Exhibit 99.1

GeoVax Reports 2021 Year-End Financial Results
And Provides Corporate Update
Advancing Phase 2 Clinical Trials for COVID-19 and Immuno-Oncology
ATLANTA, GA, March 9, 2022 – GeoVax Labs, Inc. (Nasdaq: GOVX), a biotechnology company developing immunotherapies and vaccines against infectious diseases and cancer, today announced its financial results for the year ended December 31, 2021 and provided an update on product development programs. GeoVax’s management will host a live conference call and webcast at 4:30 p.m. Eastern Standard Time on Wednesday, March 9 to discuss financial results and provide a general business update. Details are provided further below.
Two Phase 2 Clinical Trials Underway for SARS-CoV-2
GEO-CM04S1 for Immunocompromised Patients – GEO-CM04S1 is being studied in an ongoing Phase 2 clinical trial (NCT04977024) to evaluate its safety and immunogenicity, compared to the Pfizer/BioNTech mRNA-based vaccine, in patients who have previously received either an allogeneic hematopoietic cell transplant, an autologous hematopoietic cell transplant or chimeric antigen receptor (CAR) T cell therapy. GEO-CM04S1 is the only COVID-19 vaccine that includes both SARS-CoV-2 spike and nucleocapsid proteins to advance to a Phase 2 trial in cancer patients. Such vaccines tend to produce an immune response quickly – in less than 14 days – with only mild side effects. The trial is also the first to compare an investigational multi-antigenic COVID-19 vaccine to the current Food and Drug Administration (FDA)-approved mRNA vaccine from Pfizer/BioNTech in people who are immunocompromised. Such patients have often shown a weak antibody response after receiving currently available COVID-19 vaccines.
GEO-CM04S1 as a Booster Vaccine – In December 2021, patient enrollment began for the Phase 2 portion of a Phase 1/2 trial (NCT04639466) of GEO-CM04S1, evaluating its use as a universal booster vaccine to current FDA-approved two-shot mRNA vaccines from Pfizer/BioNTech and Moderna. The completed Phase 1 portion of the trial was designed as a dose-escalation safety study in healthy individuals who had not been previously infected with SARS-CoV-2. The ongoing Phase 2 booster study includes healthy individuals who were previously fully vaccinated with either the Pfizer/BioNTech or Moderna vaccine. The dose-escalation study is designed to specifically evaluate the safety profile and immunogenicity of GEO-CM04S1 as a booster. The immunological responses measured throughout the study will include the level of SARS-CoV-2 neutralizing antibodies against SARS-CoV-2 variants of concern (VOC), including the newly identified Omicron VOC, as well as specific T-cell responses.
IND-Enabling Activities Progressing for Pan Coronavirus Vaccine
GEO-CM02 as a Pan-Coronavirus Vaccine – First-generation SARS-CoV-2 vaccines were designed to encode the spike (S) protein of the SARS-CoV-2 virus with the goal of inducing high levels of neutralizing antibodies. However, potential limitations of narrowly focusing on the spike (S) protein are becoming apparent with emerging variants capable of partially escaping neutralization by vaccine induced antibodies. Thus, the effectiveness of these vaccines against new SARS-CoV-2 variants and future coronavirus spillover events remains of immense concern.
GeoVax’s vaccine candidate (GEO-CM02) encodes the spike (S) protein as the neutralizing antibody target as well as the membrane (M) and envelope (E) proteins as T-cell targets and to support in vivo virus-like particle formation to augment potency. This strategy may provide the basis for generating a single dose universal coronavirus vaccine. Unique compared to other vaccines approved or under development, the GeoVax vaccine candidate is therefore specifically designed to provide a broader and more durable level of protective immunity against SARS-CoV-2, which may protect against emerging variants while avoiding the potential side effects that can limit vaccine utility and acceptance. In small animal studies, the Company measured functional immune responses after a single dose that mediated protection from infection and pathogenesis, including protection against the more virulent Beta variant. Additional studies are planned for 2022 to prepare for IND filing and subsequent human clinical trials.
MORE
Phase 2 Clinical Trial Underway for Advanced Head and Neck Cancer
Gedeptin® – Gedeptin is a novel, patented product/technology for the treatment of solid tumors through a gene therapy strategy known as Gene-Directed Enzyme Prodrug Therapy (GDEPT). In September 2021, GeoVax entered into an assignment and license agreement with PNP Therapeutics, Inc. (“PNP”), granting GeoVax exclusive rights to develop and commercialize Gedeptin. The Gedeptin technology was developed with funding support from the National Cancer Institute (NCI), part of the NIH. GeoVax’s license to Gedeptin includes the rights to expand the use of Gedeptin to all human diseases and/or conditions including, but not limited to, other cancers.
