SQZ-PBMC-HPV-101 Clinical Program Updates at ESMO Immuno-Oncology Congress 2021 Conference Call and Webcast December 9th, 2021

Forward Looking Statements and Legal Disclaimers This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this presentation that do not relate to matters of historical fact should be considered forward-looking statements, including statements relating to our platform development, our product candidates, our preclinical and clinical activities, progress and outcomes, development plans, manufacturing, clinical safety and efficacy, therapeutic impact, and market opportunities. These forward-looking statements are based on management's current expectations. The words ”may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “estimate,” “believe,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: risks and uncertainties related to our limited operating history; our significant losses incurred since inception and expectation to incur significant additional losses for the foreseeable future; the development of our initial product candidates, upon which our business is highly dependent; the impact of the COVID-19 pandemic on our operations and clinical activities; our need for additional funding and our cash runway; the lengthy, expensive, and uncertain process of clinical drug development, including uncertain outcomes of clinical trials and potential delays in regulatory approval; our ability to maintain our relationships with our third party vendors and strategic partners; and protection of our proprietary technology, intellectual property portfolio and the confidentiality of our trade secrets. These and other important factors discussed in our filings with the US Securities and Exchange Commission could cause actual results to differ materially from those indicated by the forward-looking statements made in this presentation. Any such forward-looking statements represent management's estimates as of the date of this presentation. New risk factors and uncertainties may emerge from time to time, and it is not possible to predict all risk factors and uncertainties. While we may elect to update such forward-looking statements at some point in the future, except as required by law, we disclaim any obligation to do so, even if subsequent events cause our views to change. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this presentation. Certain information contained in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and our own internal estimates and research. While we believe these third-party sources to be reliable as of the date of this presentation, we have not independently verified, and we make no representation as to the adequacy, fairness, accuracy or completeness of any information obtained from third-party sources. In addition, all of the market data included in this presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Neither we nor our affiliates, advisors or representatives makes any representation as to the accuracy or completeness of the data included in this presentation or undertake to update such data after the date of this presentation. Finally, while we believe our own internal research is reliable, such research has not been verified by any independent source.

