Exhibit 99.1

TLR9 activation and STAT3 inhibition: a bifunctional approach to immuno - oncology March 2022 STING & TLR - Targeting Therapies Summit 2022
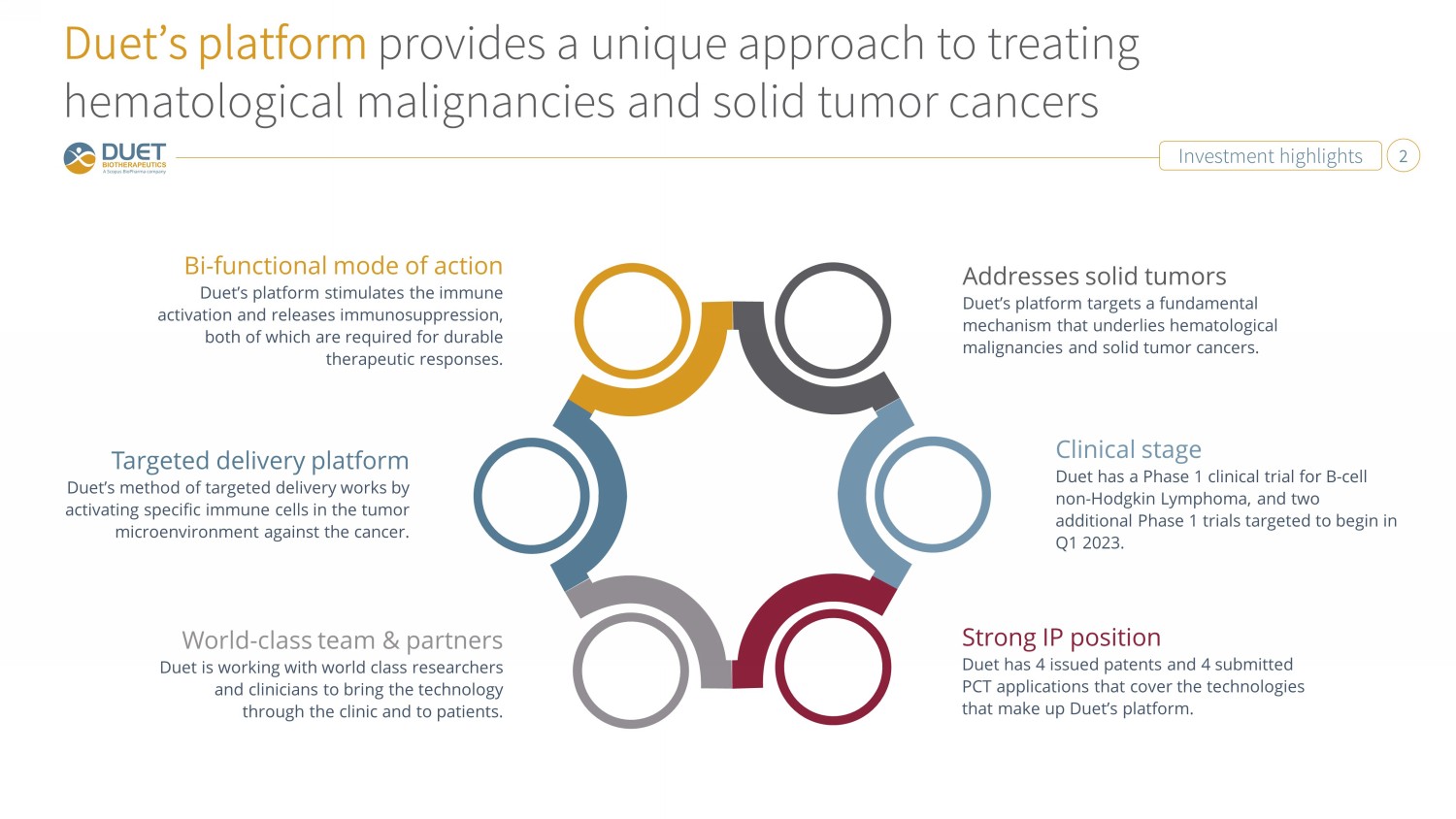
Investment highlights 2 Duet’s platform provides a unique approach to treating hematological malignancies and solid tumor cancers Strong IP position Duet has 4 issued patents and 4 submitted PCT applications that cover the technologies that make up Duet’s platform. Bi - functional mode of action Duet’s platform stimulates the immune activation and releases immunosuppression, both of which are required for durable therapeutic responses. Addresses solid tumors Duet’s platform targets a fundamental mechanism that underlies hematological malignancies and solid tumor cancers. World - class team & partners Duet is working with world class researchers and clinicians to bring the technology through the clinic and to patients. Targeted delivery platform Duet’s method of targeted delivery works by activating specific immune cells in the tumor microenvironment against the cancer. Clinical stage Duet has a Phase 1 clinical trial for B - cell non - Hodgkin Lymphoma, and two additional Phase 1 trials targeted to begin in Q1 2023.
