0001551152false2022FYhttp://fasb.org/us-gaap/2022#CostOfGoodsAndServicesSold http://fasb.org/us-gaap/2022#ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost http://fasb.org/us-gaap/2022#SellingGeneralAndAdministrativeExpensehttp://fasb.org/us-gaap/2022#CostOfGoodsAndServicesSold http://fasb.org/us-gaap/2022#ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost http://fasb.org/us-gaap/2022#SellingGeneralAndAdministrativeExpensehttp://fasb.org/us-gaap/2022#CostOfGoodsAndServicesSold http://fasb.org/us-gaap/2022#ResearchAndDevelopmentExpenseExcludingAcquiredInProcessCost http://fasb.org/us-gaap/2022#SellingGeneralAndAdministrativeExpensehttp://fasb.org/us-gaap/2022#OtherAssetsNoncurrenthttp://fasb.org/us-gaap/2022#OtherAssetsNoncurrenthttp://fasb.org/us-gaap/2022#PropertyPlantAndEquipmentNethttp://fasb.org/us-gaap/2022#PropertyPlantAndEquipmentNethttp://fasb.org/us-gaap/2022#AccountsPayableAndAccruedLiabilitiesCurrenthttp://fasb.org/us-gaap/2022#AccountsPayableAndAccruedLiabilitiesCurrenthttp://fasb.org/us-gaap/2022#OtherLiabilitiesNoncurrenthttp://fasb.org/us-gaap/2022#OtherLiabilitiesNoncurrenthttp://fasb.org/us-gaap/2022#LongTermDebtAndCapitalLeaseObligationsCurrenthttp://fasb.org/us-gaap/2022#LongTermDebtAndCapitalLeaseObligationsCurrenthttp://fasb.org/us-gaap/2022#LongTermDebtAndCapitalLeaseObligationshttp://fasb.org/us-gaap/2022#LongTermDebtAndCapitalLeaseObligationshttp://fasb.org/us-gaap/2022#SellingGeneralAndAdministrativeExpensehttp://fasb.org/us-gaap/2022#CostOfGoodsAndServicesSoldhttp://fasb.org/us-gaap/2022#CostOfGoodsAndServicesSoldhttp://fasb.org/us-gaap/2022#CostOfGoodsAndServicesSoldhttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#ForeignCurrencyTransactionGainLossBeforeTaxhttp://fasb.org/us-gaap/2022#ForeignCurrencyTransactionGainLossBeforeTaxhttp://fasb.org/us-gaap/2022#ForeignCurrencyTransactionGainLossBeforeTaxhttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#InterestIncomeExpenseNonoperatingNethttp://fasb.org/us-gaap/2022#OtherNonoperatingIncomeExpensehttp://fasb.org/us-gaap/2022#OtherNonoperatingIncomeExpensehttp://fasb.org/us-gaap/2022#OtherNonoperatingIncomeExpenseP3Y333333100015511522022-01-012022-12-310001551152us-gaap:CommonStockMemberexch:XCHI2022-01-012022-12-310001551152us-gaap:CommonStockMemberexch:XNYS2022-01-012022-12-310001551152exch:XNYSabbv:Sec1500SeniorNotesDue2023Member2022-01-012022-12-310001551152exch:XNYSabbv:Sec1.375SeniorNotesDue2024Member2022-01-012022-12-310001551152exch:XNYSabbv:Sec1250SeniorNotesDue2024Member2022-01-012022-12-310001551152abbv:Sec0.750SeniorNotesDue2027Memberexch:XNYS2022-01-012022-12-310001551152exch:XNYSabbv:Sec2.125SeniorNotesdue2028Member2022-01-012022-12-310001551152exch:XNYSabbv:Sec2625SeniorNotesDue2028Member2022-01-012022-12-310001551152exch:XNYSabbv:Sec2125SeniorNotesDue2029Member2022-01-012022-12-310001551152abbv:Sec1250SeniorNotesDue2031Memberexch:XNYS2022-01-012022-12-3100015511522022-06-30iso4217:USD00015511522023-01-31xbrli:shares00015511522021-01-012021-12-3100015511522020-01-012020-12-31iso4217:USDxbrli:shares00015511522022-12-3100015511522021-12-310001551152us-gaap:CommonStockMember2019-12-310001551152us-gaap:TreasuryStockMember2019-12-310001551152us-gaap:AdditionalPaidInCapitalMember2019-12-310001551152us-gaap:RetainedEarningsMember2019-12-310001551152us-gaap:AccumulatedOtherComprehensiveIncomeMember2019-12-310001551152us-gaap:NoncontrollingInterestMember2019-12-3100015511522019-12-310001551152us-gaap:CommonStockMember2020-01-012020-12-310001551152us-gaap:TreasuryStockMember2020-01-012020-12-310001551152us-gaap:AdditionalPaidInCapitalMember2020-01-012020-12-310001551152us-gaap:RetainedEarningsMember2020-01-012020-12-310001551152us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-01-012020-12-310001551152us-gaap:NoncontrollingInterestMember2020-01-012020-12-310001551152us-gaap:CommonStockMember2020-12-310001551152us-gaap:TreasuryStockMember2020-12-310001551152us-gaap:AdditionalPaidInCapitalMember2020-12-310001551152us-gaap:RetainedEarningsMember2020-12-310001551152us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-12-310001551152us-gaap:NoncontrollingInterestMember2020-12-3100015511522020-12-310001551152us-gaap:CommonStockMember2021-01-012021-12-310001551152us-gaap:TreasuryStockMember2021-01-012021-12-310001551152us-gaap:AdditionalPaidInCapitalMember2021-01-012021-12-310001551152us-gaap:RetainedEarningsMember2021-01-012021-12-310001551152us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-01-012021-12-310001551152us-gaap:NoncontrollingInterestMember2021-01-012021-12-310001551152us-gaap:CommonStockMember2021-12-310001551152us-gaap:TreasuryStockMember2021-12-310001551152us-gaap:AdditionalPaidInCapitalMember2021-12-310001551152us-gaap:RetainedEarningsMember2021-12-310001551152us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-12-310001551152us-gaap:NoncontrollingInterestMember2021-12-310001551152us-gaap:CommonStockMember2022-01-012022-12-310001551152us-gaap:TreasuryStockMember2022-01-012022-12-310001551152us-gaap:AdditionalPaidInCapitalMember2022-01-012022-12-310001551152us-gaap:RetainedEarningsMember2022-01-012022-12-310001551152us-gaap:AccumulatedOtherComprehensiveIncomeMember2022-01-012022-12-310001551152us-gaap:NoncontrollingInterestMember2022-01-012022-12-310001551152us-gaap:CommonStockMember2022-12-310001551152us-gaap:TreasuryStockMember2022-12-310001551152us-gaap:AdditionalPaidInCapitalMember2022-12-310001551152us-gaap:RetainedEarningsMember2022-12-310001551152us-gaap:AccumulatedOtherComprehensiveIncomeMember2022-12-310001551152us-gaap:NoncontrollingInterestMember2022-12-3100015511522013-01-012013-01-01xbrli:pure0001551152us-gaap:LandMember2022-12-310001551152us-gaap:LandMember2021-12-310001551152us-gaap:BuildingMember2022-12-310001551152us-gaap:BuildingMember2021-12-310001551152us-gaap:EquipmentMember2022-12-310001551152us-gaap:EquipmentMember2021-12-310001551152us-gaap:ConstructionInProgressMember2022-12-310001551152us-gaap:ConstructionInProgressMember2021-12-310001551152us-gaap:BuildingMembersrt:MinimumMember2022-01-012022-12-310001551152us-gaap:BuildingMembersrt:MaximumMember2022-01-012022-12-310001551152us-gaap:EquipmentMembersrt:MinimumMember2022-01-012022-12-310001551152srt:MaximumMemberus-gaap:EquipmentMember2022-01-012022-12-310001551152abbv:DJSAntibodiesLTDMember2022-10-012022-10-310001551152abbv:DJSAntibodiesLTDMember2022-10-310001551152abbv:SolitonIncMember2021-12-012021-12-310001551152abbv:SolitonIncMember2021-12-310001551152abbv:LumineraMember2020-10-012020-10-310001551152abbv:LumineraMember2020-10-310001551152abbv:AllerganplcMember2020-05-080001551152abbv:AllerganplcMember2020-05-082020-05-0800015511522020-05-080001551152us-gaap:FiniteLivedIntangibleAssetsMemberabbv:AllerganplcMember2021-01-012021-06-300001551152abbv:AllerganplcMemberus-gaap:IndefinitelivedIntangibleAssetsMember2021-01-012021-06-300001551152abbv:AllerganplcMember2021-01-012021-06-300001551152abbv:AllerganplcMember2020-05-082020-12-310001551152abbv:AllerganplcMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2020-01-012020-12-310001551152abbv:AllerganplcMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2021-01-012021-12-310001551152abbv:AllerganplcMember2020-01-012020-12-310001551152abbv:SyndesiTherapeuticsSAMember2022-01-012022-03-310001551152us-gaap:CollaborativeArrangementMemberabbv:SyndesiTherapeuticsSAMember2022-02-280001551152abbv:JuvisePharmaceuticalsMember2022-04-012022-06-300001551152us-gaap:OtherOperatingIncomeExpenseMemberabbv:JuvisePharmaceuticalsMember2022-04-012022-06-300001551152us-gaap:CollaborativeArrangementMemberabbv:CalicoLifeSciencesLLCMember2021-07-012021-09-300001551152us-gaap:CollaborativeArrangementMemberabbv:TeneoOneMember2021-07-012021-09-300001551152srt:MaximumMemberus-gaap:CollaborativeArrangementMemberabbv:TeneoOneMember2021-09-300001551152us-gaap:CollaborativeArrangementMemberabbv:REGENXBIOIncMember2021-10-012021-12-310001551152srt:MaximumMemberus-gaap:CollaborativeArrangementMemberabbv:REGENXBIOIncMember2021-09-300001551152us-gaap:CollaborativeArrangementMemberabbv:AcquiredIPRDAndMilestonesMemberabbv:IMabBiopharmaMember2020-09-012020-09-300001551152us-gaap:CollaborativeArrangementMemberabbv:IMabBiopharmaMember2020-10-012020-12-310001551152srt:MaximumMemberus-gaap:CollaborativeArrangementMemberabbv:IMabBiopharmaMember2020-09-300001551152us-gaap:CollaborativeArrangementMemberabbv:GenmabASMember2020-01-012020-12-310001551152srt:MaximumMemberus-gaap:CollaborativeArrangementMemberabbv:GenmabASMember2020-06-300001551152us-gaap:CollaborativeArrangementMemberabbv:OtherCollaborationArrangementsMember2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:OtherCollaborationArrangementsMember2021-01-012021-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:OtherCollaborationArrangementsMember2020-01-012020-12-310001551152srt:MaximumMemberus-gaap:CollaborativeArrangementMemberabbv:OtherCollaborationArrangementsMember2022-12-310001551152us-gaap:CollaborativeArrangementMember2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMember2021-01-012021-12-310001551152us-gaap:CollaborativeArrangementMember2020-01-012020-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMember2022-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMember2022-01-012022-12-310001551152abbv:JanssenBiotechIncMember2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMembercountry:USabbv:JanssenBiotechIncMember2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMembercountry:USabbv:JanssenBiotechIncMember2021-01-012021-12-310001551152us-gaap:CollaborativeArrangementMembercountry:USabbv:JanssenBiotechIncMember2020-01-012020-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMemberus-gaap:NonUsMember2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMemberus-gaap:NonUsMember2021-01-012021-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMemberus-gaap:NonUsMember2020-01-012020-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMember2021-01-012021-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMember2020-01-012020-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:JanssenBiotechIncMember2021-12-310001551152abbv:GenentechInc.Member2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:GenentechInc.Member2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:GenentechInc.Member2021-01-012021-12-310001551152us-gaap:CollaborativeArrangementMemberabbv:GenentechInc.Member2020-01-012020-12-310001551152us-gaap:CollaborativeArrangementMembercountry:USabbv:GenentechInc.Member2022-01-012022-12-310001551152us-gaap:CollaborativeArrangementMembercountry:USabbv:GenentechInc.Member2021-01-012021-12-310001551152us-gaap:CollaborativeArrangementMembercountry:USabbv:GenentechInc.Member2020-01-012020-12-310001551152us-gaap:DevelopedTechnologyRightsMember2022-12-310001551152us-gaap:DevelopedTechnologyRightsMember2021-12-310001551152us-gaap:LicensingAgreementsMember2022-12-310001551152us-gaap:LicensingAgreementsMember2021-12-3100015511522022-07-012022-09-300001551152srt:MinimumMember2022-01-012022-12-310001551152srt:MaximumMember2022-01-012022-12-310001551152srt:WeightedAverageMemberus-gaap:DevelopedTechnologyRightsMember2022-01-012022-12-310001551152srt:WeightedAverageMemberus-gaap:LicensingAgreementsMember2022-01-012022-12-310001551152abbv:AllerganIntegrationPlanMember2022-12-310001551152abbv:AllerganIntegrationPlanMember2022-01-012022-12-310001551152us-gaap:EmployeeSeveranceMemberus-gaap:CostOfSalesMemberabbv:AllerganIntegrationPlanMember2022-01-012022-12-310001551152us-gaap:EmployeeSeveranceMemberus-gaap:CostOfSalesMemberabbv:AllerganIntegrationPlanMember2021-01-012021-12-310001551152us-gaap:EmployeeSeveranceMemberus-gaap:CostOfSalesMemberabbv:AllerganIntegrationPlanMember2020-01-012020-12-310001551152us-gaap:CostOfSalesMemberabbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2022-01-012022-12-310001551152us-gaap:CostOfSalesMemberabbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2021-01-012021-12-310001551152us-gaap:CostOfSalesMemberabbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2020-01-012020-12-310001551152us-gaap:EmployeeSeveranceMemberus-gaap:ResearchAndDevelopmentExpenseMemberabbv:AllerganIntegrationPlanMember2022-01-012022-12-310001551152us-gaap:EmployeeSeveranceMemberus-gaap:ResearchAndDevelopmentExpenseMemberabbv:AllerganIntegrationPlanMember2021-01-012021-12-310001551152us-gaap:EmployeeSeveranceMemberus-gaap:ResearchAndDevelopmentExpenseMemberabbv:AllerganIntegrationPlanMember2020-01-012020-12-310001551152us-gaap:ResearchAndDevelopmentExpenseMemberabbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2022-01-012022-12-310001551152us-gaap:ResearchAndDevelopmentExpenseMemberabbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2021-01-012021-12-310001551152us-gaap:ResearchAndDevelopmentExpenseMemberabbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2020-01-012020-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2022-01-012022-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2021-01-012021-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2020-01-012020-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2022-01-012022-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2021-01-012021-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMemberus-gaap:SellingGeneralAndAdministrativeExpensesMember2020-01-012020-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMember2022-01-012022-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMember2021-01-012021-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMember2020-01-012020-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2022-01-012022-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2021-01-012021-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2020-01-012020-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMember2020-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2020-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMember2021-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2021-12-310001551152us-gaap:EmployeeSeveranceMemberabbv:AllerganIntegrationPlanMember2022-12-310001551152abbv:AllerganIntegrationPlanMemberus-gaap:OtherRestructuringMember2022-12-310001551152abbv:OtherRestructuringPlansMemberus-gaap:EmployeeSeveranceMember2022-01-012022-12-310001551152abbv:OtherRestructuringPlansMemberus-gaap:EmployeeSeveranceMember2021-01-012021-12-310001551152abbv:OtherRestructuringPlansMemberus-gaap:EmployeeSeveranceMember2020-01-012020-12-310001551152abbv:OtherRestructuringPlansMember2022-01-012022-12-310001551152abbv:OtherRestructuringPlansMember2019-12-310001551152abbv:OtherRestructuringPlansMember2020-01-012020-12-310001551152abbv:OtherRestructuringPlansMember2020-12-310001551152abbv:OtherRestructuringPlansMember2021-01-012021-12-310001551152abbv:OtherRestructuringPlansMember2021-12-310001551152abbv:OtherRestructuringPlansMember2022-12-3100015511522022-12-012022-12-310001551152us-gaap:LeaseholdImprovementsMember2022-12-012022-12-310001551152abbv:AggregateNotesDue2022Membersrt:MinimumMember2022-12-310001551152abbv:AggregateNotesDue2022Membersrt:MaximumMember2022-12-310001551152abbv:AggregateNotesDue2022Member2022-12-310001551152abbv:AggregateNotesDue2022Membersrt:MinimumMember2021-12-310001551152abbv:AggregateNotesDue2022Membersrt:MaximumMember2021-12-310001551152abbv:AggregateNotesDue2022Member2021-12-310001551152abbv:SeniorNotesDuein2023At3.75PercentMemberus-gaap:SeniorNotesMember2022-12-310001551152abbv:SeniorNotesDuein2023At3.75PercentMemberus-gaap:SeniorNotesMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes2.85PercentDue2023Member2022-12-310001551152us-gaap:SeniorNotesMember2022-12-310001551152us-gaap:SeniorNotesMember2021-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2023Memberus-gaap:LoansPayableMember2022-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2023Memberus-gaap:LoansPayableMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2023At1.500PercentMember2022-12-31iso4217:EUR0001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2023At1.500PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2023At2.800PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2023At2.800PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2024At2.60PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2024At2.60PercentMember2021-12-310001551152abbv:SeniorEuroNotes1375PercentDue2024Memberus-gaap:SeniorNotesMember2022-12-310001551152abbv:SeniorEuroNotes1375PercentDue2024Memberus-gaap:SeniorNotesMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2024at3.850PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2024at3.850PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2024At1.250PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2024At1.250PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2025At3.60PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2025At3.60PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2025at3.800PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2025at3.800PercentMember2021-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2025Memberus-gaap:LoansPayableMember2022-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2025Memberus-gaap:LoansPayableMember2021-12-310001551152us-gaap:LoansPayableMemberabbv:FloatingRateTermLoanTrancheDueMay2025RefinancedMember2022-12-310001551152us-gaap:LoansPayableMemberabbv:FloatingRateTermLoanTrancheDueMay2025RefinancedMember2021-12-310001551152abbv:SeniorNotesDuein2026At2.95PercentMemberus-gaap:SeniorNotesMember2022-12-310001551152abbv:SeniorNotesDuein2026At2.95PercentMemberus-gaap:SeniorNotesMember2021-12-310001551152abbv:SeniorNotes3.20PercentDue2026Memberus-gaap:SeniorNotesMember2022-12-310001551152abbv:SeniorNotes3.20PercentDue2026Memberus-gaap:SeniorNotesMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDuein2027at0.750PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDuein2027at0.750PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2028At4.25PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2028At4.25PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotes2125PercentDue2028Member2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotes2125PercentDue2028Member2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2028At2.625PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2028At2.625PercentMember2021-12-310001551152abbv:SeniorNotesDuein2029At3.20PercentMemberus-gaap:SeniorNotesMember2022-12-310001551152abbv:SeniorNotesDuein2029At3.20PercentMemberus-gaap:SeniorNotesMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2029At2.125PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2029At2.125PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDuein2031at1.250PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDuein2031at1.250PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2035at4.550PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2035at4.550PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2035At4.50PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2035At4.50PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes4.30PercentDue2036Member2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes4.30PercentDue2036Member2021-12-310001551152abbv:SeniorNotesDuein2039At4.05PercentMemberus-gaap:SeniorNotesMember2022-12-310001551152abbv:SeniorNotesDuein2039At4.05PercentMemberus-gaap:SeniorNotesMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2042At4.40PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2042At4.40PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2042at4.625PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2042at4.625PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2044at4.850Member2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2044at4.850Member2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2045At4.70PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2045At4.70PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2045at4.750PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2045at4.750PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes4.45PercentDue2046Member2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes4.45PercentDue2046Member2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2048At4.875PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2048At4.875PercentMember2021-12-310001551152abbv:SeniorNotesDuein2049At4.25PercentMemberus-gaap:SeniorNotesMember2022-12-310001551152abbv:SeniorNotesDuein2049At4.25PercentMemberus-gaap:SeniorNotesMember2021-12-310001551152abbv:OtherLongTermDebtMember2022-12-310001551152abbv:OtherLongTermDebtMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes3450PercentDue2022Member2022-01-012022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes3450PercentDue2022Member2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes325PercentDue2022Member2022-01-012022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes325PercentDue2022Member2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2022At3.20PercentMember2022-01-012022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2022At3.20PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2022At2.90PercentMember2022-01-012022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueIn2022At2.90PercentMember2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes23PercentDue2022Member2022-01-012022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes23PercentDue2022Member2022-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2022AtFloatingRatesMember2022-01-012022-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2025Memberus-gaap:LoansPayableMember2022-01-012022-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2023RefinancedMemberus-gaap:LoansPayableMemberus-gaap:SubsequentEventMember2023-01-012023-02-170001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes2.30PercentDue2021Member2021-01-012021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotes2.30PercentDue2021Member2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2021At0.500PercentMember2021-01-012021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorEuroNotesDue2021At0.500PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2021At5.000PercentMember2021-01-012021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDue2021At5.000PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2021At3.375PercentMember2021-01-012021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2021At3.375PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2021At2.15PercentMember2021-01-012021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDuein2021At2.15PercentMember2021-12-310001551152us-gaap:SeniorNotesMemberabbv:SeniorNotesDueinMay2021AtFloatingRatesMember2021-01-012021-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2023Memberus-gaap:LoansPayableMember2021-01-012021-12-310001551152abbv:FloatingRateTermLoanTrancheDueMay2023RefinancedMemberus-gaap:LoansPayableMember2021-12-310001551152us-gaap:CommercialPaperMember2022-12-310001551152us-gaap:CommercialPaperMember2021-12-310001551152us-gaap:RevolvingCreditFacilityMember2022-01-012022-12-310001551152us-gaap:RevolvingCreditFacilityMember2022-12-310001551152us-gaap:RevolvingCreditFacilityMember2021-12-31abbv:derivative0001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2022-01-012022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:TreasuryLockMemberus-gaap:CashFlowHedgingMember2019-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMember2022-12-310001551152us-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMember2021-12-310001551152us-gaap:SeniorNotesMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:NetInvestmentHedgingMember2021-12-310001551152us-gaap:SeniorNotesMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:NetInvestmentHedgingMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2022-12-31iso4217:SEKiso4217:CADiso4217:CHF0001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2021-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2021-12-310001551152us-gaap:OtherNoncurrentAssetsMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2022-12-310001551152us-gaap:OtherNoncurrentAssetsMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:OtherLiabilitiesMemberus-gaap:ForeignExchangeForwardMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:OtherLiabilitiesMemberus-gaap:ForeignExchangeForwardMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2021-12-310001551152us-gaap:OtherNoncurrentAssetsMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2022-12-310001551152us-gaap:OtherNoncurrentAssetsMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:OtherLiabilitiesMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:OtherLiabilitiesMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2021-12-310001551152us-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMember2022-12-310001551152us-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMember2021-12-310001551152us-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMember2022-12-310001551152us-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMemberus-gaap:InterestRateSwapMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:CashFlowHedgingMemberus-gaap:InterestRateSwapMember2022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:CashFlowHedgingMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMemberus-gaap:InterestRateSwapMember2022-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:PrepaidExpensesAndOtherCurrentAssetsMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:InterestRateSwapMember2022-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:AccountsPayableAndAccruedLiabilitiesMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:OtherNoncurrentAssetsMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2022-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:OtherNoncurrentAssetsMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:OtherLiabilitiesMemberus-gaap:InterestRateSwapMember2022-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:OtherLiabilitiesMemberus-gaap:InterestRateSwapMember2021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMember2022-01-012022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMember2021-01-012021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMember2020-01-012020-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2022-01-012022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2021-01-012021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2020-01-012020-12-310001551152us-gaap:CostOfSalesMember2022-12-310001551152us-gaap:InterestExpenseMember2022-12-310001551152us-gaap:SeniorNotesMemberus-gaap:DesignatedAsHedgingInstrumentMember2022-01-012022-12-310001551152us-gaap:SeniorNotesMemberus-gaap:DesignatedAsHedgingInstrumentMember2021-01-012021-12-310001551152us-gaap:SeniorNotesMemberus-gaap:DesignatedAsHedgingInstrumentMember2020-01-012020-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2021-01-012021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:ForeignExchangeForwardMember2020-01-012020-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2022-01-012022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2020-01-012020-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:ForeignExchangeForwardMemberus-gaap:NetInvestmentHedgingMember2021-01-012021-12-310001551152us-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMember2022-01-012022-12-310001551152us-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMember2020-01-012020-12-310001551152us-gaap:ForeignExchangeForwardMemberus-gaap:NondesignatedMember2021-01-012021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:TreasuryLockMemberus-gaap:CashFlowHedgingMember2021-01-012021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:TreasuryLockMemberus-gaap:CashFlowHedgingMember2022-01-012022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:TreasuryLockMemberus-gaap:CashFlowHedgingMember2020-01-012020-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:InterestRateSwapMember2021-01-012021-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:InterestRateSwapMember2022-01-012022-12-310001551152us-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:CashFlowHedgingMemberus-gaap:InterestRateSwapMember2020-01-012020-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2022-01-012022-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2021-01-012021-12-310001551152us-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMemberus-gaap:InterestRateSwapMember2020-01-012020-12-310001551152us-gaap:InterestExpenseMemberus-gaap:SeniorNotesMemberus-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMember2022-01-012022-12-310001551152us-gaap:InterestExpenseMemberus-gaap:SeniorNotesMemberus-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMember2021-01-012021-12-310001551152us-gaap:InterestExpenseMemberus-gaap:SeniorNotesMemberus-gaap:FairValueHedgingMemberus-gaap:DesignatedAsHedgingInstrumentMember2020-01-012020-12-310001551152us-gaap:FairValueMeasurementsRecurringMember2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2022-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2022-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2022-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2022-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:ForeignExchangeContractMember2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:ForeignExchangeContractMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:ForeignExchangeContractMember2022-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Memberus-gaap:ForeignExchangeContractMember2022-12-310001551152us-gaap:FairValueMeasurementsRecurringMember2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2021-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2021-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2021-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2021-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:ForeignExchangeContractMember2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:ForeignExchangeContractMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:ForeignExchangeContractMember2021-12-310001551152us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Memberus-gaap:ForeignExchangeContractMember2021-12-310001551152us-gaap:MeasurementInputDiscountRateMembersrt:MinimumMember2022-12-310001551152srt:MaximumMemberus-gaap:MeasurementInputDiscountRateMember2022-12-310001551152srt:WeightedAverageMemberus-gaap:MeasurementInputDiscountRateMember2022-12-310001551152us-gaap:MeasurementInputDiscountRateMembersrt:MinimumMember2021-12-310001551152srt:MaximumMemberus-gaap:MeasurementInputDiscountRateMember2021-12-310001551152srt:WeightedAverageMemberus-gaap:MeasurementInputDiscountRateMember2021-12-310001551152abbv:ProbabilityofPaymentforUnachievedMilestonesMembersrt:MinimumMember2022-12-310001551152abbv:ProbabilityofPaymentforUnachievedMilestonesMembersrt:MaximumMember2022-12-310001551152abbv:ProbabilityofPaymentforUnachievedMilestonesMembersrt:WeightedAverageMember2022-12-310001551152abbv:ProbabilityofPaymentforUnachievedMilestonesMembersrt:MinimumMember2021-12-310001551152abbv:ProbabilityofPaymentforUnachievedMilestonesMembersrt:MaximumMember2021-12-310001551152abbv:ProbabilityofPaymentforUnachievedMilestonesMembersrt:WeightedAverageMember2021-12-310001551152abbv:ProbabilityofPaymentforRoyaltiesbyIndicationMembersrt:MinimumMember2022-12-310001551152srt:MaximumMemberabbv:ProbabilityofPaymentforRoyaltiesbyIndicationMember2022-12-310001551152srt:WeightedAverageMemberabbv:ProbabilityofPaymentforRoyaltiesbyIndicationMember2022-12-310001551152abbv:ProbabilityofPaymentforRoyaltiesbyIndicationMembersrt:MinimumMember2021-12-310001551152srt:MaximumMemberabbv:ProbabilityofPaymentforRoyaltiesbyIndicationMember2021-12-310001551152srt:WeightedAverageMemberabbv:ProbabilityofPaymentforRoyaltiesbyIndicationMember2021-12-310001551152abbv:ProjectedYearofPaymentsMembersrt:MinimumMember2022-12-310001551152srt:MaximumMemberabbv:ProjectedYearofPaymentsMember2022-12-310001551152abbv:ProjectedYearofPaymentsMembersrt:WeightedAverageMember2022-12-310001551152abbv:ProjectedYearofPaymentsMembersrt:MinimumMember2021-12-310001551152srt:MaximumMemberabbv:ProjectedYearofPaymentsMember2021-12-310001551152abbv:ProjectedYearofPaymentsMembersrt:WeightedAverageMember2021-12-310001551152abbv:ProbabilityofPaymentforRoyaltiesExcludingApprovedIndicationsMember2022-12-310001551152abbv:ProbabilityofPaymentforRoyaltiesExcludingApprovedIndicationsMembersrt:MinimumMember2021-12-310001551152srt:MaximumMemberabbv:ProbabilityofPaymentforRoyaltiesExcludingApprovedIndicationsMember2021-12-310001551152us-gaap:CarryingReportedAmountFairValueDisclosureMember2022-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMember2022-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMemberus-gaap:FairValueInputsLevel1Member2022-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMemberus-gaap:FairValueInputsLevel2Member2022-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMemberus-gaap:FairValueInputsLevel3Member2022-12-310001551152us-gaap:CarryingReportedAmountFairValueDisclosureMember2021-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMember2021-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMemberus-gaap:FairValueInputsLevel1Member2021-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMemberus-gaap:FairValueInputsLevel2Member2021-12-310001551152us-gaap:EstimateOfFairValueFairValueDisclosureMemberus-gaap:FairValueInputsLevel3Member2021-12-310001551152us-gaap:SalesRevenueNetMember2022-01-012022-12-31abbv:wholesaler0001551152abbv:PrincipalUSCustomersMemberus-gaap:AccountsReceivableMemberus-gaap:GeographicConcentrationRiskMember2022-01-012022-12-310001551152abbv:PrincipalUSCustomersMemberus-gaap:AccountsReceivableMemberus-gaap:GeographicConcentrationRiskMember2021-01-012021-12-310001551152us-gaap:SalesRevenueNetMemberus-gaap:ProductConcentrationRiskMemberabbv:HUMIRAMember2022-01-012022-12-310001551152us-gaap:SalesRevenueNetMemberus-gaap:ProductConcentrationRiskMemberabbv:HUMIRAMember2021-01-012021-12-310001551152us-gaap:SalesRevenueNetMemberus-gaap:ProductConcentrationRiskMemberabbv:HUMIRAMember2020-01-012020-12-310001551152us-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:PensionPlansDefinedBenefitMember2020-12-310001551152us-gaap:OtherPostretirementBenefitPlansDefinedBenefitMember2021-12-310001551152us-gaap:OtherPostretirementBenefitPlansDefinedBenefitMember2020-12-310001551152us-gaap:PensionPlansDefinedBenefitMember2022-01-012022-12-310001551152us-gaap:PensionPlansDefinedBenefitMember2021-01-012021-12-310001551152us-gaap:OtherPostretirementBenefitPlansDefinedBenefitMember2022-01-012022-12-310001551152us-gaap:OtherPostretirementBenefitPlansDefinedBenefitMember2021-01-012021-12-310001551152us-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:OtherPostretirementBenefitPlansDefinedBenefitMember2022-12-310001551152us-gaap:ForeignPlanMember2022-12-310001551152us-gaap:ForeignPlanMember2021-12-310001551152us-gaap:OtherPostretirementBenefitPlansDefinedBenefitMember2020-01-012020-12-310001551152us-gaap:PensionPlansDefinedBenefitMember2020-01-012020-12-310001551152abbv:PreSixtyFiveYearsOfAgeMember2022-01-012022-12-310001551152abbv:PostSixtyFiveYearsOfAgeMember2022-01-012022-12-310001551152abbv:PreSixtyFiveYearsOfAgeMember2022-12-310001551152abbv:PostSixtyFiveYearsOfAgeMember2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMember2022-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMember2022-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMember2022-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMember2022-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMember2022-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMember2022-12-310001551152us-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:MutualFundMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:MutualFundMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:MutualFundMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:MutualFundMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2022-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueMeasuredAtNetAssetValuePerShareMember2022-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsLargeCapMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesUsMidCapMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMember2021-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:DefinedBenefitPlanEquitySecuritiesNonUsMember2021-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMember2021-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:USTreasuryAndGovernmentMember2021-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:CorporateDebtSecuritiesMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMember2021-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:ForeignGovernmentDebtSecuritiesMember2021-12-310001551152us-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:OtherDebtSecuritiesMemberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:MutualFundMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:MutualFundMemberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:MutualFundMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:MutualFundMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:RealEstateMemberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueInputsLevel12And3Member2021-12-310001551152us-gaap:FairValueInputsLevel1Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:FairValueInputsLevel2Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:FairValueInputsLevel3Memberus-gaap:PensionPlansDefinedBenefitMember2021-12-310001551152us-gaap:PensionPlansDefinedBenefitMemberus-gaap:FairValueMeasuredAtNetAssetValuePerShareMember2021-12-310001551152us-gaap:DefinedBenefitPlanEquitySecuritiesMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152us-gaap:DefinedBenefitPlanDebtSecurityMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152abbv:DefinedBenefitPlanOtherAssetsMemberus-gaap:PensionPlansDefinedBenefitMember2022-12-310001551152abbv:AbbVieSavingsPlanMember2022-01-012022-12-310001551152abbv:AbbVieSavingsPlanMember2021-01-012021-12-310001551152abbv:AbbVieSavingsPlanMember2020-01-012020-12-310001551152us-gaap:CostOfSalesMember2022-01-012022-12-310001551152us-gaap:CostOfSalesMember2021-01-012021-12-310001551152us-gaap:CostOfSalesMember2020-01-012020-12-310001551152us-gaap:ResearchAndDevelopmentExpenseMember2022-01-012022-12-310001551152us-gaap:ResearchAndDevelopmentExpenseMember2021-01-012021-12-310001551152us-gaap:ResearchAndDevelopmentExpenseMember2020-01-012020-12-310001551152us-gaap:SellingGeneralAndAdministrativeExpensesMember2022-01-012022-12-310001551152us-gaap:SellingGeneralAndAdministrativeExpensesMember2021-01-012021-12-310001551152us-gaap:SellingGeneralAndAdministrativeExpensesMember2020-01-012020-12-310001551152us-gaap:EmployeeStockOptionMember2022-01-012022-12-310001551152us-gaap:EmployeeStockOptionMember2021-01-012021-12-310001551152us-gaap:EmployeeStockOptionMember2020-01-012020-12-310001551152us-gaap:EmployeeStockOptionMember2021-12-310001551152us-gaap:EmployeeStockOptionMember2022-12-310001551152us-gaap:RestrictedStockUnitsRSUMembersrt:MinimumMember2022-01-012022-12-310001551152srt:MaximumMemberus-gaap:RestrictedStockUnitsRSUMember2022-01-012022-12-310001551152abbv:PerformancebasedRestrictedStockUnitsMember2022-01-012022-12-310001551152us-gaap:PerformanceSharesMember2022-01-012022-12-310001551152abbv:RestrictedStockUnitsAndPerformanceSharesMember2021-12-310001551152abbv:RestrictedStockUnitsAndPerformanceSharesMember2022-01-012022-12-310001551152abbv:RestrictedStockUnitsAndPerformanceSharesMember2022-12-310001551152abbv:RestrictedStockUnitsAndPerformanceSharesMember2021-01-012021-12-310001551152abbv:RestrictedStockUnitsAndPerformanceSharesMember2020-01-012020-12-3100015511522022-10-282022-10-2800015511522021-10-292021-10-2900015511522020-10-302020-10-3000015511522022-09-092022-09-0900015511522021-09-102021-09-1000015511522020-09-112020-09-1100015511522022-06-232022-06-2300015511522021-06-172021-06-1700015511522020-06-172020-06-1700015511522022-02-172022-02-1700015511522021-02-182021-02-1800015511522020-02-202020-02-200001551152abbv:December2018StockRepurchaseAuthorizationMember2022-01-012022-12-310001551152abbv:December2018StockRepurchaseAuthorizationMember2021-01-012021-12-310001551152abbv:December2018StockRepurchaseAuthorizationMember2020-01-012020-12-310001551152abbv:December2018StockRepurchaseAuthorizationMember2022-12-310001551152abbv:December2018StockRepurchaseAuthorizationMemberus-gaap:SubsequentEventMember2023-02-160001551152us-gaap:AccumulatedForeignCurrencyAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2019-12-310001551152abbv:AccumulatedNetGainLossFromNetInvestmentHedgesMember2019-12-310001551152us-gaap:AccumulatedDefinedBenefitPlansAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2019-12-310001551152us-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2019-12-310001551152us-gaap:AociIncludingPortionAttributableToNoncontrollingInterestMember2019-12-310001551152us-gaap:AccumulatedForeignCurrencyAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2020-01-012020-12-310001551152abbv:AccumulatedNetGainLossFromNetInvestmentHedgesMember2020-01-012020-12-310001551152us-gaap:AccumulatedDefinedBenefitPlansAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2020-01-012020-12-310001551152us-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2020-01-012020-12-310001551152us-gaap:AociIncludingPortionAttributableToNoncontrollingInterestMember2020-01-012020-12-310001551152us-gaap:AccumulatedForeignCurrencyAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2020-12-310001551152abbv:AccumulatedNetGainLossFromNetInvestmentHedgesMember2020-12-310001551152us-gaap:AccumulatedDefinedBenefitPlansAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2020-12-310001551152us-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2020-12-310001551152us-gaap:AociIncludingPortionAttributableToNoncontrollingInterestMember2020-12-310001551152us-gaap:AccumulatedForeignCurrencyAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2021-01-012021-12-310001551152abbv:AccumulatedNetGainLossFromNetInvestmentHedgesMember2021-01-012021-12-310001551152us-gaap:AccumulatedDefinedBenefitPlansAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2021-01-012021-12-310001551152us-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2021-01-012021-12-310001551152us-gaap:AociIncludingPortionAttributableToNoncontrollingInterestMember2021-01-012021-12-310001551152us-gaap:AccumulatedForeignCurrencyAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2021-12-310001551152abbv:AccumulatedNetGainLossFromNetInvestmentHedgesMember2021-12-310001551152us-gaap:AccumulatedDefinedBenefitPlansAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2021-12-310001551152us-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2021-12-310001551152us-gaap:AociIncludingPortionAttributableToNoncontrollingInterestMember2021-12-310001551152us-gaap:AccumulatedForeignCurrencyAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2022-01-012022-12-310001551152abbv:AccumulatedNetGainLossFromNetInvestmentHedgesMember2022-01-012022-12-310001551152us-gaap:AccumulatedDefinedBenefitPlansAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2022-01-012022-12-310001551152us-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2022-01-012022-12-310001551152us-gaap:AociIncludingPortionAttributableToNoncontrollingInterestMember2022-01-012022-12-310001551152us-gaap:AccumulatedForeignCurrencyAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2022-12-310001551152abbv:AccumulatedNetGainLossFromNetInvestmentHedgesMember2022-12-310001551152us-gaap:AccumulatedDefinedBenefitPlansAdjustmentIncludingPortionAttributableToNoncontrollingInterestMember2022-12-310001551152us-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2022-12-310001551152us-gaap:AociIncludingPortionAttributableToNoncontrollingInterestMember2022-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:NetInvestmentHedgingMemberus-gaap:AociDerivativeQualifyingAsHedgeExcludedComponentParentMember2022-01-012022-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:NetInvestmentHedgingMemberus-gaap:AociDerivativeQualifyingAsHedgeExcludedComponentParentMember2021-01-012021-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:NetInvestmentHedgingMemberus-gaap:AociDerivativeQualifyingAsHedgeExcludedComponentParentMember2020-01-012020-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:ForeignExchangeForwardMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2022-01-012022-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:ForeignExchangeForwardMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2021-01-012021-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:ForeignExchangeForwardMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2020-01-012020-12-310001551152us-gaap:TreasuryLockMemberus-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2022-01-012022-12-310001551152us-gaap:TreasuryLockMemberus-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2021-01-012021-12-310001551152us-gaap:TreasuryLockMemberus-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2020-01-012020-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMemberus-gaap:InterestRateSwapMember2022-01-012022-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMemberus-gaap:InterestRateSwapMember2021-01-012021-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMemberus-gaap:InterestRateSwapMember2020-01-012020-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2022-01-012022-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2021-01-012021-12-310001551152us-gaap:ReclassificationOutOfAccumulatedOtherComprehensiveIncomeMemberus-gaap:AccumulatedGainLossCashFlowHedgeIncludingNoncontrollingInterestMember2020-01-012020-12-310001551152abbv:AllerganplcMember2022-01-012022-06-300001551152abbv:DomesticTaxAuthorityAndStateAndLocalJurisdictionMember2022-12-310001551152abbv:DomesticTaxAuthorityStateAndLocalJurisdictionAndForeignTaxAuthorityMember2022-01-012022-12-310001551152abbv:DomesticAndForeignTaxAuthorityMember2022-12-310001551152abbv:NiaspanMember2022-01-012022-12-31abbv:lawsuitabbv:class_actionabbv:direct_purchaserabbv:end_payor_purchaser0001551152abbv:AndroGelAntitrustLitigationKingDrugCoOfFlorenceIncEtAlVAbbVieIncEtAlMember2019-08-012019-08-31abbv:company0001551152abbv:BystolicAntitrustLitigationMember2022-01-012022-12-310001551152abbv:PrescriptionDrugAbuseLitigationMember2022-12-31abbv:claim0001551152abbv:PrescriptionDrugAbuseLitigationInStateCourtsMember2022-12-310001551152us-gaap:SellingGeneralAndAdministrativeExpensesMemberabbv:PrescriptionDrugAbuseLitigationInStateCourtsMember2022-06-300001551152abbv:ElliottAssociatesL.P.Member2016-06-012016-06-30abbv:investment_fund0001551152country:USabbv:ImmunologyMemberabbv:HUMIRAMember2022-01-012022-12-310001551152country:USabbv:ImmunologyMemberabbv:HUMIRAMember2021-01-012021-12-310001551152country:USabbv:ImmunologyMemberabbv:HUMIRAMember2020-01-012020-12-310001551152abbv:ImmunologyMemberabbv:HUMIRAMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:ImmunologyMemberabbv:HUMIRAMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:ImmunologyMemberabbv:HUMIRAMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:ImmunologyMemberabbv:HUMIRAMember2022-01-012022-12-310001551152abbv:ImmunologyMemberabbv:HUMIRAMember2021-01-012021-12-310001551152abbv:ImmunologyMemberabbv:HUMIRAMember2020-01-012020-12-310001551152abbv:SKYRIZIMembercountry:USabbv:ImmunologyMember2022-01-012022-12-310001551152abbv:SKYRIZIMembercountry:USabbv:ImmunologyMember2021-01-012021-12-310001551152abbv:SKYRIZIMembercountry:USabbv:ImmunologyMember2020-01-012020-12-310001551152abbv:SKYRIZIMemberabbv:ImmunologyMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:SKYRIZIMemberabbv:ImmunologyMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:SKYRIZIMemberabbv:ImmunologyMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:SKYRIZIMemberabbv:ImmunologyMember2022-01-012022-12-310001551152abbv:SKYRIZIMemberabbv:ImmunologyMember2021-01-012021-12-310001551152abbv:SKYRIZIMemberabbv:ImmunologyMember2020-01-012020-12-310001551152abbv:RINVOQMembercountry:USabbv:ImmunologyMember2022-01-012022-12-310001551152abbv:RINVOQMembercountry:USabbv:ImmunologyMember2021-01-012021-12-310001551152abbv:RINVOQMembercountry:USabbv:ImmunologyMember2020-01-012020-12-310001551152abbv:RINVOQMemberabbv:ImmunologyMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:RINVOQMemberabbv:ImmunologyMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:RINVOQMemberabbv:ImmunologyMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:RINVOQMemberabbv:ImmunologyMember2022-01-012022-12-310001551152abbv:RINVOQMemberabbv:ImmunologyMember2021-01-012021-12-310001551152abbv:RINVOQMemberabbv:ImmunologyMember2020-01-012020-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMembercountry:US2022-01-012022-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMembercountry:US2021-01-012021-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMembercountry:US2020-01-012020-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMember2022-01-012022-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMember2021-01-012021-12-310001551152abbv:HematologicOncologyMemberabbv:ImbruvicaMember2020-01-012020-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMembercountry:US2022-01-012022-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMembercountry:US2021-01-012021-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMembercountry:US2020-01-012020-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMember2022-01-012022-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMember2021-01-012021-12-310001551152abbv:VENCLEXTAMemberabbv:HematologicOncologyMember2020-01-012020-12-310001551152abbv:AestheticsMembercountry:USabbv:BotoxCosmeticMember2022-01-012022-12-310001551152abbv:AestheticsMembercountry:USabbv:BotoxCosmeticMember2021-01-012021-12-310001551152abbv:AestheticsMembercountry:USabbv:BotoxCosmeticMember2020-01-012020-12-310001551152abbv:AestheticsMemberabbv:BotoxCosmeticMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:AestheticsMemberabbv:BotoxCosmeticMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:AestheticsMemberabbv:BotoxCosmeticMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:AestheticsMemberabbv:BotoxCosmeticMember2022-01-012022-12-310001551152abbv:AestheticsMemberabbv:BotoxCosmeticMember2021-01-012021-12-310001551152abbv:AestheticsMemberabbv:BotoxCosmeticMember2020-01-012020-12-310001551152abbv:AestheticsMembercountry:USabbv:JuvedermCollectionMember2022-01-012022-12-310001551152abbv:AestheticsMembercountry:USabbv:JuvedermCollectionMember2021-01-012021-12-310001551152abbv:AestheticsMembercountry:USabbv:JuvedermCollectionMember2020-01-012020-12-310001551152abbv:AestheticsMemberabbv:JuvedermCollectionMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:AestheticsMemberabbv:JuvedermCollectionMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:AestheticsMemberabbv:JuvedermCollectionMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:AestheticsMemberabbv:JuvedermCollectionMember2022-01-012022-12-310001551152abbv:AestheticsMemberabbv:JuvedermCollectionMember2021-01-012021-12-310001551152abbv:AestheticsMemberabbv:JuvedermCollectionMember2020-01-012020-12-310001551152abbv:AestheticsMembercountry:USabbv:OtherAestheticsMember2022-01-012022-12-310001551152abbv:AestheticsMembercountry:USabbv:OtherAestheticsMember2021-01-012021-12-310001551152abbv:AestheticsMembercountry:USabbv:OtherAestheticsMember2020-01-012020-12-310001551152abbv:AestheticsMemberabbv:OtherAestheticsMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:AestheticsMemberabbv:OtherAestheticsMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:AestheticsMemberabbv:OtherAestheticsMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:AestheticsMemberabbv:OtherAestheticsMember2022-01-012022-12-310001551152abbv:AestheticsMemberabbv:OtherAestheticsMember2021-01-012021-12-310001551152abbv:AestheticsMemberabbv:OtherAestheticsMember2020-01-012020-12-310001551152abbv:NeuroscienceMembercountry:USabbv:BotoxTherapeuticMember2022-01-012022-12-310001551152abbv:NeuroscienceMembercountry:USabbv:BotoxTherapeuticMember2021-01-012021-12-310001551152abbv:NeuroscienceMembercountry:USabbv:BotoxTherapeuticMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberus-gaap:NonUsMemberabbv:BotoxTherapeuticMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberus-gaap:NonUsMemberabbv:BotoxTherapeuticMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberus-gaap:NonUsMemberabbv:BotoxTherapeuticMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:BotoxTherapeuticMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:BotoxTherapeuticMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:BotoxTherapeuticMember2020-01-012020-12-310001551152abbv:NeuroscienceMembercountry:USabbv:VraylarMember2022-01-012022-12-310001551152abbv:NeuroscienceMembercountry:USabbv:VraylarMember2021-01-012021-12-310001551152abbv:NeuroscienceMembercountry:USabbv:VraylarMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:VraylarMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:VraylarMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:VraylarMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:VraylarMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:VraylarMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:VraylarMember2020-01-012020-12-310001551152abbv:NeuroscienceMembercountry:USabbv:DuodopaMember2022-01-012022-12-310001551152abbv:NeuroscienceMembercountry:USabbv:DuodopaMember2021-01-012021-12-310001551152abbv:NeuroscienceMembercountry:USabbv:DuodopaMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:DuodopaMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:DuodopaMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:DuodopaMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:DuodopaMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:DuodopaMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:DuodopaMember2020-01-012020-12-310001551152abbv:NeuroscienceMembercountry:USabbv:UbrelvyMember2022-01-012022-12-310001551152abbv:NeuroscienceMembercountry:USabbv:UbrelvyMember2021-01-012021-12-310001551152abbv:NeuroscienceMembercountry:USabbv:UbrelvyMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:QuliptaMembercountry:US2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:QuliptaMembercountry:US2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:QuliptaMembercountry:US2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMembercountry:US2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMembercountry:US2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMembercountry:US2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMember2022-01-012022-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMember2021-01-012021-12-310001551152abbv:NeuroscienceMemberabbv:OtherNeuroscienceMember2020-01-012020-12-310001551152abbv:EyeCareMembercountry:USabbv:LumiganGanfortMember2022-01-012022-12-310001551152abbv:EyeCareMembercountry:USabbv:LumiganGanfortMember2021-01-012021-12-310001551152abbv:EyeCareMembercountry:USabbv:LumiganGanfortMember2020-01-012020-12-310001551152abbv:EyeCareMemberus-gaap:NonUsMemberabbv:LumiganGanfortMember2022-01-012022-12-310001551152abbv:EyeCareMemberus-gaap:NonUsMemberabbv:LumiganGanfortMember2021-01-012021-12-310001551152abbv:EyeCareMemberus-gaap:NonUsMemberabbv:LumiganGanfortMember2020-01-012020-12-310001551152abbv:EyeCareMemberabbv:LumiganGanfortMember2022-01-012022-12-310001551152abbv:EyeCareMemberabbv:LumiganGanfortMember2021-01-012021-12-310001551152abbv:EyeCareMemberabbv:LumiganGanfortMember2020-01-012020-12-310001551152abbv:EyeCareMembercountry:USabbv:AlphaganCombiganMember2022-01-012022-12-310001551152abbv:EyeCareMembercountry:USabbv:AlphaganCombiganMember2021-01-012021-12-310001551152abbv:EyeCareMembercountry:USabbv:AlphaganCombiganMember2020-01-012020-12-310001551152abbv:EyeCareMemberus-gaap:NonUsMemberabbv:AlphaganCombiganMember2022-01-012022-12-310001551152abbv:EyeCareMemberus-gaap:NonUsMemberabbv:AlphaganCombiganMember2021-01-012021-12-310001551152abbv:EyeCareMemberus-gaap:NonUsMemberabbv:AlphaganCombiganMember2020-01-012020-12-310001551152abbv:EyeCareMemberabbv:AlphaganCombiganMember2022-01-012022-12-310001551152abbv:EyeCareMemberabbv:AlphaganCombiganMember2021-01-012021-12-310001551152abbv:EyeCareMemberabbv:AlphaganCombiganMember2020-01-012020-12-310001551152abbv:EyeCareMemberabbv:RestasisMembercountry:US2022-01-012022-12-310001551152abbv:EyeCareMemberabbv:RestasisMembercountry:US2021-01-012021-12-310001551152abbv:EyeCareMemberabbv:RestasisMembercountry:US2020-01-012020-12-310001551152abbv:EyeCareMemberabbv:RestasisMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:EyeCareMemberabbv:RestasisMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:EyeCareMemberabbv:RestasisMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:EyeCareMemberabbv:RestasisMember2022-01-012022-12-310001551152abbv:EyeCareMemberabbv:RestasisMember2021-01-012021-12-310001551152abbv:EyeCareMemberabbv:RestasisMember2020-01-012020-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMembercountry:US2022-01-012022-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMembercountry:US2021-01-012021-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMembercountry:US2020-01-012020-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMember2022-01-012022-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMember2021-01-012021-12-310001551152abbv:OtherEyeCareMemberabbv:EyeCareMember2020-01-012020-12-310001551152abbv:OtherKeyProductsMembercountry:USabbv:MAVYRETMember2022-01-012022-12-310001551152abbv:OtherKeyProductsMembercountry:USabbv:MAVYRETMember2021-01-012021-12-310001551152abbv:OtherKeyProductsMembercountry:USabbv:MAVYRETMember2020-01-012020-12-310001551152abbv:OtherKeyProductsMemberabbv:MAVYRETMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:OtherKeyProductsMemberabbv:MAVYRETMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:OtherKeyProductsMemberabbv:MAVYRETMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:OtherKeyProductsMemberabbv:MAVYRETMember2022-01-012022-12-310001551152abbv:OtherKeyProductsMemberabbv:MAVYRETMember2021-01-012021-12-310001551152abbv:OtherKeyProductsMemberabbv:MAVYRETMember2020-01-012020-12-310001551152abbv:CreonMemberabbv:OtherKeyProductsMembercountry:US2022-01-012022-12-310001551152abbv:CreonMemberabbv:OtherKeyProductsMembercountry:US2021-01-012021-12-310001551152abbv:CreonMemberabbv:OtherKeyProductsMembercountry:US2020-01-012020-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMembercountry:US2022-01-012022-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMembercountry:US2021-01-012021-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMembercountry:US2020-01-012020-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMemberus-gaap:NonUsMember2022-01-012022-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMemberus-gaap:NonUsMember2021-01-012021-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMemberus-gaap:NonUsMember2020-01-012020-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMember2022-01-012022-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMember2021-01-012021-12-310001551152abbv:LinzessConstellaMemberabbv:OtherKeyProductsMember2020-01-012020-12-310001551152abbv:OtherProductsMember2022-01-012022-12-310001551152abbv:OtherProductsMember2021-01-012021-12-310001551152abbv:OtherProductsMember2020-01-012020-12-310001551152country:US2022-01-012022-12-310001551152country:US2021-01-012021-12-310001551152country:US2020-01-012020-12-310001551152country:DE2022-01-012022-12-310001551152country:DE2021-01-012021-12-310001551152country:DE2020-01-012020-12-310001551152country:CA2022-01-012022-12-310001551152country:CA2021-01-012021-12-310001551152country:CA2020-01-012020-12-310001551152country:JP2022-01-012022-12-310001551152country:JP2021-01-012021-12-310001551152country:JP2020-01-012020-12-310001551152country:CN2022-01-012022-12-310001551152country:CN2021-01-012021-12-310001551152country:CN2020-01-012020-12-310001551152country:FR2022-01-012022-12-310001551152country:FR2021-01-012021-12-310001551152country:FR2020-01-012020-12-310001551152country:AU2022-01-012022-12-310001551152country:AU2021-01-012021-12-310001551152country:AU2020-01-012020-12-310001551152country:ES2022-01-012022-12-310001551152country:ES2021-01-012021-12-310001551152country:ES2020-01-012020-12-310001551152country:GB2022-01-012022-12-310001551152country:GB2021-01-012021-12-310001551152country:GB2020-01-012020-12-310001551152country:IT2022-01-012022-12-310001551152country:IT2021-01-012021-12-310001551152country:IT2020-01-012020-12-310001551152country:BR2022-01-012022-12-310001551152country:BR2021-01-012021-12-310001551152country:BR2020-01-012020-12-310001551152abbv:OtherCountriesMember2022-01-012022-12-310001551152abbv:OtherCountriesMember2021-01-012021-12-310001551152abbv:OtherCountriesMember2020-01-012020-12-310001551152abbv:UnitedStatesAndPuertoRicoMember2022-12-310001551152abbv:UnitedStatesAndPuertoRicoMember2021-12-310001551152srt:EuropeMember2022-12-310001551152srt:EuropeMember2021-12-310001551152abbv:AllOtherCountriesMember2022-12-310001551152abbv:AllOtherCountriesMember2021-12-31abbv:segment00015511522022-10-012022-12-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D. C. 20549
FORM 10-K | | | | | | | | |
| (MARK ONE) | | |
☒ | | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2022
OR | | | | | | | | |
☐ | | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number 001-35565
AbbVie Inc.
