|
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
|
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
|
|
|
|
|
(State or other jurisdiction of incorporation or organization)
|
(I.R.S. Employer Identification No.)
|
|
|
||
|
|
|
|
|
(Address of principal executive offices)
|
(Zip Code)
|
|
Title of each class
|
Trading Symbols(s)
|
Name of each exchange on which registered
|
||
|
|
|
|
||
|
|
|
|
|
|
☒
|
Accelerated filer
|
☐
|
|
Non-accelerated filer
|
☐
|
Smaller reporting company
|
|
|
Emerging growth company
|
|
|
|
|
Page
|
|
|
|
|
|
|
|
|
3 |
||
|
|
6 | ||
|
Item 1.
|
6 | ||
|
Item 1A.
|
37 |
||
|
Item 1B.
|
74 |
||
|
Item 2.
|
74 |
||
|
Item 3.
|
74 |
||
|
Item 4.
|
74 |
||
|
|
|||
|
|
75 |
||
|
Item 5.
|
75 |
||
|
Item 6.
|
75 |
||
|
Item 7.
|
76 |
||
|
Item 7A.
|
87 |
||
|
Item 8.
|
87 |
||
|
Item 9.
|
87 |
||
|
Item 9A.
|
87 |
||
|
Item 9B.
|
89 |
||
|
Item 9C.
|
89 |
||
|
|
89 |
||
|
Item 10.
|
89 |
||
|
Item 11.
|
96 |
||
|
Item 12.
|
115 |
||
|
Item 13.
|
118 |
||
|
Item 14.
|
123 |
||
|
|
|
||
| 125 |
|||
|
Item 15.
|
125 |
||
|
Item 16.
|
128 |
||
|
129
|
|||
| ● |
the ability to recognize the anticipated benefits of the Business Combination, which may be affected by, among other things, competition and our ability to grow and
manage growth profitably and retain our key employees;
|
| ● |
the ability to maintain the listing of our Class A common stock on The Nasdaq Stock Market LLC (“Nasdaq”);
|
| ● |
changes in applicable laws or regulations;
|
| ● |
our ability to raise financing in the future;
|
| ● |
the success, cost and timing of our product development activities;
|
| ● |
the commercialization and adoption of our existing products and the success of any product we may offer in the future;
|
| ● |
the potential attributes and benefits of our products once commercialized;
|
| ● |
our ability to obtain and maintain regulatory approval for our products, and any related restrictions and limitations of any approved product;
|
| ● |
our ability to identify, in-license or acquire additional technology;
|
| ● |
our ability to maintain our existing license agreements and manufacturing arrangements;
|
| ● |
our ability to compete with other companies currently marketing or engaged in the development of products and services that serve customers engaged in proteomic
analysis, many of which have greater financial and marketing resources than us;
|
| ● |
the size and growth potential of the markets for our products, and the ability of each product to serve those markets once commercialized, either alone or in
partnership with others;
|
| ● |
our estimates regarding expenses, future revenue, capital requirements and needs for additional financing;
|
| ● |
our financial performance; and
|
| ● |
the impact of the COVID-19 pandemic on our business.
|
|
•
|
We are an early-stage life sciences technology company with a history of net losses, which we expect to continue, and we may not be able to generate
meaningful revenues or achieve and sustain profitability in the future.
|
|
•
|
We have a limited operating history, which may make it difficult to evaluate the prospects for our future viability and predict our future
performance.
|
|
•
|
We may need to raise additional capital to fund commercialization plans for our products, including manufacturing, sales and marketing
activities, expand our investments in research, and development and commercialize new products and applications.
|
|
•
|
We have identified material weaknesses in our internal control over financial reporting. If we are unable to develop and maintain an effective
system of internal control over financial reporting, we may not be able to accurately report our financial results in a timely manner, which may adversely affect investor confidence in us and materially and adversely affect our business
and operating results.
|
|
•
|
We have not yet commercially launched our products, and we may not be able to successfully commercially launch our products as planned.
|
|
•
|
Because we are a “controlled company” within the meaning of the Nasdaq rules, our stockholders may not have certain corporate governance
protections that are available to stockholders of companies that are not controlled companies.
|
|
•
|
The dual class structure of our common stock has the effect of concentrating voting power with Jonathan M. Rothberg, Ph.D., our Interim Chief
Executive Officer and Executive Chairman of the board of directors and Legacy Quantum-Si’s Founder, which will limit an investor’s ability to influence the outcome of important transactions, including a change in control.
|
|
•
|
Even if we commercially launch our products, our success depends on broad scientific and market acceptance, which we may fail to achieve.
|
|
•
|
The size of the markets for our products may be smaller than estimated, and new market opportunities may not develop as quickly as we expect, or
at all, limiting our ability to successfully sell our products.
|
|
•
|
The COVID-19 pandemic and efforts to reduce its spread have adversely impacted, and are expected to continue to materially and adversely impact,
our business and operations.
|
|
•
|
If we do not sustain or successfully manage our anticipated growth, our business and prospects will be harmed.
|
|
•
|
We are currently undergoing a leadership transition, and we depend on our key personnel and other highly qualified personnel, and
if we are unable to recruit, train and retain our personnel in the future, we may not achieve our goals.
|
|
•
|
We expect to be dependent upon revenue generated from the sales of our initial products from the time they are commercialized through the
foreseeable future.
|
|
•
|
We rely on a small number of contract manufacturers to manufacture and supply our instruments. If these manufacturers should fail or not perform
satisfactorily, our ability to commercialize and supply our instruments would be adversely affected.
|
|
•
|
If we do not successfully develop and deploy our software, our commercialization efforts and therefore business and results of operations could
suffer.
|
|
•
|
We have limited experience producing and supplying our products, and we may be unable to consistently manufacture or source our instruments and
consumables to the necessary specifications or in quantities necessary to meet demand on a timely basis and at acceptable performance and cost levels.
|
|
•
|
The life sciences technology market is highly competitive. If we fail to compete effectively, our business and results of operations will
suffer.
|
|
•
|
If we elect to label and promote any of our products as clinical diagnostics or medical devices, we would be required to obtain prior marketing
authorization from the U.S. Food and Drug Administration (“FDA”), which would take significant time and expense and could fail to result in FDA marketing authorization of the device for the intended use or uses we believe are commercially
attractive.
|
|
•
|
Our products, if used for the diagnosis of disease, could be subject to government regulation, and the regulatory approval and maintenance
process for such products may be expensive, time-consuming, and uncertain both in timing and in outcome.
|
|
•
|
Our research use only (“RUO”) products could become subject to government regulation as medical devices by the FDA and other regulatory agencies
even if we do not elect to seek regulatory authorization to market our products for diagnostic purposes, which would adversely impact our ability to market and sell our products and harm our business.
|
|
•
|
If we are unable to obtain and maintain and enforce sufficient intellectual property protection for our products and technology, or if the scope
of the intellectual property protection obtained is not sufficiently broad, our competitors could develop and commercialize products similar or identical to ours, and our ability to successfully commercialize our products may be impaired.
|
|
•
|
We may not be able to protect our intellectual property rights throughout the world.
|
| • |
Personalized medicine: tailoring of disease treatment based on genomic data and real-time proteomic data;
|
| • |
Biomarker discovery: identification of protein markers for disease identification;
|
| • |
Drug discovery and development: identification of potential drug candidates and aid in the development of the drug;
|
| • |
Systems biology: system-wide investigations of disease pathways to identify biomarkers, drug action, toxicity, efficacy and
resistance;
|
| • |
Industry / agriculture: bioproduction and study of plant-pathogen interaction (e.g. crop engineering for drought
resistance); and
|
| • |
Food science: identification of allergies, understanding an improvement of nutritional values and food quality and safety
control.
|
| • |
Lower-plex methods. Lower-plex proteomic analysis methods include immunoassay, Gel, and chromatography based methods.
Immunoassay based methods rely on the availability of antibodies targeting specific proteins or epitopes as a way to identify and quantify protein expression levels. Changes or modifications to the protein may prevent the antibody from
binding, resulting in missed identification. Gel based methods like Western blots were the first proteomic technique developed. They utilize an electric current to separate proteins in a gel based on their size and charge, prior to
further analysis by a mass spectrometer (MS) instrument. Chromatography based methods use ion-exchange chromatography to separate and purify proteins from complex biological mixtures. The purified proteins can then be analyzed using a
mass spectrometer.
|
| • |
Higher-plex methods. Higher-plex proteomic analysis methods include protein microarrays and mass spectrometry instruments.
Existing high-plex proteomic technologies, however, often have tradeoffs between sensitivity and dynamic range — current technologies that are able to analyze the proteome at higher plex, often do so with lower sensitivity and resolution.
Protein microarrays apply small amounts of sample to a “glass chip” where specific antibodies are used to capture target proteins to measure the expression levels and binding affinities of proteins. The most common way researchers
currently analyze proteins is through the use of mass spectrometry. Mass spectrometry is a method for the mass determination and characterization of proteins, and its direct applications include protein identification and
post-translational modifications, elucidation of protein complexes, their subunits and functional interactions, as well as global measurement of proteins in proteomics. Some newer technologies have addressed certain limitations of these
methods, yet still require separate peptide drying or are reliant on existing mass spectrometry instruments. With an estimated 16,000 mass spectrometry instruments installed worldwide specifically for proteomics analysis, we believe the
cost of $250,000 to $1,000,000 or more per new instrument, according to research by DeciBio, LLC, limits access to proteomics research and we believe currently limits the size and growth of the overall proteomics industry.
|
| • |
Limitations of biased approaches. Unlike with nucleic acids, there is no ability to amplify individual proteins for analysis.
Without an amplification method, typical workflows rely on analyte-specific reagents (ASRs) for protein detection. ASRs comprise a variety of molecules, such as antibodies or aptamers, that bind to specific regions, rather than individual
amino acids, and therefore may not detect the presence of a known protein variants. For instance, the average binding site of an ASR is an epitope with a length of five (5) to eight (8) amino acids, whereas the average length of a human
protein is approximately 470 amino acids. While ASRs are prevalent and readily available, inherent limitations in how these molecules interact with proteins for various detection platforms limit their use for resolving protein sequences at
single amino acid resolution.
|
| • |
Mass spectrometry tools have a high cost of purchase and ownership. For more than a decade, mass spectrometry has been the
dominant tool for an unbiased approach to protein analysis. Shotgun proteomics, or studying pieces of proteins that have been broken apart, typically utilizes mass spectrometry and mass spectrometry workflows, allowing for the
interrogation of individual peptides and protein sequences. However, these techniques are generally complex, lengthy, expensive, laborious and require extensive data analysis. Taken together, these factors limit the scalability of this
approach and broad adoption of the technology in the market. Comparatively, targeted or biased methods are scalable but only enable interrogation of a small number of targeted proteins per sample. Biased approaches lack the breadth and
depth necessary to catalog new protein variants. Users are therefore forced to choose between breadth with mass spectrometry or scalability with other biased technologies, or limited alternatives that can address both needs.
|
| • |
Low levels of resolution and sensitivity. We believe successful technologies for use in broad proteomic and clinical testing
generally require high levels of specificity and sensitivity as well as the ability to scale to reliably meet volume demand. Current sensitivity and dynamic range restrictions make legacy technologies, such as mass spectrometry, difficult
to use with liquid samples and restrict the ability to analyze at single molecule resolution.
|
| • |
There is no method that allows for massively parallel proteomic sequencing. The ability to perform massively parallel
sampling in genomics has helped transform unbiased genomic analysis. Prior to NGS, large scale genomic analysis was limited, as it required expensive instruments and intensive labor for sample preparation and data analysis. The
introduction of NGS enabled massively parallel sampling of small fragments of DNA, enabling sequencing of tens of billions of DNA fragments per sample. By allowing the technology to scale analysis while also reducing costs, NGS enabled
numerous end-market opportunities, including routine cancer panel testing, clinical exomes and other DNA-based assays. Proteomics is currently facing similar limitations, with no existing technology that enables massively parallel sampling
of proteins.
|
| • |
There is no end-to-end platform to enable a true sample to answer assay. While there have been some improvements to proteomic
technologies, there remain numerous key limitations in typical proteomic analysis. Experiments often require input and oversight from highly trained mass spectrometry technicians, which often requires specialty training for both mass
spectrometry instrument operation and data analysis. Further, these workflows can be tedious and require extensive hands-on-time to perform, inherently limiting sample throughput.
|
| • |
Costly and complex data analysis. We believe the critical unmet needs remaining in proteomic analysis relate to cost,
accessibility and simplicity. Given the complex and dynamic aspects of proteins, proteomic analysis can generate vast amounts of data that can be difficult to analyze to arrive at a biologically relevant answer. Currently, the complexity
of the analysis is also costly, due to the data processing and analysis infrastructure that is often required.
|




| • |
Transport and meter out small volumes of reagents/samples between reservoirs;
|
| • |
Perform chemical or enzymatic incubations with or without temperature control;
|
| • |
Purify target analyte; and
|
| • |
Automate sample prep through to library creation.
|

| • |
What protein is present? Amino acid resolution can provide insight into more than just whether a protein is present or
absent. The sequence information could also indicate what version of the protein is present and how it has been changed from the normal version.
|
| • |
How much of the protein is present? A digital quantification provides precise protein abundance, not an analog theoretical
abundance based on a colorimetric or mass abundance readout.
|
| • |
How has the protein been modified? Single-molecule sensitivity could show how the protein has been post-translationally
modified thus providing greater insights to its role in the context of biological processes within the cell.
|


| • |
User management for secured data access;
|
| • |
Light-weight library information management system for data management;
|
| • |
Multi-tenancy to enable data sharing and collaborations; and
|
| • |
Application store to power a new generation of applications.
|
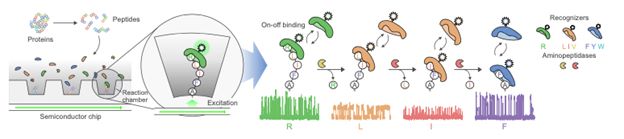
| • |
Addressing a large and growing proteomics market poised for technological disruption. We aim to transform single molecule
protein analysis and to democratize proteomic analysis by directly enabling users to unlock significant and unbiased biological insights through improved resolution and access to the proteome. We are developing products to serve customers
within the broader proteomics market, which was estimated to be $36.0 billion in 2020 according to Allied Market Research and is expected to grow to over $70.0 billion by 2025, which represents an approximate 14% CAGR. We believe that the
current addressable market for the products we are developing is $21.0 billion and comprises users across three core groups — users of legacy proteomics technologies, users of benchtop DNA sequencing technologies, and users of other protein
analyzers. Some of these technologies have existed for decades, yet have not provided users unbiased access to the proteome in a simple, cost effective, and scalable manner, which we believe our platform will provide. We believe that our
platform has the potential to enable users to study the proteome similar to the manner in which NGS technologies have transformed the study of the genome.
|
| • |
Differentiated single molecule detection providing the ultimate level of protein sensitivity and specificity. Our platform is
based on our proprietary semiconductor chip designed to enable measurements at the ultimate level of sensitivity and specificity, single molecules. By enabling true single molecule detection, we are not reliant on ensemble measurements,
which can often vary from sample to sample and even run to run.
|
| • |
Amino acid resolution and Post-Translational Modification (PTM) detection. Moving beyond simple confirmatory information
provided by affinity-based platforms, our platform delivers amino acid resolution shifting the output from analog to digital. The ability to also identify PTMs could provide novel insights into how pathways are turned on/off during in the
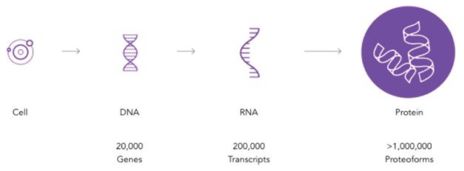
context of disease and ultimately improve our understanding of the estimated 1 million + proteoforms.
|
| • |
Real-time data processing and open cloud platform provides fast, simple data analysis. During sequencing our Platinum
instrument is designed to stream data to the cloud in real-time, which could allow for real-time analysis to enable faster time to results. In addition, we have developed our cloud-based platform to provide key tools needed to streamline
use of the platform such as secure access, data management, and an open platform where developers can create new analytical workflows to run in our cloud and share them easily with other users.
