12-312023FYFalse00016686730.0337268P1Yhttp://www.petiq.com/20231231#LongTermDebtAndFinanceLeasesCurrentConsolidated Statements of Operations00016686732023-01-012023-12-3100016686732023-06-30iso4217:USD0001668673us-gaap:CommonClassAMember2024-02-21xbrli:shares0001668673us-gaap:CommonClassBMember2024-02-210001668673petq:TermLoansMember2023-12-3100016686732023-12-3100016686732022-12-310001668673us-gaap:CommonClassAMember2023-12-31iso4217:USDxbrli:shares0001668673us-gaap:CommonClassAMember2022-12-310001668673us-gaap:CommonClassBMember2023-12-310001668673us-gaap:CommonClassBMember2022-12-310001668673us-gaap:ProductMember2023-01-012023-12-310001668673us-gaap:ProductMember2022-01-012022-12-310001668673us-gaap:ProductMember2021-01-012021-12-310001668673us-gaap:ServiceMember2023-01-012023-12-310001668673us-gaap:ServiceMember2022-01-012022-12-310001668673us-gaap:ServiceMember2021-01-012021-12-3100016686732022-01-012022-12-3100016686732021-01-012021-12-3100016686732021-12-3100016686732020-12-310001668673us-gaap:RetainedEarningsMember2020-12-310001668673us-gaap:AccumulatedDistributionsInExcessOfNetIncomeMember2020-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassAMember2020-12-310001668673us-gaap:TreasuryStockCommonMember2020-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassBMember2020-12-310001668673us-gaap:AdditionalPaidInCapitalMember2020-12-310001668673us-gaap:NoncontrollingInterestMember2020-12-310001668673us-gaap:AccumulatedDistributionsInExcessOfNetIncomeMember2021-01-012021-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassAMember2021-01-012021-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassBMember2021-01-012021-12-310001668673us-gaap:AdditionalPaidInCapitalMember2021-01-012021-12-310001668673us-gaap:NoncontrollingInterestMember2021-01-012021-12-310001668673us-gaap:RetainedEarningsMember2021-01-012021-12-310001668673us-gaap:RetainedEarningsMember2021-12-310001668673us-gaap:AccumulatedDistributionsInExcessOfNetIncomeMember2021-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassAMember2021-12-310001668673us-gaap:TreasuryStockCommonMember2021-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassBMember2021-12-310001668673us-gaap:AdditionalPaidInCapitalMember2021-12-310001668673us-gaap:NoncontrollingInterestMember2021-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassAMember2022-01-012022-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassBMember2022-01-012022-12-310001668673us-gaap:AdditionalPaidInCapitalMember2022-01-012022-12-310001668673us-gaap:NoncontrollingInterestMember2022-01-012022-12-310001668673us-gaap:AccumulatedDistributionsInExcessOfNetIncomeMember2022-01-012022-12-310001668673us-gaap:TreasuryStockCommonMember2022-01-012022-12-310001668673us-gaap:RetainedEarningsMember2022-01-012022-12-310001668673us-gaap:RetainedEarningsMember2022-12-310001668673us-gaap:AccumulatedDistributionsInExcessOfNetIncomeMember2022-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassAMember2022-12-310001668673us-gaap:TreasuryStockCommonMember2022-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassBMember2022-12-310001668673us-gaap:AdditionalPaidInCapitalMember2022-12-310001668673us-gaap:NoncontrollingInterestMember2022-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassAMember2023-01-012023-12-310001668673us-gaap:AdditionalPaidInCapitalMember2023-01-012023-12-310001668673us-gaap:NoncontrollingInterestMember2023-01-012023-12-310001668673us-gaap:AccumulatedDistributionsInExcessOfNetIncomeMember2023-01-012023-12-310001668673us-gaap:RetainedEarningsMember2023-01-012023-12-310001668673us-gaap:RetainedEarningsMember2023-12-310001668673us-gaap:AccumulatedDistributionsInExcessOfNetIncomeMember2023-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassAMember2023-12-310001668673us-gaap:TreasuryStockCommonMember2023-12-310001668673us-gaap:CommonStockMemberus-gaap:CommonClassBMember2023-12-310001668673us-gaap:AdditionalPaidInCapitalMember2023-12-310001668673us-gaap:NoncontrollingInterestMember2023-12-31petq:segmentpetq:distributionCenterpetq:locationpetq:state0001668673us-gaap:TradeAccountsReceivableMember2023-12-310001668673us-gaap:TradeAccountsReceivableMember2022-12-310001668673us-gaap:NotesReceivableMember2023-12-310001668673us-gaap:NotesReceivableMember2022-12-310001668673petq:ComputerEquipmentAndSoftwareMember2023-12-310001668673srt:MinimumMemberpetq:VehicleAndVehicleAccessoriesMember2023-12-310001668673srt:MaximumMemberpetq:VehicleAndVehicleAccessoriesMember2023-12-310001668673us-gaap:BuildingMember2023-12-310001668673srt:MinimumMemberus-gaap:EquipmentMember2023-12-310001668673us-gaap:EquipmentMembersrt:MaximumMember2023-12-310001668673us-gaap:LeaseholdImprovementsMembersrt:MinimumMember2023-12-310001668673us-gaap:LeaseholdImprovementsMembersrt:MaximumMember2023-12-310001668673srt:MinimumMemberus-gaap:FurnitureAndFixturesMember2023-12-310001668673srt:MaximumMemberus-gaap:FurnitureAndFixturesMember2023-12-310001668673us-gaap:ConvertibleNotesPayableMember2020-05-19petq:product0001668673petq:RoccoRoxieMember2023-01-132023-01-130001668673petq:RoccoRoxieMember2023-03-010001668673petq:RoccoRoxieMember2023-01-130001668673us-gaap:TradeNamesMemberpetq:RoccoRoxieMember2023-01-130001668673us-gaap:CustomerRelationshipsMemberpetq:RoccoRoxieMember2023-01-130001668673petq:RoccoRoxieMember2023-01-012023-12-310001668673petq:RoccoRoxieMember2023-12-310001668673us-gaap:LeaseholdImprovementsMember2023-12-310001668673us-gaap:LeaseholdImprovementsMember2022-12-310001668673us-gaap:EquipmentMember2023-12-310001668673us-gaap:EquipmentMember2022-12-310001668673us-gaap:VehiclesMember2023-12-310001668673us-gaap:VehiclesMember2022-12-310001668673petq:ComputerEquipmentAndSoftwareMember2022-12-310001668673us-gaap:BuildingMember2022-12-310001668673us-gaap:FurnitureAndFixturesMember2023-12-310001668673us-gaap:FurnitureAndFixturesMember2022-12-310001668673us-gaap:LandMember2023-12-310001668673us-gaap:LandMember2022-12-310001668673us-gaap:ConstructionInProgressMember2023-12-310001668673us-gaap:ConstructionInProgressMember2022-12-310001668673us-gaap:CertificationMarksMember2023-12-310001668673us-gaap:CertificationMarksMember2022-12-310001668673us-gaap:CustomerRelationshipsMembersrt:MinimumMember2023-12-310001668673us-gaap:CustomerRelationshipsMembersrt:MaximumMember2023-12-310001668673us-gaap:CustomerRelationshipsMember2023-12-310001668673us-gaap:CustomerRelationshipsMember2022-12-310001668673srt:MinimumMemberus-gaap:PatentedTechnologyMember2023-12-310001668673srt:MaximumMemberus-gaap:PatentedTechnologyMember2023-12-310001668673us-gaap:PatentedTechnologyMember2023-12-310001668673us-gaap:PatentedTechnologyMember2022-12-310001668673srt:MinimumMemberus-gaap:TradeNamesMember2023-12-310001668673srt:MaximumMemberus-gaap:TradeNamesMember2023-12-310001668673us-gaap:TradeNamesMember2023-12-310001668673us-gaap:TradeNamesMember2022-12-310001668673us-gaap:TrademarksMember2023-12-310001668673us-gaap:TrademarksMember2022-12-310001668673us-gaap:InProcessResearchAndDevelopmentMember2023-12-310001668673us-gaap:InProcessResearchAndDevelopmentMember2022-12-310001668673us-gaap:InProcessResearchAndDevelopmentMember2023-01-012023-12-310001668673us-gaap:ProductMember2021-12-310001668673us-gaap:ServiceMember2021-12-310001668673us-gaap:ProductMember2022-12-310001668673us-gaap:ServiceMember2022-12-310001668673us-gaap:ProductMember2023-12-310001668673us-gaap:ServiceMember2023-12-310001668673petq:NotesPayableVipAcquisitionMember2021-04-132021-04-130001668673petq:SeniorSecuredAssetBasedRevolvingCreditFacilityMember2021-04-130001668673petq:SeniorSecuredAssetBasedRevolvingCreditFacilityMember2021-04-132021-04-13xbrli:pure0001668673petq:SeniorSecuredAssetBasedRevolvingCreditFacilityMember2023-12-310001668673petq:SeniorSecuredTermLoanFacilityMember2021-04-130001668673srt:MinimumMemberpetq:SeniorSecuredTermLoanFacilityMemberpetq:LondonInterbankOfferedRateLIBOR1Member2021-04-132021-04-130001668673srt:MaximumMemberpetq:SeniorSecuredTermLoanFacilityMemberpetq:LondonInterbankOfferedRateLIBOR1Member2021-04-132021-04-130001668673petq:SeniorSecuredTermLoanFacilityMember2021-04-132021-04-130001668673petq:SeniorSecuredTermLoanFacilityMember2023-12-310001668673us-gaap:ConvertibleNotesPayableMemberus-gaap:CommonClassAMember2020-05-190001668673us-gaap:ConvertibleNotesPayableMemberus-gaap:CommonClassAMember2020-05-192020-05-190001668673us-gaap:ConvertibleNotesPayableMembersrt:MinimumMemberus-gaap:CommonClassAMember2020-05-192020-05-19petq:day0001668673us-gaap:ConvertibleNotesPayableMemberus-gaap:CommonClassAMember2023-12-3100016686732020-05-190001668673us-gaap:CommonClassAMember2020-05-192020-05-190001668673petq:CappedCallTransactionsMemberus-gaap:CommonClassAMember2020-05-142020-05-1900016686732022-12-312022-12-3100016686732020-07-012020-07-3100016686732020-07-310001668673us-gaap:ConvertibleDebtMember2023-12-310001668673us-gaap:ConvertibleDebtMember2022-12-310001668673petq:TermLoansMember2022-12-310001668673us-gaap:RevolvingCreditFacilityMember2023-12-310001668673us-gaap:RevolvingCreditFacilityMember2022-12-310001668673petq:SeniorSecuredTermLoanFacilityMember2021-01-012021-12-310001668673petq:SeniorSecuredAssetBasedRevolvingCreditFacilityMember2021-01-012021-12-310001668673petq:MortgageMember2021-01-012021-12-310001668673srt:MinimumMember2023-12-310001668673srt:MaximumMember2023-12-310001668673petq:ContinuingLlcOwnersMember2023-12-310001668673us-gaap:StateAndLocalJurisdictionMember2023-12-310001668673us-gaap:RestrictedStockUnitsRSUMember2023-01-012023-12-310001668673us-gaap:RestrictedStockUnitsRSUMember2022-01-012022-12-310001668673us-gaap:RestrictedStockUnitsRSUMember2021-01-012021-12-310001668673us-gaap:StockCompensationPlanMember2023-01-012023-12-310001668673us-gaap:ConvertibleDebtSecuritiesMember2023-01-012023-12-310001668673us-gaap:ConvertibleDebtSecuritiesMember2021-01-012021-12-310001668673us-gaap:ConvertibleDebtSecuritiesMember2022-01-012022-12-310001668673petq:OmnibusIncentivePlanMemberus-gaap:CommonClassAMember2023-12-310001668673petq:OmnibusIncentivePlanMemberus-gaap:CommonClassAMember2022-12-310001668673petq:InductionAndRetentionStockPlanMemberus-gaap:CommonClassAMember2018-12-310001668673petq:InductionAndRetentionStockPlanMemberus-gaap:CommonClassAMember2018-01-012023-12-310001668673srt:MaximumMember2023-01-012023-12-310001668673us-gaap:EmployeeStockOptionMember2023-01-012023-12-310001668673us-gaap:EmployeeStockOptionMember2022-01-012022-12-310001668673us-gaap:EmployeeStockOptionMember2021-01-012021-12-310001668673us-gaap:RestrictedStockUnitsRSUMember2023-12-310001668673us-gaap:RestrictedStockUnitsRSUMember2021-12-310001668673us-gaap:RestrictedStockUnitsRSUMember2022-12-31petq:vote00016686732022-09-060001668673petq:AmendedHoldcoLLCAgreementMember2023-12-310001668673petq:PetIQLLCOpcoMember2023-12-310001668673petq:PetiqHoldingsLlcMember2023-01-012023-12-310001668673petq:PetiqHoldingsLlcMember2022-01-012022-12-310001668673petq:ContinuingLlcOwnersMember2021-12-310001668673petq:PetIQLLCOpcoMember2021-12-310001668673petq:AmendedHoldcoLLCAgreementMember2021-12-310001668673petq:ContinuingLlcOwnersMember2022-01-012022-12-310001668673petq:PetIQLLCOpcoMember2022-01-012022-12-310001668673petq:AmendedHoldcoLLCAgreementMember2022-01-012022-12-310001668673petq:ContinuingLlcOwnersMember2022-12-310001668673petq:PetIQLLCOpcoMember2022-12-310001668673petq:AmendedHoldcoLLCAgreementMember2022-12-310001668673petq:ContinuingLlcOwnersMember2023-01-012023-12-310001668673petq:PetIQLLCOpcoMember2023-01-012023-12-310001668673petq:AmendedHoldcoLLCAgreementMember2023-01-012023-12-310001668673us-gaap:CustomerConcentrationRiskMemberpetq:TwoCustomersMemberus-gaap:SalesRevenueNetMember2023-01-012023-12-310001668673us-gaap:CustomerConcentrationRiskMemberpetq:TwoCustomersMemberus-gaap:SalesRevenueNetMember2022-01-012022-12-310001668673us-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMemberpetq:OneCustomerMember2021-01-012021-12-310001668673us-gaap:CustomerConcentrationRiskMemberpetq:OneCustomerMemberus-gaap:AccountsReceivableMember2023-01-012023-12-310001668673us-gaap:CustomerConcentrationRiskMemberpetq:OneCustomerMemberus-gaap:AccountsReceivableMember2022-01-012022-12-310001668673petq:MedVetsInc.AndBayMedicalSolutionsIncMember2023-01-012023-12-310001668673petq:MedVetsInc.AndBayMedicalSolutionsIncMember2022-01-012022-12-310001668673petq:MedVetsInc.AndBayMedicalSolutionsIncMember2021-01-012021-12-310001668673petq:MediationWithThirdPartyMember2021-12-310001668673petq:LawsuitByFormerSupplierForRedemptionOfOwnershipInterestMember2021-12-310001668673petq:LawsuitByFormerSupplierForRedemptionOfOwnershipInterestMember2022-01-012022-12-310001668673petq:ProductsMemberus-gaap:OperatingSegmentsMember2023-01-012023-12-310001668673petq:ServicesMemberus-gaap:OperatingSegmentsMember2023-01-012023-12-310001668673petq:ProductsMemberus-gaap:OperatingSegmentsMember2022-01-012022-12-310001668673petq:ServicesMemberus-gaap:OperatingSegmentsMember2022-01-012022-12-310001668673petq:ProductsMemberus-gaap:OperatingSegmentsMember2021-01-012021-12-310001668673petq:ServicesMemberus-gaap:OperatingSegmentsMember2021-01-012021-12-310001668673petq:ProductsMemberus-gaap:OperatingSegmentsMember2023-12-310001668673petq:ProductsMemberus-gaap:OperatingSegmentsMember2022-12-310001668673petq:ServicesMemberus-gaap:OperatingSegmentsMember2023-12-310001668673petq:ServicesMemberus-gaap:OperatingSegmentsMember2022-12-310001668673petq:DepreciationAndAmortizationMember2023-01-012023-12-310001668673petq:ProductsMembercountry:US2023-01-012023-12-310001668673us-gaap:NonUsMemberpetq:ProductsMember2023-01-012023-12-310001668673petq:ProductsMember2023-01-012023-12-310001668673petq:ServicesMembercountry:US2023-01-012023-12-310001668673us-gaap:NonUsMemberpetq:ServicesMember2023-01-012023-12-310001668673petq:ServicesMember2023-01-012023-12-310001668673country:US2023-01-012023-12-310001668673us-gaap:NonUsMember2023-01-012023-12-310001668673petq:ProductsMembercountry:US2022-01-012022-12-310001668673us-gaap:NonUsMemberpetq:ProductsMember2022-01-012022-12-310001668673petq:ProductsMember2022-01-012022-12-310001668673petq:ServicesMembercountry:US2022-01-012022-12-310001668673us-gaap:NonUsMemberpetq:ServicesMember2022-01-012022-12-310001668673petq:ServicesMember2022-01-012022-12-310001668673country:US2022-01-012022-12-310001668673us-gaap:NonUsMember2022-01-012022-12-310001668673petq:ProductsMembercountry:US2021-01-012021-12-310001668673us-gaap:NonUsMemberpetq:ProductsMember2021-01-012021-12-310001668673petq:ProductsMember2021-01-012021-12-310001668673petq:ServicesMembercountry:US2021-01-012021-12-310001668673us-gaap:NonUsMemberpetq:ServicesMember2021-01-012021-12-310001668673petq:ServicesMember2021-01-012021-12-310001668673country:US2021-01-012021-12-310001668673us-gaap:NonUsMember2021-01-012021-12-310001668673country:US2023-12-310001668673country:US2022-12-310001668673srt:EuropeMember2023-12-310001668673srt:EuropeMember2022-12-31petq:center0001668673petq:LeaseTerminationGainMember2023-01-012023-12-310001668673petq:SettlementOfOperatingLeaseLiabilityMember2023-01-012023-12-310001668673us-gaap:ContractTerminationMember2023-01-012023-12-310001668673petq:CostOfServicesMember2023-01-012023-12-310001668673us-gaap:PropertyPlantAndEquipmentMember2023-01-012023-12-310001668673petq:OperatingLeaseRightOfUseAssetMember2023-01-012023-12-310001668673us-gaap:OtherRestructuringMember2023-01-012023-12-310001668673petq:TotalRestructuringMember2023-01-012023-12-310001668673us-gaap:PropertyPlantAndEquipmentMember2023-12-310001668673petq:OperatingLeaseRightOfUseAssetMember2023-12-310001668673us-gaap:InventoriesMember2023-01-012023-12-310001668673us-gaap:InventoriesMember2023-12-310001668673us-gaap:EmployeeSeveranceMember2023-01-012023-12-310001668673us-gaap:EmployeeSeveranceMember2023-12-310001668673petq:LeaseTerminationGainMember2023-12-310001668673us-gaap:OtherRestructuringMember2023-12-310001668673petq:RelevantInsuranceCompanyMemberpetq:InsurancePolicyPremiumExpenseMember2023-01-012023-12-310001668673petq:RelevantInsuranceCompanyMemberpetq:InsurancePolicyPremiumExpenseMember2022-01-012022-12-310001668673petq:RelevantInsuranceCompanyMemberpetq:InsurancePolicyPremiumExpenseMember2021-01-012021-12-310001668673petq:ColliersInternationalMemberpetq:CorporateOfficeInEagleIdahoMemberpetq:RealEstateCommissionsExpenseMember2021-08-310001668673petq:ColliersInternationalMemberpetq:CorporateOfficeInEagleIdahoMemberpetq:RealEstateCommissionsExpenseMember2021-08-012021-08-310001668673petq:ColliersInternationalMemberpetq:RealEstateCommissionsExpenseMember2021-12-012021-12-310001668673petq:ColliersInternationalMemberpetq:RealEstateCommissionsExpenseMember2020-04-012020-04-300001668673us-gaap:ImmediateFamilyMemberOfManagementOrPrincipalOwnerMemberpetq:MasterServicesAgreementMember2023-12-310001668673us-gaap:ImmediateFamilyMemberOfManagementOrPrincipalOwnerMemberpetq:MasterServicesAgreementMember2023-01-012023-12-310001668673petq:AcadiaInvestorRelationsLLCMemberpetq:InvestorRelationsConsultantMember2022-01-012022-12-310001668673petq:AcadiaInvestorRelationsLLCMemberpetq:InvestorRelationsConsultantMember2023-01-012023-12-31
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| | | | | |
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2023
| | | | | |
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number: 001-38163
| | |
| PetIQ, Inc. |
| (Exact name of registrant as specified in its charter) |
| | | | | |
| Delaware | 35-2554312 |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
| |
230 E. Riverside Drive | 83616 |
Eagle, Idaho | (Zip Code) |
| (Address of principal executive offices) | |
| | |
208-939-8900 |
| (Registrant’s telephone number, including area code) |
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
| Title of Each Class | Trading Symbol | Name of Each Exchange on Which Registered |
| Class A Common Stock, $0.001 par value | PETQ | The NASDAQ Global Select Market |
Securities registered pursuant to Section 12(g) of the Act: None.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes o No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days Yes x No o
Indicate by check mark whether the registrant has submitted electronically Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes x No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | |
| Large Accelerated Filer o | | Accelerated filer x |
| Non-accelerated filer o | | Smaller reporting company o |
| Emerging growth company o | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. x
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. o
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b).
o
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). o Yes x No
As of June 30, 2023, the last business day of the registrant’s most recently completed second fiscal quarter, the aggregate market value of common equity held by non-affiliates of the registrant was $402.6 million. Shares of Class A common stock held by each executive officer, director and by certain persons that own 10 percent or more of the outstanding Class A common stock have been excluded in that such persons may be deemed to be affiliates. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of February 21, 2024 we had 29,199,778 shares of Class A common stock and 231,540 shares of Class B common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
We intend to file with the Securities and Exchange Commission, not later than 120 days after the close of our fiscal year ended December 31, 2023, a definitive proxy statement or an amendment to this report filed under cover of Form 10-K/A containing the information required to be disclosed under Part III of Form 10-K.
PetIQ, Inc.
Table of Contents
Cautionary Note Regarding Forward-Looking Statements
This Annual Report on Form 10-K (this “Annual Report”) contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements involve risks and uncertainties, such as statements about our plans, objectives, expectations, assumptions or future events. In some cases, you can identify forward-looking statements by terminology such as “anticipate,” “estimate,” “plan,” “project,” “continuing,” “ongoing,” “expect,” “believe,” “intend,” “may,” “will,” “should,” “could” and similar expressions. Examples of forward-looking statements include, without limitation:
•statements regarding our strategies, results of operations or liquidity;
•statements concerning projections, predictions, expectations, estimates or forecasts as to our business, financial and operational results and future economic performance;
•statements of management’s goals and objectives; and
•assumptions underlying statements regarding us or our business.
Forward-looking statements involve estimates, assumptions, known and unknown risks, uncertainties and other factors that could cause actual results to differ materially from any future results, performances, or achievements expressed or implied by the forward-looking statements. Forward-looking statements should not be read as a guarantee of future performance or results, and will not necessarily be accurate indications of the times at, or by, which such performance or results will be achieved. Forward-looking statements are based on information available at the time those statements are made or management’s good faith belief as of that time with respect to future events, are subject to risks and uncertainties that could cause actual performance or results to differ materially from those expressed in or suggested by the forward-looking statements. Important factors that could cause such differences include, but are not limited to, factors discussed under the heading “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in this Annual Report; general economic or market conditions, including inflation, and interest rates, ; overall consumer spending in our industry;our ability to successfully grow our business through acquisitions and our ability to integrate acquisitions; our dependency on a limited number of customers; our ability to implement our growth strategy effectively our ability to continue to grow our Services segment; disruptions in our manufacturing, shipping, transportation and distribution chains; competition from veterinarians and others in our industry; reputational damage to our brands; the effectiveness of our marketing and trade promotion programs; recalls or withdrawals of our products or product liability claims; to introduce new products and improve existing products; our failure to protect our intellectual property; costs associated with governmental regulation; our ability to keep and retain key employees; our ability to sustain profitability; cyber security risks including breaches that result in business interruption and data loss; our substantial indebtedness and our ability to raise additional capital as needed; and the risks set forth under the “Risk Factors” section of this Annual Report.
Additional risks and uncertainties not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, financial condition or operating results. The forward-looking statements speak only as of the date on which they are made, and, except as required by law, we undertake no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events. In addition, we cannot assess the impact of each factor on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements. Consequently, you should not place undue reliance on forward-looking statements.
Summary of Selected Risk Factors Associated with Our Business
Our business operations are subject to numerous risks and uncertainties, including the risks described in the section titled “Risk Factors” included under Part I, Item 1A of this Annual Report. The following is only a summary of the principal risks associated with an investment in our Class A common stock. Material risks that may adversely affect our business, financial condition or results of operations included, but are not limited to, the following:
•Unfavorable economic conditions could reduce spending on our products and limit our ability to grow our business and negatively affect our results of operations.
•We may seek to grow our business through acquisitions of or investments in new or complementary businesses which necessitate incurring significant debt, facilities, technologies or products, or through strategic alliances, and the failure to manage acquisitions, investments or strategic alliances, or the failure to integrate them with our existing business, could have a material adverse effect on us.
•We are dependent on a relatively limited number of customers for a significant portion of our net sales.
•We may not be able to successfully implement our growth strategy in our Products segment on a timely basis or at all.
•We may not continue to grow our Services business.
•We operate in a highly competitive industry and may lose market share or experience margin erosion if we are unable to compete effectively as competitors develop new offerings or improve existing offerings.
•Resistance from veterinarians to authorize prescriptions, or attempts/efforts on their part to discourage pet owners to purchase from retailers and pharmacies could cause our net sales to decrease and could materially adversely affect our financial condition and results of operations.
•Our growth and business are dependent on consumer, product, brand or ingredient trends that may change, and our historical growth may not be indicative of our future growth.
•We may not be able to manage our manufacturing and supply chain effectively, which may adversely affect our results of operations.
•Shipping and transportation is a critical part of our business and any changes in, or disruptions to, our shipping arrangements could adversely affect our business, financial condition, and results of operations.
•The growth of our business depends in part on our ability to accurately predict consumer trends, engage consumers with effective marketing, successfully develop and introduce new products, improve existing products, and expand into new offerings.
•Failure or perceived failure to comply with existing or future laws, regulations, contracts, self-regulatory schemes, standards, and other obligations related to data privacy and security (including security incidents) could harm our business. Compliance or the actual or perceived failure to comply with such obligations could increase the costs of our products and services, limit their use or adoption, and otherwise negatively affect our operating results and business.
•Pandemics and disease outbreaks and any related economic downturn have in the past and could continue to negatively impact our business, financial condition and results of operations.
•We have a substantial amount of debt, which could adversely affect our financial position and our ability to raise additional capital and prevent us from fulfilling our obligations.
•If our cash from operations is not sufficient to meet our current or future operating needs, expenditures and debt service obligations, our business, financial condition and results of operations may be materially adversely affected.
•The trading price of our Class A common stock is highly volatile and the market price of our shares of Class A common stock may decline in spite of our operating performance.
•We are subject to extensive and ongoing governmental regulation and we may incur material costs in order to comply with existing or future laws and regulations, and our failure to comply may result in enforcement, recalls and other adverse actions or significant penalties.
•Our principal asset is our interest in HoldCo, (defined below), and, accordingly, we depend on distributions from HoldCo to pay our taxes and expenses. HoldCo’s ability to make such distributions may be subject to various limitations and restrictions.
PART I
Unless the context requires otherwise, references in this Annual Report to "PetIQ, Inc.," "PetIQ," the "Company," "we," "our" or "us" refer collectively to PetIQ, Inc. and its consolidated subsidiaries, including PetIQ Holdings, LLC, a Delaware limited liability company, which we refer to as “HoldCo.” PetIQ, Inc. is the sole managing member of HoldCo, which is the sole member of PetIQ, LLC (“Opco”) and, through HoldCo, operate and control all of the business and affairs of Opco.
PetIQ, Inc. and HoldCo are holding companies with no other operations, cash flows, material assets or liabilities other than the equity interests in Opco.
Item 1 - Business
Business Overview
PetIQ is a leading pet medication, product and wellness company delivering a smarter way for pet parents to help pets live their best lives through convenient access to affordable health and wellness products and veterinary services. We have two reporting segments: (i) Products; and (ii) Services. PetIQ believes that pets are an important part of the family and deserve the best products and care we can provide.
Our Products segment consists of our product manufacturing and distribution business through which we manufacture and distribute pet medication and health and wellness products to major U.S. retail and e-commerce channels through more than 60,000 points of distribution. We focus our offerings on innovative, proprietary value-branded products, and leading third-party branded products for dogs and cats, including pet Rx medications, OTC medications and wellness products. Our Products segment is further supported by our world-class medications manufacturing facility in Omaha, Nebraska and health and wellness manufacturing facility in Springville, Utah.
Our Services segment consists of veterinary services, and related product sales, provided by the Company directly to consumers. Our national veterinarian service platform operates at over 2,600 community clinic locations and wellness centers hosted at retailers across 39 states providing cost effective and convenient veterinary wellness services. We offer diagnostic tests, vaccinations, prescription medications, microchipping, grooming and hygiene and wellness checks.
Our Industry
Attractive Pet Industry Trends. By year-end 2023, approximately 50% of total U.S. households owned a dog or cat. Packaged Facts estimates that by 2026 approximately 55% of United States households will own a pet:
▪Pet Humanization: In the United States, according to The Human Animal Bond Research Institute (HABRI) and Zoetis data for 2021, 95% of pet owners consider their pet a part of their family, and 98% reported that they have personally experienced health benefits from having a pet in their lives. With pets increasingly viewed as companions, friends and family members, pet owners behave like “pet parents” with a strong inclination for spending disposable income to meet all of their pets’ needs during all economic cycles. Pets have become a household and individual spending priority. In a Packaged Facts survey conducted in 2022, 68% of consumers reported that they have been cutting back on household expenses due to price inflation, economic uncertainty, or other factors, however only 14% to 15% reported cutting back in pet care categories.
▪Increasing Consumer Focus on Pet Health and Wellness: Consumers are exhibiting greater interest in improved health for their pets and, as a result, are increasing their spending on veterinary care as well as purchases of the most effective veterinarian-grade pet products and supplies. In 2022, in the wake of COVID-19, Packaged Facts survey data showed that 58% of dog owners and 54% of cat owners strongly agree that they "look for products to improve my pet's’ health and wellness."
▪Increasing Focus on Affordable Products and Services: According to Packaged Facts, 32% of dog/cat owners who consider their pets part of the family are concerned about the affordability of routine health care for their pets and 42% are concerned about the affordability of emergency care for their pets. In a 2021 survey, 68% of pet owners agreed that they were seeking lower prices, special offers, and sales on pet products. Pet owners of all demographic and income levels aspire to purchase leading veterinarian-grade treatments.
▪Increasing Market Size and Consumer Spending: Pet spending in the United States has steadily increased every year since 1994, with Americans spending approximately $122.8 billion on pet products and services for their pets in 2021, up from $81.8 billion in 2016, representing a Compound Annual Growth Rate ("CAGR)" of 8.5%. Packaged Facts projects the total U.S. pet products and services market to grow at a CAGR of 7.3% from 2021 to 2026.
Strong Growth in Pet Products. According to Packaged Facts, the $134.5 billion U.S. consumers spent on pet products and services in 2022 more than doubled 2010 spending of $53.7 billion. Veterinary channel sales of pet medications grew from an estimated $6.7 billion in 2018 to $8.6 billion in 2022, and overall retail and veterinary channel sales of pet medications and supplements are estimated to have grown from $10.1 billion in 2018 to $14.3 billion in 2022, according to Packaged
Facts, with pet supplement sales growing from $1.1 billion in 2018 to $2.3 billion in 2022 in keeping with increasing consumer attention to pet health and wellness.
Growth of Pet Medication Purchases from Retail and E-commerce Channels. U.S. retail sales of pet medications reached $12.1 billion in 2022, inclusive of sales through veterinarians, brick-and-mortar stores and online retailers. Packaged Facts projects that pet medication sales will grow to $13.8 billion by 2026. The COVID-19 pandemic accelerated the migration of pet owner purchases of veterinary-grade pet products from retail and e-commerce channels including both brick-and-mortar and online offerings and away from purchases directly from veterinarians. We believe this migration will continue in the future as more consumers take advantage of the convenience of both local brick-and-mortar and online retail options.
Our Business Strategy
There are significant opportunities to grow our brand awareness, increase our net sales and profitability and deliver shareholder value by executing on the following initiatives:
Grow Consumer Awareness of and Engagement with Our Brands/Products in the Retail & E-commerce Channels. We are an established category leader in the pet health and wellness and medication market. We maintain strong relationships with the veterinary channel and distributors. Our brands have strong penetration of major retail and e-commerce channels and high awareness among pet parents. We believe we will increase our share of the overall pet Rx and OTC medication and health and wellness product markets through our broad network that includes leading U.S. retailers and e-commerce partners. We continue to prioritize building broader consumer awareness, converting more pet owners to use products we manufacture or distribute and providing excellent value. In addition, our retail and e-commerce partners continue to see that our proprietary manufactured products bring significant value to their pet health and wellness category sales and profit. We build and maintain awareness among pet owners that our proprietary manufactured products offer the same quality and active ingredients at a significant savings versus national veterinary brands.
Increase Volume of Products with Existing Retailer & E-commerce Partners. A majority of leading U.S. retailers and e-commerce partners purchase and resell our core product offerings. We believe our net sales and profitability will continue to grow as we expand the number of products we have available for sale through these channels. We invest in research and development to support our own proprietary manufactured products that we expect to help us expand SKU placement within existing accounts and new accounts. Additionally, we believe we are positioned to gain additional item placement and distribution in retail partners where we are also operating our veterinarian clinics.
