kin-2020123100015617432020FYfalseP2YP15YP1YP1Y0.3300015617432020-01-012020-12-310001561743us-gaap:CommonStockMember2020-01-012020-12-310001561743us-gaap:PreferredStockMember2020-01-012020-12-31iso4217:USD00015617432020-06-30xbrli:shares00015617432021-02-2800015617432020-12-3100015617432019-12-31iso4217:USDxbrli:shares0001561743kin:ProductRevenueMember2020-01-012020-12-310001561743kin:ProductRevenueMember2019-01-012019-12-310001561743kin:ProductRevenueMember2018-01-012018-12-310001561743kin:AssetSaleMember2020-01-012020-12-310001561743kin:AssetSaleMember2019-01-012019-12-310001561743kin:AssetSaleMember2018-01-012018-12-310001561743us-gaap:RoyaltyMember2020-01-012020-12-310001561743us-gaap:RoyaltyMember2019-01-012019-12-310001561743us-gaap:RoyaltyMember2018-01-012018-12-310001561743kin:ManufacturingMember2020-01-012020-12-310001561743kin:ManufacturingMember2019-01-012019-12-310001561743kin:ManufacturingMember2018-01-012018-12-310001561743kin:PartnerLicensingMember2020-01-012020-12-310001561743kin:PartnerLicensingMember2019-01-012019-12-310001561743kin:PartnerLicensingMember2018-01-012018-12-3100015617432019-01-012019-12-3100015617432018-01-012018-12-310001561743us-gaap:CommonStockMember2017-12-310001561743us-gaap:AdditionalPaidInCapitalMember2017-12-310001561743us-gaap:AccumulatedOtherComprehensiveIncomeMember2017-12-310001561743us-gaap:RetainedEarningsMember2017-12-3100015617432017-12-310001561743us-gaap:RetainedEarningsMember2018-01-012018-12-310001561743us-gaap:AccumulatedOtherComprehensiveIncomeMember2018-01-012018-12-310001561743us-gaap:CommonStockMember2018-01-012018-12-310001561743us-gaap:AdditionalPaidInCapitalMember2018-01-012018-12-310001561743us-gaap:CommonStockMemberkin:AtTheMarketOfferingMember2018-01-012018-12-310001561743us-gaap:AdditionalPaidInCapitalMemberkin:AtTheMarketOfferingMember2018-01-012018-12-310001561743kin:AtTheMarketOfferingMember2018-01-012018-12-310001561743kin:PublicOfferingMemberus-gaap:CommonStockMember2018-01-012018-12-310001561743kin:PublicOfferingMemberus-gaap:AdditionalPaidInCapitalMember2018-01-012018-12-310001561743kin:PublicOfferingMember2018-01-012018-12-310001561743us-gaap:CommonStockMember2018-12-310001561743us-gaap:AdditionalPaidInCapitalMember2018-12-310001561743us-gaap:AccumulatedOtherComprehensiveIncomeMember2018-12-310001561743us-gaap:RetainedEarningsMember2018-12-3100015617432018-12-310001561743us-gaap:RetainedEarningsMember2019-01-012019-12-310001561743us-gaap:AccumulatedOtherComprehensiveIncomeMember2019-01-012019-12-310001561743us-gaap:CommonStockMember2019-01-012019-12-310001561743us-gaap:AdditionalPaidInCapitalMember2019-01-012019-12-310001561743kin:PublicOfferingMemberus-gaap:CommonStockMember2019-01-012019-12-310001561743kin:PublicOfferingMemberus-gaap:AdditionalPaidInCapitalMember2019-01-012019-12-310001561743kin:PublicOfferingMember2019-01-012019-12-310001561743us-gaap:CommonStockMember2019-12-310001561743us-gaap:AdditionalPaidInCapitalMember2019-12-310001561743us-gaap:AccumulatedOtherComprehensiveIncomeMember2019-12-310001561743us-gaap:RetainedEarningsMember2019-12-310001561743us-gaap:RetainedEarningsMember2020-01-012020-12-310001561743us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-01-012020-12-310001561743us-gaap:CommonStockMember2020-01-012020-12-310001561743us-gaap:AdditionalPaidInCapitalMember2020-01-012020-12-310001561743us-gaap:CommonStockMemberkin:AtTheMarketOfferingMember2020-01-012020-12-310001561743us-gaap:AdditionalPaidInCapitalMemberkin:AtTheMarketOfferingMember2020-01-012020-12-310001561743kin:AtTheMarketOfferingMember2020-01-012020-12-310001561743us-gaap:CommonStockMember2020-12-310001561743us-gaap:AdditionalPaidInCapitalMember2020-12-310001561743us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-12-310001561743us-gaap:RetainedEarningsMember2020-12-31kin:class_of_stock0001561743kin:KindredBioEquineIncMember2016-04-252016-04-250001561743kin:KindredBioEquineIncMember2016-04-250001561743kin:CentaurBiopharmaceuticalServicesMember2016-04-252016-04-250001561743kin:CentaurBiopharmaceuticalServicesMember2016-04-250001561743kin:AssetSaleMiratazMember2020-03-162020-03-160001561743kin:AssetSaleMember2020-03-162020-03-1600015617432020-04-1500015617432020-04-152020-04-15kin:position00015617432020-03-162020-03-1600015617432020-06-082020-06-0800015617432020-10-012020-12-3100015617432020-07-012020-09-3000015617432020-12-012020-12-310001561743us-gaap:IPOMember2013-12-012019-12-310001561743kin:HCWainwrightCoLLCMember2020-04-082020-04-080001561743us-gaap:FurnitureAndFixturesMember2020-01-012020-12-310001561743us-gaap:ComputerEquipmentMember2020-01-012020-12-310001561743us-gaap:LandImprovementsMember2020-01-012020-12-310001561743us-gaap:BuildingAndBuildingImprovementsMember2020-01-012020-12-310001561743kin:AssetSaleMiratazMember2020-03-012020-03-3100015617432020-01-012020-03-31xbrli:pure0001561743kin:FourDistributorCustomersMemberus-gaap:SalesRevenueNetMemberus-gaap:CustomerConcentrationRiskMember2020-01-012020-12-310001561743us-gaap:SalesRevenueNetMemberkin:ThreeDistributorCustomersMemberus-gaap:CustomerConcentrationRiskMember2019-01-012019-12-310001561743kin:FourDistributorCustomersMemberus-gaap:SalesRevenueNetMemberus-gaap:CustomerConcentrationRiskMember2018-01-012018-12-310001561743kin:ZimetaMember2020-01-012020-12-310001561743kin:ContractManufacturingMember2020-01-012020-12-310001561743kin:ContractManufacturingMember2019-01-012019-12-310001561743kin:ContractManufacturingMember2018-01-012018-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2020-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Memberus-gaap:MoneyMarketFundsMember2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2020-12-310001561743us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2020-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2020-12-310001561743us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Member2020-12-310001561743us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2020-12-310001561743us-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMember2020-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMember2020-12-310001561743us-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Member2020-12-310001561743us-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2020-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Memberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMemberus-gaap:FairValueInputsLevel3Member2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberkin:CorporateNoteMember2020-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberkin:CorporateNoteMember2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Memberkin:CorporateNoteMember2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberkin:CorporateNoteMemberus-gaap:FairValueInputsLevel3Member2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMember2020-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Member2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2020-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2019-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Memberus-gaap:MoneyMarketFundsMember2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2019-12-310001561743us-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMember2019-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMember2019-12-310001561743us-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Member2019-12-310001561743us-gaap:CommercialPaperMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2019-12-310001561743us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2019-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMember2019-12-310001561743us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Member2019-12-310001561743us-gaap:USTreasuryBillSecuritiesMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2019-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Memberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMemberus-gaap:FairValueInputsLevel3Member2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberkin:CorporateNoteMember2019-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberkin:CorporateNoteMember2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Memberkin:CorporateNoteMember2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberkin:CorporateNoteMemberus-gaap:FairValueInputsLevel3Member2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMember2019-12-310001561743us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel2Member2019-12-310001561743us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2019-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMemberus-gaap:USTreasuryBillSecuritiesMember2020-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2020-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMemberus-gaap:CorporateNoteSecuritiesMember2020-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMember2020-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodTwoMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2020-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodTwoMember2020-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMemberus-gaap:USTreasuryBillSecuritiesMember2019-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMemberus-gaap:CommercialPaperMember2019-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2019-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMemberus-gaap:CorporateNoteSecuritiesMember2019-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodOneMember2019-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodTwoMemberus-gaap:USGovernmentAgenciesDebtSecuritiesMember2019-12-310001561743us-gaap:CorporateNoteSecuritiesMemberkin:AvailableForSaleSecuritiesMaturityPeriodTwoMember2019-12-310001561743kin:AvailableForSaleSecuritiesMaturityPeriodTwoMember2019-12-3100015617432019-09-300001561743kin:TermLoanMemberkin:SolarCapitalLtd.Member2019-09-300001561743kin:TermLoanMemberkin:SolarCapitalLtd.Memberkin:LoanAgreementTermALoanMember2019-09-300001561743kin:LoanAgreementTermBLoanMemberkin:TermLoanMemberkin:SolarCapitalLtd.Member2019-09-300001561743kin:TermLoanMemberkin:LoanAgreementTermCLoanMemberkin:SolarCapitalLtd.Member2019-09-300001561743us-gaap:LondonInterbankOfferedRateLIBORMemberkin:TermLoanMemberkin:SolarCapitalLtd.Member2019-01-012019-09-300001561743kin:TermLoanMemberkin:SolarCapitalLtd.Member2019-01-012019-09-300001561743kin:TermLoanMemberkin:SolarCapitalLtd.Membersrt:MinimumMember2019-01-012019-09-300001561743kin:TermLoanMembersrt:MaximumMemberkin:SolarCapitalLtd.Member2019-01-012019-09-300001561743kin:TermLoanMembersrt:ScenarioForecastMemberkin:SolarCapitalLtd.Member2020-10-012021-09-300001561743kin:TermLoanMembersrt:ScenarioForecastMemberkin:SolarCapitalLtd.Member2021-10-012024-09-300001561743kin:TermLoanMemberkin:SolarCapitalLtd.Memberkin:LoanAgreementTermALoanMember2020-03-162020-03-160001561743kin:LoanAgreementTermBLoanMemberkin:TermLoanMemberkin:SolarCapitalLtd.Member2020-03-162020-03-160001561743kin:TermLoanMemberkin:LoanAgreementTermCLoanMemberkin:SolarCapitalLtd.Member2020-03-162020-03-160001561743kin:TermLoanMemberkin:LoanAgreementTermCLoanMemberkin:SolarCapitalLtd.Member2020-03-160001561743kin:TermLoanMemberkin:SolarCapitalLtd.Member2020-03-162020-03-160001561743kin:TermLoanMemberkin:SolarCapitalLtd.Member2020-12-230001561743us-gaap:ComputerEquipmentMember2020-12-310001561743us-gaap:ComputerEquipmentMember2019-12-310001561743us-gaap:FurnitureAndFixturesMember2020-12-310001561743us-gaap:FurnitureAndFixturesMember2019-12-310001561743us-gaap:LeaseholdImprovementsMember2020-12-310001561743us-gaap:LeaseholdImprovementsMember2019-12-310001561743us-gaap:BuildingMember2020-12-310001561743us-gaap:BuildingMember2019-12-310001561743us-gaap:BuildingImprovementsMember2020-12-310001561743us-gaap:BuildingImprovementsMember2019-12-310001561743us-gaap:LandMember2020-12-310001561743us-gaap:LandMember2019-12-310001561743us-gaap:LandImprovementsMember2020-12-310001561743us-gaap:LandImprovementsMember2019-12-310001561743us-gaap:ConstructionInProgressMember2020-12-310001561743us-gaap:ConstructionInProgressMember2019-12-31utr:sqft0001561743kin:StrategicVeterinaryPharmaceuticalsInc.Member2020-12-310001561743kin:StrategicVeterinaryPharmaceuticalsInc.Member2017-06-212017-06-21utr:acre0001561743us-gaap:SeriesAPreferredStockMember2020-12-310001561743kin:UndesignatedMember2020-12-31kin:vote0001561743us-gaap:EmployeeStockOptionMemberkin:EmployeeMemberus-gaap:CommonStockMember2018-01-012018-12-310001561743us-gaap:EmployeeStockOptionMemberkin:EmployeeMemberus-gaap:CommonStockMember2019-01-012019-12-310001561743us-gaap:EmployeeStockOptionMemberkin:EmployeeMemberus-gaap:CommonStockMember2020-01-012020-12-3100015617432018-01-012018-01-310001561743kin:AtTheMarketOfferingMember2018-05-310001561743kin:AtTheMarketOfferingMember2018-06-012018-06-300001561743kin:PublicStockOfferingMember2018-06-222018-06-220001561743kin:PublicStockOfferingMember2018-06-220001561743kin:PublicStockOfferingMember2019-01-232019-01-230001561743us-gaap:OverAllotmentOptionMember2019-01-232019-01-230001561743kin:PublicStockOfferingMember2019-01-2300015617432019-01-232019-01-2300015617432020-04-082020-04-080001561743kin:A2012EquityIncentivePlanMemberus-gaap:EmployeeStockOptionMember2012-11-042012-11-040001561743kin:A2012EquityIncentivePlanMember2012-11-040001561743kin:A2012EquityIncentivePlanMemberus-gaap:EmployeeStockOptionMember2020-12-310001561743us-gaap:EmployeeStockOptionMemberkin:A2016EquityIncentivePlanMember2016-05-310001561743us-gaap:EmployeeStockOptionMemberkin:A2016EquityIncentivePlanMember2016-05-012016-05-310001561743us-gaap:EmployeeStockOptionMemberkin:A2016EquityIncentivePlanMemberkin:OptioneeMember2016-05-310001561743kin:A2016EquityIncentivePlanMemberkin:A10StockOptionsMember2016-05-012016-05-310001561743us-gaap:EmployeeStockOptionMemberkin:A2016EquityIncentivePlanMembersrt:MinimumMember2016-05-012016-05-310001561743us-gaap:EmployeeStockOptionMemberkin:A2016EquityIncentivePlanMembersrt:MaximumMember2016-05-012016-05-310001561743us-gaap:EmployeeStockOptionMemberkin:A2016EquityIncentivePlanMember2020-12-310001561743kin:A2016EquityIncentivePlanMemberus-gaap:RestrictedStockMember2020-01-012020-12-310001561743us-gaap:RestrictedStockUnitsRSUMemberkin:A2016EquityIncentivePlanMember2020-12-310001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMember2018-06-300001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMember2020-06-152020-06-150001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMember2018-06-012018-06-300001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMemberkin:OptioneeMember2018-06-300001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMembersrt:MinimumMember2018-06-012018-06-300001561743us-gaap:EmployeeStockOptionMembersrt:MaximumMemberkin:A2018EquityIncentivePlanMember2018-06-012018-06-300001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMember2020-12-310001561743us-gaap:RestrictedStockUnitsRSUMemberkin:A2018EquityIncentivePlanMember2020-12-310001561743us-gaap:EmployeeStockMemberkin:A2014EmployeeStockPurchasePlanMember2014-12-310001561743us-gaap:EmployeeStockMemberkin:A2014EmployeeStockPurchasePlanMembersrt:MinimumMember2014-12-310001561743us-gaap:EmployeeStockMemberkin:A2014EmployeeStockPurchasePlanMembersrt:MaximumMember2014-12-310001561743us-gaap:EmployeeStockMemberkin:A2014EmployeeStockPurchasePlanMember2014-12-012014-12-310001561743us-gaap:EmployeeStockMemberkin:A2014EmployeeStockPurchasePlanMember2020-01-012020-12-310001561743us-gaap:EmployeeStockMemberkin:A2014EmployeeStockPurchasePlanMember2020-12-310001561743kin:A2014EmployeeStockPurchasePlanMemberus-gaap:CommonStockMember2020-01-012020-12-310001561743us-gaap:EmployeeStockMemberkin:A2014EmployeeStockPurchasePlanMember2019-12-310001561743kin:A2018EquityIncentivePlanMember2020-12-310001561743kin:A2014EmployeeStockPurchasePlanMember2020-12-3100015617432017-01-012017-12-310001561743kin:A2012EquityIncentivePlanMember2018-01-012018-12-310001561743kin:A2016EquityIncentivePlanMemberus-gaap:RestrictedStockMember2018-01-012018-12-310001561743us-gaap:RestrictedStockUnitsRSUMemberkin:A2016EquityIncentivePlanMember2018-01-012018-12-310001561743kin:A2016EquityIncentivePlanMember2018-01-012018-12-310001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMember2018-01-012018-12-310001561743kin:A2012EquityIncentivePlanMember2019-01-012019-12-310001561743kin:A2016EquityIncentivePlanMember2019-01-012019-12-310001561743kin:A2016EquityIncentivePlanMemberus-gaap:RestrictedStockMember2019-01-012019-12-310001561743us-gaap:RestrictedStockUnitsRSUMemberkin:A2018EquityIncentivePlanMember2019-01-012019-12-310001561743kin:A2012EquityIncentivePlanMember2020-01-012020-12-310001561743kin:A2016EquityIncentivePlanMember2020-01-012020-12-310001561743us-gaap:RestrictedStockUnitsRSUMemberkin:A2018EquityIncentivePlanMember2020-01-012020-12-310001561743kin:A2018EquityIncentivePlanMemberus-gaap:RestrictedStockMember2020-01-012020-12-310001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMember2020-01-012020-12-310001561743us-gaap:RestrictedStockUnitsRSUMember2020-01-012020-12-310001561743us-gaap:RestrictedStockMember2018-01-232018-01-230001561743us-gaap:RestrictedStockUnitsRSUMember2018-01-222018-01-220001561743us-gaap:RestrictedStockMember2019-01-232019-01-230001561743us-gaap:RestrictedStockMember2019-01-222019-01-220001561743kin:A2018EquityIncentivePlanMemberus-gaap:RestrictedStockMember2019-01-012019-03-310001561743us-gaap:RestrictedStockMember2020-01-232020-01-230001561743us-gaap:RestrictedStockMember2020-07-312020-07-310001561743us-gaap:RestrictedStockUnitsRSUMember2020-01-222020-01-220001561743us-gaap:RestrictedStockUnitsRSUMember2020-07-312020-07-310001561743us-gaap:EmployeeStockOptionMemberkin:A2018EquityIncentivePlanMember2020-06-012020-06-300001561743us-gaap:CommonStockMember2020-01-012020-12-310001561743us-gaap:CommonStockMember2019-01-012019-12-310001561743us-gaap:CommonStockMember2018-01-012018-12-310001561743us-gaap:RestrictedStockUnitsRSUMember2018-01-220001561743us-gaap:RestrictedStockUnitsRSUMember2019-01-012019-03-310001561743us-gaap:RestrictedStockMember2019-01-012019-03-310001561743us-gaap:RestrictedStockUnitsRSUMember2020-01-012020-03-310001561743us-gaap:RestrictedStockUnitsRSUMember2020-07-012020-07-310001561743us-gaap:RestrictedStockMember2020-12-310001561743us-gaap:RestrictedStockMember2020-01-012020-12-310001561743us-gaap:RestrictedStockMember2019-12-310001561743us-gaap:ResearchAndDevelopmentExpenseMember2020-01-012020-12-310001561743us-gaap:ResearchAndDevelopmentExpenseMember2019-01-012019-12-310001561743us-gaap:ResearchAndDevelopmentExpenseMember2018-01-012018-12-310001561743us-gaap:GeneralAndAdministrativeExpenseMember2020-01-012020-12-310001561743us-gaap:GeneralAndAdministrativeExpenseMember2019-01-012019-12-310001561743us-gaap:GeneralAndAdministrativeExpenseMember2018-01-012018-12-310001561743us-gaap:EmployeeStockOptionMember2020-01-012020-12-310001561743us-gaap:EmployeeStockOptionMember2019-01-012019-12-310001561743us-gaap:EmployeeStockOptionMember2018-01-012018-12-310001561743us-gaap:EmployeeStockMember2020-01-012020-12-310001561743us-gaap:EmployeeStockMember2019-01-012019-12-310001561743us-gaap:EmployeeStockMember2018-01-012018-12-310001561743us-gaap:RestrictedStockUnitsRSUMember2019-01-012019-12-310001561743us-gaap:RestrictedStockUnitsRSUMember2018-01-012018-12-3100015617432020-07-312020-07-310001561743us-gaap:PropertySubjectToOperatingLeaseMemberus-gaap:BuildingMember2020-02-290001561743kin:OfficeSpaceMemberus-gaap:PropertySubjectToOperatingLeaseMember2015-08-310001561743kin:OfficeSpaceMemberus-gaap:PropertySubjectToOperatingLeaseMember2020-11-300001561743kin:OfficeSpaceMemberus-gaap:PropertySubjectToOperatingLeaseMember2020-09-300001561743kin:OfficeSpaceMemberus-gaap:PropertySubjectToOperatingLeaseMember2019-05-31kin:numberOfLeases00015617432018-06-012018-06-3000015617432018-06-300001561743us-gaap:DomesticCountryMember2020-12-310001561743us-gaap:StateAndLocalJurisdictionMember2020-12-310001561743us-gaap:DomesticCountryMemberus-gaap:TaxYear2017Member2020-12-310001561743us-gaap:DomesticCountryMemberus-gaap:ResearchMember2020-12-310001561743us-gaap:StateAndLocalJurisdictionMemberus-gaap:ResearchMember2020-12-310001561743us-gaap:EmployeeStockOptionMember2020-01-012020-12-310001561743us-gaap:EmployeeStockOptionMember2019-01-012019-12-310001561743us-gaap:EmployeeStockOptionMember2018-01-012018-12-310001561743us-gaap:RestrictedStockMember2020-01-012020-12-310001561743us-gaap:RestrictedStockMember2019-01-012019-12-310001561743us-gaap:RestrictedStockMember2018-01-012018-12-310001561743us-gaap:RestrictedStockUnitsRSUMember2020-01-012020-12-310001561743us-gaap:RestrictedStockUnitsRSUMember2019-01-012019-12-310001561743us-gaap:RestrictedStockUnitsRSUMember2018-01-012018-12-310001561743srt:MaximumMember2020-01-012020-12-310001561743us-gaap:SubsequentEventMember2021-01-012021-03-10
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
______________________
FORM 10-K
______________________
| | | | | |
| (Mark one) |
☒
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| For the fiscal year ended December 31, 2020 |
| OR |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| For the transition period from _____ to ______ |
Commission file number: 001-36225
KINDRED BIOSCIENCES, INC.
(Exact name of registrant as specified in its charter)
___________________
| | | | | | | | |
| Delaware | | 46-1160142 |
| (State or other jurisdiction of incorporation or organization) | | (I.R.S. Employer
Identification Number) |
1555 Bayshore Highway, Suite 200
Burlingame, California 94010
(Address of principal executive offices) (Zip code)
(650) 701-7901
(Registrant's telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act: | | | | | | | | | | | | | | |
| Title of each class | | Trading Symbol | | Name of each exchange on which registered |
| Common Stock, $0.0001 par value | | KIN | | The NASDAQ Stock Market LLC |
| Preferred Stock Purchase Rights | | KIN | | The NASDAQ Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act: None.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | | | | |
| Large accelerated filer | ☐ | | Accelerated filer | ☐ |
| Non-accelerated filer | ☒ | | Smaller reporting company | ☒ |
| | | Emerging growth company
| ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. Yes ☐ No ☒
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act).
Yes ☐ No ☒
On June 30, 2020, (the last business day of the registrant’s most recently completed second fiscal quarter), the aggregate market value of the common stock of the registrant held by non-affiliates of the registrant was approximately $136.3 million.
The outstanding number of shares of the registrant’s common stock as of March 10, 2021 was 41,449,218.
DOCUMENTS INCORPORATED BY REFERENCE
Certain portions of the registrant’s Proxy Statement for the 2021 annual meeting of stockholders, to be filed with the Securities and Exchange Commission pursuant to Regulation 14A not later than 120 days after the close of the registrant’s fiscal year, are incorporated by reference into Part III of this Form 10-K.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995. All statements contained in this Annual Report that do not relate to matters of historical fact should be considered forward-looking statements, including, but not limited to, statements regarding our expectations about the trials, regulatory approval, manufacturing, distribution and commercialization of our current and future products and product candidates, and statements regarding our anticipated revenues, expenses, margins, profits and use of cash. These forward-looking statements are based on our current expectations. We have identified some of these forward-looking statements with words such as “believe,” “may,” “will,” “should,” “could,” “expect,” “intend,” “plan,” “predict,” “anticipate,” “estimate,” “continue” or other words and terms of similar meaning and the use of future dates. These statements are not promises or guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results to be materially different from any future results expressed or implied by the forward-looking statements. These risks and uncertainties include, but are not limited to, the following:
•our limited operating history and expectations of losses for the foreseeable future;
•the absence of significant revenue from our products and our product candidates for the foreseeable future;
•the likelihood that our revenue will vary from quarter to quarter;
•our potential inability to obtain any necessary additional financing;
•our substantial dependence on the success of our products and our lead product candidates which may not be successfully commercialized even if they are approved for marketing;
•the effect of competition;
•our potential inability to obtain regulatory approval for our existing or future product candidates;
•our dependence on third parties to conduct some of our development activities;
•our dependence upon third-party manufacturers for supplies related to our products and our product candidates and the potential inability of these manufacturers to deliver a sufficient amount of supplies on a timely basis;
•the uncertain effect of the COVID-19 pandemic on our business, results of operations and financial condition;
•uncertainties regarding the outcomes of trials regarding our product candidates;
•our potential failure to attract and retain senior management and key scientific personnel;
•uncertainty about our ability to enter into satisfactory agreements with third-party licensees of our biologic products and uncertainty about the amount of revenue that we will receive from such agreements;
•our significant costs of operating as a public company;
•potential cyber-attacks on our information technology systems or on our third-party providers’ information technology systems, which could disrupt our operations;
•our potential inability to repay the secured indebtedness that we have incurred from third-party lenders, and the restrictions on our business activities that are contained in our loan agreement with these lenders;
•the risk that our 2020 strategic realignment and restructuring plans will result in unanticipated costs or revenue shortfalls;
•uncertainty about the amount of royalties that we will receive from the sale of Mirataz® to Dechra Pharmaceuticals PLC;
•the risk that the revenue from our delivery of services or products under any contract may be less than we anticipate if the other party to the contract exercises its right to terminate the contract prior to the completion of the contract or if such party is unable or unwilling to satisfy its payment obligations under the contract;
•our potential inability to obtain and maintain patent protection and other intellectual property protection for our products and our product candidates;
•potential claims by third parties alleging our infringement of their patents and other intellectual property rights;
•our potential failure to comply with regulatory requirements, which are subject to change on an ongoing basis;
•the potential volatility of our stock price; and
•the significant control over our business by our principal stockholders and management.
For a further description of these risks and other risks that we face, please see the risk factors described in Item 1A of this Annual Report under the caption “Risk Factors” and any subsequent updates that may be contained in our Quarterly Reports on Form 10-Q and other documents we file with the Securities and Exchange Commission (the “SEC”). As a result of the risks described above and in our filings with the SEC, actual results may differ materially from those indicated by the forward-looking statements made in this Annual Report. Forward-looking statements contained in this Annual Report speak only as of the date of this Annual Report, and we undertake no obligation to update or revise these statements, except as may be required by law.
PART I.
ITEM 1. BUSINESS.
Overview
We are a biopharmaceutical company developing innovative biologics focused on saving and improving the lives of pets. Our mission is to bring to pets the same kinds of safe and effective medicines that our human family members enjoy. Our core strategy is to identify targets that have already demonstrated safety and efficacy in humans and to develop therapeutics based on these validated targets for dogs and cats. We believe that this approach will lead to shorter development times and higher approval rates than pursuing new, non-validated targets. Our current portfolio includes over 20 product candidates in development, predominantly biologics. We also have state-of-the-art biologics manufacturing capabilities and a broad intellectual property portfolio.
Our first product, Mirataz® (mirtazapine transdermal ointment) was approved in May 2018 and became commercially available to veterinarians in the United States in July 2018. In November 2019, our second product, Zimeta™ (dipyrone injection) for the control of fever in horses was approved by the Food and Drug Administration (FDA) and became commercially available in December 2019. In addition, we have a portfolio of other product candidates, predominantly biologics, in various stages of development.
On March 16, 2020, we announced we will further prioritize biologics programs for dogs and cats and rely primarily on a partnership-focused commercialization model, which is expected to significantly reduce the amount of additional dilutive capital the company will require. We believe monoclonal antibodies are the future of veterinary medicine, and represent the greatest opportunity for value creation, given large potential markets for our programs and our competitive advantage in biologics. As part of the strategic realignment, we substantially reduced our commercial footprint and discontinued development of most canine and feline small molecule programs. This business model is expected to achieve a better return for our stockholders.
We were incorporated in Delaware in September 2012. The address of our principal executive offices is 1555 Bayshore Highway, Suite 200, Burlingame, CA 94010. Unless the context requires otherwise, references to “KindredBio,” “the Company,” “we,” “us” or “our” in this Annual Report on Form 10-K for the fiscal year ended December 31, 2020 (the “2020 Annual Report”) refer to Kindred Biosciences, Inc., a Delaware corporation, and its subsidiaries.
Recent Milestones
Product Highlights


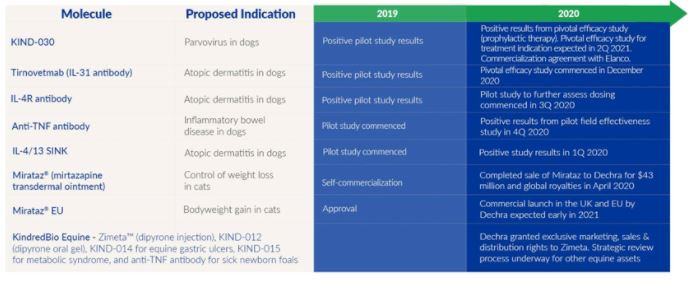
On March 16, 2020, we signed an agreement to sell Mirataz® (mirtazapine transdermal ointment), our transdermal drug for the management of weight loss in cats, to Dechra Pharmaceuticals PLC ("Dechra") for a cash purchase price of $43 million, of which $38.7 million was paid on the closing date and $4.3 million will be paid out of escrow beginning in 12 months assuming no escrow claims, alongside an ongoing royalty on global net sales. The acquisition comprises worldwide marketing rights, intellectual property rights, marketing authorizations and associated regulatory documentation, third party supply contracts related to raw material and manufacture of the finished product, and certain product inventory. With commercial sales and marketing teams in 25 countries, and distributor relationships in an additional 72 countries, Dechra is strongly positioned to market Mirataz in the United States, Europe, and globally. Their focus on the sale of technical and value-added specialty pharmaceuticals has led to the development of market-leading brands in the specialty veterinarian sector, particularly within the field of chronic disease management. With a complementary feline product portfolio targeting diseases linked to feline weight loss, Mirataz will represent an important cross-promotional product for Dechra worldwide.
Mirataz is the first and only FDA approved transdermal medication specifically developed for the management of weight loss in cats. Unintended weight loss is a serious unmet medical need in cats, and may be caused by multiple factors, including chronic illness, like chronic kidney disease, or behavioral issues, such as stress. If untreated, it may lead to hepatic lipidosis, which can be a life-threatening condition.
On December 12, 2019, KindredBio announced that the European Commission granted marketing authorization of Mirataz for bodyweight gain in cats experiencing poor appetite and weight loss resulting from chronic medical conditions. Mirataz is the first and only medication approved in the EU to induce bodyweight gain in cats experiencing poor appetite and weight loss resulting from chronic medical conditions. Europe represents the second largest market for veterinary therapeutics internationally. The authorization is valid in all 28-member states of the European Union, together with Iceland, Liechtenstein, and Norway, and including the UK. Dechra, which is based in the United Kingdom, plans to launch Mirataz in the UK and the European Union, and intends to conduct the necessary regulatory activities to achieve approvals in other key international markets.
We recorded $822,000 in Mirataz net product revenues in 2020, which reflects our product sales up through the date of the sale of Mirataz to Dechra on March 16, 2020. This compares with net product revenues of $4.1 million in 2019 and $2.0 million in 2018, when the product was launched in July of that year. Mirataz royalty revenue for the year ended 2020 was $535,000.
ZimetaTM (dipyrone injection) (Zimeta IV)
On November 25, 2019 KindredBio announced that the U.S. Food and Drug Administration approved Zimeta™ (dipyrone injection) for the control of pyrexia in horses. Pyrexia, or fever, is associated with a number of underlying diseases and can result in significant negative outcomes, including dehydration, laminitis, muscle wasting, weight loss, and in some cases death. Zimeta is the first injectable dipyrone product to receive FDA approval for use in horses. Dipyrone, the active ingredient in Zimeta, is a member of the non-steroidal anti-inflammatory drug (NSAID) class and has a centrally acting mechanism of action on the hypothalamus where fever originates and is regulated.
As part of the strategic restructuring announcement on March 16, 2020 and our further prioritization on biologics programs for dogs and cats, we evaluated the potential spin-out or divestiture of our equine assets. In December 2020, we entered into a Distribution and Licensing Agreement granting Dechra Veterinary Products LLC (Dechra), a corporation registered in Delaware, an exclusive license under our Patents and Marketing Authorizations to promote, market, sell and distribute Zimeta in the territory (US and Canada). We are responsible for the performance of our third party manufacturer for Zimeta, including the performance of all duties and including but not limited to, timely delivery of product meeting the applicable specifications. In consideration for the exclusive license and manufacturing services to be performed by us, Dechra will pay KindredBio a milestone payment upon achievement of a certain sales milestone, a royalty fee and a price per unit of Zimeta.
For the year ended December 31, 2020, we recorded $27,000 in Zimeta net product revenues compared to $127,000 in 2019, when the product was launched in December of that year.
Biologic Product Candidates
On March 16, 2020, we announced we will further prioritize our biologics programs for dogs and cats, which we view as our highest potential assets.
KIND-016, Tirnovetmab (Interleukin-31)
In October 2018, we announced positive topline results from our pilot laboratory effectiveness study of tirnovetmab, KIND-016, a fully caninized, high-affinity monoclonal antibody targeting interleukin-31 (IL-31), for the treatment of atopic dermatitis in dogs. In addition, we announced that the U.S. Patent and Trademark Office has issued a patent (Patent No. 10,093,731) for KindredBio's anti-IL31 antibody.
In July 2019, we reported positive topline results from a pilot field effectiveness study for our IL-31 antibody that confirmed the results from our pilot laboratory study. The manufacturing scale up process proceeded and the pivotal efficacy study of KIND-016 was initiated in December 2020.
Canine atopic dermatitis is an immune-mediated inflammatory skin condition in dogs and is the leading reason owners take their dog to the veterinarian. Atopic dermatitis is a large market, with the leading two products on the market selling over $900 million per year. We are pursuing a multi-pronged approach toward atopic dermatitis, with a portfolio of promising biologics. Our market research tells us there is strong demand for new biological treatments for pruritic dogs, with 70% of veterinarians, and a higher percentage of dermatologists, expressing a need for alternatives to current therapies.
KIND-032
In December 2019, we announced the outcome of a positive pilot laboratory study of KIND-032, a fully caninized monoclonal antibody targeting interleukin-4 (IL-4) receptor, for the treatment of atopic dermatitis in dogs. In the study, 14 laboratory dogs with clinical signs consistent with atopic dermatitis were dosed with placebo or with KIND-032 at two different doses. The Canine Atopic Dermatitis Extent and Severity Index (CADESI) scores were assessed by board-certified veterinary dermatologists who were blinded to treatment assignments. The study demonstrated that KindredBio's antibody was well-tolerated. Although the
study was a single-dose study designed primarily to assess safety and pharmacokinetics, evidence of positive efficacy and dose response was observed at Week 1, as measured by CADESI-04. A second pilot study to further assess dosing commenced in the third quarter of 2020.
The IL-4 pathway is a key driver of the inflammation that underlies atopic dermatitis and several other allergic diseases. Unlike KIND-025, which binds to IL-4 and IL-13 circulating in blood, KIND-032 binds to the IL-4 receptor on the surface of immune cells.
KIND-025
On March 24, 2020, we announced positive results from our pilot field efficacy study of KIND-025, a canine fusion protein targeting IL-4 and IL-13, for the treatment of atopic dermatitis in dogs. A higher treatment success rate was observed in the KIND-025 group over the placebo group from week 1 through week 4. Positive efficacy signals were also detected with other endpoints including 20mm or higher reduction from baseline in PVAS score. Cell line development is being continued as we further evaluate this program. The IL-4 and IL-13 pathways are key drivers of the inflammation that underlies atopic dermatitis and other allergic diseases. The IL-4/13 SINK molecule binds to both IL-4 and IL-13 circulating in the blood and inhibits their interactions with their respective receptors, thereby modifying the clinical signs associated with atopic dermatitis. We currently do not have plans to prioritize KIND-025 ahead of our other programs.
KIND-030
In August 2019, we announced positive results from our pilot efficacy study of KIND-030, a chimeric, high-affinity monoclonal antibody targeting canine parvovirus (CPV). This was a 12-dog study, of which 4 dogs were treated prophylactically and 2 dogs were treated after establishment of the infection. All treated dogs survived, compared to none in the applicable placebo group. The effect was seen in both prophylaxis setting, as well as in a treatment setting after establishment of infection. On September 16, 2020, we reported positive results from our pivotal efficacy study of KIND-030 in prevention of parvovirus infection in prophylactic treatment. In the randomized, blinded, placebo-controlled study, KIND-030 was administered to dogs as prophylactic therapy to prevent clinical signs of CPV infection. The primary objectives of the study were met. All of the placebo-control dogs developed parvovirus infection as predefined in the study protocol, while none of the KIND-030 treated dogs developed the disease. Furthermore, the parvovirus challenge resulted in 60% mortality rate in the control dogs compared to 0% mortality rate in the KIND-030 treated dogs.
KIND-030 is being pursued for two indications in dogs: prophylactic therapy to prevent clinical signs of canine parvovirus infection and treatment of established parvovirus infection. The pivotal efficacy study for the treatment indication is expected to be completed in the second quarter of 2021. There is no set review timeline at the United States Department of Agriculture Center for Veterinary Biologics. Regulatory approval and review timeline are subject to the typical risks inherent in such a process.
CPV is the most significant cause of viral enteritis in dogs, especially puppies, with a mortality rate reportedly as high as 91% if untreated. A 2014 Banfield State of Pet Health report noted at least 250,000 dogs are infected with parvoviruses each year, excluding emergency hospitals, shelters, specialty hospitals or undiagnosed cases. While there are vaccines available for CPV, they have to be administered multiple times and many puppies do not receive the vaccine at all, or do not receive the complete series. This will not replace the need for vaccination; it may just change the timing of the vaccination post administration. There are currently no approved or unapproved treatments for CPV. Currently, owners spend up to thousands of dollars for supportive care for dogs infected with CPV.
In December 2020, we announced an agreement granting Elanco Animal Health, Inc. ("Elanco") exclusive global rights to KIND-030. Under the terms of the agreement, KindredBio will receive an upfront payment of $500,000, development milestone payments of up to $16 million upon achievement of certain development, regulatory and manufacturing targets, and sales milestones in an aggregate amount of up to $94
million payable throughout the term of the agreement. Furthermore, royalty payments are to range from the low to high teens. The agreement specifies that KindredBio will supply the licensed product to Elanco, and that Elanco will conduct the necessary regulatory activities to achieve approvals in Europe and other key international markets.
KIND-509
On December 21, 2020, we announced positive results from the pilot field efficacy study of our monoclonal antibody against tumor necrosis factor alpha (anti-TNF antibody) for canine inflammatory bowel disease (IBD). The study was a randomized, blinded, placebo-controlled pilot effectiveness study that enrolled 10 dogs diagnosed with IBD to assess the efficacy and safety of KindredBio's anti-TNFα antibody over a 4-week treatment period. The primary effectiveness variable for this exploratory study was reduction in Canine Inflammatory Bowel Disease Activity Index (CIBDAI) score, which was assessed at Screening and Days 0, 7, 14, 21 and 28. Complete remission, defined as ≥ 75% reduction in average post-dose CIBDAI score from baseline, was achieved in 75% of the anti-TNFα group compared to 17% in the placebo group. The treatment effect was early-onset and durable. At Day 7, the first post-dose visit, 75% of the anti-TNFα treated dogs showed ≥ 75% reduction of CIBDAI score from baseline, compared to 17% in the placebo group. Furthermore, 50% of the anti-TNFα treated dogs achieved and maintained 100% reduction of CIBDAI score from baseline throughout all post-dose visits, whereas none in the placebo group achieved the same result.
IBD is a chronic disease of the gastrointestinal tract and can affect dogs at any age, but is more common in middle-aged and older dogs. The majority of canine IBD cases involve chronic states of diarrhea, vomiting, gastroenteritis, inappetence, and other symptoms, certain of which are cited as among the most frequent disorders impacting dogs. For certain dog breeds, the prevalence of diarrhea exceeds 5%. Existing treatments can have significant drawbacks, including limited diets and excessive antibiotic use, which can lead to owner frustration, lapses in treatment adherence, or poor quality of life for the affected animal.
KIND-510a
In January 2019, we announced positive topline results from our pilot field effectiveness study of KIND-510a, a long-acting feline recombinant erythropoietin being developed for the management of non-regenerative anemia in cats. It has been engineered by KindredBio to have a prolonged half-life compared to endogenous erythropoietin, a protein that regulates and stimulates production of red blood cells. The pivotal efficacy study for KIND-510a was initiated in the fourth quarter of 2019.
