Cayman Islands | | | 2834 | | | Not Applicable |
(State or Other Jurisdiction of Incorporation or Organization) | | | (Primary Standard Industrial Classification Code Number) | | | (I.R.S. Employer Identification Number) |
Will H. Cai, Esq. Timothy Pitrelli, Esq. Cooley LLP c/o 35th Floor Two Exchange Square 8 Connaught Place Central, Hong Kong +852 3758-1200 | | | Reid S. Hooper, Esq. Cooley LLP 1299 Pennsylvania Avenue NW, Suite 700 Washington, DC 20004 (202) 842 7899 | | | Michael J. Blankenship Winston & Strawn LLP 800 Capitol Street, Suite 2400 Houston, Texas 77002 (713) 651 2600 |
† | The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification (“ASC”) after April 5, 2012. |

| | | Per ordinary share | | | Total | |
Public offering price | | | $ | | | $ |
Underwriting discounts and commissions(1) | | | $ | | | $ |
Proceeds, before expenses, to APRINOIA Therapeutics Inc. | | | $ | | | $ |
(1) | See “Underwriting” for additional disclosure regarding compensation payable by us to the underwriters. |
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
• | Tau & Tauopathies. Tau is an important protein in the brain that exists in different forms and plays a critical role in brain function. Tauopathies are neurodegenerative diseases characterized by accumulation of aggregated tau protein in distinct brain regions, such as Progressive Supranuclear Palsy (“PSP”), Alzheimer’s disease (“AD”) and Pick’s disease (“PiD”). Cumulative increases of these abnormal aggregates correlate with disease progression, and depending on the disorder can result in loss of memory, balance, walking and control of eye movements with the eventual loss of independent neurologic function. |
• | α-Syn & synucleinopathies. α-Syn is an important and highly abundant protein in the brain that regulates the release of neurochemicals between brain cells. Synucleinopathies are neurodegenerative diseases characterized by aggregation of abnormal α-Syn proteins. In synucleinopathies such as Parkinson’s disease (“PD”), Lewy Body Dementia (“LBD”) and Multiple System Atrophy (“MSA”), abnormal α-Syn aggregates accumulate in specific brain regions, and depending on the disorder can result in progressive loss of neurologic function, leading to problems with motor control, walking, balance, behavior and memory. |
• | PET diagnostic tracer APN-1607. We are developing APN-1607 as a PET imaging tracer for the detection of 3R and 4R tau aggregates, which contribute to the pathogenesis of various tauopathies, including PSP, |
• | Antibody platform and APNmAb005. APNmAb005 is a humanized anti-tau antibody we are developing for the treatment of AD, non-AD primary tauopathies including rare neurodegenerative disorders, such as PSP, cortico-basal degeneration (“CBD”) and behavioral variant Frontal Temporal Dementia (“FTD”) or its subcategory, Pick’s Disease (“PiD”). Unlike most other anti-tau antibodies currently in clinical development that bind to all forms of tau or phospho-tau (i.e., sites on tau protein that undergo phoshoprylation in disease state), APNmAb005 is designed to target a specific conformation epitope in the mid region that confers selectivity for misfolded tau oligomers/aggregates formed at axons/dendrites at early stages of disease that may contribute to disease progression. Based on existing clinical studies, we believe that blocking the pathological tau transmission has the potential to offer an effective treatment to slow down the disease progression for AD patients. While the recent approval of two anti-amyloid antibody treatments – aducanumab (Aduhelm®) and lecanemab (Leqembi®) – represents a significant advance in the field, their widespread use will be likely limited due to safety concerns arising from anti-amyloid imaging related abnormalities (“ARIA”). Furthermore, despite the dramatic reductions in amyloid pathology, suggested by recent studies, their efficacy in slowing cognitive decline in such studies was modest. These findings are consistent with the long-standing notion that although the accumulation of amyloid plays a critical role in the pathogenesis of AD, clearing amyloid alone is insufficient to completely block or prevent disease progression and argues for the discovery of other disease modifying targets such pathological forms of tau, which correlate with disease progression and cognitive decline. An IND for APNmAb005 was filed on February 24, 2022, and the FDA granted a Study May Proceed letter on April 20, 2022, for the Phase 1 trial to evaluate the safety of APNmAb005 in healthy volunteers. The first cohort of 8 subjects was dosed at 5mg/kg and the safety review was completed on August 18, 2023. There were no clinically significant safety findings. The study is currently active and not recruiting for the time being due to a reprioritization of resources. To accelerate development of APNmAb005, cohorts composed of patients with early AD and PSP will be dosed using a staggered parallel group design. Dosing is anticipated to resume in the fourth quarter of 2024. It is our intention to transition the study to evaluate single and |
• | Protein degrader platform and PROTAC degraders. Our proprietary PROTAC degrader programs for α-Syn and tau are our most innovative and cutting-edge platforms and have the potential to herald an entirely new class of drugs for the treatment of neurodegenerative disorders such as AD and PD. Our tau and α-Syn degrader programs are currently in the preclinical stages. Empowered by our knowledge of aggregated protein binder chemistry from our PET tracer programs we have generated a proprietary degrader library of 800+ compounds in α-Syn and tau degrader space. α-Syn degraders are identified from a cellular model of human dopaminergic neurons and the active degraders have been validated in animal models for selectivity, mechanism of action, physicochemical and metabolic properties. Based on our preclinical data disclosed in “Business — Our Differentiated Therapeutic Platforms and Pipeline — Our Therapeutic Product Candidates — α-Syn Degrader — Preclinical Results” and “Business — Our Differentiated Therapeutic Platforms and Pipeline — Our Therapeutic Product Candidates — Tau Degrader — Preclinical Results”, we believe that it would be feasible for this class of molecules to achieve reasonable brain penetration. Importantly, in preliminary studies we have observed significant reduction of pathological α-Syn in transgenic mice. Our tau degrader program is supported by Alzheimer’s Drug Discovery Foundation (“ADDF”) following their rigorous scientific review process. In the ADDF proposal, submitted to ADDF in December 2022, we described an experimental tau degrader RAC-1480, our lead compound at the time, which showed selective reduction of seed-induced tau aggregated formation in HEK293 cells and primary neurons. Intravenous injection of RAC-1480 at 25 mg/kg in rTg4510 tau transgenic mice reduced pathological tau species. The program has evolved rapidly since then and several new degraders have emerged with improved potency and ADME properties. TPD3 described in this document is one such improved tau degrader. The active compounds identified from our degrader library are highly selective for aggregated pathological tau while sparing the normal monomer tau. Similar to that with α-Syn degraders, we have conducted extensive work to validate the degrader mechanism and characterize metabolic and pharmacokinetic features. Both degrader programs are currently at lead optimization stages aiming for improved physicochemical properties, brain exposure and eventually robust in vivo efficacy by oral dosing. Our goal is to advance at least one degrader compound to IND-enabling GLP toxicology studies in 2025. |
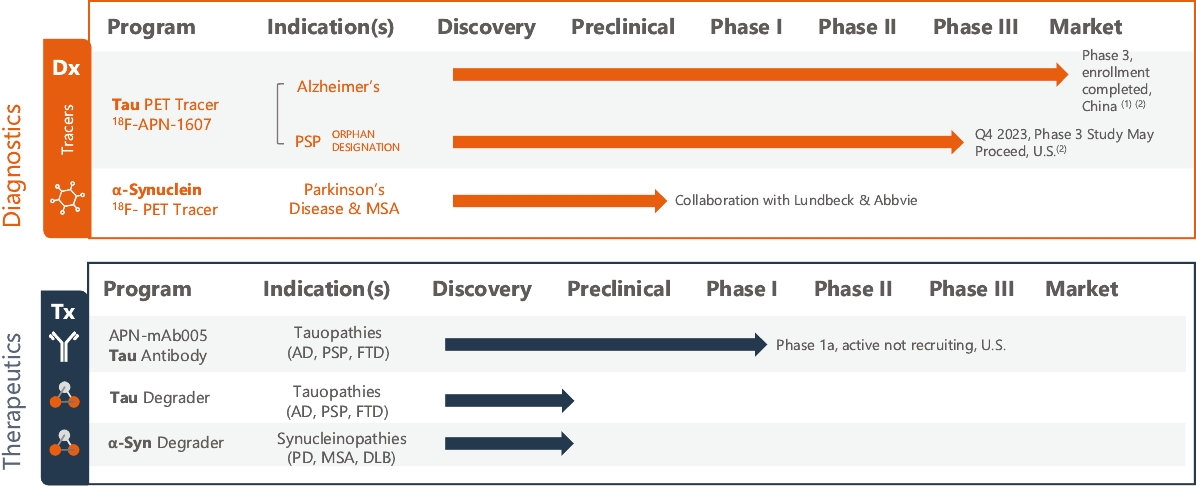
(1) | Phase 2 clinical trial of APN-1607 tau tracer for AD in the United States, Japan and Taiwan, is active non-recruiting. |
(2) | Our tau PET tracer APN-1607 improved upon a previously developed first generation compound from National Institute for Quantum Science and Technology (“QST”) in Japan. We obtained an exclusive worldwide license from QST for its patent for APN-1607 in 2016. We have an exclusive license for worldwide rights to develop and commercialize APN-1607, except for mainland China, where we granted an exclusive sublicense for its development, manufacture, marketing and distribution to Yantai Yitai Pharmaceutical Technology Co., Ltd. |
• | Our lead diagnostic product candidate (APN-1607). APN-1607 is our 3R/4R tau PET tracer and most clinically advanced diagnostic product candidate. APN-1607 is designed as a new generation tau PET tracer to achieve a higher specificity for the pathological tau aggregates. We believe that APN-1607, if approved, has the potential to be a powerful enabling tool for the diagnosis of various tauopathies, as it has shown low non-specific binding to other brain proteins, and the ability to detect different forms of tau in clinical studies. APN-1607 may therefore potentially be used in more precise diagnosis and stage classification of various tauopathies, including PSP, AD and PiD. |
• | Our lead therapeutic product candidate (APNmAb005). APNmAb005 is a humanized anti-tau antibody and our most clinically advanced therapeutic product candidate. APNmAb005 is designed to preferentially bind pathological tau aggregates, not normal tau, that accumulate at the neuronal synapses with disease. In addition, based on preclinical studies we conducted, APNmAb005 recognizes a three-dimensional conformation-dependent epitope that is only present in tau abnormal aggregates but not in normal tau protein, thus suggesting this product candidate may achieve a high level of selectivity for pathological forms of tau. |
• | Our lead therapeutic product candidate (Degrader). PROTACs offer a highly novel platform for the targeted degradation of toxic proteins that are causative in a number of neurodegenerative disorders as described above. These are bifunctional molecules that combine an active site selective for binding to the target of interest (tau or α-Syn) and a ligand (a binding site) for E3 ubiquitin ligase to drive the selective degradation of these proteins inside the cell’s proteasome. First-generation degraders are now entering clinical trials for cancer treatment, but currently there are no degraders in clinical development for neurodegenerative diseases. PROTAC is a potentially ground-breaking approach for the treatment of proteinopathies in neurodegenerative diseases that have been considered undruggable targets for traditional small molecules. Most of the PROTAC drugs currently in development are based on published E3 ligand structures, which leaves the warhead that targets the protein of interest as the key component that drives target specificity. It is noteworthy that the popular PROTAC targets in cancer are well characterized by soluble proteins such as CDK2, BTK4 and ER and in some cases allow for structure guided drug design. By contrast, the pathological proteins implicated in neurodegenerative diseases are more complex for warhead discovery as well as degrader screening. In this regard, we believe our PROTAC program is highly competitive in this field due to innovation in our PET ligand chemistry and expertise in disease biology which have enabled us to generate a diverse library to screen tau and α-Syn degraders and characterize them in cellular assays and animal models that recapitulate pathological α-Syn or tau formation. |
• | Develop novel solutions to overcome the challenges in diagnosing and treating neurodegenerative diseases. |
• | Continue to execute our versatile R&D and commercialization strategy to maximize asset value. |
• | Create product-by-product and region-by-region commercialization strategies in anticipation of our future product launches. |
• | Our founder and chairman of the board, Dr. Ming-Kuei Jang, Ph.D., has over 20 years of experience in neurodegenerative diseases. He also serves as the Chief Scientific Officer of APRINOIA USA and President of our Asia operations. Prior to founding our company, Dr. Jang held an associate director role at GlaxoSmithKline in Shanghai, served as senior research biologist of Merck & Co in Boston, Massachusetts, and led Neurodegeneration Consortium at MD Anderson Cancer Center in Houston, Texas. |
• | Our Chief Executive Officer, Dr. Mark S. Shearman, Ph.D., has extensive experience in pharmaceutical research, drug development and strategic partnerships. Prior to joining us, Dr. Shearman served as the Chief Scientific Officer at Editas Medicine, Chief Scientific Officer at Applied Genetic Technologies Corporation, a Senior Vice President of research and early development at Merck KGAa. He also served at Merck & Co., with his last position as an executive director, and Merck, Sharp & Dohme, with his last position as a senior director of department of cellular & molecular neuroscience responsible for the research and development of AD. |
• | Our Chief Medical Officer, Dr. Bradford A. Navia, M.D., Ph.D., has over 17 years of experience in clinical development (including Phase 1 through Phase 3), neuroimaging and biomarkers in psychiatry and neurology, including several INDs, sNDAs and an NDA. Prior to joining us, Dr. Navia was an Associate |
• | Our Chief Financial Officer, Brian Achenbach, M.B.A., has over 30 years of experience in finance and accounting primarily in the biotech, pharmaceutical and medical device industries. Prior to joining us, Mr. Achenbach served as Chief Financial Officer at On Demand Pharmaceuticals, Senior Vice President of Finance & Corporate Controller at Mustang Bio (Nasdaq: MBIO), and has held leadership positions in finance and accounting in multiple life sciences companies. |
• | Our General Counsel, Lana Gladstein, J.D., has over 23 years of experience in legal and pharmaceutical industry. Prior to joining us, Ms. Gladstein served as Chief Legal Officer and General Counsel of Arranta Bio (acquired by Recipharm), Chief Legal Officer of Recipharm (Americas) post acquisition of Arranta Bio, Executive VP and General Counsel at Brammer Bio (acquired by Thermo Fisher Scientific), and General Counsel of Viral Vector Services of Thermo Fisher Scientific post acquisition of Brammer Bio. Prior to that, Ms. Gladstein spent over 16 years in private practice, including as partner at Nutter McClennen & Fish LLP and Pepper Hamilton LLP, where she focused her practices in intellectual property litigation and strategies. |
• | We are a clinical-stage biotechnology company with a limited operating history and face significant challenges and expenses as we build our capabilities and develop our pipeline of diagnostic and therapeutic product candidates. |
• | We have incurred net losses since our inception and anticipate that we will continue to incur significant losses for the foreseeable future. We have never generated any revenue from product sales and may never be profitable. |
• | We recorded net cash outflow from operating activities since our inception. We may need to acquire funding from time to time to complete the development and commercialization of our pipeline candidates, which may not be available on acceptable terms, or at all. If we are unable to raise capital when needed, we may be forced to delay, reduce or eliminate certain of our product development programs or other operations. |
• | Our independent registered public accounting firm’s report contains an explanatory paragraph that expresses substantial doubt about our ability to continue as a “going concern.” |
• | Raising additional capital may cause dilution to the interests of our shareholders, restrict our operations or require us to relinquish rights to our technologies or product candidates. |
• | We depend heavily on the success of our lead diagnostic product candidate APN-1607, and, to a lesser extent, our anti-tau antibody product candidate, APNmAb005, and our degrader programs, all of which are currently or expected to be in clinical development. If our clinical trials are unsuccessful, we or our collaboration partner, Yantai Yitai Pharmaceutical Technology Co., Ltd. (“Yitai”), a wholly-owned subsidiary of Dongcheng Pharma, do not obtain regulatory approval in the targeted jurisdictions, or we or Yitai are unable to commercialize APN-1607, APNmAb005 or degraders, or experience significant delays in doing so, our business, our financial condition and results of operations will be materially adversely affected. |
• | We operate in highly competitive and rapidly changing industries. Our competitors are evaluating diagnostic product candidates in the same indication as our lead diagnostic product candidate, APN-1607, such as AD and PSP, and could enter the market with competing products of our product candidates, which may result in a material decline in sales of affected product candidates. |
• | A fast track, breakthrough therapy or other designation by the FDA may not actually lead to a faster development or regulatory review or approval process. |
• | Even if we successfully obtain regulatory approvals for our product candidates, they may not gain market acceptance, in which case we may not be able to generate product revenues, which will materially adversely affect our business, financial condition and results of operations. |
• | As a company with operations outside of the United States, our business is subject to economic, political, regulatory and other risks associated with international operations. |
• | Our future growth and ability to compete depend on retaining our key personnel and recruiting additional qualified personnel. |
• | We are a fast-growing emerging company and expect to expand our development and regulatory capabilities, and as a result, we may encounter difficulties in managing our growth, which could disrupt our operations. |
• | If we fail to maintain our relationships with our current or future business and licensing partners, our business, commercialization prospects and financial condition may be materially adversely affected. |
• | We may seek to form additional strategic alliances in the future with respect to our product candidates, and if we do not realize the benefits of such alliances, our business, financial condition, commercialization prospects and results of operations may be materially adversely affected. |
• | We rely on third parties to conduct our nonclinical studies and clinical trials and perform other tasks for us. If these third parties do not successfully carry out their contractual duties, meet expected deadlines, or comply with regulatory requirements, we may not be able to obtain regulatory approval for or commercialize our product candidates and our business could be substantially harmed. |
• | All material aspects of the research, development, manufacturing and commercialization of pharmaceutical products are heavily regulated, and we may face difficulties in complying with or be unable to comply with such regulations, which could have a material adverse effect on our business. |
• | The approval processes of regulatory authorities in the United States are lengthy, time-consuming and inherently unpredictable. If we are ultimately unable to obtain regulatory approval for our product candidates, our business will be substantially harmed. |
• | Even if we complete the necessary preclinical studies and clinical trials, the regulatory approval process is expensive, time-consuming and uncertain and may prevent us from obtaining approvals for the commercialization of some or all of our product candidates. As a result, we cannot predict when or if, and in which territories, we will obtain marketing approval to commercialize product candidates. |
• | Obtaining and maintaining regulatory approval of our product candidates in one jurisdiction does not mean that we will be successful in obtaining regulatory approval of our product candidates in other jurisdictions. |
• | If we are unable to establish sales, marketing and distribution capabilities for our product candidates, or enter into sales, marketing and distribution agreements with third parties, we may not be successful in commercializing our product candidates, if and when they are approved. |
• | The successful commercialization of our product candidates will depend in part on the extent to which governmental authorities and health insurers establish adequate coverage and reimbursement levels and pricing policies. |
• | We have never commercialized a product candidate before, which may make it difficult to evaluate the prospects for our future viability, and may lack the necessary expertise, personnel and resources to successfully commercialize our product candidates on our own or together with suitable partners. |
• | We may not have sufficient patent terms to effectively protect our future approved product candidates and business. |
• | If we or our collaboration partner are unable to obtain, maintain, defend and enforce patent and other intellectual property rights for our technologies and product candidates, or if the scope of the patent and other intellectual property rights obtained is not sufficiently broad, our competitors and other third parties could develop and commercialize technology and biologics similar or identical to ours, and our ability to successfully commercialize our technology and product candidates may be impaired. |
• | We or our collaboration partner may become subject to intellectual property-related litigation or other proceedings to protect or enforce our patents or the patents of our licensors or collaborators, any of which could be expensive, time-consuming, and unsuccessful, and may ultimately result in our loss of ownership of intellectual property. |
• | Business interruptions could seriously harm our future revenue and financial condition, increase our costs and expenses and delay us in the process of developing our product candidates. |
• | We may be subject to claims by third parties asserting that we or our employees, consultants or independent contractors have misappropriated, wrongfully used or disclosed their confidential information or trade secrets or other intellectual property or claiming ownership of what we regard as our own intellectual property. |
 as our wholly-owned subsidiary in Japan, and APRINOIA Therapeutics, LLC as our wholly-owned subsidiary in the United States. In December 2023, we incorporated APRINOIA Therapeutics Limited as our wholly-owned subsidiary in Ireland. Our principal executive offices are located at 245 Main Street, 2nd Floor, Cambridge, MA 02142. Our telephone number at this address is 617-225-4415. Our registered office is located at the offices of Maples Corporate Services Limited, PO Box 309, Ugland House, Grand Cayman, KY1-1104, Cayman Island. Investors should submit any inquiries to the address and telephone number of our principal executive offices set forth above.