Provide Veterinarian Services in Conjunction with our Retail Partners. Through our Services segment, we participate in the veterinary services industry, which grew from $28.5 billion in 2018 to $36.8 billion in 2022, according to Packaged Facts. This growth equally reflects increased consumer focus on pet health and wellness. We provide a comprehensive suite of services at over 2,600 community clinic locations and wellness centers hosted at retailers across 39 states. We offer diagnostic tests, vaccinations, prescription medications, microchipping, grooming and hygiene and wellness checks.
Our Products
Through our Products segment, we are a manufacturer and distributor of pet medication and health and wellness products to major U.S. retail and e-commerce channels. We focus our offerings on innovative, proprietary value-branded products, and leading third-party branded products for dogs and cats, including pet Rx medications, OTC medications, and wellness products. We offer and supply these products to customers primarily in the United States.
Rx Medications
The Rx pet medications we sell include flea and tick control, heartworm preventatives, arthritis, thyroid, diabetes and pain treatments, antibiotics and other specialty medications, all of which require a prescription from a veterinarian.
We sell over 350 stock keeping units ("SKUs") of the most popular pet Rx medications to retailers and e-commerce partners, in multiple formats, that previously had been available primarily through the veterinarian channel. Our retail and e-commerce partners then sell these pet Rx medications to pet owners who have a prescription. We source these pet Rx medications directly from manufacturers or through licensed distributors. Several of the top-selling Rx pet medications that we distribute include Nexgard®, Heartgard® Plus and Vetmedin®.
OTC Medications
The pet OTC medications we sell are primarily within the flea and tick control, dog dewormers, along with behavior management categories and do not require a prescription from a veterinarian. These products are available in multiple forms that consumers choose between, such as spot on (topical) treatments, chewables, oral tablets and collars. We develop and manufacture our own propriety value-branded products and distribute well-known leading third-party branded OTC medications.
We sell over 320 SKUs of the leading OTC-branded and value-branded medications within the animal health OTC category to U.S. retail and e-commerce channels. Most of our manufactured OTC medication volume is represented by PetArmor®, Capstar®, Nextstar®, Sentry® and Sergeants® brands and we have manufacturing capabilities to produce multiple product forms within flea and tick control.
Health and Wellness Products
Our health and wellness products include dog and cat treats, oral health solutions, stain and odor treatments and pet nutritional supplements. We manufacture and distribute more than 400 SKUs of proprietary wellness products for dogs and cats, mainly under our PetArmor®, Rocco & Roxie®, VetIQ®, Minties® and Sentry® brands.
Our Services
Our Services segment consists of veterinary services, and related product sales, provided by the Company directly to consumers. We operate our national veterinarian service platform through community clinics, or pop-up locations, and wellness centers, or permanent locations that provide cost effective and convenient veterinary wellness services, including diagnostic tests, vaccinations, prescription medications, microchipping, grooming and hygiene and wellness checks. In 2023, we operated over 2,600 community clinic locations and wellness centers hosted at retailers across 39 states. In addition, in 2023, we collaborated with Walmart, an existing partner, on a new, pilot wellness center to offer a variety of pet services, including veterinary care, grooming and hygiene care.
Seasonality
Products
While many of our products are sold consistently throughout the year, we experience seasonality in the form of increased demand for our flea and tick products in the spring and summer. Additionally, we may experience fluctuations in net sales related to the inventory management strategies of our retail customers.
Services
Similarly, the practice of veterinary medicine is subject to seasonal fluctuation. In particular, demand for veterinary services is significantly higher during the warmer months as pets and pet parents tend to be more active and outdoors and there are more fleas, ticks, and mosquitos during these months and products and services sold to prevent or treat illness or diseases related to these insects.
Product Innovation
We sell a broad portfolio of pet medications and health and wellness products to our retail and e-commerce customers, including an array of products that we develop, manufacture and distribute. To continue to grow our OTC medications and other health and wellness product offerings, we invest in research and development on an ongoing basis. We use a combination of in-house specialists, third-party consultants and animal health research and development experts to expand our proprietary value-branded portfolio and develop next-generation versions of our existing pet products and secure patent protection for these innovations where doing so will allow us to maintain a competitive advantage in the market.
In addition, we have harnessed our position to emerge as an attractive partner for outside research and development researchers and entrepreneurs developing and manufacturing new products and technologies in the strategic pet health and wellness field. We believe these scientists and entrepreneurs seek out our partnership on innovative products given our experience in proprietary value-branded manufacturing and relationships with key retail and e-commerce channel contacts. Our process of assessing partnerships with any outside research and development opportunity includes performing our own internal research and development review, testing and quality control procedures.
Product Sales Channels
Traditional industry sales channels for pet Rx medications, OTC medications, and other health and wellness products include sales through the veterinarian, retail and e-commerce channels. In recent years the retail and e-commerce channels have become intertwined with brick and mortar retailers expanding their online presence and online retailers opening brick and mortar stores.
Historically, non-prescription pet Rx and flea and tick medications have been sold through veterinarian offices and, to a lesser extent, e-commerce. We have focused on making these products, as well as our proprietary value-branded products, available directly to consumers through retail and e-commerce partners, which offer consumers convenient access to these products at lower prices. Our sales channels are primarily concentrated in five sub-channels of retail: (i) food, drug and mass market sales (e.g., Kroger, Target and Walmart); (ii) club stores (e.g., BJ’s Wholesale Club, Costco Wholesale and Sam's Club); (iii) pet specialty stores (e.g., Petco, PetSmart and independent pet stores); (iv) e-commerce (e.g., Amazon.com and Chewy.com); and (v) independent pharmacies and pharmacy distributors. We believe we are a key participant in the sales growth of pet medication products to the retail channel, with the additional benefit of having access to the veterinary channel through solid relationships with established distributors.
Customers
Products
Approximately 99% of our 2023 and 2022 net sales were generated from customers located in the United States and Canada. Our Products segment customers are primarily national store chains, e-commerce, and national pet superstore chains, such as Amazon, Chewy.com, Costco, Kroger, Petco, PetSmart, Sam's Club, Target, The Tractor Supply Company, and Walmart. We supply each of these customers on a national basis. Our largest retail customers in 2023 were Chewy.com and Amazon, which represented 28% and 15%, respectively, of our net sales. Our largest retail customers in 2022 were Chewy.com and Walmart, which represented 25% and 10%, respectively, of our net sales.
Services
Our Services segment customers are consumers purchasing veterinary and related services and products directly from us through one of our community clinics or wellness centers.
Supply Chain
Proprietary Value-Branded Products
None of our suppliers for our proprietary value-branded products are individually significant. We believe there is ample available capacity, including of active pharmaceutical ingredients, for our value-branded products, including at contract manufacturing organizations around the world. Our proprietary value-branded products are currently manufactured by us at our facilities in Omaha, Nebraska and Springville, Utah and through a network of manufacturing facilities owned and operated by contract manufacturing partners across the United States and in Europe. We expect that the combined capacities of our facilities and those of our contract manufacturing partners will meet our forecasted needs for our proprietary value-branded products for the foreseeable future.
Distributed Products
We purchase branded and other products that we distribute, but do not manufacture, from a variety of sources in the United States and Europe, including certain manufacturers and licensed distributors. We believe that having strong relationships with our suppliers will ensure the availability of an adequate volume of products ordered by our retail customers and will enable us to provide more and better product information.
Fulfillment, Warehousing and Shipping
To accomplish efficient fulfillment for pet Rx medication products across the United States into retail, we utilize our established medication distribution channels with our distribution partner, Anda, Inc. We have a multi-year contract with Anda, Inc., which automatically renews for successive two year terms.
For most products, our in-house fulfillment and distribution operations manage the entire supply chain, beginning with the placement of the order, continuing through order processing and then fulfilling and shipping to the customer. All customer orders are processed by our customer service team. We store product inventory and fill most customer orders from our distribution centers in Daytona Beach, Florida, Omaha, Nebraska and Springville, Utah. We also use third-party warehouse providers to fulfill a small portion of our orders. We ship our products using common carriers.
Product Quality and Safety
We believe that product safety and quality are critical. We have developed, implemented and enforce a robust regulatory compliance, product safety and quality program. We have established critical control points throughout the entire supply chain from ingredient sourcing to finished goods to ensure compliance with our quality program.
The food safety program at our Utah plant, where our pet treats are made, is certified at Safe Quality Food (“SQF”) Level II (Food Safety) under Global Food Safety Initiative Benchmarks. To achieve this qualification level, our Utah facility has been built to comply with particular food safety specifications and allows for correct airflow to prevent cross-contamination, among other things. This qualification level also requires us to have certain standard operating procedures in place written to SQF code specifications, hold regular training seminars for manufacturing employees and maintain reporting documentation evidencing compliance with such standard operating procedures.
In addition, our food safety and quality program includes strict guidelines for incoming ingredients, batching, processing, storing, handling, packaging and finished goods. As part of our focus on food safety and quality, we have implemented batch and lot traceability controls across our manufacturing network, including at our manufacturing facilities, where such controls have been implemented into our enterprise resource planning system. These controls allow us to track and tie discreet, inbound raw material components through the manufacturing process to the ultimate finished product, allowing us to maintain and control all finished product lot details and quickly access process manufacturing details.
At the Florida facility where our Rx and some OTC medications are held for distribution, we maintain a Veterinary Prescription Drug Wholesale Distributor license with the State of Florida Department of Business and Professional Regulation, which is the same government entity that regulates distribution facilities for human medications. In connection with our maintenance of this license, the State of Florida conducts random inspections of our facility. To pass these inspections, we must demonstrate safety compliance at the highest standard, including maintaining correct plant temperatures and environmental controls.
As described above, we use contract manufacturers to produce certain of our proprietary value-branded products. To ensure product quality, consistency and safety standards, we actively monitor each contract manufacturer’s operations through the standard operating procedures and facility audits.
At our Omaha location U.S. Environmental Protection Agency ("EPA") and U.S. Food and Drug Administration ("FDA") regulated products are produced, packaged, and distributed from our nearby distribution center. Products include dog and cat flea and tick spot-on, shampoo, collars, toothpaste and hairball paste. We have a robust quality management program that includes quality processes for incoming inspection, manufacturing and packaging inspections, supplier quality, change control, deviations, and corrective and preventative actions. We manage customer interaction through our call centers and social media to ensure that products maintain the highest quality. All call data is tracked, trended and reviewed for signals that may indicate product quality issues. The Omaha site is inspected several times annually by external auditors and we perform annual internal audits and mock recalls. We have received high marks and consistently maintain compliance with current good manufacturing practices and retain certifications as required.
All of our contract manufacturing facilities are required to have written quality control standard operating procedures. We require our contract manufacturing facilities to maintain third-party certifications and pass our own quality system and safety audits, and for FDA-regulated products, to comply with the Good Manufacturing Practices of the FDA. Third-party certifications provide an independent and external assessment that a product and/or process complies with applicable safety regulations and standards. In addition, our quality control team conducts reviews of all aspects of our supply chain to ensure that ingredients, finished goods and manufacturing processes meet our strict safety and quality requirements and that all of our ingredients are rigorously tested prior to being used in our products.
We maintain a customer service line and have trained representatives to assist customers. Any call reporting an adverse event relating to our products is addressed by our third-party vendor, SafetyCall, through its own on-site veterinarians. On
a quarterly basis, we submit filings in accordance with the EPA specifications reporting any adverse event associated with our flea and tick products.
Marketing and Advertising
Our marketing strategy largely focuses on building awareness and educating pet owners about our various brands and products. To accomplish this goal, we use a combination of television, digital marketing (e.g. search, display ads, email), social media marketing and in-store displays and promotions. Our marketing message highlights the quality and cost-savings our products offer customers such as our proprietary, value-branded flea and tick products that contain the same active ingredients as leading brands at lower prices.
Competition
Products
The pet medication, snack, food and health and wellness industry is highly competitive. We compete on the basis of product quality, product availability, palatability, loyalty and trust, product variety and ingredients, product packaging and design, shelf space, reputation and brand, price point and promotional efforts. We compete directly and indirectly with both manufacturers and distributors of pet medication and health and wellness products and online distributors, as well as with veterinarians. We face competition from companies that distribute various pet medications to traditional retailers and pet health and wellness products companies such as Elanco (formerly Bayer AG), Hartz (Unicharm Corp.), Mars, Inc. (“Mars”), Manna Pro, Nestlé S.A., Spectrum Holdings, Promika LLC, Tevra Brands, and The J.M. Smucker Company, most of which are larger than we are and have greater financial resources. Similarly, we face intense competition from manufacturers who sell pet medications and pet health and wellness products to e-commerce and other retailers and to veterinarians, who compete directly with our retailers to offer consumers pet flea and tick and other pet health and wellness products.
Our retail customers compete with veterinarians for the sale of pet Rx and OTC pet medications and other health and wellness products. Many pet owners may prefer the convenience of purchasing their pet medications or other health products during a veterinarian visit. In order to effectively compete with veterinarians, we and retail partners must continue to price competitively and to educate pet owners about the product availability, service and savings offered by purchasing pet medications and other health products in their retail stores or from their websites.
Services
Within our Services segment, we compete directly with veterinarians. Our primary competitors for our veterinary clinics in most markets are individual practitioners or small, regional multi-clinic practices. In addition, some national companies such as Banfield Pet Hospitals, VCA Animal Hospitals, or Petco are developing or have developed networks of veterinary clinics or hospitals in markets in which we currently operate.
Our Trademarks and Other Intellectual Property
We believe that our intellectual property is valuable and has contributed to the success of our business. Our primary trademarks include “PetIQ,” “PetArmor,” “VIP Petcare,” “VetIQ PetCare,” “VetIQ,” “Capstar,” “Advecta,” “SENTRY,” “Sergeants,” “PurLuv,” “Rocco & Roxie,” and “Minties” all of which are registered with the U.S. Patent and Trademark Office. We also have numerous other trademark registrations and pending applications, in the U.S., Canada, China, Europe, and other countries, for product names that are central to our branding. Our trademarks are assets that reinforce our brand, our sub-brands and consumers perception of our products. The current registrations of these trademarks in the U.S. and foreign countries are effective for varying periods of time and may be renewed periodically, provided that we, as the registered owner, or our licensees where applicable, comply with all applicable renewal requirements including, where necessary, the continued use of the trademarks in connection with the goods or services identified in the applicable registrations. In addition to trademark protection, we own numerous URL designations, including www.petarmor.com, www.vetiqpetcare.com, www.vippetcare.com, www.petvet.vippetcare.com, www.vetiq.com, www.advecta.com, www.sentrypetcare.com, www.sergeants.com, www.delightibles.com and www.mintiestreats.com, which are important to the successful implementation of our marketing and advertising strategy. We also own patents and have pending patent applications for products, formulas, ingredient combinations and packaging that we consider important to our business. Including various methods of use, interomone, pheromone compositions and spot-on pesticide compositions. We rely on and carefully protect unpatented proprietary expertise, recipes and formulations, continuing innovation and other trade
secrets to develop and maintain our competitive position. The contents of our websites are not intended to be incorporated by reference into this Annual Report on Form 10-K or in any other report or document we file with the Securities and Exchange Commission ("SEC"), and any references to our websites are intended to be inactive textual references only.
Government Regulation
Along with our contract manufacturers, ingredient and packaging suppliers and third-party shipping providers, we are subject to a broad range of laws and regulations, both in the U.S. and elsewhere, intended to protect public health and safety, natural resources and the environment. Our products and operations in the U.S. are subject to regulation by the FDA, the EPA, the Florida Department of Health and the U.S. Department of Agriculture ("USDA") and by various other federal, state, local and foreign authorities regarding the registration, manufacturing, processing, packaging, storage, distribution, advertising, labeling and export of our products, including drug and food safety standards.
All Rx animal drugs are required to be approved by the FDA through either a New Animal Drug Application or, in the case of generic Rx animal drugs, an Abbreviated New Animal Drug Application (“ANADA”). Two of our proprietary value-branded products, TruProfen and Heart Shield Plus, have been approved by the FDA under ANADAs submitted to the FDA by third parties. We have agreements with these third parties that hold approved ANADAs to private label or proprietary value-branded products under such ANADAs. However, the third parties that hold the ANADAs are ultimately responsible for compliance with regulatory obligations associated with these products.
In addition, our foreign subsidiaries are subject to the laws of the United Kingdom, the Republic of Ireland and the European Union, as well as provincial and local regulations.
Under various statutes and regulations, these agencies and authorities, among other things, (i) prescribe the requirements for registration and establish the standards for quality and safety, (ii) regulate our marketing, advertising and sales to consumers and (iii) control the importing and exporting of our products. Some of these agencies, in certain circumstances, must not only approve our products, but also review the manufacturing processes and facilities used to produce these products before they can be marketed in the United States and elsewhere. In particular, certain of our pet products require EPA or FDA approval prior to marketing. To market such a regulated pet product, the regulatory agency must approve a new product, supported by data from animal safety and effectiveness studies that adequately demonstrate the safety and efficacy of that product in the target animal for the intended indication; or, in the case of generic versions of previously approved reference-listed pet products, the regulatory agency, supported by data to demonstrate, among other things, that the proposed generic product has the same active ingredients in the same concentration as the reference-listed product and is bioequivalent to the reference listed product. After approval, manufacturers are required to collect reports of adverse events and submit them on a regular basis to either the EPA or FDA. Some of the approved products we distribute are held by third parties with whom we contract to distribute those products under our own label.
We are subject to labor and employment laws, safety and health regulations and other laws, including those promulgated by the EPA and the National Labor Relations Board. Our operations, and those of our contract manufacturers, ingredient and packaging suppliers and third-party shipping providers, are subject to various laws and regulations relating to worker health and safety matters as well as environmental and natural resource protection, including the availability and use of pesticides, emissions and discharges to the environment, and the treatment, handling, storage and disposal of materials and wastes. We monitor changes in these laws and believe that we are in material compliance with applicable laws and regulations. No assurance can be given, however, that material costs and liabilities will not arise in the future, such as due to a change in the law or the discovery of currently unknown conditions.
Certain states have laws, rules and regulations which require that veterinary medical practices be either wholly-owned or majority-owned by licensed veterinarians and that corporations which are not wholly-owned or majority-owned by licensed veterinarians refrain from providing, or holding themselves out as providers of, veterinary medical care. In these states and provinces, we provide management and other administrative services to veterinary practices rather than owning such practices or providing such care. In some cases, in addition to providing management and administrative services we may lease the veterinary facility and equipment to the veterinary practice. Although we have structured our operations to comply with our understanding of the veterinary medicine laws of each state and province in which we operate, interpretive legal precedent and regulatory guidance varies by jurisdiction and is often sparse and not fully developed.
In addition, all of the states in which we operate impose various registration permit and/or licensing requirements. To fulfill these requirements, we have registered each of our facilities with appropriate governmental agencies and, where required,
have appointed a licensed veterinarian to act on behalf of each facility. All veterinarians practicing in our animal wellness centers are required to maintain valid state licenses to practice.
Human Capital
We employed approximately 1,933 people as of December 31, 2023, of which 1,890 are employed within the United States. Our workforce is comprised of approximately 58% full time and 42% part time employees. Of our total employees, approximately 1,330 of our employees worked in our Services division. In addition, we regularly contract with veterinarians to provide veterinary services in our mobile community clinics and wellness centers. During the year ending December 31, 2023, we engaged approximately 1,444 veterinarians that were independent contractors.
The animal health industry is highly competitive and PetIQ is a fast growing company. PetIQ’s benefit offerings are designed to meet the evolving needs of a diverse workforce. Attraction and retention of key talent is a focal point for the Company. To support these objectives, our human resources programs are designed to reward and support employees through competitive pay and benefits; support and facilitate internal talent mobility; and evolve and invest in technology, tools, and resources to enable employees at work. Some examples of key programs and initiatives that are focused to attract and retain our workforce include:
•Four core values that serve as the foundation for our business: Customer Focused, Adaptive and Agile, Humble and Hungry, and Results Oriented.
•An annual review process to focus on employee skills and career growth and strengthen supervisor-employee relationships.
•Quarterly town hall meetings to invite dialogue among employees and leaders.
•Free mental and behavioral health resources, including on-demand access to an employee assistance program for employees and their dependents.
•Encouraging and supporting renewal of all professional licenses and professional memberships.
•Total rewards to all employees to include competitive pay, various output related bonus plans in both the Services and Product segments, a 401(k) plan with a four percent Company match, paid time off, parental leave, health, vision and dental insurance, and other ancillary benefits.
Our Corporate Information
Our principal executive offices are located at 230 E. Riverside Drive, Eagle, Idaho 83616. Our telephone number is 208-939-8900. The address of our corporate website is www.petiq.com, and our investor relations website is located at http://ir.petiq.com. The contents of our website are not intended to be incorporated by reference into this Annual Report or in any other report or document we file with the SEC, and any references to our websites are intended to be inactive textual references only.
Available Information
Our annual reports on Form 10-K, annual proxy statements and related proxy cards are made available on our website at the same time they are mailed to stockholders. Our quarterly reports on Form 10-Q, periodic reports on Form 8-K and amendments to those reports that we file or furnish pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), are available through our website, free of charge, as soon as reasonably practicable after they have been electronically filed or furnished to the SEC. Our website also provides access to reports filed by our directors, executive officers and certain significant shareholders pursuant to Section 16 of the Exchange Act. In addition, General Code of Ethics and charters for the committees of our board of directors are available on our website as well as other shareholder communications. The information contained in or that can be accessed through our website does not constitute a part of, and is not incorporated by reference into, this Annual Report. The SEC maintains an internet site (http://www.sec.gov) that contains reports, proxy information statements and other information related to issuers that file electronically with the SEC.
Item 1A – Risk Factors
You should carefully consider the risks described below in addition to the other information set forth in this Annual Report. If any of the risks, events, and uncertainties described in the risk factors listed below actually occurs, our business, results of operations, cash flows, and financial condition may be materially and adversely affected. The risk factors listed below are not exhaustive. Additional risks not currently known to us or that we presently deem immaterial may emerge or become
material at any time and may negatively impact our business, reputation, financial condition, results of operations or the trading price of our common stock.
Risks Related to our Business and Industry
Unfavorable economic conditions and the consumer behavior trends they drive could reduce spending on our products and services and limit our ability to grow our business and negatively affect our results of operations.
Our results of operations may be affected by unfavorable changes in the domestic and global economy on us or our customers and potential customers. The keeping of pets and the purchase of pet-related products and services may constitute discretionary spending for some consumers and any material decline in the amount of consumer discretionary spending may reduce overall levels of pet ownership or spending on pets. Unfavorable economic conditions, including conditions resulting from an economic recession in the United States or other major markets, financial and credit market fluctuations, high levels of inflation and/or interest rates, international trade relations, political turmoil, natural catastrophes, outbreaks of contagious diseases, lower corporate earnings, reduction in business confidence and activity, global geopolitical conflicts, and terrorist attacks, could cause a decrease in consumer sentiment, adversely impact our retail customers and suppliers and our community clinics and wellness centers, and negatively affect the growth of our business and our results of operations. Our competitors, many of which are larger and have greater financial resources than we do, may respond to challenging market conditions by lowering prices in an attempt to attract our customers. We cannot predict the timing, strength, or duration of any economic slowdown, instability, or recovery, generally or how any such event may impact our business.
We may seek to grow our business through acquisitions of or investments in new or complementary businesses, facilities, technologies or products, or through strategic alliances, and the failure to manage acquisitions, investments or strategic alliances, or the failure to integrate them with our existing business, could have a material adverse effect on us.
From time to time we have considered and anticipate that in the future we may consider opportunities to acquire or make investments in new or complementary businesses, facilities, technologies or products, or enter into strategic alliances, that may enhance our capabilities, expand our manufacturing network, complement our current products or expand the breadth of our markets. Potential and completed acquisitions and investments and other strategic alliances involve numerous risks, including:
•problems integrating the purchased business, facilities, technologies, products or brands;
•issues maintaining uniform standards, procedures, controls and policies;
•unanticipated costs;
•diversion of management’s attention from our existing business;
•adverse effects on existing business relationships with suppliers, contract manufacturers, and retail customers;
•risks associated with entering new markets in which we have limited or no experience;
•potential loss of key employees of acquired businesses; and
•increased legal and accounting expenses.
We do not know if we will be able to identify acquisitions or strategic relationships we deem suitable, whether we will be able to successfully complete any such transactions on favorable terms or at all or whether we will be able to successfully integrate any acquired business, facilities, technologies or products into our business or retain any key personnel, suppliers or customers. Our ability to successfully grow through strategic transactions depends upon our ability to identify, negotiate, complete and integrate suitable target businesses, facilities, technologies and products and to obtain any necessary financing. These efforts could be expensive and time-consuming and may disrupt our ongoing business and prevent management from focusing on our operations. If we are unable to integrate any acquired businesses, facilities, technologies and products effectively, our business, results of operations and financial condition could be materially adversely affected.
Completed acquisitions may result in additional goodwill and/or an increase in other intangible assets on our balance sheet. We are required annually, or as facts and circumstances exist, to test goodwill and other intangible assets to determine if impairment has occurred. If the testing performed indicates that impairment has occurred, we are required to record a non-cash impairment charge for the difference between the carrying value of the goodwill or other intangible assets and the implied fair value of the goodwill or the fair value of other intangible assets in the period the determination is made. Should the value of goodwill or other intangible assets become impaired, there could be a material adverse effect on our financial condition and results of operations.
We are dependent on a relatively limited number of customers for a significant portion of our net sales.
Our largest retail customers in 2023 were Chewy.com and Amazon, which accounted for 28% and 15% respectively, of our net sales. Our largest retail customers in 2022 were Chewy.com and Walmart, which accounted for 25% and 10%, respectively, of our net sales. No other retail customer has accounted for 10% or more of our net sales during these two years. If we were to lose any of our key retail customers, or if any of our key retail customers reduce the amount of their orders, consolidate, reduce their store footprint, experience financial or operational difficulties or generate less traffic, our business, financial condition and results of operations may be materially adversely affected.
In addition, we generally do not enter into long-term contracts with our retail customers. As a result, we rely on consumers’ continuing demand for our products and our position in the market for all purchase orders. Our customers are sophisticated and have the ability to replace our proprietary value brands with various other supply options if we do not compete aggressively for their business. If our retail customers change their pricing, margin expectations or business terms (including through the imposition of warehouse and other fees), change their business strategies as a result of industry consolidation or otherwise, reduce the number of brands or product lines they carry, decrease their advertising or promotional efforts for, or the amount of shelf space they allocate to, our products or allocate greater shelf space to other products, our net sales could decrease and our business, financial condition and results of operations would be materially adversely affected.
We may not be able to successfully implement our growth strategy in our Products segment on a timely basis or at all.
Our future success depends, in large part, on our ability to implement our growth strategy, including developing and introducing new products, expanding into new markets, attracting new consumers to our brand and sub-brands, improving placement of our products in the stores of our retail customers, and expanding our distribution and online sales through our retail partners. Our ability to implement this growth strategy depends, among other things, on our ability to:
•develop new proprietary value-branded products and product line extensions that appeal to consumers;
•continue to effectively compete in our industry;
•increase our brand and sub-brand recognition by effectively implementing our marketing strategy and advertising initiatives;
•maintain and, to the extent necessary, improve our high standards for product quality, safety and integrity;
•expand and maintain brand and sub-brand loyalty;
•secure shelf space in the stores of our retail customers; and
•enter into distribution and other strategic arrangements with traditional retailers and other potential distributors of our products.
We may not be able to successfully implement our growth strategy and may need to change our strategy in order to maintain our growth. If we fail to implement our growth strategy or if we invest resources in a growth strategy that ultimately proves unsuccessful, our business, financial condition and results of operations would be materially adversely affected.
We may not continue to grow our Services business.
As of December 31, 2023, we had over 2,600 community clinics and 133 wellness centers. Growth in our Services business will require opening new clinics, both wellness centers and mobile clinics, and operating those on a profitable basis. Our ability to open new clinics is dependent upon a number of factors, many of which are beyond our control, including our ability to:
•hire, train, and retain the skilled veterinarians and skilled employees necessary to staff the clinics and wellness centers;
•identify locations and retail partners that can support our wellness centers;
•compete for sites and secure wellness center space in the stores of our retail partners;
•reach acceptable lease or host arrangement terms;
•obtain, in a timely manner and for an acceptable cost, required licenses, permits, and regulatory approvals;
•increase profitability of our mobile clinics and wellness centers;
•respond effectively to any changes in local, state, and federal law and regulations that adversely affect our ability to open new wellness centers or clinics; and
•control construction and other launch costs to open the wellness centers and clinics.
There is no guarantee that we will continue to open wellness centers or community clinics and, if we do so, that we will be profitable. Specifically, during the third quarter of 2023, we implemented a Services segment optimization (the "optimization") to improve the functioning of the Services segment and profitability. The optimization included assessing the operational and financial performance of the Company's wellness centers since re-opening after the pandemic as well as the assessment of the veterinary labor market in each geographic market. The Company also evaluated its ability to potentially convert these locations to a more hygiene-focused offering and determined they would be unable to convert these locations in the future based on the aforementioned assessment and the available square footage within the respective wellness centers. As a result of the optimization, the Company closed 149 wellness centers during the year ended December 31, 2023. Restructuring and related charges attributable to the optimization were $13.6 million recorded on the consolidated statements of operations for the twelve months ended December 31, 2023, approximately $11.0 million of depreciation and amortization as well as $0.9 million inventory valuation adjustments. Our ability to improve profitability and achieve the anticipated cost savings from the optimization, as well as our ability to reinvest those cost savings into other areas of our business, is subject to many estimates and assumptions, some of which are beyond our control. If our estimates and assumptions are incorrect or if other unforeseen events occur, we may not achieve the benefits of the optimization and our business and results of operations could be adversely affected.
We may experience difficulties hiring skilled veterinarians due to shortages that could disrupt our business.
From time to time we may experience shortages of skilled veterinarians in markets in which we operate mobile clinics and wellness centers, which may require us to enhance wages and benefits to recruit and retain enough qualified veterinarians to adequately staff mobile clinics and wellness centers. If we are unable to recruit and retain qualified veterinarians, or to control our labor costs, our business, financial conditions and results of operations may be materially adversely affected.
We operate in a highly competitive industry and may lose market share or experience margin erosion if we are unable to compete effectively.
The pet medication, snack, food and health and wellness industry is highly competitive. We compete on the basis of product quality, product availability, palatability, loyalty and trust, product variety and ingredients, product packaging and design, shelf space, reputation and brand, price point and promotional efforts. We compete directly and indirectly with both manufacturers and distributors of pet medication and health and wellness products and online distributors, as well as with veterinarians. We face competition from companies that distribute various pet medications to traditional retailers and pet health and wellness products companies such as Elanco (formerly Bayer AG), Hartz (Unicharm Corp.), Mars, Inc. (“Mars”), Manna Pro, Nestlé S.A., Spectrum Holdings, Promika LLC, Tevra Brands, and The J.M. Smucker Company, most of which are larger than we are and have greater financial resources. Similarly, we face intense competition from manufacturers who sell pet medications and pet health and wellness products to e-commerce and other retailers and to veterinarians, who compete directly with our retailers to offer consumers pet flea and tick and other pet health and wellness products.
Our retail customers compete with veterinarians for the sale of pet Rx and OTC pet medications and other health and wellness products. Many pet owners may prefer the convenience of purchasing their pet medications or other health products during a veterinarian visit. In order to effectively compete with veterinarians, we and retail partners must continue to price competitively and to educate pet owners about the product availability, service and savings offered by purchasing pet medications and other health products in their retail stores or from their websites.
Within our Services segment, we compete directly with veterinarians. Our primary competitors for our veterinary clinics in most markets are individual practitioners or small, regional multi-clinic practices. In addition, some national companies such as Banfield Pet Hospitals, VCA Animal Hospitals, or Petco are developing or have developed networks of veterinary clinics or hospitals in markets in which we currently operate.
These competitors may be able to identify and adapt to changes in consumer preferences more quickly than us due to their resources and scale. They may also be more successful in marketing and selling their products, or services, better able to increase prices to reflect cost pressures and better able to increase their promotional activity, which may impact us and the entire pet health and wellness industry. If these or other competitive pressures cause our products to lose market share or
experience margin erosion, our business, financial condition and results of operations would be materially adversely affected.
Resistance from veterinarians to authorize prescriptions, or attempts/efforts on their part to discourage pet owners to purchase from retailers and pharmacies could cause our net sales to decrease and could materially adversely affect our financial condition and results of operations.
Since we began our operations, some veterinarians have resisted providing, or simply refuse to provide, pet owners with a copy of their pet’s prescription or authorizing the prescription to an outside pharmacy, thereby effectively preventing outside pharmacies from filling such prescriptions under state law. We have also been informed by customers and consumers that veterinarians on certain occasions have tried to discourage pet owners from purchasing from the retail channel. If the number of veterinarians who refuse to authorize prescriptions should increase, or if veterinarians are successful in discouraging pet owners from purchasing from outside retailers and pharmacies, our net sales could decrease and our financial condition and results of operations would be materially adversely affected.
Any damage to our reputation or our brand or sub-brands may materially adversely affect our business, financial condition and results of operations.