|
| • |
Innovative proprietary end-to-end proteomic platform offering differentiated full suite of protein sequencing solutions. We
believe that our platform will enable full end-to-end proteomics workflow solution spanning sample preparation through protein sequencing and analysis, allowing our customers a seamless opportunity to perform proteomic studies at scale. We
also believe that we are the first company to successfully enable NGPS on a semiconductor chip, thus digitizing a substantial proteomics opportunity. We believe the digital nature of our readout provides an accurate and repeatable
quantification of proteins in the sample and could scale to enable billions of data points working at the ultimate level of sensitivity — single molecule resolution.
|
| • |
Platform to enable democratized access to proteomics tools. Our platform is designed to provide an easy-to-use workflow with
the potential to enable users the ability to better characterize and understand the full complexity of the proteome in an unbiased fashion. Current workflows are typically disaggregated, expensive, require significant training to operate,
and are often performed in a separate specialty laboratory. We aim for our technology platform to be broadly available across pharmaceutical and academic research centers, basic research labs, and other healthcare centers and clinical
laboratories (for RUO until appropriate regulatory authorization is secured to allow clinical or diagnostic uses) at a price point that is a significant discount to most legacy technologies. The reduction in both cost and complexity could
allow for rapid adoption, whether a user is replacing a legacy technology or buying a new instrument. In addition to appealing to users of existing proteomics tools, we believe that our proteomics platform will appeal to users of DNA
sequencing technologies who seek to augment their research and discovery of biomarkers and further deepen their understanding of biology.
|
| • |
Business model that leverages growing install base of instruments. We have initiated our early access limited release phase to
first enable key thought leaders with early access to our platform in 2021 and we seek to broadly commercialize our platform, for RUO, in the second half of 2022. After our full commercial launch, we will aim to grow our install base,
optimize workflows, and expand our applications, which we expect will then generate revenues from our consumables. Our goal is that the integration of our instruments into our users’ projects will provide ongoing sales of consumables,
resulting in a growing recurring revenue stream.
|
| • |
Robust patent protection. We have a strong intellectual property strategy in which we have 140 issued patents and 593 pending
applications as of December 31, 2021. Many from our management team worked directly with our Founder, Dr. Jonathan Rothberg, as he revolutionized the creation of next generation DNA sequencing while founding Ion Torrent, which was acquired
by Life Technologies in 2010. Our team has similarly devoted its efforts to revolutionizing unbiased proteomic analysis using a similar scientific and technical validation approach since our founding in 2013.
|
| • |
Visionary founder backed by strong executive leadership team that has developed and commercialized multiple sequencing technologies
and experienced financial partners with deep experience in healthcare. Our Founder, Interim Chief Executive Officer and Executive Chairman, Dr. Jonathan Rothberg, has dedicated his career to developing breakthrough technologies
to revolutionize healthcare. He has founded more than 10 healthcare technology companies and has received numerous awards, including the Presidential Medal of Technology & Innovation in 2016. Dr. Rothberg previously founded 454 Life
Sciences, a high throughput DNA sequencing platform which was later sold to Roche, as well as founded Ion Torrent, a next generation sequencing platform which was later sold to Life Technologies. He is supported by a world-class management
team, including our executive officers and other senior management, with decades of cumulative experience in the healthcare and life sciences end-markets. Many members of the team worked directly with Dr. Rothberg to successfully
commercialize previous DNA sequencing technologies. We believe this leadership team positions us as a potentially disruptive force in creating a new market of next generation protein sequencing.
|
| • |
Systematic and phased approach to broad commercialization and adoption, directed at potential customers we extensively know. We
intend to follow a systematic and phased approach to successfully launch and commercialize our platform, for RUO, in the second half of 2022. This strategy included partnering with key thought leaders to obtain initial evidence and
feedback in 2021 under an early access program. Members of our team have previously utilized this approach to successfully launch other disruptive sequencing technologies, including the roll out of Ion Torrent’s next generation DNA
sequencing technology. We believe this approach will allow us to introduce our platform in a structured manner to demonstrate its use and practicality, while working directly with our key potential customers and industry thought leaders to
help ensure a positive experience. Our core leadership team has decades of cumulative experience working directly in the life sciences industry with many of the companies and research centers that have the potential to become key customers
and that we will seek to build into our prospective customer pipeline.
|
| • |
Rapidly build our commercial infrastructure to help ensure successful initial commercial launch in the U.S. We expect to
rapidly build out our commercial and operational infrastructure to sell and support our platform as we launch and commercialize our technology. We also have manufacturing partnerships that we believe will allow us to rapidly expand our
capacity, with the ability to create new manufacturing lines to meet potential customer demand. In November 2021, we acquired Majelac, a semiconductor packaging company based in Garnet Valley, Pennsylvania. The acquisition brings our
semiconductor chip assembly and packaging capabilities in-house in order to secure our supply chain and support scaling commercialization efforts.
|
| • |
Invest in market development activities to increase awareness of the importance of the proteome and the strengths of our platform. We
believe our platform has the capability to enable users to generate significant amounts of proteomic information at speed, scale, and simplicity through a solution that is not available today. We believe the utility of our platform will
span basic and discovery applications and translational research in which there is a strong market need for proteomic analysis for novel discoveries and better insights into the complexity of disease. We plan to invest in market
development activities and partnerships to increase awareness of the importance and utility of proteomics to expand and accelerate demand for our products.
|
| • |
Continued technical innovation to drive product enhancements, new products, and additional applications. Our leadership team
has deep expertise in scientific and technological development and commercialization. After we commercialize our initial products, we aim to continually innovate and develop new products, product enhancements, applications, workflows, and
other tools to enable our customers to generate unbiased proteomic information at scale on a benchtop platform.
|
| • |
Accessibility and Enablement: Enable broad adoption of protein sequencing. Our mission is to democratize single molecule
proteomic analysis by providing a full workflow of solutions at an affordable cost. We believe that our platform will directly address many of the key bottlenecks that exist within legacy proteomic technologies, namely low sensitivity,
lack of dynamic range, complex workflow, complex analysis, and high cost. We believe our platform offers the potential for a more practical, affordable, and intuitive end-to-end workflow solution relative to many legacy proteomic
technologies. We have specifically developed our platform to be adopted and integrated into any existing lab. We believe that our platform will have wide utility across the study of proteins, including basic and discovery research and,
subject to regulatory authorization, clinical diagnostics, and potentially industrial applications like bioproduction. Our ability to develop our platform such that it will be offered at a significant discount to many legacy instruments
and other proteomic technologies, may allow proteomic analysis to reach new markets and new users, potentially enabling and accelerating innovative discoveries.
|
| • |
Continue to strengthen our intellectual property portfolio for existing and new technologies. We
have a broad and deep patent protection strategy, which includes 140 issued patents and over 593 pending applications as of December 31, 2021. Protection of our intellectual property is a strategic priority for the business. We have
taken, and will continue to take, steps to protect our current and future intellectual property and proprietary technology. We believe our broad patent portfolio and continued rigorous patent protection strategy will help to allow us to
focus on our key priorities of commercializing our platform, continuing to innovate with new technologies, and preventing fast-followers.
|
| • |
Foster extraordinary talent inspired and unified by our mission. With decades of cumulative experience in the healthcare and
life sciences markets among our executive officers and other senior management, our world-class management team is unified by our mission to democratize single molecule proteomic analysis by making protein sequencing accessible globally.
We seek to execute at scale the vision of our Founder, Interim Chief Executive Officer and Executive Chairman, Dr. Jonathan Rothberg. He has dedicated his career to enabling breakthrough technologies to revolutionize healthcare, including
a novel genome sequencing method brought to market through his company 454 Life Sciences and has founded more than 10 companies. Dr. Rothberg is supported by a leadership team with many years of sequencing, technology, and healthcare
experience at other leading companies, including Affymetrix, Becton Dickinson, Illumina, Ion Torrent, Life Technologies, Pacific Biosciences, and Thermo Fisher Scientific, among others. We plan to continue to add talented and experienced
members to our team and maintain our commitment to our mission of democratizing proteomic analysis by making protein sequencing accessible globally.
|
| • |
resolution and sensitivity;
|
| • |
cost of instruments and consumables;
|
| • |
efficiency and speed of workflows;
|
| • |
the scale required to address the complexity and dynamic range of the proteome;
|
| • |
throughput to meet lab testing volume;
|
| • |
reputation among customers and key thought leaders;
|
| • |
innovation in product offerings;
|
| • |
accuracy and reproducibility of results;
|
| • |
strength of intellectual property portfolio;
|
| • |
operational and manufacturing footprint;
|
| • |
customer support infrastructure; and
|
| • |
a leadership and commercial team with extensive execution and scientific background.
|
| • |
Development of comprehensive product description and indications for use.
|
| • |
Completion of extensive nonclinical tests and/or animal studies, performed in accordance with the FDA’s Good Laboratory Practice (“GLP”) regulations, as well as any
performance standards or other testing requirements established by the FDA through regulations or device-specific guidance.
|
| • |
Comprehensive review of one or more predicate devices and development of data supporting the new product’s substantial equivalence to such predicate devices.
|
| • |
the product may not be safe or effective for its intended use(s) to the FDA’s satisfaction;
|
| • |
the data from the applicant’s nonclinical studies and clinical trials may be insufficient to support approval;
|
| • |
the manufacturing process or facilities that the applicant uses may not meet applicable requirements; and
|
| • |
changes in FDA approval policies or adoption of new regulations may require additional data to demonstrate the safety or effectiveness of the device.
|
| • |
the FDA, the IRB(s), or other regulatory authorities do not approve a clinical trial protocol or a clinical trial, or place a clinical trial on hold;
|
| • |
participants do not enroll in clinical trials at the expected rate;
|
| • |
participants do not comply with trial protocols;
|
| • |
participant follow-up is not at the expected rate;
|
| • |
participants experience adverse side effects;
|
| • |
participants die during a clinical trial, even though their death may not be related to the investigational products;
|
| • |
third-party clinical investigators decline to participate in a trial or do not perform a trial on the sponsor’s anticipated schedule or consistent with the clinical
trial protocol, GCPs or other FDA requirements;
|
| • |
the sponsor or third-party organizations do not perform data collection, monitoring and analysis in a timely or accurate manner or consistent with the clinical trial
protocol or investigational or statistical plans;
|
| • |
third-party clinical investigators have significant financial interests related to the sponsor or the study that the FDA deems to make the study results unreliable, or
the sponsor or investigators fail to disclose such interests;
|
| • |
unfavorable regulatory inspections of the sponsor’s clinical trial sites or manufacturing facilities, which may, among other things, require the sponsor to undertake
corrective action or suspend or terminate the sponsor’s clinical trials;
|
| • |
changes in governmental regulations or administrative actions applicable to the sponsor’s trial protocols;
|
| • |
the interim or final results of the clinical trial are inconclusive or unfavorable as to safety or effectiveness; and
|
| • |
the FDA concludes that the results from the sponsor’s trial and/or trial design are inadequate to demonstrate safety and effectiveness of the product.
|
| • |
establishment registration and device listing;
|
| • |
the QSR, which requires manufacturers, including third-party manufacturers, to follow design, testing, control, storage, supplier/contractor selection, complaint
handling, documentation and other quality assurance procedures;
|
| • |
labeling regulations, which govern the mandatory elements of the device labels and packaging (including Unique Device Identifier markings for certain categories of
products);
|
| • |
FDA’s prohibitions against the promotion of products for uncleared, unapproved or “off-label” uses and other requirements related to promotional activities;
|
| • |
the MDR regulations, which require that manufacturers report to the FDA if a device may have caused or contributed to a death or serious injury or malfunctioned in a
way that would likely cause or contribute to a death or serious injury if it were to recur;
|
| • |
voluntary and mandatory device recalls to address problems when a device is defective and/or could be a risk to health;
|
| • |
correction and removal reporting regulations, which require that manufacturers report to the FDA field corrections and product recalls or removals if undertaken to
reduce a risk to health posed by the device or to remedy a violation of the FDCA that may present a risk to health; and
|
| • |
post-market surveillance regulations, which apply to certain Class II or III devices when necessary to protect the public health or to provide additional safety and
effectiveness data for the device.
|
| • |
Warning Letters or Untitled Letters that require corrective action;
|
| • |
fines and civil penalties;
|
| • |
unanticipated expenditures;
|
| • |
delays in approving/clearing or refusal to approve/clear any of our future products;
|
| • |
FDA refusal to issue certificates to foreign governments needed to export our products for sale in other countries;
|
| • |
suspension or withdrawal of FDA approval or clearance (as may be applicable);
|
| • |
product recall or seizure;
|
| • |
partial suspension or total shutdown of production;
|
| • |
operating restrictions;
|
| • |
injunctions or consent decrees; and
|
| • |
civil or criminal prosecution.
|
|
•
|
the timing and amount of expenditures that we may incur to develop, commercialize or acquire additional products and technologies or for other
purposes, such as the expansion of our facilities;
|
|
•
|
changes in governmental funding of life sciences research and development or changes that impact budgets or budget cycles;
|
|
•
|
seasonal spending patterns of our customers;
|
|
•
|
the timing of when we recognize any revenues;
|
|
•
|
future accounting pronouncements or changes in our accounting policies;
|
|
•
|
the outcome of any future litigation or governmental investigations involving us, our industry or both;
|
|
•
|
higher than anticipated service, replacement and warranty costs;
|
|
•
|
the impact of the COVID-19 pandemic on the economy, investment in life sciences and research industries, our business operations, and resources
and operations of our suppliers, distributors and potential customers; and
|
|
•
|
general industry, economic and market conditions and other factors, including factors unrelated to our operating performance or the operating
performance of our competitors.
|
|
•
|
the inability to establish the capabilities and value proposition of our products with key opinion leaders in a timely fashion;
|
|
•
|
the potential need or desire to modify aspects of our products prior to entering into the second or third phases of our commercial launch plan;
|
|
•
|
changing industry or market conditions, customer requirements or competitor offerings over the span of our commercial launch plan;
|
|
•
|
delays in building out our sales, customer support and marketing organization as needed for each of the phases of our commercial launch plan;
and
|
|
•
|
delays in ramping up manufacturing, either internally or through our suppliers to meet the expected demand in each of the phases of our
commercial launch plan.
|
|
•
|
our ability to market and increase awareness of the capabilities of our products;
|
|
•
|
the ability of our products to demonstrate comparable performance in intended use applications broadly in the hands of customers consistent with
the early access limited release phase of our commercialization plan;
|
|
•
|
our potential customers’ willingness to adopt new products and workflows;
|
|
•
|
our product’s ease of use and whether it reliably provides advantages over other alternative technologies;
|
|
•
|
the rate of adoption of our products by academic institutions, laboratories, biopharmaceutical companies and others;
|
|
•
|
the prices we charge for our products;
|
|
•
|
our ability to develop new products and workflows and solutions for customers;
|
|
•
|
if competitors develop and commercialize products that perform similar functions as our products; and
|
|
•
|
the impact of our investments in product innovation and commercial growth.
|
|
•
|
our ability to attract, retain and manage the sales, marketing and customer service and support force necessary to commercialize and gain market
acceptance of our products;
|
|
•
|
the time and cost of establishing a specialized sales, marketing and customer service and support force; and
|
|
•
|
our sales, marketing and customer service and support force may be unable to initiate and execute successful commercialization activities.
|
|
•
|
decreases in government funding of research and development;
|
|
•
|
changes to programs that provide funding to research laboratories and institutions, including changes in the amount of funds allocated to
different areas of research or changes that have the effect of increasing the length of the funding process;
|
|
•
|
macroeconomic conditions and the political climate;
|
|
•
|
potential changes in the regulatory environment;
|
|
•
|
differences in budgetary cycles, especially government- or grant-funded customers, whose cycles often coincide with government fiscal year ends;
|
|
•
|
competitor product offerings or pricing;
|
|
•
|
market-driven pressures to consolidate operations and reduce costs; and
|
|
•
|
market acceptance of relatively new technologies.
