ASSET PURCHASE AGREEMENT
among
APPLIED UV, INC.
STERILUMEN, INC.
and
AKIDA HOLDINGS, LLC
SIMBA PARTNERS, LLC
JJH HOLDINGS, LLC
FAKHRUDDIN HOLDINGS FZC
dated as of
February 8, 2021
| i |
TABLE OF CONTENTS
| Article I Definitions | 1 |
| Article II Purchase and Sale. | 6 |
| Section 2.01 Purchase and Sale of Assets. | 6 |
| Section 2.02 Liabilities. | 8 |
| Section 2.03 Purchase Price. | 8 |
| Section 2.04 Allocation of Purchase Price. | 8 |
| Article III Closing | 8 |
| Section 3.01 Closing. | 8 |
| Section 3.02 Closing Deliverables. | 8 |
| Article IV Representations and warranties of seller and each member | 10 |
| Section 4.01 Organization and Qualification of Seller. | 10 |
| Section 4.02 Authority of Seller. | 10 |
| Section 4.03 No Conflicts; Consents. | 11 |
| Section 4.04 Financial Statements. | 11 |
| Section 4.05 Bankruptcy. | 11 |
| Section 4.06 Undisclosed Liabilities. | 11 |
| Section 4.07 Absence of Certain Changes, Events and Conditions. | 12 |
| Section 4.08 Assigned Contracts. | 12 |
| Section 4.09 Title to Acquired Assets. | 12 |
| Section 4.10 Condition of Assets. | 12 |
| Section 4.11 Intellectual Property. | 13 |
| Section 4.12 Insurance. | 14 |
| Section 4.13 Business Relationships. | 14 |
| Section 4.14 Absence of Certain Business Practices. | 14 |
| Section 4.15 Legal Proceedings; Governmental Orders. | 15 |
| Section 4.16 Compliance with Laws; Permits. | 15 |
| Section 4.17 Employee Benefit Matters. | 15 |
| Section 4.18 Employment Matters. | 16 |
| Section 4.19 Environmental Matters. | 16 |
| Section 4.20 Restricted Securities. | 16 |
| Section 4.21 Status of Seller and Members. | 16 |
| Section 4.22 Investment Risk. | 16 |
| Section 4.23 Restrictive Legends. | 16 |
| Section 4.24 Commissions. | 17 |
| Section 4.25 Taxes. | 17 |
| Section 4.26 FDA Compliance. | 17 |
| Section 4.27 Accounts Receivable. | 17 |
| Article V Representations and warranties of Purchaser | 18 |
| Section 5.01 Organization of Parent. | 18 |
| Section 5.02 Organization of Purchaser. | 18 |
| Section 5.03 Authority of Purchaser. | 18 |
| Section 5.04 No Conflicts; Consents. | 18 |
| Section 5.05 Legal Proceedings. | 18 |
| Section 5.06 Shares. | 19 |
| Section 5.07 No Commissions. | 19 |
| Section 5.08 Access and Investigation; No Other Representations and Warranties | 19 |
| Article VI Covenants | 19 |
| Section 6.01 Post-Closing Covenants. | 19 |
| Section 6.02 Non-solicitation | 20 |
| Section 6.03 Governmental Approvals and Consents. | 21 |
| Section 6.04 Licensing Agreement. | 21 |
| Section 6.05 Governmental Approvals and Consents. | 21 |
| Section 6.06 Books and Records. | 21 |
| Section 6.07 Public Announcements. | 21 |
| Section 6.08 Receivables. | 22 |
| Section 6.09 Transfer Taxes. | 22 |
| Section 6.10 Further Assurances. | 22 |
| Article VII Indemnification | 22 |
| Section 7.01 Survival. | 22 |
| Section 7.02 Indemnification by Seller and the Members. | 23 |
| Section 7.03 Purchaser Indemnification. | 24 |
| Section 7.04 Indemnification Procedures. | 24 |
| Section 7.05 Payments. | 26 |
| Section 7.06 Tax Treatment of Indemnification Payments. | 26 |
| Section 7.07 Exclusive Remedies. | 26 |
| Article VIII Miscellaneous | 26 |
| Section 8.01 Expenses. | 26 |
| Section 8.02 Notices. | 26 |
| Section 8.03 Headings. | 27 |
| Section 8.04 Severability. | 27 |
| Section 8.05 Entire Agreement. | 27 |
| Section 8.06 Successors and Assigns. | 27 |
| Section 8.07 No Third-party Beneficiaries. | 28 |
| Section 8.08 Amendment and Modification; Waiver. | 28 |
| Section 8.09 Governing Law; Jurisdiction and Venue. | 28 |
| Section 8.10 Counterparts. | 28 |
| ii |
ASSET PURCHASE AGREEMENT
This Asset Purchase Agreement (this “Agreement”), dated as of February 8, 2021, is entered into by and among Applied UV, Inc., a Delaware corporation having its principal place of business at 150 N. Macquesten Parkway, Mount Vernon, NY 10550 (“Parent”) and SteriLumen, Inc., a New York corporation having its principal place of business at 8480 East Orchard Road, Suite 2400, Greenwood Village, Colorado 80111 (“Purchaser”) on the one hand and Akida Holdings LLC, a Florida limited liability company, having its principal place of business at 2300 Marshpoint Road, Suite 202, Neptune Beach, FL 32266 (“Seller”) and the Seller’s members, Simba Partners, LLC, JJH Holdings, LLC and Fakhruddin Holdings FZC on the other.
Each party may be individually referred to as a “Party,” and all parties may be collectively referred to as the “Parties.”
RECITALS
WHEREAS, Seller owns the exclusive world-wide rights to Airocide™, a unique NASA developed air purification technology and manufactures (through third parties), markets and sells devices based on such technology (the “Business”);
WHEREAS, Seller wishes to sell to Purchaser, the wholly-owned subsidiary of the Parent, and Parent and Purchaser wishes the Purchaser to purchase and assume from Seller, substantially all of the assets of the Seller, subject to the terms and conditions set forth herein; and
NOW, THEREFORE, in consideration of the mutual covenants and agreements hereinafter set forth and for other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto agree as follows:
Article
I
Definitions
Any capitalized terms not defined in this Article I have the meanings given to them in the respective section contained in this Agreement in which the first appear. The following terms have the meanings specified or referred to in this Article I:
“Acquired Assets” has the meaning set forth in Section 2.01(p).
“Acquired Bank Account” has the meaning set forth in Section 3.02(a)(xii).
“Action” means any claim, action, cause of action, demand, lawsuit, arbitration, inquiry, audit, notice of violation, proceeding, litigation, citation, summons, subpoena or investigation of any nature, whether civil, criminal, administrative, regulatory or otherwise, and whether at law or in equity.
“Affiliate” of a Person means any other Person that directly or indirectly, through one or more intermediaries, controls, is controlled by, or is under common control with, such Person. The term “control” (including the terms “controlled by” and “under common control with”) means the possession, directly or indirectly, of the power to direct or cause the direction of the management and policies of a Person, whether through the ownership of voting securities, by contract or otherwise. For all purposes of this Agreement, Kes shall be deemed to be an “Affiliate” of the Seller.
| 1 |
“Agreement” has the meaning set forth in the preamble.
“Allocation Schedule” has the meaning set forth in Section 2.04.
“Annual Financial Statements” has the meaning set forth in Section 4.04.
“Airocide” means the AiroCide™ Air Quality-Improvement™ System.
“Assigned Contracts” means the Contracts listed on Exhibit H.
“Assignment and Assumption Agreement” has the meaning set forth in Section 3.02(a)(iii).
“Basket Amount” has the meaning set forth in Section 7.02(c).
“Bill of Sale” has the meaning set forth in Section 3.02(a)(i).
“Books and Records” has the meaning set forth in Section 2.01(o).
“Business” has the meaning set forth in the recitals.
“Business Day” means any day except Saturday, Sunday or any other day on which commercial banks located in New York, NY are authorized or required by Law to be closed for business.
“Closing” has the meaning set forth in Section 3.01.
“Closing Date” has the meaning set forth in Section 3.01.
“Contracts” means all contracts, leases, deeds, mortgages, licenses, instruments, notes, commitments, undertakings, indentures, joint ventures and all other legally binding agreements, commitments and arrangements, whether written or oral.
“Current Assets” means the current assets of the Seller set forth on Schedule 1.
“Current Assets Value” means the aggregate value of the Current Assets on the Closing Date set forth on Schedule 1.
“Current Liabilities” means the current liabilities of the Seller set forth on Schedule 1.
“Current Liabilities Amount” means the aggregate outstanding amount of the Current Liabilities on the Closing Date as set forth on Schedule 1.
“Direct Claim” has the meaning set forth in Section 7.04(c).
“Employee Benefit Program” has the meaning set forth in Section 4.17.
“Environmental Laws” means any Law (x) relating to pollution (or the cleanup thereof or the filing of information with respect thereto), human health or the protection of air, surface water, ground water, drinking water supply, land (including land surface or subsurface), plant and animal life or Losses for injury or loss of natural resources, (y) concerning exposure to, or the use, storage, recycling, treatment, generation, transportation, processing, handling, labeling, production or disposal of regulated substances, or (z) that may impose liability or obligations for injuries or damages due to or threatened as a result of the presence of, exposure to, or ingestion of, any regulated substance, in each case as amended and as now or hereafter in effect.
| 2 |
“Encumbrance” means any charge, claim, community property interest, pledge, condition, equitable interest, lien (statutory or other), option, security interest, mortgage, easement, encroachment, right of way, right of first refusal, or restriction of any kind, including any restriction on use, voting, transfer, receipt of income or exercise of any other attribute of ownership.
“Excluded Assets” means the following assets owned by the Seller immediately prior to the Closing:
(a) The Purchase Price delivered to the Seller pursuant to the provisions contained in Section 2.03;
(b) The rights of the parties under this Agreement;
(c) Any and all rights and title for real property, except for any lease agreements included in the Assigned Contracts;
(d) Payroll and personnel records;
(e) Seller’s minute books and ownership record books;
(f) Prepaid taxes and any Tax refunds; and
(g) Seller’s Insurance Policies.
“Financial Statements” has the meaning set forth in Section 4.04.
“Fundamental Reps” means the representation and warranties of the Seller set forth in Section 4.01, Section 4.02, Section 4.09, Section 4.11(a), the first sentence of Section 4.11(b), Section 4.19, Section 4.21, Section 4.22, Section 4.24 and Section 4.25.
“GAAP” means United States generally accepted accounting principles in effect from time to time.
“Governmental Authority” means any federal, state, local or foreign government or political subdivision thereof, or any agency or instrumentality of such government or political subdivision, or any self-regulated organization or other non-governmental regulatory authority or quasi-governmental authority (to the extent that the rules, regulations or orders of such organization or authority have the force of Law), or any arbitrator, court or tribunal of competent jurisdiction.
“Governmental Order” means any order, writ, judgment, injunction, decree, stipulation, determination or award entered by or with any Governmental Authority.
“Hybrid Rep” means the representations and warranties contained in the second sentence of Section 4.11(b) and Section 4.11(c) through Section 4.11(i).
“Indemnified Party” has the meaning set forth in Section 7.04.
| 3 |
“Indemnifying Party” has the meaning set forth in Section 7.04.
“Insurance Policies” has the meaning set forth in Section 4.12.
“Intellectual Property” means with respect to the Business, all domestic or foreign rights in, to and concerning: (i) inventions and discoveries (whether patented, patentable or unpatentable and whether or not reduced to practice), including research and techniques, technical designs, and specifications (written or otherwise), improvements, modifications, adaptations, and derivations thereto, and patents, patent applications, inventor’s certificates, and patent disclosures, together with divisions, continuations, continuations-in-part, revisions, reissuances and reexaminations thereof; (ii) trademarks, service marks, brand names, certification marks, collective marks, d/b/a’s, trade dress, logos, symbols, trade names, assumed names, fictitious names, corporate names and other indications or indicia of origin, including translations, adaptations, derivations, modifications, combinations and renewals thereof; (iii) published and unpublished works of authorship, whether copyrightable or not (including databases and other compilations of data or information), copyrights therein and thereto, moral rights, and rights equivalent thereto, including the rights of attribution, assignation and integrity; (iv) trade secrets, confidential and/or proprietary information (including research and development, know-how, formulas, compositions, manufacturing and production processes and techniques, technical data, schematics, designs, discoveries, drawings, prototypes, specifications, hardware configurations, customer and supplier lists, financial information, pricing and cost information, financial projections, and business and marketing methods plans and proposals) (collectively “Trade Secrets”); (v) computer software, including programs, applications, source and object code, data bases, data, models, algorithms, flowcharts, tables and documentation related to the foregoing; (vi) other similar tangible or intangible intellectual property or proprietary rights, information and technology and copies and tangible embodiments thereof (in whatever form or medium); (vii) all applications to register, registrations, restorations, reversions and renewals or extensions of the foregoing; (viii) internet domain names; and (ix) all the goodwill associated with each of the foregoing and symbolized thereby; and (x) all other intellectual property or proprietary rights and claims or causes of action arising out of or related to any infringement, misappropriation or other violation of any of the foregoing, including rights to recover for past, present and future violations thereof.
“Interim Financial Statements” has the meaning set forth in Section 4.04.
“Kes” shall mean Kesair Technologies, LLC.
“Knowledge” means with respect to any Person, (x) such Person is actually aware of such fact or matter or (y) such Person should reasonably have been expected to discover or otherwise become aware of such fact or matter after reasonable investigation.
“Law” means any statute, law, ordinance, regulation, rule, code, order, constitution, treaty, common law, equitable doctrine, judgment, decree, other requirement or rule of law of any Governmental Authority.
“Liabilities” means liabilities, obligations or commitments of any nature whatsoever, asserted or unasserted, known or unknown, absolute or contingent, accrued or unaccrued, matured or unmatured or otherwise.
| 4 |
“Losses” means losses, damages, liabilities, deficiencies, judgments, interest, awards, penalties, fines, costs or expenses of whatever kind, including reasonable attorneys' fees and the cost of enforcing any right to indemnification hereunder and the cost of pursuing any insurance providers; provided, however, that “Losses” shall not include consequential, incidental or punitive damages, except in the case of fraud.
“Material Adverse Effect” means any event, occurrence, fact, condition or change that is, or could reasonably be expected to become, individually or in the aggregate, materially adverse to (a) the business, results of operations, condition (financial or otherwise) or assets of the Business, or (b) the value of the Acquired Assets.
“Members” means Simba Partners, LLC, JJH Holdings, LLC and Fakhruddin Holdings FZC.
“Parent” has the meaning set forth in the preamble.
“Permits” means all permits, licenses, franchises, approvals, authorizations, registrations, certificates, variances and similar rights obtained, or required to be obtained, from Governmental Authorities.
“Permitted Encumbrance” shall have the meaning set forth in Section 4.09.
“Person” means an individual, corporation, partnership, joint venture, limited liability company, Governmental Authority, unincorporated organization, trust, association or other entity.
“Pre-Closing Tax Period” means any taxable period ending on or before the Closing Date and, with respect to any taxable period beginning before and ending after the Closing Date, the portion of such taxable period ending on and including the Closing Date.
“Purchase Price” has the meaning set forth in Section 2.03.
“Purchase Price Value” means an amount equal to the (x) cash portion of the Purchase Price plus (y)(1) the number of Shares included in the Purchase Price multiplied by (2) the closing price of the Parent’s common stock on the Closing Date.
“Purchaser” has the meaning set forth in the preamble.
“Purchaser Indemnitees” has the meaning set forth in Section 7.02.
“Purchaser’s Closing Certificate” means a certificate of an officer of the Parent certifying that attached thereto are true and complete copies of (i) all formation documents of the Parent and Purchaser and (ii) all resolutions adopted by the board of directors of the Parent and the Purchaser, in each case, authorizing the execution, delivery and performance of this Agreement and the other Transaction Documents and the consummation of the transactions contemplated hereby and thereby, and that all such resolutions are in full force and effect and are all the resolutions adopted in connection with the transactions contemplated hereby and thereby.