(Exact name of registrant as specified in its charter) | | | | | | | | |
| Delaware | | 32-0375147 |
(State or other jurisdiction of
incorporation or organization) | | (I.R.S. employer
identification number) |
1 North Waukegan Road
North Chicago, Illinois 60064-6400
(847) 932-7900
(Address, including zip code, and telephone number of principal executive offices)
Securities Registered Pursuant to Section 12(b) of the Act: | | | | | | | | | | | | | | |
| Title of Each Class | | Trading Symbol(s) | | Name of Each Exchange on Which Registered |
| Common Stock, par value $0.01 per share | | ABBV | | New York Stock Exchange |
| | | | Chicago Stock Exchange |
| 1.500% Senior Notes due 2023 | | ABBV23B | | New York Stock Exchange |
| 1.375% Senior Notes due 2024 | | ABBV24 | | New York Stock Exchange |
| 1.250% Senior Notes due 2024 | | ABBV24B | | New York Stock Exchange |
| 0.750% Senior Notes due 2027 | | ABBV27 | | New York Stock Exchange |
| 2.125% Senior Notes due 2028 | | ABBV28 | | New York Stock Exchange |
| 2.625% Senior Notes due 2028 | | ABBV28B | | New York Stock Exchange |
| 2.125% Senior Notes due 2029 | | ABBV29 | | New York Stock Exchange |
| 1.250% Senior Notes due 2031 | | ABBV31 | | New York Stock Exchange |
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes ☒ No ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act.
Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports) and (2) has been subject to such filing requirements for the past 90 days.
Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of "large accelerated filer," "accelerated filer" and "smaller reporting company" in Rule 12b-2 of the Exchange Act. | | | | | | | | | | | |
| Large Accelerated Filer | ☒ | Accelerated Filer | ☐ |
| Non-Accelerated Filer | ☐ | Smaller reporting company | ☐ |
| | Emerging growth company | ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☒
If securities are registered pursuant to Section 12(b) of the Act, indicate by checkmark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act).
Yes ☐ No ☒
The aggregate market value of the 1,751,453,358 shares of voting stock held by non-affiliates of the registrant, computed by reference to the closing price as reported on the New York Stock Exchange, as of the last business day of AbbVie Inc.'s most recently completed second fiscal quarter (June 30, 2022), was $268,252,598,311. AbbVie has no non-voting common equity.
Number of common shares outstanding as of January 31, 2023: 1,769,399,971
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the 2023 AbbVie Inc. Proxy Statement are incorporated by reference into Part III. The Definitive Proxy Statement will be filed on or about March 20, 2023.
ABBVIE INC.
FORM 10-K
FOR THE YEAR ENDED DECEMBER 31, 2022
TABLE OF CONTENTS
PART I
Overview
AbbVie or "the company" refer to AbbVie Inc., or AbbVie Inc. and its consolidated subsidiaries, as the context requires. AbbVie is a global, diversified research-based biopharmaceutical company positioned for success with a comprehensive product portfolio that has leadership positions across immunology, oncology, aesthetics, neuroscience and eye care. AbbVie uses its expertise, dedicated people and unique approach to innovation to develop and market advanced therapies that address some of the world’s most complex and serious diseases. AbbVie was incorporated in Delaware on April 10, 2012. On January 1, 2013, AbbVie became an independent, publicly-traded company as a result of the distribution by Abbott Laboratories (Abbott) of 100% of the outstanding common stock of AbbVie to Abbott's shareholders.
Segments
AbbVie operates as a single global business segment dedicated to the research and development, manufacturing, commercialization and sale of innovative medicines and therapies. This operating structure enables the Chief Executive Officer, as chief operating decision maker (CODM), to allocate resources and assess business performance on a global basis in order to achieve established long-term strategic goals. Consistent with this structure, a global research and development and supply chain organization is responsible for the discovery, development, manufacturing and supply of products. Commercial efforts that coordinate the marketing, sales and distribution of these products are organized by geographic region or therapeutic area. All of these activities are supported by a global corporate administrative staff. The determination of a single business segment is consistent with the consolidated financial information regularly reviewed by the CODM for purposes of assessing performance, allocating resources and planning and forecasting future periods. See Note 16, "Segment and Geographic Area Information" to the Consolidated Financial Statements included under Item 8, "Financial Statements and Supplementary Data" and the sales information related to AbbVie's key products and geographies included under Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations."
Products
AbbVie's portfolio of products includes a broad line of therapies that address some of the world's most complex and serious diseases.
Immunology products. AbbVie maintains an extensive immunology portfolio across rheumatology, dermatology and gastroenterology. AbbVie's immunology products address unmet needs for patients with autoimmune diseases. These products are:
Humira. Humira (adalimumab) is a biologic therapy administered as a subcutaneous injection. It is approved to treat the following autoimmune diseases in the United States, Canada and Mexico (collectively, North America) and in the European Union:
| | | | | | | | |
| Condition | | Principal Markets |
| Rheumatoid arthritis (moderate to severe) | | North America, European Union |
| Psoriatic arthritis | | North America, European Union |
| Ankylosing spondylitis | | North America, European Union |
| Adult Crohn's disease (moderate to severe) | | North America, European Union |
| Plaque psoriasis (moderate to severe chronic) | | North America, European Union |
| Juvenile idiopathic arthritis (moderate to severe polyarticular) | | North America, European Union |
| Ulcerative colitis (moderate to severe) | | North America, European Union |
| Axial spondyloarthropathy | | European Union |
| Pediatric Crohn's disease (moderate to severe) | | North America, European Union |
| Hidradenitis suppurativa (moderate to severe) | | North America, European Union |
| Pediatric enthesitis-related arthritis | | European Union |
| Non-infectious intermediate, posterior and panuveitis | | North America, European Union |
| Pediatric ulcerative colitis (moderate to severe) | | U.S., Canada, European Union |
| Pediatric uveitis | | North America, European Union |
| | | | | | | | | | | |
1 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Humira is also approved in Japan for the treatment of intestinal Behçet's disease and pyoderma gangrenosum.
Humira is sold in numerous other markets worldwide, including Japan, China, Brazil and Australia, and accounted for approximately 37% of AbbVie's total net revenues in 2022.
Skyrizi. Skyrizi (risankizumab) is an interleukin-23 (IL-23) inhibitor that selectively blocks IL-23 by binding to its p19 subunit. It is a biologic therapy approved to treat the following autoimmune diseases in North America, the European Union and Japan: | | | | | | | | |
Condition | | Principal Markets |
Plaque psoriasis (moderate to severe) | | North America, European Union, Japan |
Psoriatic arthritis | | U.S., European Union |
Adult Crohn's disease (moderate to severe) | | U.S., Canada, European Union |
Skyrizi is also approved in Japan for the treatment of plaque psoriasis, psoriatic arthritis, erythrodermic psoriasis in patients who have an inadequate response to conventional therapies, and for induction and maintenance in moderately to severely active Crohn's disease.
Skyrizi is approved in multiple countries globally, including the United States, Canada and the European Union, for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy. In psoriatic disease (psoriasis or psoriatic arthritis) Skyrizi is administered as a quarterly subcutaneous injection following two induction doses. When administered for Crohn’s disease, Skyrizi is given in three induction doses via IV infusion, followed by subcutaneous injection via an on-body injector every eight weeks.
Rinvoq. Rinvoq (upadacitinib) is an oral, once-daily selective and reversible JAK inhibitor that is approved to treat the following inflammatory diseases in North America, the European Union and Japan:
| | | | | | | | |
| Condition | | Principal Markets |
| Rheumatoid arthritis (moderate to severe) | | North America, European Union, Japan |
| Psoriatic arthritis | | U.S., Canada, European Union, Japan |
| Ankylosing spondylitis | | U.S., European Union |
| Atopic dermatitis (moderate to severe) | | U.S., Canada, European Union, Japan |
| Axial spondyloarthropathy | | U.S., European Union |
| Ulcerative colitis | | U.S., European Union |
In the United States, Rinvoq is indicated for both the treatment of moderate to severe active rheumatoid arthritis, for active psoriatic arthritis, for moderately to severely active ulcerative colitis, for active ankylosing spondylitis and for active non-radiographic axial spondyloarthritis in adult patients who have an inadequate response or intolerance to one or more TNF blockers. It is also indicated for the treatment of moderate to severe atopic dermatitis in adults and children 12 years of age and older whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies are inadvisable.
In the European Union, Rinvoq is indicated for the treatment of moderate to severe rheumatoid arthritis in adults, for active psoriatic arthritis in adults who have an inadequate response or intolerance to disease-modifying anti-rheumatic medicines (DMARDs), and for active axial spondyloarthritis in adults. It is also indicated for the treatment of moderate to severe atopic dermatitis in adults and children 12 years of age and older, and for moderately to severely active ulcerative colitis in adults.
Oncology products. AbbVie’s oncology products target some of the most complex and difficult-to-treat cancers. These products are:
Imbruvica. Imbruvica (ibrutinib) is an oral, once-daily therapy that inhibits a protein called Bruton's tyrosine kinase. Imbruvica was one of the first medicines to receive a United States Food and Drug Administration (FDA) approval after being granted a Breakthrough Therapy Designation and is one of the few therapies to receive four separate designations. Imbruvica currently is approved for the treatment of adult patients with blood cancers such as chronic lymphocytic leukemia (CLL), as well as certain forms of non-Hodgkin lymphoma. Imbruvica is approved in adult and pediatric patients one year and older with chronic graft versus host disease after failure of one or more lines of systemic therapy.
Venclexta/Venclyxto. Venclexta (venetoclax) is a B-cell lymphoma 2 (BCL-2) inhibitor used to treat hematological malignancies. Venclexta is approved by the FDA for adults with CLL or small lymphocytic lymphoma. In addition, Venclexta
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 2 |
is approved in combination with azacitidine, or decitabine, or low-dose cytarabine to treat adults with newly-diagnosed acute myeloid leukemia who are 75 years of age or older or have other medical conditions that prevent the use of standard chemotherapy.
Aesthetics products. AbbVie’s Aesthetics portfolio consists of facial injectables, plastics and regenerative medicine, body contouring and skincare products, which hold market-leading positions in the U.S. and in key markets around the world. These products are:
Botox Cosmetic. Botox Cosmetic is an acetylcholine release inhibitor and a neuromuscular blocking agent indicated for treatment in three areas: temporary improvement in the appearance of moderate to severe glabellar lines (frown lines between the eyebrows), moderate to severe crow's feet and moderate to severe forehead lines in adults. Having received its initial FDA approval in 2002, Botox Cosmetic is now approved for use in all major markets around the world.
The Juvederm Collection of Fillers. The Juvederm Collection of Fillers is a portfolio of hyaluronic acid-based dermal fillers with a variety of approved indications in the U.S. and in other major markets around the world to augment or treat volume loss in the cheeks, chin, lips and lower face.
Other aesthetics. Other aesthetics products include, but are not limited to, Alloderm regenerative dermal tissue, Coolsculpting body contouring technology, Natrelle breast implants, the SkinMedica skincare line, Latisse eyelash solution and DiamondGlow dermabrasion technology.
Neuroscience products. AbbVie’s neuroscience products address some of the most difficult-to-treat neurologic diseases. These products are:
Botox Therapeutic. Botox Therapeutic (onabotulinumtoxinA injection) is an acetylcholine release inhibitor and a neuromuscular blocking agent that is injected into muscle tissue. In the United States, it is approved to treat numerous indications, including chronic migraine, overactive bladder in adults who have an inadequate response to an anticholinergic medication, and urinary incontinence due to detrusor overactivity associated with a neurologic condition in adults who have an inadequate response to an anticholinergic medication. In addition, Botox Therapeutic is approved to treat spasticity in patients two years of age and older, cervical dystonia in adults, as well as other conditions. Botox is marketed in other countries around the world and licenses will vary. Botox Therapeutic is marketed by GSK in Japan.
Vraylar. Vraylar (cariprazine) is a dopamine D3-preferring D3/D2 receptor partial agonist and a 5-HT1A receptor partial agonist. Vraylar is indicated for acute and maintenance treatment of schizophrenia in adults, acute treatment of manic or mixed episodes associated with bipolar disorder in adults, acute treatment of depressive episodes associated with bipolar I disorder in adults and as an adjunctive treatment in major depressive disorder.
Duopa and Duodopa (carbidopa and levodopa). AbbVie's levodopa-carbidopa intestinal gel for the treatment of advanced Parkinson's disease is marketed as Duopa in the United States and as Duodopa outside of the United States.
Ubrelvy. Ubrelvy (ubrogepant) is a calcitonin gene-related peptide receptor antagonist indicated for the acute treatment of migraine with or without aura in adults. Ubrelvy is commercialized in the United States, Israel, Saudi Arabia, United Arab Emirates and has been approved in Canada.
Qulipta. Qulipta (atogepant) is a calcitonin gene-related peptide receptor antagonist indicated for the preventive treatment of episodic migraine in adults. Qulipta is commercialized in the United States and has recently been approved for use in Canada.
Eye care products. AbbVie’s eye care products address unmet needs and new approaches to help preserve and protect patients’ vision. These products are:
Lumigan/Ganfort. Lumigan (bimatoprost ophthalmic solution) 0.01% is a once daily, topical prostaglandin analog indicated for the reduction of elevated intraocular pressure (IOP) in patients with open angle glaucoma (OAG) or ocular hypertension (OHT). Ganfort is a once daily topical fixed combination of bimatoprost 0.03% and timolol 0.5% for the reduction of IOP in adult patients with OAG or OHT. Lumigan is sold in the United States and numerous markets around the world, while Ganfort is approved in the European Union and some markets in South America, the Middle East and Asia.
Alphagan/Combigan. Alphagan (brimonidine tartrate ophthalmic solution) is an alpha-adrenergic receptor agonist indicated for the reduction of elevated IOP in patients with open-angle glaucoma or ocular hypertension. Combigan (brimonidine tartrate/timolol maleate ophthalmic solution) is approved for reducing elevated IOP in patients with glaucoma who require additional or adjunctive IOP-lowering therapy. Both Alphagan and Combigan are available for sale in the United States and numerous markets around the world.
| | | | | | | | | | | |
3 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Restasis. Restasis is a calcineurin inhibitor immunosuppressant indicated to increase tear production in patients whose tear production is presumed to be suppressed due to ocular inflammation associated with keratoconjunctivitis sicca. Restasis is approved in the United States and a number of other markets in South America, the Middle East and Asia.
Other eye care. Other eye care products include Ozurdex, Refresh/Optive, Xen and Durysta.
Other key products. AbbVie’s other key products include, among other things, treatments for patients with hepatitis C virus (HCV), metabolic and hormone products that target a number of conditions, including exocrine pancreatic insufficiency and hypothyroidism, as well as endocrinology products for the palliative treatment of advanced prostate cancer, treatment of endometriosis and central precocious puberty and for the preoperative treatment of patients with anemia caused by uterine fibroids. These products are:
Mavyret/Maviret. Mavyret (glecaprevir/pibrentasvir) is approved in the United States and European Union (Maviret) for the treatment of adult and pediatric patients (12 years and older or weighing at least 45 kilograms) with chronic HCV genotype 1-6 infection without cirrhosis and with compensated cirrhosis (Child-Pugh A). It is also indicated for the treatment of adult and pediatric patients (12 years and older or weighing at least 45 kilograms) with HCV genotype 1 infection, who previously have been treated with a regimen containing an HCV NS5A inhibitor or an NS3/4A protease inhibitor, but not both. It is an 8-week, pan-genotypic treatment for patients without cirrhosis and following the EXPEDITION-8 study, also in patients with compensated cirrhosis who are new to treatment.
Creon. Creon (pancrelipase) is a pancreatic enzyme therapy for exocrine pancreatic insufficiency, a condition that occurs in patients with cystic fibrosis, chronic pancreatitis and several other conditions.
Lupron. Lupron (leuprolide acetate), which is also marketed as Lucrin and Lupron Depot, is a product for the palliative treatment of advanced prostate cancer, treatment of endometriosis and central precocious puberty and for the preoperative treatment of patients with anemia caused by uterine fibroids. Lupron is approved for daily subcutaneous injection and one-month, three-month, four-month and six-month intramuscular injection.
Linzess/Constella. Linzess (linaclotide) is a once-daily guanylate cyclase-C agonist used in adults to treat irritable bowel syndrome with constipation (IBS‑C) and chronic idiopathic constipation. The product is marketed as Linzess in the United States and as Constella outside of the United States.
Synthroid. Synthroid (levothyroxine sodium tablets, USP) is used in the treatment of hypothyroidism.
AbbVie has the rights to sell Creon and Synthroid only in the United States.
Marketing, Sales and Distribution Capabilities
AbbVie utilizes a combination of dedicated commercial resources, regional commercial resources and distributorships to market, sell and distribute its products worldwide. AbbVie directs its primary marketing efforts toward securing the prescription, or recommendation, of its brand of products by physicians, external experts and other health care providers. Managed care providers (for example, health maintenance organizations and pharmacy benefit managers), hospitals and state and federal government agencies (for example, State Medicaid programs, the United States Department of Veterans Affairs and the United States Department of Defense) are also important customers. AbbVie also markets directly to consumers themselves, although in the United States many of the company's products must be sold pursuant to a prescription. Outside of the United States, AbbVie focuses its promotional and market access efforts on external experts, payers, physicians and health systems. AbbVie also provides patient support programs closely related to its products.
AbbVie's products are generally sold worldwide directly to wholesalers, distributors, government agencies, health care facilities, specialty pharmacies and independent retailers from AbbVie-owned distribution centers and public warehouses. Certain products (including aesthetic products and devices) are also sold directly to physicians and other licensed healthcare providers. Although AbbVie's business does not have significant seasonality, AbbVie's product revenues may be affected by end customer and retail buying patterns, fluctuations in wholesaler inventory levels and other factors.
In the United States, AbbVie distributes pharmaceutical products principally through independent wholesale distributors, with some sales directly to retailers, pharmacies, patients or other customers. In 2022, three wholesale distributors (McKesson Corporation, Cardinal Health, Inc. and AmerisourceBergen Corporation) accounted for substantially all of AbbVie's pharmaceutical product sales in the United States. No individual wholesaler accounted for greater than 39% of AbbVie's 2022 gross revenues in the United States. Outside the United States, AbbVie sells products primarily to wholesalers or through distributors, and depending on the market works through largely centralized national payers system to agree on reimbursement terms.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 4 |
Certain products are co-marketed or co-promoted with other companies. AbbVie has no single customer that, if the customer were lost, would have a material adverse effect on the company's business. No material portion of AbbVie's business is subject to renegotiation of profits or termination of contracts at the election of the government. Orders are generally filled on a current basis and order backlog is not material to AbbVie's business.
Competition
The markets for AbbVie's products are highly competitive. AbbVie competes with other research-based pharmaceuticals and biotechnology companies that discover, manufacture, market and sell proprietary pharmaceutical products, therapies and biologics. For example, AbbVie's immunology products compete with anti-TNF products, JAK inhibitors and other competitive products intended to treat a number of disease states, and AbbVie's oncology products compete with BTK inhibitors. In addition, in the past few years, a number of other companies have started to develop, have successfully developed and/or are currently marketing products that are being positioned as competitors to Botox. The search for technological innovations in pharmaceutical products is a significant aspect of competition. The introduction of new products by competitors and changes in medical practices and procedures can result in product obsolescence. Price is also a competitive factor. In addition, the substitution of generic and biosimilar pharmaceutical products for branded pharmaceutical products creates competitive pressures on AbbVie's products that do not have patent protection. New products or treatments brought to market by AbbVie’s competitors could cause revenues for AbbVie’s products to decrease due to price reductions and sales volume decreases.
Biosimilars. Competition for AbbVie’s biologic products is affected by the approval of follow-on biologics, also known as “biosimilars.” Biologics have added major therapeutic options for the treatment of many diseases, including some for which therapies were unavailable or inadequate. The cost of developing and producing biologic therapies is typically dramatically higher than for small molecule medications, and many biologic medications are used for ongoing treatment of chronic diseases, such as rheumatoid arthritis or inflammatory bowel disease, or for the treatment of previously untreatable cancer. Significant investments in biologics infrastructure and manufacturing are necessary to produce biologic products.
Humira is now facing direct biosimilar competition in Europe, the United States and other countries, and AbbVie will continue to face competitive pressure from these biologics and from orally administered products.
In the United States, the FDA regulates biologics under the Federal Food, Drug, and Cosmetic Act (the FFDCA), the Public Health Service Act (PHSA) and the regulations implementing these statutes. The enactment of federal health care reform legislation in March 2010 provided a pathway for approval of biosimilars under the PHSA, but the approval process for, and science behind, biosimilars is complex. Approval by the FDA is dependent upon many factors, including a showing that the biosimilar is "highly similar" to the original product and has no clinically meaningful differences from the original product in terms of safety, purity and potency. The types of data that could ordinarily be required in an application to show similarity may include analytical data, bioequivalence studies and studies to demonstrate chemical similarity, animal studies (including toxicity studies) and clinical studies.
Furthermore, the law provides that only a biosimilar product that is determined to be "interchangeable" will be considered by the FDA as substitutable for the original biologic product without the intervention of the health care provider who prescribed the original biologic product. To prove that a biosimilar product is interchangeable, the applicant must demonstrate that the product can be expected to produce the same clinical results as the original biologic product in any given patient, and if the product is administered more than once in a patient, that safety risks and potential for diminished efficacy of alternating or switching between the use of the interchangeable biosimilar biologic product and the original biologic product is no greater than the risk of using the original biologic product without switching. The law continues to be interpreted and implemented by the FDA. As a result, its full ultimate impact, implementation and meaning remains subject to uncertainty.
Intellectual Property Protection and Regulatory Exclusivity
Generally, upon approval, products may be entitled to certain kinds of exclusivity under applicable intellectual property and regulatory regimes. AbbVie’s intellectual property is materially valuable to the company, and AbbVie seeks patent protection, where available, in all significant markets and/or countries for each product in development. In the United States, the expiration date for patents is 20 years after the filing date. Given that patents relating to pharmaceutical products are often obtained early in the development process and given the amount of time needed to complete clinical trials and other development activities required for regulatory approval, the length of time between product launch and patent expiration is significantly less than 20 years. The Drug Price Competition and Patent Term Restoration Act of 1984 (commonly known as the Hatch-Waxman Act) permits a patent holder to seek a patent extension, commonly called a “patent term restoration,” for patents on products (or processes for making the product) regulated by the FFDCA. The length of the patent extension is roughly based on 50 percent of the period of time from the filing of an Investigational New Drug Application (NDA) for a
| | | | | | | | | | | |
5 |  | 2022 Form 10-K | 2022 Form 10-K | | |
compound to the submission of the NDA for such compound, plus 100 percent of the time period from NDA submission to regulatory approval. The extension, however, cannot exceed five years and the patent term remaining after regulatory approval cannot exceed 14 years. Biological products licensed under the PHSA are similarly eligible for terms of patent restoration.
Pharmaceutical products may be entitled to other forms of legal or regulatory exclusivity upon approval. The scope, length and requirements for each of these exclusivities vary both in the United States and in other jurisdictions. In the United States, if the FDA approves a conventional drug product that contains an active ingredient not previously approved, the product is typically entitled to five years of non-patent regulatory exclusivity. Specific conditions of use approved for individual products may also be entitled to three years of exclusivity if approval was based on the FDA’s reliance on new clinical studies essential to approval submitted by the NDA applicant. If the NDA applicant studies the product for use by children, the FDA may grant pediatric exclusivity, which extends by 180 days all existing exclusivities (patent and regulatory) related to the product. For products that are either used to treat conditions that afflict a relatively small population or for which there is not a reasonable expectation that the research and development costs will be recovered, the FDA may designate the pharmaceutical as an orphan drug and grant it seven years of exclusivity. Other types of regulatory exclusivity may also be available, such as Generating New Antibiotic Incentives Now (GAIN) exclusivity, which can provide new antibiotic or new antifungal drugs an additional five years of exclusivity to be added to certain exclusivities already provided for by law.
Applicable laws and regulations dictate the scope of any exclusivity to which a product or particular characteristics of a product is entitled upon approval in any particular country. In certain instances, regulatory exclusivity may offer protection where patent protection is no longer available or for a period of time in excess of patent protection. It is not possible to estimate for each product in development the total period and scope of exclusivity to which it may become entitled until regulatory approval is obtained or sometimes even later. However, given the length of time required to complete clinical development of a pharmaceutical product, the periods of exclusivity that might be achieved in any individual case would not generally be expected to exceed a minimum of three years and a maximum of 14 years. These estimates do not consider other factors, such as the difficulty of recreating the manufacturing process for a particular product or other proprietary knowledge that may delay the introduction of a generic or other follow-on product after the expiration of applicable patent and other regulatory exclusivity periods.
Biologics may be entitled to exclusivity under the Biologics Price Competition and Innovation Act, which was passed on March 23, 2010 as Title VII to the Patient Protection and Affordable Care Act. The law provides a pathway for approval of biosimilars following the expiration of 12 years of regulatory exclusivity for the innovator biologic and a potential additional 180 day-extension term for conducting pediatric studies. Biologics are also eligible for orphan drug exclusivity, as discussed above. The law also includes an extensive process for the innovator biologic and biosimilar manufacturer to litigate patent infringement, validity and enforceability. The European Union has also created a pathway for approval of biosimilars and has published guidelines for approval of certain biosimilar products. The more complex nature of biologics and biosimilar products has led to close regulatory scrutiny over follow-on biosimilar products, which can reduce the effect of biosimilars on sales of the innovator biologic as compared to the sales erosion caused by generic versions of small molecule pharmaceutical products.
AbbVie owns or has licensed rights to a substantial number of patents and patent applications. AbbVie licenses or owns a patent portfolio of thousands of patent families, each of which includes United States patent applications and/or issued patents and may also contain the non-United States counterparts to these patents and applications.
These patents and applications, including various patents that expire during the period 2023 to the early 2040s, in aggregate are believed to be of material importance in the operation of AbbVie’s business. However, AbbVie believes that no single patent, license, trademark (or related group of patents, licenses, or trademarks), except for those related to adalimumab (which is sold under the trademark Humira), are material in relation to the company’s business as a whole. AbbVie has entered into settlement and license agreements with several adalimumab biosimilar manufacturers. Under the agreements, the licenses in the United States begin in 2023 and the licenses in Europe began in 2018.
In addition, the following patents, licenses and trademarks are significant: those related to ibrutinib (which is sold under the trademark Imbruvica), those related to risankizumab (which is sold under the trademark Skyrizi) and those related to upadacitinib (which is sold under the trademark Rinvoq). The United States composition of matter patent covering ibrutinib is expected to expire in 2027, with pediatric regulatory exclusivity then extending until May 2028. However, no generic entry for any ibrutinib product is expected prior to March 30, 2032. The United States composition of matter patent covering risankizumab is expected to expire in 2033. And the United States composition of matter patent covering upadacitinib is expected to expire in 2033.
AbbVie may rely, in some circumstances, on trade secrets to protect its technology. AbbVie seeks to protect its technology and product candidates, in part, by confidentiality agreements with its employees, consultants, advisors,
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 6 |
contractors and collaborators. These agreements may be breached and AbbVie may not have adequate remedies for any breach. In addition, AbbVie’s trade secrets may otherwise become known or be independently discovered by competitors. To the extent that AbbVie’s employees, consultants, advisors, contractors and collaborators use intellectual property owned by others in their work for the company, disputes may arise as to the rights in related or resulting know-how and inventions.
Licensing, Acquisitions and Other Arrangements
In addition to its independent efforts to develop and market products, AbbVie enters into arrangements such as acquisitions, option-to-acquire agreements, licensing arrangements, option-to-license arrangements, strategic alliances, co-promotion arrangements, co-development and co-marketing agreements and joint ventures. The acquisitions and option-to-acquire agreements typically include, among other terms and conditions, non-refundable purchase price payments or option fees, option exercise payments, milestones or earn-outs and other customary terms and obligations. The licensing and other arrangements typically include, among other terms and conditions, non-refundable upfront license fees, option fees and option exercise payments, milestone payments and royalty and/or profit sharing obligations. See Note 5, "Licensing, Acquisitions and Other Arrangements—Other Licensing & Acquisitions Activity," to the Consolidated Financial Statements included under Item 8, "Financial Statements and Supplementary Data."
Third Party Agreements
AbbVie has agreements with third parties for process development, product distribution, analytical services and manufacturing of certain products. AbbVie procures certain products and services from a limited number of suppliers and, in some cases, a single supply source. In addition, AbbVie has agreements with third parties for active pharmaceutical ingredient and product manufacturing, formulation and development services, fill, finish and packaging services, transportation and distribution and logistics services for certain products. AbbVie does not believe that these manufacturing-related agreements are material because AbbVie's business is not substantially dependent on any individual agreement. In most cases, AbbVie maintains alternate supply relationships that it can utilize without undue disruption of its manufacturing processes if a third party fails to perform its contractual obligations. AbbVie seeks to maintain sufficient inventory of product to minimize the impact of any supply disruption.
AbbVie is also party to certain collaborations and other arrangements, as discussed in Note 5, "Licensing, Acquisitions and Other Arrangements—Other Licensing & Acquisitions Activity," to the Consolidated Financial Statements included under Item 8, "Financial Statements and Supplementary Data."
Sources and Availability of Raw Materials
AbbVie purchases, in the ordinary course of business, raw materials and supplies essential to its operations from numerous suppliers around the world. In addition, certain medical devices and components necessary for the manufacture of AbbVie products are provided by unaffiliated third party suppliers. Despite the disruption to the global supply chain caused by COVID-19, AbbVie has continued to supply patients with no material supply impact, except for the previously-disclosed supply issues impacting Lupron. Given the general increased global volatility due to the pandemic, AbbVie is monitoring and taking actions to mitigate potential supply shortages which may impact the fulfillment of product demand.
Research and Development Activities
AbbVie makes a significant investment in research and development and has numerous compounds (and complementary devices) in clinical development, including potential treatments for complex, life-threatening diseases. AbbVie's ability to discover and develop new compounds is enhanced by the company's use of integrated discovery and development project teams, which include chemists, biologists, physicians and pharmacologists who work on the same compounds as a team. AbbVie also partners with third parties, such as biotechnology companies, other pharmaceutical companies and academic institutions to identify and prioritize promising new treatments that complement and enhance AbbVie’s existing portfolio. AbbVie also supplements its research and development efforts with acquisitions.
The research and development process generally begins with discovery research which focuses on the identification of a molecule that has a desired effect against a given disease. If preclinical testing of an identified compound proves successful, the compound moves into clinical development which generally includes the following phases:
•Phase 1— involves the first human tests in a small number of healthy volunteers or patients to assess safety, tolerability and doses for later phases.
•Phase 2— tests different doses of the drug in a disease state in order to assess efficacy.
•Phase 3— tests a drug that demonstrates favorable results in the earlier phases in a significantly larger patient population to further demonstrate efficacy and safety in order to meet requirements to enable global approval.
| | | | | | | | | | | |
7 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Preclinical data and clinical trials from all of the development phases provide the data required to prepare and submit an NDA, a Biological License Application (BLA) or other submission for regulatory approval to the FDA or similar government agencies outside the United States. The specific requirements (e.g., scope of clinical trials) for obtaining regulatory approval vary across different countries and geographic regions.
The research and development process from discovery through a new drug launch typically takes 8 to 12 years and can be even longer. The research and development of new pharmaceutical products has a significant amount of inherent uncertainty. There is no guarantee when, or if, a molecule will receive the regulatory approval required to launch a new drug or indication.
In addition to the development of new products, delivery devices and new formulations, research and development projects also may include Phase 4 trials, sometimes called post-marketing studies. For such projects, clinical trials are designed and conducted to collect additional data regarding, among other parameters, the benefits and risks of an approved drug.
Regulation—Discovery and Clinical Development
United States. Securing approval to market a new pharmaceutical product in the United States requires substantial effort and financial resources and takes several years to complete. The applicant must complete preclinical tests and submit protocols to the FDA before commencing clinical trials. Clinical trials are intended to establish the safety and efficacy of the pharmaceutical product and typically are conducted in sequential phases, although the phases may overlap or be combined. If the required clinical testing is successful, the results are submitted to the FDA in the form of an NDA or BLA requesting approval to market the product for one or more indications. The FDA reviews an NDA or BLA to determine whether a product is safe and effective for its intended use and whether its manufacturing is compliant with current Good Manufacturing Practices (cGMP).
Compliance with regulatory requirements is assured through periodic, announced or unannounced inspections by the FDA and other regulatory authorities, and these inspections associated with clinical development may include the sponsor, investigator sites, laboratories, hospitals and manufacturing facilities of AbbVie's subcontractors or other third-party manufacturers. Failure to comply with applicable regulatory requirements can result in enforcement action by the FDA, including rejection of an NDA or BLA.
Even if an NDA or a BLA receives approval, the applicant must comply with post-approval requirements. For example, holders of an approval must report adverse reactions, provide updated safety and efficacy information and comply with requirements concerning advertising and promotional materials and activities. Also, quality control and manufacturing procedures must continue to conform to cGMP after approval, and certain changes to the manufacturing procedures and finished product must be submitted and approved by the FDA prior to implementation. The FDA periodically inspects manufacturing facilities to assess compliance with cGMP, which imposes extensive procedural and record keeping requirements. In addition, as a condition of approval, the FDA may require post-marketing testing and surveillance to further assess and monitor the product's safety or efficacy after commercialization, which may require additional clinical trials, patient registries, observational data or additional work on chemistry, manufacturing and controls. Any post-approval regulatory obligations, and the cost of complying with such obligations, could expand in the future. Further, the FDA continues to regulate product labeling, and prohibits the promotion of products for unapproved or “off-label” uses along with other labeling restrictions.
Outside the United States. AbbVie is subject to similar regulatory requirements outside the United States for approval and marketing of pharmaceutical products. AbbVie must obtain approval of a clinical trial application or product from applicable supervising regulatory authorities before it can commence clinical trials or marketing of the product in target markets. The approval requirements and process for each country can vary, and the time required to obtain approval may be longer or shorter than that required for FDA approval in the United States. For example, AbbVie may submit marketing authorizations in the European Union under either a centralized or decentralized procedure. The centralized procedure is mandatory for the approval of biotechnology products and many pharmaceutical products and provides for a single marketing authorization that is valid for all European Union member states. Under the centralized procedure, a single marketing authorization application is submitted to the European Medicines Agency. After the agency evaluates the application, it makes a recommendation to the European Commission, which then makes the final determination on whether to approve the application. The decentralized procedure provides for mutual recognition of individual national approval decisions and is available for products that are not subject to the centralized procedure.
In Japan, applications for approval of a new product are made through the Pharmaceutical and Medical Devices Agency (PMDA). Japan-specific trials and/or bridging studies to demonstrate that the non-Japanese clinical data applies to Japanese patients may be required. After completing a comprehensive review, the PMDA reports to the Ministry of Health, Labour and Welfare, which then approves or denies the application.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 8 |
Similarly, applications for a new product in China are submitted to the Center for Drug Evaluation of the National Medical Products Administration for technical review and approval of a product for marketing in China. Clinical data in Chinese subjects are usually required to support approval in China, requiring the inclusion of China in global pivotal studies, or a separate China/Asian clinical trial.
The regulatory process in many emerging markets continues to evolve. Many emerging markets, including those in Asia, generally require regulatory approval to have been obtained in a large developed market (such as the United States or Europe) before the country will begin or complete its regulatory review process. Similar to the requirements in Japan and China, certain countries (notably South Korea, Taiwan, India and Russia) also generally require that clinical studies that include data from patients in those countries be conducted in order to support local regulatory approval.
The requirements governing the conduct of clinical trials and product licensing also vary. In addition, post-approval regulatory obligations such as adverse event reporting and cGMP compliance generally apply and may vary by country. For example, after a marketing authorization has been granted in the European Union, periodic safety reports must be submitted and other pharmacovigilance measures may be required (such as Risk Management Plans).
