
Reimagining The Potential Of Human Enzyme Therapeutics Corporate Overview – August 2022 Nasdaq: AGLE Exhibit 99.2

Forward Looking Statements ©2022 Aeglea BioTherapeutics. External 2 This presentation and the accompanying oral presentation contain “forward-looking” statements that are based on our management’s beliefs and assumptions and on information currently available to management. Forward-looking statements include all statements other than statements of historical fact contained in this presentation, including information concerning our current and future financial performance, business plans and objectives, current and future clinical and preclinical development activities, timing and success of our ongoing and planned clinical trials and related data, the timing of announcements, updates, results of our clinical trials and related data, our ability to obtain and maintain regulatory approval, the potential therapeutic benefits, safety profile and economic value of pegzilarginase and our other product candidates, potential growth opportunities, financing plans, use and adequacy of financing plans, the length of time that we believe our existing cash resources will fund operations, competitive position, industry environment and potential market opportunities.
Forward-looking statements are subject to known and unknown risks, uncertainties, assumptions and other factors including, but not limited to, those related to the success, cost and timing of our product candidate development activities and ongoing and planned clinical trials; our plans to develop and commercialize targeted therapeutics, including our lead product candidate pegzilarginase, AGLE-177, and our product candidate for the treatment of cystinuria; the design, progress of patient enrollment and dosing in our clinical trials, including our phase 1/2 trial of AGLE-177 in Homocystinuria; the ability of our product candidates to achieve applicable endpoints in clinical trials; the safety profile of our product candidates in clinical trials; the potential for data from our current and future clinical trials to support a marketing application, as well as the timing of these events, including data for our phase 1/2 trial of AGLE-177 in Homocystinuria; the potential for preclinical studies to be predictive of current or future clinical trials; our ability to obtain funding for our operations, development and commercialization of our product candidates; the impact of the COVID-19 pandemic on our operations and clinical development activities, including on the timing of enrollment of our clinical trials; the timing of and our ability to obtain and maintain regulatory approvals; our ability to obtain regulatory approval for, and commercialize, pegzilarginase, and recognize milestone and royalty payments from our licensing and supply agreement with Immedica; the potential for expedited review of pegzilarginase as a result of its Breakthrough Therapy designation; the potential addressable markets of our product candidates; the rate and degree of market acceptance and clinical utility of our product candidates; the size and growth potential of the markets for our product candidates, and our ability to serve those markets; our commercialization, marketing and manufacturing capabilities and strategy; future agreements with third parties in connection with the potential commercialization of our product candidates; our expectations regarding our ability to obtain and maintain intellectual property protection; our dependence on third party manufacturers; our ability to develop our own commercial manufacturing facility; the success of competing therapies that are or may become available; our ability to attract and retain key scientific or management personnel; our ability to identify additional product candidates with significant commercial potential consistent with our commercial objectives; and our estimates regarding expenses, future revenue, capital requirements and needs for additional financing.
Moreover, we operate in a very competitive and rapidly changing environment, and new risks may emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed herein may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. Further information on these and other factors that could affect these forward-looking statements is contained in our most recent Annual Report on Form 10-K filed with the U.S. Securities and Exchange Commission (SEC) and other reports filed by the SEC. You should not rely upon forward-looking statements as predictions of future events. Although our management believes that the expectations reflected in our forward-looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances described in the forward-looking statements will be achieved or occur. We undertake no obligation to publicly update any forward-looking statements, whether written or oral, that may be made from time to time, whether as a result of new information, future developments or otherwise.

Addressing Metabolic Imbalances to Improve the Lives of Patients ©2022 Aeglea BioTherapeutics. External 3 Aeglea BioTherapeutics
At Aeglea, we believe that every patient deserves a chance at a better life. We are committed to helping people with rare and devastating metabolic diseases who have limited treatment options. Having a rare disease does not mean that you are in the fight alone.

©2022 Aeglea BioTherapeutics. External 4 Program Highlights and Upcoming Milestones Pegzilarginase in ARG1-D:
Actively engaged with FDA to identify potential paths to address clinical request in Refusal to File letter and progress the program towards potential U.S. approval
On track with EU regulatory activities AGLE-177 in Homocystinuria:
Expecting clinical data from Phase 1/2 clinical trial in fourth quarter of 2022
Enrolling patients in sites in UK, Australia and U.S. Pipeline:
Postponing certain activities for AGLE-325 in Cystinuria due to strategic prioritization and manufacturing constraints
Strategically advancing research and molecule optimization for undisclosed rare diseases
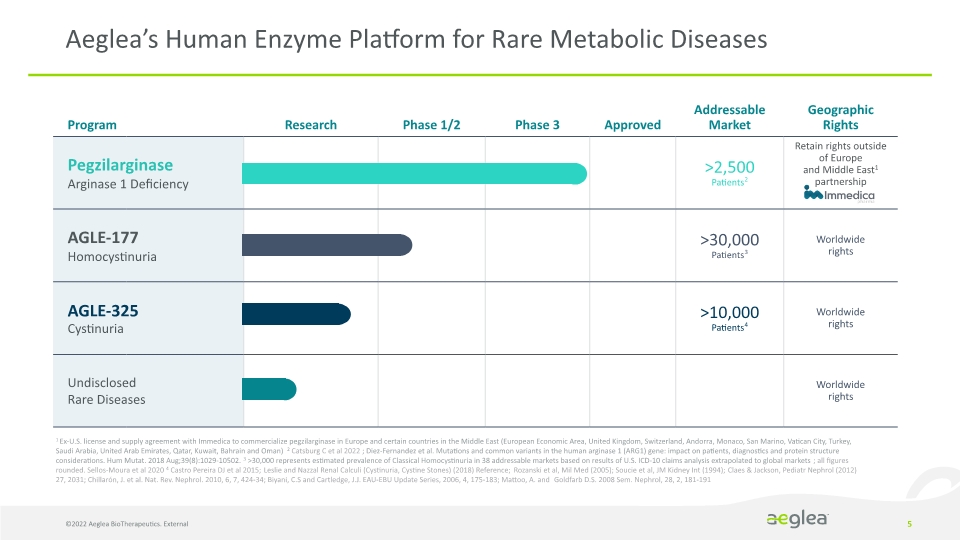
5 Aeglea’s Human Enzyme Platform for Rare Metabolic Diseases 1 Ex-U.S. license and supply agreement with Immedica to commercialize pegzilarginase in Europe and certain countries in the Middle East (European Economic Area, United Kingdom, Switzerland, Andorra, Monaco, San Marino, Vatican City, Turkey, Saudi Arabia, United Arab Emirates, Qatar, Kuwait, Bahrain and Oman) 2 Catsburg C et al 2022 ; Diez-Fernandez et al. Mutations and common variants in the human arginase 1 (ARG1) gene: impact on patients, diagnostics and protein structure considerations. Hum Mutat. 2018 Aug;39(8):1029-10502. 3 >30,000 represents estimated prevalence of Classical Homocystinuria in 38 addressable markets based on results of U.S. ICD-10 claims analysis extrapolated to global markets; all figures rounded. Sellos-Moura et al 2020 4 Castro Pereira DJ et al 2015; Leslie and Nazzal Renal Calculi (Cystinuria, Cystine Stones) (2018) Reference; Rozanski et al, Mil Med (2005); Soucie et al, JM Kidney Int (1994); Claes & Jackson, Pediatr Nephrol (2012) 27, 2031; Chillarón, J. et al. Nat. Rev. Nephrol. 2010, 6, 7, 424-34; Biyani, C.S and Cartledge, J.J. EAU-EBU Update Series, 2006, 4, 175-183; Mattoo, A. and Goldfarb D.S. 2008 Sem. Nephrol, 28, 2, 181-191 ©2022 Aeglea BioTherapeutics. External