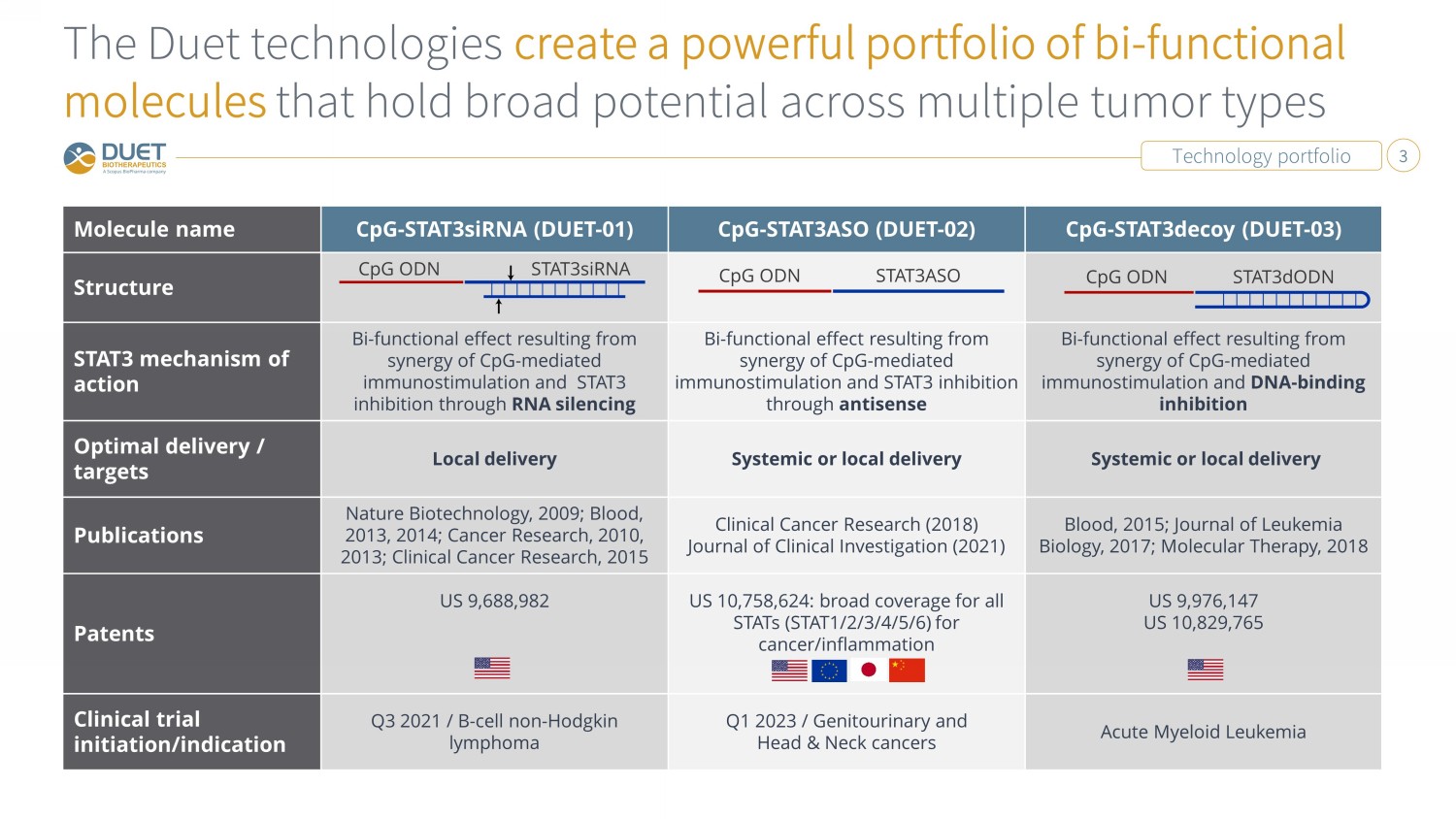
Technology portfolio 3 The Duet technologies create a powerful portfolio of bi - functional molecules that hold broad potential across multiple tumor types Molecule name CpG - STAT3siRNA (DUET - 01) CpG - STAT3ASO (DUET - 02) CpG - STAT3decoy (DUET - 03) Structure STAT3 mechanism of action Bi - functional effect resulting from synergy of CpG - mediated immunostimulation and STAT3 inhibition through RNA silencing Bi - functional effect resulting from synergy of CpG - mediated immunostimulation and STAT3 inhibition through antisense Bi - functional effect resulting from synergy of CpG - mediated immunostimulation and DNA - binding inhibition Optimal delivery / targets Local delivery Systemic or local delivery Systemic or local delivery Publications Nature Biotechnology, 2009; Blood, 2013, 2014; Cancer Research, 2010, 2013; Clinical Cancer Research, 2015 Clinical Cancer Research (2018) Journal of Clinical Investigation (2021) Blood, 2015; Journal of Leukemia Biology, 2017; Molecular Therapy, 2018 Patents US 9,688,982 US 10,758,624: broad coverage for all STATs (STAT1/2/3/4/5/6) for cancer/inflammation US 9,976,147 US 10,829,765 Clinical trial initiation/indication Q3 2021 / B - cell non - Hodgkin lymphoma Q1 2023 / Genitourinary and Head & Neck cancers Acute Myeloid Leukemia CpG ODN STAT3siRNA CpG ODN STAT3ASO CpG ODN STAT3dODN
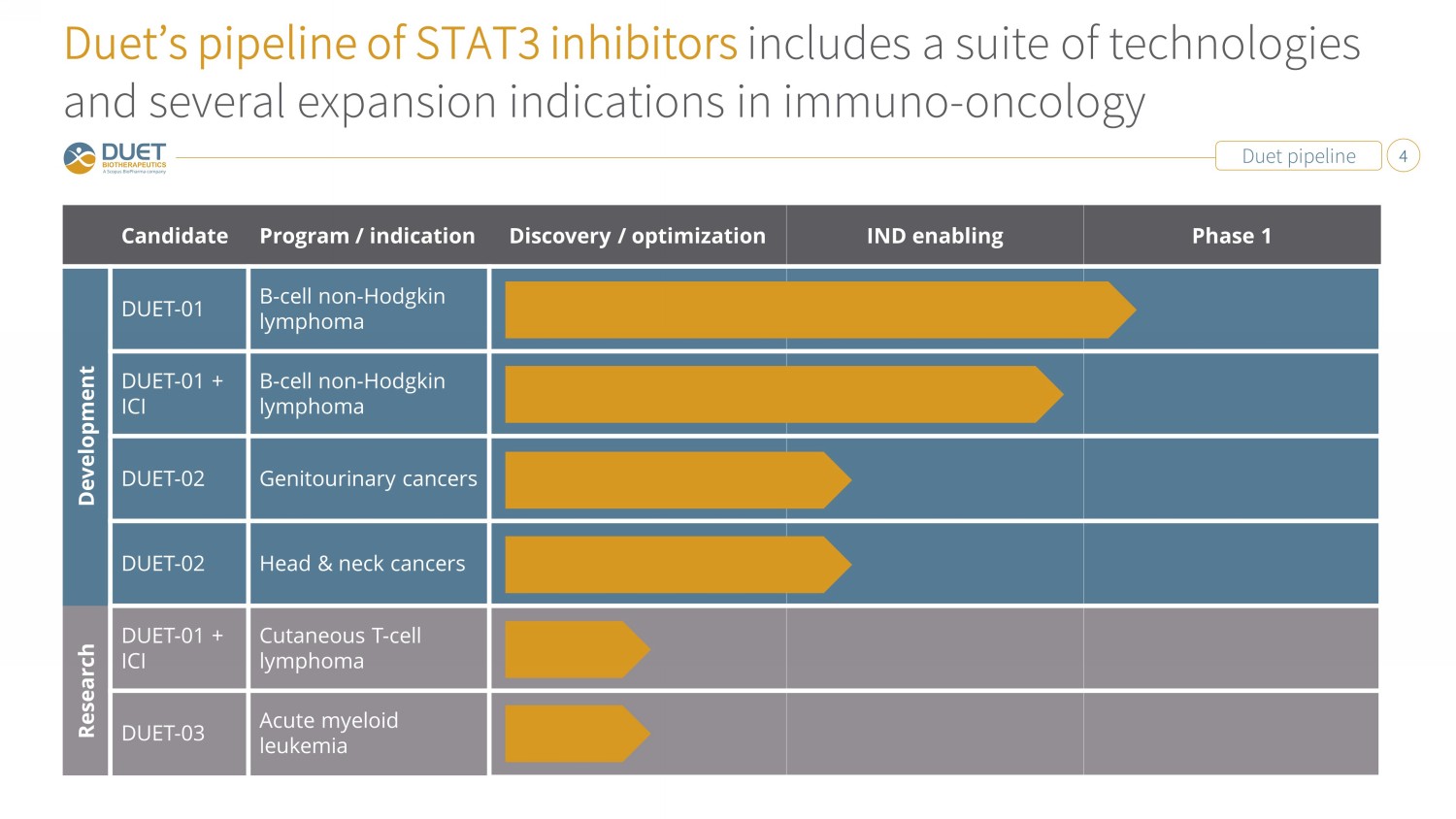