Regulation—Commercialization, Distribution and Manufacturing
The manufacturing, marketing, sale, promotion and distribution of AbbVie's products are subject to comprehensive government regulation. Government regulation by various national, regional, federal, state and local agencies, both in the United States and other countries, addresses (among other matters) inspection of, and controls over, research and laboratory procedures, clinical investigations, product approvals and manufacturing, labeling, packaging, marketing and promotion, pricing and reimbursement, sampling, distribution, quality control, post-marketing surveillance, record keeping, storage and disposal practices. AbbVie's operations are also affected by trade regulations in many countries that limit the import of raw materials and finished products and by laws and regulations that seek to prevent corruption and bribery in the marketplace (including the United States Foreign Corrupt Practices Act and the United Kingdom Bribery Act, which provide guidance on corporate interactions with government officials) and require safeguards for the protection of personal data. In addition, AbbVie is subject to laws and regulations pertaining to health care fraud and abuse, including state and federal anti-kickback and false claims laws in the United States. Prescription drug manufacturers such as AbbVie are also subject to taxes, as well as application, product, user and other fees.
Compliance with these laws and regulations is costly and materially affects AbbVie's business. Among other effects, health care regulations substantially increase the time, difficulty and costs incurred in obtaining and maintaining approval to market newly developed and existing products. AbbVie expects compliance with these regulations to continue to require significant technical expertise and capital investment to ensure compliance. Failure to comply can delay the release of a new product or result in regulatory and enforcement actions, the seizure or recall of a product, the suspension or revocation of the authority necessary for a product's production and sale and other civil or criminal sanctions, including fines and penalties.
In addition to regulatory initiatives, AbbVie's business can be affected by ongoing studies of the utilization, safety, efficacy and outcomes of health care products and their components that are regularly conducted by industry participants, government agencies and others. These studies can lead to updates to the data regarding utilization, safety and efficacy of previously marketed products. In some cases, these studies have resulted, and may in the future result, in the discontinuance of, or limitations on, marketing of such products domestically or worldwide, and may give rise to claims for damages from persons who believe they have been injured as a result of their use.
Access to human health care products continues to be a subject of oversight, investigation and action by governmental agencies, legislative bodies and private organizations in the United States and other countries. A major focus is cost containment. Efforts to reduce health care costs are also being made in the private sector, notably by health care payers and providers, which have instituted various cost reduction and containment measures. AbbVie expects insurers and providers to continue attempts to reduce the cost of health care products. Outside the United States, many countries control the price of health care products directly or indirectly, through reimbursement, payment, pricing, coverage limitations, or compulsory licensing. Political and budgetary pressures in the United States and in other countries may also heighten the scope and severity of pricing pressures on AbbVie's products for the foreseeable future.
United States. Specifically, U.S. federal laws require pharmaceutical manufacturers to pay certain statutorily-prescribed rebates to state Medicaid programs on prescription drugs reimbursed under state Medicaid plans, and the efforts by states to seek additional rebates may affect AbbVie's business. Similarly, the Veterans Health Care Act of 1992, as a prerequisite to participation in Medicaid and other federal health care programs, requires that manufacturers extend additional discounts on pharmaceutical products to various federal agencies, including the United States Department of Veterans Affairs, Department of Defense and Public Health Service entities and institutions. In addition, recent legislative changes would require similarly discounted prices to be offered to TRICARE program beneficiaries. The Veterans Health Care
| | | | | | | | | | | |
9 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Act of 1992 also established the 340B drug discount program, which requires pharmaceutical manufacturers to provide products at reduced prices to various designated health care entities and facilities.
In the United States, most states also have generic substitution legislation requiring or permitting a dispensing pharmacist to substitute a different manufacturer's generic version of a pharmaceutical product for the one prescribed. In addition, the federal government follows a diagnosis-related group (DRG) payment system for certain institutional services provided under Medicare or Medicaid and has implemented a prospective payment system (PPS) for services delivered in hospital outpatient, nursing home and home health settings. DRG and PPS entitle a health care facility to a fixed reimbursement based on the diagnosis and/or procedure rather than actual costs incurred in patient treatment, thereby increasing the incentive for the facility to limit or control expenditures for many health care products. Medicare reimburses Part B drugs based on average sales price plus a certain percentage to account for physician administration costs, which have been reduced in the hospital outpatient setting. Medicare enters into contracts with private plans to negotiate prices for most patient-administered medicine delivered under Part D.
Under the Patient Protection and Affordable Care Act and the Health Care and Education Reconciliation Act (together, the Affordable Care Act), AbbVie pays a fee related to its pharmaceuticals sales to government programs. In addition, AbbVie provides a discount of 70% for branded prescription drugs sold to patients who fall into the Medicare Part D coverage gap, or "donut hole."
The Affordable Care Act also includes provisions known as the Physician Payments Sunshine Act, which require manufacturers of drugs and biologics covered under Medicare and Medicaid to record any transfers of value to physicians and teaching hospitals and to report this data to the Centers for Medicare and Medicaid Services for subsequent public disclosure. Similar reporting requirements have also been enacted on the state level in the United States, and an increasing number of countries worldwide either have adopted or are considering similar laws requiring disclosure of interactions with health care professionals. Failure to report appropriate data may result in civil or criminal fines and/or penalties.
European Union. The European Union has adopted directives and other legislation governing labeling, advertising, distribution, supply, pharmacovigilance and marketing of pharmaceutical products. Such legislation provides mandatory standards throughout the European Union and permits member states to supplement these standards with additional regulations. European governments also regulate pharmaceutical product prices through their control of national health care systems that fund a large part of the cost of such products to consumers. As a result, patients are unlikely to use a pharmaceutical product that is not reimbursed by the government. In many European countries, the government either regulates the pricing of a new product at launch or subsequent launch through direct price controls or reference pricing. In recent years, many countries have also imposed new or additional cost containment measures on pharmaceutical products. Differences between national pricing regimes create price differentials within the European Union that can lead to significant parallel trade in pharmaceutical products.
Most governments also promote generic substitution by mandating or permitting a pharmacist to substitute a different manufacturer's generic version of a pharmaceutical product for the one prescribed and by permitting or mandating that health care professionals prescribe generic versions in certain circumstances. Many governments are also following a similar path for biosimilar therapies. In addition, governments use reimbursement lists to limit the pharmaceutical products that are eligible for reimbursement by national health care systems.
Japan. In Japan, the National Health Insurance system maintains a Drug Price List specifying which pharmaceutical products are eligible for reimbursement, and the Ministry of Health, Labour and Welfare sets the prices of the products on this list. The government generally introduces price cut rounds every other year and also mandates price decreases for specific products. New products judged innovative or useful, that are indicated for pediatric use, or that target orphan or small population diseases, however, may be eligible for a pricing premium. The government has also promoted the use of generics, where available.
Emerging Markets. Many emerging markets take steps to reduce pharmaceutical product prices, in some cases through direct price controls and in others through the promotion of generic/biosimilar alternatives to branded pharmaceuticals.
Since AbbVie markets its products worldwide, certain products of a local nature and variations of product lines must also meet other local regulatory requirements. Certain additional risks are inherent in conducting business outside the United States, including price and currency exchange controls, changes in currency exchange rates, limitations on participation in local enterprises, expropriation, nationalization and other governmental action.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 10 |
Regulation – Medical Devices
Medical devices are subject to regulation by the FDA, state agencies and foreign government health authorities. FDA regulations, as well as various U.S. federal and state laws, govern the development, clinical testing, manufacturing, labeling, record keeping and marketing of medical device products agencies in the United States. AbbVie’s medical device product candidates, including AbbVie’s breast implants, must undergo rigorous clinical testing and an extensive government regulatory clearance or approval process prior to sale in the United States and other countries. The lengthy process of clinical development and submissions for clearance or approval, and the continuing need for compliance with applicable laws and regulations, require the expenditure of substantial resources. Regulatory clearance or approval, when and if obtained, may be limited in scope, and may significantly limit the indicated uses for which a product may be marketed. Cleared or approved products and their manufacturers are subject to ongoing review, and discovery of previously unknown problems with products may result in restrictions on their manufacture, sale and/or use or require their withdrawal from the market.
United States. AbbVie’s medical device products are subject to extensive regulation by the FDA in the United States. Unless an exemption applies, each medical device AbbVie markets in the United States must have a 510(k) clearance or a Premarket Approval Application (PMA) in accordance with the FFDCA and its implementing regulations. The FDA classifies medical devices into one of three classes, depending on the degree of risk associated with each medical device and the extent of controls that are needed to ensure safety and effectiveness. Devices deemed to pose a lower risk are placed in either Class I or Class II, and devices deemed by the FDA to pose the greatest risk, such as life-sustaining, life-supporting or implantable devices, or a device deemed to be not substantially equivalent to a previously cleared 510(k) device, are placed in Class III. In general, a Class III device cannot be marketed in the United States unless the FDA approves the device after submission of a PMA, and any changes to the device subsequent to initial FDA approval must also be reviewed and approved by the FDA. The majority of AbbVie’s medical device products, including AbbVie’s breast implants, are regulated as Class III medical devices. A Class III device may have significant additional obligations imposed in its conditions of approval, and the time in which it takes to obtain approval can be long. Compliance with regulatory requirements is assured through periodic, unannounced facility inspections by the FDA and other regulatory authorities, and these inspections may include the manufacturing facilities of AbbVie’s subcontractors or other third-party manufacturers. Failure to comply with applicable regulatory requirements can result in enforcement action by the FDA, which may include any of the following sanctions: warning letters or untitled letters; fines, injunctions and civil penalties; recall or seizure of AbbVie’ products; operating restrictions, partial suspension or total shutdown of production; refusing AbbVie’ request for 510(k) clearance or PMA approval of new products; withdrawing 510(k) clearance or PMA approvals that are already granted; and criminal prosecution.
A clinical trial is almost always required to support a PMA application and is sometimes required for a 510(k) premarket notification. Clinical trials generally require submission of an application for an investigational device exemption (IDE), which must be supported by appropriate data, such as animal and laboratory testing results, showing that it is safe to test the device in humans and that the testing protocol is scientifically sound. A study sponsor must obtain approval for its IDE from the FDA, and it must also obtain approval of its study from the Institutional Review Board overseeing the trial. The results of clinical testing may not be sufficient to obtain approval of the investigational device.
Once a device is approved, the manufacture and distribution of the device remains subject to continuing regulation by the FDA, including Quality System Regulation requirements, which involve design, testing, control, documentation and other quality assurance procedures during the manufacturing process. Medical device manufacturers and their subcontractors are required to register their establishments and list their manufactured devices with the FDA and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with regulatory requirements. Manufacturers must also report to the FDA if their devices may have caused or contributed to a death or serious injury or malfunctioned in a way that could likely cause or contribute to a death or serious injury, or if the manufacturer conducts a field correction or product recall or removal to reduce a risk to health posed by a device or to remedy a violation of the FFDCA that may present a health risk. Further, the FDA continues to regulate device labeling, and prohibits the promotion of products for unapproved or “off-label” uses along with other labeling restrictions.
European Union. Medical device products that are marketed in the European Union must comply with the requirements of the Medical Device Regulation (the MDR), which came into effect in May 2021. The MDR provides for regulatory oversight with respect to the design, manufacture, clinical trials, labeling and adverse event reporting for medical devices to ensure that medical devices marketed in the European Union are safe and effective for their intended uses. Medical devices that comply with the MDR are entitled to bear a Conformité Européenne marking evidencing such compliance and may be marketed in the European Union. Failure to comply with these domestic and international regulatory requirements could affect AbbVie’s ability to market and sell AbbVie’s products in these countries.
| | | | | | | | | | | |
11 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Environmental Matters
AbbVie believes that its operations comply in all material respects with applicable laws and regulations concerning environmental protection. Regulations under federal and state environmental laws impose stringent limitations on emissions and discharges to the environment from various manufacturing operations. AbbVie's capital expenditures for pollution control in 2022 were approximately $12 million and operating expenditures were approximately $35 million. In 2023, capital expenditures for pollution control are estimated to be approximately $9 million and operating expenditures are estimated to be approximately $37 million.
Abbott was identified as one of many potentially responsible parties in investigations and/or remediations at several locations in the United States, including Puerto Rico, under the Comprehensive Environmental Response, Compensation and Liability Act, commonly known as Superfund. Some of these locations were transferred to AbbVie in connection with the separation and distribution, and AbbVie has become a party to these investigations and remediations. Abbott was also engaged in remediation at several other sites, some of which have been transferred to AbbVie in connection with the separation and distribution, in cooperation with the Environmental Protection Agency or similar agencies. While it is not feasible to predict with certainty the final costs related to those investigations and remediation activities, AbbVie believes that such costs, together with other expenditures to maintain compliance with applicable laws and regulations concerning environmental protection, should not have a material adverse effect on the company's financial position, cash flows, or results of operations.
Employees
AbbVie employed approximately 50,000 employees in over 70 countries as of January 31, 2023. Outside the United States, some of AbbVie's employees are represented by unions or works councils. AbbVie believes that it has good relations with its employees.
Human Capital Management
Attracting, retaining and providing meaningful growth and development opportunities to AbbVie's employees is critical to the company's success in making a remarkable impact on people’s lives around the world. AbbVie leverages numerous resources to identify and enhance strategic and leadership capability, foster employee engagement and create a culture where diverse talent is productive and engaged. AbbVie invests in its employees through competitive compensation, benefits and employee support programs and offers best-in-class development and leadership opportunities. AbbVie has developed a deep talent base through ongoing investment in functional and leadership training and by sourcing world-class external talent, ensuring a sustainable talent pipeline. AbbVie continuously cultivates and enhances its working culture and embraces equality, diversity and inclusion as fundamental to the company's mission.
Attracting and Developing Talent. Attracting and developing high-performing talent is essential to AbbVie’s continued success. AbbVie implements detailed talent attraction strategies, with an emphasis on STEM skill sets, a diverse talent base and other critical skillsets, including drug discovery, clinical development, market access and business development. AbbVie has also invested in integrated inclusive practices across the end-to-end hiring process and introduced Inclusive Recruitment Live Trainings, as a requirement for all people leaders and recruiters. AbbVie also invests in competitive compensation and benefits programs. In addition to offering a comprehensive suite of benefits ranging from medical and dental coverage to retirement, disability and life insurance programs, AbbVie also provides health promotion programs, mental health awareness campaigns and employee assistance programs in several countries, financial wellness support, on-site health screenings and immunizations in several countries and on-site fitness and rehabilitation centers. In 2022, AbbVie launched on-site health care clinics at certain locations, offering convenient and affordable access to quality healthcare, flu shots and vaccines. In addition, the AbbVie Employee Assistance Fund (a part of the AbbVie Foundation) supports two programs for global employees: the AbbVie Possibilities Scholarship for children of employees, which is an annual merit-based scholarship for use at accredited colleges, universities or vocational-technical schools; and the Employee Relief Program, which is financial assistance to support short term needs of employees when faced with large-scale disasters (e.g. a hurricane), individual disasters (e.g. a home fire) or financial hardship (e.g. the death of a spouse). Finally, AbbVie empowers managers and their teams with tools, tips and guidelines on effectively managing workloads, managing teams from a distance and supporting flexible work practices. In 2022, we implemented "Where We Work", AbbVie's hybrid work model, offering eligible employees predictable flexibility.
New AbbVie employees are given a tailored onboarding experience for faster integration and to support performance. One of AbbVie's mentorship programs allows employees to self-nominate as mentors or mentees and facilitates meaningful relationships supporting employees’ career and development goals.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 12 |
AbbVie also provides structured, broad-based development opportunities, focusing on high-performance skills and leadership training. AbbVie's talent philosophy holds leaders accountable for building a high-performing organization, and the company provides development opportunities for all levels of leadership. AbbVie's Learn, Develop, Perform program offers year-long, self-directed leadership education, supplemented with tools and resources, and leverages leaders as role models and teachers. In addition, a foundational success factor to AbbVie's leadership pipeline is the company's Professional Development Programs, which attract graduates, postgraduates and post-doctoral talent to participate in formal development programs lasting up to three years, with the objective of strengthening functional and leadership capabilities.
Culture. AbbVie’s shared values of transforming lives, acting with integrity, driving innovation, embracing diversity and inclusion and serving the community form the core of the company's culture. AbbVie articulates the behaviors associated with these values in the Ways We Work, a core set of working behaviors that emphasize how the company achieves results is equally as important as achieving them. The Ways We Work are designed to ensure that every AbbVie employee is aware of the company's cultural expectations. AbbVie integrates the Ways We Work into all talent processes, forming the basis for assessing performance, prioritizing development and ultimately rewarding employees. AbbVie believes its culture creates strong engagement, which is measured regularly through a confidential, third-party all-employee survey, and this engagement supports AbbVie’s mission of making a remarkable impact on people’s lives.
Equity, Equality, Diversity & Inclusion (EED&I). A cornerstone of AbbVie’s human capital management approach is to prioritize fostering an inclusive and diverse workforce. Recently, AbbVie adopted a five-year Equality, Diversity & Inclusion roadmap that defines key global focus areas, objectives and associated initiatives, and includes implementation plans organized by business function and geography. AbbVie’s senior leaders have adopted formal goals aligned with executing this strategy. In recent years, AbbVie's board of directors has prioritized oversight of AbbVie's response to the U.S. racial justice movement, including overseeing internal programs designed to ensure that AbbVie is attracting, retaining and developing diverse talent. Through December 2022, women represented 52 percent of management positions globally and in the United States, 37 percent of AbbVie's workforce was comprised of members of historically underrepresented populations, an increase from 2021. Further, AbbVie is committed to pay equity and conducts pay equity analyses annually. A critical component of AbbVie's strategy is to instill an inclusive mindset in all AbbVie leaders and employees, so the company continues to realize the full value of its diverse workforce from recruitment through retirement. AbbVie's Employee Resource Groups also help the company nurture an inclusive culture by building community and creating connections and opportunities for mentoring, professional development, talent attraction and networking. In 2021, AbbVie reiterated its commitment to racial equality and social justice by, among other things, expanding its employee matching program to $3-to-$1 for donations to civil rights nonprofits fostering racial equity and by reaffirming its commitment to clinical trial diversity. Additional information about AbbVie's efforts on racial equality and social justice is provided on the company's website at: https://abbvie.com/our-company/equality-inclusion-diversity/our-commitment-to-racial-justice.html.
Internet Information
Copies of AbbVie's Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934 are available free of charge through AbbVie's investor relations website (investors.abbvie.com) as soon as reasonably practicable after AbbVie electronically files the material with, or furnishes it to, the Securities and Exchange Commission (SEC).
AbbVie's corporate governance guidelines, outline of directorship qualifications, code of business conduct and the charters of AbbVie's audit committee, compensation committee, nominations and governance committee and public policy committee are all available on AbbVie's investor relations website (investors.abbvie.com).
You should carefully consider the following risks and other information in this Form 10-K in evaluating AbbVie and AbbVie's common stock. Any of the following risks could materially and adversely affect AbbVie's results of operations, financial condition or cash flows. The risk factors generally have been separated into two groups: risks related to AbbVie's business and risks related to AbbVie's common stock. Based on the information currently known to it, AbbVie believes that the following information identifies the most significant risk factors affecting it in each of these categories of risks. However, the risks and uncertainties AbbVie faces are not limited to those set forth in the risk factors described below and may not be in order of importance or probability of occurrence. Additional risks and uncertainties not presently known to AbbVie or that AbbVie currently believes to be immaterial may also adversely affect its business. In addition, past financial performance may not be a reliable indicator of future performance and historical trends should not be used to anticipate results or trends in future periods.
| | | | | | | | | | | |
13 |  | 2022 Form 10-K | 2022 Form 10-K | | |
If any of the following risks and uncertainties develops into actual events, these events could have a material adverse effect on AbbVie's business, results of operations, financial condition or cash flows. In such case, the trading price of AbbVie's common stock could decline.
Risks Related to AbbVie's Business
The expiration or loss of patent protection and licenses, including the loss of exclusivity for Humira and increased competition from biosimilars, may adversely affect AbbVie's revenues and operating earnings.
AbbVie relies on patent, trademark and other intellectual property protection in the discovery, development, manufacturing and sale of its products. In particular, patent protection is, in the aggregate, important in AbbVie's marketing of pharmaceutical products in the United States and most major markets outside of the United States. Patents covering AbbVie products normally provide market exclusivity, which is important for the profitability of many of AbbVie's products.
As patents for certain of its products expire, AbbVie could face competition from lower priced generic or biosimilar products. The expiration or loss of patent protection for a product typically is followed promptly by substitutes that may significantly reduce sales for that product in a short amount of time. If AbbVie's competitive position is compromised because of generics, biosimilars or otherwise, it could have a material adverse effect on AbbVie's business and results of operations. In addition, proposals emerge from time to time for legislation to further encourage the early and rapid approval of generic drugs or biosimilars. Any such proposals that are enacted into law could increase the impact of generic competition.
Large pharmaceutical companies and generics manufacturers of pharmaceutical products continue to expand into the biotechnology field and form partnerships to pursue biosimilars. Companies have developed and are developing biosimilars that compete with AbbVie’s biologic products, including Humira. As competitors obtain marketing approval for biosimilars referencing AbbVie’s biologic products, AbbVie’s products may become subject to competition from such biosimilars, with the attendant competitive pressure and consequences. Expiration of or successful challenges to AbbVie’s applicable patent rights could also trigger competition from other products, assuming any relevant exclusivity period has expired. As a result, AbbVie could face increased litigation and administrative proceedings with respect to the validity and/or scope of patents relating to its biologic products.
For example, Humira accounted for approximately 37% of AbbVie's total net revenues in 2022. Humira is facing competition from biosimilar products in the United States beginning in 2023 due to the loss of exclusivity, which AbbVie anticipates will cause a significant decline in Humira's revenue and could adversely affect AbbVie’s revenues and operating earnings.
AbbVie's principal patents and trademarks are described in greater detail in Item 1, "Business—Intellectual Property Protection and Regulatory Exclusivity" and Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations—Results of Operations," and litigation regarding these patents is described in Item 3, "Legal Proceedings."
AbbVie's major products could lose patent protection earlier than expected, which could adversely affect AbbVie's revenues and operating earnings.
A significant portion of AbbVie's revenue and operating earnings are derived from several major products. Third parties or government authorities may challenge or seek to invalidate or circumvent AbbVie's patents and patent applications. For example, manufacturers of generic pharmaceutical products file, and may continue to file, Abbreviated New Drug Applications with the FDA seeking to market generic forms of AbbVie's products prior to the expiration of relevant patents owned or licensed by AbbVie by asserting that the patents are invalid, unenforceable and/or not infringed. In addition, petitioners have filed, and may continue to file, challenges to the validity of AbbVie's patents under the 2011 Leahy-Smith America Invents Act, which created inter partes review and post grant review procedures for challenging patent validity in administrative proceedings at the United States Patent and Trademark Office.
Although most of the challenges to AbbVie's intellectual property have come from other businesses, governments may also challenge intellectual property rights. For example, court decisions and potential legislation relating to patents, such as legislation regarding biosimilars, and other regulatory initiatives may result in further erosion of intellectual property protection. In addition, certain governments outside the United States have indicated that compulsory licenses to patents may be sought to further their domestic policies or on the basis of national emergencies, such as HIV/AIDS. If triggered, compulsory licenses may diminish or eliminate sales and profits from those jurisdictions and negatively affect AbbVie's results of operations.
AbbVie normally responds to challenges by vigorously defending its patents, including by filing patent infringement lawsuits. Patent litigation, administrative proceedings and other challenges to AbbVie's patents are costly and unpredictable and may deprive AbbVie of market exclusivity for a patented product. To the extent AbbVie's intellectual property is
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 14 |
successfully challenged, circumvented or weakened, or to the extent such intellectual property does not allow AbbVie to compete effectively, AbbVie's business will suffer. To the extent that countries do not enforce AbbVie's intellectual property rights or require compulsory licensing of AbbVie's intellectual property, AbbVie's revenues and operating earnings will be reduced.
A third party's intellectual property may prevent AbbVie from selling its products or have a material adverse effect on AbbVie's profitability and financial condition.
Third parties may claim that an AbbVie product infringes upon their intellectual property. In addition, in its pursuit of valid business opportunities, AbbVie may be required to challenge intellectual property rights held by others that it believes were improperly granted. Resolving an intellectual property infringement or other claim can be costly and time consuming and may require AbbVie to enter into license agreements. AbbVie cannot guarantee that it would be able to obtain license agreements on commercially reasonable terms. A successful claim of patent or other intellectual property infringement could subject AbbVie to significant damages or an injunction preventing the manufacture, sale, or use of the affected AbbVie product or products. Any of these events could have a material adverse effect on AbbVie's profitability and financial condition.
AbbVie's research and development efforts may not succeed in developing and marketing commercially successful products and technologies, which may cause its revenues and profitability to decline.
To remain competitive, AbbVie must continue to launch new products and new indications and/or brand extensions for existing products. Such launches must generate revenue sufficient both to cover its substantial research and development costs and to replace revenues of profitable products that are lost to or displaced by competing products or therapies. Failure to do so would have a material adverse effect on AbbVie's revenue and profitability. Accordingly, AbbVie commits substantial effort, funds and other resources to research and development and must make ongoing substantial expenditures without any assurance that its efforts will be commercially successful. A high rate of failure in the biopharmaceutical industry is inherent in the research and development of new products, and failure can occur at any point in the research and development process, including after significant funds have been invested. Products that appear promising in development may fail to reach the market for numerous reasons, including failure to demonstrate effectiveness, safety concerns, superior safety or efficacy of competing therapies, failure to achieve positive clinical or pre-clinical outcomes beyond the current standards of care, inability to obtain necessary regulatory approvals or delays in the approval of new products and new indications, limited scope of approved uses, excessive costs to manufacture or the failure to obtain or maintain intellectual property rights, or infringement of the intellectual property rights of others.
Decisions about research studies made early in the development process of a pharmaceutical product candidate can affect the marketing strategy once such candidate receives approval. More detailed studies may demonstrate additional benefits that can help in the marketing, but they also consume time and resources and may delay submitting the pharmaceutical product candidate for regulatory approval. AbbVie cannot guarantee that a proper balance of speed and testing will be made with respect to each pharmaceutical product candidate or that decisions in this area would not adversely affect AbbVie's results of operations.
Even if AbbVie successfully develops and markets new products or enhancements to its existing products, they may be quickly rendered obsolete by changing clinical preferences, changing industry standards, or competitors' innovations. AbbVie's innovations may not be accepted quickly in the marketplace because of existing clinical practices or uncertainty over third-party reimbursement. AbbVie cannot state with certainty when or whether any of its products under development will be launched, whether it will be able to develop, license, or otherwise acquire compounds or products, or whether any products will be commercially successful. Failure to launch successful new products or new indications for existing products may cause AbbVie's products to become obsolete, causing AbbVie's revenues and operating results to suffer.
AbbVie is subject to cost-containment efforts and pricing pressures that could cause a reduction in revenues and operating earnings, and changes in the terms of rebate and chargeback programs, which are common in the pharmaceuticals industry, could have a material adverse effect on AbbVie's operations.
Cost-containment efforts by governments and private organizations are described in greater detail in Item 1, "Business—Regulation—Commercialization, Distribution and Manufacturing." To the extent these cost containment efforts are not offset by greater demand, increased patient access to health care, or other factors, AbbVie's revenues and operating earnings will be reduced. In the United States, the European Union and other countries, AbbVie's business has experienced downward pressure on product pricing, and this pressure could increase in the future.
AbbVie is subject to increasing public and legislative pressure with respect to pharmaceutical pricing. In the United States, practices of managed care groups, and institutional and governmental purchasers, as well as federal laws and regulations related to Medicare and Medicaid, contribute to pricing pressures. In particular, the Inflation Reduction Act of
| | | | | | | | | | | |
15 |  | 2022 Form 10-K | 2022 Form 10-K | | |
2022 requires (i) the government to negotiate prices for select high expenditure Medicare Part D drugs (prices effective beginning in 2026) and Part B drugs (prices effective beginning in 2028), (ii) manufacturers to pay a rebate for Medicare Part B and Part D drugs when prices for those drugs increase faster than inflation beginning in 2022 for Part D and 2023 for Part B, and (iii) a Medicare Part D redesign replacing the current coverage gap provisions and establishing a $2,000 cap for out-of-pocket costs for Medicare beneficiaries beginning in 2025, with manufacturers being responsible for 10% of costs up to the $2,000 cap and 20% after that cap is reached. The Inflation Reduction Act is to be implemented through forthcoming agency action, the outcome of which cannot be reasonably determined with certainty. Additionally, changes to U.S. tax laws now require (i) a 15% minimum tax generally applied to U.S. corporations on adjusted financial statement income beginning in 2023 and (ii) a non-deductible 1% excise tax provision on net stock repurchases, to be applied to repurchases beginning in 2023. AbbVie continues to evaluate the impact that the Inflation Reduction Act may have on the company. The potential for continuing changes to the health care system in the United States and the increased purchasing power of entities that negotiate on behalf of Medicare, Medicaid and private sector beneficiaries may result in additional pricing pressures.
In major markets worldwide, governments play a significant role in funding health care services and determining the pricing and reimbursement of pharmaceutical products. Consequently, in those markets, AbbVie is subject to government decision-making and budgetary actions with respect to its products. In particular, many European countries have ongoing government-mandated price reductions for many pharmaceutical products, and AbbVie anticipates continuing pricing pressures in Europe. Differences between countries' pricing regulations could lead to third-party cross-border trading in AbbVie's products that results in a reduction in revenues and operating earnings.
Rebates related to government programs, such as fee-for-service Medicaid or Medicaid managed care programs, arise from laws and regulations. AbbVie cannot predict with certainty if additional government initiatives to contain health care costs or other factors could lead to new or modified regulatory requirements that include higher or incremental rebates or discounts. Other rebate and discount programs arise from contractual agreements with private payers. Various factors, including market factors and the ability of private payers to control patient access to products, may provide payers the leverage to negotiate higher or additional rebates or discounts that could have a material adverse effect on AbbVie's operations.
A portion of AbbVie's near-term pharmaceutical pipeline relies on collaborations with third parties, which may adversely affect the development and sale of its products.
AbbVie depends on alliances and joint ventures with pharmaceutical and biotechnology companies for a portion of the products in its near-term pharmaceutical pipeline. Failures by these parties to meet their contractual, regulatory, or other obligations to AbbVie, or any disruption in the relationships between AbbVie and these third parties, could have an adverse effect on AbbVie's pharmaceutical pipeline and business. In addition, AbbVie's collaborative relationships for research and development extend for many years and may give rise to disputes regarding the relative rights, obligations and revenues of AbbVie and its collaboration partners, including the ownership of intellectual property and associated rights and obligations. This could result in the loss of intellectual property rights or protection, delay the development and sale of potential pharmaceutical products, affect the effective sale and delivery of AbbVie's commercialized products and lead to lengthy and expensive litigation, administrative proceedings or arbitration.
Biologics carry unique risks and uncertainties, which could have a negative impact on AbbVie's business and results of operations.
The successful discovery, development, manufacturing and sale of biologics is a long, expensive and uncertain process. There are unique risks and uncertainties with biologics. For example, access to and supply of necessary biological materials, such as cell lines, may be limited and current governmental regulations restrict access to and regulate the transport and use of such materials. In addition, the development, manufacturing and sale of biologics is subject to regulations that are often more complex and extensive than the regulations applicable to other pharmaceutical products. As a result, manufacturing biologics, especially in large quantities, is often complex and may require the use of innovative technologies. Such manufacturing also requires facilities specifically designed and validated for this purpose and sophisticated quality assurance and quality control procedures. Biologics are also frequently costly to manufacture because production inputs are derived from living animal or plant material, and some biologics cannot be made synthetically. Failure to successfully discover, develop, manufacture and sell biologics—including Humira and Skyrizi —could have a negative impact on AbbVie's business and results of operations.
New products and technological advances by AbbVie's competitors may negatively affect AbbVie's results of operations.
AbbVie competes with other research-based pharmaceutical and biotechnology companies that research, develop, manufacture, market and sell proprietary pharmaceutical products and biologics. For example, Humira competes with anti-
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 16 |
TNF products and other competitive products intended to treat a number of disease states and Mavyret/Maviret competes with other available hepatitis C treatment options. In addition, in the past few years, a number of other companies have started to develop, have successfully developed and/or are currently marketing products that are being positioned as competitors to Botox. All of these competitors may introduce new products or develop technological advances that compete with AbbVie’s products in therapeutic areas such as immunology, hematologic oncology, aesthetics, neuroscience and eye care. In addition, as AbbVie products lose exclusivity, competition surrounding such products will increase and generic and biosimilar products will increasingly penetrate the markets. AbbVie cannot predict with certainty the timing or impact of the introduction by competitors of new products or technological advances. Such competing products may be safer, more effective, more effectively marketed or sold, have lower prices or better insurance coverage or reimbursement levels, or have superior performance features than AbbVie’s products, and this may negatively impact AbbVie’s business and results of operations.
The manufacture of many of AbbVie's products is a highly exacting and complex process, and if AbbVie or one of its suppliers encounters problems manufacturing AbbVie's products, AbbVie's business could suffer.
The manufacture of many of AbbVie's products is a highly exacting and complex process, due in part to strict regulatory requirements. Problems may arise during manufacturing for a variety of reasons, including equipment malfunction, failure to follow specific protocols and procedures, problems with raw materials, delays related to the construction of new facilities or the expansion of existing facilities, including those intended to support future demand for AbbVie's products, changes in manufacturing production sites and limits to manufacturing capacity due to regulatory requirements, changes in the types of products produced, physical limitations that could inhibit continuous supply, man-made or natural disasters and environmental factors. If problems arise during the production of a batch of product, such batch of product may have to be discarded and AbbVie may experience product shortages or incur added expenses. This could, among other things, lead to increased costs, lost revenue, damage to customer relations, time and expense spent investigating the cause and, depending on the cause, similar losses with respect to other batches or products. If problems are not discovered before the product is released to the market, recall and product liability costs may also be incurred.
AbbVie uses raw materials and components in its pharmaceutical and biologic manufacturing processes, including those sourced from single suppliers, and an interruption in the supply of those raw materials and components could adversely affect AbbVie's business and results of operations.
AbbVie uses raw materials and components in its pharmaceutical and biologic manufacturing processes that may be sourced from single suppliers. The failure of AbbVie's suppliers, and particularly its single-source suppliers, to fulfill their contractual obligations in a timely manner or as a result of regulatory noncompliance or physical disruption at a manufacturing site may impair AbbVie's ability to deliver its products to customers on a timely and competitive basis, which could adversely affect AbbVie's business and results of operations. Increases in demand on any of AbbVie's suppliers could result in delays and disruptions in the manufacturing, distribution and sale of its products and/or product shortages, leading to lost revenue. Finding an alternative supplier could take a significant amount of time and involve significant expense due to the nature of the products and the need to obtain regulatory approvals. AbbVie cannot guarantee that it will be able to reach agreement with alternative providers or that regulatory authorities would approve AbbVie's use of such alternatives. Business interruption insurance may not provide adequate compensation in the case of a failure by a supplier.
Certain aspects of AbbVie’s operations are highly dependent upon third party service providers.
AbbVie relies on suppliers, vendors and other third party service providers to research, develop, manufacture, commercialize, promote and sell its products. Reliance on third party manufacturers reduces AbbVie’s oversight and control of the manufacturing process. Some of these third party providers are subject to legal and regulatory requirements, privacy and security risks and market risks of their own. The failure of a critical third party service provider to meet its obligations could have a material adverse impact on AbbVie’s operations and results. If any third party service providers have violated or are alleged to have violated any laws or regulations during the performance of their obligations to AbbVie, it is possible that AbbVie could suffer financial and reputational harm or other negative outcomes, including possible legal consequences.
Significant safety or efficacy issues could arise for AbbVie's products, which could have a material adverse effect on AbbVie's revenues and financial condition.
Pharmaceutical products receive regulatory approval based on data obtained in controlled clinical trials of limited duration. Following regulatory approval, these products will be used over longer periods of time in many patients. Additional, and perhaps more extensive, studies may also be conducted, which may be sponsored by AbbVie but could also be sponsored by competitors, insurance companies, government institutions, scientists, investigators or other interested parties. If new safety or efficacy issues are reported or if new scientific information becomes available (including results of post-marketing Phase 4 trials), or if governments change standards regarding safety, efficacy or labeling, AbbVie may be required to amend
| | | | | | | | | | | |
17 |  | 2022 Form 10-K | 2022 Form 10-K | | |
the conditions of use for a product. For example, AbbVie may voluntarily provide or be required to provide updated information on a product's label or narrow its approved indication, either of which could reduce the product's market acceptance. If safety or efficacy issues with an AbbVie product arise, sales of the product could be halted by AbbVie or by regulatory authorities and regulatory action could be taken by such regulatory authorities. Safety or efficacy issues affecting suppliers' or competitors' products also may reduce the market acceptance of similar AbbVie products.
New data about AbbVie's products, or products similar to its products, could negatively impact demand for AbbVie's products due to actual or perceived safety issues or uncertainty regarding efficacy and, in some cases, could result in product withdrawal. Furthermore, new data and information, including information about product misuse, may lead government agencies, professional societies, practice management groups or organizations involved with various diseases to publish guidelines or recommendations related to the use of AbbVie's products or the use of related therapies or place restrictions on sales. Such guidelines or recommendations may lead to lower sales of AbbVie's products.
AbbVie is subject to product liability claims and other lawsuits that may adversely affect its business, results of operations and reputation.
In the ordinary course of business, AbbVie is the subject of product liability claims and lawsuits alleging that AbbVie's current or historical products or the products of other companies that it promotes have resulted or could result in an unsafe condition for or injury to patients. For example, lawsuits are pending against Allergan, AbbVie’s subsidiary, and certain of its former officers alleging they made misrepresentations and omissions regarding Allergan’s textured breast implants. Product liability claims and lawsuits and safety alerts or product recalls, regardless of their ultimate outcome, may have a material adverse effect on AbbVie's business, results of operations and reputation and on its ability to attract and retain customers. Consequences may also include additional costs, a decrease in market share for the product in question, lower revenue and exposure to other claims. AbbVie evaluates its risks and has determined that the cost of obtaining product liability insurance outweighs the likely benefits of the coverage that is available and, as such, AbbVie's product liability losses are self-insured.
AbbVie is also the subject of other claims, legal proceedings and investigations in the ordinary course of business, which relate to intellectual property, commercial, securities and other matters. Adverse outcomes in such claims, legal proceedings and investigations may also adversely affect AbbVie’s business, results of operations and reputation. For example, Allergan was named as a defendant in approximately 3,000 matters relating to the promotion and sale of prescription opioid pain relievers. Allergan recently agreed to a nationwide settlement to resolve such matters, which will provide for up to $2.02 billion to help support state and local efforts to address opioid-related issues in the United States, among other things. However, AbbVie cannot guarantee that the conditions to such settlement will be satisfied, or that additional suits will not be filed. See Note 15, "Legal Proceedings and Contingencies" to the Consolidated Financial Statements included under Item 8, "Financial Statements and Supplementary Data." AbbVie cannot predict with certainty the outcome of these proceedings.
AbbVie is subject to governmental regulations, and it can be costly to comply with these regulations and to develop compliant products and processes.
AbbVie's products are subject to rigorous regulation by numerous international, supranational, federal and state authorities, as described in Item 1, "Business—Regulation—Discovery and Clinical Development,” “Business—Regulation—Commercialization, Distribution and Manufacturing,” and “Business—Regulation—Medical Devices.” The process of obtaining regulatory approvals to market a pharmaceutical product can be costly and time consuming, and approvals may not be granted for future products, or additional indications or uses of existing products, on a timely basis, if at all. Delays in the receipt of, or failure to obtain approvals for, future products, or new indications and uses, could result in delayed realization of product revenues, reduction in revenues and substantial additional costs.
The U.S. healthcare industry, in particular, is highly regulated and subject to frequent and substantial regulatory changes. It is expected that the U.S. healthcare industry will continue to be subject to increasing regulation as well as political and legal action, as future proposals to reform the healthcare system are considered by the executive branch, Congress and state legislatures. AbbVie cannot predict with certainty when additional changes in the healthcare industry in general, or the pharmaceutical industry in particular, will occur, or what the impact of such changes may be. For example, the Inflation Reduction Act is to be implemented through forthcoming agency action, the outcome of which cannot be reasonably determined with certainty.
In addition, AbbVie cannot guarantee that it will remain compliant with applicable regulatory requirements once approval has been obtained for a product. These requirements include, among other things, regulations regarding manufacturing practices, product labeling and advertising and post-marketing reporting, including adverse event reports and field alerts due to manufacturing quality concerns. AbbVie must incur expense and spend time and effort to ensure compliance with these complex regulations.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 18 |
Possible regulatory actions could result in substantial modifications to AbbVie's business practices and operations; refunds, recalls or seizures of AbbVie's products; a total or partial shutdown of production in one or more of AbbVie's or its suppliers' facilities while AbbVie or its supplier remedies the alleged violation; the inability to obtain future approvals; and withdrawals or suspensions of current products from the market. Any of these events could disrupt AbbVie's business and have a material adverse effect on its business and results of operations.
Laws and regulations affecting government benefit programs could impose new obligations on AbbVie, require it to change its business practices, and restrict its operations.
The health care industry is subject to federal, state and international laws and regulations pertaining to government benefit program reimbursements, rebates, price reporting and regulation and health care fraud and abuse. In the United States, these laws include anti-kickback and false claims laws, the Medicaid Rebate Statute, the Veterans Health Care Act, the U.S. Physician Payments Sunshine Act, the TRICARE program, the government pricing rules applicable to the Medicaid, Medicare Part B, 340B Drug Pricing Program and individual state laws relating to pricing and sales and marketing practices. Violations of such laws and regulations may be punishable by criminal and/or civil sanctions, including, in some instances, substantial fines, imprisonment and exclusion from participation in federal and state health care programs, including Medicare, Medicaid and Veterans Administration health programs. Such violations may also lead to product recalls and seizures, interruption of production leading to product shortages, import bans or denials of import certifications, delays or denials in the approvals of new products or supplemental approvals of current products pending resolution of the issues, and reputational harm, any of which would adversely affect AbbVie's business. These laws and regulations are broad in scope and are subject to change and evolving interpretations, which could require AbbVie to incur substantial costs associated with compliance or to alter one or more of its sales or marketing practices. In addition, violations of these laws and regulations, or allegations of such violations, could impose new obligations on AbbVie, require it to change its business practices and restrict its operations.
Public health outbreaks, epidemics or pandemics, such as the coronavirus (COVID-19), have had, and could in the future have, an adverse impact on AbbVie’s operations and financial condition.
Public health outbreaks, epidemics or pandemics have had, and could in the future have, an adverse impact on AbbVie’s operations and financial condition. The pandemic caused by the novel strain of coronavirus (COVID-19) caused many countries, including the United States, to declare national emergencies and implement preventive measures such as travel bans and shelter in place or total lock-down orders. The continuation or re-implementation of these bans and orders remains uncertain. The COVID-19 pandemic caused AbbVie to modify certain of its business practices, and AbbVie may take further actions as may be required by government authorities or as AbbVie determines are in the best interests of AbbVie’s employees, patients, customers and business partners.
While the impact of COVID-19 on AbbVie’s operations, including, among others, its manufacturing and supply chain, sales and marketing, commercial and clinical trial operations, to date was not material, AbbVie experienced lower new patient starts in certain products and markets. The impact of COVID-19 and other public health outbreaks on AbbVie over the long-term is uncertain and cannot be predicted with confidence. The extent of the adverse impact of COVID-19 or any other public health outbreak on AbbVie’s operations will depend on the extent and severity of the continued spread of the disease globally, the timing and nature of actions taken to respond to it and the resulting economic consequences. Ultimately, efforts to mitigate the impact of COVID-19 or any other public health outbreak may not completely prevent AbbVie's business from being adversely affected and future impacts remain uncertain.
The international nature of AbbVie's business subjects it to additional business risks that may cause its revenue and profitability to decline.
AbbVie's business is subject to risks associated with doing business internationally, including in emerging markets. Net revenues outside of the United States made up approximately 21% of AbbVie's total net revenues in 2022. The risks associated with AbbVie's operations outside the United States include:
•fluctuations in currency exchange rates;
•changes in medical reimbursement policies and programs and pricing restrictions;
•multiple legal and regulatory requirements that are subject to change and that could restrict AbbVie's ability to manufacture, market and sell its products;
•differing local product preferences and product requirements;
•trade protection measures and import or export licensing requirements;
| | | | | | | | | | | |
19 |  | 2022 Form 10-K | 2022 Form 10-K | | |
•international trade disruptions or disputes;
•difficulty in establishing, staffing and managing operations;
•differing labor regulations;
•potentially negative consequences from changes in or interpretations of tax laws;
•political and economic instability, including as a result of the COVID-19 pandemic;
•sovereign debt issues;
•price and currency exchange controls, limitations on participation in local enterprises, expropriation, nationalization and other governmental action and regulation;
•inflation, recession and fluctuations in interest rates;
•restrictions on transfers of funds;
•potential deterioration in the economic position and credit quality of certain non-U.S. countries; and
•potential penalties or other adverse consequences for violations of anti-corruption, anti-bribery and other similar laws and regulations, including the United States Foreign Corrupt Practices Act and the United Kingdom Bribery Act.
In particular, in February 2022, armed conflict escalated between Russia and Ukraine. In response thereto, the United States and other North Atlantic Treaty Organizations member states, as well as certain non-member states, announced targeted economic sanctions, embargoes and export controls on Russia and Belarus. Although AbbVie is continuing to supply essential pharmaceutical products in Russia for humanitarian reasons while working to maintain compliance with evolving international sanctions, it has suspended operations for all aesthetics products in Russia. AbbVie's operations in Russia, Belarus and Ukraine are not significant. However, it is not possible to predict with certainty the continued consequences of this conflict, which, in addition to sanctions, have included regional instability, geopolitical shifts and adverse effects on macroeconomic conditions, currency exchange rates and financial markets. Any such consequences could have an adverse impact on AbbVie's business and results of operations.
If AbbVie does not effectively and profitably commercialize its products, AbbVie's revenues and financial condition could be adversely affected.
