
Q4 2019 Earnings Conference Call March 12, 2020 ©2020 ViewRay, Inc. All rights reserved.

FORWARD-LOOKING STATEMENTS This presentation has been prepared solely for use at this meeting and is intended for investors and analysts only. The material is given in conjunction with an oral presentation and should not be taken out of context. Unless the context requires otherwise, references to “ViewRay,” “the company,” “we,” “us” and “our,” refer to ViewRay, Inc. Except for historical information, ViewRay’s written and accompanying oral presentation may contain forward-looking statements, including statements about the overall industry, including but not limited to: our current expectations of the market; growth drivers; future trends; demand for radiation oncology products and features; and innovation and growth opportunities. Forward-looking statements also include, but are not limited to, statements about ViewRay’s: future orders; backlog or earnings growth; future financial results; and market acceptance of ViewRay’s existing products, future products, or technology. Words such as “could,” “anticipates,” “expects,” “outlook,” “intends,” “plans,” “believes,” “seeks,” “vision,” “estimates,” “may,” “will,” “future,” “horizon,” “aiming,” “driving,” “target” (or variations of them,) and similar statements, are forward-looking statements. These types of statements express management’s beliefs based on the information available to us as of the date of this presentation, are subject to change, and are not guarantees of future performance. Forward-looking statements involve risks, uncertainties, and assumptions that are difficult to predict and could cause ViewRay’s results to differ materially from those presented. These risks, uncertainties, and assumptions include, but are not limited to, changes in: the regulatory environment; global economics; trade compliance requirements, duties or tariffs; third-party reimbursement levels; currency exchange rates; taxation, healthcare law, and product clearance requirements, as well as those related to: adverse publicity about ViewRay and our products; our reliance on sole or limited source suppliers; our ability to commercialize our products successfully; the impact of competitive products and pricing, and all other risks listed from time to time in the company’s filings with the Securities and Exchange Commission, which are incorporated into this Forward-Looking Statements disclosure by this reference. We do not assume any obligation to update or revise the forward-looking statements in ViewRay’s written or oral presentation, whether based on future events, new or additional information or otherwise. ViewRay’s written and oral presentation does not constitute an offer to sell, or the solicitation of an offer to buy, securities. The opinions and clinical experiences presented herein are specific to the featured physicians and/or the featured patients and are for information purposes only. Nothing in this material is intended to provide specific medical advice or to take the place of written law or regulations. Individual customer results are illustrative only and are not predictive of future results. MEDICAL ADVICE DISCLAIMER ViewRay is a medical device manufacturer and cannot and does not recommend specific treatment approaches. Individual results may vary. Forward-Looking Statements & Disclaimer ©2020 ViewRay, Inc. All rights reserved. 2

Our Mission: Treat and Prove What Others Can’t. ©2020 ViewRay, Inc. All rights reserved. patients treated to date1 systems1 Merit demonstrated in of peer-reviewed journal articles and abstracts1 >8,000 34 Hundreds 3

2020 Guidance ©2020 ViewRay, Inc. All rights reserved. Revenue in the range of $58 - $95 million Cash use in the range of $60 to $80 million Full year impact is uncertain 9 installations planned for 2020 in regions with travel restrictions currently in place REVENUE CASH USE CORONAVIRUS 4

Current Technology for SBRT Evidence has shown that ablative dose, 75-100 Gy biological effective doses (BED10), drive favorable outcomes2 Conventional LINACs are limited by dose constraints of surrounding health tissue Clinicians balance the need for ablative doses/tumor coverage vs the risk of toxicity/injury to organs at risk ©2020 ViewRay, Inc. All rights reserved. LOCAL CONTROL TOXICITY Trade-off Between Safety and Effectiveness Limits Adoption of New Treatment Paradigms 5

MRIdian: No Compromise Necessary On-table adaptation provides clinicians: Dose escalation in multiple tumor sites to 75-100 Gy BED103 Maintenance of normal organ dose limits or constraints Tighter treatment margins, PTV ≅ CTV3 Real time tumor tracking & gating: Treat the tumor and spare healthy tissue3 Reduces dose to organs at risk5 by reducing treatment margins Real-time MR Imaging Enables: High-dose, ablative SBRT Reduced toxicity Improved outcomes No fiducials/surrogates required ©2020 ViewRay, Inc. All rights reserved. 6

