Exhibit 99.1

NASDAQ/TASE: PPBT JANUARY 2021 CORPORATE PRESENTATION

2 Forward - looking Statements and Safe Harbor Certain statements in this presentation that are forward - looking and not statements of historical fact are forward - looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 . Such forward - looking statements include, but are not limited to, statements that are not statements of historical fact, and may be identified by words such as “ believe ” , “ expect ” , “ intend ” , “ plan ” , “ may ” , “ should ” , “ could ” , “ might ” , “ seek ” , “ target ” , “ will ” , “ project ” , “ forecast ” , “ continue ” or “ anticipate ” or their negatives or variations of these words or other comparable words . You should not place undue reliance on these forward - looking statements, which are not guarantees of future performance . Forward - looking statements reflect our current views, expectations, beliefs or intentions with respect to future events, and are subject to a number of assumptions, involve known and unknown risks, many of which are beyond our control, as well as uncertainties and other factors that may cause our actual results, performance or achievements to be significantly different from any future results, performance or achievements expressed or implied by the forward - looking statements . Important factors that could cause or contribute to such differences include, among others, risks relating to : the plans, strategies and objectives of management for future operations ; product development for NT 219 and CM 24 ; the process by which early stage therapeutic candidates such as NT 219 and CM 24 could potentially lead to an approved drug product is long and subject to highly significant risks, particularly with respect to a joint development collaboration ; the fact that drug development and commercialization involves a lengthy and expensive process with uncertain outcomes ; our ability to successfully develop and commercialize our pharmaceutical products ; the expense, length, progress and results of any clinical trials ; the lack of sufficient funding to finance the clinical trials ; the impact of any changes in regulation and legislation that could affect the pharmaceutical industry ; the difficulty in receiving the regulatory approvals necessary in order to commercialize our products ; the difficulty of predicting actions of the U . S . Food and Drug Administration or any other applicable regulator of pharmaceutical products ; the regulatory environment and changes in the health policies and regimes in the countries in which we operate ; the uncertainty surrounding the actual market reception to our pharmaceutical products once cleared for marketing in a particular market ; the introduction of competing products ; patents attained by competitors ; dependence on the effectiveness of our patents and other protections for innovative products ; our ability to obtain, maintain and defend issued patents ; the commencement of any patent interference or infringement action against our patents, and our ability to prevail, obtain a favorable decision or recover damages in any such action ; and the exposure to litigation, including patent litigation, and/or regulatory actions, and other factors that are discussed in our in our Annual Report on Form 20 - F for the year ended December 31 , 2019 and in our other filings with the U . S . Securities and Exchange Commission (the “ SEC ” ), including our cautionary discussion of risks and uncertainties under “ Risk Factors ” in our Registration Statements and Annual Reports . These are factors that we believe could cause our actual results to differ materially from expected results . Other factors besides those we have listed could also adversely affect us . Any forward - looking statement in this presentation speaks only as of the date which it is made . We disclaim any intention or obligation to publicly update or revise any forward - looking statement, or other information contained herein, whether as a result of new information, future events or otherwise, except as required by applicable law . You are advised, however, to consult any additional disclosures we make in our reports to the SEC, which are available on the SEC ’ s website, http : //www . sec . gov .
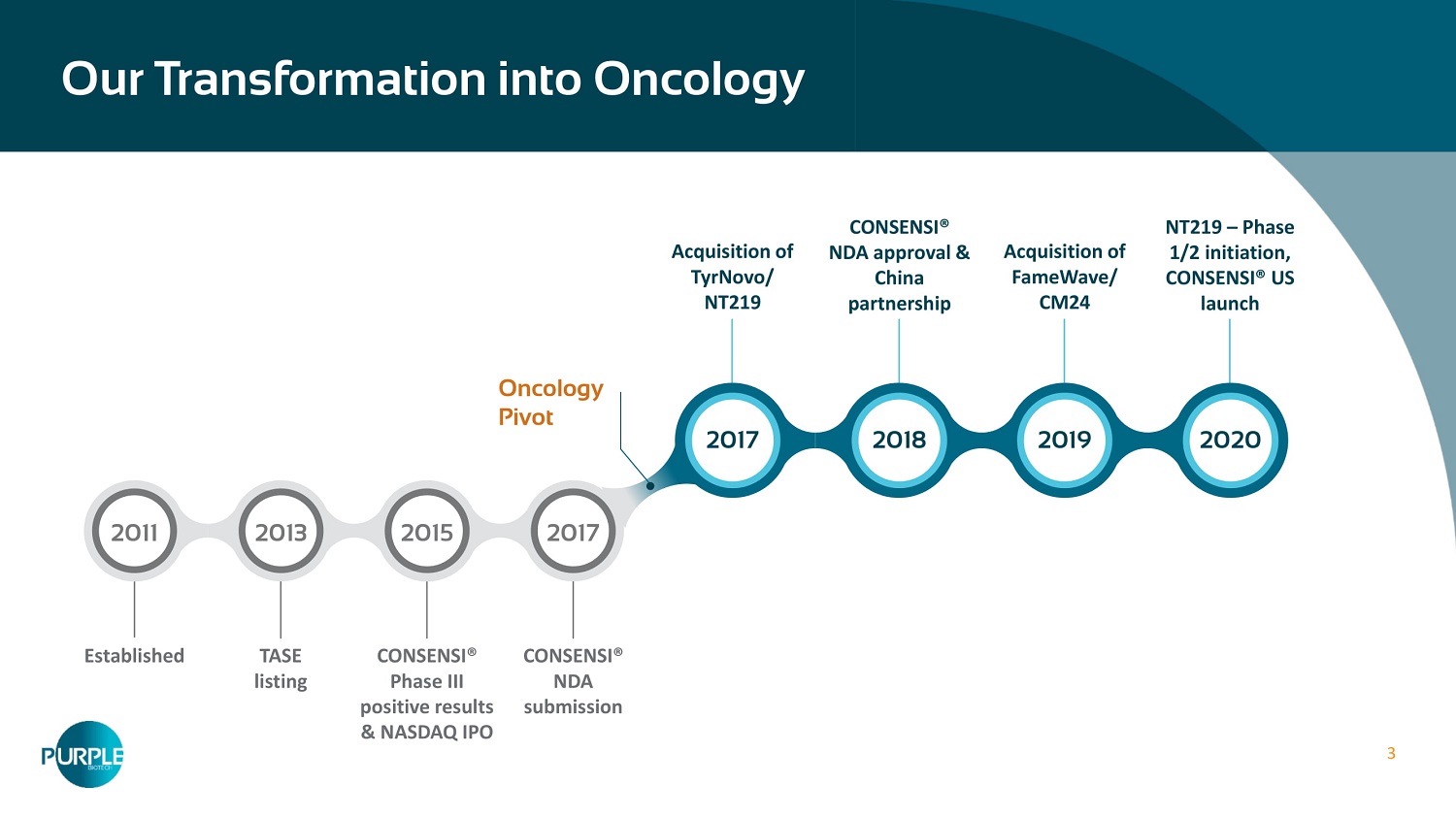
3 Our Transformation into Oncology Acquisition of TyrNovo/ NT 219 CONSENSI® NDA approval & China partnership Acquisition of FameWave/ CM 24 NT 219 – Phase 1 / 2 initiation, CONSENSI® US launch Established TASE listing CONSENSI® Phase III positive results & NASDAQ IPO CONSENSI® NDA submission 2011 2013 2015 2017 2017 2018 2019 2020 Oncology Pivot

4 Advancing Clinical - stage Novel Oncology Therapies Strong balance sheet and cash position Purple Biotech (NASDAQ/TASE: PPBT) CM 24 - First - in - class α - CEACAM 1 mAb, Validating clinical collaboration with NT 219 - First - in - class, small molecule, dual inhibitor of IRS 1 / 2 and STAT 3 H 2:21 - Two phase 1 study readouts $61M cash as of December 31 st , 2020 CONSENSI® commercial royalties supports pipeline development Cash runway through 2024 ADSs outstanding: 17.2 M

5 Expanding Pipeline Multiple data read - outs expected in the next 12 months Program Indication Preclinical Phase 1 Phase 2 Phase 3 Partner Value Drivers CM24 (CEACAM - 1) S olid tumors (monotherapy) (Completed) Expansion arms on RP 2 D: Initiation Q 4:21 Topline data: H 2:21 S olid tumors (combination with nivolumab) NSCLC (combination with nivolumab) Pancreatic Cancer (combination with nivolumab and nab - paclitaxale ) NT 219 (IRS 1 / 2 & STAT 3 ) Solid tumors (monotherapy) Expansion arms on RP 2 D: Initiation Q 4:21 Topline data: H 2:21 R/M HNSCC & CRC (dose escalation with cetuximab) ; R/M HNSCC (expansion - combination with cetuximab on RP2D) Done / Ongoing Planned study Done / Ongoing Expansion

6 Experienced Leadership Isaac Israel Chief Executive Officer Former CEO of BeeContact Ltd. (TASE:BCNT). NextGen Biomed (TASE: NXGN) Eric K. Rowinsky , MD Chairman of the Board Former CMO at ImClone, Stemline , Board member at Biogen Inc. Gil Efron Deputy CEO and Chief Financial Officer Former Deputy CEO & CFO at Kamada (NASDAQ:KMDA) Hadas Reuveni , Ph.D. Vice President, R&D Formerly at Keryx (NASDAQ:KERX) Bertrand Liang, MD, PhD, MBA, AMP Chief Medical Officer Formerly at Biogen Idec, Amgen, NCI Michael Schickler , Ph.D Head of Clinical & Regulatory Affairs Formerly at Hoffmann - La Roche, CEO at CureTech