Pegzilarginase for Arginase 1 Deficiency

The Progressive Impact of Persistently High Plasma Arginine1-4 Arginase 1 Deficiency (ARG1-D) Disease Overview 7 Infancy2-4,7,8
Initial 6-12 months may be uneventful
May present with seizures, episodes of hyperammonemia: irritability, feeding difficulties, poor appetite, nausea/vomiting, decreased alertness
Toddlerhood (2-4 years) 2,3,7-9
Spasticity in lower limbs (toe walking)
Seizures, avoidance of protein is common and intellectual disability Childhood (5-10 years) 2,3,5,6,9
Progressive spasticity and growth impairment
Declining neuromotor and intellectual capabilities
Increasing dependence on walking aids
Loss of mobility
Decrease/loss of vocabulary/language
Adolescence (11-17 years)2,3,5,10
Potential loss of ambulation and bowel/bladder control
Severe intellectual disability Adulthood (18+ years) 2,3,11,12
Continued decline with increasing disability that may result in early mortality 1 Diez-Fernandez C, et al. Hum Mutat. 2018;39:1029-1050. 2 Carvalho DR, et al. Pediatr Neurol. 2012;46:369-374. 3 Crombez EA, Cederbaum SD. Mol Genet Metab. 2005;84:243-251. 4 De Deyn PP, et al. Hyperargininemia: a treatable inborn error of metabolism. In: Guanidino Compounds in Biology and Medicine. London, UK: John Libbey Company Ltd.; 1997:53-69. 5 Prasad A, et al. J Child Neurol. 1997;12:301-309. 6 Amayreh W, et al. Dev Med Child Neurol. 2014;56:1021-1024. 7 Scaglia F, Lee B. Am J Med Genet C Semin Med Genet. 2006;142C:113-120. 8 Sin YY, et al. J Mol Med (Berl). 2015;93:1287-1296. 9 Cai X, et al. Medicine (Baltimore). 2018;97:e9880. 10 Schlune A, et al. J. Amino Acids. 2015;47:1751-1762. 11 Sun A, et al. Arginase deficiency. In: Adam MP, et al, eds. GeneReviews®. Seattle, WA: University of Washington, Seattle; 2020. 12 Diaz GA, et al. Poster presented at: 13th European Paediatric Neurology Society (EPNS) Congress; September 17-21, 2019; Athens, Greece. Poster P06-34. Current Standard of Care
Focused on lowering plasma arginine levels and controlling hyperammonemia with:
Severe dietary protein restriction
Amino acid supplementation
Ammonia scavengers
Ineffective at controlling plasma arginine levels
Significant Unmet Need
High arginine levels
Severe and progressive disease with early mortality
Easily diagnosed but often missed due to lack of awareness
No approved therapies to address high arginine levels
ARG1-D is a serious, progressive disease with early mortality and high unmet medical need. It is caused by a mutation in the arginase 1 enzyme, resulting in persistently high levels of arginine. ©2022 Aeglea BioTherapeutics. External
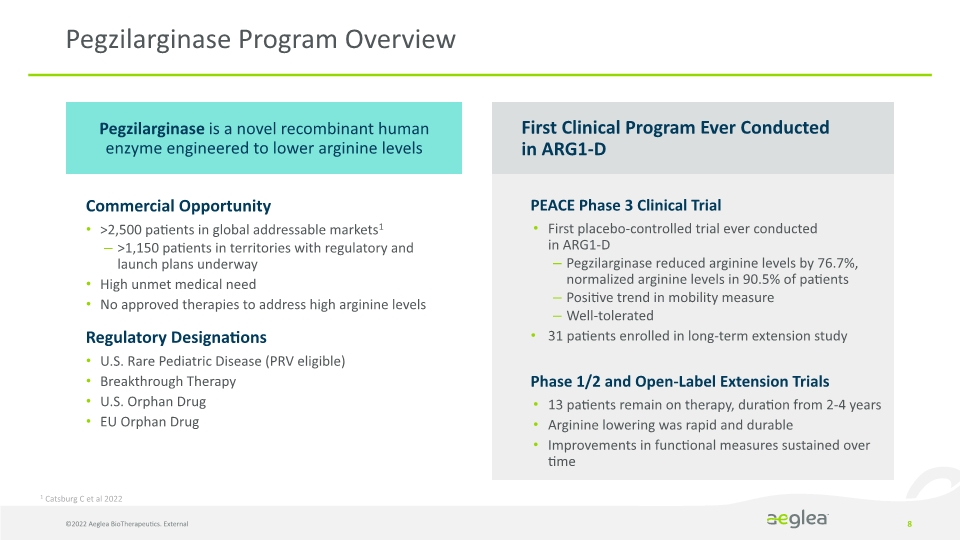
8 Pegzilarginase Program Overview Commercial Opportunity
>2,500 patients in global addressable markets1
>1,150 patients in territories with regulatory and launch plans underway
High unmet medical need
No approved therapies to address high arginine levels
Regulatory Designations
U.S. Rare Pediatric Disease (PRV eligible)
Breakthrough Therapy
U.S. Orphan Drug
EU Orphan Drug
Pegzilarginase is a novel recombinant human enzyme engineered to lower arginine levels PEACE Phase 3 Clinical Trial
First placebo-controlled trial ever conducted in ARG1-D
Pegzilarginase reduced arginine levels by 76.7%, normalized arginine levels in 90.5% of patients
Positive trend in mobility measure
Well-tolerated
31 patients enrolled in long-term extension study
Phase 1/2 and Open-Label Extension Trials
13 patients remain on therapy, duration from 2-4 years
Arginine lowering was rapid and durable
Improvements in functional measures sustained over time First Clinical Program Ever Conducted in ARG1-D ©2022 Aeglea BioTherapeutics. External 1 Catsburg C et al 2022
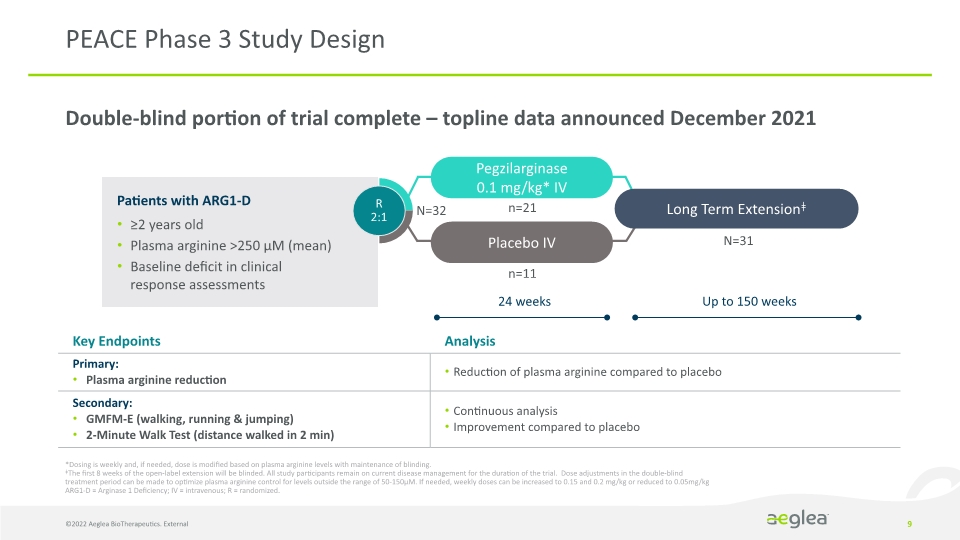
Double-blind portion of trial complete – topline data announced December 2021 9 PEACE Phase 3 Study Design *Dosing is weekly and, if needed, dose is modified based on plasma arginine levels with maintenance of blinding.
ǂThe first 8 weeks of the open-label extension will be blinded. All study participants remain on current disease management for the duration of the trial. Dose adjustments in the double-blind treatment period can be made to optimize plasma arginine control for levels outside the range of 50-150µM. If needed, weekly doses can be increased to 0.15 and 0.2 mg/kg or reduced to 0.05mg/kg
ARG1-D = Arginase 1 Deficiency; IV = intravenous; R = randomized. Patients with ARG1-D
≥2 years old
Plasma arginine >250 µM (mean)
Baseline deficit in clinical response assessments Placebo IV R 2:1 Long Term Extensionǂ Pegzilarginase
0.1 mg/kg* IV 24 weeks Up to 150 weeks n=11 N=32 n=21 N=31 ©2022 Aeglea BioTherapeutics. External
10
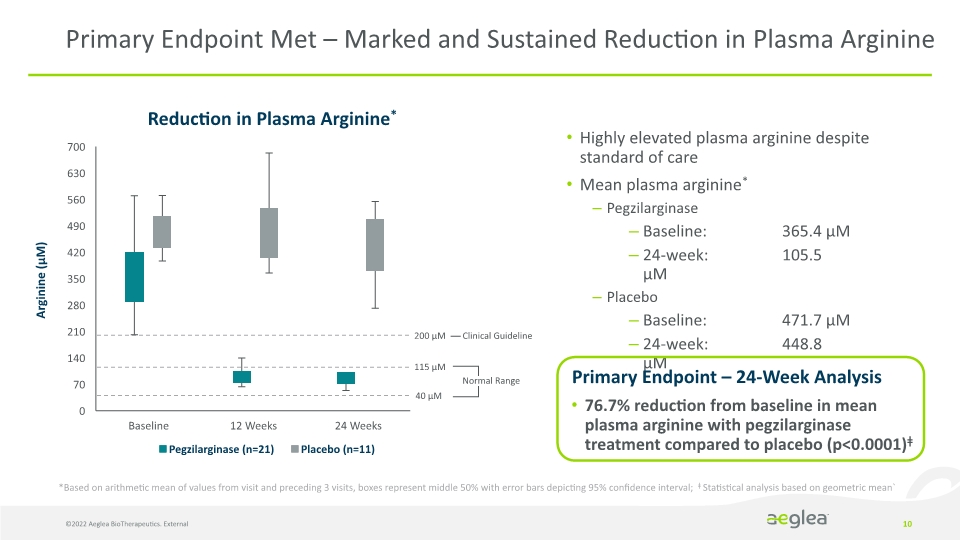
Highly elevated plasma arginine despite standard of care
Mean plasma arginine*
Pegzilarginase
Baseline: 365.4 µM
24-week: 105.5 µM
Placebo
Baseline: 471.7 µM
24-week: 448.8 µM
Primary Endpoint Met – Marked and Sustained Reduction in Plasma Arginine Reduction in Plasma Arginine* *Based on arithmetic mean of values from visit and preceding 3 visits, boxes represent middle 50% with error bars depicting 95% confidence interval; ǂ Statistical analysis based on geometric mean` Clinical Guideline ©2022 Aeglea BioTherapeutics. External Primary Endpoint – 24-Week Analysis
76.7% reduction from baseline in mean plasma arginine with pegzilarginase treatment compared to placebo (p<0.0001)ǂ