|
|
•
|
required compliance with existing and changing foreign regulatory requirements and laws that are or may be applicable to our business in the
future, such as the European Union’s General Data Protection Regulation (“GDPR”) and other data privacy requirements, labor and employment regulations, anti-competition regulations, the U.K. Bribery Act of 2010 and other anti-corruption
laws, regulations relating to the use of certain hazardous substances or chemicals in commercial products, and require the collection, reuse, and recycling of waste from products we manufacture;
|
|
•
|
required compliance with U.S. laws such as the Foreign Corrupt Practices Act, and other U.S. federal laws and regulations established by the
Office of Foreign Assets Control of the U.S. Department of the Treasury;
|
|
•
|
export requirements and import or trade restrictions;
|
|
•
|
laws and business practices favoring local companies;
|
|
•
|
foreign currency exchange, longer payment cycles and difficulties in enforcing agreements and collecting receivables through certain foreign
legal systems;
|
|
•
|
changes in social, economic, and political conditions or in laws, regulations and policies governing foreign trade, manufacturing, research and
development, and investment both domestically as well as in the other countries and jurisdictions in which we operate and into which it may sell our products including as a result of the separation of the United Kingdom from the European
Union (“Brexit”);
|
|
•
|
potentially adverse tax consequences, tariffs, customs charges, bureaucratic requirements, and other trade barriers;
|
|
•
|
difficulties and costs of staffing and managing foreign operations; and
|
|
•
|
difficulties protecting, maintaining, enforcing or procuring intellectual property rights.
|
|
•
|
a failure to achieve market acceptance for our products or expansion of our product sales;
|
|
•
|
loss of customer orders and delay in order fulfillment;
|
|
•
|
damage to our brand reputation;
|
|
•
|
loss of revenue;
|
|
•
|
increased warranty and customer service and support costs due to product repair or replacement;
|
|
•
|
product recalls or replacements;
|
|
•
|
inability to attract new customers;
|
|
•
|
diversion of resources from our manufacturing and research and development team into our service team; and
|
|
•
|
legal claims against us, including product liability claims, which could be costly and time consuming to defend and result in substantial
damages.
|
|
•
|
greater name and brand recognition;
|
|
•
|
greater financial and human resources;
|
|
•
|
broader product lines;
|
|
•
|
larger sales forces and more established distributor networks;
|
|
•
|
substantial intellectual property portfolios;
|
|
•
|
larger and more established customer bases and relationships; and
|
|
•
|
better established, larger scale and lower cost manufacturing capabilities.
|
|
•
|
the federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering,
receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce either the referral of an individual or furnishing or arranging for a good or service, for which payment may be made, in whole or in part, under
federal healthcare programs, such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation;
|
|
•
|
the federal civil and criminal false claims laws, including the federal civil False Claims Act, which prohibit, among other things, individuals
or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid or other federal healthcare programs that are false or fraudulent. Private individuals can bring False Claims Act “qui tam”
actions, on behalf of the government and such individuals, commonly known as “whistleblowers,” may share in amounts paid by the entity to the government in fines or settlement.
|
|
•
|
the federal Civil Monetary Penalties Law, which prohibits, among other things, offering or transferring remuneration to a federal healthcare
beneficiary that a person knows or should know is likely to influence the beneficiary’s decision to order or receive items or services reimbursable by the government from a particular provider or supplier;
|
|
•
|
HIPAA, which created additional federal criminal statutes that prohibit, among other things, executing a scheme to defraud any healthcare
benefit program and making false statements relating to healthcare matters;
|
|
•
|
the federal Physician Sunshine Act, which requires certain manufacturers of drugs, devices, biologics and medical supplies for
which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, to report annually to CMS, information related to payments and other transfers of value to physicians, which is defined broadly to include
doctors, dentists, optometrists, podiatrists and chiropractors teaching hospitals and certain advanced non-physician healthcare practitioners; and
|
|
•
|
analogous state and foreign law equivalents of each of the above federal laws, such as anti-kickback and false claims laws, which may apply to
items or services reimbursed by any third-party payor, including commercial insurers or patients.
|
| • |
the scope of rights granted under the license agreement and other interpretation-related issues;
|
| • |
our financial or other obligations under the license agreement;
|
| • |
whether, and the extent to which, our products, technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement;
|
| • |
our diligence obligations under the license agreement and what activities satisfy those diligence obligations;
|
| • |
the inventorship and ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensor(s); and
|
| • |
the priority of invention of patented technology.
|
| • |
others may be able to make products that are similar to products and technologies we may develop or utilize similar technology that are not covered by the claims of the patents that we own or license now
or in the future;
|
| • |
we, or our licensor(s), might not have been the first to make the inventions covered by the issued patent or pending patent application that we license or may own in the future;
|
| • |
we, or our licensor(s), might not have been the first to file patent applications covering certain of our or their inventions;
|
| • |
others may independently develop similar or alternative technologies or duplicate any of our technologies without infringing, misappropriating or otherwise violating our owned or licensed intellectual
property rights;
|
| • |
it is possible that our pending licensed patent applications or those that we may own in the future will not lead to issued patents;
|
| • |
issued patents that we hold rights to may be held invalid or unenforceable, including as a result of legal challenges by our competitors;
|
| • |
our competitors might conduct research and development activities in countries where we do not have patent rights and then use the information learned from such activities to develop competitive products
for sale in our major commercial markets;
|
| • |
we may not develop additional proprietary technologies that are patentable;
|
| • |
the patents of others may harm our business; and
|
| • |
we may choose not to file a patent for certain trade secrets or know-how, and a third party may subsequently file a patent covering such intellectual property.
|
| • |
the ability of our board of directors to issue one or more series of preferred stock;
|
| • |
stockholder action by written consent only until the first time when Dr. Rothberg ceases to beneficially own a majority of the voting power of our capital stock;
|
| • |
certain limitations on convening special stockholder meetings;
|
| • |
advance notice for nominations of directors by stockholders and for stockholders to include matters to be considered at our annual meetings;
|
| • |
amendment of certain provisions of the organizational documents only by the affirmative vote of (i) a majority of the voting power of our capital stock so long as Dr. Rothberg beneficially owns shares
representing a majority of the voting power of our capital stock and (ii) at least two-thirds of the voting power of the capital stock from and after the time that Dr. Rothberg ceases to beneficially own shares representing a majority
of our voting power; and
|
| • |
a dual-class common stock structure with 20 votes per share of our Class B common stock, the result of which is that Dr. Rothberg has the ability to control the outcome of matters requiring stockholder
approval, even though Dr. Rothberg owns less than a majority of the outstanding shares of our capital stock.
|
| ITEM 2. |
PROPERTIES
|
| ITEM 3. |
LEGAL PROCEEDINGS
|
| ITEM 4. |
MINE SAFETY DISCLOSURES
|
| ITEM 5. |
MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
|
| ITEM 6. |
[RESERVED]
|
| ITEM 7. |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
|
|
Years ended December 31,
|
||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
% Change
|
|||||||||
|
Operating expenses:
|
||||||||||||
|
Research and development
|
$
|
46,575
|
$
|
27,555
|
69.0
|
%
|
||||||
|
General and administrative
|
46,377
|
7,984
|
480.9
|
%
|
||||||||
|
Sales and marketing
|
3,956
|
1,152
|
243.4
|
%
|
||||||||
|
Total operating expenses
|
96,908
|
36,691
|
164.1
|
%
|
||||||||
|
Loss from operations
|
(96,908
|
)
|
(36,691
|
)
|
164.1
|
%
|
||||||
|
Interest expense
|
(5
|
)
|
(9
|
)
|
(44.4
|
%)
|
||||||
|
Dividend income
|
2,549
|
97
|
2527.8
|
%
|
||||||||
|
Change in fair value of warrant liabilities
|
4,379
|
-
|
nm
|
|||||||||
|
Other (expense) income, net
|
(5,004
|
)
|
(10
|
)
|
49940.0
|
%
|
||||||
|
Loss before provision for income taxes
|
(94,989
|
)
|
(36,613
|
)
|
159.4
|
%
|
||||||
|
Provision for income taxes
|
-
|
-
|
nm
|
|||||||||
|
Net loss and comprehensive loss
|
$
|
(94,989
|
)
|
$
|
(36,613
|
)
|
159.4
|
%
|
||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
Amount
|
%
|
||||||||||||
|
Research and development
|
$
|
46,575
|
$
|
27,555
|
$
|
19,020
|
69.0
|
%
|
||||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
Amount
|
%
|
||||||||||||
|
General and administrative
|
$
|
46,377
|
$
|
7,984
|
$
|
38,393
|
480.9
|
%
|
||||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
Amount
|
%
|
||||||||||||
|
Sales and marketing
|
$
|
3,956
|
$
|
1,152
|
$
|
2,804
|
243.4
|
%
|
||||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
Amount
|
%
|
||||||||||||
|
Interest expense
|
$
|
(5
|
)
|
$
|
(9
|
)
|
$
|
4
|
(44.4
|
%)
|
||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
Amount
|
%
|
||||||||||||
|
Dividend income
|
$
|
2,549
|
$
|
97
|
$
|
2,452
|
2527.8
|
%
|
||||||||
| Years Ended December 31, | Change | |||||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
Amount
|
%
|
||||||||||||
|
Change in fair value of warrant liabilities
|
$
|
4,379
|
$
|
-
|
$
|
4,379
|
|
nm | ||||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2021
|
2020
|
Amount
|
%
|
||||||||||||
|
Other (expense), net
|
$
|
(5,004
|
)
|
$
|
(10
|
)
|
$
|
(4,994
|
)
|
49940.0
|
%
|
|||||
|
Years ended December 31,
|
||||||||||||
|
(in thousands, except for % changes)
|
2020
|
2019
|
% Change
|
|||||||||
|
Operating expenses:
|
||||||||||||
|
Research and development
|
$
|
27,555
|
$
|
28,102
|
(1.9
|
%)
|
||||||
|
General and administrative
|
7,984
|
7,884
|
1.3
|
%
|
||||||||
|
Sales and marketing
|
1,152
|
634
|
81.7
|
%
|
||||||||
|
Total operating expenses
|
36,691
|
36,620
|
0.2
|
%
|
||||||||
|
Loss from operations
|
(36,691
|
)
|
(36,620
|
)
|
0.2
|
%
|
||||||
|
Interest expense
|
(9
|
)
|
-
|
nm
|
||||||||
|
Dividend income
|
97
|
823
|
(88.2
|
%)
|
||||||||
|
Change in fair value of warrant liabilities
|
-
|
-
|
nm
|
|||||||||
|
Other (expense) income, net
|
(10
|
)
|
5
|
(300.0
|
%)
|
|||||||
|
Loss before provision for income taxes
|
(36,613
|
)
|
(35,792
|
)
|
2.3
|
%
|
||||||
|
Provision for income taxes
|
-
|
-
|
nm
|
|||||||||
|
Net loss and comprehensive loss
|
$
|
(36,613
|
)
|
$
|
(35,792
|
)
|
2.3
|
%
|
||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2020
|
2019
|
Amount
|
%
|
||||||||||||
|
Research and development
|
$
|
27,555
|
$
|
28,102
|
$
|
(547
|
)
|
(1.9
|
%)
|
|||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2020
|
2019
|
Amount
|
%
|
||||||||||||
|
General and administrative
|
$
|
7,984
|
$
|
7,884
|
$
|
100
|
1.3
|
%
|
||||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2020
|
2019
|
Amount
|
%
|
||||||||||||
|
Sales and marketing
|
$
|
1,152
|
$
|
634
|
$
|
518
|
81.7
|
%
|
||||||||
|
Years Ended December 31,
|
Change
|
||||||||||||||||
|
(in thousands, except for % changes)
|
2020
|
2019
|
Amount
|
% |
|
||||||||||||
|
Interest expense
|
$
|
(9
|
)
|
$
|
-
|
$
|
(9
|
)
|
|
nm |
|
||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2020
|
2019
|
Amount
|
%
|
||||||||||||
|
Dividend income
|
$
|
97
|
$
|
823
|
$
|
(726
|
)
|
(88.2
|
%)
|
|||||||
|
Years Ended December 31,
|
Change
|
|||||||||||||||
|
(in thousands, except for % changes)
|
2020
|
2019
|
Amount
|
%
|
||||||||||||
|
Other (expense) income, net
|
$
|
(10
|
)
|
$
|
5
|
$
|
(15
|
)
|
(300.0
|
%)
|
||||||
|
Years Ended December 31,
|
||||||||||||
|
(in thousands)
|
2021
|
2020
|
2019
|
|||||||||
|
Net loss
|
$
|
(94,989
|
)
|
(36,613
|
)
|
(35,792
|
)
|
|||||
|
Interest expense
|
5
|
9
|
-
|
|||||||||
|
Dividend income
|
(2,549
|
)
|
(97
|
)
|
(823
|
)
|
||||||
|
Change in fair value of warrant liabilities
|
(4,379
|
)
|
-
|
-
|
||||||||
|
Other expense (income), net
|
5,004
|
10
|
(5
|
)
|
||||||||
|
Stock-based compensation expense
|
24,918
|
1,924
|
2,715
|
|||||||||
|
Depreciation
|
1,041
|
894
|
780
|
|||||||||
|
Transaction related costs - business combination
|
6,920
|
-
|
-
|
|||||||||
|
Adjusted EBITDA
|
$
|
(64,029
|
)
|
$
|
(33,873
|
)
|
$
|
(33,125
|
)
|
|||
|
Years Ended December 31,
|
||||||||||||
|
(in thousands)
|
2021
|
2020
|
2019
|
|||||||||
|
Net cash (used in) provided by:
|
||||||||||||
|
Net cash used in operating activities
|
$
|
(66,813
|
)
|
$
|
(32,573
|
)
|
$
|
(30,708
|
)
|
|||
|
Net cash used in investing activities
|
(450,937
|
)
|
(461
|
)
|
(1,241
|
)
|
||||||
|
Net cash provided by financing activities
|
516,625
|
37,014
|
18,217
|
|||||||||
|
Net (decrease) increase in cash and cash equivalents
|
$
|
(1,125
|
)
|
$
|
3,980
|
$
|
(13,732
|
)
|
||||
|
•
|
Risk-free interest rate: The risk-free interest rate for periods within the expected term of the awards is based on the U.S. Treasury yield curve in effect at the time of the grant;
|
|
•
|
Expected dividend yield: We have never declared or paid any cash dividends and do not expect to pay any cash dividends in the foreseeable future;
|
|
•
|
Expected term: For awards, we calculate the expected term using the “simplified” method, which is the simple average of the vesting period and the contractual term; and
|
|
•
|
Expected volatility: We determined expected annual equity volatility to be 70% based on the historical volatility of guideline public companies for the years ended December 31, 2019 and 2020 and from
January to June 10, 2021. After June 10, 2021, the volatility is calculated by a third-party professional services firm and reviewed by the Company.
|
| ITEM 8. |
FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
|
| ITEM 9. |
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
|
| ITEM 9B. |
OTHER INFORMATION
|
| ITEM 9C. |
DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
|
| ITEM 10. |
DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
|
|
Name
|
Age
|
Position
|
||
|
Jonathan M. Rothberg, Ph.D.
|
58
|
Interim Chief Executive Officer and Executive Chairman of the Board
|
||
|
Claudia Drayton
|
54
|
Chief Financial Officer
|
||
|
Michael P. McKenna, Ph.D.
|
59
|
President and Chief Operating Officer
|
||
|
Matthew Dyer, Ph.D.
|
40
|
Chief Business Officer
|
||
|
Christian LaPointe, Ph.D.
|
51
|
General Counsel and Corporate Secretary
|
||
|
Marijn Dekkers, Ph.D.
|
64
|
Director
|
||
|
Ruth Fattori
|
69
|
Director
|
||
|
Brigid A. Makes
|
66
|
Director
|
||
|
Michael Mina, M.D., Ph.D.
|
38
|
Director
|
||
|
Kevin Rakin
|
61
|
Director
|
||
|
James Tananbaum, M.D.
|
58
|
Director
|
| ITEM 11. |
EXECUTIVE COMPENSATION
|
|
Name
|
Principal Position
|
|
|
John Stark
|
Former Chief Executive Officer
|
|
|
Claudia Drayton
|
Chief Financial Officer
|
|
|
Michael P. McKenna, Ph.D.
|
President and Chief Operating Officer
|
|
|
Matthew Dyer, Ph.D.
|
Chief Business Officer
|
|
|
Christian LaPointe, Ph.D.
|
General Counsel and Corporate Secretary
|
|
•
|
Attract, motivate and retain executive officers of outstanding ability and potential;
|
|
•
|
Reinforce the execution of our business strategy and the achievement of our business objectives; and
|
|
•
|
Align the interests of our executive officers with the interests of our stockholders, with the ultimate objective of increasing stockholder value.