“Representative” means, with respect to any Person, any and all directors, officers, employees, consultants, financial advisors, counsel, accountants and other agents of such Person.
“ROFR Shares” has the meaning set forth in Section 6.01(c).
| 5 |
“Seller” has the meaning set forth in the preamble.
“Seller Indemnities” has the meaning set forth in Section 7.03.
“Seller’s Closing Certificate” means a certificate of a Manager (or equivalent officer) of Seller certifying that attached thereto are true and complete copies of (i) all formation documents of the Seller and (ii) all resolutions adopted by the board of managers of Seller authorizing the execution, delivery and performance of this Agreement and the other Transaction Documents and the consummation of the transactions contemplated hereby and thereby, and that all such resolutions are in full force and effect and are all the resolutions adopted in connection with the transactions contemplated hereby and thereby.
“Selling Shareholder” has the meaning set forth in Section 6.01(b).
“Series A Units” The Series A Units of the Seller, issued pursuant to the Seller’s Second Amended and Restated Operating Agreement, dated September 10, 2014.
“Series A Consent” The consent of the holders of the Series A Units to the transactions contemplated by this Agreement, in form and substance reasonably acceptable to Purchaser.
“Taxes” means all federal, state, local, foreign and other income, gross receipts, sales, use, production, ad valorem, transfer, documentary, franchise, registration, profits, license, lease, service, service use, withholding, payroll, employment, unemployment, estimated, excise, severance, environmental, stamp, occupation, premium, property (real or personal), real property gains, windfall profits, customs, duties or other taxes, fees, assessments or charges of any kind whatsoever, together with any interest, additions or penalties with respect thereto and any interest in respect of such additions or penalties.
“Tax Return” means any return, declaration, report, claim for refund, information return or statement or other document relating to Taxes, including any schedule or attachment thereto, and including any amendment thereof.
“Third-Party Claim” has the meaning set forth in Section 7.04(a).
“Transaction Documents” means this Agreement, the Bill of Sale, the Assignment and Assumption Agreement, and the other agreements, instruments and documents required to be delivered at the Closing.
“Wire Transfer Instructions” has the meaning set forth in Section 3.02(a)(vii).
Article
II
Purchase and Sale.
Section 2.01 Purchase and Sale of Assets. Subject to the terms and conditions set forth herein, at the Closing, Seller shall sell, assign, transfer, convey and deliver to Purchaser, and Purchaser shall purchase the Acquired Assets from Seller, free and clear of any Encumbrances other than Permitted Encumbrances. The purchase of the Acquired Assets shall include all of Seller's right, title and interest in, to and under all of the assets, properties and rights of every kind and nature, whether real, personal or mixed, tangible or intangible (including goodwill), wherever located and whether now existing or hereafter acquired, which relate to, or are used or held for use in connection with the Business, including the following:
| 6 |
(a) all furniture, fixtures, equipment, plant tooling, scanners, machinery, tools, vehicles, office equipment, printers, supplies, computers, telephones and other tangible personal property owned by the Seller;
(b) all accounts and unbilled receivables held by Seller, and any security, claim, remedy or other right related to any of the foregoing to the extent not contributed as set forth on the Allocation Schedule;
(c) all commercial and consumer products related to the Business, including work in progress, deferred cost of services, supplies, parts and other inventories (“Inventory”);
(d) all deposits made to manufactures of AirocideTM consumer products;
(e) all cash on deposit in the Seller’s accounts as set forth in Schedule 1, which shall be deposited into the Acquired Bank Account prior to the Closing;
(f) all Assigned Contracts;
(g) the Intellectual Property set forth on Schedule 4.11(a);
(h) manufacturer, distributor and customer relationships in existence on the Closing Date;
(i) all marketing materials related to the Business;
(j) all Permits which are held by Seller and required for the conduct of the Business as currently conducted or for the ownership and use of the Acquired Assets to the extent such Permits are transferable;
(k) all rights to any Actions of any nature available to or being pursued by Seller to the extent related to the Business or the Acquired Assets, whether arising by way of counterclaim or otherwise;
(l) all prepaid expenses, un-deposited funds, credits, advance payments, claims, security, refunds, security deposits, rights of recovery, rights of set-off, rights of recoupment, deposits, charges, sums and fees (including any such items relating to the payment of Taxes);
(m) all of Seller's rights under warranties, indemnities and all similar rights against third parties to the extent related to any Acquired Assets;
(n) all insurance benefits, for policies held by Seller, including rights and proceeds, arising from or relating to the Business or the Acquired Assets;
(o) originals, or where not available, copies, of all books and records, including books of account, ledgers and general, financial and accounting records, machinery and equipment maintenance files, computer databases and software, customer lists, email addresses, customer purchasing histories, price lists, distribution lists, supplier lists, production data, quality control records and procedures, customer complaints and inquiry files, research and development files, records and data (including all correspondence with any Governmental Authority), sales material and records (including pricing history, total sales, terms and conditions of sale, sales and pricing policies and practices), strategic plans, internal financial statements, marketing and promotional surveys, material and research and files relating to the Intellectual Property, other than payroll and personnel records (“Books and Records”); and
| 7 |
(p) all other assets owned by the Seller (all of the Seller’s assets set forth in clauses (a) through (n) in this Section 2.01 and more specifically described in Exhibit A shall be referred to herein as “Acquired Assets;” provided, however, Acquired Assets shall not include Excluded Assets).
Section 2.02 Liabilities. Purchaser shall not assume and shall not be responsible to pay, perform or discharge any Liabilities or obligations of Seller or any of its Affiliates of any kind or nature whatsoever other than Liabilities of the Seller set forth in the Assigned Contracts and the Seller’s Current Liabilities.
Section 2.03 Purchase Price. The purchase price for the Acquired Assets shall be (i) $901,274.96 in cash and (ii) 1,375,000 unregistered shares of the Parent’s common stock (the “Purchase Price”).
The Parent shall pay the cash portion of the Purchase Price (the “Cash Portion”) on behalf of the Purchaser by a wire transfer of immediately available funds to the account set forth in the Wire Transfer Instructions. The shares of Parent common stock included in the Purchase Price (the “Shares”) shall be issued by the Parent to the Seller on the Closing Date in book-entry form.
Section 2.04 Allocation of Purchase Price. Seller, Parent and Purchaser agree that the Purchase Price plus other relevant items shall be allocated among the Acquired Assets for all purposes (including Tax and financial accounting) as shown on Exhibit B (the “Allocation Schedule”). Any state or federal income tax resulting from sale of the Acquired Assets shall be the sole responsibility of the Seller and the Members.
Article
III
Closing
Section 3.01 Closing. Subject to the terms and conditions of this Agreement, the consummation of the transactions contemplated by this Agreement (the “Closing”) shall take place simultaneously with the execution and delivery of this Agreement and the Transaction Documents by the delivery and exchange of signature pages sent by e-mail and/or facsimile. The date on which the Closing is to occur is herein referred to as the “Closing Date.” The Closing shall be effective as of Seller’s close of business on the Closing Date.
Section 3.02 Closing Deliverables.
(a) At or before the Closing, Seller shall deliver (or cause its Affiliates to deliver) to Purchaser the following:
| 8 |
(i) the Bill of Sale with Assignment and Assumption Agreement in the form of Exhibit C hereto (the “Bill of Sale”), duly executed by Seller;
(ii) the Assigned Contracts duly executed by each of the parties thereto;
(iii) the Assignment and Assumption Agreement, substantially in the form of Exhibit D (the “Assignment and Assumption Agreement”), duly executed by the Seller;
(iv) the Employment Agreement, duly executed by Keith Frein;
(v) the Consulting Agreement, duly executed by David Kight;
(vi) the Series A Consent, executed by 100% of the holders of the Series A Units;
(vii) wire transfer instructions, executed by the Seller, which sets forth the wiring information necessary for wiring funds into the account of the Seller (“Wire Transfer Instructions”);
(viii) the Seller’s Closing Certificate;
(ix) a certificate of good standing for the Seller from its state of formation; and
(x) a Settlement Statement, which sets forth the amount of the cash portion of the Purchase Price and any other payments to be made at Closing, if any, (the “Settlement Statement”) duly executed by Seller;
(xi) a complete written description of the Catalyst, its ingredients and components and instructions for producing the Catalyst and a video that shows how the glass tubules or similar substrates are coated with the proprietary titanium dioxide (TiO2) solution and placed inside Airocide’s reaction chambers;
(xii) the forms required by Valley National Bank necessary to transfer signature authority for Seller’s bank account (account number 42296137) (the “Acquired Bank Account”) to Keyoumars Saeed, Chief Executive Officer of Purchaser, duly executed by Seller; and
(xiii) such other customary instruments of transfer, assumption, filings or documents, in form and substance reasonably satisfactory to Purchaser, as may be required to give effect to this Agreement.
(b) At or before the Closing, the Parent or the Purchaser, as applicable, shall deliver to Seller the following:
(i) The cash portion of the Purchase Price by wire transfer of immediately available funds pursuant to the Wire Transfer Instructions;
(ii) Evidence of the issuance of the Shares to the Seller from the Parent’s transfer agent;
(iii) the Bill of Sale, duly executed by Purchaser;
(iv) the Assignment and Assumption Agreement, duly executed by Purchaser;
(v) the Employment Agreement, duly executed by the Purchaser;
| 9 |
(vi) the Consulting Agreement, duly executed by the Purchaser;
(vii) the Settlement Statement, duly executed by the Purchaser;
(viii) the Purchaser’s Closing Certificate; and
(ix) certificates of good standing for the Parent and the Purchaser from each of their states of formation.
Article
IV
Representations and warranties of seller and each member
Seller and each Member hereby represents and warrants to Purchaser that the statements contained in this Article IV are true and correct as of the Closing Date.
Section 4.01 Organization and Qualification of Seller. Seller is a Florida limited liability company, having its principal place of business at 2300 Marshpoint Road, Suite 202, Neptune Beach, FL 32266. The only members of the Seller are Simba Partners, LLC, JJH Holdings, LLC and Fakhruddin Holdings FZC. Seller is licensed or qualified to do business and is in good standing in each jurisdiction in which the ownership of the Acquired Assets or the operation of the Business as currently conducted makes such licensing or qualification necessary.
Section 4.02 Authority of Seller. Seller has full power and authority to enter into this Agreement and the other Transaction Documents to which it is a party, to carry out its obligations hereunder and thereunder and to consummate the transactions contemplated hereby and thereby. The execution and delivery by Seller of this Agreement and any other Transaction Documents to which Seller is a party, the performance by Seller of its obligations hereunder and thereunder and the consummation by Seller of the transactions contemplated hereby and thereby have been duly authorized by all requisite limited liability company action on the part of Seller. This Agreement has been duly executed and delivered by Seller, and (assuming due authorization, execution and delivery by Purchaser) this Agreement constitutes a legal, valid and binding obligation of Seller enforceable against Seller in accordance with its terms, subject to applicable bankruptcy, fraudulent conveyance, moratorium and similar Laws affecting creditors’ remedies and rights generally. When each other Transaction Document to which Seller is or will be a party has been duly executed and delivered by Seller (assuming due authorization, execution and delivery by each other party thereto), such Transaction Document will constitute a legal and binding obligation of Seller enforceable against it in accordance with its terms, subject to applicable bankruptcy, fraudulent conveyance, moratorium and similar Laws affecting creditors’ remedies and rights generally.
| 10 |
Section 4.03 No Conflicts; Consents. Except as set forth on Schedule 4.03, the execution, delivery and performance by Seller of this Agreement and the other Transaction Documents to which it is a party, and the consummation of the transactions contemplated hereby and thereby, do not and will not: (a) conflict with or result in a violation or breach of, or default under, any provision of the operating agreement, the articles of organization or other organizational documents of Seller; (b) conflict with or result in a violation or breach of any provision of any Law or Governmental Order applicable to Seller, the Business or the Acquired Assets; (c) require the consent, notice or other action by any Person under, conflict with, result in a violation or breach of, constitute a default or an event that, with or without notice or lapse of time or both, would constitute a default under, result in the acceleration of or create in any party the right to accelerate, terminate, modify or cancel any Contract or Permit to which Seller is a party or by which Seller or the Business is bound or to which any of the Acquired Assets are subject (including any Assigned Contract); or (d) result in the creation or imposition of any Encumbrance other than Permitted Encumbrances on the Acquired Assets. No consent, approval, Permit, Governmental Order, declaration or filing with, or notice to, any Governmental Authority is required by or with respect to Seller in connection with the execution and delivery of this Agreement or any of the other Transaction Documents and the consummation of the transactions contemplated hereby and thereby.
Section 4.04 Financial Statements. Attached hereto as Schedule 4.04 are the complete copies of the audited financial statements of the Seller as at fiscal year-end September 30 in each of the years 2019 and 2020 (the “Annual Financial Statements”), and unaudited interim financial statements for the three month period ending December 31, 2020 (the “Interim Financial Statements” and together with the Annual Financial Statements, the “Financial Statements”), which in each case reflect the results of operations and financial condition of Seller for such periods and at such dates. The Financial Statements have been prepared in accordance with GAAP, applied on a consistent basis throughout the period involved, subject, in the case of the Interim Financial Statements, to normal and recurring year-end adjustments (the effect of which will not have a Material Adverse Effect). The Financial Statements are based on the books and records of the Seller, fairly present in all material respects the financial condition and results of operations and the cash flows of the Seller and the Business as of the respective dates they were prepared and the results of the operations of the Seller and the Business for the periods indicated.
Section 4.05 Bankruptcy. None of the Members or the Seller have: (i) admitted in writing an inability to pay their respective debts, generally as they become due; (ii) filed or consented to the filing of a petition in bankruptcy or a petition to take advantage of an insolvency act; (iii) made an assignment for the benefit of their creditors; (iv) consented to the appointment of a receiver for themselves or for the whole or any substantial part of their property; (v) had a petition in bankruptcy filed against them; or (vi) been adjudged a bankrupt or filed a petition or answer seeking reorganization or arrangement under the federal bankruptcy laws or any law or statute of the United States of America or any other jurisdiction.
Section 4.06 Undisclosed Liabilities. Except as set forth on Schedule 4.06, Seller has no Liabilities, other than the liabilities set forth in the Interim Financial Statements and current liabilities incurred in Seller’s ordinary course of business since the date of the Interim Financial Statements.
| 11 |
Section 4.07 Absence of Certain Changes, Events and Conditions. Since the date of the Interim Financial Statements, and other than in the ordinary course of business consistent with past practice, there has not been any:
(a) occurrence or development that has had, or could reasonably be expected to have, individually or in the aggregate, a Material Adverse Effect;
(b) incurrence, assumption or guarantee of any indebtedness for borrowed money in connection with the Business except unsecured current obligations and Liabilities incurred in the ordinary course of business consistent with past practice;
(c) transfer, assignment, sale or other disposition of any of the Acquired Assets;
(d) cancellation of any debts or claims or amendment, termination or waiver of any rights constituting Acquired Assets;
(e) capital expenditures singularly or in the aggregate in excess of $25,000;
(f) imposition of any Encumbrance upon any of the Acquired Assets;
(g) any Contract to do any of the foregoing, or any action or omission that would result in any of the foregoing.
Section 4.08 Assigned Contracts. Each Assigned Contract is valid and binding on Seller in accordance with its terms and is in full force and effect. Neither the Seller nor, to Seller's Knowledge, any other party thereto is in breach of or default under (or is alleged to be in breach of or default under), or has provided or received any notice of any intention to terminate, any Assigned Contract. No event or circumstance has occurred that, with or without notice or lapse of time or both, would constitute an event of default by Seller under any Assigned Contract or result in a termination thereof or would cause or permit the acceleration or other changes of any right or obligation or the loss of benefit thereunder. There are no disputes pending or, to Seller’s Knowledge, threatened under any Assigned Contract.