In GDEPT, a vector is used to selectively transduce tumor cells with a nonhuman gene, which expresses an enzyme that can convert a nontoxic prodrug into a very toxic antitumor compound, in situ. A cycle of Gedeptin therapy consists of three intra-tumoral injections of Gedeptin over a two-day period followed by infusion of a prodrug, fludarabine phosphate, once a day for three days. A Phase 1 dose ranging study, evaluating the safety of a single cycle of Gedeptin therapy, found the therapy to be well tolerated, with evidence of a reduction in tumor size in patients with solid tumors.
A Phase 1/2 trial (NCT03754933), evaluating the safety and efficacy of repeat cycles of Gedeptin therapy in patients with recurrent head and neck squamous cell carcinoma (HNSCC), with tumor(s) accessible for injection and no curable treatment options, is currently enrolling at Stanford University in collaboration with Emory University. The trial design involves repeat administration using Gedeptin followed by systemic fludarabine, to gain additional information prior to expansion towards a larger patient trial. The initial stage of the study is being funded by the FDA under its Orphan Products Clinical Trials Grants Program. The FDA has also granted Gedeptin orphan drug status for the intra-tumoral treatment of anatomically accessible oral and pharyngeal cancers, including cancers of the lip, tongue, gum, floor of mouth, salivary gland and other oral cavities. In January 2022, we engaged CATO SMS, a global provider of clinical research solutions, to manage the ongoing Phase 1/2 trial and to assist with the expansion of clinical sites and acceleration of patient enrollment and evaluation.
IND-Enabling Activities Progressing for MUC1-based Cancer Immunotherapy
MVA-VLP-MUC1 – Using our GV-MVA-VLP™ vaccine platform, we are developing a cancer immunotherapy based on the abnormal, aberrantly glycosylated forms of the cell surface-associated MUC1 protein that is expressed on a wide range of cancers, including breast, colon, ovarian, prostate, pancreatic, and lung, with the goal of raising therapeutic anti-tumor antibodies and T cell responses in cancer patients. Our cancer immunotherapy program is based on the concept of combining a tumor-associated antigen vaccine with a potent anti-tumor agent, such as an Immune Checkpoint Inhibitor (“ICI”), with the goal of achieving regression of tumor growth and development.
The initial animal studies of our MVA-VLP-MUC1 vaccine and ICI combination, have been encouraging, showing that a combination of our MVA-VLP-MUC1 vaccine candidate with a MUC1 synthetic peptide was capable of breaking tolerance to human MUC1 in transgenic mice and inducing immune responses with efficacy against challenge in a lymphoma tumor model. Our studies also demonstrated a significant reduction of the tumor burden in a mouse model for colorectal cancer.
In 2022 we plan to further these animal studies in collaboration to define the optimal course and schedule of vaccination to define a protocol that can be evaluated in a Phase 1 clinical trial.
MORE
Other Infectious Disease Vaccine Programs
Hemorrhagic Fever Virus Vaccines (Ebola, Sudan, Marburg and Lassa) – Our initial preclinical studies in rodents and nonhuman primates for our MVA-VLP-EBOV vaccine candidate have shown 100% protection against a lethal dose of EBOV upon a single immunization. Recent studies in lethal challenge guinea pig models demonstrated that GeoVax vaccines MVA-VLP-SUDV and MVA-VLP-MARV conferred 100% protection from death. These vaccines were subsequently evaluated in a rigorous cynomolgus macaque infectious challenge model. Vaccination protected nonhuman primates from viremia, weight loss and death following challenge with a dose of Sudan or Marburg virus that is lethal in nonvaccinated animals. Evaluation of immune responses following vaccination demonstrated presence of both neutralizing antibodies and functional T cells, indicating a breadth of responses that combine for optimal protection. Likewise, our initial preclinical studies in rodents for our LASV vaccine candidate have shown 100% single-dose protection against a lethal dose of LASV challenge composed of multiple strains delivered directly into the brain. The nonhuman primate studies are ongoing in collaboration with NIAID and the U.S. Army and clinical development programs will be defined based on efficacy data and global priorities as potentially dangerous outbreaks occur.