as our wholly-owned subsidiary in Japan, and APRINOIA Therapeutics, LLC as our wholly-owned subsidiary in the United States. In December 2023, we incorporated APRINOIA Therapeutics Limited as our wholly-owned subsidiary in Ireland. Our principal executive offices are located at 245 Main Street, 2nd Floor, Cambridge, MA 02142. Our telephone number at this address is 617-225-4415. Our registered office is located at the offices of Maples Corporate Services Limited, PO Box 309, Ugland House, Grand Cayman, KY1-1104, Cayman Island. Investors should submit any inquiries to the address and telephone number of our principal executive offices set forth above. • | the sections of the Exchange Act regulating the solicitation of proxies, consents or authorizations in respect of a security registered under the Exchange Act; |
• | the sections of the Exchange Act requiring insiders to file public reports of their share ownership and trading activities and liability for insiders who profit from trades made in a short period of time; and |
• | the rules under the Exchange Act requiring the filing with the SEC of quarterly reports on Form 10-Q containing unaudited financial and other specified information, or current reports on Form 8-K, upon the occurrence of specified significant events. |
• | the requirement that a majority of the board of directors must be comprised of independent directors as defined in Nasdaq Rule 5605(a)(2); |
• | the requirement that each member of the compensation committee must be an independent director as set forth in Nasdaq Rule 5605(d)(2)(A); |
• | the requirement that director nomination should be made by a vote in which only independent directors participate or by a nominations committee comprised solely of independent directors as set forth in Nasdaq Rule 5605(e)(1); |
• | the requirement to obtain shareholder approval for certain issuances of securities, including shareholder approval of stock option plans; |
• | the requirement that the board of directors shall have regularly scheduled meetings at which only independent directors are present as set forth in Nasdaq Rule 5605(b)(2); and |
• | the requirement that an annual shareholders meeting must be held no later than one year after the end of the fiscal year-end as set forth in Nasdaq Rule 5620(a). |
• | is based on the (i) 23,528,707 ordinary shares issued and outstanding as of the date of this prospectus, which consists of (a) 10,123,057 ordinary shares issued and outstanding as of the date of this prospectus, |
• | excludes 3,456,098 ordinary shares issuable upon the exercise of options outstanding as of the date of this prospectus; and |
• | excludes 11,250 ordinary shares available for future issuance under our equity incentive plans upon the completion of this offering. |
• | a 1-for-4 reverse share split of our ordinary shares and preferred shares effected on , 2024; |
• | no exercise of the outstanding options described above; |
• | no exercise of the underwriters’ option to purchase additional ordinary shares; and |
• | the effectiveness of our amended and restated memorandum and articles of association, which will occur immediately prior to the completion of this offering. |
| | | For the six months ended June 30, | | | | | | | Year Ended December 31, | | ||||||||||||||
| | | 2023 | | | 2022 | | | Change | | | % | | | 2022 | | | 2021 | | | Change | | | % | |
| | | ($ in thousands, except percentages) | ||||||||||||||||||||||
Selected consolidated statements of operations: | | | ||||||||||||||||||||||
Revenue | | | $496 | | | $295 | | | $201 | | | 68 | | | $394 | | | $550 | | | $(156) | | | (28) |
Revenue – related party | | | 8,538 | | | — | | | 8,538 | | | — | | | — | | | — | | | — | | | — |
Total revenue | | | 9,034 | | | 295 | | | 8,739 | | | 2,962 | | | 394 | | | 550 | | | (156) | | | (28) |
Operating expenses | | | | | | | | | | | | | | | | | ||||||||
Research and development | | | 11,067 | | | 11,289 | | | (222) | | | (2) | | | 21,617 | | | 19,660 | | | 1,957 | | | 10 |
General and administrative | | | 4,704 | | | 2,849 | | | 1,855 | | | 65 | | | 7,041 | | | 4,787 | | | 2,254 | | | 47 |
Total operating expenses | | | 15,771 | | | 14,138 | | | 1,633 | | | 12 | | | 28,658 | | | 24,447 | | | 4,211 | | | 17 |
Loss from operations | | | (6,737) | | | (13,843) | | | 7,106 | | | (51) | | | (28,264) | | | (23,897) | | | (4,367) | | | 18 |
Other (expense) income: | | | | | | | | | | |||||||||||||||
Interest expense, net | | | (1,444) | | | (13) | | | (1,431) | | | 11,008 | | | (67) | | | (36) | | | (31) | | | 86 |
Change in fair value of derivative liabilities | | | 498 | | | — | | | 498 | | | — | | | — | | | — | | | — | | | — |
Other income (expense), net | | | 551 | | | 186 | | | 365 | | | 196 | | | 117 | | | 672 | | | (555) | | | (83) |
Total other income (expense), net | | | (395) | | | 173 | | | (568) | | | (328) | | | 50 | | | 636 | | | (586) | | | (92) |
Loss before income taxes | | | (7,132) | | | (13,670) | | | 6,538 | | | (48) | | | (28,214) | | | (23,261) | | | (4,953) | | | 21 |
Provision for income taxes | | | (53) | | | (6) | | | (47) | | | 783 | | | (17) | | | — | | | (17) | | | — |
Net loss | | | $(7,185) | | | $(13,676) | | | $6,491 | | | (47) | | | $(28,231) | | | $(23,261) | | | $(4,970) | | | 21 |
| | | As of June 30, | | | As of December 31, | ||||
| | | 2023 | | | 2022 | | | 2021 | |
| | | ($ in thousands) | |||||||
Selected consolidated balance sheets: | | | | | | | |||
Assets | | | | | | | |||
Current assets: | | | | | | | |||
Cash | | | $7,731 | | | $1,221 | | | $9,674 |
Prepaid expenses and other current assets | | | 1,162 | | | 590 | | | 429 |
Note receivable - related party | | | 660 | | | — | | | 155 |
Total current assets | | | 9,553 | | | 1,811 | | | 10,258 |
Property and equipment, net | | | 1,957 | | | 2,153 | | | 509 |
Deferred offering costs | | | 2,807 | | | 1,288 | | | — |
Operating lease right-of-use assets | | | 115 | | | 154 | | | 322 |
Prepaid expenses, net of current portion and other long-term assets | | | 44 | | | 237 | | | 1,677 |
Total assets | | | $14,476 | | | $5,643 | | | $12,766 |
| | | As of June 30, | | | As of December 31, | ||||
| | | 2023 | | | 2022 | | | 2021 | |
| | | ($ in thousands) | |||||||
| | | | | | | ||||
Liabilities, Redeemable Convertible Preferred Shares, and Shareholders' Deficit | | | | | | | |||
Current liabilities: | | | | | | | |||
Accounts payable | | | $7,318 | | | $8,887 | | | $432 |
Accrued expenses and other current liabilities | | | 4,940 | | | 2,466 | | | 2,993 |
Operating lease liabilities, current | | | 102 | | | 124 | | | 154 |
Related party payable | | | — | | | 904 | | | — |
Short-term borrowings | | | 690 | | | 1,450 | | | 785 |
Convertible notes (including related parties convertible notes of $10,762 and $753 as of June 30, 2023 and December 31, 2022, respectively, net of debt discount and issuance costs) | | | 14,788 | | | 1,093 | | | — |
Derivative liabilities (including related parties derivative liabilities of $1,685 and $173 as of June 30, 2023 and December 31, 2022, respectively) | | | 2,679 | | | 251 | | | — |
Total current liabilities | | | 30,517 | | | 15,175 | | | 4,364 |
Operating lease liabilities, net of current portion | | | 15 | | | 42 | | | 177 |
Total liabilities | | | 30,532 | | | 15,217 | | | 4,541 |
Commitments and Contingencies | | | | | | | |||
Redeemable convertible preferred shares (Series B, Pre-C and C), $0.1 par value; 56,973,336 shares authorized; 53,622,601 shares issued and outstanding; redemption and liquidation value of $66,166 as of June 30, 2023 and December 31, 2022 | | | 65,876 | | | 65,876 | | | 56,913 |
Shareholders’ deficit: | | | | | | | |||
Ordinary shares, $0.1 par value, 443,026,664 shares authorized; 40,492,206 and 38,617,056 shares issued and outstanding as of June 30, 2023 and December 31, 2022, respectively | | | 4,049 | | | 3,862 | | | 3,835 |
Additional paid-in capital | | | 13,176 | | | 12,296 | | | 10,373 |
Accumulated deficit | | | (97,823) | | | (90,638) | | | (62,407) |
Accumulated other comprehensive loss | | | (1,334) | | | (970) | | | (489) |
Total shareholders’ deficit | | | (81,932) | | | (75,450) | | | (48,688) |
Total liabilities, redeemable convertible preferred shares, and shareholders’ deficit | | | $14,476 | | | $5,643 | | | $12,766 |
| | | For the Six Months Ended June 30, | | | For the Year Ended December 31, | |||||||
| | | 2023 | | | 2022 | | | 2022 | | | 2021 | |
| | | ($ in thousands) | ||||||||||
Selected consolidated cash flow: | | | | | | | | | ||||
Net cash used in operating activities | | | $(5,451) | | | $(9,808) | | | $(17,237) | | | $(24,303) |
Net cash used in investing activities | | | (781) | | | (878) | | | (2,016) | | | (317) |
Net cash provided by financing activities | | | 13,287 | | | 9,002 | | | 11,354 | | | 31,492 |
Effect of exchange rates on cash | | | (545) | | | (303) | | | (554) | | | (60) |
Net increase (decrease) in cash | | | $6,510 | | | $(1,987) | | | $(8,453) | | | $6,812 |
• | continue our ongoing and planned research and development of our lead diagnostic product candidate, APN-1607; |
• | continue our ongoing and planned research and development of our pipeline of diagnostic and therapeutic product candidates, such as α-Syn PET tracers and APNmAb005; |
• | conduct preclinical studies and clinical trials for any additional product candidates that we may pursue in the future, including ongoing and planned development of additional diagnostic or therapeutic product candidates for the diagnosis or treatment of tauopathies, such as Alzheimer’s Disease (“AD”) and Progressive Supranuclear Palsy (“PSP”), and α-synucleinopathies, such as Parkinson’s Disease (“PD”) and Multiple System Atrophy (“MSA”); |
• | seek to discover and develop additional diagnostic and therapeutic product candidates and further expand our current pipeline; |
• | seek regulatory approvals for any product candidates that successfully complete clinical trials; |
• | meet the requirements for clinical trials and potential commercialization; |
• | establish sales, marketing and distribution infrastructure to commercialize any product candidate for which we may obtain regulatory approval; |
• | develop, maintain, expand and protect our intellectual property portfolio; |
• | add clinical, operational, financial and management information systems and/or personnel, including personnel to support our product development and planned future commercialization efforts; |
• | expand our operations in the United States, Japan, Taiwan and other geographic regions; and |
• | incur additional legal, accounting and other expenses associated with operating as a public company. |
• | the progress, results and costs of laboratory testing, manufacturing, and preclinical and clinical development for our current product candidates; |
• | the scope, progress, results and costs of preclinical development, laboratory testing and clinical trials of other diagnostic and/or therapeutic product candidates that we may pursue; |
• | the development requirements of other diagnostic and/or therapeutic product candidates that we may pursue; |
• | the timing and amounts of any milestone or royalty payments we may be required to make under future license agreements if we enter into such agreements; |
• | the costs of improving our research and development capacities and infrastructure, including hiring additional research and development, clinical, and quality control personnel; |
• | the costs, timing and outcome of regulatory review of our product candidates; |
• | the costs and timing of future commercialization activities, including product manufacturing, marketing, sales and distribution, for any of our product candidates for which we receive marketing approval; |
• | the amount of revenue, if any, received from commercial sales of our product candidates for which we receive marketing approval; |
• | the costs and timing of preparing, filing and prosecuting patent applications, obtaining, maintaining, protecting and enforcing our intellectual property rights and defending against any intellectual property-related claims; |
• | the costs associated with operating as a public company; and |
• | the extent to which we acquire or in-license other product candidates and technologies. |
• | completing clinical trials that demonstrate the efficacy and safety of our product candidates; |
• | receiving marketing approvals from applicable regulatory authorities; |
• | obtaining commercial manufacturing capabilities; |
• | launching commercial sales, marketing and distribution operations; |
• | acceptance of our diagnostic and therapeutic product candidates by patients, the medical community and/or third-party payors, if such product candidates are approved; |
• | a continued acceptable safety profile following approval; |
• | competing effectively with other diagnostics or therapies; and |
• | qualifying for, obtaining, maintaining, enforcing and defending our intellectual property rights and claims and not infringing on third parties’ intellectual property rights. |
• | the accuracy of diagnosis for which our diagnostic product candidates are approved; |
• | the clinical indications for which our product candidates are approved; |
• | the accuracy and specificity to the pathological protein aggregates of our PET tracer candidates; |
• | hospitals and medical imaging centers establishing the infrastructure required, such as the PET scanners, for the administration of PET scan; |
• | the cost of our PET tracer candidates and the cost of administrating PET scan in relation to alternative diagnostic methods; |
• | the cost of product candidates in relation to alternative therapies; |
• | physicians, hospitals, and patients considering our product candidates as safe and effective therapies; |
• | the potential and perceived advantages of our product candidates over alternative therapies; |
• | the prevalence and severity of any side effects; |
• | product labeling or product insert requirements of the FDA or other regulatory authorities; |
• | limitations or warnings contained in the labeling approved by the FDA or other regulatory authorities; |
• | the timing of market introduction of our product candidates as well as competitive products; |
• | the amount of upfront costs or training required for physicians to administer our product candidates; |
• | the availability of coverage, adequate reimbursement, and pricing by third-party payors and government authorities; |
• | the willingness of patients to pay out-of-pocket in the absence of comprehensive coverage and reimbursement by third-party payors and government authorities; |
• | relative convenience and ease of administration of our diagnostics, including as compared to alternative diagnostic methods and competitive diagnostics; |
• | relative convenience and ease of administration of our product candidates including as compared to alternative treatments and therapies; and |
• | the effectiveness of our sales and marketing efforts and distribution support. |
• | be delayed in obtaining marketing approval for our product candidates; |
• | not obtain marketing approval; |
• | obtain approval for indications or patient populations that are not as broad as intended or desired; |
• | obtain approval with labeling that includes significant use or distribution restrictions or significant safety warnings, including boxed warnings; |
• | be subject to additional post-marketing testing or other requirements; or |
• | remove the product from the market after obtaining marketing approval. |
• | the patient eligibility criteria defined in the protocol; |
• | the number of patients with the disease or condition being studied; |
• | the understanding of risks and benefits of the product candidate in the clinical trial; |
• | clinicians’ and patients’ perceptions as to the potential advantages of the product candidate being studied in relation to other available diagnostic methods or therapeutics, including any diagnostics that may be approved to diagnose the indications or new therapeutics that may be approved for the indications we are investigating or therapeutics that may be used off-label for these indications; |
• | the size and nature of the patient population who meet inclusion criteria; |
• | the proximity of patients to study sites; |
• | sufficient supply of PET tracer by third-party suppliers, including contract manufacturing organizations (“CMOs”); |
• | the design of the clinical trial; |
• | competing clinical trials for similar or other new diagnostics or therapeutics for tauopathies and α-synucleinopathies; and |
• | our ability to obtain and maintain patient consents. |
• | regulatory authorities may withdraw approvals of such products and require us to take any approved products off the market; |
• | regulatory authorities may require the addition of labeling statements, specific warnings, a contraindication or field alerts to physicians and pharmacies; |
• | we may be required to create a medication guide outlining the risks of such side effects and make it available to patients; |
• | we may be required to change the way the product is administered, conduct additional studies or change the labeling of such products; |
• | we may be subject to limitations in how we promote such products; |
• | sales of the product may decrease substantially; |
• | we could be sued and held liable for any harm that such products caused to patients; and |
• | our reputation and physician or patient acceptance of our products may suffer. |
• | the FDA or other regulatory authorities may disagree as to the number, design or implementation of our clinical trials, or may not interpret the results from clinical trials as we do; |
• | regulators or institutional review boards may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site; |
• | we may not reach an agreement on acceptable terms with prospective clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different clinical trial sites; |
• | clinical trials of our product candidates may produce negative or inconclusive results; |
• | we may decide, or regulators may require us, to conduct additional clinical trials or abandon product development programs; |
• | the number of patients required for clinical trials of our product candidates may be larger than we anticipate, enrollment in these clinical trials may be slower than we anticipate, participants may drop out of these clinical trials at a higher rate than we anticipate or we may fail to recruit eligible patients to participate in a trial; |
• | our third-party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us promptly, or at all; |
• | regulators may issue a clinical hold, or regulators or institutional review boards may require that we or our investigators suspend or terminate clinical research for various reasons, including non-compliance with regulatory requirements or a finding that the participants are being exposed to unacceptable health risks; |
• | the cost of clinical trials of our product candidates may be greater than we anticipate; |
• | the FDA or other regulatory authorities may fail to approve our manufacturing processes or facilities; |
• | the supply or quality of our product candidates or other materials necessary to conduct clinical trials of our product candidates may be insufficient or inadequate; |
• | our product candidates may have undesirable side effects or other unexpected characteristics, leading to possible suspension or termination of the clinical trials; and |
• | the approval policies or regulations of the FDA or other regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval. |
• | we may not be successful in identifying additional product candidates; |
• | we may not be able or willing to assemble sufficient resources to acquire or discover additional product candidates; |
• | our product candidates may not succeed in preclinical or clinical studies; |
• | a product candidate may on further study be shown to have harmful side effects or other characteristics that indicate it is unlikely to be effective or otherwise does not meet applicable regulatory criteria; |
• | competitors may develop alternatives that render our product candidates obsolete or less attractive; |
• | product candidates we develop may nevertheless be covered by third parties’ patents or other exclusive rights; |
• | the market for a product candidate may change during our development program so that the continued development of that product candidate is no longer reasonable; |
• | a product candidate may not be capable of being produced in commercial quantities at an acceptable cost, or at all; and |
• | a product candidate may not be accepted as safe and effective by patients, the medical community or third-party payors, if applicable. |
• | economic weakness, including inflation, or political instability in particular non-U.S. economics and markets; |
• | changes in a specific country’s or region’s political or economic environment; |
• | different and changing regulatory requirements for product approvals in different jurisdictions; |
• | differing reimbursement regimes and price controls in certain non-U.S. markets; |
• | potentially reduced protection for intellectual property rights; |