When ‘Good Enough’ Is Not Good Enough MRIdian vs. Traditional CT Linac Images courtesy of the Siteman Cancer Center at Washington University School of Medicine MRIdian Market-leading CT LINAC 15 – 30mm Treatment Margins 1 – 5mm Treatment Margins Visibly Different Treatment Margins Software differentiates soft tissue from organs and spares healthy tissue ©2020 ViewRay, Inc. All rights reserved. 7

Where We Are Today: MRIdian has been utilized to treat over 50 ICD cancer types, and its evidence has been documented in hundreds of peer-reviewed journal articles and abstracts ©2020 ViewRay, Inc. All rights reserved. Red Journal6 AUMC treated high-risk lung patients, demonstrating the ability to deliver high doses in a complex patient population with low resulting toxicity Red Journal4 Washington University treating APBI in a single fraction, enabled by MRIdian’s small treatment margins MRIdian “provides a convenient, less-toxic, and more feasible option for breast cancer patients post surgery.” Lancet Oncology5 Identified MR-guided therapy as a “practice changing technology that allows for precise delivery of high dose in liver and elsewhere.” 8
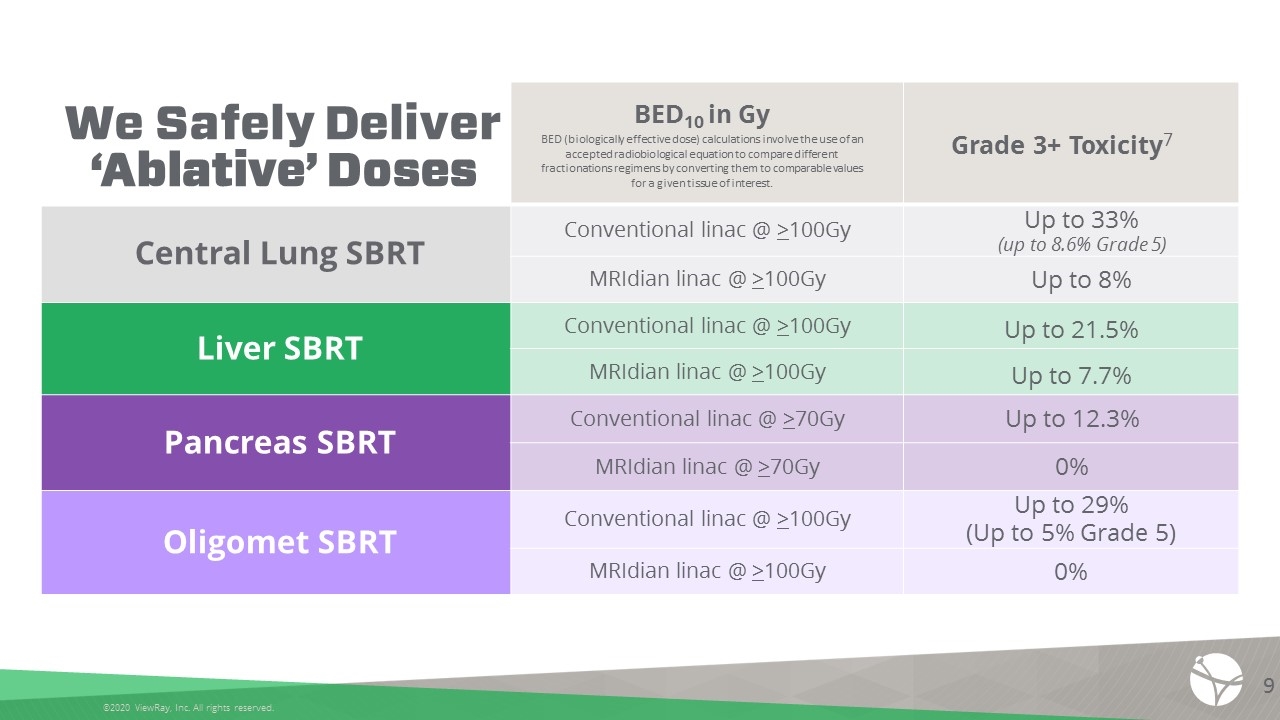
BED10 in Gy BED (biologically effective dose) calculations involve the use of an accepted radiobiological equation to compare different fractionations regimens by converting them to comparable values for a given tissue of interest. Grade 3+ Toxicity7 Central Lung SBRT Conventional linac @ >100Gy Up to 33% (up to 8.6% Grade 5) MRIdian linac @ >100Gy Up to 8% Liver SBRT Conventional linac @ >100Gy Up to 21.5% MRIdian linac @ >100Gy Up to 7.7% Pancreas SBRT Conventional linac @ >70Gy Up to 12.3% MRIdian linac @ >70Gy 0% Oligomet SBRT Conventional linac @ >100Gy Up to 29% (Up to 5% Grade 5) MRIdian linac @ >100Gy 0% We Safely Deliver ‘Ablative’ Doses ©2020 ViewRay, Inc. All rights reserved. 9