Malaria – We have collaborated with the Burnet Institute, a leading infectious diseases research institute in Australia, for the development of a vaccine to prevent malaria infection. The project included the design, construction, and characterization of multiple malaria vaccine candidates using GeoVax’s GV-MVA-VLP™ vaccine platform combined with malaria Plasmodium falciparum and Plasmodium vivax sequences identified by the Burnet Institute. We also collaborated separately with Leidos, Inc. with work funded by a grant to Leidos from the United States Agency for International Development (USAID) Malaria Vaccine Development Program (MVDP). This program has recently been inactive as we have prioritized our other development programs. However, pending additional funding support via federal grants or other sources, we may further pursue this area.
Zika – To address the unmet need for a vaccine against Zika virus, we are developing novel vaccine candidates constructed using our GV-MVA-VLPTM platform. MVA has an outstanding safety record, which is particularly important given the need to include women of child-bearing age and newborns among those being vaccinated. Our Zika vaccine is designed around the NS1 gene product to eliminate the risk of Antibody Dependent Enhancement (ADE), which is a serious side effect observed when a vaccinated individual does not have a fully protective immune response which causes a more virulent reaction if infected. Our initial preclinical studies in rodents using our GEO-ZM02 vaccine candidate demonstrated 100% single-dose protection against a lethal dose of ZIKV delivered directly into the brain. In rhesus macaques, vaccination with GEO-ZM02 induced immune responses that effectively controlled the virus replication despite the fact the vaccine is not designed to induce ZIKV neutralizing antibodies. Further development of our Zika vaccine will be dependent upon partnering support.
HIV – Due to our corporate refocusing of development efforts prioritizing our SARS-CoV-2 and cancer immunotherapy programs, and to a lack of continuing government support for our HIV vaccine development efforts, we recently decided to discontinue active development of these programs. Our technology and intellectual property in this will remain available for out-license or partnering, but we will no longer devote any corporate resources to the programs.
Management Commentary
David Dodd, GeoVax’s Chairman and CEO, commented, “Our primary corporate focus continues to be on our COVID-19 vaccine and our cancer immunotherapy programs, and the three ongoing Phase 2 clinical trials in those areas represent the achievement of highly important goals we established at the beginning of 2021. As recently communicated in our shareholder update letter in January, our 2022 goals include the acceleration and expansion of these clinical programs focused on generating relevant data as soon as possible. To that end, we have added organizational and operational resources to support advancing through clinical development into regulatory registration. We look forward to providing additional updates as we make progress in these, and other, programs.”
MORE
Financial Review
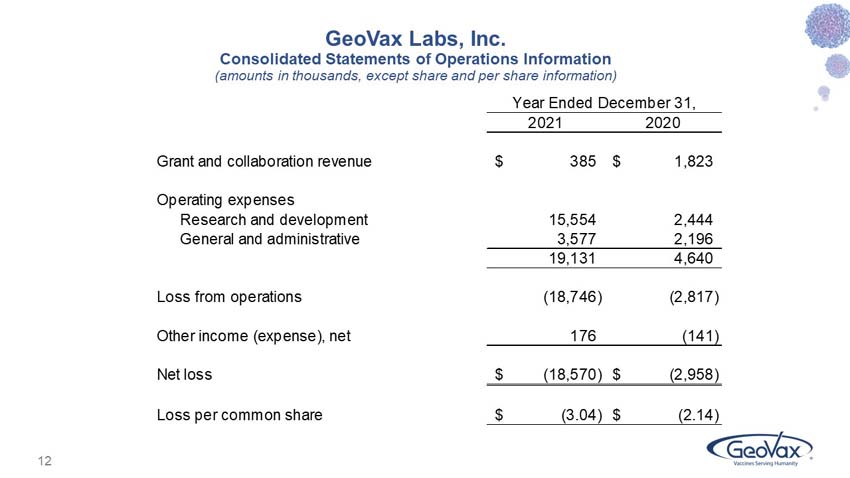
GeoVax reported a net loss for the year ended December 31, 2021 of $18.6 million, as compared to $3.0 million for the year ended December 31, 2020.
Grant and collaboration revenues were $0.4 million for 2021, as compared to $1.8 million in 2020. These amounts primarily relate to grants from NIAID for our Covid-19 vaccine program, and from the U.S. Department of Defense (DoD) for our Lassa Fever vaccine program. As of December 31, 2021, there were $81,526 of approved funds remaining and available for use related to GeoVax’s grant from the DoD.