90.5% of pegzilarginase treated patients achieved normal plasma arginine levels (p<0.0001)*
No meaningful change in plasma arginine levels in placebo arm
Treatment with pegzilarginase surpassed current clinical guideline goals of reducing arginine levels below 200 µM 11 Normalization of Plasma Arginine Levels in ARG1-D Patients Proportion of Patients Reaching Normal Arginine Levels (40-115 µM) Potential Transformative Therapy for a Patient Population with Previously Unachievable Control of Arginine to Normal Levels * Nominal p value, based on arithmetic mean of values from visit and preceding 3 visits ©2022 Aeglea BioTherapeutics. External

Mobility Assessed by GMFM-E and 2MWT 12 Mean Change from Baseline in 2MWT Mean Change from Baseline in GMFM-E 1 MCID is minimal clinically important difference defined in Cerebral Palsy population. 2Oeffinger D et al 2008; GMFM-E = Gross Motor Function Measure Part E; 2MWT = 2-minute walk test; GMFCS = Gross Motor Function Classification System ; LS = least squares ©2022 Aeglea BioTherapeutics. External
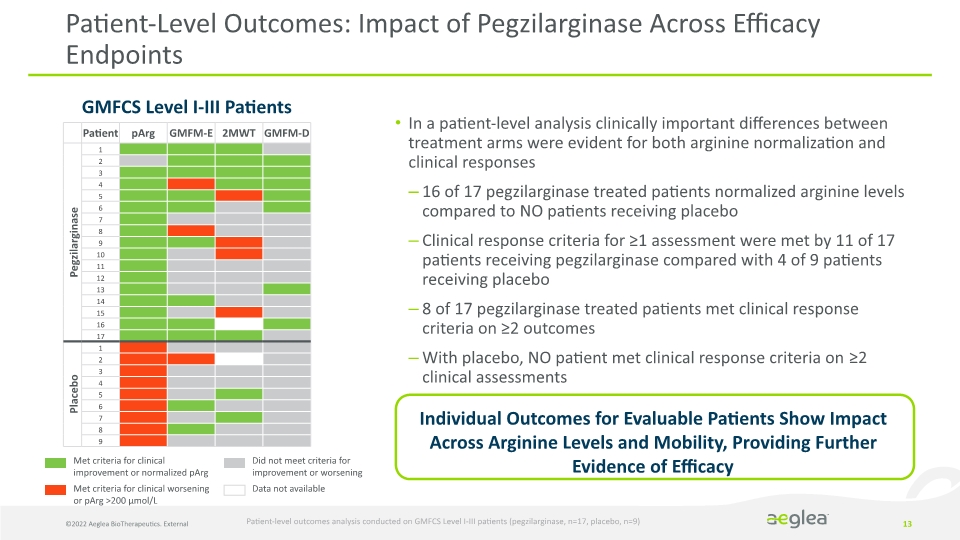
©2022 Aeglea BioTherapeutics. External 13 In a patient-level analysis clinically important differences between treatment arms were evident for both arginine normalization and clinical responses
16 of 17 pegzilarginase treated patients normalized arginine levels compared to NO patients receiving placebo
Clinical response criteria for ≥1 assessment were met by 11 of 17 patients receiving pegzilarginase compared with 4 of 9 patients receiving placebo
8 of 17 pegzilarginase treated patients met clinical response criteria on ≥2 outcomes
With placebo, NO patient met clinical response criteria on ≥2 clinical assessments
Patient-Level Outcomes: Impact of Pegzilarginase Across Efficacy Endpoints Patient-level outcomes analysis conducted on GMFCS Level I-III patients (pegzilarginase, n=17, placebo, n=9) GMFCS Level I-III Patients Met criteria for clinical improvement or normalized pArg
Met criteria for clinical worsening or pArg >200 µmol/L Did not meet criteria for improvement or worsening
Data not available