|
|
What We Do
|
What We Don’t Do
|
|
|
✓ Emphasize “at-risk” compensation and long-term equity incentives
|
× No guaranteed “single-trigger” change in control cash payments
|
|
|
✓ Tie performance bonus opportunities to defined corporate objectives
|
× No tax reimbursements or tax gross-ups on severance or change in control payments
|
|
|
✓ Structure severance payments as “double-trigger” requiring both a change in control and an involuntary termination for payout
|
× No special executive welfare or health benefits, or retirement plans not available to our employees generally
|
|
|
✓ Assess risks of our compensation program annually
|
× No guaranteed salary increases or bonuses
× No extensive perquisites
|
|
|
✓ Maintain a compensation committee comprised entirely of independent directors
|
||
|
✓ Retain an independent compensation advisor
|
|
•
|
Assisting in developing a peer group of publicly traded companies to be used to help assess executive compensation;
|
|
•
|
Assisting in developing a competitive compensation strategy and consistent executive compensation assessment practices relevant to a public company, including review and
recommendation of the target values of the annual performance-based cash incentive program as well as the equity strategy for the Company covering dilution, grant levels and type of equity; and
|
|
•
|
Meeting regularly with the compensation committee to review all elements of executive compensation including the competitiveness of the executive compensation program against
approved peer companies.
|
|
• Accelerate Diagnostics, Inc.
|
• Inovio Pharmaceuticals, Inc.
|
|
• Acutus Medical, Inc.
|
• Maravai LifeSciences Holdings, Inc.
|
|
• Berkeley Lights, Inc.
|
• NanoString Technologies, Inc.
|
|
• Bionano Genomics, Inc.
|
• Personalis, Inc.
|
|
• Butterfly Network, Inc.
|
• PureTech Health plc
|
|
• Castle Biosciences, Inc.
|
• Quanterix Corporation
|
|
• Cerevel Therapeutics Holdings, Inc.
|
• Seer, Inc.
|
|
• Co-Diagnostics, Inc.
|
• T2 Biosystems, Inc.
|
|
• Codexis, Inc.
|
• SeerVeracyte, Inc.
|
|
• Fluidigm Corporation
|
|
•
|
Base salary;
|
|
•
|
Annual performance-based cash incentive compensation; and
|
|
•
|
Equity incentive awards.
|
|
Name
|
2020 Base Salary
|
2021 Base Salary
|
% Increase
|
||||||||||
|
John Stark (2)
|
$
|
350,000
|
$
|
500,000
|
(1)
|
42.9
|
%
|
||||||
|
Claudia Drayton (3)
|
-
|
$
|
385,000
|
-
|
|||||||||
|
Michael P. McKenna, Ph.D.
|
$
|
262,500
|
$
|
440,000
|
(4)
|
67.6
|
%
|
||||||
|
Matthew Dyer, Ph.D.
|
$
|
262,500
|
$
|
400,000
|
(4)
|
52.4
|
%
|
||||||
|
Christian LaPointe, Ph.D.
|
$
|
240,000
|
$
|
375,000
|
(1)
|
56.3
|
%
|
||||||
|
(1)
|
The increases in base salaries for Mr. Stark and Dr. LaPointe were effective as of July 1, 2021.
|
|
(2)
|
Mr. Stark’s employment as our CEO terminated effective as of February 8, 2022.
|
|
(3)
|
Ms. Drayton joined Legacy Quantum-Si as its Chief Financial Officer in April 2021. Her initial base salary was $330,000, which was increased to $385,000 effective as of July 1, 2021.
|
|
(4)
|
The base salaries for Dr. McKenna and Dr. Dyer were increased to $275,625 effective as of January 1, 2021, and then were increased to the amounts shown in the table above effective as of July 1, 2021.
|
|
•
|
Achieve early access performance specifications
|
|
•
|
Establish/secure supply chain inventory to achieve 2022 revenue targets (instruments, chips and assays)
|
|
•
|
Enable (ship and train) more than ten early access customers
|
|
•
|
Execute on approved hiring plan and The Sarbanes–Oxley Act of 2002 compliance readiness
|
|
•
|
Establish development programs for single molecule methods/assays (nucleic acids/proteomics/digital analyte)
|
|
•
|
Establishment of a robust governance and communication protocol (applicable to our former CEO only)
|
|
Name
|
Incentive Target Amount
(as a % of Base Salary)
|
Annual Target Bonus (1)
|
Actual Award
|
|||||||||
|
John Stark
|
100
|
%
|
$
|
425,000
|
$
|
352,750
|
(2)
|
|||||
|
Claudia Drayton
|
50
|
%
|
$
|
129,062
|
$
|
115,000
|
||||||
|
Michael P. McKenna, Ph.D.
|
50
|
%
|
$
|
178,906
|
$
|
150,000
|
||||||
|
Matthew Dyer, Ph.D.
|
50
|
%
|
$
|
168,906
|
$
|
145,000
|
||||||
|
Christian LaPointe, Ph.D.
|
50
|
%
|
$
|
153,750
|
$
|
132,500
|
||||||
|
(1)
|
The annual target bonus takes into consideration salary adjustments made during 2021
|
|
(2)
|
Mr. Stark’s employment with the Company terminated effective as of February 8, 2022 and he received an amount equal to his 2021 bonus under the Stark Separation Agreement.
|
|
Name and Position
|
Year
|
Salary
($) |
Bonus
($) |
Stock Awards
($) (2) |
Option Awards
($) (3) |
Non-Equity Incentive Plan Compensation
($) |
All Other Compensation
($) |
Total
($) |
|||||||||||||||||||||
|
John Stark
|
2021
|
$
|
425,000
|
$
|
8,750
|
(1)
|
$
|
15,711,346
|
(10) |
$
|
-
|
$
|
352,750
|
$
|
43,096
|
(5)
|
$
|
16,540,942
|
|||||||||||
|
Former Chief Executive Officer and Director (4)
|
2020
|
$
|
58,333
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
7,564
|
$
|
65,897
|
||||||||||||||
|
2019
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
|||||||||||||||
|
|
|||||||||||||||||||||||||||||
|
Claudia Drayton
|
2021
|
$
|
247,500
|
$
|
-
|
$
|
905,322
|
$
|
1,119,239
|
$
|
115,000
|
$
|
-
|
$
|
2,387,061
|
||||||||||||||
|
Chief Financial Officer (6)
|
2020
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
||||||||||||||
|
2019
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
|||||||||||||||
|
|
|||||||||||||||||||||||||||||
|
Michael P. McKenna, PhD.
|
2021
|
$
|
357,813
|
$
|
250,000
|
(1)
|
$
|
680,268
|
$
|
492,946
|
$
|
150,000
|
$
|
100,000
|
(7)
|
$
|
2,031,027
|
||||||||||||
|
President and Chief Operating Officer
|
2020
|
$
|
262,500
|
$
|
75,000
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
337,500
|
||||||||||||||
|
2019
|
$
|
250,000
|
$
|
50,000
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
300,000
|
|||||||||||||||
|
|
|||||||||||||||||||||||||||||
|
Matthew Dyer, PhD.
|
2021
|
$
|
337,813
|
$
|
250,000
|
(1)
|
$
|
680,268
|
$
|
-
|
$
|
145,000
|
$
|
25,903
|
(8)
|
$
|
1,438,984
|
||||||||||||
|
Chief Business Officer
|
2020
|
$
|
262,500
|
$
|
75,000
|
$
|
-
|
$
|
257,500
|
$
|
-
|
$
|
58,868
|
$
|
653,868
|
||||||||||||||
|
2019
|
$
|
250,000
|
$
|
20,000
|
$
|
-
|
$
|
742,788
|
$
|
-
|
$
|
44,747
|
$
|
1,057,535
|
|||||||||||||||
|
|
|||||||||||||||||||||||||||||
|
Christian LaPointe, PhD.
|
2021
|
$
|
307,500
|
$
|
50,000
|
(1)
|
$
|
1,333,809
|
$
|
246,592
|
$
|
132,500
|
$
|
-
|
$
|
2,070,401
|
|||||||||||||
|
General Counsel and Corporate Secretary (9)
|
2020
|
$
|
36,000
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
36,000
|
||||||||||||||
|
2019
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
|||||||||||||||
| (1) |
The amount represents discretionary transaction bonuses paid in connection with the consummation of the Business Combination.
|
| (2) |
The amount represents the aggregate grant date fair value for restricted stock unit (“RSU”) awards computed in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718 (“ASC 718”). A
discussion of our methodology for determining grant date fair value may be found in Note 12 “Equity Incentive Plan” in our consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
|
| (3) |
The amount represents the aggregate grant date fair value for option awards computed in accordance with ASC 718. A discussion of our methodology for determining grant date fair value may be found in Note 12 “Equity Incentive
Plan” in our consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
|
| (4) |
Mr. Stark joined Legacy Quantum-Si as its Chief Executive Officer in November 2020 and his employment and service as a member of the Board ended effective as of February 8, 2022.
|
| (5) |
Consists of a temporary housing allowance for housing and travel to our principal executive office in Connecticut.
|
| (6) |
Ms. Drayton joined Legacy Quantum-Si as its Chief Financial Officer in April 2021. Her current annual base salary is $385,000.
|
|
(7)
|
Consists of a loan amount forgiven by us in 2021 prior to the Business Combination which was provided in connection with Dr. McKenna’s commencement of employment. The company forgave the loan
as consideration for Dr. McKenna’s performance throughout his time at Legacy Quantum-Si.
|
| (8) |
Consists of a housing allowance of $12,437 provided to Dr. Dyer in January and February 2021 for housing and travel to our principal executive office in Connecticut and $13,466 provided to Dr. Dyer in February 2021 for
relocation expenses.
|
|
(9)
|
Dr. LaPointe joined Legacy Quantum-Si as its General Counsel and Corporate Secretary in November 2020.
|
|
(10)
|
Includes a performance-based RSU award granted to Mr. Stark in 2021. The maximum grant date fair value of this performance-based RSU award, assuming the performance conditions had been
achieved in full, is the same ($2,373,254) for Mr. Stark. These RSUs were forfeited in accordance with the Stark Separation Agreement (as defined below).
|
|
Name
|
Grant Date
|
Estimated Future Payouts Under Non-Equity Incentive Plan Awards: Target ($) (1)
|
Estimated Future Payouts Under Equity Incentive Plan Awards: Target (#)
|
All Other Stock Awards: Number of Shares of Stock or Units (#)
|
All Other Option Awards: Number of Securities Underlying Options (#)
|
Exercise or Base Price of Option Awards ($/Sh)
|
Grant Date Fair Value of Stock and Option Awards ($) (2)
|
|||||||||||||||||||||
|
John Stark
|
-
|
$
|
425,000
|
-
|
-
|
-
|
$
|
-
|
$
|
-
|
||||||||||||||||||
|
2/17/2021
|
$
|
-
|
-
|
1,703,460
|
(3)
|
-
|
$
|
-
|
$
|
13,338,092
|
||||||||||||||||||
|
2/17/2021
|
$
|
-
|
453,777
|
(4)
|
-
|
-
|
$
|
-
|
$
|
2,373,254
|
(5)
|
|||||||||||||||||
|
Claudia Drayton
|
-
|
$
|
129,062
|
-
|
-
|
-
|
$
|
-
|
$
|
-
|
||||||||||||||||||
|
4/20/2021
|
$
|
-
|
-
|
95,700
|
(6)
|
-
|
$
|
-
|
$
|
905,322
|
||||||||||||||||||
|
4/20/2021
|
$
|
-
|
-
|
-
|
191,399
|
(7)
|
$
|
9.46
|
$
|
1,119,239
|
||||||||||||||||||
|
Michael P. McKenna, Ph.D.
|
-
|
$
|
178,906
|
-
|
-
|
-
|
$
|
-
|
-
|
|||||||||||||||||||
|
3/12/2021
|
$
|
-
|
-
|
79,750
|
(8)
|
-
|
$
|
-
|
$
|
680,268
|
||||||||||||||||||
|
8/31/2021
|
$
|
-
|
-
|
-
|
100,000
|
(9)
|
$
|
9.72
|
$
|
492,946
|
||||||||||||||||||
|
Matthew Dyer, Ph.D.
|
-
|
$
|
168,906
|
-
|
-
|
-
|
$
|
-
|
$
|
-
|
||||||||||||||||||
|
3/12/2021
|
$
|
-
|
-
|
79,750
|
(10)
|
-
|
$
|
-
|
$
|
680,268
|
||||||||||||||||||
|
Christian LaPointe, Ph.D.
|
-
|
$
|
153,750
|
-
|
-
|
-
|
$
|
-
|
-
|
|||||||||||||||||||
|
2/17/2021
|
$
|
-
|
-
|
170,346
|
(11)
|
-
|
$
|
-
|
$
|
1,333,809
|
||||||||||||||||||
|
8/31/2021
|
$
|
-
|
-
|
-
|
50,000
|
(12)
|
$
|
9.72
|
$
|
246,592
|
||||||||||||||||||
| (1) |
Represents the potential 2021 cash incentive bonus payouts assuming target achievement of goals, based upon the NEO’s cash incentive
bonus target and base salary in effect on December 31, 2021. No minimum threshold amount or maximum amount beyond the target amount was established. See the column entitled “Non-Equity Incentive Plan Compensation” in the
Summary Compensation Table for the cash incentive bonuses earned by the NEOs in 2021. See “Compensation Discussion and Analysis — Components of Executive Compensation — Annual Performance-Based Cash Incentive Compensation” for
a description of our 2021 Plan.
|
|
(2)
|
The amount represents the grant date fair value for RSU awards and options computed in accordance with ASC 718. A discussion of our methodology for determining grant date fair value may be
found in Note 12 “Equity Incentive Plan” in our consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
|
| (3) |
Represents the grant of RSUs made to Mr. Stark. The RSUs vested as to 25% on January 7, 2022, with the remainder vesting in 12 equal
quarterly installments thereafter beginning with the quarter ending March 31, 2022, subject to Mr. Stark’s continued service through the applicable vesting date. All of Mr. Stark’s unvested RSUs were forfeited on February 8,
2022 in connection with Mr. Stark’s separation.
|
|
(4)
|
Represents the grant of performance-based RSUs made to Mr. Stark. The performance-based RSUs vest (i) on the closing of a financing in excess of $50 million within three years of Mr. Stark’s
commencement of employment with Legacy Quantum-Si at a share price greater than $16.08, or (ii) if within three years of Mr. Stark’s commencement of employment with Legacy Quantum-Si the publicly-listed closing price of our
shares is $16.08 or more for any 20 trading days within any 30 consecutive trading day period, subject to Mr. Stark’s continued service through the applicable vesting date. These RSUs were forfeited in accordance with the
Stark Separation Agreement.
|
| (5) |
The amount represents the maximum grant date fair value for the performance-based RSUs, computed in accordance with FASB ASC Topic 718. A discussion of the assumptions used in determining grant date fair value may be found in
Note 12 “Equity Incentive Plan” in our consolidated financial statements included elsewhere in this Annual Report on Form 10-K. The maximum grant date fair value of this performance-based RSU award, assuming that the performance
conditions are achieved in full, is the same ($2,373,254).
|
| (6) |
Represents the grant of RSUs made to Ms. Drayton. The RSUs vest as to 25% on June 30, 2022, with the remainder vesting in 12 equal quarterly installments thereafter, subject to Ms. Drayton’s continued service through the
applicable vesting date.
|
| (7) |
Represents the grant of stock options made to Ms. Drayton. The shares underlying this option vest as to 25% on June 30, 2022, with the remainder vesting in 36 equal monthly installments thereafter, subject to Ms. Drayton’s
continued service through the applicable vesting date.
|
| (8) |
Represents the grant of RSUs made to Dr. McKenna. The RSUs vest as to 25% on March 12, 2022, with the remainder vesting in 12 equal quarterly installments thereafter, subject to Dr. McKenna’s continued service through the
applicable vesting date.
|
| (9) |
Represents the grant of stock options made to Dr. McKenna. The shares underlying this option vest as to 2.083% for 48 months in equal installments beginning on August 31, 2021, thereafter, subject to McKenna’s continued
service through the applicable vesting date.
|
| (10) |
Represents the grant of RSUs made to Dr. Dyer. The RSUs vest as to 25% on March 12, 2022, with the remainder vesting in 12 equal quarterly installments thereafter, subject to Dr. Dyer’s continued service through the
applicable vesting date.
|
| (11) |
Represents the grant of RSUs made to Dr. LaPointe. The RSUs vested as to 25% on January 7, 2022, with the remainder vesting in 12 equal quarterly installments thereafter beginning with the quarter ending March 31, 2022,
subject to Dr. LaPointe’s continued service through the applicable vesting date.
|
| (12) |
Represents the grant of stock options made to Dr. LaPointe. The shares underlying this option vest as to 25% on August 31, 2022, with the remainder vesting in 36 equal monthly installments thereafter, subject to Dr.