Research and development (R&D) expenses were $15.6 million for 2021, as compared to $2.4 million in 2020. Contributing to the year-over-year increase in R&D expense were upfront payments and clinical trial expense reimbursements made pursuant to our in-license agreements with City of Hope and PNP Therapeutics, expenditures associated with our pan coronavirus vaccine program, manufacturing process development costs, and a generally higher level of activity.
General and administrative expenses were $3.6 million for 2021, as compared to $2.2 million in 2020, with the increase primarily attributable to higher Delaware franchise taxes; stock-based compensation expense; legal, accounting and patent costs; insurance costs; consulting fees; and investor relations costs.
Other income (expense) was $175,506 for 2021, as compared to $(141,253) in 2020. The 2021 amount includes a gain of $172,056 recorded upon the extinguishment of the Company’s PPP loan principal and accrued interest. The 2020 amount includes $138,851 of interest expense and amortized debt discount related to convertible debentures that were retired during 2020.
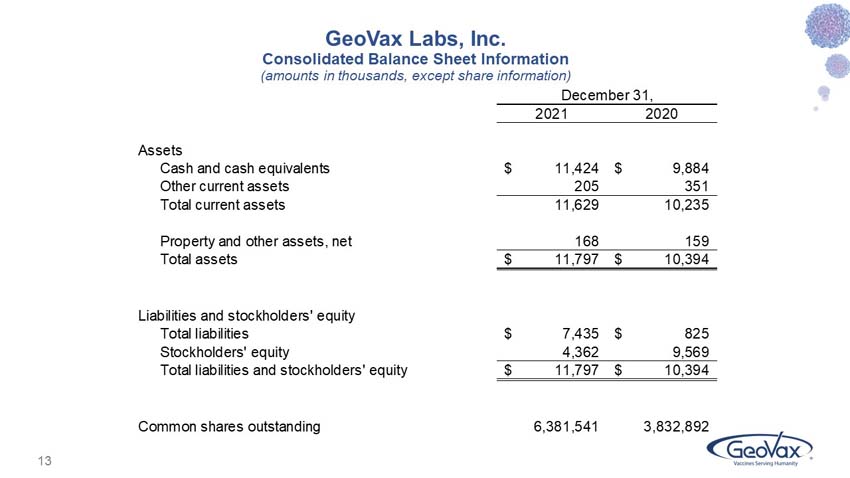
GeoVax reported cash balances of $11.4 million at December 31, 2021, as compared to $9.9 million at December 31, 2020. During the first quarter of 2022, the Company further supplemented its cash resources with net proceeds of $9.4 million from a private placement of its common stock and warrants.
Summarized financial information is attached. Further information concerning the Company’s financial position and results of operations are included in its Annual Report on Form 10-K filed with the Securities and Exchange Commission.
Conference Call
Management will host a conference call at 4:30 p.m. ET on Wednesday, March 9, 2022 to review financial results and provide an update on corporate developments. Following management’s formal remarks, there will be a question-and-answer session.
Participants are asked to pre-register for the call via the following link:
https://dpregister.com/sreg/10164185/f1aa6acaf6
Please note that registered participants will receive their dial-in number upon registration and will dial directly into the call without delay. Those without Internet access or who are unable to pre-register may dial in by calling 1-866-777-2509 (domestic) or 1-412-317-5413 (international). All callers should dial in approximately 10 minutes prior to the scheduled start time and ask to be joined into the GeoVax Labs call.
The conference call will be available through a live webcast found here:
https://services.choruscall.com/mediaframe/webcast.html?webcastid=liKBkNK2
MORE
A webcast replay of the call will be available via the same link as the live webcast approximately one hour after the end of the call through June 9, 2022. A telephonic replay of the call can be accessed by calling 1-877-344-7529 (domestic) or 1-412-317-0088 (international) and using access code 3389318. The telephonic replay will be available until March 23, 2022.
