EX-99.2

FY2021 Q2 Results May 7, 2021 Exhibit
99.2

The Private Securities Litigation
Reform Act of 1995 provides a safe harbor from civil litigation for forward-looking statements accompanied by meaningful cautionary statements. Except for historical information, this presentation contains forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, which may be identified by words such as “continues”, “estimates”, “anticipates”,
“projects”, “plans”, “seeks”, “may”, “will”, “expects”, “intends”, “believes”, “signals”, “should”, “can”,
“guidance” and similar expressions or the negative versions thereof and which also may be identified by their context. All statements that address operating performance or events or developments that Meridian Bioscience, Inc.
(“Meridian” or “the Company”) expects or anticipates will occur in the future, including, but not limited to, statements relating to per share diluted net earnings, sales, product demand, net revenues, operating margin, other
guidance and the impact of COVID-19 on its business and prospects, are forward-looking statements. Such statements, whether expressed or implied, are based upon current expectations of the Company and speak only as of the date made. Specifically,
Meridian’s forward-looking statements are, and will be, based on management’s then-current views and assumptions regarding future events and operating performance. Meridian assumes no obligation to publicly update or revise any
forward-looking statements even if experience or future changes make it clear that any projected results expressed or implied therein will not be realized. These statements are subject to various risks, uncertainties and other factors that could
cause actual results to differ materially, including, without limitation, the following: Meridian’s operating results, financial condition and continued growth depends, in part, on its ability to introduce into the marketplace enhancements of
existing products or new products that incorporate technological advances, meet customer requirements and respond to products developed by Meridian’s competition, its ability to effectively sell such products and its ability to successfully
expand and effectively manage increased sales and marketing operations. While Meridian has introduced a number of internally developed products and acquired products, there can be no assurance that it will be successful in the future in introducing
such products on a timely basis or in protecting its intellectual property, and unexpected or costly manufacturing costs associated with its introduction of new products or acquired products could cause actual results to differ from expectations.
Meridian relies on proprietary, patented and licensed technologies. As such, the Company’s ability to protect its intellectual property rights, as well as the potential for intellectual property litigation, would impact its results. Ongoing
consolidations of reference laboratories and formation of multi-hospital alliances may cause adverse changes to pricing and distribution. Recessionary pressures on the economy and the markets in which the Company’s customers operate, as well
as adverse trends in buying patterns from customers, can change expected results. Costs and difficulties in complying with laws and regulations, including those administered by the United States Food and Drug Administration, can result in
unanticipated expenses and delays and interruptions to the sale of new and existing products, as can the uncertainty of regulatory approvals and the regulatory process (including the currently ongoing study and other FDA actions regarding the
Company’s LeadCare products). The international scope of Meridian’s operations, including changes in the relative strength or weakness of the U.S. dollar and general economic conditions in foreign countries, can impact results and make
them difficult to predict. One of Meridian’s growth strategies is the acquisition of companies and product lines. There can be no assurance that additional acquisitions will be consummated or that, if consummated, will be successful and the
acquired businesses will be successfully integrated into Meridian’s operations. There may be risks that acquisitions may disrupt operations and may pose potential difficulties in employee retention, and there may be additional risks with
respect to Meridian’s ability to recognize the benefits of acquisitions, including potential synergies and cost savings or the failure of acquisitions to achieve their plans and objectives. Meridian cannot predict the outcome of future
goodwill impairment testing and the impact of possible goodwill impairments on Meridian’s earnings and financial results. Meridian cannot predict the possible impact of U.S. health care legislation enacted in 2010 – the Patient
Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act – and any modification or repeal of any of the provisions thereof initiated by Congress or the presidential administration, and any similar
initiatives in other countries on its results of operations. Efforts to reduce the U.S. federal deficit, breaches of Meridian’s information technology systems, trade wars, increased tariffs, and natural disasters and other events could have a
materially adverse effect on Meridian’s results of operations and net revenues. The Company can make no assurances that a material weakness in its internal control over financial reporting will not be identified in the future, which if
identified and not properly corrected, could materially and adversely affect its operations and result in material misstatements in its consolidated financial statements. Meridian also is subject to risks and uncertainties related to disruptions to
or reductions in business operations or prospects due to pandemics, epidemics, widespread health emergencies, or outbreaks of infectious diseases such as COVID-19. In addition to the factors described in this paragraph, please also refer to
additional factors identified from time to time in the Company’s filings with the Securities and Exchange Commission, including in Part I, Item 1A Risk Factors of the Company’s most recent Annual Report on Form 10-K, which contains a
list and description of uncertainties, risks and other matters that may affect the Company. Readers should carefully review these forward-looking statements and risk factors, and not place undue reliance on the Company’s forward-looking
statements. Forward Looking Statements