Advancing First - in - Class Oncology Therapies CM24 - an α - CEACAM1 mAb
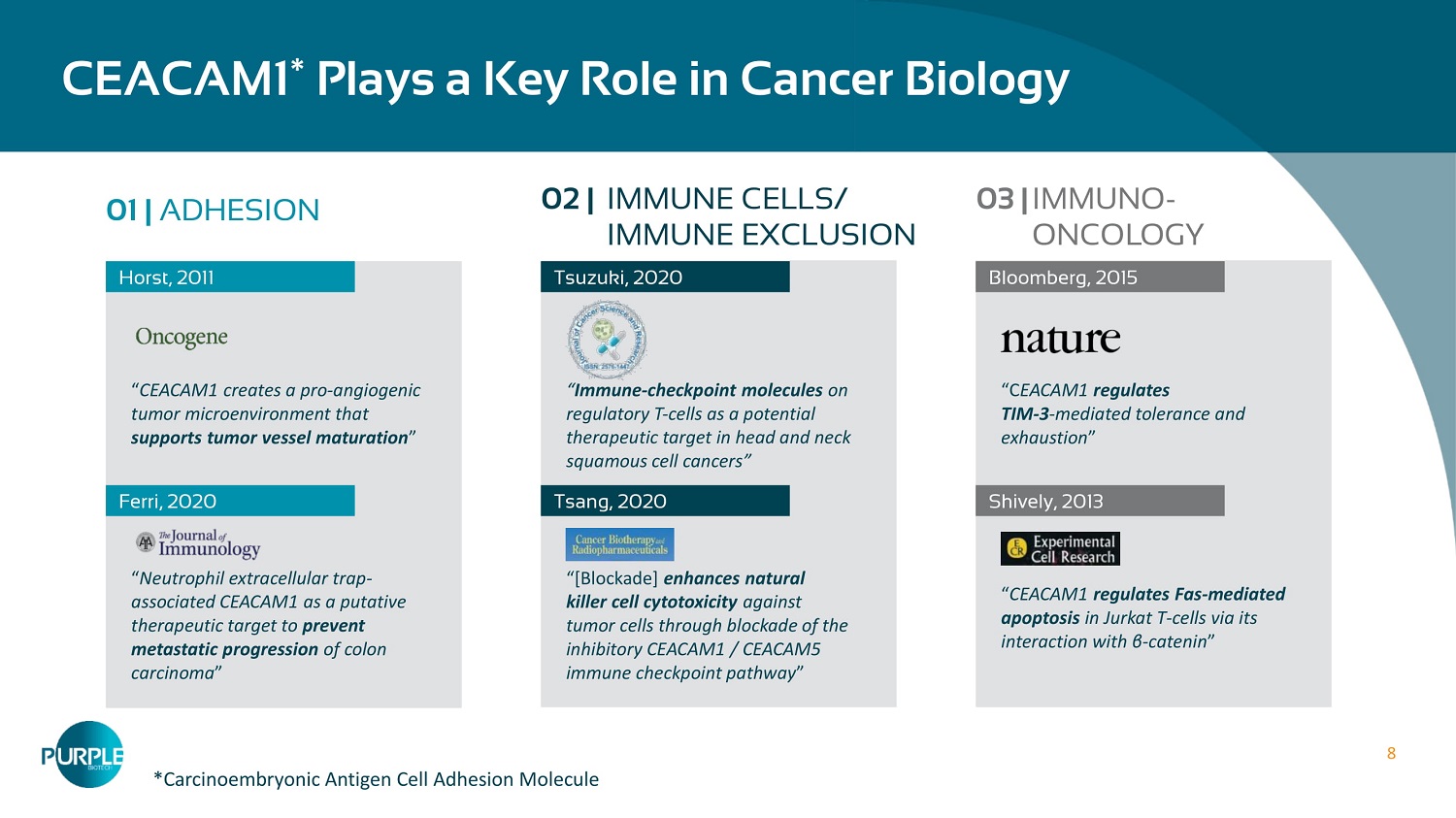
8 CEACAM 1 Ύ Plays a Key Role in Cancer Biology 01 | ADHESION Horst, 2011 “ CEACAM1 creates a pro - angiogenic tumor microenvironment that supports tumor vessel maturation ” “ Neutrophil extracellular trap - associated CEACAM 1 as a putative therapeutic target to prevent metastatic progression of colon carcinoma ” Ferri , 2020 Tsuzuki, 2020 Tsang, 2020 Bloomberg, 2015 “C EACAM1 regulates TIM - 3 - mediated tolerance and exhaustion ” “ CEACAM 1 regulates Fas - mediated apoptosis in Jurkat T - cells via its interaction with β - catenin ” Shively, 2013 02 | IMMUNE CELLS/ IMMUNE EXCLUSION 03 | IMMUNO - ONCOLOGY “ Immune - checkpoint molecules on regulatory T - cells as a potential therapeutic target in head and neck squamous cell cancers ” “[Blockade] enhances natural killer cell cytotoxicity against tumor cells through blockade of the inhibitory CEACAM1 / CEACAM5 immune checkpoint pathway ” *Carcinoembryonic Antigen Cell Adhesion Molecule
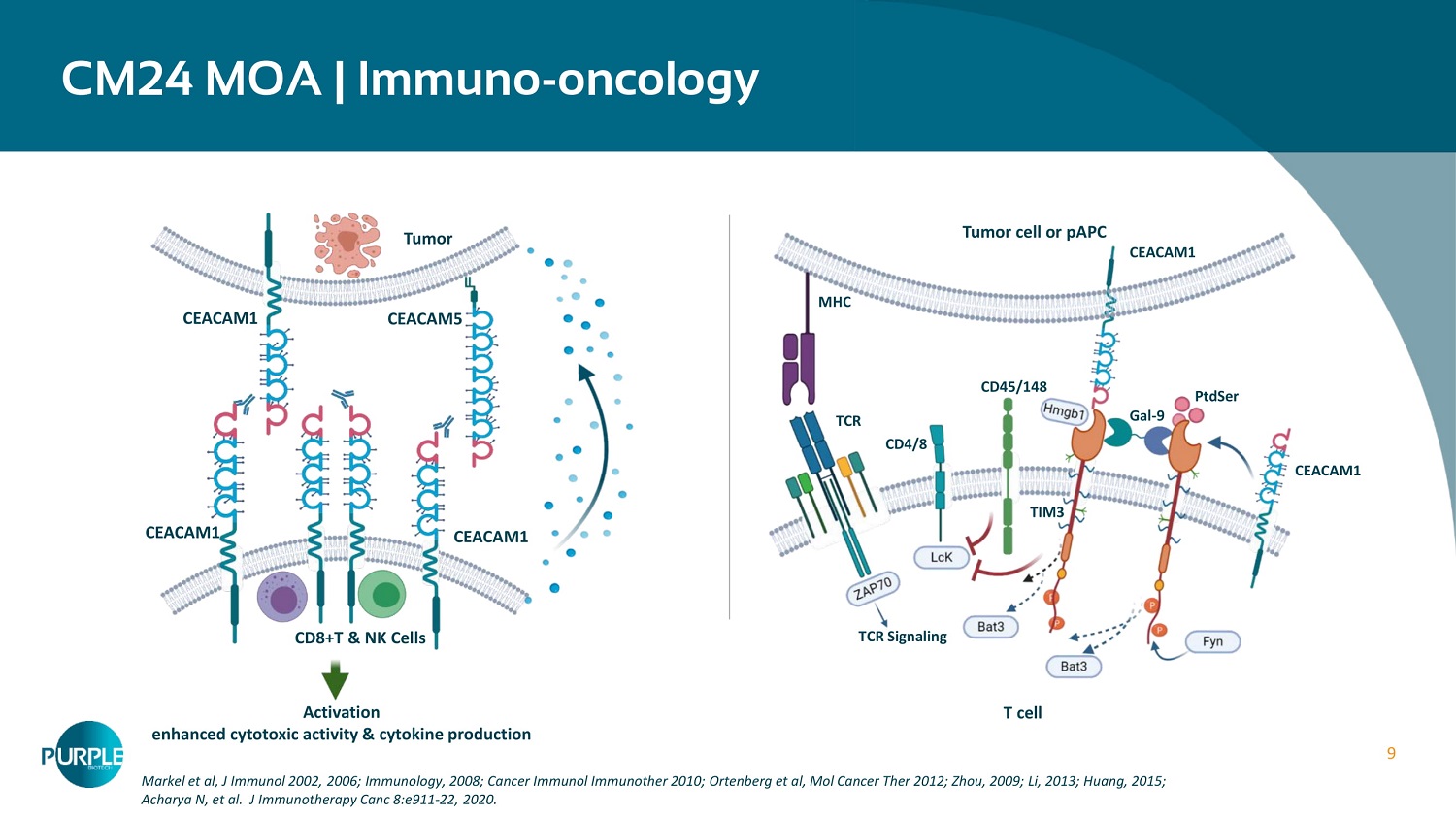
9 CM 24 MOA | Immuno - oncology Markel et al, J Immunol 2002 , 2006 ; Immunology, 2008 ; Cancer Immunol Immunother 2010 ; Ortenberg et al, Mol Cancer Ther 2012 ; Zhou, 2009 ; Li, 2013 ; Huang, 2015 ; Acharya N, et al. J Immunotherapy Canc 8 :e 911 - 22 , 2020 . Tumor CD8+T & NK Cells CEACAM 1 CEACAM1 CEACAM 1 CEACAM5 Activation enhanced cytotoxic activity & cytokine production Tumor cell or pAPC T cell TCR Signaling TIM 3 MHC TCR CD4/8 CD 45 / 148 CEACAM 1 CEACAM1 Gal - 9 PtdSer

10 CM 24 MOA | Adhesion Neutrophil extracellular trap - associated CEACAM1 as a putative therapeutic target to prevent metastatic progression: CEACAM 1 creates a pro - angiogenic tumor microenvironment that supports tumor vessel maturation. CM 24 is proposed to attenuate/block tumor growth and metastatic processes. Rayes RF, et al. Neutrophil Extracellular Trap - Associated CEACAM 1 as a Putative Therapeutic Target to Prevent Metastatic Progression of Colon Carcinoma. J Immunol. 2020 ; Gerstel, D. et al. CEACAM 1 creates a pro - angiogenic tumor microenvironment that supports tumor vessel maturation. Oncogene 30 , 4275 – 4288 ( 2011 ) Primary Tumor Metastasizing CM24 Metastatic Tumor
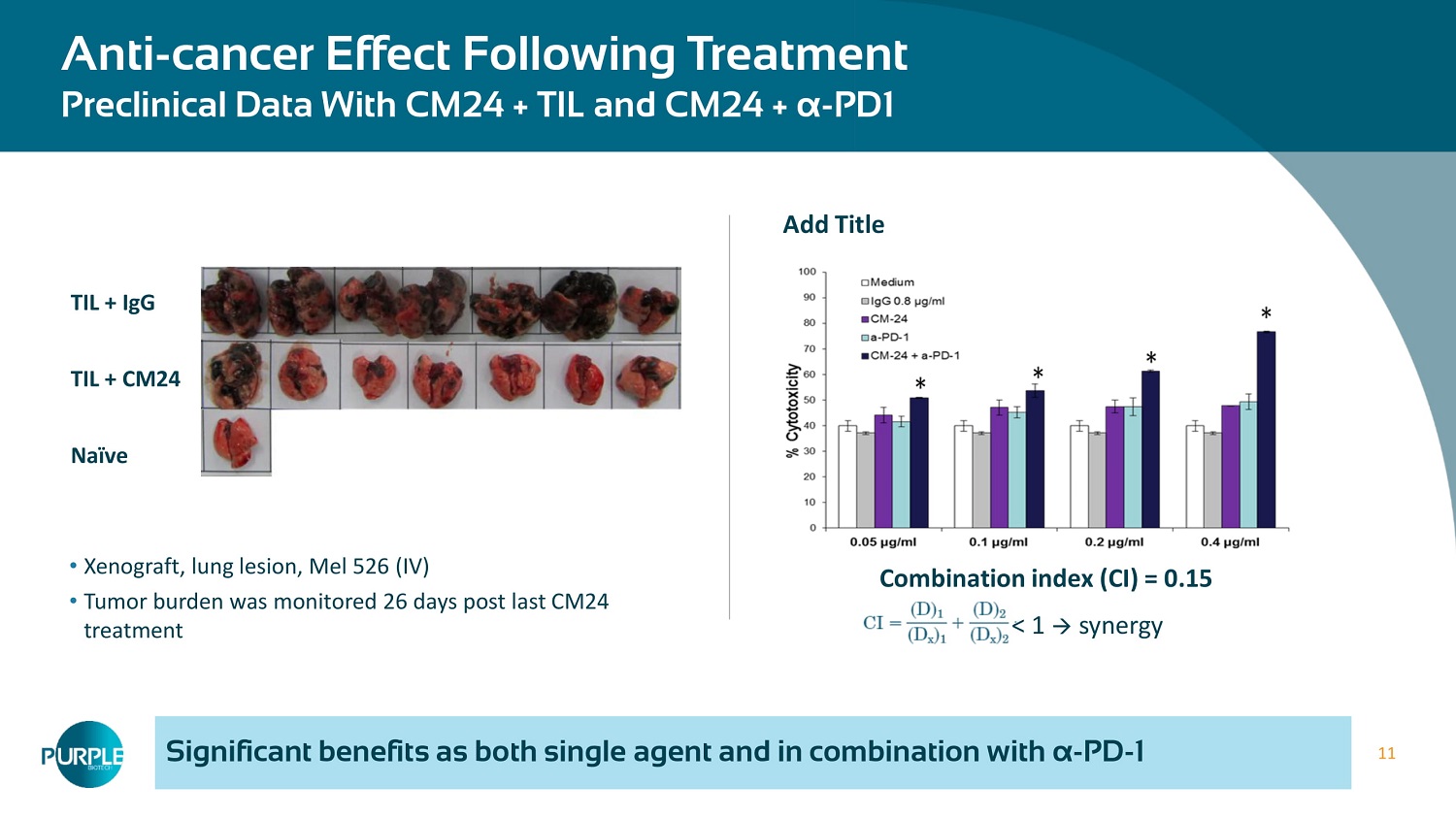
11 Significant benefits as both single agent and in combination with α - PD - 1 Anti - cancer Effect Following Treatment Preclinical Data With CM24 + TIL and CM24 + α - PD1 • Xenograft, lung lesion, Mel 526 (IV) • Tumor burden was monitored 26 days post last CM 24 treatment TIL + IgG TIL + CM 24 Naïve Combination index ( CI) = 0.15 < 1 synergy Add Title
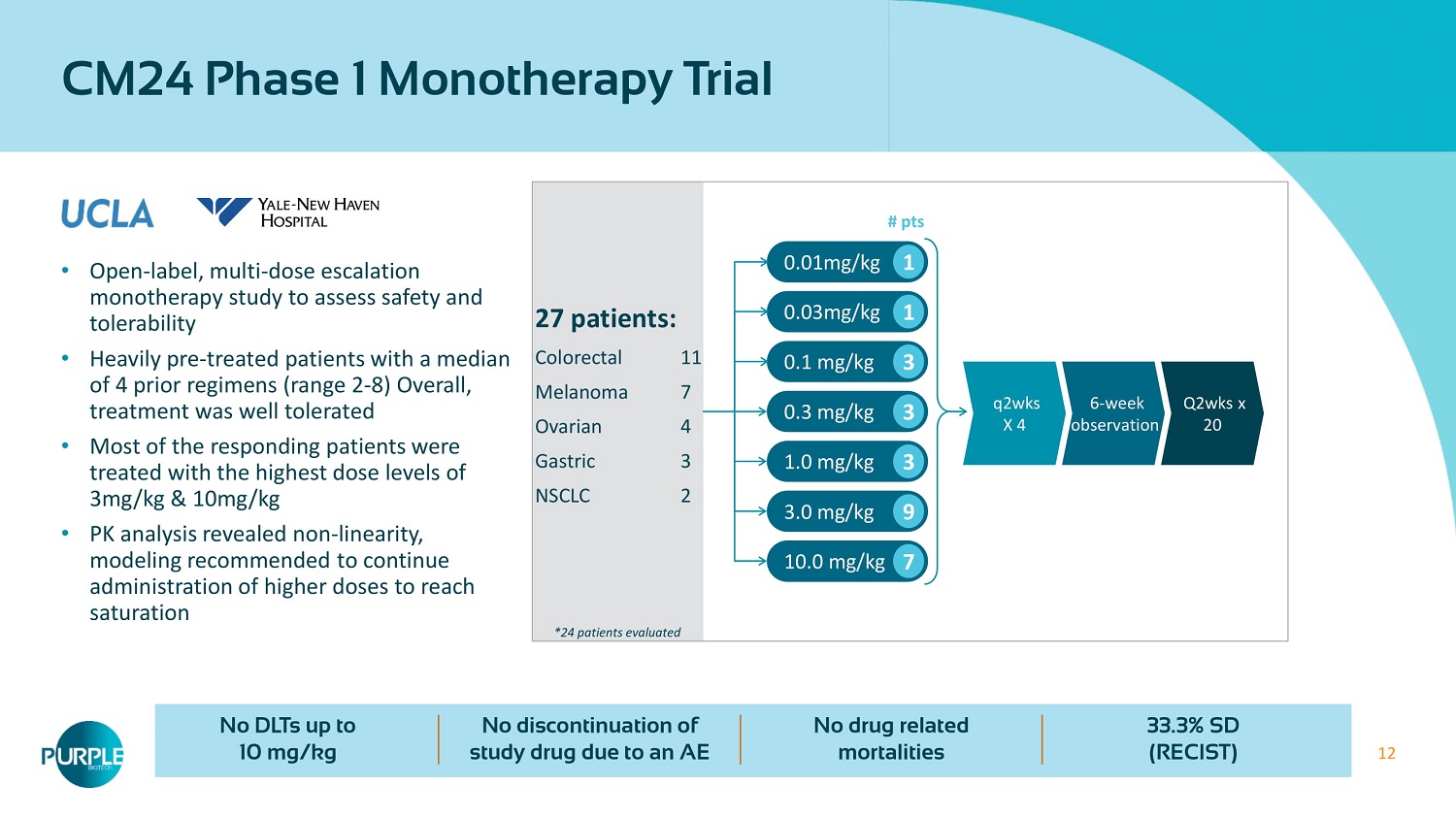
12 • Open - label, multi - dose escalation monotherapy study to assess safety and tolerability • Heavily pre - treated patients with a median of 4 prior regimens (range 2 - 8 ) Overall, treatment was well tolerated • Most of the responding patients were treated with the highest dose levels of 3 mg/kg & 10 mg/kg • PK analysis revealed non - linearity, modeling recommended to continue administration of higher doses to reach saturation CM24 Phase 1 Monotherapy Trial No DLTs up to 10 mg/kg No discontinuation of study drug due to an AE No drug related mortalities 33.3 % SD (RECIST) * 24 patients evaluated # pts 27 patients: Colorectal 11 Melanoma 7 Ovarian 4 Gastric 3 NSCLC 2 0.01mg/kg ϭ 0.03 mg/kg ϭ 0.1 mg/kg ϯ 0.3 mg/kg ϯ 1.0 mg/kg ϯ 3.0 mg/kg ϵ 10.0 mg/kg ϳ Q2wks x 20 6 - week observation q2wks X 4