On November 25, 2020, KindredBio made the decision to suspend the feline recombinant erythropoietin program following an adverse event. Patient safety is paramount to KindredBio. Given the impact of safety profile on the program's commercial value, and amid the continued rapid growth of the canine dermatitis market, the decision was made to redirect resources toward accelerating KindredBio's programs for canine atopic dermatitis. KindredBio maintains backup programs for its feline recombinant erythropoietin and may seek to develop these at a later date.
Anemia is a common condition that is estimated to affect millions of older cats. It is often associated with chronic kidney disease, because kidneys produce erythropoietin and chronic kidney disease leads to decreased levels of endogenous erythropoietin. Chronic kidney disease affects approximately half of older cats, making it a leading cause of feline mortality. Human erythropoietins, which are multi-billion-dollar products in the human market, are immunogenic in cats.
KIND-511
KIND-511 is an anti-Tumor Necrosis Factor (“anti-TNF”) treatment for newborn foals. Sick newborn foals, defined as sepsis score ≥ 11 or positive blood culture, are challenging, and difficult to treat and result in approximately 50% mortality. We have completed a pilot field study in sick or septic foals to assess safety and
efficacy of anti-TNF monoclonal antibody, with positive results. By Kaplan-Meier analysis, the difference in survival between the control and placebo groups was statistically significant (p=0.0293). There is currently no FDA-approved therapy. We have placed this program on indefinite hold to focus on accelerating our late-stage biologic candidates in the canine and feline markets.
We also have promising new antibody candidates under development, which will be disclosed at a later stage of development and as lead programs further advance.
Small Molecule Product Candidates
On March 16, 2020, KindredBio announced the discontinuation of the development of canine and feline small molecule candidates as we prioritize our pipeline of innovative biologic candidates.
We completed a strategic review of our equine candidates. In December 2020, we entered into a Distribution and Licensing Agreement granting Dechra an exclusive license under our Patents and Marketing Authorizations to promote, market, sell and distribute Zimeta in the US and Canadian territories in return for a royalty on sales and milestone payment upon achievement of a certain sales milestone. We made the decision to discontinue the development of KIND-012 (dipyrone oral gel) and pause the development of KIND-014 to focus on accelerating our late-stage biologic candidates.
Market Overview
We believe there are significant unmet medical needs for pets, and that the pet therapeutics segment of the animal health industry is likely to grow substantially as new therapeutics are identified, developed and marketed specifically for pets. We plan to commercialize our feline and canine biologics in the United States through commercial partnerships, and in the European Union (the “EU”) primarily through commercial partnerships, distributors and other third parties.
Relative to human drug development, the development of pet therapeutics is generally faster and less expensive, since it requires fewer clinical studies involving fewer subjects and can be conducted directly in the target species. For example, studies that are typically required for approval of human drugs such as QTC studies, which detect cardiac irregularities, elderly patient studies, renal impairment studies, hepatic impairment studies or costly, long-term genotoxicity studies are not required for pet therapeutics. Based on our progress since inception in September 2012, we believe we can develop biologics in around six years at an average cost of approximately $8 million. The lower cost associated with the development of pet therapeutics permits us to pursue multiple product candidates simultaneously and avoid the binary outcome associated with the development of a single lead therapy by some human biotechnology companies. Because our strategy is to identify targets that have already demonstrated safety and efficacy in humans and to develop therapeutics based on these validated targets, we can often advance our programs more rapidly than if we were pursuing unvalidated targets. Biologics could be submitted under USDA, FDA or EMA regulatory authorities for approval.
We estimate that the total U.S. market for veterinary care was approximately $99.0 billion in 2020, of which veterinary care and product sales comprised $30.2 billion. According to the 2019-2020 American Pet Products Association (APPA) National Pet Owners Survey, 67% of households owned a pet, which equates to 84.9 million homes. It is estimated that there are 97.0 million dogs and 76.0 million cats currently living in the United States. We believe there are many unmet or underserved medical needs and that the pet therapeutics portion of the market can grow significantly as new, safe and effective therapeutics are identified, developed and marketed. We expect continued market growth as new pet therapeutics are developed and owners grow more familiar with the treatment of pets with such therapeutics.
The equine sector shares many similarities with the orphan human market. There are fewer horses than dogs or cats, but the willingness to pay is substantially higher.
Management Team
Our small molecule approvals at both the FDA and EMA jurisdiction and the progress of the biologicals portfolio are testament to the management team’s extensive experience in both human and animal drug development.
Richard Chin, M.D., our Chief Executive Officer, was previously Head of Clinical Research for the Biotherapeutics Unit at Genentech, Inc., where he oversaw Phase I through Phase IV clinical programs for all products except oncology. Wendy Wee, our Chief Financial Officer, has over 30 years of experience and most recently was Vice President of Finance and Principal Accounting Officer at Telik, Inc. Hangjun Zhan, Ph.D., our Chief Scientific Officer, is a well-established protein biochemist and biophysicist with over 20 years of drug discovery experience in the biotechnology industry. Ms. Wee recently announced her intention to retire as our Chief Financial Officer on August 31, 2021, and we are in the process of identifying a new Chief Financial Officer.
Product Pipeline
As of March 16, 2021, our product pipeline is focused on innovative biologics for dogs and cats.
The US Department of Agriculture’s (USDA) Center for Veterinary Biologics and the FDA’s Center for Veterinary Medicine have a memorandum of understanding under which animal products are to be regulated by the USDA as biologics, if they are intended for use to diagnose, cure, mitigate, treat, or prevent disease in animals and they work primarily through an immune process, or by the FDA as drugs, if they are intended for use in the diagnosis, cure, mitigation, treatment, or prevention of animal disease if the primary mechanism of action is not immunological or is undefined. Although we believe that most of our current animal biologics will be regulated by the USDA based on their mechanisms of action, certain of our animal biologics will be regulated by the FDA instead of the USDA.
We are currently pursuing over 20 indications with a focus on biologics product candidates. The following table illustrates some of the product candidates that we are developing.
Product Selection and Development
We utilize a rigorous screening and review process to identify targets that have demonstrated safety and efficacy in humans and address unmet medical needs in veterinary medicine. In some cases, we identify a chemical or functional equivalent of a validated human drug that addresses the same biological target or pathway. We review these targets with a view to differentiating them from existing treatments, including human products used extra-label in animals, based on ease of administration, method of delivery, dosing regimen, and other similar factors.
Biologic therapies are typically derived from living organisms. A biologic can be defined as a large complex molecule (nucleic acid and protein platforms) produced from or extracted from a biological or living system. They are made by genetically engineering living cells, and a high level of precision is required in the manufacturing process to produce a consistent biologic product each time. A biologic product can be a monoclonal antibody, a vaccine, a tissue, or various proteins such as cytokines, enzymes, fusion proteins, whole cells, and viral and non-viral gene therapies. Our biologic product candidates are usually based on therapies and targets for which products have been successfully commercialized for humans. Human antibody therapies are expensive and are often ineffective in other species since they are usually immunogenic or recognized as foreign bodies and rejected by the immune systems of dogs, cats, horses, and other animals. We identify or create biologics, including antibodies, that are fully or mostly canine or feline. We generally intend to seek composition-of-matter and other patents for these new biologics.
In January 2020, we announced that we developed a technology to extend canine antibody half-life by up to three-fold. A patent application for this technology has been filed.
The study comprised 12 dogs, including four groups with various modifications incorporating KindredBio technology and one wild type canine antibody as control. Half-life extension was observed in all dogs across all groups other than the wild type, with the magnitude of extension over native antibody ranging from two to three-fold.
Half-life extension technologies have the potential to improve therapeutic performance in numerous ways. Reduced dosing frequency and/or amount of dosing can lead to improved pet owner convenience and compliance. It can also substantially lower the cost of goods and enhance profitability and market positioning. In addition, higher drug concentration using the same dose and dosing interval as the parent antibody can result in extended drug exposure and potentially improved efficacy.
In addition, KIND-Bodies, a unique biologics scaffold with certain advantages over traditional monoclonal antibodies, including bi-specific binding, is under development. We have also developed Fc engineering technologies that can improve affinity of canine antibodies to protein A, which is important for manufacturing of antibodies, and other technologies to modify the immune function of antibodies, and we have filed for IP on those technologies.
We have an in-house laboratory capable of protein engineering, cell line development, analytics, and other activities necessary for advancing a world-class biologics pipeline. We believe that we have one of the best biologics teams in the pharmaceutical industry, drawn from some of the top biotechnology companies.
Our biologic product candidates are not expected to face generic competition in the United States as there is no pathway for approval of a generic veterinary biologic regulated by the USDA.
Contract Manufacturing
In May 2020, we entered into an agreement with Vaxart, Inc. for the manufacture of Vaxart's oral vaccine candidate for COVID-19. We recorded contract manufacturing revenue based on the percentage completion of specific milestones for the year ended December 31, 2020. In October 2020, we announced the expansion of our manufacturing agreement with Vaxart for COVID-19 and other vaccine candidates. In addition, we will begin manufacturing, formulation and analytical services for the National Cancer Institute's PREVENT Cancer Preclinical Drug Development Program in 2021. KindredBio was selected as one of the three companies that were awarded the NCI cGMP manufacturing pool contract in 2019.
We have constructed a state-of-the-art manufacturing plant in Burlingame, California for our initial biologic product candidates, which we believe is one of the first GMP biologics plants for veterinary products. We started GMP manufacturing in January 2018 and believe that the plant will position us as a leader in the veterinary biologics field, and potentially afford us an advantage in cost of goods for our products. We
acquired a second manufacturing plant in August 2017 in Elwood, Kansas and construction to support initial production lines on our biologics manufacturing was completed in 2019. The bioreactors and fill & finish equipment are fully commissioned. The Elwood facility includes approximately 180,000 square feet with clean rooms, utility, equipment, and related quality documentation suitable for small molecule and biologics manufacturing.
Business Strategy
Key elements of our business strategy are as follows:
Continue to focus on the development of our pipeline
In addition to our focus on patient enrollment for our pivotal field study of KIND-016 and completing our pivotal treatment efficacy study of KIND-030, we expect to initiate a field pilot studies for KIND-032 and KIND-509 in 2021. Additionally, we are also developing multiple other products. In all, we have over 20 programs for various indications.
Continue to focus on cost-effective research and development execution
In order to execute our studies rapidly and efficiently, we have built an experienced team drawn from both the veterinary and human pharmaceutical industries. We rely primarily on our own personnel or independent contractors, rather than on contract research organizations (“CROs”), for many business-critical tasks, including protocol designs, regulatory interactions, statistics, data management and clinical operations. By doing so, we believe we can maintain higher quality, achieve lower costs and seek regulatory approval more quickly.
Leverage our antibody and biologics experience
Members of our team have extensive experience developing biologics such as antibodies. We are leveraging their expertise to identify and develop antibody-based therapies for pets based on approved human therapies, and to identify appropriate manufacturing technologies for these product candidates.
Leverage our current product pipeline in additional animal species
We intend to develop our product candidates primarily for approval in one or more indications in predominantly dogs and cats. We may consider the development of our current or future product candidates for additional species in the future.
Expand our pipeline with additional product candidates
We identify biologic therapeutics, including therapeutic targets, that have demonstrated safety and efficacy in humans. We discover the biologics that are specific to canines or felines. We have already identified a number of additional product candidates in the pre-INAD stage that we may potentially pursue. We continuously seek to protect our product candidates through a combination of patents, know-how and other customary means. It is worth noting that there is no biosimilar pathway for veterinary medicine currently.
Commercialize our canine and feline products through commercial partners in the United States and other regions
As disclosed in our March 16, 2020 press release, KindredBio is transitioning to a partnership-focused commercialization model whereby pipeline assets are out-licensed to larger commercial partners that can maximize product opportunity in return for upfront payments, contingent milestones, and royalties on future sales. Accordingly, our companion animal commercial infrastructure was dismantled in mid-2020.
Commercialize our equine products through distribution and licensing agreements with partners
We executed a distribution and licensing agreement with Dechra to commercialize Zimeta. While KindredBio's current development activities are predominantly focused on biologics for dogs and cats, select distributor relationships may be used to market other equine products directly to veterinarians in the United States. We also intend to establish collaborations with distributors or licensing partners to commercialize any products that may be approved by the EMA.
Fund development of our pipeline through contract manufacturing agreements
We have set up a full-service contract development and manufacturing organization, Centaur Biopharmaceutical Services, Inc., proceeds of which are used to fund development of our companion animal product pipeline. Centaur's capabilities span production and process development, analytical development and process characterization, and leverage our manufacturing facilities in California and Kansas.
Pet Therapeutics Market
Overview
U.S. consumers spent an estimated $99.0 billion on their pets in 2020, according to the APPA. The veterinary care segment has been among the fastest growing segments of the overall U.S. pet market. This segment accounted for an estimated $30.2 billion spent on veterinary care in 2020.
Researchers who specialize in studying the pet industry have noted its constant rise and recession-proof nature. Specifically, American pet spending has continued to rise each year since 1994 – even during the 2007-2009 recession – according to the APPA. In 2020, positive pet care trends during the pandemic based on increased adoptions and owners spending more time with their pets drove market growth.
We believe several factors will contribute to an increase in spending on pet therapeutics. Pets are generally living longer and as a result, are increasingly exhibiting many of the same diseases associated with aging in humans such as cardiovascular disease, arthritis, and diabetes. Among pet owners, there is growing familiarity in treating these pet diseases with medications. According to the APPA, in the U.S., approximately 63% of dog owners and 43% of cat owners treated their pets with medications in 2018.
In addition, pet ownership numbers may increase as more people become aware of the myriad health benefits of pet ownership. According to the Human Animal Bond Research Institute, studies show that some of the benefits of having a dog include helping to lower blood pressure, decrease risk of heart disease, and preventing allergies in children.
Several studies have shown that pet owners are willing to make financial sacrifices for their pets. In a 2017 Harris Poll conducted for the American Institute of Certified Public Accountant, 76% of U.S. adults indicated they would make financial sacrifices for their pets to pay for an emergency expense such as medical care.
We expect pet owners to spend more on their pets' health and welfare as new therapeutics are developed specifically for pets. The trend of anthropomorphizing or humanizing pets continues as pet owners increasingly view pets as members of their family, or even as surrogate children. Numerous studies have shown pet owners celebrate pet birthdays, purchase Christmas gifts for their pets, and have embraced technology by purchasing electronic devices to monitor the location or health of their pets.
Finally, the pet ownership levels and spending activity of millennials, which make up a quarter of the U.S. population, suggest these trends will continue into the future.
Pet Therapeutics Market Dynamics
The respective businesses of developing and commercializing therapeutics for pets and for humans share a number of characteristics, including the need to demonstrate safety and efficacy in clinical trials, obtain FDA or other regulatory approval for marketing, manufacture the therapeutics in facilities compliant with GMP requirements and market the therapeutics only for their intended indication based on claims permitted in the product label, and not for other uses, which is referred to as extra-label use.
Despite their similarities, there are a number of important differences between the pet therapeutics and human therapeutics businesses, including:
• Faster, less expensive and more predictable development. The development of pet therapeutics requires fewer clinical studies in fewer subject animals than the development of human therapeutics and, unlike human therapeutics, is conducted directly in the target animals. We believe our strategy of selecting targets with demonstrated efficacy and safety in humans enhances the predictability of results and probability of success of our pivotal trials relative to targets that have not been previously validated.
• Role and incentives for veterinary practices. In the United States, veterinarians generally serve the dual role of doctor and pharmacist, and pet owners typically purchase medicines directly from their veterinarians. Therapeutics specifically developed for pets enable veterinarians to provide potentially superior treatment options, while also increasing revenue from the sale of these therapeutics.
• Primarily private-pay nature of veterinary market. Pet owners in the United States generally pay for pet therapeutics out-of-pocket, and approximately 14% of dogs and 7% of cats have health insurance. As a result, pet owners must make decisions primarily on their veterinarians’ advice regarding available treatment options, rather than on the treatment options’ eligibility for reimbursement by insurance companies or government payers. We believe this results in less pricing pressure than in human healthcare, although the limited adoption of insurance may also reduce pet owners’ ability to pay for therapeutics recommended by their veterinarians.
• Less generic competition and strong brand loyalty. There is less generic competition in the pet therapeutics industry than in the human healthcare industry. Approximately 14% of veterinary drugs face generic competition, and the percentage of generic prescriptions in the veterinary space is only 7% as compared to approximately 81% for human drugs. For example, Rimadyl, the leading U.S. pet NSAID, lost regulatory exclusivity in 2001, but its sales have grown since generic competition was introduced in 2005. We believe that stronger brand loyalty and lack of mandatory generic drug substitution, as in human pharmaceuticals, partially explains the low penetration of generics in veterinary medicine.
Unmet Medical Needs in the Pet Therapeutics Market
Despite the growing market for pet therapeutics, there are relatively few treatment options approved for use in pets as compared to human therapeutic treatments. As a result, veterinarians often must resort to prescribing products approved for use in humans but not approved, formulated or even formally studied in pets. Veterinarians must then rely upon trial and error or untested rules of thumb to assess the proper dosage needed to be effective in the particular species without undue risk of side effects. The veterinarian must also find a way to administer the human product in animals and determine the appropriate dose to treat the disease in the species, which are important and potentially overlooked practical considerations in the treatment of pets.
Even in disease categories with approved pet therapeutics, significant unmet medical needs remain. For example, the NSAID class of products, commonly prescribed for pain, have potentially serious side effects in dogs that limit their long-term use and may require ongoing monitoring by veterinarians. The treatment of pain in cats is further complicated as a result of their differing biology, which makes NSAIDs toxic.
Animal health companies have been relatively slow to develop new therapeutics for pets and have tended to focus primarily on the larger market for the treatment of livestock and other farm animals. In 2020, human pharmaceutical companies received FDA/CDER approval for 141 original NDA and BLA drugs (non-generic), while animal health companies received only 16 FDA/CVM drug approvals. In the EU, the EMA recommended for authorization 97 new medicines (39 new active substances) for human use in 2020, compared only to 20 new medicines (10 new active substances) recommended for veterinary use.
We believe that therapeutics specifically developed for pets can extend and improve pets’ quality of life, help veterinarians achieve improved medical outcomes and make the process of administering therapeutics to pets much more convenient. Advances in human medicines have created new therapeutics for managing chronic diseases associated with aging, such as osteoarthritis, cancer, diabetes and cardiovascular diseases. Pets often suffer from the same disease as humans, including diabetes, arthritis, cancer, Alzheimer’s disease (canine cognitive dysfunction), lupus, Crohn’s disease, Lou Gehrig’s disease (degenerative myelopathy) and others. In most cases, the biologies of the diseases in pets are very similar to those in humans. Because of the similarity of the diseases, many human drugs, when formulated properly and administered in proper doses, are effective in pets. However, most human drugs are neither formulated nor approved for animals.
Commercialization
We have substantially reduced our companion animal commercial infrastructure and rely primarily on a partnership-focused commercialization model, whereby pipeline assets are out-licensed to larger commercial partners that can maximize product opportunity in return for upfront payments, contingent milestones, and royalties on future sales. We believe this strategy will maximize the value of our pipeline, generate attractive commercial terms, and rely less on dilutive capital.
According to industry sources, approximately one-third of pet veterinary practice revenue comes from prescription drug sales, vaccinations and non-prescription medicines. We believe veterinarians are self-motivated to prescribe innovative therapeutics that are safe, effective, and supported by reliable clinical data and regulatory approval in order to improve the health of pets, while also generating additional revenue.
Manufacturing
For biologics, we have established our own GMP manufacturing capabilities in Burlingame, California and proceeded to GMP manufacturing in January 2018. In August 2017, we acquired a manufacturing facility in Elwood, Kansas and completed construction and commissioning in 2019. The Elwood facility includes approximately 180,000 square feet with clean rooms, utility, equipment, and related quality documentation suitable for biologics and small molecule manufacturing. The USDA regulates the manufacture of pet biologics under standards that are less stringent than those for human biologics, which may reduce the cost of goods of our biologic product candidates relative to human biologics.
Competition
While there are fewer competitors in the pet therapeutics industry than in the human pharmaceutical industry, the development and commercialization of new animal health medicines is highly competitive, and we expect considerable competition from major pharmaceutical, biotechnology and specialty animal health medicines companies.
Our potential competitors include large animal health companies, which currently derive the majority of their revenue from livestock medications. For example, in 2020 livestock accounted for 44%, and pets 55%, of sales for Zoetis, a large company focused on animal health. Within the pet therapeutics market, vaccines and parasiticides are currently the greatest sources of revenue.
Large animal health companies include Merck Animal Health, the animal health division of Merck & Co., Inc.; Boehringer Ingelheim Animal Health, the animal health division of Boehringer Ingelheim GmbH;
Elanco Animal Health Incorporated; and Zoetis, Inc. We will also compete against several animal health companies, such as the Virbac Group, Ceva Animal Health and Dechra Pharmaceuticals PLC. We are also aware of smaller companies that are developing products for use in the pet therapeutics market, including Zomedica, Scout Bio, and Anivive Lifesciences.
At the product level, we will face competition for Zimeta from Flunixin and phenylbutazone, even though they are not approved for control of fever in horses. Mirataz, which was sold to Dechra in March 2020, will face competition from Elura. In addition, we may face competition from various products including additional products in development. Our products may also face competition from generic medicines and products approved for use in humans that are used extra-label for pets. Some of our other products also may face competition from their human generic equivalents in countries where such equivalents are available.
Many of our competitors and potential competitors have substantially more financial, technical and human resources than we do. Many also have far more experience than we have in the development, manufacture, regulation and worldwide commercialization of animal health medicines, including pet therapeutics. In addition, these and other potential competing products may benefit from greater brand recognition and brand loyalty than any that our product candidates may achieve. Accordingly, there is no assurance that we and our products can compete effectively.
Intellectual Property
We intend to rely primarily upon a combination of regulatory exclusivity, patents, trade secret protection, proprietary know-how, license agreements, and confidentiality agreements to protect our product formulations, biologics, processes, therapeutic methods and other technologies, and to operate without infringing on the proprietary rights of other parties, both in the United States and in other countries. We currently have numerous provisional, nonprovisional, and international patent applications pending, and two U.S. issued patents, for our IL-31 antibody compositions-of-matter and corresponding methods of use. Because our approved product (Zimeta) and our other small molecule product candidates are based on generic human drugs, there is little, if any, composition-of-matter patent protection available for the API in such product candidates. We have filed patent applications on many of our biologic products. However, even intellectual property protection, if available to us, may not afford us with complete protection against competitors. See “Risk Factors-Risks Related to Intellectual Property.”
We depend upon the skills, knowledge and experience of our management personnel, as well as that of our other employees, advisors, consultants and contractors, none of which are patentable. To help protect our know-how, and any inventions for which patents may be difficult to obtain or enforce, we require all of our employees, consultants, advisors and other contractors to enter into customary confidentiality and inventions agreements that prohibit the disclosure of confidential information and, where applicable, require disclosure and assignment to us of the ideas, developments, discoveries and inventions important to our business.
Regulatory
The development, approval and sale of animal health products are governed by the laws and regulations of each country in which we intend to sell our products. To comply with these regulatory requirements, we have established processes and resources to provide oversight of the development and launch of our products and their maintenance in the market.
United States
Three federal regulatory agencies regulate the health aspects of animal health products in the United States: the FDA; the USDA; and the Environmental Protection Agency (the “EPA”). In addition, the Drug Enforcement Administration (the “DEA”) regulates animal therapeutics that are classified as controlled substances.
The FDA Center for Veterinary Medicine (the “CVM”), regulates animal pharmaceuticals under the Federal Food, Drug and Cosmetic Act. The USDA Center for Veterinary Biologics (the “CVB”), regulates veterinary vaccines and certain biologics pursuant to the Virus, Serum, Toxin Act. The EPA Office of Pesticide Programs (the “OPP”) regulates veterinary pesticides under the Federal Insecticide, Fungicide and Rodenticide Act. Many topical products used for treatment of flea and tick infestations are regulated by the EPA.
All of our current product candidates are animal pharmaceuticals or biologics regulated by the CVM or the CVB, respectively. Manufacturers of animal health pharmaceuticals and biologics, including us, must show their products to be safe, effective and produced by a consistent method of manufacture. We are also required to conduct post-approval monitoring of products and to submit reports of product quality defects, adverse events or unexpected results, and are subject to regulatory inspection from time to time. In addition, for our controlled substance product candidates, we are required to comply with the Controlled Substances Act (the “CSA”) and related state laws regarding manufacturing, labeling, packaging, testing, dispensing, production and procurement quotas, recordkeeping, reporting, handling, shipment and disposal.
Requirements for Approval of Veterinary Pharmaceuticals for Pets
As a condition to regulatory approval for the sale of animal products, regulatory agencies worldwide generally require that a product to be used for pets be demonstrated to:
• be safe for the intended use in the intended species;
• have substantial evidence of effectiveness for the intended use;
• have a defined manufacturing process that ensures the product can be made with high quality consistency; and
• be safe for humans handling the product and for the environment.
Safety. To determine that a new veterinary drug is safe for use, most regulatory authorities will require us to provide data from a safety study generated in laboratory cats and dogs tested at doses higher than the intended label dose, over a period of time determined by the intended length of dosing of the product. In the case of the FDA, the design and review of the safety study and the study protocol can be completed prior to initiation of the study to help assure that the data generated will meet FDA requirements. These studies are conducted under rigorous quality control, including GLP, to assure integrity of the data. They are designed to clearly define a safety margin, identify any potential safety concerns, and establish a safe dose for the product. In addition, safety data from pivotal field studies conducted under GCP standards are evaluated to assure that the product will be safe in the target population. Furthermore, because safety and effectiveness studies must conform to VICH guidelines, which are established under an international program aimed at harmonizing technical requirements for veterinary product registration, they can be utilized by regulatory bodies in the European Union, Japan, Canada, New Zealand and Australia.
Effectiveness. Early pilot studies may be conducted in laboratory cats and dogs to establish effectiveness and the dose range for each product. Data on how well the drug is absorbed when dosed by different routes of administration and the relationship of the dose to the effectiveness are studied. When an effective dose is established, a study protocol to test the product in real world conditions is developed prior to beginning the study. In the case of the FDA, the pivotal effectiveness field study protocol can be submitted for review and concurrence prior to study initiation, to help assure that the data generated will meet requirements.
The pivotal field effectiveness study must be conducted with the formulation of the product that is intended to be commercialized, and is a multi-site, randomized, controlled study, generally with a placebo control. To reduce bias in the study, individuals doing the assessment are not told whether the subject is in the group receiving the treatment being tested or the placebo group. In both the United States and the European Union, the number of subjects enrolled in pivotal field effectiveness studies is required to be approximately
100 to 150 animal subjects treated with the test product and a comparable number of subjects in the control group that receive the placebo. In many cases, a pivotal field study may be designed with clinical sites in both the European Union and the United States, and this single study may satisfy regulatory requirements in both jurisdictions.
Chemistry, Manufacturing and Controls ("CMC"). To assure that the product can be manufactured consistently, regulatory agencies will require us to provide documentation of the process by which the API is made and the controls applicable to that process that assure the API and the formulation of the final commercial product meet certain criteria, including quality, purity and stability. After a product is approved, we will be required to communicate with the regulatory bodies any changes in the procedures or manufacturing site. Both API and commercial formulations are required to be manufactured at facilities that practice pharmaceutical GMP.
Environmental and Human Safety. We will not be required under United States law to provide an environmental impact statement for products currently in development if the products are given at the home of the pet’s owner or in a veterinary hospital. If products might result in some type of environmental exposure or release, the environmental impact must be assessed. For approval in the EU, a risk assessment for potential human exposure will be required.
Labeling, All Other Information, and Freedom of Information Summary. We also will be required to submit the intended label for the product, and also any information regarding additional research that has been conducted with the drug, to the CVM and other regulatory bodies for review. We will draft, and submit for regulatory review, the Freedom of Information Summary for use in the United States. This summary outlines the studies and provides substantial information that the FDA uses to assess the drug’s safety and effectiveness and then publishes on its website.
Regulatory Process at the FDA
To begin the development process for products in the United States, we must file an Investigational New Animal Drug (INAD) submission with the FDA. We will then usually hold a pre-development meeting with the FDA to reach a general agreement on the plans for providing the data necessary to fulfill requirements for an NADA. We evaluate if drug candidates can benefit from approval under the minor use minor species (MUMS) or expanded conditional approval programs. During development, we will usually submit pivotal protocols to the FDA for review and concurrence prior to conducting the required studies. We will gather and submit data on manufacturing, safety and effectiveness to the FDA for review, and this review will be conducted according to timelines specified in the Animal Drug User Fee Act. These are called technical sections, which collectively form the basis of the NADA. Once all data have been submitted and reviewed for each technical section - safety, effectiveness and CMC - the FDA will issue us a technical section complete letter as each section review is completed, and when the three letters have been issued, we will compile a draft of the Freedom of Information Summary, the proposed labeling, and all other relevant information, and submit these for FDA review. An administrative NADA is a NADA that is submitted after all of the technical sections that fulfill the requirements for the approval of the new animal drug have been reviewed by the CVM and the CVM has issued a technical section complete letter for each of those technical sections. Although this process is not required and submission of a non-administrative NADA is also acceptable, we plan to take advantage of the administrative NADA process to obtain a more timely, phased review. Because the CVM has already reviewed the individual technical sections before the administrative NADA is filed, the CVM is committed under its user fee agreements to reviewing and acting on 90% of administrative NADAs within 60 days after submission. The CVM user fee goal is to review and act on 90% of non-administrative NADAs within 180 days after submission. After approval, we will be required to collect reports of adverse events and product quality complaints, and submit them on a regular basis to the FDA.
Regulatory Process at the USDA
To begin the development process for veterinary biologics products in the United States, we typically file an Application for United States Veterinary Biological Product License with the USDA. For the biologics products that we develop, we may then meet with the USDA to reach a general agreement on the plans for providing the data necessary to fulfill requirements for an approval. During development, we gather and submit data on manufacturing, purity and potency to the USDA for review. Once all data have been submitted and reviewed, the USDA will issue its decision. Unlike the FDA, there are no timelines specified by law for the USDA’s review.
In some cases, it may be unclear whether our product candidates meet the definition of a biological product subject to regulation by the USDA or a drug subject to regulation by the FDA. The USDA’s Center for Veterinary Biologics and the FDA’s Center for Veterinary Medicine have a memorandum of understanding concerning their joint responsibilities for resolving jurisdictional issues over products of this nature. Under the memorandum of understanding, animal products are to be regulated by the USDA as biologics if they are intended for use to diagnose, cure, mitigate, treat, or prevent disease in animals and they work primarily through an immune process, or by the FDA as drugs, if they are intended for use in the diagnosis, cure, mitigation, treatment, or prevention of animal disease if the primary mechanism of action is not immunological or is undefined. There is a process to gain a jurisdiction decision.
Regulatory Process at the EMA
The EMA is responsible for coordinating scientific evaluation of applications for marketing approval for pet therapeutics in the EU. Its veterinary review section is distinct from the review section for human drugs. To perform these evaluations the EMA established a specific scientific committee, the Committee for Medicinal Products for Veterinary Use ("CVMP"). The CVMP considers applications submitted by companies for the marketing approval of individual pet therapeutics and evaluates whether or not the medicines meet the necessary quality, safety and efficacy requirements. Assessments conducted by the CVMP are based on scientific criteria and are intended to ensure that pet therapeutics reaching the marketplace have a positive benefit-risk balance in the pet population for which they are intended. Based on the CVMP’s recommendation, a centralized marketing authorization is granted by the EMA, which allows the product to be marketed in any of the EU states, Norway, Lichtenstein and Iceland. The CVMP is also responsible for various post-authorization and maintenance activities, including the assessment of modifications or extensions to an existing marketing authorization.
To obtain authorization from the EMA, we must submit a marketing authorization application called a dossier. The dossier is the EMA’s equivalent of the FDA’s NADA and includes data from studies showing the quality, safety and efficacy of the product. The CVMP reviews and evaluates the dossier. For any dossier, a rapporteur and co-rapporteur are appointed from the members of the CVMP. Their role is to lead the scientific evaluation and prepare the assessment report. The rapporteur can utilize experts to assist it in performing its assessment. The report is critiqued by the co-rapporteur and other members of the CVMP before the CVMP makes its determination. The final opinion of the CVMP is generally given within 210 days of the submission of a dossier, but the EMA makes the final decision on the approval of products. In general, the requirements for regulatory approval of an animal health product in the EU are similar to those in the United States, requiring demonstrated evidence of purity, safety, efficacy and consistency of manufacturing processes.
Alternatively, product approval applications may be submitted directly to the regulatory authority in each country rather than by centralized approval by the EMA.
Regulatory Processes at the DEA
The DEA regulates controlled substances as Schedule I, II, III, IV or V substances. Schedule I substances by definition have no established medicinal use and may not be marketed or sold in the United States. An animal drug may be listed as Schedule II, III, IV or V, with Schedule II substances considered to
present the highest risk of abuse and Schedule V substances the lowest relative risk of abuse among such substances. Certain of our product candidates are likely to be scheduled as controlled substances under the CSA. Consequently, their manufacture, shipment, storage, sale and use will be subject to a high degree of regulation.
Annual registration is required for any facility that manufactures, distributes, dispenses, imports or exports any controlled substance. The registration is specific to the particular location, activity and controlled substance schedule. For example, separate registrations are needed for import and manufacturing, and each registration will specify which schedules of controlled substances are authorized.
The DEA typically inspects a facility to review its security measures prior to issuing a registration. Security requirements vary by controlled substance schedule, with the most stringent requirements applying to Schedule I and Schedule II substances. Required security measures include background checks on employees and physical control of inventory through measures such as cages, surveillance cameras and inventory reconciliations. Records must be maintained for the handling of all controlled substances, and periodic reports must be made to the DEA, for example, distribution reports for Schedule II controlled substances, Schedule III substances that are narcotics, and other designated substances. Reports must also be made for thefts or losses of any controlled substance, and to obtain authorization to destroy any controlled substance. In addition, special authorization and notification requirements apply to imports and exports.
In addition, a DEA quota system controls and limits the availability and production of controlled substances in Schedule I or II. Distributions of any Schedule I or II controlled substance must also be accompanied by special order forms, with copies provided to the DEA. The DEA may adjust aggregate production quotas and individual production and procurement quotas from time to time during the year, although the DEA has substantial discretion in whether or not to make such adjustments.
Other Regulatory Considerations
Regulatory rules relating to human food safety, food additives, or drug residues in food will not apply to the products we currently are developing because our products are not intended for use in food animals or food production animals, with the exception of horses, which qualify as food animals in Europe and Canada.
Advertising and promotion of animal health products is controlled by regulations in the United States and other countries. These rules generally restrict advertising and promotion to those claims and uses that have been reviewed and authorized by the applicable agency. We will conduct a review of advertising and promotional material for compliance with local and regional requirements in the markets where we sell pet therapeutics.
While small molecule product drugs may eventually face generic competition in the United States, there is no pathway for approval of a generic veterinary biologic regulated by the USDA.
Human Capital
Our success depends on our ability to retain and attract the top talent. We offer a diverse, safe and fun workplace for our employees to grow and develop in their careers.
As of December 31, 2020, we had 63 employees, including 11 employees with D.V.M., M.D. or Ph.D. degrees. Of our employees, 17, including Dr. Chin, are engaged in one or more aspects of our research and development activities. Dr. Chin also is engaged in corporate and administrative activities. None of our employees are represented by labor unions or covered by collective bargaining agreements.
Diversity and Inclusion
As of December 31, 2020, our employee base was comprised of 46% females and 54% males, and of the following ethnicities: 34% Asian, 59% White, 3% Hispanic, 3% two or more ethnicities and 1% additional groups (including American Indian or Alaska Native and Native Hawaiian or Other Pacific Islander). We will also continue our ongoing efforts to increase the representation of women in leadership.
We diversify our workforce through recruiting, retention, people development, and inclusion. We create more opportunities to consider qualified people from underrepresented groups for open roles. We promote an inclusive workplace by helping people understand the issues that affect underrepresented communities and how to reduce the effects of bias in the workplace.
Compensation and Benefits
We offer competitive compensation to attract and retain the best people to work on our mission. Our employees' total compensation package includes salary, bonuses, and equity. We offer full-time employees equity at the time of hire and through annual equity grants because we want them to become owners of the company and be committed to our long-term success. We ensure fair and equitable pay across gender and race in the United States for people in similar jobs, accounting for factors such as location, role, and level. We offer a wide range of benefits across areas such as health, family, and paid-time off, including healthcare and wellness benefits, family care resources, a 401(k) plan.
Health, Safety, Wellness and Covid-19 responses
We provide our employees and their families with access to a variety of health and wellness programs. We encourage employees to engage in healthy behaviors, and we offer choices where possible so they are customized to meet their needs and the needs of their families.
In response to the COVID-19 pandemic, we made important trainings and changes that we determined were in the best interest of our employees, and the communities in which we operate, in compliance with government regulations. This includes having the vast majority of our employees work from home, while implementing additional safety measures for employees continuing critical on-site work.
Available Information
We file annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy statements and other documents with the SEC. All of our filings with the SEC (including documents that we “furnish” with the SEC rather than “file” with the SEC) are available on the SEC’s website at www.sec.gov. Our filings with the SEC are also available free of charge on our website at www.kindredbio.com as soon as reasonably practicable after each document is electronically filed with or furnished to the SEC. The information contained in, or accessible through, our website is not a part of this Annual Report on Form 10-K and is not incorporated by reference into any other filings that we make with the SEC.
ITEM 1A. RISK FACTORS
Investing in our common stock involves a high degree of risk. You should carefully consider the risks described below, as well as the other information contained in or incorporated by reference into our other public filings with the Securities and Exchange Commission, before deciding whether to invest in our common stock. The occurrence of any of the events or developments described below could harm our business, financial condition, results of operations and growth prospects. In such an event, the market price of our common stock could decline, and you may lose all or part of your investment. Additional risks and uncertainties not presently known to us or that we currently deem immaterial also may impair our business operations.
Summary of Principal Risks
The following bullet points summarize the principal risks we face, each of which could adversely affect our business, operations, and financial results. For clarity of presentation, we have arranged these risks by what they most directly affect – (1) our business, (2) our intellectual property, (3) government regulation, and (4) our stock price. A fifth group of “general risk factors” lists risks that apply to businesses generally.
Risks Related to Our Business
•The current pandemic of COVID-19 and the future outbreak of other highly infectious or contagious diseases could have a material adverse impact on our business, financial condition and results of operations, including research and development, regulatory approvals and manufacturing of our product candidates, and our partnership-based model for commercialization.
•We have a limited operating history, are not profitable and may never become profitable.
•We are substantially dependent on the success of the product candidates in our pipeline.
•The results of earlier studies may not be predictive of the results of our pivotal trials, and we may be unable to obtain regulatory approval for our existing or future product candidates under applicable regulatory requirements.
•The commercial potential of a product candidate in development is difficult to predict.
•Our secured loan agreement contains restrictions that limit our flexibility in operating our business.
•There is no assurance as to the amount of royalties that we will receive from the sale of Mirataz® to Dechra or as to the amount of revenue that we will receive from third-party licensees of our products.
Risks Related to Our Intellectual Property
•Our commercial success will depend, in part, on obtaining and maintaining patent protection for our products.
•We may become subject to third parties’ claims alleging infringement of patents and proprietary rights or priority of invention.
•If our efforts to protect the proprietary nature of the intellectual property related to our products or any of our current or future product candidates are not adequate, we may not be able to compete effectively in our market.
Risks Related to Government Regulation
•Even if we receive regulatory approval for any of our current or future product candidates, we will be subject to ongoing regulatory obligations, review, requirements, and restrictions.
•Any of our current or future approved products may cause or contribute to adverse medical events that we are required to report to regulatory authorities and, if we fail to do so, we could be subject to sanctions that would materially harm our business.
•Legislative or regulatory reforms with respect to pet therapeutics may make it more difficult and costly for us to obtain regulatory clearance or approval of any of our current or future product candidates and to produce, market, and distribute our products after clearance or approval is obtained.
•Certain of our product candidates currently in development may be classified as controlled substances, the manufacture, use, sale, importation, exportation, and distribution of which are subject to additional regulation by state, federal, and foreign law enforcement and other regulatory agencies.
Risks Related to Our Common Stock
•The price of our common stock could be subject to volatility related or unrelated to our operations.
•Our quarterly and annual operating results may be volatile and may vary significantly from the estimates and expectations of investors and third parties, including securities or industry analysts, which could cause the market price of our common stock to decline.
•Our principal stockholders and management own a significant percentage of our stock and will be able to exert significant control over matters subject to stockholder approval.
General Risk Factors
•If we fail to retain current members of our senior management, or to attract and keep additional key personnel, our business and prospects could be materially adversely impacted.
•Our information technology systems and the information technology systems of third parties with whom we do business are vulnerable to cyber-attacks, breaches of security and misappropriation of data, which could result in substantial damage to our business and operations.
Risk Factors - Discussion
The following section discusses the principal risks listed above, as well as other risks we believe to be material.