Maintaining, developing and expanding our reputation with consumers, our retail customers and our suppliers is critical to our success. Our brand and sub-brands may suffer if our marketing plans or product initiatives are not successful. The importance of our brand and sub-brands may decrease if competitors offer more products with formulations similar to the products that we manufacture. Further, our brand and sub-brands may be negatively impacted due to real or perceived quality issues or if consumers perceive us as being untruthful in our marketing and advertising, even if such perceptions are not accurate. Product contamination, the failure to maintain high standards for product quality, safety and integrity, including raw materials and ingredients obtained from suppliers, or allegations of product quality issues, mislabeling or contamination, even if untrue or caused by our contract manufacturing partners or raw material suppliers, may reduce demand for our products or cause production and delivery disruptions. We maintain guidelines and procedures to ensure the quality, safety and integrity of our products. However, we may be unable to detect or prevent product and/or ingredient quality issues, mislabeling or contamination, particularly in instances of fraud or attempts to cover up or obscure deviations from our guidelines and procedures. If any of our products become unfit for consumption, cause injury or are mislabeled, we may have to engage in a product recall and/or be subject to liability. Damage to our reputation or our brand or sub-brands or loss of consumer confidence in our products for any of these or other reasons could result in decreased demand for our products and our business, financial condition and results of operations may be materially adversely affected.
Our growth and business are dependent on trends that may change, and our historical growth may not be indicative of our future growth.
The growth of our business depends primarily on the continued shift from consumers purchasing pet health and wellness products from veterinarians to purchasing such products through traditional retail channels, growth of the pet health and wellness products market and popularity of pet ownership, transitions from traditional veterinarians to mobile clinics and wellness centers, as well as on general economic conditions. These trends may not continue or may change. In the event of a decline in consumers purchasing pet health and wellness products through traditional retail channels, a change in pet health and wellness trends or a decrease in the overall number of pets, or during challenging economic times, we may be unable to persuade our retail customers and consumers to purchase our products, and our business, financial condition and results of operations would be materially adversely affected and our growth rate may slow or stop.
In addition, our historical growth has placed and, if continued, may continue to place significant demands on our management and our operational and financial resources. Our organizational structure may become more complex as we add additional staff, and we would likely require more resources to grow and continue to improve our operational, management and financial controls. If we are not able to manage our growth effectively, our business, financial condition and results of operations would be materially adversely affected.
Our business depends, in part, on the sufficiency and effectiveness of our marketing and trade promotion programs and incentives.
Due to the competitive nature of our industry, we must effectively and efficiently promote and market our products and services through television, internet and print advertisements as well as through trade promotions and incentives to sustain and improve our competitive position in our market. Marketing investments may be costly. In addition, we may, from time
to time, change our marketing strategies and spending, including the timing or nature of our trade promotions and incentives. We may also change our marketing strategies and spending in response to actions by our customers, competitors and other companies that manufacture and/or distribute pet health and wellness products or provide health and wellness services. The sufficiency and effectiveness of our marketing and trade promotions and incentives are important to our ability to retain and improve our market share and margins. If our marketing and trade promotions and incentives are not successful or if we fail to implement sufficient and effective marketing and trade promotions and incentives or adequately respond to changes in industry marketing strategies, our business, financial condition and results of operations would be adversely affected.
To the extent our retail customers purchase products in excess of consumer consumption in any period, our net sales in a subsequent period may be adversely affected as our retail customers seek to reduce their inventory levels.
From time to time, our retail customers may purchase more products than they expect to sell to consumers during a particular time period. Our retail customers may grow their inventory in anticipation of, or during, our promotional events, which typically provide for reduced prices during a specified time or other incentives. Our retail customers may also increase inventory in anticipation of a price increase for our products, or otherwise over-order our products as a result of overestimating demand for our products. If a retail customer increases its inventory during a particular reporting period as a result of a promotional event, anticipated price increase or otherwise, then our net sales during the subsequent reporting period may be adversely impacted as our retail customers seek to reduce their inventory to customary levels. This effect may be particularly pronounced when the promotional event, price increase or other event occurs near the end or beginning of a reporting period or when there are changes in the timing of a promotional event, price increase or similar event, as compared to the prior year. To the extent our retail customers seek to reduce their usual or customary inventory levels or change their practices regarding purchases in excess of consumer consumption, our net sales and results of operations would be materially adversely affected in that or subsequent periods.
We may not be able to manage our manufacturing and supply chain effectively, which may adversely affect our results of operations.
We must accurately forecast demand for all of our products in order to ensure that we have enough products available to meet the needs of our retail customers. Our forecasts are based on multiple assumptions that may cause our estimates to be inaccurate and affect our ability to obtain adequate manufacturing capacity (whether our own manufacturing capacity or contract manufacturing capacity) in order to meet the demand for our proprietary value-branded products, which could prevent us from meeting increased retail customer or consumer demand and harm our brand, our sub-brands and our business. If we do not accurately align our manufacturing capabilities with demand, our business, financial condition and results of operations may be materially adversely affected.
If for any reason we were to change any one of our contract manufacturers, we could face difficulties that might adversely affect our ability to maintain an adequate supply of our proprietary value-branded products, and we would incur costs and expend resources in the course of making the change. Moreover, we might not be able to obtain terms as favorable as those received from our current contract manufacturers, which in turn would increase our costs.
In addition, we must continuously monitor our inventory and product mix against forecasted demand. If we underestimate demand, we risk having inadequate supplies. We also face the risk of having too much inventory on hand that may reach its expiration date and become unsalable, and we may be forced to rely on markdowns or promotional sales to dispose of excess or slow-moving inventory. If we are unable to manage our supply chain effectively, our operating costs could increase and our profit margins could decrease.
We rely on third parties to provide us with materials and services, and are subject to increased labor and material costs and potential disruptions in supply.
The materials used to manufacture our products may be subject to availability constraints and price volatility caused by changes in demand, weather conditions, supply conditions, government regulations, economic climate, disease outbreaks and other factors. In addition, labor costs may be subject to volatility caused by the supply of labor, governmental regulations, economic climate and other factors. Increases in the demand for, availability or the price of, materials used to manufacture our products and increases in labor costs could increase the costs to manufacture our products, result in product delivery delays or shortages, and impact our ability to launch new products on a timely basis or at all. We may not be able to pass all or a material portion of any higher material or labor costs on to our customers, which could materially adversely affect our operating results and financial condition.
Certain third-party suppliers are the sole or exclusive source of certain materials and services necessary for production of our products. We may be unable to meet demand for certain of our products if any of our third-party suppliers cease or interrupt operations, fail to renew contracts with us or otherwise fail to meet their obligations to us.
Shipping is a critical part of our business and any changes in, or disruptions to, our shipping arrangements could adversely affect our business, financial condition, and results of operations.
We currently rely on third-party national and regional logistics providers to deliver products to our manufacturing and distribution warehouses from our third-party suppliers and contract manufacturers and to deliver products from our manufacturing and distribution warehouses to our retail customers. If we are not able to negotiate acceptable pricing and other terms with these providers, or if these providers experience performance problems or other difficulties in processing our orders or delivering our products, it could negatively impact our results of operations and our customers’ experience. For example, changes to the terms of our shipping arrangements may adversely impact our margins and profitability. In addition, our ability to receive inbound inventory efficiently and ship merchandise to our retail customers may be negatively affected by factors beyond our and these providers’ control, including inclement weather, fire, flood, power loss, earthquakes, acts of war or terrorism or other events specifically impacting our or other shipping partners, such as labor disputes, financial difficulties, system failures and other disruptions to the operations of the shipping companies on which we rely. We are also subject to risks of damage or loss during delivery by our shipping vendors. If any of the foregoing occurs, our business, financial condition and results of operations may be materially adversely affected.
The growth of our business depends in part on our ability to accurately predict consumer trends, successfully introduce new products and improve existing products, and expand into new offerings.
Our growth depends, in part, on our ability to successfully introduce new products, including our manufactured products, and improve and reposition our existing products to meet the requirements of our retail partners and those of pet parents. This, in turn, depends on our ability to predict and respond to evolving consumer trends, demands and preferences. The success of our innovation and product development efforts is affected by the technical capability of our product development staff and third-party consultants in developing and testing new products, including complying with governmental regulations, our attractiveness as a partner for outside research and development scientists and entrepreneurs and the success of our management and sales team in introducing and marketing new products.
We may be unable to determine with accuracy when or whether any of our products now under development will be approved or launched, and we may be unable to develop or otherwise acquire product candidates or products. Additionally, we cannot predict whether any such products, once launched, will be commercially successful. Furthermore, the timing and cost of our R&D initiatives may increase as a result of additional government regulation or otherwise, making it more time-consuming and/or costly to research, test and develop new products. If we are unable to successfully develop or otherwise acquire new products, our financial condition and results of operations may be materially adversely affected.
We are subject to stringent and evolving U.S. and foreign laws, regulations, and rules, contractual obligations, industry standards, policies and other obligations related to data privacy and security. Our actual or perceived failure to comply with such obligations could lead to regulatory investigations or actions; litigation (including class claims) and mass arbitration demands; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; and other adverse business consequences.
In the ordinary course of business, we collect, receive, store, process, generate, use, transfer, disclose, make accessible, protect, secure, dispose of, transmit, and share (collectively, process) personal data and other sensitive information, including proprietary and confidential business data, trade secrets, intellectual property, business plans, transactions, and financial information (collectively, sensitive data).
Our data processing activities subject us to numerous data privacy and security obligations, such as various laws, regulations, guidance, industry standards, external and internal privacy and security policies, contractual requirements, and other obligations relating to data privacy and security.
In the United States, federal, state, and local governments have enacted numerous data privacy and security laws, including data breach notification laws, personal data privacy laws, consumer protection laws (e.g., Section 5 of the Federal Trade Commission Act), and other similar laws (e.g., wiretapping laws). In the past few years, numerous U.S. states—including California, Virginia, Colorado, Connecticut, and Utah—have enacted comprehensive privacy laws that impose certain obligations on covered businesses, including providing specific disclosures in privacy notices and affording residents with
certain rights concerning their personal data. These state laws allow for statutory fines for noncompliance. For example, the California Consumer Privacy Act of 2018, as amended by the California Privacy Rights Act of 2020 (“CPRA”) (collectively, “CCPA”), applies to personal data of consumers, business representatives, and employees who are California residents, and requires businesses to provide specific disclosures in privacy notices and honor requests of such individuals to exercise certain privacy rights. The CCPA provides for fines of up to $7,500 per intentional violation and allows private litigants affected by certain data breaches to recover significant statutory damages. Similar laws are being considered in several other states, as well as at the federal and local levels, and we expect more states to pass similar laws in the future. These developments may further complicate compliance efforts and increase legal risk and compliance costs for us and the third parties upon whom we rely.
Outside the United States, an increasing number of laws, regulations, and industry standards may govern data privacy and security. For example, the European Union’s General Data Protection Regulation (“EU GDPR”) and the United Kingdom’s GDPR (“UK GDPR”) impose strict requirements for processing personal data. For example, under the EU GDPR and the UK GDPR, companies may face temporary or definitive bans on data processing and other corrective actions; fines of up to 20 million Euros under the EU GDPR, 17.5 million pounds sterling under the UK GDPR or, in each case, 4% of annual global revenue, whichever is greater; or private litigation related to processing of personal data brought by classes of data subjects or consumer protection organizations authorized at law to represent their interests. In addition, we may be unable to transfer personal data from Europe and other jurisdictions to the United States or other countries due to foreign data localization requirements or limitations on cross-border data flows.
Our employees and personnel may use generative artificial intelligence (“AI”) technologies to perform portions of their work, and the disclosure and use of personal data in generative AI technologies is subject to various privacy laws and other privacy obligations. Governments have passed and are likely to pass additional laws regulating generative AI. Our use of this technology could result in additional compliance costs, regulatory investigations and actions, and lawsuits.
In addition to data privacy and security laws, we are bound by other contractual obligations related to data privacy and security, and our efforts to comply with such obligations may not be successful. We also publish privacy policies, marketing materials, and other statements regarding data privacy and security and if these policies, materials, or statements are found to be deficient, lacking in transparency, deceptive, unfair, or misrepresentative of our practices, we may be subject to investigation, enforcement actions by regulators, or other adverse consequences.
Obligations related to data privacy and security (and consumers’ data privacy expectations) are quickly changing, becoming increasingly stringent, and creating uncertainty. Additionally, these obligations may be subject to differing applications and interpretations, which may be inconsistent or conflict among jurisdictions. Preparing for and complying with these obligations requires us to devote significant resources and may necessitate changes to our services, information technologies, systems, and practices and to those of any third parties that process personal data on our behalf.
We may at times fail (or be perceived to have failed) in our efforts to comply with our data privacy and security obligations. Moreover, despite our efforts, our personnel or third parties on whom we rely may fail to comply with such obligations, which could negatively impact our business operations. If we or the third parties on which we rely fail, or are perceived to have failed, to address or comply with applicable data privacy and security obligations, we could face significant consequences, including but not limited to: government enforcement actions (e.g., investigations, fines, penalties, audits, inspections, and similar); litigation (including class-action claims) and mass arbitration demands; additional reporting requirements and/or oversight; bans on processing personal data; and orders to destroy or not use personal data.
If our information technology systems or those of third parties upon which we rely, or our data are or were compromised, we could experience adverse consequences resulting from such compromise, including but not limited to regulatory investigations or actions; litigation; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; and other adverse consequences.
In the ordinary course of our business, we and the third parties upon which we rely process sensitive data, and, as a result, we and the third parties upon which we rely face a variety of evolving threats that could cause security incidents. Cyber-attacks, malicious internet-based activity, online and offline fraud, and other similar activities threaten the confidentiality, integrity, and availability of our sensitive data and information technology systems, and those of the third parties upon which we rely. Such threats are prevalent and continue to rise, are increasingly difficult to detect, and come from a variety
of sources, including traditional computer “hackers,” threat actors, “hacktivists,” organized criminal threat actors, personnel (such as through theft or misuse), sophisticated nation states, and nation-state-supported actors.
Some actors now engage and are expected to continue to engage in cyber-attacks, including without limitation nation-state actors for geopolitical reasons and in conjunction with military conflicts and defense activities. During times of war and other major conflicts, we and the third parties upon which we rely may be vulnerable to a heightened risk of these attacks, including retaliatory cyber-attacks, that could materially disrupt our systems and operations, supply chain, and ability to produce, sell and distribute our products and services.
We and the third parties upon which we rely are subject to a variety of evolving threats, including but not limited to social-engineering attacks (including through deep fakes, which may be increasingly more difficult to identify as fake, and phishing attacks), malicious code (such as viruses and worms), malware (including as a result of advanced persistent threat intrusions), denial-of-service attacks, credential stuffing, credential harvesting, personnel misconduct or error, ransomware attacks, supply-chain attacks, software bugs, server malfunctions, software or hardware failures, loss of data or other information technology assets, adware, attacks enhanced or facilitated by AI, telecommunications failures, earthquakes, fires, floods, and other similar threats.
In particular, severe ransomware attacks are becoming increasingly prevalent and can lead to significant interruptions in our operations, ability to provide our products or services, loss of sensitive data and income, reputational harm, and diversion of funds. Extortion payments may alleviate the negative impact of a ransomware attack, but we may be unwilling or unable to make such payments due to, for example, applicable laws or regulations prohibiting such payments.
Remote work has become more common and has increased risks to our information technology systems and data, as more of our employees utilize network connections, computers, and devices outside our premises or network, including working at home, while in transit and in public locations. Additionally, future or past business transactions (such as acquisitions or integrations) could expose us to additional cybersecurity risks and vulnerabilities, as our systems could be negatively affected by vulnerabilities present in acquired or integrated entities’ systems and technologies. We may discover security issues that were not found during due diligence of such acquired or integrated entities, and it may be difficult to integrate companies into our information technology environment and security program.
We rely on our information technology systems to effectively manage our sales and marketing, accounting, financial and legal and compliance functions, engineering and product development tasks, research and development data, communications, supply chain, order entry and fulfillment and other business processes. We also rely on third parties and virtualized infrastructure to operate and support our information technology systems. Our ability to monitor these third parties’ information security practices is limited, and these third parties may not have adequate information security measures in place. If our third-party service providers experience a security incident or other interruption, we could experience adverse consequences. While we may be entitled to damages if our third-party service providers fail to satisfy their privacy or security-related obligations to us, any award may be insufficient to cover our damages, or we may be unable to recover such award. In addition, supply-chain attacks have increased in frequency and severity, and we cannot guarantee that third parties’ infrastructure in our supply chain or our third-party partners’ supply chains have not been compromised.
While we have implemented security measures designed to protect against security incidents, there can be no assurance that these measures will be effective. We take steps designed to detect, mitigate, and remediate vulnerabilities in our information systems (such as our hardware and/or software, including that of third parties upon which we rely). We may not, however, detect and remediate all such vulnerabilities including on a timely basis. Further, we may experience delays in deploying remedial measures and patches designed to address identified vulnerabilities. Vulnerabilities could be exploited and result in a security incident.
Any of the previously identified or similar threats could cause a security incident or other interruption that could result in unauthorized, unlawful, or accidental acquisition, modification, destruction, loss, alteration, encryption, disclosure of, or access to our sensitive data or our information technology systems, or those of the third parties upon whom we rely. A security incident or other interruption could disrupt our ability (and that of third parties upon whom we rely) to provide our services.
We may expend significant resources or modify our business activities to try to protect against security incidents. Additionally, certain data privacy and security obligations may require us to implement and maintain specific security
measures or industry-standard or reasonable security measures to protect our information technology systems and sensitive data.
Applicable data privacy and security obligations may require us to notify relevant stakeholders, including affected individuals, customers, regulators, and investors, of security incidents. Such disclosures are costly, and the disclosure or the failure to comply with such requirements could lead to adverse consequences.
If we (or a third party upon whom we rely) experience a security incident or are perceived to have experienced a security incident, we may experience adverse consequences, such as government enforcement actions (for example, investigations, fines, penalties, audits, and inspections); additional reporting requirements and/or oversight; restrictions on processing sensitive data (including personal data); litigation (including class claims); indemnification obligations; negative publicity; reputational harm; monetary fund diversions; diversion of management attention; interruptions in our operations (including availability of data); financial loss; and other similar harms. Security incidents and attendant consequences may prevent or cause customers to stop using our services, deter new customers from using our services, and negatively impact our ability to grow and operate our business.
Our contracts may not contain limitations of liability, and even where they do, there can be no assurance that limitations of liability in our contracts are sufficient to protect us from liabilities, damages, or claims related to our data privacy and security obligations. We cannot be sure that our insurance coverage will be adequate or sufficient to protect us from or to mitigate liabilities arising out of our privacy and security practices, that such coverage will continue to be available on commercially reasonable terms or at all, or that such coverage will pay future claims.
In addition to experiencing a security incident, third parties may gather, collect, or infer sensitive data about us from public sources, data brokers, or other means that reveals competitively sensitive details about our organization and could be used to undermine our competitive advantage or market position. Additionally, sensitive data of the Company or our customers could be leaked, disclosed, or revealed as a result of or in connection with our employees’, personnel’s, or vendors’ use of generative AI technologies.
Our success depends on our ability to attract and retain key employees and the succession of senior management.
Our continued growth and success requires us to hire, retain and develop our leadership team. If we are unable to attract and retain talented, highly qualified senior management and other key executives, as well as provide for the succession of senior management, our growth and results of operations may be adversely impacted.
Pandemics or disease outbreaks and any related economic downturn have impacted and in the future may continue to negatively impact our business, financial condition and results of operations.
Pandemics or disease outbreaks, such as the COVID-19 pandemic, have impacted and may continue to impact our business through adversely affected workforces, economies and financial markets globally, leading to a reduction or inability for our customers, partners, suppliers or vendors or other parties with whom we do business to meet their contractual obligations, and for a period of time, a reduction in customer spending on our products and services, and such conditions may reoccur in the future. For example, in our Services segment, we closed or cancelled clinics in 2020 and 2021 in response to COVID-19 and related public health measures and following initial reopening we experienced an elevated level of clinic closures due to labor shortages related to the COVID-19 pandemic.The extent to which pandemics or disease outbreaks in the future will impact our business, financial condition and results of operations in the future will depend on future developments, which are highly uncertain.
Risks Related to Finance and Accounting
We have a substantial amount of debt, which could adversely affect our financial position and our ability to raise additional capital and prevent us from fulfilling our obligations under our obligations.
As of December 31, 2023, we had total outstanding indebtedness of approximately $451.8 million consisting of $292.5 million outstanding under a term loan due 2028 (the "Term Loan B"), $143.8 million of outstanding 4.0% convertible senior notes due 2026 (the “Notes”) and $15.6 million in other debt. Additionally, we had an unused credit facility with $125.0 million of availability as of December 31, 2023 the ("ABL"). Our substantial indebtedness may:
•make it difficult for us to satisfy our financial obligations, including with respect to our indebtedness;
•limit our ability to borrow additional funds for working capital, capital expenditures, acquisitions, or other general business purposes;
•require us to use a substantial portion of our cash flow from operations to make debt service payments instead of other purposes, thereby reducing the amount of cash flow available for future working capital, capital expenditures, acquisitions, or other general business purposes, which is exacerbated given recent increases in interest rates;
•expose us to the risk of continued increased interest rates as certain of our borrowings, including under our credit facilities, are at variable rates of interest;
•limit our ability to pay dividends;
•limit our flexibility to plan for, or react to, changes in our business and industry;
•place us at a competitive disadvantage compared with our less-leveraged competitors;
•increase our vulnerability to the impact of adverse economic, competitive, and industry conditions; and
•increase our cost of borrowing.
In addition, the credit agreement governing our credit facility contains, and the agreements governing our future indebtedness may contain, restrictive covenants that may limit our ability to engage in activities that may be in our long-term best interest. These restrictive covenants include, among others, limitations on our ability and the ability of our affiliates, including HoldCo, to incur additional indebtedness and liens, engage in sale leaseback transactions, pay dividends or make other distributions in respect of, or repurchase or redeem, capital stock, prepay, redeem, or repurchase certain debt, make acquisitions, investments, loans, and advances, or sell or otherwise dispose of assets. Our failure to comply with those covenants could result in an event of default which, if not cured or waived, could result in the acceleration of substantially all of our debt.
The terms of the agreements governing our indebtedness limit, but do not prohibit, us from incurring additional indebtedness, and the additional indebtedness incurred in compliance with these restrictions could be substantial. If new indebtedness is added to our current debt levels, the related risks that we now face could intensify.
Operating our business requires a significant amount of cash, and we may not have sufficient cash flow from our business to pay the Notes and any other debt when due, which may seriously harm our business.
Our ability to generate cash to meet our operating needs, expenditures and debt service obligations will depend on our future performance and financial condition, which will be affected by financial, business, economic, legislative, regulatory and other factors, including potential changes in costs, pricing, the success of product innovation and marketing, competitive pressure and consumer preferences. If our cash flow and capital resources are insufficient to fund our debt service obligations and other cash needs, we could face substantial liquidity problems and could be forced to reduce or delay investments and capital expenditures or to dispose of material assets or operations, seek additional debt or equity capital or restructure or refinance our indebtedness. Our current and future debt agreements, including the Notes, the ABL Facility and the Term Loan B restrict our ability to take these actions and we may not be able to affect any such alternative measures on commercially reasonable terms or at all. If we cannot make scheduled payments on our debt, holders of our indebtedness can terminate their commitments to loan money, can declare all outstanding principal and interest to be due and payable, and, to the extent such debt is secured, foreclose against the assets securing their borrowings and we could be forced into bankruptcy or liquidation. In addition, any downgrade of our debt ratings by any of the major rating agencies, which could result from our financial performance, acquisitions or other factors, would also negatively impact our access to additional debt financing (including leasing) or refinancing on favorable terms, or at all. Even if we are successful in taking any such alternative actions, such actions may not allow us to meet our scheduled debt service obligations and, as a result, our business, financial condition and results of operations may be materially adversely affected.
In addition, holders of the Notes have the right to require us to repurchase all or a portion of the Notes on the occurrence of a fundamental change, as defined in the Indenture at a repurchase price equal to 100% of the principal amount of the Notes to be repurchased, plus accrued and unpaid interest, if any, to, but excluding, the fundamental change repurchase date. Further, if a make-whole fundamental change as defined in the indenture governing the Notes (the “Indenture”) occurs prior to the maturity date of the Notes, we will in some cases be required to increase the conversion rate for a holder that elects to convert its Notes in connection with such make-whole fundamental change. On the conversion of the Notes, unless we elect to deliver solely shares of our Class A common stock to settle such conversion (other than paying cash in lieu of delivering any fractional share), we will be required to make cash payments for the Notes being converted. However, we may not have enough available cash or be able to obtain financing at the time we are required to make such repurchases of the Notes surrendered or pay cash with respect to the Notes being converted.
We cannot be certain that additional financing will be available on reasonable terms when needed, or at all.
We have historically incurred net losses and we may not attain and sustain profitability in future periods. As a result, we may need additional financing. Our ability to obtain additional financing, if and when required, will depend on investor demand, our operating performance, our credit rating, the condition of the capital markets, and other factors. To the extent we use available funds or draw on our ABL Facility, we may need to raise additional funds and we cannot assure investors that additional financing will be available to us on favorable terms when required, or at all. If we raise additional funds through the issuance of equity, equity-linked, or debt securities, those securities may have rights, preferences, or privileges senior to the rights of our Class A common stock, and our existing stockholders may experience dilution. In the event that we are unable to obtain additional financing on favorable terms, our interest expense and principal repayment requirements could increase significantly, which could have a material adverse effect on our business, financial condition and results of operations.
We have incurred net losses in the past and may be unable to achieve or sustain profitability in the future.
As of December 31, 2023, we had an accumulated deficit of $160.6 million. We expect to continue to incur significant product commercialization and regulatory, sales and marketing, clinic opening, and other expenses. In addition, our selling, general and administrative expenses increased following prior acquisitions to support the larger combined Company and product portfolio. The net income (loss) we earn may fluctuate significantly from quarter to quarter. We will need to generate additional net sales or increased gross margin to attain and sustain profitability, and we cannot be sure that we will achieve or sustain profitability for any substantial period of time. Our failure to achieve or sustain profitability could negatively impact the value of our Class A common stock.
The trading price of our Class A common stock is highly volatile and could reduce the market price of our shares of Class A common stock in spite of our operating performance.
The volatility in the trading price of our Class A common stock, as well as general economic, market or political conditions, could reduce the market price of shares of our Class A common stock in spite of our operating performance. In addition, our results of operations could be below the expectations of public market analysts and investors due to a number of potential factors, including variations in our quarterly results of operations, additions or departures of key management personnel, failure to meet analysts’ earnings estimates, publication of research reports about our industry, litigation and government investigations, changes or proposed changes in laws or regulations or differing interpretations or enforcement thereof affecting our business, adverse market reaction to any indebtedness we may incur or securities we may issue in the future, changes in market valuations of similar companies or speculation in the press or investment community, announcements by our competitors of significant contracts, acquisitions, dispositions, strategic partnerships, joint ventures or capital commitments and adverse publicity about our industry in or individual scandals, and in response the market price of shares of our Class A common stock could decrease significantly.
In the past few years, stock markets have experienced extreme price and volume fluctuations that have affected and continue to affect the market prices of many companies, including our own. Fluctuations have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry fluctuations, as well as general economic, political, regulatory and market conditions, may continue to negatively impact investor confidence and the market price of equity securities, including our Class A common stock. In the past, following periods of volatility in the overall market and the market price of a company’s securities, securities class action litigation has often been instituted against these companies. This litigation, if instituted against us, could result in substantial costs and a diversion of our management’s attention and resources.
Our quarterly operating results may fluctuate significantly and could fall below the expectations of securities analysts and investors due to seasonality and other factors, some of which are beyond our control, resulting in a decline in our stock price.
Our quarterly operating results may fluctuate significantly because of several factors, including:
•the timing of new product and clinic launches;
•the timing and extent of customer inventory management decisions;
•our ability to procure product in a cost effective manner;
•expansion to new customers or product categories;
•seasonality of services;
•macroeconomic conditions, both nationally and locally;
•negative publicity relating to use of pet products outside the veterinary channel; and
•taxes.
Seasonal factors and the timing of holidays cause our revenue to fluctuate from quarter to quarter. Our flea and tick product offerings are most significant in the first half of the year, both leading up to and throughout the spring and summer seasons. Adverse weather conditions may also affect customer traffic to our customers or our ability to meet customer delivery requirements.
Risk Related to Legal and Regulatory
If our products or services are alleged to cause injury or illness or our products fail to comply with governmental regulations, we may need to recall our products and/or may experience related claims and reputational damage.
Our products may be subject to product recalls, including voluntary recalls or withdrawals, if they are alleged to pose a risk of injury or illness, or if they are alleged to have been mislabeled, misbranded or adulterated or to otherwise be in violation of governmental regulations. We may also voluntarily recall or withdraw products in order to protect our brand or reputation if we determine that they do not meet our standards, whether for quality, palatability, appearance or otherwise. If there is any future product recall or withdrawal, it could result in substantial and unexpected expenditures, destruction of product inventory, damage to our reputation and lost sales due to the unavailability of the product for a period of time, and our business, financial condition and results of operations may be materially adversely affected. In addition, a product recall or withdrawal may require significant management attention and could result in enforcement action by regulatory authorities.
We also may be subject to product liability claims if the consumption or use of our products is alleged to cause injury or illness. Although we carry product liability insurance, our insurance may not be adequate to cover all liabilities that we may incur in connection with product liability claims. For example, punitive damages are generally not covered by insurance. If we are subject to substantial product liability claims in the future, we may not be able to continue to maintain our existing insurance, obtain comparable insurance at a reasonable cost, if at all, or secure additional coverage. This could result in future product liability claims being uninsured. If there is a product liability judgment against us or a settlement agreement related to a product liability claim, our business, financial condition and results of operations may be materially adversely affected. In addition, even if product liability claims against us are not successful or are not fully pursued, these claims could be costly and time-consuming and may require management to spend time defending claims rather than operating our business.
Additionally, we may be subject to claims for veterinary malpractice or negligence in the event as a result of services provided by our veterinarians. Although we carry appropriate insurance, our insurance may not be adequate to cover all liabilities that we may incur in connection with veterinary malpractice or negligence claims. Additionally, any such claims may result in reputational damage to our services segment and our business, financial condition and results of operations may be materially adversely affected.
Failure to protect our intellectual property could harm our competitive position or require us to incur significant expenses to enforce our rights.
Our success depends in part on our ability to protect our intellectual property rights. Our trademarks such as “PetIQ,” “PetArmor,” “VIP Petcare,” “VetIQ PetCare,” “VetIQ,” “Capstar,” “Advecta,” “SENTRY,” “Sergeants,” “PurLuv,” “Rocco & Roxie,” and “Minties” and others are assets that support our brand, sub-brands and consumers’ perception of our products. We rely on trademark, copyright, trade secret, patent and other intellectual property laws, as well as nondisclosure and confidentiality agreements and other methods, to protect our trademarks, trade names, proprietary information, technologies and/or processes. Our non-disclosure agreements and confidentiality agreements may not effectively prevent disclosure of our proprietary information, technologies and processes and may not provide an adequate remedy in the event of unauthorized disclosure of such information, which could harm our competitive position. In addition, effective patent, copyright, trademark and trade secret protection may be unavailable or limited for some of our intellectual property rights and trade secrets in foreign countries. We may need to engage in litigation or similar activities to enforce our intellectual property rights, to protect our trade secrets or to determine the validity and scope of proprietary rights of others. Any such litigation could require us to expend significant resources and divert the efforts and attention of our management and other personnel from our business operations. If we fail to protect our intellectual property, our business, financial condition and results of operations may be materially adversely affected.
We may be subject to intellectual property infringement claims or other allegations, which could result in substantial damages and diversion of management’s efforts and attention.
We have obligations to respect third-party intellectual property. The steps we take to prevent misappropriation, infringement or other violation of the intellectual property of others may not be successful. From time to time, third parties have asserted intellectual property infringement claims against us, our suppliers, or our retail customers and may continue to do so in the future. Although we believe that our products and manufacturing processes do not infringe in any material respect upon proprietary rights of other parties and/or that meritorious defenses would exist with respect to any assertions of infringement of other parties, we may from time to time be found to infringe on the proprietary rights. For example, patent applications in the United States and some foreign countries are generally not publicly disclosed until the patent application is published, and we may not be aware of currently filed patent applications that relate to our products or processes. If patents later are issued on these applications, we may be found liable for subsequent infringement. Such claims that our products or processes infringe these rights, regardless of their merit or resolution, could be costly and may divert the efforts and attention of our management and technical personnel. In part due to the complex technical issues and inherent uncertainties in intellectual property litigation, we cannot predict whether we will prevail in such proceedings. If such proceedings result in an adverse outcome, we could, among other things, be required to:
•Pay substantial damages (potentially treble damages in the United States);
•cease the manufacture, use or sale of the infringing products;
•discontinue the use of the infringing processes;
•expend significant resources to develop non-infringing processes;
•expend significant resources to litigate matters or to develop non-infringing processes; and
•enter into licensing arrangements with the third party claiming infringement, which may not be available on commercially reasonable terms, or may not be available at all.
If any of the foregoing occurs, our ability to compete could be affected and our business, financial condition and results of operations may be materially adversely affected.
Adverse litigation judgments or settlements resulting from legal proceedings relating to our business operations could materially adversely affect our business, financial condition and results of operations.
From time to time, we are subject to allegations, and may be party to legal claims and regulatory proceedings, relating to our business operations. Such allegations, claims and proceedings may be brought by third parties, including our customers, employees, governmental or regulatory bodies or competitors. Defending against such claims and proceedings, regardless of their merits or outcomes, is costly and time consuming and may divert management’s attention and personnel resources from our normal business operations, and the outcome of many of these claims and proceedings cannot be predicted. If any of these claims or proceedings were to be determined adversely to us, a judgment, a fine or a settlement involving a payment of a material sum of money were to occur, or injunctive relief were issued against us, our reputation could be affected and our business, financial condition and results of operations could be materially adversely affected.