Duet pipeline 4 Duet’s pipeline of STAT3 inhibitors includes a suite of technologies and several expansion indications in immuno - oncology Candidate Program / indication Discovery / optimization IND enabling Phase 1 Development DUET - 01 B - cell non - Hodgkin lymphoma DUET - 01 + ICI B - cell non - Hodgkin lymphoma DUET - 02 Genitourinary cancers DUET - 02 Head & neck cancers Research DUET - 01 + ICI Cutaneous T - cell lymphoma DUET - 03 Acute myeloid leukemia

The problem 5 Tumor cells co - opt the immune system to evade adaptive immunity and promote oncogenesis in the TME Tumors use checkpoints to evade immune response and block activity of cytotoxic T cells and NK cells 2 Tumors recruit T reg and immature myeloid cells to create an immunosuppressive environment and promote oncogenesis 1 Tumor Microenvironment (TME)
STAT3 as an I - O target 6 STAT3 has been implicated in maintaining immunosuppression within the microenvironment of many solid tumors • STAT3 has been shown as key factor in maintenance of immunosuppressive environment found in many solid malignancies, and is activated by many oncogenic pathways • STAT3 in tumor cells downregulates production of cytokines conducive to anti - cancer immunostimulatory effects • creates crosstalk between tumor cells and immune cells in tumor microenvironment driving “feed - forward” loop stimulating tumor cells to upregulate STAT3 • STAT3 promotes expansion of MDSCs, which: • Inhibit activation of CD4+ & CD8+ T - cells • Promote T reg expansion