Welcome to the SQZ Conference Call and Webcast SQZ-PBMC-HPV-101 Clinical Program Updates at ESMO Immuno-Oncology Congress 2021

Agenda Welcome Micah Zajic, Chief Business Officer SQZ Oncology Portfolio Armon Sharei, PhD, Chief Executive Officer and Founder SQZ-PBMC-HPV-101 Phase 1/2 Trial Highest-Dose Cohort Findings �Jong Chul Park, MD, Massachusetts General Hospital Monotherapy Summary & Clinical Development �Ricardo Zwirtes, MD, Oncology Clinical Development Combination Strategies and Manufacturing �Howard Bernstein, MD, PhD, Chief Scientific Officer Summary & 2022 Oncology Portfolio Outlook Armon Sharei, PhD, Chief Executive Officer and Founder Questions & Answers Management Team Members

Agenda Welcome Micah Zajic, Chief Business Officer SQZ Oncology Portfolio Armon Sharei, PhD, Chief Executive Officer and Founder SQZ-PBMC-HPV-101 Phase 1/2 Trial Highest-Dose Cohort Findings �Jong Chul Park, MD, Massachusetts General Hospital Monotherapy Summary & Clinical Development �Ricardo Zwirtes, MD, Oncology Clinical Development Combination Strategies and Manufacturing �Howard Bernstein, MD, PhD, Chief Scientific Officer Summary & 2022 Oncology Portfolio Outlook Armon Sharei, PhD, Chief Executive Officer and Founder Questions & Answers Management Team Members

An exciting milestone for the SQZTM APC approach in solid tumors Biomarkers support expected APC mechanism Clinical and immune response in heavily treated patient Motivates program expansion with systemic combos and eAPCs Manufacturing data highlights rapid production Additional clinical data readouts and INDs expected in 2022
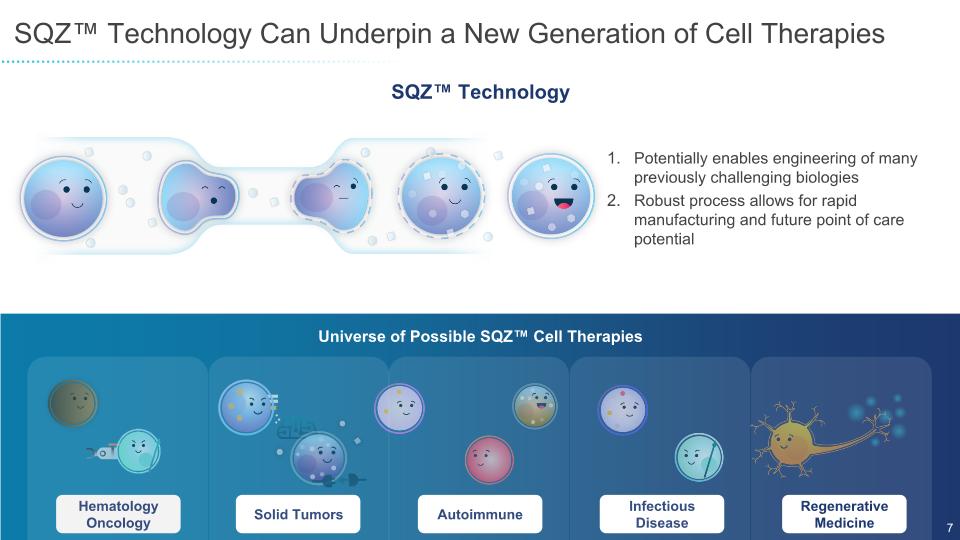
SQZ™ Technology Can Underpin a New Generation of Cell Therapies Universe of Possible SQZ™ Cell Therapies Hematology Oncology Solid Tumors Autoimmune Infectious Disease Regenerative Medicine SQZ™ Technology Potentially enables engineering of many previously challenging biologies Robust process allows for rapid manufacturing and future point of care potential
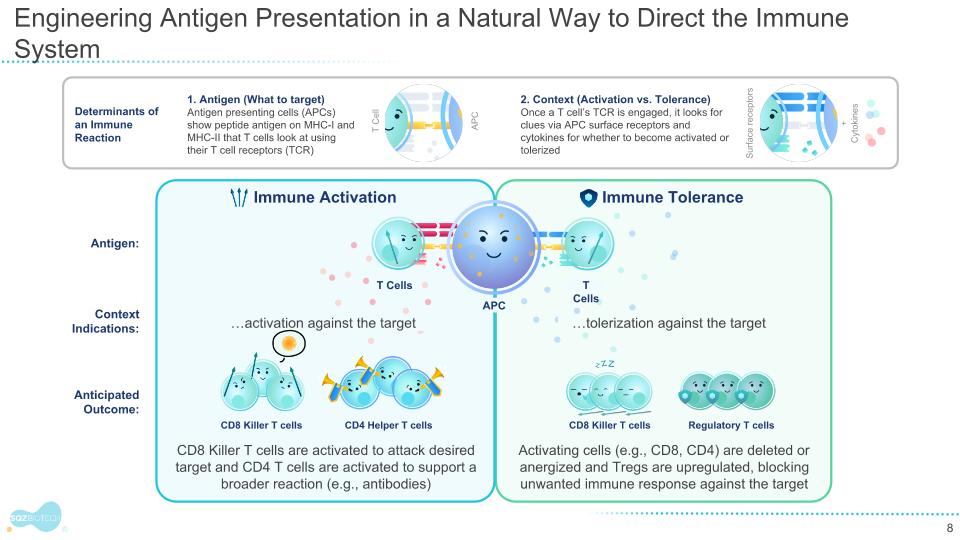
Engineering Antigen Presentation in a Natural Way to Direct the Immune System 1. Antigen (What to target) Antigen presenting cells (APCs) show peptide antigen on MHC-I and MHC-II that T cells look at using their T cell receptors (TCR) 2. Context (Activation vs. Tolerance) Once a T cell’s TCR is engaged, it looks for clues via APC surface receptors and cytokines for whether to become activated or tolerized Determinants of an Immune Reaction APC T Cell Cytokines Surface receptors + Context Indications: Activating cells (e.g., CD8, CD4) are deleted or anergized and Tregs are upregulated, blocking unwanted immune response against the target CD8 Killer T cells are activated to attack desired target and CD4 T cells are activated to support a broader reaction (e.g., antibodies) Anticipated Outcome: Antigen: Immune Tolerance Immune Activation CD8 Killer T cells CD8 Killer T cells CD4 Helper T cells Regulatory T cells T Cells APC …tolerization against the target …activation against the target T Cells