©2022 Aeglea BioTherapeutics. External 14 Patient-Level Outcomes: Mobility in Select Patients Pegzilarginase-Treated Patients Achieving Clinical Response on ≥2 Mobility Outcomes and No Worsening at Week 24 Ages are at the time of enrollment; Normalization defined at ±15% of age/sex-matched mean distance per the NIH toolbox; * Reflects change from Week 12, distance at baseline not assessed owing to age
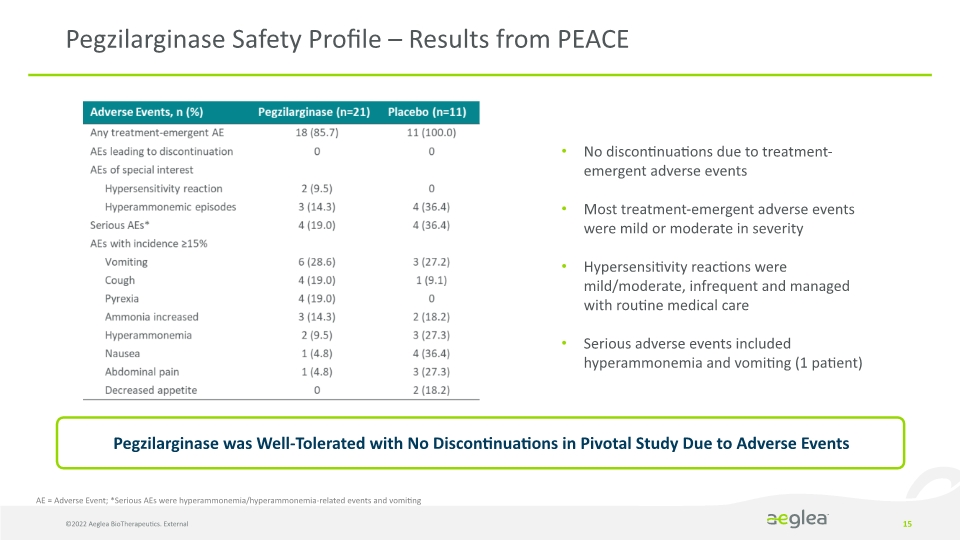
15 Pegzilarginase Safety Profile – Results from PEACE AE = Adverse Event; *Serious AEs were hyperammonemia/hyperammonemia-related events and vomiting Pegzilarginase was Well-Tolerated with No Discontinuations in Pivotal Study Due to Adverse Events No discontinuations due to treatment-emergent adverse events
Most treatment-emergent adverse events were mild or moderate in severity
Hypersensitivity reactions were mild/moderate, infrequent and managed with routine medical care
Serious adverse events included hyperammonemia and vomiting (1 patient) ©2022 Aeglea BioTherapeutics. External
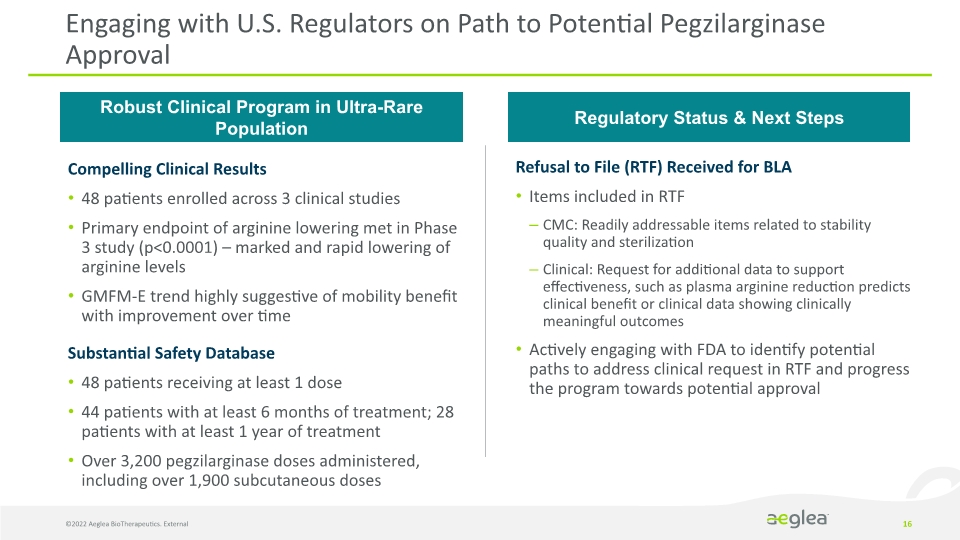
©2022 Aeglea BioTherapeutics. External 16 Engaging with U.S. Regulators on Path to Potential Pegzilarginase Approval Compelling Clinical Results
48 patients enrolled across 3 clinical studies
Primary endpoint of arginine lowering met in Phase 3 study (p<0.0001) – marked and rapid lowering of arginine levels
GMFM-E trend highly suggestive of mobility benefit with improvement over time
Substantial Safety Database
48 patients receiving at least 1 dose
44 patients with at least 6 months of treatment; 28 patients with at least 1 year of treatment
Over 3,200 pegzilarginase doses administered, including over 1,900 subcutaneous doses Robust Clinical Program in Ultra-Rare Population Regulatory Status & Next Steps Refusal to File (RTF) Received for BLA
Items included in RTF
CMC: Readily addressable items related to stability quality and sterilization
Clinical: Request for additional data to support effectiveness, such as plasma arginine reduction predicts clinical benefit or clinical data showing clinically meaningful outcomes
Actively engaging with FDA to identify potential paths to address clinical request in RTF and progress the program towards potential approval