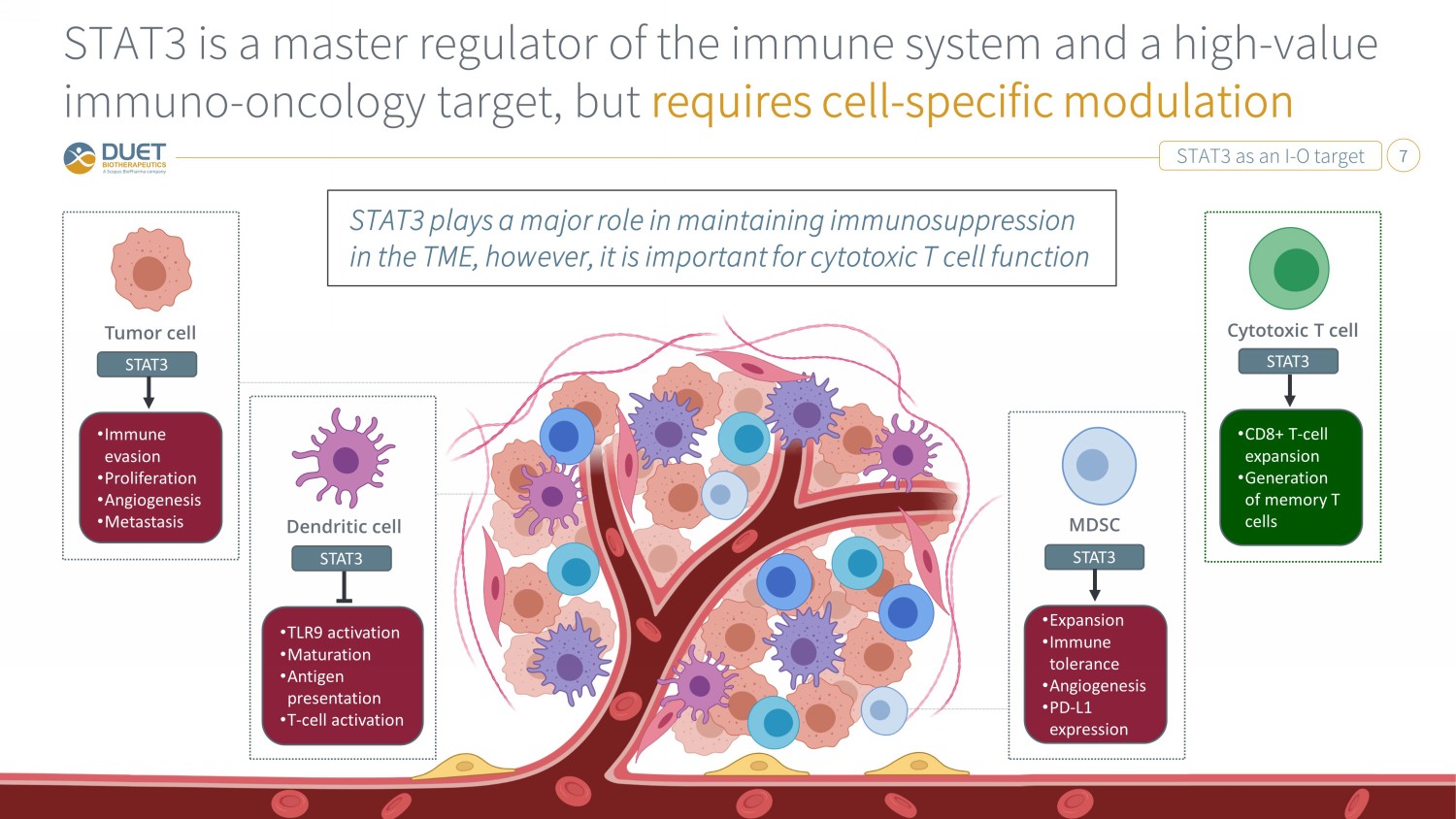
STAT3 as an I - O target 7 STAT3 is a master regulator of the immune system and a high - value immuno - oncology target, but requires cell - specific modulation Tumor cell STAT3 • Immune evasion • Proliferation • Angiogenesis • Metastasis STAT3 plays a major role in maintaining immunosuppression in the TME, however, it is important for cytotoxic T cell function MDSC STAT3 • Expansion • Immune tolerance • Angiogenesis • PD - L1 expression Cytotoxic T cell STAT3 • CD8+ T - cell expansion • Generation of memory T cells Dendritic cell STAT3 • TLR9 activation • Maturation • Antigen presentation • T - cell activation
8 TLR9 activation provokes both innate and adaptive immune responses • Toll like Receptor 9 (TLR9) is an internal cellular receptor in immune cells involved in activation of innate immune responses • Recognizes unmethylated sections of microbial DNA • Synthetic CpG oligonucleotides recognized by receptor as well • Activation of TLR9 by CpG oligonucleotides results in multiple immunomodulatory effects: • Activation of plasmacytoid dendritic cells • Secretion of pro - inflammatory cytokines • Expansion of NK and T - cells TLR9 as an I - O target