Some may prescribe very high dose but allow the target coverage to be potentially horribly low. This is how toxicity might be mitigated but on the other hand they are not giving ablative doses to most or all of the tumor in some cases. I am able to treat with ablative doses to all or nearly all of the tumor with MRIdian and still cause almost no major toxicity. ©2020 ViewRay, Inc. All rights reserved. Michael Chuong, M.D. Department of Radiation Oncology Director of Proton Therapy and MR-Guided Radiation Therapy Miami Cancer Institute KOL Perspective “ “ 10
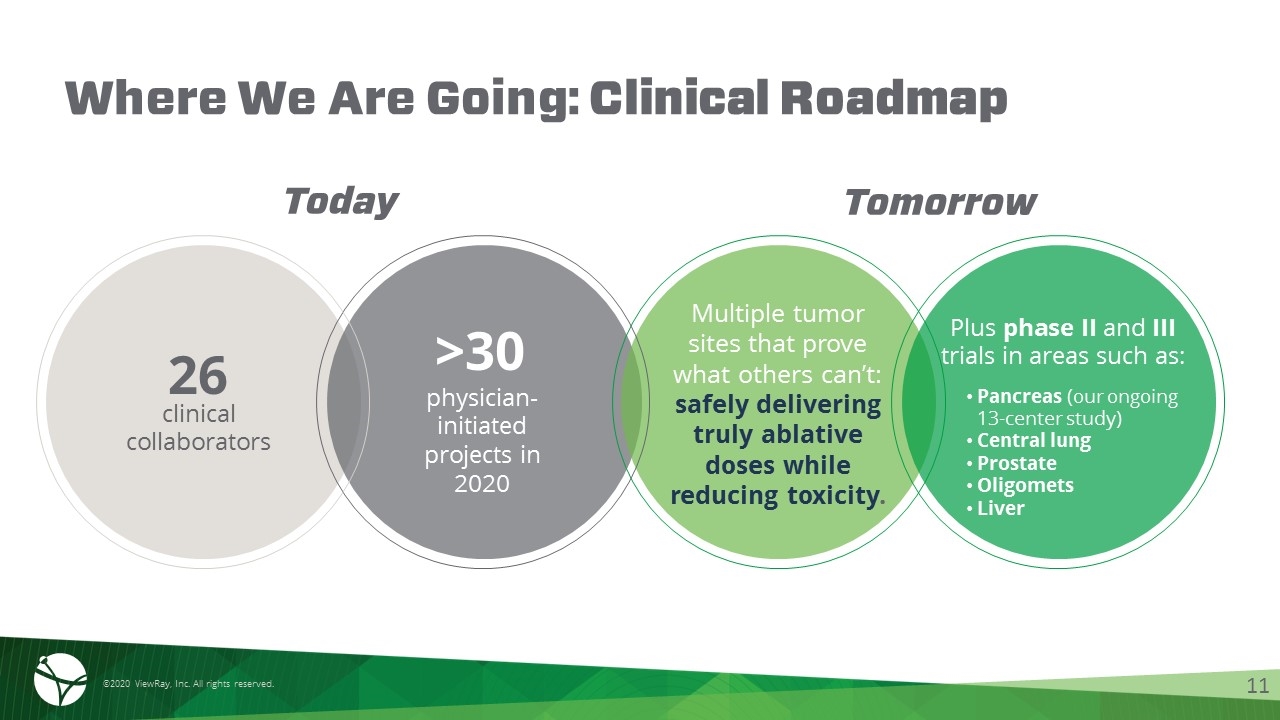
Where We Are Going: Clinical Roadmap clinical collaborators physician-initiated projects in 2020 Multiple tumor sites that prove what others can’t: safely delivering truly ablative doses while reducing toxicity. 26 >30 Plus phase II and III trials in areas such as: Pancreas (our ongoing 13-center study) Central lung Prostate Oligomets Liver Today Tomorrow ©2020 ViewRay, Inc. All rights reserved. 11