AGLE-177 for Homocystinuria
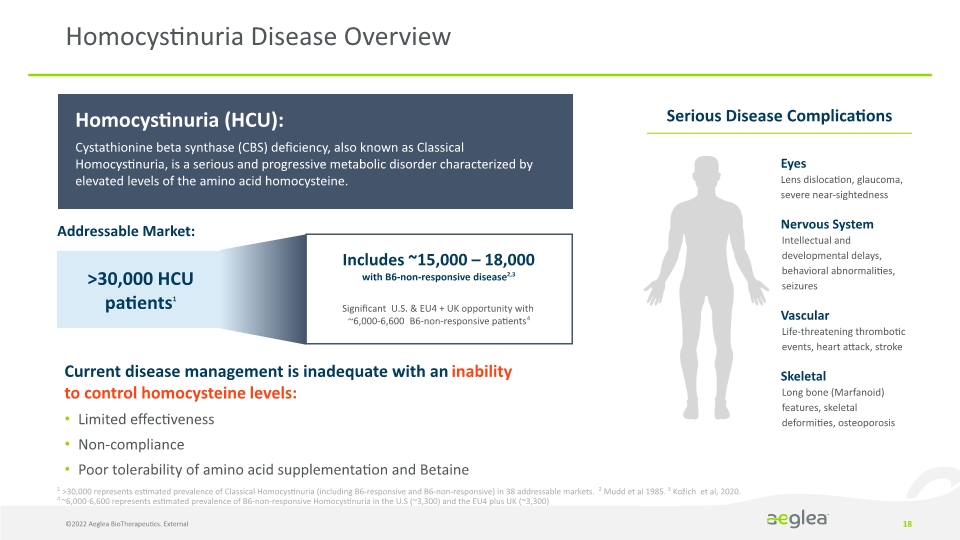
Homocystinuria Disease Overview 18 1 >30,000 represents estimated prevalence of Classical Homocystinuria (including B6-responsive and B6-non-responsive) in 38 addressable markets. 2 Mudd et al 1985. 3 Kožich et al, 2020. 4 ~6,000-6,600 represents estimated prevalence of B6-non-responsive Homocystinuria in the U.S (~3,300) and the EU4 plus UK (~3,300) Homocystinuria (HCU):
Cystathionine beta synthase (CBS) deficiency, also known as Classical Homocystinuria, is a serious and progressive metabolic disorder characterized by elevated levels of the amino acid homocysteine.
Addressable Market: >30,000 HCU patients1 Includes ~15,000 – 18,000
with B6-non-responsive disease2,3
Significant U.S. & EU4 + UK opportunity with
~6,000-6,600 B6-non-responsive patients4
Serious Disease Complications Eyes
Lens dislocation, glaucoma, severe near-sightedness
Nervous System
Intellectual and developmental delays, behavioral abnormalities, seizures
Vascular
Life-threatening thrombotic events, heart attack, stroke
Skeletal
Long bone (Marfanoid) features, skeletal deformities, osteoporosis
Current disease management is inadequate with an inability to control homocysteine levels:
Limited effectiveness
Non-compliance
Poor tolerability of amino acid supplementation and Betaine ©2022 Aeglea BioTherapeutics. External
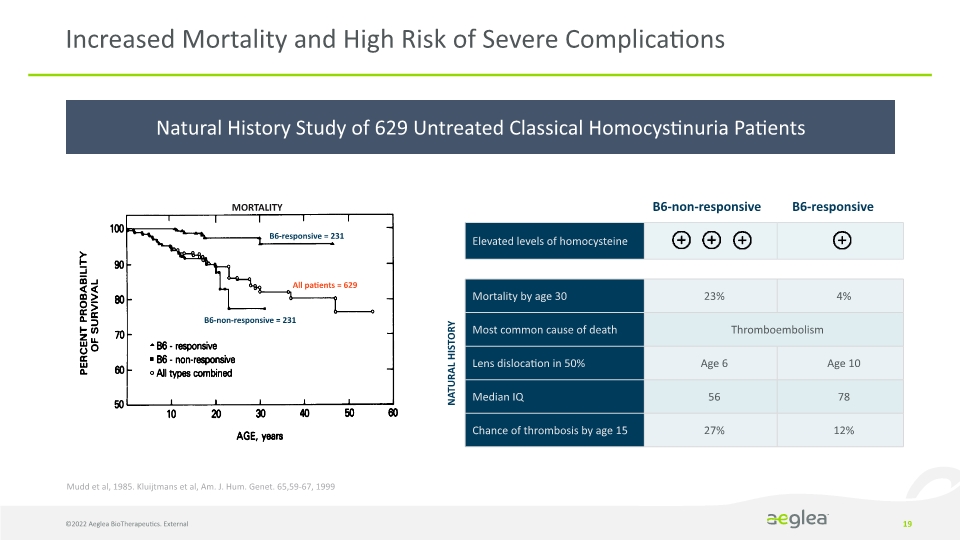
Natural History Study of 629 Untreated Classical Homocystinuria Patients 19 Increased Mortality and High Risk of Severe Complications B6-responsive = 231 All patients = 629 B6-non-responsive = 231 MORTALITY NATURAL HISTORY Mudd et al, 1985. Kluijtmans et al, Am. J. Hum. Genet. 65,59-67, 1999 B6-non-responsive B6-responsive ©2022 Aeglea BioTherapeutics. External
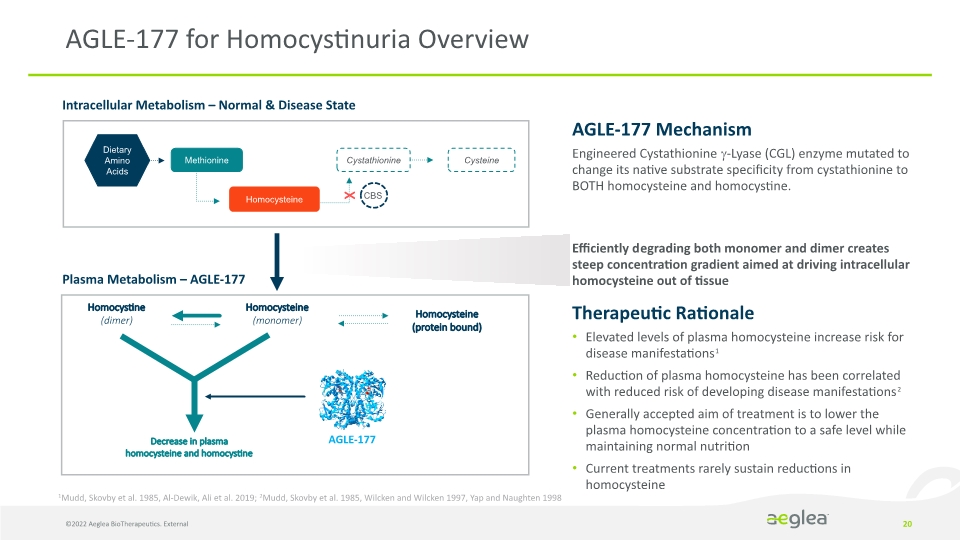
AGLE-177 for Homocystinuria Overview 20 Intracellular Metabolism – Normal & Disease State Plasma Metabolism – AGLE-177 Homocysteine
(monomer) Homocystine
(dimer) AGLE-177 Homocysteine
(protein bound)
Decrease in plasma homocysteine and homocystine X AGLE-177 Mechanism
Engineered Cystathionine -Lyase (CGL) enzyme mutated to change its native substrate specificity from cystathionine to BOTH homocysteine and homocystine.
Efficiently degrading both monomer and dimer creates steep concentration gradient aimed at driving intracellular homocysteine out of tissue
Therapeutic Rationale
Elevated levels of plasma homocysteine increase risk for disease manifestations1
Reduction of plasma homocysteine has been correlated with reduced risk of developing disease manifestations2
Generally accepted aim of treatment is to lower the plasma homocysteine concentration to a safe level while maintaining normal nutrition
Current treatments rarely sustain reductions in homocysteine 1Mudd, Skovby et al. 1985, Al-Dewik, Ali et al. 2019; 2Mudd, Skovby et al. 1985, Wilcken and Wilcken 1997, Yap and Naughten 1998
©2022 Aeglea BioTherapeutics. External