The Duet solution 9 Duet strategy: bi - functional oligonucleotide activates the immune system (TLR9) and turns off immunosuppression (STAT3) Scavenger receptors on the surface of myeloid cells have a high affinity for Duet’s oligos, leading to rapid and specific uptake 1 Duet’s oligos are internalized in the cell, where they bind to TLR9, triggering immune system activation 2 Duet’s oligos are then released into the cytosol and inhibit STAT3, allowing for full TLR9 - driven immune activation 3 Tumor Tumor cell death
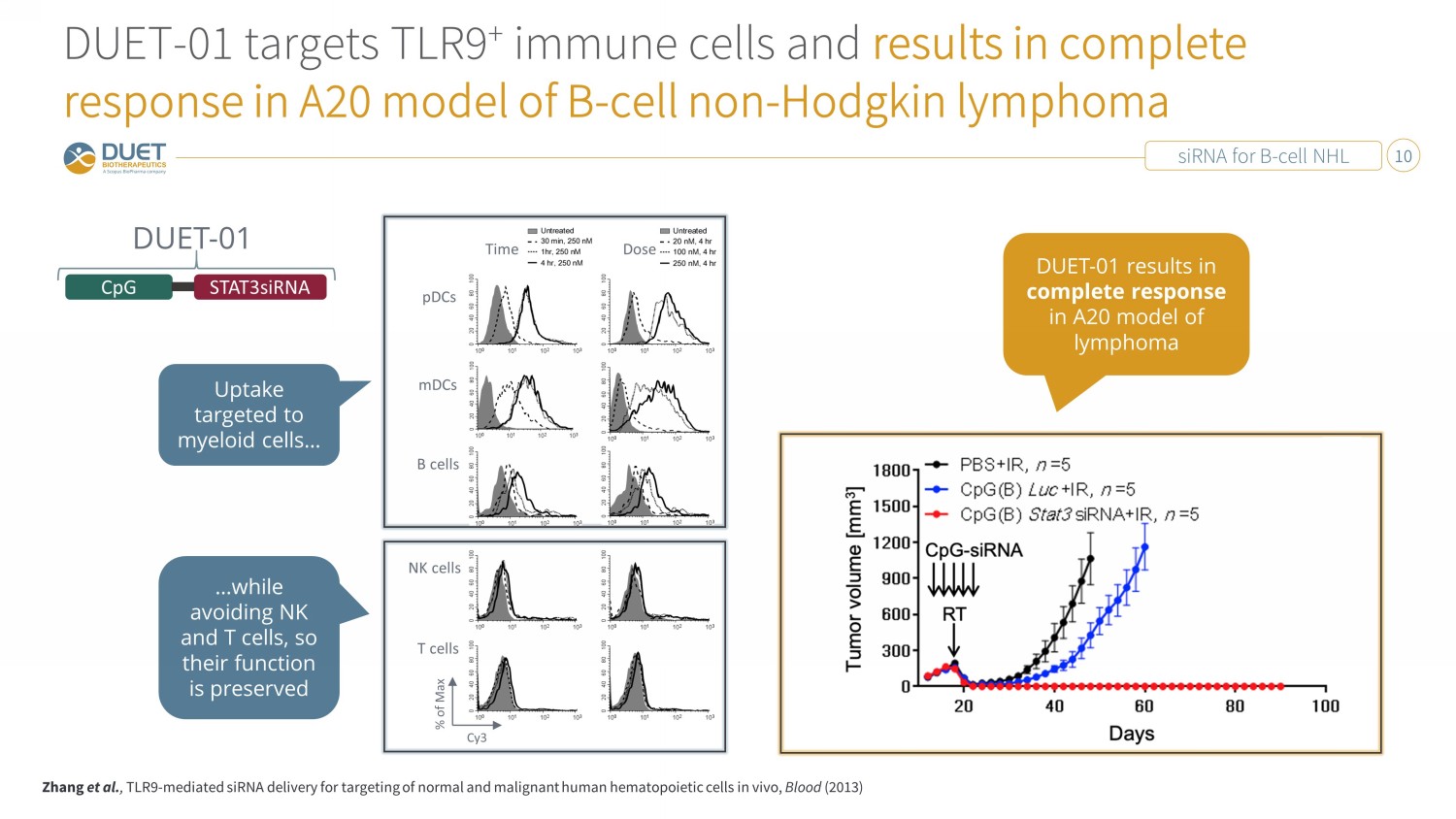
siRNA for B - cell NHL 10 DUET - 01 targets TLR9 + immune cells and results in complete response in A20 model of B - cell non - Hodgkin lymphoma Zhang et al. , TLR9 - mediated siRNA delivery for targeting of normal and malignant human hematopoietic cells in vivo, Blood (2013) CpG STAT3siRNA DUET - 01 % of Max B cells NK cells T cells Dose Time pDCs mDCs Cy3 Uptake targeted to myeloid cells… …while avoiding NK and T cells, so their function is preserved DUET - 01 results in complete response in A20 model of lymphoma
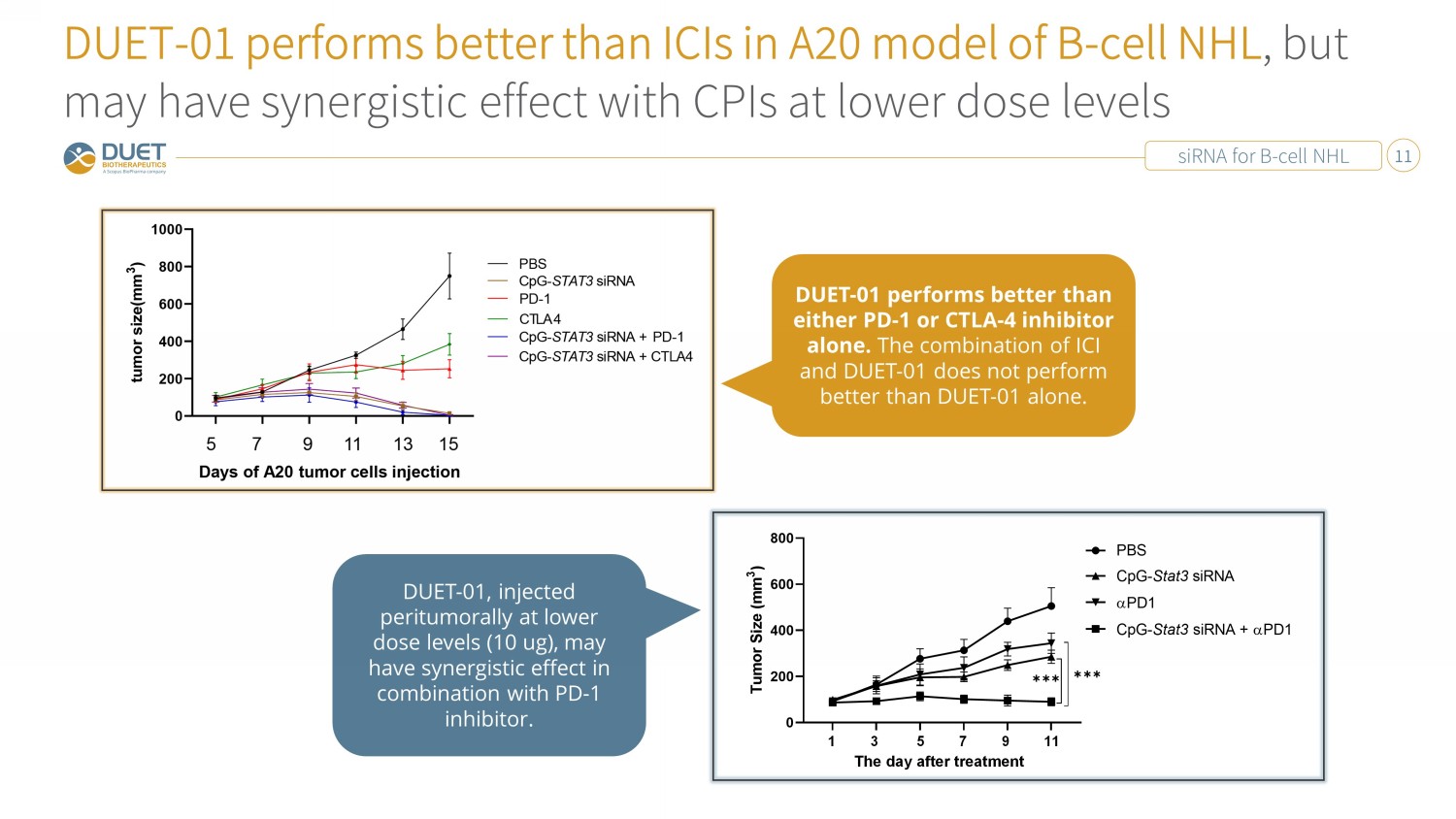
siRNA for B - cell NHL 11 DUET - 01 performs better than ICIs in A20 model of B - cell NHL , but may have synergistic effect with CPIs at lower dose levels 1 3 5 7 9 11 The day after treatment DUET - 01, injected peritumorally at lower dose levels (10 ug), may have synergistic effect in combination with PD - 1 inhibitor. DUET - 01 performs better than either PD - 1 or CTLA - 4 inhibitor alone. The combination of ICI and DUET - 01 does not perform better than DUET - 01 alone.