• | different issues for securing, maintaining or obtaining freedom to operate presented in different jurisdictions; |
• | difficulties to comply with different, complex and changing laws, regulations and court systems of multiple jurisdictions and a wide variety of foreign laws, treaties and regulations, as well as potential negative consequences from any non-compliance of such laws, treaties and regulations; |
• | potential negative consequences from changes in tax laws; compliance with tax, employment, immigration and labor laws for employees living or traveling abroad, including, for example, the variable tax treatment in different jurisdictions of options granted under our share incentive plans; |
• | litigation or administrative actions resulting from claims against us by current or former employees or consultants individually or as part of class actions, including claims of wrongful terminations, discrimination, misclassification or other violations of labor law or other alleged conduct; |
• | changes in non-U.S. regulations and customs, tariffs and trade barriers; |
• | risks relating foreign exchange and currency controls; |
• | trade protection measures, import or export licensing requirements or other restrictive actions by governments; |
• | transportation or supply chain interruption due to cancellation of public transport or restrictions on logistics; |
• | governments imposed lockdowns such as stringent quarantine measures and mandate temporary shutdown of business operations; |
• | production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad; |
• | difficulties associated with staffing and managing international operations, including differing labor relations; |
• | workforce uncertainty in countries where labor unrest is more common than in the United States; and |
• | business interruptions resulting from geo-political actions, including war and terrorism, health epidemics, or natural disasters including earthquakes, typhoons, floods and fires. |
• | identifying, recruiting, integrating, maintaining and motivating additional employees; |
• | managing our internal development efforts effectively, including the clinical, FDA review processes for our product candidates; and |
• | improving our operational, financial and management controls, reporting systems and procedures. |
• | disruptions to our business and clinical activities caused by potential future governments-imposed lockdowns such as stringent quarantine measures, mandate of temporary shutdown of business operations, limitation in patient enrollment, disruptions to patient follow-up, and curtailed screening visits, delays or difficulties in enrolling patients; |
• | interruption to supply chain due to cancellation of public transport or restrictions on logistics; |
• | delays in clinical sites receiving the supplies and materials needed to conduct our clinical trials; |
• | diversion of healthcare resources away from the conduct of clinical trials, including the diversion of hospitals serving as our clinical trial sites and hospital staff supporting the conduct of our clinical trials; |
• | interruption of key clinical trial activities, such as clinical trial site monitoring, due to limitations on travel imposed or recommended by governments, employers and others; |
• | restrictions on employee resources that would otherwise be focused on the conduct of our clinical trials, including because of sickness of employees or their families or the desire of employees to avoid contact with crowds; |
• | delays in receiving approval from local regulatory authorities to initiate our planned clinical trials; |
• | interruption in global shipping that may affect the transport of clinical trial materials; |
• | changes in local regulations as part of a response to the health pandemics or epidemics which may require us to change how our clinical trials are conducted, which may result in unexpected costs, or to pause the clinical trials altogether; |
• | delays in necessary interactions with local regulators, ethics committees and other important agencies and contractors due to limitations in employee resources or forced furlough of government employees; and |
• | refusal of the relevant regulatory authorities to accept data from clinical trials in health pandemics or epidemics affected geographic regions. |
• | we may not be able to control the amount and timing of resources that our collaboration partner chooses to dedicate to the development of such product candidates; |
• | the collaboration partner may itself experience financial difficulties and disrupt our collaboration efforts; |
• | we may be required to grant or otherwise relinquish important rights such as marketing, distribution and intellectual property rights; |
• | a collaboration partner could move forward with a competing product developed either independently or in collaboration with third parties, including our competitors; or |
• | business combinations or significant changes in a collaboration partner’s business strategy may adversely affect our willingness to complete our obligations under any arrangement. |
• | increased operating expenses and cash requirements; |
• | the assumption of additional indebtedness or contingent liabilities; |
• | assimilation of operations, intellectual property and products of an acquired company, including difficulties associated with integrating new personnel; |
• | the diversion of our management’s attention from our existing programs and initiatives in pursuing such a strategic partnership, merger or acquisition; |
• | retention of key employees, the loss of key personnel and uncertainties in our ability to maintain key business relationships; |
• | risks and uncertainties associated with the other party to such a transaction, including the prospects of that party and their existing products or product candidates and regulatory approvals; and |
• | our inability to generate revenue from acquired technology sufficient to meet our objectives in undertaking the acquisition or even to offset the associated acquisition and maintenance costs. |
• | failure to begin or complete clinical trials due to disagreements with regulatory authorities; |
• | failure to demonstrate that a product candidate is safe and effective; |
• | failure of clinical trial results to meet the level of statistical significance required for approval; |
• | reporting or data integrity issues related to our clinical trials; |
• | disagreement with our interpretation of data from preclinical studies or clinical trials; |
• | changes in approval policies or regulations that render our preclinical and clinical data insufficient for approval or require us to amend our clinical trial protocols; |
• | regulatory requests for additional analyses, reports, data, nonclinical studies and clinical trials, or questions regarding interpretations of data and results and the emergence of new information regarding our product candidates or other products; |
• | failure to satisfy regulatory conditions regarding endpoints, patient population, available therapies and other requirements for our clinical trials in order to support marketing approval on an accelerated basis or at all; |
• | a delay in or the inability of health authorities to complete regulatory inspections of our development activities, regulatory filings or manufacturing operations, whether as a result of the COVID-19 pandemic or other reasons, or our failure to satisfactorily complete such inspections; |
• | our failure to conduct a clinical trial in accordance with regulatory requirements or our clinical trial protocols; and |
• | clinical sites, investigators or other participants in our clinical trials deviating from a trial protocol, failing to conduct the trial in accordance with regulatory requirements, or dropping out of a trial. |
• | litigation involving patients taking such products; |
• | restrictions on such products, manufacturers or manufacturing processes; |
• | restrictions on the labeling or marketing of such product; |
• | restrictions on product distribution or use; |
• | requirements to conduct post-marketing studies or clinical trials; |
• | warning or untitled letters; |
• | withdrawal of such products from the market; |
• | refusal to approve pending applications or supplements to approved applications that we submit; |
• | recall of products; |
• | fines, restitution or disgorgement of profits or revenue; |
• | suspension or withdrawal of marketing approvals; |
• | suspension of any ongoing clinical trials; |
• | damage to relationships with any potential collaborators; |
• | unfavorable press coverage and damage to our reputation; |
• | refusal to permit the import or export of our products; |
• | product seizure; or |
• | injunctions or the imposition of civil or criminal penalties. |
• | the U.S. federal Anti-Kickback Statute, which prohibits, among other things, persons from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, lease, order or recommendation of, any good, facility, item or service, for which payment may be made, in whole or in part, under federal and state healthcare programs such as Medicare and Medicaid. The term “remuneration” has been broadly interpreted to include anything of value. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers and formulary managers on the other hand. Although there are several statutory exceptions and regulatory safe harbors protecting certain common activities from prosecution or other regulatory sanctions, the exceptions and safe harbors are drawn narrowly, and practices that involve remuneration that are alleged to be intended to induce prescribing, purchases or recommendations may be subject to scrutiny if they do not qualify for an exception or safe harbor. Failure to meet all of the requirements of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the federal Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review of all its facts and circumstances. Several courts have interpreted the statute’s intent requirement to mean that if any one purpose of an arrangement involving remuneration is to induce referrals of federal healthcare covered business, the federal Anti-Kickback Statute has been violated; |
• | U.S. federal civil and criminal false claims laws, including the federal False Claims Act, which can be enforced through civil whistleblower or qui tam actions, and civil monetary penalty laws, which, among other things, impose criminal and civil penalties, against individuals or entities for, among other things, knowingly presenting, or causing to be presented, to the federal government, including the Medicare and Medicaid programs, claims for payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government. Pharmaceutical and other healthcare companies have been prosecuted under these laws for, among other things, allegedly inflating drug prices they report to pricing services, which in turn were used by the government to set Medicare and Medicaid reimbursement rates, and for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. In addition, certain marketing practices, including off-label promotion, may also violate false claims laws. Further, pharmaceutical manufacturers can be held liable under the False Claims Act even when they do not submit claims directly to government payors if they are deemed to “cause” the submission of false or fraudulent claims. Criminal prosecution is also possible for making or presenting a false, fictitious or fraudulent claim to the federal government; |
• | HIPAA, which contains new federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of whether the payor is public or private, knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters; |
• | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (“HITECH”) and their respective implementing regulations, which impose obligations on “covered entities,” including certain healthcare providers, health plans, and healthcare clearinghouses, as well as their respective “business associates “that create, receive, maintain or transmit individually identifiable health information for or on behalf of a covered entity, with respect to safeguarding the privacy, security and transmission of individually identifiable health information. Additionally, HITECH also contains four new tiers of civil monetary penalties; amends HIPAA to make civil and criminal penalties directly applicable to business associates and gave state attorneys general new authority to file civil actions for damages or injunctions in U.S. federal courts to enforce the federal HIPAA laws and to seek attorneys’ fees and costs associated with pursuing federal civil actions; |
• | the U.S. federal Food, Drug and Cosmetic Act, which prohibits, among other things, the adulteration or misbranding of drugs, biologics and medical devices; |
• | the U.S. federal Physician Payments Sunshine Act, created under Section 6002 of the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, or collectively, the ACA, and its implementing regulations, created annual reporting requirements for certain manufacturers of drugs, devices, biologicals and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions), to report information related for certain payments and “transfers of value” provided to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members; and analogous state laws and regulations and foreign laws, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers; state and foreign laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government or to adopt compliance programs as prescribed by state laws and regulations, or that otherwise restrict payments that may be made to healthcare providers; state and foreign laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers, marketing expenditures or drug pricing; state and local laws that require the registration of pharmaceutical sales representatives; and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
• | it takes time and resources to negotiate and execute sales, marketing or distribution agreements and we may not end up with an agreement being signed and may delay the development or commercialization of the affected product candidate; |
• | sales, marketing or distribution agreements are subject to cancellation or nonrenewal by our collaborators, or may not be fully complied with by our collaborators; |
• | in the case of a license granted by us, we lose control of the development of the product candidate licensed; |
• | in such cases we would have only limited control over the means and resources allocated by our partner for the sales, marketing or distribution of our future approved product; and |
• | collaborators may not properly obtain, maintain, enforce, or defend our intellectual property or proprietary rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our proprietary information or expose us to potential litigation. |
• | reduced resources of our management to pursue our business strategy; |
• | decreased demand for any product candidates or products that we may develop; |
• | injury to our reputation and significant negative media attention; |
• | withdrawal of clinical trial participants; |
• | initiation of investigations by regulators; |
• | product recalls, withdrawals or labeling, marketing or promotional restrictions; |
• | significant costs to defend the resulting litigation; |
• | substantial monetary awards paid to clinical trial participants or patients; |
• | loss of revenue; and |
• | the inability to commercialize any products that we may develop. |
• | changes in the industries in which we operate; |
• | developments involving our competitors; |
• | changes in laws and regulations affecting our business; |
• | variations in our operating performance and the performance of its competitors in general; |
• | actual or anticipated fluctuations in our quarterly or annual operating results; |
• | publication of research reports by securities analysts about us or our competitors or our industry; |
• | the public’s reaction to our press releases, its other public announcements and its filings with the SEC; |
• | actions by holders in respect of any of their ordinary shares; |
• | additions and departures of key personnel; |
• | commencement of, or involvement in, litigation involving us; |
• | changes in our capital structure, such as future issuances of securities or the incurrence of debt; |
• | the volume of our ordinary shares available for public sale; and |
• | general economic and political conditions, recessions, volatility in the markets, interest rates, local and national elections, fuel prices, international currency fluctuations, corruption, political instability, and acts of war or terrorism. |
• | our ability to execute our strategies and develop our pipeline; |
• | our ability to obtain regulatory approvals for our product candidates and gain market acceptance; |
• | our ability to generate revenue from product sales and become profitable in the future; |
• | our ability to develop and protect intellectual property; |
• | our ability to properly manage a public company; |
• | our manufacturing, commercialization, and marketing capabilities and strategy; |
• | our plans relating to commercializing our product candidates, if approved, including the geographic areas of focus and sales strategy; |
• | the need to hire additional personnel and our ability to attract and retain such personnel; |
• | the size of the market opportunity for our product candidates, including our estimates of the number of patients who suffer from the diseases we are targeting; |
• | our ability to implement measures to address the material weakness that has been identified; |
• | our competitive position and the success of competing therapies that are or may become available; |
• | our estimates regarding expenses, future revenue, capital requirements and needs for additional financing; |
• | our financial performance; |
• | the period over which we estimate our existing cash and cash equivalents will be sufficient to fund our future operating expenses and capital expenditure requirements; |
• | the impact of laws and regulations; |
• | our ability to obtain, and negotiate favorable terms of, any collaboration, licensing or other arrangements that may be necessary or desirable to develop, manufacture or commercialize our product candidates; |
• | our expectations regarding the period during which we will qualify as an emerging growth company under the JOBS Act; and |
• | our anticipated use of our existing resources and the proceeds from this offering. |
• | 30% of the net proceeds to advance the clinical development of our lead diagnostic product candidate, APN-1607, including the initiation of a phase 3 clinical trial in the second half of 2024 in the United States, Europe, Japan and Taiwan and Asia for patients clinically suspected to have Progressive Supranuclear Palsy (“PSP”); |
• | 10% of the net proceeds to advance the clinical development of our lead therapeutic product candidate, APNmAb005, including the advancement of our ongoing phase 1 clinical trial in the United States; |
• | 10% of the net proceeds to advance the preclinical development of our lead protein degraders toward IND-enabling studies; and |
• | the remainder to fund other research and development activities, working capital requirements and general corporate purposes. |
• | on an actual basis; |
• | on a pro forma basis to reflect (i) the conversion of all of our issued and outstanding convertible preferred shares into 13,405,650 ordinary shares on a one-for-one basis upon the completion of this offering; and (ii) the conversion of all of our issued convertible promissory notes and associated interest into 2,300,119 ordinary shares upon the completion of this offering based on 80% of an assumed initial public offering price of $12.00 per ordinary share, which is the midpoint of the price range set forth on the cover page of this prospectus; and |
• | on a pro forma as adjusted basis to reflect (i) the pro forma adjustments set forth above, and (ii) the sale of 2,000,000 ordinary shares by us in this offering at an assumed initial public offering price of $12.00 per ordinary share, which is the midpoint of the estimated range of the initial public offering price shown on the front cover of this prospectus, after deducting the underwriting discounts and commissions and estimated offering expenses payable by us, assuming the underwriters do not exercise their option to purchase additional ordinary shares. |
| | | As of June 30, 2023 | |||||||
| | | Actual (unaudited) | | | Pro Forma (1) | | | Pro Forma As Adjusted (1)(2) | |
| | | (Amounts in thousands, except share and per share data) | |||||||
Cash | | | $7,731 | | | $12,181 | | | $32,651 |
Convertible notes (including related parties convertible notes of $10,762, net of debt discount and issuance costs) | | | $ 14,788 | | | $ — | | | $— |
Derivative liabilities (including related parties derivative liabilities of $1,685) | | | 2,679 | | | — | | | — |
Redeemable convertible preferred shares (Series B, Pre-C and C), $0.4 par value; 14,243,334 shares authorized; 13,405,650 shares issued and outstanding, actual; no shares authorized, issued and outstanding, pro forma and pro forma as adjusted | | | 65,876 | | | — | | | — |