21 Preclinical Data Support Potential Advantages of AGLE-177 Marked survival benefit with total weekly dose as low as 2 mg/kg 1 Daige C. et al. Poster presented at ASHG 2020 Virtual Meeting. 2 Majtan T. et al., Life Sci. 2018; May 1;200:15-25. 3 Mice were maintained on betaine until weaning; tHcy = total homocysteine Twice weekly subcutaneous doses AGLE-177 in CBS -/- mouse model1,3
Significant survival benefit with AGLE-177 at substantially lower total weekly dose of 2 mg/kg compared to a total weekly dose of 22.5 mg/kg reported for a competitor enzyme2 Demonstrated pharmacological effect on homocysteine level in normal animals in toxicology studies Substantial decreases in homocysteine after single subcutaneous dose in toxicology studies
Pharmacological effect in normal animals dosed with AGLE-177 differentiated from reported data for a competitor enzyme2 ©2022 Aeglea BioTherapeutics. External
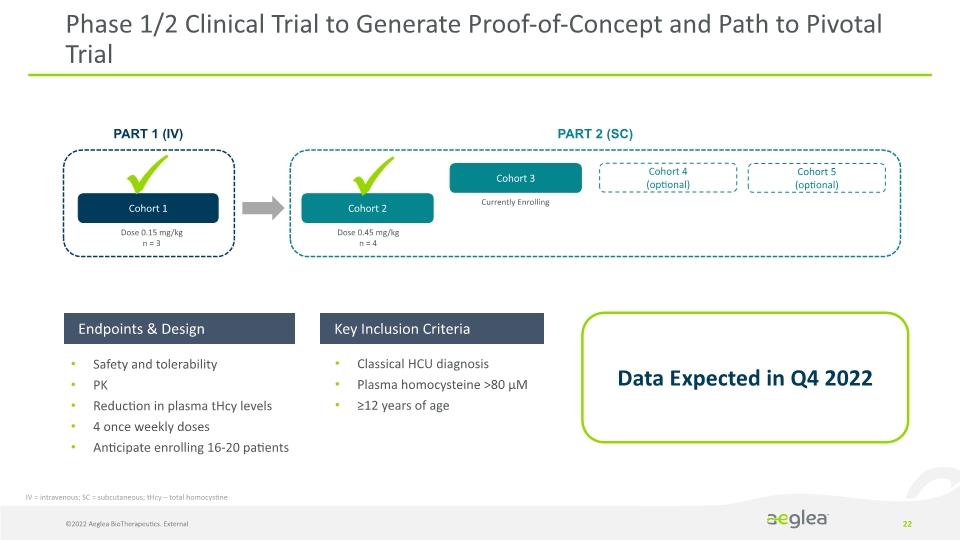
©2022 Aeglea BioTherapeutics. External 22 Phase 1/2 Clinical Trial to Generate Proof-of-Concept and Path to Pivotal Trial Cohort 2 Cohort 3 PART 1 (IV) PART 2 (SC) Dose 0.45 mg/kg
n = 4 Currently Enrolling
Dose 0.15 mg/kg
n = 3 Cohort 1 Cohort 5
(optional) Data Expected in Q4 2022 IV = intravenous; SC = subcutaneous; tHcy – total homocystine Cohort 4
(optional) Endpoints & Design Key Inclusion Criteria Safety and tolerability
PK
Reduction in plasma tHcy levels
4 once weekly doses
Anticipate enrolling 16-20 patients Classical HCU diagnosis
Plasma homocysteine >80 µM
≥12 years of age

23 AGLE-177: a Potentially Differentiated Treatment for Homocystinuria Metabolizes both homocysteine (monomer) and homocystine (dimer)
Showed total homocysteine reduction, no safety concerns in cohort 1 of Phase 1/2 trial
Demonstrated significant survival benefit in CBS -/- mice at low total weekly doses
Reduces homocysteine levels in CBS -/- mice with single and twice weekly dosing
Demonstrated potency to reduce homocysteine from normal levels in normal rats and cynos 1 Dosed subcutaneously; 2 Dosed intravenously; NOAEL = no observed adverse effect level AGLE-177 Key Attributes Phase 1/2 Clinical Data Expected in Q4 2022 and Well-Positioned to Advance into Potential Pivotal Study ©2022 Aeglea BioTherapeutics. External

Human Enzyme Engineering Platform

Cystinuria Overview 25 Cystinuria:
Cystinuria is a metabolic disorder characterized by high levels of amino acid cystine in the urine, leading to the formation of stones in the kidney, ureter and bladder. Persistent stones can result in serious damage to the kidneys, causing an increased risk of hypertension and chronic kidney failure. Addressable Market: Estimated prevalence rate 1:15,000 Based on literature >10,000
Patients in addressable markets
Low cystine in urine Healthy High cystine in urine cystine precipitates stone formation Severe pain, surgical interventions, kidney failure Biyani and Cartledge EAU-EBU Update Series 4 (2006) 175-183, Leslie and Nazzal Renal Calculi (Cystinuria, Cystine Stones) (2018) ©2022 Aeglea BioTherapeutics. External

26 AGLE-325 Preclinical Advancement New candidate advanced through lead optimization with preclinical efficacy and PK/PD profile which support attractive clinical dosing schedule µCT Kidney Stone Volume1 Kidney Pathology1 N=16 N=32 INNOVATIVE ENZYME APPROACH Enzymatic reduction of plasma cystine should lower urine cystine and reduce cystine stone formation 1 Experiments performed using a clinically relevant dose range; PK = pharmacokinetics; PD = pharmacodynamics ©2022 Aeglea BioTherapeutics. External

Leveraging Aeglea’s Next Generation Human Enzyme Platform 27 High activity human pegylated investigational enzyme
Degrades key accumulating metabolite
Results in restorative phenotypic improvements in genetically engineered mouse model PROOF OF CONCEPT Vehicle Control Therapeutic Enzyme ©2022 Aeglea BioTherapeutics. External

Summary

©2022 Aeglea BioTherapeutics. External 29 Program Highlights and Upcoming Milestones Pegzilarginase in ARG1-D:
Actively engaged with FDA to identify potential paths to address clinical request in Refusal to File letter and progress the program towards potential U.S. approval
On track with EU regulatory activities AGLE-177 in Homocystinuria:
Expecting clinical data from Phase 1/2 clinical trial in fourth quarter of 2022
Enrolling patients in sites in UK, Australia and U.S. Pipeline:
Postponing certain activities for AGLE-325 in Cystinuria due to strategic prioritization and manufacturing constraints
Strategically advancing research and molecule optimization for undisclosed rare diseases