Risks Related to Our Business
The current pandemic of COVID-19 and the future outbreak of other highly infectious or contagious diseases, could have a material adverse impact on our business, financial condition and results of operations, including our research and development of our product candidates, regulatory approvals of our product candidates, manufacturing of our product candidates, and our partnership-based model for commercialization.
In December 2019, a novel strain of coronavirus, or COVID-19, was reported to have surfaced and it has since reached multiple regions, countries and cities, including in California where our headquarters are located, and all cities in which we have offices and facilities. The COVID-19 pandemic continues to evolve and the ultimate impact of this pandemic is highly uncertain and subject to change. To date, the pandemic has led to the implementation of various responses, including government-imposed quarantines, travel restrictions and other public health safety measures. Certain jurisdictions have begun re-opening only to return to restrictions in the face of increases in new COVID-19 cases. COVID-19 has and will likely continue to impact our operations and those of our third-party partners and the ultimate impact on our business and financial results remains uncertain and cannot be predicted with confidence, and will depend on many factors, including the scope, severity, duration and any recurrence of the COVID-19 pandemic, including through any new variant strains of the underlying virus, the actions taken to contain the pandemic or mitigate its impact, the direct and indirect economic effects of the pandemic and containment measures, and the availability and efficacy of vaccines and treatments for COVID-19,
among others. The continued development and fluidity of the COVID-19 pandemic precludes any prediction as to its full impact on our business. Nevertheless, the COVID-19 pandemic may adversely affect our business, financial condition and results of operations.
In response to the spread of COVID-19, we took, and have continued to take, temporary precautionary measures intended to help minimize the risk of the virus to our employees and their families, including implementing a work from home policy for nearly all employees who are able to perform their duties remotely, and have generally restricted on-site staff to only those personnel and contractors who perform activities that need to be completed on-site, limited the number of staff in any given laboratory, manufacturing facility or other facility and implemented safety practices and procedures for those individuals who are required to work in our facilities, including but not limited to mandatory health screening, the use of face coverings, physical distancing requirements and increased cleaning protocols, as well as voluntary onsite testing. We expect to adopt and implement additional precautions commensurate with any expansion of employees returning to physical locations, and at this time, we cannot predict when certain restrictions that are in place to protect our employees can be further reduced or will no longer be needed. The effects of government-imposed quarantines and our work-from-home policies may negatively impact productivity, or disrupt, delay, or otherwise adversely impact our business. Compliance with governmental measures imposed in response to COVID-19 has caused and will continue to cause us to incur additional costs, and any inability to comply with such measures can subject us to restrictions on our business activities, fines, and other penalties, any of which can adversely affect our business. In addition, the increase in certain of our employees working remotely has amplified certain risks to our business, including increased demand on our information technology resources and systems, increased phishing and other cybersecurity attacks, and any failure to effectively manage these risks, including to timely identify and appropriately respond to any cyberattacks, could adversely impact our business operations.
As a result of the COVID-19 pandemic, we may experience disruptions that could severely impact our business and operations, including our ability to successfully commercialize our products and our product candidates, and due to the current pandemic, we may not be able to meet expectations with respect to commercial sales. For example, due to the impact of the COVID-19 pandemic, the downturn in equine events and transportation affected Zimeta sales. In addition, business interruptions from the current or future pandemics, including staffing shortages, production slowdowns and disruptions in delivery systems, may also adversely impact the third parties we or our partners rely on in the U.S. and abroad to sufficiently provide supplies related to the manufacture of our products and product candidates and to provide supplies in quantities we require, which may impair our commercialization efforts, our research and development activities and the commercialization of our products and product candidates.
Additionally, timely completion of pre-clinical activities and initiation of planned clinical trials is dependent upon the availability of, for example, pre-clinical and clinical trial sites, researchers and investigators, pet patients or healthy volunteer pet subjects available for recruitment and enrollment, and regulatory agency personnel, which may be adversely affected by global health matters, such as the COVID-19 pandemic. We are conducting and plan to continue to conduct pre-clinical activities and clinical trials for our product candidates in geographies which have been and continue to be affected by COVID-19, and believe that the COVID-19 pandemic will have an impact on various aspects of our ongoing clinical trials and on the clinical trials and pre-clinical studies we expect to initiate in 2021. For example, certain veterinary clinics in our pivotal study for KIND-510a suspended clinical trials due to the COVID-19 pandemic. Any business interruptions caused by the COVID-19 pandemic could also delay necessary interactions with local regulators, ethics committees, manufacturing sites, research or clinical trial sites and other important agencies and contractors, which could adversely impact the clinical trials of our product candidates.
Health regulatory agencies globally may also experience disruptions in their operations as a result of the COVID-19 pandemic, which may impact review, inspection and approval timelines.
Some factors from the COVID-19 pandemic that may delay or otherwise adversely affect enrollment in and the conduct of the clinical trials of our product candidates, as well as adversely impact our business generally, include:
•limitations on travel that could interrupt key trial activities, such as clinical trial site initiations and monitoring, travel by employees, contractors or pets to clinical trial sites, including any government-imposed travel restrictions or quarantines that will impact the ability or willingness of volunteers, employees or contractors to travel to our research, manufacturing and clinical trial sites, any of which could delay or adversely impact the conduct or progress of our clinical trials; and
•interruption of, or delays in receiving, supplies of our product candidates from our suppliers due to staffing shortages, production slowdowns or stoppages and disruptions in delivery systems.
These and other factors arising from the COVID-19 pandemic could worsen in countries that are already afflicted with COVID-19 or could return to countries where the pandemic has been partially contained, each of which could further adversely impact our ability to conduct clinical trials and our business generally, and could have a material adverse impact on our operations and financial condition and results. In addition, a recession, depression or other sustained adverse market event resulting from the COVID-19 pandemic could materially and adversely affect our business and the value of our common stock.
The ultimate impact of the COVID-19 pandemic remains highly uncertain and subject to change. We do not yet know the full extent of potential delays or impacts on our business, our commercial results, clinical trials, healthcare systems or the global economy as a whole. These effects could have a material impact on our operations, and we will continue to monitor the COVID-19 situation closely.
We have a limited operating history, are not profitable and may never become profitable.
We are a commercial stage biopharmaceutical company. Since our formation in September 2012 and until June 2018, our operations were limited to the identification of product candidates and research and development of our product candidates, including our lead product candidates, Mirataz and Zimeta. Mirataz became commercially available in July 2018 and Zimeta in December 2019. As a result, we have limited historical operations upon which to evaluate our business and prospects, we have demonstrated an ability to obtain marketing approval for only two of our product candidates and have not yet successfully overcome the risks and uncertainties frequently encountered by companies in emerging fields such as the pet therapeutics industry. We have only generated revenue on Mirataz for twenty-two months and thirteen months for Zimeta, and will continue to incur significant research and development and other expenses. As of December 31, 2020, we had an accumulated deficit of $244.9 million. For the foreseeable future, we expect to continue to incur losses, which will increase significantly from historical levels as we expand our product development activities, seek regulatory approvals for our product candidates and begin to commercialize them if they are approved by the Center for Veterinary Medicine branch of the FDA, the USDA or the EMA. It is also likely that our revenue will vary from quarter to quarter, and we face the risk that the revenue from our delivery of services or products under any contract may be less than we anticipate if the other party to the contract exercises its right to terminate the contract prior to completion of the contract or if such party is unable or unwilling to satisfy its payment obligations under the contract. Even if we succeed in developing and commercializing one or more product candidates, we expect to continue to incur losses for the foreseeable future, and we may never become profitable. If we fail to achieve or maintain profitability, it would adversely affect the value of our common stock.
We may need to raise additional capital to achieve our goals.
Our biologics candidates typically require four to six years of further development at an average cost of approximately $8 million per product candidate before we expect to be able to apply for marketing approval in the United States. We also are actively involved in identifying additional human therapeutics for development and commercialization as pet therapeutics and will continue to expend substantial resources for the foreseeable future to develop our current product candidates and any other product candidates we may develop or acquire. These expenditures will include: costs of identifying additional potential product candidates; costs associated with drug formulation; costs associated with conducting pilot, pivotal, and toxicology studies; costs associated with completing other research and development activities; costs associated with payments to technology licensors and maintaining other intellectual property; costs of obtaining regulatory approvals; costs associated with establishing commercial manufacturing and supply capabilities; and costs associated with marketing and selling any of our products approved for sale, either directly by us or through licensees. We also may incur unanticipated costs.
Because the outcome of these activities is inherently uncertain, the actual amounts necessary to successfully complete the development and commercialization of our current or future product candidates may be greater or less than we anticipate.
We believe we have sufficient cash and cash equivalents to fund our operating plan for at least another 24 months. However, we may seek additional funds through public or private equity or debt financings or other sources such as strategic collaborations. Additionally, we do not expect our existing cash and cash equivalents to be sufficient to complete the development of all of our current product candidates, or of any additional product candidates that we may identify, and we may need to raise additional capital to fund these activities. Even if we believe we have sufficient funds on hand for our current or planned future business and operations, we may seek from time to time to raise additional capital based upon favorable market conditions or strategic considerations such as potential acquisitions.
Our future capital requirements depend on many factors, including, but not limited to:
•the scope, progress, results and costs of researching and developing our current or future product candidates;
•the timing of, and the costs involved in, obtaining regulatory approvals for any of our current or future product candidates;
•the number and characteristics of the product candidates we pursue;
•the cost of manufacturing our current and future product candidates and any products we successfully commercialize;
•the cost of commercialization activities for equine products and any other products that we elect to sell directly rather than through licensees, including marketing, sales and distribution costs;
•the amount of licensing and royalty payments that we receive with respect to our products that are out-licensed to commercial partners or are sold to other companies;
•the expenses needed to attract and retain skilled personnel;
•the costs associated with being a public company;
•our ability to establish and maintain strategic collaborations, licensing or other arrangements and the financial terms of such agreements; and
•the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing current and future patents, including litigation costs and the outcome of any such litigation.
Additional funds may not be available when we need them on terms that are acceptable to us, or at all. If adequate funds are not available to us on a timely basis, we may be required to delay, limit, reduce or terminate one or more of our product development programs or any future commercialization efforts.
We are substantially dependent on the success of the product candidates in our pipeline and cannot be certain that any of our product candidates will be approved for marketing or successfully commercialized even if approved.
A substantial portion of our efforts over the foreseeable future will be focused on the long-term commercial success of our product candidates that are in our pipeline. Accordingly, our prospects, including our ability to generate material product revenue, obtain any new financing if needed to fund our business and operations, or enter into potential strategic transactions, will depend heavily on the successful development and commercialization of our lead product candidates, which in turn will depend on a number of factors, including the following:
•the successful completion of the pivotal trials and toxicology studies of one or more of our current product candidates, which may take significantly longer than we currently anticipate and will depend, in part, upon the satisfactory performance of third-party contractors;
•our ability to demonstrate to the satisfaction of the FDA, the USDA and the EMA the safety and efficacy of our product candidates and to obtain regulatory approvals;
•our ability or the ability of our third-party manufacturers to manufacture satisfactory supplies of our products and product candidates, or supplies used in the manufacture of our products and product candidates, and to develop, validate and maintain commercially viable manufacturing processes that are compliant with GMP and to manufacture them at an acceptable cost as well as the ability to sell them at an acceptable price with reasonable margins;
•our ability to successfully launch commercial sales of our current product candidates, assuming marketing approval is obtained, whether through our own efforts or through licensing agreements with commercial partners;
•the effectiveness of the commercialization efforts for our products and product candidates, including the effectiveness of marketing, sales and distribution strategy and operations, whether performed by us or by our commercial partners through licensing agreements.
•the availability, perceived advantages, relative cost, relative safety and relative efficacy of our products and product candidates compared to alternative and competing treatments;
•the acceptance of our products and product candidates as safe and effective by veterinarians, pet owners and the animal health community;
•the prevalence and severity of adverse side effects and our ability to maintain a continued acceptable safety profile of a product following approval;
•our ability to obtain supplemental indications for our products and product candidates;
•any product liability claim or lawsuit we may be involved in from time to time with regards to our products and product candidates;
•our ability to achieve and maintain compliance with all regulatory requirements applicable to our business; and
•our ability to obtain and enforce our intellectual property rights and obtain marketing exclusivity for our products and product candidates, and avoid or prevail in any third-party patent interference, patent infringement claims or administrative patent proceedings initiated by third parties or the U.S. Patent and Trademark Office, or USPTO.
Many of these factors are beyond our control.
If our product candidates are approved, they may face significant competition and may be unable to compete effectively.
The development and commercialization of pet therapeutics is highly competitive and our success will depend on our ability to compete effectively with other products in the market. We expect to compete with animal health divisions of major pharmaceutical and biotechnology companies such as Merck Animal Health, Zoetis, Elanco, and Boehringer Ingelheim Animal Health, as well as specialty animal health medicines companies such as Virbac Group, Ceva Animal Health and Dechra Pharmaceuticals PLC. Additionally, we are aware of smaller companies that are developing products for use in the pet therapeutics market, including Zomedica, Scout Bio, and
Anivive Lifesciences. We also expect to compete with academic institutions, governmental agencies and private organizations that are conducting research in the field of animal health medicines.
Many of our competitors and potential competitors have substantially more financial, technical and human resources than we do. Many also have far more experience than we have in the development, manufacture, regulation and worldwide commercialization of animal health medicines, including pet therapeutics.
For these reasons, there is no assurance that we and any of our approved products can compete effectively.
The development of our biologic product candidates is dependent upon relatively novel technologies and uncertain regulatory pathways.
We have developed and plan to continue to develop biologics, including animal antibodies, for pets. Identification, optimization, and manufacture of therapeutic animal biologics is a relatively new field in which unanticipated difficulties or challenges could arise, and we expect the discovery, development, manufacturing and sale of biologic products to be a long, expensive and uncertain process. While many biologics have been approved for use in humans, apart from vaccines, relatively few recombinant proteins or antibodies have been approved for use in animals. There are unique risks and uncertainties with biologics, the development, manufacturing, and sale of which are subject to regulations that are often more complex and extensive than the regulations applicable to other small molecule products. We may be unable to identify biologics suitable for development or to achieve the potency and stability required for use in pets. In particular, canine and feline antibodies represent new types of product candidates that may be difficult to develop successfully.
In some cases, it may be unclear whether our product candidates meet the definition of a biological product subject to regulation by the USDA or a drug subject to regulation by the FDA. The USDA’s Center for Veterinary Biologics and the FDA’s Center for Veterinary Medicine have a memorandum of understanding concerning their joint responsibilities for resolving jurisdictional issues over products of this nature. Under the memorandum of understanding, animal products are to be regulated by the USDA as biologics, if they are intended for use to diagnose, cure, mitigate, treat, or prevent disease in animals and they work primarily through an immune process, or by the FDA as drugs, if they are intended for use in the diagnosis, cure, mitigation, treatment, or prevention of animal disease if the primary mechanism of action is not immunological or is undefined.
Although we believe that most of our current animal biologics will be regulated by the USDA based on their mechanisms of action, the USDA and the FDA may not agree with our assessment, or disputes may arise between the USDA and the FDA over regulatory jurisdiction for one or more of such biologics. If so, the development of our biologics may be delayed while any such disputes are adjudicated by the agencies. Furthermore, if the agencies were to determine that one or more of our animal biologics will be regulated by the FDA instead of the USDA, the time and cost of developing such biologics may be longer and more expensive than we currently anticipate, and we may determine to discontinue development of such biologics. It is also possible that the USDA’s regulatory standards for novel biologics may be more difficult to satisfy than we anticipate. The current trend indicates that the FDA is attempting to exert jurisdiction over more biologics.
We believe that some veterinarians prefer to see further efficacy data before making a new biologic product prescribing decision. Accordingly, we may also find it necessary to conduct additional studies of our biologic product candidates in order to achieve commercial success.
The results of earlier studies may not be predictive of the results of our pivotal trials, and we may be unable to obtain regulatory approval for our existing or future product candidates under applicable regulatory requirements. The denial or delay of any regulatory approval would prevent or delay our commercialization efforts and adversely affect our potential to generate material product revenue and our financial condition and results of operations.
The research, testing, manufacturing, labeling, approval, sale, marketing and distribution of pet therapeutics are subject to extensive regulation. We are usually not permitted to market our products in the United States until we receive approval of an NADA from the FDA or a PLA from the USDA, or in the EU or in other EEA countries until we receive marketing approval from the EMA. To gain approval to market a pet therapeutic for a particular species,
we must provide the FDA, the USDA and the EMA, as applicable, with efficacy data from pivotal trials that adequately demonstrate that our product candidates are safe and effective in the target species (e.g., dogs, cats or horses) for the intended indications. In addition, we must provide manufacturing data. For the FDA and EMA, we must provide data from toxicology studies, also called target animal safety studies, and in some cases environmental impact data. We are conducting the pivotal trial of our compounds internally without significant outsourcing, but we rely on contract research organizations ("CROs") and other third parties to conduct our toxicology studies and for certain other development activities. The results of toxicology studies and other initial development activities, and of any previous studies in humans or animals conducted by us or third parties, may not be predictive of future results of pivotal trials or other future studies, and failure can occur at any time during the conduct of pivotal trials and other development activities by us or our CROs. Our pivotal trial may fail to show the desired safety or efficacy of our product candidates despite promising initial data or the results in previous human or animal studies conducted by others, and success of a product candidate in prior animal studies, or in the treatment of human beings, does not ensure success in subsequent studies. Clinical trials in humans and pivotal trials in animals sometimes fail to show a benefit even for drugs that are effective, because of statistical limitations in the design of the trials or other statistical anomalies. Therefore, even if our studies and other development activities are completed as planned, the results may not be sufficient to obtain regulatory approval for our product candidates.
The FDA, USDA or EMA can delay, limit or deny approval of any of our product candidates for many reasons, including:
•if the FDA, USDA or EMA disagrees with our interpretation of data from our pivotal studies or other development efforts;
•if we are unable to demonstrate to the satisfaction of the FDA, USDA or EMA that the product candidate is safe and effective for the target indication;
•if the FDA, USDA or EMA requires additional studies or changes its approval policies or regulations;
•if the FDA, USDA or EMA does not approve of the formulation, labeling or the specifications of our current and future product candidates; and
•if the FDA, USDA or EMA fails to approve the manufacturing processes of our third-party contract manufacturers.
Further, even if we receive approval of our product candidates, such approval may be for a more limited indication than we originally requested, and the FDA, USDA or EMA may not approve the labeling that we believe is necessary or desirable for the successful commercialization of our product candidates.
Any delay or failure in obtaining applicable regulatory approval for the intended indications of our product candidates would delay or prevent commercialization of such product candidates and would materially adversely impact our business and prospects.
Our Protocol Concurrences with the FDA for our pivotal studies do not guarantee marketing approval in the United States.
We may conduct pivotal trials under Protocol Concurrences with the FDA. A Protocol Concurrence in animal drug development is analogous to a Special Protocol Assessment in human drug development, and means that the FDA agrees that the design and analyses proposed in a protocol are acceptable to support regulatory approval of the product candidate with respect to effectiveness of the indication studied and will not change its view of these matters, unless public or animal health concerns arise that were not recognized at the time of protocol concurrence or we change the protocol. Even under a Protocol Concurrence, approval of a NADA by the FDA is not guaranteed, because a final determination that the agreed-upon protocol satisfies a specific objective, such as the demonstration of efficacy, or supports an approval decision, will be based on a complete review of all the data submitted to the FDA.
Development of pet therapeutics is inherently expensive, time-consuming and uncertain, and any delay or discontinuance of our current or future pivotal trials would significantly harm our business and prospects.
Development of pet therapeutics remains an inherently lengthy, expensive and uncertain process, and there is no assurance that our development activities will be successful. We do not know whether the trials of our current or future product candidates will begin or conclude on time, and they may be delayed or discontinued for a variety of reasons, including if we are unable to:
•address any safety concerns that arise during the course of the studies;
•complete the studies due to deviations from the study protocols or the occurrence of adverse events;
•add new study sites;
•address any conflicts with new or existing laws or regulations; or
•reach agreement on acceptable terms with study sites, which can be subject to extensive negotiation and may vary significantly among different sites.
Any delays in completing our development efforts will increase our costs, delay our product candidate development and approval process and jeopardize our ability to commence product sales and generate revenue. Any of these occurrences may significantly harm our business, financial condition and prospects. In addition, factors that may cause a delay in the commencement or completion of our development efforts may also ultimately lead to the denial of regulatory approval of our product candidates which, as described above, would materially, adversely impact our business and prospects.
We currently rely on third parties to conduct some of our development activities and may rely more heavily on such third parties in the future. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, we may be unable to obtain regulatory approval for or commercialize our current or future product candidates as planned.
We currently plan to conduct our own pivotal trials, but we rely upon CROs to conduct our toxicology studies and for other development activities. We also may rely on CROs in the future to conduct one or more pivotal trials. These CROs are not our employees, and except for contractual duties and obligations, we have limited ability to control the amount or timing of resources that they devote to our programs or manage the risks associated with their activities on our behalf. We are responsible to regulatory authorities for ensuring that each of our studies is conducted in accordance with the development plans and trial protocols, and any failure by our CROs to do so may adversely affect our ability to obtain regulatory approvals, subject us to penalties, or harm our credibility with regulators. The FDA and foreign regulatory authorities also require us and our CROs to comply with regulations and standards, commonly referred to as good clinical practices ("GCPs"), or good laboratory practices ("GLPs"), for conducting, monitoring, recording and reporting the results of our studies to ensure that the data and results are scientifically credible and accurate.
Our agreements with CROs may allow termination by the CROs in certain circumstances with little or no advance notice to us. These agreements generally will require our CROs to reasonably cooperate with us at our expense for an orderly winding down of the CROs’ services under the agreements. If the CROs conducting our studies do not comply with their contractual duties or obligations to us, or if they experience work stoppages, do not meet expected deadlines, terminate their agreements with us or need to be replaced, or if the quality or accuracy of the data they obtain is compromised due to the failure to adhere to our development protocols or GCPs or for any other reason, we may need to secure new arrangements with alternative CROs, which could be difficult and costly. In such event, our studies also may need to be extended, delayed or terminated as a result, or may need to be repeated. If any of the foregoing were to occur, regulatory approval and commercialization of our product candidates may be delayed and we may be required to expend substantial additional resources.
Even if we obtain regulatory approval of one or more of our current or future product candidates, they may never achieve market acceptance or commercial success.
If we obtain FDA, USDA or EMA approvals for one or more of our current or future product candidates, they may not achieve market acceptance among veterinarians and pet owners and may not be commercially successful. Market acceptance of any of our current or future product candidates for which we may receive approval depends on a number of factors, including:
•the indications for which our products are approved;
•the potential and perceived advantages of our product candidates over alternative treatments, including generic medicines and competing products currently prescribed by veterinarians, and products approved for use in humans that are used extra-label in animals;
•the cost of treatment in relation to alternative treatments and willingness on the part of veterinarians and pet owners to pay for our products, including other discretionary items, especially during economically challenging times;
•the prevalence and severity of any adverse side effects of our products;
•the relative convenience and ease of administration of our products;
•the effectiveness of our sales and marketing efforts; and
•the proper training and administration of our products by veterinarians and acceptance by veterinarians and pet owners of our products as safe and effective.
Any failure by our product candidates that obtain regulatory approval to achieve market acceptance or commercial success would adversely affect our financial condition and results of operations.
Pet therapeutics, like human therapeutics, are subject to unanticipated post-approval safety or efficacy concerns, which may harm our business and reputation.
The success of our commercialization efforts will depend upon the perceived safety and effectiveness of pet therapeutics, in general, and of our products, in particular. Unanticipated safety or efficacy concerns can arise with respect to approved pet therapeutics after they enter into commerce, which may result in product recalls or withdrawals or suspension of sales, as well as product liability and other claims. Because reliable detection of rare events might require exposure of millions of animals, it is not possible to rule out the risk until well after the launch of the product.
It is also possible that the occurrence of significant adverse side effects in approved human generic compounds upon which our product candidates are based could impact our products. Any safety or efficacy concerns, or recalls, withdrawals or suspensions of sales of our products or other pet therapeutics, or of their human equivalents, could harm our reputation, in particular, or pet therapeutics, generally, and materially, adversely affect our business and prospects or the potential growth of the pet therapeutics industry, regardless of whether such concerns or actions are justified.
Future federal and state legislation may result in increased exposure to product liability claims, which could result in substantial losses to us.
Under current federal and state laws, pets are generally considered to be personal property of their pet owners and, as such, pet owners’ recovery for product liability claims involving their pets may be limited to the replacement value of the pets. Pet owners and their advocates, however, have filed lawsuits from time to time seeking non-economic damages such as pain and suffering and emotional distress for harm to their pets based on theories applicable to personal injuries to humans. If new legislation is passed to allow recovery for such non-economic damages, or if precedents are set allowing for such recovery, we could be exposed to increased product liability claims that could result in substantial losses to us if successful. In addition, some horses can be worth millions of dollars or more, and product liability for horses may be very high.
It is also possible that our product liability insurance will not be sufficient to cover any future product liability claims against us.
We rely on third parties to provide us with certain supplies and services relating to the manufacture of our products and product candidates and are subject to increased labor and material costs and potential disruptions in supply.
Certain supplies obtained by us from third parties relating to the manufacture of our products and product candidates may be subject to availability constraints and price volatility caused by changes in demand, weather conditions, supply conditions, government regulations, economic climate and other factors, including any impacts caused by the COVID-19 pandemic. In addition, labor costs may be subject to volatility caused by the supply of labor, governmental regulations, economic climate and other factors. Increases in the demand for, availability or the price of, such supplies and increases in labor costs could increase the costs to manufacture our products and product candidates, result in product delivery delays or shortages, and impact our ability to launch new products on a timely basis or at all. We may not be able to pass all or a material portion of any higher supply or labor costs on to our customers, which could materially adversely affect our operating results and financial condition.
Certain third-party suppliers are the sole or exclusive source of certain supplies and services necessary for the manufacture of our products or product candidates. We may be unable to meet demand for certain of our products or manufacture certain of our product candidates if any of our third-party suppliers cease or interrupt operations, fail to renew contracts with us or otherwise fail to meet their obligations to us.
Biologics manufacturing is difficult and costly and may not be commercially viable.
There are no established sources of the active ingredients in our biologic product candidates, so we will be required to develop and validate the manufacturing process for pet biologics. Manufacturing of pet biologics, apart from vaccines, is a relatively new field in which unanticipated difficulties or challenges could arise. Small changes in the manufacturing process can have significant impact on product quality, consistency and yield. Manufacturing biologics, especially in large quantities, is complex and may require the use of innovative technologies that we may need to develop ourselves or in conjunction with third-party collaborators. Such manufacturing requires facilities specifically designed and validated for this purpose and sophisticated quality assurance and quality control procedures. Biologics are also usually costly to manufacture, because production usually requires the use of living organisms. Factors such as these may make it more technically challenging, time-consuming and expensive than we anticipate to manufacture biologics. Animal antibodies also must be manufactured at a sufficiently low cost that they are economically viable for us and for our customers. We built a manufacturing plant for biologics manufacturing and if it is not utilized to full capacity, it may result in increased annual costs and increase in cost of goods. There is no assurance that we will be able to manufacture biologics at an economical cost, if at all.
Our facilities used to manufacture the biologics are subject to inspections by the FDA, USDA and the EMA. If we cannot successfully manufacture material in compliance with these strict regulatory requirements, we will not be able to secure or maintain regulatory approval for the manufacturing facility. If the FDA or a comparable foreign regulatory authority does not approve the manufacturing facilities, or if it withdraws any such approval in the future, we may need to find alternative manufacturing facilities, which could result in delays in, or adversely affect our ability to, develop or commercialize our product candidates. We also may be subject to penalties and sanctions from the FDA and other regulatory authorities for any violations of applicable regulatory requirements. The USDA and EMA employ different regulatory standards than the FDA, so we may require multiple manufacturing processes and facilities for the same product candidate or any approved product.
There is no assurance that our strategic restructuring transition to a partnership-based model for the commercialization of our biologic products will be successful or that it will result in greater profits for us than if we directly sold these biologic products.
As we announced on March 16, 2020, our strategic restructuring involves a transition to a partnership-based model for commercialization in which our canine and feline biologic products will be out-licensed to larger commercial partners in return for upfront payments, contingent milestone payments and royalties on future sales. The strategic restructuring involves discontinuing our canine, feline and equine commercial infrastructure.
There is no assurance that our transition to a partnership-based model for commercialization of our canine and feline biologic products will be successful or that it will result in greater profits for us than if we directly sold these biologic products. We may be unable to enter into licensing agreements that contain favorable upfront payments, contingent milestone payments and royalties. In addition, there is no assurance that the parties to which we out-license our biologic products will fulfill the terms of their agreements or be successful in their efforts to sell our products at favorable prices and in satisfactory amounts.
There is no assurance as to the amount of revenue that we will receive with respect to our equine assets.
We previously announced we were evaluating various options for our equine products candidates, including a possible spin-out or other divesture of our equine assets. We have completed our strategic evaluation, and there is no assurance that we will be successful in developing or implementing a profitable strategy for our equine assets.
Furthermore, in December 2020, we entered into a distribution and licensing agreement with Dechra in which we granted Dechra an exclusive license to promote, market, sell and distribute Zimeta, although there is no assurance as to the amount of revenue that we will ultimately receive from that agreement.
Even if we successfully identify product candidates, they may be unsuccessfully commercialized for many reasons.
Even if we successfully identify product candidates, they may be unsuccessfully commercialized for many reasons, including the following:
•a product candidate may be covered by third parties’ patents or other exclusive rights unknown to us;
•a product candidate may on further study be shown to have harmful side effects in pets or other characteristics that indicate it is unlikely to be effective or otherwise does not meet applicable regulatory criteria;
•a product candidate may not be capable of being produced in commercial quantities at an acceptable cost, or at all;
•a product candidate may not be accepted as safe and effective by veterinarians, pet owners and the pet therapeutic community; and
•competitors may develop alternatives that render our product candidates obsolete.
Failure to identify further product candidates ultimately suitable for development and commercialization would have an adverse impact on our growth strategy and future business prospects.
Changes in distribution channels for pet therapeutics may make it more difficult or expensive to distribute our products.
In the United States, pet owners typically purchase their pet therapeutics from their local veterinarians who also prescribe such therapeutics. There is a trend, however, toward increased purchases of pet therapeutics from Internet-based retailers, “big-box” retail stores and other over-the-counter distribution channels, which follows a significant shift in recent years away from the traditional veterinarian distribution channel in the sale of parasiticides and vaccines. It is also possible that pet owners may come to rely increasingly on internet-based animal health information rather than on their veterinarians. We currently expect that our pet therapeutics will be marketed directly to veterinarians, so any reduced reliance on veterinarians by pet owners could materially adversely affect our business and prospects. Pet owners also may substitute human health products for pet therapeutics if the human health products are less expensive or more readily available, which substitution also could adversely affect our business.
Legislation has been or may be proposed in the United States or abroad that would require veterinarians to provide pet owners with written prescriptions and disclosures that the pet owner has the right to fill the prescriptions
through other means. If enacted, such legislation could lead to a reduction in the number of pet owners who purchase their pet therapeutics directly from veterinarians, which also could adversely affect our business.
Consolidation of our customers could negatively affect the pricing of our products.
Veterinarians will be our primary customers for any approved products. In recent years, there has been a trend towards the consolidation of veterinary clinics and animal hospitals. If this trend continues, these large clinics and hospitals could attempt to leverage their buying power to obtain favorable pricing from us and other pet therapeutics companies. Any resulting downward pressure on the prices of any of our approved products could have a material adverse effect on our results of operations and financial condition.
Our research and development relies on evaluations in animals, which is controversial and may become subject to bans or additional regulations.
The evaluation of our product candidates in target animals is required to develop and commercialize our product candidates. Although our animal testing is subject to GLP and GCP requirements, as applicable, animal testing in the human pharmaceutical industry and in other industries has been the subject of controversy and adverse publicity. Some organizations and individuals have sought to ban animal testing or encourage the adoption of additional regulations applicable to animal testing. To the extent that such bans or regulations are imposed, our research and development activities, and by extension our operating results and financial condition, could be materially adversely affected. In addition, negative publicity about animal practices by us or in our industry could harm our reputation among potential customers for our products.
Our products and, if approved, our product candidates may be marketed in the United States only in the target animals and for the indications for which they are approved, and if we want to expand the approved animals or indications, we will need to obtain additional FDA or USDA approvals, which may not be granted.
We and our licensees may market or advertise our products and any of our product candidates that are approved only in the specific species and for treatment of the specific indications for which they were approved, which could limit use of the products by veterinarians and pet owners.
Use of a drug outside its cleared or approved indications in the animal context is known as extra-label use. Under the Animal Medicinal Drug Use Clarification Act of 1994 (the "AMDUCA"), veterinarians are permitted to prescribe extra-label uses of certain approved animal drugs and approved human drugs for animals under certain conditions. Thus, although veterinarians may in the future prescribe and use human-approved products or our products for extra-label uses, we may not promote our products for extra-label uses. If the FDA determines that any of our marketing activities constitute promotion of an extra-label use, it could subject us to regulatory enforcement, which could have an adverse impact on our reputation and potential liability to us.
The commercial potential of a product candidate in development is difficult to predict. The market for our product candidates, or for the pet therapeutics industry as a whole, is uncertain and may be smaller than we anticipate, which could significantly and negatively impact our revenue, results of operations and financial condition.
It is very difficult to estimate the commercial potential of any of our product candidates because of the emerging nature of our industry as a whole. The pet therapeutics market continues to evolve, and it is difficult to predict the market potential for what we believe to be the unmet medical needs of pets. The market will depend on important factors such as safety and efficacy compared to other available treatments, including potential human generic therapeutic alternatives with similar efficacy profiles, changing standards of care, preferences of veterinarians, the willingness of pet owners to pay for such products, and the availability of competitive alternatives that may emerge either during the product development process or after commercial introduction. If the market potential for our product candidates is less than we anticipate due to one or more of these factors, it could negatively impact our business, financial condition and results of operations. Furthermore, the willingness of pet owners to pay for our product candidates, if approved, may be less than we anticipate, and may be negatively affected by overall economic conditions. The current penetration of pet insurance in the United States is low, pet owners are likely to
have to pay for our products, if at all, out-of-pocket, and pet owners may not be willing or able to pay for any approved products of ours.
Our manufacturing services are highly complex, and if we are unable to provide quality and timely services to our customers, it may impact our future capital requirements.
The manufacturing services we offer are highly complex, due in part to strict regulatory requirements. A failure of our quality control systems in our facilities could cause problems to arise in connection with facility operations for a variety of reasons, including equipment malfunction, viral contamination, failure to follow specific manufacturing instructions, protocols and standard operating procedures, problems with raw materials or environmental factors. Such problems could affect production of a single manufacturing run or a series of runs, requiring the destruction of products, or could halt manufacturing operations altogether. In addition, our failure to meet required quality standards may result in our failure to timely deliver products to our customers which, in turn, could damage our reputation for quality and service. Any such incident could, among other things, lead to increased costs, lost revenue, reimbursement to customers for lost drug substance, damage to and possibly termination of existing customer relationships, time and expense spent investigating the cause and, depending on the cause, similar losses with respect to other manufacturing runs. With respect to our commercial manufacturing, if problems are not discovered before the product is released to the market, we may be subject to regulatory actions, including product recalls, product seizures, injunctions to halt manufacture and distribution, restrictions on our operations, civil sanctions, including monetary sanctions, and criminal actions. In addition, such issues could subject us to litigation, the cost of which could be significant.
We may acquire other businesses or form joint ventures that may be unsuccessful and could adversely dilute your ownership of our company.
As part of our business strategy, we may pursue acquisitions of other complementary assets and businesses and may also pursue strategic alliances. Our company has no experience in acquiring other assets or businesses and has limited experience in forming such alliances. We may not be able to successfully integrate any acquisitions into our existing business, and we could assume unknown or contingent liabilities or become subject to possible stockholder claims in connection with any related-party or third-party acquisitions or other transactions. We also could experience adverse effects on our reported results of operations from acquisition-related charges, amortization of acquired technology and other intangibles and impairment charges relating to write-offs of goodwill and other intangible assets from time to time following an acquisition. Integration of an acquired company requires management resources that otherwise would be available for ongoing development of our existing business. We may not realize the anticipated benefits of any acquisition, technology license or strategic alliance.
To finance future acquisitions, we may choose to issue shares of our common stock as consideration, which would dilute your ownership interest in us. Alternatively, it may be necessary for us to raise additional funds through public or private financings. Additional funds may not be available on terms that are favorable to us and, in the case of equity financings, may result in dilution to our stockholders.
Our secured loan agreement contains restrictions that limit our flexibility in operating our business.
On September 30, 2019, we entered into a Loan and Security Agreement (the “Loan Agreement”) with Solar Capital Ltd., as collateral agent and lender, and the other lenders named in the Loan Agreement (Solar Capital Ltd. and the other lenders collectively, the “Lenders”). The Lenders have agreed to make available to KindredBio an aggregate principal amount of up to $50.0 million under the Loan Agreement. The Loan Agreement provides for a term loan commitment of $50.0 million in three tranches: (1) a $20.0 million term A loan that was funded on September 30, 2019; (2) a $15.0 million term B loan that is to be funded at our request, subject to certain conditions described in the Loan Agreement being satisfied, no later than December 31, 2020; and (3) a $15.0 million term C loan that is to be funded at our request, subject to certain conditions described in the Loan Agreement being satisfied, on or before June 30, 2021. Each term loan has a maturity date of September 30, 2024. Each term loan bears interest at a floating per annum rate equal to the one-month LIBOR rate (with a floor of 2.17%) plus 6.75%. We are permitted to make interest-only payments on each term loan through October 31, 2021. The interest-only period can be extended by six months upon our satisfaction of the minimum liquidity requirements described in the Loan Agreement.
The Loan Agreement contains numerous negative covenants that prohibit us from taking certain actions in connection with the operation of our business without obtaining the prior written consent of the Lenders. For example, without the prior written consent of the Lenders, we are not permitted to (1) sell, license or otherwise transfer specified assets or portions of our business, (2) engage in specified new businesses, (3) issue more than a specified amount of securities, (4) merge with another entity, (5) acquire another business, (6) incur additional indebtedness or encumber assets, subject to certain exceptions, (7) pay cash dividends, (8) enter into specified material contracts, or (9) permit third parties to acquire ownership interests in our subsidiaries. There is no assurance that the Lenders will provide their written consent to any of these actions even if the actions are in the best interests of our stockholders.
We may be unable to repay the outstanding principal and accrued interest under the Loan Agreement, in which event the Lenders could exercise their default remedies under the Loan Agreement.
Our obligations under the Loan Agreement are secured by a first-priority security interest in substantially all of our assets, including our intellectual property, and a lien on our real property. The Loan Agreement contains customary representations, warranties and covenants and also includes customary events of default, including payment defaults, breaches of covenants, and a default upon the occurrence of a material adverse change affecting us. Upon the occurrence of an event of default, a default interest rate of an additional 5.00% per annum may be applied to the outstanding loan balance, and the Lenders may declare all outstanding obligations immediately due and payable and exercise all of their rights and remedies as set forth in the Loan Agreement and under applicable law. Among other things, the Lenders could attempt to take possession of and sell substantially all of our assets, which would have a material adverse effect on the market value of our common stock.
There is no assurance that we will be able to repay all outstanding principal and accrued interest under the Loan Agreement. In order to attempt to prevent the occurrence of an event of default under the Loan Agreement, we might be required to take actions that might not be in our long-term best interests such as (1) dedicating a substantial portion of our cash flow from operations to the payment of principal and accrued interest under the Loan Agreement, thereby reducing funds available to us for other purposes, (2) divesting valuable assets in order to raise funds with which to repay the principal and accrued interest under the Loan Agreement, and (3) delaying capital expenditures, new product candidate initiatives and acquisitions of other businesses. The existence of the Loan Agreement and the obligations under the Loan Agreement might also limit our ability to obtain additional equity or debt funding from third parties.
There is no assurance as to the amount of royalties that we will receive from the sale of Mirataz® to Dechra or as to the amount of revenue that we will receive from third-party licensees of our products.
A portion of the consideration to us from the sale of Mirataz® to Dechra will be based on royalties from sales of Mirataz® by Dechra pursuant to the sale agreement that we have entered into with Dechra. The amount of such sales and royalties cannot be predicted by us with any degree of certainty, and the total purchase price that we will receive from Dechra under the sale agreement therefore is uncertain.
There is also no assurance as to the amount of revenue that we will receive from third-party licensees of our products including, without limitation, from our recently announced agreement with Dechra regarding Zimeta and our recently announced agreement with Elanco regarding KIND-030.
Vaxart, Inc. requires additional capital to fund its operations, which may affect Vaxart's ability to pay us for our manufacture of its COVID-19 and other vaccine candidates.
On May 19, 2020, we entered into an agreement with Vaxart, Inc. for the manufacture of Vaxart's oral vaccine candidate for COVID-19. In October 2020, we announced the expansion of our manufacturing agreement with Vaxart for COVID-19 and other vaccine candidates. Total revenue from the partnership is expected to be approximately $20.5 million. Despite contractual obligations, Vaxart's ability to pay us may be subject to Vaxart's requirement for additional capital to fund its operations, as disclosed by Vaxart in its Annual Report on Form 10-K filed with the SEC on February 25, 2021.
Risks Related to Intellectual Property
Our commercial success will depend, in part, on obtaining and maintaining patent protection for our products.