Shareholders' equity (deficit): | | | | | | | |||
Ordinary shares, $0.4 par value, 110,756,666 shares authorized; 10,123,057 shares issued and outstanding, actual; $0.4 par value, 110,756,666 shares authorized, 25,828,826 shares issued and outstanding, pro forma; and $0.4 par value, 110,756,666 shares authorized, 27,828,826 shares issued and outstanding, pro forma as adjusted | | | 4,049 | | | 10,331 | | | 11,131 |
Additional paid-in capital | | | 13,176 | | | 94,851 | | | 114,521 |
Accumulated deficit | | | (97,823) | | | (97,987) | | | (100,678) |
Accumulated other comprehensive loss | | | (1,334) | | | (1,334) | | | (1,334) |
Total shareholders' equity (deficit) | | | (81,932) | | | 5,861 | | | 23,640 |
Total capitalization | | | $1,411 | | | $5,861 | | | $23,640 |
(1) | The unaudited pro forma and pro forma as adjusted information does not include the impact of share-based compensation expense for share options which we expect to record upon the completion of this offering. |
(2) | The pro forma as adjusted information discussed above is illustrative only. Our additional paid-in capital and total shareholders’ equity (deficit) following the completion of this offering are subject to adjustment based on the actual initial public offering price and other terms of this offering determined at pricing. A $1.00 increase (decrease) in the assumed initial public offering price of $12.00 per ordinary share, the midpoint of the estimated range of the initial public offering price shown on the cover page of this prospectus, would increase (decrease) |
• | 3,292,493 ordinary shares issuable upon exercise of share options outstanding as of June 30, 2023, at a weighted average exercise price of $0.62 per share; and |
• | 274,855 ordinary shares reserved for future issuance under our Equity Incentive Plan #4, or the 2022 Plan as of June 30, 2023. |
| | | Per Ordinary Share | |
Assumed initial public offering price per ordinary share | | | $12.00 |
Net tangible book value per ordinary share | | | $(8.37) |
Pro forma net tangible book value per ordinary share after giving effect to the pro forma adjustments described above | | | $0.12 |
Pro forma net tangible book value per ordinary share as adjusted to give effect to the pro forma adjustments described above, and this offering | | | $0.85 |
Amount of dilution in net tangible book value per ordinary share to new investors in the offering | | | $11.15 |
| | | Ordinary Shares Purchased | | | Total Consideration | | | Average Price Per Ordinary Share | |||||||
| | | Number | | | Percent | | | Amount | | | Percent | | |||
(in thousands, except share, per share and percent data) | | | | | | | | | | | |||||
Existing shareholders | | | 25,828,826 | | | 92.8% | | | $101,335 | | | 80.9% | | | $3.92 |
New investors | | | 2,000,000 | | | 7.2% | | | $24,000 | | | 19.1% | | | $12.00 |
Total | | | 27,828,826 | | | 100.0% | | | $125,335 | | | 100.0% | | | |
• | political and economic stability; |
• | an effective judicial system; |
• | tax neutrality; |
• | the absence of exchange control or currency restrictions; and |
• | the availability of professional and support services. |
• | the Cayman Islands has a less-developed body of securities laws as compared to the United States and these securities laws provide significantly less protection to investors as compared to those of the United States; and |
• | Cayman Islands companies may not have standing to sue before the federal courts of the United States. |
• | continue our ongoing and planned clinical research and development of our lead diagnostic product candidate, APN-1607, in the United States for the diagnosis of AD and PSP; |
• | continue our ongoing and planned preclinical studies and clinical research and development of our other diagnostic and therapeutic product candidates, including our lead therapeutic product candidate, APNmAb005, and Tau and α-Syn degrader candidates; |
• | continue our other ongoing and planned discovery and research and development activities; |
• | seek to discover and develop additional product candidates and further expand our clinical product pipeline; |
• | seek regulatory approvals for any product candidates that successfully complete clinical trials; |
• | establish sales, marketing and distribution infrastructure to commercialize any product candidate for which we may obtain regulatory approval; |
• | develop, maintain, expand and protect our intellectual property portfolio; |
• | hire additional research, clinical, quality control, and administrative personnel; |
• | expand our operations globally; and |
• | incur additional legal, accounting, investor relations, insurance and other expenses associated with operating as a public company following the completion of this offering. |
• | expenses incurred under agreements with organizations that support our drug discovery and development activities; |
• | expenses incurred in connection with the preclinical and clinical development of our product candidates and programs; |
• | costs related to contract research organizations (“CROs”) and contract development and manufacturing organizations (“CDMOs”), that are primarily engaged to provide drug substance and product for our clinical trials, research and development programs, as well as investigative sites and consultants that conduct our clinical trials, nonclinical studies and other scientific development services; |
• | costs of acquiring and manufacturing nonclinical and clinical trial materials, including manufacturing registration and validation batches; |
• | employee-related expenses, including salaries, related benefits and equity-based compensation expense, for employees engaged in research and development functions; |
• | costs related to compliance with quality and regulatory requirements; |
• | payments made under third-party licensing agreements; and |
• | direct and allocated costs related to facilities, information technology, personnel and other overhead. |
• | per patient trial costs; |
• | the number of patients that participate in the trials; |
• | the number of sites included in the trials; |
• | the countries in which the trials are conducted; |
• | the length of time required to enroll eligible patients; |
• | the drop-out or discontinuation rates of patients; |
• | potential additional safety monitoring or other studies requested by regulatory agencies; |
• | the duration of patient follow-up; |
• | the efficacy and safety profile of the product candidates; |
• | the number of trials required for regulatory approval; |
• | the receipt of regulatory approvals from applicable regulatory authorities; |
• | the timing, receipt and terms of any marketing approvals from applicable regulatory authorities; and |
• | the extent to which we establish collaboration, licensing or similar arrangements and the performance of any related third parties. |
| | | For the six months ended June 30, | | | | | | | Year Ended December 31, | | ||||||||||||||
| | | 2023 | | | 2022 | | | Change | | | % | | | 2022 | | | 2021 | | | Change | | | % | |
| | | ($ in thousands, except percentages) | ||||||||||||||||||||||
Revenue | | | $496 | | | $295 | | | $201 | | | 68 | | | $394 | | | $550 | | | $(156) | | | (28) |
Revenue - related party | | | 8,538 | | | — | | | 8,538 | | | — | | | — | | | — | | | — | | | — |
Total revenue | | | 9,034 | | | 295 | | | 8,739 | | | 2,962 | | | 394 | | | 550 | | | (156) | | | (28) |
Operating expenses | | | | | | | | | | |||||||||||||||
Research and development | | | 11,067 | | | 11,289 | | | (222) | | | (2) | | | 21,617 | | | 19,660 | | | 1,957 | | | 10 |
General and administrative | | | 4,704 | | | 2,849 | | | 1,855 | | | 65 | | | 7,041 | | | 4,787 | | | 2,254 | | | 47 |
Total operating expenses | | | 15,771 | | | 14,138 | | | 1,633 | | | 12 | | | 28,658 | | | 24,447 | | | 4,211 | | | 17 |
Loss from operations | | | (6,737) | | | (13,843) | | | 7,106 | | | (51) | | | (28,264) | | | (23,897) | | | (4,367) | | | 18 |
Other (expense) income: | | | | | | | | | | | | | | | | | ||||||||
Interest expense, net | | | (1,444) | | | (13) | | | (1,431) | | | 11,008 | | | (67) | | | (36) | | | (31) | | | 86 |
Change in fair value of derivative liabilities | | | 498 | | | — | | | 498 | | | — | | | — | | | — | | | — | | | — |
Other income (expense), net | | | 551 | | | 186 | | | 365 | | | 196 | | | 117 | | | 672 | | | (555) | | | (83) |
Total other income (expense), net | | | (395) | | | 173 | | | (568) | | | (328) | | | 50 | | | 636 | | | (586) | | | (92) |
| | | For the six months ended June 30, | | | | | | | Year Ended December 31, | | ||||||||||||||
| | | 2023 | | | 2022 | | | Change | | | % | | | 2022 | | | 2021 | | | Change | | | % | |
| | | ($ in thousands, except percentages) | ||||||||||||||||||||||
Loss before income taxes | | | (7,132) | | | (13,670) | | | 6,538 | | | (48) | | | (28,214) | | | (23,261) | | | (4,953) | | | 21 |
Provision for income taxes | | | (53) | | | (6) | | | (47) | | | 783 | | | (17) | | | — | | | (17) | | | — |
Net loss | | | $(7,185) | | | $(13,676) | | | $6,491 | | | (47) | | | $(28,231) | | | $(23,261) | | | $(4,970) | | | 21 |
| | | For the six months ended June 30, | | | | | ||||||
| | | 2023 | | | 2022 | | | Change | | | % | |
| | | ($ in thousands, except percentages) | ||||||||||
Outsourced research services | | | $5,344 | | | $7,423 | | | $(2,079) | | | (28) |
Personnel expenses (including share-based compensation) | | | 3,426 | | | 2,346 | | | 1,080 | | | 46 |
Facilities and lab supplies | | | 907 | | | 533 | | | 374 | | | 70 |
Legal, professional and consulting fees | | | 494 | | | 674 | | | (180) | | | (27) |
Other expenses | | | 896 | | | 313 | | | 583 | | | 186 |
| | | $11,067 | | | $11,289 | | | $(222) | | | (2) | |
| | | For the six months ended June 30, | | | | | ||||||
| | | 2023 | | | 2022 | | | Change | | | % | |
| | | ($ in thousands, except percentages) | ||||||||||
Personnel expenses (including share-based compensation) | | | $2,550 | | | $1,021 | | | $1,529 | | | 150 |
Legal, professional and consulting fees | | | 1,585 | | | 1,432 | | | 153 | | | 11 |
Facilities and lab supplies | | | 246 | | | 241 | | | 5 | | | 2 |
Other expenses | | | 323 | | | 155 | | | 168 | | | 108 |
| | | $4,704 | | | $2,849 | | | $1,855 | | | 65 | |
| | | For the Year Ended December 31, | | | | | ||||||
| | | 2022 | | | 2021 | | | Change | | | % | |
| | | ($ in thousands, except percentages) | ||||||||||
Outsourced research services | | | $12,583 | | | $14,940 | | | $(2,357) | | | (16) |
Personnel expenses (including share-based compensation) | | | 6,072 | | | 2,384 | | | 3,688 | | | 155 |
Facilities and lab supplies | | | 1,332 | | | 936 | | | 396 | | | 42 |
Legal, professional and consulting fees | | | 893 | | | 735 | | | 158 | | | 21 |
Other expenses | | | 737 | | | 665 | | | 72 | | | 11 |
| | | $21,617 | | | $19,660 | | | $1,957 | | | 10 | |
| | | For the Year Ended December 31, | | | | | ||||||
| | | 2022 | | | 2021 | | | Change | | | % | |
| | | ($ in thousands, except percentages) | ||||||||||
Personnel expenses (including share-based compensation) | | | $3,549 | | | $2,056 | | | $1,493 | | | 73 |
Legal, professional and consulting fees | | | 2,709 | | | 2,011 | | | 698 | | | 35 |
Facilities and lab supplies | | | 412 | | | 237 | | | 175 | | | 74 |
Other expenses | | | 371 | | | 483 | | | (112) | | | (23) |
| | | $7,041 | | | $4,787 | | | $2,254 | | | 47 | |
• | the timing, receipt and amount of sales of any future approved or cleared products, if any; |
• | the scope, progress, results and costs of researching and developing our existing product candidates or any future product candidates, and conducting preclinical studies and clinical trials; |
• | the timing of, and the costs involved in, obtaining regulatory approvals or clearances for our existing product candidates or any future product candidates; |
• | the time and costs involved in obtaining regulatory approval for our product candidates and any delays we may encounter as a result of evolving regulatory requirements or adverse results with respect to any of these product candidates; |
• | the number and characteristics of any additional product candidates we develop or acquire; |
• | the cost of manufacturing our product candidates and any products we successfully commercialize, including costs associated with developing our manufacturing capabilities; |
• | the costs of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending intellectual property-related claims; |
• | the extent to which we acquire or in-license other product candidates and technologies; |
• | our ability to establish and maintain strategic collaborations, licensing or other arrangements and the financial terms of any such agreements that we may enter into; |
• | the expenses needed to attract and retain skilled personnel and senior management; and |
• | the costs associated with being a public company. |
| | | For the Six Months Ended June 30, | | | For the Year Ended December 31, | |||||||
| | | 2023 | | | 2022 | | | 2022 | | | 2021 | |
| | | ($ in thousands) | ||||||||||
Net cash used in operating activities | | | $(5,451) | | | $(9,808) | | | $(17,237) | | | $(24,303) |
Net cash used in investing activities | | | (781) | | | (878) | | | (2,016) | | | (317) |
Net cash provided by financing activities | | | 13,287 | | | 9,002 | | | 11,354 | | | 31,492 |
Effect of exchange rates on cash | | | (545) | | | (303) | | | (554) | | | (60) |
Net increase (decrease) in cash | | | $6,510 | | | $(1,987) | | | $(8,453) | | | $6,812 |
• | Tau & Tauopathies. Tau is an important protein in the brain that exists in different forms and plays a critical role in brain function. Tauopathies are neurodegenerative diseases characterized by accumulation of aggregated tau protein in distinct brain regions, such as Progressive Supranuclear Palsy (“PSP”), Alzheimer’s disease (“AD”) and Pick’s disease (“PiD”). Cumulative increases of these abnormal aggregates correlate with disease progression, and depending on the disorder can result in loss of memory, balance, walking and control of eye movements with the eventual loss of independent neurologic function. |
• | α-Syn & synucleinopathies. α-Syn is an important and highly abundant protein in the brain that regulates the release of neurochemicals between brain cells. Synucleinopathies are neurodegenerative diseases characterized by aggregation of abnormal α-Syn proteins. In synucleinopathies such as Parkinson’s disease (“PD”), Lewy Body Dementia (“LBD”) and Multiple System Atrophy (“MSA”), abnormal α-Syn aggregates accumulate in specific brain regions, and depending on the disorder can result in progressive loss of neurologic function, leading to problems with motor control, walking, balance, behavior and memory. |
• | PET diagnostic tracer APN-1607. We are developing APN-1607 as a PET imaging tracer for the detection of 3R and 4R tau aggregates, which contribute to the pathogenesis of various tauopathies, including PSP, a rare neurodegenerative disease. Based on Title 21 of the Code of Federal Regulations, Part 315, Diagnostics Radiopharmaceuticals, we seek two indication claims for APN-1607: (1) as a pathological or disease marker of 4R tau in PSP and (2) as clinically useful marker for the diagnostic management in patients who present with parkinsonian syndromes in whom the diagnosis of PSP is uncertain or requires confirmation. We have received an Orphan Drug Designation (“ODD”) from the U.S. Food and Drug Administration (the “FDA”) in 2017 for APN-1607 as a diagnostic agent for PSP. Under the U.S. Orphan Drug Act, the FDA may grant ODDs to drugs or biologics intended to treat a “rare disease or condition” |
• | Antibody platform and APNmAb005. APNmAb005 is a humanized anti-tau antibody we are developing for the treatment of AD, non-AD primary tauopathies including rare neurodegenerative disorders, such as PSP, cortico-basal degeneration (“CBD”) and behavioral variant Frontal Temporal Dementia (“FTD”) or its subcategory, Pick’s Disease (“PiD”). Unlike most other anti-tau antibodies currently in clinical development that bind to all forms of tau or phospho-tau (i.e., sites on tau protein that undergo phoshoprylation in disease state), APNmAb005 is designed to target a specific conformation epitope in the mid region that confers selectivity for misfolded tau oligomers/aggregates formed at axons/dendrites at early stages of disease that may contribute to disease progression. Based on existing clinical studies, we believe blocking the pathological tau transmission has the potential to offer an effective treatment to slow down the disease progression for AD patients. While the recent approval of two anti-amyloid antibody treatments – aducanumab (Aduhelm®) and lecanemab (Leqembi®) – represents a significant advance in the field, their widespread use will be likely limited due to safety concerns arising from anti-amyloid imaging related abnormalities (“ARIA”). Furthermore, despite the dramatic reductions in amyloid pathology, suggested by recent studies, their efficacy in slowing cognitive decline in such studies was modest. These findings are consistent with the long-standing notion that although the accumulation of amyloid plays a critical role in the pathogenesis of AD, clearing amyloid alone is insufficient to completely block or prevent disease progression and argues for the discovery of other disease modifying targets such pathological forms of tau, which correlate with disease progression and cognitive decline. An IND for the APNmAb005 program was filed on February 24, 2022, and the FDA granted a Study May Proceed letter on April 20, 2022 for the Phase 1 trial to evaluate the safety of APNmAb005 in healthy volunteers. The first cohort of 8 subjects was dosed at 5mg/kg and the safety review was completed on August 18, 2023. There were no clinically significant safety findings. The study is currently active and not recruiting for the time being due to a reprioritization of resources. To accelerate development of APNmAb005, cohorts composed of patients with early AD and PSP will be dosed using a staggered parallel group design. Dosing is anticipated to resume in the fourth quarter of 2024. It is our intention to transition the study to evaluate single and multiple dosing in patients, in order to continue to evaluate safety and tolerability at higher doses but also to acquire exploratory biomarker data. We will also seek a partnership to advance this program beyond Phase 1 into potential proof-of-concept clinical studies. |
• | Protein degrader platform and PROTAC degraders. Our proprietary PROTAC degrader programs for α-Syn and tau are our most innovative and cutting-edge platforms and have the potential to herald an entirely new class of drugs for the treatment of neurodegenerative disorders such as AD and PD. Our tau and α-Syn degrader programs are currently in the preclinical stages. Empowered by our knowledge of aggregated protein binder chemistry from our PET tracer programs we have generated a proprietary degrader library of 800+ compounds in α-Syn and tau degrader space. α-Syn degraders are identified from |
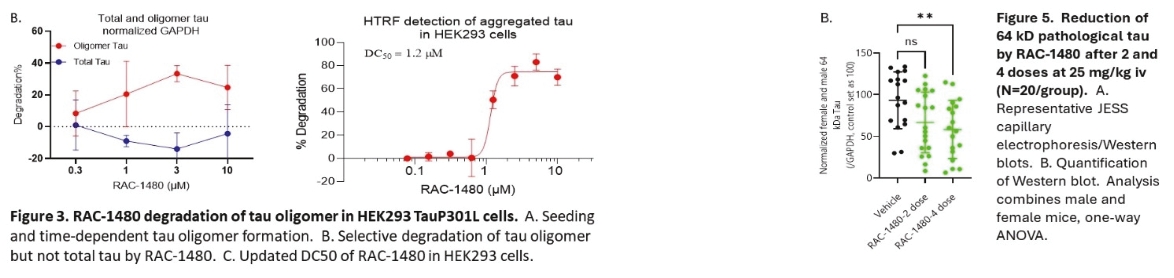

(1) | Phase 2 clinical trial of APN-1607 tau tracer for AD in the United States, Japan and Taiwan, is active non-recruiting. |
(2) | Our tau PET tracer APN-1607 improved upon a previously developed first generation compound from National Institute for Quantum Science and Technology ("QST") in Japan. We obtained an exclusive worldwide license from QST for its patent for APN-1607 in 2016. We have an exclusive license for worldwide rights to develop and commercialize APN-1607, except for mainland China, where we granted an exclusive sublicense for its development, manufacture, marketing and distribution to Yantai Yitai Pharmaceutical Technology Co., Ltd. |
• | Our lead diagnostic product candidate (APN-1607). APN-1607 is our 3R/4R tau PET tracer and most clinically advanced diagnostic product candidate. APN-1607 is designed as a new generation tau PET tracer to achieve a higher specificity for the pathological tau aggregates. We believe that APN-1607, if approved, has the potential to be a powerful enabling tool for the diagnosis of various tauopathies, as it has shown low non-specific binding to other brain proteins, and the ability to detect different forms of tau in clinical studies. APN-1607 may therefore potentially be used in more precise diagnosis and stage classification of various tauopathies, including PSP, AD and PiD. |
• | Our lead therapeutic product candidate (APNmAb005). APNmAb005 is a humanized anti-tau antibody and our most clinically advanced therapeutic product candidate. APNmAb005 is designed to preferentially bind pathological tau aggregates, not normal tau, that accumulate at the neuronal synapses with disease. In addition, based on preclinical studies we conducted, APNmAb005 recognizes a three-dimensional conformation-dependent epitope that is only present in tau abnormal aggregates but not in normal tau protein, thus suggesting this product candidate may achieve a high level of selectivity for pathological forms of tau. |