13 Nivolumab (OPDIVO®) administered Q 2 W or Q 4 W, representing good clinical and commercial fit for CM 24 PK/PD Modeling Provides Dosage & Schedule Guidance Simulated TMDD 1 saturation at Ctrough with Q 2 W regimen Predictions with Q 3 W regimen • Modeling with increased interval between doses demonstrates lower TMDD at 10mg/kg dose • With Q3W, 20mg/kg dosing is not sufficient for saturation across population • Consistent with observed PK showing high clearance at doses < 10 mg/kg, plot shows low TMDD saturation at low doses • 10 mg/kg has a broad range of saturation, and > 10 mg/kg dose is needed for saturation across population 1 Target - mediated drug disposition. 2 OPDIVO® is a registered trademark of Bristol - Myers Squibb. Ctrough is the drug concentration reached by CM24 before the next dose is administered

14 14 Large Market Opportunity in NSCLC & Pancreatic Cancer • CEACAM 1 expression correlates with poor prognosis in NSCLC and Pancreatic cancer 1 • Preclinical data support significant synergy • Receptor saturation with CM 24 is better achieved with a Q 2 W regimen, aligning with the nivolumab regimen • NSCLC accounts for ~ 200 K new cases/year in the US; with a 5 - year relative survival rate of 23 % 2 • Immunotherapy is now recommended as first line therapy in all patients with NSCLC and no driver mutations 3 • 5 - year overall survival rates with chemotherapy in 2 L is 2.6 % and with I/O Opdivo ® is 13.4 % 4 • Pancreatic Cancer accounts for ~60K new cases/year in the US; with a 5 - year relative survival rate of 10% 2 • I/O approaches have been limited to patients with high microsatellite instability (MSI - H) or high tumor mutational burden (TMB - H) • 5 - year overall survival rates with chemotherapy in 2L is 3% 2 Combining nivolumab with CM 24 in a clinical collaboration with 1 Dango et al, Lung Cancer 2008; 60:426 & Calinescu et al, Journal of Immunology Research 2018: 7169081. 2 American Cancer Society, Cancer Facts & Figures 2019, and the ACS website, https://seer.cancer.gov/statfacts/html/pancreas.ht ml 3 Economopoulou P, Mountzios G. The emerging treatment landscape of advanced non - small cell lung cancer. Ann Transl Med. 2018;6(8):138. doi:10.21037/atm.2017.11.07 4 Gettinger S, et al "Five - year outcomes from the randomized, phase III trials CheckMate 017/057: Nivolumab vs docetaxel in previously treated NSCLC" WCLC 2019; Abstract OA14.04.
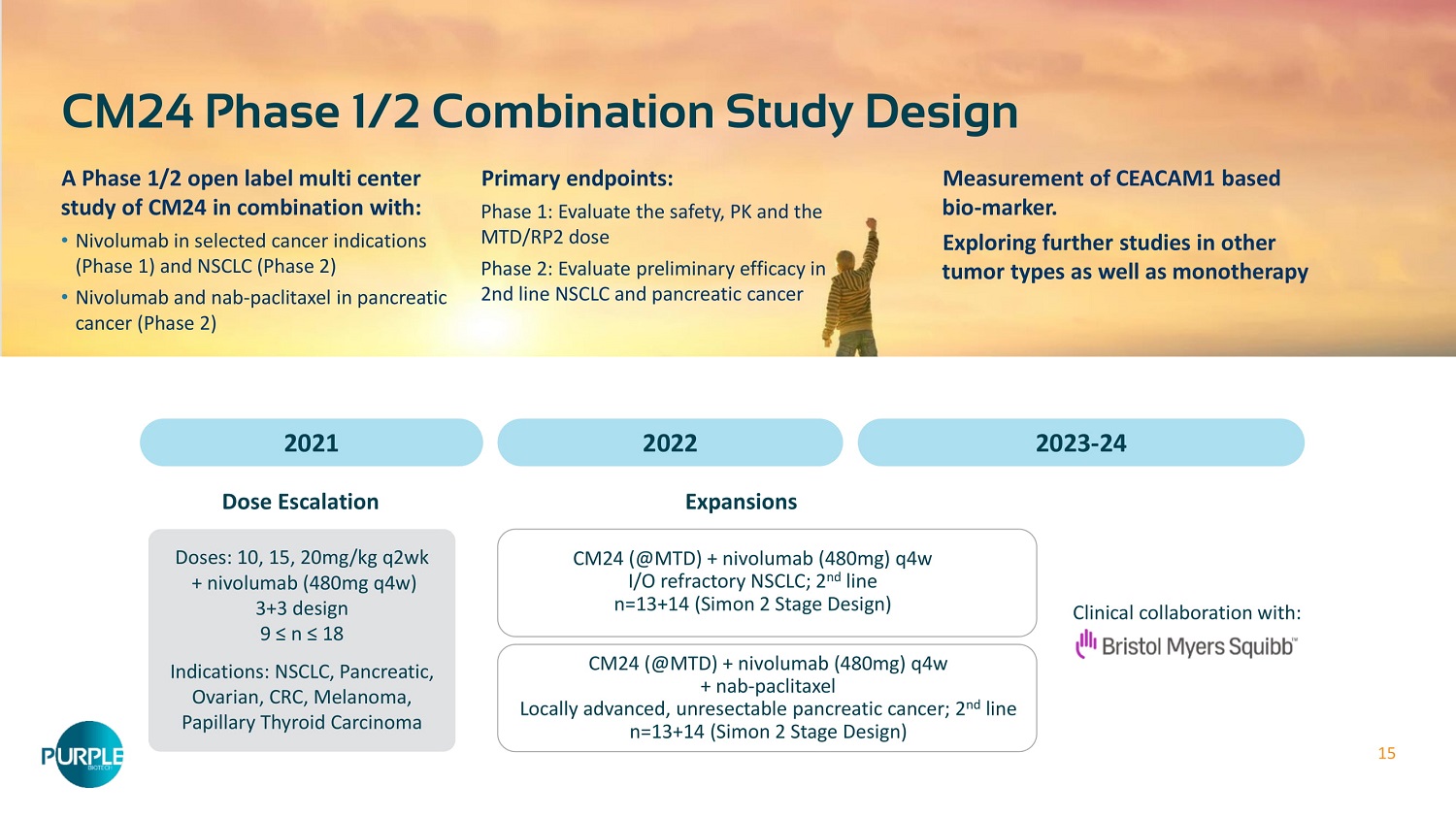
15 CM 24 Phase 1 / 2 Combination Study Design A Phase 1 / 2 open label multi center study of CM 24 in combination with: • Nivolumab in selected cancer indications (Phase 1 ) and NSCLC (Phase 2 ) • Nivolumab and nab - paclitaxel in pancreatic cancer (Phase 2 ) Primary endpoints: Phase 1 : Evaluate the safety, PK and the MTD/RP 2 dose Phase 2 : Evaluate preliminary efficacy in 2 nd line NSCLC and pancreatic cancer Measurement of CEACAM 1 based bio - marker. Exploring further studies in other tumor types as well as monotherapy 2021 2022 2023 - 24 Doses: 10 , 15 , 20 mg/kg q 2 wk + nivolumab ( 480 mg q 4 w) 3 + 3 design 9 ≤ n ≤ 18 Indications: NSCLC, Pancreatic, Ovarian, CRC, Melanoma, Papillary Thyroid Carcinoma Dose Escalation Expansions CM 24 (@MTD) + nivolumab ( 480 mg) q 4 w I/O refractory NSCLC; 2 nd line n= 13 + 14 (Simon 2 Stage Design) CM 24 (@MTD) + nivolumab ( 480 mg) q 4 w + nab - paclitaxel Locally advanced, unresectable pancreatic cancer; 2 nd line n= 13 + 14 (Simon 2 Stage Design) Clinical collaboration with:

Advancing First - in - Class Oncology Therapies NT219 – A Dual Inhibitor of IRS 1/2 and STAT3
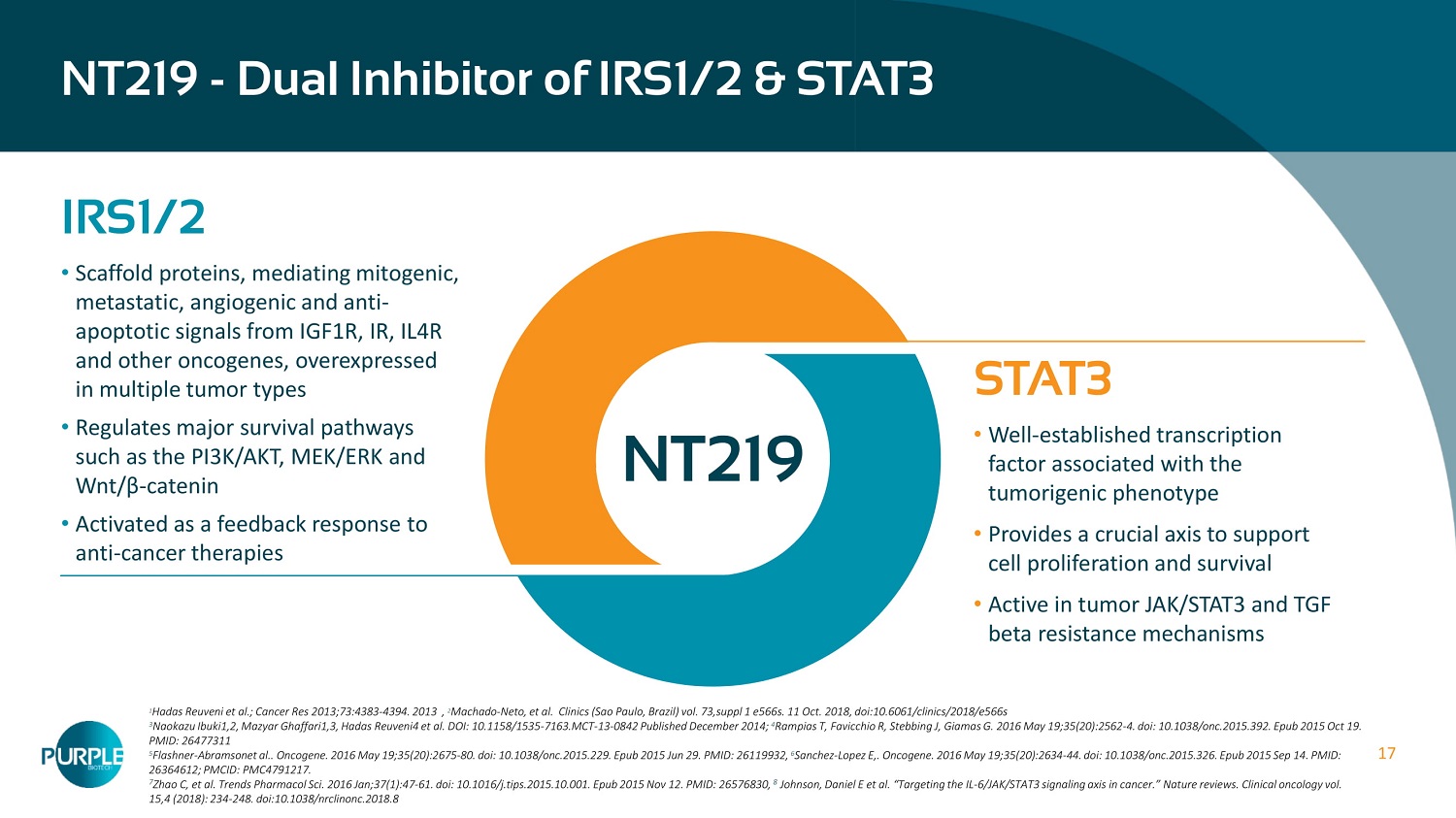
17 NT 219 - Dual Inhibitor of IRS 1 / 2 & STAT 3 NT 219 IRS 1 / 2 • Scaffold proteins, mediating mitogenic, metastatic, angiogenic and anti - apoptotic signals from IGF 1 R, IR, IL 4 R and other oncogenes, overexpressed in multiple tumor types • Regulates major survival pathways such as the PI 3 K/AKT, MEK/ERK and Wnt / β - catenin • Activated as a feedback response to anti - cancer therapies STAT3 • Well - established transcription factor associated with the tumorigenic phenotype • Provides a crucial axis to support cell proliferation and survival • Active in tumor JAK/STAT 3 and TGF beta resistance mechanisms 1 Hadas Reuveni et al.; Cancer Res 2013;73:4383 - 4394. 2013 , 2 Machado - Neto, et al. Clinics (Sao Paulo, Brazil) vol. 73,suppl 1 e566s. 11 Oct. 2018, doi:10.6061/clinics/2018/e566s 3 Naokazu Ibuki1,2, Mazyar Ghaffari1,3, Hadas Reuveni4 et al. DOI: 10.1158/1535 - 7163.MCT - 13 - 0842 Published December 2014; 4 Rampias T, Favicchio R, Stebbing J, Giamas G. 2016 May 19;35(20):2562 - 4. doi: 10.1038/onc.2015.392. Epub 2015 Oct 19. PMID: 26477311 5 Flashner - Abramsonet al.. Oncogene. 2016 May 19;35(20):2675 - 80. doi: 10.1038/onc.2015.229. Epub 2015 Jun 29. PMID: 26119932, 6 Sanchez - Lopez E,. Oncogene. 2016 May 19;35(20):2634 - 44. doi: 10.1038/onc.2015.326. Epub 2015 Sep 14. PMID: 26364612; PMCID: PMC4791217. 7 Zhao C, et al. Trends Pharmacol Sci. 2016 Jan;37(1):47 - 61. doi: 10.1016/j.tips.2015.10.001. Epub 2015 Nov 12. PMID: 26576830, 8 Johnson, Daniel E et al. “Targeting the IL - 6/JAK/STAT3 signaling axis in cancer.” Nature reviews. Clinical oncology vol. 15,4 (2018): 234 - 248. doi:10.1038/nrclinonc.2018.8
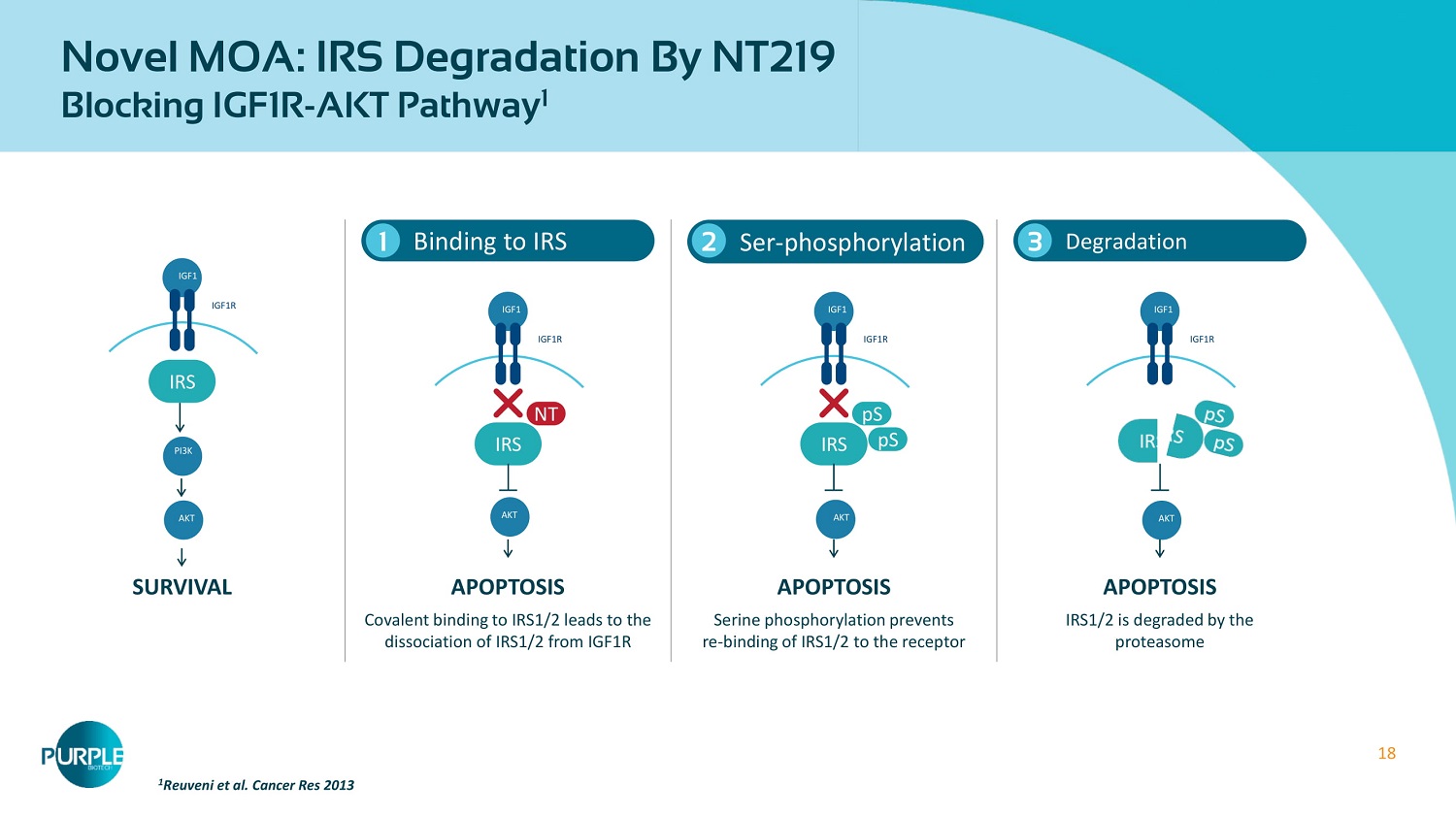
18 Novel MOA: IRS Degradation By NT 219 Blocking IGF 1 R - AKT Pathway 1 Binding to IRS 1 1 Reuveni et al. Cancer Res 2013 Ser - phosphorylation 2 Degradation 3 Covalent binding to IRS 1 / 2 leads to the dissociation of IRS 1 / 2 from IGF 1 R Serine phosphorylation prevents re - binding of IRS 1 / 2 to the receptor SURVIVAL APOPTOSIS APOPTOSIS APOPTOSIS IRS1/2 is degraded by the proteasome IGF 1 IGF 1 R IRS AKT PI 3 K IGF 1 IGF 1 R IRS NT AKT IGF 1 IGF 1 R IRS pS pS AKT IGF 1 IGF 1 R AKT
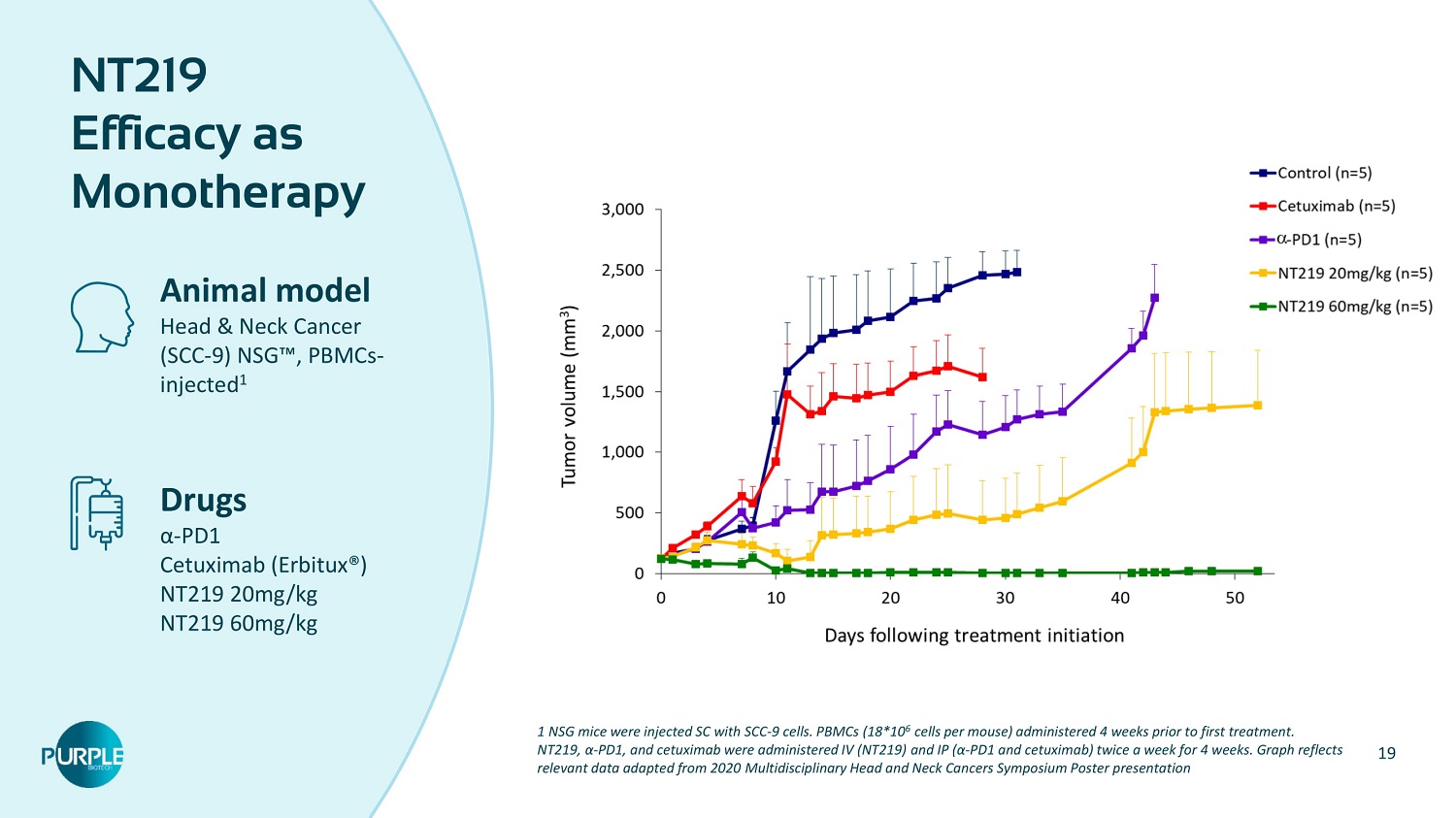
19 NT 219 Efficacy as Monotherapy Animal model Head & Neck Cancer (SCC - 9 ) NSG Ρ , PBMCs - injected 1 Drugs α - PD 1 Cetuximab (Erbitux®) NT 219 20 mg/kg NT 219 60 mg/kg 1 NSG mice were injected SC with SCC - 9 cells. PBMCs ( 18 * 10 6 cells per mouse) administered 4 weeks prior to first treatment. NT 219 , α - PD 1 , and cetuximab were administered IV (NT 219 ) and IP ( α - PD 1 and cetuximab) twice a week for 4 weeks. Graph reflects relevant data adapted from 2020 Multidisciplinary Head and Neck Cancers Symposium Poster presentation