About GeoVax
GeoVax Labs, Inc. is a clinical-stage biotechnology company developing human vaccines and immunotherapies against infectious diseases and cancer using novel proprietary platforms. GeoVax’s product pipeline includes two ongoing Phase 2 clinical trials of GEO-CM04S1 (formerly COH04S1) for COVID-19 as a universal booster vaccine to mRNA vaccines authorized by the U.S. Food and Drug Administration (FDA) and as a primary vaccine for use in immunocompromised patients. In addition to GEO-CM04S1 for COVID-19, GeoVax is developing GEO-CM02 as a pan-coronavirus vaccine. The Company is also conducting a Phase 1/2 clinical trial of Gedeptin® for treatment of head and neck cancer. Gedeptin® has been granted orphan drug status by the FDA. Additional research and development programs include preventive vaccines against Zika Virus, hemorrhagic fever viruses (Ebola, Sudan, Marburg, and Lassa) and malaria, as well as immunotherapies for multiple solid tumors. The Company’s portfolio of wholly owned, co-owned, and in-licensed intellectual property stands at over 70 granted or pending patent applications spread over 20 patent families.
For additional information about GeoVax, visit our website: www.geovax.com.
Forward-Looking Statements
This release contains forward-looking statements regarding GeoVax’s business plans. The words “believe,” “look forward to,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “could,” “target,” “potential,” “is likely,” “will,” “expect” and similar expressions, as they relate to us, are intended to identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. Actual results may differ materially from those included in these statements due to a variety of factors, including whether: GeoVax is able to obtain acceptable results from ongoing or future clinical trials of its investigational products, GeoVax’s immuno-oncology products and preventive vaccines can provoke the desired responses, and those products or vaccines can be used effectively, GeoVax’s viral vector technology adequately amplifies immune responses to cancer antigens, GeoVax can develop and manufacture its immuno-oncology products and preventive vaccines with the desired characteristics in a timely manner, GeoVax’s immuno-oncology products and preventive vaccines will be safe for human use, GeoVax’s vaccines will effectively prevent targeted infections in humans, GeoVax’s immuno-oncology products and preventive vaccines will receive regulatory approvals necessary to be licensed and marketed, GeoVax raises required capital to complete development, there is development of competitive products that may be more effective or easier to use than GeoVax’s products, GeoVax will be able to enter into favorable manufacturing and distribution agreements, and other factors, over which GeoVax has no control.
Further information on our risk factors is contained in our registration statement on Form S-1 and in our periodic reports on Form 10-Q and Form 10-K that we have filed and will file with the SEC. Any forward-looking statement made by us herein speaks only as of the date on which it is made. Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We undertake no obligation to publicly update any forward-looking statement, whether as a result of new information, future developments or otherwise, except as may be required by U.S. federal securities law.
Contact:
GeoVax Labs, Inc.
investor@geovax.com
678-384-7220
FINANCIAL TABLES FOLLOW
MORE
GEOVAX LABS, INC.
Condensed Consolidated Statements of Operations Information
(amounts in thousands, except common share information)
|
Year Ended December 31, |
||||||||
|
2021 |
2020 |
|||||||
|
Grant and collaboration revenue |
$ | 385 | $ | 1,823 | ||||
|
Operating expenses: |
||||||||
|
Research and development |
15,554 | 2,444 | ||||||
|
General and administrative |
3,577 | 2,196 | ||||||
| 19,131 | 4,640 | |||||||
|
Loss from operations |
(18,746 | ) | (2,817 | ) | ||||
|
Other income (expense), net |
176 | (141 | ) | |||||
|
Net loss |
$ | (18,570 | ) | $ | (2,958 | ) | ||
|
Net loss per common share |
$ | (3.04 | ) | $ | (2.14 | ) | ||
|
Weighted average shares outstanding |
6,099,933 | 1,383,523 | ||||||
Condensed Consolidated Balance Sheet Information
(amounts in thousands, except common share information)
|
December 31, |
||||||||
|
2021 |
2020 |
|||||||
|
Assets: |
||||||||
|
Cash and cash equivalents |
$ | 11,424 | $ | 9,884 | ||||
|
Other current assets |
205 | 351 | ||||||
|
Total current assets |
11,629 | 10,235 | ||||||
|
Property and other assets |
168 | 159 | ||||||
|
Total assets |
$ | 11,797 | $ | 10,394 | ||||
|
Liabilities and stockholders’ equity |
||||||||
|
Total liabilities |
$ | 7,435 | $ | 825 | ||||
|
Stockholders’ equity |
4,362 | 9,569 | ||||||
|
Total liabilities and stockholders’ equity |
$ | 11,797 | $ | 10,394 | ||||
|
Common Shares Outstanding |
6,381,541 | 3,832,892 | ||||||
###