Section 4.09 Title to Acquired Assets. Seller has good and valid title to all of the Acquired Assets. All such Acquired Assets are free and clear of Encumbrances except for the following (collectively referred to as “Permitted Encumbrances”):
(a) liens for Taxes not yet due and payable; and
(b) mechanics', carriers', workmen's, repairmen's or other like liens arising or incurred in the ordinary course of business consistent with past practice or amounts that are not delinquent and which are not, individually or in the aggregate, material to the Business or the Acquired Assets.
Section 4.10 Condition of Inventory. The Inventory included in the Acquired Assets is current, merchantable, saleable and usable in the ordinary course of business, consistent with past practice.
| 12 |
Section 4.11 Intellectual Property.
(a) Schedule 4.11(a) sets forth an accurate and complete list of all Intellectual Property. Schedule 4.11(a) lists the jurisdictions in which each such item of the Seller Intellectual Property has been issued or registered or in which any such application for such issuance and registration has been filed, and the name of the owner of each such registration or application. To the Seller’s Knowledge, all of the Seller’s patents are valid.
(b) The Seller owns or possesses adequate rights to use all Intellectual Property necessary to carry on the Business, consistent with past practice. The Seller has taken all commercially reasonable steps to maintain its ownership of and interest in its Intellectual Property.
(c) The Seller’s products and services, and the conduct of the Business as presently conducted does not infringe, violate or constitute an unauthorized use or misappropriation of any intellectual property right or other similar right, or any contractual right, of any Person.
(d) Each item of the Seller’s Intellectual Property that has been issued and registered in any jurisdiction by the Seller is valid and subsisting, all necessary registration, maintenance and renewal fees currently due in connection with such registered Intellectual Property have been paid and all necessary documents and certificates in connection with such registered Intellectual Property have been filed with the relevant patent, copyright, trademark or other authorities in the United States or foreign jurisdictions, as the case may be, for the purposes of maintaining such registered Intellectual Property.
(e) Except as set forth in the Intellectual Property Assignment and License Agreement, dated as of January 1, 2021, between KES Science & Technology, Inc., KES Air Technologies, LLC and Seller, no other Person has any rights to any Intellectual Property owned by the Seller.
(f) The Seller has taken all commercially reasonable steps to protect the secrecy and confidentiality of all Trade Secrets.
(g) The Seller is not, or has not been at any time, the subject of any pending or, to Seller’s Knowledge, threatened Actions which involve a claim of infringement, misappropriation, unauthorized use or violation of any Intellectual Property rights of any Person, or challenge the Seller’s ownership, use, validity or enforceability of any Intellectual Property. The Seller has not received written notice of any such threatened claim and to Seller’s Knowledge, there are no facts or circumstances that would form the basis for any such claim. To Seller’s Knowledge, all of the Seller’s rights in and to its Intellectual Property are valid and enforceable in all material respects.
(h) To Seller’s Knowledge, no Person is infringing, violating, misusing or misappropriating any of the Seller’s Intellectual Property, and no claims of such infringements, violations, misuse or misappropriations have been made against any Person by Seller.
| 13 |
(i) No present or former employee or consultant of the Seller has any right, title, or interest, directly or indirectly, in whole or in part, in any Intellectual Property. To Seller’s Knowledge, no employee, consultant or independent contractor of Seller is, as a result of or in the course of such employee, consultant or independent contractor’s engagement by the Seller, in default or breach of any material term of any employment agreement, non-disclosure agreement, assignment of invention agreement or similar agreement.
Section 4.12 Insurance. Schedule 4.12 contains a (a) a true and complete list of all current policies or binders of fire, liability, product liability, umbrella liability, real and personal property, workers' compensation, vehicular, fiduciary liability and other casualty and property insurance maintained by Seller relating to the Business or the Acquired Assets (collectively, the “Insurance Policies”); and (b) a list of all pending claims and the claims history for Seller since January 1, 2019. Seller represents and acknowledges that there are no claims related to the Business or the Acquired Assets pending under any such Insurance Policies as to which coverage has been questioned, denied or disputed or in respect of which there is an outstanding reservation of rights. Seller has not received any written notice of cancellation of, premium increase with respect to, or alteration of coverage under, any of such Insurance Policies. All premiums due on such Insurance Policies have either been paid or, if not yet due, accrued. All such Insurance Policies are in full force and effect and enforceable in accordance with their terms.
Section 4.13 Business Relationships.
(a)(i) The Seller has not received an oral or written notice of termination of any written agreements from any of its current manufacturers, distributors or customers (each a “Counterparty”), (ii) to Seller’s Knowledge, there are no complaints, claims or threats of any Counterparty against Seller which could reasonably be expected to result in a reduction of the Seller’s revenue in excess of ten percent (10%) or more; and (iii) outside of the ordinary course of business, to the Seller’s Knowledge, there are no material service issues which may curtail the relationship of a Counterparty with Seller.
(b) Schedule 4.13(b) contains a complete and accurate list of all contracts of the Seller relating to the sale of Airocide products and/or related services, including all agreements between the Seller and Kes.
(c) To Seller’s Knowledge, there is not any present condition which would prevent Purchaser from carrying on the Business after the Closing Date in the same manner as it is presently being carried on.
Section 4.14 Absence of Certain Business Practices. Neither Seller nor any Member, officer, director or manager, of Seller, nor to Seller’s Knowledge, any other Person acting on behalf of Seller, has (i) received, directly or indirectly, any material rebates, payments, commissions, promotional allowances or any other economic benefits, regardless of their nature or type, from any manufacturer, distributor, customer, governmental employee or other Person with whom the Seller has done business directly or indirectly, or (ii) directly or indirectly, given or agreed to give any gift or similar benefit to any customer, governmental employee or other person or entity who is or may be in a position to help or hinder Seller (or assist Seller in connection with any actual or proposed transaction) which, in the case of either clause (i) or clause (ii) above, would reasonably be expected to subject Seller to any Losses in any Action. Neither Seller, nor, to Seller’s Knowledge, any officer or manager thereof has used any funds for unlawful contributions, gifts, entertainment or other expenses relating to political activity or otherwise, or has made any direct or indirect unlawful payment to governmental officials or employees from the Seller’s funds or been reimbursed from the Seller’s funds for any such payment, or is aware that any other Person associated with or acting on behalf of Seller has engaged in any such activities.
| 14 |
Section 4.15 Legal Proceedings; Governmental Orders.
(a) There are no Actions pending or, to Seller's Knowledge, threatened against or by Seller or any of its Members (a) relating to or affecting the Business or the Acquired Assets; or (b) that challenge or seek to prevent, enjoin or otherwise delay the transactions contemplated by this Agreement. No event has occurred or circumstances exist that may give rise to, or serve as a basis for, any such Action.
(b) There are no outstanding Governmental Orders and no unsatisfied judgments, penalties or awards against, relating to or affecting Seller or the Business.
Section 4.16 Compliance with Laws; Permits.
(a) Since January 1, 2019, Seller has materially complied with all Laws applicable to the conduct of the Business as currently conducted or the ownership and use of the Acquired Assets.
(b) All Permits required for Seller to conduct the Business as currently conducted or for the ownership and use of the Acquired Assets have been obtained by Seller and are valid and in full force and effect. Schedule 4.16(b) contains a list of all current Permits issued to Seller which are related to the conduct of the Business as currently conducted or the ownership and use of the Acquired Assets which, if not held by the Seller would result in a Materially Adverse Effect. To Seller’s Knowledge, no event has occurred that, with or without notice or lapse of time or both, would reasonably be expected to result in the revocation, suspension, lapse or limitation of any Permit set forth in said list.
Section 4.17 Employee Benefit Matters. Seller does not maintain or contribute to and have not maintained or contributed to, any employee benefit plan, fringe benefit, stock option, equity-based compensation, phantom stock, bonus or incentive plan, severance pay policy or agreement, retirement, pension, profit sharing or deferred compensation plan or agreement, or any similar plan or agreement or any plan or arrangement providing compensation to employees or non-employee directors (an “Employee Benefit Program”). Seller has no obligations or liabilities with respect to any Employee Benefit Program.
| 15 |
Section 4.18 Employment Matters. Seller is and has been in compliance in all material respects with all applicable Laws pertaining to employment and employment practices to the extent they relate to employees of the Business, including all Laws relating to labor relations, equal employment opportunities, fair employment practices, employment discrimination, harassment, retaliation, reasonable accommodation, disability rights or benefits, immigration, wages, hours, overtime compensation, child labor, hiring, promotion and termination of employees, working conditions, meal and break periods, privacy, health and safety, workers' compensation, leaves of absence and unemployment insurance. There are no Actions concerning the employment practices of Seller pending or, to Seller’s Knowledge, threatened before any Governmental Authority that could reasonably be expected to have a Material Adverse Effect and, to Seller’s Knowledge, no basis for any such matter exists.
Section 4.19 Environmental Matters. The Seller is in material compliance with all Environmental Laws and the requirements of all Permits issued under such Environmental Laws with respect to the Seller in all material respects.
Section 4.20 Restricted Securities. The Seller acknowledges that the Shares will not be registered pursuant to the Securities Act or any applicable state securities laws, that the Shares will be characterized as "restricted securities" under federal securities laws, and that under such laws and applicable regulations the Shares cannot be sold or otherwise disposed of without registration under the Securities Act or an exemption therefrom. In this regard, the Seller, each Member and the controlling Person of such Member is familiar with Rule 144 promulgated under the Securities Act, as currently in effect, and understands the resale limitations imposed thereby and by the Securities Act.
Section 4.21 Status of Seller and Members. The Seller and each Member hereby represents and warrants that it is an "accredited investor" as that term is defined in Rule 501 of Regulation D promulgated under the Securities Act.
Section 4.22 Investment Risk. The Seller and each Member is able to bear the economic risk of directly or indirectly acquiring its Shares pursuant to the terms of this Agreement, including a complete loss of its investment in the Shares. The Seller and such Member acknowledge that Purchaser makes no representations or warranties concerning the merits of an investment in the Shares, and the Seller and such Member understands and acknowledges that Purchaser makes no representations or warranties concerning the liquidity of the Shares or the Seller’s or such Member’s ability to sell his Shares at a price he deems favorable.
Section 4.23 Restrictive Legends. The Seller acknowledges that any certificate(s) representing the Shares shall each conspicuously set forth on the face or back thereof a legend in substantially the following form:
“The securities represented hereby have not been registered with the SEC or the securities commission of any state in reliance upon an exemption from registration under the Securities Act of 1933, as amended, and, accordingly, may not be transferred unless (i) such securities have been registered for sale pursuant to the Securities Act of 1933, as amended, or (ii) such securities may be sold pursuant to Rule 144 or other applicable exemption from applicable securities laws. The Company may require an opinion of counsel to the holder of these securities, reasonably satisfactory to the Company, that such transfer may lawfully be made without registration under the Securities Act of 1933, as amended.”
| 16 |
Section 4.24 No Commissions. The Seller has not incurred any current or future obligation for any finder’s or broker’s or agent’s fees or commissions or similar compensation in connection with the transactions contemplated hereby.
Section 4.25 Taxes.
(a) All Tax Returns with respect to the Seller or the Business required to be filed by Seller for any Pre-Closing Tax Period have been, or will be, timely filed. Such Tax Returns are, or will be, true, complete and correct in all material respects. All Taxes, due and owing by Seller (whether or not shown on any Tax Return) have been, or will be, timely paid.
(b) Seller has withheld and paid each Tax required to have been withheld and paid in connection with amounts paid or owing to any employee, independent contractor, creditor, customer, shareholder or other party, and materially complied with all information reporting and backup withholding provisions of applicable Law.
(c) No extensions or waivers of statutes of limitations have been given or requested with respect to any Taxes of Seller.
(d) All deficiencies asserted, or assessments made, for Taxes against Seller as a result of any examinations by any Governmental Authority have been fully paid.
Section 4.26 FDA Compliance
All of the representations and warranties set forth on Schedule 4.26 are hereby incorporated by reference into this Section 4.26.
Section 4.27 Schedule 4.27 sets forth all of the Seller’s accounts receivables as of the Closing Date (the “Accounts Receivables”). The Seller has no reason to believe that any of the Accounts Receivable will not be paid by the applicable obligor thereof in due course. The Seller and the Members hereby jointly and severally agree to pay the Purchaser the outstanding amount of any Accounts Receivable that is not paid in full within 120 days of the Closing Date (the “Cut-off Date”). If the Purchaser does not receive payment in full for any amounts owing under this Section 4.27 within 10 business days of notice provided to the Sellers and the Members it may cancel on a pro rata basis a number of the Parent’s shares owned by the Members equal to (i) the aggregate amount of Accounts Receivable outstanding on the Cut-off Date divided by (ii) $7.50.
| 17 |
Article
V
Representations and warranties of Purchaser
Each of the Parent and the Purchaser represents and warrants to Seller that the statements contained in this Article V are true and correct as of the Closing Date.
Section 5.01 Organization of Parent. Parent is a business corporation duly organized, validly existing and in good standing under the Laws of the State of Delaware
Section 5.02 Organization of Purchaser. Purchaser is a business corporation duly organized, validly existing and in good standing under the Laws of the State of New York.
Section 5.03 Authority of Parent and Purchaser. Each of Parent and Purchaser has full power and authority to enter into this Agreement and the other Transaction Documents to which it is a party, to carry out its obligations hereunder and thereunder and to consummate the transactions contemplated hereby and thereby. The execution and delivery by Parent or the Purchaser of this Agreement and any other Transaction Document to which it is a party, the performance by Parent or Purchaser of its obligations hereunder and thereunder and the consummation by Parent or Purchaser of the transactions contemplated hereby and thereby, as applicable, have been duly authorized by all requisite corporate action on the part of Parent and/or Purchaser, as applicable. This Agreement has been duly executed and delivered by Parent and Purchaser, and (assuming due authorization, execution and delivery by Seller) this Agreement constitutes a legal, valid and binding obligation of Parent and Purchaser enforceable against Parent and Purchaser in accordance with its terms. When each other Transaction Document to which Parent or Purchaser is or will be a party has been duly executed and delivered by Parent or Purchaser (assuming due authorization, execution and delivery by each other party thereto), as applicable, such Transaction Document will constitute a legal and binding obligation of Parent or Purchaser, as applicable, enforceable against it in accordance with its terms.
Section 5.04 No Conflicts; Consents. The execution, delivery and performance by Parent or the Purchaser of this Agreement and the other Transaction Documents to which it is a party, and the consummation of the transactions contemplated hereby and thereby, do not and will not: (a) conflict with or result in a violation or breach of, or default under, any provision of the certificate of incorporation, by-laws or other organizational documents of Parent or Purchaser; (b) conflict with or result in a violation or breach of any provision of any Law or Governmental Order applicable to Parent or Purchaser; or (c) require the consent, notice or other action by any Person under any Contract to which Parent or Purchaser is a party. No consent, approval, Permit, Governmental Order, declaration or filing with, or notice to, any Governmental Authority is required by or with respect to Parent or Purchaser in connection with the execution and delivery of this Agreement and the other Transaction Documents to which it is a party and the consummation of the transactions contemplated hereby and thereby.
Section 5.05 Legal Proceedings. There are no Actions pending or, to Purchaser's or Parent’s Knowledge, threatened against or by Purchaser or any Affiliate of Purchaser that challenge or seek to prevent, enjoin or otherwise delay the transactions contemplated by this Agreement. No event has occurred or circumstances exist that may give rise or serve as a basis for any such Action.
| 18 |
Section 5.06 Shares. The Shares to be issued to the Seller pursuant to this Agreement will be duly authorized and reserved for issuance and when issued in accordance with this Agreement, will be validly issued and outstanding, with good and marketable title to such Shares (subject to the legend referenced in Section 4.23), fully paid and non-assessable and vest in the holder thereof free and clear of any restrictions on transfer and voting rights (other than any restrictions under applicable state or federal securities laws), Taxes, Encumbrances, options, warrants, purchase rights, Contracts, commitments, equities, claims, and demands and will not be subject to any pre-emptive or other similar rights.