LaPointe’s continued service through the applicable vesting date.
|
|
|
Option Awards
|
Stock Awards
|
|||||||||||||||||||||||||||||||
|
Name
|
Grant Date
|
Number of Securities Underlying Unexercised Options Exercisable (#)
|
Number of Securities Underlying Unexercised Options Unexercisable (#)
|
Option Exercise Price
|
Options Expiration Date
|
Number of Shares or Units That Have Not Vested
|
Market Value of Shares or Units of Stock That Have Not Vested (1)
|
Equity Incentive Plan Awards: Number of Unearned Shares, Units or Other Rights That Have Not Vested
|
Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Unit Rights That Have Not Vested
|
||||||||||||||||||||||||
|
John
Stark |
2/17/2021
|
-
|
-
|
$
|
-
|
-
|
1,703,460
|
(2)
|
$
|
13,406,230
|
-
|
-
|
|||||||||||||||||||||
|
2/17/2021
|
-
|
-
|
$
|
-
|
-
|
-
|
$
|
-
|
453,777
|
(3)
|
$
|
3,571,225
|
|||||||||||||||||||||
|
|
|||||||||||||||||||||||||||||||||
|
Claudia
Drayton |
4/20/2021
|
-
|
191,399
|
(4)
|
$
|
9.46
|
4/20/2031
|
-
|
$
|
-
|
-
|
-
|
|||||||||||||||||||||
|
4/20/2021
|
-
|
-
|
$
|
-
|
-
|
95,700
|
(5)
|
$
|
753,159
|
-
|
-
|
||||||||||||||||||||||
|
|
|||||||||||||||||||||||||||||||||
|
Michael P.
McKenna, Ph.D. |
3/12/2021
|
-
|
-
|
$
|
-
|
-
|
79,750
|
(7)
|
$
|
627,633
|
-
|
-
|
|||||||||||||||||||||
|
8/31/2021
|
10,415
|
(6)
|
89,585
|
$
|
9.72
|
8/31/2031
|
-
|
$
|
-
|
-
|
-
|
||||||||||||||||||||||
|
|
|||||||||||||||||||||||||||||||||
|
Matthew
Dyer, Ph.D. |
1/11/2018
|
7,490
|
(8)
|
-
|
$
|
2.56
|
1/11/2028
|
-
|
$
|
-
|
-
|
-
|
|||||||||||||||||||||
|
8/23/2019
|
159,506
|
(9)
|
79,744
|
$
|
3.03
|
8/23/2029
|
-
|
$
|
-
|
-
|
-
|
||||||||||||||||||||||
|
8/23/2019
|
115,135
|
(10)
|
39,886
|
$
|
3.03
|
8/23/2029
|
-
|
$
|
-
|
-
|
-
|
||||||||||||||||||||||
|
5/17/2020
|
60,766
|
(11)
|
73,228
|
$
|
2.90
|
5/17/2030
|
-
|
$
|
-
|
-
|
-
|
||||||||||||||||||||||
|
3/12/2021
|
-
|
-
|
$
|
-
|
-
|
79,750
|
(12)
|
$
|
627,633
|
-
|
-
|
||||||||||||||||||||||
|
|
|||||||||||||||||||||||||||||||||
|
Christian
LaPointe, Ph.D. |
2/17/2021
|
-
|
-
|
$
|
-
|
-
|
170,346
|
(14)
|
$
|
1,340,623
|
-
|
-
|
|||||||||||||||||||||
|
8/31/2021
|
-
|
50,000
|
(13)
|
$
|
9.72
|
8/31/2031
|
-
|
$
|
-
|
-
|
-
|
||||||||||||||||||||||
| (1) |
The market value of the stock awards is based on the closing price of our Class A common stock of $7.87 per share on December 31, 2021.
|
| (2) |
25% of the RSUs vested on January 7, 2022 and the remainder vests, subject to continued service, in 12 equal quarterly installments
thereafter beginning with the quarter ending March 31, 2022. All of Mr. Stark’s unvested RSUs were forfeited on February 8, 2022 in connection with Mr. Stark’s separation.
|
| (3) |
The RSUs vest, subject to continued service (i) on the closing of a financing in excess of $50 million within three years of Mr.
Stark’s commencement of employment with Legacy Quantum-Si at a share price greater than $16.08, or (ii) if within three years of Mr. Stark’s start date the publicly-listed closing price of our shares is $16.08 or more for any
20 trading days within any 30 consecutive trading day period. These RSUs were forfeited on February 8, 2022 in connection with Mr. Stark’s separation.
|
|
(4)
|
The shares underlying this option vest, subject to continued service, as follows: 25% on June 30, 2022, with the remainder vesting in 36 equal monthly installments thereafter.
|
| (5) |
The RSUs vest, subject to continued service, as follows: 25% on June 30, 2022, with the remainder vesting in 12 equal quarterly installments thereafter.
|
|
(6)
|
The shares underlying this option vest, subject to continued service, in 48 equal monthly installments beginning on August 31, 2021.
|
| (7) |
The RSUs vest, subject to continued service, as follows: 25% on March 12, 2022, with the remainder vesting in 12 equal quarterly installments thereafter.
|
| (8) |
The shares underlying this option vest, subject to continued service, as follows: 25% of the shares vested on December 31, 2018, with the remainder vesting in equal monthly installments over the following 36 months.
|
| (9) |
The shares underlying this option vest, subject to continued service, in 48 equal monthly installments beginning on January 31, 2019.
|
| (10) |
The shares underlying this option vest, subject to continued service, in 48 equal monthly installments beginning on May 31, 2019.
|
| (11) |
The shares underlying this option vest, subject to continued service, in 48 equal monthly installments beginning on January 31, 2020.
|
| (12) |
The RSUs vest, subject to continued service, as follows: 25% on March 12, 2022, with the remainder vesting in 12 equal quarterly installments thereafter.
|
|
(13)
|
The shares underlying this option vest, subject to continued service, as follows: 25% on August 31, 2022, with the remainder vesting in 36 equal monthly installments thereafter.
|
| (14) |
25% of the RSUs vested on January 7, 2022 and remainder vests, subject to continued service, in 12 equal quarterly installments thereafter beginning with the quarter ending March 31, 2022.
|
|
Option Awards
|
||||||||
|
Name
|
Number of Shares Acquired on Exercise (#)
|
Value Realized on Exercise ($) (1)
|
||||||
|
John Stark
|
-
|
$
|
-
|
|||||
|
Claudia Drayton
|
-
|
$
|
-
|
|||||
|
Michael P. McKenna, Ph.D.
|
-
|
$
|
-
|
|||||
|
Matthew Dyer, Ph.D.
|
142,114
|
$
|
987,744
|
|||||
|
Christian LaPointe, Ph.D.
|
-
|
$
|
-
|
|||||
|
(1)
|
The value realized on exercise is based on the difference between the closing price of our Class A common stock on Nasdaq on the date of exercise and the applicable exercise price of those
options and does not represent actual amounts received by the individual as a result of the option exercises.
|
|
•
|
Severance payable in the form of salary continuation or a lump sum payment. The severance amount is equal to participant’s then-current base salary times a multiplier determined based on the
participant’s title or role with us. The multiplier for our Chief Executive Officer is 1.0 and the multiplier for our other executive officers is 0.75.
|
|
•
|
The portion of any outstanding unvested equity award that would vest on an annual cliff vesting date in accordance with the terms of the award during the three months following the participant’s
termination date will vest as of the date the termination of such participant’s employment becomes effective.
|
|
•
|
We will pay for company contribution for continuation coverage under the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended (“COBRA”) during the severance period.
|
|
•
|
Severance payable in a single lump sum. The severance amount is equal to participant’s then-current base salary and then-current target annual bonus opportunity, times a change in control
multiplier determined based on the participant’s title or role with us. The multiplier for our Chief Executive Officer is 1.5 and the multiplier for our other executive officers is 1.0.
|
|
•
|
Any outstanding unvested equity awards held by the participant under any then-current outstanding equity incentive plan(s) will become fully vested as of the date the termination of such
participant’s employment becomes effective.
|
|
•
|
We will pay for company contribution for continuation coverage under COBRA during the severance period.
|
|
Name
|
Compensation Component
|
Termination Without Cause
Absent a Change in Control ($)
|
Termination Without Cause or
For Good Reason Within 12
Months Following a Change of
Control ($)
|
|||||||
|
John Stark (2)
|
Cash compensation
|
$ |
500,000
|
(3)
|
$
|
1,250,000
|
(5)
|
|||
|
Acceleration of unvested
options and RSUs |
$ |
3,351,565
|
(1)
|
$
|
16,757,787
|
(1)
|
||||
|
Benefits and Perquisites
|
$ |
26,905
|
$
|
40,357
|
||||||
|
|
|
|||||||||
|
Claudia Drayton
|
Cash compensation
|
$ |
288,750
|
(3)
|
$
|
577,500
|
(5)
|
|||
|
Acceleration of unvested
options and RSUs |
$ |
-
|
$
|
753,159
|
(1)
|
|||||
|
Benefits and Perquisites
|
$ |
14,457
|
(4)
|
$
|
19,275
|
(4)
|
||||
|
|
|
|||||||||
|
Michael P. McKenna, Ph.D.
|
Cash Compensation
|
$ |
330,000
|
(3)
|
$
|
660,000
|
(5)
|
|||
|
Acceleration of unvested
options and RSUs |
$ |
156,904
|
(1)
|
$
|
627,633
|
(1)
|
||||
|
Benefits and Perquisites
|
$ |
15,394
|
(4)
|
$
|
20,525
|
(4)
|
||||
|
|
|
|||||||||
|
Matthew Dyer, Ph.D.
|
Cash compensation
|
$ |
300,000
|
(3)
|
$
|
600,000
|
(5)
|
|||
|
Acceleration of unvested
options and RSUs |
$ |
156,904
|
(1)
|
$
|
1,570,585
|
(1)
|
||||
|
Benefits and Perquisites
|
$ |
20,179
|
(4)
|
$
|
26,905
|
(4)
|
||||
|
|
|
|||||||||
|
Christian LaPointe, Ph.D.
|
Cash compensation
|
$ |
281,250
|
(3)
|
$
|
562,500
|
(5)
|
|||
|
Acceleration of unvested
options and RSUs |
$ |
335,152
|
(1)
|
$
|
1,340,623
|
(1)
|
||||
|
Benefits and Perquisites
|
$ |
15,394
|
$
|
20,525
|
||||||
| (1) |
Value attributable to accelerated vesting of (i) then unvested options, determined by multiplying the number of shares accelerated by the difference between the exercise price of the option
and the closing price of our shares on December 31, 2021, and (ii) then unvested RSUs, determined by multiplying the number of RSUs accelerated by the closing price of our shares on December 31, 2021. The closing price of
our shares on December 31, 2021 was $7.87.
|
| (2) |
Mr. Stark’s employment with us, and his service as a member of the Board, terminated effective February 8, 2022. The terms of his separation agreement are discussed above under “Employment
Arrangements – John Stark.”
|
| (3) |
Twelve months of 2021 base salary continuation for our former CEO and nine months of 2021 base salary continuation for our other NEOs.
|
| (4) |
Payment of COBRA premiums during the base salary continuation period.
|
| (5) |
Eighteen months of 2021 base salary continuation for our former CEO and twelve months of 2021 base salary continuation for our other NEOs including full bonus payout.
|
|
Name
|
Fees Earned or
Paid in Cash ($)(1) |
Stock Awards ($) (2)
|
Option Awards ($) (2)
|
All Other
Compensation ($) |
Total
|
|||||||||||||||
|
Jonathan M. Rothberg, Ph.D.
|
$
|
30,673
|
$
|
12,994,992
|
(3)
|
$
|
-
|
$
|
221,831
|
(3)
|
$
|
13,247,496
|
||||||||
|
Marijn Dekkers, Ph.D.
|
$
|
37,459
|
$
|
1,479,492
|
(5) |
$
|
-
|
$
|
-
|
$
|
1,516,951
|
|||||||||
|
Ruth Fattori
|
$
|
41,621
|
$
|
1,479,492
|
(5) |
$
|
-
|
$
|
-
|
$
|
1,521,113
|
|||||||||
|
Brigid A. Makes
|
$
|
38,846
|
$
|
199,992
|
$
|
-
|
$
|
-
|
$
|
238,838
|
||||||||||
|
Michael Mina, M.D., Ph.D. (4)
|
$
|
27,747
|
$
|
199,992
|
$
|
2,094,023
|
$
|
-
|
$
|
2,321,762
|
||||||||||
|
Kevin Rakin
|
$
|
30,522
|
$
|
199,992
|
$
|
-
|
$
|
-
|
$
|
230,514
|
||||||||||
|
James Tananbaum, M.D
|
$
|
31,909
|
$
|
199,992
|
$
|
-
|
$
|
-
|
$
|
231,901
|
||||||||||
| (1) |
Amounts represent fees earned during 2021 under our Non-Employee Director Compensation Policy.
|
| (2) |
Amount represents the aggregate grant date fair value for options and RSUs, computed in accordance with FASB ASC Topic 718. Each non-employee director was granted 20,512 RSUs upon their
appointment as directors of the Company following the Business Combination on June 11, 2021. A discussion of the assumptions used in determining grant date fair value may be found in Note 12 “Equity Incentive Plan” in our
consolidated financial statements included elsewhere in this Annual Report on Form 10-K. The RSUs vest in equal annual installments over three years beginning on June 11, 2022, subject to the director’s continued service
through the applicable vesting date.
|
| (3) |
In connection with the Business Combination Agreement, Legacy Quantum-Si and Dr. Rothberg, the founder of Legacy Quantum-Si,
Interim CEO and Executive Chairman of our Board, entered into the Executive Chairman Agreement, effective as of the Closing of the Business Combination pursuant to which Dr. Rothberg advised our CEO and provide guidance to
the Board. The amount included in the table represents the grant date fair value of a restricted stock unit award granted to Dr. Rothberg in connection with entering into the Executive Chairman Agreement and cash payments
paid pursuant to the Executive Chairman Agreement in 2021. A discussion of the terms of the Executive Chairman Agreement can be found below under “Item 13 - Certain Relationships and Related Transactions, and Director
Independence - Executive Chairman Agreement with Jonathan M. Rothberg, Ph.D.” Dr. Rothberg will not receive any additional compensation for serving as Interim CEO.
|
| (4) |
On April 19, 2021, Michael Mina, M.D., Ph.D. entered into a consulting agreement with Quantum-Si to serve as Legacy Quantum-Si’s
Chief Medical Advisor. Under the terms of the consulting agreement, Dr. Mina was eligible to receive $22,500 per month for 60% of full-time service to us. Also pursuant to the terms of the consulting agreement, Dr. Mina
was granted an option to purchase shares of Legacy Quantum-Si common stock with an exercise price equal to the fair market value of the common stock on the grant date. The option to purchase 358,875 shares has a per share
exercise price of $9.46 and vests in equal monthly installments over three years beginning on May 31, 2021, subject to Dr. Mina’s continued service on each vesting date provided, however, that during any monthly period
when Dr. Mina’s commitment to us is less than 60% of full time service, the number of shares that vest that month would be reduced proportionately based on the reduction in service relative to Dr. Mina’s 60% of full time
service commitment, and those unvested shares will be forfeited back to us. During 2021, Dr. Mina did not provide any consulting services to us under the consulting agreement and therefore none of the shares underlying
his options have vested and he did not receive any cash compensation. The consulting agreement was terminated on February 14, 2022 and his options were cancelled in its entirety on February 14, 2022 and nothing was
exercisable.
|
| (5) |
Each of Dr. Dekkers and Ms. Fattori received 150,000 RSUs in their capacity as directors of Legacy Quantum-Si. The RSUs vest in
equal annual installments over three years beginning on June 11, 2022, subject to continued service through the applicable vesting date.
|
|
Name
|
Number of Stock
Options Held at
Fiscal Year-End |
Number of Restricted
Stock Units Held at
Fiscal Year-End |
|||||||
|
Jonathan M. Rothberg, Ph.D.