20 By blocking both STAT 3 and IRS pathways, NT 219 prevents tumor resistance and re - sensitizes tumors to anti - cancer therapies STAT 3 and IRS are Essential in Therapeutic Resistance STAT 3 IRS EGFR MEK Tumor Regression ERK PI 3 K AKT Tumor Survival Tumor Survival Buparlisib Ruxolitinib Erlotinib Blocking survival pathways NT 219 NT 219 STAT 3 IRS ONCOPROTEIN DRUG MEK / ERK TUMOR REGRESSION Proof of Concept: PDX model of Head and Neck Cancer Erlotinib+Ruxolitinib (n= 6 ) Erlotinib+Buparlisib (n= 6 ) Control (n= 6 ) Erlotinib (n= 8 ) Erlotinib+NT 219 (n= 6 ) Erlotinib+Ruxolitinib+Buparlisib (n= 8 )

21 NT 219 + Targeted Therapies Established Efficacy in PDX Models NSCLC Exon 19 deletion EGFR and T 790 M, biopsy of bone marrow metastasis, patient previously progressed on afatinib and osimertinib R/M HNSCC metastasis, patient previously progressed on chemoradiation, several chemotherapies, and pembrolizumab Treatments on days 0 , 3 and 10 , cetuximab - 1 mg/mouse, 3 mice/group; PBMCs ( 1.4 M cells/mouse) were injected on day 6 Osimertinib 5 mg/kg, NT 219 65 mg/kg, mean tumor volume at the end point, 3 mice/group; 0 500 1,000 1,500 2,000 Control NT219 Osimertinib Osimertinib+NT219 Tumor Volume (mm 3 ) * ** ** * Tumor Volume (mm 3 ) ** p< 0.01 , * p< 0.02 based on one - way ANOVA with post hoc Tukey ’ s HSD test Control NT 219 Cetuximab Cetuximab+NT 219
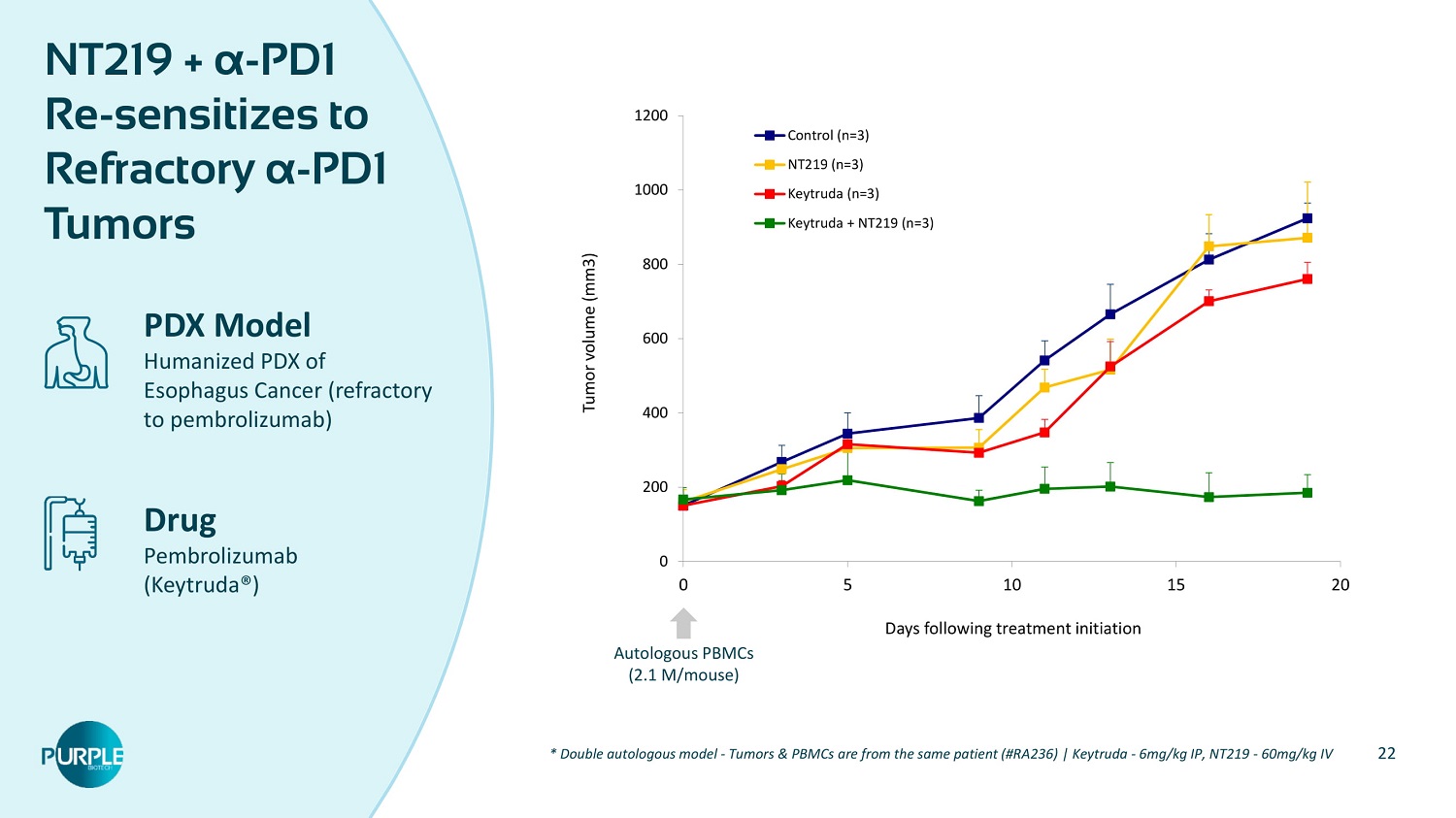
22 NT 219 + α - PD 1 Re - sensitizes to Refractory α - PD 1 Tumors PDX Model Humanized PDX of Esophagus Cancer (refractory to pembrolizumab) Drug Pembrolizumab (Keytruda®) * Double autologous model - Tumors & PBMCs are from the same patient (#RA 236 ) | Keytruda - 6 mg/kg IP, NT 219 - 60 mg/kg IV 0 200 400 600 800 1000 1200 0 5 10 15 20 Tumor volume (mm 3 ) Days following treatment initiation Control (n=3) NT219 (n=3) Keytruda (n=3) Keytruda + NT219 (n=3) Autologous PBMCs ( 2.1 M/mouse)
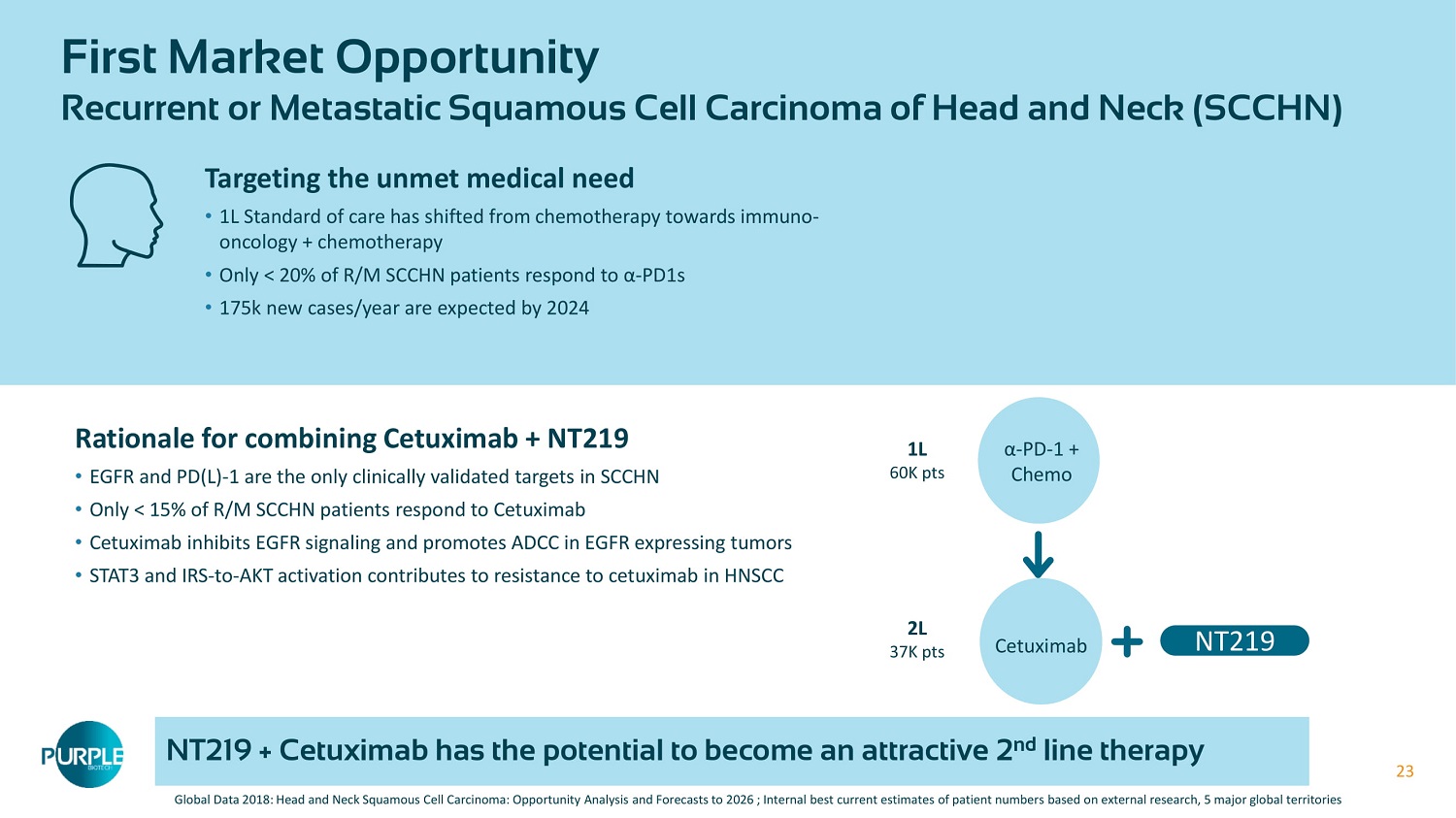
23 23 First Market Opportunity Recurrent or Metastatic Squamous Cell Carcinoma of Head and Neck (SCCHN) Global Data 2018 : Head and Neck Squamous Cell Carcinoma: Opportunity Analysis and Forecasts to 2026 ; Internal best current estimates of patient numbers based on external research, 5 major global territories Rationale for combining Cetuximab + NT 219 • EGFR and PD(L) - 1 are the only clinically validated targets in SCCHN • Only < 15 % of R/M SCCHN patients respond to Cetuximab • Cetuximab inhibits EGFR signaling and promotes ADCC in EGFR expressing tumors • STAT 3 and IRS - to - AKT activation contributes to resistance to cetuximab in HNSCC Targeting the unmet medical need • 1 L Standard of care has shifted from chemotherapy towards immuno - oncology + chemotherapy • Only < 20 % of R/M SCCHN patients respond to α - PD 1 s • 175 k new cases/year are expected by 2024 α - PD - 1 + Chemo 1 L 60 K pts Cetuximab 2 L 37 K pts NT 219 NT 219 + Cetuximab has the potential to become an attractive 2 nd line therapy
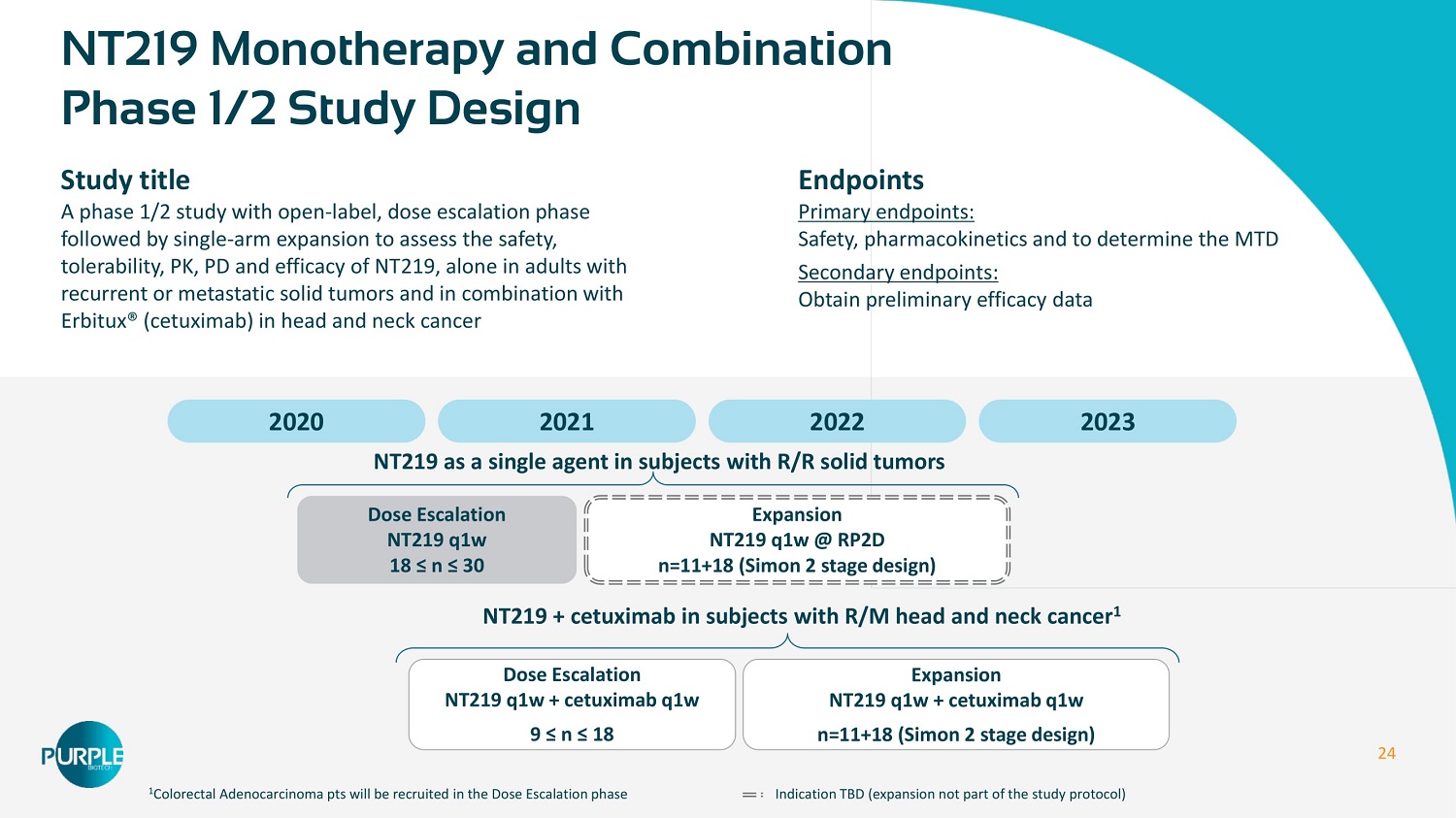
24 2020 2021 2022 2023 Dose Escalation NT 219 q 1 w 18 ≤ n ≤ 30 Expansion NT 219 q 1 w @ RP 2 D n= 11 + 18 (Simon 2 stage design) Dose Escalation NT 219 q 1 w + cetuximab q 1 w 9 ≤ n ≤ 18 NT 219 + cetuximab in subjects with R/M head and neck cancer 1 Expansion NT 219 q 1 w + cetuximab q 1 w n= 11 + 18 (Simon 2 stage design) NT 219 Monotherapy and Combination Phase 1 / 2 Study Design Study title A phase 1 / 2 study with open - label, dose escalation phase followed by single - arm expansion to assess the safety, tolerability, PK, PD and efficacy of NT 219 , alone in adults with recurrent or metastatic solid tumors and in combination with Erbitux® (cetuximab) in head and neck cancer Endpoints Primary endpoints: Safety, pharmacokinetics and to determine the MTD Secondary endpoints: Obtain preliminary efficacy data 1 Colorectal Adenocarcinoma pts will be recruited in the Dose Escalation phase Indication TBD (expansion not part of the study protocol) NT 219 as a single agent in subjects with R/R solid tumors