We are subject to extensive and ongoing governmental regulation and we may incur material costs in order to comply with existing or future laws and regulations, and our failure to comply may result in enforcement, recalls and other adverse actions or significant penalties.
We are subject to a broad range of federal, state, local and foreign laws and regulations intended to protect public health and safety, natural resources and the environment. See “Business—Government Regulation.” Our operations are subject to extensive and ongoing regulation by the FDA, EPA, USDA, the Florida Department of Health and by various other federal, state, local and foreign authorities regarding the manufacturing, processing, packaging, storage, distribution, advertising, labeling and import and export of our products, including drug and food safety standards. Our operations also are subject to regulation regarding the availability and use of pesticides, emissions and discharges to the environment, and the treatment, handling, storage and disposal of materials and wastes. Many of these laws and regulations are becoming increasingly stringent and compliance with them is becoming increasingly expensive. Costs of compliance, and the impacts on us of any non-compliance, with any such laws and regulations could materially adversely affect our business, financial condition and results of operations.
Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with our third-party manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
•restrictions on the marketing or manufacturing of the product, withdrawal of the product from the market, or voluntary or mandatory product recalls;
•fines, warning letters or holds on target animal studies;
•refusal by applicable regulatory authorities to approve pending applications or supplements to approved applications, or suspension or revocation of product approvals;
•product seizure or detention, or refusal to permit the import or export of products; and
•injunctions or the imposition of civil or criminal penalties.
Regulatory policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of any current or future product candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained, which would adversely affect our business.
Our business is also affected by export and import controls and similar laws and regulations, both in the United States and elsewhere. Issues such as national security or health and safety, which may slow or otherwise restrict imports or exports, may adversely affect our business, financial condition and results of operations.
Violations of or liability under any of these laws and regulations may result in administrative, civil or criminal fines or penalties against us, revocation or modification of applicable permits, environmental investigations or remedial activities, voluntary or involuntary product recalls, warning or untitled letters or cease and desist orders against or restrictions on operations that are not in compliance, among other things. Liability may be imposed under some laws and regulations regardless of fault or knowledge and regardless of the legality of the original action. These laws and regulations, or their interpretation, may change in the future and we may incur (directly, or indirectly through our contract manufacturers) material costs to comply with current or future laws and regulations or in any required product recalls.
Certain states have laws, rules and regulations which require that veterinary medical practices be owned by licensed veterinarians and that corporations which are not owned by licensed veterinarians refrain from providing, or holding themselves out as providers of, veterinary medical care. We may experience difficulty in expanding our operations into other states or provinces with similar laws, rules and regulations. Although we have structured our operations to comply with our understanding of the veterinary medicine laws of each state and province in which we operate, interpretive legal precedent and regulatory guidance varies by jurisdiction and is often sparse and not fully developed. A determination that we are in violation of applicable restrictions on the practice of veterinary medicine in any jurisdiction in which we operate, could have a material adverse effect on us, particularly if we are unable to restructure our operations to comply with the requirements of that jurisdiction.
All of the states in which we operate impose various registration permit and/or licensing requirements. To fulfill these requirements, we have registered each of our facilities with appropriate governmental agencies and, where required, have appointed a licensed veterinarian to act on behalf of each facility. All veterinarians practicing in our animal hospitals are required to maintain valid state licenses to practice.
Failure to comply with federal, state and international laws and regulations relating to permit and/or licensing requirements, or the expansion of existing or the enactment of new laws or regulation relating to permit and/or licensing requirements, could adversely affect our business and our financial condition.
We strive to comply with all applicable laws, regulations and other legal obligations relating to permit and/or licensing requirements. It is possible, however, that these requirements may be interpreted and applied in a manner that is inconsistent from one jurisdiction to another or may conflict with other rules or our practices. We cannot guarantee that our practices have complied, comply or will comply fully with all such laws, regulations, requirements and obligations. Any failure, or perceived failure, by us to comply with our filed permits and licenses with any applicable federal, state or international related laws, industry standards or codes of conduct, regulatory guidance, orders to which we may be subject or other legal obligations relating to privacy or consumer protection could adversely affect our reputation, brand and business, and may result in claims, proceedings or actions against us by governmental entities or others or other liabilities.
Any such claim, proceeding or action could hurt our reputation, brand and business, force us to incur significant expenses in defense of such proceedings, distract our management, increase our costs of doing business, result in a loss of customers and suppliers and may result in the imposition of monetary liability. We may also be contractually liable to indemnify and hold harmless third parties from the costs or consequences of non-compliance with any laws, regulations or other legal obligations relating to permit and/or licensing requirements. In addition, various federal, state and foreign legislative and regulatory bodies may expand existing laws or regulations, enact new laws or regulations or issue revised rules or guidance regarding permit and/or licensing requirements. Any such changes may force us to incur substantial costs or require us to change our business practices. This could compromise our ability to pursue our growth strategy effectively and may adversely affect our ability to acquire customers or otherwise harm our business, financial condition and results of operations.
If we fail to comply with governmental regulations applicable to our Services business, various governmental agencies may impose fines, institute litigation or preclude us from operating in certain states.
Certain states and provinces have laws, rules and regulations which require that veterinary medical practices be owned by licensed veterinarians and that corporations which are not owned by licensed veterinarians refrain from providing, or holding themselves out as providers of, veterinary medical care. We may experience difficulty in expanding our operations into other states or provinces with similar laws, rules and regulations. Although we have structured our operations to comply with our understanding of the veterinary medicine laws of each state in which we operate, interpretive legal precedent and regulatory guidance varies by jurisdiction and is often sparse and not fully developed. A determination that we are in violation of applicable restrictions on the practice of veterinary medicine in any jurisdiction in which we operate, could have a material adverse effect on us, particularly if we are unable to restructure our operations to comply with the requirements of that jurisdiction. All of the states in which we operate impose various registration requirements. To fulfill these requirements, we have registered each of our facilities with appropriate governmental agencies and, where required, have appointed a licensed veterinarian to act on behalf of each facility. All veterinarians practicing in our animal hospitals are required to maintain valid state licenses to practice.
We are subject to environmental, health, and safety laws and regulations that could result in costs to us.
In connection with the ownership and operations of our business, we are subject to laws and regulations relating to the protection of the environment and health and safety matters, including those governing the management and disposal of wastes and the cleanup of contaminated sites. We could incur costs, including fines and other sanctions, cleanup costs, and third-party claims, as a result of violations of or liabilities under environmental laws and regulations. Although we are not aware of any of our sites at which we currently have material remedial obligations, the imposition of remedial obligations as a result of the discovery of contaminants in the future could result in additional costs.
Continuing political and social attention to the issue of climate change has resulted in both existing and pending international agreements and national, regional, or local legislation and regulatory measures to limit greenhouse gas emissions, such as cap and trade regimes, carbon taxes, restrictive permitting, increased fuel efficiency standards, and incentives or mandates for renewable energy. Such measures could subject us and our vendors to additional costs and restrictions and require significant operating and capital expenditures, including with respect to waste and energy reduction, compliance costs, and workforce initiatives, which could adversely impact our business, financial condition, results of operations and cash flows.
Risks Related to our Organizational Structure
Our principal asset is our interest in HoldCo, and, accordingly, we depend on distributions from HoldCo to pay our taxes and expenses. HoldCo’s ability to make such distributions may be subject to various limitations and restrictions.
We are a holding company and have no material assets other than our ownership of LLC membership interests in HoldCo (“LLC Interests”). As such, we have no independent means of generating revenue or cash flow, and our ability to pay our taxes and operating expenses or declare and pay dividends in the future, if any, will be dependent upon the financial results and cash flows of HoldCo and its subsidiaries and distributions we receive from HoldCo. There can be no assurance that our subsidiaries will generate sufficient cash flow to distribute funds to us or that applicable state law and contractual restrictions, including negative covenants in our debt instruments, will permit such distributions.
We anticipate that HoldCo will continue to be treated as a partnership for U.S. federal income tax purposes and, as such, generally will not be subject to any entity-level U.S. federal income tax. Instead, taxable income will be allocated to
holders of LLC Interests, including us. Accordingly, we will incur income taxes on our allocable share of any net taxable income of HoldCo, as well as expenses related to our operations.
We intend, as its managing member, to cause HoldCo to make cash distributions to the owners of LLC Interests, including us, in an amount sufficient to (i) fund all or part of their tax obligations in respect of taxable income allocated to them and (ii) cover our operating expenses. However, HoldCo’s ability to make such distributions may be subject to various limitations and restrictions, such as restrictions on distributions that would either violate any contract or agreement to which HoldCo is then a party, including debt agreements, or any applicable law, or that would have the effect of rendering HoldCo insolvent. Our credit agreements do not currently restrict our ability to make tax distributions, except under limited circumstances. In addition, for taxable years beginning after December 31, 2017, liability for adjustments to a partnership’s tax return can be imposed on the partnership itself in certain circumstances, absent an election to the contrary. HoldCo could be subject to material liabilities pursuant to adjustments to its partnership tax returns if, for example, its calculations or allocations of taxable income or loss are incorrect, which also could limit its ability to make distributions to us.
Under the terms of the Limited Liability Company Agreement of HoldCo Agreement, tax distributions payable to us are computed based on our actual tax liability, whereas tax distributions payable to members of HoldCo besides us are computed based on an assumed combined tax rate equal to the maximum rate applicable to an individual resident in New York, New York (taking into account the deductibility of state and local taxes and other applicable adjustments). We expect such calculation to result in us often receiving less, and continuing partners of HoldCo (the "LLC Owners") often receiving more, than the respective pro rata shares of the total tax distributions paid by HoldCo.
If we do not have sufficient funds to pay tax or other liabilities or to fund our operations, we may have to borrow funds, which could materially adversely affect our liquidity and financial condition and subject us to various restrictions imposed by any such lenders. In addition, if HoldCo does not have sufficient funds to make distributions, our ability to declare and pay cash dividends will also be restricted or impaired.
Anti-takeover provisions in our organizational documents and Delaware law might discourage or delay acquisition attempts for us that you might consider favorable.
Our amended and restated certificate of incorporation and amended and restated bylaws contain provisions that may make the merger or acquisition of the Company more difficult without the approval of our board of directors. Among other things:
•removal of directors, only for cause, by a supermajority of the voting power of stockholders entitled to vote;
•a provision denying stockholders the ability to call special meetings;
•a provision denying stockholders the ability to act by written consent;
•provisions waiving the corporate opportunity doctrine with respect to certain stockholders and their affiliates;
•advance notice requirements for stockholder proposals and nominations; and
•the authorization of undesignated preferred stock, the terms of which may be established and shares of which may be issued without stockholder approval.
These provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors, which is responsible for appointing the members of our management, and may discourage, delay or prevent a transaction involving a change of control of our Company that is in the best interest of our stockholders. Even in the absence of a takeover attempt, the existence of these provisions may adversely affect the prevailing market price of our Class A common stock if they are viewed as discouraging future takeover attempts. In addition, because we are incorporated in Delaware, we have opted out of Section 203 of the General Corporation Law of the State of Delaware.
Unanticipated changes in effective tax rates or adverse outcomes resulting from examination of our income or other tax returns could adversely affect our results of operations and financial condition.
We are subject to U.S. federal, state and local and non-U.S. taxes, and our tax liabilities will be affected by the allocation of expenses to differing jurisdictions. Our future effective tax rates could be subject to volatility or adversely affected by a number of factors, including:
•changes in the valuation of our deferred tax assets and liabilities;
•expected timing and amount of the release of any tax valuation allowances;
•tax effects of stock-based compensation; or
•changes in tax laws, regulations or interpretations thereof.
For example, the Inflation Reduction Act of 2022 introduced a 15% alternative minimum tax on the “adjusted financial statement income” of certain large corporations and a 1% excise tax on certain actual and deemed stock repurchases, both of which became effective in 2023. While we do not expect to be an applicable corporation that is subject to the alternative minimum tax as currently enacted, we will be a covered corporation that could be subject to the stock repurchase excise tax.
In addition, we may be subject to audits of our income, sales and other transaction taxes by U.S. federal, state and local and non-U.S. taxing authorities. Outcomes from these audits could have an adverse effect on our operating results and financial condition.
Our ability to use our net operating losses to offset future taxable income may be subject to certain limitations.
We have U.S. federal net operating loss (“NOL”) carryforwards as a result of prior period losses, some of which, if not utilized will begin to expire in fiscal year 2037 for federal purposes. These net operating loss carryforwards could expire unused and be unavailable to offset future income tax liabilities, which could adversely affect our potential profitability.
In addition, under Section 382 of the Internal Revenue Code of 1986, as amended, if a corporation undergoes an “ownership change,” its ability to use its pre-change net operating loss carryforwards to offset its post-change taxable income or tax liability may be limited. Such an “ownership change” generally occurs if there is a greater than 50 percentage point change (by value) in its equity ownership by one or more stockholders or groups of stockholders who own at least 5% of our stock over a three-year period. We have experienced ownership changes in the past and may experience ownership changes in the future as a result of subsequent shifts in our stock ownership. As a result, if we earn net taxable income, our ability to use our pre-change net operating loss carryforwards and other pre-change tax attributes to offset U.S. federal and state taxable income or tax liability may be subject to limitations, which could potentially result in increased future tax liability to us. Furthermore, under the current U.S. federal tax laws, the amount of net operating loss carryforwards from tax years beginning after December 31, 2017 that we are permitted to use in any taxable year beginning after December 31, 2020 is limited to 80% of our taxable income in such year, where taxable income is determined without regard to the net operating loss deduction itself. Under current U.S. federal tax laws, net operating losses are generally not permitted to be carried back to prior taxable years. There is also a risk that, due to regulatory changes, such as suspensions of the use of NOLs, or other unforeseen reasons, our existing NOLs could expire or otherwise be unavailable to offset future income tax liabilities. For these reasons, we may not be able to realize a tax benefit from the use of our NOLs, whether or not we attain profitability.
Since we have no current plans to pay regular cash dividends on our Class A common stock, you may not receive any return on investment unless you sell your Class A common stock for a price greater than that which you paid for it.
We have not paid, and do not anticipate paying, any regular cash dividends on our Class A common stock. Any decision to declare and pay dividends in the future will be made at the discretion of our board of directors and will depend on, among other things, our financial condition, results of operations, cash requirements, contractual restrictions and other factors that our board of directors may deem relevant. In addition, our ability to pay dividends is, and may be, limited by covenants of existing and any future outstanding indebtedness we or our subsidiaries incur, including under the ABL Facility and Term Loan B. Therefore, any return on investment in our Class A common stock is solely dependent upon the appreciation of the price of our Class A common stock on the open market, which may not occur. Please read “Dividend Policy” for more detail.
Item 1B. Unresolved Staff Comments
None.
Item 1C. Cybersecurity
Cybersecurity Risk Management & Strategy
PetIQ has implemented and maintains various information security processes and systems designed to identify, assess, and manage material risks from cybersecurity threats to our critical computer networks, third party hosted services,
communications systems, hardware and software, and our critical data, including confidential information that is proprietary, strategic, or competitive in nature (“Information Systems and Data”).
Our cybersecurity function, led by our Cybersecurity Manager and supported by our Chief Information Officer (“CIO”) and third-party service providers, helps identify, assess, and manage the Company’s cybersecurity threats and risks. This group identifies and assesses cybersecurity threats by monitoring and evaluating our threat environment using various methods including: using manual and automated tools, subscribing to reports and services that identify cybersecurity threats, conducting scans of the threat environment, conducting internal and external cybersecurity audits, engaging third-party threat assessments, conducting vulnerability assessments, leveraging external intelligence feeds, and completing third-party red/blue team exercises and tabletop incident response exercises.
Depending on the environment and system, we implement and maintain various technical, physical, and organizational measures, processes, standards, practices and policies designed to manage and mitigate material risks from cybersecurity threats to our Information Systems and Data, including: incident management processes (for both internal and third-party hosted systems), maintaining certain security certifications, encryption of certain data, network security controls, access controls, physical security, asset management (such as tracking and disposal), systems monitoring, vendor risk management processes, employee training, penetration testing, dedicated cybersecurity staff, and cybersecurity insurance.
We use third-party service providers to assist us to identify, assess, and manage material risks from cybersecurity threats, including threat intelligence service providers, cybersecurity software and managed cybersecurity providers, penetration testing firms, and forensic investigators.
Further, we use third-party service providers to perform a variety of functions throughout our business, such as software-as-a-service providers and data/computing hosting companies. We have a vendor management program that, depending on the nature of the services provided, the sensitivity of the Information Systems and Data at issue, and the identity of the provider, may involve different levels of assessment designed to help identify cybersecurity risks associated with a provider. This vendor management program includes a security questionnaire, reviews of security assessments, and vulnerability scans related to the vendor, as well as the imposition of contractual obligations related to cybersecurity.
At PetIQ, cybersecurity is an overall company risk that is managed as a part of the Enterprise Risk Management Program which is updated and reviewed quarterly and is overseen by both senior management and the Board of Directors. For a description of the risks from cybersecurity threats that may materially affect PetIQ and how they may do so, see our risk factors under Part 1. Item 1A. Risk Factors in this Annual Report on Form 10-K, including “If our information technology systems or those of third parties upon which we rely, or our data are or were compromised, we could experience adverse consequences resulting from such compromise, including but not limited to regulatory investigations or actions; litigation; fines and penalties; disruptions of our business operations; reputational harm; loss of revenue or profits; and other adverse consequences.”
Governance
PetIQ’s Board of Directors is responsible for overseeing the Company’s cybersecurity risks and threats. Specifically, the Audit Committee of the Board of Directors reviews the Company’s cybersecurity status, risks, and threats periodically. Additionally, as needed, individual board members may reach out to Company management directly with cybersecurity questions or clarifications.
PetIQ has implemented cybersecurity processes and procedures in coordination with cybersecurity risk mitigation tools and services designed to help prevent, detect, and eradicate cybersecurity incidents. The CIO, who has more than 30 years of IT experience, has overall accountability for cybersecurity. The Cybersecurity Manager reports to the CIO. The Cybersecurity manager, who has overall responsibility for assessing and managing cybersecurity risk as well as managing and monitoring the cybersecurity technology stack, has more than 20 years of IT experience and is an ANSI/EC-Council certified CISO.
PetIQ’s cybersecurity incident management processes include processes to assess the impact of an incident for reporting purposes, as well as escalation procedures for incidents (based on severity, risk, and impact) that can flow communications and decisions up through the CIO, Executive/Senior Leadership, and the Audit Committee of the Board of Directors as needed.
Item 2 - Properties
The following table sets forth the location, size, use and lease expiration date of our material properties as of December 31, 2023.
| | | | | | | | | | | | | | | | | | | | |
| LOCATION | | APPROXIMATE SIZE | | PRINCIPAL USE(S) | | LEASE EXPIRATION DATE |
| Daytona Beach, Florida | | 142,900 square feet | | Manufacturing and distribution warehouse; office | | November 30, 2025 |
| Springville, Utah | | 242,000 square feet | | Manufacturing and distribution warehouse; office | | January 31, 2029 |
| Omaha, Nebraska | | 131,150 square feet | | Manufacturing; office | | Owned |
| Omaha, Nebraska | | 349,680 square feet | | Distribution warehouse | | September 30, 2026 |
| Eagle, Idaho | | 65,000 square feet | | Corporate Headquarters | | Owned |
We are obligated under non-cancelable leases for the facilities we do not own. Our leases have varying terms, typically with three to five year renewal options.
We believe that our current properties are adequate for our intended purposes and represent sufficient capacity for our near term plans.
Item 3 – Legal Proceedings
For a discussion of our “Legal Proceedings,” refer to Note 13 – Commitments and Contingencies in the notes to our audited consolidated financial statements of this Annual Report.
Item 4 – Mine Safety Disclosures
Not Applicable
PART II
Item 5 – Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Holders of Record
As of February 21, 2024, there were approximately 10 holders of record of our Class A common stock and 6 holders of record of our Class B common stock. The holders of our Class B common stock also hold LLC interests in HoldCo. There is no public market for these interests. A substantially greater number of holders of our stock are held in “street name” and held of record by banks, brokers, and other financial institutions.
Dividend Policy
We have not historically paid cash dividends on our common stock, and have no current plans to pay cash dividends on our Class A common stock. The declaration, amount and payment of any future dividends will be at the sole discretion of our board of directors. Our board of directors may take into account general and economic conditions, our financial condition and results of operations, our available cash and current and anticipated cash needs, capital requirements, contractual, legal, tax and regulatory restrictions and implications on the payment of dividends by us to our stockholders or by our subsidiaries to us, including restrictions under our senior secured credit facilities and other indebtedness we may incur, and such other factors as our board of directors may deem relevant.
Stock Repurchase Program
On September 6, 2022, the Company's Board of Directors authorized a stock repurchase program for up to $30 million of the Company’s outstanding shares of Class A common stock. Repurchases of Class A common stock may be made at
management’s discretion from time to time in one or more transactions on the open market or in privately negotiated purchase and/or through other legally permissible means, depending on market conditions and in accordance with applicable rules and regulations promulgated under Securities Exchange Act. The Company did not purchase any shares during the year ended December 31, 2023. As of December 31, 2023, $26.1 million in aggregate dollar value of shares remained available for purchase under the stock repurchase program.
Stock Performance Graph
The information contained in the following chart is not considered to be “soliciting material,” or “filed,” or incorporated by reference in any past or future filing by the Company under the Securities Act or Exchange Act unless and only to the extent that, the Company specifically incorporates it by reference.
The following graph compares our total common stock return with the total return for (i) the NASDAQ Composite Index (the “NASDAQ Composite”) and (ii) the Russell 2000 Index (the “Russell 2000”) for the period from December 31, 2018 through December 31, 2023. The figures represented below assume an investment of $100 in our common stock on December 31, 2018 and in the NASDAQ Composite and the Russell 2000 on the same date. The comparisons in the table are required by the SEC and are not intended to forecast or be indicative of possible future performance of our common stock.
| | | | | | | | | | | | | | | | | | | | |
| Date | | PetIQ | | NASDAQ Composite | | Russell 2000 |
| December 31, 2018 | | $ | 100.00 | | | $ | 100.00 | | | $ | 100.00 | |
| December 31, 2019 | | 106.73 | | | 135.23 | | | 123.72 | |
| December 31, 2020 | | 163.83 | | | 194.24 | | | 146.44 | |
| December 31, 2021 | | 96.76 | | | 235.78 | | | 166.50 | |
| December 31, 2022 | | 39.28 | | | 157.74 | | | 130.60 | |
| December 31, 2023 | | 84.15 | | | 226.24 | | | 150.31 | |
Item 6 – [Reserved]
Item 7 – Management’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis of our financial condition and results of operations should be read together with our audited consolidated financial statements and related notes and other financial information appearing elsewhere in this Annual Report. This section of this Annual Report generally discusses 2023 and 2022 items and year-to-year comparisons of 2023 to 2022. Discussions of 2021 items and year-to-year comparisons of 2022 and 2021 that are not included in this Annual Report can be found in “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in Part II, Item 7 on our Annual Report on Form 10-K for the year ended December 31, 2022, which is incorporated by reference herein. This discussion contains forward-looking statements that reflect our plans, estimates, and beliefs and involve numerous risks and uncertainties. Actual results may differ materially from those contained in any forward-looking statements. See “Cautionary Note Regarding Forward-Looking Statements.”
Business Overview
PetIQ is a leading pet medication, product and wellness company delivering a smarter way for pet parents to help pets live their best lives through convenient access to affordable health and wellness products and veterinary services. We have two reporting segments: (i) Products; and (ii) Services. PetIQ believes that pets are an important part of the family and deserve the best products and care we can provide.
Our Products segment consists of our product manufacturing and distribution business through which we manufacture and distribute pet medication and health and wellness products to major U.S. retail and e-commerce channels through more than 60,000 points of distribution. We focus our offerings on innovative, proprietary value-branded products, and leading third-party branded products for dogs and cats, including pet Rx medications, OTC medications and wellness products. Our Products segment is further supported by our world-class medications manufacturing facility in Omaha, Nebraska and health and wellness manufacturing facility in Springville, Utah.
Our Services segment consists of veterinary services, and related product sales, provided by the Company directly to consumers. Our national veterinarian service platform operates at over 2,600 community clinic locations and wellness centers hosted at retailers across 39 states providing cost effective and convenient veterinary wellness services. We offer diagnostic tests, vaccinations, prescription medications, microchipping, grooming and hygiene and wellness checks.
During the year ended December 31, 2023, we implemented a Services segment optimization (the "optimization") to improve the functioning of the Services segment and profitability. The optimization included assessing the operational and financial performance of the Company's wellness centers since re-opening after the pandemic as well as the assessment of the veterinary labor market in each geographic market. The Company also evaluated its ability to potentially convert these locations to a more hygiene-focused offering and determined they would be unable to convert these locations in the future based on the aforementioned assessment and the available square footage within the respective wellness centers. As a result of the optimization, the Company identified 149 underperforming wellness centers for closure. The Company closed 149 wellness centers during the year ended December 31, 2023 and finished the year with 133 wellness centers. Restructuring and related charges attributable to the optimization were $13.6 million recorded on the consolidated statements of operations for the year ended December 31, 2023. These charges include approximately $11.0 million of depreciation and amortization as well as $0.9 million inventory valuation adjustments, and variable lease expenses, severance and other termination costs of approximately $2.6 million. For additional information about the restructuring, see "Note 15 - Restructuring" to our condensed consolidated financial statements included herein.
On January 13, 2023, the Company completed the acquisition of all of the membership units of Rocco and Roxie Supply Co, LLC ("R&R"), after which R&R became a wholly owned and consolidated subsidiary of the Company. The acquisition expands the Company's product portfolio to include stain and odor products, jerky treats and behavioral products as well as expands sales channels in e-commerce.
PetIQ, Inc. is the managing member of PetIQ Holdings, LLC (“HoldCo”), a Delaware limited liability company, which is the sole member of PetIQ, LLC (“Opco”) and, through HoldCo, operate and control all the business and affairs of Opco.
Macroeconomic Trends
We, like other businesses, face challenges related to global economic conditions, the economic downturns and high inflation. Growth in the companion animal sectors is driven by overall economic development and related growth. In the
past, certain of our customers and suppliers have been affected directly by economic downturns and inflation, which decreased the demand for our products and services.
Industry sources have reported that pet owners indicated a preference for reducing spending on other aspects of their lifestyle, including entertainment, clothing and household goods, before reducing spending on pet care. Each of these factors, contributes to our ability to incorporate inflationary challenges into our product pricing and mitigate the impact on our results. While these factors have mitigated the impact of prior downturns in the global economy, economic challenges, including the current economic downturn and inflation, could increase cost sensitivity among our customers, which may result in reduced demand for our products, which could have a material adverse effect on our operating results and financial condition.
Components of our Results of Operations
Net Sales
Our Product Segment net sales consist of our total product sales net of product returns, allowances (discounts), trade promotions and incentives. We offer a variety of trade promotions and incentives to our customers, such as cooperative advertising programs and in-store displays. We recognize revenue when control transfers to our customers, in accordance with the terms of our contracts, which generally occurs upon shipment of product. Most contracts contain variable consideration, which is estimated at the time of sale and updated at each period end. Trade promotions are used to increase our aggregate net sales. Our net sales are periodically influenced by the timing, extent and amount of such trade promotions and incentives.
Key factors that may affect our future Product sales growth include new product introductions; expansion into other customer bases; expansion of items sold to existing customers, addition of new retail customers; price competition; as well as whether maintaining and developing positive relationships with key retail customers. In addition, our products are primarily consumables and, as such, they experience a replenishment cycle.
Our Service Segment revenue consists of providing veterinary services to consumers and selling products to the consumer in conjunction with providing those services. The customer generally renders payment at the time the service is provided. Services Segment revenue is dependent on the number of clinics and wellness centers we run, dollars per pet and the number of pets we see in our clinics and wellness centers.
While many of our products are sold consistently throughout the year, we experience seasonality in the form of increased demand for our flea and tick product offerings in the first half of the year, both leading up to and throughout the spring and summer seasons. Additionally, our veterinary services experience seasonality as consumers typically seek more services in the warmer months.
Gross Profit
Gross profit consists of net product sales plus service revenue less cost of product sales and services. Our cost of product sales consists primarily of costs of raw goods, finished goods, packaging materials, manufacturing, shipping and handling costs and costs associated with our warehouses and distribution network. Cost of services are comprised of all service and product costs related to providing veterinary services, including but not limited to, salaries or contract costs of veterinarians, technicians and other clinic based personnel, transportation and delivery costs, facilities rent, occupancy costs, supply costs, depreciation and amortization of clinic assets, certain marketing and promotional expenses and costs of goods sold.
Gross margin measures our gross profit as a percentage of net sales. With respect to our proprietary products, we have a manufacturing network that includes leased and owned manufacturing facilities where we manufacture finished goods, as well as third-party contract manufacturing facilities from which we purchase finished products. The gross margin on our proprietary value-branded products is higher than on our distributed products. For distributed products, our costs are driven by the extent of value-added products and services we render with the distributed product. Gross profit in the services segment is driven by the number of pets that seek services in the individual clinics and wellness centers due to the relatively fixed cost nature of operating the clinic or wellness center.
Selling, General and Administrative Expenses
Our selling, general and administrative expenses primarily consist of employee compensation and benefits expenses, sales and merchandising expenses, advertising and marketing expenses, a portion of rent and lease expenses, information technology (“IT”), utility expenses, professional fees, insurance costs, R&D costs, host fees, banking charges, and consulting fees.
Our advertising and marketing expenses primarily consist of digital marketing (e.g. social, display and search, etc.), online video and streaming TV, e-mail, in-store merchandising and trade shows in an effort to build awareness and drive demand for our products and services. Our Product Segment focuses on promoting our manufactured brands through direct-to-consumer, supported by trade promotions and merchandising. Our Services Segment focuses on promoting our veterinary services direct-to-consumer, geo-targeted around our retail locations, supported by in-store signage. We generally expect our marketing expenses to increase commensurate with increases in revenue and market share for both segments.
As noted above, we experience seasonality in the form of increased demand for our flea and tick product offerings in the first half of the year, both leading up to and throughout the spring and summer seasons and, as a result, the sales and merchandising expenses component of our selling, general and administrative expenses generally increases during this period due to promotional spending relating to our flea and tick product lines.
Restructuring
During the year ended December 31, 2023, the Company implemented a Services segment optimization (the "optimization") to improve the functioning of the Services segment and profitability. The optimization included assessing the operational and financial performance of the Company's wellness centers since re-opening after the pandemic as well as the assessment of the veterinary labor market in each geographic market. The Company also evaluated its ability to potentially convert these locations to a more hygiene-focused offering and determined we would be unable to convert these locations in the future based on the aforementioned assessment and the available square footage within the respective wellness centers. As a result of the optimization, the Company identified 149 underperforming wellness centers for closure. Restructuring charges totaled $13.6 million for the year ended 2023 and had no comparable event in 2022.
Impairment and other asset charges
During the year ended December 31, 2023, the Company committed to a plan to sell its foreign subsidiary, Mark & Chappell, and initiated the required actions to complete such a sale. In connection with the expected sale of the business and classification of related assets and liabilities as held for sale, we recorded a non-cash asset charge of $7.7 million for the year ended December 31, 2023. During the year ended December 31, 2022, the Company recorded an impairment charge of $47.3 million related to the write-down of the full goodwill balance of the Services segment.
Net income (loss)
Our net income (loss) for future periods may be affected by the various factors described above. In addition, our historical results are impacted by Opco’s status as a pass-through entity for U.S. federal income tax purposes and our ownership percentage of HoldCo. Our financials include a valuation allowance which effectively removed our deferred tax assets based on the likelihood of realization. Improved profitability could result in reversal of the valuation allowance, resulting in significant swings in net income (loss). Our tax expense is impacted by our structure and, as a result, we expect our tax
expense to fluctuate on a quarterly basis depending on the number of exchanges of LLC interests that occur during each
period
Non-Controlling Interest
We consolidate the financial position and results of operations of HoldCo. Our continuing LLC Owners hold their equity investment in us primarily through LLC Interests in the Company’s subsidiary, HoldCo, and an equal number of shares of the Company’s Class B common stock. Our Class B Stock has voting, but no economic rights. Each LLC Interest, together with a share of Class B Stock held by the Continuing LLC, is exchangeable for a share of the Company’s Class A common stock (or at the option of the Company, the cash equivalent thereof). The Company is the managing member of HoldCo and owns a majority of the LLC Interests, and consolidates HoldCo in the Company’s Consolidated Financial Statements. The interest of the continuing LLC Owners in HoldCo is reflected in our Consolidated Financial Statements as a non-controlling interest.