• | Our lead therapeutic product candidate (Degrader). PROTACs offer a highly novel platform for the targeted degradation of toxic proteins that are causative in a number of neurodegenerative disorders as described above. These are bifunctional molecules that combine an active site selective for binding to the |
• | Our founder and chairman of the board, Dr. Ming-Kuei Jang, Ph.D., has over 20 years of experience in neurodegenerative diseases. He also serves as the Chief Scientific Officer of APRINOIA USA and President of our Asia operations. Prior to founding the Company, Dr. Jang held an associate director role at GlaxoSmithKline in Shanghai, served as senior research biologist of Merck & Co in Boston, Massachusetts, and led Neurodegeneration Consortium at MD Anderson Cancer Center in Houston, Texas. |
• | Our Chief Executive Officer, Dr. Mark S. Shearman, Ph.D., has extensive experience in pharmaceutical research, drug development and strategic partnerships. Prior to joining us, Dr. Shearman served as the Chief Scientific Officer at Editas Medicine, Chief Scientific Officer at Applied Genetic Technologies Corporation, a Senior Vice President of research and early development at Merck KGAa. He also served at Merck & Co., with his last position as an executive director, and Merck, Sharp & Dohme, with his last position as a senior director of department of cellular & molecular neuroscience responsible for the research and development of AD. |
• | Our Chief Medical Officer, Dr. Bradford A. Navia, M.D., Ph.D., has over 17 years of experience in clinical development (including Phase 1 through Phase 3), neuroimaging and biomarkers in psychiatry and neurology, including several INDs, sNDAs and an NDA. Prior to joining us, Dr. Navia was an Associate Professor of Neurology and Psychiatry at Tufts Medical School, and the recipient of numerous awards and funding from National Institute of Health; executive director of Sunovion Pharmaceuticals, where he was the global project lead for the development of KYNMOBI; senior director, strategic and clinical lead in the neuroscience division at AbbVie Inc.; senior director and head of neuroimaging at Eisai Co., Ltd. and director in the neuroscience clinical development division at Johnson & Johnson. |
• | Our Chief Financial Officer, Brian Achenbach, M.B.A., has over 30 years of experience in finance and accounting primarily in the biotech, pharmaceutical and medical device industries. Prior to joining us, Mr. Achenbach served as Chief Financial Officer at On Demand Pharmaceuticals, Senior Vice President of Finance & Corporate Controller at Mustang Bio (Nasdaq: MBIO), and has held leadership positions in finance and accounting in multiple life sciences companies. |
• | Our General Counsel, Lana Gladstein, J.D., has over 23 years of experience in legal and pharmaceutical industry. Prior to joining us, Ms. Gladstein served as Chief Legal Officer and General Counsel of Arranta |
• | Develop novel solutions to overcome the challenges in diagnosing and treating neurodegenerative diseases. We aim to develop solutions to overcome past failures in drug development by other companies in central nervous system (“CNS”) related clinical trials. These failures were attributable to one or more reasons, including wrong targets, molecules, patients, drug doses, and treatment timing or duration. We will focus our discovery and research capabilities on tau and α-Syn protein aggregates. We aim to leverage our unique PET tracers and protein binders, which specifically recognize tau and α-Syn in pathological aggregates instead of their normal forms, to develop and commercialize our diagnostic and therapeutic assets. |
• | Continue to execute our versatile R&D and commercialization strategy to maximize asset value. We plan to seek collaboration opportunities with leading pharmaceutical and biotechnology companies to develop and commercialize our product candidates at different stages of clinical development. We expect this strategy to provide us with the flexibility to optimize the value of our product pipeline and the ability to generate cash inflow with a higher degree of certainty prior to drug commercialization. |
○ | Diagnostic programs strategy. We have adopted different strategies for our early- and late-stage PET assets. We formed early research collaborations with pharmaceutical companies to support the development of our early-stage programs, such as α-Syn PET tracers. For our late-stage clinical programs, we plan to provide non-exclusive licenses to pharmaceutical companies while retaining the rights to fund our clinical trials to NDA to allow us to capture the full commercialization value. |
○ | Therapeutic programs strategy. Our clinical development strategy is to start with rare diseases attributable to pathological mechanisms involving tau or α-Syn such as PSP, PiD, and MSA, and fund the clinical trials to NDA through internal resources. For more prevalent diseases, such as AD and PD, which will require larger and more complex trials, we may consider forming partnerships with other biotechnology and pharmaceutical companies to help fund those clinical trials. In both situations, we will leverage our proprietary PET tracer, APN-1607, for patient selection and monitoring treatment response, thereby improving the probability of success of these trials. |
• | Create product-by-product and region-by-region commercialization strategies in anticipation of our future product launches. We will evaluate commercialization strategies on a product-by-product and region-by-region basis in order to maximize the value of our future approved products. We will take into consideration a matrix of factors, including capital investment necessary to execute on each option, manufacturing and distribution partners and infrastructure available in each market, availabilities of sales and marketing specialists, size of the market, competition, and availability of pharmaceutical and biotechnology partners to form a tailored commercialization strategy for each product candidate. |
• | PSP. PSP is a rare neurodegenerative disorder with a prevalence ranging from 1.39 to 17.3 affected individuals per 100,000 people. The characteristic signs of PSP are postural instability, falls, disturbances controlling in eye movements, cognitive or behavioral changes. The condition gradually worsens until death, an average of seven years after onset of symptoms. Reliable diagnosis, particularly during the early stages of disease, remains a major clinical challenge and currently a definitive diagnosis can be made only by postmortem examination. There is a significant unmet need to identify biomarkers for the diagnosis of PSP and related tauopathies, particularly during the early stages of the disease. |
• | AD. AD is the most common cause of dementia. AD and neurodegenerative diseases affected approximately 50 million people worldwide in 2020 and are expected to affect 139 million people worldwide by 2050. In the United States, approximately 6.5 million people have AD and the number could reach 12 million by 2050. AD is projected to cost the United States more than $379 billion in economic burden annually by 2040. Approximately 90% of physicians in the United States believe that early diagnosis of AD is critical to be able to delay the progression of the disease. Regardless, many physicians remain uncomfortable adopting early diagnosis given the scarcity of reliable diagnostic tools for early diagnosis, underscoring the unmet need to identify more precise diagnostics. |
• | PD. PD is the most prevalent movement disorder in the elderly and affects approximately 10 million people worldwide in 2020 and the number is growing with an aging population. Currently, PD affects about 1 million people in the United States. |
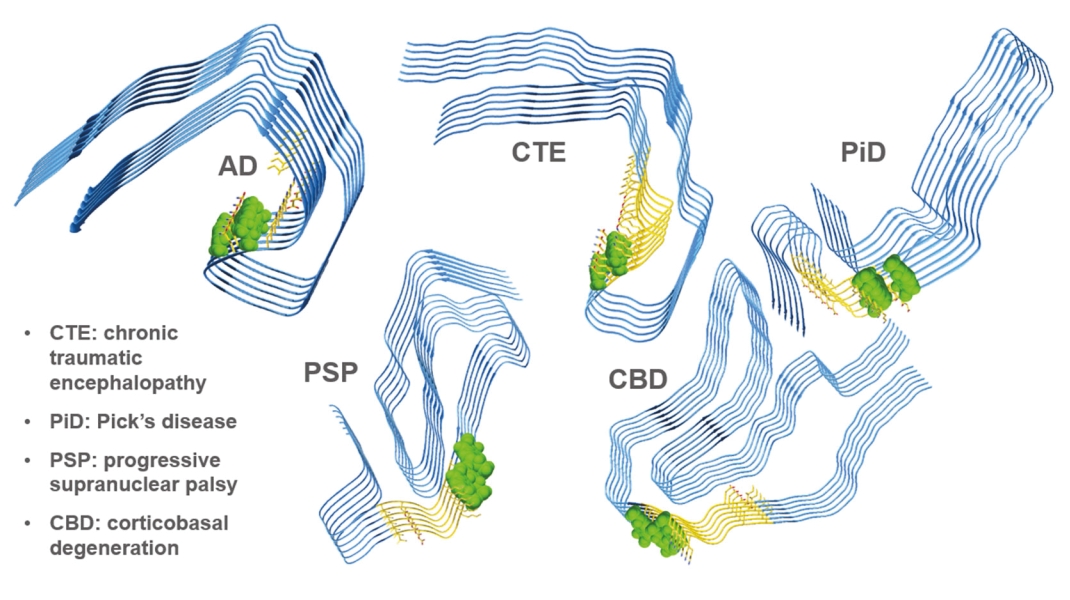
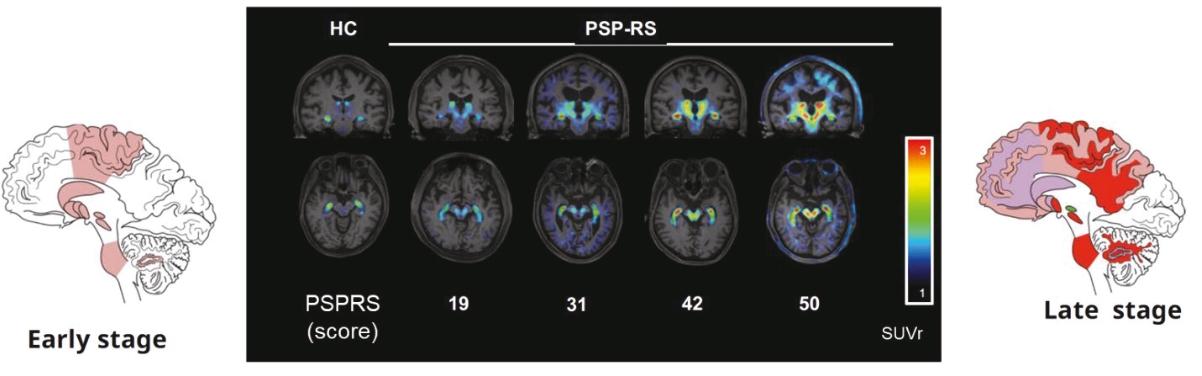
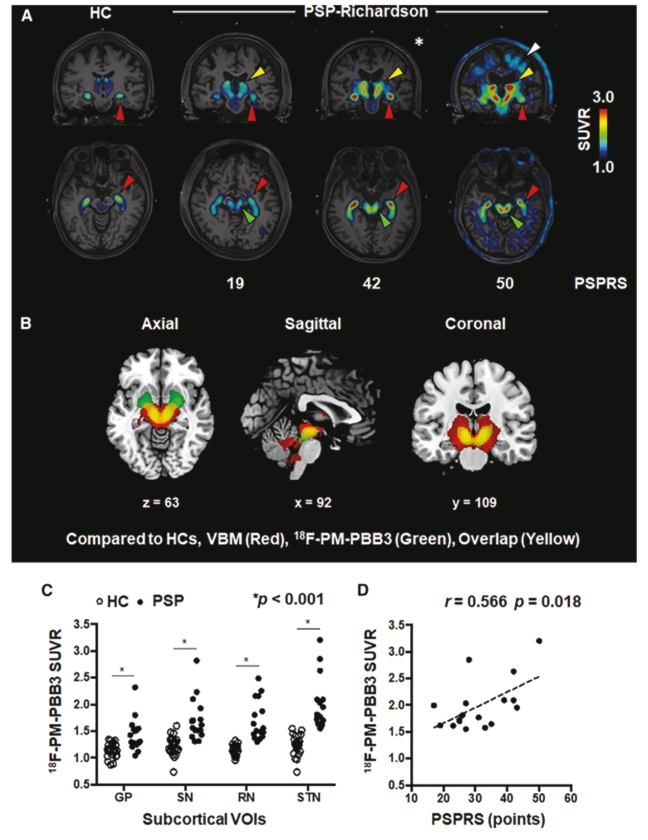
(A) | Coronal (upper) and axial (lower) APN-1607-PET images of HC and PSP-Richardson patients with different disease severities scored by PSPRS. Signals are localized to basal ganglia areas (yellow arrowhead) and midbrain (green arrowhead) and expansion to the primary motor and adjacent cerebral cortices containing white matter (white arrowhead) with clinical advancement. The asterisked image was derived from an autopsy-confirmed PSP case. |
(B) | Voxel-based analyses of brain atrophy (voxel-based morphometry [VBM]; red), APN-1607signal increase (green), and their spatial overlaps (yellow) in PSP-Richardson patients relative to HCs (p < 0.05, familywise error corrected at cluster level). Statistical maps are displayed in the Montreal Neurological Institute coordinate space. |
(C) | Comparisons of APN-1607 uptake in subcortical regions, including the globus pallidus (GP), substantia nigra (SN), raphe nucleus (RN), and subthalamic nucleus (STN) between 23 HCs (white circles) and 16 PSP-Richardson patients (black circles). *p < 0.001 by two-sample t test. |
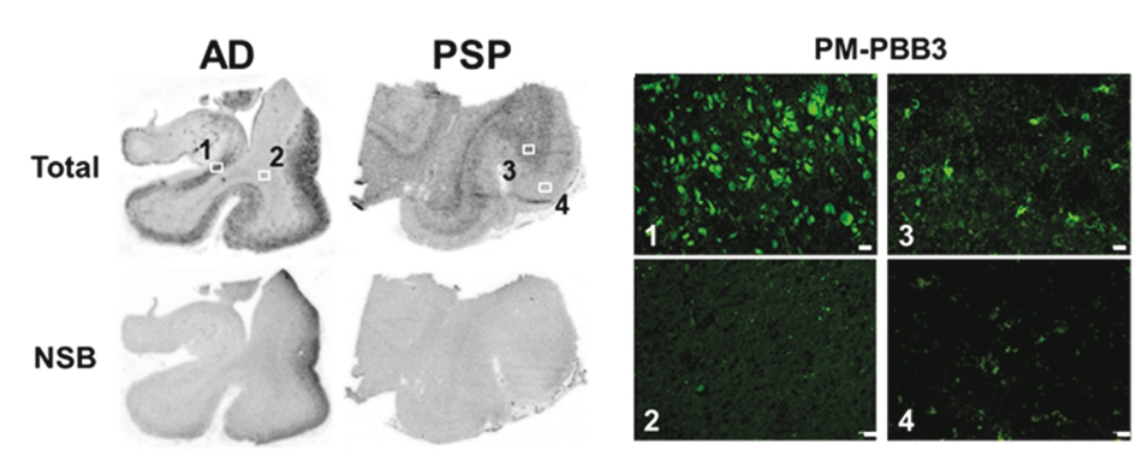
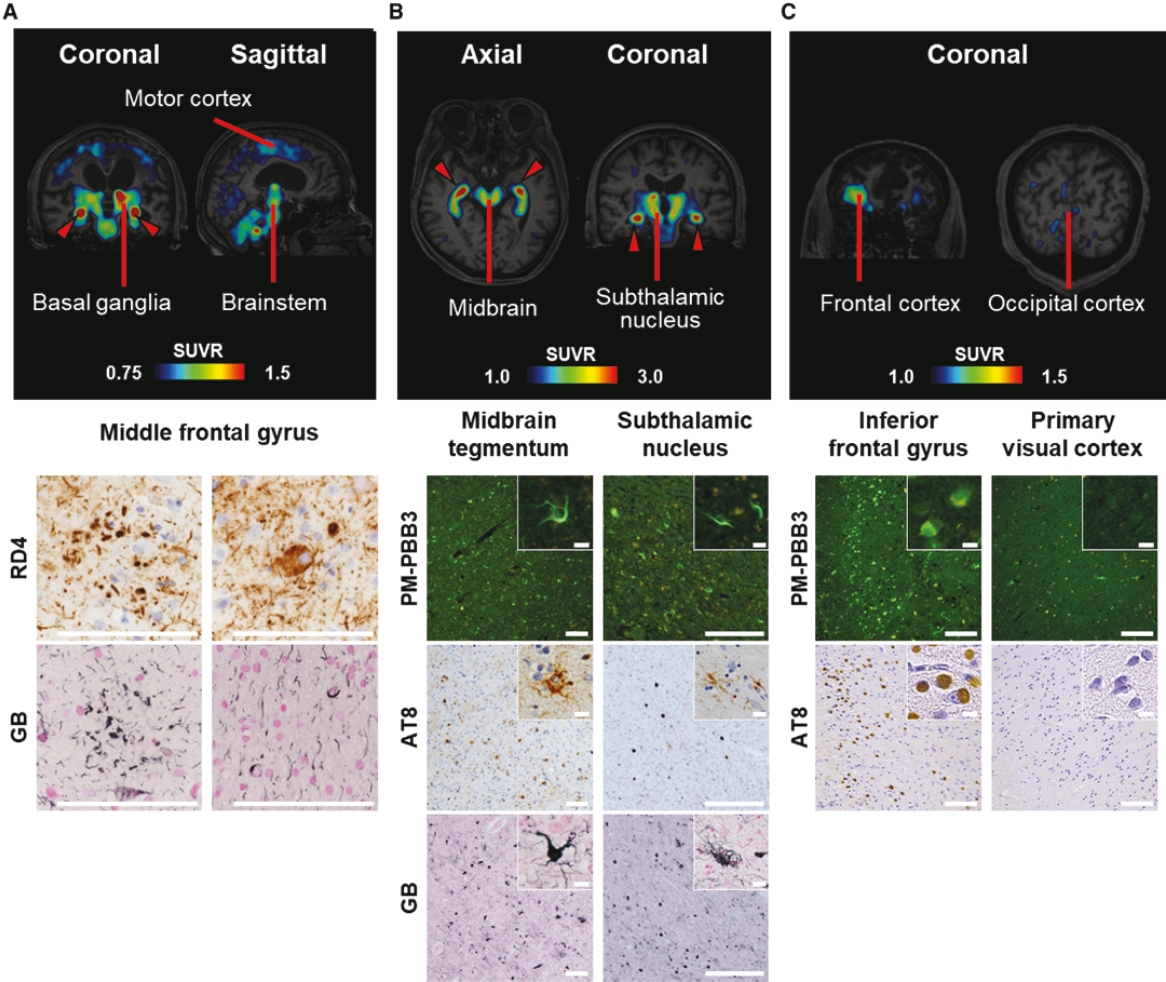
(A) | Coronal and sagittal brain images of a 68-year-old subject clinically diagnosed with CBS (upper panels). Enhanced radioligand binding was observed in the primary motor and adjacent cortices and subcortical regions, including basal ganglia, subthalamic nucleus, midbrain, pons, and choroid plexus (red arrowheads). Neuropathological assays of biopsy tissues collected from the middle frontal gyrus revealed the existence of astrocytic plaques, ballooned neurons, and coiled bodies stained with RD4 and/or GB in the cortex and corticomedullary junction (lower panels), in agreement with CBD tau pathologies. |
(B) | Axial and coronal 18F-PM-PBB3 PET images of a 65-year-old patient with a clinical diagnosis of PSP-Richardson (PSP-3, upper panels). The radioligand binding was augmented in the midbrain, subthalamic nucleus, neighboring subcortical structures, and choroid plexus (red arrowheads). Brain autopsy conducted 2 years after the PET scan demonstrated abundant accumulation of tufted astrocytes stained with non-radiolabeled PM-PBB3,AT8, and GB in the midbrain tegmentum and subthalamic nucleus (lower panels), indicating PSP as a definite diagnosis of this individual. |
(C) | Coronal 18F-PM-PBB3 PET images of a 59-year-old patient clinically diagnosed with bvFTD (PiD-2, upper panels). Accumulations of radio signals were noticeable in the frontal cortex, in contrast with a lack of radioligand binding in the occipital cortex. Brain autopsy was carried out 1 year after the PET scan, showing great abundance of Pick bodies and neuropil threads stained with non-radiolabeled PM-PBB3 and AT8 in the inferior frontalgyrus (lower panels). This was in sharp distinction from the few tau pathologies in the primary visual cortex (lower panels), collectively supporting a definite diagnosis of this case as PiD. |
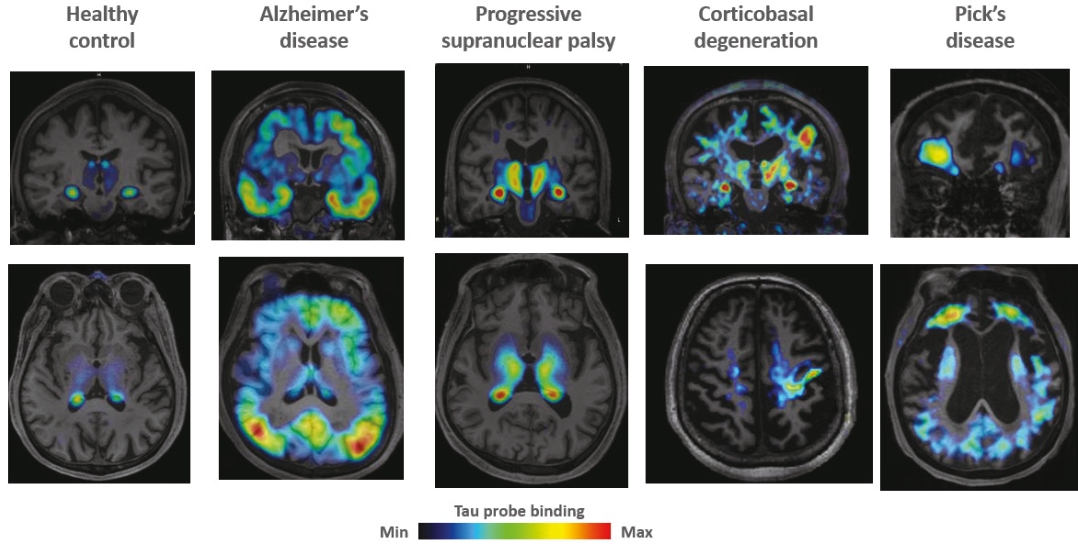
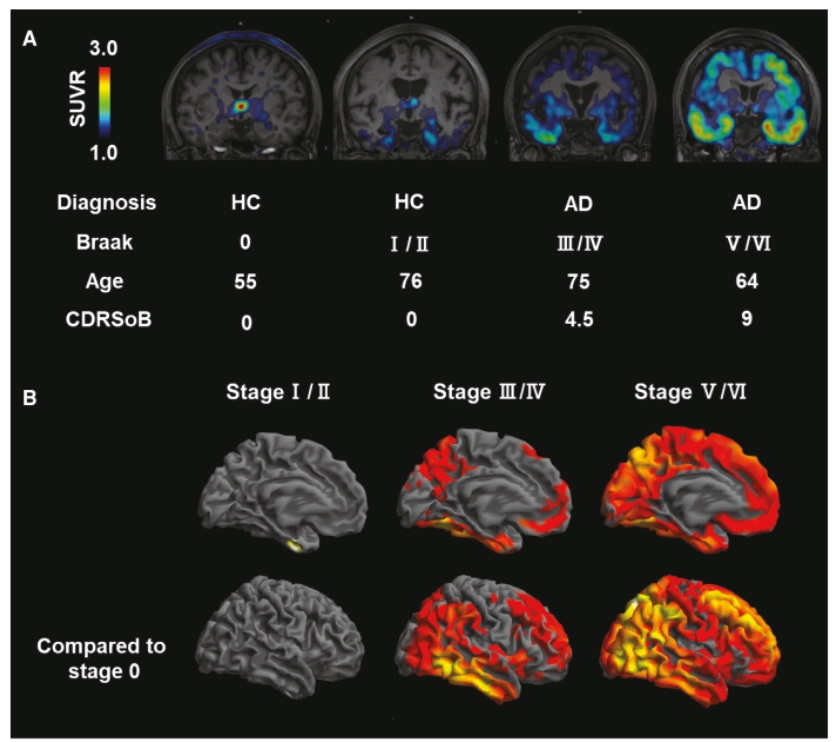
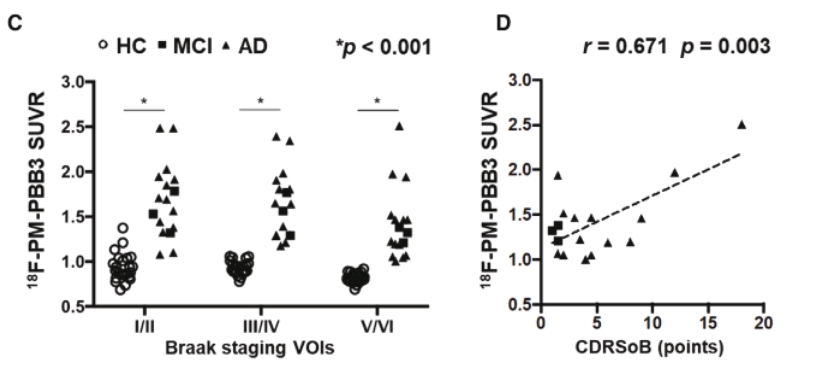
(A) | Coronal 18F-PM-PBB3-PET images of HCs and AD patients classified into different Braak tau stages. |
(B) | The topology of increased 18F-PM-PBB3 binding in subjects at each Braak stage compared to 22 HCs (stage zero). p <0.005, uncorrected, for one HC (stage I/II); p <0.05, familywise error corrected at cluster level, for four MCI/AD patients (stage III/IV) and for 13 MCI/AD patients (stage V/VI). |
(C) | Comparisons of 18F-PM-PBB3 binding in Braak stage VOIs between 23 HCs (white circles) and three MCI (black squares) and 14 AD (black triangles) cases. *p <0.001 by two-sample t test. |
(D) | Correlation of 18F-PM-PBB3 binding in the Braak stage V/VI VOI with CDRSoB points in MCI (black squares) and AD (black triangles) patients. r = 0.671 and p =0.003 by Pearson’s correlation analysis. Associations between the clinical disease severity and the extension of 18F-PM-PBB3 binding among MCI/AD patients. |
• | α-Syn PET Tracer. The discovery and development of a PET imaging tracer that can detect α-synuclein aggregates in patients with PD and related α-synucleinopathies such as MSA represents a significant unmet need in the field. There are no approved tracers for this indication. We have entered into a Research Collaboration Agreement with H. Lundbeck A/S (“Lundbeck”) and Abbvie Inc. (“Abbvie”), in which we share the responsibilities to advance development of aSyn PET tracer candidates. We initially identified 16 lead compounds. Among them, we transferred four compounds to Lundbeck and another four compound to Abbvie for further evaluation, including autoradiography, brain tissue binding, and PK studies. The remaining eight compounds were evaluated in-house by us. These results were shared among the three parties. Our α-Syn PET tracer lead candidate (RAC0523 binds to abnormal α-Syn aggregates in multiple α-Synucleinopathies including PD and MSA. Non-human primate imaging study indicates moderate brain penetration, promising kinetics and slow washout. RAC 0523 was observed to be well tolerated in a rodent toxicology study conducted by our vendor under GLP conditions, which is an important stage-gate in determining the possibility of advancing the molecule into clinical development. Lundbeck continues to perform binding studies and Abbvie is contributing radiological chemistry resources. See “— License Agreements and Collaborations” for more details. |
• | We believe our diagnostic platform has broad applicability across a wide array of targets and indications. We are also exploring other potential promising targets and indications. |
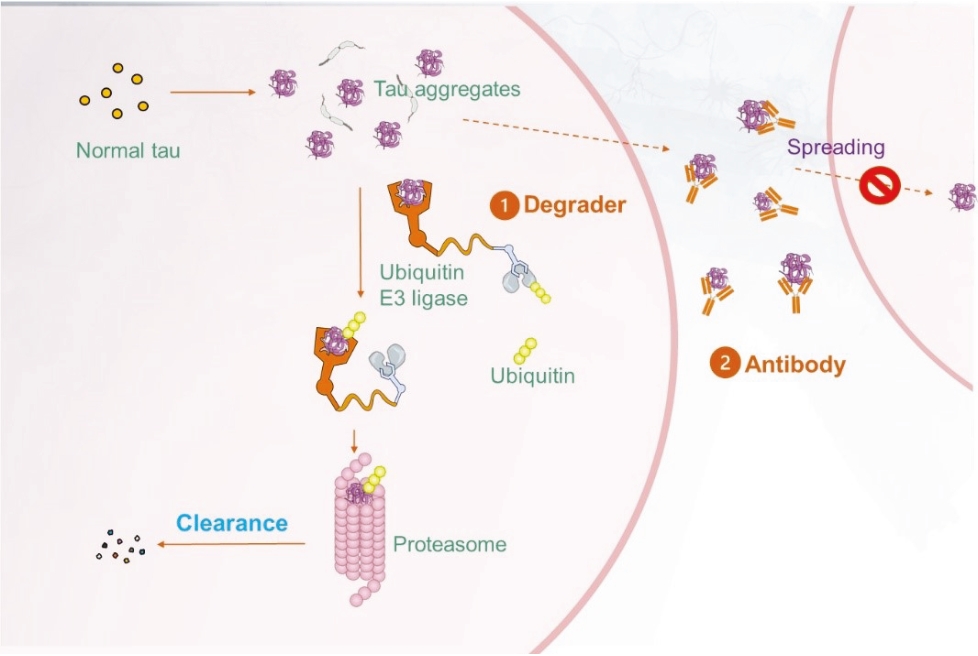
• | Degrader platform. Our degrader platform has four parts, namely (i) a proprietary PROTAC degrader compound library, (ii) cryogenic electron microscopy (“cryoEM”) technology to validate the binding of select compounds at the molecular level, (iii) diagnostic tools in the form of PET tracers which can potentially be used as pharmacodynamic biomarkers to monitor the removal of pathological tau, and (iv) animal models to confirm compounds’ activity therapeutic value in vivo. Leveraging our degrader platform, we can quickly assay our CNS protein binding compound collection to select promising candidates. Once promising candidates are identified, we can use cryoEM to confirm a binding mode and to facilitate the development and optimization of structural activity relationship (“SAR”). Finally, using our PET tracers, we can measure target engagement of our candidates in vivo. We are building a small molecule library designed to target pathological aggregates. |
• | Antibody platform. Both hybridoma and phage display library approaches were employed to generate more than 40 anti-tau antibodies for our antibody library. Moreover, we designed a disease-focus screening framework to identify several anti-tau conformational antibodies recognizing pathological tau species enriched at neuronal synapses. Our most clinically advanced product candidate from the antibody platform is APNmAb005. Based on this experience, we believe we have the capability of generating novel antibodies targeting other pathological protein aggregates. |
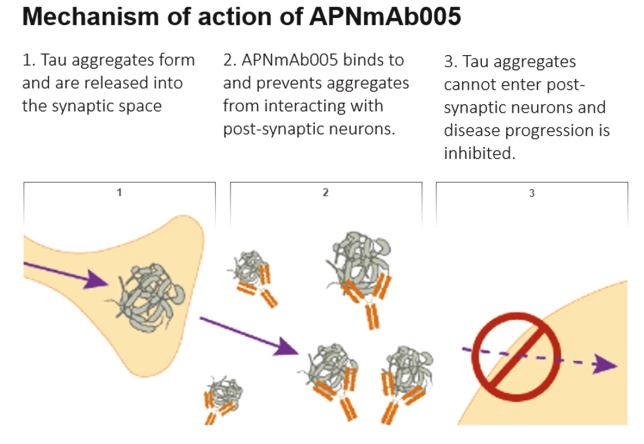
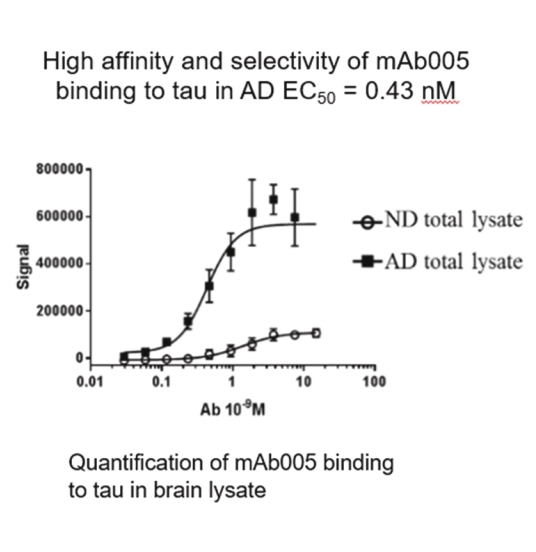
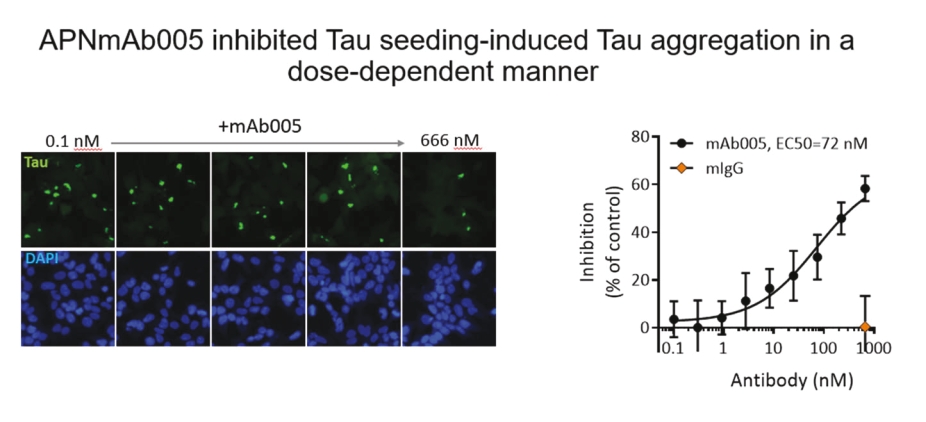
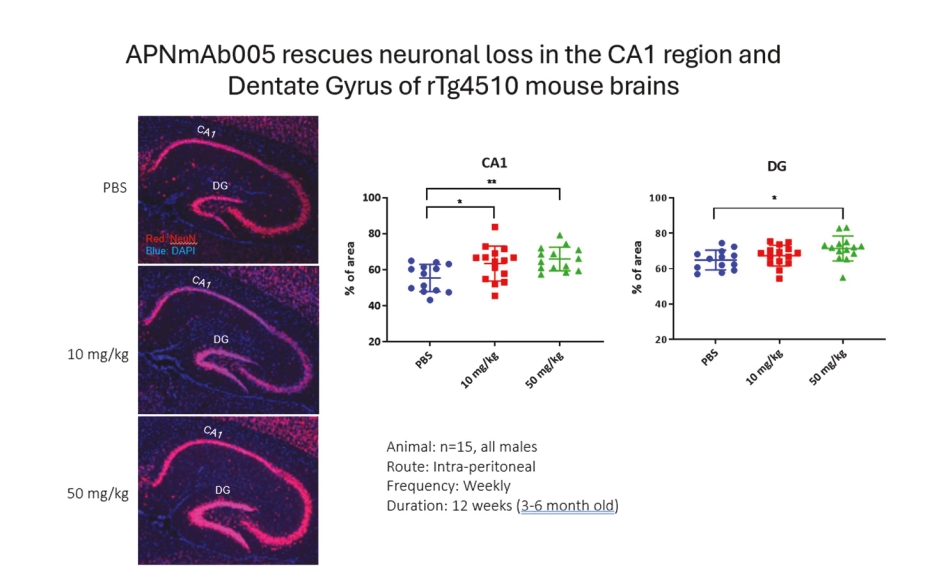
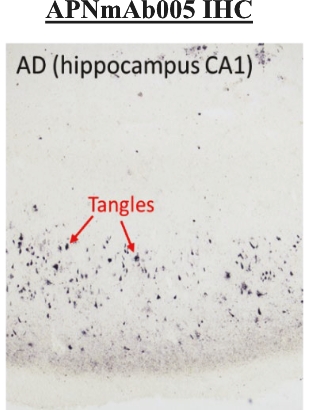 | | | 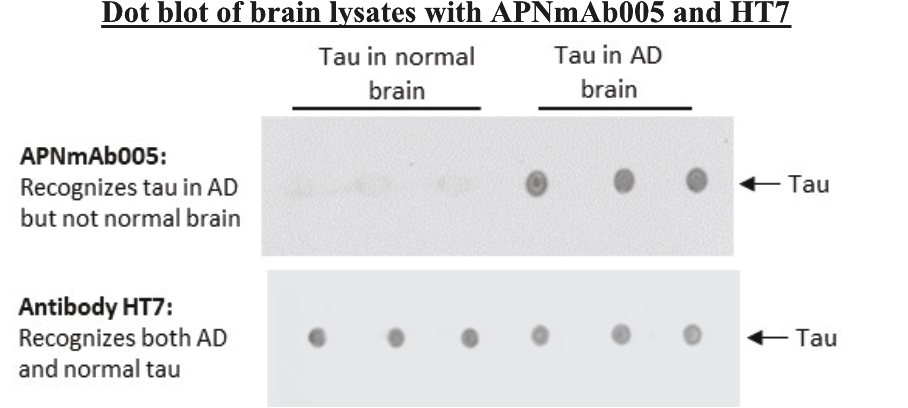 |

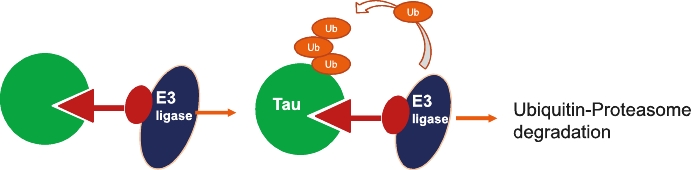 | | | Upon entering the cells, a degrader forms a ternary complex with tau and E3 ligase. The latter transfers ubiquitin to tau and marks it as substrate to be removed by proteasome degradation. |
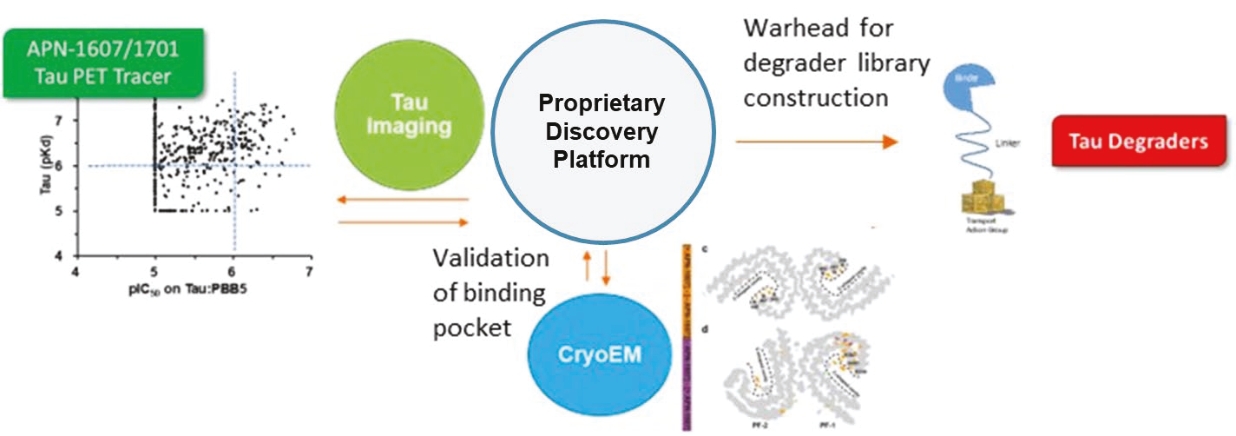
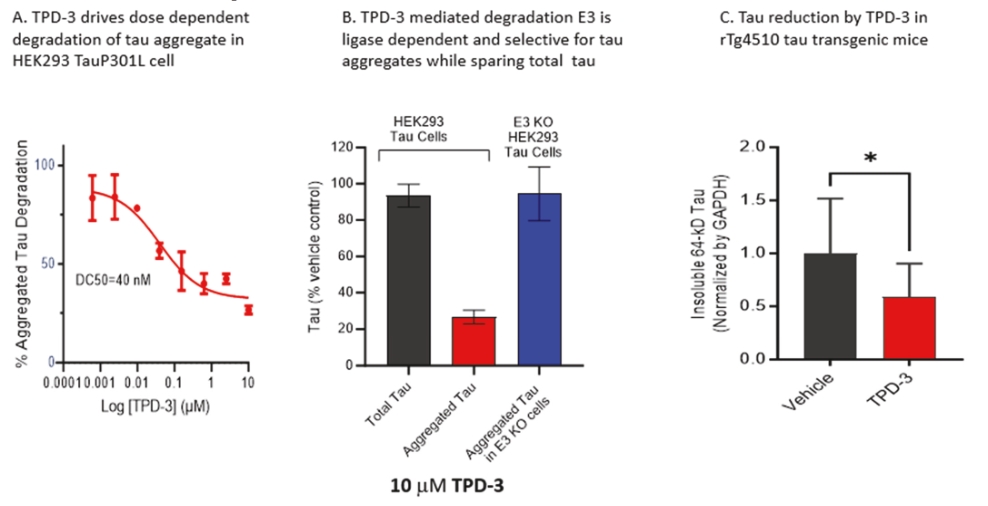
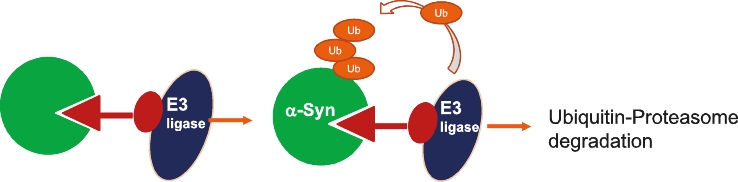 | | | Similar to that described for tau degraders, α-Syn degraders drive the formation of a ternary complex with α-Syn and E3 ligase. α-Syn labeled with ubiquitin by E3 ligase is then removed by proteasome degradation. |
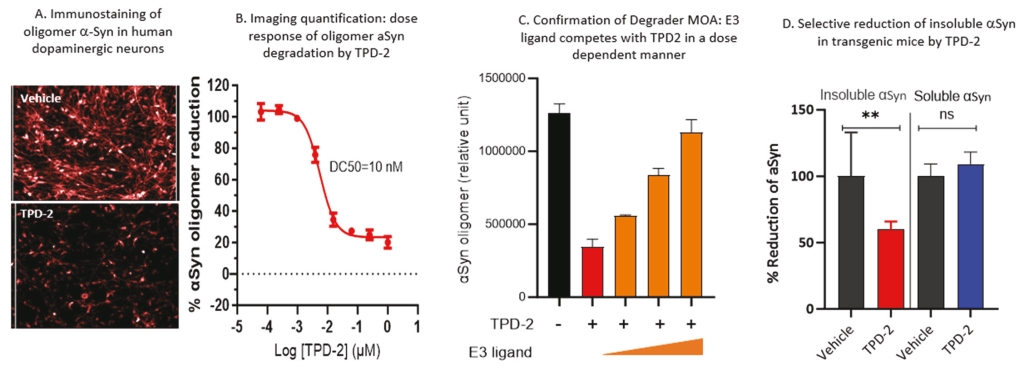
• | Tau PET tracer patent family (including APN-1607 related patents licensed from QST). We exclusively license a portfolio of patents and patent applications describing composition-of-matter claims encompassing tau PET tracers including APN-1607 as well as claims to associated methods of use for PET imaging from QST in Japan. See “—License Agreements and Collaborations—License Agreement with National Institutes for Quantum Science and Technology” above for more details. As of the date of this prospectus, our tau PET tracers’ patent portfolio, licensed from QST, includes two issued U.S. patents and one pending U.S. patent application, as well as 21 issued patents in Taiwan, Australia, Canada, China, Hong Kong, Japan, South Korea, Singapore, European Patent Office, Switzerland, Germany, Denmark, Estonia, Finland, France, United Kingdom, Italy, Netherlands, and Sweden, and one pending patent application in India. The issued U.S. patents will expire in December 2032 and October 2033, respectively, and other patents granted in other jurisdictions of this patent family will expire no earlier than December 2032, without taking potential patent term extensions into account. |
• | α-Syn PET tracer patent family. As of the date of this prospectus, under our α-syn PET tracer patent family, we owned seven issued patents in Australia, China, India, Japan, South Korea, New Zealand, and South Africa, as well as two pending patent applications in the United States and 12 pending patent applications in Taiwan, Brazil, Canada, Hong Kong, European Patent Office, Israel, Japan, South Korea, Malaysia, Mexico, New Zealand, Thailand, and Vietnam. These pending patent applications contain composition-of-matter claims encompassing various α-Syn small molecules as well as claims to associated methods of manufacture and methods of use. These patents, if granted, will expire in 2039 without taking potential patent term extensions into account. |
• | Degrader patent families. As of the date of this prospectus, under our degrader patent families, we owned two issued patents in the United State claiming degrader compositions of matter targeting tau and α-syn, respectively and one issued patent in Taiwan. The issued U.S. patents will expire in November 2040, |
• | Anti-tau antibody patent family (including APNmAb005). As of the date of this prospectus, under our anti-tau antibody patent family, we owned two issued patents in the United States and five issued patents in Australia, China, Japan, South Korea and Taiwan, as well as one pending patent application in the United States and five pending patent applications in Taiwan, Canada, Hong Kong, European Patent Office, and Singapore. These issued patents and pending patent applications are directed to composition-of-matter claims encompassing anti-tau antibodies including our product candidates APNmAb005. The issued U.S. patents will expire in August 2040, and other patents granted in other jurisdictions of this patent family will expire no earlier than 2040, without taking potential patent term extensions into account. |
• | the completion of preclinical laboratory tests and animal tests conducted under cGLP regulations; |
• | the submission to the FDA of an IND application for human clinical testing, which must become effective before human clinical studies commence; |
• | obtaining a positive opinion from the ethics committee (Europe)/institutional review board (U.S.) to commence study on human subjects; |
• | the performance of adequate and well-controlled human clinical studies to establish the safety and efficacy of the product candidate for each proposed indication and conducted in accordance with cGCP requirements; |
• | pre-NDA submission meeting with FDA (highly recommended); |
• | the submission to the FDA of an NDA; |
• | the FDA’s acceptance of the NDA; |
• | satisfactory completion of an FDA Pre-Approval Inspection (“PAI”) of the manufacturing facilities at which the product is made to assess compliance with cGMP requirements; |
• | the FDA’s review and approval of an NDA prior to any commercial marketing or sale of the drug in the United States; and |
• | having parallel scientific advice from the EMA or Health Technology Assessment body whereby the payors are involved at the outset (Phase 2), which is intended to facilitate the design of clinical studies to target primarily populations with a high chance of obtaining reimbursement and accelerate the process of time to reimbursement. |
• | restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
• | fines, warning letters or holds on post-approval clinical studies; |
• | refusal of the FDA to approve pending NDAs or supplements to approved NDAs, or suspension or revocation of product license approvals; |
• | product seizure or detention, or refusal to permit the import or export of products; or |
• | injunctions or the imposition of civil or criminal penalties. |
Name | | | Age | | | Position(s) |
Ming-Kuei Jang, Ph.D. | | | 52 | | | Chairman of the Board of Directors |
Mark S. Shearman, Ph.D. | | | 62 | | | Director and Chief Executive Officer |
Michael Xin Hui, M.B.A. | | | 52 | | | Director |
Zhigang Luo, M.B.A. | | | 53 | | | Director |
Roger James Pomerantz, M.D., F.A.C.P. | | | 66 | | | Director |
Bradford A. Navia, M.D., Ph.D. | | | 69 | | | Chief Medical Officer |
Brian Achenbach, M.B.A. | | | 59 | | | Chief Financial Officer |