25 Advancing Clinical - stage Novel Oncology Therapies Strong balance sheet and cash position Purple Biotech (NASDAQ/TASE: PPBT) CM 24 - First - in - class α - CEACAM 1 mAb , Validating clinical collaboration with NT 219 - First - in - class, small molecule, dual inhibitor of IRS 1 / 2 and STAT 3 H 2:21 - Two phase 1 study readouts $ 61 M cash as of December 31 st , 2020 CONSENSI® commercial royalties supports pipeline development Cash runway through 2024 ADSs outstanding: 17.2 M

26 We are committed to providing cancer patients with first - in - class therapies to OVERCOME tumor drug resistance, ENHANCE treatment response and SLOW tumor progression

THANK YOU Contact Us: ir@purple - biotech.com

Appendix A - CM 24
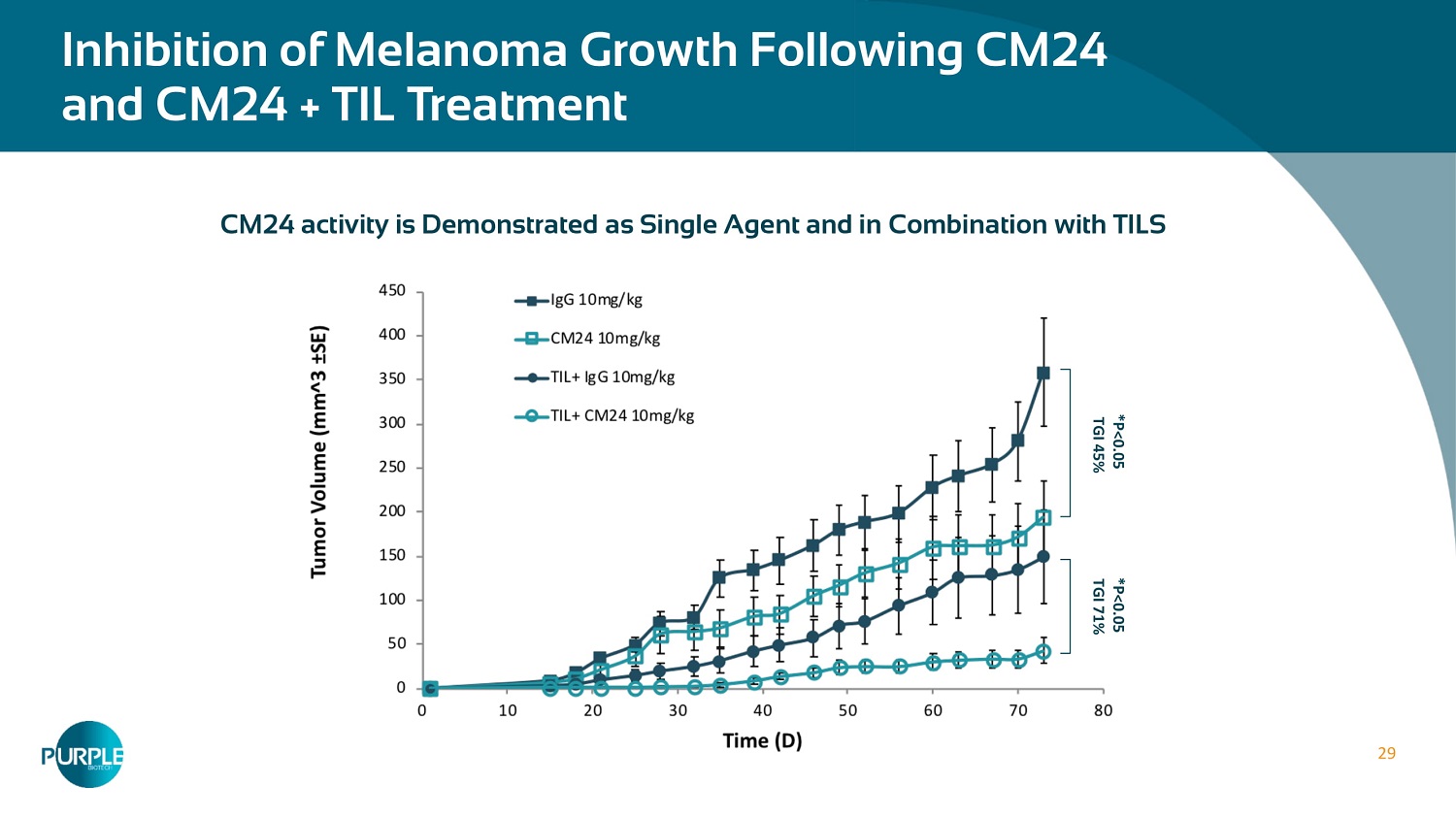
29 CM 24 activity is Demonstrated as Single Agent and in Combination with TILS Inhibition of Melanoma Growth Following CM 24 and CM 24 + TIL Treatment * P< 0.05 TGI 71 % * P< 0.05 TGI 45 %
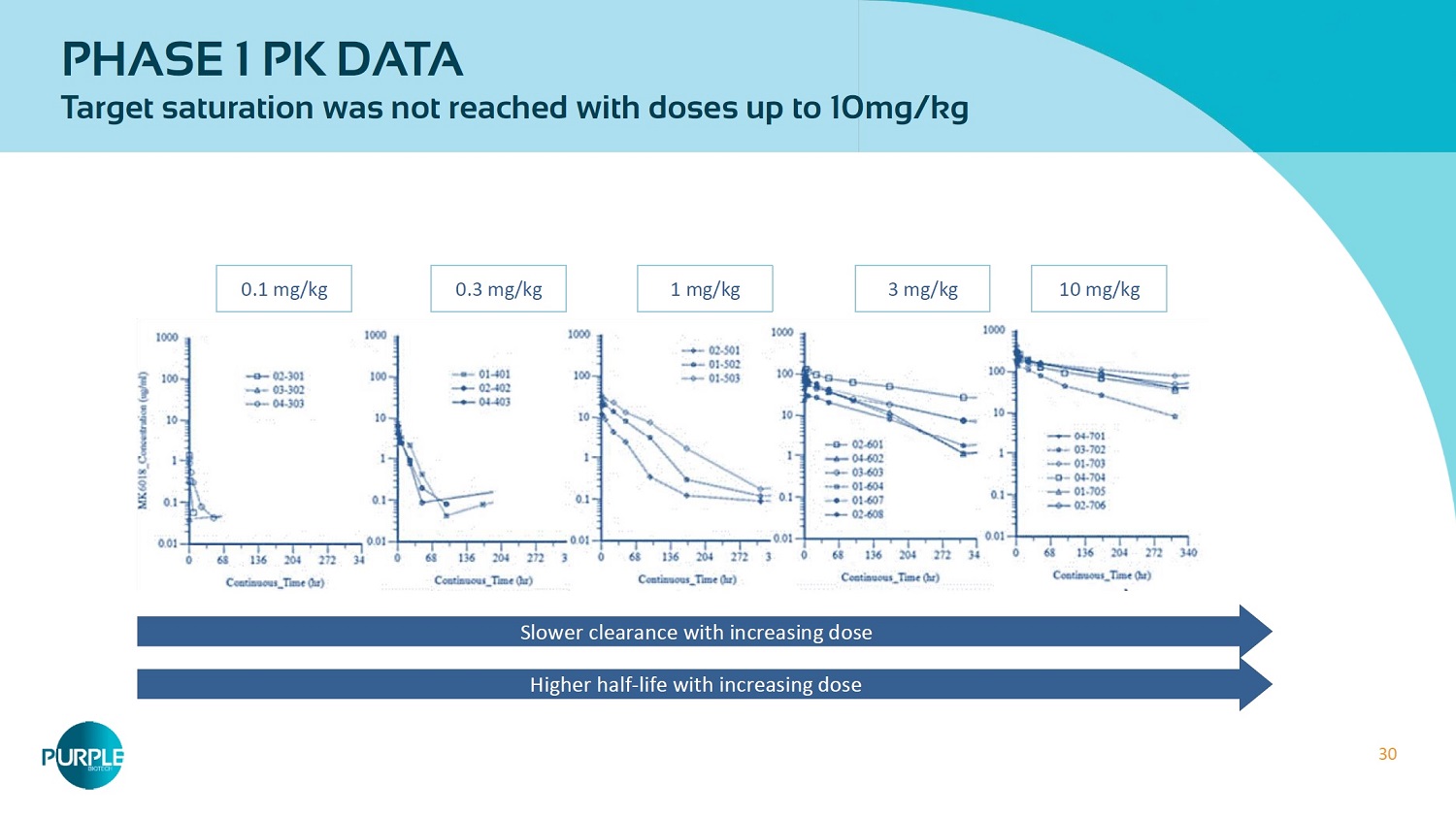
30 PHASE 1 PK DATA Target saturation was not reached with doses up to 10 mg/kg Slower clearance with increasing dose Higher half - life with increasing dose 0.1 mg/ kg 0.3 mg/ kg 1 mg/ kg 3 mg/ kg 10 mg/ kg