In so far as part of our business strategy is to develop successful human drugs and biologics for veterinary use, our ability to obtain a proprietary intellectual property position for our products is uncertain. We have two issued U.S. patents for our IL-31 antibody, but we do not have an issued patent for Zimeta or any of our other lead product candidates at this time. However, we have filed patent applications covering various aspects of our products and product candidates, and the U.S. Patent and Trademark Office (the “USPTO”) has issued patents for our anti IL-31 antibody (U.S. Patent No. 10,093,731) and for methods of its use (U.S. Patent No. 10,150,810). Our other patent applications may never result in the issuance of patents, and/or patents issued to us may be dominated by the patents of third parties, including, for example, patents issued to analogous human drug or biological compositions and their usages. Furthermore, even if they are unchallenged by third parties, our current or future patents, if issued, may not adequately protect our intellectual property or prevent others from designing around their claims. In order to commercialize our products and product candidates in one or more species, we could be required to enter into third party licenses or, if a license is not available on terms that we consider reasonable, we could be required to cease development or commercialization of one or more of our drug or biologic products or product candidates. Thus, if we cannot obtain ownership of issued patents covering our products and product candidates, our business and prospects would be adversely affected.
It is possible that no patents will issue to us to cover an approved product, and/ or that we will have little to no commercial protection against competing products. In such cases, we would then rely solely on other forms of exclusivity, such as regulatory exclusivity provided by the Federal Food, Drug and Cosmetic Act, if available, which may provide less protection to our competitive position.
Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of any patents that issue. On September 16, 2011, the Leahy-Smith America Invents Act (the “Leahy-Smith Act”) was signed into law. The Leahy-Smith Act includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted, redefine prior art, may affect patent litigation, and switch the U.S. patent system from a “first-to-invent” system to a “first-to-file” system. Under a “first-to-file” system, assuming the other requirements for patentability are met, the first inventor to file a patent application generally will be entitled to the patent on an invention regardless of whether another inventor had made the invention earlier. The USPTO recently developed new regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, and in particular, the first-to-file provisions, only became effective on March 16, 2013. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of any patents that issue, all of which could have a material adverse effect on our business and financial condition.
We may become subject to third parties’ claims alleging infringement of patents and proprietary rights or priority of invention, which would be costly, time-consuming and, if successfully asserted against us, delay or prevent the development and commercialization of our current or future products and product candidates.
There has been substantial litigation regarding patents and other intellectual property rights in the field of therapeutics, as well as patent challenge proceedings, including interference and administrative law proceedings before the USPTO, and oppositions and other comparable proceedings in foreign jurisdictions. Under U.S. patent reform laws, new procedures, including inter partes review and post-grant review, were implemented as of March 16, 2013, and the implementation of such reform laws presents uncertainty regarding the outcome of any challenges to our patents. We are aware of two European patents that claim antibodies that bind to dog IL-31 and feline IL-31with certain functional characteristics, and a European Patent Office opposition decision maintaining one of the patents is currently under appeal. The patents may or may not be relevant to one of our product candidates in Europe. There may be issued patents and pending patent applications with claims directed to long-acting or extended-release pharmaceutical formulations and uses of the same small molecules as in some of our small molecule products or product candidates, and other patents and pending patent applications with claims directed to pharmaceutical formulations and use of biologics conceptually similar to some of our biologics product candidates. There also may be other patents already issued of which we are unaware that might be infringed by one of our current or future products or product candidates. Because patent applications can take many years to issue and may
be confidential for eighteen months or more after filing, there may be applications now pending of which we are unaware and which may later result in issued patents that may be infringed by our current or future products or product candidates. There is no assurance that our current or future products or product candidates will not infringe these or other existing or future third-party patents. In addition, third parties may obtain patents in the future and claim that use of our technologies infringes upon these patents.
To the extent we become subject to future third-party claims against us or our collaborators, we could incur substantial expenses and, if any such claims are successful, we could be liable to pay substantial damages, including treble damages and attorney’s fees if we or our collaborators are found to be willfully infringing a third-party’s patents. If a patent infringement suit were brought against us or our collaborators, we or they could be forced to stop or delay research, development, manufacturing or sales of our product or product candidate that is the subject of the suit. Even if we are successful in defending such claims, infringement and other intellectual property claims can be expensive and time-consuming to litigate and divert management’s attention from our business and operations. As a result of or in order to avoid potential patent infringement claims, we or our collaborators may be compelled to seek a license from a third party for which we would be required to pay license fees or royalties, or both. Moreover, these licenses may not be available on acceptable terms, or at all. Even if we or our collaborators were able to obtain such a license, the rights may be nonexclusive, which could allow our competitors access to the same intellectual property. Any of these events could harm our business and prospects.
In addition to possible infringement claims against us, we may be subject to third-party preissuance submission of prior art to the USPTO, or become involved in opposition, interference, derivation, reexamination, inter partes review, post-grant review, or other patent office proceedings or litigation in the United States or elsewhere, challenging our patent rights or the patent rights of others. We may also become involved in similar opposition proceedings in the European Patent Office or similar offices in other jurisdictions regarding our intellectual property rights with respect to our products and technology. An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our current or future patent rights, if any, allow third parties to commercialize our technology or products and compete directly with us, without payment to us, or result in our inability to manufacture or commercialize products without infringing third-party patent rights.
If our efforts to protect the proprietary nature of the intellectual property related to our products or any of our current or future product candidates are not adequate, we may not be able to compete effectively in our market.
We intend to rely upon a combination of regulatory exclusivity periods, patents, trade secret protection, confidentiality agreements, and license agreements to protect the intellectual property related to our products and our product candidates and our development programs.
Composition-of-matter patents on the active pharmaceutical ingredients in pharmaceutical products, including pet therapeutics, are generally considered to be the strongest form of intellectual property protection, since such patents provide protection without regard to any particular method of use or manufacture. Because our current small molecule product is based on a generic human drug, and because there is little, if any, composition of matter patent protection still available for the API of such drug, we do not seek to obtain such composition-of-matter patents for the API in our small molecule product. In addition, we cannot be certain that all of the composition-of-matter claims in our patent applications for our biologics product candidates will be considered patentable by the USPTO and courts in the United States, or by the patent offices and courts in foreign countries.
Method-of-use patents protect the use of a product for the claimed method. This type of patent does not prevent a competitor from developing or marketing an identical product for an indication that is outside the scope of the issued claims. Moreover, even if competitors do not actively promote their product for our targeted indications for which we may obtain patents, veterinarians may recommend that pet owners use these products extra-label, or pet owners may do so themselves. Although extra-label use may infringe or contribute to the infringement of method-of-use patents, the practice is common and such infringement is difficult to prevent or prosecute.
If the breadth or strength of protection provided by any patent applications or patents we may own, in-license, or pursue with respect to any of our current or future product candidates is threatened, it could threaten our ability to commercialize any of our current or future product candidates. Further, if we encounter delays in our
development efforts, the period of time during which we could market any of our current or future product candidates under any patent protection we obtain would be reduced.
Even where laws provide protection or we are able to obtain patents, costly and time-consuming litigation may be necessary to enforce and determine the scope of our proprietary rights, and the outcome of such litigation would be uncertain. Moreover, any actions we may bring to enforce our intellectual property against our competitors could provoke them to bring counterclaims against us, and some of our competitors have substantially greater intellectual property portfolios than we have.
We also rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or for which we have not filed patent applications, processes for which patents are difficult to enforce and other elements of our product development processes that involve proprietary know-how, information or technology that is not covered by patents. Although we require all of our employees to assign their inventions to us, and endeavor to execute confidentiality agreements with all of our employees, consultants, advisors and any third parties who have access to our proprietary know-how, information or technology, we cannot be certain that we have executed such agreements with all parties who may have helped to develop our intellectual property or had access to our proprietary information, or that our agreements will not be breached. We cannot guarantee that our trade secrets and other confidential proprietary information will not be disclosed or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. If we are unable to prevent material disclosure of the intellectual property related to our technologies to third parties, we will not be able to establish or maintain a competitive advantage in our market, which could materially adversely affect our business, results of operations and financial condition.
Any disclosure to or misappropriation by third parties of our confidential proprietary information could enable competitors to quickly duplicate or surpass our technological achievements, thus eroding our competitive position in our market.
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing our ability to protect our products.
As is the case with other biopharmaceutical companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biopharmaceutical industry involves both technological and legal complexity. Therefore, obtaining and enforcing biopharmaceutical patents is costly, time-consuming and inherently uncertain. In addition, the United States has recently enacted and implemented wide-ranging patent reform legislation. The U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce patents that we might obtain in the future.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on product and product candidates throughout the world would be prohibitively expensive. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where we may obtain patent protection, but where patent enforcement is not as strong as that in the United States. These products may compete with our products in jurisdictions where we do not have any issued or licensed patents and any future patent claims or other intellectual property rights may not be effective or sufficient to prevent them from so competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biopharmaceuticals, which could make it difficult for us to stop the infringement of our patents, if any, or marketing of competing products in violation of our proprietary rights generally. Further, the laws of some foreign countries do
not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant problems in protecting and defending our intellectual property both in the United States and abroad. Proceedings to enforce our future patent rights, if any, in foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business.
We may be involved in lawsuits to protect or enforce any patents issued to us, which could be expensive, time-consuming and unsuccessful.
Competitors may infringe any patents that issue to us, or any patents that we may license. To counter infringement or unauthorized use of any patents we may obtain, we may be required to file infringement claims, which can be expensive and time-consuming to litigate. In addition, if we or one of our future collaborators were to initiate legal proceedings against a third party to enforce a patent covering our current product candidates, or one of our future products, the defendant could counterclaim that the patent is invalid or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a materially misleading statement, during prosecution. Third parties may also raise similar claims before the USPTO, even outside the context of litigation. The outcome following legal assertions of invalidity and unenforceability is unpredictable. We cannot be certain that there is no invalidating prior art, of which we and the patent examiner were unaware during prosecution. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, we would lose at least part, and perhaps all, of any current or future patent protection on our current or future product candidates. Such a loss of patent protection could have a material adverse impact on our business.
Litigation proceedings may fail and, even if successful, may result in substantial costs and distract our management and other employees. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be unsuccessful, it could have an adverse effect on the price of our common stock. Finally, we may not be able to prevent, alone or with the support of our licensors, misappropriation of our trade secrets or confidential information, particularly in countries where the laws may not protect those rights as fully as in the United States.
We have the following seven (7) Registered Trademarks and no pending trademark applications.
We have one registered US trademark for ZIMETA, one International Trademark Registration for ZIMETA, and National Registrations for ZIMETA in Australia, Canada, European Union, New Zealand, and United Kingdom.
The Opposition Period for the Canada ZIMETA National Registration is still active, and therefore it is possible that a third-party opposition may be filed.
We have no registered trademarks for our company name or for various of our current product candidates in the United States or any other countries, and failure to obtain those registrations could adversely affect our business.
While various of our trademarks have been registered, and some of our pending trademark applications have been allowed but not yet registered, various other of our pending trademark applications have neither been allowed nor registered. During trademark registration proceedings, we may receive rejections. If so, we will have an opportunity to respond, but we may be unable to overcome such rejections. In addition, USPTO and comparable agencies in many foreign jurisdictions may permit third parties to oppose pending trademark applications and to seek to cancel registered trademarks. Such opposition proceedings have been filed and are currently pending for three European trademark applications. Current and future opposition or cancellation proceedings, if any, filed against any of our trademark applications or any registered trademarks may result in cancellation of these trademarks. Moreover, any name we propose to use with our product candidates in the United States must be approved by the FDA or the USDA, regardless of whether we have registered or applied to register as a trademark. The FDA typically conducts a review of proposed product names, including an evaluation of potential for confusion with other product names. If the FDA or the USDA objects to any of our proposed proprietary product names, we
may be required to expend significant additional resources in an effort to identify a suitable substitute name that would qualify under applicable trademark laws, not infringe the existing rights of third parties and be acceptable to the FDA or USDA.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties.
We have received confidential and proprietary information from third parties. In addition, we employ individuals who were previously employed at other biotechnology, pharmaceutical or animal health companies. We may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise improperly used or disclosed confidential information of these third parties or our employees’ former employers. Litigation may be necessary to defend against any such claims. Even if we are successful in defending against any such claims, such litigation could result in substantial cost and be a distraction to our management and employees.
Risks Related to Government Regulation
Even after receiving regulatory approval of Mirataz and Zimeta, and even if we receive regulatory approval for any of our current or future product candidates, we will be subject to ongoing FDA, USDA, and EMA obligations and continued regulatory review, which may result in significant additional expense. Additionally, Mirataz, Zimeta and any product candidates, if approved, will be subject to labeling and manufacturing requirements and could be subject to other restrictions. Failure to comply with these regulatory requirements or the occurrence of unanticipated problems with our products could result in significant penalties.
For Mirataz, Zimeta and for any of our current or future product candidates approved by the FDA, USDA, or EMA, the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and recordkeeping for Mirataz, Zimeta and every other approved product will be subject to extensive and ongoing regulatory requirements. These requirements include submissions of safety and other post-marketing information and reports, establishment registration, and product listing, as well as continued compliance with GMP, GLP and GCP for any studies that we conduct post-approval. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with our third-party manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
•restrictions on the marketing or manufacturing of the product, withdrawal of the product from the market, or voluntary product recalls;
•fines, warning letters or holds on target animal studies;
•refusal by the FDA, USDA, or EMA to approve pending applications or supplements to approved applications filed by us or our strategic collaborators, or suspension or revocation of product license approvals;
•product seizure or detention, or refusal to permit the import or export of products; and
•injunctions or the imposition of civil or criminal penalties.
The FDA, USDA, or EMA’s policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the United States or abroad. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained and we may not achieve or sustain profitability, which would adversely affect our business.
Any of our current or future approved products may cause or contribute to adverse medical events that we are required to report to regulatory authorities and, if we fail to do so, we could be subject to sanctions that would materially harm our business.
Certain regulatory authorities will require that we report certain information about adverse medical events regarding an approved product if the product may have caused or contributed to those adverse events. The timing of our obligation to report would be triggered by the date we become aware of the adverse event as well as the nature of the event. We may fail to report adverse events we become aware of within the prescribed timeframe. We may also fail to appreciate that we have become aware of a reportable adverse event, especially if it is not reported to us as an adverse event or if it is an adverse event that is unexpected or removed in time from the use of our product. If we fail to comply with our reporting obligations, the regulatory authorities could take action including criminal prosecution, seizure of our product or delay in approval or clearance of future products.
Legislative or regulatory reforms with respect to pet therapeutics may make it more difficult and costly for us to obtain regulatory clearance or approval of any of our current or future product candidates and to produce, market, and distribute our products after clearance or approval is obtained.
From time to time, legislation is drafted and introduced in the U.S. Congress or EU that could significantly change the statutory provisions governing the testing, regulatory clearance or approval, manufacture, and marketing of regulated products. In addition, FDA and USDA regulations and guidance are often revised or reinterpreted by the FDA and USDA in ways that may significantly affect our business and our products. Similar changes in laws or regulations can occur in other countries. Any new regulations or revisions or reinterpretations of existing regulations in the United States or in other countries may impose additional costs or lengthen review times of any of our current or future product candidates. We cannot determine what effect changes in regulations, statutes, legal interpretation or policies, when and if promulgated, enacted or adopted may have on our business in the future. Such changes could, among other things, require:
•changes to manufacturing methods;
•recall, replacement, or discontinuance of certain products; and
•additional record keeping.
Each of these would likely entail substantial time and cost and could materially harm our financial results. In addition, delays in receipt of or failure to receive regulatory clearances or approvals for any future products would harm our business, financial condition, and results of operations.
Certain of our product candidates currently in development may be classified as controlled substances, the manufacture, use, sale, importation, exportation, and distribution of which are subject to additional regulation by state, federal, and foreign law enforcement and other regulatory agencies.
Certain of our product candidates may be subject to regulation as controlled substances under the federal Controlled Substances Act of 1970, or CSA, and regulations of the U.S. Drug Enforcement Administration, or DEA. The DEA regulates controlled substances as Schedule I, II, III, IV or V substances. Schedule I substances by definition have no established medicinal use and may not be marketed or sold in the United States. An animal drug product may be listed as Schedule II, III, IV or V, with Schedule II substances considered to present the highest risk of abuse and Schedule V substances the lowest relative risk of abuse among such substances.
Various states also independently regulate controlled substances. Though state-controlled substances laws often mirror federal law, because the states are separate jurisdictions, they may separately schedule drugs as well. While some states automatically schedule a drug when the DEA does so, in other states there must be rulemaking or a legislative action. State scheduling may delay commercial sale of any controlled substance drug product for which we obtain federal regulatory approval and adverse scheduling could impair the commercial attractiveness of such product. We would also be required to obtain separate state registrations in order to be able to obtain, handle and distribute controlled substances for target animal studies, and failure to meet applicable regulatory requirements
could lead to enforcement and sanctions from the states in addition to those from the DEA or otherwise arising under federal law.
For any of our product candidates classified as controlled substances, we and our suppliers, manufacturers, contractors, customers and distributors will be required to obtain and maintain applicable registrations from state, federal and foreign law enforcement and regulatory agencies and comply with state, federal and foreign laws and regulations regarding the manufacture, use, sale, importation, exportation and distribution of controlled substances. There is a risk that DEA regulations may limit the supply of the compounds used in pivotal trials of our product candidates, and, in the future, the ability to produce and distribute our products in the volume needed to meet commercial demand.
Regulations associated with controlled substances govern manufacturing, labeling, packaging, testing, dispensing, production and procurement quotas, recordkeeping, reporting, handling, shipment and disposal. These regulations increase the personnel needs and the expense associated with development and commercialization of product candidates containing controlled substances. The DEA and some states conduct periodic inspections of registered establishments that handle controlled substances. Failure to obtain and maintain required registrations or comply with any applicable regulations could delay or preclude us from developing and commercializing our product candidates containing controlled substances and subject us to enforcement action. The DEA may seek civil penalties, refuse to renew necessary registrations or initiate proceedings to revoke those registrations. In some circumstances, violations could lead to criminal proceedings. Because of their restrictive nature, these regulations could limit commercialization of any of our product candidates that are classified as controlled substances.
Risks Related to Our Common Stock
The price of our common stock could be subject to volatility related or unrelated to our operations.
Our stock prices and the market prices for securities of biotechnology companies in general have been highly volatile, with recent significant price and volume fluctuations, and may continue to be highly volatile in the future. Since our initial public offering in December 2013, the trading price of our common stock has ranged from a low of $2.90 to a high of $26.99. The trading price may continue to be subject to wide fluctuations in response to various factors, some of which are beyond our control. These factors include those discussed previously in this “Risk Factors” section of this Annual Report on Form 10-K and others, such as:
•any delays in, or suspension or failure of, our current and future studies;
•announcements of regulatory approval or disapproval of any of our current or future product candidates or of regulatory actions affecting us or our industry;
•delays in the commercialization of our products or product candidates;
•manufacturing and supply issues related to our development programs and commercialization of our product or product candidates;
•quarterly variations in our results of operations or those of our competitors;
•changes in our earnings estimates or recommendations by securities analysts or adverse publicity regarding us or products or product candidates;
•announcements by us or our competitors of new product candidates, significant contracts, commercial relationships, acquisitions or capital commitments;
•announcements relating to future development or license agreements including termination of such agreements;
•adverse developments with respect to our intellectual property rights or those of our principal collaborators;
•commencement of litigation involving us or our competitors;
•any major changes in our board of directors or management;
•new legislation in the United States relating to the prescription, sale, distribution or pricing of pet therapeutics;
•product liability claims, other litigation or public concern about the safety of our products or product candidates;
•market conditions in the animal health industry, in general, or in the pet therapeutics sector, in particular, including performance of our competitors; and
•general economic conditions in the United States and abroad.
In addition, the stock market, in general, or the market for stocks in our industry, in particular, may experience broad market fluctuations, which may adversely affect the market price or liquidity of our common stock. Any sudden decline in the market price of our common stock could trigger securities class-action lawsuits against us. If any of our stockholders were to bring such a lawsuit against us, we could incur substantial costs defending the lawsuit and the time and attention of our management would be diverted from our business and operations. We also could be subject to damages claims if we are found to be at fault in connection with a decline in our stock price.
Our quarterly and annual operating results may be volatile and may vary significantly from the estimates and expectations of investors and third parties, including securities or industry analysts, which could cause the market price of our common stock to decline.
It has been our practice not to provide forward-looking sales, revenue or earnings guidance and not to endorse any third party’s sales, revenue or earnings estimates, including the estimates of securities or industry analysts. As a result, our actual operating results may be below the expectations of our investors and third parties, including securities or industry analysts. Investors should not rely on any estimates, research or reports published by third parties, including analysts. Furthermore, many factors could cause our revenues and operating results to vary significantly in the future, including, but not limited to, those set out in the “Risk Factors” section of this Annual Report on Form 10-K. Accordingly, we believe that quarter-to-quarter comparisons of our operating results are not necessarily meaningful. Investors should not rely on the results of one quarter as an indication of our future performance.
Our principal stockholders and management own a significant percentage of our stock and will be able to exert significant control over matters subject to stockholder approval.
As of December 31, 2020, our executive officers, directors, holders of 5% or more of our capital stock and their respective affiliates beneficially own in the aggregate approximately 39.1% of our outstanding shares of common stock, excluding shares they may acquire upon exercise of stock options or upon the vesting or restricted stock units they hold. As a result of their stock ownership, these stockholders may have the ability to influence our management and policies and will be able to significantly affect the outcome of matters requiring stockholder approval such as elections of directors, amendments of our organizational documents or approvals of any merger, sale of assets or other major corporate transaction. This may prevent or discourage unsolicited acquisition proposals or offers for our common stock that you may feel are in your best interest as one of our stockholders.
Provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable and may lead to entrenchment of management.
Our amended and restated certificate of incorporation and amended and restated bylaws contain provisions that could delay or prevent changes in control or changes in our management without the consent of our board of directors. These provisions include the following:
•a classified board of directors with three-year staggered terms, which may delay the ability of stockholders to change the membership of a majority of our board of directors;
•no cumulative voting in the election of directors, which limits the ability of minority stockholders to elect director candidates;
•the exclusive right of our board of directors to elect a director to fill a vacancy created by the expansion of the board of directors or the resignation, death or removal of a director, which prevents stockholders from being able to fill vacancies on our board of directors;
•the ability of our board of directors to authorize the issuance of shares of preferred stock and to determine the terms of those shares, including preferences and voting rights, without stockholder approval, which could adversely affect the rights of our common stockholders or be used to deter a possible acquisition of our company;
•the ability of our board of directors to amend our bylaws without obtaining stockholder approval;
•the required approval of the holders of at least two-thirds of the shares entitled to vote at an election of directors to adopt, amend or repeal our bylaws or repeal the provisions of our amended and restated certificate of incorporation regarding the election and removal of directors;
•a prohibition on stockholder action by written consent, which forces stockholder action to be taken at an annual or special meeting of our stockholders;
•the requirement that a special meeting of stockholders may be called only by the chairman of the board of directors, the chief executive officer, the president or the board of directors, which may delay the ability of our stockholders to force consideration of a proposal or to take action, including the removal of directors; and
•advance notice procedures that stockholders must comply with in order to nominate candidates to our board of directors or to propose matters to be acted upon at a stockholders’ meeting, which may discourage or deter a potential acquirer from conducting a solicitation of proxies to elect the acquirer’s own slate of directors or otherwise attempting to obtain control of us.
These provisions could inhibit or prevent possible transactions that some stockholders may consider attractive.
We are also subject to the anti-takeover provisions contained in Section 203 of the Delaware General Corporation Law. Under Section 203, a corporation generally may not engage in a business combination with any holder of 15% or more of its capital stock unless the holder has held the stock for three years or, among other exceptions, the board of directors has approved the transaction.
Our stockholder rights agreement could discourage a takeover that stockholders may consider favorable and may lead to an entrenchment of management.
On May 19, 2017, our board of directors approved and adopted a rights agreement with American Stock Transfer & Trust Company, LLC, as rights agent, and, on July 24, 2017, our stockholders approved the adoption of the rights agreement. The rights agreement is intended to protect our stockholders from coercive or otherwise unfair proposals to acquire control of KindredBio by significantly diluting the ownership interest of any person who acquires at least 20% of our outstanding common stock by providing all other stockholders with the right to acquire additional shares of our preferred stock or common stock at a significant discount. Although the rights agreement is intended to encourage an acquiring person to negotiate a proposed merger or other business combination with our board of directors and management, it could discourage a takeover transaction that stockholders may consider favorable and may lead to an entrenchment of management. On May 11, 2020, our Board of Directors extended the expiration date of the rights agreement to May 18, 2023.
Our amended and restated by-laws designate the Court of Chancery of the State of Delaware as the sole and exclusive forum for certain types of actions and proceedings that may be initiated by our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or other employees.
Our amended and restated by-laws provide that, unless we consent in writing to an alternative forum, the Court of Chancery of the State of Delaware will be the sole and exclusive forum for (1) any derivative action or proceeding brought on our behalf, (2) any action asserting a claim of breach of a fiduciary duty owed by any director, officer or other employee to us or our stockholders, (3) any action asserting a claim arising pursuant to any provision of the Delaware General Corporation Law, or (4) any action asserting a claim that is governed by the internal affairs doctrine. Any person purchasing or otherwise acquiring any interest in any shares of our capital stock shall be deemed to have notice of and to have consented to this provision of our amended and restated by-laws. This choice-of-forum provision may limit our stockholders’ ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees, which may discourage such lawsuits. Alternatively, if a court were to find this provision of our amended and restated by-laws inapplicable or unenforceable with respect to one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, which could adversely affect our business and financial condition.
We do not intend to pay dividends on our common stock, and the ability of investors in our common stock to achieve a return on their investment will depend on appreciation in the market price of our common stock.
We currently intend to invest any future earnings to fund our growth and not to pay any cash dividends on our common stock. Since we do not intend to pay dividends, the ability of investors in our common stock to receive a return on their investment will depend on any appreciation in the market price of our common stock. There is no assurance that our common stock will appreciate in price.
General Risk Factors
If we fail to retain current members of our senior management, or to attract and keep additional key personnel, our business and prospects could be materially adversely impacted.
Our success depends on our continued ability to attract, retain and motivate highly qualified management and scientific personnel. We are highly dependent upon our senior management, particularly Richard Chin, M.D., our Chief Executive Officer, Wendy Wee, our Chief Financial Officer and Hangjun Zhan, Ph.D., our Chief Scientific Officer. The loss of services of any of our key personnel could adversely affect our ability to successfully develop our current or future product pipeline and commercialize our product candidates. Although we have entered into employment agreements with these key members of senior management, such agreements generally do not prohibit them from leaving our employ at any time. We currently do not maintain “key man” life insurance on any of our senior management team. The loss of Dr. Chin or other members of our current senior management could adversely affect the timing or outcomes of our current and planned studies, as well as longer-term prospects for commercializing our product candidates. Ms. Wee recently announced her intention to retire as our Chief Financial Officer on August 31, 2021, and we are in the process of identifying a new Chief Financial Officer.
In addition, competition for qualified personnel in the animal health fields is intense, because there is a limited number of individuals who are trained or experienced in the field. We will need to hire additional personnel as we expand our biologics product development activities, and we may not be able to attract and retain qualified personnel on acceptable terms, or at all.
Our information technology systems and the information technology systems of third parties with whom we do business are vulnerable to cyber-attacks, breaches of security and misappropriation of data, which could result in substantial damage to our business and operations.
Our internal computer systems and those of our current and future employees and third-party vendors, manufacturers, licensees and consultants are vulnerable to damage from unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. The secure processing, maintenance and transmission
of electronic information, including customer, employee and company data, is critical to our operations and the legal environment surrounding information security, storage, use, processing, disclosure and privacy is demanding with the frequent imposition of new and changing requirements. We also store certain information with third parties, and we utilize third-party service providers to process, manage or transmit data, which may also increase our risk. Our information systems and those of third parties with whom we do business are subjected to computer viruses or other malicious codes, cyber- or phishing-attacks and also are vulnerable to an increasing threat of continually evolving cybersecurity risks and external hazards, as well as improper or inadvertent employee behavior, all of which could expose confidential company and personal data systems and information to security breaches. Any system failure or security breach by employees or others may pose a risk that sensitive data, including data from our target animal studies, intellectual property, trade secrets, confidential information or personal information belonging to us may be exposed to unauthorized persons or to the public. If such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations. For example, the loss of data from completed or future studies could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. Likewise, we rely on third parties to manufacture our therapeutics and therapeutic candidates, and similar events relating to their computer systems could also have a material adverse effect on our business. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability, the further development of our therapeutic candidates and commercialization of our therapeutics could be delayed, and the trading price of our common stock could be adversely affected. To date, we have not experienced any material impact to our business or operations resulting from security breaches, including from information or cybersecurity attacks; however, because of the frequently changing attack techniques, along with the increased volume and sophistication of the attacks, there is the potential for us to be adversely impacted.
If securities or industry analysts do not publish research or reports about our company, or if they issue adverse or misleading opinions regarding us or our stock, our stock price and trading volume could decline.
Although we have research coverage by securities and industry analysts, if coverage is not maintained, the market price for our common stock may be adversely affected. Our stock price also may decline if any analyst who covers us issues an adverse or erroneous opinion regarding us, our business model, our intellectual property or our stock performance, or if our target animal studies and operating results fail to meet analysts’ expectations. If one or more analysts cease coverage of us or fail to regularly publish reports on us, we could lose visibility in the financial markets, which could cause our stock price or trading volume to decline and possibly adversely affect our ability to engage in future financings.
If we fail to maintain an effective system of disclosure controls and internal control over financial reporting, our ability to produce timely and accurate financial statements or comply with applicable regulations could be impaired.
As a public company, we are subject to the reporting requirements of the Securities Exchange Act of 1934, as amended (the "Exchange Act"), the Sarbanes-Oxley Act of 2002, as amended (the "Sarbanes-Oxley Act"), and the listing standards of the NASDAQ Stock Market. We expect that the requirements of these rules and regulations will continue to increase our legal, accounting and financial compliance costs, make some activities more difficult, time consuming and costly, and place significant strain on our personnel, systems and resources.
The Sarbanes-Oxley Act requires, among other things, that we maintain effective disclosure controls and procedures and internal control over financial reporting. We are continuing to develop and refine our disclosure controls and other procedures that are designed to ensure that information required to be disclosed by us in the reports that we will file with the SEC is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms, and that information required to be disclosed in reports under the Exchange Act is accumulated and communicated to our principal executive and financial officers. We are also continuing to improve our internal control over financial reporting. In order to maintain and improve the effectiveness of our disclosure controls and procedures and internal control over financial reporting, we have expended, and anticipate that we will continue to expend, significant resources, including accounting-related costs and significant management oversight.
Our current controls and any new controls that we develop may become inadequate because of changes in conditions in our business. Further, weaknesses in our disclosure controls or our internal control over financial reporting may be discovered in the future. Any failure to develop or maintain effective controls, or any difficulties encountered in their implementation or improvement, could harm our operating results or cause us to fail to meet our reporting obligations and may result in a restatement of our financial statements for prior periods. Any failure to implement and maintain effective internal control over financial reporting also could adversely affect the results of management evaluations of our internal control over financial reporting that we are required to include in our periodic reports that we file with the SEC. Ineffective disclosure controls and procedures and internal control over financial reporting could also cause investors to lose confidence in our reported financial and other information, which would likely have a negative effect on the trading price of our common stock. In addition, if we are unable to continue to meet these requirements, we may not be able to remain listed on the NASDAQ Stock Market.
Any failure to maintain effective disclosure controls and internal control over financial reporting could have a material and adverse effect on our business and operating results and cause a decline in the price of our common stock.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
Under Section 382 of the Internal Revenue Code of 1986, as amended, if a corporation undergoes an “ownership change,” the corporation’s ability to use its pre-change net operating loss carryforwards and other pre-change tax attributes (such as research and development tax credits) to offset its post-change income and taxes may be limited. In general, an “ownership change” occurs if there is a cumulative change in our ownership by “5% shareholders” that exceeds 50 percentage points over a rolling three-year period. Similar rules may apply under state tax laws. If we experience one or more ownership changes as a result of future transactions in our stock, we may be limited in our ability to use our net operating loss carryforwards and other tax assets to reduce taxes owed on the net taxable income that we earn. Any such limitations on the ability to use our net operating loss carryforwards and other tax assets could potentially result in increased future tax liability to us.
ITEM 1B. UNRESOLVED STAFF COMMENTS.
None.
ITEM 2. PROPERTIES.
We have non-cancelable operating leases for laboratory space in Burlingame, California with several amendments to expand the facility. In February 2020, we further amended non-cancelable operating leases for laboratory space in Burlingame, California for an expansion of an additional 2,260 square feet of laboratory space commencing on May 1, 2020 and expiring on May 31, 2025. The total non-cancellable operating lease for the entire existing laboratory space is 13,736 square feet, expiring May 31, 2025. In August 2015, we entered into a new non-cancelable operating lease for 3,126 square feet of office space in San Diego, California and in June 2019, renewed the lease through February 2025. Our headquarters office lease for 8,090 square feet of office space in Burlingame, California expired November 30, 2020. In September 2020, we renewed our headquarters for only 6,900 square feet of office space for another 3 years, expiring November 30, 2023. In May 2019, we signed another lease in Burlingame, consisting of 1,346 square feet of space through April 2022. In addition, we have five equipment leases expiring through 2027.
On June 21, 2017, we entered into a purchase agreement with Strategic Veterinary Pharmaceuticals, Inc. ("SVP") for the purchase of an approximately 180,000 sq. ft. biologics plant (the "Plant") with clean rooms, utility, equipment, and related quality documentation suitable for small molecule and biologics manufacturing, that is located in Elwood, Kansas. The purchase was finalized on August 7, 2017 upon completion of the diligence period and satisfaction of the conditions of escrow. The Plant was purchased for $3,750,000, which includes approximately eight acres of land located at 1411 Oak Street, Elwood, Kansas, all improvements located at the Plant, and all personal property and intangible property owned by SVP and located at the Plant or used in connection with the operation of the Plant.
ITEM 3. LEGAL PROCEEDINGS.
We are not currently a party to any material legal proceedings.
ITEM 4. MINE SAFETY DISCLOSURES.
Not applicable.
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES.
Market Information
Since December 12, 2013, our common stock has been traded on The NASDAQ Capital Market under the symbol “KIN.” Prior to December 12, 2013, there was no public trading market for our common stock.
Common Stock Information
As of March 10, 2021, there were 41,449,218 outstanding shares of our common stock outstanding held of record by approximately 22 holders.
Dividends
We have never declared or paid any cash dividends on our capital stock. We intend to retain any future earnings and do not expect to pay any dividends in the foreseeable future. Any future determination to declare cash dividends will be made at the discretion of our board of directors, subject to applicable laws, and will depend on a number of factors, including our financial condition, results of operations, capital requirements, contractual restrictions, general business conditions and other factors that our board of directors may deem relevant.
Equity Compensation Plan Information
The following table sets forth certain information as of December 31, 2020 regarding securities authorized for issuance under our equity compensation plans:
| | | | | | | | | | | | | | | | | | | | |
| Plan Category | | Number of securities
to be issued upon
exercise of
outstanding options,
warrants and rights | | Weighted-average
exercise price of
outstanding options,
warrants and rights | | Number of securities
remaining available
for future issuance
under equity
compensation plans
(excluding securities
reflected in column (a)) |
| | (a) | | (b) | | (c) |
| Equity compensation plans approved by stockholders: | | | | | | |
2012 Equity Incentive Plan (terminated) | | 2,586,792 | | | $6.68 | | — | |
2016 Equity Incentive Plan (terminated) | | 1,726,019 | | | $7.63 | | — | |
2018 Equity Incentive Plan | | 2,064,921 | | | $10.01 | | 1,936,422 | |
Equity compensation plans not approved by stockholders | | — | | | — | | | — | |
Total | | 6,377,732 | | | $8.02 | | 1,936,422 | |
Stock Performance Graph
The following line graph presentation compares cumulative total stockholder returns of Kindred Biosciences, Inc. with The NASDAQ Stock Market Index and The NASDAQ Biotechnology Index (the “Peer Index”) for the period from December 31, 2015 to December 31, 2020. The graph and table assume that $100
was invested in each of our common stock, The NASDAQ Stock Market Index and the Peer Index on December 31, 2015, and that all dividends were reinvested. The returns shown are based on historical results and are not intended to suggest future performance. The graph shall not be deemed “soliciting material” or to be “filed” with the SEC for purposes of Section 18 of the Exchange Act or otherwise subject to the liabilities under that Section, and shall not be deemed to be incorporated by reference into any of our filings of under the Securities Act of 1933, as amended, or the Exchange Act.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 12/31/15 | | 6/30/16 | | 12/31/16 | | 6/30/17 | | 12/31/17 | | 6/30/18 | | 12/31/18 | | 6/30/19 | | 12/31/19 | | 6/30/20 | | 12/31/20 |
| Kindred Biosciences, Inc. | 100.00 | | | 104.12 | | | 125.00 | | | 252.94 | | | 277.94 | | | 313.24 | | | 322.06 | | | 244.99 | | | 249.40 | | | 132.05 | | | 126.76 | |
| NASDAQ Composite-Total Returns | 100.00 | | | 97.34 | | | 108.87 | | | 124.89 | | | 141.13 | | | 154.36 | | | 137.12 | | | 166.37 | | | 187.44 | | | 211.20 | | | 271.64 | |
| NASDAQ Biotechnology Index | 100.00 | | | 76.25 | | | 78.65 | | | 92.31 | | | 95.69 | | | 98.69 | | | 87.21 | | | 98.50 | | | 109.11 | | | 124.23 | | | 137.94 | |
Unregistered Sales of Securities
During the fiscal year ended December 31, 2020, we did not sell any securities that were not registered under the Securities Act of 1933, as amended.
Repurchase of Shares
We did not repurchase any of our shares of capital stock during the fiscal year ended December 31, 2020.
ITEM 6. SELECTED FINANCIAL DATA.
The following selected historical information has been derived from the audited consolidated financial statements of Kindred Biosciences and should be read in conjunction with “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Item 8. Financial Statements and Supplementary Data” included elsewhere in this Annual Report on Form 10-K. The Statement of Operations and Comprehensive Loss Data and the Balance Sheet Data are derived from the audited consolidated financial statements which are included in the Form 10-K. The historical results are not necessarily indicative of the results of operations to be expected in the future.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands, except per share amounts) | Years ended December 31, |
| 2020 | | 2019 | | 2018 | | 2017 | | 2016 | | | | |
Statement of Operations and Comprehensive Loss Data: | | | | | | | | | | | | | |
| Revenues: | | | | | | | | | | | | | |
Net product revenues | $ | 878 | | | $ | 4,256 | | | $ | 1,966 | | | $ | — | | | $ | — | | | | | |
Revenue from asset sale | 38,700 | | | — | | | — | | | — | | | — | | | | | |
Partner royalty revenue | 535 | | | — | | | — | | | — | | | — | | | | | |
Contract manufacturing revenue | 1,551 | | | — | | | — | | | — | | | — | | | | | |
Partner licensing revenue | 500 | | | — | | | — | | | — | | | — | | | | | |
| Total revenues | 42,164 | | | 4,256 | | | 1,966 | | | — | | | — | | | | | |
| | | | | | | | | | | | | |
Operating costs and expenses: | | | | | | | | | | | | | |
Cost of product sales (1) | 3,945 | | | 587 | | | 324 | | | — | | | — | | | | | |
Contract manufacturing costs | 681 | | | — | | | — | | | — | | | — | | | | | |
Research and development | 31,281 | | | 28,310 | | | 26,399 | | | 17,665 | | | 13,861 | | | | | |
Selling, general and administrative | 21,979 | | | 37,926 | | | 26,499 | | | 13,988 | | | 8,308 | | | | | |
Restructuring costs | 4,246 | | | — | | | — | | | — | | | 655 | | | | | |
| Total operating costs and expenses | 62,132 | | | 66,823 | | | 53,222 | | | 31,653 | | | 22,824 | | | | | |
| | | | | | | | | | | | | |
Loss from operations | (19,968) | | | (62,567) | | | (51,256) | | | (31,653) | | | (22,824) | | | | | |
Interest and other income (expense), net | (1,828) | | | 1,178 | | | 1,566 | | | 774 | | | 325 | | | | | |
Net loss | (21,796) | | | (61,389) | | | (49,690) | | | (30,879) | | | (22,499) | | | | | |
| Change in unrealized gains or losses on available-for-sale securities | (1) | | | 24 | | | 20 | | | — | | | 19 | | | | | |
| Comprehensive loss | $ | (21,797) | | | $ | (61,365) | | | $ | (49,670) | | | $ | (30,879) | | | $ | (22,480) | | | | | |
| | | | | | | | | | | | | |
Net loss per share, basic and diluted (2) | $ | (0.55) | | | $ | (1.59) | | | $ | (1.60) | | | $ | (1.23) | | | $ | (1.13) | | | | | |
| Weighted-average number of common shares outstanding, basic and diluted | 39,289 | | | 38,657 | | | 31,001 | | | 25,084 | | | 19,873 | | | | | |
| | | | | | | | | | | | | |
| (1) Includes $3,826 finished goods write-off related to the Dechra agreements signed in 2020. | | | | |
| | | | | | | | | | | | | |
| (2) See Note 14 of the notes to consolidated financial statements included elsewhere in this annual report for an explanation of the method used to calculate the basic and diluted net loss per share attributable to common stockholders and the number of shares used in the computation of the per share amounts. | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands) | As of December 31, |
| 2020 | | 2019 | | 2018 | | 2017 | | 2016 | | | | |
Balance Sheet Data: | | | | | | | | | | | | | |
Cash and cash equivalents | $ | 11,620 | | | $ | 15,986 | | | $ | 56,302 | | | $ | 34,813 | | | $ | 6,687 | | | | | |
Short-term investments | 46,758 | | | 55,723 | | | 17,630 | | | 46,207 | | | 50,068 | | | | | |
Long-term investments | 1,500 | | | 1,837 | | | — | | | 1,499 | | | 1,052 | | | | | |
Working capital | 55,728 | | | 69,121 | | | 64,888 | | | 75,790 | | | 54,170 | | | | | |
Total assets | 95,814 | | | 114,024 | | | 106,482 | | | 90,822 | | | 61,576 | | | | | |
Total liabilities | 28,332 | | | 32,103 | | | 15,275 | | | 6,142 | | | 3,896 | | | | | |
Accumulated deficit | (244,855) | | | (223,059) | | | (161,670) | | | (111,980) | | | (81,101) | | | | | |
Total stockholders' equity | $ | 67,482 | | | $ | 81,921 | | | $ | 91,207 | | | $ | 84,680 | | | $ | 57,680 | | | | | |
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS.
Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and the related notes and other financial information included elsewhere in this Annual Report on Form 10-K. Some of the information contained in this discussion and analysis, including information with respect to our plans and strategy for our business, includes forward-looking statements that involve risks and uncertainties. You should review the “Risk Factors” section of this Annual Report for a discussion of important factors that could cause our actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis or elsewhere in this Annual Report.
Overview
We are a commercial-stage biopharmaceutical company focused on saving and improving the lives of pets. Our mission is to bring to our pets the same kinds of safe and effective medicines that our human family members enjoy. Our core strategy is to identify targets that have already demonstrated safety and efficacy in humans and to develop therapeutics based on these validated targets for pets, primarily dogs and cats. We believe this approach will lead to shorter development times and higher approval rates than pursuing new, non-validated targets. Our current portfolio includes over 20 product candidates in development, predominantly biologics. We also have state-of-the-art biologics manufacturing capabilities and a broad intellectual property portfolio.
Our first product, Mirataz® was approved in May 2018 and became commercially available to veterinarians in the United States in July 2018. In November 2019, our second product, Zimeta™ (dipyrone injection) for the control of fever in horses was approved by the FDA and became commercially available in December 2019. In addition, we have multiple other product candidates, predominantly biologics, in various stages of development. We believe there are significant unmet medical needs for pets, and that the pet therapeutics segment of the animal health industry is likely to grow substantially as new therapeutics are identified, developed and marketed specifically for pets.
Mirataz is the first and only transdermal medication specifically developed and FDA-approved for the management of weight loss in cats. Weight loss is a serious and potentially fatal condition that represents the leading cause of visits to the veterinarian for cats. Mirataz, which is formulated with the proprietary Accusorb™ technology, is applied topically to the cat’s inner ear (pinna) once a day, providing a more attractive application route compared to oral administration. The product is classified as a weight gain drug and can be used in cats with various underlying diseases associated with unintended weight loss.
Zimeta is the first injectable dipyrone product to receive FDA approval for use in horses. Dipyrone, the active ingredient in Zimeta, is a member of the non-steroidal anti-inflammatory drug (NSAID) class and has a centrally acting mechanism of action on the hypothalamus where fever originates and is regulated.
On March 16, 2020, we entered into an Asset Purchase Agreement whereby we agreed to sell Mirataz, our
transdermal drug for the management of weight loss in cats, to Dechra for a cash purchase price of $43 million, of which $38.7 million will be paid on the closing date and $4.3 million will be paid out of escrow beginning in 12 months assuming no escrow claims, alongside an ongoing royalty on global net sales. The acquisition comprises worldwide marketing rights, intellectual property rights, marketing authorizations and associated regulatory documentation, third party supply contracts related to raw material and manufacture of the finished product, and certain product inventory. On April 15, 2020, we completed the sale of Mirataz to Dechra.
The European Commission granted marketing authorization of Mirataz in December 2019. Dechra, which is based in the United Kingdom, plans to launch Mirataz in the UK and the European Union in 2021, and intends to conduct the necessary regulatory activities to achieve approvals in other key international markets.
Concurrent with the sale of Mirataz, we announced a strategic realignment of our business model whereby we plan to rely more on a partnership-based model for commercialization strategy similar to the traditional human biotech commercialization strategy whereby pipeline assets are partnered with larger commercial partners that can maximize product opportunity in return for upfront payments, contingent milestones, and royalties on future sales. Our focus will be on accelerating our deep pipeline of late-stage biologics candidates in canine and feline markets, while stopping most small molecule development for these species. We believe monoclonal antibodies are the future of veterinary medicine, and represent the greatest opportunity for value creation, given large potential markets for our programs and our competitive advantage in biologics. Accordingly, the companion animal commercial infrastructure was substantially reduced. In connection with this restructuring, we eliminated 53 positions, representing about one-third of our workforce. The eliminated positions primarily relate to the companion animal sales force and research and development for small molecule programs. On June 8, 2020, we announced a second restructuring to eliminate an additional 24 positions to streamline our operations and reduce operating expenditures by prioritizing investment in our highest value, late stage programs, especially the interleukin-31 (IL-31) antibody, interleukin-4 receptor (IL-4R) antibody, and parvovirus antibody programs. We completed our restructuring by eliminating the last 5 remaining positions in the third quarter of 2020 and do not anticipate any further reductions in the foreseeable future.
In December 2020, we entered into a Distribution and Licensing Agreement granting Dechra Veterinary Products, LLC, an exclusive license under our Patents and Marketing Authorizations to promote, market, sell and distribute Zimeta in the U.S. and Canadian territories, through channels of distribution permitted by applicable laws and regulations in the relevant countries forming part of the territory. We are responsible for the performance of the third party manufacturer for Zimeta, including the performance of all of the duties and including but not limited to, timely delivery of product meeting the applicable specifications. In consideration for the exclusive license and manufacturing services to be performed by us, Dechra will pay KindredBio a milestone payment upon achievement of certain sales milestone, a royalty fee and a price per unit of Zimeta.
On May 19, 2020, we entered into an agreement with Vaxart, Inc. for the manufacture of Vaxart's oral vaccine candidate for COVID-19. We recorded contract manufacturing revenue based on the percentage completion of specific milestones for the quarter. In October 2020, we announced the expansion of our manufacturing agreement with Vaxart for COVID-19 and other vaccine candidates.
Biologic Product Development Updates
On March 16, 2020, we announced further prioritization our biologics programs for dogs and cats, which we view as our highest potential assets.
KIND-016, Tirnovetmab (Interleukin-31)
In October 2018, we announced positive topline results from our pilot laboratory effectiveness study of tirnovetmab, KIND-016, a fully caninized, high-affinity monoclonal antibody targeting interleukin-31 (IL-31), for the treatment of atopic dermatitis in dogs. In addition, we announced that the U.S. Patent and Trademark Office has issued a patent (Patent No. 10,093,731) for KindredBio's anti-IL31 antibody.
In July 2019, we reported positive topline results from a pilot field effectiveness study for our IL-31 antibody that confirmed the results from our pilot laboratory study. The manufacturing scale up process proceeded and the pivotal efficacy study of KIND-016 was initiated in December 2020.
Canine atopic dermatitis is an immune-mediated inflammatory skin condition in dogs and is the leading reason owners take their dog to the veterinarian. Atopic dermatitis is a large market, with the leading two products on the market selling over $900 million per year. We are pursuing a multi-pronged approach toward atopic dermatitis, with a portfolio of promising biologics. Our market research tells us there is strong demand for new biological treatments for pruritic dogs, with 70% of veterinarians, and a higher percentage of dermatologists, expressing a need for alternatives to current therapies.
KIND-032
In December 2019 we announced the outcome of a positive pilot laboratory study of KIND-032, a fully caninized monoclonal antibody targeting interleukin-4 (IL-4) receptor, for the treatment of atopic dermatitis in dogs. In the study, 14 laboratory dogs with clinical signs consistent with atopic dermatitis were dosed with placebo or with KIND-032 at two different doses. The Canine Atopic Dermatitis Extent and Severity Index (CADESI) scores were assessed by board-certified veterinary dermatologists who were blinded to treatment assignments. The study demonstrated that KindredBio's antibody was well-tolerated. Although the study was a single-dose study designed primarily to assess safety and pharmacokinetics, evidence of positive efficacy and dose response was observed at Week 1, as measured by CADESI-04. A second pilot study to further assess dosing commenced in the third quarter of 2020.
The IL-4 pathway is a key driver of the inflammation that underlies atopic dermatitis and several other allergic diseases. Unlike KIND-025, which binds to IL-4 and IL-13 circulating in blood, KIND-032 binds to the IL-4 receptor on the surface of immune cells.
KIND-025
On March 24, 2020, we announced positive results from our pilot field efficacy study of KIND-025, a canine fusion protein targeting IL-4 and IL-13, for the treatment of atopic dermatitis in dogs. A higher treatment success rate was observed in the KIND-025 group over the placebo group from week 1 through week 4. Positive efficacy signals were also detected with other endpoints including 20mm or higher reduction from baseline in PVAS score. Cell line development is being continued as we further evaluate this program. The IL-4 and IL-13 pathways are key drivers of the inflammation that underlies atopic dermatitis and other allergic diseases. The IL-4/13 SINK molecule binds to both IL-4 and IL-13 circulating in the blood and inhibits their interactions with their respective receptors, thereby modifying the clinical signs associated with atopic dermatitis. We currently do not have plans to prioritize KIND-025 ahead of our other programs.
KIND-030
In August 2019, we announced positive results from our pilot efficacy study of KIND-030, a chimeric, high-affinity monoclonal antibody targeting canine parvovirus (CPV). This was a 12-dog study, of which 4 dogs were treated prophylactically and 2 dogs were treated after establishment of the infection. All treated dogs survived, compared to none in the applicable placebo group. The effect was seen in both prophylaxis setting, as well as in a treatment setting after establishment of infection. On September 16, 2020, we reported positive results from our pivotal efficacy study of KIND-030 in prevention of parvovirus infection in prophylactic treatment. In the randomized, blinded, placebo-controlled study, KIND-030 was administered to dogs as prophylactic therapy to prevent clinical signs of CPV infection. The primary objectives of the study were met. All of the placebo-control dogs developed parvovirus infection as predefined in the study protocol, while none of the KIND-030 treated dogs developed the disease. Furthermore, the parvovirus challenge resulted in 60% mortality rate in the control dogs compared to 0% mortality rate in the KIND-030 treated dogs.
KIND-030 is being pursued for two indications in dogs: prophylactic therapy to prevent clinical signs of canine parvovirus infection and treatment of established parvovirus infection. The pivotal efficacy study for the treatment indication is expected to complete in the second quarter of 2021. There is no set review timeline at the United States Department of Agriculture Center for Veterinary Biologics. Regulatory approval and review timeline are subject to the typical risks inherent in such a process.
CPV is the most significant cause of viral enteritis in dogs, especially puppies, with mortality rates reportedly as high as 91% if untreated. Banfield Medical records report that at least 250,000 dogs are infected with parvoviruses each year, excluding emergency hospitals, shelters, specialty hospitals or undiagnosed cases. While there are vaccines available for CPV, they have to be administered multiple times and many puppies don't receive the vaccine at all, or don't get the complete series. This will not replace the need for vaccination; it may just change the timing of the vaccination post administration. There are currently no approved or unapproved treatments for CPV. Currently, owners spend up to thousands of dollars for supportive care for dogs infected with CPV.
In December 2020, we announced an agreement granting Elanco Animal Health, Inc. ("Elanco") exclusive global rights to KIND-030. Under the terms of the agreement, KindredBio will receive an upfront payment of $500,000, development milestone payments of up to $16 million upon achievement of certain development, regulatory and manufacturing targets, and sales milestones in an aggregate amount of up to $94 million payable throughout the term of the agreement. Furthermore, royalty payments are to range from the low to high teens. The agreement specifies that KindredBio will supply the licensed product to Elanco, and that Elanco will conduct the necessary regulatory activities to achieve approvals in Europe and other key international markets.
KIND-509
On December 21, 2020, we announced positive results from the pilot field efficacy study of our monoclonal antibody against tumor necrosis factor alpha (anti-TNF antibody) for canine inflammatory bowel disease (IBD). The study was a randomized, blinded, placebo-controlled pilot effectiveness study that enrolled 10 dogs diagnosed with IBD to assess the efficacy and safety of KindredBio's anti-TNFα antibody over a 4-week treatment period. The primary effectiveness variable for this exploratory study was reduction in Canine Inflammatory Bowel Disease Activity Index (CIBDAI) score, which was assessed at Screening and Days 0, 7, 14, 21 and 28. Complete remission, defined as ≥ 75% reduction in average post-dose CIBDAI score from baseline, was achieved in 75% of the anti-TNFα group compared to 17% in the placebo group. The treatment effect was early-onset and durable. At Day 7, the first post-dose visit, 75% of the anti-TNFα treated dogs showed ≥ 75% reduction of CIBDAI score from baseline, compared to 17% in the placebo group. Furthermore, 50% of the anti-TNFα treated dogs achieved and maintained 100% reduction of CIBDAI score from baseline throughout all post-dose visits, whereas none in the placebo group achieved the same result.
IBD is a chronic disease of the gastrointestinal tract and can affect dogs at any age, but is more common in middle-aged and older dogs. The majority of canine IBD cases involve chronic states of diarrhea, vomiting, gastroenteritis, inappetence, and other symptoms, certain of which are cited as among the most frequent disorders impacting dogs. For certain dog breeds, the prevalence of diarrhea exceeds 5%. Existing treatments can have significant drawbacks, including limited diets and excessive antibiotic use, which can lead to owner frustration, lapses in treatment adherence, or poor quality of life for the affected animal.
KIND-510a
In January 2019, we announced positive topline results from our pilot field effectiveness study of KIND-510a, a long-acting feline recombinant erythropoietin being developed for the management of non-regenerative anemia in cats. It has been engineered by KindredBio to have a prolonged half-life compared to endogenous erythropoietin, a protein that regulates and stimulates production of red blood cells. The pivotal efficacy study for KIND-510a was initiated in the fourth quarter of 2019.
On November 25, 2020, KindredBio made the decision to suspend the feline recombinant erythropoietin program following an adverse event. Patient safety is paramount to KindredBio. Given the impact of safety profile on the program's commercial value, and amid the continued rapid growth of the canine dermatitis market, the decision was made to redirect resources toward accelerating the Company's programs for canine atopic dermatitis. KindredBio maintains backup programs for its feline recombinant erythropoietin and may seek to develop these at a later date.
Anemia is a common condition that is estimated to affect millions of older cats. It is often associated with chronic kidney disease, because kidneys produce erythropoietin and chronic kidney disease leads to decreased levels of endogenous erythropoietin. Chronic kidney disease affects approximately half of older cats, making it a leading cause of feline mortality. Human erythropoietins, which are multi-billion-dollar products in the human market, are immunogenic in cats.
KIND-511
KIND-511 is an anti-Tumor Necrosis Factor (“anti-TNF”) treatment for newborn foals. Sick newborn foals, defined as sepsis score ≥ 11 or positive blood culture, are challenging, and difficult to treat and result in
approximately 50% mortality. We have completed a pilot field study in sick or septic foals to assess safety and efficacy of anti-TNF monoclonal antibody, with positive results. By Kaplan-Meier analysis, the difference in survival between the control and placebo groups was statistically significant (p=0.0293). There is currently no FDA-approved therapy. We have placed this program on indefinite hold to focus on accelerating our late-stage biologic candidates in the canine and feline markets.
In addition to the product candidates discussed above, we are in the early stages of development for multiple additional indications, including interleukin antibodies and canine checkpoint inhibitors, with the potential to attain approval for one or more products annually for several years. In all, we have over 20 programs for various indications for dogs and cats.
Equine Product Development Updates
We completed a strategic review of our equine candidates. In December 2020, we entered into a Distribution and Licensing Agreement granting Dechra an exclusive license under our Patents and Marketing Authorizations to promote, market, sell and distribute Zimeta in the US and Canadian territories in return for a royalty on sales and milestone payment upon achievement of a certain sales milestone. We made the decision to discontinue the development of KIND-012 (dipyrone oral gel) and pause the development of KIND-014 to focus on accelerating our late-stage biologic candidates.
Manufacturing
We have constructed a Good Manufacturing Practice, or GMP, biologics manufacturing plant in Burlingame, CA which is fully commissioned. We have proceeded to GMP manufacturing of our feline erythropoietin product candidate in January 2018. In addition, we have completed construction and commissioning of our biologics manufacturing lines in our manufacturing plant in Elwood, Kansas in 2019. The Elwood facility includes approximately 180,000 square feet with clean rooms, utility, equipment, and related quality documentation suitable for biologics and small molecule manufacturing.
Funding
We are a commercial-stage company with two products approved for marketing and sale. We have incurred significant net losses since our inception. We incurred cumulative net losses of $244.9 million through December 31, 2020. These losses have resulted principally from costs incurred in connection with investigating and developing our product candidates, research and development activities and general and administrative costs associated with our operations.
Historically, our funding has been a combination of private and public offerings. From our initial public offering in December 2013 through December 2019, we raised approximately $257.4 million in net proceeds, after deducting underwriting discounts and commissions and offering expenses. On April 8, 2020, we entered into an At Market Offering ("ATM") whereby we may offer and sell shares of our common stock from time to time up to $25 million. Through December 31, 2020, 59,211 shares were sold through the ATM, for total gross proceeds of approximately $298,000. Net proceeds, after deducting underwriting discounts and commissions and offering expense, were approximately $201,000. As of December 31, 2020, we had cash, cash equivalents and investments in available-for-sale securities of $59.9 million.
For the foreseeable future, we expect to continue to incur losses, which will increase significantly from historical levels as we expand our product development activities, seek regulatory approvals for our product candidates and begin to commercialize them if they are approved by the Center for Veterinary Medicine branch, or CVM, of the FDA, the U.S. Department of Agriculture, or USDA, or the European Medicines Agency, or EMA. If we are required to further fund our operations, we expect to do so through public or private equity offerings, debt financings, corporate collaborations and licensing arrangements. We cannot assure you that such funds will be available on terms favorable to us, if at all. Arrangements with collaborators or others may require us to relinquish rights to certain of our technologies or product candidates. In addition, we may never successfully complete development of, obtain adequate patent protection for, obtain necessary regulatory approval, or achieve commercial viability for any of our biologic product candidates. If we are not able to raise additional capital on terms acceptable
to us, or at all, as and when needed, we may be required to curtail our operations, and we may be unable to continue as a going concern.
Financial Overview
Revenues
Our revenues consist of product revenue resulting from the sale of Mirataz for the management of weight loss in cats and Zimeta for the treatment of fever in horses. In addition, our revenues in 2020 also included the sale of our Mirataz asset, partner royalties, contract manufacturing and partner licensing revenue.
Cost of Revenues
Cost of product revenues consists primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, inbound shipping and other third-party logistics costs.
Contract manufacturing costs consist primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, rent, facility costs and related machinery depreciation.
Research and Development Expense
All costs of research and development are expensed in the period incurred. Research and development costs primarily consist of contracted development costs, manufacturing costs, salaries and related expenses for personnel, stock-based compensation expense, regulatory, outside service providers, professional and consulting services, travel costs and materials used in clinical trials and research and development.
We are currently pursuing over 10 indications. We typically use our employee and infrastructure resources across multiple development programs. We track outsourced development costs by development compound but do not allocate personnel or other internal costs related to development to specific programs or development compounds as these expenses are included in personnel costs and other internal costs.
Selling, General and Administrative Expense
Selling, general and administrative expense consists primarily of personnel costs, including salaries, related benefits and stock-based compensation for employees, consultants and directors. Selling, general and administrative expenses also include rent and other facilities costs, conference and sponsorship activities, travel costs, professional fees for legal, accounting and tax, information technology services, business development activities, costs associated with being a public company and other general and commercial business services.
As announced in the strategic realignment of our business model on March 16, 2020, KindredBio transitioned to a partnership-based model for commercialization strategy similar to the traditional human biotech commercialization strategy whereby pipeline assets are partnered with larger commercial partners that can maximize product opportunity in return for upfront payments, contingent milestones, and royalties on future sales. As a result of the sale of our Mirataz asset, our companion animal commercial infrastructure was substantially reduced. Furthermore, the Distribution and Licensing Agreement of Zimeta to Dechra resulted in the dismantling our commercial infrastructure.
Interest and Other Income (Expenses), Net
Consist of interest earned on our cash, cash equivalents and short-term investments, interest expenses on our long-term loan and asset disposals.
Income Taxes
As of December 31, 2020, we had net operating loss carryforwards for federal and state income tax purposes of $211,817,000 and $117,203,000, respectively, which will begin to expire in fiscal year 2032. The
Federal NOL generated after 2017 of $118,704,000 will carryforward indefinitely and be available to offset up to 80% of future taxable income each year. Our management has evaluated the factors bearing upon the realizability of our deferred tax assets, which are comprised principally of net operating loss carryforwards. Our management concluded that, due to the uncertainty of realizing any tax benefits as of December 31, 2020, a valuation allowance was necessary to fully offset our deferred tax assets.
Critical Accounting Policies and Significant Judgments and Estimates
Our management’s discussion and analysis of financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States, or U.S. GAAP. The preparation of our consolidated financial statements and related disclosures requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities, and revenue, costs and expenses and related disclosures during the reporting periods. On an ongoing basis, we evaluate our estimates and judgments, including those described below. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully described in Note 2 of the notes to our consolidated financial statements appearing elsewhere in this document, we believe that the estimates and assumptions involved in the following accounting policies may have the greatest potential impact on our consolidated financial statements.
Revenue Recognition
Our revenues consist of product revenues resulting from the sale of Mirataz and Zimeta, revenue from the sale of our Mirataz asset, the associated partner royalties, revenue from our contract manufacturing service and partner licensing revenue.
In accordance with Accounting Standards Codification ("ASC") 606, we applied the following steps to recognize revenue that reflects the consideration to which we expect to be entitled to receive in exchange for the promised goods or services (See Note 2):
1. Identify the contract with a customer
2. Identify the performance obligations in the contract
3. Determine the transaction price
4. Allocate the transaction price to the performance obligations
5. Determine the satisfaction of performance obligation
Product Revenue
Our product revenues consist of sales of Mirataz and Zimeta. We account for a contract with a customer when there is a legally enforceable contract between us and our customer, the rights of the parties are identified, the contract has commercial substance, and collectability of the contract consideration is probable. Our customers could either be distributors who subsequently resell our products to third parties such as veterinarians, clinics or animal hospitals, licensing partners or other third parties.
Revenue from the sale of our Mirataz asset
On March 16, 2020, we entered into an Asset Purchase Agreement to sell Mirataz to Dechra for a cash purchase price of $43 million. On April 15, 2020, we completed the sale of Mirataz to Dechra and received payment of $38.7 million on the closing date. The remaining $4.3 million will be paid out of escrow beginning in 12 months assuming no escrow claims. We concluded our accounting treatment of the asset sale to Dechra meets the scope of ASC Topic 610-20-15-2, "Gains and Losses from the Derecognition of Nonfinancial Assets". We considered our strategic realignment of our business model whereby we become a biologics-only company focused on accelerating our deep pipeline of late-stage biologics candidates in canine and feline markets. Accordingly, we plan to rely more on a partnership-based model for commercialization strategy whereby pipeline assets are partnered with larger
commercial partners that can maximize product opportunity in return for upfront payment, contingent milestones, and royalties on future sales. Based on the above evaluation in the aggregate, we concluded the proper presentation of the sale of Mirataz to be within operating income as part of revenue. This is presented as a separate line item and described as revenue from asset sale.
Partner Royalties
We recognize royalty revenue in connection with licenses granted under license and development arrangements with partners. Royalties are based upon a percentage of commercial sales of partnered products based on levels of net sales. These sales-based royalties, for which the license was deemed the predominant element to which the royalties relate, are estimated and recognized in the period in which the partners’ commercial sales occur. The royalties are generally reported and payable to us within 45 to 60 days of the end of each calendar quarter in which the commercial sales are made. We base our estimates of royalties earned on actual sales information from our partners when available. If actual royalties received are different than amounts estimated, we would adjust the royalty revenue in the period in which the adjustment becomes known.
Contract Manufacturing Revenue
The manufacturing revenue stream generally represents revenue from the manufacturing of customer product(s). Under a manufacturing contract, a quantity of manufacturing runs is ordered and the product is manufactured according to the customer’s specifications and typically only one performance obligation is included. Each manufacturing run represents a distinct service that is sold separately and has stand-alone value to the customer. The product(s) are manufactured exclusively for a specific customer and have no alternative use. The customer retains control of their product during the entire manufacturing process and can make changes to the process or specifications at their request.
The customer and our project team typically have a timeline on each milestone and duration time. They also have an estimated start and finish date. When the project is moving forward, they constantly change to the actual date to track the project progress. The timing has been shared by both parties. This becomes the most important basis for our revenue recognition.
Because of the timing effect of revenue recognition, billings and cash collections can be recorded into three different ways: billed trade receivables, contract assets (unbilled receivables), and contract liabilities (customer deposits and deferred revenue). Contract assets are recorded when our right to consideration is conditioned on something other than the passage of time. Contract assets are reclassified to trade receivables on the balance sheet when our rights become unconditional. Contract liabilities represent customer deposits and deferred revenue billed and/or received in advance of our fulfillment of performance obligations. Contract liabilities convert to revenue as we perform our obligations under the contract.
Partner Licensing Revenue
Partner licensing revenue consists of revenue that compensates us for services performed, such as formulation, process development, and preparation of pre-clinical and clinical drug product materials under research and development arrangements with partners. Revenues related to research and development are generally recognized as the related services or activities are performed using the output method and in accordance with the contract terms. To the extent that the agreements specify services are to be performed on a fixed basis, revenues are
recognized consistent with the pattern of the work performed. In agreements that specify milestones, we evaluate whether the milestones are considered probable of being achieved and estimate the amount to be included in the transaction price using the most likely amount method. If it is probable that a significant revenue reversal would not occur, the value of the associated milestone is recognized at a point in time. Non-refundable milestone payments related to arrangements under which we have continuing performance obligations would be deferred and recognized over the period of performance. Milestone payments that are not within our control, such as submission for approval to regulators by a partner or approvals from regulators, are not considered probable of being achieved until those submissions are submitted by the customer or approvals are received.
Product Returns
Consistent with the industry practice, we generally offer customers a limited right of return of damaged or expired product that has been purchased directly from us. Our return policy generally allows customers to receive credit for expired products within 90 days after the product’s expiration date. We estimate the amount of our product revenues that may be returned by our customers and record these estimates as a reduction of product revenues in the period the related product revenues are recognized, as well as within accrued liabilities, in the consolidated balance sheets.
Our contract manufacturing customer retains control of their product during the entire manufacturing process and can make changes to the process or specifications at their request. There were no product returns.
Fair Value Measurements
We invest our cash in money market funds, cash deposits and debt instruments of the U.S. government agency securities. In the current market environment, the assessment of the fair value of the debt securities can be difficult and subjective. Accounting Standards Codification, or ASC, 820, “Fair Value Measurements and Disclosure" standard describes a fair value hierarchy based on three levels of inputs, of which the first two are considered observable and the last unobservable, that may be used to measure fair value which are the following:
| | | | | | | | | | | | | | |
| Level 1 | | Quoted prices in active markets for identical assets or liabilities; |
| Level 2 | | Inputs other than Level 1 that are observable, either directly or indirectly, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities; and |
| Level 3 | | Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities. The determination of fair value for Level 3 instruments requires the most management judgment and subjectivity. |
Inventories
We value inventory at the lower of cost or net realizable value. We determine the cost of inventory using the standard-cost method, which approximates actual cost based on a first-in, first-out method. We analyze our inventory levels quarterly and write down inventory subject to expire in excess of expected requirements, or that has a cost basis in excess of its expected net realizable value. These inventory related costs are recognized as cost of product revenues on the accompanying consolidated statements of operations. Currently our inventory consists of finished goods only.
Research and Development
As part of the process of preparing our consolidated financial statements, we are required to estimate accrued research and development expenses. Examples of estimated accrued expenses include fees paid to vendors and clinical sites in connection with our pivotal studies, to CROs in connection with our toxicology studies, and to contract manufacturers in connection with the production of API and formulated drug.
We review new and open contracts and communicate with applicable internal and vendor personnel to identify services that have been performed on our behalf and estimate the level of service performed and the associated costs incurred for the service when we have not yet been invoiced or otherwise notified of the actual cost for accrued expenses. The majority of our service providers invoice us monthly in arrears for services performed or as milestones are achieved in relation to our contract manufacturers. We make estimates of our accrued expenses as of each balance sheet date.
We base our accrued expenses related to pivotal studies on our estimates of the services received and efforts expended pursuant to contracts with vendors, our internal resources, and payments to clinical sites based on enrollment projections. The financial terms of the vendor agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. Payments under some of these contracts depend on factors such
as the successful enrollment of animals and the completion of development milestones. We estimate the time period over which services will be performed and the level of effort to be expended in each period. If the actual timing of the performance of services or the level of effort varies from our estimate, we adjust the related expense accrual accordingly on a prospective basis. If we do not identify costs that have been incurred or if we underestimate or overestimate the level of services performed or the costs of these services, our actual expenses could differ from our estimates. To date, we have not made any material adjustments to our estimates of accrued research and development expenses or the level of services performed in any reporting period presented.
Stock-Based Compensation
We measure stock-based awards at fair value on the date of grant and recognize the corresponding compensation expense of the awards, net of estimated forfeitures, over the requisite service periods, which correspond to the vesting periods of the awards. Generally, we issue stock-based awards with only service-based vesting conditions, and record compensation expense for these awards using the straight-line method. Our intention is to grant stock-based awards with exercise prices equivalent to the fair value of our common stock as of the date of grant.
The fair value of each stock-based award is estimated using the Black-Scholes option-pricing model. The expected volatility is based on the historic volatility of our own stock. The expected terms of our awards have been determined utilizing the “simplified” method, since our historical experience for option grants is not relevant to our expectations for recent grants. The risk-free interest rate is determined by reference to the U.S. Treasury yield curve in effect at the time of grant of the award for time periods approximately equal to the expected term of the award. Expected dividend yield is zero, based on the fact that we have never paid cash dividends and do not expect to pay any cash dividends in the foreseeable future. See Note 10 in Notes to Consolidated Financial Statements for further information.
Results of Operations
The following table summarizes the results of our operations for the periods indicated:
| | | | | | | | | | | | | | | | | |
| (In thousands, except per share amounts) | Years Ended December 31, |
| 2020 | | 2019 | | 2018 |
| Revenues: | | | | | |
Net product revenues | $ | 878 | | | $ | 4,256 | | | $ | 1,966 | |
Revenue from asset sale | 38,700 | | | — | | | — | |
Partner royalty revenue | 535 | | | — | | | — | |
Contract manufacturing revenue | 1,551 | | | — | | | — | |
| Partner licensing revenue | 500 | | | — | | | — | |
| Total revenues | 42,164 | | | 4,256 | | | 1,966 | |
| Operating costs and expenses | | | | | |
Cost of product revenues (1) | 3,945 | | | 587 | | | 324 | |
Contract manufacturing costs | 681 | | | — | | | — | |
Research and development | 31,281 | | | 28,310 | | | 26,399 | |
Selling, general and administrative | 21,979 | | | 37,926 | | | 26,499 | |
Restructuring costs | 4,246 | | | — | | | — | |
| Total operating costs and expenses | 62,132 | | | 66,823 | | | 53,222 | |
| | | | | |
| Loss from operations | (19,968) | | | (62,567) | | | (51,256) | |
| Interest and other income (expenses), net | (1,828) | | | 1,178 | | | 1,566 | |
| Net loss | (21,796) | | | (61,389) | | | (49,690) | |
| Change in unrealized gains or losses on available-for-sale securities | (1) | | | 24 | | | 20 | |
| Comprehensive loss | $ | (21,797) | | | $ | (61,365) | | | $ | (49,670) | |
| | | | | |
Net loss per share attributable to common stockholders, basic and diluted | $ | (0.55) | | | $ | (1.59) | | | $ | (1.60) | |
| | | | | |
Weighted-average number of common shares outstanding, basic and diluted | 39,289 | | | 38,657 | | | 31,001 | |
| | | | | |
| (1) Includes $3,826 finished goods write-off related to the Dechra agreements signed in 2020. |
Revenues
Our product revenues consist of sale of Mirataz and Zimeta, revenue from the sale of our Mirataz asset, the associated partner royalties, revenue from our contract manufacturing service and partner licensing revenue.
Product revenue
Our product revenues consist of sales of Mirataz and Zimeta. Also included in product sale is revenue derived from co-marketing products for our partners in conjunction with sales of Mirataz and Zimeta.
Our net product revenue for the year ended December 31, 2020 decreased by 79.4% to $878,000 compared with $4,256,000 for the same period in 2019. The decrease is mainly due our sale of the Mirataz asset to Dechra in April 2020 compared with twelve months of Mirataz product sales in 2019. Net product revenue for Zimeta in 2020 was $27,000 compared with $127,000 in 2019, reflecting a downturn in equine events and transportation as a result of COVID-19. In conjunction with Mirataz and Zimeta, we also recorded $29,000 in revenue derived from co-marketing products for our partners, Butterfly Networks and Astaria Global in 2020.
Our product sale revenue for the year ended December 31, 2019 increased by 116.5% to $4,256,000 compared with $1,966,000 for the same period in 2018. The increase is mainly due to a full year of Mirataz product
sales while Mirataz only became commercial available to veterinarians in the United States in July 2018. In addition, our second product, Zimeta™ (dipyrone injection) for the control of fever in horses was approved by the FDA and became commercially available in December 2019.
As a result of our licensing agreements with Dechra, we will be recording partner royalty revenues related to these products instead of product revenues going forward.
Revenue from asset sale
Our revenue from the Mirataz asset sale to Dechra for the year ended December 31, 2020 was $38.7 million. The remaining $4.3 million will be paid out of escrow beginning in 12 months assuming no escrow claims. There were no asset sales in 2019 or 2018.
Partner royalties
Our partner royalty revenue for the year ended December 31, 2020 was $535,000, resulting from Dechra's net sales of Mirataz. There were no partner royalty revenues in 2019 or 2018.
Contract manufacturing revenue
On May 19, 2020, we entered into an agreement with Vaxart, Inc. for the manufacture of Vaxart's oral vaccine candidate for COVID-19. We recorded contract manufacturing revenue of $1.6 million based on the percentage completion of specific milestones. We did not have any contract manufacturing revenue in prior years.
Partner licensing revenue
Our partner licensing revenue for the year ended December 31, 2020 was $0.5 million due to a non-refundable upfront payment from Elanco for the exclusive global rights to KIND-030. There were no partner licensing revenues in prior years.
Cost of Revenues
Our cost of product sales for the year ended December 31, 2020 increased by 572.1% to $3,945,000 compared with $587,000 for the same period in 2019. Cost of product revenues consists primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, inbound shipping and other third-party logistics costs. The increase is mainly due to the $3.8 million finished goods write-off related to the Dechra agreements signed in 2020, the majority of which is related to Mirataz. All remaining Mirataz products not included in the transferred assets to Dechra due to the transition to proprietary Dechra brand labelling was written off.
Our cost of product sales for the year ended December 31, 2019 increased by 81.2% to $587,000 compared with $324,000 for the same period in 2018. Cost of product revenues consists primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, inbound shipping and other third-party logistics costs. The increase reflected a full year of Mirataz product sales in 2019.
Our cost of contract manufacturing for the year ended December 31, 2020 was $681,000. Contract manufacturing costs consist primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, rent, facility costs and related machinery depreciation. We did not have any contract manufacturing costs in prior years.
Concentrations of credit risk
Our revenue was generated entirely from sales within the United States. Our product sales to three large distributors, namely MWI Animal Health, Henry Schein (now Covetrus North America) and Midwest Veterinary Supply, each accounted for more than 10% of gross product revenues for the year ended December 31, 2020. On a combined basis, in 2020, these distributors accounted for approximately 78% of our product sales.
Our accounts receivable from amounts billed for contract manufacturing services in 2020 is derived from one customer. The contract requires up-front payment and installment payments during the service period. We perform periodic evaluations of the financial condition of our customers and generally do not require collateral, but we can terminate any contract if a material default occurs.
For the year ended December 31, 2019, our product sales to three large distributors, namely Henry Schein (now Covetrus North America), MWI Animal Health and Patterson Veterinary Supply each accounted for more than 10% of total revenues. On a combined basis, in 2019, these distributors accounted for approximately 85% of our product sales. We did not provide any contract manufacturing services in 2019 or 2018.
Product returns
Our return policy generally allows customers to receive credit for expired products within 90 days after the product’s expiration date. We estimated product return liabilities of 3% for Zimeta of gross revenue using probability-weighted available industry data and data provided by our distributors such as the inventories remaining in the distribution channel. Adjustments will be made in the future if actual results vary from our estimates.
Accounts receivable and allowance for doubtful accounts
Accounts receivable are stated at their carrying values, net of any allowances for doubtful accounts. Accounts receivable consist primarily of amounts due from distributors, our contract manufacturing customer, and licensing partner for which collection is probable based on the customer's intent and ability to pay. Receivables are evaluated for collection probability on a regular basis and an allowance for doubtful accounts is recorded, if necessary. We have no allowance for doubtful accounts as of December 31, 2020 and 2019, as our analysis did not uncover any collection risks.
Research and Development Expense
All costs of research and development are expensed in the period incurred. Research and development costs consist primarily of salaries and related expenses for personnel, stock-based compensation expense, fees paid to consultants, outside service providers, professional services, travel costs and materials used in clinical trials and research and development. We typically use our employee and infrastructure resources across multiple development programs.
Research and development expense was as follows for the periods indicated:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands except percentages) | Years Ended December 31, | | Annual percent change |
| 2020 | | 2019 | | 2018 | | 2020/2019 | | 2019/2018 |
Payroll and related | $ | 10,868 | | | $ | 12,646 | | | $ | 10,204 | | | (14) | % | | 24 | % |
Consulting | 800 | | | 2,406 | | | 2,360 | | | (67) | | | 2 | |
Field trial costs, including materials | 3,097 | | | 4,015 | | | 3,915 | | | (23) | | | 3 | |
Biologics development and supplies | 7,269 | | | 2,526 | | | 3,890 | | | 188 | | | (35) | |
Stock-based compensation | 1,883 | | | 1,848 | | | 1,746 | | | 2 | | | 6 | |
Other | 7,364 | | | 4,869 | | | 4,284 | | | 51 | | | 14 | |
| $ | 31,281 | | | $ | 28,310 | | | $ | 26,399 | | | 10 | % | | 7 | % |
During the year ended December 31, 2020, research and development expense related primarily to advancing the development of KIND-030, KIND-016, KIND-032, KIND-510a and other early stage biologic programs.
Research and development expenses for the year ended December 31, 2020 increased by 10% to $31.3 million compared with $28.3 million for the same period in 2019. The increase was primarily due to the inclusion of expenses from the Kansas facility as it began to manufacture clinical trial material offset by lower costs as we stopped most small molecule development and prioritize our focus on late stage biologic candidates. Prior to 2020, construction and commissioning expenditures associated with the Kansas facility had been categorized as general
and administrative expenses. Outsourced research and development expense related to KIND-030, KIND-510a, KIND-014 and other product development programs for the year ended December 31, 2020 were $1.3 million, $0.8 million, $0.4 million and $0.9 million, respectively.
Research and development expenses for the year ended December 31, 2019 increased by 7% to $28.3 million compared with $26.4 million for the same period in 2018. Payroll and related expenses increased by $2.4 million due to increase in headcount as we advance our biologics development and manufacturing programs. Higher regulatory and research fees, consulting, depreciation, rent as a result of expanded lab spaces and other facility costs also contributed to the increase in expenses, offset by lower non-GMP production and testing expenses. Outsourced research and development expense related to KIND-014, KIND-012, CAD programs, KIND-510a and other product development programs for the year ended December 31, 2019 were $1.7 million, $0.6 million, $0.5 million, $0.4 million and $1.3 million, respectively.
We expect research and development expense to decrease for the foreseeable future. While we expect biologics manufacturing expenses to increase due to the manufacture of KIND-016 and KIND-030 antibodies for our pivotal field studies, our excess manpower and capacity allow us to take on some contract manufacturing services thereby subsidizing the cost of our manufacturing facilities. Labor and associated benefits, supplies and facility expenses related to contract manufacturing services are recorded as cost of goods sold and would offset increases in our biologics manufacturing expenses, resulting in a net decrease in research and development expenses. Due to the inherently unpredictable nature of our development, we cannot reasonably estimate or predict the nature, specific timing or estimated costs of the efforts that will be necessary to complete the development of our product candidates.