Section 5.07 No Commissions. Neither the Parent nor the Purchaser have incurred any current or future obligation for any finder’s or broker’s or agent’s fees or commissions or similar compensation in connection with the transactions contemplated hereby.
Section 5.08 Access and Investigation; No Other Representations and Warranties; Non-Reliance. Purchaser has conducted, to its satisfaction, its own independent investigation of the Acquired Assets, the Business, Liabilities, operations and prospects (financial or otherwise) of the Seller. Purchaser acknowledges that neither the Seller nor any Member has made or is making any representations or warranties, express or implied, regarding the Seller or any Member, their respective assets, businesses, liabilities, operations and prospects (financial or otherwise) or the subject matter of this Agreement and the Transaction Documents, except as contained in Article IV (as modified by the Schedules to this Agreement), the Transaction Documents and the certificates executed and delivered by the Seller and the Members at Closing, all of which representations and warranties are only contractual in nature.
Article
VI
Covenants
Section 6.01 Post-Closing Covenants.
(a) The Seller and the Members hereby agree to enter into lock-up agreements with respect to the Shares to the extent required by an underwriter or placement agent engaged by the Parent in connection with a future financing as long as the Parent’s officers, directors and shareholders holding 5% or more of the issued and outstanding shares of the Parent are required to enter lock-ups. Any lock-up agreements entered into by the Seller and the Members will be on substantially the same terms as those lock-up agreements entered into by the Parent’s officers, directors and other shareholders. Anything in this Section 6.01(a) to the contrary notwithstanding, no Member will be required to enter into any such lock-up agreement, unless he, she or it holds more than 5% of the issued and outstanding shares of the Parent; provided, however, that the Parent may request that any Member holding less than 5% of the issued and outstanding shares of the Parent or any member of such Member, and such Member or, as applicable, such member of such Member will consider, entering into lock-up agreements with respect to the Shares; provided, further, that no consideration will be due from the Parent with respect to any such lock-up agreements with respect to the Shares. The Seller, the Members and each member of the Members hereby agree not to transfer any of the Shares for the sole purpose of avoiding entering into any such lock-up agreement.
| 19 |
(b) The Seller and each Member will not, and each will cause their members and other Affiliates to whom any portion of the Shares are transferred (each, a “Selling Shareholder”) not to (i) until the date which is six months from the Closing Date, transfer the Shares to any Person, including Affiliates who are not the Seller or a Member and (ii) until the first anniversary of the Closing Date, transfer Shares without providing notice to the Seller within one business day of such transfer.
(c) Each Selling Shareholder agrees or will be caused by the Seller or the applicable Member to agree that until the date that is eighteen months from the Closing Date, the Parent has the right of first refusal to purchase the number of Shares that will be sold in aggregate by the Selling Shareholders in excess of 15,000 during a 5 consecutive trading day period (“ROFR Shares”), provided, however, any Shares transferred by the Seller or applicable Member to any of their respective Affiliates, after the date which is six months after Closing Date, will be excluded from the ROFR Shares. The applicable Selling Shareholder will provide written notice to the Parent of its intention to sell ROFR Shares and the Parent shall have the right to purchase the ROFR Shares from such Selling Shareholder for a period of one trading day after such notice is received at a price equal to the average of the closing prices of the Parent’s common stock as stated by Nasdaq during the three trading days immediately prior to the date the right of first refusal notice is received by the Purchaser less a discount of five percent (5%).
(d) After the Closing Date, the Parent agrees that David Kight, a manager of the Seller, may observe meetings of the Parent’s board of directors at the invitation of the chairman.
(e) The Parent will use high ethical and moral standards in the operation of its business. In the event the Parent elects to execute a debt financing transaction, the Parent will provide Fakhruddin Holdings FZC written notice of such financing transaction no less than 7 calendar days prior to closing of such financing transaction; provided that such written notice to Fakhruddin Holdings FZC will be provided via e-mail to yhamid@fakhruddin.ae.
(f) The Seller shall use reasonable commercial efforts to assign the Seller’s accounts with Shopify Merchant Services and Intuit Merchant Services over to the Purchaser promptly after the Closing.
Section 6.02 Non-solicitation. Until the second anniversary of the Closing Date, Parent shall not, and shall not permit any of its Affiliates to, directly or indirectly, hire or solicit any person for employment any employee of the Seller (other than Keith Frein and David Ghelerter), except pursuant to a general solicitation which is not directed specifically to any such employees; without first obtaining the written consent of the Seller.
| 20 |
Section 6.03 Employment and Consulting Agreements. Upon the Closing, (i) Keith Frein shall be employed by the Purchaser pursuant to an employment agreement in substantially the form of Exhibit E hereto (the “Employment Agreement”) and (ii) David Kight shall be engaged by the Purchaser as consultant pursuant to a consulting agreement substantially in the form of Exhibit F hereto (the “Consulting Agreement”).
Section 6.04 [Intentionally Omitted].
Section 6.05 Approvals and Consents. Seller, Parent and Purchaser shall use commercially reasonable efforts to give all notices to, and obtain all consents from, any third parties to whom notice to, or a consent from, is required by Law or Contract.
Section 6.06 Books and Records.
(a) In order to facilitate the resolution of any claims made against or incurred by Seller prior to the Closing, or for any other reasonable purpose, for a period of five years after the Closing, Purchaser shall:
(i) allow Seller to keep copies of Books and Records; and
(ii) retain the Books and Records (but only a copy of payroll and personnel files) relating to periods prior to the Closing in a manner reasonably consistent with the prior practices of Seller; and
(iii) upon reasonable notice, afford the Seller's Representatives reasonable access (including the right to make, at Seller's expense, photocopies), during normal business hours, to such Books and Records.
(b) In order to facilitate the resolution of any claims made by or against or incurred by Purchaser after the Closing, or for any other reasonable purpose, for a period of five years following the Closing, Seller shall:
(i) retain any Books and Records (including personnel files) of Seller which relate to the Business and its operations for periods prior to the Closing which have not been provided to the Purchaser; and
(ii) upon reasonable notice, afford the Purchaser's Representatives reasonable access (including the right to make, at Purchaser's expense, photocopies), during normal business hours, to such books and records.
(c) Neither Purchaser nor Seller shall be obligated to provide the other party with access to any books or records (including personnel files) pursuant to this Section 6.06 where such access would violate any Law.
Section 6.07 Public Announcements. Unless otherwise required by applicable Law (based upon the reasonable advice of counsel), neither Party, nor any of their Affiliates, shall make any public announcements in respect of this Agreement or the transactions contemplated hereby or otherwise communicate with any news media without the prior written consent of the other Party, and the Parties shall cooperate as to the timing and contents of any announcement made in respect of this Agreement of the transactions contemplated hereby.
| 21 |
Section 6.08 Receivables. From and after the Closing, if Seller or any of its Affiliates receives or collects any funds relating to any Acquired Asset, Seller shall remit such funds to Purchaser within five Business Days after its receipt thereof. From and after the Closing, if Purchaser or its Affiliate receives or collects any funds relating to any Excluded Asset, Purchaser or its Affiliate shall remit any such funds to Seller within five Business Days after its receipt thereof.
Section 6.09 Transfer Taxes. All transfer, documentary, sales, use, stamp, registration, value added and other such Taxes and fees (including any penalties and interest) incurred in connection with this Agreement and the other Transaction Documents (including any real property transfer Tax and any other similar Tax) shall be borne and paid by Seller and/or its Members when due. Seller and its Members shall, at its own expense, timely file any Tax Return or other document with respect to such Taxes or fees (and Purchaser shall cooperate with respect thereto as necessary).
Section 6.10 Further Assurances. Following the Closing and at the Purchaser’s sole cost and expense, Seller shall, and shall cause its Affiliates to, execute and deliver such additional documents, instruments, conveyances and assurances and take such further actions as may be reasonably requested by Purchaser to carry out the provisions hereof and give effect to the transactions contemplated by this Agreement and the other Transaction Documents.
Article
VII
Indemnification
Section 7.01 Survival. Subject to the limitations and other provisions of this Agreement, the representations and warranties contained in Article IV shall survive the Closing and will expire on and be of no further force or effect on and after six (6) months following the Closing Date; provided, however, that (i) the Fundamental Reps and the representations and warranties in Section 5.01, Section 5.02, Section 5.03 and Section 5.07 shall survive for the full period of all applicable statutes of limitations (giving effect to any waiver, mitigation or extension thereof) plus 60 days and (ii) the Hybrid Rep shall survive the Closing and will expire on and be of no further force or effect on and after eighteen (18) months following the Closing Date. All covenants and agreements of the parties contained herein shall survive the Closing indefinitely or for the period explicitly specified therein. Notwithstanding the foregoing, any claims asserted in good faith with reasonable specificity (to the extent known at such time) and in writing by notice from the non-breaching party to the breaching party prior to the expiration date of the applicable survival period shall not thereafter be barred by the expiration of the relevant representation or warranty and such claims shall survive until finally resolved.
| 22 |
Section 7.02 Indemnification by Seller and the Members.
(a) Subject to the other terms and conditions of this Article VII, including Section 7.07, Seller and the Members (severally in their applicable pro rata ownership percentage of Seller, but not jointly) shall indemnify and defend each of Purchaser and its Affiliates and their respective Representatives (collectively, the “Purchaser Indemnitees”) against, and shall hold each of them harmless from and against, and shall pay and reimburse each of them for, any and all Losses, up to an amount equal to 15% of the Purchase Price Value, incurred or sustained by, or imposed upon, the Purchaser Indemnitees based upon, arising out of, with respect to or by reason of:
(i) any inaccuracy in or breach of any of the representations or warranties of Seller contained in this Agreement, the other Transaction Documents or in any certificate or instrument delivered by or on behalf of Seller pursuant to this Agreement, as of the date such representation or warranty was made or as if such representation or warranty was made on and as of the Closing Date (except for representations and warranties that expressly relate to a specified date, the inaccuracy in or breach of which will be determined with reference to such specified date);
(ii) any breach or non-fulfillment of any covenant, agreement or obligation to be performed by Seller pursuant to this Agreement, the other Transaction Documents or any certificate or instrument delivered by or on behalf of Seller pursuant to this Agreement;
(iii) any Excluded Asset or any Liability of the Seller (including any Liabilities related to products sold by the Seller prior to the Closing) not expressly assumed by the Purchaser pursuant to this Agreement or any Transaction Document; or
(iv) any Third-Party Claim based upon, resulting from or arising out of the business, operations, properties, assets or obligations of Seller or any of its Affiliates (including the Business and the Acquired Assets) conducted, existing or arising on or prior to the Closing Date.
(b) Anything in this Agreement to the contrary notwithstanding, the limitation on indemnification contained in Section 7.02(a) with respect to (i) the inaccuracy in or breach of (or any facts or circumstances constituting any such inaccuracy or breach) the Fundamental Reps or any breach or non-fulfillment of the Seller’s covenants contained in Article VI shall be capped at the Purchase Price Value and (ii) the inaccuracy in or breach of (or any facts or circumstances constituting any such inaccuracy or breach) the Hybrid Rep shall be capped at 30% of the Purchase Price Value. Anything in this Agreement to the contrary notwithstanding, the indemnification provided by the Seller and the Members for the inaccuracy in or breach of (or any facts or circumstances constituting any such inaccuracy or breach) of the Fundamental Reps or a breach of the Seller’s covenants contained in Article VI shall be joint and several.
(c) From and after the Closing, the Purchaser Indemnitees will not have the right to be indemnified pursuant to the provisions contained in Section 7.02(a) unless and until the Purchaser Indemnitees (or any member thereof) have incurred on a cumulative basis aggregate Losses in an amount exceeding $100,000 (the “Basket Amount”), whereupon the Purchaser Indemnitees (or any member thereof) will be entitled to indemnification for all Losses incurred by the Purchaser Indemnitees (or any member thereof) in excess of the Basket Amount; provided, however, that the Basket Amount with respect to the inaccuracy or breach of (or any facts or circumstances constituting any such inaccuracy or breach) of the Fundamental Reps, the Hybrid Rep or a breach of the Seller’s covenants contained in Article VI shall be $50,000.
| 23 |
Section 7.03 Parent and Purchaser Indemnification. Subject to the other terms and conditions of this Article VII, including Section 7.07, the Parent and the Purchaser shall indemnify and defend each of Seller and its Affiliates and their respective Representatives (collectively, the “Seller Indemnitees”) against, and shall hold each of them harmless from and against, and shall pay and reimburse each of them for, any and all Losses, up to an amount equal to 15% of the Purchase Price Value, incurred or sustained by, or imposed upon, the Seller Indemnitees based upon, arising out of, with respect to or by reason of:
(a) any breach by the Parent or the Purchaser of its representations and warranties set forth in Section 5.06;
(b) any breach or non-fulfillment of any covenant, agreement or obligation to be performed by Parent or Purchaser pursuant to this Agreement, the other Transaction Documents or any certificate or instrument delivered by or on behalf of Seller pursuant to this Agreement;
(c) any Liability of the Seller expressly assumed by the Purchaser pursuant to this Agreement or any Transaction Document; or
(d) any Third-Party Claim based upon, resulting from or arising out of the business, operations, properties, assets or obligations of Purchaser or any of its Affiliates (including the Business and the Acquired Assets) after the Closing Date.
Section 7.04 Indemnification Procedures. The party making a claim under this Article VII is referred to as the “Indemnified Party”, and the party against whom such claims are asserted under this Article VII is referred to as the “Indemnifying Party”.
(a) Third-Party Claims. If any Indemnified Party receives notice of the assertion or commencement of any Action made or brought by any Person who is not a party to this Agreement or an Affiliate of a party to this Agreement or a Representative of the foregoing (a “Third-Party Claim”) against such Indemnified Party with respect to which the Indemnifying Party is obligated to provide indemnification under this Agreement, the Indemnified Party shall give the Indemnifying Party reasonably prompt written notice thereof, but in any event not later than 10 calendar days after receipt of such notice of such Third-Party Claim. The failure to give such prompt written notice shall not, however, relieve the Indemnifying Party of its indemnification obligations, except and only to the extent that the Indemnifying Party forfeits rights or defenses by reason of such failure. Such notice by the Indemnified Party shall describe the Third-Party Claim in reasonable detail, shall include copies of all material written evidence thereof and shall indicate the estimated amount, if reasonably practicable, of the Loss that has been or may be sustained by the Indemnified Party. The Indemnifying Party shall have the right to participate in, or by giving written notice to the Indemnified Party, to assume the defense of any Third-Party Claim at the Indemnifying Party's expense and by the Indemnifying Party's own counsel, and the Indemnified Party shall cooperate in good faith in such defense. In the event that the Indemnifying Party assumes the defense of any Third-Party Claim, subject to Section 7.04(b), it shall have the right to take such action as it deems necessary to avoid, dispute, defend, appeal or make counterclaims pertaining to any such Third-Party Claim in the name and on behalf of the Indemnified Party. The Indemnified Party shall have the right to participate in the defense of any Third-Party Claim with counsel selected by it subject to the Indemnifying Party's right to control the defense thereof. The fees and disbursements of such counsel shall be at the expense of the Indemnified Party, provided, that if in the reasonable opinion of counsel to the Indemnified Party, (A) there are legal defenses available to an Indemnified Party that are materially different from or additional to those available to the Indemnifying Party; or (B) there exists a legal conflict of interest between the Indemnifying Party and the Indemnified Party that cannot be waived, the Indemnifying Party shall be liable for the reasonable fees and expenses of counsel to the Indemnified Party. If the Indemnifying Party elects not to compromise or defend such Third-Party Claim, fails to promptly notify the Indemnified Party in writing of its election to defend as provided in this Agreement, or fails to diligently prosecute the defense of such Third-Party Claim, the Indemnified Party may, subject to Section 7.04(b), pay, compromise, defend such Third-Party Claim and seek indemnification for any and all Losses based upon, arising from or relating to such Third-Party Claim. Seller and Purchaser shall cooperate with each other in all reasonable respects in connection with the defense of any Third-Party Claim, including making available records relating to such Third-Party Claim and furnishing, without expense (other than reimbursement of actual out-of-pocket expenses) to the defending party, management employees of the non-defending party as may be reasonably necessary for the preparation of the defense of such Third-Party Claim.
| 24 |
(b) Settlement of Third-Party Claims. Notwithstanding any other provision of this Agreement, the Indemnifying Party shall not enter into settlement of any Third-Party Claim without the prior written consent of the Indemnified Party (which consent will not be unreasonably conditioned, delayed or withheld), provided, however, that no prior written consent of the Indemnified party will be required if such settlement (A) fully releases the Indemnified Party from further liability or other consequences and (B) involves only the payment of monetary damages for which the Indemnified Party is at the time entitled to indemnification for the full amount of Losses and (C) the Indemnifying Party provides the Indemnified Party with evidence of the Indemnifying Party’s ability to pay such monetary damages.