Results of Operations
The following table sets forth our consolidated statements of operations in dollars and as a percentage of net sales for the periods presented:
| | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | % of Net Sales for the Years ended December 31, |
| $'s in 000's | 2023 | | 2022 | | 2023 | | 2022 |
| Product sales | $ | 968,151 | | | $ | 800,305 | | | 87.9% | | 86.8% |
| Services revenue | 133,812 | | | 121,208 | | | 12.1% | | 13.2% |
| Total net sales | 1,101,963 | | | 921,513 | | | 100.0% | | 100.0% |
| Cost of products sold | 732,422 | | | 606,548 | | | 66.5% | | 65.8% |
| Cost of services | 116,801 | | | 105,302 | | | 10.6% | | 11.4% |
| Total cost of sales | 849,223 | | | 711,850 | | | 77.1% | | 77.2% |
| Gross profit | 252,740 | | | 209,663 | | | 22.9% | | 22.8% |
| Operating expenses | | | | | | | |
| Selling, general and administrative expenses | 196,236 | | | 182,561 | | | 17.8% | | 19.8% |
| Restructuring | 11,751 | | | — | | | 1.1% | | —% |
| Impairment and other asset charges | 7,680 | | | 47,264 | | | 0.7% | | 5.1% |
| Operating income (loss) | 37,073 | | | (20,162) | | | 3.4% | | (2.2)% |
| Interest expense, net | 34,547 | | | 27,374 | | | 3.1% | | 3.0% |
| | | | | | | |
| | | | | | | |
| Other expense (income), net | 160 | | | (130) | | | —% | | —% |
| Total other expense, net | 34,707 | | | 27,244 | | | 3.1% | | 3.0% |
| Pretax net income (loss) | 2,366 | | | (47,406) | | | 0.2% | | (5.1)% |
| Income tax benefit (expense) | 173 | | | (1,214) | | | —% | | (0.1)% |
| Net income (loss) | $ | 2,539 | | | $ | (48,620) | | | 0.2% | | (5.3)% |
Year Ended December 31, 2023 Compared With Year Ended December 31, 2022
Net sales
Consolidated Net Sales
Consolidated net sales increased approximately $180.5 million, or 19.6%, to $1,102.0 million for the year ended December 31, 2023, compared to $921.5 million for the year ended December 31, 2022. Net sales growth of $167.9 million within the Products segment was driven by broad strength across most product categories on strong consumer demand, including distributed flea and tick, prescription medication, and manufacturing categories, as well as sales from the acquisition of R&R, which occurred in January 2023. The Services segment revenues grew by $12.6 million driven by increased average dollar per clinic and average dollar per pet served.
Products Segment
Product sales increased approximately $167.9 million, or 21.0%, to $968.2 million for the year ended December 31, 2023, compared to $800.3 million for the year ended December 31, 2022. The increase was driven by manufactured products which grew by 28% driven by broad strength across most categories as well as growth in distributed products of 20%.
Services Segment
Service revenue increased approximately $12.6 million, or 10.4%, from $121.2 million to $133.8 million for the year ended December 31, 2023, compared to the year ended December 31, 2022. The increased service revenue was driven by operational improvements which drove increases in clinic counts, average dollar per clinic, and average dollar per pet served.
Gross profit
Gross profit increased by approximately $43.0 million, or 20.5%, to $252.7 million for the year ended December 31, 2023, compared to $209.7 million for the year ended December 31, 2022. This increase is driven by the Products segment gross profit increasing by $42.0 million due to net sales growth.
Gross margin was up slightly at 22.9% for the year ended December 31, 2023, compared to 22.8% for the year ended December 31, 2022. The increased margin was primarily due to operational efficiencies and a favorable product mix offset by incremental costs from wellness centers closed as a part of service segment optimization.
Selling, general and administrative expenses
Consolidated selling, general and administrative expenses (“SG&A”) increased approximately $13.6 million, or 7.5%, to $196.2 million for the year ended December 31, 2023, compared to $182.6 million for the year ended December 31, 2022. This increase was primarily driven by the following:
•$10.4 million increase in a variety of marketing and advertising expenses,
•$4.0 million increase in variable selling expenses,
•$2.0 million increase in compensation costs,
•$1.7 million amortization expenses related to termination of an in process research and development agreement.
•Partially offset by $3.9 million decrease in legal expenses.
As a percentage of net sales, SG&A decreased from 19.8% for the year ended December 31, 2022 to 17.8% for the year ended December 31, 2023, this decrease was driven by operational efficiencies.
Products Segment
Products segment SG&A increased approximately $19.2 million, or 13.5%, to $161.6 million for the year ended December 31, 2023, compared to $142.4 million for the year ended December 31, 2022. This increase was driven by variable selling expenses on higher sales, professional services, amortization expenses related to termination of an in process research and development agreement, and increased marketing spend to support our branded portfolio, partially offset by decreased licensing fees.
Services Segment
Services segment SG&A decreased approximately $5.5 million, or 13.8%, to $34.6 million for the year ended December 31, 2023, compared to $40.1 million for the year ended December 31, 2022. This decrease was primarily driven by operational improvements, reduced marketing expenditures, and legal expenses incurred in the prior year for which there was no comparable event in 2023.
Restructuring
During the year ended December 31, 2023, the Company implemented a Services segment optimization (the "optimization") to improve the functioning of the Services segment and profitability. The optimization included assessing the operational and financial performance of the Company's wellness centers since re-opening after the pandemic as well as the assessment of the veterinary labor market in each geographic market. The Company also evaluated its ability to potentially convert these locations to a more hygiene-focused offering and determined they would be unable to convert these locations in the future based on the aforementioned assessment and the available square footage within the respective wellness centers. As a result of the optimization, the Company identified and closed 149 underperforming wellness centers during the year ended December 31, 2023. Restructuring totaled $11.8 million plus an additional $1.8 million included in cost of services equating to a total restructuring charge of $13.6 million and had no comparable event for the year ended December 31, 2022.
Impairment and other asset charges
During the year ended December 31, 2023, the Company committed to a plan to sell its foreign subsidiary, Mark & Chappell, and initiated the required actions to complete such a sale. In connection with the expected sale of the business and classification of related assets and liabilities as held for sale, we recorded a non-cash asset charge of $7.7 million for
the year ended December 31, 2023. During the year ended December 31, 2022, the Company recorded an impairment charge of $47.3 million related to the write-down of the full goodwill balance of the Services segment.
Interest expense, net
Interest expense, net, increased $7.1 million, to $34.5 million for the year ended December 31, 2023, compared to $27.4 million for the year ended December 31, 2022. This increase was driven by the higher rates on the Company's variable rate debt due to rising interest rates, partially offset by interest earned on the Company's cash and cash equivalents.
Pre-tax net income (loss)
As a result of the factors described above for the year ended December 31, 2023, pre-tax net income (loss) increased $49.8 million to a pre-tax net income of $2.4 million for the year ended December 31, 2023 compared to a pre-tax net loss of $47.4 million for the year ended December 31, 2022.
Tax benefit (expense)
The Company owns approximately 99.2% of HoldCo with the LLC Interests not held by the Company considered non-controlling interest. HoldCo is treated as a partnership for income tax reporting. HoldCo’s members, including the Company, are liable for federal, state, and local income taxes based on their share of HoldCo’s taxable income.
Income tax benefit (expense) totaled (7.3%) and (2.6%) of pre-tax net income (loss) for the years ended December 31, 2023 and 2022, respectively. Our tax rate is affected primarily by share-based compensation, return to provision adjustments, change in investments held for sale, changes in the valuation allowance, and state and local taxes during the year ended December 31, 2023. It is also affected by discrete items that may occur in any given year, which are not consistent from year to year.
Consolidated Non-GAAP Financial Measures
EBITDA and Adjusted EBITDA are non-GAAP financial measures. We calculate EBITDA as net income (loss) adjusted for income tax expense, depreciation, amortization, goodwill impairment, and interest expense, net. We calculate Adjusted EBITDA as EBITDA adjusted for acquisition costs, stock-based compensation expense, integration and business transformation, restructuring costs, litigation expenses, and other one-time transactions that management does not believe are representative of our core ongoing business. Adjusted EBITDA is utilized by management in evaluating the Company's performance and the effectiveness of our business strategies. The Company presents EBITDA because it is a necessary component for computing Adjusted EBITDA.
We believe that the use of EBITDA and Adjusted EBITDA provides an additional tool for investors to use in evaluating ongoing operating results and trends. In addition, you should be aware when evaluating EBITDA and Adjusted EBITDA that in the future we may incur expenses similar to those excluded when calculating these measures. Our presentation of these measures should not be construed as an inference that our future results will be unaffected by these or other unusual or non-recurring items. Our computation of EBITDA and Adjusted EBITDA may not be comparable to other similarly titled measures computed by other companies, because all companies do not calculate EBITDA and Adjusted EBITDA in the same manner.
Our management does not, and you should not, consider EBITDA or Adjusted EBITDA in isolation or as an alternative to financial measures determined in accordance with GAAP. The principal limitation of EBITDA and Adjusted EBITDA is that they exclude significant expenses and income that are required by GAAP to be recorded in our financial statements. Some of these limitations are:
•EBITDA does not reflect our cash expenditures, or future requirements, for capital expenditures or contractual commitments;
•EBITDA does not reflect changes in, or cash requirements for, our working capital needs;
•EBITDA does not reflect the interest expenses, or the cash requirements necessary to service interest or principal payments, on our debts;
•Although depreciation and amortization are non-cash charges, the assets being depreciated and amortized will often have to be replaced in the future, and Adjusted EBITDA does not reflect any cash requirements for such replacements;
•Adjusted EBITDA does not reflect the impact of certain cash charges resulting from matters we consider not to be indicative of our ongoing core operations; and
•Other companies in our industry may calculate Adjusted EBITDA differently than we do, limiting its usefulness as a comparative measure.
Because of these limitations, EBITDA and Adjusted EBITDA should not be considered in isolation or as a substitute for performance measures calculated in accordance with GAAP. We compensate for these limitations by relying primarily on our GAAP results and using EBITDA and Adjusted EBITDA only supplementary. You should review the reconciliations of net income (loss) to EBITDA and Adjusted EBITDA below and not rely on any single financial measure to evaluate our business.
The following table reconciles net income (loss) to EBITDA and Adjusted EBITDA for the periods presented.
| | | | | | | | | | | |
| Year Ended December 31, |
| $'s in 000's | 2023 | | 2022 |
| Net income (loss) | $ | 2,539 | | | $ | (48,620) | |
| Plus: | | | |
| Tax (benefit) expense | (173) | | | 1,214 | |
Depreciation(1) | 24,773 | | | 14,520 | |
| Amortization | 19,797 | | | 18,079 | |
Impairment and other asset charges(2) | 7,680 | | | 47,264 | |
| | | |
| Interest expense, net | 34,547 | | | 27,374 | |
| EBITDA | $ | 89,163 | | | $ | 59,831 | |
| | | |
Acquisition costs(3) | 1,164 | | | 1,464 | |
| Stock based compensation expense | 9,468 | | | 11,363 | |
Integration and business transformation costs(4) | 2,316 | | | 1,171 | |
| Litigation expenses | 31 | | | 3,862 | |
Restructuring(5) | 2,564 | | | — | |
| Adjusted EBITDA | $ | 104,706 | | | $ | 77,691 | |
(1) Depreciation includes $11.0 million of accelerated depreciation recognized during the year ended December 31, 2023, associated with Services segment optimization.
(2) Impairment and other asset charges includes asset charges associated with the Company committing to a plan to sell its foreign subsidiary, Mark & Chappell during the year ended December 31, 2023. For the year ended December 31, 2022, impairment and other asset charges includes write-down of the full goodwill balance of the Services segment.
(3) Acquisition costs include legal, accounting, banking, consulting, diligence, and other costs related to completed and contemplated acquisitions.
(4) Integration and business transformation costs, including personnel costs such as severance and retention bonuses, consulting costs, contract termination costs and IT and ERP implementation costs.
(5) Restructuring consists of variable lease expenses, inventory reserves, lease termination costs, severance, and other miscellaneous costs.
Financial Condition, Liquidity, and Capital Resources
Historically, our primary sources of liquidity have been cash flow from operations, borrowings, and equity contributions. As of December 31, 2023 and December 31, 2022, our cash and cash equivalents were $116.4 million and $101.3 million, respectively. As of December 31, 2023, we had an unused revolving credit facility with availability of $125.0 million,
$292.5 million outstanding under a term loan, $143.8 million of outstanding 4.0% Convertible Senior Notes due 2026 (the “Notes”), and $15.6 million in other debt. The debt agreements bear interest at rates between 4.00% and 10.17%.
Our primary cash needs are for working capital and to support our growth plans, which may include acquisitions. Our maintenance capital expenditures have typically been less than 1.0% of net sales, but we may make additional capital expenditures as necessary to support our growth. Our primary working capital requirements are to fund inventory and accounts receivable to support sales. Fluctuations in working capital are primarily driven by the timing of new product launches and seasonal retailer demand, which is typically higher in the first half of the year. As the flea and tick season ends in the second half of the year and retailers reduce inventory, working capital typically decreases. As of December 31, 2023 and December 31, 2022, we had working capital (current assets less current liabilities) of $241.3 million and $220.6 million, respectively. The Company has not historically made significant non-contractual debt pay downs, but may choose to do so in the future as part of its capital allocation strategy.
We believe that our operating cash flow, cash on hand, and debt proceeds from our borrowings under our debt facilities will be adequate to meet our operating, investing, and financing needs for at least the next 12 months. We believe we will meet our longer-term expected future cash requirements primarily from a combination of cash flow from operating activities, borrowings under our debt facilities and available cash and cash equivalents. To the extent additional funds are necessary to meet long-term liquidity needs as we continue to execute our business strategy, we anticipate that they will be obtained through the incurrence of additional indebtedness, equity financings, or a combination of these potential sources of funds, although we can provide no assurance that these sources of funding will be available on reasonable terms or at all. As in the past, we will continue to explore opportunities to optimize our capital structure.
Cash Flows
Cash provided by Operating Activities
Net cash provided by operating activities was $61.9 million for the year ended December 31, 2023, compared to $48.0 million for the year ended December 31, 2022. The change in operating cash flows is primarily attributed to $51.2 million increase in profitability offset by decreases in non-cash items of $33.4 million primarily due to $1.9 million decreased stock compensation in 2023 and goodwill impairment of $47.3 million which occurred in 2022 partially offset by accelerated depreciation and amortization in 2023 of $11.0 million incurred as a part of the service segment optimization and asset charges of $7.7 million in 2023. Additionally, there was a $3.9 million decrease in cash provided by working capital due to timing.
Cash used in Investing Activities
Net cash used in investing activities was $36.8 million for the year ended December 31, 2023, compared to $12.0 million for the year ended December 31, 2022. The increase in net cash used in investing activities was primarily the result of $27.6 million of the cash utilized in the acquisition of R&R partially offset by lower purchases of fixed assets.
Cash used in Financing Activities
Net cash used in financing activities was $10.2 million for the year ended December 31, 2023, compared to $13.7 million in net cash used in financing activities for the year ended December 31, 2022. The change in cash used in financing activities is primarily driven by $3.9 million stock repurchase made in the prior year period with no comparable events in the current period.
Description of Indebtedness
Senior Secured Asset-Based Revolving Credit Facility – ABL
On April 13, 2021, Opco entered into an asset-based revolving credit agreement with KeyBank National Association, as administrative agent and collateral agent, and the lenders’ party thereto, that provides revolving credit commitments of $125.0 million, subject to a borrowing base limitation (the “ABL Facility”). The borrowing base for the ABL Facility at any time equals the sum of: (i) 90% of eligible investment-grade accounts; plus (ii) 85% of eligible other accounts; plus, (iii) 85% of the net orderly liquidation value of the cost of certain eligible on-hand and in-transit inventory; plus, (iv) at the option of Opco, 100% of qualified cash; minus (v) reserves. The ABL Facility bears interest at a variable rate plus a margin, with the variable rate being based on a base rate or LIBOR at the option of the Company. On February 3, 2023, Opco entered into the First Amendment Agreement to the ABL Facility to replace the interest rate benchmark from LIBOR
to the Secured Overnight Financing Rate (“SOFR”). The rate at December 31, 2023 was 5.92%. The Company also pays a commitment fee on unused borrowings at a rate of 0.35%.
The ABL Facility is secured by substantially all the assets of HoldCo and its wholly-owned domestic subsidiaries including a first-priority security interest in personal property consisting of accounts receivable, inventory, cash, and deposit accounts (such collateral subject to such first-priority security interest, “ABL Priority Collateral”), and a second-priority security interest in all other personal and real property of HoldCo and its wholly-owned domestic subsidiaries (such collateral subject to such second-priority security interest, “Term Priority Collateral”), in each case, subject to customary exceptions. The ABL contains customary representations and warranties, affirmative and negative covenants and events of default, including negative covenants that restrict the ability of HoldCo and its restricted subsidiaries to incur additional indebtedness, pay dividends, make investments, loans, and acquisitions, among other restrictions.
Senior Secured Term Loan Facility – Term Loan B
On April 13, 2021, Opco entered into a term credit agreement with Jefferies Finance LLC, as administrative agent and collateral agent, and the lenders’ party thereto, that provides senior secured term loans of $300.0 million (the “Term Loan B”). The Term Loan B bears interest at a variable rate (with the variable rate being based on a base rate or LIBOR at the option of the Company) plus a margin of 3.25% in the case of base rate loans, or 4.25% in the case of LIBOR loans. LIBOR rates are subject to a 0.50% floor. The Term Loan B requires quarterly payments of 0.25% of the original principal amount, with the balance due on April 13, 2028. On May 25, 2023, the Term Loan B was amended to replace the interest rate benchmark from the Adjusted Eurodollar Rate to SOFR. The interest rate at December 31, 2023 was 10.17%.
The Term Loan B is secured by substantially all the assets of HoldCo and its wholly-owned domestic subsidiaries, including a first-priority security interest in Term Priority Collateral and a second-priority security interest in ABL Priority Collateral, in each case, subject to customary exceptions. The Term Loan B contains customary representations and warranties, affirmative and negative covenants and events of default, including negative covenants that restrict the ability of HoldCo and its restricted subsidiaries to incur additional indebtedness, pay dividends, make investments, loans, and acquisitions, among other restrictions.
The Company’s Term Loan B allows for certain incremental transactions such as additional borrowings, making restricted payments, or making acquisitions, as long as the Company’s net leverage ratio is within pre-established ranges that vary depending on the proposed incremental transaction. The Company’s net leverage ratio as defined in the Term Loan B as of December 31, 2023 was 2.9x.
Convertible Notes
On May 19, 2020, the Company issued $143.8 million in aggregate principal amount of 4.00% Convertible Senior Notes due 2026 (the “Notes”) pursuant to the indenture (the “Indenture”), dated as of May 19, 2020. The total net proceeds from the Notes offering, after deducting debt issuance costs paid or payable by us, was $137.9 million. The Notes accrue interest at a rate of 4.00% per annum, payable semi-annually in arrears on June 1 and December 1 of each year, beginning on December 1, 2020. The Notes will mature on June 1, 2026, unless earlier repurchased, redeemed or converted. Before January 15, 2026, holders will have the right to convert their Notes only upon the occurrence of certain events. From and after January 15, 2026, holders may convert their Notes at any time at their election until the close of business on the scheduled trading day immediately before the maturity date. The Company will settle conversions by paying or delivering, as applicable, cash, shares of its Class A common stock, or a combination of cash and shares of its Class A common stock, at its election. The initial conversion rate is 33.7268 shares of Class A common stock per $1,000 principal amount of Notes. The conversion rate and conversion price will be subject to customary adjustments upon the occurrence of certain events. In addition, if certain corporate events that constitute a “Make-Whole Fundamental Change” (as defined in the Indenture) occur, then the conversion rate will, in certain circumstances, be increased for a specified period of time.
The Notes are redeemable, in whole or in part, at the Company’s option at any time, and from time to time, on or after June 1, 2023 and on or before the 40th scheduled trading day immediately before the maturity date, at a cash redemption price equal to the principal amount of the Notes to be redeemed, plus accrued and unpaid interest, if any, to, but excluding, the redemption date, but only if the last reported sale price per share of the Company’s Class A common stock exceeds 130% of the conversion price on (i) each of at least 20 trading days, whether or not consecutive, during the 30 consecutive trading days ending on, and including, the trading day immediately before the date the Company sends the related redemption notice; and (ii) the trading day immediately before the date the Company sends such notice. In addition, calling any Notes will constitute a Make-Whole Fundamental Change with respect to such Notes, which will result in an increase to the conversion rate if such Notes are converted after they are called for redemption.
If certain corporate events that constitute a “Fundamental Change” (as defined in the Indenture) occur, then noteholders may require the Company to repurchase their Notes at a cash repurchase price equal to the principal amount of the Notes to be repurchased, plus accrued and unpaid interest, if any, to, but excluding, the fundamental change repurchase date. The definition of Fundamental Change includes certain business combination transactions involving the Company and certain de-listing events with respect to the Company’s Class A common stock.
The Notes are the Company’s senior, unsecured obligations and are (i) equal in right of payment with the Company’s existing and future senior, unsecured indebtedness; (ii) senior in right of payment to the Company’s existing and future indebtedness that is expressly subordinated to the Notes; (iii) effectively subordinated to the Company’s existing and future secured indebtedness, to the extent of the value of the collateral securing that indebtedness; and (iv) structurally subordinated to all existing and future indebtedness and other liabilities, including trade payables, and (to the extent the Company is not a holder thereof) preferred equity, if any, of the Company’s subsidiaries. The Notes contain customary events of default.
The fair value of the Notes was $146.1 million as of December 31, 2023. The estimated fair value of the Notes is based on market rates at the closing trading price of the Convertible Notes as of December 31, 2023 and is classified as Level 2 in the fair value hierarchy.
Capped Call Transactions
On May 14, 2020 and May 19, 2020, the Company entered into capped call transactions (the “Capped Call Transactions”) with two counterparties. The Capped Call Transactions cover, subject to anti-dilution adjustments substantially similar to the Notes, the underlying shares of Class A common stock and are intended to reduce, subject to a limit, the potential dilution with respect to the Class A common stock upon conversion of the Notes. The cap price of the Capped Call Transactions is $41.51 per share of Class A common stock, and is subject to certain adjustments under the terms of the Capped Call Transactions.
For additional discussion of our “indebtedness,” refer to Note 5 – “Debt” in the notes to our audited consolidated financial statements included in this Annual Report.
Contractual Obligations and Commitments
The following table summarizes our contractual obligations as of December 31, 2023:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Payment Due by Period |
| $'s in 000's | | Total | | 2024 | | 2025-2026 | | 2027-2028 | | Thereafter |
| Long-term debt | | $ | 451,814 | | | $ | 7,400 | | | $ | 151,914 | | | $ | 284,700 | | | $ | 7,800 | |
| Interest on debt | | 169,339 | | | 37,157 | | | 72,514 | | | 59,086 | | | 582 | |
| Operating lease obligations | | 23,533 | | | 8,468 | | | 10,159 | | | 4,729 | | | 177 | |
| Finance lease obligations | | 1,849 | | | 1,288 | | | 458 | | | 103 | | | — | |
| Product purchase obligations | | 74,432 | | | 72,534 | | | 1,898 | | | — | | | — | |
| Total contractual obligations | | $ | 720,967 | | | $ | 126,847 | | | $ | 236,943 | | | $ | 348,618 | | | $ | 8,559 | |
Critical Accounting Policies and Estimates
Our accounting policies have been established to conform to GAAP. The preparation of financial statements in accordance with GAAP requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the revenue and expenses incurred during the reported periods. On an ongoing basis, we evaluate our estimates and judgments, including those related to accrued expenses and stock-based compensation to ensure they are reasonable for reporting purposes. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not apparent from other sources. Changes in estimates are reflected in reported results for the period in which they become known. However, actual results may differ from these estimates under different assumptions or conditions.
This summary should be read in conjunction with the more complete discussion of our accounting policies and procedures included in Note 1 - Principal business Activity and Significant Accounting Policies in the notes to our consolidated financial statements.
Inventories
Inventories are stated at the lower of cost or net realizable value. Cost is determined on the first-in first-out (“FIFO”) method and includes estimated rebate amounts. The Company maintains reserves for estimated obsolete or unmarketable inventory based on the difference between the cost of inventory and its estimated net realizable value. In estimating the reserves, management considers factors such as excess or slow-moving inventories, product expiration dating, and market conditions. Changes in these conditions may result in additional reserves.
Impairment of Goodwill
Goodwill is the excess of the consideration paid over the fair value of specifically identifiable assets, liabilities and contingent liabilities in a business combination. Intangible assets acquired are recorded at estimated fair value. Goodwill and intangible assets deemed to have indefinite lives are not amortized, but are tested for impairment annually during the fourth quarter, and at any time when events suggest an impairment more likely than not occurred.
To assess goodwill for impairment, the Company, depending on relevant facts and circumstances, performs either a qualitative assessment or a quantitative analysis utilizing a discounted cash flow valuation model. In performing the qualitative assessment, the Company evaluates relevant factors such as macroeconomic conditions, industry and market considerations, cost factors and overall financial performance, as well as company and reporting unit specific items. If, after assessing these qualitative factors, the Company determines that it is more likely than not that the carrying value of the reporting unit is less than its fair value, then no further testing is required. In performing a quantitative analysis, the Company determines the fair value of a reporting unit using management’s assumptions about future cash flows based on long-range strategic plans. This approach incorporates many assumptions including discount rates and future growth rates. In the event the carrying amount of a reporting unit exceeded its fair value, an impairment loss would be recognized.
During the third quarter of 2022, the Company's market capitalization declined significantly, driven by rising interest rates and macroeconomic conditions. Additionally, the Company slowed its expansion plans for the Services reporting unit. Based on these events, the Company concluded that an indicator of impairment existed for the Services reporting unit related to goodwill. As a result of the Company's impairment test, the Company determined that the fair value of the Services reporting unit was less than it's carrying value resulting in a non-cash goodwill impairment charge of $47.3 million, representing all of the goodwill in the Services reporting unit, during the year ended December 31, 2022. The associated market capitalization reconciliation indicated that increasing the weighted average cost of capital used in the analysis of the Products reporting could indicate impairment to that reporting unit. Subsequent to the quantitative assessment performed in prior year for the products segment, the Company's stock price and market capitalization has increased significantly. We performed a qualitative assessment in the current year, noting no indicators of impairment. No impairment was recognized for the years ended December 31, 2023 and 2021, respectively. For further discussion and details relating to the Company’s goodwill balances refer to Note 4 – Intangible Assets and Goodwill in the notes to our audited consolidated financial statements of this Annual Report.
Accounting for Income Taxes
The Company’s annual income tax rate is based on its income, statutory tax rates, changes in prior tax positions and tax planning opportunities available in the various jurisdictions in which it operates. Significant judgment and estimates are required to determine the Company’s annual tax rate and evaluate its tax positions. Despite the Company’s belief that its tax return positions are fully supportable, these positions are subject to challenge, and the Company may not be successful in defending these challenges.
Deferred income taxes arise from temporary differences between the tax basis of assets and liabilities and their reported amounts in the financial statements, which will result in taxable or deductible amounts in the future. In evaluating our ability to recover our deferred tax assets in the jurisdiction from which they arise, we consider all available positive and negative evidence, including scheduled reversals of deferred tax liabilities, projected future taxable income, tax-planning strategies, and results of recent operations. In the event that it is determined that an asset is not more likely than not to be realized, a valuation allowance is recorded against the asset. Valuation allowances related to deferred tax assets can be impacted by changes to tax laws, changes to statutory tax rates and future taxable income levels. In the event the Company
were to determine that it would not be able to realize all or a portion of its deferred tax assets in the future, the unrealizable amount would be charged to earnings in the period in which that determination is made. Conversely, if the Company were to determine that it would be able to realize deferred tax assets in the future in excess of the net carrying amounts, it would decrease the recorded valuation allowance through a favorable adjustment to earnings in the period that the determination was made. The Company has assessed the realizability of the net deferred tax assets as of December 31, 2023 and in that analysis has considered the relevant positive and negative evidence available to determine whether it is more likely than not that some portion or all of the deferred income tax assets will not be realized. The Company believes it is more likely than not that the benefit from the recorded deferred tax assets will not be realized and has recorded a valuation allowance.
Based on our current earnings and anticipated future earnings, we believe that there is a reasonable possibility that in the foreseeable future, sufficient positive evidence may become available that results in a conclusion that all or a portion of the valuation allowance will no longer be needed. Release of the valuation allowance would result in the recognition of certain deferred tax assets and a decrease to income tax expense for the period the release is recorded. However, the exact timing and amount of the valuation allowance release are subject to change on the basis of the level of profitability that we are able to actually achieve.
Item 7A – Quantitative and Qualitative Disclosures About Market Risk
We are exposed to certain market risks arising from transactions in the normal course of our business. Such risk is principally associated with interest rates. We currently do not enter into derivatives or other financial instruments for trading or speculative purposes.
Interest Rate Risk
We are exposed to changes in interest rates because the indebtedness incurred under our ABL Facility and Term Loan B are variable rate debt. Interest rate changes generally do not affect the market value of our credit agreement but do affect the amount of our interest payments and, therefore, our future earnings and cash flows. As of December 31, 2023, we had variable rate debt of approximately $292.5 million under our ABL Facility and Term Loan B. An increase of 1% would have increased our interest expense for the year ended December 31, 2023 by approximately $2.9 million.
Item 8 – Financial Statements and Supplementary Data
Table of Contents
KPMG LLP
Suite 600
205 North 10th Street
Boise, ID 83702-5798
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors
PetIQ, Inc.:
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of PetIQ, Inc. and subsidiaries (the Company) as of December 31, 2023 and 2022, the related consolidated statements of operations, comprehensive income (loss), stockholders’ equity, and cash flows for each of the years in the three-year period ended December 31, 2023, and the related notes (collectively, the consolidated financial statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the years in the three-year period ended December 31, 2023, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company’s internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission, and our report dated February 29, 2024 expressed an unqualified opinion on the effectiveness of the Company’s internal control over financial reporting.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the consolidated financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of a critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
KPMG LLP, a Delaware limited liability partnership and a member firm of
the KPMG global organization of independent member firms affiliated with
KPMG International Limited, a private English company limited by guarantee.
Evaluation of the assessment of the realizability of deferred tax assets
As discussed in Notes 1 and 7 to the consolidated financial statements, the Company has assessed the realizability of the deferred tax assets as of December 31, 2023, and has considered the relevant positive and negative evidence available to determine whether it is more likely than not that some portion, or all, of the deferred tax assets will not be realized. The realization of the deferred tax assets is dependent on several factors, including the generation of sufficient taxable income to realize its deferred tax assets. The Company believes it is more likely than not that the benefit from the recorded deferred tax assets will not be realized. The Company has recorded a valuation allowance for these deferred tax assets of $112.2 million as of December 31, 2023.
We identified the evaluation of the realizability of the US deferred tax assets as a critical audit matter. Subjective auditor judgment, including the involvement of tax professionals with specialized skills and knowledge, was required to evaluate the key evidence, including the amount of, adjustments to, and timing of cumulative losses in recent years, recent period pretax income trends, and expected increases in future taxable income from restructuring activities as it relates to the Company’s determination of whether it is more likely than not that some portion, or all, of the deferred tax assets will not be realized. Changes in the estimated impact of key evidence on future earnings potential could have a significant impact on the realization of the Company’s deferred tax assets and the amount of the valuation allowance.
The following are the primary procedures we performed to address this critical audit matter. We evaluated the design and tested the operating effectiveness of certain internal controls related to the Company’s assessment of the realizability of the US deferred tax assets. This included controls related to the evaluation of the key evidence. We evaluated the amount of, adjustments to, and timing of cumulative losses in recent years by assessing the appropriateness of adjustments to historical amounts as well as the weight given to the key evidence in the context of the applicable accounting standards. We evaluated recent period pretax income trends and expected increases in future taxable income from restructuring activities by performing sensitivity analyses over these pieces of evidence to determine the potential impact they could have on the realizability of the US deferred tax assets. We involved tax professionals with specialized skills and knowledge, who assisted in evaluating the Company’s assessment of realizability of the US deferred tax assets and the resulting valuation allowance by assessing the appropriateness of management's evaluation of the impact of the key evidence on the realizability of the US deferred tax assets in the context of the applicable accounting standards.
We have served as the Company’s auditor since 2014.