Lana Gladstein, J.D. | | | 48 | | | General Counsel |
• | conducting and managing the business of our company; |
• | representing our company in contracts and deals; |
• | appointing attorneys for our company; |
• | selecting and removing senior management; |
• | providing employee benefits and pensions; |
• | managing our company’s finance and bank accounts; |
• | evaluating the performance and determining the compensation level of chief executive officer; |
• | exercising the borrowing powers of our company and mortgaging the property of our company; and |
• | exercising any other powers conferred by the shareholders meetings or under our amended and restated memorandum and articles of association. |
• | Class I, which will consist of , whose term will expire at our first annual general meeting of shareholders to be held after the closing of this offering or until their successors are elected and qualified; |
• | Class II, which will consist of , whose term will expire at our second annual general meeting of shareholders to be held after the closing of this offering or until their successors are elected and qualified; and |
• | Class III, which will consist of , whose term will expire at our third annual general meeting of shareholders to be held after the closing of this offering or until their successors are elected and qualified. |
• | selecting the independent auditor; |
• | pre-approving auditing and non-auditing services permitted to be performed by the independent auditor; |
• | annually reviewing the independent auditor’s report describing the auditing firm’s internal quality control procedures, any material issues raised by the most recent internal quality control review, or peer review, of the independent auditors and all relationships between the independent auditor and our company; |
• | review responsibilities, budget, compensation and staffing of our internal audit function; |
• | reviewing with the independent auditor any audit problems or difficulties and management’s response; |
• | reviewing and, if material, approving all related party transactions on an ongoing basis; |
• | reviewing and discussing the annual audited financial statements with management and the independent auditor; |
• | reviewing and discussing with management and the independent auditors major issues regarding accounting principles and financial statement presentations; |
• | reviewing reports prepared by management or the independent auditors relating to significant financial reporting issues and judgments; |
• | discussing earnings press releases with management, as well as financial information and earnings guidance provided to analysts and rating agencies; |
• | reviewing with management and the independent auditors the effect of regulatory and accounting initiatives, as well as off-balance sheet structures, on our financial statements; |
• | discussing policies with respect to risk assessment and risk management with management and internal auditors; |
• | timely reviewing reports from the independent auditor regarding all critical accounting policies and practices to be used by our company, all alternative treatments of financial information within IFRS that have been discussed with management and all other material written communications between the independent auditor and management; |
• | establishing procedures for the receipt, retention and treatment of complaints received from our employees regarding accounting, internal accounting controls or auditing matters and the confidential, anonymous submission by our employees of concerns regarding questionable accounting or auditing matters; |
• | such other matters that are specifically delegated to our audit committee by our board of directors from time to time; and |
• | meeting separately, periodically, with management, internal auditors and the independent auditor. |
• | reviewing, evaluating and, if necessary, revising our overall compensation policies; |
• | reviewing and evaluating the performance of our directors and relevant senior officers and determining the compensation of relevant senior officers; |
• | reviewing and approving our senior officers’ employment agreements with us; |
• | setting performance targets for relevant senior officers with respect to our incentive compensation plan and equity-based compensation plans; |
• | administering our equity-based compensation plans in accordance with the terms thereof; and |
• | such other matters that are specifically delegated to the compensation committee by our board of directors from time to time. |
• | selecting and recommending to our board of directors nominees for election by the shareholders or appointment by the board; |
• | reviewing annually with our board of directors the current composition of our board of directors with regards to characteristics such as independence, knowledge, skills, experience and diversity; |
• | making recommendations on the frequency and structure of our board of directors meetings and monitoring the functioning of the committees of our board of directors; and |
• | advising our board of directors periodically with regards to significant developments in the law and practice of corporate governance as well as our compliance with applicable laws and regulations, and making recommendations to the board on all matters of corporate governance and on any remedial action to be taken.] |
• | Types of Awards. The awards granted under the plan mean the option to purchase the ordinary shares of our company. |
• | Plan Administration. The plan will be administered by a committee appointed by our board of directors, which shall consist of four directors. |
• | Award Agreement. Awards granted under the plan are evidenced by a written instrument executed by the relevant participant to whom an award is granted and our company, containing such terms, conditions, limitations and restrictions as the committee that administers the plan shall deem advisable which are not inconsistent with the plan. |
• | Exercise Price. The exercise price for the ordinary shares purchased under an award shall be determined by the committee that administers the plan, but shall be no less than the fair market value per share on the date of grant. |
• | Eligibility. Persons eligible to participate in the plan include our employees, consultants and advisors. |
• | Term of the Awards. The maximum of an award shall be established by the committee that administers the plan or, if not so established, shall be ten (10) years from the date of grant. |
• | Vesting Schedule. The vesting schedule will be set forth in the written instrument evidencing the awards granted. |
• | Assignability. No award or interest in an award under the plan may be sold, assigned, pledged or transferred by the participants or made subject to attachment or similar proceedings otherwise than by will or by the applicable laws of descent and distribution. However, the committee may in its sole discretion permit the transfer of an award subject to the terms and conditions of the plan and the relevant instrument. |
• | Term and Termination. The plan shall terminate ten (10) years from its effective date, unless sooner terminated by our board of directors. |
Name | | | Ordinary Shares Underlying Options | | | Exercise Price ($/Share) | | | Date of Grant | | | Date of Expiration |
Ming-Kuei Jang | | | 250,000 | | | 0.400 | | | November 1, 2018 | | | October 31, 2023 |
| | | 341,389 | | | 0.400 | | | October 1, 2019 | | | September 30, 2024 | |
| | | 805,000 | | | 0.624 | | | November 1, 2019 | | | October 31, 2024 | |
| | | * | | | 0.624 | | | August 15, 2022 | | | August 14, 2027 | |
| | | 700,000 | | | 0.632 | | | August 15, 2022 | | | August 14, 2027 | |
Mark S. Shearman | | | * | | | 0.400 | | | November 1, 2018 | | | October 31, 2023 |
| | | * | | | 0.632 | | | August 15, 2022 | | | August 14, 2027 | |
| | | * | | | 0.632 | | | July 17, 2023 | | | July 16, 2028 | |
| | | * | | | 0.632 | | | December 1, 2023 | | | November 30, 2028 | |
Michael Xin Hui | | | — | | | — | | | — | | | — |
| | | | | | | | | |||||
Zhigang Luo | | | — | | | — | | | — | | | — |
Roger James Pomerantz | | | — | | | — | | | — | | | — |
Bradford A. Navia | | | 337,500 | | | 0.632 | | | December 1, 2021 | | | November 30, 2026 |
Brian Achenbach | | | 250,000 | | | 0.632 | | | February 1, 2023 | | | January 31, 2028 |
Lana Gladstein | | | * | | | 0.632 | | | July 17, 2023 | | | July 16, 2028 |
| | | * | | | 0.632 | | | December 1, 2023 | | | November 30, 2028 | |
| | | * | | | 0.632 | | | January 12, 2024 | | | January 11, 2029 | |
All directors and executive officers as a group | | | 2,961,771 | | | | | | |
* | Less than 1% of our total outstanding ordinary shares on an as-converted basis. |
| | | Ordinary Shares Beneficially Owned Prior to This Offering | | | Ordinary Shares Beneficially Owned After This Offering | |||||||
| | | Number | | | % | | | Number | | | % | |
Directors and Executive Officers**: | | | | | | | | | ||||
Ming-Kuei Jang(1) | | | 2,181,166 | | | 8.0% | | | 2,181,166 | | | 7.4% |
Mark S. Shearman | | | * | | | * | | | * | | | * |
Michael Xin Hui | | | — | | | — | | | — | | | — |
Zhigang Luo | | | — | | | — | | | — | | | — |
Roger James Pomerantz | | | — | | | — | | | — | | | — |
Bradford A. Navia(2) | | | 337,500 | | | 1.3% | | | 337,500 | | | 1.2% |
Brian Achenbach | | | * | | | * | | | * | | | * |
Lana Gladstein | | | * | | | * | | | * | | | * |
All Directors and Executive Officers as a Group | | | 2,882,145 | | | 11.2% | | | 2,882,145 | | | 10.4% |
| | | | | | | | | |||||
Principal Shareholders: | | | | | | | | | ||||
Entities Affiliated with Dongcheng Pharma(3) | | | 4,810,052 | | | 18.6% | | | 4,810,052 | | | 17.3% |
Wealth Path Investments Limited(4) | | | 2,475,000 | | | 9.6% | | | 2,475,000 | | | 8.9% |
Daiwa Taiwan-Japan Biotech Fund(5) | | | 2,437,500 | | | 9.4% | | | 2,437,500 | | | 8.8% |
KTB China Synergy Fund(6) | | | 2,149,067 | | | 8.3% | | | 2,149,067 | | | 7.7% |
ShangPharma Investment Group Limited(7) | | | 1,625,750 | | | 6.3% | | | 1,625,750 | | | 5.8% |
* | Less than 1% of our total outstanding shares. |
** | Except as otherwise indicated below, the business address of our directors and executive officers. |
(1) | Represents 639,264 ordinary shares held by Mr. Ming-Kuei Jang and 1,541,902 ordinary shares issuable upon the exercise of options held by Mr. Jang within 60 days of the date of this prospectus. |
(2) | Represents 337,500 ordinary shares issuable upon the exercise of options held by Mr. Bradford A. Navia within 60 days of the date of this prospectus. |
(3) | Represents 963,563 ordinary shares and 3,371,805 Series C preferred shares directly held by Yantai Dongcheng Biochemicals Co., Ltd. (“Dongcheng Pharma”) and 474,684 Series C preferred shares directly held by DongCheng International (HongKong) Limited (“Dongcheng HK”), a wholly owned subsidiary of Dongcheng Pharma. The registered address of Dongcheng Pharma is No.7 Chang Bai Shan Road, Yantai Economic and Technological Development Zone, Shandong Province, China, and the registered address of Dongcheng HK is Room 413, 4/F, Lucky Centre, 165-171 Wan Chai Road, Wan Chai, Hong Kong. Dongcheng Pharma is a company publicly listed on the Shenzhen Stock Exchange (stock code: 002675). |
(4) | Represents 2,475,000 ordinary shares held by Wealth Path Investments Limited (“Wealth Path”). Wealth Path is wholly owned by Mr. Spencer Lee. By virtue of being the controlling shareholder and the director of Wealth Path, Mr. Spencer Lee may be deemed to have beneficial ownership over shares held by Wealth Path. The registered address of Wealth Path is Vistra Corporate Services Centre, Wickhams Cay II, Road Town, Tortola, VG1110, British Virgin Islands. |
(5) | Represents 1,500,000 ordinary shares and 937,500 Series B preferred shares directly held by Daiwa Taiwan-Japan Biotech Fund (“Daiwa”). DCI Partners Co., Ltd. (“DCIP”) is the general partner of Daiwa. The voting and investment power of shares held by Daiwa is exercised by majority vote of an investment committee consisting of DCIP’s employees, each of whom disclaims beneficial ownership for the shares held by Daiwa, except to the extent of any pecuniary interest therefrom. The principal business address of Daiwa is 1-9-1 Marunouchi, Chiyoda-ku, Tokyo, 100-6756, Japan. |
(6) | Represents 1,562,500 Series B preferred shares, 400,000 Series Pre-C preferred shares and 186,567 Series C preferred shares directly held by KTB China Synergy Fund (“KTB”). The voting and investment power of shares held by KTB is exercised by its general partner, Woori Venture Partners, which disclaims beneficial ownership for the shares held by KTB. The principal business address of KTB is 670 Daewangpangyo-ro, Bundang-gu, Seongnam-si, Gyeonggi-do 10FL, USpace 2A dong, Republic of Korea. |
(7) | Represents 1,313,250 ordinary shares and 312,500 Series B preferred shares directly held by ShangPharma Investment Group Limited (“ShangPharma”). The voting and investment power of shares held by ShangPharma is exercised by its managing director, Mr. Michael Xin Hui. The registered address of ShangPharma is Kingston Chambers, P.O. Box 173, Road Town, Tortola, British Virgin Islands. |
• | the instrument of transfer is lodged with us, accompanied by the certificate for the ordinary shares to which it relates and such other evidence as our board of directors may reasonably require to show the right of the transferor to make the transfer; |
• | the instrument of transfer is in respect of only one class of ordinary shares; |
• | the instrument of transfer is properly stamped, if required; |
• | in the case of a transfer to joint holders, the number of joint holders to whom the ordinary share is to be transferred does not exceed four; and |
• | a fee of such maximum sum as the Nasdaq may determine to be payable or such lesser sum as our directors may from time to time require is paid to us in respect thereof. |
• | the designation of the series; |
• | the number of shares of the series; |
• | the dividend rights, dividend rights, conversion rights, voting rights; |
• | the rights and terms of redemption and liquidation preferences; and |
• | any other powers, preferences and relative, participating, optional and other special rights. |
• | authorize our board of directors to issue preference shares in one or more series and to designate the price, rights, preferences, privileges and restrictions of such preference shares without any further vote or action by our shareholders; and |
• | limit the ability of shareholders to requisition and convene general meetings of shareholders. |
• | does not have to file an annual return of its shareholders with the Registrar of Companies; |
• | is not required to open its register of members for inspection; |
• | does not have to hold an annual general meeting; |
• | may issue shares or shares with no par value; |
• | may obtain an undertaking against the imposition of any future taxation (such undertakings are usually given for 20 years in the first instance); |
• | may register by way of continuation in another jurisdiction and be deregistered in the Cayman Islands; |
• | may register as a limited duration company; and |
• | may register as a segregated portfolio company. |
• | the statutory provisions as to the required majority vote have been met; |
• | the shareholders have been fairly represented at the meeting in question and the statutory majority are acting bona fide without coercion of the minority to promote interests adverse to those of the class; |
• | the arrangement is such that may be reasonably approved by an intelligent and honest man of that class acting in respect of his interest; and |
• | the arrangement is not one that would more properly be sanctioned under some other provision of the Companies Act. |
• | a company acts or proposes to act illegally or ultra vires; |
• | the act complained of, although not ultra vires, could only be effected duly if authorized by more than a simple majority vote that has not been obtained; and |
• | those who control the company are perpetrating a “fraud on the minority.” |
• | 1% of our then outstanding ordinary shares, which will equal approximately 278,288 ordinary shares immediately after this offering, assuming the underwriters do not exercise their over-allotment option; or |
• | the average weekly trading volume of our ordinary shares on the Nasdaq, during the four calendar weeks preceding the date on which notice of the sale is filed with the SEC. |
• | banks, insurance companies, and certain other financial institutions; |
• | pension plans; |
• | U.S. expatriates and certain former citizens or long-term residents of the United States; |
• | persons holding ordinary shares as part of a hedging transaction, “straddle,” wash sale, conversion transaction or integrated transaction or persons entering into a constructive sale with respect to ordinary shares; |
• | persons whose “functional currency” for U.S. federal income tax purposes is not the U.S. dollar; |
• | brokers, dealers or traders in securities, commodities or currencies; |
• | tax-exempt entities or government organizations; |
• | S corporations, partnerships, or other entities or arrangements classified as partnerships for U.S. federal income tax purposes (and investors therein); |
• | regulated investment companies or real estate investment trusts; |
• | persons investing through individual retirement accounts or other tax deferred accounts; |
• | persons who acquired our ordinary shares pursuant to the exercise of any employee stock option or otherwise as compensation; |
• | persons that own or are deemed to own ten percent or more of our shares (by vote or value); and |
• | persons holding our ordinary shares in connection with a trade or business, permanent establishment, or fixed place of business outside the United States. |
(1) | an individual who is a citizen or resident of the United States; |
(2) | a corporation, or other entity taxable as a corporation for U.S. federal income tax purposes, created or organized in or under the laws of the United States, any state therein or the District of Columbia; |
(3) | an estate the income of which is subject to U.S. federal income taxation regardless of its source; or |
(4) | a trust if (1) a U.S. court is able to exercise primary supervision over the administration of the trust and one or more U.S. persons have authority to control all substantial decisions of the trust or (2) the trust has a valid election to be treated as a U.S. person under applicable U.S. Treasury Regulations. |
• | the excess distribution or gain will be allocated ratably over a U.S. Holder’s holding period for the ordinary shares; |
• | the amount allocated to the taxable year of the disposition or distribution (as applicable), and any taxable year prior to the first taxable year in which we became a PFIC, will be treated as ordinary income; and |
• | the amount allocated to each other year will be subject to the highest tax rate in effect for that year and the interest charge generally applicable to underpayments of tax will be imposed on the resulting tax attributable to each such year. |
Underwriter | | | Number of Shares |
US Tiger Securities, Inc. | | | [•] |
Total | | | 2,000,000 |
| | | Per Share | | | Total Without Exercise of Over- allotment Option | | | Total With Full Exercise of Over- allotment Option | |
Initial public offering price | | | $[•] | | | [•] | | | [•] |
Underwriting discounts and commissions to be paid by us | | | $[•] | | | [•] | | | [•] |
Proceeds, before expenses, to us | | | $[•] | | | [•] | | | [•] |
• | is an “investment business” within the meaning of clause 37 of Schedule 1 of the FMCA; |
• | meets the “investment activity criteria” specified in clause 38 of Schedule 1 of the FMCA; |
• | is “large” within the meaning of clause 39 of Schedule 1 of the FMCA; or |
• | is a “government agency” within the meaning of clause 40 of Schedule 1 of the FMCA. |
SEC Registration Fee | | | $7,380 |
FINRA Filing Fee | | | 4,250 |
Stock Exchange Application and Listing Fee | | | 295,000 |
Transfer Agent’s fees and expenses | | | 3,500 |
Printing and Engraving Expenses | | | 50,000 |
Legal Fees and Expenses | | | 800,000 |
Accounting Fees and Expenses | | | 150,000 |
Miscellaneous | | | 540,000 |
Total | | | $1,850,130 |
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
| | | ||
| | | ||
| | | ||
| | | ||
| | |
| | | December 31, | ||||
| | | 2022 | | | 2021 | |
Assets | | | | | ||
Current assets: | | | | | ||
Cash | | | $1,221 | | | $9,674 |
Prepaid expenses and other current assets | | | 590 | | | 429 |
Accounts receivable | | | — | | | 155 |
Total current assets | | | 1,811 | | | 10,258 |
Property and equipment, net | | | 2,153 | | | 509 |
Deferred offering costs | | | 1,288 | | | — |
Operating lease right-of-use assets | | | 154 | | | 322 |
Prepaid expenses, net of current portion and other long-term assets | | | 237 | | | 1,677 |
Total assets | | | $5,643 | | | $12,766 |
Liabilities, Redeemable Convertible Preferred Shares, and Shareholders' Deficit | | | | | ||
Current liabilities: | | | | | ||
Accounts payable | | | $8,887 | | | $432 |
Accrued expenses and other current liabilities | | | 2,466 | | | 2,993 |