AbbVie must effectively and profitably commercialize its principal products by creating and meeting continued market demand; achieving market acceptance and generating product sales; ensuring that the active pharmaceutical ingredient(s) for a product and the finished product are manufactured in sufficient quantities and in compliance with requirements of the FDA and similar foreign regulatory agencies and with acceptable quality and pricing to meet commercial demand; and ensuring that the entire supply chain efficiently and consistently delivers AbbVie's products to its customers. The commercialization of AbbVie products may not be successful due to, among other things, unexpected challenges from competitors, new safety issues or concerns being reported that may impact or narrow approved indications, the relative price of AbbVie's product as compared to alternative treatment options and changes to a product's label that further restrict its marketing. If the commercialization of AbbVie's principal products is unsuccessful, AbbVie's revenues and financial condition could be adversely affected.
AbbVie may acquire other businesses, license rights to technologies or products, form alliances, or dispose of assets, which could cause it to incur significant expenses and could negatively affect profitability.
AbbVie may pursue acquisitions, technology licensing arrangements, joint ventures and strategic alliances, or dispose of some of its assets, as part of its business strategy. AbbVie may not complete these transactions in a timely manner, on a cost-effective basis, or at all, and may not realize the expected benefits. If AbbVie is successful in making an acquisition, the products and technologies that are acquired may not be successful or may require significantly greater resources and investments than originally anticipated. AbbVie may not be able to integrate acquisitions successfully into its existing business and could incur or assume significant debt and unknown or contingent liabilities. AbbVie could also experience negative effects on its reported results of operations from acquisition or disposition-related charges, amortization of expenses related to intangibles and charges for impairment of long-term assets. These effects could cause a deterioration of AbbVie's credit rating and result in increased borrowing costs and interest expense.
Additionally, changes in AbbVie's structure, operations, revenues, costs, or efficiency resulting from major transactions such as acquisitions, divestitures, mergers, alliances, joint ventures, restructurings or other strategic initiatives, may result in
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 20 |
greater than expected costs, may take longer than expected to complete or encounter other difficulties, including the need for regulatory approval where appropriate.
AbbVie is dependent on wholesale distributors for distribution of its products in the United States and, accordingly, its business and results of operations could be adversely affected if they encounter financial or other difficulties.
In 2022, three wholesale distributors (McKesson Corporation, Cardinal Health, Inc. and AmerisourceBergen Corporation) accounted for substantially all of AbbVie's pharmaceutical product sales in the United States. If one of its significant wholesale distributors encounters financial or other difficulties, such distributor may decrease the amount of business that it does with AbbVie, and AbbVie may be unable to collect all the amounts that the distributor owes it on a timely basis or at all, which could adversely affect AbbVie's business and results of operations.
AbbVie has debt obligations that could adversely affect its business and its ability to meet its obligations.
The amount of debt that AbbVie has incurred and intends to incur could have important consequences to AbbVie and its investors. These consequences include, among other things, requiring a portion of AbbVie's cash flow from operations to make interest payments on this debt and reducing the cash flow available to fund capital expenditures and other corporate purposes and to grow AbbVie's business. To the extent AbbVie incurs additional indebtedness or interest rates increase, these risks could increase further. In addition, AbbVie's cash flow from operations may not be sufficient to repay all of the outstanding debt as it becomes due, and AbbVie may not be able to borrow money, sell assets, or otherwise raise funds on acceptable terms, or at all, to refinance its debt.
AbbVie may need additional financing in the future to meet its capital needs or to make opportunistic acquisitions, and such financing may not be available on favorable terms, if at all.
AbbVie may need additional financing in the future to meet its capital needs or to make opportunistic acquisitions. For example, it may need to increase its investment in research and development activities. The capital and credit markets may experience extreme volatility and disruption, which may lead to uncertainty and liquidity issues for both borrowers and investors, and AbbVie may be unable to obtain any desired additional financing on terms favorable to it, if at all. If AbbVie loses its investment grade credit rating or adequate funds are not available on acceptable terms, AbbVie may be unable to fund its expansion, successfully develop or enhance products, or respond to competitive pressures, any of which could negatively affect AbbVie's business. If AbbVie raises additional funds by issuing debt or entering into credit facilities, it may be subject to limitations on its operations due to restrictive covenants. Failure to comply with these covenants could adversely affect AbbVie's business.
AbbVie depends on information technology and a failure of, or significant disruption to, those systems could have a material adverse effect on AbbVie's business.
AbbVie relies on sophisticated software applications and complex information technology systems to operate its business, which may be vulnerable to malicious intrusion, random attack, loss of data privacy, disruption, degradation or breakdown. Data privacy or security breaches by employees or others may in the future result in the failure of critical business operations. Such breaches may cause sensitive data, including intellectual property, trade secrets or personal information belonging to AbbVie, its patients, customers or business partners, to be exposed to unauthorized persons or to the public. To date, neither AbbVie’s business nor operations have been materially impacted by such incidents. Although AbbVie has invested in the protection of its data and information technology and monitors its systems on an ongoing basis, there can be no assurance that such efforts will prevent material breakdowns or breaches in AbbVie's information technology systems that could have a material adverse effect on AbbVie's business. Such adverse consequences could include loss of revenue or the loss of critical or sensitive information from AbbVie’s or third-party providers’ databases or IT systems and could also result in legal, financial, reputational or business harm to AbbVie and potentially substantial remediation costs. In addition, while AbbVie maintains cyber insurance, it cannot guarantee that such insurance will be sufficient to cover the financial, legal, business or reputational losses that may result from an interruption or breach of AbbVie systems.
AbbVie’s balances of intangible assets, including developed product rights and goodwill acquired, are subject to impairment testing and may result in impairment charges, which may adversely affect AbbVie’s results of operations and financial condition.
A significant amount of AbbVie’s total assets is related to acquired intangibles and goodwill. As of December 31, 2022, the carrying value of AbbVie’s developed product rights and other intangible assets was $67.4 billion and the carrying value of AbbVie’s goodwill was $32.2 billion.
AbbVie’s developed product rights are stated at cost, less accumulated amortization. AbbVie determines original fair value and amortization periods for developed product rights based on its assessment of various factors impacting estimated
| | | | | | | | | | | |
21 |  | 2022 Form 10-K | 2022 Form 10-K | | |
useful lives and cash flows of the acquired products. Significant adverse changes to any of these factors require AbbVie to perform an impairment test on the affected asset and, if evidence of impairment exists, require AbbVie to take an impairment charge with respect to the asset. For assets that are not impaired, AbbVie may adjust the remaining useful lives. Such a charge could adversely affect AbbVie’s results of operations and financial condition.
AbbVie’s other significant intangible assets include in-process research and development (IPR&D) intangible projects, acquired in recent business combinations, which are indefinite-lived intangible assets. For IPR&D assets, the risk of failure is significant, and there can be no certainty that these assets ultimately will yield successful products. AbbVie's ability to realize value on these significant investments is often contingent upon, among other things, regulatory approvals and market conditions. As such, IPR&D assets may become impaired and/or be written off at some point in the future if the associated research and development effort is abandoned or is curtailed.
Goodwill and AbbVie’s IPR&D intangible assets are tested for impairment annually, or when events occur, or circumstances change that could potentially reduce the fair value of the reporting unit or intangible asset. Impairment testing compares the fair value of the reporting unit or intangible asset to its carrying amount. A goodwill or IPR&D impairment, if any, would be recorded in operating income and could have a material adverse effect on AbbVie’s results of operations and financial condition.
Failure to attract, develop and retain highly qualified personnel could affect AbbVie’s ability to successfully develop and commercialize products.
AbbVie’s success is largely dependent on its continued ability to attract, develop and retain diverse, highly qualified scientific, technical and management personnel, as well as personnel with expertise in clinical research and development, governmental regulation and commercialization. Competition for qualified personnel in the biopharmaceutical field is intense and increasing. AbbVie cannot be sure that it will be able to attract and retain quality personnel or that the costs of doing so will not materially increase.
The illegal distribution and sale by third parties of counterfeit or unregistered versions of AbbVie products could have a material adverse impact on its reputation, business and results of operations.
Third parties may illegally obtain, distribute, and sell counterfeit or illegally diverted from their intended market versions of AbbVie products. These versions of product would not meet AbbVie's rigorous manufacturing, testing, distribution and quality standards. A patient who receives a counterfeit, stolen, or diverted drug may be at risk for a number of dangerous health consequences. The prevalence of counterfeit/diverted medicines is an industry-wide issue due to a variety of factors, including the adoption of e-commerce, which increased during the COVID-19 pandemic, greatly enhancing consumers' ability to obtain prescriptions and other medical treatments via the internet in lieu of traditional brick and mortar pharmacies. This can expose patients to greater risks as the internet is a preferred vehicle for dangerous counterfeit/diverted product offers and scams because of the anonymity it affords. AbbVie's reputation and business could suffer harm as a result of counterfeit or diverted drugs sold under its brand name which may also result in reduced revenues that could negatively affect our results of operation.
Other factors can have a material adverse effect on AbbVie's profitability and financial condition.
Many other factors can affect AbbVie's results of operations, cash flows and financial condition, including:
•changes in or interpretations of laws and regulations, including changes in accounting standards, taxation requirements, product marketing application standards, data privacy laws, particularly in the European Union and the United States and environmental laws;
•differences between the fair value measurement of assets and liabilities and their actual value, particularly for pension and post-employment benefits, stock-based compensation, intangibles and goodwill; and for contingent liabilities such as litigation and contingent consideration, the absence of a recorded amount, or an amount recorded at the minimum, compared to the actual amount;
•changes in the rate of inflation (including the cost of raw materials, commodities and supplies), interest rates, market value of AbbVie's equity investments and the performance of investments held by it or its employee benefit trusts;
•changes in the creditworthiness of counterparties that transact business with or provide services to AbbVie or its employee benefit trusts;
•environmental liabilities in connection with AbbVie’s manufacturing processes and distribution logistics, including the handling of hazardous materials;
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 22 |
•changes in the ability of third parties that provide information technology, accounting, human resources, payroll and other outsourced services to AbbVie to meet their contractual obligations to AbbVie;
•the failure, or perceived failure, of achieving environmental, social and governance objectives;
•information loss or damage to AbbVie's reputation, brand, image or goodwill due to increased use of social media platforms;
•business interruptions stemming from natural disasters, such as climate change, earthquakes, hurricanes, flooding, fires, or efforts taken by third parties to prevent or mitigate such disasters; and
•changes in business, economic and political conditions, including: war, political instability, terrorist attacks, the threat of future terrorist activity and related military action; natural disasters; the cost and availability of insurance due to any of the foregoing events; labor disputes, strikes, slow-downs, or other forms of labor or union activity; and pressure from third-party interest groups.
Risks Related to AbbVie's Common Stock
AbbVie cannot guarantee the timing, amount, or payment of dividends on its common stock or the repurchase of its common stock.
Although AbbVie expects to pay regular cash dividends, the timing, declaration, amount and payment of future dividends to stockholders will fall within the discretion of AbbVie's board of directors. The board's decisions regarding the payment of dividends will depend on many factors, such as AbbVie's financial condition, earnings, capital requirements, debt service obligations, industry practice, legal requirements, regulatory constraints and other factors that the board deems relevant. For more information, see Item 5, "Market for Registrant's Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities." AbbVie's ability to pay dividends and repurchase shares under its share repurchase program will depend on its ongoing ability to generate cash from operations and access capital markets. AbbVie cannot guarantee that it will continue to pay a dividend in the future.
An AbbVie stockholder's percentage of ownership in AbbVie may be diluted in the future.
In the future, a stockholder's percentage ownership in AbbVie may be diluted because of equity issuances for capital market transactions, equity awards that AbbVie will be granting to AbbVie's directors, officers and employees, acquisitions or other purposes. AbbVie's employees have options to purchase shares of its common stock as a result of conversion of their Abbott stock options (in whole or in part) to AbbVie stock options. AbbVie anticipates its compensation committee will grant additional stock options or other stock-based awards to its employees. Such awards will have a dilutive effect on AbbVie's earnings per share, which could adversely affect the market price of AbbVie's common stock. From time to time, AbbVie will issue additional options or other stock-based awards to its employees under AbbVie's employee benefits plans.
In addition, AbbVie's amended and restated certificate of incorporation authorizes AbbVie to issue, without the approval of AbbVie's stockholders, one or more classes or series of preferred stock having such designation, powers, preferences and relative, participating, optional and other special rights, including preferences over AbbVie's common stock respecting dividends and distributions, as AbbVie's board of directors generally may determine. The terms of one or more classes or series of preferred stock could dilute the voting power or reduce the value of AbbVie's common stock. For example, AbbVie could grant the holders of preferred stock the right to elect some number of AbbVie's directors in all events or on the happening of specified events or the right to veto specified transactions. Similarly, the repurchase or redemption rights or liquidation preferences AbbVie could assign to holders of preferred stock could affect the residual value of the common stock.
Certain provisions in AbbVie's amended and restated certificate of incorporation and amended and restated by-laws, and of Delaware law, may prevent or delay an acquisition of AbbVie, which could decrease the trading price of AbbVie's common stock.
AbbVie's amended and restated certificate of incorporation and amended and restated by-laws contain, and Delaware law contains, provisions that are intended to deter coercive takeover practices and inadequate takeover bids by encouraging prospective acquirors to negotiate with AbbVie's board of directors rather than to attempt a hostile takeover. These provisions include, among others:
•the inability of AbbVie's stockholders to call a special meeting;
•the division of AbbVie's board of directors into three classes of directors, with each class serving a staggered three-year term;
| | | | | | | | | | | |
23 |  | 2022 Form 10-K | 2022 Form 10-K | | |
•a provision that stockholders may only remove directors for cause;
•the ability of AbbVie's directors, and not stockholders, to fill vacancies on AbbVie's board of directors; and
•the requirement that the affirmative vote of stockholders holding at least 80% of AbbVie's voting stock is required to amend certain provisions in AbbVie's amended and restated certificate of incorporation and AbbVie's amended and restated by-laws relating to the number, term and election of AbbVie's directors, the filling of board vacancies, the calling of special meetings of stockholders and director and officer indemnification provisions.
In addition, Section 203 of the Delaware General Corporation Law provides that, subject to limited exceptions, persons that acquire, or are affiliated with a person that acquires, more than 15% of the outstanding voting stock of a Delaware corporation shall not engage in any business combination with that corporation, including by merger, consolidation or acquisitions of additional shares, for a three-year period following the date on which that person or its affiliates becomes the holder of more than 15% of the corporation's outstanding voting stock.
AbbVie believes these provisions protect its stockholders from coercive or otherwise unfair takeover tactics by requiring potential acquirors to negotiate with AbbVie's board of directors and by providing AbbVie's board of directors with more time to assess any acquisition proposal. These provisions are not intended to make the company immune from takeovers. However, these provisions apply even if the offer may be considered beneficial by some stockholders and could delay or prevent an acquisition that AbbVie's board of directors determines is not in the best interests of AbbVie and AbbVie's stockholders. These provisions may also prevent or discourage attempts to remove and replace incumbent directors.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 24 |
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains certain forward-looking statements regarding business strategies, market potential, future financial performance and other matters. The words "believe," "expect," "anticipate," "project" and similar expressions and uses of future or conditional verbs, generally identify "forward looking statements," which speak only as of the date the statements were made. The matters discussed in these forward-looking statements are subject to risks, uncertainties and other factors that could cause actual results to differ materially from those expressed or implied in the forward-looking statements. In particular, information included under Item 1, "Business," Item 1A, "Risk Factors," and Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations" contain forward looking statements. Where, in any forward looking statement, an expectation or belief as to future results or events is expressed or implied, such expectation or belief is based on the current plans and expectations of AbbVie management and expressed in good faith and believed to have a reasonable basis, but there can be no assurance that the expectation or belief will result or be achieved or accomplished. Factors that could cause actual results or events to differ materially from those anticipated include, but are not limited to, the matters described under Item 1A, "Risk Factors" and Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations." AbbVie does not undertake, and specifically declines, any obligation to update the forward-looking statements included in this Annual Report on Form 10-K to reflect events or circumstances after the date hereof, unless AbbVie is required by applicable securities law to do so.
ITEM 1B. UNRESOLVED STAFF COMMENTS None.
AbbVie's corporate offices are located at 1 North Waukegan Road, North Chicago, Illinois 60064-6400. As of December 31, 2022, AbbVie owns or leases approximately 637 facilities worldwide, containing an aggregate of approximately 19 million square feet of floor space dedicated to production, distribution and administration. AbbVie's significant manufacturing facilities are in the following locations:
| | | | | | | | |
| United States | | Outside the United States |
| Abbott Park, Illinois* | | Campoverde di Aprilia, Italy |
| Barceloneta, Puerto Rico | | Clonshaugh, Ireland |
| Branchburg, New Jersey* | | La Aurora, Costa Rica |
| Campbell, California | | Ludwigshafen, Germany |
| Cincinnati, Ohio | | Pringy, France |
| Dublin, California* | | Singapore* |
| Irvine, California | | Sligo, Ireland |
| North Chicago, Illinois | | Westport, Ireland* |
| Waco, Texas | | |
| Worcester, Massachusetts* | | |
| Wyandotte, Michigan* | | |
_______________________________________________________________________________
* Leased property.
AbbVie believes its facilities are suitable and provide adequate production capacity for its current and projected operations. There are no material encumbrances on AbbVie's owned properties.
In the United States, including Puerto Rico, AbbVie has two central distribution centers. AbbVie also has research and development facilities in the United States located at: Abbott Park, Illinois; Branchburg, New Jersey; Cambridge, Massachusetts; Irvine, California; Madison, New Jersey; North Chicago, Illinois; Pleasanton, California; Santa Cruz, California; South San Francisco, California; and Worcester, Massachusetts. Outside the United States, AbbVie's principal research and development facilities are located in Ludwigshafen, Germany.
| | | | | | | | | | | |
25 |  | 2022 Form 10-K | 2022 Form 10-K | | |
ITEM 3. LEGAL PROCEEDINGS Information pertaining to legal proceedings is provided in Note 15, "Legal Proceedings and Contingencies" to the Consolidated Financial Statements included under Item 8, "Financial Statements and Supplementary Data," and is incorporated by reference herein.
ITEM 4. MINE SAFETY DISCLOSURES Not applicable.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 26 |
INFORMATION ABOUT OUR EXECUTIVE OFFICERS
The following table lists AbbVie's executive officers:
| | | | | | | | | | | | | | |
| Name | | Age | | Position |
| Richard A. Gonzalez | | 69 | | Chairman of the Board and Chief Executive Officer |
| Robert A. Michael | | 52 | | Vice Chairman and President |
| Scott T. Reents | | 55 | | Executive Vice President, Chief Financial Officer |
| Timothy J. Richmond | | 56 | | Executive Vice President, Chief Human Resources Officer |
| Azita Saleki-Gerhardt, Ph.D. | | 59 | | Executive Vice President, Operations |
| Perry C. Siatis | | 48 | | Executive Vice President, General Counsel and Secretary |
| Jeffrey R. Stewart | | 54 | | Executive Vice President, Chief Commercial Officer |
| Thomas J. Hudson, M.D. | | 61 | | Senior Vice President, Research & Development and Chief Scientific Officer |
| Elaine K. Sorg | | 56 | | Senior Vice President, AbbVie and President, U.S. Commercial Operations |
| Carrie Strom | | 45 | | Senior Vice President, AbbVie and President, Global Allergan Aesthetics |
| Brian L. Durkin | | 62 | | Vice President, Controller |
_______________________________________________________________________________
Mr. Gonzalez is the Chairman and Chief Executive Officer of AbbVie. He served as Abbott’s Executive Vice President of the Pharmaceutical Products Group from July 2010 to December 2012, and was responsible for Abbott’s worldwide pharmaceutical business, including commercial operations, research and development and manufacturing. He also served as President, Abbott Ventures Inc., Abbott’s medical technology investment arm, from 2009 to 2011. Mr. Gonzalez joined Abbott in 1977 and held various management positions. He was first appointed as an AbbVie corporate officer in December 2012.
Mr. Michael is AbbVie’s Vice Chairman and President. Mr. Michael previously served as Vice Chairman, Finance and Commercial Operations and Chief Financial Officer from June 2021 to June 2022, as Executive Vice President, Chief Financial Officer from 2019 to 2021, as Senior Vice President, Chief Financial Officer from 2018 to 2019 and as Vice President, Controller from 2017 to 2018. He served as AbbVie’s Vice President, Treasurer from 2015 to 2016, as Vice President, Controller, Commercial Operations from 2013 to 2015 and Vice President, Financial Planning and Analysis from 2012 to 2013. At Abbott, Mr. Michael served as Division Controller, Nutrition Supply Chain from 2010 to 2012. Mr. Michael joined Abbott in 1993 and was first appointed as an AbbVie corporate officer in March 2017.
Mr. Reents is AbbVie’s Executive Vice President, Chief Financial Officer. He previously served as Senior Vice President, Chief Financial Officer from June 2022 to November 2022, as Vice President, Tax and Treasury from 2019 to June 2022, and as Vice President, Tax from 2013 to 2019. Mr. Reents joined Abbott in 2008 and was first appointed as an AbbVie corporate officer in June 2022.
Mr. Richmond is AbbVie’s Executive Vice President, Chief Human Resources Officer. He served as Senior Vice President, Human Resources from 2013 to 2018. Mr. Richmond served as Abbott’s Divisional Vice President of Compensation & Benefits from 2008 to 2012, as Group Vice President of Talent and Rewards from 2007 to 2008 and as Divisional Vice President of Talent Acquisition from 2006 to 2007. Mr. Richmond joined Abbott in 2006 and was first appointed as an AbbVie corporate officer in December 2012.
Dr. Saleki-Gerhardt is AbbVie’s Executive Vice President, Operations. She served as Senior Vice President, Operations from 2013 to 2018. Dr. Saleki-Gerhardt served as Abbott’s Vice President, Pharmaceuticals Manufacturing and Supply from 2011 to 2012, and as Divisional Vice President, Quality Assurance, Global Pharmaceutical Operations from 2008 to 2011. Dr. Saleki-Gerhardt joined Abbott in 1993 and was first appointed as an AbbVie corporate officer in December 2012. She serves on the board of Entegris Inc.
Mr. Siatis is AbbVie’s Executive Vice President, General Counsel and Secretary. Mr. Siatis previously served as Senior Vice President, Deputy General Counsel from September 2021 until October 2022. From 2013 until 2021, Mr. Siatis also served in various roles including as Senior Vice President, Legal and Chief Ethics and Compliance Officer; Senior Vice President of Legal Transactions and R&D/Alliance Management and Chief Ethics and Compliance Officer; and Vice President, Biologic Strategic Development and Legal Regulatory. Mr. Siatis joined Abbott in 2005 and was first appointed as an AbbVie corporate officer in October 2022.
Mr. Stewart is AbbVie’s Executive Vice President, Chief Commercial Officer. He previously served as Senior Vice President, U.S. Commercial Operations from 2018 to 2020 and as AbbVie’s President, Commercial Operations from 2013 to
| | | | | | | | | | | |
27 |  | 2022 Form 10-K | 2022 Form 10-K | | |
2018. Prior to AbbVie’s separation from Abbott, he served as Vice President, Abbott Proprietary Pharmaceutical Division, United States. Mr. Stewart joined Abbott in 1992 and was first appointed as an AbbVie corporate officer in December 2018.
Dr. Hudson is AbbVie's Senior Vice President, Research & Development and Chief Scientific Officer. He previously served as Vice President, Head of Oncology Discovery and Early Development from 2016 to 2019. Prior to joining AbbVie, Dr. Hudson served at the Ontario Institute for Cancer Research as President and Scientific Director. He also previously served as Founder and Director of the McGill University and Genome Quebec Innovation Centre and Assistant Director of the Whitehead/MIT Center for Genome Research. Dr. Hudson was first appointed as an AbbVie corporate officer in July 2019.
Ms. Sorg is AbbVie’s Senior Vice President, AbbVie, and President U.S. Commercial Operations. She previously served as AbbVie’s President, U.S. Immunology and Patient Services from 2019 to 2020 and as Vice President, Immunology and Oncology from 2016 to 2018. She served as Vice President, Immunology prior to AbbVie’s separation from Abbott and until 2016 at AbbVie. Ms. Sorg joined Abbott in 2012 and was first appointed as an AbbVie corporate officer in November 2020. Prior to joining Abbott, Ms. Sorg served in management roles at Eli Lilly and Company for 23 years.
Ms. Strom is AbbVie’s Senior Vice President, AbbVie, and President, Global Allergan Aesthetics, responsible for the worldwide operations of the aesthetics franchise. She was appointed to the position upon AbbVie’s acquisition of Allergan in 2020 and was first appointed as an AbbVie corporate officer in May 2020. At Allergan, Ms. Strom previously served as Senior Vice President, U.S. Medical Aesthetics from 2018 to 2020. She joined Allergan in 2011.
Mr. Durkin is AbbVie’s Vice President, Controller. Mr. Durkin previously served as Vice President, Internal Audit from 2016 to 2018. Prior to joining AbbVie, he served as Vice President of Finance and Division Controller for Abbott’s Vision Care business from 2009 to 2016 and Controller Pharmaceutical Research and Development from 2005 to 2009. Mr. Durkin joined Abbott in 1986 and was first appointed as an AbbVie corporate officer in October 2018.
The executive officers of AbbVie are elected annually by the board of directors. All other officers are elected by the board or appointed by the Chairman of the Board. All officers are either elected at the first meeting of the board of directors held after the annual stockholder meeting or appointed by the Chairman of the Board after that board meeting. Each officer holds office until a successor has been duly elected or appointed and qualified or until the officer's death, resignation, or removal. There are no family relationships between any of the executive officers listed above.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 28 |
PART II
ITEM 5. MARKET FOR REGISTRANT'S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES Principal Market
The principal market for AbbVie's common stock is the New York Stock Exchange (Symbol: ABBV). AbbVie's common stock is also listed on the Chicago Stock Exchange and traded on various regional and electronic exchanges.
Stockholders
There were 44,487 stockholders of record of AbbVie common stock as of January 31, 2023.
Performance Graph
The following graph compares the cumulative total returns of AbbVie, the S&P 500 Index and the NYSE Arca Pharmaceuticals Index for the period from December 31, 2017 through December 31, 2022. This graph assumes $100 was invested in AbbVie common stock and each index on December 31, 2017 and also assumes the reinvestment of dividends. The stock price performance on the following graph is not necessarily indicative of future stock price performance.
This performance graph is furnished and shall not be deemed "filed" with the SEC or subject to Section 18 of the Securities Exchange Act of 1934, nor shall it be deemed incorporated by reference in any of AbbVie's filings under the Securities Act of 1933, as amended.
| | | | | | | | | | | |
29 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Dividends
On October 28, 2022, AbbVie's board of directors declared an increase in the quarterly cash dividend from $1.41 per share to $1.48 per share, payable on February 15, 2023, to stockholders of record as of January 13, 2023. The timing, declaration, amount of and payment of any dividends by AbbVie in the future is within the discretion of its board of directors and will depend upon many factors, including AbbVie's financial condition, earnings, capital requirements of its operating subsidiaries, covenants associated with certain of AbbVie's debt service obligations, legal requirements, regulatory constraints, industry practice, ability to access capital markets and other factors deemed relevant by its board of directors. Moreover, if AbbVie determines to pay any dividend in the future, there can be no assurance that it will continue to pay such dividends or the amount of such dividends.
Issuer Purchases of Equity Securities | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Period | Total
Number
of Shares
(or Units)
Purchased | | Average
Price
Paid per Share
(or Unit) | | Total
Number of
Shares (or Units)
Purchased as Part
of Publicly
Announced
Plans or
Programs | | Maximum Number (or
Approximate Dollar Value) of
Shares (or Units) that May
Yet Be Purchased Under the
Plans or Programs | |
| October 1, 2022 - October 31, 2022 | 926 | | (1) | $ | 141.34 | | (1) | — | | | $ | 1,393,714,917 | | |
| November 1, 2022 - November 30, 2022 | 940 | | (1) | $ | 146.61 | | (1) | — | | | $ | 1,393,714,917 | | |
| December 1, 2022 - December 31, 2022 | 26,019 | | (1) | $ | 161.87 | | (1) | — | | | $ | 1,393,714,917 | | |
| Total | 27,885 | | (1) | $ | 160.67 | | (1) | — | | | $ | 1,393,714,917 | | |
1. In addition to AbbVie shares repurchased on the open market under a publicly announced program, if any, these shares also included the shares purchased on the open market for the benefit of participants in the AbbVie Employee Stock Purchase Plan – 926 in October; 940 in November; and 26,019 in December.
These shares do not include the shares surrendered to AbbVie to satisfy minimum tax withholding obligations in connection with the vesting or exercise of stock-based awards.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 30 |
| | | | | | | | | | | |
31 |  | 2022 Form 10-K | 2022 Form 10-K | | |
ITEM 7. MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS The following is a discussion and analysis of the financial condition of AbbVie Inc. (AbbVie or the company). This commentary should be read in conjunction with the Consolidated Financial Statements and accompanying notes appearing in Item 8, "Financial Statements and Supplementary Data." This section of Form 10-K generally discusses 2022 and 2021 items and year-to-year comparisons between 2022 and 2021. Discussions of 2020 items and year-to-year comparisons between 2021 and 2020 that are not included in this Form 10-K can be found in “Management's Discussion and Analysis of Financial Condition and Results of Operations” in Part II, Item 7 of the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2021.
EXECUTIVE OVERVIEW
Company Overview
AbbVie is a global, diversified research-based biopharmaceutical company positioned for success with a comprehensive product portfolio that has leadership positions across immunology, oncology, aesthetics, neuroscience and eye care. AbbVie uses its expertise, dedicated people and unique approach to innovation to develop and market advanced therapies that address some of the world’s most complex and serious diseases.
AbbVie's products are generally sold worldwide directly to wholesalers, distributors, government agencies, health care facilities, specialty pharmacies and independent retailers from AbbVie-owned distribution centers and public warehouses. Certain products (including aesthetic products and devices) are also sold directly to physicians and other licensed healthcare providers. In the United States, AbbVie distributes pharmaceutical products principally through independent wholesale distributors, with some sales directly to retailers, pharmacies, patients or other customers. Outside the United States, AbbVie sells products primarily to wholesalers or through distributors, and depending on the market works through largely centralized national payers system to agree on reimbursement terms. Certain products are co-marketed or co-promoted with other companies. AbbVie operates as a single global business segment and has approximately 50,000 employees.
2022 Financial Results
AbbVie's strategy has focused on delivering strong financial results, maximizing the benefits of the Allergan acquisition, advancing and investing in its pipeline and returning value to shareholders while ensuring a strong, sustainable growth business over the long term. The company's financial performance in 2022 included delivering worldwide net revenues of $58.1 billion, operating earnings of $18.1 billion, diluted earnings per share of $6.63 and cash flows from operations of $24.9 billion. Worldwide net revenues increased by 3% on a reported basis and 5% on a constant currency basis, reflecting growth across its immunology, neuroscience and aesthetics portfolios.
Diluted earnings per share in 2022 was $6.63 and included the following after-tax costs: (i) $6.4 billion related to the amortization of intangible assets; (ii) $2.8 billion for the change in fair value of contingent consideration liabilities; (iii) $2.0 billion for charges related to litigation matters; (iv) $766 million of acquisition and integration expenses; and (v) $604 million related to intangible asset impairment. These costs were partially offset by an after-tax gain of $126 million related to the divestiture of Pylera and a benefit of $26 million related to certain tax items. Additionally, financial results reflected continued funding to support all stages of AbbVie’s pipeline assets and continued investment in AbbVie’s on-market brands.
Following the closing of the Allergan acquisition in 2020, AbbVie implemented an integration plan designed to reduce costs, integrate and optimize the combined organization. As a result of the successful execution of the integration plan, AbbVie realized $2.5 billion of annual cost synergies in 2022.
To achieve these integration objectives, AbbVie incurred total cumulative charges of $2.3 billion through 2022. These costs consisted of severance and employee benefit costs (cash severance, non-cash severance, including accelerated equity award compensation expense, retention and other termination benefits) and other integration expenses.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 32 |
Recent Global Events
Russia/Ukraine
In response to the military conflict between Russia and Ukraine, the United States and other North Atlantic Treaty Organization member states, as well as certain non-member states, announced targeted economic sanctions and export controls on Russia and Belarus. These include restrictions on the export and transfer of products containing certain toxins, including Botox, to Russia and Belarus. However, AbbVie is not prohibited to continue the sale of essential pharmaceutical products to help ensure patients receive an uninterrupted supply of their medicines. In March 2022, AbbVie announced the suspension of operations for all aesthetics products in Russia. In April 2022, AbbVie also announced that all profits from the sales of essential medicines in Russia will be donated to support direct humanitarian relief efforts in Ukraine. While the company’s operations in Russia, Belarus and Ukraine are not significant, if the conflict escalates and results in broader economic and political concerns, AbbVie’s business could be adversely impacted.
Impact of the Coronavirus Disease 2019 (COVID-19)
In response to COVID-19, AbbVie continues to closely manage manufacturing and supply chain resources around the world to help ensure that patients continue to receive an uninterrupted supply of their medicines. Clinical trial sites are being monitored locally to protect the safety of study participants, staff and employees. While the impact of COVID-19 on AbbVie's operations to date has not been material, AbbVie continues to experience lower new patient starts in certain products and markets. AbbVie expects this matter could continue to negatively impact its results of operations throughout the duration of the pandemic.
The extent to which COVID-19 may impact AbbVie's financial condition and results of operations remains uncertain and is dependent on numerous evolving factors, including the measures being taken by authorities to mitigate against the spread of COVID-19, the emergence of new variants and the effectiveness of vaccines and therapeutics.
2023 Strategic Objectives
AbbVie's mission is to discover and develop innovative medicines and products that solve serious health issues today and address the medical challenges of tomorrow while achieving top-tier financial performance through outstanding execution. AbbVie intends to execute its strategy and advance its mission in a number of ways, including: (i) maximizing the benefits of a diversified revenue base with multiple long-term growth drivers; (ii) leveraging AbbVie's commercial strength and international infrastructure across therapeutic areas and ensuring strong commercial execution of new product launches; (iii) continuing to invest in and expand its pipeline in support of opportunities in immunology, oncology, aesthetics, neuroscience and eye care as well as continued investment in key on-market products; (iv) generating substantial operating cash flows to support investment in innovative research and development, and return cash to shareholders via a strong and growing dividend while also reducing debt. In addition, AbbVie anticipates several regulatory submissions and data readouts from key clinical trials in the next 12 months.
AbbVie expects to achieve its strategic objectives through:
•Skyrizi and Rinvoq revenue growth driven by increasing market share and indication expansion.
•Advancing our hematologic oncology portfolio by increasing Venclexta market share and new indications, strong commercial execution of new product launches and effectively managing market and competitive challenges impacting Imbruvica.
•Continuing investment in the global expansion of aesthetics and increasing market penetration of Botox and Juvederm Collection.
•Neuroscience revenue growth driven by Vraylar, Botox Therapeutic, Ubrelvy and Qulipta.
•Maximizing AbbVie's existing eye care portfolio.
•Effectively managing the impact of Humira biosimilar erosion.
•The favorable impact of pipeline products and indications recently approved or currently under regulatory review where approval is expected in 2023. These products are described in greater detail in the section labeled "Research and Development" included as part of this Item 7.
| | | | | | | | | | | |
33 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Research and Development
Research and innovation are the cornerstones of AbbVie's business as a global biopharmaceutical company. AbbVie's long-term success depends to a great extent on its ability to continue to discover and develop innovative products and acquire or collaborate on compounds currently in development by other biotechnology or pharmaceutical companies.
AbbVie's pipeline currently includes over 80 compounds, devices or indications in development individually or under collaboration or license agreements and is focused on such important specialties as immunology, oncology, aesthetics, neuroscience and eye care. Of these programs, approximately 50 are in mid- and late-stage development.
The following sections summarize transitions of significant programs from mid-stage development to late-stage development as well as developments in significant late-stage and registration programs. AbbVie expects multiple mid-stage programs to transition into late-stage programs in the next 12 months.
Significant Programs and Developments
Immunology
Skyrizi
•In January 2022, AbbVie announced that the U.S. Food and Drug Administration (FDA) approved Skyrizi for the treatment of adults with active psoriatic arthritis.
•In June 2022, AbbVie announced that the FDA approved Skyrizi for the treatment of adults with moderately to severely active Crohn’s disease.
•In November 2022, AbbVie announced that the European Commission (EC) approved Skyrizi for the treatment of adults with moderately to severely active Crohn's disease who have had inadequate response, lost response or were intolerant to conventional or biologic therapy.
Rinvoq
•In January 2022, AbbVie announced that the FDA approved Rinvoq for the treatment of moderate to severe atopic dermatitis in adults and children 12 years of age and older whose disease did not respond to previous treatment and is not well controlled with other pills or injections, including biologic medicines, or when use of other pills or injections is not recommended.
•In February 2022, AbbVie announced top-line results from its second Phase 3 induction study, U-Excel, for Rinvoq in patients with moderate to severe Crohn’s disease who had an inadequate response or were intolerant to conventional or biologic therapy met the primary and most key secondary endpoints.
•In March 2022, AbbVie announced that the FDA approved Rinvoq for the treatment of adults with moderately to severely active ulcerative colitis (UC) who have had an inadequate response or intolerance to one or more tumor necrosis factor (TNF) blockers.
•In April 2022, AbbVie announced that the FDA approved Rinvoq for the treatment of adults with active ankylosing spondylitis who have had an inadequate response or intolerance to one or more TNF blockers.
•In May 2022, AbbVie announced positive top-line results from U-ENDURE, a Phase 3 maintenance study for Rinvoq in adult patients with moderate to severe Crohn's disease who had an inadequate response or were intolerant to a conventional or biologic therapy. The results showed that more patients treated with Rinvoq achieved the co-primary and secondary endpoints at one year compared to placebo.
•In July 2022, AbbVie announced that the EC approved Rinvoq for the treatment of adults with moderately to severely active UC who have had an inadequate response, lost response or were intolerant to either conventional therapy or a biologic agent.
•In July 2022, AbbVie announced its submission of a supplemental New Drug Application (sNDA) to the FDA and a marketing authorization application (MAA) to the EMA for Rinvoq for the treatment of adult patients with moderately to severely active Crohn’s disease.
•In July 2022, AbbVie announced that the EC approved Rinvoq for the treatment of adult patients with active non-radiographic axial spondyloarthritis (nr-axSpA).
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 34 |
•In October 2022, AbbVie announced that the FDA approved Rinvoq for the treatment of adults with active nr-axSpA with objective signs of inflammation who have had an inadequate response or intolerance to TNF blocker therapy.
•In November 2022, AbbVie announced that the EMA's Committee for Medical Products for Human Use (CHMP) adopted an opinion following a review of the benefit-risk of medicines within the JAK inhibitor class for the treatment of inflammatory diseases, including Rinvoq. Confirming the recommendation from the Pharmacovigilance Risk Assessment Committee (PRAC), the CHMP did not recommend changes to the current Rinvoq indication statements and recommended updates to dosage and special warnings for all JAK inhibitor products indicated for the treatment of inflammatory diseases. These recommendations will be forwarded to the EC, which is expected to issue a final decision.
Oncology
Teliso-V
•In January 2022, AbbVie announced that the FDA granted Breakthrough Therapy Designation to investigational telisotuzumab vedotin (Teliso-V) for the treatment of patients with advanced/metastatic epidermal growth factor receptor wild type, nonsquamous non-small cell lung cancer with high levels of c-Met overexpression whose disease has progressed on or after platinum-based therapy.
•In May 2022, AbbVie initiated a Phase 3 clinical trial to evaluate Teliso-V versus docetaxel for the treatment of patients with previously treated c-Met overexpressing, epidermal growth factor receptor wild type, advanced/metastatic non-squamous non-small cell lung cancer.
Epcoritamab
•In March 2022, Genmab A/S (Genmab) announced that the FDA granted orphan-drug designation to the investigational medicine, epcoritamab (DuoBody-CD3xCD20), for the treatment of follicular lymphoma. Genmab and AbbVie are co-developing epcoritamab and will share commercial responsibilities in the U.S. and Japan, with AbbVie responsible for further global commercialization.
•In June 2022, AbbVie and Genmab announced primary results from the large B-cell lymphoma expansion cohort in the EPCORE NHL-1 phase 2 clinical trial evaluating epcoritamab, an investigational subcutaneous bispecific antibody. In this study, epcoritamab demonstrated efficacy with durable responses in patients who had previously received at least two prior lines of anti-lymphoma therapy including chimeric antigen receptor T-cell therapy.
•In September 2022, AbbVie and Genmab submitted a biological license application (BLA) to the FDA for epcoritamab for the treatment of patients with relapsed/refractory large B-cell lymphoma.
•In October 2022, AbbVie and Genmab submitted an MAA to the EMA for epcoritamab for the treatment of patients with relapsed/refractory diffuse large B-cell lymphoma.
•In October 2022, AbbVie initiated a Phase 3 clinical trial to evaluate epcoritamab in combination with rituximab and lenalidomide compared to rituximab and lenalidomide in patients with relapsed or refractory follicular lymphoma.
•In November 2022, AbbVie announced that the FDA has accepted for priority review the BLA for epcoritamab for the treatment of relapsed/refractory large B-cell lymphoma.
Imbruvica
•In August 2022, AbbVie announced that the FDA approved the use of Imbruvica for the treatment of pediatric patients one year and older with chronic graft versus host disease after failure of one or more lines of systemic therapy.
•In August 2022, the National Comprehensive Cancer Network (NCCN) in the United States issued updated guidelines for the management of chronic lymphocytic leukemia (CLL) re-categorizing Imbruvica from “Preferred Regimen” to “Other Recommended Regimen”.
| | | | | | | | | | | |
35 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Aesthetics
Juvederm Collection
•In February 2022, AbbVie announced that the FDA approved Juvederm Volbella XC for improvement of infraorbital hollows in adults over the age of 21.
•In August 2022, AbbVie announced that the FDA approved Juvederm Volux XC for the improvement of jawline definition in adults over the age of 21 with moderate to severe loss of jawline definition.
BoNTE
•In March 2022, AbbVie initiated three Phase 3 clinical trials to evaluate the efficacy and safety of BoNTE (AGN-151586) for the treatment of glabellar lines.
Neuroscience
Vraylar
•In December 2022, AbbVie announced that the FDA approved Vraylar as an adjunctive therapy to antidepressants for the treatment of major depressive disorder in adults.
Qulipta
•In March 2022, AbbVie announced results from the Phase 3 PROGRESS trial for Qulipta in the preventive treatment of chronic migraine in adults met the primary endpoint and resulted in significant improvements in all secondary endpoints after adjustment for multiple comparisons.
•In June 2022, AbbVie submitted an sNDA to the FDA for Qulipta for the preventative treatment of chronic migraine in adults.
•In July 2022, AbbVie submitted an MAA to the EMA for Qulipta for the prophylactic treatment of migraine in adult patients who have at least four migraine days per month.