(c) Direct Claims. Any Action by an Indemnified Party on account of a Loss which does not result from a Third-Party Claim (a “Direct Claim”) shall be asserted by the Indemnified Party giving the Indemnifying Party reasonably prompt written notice thereof, but in any event not later than 30 days after the Indemnified Party becomes aware of such Direct Claim. The failure to give such prompt written notice shall not, however, relieve the Indemnifying Party of its indemnification obligations, except and only to the extent that the Indemnifying Party forfeits rights or defenses by reason of such failure. Such notice by the Indemnified Party shall describe the Direct Claim in reasonable detail, shall include copies of all material written evidence thereof and shall indicate the amount of the Loss that has been sustained by the Indemnified Party. The Indemnifying Party shall have 30 days after its receipt of such notice to respond in writing to such Direct Claim. The Indemnified Party shall allow the Indemnifying Party and its Representatives to investigate the matter or circumstance alleged to give rise to the Direct Claim, and whether and to what extent any amount is payable in respect of the Direct Claim and the Indemnified Party shall assist the Indemnifying Party's investigation by giving such information and assistance (including access to the Indemnified Party's premises and personnel and the right to examine and copy any accounts, documents or records) as the Indemnifying Party or any of its Representatives may reasonably request. If the Indemnifying Party does not so respond within such 30-day period, the Indemnifying Party shall be deemed to have rejected such claim, in which case the Indemnified Party shall be free to pursue such remedies as may be available to the Indemnified Party on the terms and subject to the provisions of this Agreement.
| 25 |
Section 7.05 Payments. Once a Loss is agreed to by the Indemnifying Party or finally adjudicated to be payable pursuant to this Article VII, the Indemnifying Party shall satisfy its obligations within 15 Business Days of such final, non-appealable adjudication by wire transfer of immediately available funds. The parties hereto agree that should an Indemnifying Party not make full payment of any such obligations within such 15 Business Day period, any amount payable shall accrue interest from and including the date of agreement of the Indemnifying Party or final, non-appealable adjudication to the date such payment has been made at a rate per annum equal to eight percent (8%). Such interest shall be calculated daily on the basis of a 365-day year and the actual number of days elapsed.
Section 7.06 Tax Treatment of Indemnification Payments. All indemnification payments made under this Agreement shall be treated by the parties as an adjustment to the Purchase Price for Tax purposes, unless otherwise required by Law.
Section 7.07 Exclusive Remedies. The parties acknowledge and agree that their sole and exclusive remedy with respect to any and all claims (other than claims arising from fraud, criminal activity or willful misconduct on the part of a party hereto in connection with the transactions contemplated by this Agreement) for which indemnification is available pursuant to this Article VII, shall be pursuant to the indemnification provisions set forth in this Article VII. In furtherance of the foregoing, each party hereby waives, to the fullest extent permitted under Law, any and all rights, claims and causes of action for covered by this Article VII. Nothing in this Article VII shall limit any Person's right to seek and obtain any amount of indemnification or equitable relief or to seek any other remedy on account of any party's fraudulent, criminal or intentional misconduct.
Article
VIII
Miscellaneous
Section 8.01 Expenses. Except as otherwise expressly provided herein, all costs and expenses, including fees and disbursements of counsel, financial advisors and accountants, incurred in connection with this Agreement and the transactions contemplated hereby shall be paid by the party incurring such costs and expenses, whether or not the Closing shall have occurred.
Section 8.02 Notices. All notices, requests, consents, claims, demands, waivers and other communications hereunder shall be in writing and shall be deemed to have been given (a) when delivered by hand (with written confirmation of receipt); (b) when received by the addressee if sent by a nationally recognized overnight courier (receipt requested); (c) on the date sent by facsimile or e-mail of a PDF document (with confirmation of transmission) if sent during normal business hours of the recipient, and on the next Business Day if sent after normal business hours of the recipient or (d) on the third day after the date mailed, by certified or registered mail, return receipt requested, postage prepaid. Such communications must be sent to the respective parties at the following addresses (or at such other address for a party as shall be specified in a notice given in accordance with this Section 8.02):
| 26 |
| If to Seller or any Member: | Akida Holdings LLC 2300
Marshpoint Road, Suite 202, Neptune Beach, FL 32266 |
If to Purchaser: |
SteriLumen, Inc. 8480 East Orchard Road, Suite 2400, Greenwood Village, Colorado 80111 E-mail: q@sterilumen.com; m.munn@sterlilumen.com Attn: Keyoumars Saeed and Max Munn |
Section 8.03 Headings. The headings in this Agreement are for reference only and shall not affect the interpretation of this Agreement.
Section 8.04 Severability. If any term or provision of this Agreement is invalid, illegal or unenforceable in any jurisdiction, such invalidity, illegality or unenforceability shall not affect any other term or provision of this Agreement or invalidate or render unenforceable such term or provision in any other jurisdiction. Upon such determination that any term or other provision is invalid, illegal or unenforceable, the parties hereto shall negotiate in good faith to modify this Agreement so as to effect the original intent of the parties as closely as possible in a mutually acceptable manner in order that the transactions contemplated hereby be consummated as originally contemplated to the greatest extent possible.