Boise, Idaho
February 29, 2024
| | |
PetIQ, Inc. Consolidated Balance Sheets (In 000’s except for per share amounts) |
| | | | | | | | | | | | | | |
| | | | |
| | December 31, 2023 | | December 31, 2022 |
| Current assets | | | | |
| Cash and cash equivalents | | $ | 116,369 | | | $ | 101,265 | |
| Accounts receivable, net | | 142,511 | | | 118,004 | |
| Inventories | | 159,309 | | | 142,605 | |
| Other current assets | | 12,645 | | | 8,238 | |
| Total current assets | | 430,834 | | | 370,112 | |
| Property, plant and equipment, net | | 57,097 | | | 73,395 | |
| Operating lease right of use assets | | 19,079 | | | 18,231 | |
| Other non-current assets | | 2,083 | | | 1,373 | |
| Intangible assets, net | | 159,729 | | | 172,479 | |
| Goodwill | | 199,404 | | | 183,306 | |
| Total assets | | $ | 868,226 | | | $ | 818,896 | |
| Liabilities and equity | | | | |
| Current liabilities | | | | |
| Accounts payable | | $ | 139,264 | | | $ | 112,995 | |
| Accrued wages payable | | 16,734 | | | 11,512 | |
| Accrued interest payable | | 6,636 | | | 1,912 | |
| Other accrued expenses | | 10,692 | | | 7,725 | |
| Current portion of operating leases | | 7,608 | | | 6,595 | |
| Current portion of long-term debt and finance leases | | 8,595 | | | 8,751 | |
| Total current liabilities | | 189,529 | | | 149,490 | |
| Operating leases, less current installments | | 13,763 | | | 12,405 | |
| Long-term debt, less current installments | | 437,820 | | | 443,276 | |
| Finance leases, less current installments | | 516 | | | 907 | |
| Other non-current liabilities | | 3,600 | | | 1,025 | |
| Total non-current liabilities | | 455,699 | | | 457,613 | |
| Equity | | | | |
| Additional paid-in capital | | 387,349 | | | 378,709 | |
Class A common stock, par value $0.001, 125,000 shares authorized; 29,570 and 29,348 shares issued, respectively | | 29 | | | 29 | |
Class B common stock, par value $0.001 per share, 8,402 shares authorized; 231 and 252 shares issued and outstanding, respectively | | — | | | — | |
Class A treasury stock, at cost, 373 and 373 shares, respectively | | (3,857) | | | (3,857) | |
| Accumulated deficit | | (160,602) | | | (162,733) | |
| Accumulated other comprehensive loss | | (1,706) | | | (2,224) | |
| Total stockholders' equity | | 221,213 | | | 209,924 | |
| Non-controlling interest | | 1,785 | | | 1,869 | |
| Total equity | | 222,998 | | | 211,793 | |
| Total liabilities and equity | | $ | 868,226 | | | $ | 818,896 | |
See accompanying notes to the consolidated financial statements.
| | |
PetIQ, Inc. Consolidated Statements of Operations (In 000’s except for per share amounts) |
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Product sales | $ | 968,151 | | | $ | 800,305 | | | $ | 825,395 | |
| Services revenue | 133,812 | | | 121,208 | | | 107,133 | |
| Total net sales | 1,101,963 | | | 921,513 | | | 932,528 | |
| Cost of products sold | 732,422 | | | 606,548 | | | 646,402 | |
| Cost of services | 116,801 | | | 105,302 | | | 99,733 | |
| Total cost of sales | 849,223 | | | 711,850 | | | 746,135 | |
| Gross profit | 252,740 | | | 209,663 | | | 186,393 | |
| Operating expenses | | | | | |
| Selling, general and administrative expenses | 196,236 | | | 182,561 | | | 170,521 | |
| Restructuring | 11,751 | | | — | | | — | |
| | | | | |
| Impairment and other asset charges | 7,680 | | | 47,264 | | | — | |
| Operating income (loss) | 37,073 | | | (20,162) | | | 15,872 | |
| Interest expense, net | 34,547 | | | 27,374 | | | 24,696 | |
| Loss on debt extinguishment | — | | | — | | | 5,453 | |
| Other expense (income), net | 160 | | | (130) | | | (1,763) | |
| Total other expense, net | 34,707 | | | 27,244 | | | 28,386 | |
| Pretax net income (loss) | 2,366 | | | (47,406) | | | (12,514) | |
| Income tax benefit (expense) | 173 | | | (1,214) | | | (3,869) | |
| Net income (loss) | 2,539 | | | (48,620) | | | (16,383) | |
| Net income (loss) attributable to non-controlling interest | 408 | | | (412) | | | (416) | |
| Net income (loss) attributable to PetIQ, Inc. | $ | 2,131 | | | $ | (48,208) | | | $ | (15,967) | |
| | | | | |
| Net income (loss) per share attributable to PetIQ, Inc. Class A common stock | | | | | |
| Basic | $ | 0.07 | | | $ | (1.65) | | | $ | (0.57) | |
| Diluted | $ | 0.07 | | | $ | (1.65) | | | $ | (0.57) | |
| Weighted Average shares of Class A common stock outstanding | | | | | |
| Basic | 29,135 | | | 29,159 | | | 28,242 | |
| Diluted | 29,530 | | | 29,159 | | | 28,242 | |
See accompanying notes to the consolidated financial statements.
| | |
PetIQ, Inc. Consolidated Statements of Comprehensive Income (Loss) ($’s in 000’s) |
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Net income (loss) | $ | 2,539 | | | $ | (48,620) | | | $ | (16,383) | |
| Foreign currency translation adjustment | 522 | | | (1,553) | | | (65) | |
| Comprehensive income (loss) | 3,061 | | | (50,173) | | | (16,448) | |
| Comprehensive income (loss) attributable to non-controlling interest | 412 | | | (425) | | | (417) | |
| Comprehensive income (loss) attributable to PetIQ, Inc. | $ | 2,649 | | | $ | (49,748) | | | $ | (16,031) | |
See accompanying notes to the consolidated financial statements.
| | |
PetIQ, Inc.
Consolidated Statements of Cash Flows
($’s in 000’s) |
| | | | | | | | | | | | | | | | | | | | |
| | Year Ended December 31, |
| | 2023 | | 2022 | | 2021 |
| Cash flows from operating activities | | | | | | |
| Net income (loss) | | $ | 2,539 | | | $ | (48,620) | | | $ | (16,383) | |
| Adjustments to reconcile net income (loss) to net cash provided by operating activities | | | | | | |
| Depreciation and amortization of intangible assets and loan fees | | 44,498 | | | 35,468 | | | 39,300 | |
| Loss on debt extinguishment | | — | | | — | | | 5,453 | |
| Loss (gain) on disposition of property, plant, and equipment | | 8 | | | 438 | | | (1,183) | |
| Stock based compensation expense | | 9,468 | | | 11,363 | | | 9,428 | |
| Deferred tax adjustment | | (172) | | | 599 | | | 3,487 | |
| Impairment and other asset charges | | 7,680 | | | 47,264 | | | — | |
| | | | | | |
| Other non-cash activity | | (85) | | | (385) | | | 233 | |
| Changes in assets and liabilities, net of business acquisition | | | | | | |
| Accounts receivable | | (24,457) | | | (4,137) | | | (11,197) | |
| Inventories | | (16,041) | | | (46,297) | | | 1,283 | |
| Other assets | | (4) | | | 1,093 | | | (1,380) | |
| Accounts payable | | 25,950 | | | 58,546 | | | (12,131) | |
| Accrued wages payable | | 5,342 | | | (1,225) | | | 2,194 | |
| Other accrued expenses | | 7,161 | | | (6,083) | | | 4,663 | |
| Net cash provided by operating activities | | 61,887 | | | 48,024 | | | 23,767 | |
| Cash flows from investing activities | | | | | | |
| Proceeds from disposition of property, plant, and equipment | | — | | | — | | | 5,132 | |
| Purchase of property, plant, and equipment | | (9,145) | | | (11,973) | | | (31,270) | |
| Business acquisitions (net of cash acquired) | | (27,634) | | | — | | | — | |
| Net cash used in investing activities | | (36,779) | | | (11,973) | | | (26,138) | |
| Cash flows from financing activities | | | | | | |
| Proceeds from issuance of long-term debt | | 35,000 | | | 59,000 | | | 642,568 | |
| Principal payments on long-term debt | | (42,600) | | | (66,600) | | | (597,071) | |
| Tax distributions to LLC Owners | | (165) | | | — | | | (70) | |
| Principal payments on finance lease obligations | | (1,494) | | | (1,493) | | | (1,926) | |
| Payment of deferred financing fees and debt discount | | — | | | — | | | (7,656) | |
| Tax withholding payments on Restricted Stock Units | | (984) | | | (875) | | | (937) | |
| Stock repurchase | | — | | | (3,857) | | | — | |
| Exercise of options to purchase Class A common stock | | 54 | | | 115 | | | 13,426 | |
| Net cash (used in) provided by financing activities | | (10,189) | | | (13,710) | | | 48,334 | |
| Net change in cash and cash equivalents | | 14,919 | | | 22,341 | | | 45,963 | |
| Effect of exchange rate changes on cash and cash equivalents | | 185 | | | (482) | | | (13) | |
| Cash and cash equivalents, beginning of period | | 101,265 | | | 79,406 | | | 33,456 | |
| Cash and cash equivalents, end of period | | $ | 116,369 | | | $ | 101,265 | | | $ | 79,406 | |
See accompanying notes to the consolidated financial statements.
| | |
PetIQ, Inc.
Consolidated Statements of Cash Flows
($’s in 000’s) |
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| Supplemental cash flow information | 2023 | | 2022 | | 2021 |
| Interest paid, net | $ | 27,194 | | | $ | 26,404 | | | $ | 19,189 | |
| Net change in property, plant, and equipment acquired through accounts payable | 89 | | | 509 | | | 735 | |
| Finance lease additions | 775 | | | 59 | | | 1,191 | |
| Net change of deferred tax asset from step-up in basis | — | | | — | | | 3,348 | |
| Income taxes paid, net of refunds | 558 | | | 359 | | | 418 | |
| Net change in accrued tax distribution | 299 | | | — | | | 7 | |
See accompanying notes to the consolidated financial statements.
| | |
PetIQ, Inc. Consolidated Statements of Stockholders’ Equity (In 000’s) |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Accumulated
Deficit | | Accumulated
Other
Comprehensive
Loss | | Class A Common | | Treasury Stock | | Class B Common | | Additional
Paid-in
Capital | | Non-controlling
Interest | | Total
Equity |
| | | | Shares | | Dollars | | Shares | | Dollars | | Shares | | Dollars | | | |
| Balance - January 1, 2021 | | $ | (98,558) | | | $ | (686) | | | 25,711 | | | $ | 26 | | | — | | | $ | — | | | 3,040 | | | $ | 3 | | | $ | 319,642 | | | $ | 25,983 | | | $ | 246,410 | |
| Exchange of LLC Interests held by LLC Owners | | — | | | 66 | | | 2,768 | | | 3 | | | — | | | — | | | (2,768) | | | (3) | | | 23,531 | | | (23,597) | | | — | |
| Net increase in deferred tax asset from LLC Interest transactions | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | 3,235 | | | 113 | | | 3,348 | |
| Accrued tax distributions | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (7) | | | (7) | |
| Other comprehensive loss | | — | | | (64) | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (1) | | | (65) | |
| Stock based compensation expense | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | 9,109 | | | 319 | | | 9,428 | |
| Exercise of options to purchase common stock | | — | | | — | | | 583 | | | — | | | — | | | — | | | — | | | — | | | 13,426 | | | — | | | 13,426 | |
| Issuance of stock vesting of RSU's, net of tax withholdings | | — | | | — | | | 77 | | | — | | | — | | | — | | | — | | | — | | | (937) | | | — | | | (937) | |
| Net loss | | (15,967) | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (416) | | | (16,383) | |
| Balance - December 31, 2021 | | $ | (114,525) | | | $ | (684) | | | 29,139 | | | $ | 29 | | | — | | | $ | — | | | 272 | | | $ | — | | | $ | 368,006 | | | $ | 2,394 | | | $ | 255,220 | |
| Exchange of LLC Interests held by LLC Owners | | — | | | — | | | 20 | | | — | | | — | | | — | | | (20) | | | — | | | 199 | | | (199) | | | — | |
| | | | | | | | | | | | | | | | | | | | | | |
| Other comprehensive loss | | — | | | (1,540) | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (13) | | | (1,553) | |
| Treasury stock purchased | | — | | | — | | | — | | | — | | | 373 | | | (3,857) | | | — | | | — | | | — | | | — | | | (3,857) | |
| Stock based compensation expense | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | 11,264 | | | 99 | | | 11,363 | |
| Exercise of options to purchase common stock | | — | | | — | | | 2 | | | — | | | — | | | — | | | — | | | — | | | 115 | | | — | | | 115 | |
| Issuance of stock vesting of RSU's, net of tax withholdings | | — | | | — | | | 187 | | | — | | | — | | | — | | | — | | | — | | | (875) | | | — | | | (875) | |
| Net loss | | (48,208) | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (412) | | | (48,620) | |
| Balance - December 31, 2022 | | $ | (162,733) | | | $ | (2,224) | | | 29,348 | | | $ | 29 | | | 373 | | | $ | (3,857) | | | 252 | | | $ | — | | | $ | 378,709 | | | $ | 1,869 | | | $ | 211,793 | |
| Exchange of LLC Interests held by LLC Owners | | — | | | — | | | 21 | | | — | | | — | | | — | | | (21) | | | — | | | 180 | | | (180) | | | — | |
| Accrued tax distributions | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (394) | | | (394) | |
| Other comprehensive income | | — | | | 518 | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | 4 | | | 522 | |
| Stock based compensation expense | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | 9,390 | | | 78 | | | 9,468 | |
| Exercise of options to purchase common stock | | — | | | — | | | 3 | | | — | | | — | | | — | | | — | | | — | | | 54 | | | — | | | 54 | |
| Issuance of stock vesting of RSU's, net of tax withholdings | | — | | | — | | | 198 | | | — | | | — | | | — | | | — | | | — | | | (984) | | | — | | | (984) | |
| Net income | | 2,131 | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | — | | | 408 | | | 2,539 | |
| Balance - December 31, 2023 | | $ | (160,602) | | | $ | (1,706) | | | 29,570 | | | $ | 29 | | | 373 | | | $ | (3,857) | | | 231 | | | $ | — | | | $ | 387,349 | | | $ | 1,785 | | | $ | 222,998 | |
Note that certain figures shown in the table above may not recalculate due to rounding.
See accompanying notes to the consolidated financial statements.
Notes to the consolidated financial statements
Note 1 – Principal Business Activity and Significant Accounting Policies
Principal Business Activity and Principals of Consolidation
PetIQ is a leading pet medication, product and wellness company delivering a smarter way for pet parents to help pets live their best lives through convenient access to affordable health and wellness products and veterinary services. We have two reporting segments: (i) Products; and (ii) Services. PetIQ believes that pets are an important part of the family and deserve the best products and care we can provide.
Our Products segment consists of our product manufacturing and distribution business through which we manufacture and distribute pet medication and health and wellness products to major U.S. retail and e-commerce channels through more than 60,000 points of distribution. We focus our offerings on innovative, proprietary value-branded products, and leading third-party branded products for dogs and cats, including pet Rx medications, OTC medications and wellness products. Our Products segment is further supported by our world-class medications manufacturing facility in Omaha, Nebraska and health and wellness manufacturing facility in Springville, Utah.
Our Services segment consists of veterinary services, and related product sales, provided by the Company directly to consumers. Our national veterinarian service platform operates at over 2,600 community clinic locations and wellness centers hosted at retailers across 39 states providing cost effective and convenient veterinary wellness services. We offer diagnostic tests, vaccinations, prescription medications, microchipping, grooming and hygiene and wellness checks.
PetIQ is the managing member of PetIQ Holdings, LLC (“HoldCo”), a Delaware limited liability company, which is the sole member of PetIQ, LLC (“Opco”) and, through HoldCo, operate and control all the business and affairs of Opco. PetIQ and HoldCo are holding companies with no other operations, cash flows, material assets or liabilities other than the equity interests in Opco.
The consolidated financial statements include the accounts of the Company and all majority-owned subsidiaries. All intercompany transactions and balances have been eliminated in consolidation.
Use of Estimates
The preparation of consolidated financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of sales and expenses during the reporting period. Actual results could differ from those estimates. Significant items subject to such estimates and assumptions include the useful lives of property, plant, and equipment and intangible assets; the valuation of property, plant, and equipment, intangible assets and goodwill, the valuation of assets and liabilities in connection with acquisitions, the valuation of deferred tax assets, the valuation of inventories, and reserves for legal contingencies.
Fair Value of Financial Instruments
Fair value is defined as the price that would be received to sell an asset or paid to transfer a liability (an exit price) in an orderly transaction between market participants at the reporting date. The accounting guidance establishes a three-tiered hierarchy, which prioritizes the inputs used in the valuation methodologies in measuring fair value:
Level 1—Quoted prices in active markets for identical assets or liabilities.
Level 2—Inputs other than Level 1 that are observable, either directly or indirectly, such as quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities.
Level 3—Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
The categorization of a financial instrument within the valuation hierarchy is based on the lowest level of input that is significant to the fair value measurement.
The carrying amounts of the Company’s financial instruments, including cash and cash equivalents, accounts receivable, accounts payable and accrued liabilities, are at cost, which approximates fair value due to their relatively short maturities.
Cash and Cash Equivalents
Cash equivalents consist of highly liquid investments with an original maturity of three months or less at the date of acquisition. All credit card, debit card and electronic transfer transactions that process in less than seven days are classified as cash and cash equivalents. The Company maintains its cash accounts in various deposit accounts, the balances of which at times exceeded federal deposit insurance limits during the periods presented.
Receivables and Credit Policy
Trade receivables due from customers are uncollateralized customer obligations due under normal trade terms requiring payment within 45 days from the invoice date. Accounts receivable are stated at the amount billed to the customer, net of discounts and estimated deductions. The Company does not have a policy for charging interest on overdue customer account balances. The Company provides an allowance for credit losses equal to expected losses. The Company’s estimate is based on historical collection experience, a review of the current status of trade accounts receivable and known current economic conditions. Payments of trade receivables are allocated to the specific invoices identified on the customer’s remittance advice.
Other receivables consists of various receivables due from vendors, banking institutions, employees, and government agencies.
Accounts receivable, net consists of the following as of:
| | | | | | | | | | | |
| $'s in 000's | December 31, 2023 | | December 31, 2022 |
| Trade receivables | $ | 126,835 | | | $ | 104,612 | |
| Other receivables | 15,821 | | | 13,790 | |
| 142,656 | | | 118,402 | |
| Less: Allowance for doubtful accounts | (145) | | | (398) | |
| Total accounts receivable, net | $ | 142,511 | | | $ | 118,004 | |
Inventories
Inventories are stated at the lower of cost or net realizable value. Cost is determined on the FIFO method and includes estimated rebate amounts. The Company maintains reserves for estimated obsolete or unmarketable inventory based on the difference between the cost of inventory and its estimated net realizable value. In estimating the reserves, management considers factors such as excess or slow-moving inventories, product expiration dating, and market conditions. Changes in these conditions may result in additional reserves. Major components of inventories consist of the following as of:
| | | | | | | | | | | |
| $'s in 000's | December 31, 2023 | | December 31, 2022 |
| Raw materials | $ | 16,055 | | | $ | 17,464 | |
| Work in progress | 2,591 | | | 2,234 | |
| Finished goods | 140,663 | | | 122,907 | |
| Total inventories | $ | 159,309 | | | $ | 142,605 | |
Property, Plant, and Equipment
Property, plant, and equipment are recorded at cost. Expenditures for improvements that significantly add to the productive capacity or extend the useful life of an asset are capitalized. Expenditures for maintenance and repairs are charged to expense as incurred. Long-lived assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. No impairment charge was recorded for the years ended December 31, 2023, 2022 and 2021.
Depreciation and amortization is provided using the straight-line method, based on estimated useful lives of the assets, except for leasehold improvements and finance leased assets which are depreciated over the shorter of the expected useful life or the lease term. Depreciation and amortization expense is recorded in cost of sales and selling, general and administrative expenses in the consolidated statements of operations, depending on the use of the asset. The estimated useful lives of property, plant, and equipment are as follows:
| | | | | |
| Computer equipment and software | 3 years |
| Vehicle and vehicle accessories | 3-5 years |
| Buildings | 33 years |
| Equipment | 2-15 years |
| Leasehold improvements | 2-15 years |
| Furniture and fixtures | 5-10 years |
Goodwill and Intangible Assets
Goodwill is the excess of the consideration paid over the fair value of specifically identifiable assets, liabilities and contingent liabilities in a business combination. Intangible assets acquired are recorded at estimated fair value. Goodwill and intangible assets deemed to have indefinite lives are not amortized, but are tested for impairment annually during the fourth quarter, and at any time when events suggest an impairment more likely than not occurred.
To assess goodwill for impairment, the Company, depending on relevant facts and circumstances, performs either a qualitative assessment or a quantitative analysis utilizing a discounted cash flow valuation model. In performing the qualitative assessment, the Company evaluates relevant factors such as macroeconomic conditions, industry and market considerations, cost factors and overall financial performance, as well as company and reporting unit specific items. If, after assessing these qualitative factors, the Company determines that it is more likely than not that the carrying value of the reporting unit is less than its fair value, then no further testing is required. In performing a quantitative analysis, the Company determines the fair value of a reporting unit using management’s assumptions about future cash flows based on long-range strategic plans. This approach incorporates many assumptions including discount rates and future growth rates. In the event the carrying amount of a reporting unit exceeded its fair value, an impairment loss would be recognized.
As a result of the Company's annual impairment test, no impairment charges were recorded for the years ended December 31, 2023 and 2021, respectively. However, during the year ended December 31, 2022 the Company determined that the fair value of the Services reporting unit was less than it's carrying value, resulting in a non-cash goodwill impairment charge of $47.3 million.
Indefinite-lived intangible assets are tested for impairment utilizing either a qualitative assessment or a quantitative analysis. For a qualitative assessment, the Company identifies and considers relevant key factors, events, and circumstances to determine whether it is necessary to perform a quantitative impairment test. The key factors considered include macroeconomic, industry, and market conditions, as well as the asset's actual and forecasted results. For the quantitative impairment tests, the Company compares the carrying amounts to the current fair market values. Intangible assets with definite lives are amortized over their estimated useful lives to reflect the pattern over which the economic benefits of the intangible assets are consumed. Definite-lived intangible assets are also evaluated for impairment when impairment indicators are present. No impairment was recorded as a result of this test or was recorded for the years ended December 31, 2023, 2022, and 2021.
Assets and Liabilities Held for Sale
During the fourth quarter of 2023, the Company committed to a plan to sell its foreign subsidiary within the Products segment, Mark & Chappell ("M&C"), and initiated the required actions to complete such a sale. Based on the applicable requirements, the Company concluded that the M&C business met the held for sale criteria as of December 31, 2023. As such, the assets and liabilities of M&C are classified as held for sale and presented as $4.2 million of assets within Other current assets and $1.2 million of liabilities within Other accrued expenses in our Consolidated Balance Sheets as of December 31, 2023. In connection with the expected sale of the business and classification of related assets and liabilities as held for sale, we recorded asset charges of $7.7 million during the year ended December 31, 2023, which is included on our Consolidated Statements of Operations within Impairment and other asset charges. We expect the sale of Mark & Chappell will close during the first quarter of 2024.
Convertible Debt
On May 19, 2020, the Company issued $143.8 million aggregate principal amount of Convertible Notes due 2026 (the “Notes”). See Note 5 – “Debt.” Simultaneously, with the issuance of the Notes, we bought capped call options from certain financial institutions to minimize the impact of potential dilution of our Class A common stock upon conversion of the Notes. The premium for the capped call options was recorded as additional paid-in capital in our consolidated balance sheets as the options are settleable in our Class A common stock.
Revenue Recognition
When Performance Obligations Are Satisfied
A performance obligation is a promise in a contract to transfer a distinct good or service to the customer and is the unit of account for revenue recognition. A contract’s transaction price is allocated to each distinct performance obligation and recognized as revenue when, or as, the performance obligation is satisfied. The Company’s performance obligations are product sales and the delivery of veterinary services.
Revenue is generally recognized for product sales on a point in time basis when product control is transferred to the customer. In general, control transfers to the customer when the product is shipped or delivered to the customer based upon applicable shipping terms, as the customer can direct the use and obtain substantially all of the remaining benefits from the asset at this point in time.
Revenue for services is recognized over time as the service is delivered, typically over a single day. Payment is typically rendered at the time of service. Customer contracts generally do not include more than one performance obligation. When a contract does contain more than one performance obligation, we allocate the contract’s transaction price to each performance obligation based on its relative standalone selling price. The standalone selling price for each distinct good is generally determined by directly observable data. The Company began offering subscription based veterinary service plans to customers of the Services segment in 2021; however, total activity during the years ended December 31, 2023, 2022, and 2021 was immaterial.
The performance obligations in our contracts are satisfied within one year. As such, we have not disclosed the transaction price allocated to remaining performance obligations as of December 31, 2023 and 2022.
Variable Consideration
In addition to fixed contract consideration, most contracts include some form of variable consideration. The most common forms of variable consideration include discounts, rebates, and sales returns and allowances. Variable consideration is treated as a reduction in revenue when product revenue is recognized. Depending on the specific type of variable consideration, we use either the expected value or most likely amount method to determine the variable consideration. We believe there will not be significant changes to our estimates of variable consideration when any related uncertainties are resolved with our customers. The Company reviews and updates its estimates and related accruals of variable consideration each period based on the terms of the agreements, historical experience, and any recent changes in the market. Any uncertainties in the ultimate resolution of variable consideration due to factors outside of the Company’s influence are typically resolved within a short timeframe therefore not requiring any additional constraint on the variable consideration.
Trade marketing expense, consisting primarily of customer pricing allowances and merchandising funds are offered through various programs to customers and are designed to promote our products. They include the cost of in-store product displays, feature pricing in retailers' advertisements and other temporary price reductions. These programs are offered to our customers both in fixed and variable amounts. The ultimate cost of these programs depends on retailer performance and is subject to management estimates.
Certain retailers require the payment of product introductory fees in order to obtain space for the Company's products on the retailer's store shelves. This cost is typically a lump sum and is determined using the expected value based on the contract between the two parties.
Both trade marketing expense and product introductory fees are recognized as reductions of revenue at the time the transfer of control of the associated products occurs. Accruals for expected payouts, or amounts paid in advance, under these programs are included as accounts payable or other current assets in the consolidated balance sheets.
Significant Payment Terms
Our customer contracts identify the product, quantity, price, payment and final delivery terms. Payment terms usually include early pay discounts. We grant payment terms consistent with industry standards. Although some payment terms may be more extended, terms beyond one year are not typically granted at contract inception. As a result, we do not adjust the promised amount of consideration for the effects of a significant financing component because the period between our transfer of a promised good or service to a customer and the customer’s payment for that good or service is typically one year or less.
Shipping and other costs
All shipping and handling costs associated with outbound freight are accounted for as fulfillment costs and are included in the cost of sales. This includes shipping and handling costs after control over a product has transferred to a customer.
Sales tax collected from customers and remitted to governmental authorities is not included in revenue and is reflected as a liability on the Company’s consolidated balance sheets.
Warranties & Returns
PetIQ provides all customers with a standard or assurance type warranty. Either stated or implied, the Company provides assurance the related products will comply with all agreed-upon specifications and other warranties provided under the law. No significant services beyond an assurance warranty are provided to customers.
The Company does not grant a general right of return. However, customers may return defective or non-conforming products. Additionally, customers from time to time will negotiate certain return provisions to facilitate seasonal retail inventory changes or to reset placement within stores. Customer remedies may include either a cash refund or an exchange of the product. As a result, the right of return and related refund liability is estimated and recorded as a reduction in revenue. This return estimate is reviewed and updated each period and is based on historical sales and return experience.
Contract balances
Contract asset and liability balances as of December 31, 2023 and 2022 are immaterial. The Company does not have significant deferred revenue or unbilled receivable balances.
Cost of Services
Cost of Services are comprised of all service and product costs related to the delivery of veterinary services, including but not limited to, salaries and contract costs of veterinarians, technicians and other clinic based personnel, transportation and delivery costs, rent, occupancy costs, supply costs, depreciation and amortization of clinic assets, certain marketing and promotional expenses and costs of goods sold.
Research and Development and Advertising Costs
Research and development and advertising costs are expensed as incurred and are included in selling, general and administrative expenses. Research and development costs amounted to $1.1 million, $2.1 million, and $8.0 million and advertising costs were $45.6 million, $20.1 million, and $16.2 million for the years ended December 31, 2023, 2022, and 2021, respectively.
Collaboration Agreements
Through the Perrigo Animal Health Acquisition, we entered into a product development and asset purchase agreement with a third party for certain product formulations in development by the third party. During the year ended December 31, 2021, the Company opted out of the arrangement for two of the product formulations, and during the year ended December 31, 2023 the Company opted out of the final project. Product development costs were expensed as incurred or as milestone payments become probable, and the Company wrote off the remaining intangible asset upon abandonment of the project. All expenses are included in selling, general and administrative expenses on the consolidated statements of operations. The Company accrued $2.0 million in research and development expense within accounts payable in the consolidated balance sheets related to the agreement as a milestone was determined to be probable during the year ended December 31, 2021.
The amount was paid during the year ended December 31, 2022. There was $1.7 million of expenses incurred upon abandonment of the project for the period ended December 31, 2023 and no expenses incurred under the agreement for the period ended December 31, 2022. See Note 4 for more information.
Litigation
The Company is subject to various legal proceedings, claims, litigation, investigations and contingencies arising out of the ordinary course of business. If the likelihood of an adverse legal outcome is determined to be probable and the amount of loss is estimable, then a liability is accrued in accordance with accounting guidance for Contingencies. If the assessment indicates a potentially material loss contingency is not probable but is reasonably possible, or is probable but cannot be estimated, then the nature of the contingent liability, together with an estimate of the range of possible loss if determinable and material, is disclosed. The Company consults with both internal and external legal counsel related to litigation. See Note 13 for more information.
Stock Based Compensation
The Company expenses employee share-based awards under ASC Topic 718, Compensation—Stock Compensation, which requires compensation cost for the grant-date fair value of share-based awards to be recognized over the requisite service period. Stock options granted to executives and other employees are valued using the Black-Scholes option pricing model. See Note 9 for more information.
Accounting for Income Taxes
The Company uses the asset and liability approach for financial accounting and reporting of income taxes. Deferred income taxes reflect the net tax effect of temporary differences between the carrying amount of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. Deferred taxes are measured using rates expected to apply to taxable income in years in which those temporary differences are expected to reverse. The effect of a change in tax rates on deferred tax assets and liabilities is recognized in income in the period that includes the enactment date.
We recognize deferred tax assets to the extent that we believe that these assets are more likely than not to be realized. In making such a determination, we consider all available positive and negative evidence, including future reversals of existing taxable temporary differences, projected future taxable income, tax-planning strategies, and results of recent operations. If we determine that we would be able to realize our deferred tax assets in the future in excess of their net recorded amount, we would make an adjustment to the deferred tax asset valuation allowance, which would reduce the provision for income taxes.
The Company uses a two-step process for the measurement of uncertain tax positions that have been taken or are expected to be taken in a tax return. The first step is a determination of whether the tax position should be recognized in the consolidated financial statements. The second step determines the measurement of the tax position. The Company records potential interest and penalties on uncertain tax positions as a component of income tax expense.
Interest Expense, net
Interest expense, net, is comprised primarily of interest expense related to (i) our debt agreements, (ii) unused line fees, (iii) amortization of deferred loan fees and discounts, (iv) finance lease obligations and the mortgage note outstanding, offset by interest income earned on our demand deposits and other assets. Interest expense was $38.3 million, $28.2 million, and $24.7 million for the years ended December 31, 2023, 2022, and 2021, respectively, offset by $3.7 million, $0.8 million, and $0.0 million of interest income, respectively.
Non-Controlling Interest
The non-controlling interests on the consolidated statements of operations represents the portion of earnings or loss attributable to the economic interest in the Company’s subsidiary, HoldCo, held by the non-controlling holders of Class B common stock and limited liability company interests in HoldCo. Non-controlling interests on the consolidated balance sheet represents the portion of net assets of the Company attributable to the non-controlling holders of Class B common stock and Limited Liability Company interests in HoldCo.
Earnings (Loss) Per Share
Basic earnings (loss) per share is computed by dividing net income (loss) attributable to PetIQ, Inc. by the weighted average shares outstanding during the period. Diluted earnings (loss) per share is computed by dividing net income (loss) attributable to PetIQ, Inc., adjusted as necessary for the impact of potentially dilutive securities, by the weighted-average shares outstanding during the period and the impact of securities that would have a dilutive effect on income (loss) per share. See Note 8 for further discussion.
Recently Issued Accounting Pronouncements / Adopted Accounting Standard Updates
From time to time, the Financial Accounting Standards Board or other standards setting bodies issue new accounting pronouncements. Updates to the Accounting Standards Codification are communicated through issuance of an Accounting Standards Update (“ASU”)
In November of 2023, the Financial Accounting Standards Board (FASB) issued ASU No. 2023-07, Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures. The amendments are intended to increase reportable segment disclosure requirements primarily through enhanced disclosures about significant segment expenses. The ASU is effective on a retrospective basis for fiscal years beginning after December 15, 2023, and interim periods within fiscal years beginning after December 15, 2024. We are currently evaluating the impact of this guidance on the disclosures within our consolidated financial statements.
In December 2023, the FASB issued ASU No. 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures. The amendments require disclosure of specific categories in the rate reconciliation and provide additional information for reconciling items that meet a quantitative threshold and further disaggregation of income taxes paid for individually significant jurisdictions. The ASU is effective for fiscal years beginning after December 15, 2024, with early adoption permitted. We are currently evaluating the impact of this guidance on the disclosures within our consolidated financial statements.
Note 2 – Business Combination
Rocco & Roxie
The Company completed the acquisition of all of the membership units of Rocco & Roxie Supply Co, LLC ("R&R") on January 13, 2023 (the "R&R Acquisition"), which resulted in R&R becoming a wholly owned subsidiary of the Company. The R&R Acquisition expands the Company's brand and product portfolio to include stain and odor products and enables the Company to extend its offerings into premium dog supplements and jerky treats. The Company paid $26.5 million for the membership interests of R&R using cash on hand. The purchase was subject to normal working capital adjustments.
The following table summarizes the final allocations of the consideration paid of $27.6 million, which included the $26.5 million purchase price plus $1.1 million of working capital, to the assets acquired and liabilities assumed, based on the fair value at the date of the R&R Acquisition:
| | | | | | | | | | | |
| $'s in 000's | | | | | Fair Value |
| Current assets | | | | | $ | 3,020 | |
| Other assets | | | | | 1,208 | |
| Amortizable intangibles | | | | | |
| Trade name | | | | | 7,100 | |
| Customer relationships | | | | | 320 | |
| Total amortizable intangibles | | | | | $ | 7,420 | |
| Goodwill | | | | | 20,641 | |
| Total assets | | | | | $ | 32,289 | |
| | | | | |
| Current liabilities | | | | | 1,000 | |
| Other tax liabilities | | | | | 3,655 | |
| Total liabilities | | | | | 4,655 | |
| | | | | |
| Purchase price, net of cash acquired | | | | | $ | 27,634 | |
Intangible assets will be amortized over the estimated useful lives of the assets through January 2033. The weighted average amortization period of the amortizable intangible assets is approximately 9.9 years. The identifiable intangible assets are measured at fair value as Level III in accordance with the fair value hierarchy.
Goodwill represents the future economic benefits that do not qualify for separate recognition and primarily includes the assembled workforce and other non-contractual relationships, as well as expected future synergies. Approximately $19.4 million of the $20.6 million of Goodwill will not be tax deductible, and the remaining balance is expected to be deductible for tax purposes. Goodwill was allocated to the Products segment. Transaction costs of $0.5 million were incurred and are recorded in Selling, General, and Administrative costs on the consolidated statement of operations.
