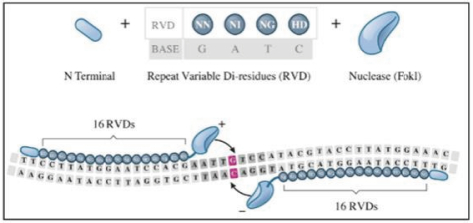
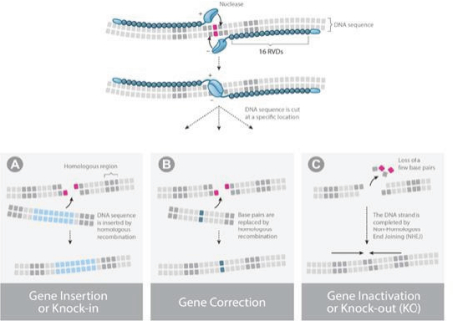
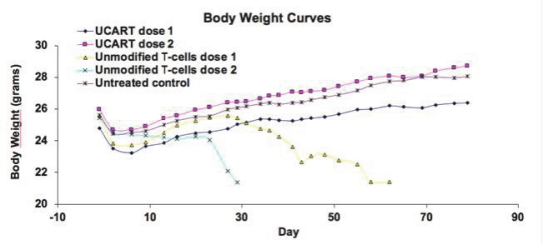
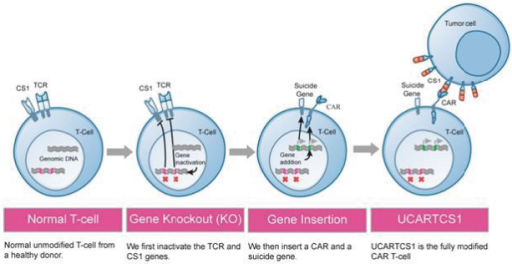
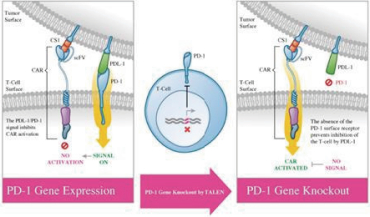
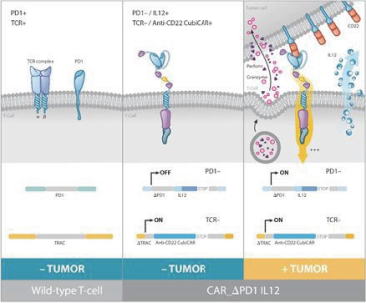
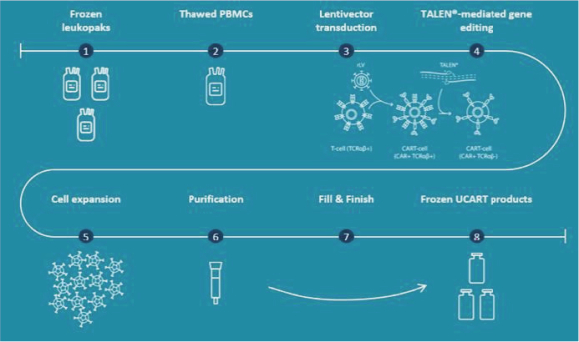
UCART Pipeline
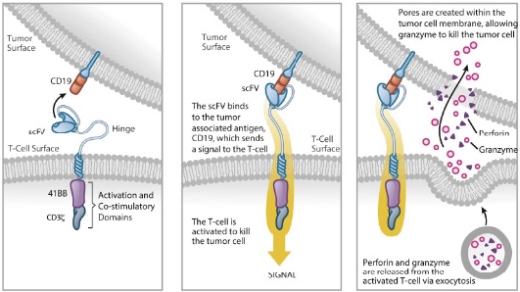
We are developing a series of product candidates for advanced hematologic cancers. Our lead immuno-oncology product candidates, which we refer to as Universal CAR treatment. Each UCART product candidate is designed to target a selected antigen expressed on tumor cells and bears specific engineered attributes, such as inhibition of alloreactivity and compatibility with specific medical regimens that cancer patients may undergo. UCART is the first therapeutic product line that we are developing with our gene-editing platform to address unmet medical needs in oncology. We are focusing our initial internal pipeline in the hematologic cancer space, targeting diseases with high unmet needs such as ALL, AML, NHL, MM and other types of cancers.
T-cells
(UCARTs), are allogeneic CAR T-cells
engineered to be used as an “off-the-shelf”
The following chart highlights our key product candidates:
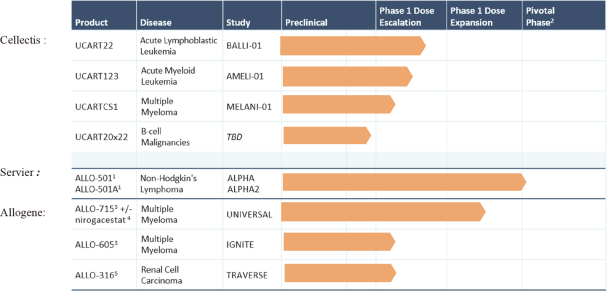
| 1 | ALLO-501 and ALLO-501A are exclusively licensed to Servier and under a joint clinical development program between Servier and Allogene. The ALPHA and ALPHA2 studies targets Diffuse Large B-Cell Lymphoma (DLBCL) and Follicular Lymphoma (FL) indications, which are subtypes of NHL. |
| 2 | Phase 3 may not be required if Phase 2 is registrational. |
| 3 | ALLO-715 and ALLO-605 target BCMA which is a licensed target from Cellectis. ALLO-715 and ALLO-605 utilize TALEN® gene-editing technology pioneered and owned by Cellectis. Allogene has an exclusive license to the Cellectis technology for allogeneic products directed at the BCMA target. Allogene holds global development and commercial rights for this investigational candidate. |
| 4 | Allogene sponsored trial in combination with SpringWorks Therapeutics. |
| 5 | ALLO-316 targets CD70 which is a licensed target from Cellectis. ALLO-316 utilize TALEN® gene-editing technology pioneered and owned by Cellectis. Allogene has an exclusive license to the Cellectis technology for allogeneic products directed at the CD70 target. Allogene holds global development and commercial rights for this investigational candidate. |
Targeted Indications
r/r Acute Lymphoblastic Leukemia (ALL)
ALL is a heterogeneous hematologic disease characterized by the proliferation of immature lymphoid cells in the bone marrow, peripheral blood, and other organs. The proliferation and accumulation of blast cells in the marrow results in suppression of hematopoiesis and, thereafter, anemia, thrombocytopenia, and neutropenia. Extramedullary accumulations of lymphoblasts may occur in various sites, especially the meninges, gonads, thymus, liver, spleen, or lymph nodes. Data from the Surveillance, Epidemiology, and End Results (SEER) database have shown an
age-adjusted
incidence rate of ALL in the United States of 1.7 62