Certain financial measures presented in
this presentation, such as operating expenses, operating income, operating margin, net earnings and net earnings per diluted share, each on an adjusted basis, excluding as applicable the effects of acquisition-related costs, changes in fair value of
acquisition consideration, restructuring costs and selected legal costs, are not recognized under United States generally accepted accounting principles, or GAAP. Management believes this non-GAAP financial information is useful to investors in
evaluating our performance, as these measures: (i) help investors to more meaningfully evaluate and compare the results of operations from period to period by removing the impacts of these non-routine items; and (ii) are used by management for
various purposes, including evaluating performance from period to period in presentations to our board of directors, and as a basis for strategic planning and forecasting. While we believe these financial measures are commonly used by investors to
evaluate our performance and that of our competitors, the non-GAAP measures in this presentation may be different from non-GAAP measures used by other companies and should not be considered as an alternative to performance measures derived in
accordance with GAAP. In addition, the non-GAAP measures presented herein are not based on any comprehensive set of accounting rules or principles. These non-GAAP measures have limitations, in that they do not reflect all amounts associated with our
results as determined in accordance with GAAP, and they should not be considered as alternatives to information attributable to Meridian Bioscience, Inc. determined in accordance with GAAP. See the consolidated financial statements included in our
reports filed with the U.S. Securities and Exchange Commission for our GAAP results. Additionally, for reconciliations of the non-GAAP measures included herein to our closest reported GAAP measures, refer to the reconciliations included in the press
release of Meridian Bioscience, Inc. dated May 7, 2021. Non-GAAP Financial Measures