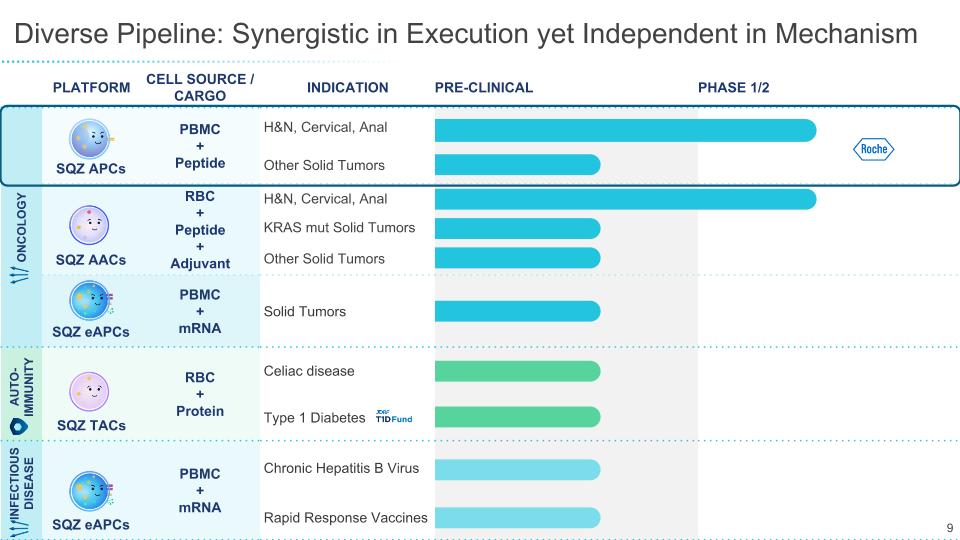
Diverse Pipeline: Synergistic in Execution yet Independent in Mechanism 9 PLATFORM CELL SOURCE / CARGO INDICATION PRE-CLINICAL PHASE 1/2 ONCOLOGY SQZ APCs PBMC + Peptide H&N, Cervical, Anal Other Solid Tumors SQZ AACs RBC �+ Peptide + Adjuvant H&N, Cervical, Anal KRAS mut Solid Tumors Other Solid Tumors SQZ eAPCs PBMC + mRNA Solid Tumors AUTO-IMMUNITY SQZ TACs RBC + Protein Celiac disease Type 1 Diabetes INFECTIOUS DISEASE SQZ eAPCs PBMC + mRNA Chronic Hepatitis B Virus Rapid Response Vaccines 9
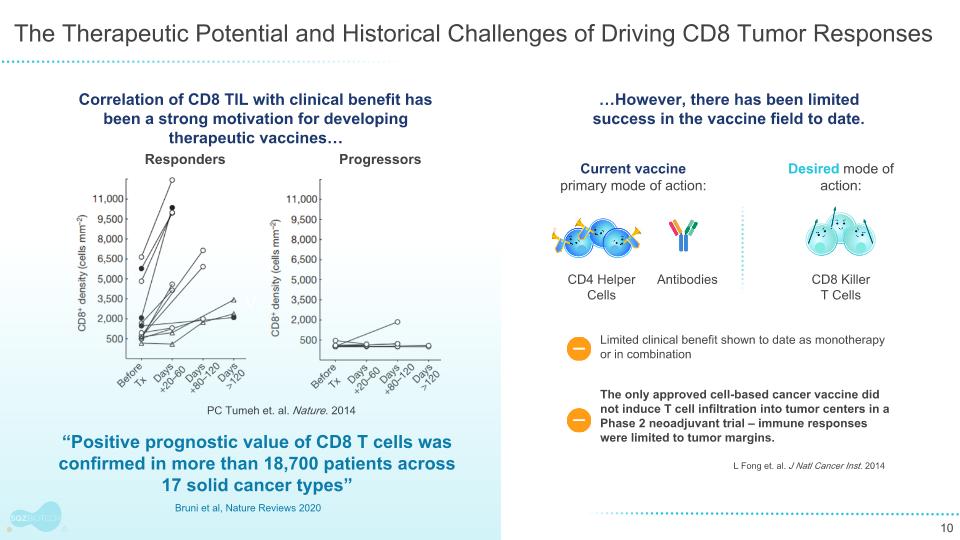
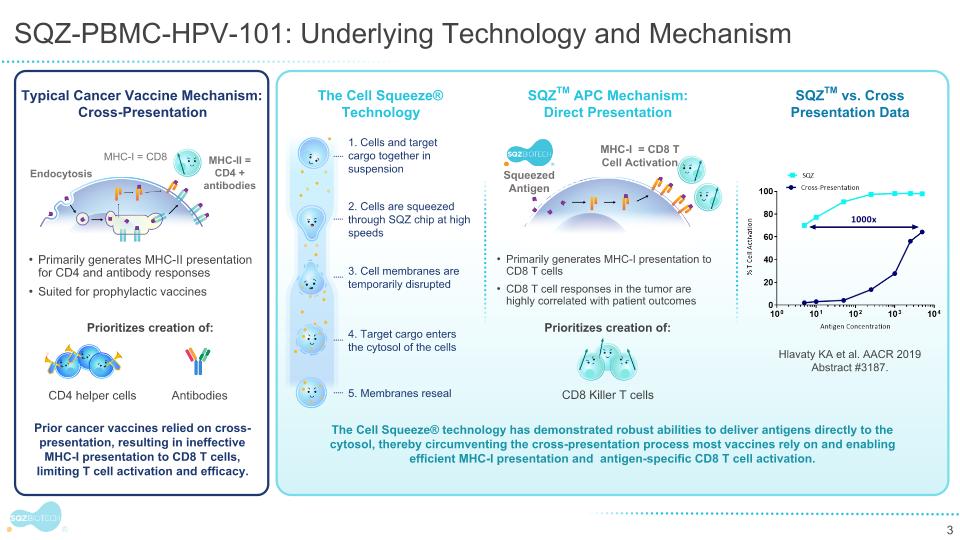
The Therapeutic Potential and Historical Challenges of Driving CD8 Tumor Responses Responders Progressors …However, there has been limited success in the vaccine field to date. The only approved cell-based cancer vaccine did not induce T cell infiltration into tumor centers in a Phase 2 neoadjuvant trial – immune responses were limited to tumor margins. L Fong et. al. J Natl Cancer Inst. 2014 Limited clinical benefit shown to date as monotherapy or in combination V “Positive prognostic value of CD8 T cells was confirmed in more than 18,700 patients across 17 solid cancer types” Bruni et al, Nature Reviews 2020 PC Tumeh et. al. Nature. 2014 Correlation of CD8 TIL with clinical benefit has been a strong motivation for developing therapeutic vaccines… Current vaccine primary mode of action: Desired mode of action: CD8 Killer T Cells Antibodies CD4 Helper Cells
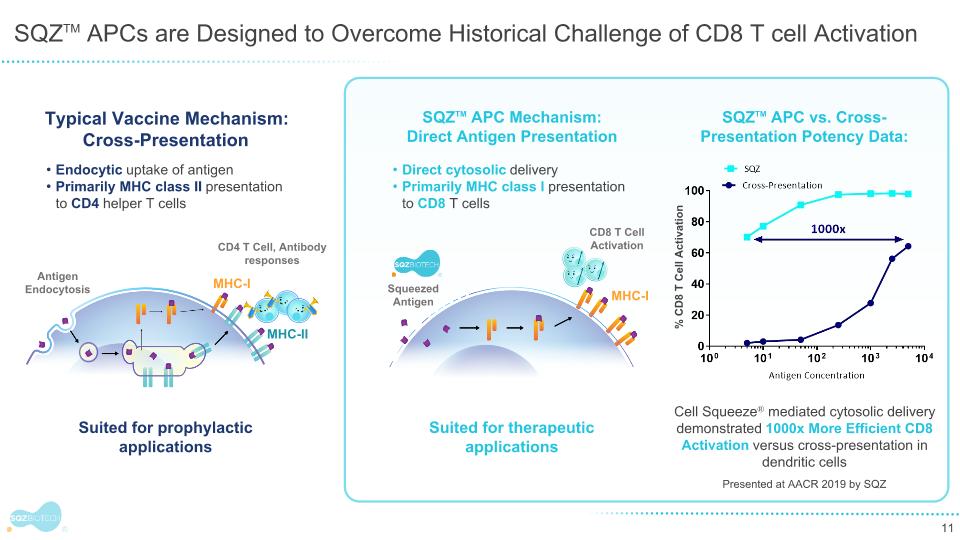
SQZTM APCs are Designed to Overcome Historical Challenge of CD8 T cell Activation Squeezed Antigen SQZTM APC vs. Cross-Presentation Potency Data: Cell Squeeze® mediated cytosolic delivery demonstrated 1000x More Efficient CD8 Activation versus cross-presentation in dendritic cells % CD8 T Cell Activation Typical Vaccine Mechanism: Cross-Presentation CD4 T Cell, Antibody responses Antigen Endocytosis MHC-I MHC-II Endocytic uptake of antigen Primarily MHC class II presentation to CD4 helper T cells SQZTM APC Mechanism: Direct Antigen Presentation MHC-I CD8 T Cell Activation Suited for prophylactic applications Direct cytosolic delivery Primarily MHC class I presentation to CD8 T cells Suited for therapeutic applications Presented at AACR 2019 by SQZ
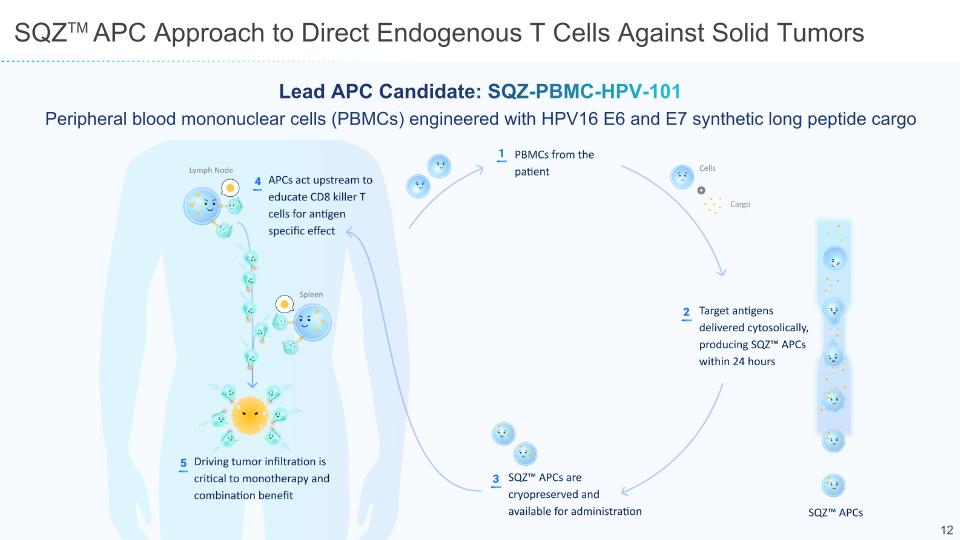
SQZTM APC Approach to Direct Endogenous T Cells Against Solid Tumors Lead APC Candidate: SQZ-PBMC-HPV-101 Peripheral blood mononuclear cells (PBMCs) engineered with HPV16 E6 and E7 synthetic long peptide cargo