Phase 1 clinical trial 12 A Phase I study of intratumoral injections of DUET - 01 with local radiation in patients with relapsed/refractory B - cell NHL ELIZABETH BUDDE, MD, PHD Associate Professor, Division of Lymphoma, Department of Hematology & Hematopoietic Cell Transplantation at City of Hope PRIMARY OBJECTIVES: • Determine the recommended Phase 2 dose of DUET - 01 when given in combination with local radiation therapy. • Evaluate safety and feasibility of intratumoral injections when combined with radiation, by evaluation of toxicities. SECONDARY OBJECTIVES: • Characterize the clinical activity of DUET - 01 through the assessment of disease response, and response duration. • Characterize the biologic activity when combined with radiation by assessing through immunologic correlative studies. • Characterize silencing of the STAT3 gene and its key downstream genes through correlative studies. • Characterize local and systemic immune responses , including evidence/extent of immune cell intratumoral infiltration, immune cell activation, and changes in proinflammatory mediators in plasma.
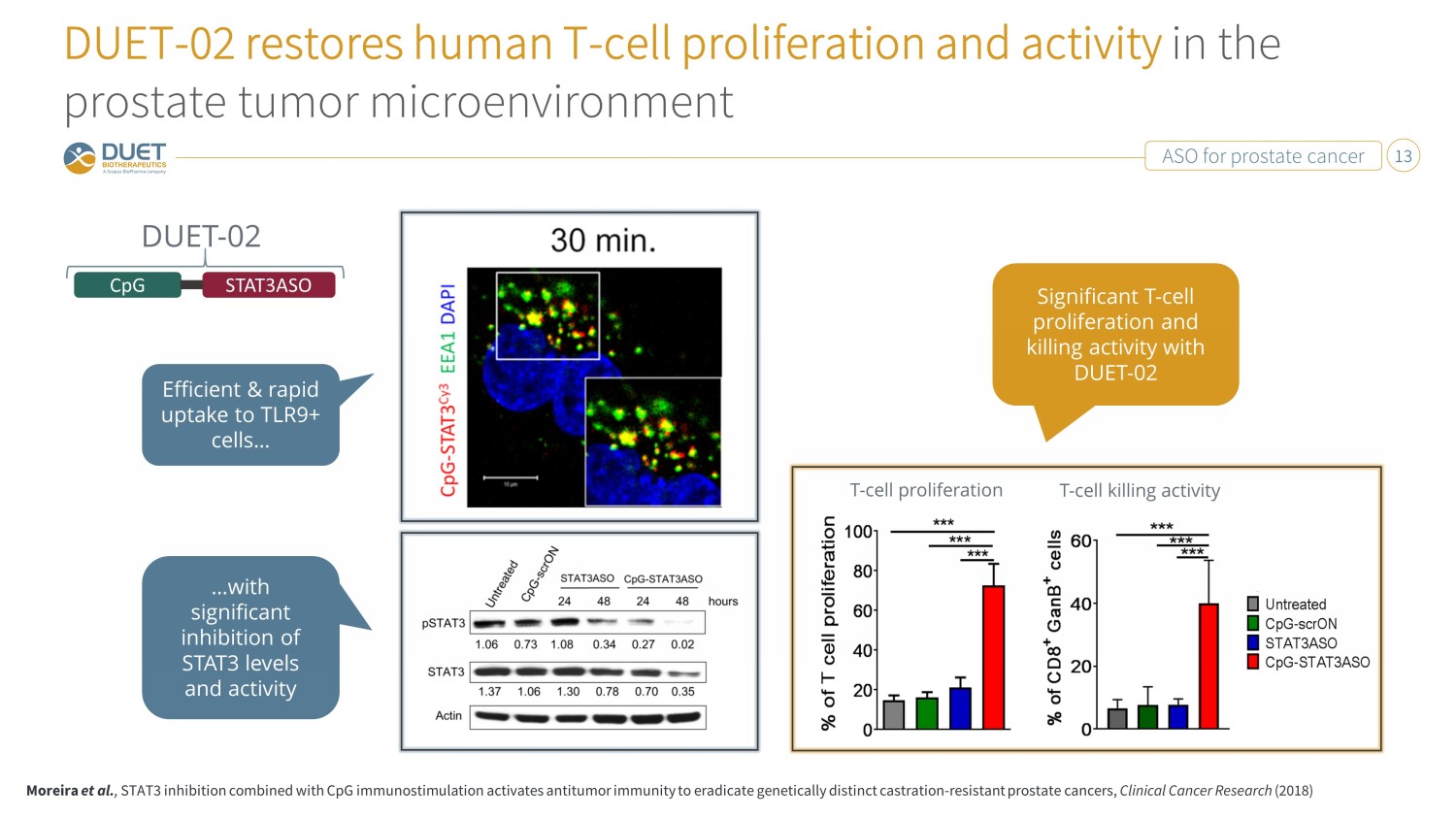
ASO for prostate cancer 13 DUET - 02 restores human T - cell proliferation and activity in the prostate tumor microenvironment T - cell proliferation T - cell killing activity CpG STAT3ASO DUET - 02 Moreira et al. , STAT3 inhibition combined with CpG immunostimulation activates antitumor immunity