805 Las Cimas Parkway Suite 100 Austin, TX 78746 aeglea.com

Financial Summary 31 “Non-GAAP” cash, cash equivalents and marketable securities as of June 30, 2022: $90.41 million (no debt)
As of June 30, 2022, 94.2 million2 common shares and pre-funded warrants outstanding Expected funding runway: through second quarter of 2023 1 Includes $1.8mm of restricted cash. 2Includes 61.2 million common shares outstanding and 33.0 million pre-funded warrants. ©2022 Aeglea BioTherapeutics. External
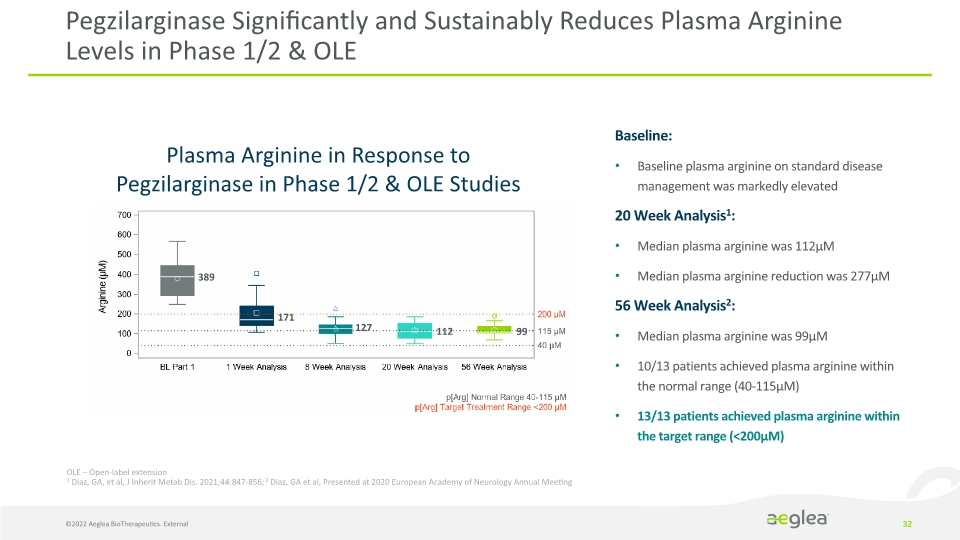
Pegzilarginase Significantly and Sustainably Reduces Plasma Arginine Levels in Phase 1/2 & OLE 32 Plasma Arginine in Response to Pegzilarginase in Phase 1/2 & OLE Studies Baseline:
Baseline plasma arginine on standard disease management was markedly elevated
20 Week Analysis1:
Median plasma arginine was 112µM
Median plasma arginine reduction was 277µM
56 Week Analysis2:
Median plasma arginine was 99µM
10/13 patients achieved plasma arginine within the normal range (40-115µM)
13/13 patients achieved plasma arginine within the target range (<200µM) 389 171 127 112 99 OLE – Open-label extension
1 Diaz, GA, et al, J Inherit Metab Dis. 2021;44:847-856; 2 Diaz, GA et al, Presented at 2020 European Academy of Neurology Annual Meeting ©2022 Aeglea BioTherapeutics. External
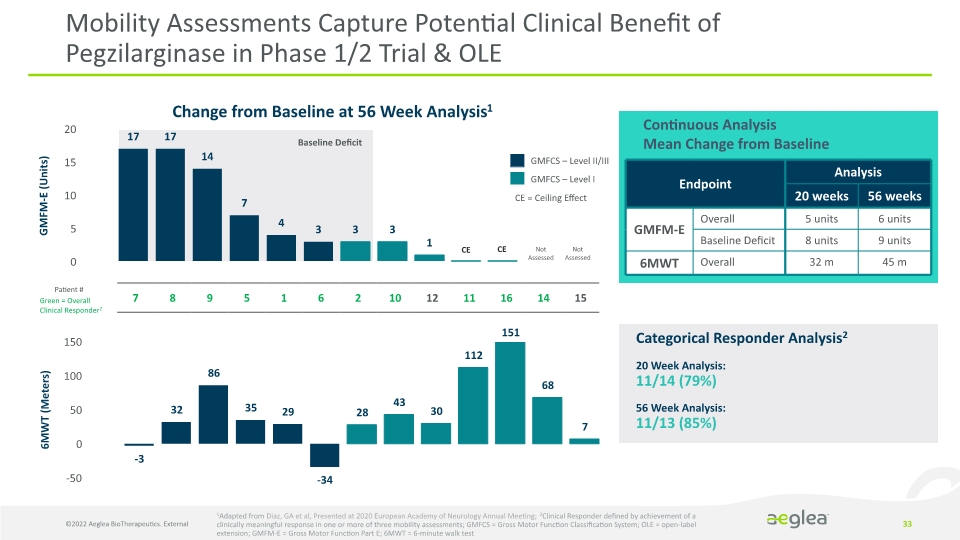
Mobility Assessments Capture Potential Clinical Benefit of Pegzilarginase in Phase 1/2 Trial & OLE 33 Continuous Analysis
Mean Change from Baseline GMFCS – Level II/III GMFCS – Level I CE = Ceiling Effect 20 Week Analysis: 11/14 (79%)
56 Week Analysis: 11/13 (85%) Categorical Responder Analysis2 1Adapted from Diaz, GA et al, Presented at 2020 European Academy of Neurology Annual Meeting; 2Clinical Responder defined by achievement of a clinically meaningful response in one or more of three mobility assessments; GMFCS = Gross Motor Function Classification System; OLE = open-label extension; GMFM-E = Gross Motor Function Part E; 6MWT = 6-minute walk test Change from Baseline at 56 Week Analysis1 Not Assessed Not Assessed CE CE Green = Overall Clinical Responder2 Patient # GMFM-E (Units) 6MWT (Meters) Baseline Deficit 151 ©2022 Aeglea BioTherapeutics. External
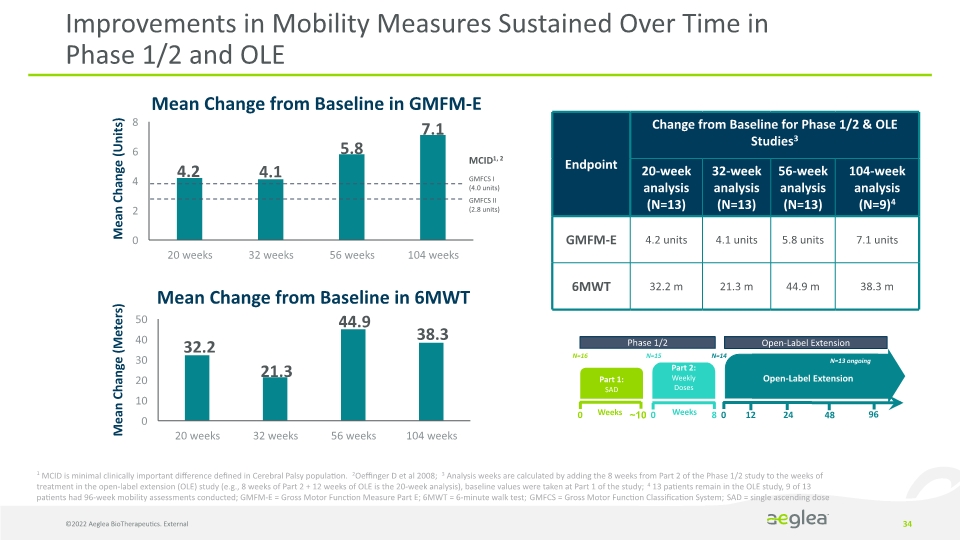
34 Improvements in Mobility Measures Sustained Over Time in Phase 1/2 and OLE 48 1 MCID is minimal clinically important difference defined in Cerebral Palsy population. 2Oeffinger D et al 2008; 3 Analysis weeks are calculated by adding the 8 weeks from Part 2 of the Phase 1/2 study to the weeks of treatment in the open-label extension (OLE) study (e.g., 8 weeks of Part 2 + 12 weeks of OLE is the 20-week analysis), baseline values were taken at Part 1 of the study; 4 13 patients remain in the OLE study, 9 of 13 patients had 96-week mobility assessments conducted; GMFM-E = Gross Motor Function Measure Part E; 6MWT = 6-minute walk test; GMFCS = Gross Motor Function Classification System; SAD = single ascending dose Mean Change from Baseline in GMFM-E Mean Change from Baseline in 6MWT Mean Change (Units) Mean Change (Meters) ©2022 Aeglea BioTherapeutics. External
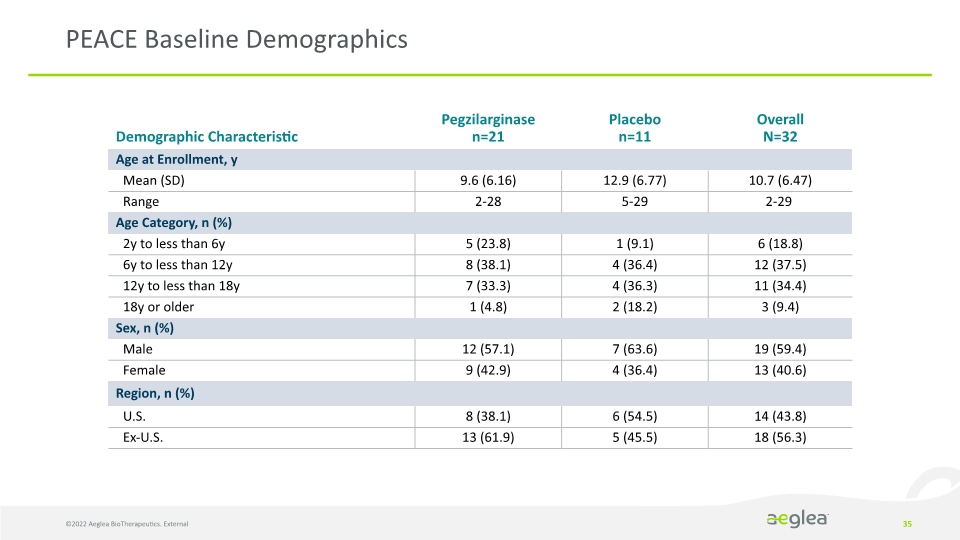
PEACE Baseline Demographics 35 ©2022 Aeglea BioTherapeutics. External
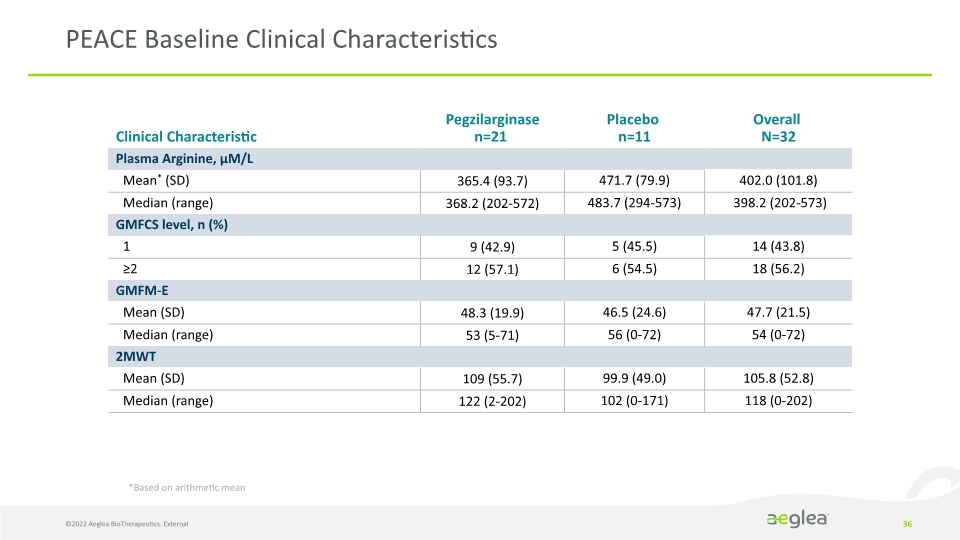
PEACE Baseline Clinical Characteristics 36 *Based on arithmetic mean ©2022 Aeglea BioTherapeutics. External