|
-
|
1,520,512
|
|||||||
|
Marijn Dekkers, Ph.D.
|
-
|
170,512
|
|||||||
|
Ruth Fattori
|
-
|
170,512
|
|||||||
|
Brigid A. Makes
|
-
|
20,512
|
|||||||
|
Michael Mina, M.D., Ph.D.
|
279,123
|
20,512
|
|||||||
|
Kevin Rakin
|
-
|
20,512
|
|||||||
|
James Tananbaum, M.D.
|
-
|
20,512
|
|||||||
|
Position
|
Retainer
|
|||
|
Audit committee chairperson
|
$
|
20,000
|
||
|
Audit committee member
|
$
|
10,000
|
||
|
Compensation committee chairperson
|
$
|
15,000
|
||
|
Compensation committee member
|
$
|
7,500
|
||
|
Nominating and corporate governance committee chairperson
|
$
|
10,000
|
||
|
Nominating and corporate governance committee member
|
$
|
5,000
|
||
|
ITEM 12.
|
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
|
| • |
each person known to us to be the beneficial owner of more than 5% of our outstanding common stock;
|
| • |
each of our executive officers and directors; and
|
| • |
all of our executive officers and directors as a group.
|
|
Name and Address of Beneficial Owner
|
Number of
shares of
Class A
Common
Stock |
%
|
Number of
shares
Class B
Common
stock
|
%
|
% of Total
Voting Power**
|
|||||||||||||||
|
Directors and Executive Officers:
|
||||||||||||||||||||
|
Jonathan M. Rothberg, Ph.D. (1)
|
15,692,967
|
13.2
|
%
|
19,937,500
|
100.0
|
%
|
80.1
|
%
|
||||||||||||
|
John Stark (2)
|
245,996
|
*
|
-
|
-
|
*
|
|||||||||||||||
|
Claudia Drayton
|
-
|
-
|
-
|
-
|
-
|
|||||||||||||||
|
Michael P. McKenna, Ph.D. (3)
|
834,105
|
*
|
-
|
-
|
*
|
|||||||||||||||
|
Matthew Dyer, Ph.D. (4)
|
658,647
|
*
|
-
|
-
|
*
|
|||||||||||||||
|
Christian LaPointe, Ph.D. (5)
|
90,580
|
*
|
-
|
-
|
*
|
|||||||||||||||
|
Marijn Dekkers, Ph.D. (6)
|
549,980
|
*
|
-
|
-
|
*
|
|||||||||||||||
|
Ruth Fattori (7)
|
49,980
|
*
|
-
|
-
|
*
|
|||||||||||||||
|
Brigid A. Makes
|
-
|
-
|
-
|
-
|
-
|
|||||||||||||||
|
Michael Mina, M.D., Ph.D.
|
-
|
-
|
-
|
-
|
-
|
|||||||||||||||
|
Kevin Rakin (8)
|
1,890,000
|
1.6
|
%
|
-
|
-
|
*
|
||||||||||||||
|
James Tananbaum, M.D. (9)
|
8,403,805
|
7.1
|
%
|
-
|
-
|
1.6
|
%
|
|||||||||||||
|
All Current Directors and Executive Officers as a Group (11 Individuals) (10)
|
28,170,064
|
23.6
|
%
|
19,937,500
|
100.0
|
%
|
82.4
|
%
|
||||||||||||
|
Five Percent Holders:
|
||||||||||||||||||||
|
Jonathan M. Rothberg, Ph.D. (1)
|
15,692,967
|
13.2
|
%
|
19,937,500
|
100.0
|
%
|
8.1
|
%
|
||||||||||||
|
ARK Investment Management LLC (11)
|
13,067,150
|
11.0
|
%
|
-
|
-
|
2.5
|
%
|
|||||||||||||
|
Foresite Capital (9)
|
8,403,805
|
7.1
|
%
|
-
|
-
|
1.6
|
%
|
|||||||||||||
|
Glenview Capital Management, LLC (12)
|
6,000,000
|
5.1
|
%
|
-
|
-
|
1.2
|
%
|
|||||||||||||
| * |
Indicates beneficial ownership of less than 1%.
|
| ** |
Percentage of total voting power represents voting power with respect to all shares of our Class A common stock and our Class B common stock as a single class. Each share of our Class B
common stock is entitled to 20 votes per share and each share of our Class A common stock is entitled to 1 vote per share.
|
| (1) |
Consists of 15,692,967 shares of our Class A common stock and 19,937,500 shares of our Class B common stock held by Jonathan M. Rothberg, Ph.D., Dr. Rothberg’s spouse, 4C Holdings I, LLC, 4C
Holdings V, LLC, 2012 JMR Trust Common, LLC and 23rd Century Capital LLC. Dr. Rothberg, Legacy Quantum-Si’s founder and our Interim CEO and Executive Chairman, is the sole manager of 4C Holdings I, LLC, 4C Holdings V, LLC
and 2012 JMR Trust Common, LLC and has sole voting and investment control of our Class A common stock and our Class B common stock owned by those entities. Dr. Rothberg’s son is the manager of 23rd Century Capital LLC. Dr. Rothberg disclaims beneficial ownership of the shares held by his spouse and 23rd Century Capital LLC.
|
| (2) |
Consists of shares of our Class A common stock held by Mr. Stark, our former CEO.
|
| (3) |
Consists of (i) 797,500 shares of our Class A common stock held by Dr. McKenna, (ii) 19,939 shares of our Class A common stock issuable upon vesting of RSUs within 60 days of February 15, 2022
held by Dr. McKenna, and (iii) options to purchase 16,666 shares of our Class A common stock issuable upon the exercise of options to purchase shares of our Class A common stock exercisable within 60 days of February 15,
2022 held by Dr. McKenna.
|
| (4) |
Consists of (i) 261,743 shares of our Class A common stock held by Dr. Dyer, (ii) 19,939 shares of our Class A common stock issuable upon vesting of RSUs within 60 days of February 15, 2022
held by Dr. Dyer, and (iii) options to purchase 376,965 shares of our Class A common stock issuable upon the exercise of options to purchase shares of our Class A common stock exercisable within 60 days of February 15, 2022
held by Dr. Dyer.
|
| (5) |
Consists of (i) 79,933 shares of our Class A common stock held by Dr. LaPointe. and (ii) 10,647 shares of our Class A common stock issuable upon vesting of RSUs within 60 days of February 15,
2022 held by Dr. LaPointe.
|
| (6) |
Consists of (i) 37,485 shares of our Class A common stock held by Dr. Dekkers, (ii) 12,495 shares of our Class A common stock issuable upon vesting of RSUs within 60 days of February 15, 2022
held by Dr. Dekkers, and (iii) 500,000 shares of our Class A common stock held by Novalis Lifesciences Investments I, LP (“Novalis”). Dr. Dekkers has sole voting and investment control over the shares held by Novalis.
|
| (7) |
Consists of (i) 37,485 shares of our Class A common stock held by Ms. Fattori and (ii) 12,495 shares of our Class A common stock issuable upon vesting of RSUs within 60 days of February 15,
2022 held by Ms. Fattori.
|
| (8) |
Consists of (i) 89,000 shares of our Class A common stock held by Mr. Rakin and the Kevin L. Rakin Irrevocable Trust, (ii) 601,000 shares of our Class A common stock held by HighCape Partners
QSI II Invest, L.P, (iii) 24,527 shares of our Class A common stock held by HighCape Partners II, L.P. and (iv) 1,175,473 shares of our Class A common stock held by HighCape Partners QP II, L.P. Mr. Rakin and Matt Zuga are
the managing members of HighCape Capital II GP, LLC, which is the general partner of HighCape Partners II GP, L.P., which is the general partner of each of HighCape Partners QSI II Invest, L.P, HighCape Partners II, L.P. and
HighCape Partners QP II, L.P., and as a result each may be deemed to share voting and investment discretion with respect to the common stock held by such entities. Mr. Rakin disclaims any beneficial ownership of the
securities to be held by HighCape Partners QSI II Invest, L.P, HighCape Partners II, L.P. and HighCape Partners QP II, L.P. other than to the extent of any pecuniary interest he may have therein, directly or indirectly. The
business address of each of these entities or individuals is 452 Fifth Avenue, 21st Floor, New York, NY 10018.
|
| (9) |
Based on Schedule 13D filed by Foresite Capital Management, LLC on June 21, 20221. Consists of 4,463,619 shares of our Class A common stock held by Foresite Capital Fund IV, L.P. (“Foresite
IV”) 2,342,061 shares of our Class A common stock held by Foresite Capital Fund V, L.P. (“Foresite V”) and 1,598,125 shares of our Class A common stock held by Foresite Capital Opportunity Fund V, L.P. (“Foresite
Opportunity”). Foresite Capital Management IV, LLC (“FCM IV”) is the general partner of Foresite IV and may be deemed to have sole voting and dispositive power over shares held by Foresite IV. Foresite Capital Management
V, LLC (“FCM V”) is the general partner of Foresite V and Foresite Opportunity and may be deemed to have sole voting and dispositive power over shares held by Foresite V and Foresite Opportunity. Dr. James Tananbaum is the
sole managing member of FCM IV and FCM V and may be deemed to have sole voting and dispositive power over shares held by Foresite IV, Foresite V and Foresite Opportunity. Each of FCM IV, FCM V and Dr. Tananbaum disclaims
beneficial ownership of shares held by Foresite IV, Foresite V and Foresite Opportunity except to the extent of any pecuniary interest therein. The address of Foresite IV, Foresite V, Foresite Opportunity, FCM IV, FCM V and
Dr. Tananbaum is 600 Montgomery Street, Suite 4500, San Francisco, CA 94111.
|
| (10) |
See footnotes 1 and 3 through 9.
|
| (11) |
Based on Schedule 13G/A filed by ARK Investment Management LLC (“ARK”) on February 9, 2022. Consists of shares of our Class A common stock held by ARK. The business address of ARK is 3 East
28th Street, 7th Floor, New York, New York 10016.
|
| (12) |
Based on Schedule 13G/A filed by Glenview Capital Management, LLC (“Glenview Capital Management”) on February 14, 2022. Consists of 261,362 shares of our Class A common stock held for the
account of Glenview Capital Partners, L.P. (“Glenview Capital Partners”), 1,913,372 shares of our Class A common stock held for the account of Glenview Capital Master Fund, Ltd., 641,271 shares of our Class A common stock
held for the account of Glenview Institutional Partners, L.P., 1,369,620 shares of our Class A common stock held for the account of Glenview Offshore Opportunity Master Fund, Ltd., 1,673,485 shares of our Class A common
stock held for the account of Glenview Capital Opportunity Fund, L.P., and 140,890 shares of our Class A common stock held for the account of Glenview Healthcare Master Fund (collectively, the Glenview Investment Funds).
Glenview Capital Management serves as investment manager to each of the Glenview Investment Funds. Larry Robbins is the Chief Executive Officer of Glenview Capital Management. The address of the principal business office
for Mr. Robbins, Glenview Capital Management and the Glenview Investment Funds is 767 Fifth Avenue, 44th Floor, New York, New York 10153.
|
| (a) |
(b) |
(c) |
|||||||||||||||||||
|
Plan category
|
Plan category
Number of securities to be issued upon exercise of outstanding options, warrants and rights |
Weighted-average exercise
price of outstanding
options,
warrants and rights
|
Number of securities
remaining available for
future issuance under
equity compensation plans
(excluding securities
reflected in column (a))
|
||||||||||||||||||
|
Equity compensation plans approved by security holders
|
12,313,944
|
(1)
|
$
|
5.14
|
(2)
|
11,891,127
|
(3)
|
||||||||||||||
|
Equity compensation plans not approved by security holders
|
-
|
-
|
-
|
||||||||||||||||||
|
Total
|
12,313,944
|
$
|
5.14
|
11,891,127
|
(4)
|
||||||||||||||||
| (1) |
Consists of (i) 10,935,482 shares to be issued upon exercise of outstanding options and RSUs under the 2013 Plan and (ii) 1,378,462 shares to be issued upon exercise of outstanding options and
RSUs under the 2021 Plan.
|
| (2) |
Consists of the weighted-average exercise price of the $5.14 stock options outstanding on December 31, 2021.
|
| (3) |
Consists of shares that remained available for future issuance under the 2021 Plan as of December 31, 2021. No shares remained available for future issuance under the 2013 Plan as of December
31, 2021.
|
| (4) |
The 2021 Plan has an evergreen provision that allows for an annual increase in the number of shares available for issuance under the 2021 Plan to be added on the first day of each fiscal year,
beginning in fiscal year 2022 and ending on the second day of fiscal year 2031. The evergreen provides for an automatic increase in the number of shares available for issuance equal to the lesser of (i) 4% of the number of
outstanding shares of common stock on such date and (ii) an amount determined by the plan administrator.
|
|
Name
|
Shares
|
Aggregate
Purchase Price
|
Date of Issuance
|
||||||
|
Foresite Capital Fund IV, L.P.
|
1,865,672
|
$
|
10,000,002
|
February 21, 2020
|
|||||
|
Foresite Capital Fund IV, L.P.
|
3,731,343
|
$
|
19,999,998
|
December 29, 2020
|
|||||
|
Foresite Capital Fund V, L.P.
|
932,836
|
$
|
5,000,001
|
December 29, 2020
|
|||||
| • |
any person who is or was an executive officer, director, or director nominee of ours at any time since the beginning of our last fiscal year;
|
| • |
a person who is or was an Immediate Family Member (as defined below) of an executive officer, director, director nominee at any time since the beginning of our last fiscal year;
|
| • |
any person who, at the time of the occurrence or existence of the transaction, is the beneficial owner of more than 5% of any class of our voting securities, or a Significant Stockholder; or
|
| • |
any person who, at the time of the occurrence or existence of the transaction, is an Immediate Family Member of a Significant Stockholder of ours.
|
| • |
the related person’s interest in the transaction;
|
| • |
the approximate dollar value of the amount involved in the transaction;
|
| • |
the approximate dollar value of the amount of the related person’s interest in the transaction without regard to the amount of any profit or loss;
|
| • |
whether the transaction was undertaken in the ordinary course of our business;
|
| • |
whether the transaction with the related person is proposed to be, or was, entered into on terms no less favorable to us than terms that could have been reached with an unrelated third party;
|
| • |
the purpose of, and the potential benefits to us of, the transaction; and
|
| • |
any other information regarding the transaction or the related person in the context of the proposed transaction that would be material to investors in light of the circumstances of the
particular transaction.
|
|
|
2021
|
2020
|
||||||
|
Audit fees (1)
|
$
|
1,431,200
|
$
|
43,775
|
||||
|
Audit-related fees (2)
|
1,155,000
|
-
|
||||||
|
Tax fees (2)
|
-
|
-
|
||||||
|
All other fees (2)
|
-
|
-
|
||||||
|
Total
|
$
|
2,586,200
|
$
|
43,775
|
||||
| (1) |
Audit fees consisted of audit work performed in the preparation of consolidated financial statements, as well as work generally only the independent registered public accounting firm can
reasonably be expected to provide, such as quarterly review procedures and the provision of consents in connection with the filing of registration statements and related amendments, as well as other filings.
|
| (2) |
Audited-related fees consisted of services related to the Business Combination in 2021. There were no tax and other related fees in 2021 or 2020, and audit-related fees in 2020.
|
|
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
|
| Page | |
|
(a). 1. Index to Consolidated Financial Statements as of December 31, 2021 and 2020 and for the years ended December 31, 2021, 2020
and 2019
|
|
|
|
|
|
Report of Independent Registered Public Accounting Firm (
|
130 |
| 132 | |
| 133 | |
| 134 | |
| 135 | |
| 136 |
|
Exhibit
Number
|
Exhibit Description
|
|
Filed Herewith |
|
Incorporated by
Reference Herein
from
Form or Schedule
|
|
Filing Date
|
SEC File/
Reg. Number
|
||
|
Business Combination Agreement, dated as of February 18, 2021, by and among Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.), Clay Merger Sub, Inc., and Q-SI Operations Inc. (formerly
Quantum-Si Incorporated)
|
Form 8-K
(Exhibit 2.1)
|
|
2/18/2021
|
001-39486
|
||||||
|
Second Amended and Restated Certificate of Incorporation of Quantum-Si Incorporated
|
Form 8-K
(Exhibit 3.1)
|
|
6/15/2021
|
001-39486
|
||||||
|
Amended and Restated Bylaws of Quantum-Si Incorporated
|
X
|
|||||||||
|
Description of Securities
|
X
|
|||||||||
|
Specimen Class A Common Stock Certificate
|
Form S-4/A
(Exhibit 4.1)
|
|
5/11/2021
|
333-253691
|
||||||
|
Warrant Agreement, dated as of September 3, 2020, by and between Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.) and Continental Stock Transfer & Trust Company
|
Form 8-K
(Exhibit 4.1)
|
|
9/9/2020
|
001-39486
|
|
Form of PIPE Investor Subscription Agreement for institutional investors, dated as of February 18, 2021, by and between Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.) and the
subscriber parties thereto
|
Form 8-K
(Exhibit 10.1)
|
|
2/18/2021
|
001-39486
|
||||||
|
Form of PIPE Investor Subscription Agreement for accredited investors, dated as of February 18, 2021, by and between Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.) and the subscriber
parties thereto
|
Form 8-K/A
(Exhibit 10.2)
|
|
2/19/2021
|
001-39486
|
||||||
|
Form of Subscription Agreement, dated as of February 18, 2021, by and between Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.) and the Foresite Funds
|
Form 8-K/A
(Exhibit 10.3)
|
|
2/19/2021
|
001-39486
|
||||||
|
Transaction Support Agreement, dated as of February 19, 2021, by and among Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.), and certain supporting stockholders of Q-SI Operations Inc.