Note 3 – Property, Plant, and Equipment
Property, plant, and equipment consists of the following at:
| | | | | | | | | | | |
| $'s in 000's | December 31, 2023 | | December 31, 2022 |
| Leasehold improvements | $ | 18,379 | | | $ | 27,694 | |
| Equipment | 26,892 | | | 27,187 | |
| Vehicles and accessories | 5,592 | | | 6,771 | |
| Computer equipment and software | 21,256 | | | 17,501 | |
| Buildings | 22,733 | | | 26,176 | |
| Furniture and fixtures | 3,481 | | | 3,499 | |
| Land | 8,934 | | | 8,934 | |
| Construction in progress | 4,234 | | | 4,797 | |
| 111,501 | | | 122,559 | |
| Less accumulated depreciation | (54,404) | | | (49,164) | |
| Total property, plant, and equipment | $ | 57,097 | | | $ | 73,395 | |
Depreciation and amortization expense related to these assets total $24.8 million, $14.5 million, and $14.4 million for the years ended December 31, 2023, 2022, and 2021, respectively. Depreciation for the year ended December 31, 2023, 2022, and 2021 includes approximately $8.2 million, $0.0 million, and $2.0 million of accelerated depreciation related to fixed assets with shortened useful lives, respectively.
Note 4 – Intangible Assets and Goodwill
Intangible assets consist of the following at:
| | | | | | | | | | | | | | | | | | | | |
| $'s in 000's | | Useful Lives | | December 31, 2023 | | December 31, 2022 |
| Amortizable intangibles | | | | | | |
| Certification | | 7 years | | $ | 350 | | | $ | 350 | |
| Customer relationships | | 12-20 years | | 159,291 | | | 160,040 | |
| Patents and processes | | 5-10 years | | 12,855 | | | 14,634 | |
| Brand names | | 5-15 years | | 30,906 | | | 24,633 | |
| Total amortizable intangibles | | | | 203,402 | | | 199,657 | |
| Less accumulated amortization | | | | (76,912) | | | (62,085) | |
| Total net amortizable intangibles | | | | 126,490 | | | 137,572 | |
| Non-amortizable intangibles | | | | | | |
| Trademarks and other | | | | 33,239 | | | 33,239 | |
| In-process research and development | | | | — | | | 1,668 | |
| Intangible assets, net of accumulated amortization | | | | $ | 159,729 | | | $ | 172,479 | |
Certain intangible assets are denominated in currencies other than the U.S. Dollar; therefore, their gross and net carrying values are subject to foreign currency movements. Amortization expense for the years ended December 31, 2023, 2022, and 2021 was $19.8 million, $18.1 million, and $22.3 million, respectively.
During the year ended December 31, 2023, the Company opted out of the final acquired project from the Perrigo Animal Health acquisition included in in-process research and development, effectively abandoning the associated research and development effort. Accordingly, the Company wrote off the associated intangible asset of $1.7 million in the Products segment, the expense for which is included as amortization expense in selling, general and administrative expenses on the consolidated statement of operations for the year ended December 31, 2023.
Estimated future amortization expense for each of the following years is as follows:
| | | | | |
| Years ending December 31, ($'s in 000's) | |
| 2024 | $ | 15,876 | |
| 2025 | 14,899 | |
| 2026 | 14,303 | |
| 2027 | 13,640 | |
| 2028 | 12,295 | |
| Thereafter | 55,477 | |
The following is a summary of the changes in the carrying value of goodwill for the years ended December 31, 2023 and 2022.
| | | | | | | | | | | | | | | | | | | | |
| | Reporting Unit | | |
| ($'s in 000's) | | Products | | Services | | Total |
| Goodwill as of January 1, 2022 | | $ | 183,846 | | | $ | 47,264 | | | $ | 231,110 | |
| Foreign currency translation | | (540) | | | — | | | (540) | |
| Impairment | | — | | | (47,264) | | | (47,264) | |
| Goodwill as of December 31, 2022 | | 183,306 | | | — | | | 183,306 | |
| Foreign currency translation | | 252 | | | — | | | 252 | |
| R&R Acquisition | | 20,641 | | | — | | | 20,641 | |
| Transfer to held for sale | | (4,795) | | | — | | | (4,795) | |
| Goodwill as of December 31, 2023 | | $ | 199,404 | | | $ | — | | | $ | 199,404 | |
Note 5 – Debt
On April 13, 2021, the Company entered into the ABL Facility and the Term Loan B (each as defined below), which replaced both the Amended Revolving Credit Agreement on July 8, 2019 (the "Amended Revolving Credit Agreement") and A&R Term Loan Agreement as well as fully repaid $27.5 million in other debt.
As part of the termination of the Amended Revolving Credit Agreement and the A&R Term Loan Credit agreement, and the repayment in full of other debt, the Company wrote off $5.5 million in deferred financing fees to loss on debt extinguishment and incurred an additional $0.9 million in costs related to the transaction which are included in selling, general and administrative expenses for the year ended December 31, 2021.
Senior Secured Asset-Based Revolving Credit Facility – ABL
On April 13, 2021, Opco entered into an asset-based revolving credit agreement with KeyBank National Association, as administrative agent and collateral agent, and the lenders’ party thereto, that provides revolving credit commitments of $125.0 million, subject to a borrowing base limitation (the “ABL Facility”). The borrowing base for the ABL Facility at any time equals the sum of: (i) 90% of eligible investment-grade accounts; plus (ii) 85% of eligible other accounts; plus, (iii) 85% of the net orderly liquidation value of the cost of certain eligible on-hand and in-transit inventory; plus, (iv) at the option of Opco, 100% of qualified cash; minus (v) reserves. The ABL Facility bears interest at a variable rate plus a margin, with the variable rate being based on a base rate or London Interbank Offered Rate, ("LIBOR") at the option of the Company. On February 3, 2023, Opco entered into the First Amendment Agreement to the ABL Facility to replace the interest rate benchmark from LIBOR to the Secured Overnight Financing Rate (“SOFR”). The rate at December 31, 2023 was 5.92%. The Company also pays a commitment fee on unused borrowings at a rate of 0.35%.
The ABL Facility is secured by substantially all the assets of HoldCo and its wholly-owned domestic subsidiaries including a first-priority security interest in personal property consisting of accounts receivable, inventory, cash, and deposit accounts (such collateral subject to such first-priority security interest, “ABL Priority Collateral”), and a second-priority security interest in all other personal and real property of HoldCo and its wholly-owned domestic subsidiaries (such collateral subject to such second-priority security interest, “Term Priority Collateral”), in each case, subject to customary exceptions. The ABL contains customary representations and warranties, affirmative and negative covenants and events of default, including negative covenants that restrict the ability of HoldCo and its restricted subsidiaries to incur additional indebtedness, pay dividends, make investments, loans, and acquisitions, among other restrictions. No amounts were outstanding under the ABL as of December 31, 2023.
Senior Secured Term Loan Facility – Term Loan B
On April 13, 2021, Opco entered into a term credit agreement with Jefferies Finance LLC, as administrative agent and collateral agent, and the lenders’ party thereto, that provides senior secured term loans of $300.0 million (the “Term Loan B”). The Term Loan B bears interest at a variable rate (with the variable rate being based on a base rate or LIBOR at the option of the Company) plus a margin of 3.25% in the case of base rate loans, or 4.25% in the case of LIBOR loans. LIBOR rates are subject to a 0.50% floor. The Term Loan B requires quarterly payments of 0.25% of the original principal
amount, with the balance due on April 13, 2028. On May 25, 2023, the Term Loan B was amended to replace the interest rate benchmark from the Adjusted Eurodollar Rate to SOFR. The interest rate at December 31, 2023 was 10.17%.
The Term Loan B is secured by substantially all the assets of HoldCo and its wholly-owned domestic subsidiaries, including a first-priority security interest in Term Priority Collateral and a second-priority security interest in ABL Priority Collateral, in each case, subject to customary exceptions. The Term Loan B contains customary representations and warranties, affirmative and negative covenants and events of default, including negative covenants that restrict the ability of HoldCo and its restricted subsidiaries to incur additional indebtedness, pay dividends, make investments, loans, and acquisitions, among other restrictions.
The fair value of the Term Loan B was $281.9 million as of December 31, 2023. The estimated fair value of the Notes is based on market rates at the closing trading price as of December 31, 2023 and is classified as Level 2 in the fair value hierarchy.
Convertible Notes
On May 19, 2020, the Company issued $143.8 million in aggregate principal amount of 4.00% Convertible Senior Notes due 2026 (the “Notes”) pursuant to the indenture (the “Indenture”), dated as of May 19, 2020. The total net proceeds from the Notes offering, after deducting debt issuance costs paid or payable by us, was $137.9 million. The Notes accrue interest at a rate of 4.00% per annum, payable semi-annually in arrears on June 1 and December 1 of each year, beginning on December 1, 2020. The Notes will mature on June 1, 2026, unless earlier repurchased, redeemed or converted. Before January 15, 2026, holders will have the right to convert their Notes only upon the occurrence of certain events. From and after January 15, 2026, holders may convert their Notes at any time at their election until the close of business on the scheduled trading day immediately before the maturity date. The Company will settle conversions by paying or delivering, as applicable, cash, shares of its Class A common stock, or a combination of cash and shares of its Class A common stock, at its election. The initial conversion rate is 33.7268 shares of Class A common stock per $1,000 principal amount of Notes. The conversion rate and conversion price will be subject to customary adjustments upon the occurrence of certain events. In addition, if certain corporate events that constitute a “Make-Whole Fundamental Change” (as defined in the Indenture) occur, then the conversion rate will, in certain circumstances, be increased for a specified period of time.
The Notes are redeemable, in whole or in part, at the Company’s option at any time, and from time to time, on or after June 1, 2023 and on or before the 40th scheduled trading day immediately before the maturity date, at a cash redemption price equal to the principal amount of the Notes to be redeemed, plus accrued and unpaid interest, if any, to, but excluding, the redemption date, but only if the last reported sale price per share of the Company’s Class A common stock exceeds 130% of the conversion price on (i) each of at least 20 trading days, whether or not consecutive, during the 30 consecutive trading days ending on, and including, the trading day immediately before the date the Company sends the related redemption notice; and (ii) the trading day immediately before the date the Company sends such notice. In addition, calling any Notes will constitute a Make-Whole Fundamental Change with respect to such Notes, which will result in an increase to the conversion rate if such Notes are converted after they are called for redemption.
If certain corporate events that constitute a “Fundamental Change” (as defined in the Indenture) occur, then noteholders may require the Company to repurchase their Notes at a cash repurchase price equal to the principal amount of the Notes to be repurchased, plus accrued and unpaid interest, if any, to, but excluding, the fundamental change repurchase date. The definition of Fundamental Change includes certain business combination transactions involving the Company and certain de-listing events with respect to the Company’s Class A common stock.
The Notes are the Company’s senior, unsecured obligations and are (i) equal in right of payment with the Company’s existing and future senior, unsecured indebtedness; (ii) senior in right of payment to the Company’s existing and future indebtedness that is expressly subordinated to the Notes; (iii) effectively subordinated to the Company’s existing and future secured indebtedness, to the extent of the value of the collateral securing that indebtedness; and (iv) structurally subordinated to all existing and future indebtedness and other liabilities, including trade payables, and (to the extent the Company is not a holder thereof) preferred equity, if any, of the Company’s subsidiaries. The Notes contain customary events of default.
The fair value of the Notes was $146.1 million as of December 31, 2023. The estimated fair value of the Notes is based on market rates at the closing trading price of the Convertible Notes as of December 31, 2023 and is classified as Level 2 in the fair value hierarchy.
Capped Call Transactions
On May 14, 2020 and May 19, 2020, the Company entered into capped call transactions (the “Capped Call Transactions”) with two counterparties. The Capped Call Transactions cover, subject to anti-dilution adjustments substantially similar to the Notes, the underlying shares of Class A common stock and are intended to reduce, subject to a limit, the potential dilution with respect to the Class A common stock upon conversion of the Notes. The cap price of the Capped Call Transactions is $41.51 per share of Class A common stock, and is subject to certain adjustments under the terms of the Capped Call Transactions.
The Company paid approximately $14.8 million for the Capped Call Transactions, which was recorded as additional paid-in capital, using a portion of the gross proceeds from the sale of the Notes. The capped call is expected to be tax deductible as the Company elected to integrate the capped call into the Notes for tax purposes.
General Other Debt
The Company entered into a mortgage with Huntington Bank to finance a commercial building in Eagle, Idaho, in December 2022. The mortgage bears interest at a fixed rate of 2.0% plus LIBOR and utilizes a 10 year amortization schedule with a balloon payment of the balance due at that time. As of June 30, 2023 the interest rate benchmark changed from LIBOR to SOFR. The rate at December 31, 2023 was 7.47%.
In July 2020, the Company entered an asset purchase agreement that called for PetIQ to pay $20.6 million, $2.6 million at signing and $1.0 million per quarter thereafter with no interest. The Company discounted the payment stream using a market interest rate of 8.3%, resulting in an obligation of $17.5 million at the time it was entered into.
The following represents the Company’s long-term debt as of:
| | | | | | | | | | | |
| | | |
| $'s in 000's | December 31, 2023 | | December 31, 2022 |
| Convertible notes | $ | 143,750 | | | $ | 143,750 | |
| Term loans | 292,500 | | | 295,500 | |
| Revolving credit facility | — | | | — | |
| Other debt | 15,564 | | | 19,690 | |
| Net discount on debt and deferred financing fees | (6,587) | | | (8,531) | |
| $ | 445,227 | | | $ | 450,409 | |
| Less current maturities of long-term debt | (7,407) | | | (7,133) | |
| Total long-term debt | $ | 437,820 | | | $ | 443,276 | |
Future maturities of long-term debt, excluding net discount on debt and deferred financing fees, as of December 31, 2023, are as follows:
| | | | | |
| ($'s in 000's) | |
| 2024 | $ | 7,400 | |
| 2025 | 4,600 | |
| 2026 | 147,314 | |
| 2027 | 3,600 | |
| 2028 | 281,100 | |
| Thereafter | 7,800 | |
The Company incurred $6.4 million in debt issuance costs related to the Term Loan B, $1.0 million related to the ABL, and $0.2 million related to the new mortgage during the year ended December 31, 2021.
Note 6 – Leases
The Company leases certain real estate for commercial, production, and retail purposes, as well as equipment from third parties. Lease expiration dates are between 2024 and 2029.
For both operating and finance leases, the Company recognizes a right-of-use asset, which represents the right to use the underlying asset for the lease term, and a lease liability, which represents the present value of our obligation to make payments arising over the lease term.
We elected the short-term lease exemption for all leases that qualify. This means leases having an initial term of twelve months or less are not recorded on the balance sheet and the related lease expense is recognized on a straight-line basis over the term of the lease.
The Company’s leases may include options to extend or terminate the lease. Renewal options generally range from one to ten years and the options to extend are included in the lease term when it is reasonably certain that we will exercise that option. Some leases have variable payments, however, because they are not based on an index or rate, they are not included in the ROU assets and liabilities. Variable payments for real estate leases primarily relate to common area maintenance, insurance, taxes and utilities. Variable payments for equipment and vehicles primarily relate to usage, repairs, and maintenance. As the implicit rate is not readily determinable for most of the Company’s leases, the Company applies a portfolio approach using an estimated incremental borrowing rate, giving consideration to company specific information and publicly available interest rates for instruments with similar characteristics, to determine the initial present value of lease payments over the lease terms.
The components of lease expense consists of the following:
| | | | | | | | | | | | | | | | | |
| For the Year Ended |
| $'s in 000's | December 31, 2023 | | December 31, 2022 | | December 31, 2021 |
| Finance lease cost | | | | | |
| Amortization of right-of-use assets | $ | 1,398 | | | $ | 1,185 | | | $ | 2,215 | |
| Interest on lease liabilities | 134 | | | 243 | | | 338 | |
| Operating lease cost | 7,845 | | | 6,603 | | | 5,556 | |
Variable lease cost(1) | 4,191 | | | 1,318 | | | 1,283 | |
| Short-term lease cost | 115 | | | 17 | | | 13 | |
| Sublease income | (387) | | | (269) | | | (238) | |
| Total lease cost | $ | 13,296 | | | $ | 9,097 | | | $ | 9,167 | |
(1) Variable lease cost primarily relates to maintenance expenses incurred as a part of service segment optimization, common area maintenance, property taxes and insurance on leased real estate.
Other information related to leases was as follows as of:
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
| Weighted-average remaining lease term (years) | | | |
| Operating leases | 3.74 | | 3.19 |
| Finance leases | 2.32 | | 1.82 |
| Weighted-average discount rate | | | |
| Operating leases | 5.5% | | 4.4% |
| Finance leases | 5.4% | | 4.5% |
Annual future commitments under non-cancelable leases as of December 31, 2023, consist of the following:
| | | | | | | | | | | |
| Lease Obligations |
| $'s in 000's | Operating Leases | | Finance Leases |
| 2024 | $ | 8,468 | | | $ | 1,288 | |
| 2025 | 6,068 | | | 306 | |
| 2026 | 4,091 | | | 152 | |
| 2027 | 2,466 | | | 73 | |
| 2028 | 2,263 | | | 30 | |
| Thereafter | 177 | | | — | |
| Total minimum future obligations | $ | 23,533 | | | $ | 1,849 | |
| Less interest | (2,162) | | | (145) | |
| Present value of net future minimum obligations | 21,371 | | | 1,704 | |
| Less current lease obligations | (7,608) | | | (1,188) | |
| Long-term lease obligations | $ | 13,763 | | | $ | 516 | |
Supplemental cash flow information:
| | | | | | | | | | | | | | | | | |
| For the Year Ended |
| $'s in 000's | December 31, 2023 | | December 31, 2022 | | December 31, 2021 |
| Cash paid for amounts included in the measurement of lease liabilities | | | | | |
| Operating cash flows from finance leases | $ | 134 | | | $ | 243 | | | $ | 338 | |
| Operating cash flows from operating leases | 8,005 | | | 6,730 | | | 3,928 | |
| Financing cash flows from finance leases | 1,494 | | | 1,493 | | | 1,926 | |
| (Noncash) right-of-use assets obtained in exchange for lease obligations | | | | | |
| Operating leases | 8,524 | | | 4,087 | | | 5,212 | |
| Finance leases | 775 | | | 59 | | | 1,191 | |
Note 7 - Income Taxes
The Company is the sole managing member of HoldCo. HoldCo is treated as a partnership for U.S. federal income tax purposes with the remaining members of HoldCo (the “LLC Owners”) owning a non-controlling interest. The LLC Owners have an exchange right which grants them the right to exchange a HoldCo partnership interest and a PetIQ Class B Common Stock share for a PetIQ Class A Common Stock share. Upon such an exchange, the Company is treated as purchasing an additional interest in HoldCo from the continuing LLC Owners in a taxable exchange which generates deferred tax assets as a result of an increase in tax basis for the Company. As of December 31, 2023, the Company had $67.6 million of deferred tax assets associated with these exchanges, which currently have a full valuation allowance against the deferred tax asset. The non-controlling interests totaled approximately 0.8% of the ownership of HoldCo as of December 31, 2023. See Note 11 – Non-controlling interests for more information.
HoldCo’s members, including the Company, are liable for federal, state, and local income taxes based on their share of HoldCo’s taxable income.
The components of pretax net income (loss), determined by tax jurisdiction, are as follows:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| | | | | |
| $'s in 000's | 2023 | | 2022 | | 2021 |
| United States | $ | 2,037 | | | $ | (47,767) | | | $ | (12,816) | |
| Foreign | 329 | | | 361 | | | 302 | |
| Total | $ | 2,366 | | | $ | (47,406) | | | $ | (12,514) | |
The provision for income taxes for 2023, 2022, and 2021 consisted of the following:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| | | | | |
| $'s in 000's | 2023 | | 2022 | | 2021 |
| Current: | | | | | |
| Federal | $ | 149 | | | $ | — | | | $ | — | |
| State | 294 | | | 498 | | | 323 | |
| Foreign | 226 | | | 117 | | | 26 | |
| $ | 669 | | | $ | 615 | | | $ | 349 | |
| Deferred and other: | | | | | |
| Federal | (573) | | | 574 | | | 2,661 | |
| State | (61) | | | 154 | | | 717 | |
| Foreign | (208) | | | (129) | | | 142 | |
| (842) | | | 599 | | | 3,520 | |
| Total income tax (benefit) expense | $ | (173) | | | $ | 1,214 | | | $ | 3,869 | |
Reconciliation between the effective tax rate on income from continuing operations and the statutory tax rate is as follows:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| | | | | |
| 2023 | | 2022 | | 2021 |
| Income tax expense at federal statutory rate | 21.0 | % | | 21.0 | % | | 21.0 | % |
| State and local income taxes net of federal tax benefit | 29.3 | | | 3.6 | | | (0.2) | |
| Non-controlling interest and nontaxable income | (0.6) | | | (0.2) | | | (0.1) | |
| Deferred tax rate changes | 13.9 | | | — | | | 1.0 | |
| Share-based compensation | 57.3 | | | (1.6) | | | 6.8 | |
| Foreign income inclusions | 3.7 | | | — | | | (0.6) | |
| Executive compensation limitations | 5.3 | | | — | | | — | |
| Unrecognized tax benefits | (14.5) | | | — | | | — | |
| Nontaxable indemnification asset change | 2.9 | | | — | | | — | |
| Investments held for sale | (43.8) | | | — | | | — | |
| Goodwill impairment | — | | | (9.2) | | | — | |
| Return-to-provision | (124.7) | | | (6.8) | | | (15.7) | |
| Valuation allowance | 41.1 | | | (9.4) | | | (42.9) | |
| Other | 1.8 | | | — | | | (0.2) | |
| Effective income tax rate | (7.3) | % | | (2.6) | % | | (30.9) | % |
Our tax rate is affected primarily by share-based compensation, return to provision adjustments, change in investments held for sale, the changes in valuation allowance, and state and local taxes during the year ended December 31, 2023. It is also affected by discrete items that may occur in any given year, which are not consistent from year to year.
The Company has recorded deferred tax assets and liabilities based on the differences between the book value of assets and liabilities for financial reporting purposes and those amounts applicable for income tax purposes. Deferred tax assets have been recorded for the basis differences resulting from the purchase of LLC Interests from existing members and newly issued LLC Interests acquired directly from HoldCo. The tax effects of temporary differences that give rise to significant portions of the deferred tax assets and liabilities at December 31, 2023 and 2022 are as follows:
| | | | | | | | | | | |
| $'s in 000's | 2023 | | 2022 |
| Deferred tax assets | | | |
| Investment in partnership | $ | 73,111 | | | $ | 69,656 | |
| Outside basis in foreign subsidiary | 2,648 | | | — | |
| Fixed assets | — | | | 62 | |
| Net operating loss carryforwards and tax credits | 26,385 | | | 33,271 | |
| Disallowed business interest carryforward | 10,472 | | | 8,389 | |
| Other accruals and reversals | 57 | | | 5 | |
| Subtotal | 112,673 | | | 111,383 | |
| Less: valuation allowance | (112,184) | | | (111,218) | |
| Net deferred tax assets | $ | 489 | | | $ | 165 | |
| | | |
| Deferred tax liabilities | | | |
| Fixed assets | $ | — | | | $ | (112) | |
| Intangible assets | (2,690) | | | (1,069) | |
| Other | (472) | | | (9) | |
| Net deferred tax liabilities | $ | (3,162) | | | $ | (1,190) | |
At December 31, 2023, the Company has federal net operating loss (“NOL”) carryforwards of $94.8 million, which do not expire. The NOL carryforwards will have an indefinite carryforward period but can generally only be used to offset 80% of taxable income in any particular year. The Company has a federal business interest expense carryover totaling $49.9 million as of December 31, 2023, which has an indefinite carryforward period but is limited in any particular year based on certain provisions. As of December 31, 2023, the Company has charitable contribution carryforwards of $0.6 million, which if unused will expire between 2024 and 2028. The Company has state NOL carryforwards of $79.2 million as of December 31, 2023 which expire between 2024 and 2041 and others that have an indefinite carryforward period. At December 31, 2023 the Company has state business tax credits related to tax incentive arrangements of $0.6 million which if unused will expire between 2036 and 2038.
The Company has assessed the realizability of the net deferred tax assets as of December 31, 2023 and has considered the relevant positive and negative evidence available to determine whether it is more likely than not that some portion or all of the deferred income tax assets will not be realized. The realization of the gross deferred tax assets is dependent on several factors, including the generation of sufficient taxable income to realize its deferred tax assets. The Company believes it is more likely than not that the benefit from the recorded deferred tax assets in the United States will not be realized. The Company has recorded a valuation allowance for these deferred tax assets of $112.2 million and $111.2 million as of December 31, 2023 and 2022, respectively. Additionally, the Company has recorded a net deferred tax liability related to its indefinite lived intangible assets in the United States. In future periods, if we conclude we have future taxable income sufficient to recognize the deferred tax assets, we may reduce or eliminate the valuation allowance.
The following table summarizes the changes related to our gross unrecognized tax benefits excluding interest and penalties:
| | | | | | | | | | | | | | | | | |
| $'s in 000's | 2023 | | 2022 | | 2021 |
| Balance as of January 1, | $ | — | | | $ | — | | | $ | — | |
| Increases related to prior years' tax positions | (1,079) | | | — | | | — | |
| Increases related to current years' tax positions | — | | | — | | | — | |
| Decreases related to prior years' tax positions | — | | | — | | | — | |
| Decreases related to current years' tax positions | — | | | — | | | — | |
| Lapse of statute of limitations | 338 | | | — | | | — | |
| Balance as of December 31, | $ | (741) | | | $ | — | | | $ | — | |
As of December 31, 2023, 2022, and 2021, we had $0.7 million, $0.0 million, and $0.0 million, respectively, of unrecognized tax benefits recorded in our consolidated balance sheets, excluding interest and penalties. Of the total unrecognized tax benefits recorded, $0.7 million, $0.0 million, and $0.0 million (net of the federal benefit for state taxes), respectively, would impact the effective tax rate if recognized. The Company is fully indemnified with respect to these recorded unrecognized tax benefits.
We recognize interest and penalties related to uncertain tax positions as income tax expense. For the years ended December 31, 2023, 2022, and 2021, we recognized an insignificant amount of interest and penalties related to taxes. We recognize tax liabilities and adjust these liabilities when our judgement changes as a result of the evaluation of new information not previously available or as new uncertainties occur.
The Company operates in certain jurisdictions where we have tax incentive arrangements. These incentives are conditional upon certain business operations and employment thresholds. The effect of these tax incentive arrangements are recognized in the Company's tax provision as the appropriate conditions are met and credits are earned.
We are subject to taxation in the United States and various states and foreign jurisdictions. As of December 31, 2023, tax years from 2020 to present are subject to examination by the tax authorities.
Note 8 – Earnings (Loss) per Share
Basic and Diluted Earnings (Loss) per share
Basic earnings (loss) per share of Class A common stock is computed by dividing net earnings (loss) available to PetIQ, Inc. by the weighted-average number of shares of Class A common stock outstanding during the period. Diluted earnings (loss) per share of Class A common stock is computed by dividing net earnings (loss) available to PetIQ, Inc. by the weighted-average number of shares of Class A common stock outstanding adjusted to give effect to potentially dilutive securities.
The following table sets forth reconciliations of the numerators and denominators used to compute basic and diluted earnings (loss) per share of Class A common stock:
| | | | | | | | | | | | | | | | | |
| Year ended December 31, |
| (in 000's, except for per share amounts) | 2023 | | 2022 | | 2021 |
| Numerator: | | | | | |
| Net income (loss) | $ | 2,539 | | | $ | (48,620) | | | $ | (16,383) | |
| Less: net income (loss) attributable to non-controlling interests | 408 | | | (412) | | | (416) | |
| Net income (loss) attributable to PetIQ, Inc. — basic and diluted | $ | 2,131 | | | $ | (48,208) | | | $ | (15,967) | |
Denominator(1): | | | | | |
| Weighted-average shares of Class A common stock outstanding — basic | 29,135 | | | 29,159 | | | 28,242 | |
Dilutive effect of share-based compensation awards(2) | 395 | | | — | | | — | |
Dilutive effect of conversion of Notes(3) | — | | | — | | | — | |
| Weighted-average shares of Class A common stock outstanding — diluted | 29,530 | | | 29,159 | | | 28,242 | |
| | | | | |
| Earnings (loss) per share of Class A common stock — basic | $ | 0.07 | | | $ | (1.65) | | | $ | (0.57) | |
| Earnings (loss) per share of Class A common stock — diluted | $ | 0.07 | | | $ | (1.65) | | | $ | (0.57) | |
(1) Shares of the Company’s Class B common stock do not share in the earnings of the Company and are therefore not participating securities. As such, separate presentation of basic and diluted earnings per share of Class B common stock under the two-class method has not been presented.
(2) For the year ended December 31, 2023, 1,737 thousand of outstanding share-based compensation awards were excluded from the computation of diluted earnings per share, as the effect of including those shares would be anti-dilutive. Additionally, all outstanding share-based compensation awards were not included in the diluted earnings per share calculation for the years ended December 31, 2022 and 2021, as they have been determined to be anti-dilutive under the treasury stock method.
(3) For the years ended December 31, 2023, 2022, and 2021, 4,848 shares of common stock associated with the assumed conversion of the Notes have been excluded from the computation of diluted earnings per share, as the effect of including those shares would be anti-dilutive under the if-converted method.
Note 9 – Stock Based Compensation
PetIQ, Inc. Omnibus Incentive Plan
The Amended and Restated PetIQ, Inc. Omnibus Incentive Plan, as amended (the “Plan”), provides for the grant of various equity-based incentive awards to directors of the Company, employees, and consultants. The types of equity-based awards that may be granted under the Plan include: stock options, stock appreciation rights ("SARs"), restricted stock, restricted stock units ("RSUs"), and other stock-based awards. On June 22, 2022, the Company's stockholders approved an amendment and restatement of the Plan to, among other things, increase the total number of shares of the Company's Class A common stock reserved and available for issuance thereunder by 1,890,000 shares resulting in a total of 5,804,000 shares of Class A common stock issuable under the Plan. As of December 31, 2023 and 2022, 1,213,177 and 2,143,955 shares were available for issuance under the Plan, respectively. All awards issued under the Plan may only be settled in shares of Class A common stock. Shares issued pursuant to awards under the incentive plans are from our authorized but unissued shares.
PetIQ, Inc. 2018 Inducement and Retention Stock Plan for CVC Employees
The PetIQ, Inc. 2018 Inducement and Retention Stock Plan for CVC Employees (the “Inducement Plan”) provided for the grant of stock options to employees hired in connection with an acquisition in 2018 as employment inducement awards pursuant to NASDAQ Listing Rule 5635(c)(4). The Inducement Plan reserved 800,000 shares of Class A Common Stock of the Company, of which 760,000 were granted. No further grants may be made under the Inducement Plan. All awards issued under the Inducement Plan may only be settled in shares of Class A common stock.
Stock Options
The Company awards stock options to certain employees under the Plan and previously issued stock options under the Inducement Plan, which are subject to time-based vesting conditions, typically 25% on each anniversary of the grant date until fully vested. Upon a termination of service relationship by the Company, all unvested options will be forfeited and the shares of common stock underlying such awards will become available for issuance under the Plan. The maximum contractual term for stock options is 10 years.
The fair value of these equity awards is amortized to equity based compensation expense over the vesting period. Expense recognized totaled $1.4 million, $3.2 million, and $5.2 million for the years ended December 31, 2023, 2022, and 2021, respectively. There were no options granted in 2023. All stock based compensation expense is included in selling, general and administrative expenses based on the role of recipients. The fair value of the stock option awards was determined on the grant dates using the Black-Scholes valuation model based on the following weighted-average assumptions for the period ended:
| | | | | | | | | | | | | | |
| | December 31, 2022 | | December 31, 2021 |
Expected term (years) (1) | | 6.25 | | 6.17 |
Expected volatility (2) | | 37.21 | % | | 33.45 | % |
Risk-free interest rate (3) | | 1.44 | % | | 0.89 | % |
Dividend yield (4) | | 0.00 | % | | 0.00 | % |
(1)The Company utilized the simplified method to determine the expected term of the stock options since we do not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term.
(2)The expected volatility assumption was calculated based on a peer group analysis of stock price volatility with a look back period consistent with the expected option term.
(3)The risk-free interest rate was based on the U.S. Treasury yield curve in effect at the time of grant, which corresponds to the expected term of the stock options.
(4)The Company has not paid and does not anticipate paying a cash dividend on our common stock.
The weighted average grant date fair value of stock options granted during the period ended 2022 and 2021 was $9.14 and $12.39, per option respectively. There were no options granted during the year ended December 31, 2023. The following table summarizes the activity of the Company’s unvested stock options for the period ended December 31, 2023:
| | | | | | | | | | | | | | | | | | | | | | | |
| Stock
Options
(in 000's) | | Weighted
Average
Exercise
Price | | Aggregate
Intrinsic
Value
(in 000's) | | Weighted
Average
Remaining
Contractual
Life
(years) |
| Outstanding at January 1, 2022 | 1,768 | | | $ | 26.51 | | | $ | 2,897 | | | 7.3 |
| Granted | 83 | | | 14.16 | | | | | |
| Exercised | (2) | | | 19.49 | | | $ | 10 | | | |
| Forfeited and canceled | (196) | | | 30.06 | | | | | |
| Outstanding at December 31, 2022 | 1,653 | | | $ | 25.48 | | | $ | 53 | | | 6.2 |
| Granted | — | | | — | | | | | |
| Exercised | (3) | | | 19.49 | | | $ | 5 | | | |
| Forfeited and canceled | (196) | | | 24.52 | | | | | |
| Outstanding at December 31, 2023 | 1,454 | | | $ | 25.62 | | | $ | 986 | | | 4.9 |
| Options exercisable at December 31, 2023 | 1,246 | | | | | | | |
The aggregate intrinsic values disclosed in the above table were calculated as the excess, if any, between the Company’s closing share price per share on the respective period end date and the strike price of the underlying awards.