Measuring the Functional Impact of Spasticity in ARG1-D 37 Gross Motor Function Classification System (GMFCS)
Overall description of current motor function based on:
Movements such as sitting, walking
Use of mobility devices Gross Motor Function Measure (GMFM) Part E
Assesses unaided mobility without bracing or assistive devices
24 tasks involving walking forward/backward, running, jumping and ascending/descending stairs with a score range of 0-72
Timed Walk Tests
Evaluates aided mobility with any needed bracing or assistive devices over a defined period of time
2 mins (2MWT)
6 mins (6MWT) Classification of Mobility: Measures of Change in Mobility: Level V not pictured: adapted seating and assistance with transfers, and utilize wheeled power mobility independently or manual mobility with assistance in most settings GMFCS graphic adapted with permission from The Royal Children’s Hospital Melbourne, Australia ©2022 Aeglea BioTherapeutics. External

38 Attractive Commercial Opportunity and a Targeted Launch Plan 1 Catsburg C et al 2022 2 Licensing and supply agreement signed with Immedica Pharma AB in March 2021; countries in agreement include European Economic Area, United Kingdom, Switzerland, Andorra, Monaco, San Marino, Vatican City, Turkey, Saudi Arabia, United Arab Emirates, Qatar, Kuwait, Bahrain and Oman.
Prevalence of >2,500 Patients1 with Significant Unmet Need United States
>250 patients Western Europe and Middle East2
>900 patients Rest of World
>1,350 patients (including 700 in LatAm, 400 in Asia Pacific) Patient Identification Accelerating the diagnosis and identification of patients through ongoing HCP engagement Disease Education and Awareness Driving HCP dialogue about
the devastating and progressive nature of ARG1-D Access and Reimbursement Ensuring payer understanding of the significant clinical and economic burden of ARG1-D Organizational
Readiness Establishing an internal mindset and infrastructure to meet the needs of the global ARG1-D patient community A Focused Effort to Ensure a Successful Launch ©2022 Aeglea BioTherapeutics. External

Continued patient identification momentum across key markets
Gaining valuable insights into disease management and patient characteristics
Progress is outpacing relevant benchmarks
Driving increased awareness of ARG1-D to facilitate accurate diagnosis
THINK ARGININE™: a disease awareness initiative with diagnostic testing for eligible patients
Focus on key specialties with highest likelihood of potential un- or misdiagnosed patients
Ongoing scientific discourse to broaden awareness and deepen ARG1-D understanding and advocacy among HCP community
High concentration of identified patients in relatively few metabolic institutions with very knowledgeable specialists
Thoughtful engagement with the patient community to strengthen understanding of patient journey and unmet needs Engaging with HCP and Patient Communities to Accelerate Diagnosis, Broaden Awareness, and Deepen Understanding of ARG1-D 39 Patient Identification Disease Education and Awareness ©2022 Aeglea BioTherapeutics. External

Laying the Groundwork to Transition Smoothly to Commercial Operations 40 High-touch patient services model is being developed based on our deep understanding of the needs of the ARG1-D patient community
Thoughtful, targeted approach to payer engagement focused on payers most likely to have ARG1-D covered lives Core commercial and medical team in place with deep ultra-rare launch experience
Commercial formulation on stability and distribution strategy in place
Tight coordination with Immedica, our commercial partner in certain countries in Europe and the Middle East
Commercial infrastructure built with pegzilarginase and earlier-stage pipeline programs in mind Access and Reimbursement Organizational
Readiness ©2022 Aeglea BioTherapeutics. External

AGLE-177 Improves Pathologies and Correction of Disease Manifestations in a Preclinical Model of Homocystinuria 41 Disease Resolution Disease Reversal Dosing Begins Day 10 CBS-/-
Macro-steatosis Disease Progression AGLE-177-HIS 25 mg/kg + Betaine Day 23 CBS-/- AGLE-177-HIS 25 mg/kg
Day 60 CBS-/- PBS + Betaine Day 23 CBS-/-
Macro-steatosis and necrosis Premature Death Healthy liver from
wild-type animal Reversal of Severe Liver Abnormalities in CBS-/- Mouse Model1 Reductions in total plasma homocysteine leads to improvements in disease related abnormalities Effects on the Long-term Pathologies of Osteoporosis2,3 Increased BMD in preclinical model of Homocystinuria with multiple doses of AGLE-177 1 Daige C. et al. Poster presented at ASHG 2018 Annual Meeting 2 Daige C. et al. Poster presented at ASHG 2020 Annual Meeting 3 CBS -/- mice were dosed subcutaneously twice weekly with AGLE-177-HIS starting at D10 through Day 169 were evaluated for bone mineral density (BMD) by dual-energy X-ray absorptiometry; HED = human equivalent dose ©2022 Aeglea BioTherapeutics. External









