ABBV-951
•In May 2022, AbbVie submitted a New Drug Application to the FDA for ABBV-951 (foscarbidopa/foslevodopa) for the treatment of motor fluctuations in patients with advanced Parkinson's disease.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 36 |
RESULTS OF OPERATIONS
Net Revenues
The comparisons presented at constant currency rates reflect comparative local currency net revenues at the prior year's foreign exchange rates. This measure provides information on the change in net revenues assuming that foreign currency exchange rates had not changed between the prior and current periods. AbbVie believes that the non-GAAP measure of change in net revenues at constant currency rates, when used in conjunction with the GAAP measure of change in net revenues at actual currency rates, may provide a more complete understanding of the company's operations and can facilitate analysis of the company's results of operations, particularly in evaluating performance from one period to another. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | Percent change |
| | | | | | | At actual currency rates | | At constant currency rates |
| years ended (dollars in millions) | 2022 | | 2021 | | 2020 | | 2022 | | 2021 | | 2022 | | 2021 |
| United States | $ | 45,713 | | | $ | 43,510 | | | $ | 34,879 | | | 5.1 | % | | 24.7 | % | | 5.1 | % | | 24.7 | % |
| International | 12,341 | | | 12,687 | | | 10,925 | | | (2.7) | % | | 16.1 | % | | 5.5 | % | | 12.6 | % |
| Net revenues | $ | 58,054 | | | $ | 56,197 | | | $ | 45,804 | | | 3.3 | % | | 22.7 | % | | 5.1 | % | | 21.9 | % |
| | | | | | | | | | | |
37 |  | 2022 Form 10-K | 2022 Form 10-K | | |
The following table details AbbVie's worldwide net revenues: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | Percent change |
| | | | | | | | | At actual currency rates | | At constant currency rates |
| years ended December 31 (dollars in millions) | | 2022 | | 2021 | | 2020 | | 2022 | | 2021 | | 2022 | | 2021 |
| Immunology | | | | | | | | | | | | | | |
| Humira | United States | | $ | 18,619 | | | $ | 17,330 | | | $ | 16,112 | | | 7.4 | % | | 7.6 | % | | 7.4 | % | | 7.6 | % |
| International | | 2,618 | | | 3,364 | | | 3,720 | | | (22.2) | % | | (9.6) | % | | (14.9) | % | | (12.8) | % |
| Total | | $ | 21,237 | | | $ | 20,694 | | | $ | 19,832 | | | 2.6 | % | | 4.3 | % | | 3.8 | % | | 3.7 | % |
| Skyrizi | United States | | $ | 4,484 | | | $ | 2,486 | | | $ | 1,385 | | | 80.4 | % | | 79.6 | % | | 80.4 | % | | 79.6 | % |
| International | | 681 | | | 453 | | | 205 | | | 50.4 | % | | >100.0 % | | 67.1 | % | | >100.0 % |
| Total | | $ | 5,165 | | | $ | 2,939 | | | $ | 1,590 | | | 75.7 | % | | 84.9 | % | | 78.3 | % | | 84.0 | % |
| Rinvoq | United States | | $ | 1,794 | | | $ | 1,271 | | | $ | 653 | | | 41.2 | % | | 94.8 | % | | 41.2 | % | | 94.8 | % |
| International | | 728 | | | 380 | | | 78 | | | 91.4 | % | | >100.0 % | | >100.0 % | | >100.0 % |
| Total | | $ | 2,522 | | | $ | 1,651 | | | $ | 731 | | | 52.8 | % | | >100.0 % | | 58.1 | % | | >100.0 % |
| Hematologic Oncology | | | | | | | | | | | | | | |
| Imbruvica | United States | | $ | 3,426 | | | $ | 4,321 | | | $ | 4,305 | | | (20.7) | % | | 0.4 | % | | (20.7) | % | | 0.4 | % |
| Collaboration revenues | | 1,142 | | | 1,087 | | | 1,009 | | | 5.1 | % | | 7.7 | % | | 5.1 | % | | 7.7 | % |
| Total | | $ | 4,568 | | | $ | 5,408 | | | $ | 5,314 | | | (15.5) | % | | 1.8 | % | | (15.5) | % | | 1.8 | % |
| Venclexta | United States | | $ | 1,009 | | | $ | 934 | | | $ | 804 | | | 8.0 | % | | 16.1 | % | | 8.0 | % | | 16.1 | % |
| International | | 1,000 | | | 886 | | | 533 | | | 12.9 | % | | 66.2 | % | | 24.6 | % | | 60.9 | % |
| Total | | $ | 2,009 | | | $ | 1,820 | | | $ | 1,337 | | | 10.4 | % | | 36.1 | % | | 16.1 | % | | 34.0 | % |
| Aesthetics | | | | | | | | | | | | | | |
Botox Cosmetic (a) | United States | | $ | 1,654 | | | $ | 1,424 | | | $ | 687 | | | 16.2 | % | | >100.0 % | | 16.2 | % | | >100.0 % |
| International | | 961 | | | 808 | | | 425 | | | 18.9 | % | | 90.0 | % | | 28.8 | % | | 83.9 | % |
| Total | | $ | 2,615 | | | $ | 2,232 | | | $ | 1,112 | | | 17.2 | % | | >100.0 % | | 20.8 | % | | 98.4 | % |
Juvederm Collection (a) | United States | | $ | 548 | | | $ | 658 | | | $ | 318 | | | (16.7) | % | | >100.0 % | | (16.7) | % | | >100.0 % |
| International | | 880 | | | 877 | | | 400 | | | 0.3 | % | | >100.0 % | | 8.9 | % | | >100.0 % |
| Total | | $ | 1,428 | | | $ | 1,535 | | | $ | 718 | | | (7.0) | % | | >100.0 % | | (2.1) | % | | >100.0 % |
Other Aesthetics (a) | United States | | $ | 1,122 | | | $ | 1,268 | | | $ | 666 | | | (11.5) | % | | 90.2 | % | | (11.5) | % | | 90.2 | % |
| International | | 168 | | | 198 | | | 94 | | | (14.9) | % | | >100.0 % | | (8.3) | % | | >100.0 % |
| Total | | $ | 1,290 | | | $ | 1,466 | | | $ | 760 | | | (12.0) | % | | 93.0 | % | | (11.1) | % | | 91.9 | % |
| Neuroscience | | | | | | | | | | | | | | | |
Botox Therapeutic (a) | United States | | $ | 2,255 | | | $ | 2,012 | | | $ | 1,155 | | | 12.1 | % | | 74.3 | % | | 12.1 | % | | 74.3 | % |
| International | | 464 | | | 439 | | | 232 | | | 5.6 | % | | 89.0 | % | | 15.3 | % | | 78.8 | % |
| Total | | $ | 2,719 | | | $ | 2,451 | | | $ | 1,387 | | | 10.9 | % | | 76.7 | % | | 12.6 | % | | 75.0 | % |
Vraylar (a) | United States | | $ | 2,037 | | | $ | 1,728 | | | $ | 951 | | | 17.9 | % | | 81.7 | % | | 17.9 | % | | 81.7 | % |
| International | | 1 | | | — | | | — | | | n/m | | n/m | | n/m | | n/m |
| Total | | $ | 2,038 | | | $ | 1,728 | | | $ | 951 | | | 17.9 | % | | 81.7 | % | | 17.9 | % | | 81.7 | % |
| Duodopa | United States | | $ | 95 | | | $ | 102 | | | $ | 103 | | | (6.7) | % | | (1.0) | % | | (6.7) | % | | (1.0) | % |
| International | | 363 | | | 409 | | | 391 | | | (11.3) | % | | 4.6 | % | | (0.8) | % | | (0.1) | % |
| Total | | $ | 458 | | | $ | 511 | | | $ | 494 | | | (10.4) | % | | 3.4 | % | | (2.0) | % | | (0.3) | % |
Ubrelvy (a) | United States | | $ | 680 | | | $ | 552 | | | $ | 125 | | | 23.2 | % | | >100.0 % | | 23.2 | % | | >100.0 % |
| Qulipta | United States | | $ | 158 | | | $ | — | | | $ | — | | | >100.0 % | | n/m | | >100.0 % | | n/m |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
Other Neuroscience (a) | United States | | $ | 456 | | | $ | 667 | | | $ | 528 | | | (30.5) | % | | 26.3 | % | | (30.5) | % | | 26.3 | % |
| International | | 19 | | | 18 | | | 11 | | | 4.8 | % | | 77.4 | % | | 9.0 | % | | 64.7 | % |
| Total | | $ | 475 | | | $ | 685 | | | $ | 539 | | | (29.6) | % | | 27.2 | % | | (29.5) | % | | 27.0 | % |
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 38 |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | Percent change |
| | | | | | | | | At actual currency rates | | At constant currency rates |
| years ended December 31 (dollars in millions) | | 2022 | | 2021 | | 2020 | | 2022 | | 2021 | | 2022 | | 2021 |
| Eye Care | | | | | | | | | | | | | | |
Lumigan/Ganfort (a) | United States | | $ | 242 | | | $ | 273 | | | $ | 165 | | | (11.0) | % | | 64.7 | % | | (11.0) | % | | 64.7 | % |
| International | | 272 | | | 306 | | | 213 | | | (11.3) | % | | 44.1 | % | | (3.0) | % | | 38.1 | % |
| Total | | $ | 514 | | | $ | 579 | | | $ | 378 | | | (11.2) | % | | 53.1 | % | | (6.8) | % | | 49.7 | % |
Alphagan/Combigan (a) | United States | | $ | 202 | | | $ | 373 | | | $ | 223 | | | (45.8) | % | | 66.5 | % | | (45.8) | % | | 66.5 | % |
| International | | 144 | | | 156 | | | 103 | | | (7.9) | % | | 52.5 | % | | 2.5 | % | | 50.6 | % |
| Total | | $ | 346 | | | $ | 529 | | | $ | 326 | | | (34.6) | % | | 62.1 | % | | (31.5) | % | | 61.5 | % |
Restasis (a) | United States | | $ | 621 | | | $ | 1,234 | | | $ | 755 | | | (49.6) | % | | 63.3 | % | | (49.6) | % | | 63.3 | % |
| International | | 45 | | | 56 | | | 32 | | | (20.2) | % | | 75.3 | % | | (13.8) | % | | 80.1 | % |
| Total | | $ | 666 | | | $ | 1,290 | | | $ | 787 | | | (48.3) | % | | 63.8 | % | | (48.0) | % | | 64.0 | % |
Other Eye Care (a) | United States | | $ | 538 | | | $ | 523 | | | $ | 305 | | | 2.3 | % | | 72.7 | % | | 2.3 | % | | 72.7 | % |
| International | | 637 | | | 646 | | | 388 | | | (1.2) | % | | 66.1 | % | | 8.7 | % | | 61.0 | % |
| Total | | $ | 1,175 | | | $ | 1,169 | | | $ | 693 | | | 0.4 | % | | 69.0 | % | | 5.9 | % | | 66.1 | % |
| Other Key Products | | | | | | | | | | | | | | |
| Mavyret | United States | | $ | 755 | | | $ | 754 | | | $ | 785 | | | 0.2 | % | | (4.0) | % | | 0.2 | % | | (4.0) | % |
| International | | 786 | | | 956 | | | 1,045 | | | (17.8) | % | | (8.5) | % | | (8.5) | % | | (10.8) | % |
| Total | | $ | 1,541 | | | $ | 1,710 | | | $ | 1,830 | | | (9.9) | % | | (6.5) | % | | (4.7) | % | | (7.8) | % |
| Creon | United States | | $ | 1,278 | | | $ | 1,191 | | | $ | 1,114 | | | 7.3 | % | | 6.9 | % | | 7.3 | % | | 6.9 | % |
Linzess/Constella (a) | United States | | $ | 1,003 | | | $ | 1,006 | | | $ | 649 | | | (0.4) | % | | 55.1 | % | | (0.4) | % | | 55.1 | % |
| International | | 32 | | | 32 | | | 18 | | | 0.3 | % | | 77.3 | % | | 7.6 | % | | 66.4 | % |
| Total | | $ | 1,035 | | | $ | 1,038 | | | $ | 667 | | | (0.3) | % | | 55.7 | % | | (0.1) | % | | 55.4 | % |
| All other | | | $ | 4,137 | | | $ | 5,019 | | | $ | 5,119 | | | (17.6) | % | | (2.0) | % | | (16.3) | % | | (2.8) | % |
| Total net revenues | | $ | 58,054 | | | $ | 56,197 | | | $ | 45,804 | | | 3.3 | % | | 22.7 | % | | 5.1 | % | | 21.9 | % |
n/m – Not meaningful
(a)Net revenues include Allergan product revenues after the acquisition closing date of May 8, 2020.
The following discussion and analysis of AbbVie's net revenues by product is presented on a constant currency basis.
Global Humira sales increased 4% in 2022 primarily driven by market growth across therapeutic categories, partially offset by direct biosimilar competition in international markets. In the United States, Humira sales increased 7% in 2022 primarily driven by market growth across all indications and favorable pricing. This increase was partially offset by a lower market share following the corresponding market share gains of Skyrizi and Rinvoq. Internationally, Humira revenues decreased 15% in 2022 primarily driven by direct biosimilar competition. On January 31, 2023, Humira lost exclusivity in the United States. Following this loss of exclusivity, AbbVie expects direct biosimilar competition and Humira net revenues to decline in the United States. AbbVie continues to pursue strategies to maintain broad formulary access of Humira and manage the impact of biosimilar erosion.
Net revenues for Skyrizi increased 78% in 2022 primarily driven by continued strong volume and market share uptake since launch as a treatment for plaque psoriasis as well as market growth. Net revenues were also favorably impacted by recent regulatory approvals and expansion of Skyrizi for the treatment of psoriatic arthritis and Crohn’s disease.
Net revenues for Rinvoq increased 58% in 2022 primarily driven by continued strong volume and market share uptake since launch for the treatment of moderate to severe rheumatoid arthritis as well as market growth. Net revenues were also favorably impacted by recent regulatory approvals and expansion of Rinvoq for the treatment of psoriatic arthritis, atopic dermatitis, ankylosing spondylitis, ulcerative colitis and non-radiographic axial spondyloarthritis.
Net revenues for Imbruvica represent product revenues in the United States and collaboration revenues outside of the United States related to AbbVie's 50% share of Imbruvica profit. AbbVie's global Imbruvica revenues decreased 16% in 2022 as a result of decreased market demand and lower market share in the United States. The decrease in net revenues was also partially offset by increased collaboration revenues.
Net revenues for Venclexta increased 16% in 2022 primarily due to continued expansion of Venclexta for the treatment of patients with CLL and acute myeloid leukemia.
| | | | | | | | | | | |
39 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Net revenues for Botox Cosmetic increased 21% in 2022 due to sustained consumer demand in the United States, which was moderated in the second half of the year by economic pressures impacting consumer discretionary spending, and increased investment in key international markets.
Net revenues for Juvederm Collection decreased 2% in 2022 due to economic pressures impacting consumer discretionary spending and increased pricing promotions to support the market. International net revenues increased by 9% due to increased investment in key markets, partially offset by the suspension of aesthetic operations in Russia and the impact of COVID-19 in China.
Net revenues for Botox Therapeutic increased 13% in 2022 due to market growth.
Net revenues for Vraylar increased 18% in 2022 due to higher market share and market growth.
Net revenues for Ubrelvy increased 23% in 2022 primarily due to increased market share uptake since launch, partially offset by unfavorable pricing.
Net revenues for Qulipta increased greater than 100% in 2022 due to strong volume and market share uptake since launch for the preventative treatment of episodic migraine in adults.
Net revenues for Mavyret decreased 5% in 2022 due to the continued disruption of global hepatitis C virus markets due to the COVID-19 pandemic.
Gross Margin | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | Percent change |
| years ended December 31 (dollars in millions) | 2022 | | 2021 | | 2020 | | 2022 | | 2021 |
| Gross margin | $ | 40,640 | | | $ | 38,751 | | | $ | 30,417 | | | 5 | % | | 27 | % |
| as a percent of net revenues | 70 | % | | 69 | % | | 66 | % | | | | |
Gross margin as a percentage of net revenues in 2022 increased compared to 2021. Gross margin percentage for 2022 was favorably impacted by changes in product mix, partially offset by an intangible asset impairment charge of $770 million.
Selling, General and Administrative | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | Percent change |
| years ended December 31 (dollars in millions) | 2022 | | 2021 | | 2020 | | 2022 | | 2021 |
| Selling, general and administrative | $ | 15,260 | | | $ | 12,349 | | | $ | 11,299 | | | 24 | % | | 9 | % |
| as a percent of net revenues | 26 | % | | 22 | % | | 25 | % | | | | |
Selling, general and administrative (SG&A) expenses as a percentage of net revenues increased in 2022 compared to the prior year primarily due to the unfavorable impact of litigation reserve charges of $2.5 billion, partially offset by leverage from revenue growth and increased synergies realized.
Research and Development and Acquired IPR&D and Milestones | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | Percent change |
| years ended December 31 (dollars in millions) | 2022 | | 2021 | | 2020 | | 2022 | | 2021 |
| Research and development | $ | 6,510 | | | $ | 6,922 | | | $ | 6,379 | | | (6) | % | | 9 | % |
| as a percent of net revenues | 11 | % | | 12 | % | | 14 | % | | | | |
| Acquired IPR&D and milestones | $ | 697 | | | $ | 1,124 | | | $ | 1,376 | | | (38) | % | | (18) | % |
R&D expenses as a percentage of net revenues decreased in 2022 compared to 2021. R&D expense percentage for 2022 was favorably impacted by increased scale of the combined company and synergies realized, the purchase of priority review vouchers from third parties in the prior year as well as lower integration costs related to the acquisition of Allergan.
Acquired IPR&D and milestones expense represents upfront and subsequent development milestone payments incurred prior to regulatory approval to acquire rights to in-process R&D projects through R&D collaborations, licensing arrangements or other asset acquisitions. Acquired IPR&D and milestones expense in 2022 included a charge of $130 million related to acquiring Syndesi Therapeutics SA, charges related to other upfront payments totaling $315 million and development milestones of $252 million. Acquired IPR&D and milestones expense in 2021 included a charge of $400 million related to exercising the company's exclusive right to acquire TeneoOne, a charge of $370 million related to a collaboration
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 40 |
agreement with REGENXBIO Inc, charges related to other upfront payments totaling $192 million and development milestones of $162 million. See Note 5 to the Consolidated Financial Statements for additional information.
Other Operating Expense, Net
Other operating expense, net in 2022 included a one-time charge of $229 million related to an asset divested as part of the Allergan acquisition, partially offset by $172 million of income related to the sale of worldwide commercial rights of a mature brand Pylera. Other operating expense, net in 2021 included a $500 million charge related to the extension of the Calico Life Sciences LLC collaboration. See Note 5 to the Consolidated Financial Statements for additional information.
Other Non-Operating Expenses | | | | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | | 2022 | | 2021 | | 2020 |
Interest expense | | $ | 2,230 | | | $ | 2,423 | | | $ | 2,454 | |
Interest income | | (186) | | | (39) | | | (174) | |
Interest expense, net | | $ | 2,044 | | | $ | 2,384 | | | $ | 2,280 | |
| | | | | | |
| Net foreign exchange loss | | $ | 148 | | | $ | 51 | | | $ | 71 | |
| Other expense, net | | 2,448 | | | 2,500 | | | 5,614 | |
Interest expense in 2022 decreased compared to 2021 primarily due to a lower average debt balance as a result of deleveraging, partially offset by the impact of higher interest rates.
Interest income in 2022 increased compared to 2021 primarily due to the impact of higher interest rates.
Other expense, net included charges related to changes in fair value of contingent consideration liabilities of $2.8 billion in 2022 and $2.7 billion in 2021. The fair value of contingent consideration liabilities is impacted by the passage of time and multiple other inputs, including the probability of success of achieving regulatory/commercial milestones, discount rates, the estimated amount of future sales of the acquired products and other market-based factors. In 2022, the change in fair value reflected higher estimated Skyrizi sales driven by stronger market share uptake and the passage of time, partially offset by higher discount rates. In 2021, the change in fair value reflected higher estimated Skyrizi sales driven by stronger market share uptake, favorable clinical trial results and the passage of time, partially offset by higher discount rates.
Income Tax Expense
The effective income tax rate was 12% in 2022, 11% in 2021 and negative 36% in 2020. The effective income tax rates differed from the U.S. statutory tax rate of 21% principally due to the impact of foreign operations which reflects the impact of lower income tax rates in locations outside the United States, tax incentives in Puerto Rico and other foreign tax jurisdictions, business development activities and changes in fair value of contingent consideration. The effective tax rates for these periods also reflected the benefit from U.S. tax credits principally related to research and development credits, the orphan drug tax credit and Puerto Rico excise tax credits. The Puerto Rico tax credits relate to excise tax on certain products manufactured in Puerto Rico. The tax is levied on gross inventory purchases from entities in Puerto Rico and is included in cost of products sold in the consolidated statements of earnings. The majority of the tax is creditable for U.S. income tax purposes. In 2022, Puerto Rico enacted Act 52-2002 (the “Puerto Rico Act”) allowing for a transition from a Puerto Rico excise tax levied on gross inventory purchases to an income-based tax beginning in 2023. The company completed the transition requirements of the Puerto Rico Act in 2022, resulting in the remeasurement of certain deferred tax assets and liabilities based on income tax rates at which they are expected to reverse in the future. The net tax benefit from the remeasurement of deferred taxes related to the Puerto Rico Act was $323 million.
FINANCIAL POSITION, LIQUIDITY AND CAPITAL RESOURCES | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Cash flows provided by (used in) | | | | | |
| Operating activities | $ | 24,943 | | | $ | 22,777 | | | $ | 17,588 | |
| Investing activities | (623) | | | (2,344) | | | (37,557) | |
| Financing activities | (24,803) | | | (19,039) | | | (11,501) | |
Operating cash flows in 2022 increased from 2021 primarily due to improved results of operations resulting from revenue growth and lower income tax payments, partially offset by the timing of working capital. Operating cash flows also reflected AbbVie’s contributions to its defined benefit plans of $357 million in 2022 and $376 million in 2021.
| | | | | | | | | | | |
41 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Investing cash flows in 2022 included payments made for capital expenditures of $695 million, other acquisitions and investments of $539 million, $255 million cash consideration paid to acquire DJS Antibodies Ltd offset by cash acquired and net sales and maturities of investments securities totaling $92 million. Investment cash flows in 2021 included $535 million cash consideration paid to acquire Soliton, Inc. offset by cash acquired, payments made for other acquisitions and investments of $1.4 billion, capital expenditures of $787 million and net purchases of investment securities totaling $21 million.
Financing cash flows in 2022 included repayment of $3.1 billion aggregate principal amount of the company's 2.9% senior notes, $3.0 billion aggregate principal amount of the company's 2.3% senior notes, $2.9 billion aggregate principal amount of the company's 3.45% senior notes, $1.7 billion aggregate principal amount of the company's 3.25% senior notes, $1.0 billion aggregate principal amount of the company’s 3.2% senior notes and $750 million aggregate principal amount of the company's floating rate senior notes. Additionally financing cash flows included repayment of a $2.0 billion floating term loan due May 2025 and issuance of a new $2.0 billion floating rate term loan as part of the term loan refinancing in February 2022. Subsequent to December 31, 2022, the company repaid a $1.0 billion floating rate three-year term loan that was scheduled to mature in May 2023.
Financing cash flows in 2021 included early repayments of $1.8 billion aggregate principal amount of the company's 2.3% principal notes, $1.2 billion aggregate principal amount of the company's 5.0% senior notes and €750 million aggregate principal amount of the company's 0.5% senior Euro notes. Financing cash flows also included repayment of $750 million aggregate principal amount of floating rate senior notes, $1.3 billion aggregate principal amount of 3.375% senior notes, $1.8 billion aggregate principal amount of 2.15% senior notes and $750 million aggregate principal amount of floating rate senior notes at maturity. Additionally, financing cash flows included repayment of a $1.0 billion floating rate term loan due May 2023 and issuance of a new $1.0 billion floating rate term loan as part of the term loan refinancing in September 2021.
Financing cash flows also included cash dividend payments of $10.0 billion in 2022 and $9.3 billion in 2021. The increase in cash dividend payments was primarily driven by an increase of the dividend rate.
The company's stock repurchase authorization permits purchases of AbbVie shares from time to time in open-market or private transactions at management’s discretion. The program has no time limit and can be discontinued at any time. AbbVie repurchased 8 million shares for $1.1 billion in 2022 and 6 million shares for $670 million in 2021. AbbVie's remaining stock repurchase authorization was $1.4 billion as of December 31, 2022. On February 16, 2023, AbbVie's board of directors authorized a $5.0 billion increase to the existing stock repurchase authorization.
No commercial paper borrowings were issued during 2022 or 2021 and there were no commercial paper borrowings outstanding as of December 31, 2022 or December 31, 2021. AbbVie may issue additional commercial paper or retire commercial paper to meet liquidity requirements as needed.
Credit Risk
AbbVie monitors economic conditions, the creditworthiness of customers and government regulations and funding, both domestically and abroad. AbbVie regularly communicates with its customers regarding the status of receivable balances, including their payment plans and obtains positive confirmation of the validity of the receivables. AbbVie establishes an allowance for credit losses equal to the estimate of future losses over the contractual life of outstanding accounts receivable. AbbVie may also utilize factoring arrangements to mitigate credit risk, although the receivables included in such arrangements have historically not been a significant amount of total outstanding receivables.
Credit Facility, Access to Capital and Credit Ratings
Credit Facility
AbbVie currently has a $4.0 billion five-year revolving credit facility that matures in August 2024. This credit facility enables the company to borrow funds on an unsecured basis at variable interest rates and contains various covenants. At December 31, 2022, the company was in compliance with all covenants, and commitment fees under the credit facility were insignificant. No amounts were outstanding under the company's credit facility as of December 31, 2022 and December 31, 2021.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 42 |
Access to Capital
The company intends to fund short-term and long-term financial obligations as they mature through cash on hand, future cash flows from operations or has the ability to issue additional debt. The company's ability to generate cash flows from operations, issue debt or enter into financing arrangements on acceptable terms could be adversely affected if there is a material decline in the demand for the company's products or in the solvency of its customers or suppliers, deterioration in the company's key financial ratios or credit ratings, or other material unfavorable changes in business conditions. At the current time, the company believes it has sufficient financial flexibility to issue debt, enter into other financing arrangements and attract long-term capital on acceptable terms to support the company's growth objectives.
Credit Ratings
In 2022, Moody’s Investors Service upgraded AbbVie's senior unsecured long-term credit rating to Baa1 from Baa2, affirmed its Prime-2 short-term credit rating and revised its outlook to positive from stable. In addition, Standard and Poor's Global Ratings revised its outlook to positive from stable and affirmed its long-term issuer credit rating of BBB+.
Unfavorable changes to the ratings may have an adverse impact on future financing arrangements; however, they would not affect the company’s ability to draw on its credit facility and would not result in an acceleration of scheduled maturities of any of the company’s outstanding debt.
Future Cash Requirements
Contractual Obligations
The following table summarizes AbbVie's estimated material contractual obligations as of December 31, 2022: | | | | | | | | | | | | | | | | | |
| (in millions) | Total | | Current | | Long-term |
| Long-term debt, including current portion | $ | 63,128 | | | $ | 4,132 | | | $ | 58,996 | |
Interest on long-term debt(a) | 28,445 | | | 2,363 | | | 26,082 | |
Contingent consideration liabilities(b) | 16,384 | | | 1,469 | | | 14,915 | |
(a)Includes estimated future interest payments on long-term debt. Interest payments on debt are calculated for future periods using forecasted interest rates in effect at the end of 2022. Projected interest payments include the related effects of interest rate swap agreements. Certain of these projected interest payments may differ in the future based on changes in floating interest rates or other factors or events. The projected interest payments only pertain to obligations and agreements outstanding at December 31, 2022. See Note 10 to the Consolidated Financial Statements for additional information regarding the company's debt instruments and Note 11 for additional information on the interest rate swap agreements outstanding at December 31, 2022.
(b)Includes contingent consideration liabilities which are recorded at fair value on the consolidated balance sheet. Potential contingent consideration payments that exceed the fair value recorded on the consolidated balance sheet are not included in the table of contractual obligations. See Note 11 to the Consolidated Financial Statements for additional information regarding these liabilities.
AbbVie enters into certain unconditional purchase obligations and other commitments in the normal course of business. There have been no changes to these commitments that would have a material impact on the company’s ability to meet either short-term or long-term future cash requirements.
Income Taxes
Future income tax cash requirements include a one-time transition tax liability on a mandatory deemed repatriation of previously untaxed earnings of foreign subsidiaries resulting from U.S. tax reform enacted in 2017. The one-time transition tax liability was $3.4 billion as of December 31, 2022 and is payable in four future annual installments.
Liabilities for unrecognized tax benefits totaled $6.5 billion as of December 31, 2022. It is not possible to reliably estimate the timing of the future cash outflows related to these liabilities. See Note 14 to the Consolidated Financial Statements for additional information on these unrecognized tax benefits.
Quarterly Cash Dividend
On October 28, 2022, AbbVie announced that its board of directors declared an increase in the quarterly cash dividend from $1.41 per share to $1.48 per share beginning with the dividend payable on February 15, 2023, to stockholders of record as of January 13, 2023. This reflects an increase of approximately 5.0% over the previous quarterly rate. The timing,
| | | | | | | | | | | |
43 |  | 2022 Form 10-K | 2022 Form 10-K | | |
declaration, amount of and payment of any dividends by AbbVie in the future is within the discretion of its board of directors and will depend upon many factors, including AbbVie's financial condition, earnings, capital requirements of its operating subsidiaries, covenants associated with certain of AbbVie's debt service obligations, legal requirements, regulatory constraints, industry practice, ability to access capital markets and other factors deemed relevant by its board of directors.
Collaborations, Licensing and Other Arrangements
AbbVie enters into collaborative, licensing and other arrangements with third parties that may require future milestone payments to third parties contingent upon the achievement of certain development, regulatory, or commercial milestones. Individually, these arrangements are insignificant in any one annual reporting period. However, if milestones for multiple products covered by these arrangements happen to be reached in the same reporting period, the aggregate charge to expense could be material to the results of operations in that period. From a business perspective, the payments are viewed as positive because they signify that the product is successfully moving through development and is now generating or is more likely to generate future cash flows from product sales. It is not possible to predict with reasonable certainty whether these milestones will be achieved or the timing for achievement. See Note 5 to the Consolidated Financial Statements for additional information on these collaboration arrangements.
CRITICAL ACCOUNTING POLICIES AND ESTIMATES
The preparation of financial statements in accordance with generally accepted accounting principles in the United States requires the use of estimates and assumptions that affect the reported amounts of assets and liabilities and the reported amounts of revenue and expenses. A summary of the company's significant accounting policies is included in Note 2 to the Consolidated Financial Statements. Certain of these policies are considered critical as these most significantly impact the company's financial condition and results of operations and require the most difficult, subjective, or complex judgments, often as a result of the need to make estimates about the effect of matters that are inherently uncertain. Actual results may vary from these estimates.
Revenue Recognition
AbbVie recognizes revenue when control of promised goods or services is transferred to the company’s customers, in an amount that reflects the consideration AbbVie expects to be entitled to in exchange for those goods or services. Sales, value add and other taxes collected concurrent with revenue-producing activities are excluded from revenue. AbbVie generates revenue primarily from product sales. For the majority of sales, the company transfers control, invoices the customer and recognizes revenue upon shipment to the customer.
Rebates
AbbVie provides rebates to pharmacy benefit managers, state government Medicaid programs, insurance companies that administer Medicare drug plans, wholesalers, group purchasing organizations and other government agencies and private entities.
Rebate and chargeback accruals are accounted for as variable consideration and are recorded as a reduction to revenue in the period the related product is sold. Provisions for rebates and chargebacks totaled $41.4 billion in 2022, $33.9 billion in 2021 and $27.0 billion in 2020. Rebate amounts are typically based upon the volume of purchases using contractual or statutory prices, which may vary by product and by payer. For each type of rebate, the factors used in the calculations of the accrual for that rebate include the identification of the products subject to the rebate, the applicable price terms and the estimated lag time between sale and payment of the rebate, which can be significant.
In order to establish its rebate and chargeback accruals, the company uses both internal and external data to estimate the level of inventory in the distribution channel and the rebate claims processing lag time for each type of rebate. To estimate the rebate percentage or net price, the company tracks sales by product and by customer or payer. The company evaluates inventory data reported by wholesalers, available prescription volume information, product pricing, historical experience and other factors in order to determine the adequacy of its reserves. AbbVie regularly monitors its reserves and records adjustments when rebate trends, rebate programs and contract terms, legislative changes, or other significant events indicate that a change in the reserve is appropriate. Historically, adjustments to rebate accruals have not been material to net earnings.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 44 |
The following table is an analysis of the three largest accruals for rebates and chargebacks, which comprise approximately 94% of the total consolidated rebate and chargebacks recorded as reductions to revenues in 2022. Remaining rebate provisions charged against gross revenues are not significant in the determination of operating earnings. | | | | | | | | | | | | | | | | | |
| (in millions) | Medicaid
and
Medicare
Rebates | | Managed
Care
Rebates | | Wholesaler
Chargebacks |
| Balance at December 31, 2019 | $ | 1,765 | | | $ | 1,936 | | | $ | 686 | |
Additions(a) | 1,266 | | | 649 | | | 71 | |
| Provisions | 6,715 | | | 8,656 | | | 8,677 | |
| Payments | (6,801) | | | (8,334) | | | (8,693) | |
| Balance at December 31, 2020 | 2,945 | | | 2,907 | | | 741 | |
| Provisions | 9,622 | | | 11,306 | | | 11,286 | |
| Payments | (8,751) | | | (11,116) | | | (11,125) | |
| Balance at December 31, 2021 | 3,816 | | | 3,097 | | | 902 | |
| Provisions | 11,713 | | | 14,119 | | | 13,070 | |
| Payments | (10,331) | | | (12,974) | | | (12,829) | |
| Balance at December 31, 2022 | $ | 5,198 | | | $ | 4,242 | | | $ | 1,143 | |
(a)Represents rebate accruals and chargeback allowances assumed in the Allergan acquisition.
Other Allowances
Other allowances include cash discounts, product returns, sales incentives, and other adjustments, which are accounted for as variable consideration and are recorded as a reduction to revenue in the same period the related product is sold. Reserves for cash discounts and sales incentives are readily determinable because the company's experience of payment history is fairly consistent. Product returns can be reliably estimated based on the company's historical return experience. Cash discounts totaled $1.8 billion in 2022, $1.6 billion in 2021 and $1.2 billion in 2020. Allowances other than cash discounts are not significant.
Pension and Other Post-Employment Benefits
AbbVie engages outside actuaries to assist in the determination of the obligations and costs under the pension and other post-employment benefit plans that are direct obligations of AbbVie. The valuation of the funded status and the net periodic benefit cost for these plans are calculated using actuarial assumptions. The significant assumptions, which are reviewed annually, include the discount rate, the expected long-term rate of return on plan assets and the health care cost trend rates and are disclosed in Note 12 to the Consolidated Financial Statements.
The discount rate is selected based on current market rates on high-quality, fixed-income investments at December 31 each year. AbbVie employs a yield-curve approach for countries where a robust bond market exists. The yield curve is developed using high-quality bonds. The yield-curve approach reflects the plans' specific cash flows (i.e. duration) in calculating the benefit obligations by applying the corresponding individual spot rates along the yield curve. AbbVie reflects the plans' specific cash flows and applies them to the corresponding individual spot rates along the yield curve in calculating the service cost and interest cost portions of expense. For other countries, AbbVie reviews various indices such as corporate bond and government bond benchmarks to estimate the discount rate.
| | | | | | | | | | | |
45 |  | 2022 Form 10-K | 2022 Form 10-K | | |
AbbVie's assumed discount rates have a significant effect on the amounts reported for defined benefit pension and other post-employment plans as of December 31, 2022. A 50 basis point change in the assumed discount rate would have had the following effects on AbbVie's calculation of net periodic benefit costs in 2023 and projected benefit obligations as of December 31, 2022: | | | | | | | | | | | |
| 50 basis point |
| (in millions) (brackets denote a reduction) | Increase | | Decrease |
| Defined benefit plans | | | |
| Net periodic benefit cost | $ | (34) | | | $ | 57 | |
| Projected benefit obligation | (612) | | | 687 | |
| Other post-employment plans | | | |
| Net periodic benefit cost | $ | (5) | | | $ | 6 | |
| Projected benefit obligation | (44) | | | 49 | |
The expected long-term rate of return is based on the asset allocation, historical performance and the current view of expected future returns. AbbVie considers these inputs with a long-term focus to avoid short-term market influences. The current long-term rate of return on plan assets for each plan is supported by the historical performance of the trust's actual and target asset allocation. AbbVie's assumed expected long-term rate of return has a significant effect on the amounts reported for defined benefit pension plans as of December 31, 2022 and will be used in the calculation of net periodic benefit cost in 2023. A one percentage point change in assumed expected long-term rate of return on plan assets would increase or decrease the net period benefit cost of these plans in 2023 by $98 million.
The health care cost trend rate is selected by reviewing historical trends and current views on projected future health care cost increases. The current health care cost trend rate is supported by the historical trend experience of each plan. Assumed health care cost trend rates have a significant effect on the amounts reported for health care plans as of December 31, 2022 and will be used in the calculation of net periodic benefit cost in 2023.
Income Taxes
AbbVie accounts for income taxes under the asset and liability method. Provisions for federal, state and foreign income taxes are calculated on reported pre-tax earnings based on current tax laws. Deferred taxes are provided using enacted tax rates on the future tax consequences of temporary differences, which are the differences between the financial statement carrying amount of assets and liabilities and their respective tax bases and the tax benefits of carryforwards. A valuation allowance is established or maintained when, based on currently available information, it is more likely than not that all or a portion of a deferred tax asset will not be realized.
Litigation
The company is subject to contingencies, such as various claims, legal proceedings and investigations regarding product liability, intellectual property, commercial, securities and other matters that arise in the normal course of business. See Note 15 to the Consolidated Financial Statements for additional information. Loss contingency provisions are recorded for probable losses at management's best estimate of a loss, or when a best estimate cannot be made, a minimum loss contingency amount within a probable range is recorded. Accordingly, AbbVie is often initially unable to develop a best estimate of loss and therefore, the minimum amount, which could be zero, is recorded. As information becomes known, either the minimum loss amount is increased, resulting in additional loss provisions, or a best estimate can be made, also resulting in additional loss provisions. Occasionally, a best estimate amount is changed to a lower amount when events result in an expectation of a more favorable outcome than previously expected.
Valuation of Goodwill and Intangible Assets
AbbVie has acquired and may continue to acquire significant intangible assets in connection with business combinations that AbbVie records at fair value. Transactions involving the purchase or sale of intangible assets occur between companies in the pharmaceuticals industry and valuations are usually based on a discounted cash flow analysis incorporating the stage of completion. The discounted cash flow model requires assumptions about the timing and amount of future net cash flows, risk, cost of capital, terminal values and market participants. Each of these factors can significantly affect the value of the intangible asset. In-process research and development (IPR&D) acquired in a business combination is capitalized as an indefinite-lived intangible asset until regulatory approval is obtained, at which time it is accounted for as a definite-lived asset and amortized over its estimated useful life, or discontinuation, at which point the intangible asset will be written off. IPR&D acquired in transactions that are not business combinations is expensed immediately, unless deemed to have an alternative
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 46 |
future use. Payments made to third parties subsequent to regulatory approval are capitalized and amortized over the remaining useful life.
AbbVie reviews the recoverability of definite-lived intangible assets whenever events or changes in circumstances indicate the carrying value of an asset may not be recoverable. Goodwill and indefinite-lived intangible assets are reviewed for impairment annually or when an event occurs that could result in an impairment. See Note 2 to the Consolidated Financial Statements for additional information.
Annually, the company tests its goodwill for impairment by first assessing qualitative factors to determine whether it is more likely than not that the fair value is less than its carrying amount. Some of the factors considered in the assessment include general macro-economic conditions, conditions specific to the industry and market, cost factors, the overall financial performance and whether there have been sustained declines in the company's share price. If the company concludes it is more likely than not that the fair value of the reporting unit is less than its carrying amount, a quantitative impairment test is performed. AbbVie tests indefinite-lived intangible assets for impairment by first assessing qualitative factors to determine whether it is more likely than not that the fair value is less than its carrying amount. If the company concludes it is more likely than not that the fair value is less than its carrying amount, a quantitative impairment test is performed.
For its quantitative impairment tests, the company uses an estimated future cash flow approach that requires significant judgment with respect to future volume, revenue and expense growth rates, changes in working capital use, the selection of an appropriate discount rate, asset groupings and other assumptions and estimates. The estimates and assumptions used are consistent with the company's business plans and a market participant's views. The use of alternative estimates and assumptions could increase or decrease projected cash flows and the estimated fair value of the related intangible assets. Future changes to these estimates and assumptions could have a material impact on the company's results of operations. Actual results may differ from the company's estimates.
Contingent Consideration
The fair value measurements of contingent consideration liabilities are determined as of the acquisition date based on significant unobservable inputs, including the discount rate, estimated probabilities and timing of achieving specified development, regulatory and commercial milestones and the estimated amount of future sales of the acquired products. Contingent consideration liabilities are revalued to fair value at each subsequent reporting date until the related contingency is resolved. The potential contingent consideration payments are estimated by applying a probability-weighted expected payment model for contingent milestone payments and a Monte Carlo simulation model for contingent royalty payments, which are then discounted to present value. Changes to the fair value of the contingent consideration liabilities can result from changes to one or a number of inputs, including discount rates, the probabilities of achieving the milestones, the time required to achieve the milestones and estimated future sales. Significant judgment is employed in determining the appropriateness of certain of these inputs, which are disclosed in Note 11 to the Consolidated Financial Statements. Changes to the inputs described above could have a material impact on the company's financial position and results of operations in any given period.
| | | | | | | | | | | |
47 |  | 2022 Form 10-K | 2022 Form 10-K | | |
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK The company is exposed to risk that its earnings, cash flows and equity could be adversely impacted by changes in foreign exchange rates and interest rates. Certain derivative instruments are used when available on a cost-effective basis to hedge the company's underlying economic exposures. See Note 11 to the Consolidated Financial Statements for additional information regarding the company's financial instruments and hedging strategies.
Foreign Currency Risk
AbbVie's primary net foreign currency exposures are the Euro, Japanese yen, Canadian dollar, Chinese yuan and British pound. The following table reflects the total foreign currency forward exchange contracts outstanding at December 31, 2022 and 2021: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 2022 | | 2021 |
| as of December 31 (in millions) | Contract amount | | Weighted average exchange rate | | Fair and carrying value receivable/(payable) | | Contract amount | | Weighted average exchange rate | | Fair and carrying value receivable/(payable) |
| Receive primarily U.S. dollars in exchange for the following currencies: | | | | | | | | | | | |
| Euro | $ | 8,507 | | | 1.071 | | | $ | 9 | | | $ | 10,253 | | | 1.155 | | | $ | 195 | |
| Canadian dollar | 1,302 | | | 1.312 | | | 40 | | | 571 | | | 1.258 | | | 9 | |
| British pound | 772 | | | 1.234 | | | (8) | | | 605 | | | 1.331 | | | 9 | |
| Chinese yuan | 596 | | | 7.024 | | | (5) | | | 673 | | | 6.400 | | | (1) | |
| Japanese yen | 567 | | | 133.3 | | | (3) | | | 602 | | | 113.3 | | | 9 | |
| All other currencies | 1,954 | | | n/a | | (2) | | | 1,549 | | | n/a | | 5 | |
| Total | $ | 13,698 | | | | | $ | 31 | | | $ | 14,253 | | | | | $ | 226 | |
The company estimates that a 10% appreciation in the underlying currencies being hedged from their levels against the U.S. dollar, with all other variables held constant, would decrease the fair value of foreign exchange forward contracts by $1.4 billion at December 31, 2022. If realized, this appreciation would negatively affect earnings over the remaining life of the contracts. However, gains and losses on the hedging instruments offset losses and gains on the hedged transactions and reduce the earnings and stockholders' equity volatility relating to foreign exchange. A 10% appreciation is believed to be a reasonably possible near-term change in foreign currencies.
As of December 31, 2022, the company has €5.9 billion aggregate principal amount of unsecured senior Euro notes outstanding, which are exposed to foreign currency risk. The company designated these foreign currency denominated notes as hedges of its net investments in certain foreign subsidiaries and affiliates. As a result, any foreign currency translation gains or losses related to the Euro notes will be included in accumulated other comprehensive loss. See Note 10 to the Consolidated Financial Statements for additional information regarding the senior Euro notes and Note 11 to the Consolidated Financial Statements for additional information regarding the net investment hedging program.