Section 8.05 Entire Agreement. This Agreement and the other Transaction Documents constitute the sole and entire agreement of the parties to this Agreement with respect to the subject matter contained herein and therein, and supersede all prior and contemporaneous understandings and agreements, both written and oral, with respect to such subject matter.
Section 8.06 Successors and Assigns. This Agreement shall be binding upon and shall inure to the benefit of the parties hereto and their respective successors and permitted assigns. Neither party may assign its rights or obligations hereunder without the prior written consent of the other party. No assignment shall relieve the assigning party of any of its obligations hereunder.
| 27 |
Section 8.07 No Third-party Beneficiaries. Except as provided in Article III, this Agreement is for the sole benefit of the parties hereto and their respective successors and permitted assigns and nothing herein, express or implied, is intended to or shall confer upon any other Person or entity any legal or equitable right, benefit or remedy of any nature whatsoever under or by reason of this Agreement.
Section 8.08 Amendment and Modification; Waiver. This Agreement may only be amended, modified or supplemented by an agreement in writing signed by each party hereto. No waiver by any party of any of the provisions hereof shall be effective unless explicitly set forth in writing and signed by the party so waiving. No failure to exercise, or delay in exercising, any right, remedy, power or privilege arising from this Agreement shall operate or be construed as a waiver thereof; nor shall any single or partial exercise of any right, remedy, power or privilege hereunder preclude any other or further exercise thereof or the exercise of any other right, remedy, power or privilege.
Section 8.09 Governing Law; Jurisdiction and Venue. This Agreement shall be governed by and construed in accordance with the internal laws of the State of Delaware without giving effect to any choice or conflict of law provision or rule (whether of the State of Delaware or any other jurisdiction). The parties hereby consent to the concurrent exclusive jurisdiction of the courts of the State of Delaware and the United States courts located in New Castle County, Delaware in connection with any suit, action or proceeding arising out of or relating in any manner to this Agreement, and each of the Parties further irrevocably agrees to waive any objection to the venue of any such suit or proceeding in either court, or to in personam jurisdiction providing that service is effective.
Section 8.10 Counterparts. This Agreement may be executed in counterparts, each of which shall be deemed an original, but all of which together shall be deemed to be one and the same agreement. A signed copy of this Agreement delivered by facsimile, e-mail or other means of electronic transmission shall be deemed to have the same legal effect as delivery of an original signed copy of this Agreement.
[SIGNATURE PAGE FOLLOWS]
| 28 |
IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be executed as of the date first written above by their respective officers thereunto duly authorized.
| SELLER: | PURCHASER AND PARENT: | |
| AKIDA HOLDINGS LLC | STERILUMEN, INC. | |
| By: | By: | |
| Name: David Knight | Name: Keyoumars Saeed | |
| Title: Manager | Title: Chief Executive Officer | |
| MEMBERS: | APPLIED UV, INC. | |
| SIMBA PARTNERS, LLC | By: | |
| Name: Keyoumars Saeed | ||
| By: | Title: Chief Executive Officer | |
| Name: David Kight | ||
| Title: Manager | ||
| JJH HOLDINGS, LLC | ||
| By: | ||
| Name: John J. Hayman, III | ||
| Title: Managing Member | ||
| FAKHRUDDIN HOLDINGS FZC | ||
| By: | ||
| Name: Yusuf Fakhruddin | ||
| Title: Manager |
[SIGNATURE PAGE CONTINUES ON NEXT PAGE]
| 29 |
As an inducement for the Purchaser to enter into this Agreement, the following individuals, each of whom is an equity holder of a Member, hereby agrees that he will be liable for the obligations of the applicable Member for which he is an equity holder pursuant to Article VII and agrees to be bound by Section 6.01(a):
________________________________
John J. Hayman, Jr., with respect to the
obligations of JJH Holdings, LLC
________________________________
John J. Hayman, III, with respect to the
obligations of JJH Holdings, LLC
________________________________
Scott Hayman, with respect to the
obligations of JJH Holdings, LLC
________________________________
David Heffner, with respect to the
obligations of JJH Holdings, LLC
________________________________
David E. Kight, with respect to the
obligations of Simba Partners, LLC
________________________________
Barney Freedman, with respect to the
obligations of Simba Partners, LLC
________________________________
Yusuf Fakhruddin, with respect to the
obligations of Fakhruddin Holdings FCZ
| 30 |
EXHIBIT A
List of Acquired Assets
Exhibit A – Acquired Assets
Furniture & Fixtures
None
Production Tooling
As Detailed Below
Accounts Receivable
As Detailed Below
Inventory
As set forth on Schedule 1
Factory and Vendor Deposits with Season as a2b Fulfillment
As set forth on Schedule 1
Cash on Hand
As set forth on Schedule 1
Intellectual Property
As detailed on Schedule 4.11(a)
| 31 |
| EXHIBIT A | ||||||||||||||||||||||||
| Akida Holdings LLC | ||||||||||||||||||||||||
| A/R Aging Summary | ||||||||||||||||||||||||
| As of February 4, 2021 | ||||||||||||||||||||||||
| Current | 1-30 | 31-60 | 61-90 | >90 | Total | |||||||||||||||||||
| Interactive Group | 0.00 | 99.00 | 0.00 | 0.00 | 0.00 | 99.00 | ||||||||||||||||||
| Air Purifier Systems | 0.00 | 99.00 | 0.00 | 0.00 | 0.00 | 99.00 | ||||||||||||||||||
| Shawano County Finance Department | 0.00 | 246.19 | 0.00 | 0.00 | 0.00 | 246.19 | ||||||||||||||||||