Appendix B - NT 219

32 Selected Publications Menashe Bar - Eli Michael Karin Michael Cox Alexander Levitzki

33 Ido Wolf, MD, Head of Oncology Division, Tel Aviv Sourasky Medical Center AACR Virtual Special Conference on Epigenetics and Metabolism, Oct 2020 Colon cancer LS - 513 cells overexpressing IRS 2 demonstrate enhanced b - catenin activity. Targeted inhibition of IRS 2 by NT 219 or IRS 2 - SH RNA, suppresses the increased b - catenin activity and inhibit LS - 513 cell viability. Active β - Catenin IRS 2 β - actin NT 219 + - NT 219 concentrations ( m M) CRC Cell Viability ( 72 hr) IRS 2 NT 219 downregulates IRS 2 and suppresses the b - catenin activity Silencing IRS 2 inhibits b - catenin activity NT 219 | Suppresses β - Catenin activity in CRC Cells
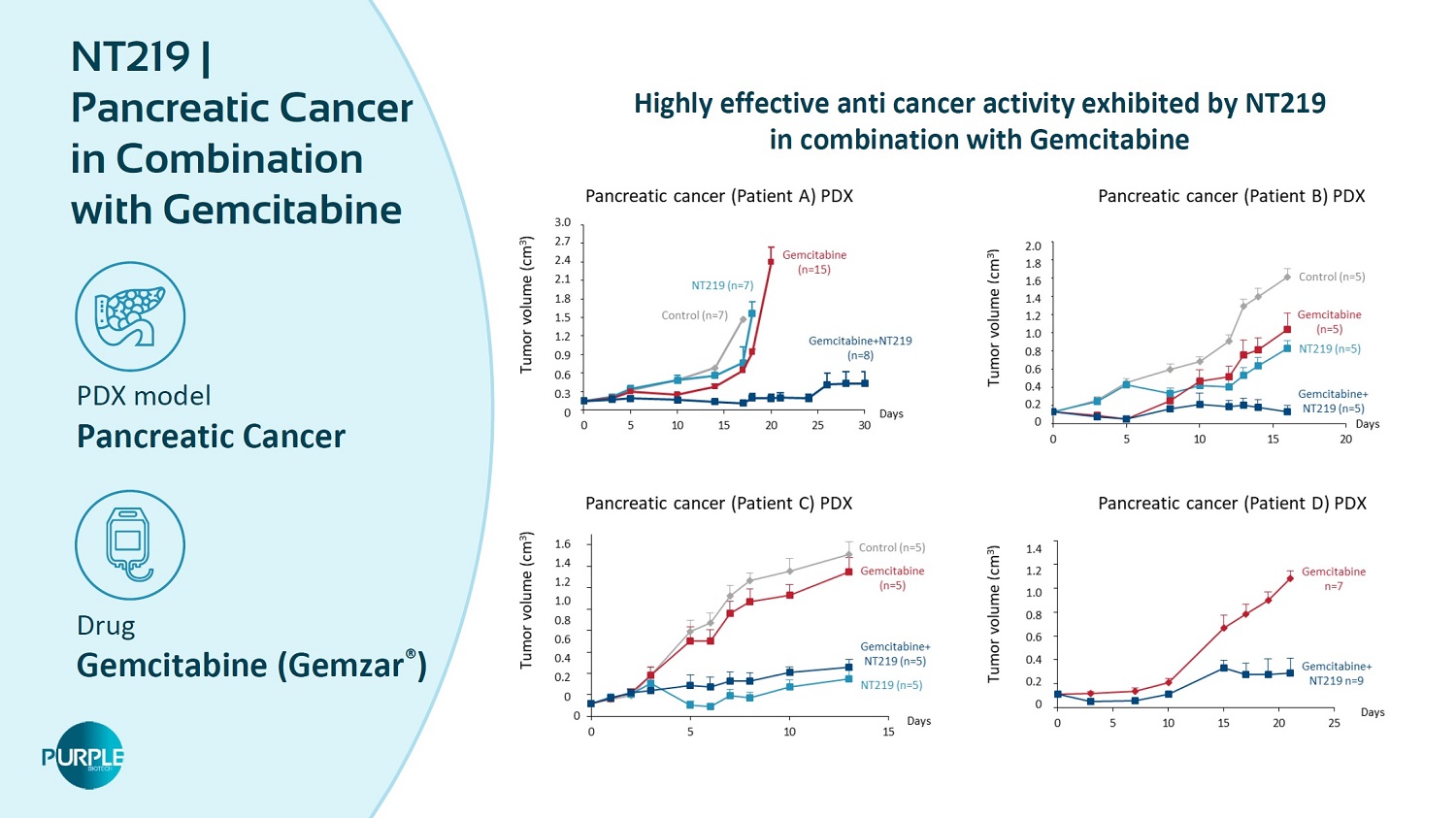
34 PDX model Pancreatic Cancer Drug Gemcitabine ( Gemzar ® ) NT 219 | Pancreatic Cancer in Combination with Gemcitabine Highly effective anti cancer activity exhibited by NT 219 in combination with Gemcitabine
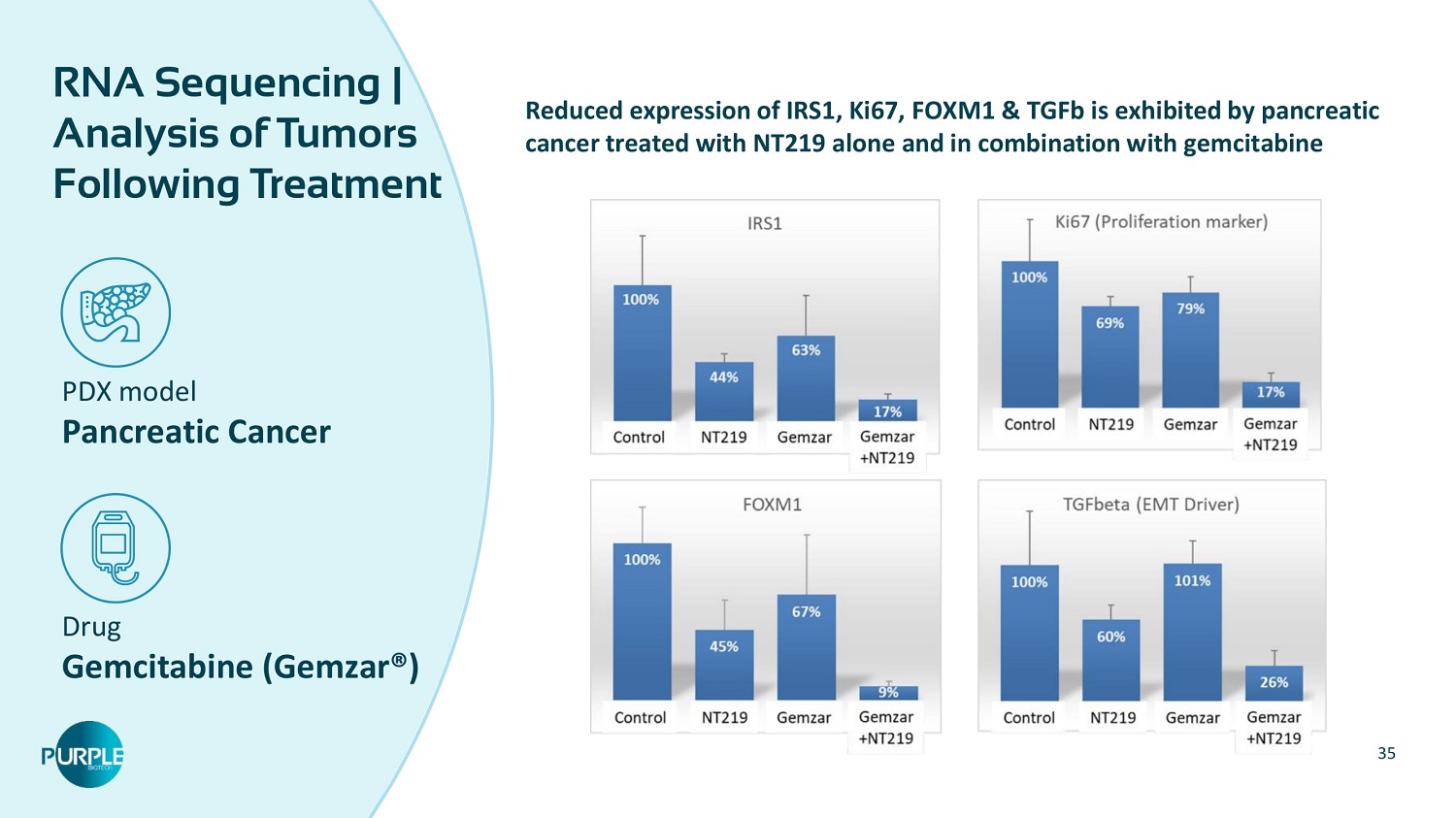
35 PDX model Pancreatic Cancer Drug Gemcitabine ( Gemzar ®) RNA Sequencing | Analysis of Tumors Following Treatment Reduced expression of IRS 1 , Ki 67 , FOXM 1 & TGFb is exhibited by pancreatic cancer treated with NT 219 alone and in combination with gemcitabine