Interest Rate Risk
The company estimates that an increase in interest rates of 100 basis points would adversely impact the fair value of AbbVie's interest rate swap contracts by approximately $155 million at December 31, 2022. If realized, the fair value reduction would affect earnings over the remaining life of the contracts. The company estimates that an increase of 100 basis points in long-term interest rates would decrease the fair value of long-term debt by $4.2 billion at December 31, 2022. A 100 basis point change is believed to be a reasonably possible near-term change in interest rates.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 48 |
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA | | | | | |
| | Page |
| Consolidated Financial Statements | |
| |
| |
| |
| |
| |
| | | | | |
| Notes to Consolidated Financial Statements | |
| | | | | | | | | | | |
49 |  | 2022 Form 10-K | 2022 Form 10-K | | |
AbbVie Inc. and Subsidiaries
Consolidated Statements of Earnings
| | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions, except per share data) | 2022 | | 2021 | | 2020 |
| Net revenues | $ | 58,054 | | | $ | 56,197 | | | $ | 45,804 | |
| | | | | |
| Cost of products sold | 17,414 | | | 17,446 | | | 15,387 | |
| Selling, general and administrative | 15,260 | | | 12,349 | | | 11,299 | |
| Research and development | 6,510 | | | 6,922 | | | 6,379 | |
| Acquired IPR&D and milestones | 697 | | | 1,124 | | | 1,376 | |
| Other operating expense, net | 56 | | | 432 | | | — | |
| Total operating costs and expenses | 39,937 | | | 38,273 | | | 34,441 | |
| Operating earnings | 18,117 | | | 17,924 | | | 11,363 | |
| | | | | |
| Interest expense, net | 2,044 | | | 2,384 | | | 2,280 | |
| Net foreign exchange loss | 148 | | | 51 | | | 71 | |
| Other expense, net | 2,448 | | | 2,500 | | | 5,614 | |
| Earnings before income tax expense | 13,477 | | | 12,989 | | | 3,398 | |
| Income tax expense (benefit) | 1,632 | | | 1,440 | | | (1,224) | |
| Net earnings | 11,845 | | | 11,549 | | | 4,622 | |
| Net earnings attributable to noncontrolling interest | 9 | | | 7 | | | 6 | |
| Net earnings attributable to AbbVie Inc. | $ | 11,836 | | | $ | 11,542 | | | $ | 4,616 | |
| | | | | |
| Per share data | | | | | |
| Basic earnings per share attributable to AbbVie Inc. | $ | 6.65 | | | $ | 6.48 | | | $ | 2.73 | |
| Diluted earnings per share attributable to AbbVie Inc. | $ | 6.63 | | | $ | 6.45 | | | $ | 2.72 | |
| | | | | |
| Weighted-average basic shares outstanding | 1,771 | | | 1,770 | | | 1,667 | |
| Weighted-average diluted shares outstanding | 1,778 | | | 1,777 | | | 1,673 | |
The accompanying notes are an integral part of these consolidated financial statements.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 50 |
AbbVie Inc. and Subsidiaries
Consolidated Statements of Comprehensive Income
| | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Net earnings | $ | 11,845 | | | $ | 11,549 | | | $ | 4,622 | |
| | | | | |
Foreign currency translation adjustments, net of tax expense (benefit) of $(10) in 2022, $(35) in 2021 and $28 in 2020 | (943) | | | (1,153) | | | 1,511 | |
Net investment hedging activities, net of tax expense (benefit) of $152 in 2022, $193 in 2021 and $(221) in 2020 | 555 | | | 699 | | | (799) | |
Pension and post-employment benefits, net of tax expense (benefit) of $272 in 2022, $124 in 2021 and $(47) in 2020 | 1,088 | | | 521 | | | (102) | |
| | | | | |
Cash flow hedging activities, net of tax expense (benefit) of $5 in 2022, $20 in 2021 and $(23) in 2020 | — | | | 151 | | | (131) | |
| Other comprehensive income | $ | 700 | | | $ | 218 | | | $ | 479 | |
| Comprehensive income | 12,545 | | | 11,767 | | | 5,101 | |
| Comprehensive income attributable to noncontrolling interest | 9 | | | 7 | | | 6 | |
| Comprehensive income attributable to AbbVie Inc. | $ | 12,536 | | | $ | 11,760 | | | $ | 5,095 | |
The accompanying notes are an integral part of these consolidated financial statements.
| | | | | | | | | | | |
51 |  | 2022 Form 10-K | 2022 Form 10-K | | |
AbbVie Inc. and Subsidiaries
Consolidated Balance Sheets
| | | | | | | | | | | |
| as of December 31 (in millions, except share data) | 2022 | | 2021 |
| Assets | | | |
| Current assets | | | |
| Cash and equivalents | $ | 9,201 | | | $ | 9,746 | |
| Short-term investments | 28 | | | 84 | |
| Accounts receivable, net | 11,254 | | | 9,977 | |
| Inventories | 3,579 | | | 3,128 | |
| Prepaid expenses and other | 4,401 | | | 4,993 | |
| Total current assets | 28,463 | | | 27,928 | |
| | | |
| Investments | 241 | | | 277 | |
| Property and equipment, net | 4,935 | | | 5,110 | |
| Intangible assets, net | 67,439 | | | 75,951 | |
| Goodwill | 32,156 | | | 32,379 | |
| Other assets | 5,571 | | | 4,884 | |
| Total assets | $ | 138,805 | | | $ | 146,529 | |
| | | |
| Liabilities and Equity | | | |
| Current liabilities | | | |
| Short-term borrowings | $ | 1 | | | $ | 14 | |
| Current portion of long-term debt and finance lease obligations | 4,135 | | | 12,481 | |
| Accounts payable and accrued liabilities | 25,402 | | | 22,699 | |
| Total current liabilities | 29,538 | | | 35,194 | |
| | | |
| Long-term debt and finance lease obligations | 59,135 | | | 64,189 | |
| Deferred income taxes | 2,190 | | | 3,009 | |
| Other long-term liabilities | 30,655 | | | 28,701 | |
| | | |
| Commitments and contingencies | | | |
| | | |
| Stockholders' equity | | | |
Common stock, $0.01 par value, 4,000,000,000 shares authorized, 1,813,770,294 shares issued as of December 31, 2022 and 1,803,195,293 as of December 31, 2021 | 18 | | | 18 | |
Common stock held in treasury, at cost, 44,589,000 shares as of December 31, 2022 and 34,857,597 as of December 31, 2021 | (4,594) | | | (3,143) | |
| Additional paid-in capital | 19,245 | | | 18,305 | |
| Retained earnings | 4,784 | | | 3,127 | |
| Accumulated other comprehensive loss | (2,199) | | | (2,899) | |
| Total stockholders' equity | 17,254 | | | 15,408 | |
| Noncontrolling interest | 33 | | | 28 | |
| Total equity | 17,287 | | | 15,436 | |
| | | |
| Total liabilities and equity | $ | 138,805 | | | $ | 146,529 | |
The accompanying notes are an integral part of these consolidated financial statements.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 52 |
AbbVie Inc. and Subsidiaries
Consolidated Statements of Equity
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | Common shares outstanding | | Common stock | | Treasury stock | | Additional paid-in capital | | Retained earnings | | Accumulated other comprehensive loss | | Noncontrolling interest | | Total |
| Balance at December 31, 2019 | 1,479 | | | $ | 18 | | | $ | (24,504) | | | $ | 15,193 | | | $ | 4,717 | | | $ | (3,596) | | | $ | — | | | $ | (8,172) | |
| Net earnings attributable to AbbVie Inc. | — | | | — | | | — | | | — | | | 4,616 | | | — | | | — | | | 4,616 | |
| Other comprehensive income, net of tax | — | | | — | | | — | | | — | | | — | | | 479 | | | — | | | 479 | |
| Dividends declared | — | | | — | | | — | | | — | | | (8,278) | | | — | | | — | | | (8,278) | |
| Common shares and equity awards issued for acquisition of Allergan plc | 286 | | | — | | | 23,166 | | | 1,243 | | | — | | | — | | | — | | | 24,409 | |
| Purchases of treasury stock | (10) | | | — | | | (978) | | | — | | | — | | | — | | | — | | | (978) | |
| Stock-based compensation plans and other | 10 | | | — | | | 52 | | | 948 | | | — | | | — | | | — | | | 1,000 | |
| Change in noncontrolling interest | — | | | — | | | — | | | — | | | — | | | — | | | 21 | | | 21 | |
| Balance at December 31, 2020 | 1,765 | | | 18 | | | (2,264) | | | 17,384 | | | 1,055 | | | (3,117) | | | 21 | | | 13,097 | |
| Net earnings attributable to AbbVie Inc. | — | | | — | | | — | | | — | | | 11,542 | | | — | | | — | | | 11,542 | |
| Other comprehensive income, net of tax | — | | | — | | | — | | | — | | | — | | | 218 | | | — | | | 218 | |
| Dividends declared | — | | | — | | | — | | | — | | | (9,470) | | | — | | | — | | | (9,470) | |
| Purchases of treasury stock | (8) | | | — | | | (934) | | | — | | | — | | | — | | | — | | | (934) | |
| Stock-based compensation plans and other | 11 | | | — | | | 55 | | | 921 | | | — | | | — | | | — | | | 976 | |
| Change in noncontrolling interest | — | | | — | | | — | | | — | | | — | | | — | | | 7 | | | 7 | |
| Balance at December 31, 2021 | 1,768 | | | 18 | | | (3,143) | | | 18,305 | | | 3,127 | | | (2,899) | | | 28 | | | 15,436 | |
| Net earnings attributable to AbbVie Inc. | — | | | — | | | — | | | — | | | 11,836 | | | — | | | — | | | 11,836 | |
| Other comprehensive income, net of tax | — | | | — | | | — | | | — | | | — | | | 700 | | | — | | | 700 | |
| Dividends declared | — | | | — | | | — | | | — | | | (10,179) | | | — | | | — | | | (10,179) | |
| Purchases of treasury stock | (10) | | | — | | | (1,487) | | | — | | | — | | | — | | | — | | | (1,487) | |
| Stock-based compensation plans and other | 11 | | | — | | | 36 | | | 940 | | | — | | | — | | | — | | | 976 | |
| Change in noncontrolling interest | — | | | — | | | — | | | — | | | — | | | — | | | 5 | | | 5 | |
| Balance at December 31, 2022 | 1,769 | | | $ | 18 | | | $ | (4,594) | | | $ | 19,245 | | | $ | 4,784 | | | $ | (2,199) | | | $ | 33 | | | $ | 17,287 | |
The accompanying notes are an integral part of these consolidated financial statements.
| | | | | | | | | | | |
53 |  | 2022 Form 10-K | 2022 Form 10-K | | |
AbbVie Inc. and Subsidiaries
Consolidated Statements of Cash Flows | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) (brackets denote cash outflows) | 2022 | | 2021 | | 2020 |
| Cash flows from operating activities | | | | | |
| Net earnings | $ | 11,845 | | | $ | 11,549 | | | $ | 4,622 | |
| Adjustments to reconcile net earnings to net cash from operating activities: | | | | | |
| Depreciation | 778 | | | 803 | | | 666 | |
| Amortization of intangible assets | 7,689 | | | 7,718 | | | 5,805 | |
| Deferred income taxes | (1,931) | | | (898) | | | (2,325) | |
| Change in fair value of contingent consideration liabilities | 2,761 | | | 2,679 | | | 5,753 | |
| Stock-based compensation | 671 | | | 692 | | | 753 | |
| Acquired IPR&D and milestones | 697 | | | 1,124 | | | 1,376 | |
| Other charges related to collaborations | — | | | 500 | | | — | |
| Gain on divestitures | (172) | | | (68) | | | — | |
| Non-cash litigation reserve adjustments, net of cash payments | 2,243 | | | 163 | | | (31) | |
| Impairment of intangible assets | 770 | | | 50 | | | — | |
| Other, net | (150) | | | (213) | | | 863 | |
| Changes in operating assets and liabilities, net of acquisitions: | | | | | |
| Accounts receivable | (1,455) | | | (1,321) | | | (929) | |
| Inventories | (686) | | | (142) | | | (40) | |
| Prepaid expenses and other assets | (264) | | | (197) | | | 134 | |
| Accounts payable and other liabilities | 1,605 | | | 1,628 | | | 1,514 | |
| Income tax assets and liabilities, net | 542 | | | (1,290) | | | (573) | |
| Cash flows from operating activities | 24,943 | | | 22,777 | | | 17,588 | |
| | | | | |
| Cash flows from investing activities | | | | | |
| Acquisition of businesses, net of cash acquired | (255) | | | (525) | | | (38,260) | |
| Other acquisitions and investments | (539) | | | (1,377) | | | (1,350) | |
| Acquisitions of property and equipment | (695) | | | (787) | | | (798) | |
| Purchases of investment securities | (1,438) | | | (119) | | | (61) | |
| Sales and maturities of investment securities | 1,530 | | | 98 | | | 1,525 | |
| Other, net | 774 | | | 366 | | | 1,387 | |
| Cash flows from investing activities | (623) | | | (2,344) | | | (37,557) | |
| | | | | |
| Cash flows from financing activities | | | | | |
| Proceeds from issuance of long-term debt | 2,000 | | | 1,000 | | | 3,000 | |
| Repayments of long-term debt and finance lease obligations | (14,433) | | | (9,414) | | | (5,683) | |
| Debt issuance costs | — | | | — | | | (20) | |
| Dividends paid | (10,043) | | | (9,261) | | | (7,716) | |
| Purchases of treasury stock | (1,487) | | | (934) | | | (978) | |
| Proceeds from the exercise of stock options | 262 | | | 244 | | | 209 | |
| Payments of contingent consideration liabilities | (1,132) | | | (698) | | | (321) | |
| Other, net | 30 | | | 24 | | | 8 | |
| Cash flows from financing activities | (24,803) | | | (19,039) | | | (11,501) | |
| Effect of exchange rate changes on cash and equivalents | (62) | | | (97) | | | (5) | |
| Net change in cash and equivalents | (545) | | | 1,297 | | | (31,475) | |
| Cash and equivalents, beginning of year | 9,746 | | | 8,449 | | | 39,924 | |
| | | | | |
| Cash and equivalents, end of year | $ | 9,201 | | | $ | 9,746 | | | $ | 8,449 | |
| | | | | |
| Other supplemental information | | | | | |
| Interest paid, net of portion capitalized | $ | 2,546 | | | $ | 2,712 | | | $ | 2,619 | |
| Income taxes paid | 2,988 | | | 3,648 | | | 1,674 | |
| Supplemental schedule of non-cash investing and financing activities | | | | | |
| Issuance of common shares associated with acquisitions of businesses | — | | | — | | | 23,979 | |
The accompanying notes are an integral part of these consolidated financial statements.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 54 |
AbbVie Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Background
The principal business of AbbVie Inc. (AbbVie or the company) is the discovery, development, manufacturing and sale of a broad line of therapies that address some of the world's most complex and serious diseases. AbbVie's products are generally sold worldwide directly to wholesalers, distributors, government agencies, health care facilities, specialty pharmacies and independent retailers from AbbVie-owned distribution centers and public warehouses. Certain products (including aesthetic products and devices) are also sold directly to physicians and other licensed healthcare providers. In the United States, AbbVie distributes pharmaceutical products principally through independent wholesale distributors, with some sales directly to retailers, pharmacies, patients or other customers. Outside the United States, AbbVie sells products primarily to wholesalers or through distributors, and depending on the market works through largely centralized national payers system to agree on reimbursement terms.
AbbVie was incorporated in Delaware on April 10, 2012. On January 1, 2013, AbbVie became an independent, publicly-traded company as a result of the distribution by Abbott Laboratories (Abbott) of 100% of the outstanding common stock of AbbVie to Abbott's shareholders.
On May 8, 2020, AbbVie completed its acquisition of Allergan plc (Allergan). Refer to Note 5 for additional information regarding this acquisition.
Note 2 Summary of Significant Accounting Policies Use of Estimates
The consolidated financial statements have been prepared in accordance with U.S. generally accepted accounting principles (GAAP) and necessarily include amounts based on estimates and assumptions by management. Actual results could differ from those amounts. Significant estimates include amounts for rebates, pension and other post-employment benefits, income taxes, litigation, valuation of goodwill and intangible assets, contingent consideration liabilities, financial instruments and inventory and accounts receivable exposures.
Basis of Consolidation
The consolidated financial statements include the accounts of AbbVie and all of its subsidiaries in which a controlling interest is maintained. Controlling interest is determined by majority ownership interest and the absence of substantive third-party participating rights or, in the case of variable interest entities, where AbbVie is determined to be the primary beneficiary. Investments in companies over which AbbVie has a significant influence but not a controlling interest are accounted for using the equity method with AbbVie's share of earnings or losses reported in other expense, net in the consolidated statements of earnings. Intercompany balances and transactions are eliminated.
During 2022, AbbVie revised its classification of development milestone expense associated with licensing and collaboration arrangements in the consolidated statements of earnings. Milestone payments incurred prior to regulatory approval, which were previously included in research and development (R&D) expense, are now presented as acquired IPR&D and milestones expense in the consolidated statements of earnings. The reclassification decreased R&D expense and increased acquired IPR&D and milestones expense by $162 million in 2021 and $178 million in 2020. The company believes this presentation assists users of the financial statements to better understand the total upfront and subsequent development milestone payments incurred to acquire in-process research and development projects. Prior periods have been reclassified to conform to the current period presentation. Certain other reclassifications have been made to conform the prior period consolidated financial statements to the current period presentation.
Revenue Recognition
AbbVie recognizes revenue when control of promised goods or services is transferred to the company’s customers, in an amount that reflects the consideration AbbVie expects to be entitled to in exchange for those goods or services. Sales, value add and other taxes collected concurrent with revenue-producing activities are excluded from revenue. AbbVie generates revenue primarily from product sales. For the majority of sales, the company transfers control, invoices the customer and recognizes revenue upon shipment to the customer. The company recognizes shipping and handling costs as an
| | | | | | | | | | | |
55 |  | 2022 Form 10-K | 2022 Form 10-K | | |
expense in cost of products sold when the company transfers control to the customer. Payment terms vary depending on the type and location of the customer, are based on customary commercial terms and are generally less than one year. AbbVie does not adjust revenue for the effects of a significant financing component for contracts where AbbVie expects the period between the transfer of the good or service and collection to be one year or less.
Cash discounts, rebates and chargebacks, sales incentives, product returns and certain other adjustments are accounted for as variable consideration. Provisions for variable consideration are based on current pricing, executed contracts, government pricing legislation and historical data and are provided for in the period the related revenues are recorded. Rebate amounts are typically based upon the volume of purchases using contractual or statutory prices, which may vary by product and by payer. For each type of rebate, factors used in the calculation of the accrual include the identification of the products subject to the rebate, the applicable price terms and the estimated lag time between sale and payment of the rebate, which can be significant.
In addition to revenue from contracts with customers, the company also recognizes certain collaboration revenues. See Note 6 for additional information related to the collaborations with Janssen Biotech, Inc. and Genentech, Inc. Additionally, see Note 16 for disaggregation of revenue by product and geography.
Research and Development Expenses
Internal R&D costs are expensed as incurred. Clinical trial costs incurred by third parties are expensed as the contracted work is performed.
Acquired IPR&D and Milestones Expenses
In an asset acquisition, payments incurred prior to regulatory approval to acquire rights to in-process R&D projects are expensed as acquired IPR&D and milestones expense in the consolidated statements of earnings unless the project has an alternative future use. These costs include upfront and development milestone payments related to R&D collaborations, licensing arrangements, or other asset acquisitions that provide rights to develop, manufacture and/or sell pharmaceutical products. Where contingent development milestone payments are due to third parties, prior to regulatory approval, the payment obligations are expensed when the milestone results are achieved. Regulatory and commercial milestone payments made to third parties subsequent to regulatory approval are capitalized as intangible assets and amortized to cost of products sold over the remaining useful life of the related product.
Business Combinations
AbbVie utilizes the acquisition method of accounting for business combinations. This method requires, among other things, that results of operations of acquired companies are included in AbbVie's results of operations beginning on the acquisition date and that assets acquired and liabilities assumed are recognized at fair value as of the acquisition date. Any excess of the fair value of consideration transferred over the fair value of the net assets acquired is recognized as goodwill. Contingent consideration liabilities are recognized at the estimated fair value on the acquisition date. Subsequent changes to the fair value of contingent consideration liabilities are recognized in other expense, net in the consolidated statements of earnings. The fair value of assets acquired and liabilities assumed in certain cases may be subject to revision based on the final determination of fair value during a period of time not to exceed 12 months from the acquisition date. Legal costs, due diligence costs, business valuation costs and all other business acquisition costs are expensed when incurred.
In a business combination, the fair value of IPR&D projects acquired is capitalized and accounted for as indefinite-lived intangible assets until the underlying project receives regulatory approval, at which point the intangible asset will be accounted for as a definite-lived intangible asset, or discontinuation, at which point the intangible asset will be written off. R&D costs incurred by the company after the acquisition are expensed to R&D as incurred.
Collaborations and Other Arrangements
The company enters into collaborative agreements with third parties to develop and commercialize drug candidates. Collaborative activities may include joint research and development and commercialization of new products. AbbVie generally receives certain licensing rights under these arrangements. These collaborations often require upfront payments and may include additional milestone, research and development cost sharing, royalty or profit share payments, contingent upon the occurrence of certain future events linked to the success of the asset in development and commercialization. Upfront payments associated with collaborative arrangements and subsequent payments made to the partner for the achievement of development milestones prior to regulatory approval are expensed to acquired IPR&D and milestones expense in the consolidated statements of earnings. Regulatory and commercial milestone payments made to the partner subsequent to regulatory approval are capitalized as intangible assets and amortized to cost of products sold over the estimated useful life of the related asset. Royalties are expensed to cost of products sold in the consolidated statements of earnings when incurred.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 56 |
Advertising
Costs associated with advertising are expensed as incurred and are included in selling, general and administrative (SG&A) expense in the consolidated statements of earnings. Advertising expenses were $2.0 billion in 2022, $2.1 billion in 2021 and $1.8 billion in 2020.
Pension and Other Post-Employment Benefits
AbbVie records annual expenses relating to its defined benefit pension and other post-employment benefit plans based on calculations which utilize various actuarial assumptions including discount rates, rates of return on assets, compensation increases, turnover rates and health care cost trend rates. AbbVie reviews its actuarial assumptions on an annual basis and makes modifications to the assumptions based on current rates and trends. Actuarial gains and losses are deferred in accumulated other comprehensive income (loss) (AOCI), net of tax and are amortized over the remaining service attribution periods of the employees under the corridor method. Differences between the expected long-term return on plan assets and the actual annual return are generally amortized to net periodic benefit cost over a five-year period.
Income Taxes
Income taxes are accounted for under the asset and liability method. Provisions for federal, state and foreign income taxes are calculated on reported pre-tax earnings based on current tax laws. Deferred taxes are provided using enacted tax rates on the future tax consequences of temporary differences, which are the differences between the financial statement carrying amounts of assets and liabilities and their respective tax bases and the tax benefits of carryforwards. A valuation allowance is established or maintained when, based on currently available information, it is more likely than not that all or a portion of a deferred tax asset will not be realized.
Cash and Equivalents
Cash and equivalents include money market funds and time deposits with original maturities of three months or less.
Investments
Investments consist primarily of equity securities, held-to-maturity debt securities, marketable debt securities and time deposits. Investments in equity securities that have readily determinable fair values are recorded at fair value. Investments in equity securities that do not have readily determinable fair values are recorded at cost and are remeasured to fair value based on certain observable price changes or impairment events as they occur. Held-to-maturity debt securities are recorded at cost. Gains or losses on investments are included in other expense, net in the consolidated statements of earnings. Investments in marketable debt securities are classified as available-for-sale and are recorded at fair value with any unrealized holding gains or losses, net of tax, included in AOCI on the consolidated balance sheets until realized, at which time the gains or losses are recognized in earnings.
AbbVie periodically assesses its marketable debt securities for impairment and credit losses. When a decline in fair value of marketable debt security is due to credit related factors, an allowance for credit losses is recorded with a corresponding charge to other expense, net in the consolidated statements of earnings. When AbbVie determines that a non-credit related impairment has occurred, the amortized cost basis of the investment, net of allowance for credit losses, is written down with a charge to other expense, net in the consolidated statements of earnings and an available-for-sale investment's unrealized loss is reclassified from AOCI to other expense, net in the consolidated statements of earnings. Realized gains and losses on sales of investments are computed using the first-in, first-out method adjusted for any impairments and credit losses that were recorded in net earnings.
Accounts Receivable
Accounts receivable are stated at amortized cost less allowance for credit losses. The allowance for credit losses reflects the best estimate of future losses over the contractual life of outstanding accounts receivable and is determined on the basis of historical experience, specific allowances for known troubled accounts, other currently available information including customer financial condition and both current and forecasted economic conditions.
| | | | | | | | | | | |
57 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Inventories
Inventories are valued at the lower of cost (first-in, first-out basis) or market. Cost includes material and conversion costs. Inventories consisted of the following: | | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| Finished goods | $ | 1,162 | | | $ | 932 | |
| Work-in-process | 1,417 | | | 1,193 | |
| Raw materials | 1,000 | | | 1,003 | |
| Inventories | $ | 3,579 | | | $ | 3,128 | |
Property and Equipment | | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| Land | $ | 286 | | | $ | 287 | |
| Buildings | 2,737 | | | 2,791 | |
| Equipment | 7,107 | | | 6,850 | |
| Construction in progress | 856 | | | 799 | |
| Property and equipment, gross | 10,986 | | | 10,727 | |
| Less accumulated depreciation | (6,051) | | | (5,617) | |
| Property and equipment, net | $ | 4,935 | | | $ | 5,110 | |
Depreciation for property and equipment is recorded on a straight-line basis over the estimated useful lives of the assets. The estimated useful life for buildings ranges from 10 to 50 years. Buildings include leasehold improvements which are amortized over the lesser of the remainder of the lease term or the useful life of the leasehold improvement. The estimated useful life for equipment ranges from 2 to 25 years. Equipment includes certain computer software and software development costs incurred in connection with developing or obtaining software for internal use and is amortized over 3 to 10 years. Depreciation expense was $778 million in 2022, $803 million in 2021 and $666 million in 2020.
Leases
Short-term leases with a term of 12 months or less are not recorded on the balance sheet. For leases commencing or modified in 2019 or later, AbbVie does not separate lease components from non-lease components.
The company records lease liabilities based on the present value of lease payments over the lease term. AbbVie generally uses an incremental borrowing rate to discount its lease liabilities, as the rate implicit in the lease is typically not readily determinable. Certain lease agreements include renewal options that are under the company's control. AbbVie includes optional renewal periods in the lease term only when it is reasonably certain that AbbVie will exercise its option.
Variable lease payments include payments to lessors for taxes, maintenance, insurance and other operating costs as well as payments that are adjusted based on an index or rate. The company's lease agreements do not contain any significant residual value guarantees or restrictive covenants.
Litigation and Contingencies
Loss contingency provisions are recorded when it is probable that a liability has been incurred and the amount of the liability can be reasonably estimated based on existing information. When a best estimate cannot be made, the minimum loss contingency amount in a probable range is recorded. Legal fees are expensed as incurred. AbbVie accrues for product liability claims on an undiscounted basis. The liabilities are evaluated quarterly and adjusted if necessary as additional information becomes available. Receivables for insurance recoveries for product liability claims, if any, are recorded as assets on an undiscounted basis when it is probable that a recovery will be realized.
Goodwill and Intangible Assets
Intangible assets acquired in a business combination are recorded at fair value using a discounted cash flow model. The discounted cash flow model requires assumptions about the timing and amount of future net cash flows, risk, the cost of capital and terminal values of market participants. Definite-lived intangibles are amortized over their estimated useful lives using the estimated pattern of economic benefit. AbbVie reviews the recoverability of definite-lived intangible assets whenever events or changes in circumstances indicate the carrying value of an asset may not be recoverable. AbbVie first compares the projected undiscounted cash flows to be generated by the asset to its carrying value. If the undiscounted cash
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 58 |
flows of an intangible asset are less than the carrying value, the intangible asset is written down to its fair value. Where cash flows cannot be identified for an individual asset, the review is applied at the lowest level for which cash flows are largely independent of the cash flows of other assets and liabilities.
Goodwill and indefinite-lived assets are not amortized, but are subject to an impairment review annually and more frequently when indicators of impairment exist. An impairment of goodwill could occur if the carrying amount of a reporting unit exceeded the fair value of that reporting unit. An impairment of indefinite-lived intangible assets would occur if the fair value of the intangible asset is less than the carrying value.
The company tests its goodwill for impairment by first assessing qualitative factors to determine whether it is more likely than not that the fair value is less than its carrying amount. If the company concludes it is more likely than not that the fair value of the reporting unit is less than its carrying amount, a quantitative impairment test is performed. AbbVie tests indefinite-lived intangible assets for impairment by first assessing qualitative factors to determine whether it is more likely than not that the fair value is less than its carrying amount. If the company concludes it is more likely than not that the fair value is less than its carrying amount, a quantitative impairment test is performed. For its quantitative impairment tests, the company uses an estimated future cash flow approach that requires significant judgment with respect to future volume, revenue and expense growth rates, changes in working capital use, the selection of an appropriate discount rate, asset groupings and other assumptions and estimates. The estimates and assumptions used are consistent with the company's business plans and a market participant's views. The use of alternative estimates and assumptions could increase or decrease projected cash flows and the estimated fair value of the related intangible assets. Future changes to these estimates and assumptions could have a material impact on the company's results of operations. Actual results may differ from the company's estimates.
Foreign Currency Translation
Foreign subsidiary earnings are translated into U.S. dollars using average exchange rates. The net assets of foreign subsidiaries are translated into U.S. dollars using period-end exchange rates. The U.S. dollar affects that arise from translating the net assets of these subsidiaries at changing rates are recognized in other comprehensive income (loss) in the consolidated statements of comprehensive income. The net assets of subsidiaries in highly inflationary economies are remeasured as if the functional currency were the reporting currency. The remeasurement is recognized in net foreign exchange loss in the consolidated statements of earnings.
Derivatives
All derivative instruments are recognized as either assets or liabilities at fair value on the consolidated balance sheets and are classified as current or long-term based on the scheduled maturity of the instrument.
For derivatives formally designated as hedges, the company assesses at inception and quarterly thereafter whether the hedging derivatives are highly effective in offsetting changes in the fair value or cash flows of the hedged item. The changes in fair value of a derivative designated as a fair value hedge and of the hedged item attributable to the hedged risk are recognized in earnings immediately. The effective portions of changes in the fair value of a derivative designated as a cash flow hedge are reported in AOCI and are subsequently recognized in earnings consistent with the underlying hedged item. If it is determined that a derivative is no longer highly effective as a hedge, the company discontinues hedge accounting prospectively. If a hedged forecasted transaction becomes probable of not occurring, any gains or losses are reclassified from AOCI to earnings. Derivatives that are not designated as hedges are adjusted to fair value through current earnings.
The company also uses derivative instruments or foreign currency denominated debt to hedge its net investments in certain foreign subsidiaries and affiliates. Realized and unrealized gains and losses from these hedges are included in AOCI.
Derivative cash flows, with the exception of net investment hedges, are principally classified in the operating section of the consolidated statements of cash flows, consistent with the underlying hedged item. Cash flows related to net investment hedges are classified in the investing section of the consolidated statements of cash flows.
| | | | | | | | | | | |
59 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Note 3 Supplemental Financial Information Interest Expense, Net | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Interest expense | $ | 2,230 | | | $ | 2,423 | | | $ | 2,454 | |
| Interest income | (186) | | | (39) | | | (174) | |
| Interest expense, net | $ | 2,044 | | | $ | 2,384 | | | $ | 2,280 | |
Accounts Payable and Accrued Liabilities | | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| Sales rebates | $ | 10,717 | | | $ | 8,254 | |
| Dividends payable | 2,680 | | | 2,543 | |
| Accounts payable | 2,934 | | | 2,882 | |
| Current portion of contingent consideration liabilities | 1,469 | | | 1,249 | |
| Salaries, wages and commissions | 1,371 | | | 1,785 | |
| Royalty and license arrangements | 412 | | | 661 | |
| Other | 5,819 | | | 5,325 | |
| Accounts payable and accrued liabilities | $ | 25,402 | | | $ | 22,699 | |
Other Long-Term Liabilities | | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| Contingent consideration liabilities | $ | 14,915 | | | $ | 13,638 | |
| Liabilities for unrecognized tax benefits | 6,502 | | | 5,970 | |
| Income taxes payable | 2,985 | | | 3,442 | |
| Pension and other post-employment benefits | 1,638 | | | 3,153 | |
| Other | 4,615 | | | 2,498 | |
| Other long-term liabilities | $ | 30,655 | | | $ | 28,701 | |
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 60 |
Note 4 Earnings Per Share AbbVie grants certain restricted stock units (RSUs) that are considered to be participating securities. Due to the presence of participating securities, AbbVie calculates earnings per share (EPS) using the more dilutive of the treasury stock or the two-class method. For all periods presented, the two-class method was more dilutive.
The following table summarizes the impact of the two-class method: | | | | | | | | | | | | | | | | | |
| Years ended December 31, |
| (in millions, except per share data) | 2022 | | 2021 | | 2020 |
| Basic EPS | | | | | |
| Net earnings attributable to AbbVie Inc. | $ | 11,836 | | | $ | 11,542 | | | $ | 4,616 | |
| Earnings allocated to participating securities | 54 | | | 74 | | | 60 | |
| Earnings available to common shareholders | $ | 11,782 | | | $ | 11,468 | | | $ | 4,556 | |
| Weighted average basic shares of common stock outstanding | 1,771 | | | 1,770 | | | 1,667 | |
| Basic earnings per share attributable to AbbVie Inc. | $ | 6.65 | | | $ | 6.48 | | | $ | 2.73 | |
| | | | | |
| Diluted EPS | | | | | |
| Net earnings attributable to AbbVie Inc. | $ | 11,836 | | | $ | 11,542 | | | $ | 4,616 | |
| Earnings allocated to participating securities | 54 | | | 74 | | | 60 | |
| Earnings available to common shareholders | $ | 11,782 | | | $ | 11,468 | | | $ | 4,556 | |
| Weighted average shares of common stock outstanding | 1,771 | | | 1,770 | | | 1,667 | |
| Effect of dilutive securities | 7 | | | 7 | | | 6 | |
| Weighted average diluted shares of common stock outstanding | 1,778 | | | 1,777 | | | 1,673 | |
| Diluted earnings per share attributable to AbbVie Inc. | $ | 6.63 | | | $ | 6.45 | | | $ | 2.72 | |
Certain shares issuable under stock-based compensation plans were excluded from the computation of EPS because the effect would have been antidilutive. The number of common shares excluded was insignificant for all periods presented.
| | | | | | | | | | | |
61 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Note 5 Licensing, Acquisitions and Other Arrangements Acquisition of DJS Antibodies Ltd
In October 2022, AbbVie entered into an agreement to acquire DJS Antibodies Ltd (DJS) including its lead program DJS-002 and proprietary HEPTAD platform. DJS-002 is an LPAR1 antagonist antibody currently in preclinical studies for the treatment of Idiopathic Pulmonary Fibrosis and other fibrotic diseases. HEPTAD platform is a potential novel approach to antibody discovery with specific capabilities targeting transmembrane protein targets. The aggregate purchase price of $287 million was comprised of a $255 million upfront cash payment and $32 million for the acquisition date fair value of contingent consideration liabilities, for which AbbVie may owe up to $95 million in future payments upon achievement of certain development milestones. The transaction was accounted for as a business combination using the acquisition method of accounting. As of the acquisition date, AbbVie acquired $233 million of intangible assets for in-process research and development, $22 million of intangible assets for developed product rights and $60 million of deferred tax liabilities. Other assets and liabilities assumed were insignificant. The acquisition resulted in the recognition of $92 million of goodwill which is not deductible for tax purposes.
Acquisition of Soliton, Inc.
In December 2021, AbbVie completed its previously announced acquisition of Soliton, Inc. (Soliton). Soliton's RESONIC (Rapid Acoustic Pulse device) has U.S. Food and Drug Administration (FDA) 510(k) clearance for the long-term improvement in the appearance of cellulite up to one year. The transaction was accounted for as a business combination using the acquisition method of accounting. Total consideration transferred allocated to the purchase price consisted of cash consideration of $535 million paid to holders of Soliton common stock, equity-based awards and warrants. As of the transaction date, AbbVie acquired $407 million of intangible assets for developed product rights and assumed deferred tax liabilities totaling $63 million. Other assets and liabilities were insignificant. The acquisition resulted in the recognition of $177 million of goodwill which is not deductible for tax purposes.
Acquisition of Luminera
In October 2020, AbbVie entered into an agreement with Luminera, a privately held aesthetics company based in Israel, to acquire Luminera's full dermal filler portfolio and R&D pipeline including HArmonyCa, a dermal filler intended for facial soft tissue augmentation. The aggregate accounting purchase price of $186 million was comprised of a $122 million upfront cash payment and $64 million for the acquisition date fair value of contingent consideration liabilities, for which AbbVie may owe up to $90 million in future payments upon achievement of certain commercial milestones. The agreement was accounted for as a business combination using the acquisition method of accounting. As of the acquisition date, AbbVie acquired $127 million of intangible assets for in-process research and development and $33 million of intangible assets for developed product rights. Other assets and liabilities assumed were insignificant. The acquisition resulted in the recognition of $12 million of goodwill which is not deductible for tax purposes.
Acquisition of Allergan
On May 8, 2020, AbbVie completed its acquisition of all outstanding equity interests in Allergan in a cash and stock transaction. Allergan is a global pharmaceutical leader focused on developing, manufacturing and commercializing branded pharmaceutical, device, biologic, surgical and regenerative medicine products for patients around the world. The combination created a diverse entity with leadership positions across immunology, hematologic oncology, aesthetics, neuroscience and eye care. AbbVie's existing product portfolio and pipeline is enhanced with numerous Allergan assets and Allergan's product portfolio benefits from AbbVie's commercial strength, expertise and international infrastructure. Under the terms of the acquisition, each ordinary share of Allergan common stock was converted into the right to receive (i) $120.30 in cash and (ii) 0.8660 of a share of AbbVie common stock.
Total consideration for the acquisition of Allergan is summarized as follows: | | | | | |
| (in millions) | |
Cash consideration paid to Allergan shareholders (a) | $ | 39,675 | |
Fair value of AbbVie common stock issued to Allergan shareholders (b) | 23,979 | |
Fair value of AbbVie equity awards issued to Allergan equity award holders (c) | 430 | |
| Total consideration | $ | 64,084 | |
(a)Represents cash consideration transferred of $120.30 per outstanding Allergan ordinary share based on 330 million Allergan ordinary shares outstanding at closing.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 62 |
(b)Represents the acquisition date fair value of 286 million shares of AbbVie common stock issued to Allergan shareholders based on the exchange ratio of 0.8660 AbbVie shares for each outstanding Allergan ordinary share at the May 8, 2020, closing price of $83.96 per share.
(c)Represents the pre-acquisition service portion of the fair value of 11 million AbbVie stock options and 8 million RSUs issued to Allergan equity award holders.
The acquisition of Allergan has been accounted for as a business combination using the acquisition method of accounting. The acquisition method requires, among other things, that assets acquired and liabilities assumed in a business combination be recognized at their fair values as of the acquisition date. The valuation of assets acquired and liabilities assumed was finalized during the second quarter of 2021. Measurement period adjustments to the preliminary purchase price allocation during 2021 included (i) an increase to intangible assets of $710 million; (ii) an increase to deferred income tax liabilities of $148 million; (iii) other individually insignificant adjustments for a net increase to identifiable net assets of $2 million; and (iv) a corresponding decrease to goodwill of $564 million. The measurement period adjustments primarily resulted from the completion of the valuation of certain license agreement intangible assets based on facts and circumstances that existed as of the acquisition date and did not result from intervening events subsequent to such date. These adjustments did not have a significant impact on AbbVie's results of operations in 2021 and would not have had a significant impact on prior period results if these adjustments had been made as of the acquisition date.
The following table summarizes the final fair value of assets acquired and liabilities assumed as of the acquisition date: | | | | | |
| (in millions) | |
| Assets acquired and liabilities assumed | |
| Cash and equivalents | $ | 1,537 | |
| Short-term investments | 1,421 | |
| Accounts receivable | 2,374 | |
| Inventories | 2,340 | |
| Prepaid expenses and other current assets | 1,982 | |
| Investments | 137 | |
| Property and equipment | 2,129 | |
| Intangible assets | |
| Definite-lived intangible assets | 68,190 | |
| In-process research and development | 1,600 | |
| Other noncurrent assets | 1,395 | |
| Short-term borrowings | (60) | |
| Current portion of long-term debt and finance lease obligations | (1,899) | |
| Accounts payable and accrued liabilities | (5,852) | |
| Long-term debt and finance lease obligations | (18,937) | |
| Deferred income taxes | (3,940) | |
| Other long-term liabilities | (4,765) | |
| |
| Total identifiable net assets | 47,652 | |
| Goodwill | 16,432 | |
| Total assets acquired and liabilities assumed | $ | 64,084 | |
The fair value step-up adjustment to inventories of $1.2 billion was amortized to cost of products sold when the inventory was sold to customers and was fully amortized as of December 31, 2021.
Intangible assets relate to $68.2 billion of definite-lived intangible assets and $1.6 billion of IPR&D. The acquired definite-lived intangible assets consist of developed product rights and license agreements and are being amortized over a weighted-average estimated useful life of approximately twelve years using the estimated pattern of economic benefit. The estimated fair values of identifiable intangible assets were determined using the "income approach" which is a valuation technique that provides an estimate of the fair value of an asset based on market participant expectations of the cash flows an asset would generate over its remaining useful life. Some of the more significant assumptions inherent in the development of these asset valuations include the estimated net cash flows for each year for each asset or product, the appropriate discount rate necessary to measure the risk inherent in each future cash flow stream, the life cycle of each asset, the potential regulatory and commercial success risk, competitive trends impacting the asset and each cash flow stream, as well as other factors.
| | | | | | | | | | | |
63 |  | 2022 Form 10-K | 2022 Form 10-K | | |
The fair value of long-term debt was determined by quoted market prices as of the acquisition date and the total purchase price adjustment of $1.3 billion is being amortized as a reduction to interest expense, net over the lives of the related debt.
Goodwill was calculated as the excess of the consideration transferred over the net assets recognized and represents the future economic benefits arising from the other assets acquired that could not be individually identified and separately recognized. Specifically, the goodwill recognized from the acquisition of Allergan represents the value of additional growth platforms and an expanded revenue base as well as anticipated operational synergies and cost savings from the creation of a single combined global organization. The goodwill is not deductible for tax purposes.
Following the acquisition date, the operating results of Allergan have been included in the consolidated financial statements. For the period from the acquisition date through December 31, 2020, net revenues attributable to Allergan were $10.3 billion and operating losses attributable to Allergan were $1.1 billion, inclusive of $4.0 billion of intangible asset amortization and $1.2 billion of inventory fair value step-up amortization.
Acquisition-related expenses, which were comprised primarily of regulatory, financial advisory and legal fees, totaled $781 million for the year ended December 31, 2020 which were included in SG&A expenses in the consolidated statements of earnings. In the fourth quarter of 2021, AbbVie recovered certain acquisition-related regulatory fees totaling $401 million which was recorded as a reduction to SG&A expenses in the consolidated statement of earnings for the year ended December 31, 2021.
Pro Forma Financial Information
The following table presents the unaudited pro forma combined results of AbbVie and Allergan for 2020 as if the acquisition of Allergan had occurred on January 1, 2019: | | | | | | | | |
| years ended December 31 (in millions) | | 2020 |
| Net revenues | | $ | 50,521 | |
| Net earnings | | 6,746 | |
The unaudited pro forma combined financial information was prepared using the acquisition method of accounting and was based on the historical financial information of AbbVie and Allergan. In order to reflect the occurrence of the acquisition on January 1, 2019 as required, the unaudited pro forma financial information includes adjustments to reflect incremental amortization expense to be incurred based on the final fair values of the identifiable intangible assets acquired; the incremental cost of products sold related to the fair value adjustments associated with acquisition date inventory; the additional interest expense associated with the issuance of debt to finance the acquisition; and the reclassification of acquisition-related costs incurred during the year ended December 31, 2020 to the year ended December 31, 2019. The unaudited pro forma financial information is not necessarily indicative of what the consolidated results of operations would have been had the acquisition been completed on January 1, 2019. In addition, the unaudited pro forma financial information is not a projection of future results of operations of the combined company nor does it reflect the expected realization of any synergies or cost savings associated with the acquisition.
Other Licensing & Acquisitions Activity
Cash outflows related to other acquisitions and investments totaled $539 million in 2022, $1.4 billion in 2021 and $1.4 billion in 2020. AbbVie recorded acquired IPR&D and milestones expense of $697 million in 2022, $1.1 billion in 2021 and $1.4 billion in 2020. Significant arrangements impacting 2022, 2021 and 2020, some of which require contingent milestone payments, are summarized below.
Syndesi Therapeutics SA
In February 2022, AbbVie acquired Syndesi Therapeutics SA and its portfolio of novel modulators of the synaptic vesicle protein 2A, including its lead molecule SDI-118 and accounted for the transaction as an asset acquisition. SDI-118 is a small molecule currently in Phase 1b studies, which is being evaluated to target nerve terminals to enhance synaptic efficiency. Under the terms of the agreement, AbbVie made an upfront payment of $130 million which was recorded to acquired IPR&D and milestones expense in the consolidated statement of earnings in the first quarter of 2022. The agreement also includes
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 64 |
additional future payments of up to $870 million upon the achievement of certain development, regulatory and commercial milestones.
Juvise Pharmaceuticals
In June 2022, AbbVie and Laboratories Juvise Pharmaceuticals (Juvise) entered into an asset purchase agreement where Juvise acquired worldwide commercial rights of a mature brand Pylera, which is used for the treatment of peptic ulcers with an infection by the bacterium Helicobacter pylori. The transaction was accounted for as the sale of an asset. Upon completion of the transaction, AbbVie received net cash proceeds of $215 million and recognized a pre-tax gain of $172 million which was recorded in other operating income in the consolidated statement of earnings in the second quarter of 2022.
Calico Life Sciences LLC
In July 2021, AbbVie and Calico Life Sciences LLC (Calico) entered into an extension of their collaboration to discover, develop and bring to market new therapies for patients with age-related diseases, including neurodegeneration and cancer. This is the second collaboration extension and builds on the partnership established in 2014 and extended in 2018. Under the terms of the agreement, AbbVie and Calico will each contribute an additional $500 million, and the term is extended for an additional three years. AbbVie’s contribution is payable in two equal installments beginning in 2023. Calico will be responsible for research and early development until 2025 and will advance collaboration projects into Phase 2a through 2030. Following completion of the Phase 2a studies, AbbVie will have the option to exclusively license the collaboration compounds. Upon exercise, AbbVie would be responsible for late-stage development and commercial activities. Collaboration costs and profits will be shared equally by both parties post option exercise. During the third quarter of 2021, AbbVie recorded $500 million as other operating expense in the consolidated statement of earnings related to its commitments under the agreement.
TeneoOne and TNB-383B
In September 2021, AbbVie acquired TeneoOne, an affiliate of Teneobio, Inc., and TNB-383B, a BCMA-targeting immunotherapeutic for the potential treatment of relapsed or refractory multiple myeloma (R/R MM). In February 2019, AbbVie and TeneoOne entered a strategic transaction to develop and commercialize TNB-383B, a bispecific antibody that simultaneously targets BCMA and CD3 and is designed to direct the body's own immune system to target and kill BCMA-expressing tumor cells. AbbVie exercised its exclusive right to acquire TeneoOne and TNB-383B based on an interim analysis of an ongoing Phase 1 study and accounted for the transaction as an asset acquisition. Under the terms of the agreement, AbbVie made an exercise payment of $400 million which was recorded to acquired IPR&D and milestones expense in the consolidated statement of earnings in the third quarter of 2021. The agreement also included additional payments of up to $250 million upon the achievement of certain development, regulatory and commercial milestones.
REGENXBIO Inc.
In September 2021, AbbVie and REGENXBIO Inc. (REGENXBIO) entered into a collaboration to develop and commercialize RGX-314, an investigational gene therapy for wet age-related macular degeneration, diabetic retinopathy and other chronic retinal diseases. The collaboration provides AbbVie with an exclusive global license to develop and commercialize RGX-314. REGENXBIO will be responsible for completion of ongoing trials, AbbVie and REGENXBIO will collaborate and share costs of additional trials and AbbVie will lead the clinical development and commercialization of RGX-314 globally. REGENXBIO and AbbVie will share equally in pre-tax profits from net revenues of RGX-314 in the U.S. and AbbVie will pay REGENXBIO tiered royalties on net revenues outside the U.S. Upon closing in the fourth quarter of 2021, AbbVie made an upfront payment of $370 million to exclusively license RGX-314 which was recorded to acquired IPR&D and milestones expense in the consolidated statement of earnings for the year ended December 31, 2022. The agreement also included additional payments of up to $1.4 billion upon the achievement of certain development, regulatory and commercial milestones.
I-Mab Biopharma
In September 2020, AbbVie and I-Mab Biopharma (I-Mab) entered into a collaboration agreement for the development and commercialization of lemzoparlimab, an anti-CD47 monoclonal antibody internally discovered and developed by I-Mab for the treatment of multiple cancers. Both companies will collaborate to design and conduct further global clinical trials to evaluate lemzoparlimab. The collaboration provides AbbVie an exclusive global license, excluding greater China, to develop and commercialize lemzoparlimab. The companies will share manufacturing responsibilities with AbbVie being the primary manufacturer for global supply. The agreement also allows for potential collaboration on future CD47-related therapeutic agents, subject to further licenses to explore each other's related programs in their respective territories. The terms of the arrangement include an initial upfront payment of $180 million to exclusively license lemzoparlimab along with a milestone payment of $20 million based on the Phase I results, for a total of $200 million, which was recorded to acquired IPR&D and milestones expense in the consolidated statement of earnings in the fourth quarter of 2020 after regulatory approval of the
| | | | | | | | | | | |
65 |  | 2022 Form 10-K | 2022 Form 10-K | | |
transaction. In addition, I-Mab will be eligible to receive up to $1.7 billion upon the achievement of certain clinical development, regulatory and commercial milestones, and AbbVie will pay tiered royalties from low-to-mid teen percentages on global net revenues outside of greater China.
