
(c) (xlii) |
|
X |
|
|
|
|
|
|
|
|
|
(xliii) |
Letter of Re-Appointment of The Rt. Hon Baroness Virginia Bottomley |
|
X |
|
|
|
|
|
|
|
|
(xliv) |
|
X |
|
|
|
|
|
|
|
|
8 |
|
|
X |
|
|
|
|
|
|
|
|
12 |
(a) |
Certification of Namal Nawana, filed pursuant to Exchange Act Rule 13a -14(a) |
|
X |
|
|
|
|
|
|
|
|
(b) |
Certification of Graham Baker filed pursuant to Exchange Act Rule 13a -14(a) |
|
X |
|
|
|
|
|
|
|
13 |
(a) |
Certification of Namal Nawana and Graham Baker furnished pursuant to Exchange Act Rule 13a – 14(b) |
|
X |
|
|
|
|
|
|
|
15.1 |
|
Consent of KPMG LLP, Independent Registered Public Accounting Firm
|
|
X |
|
|
Smith & Nephew Annual Report 2018 |
|
223 |
|
Exhibit No. |
Description of Document |
Incorporated Herein by Reference To |
Filed |
|
|
|
|
|
|
|
|
101.INS* |
|
XBRL Instance Document |
|
|
|
|
|
|
|
|
|
101.SCH* |
|
XBRL Taxonomy Extension Schema Document |
|
|
|
|
|
|
|
|
|
101.CAL* |
|
XBRL Taxonomy Extension Calculation Linkbase Document |
|
|
|
|
|
|
|
|
|
101.LAB* |
|
XBRL Taxonomy Extension Label Linkbase Document |
|
|
|
|
|
|
|
|
|
101.PRE* |
|
XBRL Taxonomy Extension Presentation Linkbase Document |
|
|
|
|
|
|
|
|
|
101.DEF* |
|
XBRL Taxonomy Extension Definition Linkbase Document |
|
|
|
224 |
Other Information |
Smith & Nephew Annual Report 2018 |
|
SIGNATURE
The Registrant hereby certifies that it meets all of the requirements for filing on Form 20-F and has duly caused and authorized the undersigned to sign this Annual Report on its behalf.
|
|
Smith & Nephew plc |
|
|
(Registrant) |
|
|
|
|
By: |
/s/ Susan Swabey |
|
|
Susan Swabey |
|
|
Company Secretary |
London, England
March 4, 2019

 Page 8
Page 8 Page 28‑29
Page 28‑29
 Page 15‑17
Page 15‑17






















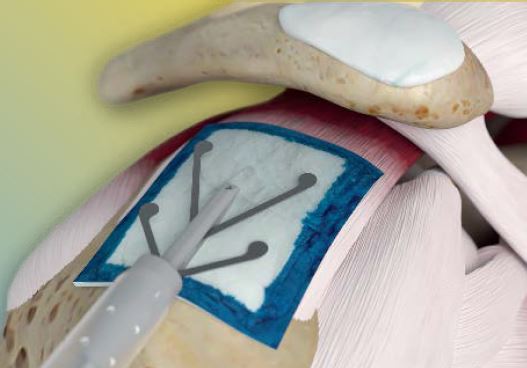
























 American/Italian
American/Italian Australian/French
Australian/French British
British


 Indian
Indian British
British Swiss
Swiss


 Swedish
Swedish British
British



 Italian
Italian American
American



 American/Canadian
American/Canadian





 American/South African
American/South African American
American Board timetable 2018
Board timetable 2018