Strategic Value Yields Economic Value ©2020 ViewRay, Inc. All rights reserved. UP TO 80 INCREMENTAL PATIENTS FOR CANCER PROGRAMS8 + = Powerful Combination ü “…had no other viable options for treatment.” – Dr. Siddiqui, Henry Ford Health Systems ü Multimodality cancer program with MRIdian enables movement of high fraction patients to fewer fraction SBRT, capturing operational efficiencies to expand program capacity ü Patients with No Other Options Net New Patients Optimizing Care Delivery 12

Optimizing Care Delivery Today ©2020 ViewRay, Inc. All rights reserved. Common IMRT Practice: 28 – 44 Fx × 15 min = 420 – 660 min POTENTIAL TO INCREASE PATIENT THROUGHPUT BY 2.3× TO 3.7× Prostate Cancer MRIdian SBRT: 5 Fx with 2 adaptions = 180 min9 13

Economic Value Development ©2020 ViewRay, Inc. All rights reserved. Flexible Alternative Acquisition Model Reduce Treatment Times Appropriate and Higher Reimbursement $1 million of COGS reduction over the next three years Sub 30-minute treatment times in prostate and breast Continue to work on differentiated reimbursement in the US, Japan, and select European countries ü ü ü 14

Summary ©2020 ViewRay, Inc. All rights reserved. ü ü Roadmap to Win: Further Expanding our Clinical, Strategic, and Economic Value Clinical: 30 physician-initiated projects in 2020, plus developing phase II and III studies Strategic and Economic: Sub 30-minute treatment times, flexible alternative acquisition model, appropriate and higher reimbursement MRIdian offers a no compromise solution to safely deliver ablative doses 15

Q4 Financial Summary Total revenue of $16.5 million in the quarter, primarily from three revenue units, including one system upgrade Received 4 MRIdian orders in the quarter totaling approximately $21.2 million Total backlog as of December 31, 2019: $227.3 million Cash and cash equivalents as of December 31, 2019: $226.8 million ©2020 ViewRay, Inc. All rights reserved. 16