Genmab A/S
In June 2020, AbbVie and Genmab A/S (Genmab) entered into a collaboration agreement to jointly develop and commercialize three of Genmab's early-stage investigational bispecific antibody therapeutics and entered into a discovery research collaboration for future differentiated antibody therapeutics for the treatment of cancer. Under the terms of the agreement, Genmab granted AbbVie an exclusive license to its epcoritamab (DuoBody-CD3xCD20), DuoHexaBody-CD37 and DuoBody-CD3x5T4 programs. For epcoritamab, the companies will share commercial responsibilities in the U.S. and Japan, with AbbVie responsible for further global commercialization. Genmab will record net revenues in the U.S. and Japan, and the parties will share equally in pre-tax profits from these sales. Genmab will receive tiered royalties on remaining global sales. For the discovery research partnership, Genmab will conduct Phase 1 studies for these programs and AbbVie retains the right to opt-in to program development. During 2020, AbbVie made an upfront payment of $750 million, which was recorded to acquired IPR&D and milestones expense in the consolidated statement of earnings. AbbVie could make additional payments of up to $3.2 billion upon the achievement of certain development, regulatory and commercial milestones for all programs.
Other Arrangements
In addition to the significant arrangements described above, AbbVie entered into several other arrangements resulting in charges related to upfront payments of $315 million in 2022, $192 million in 2021 and $248 million in 2020. In connection with the other individually insignificant early-stage arrangements entered into in 2022, AbbVie could make additional payments of up to $7.5 billion upon the achievement of certain development, regulatory and commercial milestones. Acquired IPR&D and milestones expense also included development milestones of $252 million in 2022, $162 million in 2021 and $178 million in 2020.
The company has ongoing transactions with other entities through collaboration agreements. The following represent the significant collaboration agreements impacting 2022, 2021 and 2020.
Collaboration with Janssen Biotech, Inc.
In December 2011, Pharmacyclics, a wholly-owned subsidiary of AbbVie, entered into a worldwide collaboration and license agreement with Janssen Biotech, Inc. and its affiliates (Janssen), one of the Janssen Pharmaceutical companies of Johnson & Johnson, for the joint development and commercialization of Imbruvica, a novel, orally active, selective covalent inhibitor of Bruton's tyrosine kinase and certain compounds structurally related to Imbruvica, for oncology and other indications, excluding all immune and inflammatory mediated diseases or conditions and all psychiatric or psychological diseases or conditions, in the United States and outside the United States.
The collaboration provides Janssen with an exclusive license to commercialize Imbruvica outside of the United States and co-exclusively with AbbVie in the United States. Both parties are responsible for the development, manufacturing and marketing of any products generated as a result of the collaboration. The collaboration has no set duration or specific expiration date and provides for potential future development, regulatory and approval milestone payments of up to $200 million to AbbVie. The collaboration also includes a cost sharing arrangement for associated collaboration activities. Except in certain cases, Janssen is responsible for approximately 60% of collaboration development costs and AbbVie is responsible for the remaining 40% of collaboration development costs.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 66 |
In the United States, both parties have co-exclusive rights to commercialize the products; however, AbbVie is the principal in the end-customer product sales. AbbVie and Janssen share pre-tax profits and losses equally from the commercialization of products. Sales of Imbruvica are included in AbbVie's net revenues. Janssen's share of profits is included in AbbVie's cost of products sold. Other costs incurred under the collaboration are reported in their respective expense line items, net of Janssen's share.
Outside the United States, Janssen is responsible for and has exclusive rights to commercialize Imbruvica. AbbVie and Janssen share pre-tax profits and losses equally from the commercialization of products. AbbVie's share of profits is included in AbbVie's net revenues. Other costs incurred under the collaboration are reported in their respective expense line items, net of Janssen's share.
The following table shows the profit and cost sharing relationship between Janssen and AbbVie: | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| United States - Janssen's share of profits (included in cost of products sold) | $ | 1,607 | | | $ | 2,018 | | | $ | 2,012 | |
| International - AbbVie's share of profits (included in net revenues) | 1,142 | | | 1,087 | | | 1,009 | |
| Global - AbbVie's share of other costs (included in respective line items) | 268 | | | 304 | | | 295 | |
AbbVie’s receivable from Janssen, included in accounts receivable, net, was $295 million at December 31, 2022 and $294 million at December 31, 2021. AbbVie’s payable to Janssen, included in accounts payable and accrued liabilities, was $379 million at December 31, 2022 and $509 million at December 31, 2021.
Collaboration with Genentech, Inc.
AbbVie and Genentech, Inc. (Genentech), a member of the Roche Group, are parties to a collaboration and license agreement executed in 2007 to jointly research, develop and commercialize human therapeutic products containing BCL-2 inhibitors and certain other compound inhibitors which includes Venclexta, a BCL-2 inhibitor used to treat certain hematological malignancies. AbbVie shares equally with Genentech all pre-tax profits and losses from the development and commercialization of Venclexta in the United States. AbbVie pays royalties on Venclexta net revenues outside the United States.
AbbVie manufactures and distributes Venclexta globally and is the principal in the end-customer product sales. Sales of Venclexta are included in AbbVie's net revenues. Genentech's share of United States profits is included in AbbVie's cost of products sold. AbbVie records sales and marketing costs associated with the United States collaboration as part of SG&A expenses and global development costs as part of R&D expenses, net of Genentech’s share. Royalties paid for Venclexta revenues outside the United States are also included in AbbVie’s cost of products sold.
The following table shows the profit and cost sharing relationship between Genentech and AbbVie: | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Genentech's share of profits, including royalties (included in cost of products sold) | $ | 778 | | | $ | 703 | | | $ | 533 | |
| AbbVie's share of sales and marketing costs from U.S. collaboration (included in SG&A) | 37 | | | 40 | | | 46 | |
| AbbVie's share of development costs (included in R&D) | 121 | | | 140 | | | 129 | |
| | | | | | | | | | | |
67 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Note 7 Goodwill and Intangible Assets Goodwill
The following table summarizes the changes in the carrying amount of goodwill: | | | | | |
| (in millions) | |
| Balance as of December 31, 2020 | $ | 33,124 | |
Additions(a) | 177 | |
Measurement period adjustments(b) | (564) | |
| Foreign currency translation adjustments and other | (358) | |
| Balance as of December 31, 2021 | 32,379 | |
Additions(c) | 92 | |
| Foreign currency translation adjustments and other | (315) | |
| Balance as of December 31, 2022 | $ | 32,156 | |
(a)Goodwill additions related to the acquisition of Soliton in the fourth quarter of 2021 (see Note 5).
(b)Measurement period adjustments recorded in 2021 related to the acquisition of Allergan (see Note 5).
(c)Goodwill additions related to the acquisition of DJS in the fourth quarter of 2022 (see Note 5).
The company performs its annual goodwill impairment assessment in the third quarter, or earlier if impairment indicators exist. As of December 31, 2022 and 2021, there were no accumulated goodwill impairment losses.
Intangible Assets, Net
The following table summarizes intangible assets: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 2022 | | 2021 |
| as of December 31 (in millions) | Gross
carrying
amount | | Accumulated
amortization | | Net
carrying
amount | | Gross
carrying
amount | | Accumulated
amortization | | Net
carrying
amount |
| Definite-lived intangible assets | | | | | | | | | | | |
| Developed product rights | $ | 87,698 | | | $ | (25,003) | | | $ | 62,695 | | | $ | 88,945 | | | $ | (18,463) | | | $ | 70,482 | |
| License agreements | 8,474 | | | (4,642) | | | 3,832 | | | 8,487 | | | (3,688) | | | 4,799 | |
| Total definite-lived intangible assets | 96,172 | | | (29,645) | | | 66,527 | | | 97,432 | | | (22,151) | | | 75,281 | |
| Indefinite-lived intangible assets | 912 | | | — | | | 912 | | | 670 | | | — | | | 670 | |
| Total intangible assets, net | $ | 97,084 | | | $ | (29,645) | | | $ | 67,439 | | | $ | 98,102 | | | $ | (22,151) | | | $ | 75,951 | |
Definite-Lived Intangible Assets
In September 2022, the company made a strategic decision to reduce ongoing sales and marketing investment related to Vuity, an on-market product to treat presbyopia. This strategic decision contributed to a significant decrease in the estimated future cash flows for the product and represented a triggering event which required the company to evaluate the underlying definite lived-intangible asset for impairment. The company utilized a discounted cash flow analysis to estimate the fair value of the intangible asset resulting in a full impairment of both the gross and net carrying amount. Based on the revised cash flows, the company recorded a pre-tax impairment charge of $770 million to cost of products sold in the consolidated statement of earnings for the third quarter of 2022.
Definite-lived intangible assets are amortized over their estimated useful lives, which range between 1 to 16 years with an average of 12 years for developed product rights and 11 years for license agreements. Amortization expense was $7.7 billion in 2022, $7.7 billion in 2021 and $5.8 billion in 2020 and was included in cost of products sold in the consolidated statements of earnings. The anticipated annual amortization expense for definite-lived intangible assets recorded as of December 31, 2022 is as follows: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (in billions) | 2023 | | 2024 | | 2025 | | 2026 | | 2027 |
| Anticipated annual amortization expense | $ | 7.7 | | | $ | 7.9 | | | $ | 8.0 | | | $ | 7.3 | | | $ | 5.8 | |
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 68 |
Indefinite-Lived Intangible Assets
Indefinite-lived intangible assets represent acquired IPR&D associated with products that have not yet received regulatory approval. Indefinite-lived intangible assets as of December 31, 2022 primarily relate to the acquisitions of Allergan and DJS.
The company performs its annual impairment assessment of indefinite-lived intangible assets in the third quarter, or earlier if impairment indicators exist.
Note 8 Integration and Restructuring Plans Allergan Integration Plan
Following the closing of the Allergan acquisition, AbbVie implemented an integration plan designed to reduce costs, integrate and optimize the combined organization and incurred total cumulative charges of $2.3 billion through 2022. These costs consisted of severance and employee benefit costs (cash severance, non-cash severance, including accelerated equity award compensation expense, retention and other termination benefits) and other integration expenses.
The following table summarizes the charges (benefits) associated with the Allergan acquisition integration plan: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Severance and employee benefits | | Other integration |
| year ended December 31 (in millions) | | 2022 | | 2021 | | 2020 | | 2022 | | 2021 | | 2020 |
| Cost of products sold | | $ | (4) | | | $ | 5 | | | $ | 109 | | | $ | 121 | | | $ | 127 | | | $ | 21 | |
| Research and development | | — | | | — | | | 199 | | | 23 | | | 102 | | | 177 | |
| Selling, general and administrative | | (4) | | | 64 | | | 388 | | | 403 | | | 289 | | | 237 | |
| Total charges (benefits) | | $ | (8) | | | $ | 69 | | | $ | 696 | | | $ | 547 | | | $ | 518 | | | $ | 435 | |
The following table summarizes the cash activity in the recorded liability associated with the integration plan: | | | | | | | | | | | |
| year ended December 31 (in millions) | Severance and employee benefits | | Other integration |
| Charges | $ | 594 | | | $ | 435 | |
| Payments and other adjustments | (227) | | | (415) | |
| Accrued balance as of December 31, 2020 | 367 | | | 20 | |
| Charges | 65 | | | 461 | |
| Payments and other adjustments | (210) | | | (448) | |
| Accrued balance as of December 31, 2021 | 222 | | | 33 | |
| Charges (benefits) | (8) | | | 385 | |
| Payments and other adjustments | (116) | | | (409) | |
| Accrued balance as of December 31, 2022 | $ | 98 | | | $ | 9 | |
Other Restructuring
AbbVie continuously evaluates its operations to identify opportunities to optimize its manufacturing and R&D operations, commercial infrastructure and administrative costs and to respond to changes in its business environment. As a result, AbbVie management periodically approves individual restructuring plans to achieve these objectives. In 2022, 2021 and 2020, no such plans were individually significant. Restructuring charges recorded were $241 million in 2022, $59 million in 2021 and $60 million in 2020 and were primarily related to employee severance and contractual obligations. These charges were recorded in cost of products sold, R&D expense and SG&A expenses in the consolidated statements of earnings based on the classification of the affected employees or operations.
| | | | | | | | | | | |
69 |  | 2022 Form 10-K | 2022 Form 10-K | | |
The following table summarizes the cash activity in the restructuring reserve for 2022, 2021 and 2020:
| | | | | |
| (in millions) | |
| Accrued balance as of December 31, 2019 | $ | 140 | |
| Restructuring charges | 58 | |
| Payments and other adjustments | (108) | |
| Accrued balance as of December 31, 2020 | 90 | |
| Restructuring charges | 54 | |
| Payments and other adjustments | (111) | |
| Accrued balance as of December 31, 2021 | 33 | |
| Restructuring charges | 193 | |
| Payments and other adjustments | (50) | |
| Accrued balance as of December 31, 2022 | $ | 176 | |
AbbVie's lease portfolio primarily consists of real estate properties, vehicles and equipment. The following table summarizes the amounts and location of operating and finance leases on the consolidated balance sheets:
| | | | | | | | | | | | | | |
| as of December 31 (in millions) | Balance sheet caption | 2022 | | 2021 |
| Assets | | | | |
| Operating | Other assets | $ | 737 | | | $ | 762 | |
| Finance | Property and equipment, net | 25 | | | 33 | |
| Total lease assets | | $ | 762 | | | $ | 795 | |
| Liabilities | | | | |
| Operating | | | | |
| Current | Accounts payable and accrued liabilities | $ | 166 | | | $ | 178 | |
| Noncurrent | Other long-term liabilities | 754 | | | 713 | |
| Finance | | | | |
| Current | Current portion of long-term debt and finance lease obligations | 17 | | | 9 | |
| Noncurrent | Long-term debt and finance lease obligations | 17 | | | 25 | |
| Total lease liabilities | | $ | 954 | | | $ | 925 | |
The following table summarizes the lease costs recognized in the consolidated statements of earnings:
| | | | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | | 2022 | | 2021 | | 2020 |
| Operating lease cost | | $ | 201 | | | $ | 226 | | | $ | 192 | |
| | | | | | |
| | | | | | |
| | | | | | |
| Short-term lease cost | | 67 | | | 56 | | | 59 | |
| Variable lease cost | | 71 | | | 71 | | | 60 | |
| Total lease cost | | $ | 339 | | | $ | 353 | | | $ | 311 | |
In December 2022, the company entered into an agreement to sublease a portion of its Madison, New Jersey office space through the end of the original lease maturity in 2030. As a result of this agreement, the company recognized an impairment loss on its right-of-use asset of $69 million and wrote-off the related leasehold improvements of $37 million. These losses were recorded to SG&A expense in the consolidated statements of earnings for the year ended December 31, 2022. The company used a discounted cash flows method to value the right-of-use asset to determine the impairment amount.
Sublease income and finance lease costs were insignificant in 2022, 2021 and 2020.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 70 |
The following table presents the weighted-average remaining lease term and weighted-average discount rate for operating and finance leases: | | | | | | | | | | | | | | | | | |
| years ended December 31 | 2022 | | 2021 | | 2020 |
| Weighted-average remaining lease term (years) | | | | | |
| Operating | 8 | | 7 | | 8 |
| Finance | 2 | | 3 | | 3 |
| Weighted-average discount rate | | | | | |
| Operating | 2.6 | % | | 2.4 | % | | 2.5 | % |
| Finance | 1.5 | % | | 1.1 | % | | 1.4 | % |
The following table presents supplementary cash flow information regarding the company's leases: | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Cash paid for amounts included in the measurement of lease liabilities | | | | | |
| Operating cash flows from operating leases | $ | 212 | | | $ | 236 | | | $ | 185 | |
| Right-of-use assets obtained in exchange for new operating lease liabilities | 235 | | | 66 | | | 692 | |
Finance lease cash flows were insignificant in 2022, 2021 and 2020. Right-of-use assets obtained in exchange for new operating lease liabilities as of December 31, 2020 included $453 million of right-of-use assets acquired in the Allergan acquisition.
The following table summarizes the future maturities of AbbVie's operating and finance lease liabilities as of December 31, 2022: | | | | | | | | | | | | | | | | | |
| (in millions) | Operating
leases | | Finance
leases | | Total (a) |
| 2023 | $ | 185 | | | $ | 17 | | | $ | 202 | |
| 2024 | 152 | | | 7 | | | 159 | |
| 2025 | 130 | | | 5 | | | 135 | |
| 2026 | 113 | | | 6 | | | 119 | |
| 2027 | 90 | | | — | | | 90 | |
| Thereafter | 361 | | | — | | | 361 | |
| Total lease payments | 1,031 | | | 35 | | | 1,066 | |
| Less: Interest | 111 | | | 1 | | | 112 | |
| Present value of lease liabilities | $ | 920 | | | $ | 34 | | | $ | 954 | |
(a)Lease payments recognized as part of lease liabilities for optional renewal periods are insignificant.
| | | | | | | | | | | |
71 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Note 10 Debt, Credit Facilities and Commitments and Contingencies The following table summarizes long-term debt: | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| as of December 31 (dollars in millions) | 2022 Effective interest rate (a) | | 2022 | | 2021 Effective interest rate (a) | | 2021 | | | | | |
2.30-3.45% aggregate notes due 2022 | 1.92-3.28% | | $ | — | | | 0.99-3.45% | | $ | 12,428 | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
| | | | | | | | | | | | |
3.75% senior notes due 2023 | 3.84 | % | | 1,250 | | | 3.84 | % | | 1,250 | | | | | | |
2.85% senior notes due 2023 | 2.91 | % | | 1,000 | | | 2.91 | % | | 1,000 | | | | | | |
| Floating rate term loans due 2023 | 2.45 | % | | 1,000 | | | 0.81 | % | | 1,000 | | | | | | |
1.50% senior euro notes due 2023 (€500 principal) | 0.49 | % | | 532 | | | 0.49 | % | | 567 | | | | | | |
2.80% senior notes due 2023 | 2.13 | % | | 350 | | | 2.13 | % | | 350 | | | | | | |
2.60% senior notes due 2024 | 2.69 | % | | 3,750 | | | 2.69 | % | | 3,750 | | | | | | |
1.375% senior euro notes due 2024 (€1,450 principal) | 1.46 | % | | 1,543 | | | 1.46 | % | | 1,643 | | | | | | |
3.85% senior notes due 2024 | 2.07 | % | | 1,032 | | | 2.07 | % | | 1,032 | | | | | | |
1.25% senior euro notes due 2024 (€700 principal) | 0.65 | % | | 745 | | | 0.65 | % | | 793 | | | | | | |
3.60% senior notes due 2025 | 3.66 | % | | 3,750 | | | 3.66 | % | | 3,750 | | | | | | |
3.80% senior notes due 2025 | 2.09 | % | | 3,021 | | | 2.09 | % | | 3,021 | | | | | | |
| Floating rate term loans due 2025 | 1.39 | % | | — | | | 1.36 | % | | 2,000 | | | | | | |
| Floating rate term loans due 2025 | 2.82 | % | | 2,000 | | | — | | | — | | | | | | |
2.95% senior notes due 2026 | 3.02 | % | | 4,000 | | | 3.02 | % | | 4,000 | | | | | | |
3.20% senior notes due 2026 | 3.28 | % | | 2,000 | | | 3.28 | % | | 2,000 | | | | | | |
0.75% senior euro notes due 2027 (€750 principal) | 0.86 | % | | 798 | | | 0.86 | % | | 850 | | | | | | |
4.25% senior notes due 2028 | 4.38 | % | | 1,750 | | | 4.38 | % | | 1,750 | | | | | | |
2.125% senior euro notes due 2028 (€750 principal) | 2.18 | % | | 798 | | | 2.18 | % | | 850 | | | | | | |
2.625% senior euro notes due 2028 (€500 principal) | 1.20 | % | | 532 | | | 1.20 | % | | 567 | | | | | | |
3.20% senior notes due 2029 | 3.25 | % | | 5,500 | | | 3.25 | % | | 5,500 | | | | | | |
2.125% senior euro notes due 2029 (€550 principal) | 1.19 | % | | 585 | | | 1.19 | % | | 623 | | | | | | |
1.25% senior euro notes due 2031 (€650 principal) | 1.30 | % | | 691 | | | 1.30 | % | | 737 | | | | | | |
4.55% senior notes due 2035 | 3.52 | % | | 1,789 | | | 3.52 | % | | 1,789 | | | | | | |
4.50% senior notes due 2035 | 4.58 | % | | 2,500 | | | 4.58 | % | | 2,500 | | | | | | |
4.30% senior notes due 2036 | 4.37 | % | | 1,000 | | | 4.37 | % | | 1,000 | | | | | | |
4.05% senior notes due 2039 | 4.11 | % | | 4,000 | | | 4.11 | % | | 4,000 | | | | | | |
4.40% senior notes due 2042 | 4.46 | % | | 2,600 | | | 4.46 | % | | 2,600 | | | | | | |
4.625% senior notes due 2042 | 4.00 | % | | 457 | | | 4.00 | % | | 457 | | | | | | |
4.85% senior notes due 2044 | 4.11 | % | | 1,074 | | | 4.11 | % | | 1,074 | | | | | | |
4.70% senior notes due 2045 | 4.73 | % | | 2,700 | | | 4.73 | % | | 2,700 | | | | | | |
4.75% senior notes due 2045 | 4.20 | % | | 881 | | | 4.20 | % | | 881 | | | | | | |
4.45% senior notes due 2046 | 4.50 | % | | 2,000 | | | 4.50 | % | | 2,000 | | | | | | |
4.875% senior notes due 2048 | 4.94 | % | | 1,750 | | | 4.94 | % | | 1,750 | | | | | | |
4.25% senior notes due 2049 | 4.29 | % | | 5,750 | | | 4.29 | % | | 5,750 | | | | | | |
| Fair value hedges | | | (346) | | | | | 102 | | | | | | |
| Unamortized bond discounts | | | (116) | | | | | (130) | | | | | | |
| Unamortized deferred financing costs | | | (222) | | | | | (251) | | | | | | |
Unamortized bond premiums (b) | | | 793 | | | | | 954 | | | | | | |
| Other | | | 33 | | | | | 33 | | | | | | |
| Total long-term debt and finance lease obligations | | | 63,270 | | | | | 76,670 | | | | | | |
| Current portion | | | 4,135 | | | | | 12,481 | | | | | | |
| Noncurrent portion | | | $ | 59,135 | | | | | $ | 64,189 | | | | | | |
(a)Excludes the effect of any related interest rate swaps.
(b)Represents unamortized purchase price adjustments of Allergan debt.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 72 |
Senior notes and floating rate term loans are redeemable prior to maturity at a redemption price equal to the principal amount plus a make-whole premium and AbbVie may redeem these debt securities at par generally between one and six months prior to maturity. At December 31, 2022, the company was in compliance with its senior note covenants and term loan covenants.
Maturities of Long-Term Debt
| | | | | |
| as of and for the years ending December 31 (in millions) | |
| 2023 | $ | 4,132 | |
| 2024 | 7,070 | |
| 2025 | 8,771 | |
| 2026 | 6,000 | |
| 2027 | 798 | |
| Thereafter | 36,357 | |
| Total obligations and commitments | 63,128 | |
| Fair value hedges, unamortized bond premiums/discounts, deferred financing costs and finance lease obligations | 142 | |
| Total long-term debt and finance lease obligations | $ | 63,270 | |
Repayment and Issuance of Long-Term Debt
In 2022, the company repaid $2.9 billion aggregate principal amount of 3.450% senior notes, $1.7 billion aggregate principal amount of 3.25% senior notes, $1.0 billion aggregate principal amount of 3.2% senior notes. These repayments were made by exercising, under the terms of the notes ranging between 60 and 90-day early redemptions at 100% of the principal amount. During the quarter ended December 31, 2022, the company also paid $3.1 billion aggregate principal amount of 2.9% senior notes, $3.0 billion aggregate principal amount of 2.3% senior notes and $750 million aggregate principal amount of floating rate senior notes at maturity. Additionally in 2022, the company refinanced its $2.0 billion floating rate five-year term loan. As part of the refinancing, the company repaid the existing $2.0 billion term loan due May 2025 and borrowed $2.0 billion under a new term loan at a lower floating rate. All other significant terms of the loan, including the maturity date, remained unchanged after the refinancing.
Subsequent to December 31, 2022, the company repaid a $1.0 billion floating rate three-year term loan that was scheduled to mature in May 2023.
In 2021, the company repaid $1.8 billion aggregate principal amount of 2.3% senior notes, €750 million aggregate principal amount of 0.5% senior Euro notes and $1.2 billion aggregate principal amount of 5.0% senior notes. These repayments were made by exercising, under the terms of the notes, ranging between 30 and 90-day early redemptions at 100% of the principal amounts. The company also repaid $1.3 billion aggregate principal amount of 3.375% senior notes $1.8 billion aggregate principal amount of 2.15% senior notes and $1.5 billion aggregate principal amount of floating rate senior notes at maturity. Additionally in 2021, the company refinanced its $1.0 billion floating rate three-year term loan. As part of the refinancing, the company repaid the existing $1.0 billion term loan due May 2023 and borrowed $1.0 billion under a new term loan at a lower floating rate. All other significant terms of the loan, including the maturity date, remained unchanged after the refinancing.
Short-Term Borrowings
There were no commercial paper borrowings outstanding as of December 31, 2022 and December 31, 2021. No commercial paper borrowings were issued during 2022 or 2021. There were commercial paper borrowings issued during 2020 and the weighted-average interest rate was 1.8%. AbbVie currently has a $4.0 billion five-year revolving credit facility that matures in August 2024. This credit facility enables the company to borrow funds on an unsecured basis at variable interest rates and contains various covenants, all of which the company was in compliance with as of December 31, 2022. Commitment fees under AbbVie's revolving credit facilities were insignificant in 2022, 2021 and 2020. No amounts were outstanding under the company's credit facilities as of December 31, 2022 and December 31, 2021.
Contingencies and Guarantees
In connection with the separation, AbbVie has indemnified Abbott for all liabilities resulting from the operation of AbbVie's business other than income tax liabilities with respect to periods prior to the distribution date and other liabilities as agreed to by AbbVie and Abbott. AbbVie has no material exposures to off-balance sheet arrangements and no special-purpose entities. In the ordinary course of business, AbbVie has periodically entered into third-party agreements, such as the assignment of product rights, which have resulted in AbbVie becoming secondarily liable for obligations for which AbbVie had previously been primarily liable. Based upon past experience, the likelihood of payments under these agreements is remote.
| | | | | | | | | | | |
73 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Note 11 Financial Instruments and Fair Value Measures Risk Management Policy
The company is exposed to foreign currency exchange rate and interest rate risks related to its business operations. AbbVie's hedging policy attempts to manage these risks to an acceptable level based on the company's judgment of the appropriate trade-off between risk, opportunity and costs. The company uses derivative and nonderivative instruments to reduce its exposure to foreign currency exchange rates. AbbVie also periodically enters into interest rate swaps in which the company agrees to exchange, at specified intervals, the difference between fixed and floating interest amounts calculated by reference to an agreed-upon notional amount. Derivative instruments are not used for trading purposes or to manage exposure to changes in interest rates for investment securities, and none of the company's outstanding derivative instruments contain credit risk related contingent features; collateral is generally not required.
Financial Instruments
Various AbbVie foreign subsidiaries enter into foreign currency forward exchange contracts to manage exposures to changes in foreign exchange rates for anticipated intercompany transactions denominated in a currency other than the functional currency of the local entity. These contracts, with notional amounts totaling $1.7 billion at December 31, 2022 and $1.1 billion at December 31, 2021, are designated as cash flow hedges and are recorded at fair value. The durations of these forward exchange contracts were generally less than 18 months. Accumulated gains and losses as of December 31, 2022 will be reclassified from AOCI and included in cost of products sold at the time the products are sold, generally not exceeding six months from the date of settlement.
In 2019, the company entered into treasury rate lock agreements with notional amounts totaling $10.0 billion to hedge exposure to variability in future cash flows resulting from changes in interest rates related to the issuance of long-term debt in connection with the acquisition of Allergan. The treasury rate lock agreements were designated as cash flow hedges and recorded at fair value. The agreements were net settled upon issuance of the senior notes in 2019 and the resulting net gain was recognized in other comprehensive income. This gain is reclassified to interest expense, net over the term of the related debt.
The company was a party to interest rate swap contracts designated as cash flow hedges that matured in November 2022, for which the notional amount was $750 million at December 31, 2021. The effect of the hedge contracts was to change a floating-rate interest obligation to a fixed rate for that portion of the floating-rate debt. Realized and unrealized gains or losses were included in AOCI and were reclassified to interest expense, net over the lives of the floating-rate debt.
The company also enters into foreign currency forward exchange contracts to manage its exposure to foreign currency denominated trade payables and receivables and intercompany loans. These contracts are not designated as hedges and are recorded at fair value. Resulting gains or losses are reflected in net foreign exchange loss in the consolidated statements of earnings and are generally offset by losses or gains on the foreign currency exposure being managed. These contracts had notional amounts totaling $6.5 billion at December 31, 2022 and $8.2 billion at December 31, 2021.
The company also uses foreign currency forward exchange contracts or foreign currency denominated debt to hedge its net investments in certain foreign subsidiaries and affiliates. The company had an aggregate principal amount of senior Euro notes designated as net investment hedges of €5.9 billion at December 31, 2022 and December 31, 2021. In addition, the company had foreign currency forward exchange contracts designated as net investment hedges with notional amounts totaling €4.3 billion, SEK2.0 billion, CAD750 million and CHF90 million at December 31, 2022 and €4.3 billion at December 31, 2021. The company uses the spot method of assessing hedge effectiveness for derivative instruments designated as net investment hedges. Realized and unrealized gains and losses from these hedges are included in AOCI and the initial fair value of hedge components excluded from the assessment of effectiveness is recognized in interest expense, net over the life of the hedging instrument.
The company is a party to interest rate swap contracts designated as fair value hedges with notional amounts totaling $4.5 billion at December 31, 2022 and December 31, 2021. The effect of the hedge contracts is to change a fixed-rate interest obligation to a floating rate for that portion of the debt. AbbVie records the contracts at fair value and adjusts the carrying amount of the fixed-rate debt by an offsetting amount.
No amounts are excluded from the assessment of effectiveness for cash flow hedges or fair value hedges.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 74 |
The following table summarizes the amounts and location of AbbVie's derivative instruments on the consolidated balance sheets: | | | | | | | | | | | | | | | | | | | | | | | |
| Fair value - Derivatives in asset position | | Fair value -
Derivatives in liability position |
| as of December 31 (in millions) | Balance sheet caption | 2022 | 2021 | | Balance sheet caption | 2022 | 2021 |
| Foreign currency forward exchange contracts | | | | | | | |
| Designated as cash flow hedges | Prepaid expenses and other | $ | 49 | | $ | 51 | | | Accounts payable and accrued liabilities | $ | 8 | | $ | 2 | |
| | | | | | | |
| Designated as cash flow hedges | Other assets | 1 | | — | | | Other long-term liabilities | — | | — | |
| Designated as net investment hedges | Prepaid expenses and other | 6 | | 149 | | | Accounts payable and accrued liabilities | 36 | | — | |
| Designated as net investment hedges | Other assets | 74 | | 15 | | | Other long-term liabilities | 47 | | — | |
| Not designated as hedges | Prepaid expenses and other | 33 | | 26 | | | Accounts payable and accrued liabilities | 41 | | 13 | |
| Interest rate swap contracts | | | | | | | |
| Designated as cash flow hedges | Prepaid expenses and other | — | | — | | | Accounts payable and accrued liabilities | — | | 7 | |
| Designated as fair value hedges | Prepaid expenses and other | — | | — | | | Accounts payable and accrued liabilities | 17 | | — | |
| Designated as fair value hedges | Other assets | — | | 26 | | | Other long-term liabilities | 375 | | 15 | |
| Total derivatives | | $ | 163 | | $ | 267 | | | | $ | 524 | | $ | 37 | |
While certain derivatives are subject to netting arrangements with the company's counterparties, the company does not offset derivative assets and liabilities within the consolidated balance sheets.
The following table presents the pre-tax amounts of gains (losses) from derivative instruments recognized in other comprehensive income: | | | | | | | | | | | | | | | | | | | | |
| years ended in December 31 (in millions) | | 2022 | | 2021 | | 2020 |
| Foreign currency forward exchange contracts | | | | | | |
| Designated as cash flow hedges | | $ | 103 | | | $ | 82 | | | $ | (71) | |
| Designated as net investment hedges | | 395 | | | 341 | | | (95) | |
| Interest rate swap contracts designated as cash flow hedges | | 6 | | | 2 | | | (53) | |
Assuming market rates remain constant through contract maturities, the company expects to reclassify pre-tax gains of $86 million into cost of products sold for foreign currency cash flow hedges and pre-tax gains of $24 million into interest expense, net for treasury rate lock agreement cash flow hedges during the next 12 months.
Related to AbbVie’s non-derivative, foreign currency denominated debt designated as net investment hedges, the company recognized in other comprehensive income pre-tax gains of $406 million in 2022, pre-tax gains of $577 million in 2021 and pre-tax losses of $907 million in 2020.
| | | | | | | | | | | |
75 |  | 2022 Form 10-K | 2022 Form 10-K | | |
The following table summarizes the pre-tax amounts and location of derivative instrument net gains (losses) recognized in the consolidated statements of earnings, including the net gains (losses) reclassified out of AOCI into net earnings. See Note 13 for the amount of net gains (losses) reclassified out of AOCI. | | | | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | Statement of earnings caption | 2022 | | 2021 | | 2020 |
| Foreign currency forward exchange contracts | | | | | | |
| Designated as cash flow hedges | Cost of products sold | $ | 82 | | | $ | (87) | | | $ | 23 | |
| Designated as net investment hedges | Interest expense, net | 94 | | | 26 | | | 18 | |
| Not designated as hedges | Net foreign exchange loss | (156) | | | (100) | | | 58 | |
| Treasury rate lock agreements designated as cash flow hedges | Interest expense, net | 23 | | | 24 | | | 24 | |
| Interest rate swap contracts | | | | | | |
| Designated as cash flow hedges | Interest expense, net | (1) | | | (24) | | | (17) | |
| Designated as fair value hedges | Interest expense, net | (402) | | | (127) | | | 365 | |
| Debt designated as hedged item in fair value hedges | Interest expense, net | 402 | | | 127 | | | (365) | |
Fair Value Measures
The fair value hierarchy consists of the following three levels:
•Level 1—Valuations based on unadjusted quoted prices in active markets for identical assets that the company has the ability to access;
•Level 2—Valuations based on quoted prices for similar instruments in active markets, quoted prices for identical or similar instruments in markets that are not active and model-based valuations in which all significant inputs are observable in the market; and
•Level 3—Valuations using significant inputs that are unobservable in the market and include the use of judgment by the company's management about the assumptions market participants would use in pricing the asset or liability.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 76 |
The following table summarizes the bases used to measure certain assets and liabilities carried at fair value on a recurring basis on the consolidated balance sheet as of December 31, 2022: | | | | | | | | | | | | | | | | | | | | | | | |
| | | Basis of fair value measurement |
| (in millions) | Total | | Quoted prices in active markets for
identical assets
(Level 1) | | Significant other observable
inputs
(Level 2) | | Significant unobservable inputs
(Level 3) |
| Assets | | | | | | | |
| Cash and equivalents | $ | 9,201 | | | $ | 4,201 | | | $ | 5,000 | | | $ | — | |
| Money market funds and time deposits | 21 | | | — | | | 21 | | | — | |
| Debt securities | 28 | | | — | | | 28 | | | — | |
| Equity securities | 91 | | | 59 | | | 32 | | | — | |
| | | | | | | |
| Foreign currency contracts | 163 | | | — | | | 163 | | | — | |
| | | | | | | |
| Total assets | $ | 9,504 | | | $ | 4,260 | | | $ | 5,244 | | | $ | — | |
| Liabilities | | | | | | | |
| Interest rate swap contracts | $ | 392 | | | $ | — | | | $ | 392 | | | $ | — | |
| Foreign currency contracts | 132 | | | — | | | 132 | | | — | |
| Contingent consideration | 16,384 | | | — | | | — | | | 16,384 | |
| Total liabilities | $ | 16,908 | | | $ | — | | | $ | 524 | | | $ | 16,384 | |
The following table summarizes the bases used to measure certain assets and liabilities carried at fair value on a recurring basis on the consolidated balance sheet as of December 31, 2021: | | | | | | | | | | | | | | | | | | | | | | | |
| | | Basis of fair value measurement |
| (in millions) | Total | | Quoted prices in active markets for identical assets
(Level 1) | | Significant other observable
inputs
(Level 2) | | Significant unobservable inputs
(Level 3) |
| Assets | | | | | | | |
| Cash and equivalents | $ | 9,746 | | | $ | 4,451 | | | $ | 5,295 | | | $ | — | |
| Money market funds and time deposits | 45 | | | — | | | 45 | | | — | |
| Debt securities | 46 | | | — | | | 46 | | | — | |
| Equity securities | 121 | | | 100 | | | 21 | | | — | |
| Interest rate swap contracts | 26 | | | — | | | 26 | | | — | |
| Foreign currency contracts | 241 | | | — | | | 241 | | | — | |
| Total assets | $ | 10,225 | | | $ | 4,551 | | | $ | 5,674 | | | $ | — | |
| Liabilities | | | | | | | |
| Interest rate swap contracts | $ | 22 | | | $ | — | | | $ | 22 | | | $ | — | |
| Foreign currency contracts | 15 | | | — | | | 15 | | | — | |
| Contingent consideration | 14,887 | | | — | | | — | | | 14,887 | |
| Total liabilities | $ | 14,924 | | | $ | — | | | $ | 37 | | | $ | 14,887 | |
Money market funds and time deposits are valued using relevant observable market inputs including quoted prices for similar assets and interest rate curves. Equity securities primarily consist of investments for which the fair values were determined by using the published market prices per unit multiplied by the number of units held, without consideration of transaction costs. The derivatives entered into by the company were valued using observable market inputs including published interest rate curves and both forward and spot prices for foreign currencies.
The fair value measurements of the contingent consideration liabilities were determined based on significant unobservable inputs, including the discount rate, estimated probabilities and timing of achieving specified development, regulatory and commercial milestones and the estimated amount of future sales of the acquired products. The potential contingent consideration payments are estimated by applying a probability-weighted expected payment model for contingent milestone payments and a Monte Carlo simulation model for contingent royalty payments, which are then discounted to present value. Changes to the fair value of the contingent consideration liabilities can result from changes to one or a number of inputs, including discount rates, the probabilities of achieving the milestones, the time required to achieve the milestones
| | | | | | | | | | | |
77 |  | 2022 Form 10-K | 2022 Form 10-K | | |
and estimated future sales. Significant judgment is employed in determining the appropriateness of certain of these inputs. Changes to the inputs described above could have a material impact on the company's financial position and results of operations in any given period.
The fair value of the company's contingent consideration liabilities was calculated using the following significant unobservable inputs: | | | | | | | | | | | | | | | | | |
| 2022 | | 2021 |
| years ended December 31 (in millions) | Range | Weighted Average(a) | | Range | Weighted Average(a) |
| Discount rate | 4.7% - 5.1% | 4.8% | | 0.2% - 2.6% | 1.7 | % |
| Probability of payment for unachieved milestones | 100% - 100% | 100% | | 89% - 100% | 90 | % |
Probability of payment for royalties by indication(b) | 56% - 100% | 99% | | 56% - 100% | 96 | % |
| | | | | |
| Projected year of payments | 2023 - 2034 | 2028 | | 2022 - 2034 | 2027 |
(a)Unobservable inputs were weighted by the relative fair value of the contingent consideration liabilities.
(b)Excluding approved indications, the estimated probability of payment was 56% at December 31, 2022 and ranged from 56% to 89% at December 31, 2021.
There have been no transfers of assets or liabilities into or out of Level 3 of the fair value hierarchy. The following table presents the changes in fair value of contingent consideration liabilities which are measured using Level 3 inputs: | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Beginning balance | $ | 14,887 | | | $ | 12,997 | | | $ | 7,340 | |
Additions(a) | 32 | | | — | | | 225 | |
| Change in fair value recognized in net earnings | 2,761 | | | 2,679 | | | 5,753 | |
| Payments | (1,296) | | | (789) | | | (321) | |
| Ending balance | $ | 16,384 | | | $ | 14,887 | | | $ | 12,997 | |
(a)Additions during the year ended December 31, 2022, represent contingent consideration liabilities assumed in the DJS acquisition. Additions during the year ended December 31, 2020, represent contingent consideration liabilities assumed in the Allergan and Luminera acquisitions (see Note 5).
The change in fair value recognized in net earnings is recorded in other expense, net in the consolidated statements of earnings and included charges of $2.8 billion in 2022, $2.7 billion in 2021 and $5.8 billion in 2020. In 2022, the change in fair value reflected higher estimated Skyrizi sales driven by stronger market share uptake and the passage of time, partially offset by higher discount rates. In 2021, the change in fair value reflected higher estimated Skyrizi sales driven by stronger market share uptake, favorable clinical trial results and the passage of time, partially offset by higher discount rates. In 2020, the change in fair value reflected higher estimated Skyrizi sales driven by stronger market share uptake, lower discount rates, the passage of time and favorable clinical trial results.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 78 |
Certain financial instruments are carried at historical cost or some basis other than fair value. The book values, approximate fair values and bases used to measure the approximate fair values of certain financial instruments as of December 31, 2022 are shown in the table below: | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | Basis of fair value measurement |
| (in millions) | Book value | Approximate fair values | | Quoted prices in active markets for identical assets
(Level 1) | | Significant other observable inputs
(Level 2) | | Significant unobservable inputs
(Level 3) |
| Liabilities | | | | | | | | |
| Short-term borrowings | $ | 1 | | $ | 1 | | | $ | — | | | $ | 1 | | | $ | — | |
| Current portion of long-term debt and finance lease obligations, excluding fair value hedges | 4,152 | | 4,121 | | | 3,930 | | | 191 | | | — | |
| Long-term debt and finance lease obligations, excluding fair value hedges | 59,463 | | 54,073 | | | 53,365 | | | 708 | | | — | |
| Total liabilities | $ | 63,616 | | $ | 58,195 | | | $ | 57,295 | | | $ | 900 | | | $ | — | |
The book values, approximate fair values and bases used to measure the approximate fair values of certain financial instruments as of December 31, 2021 are shown in the table below: | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | Basis of fair value measurement |
| (in millions) | Book value | Approximate fair values | | Quoted prices in active markets for identical assets
(Level 1) | | Significant other observable inputs
(Level 2) | | Significant unobservable inputs
(Level 3) |
| Liabilities | | | | | | | | |
| Short-term borrowings | $ | 14 | | $ | 14 | | | $ | — | | | $ | 14 | | | $ | — | |
| Current portion of long-term debt and finance lease obligations, excluding fair value hedges | $ | 12,455 | | $ | 11,830 | | | $ | 11,329 | | | $ | 501 | | | $ | — | |
| Long-term debt and finance lease obligations, excluding fair value hedges | 64,113 | | 71,810 | | | 70,757 | | | 1,053 | | | — | |
| Total liabilities | $ | 76,582 | | $ | 83,654 | | | $ | 82,086 | | | $ | 1,568 | | | $ | — | |
AbbVie also holds investments in equity securities that do not have readily determinable fair values. The company records these investments at cost and remeasures them to fair value based on certain observable price changes or impairment events as they occur. The carrying amount of these investments was $129 million as of December 31, 2022 and $149 million as of December 31, 2021. No significant cumulative upward or downward adjustments have been recorded for these investments as of December 31, 2022.
Concentrations of Risk
Of total net accounts receivable, three U.S. wholesalers accounted for 82% as of December 31, 2022 and 75% as of December 31, 2021, and substantially all of AbbVie's pharmaceutical product net revenues in the United States were to these three wholesalers.
Humira (adalimumab) is AbbVie's single largest product and accounted for approximately 37% of AbbVie's total net revenues in 2022, 37% in 2021 and 43% in 2020.
| | | | | | | | | | | |
79 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Note 12 Post-Employment Benefits AbbVie sponsors various pension and other post-employment benefit plans, including defined benefit, defined contribution and termination indemnity plans, which cover most employees worldwide. In addition, AbbVie provides medical benefits, primarily to eligible retirees in the United States and Puerto Rico, through other post-retirement benefit plans. Net obligations for these plans have been reflected on the consolidated balance sheets as of December 31, 2022 and 2021.
The following table summarizes benefit plan information for the global AbbVie-sponsored defined benefit and other post-employment plans: | | | | | | | | | | | | | | | | | | | | | | | |
| Defined benefit plans | | Other post-employment plans |
| as of and for the years ended December 31 (in millions) | 2022 | | 2021 | | 2022 | | 2021 |
| Projected benefit obligations | | | | | | | |
| Beginning of period | $ | 12,006 | | | $ | 11,792 | | | $ | 850 | | | $ | 795 | |
| Service cost | 454 | | | 440 | | | 51 | | | 48 | |
| Interest cost | 297 | | | 237 | | | 23 | | | 19 | |
| Employee contributions | 1 | | | 2 | | | — | | | — | |
| Amendments | — | | | — | | | (2) | | | — | |
| Actuarial (gain) loss | (3,668) | | | (8) | | | (229) | | | 10 | |
| Benefits paid | (294) | | | (281) | | | (25) | | | (22) | |
| | | | | | | |
| | | | | | | |
| Other, primarily foreign currency translation adjustments | (208) | | | (176) | | | (1) | | | — | |
| End of period | 8,588 | | | 12,006 | | | 667 | | | 850 | |
| Fair value of plan assets | | | | | | | |
| Beginning of period | 10,655 | | | 9,702 | | | — | | | — | |
| Actual return on plan assets | (2,031) | | | 1,000 | | | — | | | — | |
| Company contributions | 357 | | | 376 | | | 25 | | | 22 | |
| Employee contributions | 1 | | | 2 | | | — | | | — | |
| Benefits paid | (294) | | | (281) | | | (25) | | | (22) | |
| | | | | | | |
| | | | | | | |
| Other, primarily foreign currency translation adjustments | (216) | | | (144) | | | — | | | — | |
| End of period | 8,472 | | | 10,655 | | | — | | | — | |
| Funded status, end of period | $ | (116) | | | $ | (1,351) | | | $ | (667) | | | $ | (850) | |
| | | | | | | |
| Amounts recognized on the consolidated balance sheets | | | | | | | |
| Other assets | $ | 896 | | | $ | 991 | | | $ | — | | | $ | — | |
| Accounts payable and accrued liabilities | (14) | | | (13) | | | (27) | | | (26) | |
| Other long-term liabilities | (998) | | | (2,329) | | | (640) | | | (824) | |
| Net obligation | $ | (116) | | | $ | (1,351) | | | $ | (667) | | | $ | (850) | |
| Actuarial loss, net | $ | 2,365 | | | $ | 3,504 | | | $ | 205 | | | $ | 461 | |
| Prior service cost (credit) | 3 | | | 5 | | | (333) | | | (370) | |
| Accumulated other comprehensive loss | $ | 2,368 | | | $ | 3,509 | | | $ | (128) | | | $ | 91 | |
Related to international defined benefit plans the projected benefit obligations in the table above included $2.1 billion at December 31, 2022 and $3.2 billion at December 31, 2021.