Agenda Welcome Micah Zajic, Chief Business Officer SQZ Oncology Portfolio Armon Sharei, PhD, Chief Executive Officer and Founder SQZ-PBMC-HPV-101 Phase 1/2 Trial Highest-Dose Cohort Findings in Advanced Solid Tumors�Jong Chul Park, MD, Massachusetts General Hospital Monotherapy Summary & Clinical Development �Ricardo Zwirtes, MD, Oncology Clinical Development Combination Strategies and Manufacturing �Howard Bernstein, MD, PhD, Chief Scientific Officer Summary & 2022 Oncology Portfolio Outlook Armon Sharei, PhD, Chief Executive Officer and Founder Questions & Answers Management Team Members
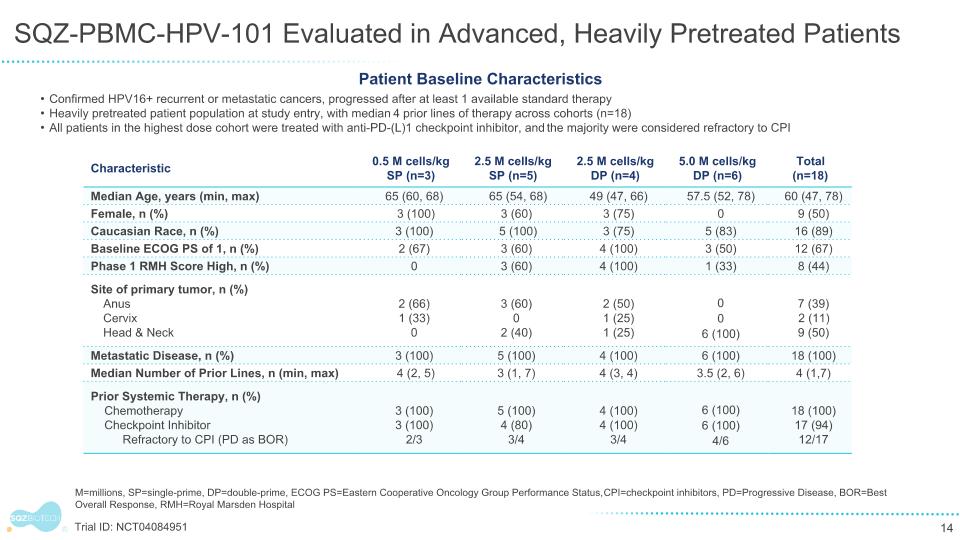
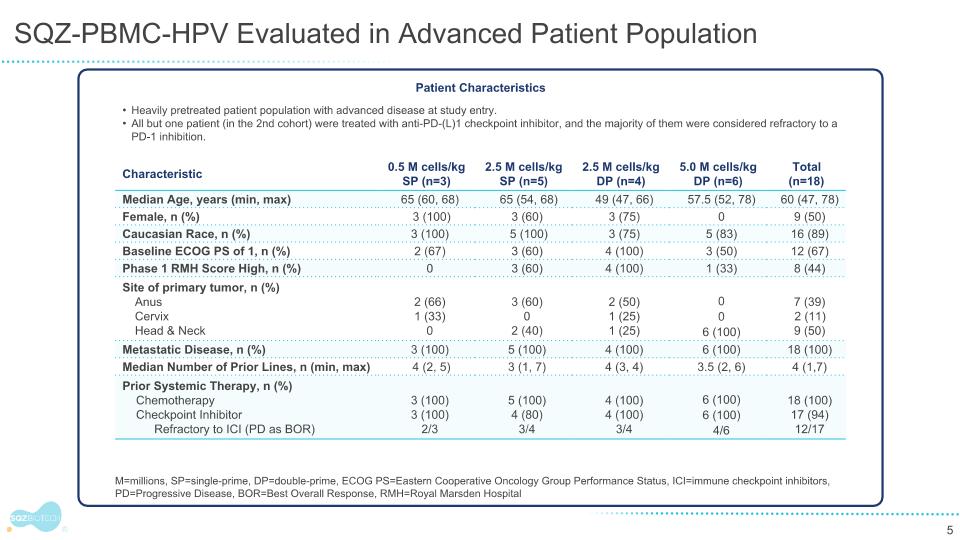
SQZ-PBMC-HPV-101 Evaluated in Advanced, Heavily Pretreated Patients Confirmed HPV16+ recurrent or metastatic cancers, progressed after at least 1 available standard therapy Heavily pretreated patient population at study entry, with median 4 prior lines of therapy across cohorts (n=18) All patients in the highest dose cohort were treated with anti-PD-(L)1 checkpoint inhibitor, and the majority were considered refractory to CPI Patient Baseline Characteristics M=millions, SP=single-prime, DP=double-prime, ECOG PS=Eastern Cooperative Oncology Group Performance Status, CPI=checkpoint inhibitors, PD=Progressive Disease, BOR=Best Overall Response, RMH=Royal Marsden Hospital Trial ID: NCT04084951 Characteristic 0.5 M cells/kg SP (n=3) 2.5 M cells/kg SP (n=5) 2.5 M cells/kg DP (n=4) 5.0 M cells/kg DP (n=6) Total (n=18) Median Age, years (min, max) 65 (60, 68) 65 (54, 68) 49 (47, 66) 57.5 (52, 78) 60 (47, 78) Female, n (%) 3 (100) 3 (60) 3 (75) 0 9 (50) Caucasian Race, n (%) 3 (100) 5 (100) 3 (75) 5 (83) 16 (89) Baseline ECOG PS of 1, n (%) 2 (67) 3 (60) 4 (100) 3 (50) 12 (67) Phase 1 RMH Score High, n (%) 0 3 (60) 4 (100) 1 (33) 8 (44) Site of primary tumor, n (%) Anus Cervix Head & Neck 2 (66) 1 (33) 0 3 (60) 0 2 (40) 2 (50) 1 (25) 1 (25) 0 0 6 (100) 7 (39) 2 (11) 9 (50) Metastatic Disease, n (%) 3 (100) 5 (100) 4 (100) 6 (100) 18 (100) Median Number of Prior Lines, n (min, max) 4 (2, 5) 3 (1, 7) 4 (3, 4) 3.5 (2, 6) 4 (1,7) Prior Systemic Therapy, n (%) Chemotherapy Checkpoint Inhibitor Refractory to CPI (PD as BOR) 3 (100) 3 (100) 2/3 5 (100) 4 (80) 3/4 4 (100) 4 (100) 3/4 6 (100) 6 (100) 4/6 18 (100) 17 (94) 12/17
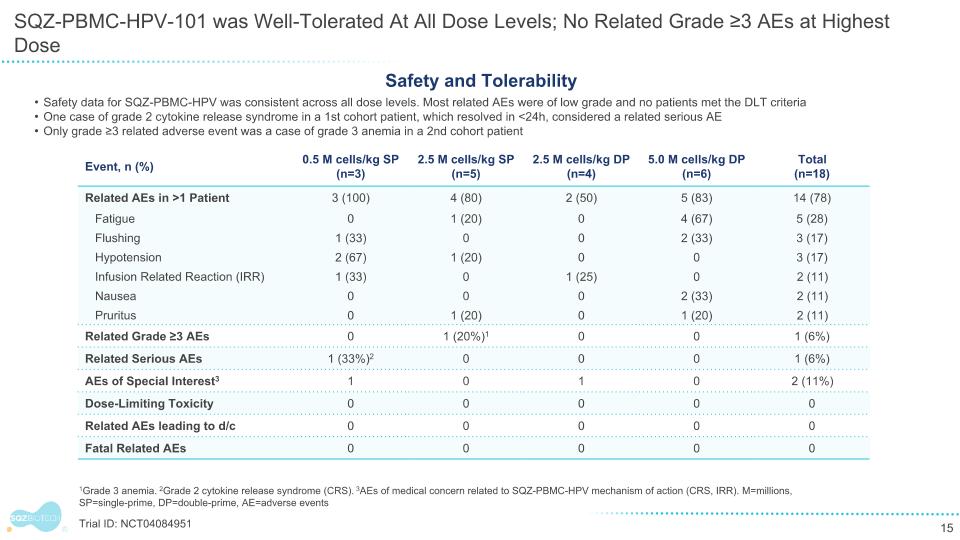
SQZ-PBMC-HPV-101 was Well-Tolerated At All Dose Levels; No Related Grade ≥3 AEs at Highest Dose 1Grade 3 anemia. 2Grade 2 cytokine release syndrome (CRS). 3AEs of medical concern related to SQZ-PBMC-HPV mechanism of action (CRS, IRR). M=millions, SP=single-prime, DP=double-prime, AE=adverse events Safety and Tolerability Safety data for SQZ-PBMC-HPV was consistent across all dose levels. Most related AEs were of low grade and no patients met the DLT criteria One case of grade 2 cytokine release syndrome in a 1st cohort patient, which resolved in <24h, considered a related serious AE Only grade ≥3 related adverse event was a case of grade 3 anemia in a 2nd cohort patient Trial ID: NCT04084951 Event, n (%) 0.5 M cells/kg SP (n=3) 2.5 M cells/kg SP (n=5) 2.5 M cells/kg DP (n=4) 5.0 M cells/kg DP (n=6) Total (n=18) Related