References ©2020 ViewRay, Inc. All rights reserved. 2020 internal and historical company data Hong JC, Ayala-Peacock DN, Lee J, Blackstock AW, Okunieff P, Sung MW, et al. (2018) Classification for long-term survival in oligometastatic patients treated with ablative radiotherapy: A multi-institutional pooled analysis. PLoS ONE 13(4): e0195149; Schneider BJ, Daly ME, Kennedy EB, et al. Stereotactic Body Radiotherapy for Early-Stage Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Endorsement of the American Society for Radiation Oncology Evidence-Based Guideline. J Clin Oncol. 2018;36(7):710–719; Finazzi T, Haasbeek CJA, Spoelstra FOB, Palacios MA, Admiraal MA, Bruynzeel AME, Slotman BJ, Lagerwaard FJ, Senan S, Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors International Journal of Radiation Oncology • Biology• Physics (2020); Toesca DA, Osmundson EC, Eyben RV, et al. Central liver toxicity after SBRT: An expanded analysis and predictive nomogram. Radiother Oncol. 2017;122(1):130–136; Rosenberg S. A. et al. (2018). A multi-institutional experience of MR-guided liver stereotactic body radiation therapy. Advances in Radiation Oncology, 4(1), 142-149; Herman JM, Chang DT, Goodman KA, et al. Phase 2 multi-institutional trial evaluating gemcitabine and stereotactic body radiotherapy for patients with locally advanced unresectable pancreatic adenocarcinoma. Cancer. 2015;121(7):1128-1137. ; Rudra S. et al. (2019). Using adaptive magnetic resonance image-guided radiation therapy for treatment of inoperable pancreatic cancer. Cancer Medicine, 8(5), 2123-2132; Palma DA, Olson R, Harrow S, et al. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): a randomised, phase 2, open-label trial. Lancet. 2019;393(10185):2051–2058. doi:10.1016/S0140-6736(18)32487-5; Henke, L., et al. (2018). Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiotherapy and Oncology, 126(3),Henke, L., et al. (2018). Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiotherapy and Oncology, 126(3), 519-526; Henke LE, et al. Stereotactic mr-guided online adaptive radiation therapy (smart) for ultracentral thorax malignancies: Results of a phase 1 trial. Adv Radiat Oncol 2019;4:201-209; Rosenberg SA, et al. A multi-institutional experience of mr-guided liver stereotactic body radiation therapy. Adv Radiat Oncol 2019;4:142-149; Finazzi T, Haasbeek CJA, Spoelstra FOB, Palacios MA, Admiraal MA, Bruynzeel AME, Slotman BJ, Lagerwaard FJ, Senan S, Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors International Journal of Radiation Oncology • Biology• Physics (2020); Rudra S. et al. (2019). Using adaptive magnetic resonance image-guided radiation therapy for treatment of inoperable pancreatic cancer. Cancer Medicine, 8(5), 2123-2132; Finazzi, et al. Role of on-table plan adaptation in MR-guided ablative radiation therapy for central lung tumors. Int J Radiat Oncol Biol Phys. 2019 Jul 15;104(4):933-941. doi: 10.1016/j.ijrobp.2019.03.035. Epub 2019 Mar 28; Kennedy WR, Thomas MA, Stanley JA, Luo J, Ochoa LL, Clifton KK, Cyr AE, Margenthaler JA, DeWees TA, Price A, Kashani R, Green O, Zoberi I, Single Institution Phase I/II Prospective Clinical Trial of Single Fraction High Gradient Adjuvant Partial Breast Irradiation for Hormone Sensitive Stage 0-I Breast Cancer, International Journal of Radiation Oncology • Biology • Physics (2020); Henke, L., et al. (2018). Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiotherapy and Oncology, 126(3), 519-526; Bruynzeel AME, Tetar SU, Oei SS, Senan S, Haasbeek CJA, Spoelstra FOB, Piet AHM, Meijnen P, Bakker van der Jagt MAB, Fraikin T, Slotman BJ, van Moorselaar RJA, Lagerwaard FJ, A prospective single-arm phase II study of stereotactic magnetic-resonance-guided adaptive radiotherapy for prostate cancer: Early toxicity results, International Journal of Radiation Oncology • Biology • Physics (2019); Finazzi T, Haasbeek CJA, Spoelstra FOB, Palacios MA, Admiraal MA, Bruynzeel AME, Slotman BJ, Lagerwaard FJ, Senan S, Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors International Journal of Radiation Oncology • Biology • Physics (2020); El-Bared N, et al. Dosimetric benefits and practical pitfalls of daily online adaptive mri-guided stereotactic radiotherapy for pancreatic cancer. Pract Radiat Oncol 2018; Hegde JV, et al. Magnetic resonance imaging guidance mitigates the effects of intrafraction prostate motion during stereotactic body radiotherapy for prostate cancer. Cureus 2018;10:e2442; Kishan AU, et al. Mri-guided dose-escalated salvage radiotherapy for bulky bladder neck recurrence of prostate cancer. Cureus 2018;10:e2360. Henke, L., et al. (2018). Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiotherapy and Oncology, 126(3), 519-526; Henke LE, et al. Stereotactic mr-guided online adaptive radiation therapy (smart) for ultracentral thorax malignancies: Results of a phase 1 trial. Adv Radiat Oncol 2019;4:201-209; Rosenberg SA, et al. A multi-institutional experience of mr-guided liver stereotactic body radiation therapy. Adv Radiat Oncol 2019;4:142-149; Finazzi T, Haasbeek CJA, Spoelstra FOB, Palacios MA, Admiraal MA, Bruynzeel AME, Slotman BJ, Lagerwaard FJ, Senan S, Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors International Journal of Radiation Oncology • Biology• Physics (2020); Rudra S. et al. (2019). Using adaptive magnetic resonance image-guided radiation therapy for treatment of inoperable pancreatic cancer. Cancer Medicine, 8(5), 2123-2132; Finazzi, et al. Role of on-table plan adaptation in MR-guided ablative radiation therapy for central lung tumors. Int J Radiat Oncol Biol Phys. 2019 Jul 15;104(4):933-941. doi: 10.1016/j.ijrobp.2019.03.035. Epub 2019 Mar 28; Kennedy WR, Thomas MA, Stanley JA, Luo J, Ochoa LL, Clifton KK, Cyr AE, Margenthaler JA, DeWees TA, Price A, Kashani R, Green O, Zoberi I, Single Institution Phase I/II Prospective Clinical Trial of Single Fraction High Gradient Adjuvant Partial Breast Irradiation for Hormone Sensitive Stage 0-I Breast Cancer, International Journal of Radiation Oncology • Biology • Physics (2020); Henke, L., et al. (2018). Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiotherapy and Oncology, 126(3), 519-526; Bruynzeel AME, Tetar SU, Oei SS, Senan S, Haasbeek CJA, Spoelstra FOB, Piet AHM, Meijnen P, Bakker van der Jagt MAB, Fraikin T, Slotman BJ, van Moorselaar RJA, Lagerwaard FJ, A prospective single-arm phase II study of stereotactic magnetic-resonance-guided adaptive radiotherapy for prostate cancer: Early toxicity results, International Journal of Radiation Oncology • Biology • Physics (2019); Finazzi T, Haasbeek CJA, Spoelstra FOB, Palacios MA, Admiraal MA, Bruynzeel AME, Slotman BJ, Lagerwaard FJ, Senan S, Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors International Journal of Radiation Oncology • Biology • Physics (2020); El-Bared N, et al. Dosimetric benefits and practical pitfalls of daily online adaptive mri-guided stereotactic radiotherapy for pancreatic cancer. Pract Radiat Oncol 2018; Hegde JV, et al. Magnetic resonance imaging guidance mitigates the effects of intrafraction prostate motion during stereotactic body radiotherapy for prostate cancer. Cureus 2018;10:e2442; Kishan AU, et al. Mri-guided dose-escalated salvage radiotherapy for bulky bladder neck recurrence of prostate cancer. Cureus 2018;10:e2360. Kennedy WR, Thomas MA, Stanley JA, Luo J, Ochoa LL, Clifton KK, Cyr AE, Margenthaler JA, DeWees TA, Price A, Kashani R, Green O, Zoberi I, Single Institution Phase I/II Prospective Clinical Trial of Single Fraction High Gradient Adjuvant Partial Breast Irradiation for Hormone Sensitive Stage 0-I Breast Cancer, International Journal of Radiation Oncology • Biology • Physics (2020) Witt, J., et al. (2020). MRI-guided adaptive radiotherapy for liver tumours: visualizing the future. Lancet Oncol 2020; 21:e74-82. Finazzi T, Haasbeek CJA, Spoelstra FOB, Palacios MA, Admiraal MA, Bruynzeel AME, Slotman BJ, Lagerwaard FJ, Senan S, Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors International Journal of Radiation Oncology • Biology • Physics (2020) Schneider BJ, Daly ME, Kennedy EB, et al. Stereotactic Body Radiotherapy for Early-Stage Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Endorsement of the American Society for Radiation Oncology Evidence-Based Guideline. J Clin Oncol. 2018;36(7):710–719; Finazzi T, Haasbeek CJA, Spoelstra FOB, Palacios MA, Admiraal MA, Bruynzeel AME, Slotman BJ, Lagerwaard FJ, Senan S, Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors International Journal of Radiation Oncology • Biology• Physics (2020); Toesca DA, Osmundson EC, Eyben RV, et al. Central liver toxicity after SBRT: An expanded analysis and predictive nomogram. Radiother Oncol. 2017;122(1):130–136; Rosenberg S. A. et al. (2018). A multi-institutional experience of MR-guided liver stereotactic body radiation therapy. Advances in Radiation Oncology, 4(1), 142-149; Herman JM, Chang DT, Goodman KA, et al. Phase 2 multi-institutional trial evaluating gemcitabine and stereotactic body radiotherapy for patients with locally advanced unresectable pancreatic adenocarcinoma. Cancer. 2015;121(7):1128-1137; Rudra S. et al. (2019). Using adaptive magnetic resonance image-guided radiation therapy for treatment of inoperable pancreatic cancer. Cancer Medicine, 8(5), 2123-2132; Palma DA, Olson R, Harrow S, et al. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): a randomised, phase 2, open-label trial. Lancet. 2019;393(10185):2051–2058. doi:10.1016/S0140-6736(18)32487-5; Henke, L., et al. (2018). Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiotherapy and Oncology, 126(3), 519-526. Based on 2020 customer survey data Internal company data; Clinical implementation of magnetic resonance imaging guided adaptive radiotherapy for localized prostate cancer Tetar, Shyama U. et al. Physics and Imaging in Radiation Oncology, Volume 9, 69 – 76.