Selling, General and Administrative Expense
The composition of general and administrative expense was as follows for the periods indicated:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands except percentages) | Years Ended December 31, | | Annual percent change |
| 2020 | | 2019 | | 2018 | | 2020/2019 | | 2019/2018 |
Payroll and related | $ | 6,294 | | | $ | 15,385 | | | $ | 9,498 | | | (59) | % | | 62 | % |
Consulting, professional and legal fees | 4,115 | | | 3,523 | | | 3,561 | | | 17 | | | (1) | |
Stock-based compensation | 5,704 | | | 5,509 | | | 4,531 | | | 4 | | | 22 | |
Corporate and marketing expenses | 2,705 | | | 5,022 | | | 4,420 | | | (46) | | | 14 | |
Other | 3,161 | | | 8,487 | | | 4,489 | | | (63) | | | 89 | |
| $ | 21,979 | | | $ | 37,926 | | | $ | 26,499 | | | (42) | % | | 43 | % |
Selling, general and administrative expenses for the year ended December 31, 2020 decreased by 42% to $22.0 million compared with $37.9 million for the same period in 2019. The $15.9 million year-over-year decrease was mainly due to the re-categorization of Kansas plant expenditures as research and development expenses, and lower payroll and related expenses including marketing expenses as a result of the elimination of our companion animal sales force. The decrease was offset by higher legal fees.
Selling, general and administrative expenses for the year ended December 31, 2019 increased by 43% to $37.9 million compared with $26.5 million for the same period in 2018. Headcount increase was due to the expansion of our commercial organization and administrative personnel to support the company's growth. Sales and marketing expenses account for a big component of the increase. Higher stock-based compensation expense also contributed to the increase.
We expect selling, general and administrative expense to decrease going forward as a result of our transition to a partnership-focused commercialization strategy whereby pipeline assets are out-licensed to larger commercial partners that can maximize product opportunity in return for upfront payment, contingent milestones, and royalties on future sales.
Restructuring Costs
We recorded restructuring charges of approximately $4.2 million for the year ended December 31, 2020. The restructuring charge of approximately $1.7 million in the first quarter of 2020 was the result of the elimination of 53 positions due to the strategic realignment of our business model whereby we became a biologics-only company while stopping most small molecule development. All charges pertaining to this restructuring have been paid. The restructuring charge of approximately $2.3 million in the second quarter was the result of prioritizing our most attractive late stage programs and substantially reducing our expenses to best position the company for success with the new business model. Another 24 employees were impacted by the restructuring and all restructuring charges have been paid by the third quarter of 2020. We further eliminated another 5 positions in the third quarter and incurred a restructuring charge of approximately $0.3 million related to severance payments and health care benefits, exclusive of stock compensation. We do not anticipate any further reductions in the foreseeable future.
Interest and Other (Expenses) Income, Net
| | | | | | | | | | | | | | | | | |
| (In thousands) | Years Ended December 31, |
| 2020 | | 2019 | | 2018 |
Interest and other (expenses) income, net | $ | (1,828) | | | $ | 1,178 | | | $ | 1,566 | |
The decrease of approximately $3.0 million in 2020 compared to 2019 was the result of $1.4 million lower interest income due to lower interest rate and cash balance. In addition, the change of further impacted by interest expense and amortization of loan issuance costs of $1.7 million.
The decrease of approximately $388,000 in 2019 compared to 2018 was mainly caused by the borrowing on a loan, which incurred approximately $461,000 of interest expenses. In addition, the change was further impacted by disposals of fixed assets of approximately $212,000, offset by an increase in interest income of $339,000 from our investments.
Quarterly Result of Operations
The following table presents selected unaudited quarterly financial data for each of the quarters in the years ended December 31, 2020 and 2019.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands, except per share amounts) | | 2020 | | 2019 |
| Quarter ended | | Dec. 31 | | Sep. 30 | | Jun. 30 | | Mar. 31 | | Dec. 31 | | Sep. 30 | | Jun. 30 | | Mar. 31 |
| | | | | | | | | | | | | | | | |
| Revenues: | | | | | | | | | | | | | | | | |
| Net product revenues | | $ | 96 | | | $ | 16 | | | $ | 163 | | | $ | 603 | | | $ | 1,401 | | | $ | 1,104 | | | $ | 1,236 | | | $ | 515 | |
| Revenue from asset sale | | — | | | — | | | 38,700 | | | — | | | — | | | — | | | — | | | — | |
| Partner royalty revenue | | 122 | | | 255 | | | 158 | | | — | | | — | | | — | | | — | | | — | |
| Contract manufacturing revenue | | 233 | | | 772 | | | 546 | | | — | | | — | | | — | | | — | | | — | |
| Partner licensing revenue | | 500 | | | — | | | — | | | — | | | — | | | — | | | — | | | — | |
| Total Revenues | | 951 | | | 1,043 | | | 39,567 | | | 603 | | | 1,401 | | | 1,104 | | | 1,236 | | | 515 | |
| | | | | | | | | | | | | | | | |
| Operating costs and expenses | | | | | | | | | | | | | | | | |
| Cost of product revenues | | 336 | | | 5 | | | 27 | | | 3,577 | | | 187 | | | 139 | | | 169 | | | 92 | |
| Contract manufacturing costs | | 45 | | | 300 | | | 336 | | | — | | | — | | | — | | | — | | | — | |
| Research and development | | 7,629 | | | 7,387 | | | 7,398 | | | 8,867 | | | 7,134 | | | 7,290 | | | 6,734 | | | 7,152 | |
| Selling, general and administrative | | 3,303 | | | 4,698 | | | 5,105 | | | 8,873 | | | 9,578 | | | 9,382 | | | 9,065 | | | 9,901 | |
| Restructuring costs | | — | | | 282 | | | 2,288 | | | 1,676 | | | — | | | — | | | — | | | — | |
| Total operating costs and expenses | | 11,313 | | | 12,672 | | | 15,154 | | | 22,993 | | | 16,899 | | | 16,811 | | | 15,968 | | | 17,145 | |
| | | | | | | | | | | | | | | | |
| Profit (loss) from operations | | (10,362) | | | (11,629) | | | 24,413 | | | (22,390) | | | (15,498) | | | (15,707) | | | (14,732) | | | (16,630) | |
| Interest and other income (expense), net | | (536) | | | (554) | | | (367) | | | (371) | | | (236) | | | 414 | | | 425 | | | 575 | |
| Net profit (loss) | | $ | (10,898) | | | $ | (12,183) | | | $ | 24,046 | | | $ | (22,761) | | | $ | (15,734) | | | $ | (15,293) | | | $ | (14,307) | | | $ | (16,055) | |
| | | | | | | | | | | | | | | | |
Net profit (loss) per share, basic(1) | | $ | (0.28) | | | $ | (0.31) | | | $ | 0.61 | | | $ | (0.58) | | | $ | (0.40) | | | $ | (0.39) | | | $ | (0.37) | | | $ | (0.42) | |
| Weighted-average number of common shares outstanding, basic | | 39,415 | | | 39,312 | | | 39,240 | | | 39,186 | | | 38,999 | | | 38,940 | | | 38,887 | | | 37,786 | |
Net profit (loss) per share, diluted(1) | | $ | (0.28) | | | $ | (0.31) | | | $ | 0.60 | | | $ | (0.58) | | | $ | (0.40) | | | $ | (0.39) | | | $ | (0.37) | | | $ | (0.42) | |
| Weighted-average number of common shares outstanding, diluted | | 39,415 | | | 39,312 | | | 40,086 | | | 39,186 | | | 38,999 | | | 38,940 | | | 38,887 | | | 37,786 | |
| | | | | | | | | | | | | | | | |
| (1) Net profit (loss) per share for each quarter are calculated as a discrete period, the sum of the quarters may not equal the calculated full year amount. |
Liquidity and Capital Resources
We have incurred losses and negative cash flows from operations since our inception in September 2012. We incurred net losses of $21.8 million, $61.4 million and $49.7 million for the years ended December 31, 2020, 2019, and 2018, respectively. These losses have resulted primarily from costs incurred in research and development activities and selling, general and administrative costs associated with our operations. As of December 31, 2020, we had an accumulated deficit of $244.9 million.
From our initial public offering in December 2013 through December 2019, we raised approximately $257.4 million in net proceeds, after deducting underwriting discounts and commissions and offering expenses. In April 2020, we entered into an At Market Offering ("ATM") whereby we may offer and sell shares of our common stock from time to time up to $25 million. Through December 31, 2020, 59,211 shares were sold through the ATM, for total gross proceeds of approximately $298,000. Net proceeds, after deducting underwriting discounts and commissions and offering expense, were approximately $201,000. As of December 31, 2020, we had cash, cash equivalents and investments in available-for-sale securities of approximately $59.9 million. We believe our cash, cash equivalents and investments along with the net reduction in our workforce, remaining proceeds from the
Mirataz sale, and revenues from royalties, partner licensing and contract manufacturing will be sufficient to fund our planned operations for at least another 24 months. In addition, our ATM facility will provide us with access to additional cash and extend our runway, if required.
The following table shows a summary of our cash flows for the periods set forth below:
| | | | | | | | | | | | | | | | | |
| (In thousands) | Years Ended December 31, |
| 2020 | | 2019 | | 2018 |
Cash flows used in operating activities | $ | (9,857) | | | $ | (56,342) | | | $ | (45,035) | |
Cash flows provided by (used in) investing activities | $ | 5,773 | | | $ | (47,816) | | | $ | 16,604 | |
Cash flows (used in) provided by financing activities | $ | (282) | | | $ | 63,842 | | | $ | 49,920 | |
Net cash used in operating activities
During the year ended December 31, 2020, net cash used in operating activities was $9.9 million. Our net loss of $21.8 million included non-cash charges primarily in the form of share-based compensation of $7.6 million, depreciation expense of $4.7 million, amortization of the debt discount of long-term loan of $348,000, Mirataz and Zimeta finished goods write-off of $3.8 million, loss on disposal of property and equipment of $56,000, partially offset by discounts and amortization of premiums on investments of $28,000. The non-cash charges were further impacted by changes in operating assets and liabilities that resulted in approximately $4.5 million of cash used in operating activities.
During the year ended December 31, 2019, net cash used in operating activities was $56.3 million. Our net loss of $61.4 million included non-cash charges primarily in the form of share-based compensation of $7.4 million, depreciation expense of $2.5 million, amortization of the debt discount of long-term loan of $84,000, shares issued for consulting services of $61,000, and loss on disposal of property and equipment of $212,000, partially offset by discounts and amortization of premiums on investments of $513,000. The non-cash charges were partly impacted by changes in operating assets and liabilities that resulted in approximately $4.7 million of cash provided by operating activities.
During the year ended December 31, 2018, net cash used in operating activities was $45.0 million. Our net loss of $49.7 million included non-cash charges primarily in the form of share-based compensation of $6.3 million, depreciation expense of $805,000, loss on disposal of property and equipment of $34,000, partially offset by discount of premiums on investments of $179,000. The non-cash charges were partly offset by changes in operating assets and liabilities that resulted in $2.3 million of cash used in operating activities.
Net cash provided by (used in) investing activities
During the year ended December 31, 2020, net cash provided by investing activities was $5.8 million, which resulted from $9.3 million related to proceeds of sales and maturities of investments, net of purchases of investments, further impacted by $3.7 million in purchases of property and equipment, of which $58,000 is included in accounts payable and accrued liabilities at December 31, 2020, also positively affected by sale of equipment of $82,000.
During the year ended December 31, 2019, net cash used in investing activities was $47.8 million, which resulted from $39.4 million related to purchase of investments, net of the proceeds of sales and maturities of investments, further impacted by $9.7 million in purchases of property and equipment, of which $1.3 million is included in accounts payable and accrued liabilities at December 31, 2019, also positively affected by sale of equipment of $5,000.
During the year ended December 31, 2018, net cash provided by investing activities was $16.6 million, which resulted from $30.3 million related to the proceeds of sales and maturities of investments, net of purchases of investments, offset by $20.1 million in purchases of property and equipment, of which $6.2 million is included in accounts payable and accrued liabilities at December 31, 2018, also positively affected by sale of equipment of $248,000.
Net cash (used in) provided by financing activities
During the year ended December 31, 2020, net cash used in financing activities consisted of approximately $201,000 in net proceeds from the sale of common stock through an ATM and follow-on public offering, and approximately $383,000 from the exercise of stock options and purchase of ESPP shares, offset by payment of $866,000 related to restricted stock awards tax liability on net settlement.
During the year ended December 31, 2019, net cash provided by financing activities consisted of approximately $43.1 million in net proceeds from the sale of common stock through an ATM and follow-on public offering, net proceeds of $19.2 million from a loan agreement and approximately $2.0 million from the exercise of stock options and purchase of ESPP shares, offset by payment of $493,000 related to restricted stock awards tax liability on net settlement.
During the year ended December 31, 2018, net cash provided by net cash provided by financing activities consisted of approximately $49.2 million in net proceeds from the sale of common stock through an ATM and follow-on public offering, and approximately $1.0 million from stock option exercises and purchase of ESPP shares, offset by payment of $247,000 related to restricted stock awards tax liability on net settlement.
Future Funding Requirements
We anticipate that we will continue to incur losses for the next several years due to expenses relating to:
•pivotal trials of our product candidates;
•toxicology studies for our product candidates; and
•biologics manufacturing.
We believe that our cash, cash equivalents and investments along with the net reduction in our workforce,
remaining proceeds from the Mirataz sale, and revenues from royalties, partner licensing and contract manufacturing will be sufficient to fund our planned operations for at least another 24 months. In addition, our April 8, 2020 ATM facility will provide us with access to additional cash and extend our runway, if required. However, our operating plan may change as a result of many factors currently unknown to us, and we may need to seek additional funds sooner than planned, through public or private equity or debt financings or other sources, such as strategic collaborations. Such financing may result in dilution to stockholders, imposition of debt covenants and repayment obligations or other restrictions that may affect our business. In addition, we may seek additional capital due to favorable market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans.
Our future capital requirements depend on many factors, including, but not limited to:
•the scope, progress, results and costs of researching and developing our current or future product candidates;
•the timing of, and the costs involved in, obtaining regulatory approvals for any of our current or future product candidates;
•the number and characteristics of the product candidates we pursue;
•the cost of manufacturing our current and future product candidates and any products we successfully out-license, including cost of building internal biologics manufacturing capacity;
•the expenses needed to attract and retain skilled personnel;
•the costs associated with being a public company;
•our ability to establish and maintain strategic collaborations, licensing or other arrangements and the financial terms of such agreements; and
•the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing current and future patents, including litigation costs and the outcome of any such litigation.
Contractual Obligations
We have non-cancelable operating leases for two office spaces and expanded laboratory space under which we are obligated to make minimum lease payments totaling $4.1 million through May 2025, the timing of which is described in more detail in the notes to the consolidated financial statements. In addition, we have five operating leases for equipment under which we are obligated to make minimum lease payments totaling $81,000 through 2027.
Off-Balance Sheet Arrangements
Since inception, we have not engaged in the use of any off-balance sheet arrangements, such as structured finance entities, special purpose entities or variable interest entities.
Recently Issued Accounting Pronouncements
In March 2020, the FASB issued ASU No. 2020-04, "Reference Rate Reform (Topic 848)", changes to the interbank offered rates (IBORs), and, particularly, the risk of cessation of the London Interbank Offered Rate ("LIBOR"). The amendments provide optional expedients and exceptions for applying U.S. GAAP to contracts that reference LIBOR expected to be discontinued because of reference rate reform. The expedients and exceptions do not apply to contract modifications made after December 31, 2022. The following optional expedients are permitted for contracts that are modified because of reference rate reform and that meet certain scope guidance: Modifications of contracts within the scope of ASC Topic 470, Debt, should be accounted for by prospectively adjusting the effective interest rate. The amendment also permits an entity to consider contract modifications due to reference rate reform to be an event that does not require contract remeasurement at the modification date or reassessment of a previous accounting determination. When elected, the optional expedients for contract modifications must be applied consistently for all contracts. It applies to all entities within the scope of the affected accounting guidance and will take effect as of March 12, 2020 through December 31, 2022. We have one loan contract which references LIBOR rate. We have not modified the contract with our lenders yet. We are currently evaluating the new guidance and have not determined the impact this standard may have on our financial statements.
We do not believe there are any other recently issued standards not yet effective that will have a material impact on our consolidated financial statements when the standards become effective.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Interest Rate Fluctuation Risk
The primary objective of our investment activities is to preserve capital. We do not utilize hedging contracts or similar instruments.
We are exposed to certain market risks relating primarily to (1) interest rate risk on our cash and cash equivalents, (2) market price risk on our short-term investments, and (3) risks relating to the financial viability of the institutions which hold our capital and through which we have invested our funds. We manage such risks by investing in short-term, liquid, highly-rated instruments. As of December 31, 2020, our cash equivalents, short-term and long-term investments are invested in money market funds, U.S. treasury bills, U.S. treasury bonds, U.S. government agencies, commercial paper and high-grade corporate notes. We do not believe we have any material exposure to interest rate risk due to the extremely low interest rate environment, the short duration of the securities we hold and our ability to hold our investments to maturity if necessary. Declines in interest rates would reduce investment income, but would not have a material effect on our financial condition or results of operations.
We do not currently have exposure to foreign currency risk.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA.
Our consolidated financial statements appear commencing on page F-1 of this Annual Report on Form 10-K, which information is incorporated herein by reference.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE.
None.
ITEM 9A. CONTROLS AND PROCEDURES.
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) that are designed to assure that information required to be disclosed in our Exchange Act reports is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms, and that such information is accumulated and communicated to management, including our Chief Executive Officer and our Chief Financial Officer (the “Certifying Officers"), as appropriate, to allow timely decisions regarding required disclosures.
In designing and evaluating the disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide reasonable assurance only of achieving the desired control objectives, and management necessarily is required to apply its judgment in weighing the costs and benefits of possible new or different controls and procedures. Limitations are inherent in all control systems, so no evaluation of controls can provide absolute assurance that all control issues and any fraud within the company have been detected.
As required by Exchange Act Rule 13a-15(b), as of the end of the period covered by this Annual Report on Form 10-K, management, under the supervision and with the participation of our Certifying Officers, evaluated the effectiveness of our disclosure controls and procedures. Based on this evaluation, the Certifying Officers have concluded that, as of the end of the period covered by this Annual Report on Form 10-K, our disclosure controls and procedures were effective at a reasonable assurance level.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act. Internal control over financial reporting is a process designed by, or under the supervision of, our Chief Executive Officer and Chief Financial Officer and effected by our board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations (COSO). Based on such evaluation, our management concluded that our internal control over financial reporting was effective at a reasonable assurance level as of December 31, 2020.
This Annual Report on Form 10-K does not include an attestation report of our independent registered public accounting firm regarding our internal control over financial reporting, in accordance with applicable SEC rules that permit us to provide only management's report in this Annual Report.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting during the quarter ended December 31, 2020 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. OTHER INFORMATION
None.
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The information required to be disclosed by this item will be contained in our Definitive Proxy Statement for the Annual Meeting of Stockholders to be filed with the SEC no later than April 30, 2021 and is incorporated herein by reference.
ITEM 11. EXECUTIVE COMPENSATION
The information required to be disclosed by this item will be contained in our Definitive Proxy Statement for the Annual Meeting of Stockholders to be filed with the SEC no later than April 30, 2021 and is incorporated herein by reference.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The information required to be disclosed by this item will be contained in our Definitive Proxy Statement for the Annual Meeting of Stockholders to be filed with the SEC no later than April 30, 2021 and is incorporated herein by reference.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The information required to be disclosed by this item will be contained in our Definitive Proxy Statement for the Annual Meeting of Stockholders to be filed with the SEC no later than April 30, 2021 and is incorporated herein by reference.
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES
The information required to be disclosed by this item will be contained in our Definitive Proxy Statement for the Annual Meeting of Stockholders to be filed with the SEC no later than April 30, 2021 and is incorporated herein by reference.
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES.
Our financial statements and related notes thereto are listed and included in this Annual Report on Form 10-K beginning on page F-1. The following exhibits are filed with, or are incorporated by reference into, this Annual Report:
EXHIBIT INDEX
| | | | | | | | |
| Exhibit No. | | Description |
| 2.1 | | |
| 3.1 | | |
| 3.2 | | |
| 3.3 | | |
| 4.1 | | |
| 4.2 | | |
| 4.3 | | |
| 4.4 | | |
| 10.1 | | |
| 10.2 | | |
| 10.3 | | |
| 10.4 | | |
| 10.5 | | |
| 10.6 | | |
| 10.7 | | |
| 10.8 | | |
| | | | | | | | |
| Exhibit No. | | Description |
| 10.9 | | |
| 10.10 | | |
| 10.11 | | |
| 10.12 | | |
| 10.13 | | |
| 10.14 | | |
| 10.15 | | |
| 10.16 | | |
| 10.17 | | |
| 10.18 | | |
| 10.19 | | |
| 10.20 | | |
| 10.21 | | |
| 10.22 | | |
| 10.23 | | |
| 10.24 | | Loan and Security Agreement among Kindred Biosciences, Inc., KindredBio Equine, Inc., Centaur Biopharmaceutical Services, Inc., Solar Capital Ltd., as collateral agent and lender, and the other lenders named therein dated September 30, 2019 (incorporated by reference to Exhibit 10.1 of the registrant's Current Report on Form 8-K filed with the SEC on October 2, 2019). |
| 10.25 | | First Amendment to Loan and Security Agreement among Kindred Biosciences, Inc., KindredBio Equine, Inc., Centaur Biopharmaceutical Services, Inc., Solar Capital, Ltd., as collateral agent and lender, and the other lenders named therein dated March 16, 2020 (incorporated by reference to Exhibit 10.1 of the registrant's Current Report on Form 8-K filed with the SEC on March 16, 2020). |
| | | | | | | | |
| Exhibit No. | | Description |
| 10.26 | | Second Amendment to Loan and Security Agreement among Kindred Biosciences, Inc., KindredBio Equine, Inc., Centaur Biopharmaceutical Services, Inc., Solar Capital, Ltd., as collateral agent and lender, and the other lenders named therein dated December 23, 2020.* |
| 10.27 | | |
| 10.28 | | |
| 10.29 | | |
| 10.30 | | |
| 21.1 | | |
| 23.1 | | |
| 31.1 | | |
| 31.2 | | |
| 32.1 | | |
| 101.INS | | Inline XBRL Instance Document |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema Document |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase Document |
| 101.DEF | | Inline XBRL Taxonomy Extension Definition Linkbase Document |
| 101.LAB | | Inline XBRL Taxonomy Extension Labels Linkbase Document |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase Document |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
| | |
* Filed with this Annual Report on Form 10-K.
† Indicates a management contract or compensatory plan or arrangement.
ITEM 16. FORM 10-K SUMMARY
None
lynSIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | |
| KINDRED BIOSCIENCES, INC. |
| | |
Date: March 16, 2021 | By: | /s/ Wendy Wee |
| | Wendy Wee |
| | Chief Financial Officer and Principal Financial and Accounting Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| | | | | | | | |
Signature | Title | Date |
/s/ Richard Chin | Chief Executive Officer and Director | March 16, 2021 |
Richard Chin, M.D. | (Principal Executive Officer) | |
| | |
/s/ Wendy Wee | Chief Financial Officer | March 16, 2021 |
Wendy Wee | (Principal Financial and Accounting Officer) | |
| | |
/s/ Denise Bevers | Director | March 16, 2021 |
Denise Bevers | | |
| | |
/s/ Lyndon Lien | Director | March 16, 2021 |
Lyndon Lien, Ph.D. | | |
| | |
/s/ Nanxi Liu | Director | March 16, 2021 |
Nanxi Liu | | |
| | |
/s/ Joseph McCracken | Director | March 16, 2021 |
Joseph McCracken, DVM | | |
| | |
/s/ Herbert Montgomery | Director | March 16, 2021 |
Herbert Montgomery | | |
| | |
/s/ Raymond Townsend | Director | March 16, 2021 |
Raymond Townsend, Pharm.D. | | |
| | |
/s/ Ervin Veszprémi | Director | March 16, 2021 |
Ervin Veszprémi | | |
Index to Consolidated Financial Statements
| | | | | |
| Page |
Financial Statements: | |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of Kindred Biosciences, Inc.
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of Kindred Biosciences, Inc. and subsidiaries (the “Company”) as of December 31, 2020 and 2019, the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity and cash flows for each of the three years in the period ended December 31, 2020, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2020 and 2019, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2020, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the consolidated financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of the critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
Revenue Recognition for the Asset Purchase Agreement with Dechra Pharmaceuticals PLC
Critical Audit Matter Description
As described further in Notes 1, 2 and 3 to the consolidated financial statements, in March 2020, the Company entered into an asset purchase agreement (“Asset Purchase Agreement”) whereby the Company agreed to sell its Mirataz asset to Dechra Pharmaceuticals PLC (“Dechra”) for a cash purchase price of $43 million, of which $38.7 million was paid on the closing date and $4.3 million will be paid out of escrow beginning in 12 months from the closing date assuming no escrow claims, alongside ongoing royalty on global net sales. The sale to Dechra comprises the following: worldwide marketing rights; intellectual property rights; marketing authorizations and associated regulatory documentation; third-party supply contracts related to raw material and manufacture of the finished product; and certain product inventory. The Asset Purchase Agreement closed on April 15, 2020. Management was required to use judgment to determine the appropriate guidance to account for the Asset Purchase Agreement as Dechra was not previously a customer of the Company, how to measure the gain related to the transaction, when to derecognize the nonfinancial assets sold to Dechra and the proper presentation of the gain in the consolidated statements of operations. For the year ended December 31, 2020, the Company recognized $38.7 million of revenue from the asset sale in the consolidated statements of operations related to the Asset Purchase Agreement.
Auditing management’s accounting for the Asset Purchase Agreement was challenging due to the complex and judgmental nature of evaluating the terms of the agreement as the Company had not previously entered into a similar transaction, determining the accounting guidance that was applicable to the transaction, evaluating the proper timing of recognition of the variable consideration of $4.3 million, and determining the appropriateness of the presentation of the gain in revenues.
How the Critical Audit Matter Was Addressed in the Audit
We obtained an understanding of management’s assessment of the accounting treatment of the Asset Purchase Agreement through inspection of the agreement and the Company’s technical accounting memo in which the Company evaluated the significant terms of the agreement, the related accounting guidance and its conclusions.
To determine the proper treatment of the accounting for the Asset Purchase Agreement, we evaluated, among other procedures, management’s identification of the relevant performance obligations, management’s allocation of the transaction price to such performance obligations and management’s decision on the accounting for the variable consideration. We also assessed management’s analogies to the accounting for gains and losses from the derecognition of nonfinancial assets, which we determined was a consistent and rational application of accounting policy, as well as management’s conclusion that the transaction did not qualify as discontinued operations and therefore gains from the transaction are appropriate to be included in operating income. Additionally, we assessed management’s conclusion that recording the gain on the transaction as revenues was appropriate as the Company’s business model was realigned to rely on a partnership-based model whereby pipeline assets are partnered with larger commercial companies that can maximize product opportunity in return for upfront payment, contingent milestones and royalties on future sales.
/s/ KMJ Corbin & Company LLP
We have served as the Company's auditor since 2013.
Irvine, California March 16, 2021
Kindred Biosciences, Inc.
Consolidated Balance Sheets
(In thousands, except share and per share amounts)
| | | | | | | | | | | |
| December 31, |
| 2020 | | 2019 |
| ASSETS | | | |
| Current assets: | | | |
| Cash and cash equivalents | $ | 11,620 | | | $ | 15,986 | |
| Short-term investments | 46,758 | | | 55,723 | |
| Accounts, royalty and license receivable | 624 | | | 923 | |
| Inventories | 207 | | | 4,218 | |
| Prepaid expenses and other | 3,415 | | | 2,495 | |
| Total current assets | 62,624 | | | 79,345 | |
| Property and equipment, net | 28,204 | | | 29,777 | |
| Long-term investments | 1,500 | | | 1,837 | |
| Operating lease right-of-use assets | 3,428 | | | 3,001 | |
| Other assets | 58 | | | 64 | |
| Total assets | $ | 95,814 | | | $ | 114,024 | |
| | | |
| LIABILITIES AND STOCKHOLDERS’ EQUITY | | | |
| | | |
| Current liabilities: | | | |
| Accounts payable | $ | 145 | | | $ | 1,256 | |
| Accrued compensation | 2,070 | | | 4,193 | |
| Accrued liabilities | 2,745 | | | 4,131 | |
| Current portion of operating lease liabilities | 825 | | | 644 | |
| Current portion of loan payable | 1,111 | | | — | |
| Total current liabilities | 6,896 | | | 10,224 | |
| Long-term liability | | | |
| Long-term operating lease liabilities | 2,934 | | | 2,614 | |
| Long-term loan payable, net of debt discount | 18,502 | | | 19,265 | |
| Total liabilities | 28,332 | | | 32,103 | |
| | | |
| Commitments and contingencies (Note 12) | | | |
| Stockholders’ equity: | | | |
Common stock; $0.0001 par value; 100,000,000 shares authorized; 39,492,134 shares and 39,203,533 shares issued and outstanding at December 31, 2020 and 2019, respectively | 4 | | | 4 | |
| Additional paid-in capital | 312,321 | | | 304,963 | |
| Accumulated other comprehensive gain | 12 | | | 13 | |
| Accumulated deficit | (244,855) | | | (223,059) | |
| Total stockholders’ equity | 67,482 | | | 81,921 | |
| Total liabilities and stockholders’ equity | $ | 95,814 | | | $ | 114,024 | |
The accompanying notes are integral part of these consolidated financial statements.
Kindred Biosciences, Inc.
Consolidated Statements of Operations and Comprehensive Loss
(In thousands, except per share amounts)
| | | | | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2020 | | 2019 | | 2018 |
| Revenues: | | | | | | |
| Net product revenues | | $ | 878 | | | $ | 4,256 | | | $ | 1,966 | |
| Revenue from asset sale | | 38,700 | | | — | | | — | |
| Partner royalty revenue | | 535 | | | — | | | — | |
| Contract manufacturing revenue | | 1,551 | | | — | | | — | |
| Partner licensing revenue | | 500 | | | — | | | — | |
| Total Revenues | | 42,164 | | | 4,256 | | | 1,966 | |
| | | | | | |
| Operating costs and expenses: | | | | | | |
Cost of product revenues (1) | | 3,945 | | | 587 | | | 324 | |
| Contract manufacturing costs | | 681 | | | — | | | — | |
| Research and development | | 31,281 | | | 28,310 | | | 26,399 | |
| Selling, general and administrative | | 21,979 | | | 37,926 | | | 26,499 | |
| Restructuring costs | | 4,246 | | | — | | | — | |
| Total operating costs and expenses | | 62,132 | | | 66,823 | | | 53,222 | |
| | | | | | |
| Loss from operations | | (19,968) | | | (62,567) | | | (51,256) | |
| Interest and other (expenses) income, net | | (1,828) | | | 1,178 | | | 1,566 | |
| Net loss | | (21,796) | | | (61,389) | | | (49,690) | |
| Change in unrealized gains or losses on available-for-sale securities | | (1) | | | 24 | | | 20 | |
| Comprehensive loss | | $ | (21,797) | | | $ | (61,365) | | | $ | (49,670) | |
| | | | | | |
| Net loss per share, basic and diluted | | $ | (0.55) | | | $ | (1.59) | | | $ | (1.60) | |
| | | | | | |
| Weighted-average number of common shares outstanding, basic and diluted | | 39,289 | | | 38,657 | | | 31,001 | |
| | | | | | |
(1) Includes $3,826 finished goods write-off related to the Dechra agreements signed in 2020. |
The accompanying notes are integral part of these consolidated financial statements.
Kindred Biosciences, Inc.
Consolidated Statements of Changes in Stockholders' Equity
(In thousands)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Common Stock | | Additional Paid-In Capital | | Accumulated Other Comprehensive income (Loss) | | Accumulated Deficit | | Total Stockholders' Equity |
| Shares | | Amount | | | | |
| Balance at December 31, 2017 | 28,183 | | | $ | 3 | | | $ | 196,688 | | | $ | (31) | | | $ | (111,980) | | | $ | 84,680 | |
| Comprehensive loss | | | | | | | | | | | |
| Net loss | — | | | — | | | — | | | — | | | (49,690) | | | (49,690) | |
| Change in unrealized gains on available for sale securities | — | | | — | | | — | | | 20 | | | — | | | 20 | |
| Total comprehensive loss | | | | | | | | | | | (49,670) | |
| Shares withheld related to net share settlement of equity awards | (27) | | | — | | | (247) | | | — | | | — | | | (247) | |
| Stock-based compensation | — | | | — | | | 6,277 | | | — | | | — | | | 6,277 | |
| Exercise of common stock options | 231 | | | — | | | 635 | | | — | | | — | | | 635 | |
At-the-Market issuance of common stock, net of $145 of issuance costs | 188 | | | — | | | 1,758 | | | — | | | — | | | 1,758 | |
Public offering of common stock, net of $3,178 of offering costs | 5,326 | | | — | | | 47,422 | | | — | | | — | | | 47,422 | |
| Common stock issued under ESPP | 47 | | | — | | | 352 | | | — | | | — | | | 352 | |
| Balance at December 31, 2018 | 33,948 | | | 3 | | | 252,885 | | | (11) | | | (161,670) | | | 91,207 | |
| Comprehensive loss | | | | | | | | | | | |
| Net loss | — | | | — | | | — | | | — | | | (61,389) | | | (61,389) | |
| Change in unrealized gains on available for sale securities | — | | | — | | | — | | | 24 | | | — | | | 24 | |
| Total comprehensive loss | | | | | | | | | | | (61,365) | |
| Shares withheld related to net share settlement of equity awards | (21) | | | — | | | (214) | | | — | | | — | | | (214) | |
| RSU Issuance of shares when vested | 51 | | | — | | | (279) | | | — | | | — | | | (279) | |
| Stock-based compensation | — | | | — | | | 7,357 | | | — | | | — | | | 7,357 | |
| Shares issued for consulting services | 8 | | | — | | | 61 | | | — | | | — | | | 61 | |
| Exercise of common stock options | 306 | | | — | | | 1,591 | | | — | | | — | | | 1,591 | |
Public offering of common stock, net of $2,924 of offering costs | 4,847 | | | 1 | | | 43,124 | | | — | | | — | | | 43,125 | |
| Common stock issued under ESPP | 65 | | | — | | | 438 | | | — | | | — | | | 438 | |
| Balance at December 31, 2019 | 39,204 | | | 4 | | | 304,963 | | | 13 | | | (223,059) | | | 81,921 | |
| Comprehensive loss | | | | | | | | | | | |
| Net loss | — | | | — | | | — | | | — | | | (21,796) | | | (21,796) | |
| Change in unrealized loss on available for sale securities | — | | | — | | | — | | | (1) | | | — | | | (1) | |
| Total comprehensive loss | | | | | | | | | | | (21,797) | |
| Shares withheld related to net share settlement of equity awards | (34) | | | — | | | (249) | | | — | | | — | | | (249) | |
| RSU Issuance of shares when vested | 143 | | | — | | | (617) | | | — | | | — | | | (617) | |
| Stock-based compensation | — | | | — | | | 7,587 | | | — | | | — | | | 7,587 | |
| Exercise of common stock options | 69 | | | — | | | 256 | | | — | | | — | | | 256 | |
At-the-Market issuance of common stock, net of $97 of issuance costs | 59 | | | — | | | 201 | | | — | | | — | | | 201 | |
| Common stock issued under ESPP | 51 | | | — | | | 180 | | | — | | | — | | | 180 | |
| Balance at December 31, 2020 | 39,492 | | | $ | 4 | | | $ | 312,321 | | | $ | 12 | | | $ | (244,855) | | | $ | 67,482 | |
The accompanying notes are integral part of these consolidated financial statements.
Kindred Biosciences, Inc.
Consolidated Statements of Cash Flows
(In thousands)
| | | | | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2020 | | 2019 | | 2018 |
| Cash Flows from Operating Activities | | | | | | |
| Net loss | | $ | (21,796) | | | $ | (61,389) | | | $ | (49,690) | |
| Adjustments to reconcile net loss to net cash used in operating activities: | | | | | | |
| Stock-based compensation expense | | 7,587 | | | 7,357 | | | 6,277 | |
| Shares issued for consulting services | | — | | | 61 | | | — | |
| Depreciation and amortization expense | | 4,676 | | | 2,539 | | | 805 | |
| Loss on disposal of property and equipment | | 56 | | | 212 | | | 34 | |
| Amortization of discount on marketable securities | | (28) | | | (513) | | | (179) | |
| Amortization of debt discount of loan payable | | 348 | | | 84 | | | — | |
| Finished goods write off related to Dechra agreements | | 3,826 | | | — | | | — | |
| Changes in operating assets and liabilities: | | | | | | |
| Accounts, royalty and license receivable | | 352 | | | (20) | | | (903) | |
| Inventories | | 185 | | | (648) | | | (3,570) | |
| Prepaid expenses and other | | (914) | | | (825) | | | (867) | |
| Other assets | | — | | | — | | | (21) | |
| Accounts payable | | (1,169) | | | 1,838 | | | 277 | |
| Accrued liabilities and accrued compensation | | (2,980) | | | (5,038) | | | 2,802 | |
| Net cash used in operating activities | | (9,857) | | | (56,342) | | | (45,035) | |
| Cash Flows from Investing Activities | | | | | | |
| Purchases of investments | | (75,666) | | | (125,020) | | | (25,100) | |
| Sales of investments | | — | | | 2,999 | | | 800 | |
| Maturities of investments | | 84,995 | | | 82,628 | | | 54,575 | |
| Purchases of property and equipment | | (3,638) | | | (8,428) | | | (13,919) | |
| Proceeds from sale of property and equipment | | 82 | | | 5 | | | 248 | |
| Net cash provided by (used in) investing activities | | 5,773 | | | (47,816) | | | 16,604 | |
| Cash Flows from Financing Activities | | | | | | |
| Exercise of stock options and purchase of ESPP shares | | 383 | | | 2,029 | | | 987 | |
| Proceeds from loan payable, net of issuance costs | | — | | | 19,181 | | | — | |
| Payment of restricted stock awards tax liability on net settlement | | (866) | | | (493) | | | (247) | |
| Net proceeds from sales of common stock | | 201 | | | 43,125 | | | 49,180 | |
| Net cash (used in) provided by financing activities | | (282) | | | 63,842 | | | 49,920 | |
| Net change in cash and cash equivalents | | (4,366) | | | (40,316) | | | 21,489 | |
| Cash and cash equivalents at beginning of year | | 15,986 | | | 56,302 | | | 34,813 | |
| Cash and cash equivalents at end of year | | $ | 11,620 | | | $ | 15,986 | | | $ | 56,302 | |
| | | | | | |
| Supplemental disclosure of non-cash financing activities: | | | | | | |
| Purchases of property and equipment included in accounts payable and accrued liabilities | | $ | 58 | | | $ | 1,297 | | | $ | 6,205 | |
| Proceeds due from exercise of stock options | | $ | 53 | | | $ | — | | | $ | — | |
| | | | | | |
The accompanying notes are integral part of these consolidated financial statements.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
1. Organization and Description of Business
Kindred Biosciences, Inc. (“we”, "us" or "our") was incorporated on September 25, 2012 (inception) in the State of Delaware. On April 25, 2016, we filed a Certificate of Incorporation with the State of Delaware for a wholly owned subsidiary, KindredBio Equine, Inc. ("KindredBio Equine"). KindredBio Equine has one class of capital stock which is designated common stock, $0.0001 par value per share. The authorized number of shares of common stock for KindredBio Equine is 1,000. On February 1, 2019, we filed a Certificate of Incorporation with the State of Delaware for a wholly owned subsidiary, Centaur Biopharmaceutical Services, Inc. ("Centaur Biopharmaceutical Services"). Centaur Biopharmaceutical Services has one class of capital stock which is designated common stock, $0.0001 par value per share. The authorized number of shares of common stock for Centaur Biopharmaceutical Services is 1,000.
We are a commercial-stage biopharmaceutical company focused on saving and improving the lives of pets. Our activities since inception have consisted principally of raising capital, establishing facilities, recruiting management and technical staff and performing research and development and advancing our product candidates seeking regulatory approval. Our headquarters are in Burlingame, California.
Our first product, Mirataz® (mirtazapine transdermal ointment) was approved in May 2018 and became commercially available to veterinarians in the United States in July 2018. In November 2019, our second product, Zimeta™ (dipyrone injection) for the control of fever in horses was approved by the FDA and became commercially available in December 2019. In addition, we have multiple other product candidates, including several biologics, in various stages of development.
On March 16, 2020, we entered into an Asset Purchase Agreement whereby we agreed to sell Mirataz, our transdermal drug for the management of weight loss in cats, to Dechra Pharmaceuticals PLC ("Dechra") for a cash purchase price of $43 million, of which $38.7 million will be paid on the closing date and $4.3 million will be paid out of escrow beginning in 12 months assuming no escrow claims, alongside an ongoing royalty on global net sales. The acquisition comprises worldwide marketing rights, intellectual property rights, marketing authorizations and associated regulatory documentation, third party supply contracts related to raw material and manufacture of the finished product, and certain product inventory. On April 15, 2020, we completed the sale of Mirataz to Dechra.