AEs in >1 Patient 3 (100) 4 (80) 2 (50) 5 (83) 14 (78) Fatigue 0 1 (20) 0 4 (67) 5 (28) Flushing 1 (33) 0 0 2 (33) 3 (17) Hypotension 2 (67) 1 (20) 0 0 3 (17) Infusion Related Reaction (IRR) 1 (33) 0 1 (25) 0 2 (11) Nausea 0 0 0 2 (33) 2 (11) Pruritus 0 1 (20) 0 1 (20) 2 (11) Related Grade ≥3 AEs 0 1 (20%)1 0 0 1 (6%) Related Serious AEs 1 (33%)2 0 0 0 1 (6%) AEs of Special Interest3 1 0 1 0 2 (11%) Dose-Limiting Toxicity 0 0 0 0 0 Related AEs leading to d/c 0 0 0 0 0 Fatal Related AEs 0 0 0 0 0
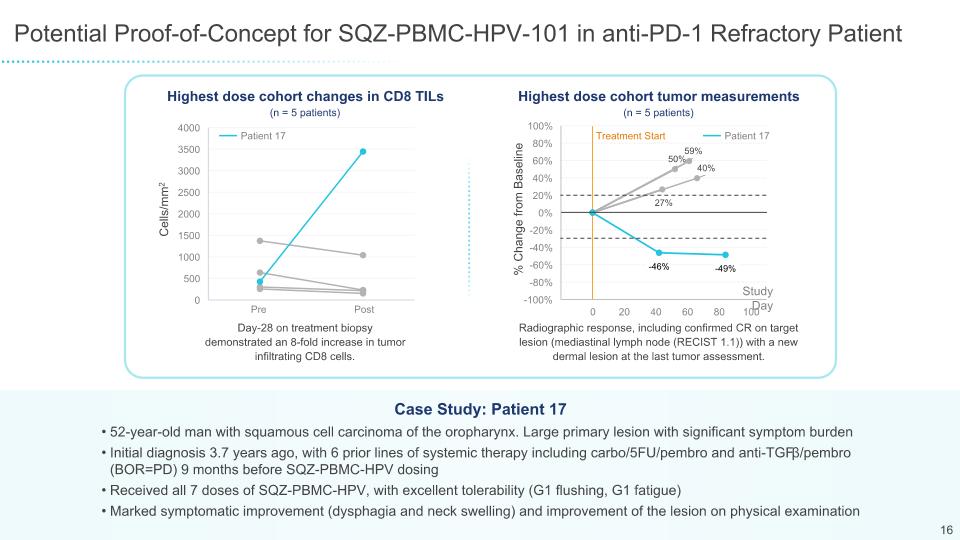
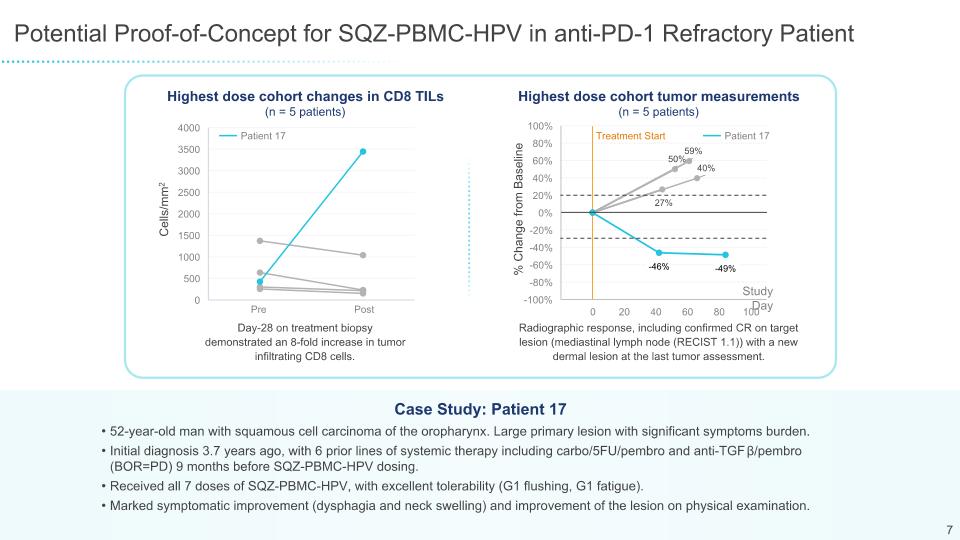
Potential Proof-of-Concept for SQZ-PBMC-HPV-101 in anti-PD-1 Refractory Patient 52-year-old man with squamous cell carcinoma of the oropharynx. Large primary lesion with significant symptom burden Initial diagnosis 3.7 years ago, with 6 prior lines of systemic therapy including carbo/5FU/pembro and anti-TGFβ/pembro (BOR=PD) 9 months before SQZ-PBMC-HPV dosing Received all 7 doses of SQZ-PBMC-HPV, with excellent tolerability (G1 flushing, G1 fatigue) Marked symptomatic improvement (dysphagia and neck swelling) and improvement of the lesion on physical examination Case Study: Patient 17 % Change from Baseline Study Day Highest dose cohort changes in CD8 TILs Highest dose cohort tumor measurements Cells/mm2 Patient 17 Post Pre Day-28 on treatment biopsy demonstrated an 8-fold increase in tumor infiltrating CD8 cells. Radiographic response, including confirmed CR on target lesion (mediastinal lymph node (RECIST 1.1)) with a new dermal lesion at the last tumor assessment. 16 Patient 17 Treatment Start (n = 5 patients) (n = 5 patients)
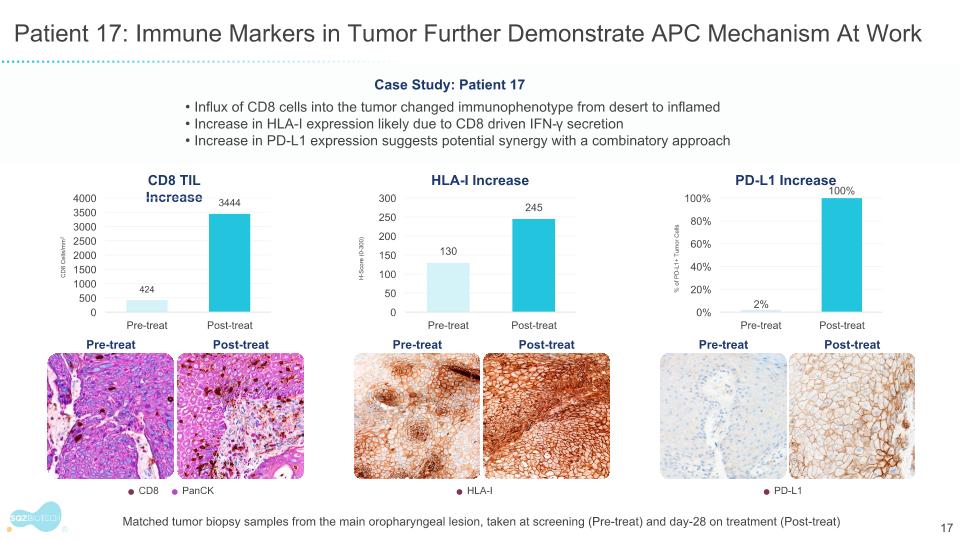
Patient 17: Immune Markers in Tumor Further Demonstrate APC Mechanism At Work Pre-treat Post-treat CD8 TIL Increase CD8 PanCK CD8 Cells/mm2 HLA-I Increase HLA-I H-Score (0-300) PD-L1 Increase PD-L1 % of PD-L1+ Tumor Cells Pre-treat Post-treat Pre-treat Post-treat Case Study: Patient 17 Influx of CD8 cells into the tumor changed immunophenotype from desert to inflamed Increase in HLA-I expression likely due to CD8 driven IFN-γ secretion Increase in PD-L1 expression suggests potential synergy with a combinatory approach Matched tumor biopsy samples from the main oropharyngeal lesion, taken at screening (Pre-treat) and day-28 on treatment (Post-treat)
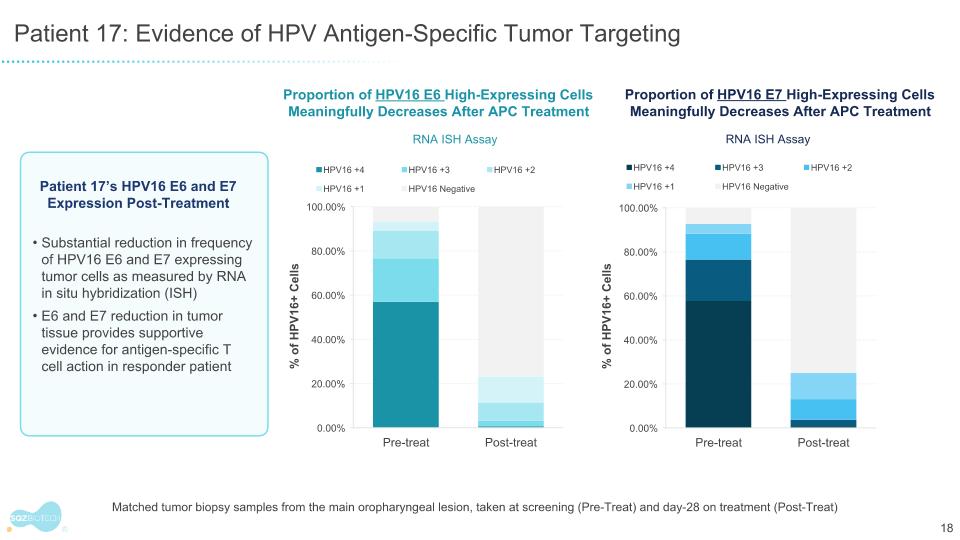
Patient 17: Evidence of HPV Antigen-Specific Tumor Targeting Proportion of HPV16 E6 High-Expressing Cells Meaningfully Decreases After APC Treatment RNA ISH Assay Proportion of HPV16 E7 High-Expressing Cells Meaningfully Decreases After APC Treatment RNA ISH Assay Substantial reduction in frequency of HPV16 E6 and E7 expressing tumor cells as measured by RNA in situ hybridization (ISH) E6 and E7 reduction in tumor tissue provides supportive evidence for antigen-specific T cell action in responder patient Patient 17’s HPV16 E6 and E7 Expression Post-Treatment % of HPV16+ Cells % of HPV16+ Cells Matched tumor biopsy samples from the main oropharyngeal lesion, taken at screening (Pre-Treat) and day-28 on treatment (Post-Treat)