| Rohi Green International Ltd. | 0.00 | 0.00 | 0.00 | 0.00 | 319.54 | 319.54 | ||||||||||||||||||
| Children's Hospital Boston | 0.00 | 0.00 | 426.85 | 0.00 | 0.00 | 426.85 | ||||||||||||||||||
| Medifacts | 0.00 | 787.48 | 0.00 | 0.00 | 0.00 | 787.48 | ||||||||||||||||||
| Macy's | 0.00 | 0.00 | 0.00 | 0.00 | 872.17 | 872.17 | ||||||||||||||||||
| Novatek Pakistan | 0.00 | 98.50 | 0.00 | 0.00 | 847.00 | 945.50 | ||||||||||||||||||
| America Nanocoat | 0.00 | 0.00 | 0.00 | 1010.46 | 0.00 | 1010.46 | ||||||||||||||||||
| AMI Services | 0.00 | 1188.00 | 0.00 | 0.00 | 0.00 | 1188.00 | ||||||||||||||||||
| North Bay Air Purification, LLC | 0.00 | 4671.18 | 0.00 | 0.00 | 0.00 | 4671.18 | ||||||||||||||||||
| SteriLumen | 0.00 | 7448.38 | 690.04 | 0.00 | 0.00 | 8138.42 | ||||||||||||||||||
| Batta Technologies, LLC | 39710.49 | 29630.49 | 0.00 | 0.00 | 945.29 | 70286.27 | ||||||||||||||||||
| Airocide Nordic Ltd. | 0.00 | 144151.13 | 0.00 | 0.00 | 0.00 | 144151.13 | ||||||||||||||||||
| TOTAL | 39710.49 | 188419.35 | 1116.89 | 1010.46 | 2984.00 | 233241.19 | ||||||||||||||||||
| 32 |
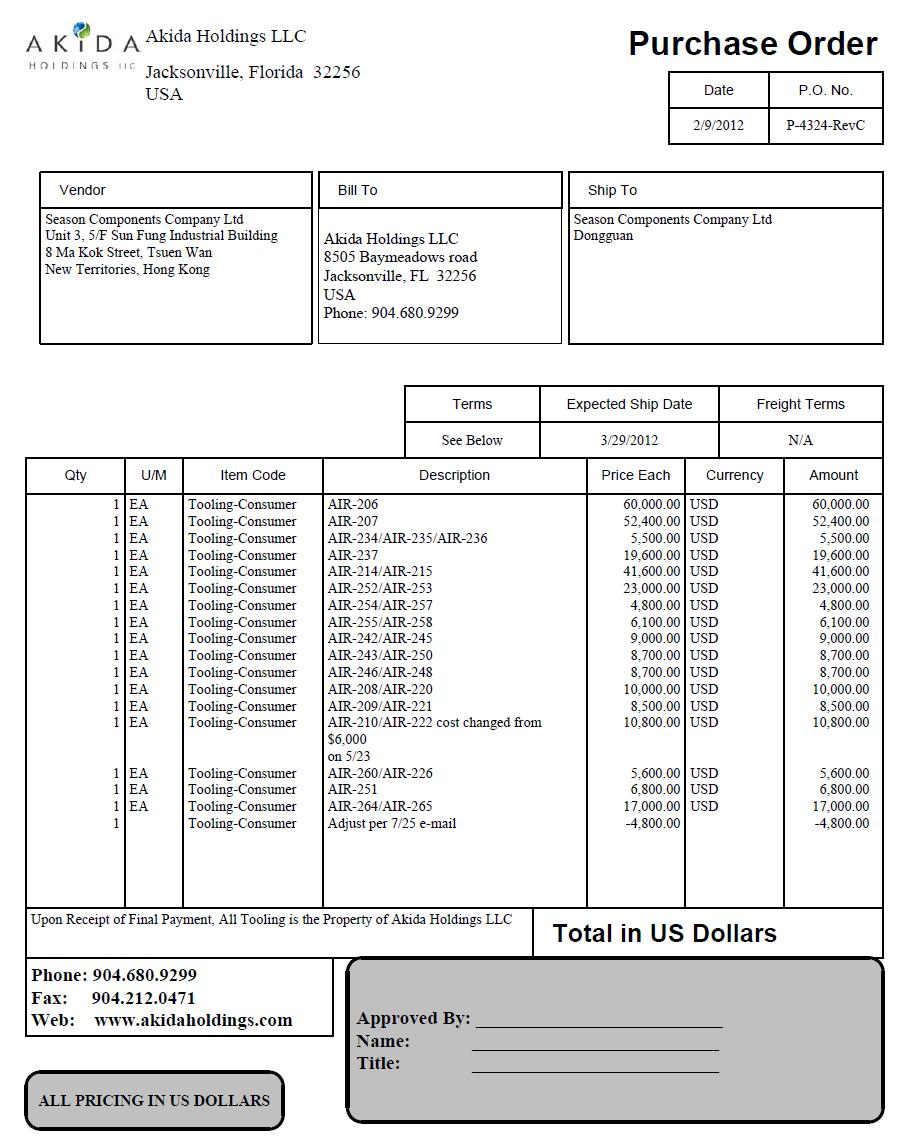
| 33 |

| 34 |
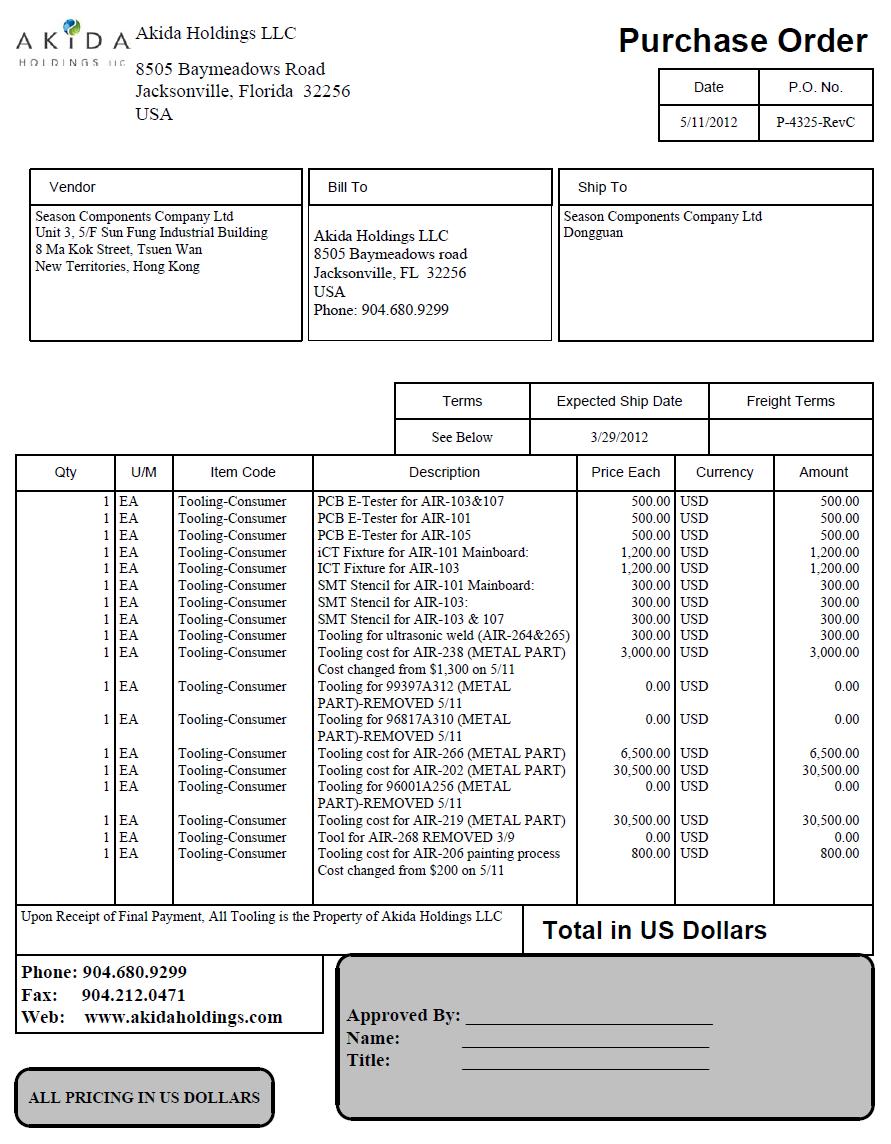
| 35 |

| 36 |
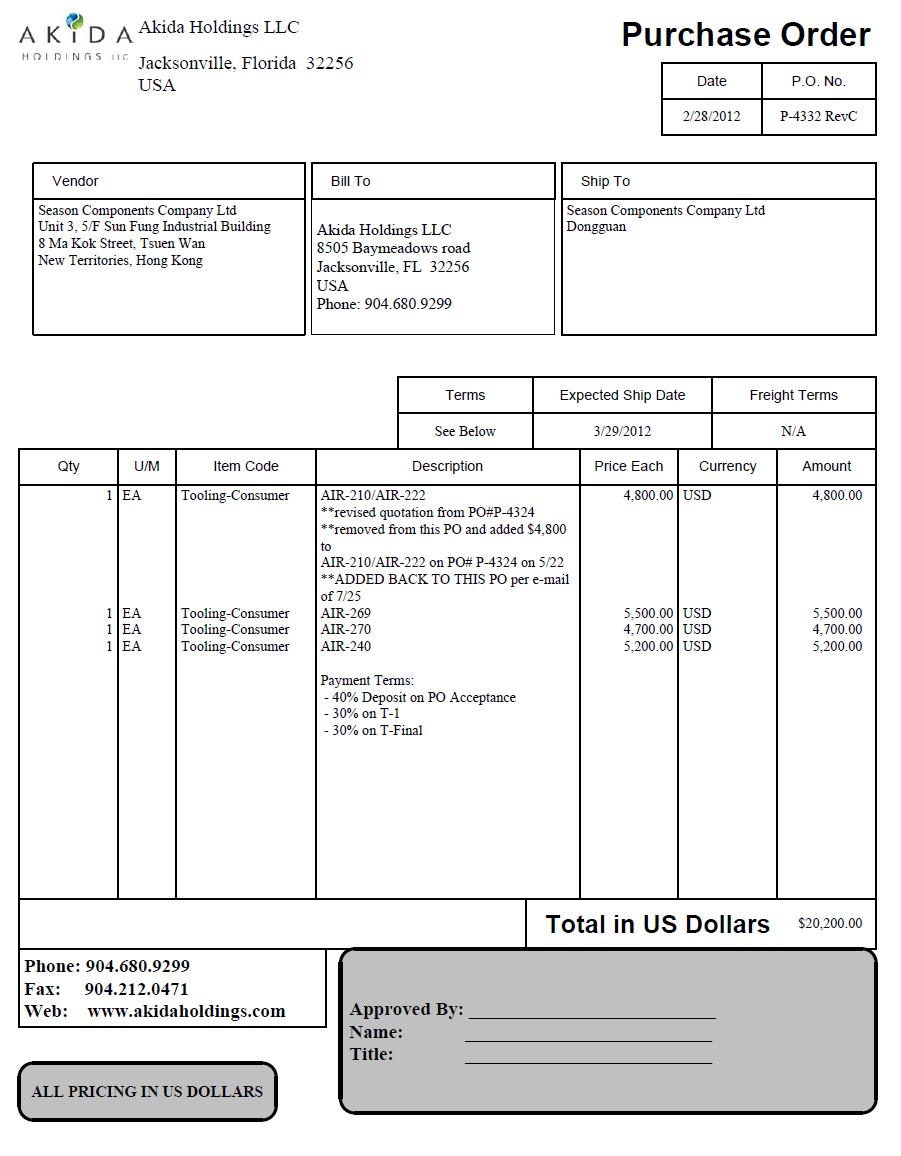
| 37 |

| 38 |
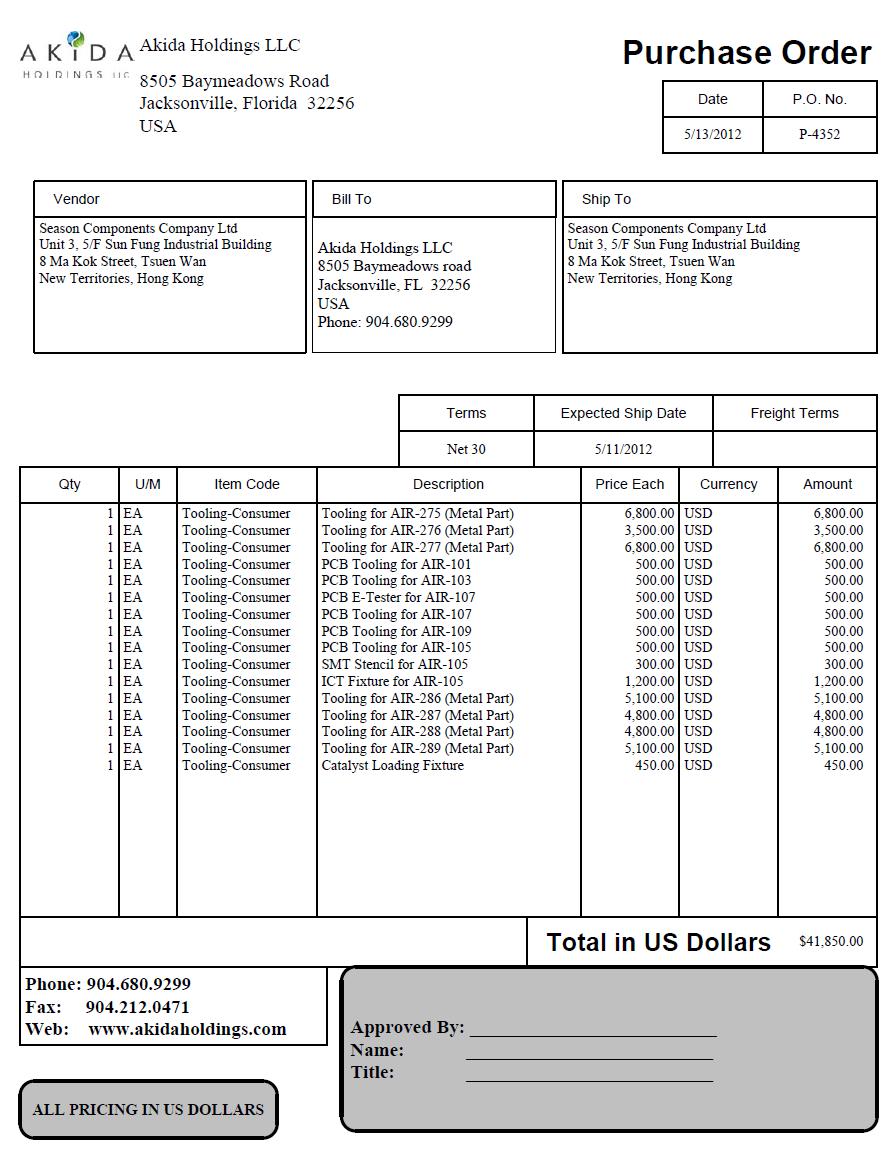
| 39 |

| 40 |
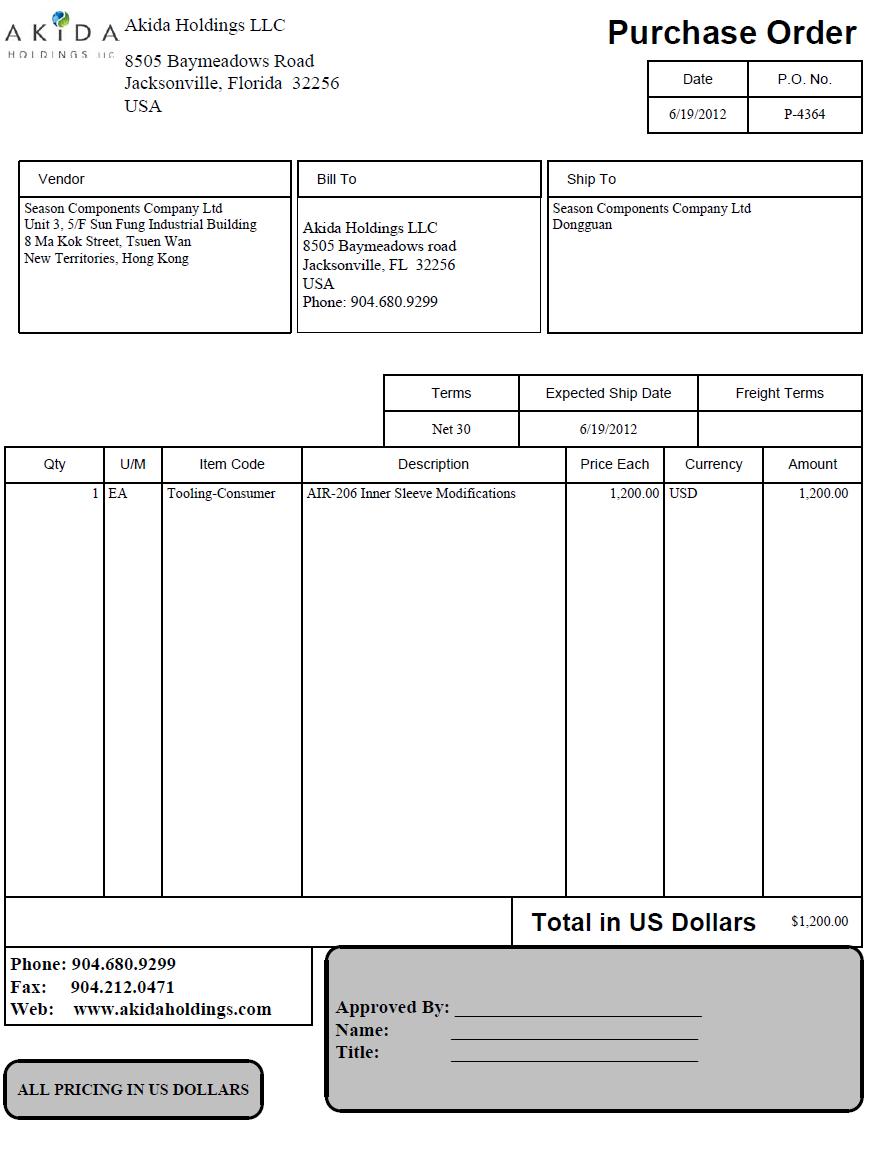
| 41 |
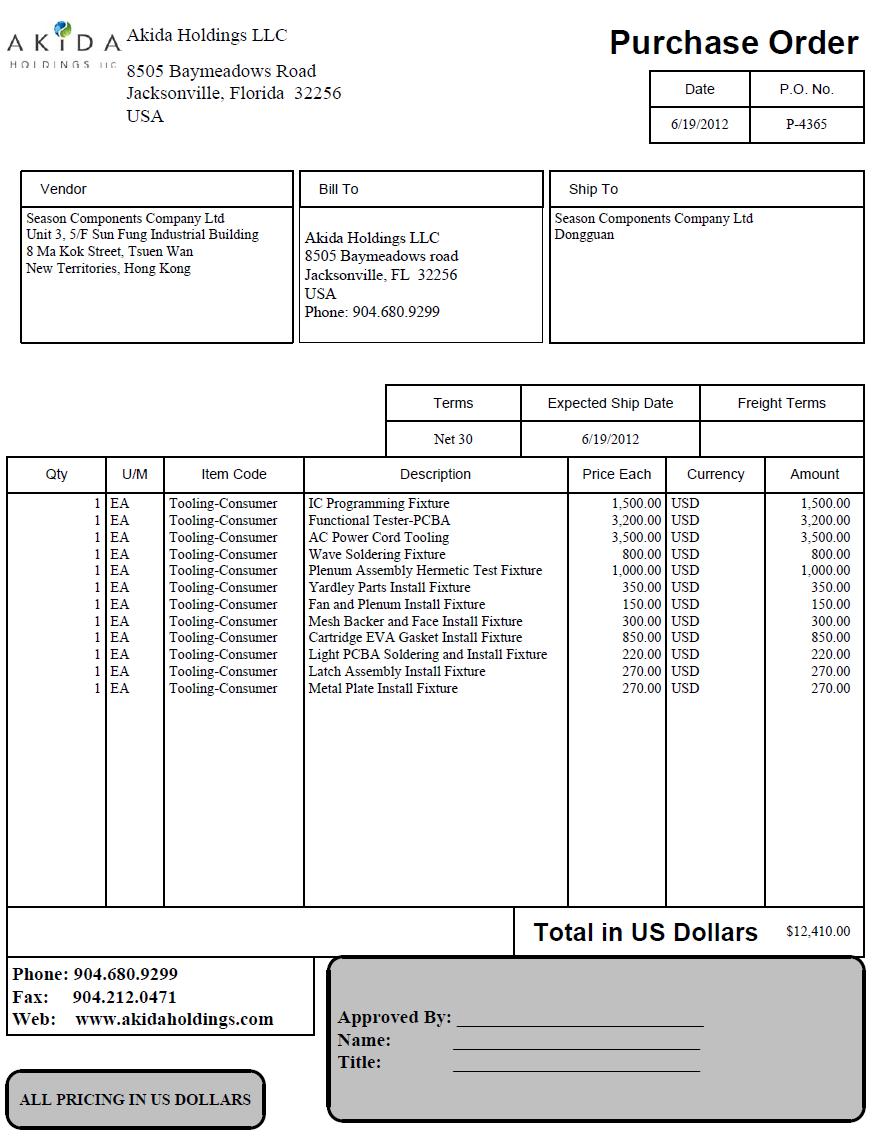
| 42 |
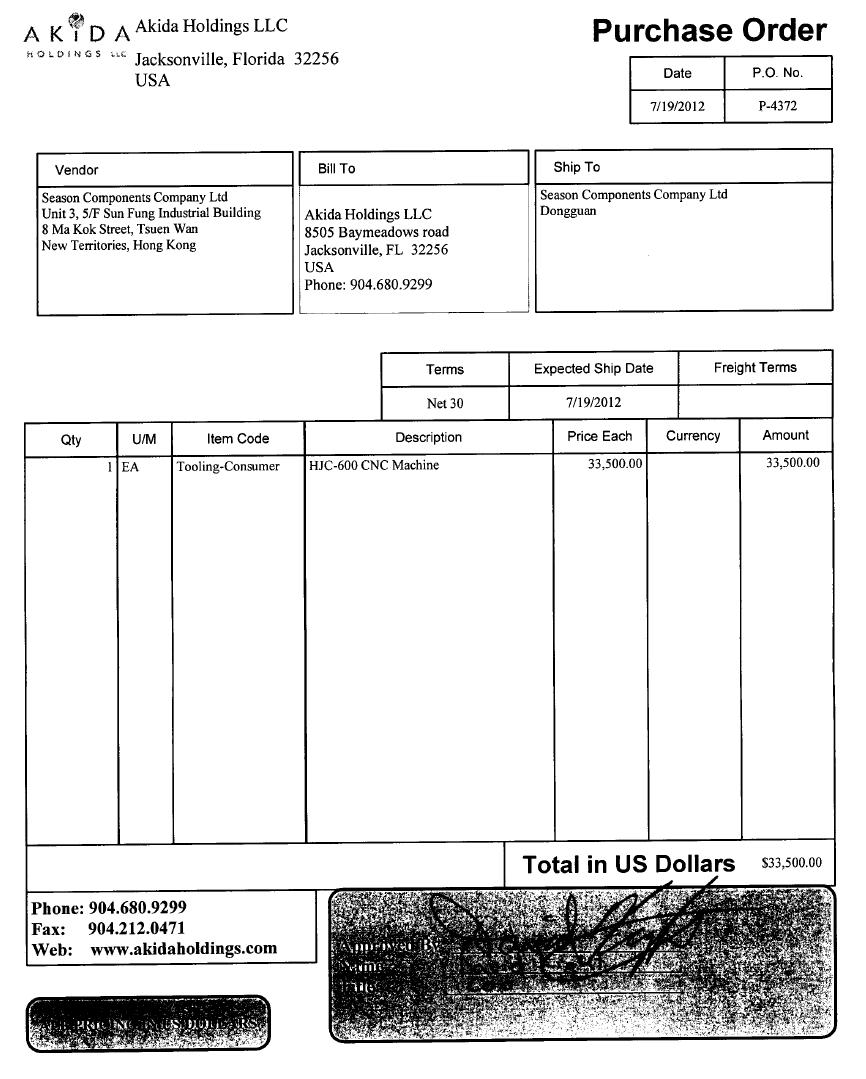
| 43 |
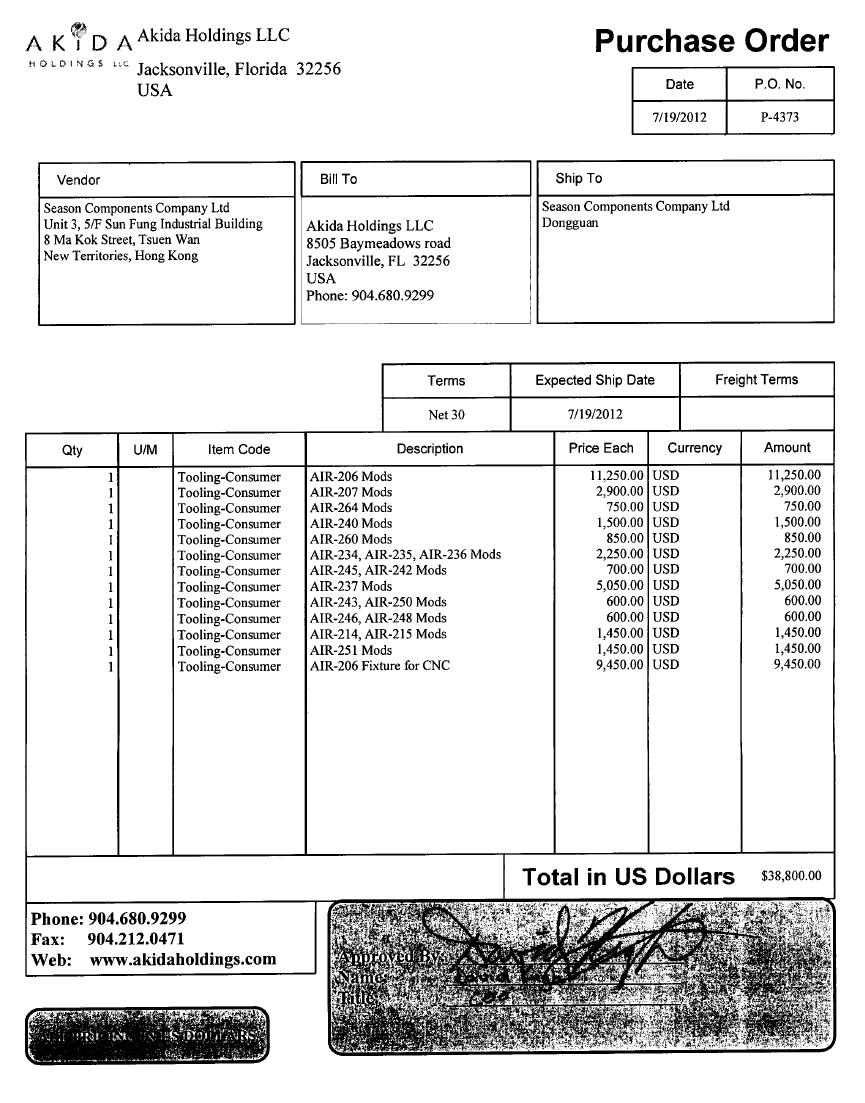
| 44 |

| 45 |
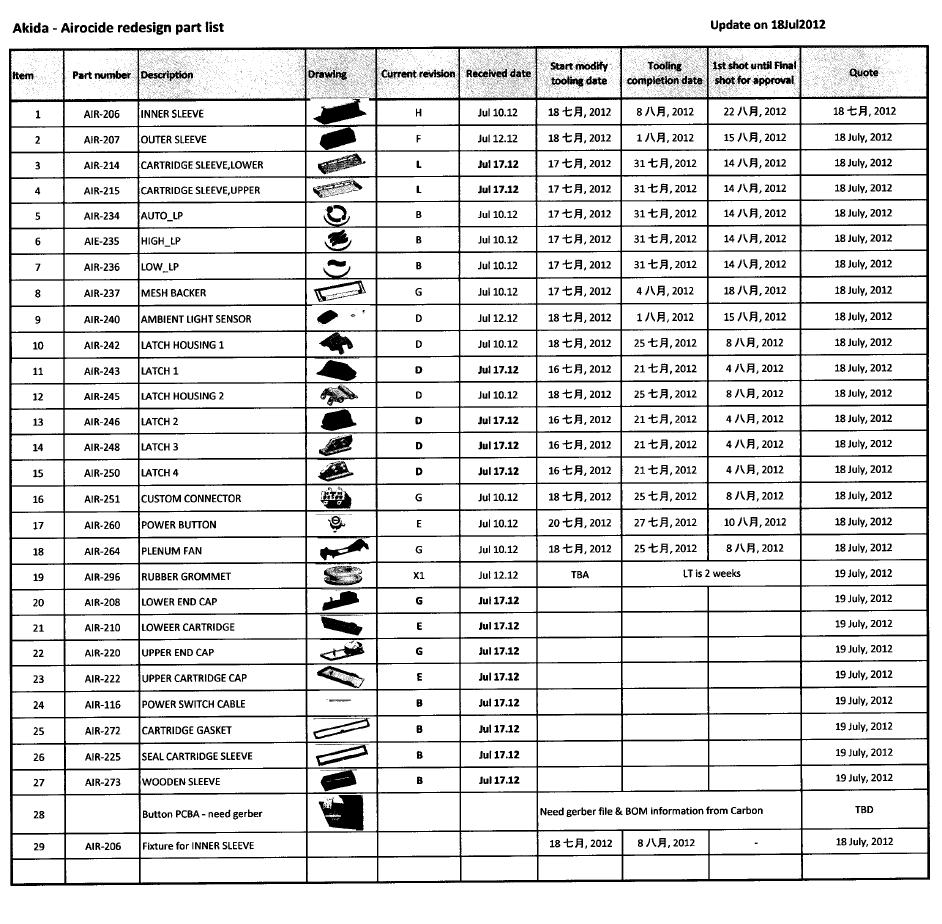
| 46 |

| 47 |
| 48 |
| 49 |
| 50 |

| 51 |

| 52 |
| 53 |

| 54 |

| 55 |
EXHIBIT B
Allocation Schedule
| 56 |
EXHIBIT C
BILL OF SALE
WITH ASSIGNMENT AND ASSUMPTION AGREEMENT
This Bill of Sale and Assignment and Assumption Agreement (the “Agreement”) is made and entered into as of this 8th day of February, 2021, by and between Akida Holdings, LLC, a Florida limited liability corporation and SteriLumen, Inc., a New York corporation having its principal place of business at 8480 East Orchard Road, Suite 2400, Greenwood Village, Colorado 80111 (“Purchaser”).
All capitalized terms used in this Agreement and not otherwise defined in this Agreement shall have the respective meanings assigned to them in the Asset Purchase Agreement described below.
RECITALS
WHEREAS, pursuant to an Asset Purchase Agreement dated as of February 8, 2021 between Seller, the Members, Purchaser and Applied UV, Inc. (the “Asset Purchase Agreement”), Seller has agreed to sell, and Purchaser has agreed to purchase, certain assets, business, property, and rights described in the Asset Purchase Agreement; and
NOW, THEREFORE, in consideration of the foregoing, and the agreements contained herein, the parties hereto hereby agree as follows:
ARTICLE I. SALE AND ASSIGNMENT
1.01. Rights Conveyed
For and in consideration of the Purchase Price and other valuable consideration, receipt of which is hereby acknowledged, Seller hereby sells, transfers, assigns, delivers and conveys to Purchaser, its successors and assigns, to have and to hold forever, and Purchaser hereby assumes, acquires and accepts all of Seller’s right, title and interest in the Acquired Assets;
ARTICLE II. MISCELLANEOUS
3.01. Successors and Assigns
This Agreement shall be binding upon and shall ensure to the benefit of the parties hereto and their respective successors, transferees and assigns.
3.02. Governing Law
This Agreement shall be governed by and construed and interpreted in accordance with the laws of the State of Delaware.
IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be executed as of the date first written above by their respective officers thereunto duly authorized.
| SELLER: | PURCHASER: |
| AKIDA HOLDINGS LLC | STERILUMEN, INC. |
| By: | By: |
| Name: David Kight | Name: Keyoumars Saeed |
| Title: Manager | Title: Chief Executive Officer |
| 57 |
EXHIBIT D
Form of Assignment and Assumption Agreement
ASSIGNMENT AND ASSUMPTION AGREEMENT
THIS ASSIGNMENT AND ASSUMPTION AGREEMENT (this "Agreement") is entered into as of the __ day of February, 2021 between Akida Holdings, LLC, a Florida limited liability company ("Assignor"), SteriLumen, Inc., a New York corporation ("Assignee"). Capitalized terms used but not otherwise defined in this Agreement have the meanings set forth in the Asset Purchase Agreement (as defined below).
RECITALS
WHEREAS, the Assignor, the members of the Assignor, the Assignee and Applied UV, Inc., the parent of the Assignee, have entered into an Asset Purchase Agreement dated the date hereof (the "Asset Purchase Agreement"), pursuant to which the Assignor has agreed to sell and transfer to Assignee all of its right, title and interest in and to the Acquired Assets;
WHEREAS, pursuant to the Asset Purchase Agreement, all of the rights and benefits of the Assignor in and to the Acquired Assets have been sold, transferred and assigned to the Assignee;
WHEREAS, the Acquired Assets include (i) the Intellectual Property Assignment and License Agreement, dated January 1, 2021, between KES Science & Technology, Inc., KES Air Technologies, LLC and Assignor (the “License Agreement”), (ii) the Management Services Agreement, dated January 1, 2021, between KES Science & Technology, Inc. and Assignor (the “Management Agreement”), (iii) the Contract Manufacturing Agreement, dated January 1, 2021, between KES Science & Technology, Inc. and Assignor (the “Manufacturing Agreement” and, together with the License Agreement and the Management Agreement, the “KES Assigned Contracts”) and (iv) the Manufacturing Services Agreement dated May 10, 2012 by and between the Assignor and Season Components Co., Ltd., a Hong Kong corporation (the “MSA” and together with the KES Assigned Contracts, the “Assigned Contracts”) are included with the Acquired Assets; and
WHEREAS, the Assignor desires to assign all of its obligations under the Assigned Contracts and the Assignee desires to assume such obligations.