(formerly Quantum-Si Incorporated)
|
Form 8-K
(Exhibit 10.1)
|
|
2/22/2021
|
001-39486
|
||||||
|
Sponsor Letter Agreement, dated as of February 18, 2021, by and among HighCape Capital Acquisition LLC, Deerfield Partners, L.P., Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.) and
Q-SI Operations Inc. (formerly Quantum-Si Incorporated)
|
Form 8-K
(Exhibit 10.4)
|
|
2/18/2021
|
001-39486
|
||||||
|
Executive Chairman Agreement, dated as of June 10, 2021, by and between Quantum-Si Incorporated and Jonathan M. Rothberg, Ph.D.
|
Form 8-K
(Exhibit 10.6)
|
|
6/15/2021
|
001-39486
|
||||||
|
10.7+
|
Offer Letter of Employment, dated as of October 28, 2020, by and between Q-SI Operations Inc. (formerly Quantum-Si Incorporated) and John Stark
|
|
Form S-4 (Exhibit 10.9) |
|
3/1/2021
|
333-253691
|
||||
|
Separation Agreement, dated as of February 11, 2022, by and between Quantum-Si Incorporated and John Stark
|
|
|
Form 8-K
(Exhibit 10.1)
|
|
2/14/2022
|
001-39486
|
||||
|
Offer Letter of Employment, dated as of March 23, 2021, by and between Q-SI Operations Inc. (formerly Quantum-Si Incorporated) and Claudia Drayton
|
Form S-4/A
(Exhibit 10.10)
|
|
5/11/2021
|
333-253691
|
||||||
|
Offer Letter of Employment, dated as of June 1, 2015, by and between Q-SI Operations Inc. (formerly Quantum-Si Incorporated) and Michael P. McKenna, Ph.D.
|
Form S-4
(Exhibit 10.10)
|
|
3/1/2021
|
333-253691
|
|
Offer Letter of Employment, dated as of March 16, 2016, by and between Q-SI Operations Inc. (formerly Quantum-Si Incorporated) and Matthew Dyer, Ph.D.
|
Form S-4
(Exhibit 10.11)
|
|
3/1/2021
|
333-253691
|
||||||
|
Offer Letter of Employment, dated as of November 4, 2020, by and between Q-SI Operations Inc. (formerly Quantum-Si Incorporated) and Christian LaPointe, Ph.D., as supplemented by the Letter Agreement, dated
as of February 16, 2021, by and between Q-SI Operations Inc. and Christian LaPointe, Ph.D.
|
X
|
|||||||||
|
Consulting Agreement, dated as of April 19, 2021, by and between Q-SI Operations Inc. (formerly Quantum-Si Incorporated) and Michael
Mina, M.D., Ph.D.
|
Form S-4/A
(Exhibit 10.13)
|
|
5/11/2021
|
333-253691
|
||||||
|
Technology and Services Exchange Agreement, dated as of February 17, 2021, by and among Q-SI Operations Inc. (formerly Quantum-Si Incorporated) and the participants named therein
|
|
Form 10-Q
(Exhibit 10.1)
|
|
11/15/2021
|
001-39486
|
|||||
|
Binders Collaboration Agreement, dated as of September 20, 2021, by and between Quantum-Si Incorporated and Protein Evolution, Inc.
|
|
Form 10-Q
(Exhibit 10.2)
|
|
11/15/2021
|
001-39486
|
|||||
|
Quantum-Si Incorporated 2021 Equity Incentive Plan
|
|
|
Form 8-K
(Exhibit 10.13.1)
|
|
6/15/2021
|
001-39486
|
||||
|
Form of Stock Option Agreement under 2021 Equity Incentive Plan
|
Form 8-K
(Exhibit 10.13.2)
|
6/15/2021
|
001-39486
|
|||||||
|
Form of Restricted Stock Unit Agreement under 2021 Equity Incentive Plan
|
|
|
Form S-8
(Exhibit 99.3)
|
|
9/2/2021
|
333-259271
|
||||
|
Q-SI Operations Inc. 2013 Employee, Director and Consultant Equity Incentive Plan, as amended
|
|
|
Form 8-K
(Exhibit 10.14.1)
|
|
6/15/2021
|
001-39486
|
||||
|
Form of Stock Option Agreement under 2013 Employee, Director and Consultant Equity Incentive Plan, as amended
|
Form 8-K
(Exhibit 10.14.2)
|
6/15/2021
|
001-39486
|
|||||||
|
Form of Restricted Stock Unit Agreement under 2013 Employee, Director and Consultant Equity Incentive Plan, as amended
|
|
|
Form 8-K
(Exhibit 10.14.3)
|
|
6/15/2021
|
001-39486
|
||||
|
Nonemployee Director Compensation Policy
|
|
|
Form 8-K
(Exhibit 10.15)
|
|
6/15/2021
|
001-39486
|
||||
|
Form of Indemnification Agreement
|
Form 8-K
(Exhibit 10.16)
|
6/15/2021
|
001-39486
|
|
Amended and Restated Registration Rights Agreement, dated as of June 10, 2021, by and among Quantum-Si Incorporated (formerly HighCape Capital Acquisition Corp.) and certain of its securityholders
|
Form 8-K
(Exhibit 10.17)
|
6/15/2021
|
001-39486
|
|||||||
|
Lease Agreement between Quantum-Si Incorporated and BP3-SD5 5510 Morehouse Drive LLC, dated June 18, 2021
|
Form 8-K
(Exhibit 10.1)
|
6/24/2021
|
001-39486
|
|||||||
|
Lease Agreement between Quantum-Si Incorporated and Winchester Office LLC, dated December 28, 2021
|
Form 8-K
(Exhibit 10.1)
|
1/24/2022
|
001-39486
|
|||||||
|
Quantum-Si Incorporated Executive Severance Plan
|
Form 8-K
(Exhibit 10.1)
|
7/6/2021
|
001-39486
|
|||||||
|
List of Subsidiaries
|
Form 8-K
(Exhibit 21.1)
|
6/15/2021
|
001-39486
|
|||||||
|
Consent of Deloitte & Touche LLP
|
X
|
|||||||||
|
Certification of the Principal Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
|
|
X
|
|
|
|
|||||
|
Certification of the Principal Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
|
|
X
|
|
|
|
|||||
|
Certifications of the Chief Executive Officer and Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
|
|
X
|
|
|
|
|
|
|||
|
101.INS
|
Inline XBRL Instance Document (the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document)
|
|
X
|
|
|
|
|
|||
|
101.SCH
|
Inline XBRL Taxonomy Extension Schema Document
|
|
X
|
|
|
|
|
|||
|
101.CAL
|
Inline XBRL Taxonomy Extension Calculation Linkbase Document
|
|
X
|
|
|
|
|
|||
|
101.DEF
|
Inline XBRL Taxonomy Extension Definition Linkbase Document
|
|
X
|
|
|
|
||||
|
101.LAB
|
Inline XBRL Taxonomy Extension Label Linkbase Document
|
|
X
|
|
|
|
||||
|
101.PRE
|
Inline XBRL Taxonomy Extension Presentation Linkbase Document
|
|
X
|
|
|
|
||||
|
104
|
Cover Page Interactive Data File (embedded within the Inline XBRL document)
|
|
X
|
|
|
|
|
|
QUANTUM-SI INCORPORATED
|
|
|
March 1, 2022
|
|
|
|
By:
|
/s/ Jonathan M. Rothberg, Ph.D.
|
|
|
|
|
Jonathan M. Rothberg, Ph.D.
|
|
|
|
Interim Chief Executive Officer
|
|
Name
|
Title
|
Date
|
||
|
/s/ Jonathan M. Rothberg, Ph.D.
|
Interim Chief Executive Officer and Executive Chairman
|
March 1, 2022
|
||
|
Jonathan M. Rothberg, Ph.D.
|
(Principal Executive Officer) | |||
|
/s/ Claudia Drayton
|
Chief Financial Officer
|
March 1, 2022
|
||
|
Claudia Drayton
|
(Principal Financial and Accounting Officer) | |||
|
/s/ Marijn Dekkers, Ph.D.
|
Director
|
March 1, 2022
|
||
|
Marijn Dekkers, Ph.D.
|
||||
|
/s/ Ruth Fattori
|
Director
|
March 1, 2022
|
||
|
Ruth Fattori
|
||||
|
/s/ Brigid A. Makes
|
Director
|
March 1, 2022
|
||
|
Brigid A. Makes
|
||||
|
/s/ Michael Mina, M.D., Ph.D.
|
Director
|
March 1, 2022
|
||
|
Michael Mina, M.D., Ph.D.
|
||||
|
/s/ Kevin Rakin
|
Director
|
March 1, 2022
|
||
|
Kevin Rakin
|
||||
|
/s/ James Tananbaum, M.D.
|
Director
|
March 1, 2022
|
||
|
James Tananbaum, M.D.
|
| • |
We tested management’s computation of the purchase price and determination of goodwill recognized focusing on the completeness and accuracy of the
assets acquired, and liabilities assumed and related fair value purchase price allocations made to identified assets acquired and liabilities assumed and the fair value of the consideration paid, specifically, equity instruments issued and
contingent payments.
|
| • |
We obtained and evaluated the valuation estimates prepared by specialists engaged by the Company, and challenged management’s review of the
appropriateness of the valuations assessed and allocation to assets acquired and liabilities assumed. The procedures included but were not limited to, testing critical inputs, including discount rates and the valuation models utilized by the
Company’s specialists.
|
| • |
With the assistance of our fair value specialists, we evaluated the
reasonableness of the (1) valuation models and (2) discount rates by:
|
| – |
Testing the source information underlying the determination of the discount rates and testing the mathematical accuracy of the calculations.
|
| – |
Developing a range of independent estimates and comparing those to the discount rates selected by management.
|
|
December 31,
2021
|
December 31,
2020
|
|||||||
|
Assets
|
||||||||
|
Current assets:
|
||||||||
|
Cash and cash equivalents
|
$
|
|
$
|
|
||||
|
Marketable securities
|
|
|
||||||
|
Prepaid expenses and other current assets
|
|
|
||||||
|
Total current assets
|
|
|
||||||
|
Property and equipment, net
|
|
|
||||||
|
Goodwill
|
|
|
||||||
|
Other assets
|
|
|
||||||
|
Other assets - related party
|
|
|
||||||
|
Operating lease right-of-use assets
|
|
|
||||||
|
Total assets
|
$
|
|
$
|
|
||||
|
Liabilities, convertible preferred stock and stockholders’ equity (deficit)
|
||||||||
|
Current liabilities:
|
||||||||
|
Accounts payable
|
$
|
|
$
|
|
||||
|
Accrued expenses and other current liabilities
|
|
|
||||||
|
Short-term operating lease liabilities
|
|
|
||||||
|
Total current liabilities
|
|
|
||||||
|
Long-term liabilities:
|
||||||||
|
Warrant liabilities
|
|
|
||||||
|
Notes payable
|
|
|
||||||
|
Other long-term liabilities
|
|
|
||||||
|
Operating lease liabilities
|
|
|
||||||
|
Total liabilities
|
|
|
||||||
|
Commitments and contingencies (Note 17)
|
||||||||
|
Convertible preferred stock
|
||||||||
|
Convertible preferred stock (Series A, B, C, D, and E) $
|
|
|
||||||
|
Stockholders’ equity (deficit)
|
||||||||
|
Class A Common stock, $
|
|
|
||||||
|
Class B Common stock, $
|
|
|
||||||
|
Additional paid-in capital
|
|
|
||||||
|
Accumulated deficit
|
(
|
)
|
(
|
)
|
||||
|
Total stockholders’ equity (deficit)
|
|
(
|
)
|
|||||
|
Total liabilities, convertible preferred stock and stockholders’ equity (deficit)
|
$
|
|
$
|
|
||||
|
Years ended December 31,
|
||||||||||||
|
2021
|
2020
|
2019
|
||||||||||
|
Operating expenses:
|
||||||||||||
|
Research and development
|
$
|
|
$
|
|
$
|
|
||||||
|
General and administrative
|
|
|
|
|||||||||
|
Sales and marketing
|
|
|
|
|||||||||
|
Total operating expenses
|
|
|
|
|||||||||
|
Loss from operations
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Interest expense
|
(
|
)
|
(
|
)
|
|
|||||||
|
Dividend income
|
|
|
|
|||||||||
|
Change in fair value of warrant liabilities
|
|
|
|
|||||||||
|
Other (expense) income, net
|
(
|
)
|
(
|
)
|
|
|||||||
|
Loss before provision for income taxes
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Provision for income taxes
|
|
|
|
|||||||||
|
Net loss and comprehensive loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Net loss per common share attributable to common stockholders, basic and diluted
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Weighted-average shares used to compute net loss per share attributable to common stockholders, basic and diluted
|
|
|
|
|||||||||
|
Convertible preferred stock
|
Class A
common stock
|
Class B
common stock
|
Additional
paid-in
|
Accumulated |
Total
stockholders’
equity
|
|||||||||||||||||||||||||||||||
|
Shares
|
Amount
|
Shares
|
Amount
|
Shares
|
Amount
|
capital
|
deficit
|
(deficit)
|
||||||||||||||||||||||||||||
|
Balance - January 1, 2019
|
|
$
|
|
|
$
|
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
(
|
)
|
|||||||||||||||||||
|
Net loss
|
-
|
-
|
-
|
|
-
|
|
|
(
|
)
|
(
|
)
|
|||||||||||||||||||||||||
|
Issuance of Series E convertible preferred stock, net of issuance costs
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
|
Common stock issued upon exercise of stock options
|
-
|
-
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
|
Stock-based compensation expense
|
-
|
-
|
-
|
|
-
|
|
|
|
|
|||||||||||||||||||||||||||
|
Balance - December 31, 2019
|
|
|
|
|
|
|
|
(
|
)
|
(
|
)
|
|||||||||||||||||||||||||
|
Net loss
|
-
|
-
|
-
|
|
-
|
|
|
(
|
)
|
(
|
)
|
|||||||||||||||||||||||||
|
Issuance of Series E convertible preferred stock, net of issuance costs
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
|
Common stock issued upon exercise of stock options
|
-
|
-
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
|
Stock-based compensation expense
|
-
|
-
|
-
|
|
-
|
|
|
|
|
|||||||||||||||||||||||||||
|
Balance - December 31, 2020
|
|
|
|
|
|
|
|
(
|
)
|
(
|
)
|
|||||||||||||||||||||||||
|
Net loss
|
-
|
-
|
-
|
|
-
|
|
|
(
|
)
|
(
|
)
|
|||||||||||||||||||||||||
|
Issuance of Series E convertible preferred stock, net of issuance costs
|
|
(
|
)
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||
|
Common stock issued upon exercise of stock options and vesting of restricted stock units
|
-
|
-
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
|
Conversion of the convertible preferred stock into Class A and Class B common stock
|
(
|
)
|
(
|
)
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||
|
Net equity infusion from the Business Combination
|
-
|
-
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
|
Majelac Technologies LLC Acquisition
|
-
|
-
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||
|
Stock-based compensation expense
|
-
|
-
|
-
|
|
-
|
|
|
|
|
|||||||||||||||||||||||||||
|
Balance - December 31, 2021
|
|
$
|
|
|
$
|
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
||||||||||||||||||||
|
Years ended December 31,
|
||||||||||||
|
2021
|
2020
|
2019
|
||||||||||
|
Cash flows from operating activities:
|
||||||||||||
|
Net loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
||||||||||||
|
Depreciation
|
|
|
|
|||||||||
|
Unrealized losses of marketable securities
|
|
|
|
|||||||||
|
Loss on disposal of fixed assets
|
|
|
|
|||||||||
|
Change in fair value of warrant liabilities
|
(
|
)
|
|
|
||||||||
|
Change in fair value of contingent consideration
|
|
|
|
|||||||||
|
Stock-based compensation expense
|
|
|
|
|||||||||
|
Write-off of intellectual property
|
|
|
|
|||||||||
|
Changes in operating assets and liabilities:
|
||||||||||||
|
Prepaid expenses and other current assets
|
(
|
)
|
(
|
)
|
|
|||||||
|
Other assets
|
(
|
)
|
|
|
||||||||
|
Other assets - related party
|
|
|
(
|
)
|
||||||||
|
Operating lease right-of-use assets
|
(
|
)
|
|
|
||||||||
|
Accounts payable
|
|
|
(
|
)
|
||||||||
|
Accrued expenses and other current liabilities
|
|
|
|
|||||||||
|
Short-term operating lease liabilities
|
|
|
|
|||||||||
|
Operating lease liabilities
|
|
|
|
|||||||||
|
Net cash used in operating activities
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Cash flows from investing activities:
|
||||||||||||
|
Purchases of property and equipment
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Purchases of marketable securities
|
(
|
)
|
|
|
||||||||
|
Business acquisition
|
(
|
)
|
|
|
||||||||
|
Net cash used in investing activities
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Cash flows from financing activities:
|
||||||||||||
|
Proceeds from exercise of stock options
|
|
|
|
|||||||||
|
Proceeds from issuance of Series E convertible preferred stock
|
|
|
|
|||||||||
|
Net proceeds from equity infusion from the Business Combination
|
|
|
|
|||||||||
|
Proceeds from issuance of notes payable
|
|
|
|
|||||||||
|
Payment of notes payable
|
(
|
)
|
|
|
||||||||
|
Stock issuance costs for Series E convertible preferred stock
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Principal payments under finance lease obligations
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Net cash provided by financing activities
|
$
|
|
$
|
|
$
|
|
||||||
|
Net (decrease) increase in cash and cash equivalents
|
(
|
)
|
|
(
|
)
|
|||||||
|
Cash and cash equivalents at beginning of period
|
|
|
|
|||||||||
|
Cash and cash equivalents at end of period
|
$
|
|
$
|
|
$
|
|
||||||
|
Supplemental disclosure of cash flow information:
|
||||||||||||
|
Cash received from exchange of research and development tax credits
|
$
|
|
$
|
|
$
|
|
||||||
|
Supplemental disclosure of noncash information:
|
||||||||||||
|
Noncash acquisition of property and equipment
|
$
|
|
$
|
|
$
|
|
||||||
|
Forgiveness of related party promissory notes
|
$
|
|
$
|
|
$
|
|
||||||
|
Noncash equity issuance - business acquisition
|
$
|
|
$
|
|
$
|
|
||||||
|
Noncash equity related warrants from the Business Combination
|
$
|
|
$
|
|
$
|
|
||||||
|
Conversion of the convertible preferred stock into Class A and Class B common stock
|
$
|
|
$
|
|
$
|
|
||||||
|
Noncash contingent consideration and holdbacks - business acquisition
|
$ |
$ |
$ |
|||||||||
| • |
valuation allowances with respect to deferred tax assets;
|
| • |
valuation for acquisitions;
|
| • |
assumptions used for leases;
|
| • |
valuation of warrant liabilities; and
|
| • |
assumptions underlying the fair value used in the calculation of the stock-based compensation.