Agenda Welcome Micah Zajic, Chief Business Officer SQZ Oncology Portfolio Armon Sharei, PhD, Chief Executive Officer and Founder SQZ-PBMC-HPV-101 Phase 1/2 Trial Highest-Dose Cohort Findings �Jong Chul Park, MD, Massachusetts General Hospital Monotherapy Summary & Clinical Development �Ricardo Zwirtes, MD, Oncology Clinical Development Combination Strategies and Manufacturing �Howard Bernstein, MD, PhD, Chief Scientific Officer Summary & 2022 Oncology Portfolio Outlook Armon Sharei, PhD, Chief Executive Officer and Founder Questions & Answers Management Team Members
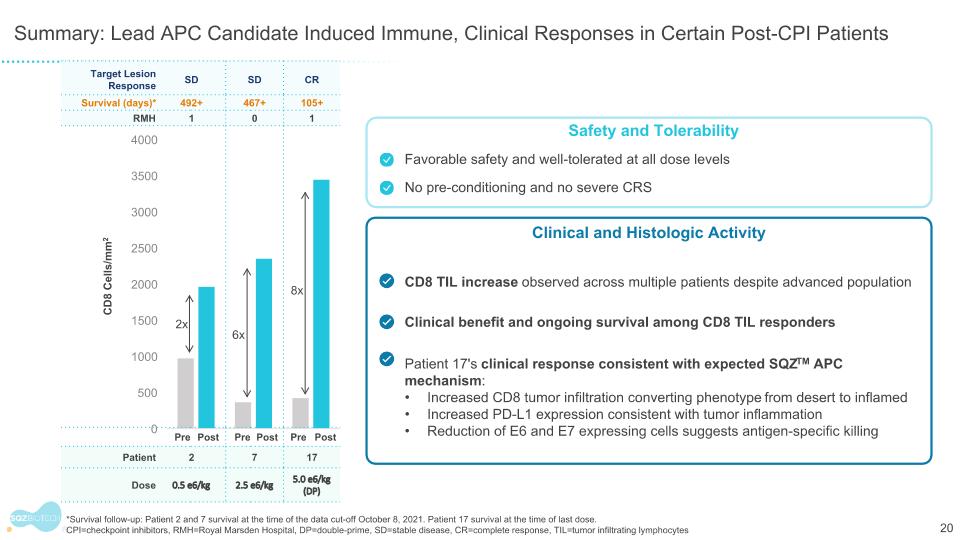
Target Lesion Response SD SD CR Survival (days)* 492+ 467+ 105+ RMH 1 0 1 Pre Post Pre Post Pre Post Patient 2 7 17 Dose 0.5 e6/kg 2.5 e6/kg 5.0 e6/kg (DP) Summary: Lead APC Candidate Induced Immune, Clinical Responses in Certain Post-CPI Patients 2x 6x 8x Safety and Tolerability Favorable safety and well-tolerated at all dose levels No pre-conditioning and no severe CRS Clinical and Histologic Activity CD8 TIL increase observed across multiple patients despite advanced population Clinical benefit and ongoing survival among CD8 TIL responders Patient 17's clinical response consistent with expected SQZTM APC mechanism: Increased CD8 tumor infiltration converting phenotype from desert to inflamed Increased PD-L1 expression consistent with tumor inflammation Reduction of E6 and E7 expressing cells suggests antigen-specific killing CD8 Cells/mm2 *Survival follow-up: Patient 2 and 7 survival at the time of the data cut-off October 8, 2021. Patient 17 survival at the time of last dose. CPI=checkpoint inhibitors, RMH=Royal Marsden Hospital, DP=double-prime, SD=stable disease, CR=complete response, TIL=tumor infiltrating lymphocytes
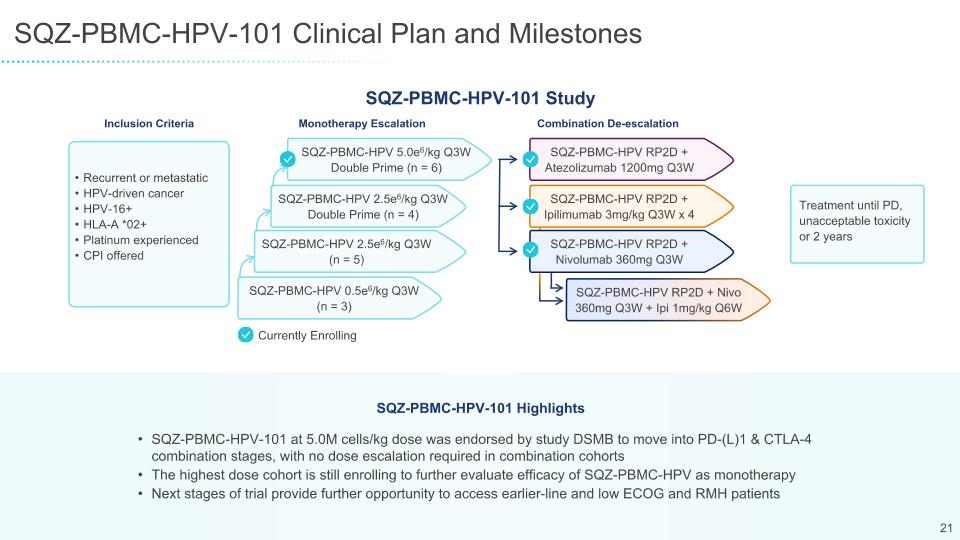
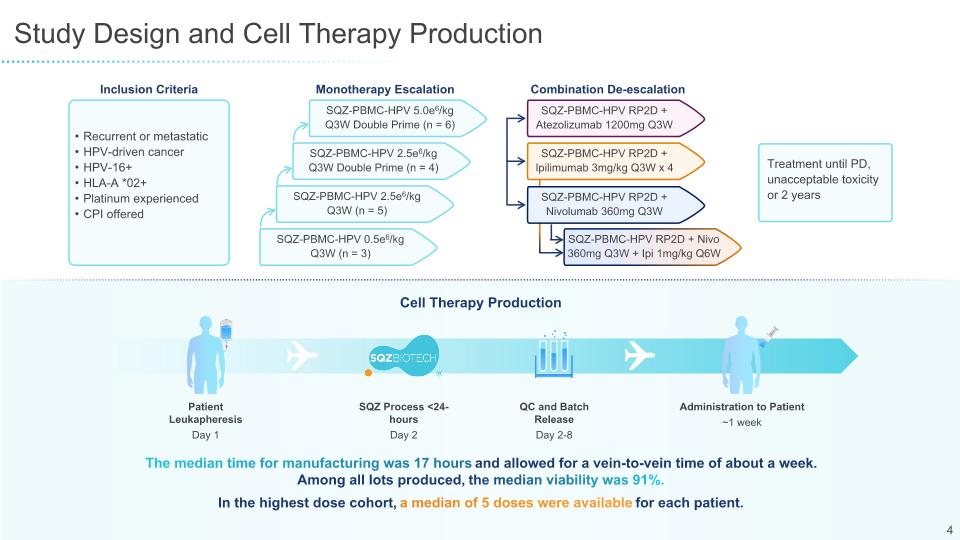
SQZ-PBMC-HPV-101 Clinical Plan and Milestones SQZ-PBMC-HPV-101 at 5.0M cells/kg dose was endorsed by study DSMB to move into PD-(L)1 & CTLA-4 combination stages, with no dose escalation required in combination cohorts The highest dose cohort is still enrolling to further evaluate efficacy of SQZ-PBMC-HPV as monotherapy Next stages of trial provide further opportunity to access earlier-line and low ECOG and RMH patients SQZ-PBMC-HPV-101 Highlights Combination De-escalation Recurrent or metastatic HPV-driven cancer HPV-16+ HLA-A *02+ Platinum experienced CPI offered Inclusion Criteria Treatment until PD, unacceptable toxicity or 2 years Monotherapy Escalation SQZ-PBMC-HPV 5.0e6/kg Q3W Double Prime (n = 6) SQZ-PBMC-HPV 2.5e6/kg Q3W Double Prime (n = 4) SQZ-PBMC-HPV 2.5e6/kg Q3W (n = 5) SQZ-PBMC-HPV 0.5e6/kg Q3W (n = 3) SQZ-PBMC-HPV RP2D + Atezolizumab 1200mg Q3W SQZ-PBMC-HPV RP2D + Ipilimumab 3mg/kg Q3W x 4 SQZ-PBMC-HPV RP2D + Nivolumab 360mg Q3W SQZ-PBMC-HPV RP2D + Nivo 360mg Q3W + Ipi 1mg/kg Q6W SQZ-PBMC-HPV-101 Study Currently Enrolling
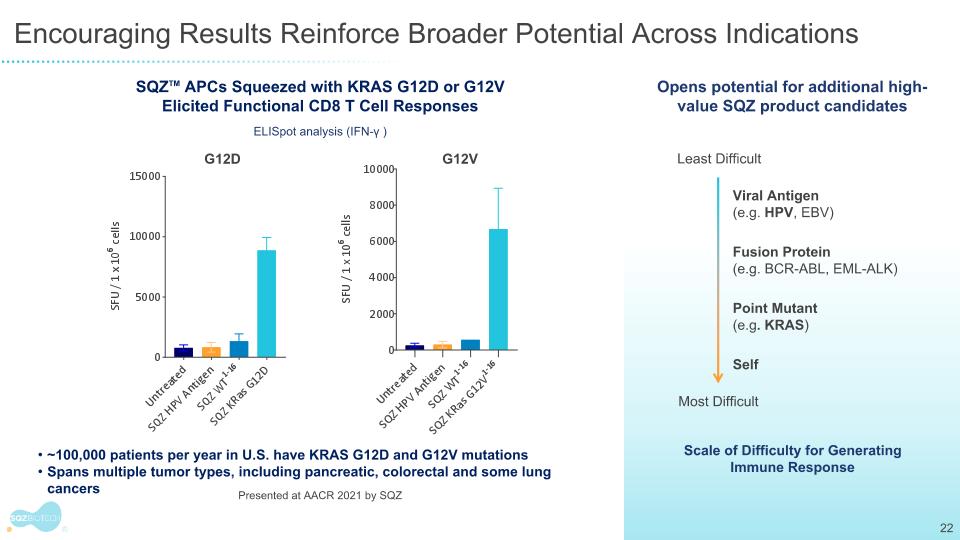