to eradicate genetically distinct castration - resistant prostate cancers, Clinical Cancer Research (2018) Efficient & rapid uptake to TLR9+ cells… …with significant inhibition of STAT3 levels and activity Significant T - cell proliferation and killing activity with DUET - 02
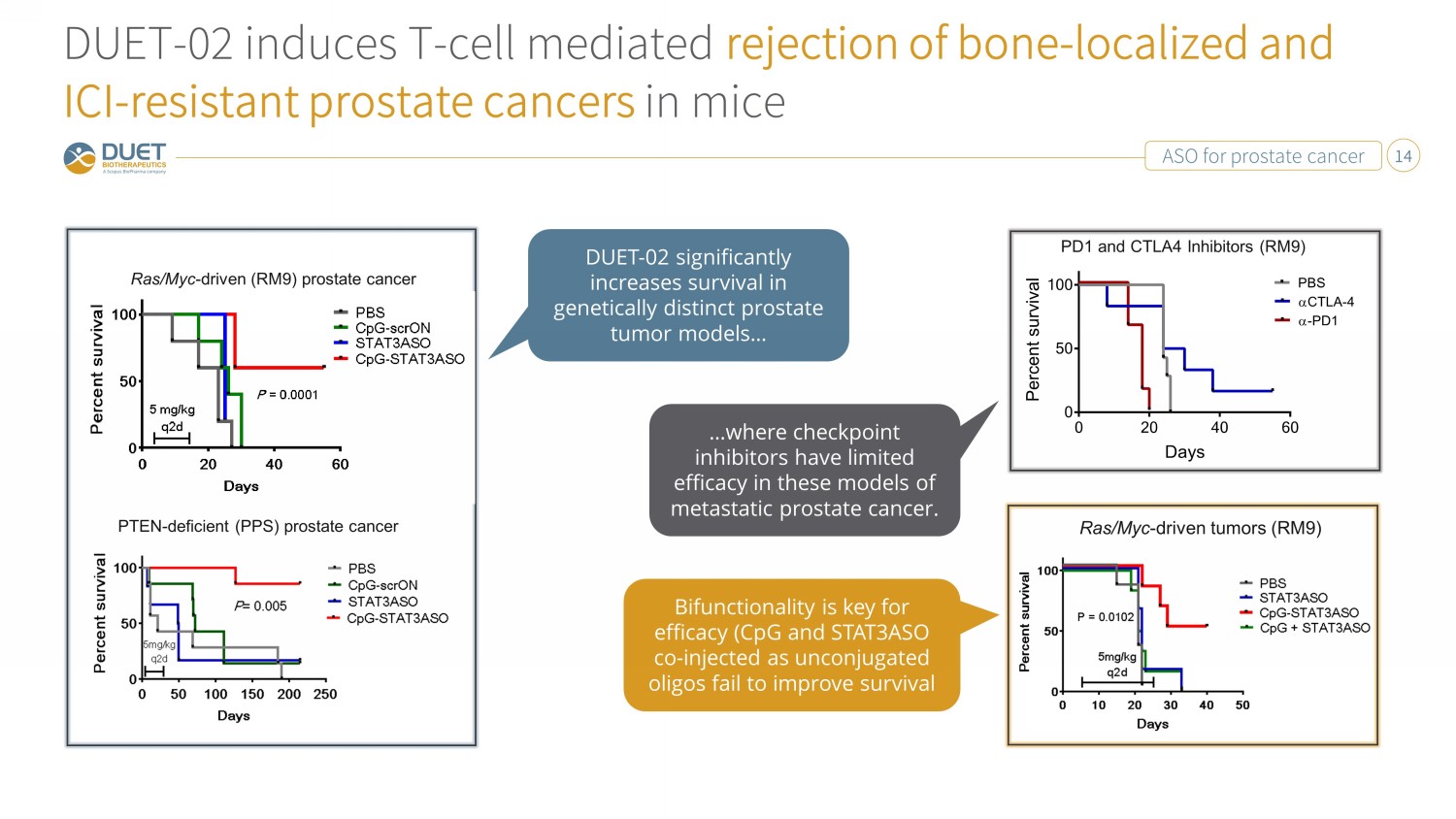
ASO for prostate cancer 14 DUET - 02 induces T - cell mediated rejection of bone - localized and ICI - resistant prostate cancers in mice Ras/Myc - driven (RM9) prostate cancer Ras/Myc-driven tumors (RM9) 5 mg/kg q2d PTEN - deficient (PPS) prostate cancer PD1 and CTLA4 Inhibitors (RM9) Ras/Myc - driven tumors (RM9) DUET - 02 significantly increases survival in genetically distinct prostate tumor models… …where checkpoint inhibitors have limited efficacy in these models of metastatic prostate cancer. Bifunctionality is key for efficacy (CpG and STAT3ASO co - injected as unconjugated oligos fail to improve survival
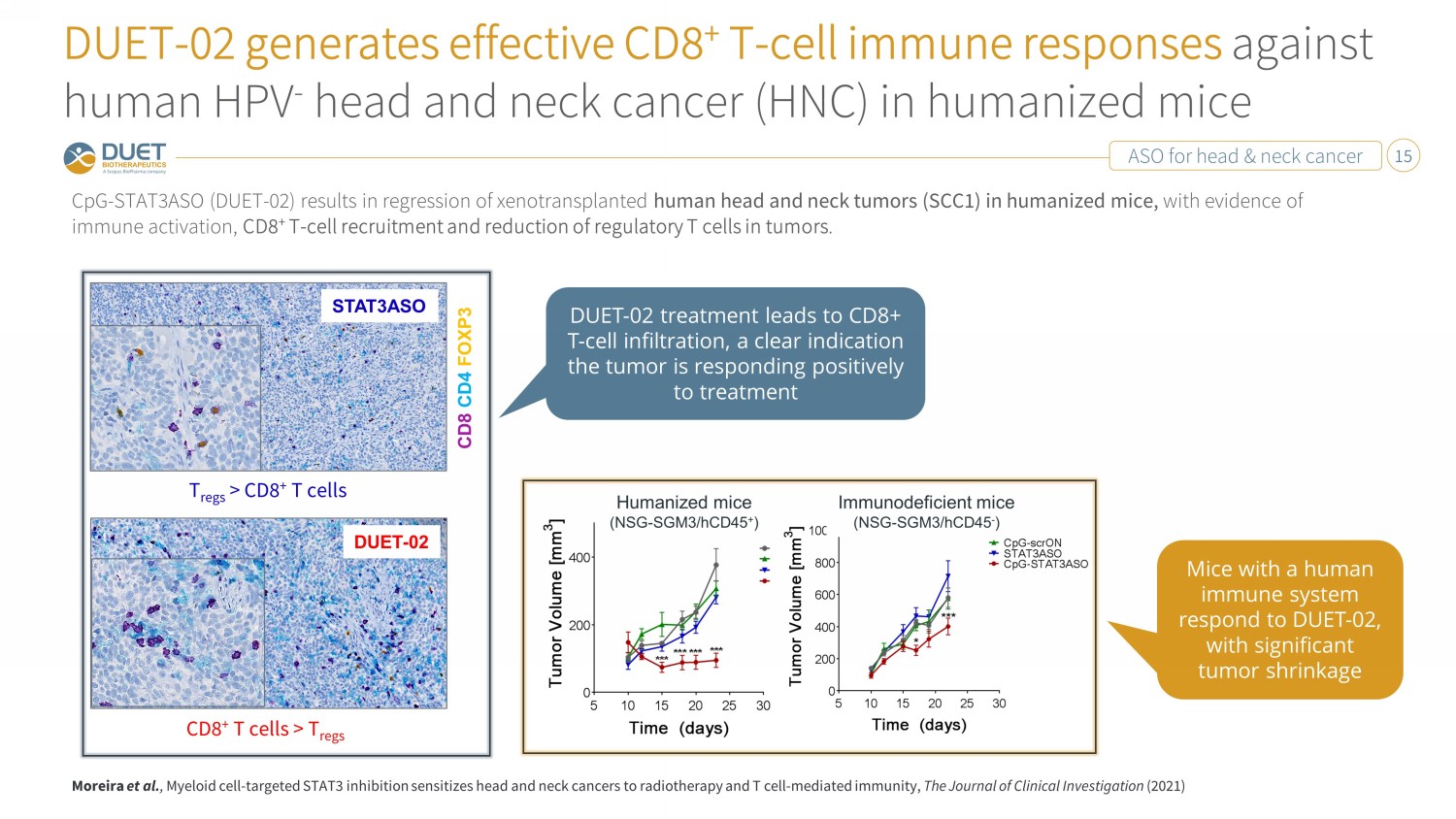
ASO for head & neck cancer 15 DUET - 02 generates effective CD8 + T - cell immune responses against human HPV - head and neck cancer (HNC) in humanized mice CpG - STAT3ASO (DUET - 02) results in regression of xenotransplanted human head and neck tumors (SCC1) in humanized mice, with evidence of immune activation, CD8 + T - cell recruitment and reduction of regulatory T cells in tumors . Humanized mice (NSG - SGM3/hCD45 + ) Immunodeficient mice (NSG - SGM3/hCD45 - ) STAT3ASO DUET - 02 CD8 CD4 FOXP3 CD8 + T cells > T regs T regs > CD8 + T cells Moreira et al. , Myeloid cell - targeted STAT3 inhibition sensitizes head and neck cancers to radiotherapy and T cell - mediated immunity, The Journal of Clinical Investigation (2021) DUET - 02 treatment leads to CD8+ T - cell infiltration, a clear indication the tumor is responding positively to treatment Mice with a human immune system respond to DUET - 02, with significant tumor shrinkage
Planned clinical trials 16 Duet is planning two clinical Phase 1 trials to begin in Q1 2023 for Genitourinary and Head & Neck Cancers Genitourinary Cancers Systemic intravenous infusions of DUET - 02 as a monotherapy for treatment of metastatic prostate, kidney, and bladder cancers Head & Neck Cancers Intratumoral injection of DUET - 02 in combination with radiotherapy for squamous cell carcinoma of head & neck cancers

17 Duet is building a leadership team that has extensive company building and clinical development experience Leadership team ALAN HORSAGER, PHD President, CEO, & Board of Directors MARCIN KORTYLEWSKI, PHD Co - Founder of Olimmune & Senior Scientific Advisor JOHN ROSSI, PHD Professor and Co - Founder of Olimmune, MiNA and Dicerna PROFESSOR NAGY HABIB Professor and Co - Founder of MiNA Therapeutics

Contact Alan Horsager, Ph.D. President – Immuno - Oncology, Scopus BioPharma President & CEO, Duet BioTherapeutics horsager@duettx.com