Operating lease liabilities, current | | | 124 | | | 154 |
Related party payable | | | 904 | | | — |
Short-term borrowings | | | 1,450 | | | 785 |
Convertible notes (including a related party convertible note of $753 and $0 as of December 31, 2022 and 2021, respectively, net of debt discount and issuance costs) | | | 1,093 | | | — |
Derivative liabilities (including a related party derivative liability of $173 and $0 as of December 31, 2022 and 2021, respectively) | | | 251 | | | — |
Total current liabilities | | | 15,175 | | | 4,364 |
Operating lease liabilities, net of current portion | | | 42 | | | 177 |
Total liabilities | | | 15,217 | | | 4,541 |
Commitments and Contingencies (Note 15) | | | | | ||
Redeemable convertible preferred shares (Series B, Pre-C and C), $0.1 par value; 56,973,336 shares authorized; 53,622,601 and 47,926,399 shares issued and outstanding; redemption and liquidation value of $66,166 and $57,166 as of December 31, 2022 and 2021, respectively | | | 65,876 | | | 56,913 |
Shareholders' deficit: | | | | | ||
Ordinary shares, $0.1 par value, 443,026,664 shares authorized; 38,617,056 and 38,352,056 shares issued and outstanding as of December 31, 2022 and 2021, respectively | | | 3,862 | | | 3,835 |
Additional paid-in capital | | | 12,296 | | | 10,373 |
Accumulated deficit | | | (90,638) | | | (62,407) |
Accumulated other comprehensive loss | | | (970) | | | (489) |
Total shareholders' deficit | | | (75,450) | | | (48,688) |
Total liabilities, redeemable convertible preferred shares, and shareholders' deficit | | | $5,643 | | | $12,766 |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Revenue | | | $394 | | | $550 |
Operating expenses | | | | | ||
Research and development | | | 21,617 | | | 19,660 |
General and administrative | | | 7,041 | | | 4,787 |
Total operating expenses | | | 28,658 | | | 24,447 |
Loss from operations | | | (28,264) | | | (23,897) |
Other (expense) income: | | | | | ||
Interest expense, net | | | (67) | | | (36) |
Other income (expense), net | | | 117 | | | 672 |
Total other income | | | 50 | | | 636 |
Loss before income taxes | | | (28,214) | | | (23,261) |
Provision for income taxes | | | (17) | | | — |
Net loss | | | (28,231) | | | (23,261) |
Net loss attributable to ordinary shareholders | | | $(28,231) | | | $(23,261) |
Net loss per share attributable to ordinary shareholders | | | | | ||
Basic and diluted | | | $(0.73) | | | $(0.69) |
Weighted-average shares outstanding | | | | | ||
Basic and diluted | | | 38,482,015 | | | 33,561,324 |
Comprehensive loss: | | | | | ||
Net loss | | | (28,231) | | | (23,261) |
Foreign currency translation adjustment | | | (481) | | | (103) |
Total comprehensive loss | | | $(28,712) | | | $(23,364) |
| | | Redeemable Convertible Preferred | | | Ordinary Shares | | | Additional Paid-In Capital | | | Accumulated Deficit | | | Accumulated Other Comprehensive Income (Loss) | | | Total Shareholders' Deficit | |||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | ||||||||||||
Balance as of December 31, 2020 | | | 25,856,336 | | | $25,917 | | | 32,449,500 | | | $3,245 | | | $9,374 | | | $(39,146) | | | $(386) | | | $(26,913) |
Issuance of series C redeemable convertible preferred shares, net of issuance costs | | | 22,070,063 | | | 30,996 | | | — | | | — | | | — | | | — | | | — | | | — |
Share-based compensation expense | | | — | | | — | | | — | | | — | | | 914 | | | — | | | — | | | 914 |
Share option exercised | | | — | | | — | | | 5,902,556 | | | 590 | | | 85 | | | — | | | — | | | 675 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (23,261) | | | — | | | (23,261) |
Foreign currency translation adjustment | | | — | | | — | | | — | | | — | | | — | | | — | | | (103) | | | (103) |
Balance as of December 31, 2021 | | | 47,926,399 | | | 56,913 | | | 38,352,056 | | | 3,835 | | | 10,373 | | | (62,407) | | | (489) | | | (48,688) |
Issuance of series C redeemable convertible preferred shares, net of issuance costs | | | 5,696,202 | | | 8,963 | | | — | | | — | | | — | | | — | | | — | | | — |
Share-based compensation expense | | | — | | | — | | | — | | | — | | | 1,911 | | | — | | | — | | | 1,911 |
Share option exercised | | | — | | | — | | | 265,000 | | | 27 | | | 12 | | | — | | | — | | | 39 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (28,231) | | | — | | | (28,231) |
Foreign currency translation adjustment | | | — | | | — | | | — | | | — | | | — | | | — | | | (481) | | | (481) |
Balance as of December 31, 2022 | | | 53,622,601 | | | $65,876 | | | 38,617,056 | | | $3,862 | | | $12,296 | | | $(90,638) | | | $(970) | | | $(75,450) |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Operating Activities: | | | | | ||
Net loss | | | $(28,231) | | | $(23,261) |
Adjustments to reconcile net loss to net cash used in operating activities: | | | | | ||
Depreciation expense | | | 295 | | | 206 |
Non-cash interest expense | | | 15 | | | — |
Amortization of operating lease right-of-use assets | | | 148 | | | 112 |
Share-based compensation expense | | | 1,911 | | | 914 |
Gain on asset disposal | | | — | | | (20) |
Changes in operating assets and liabilities: | | | | | ||
Accounts receivable | | | 148 | | | (158) |
Prepaid expenses and other current assets | | | 1,358 | | | 277 |
Accounts payable | | | 7,853 | | | (2,635) |
Operating lease obligations | | | (145) | | | (100) |
Accrued expenses and other current liabilities | | | (1,933) | | | 1,785 |
Prepaid expenses, net of current portion and other long-term assets | | | 1,344 | | | (1,423) |
Net cash used in operating activities | | | (17,237) | | | (24,303) |
Investing Activities: | | | | | ||
Purchase of property and equipment | | | (2,016) | | | (497) |
Proceeds from disposal of property and equipment | | | — | | | 180 |
Net cash used in investing activities | | | (2,016) | | | (317) |
Financing Activities: | | | | | ||
Proceeds from issuance of preferred shares, net of issuance costs | | | 8,963 | | | 30,996 |
Proceeds from exercise of share options | | | 39 | | | 675 |
Proceeds from issuance of convertible notes (including proceeds from a related party convertible note of $1,000 and $0 as of December 31, 2022 and 2021, respectively) | | | 1,450 | | | — |
Proceeds from short-term borrowings | | | 1,486 | | | 775 |
Proceeds from related party payable | | | 899 | | | — |
Repayment of short-term borrowings | | | (743) | | | (954) |
Deferred offering costs associated with the Business Combination | | | (740) | | | — |
Net cash provided by financing activities | | | 11,354 | | | 31,492 |
Effect of exchange rates on cash | | | (554) | | | (60) |
Net increase (decrease) in cash | | | (8,453) | | | 6,812 |
Cash at beginning of period | | | 9,674 | | | 2,862 |
Cash at end of period | | | $1,221 | | | $9,674 |
Supplemental disclosure of cash flow information: | | | | | ||
Cash paid for loan interest | | | $41 | | | $43 |
Supplemental cash flow information on non-cash investing and financing activities: | | | | | ||
Right-of-use assets obtained in exchange of lease liabilities | | | $— | | | $405 |
Debt issuance cost associated with convertible notes included in accounts payable | | | $116 | | | $— |
Issuance of derivative instrument related to convertible notes | | | $251 | | | $— |
Deferred offering costs associated with the Business Combination in accounts payable | | | $548 | | | $— |
 in Japan (“APRINOIA Japan”), APRINOIA Therapeutics Limited in Hong Kong (“APRINOIA HK”) and APRINOIA Therapeutics, LLC in the United States (“APRINOIA USA”). APRINOIA HK has a direct wholly-owned subsidiary, Suzhou APRINOIA Therapeutics Co, Ltd., in Suzhou (“APRINOIA Suzhou”). The Company had a subsidiary in Taiwan (APRINOIA Therapeutics Inc. (Taiwan) or “APRINOIA Taiwan”), which was inactive in late 2021 and dissolved on May 20, 2022.
in Japan (“APRINOIA Japan”), APRINOIA Therapeutics Limited in Hong Kong (“APRINOIA HK”) and APRINOIA Therapeutics, LLC in the United States (“APRINOIA USA”). APRINOIA HK has a direct wholly-owned subsidiary, Suzhou APRINOIA Therapeutics Co, Ltd., in Suzhou (“APRINOIA Suzhou”). The Company had a subsidiary in Taiwan (APRINOIA Therapeutics Inc. (Taiwan) or “APRINOIA Taiwan”), which was inactive in late 2021 and dissolved on May 20, 2022.Level 1 – | Observable inputs such as unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date; |
Level 2 – | Inputs (other than quoted prices included in Level 1) are either directly or indirectly observable for the asset or liability. These include quoted prices for similar assets or liabilities in active markets and quoted prices for identical or similar assets or liabilities in markets that are not active; and |
Level 3 – | Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities. |
• | expenses incurred under agreements with organizations that support the Company’s drug discovery and development activities; |
• | expenses incurred in connection with the preclinical and clinical development of the Company’s product candidates and programs; |
• | costs related to CROs or CDMOs, that are primarily engaged to provide drug substance and product for our clinical trials, research and development programs, as well as investigative sites and consultants that conduct the Company’s clinical trials, nonclinical studies and other scientific development services; |
• | the costs of acquiring and manufacturing nonclinical and clinical trial materials, including manufacturing registration and validation batches; |
• | employee-related expenses, including salaries, related benefits and equity-based compensation expense, for employees engaged in research and development functions; |
• | costs related to compliance with quality and regulatory requirements; |
• | payments made under third-party licensing agreements; and |
• | direct and allocated costs related to facilities, information technology, personnel and other overhead. |
| | | Estimated Useful Life (in Years) | | | December 31, | ||||
| | | 2022 | | | 2021 | ||||
Machinery and equipment | | | 3-5 | | | $2,044 | | | $190 |
Computers and purchased software | | | 3-5 | | | 65 | | | 65 |
Leasehold improvements | | | lease term | | | 141 | | | 153 |
Furniture and fixtures | | | 3-5 | | | 39 | | | 43 |
Construction in progress | | | N/A | | | 350 | | | 271 |
Property and equipment, at cost | | | | | 2,639 | | | 722 | |
Less: accumulated depreciation and amortization | | | | | (486) | | | (213) | |
Property and equipment, net | | | | | $2,153 | | | $509 | |
| | | December 31, | ||||
| | | 2022 | | | 2021 | |
Prepayment | | | $592 | | | $1,531 |
Other receivables | | | 114 | | | 457 |
Deposit | | | 121 | | | 118 |
Total | | | 827 | | | 2,106 |
Less: current portion of prepaid expenses and other assets | | | (590) | | | (429) |
Total prepaid expenses, net of current portion and other long-term assets | | | $237 | | | $1,677 |
| | | December 31, | ||||
| | | 2022 | | | 2021 | |
Accrued expense and other payables | | | $1,112 | | | $2,569 |
Payroll payable | | | 1,053 | | | 273 |
Deferred revenue | | | 213 | | | 94 |
Other current liabilities | | | 88 | | | 57 |
Total accrued expenses and other current liabilities | | | $2,466 | | | $2,993 |
Lease Cost | | | 2022 | | | 2021 |
Operating lease cost | | | $137 | | | $112 |
Short-term lease cost | | | 249 | | | 206 |
Variable lease cost | | | 241 | | | 83 |
Total lease cost | | | $627 | | | $401 |
| | | | | |||
Other Information: | | | | | ||
Cash paid for amounts included in the measurement of lease liabilities: | | | | | ||
Operating cash flows for operating leases | | | $134 | | | $102 |
Right-of-Use assets obtained in the exchange for new operating lease liabilities | | | $— | | | $405 |
| | | 2022 | | | 2021 | |
Weighted-average remaining lease terms - operating leases | | | 1.34 years | | | 2.24 years |
Weighted average discount rate - operating leases | | | 4.02% | | | 3.84% |
Years Ended December 31 | | | Amount |
2023 | | | $128 |
2024 | | | 43 |
2025 | | | — |
2026 | | | — |
2027 | | | — |
Thereafter | | | — |
Total lease payments | | | 171 |
Less: imputed interest | | | (5) |
Total operating lease liabilities | | | $166 |
| | | December 31, 2022 | |
Principal | | | $1,450 |
Accrued interest | | | 2 |
Unamortized discount | | | (359) |
Total carrying value of convertible notes | | | 1,093 |
Derivative liabilities | | | 251 |
Total convertible notes and derivative liabilities | | | $1,344 |
| | | December 31, | ||||
| | | 2022 | | | 2021 | |
2021 Bank Loan | | | $— | | | $785 |
2022 June Bank Loan | | | 725 | | | — |
2022 July Bank Loan | | | 725 | | | — |
Total short-term borrowings from third parties | | | $1,450 | | | $785 |
| | | Fair Value as of December 31, 2022 Level 3 | |
Liabilities: | | | |
Derivative liabilities | | | $251 |
Total | | | $251 |
Significant Unobservable Inputs | | | Input Range | | | Weighted Average |
Discount Rate | | | 26.69% | | | 26.69% |
Expected term (in years) | | | 0.750 - 0.978 | | | 0.61 |
Probability Scenarios: | | | | | ||
Successful Financing | | | 85% | | | |
Delay/Renegotiation | | | 10% | | | |
Dissolution | | | 5% | | |
| | | Derivative Liabilities | |
Balance as of December 31, 2021 | | | $— |
Initial fair value of instrument | | | 251 |
Change in fair value | | | — |
Foreign currency translation adjustments | | | — |
Balance as of December 31, 2022 | | | $251 |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Current | | | | | ||
Federal | | | $(17) | | | $— |
State | | | — | | | — |
Foreign | | | — | | | — |
Total Current | | | $(17) | | | $— |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Foreign rate differential | | | 17.6% | | | 19.1% |
Valuation allowance | | | -8.6% | | | -9.0% |
Foreign derived intangible income | | | 0.0% | | | 0.0% |
Taxable cancellation of debt income | | | 0.0% | | | -3.2% |
Non-deductible R&D expenses | | | -8.9% | | | -6.3% |
Other | | | -0.2% | | | -0.6% |
Effective tax rate | | | -0.1% | | | 0.0% |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Deferred tax assets | | | | | ||
Tax loss carried forward | | | $6,255 | | | $4,135 |
Deferred advertising expenses | | | 7 | | | 7 |
Others | | | — | | | 123 |
Total deferred tax assets | | | 6,262 | | | 4,265 |
Less: valuation allowance | | | (6,257) | | | (4,257) |
Total deferred tax assets | | | 5 | | | 8 |
Deferred tax liabilities | | | | | ||
Depreciation | | | (5) | | | (8) |
Total deferred tax liabilities | | | (5) | | | (8) |
Deferred tax assets, net of valuation allowance and deferred tax liabilities | | | $— | | | $— |
| | | December 31, 2022 | |||||||||||||
| | | Preferred Shares Authorized | | | Preferred Shares Issued and Outstanding | | | Carrying Value | | | Liquidation Preference | | | Ordinary Shares Issuable Upon Conversion | |
Series B Preferred Shares | | | 15,000,000 | | | 13,883,000 | | | $10,995 | | | $11,106 | | | 13,883,000 |
Pre-Series C Preferred Shares | | | 11,973,336 | | | 11,973,336 | | | 14,918 | | | 14,967 | | | 11,973,336 |
Series C Preferred Shares | | | 30,000,000 | | | 27,766,265 | | | 39,963 | | | 40,093 | | | 27,766,265 |
Total | | | 56,973,336 | | | 53,622,601 | | | $65,876 | | | $66,166 | | | 53,622,601 |
| | | December 31, 2021 | |||||||||||||
| | | Preferred Shares Authorized | | | Preferred Shares Issued and Outstanding | | | Carrying Value | | | Liquidation Preference | | | Ordinary Shares Issuable Upon Conversion | |
Series B Preferred Shares | | | 15,000,000 | | | 13,883,000 | | | $10,995 | | | $11,106 | | | 13,883,000 |
Pre-Series C Preferred Shares | | | 11,973,336 | | | 11,973,336 | | | 14,918 | | | 14,967 | | | 11,973,336 |
Series C Preferred Shares | | | 30,000,000 | | | 22,070,063 | | | 31,000 | | | 31,093 | | | 22,070,063 |
Total | | | 56,973,336 | | | 47,926,399 | | | $56,913 | | | $57,166 | | | 47,926,399 |
a. | Dividend Rights - If declared, each holder of the Preferred Shares shall be entitled to receive dividends for the respective Preferred Share held by the holder, payable out of funds or assets when and as such funds or assets become legally available therefor, prior and in preference to, and satisfied before, any dividend on the Company’s ordinary shares. |
b. | Conversion Feature - The holder of Preferred Shares shall have the right to convert all or a portion of its Preferred Shares into the Company’s ordinary shares at the then-applicable Series B, Pre-Series C and Series C conversion price at any time without payment of additional consideration. The initial conversion price for each Preferred Shares shall be the original Series B, Pre-Series C and Series C purchase price paid, with the initial conversion made on a one-for-one basis, subject to the conversion price adjustment made. The Series B, Pre-Series C and Series C conversion price for the Preferred Shares shall be adjusted appropriately for subdivision or combination of the Company’s ordinary shares, distribution of dividends on the Company’s ordinary shares, capital reorganization, recapitalization, or reclassification of the Company’s ordinary shares. If the Company issues or proposes to issue any new securities at a per share price or conversion price less than the original Series B, Pre-Series C and Series C purchase price, such Series B, Pre-Series C and Series C conversion price for the Preferred Shares will be adjusted based on the broad-based weighted average method. |
c. | Liquidation preferences – In the event of any voluntary or involuntary liquidation, change in control, sale of substantially all of the Company’s assets or upon the occurrence of other transaction defined as a deemed liquidation event pursuant to the Company’s articles of association (a “Deemed Liquidation Event”), each shareholder of Preferred Shares shall be entitled to receive, prior to, and in preference to, any distribution of assets or property of the Company to the holders of the Company’s ordinary shares, in an amount per share equal to the Preferred Shares’ original purchase price, plus any declared unpaid dividends. The assets of the Company shall be distributed to the holders of Preferred Shares in the following order: Series C Preferred Shares, Pre-Series C Preferred Shares, Series B Preferred Shares. If upon any such liquidation event or a deemed liquidation event, the assets of the Company available for distribution to its shareholders is insufficient to pay all holders of redeemable convertible preferred shares the full amount to which they shall be entitled, the holders of redeemable convertible preferred shares shall share ratably in any distribution of the assets available for distribution in proportion to the respective amounts which would otherwise have been payable in the order indicated. |
d. | Voting Rights – Each holder of outstanding shares of Preferred Shares shall be entitled to cast the number of votes equal to the number of the Company’s ordinary shares into which the shares of such Preferred Shares held by such holder are convertible as of the record date for determining shareholders entitled to vote on such matter. |
e. | Redemption Rights – The holders of the Company’s Preferred Shares have no voluntary rights to redeem shares. Upon certain change in control events that are outside of the Company’s control, including sale of substantially all of the Company’s assets or the occurrence of a Deemed Liquidation Event, the holders of the Preferred Shares may cause redemption of the Preferred Shares. Accordingly, these shares are considered contingently redeemable and are classified as temporary equity on the accompanying consolidated balance sheets. |
| | | Number of Options | | | Weighted Average Exercise Price | | | Weighted Average Remaining Contractual Term (in years) | | | Aggregate Intrinsic Value | |
Outstanding as of December 31, 2020 | | | 11,753,673 | | | $0.13 | | | 3.53 | | | $3,115 |
Granted | | | 1,350,000 | | | 0.16 | | | — | | | — |
Exercised | | | (5,902,556) | | | 0.11 | | | — | | | — |
Outstanding as of December 31, 2021 | | | 7,201,117 | | | $0.15 | | | 3.19 | | | $2,757 |
Granted | | | 7,151,608 | | | 0.16 | | | — | | | — |
Exercised | | | (265,000) | | | 0.15 | | | — | | | — |
Outstanding as of December 31, 2022 | | | 14,087,725 | | | $0.15 | | | 3.43 | | | $3,939 |
Options exercisable as of December 31, 2022 | | | 11,889,225 | | | $0.15 | | | 1.19 | | | $3,333 |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Exercise period | | | 5 years | | | 5 years |
Volatility | | | 75.85% | | | 72.16% |
Risk-free interest rate | | | 2.91% | | | 1.13% |
Expected dividend yield | | | 0.00% | | | 0.00% |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
General and administrative | | | $1,143 | | | $687 |
Research and development | | | 768 | | | 227 |
Total share-based compensation expense | | | $1,911 | | | $914 |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Numerator: | | | | | ||
Net loss | | | $(28,231) | | | $(23,261) |
Net loss attributable to ordinary shareholders | | | $(28,231) | | | $(23,261) |
| | | | | |||
Denominator: | | | | | ||
Weighted-average shares outstanding used in calculating net loss per share – basic and diluted | | | 38,482,015 | | | 33,561,324 |
Net loss per share attributable to ordinary shareholders – basic and diluted | | | $(0.73) | | | $(0.69) |
| | | Year Ended December 31, | ||||
| | | 2022 | | | 2021 | |
Redeemable convertible preferred shares (as converted to ordinary shares) | | | 53,622,601 | | | 47,926,399 |