NOW, THEREFORE, in consideration of the premises and the agreements hereinafter set forth, the parties hereto hereby agree as follows:
| 1. | Assignor hereby irrevocably assigns to Assignee, and Assignee hereby irrevocably accepts from Assignor, all of Assignor's rights, title, benefits and interests in and to the Assigned Contracts. |
| 2. | Assignee hereby irrevocably agrees to assume and perform all of the obligations of the Assignor pursuant to the Assigned Contracts, in the same manner and to the same extent as if the Assignee were the original obligor or obligors thereunder. |
| 3. | Each of the parties hereto hereby represents and warrants to the other as follows: |
| (a) | IMAGE OMITTEDThe execution, delivery and performance of this Agreement are within such party 's powers and have been duly authorized by all necessary limited liability company, corporate or other action. |
| (b) | No authorization or approval or other action by, and no notice to or filing with, any Governmental Authority, regulatory body or any other third party is required for the due execution, delivery and performance by each party to this Agreement. |
| 58 |
| 4. | This Agreement will be binding upon and will inure to the benefit of parties hereto and their respective successors and assigns. |
| 5. | This Agreement may be executed in one or more counterparts, each of which will be deemed to be an original and all of which together will be deemed to be one and the same instrument. |
| 6. | THIS AGREEMENT SHALL BE GOVERNED BY, AND CONSTRUED AND INTERPRETED IN ACCORDANCE WITH, THE LAW OF THE STATE OF DELAWARE WITHOUT REGARD TO THE PRINCIPLES OF CONFLICTS OF LAW THEREOF. |
| 7. | Any one or more of the provisions in this Agreement held to be invalid, illegal or unenforceable in any jurisdiction shall, as to such jurisdiction, be ineffective to the extent of such invalidity, illegality and unenforceability without affecting the validity, legality and enforceability of the remaining provisions of this Agreement; and the invalidity of a particular provision in a particular jurisdiction shall not invalidate such provision in any other jurisdiction. |
[Signature Page Follows]
| 59 |
IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be executed as of the date first above written by their respective duly authorized officers.
| ASSIGNOR: | ||
| AKIDA HOLDINGS, LLC | ||
| By: | ||
| Name: David Knight | ||
| Title: Manager | ||
| ASSIGNEE: | ||
| STERILUMEN, INC. | ||
| By: | ||
| Name: Keyoumars Saeed | ||
| Title: Chief Executive Officer |
The assignment and assumption set forth in Section 1 and Section 2 of this Agreement with respect to the KES Assigned Contracts only, are hereby acknowledged and agreed to:
| KES SCIENCE & TECHNOLOGY, INC. | ||
| By: | ||
| Name: John Hayman, III | ||
| Title: Chief Executive Officer | ||
| KES AIR TECHNOLOGIES, LLC | ||
| By: | ||
| Name: John Hayman, III | ||
| Title: Manager |
| 60 |
EXHIBIT E
Form of Employment Agreement
| 61 |
EXHIBIT F
Form of Consulting Agreement
| 62 |
EXHIBIT G
Assigned Contracts
1. Intellectual Property Assignment and License Agreement, dated January 1, 2021, between KES Science & Technology, Inc., KES Air Technologies, LLC and Akida Holdings, LLC
2. Management Services Agreement, dated January 1, 2021, between KES Science & Technology, Inc. and Akida Holdings, LLC
3. Contract Manufacturing Agreement, dated January 1, 2021, between KES Science & Technology, Inc. and Akida Holdings, LLC
4. Manufacturing Services Agreement dated May 10, 2012 by and between the Akida Holdings, LLC and Season Components Co., Ltd.,
| 63 |
Schedule 1
Schedule of Current Assets and Current Liabilities
| 64 |
Schedule 4.03
Required Consents
None.
| 65 |
Schedule 4.04
Financial Statements
See attachment
| 66 |
Schedule 4.06
Liabilities
| 1. | Inventory credit facility in the amount of $380,416.02 (to be paid in full at Closing); and |
| 2. | Payroll Protection Program loan in the principal amount of $36,832.00. |
| lxvii |
Schedule 4.11(a)
Intellectual Property
1. Trademarks:
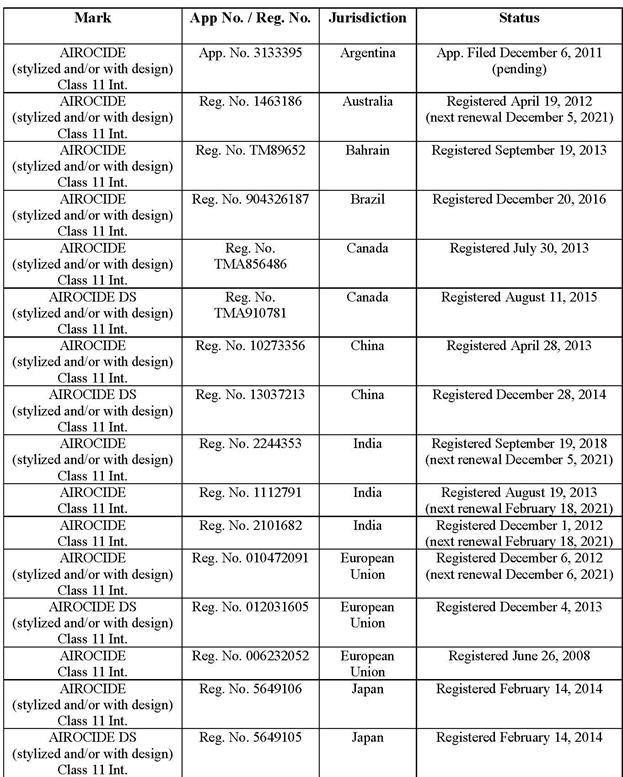
| 68 |
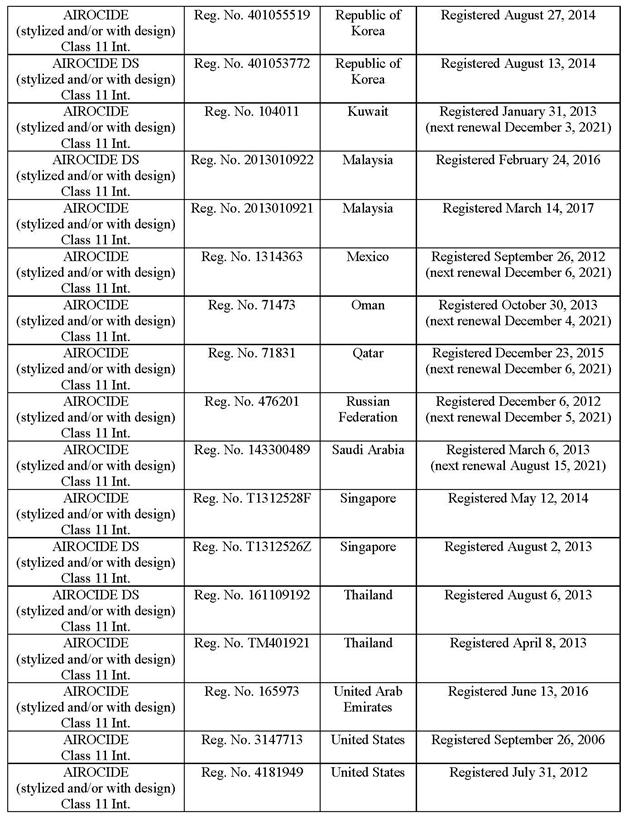
| 69 |
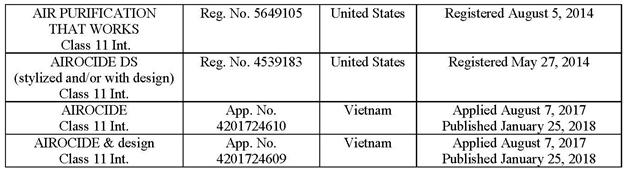
| 70 |
2. Patents:
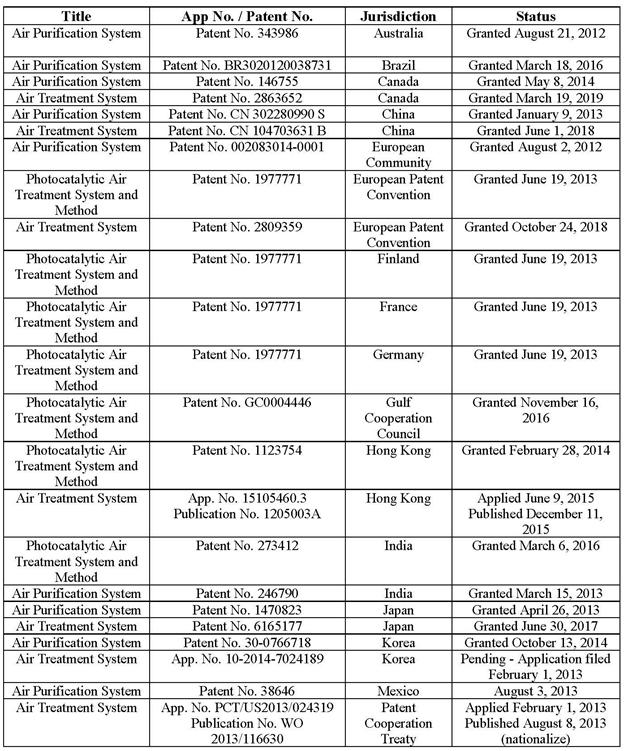
| 71 |
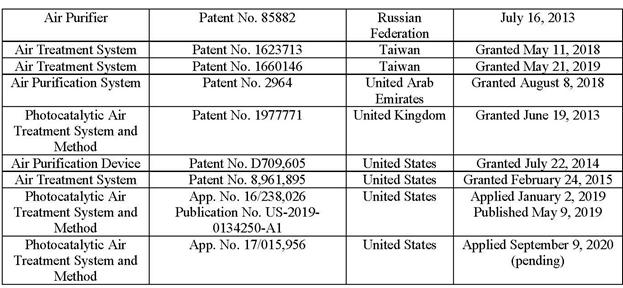
| 72 |
3. URLs:
| GoDaddy.com - Airocide Domains | ||||
| Domain Name | Status | Expiration Date | Auto-renew | Lock |
| airocide.com | Active | 4/11/2022 | On | Locked |
| 73 |
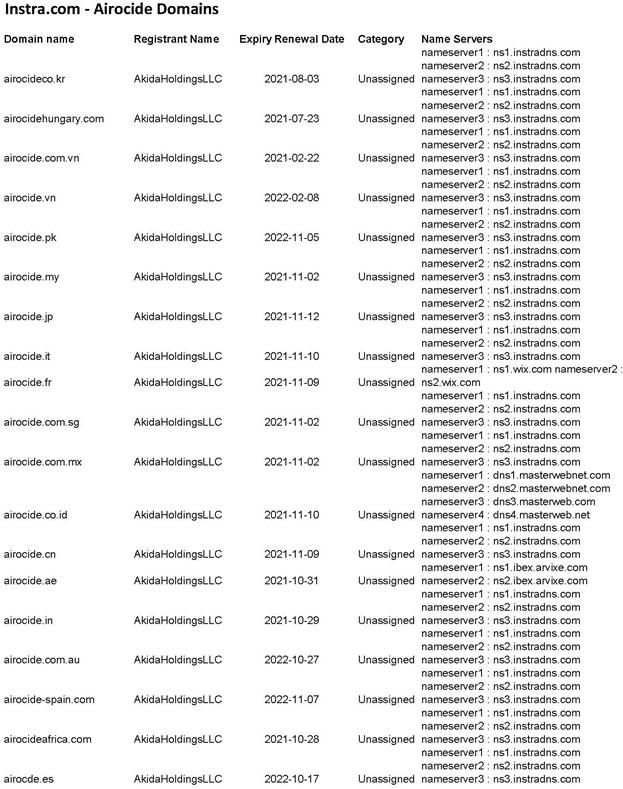
| 74 |

| 75 |

| 76 |

| 77 |
Schedule 4.12
Insurance
| 1. | Product liability policy (number 3605-35-54 EUC) issued by Chubb Custom Insurance Company with an aggregate limit of $2,000,000 and a per occurrence limit of $2,000,000; |
| 2. | Property policy (number 3593-51-24 EUC) issued by Federal Insurance Company with various limits; |
| 3. | General liability policy (number 3593-51-24 EUC) issued by Federal Insurance Company with an aggregate limit of $2,000,000 and a per occurrence limit of $2,000,000; |
| 4. | Workers compensation policy (number 3593-51-24 EUC) issued by Federal Insurance Company with statutory limits; |
| 5. | Auto liability policy (number 3593-51-24 EUC) issued by Federal Insurance Company with a per occurrence limit of $1,000,000; |
| 6. | Blanket accident policy (number 3593-51-24 EUC) issued by Federal Insurance Company with a various limits; and |
| 7. | Workers compensation and employer liability policy (number (21) 7174-30-87) issued by Federal Insurance Company with various limits. |
| 78 |
Schedule 4.13(b)
List of Airocide Contracts
| 79 |
Schedule 4.16(b)
Permits
None.
| lxxx |
Schedule 4.26
Seller FDA Compliance Representations and Warranties
1. The Seller is in material compliance with all applicable Laws and policies administered or enforced by the U.S. Food and Drug Administration (“FDA”). The Seller has operated and is in compliance in all material respects with applicable statutes and implementing regulations administered or enforced by the FDA or any other applicable Governmental Authority. The Seller has not received notice of any pending or threatened Action from the FDA or any other applicable Governmental Authority alleging that any operation or activity of the Seller has been in violation of any applicable Law.
2. To the extent applicable to Seller, the Seller is in compliance in all material respects with all reporting requirements under applicable Laws, including medical device reports (as defined by 21 C.F.R. Part 803), reports of corrections and removals (as defined by 21 C.F.R. Part 806), and the reporting and recordkeeping requirements under the Quality System Regulation of the FDA.
3. To Seller’s Knowledge, Seller does not have any material liability arising out of any injury to individuals or damage to property arising from any of Seller’s products.
4. There is no Action by any Governmental Authority pending against Seller or, to Seller’s Knowledge, threatened against Seller seeking the recall of any of the Seller’s products.
5. To Seller’s Knowledge, there are no material adverse events associated with the use of Seller’s products that Seller is required to report to the FDA that have not been reported to the FDA by Seller in accordance with applicable Law.
6. To Sellers’ Knowledge, there are no set of facts: (i) which could reasonably be expected to furnish a basis for the recall, withdrawal or suspension of any material product registration, product license, export license or other license, approval or consent of any domestic or foreign Governmental Authority with respect to the Seller or any of its commercial products; or (ii) which could reasonably be expected to furnish a basis for the recall, withdrawal or suspension of any commercial product from the market, or the termination or suspension of any clinical testing of any commercial product.
7. The Seller is in material compliance with all applicable Laws requiring the maintenance or submission of reports or records under requirements administered by the FDA or any other Governmental Authority. The Seller has not made any untrue or fraudulent statements of material facts to the FDA, or in any records and documentation prepared or maintained to comply with the applicable Laws.
8. Neither the Seller nor any of its officers or directors has been convicted of any crime or to Seller’s Knowledge engaged in any conduct that would reasonably be expected to result, or that resulted, in debarment, exclusion or disqualification by the FDA, and there are no Actions pending against Seller or, to Seller’s Knowledge, threatened against Seller that reasonably might be expected to result in criminal or civil liability or debarment, exclusion or disqualification of Seller or any of its officers or directors by the FDA. The Seller has not been subject to any other enforcement action involving the FDA or any other Governmental Authorities, including any suspension, consent decree, notice of criminal investigation, indictment, sentencing memorandum, plea agreement, court order or target or no-target letter, and none of the foregoing are pending against Seller or, to Seller’s Knowledge, threatened in writing against the Seller. Neither the Seller nor, to Seller’s Knowledge any of its officers, directors or employees, has committed any act, made any statement or failed to make any statement that would reasonably be expected to provide a basis for the FDA to invoke its policy with respect to “Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities” or for any other Governmental Authority to invoke any similar policy, in each case against the Seller.
9. At the time of the Closing, to Seller’s Knowledge, Seller’s Airocide products are manufactured, packaged, labeled, tested, distributed, sold and/or marketed by or on behalf of the Seller in compliance with, to the extent necessary for the sale of such products in healthcare facilities, the Quality System Regulation of the FDA and, to the extent applicable, counterpart regulations in all other countries where compliance is applicable and in compliance with applicable requirements under the Federal Food, Drug and Cosmetic Act, as amended, and the regulations thereunder (“FDCA”) and similar laws, rules and regulations relating to registration, investigational use, premarket clearance, licensure, or application approval, good manufacturing practices, good laboratory practices, good clinical practices, product listing, quotas, labeling, advertising, record keeping and filing of reports, except where the failure to be in compliance with any of the foregoing would not have a Material Adverse Effect.
10. The Seller or its commercial distributor are working with a qualified regulatory consultant to submit the currently applicable medical device applications and submissions, required for the manufacture and sale of the Airocide products to be cleared by the FDA for medical purposes under Section 510(k) under the FDCA and the Seller has been informed by regulatory consultant that no submissions have yet been made but are in process.
| 81 |
Schedule 4.27
Accounts Receivable
| 82 |