Concurrent with the sale of Mirataz, we announced a strategic realignment of our business model whereby we plan to rely more on a partnership-based model for commercialization strategy similar to the traditional human biotech commercialization strategy whereby pipeline assets are partnered with larger commercial partners that can maximize product opportunity in return for upfront payments, contingent milestones, and royalties on future sales. Our focus will be on accelerating our deep pipeline of late-stage biologics candidates in canine and feline markets, while stopping most small molecule development for these species. We believe monoclonal antibodies are the future of veterinary medicine, and represent the greatest opportunity for value creation, given large potential markets for our programs and our competitive advantage in biologics. Accordingly, the companion animal commercial infrastructure will be substantially reduced. In connection with this restructuring, we eliminated 53 positions and recorded a restructuring charge of approximately $1.7 million related to severance payments and health care benefits, exclusive of stock compensation, representing about one-third of our workforce. The eliminated positions primarily relate to the companion animal sales force and research and development for small molecule programs.
On June 8, 2020, we announced a plan to strengthen our strategic position by, among other things, prioritizing our most attractive late stage programs and substantially reducing our expenses to best position the Company for success with the previously announced business model. This restructuring reduced our workforce by approximately 24 employees and resulted in a restructuring charge of approximately $2.3 million related to severance payments and health care benefits, exclusive of stock compensation. We further eliminated another 5 positions and incurred a restructuring charge of approximately $0.3 million related to severance payments and health care benefits in the third quarter of 2020. We have completed our restructuring and do not anticipate any significant further reductions in our workforce for the foreseeable future.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
In December 2020, we announced an agreement granting Elanco Animal Health, Inc. ("Elanco") exclusive global rights to KIND-030. Under the terms of the agreement, KindredBio will receive an upfront payment of $500,000, development milestone payments of up to $16 million upon achievement of certain development, regulatory and manufacturing targets, and sales milestones in an aggregate amount of up to $94 million payable throughout the term of the agreement. Furthermore, royalty payments are to range from the low to high teens. The agreement specifies that KindredBio will supply the licensed product to Elanco, and that Elanco will conduct the necessary regulatory activities to achieve approvals in Europe and other key international markets.
Also in December 2020, we entered into a Distribution and Licensing Agreement granting Dechra an exclusive license under our patents and marketing authorizations to promote, market, sell and distribute Zimeta in the U.S. and Canadian territories. We are responsible for the performance of the third party manufacturer for Zimeta, including the performance of all duties and including but not limited to, timely delivery of product meeting the applicable specifications. In consideration for the exclusive license and manufacturing services to be performed by us, Dechra will pay KindredBio a milestone payment upon achievement of a certain sales milestone, royalty fees based on net Zimeta revenues and a price per unit of Zimeta.
We are subject to risks common to companies in the biotechnology and pharmaceutical industries. There can be no assurance that our research and development will be successfully completed, that adequate protection for our technology will be obtained, that any products developed will obtain necessary government regulatory approval or that any approved products will be commercially viable. We operate in an environment of substantial competition from other animal health companies. In addition, we are dependent upon the services of our employees and consultants, as well as third-party contract research organizations and manufacturers.
The December 2019 outbreak of the novel strain of coronavirus (COVID-19) may adversely impact both our ability to obtain sufficient and timely supplies of our products and other product candidates and our revenue from those products. In addition to adversely affecting our ability to obtain sufficient and timely supplies of products and product candidates from suppliers, any outbreak of contagious diseases, such as the recent novel strain of coronavirus (COVID-19) that is affecting the global community, could adversely affect our business and operations in other ways, many of which cannot currently be determined or quantified. These uncertain factors, including the duration of the outbreak, the severity of the disease and the actions to contain or treat its impact, could impair our operations including, among others, employee mobility and productivity, availability of facilities, conduct of our clinical trials, manufacturing and supply capacity, and availability and productivity of third party service suppliers.
Liquidity
We have incurred losses and negative cash flows from operations and had an accumulated deficit of $244.9 million as of December 31, 2020. We expect to continue to incur losses and negative cash flows, which will increase significantly from historical levels as we expand our product development activities, seek regulatory approvals for our product candidates, establish a biologics manufacturing capability, and commercialize approved products. We might require additional capital until such time as we can generate operating revenues in excess of operating expenses. To date, we have been funded primarily through sales of our equity and recently through an asset sale and licensing of our products. From our initial public offering in December 2013 through December 2019, we raised approximately $257.4 million in net proceeds, after deducting underwriting discounts and commissions and offering expenses. On April 8, 2020, we entered into an At Market Offering ("ATM") whereby we may offer and sell shares of our common stock from time to time up to $25 million. As of December 31, 2020, 59,211 shares were sold through the ATM, for total gross proceeds of approximately $298,000. Net proceeds, after deducting underwriting discounts and commissions and offering expense, were approximately $201,000. As of December 31, 2020, our cash, cash equivalents and investments in available-for-sale securities totaled approximately $59.9 million. We believe our cash, cash
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
equivalents and investments along with the net reduction in our workforce, remaining proceeds from the Mirataz sale, and revenues from royalties, partner licensing and contract manufacturing will be sufficient to fund our planned operations for at least another 24 months. In addition, our ATM facility will provide us with access to additional cash and extend our runway, if required.
If we require additional funding for operations, we may seek such funding through public or private equity or debt financings or other sources, such as corporate collaborations and licensing arrangements. We may not be able to obtain financing on acceptable terms, or at all, and we may not be able to enter into corporate collaborations or licensing arrangements. The terms of any financing may result in dilution or otherwise adversely affect the holdings or the rights of our stockholders. If we are unable to obtain funding, we could be forced to delay, reduce or eliminate our research and development programs or commercialization efforts, which could adversely affect our business prospects.
2. Summary of Significant Accounting Policies
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and include the accounts of the Company and its wholly owned subsidiaries (the "Company"). All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements and related disclosures in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the consolidated financial statements, and the reported amounts of revenues and expenses during the reporting periods. Estimates are based historical experiences or on forecasts, including information received from third parties and other assumptions that the Company believes are reasonable under the circumstances. Estimates are periodically reviewed in light of changes in circumstances, facts and experience. Actual results could differ from those estimates.
Cash, Cash Equivalents and Investments
We consider all highly liquid investments purchased with an original maturity of three months or less at the date of acquisition to be cash equivalents. Debt securities with original maturities greater than three months and remaining maturities less than one year are classified as short-term investments. We classify all investments as available-for-sale. Available-for-sale securities are carried at estimated fair value, with accumulated unrealized gains and losses reported as a component of accumulated other comprehensive income (loss) in the accompanying consolidated balance sheets.
Realized gains or losses on the sale of investments are determined on a specific identification method, and such gains and losses are reflected as a component of interest and other income, net in the accompanying consolidated statements of operations and comprehensive loss.
Marketable securities investments are evaluated periodically for impairment. We take into account general market conditions, changes in the economic environment as well as specific investment attributes, such as credit downgrade or illiquidity for each investment, the expected cash flows from the securities, our intent to sell the securities and whether or not we will be required to sell the securities before the recovery of their amortized cost, to estimate the fair value of our investments and to determine whether impairment is other than temporary. If it is determined that a decline in fair value of any investment is other than temporary, then the unrealized loss related to credit risk would be included in interest and other income, net.
Debt Issuance Costs
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
Debt issuance costs are amortized to interest expense over the estimated life of the related debt based on the effective interest method. In accordance with applicable accounting guidance, we present debt issuance costs on the consolidated balance sheets as a direct deduction from the associated debt. See Note 6 for more details.
Concentration of Credit Risk and of Significant Suppliers and Customers
Financial instruments that potentially subject us to concentrations of credit risk consist primarily of cash, cash equivalents and investments. From time to time, we maintain cash and cash equivalent balances in excess of amounts insured by the Federal Deposit Insurance Corporation (“FDIC”) and the Securities Investor Protection Corporation ("SIPC"). Primarily all of our cash, cash equivalents and investments at December 31, 2020 were in excess of amounts insured by the FDIC and SIPC. We do not believe that we are subject to unusual credit risk beyond the normal credit risk associated with commercial banking relationships.
We are dependent on third-party manufacturers to supply products for research and development activities in our programs. We rely on a manufacturer to supply us with our requirements for the active pharmaceutical ingredients, or API, and formulated drugs related to Zimeta. Our biologic manufacturing supplies, some of which may have longer lead times, are procured from another manufacturer. These programs would be adversely affected by a significant interruption in our supply chain.
We are also dependent on our licensing partners and contract manufacturing customer to pay us on time. We regularly review the collectability of our accounts, royalty and license receivable by considering factors such as historical experience, credit quality, the age of the receivable balances, and current economic conditions that may affect a customer’s ability to pay. For the years ended December 31, 2020, and 2019, we did not incur any bad debt expense. See Note 3 for more details.
Fair Value Measurements
We use the provisions of Accounting Standards Codification ("ASC") 820, “Fair Value Measurements and Disclosure", to determine the fair values of our financial and nonfinancial assets and liabilities where applicable. ASC 820 defines fair value, establishes a framework for measuring fair value in U.S. GAAP and expands disclosure about fair value measurements. The objective of fair value measurement is to determine the price that would be received to sell the asset or paid to transfer the liability (an exit price) in an orderly transaction between market participants at the measurement date. ASC 820 emphasizes that fair value is a market-based measurement, not an entity-specific measurement, and that market participant assumptions include assumptions about risk and effect of a restriction on the sale or use of an asset. To increase consistency and comparability in fair value measurement and related disclosures, ASC 820 establishes a fair value hierarchy that prioritizes the inputs to valuation techniques used to measure fair value into three broad levels: (1) Level 1 inputs are quoted prices (unadjusted) in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date; (2) Level 2 inputs are inputs other than quoted prices included in Level 1 that are observable for the asset or liability, either directly or indirectly through corroboration with observable market data; and (3) Level 3 inputs are unobservable inputs for the asset or liability that reflect the reporting entity’s own assumptions about risk and the assumptions market participants would use in pricing the asset or liability developed based on the best information available in the circumstances.
Government agency notes, corporate notes and commercial papers are recorded at their estimated fair value. Since these available-for-sale securities generally have market prices from multiple sources and it can be difficult to select the best individual price directly from the quoted prices in the active markets, we use Level 2 inputs for the valuation of these securities. Using the Level 2 inputs, a “consensus price” or a weighted average price for each of these securities can be derived from a distribution-curve-based algorithm which includes market prices obtained from a variety of industrial standard data providers (e.g. Bloomberg), security master files from large financial institutions, and other third-party sources.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
The carrying amount of financial instruments, including cash, accounts receivable, accounts payable and accrued liabilities approximate fair value due to the short maturities of these financial instruments. Financial assets, which consist of money market funds and available-for-sale securities, are measured at fair value on a recurring basis. (see Note 4). Based on the borrowing rates currently available for loans with similar terms, we believe the fair value of long-term debt approximates its carrying value.
Property and Equipment
Property and equipment are stated at cost less accumulated depreciation and amortization. We calculate depreciation using the straight-line method over the estimated useful lives of the assets, which range from two to five years for furniture, fixtures, lab and computer equipment and software, and fifteen to thirty-nine years for land improvements and real property. Land and assets held within construction in progress are not depreciated. Construction in progress is related to the construction or development of property and equipment that have not yet been placed in service for their intended use. Expenditures for repairs and maintenance of assets are charged to expense as incurred. We amortize leasehold improvements using the straight-line method over the estimated useful lives of the respective assets or the lease term, whichever is shorter. Upon retirement or sale, the cost and related accumulated depreciation and amortization of assets disposed of are removed from the accounts and any resulting gain or loss is included in other income/expense.
Lease Obligations
We have operating leases comprised of certain offices, lab space and equipment leases. We determine if an arrangement is a lease at inception. Our lease agreements may contain variable costs such as common area maintenance, insurance, real estate taxes or other costs. Such variable lease costs are expensed as incurred on the consolidated statements of income. For leases that have greater than 12-month lease term, Operating lease right-of-use ("ROU") assets and lease liabilities are recognized on the consolidated balance sheet at commencement date based on the present value of the future minimum lease payments over the lease term. ROU assets also include any initial direct costs incurred and any lease payments made at or before the lease commencement date, less lease incentives received.
As most of our leases do not provide an implicit rate, we use our incremental borrowing rate based on the information available at commencement date in determining the present value of lease payments. Our incremental borrowing rate is a hypothetical rate based on our understanding of what our credit rating would be in a similar economic environment.
Operating leases are included in "operating lease right-of-use assets", "current portion of operating lease liabilities", and "long-term operating lease liabilities" on our consolidated balance sheets. Lease expenses is recognized on a straight-line basis over the lease term. We also elected to apply the short-term lease measurement and recognition exemption in which ROU assets and lease liabilities are not recognized for leases with terms of 12 months or less.
Licenses
The costs incurred for the rights to use licensed technologies in the research and development process, including licensing fees and milestone payments, are charged to research and development expense as incurred in situations where we have not identified an alternative future use for the acquired rights, and are capitalized in situations where we have identified an alternative future use. No costs associated with the use of licensed technologies have been capitalized to date.
Impairment of Long-Lived Assets
We review long-lived assets, including property and equipment, for impairment whenever events or changes in business circumstances indicate that the carrying amount of the assets may not be fully recoverable.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
Factors that we consider in deciding when to perform an impairment review include significant underperformance of the business in relation to expectations, significant negative industry or economic trends, and significant changes or planned changes in the use of the assets. If an impairment review is performed to evaluate a long-lived asset for recoverability, we compare forecasts of undiscounted cash flows expected to result from the use and eventual disposition of the long-lived asset to its carrying value. An impairment loss would be recognized when estimated undiscounted future cash flows expected to result from the use of an asset are less than its carrying amount. The impairment loss would be based on the excess of the carrying value of the impaired asset over its fair value, determined based on discounted cash flows. To date, we have not recorded any impairment losses on long-lived assets.
Revenue
Our revenues consist of product revenues resulting from the sales of Mirataz and Zimeta, revenue from the sale of our Mirataz asset, the associated partner royalties, revenue from our contract manufacturing service and partner licensing revenue.
Product Revenue
Our product revenues consist of sales of Mirataz and Zimeta. We account for a contract with a customer when there is a legally enforceable contract between us and our customer, the rights of the parties are identified, the contract has commercial substance, and collectability of the contract consideration is probable. Our customers could either be distributors who subsequently resell our products to third parties such as veterinarians, clinics or animal hospitals, licensing partners or other third parties.
Revenue from the sale of our Mirataz asset
In March 2020, we entered into an Asset Purchase Agreement to sell Mirataz to Dechra for a cash purchase price of $43 million. On April 15, 2020, we completed the sale of Mirataz to Dechra and received payment of $38.7 million on the closing date. The remaining $4.3 million will be paid out of escrow beginning in 12 months assuming no escrow claims. We concluded our accounting treatment of the asset sale to Dechra meets the scope of ASC Topic 610-20-15-2, "Gains and Losses from the Derecognition of Nonfinancial Assets". We considered our strategic realignment of our business model whereby we become a biologics-only company focused on accelerating our deep pipeline of late-stage biologics candidates in canine and feline markets. Accordingly, we plan to rely more on a partnership-based model for commercialization strategy whereby pipeline assets are partnered with larger commercial partners that can maximize product opportunity in return for upfront payment, contingent milestones, and royalties on future sales. Based on the above evaluation in the aggregate, we concluded the proper presentation of the sale of Mirataz to be within operating income as part of revenue. This is presented as a separate line item and described as revenue from asset sale.
Partner Royalties
We recognize royalty revenue in connection with licenses granted under license and development arrangements with partners. Royalties are based upon a percentage of commercial sales of partnered products based on levels of net sales. These sales-based royalties, for which the license was deemed the predominant element to which the royalties relate, are estimated and recognized in the period in which the partners’ commercial sales occur. The royalties are generally reported and payable to us within 45 to 60 days of the end of each calendar quarter in which the commercial sales are made. We base our estimates of royalties earned on actual sales information from our partners when available. If actual royalties received are different than amounts estimated, we would adjust the royalty revenue in the period in which the adjustment becomes known. We do not recognize revenues if it is probable that a significant reversal of revenues will occur.
Contract Manufacturing Revenue
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
The manufacturing revenue stream generally represents revenue from the manufacturing of customer product(s). Under a manufacturing contract, a quantity of manufacturing runs is ordered and the product is manufactured according to the customer’s specifications and typically only one performance obligation is included. Each manufacturing run represents a distinct service that is sold separately and has stand-alone value to the customer. The product(s) are manufactured exclusively for a specific customer and have no alternative use. The customer retains control of their product during the entire manufacturing process and can make changes to the process or specifications at their request.
The customer and our project team typically have a timeline on each milestone and duration time. They also have an estimated start and finish date. When the project is moving forward, they constantly change to the actual date to track the project progress. The timing has been shared by both parties. This becomes the most important basis for our revenue recognition.
Because of the timing effect of revenue recognition, billings and cash collections can be recorded into three different ways: billed trade receivables, contract assets (unbilled receivables), and contract liabilities (customer deposits and deferred revenue). Contract assets are recorded when our right to consideration is conditioned on something other than the passage of time. Contract assets are reclassified to trade receivables on the balance sheet when our rights become unconditional. Contract liabilities represent customer deposits and deferred revenue billed and/or received in advance of our fulfillment of performance obligations. Contract liabilities convert to revenue as we perform our obligations under the contract.
Partner Licensing Revenue
Partner licensing revenue consists of revenue that compensates us for services performed, such as formulation, process development, and preparation of pre-clinical and clinical drug product materials under research and development arrangements with partners. Revenues related to research and development are generally recognized as the related services or activities are performed using the output method and in accordance with the contract terms. To the extent that the agreements specify services are to be performed on a fixed basis, revenues are recognized consistent with the pattern of the work performed. In agreements that specify milestones, we evaluate whether the milestones are considered probable of being achieved and estimate the amount to be included in the transaction price using the most likely amount method. If it is probable that a significant revenue reversal would not occur, the value of the associated milestone is recognized at a point in time. Non-refundable milestone payments related to arrangements under which we have continuing performance obligations would be deferred and recognized over the period of performance. Milestone payments that are not within our control, such as submission for approval to regulators by a partner or approvals from regulators, are not considered probable of being achieved until those submissions are submitted by the customer or approvals are received.
Revenue Recognition
We recognize revenues in accordance with ASC Topic 606 (“ASC 606”), "Revenue from Contracts with Customers". We account for a contract with a customer when there is a legally enforceable contract between us and our customer, the rights of the parties are identified, the contract has commercial substance, and collectability of the contract consideration is probable. Our customers could either be distributors who subsequently resell our products to third parties such as veterinarians, clinics or animal hospitals, licensing partners or the third parties.
In accordance with ASC 606, we apply the following steps to recognize revenue that reflect the consideration to which we expect to be entitled to receive in exchange for the promised goods or services:
1. Identify the contract with a customer
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
A contract with a customer exists when we enter into an enforceable contract with a customer. These contracts define each party's rights, payment terms and other contractual terms and conditions of the sale. We apply judgment in determining the customer’s ability and intention to pay, which is based on published credit and financial information pertaining to the customer.
2. Identify the performance obligations in the contract
Our product in a given purchase order is delivered at the same time and we do not separate an individual order into separate performance obligations. We have concluded the sale of finished goods and related shipping and handling are accounted for as a single performance obligation as there are no other promises to deliver goods beyond what is specified in each accepted customer order.
Under a manufacturing contract, a quantity of manufacturing runs is ordered and the product is manufactured according to the customer’s specifications and typically only one performance obligation is included. Each manufacturing run represents a distinct service that is sold separately and has stand-alone value to the customer.
3. Determine the transaction price
The transaction price is determined based on the consideration to which we will be entitled to receive in exchange for transferring goods or services to the customer, typically a fixed consideration in our contractual agreements.
4. Allocate the transaction price to the performance obligations
The transaction price is allocated to the performance obligations identified in each contract. The nature of the promises/obligations under our contracts is to transfer a distinct good or service. Accordingly, because a single performance obligation exists, including in each milestone pertaining to contract manufacturing, no allocation of the transaction price is necessary.
5. Determine the satisfaction of performance obligation
Revenue for product sales is recognized when control of the finished goods is transferred to the customer, net of applicable reserves for variable consideration. Control of the finished goods is transferred at a point in time, upon delivery to the customer.
For contract manufacturing service, revenue is recognized over time. Control of the finished manufactured products is transferred at a point in time, upon delivery to the customer.
Royalty revenues are estimated and recognized in the period in which the partners’ commercial sales occur. The royalties are generally reported and payable to us within 45 to 60 days of the end of each calendar quarter in which the commercial sales are made.
For partner licensing, revenues related to research and development are generally recognized as the related services or activities are performed using the output method and in accordance with the contract terms. To the extent that the agreements specify services are to be performed on a fixed basis, revenues are recognized consistent with the pattern of the work performed. In agreements that specify milestones, we evaluate whether the milestones are considered probable of being achieved and estimate the amount to be included in the transaction price using the most likely amount method.
Reserves for Variable Consideration
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
Revenues from product sales are recorded at the net sales price, which includes estimates of variable consideration for which reserves are established. Components of variable consideration include product returns, allowances and discounts. These estimates take into consideration a range of possible outcomes for the expected value (probability-weighted estimate) or relevant factors such as current contractual and statutory requirements, specific known market events and trends, industry data, and forecasted customer buying and payment patterns. Overall, these reserves reflect our best estimates of the amount of consideration to which we are entitled based on the terms of the respective underlying contracts. The amount of variable consideration included in the transaction price may be constrained and is included in the net sales price only to the extent that it is probable that a significant reversal in the amount of the cumulative revenue recognized where the contract will not occur in a future period. Actual amounts of consideration ultimately received may differ from our estimates. If actual results in the future vary from our estimates, we will adjust these estimates, which would affect net product revenues and earnings in the period such variances become known.
No reserves for contract manufacturing service are recorded as each manufacturing run represents a distinct service that is sold separately and has stand-alone value to the customer. The product(s) are manufactured exclusively for a specific customer and have no alternative use.
Sales-based royalty revenues recorded by us are based on the licensee’s actual net sales that occurred during the relevant period. No reserves were established and to-date, there were no adjustments made in subsequent periods.
Revenues from partner licensing is recognized when non-refundable, up-front fees are allocated to a license that is determined to be distinct from the other performance obligations identified in the license agreement. No reserves were established.
Product Returns
Consistent with the industry practice, we generally offer customers a limited right of return of damaged or expired product that has been purchased directly from us. Our return policy generally allows customers to receive credit for expired products within 90 days after the product’s expiration date. We estimate the amount of our product revenues that may be returned by our customers and record these estimates as a reduction of product revenues in the period the related product revenues are recognized, as well as within accrued liabilities, in the consolidated balance sheets. We currently estimate product return liabilities using probability-weighted available industry data and data provided by the our distributors such as the inventories remaining in the distribution channel. To-date, we have no returns and believe that returns of our product in future periods will be minimal. We do not record a return asset associated with the returned damaged or expired goods due to such asset is deemed to be fully impaired at the time of product return.
Our contract manufacturing customer retains control of their product during the entire manufacturing process and can make changes to the process or specifications at their request. There are no product returns.
Sales Discounts and Allowances
We compensate our distributors for sales order management, data and distribution and other services through sales discounts and allowances. However, such services are not distinct from our sale of products to distributors and, therefore, these discounts and allowances are recorded as a reduction of product revenues in the statements of operations, as well as a reduction to accounts receivable in the consolidated balance sheets.
No discounts and allowances are recorded for contract manufacturing service as the price of each milestone is agreed upon when the contract is signed.
Sales Commissions
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
We generally expense sales commissions when incurred because the amortization period would have been one year or less. These costs are recorded within sales and marketing expenses.
Cost of Revenues
Cost of product revenues consists primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, inbound shipping and other third-party logistics costs.
Contract manufacturing costs consist primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, rent, facility costs and related machinery depreciation.
Inventories
We value inventory at the lower of cost or net realizable value. We determine the cost of inventory using the standard-cost method, which approximates actual cost based on a first-in, first-out method. We analyze our inventory levels quarterly and write down inventory subject to expire in excess of expected requirements, or that has a cost basis in excess of its expected net realizable value. These inventory related costs are recognized as cost of product revenues on the accompanying Consolidated Statements of Operations and Comprehensive Loss. In the quarter ended March 31, 2020, we wrote off $3,494,000 Mirataz inventory related to the Dechra Asset Purchase Agreement, due to the transition to Dechra brand labelling. We wrote off $332,000 Zimeta inventory related to the Dechra Distribution and Licensing Agreement executed in December 2020. Currently our inventory consists of finished goods only.
Research and Development Costs
All costs of research and development are expensed in the period incurred. Research and development costs primarily consist of salaries and related expenses for personnel, stock-based compensation expense, fees paid to consultants, outside service providers, professional services, travel costs and materials used in clinical trials and research and development.
Patent Costs
All patent-related costs incurred in connection with filing patent applications are recorded in research and development expenses when incurred, as recoverability of such expenditures is uncertain.
Stock-Based Compensation
Our stock-based compensation plan (see Note 10) provides for the grant of stock options, restricted common stock, restricted stock units and stock appreciation rights. The estimated fair values of employee stock option grants are determined as of the date of grant using the Black-Scholes option pricing model. This method incorporates the fair value of our common stock at the date of each grant and various assumptions such as the risk-free interest rate, expected volatility based on historic volatility of our own stock prices, and expected dividend yield, and expected term of the options. The estimated fair values of restricted stock awards are determined based on the fair value of our common stock on the date of grant. The estimated fair values of stock-based awards, including the effect of estimated forfeitures, are expensed over the requisite service period, which is generally the awards’ vesting period. We classify stock-based compensation expense in the consolidated statements of operations and comprehensive loss in the same manner in which the award recipient’s payroll costs are classified.
Our accounting policy for equity instruments issued to consultants and vendors in exchange for goods and services follows Financial Accounting Standards Board ("FASB") guidance. All transactions in which goods or services are the consideration received for the issuance of equity instruments are accounted for based on the fair value of the consideration received or the fair value of the equity instrument issued, whichever is more reliably measurable. The measurement date of the fair value of the equity instrument issued is the earlier
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
of the date on which the counterparty’s performance is complete or the date at which a commitment for performance is reached.
Income Taxes
We account for income taxes using the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been recognized in the consolidated financial statements or in our tax returns. Deferred taxes are determined based on the difference between the financial statement and tax basis of assets and liabilities using enacted tax rates in effect in the years in which the differences are expected to reverse. Changes in deferred tax assets and liabilities are recorded in the provision for income taxes. We assess the likelihood that our deferred tax assets will be recovered from future taxable income and, to the extent we believe, based upon the weight of available evidence, that it is more likely than not that all or a portion of deferred tax assets will not be realized, a valuation allowance is established through a charge to income tax expense. Potential for recovery of deferred tax assets is evaluated by estimating the future taxable profits expected and considering prudent and feasible tax planning strategies.
We account for uncertainty in income taxes recognized in the consolidated financial statements by applying a two-step process to determine the amount of tax benefit to be recognized. First, the tax position must be evaluated to determine the likelihood that it will be sustained upon external examination by the taxing authorities. If the tax position is deemed more-likely-than-not to be sustained, the tax position is then assessed to determine the amount of benefit to recognize in the consolidated financial statements. The amount of the benefit that may be recognized is the largest amount that has a greater than 50% likelihood of being realized upon ultimate settlement. The provision for income taxes includes the effects of any resulting tax reserves, or unrecognized tax benefits, that are considered appropriate as well as the related net interest and penalties.
Comprehensive Loss
Our comprehensive loss includes the change in unrealized gains or losses on available-for-sale securities. The cumulative amount of gains or losses is reflected as a separate component of stockholders' equity in the accompanying consolidated balance sheets as accumulated other comprehensive loss.
Segment Data
We manage our operations as a single segment for the purposes of assessing performance and making operating decisions. We are a veterinary biotechnology company focusing on developing therapies for pets. Our chief operating decision maker is our Chief Executive Officer, who makes resource allocation decisions and assesses performance based on financial information presented on a consolidated basis. There are no segment managers who are held accountable by the chief operating decision-maker, or anyone else, for operations, operating results, and planning for levels or components below the consolidated unit level. Accordingly, we have determined that we have a single reportable segment and operating segment structure. All assets are held in the United States.
Basic and Diluted Net Loss Per Common Share
Basic net loss per common share is computed by dividing net loss attributable to common stockholders for the period by the weighted average number of common shares outstanding during the period. Diluted net loss per share is computed by dividing the net loss attributable to common stockholders for the period by the weighted average number of common shares, including potential dilutive shares of common stock assuming the dilutive effect of potentially dilutive securities. For periods in which we have reported a net loss, diluted net loss per common share is the same as basic net loss per common share, since the impact of the potentially dilutive securities would be anti-dilutive to the calculation of net loss per common share (see Note 14).
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
Recently Issued Accounting Pronouncements
In March 2020, the FASB issued ASU No. 2020-04, "Reference Rate Reform (Topic 848)", changes to the interbank offered rates (IBORs), and, particularly, the risk of cessation of the London Interbank Offered Rate ("LIBOR"). The amendments provide optional expedients and exceptions for applying U.S. GAAP to contracts that reference LIBOR expected to be discontinued because of reference rate reform. The expedients and exceptions do not apply to contract modifications made after December 31, 2022. The following optional expedients are permitted for contracts that are modified because of reference rate reform and that meet certain scope guidance: Modifications of contracts within the scope of ASC Topic 470, Debt, should be accounted for by prospectively adjusting the effective interest rate. The amendment also permits an entity to consider contract modifications due to reference rate reform to be an event that does not require contract remeasurement at the modification date or reassessment of a previous accounting determination. When elected, the optional expedients for contract modifications must be applied consistently for all contracts. It applies to all entities within the scope of the affected accounting guidance and will take effect as of March 12, 2020 through December 31, 2022. We have one loan contract which references LIBOR rate. We have not modified the contract with our lenders yet. We are currently evaluating the new guidance and have not determined the impact this standard may have on our financial statements.
We do not believe there are any other recently issued standards not yet effective that will have a material impact on our consolidated financial statements when the standards become effective.
3. Revenues and Cost of Product Revenues
Our revenues consist of product revenue resulting from the sale of Mirataz for the management of weight loss in cats and Zimeta for the treatment of fever in horses. In addition, our 2020 revenues also consist of Mirataz asset sale, partner royalties, contract manufacturing and partner licensing revenue. We account for a contract with a customer when there is a legally enforceable contract between us and our customer, the rights of the parties are identified, the contract has commercial substance, and collectability of the contract consideration is probable. Our product revenues are measured based on the consideration specified in the contract with each customer, net of product returns, discounts and allowances.
The following table presents revenues and costs for the years ended December 31, 2020, 2019 and 2018 (in thousands):
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | | | | | | | | | | | | | | | | |
| | Years Ended December 31, |
| | 2020 | | 2019 | | 2018 |
| | | | | | |
| Revenues | | | | | | |
| Net product revenues | | $ | 878 | | | $ | 4,256 | | | $ | 1,966 | |
| Revenue from asset sale | | 38,700 | | | — | | | — | |
| Partner royalty revenue | | 535 | | | — | | | — | |
| Contract manufacturing revenue | | 1,551 | | | — | | | — | |
| Partner licensing revenue | | 500 | | | — | | | — | |
| Total revenues | | 42,164 | | | 4,256 | | | 1,966 | |
| | | | | | |
| Costs of revenues | | | | | | |
Cost of product revenues (1) | | 3,945 | | | 587 | | | 324 | |
| Contract manufacturing costs | | 681 | | | — | | | — | |
| Total cost of revenues | | 4,626 | | | 587 | | | 324 | |
| | | | | | |
| Gross margin | | $ | 37,538 | | | $ | 3,669 | | | $ | 1,642 | |
| | | | | | |
(1) Includes $3,826 finished goods write-off related to the Dechra agreements signed in 2020. |
Concentrations of credit risk
Our revenue was generated entirely from sales within the United States. Our product sales to three large distributors, namely MWI Animal Health, Henry Schein (now Covetrus North America) and Midwest Veterinary Supply each accounted for more than 10% of gross product revenues for the year ended December 31, 2020. On a combined basis, in 2020, these distributors accounted for approximately 78% of our product sales.
Our accounts receivable from amounts billed for contract manufacturing services in 2020 is derived from one customer. The contract requires up-front payment and installment payments during the service period. We perform periodic evaluations of the financial condition of our customers and generally do not require collateral, but we can terminate any contract if a material default occurs. There were no contract manufacturing services in 2019 or 2018.
Our product sales to three large distributors, namely Henry Schein (now Covetrus North America), MWI Animal Health and Patterson Veterinary Supply each accounted for more than 10% of total revenues for the year ended December 31, 2019. On a combined basis, in 2019, these distributors accounted for approximately 85% of our product sales.
Our product sales to four large distributors, namely MWI Animal Health, Henry Schein (now Covetrus North America), Patterson Veterinary Supply and Midwest Veterinary Supply each accounted for more than 10% of total revenues for the year ended December 31, 2018. On a combined basis, in 2018, these distributors accounted for approximately 91% of our product sales.
Product returns
Our return policy generally allows customers to receive credit for expired products within 90 days after the product’s expiration date. We currently estimate product return liabilities of 3% for Zimeta of gross revenue using probability-weighted available industry data and data provided by our distributors such as
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
the inventories remaining in the distribution channel. Adjustments will be made in the future if actual results vary from our estimates.
Accounts receivable and allowance for doubtful accounts
Accounts receivable are stated at their carrying values, net of any allowances for doubtful accounts. Accounts receivable consist primarily of amounts due from distributors, our contract manufacturing customer, and licensing partners, for which collection is probable based on the customer's intent and ability to pay. Receivables are evaluated for collection probability on a regular basis and an allowance for doubtful accounts is recorded, if necessary. We have no allowance for doubtful accounts as of December 31, 2020 and 2019, as our analysis did not uncover any collection risks.
Cost of Revenues
Cost of product revenues consists primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, inbound shipping and other third-party logistics costs. For the year 2020, cost of product revenues included $3.8 million finished goods write-off primarily due to the transition to proprietary Dechra brand labelling on the Mirataz asset sale.
Cost of product revenues for the year 2019 consists primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, inbound shipping and other third-party logistics costs, and increased by 81.2% to $587,000 compared with $324,000 for the same period in 2018. Mirataz was launched in July 2018 while 2019 reflected a full year sale of the product.
Contract manufacturing costs of $681,000 in the year 2020 consist primarily of the cost of direct materials, direct labor and overhead costs associated with manufacturing, rent, facility costs and related machinery depreciation. There were no contract manufacturing services in 2019 or 2018.
4. Fair Value Measurements
We measure certain financial assets at fair value on a recurring basis, including cash equivalents and available-for-sale securities. The fair value of these financial assets was determined based on a three-tier fair value hierarchy as described in Note 2, which prioritizes the inputs used in measuring fair value.
The following table presents information about our financial assets that are measured at fair value on a recurring basis as of December 31, 2020 and indicates the fair value hierarchy of the valuation techniques utilized to determine such fair value:
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands) | | Fair Value Measurements as of December 31, 2020 |
| Description | | Total | | Quoted Prices in Active Markets (Level 1) | | Significant Other Observable Inputs (Level 2) | | Unobservable Inputs (Level 3) |
| Cash equivalents: | | | | | | | | |
| Money market funds | | $ | 219 | | | $ | 219 | | | $ | — | | | $ | — | |
| U.S. treasury bills | | 4,060 | | | 4,060 | | | | | |
| Commercial paper | | 3,899 | | | — | | | 3,899 | | | — | |
| U.S. government agency notes | | 2,000 | | | — | | | 2,000 | | | — | |
| Corporate notes | | 315 | | | — | | | 315 | | | — | |
| Short-term investments: | | | | | | | | |
| U.S. treasury bills | | 6,531 | | | 6,531 | | | — | | | — | |
| U.S. government agency notes | | 36,444 | | | — | | | 36,444 | | | — | |
| Corporate notes | | 3,783 | | | — | | | 3,783 | | | — | |
| Long-term investments: | | | | | | | | |
| U.S. government agency notes | | 1,500 | | | — | | | 1,500 | | | — | |
| Total | | $ | 58,751 | | | $ | 10,810 | | | $ | 47,941 | | | $ | — | |
The following table presents information about our financial assets that are measured at fair value on a recurring basis as of December 31, 2019 and indicates the fair value hierarchy of the valuation techniques utilized to determine such fair value:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands) | | Fair Value Measurements as of December 31, 2019 |
| Description | | Total | | Quoted Prices in Active Markets (Level 1) | | Significant Other Observable Inputs (Level 2) | | Unobservable Inputs (Level 3) |
| Cash equivalents: | | | | | | | | |
| Money market funds | | $ | 1,592 | | | $ | 1,592 | | | $ | — | | | $ | — | |
| Commercial paper | | 13,580 | | | — | | | 13,580 | | | — | |
| Short-term investments: | | | | | | | | — | |
| U.S. treasury bills | | 8,524 | | | 8,524 | | | — | | | — | |
| Commercial paper | | 25,573 | | | — | | | 25,573 | | | — | |
| U.S. government agency notes | | 11,461 | | | — | | | 11,461 | | | — | |
| Corporate notes | | 10,165 | | | — | | | 10,165 | | | — | |
| Long-term investments: | | | | | | | | |
| U.S. government agency notes | | 801 | | | — | | | 801 | | | — | |
| Corporate notes | | 1,036 | | | — | | | 1,036 | | | — | |
| Total | | $ | 72,732 | | | $ | 10,116 | | | $ | 62,616 | | | $ | — | |
There were no other transfers of assets between Level 1, Level 2 or Level 3 of the fair value hierarchy during the years ended December 31, 2020 and 2019.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
At December 31, 2020 and 2019, we did not have any financial liabilities which were measured at fair value on a recurring basis.
5. Investments
The following tables summarize our investments in available-for-sale securities by significant investment category reported as short-term or long-term investments as of December 31, 2020 and 2019 (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | December 31, 2020 |
| | Amortized Cost | | Gross Unrealized Gains | | Gross Unrealized Losses | | Fair Value |
| Short-term investments: | | | | | | | | |
| U.S. treasury bills | | $ | 6,531 | | | $ | — | | | $ | — | | | $ | 6,531 | |
| U.S. government agency notes | | 36,437 | | | 8 | | | (1) | | | 36,444 | |
| Corporate notes | | 3,778 | | | 5 | | | — | | | 3,783 | |
| | 46,746 | | | 13 | | | (1) | | | 46,758 | |
| Long-term investments: | | | | | | | | |
| U.S. government agency notes | | 1,500 | | | — | | | — | | | 1,500 | |
| | 1,500 | | | — | | | — | | | 1,500 | |
| Total available-for-sale investments | | $ | 48,246 | | | $ | 13 | | | $ | (1) | | | $ | 48,258 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | December 31, 2019 |
| | Amortized Cost | | Gross Unrealized Gains | | Gross Unrealized Losses | | Fair Value |
| Short-term investments: | | | | | | | | |
| U.S. treasury bills | | $ | 8,517 | | | $ | 7 | | | $ | — | | | $ | 8,524 | |
| Commercial paper | | 25,576 | | | 3 | | | (6) | | | 25,573 | |
| U.S. government agency notes | | 11,460 | | | 2 | | | (1) | | | 11,461 | |
| Corporate notes | | 10,157 | | | 8 | | | — | | | 10,165 | |
| | 55,710 | | | 20 | | | (7) | | | 55,723 | |
| Long-term investments: | | | | | | | | |
| U.S. government agency notes | | 801 | | | — | | | — | | | 801 | |
| Corporate notes | | 1,036 | | | — | | | — | | | 1,036 | |
| | 1,837 | | | — | | | — | | | 1,837 | |
| Total available-for-sale investments | | $ | 57,547 | | | $ | 20 | | | $ | (7) | | | $ | 57,560 | |
The following table summarizes the contractual maturities of our available-for-sale securities at December 31, 2020 and 2019 (in thousands):
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | | | | | | | | | | | | | | | | | | | |
| December 31, |
| 2020 | | 2019 |
| Amortized Cost | | Fair Value | | Amortized Cost | | Fair Value |
| Mature in less than one year | $ | 46,746 | | | $ | 46,758 | | | $ | 55,710 | | | $ | 55,723 | |
| Mature in one year or more | 1,500 | | | 1,500 | | | 1,837 | | | $ | 1,837 | |
| Total | $ | 48,246 | | | $ | 48,258 | | | $ | 57,547 | | | $ | 57,560 | |
6. Borrowings
On September 30, 2019, we entered into a Loan and Security Agreement (the “Loan Agreement”) with Solar Capital Ltd., as collateral agent and lender, and the other lenders named in the Loan Agreement (Solar Capital Ltd. and the other lenders collectively, the “Lenders”). The Lenders have agreed to make available to KindredBio an aggregate principal amount of up to $50.0 million under the Loan Agreement. We plan to use the loan proceeds to support the development and commercialization of our products and product candidates as well as for working capital and general corporate purposes. The Loan Agreement provides for a term loan commitment of $50.0 million in three tranches: (1) a $20.0 million term A loan that was funded on September 30, 2019; (2) a $15.0 million term B loan that is to be funded at our request, subject to certain conditions described in the Loan Agreement being satisfied, no later than December 31, 2020; and (3) a $15.0 million term C loan that is to be funded at our request, subject to certain conditions described in the Loan Agreement being satisfied, on or before June 30, 2021. Each term loan has a maturity date of September 30, 2024. We elected not to draw down on the $15.0 million term B loan that is to be funded at our request as of December 31, 2020. Each term loan bears interest at a floating per annum rate equal to the one-month LIBOR rate (with a floor of 2.17%) plus 6.75%. We are permitted to make interest-only payments on each term loan through October 31, 2021. The interest-only period can be extended by six months upon our satisfaction of the minimum liquidity requirements described in the Loan Agreement. Equal monthly payments of principal will be due and payable commencing at the end of the interest-only period of the term loans. In connection with the term loan, we incurred closing costs of $819,000, which are shown net of the proceeds and will be amortized over the term of the loan using the effective interest method. We are obligated to pay a facility fee in the amount of 0.50% of each term loan that is funded and a non-utilization fee in the amount of 0.25% of each term B loan and term C loan to the extent that such loans are not funded. We are obligated to pay a final fee equal to 3.60% of the aggregate amount of the term loans funded (or 4.35% of such funded loans if the interest-only period is extended as described above), such final fee to be due and payable upon the earliest to occur of (1) the maturity date, (2) the acceleration of the term loans, and (3) the prepayment of the term loans. This final fee is being accrued over the term of the loan agreement. We have the option to prepay all, but not less than all, of the outstanding principal balance of the term loans under the Loan Agreement. If we prepay the term loans prior to the maturity date, we must pay the Lenders a prepayment premium fee based on a percentage of the outstanding principal balance, equal to 2.0% if the prepayment occurs after September 30, 2020 but on or before September 30, 2021, or 1.0% if the prepayment occurs after September 30, 2021. Our obligations under the Loan Agreement are secured by a first-priority security interest in substantially all of KindredBio’s assets, including our intellectual property, and a lien on our real property. The Loan Agreement contains customary representations, warranties and covenants and also includes customary events of default, including payment defaults, breaches of covenants, and a default upon the occurrence of a material adverse change affecting us. Upon the occurrence of an event of default, a default interest rate of an additional 5.00% per annum may be applied to the outstanding loan balance, and the Lenders may declare all outstanding obligations immediately due and payable and exercise all their rights and remedies as set forth in the Loan Agreement and under applicable law. We were in compliance with all covenants as of December 31, 2020.