Appendix C - CONSENSI®

37 CONSENSI® | From IND to the U.S. Market CONSENSI ® is the only NSAID whose labeling indicates reduction of blood pressure and consequent risk reduction of heart attack, stroke and death Full U.S. Prescribing Information is available at http://www.consensi.com Celecoxib , a COX - 2 selective NSAID (the active ingredient in Pfizer ’ s Celebrex®) + Amlodipine a blood pressure - lowering agent (a calcium channel blocker) (the active ingredient in Pfizer's Norvasc®) Fixed dose combination of Purple Biotech ’ s clinical, regulatory and medical teams developed CONSENSI® internally from IND, through successful Phase III clinical trials, to FDA approval Launched in the USA - Coeptis Pharmaceuticals Partnered in China - CSBio Partnered in S. Korea - Kuhnil Pharmaceutical
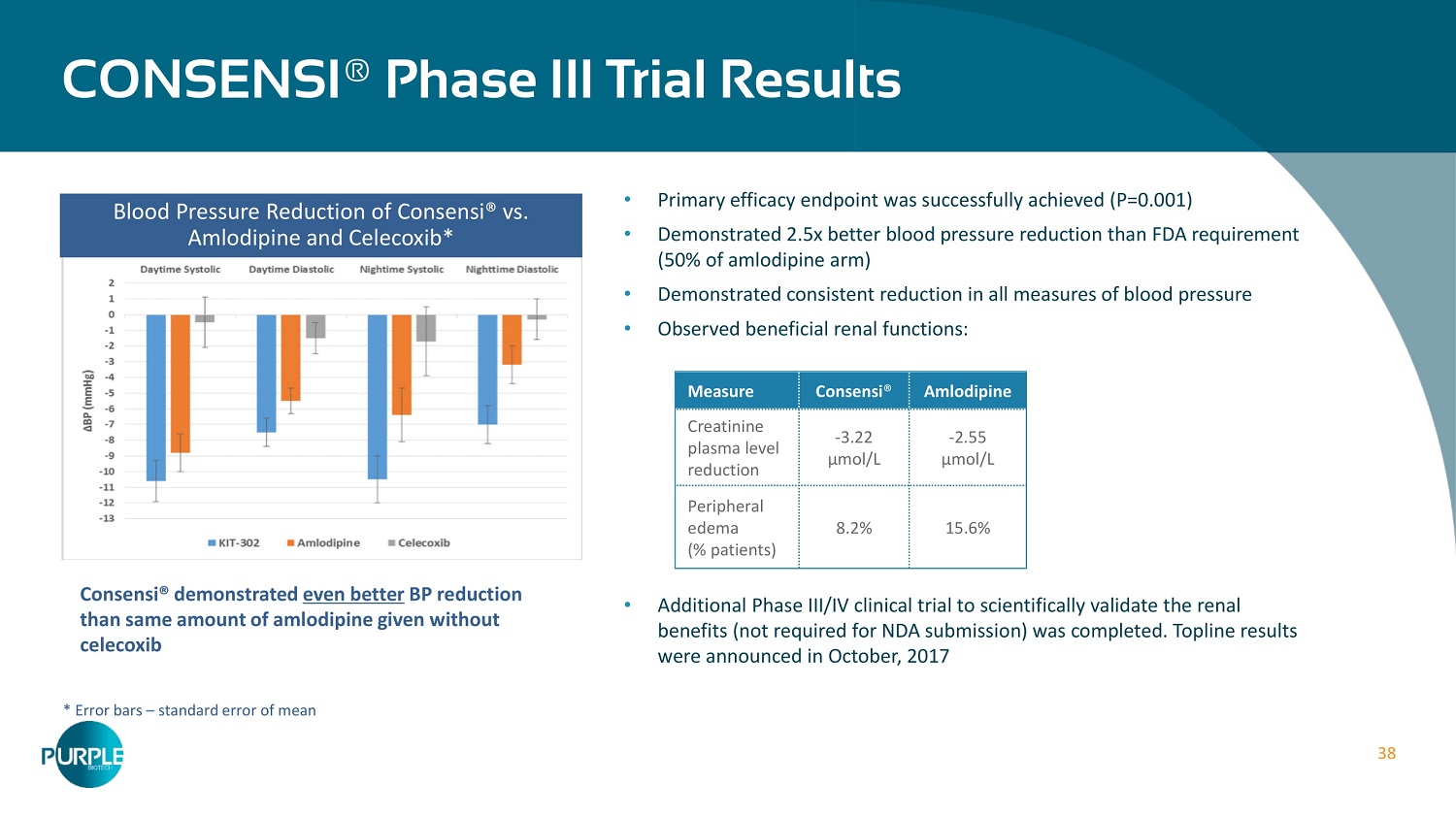
38 • Primary efficacy endpoint was successfully achieved (P= 0.001 ) • Demonstrated 2.5 x better blood pressure reduction than FDA requirement ( 50 % of amlodipine arm) • Demonstrated consistent reduction in all measures of blood pressure • Observed beneficial renal functions: • Additional Phase III/IV clinical trial to scientifically validate the renal benefits (not required for NDA submission) was completed. Topline results were announced in October, 2017 CONSENSI ® Phase III Trial Results * Error bars – standard error of mean Blood Pressure Reduction of Consensi® vs. Amlodipine and Celecoxib* Consensi ® demonstrated even better BP reduction than same amount of amlodipine given without celecoxib Measure Consensi ® Amlodipine Creatinine plasma level reduction - 3.22 μ mol/L - 2.55 μ mol/L Peripheral edema (% patients) 8.2% 15.6%

39 Purple Biotech Commercial Drug: CONSENSI® Simultaneous treatment of osteoarthritic pain and hypertension Fixed dose combination of Celecoxib , a COX - 2 selective NSAID (the active ingredient in Pfizer ’ s Celebrex®) + Amlodipine, a blood pressure - lowering agent (a calcium channel blocker) (the active ingredient in Pfizer's Norvasc®) *Celebrex® is a registered trademark of G.D. Searle LLC (a subsidiary of Pfizer Inc.). Norvasc® is a registered trademark of Pfizer Inc. CONSENSI® is the only NSAID whose labeling indicates reduction of blood pressure and consequent risk reduction of heart attack, stroke and death Full U.S. Prescribing Information is available at: : http://www.consensi.com • Approved for marketing by U.S. FDA on May 31 , 2018 • Clinical data showed Consensi Ρ was more effective at lowering blood pressure than amlodipine alone • Clinical data also demonstrated beneficial renal function measures • Formulated with 200 mg celecoxib and three different dosages ( 2.5 , 5 , 10 mg) of amlodipine • Manufactured by Dexcel Pharma – Israel ’ s largest private pharmaceutical company
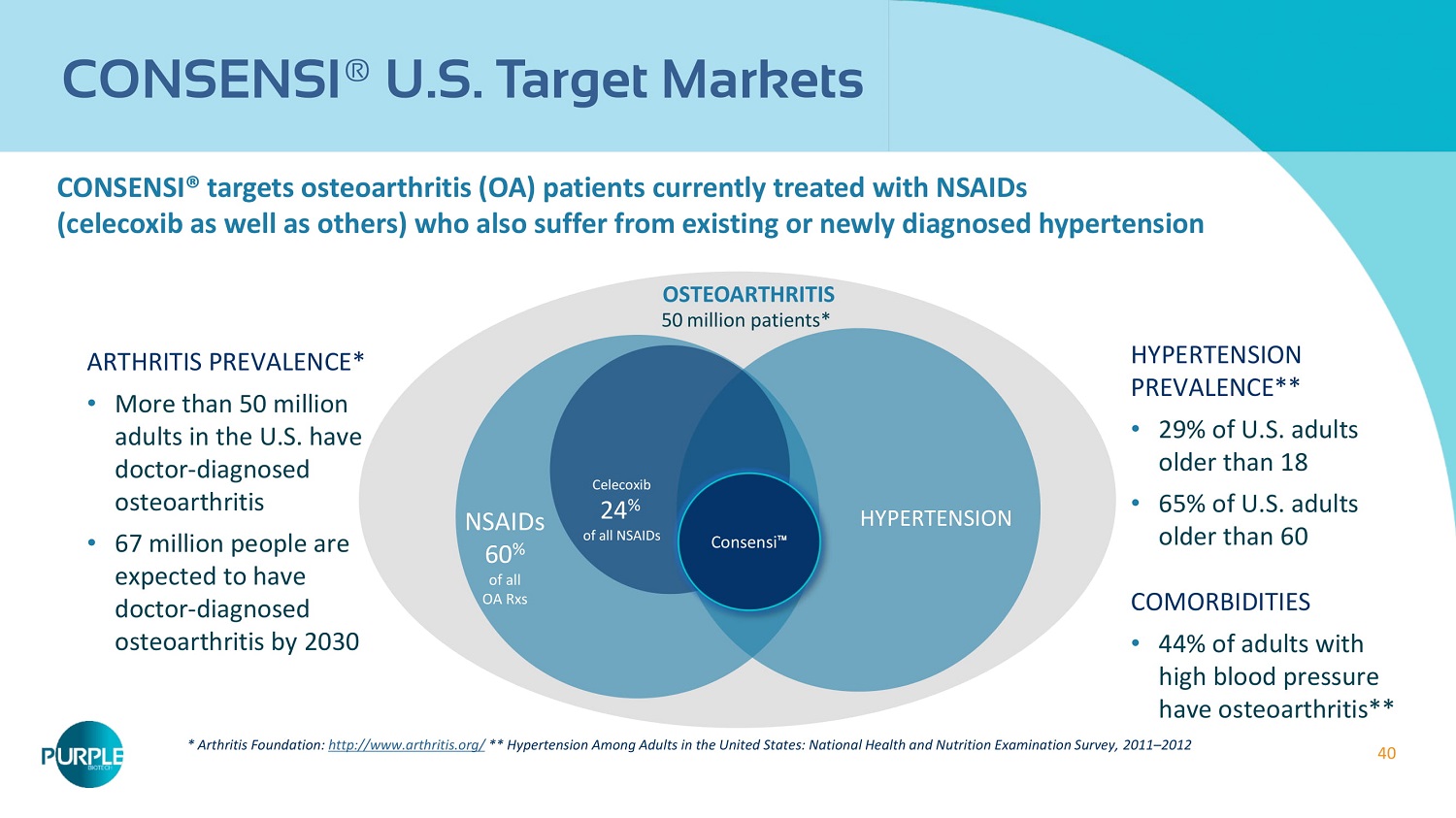
40 CONSENSI ® U.S. Target Markets CONSENSI® targets osteoarthritis (OA) patients currently treated with NSAIDs (celecoxib as well as others) who also suffer from existing or newly diagnosed hypertension OSTEOARTHRITIS 50 million patients* NSAIDs 60 % of all OA Rxs * Arthritis Foundation: http://www.arthritis.org/ ** Hypertension Among Adults in the United States: National Health and Nutrition Examination Survey, 2011 – 2012 ARTHRITIS PREVALENCE* • More than 50 million adults in the U.S. have doctor - diagnosed osteoarthritis • 67 million people are expected to have doctor - diagnosed osteoarthritis by 2030 HYPERTENSION PREVALENCE** • 29 % of U.S. adults older than 18 • 65 % of U.S. adults older than 60 COMORBIDITIES • 44 % of adults with high blood pressure have osteoarthritis** HYPERTENSION Celecoxib 24 % of all NSAIDs Consensi 䉼