|
|
Property and equipment
|
Estimated useful life
|
|
|
Laboratory and production equipment
|
|
|
|
Computer equipment
|
|
|
|
Software
|
|
|
|
Furniture and fixtures
|
|
| • |
each share of Legacy Quantum-Si capital stock (other than shares of Legacy Quantum-Si Series A preferred stock) that was issued and outstanding as of immediately prior to the Effective Time was
automatically cancelled and extinguished and converted into the right to receive a number of shares of the Company’s Class A common stock equal to the Exchange Ratio, rounded down to the nearest whole number of shares;
|
| • |
each share of Legacy Quantum-Si Series A preferred stock that was issued and outstanding as of immediately prior to the Effective Time was automatically cancelled and extinguished and converted into the
right to receive a number of shares of the Company’s Class B common stock equal to the Exchange Ratio, rounded down to the nearest whole number of shares;
|
| • |
each option to purchase shares of Legacy Quantum-Si common stock, whether vested or unvested, that was outstanding and unexercised as of immediately prior to the Effective Time was assumed by the Company
and became an option (vested or unvested, as applicable) to purchase a number of shares of the Company’s Class A common stock equal to the number of shares of Legacy Quantum-Si common stock subject to such option immediately prior to the
Effective Time multiplied by the Exchange Ratio, rounded down to the nearest whole number of shares, at an exercise price per share equal to the exercise price per share of such option immediately prior to the Effective Time divided by
the Exchange Ratio, rounded up to the nearest whole cent; and
|
| • |
each Legacy Quantum-Si restricted stock unit outstanding immediately prior to the Effective Time was assumed by the Company and became a restricted stock unit with respect to a number of shares of the
Company’s Class A common stock equal to the number of shares of Legacy Quantum-Si common stock subject to such Legacy Quantum-Si restricted stock unit immediately prior to the Effective Time multiplied by the Exchange Ratio, rounded down
to the nearest whole share.
|
| • |
|
| • |
|
| • |
|
| • |
|
| • |
|
| • |
|
|
Purchase Price
Allocation
|
||||
|
Prepaid expenses and other current assets
|
$
|
|
||
|
Property and equipment, net
|
|
|||
|
Goodwill
|
|
|||
|
Total
|
$ |
|
||
| • |
Level 1 - Valuations based on quoted prices in active markets for identical assets or liabilities that an entity has the ability to access.
|
| • |
Level 2 - Valuations based on quoted prices for similar assets or liabilities, quoted prices for identical assets or liabilities in markets that are not active, or other inputs that are observable or can be corroborated by observable
data for substantially the full term of the assets or liabilities.
|
| • |
Level 3 - Valuations based on inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
|
|
Fair Value Measurement Level
|
||||||||||||||||
|
Total
|
Level 1
|
Level 2
|
Level 3
|
|||||||||||||
|
December 31, 2021:
|
||||||||||||||||
|
Assets:
|
||||||||||||||||
|
Mutual funds - Cash and cash equivalents
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Mutual funds - Marketable securities
|
|
|
|
|
||||||||||||
|
Total assets at fair value on a recurring basis
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
|
||||||||||||||||
|
Liabilities:
|
||||||||||||||||
|
Public Warrants
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Private Warrants
|
|
|
|
|
||||||||||||
|
Total liabilities at fair value on a recurring basis
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Fair Value Measurement Level
|
||||||||||||||||
|
Total
|
Level 1
|
Level 2
|
Level 3
|
|||||||||||||
|
December 31, 2020:
|
||||||||||||||||
|
Assets:
|
||||||||||||||||
|
Mutual funds - Cash and cash equivalents
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Total assets at fair value on a recurring basis
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
|
||||||||||||||||
|
Liabilities:
|
||||||||||||||||
|
Notes payable
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Total liabilities at fair value on a recurring basis
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
December 31,
2021
|
December 31,
2020
|
|||||||
|
Laboratory and production equipment
|
$
|
|
$
|
|
||||
|
Computer equipment
|
|
|
||||||
|
Software
|
|
|
||||||
|
Furniture and fixtures
|
|
|
||||||
|
Leasehold improvements
|
|
|
||||||
|
Construction in process
|
|
|
||||||
|
|
|
|||||||
|
Less: Accumulated depreciation
|
(
|
)
|
(
|
)
|
||||
|
Property and equipment, net
|
$
|
|
$
|
|
||||
|
December 31,
2021
|
December 31,
2020
|
|||||||
|
Employee compensation
|
$
|
|
$
|
|
||||
|
Contracted services
|
|
|
||||||
|
Business acquisition costs and contingencies
|
|
|
||||||
|
Legal fees
|
|
|
||||||
|
Other
|
|
|
||||||
|
Total accrued expenses and other current liabilities
|
$
|
|
$
|
|
||||
|
Year Ended
December 31, 2021
|
||||
|
Operating lease cost
|
$
|
|
||
|
Short-term lease cost
|
|
|||
|
Variable lease cost
|
|
|||
|
Total lease cost
|
$
|
|
||
|
Operating Leases
|
||||
|
Weighted-average remaining lease term (years)
|
|
|||
|
Weighted-average discount rate
|
|
%
|
||
|
Operating Leases
|
||||
|
Operating cash paid to settle operating lease liabilities
|
$
|
|
||
|
Right-of-use assets obtained in exchange for lease liabilities
|
$
|
|
||
|
Operating Leases
|
||||
|
2022
|
$
|
|
||
|
2023
|
|
|||
|
2024
|
|
|||
|
2025
|
|
|||
|
2026
|
|
|||
|
Thereafter
|
|
|||
|
Total undiscounted lease payments
|
$
|
|
||
|
Less: Imputed interest
|
|
|||
|
Total lease liabilities
|
$
|
|
||
|
Three Months Ended
March 31, 2021
|
Three Months Ended
June 30, 2021
|
Three Months Ended
September 30, 2021
|
||||||||||
|
Operating lease cost
|
$
|
|
$
|
|
$
|
|
||||||
|
Short-term lease cost
|
|
|
|
|||||||||
|
Variable lease cost
|
|
|
|
|||||||||
|
Total lease cost
|
$
|
|
$
|
|
$
|
|
||||||
|
September 30, 2021
|
||||
|
Operating lease right-of-use assets
|
$
|
|
||
|
Short-term operating lease liabilities
|
|
|||
|
Operating lease liabilities
|
|
|||
|
Weighted-average remaining lease term (years)
|
|
|||
|
Weighted-average discount rate
|
|
%
|
||
|
Class
|
Year of
Class
Issuance
|
Issuance
Price per
Share
|
Shares
Authorized
|
Shares
Issued and
Outstanding
|
Total
Proceeds or
Exchange
Value
|
Issuance
Costs
|
Net
Carrying
Value
|
Initial
Liquidation
Price per
Share
|
|||||||||||||||||||||||
|
Series A
|
|
$
|
|
|
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||||||||||||
|
Series B
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
|
Series C
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
|
Series D
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
|
Series E
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
Number
of Options
|
Weighted
Average
Exercise
Price
|
Weighted
Average
Remaining
Contractual Term
(Years)
|
Aggregate
Intrinsic
Value
|
|||||||||||||
|
Outstanding at December 31, 2020
|
|
$
|
|
|
$
|
|
||||||||||
|
Granted
|
|
|
||||||||||||||
|
Exercised
|
(
|
)
|
|
|||||||||||||
|
Forfeited
|
(
|
)
|
|
|||||||||||||
|
Outstanding at December 31, 2021
|
|
$
|
|
|
$
|
|
||||||||||
|
Options exercisable at December 31, 2021
|
|
|
|
$
|
|
|||||||||||
|
Vested and expected to vest at December 31, 2021
|
|
$
|
|
|
$
|
|
||||||||||
|
|
|
2021
|
|
2020
|
2019
|
|
|
Risk-free interest rate
|
|
|
|
|
|
|
|
Expected dividend yield
|
|
|
|
|
|
|
|
Expected term
|
|
|
|
|
|
|
|
Expected volatility
|
|
|
|
|
|
|
|
|
2019
|
|
Risk-free interest rate
|
|
|
|
Expected dividend yield
|
|
|
|
Expected term
|
|
|
|
Expected volatility
|
|
|
|
Number
of Shares
Underlying RSUs
|
Weighted Average
Grant-Date
Fair Value
|
|||||||
|
Outstanding non-vested RSUs at December 31, 2020
|
|
$
|
|
|||||
|
Granted
|
|
|
||||||
|
Vested
|
(
|
)
|
|
|||||
|
Forfeited
|
|
|
||||||
|
Outstanding non-vested RSUs at December 31, 2021
|
|
$
|
|
|||||
|
Years ended December 31,
|
||||||||||||
|
2021
|
2020
|
2019
|
||||||||||
|
Research and development
|
$
|
|
$
|
|
$
|
|
||||||
|
General and administrative
|
|
|
|
|||||||||
|
Sales and marketing
|
|
|
|
|||||||||
|
Total stock-based compensation expense
|
$
|
|
$
|
|
$
|
|
||||||
|
Years ended December 31,
|
||||||||||||
|
2021
|
2020
|
2019
|
||||||||||
|
Numerator
|
||||||||||||
|
Net loss
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Numerator for basic and diluted EPS - loss attributable to common stockholders
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Denominator
|
||||||||||||
|
Common stock
|
|
|
|
|||||||||
|
Denominator for basic and diluted EPS - weighted-average common stock
|
|
|
|
|||||||||
|
Basic and diluted net loss per share
|
$
|
(
|
)
|
$
|
(
|
)
|
$
|
(
|
)
|
|||
|
Years ended December 31,
|
||||||||||||
|
2021
|
2020
|
2019
|
||||||||||
|
Outstanding options to purchase common stock
|
|
|
|
|||||||||
|
Outstanding restricted stock units
|
|
|
|
|||||||||
|
Outstanding warrants
|
|
|
|
|||||||||
|
Outstanding convertible preferred stock (Series A through E)
|
|
|
|
|||||||||
|
|
|
|
||||||||||
| • |
in whole and not in part;
|
| • |
at a price of $
|
| • |
upon not less than
|
| • |
if, and only if, the closing price of the Company’s common stock equals or exceeds $
|
|
Years Ended December 31,
|
||||||||||||
|
2021
|
2020
|
2019
|
||||||||||
|
Statutory tax rate
|
|
%
|
|
%
|
|
%
|
||||||
|
State taxes, net of federal benefit
|
|
|
|
|||||||||
|
Federal research and development credit
|
|
|
|
|||||||||
|
Stock-based compensation expense
|
|
(
|
)
|
(
|
)
|
|||||||
|
Other
|
|
(
|
)
|
|
||||||||
|
Valuation allowance
|
(
|
)
|
(
|
)
|
(
|
)
|
||||||
|
Effective tax rate
|
|
%
|
|
%
|
|
%
|
||||||
|
As of December 31,
|
||||||||
|
2021
|
2020
|
|||||||
|
Deferred tax assets
|
||||||||
|
Net operating loss carryforwards
|
$
|
|
$
|
|
||||
|
Tax credit carryforwards
|
|
|
||||||
|
Stock-based compensation expense
|
|
|
||||||
|
Operating lease liabilities
|
|
|
||||||
|
Other
|
|
|
||||||
|
Total deferred tax assets
|
$
|
|
$
|
|
||||
|
Deferred tax liabilities
|
||||||||
|
Operating lease right-of-use assets
|
$
|
(
|
)
|
$
|
|
|||
|
Property and equipment
|
(
|
)
|
(
|
)
|
||||
|
Other
|
(
|
)
|
|
|||||
|
Total deferred tax liabilities
|
$
|
(
|
)
|
$
|
(
|
)
|
||
| Net deferred tax assets |
$ | $ | ||||||
|
Valuation allowance
|
(
|
)
|
(
|
)
|
||||
|
Net deferred tax assets (liabilities)
|
$
|
|
$
|
|
||||
|
Amount
|
Begin to
Expire In
|
|||||||
|
Tax net operating loss carryforwards:
|
||||||||
|
Federal (pre-2018 NOLs)
|
$
|
|
|
|||||
|
Federal (post-2017 NOLs)
|
|
No Expiration
|
||||||
|
State
|
|
|
||||||
|
Tax credit carryforwards:
|
||||||||
|
Federal research and development
|
|
|
||||||
|
Connecticut research and development
|
|
N/A
|
||||||
|
Connecticut other credits
|
|
|