In conjunction with the Dechra Asset Purchase Agreement, on March 16, 2020, we entered into a First Amendment to Loan and Security Agreement (the "First Amendment") with the Lenders in connection with the sale of our Mirataz asset. Among other things, the First Amendment increases the minimum cash amount, as defined in the Loan Agreement, required to be maintained by KindredBio to $10,000,000 at any time prior
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
to the initial funding date of any term B loan, to $15,000,000 at all times on and after the initial funding date of any term B loan, and to $20,000,000 at all times on and after the initial funding date of any term C loan, and releases Solar Capital's lien in and to the assets that are being sold by KindredBio. We paid an amendment fee of $100,000. The First Amendment also requires KindredBio to receive unrestricted net proceeds of at least $10,000,000 prior to December 31, 2021 pursuant to a specified sale of preferred or common stock or convertible subordinated debt financing.
On December 23, 2020, we entered into a Second Amendment to Loan and Security Agreement (the "Second Amendment") with the Lenders in connection with the Zimeta Distribution and License Agreement with Dechra. We paid an amendment fee of $15,000. All other terms and conditions remain the same as the First Amendment.
As of December 31, 2020, assuming the principal payments start on November 1, 2021, our future debt payment obligations towards the principal and final fee, excluding interest payments and exit fee, for the respective fiscal years are as follows (in thousands):
| | | | | | | | |
| 2021 | | $ | 1,111 | |
| 2022 | | 6,667 | |
| 2023 | | 6,667 | |
| 2024 | | 6,275 | |
| Total principal and final fee payments | | 20,720 | |
| Less: unamortized debt issuance costs | | (567) | |
| Less: unaccreted value of final fee | | (540) | |
| Loan payable | | $ | 19,613 | |
| | |
| Loan payable, short-term | | $ | 1,111 | |
| Loan payable, long-term | | $ | 18,502 | |
7. Property and Equipment, Net
Property and equipment consisted of the following: | | | | | | | | | | | |
| As of December 31, |
| (In thousands) | 2020 | | 2019 |
| Computer and lab equipment | $ | 15,051 | | | $ | 10,188 | |
| Furniture and fixtures | 161 | | | 143 | |
| Leasehold improvements | 958 | | | 958 | |
| Building | 15,980 | | | 9,520 | |
| Building improvements | 1,864 | | | 1,238 | |
| Land | 85 | | | 85 | |
| Land improvement | 166 | | | 166 | |
| Construction-in-process | 975 | | | 10,932 | |
| Total | 35,240 | | | 33,230 |
| Less accumulated depreciation and amortization | (7,036) | | | (3,453) |
| Property and equipment, net | $ | 28,204 | | | $ | 29,777 | |
We constructed a Good Manufacturing Practice, or GMP, biologics manufacturing plant in Burlingame, CA which is fully commissioned and proceeded to cGMP manufacturing in January 2018. In August 2017 we acquired a manufacturing facility in Elwood, Kansas (the "Plant") and completed construction and commissioning in 2019. The Kansas plant includes approximately 180,000 square feet with clean rooms,
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
utility, equipment and related quality documentation suitable for small molecule and biologics manufacturing. The Kansas plant was purchased for $3,750,000, which includes approximately eight acres of land, all improvements located at the Plant, and all personal property and intangible property located at the Plant or used in connection with the operation of the Plant. Construction-in-process is comprised of equipment that have not been put into service for their intended use as of December 31, 2020.
Depreciation and amortization expense was $3,870,000, $1,880,000 and $805,000 for the years ended December 31, 2020, 2019 and 2018, respectively.
8. Accrued Liabilities
Accrued liabilities consisted of the following as of December 31, 2020 and 2019:
| | | | | | | | | | | |
| (In thousands) | As of December 31, |
| 2020 | | 2019 |
| Accrued consulting | $ | 468 | | | $ | 589 | |
| Accrued research and development costs | 1,654 | | | 1,336 | |
| Accrued other | 623 | | | 2,206 | |
| Total | $ | 2,745 | | | $ | 4,131 | |
9. Stockholders' Equity
Preferred Stock
Our Certificate of Incorporation, as amended, authorizes us to issue 10,000,000 shares of $0.0001 par value preferred stock. At December 31, 2020, 100,000 unissued shares of our preferred stock are designated as Series A Preferred Stock, and the remaining 9,900,000 unissued shares of our preferred stock are undesignated.
Common Stock
Our Certificate of Incorporation, as amended and restated, authorizes us to issue 100,000,000 shares of $0.0001 par value common stock.
Each share of common stock entitles the holder to one vote on all matters submitted to a vote of our stockholders, provided, however, that, except as otherwise required by law, holders of common stock shall not be entitled to vote on any amendment to the Certificate of Incorporation that relates solely to the terms of one or more outstanding shares of preferred stock if the holders of such affected series are entitled, either separately or together with the holders of one or more other series, to vote thereon pursuant to the Certificate of Incorporation or pursuant to the Delaware General Corporation Law.
In 2018, we issued 231,407 shares of common stock upon exercise of stock options for total proceeds of $635,000. In addition, we issued 46,850 shares of common stock to employees in connection with our employee stock purchase program for total proceeds of $352,000.
In 2019, we issued 305,801 shares of common stock upon exercise of stock options for total proceeds of $1,591,000. In addition, we issued 65,078 shares of common stock to employees in connection with our employee stock purchase program for total proceeds of $438,000.
In 2020, we issued 68,984 shares of common stock upon exercise of stock options for total proceeds of $256,000. In addition, we issued 51,417 shares of common stock to employees in connection with our employee stock purchase program for total proceeds of $180,000.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
As of December 31, 2020, we had 39,492,134 shares of common stock outstanding.
Stock Offerings
In January 2018, we filed a shelf registration statement on Form S-3 to offer and sell, from time to time, equity and debt securities in one or more offerings up to a total of $150.0 million. In May 2018, we entered into an At Market Issuance Sales Agreement, or the Sales Agreement, with B. Riley FBR, Inc., and Oppenheimer & Co. Inc. acting as our distribution agents, relating to the sale of up to $50,000,000 of our common stock from time to time. We terminated the Sales Agreement in June 2018 after having sold 188,100 shares, representing gross proceeds of approximately $1,903,000. Net proceeds, after deducting commission, fees and offering costs, were approximately $1,758,000. On June 20, 2018, we entered into an underwriting agreement with Cantor Fitzgerald & Co., as representative of the underwriters, and on June 22, 2018 we completed a public offering of 5,326,314 shares of common stock, which included the underwriters' option to purchase additional shares, at a public offering price of $9.50 per share for total gross proceeds of approximately $50,600,000. Net proceeds, after deducting underwriting discounts and commissions and offering expenses were approximately$47,422,000.
On January 18, 2019, we entered into an underwriting agreement with Barclays Capital Inc. and Stifel, Nicolaus & Company, Incorporated, as representatives of the underwriters, and on January 23, 2019, we completed a public offering of 4,847,250 shares of our common stock, which included the exercise in full by the underwriters of their option to purchase 632,250 additional shares of the Company’s common stock, at a public offering price of $9.50 per share for total gross proceeds of approximately $46,049,000. Net proceeds, after deducting underwriting discounts and commissions and offering expenses were approximately $43,125,000.
On April 8, 2020, we entered into an At Market Offering ("ATM") whereby we may offer and sell shares of our common stock from time to time up to $25 million. As of December 31, 2020, 59,211 shares were sold through the ATM, representing gross proceeds of approximately $298,000. Net proceeds, after deducting commission, fees and offering costs, were approximately $201,000.
The January 2018 shelf registration expired in January 2021. On January 15, 2021, we filed a new shelf registration statement on Form S-3 to offer and sell, from time to time, equity and debt securities in one or more offerings up to a total of $150.0 million. The registration statement was effective on February 2, 2021.
10. Stock-Based Awards and Benefit Plan
On November 4, 2012, our board of directors adopted the Kindred Biosciences, Inc. 2012 Equity Incentive Plan (the “2012 Plan"). The 2012 Plan provided for our board of directors to grant incentive stock options or non-qualified stock options for the purchase of common stock, to issue or sell shares of restricted common stock and to grant stock appreciation rights (“SARs”) to our employees, directors, consultants and advisers of the Company. Pursuant to the terms of the 2012 Plan, no options or SARs shall be granted under the 2012 Plan after 10 years from the date of adoption of the 2012 Plan. We reserved 4,000,000 shares of our common stock for issuance under the 2012 Plan. The 2012 Plan terminated in May 2016 and 2,586,792 stock option shares which had been granted prior to the plan’s expiration remaining outstanding as of December 31, 2020.
In May 2016, we adopted the 2016 Equity Incentive Plan (the “2016 Plan”), and reserved 3,000,000 shares of our common stock for issuance under the 2016 Plan. The 2016 Plan was the successor to our 2012 Plan and all awards made under the 2012 Plan remained subject to the terms of that plan. Options granted under the 2016 Plan were either incentive stock options or nonstatutory stock options. The 2016 Plan also provided for the grant of stock appreciation rights, restricted stock awards, restricted stock unit awards, performance stock awards, performance cash awards and other stock awards. The exercise price of a stock option was not less than 100% of the closing price of our common stock on the date of the grant. If, at any time
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
we granted an option, and the optionee directly or by attribution owned stock possessing more than 10% of the total combined voting power of all classes of our stock, the option price was at least 110% of the fair value and was not exercisable more than five years after the date of grant. Options generally vested over a period of one or four years from the date of grant. Options granted under the 2016 Plan expired no later than 10 years from the date of grant. As of December 31, 2020, there were 1,726,019 option shares outstanding, 37,500 restricted stock awards issued but unvested, and 102,500 restricted stock units granted but unvested, and no shares are available for future grants under the 2016 Plan since it was retired in June 2018.
In June 2018, we adopted the 2018 Equity Incentive Plan (the “2018 Plan”), and reserved 3,000,000 shares of our common stock for issuance under the 2018 Plan. At the Annual Meeting of Stockholders of the Company held on June 15, 2020 (the “2020 Annual Meeting”), our stockholders approved an amendment to the 2018 Equity Incentive Plan (as amended, the “2018 Plan”) to increase the number of shares of common stock authorized for issuance by 1,600,000 shares. The 2018 Plan is the successor to our 2016 Plan. All awards made under the 2016 and 2012 Plans shall remain subject to the terms of these plans. Options granted under the 2018 Plan may be either incentive stock options or nonstatutory stock options. The 2018 Plan also provides for the grant of stock appreciation rights, restricted stock awards, restricted stock unit awards, performance stock awards, performance cash awards and other stock awards. The exercise price of a stock option may not be less than 100% of the closing price of our common stock on the date of the grant. If, at any time we grant an incentive stock option, and the optionee directly or by attribution owns stock possessing more than 10% of the total combined voting power of all classes of our stock, the option price shall be at least 110% of the fair value and shall not be exercisable more than five years after the date of grant. Options generally vest over a period of one or four years from the date of grant. Options granted under the 2018 Plan expire no later than 10 years from the date of grant. As of December 31, 2020, there were 2,064,921 option shares outstanding, 493,742 restricted stock units granted but unvested, and 1,936,422 shares available for future grants under the 2018 Plan.
2014 Employee Stock Purchase Plan
In December 2014, our board of directors adopted the Kindred Biosciences, Inc. 2014 Employee Stock Purchase Plan (the “Purchase Plan”). A total of 200,000 shares of our common stock are authorized for issuance under the Purchase Plan. At the Annual Meeting of Stockholders of Kindred Biosciences, Inc. held on June 22, 2018, our stockholders approved an amendment to increase the number of shares that may be issued under the ESPP from 200,000 shares to 500,000 shares. The Purchase Plan permits eligible employees to purchase common stock at a discount through payroll deductions during defined six months consecutive offering periods beginning on December 1st. The price at which the stock is purchased is equal to the lower of 85% of the fair market value of the common stock on the first day of the offering or 85% of the fair market value of our common stock on the purchase date. A participant may purchase a maximum of 2,000 shares of common stock during each offering period, not to exceed $25,000 worth of common stock on the offering date during each calendar year. We use the Black-Scholes option pricing model, in combination with discounted employee price, in determining the value of the Purchase Plan expense to be recognized during each offering period. The weighted-average grant date fair value per share using the Black-Scholes option pricing model was $1.82 during the year ended December 31, 2020.
As of December 31, 2020, there were 300,887 shares of common stock issued under the Purchase Plan and 199,113 shares available for future issuance under the Purchase Plan. At December 31, 2020 and 2019, we had an outstanding liability of $16,000 and $40,000, respectively, which is included in accrued compensation on the consolidated balance sheets, for employee contributions to the Purchase Plan for shares pending issuance at the end of the next offering period.
Reserved Shares
At December 31, 2020, shares of common stock reserved for future issuance inclusive of outstanding option shares are as follows:
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | |
| 2018 Equity Incentive Plan | 1,936,422 | |
| 2014 Employee Stock Purchase Plan | 199,113 | |
| 2,135,535 | |
Stock Option Plan Activity Summary
A summary of activity under our stock option plans is as follows:
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Shares Available For Grant | | Shares Issuable Under Options | | Weighted Average Exercise Price | | Weighted Average Remaining Contractual Term (In Years) | | Aggregate Intrinsic Value |
| Balance, December 31, 2017 | 1,488,450 | | | 4,573,625 | | | $6.57 | | 7.2 | | $18,745,000 |
| 2012 Plan retired shares (a) | (56,953) | | | — | | | — | | | | |
| 2016 Plan RSA forfeited (b) | 26,980 | | | — | | | — | | | | |
| 2016 Plan RSU issued (c) | (315,000) | | | — | | | — | | | | |
| 2016 Plan retired shares (d) | (56,636) | | | — | | | — | | | | |
| 2018 Incentive Plan (e) | 3,000,000 | | | — | | | — | | | | |
| Granted | (1,607,193) | | | 1,607,193 | | | $9.78 | | | | |
| Exercised | — | | | (242,031) | | | $3.19 | | | | |
| Expired | 36,791 | | | (36,791) | | | $13.05 | | | | |
| Forfeited - stock options | 80,068 | | | (80,068) | | | $7.40 | | | | |
| Balance, December 31, 2018 | 2,596,507 | | | 5,821,928 | | | $7.54 | | 7.1 | | $24,780,000 |
| 2012 Plan retired shares (f) | (94,700) | | | — | | | — | | | | |
| 2016 Plan RSA forfeited (g) | 21,562 | | | — | | | — | | | | |
| 2016 Plan RSU forfeited (h) | 27,538 | | | — | | | — | | | | |
| 2016 Plan retired shares (i) | (191,491) | | | — | | | — | | | | |
| 2018 Plan RSU issued in 2019 (j) | (264,075) | | | — | | | — | | | | |
| Granted | (1,107,500) | | | 1,107,500 | | | $9.77 | | | | |
| Exercised | — | | | (305,801) | | | $5.21 | | | | |
| Expired | 104,710 | | | (104,710) | | | $13.26 | | | | |
| Forfeited - stock options | 165,547 | | | (165,547) | | | $7.97 | | | | |
| Balance, December 31, 2019 | 1,258,098 | | 6,353,370 | | $7.94 | | 6.6 | | $12,516,000 |
| 2012 Plan retired shares (k) | (92,879) | | | — | | | — | | | | |
| 2016 Plan RSA forfeited (l) | 33,959 | | | — | | | — | | | | |
| 2016 Plan RSU forfeited (m) | 51,967 | | | — | | | — | | | | |
| 2016 Plan retired shares (n) | (486,795) | | | — | | | — | | | | |
| 2018 Plan RSU issued in 2020 (o) | (638,665) | | | — | | | — | | | | |
| 2018 Plan RSU forfeited in 2020 (p) | 43,781 | | | — | | | — | | | | |
| 2018 Plan RSU cancelled in 2020 (q) | 304,052 | | | — | | | — | | | | |
| 2018 Plan add more share reserve (r) | 1,600,000 | | | — | | | — | | | | |
| RSU issued in July 2020 use 2 share reserve (s) | (43,750) | | | — | | | — | | | | |
| Granted | (781,500) | | | 781,500 | | | $9.57 | | | | |
| Exercised | — | | | (68,984) | | | $3.71 | | | | |
| Expired | 380,338 | | | (380,338) | | | $9.11 | | | | |
| Forfeited | 307,816 | | | (307,816) | | | $9.94 | | | | |
| Balance, December 31, 2020 | 1,936,422 | | | 6,377,732 | | | $8.02 | | 5.8 | | $3,441,000 |
| Options vested and expected to vest, December 31, 2020 | | | 6,377,732 | | | $8.02 | | 5.8 | | $3,441,000 |
| Options exercisable, December 31, 2020 | | | 5,331,249 | | | $7.72 | | 5.3 | | $3,434,000 |
(a) The 2012 Equity Incentive Plan terminated in May 2016. All shares available for grant under this Plan expired. True up all expired shares available for grant under the 2012 Equity Incentive Plan.
(b) Vested 62,500 RSA shares on January 23, 2018. 26,980 shares were forfeited to cover tax liability.
(c) Issued 315,000 RSU units on January 22, 2018 under the 2016 Equity Incentive Plan.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
(d) The 2016 Equity Incentive Plan terminated in June 2018. All shares available for grant under this Plan expired.
(e) The 2018 Equity Incentive Plan was adopted and approved by stockholders in June 2018.
(f) 2012 Equity Incentive Plan retired in May 2016. True up retirement in 2019.
(g) Vested 62,500 RSA shares on January 23, 2019. 21,562 shares were forfeited to cover tax liability.
(h) Vested 78,750 RSU units on January 22, 2019, 27,538 units were forfeited to cover tax liability.
(i) The 2016 Equity Incentive Plan terminated in June 2018. True up retirement in 2019.
(j) Issued 264,075 RSU units in Q1 2019 under the 2018 Equity Incentive Plan.
(k) 2012 Equity Incentive Plan retired in May 2016. True up retirement in 2020.
(l) Vested total 87,500 RSA shares on January 23, 2020 and July 31, 2020. 33,959 shares were forfeited to cover tax liability.
(m) Vested total 133,750 RSU units on January 22, 2020 and July 31, 2020, 51,967 units were forfeited to cover tax liability.
(n) The 2016 Equity Incentive Plan terminated in June 2018. All shares available for grant under this Plan expired.
(o) Issued 638,665 RSU units in 2020 under the 2018 Equity Incentive Plan.
(p) Vested 104,946 RSU units in 2020 under the 2018 Plan. 43,781 units were forfeited to cover tax liability.
(q) Due to employee termination, 304,052 units RSU has been cancelled from the 2018 Plan.
(r) In June 2020, shareholders approved to increase the number of shares of common stock authorized for issuance by 1,600,000 shares under the 2018 Plan amendment.
(s) Under the 2018 Plan amendment, each RSU granted after June 2020 under the 2018 Plan will use 2 share reverse units. The 43,750 RSU granted on July 15, 2020, will use additional 43,750 share reserve.
The aggregate intrinsic value of options is calculated as the difference between the exercise price of options and the fair value of our common stock for those options that had exercise prices lower than the fair value of our common stock on December 31, 2020, 2019 and 2018.
The aggregate intrinsic value of stock options exercised during the years ended December 31, 2020, 2019 and 2018 was $118,000, $1,046,000 and $2,261,000, respectively.
We received proceeds of $256,000, $1,591,000 and $635,000 from the exercise of common stock options during the years ended December 31, 2020, 2019 and 2018, respectively.
The weighted-average grant date fair value of options granted during the years ended December 31, 2020, 2019 and 2018 was $4.90, $5.34 and $5.58 per share, respectively.
We had an aggregate of approximately $4,577,000 of unrecognized stock-based compensation expense for unvested stock options and employee stock purchases as of December 31, 2020, which is expected to be recognized over a weighted average period of 2.2 years.
Restricted Stock
On January 22, 2018, we granted 315,000 shares of restricted stock units to four employees. Shares will vest 25% on each one year anniversary of the grant date provided that the employee is in the employment of the Company on such vesting date. In Q1 2019, we granted 300,775 shares of restricted stock units to most of our employees at the time of grant. Shares will vest 25% on each one year anniversary of the grant date provided that the employee is in the employment of the Company on such vesting date. In Q1 2020, we granted 586,915 shares of restricted stock units to most of our employees at the time of grant. Shares will vest 25% on each one year anniversary of the grant date provided that the employee is in the employment of the Company on such vesting date. In July 2020, we granted 51,750 shares of restricted stock units to most of our employees at the time of grant except officers. Shares will vest 100% on the one year anniversary of the grant date provided that the employee is in the employment of the Company on such vesting date. As of December 31, 2020, we have an aggregate of approximately $3,933,000 unrecognized stock-based compensation expense for unvested restricted stock awards and units which is expected to be recognized over a weighted-average period of 2.5 years.
Restricted stock and restricted stock unit activity for the year ended December 31, 2020, was as follows:
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | | | | | | | | | | |
| Restricted Stock Award / Restricted Stock Units | | Shares | | Weighted Average Grant Date Fair Value |
| Unvested balance at December 31, 2019 | | 625,325 | | | $9.01 |
| Granted | | 638,665 | | | 9.38 |
| Vested | | (326,196) | | | 8.60 |
| Forfeited | | (304,052) | | | 9.92 |
| Unvested balance at December 31, 2020 | | 633,742 | | | $9.16 |
Stock-Based Compensation
We recognize stock-based compensation expense for only the portion of awards that are expected to vest. In developing a forfeiture rate estimate, we have considered our historical experience to estimate pre-vesting forfeitures for service-based awards. The impact of a forfeiture rate adjustment will be recognized in full in the period of adjustment, and if the actual forfeiture rate is materially different from our estimate, we may be required to record adjustments to stock-based compensation expense in future periods.
The fair value of each stock option grant is estimated on the date of grant using the Black-Scholes option-pricing model. The Company's expected volatility is based upon the historical volatility of our common stock. The expected term of our common stock options has been determined utilizing the “simplified” method as we have insufficient historical experience for options grants overall, rendering existing historical experience irrelevant to expectations for current grants. The risk-free interest rate is determined by reference to the U.S. Treasury yield curve in effect at the time of grant of the award for time periods approximately equal to the expected term of the award. Expected dividend yield is based on the fact that we have never paid cash dividends and does not expect to pay any cash dividends in the foreseeable future.
Total stock-based compensation expense, related to all of our share-based payment awards, is comprised of the following:
| | | | | | | | | | | | | | | | | |
| (In thousands) | Years Ended December 31, |
| 2020 | | 2019 | | 2018 |
| Research and development | $1,883 | | $1,848 | | $1,746 |
| Selling, general and administrative | 5,704 | | 5,509 | | 4,531 |
| $7,587 | | $7,357 | | $6,277 |
Total stock-based compensation expense includes stock options, restricted stock awards, restricted stock units, and expense from the Purchase Plan for the years ended December 31, 2020, 2019, and 2018.
Valuation assumptions
The relevant data used to determine the fair value of stock-based awards is as follows:
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2020 | | 2019 | | 2018 |
| Stock options: | | | | | |
| Weighted average risk-free interest rate | 1.48% | | 2.49% | | 2.62% |
| Weighted average expected term (in years) | 6.2 | | 5.9 | | 5.9 |
| Weighted average expected volatility | 55.3% | | 56.9% | | 59.2% |
| Weighted average expected dividend yield | — | | — | | — |
| Fair value at grant date | $4.90 | | $5.34 | | $5.58 |
| | | | | |
| Employee stock purchase plan: | | | | | |
| Weighted average risk-free interest rate | 0.64% | | 2.17% | | 2.02% |
| Weighted average expected term (in years) | 0.5 | | 0.5 | | 0.5 |
| Weighted average expected volatility | 81.2% | | 47.5% | | 43.7% |
| Weighted average expected dividend yield | — | | — | | — |
| Fair value at grant date | $1.82 | | $2.62 | | $2.72 |
| | | | | |
| Restricted stock units: | | | | | |
| Fair value at grant date | $9.38 | | $10.49 | | $8.75 |
Equity Award Modifications
Stock Option Modifications
In connection with our restructuring, we eliminated 82 positions during the year ended December 31, 2020. We extended the post-employment exercise periods of vested stock options for 78 employees in connection with their separation from the Company. We accounted for the extension as a modification of an equity award under ASC 718 - "Stock Compensation". Accordingly, we recognized incremental stock compensation expense of approximately $120,000 during the year ended December 31, 2020.
Accelerating vesting of stock grants
On July 31, 2020, Ms. Bevers, our previous President and Chief Operating Officer, and KindredBio entered into a Severance and Release Agreement (the “Severance Agreement”) which provides, among other things, that Ms. Bevers is entitled to receive the severance compensation and benefits that are described in Section 4(c) of her Amended and Restated Executive Employment Agreement dated May 22, 2018, as amended by Amendment No. 1 dated October 19, 2018, that KindredBio previously filed with the Securities and Exchange Commission. Ms. Bevers is entitled to accelerated vesting in full of all of her outstanding stock options and other equity awards granted by KindredBio. On July 31, 2020, we accelerated the vesting of all of Ms. Bevers' stock options and other equity awards granted and accounted it as a modification of an equity award under ASC 718. Accordingly, we recognized stock compensation expense of approximately $547,000 for this transaction during the year ended December 31, 2020.
11. Leases
We have non-cancelable operating leases for laboratory space in Burlingame, California with several amendments to expand the facility. In February 2020, we further amended non-cancelable operating leases for laboratory space in Burlingame, California for an expansion of an additional 2,260 square feet of laboratory space commencing on May 1, 2020 and expiring on May 31, 2025. The total non-cancellable operating lease for the entire existing laboratory space is 13,736 square feet, expiring May 31, 2025. In August 2015, we entered into a non-cancelable operating lease for 3,126 square feet of office space in San Diego, California and
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
in June 2019, renewed the lease through February 2025. Our headquarters office lease for 8,090 square feet of office space in Burlingame, California expired November 30, 2020. In September 2020, we renewed our headquarters for only 6,900 square feet of office space for another 3 years, expiring November 30, 2023. In May 2019, we signed another lease in Burlingame, consisting of 1,346 square feet of space through April 2022. In addition, we have five equipment leases expiring through 2027.
Operating lease expense was $1,058,000, $1,204,000 and $814,000 for the years ended December 31, 2020, 2019 and 2018, respectively.
Supplemental cash flow information for the year ended December 31, 2020, related to operating leases was as follows (in thousands):
| | | | | | | | |
| Amortization of operating lease | | $ | 806 | |
| Cash paid within operating cash flows | | $ | 920 | |
| Right-of-use assets obtained in exchange for new or modified lease liabilities | | $ | 1,232 | |
Supplemental balance sheet information, as of December 31, 2020, related to operating leases was as follows (in thousands, except lease term and discount rate):
| | | | | | | | |
| Reported as: | | |
| Operating lease right-of-use assets | | $ | 3,428 | |
| | |
| Current portion of operating lease liabilities | | $ | 825 | |
| Long-term operating lease liabilities | | 2,934 | |
| Total lease liabilities | | $ | 3,759 | |
| | |
| Weighted average remaining lease term (years) | | 4.3 years |
| Weighted average discount rate | | 5.5% |
As of December 31, 2020, we are obligated to make minimum lease payments under non-cancelable operating leases, as follows (in thousands):
| | | | | | | | |
| Year ending December 31, | | Lease Payments |
| 2021 | | $ | 1,012 | |
| 2022 | | 1,058 | |
| 2023 | | 1,047 | |
| 2024 | | 837 | |
| 2025 | | 249 | |
| 2026 and thereafter | | 9 | |
| Total lease payments | | 4,212 | |
| Less: imputed interest | | (453) | |
| Total lease liabilities | | $ | 3,759 | |
12. Commitments and Contingencies
Purchase Commitments
In June 2018, we entered into a Strategic Supply Agreement (the “Agreement”), with Pall Corporation (“Pall”) for purchase of equipment and consumables to be used in support of our manufacturing requirements, including, but not limited to, the Plant. Pursuant to the Agreement, we will purchase certain pharmaceutical
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
manufacturing equipment and related services in the aggregate amount of $3.8 million with a seven-year consumable purchase obligation in the aggregate amount of approximately $16.5 million. The Agreement is subject to customary undertakings, covenants, obligations, rights and conditions. As of December 31, 2020, we have incurred the full $3.8 million in equipment purchase costs and are obligated to make consumable purchases as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | |
| Year ending December 31, | | Consumable commitments | | Consumable purchases | | Remaining commitments |
| 2021 | | $ | 3,300 | | | $ | — | | | $ | 3,300 | |
| 2022 | | 3,625 | | | — | | | 3,625 | |
| 2023 | | 3,625 | | | — | | | 3,625 | |
| 2024 | | 4,285 | | | — | | | 4,285 | |
| Total | | $ | 14,835 | | | $ | — | | | $ | 14,835 | |
Indemnities and Guarantees
We have made certain indemnities and guarantees, under which we may be required to make payments to a guaranteed or indemnified party, in relation to certain transactions. We indemnify our officers and directors to the maximum extent permitted under the laws of the State of Delaware. The duration of these indemnities and guarantees varies and, in certain cases, is indefinite. These indemnities and guarantees do not provide for any limitation of the maximum potential future payments we could be obligated to make. Historically, we have not been obligated to make any payments for these obligations and no liabilities have been recorded for these indemnities and guarantees in the accompanying consolidated balance sheets.
Legal Matters
In the ordinary course of business, we may face various claims brought by third parties and may, from time to time, make claims or take legal actions to assert our rights, including intellectual property disputes, contractual disputes and other commercial disputes. Any of these claims could subject us to litigation. Management believes there are currently no claims that are likely to have a material effect on our consolidated financial position and results of operations.
13. Income Taxes
There is no provision for income taxes because we have historically incurred operating losses and we maintain a full valuation allowance against our net deferred tax assets.
Differences between the provision (benefit) for income taxes and income taxes at the statutory federal income tax rate are as follows:
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (In thousands, except percentages) | Years Ended December 31, |
| 2020 | | 2019 | | 2018 |
| Income tax expense (benefit) at statutory federal rate | $ | (4,578) | | | 21.0 | % | | $ | (12,893) | | | 21.0 | % | | $ | (10,436) | | | 21.0 | % |
| State income tax, net of federal benefit | (1,222) | | | 5.6 | | | (3,392) | | | 5.5 | | | (3,748) | | | 7.5 | |
| Permanent items | (48) | | | 0.2 | | | 198 | | | (0.3) | | | 13 | | | — | |
| Executive compensation | 469 | | | (2.2) | | | — | | | — | | | — | | | — | |
| Research and development credits | (2,107) | | | 9.7 | | | (1,913) | | | 3.1 | | | (1,909) | | | 3.8 | |
| Stock-based compensation | 464 | | | (2.1) | | | 637 | | | (1.0) | | | (237) | | | 0.5 | |
| Reserve for uncertain tax positions | (3) | | | — | | | 861 | | | (1.4) | | | 864 | | | (1.7) | |
| Change in valuation allowance | 796 | | | (3.7) | | | 15,382 | | | (25.1) | | | 14,949 | | | (30.1) | |
| Tax attribute ownership change reduction | 6,457 | | | (29.6) | | | — | | | — | | | — | | | — | |
| Tax rate and other | (228) | | | 1.1 | | | 1,120 | | | (1.8) | | | 504 | | | (1.0) | |
| Provision (benefit) for income taxes | $ | — | | | — | % | | $ | — | | | — | % | | $ | — | | | — | % |
Deferred tax assets are recognized for temporary differences that will result in deductible amounts in future periods. The components of the deferred tax assets are as follows at December 31, 2020 and 2019:
| | | | | | | | | | | |
| December 31, |
| (In thousands) | 2020 | | 2019 |
| Deferred tax assets: | | | |
| Net operating loss carryforwards | $ | 53,506 | | | $ | 53,247 | |
| Research and development credits | 4,933 | | | 4,682 | |
| Accrued expenses | 517 | | | 1,020 | |
| Amortization and depreciation | (447) | | | (222) | |
| Stock-based compensation | 7,401 | | | 6,412 | |
| ROU Lease - Liabilities | 1,039 | | | 872 | |
| ROU Lease - Assets | (947) | | | (804) | |
| Other | 21 | | | 20 | |
| 66,023 | | | 65,227 | |
| Valuation Allowance | (66,023) | | | (65,227) | |
| Net current deferred tax assets | $ | — | | | $ | — | |
At December 31, 2020, we had net deferred tax assets of $66,023,000. Due to uncertainties surrounding our ability to generate future taxable income to realize these assets, a full valuation allowance has been established to offset the net deferred tax asset.
Additionally, the future utilization of our net operating loss and research and development tax credits carryforwards is subject to annual limitation under Sections 382 and 383 of the Internal Revenue Code of 1986, and similar state tax provisions due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes limit the amount of the net operating loss and research and development tax credit carryforward and other deferred tax assets that can be utilized to offset future taxable income and tax, respectively. In general, an ownership change, as defined by Sections 382 and 383, results from transactions increasing ownership of certain stockholders or public groups in the stock of the
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
corporation by more than 50 percent points over a three-year period. In 2020, the Company performed a preliminary IRC Section 382 study and determined that a total of $15,633,000 of federal and $18,891,000 California NOLs will be permanently disallowed. These amounts have been excluded from the tables above. The remaining net operating losses have been recognized.
At December 31, 2020, we had federal and California net operating loss carryovers of $211,817,000 and $117,203,000, respectively. The federal and California net loss carryforwards will begin to expire in 2032. The Federal NOL generated after 2017 of $118,704,000 will carryforward indefinitely and be available to offset up to 80% of future taxable income each year.
On March 27, 2020, the CARES Act was signed into law in response to the economic challenges facing US business. Under the CARES Act, the Internal Revenue Code was amended to allow for federal NOL carrybacks for five years to offset previous years income, or can be carryforward indefinitely to offset 100% of taxable income for the tax year 2020 and 80% of taxable income for tax years 2021 and thereafter.
At December 31, 2020, we had federal and state research and development tax credit carryovers of approximately $6,383,000 and $5,307,000, respectively. The federal research and development tax credit carryforwards will begin to expire in 2033. The California research and development credit carryforwards are available indefinitely.
The impact of an uncertain income tax position on the income tax return must be recognized at the largest amount that is more likely than not to be sustained upon audit by the relevant taxing authority. An uncertain income tax position will not be recognized if it has less than a 50% likelihood of being sustained. There were no unrecognized tax benefits recorded by us as of the date of adoption. As a result of the implementation, we did not recognize an increase in the liability for unrecognized tax benefits.
A roll forward of changes in our unrecognized tax benefits is shown below.
| | | | | | | | | | | | | | | | | |
| (In thousands) | December 31, |
| 2020 | | 2019 | | 2018 |
| Balance at beginning of year | $ | 4,175 | | | $ | 3,196 | | | $ | 2,252 | |
| Additions based on tax positions related to the current year | 58 | | | 942 | | | 944 | |
| Additions for tax positions of prior years | — | | | 37 | | | — | |
| Balance at end of year | $ | 4,233 | | | $ | 4,175 | | | $ | 3,196 | |
The amount of unrecognized tax benefits that would impact the effective tax rate if recognized and realized is $3,788,000.
Our practice is to recognize interest and/or penalties related to income tax matters as income tax expense. We had no accrual for interest or penalties on our accompanying consolidated balance sheets at December 31, 2020, 2019 and 2018, and have not recognized interest and/or penalties in our consolidated statements of operations and comprehensive loss for the years ended December 31, 2020, 2019 and 2018.
We do not anticipate a significant change to our unrecognized tax benefits during the next twelve months.
We file tax returns as prescribed by tax laws of the jurisdictions in which we operate. In the normal course of business, we are subject to examination by federal and state jurisdictions, where applicable. There are currently no pending tax examinations. Our federal and state tax returns are still open under statute from 2015 to present.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
14. Net Loss Per Share
Basic and diluted net loss per share was calculated as follows for the years ended December 31, 2020, 2019 and 2018:
| | | | | | | | | | | | | | | | | |
| (In thousands, except per share amounts) | Years Ended December 31, |
| 2020 | | 2019 | | 2018 |
| Basic and diluted net loss per share attributable to common stockholders: | | | | | |
| Numerator: | | | | | |
| Net loss attributable to common stockholders | $ | (21,796) | | | $ | (61,389) | | | $ | (49,690) | |
| Denominator: | | | | | |
| Weighted-average number of common shares outstanding, basic and diluted | 39,289 | | | 38,657 | | | 31,001 | |
| Net loss per common share attributable to common stockholders, basic and diluted | $ | (0.55) | | | $ | (1.59) | | | $ | (1.60) | |
There was no difference between our net loss and the net loss attributable to common stockholders for all periods presented.
Stock options to purchase 6,377,732 shares, 6,353,370 shares and 5,821,928 shares of common stock as of December 31, 2020, 2019 and 2018, respectively, were excluded from the computation of diluted net loss per share attributable to common stockholders because their effect was anti-dilutive. 37,500 shares, 125,000 shares, 187,500 shares of unvested restricted stock award as of December 31, 2020, 2019 and 2018, respectively, were also excluded from the computation of diluted net loss per share calculations because their effect was anti-dilutive. 596,242 units, 500,325 units and 315,000 units of granted but unvested restricted stock units as of December 31, 2020, 2019 and 2018, respectively, were also excluded from the computation of diluted net loss per share calculations because their effect was anti-dilutive.
15. Employee Savings Plan
We have established an employee savings plan pursuant to Section 401(k) of the Internal Revenue Code, effective May 1, 2014. The plan allows participating employees to deposit into tax deferred investment accounts up to 90% of their salary, subject to annual limits. We make contributions to the plan in an amount equal to 50% on the first 6% for a maximum of 3% of the participant’s compensation which is deferred. We contributed approximately $302,000, $452,000 and $292,000 to the plan during the years ended December 31, 2020, 2019 and 2018, respectively.
16. Restructuring Plan
On March 16, 2020, we announced a strategic realignment of our business model whereby we plan to rely more on a partnership-based model for commercialization strategy similar to the traditional human biotech commercialization strategy whereby pipeline assets are partnered with larger commercial partners that can maximize product opportunity in return for upfront payments, contingent milestones, and royalties on future sales. Our focus will be on accelerating our deep pipeline of late-stage biologics candidates in canine and feline markets, while stopping most small molecule development for these species. Accordingly, the companion animal commercial infrastructure will be substantially reduced. In connection with this restructuring, we eliminated 53 positions, representing about one-third of our workforce. The eliminated positions primarily relate to the companion animal sales force and research and development for small molecule programs. Restructuring expenses and retirement costs related to severance and health care benefits were approximately $1.7 million, exclusive of stock compensation.
Kindred Biosciences, Inc
Notes to Consolidated Financial Statements
On June 8, 2020, we announced a plan to strengthen our strategic position by, among other things, prioritizing our most attractive late stage programs and substantially reducing our expenses to best position the Company for success with the previously announced business model. This restructuring reduced our workforce by approximately 24 employees and involved a restructuring charge of approximately $2.3 million related to severance payments and health care benefits, exclusive of stock compensation. We further eliminated another 5 positions and incurred a restructuring charge of approximately $0.3 million related to severance payments and health care benefits in the third quarter of 2020. We have completed our restructuring and do not anticipate any further reductions in our workforce for the foreseeable future.
17. Subsequent Events
From January 1, 2021 through March 10, 2021, 1,831,967 shares were sold through the ATM, for total gross proceeds of approximately $9,028,000. Net proceeds, after deducting underwriting discounts and commissions and offering expense, were approximately $8,793,000.