Outstanding options to purchase ordinary shares | | | 14,087,725 | | | 7,201,117 |
Total | | | 67,710,326 | | | 55,127,516 |
| | | June 30, 2023 | | | December 31, 2022 | |
Assets | | | | | ||
Current assets: | | | | | ||
Cash | | | $7,731 | | | $1,221 |
Prepaid expenses and other current assets | | | 1,162 | | | 590 |
Note receivable - related party | | | 660 | | | — |
Total current assets | | | 9,553 | | | 1,811 |
Property and equipment, net | | | 1,957 | | | 2,153 |
Deferred offering costs | | | 2,807 | | | 1,288 |
Operating lease right-of-use assets | | | 115 | | | 154 |
Prepaid expenses, net of current portion and other long-term assets | | | 44 | | | 237 |
Total assets | | | $14,476 | | | $5,643 |
Liabilities, Redeemable Convertible Preferred Shares, and Shareholders' Deficit | | | | | ||
Current liabilities: | | | | | ||
Accounts payable | | | $7,318 | | | $8,887 |
Accrued expenses and other current liabilities | | | 4,940 | | | 2,466 |
Operating lease liabilities, current | | | 102 | | | 124 |
Related party payable | | | — | | | 904 |
Short-term borrowings | | | 690 | | | 1,450 |
Convertible notes (including related parties convertible notes of $10,762 and $753 as of June 30, 2023 and December 31, 2022, respectively, net of debt discount and issuance costs) | | | 14,788 | | | 1,093 |
Derivative liabilities (including related parties derivative liabilities of $1,685 and $173 as of June 30, 2023 and December 31, 2022, respectively) | | | 2,679 | | | 251 |
Total current liabilities | | | 30,517 | | | 15,175 |
Operating lease liabilities, net of current portion | | | 15 | | | 42 |
Total liabilities | | | 30,532 | | | 15,217 |
Commitments and Contingencies (Note 11) | | | | | ||
Redeemable convertible preferred shares (Series B, Pre-C and C), $0.1 par value; 56,973,336 shares authorized; 53,622,601 shares issued and outstanding; redemption and liquidation value of $66,166 as of June 30, 2023 and December 31, 2022 | | | 65,876 | | | 65,876 |
Shareholders' deficit: | | | | | ||
Ordinary shares, $0.1 par value, 443,026,664 shares authorized; 40,492,206 and 38,617,056 shares issued and outstanding as of June 30, 2023 and December 31, 2022, respectively | | | 4,049 | | | 3,862 |
Additional paid-in capital | | | 13,176 | | | 12,296 |
Accumulated deficit | | | (97,823) | | | (90,638) |
Accumulated other comprehensive loss | | | (1,334) | | | (970) |
Total shareholders' deficit | | | (81,392) | | | (75,450) |
Total liabilities, redeemable convertible preferred shares, and shareholders' deficit | | | $14,476 | | | $5,643 |
| | | Six Months Ended June 30, | ||||
| | | 2023 | | | 2022 | |
Revenue | | | $496 | | | $295 |
Revenue – related party | | | 8,538 | | | — |
Total revenue | | | 9,034 | | | 295 |
Operating expenses | | | | | ||
Research and development | | | 11,067 | | | 11,289 |
General and administrative | | | 4,704 | | | 2,849 |
Total operating expenses | | | 15,771 | | | 14,138 |
Loss from operations | | | (6,737) | | | (13,843) |
Other (expense) income | | | | | ||
Interest expense, net | | | (1,444) | | | (13) |
Change in fair value of derivative liabilities | | | 498 | | | — |
Other income (expense), net | | | 551 | | | 186 |
Total other income (expense), net | | | (395) | | | 173 |
Loss before income taxes | | | (7,132) | | | (13,670) |
Provision for income taxes | | | 53 | | | 6 |
Net loss | | | (7,185) | | | (13,676) |
Net loss attributable to ordinary shareholders | | | $(7,185) | | | $(13,676) |
Net loss per share attributable to ordinary shareholders | | | | | ||
Basic and diluted | | | $(0.18) | | | $(0.36) |
Weighted-average shares outstanding | | | | | ||
Basic and diluted | | | 38,991,062 | | | 38,352,056 |
Comprehensive loss: | | | | | ||
Net loss | | | $(7,185) | | | $(13,676) |
Foreign currency translation adjustment, net of tax | | | (364) | | | (296) |
Total comprehensive loss | | | $(7,549) | | | $(13,972) |
| | | Redeemable Convertible Preferred | | | Ordinary Shares | | | Additional Paid-In Capital | | | Accumulated Deficit | | | Accumulated Other Comprehensive Loss | | | Total Shareholders' Deficit | |||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | ||||||||||||
Balance as of December 31, 2022 | | | 53,622,601 | | | $65,876 | | | 38,617,056 | | | $3,862 | | | $12,296 | | | $(90,638) | | | $(970) | | | $(75,450) |
Share-based compensation expense | | | — | | | — | | | — | | | — | | | 779 | | | — | | | — | | | 779 |
Share option exercised | | | — | | | — | | | 1,875,150 | | | 187 | | | 101 | | | — | | | — | | | 288 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (7,185) | | | — | | | (7,185) |
Foreign currency translation adjustment, net of tax | | | — | | | — | | | — | | | — | | | — | | | — | | | (364) | | | (364) |
Balance as of June 30, 2023 | | | 53,622,601 | | | $65,876 | | | 40,492,206 | | | $4,049 | | | $13,176 | | | $(97,823) | | | $(1,334) | | | $(81,932) |
| | | Redeemable Convertible Preferred | | | Ordinary Shares | | | Additional Paid-In Capital | | | Accumulated Deficit | | | Accumulated Other Comprehensive Loss | | | Total Shareholders' Deficit | |||||||
| | | Shares | | | Amount | | | Shares | | | Amount | | ||||||||||||
Balance as of December 31, 2021 | | | 47,926,399 | | | $56,913 | | | 38,352,056 | | | $3,835 | | | $10,373 | | | $(62,407) | | | $(489) | | | $(48,688) |
Issuance of series C redeemable convertible preferred shares, net of issuance costs | | | 5,696,202 | | | 8,963 | | | — | | | — | | | — | | | — | | | — | | | — |
Share-based compensation expense | | | — | | | — | | | — | | | — | | | 163 | | | — | | | — | | | 163 |
Share option exercised | | | — | | | — | | | 265,000 | | | 27 | | | 12 | | | — | | | — | | | 39 |
Net loss | | | — | | | — | | | — | | | — | | | — | | | (13,676) | | | — | | | (13,676) |
Foreign currency translation adjustment, net of tax | | | — | | | — | | | — | | | — | | | — | | | — | | | (296) | | | (296) |
Balance as of June 30, 2022 | | | 53,622,601 | | | $65,876 | | | 38,617,056 | | | $3,862 | | | $10,548 | | | $(76,083) | | | $(785) | | | $(62,458) |
| | | Six Months Ended June 30, | ||||
| | | 2023 | | | 2022 | |
Operating Activities: | | | | | ||
Net loss | | | $(7,185) | | | $(13,676) |
Adjustments to reconcile net loss to net cash used in operating activities: | | | | | ||
Depreciation expense | | | 220 | | | 96 |
Non-cash interest expense | | | 1,429 | | | — |
Amortization of operating lease right-of-use assets | | | 63 | | | 86 |
Share-based compensation expense | | | 779 | | | 163 |
Change in fair value of derivative liability | | | (498) | | | — |
Changes in operating assets and liabilities: | | | | | ||
Accounts receivable | | | — | | | (49) |
Prepaid expenses and other current assets | | | (555) | | | 969 |
Accounts payable | | | (1,486) | | | 3,180 |
Accrued expenses and other current liabilities | | | 1,665 | | | (1,009) |
Operating lease obligations | | | (72) | | | (84) |
Prepaid expenses, net of current portion and other long-term assets | | | 189 | | | 516 |
Net cash used in operating activities | | | (5,451) | | | (9,808) |
Investing Activities: | | | | | ||
Purchase of property and equipment | | | (121) | | | (878) |
Note Receivable - Related Party | | | (660) | | | — |
Net cash used in investing activities | | | (781) | | | (878) |
Financing Activities: | | | | | ||
Proceeds from issuance of preferred shares, net of issuance costs | | | — | | | 8,963 |
Proceeds from exercise of share options | | | 288 | | | 39 |
Proceeds from issuance of convertible notes (including proceeds from a related party convertible note of $10,750 and $0 as of June 30, 2023 and 2022, respectively) | | | 15,208 | | | — |
Proceeds from short-term borrowings | | | — | | | 772 |
Proceeds from related party payable | | | 280 | | | — |
Repayment of related party payable | | | (1,174) | | | — |
Repayment of short-term borrowings | | | (722) | | | (772) |
Deferred offering costs associated with the Business Combination | | | (593) | | | — |
Net cash provided by financing activities | | | 13,287 | | | 9,002 |
Effect of exchange rates on cash | | | (545) | | | (303) |
Net increase (decrease) in cash | | | 6,510 | | | (1,987) |
Cash at beginning of period | | | 1,221 | | | 9,674 |
Cash at end of period | | | $7,731 | | | $7,687 |
Supplemental disclosure of cash flow information: | | | | | ||
Cash paid for loan interest | | | $38 | | | $14 |
Cash paid for income tax | | | $22 | | | $— |
Supplemental cash flow information on non-cash investing and financing activities: | | | | | ||
Right-of-use assets obtained in exchange of lease liabilities | | | $29 | | | $— |
Debt issuance cost associated with convertible notes included in accounts payable | | | $18 | | | $— |
Issuance of derivative instrument related to convertible notes | | | $2,925 | | | $— |
Deferred offering costs associated with the Business Combination in accrued expenses and other current liabilities | | | $926 | | | $— |
 Japan (“APRINOIA Japan”), APRINOIA Therapeutics Limited in Hong Kong (“APRINOIA HK”) and APRINOIA Therapeutics, LLC in the United States (“APRINOIA USA”). APRINOIA HK has a direct wholly-owned subsidiary, Suzhou APRINOIA Therapeutics Co, Ltd., in Suzhou (“APRINOIA Suzhou”). The Company had a subsidiary in Taiwan (APRINOIA Therapeutics Inc. (Taiwan) or “APRINOIA Taiwan”), which was inactive in late 2021 and dissolved on May 20, 2022.
Japan (“APRINOIA Japan”), APRINOIA Therapeutics Limited in Hong Kong (“APRINOIA HK”) and APRINOIA Therapeutics, LLC in the United States (“APRINOIA USA”). APRINOIA HK has a direct wholly-owned subsidiary, Suzhou APRINOIA Therapeutics Co, Ltd., in Suzhou (“APRINOIA Suzhou”). The Company had a subsidiary in Taiwan (APRINOIA Therapeutics Inc. (Taiwan) or “APRINOIA Taiwan”), which was inactive in late 2021 and dissolved on May 20, 2022. | | | June 30, 2023 | | | December 31, 2022 | |
Prepayment | | | $807 | | | $592 |
Other receivables | | | 290 | | | 114 |
Deposit | | | 109 | | | 121 |
Total | | | 1,206 | | | 827 |
Less: current portion of prepaid expenses and other assets | | | (1,162) | | | (590) |
Total prepaid expenses, net of current portion and other long-term assets | | | $44 | | | $237 |
| | | June 30, 2023 | | | December 31, 2022 | |
Accrued expense and other payables | | | $3,889 | | | $1,112 |
Payroll payable | | | 866 | | | 1,053 |
Deferred revenue | | | 16 | | | 213 |
Other current liabilities | | | 169 | | | 88 |
Total accrued expenses and other current liabilities | | | $4,940 | | | $2,466 |
Lease Cost | | | Six Months Ended June 30, | |||
| | | 2023 | | | 2022 | |
Operating lease cost | | | $68 | | | $70 |
Short-term lease cost | | | 105 | | | 130 |
Variable lease cost | | | 163 | | | 130 |
Total lease cost | | | $336 | | | $330 |
Other Information: | | | | | ||
Cash paid for amounts included in the measurement of lease liabilities: | | | | | ||
Operating cash flows for operating leases | | | $76 | | | $66 |
Right-of-Use assets obtained in the exchange for new operating lease liabilities | | | $29 | | | $— |
| | | June 30, 2023 | | | December 31, 2022 | |
Weighted-average remaining lease terms - operating leases | | | 1.1 years | | | 1.34 years |
Weighted average discount rate - operating leases | | | 6.68% | | | 4.02% |
Years Ending | | | Amount |
2023 | | | $61 |
2024 | | | 57 |
2025 | | | 5 |
2026 | | | — |
2027 | | | — |
2028 and thereafter | | | — |
Total lease payments | | | 123 |
Less: imputed interest | | | (6) |
Total operating lease liabilities | | | $117 |
| | | June 30, 2023 | | | December 31, 2022 | |
Principal | | | $16,658 | | | $1,450 |
Accrued interest | | | 311 | | | 2 |
Unamortized discount | | | (2,181) | | | (359) |
Total carrying value of convertible notes | | | 14,788 | | | 1,093 |
Derivative liabilities | | | 2,679 | | | 251 |
Total convertible notes and derivative liabilities | | | $17,467 | | | $1,344 |
| | | June 30, 2023 | | | December 31, 2022 | |
2022 June Bank Loan | | | $— | | | $725 |
2022 July Bank Loan | | | 690 | | | 725 |
Total short-term borrowings from third parties | | | $690 | | | $1,450 |
| | | Level 3 Fair Value | ||||
| | | As of June 30, 2023 | | | As of December 31, 2022 | |
Liabilities: | | | | | ||
Derivative liabilities | | | $2,679 | | | $251 |
Total | | | $2,679 | | | $251 |
| | | June 30, 2023 | | | December 31, 2022 | |||||||
Significant Unobservable Inputs | | | Input Range | | | Weighted Average | | | Input Range | | | Weighted Average |
Discount Rate | | | 28.41% - 32.44% | | | 30.60% | | | 26.69% | | | 26.69% |
Expected term (in years) | | | 0.500 - 1.000 | | | 0.45 | | | 0.750 - 0.978 | | | 0.61 |
Probability Scenarios: | | | | | | | | | ||||
Successful Financing/IPO | | | 85% | | | | | 85% | | | ||
Delayed Offering/Renegotiation | | | 10% | | | | | 10% | | | ||
Dissolution | | | 5% | | | | | 5% | | | ||
| | | Derivative Liabilities | |
Balance as of December 31, 2022 | | | $251 |
Initial fair value of instrument | | | 2,926 |
Change in fair value | | | (498) |
Foreign currency translation adjustments | | | — |
Balance as of June 30, 2023 | | | $2,679 |
| | | Number of Options | | | Weighted Average Exercise Price | | | Weighted Average Remaining Contractual Term (in years) | | | Aggregate Intrinsic Value | |
Outstanding as of December 31, 2022 | | | 14,087,725 | | | $0.15 | | | 3.43 | | | $3,939 |
Granted | | | 1,385,000 | | | 0.16 | | | — | | | — |
Exercised | | | (1,875,150) | | | 0.15 | | | — | | | — |
Cancelled or forfeited | | | (427,608) | | | 0.12 | | | — | | | — |
Outstanding as of June 30, 2023 | | | 13,169,967 | | | $0.16 | | | 3.11 | | | $10,232 |
Options exercisable as of June 30, 2023 | | | 11,252,292 | | | $0.15 | | | 2.92 | | | $8,747 |
Exercise period | | | 5 years |
Volatility | | | 76.80% - 79.86% |
Risk-free interest rate | | | 3.50% - 3.58% |
Expected dividend yield | | | 0.00% |
| | | Six Months Ended June 30, | ||||
| | | 2023 | | | 2022 | |
General and administrative | | | $360 | | | $9 |
Research and development | | | 419 | | | 154 |
Total share-based compensation expense | | | $779 | | | $163 |
| | | Six Months Ended June 30, | ||||
| | | 2023 | | | 2022 | |
Numerator: | | | | | ||
Net loss | | | $(7,185) | | | $(13,676) |
Net loss attributable to ordinary shareholders | | | $(7,185) | | | $(13,676) |
| | | | | |||
Denominator: | | | | | ||
Weighted-average shares outstanding used in calculating net loss per share – basic and diluted | | | 38,991,062 | | | 38,352,056 |
Net loss per share attributable to ordinary shareholders – basic and diluted | | | $(0.18) | | | $(0.36) |
| | | Six Months Ended June 30, | ||||
| | | 2023 | | | 2022 | |
Redeemable convertible preferred shares (as converted to ordinary shares) | | | 53,622,601 | | | 53,622,601 |
Outstanding options to purchase ordinary shares | | | 13,169,967 | | | 6,936,117 |
Total | | | 66,792,568 | | | 60,558,718 |
Item 6. | Indemnification of Directors and Officers |
Item 7. | Recent Sales of Unregistered Securities. |
Item 8. | Exhibits and Financial Statement Schedules. |
Item 9. | Undertakings. |
Exhibit Number | | | Description of Document |
1.1* | | | Form of Underwriting Agreement |
| | | Memorandum and Articles of Association of the Registrant | |
3.2* | | | Form of Amended and Restated Memorandum and Articles of Association of the Registrant (to be effective immediately prior to the completion of this offering) |
4.1* | | | Registrant’s Specimen Certificate for ordinary shares |
| | | Shareholders’ Agreement, dated as of September 24, 2021, among the Registrant, the holders of the Registrant’s ordinary and preferred shares and certain parties thereto | |
| | | Addendum to Shareholders’ Agreement, dated November 16, 2021, among the Registrant, the holders of the Registrant’s ordinary and preferred shares and certain parties thereto | |
5.1* | | | Opinion of Maples and Calder (Hong Kong) LLP regarding the validity of the ordinary shares being registered |
8.1* | | | Opinion of Maples and Calder (Hong Kong) LLP regarding certain Cayman Islands tax matters (included in Exhibit 5.1) |
| | | 2018 Equity Incentive Plan | |
| | | 2019 Equity Incentive Plan #2 | |
| | | 2021 Equity Incentive Plan #3 | |
| | | 2022 Equity Incentive Plan #4 | |
10.5* | | | Form of Indemnification Agreement between the Registrant and a director of Registrant |
10.6* | | | Form of Employment Agreement between the Registrant and an executive officer of Registrant |
| | | Exclusive License Agreement dated October 20, 2016 among National Institutes for Quantum and Radiological Science and Technology and APRINOIA Therapeutics Inc. (a corporation of Taiwan) | |
| | | Amendment Agreement of Exclusive License Agreement dated January 11, 2018 among National Institutes for Quantum and Radiological Science and Technology, APRINOIA Therapeutics Inc. (a corporation of Taiwan) and APRINOIA Therapeutics Inc. (a corporation of Cayman Islands) | |
| | | Amendment Number Two Agreement of Exclusive License Agreement dated May 27, 2019 among National Institutes for Quantum and Radiological Science and Technology, APRINOIA Therapeutics Inc. (a corporation of Taiwan) and APRINOIA Therapeutics Inc. (a corporation of Cayman Islands) | |
| | | Amendment Number Three Agreement of Exclusive License Agreement dated March 16, 2021 among National Institutes for Quantum and Radiological Science and Technology, APRINOIA Therapeutics Inc. (a corporation of Taiwan), APRINOIA Therapeutics Inc. (a corporation of Japan) and Suzhou APRINOIA Therapeutics Co., Ltd. | |
| | | Amendment Number Four Agreement of Exclusive License Agreement dated November 21, 2022 among National Institutes for Quantum and Radiological Science and Technology, APRINOIA Therapeutics Inc. (a corporation of Japan) and Suzhou APRINOIA Therapeutics Co., Ltd. | |
| | | License and Commercialization Agreement dated March 30, 2023 among APRINOIA Therapeutics Inc. (a corporation of Japan), APRINOIA Therapeutics Inc. (a corporation of the Cayman Islands), Yantai Yitai Pharmaceutical Technology Co., Ltd. and Yantai Dongcheng Pharmaceutical Group Co., Ltd. | |
| | | Assignment and Consulting Service Agreement dated March 30, 2023 among Suzhou Aprinoia Therapeutics Co., Ltd., APRINOIA Therapeutics Inc. (a corporation of the Cayman Islands), Yantai Yitai Pharmaceutical Technology Co., Ltd. and Yantai Dongcheng Pharmaceutical Group Co., Ltd. | |
| | | Research Collaboration Agreement dated December 20, 2018 among H. Lundbeck A/S, AbbVie Inc. and APRINOIA Therapeutics Inc. (a corporation of Taiwan) | |
| | | First Amendment, effective February 20, 2019, to the Research Collaboration Agreement among H. Lundbeck A/S, AbbVie Inc. and APRINOIA Therapeutics Inc. (a corporation of Taiwan) | |
| | | Second Amendment, effective December 1, 2019, to the Research Collaboration Agreement among H. Lundbeck A/S, AbbVie Inc. and APRINOIA Therapeutics Inc. (a corporation of Taiwan) | |
| | | Third Amendment, effective November 30, 2020, to the Research Collaboration Agreement among H. Lundbeck A/S, AbbVie Inc. and APRINOIA Therapeutics Inc. (a corporation of Taiwan) | |
| | | Assignment Agreement dated August 4, 2021 among H. Lundbeck A/S, AbbVie Inc., APRINOIA Therapeutics Inc. (a corporation of Taiwan) and APRINOIA Therapeutics Limited |
Exhibit Number | | | Description of Document |
| | | Fourth Amendment, effective January 25, 2022, to the Research Collaboration Agreement among H. Lundbeck A/S, AbbVie Inc. and APRINOIA Therapeutics Limited | |
| | | Fifth Amendment, effective January 25, 2023, to the Research Collaboration Agreement among H. Lundbeck A/S, AbbVie Inc. and APRINOIA Therapeutics Limited | |
| | | Sixth Amendment, effective September 20, 2023, to the Research Collaboration Agreement among H. Lundbeck A/S, AbbVie Inc. and APRINOIA Therapeutics Limited | |
14.1* | | | Code of Business Conduct and Ethics of the Registrant |
| | | Principal subsidiaries of the Registrant | |
| | | Consent of MaloneBailey, LLP | |
23.2* | | | Consent of Maples and Calder (Hong Kong) LLP (included in Exhibit 5.1) |
| | | Powers of Attorney (included on signature page) | |
| | | Filing Fee Table |
* | To be filed by amendment. |
** | Previously filed. |
† | Schedules and/or certain portions of the exhibits omitted pursuant to Item 601(b)(10) of Regulation S-K. The registrant agrees to furnish supplementally a copy of such schedules, or any section thereof, to the SEC upon request. |
| | | APRINOIA Therapeutics Inc. | ||||
| | | | | |||
| | | By: | | | /s/ Mark S. Shearman | |
| | | | | Name: Mark S. Shearman | ||
| | | | | Title: Chief Executive Officer | ||
Signature | | | Title | | | Date |
* | | | Founder and Chairman of the Board | | | February 26, 2024 |
Ming-Kuei Jang | | |||||
| | | | | |||
/s/ Mark S. Shearman | | | Chief Executive Officer and Director (principal executive officer) | | | February 26, 2024 |
Mark S. Shearman | | |||||
| | | | | |||
/s/ Brian Achenbach | | | Chief Financial Officer (principal financial and accounting officer) | | | February 26, 2024 |
Brian Achenbach | | |||||
| | | | | |||
| | | | | |||
* | | | Director | | | February 26, 2024 |
Michael Xin Hui | | |||||
| | | | | |||
* | | | Director | | | February 26, 2024 |
Zhigang Luo | | |||||
| | | | | |||
* | | | Director | | | February 26, 2024 |
Roger James Pomerantz | | |||||
| | ||||||
*By: /s/ Mark S. Shearman | | |||||
Mark S. Shearman | | |||||
Attorney-in-Fact | | |||||
| |