Encouraging Results Reinforce Broader Potential Across Indications SQZTM APCs Squeezed with KRAS G12D or G12V Elicited Functional CD8 T Cell Responses ~100,000 patients per year in U.S. have KRAS G12D and G12V mutations Spans multiple tumor types, including pancreatic, colorectal and some lung cancers G12D G12V Presented at AACR 2021 by SQZ Scale of Difficulty for Generating Immune Response Opens potential for additional high-value SQZ product candidates Viral Antigen (e.g. HPV, EBV) Fusion Protein (e.g. BCR-ABL, EML-ALK) Point Mutant (e.g. KRAS) Self Most Difficult Least Difficult ELISpot analysis (IFN-γ )

Agenda Welcome Micah Zajic, Chief Business Officer SQZ Oncology Portfolio Armon Sharei, PhD, Chief Executive Officer and Founder SQZ-PBMC-HPV-101 Phase 1/2 Trial Highest-Dose Cohort Findings �Jong Chul Park, MD, Massachusetts General Hospital Monotherapy Summary & Clinical Development �Ricardo Zwirtes, MD, Oncology Clinical Development Combination Strategies�Howard Bernstein, MD, PhD, Chief Scientific Officer Summary & 2022 Oncology Portfolio Outlook Armon Sharei, PhD, Chief Executive Officer and Founder Questions & Answers Management Team Members
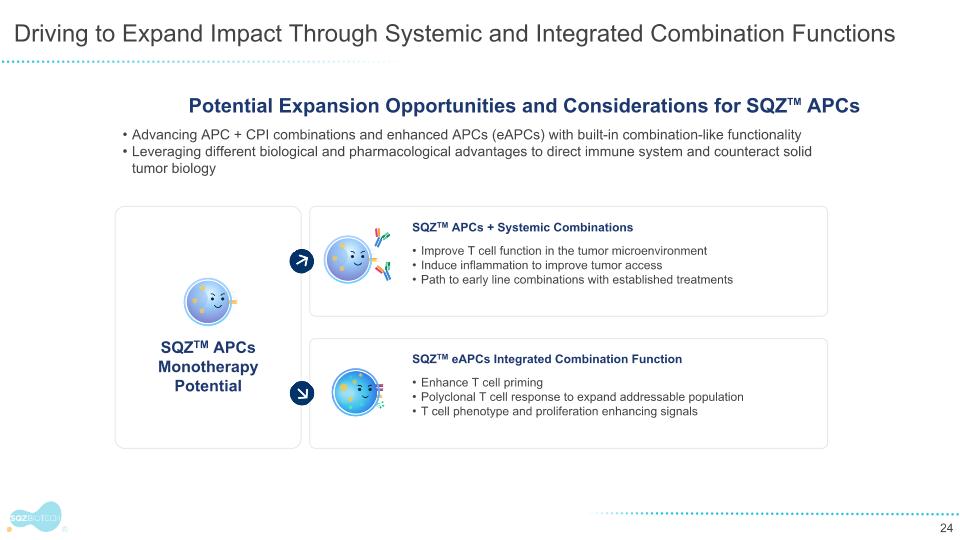
SQZTM APCs + Systemic Combinations Improve T cell function in the tumor microenvironment Induce inflammation to improve tumor access Path to early line combinations with established treatments SQZTM eAPCs Integrated Combination Function Enhance T cell priming Polyclonal T cell response to expand addressable population T cell phenotype and proliferation enhancing signals Driving to Expand Impact Through Systemic and Integrated Combination Functions Advancing APC + CPI combinations and enhanced APCs (eAPCs) with built-in combination-like functionality Leveraging different biological and pharmacological advantages to direct immune system and counteract solid tumor biology SQZTM APCs Monotherapy Potential Potential Expansion Opportunities and Considerations for SQZTM APCs
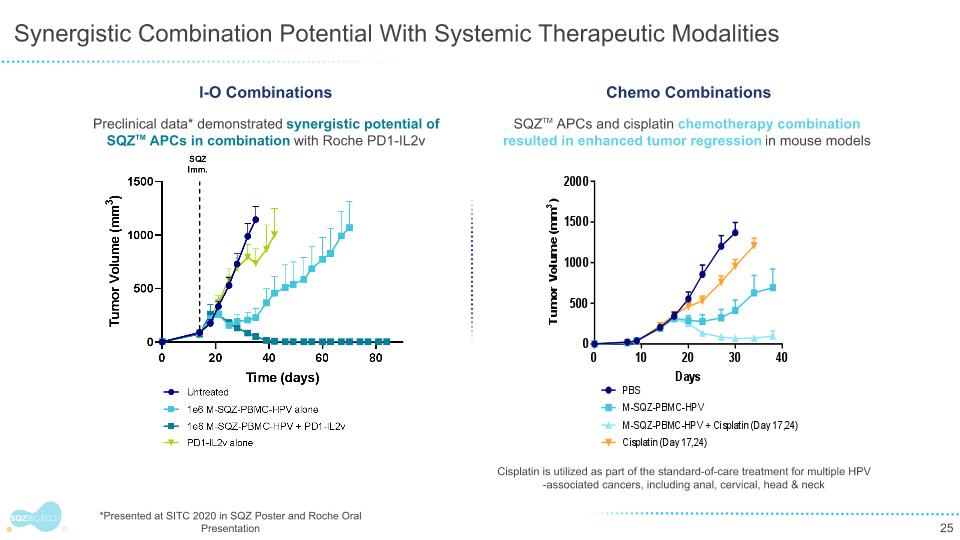
Synergistic Combination Potential With Systemic Therapeutic Modalities *Presented at SITC 2020 in SQZ Poster and Roche Oral Presentation Preclinical data* demonstrated synergistic potential of SQZTM APCs in combination with Roche PD1-IL2v I-O Combinations SQZTM APCs and cisplatin chemotherapy combination resulted in enhanced tumor regression in mouse models Cisplatin is utilized as part of the standard-of-care treatment for multiple HPV-associated cancers, including anal, cervical, head & neck Chemo Combinations
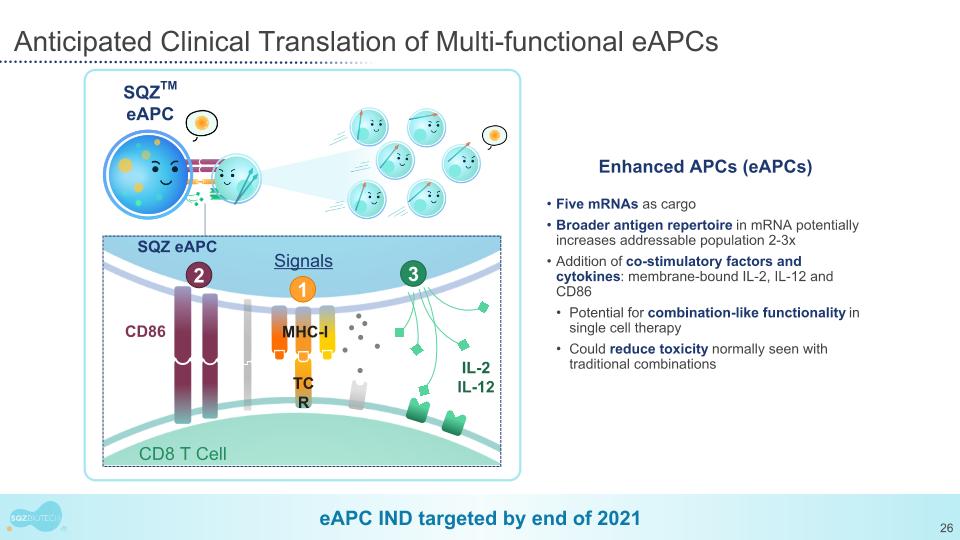
Anticipated Clinical Translation of Multi-functional eAPCs eAPC IND targeted by end of 2021 SQZTM eAPC CD86 1 3 Signals CD8 T Cell SQZ eAPC IL-2 IL-12 TCR MHC-I Five mRNAs as cargo Broader antigen repertoire in mRNA potentially increases addressable population 2-3x Addition of co-stimulatory factors and cytokines: membrane-bound IL-2, IL-12 and CD86 Potential for combination-like functionality in single cell therapy Could reduce toxicity normally seen with traditional combinations Enhanced APCs (eAPCs) 2