Signed agreement for 2nd RADx grant -
$5.5M for ramping manufacturing of Revogene® SARS-CoV-2 Assay Submitted 510(k) for Curian ® Campy assay Launched first Air-Dryable sample specific Master Mix for Blood Q2 FY2021 Business Highlights Diagnostics Life Science
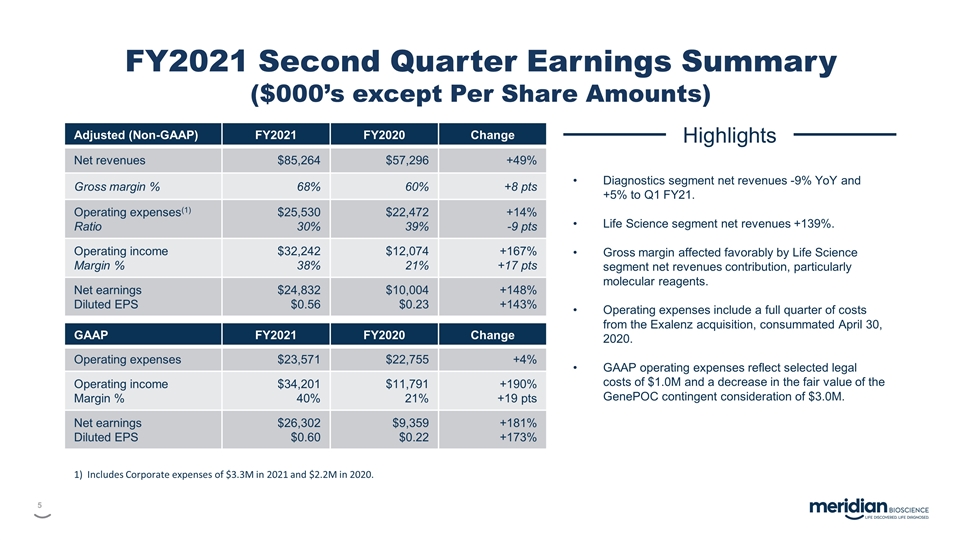
FY2021 Second Quarter Earnings Summary
($000’s except Per Share Amounts) Adjusted (Non-GAAP) FY2021 FY2020 Change Net revenues $85,264 $57,296 +49% Gross margin % 68% 60% +8 pts Operating expenses(1) Ratio $25,530 30% $22,472 39% +14% -9 pts Operating income Margin % $32,242 38%
$12,074 21% +167% +17 pts Net earnings Diluted EPS $24,832 $0.56 $10,004 $0.23 +148% +143% GAAP FY2021 FY2020 Change Operating expenses $23,571 $22,755 +4% Operating income Margin % $34,201 40% $11,791 21% +190% +19 pts Net earnings Diluted EPS
$26,302 $0.60 $9,359 $0.22 +181% +173% Highlights Diagnostics segment net revenues -9% YoY and +5% to Q1 FY21. Life Science segment net revenues +139%. Gross margin affected favorably by Life Science segment net revenues contribution, particularly
molecular reagents. Operating expenses include a full quarter of costs from the Exalenz acquisition, consummated April 30, 2020. GAAP operating expenses reflect selected legal costs of $1.0M and a decrease in the fair value of the GenePOC contingent
consideration of $3.0M. 1) Includes Corporate expenses of $3.3M in 2021 and $2.2M in 2020.
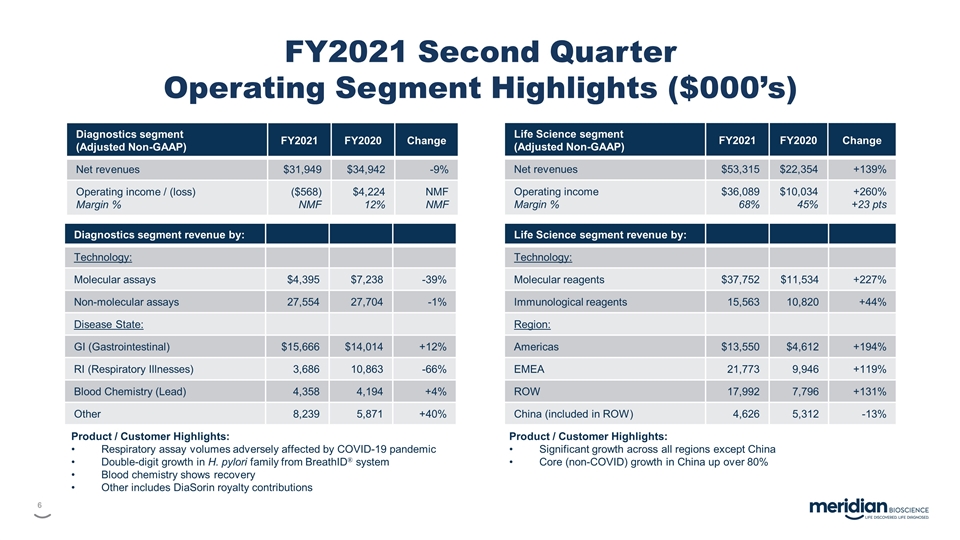
FY2021 Second Quarter Operating Segment
Highlights ($000’s) Diagnostics segment (Adjusted Non-GAAP) FY2021 FY2020 Change Net revenues $31,949 $34,942 -9% Operating income / (loss) Margin % ($568) NMF $4,224 12% NMF NMF Diagnostics segment revenue by: Technology: Molecular assays
$4,395 $7,238 -39% Non-molecular assays 27,554 27,704 -1% Disease State: GI (Gastrointestinal) $15,666 $14,014 +12% RI (Respiratory Illnesses) 3,686 10,863 -66% Blood Chemistry (Lead) 4,358 4,194 +4% Other 8,239 5,871 +40% Life Science segment
(Adjusted Non-GAAP) FY2021 FY2020 Change Net revenues $53,315 $22,354 +139% Operating income Margin % $36,089 68% $10,034 45% +260% +23 pts Life Science segment revenue by: Technology: Molecular reagents $37,752 $11,534 +227% Immunological reagents
15,563 10,820 +44% Region: Americas $13,550 $4,612 +194% EMEA 21,773 9,946 +119% ROW 17,992 7,796 +131% China (included in ROW) 4,626 5,312 -13% Product / Customer Highlights: Respiratory assay volumes adversely affected by COVID-19 pandemic
Double-digit growth in H. pylori family from BreathID® system Blood chemistry shows recovery Other includes DiaSorin royalty contributions Product / Customer Highlights: Significant growth across all regions except China Core (non-COVID) growth
in China up over 80%

FY2021 Fiscal Year Guidance Meridian
Bioscience Diagnostics Life Science Prior Guidance (2/5/2021) Updated FY2021 Guidance Meridian Bioscience Consolidated net revenues: $305 to $335 Million Adjusted operating margin: 30% to 33% Adjusted net earnings per share*: $1.60 to $1.80
Diagnostics Net revenues: $125 to $135 million Life Science Net revenues: $180 to $200 million Consolidated net revenues: $320 to $350 Million Adjusted operating margin: 31% to 33% Adjusted net earnings per share*: $1.70 to $1.90 Net revenues: $140
to $150 million Net revenues: $180 to $200 million * Assumes 44.3M diluted share count

Fluorescent analyzer Improved
performance with no subjectivity Intuitive user interface Intuitive, fast, and easy-to-use improves productivity Simple sample prep Clean, comfortable handling of samples Easy, standardized workflow Implement & train only once The Curian®
Campy Advantage

Contact:
mbi@meridianbioscience.com