At December 31, 2023, total unrecognized compensation cost related to unvested stock options was $1.1 million and is expected to be recognized over a weighted-average period of approximately 1.0 years.
Restricted Stock Units
The Company awards RSUs to certain employees under the Plan, which are subject to time-based vesting conditions. Upon a termination of service relationship by the Company, all unvested RSUs will generally be forfeited and the shares of common stock underlying such awards will become available for issuance under the Plan. The fair value of RSUs are measured based on the closing fair market value of the Company’s common stock on the date of grant. At December 31, 2023, total unrecognized compensation cost related to unvested RSUs was $19.2 million and is expected to vest over a weighted average of 2.8 years.
The fair value of these equity awards is amortized to equity based compensation expense over the vesting period, which totaled $8.1 million, $8.1 million, and $4.3 million for the years ended December 31, 2023, 2022, and 2021, respectively. All stock based compensation expense is included in selling, general and administrative expenses based on the role of recipients.
The following table summarizes the activity of the Company’s RSUs for the period ended December 31, 2023:
| | | | | | | | | | | |
| Number of
Shares
(in 000's) | | Weighted
Average
Grant Date
Fair Value |
| Outstanding at January 1, 2022 | 459 | | | $ | 31.08 | |
| Granted | 802 | | | 20.30 | |
| Settled | (231) | | | 27.81 | |
| Forfeited | (177) | | | 25.53 | |
| Outstanding at December 31, 2022 | 853 | | | $ | 23.06 | |
| Granted | 1,313 | | | 12.02 | |
| Settled | (234) | | | 22.72 | |
| Forfeited and canceled | (244) | | | 16.18 | |
| Nonvested RSUs at December 31, 2023 | 1,688 | | | $ | 15.26 | |
The total income tax benefit recognized in the income statement for share-based compensation arrangements was zero for the years ended December 31, 2023, 2022, and 2021.
Note 10 - Stockholders’ Equity
Certificate of Incorporation
The Company’s amended and restated certificate of incorporation, among other things, provides for the (i) authorization of 125,000,000 shares of Class A common stock with a par value of $0.001 per share; (ii) authorization of 8,401,521 shares of Class B common stock with a par value of $0.001 per share; and (iii) authorization of 12,500,000 shares of blank check preferred stock.
Each share of the Company’s Class A common stock and Class B common stock entitles its holders to one vote per share on all matters presented to the Company’s stockholders generally.
Holders of the Company’s Class B common stock are not entitled to receive dividends and will not be entitled to receive any distributions upon the liquidation, dissolution or winding up of the Company. Shares of Class B common stock may only be issued to the extent necessary to maintain the one-to-one ratio between the number of LLC interests of HoldCo held by continuing LLC Owners. Shares of Class B common stock are transferable only together with an equal number of LLC Interests. Shares of Class B common stock will be canceled on a one-for-one basis upon the redemption or exchange any of the outstanding LLC Interests held by the continuing LLC Owners.
The Company must, at all times, maintain a one-to-one ratio between the number of outstanding shares of Class A common stock and the number of LLC Interests owned by PetIQ, Inc. (subject to certain exceptions for treasury shares and shares underlying certain convertible or exchangeable securities).
Stock repurchase program
On September 6, 2022, the Company's Board of Directors authorized a stock repurchase program for up to $30.0 million of the Company’s outstanding shares of Class A common stock. Repurchases of Class A common stock may be made at management’s discretion from time to time in one or more transactions on the open market or in privately negotiated purchase and/or through other legally permissible means, depending on market conditions and in accordance with applicable rules and regulations promulgated under Securities Exchange Act. During the year ended December 31, 2022, the Company repurchased 373,408 shares at a weighted average price of $10.33 per share. No shares were repurchased during the year ended December 31, 2023.
Note 11 - Non-Controlling Interests
The Company reports a non-controlling interest representing the LLC interests of HoldCo held by continuing LLC Owners. Changes in PetIQ’s ownership interest in HoldCo while PetIQ retains its controlling interest in HoldCo will be accounted for as equity transactions. As such, future redemptions or direct exchanges of LLC interests of HoldCo by the continuing LLC Owners will result in a change in ownership and reduce or increase the amount recorded as non-controlling interest and increase or decrease additional paid-in capital when HoldCo has positive or negative net assets, respectively. The Company is also required to make tax distributions based on the LLC Agreement to continuing LLC Owners on a regular basis, these distributions will reduce the non-controlling interest.
As of December 31, 2023, there were 29.4 million LLC Interests outstanding, of which PetIQ owned 29.2 million, representing a 99.2% ownership interest in HoldCo. Exchange and other equity activity during the years ended December 31, 2023 and 2022 resulted in weighted average ownership of HoldCo by PetIQ of 99.2% and 99.1%, respectively.
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| LLC Interests held | | % of Total |
| $'s in 000's | LLC
Owners | | PetIQ, Inc. | | Total | | LLC
Owners | PetIQ, Inc. |
| As of January 1, 2022 | 272 | | | 29,139 | | | 29,411 | | | 0.9 | % | 99.1 | % |
| Stock based compensation transactions | — | | | 188 | | | 188 | | | | |
| Exchange transactions | (20) | | | 20 | | | — | | | | |
| Unit redemption | — | | | (373) | | | (373) | | | | |
| As of December 31, 2022 | 252 | | | 28,974 | | | 29,226 | | | 0.9 | % | 99.1 | % |
| Stock based compensation transactions | — | | | 201 | | | 201 | | | | |
| Exchange transactions | (21) | | | 21 | | | — | | | | |
| As of December 31, 2023 | 231 | | | 29,196 | | | 29,427 | | | 0.8 | % | 99.2 | % |
Note 12 - Customer Concentration
The Company has significant exposure to customer concentration. During each of the years ended December 31, 2023, 2022, and 2021, two, two, and one Product segment customers, respectively, accounted for more than 10% of sales individually and in aggregate, which accounted for 42%, 35%, and 26% of net sales, respectively.
At December 31, 2023, one Products segment customer individually accounted for more than 10% of outstanding trade receivables, accounting for 54% of outstanding trade receivables, net. At December 31, 2022 one Products segment customers individually accounting for more than 10% of outstanding trade receivables, accounting for 46% of outstanding trade receivables, net. All of our customer concentration exists in our Products segment.
Note 13 - Commitments and Contingencies
Litigation Contingencies
During the years ended December 31, 2023, 2022, and 2021, the Company recorded expense of $0.0 million, $3.5 million, and $3.5 million respectively, for contract termination costs and other litigation related matters. The expense is included within selling, general and administrative expenses for the years ended December 31, 2023, 2022, and 2021.
During the year ended December 31, 2021, the Company entered into mediation with a third party who had filed a class action lawsuit against the Company. As a result of that mediation, the Company accrued the expected settlement of $1.4 million. Final settlement of the mediation and payment of the accrued amount occurred in 2022.
Additionally, during the year ended December 31, 2022, the Company settled a lawsuit brought by a former supplier to the Company related to the redemption of ownership interests. The Company had accrued an obligation of $2.0 million as of December 31, 2021. The Company recorded an additional $3.5 million of expense in the consolidated statement of operations for the year ended December 31, 2022 and paid $5.5 million in full satisfaction of the lawsuit.
The Company records a liability when a particular contingency is probable and estimable and provides disclosure for contingencies that are at least reasonably possible of resulting in a loss including an estimate which we currently cannot make. The Company has not accrued for any contingency other than those noted above, at December 31, 2023 as the Company does not consider any other contingency to be probable or estimable. The Company expenses legal costs as incurred within selling, general, and administrative expenses on the consolidated statements of operations.
Commitments
We have commitments for leases and long-term debt that are discussed further in Note 5, Debt, and Note 6, Leases. In addition, we have purchase obligations for goods and services, capital expenditures, and raw materials entered into in the normal course of business.
Note 14 - Segments
The Company has two operating segments: Products and Services. The Products segment consists of the Company’s manufacturing and distribution business. The Services segment consists of the Company’s veterinary services and related product sales. The segments are based on the discrete financial information reviewed by the Chief Operating Decision Maker (“CODM”) to make resource allocation decisions and to evaluate performance. We measure and evaluate our reportable segments based on their respective Segment Adjusted EBITDA performance. Beginning in the fourth quarter of 2022, we allocate to our segments capital expenditures and certain costs and expenses, such as accounting, legal, human resources, information technology and corporate headquarters expenses, on a pro rata basis based on net sales to better align with the discrete financial information reviewed by our CODM. Such expenses previously were not allocated to segments. The Company has recast prior periods to give effect to this change. This change in presentation had no impact on the Consolidated Statements of Operations.
Financial information relating to the Company’s operating segments for the years ended:
| | | | | | | | | | | | | | |
| $'s in 000's | | | | |
| December 31, 2023 | | Products | | Services |
| Net sales | | $ | 968,151 | | | $ | 133,812 | |
| Segment Adjusted EBITDA | | 110,565 | | | 2,623 | |
| Depreciation expense | | 7,614 | | | 17,159 | |
| Capital expenditures | | 7,824 | | | 1,392 | |
| | | | | | | | | | | | | | |
| $'s in 000's | | | | |
| December 31, 2022 | | Products | | Services |
| Net sales | | $ | 800,305 | | | $ | 121,208 | |
| Segment Adjusted EBITDA | | 90,333 | | | 3,781 | |
| Depreciation expense | | 7,443 | | | 7,077 | |
| Capital expenditures | | 8,432 | | | 3,541 | |
| | | | | | | | | | | | | | |
| $'s in 000's | | | | |
| December 31, 2021 | | Products | | Services |
| Net sales | | $ | 825,395 | | | $ | 107,133 | |
| Segment Adjusted EBITDA | | 88,982 | | | 3,910 | |
| Depreciation expense | | 7,397 | | | 6,969 | |
| Capital expenditures | | 18,568 | | | 12,702 | |
The value of assets by segment were as follows as of:
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
| Products | $ | 798,578 | | | $ | 735,096 | |
| Services | 69,647 | | | 83,800 | |
| Total assets | $ | 868,226 | | | $ | 818,896 | |
| | | |
The following table reconciles Segment EBITDA to Net income (loss) for the periods presented.
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| $'s in 000's | 2023 | | 2022 | | 2021 |
| Segment Adjusted EBITDA: | | | | | |
| Product | $ | 110,565 | | | $ | 90,333 | | | $ | 88,982 | |
| Services | 2,623 | | | 3,781 | | | 3,910 | |
| Total | 113,188 | | | 94,114 | | | 92,892 | |
| Adjustments: | | | | | |
Depreciation(1) | (24,773) | | | (14,520) | | | (14,366) | |
| Amortization | (19,797) | | | (18,079) | | | (22,336) | |
| Interest expense, net | (34,547) | | | (27,374) | | | (24,696) | |
Acquisition costs(2) | (1,164) | | | (1,464) | | | (92) | |
Loss on debt extinguishment and related costs(3) | — | | | — | | | (6,438) | |
| Stock based compensation expense | (9,468) | | | (11,363) | | | (9,428) | |
Non same-store adjustment(4) | (8,482) | | | (16,423) | | | (23,159) | |
Integration costs(5) | (2,316) | | | (1,171) | | | 142 | |
| Litigation expenses | (31) | | | (3,862) | | | (4,105) | |
| CFO Transition | — | | | — | | | (928) | |
Restructuring(6) | (2,564) | | | — | | | — | |
Impairment and other asset charges(7) | (7,680) | | | (47,264) | | | — | |
| Pretax net income (loss) | $ | 2,366 | | | $ | (47,406) | | | $ | (12,514) | |
| Income tax benefit (expense) | 173 | | | (1,214) | | | (3,869) | |
| Net income (loss) | $ | 2,539 | | | $ | (48,620) | | | $ | (16,383) | |
(1) Depreciation includes $11.0 million of accelerated depreciation recognized during the year ended December 31, 2023, associated with Services segment optimization.
(2) Acquisition costs include legal, accounting, banking, consulting, diligence, and other costs related to completed and contemplated acquisitions.
(3) Loss on debt extinguishment and related costs are related to our entering into two new credit facilities, including the write off of deferred financing costs and related costs.
(4) Non same-store adjustment includes revenue and costs, and associated gross profit, related to our Services segment wellness centers and host partners with less than six full quarters of operating results, and also include pre-opening expenses.
(5) Integration and business transformation costs, including personnel costs such as severance and retention bonuses, consulting costs, contract termination costs and IT and ERP implementation costs.
(6) Restructuring consists of variable lease expenses, inventory valuation adjustments, lease termination costs, severance, and other miscellaneous costs.
(7) Impairment and other asset charges includes asset charges associated with the Company committing to a plan to sell its foreign subsidiary, Mark & Chappell during the year ended December 31, 2023. For the year ended December 31, 2022, impairment and other asset charges includes write-down of the full goodwill balance of the Services segment.
Supplemental geographic disclosures are below.
| | | | | | | | | | | | | | | | | |
| Year ended December 31, 2023 |
| $'s in 000's | U.S. | | Foreign | | Total |
| Product sales | $ | 960,346 | | | $ | 7,805 | | | $ | 968,151 | |
| Service revenue | 133,812 | | | — | | | 133,812 | |
| Total net sales | $ | 1,094,158 | | | $ | 7,805 | | | $ | 1,101,963 | |
| | | | | | | | | | | | | | | | | |
| Year ended December 31, 2022 |
| $'s in 000's | U.S. | | Foreign | | Total |
| Product sales | $ | 793,427 | | | $ | 6,878 | | | $ | 800,305 | |
| Service revenue | 121,208 | | | — | | | 121,208 | |
| Total net sales | $ | 914,635 | | | $ | 6,878 | | | $ | 921,513 | |
| | | | | | | | | | | | | | | | | |
| Year ended December 31, 2021 |
| $'s in 000's | U.S. | | Foreign | | Total |
| Product sales | $ | 818,593 | | | $ | 6,802 | | | $ | 825,395 | |
| Service revenue | 107,133 | | | — | | | 107,133 | |
| Total net sales | $ | 925,726 | | | $ | 6,802 | | | $ | 932,528 | |
The net book value of property plant and equipment, by geographic location was as follows as of:
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
| United States | $ | 57,097 | | | $ | 69,376 | |
| Europe | — | | | 4,019 | |
| Total | $ | 57,097 | | | $ | 73,395 | |
Note 15 - Restructuring
During the year ending December 31, 2023, the Company implemented a Services segment optimization (the "optimization") to improve the functioning of the Services segment and profitability. The optimization included assessing the operational and financial performance of the Company's wellness centers since re-opening after the pandemic as well as the assessment of the veterinary labor market in each geographic market. The Company also evaluated its ability to potentially convert these locations to a more hygiene-focused offering and determined they would be unable to convert these locations in the future based on the aforementioned assessment and the available square footage within the respective wellness centers.
As a result of the optimization, the Company identified 149 underperforming wellness centers for closure. The Company closed 149 wellness centers during the year ended December 31, 2023 and finished the year with 133 wellness centers.
Additionally, included in restructuring and related costs is a lease termination gain of approximately $0.9 million. The gain resulted from settlement of $2.7 million of operating lease liabilities for a discounted amount of$1.8 million.Closure of the wellness centers required the Company to terminate leases early, the Company agreed upon termination amounts with retail partners in 2023 and accrued for the lease termination costs on the consolidated balance sheet at December 31, 2023.
The components of restructuring expense consists of the following:
| | | | | |
| For the year ended |
| $'s in 000's | December 31, 2023 |
| Included in Cost of services | |
| Inventory reserve | $ | 888 | |
| Severance | 966 | |
| Total in Cost of services | $ | 1,854 | |
| |
| Included in Restructuring | |
| Accelerated depreciation - property, plant and equipment | $ | 8,165 | |
| Accelerated amortization - operating lease right of use assets | 2,876 | |
| Lease termination gain | (897) | |
| Variable lease expenses | 1,607 | |
| Total in Restructuring | 11,751 | |
| Total Restructuring Expenses | $ | 13,605 | |
A roll forward of our liability related to the optimization, which is included in accrued liabilities on our consolidated balance sheets, is as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| $'s in 000's | Expenses (Gains) | | Original Lease Liability | | Cash Payments | | Non-Cash Amounts | | Liability at December 31, 2023 |
| Accelerated depreciation - property, plant and equipment | $ | 8,165 | | | $ | — | | | $ | — | | | $ | (8,165) | | | $ | — | |
| Accelerated amortization - operating lease right of use assets | 2,876 | | | — | | | — | | | (2,876) | | | — | |
| Inventory reserve | 888 | | | — | | | — | | | (888) | | | — | |
| Severance | 966 | | | — | | | (966) | | | — | | | — | |
| Lease termination gain | (897) | | | 2,722 | | | — | | | — | | | 1,825 | |
| Variable lease expenses | 1,607 | | | — | | | (597) | | | — | | | 1,010 | |
| Total Restructuring | $ | 13,605 | | | $ | 2,722 | | | $ | (1,563) | | | $ | (11,929) | | | $ | 2,835 | |
Note 16 - Related Parties
Chris Christensen, the brother of CEO, McCord Christensen, acts as the Company’s agent at Moreton Insurance, which acts as a broker for a number of the Company’s insurance policies. The Company’s annual premium expense, which is paid at a variety of times throughout the year, and is generally paid directly to the relevant insurance company, amounted to $7.1 million, $6.6 million, and $6.9 million in 2023, 2022, and 2021 respectively. Mr. Chris Christensen earns various forms of compensation based on the specifics of each policy.
In August 2021, the Company sold its prior corporate office in Eagle, Idaho for $4.8 million. The Company utilized Colliers International (“Colliers”) as a broker with Mike Christensen, the brother of CEO, McCord Christensen, as agent. The Company paid approximately $0.1 million in commissions to Colliers as a result of the sale. In December 2021, the Company purchased a parcel of land for $2.5 million. Total commission paid to Colliers was approximately $0.1 million as a result of this purchase.
Mike Glasman, the brother of CFO, Zvi Glasman, acted as a broker in connection with the Company's entry into a Master Services Agreement with Syndeo, LLC d/b/a Broadvoice ("Broadvoice") in February 2023 for the provision of certain information technology related services. The amount to be paid to Broadvoice over the 39-month agreement is estimated at $0.4 million. $0.05 million was paid to Broadvoice for the year ending December 31, 2023. Mr. Michael Glasman earns various fees based on the services provided by Broadvoice.
Katie Turner, the spouse of CEO, McCord Christensen, is the owner of Acadia Investor Relations LLC, (“Acadia”) which acts as the Company’s investor relations consultant. Acadia has been paid $0.2 million for the year ending December 31, 2023, 2022, and 2021 respectively.
Note 17 – Employee Benefit Plans
The Company sponsors 401(k) defined contribution plans at certain subsidiaries. Participants may elect to defer up to 100% of compensation. The Company makes matching contributions of 100% of the employee deferrals up to 4% of compensation. The Company may also make discretionary profit sharing contributions each year, which are allocated to each eligible participant based on compensation. The Company made matching contributions of $1.8 million, $1.7 million, and $1.1 million, respectively, for the years ended December 31, 2023, 2022 and 2021.
Item 9 – Changes in and Disagreements With Accountants on Accounting and Financial Disclosure
None.
Item 9A – Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, has evaluated the effectiveness of our disclosure controls and procedures (as such term is defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”)) as of December 31, 2023. Based on this evaluation, our Chief Executive Officer and our Chief Financial Officer have concluded that, as of December 31, 2023, our disclosure
controls and procedures (a) were effective to ensure that information that we are required to disclose in reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in Securities and Exchange Commission (“SEC”) rules and forms and (b) include, without limitation, controls and procedures designed to ensure that information required to be disclosed by us in reports filed or submitted under the Exchange Act is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decisions regarding required disclosure.
Management’s Annual Report on Internal Control Over Financial Reporting
We are responsible for establishing and maintaining internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act). Internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of the effectiveness of the internal control over financial reporting to future periods are subject to the risk that the controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Our management, under the supervision and with the participation of our Chief Executive Officer and Chief Financial Officer, assessed the effectiveness of our internal control over financial reporting as of December 31, 2023. In making this assessment, our management used the Internal Control – Integrated Framework (2013) as issued by the Committee of Sponsoring Organizations (COSO) of the Treadway Commission. Based on this assessment, management concluded that our internal control over financial reporting was effective as of December 31, 2023.
The Company’s registered independent accounting firm, KPMG LLP, has audited the effectiveness of our internal controls over financial reporting as of December 31, 2023, as stated in their report which appears on the next page.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting that occurred during our fiscal quarter ended, December 31, 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
 KPMG LLP
KPMG LLPSuite 600
205 North 10th Street
Boise, ID 83702-5798
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors
PetIQ, Inc.:
Opinion on Internal Control Over Financial Reporting
We have audited PetIQ, Inc. and subsidiaries' (the Company) internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission. In our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of the Company as of December 31, 2023 and 2022, the related consolidated statements of operations, comprehensive income (loss), stockholders’ equity, and cash flows for each of the years in the three-year period ended December 31, 2023, and the related notes (collectively, the consolidated financial statements), and our report dated February 29, 2024 expressed an unqualified opinion on those consolidated financial statements.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Management's Annual Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audit also included performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Boise, Idaho
February 29, 2024
Item 9B - Other Information
Trading Plans
During the year ended December 31, 2023, no director or officer of the Company adopted or terminated a "Rule 10b5-1 trading arrangement" or "non-Rule 10b5-1 trading arrangement," as such term is defined in Item 408(a) of Regulation S-K.
Item 9C - Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
Not Applicable.
PART III
Item 10 – Directors, Executive Officers and Corporate Governance
We intend to file with the SEC, not later than 120 days after the close of our fiscal year ended December 31, 2023, a definitive proxy statement containing the information required by this Item, under the captions “Proposal
One—Election of Directors,” ‘Directors and Management,” and “Corporate Governance," which are incorporated by reference herein, or an amendment to this Annual Report filed under cover of Form 10-K/A containing the information required by this Item.
Our Code of Ethics for Senior Financial Officers applies to our principal executive officer, principal financial officer and principal accounting officer or controller or persons performing similar functions (the “Code of Ethics”). The Code of Ethics is available on our website at http://ir.petiq.com. If we make any substantive amendments to our Code of Ethics or grant any of principal executive officer, principal financial officer and principal accounting officer or controller or person performing similar functions any waiver, including any implicit waiver, from a provision of our Code of Ethics, we will disclose the nature of the amendment or waiver on our website or in a Current Report on Form 8-K.
Item 11 – Executive Compensation
We intend to file with the SEC, not later than 120 days after the close of our fiscal year ended December 31, 2023, a definitive proxy statement containing the information required by this Item, under the captions “Compensation Discussion and Analysis,” “Summary Compensation Table,” “Grants of Plan-Based Awards,” “Outstanding Equity Awards at Fiscal Year End,” “Option Exercises and Stock Vested,” “Pension Benefits,” “Nonqualified Deferred Compensation,” “Potential Payments Upon Termination or Change in Control,” “CEO Pay Ratio,” and “Non-Employee Director Compensation,” which are incorporated by reference herein, or an amendment to this Annual Report filed under cover of Form 10-K/A containing the information required by this Item .
Item 12 – Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
We intend to file with the SEC, not later than 120 days after the close of our fiscal year ended December 31, 2023, a definitive proxy statement containing the information required by this Item, under the captions “Security Ownership of Certain Beneficial Owners and Management,” and “Equity Compensation Plans at December 31, 2023” which are incorporated by reference herein, or an amendment to Annual Report filed under cover of Form 10-K/A containing the information required by this Item.
Item 13 – Certain Relationships and Related Transactions
We intend to file with the SEC, not later than 120 days after the close of our fiscal year ended December 31, 2023, a definitive proxy statement containing the information required by this Item, under the captions “Certain Relationships and Related Party Transactions,” and “Corporate Governance,” which are incorporated by reference herein, or an amendment to this Annual Report filed under cover of Form 10-K/A containing the information required by this Item.
Item 14 – Principal Accountant Fees and Services
Our independent registered public accounting firm is KPMG LLP, Boise, ID, Auditor Firm ID: 185.
We intend to file with the SEC, not later than 120 days after the close of our fiscal year ended December 31, 2023, a definitive proxy statement containing the information required by this Item, under the caption “Proposal Two—Ratification
of the Appointment of the Company’s Independent Auditors,” which is incorporated by reference herein, or an amendment to this Annual Report filed under cover of Form 10-K/A containing the information required by this Item.
PART IV
Item 15. Exhibits, Financial Statement Schedules
See "Index to Consolidated Financial Statements" in Part II, Item 8 of this Annual Report on Form 10-K. Financial statement schedules have been omitted because they are not required or are not applicable or because the information required in those schedules either is not material or is included in the consolidated financial statements or the accompanying notes.
| | | | | | | | | | | | | | | | | |
| Exhibit No. | Exhibit Description | Form | File No. | Exhibit | Filing Date |
| 3.1 | | 8-K | 000-38163 | 3.1 | 6/23/2022 |
| 3.3 | | 8-K | 001-38163 | 3.1 | 10/31/2022 |
| 4.1 | | S-1/A | 333-218955 | 4.1 | 7/17/2017 |
| 4.2 | | S-3 | 333-227186 | 4.1 | 9/4/2018 |
| 4.3 | | S-3 | 333-227186 | 4.2 | 9/4/2018 |
| 4.4 | | 8-K | 001-38163 | 4.1 | 5/20/2020 |
| 4.5 | | 10-K | 001-38163 | 4.4 | 3/11/2020 |
| 10.1 | | S-1/A | 333-218955 | 10.4 | 7/6/2017 |
| 10.2 | | 10-K | 001-38163 | 10.1 | 3/12/2019 |
| 10.3 | | 8-K | 001-38163 | 10.1 | 4/19/2021 |
| 10.4 | | 8-K | 001-38163 | 10.2 | 4/19/2021 |
| 10.5 | | 8-K | 001-38163 | 10.10 | 5/31/2023 |
| 10.6* | | S-1/A | 333-218955 | 10.13 | 7/20/2017 |
| 10.7* | | DEF 14 | 001-38163 | Appendix B | 4/29/2022 |
| 10.8* | | 10-Q | 001-38163 | 10.2 | 11/14/2018 |
| 10.9* | | 10-Q | 001-38163 | 10.3 | 11/14/2018 |
| 10.10* | | 10-Q | 001-38163 | 10.4 | 11/14/2018 |
| 10.11* | | S-8 | 333-223635 | 4.3 | 3/13/2018 |
| 10.12* | | 10-Q | 001-38163 | 10.1 | 5/9/2019 |
| 10.13* | | 8-K | 001-38163 | 10.1 | 1/5/2022 |
| 10.14* | | 8-K | 001-38163 | 10.5 | 7/9/2019 |
| 10.15* | | 10-Q | 001-38163 | 10.3 | 5/9/2019 |
| 10.16 | | 8-K | 001-38163 | 10.1 | 5/20/2020 |
| 10.17 | | 8-K | 001-38163 | 10.2 | 5/20/2020 |
| | | | | | | | | | | | | | | | | |
| 10.18 | | 10-K | 001-38163 | 10.17 | 3/1/2023 |
| 10.19 | | 10-K | 001-38163 | 10.18 | 3/1/2023 |
| 10.20 | | 10-K | 001-38163 | 10.19 | 3/1/2023 |
10.21** | | | | | |
| 21.1** | | | | | |
| 23.1** | | | | | |
| 31.1** | | | | | |
| 31.2** | | | | | |
| 32.1*** | | | | | |
| 32.2*** | | | | | |
| 97.1** | | | | | |
| 101.INS | XBRL Instance Document | | | | |
| 101.SCH | XBRL Schema Documents | | | | |
| 101.CAL | XBRL Calculation Linkbase Document | | | | |
| 101.DEF | XBRL Definition Linkbase Document | | | | |
| 101.LAB | XBRL Labels Linkbase Document | | | | |
| 101.PRE | XBRL Presentation Linkbase Document | | | | |
| 101.DEF | XBRL Definition Linkbase Document | | | | |
* Indicates management contract or compensatory plan or arrangement.
** Filed herewith
*** Furnished herewith. This exhibit should not be deemed to be "filed" for purposes of Section 18 of the Exchange Act.
Item 16. Form 10-K Summary
None.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| | | | | | | | |
| | PETIQ, INC. |
| | |
| February 29, 2024 | /s/ Zvi Glasman |
| | Zvi Glasman |
| | Chief Financial Officer |
POWER OF ATTORNEY
KNOWN BY ALL PERSONS BY THESE PRESENTS, that the individuals whose signatures appear below hereby constitute and appoint McCord Christensen and Zvi Glasman, and each of them severally, as his or her true and lawful attorneys-in-fact and agents with full power of substitution and resubstitution for him or her and in his or her name, place and stead in any and all capacities to sign any and all amendments to this Annual Report and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, full power and authority to do or perform each and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or any of them, or of his substitute or substitutes, may lawfully do to cause to be done by virtue hereof. Pursuant to the requirements of the Securities Exchange Act of 1934, this Annual Report has been signed below by the following persons on behalf of the registrant and in the capacities indicated as of February 29, 2024.
| | | | | | | | | | | |
| SIGNATURE | | TITLE | Date |
| | | |
| /s/ McCord Christensen | | Chief Executive Officer, President
(principal executive officer)
Officer and Chairman of the Board | February 29, 2024 |
| McCord Christensen | | |
| | | |
| /s/ Zvi Glasman | | Chief Financial Officer
(principal financial and accounting officer)
Officer | February 29, 2024 |
| Zvi Glasman | | |
| | | |
| /s/ Mark First | | Director | February 29, 2024 |
| Mark First | | |
| | | |
| /s/ Allan Hall | | Director | February 29, 2024 |
| Allan Hall | | |
| | | |
| /s/ Scott Huff | | Director | February 29, 2024 |
| Scott Huff | | |
| | | |
| /s/ Sheryl O'Loughlin | | Director | February 29, 2024 |
| Sheryl O'Loughlin | | |
| | | |
| /s/ Kim Lefko | | Director | February 29, 2024 |
| Kim Lefko | | |
| | | |
| /s/ Kenneth Walker | | Director | February 29, 2024 |
| Kenneth Walker | | |
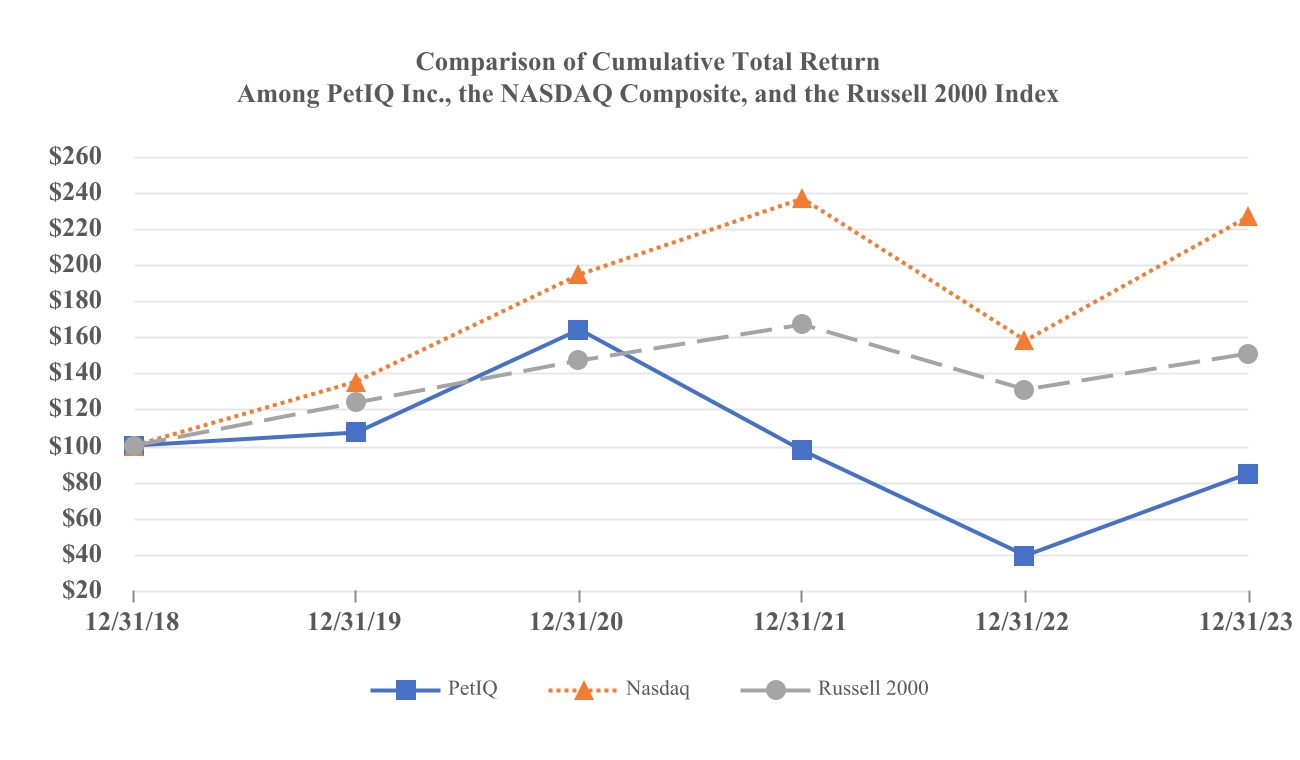



 KPMG LLP
KPMG LLP