Agenda Welcome Micah Zajic, Chief Business Officer SQZ Oncology Portfolio Armon Sharei, PhD, Chief Executive Officer and Founder SQZ-PBMC-HPV-101 Phase 1/2 Trial Highest-Dose Cohort Findings �Jong Chul Park, MD, Massachusetts General Hospital Monotherapy Summary & Clinical Development �Ricardo Zwirtes, MD, Oncology Clinical Development Manufacturing Innovation�Howard Bernstein, MD, PhD, Chief Scientific Officer Summary & 2022 Oncology Portfolio Outlook Armon Sharei, PhD, Chief Executive Officer and Founder Questions & Answers Management Team Members
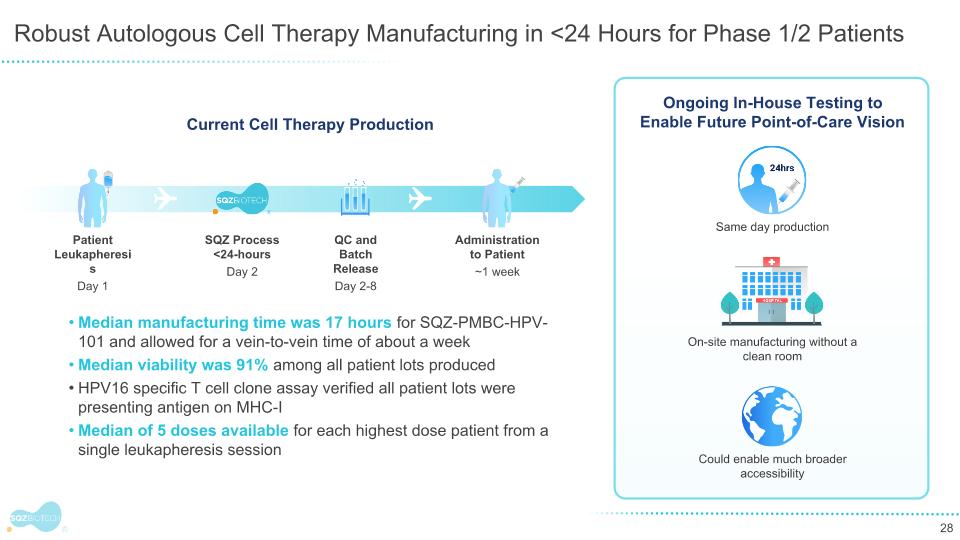
Robust Autologous Cell Therapy Manufacturing in <24 Hours for Phase 1/2 Patients Current Cell Therapy Production Patient Leukapheresis Day 1 SQZ Process <24-hours Day 2 QC and Batch Release Day 2-8 Administration to Patient ~1 week Median manufacturing time was 17 hours for SQZ-PMBC-HPV-101 and allowed for a vein-to-vein time of about a week Median viability was 91% among all patient lots produced HPV16 specific T cell clone assay verified all patient lots were presenting antigen on MHC-I Median of 5 doses available for each highest dose patient from a single leukapheresis session Ongoing In-House Testing to Enable Future Point-of-Care Vision On-site manufacturing without a clean room Same day production Could enable much broader accessibility

Agenda Welcome Micah Zajic, Chief Business Officer SQZ Oncology Portfolio Armon Sharei, PhD, Chief Executive Officer and Founder SQZ-PBMC-HPV-101 Phase 1/2 Trial Highest-Dose Cohort Findings �Jong Chul Park, MD, Massachusetts General Hospital Monotherapy Summary & Clinical Development �Ricardo Zwirtes, MD, Oncology Clinical Development Combination Strategies and Manufacturing �Howard Bernstein, MD, PhD, Chief Scientific Officer Summary & 2022 Oncology Portfolio Outlook Armon Sharei, PhD, Chief Executive Officer and Founder Questions & Answers Management Team Members
Summary & 2022 Oncology Portfolio Outlook APC platform progress supports broader development for HPV+ solid tumors and expansion to new antigens Patient 17 illustrated differentiated clinical impact potential of SQZ™ APC program Increased CD8 TIL and PD-L1 expression underscores potential CPI synergy Systemic combination cohorts with CPI are enrolling. eAPCs with multiple combination-like functions on track for 2021 IND filing Oncology clinical data through end of 2022 SQZTM eAPCs IND on track for filing by YE ‘21 Phase 1/2 monotherapy data SQZTM AACs Phase 1/2 monotherapy data SQZTM APCs Phase 1/2 combination data

Questions & Answers SQZ-PBMC-HPV-101 Clinical Program Updates at ESMO Immuno-Oncology Congress 2021
 Empowering Cells to Change Lives
Empowering Cells to Change Lives Empowering Cells to Change Lives
Empowering Cells to Change Lives Empowering Cells to Change Lives
Empowering Cells to Change Lives Empowering Cells to Change Lives
Empowering Cells to Change Lives