For plans reflected in the table above, the accumulated benefit obligations were $7.7 billion at December 31, 2022 and $10.5 billion at December 31, 2021.
The 2022 actuarial gain of $3.7 billion for qualified pension plans and actuarial gain of $229 million for other post-employment plans were primarily driven by an increase in the discount rate. The 2021 actuarial gain of $8 million for qualified pension plans and actuarial loss of $10 million for other post-employment plans were primarily driven by an increase in the assumed discount rate offset by change in demographic assumptions from 2020.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 80 |
Information For Pension Plans With An Accumulated Benefit Obligation In Excess Of Plan Assets | | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| Accumulated benefit obligation | $ | 1,211 | | | $ | 6,395 | |
| Fair value of plan assets | 746 | | | 5,412 | |
Information For Pension Plans With A Projected Benefit Obligation In Excess Of Plan Assets | | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| Projected benefit obligation | $ | 5,592 | | | $ | 7,788 | |
| Fair value of plan assets | 4,580 | | | 5,447 | |
AbbVie's U.S. pension plan was modified to close the plan to new entrants effective January 1, 2022. In addition, a change to AbbVie's U.S. retiree health benefit plan was approved in 2020 and communicated to employees and retirees in October 2020. Beginning in 2022, Medicare-eligible retirees and Medicare-eligible dependents choose health care coverage from insurance providers through a private Medicare exchange. AbbVie will continue to provide financial support to Medicare-eligible retirees. This change to the U.S. retiree health benefit plan decreased AbbVie's post-employment benefit obligation and increased AbbVie's unrecognized prior service credit as of December 31, 2020 by $397 million.
| | | | | | | | | | | |
81 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Amounts Recognized in Other Comprehensive Income
The following table summarizes the pre-tax losses (gains) included in other comprehensive income: | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Defined benefit plans | | | | | |
| Actuarial loss (gain) | $ | (925) | | | $ | (345) | | | $ | 701 | |
| | | | | |
| Amortization of prior service cost | (2) | | | (2) | | | (2) | |
| Amortization of actuarial loss | (231) | | | (288) | | | (227) | |
| Foreign exchange loss (gain) and other | 17 | | | (27) | | | 56 | |
| Total loss (gain) | $ | (1,141) | | | $ | (662) | | | $ | 528 | |
| Other post-employment plans | | | | | |
| Actuarial loss (gain) | $ | (229) | | | $ | 10 | | | $ | 40 | |
| Prior service credit | (2) | | | — | | | (397) | |
| Amortization of prior service credit | 38 | | | 39 | | | 4 | |
| Amortization of actuarial loss | (26) | | | (32) | | | (26) | |
| Total loss (gain) | $ | (219) | | | $ | 17 | | | $ | (379) | |
Net Periodic Benefit Cost | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Defined benefit plans | | | | | |
| Service cost | $ | 454 | | | $ | 440 | | | $ | 370 | |
| Interest cost | 297 | | | 237 | | | 264 | |
| Expected return on plan assets | (712) | | | (663) | | | (575) | |
| Amortization of prior service cost | 2 | | | 2 | | | 2 | |
| Amortization of actuarial loss | 231 | | | 288 | | | 227 | |
| Net periodic benefit cost | $ | 272 | | | $ | 304 | | | $ | 288 | |
| Other post-employment plans | | | | | |
| Service cost | $ | 51 | | | $ | 48 | | | $ | 42 | |
| Interest cost | 23 | | | 19 | | | 34 | |
| Amortization of prior service credit | (38) | | | (39) | | | (4) | |
| Amortization of actuarial loss | 26 | | | 32 | | | 26 | |
| Net periodic benefit cost | $ | 62 | | | $ | 60 | | | $ | 98 | |
The components of net periodic benefit cost other than service cost are included in other expense, net in the consolidated statements of earnings.
Weighted-Average Assumptions Used in Determining Benefit Obligations at the Measurement Date | | | | | | | | | | | |
| as of December 31 | 2022 | | 2021 |
| Defined benefit plans | | | |
| Discount rate | 5.0 | % | | 2.8 | % |
| Rate of compensation increases | 5.5 | % | | 5.2 | % |
| Cash balance interest crediting rate | 2.7 | % | | 2.7 | % |
| Other post-employment plans | | | |
| Discount rate | 5.3 | % | | 3.1 | % |
The assumptions used in calculating the December 31, 2022 measurement date benefit obligations will be used in the calculation of net periodic benefit cost in 2023.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 82 |
Weighted-Average Assumptions Used in Determining Net Periodic Benefit Cost | | | | | | | | | | | | | | | | | |
| years ended December 31 | 2022 | | 2021 | | 2020 |
| Defined benefit plans | | | | | |
| Discount rate for determining service cost | 3.0 | % | | 2.6 | % | | 3.1 | % |
| Discount rate for determining interest cost | 2.6 | % | | 2.2 | % | | 3.0 | % |
| Expected long-term rate of return on plan assets | 7.1 | % | | 7.1 | % | | 7.1 | % |
| Expected rate of change in compensation | 5.2 | % | | 4.6 | % | | 4.6 | % |
| Cash balance interest crediting rate | 2.7 | % | | 2.8 | % | | 2.8 | % |
| Other post-employment plans | | | | | |
| Discount rate for determining service cost | 3.3 | % | | 3.0 | % | | 3.7 | % |
| Discount rate for determining interest cost | 2.7 | % | | 2.2 | % | | 3.2 | % |
For the December 31, 2022 post-retirement health care obligations remeasurement, the company assumed a 6.2% pre-65 (2.0% post-65) annual rate of increase in the per capita cost of covered health care benefits. The pre-65 rate was assumed to decrease gradually to 4.5% (1.8% post-65) in 2030 and remain at that level thereafter. For purposes of measuring the 2022 post-retirement health care costs, the company assumed a 5.9% pre-65 (2.1% post-65) annual rate of increase in the per capita cost of covered health care benefits. The pre-65 rate was assumed to decrease gradually to 4.5% (1.8% post-65) for 2029 and remain at that level thereafter.
| | | | | | | | | | | |
83 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Defined Benefit Pension Plan Assets | | | | | | | | | | | | | | | | | | | | | | | |
| | | Basis of fair value measurement |
| as of December 31 (in millions) | 2022 | | Quoted prices in active markets for identical assets
(Level 1) | | Significant other observable inputs
(Level 2) | | Significant unobservable inputs
(Level 3) |
| Equities | | | | | | | |
U.S. large cap(a) | $ | 949 | | | $ | 949 | | | $ | — | | | $ | — | |
U.S. mid cap(b) | 157 | | | 157 | | | — | | | — | |
International(c) | 327 | | | 327 | | | — | | | — | |
| Fixed income securities | | | | | | | |
U.S. government securities(d) | 237 | | | 69 | | | 168 | | | — | |
Corporate debt instruments(d) | 680 | | | 144 | | | 536 | | | — | |
Non-U.S. government securities(d) | 548 | | | 402 | | | 146 | | | — | |
Other(d) | 84 | | | 81 | | | 3 | | | — | |
Absolute return funds(e) | 91 | | | 4 | | | 87 | | | — | |
| Real assets | 9 | | | 9 | | | — | | | — | |
Other(f) | 278 | | | 277 | | | 1 | | | — | |
| Total | $ | 3,360 | | | $ | 2,419 | | | $ | 941 | | | $ | — | |
| Total assets measured at NAV | 5,112 | | | | | | | |
| Fair value of plan assets | $ | 8,472 | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | |
| | | Basis of fair value measurement |
| as of December 31 (in millions) | 2021 | | Quoted prices in active markets for identical assets
(Level 1) | | Significant other observable inputs
(Level 2) | | Significant unobservable inputs
(Level 3) |
| Equities | | | | | | | |
U.S. large cap(a) | $ | 1,428 | | | $ | 1,428 | | | $ | — | | | $ | — | |
U.S. mid cap(b) | 198 | | | 198 | | | — | | | — | |
International(c) | 458 | | | 458 | | | — | | | — | |
| Fixed income securities | | | | | | | |
U.S. government securities(d) | 228 | | | 95 | | | 133 | | | — | |
Corporate debt instruments(d) | 945 | | | 179 | | | 766 | | | — | |
Non-U.S. government securities(d) | 602 | | | 445 | | | 157 | | | — | |
Other(d) | 273 | | | 268 | | | 5 | | | — | |
Absolute return funds(e) | 100 | | | 5 | | | 95 | | | — | |
| Real assets | 10 | | | 10 | | | — | | | — | |
Other(f) | 261 | | | 216 | | | 45 | | | — | |
| Total | $ | 4,503 | | | $ | 3,302 | | | $ | 1,201 | | | $ | — | |
| Total assets measured at NAV | 6,152 | | | | | | | |
| Fair value of plan assets | $ | 10,655 | | | | | | | |
(a)A mix of index funds and actively managed equity accounts that are benchmarked to various large cap indices.
(b)A mix of index funds and actively managed equity accounts that are benchmarked to various mid cap indices.
(c)A mix of index funds and actively managed equity accounts that are benchmarked to various non-U.S. equity indices in both developed and emerging markets.
(d)Securities held by actively managed accounts, index funds and mutual funds.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 84 |
(e)Primarily funds having global mandates with the flexibility to allocate capital broadly across a wide range of asset classes and strategies, including but not limited to equities, fixed income, commodities, financial futures, currencies and other securities, with objectives to outperform agreed upon benchmarks of specific return and volatility targets.
(f)Investments in cash and cash equivalents.
Equities and registered investment companies having quoted prices are valued at the published market prices. Fixed income securities that are valued using significant other observable inputs are quoted at prices obtained from independent financial service industry-recognized vendors. Investments held in pooled investment funds, common collective trusts or limited partnerships are valued at the net asset value (NAV) practical expedient to estimate fair value. The NAV is provided by the fund administrator and is based on the value of the underlying assets owned by the fund minus its liabilities.
The investment mix of equity securities, fixed income and other asset allocation strategies is based upon achieving a desired return, balancing higher return, more volatile equity securities and lower return, less volatile fixed income securities. Investment allocations are established for each plan and are generally made across a range of markets, industry sectors, capitalization sizes and in the case of fixed income securities, maturities and credit quality. The 2022 target investment allocation for the AbbVie Pension Plan was 62.5% in equity securities, 22.5% in fixed income securities and 15% in asset allocation strategies and other holdings. There are no known significant concentrations of risk in the plan assets of the AbbVie Pension Plan or of any other plans.
The expected return on plan assets assumption for each plan is based on management's expectations of long-term average rates of return to be achieved by the underlying investment portfolio. In establishing this assumption, management considers historical and expected returns for the asset classes in which the plans are invested, as well as current economic and capital market conditions.
Expected Benefit Payments
The following table summarizes total benefit payments expected to be paid to plan participants including payments funded from both plan and company assets: | | | | | | | | | | | |
| years ending December 31 (in millions) | Defined
benefit plans | | Other
post-employment plans |
| 2023 | $ | 310 | | | $ | 28 | |
| 2024 | 333 | | | 31 | |
| 2025 | 355 | | | 34 | |
| 2026 | 378 | | | 37 | |
| 2027 | 404 | | | 39 | |
| 2028 to 2032 | 2,427 | | | 244 | |
Defined Contribution Plan
AbbVie maintains defined contribution savings plans for the benefit of its eligible employees. The expense recognized for these plans was $474 million in 2022, $267 million in 2021 and $191 million in 2020. AbbVie provides certain other post-employment benefits, primarily salary continuation arrangements, to qualifying employees and accrues for the related cost over the service lives of the employees.
Stock-Based Compensation
In May 2021, stockholders of the company approved the AbbVie Amended and Restated 2013 Incentive Stock Program (the Amended Plan), which amends and restates the AbbVie 2013 Incentive Stock Program (2013 ISP). AbbVie grants stock-based awards to eligible employees pursuant to the Amended Plan, which provides for several different forms of benefits, including non-qualified stock options, RSUs and various performance-based awards. Under the Amended Plan, a total of 144 million shares of AbbVie common stock have been reserved for issuance as awards to AbbVie employees.
| | | | | | | | | | | |
85 |  | 2022 Form 10-K | 2022 Form 10-K | | |
AbbVie measures compensation expense for stock-based awards based on the grant date fair value of the awards and the estimated number of awards that are expected to vest. Forfeitures are estimated based on historical experience at the time of grant and are revised in subsequent periods if actual forfeitures differ from those estimates. Compensation cost for stock-based awards is amortized over the service period, which could be shorter than the vesting period if an employee is retirement eligible. Retirement eligible employees generally are those who are age 55 or older and have at least 10 years of service.
Stock-based compensation expense is principally related to awards issued pursuant to the 2013 ISP and the Amended Plan and is summarized as follows: | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Cost of products sold | $ | 38 | | | $ | 46 | | | $ | 47 | |
| Research and development | 232 | | | 226 | | | 247 | |
| Selling, general and administrative | 401 | | | 420 | | | 459 | |
| Pre-tax compensation expense | 671 | | | 692 | | | 753 | |
| Tax benefit | 122 | | | 126 | | | 131 | |
| After-tax compensation expense | $ | 549 | | | $ | 566 | | | $ | 622 | |
Realized excess tax benefits associated with stock-based compensation totaled $116 million in 2022, $50 million in 2021 and $34 million in 2020.
Stock Options
Stock options awarded to employees typically have a contractual term of 10 years and generally vest in one-third increments over a three-year period. The exercise price is equal to at least 100% of the market value on the date of grant. The fair value is determined using the Black-Scholes model. The weighted-average grant-date fair values of stock options granted were $22.83 in 2022, $16.28 in 2021 and $12.14 in 2020.
In connection with the Allergan acquisition, during the second quarter of 2020, AbbVie issued 11.2 million stock options to holders of Allergan options as a result of the conversion of such options. These options were fair-valued using a lattice valuation model. Refer to Note 5 for additional information regarding the Allergan acquisition.
The following table summarizes AbbVie stock option activity in 2022: | | | | | | | | | | | | | | | | | | | | | | | |
| (options in thousands, aggregate intrinsic value in millions) | Options | | Weighted- average
exercise price | | Weighted-average remaining
life (in years) | | Aggregate intrinsic value |
| Outstanding at December 31, 2021 | 12,374 | | | $ | 81.98 | | | 4.7 | | $ | 661 | |
| Granted | 863 | | | 144.54 | | | | | |
| Exercised | (3,764) | | | 70.85 | | | | | |
| Lapsed and forfeited | (153) | | | 107.65 | | | | | |
| Outstanding at December 31, 2022 | 9,320 | | | $ | 91.84 | | | 4.8 | | $ | 650 | |
| Exercisable at December 31, 2022 | 7,120 | | | $ | 84.54 | | | 3.7 | | $ | 549 | |
The total intrinsic value of options exercised was $295 million in 2022, $239 million in 2021 and $186 million in 2020. The total fair value of options vested during 2022 was $21 million. As of December 31, 2022, $6 million of unrecognized compensation cost related to stock options is expected to be recognized as expense over approximately the next two years.
RSUs and Performance Shares
RSUs awarded to employees other than senior executives and other key employees generally vest in ratable increments over a three or four-year period. Recipients of these RSUs are entitled to receive dividend equivalents as dividends are declared and paid during the RSU vesting period.
The majority of the equity awards AbbVie grants to its senior executives and other key employees are performance-based. Equity awards granted to senior executives and other key employees consist of a combination of performance-vested RSUs and performance shares as well as non-qualified stock options described above. The performance-vested RSUs have the potential to vest in one-third increments during a three-year performance period and may be earned based on AbbVie’s return on invested capital (ROIC) performance relative to a defined peer group of pharmaceutical, biotech and life science companies. The recipient may receive one share of AbbVie common stock for each vested award. The performance shares
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 86 |
have the potential to vest over a three-year performance period and may be earned based on AbbVie’s EPS achievement and AbbVie’s total stockholder return (TSR) (a market condition) relative to a defined peer group of pharmaceutical, biotech and life sciences companies. Dividend equivalents on performance-vested RSUs and performance shares accrue during the performance period and are payable at vesting only to the extent that shares are earned.
The weighted-average grant-date fair value of RSUs and performance shares generally is determined based on the number of shares/units granted and the quoted price of AbbVie’s common stock on the date of grant. The weighted-average grant-date fair values of performance shares with a TSR market condition are determined using the Monte Carlo simulation model.
The following table summarizes AbbVie RSU and performance share activity for 2022: | | | | | | | | | | | |
| (share units in thousands) | Share units | | Weighted-average grant date fair value |
| Outstanding at December 31, 2021 | 14,890 | | | $ | 94.93 | |
| Granted | 5,943 | | | 138.73 | |
| Vested | (6,958) | | | 89.31 | |
| Forfeited | (844) | | | 111.45 | |
| Outstanding at December 31, 2022 | 13,031 | | | $ | 116.84 | |
The fair market value of RSUs and performance shares (as applicable) vested was $1.0 billion in 2022, $718 million in 2021 and $618 million in 2020.
In connection with the Allergan acquisition, during the second quarter of 2020, AbbVie issued 8.2 million RSUs to holders of Allergan equity awards based on a conversion factor described in the transaction agreement. Refer to Note 5 for additional information regarding the Allergan acquisition.
As of December 31, 2022, $578 million of unrecognized compensation cost related to RSUs and performance shares is expected to be recognized as expense over approximately the next two years.
Cash Dividends
Cash dividends declared per common share totaled $5.71 in 2022, $5.31 in 2021 and $4.84 in 2020. The following table summarizes quarterly cash dividends declared during 2022, 2021 and 2020: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| 2022 | | 2021 | | 2020 |
| Date Declared | | Payment Date | | Dividend Per Share | | Date Declared | | Payment Date | | Dividend Per Share | | Date Declared | | Payment Date | | Dividend Per Share |
| 10/28/22 | | 02/15/23 | | $1.48 | | 10/29/21 | | 02/15/22 | | $1.41 | | 10/30/20 | | 02/16/21 | | $1.30 |
| 09/09/22 | | 11/15/22 | | $1.41 | | 09/10/21 | | 11/15/21 | | $1.30 | | 09/11/20 | | 11/16/20 | | $1.18 |
| 06/23/22 | | 08/15/22 | | $1.41 | | 06/17/21 | | 08/16/21 | | $1.30 | | 06/17/20 | | 08/14/20 | | $1.18 |
| 02/17/22 | | 05/16/22 | | $1.41 | | 02/18/21 | | 05/14/21 | | $1.30 | | 02/20/20 | | 05/15/20 | | $1.18 |
Stock Repurchase Program
The company's stock repurchase authorization permits purchases of AbbVie shares from time to time in open-market or private transactions at management’s discretion. The program has no time limit and can be discontinued at any time. Shares repurchased under these programs are recorded at acquisition cost, including related expenses and are available for general corporate purposes.
AbbVie repurchased 8 million shares for $1.1 billion in 2022, 6 million shares for $670 million in 2021 and 8 million shares for $757 million in 2020. AbbVie's remaining stock repurchase authorization was $1.4 billion as of December 31, 2022. On February 16, 2023, AbbVie's board of directors authorized a $5.0 billion increase to the existing stock repurchase authorization.
| | | | | | | | | | | |
87 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Accumulated Other Comprehensive Loss
The following table summarizes the changes in each component of accumulated other comprehensive loss, net of tax, for 2022, 2021 and 2020: | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| (in millions) (brackets denote losses) | Foreign currency translation adjustments | | Net investment hedging activities | | Pension
and post-employment benefits | | | | Cash flow hedging activities | | Total |
| Balance as of December 31, 2019 | $ | (928) | | | $ | 9 | | | $ | (2,965) | | | | | $ | 288 | | | $ | (3,596) | |
| Other comprehensive income (loss) before reclassifications | 1,511 | | | (785) | | | (300) | | | | | (108) | | | 318 | |
| Net losses (gains) reclassified from accumulated other comprehensive loss | — | | | (14) | | | 198 | | | | | (23) | | | 161 | |
| Net current-period other comprehensive income (loss) | 1,511 | | | (799) | | | (102) | | | | | (131) | | | 479 | |
| Balance as of December 31, 2020 | 583 | | | (790) | | | (3,067) | | | | | 157 | | | (3,117) | |
| Other comprehensive income (loss) before reclassifications | (1,153) | | | 720 | | | 298 | | | | | 76 | | | (59) | |
| Net losses (gains) reclassified from accumulated other comprehensive loss | — | | | (21) | | | 223 | | | | | 75 | | | 277 | |
| Net current-period other comprehensive income (loss) | (1,153) | | | 699 | | | 521 | | | | | 151 | | | 218 | |
| Balance as of December 31, 2021 | (570) | | | (91) | | | (2,546) | | | | | 308 | | | (2,899) | |
| Other comprehensive income (loss) before reclassifications | (943) | | | 629 | | | 915 | | | | | 91 | | | 692 | |
| Net losses (gains) reclassified from accumulated other comprehensive loss | — | | | (74) | | | 173 | | | | | (91) | | | 8 | |
| Net current-period other comprehensive income (loss) | (943) | | | 555 | | | 1,088 | | | | | — | | | 700 | |
| Balance as of December 31, 2022 | $ | (1,513) | | | $ | 464 | | | $ | (1,458) | | | | | $ | 308 | | | $ | (2,199) | |
Other comprehensive income for 2022 included pension and post-employment benefit plan gains of $1.1 billion primarily due actuarial gains driven by higher discount rates partially offset by losses on plan assets. Other comprehensive income for 2022 also included foreign currency translation adjustments totaling losses of $943 million principally due to the impact of the weakening of the Euro on the translation of the company’s Euro-denominated assets and the offsetting impact of net investment hedging activities totaling gains of $555 million. Other comprehensive income for 2021 included foreign currency translation adjustments totaling losses of $1.2 billion principally due to the impact of the weakening of the Euro on the translation of the company's Euro-denominated assets and the offsetting impact of net investment hedging activities totaling gains of $699 million. Other comprehensive income for 2020 included foreign currency translation adjustments totaling gains of $1.5 billion principally due to the impact of the strengthening of the Euro on the translation of the company's Euro-denominated assets and the offsetting impact of net investment hedging activities totaling losses of $799 million.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 88 |
The table below presents the impact on AbbVie's consolidated statements of earnings for significant amounts reclassified out of each component of accumulated other comprehensive loss: | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) (brackets denote gains) | 2022 | | 2021 | | 2020 |
| Net investment hedging activities | | | | | |
Gains on derivative amount excluded from effectiveness testing(a) | $ | (94) | | | $ | (26) | | | $ | (18) | |
| Tax expense | 20 | | | 5 | | | 4 | |
| Total reclassifications, net of tax | $ | (74) | | | $ | (21) | | | $ | (14) | |
| Pension and post-employment benefits | | | | | |
Amortization of actuarial losses and other(b) | $ | 221 | | | $ | 283 | | | $ | 251 | |
| Tax benefit | (48) | | | (60) | | | (53) | |
| Total reclassifications, net of tax | $ | 173 | | | $ | 223 | | | $ | 198 | |
| Cash flow hedging activities | | | | | |
Losses (gains) on foreign currency forward exchange contracts(c) | $ | (82) | | | $ | 87 | | | $ | (23) | |
Gains on treasury rate lock agreements(a) | (23) | | | (24) | | | (24) | |
Losses on interest rate swap contracts(a) | 1 | | | 24 | | | 17 | |
| Tax expense (benefit) | 13 | | | (12) | | | 7 | |
| Total reclassifications, net of tax | $ | (91) | | | $ | 75 | | | $ | (23) | |
(a)Amounts are included in interest expense, net (see Note 11).
(b)Amounts are included in the computation of net periodic benefit cost (see Note 12).
(c)Amounts are included in cost of products sold (see Note 11).
Other
In addition to common stock, AbbVie's authorized capital includes 200 million shares of preferred stock, par value $0.01. As of December 31, 2022, no shares of preferred stock were issued or outstanding.
| | | | | | | | | | | |
89 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Earnings Before Income Tax Expense | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Domestic | $ | (4,608) | | | $ | (1,644) | | | $ | (4,467) | |
| Foreign | 18,085 | | | 14,633 | | | 7,865 | |
| Total earnings before income tax expense | $ | 13,477 | | | $ | 12,989 | | | $ | 3,398 | |
Income Tax Expense | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Current | | | | | |
| Domestic | $ | 2,647 | | | $ | 1,987 | | | $ | 907 | |
| Foreign | 916 | | | 351 | | | 194 | |
| Total current taxes | $ | 3,563 | | | $ | 2,338 | | | $ | 1,101 | |
| Deferred | | | | | |
| Domestic | $ | (1,512) | | | $ | (839) | | | $ | (58) | |
| Foreign | (419) | | | (59) | | | (2,267) | |
| Total deferred taxes | $ | (1,931) | | | $ | (898) | | | $ | (2,325) | |
| Total income tax expense (benefit) | $ | 1,632 | | | $ | 1,440 | | | $ | (1,224) | |
Effective Tax Rate Reconciliation | | | | | | | | | | | | | | | | | |
| years ended December 31 | 2022 | | 2021 | | 2020 |
| Statutory tax rate | 21.0 | % | | 21.0 | % | | 21.0 | % |
| Effect of foreign operations | (4.4) | | | (5.4) | | | 2.4 | |
| U.S. tax credits | (2.8) | | | (2.8) | | | (10.6) | |
| Impacts related to U.S. tax reform | — | | | — | | | (1.1) | |
| Non-deductible expenses | 0.6 | | | 0.3 | | | 7.2 | |
| Tax law changes and related restructuring | (2.4) | | | (2.0) | | | (48.5) | |
| Tax audits and settlements | 0.9 | | | (0.4) | | | (5.1) | |
| All other, net | (0.8) | | | 0.4 | | | (1.3) | |
| Effective tax rate | 12.1 | % | | 11.1 | % | | (36.0) | % |
The effective income tax rate fluctuates year to year due to the allocation of the company's taxable earnings among jurisdictions, as well as certain discrete factors and events in each year, including changes in tax law, acquisitions and collaborations. The effective income tax rates in 2022, 2021 and 2020 differed from the statutory tax rate principally due to the impact of foreign operations which reflects the impact of lower income tax rates in locations outside the United States, tax incentives in Puerto Rico and other foreign tax jurisdictions, business development activities, changes in enacted tax rates and laws and related restructuring, tax audits and settlements and changes in fair value of contingent consideration. The effective tax rates for these periods also reflected the benefit from U.S. tax credits principally related to research and development credits, the orphan drug tax credit and Puerto Rico excise tax credits. The Puerto Rico tax credits relate to excise tax on certain products manufactured in Puerto Rico. The tax is levied on gross inventory purchases from entities in Puerto Rico and is included in cost of products sold in the consolidated statements of earnings. The majority of the tax is creditable for U.S. income tax purposes.
In 2022, Puerto Rico enacted Act 52-2002 (the Puerto Rico Act) allowing for a transition from a Puerto Rico excise tax levied on gross inventory purchases to an income-based tax beginning in 2023. The company completed the transition requirements of the Puerto Rico Act in 2022, resulting in the remeasurement of certain deferred tax assets and liabilities based on income tax rates at which they are expected to reverse in the future. The net tax benefit from the remeasurement of deferred taxes related to the Puerto Rico Act was $323 million.
The 2020 effective income tax rate included the recognition of a net tax benefit of $1.7 billion related to changes in tax laws and related restructuring, including certain intra-group transfers of intellectual property and deferred tax remeasurement.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 90 |
The Tax Cuts and Jobs Act (the Act) was signed into law in December 2017, resulting in significant changes to the U.S. corporate tax system, including a one-time transition tax on a mandatory deemed repatriation of earnings of certain foreign subsidiaries that were previously untaxed. The Act also created a minimum tax on certain foreign sourced earnings. The company’s accounting policy for the minimum tax on foreign sourced earnings is to report the tax effects on the basis that the minimum tax will be recognized in tax expense in the year it is incurred as a period expense.
Deferred Tax Assets and Liabilities | | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| Deferred tax assets | | | |
| Compensation and employee benefits | $ | 497 | | | $ | 937 | |
| Accruals and reserves | 1,023 | | | 667 | |
| Chargebacks and rebates | 991 | | | 837 | |
| Advance payments | 547 | | | 809 | |
| Net operating losses and other carryforwards | 10,391 | | | 10,095 | |
| Other | 1,710 | | | 1,234 | |
| Total deferred tax assets | 15,159 | | | 14,579 | |
| Valuation allowances | (9,627) | | | (9,391) | |
| Total net deferred tax assets | 5,532 | | | 5,188 | |
| Deferred tax liabilities | | | |
| Excess of book basis over tax basis of intangible assets | (3,590) | | | (4,711) | |
| Excess of book basis over tax basis in investments | (340) | | | (308) | |
| Other | (772) | | | (904) | |
| Total deferred tax liabilities | (4,702) | | | (5,923) | |
| Net deferred tax assets (liabilities) | $ | 830 | | | $ | (735) | |
The increase in net deferred tax assets is primarily related to capitalization of R&D expense and increases in accruals and reserves, offset by a decrease in advance payments. The decrease in deferred tax liabilities is primarily related to amortization of intangible assets.
In connection with the Allergan acquisition, the company recorded adjustments within the measurement period in 2021 related to foreign net operating losses and other credit carryforwards that are not expected to be realized. The adjustments reflected an increase of $8.2 billion to deferred tax assets and an offsetting increase to valuation allowances, resulting in no net impact to deferred tax assets.
The company had valuation allowances of $9.6 billion as of December 31, 2022 and $9.4 billion as of December 31, 2021. These were principally related to foreign and state net operating losses and other credit carryforwards that are not expected to be realized.
As of December 31, 2022, the company had U.S. federal, state and foreign credit carryforwards of $355 million as well as U.S. federal, state and foreign net operating loss carryforwards of $33.2 billion, which will expire at various times through 2042. The remaining U.S. federal and foreign loss carryforwards of $6.0 billion have no expiration.
The Act significantly changed the timing and manner in which earnings of foreign subsidiaries are subject to U.S. tax. Therefore, unremitted foreign earnings subject to the Act’s transition tax are not considered indefinitely reinvested. Post-2017 earnings subject to the U.S. minimum tax on foreign sourced earnings or eligible for the 100 percent foreign dividends received deduction are also not considered indefinitely reinvested earnings. However, the company generally considers instances of outside basis differences in foreign subsidiaries that would incur additional U.S. tax upon reversal (e.g., capital gain distribution) to be permanent in duration. The unrecognized tax liability is not practicable to determine.
| | | | | | | | | | | |
91 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Unrecognized Tax Benefits | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Beginning balance | $ | 5,489 | | | $ | 5,264 | | | $ | 2,661 | |
| Increase due to acquisition | — | | | — | | | 2,674 | |
| Increase due to current year tax positions | 88 | | | 208 | | | 91 | |
| Increase due to prior year tax positions | 243 | | | 137 | | | 59 | |
| Decrease due to prior year tax positions | (33) | | | (62) | | | (7) | |
| Settlements | (7) | | | (24) | | | (141) | |
| Lapse of statutes of limitations | (110) | | | (34) | | | (73) | |
| Ending balance | $ | 5,670 | | | $ | 5,489 | | | $ | 5,264 | |
If recognized, the net amount of potential tax benefits that would impact the company's effective tax rate is $5.5 billion in 2022 and $5.2 billion in 2021. The "Increase due to current year tax positions" and "Increase due to prior year tax positions" in the table above include amounts related to federal, state and international tax items. "Increase due to acquisition" in the table above includes amounts related to federal, state and international tax items recorded in acquisition accounting related to the Allergan acquisition.
AbbVie recognizes interest and penalties related to income tax matters in income tax expense in the consolidated statements of earnings. AbbVie recognized gross income tax expense of $339 million in 2022, $161 million in 2021 and $142 million in 2020, for interest and penalties related to income tax matters. AbbVie had an accrual for the payment of gross interest and penalties of $1.1 billion at December 31, 2022, $803 million at December 31, 2021 and $642 million at December 31, 2020.
The company is routinely audited by the tax authorities in significant jurisdictions and a number of audits are currently underway. It is reasonably possible during the next 12 months that uncertain tax positions may be settled, which could result in a decrease in the gross amount of unrecognized tax benefits. Due to the potential for resolution of federal, state and foreign examinations and the expiration of various statutes of limitation, the company's gross unrecognized tax benefits balance may change within the next 12 months up to $162 million. All significant federal, state, local and international matters have been concluded for years through 2009. The company believes adequate provision has been made for all income tax uncertainties.
Note 15 Legal Proceedings and Contingencies AbbVie is subject to contingencies, such as various claims, legal proceedings and investigations regarding product liability, intellectual property, commercial, securities and other matters that arise in the normal course of business. The most significant matters are described below. Loss contingency provisions are recorded for probable losses at management’s best estimate of a loss, or when a best estimate cannot be made, a minimum loss contingency amount within a probable range is recorded. For litigation matters discussed below for which a loss is probable or reasonably possible, the company is unable to estimate the possible loss or range of loss, if any, beyond the amounts accrued. Initiation of new legal proceedings or a change in the status of existing proceedings may result in a change in the estimated loss accrued by AbbVie. While it is not feasible to predict the outcome of all proceedings and exposures with certainty, management believes that their ultimate disposition should not have a material adverse effect on AbbVie’s consolidated financial position, results of operations or cash flows.
Subject to certain exceptions specified in the separation agreement by and between Abbott Laboratories (Abbott) and AbbVie, AbbVie assumed the liability for, and control of, all pending and threatened legal matters related to its business, including liabilities for any claims or legal proceedings related to products that had been part of its business, but were discontinued prior to the distribution, as well as assumed or retained liabilities, and will indemnify Abbott for any liability arising out of or resulting from such assumed legal matters.
Antitrust Litigation
Lawsuits are pending against AbbVie and others generally alleging that the 2005 patent litigation settlement involving Niaspan entered into between Kos Pharmaceuticals, Inc. (a company acquired by Abbott in 2006 and presently a subsidiary of AbbVie) and a generic company violates federal and state antitrust laws and state unfair and deceptive trade practices and unjust enrichment laws. Plaintiffs generally seek monetary damages and/or injunctive relief and attorneys' fees. The lawsuits pending in federal court consist of four individual plaintiff lawsuits and two consolidated purported class actions: one brought
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 92 |
by Niaspan direct purchasers and one brought by Niaspan end-payors. The cases are pending in the United States District Court for the Eastern District of Pennsylvania for coordinated or consolidated pre-trial proceedings under the MDL Rules as In re: Niaspan Antitrust Litigation, MDL No. 2460. In August 2019, the court certified a class of direct purchasers of Niaspan. In June 2020 and August 2021, the court denied the end-payors' motion to certify a class. In October 2016, the Orange County, California District Attorney’s Office filed a lawsuit on behalf of the State of California regarding the Niaspan patent litigation settlement in Orange County Superior Court, asserting a claim under the unfair competition provision of the California Business and Professions Code seeking injunctive relief, restitution, civil penalties and attorneys’ fees.
In August 2019, direct purchasers of AndroGel filed a lawsuit, King Drug Co. of Florence, Inc., et al. v. AbbVie Inc., et al., against AbbVie and others in the United States District Court for the Eastern District of Pennsylvania, alleging that 2006 patent litigation settlements and related agreements by Solvay Pharmaceuticals, Inc. (a company Abbott acquired in February 2010 and now known as AbbVie Products LLC) with three generic companies violated federal antitrust law, and also alleging that 2011 patent litigation by Abbott with two generic companies regarding AndroGel was sham litigation and the settlements of those litigations violated federal antitrust law. Plaintiffs generally seek monetary damages and/or injunctive relief and attorneys’ fees. In November 2022, the State of Oregon filed a lawsuit in the Multnomah County, Oregon Circuit Court making similar allegations regarding the 2011 patent litigation with one of the generic companies.
Lawsuits are pending against Forest Laboratories, LLC, an AbbVie subsidiary, and others generally alleging that 2009 and 2010 patent litigation settlements involving Namenda entered into between Forest and generic companies and other conduct by Forest involving Namenda, violated state antitrust, unfair and deceptive trade practices and unjust enrichment laws. Plaintiffs generally seek monetary damages and/or injunctive relief and attorneys’ fees. The lawsuits, purported class actions filed by indirect purchasers of Namenda, are consolidated as In re: Namenda Indirect Purchaser Antitrust Litigation in the United States District Court for the Southern District of New York. In November 2022, the parties reached an agreement to settle this matter that has received preliminary court approval.
Lawsuits are pending against Forest Laboratories, LLC and others generally alleging that 2012 and 2013 patent litigation settlements involving Bystolic with six generic manufacturers violated federal and state antitrust laws and state unfair and deceptive trade practices and unjust enrichment laws. Plaintiffs generally seek monetary damages and/or injunctive relief and attorneys’ fees. The lawsuits, purported class actions filed on behalf of direct and indirect purchasers of Bystolic, are consolidated as In re: Bystolic Antitrust Litigation in the United States District Court for the Southern District of New York.
Government Proceedings
Lawsuits are pending against Allergan and several other manufacturers generally alleging that they improperly promoted and sold prescription opioid products. Approximately 3,000 matters are pending against Allergan. Most of the federal court cases are consolidated for pre-trial purposes in the United States District Court for the Northern District of Ohio under the MDL rules as In re: National Prescription Opiate Litigation, MDL No. 2804. Approximately 270 matters are pending in various state courts. The plaintiffs in these cases, which include states, counties, cities, other municipal entities, Native American tribes, union trust funds and other third-party payors, private hospitals and personal injury claimants, generally seek compensatory and punitive damages. In November 2022, Allergan finalized the terms of a settlement with state and local government entities and Native American tribes. That settlement is subject to certain conditions, including Allergan's determination that a sufficient number of government entities elect to participate in the settlement. AbbVie recorded a charge of $2.1 billion to selling, general and administrative expense in the consolidated statement of earnings in the second quarter of 2022 related to this potential settlement.
Shareholder and Securities Litigation
In June 2016, a lawsuit, Elliott Associates, L.P., et al. v. AbbVie Inc., was filed by five investment funds against AbbVie in the Cook County, Illinois Circuit Court alleging that AbbVie made misrepresentations and omissions in connection with its proposed transaction with Shire. Similar lawsuits were filed between July 2017 and October 2019 against AbbVie and in some instances its chief executive officer in the same court by additional investment funds. In September 2021, the Illinois court granted AbbVie's motion for summary judgment on all pending claims in all pending cases, dismissing them with prejudice. In November 2022, the Illinois appellate court affirmed summary judgment in AbbVie's favor and, in December 2022, that court denied plaintiffs' petition for rehearing.
In October 2018, a federal securities lawsuit, Holwill v. AbbVie Inc., et al., was filed in the United States District Court for the Northern District of Illinois against AbbVie, its chief executive officer and former chief financial officer, alleging that reasons stated for Humira sales growth in financial filings between 2013 and 2018 were misleading because they omitted alleged misconduct in connection with Humira patient and reimbursement support services and other services and items of value that allegedly induced Humira prescriptions. In September 2021, the court granted plaintiffs' motion to certify a class. In May 2022, a shareholder derivative lawsuit, Ranney v. Gonzalez, et al., was filed in Delaware Chancery Court, alleging that
| | | | | | | | | | | |
93 |  | 2022 Form 10-K | 2022 Form 10-K | | |
certain AbbVie directors and officers breached their fiduciary duties based on related allegations. In December 2022, after AbbVie and the director/officer defendants filed a motion to dismiss, the plaintiff voluntarily dismissed the lawsuit with prejudice.
Lawsuits are pending against Allergan and certain of its current and former officers alleging they made misrepresentations and omissions regarding Allergan's textured breast implants. The lawsuits, which were filed by Allergan shareholders, have been consolidated in the United States District Court for the Southern District of New York as In re: Allergan plc Securities Litigation. The plaintiffs generally seek compensatory damages and attorneys’ fees. In September 2019, the court partially granted Allergan's motion to dismiss. In September 2021, the court granted plaintiffs' motion to certify a class. In December 2022, the court granted Allergan's motion for summary judgment on the remaining claims, dismissing them with prejudice. Plaintiffs are appealing the court's motion to dismiss and summary judgment rulings.
In April 2022, a federal securities lawsuit, Nakata v. AbbVie Inc., was filed in the United States District Court for the Northern District of Illinois against AbbVie and certain officers alleging misstatements regarding the potential effect that safety information about another company’s product would have on the Food and Drug Administration’s approval and labeling for AbbVie’s Rinvoq. In May and July 2022, two shareholder derivative lawsuits, Treppel Family Trust v. Gonzalez et al., and Katcher v. Gonzalez, et al., were filed in the same court, alleging that certain AbbVie directors and officers breached fiduciary and other legal duties based on related allegations.
Product Liability and General Litigation
In 2018, a qui tam lawsuit, U.S. ex rel. Silbersher v. Allergan Inc., et al., was filed in the United States District Court for the Northern District of California against several Allergan entities and others, alleging that their conduct before the U.S. Patent Office resulted in false claims for payment being made to federal and state healthcare payors for Namenda XR and Namzaric. The plaintiff-relator seeks damages and attorneys' fees under the federal False Claims Act and state law analogues. The federal government and state governments declined to intervene in the lawsuit. In August 2022, the United States Court of Appeals reversed the district court’s denial of Allergan’s motion to dismiss. The case has been remanded to the district court for further proceedings consistent with that ruling.
Intellectual Property Litigation
Pharmacyclics LLC, a wholly owned subsidiary of AbbVie, is seeking to enforce its patent rights relating to ibrutinib tablets (a drug Pharmacyclics sells under the trademark Imbruvica). Cases were filed in the United States District Court for the District of Delaware in March 2019 against Alvogen Pine Brook LLC and Natco Pharma Ltd. In August 2021, the court issued a decision holding all asserted patents infringed and valid. The judgment precludes Defendants from obtaining regulatory approval and launching until the last patent expires in 2036. On August 30, 2021, Defendants appealed. On November 15, 2022, the Court of Appeals for the Federal Circuit affirmed the judgment. Janssen Biotech, Inc. which is in a global collaboration with Pharmacyclics concerning the development and marketing of Imbruvica, is the co-plaintiff in these suits.
AbbVie Inc. is seeking to enforce patent rights relating to venetoclax (a drug sold under the trademark Venclexta). Litigation was filed in the United States District Court for the District of Delaware in July 2020 against Dr. Reddy’s Laboratories, Ltd. and Dr. Reddy’s Laboratories, Inc.: and Alembic Pharmaceuticals Ltd., Alembic Pharmaceuticals, Inc., and Alembic Global Holdings SA. AbbVie alleges defendants’ proposed generic venetoclax products infringe certain patents and seeks declaratory and injunctive relief. Genentech, Inc., which is in a global collaboration with AbbVie concerning the development and marketing of Venclexta, is the co-plaintiff in this suit.
Note 16 Segment and Geographic Area Information AbbVie operates as a single global business segment dedicated to the research and development, manufacturing, commercialization and sale of innovative medicines and therapies. This operating structure enables the Chief Executive Officer, as chief operating decision maker (CODM), to allocate resources and assess business performance on a global basis in order to achieve established long-term strategic goals. Consistent with this structure, a global research and development and supply chain organization is responsible for the discovery, manufacturing and supply of products. Commercial efforts that coordinate the marketing, sales and distribution of these products are organized by geographic region or therapeutic area. All of these activities are supported by a global corporate administrative staff. The determination of a single business segment is consistent with the consolidated financial information regularly reviewed by the CODM for purposes of assessing performance, allocating resources and planning and forecasting future periods.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 94 |
Substantially all of AbbVie's net revenues in the United States are to three wholesalers. Outside the United States, products are sold primarily to health care providers or through distributors, depending on the market served. The following tables detail AbbVie's worldwide net revenues:
| | | | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Immunology | | | | | |
| Humira | United States | $ | 18,619 | | | $ | 17,330 | | | $ | 16,112 | |
| International | 2,618 | | | 3,364 | | | 3,720 | |
| Total | $ | 21,237 | | | $ | 20,694 | | | $ | 19,832 | |
| Skyrizi | United States | $ | 4,484 | | | $ | 2,486 | | | $ | 1,385 | |
| International | 681 | | | 453 | | | 205 | |
| Total | $ | 5,165 | | | $ | 2,939 | | | $ | 1,590 | |
| Rinvoq | United States | $ | 1,794 | | | $ | 1,271 | | | $ | 653 | |
| International | 728 | | | 380 | | | 78 | |
| Total | $ | 2,522 | | | $ | 1,651 | | | $ | 731 | |
| Hematologic Oncology | | | | | |
| Imbruvica | United States | $ | 3,426 | | | $ | 4,321 | | | $ | 4,305 | |
| Collaboration revenues | 1,142 | | | 1,087 | | | 1,009 | |
| Total | $ | 4,568 | | | $ | 5,408 | | | $ | 5,314 | |
| Venclexta | United States | $ | 1,009 | | | $ | 934 | | | $ | 804 | |
| International | 1,000 | | | 886 | | | 533 | |
| Total | $ | 2,009 | | | $ | 1,820 | | | $ | 1,337 | |
| Aesthetics | | | | | |
Botox Cosmetic (a) | United States | $ | 1,654 | | | $ | 1,424 | | | $ | 687 | |
| International | 961 | | | 808 | | | 425 | |
| Total | $ | 2,615 | | | $ | 2,232 | | | $ | 1,112 | |
Juvederm Collection (a) | United States | $ | 548 | | | $ | 658 | | | $ | 318 | |
| International | 880 | | | 877 | | | 400 | |
| Total | $ | 1,428 | | | $ | 1,535 | | | $ | 718 | |
Other Aesthetics (a) | United States | $ | 1,122 | | | $ | 1,268 | | | $ | 666 | |
| International | 168 | | | 198 | | | 94 | |
| Total | $ | 1,290 | | | $ | 1,466 | | | $ | 760 | |
| Neuroscience | | | | | |
Botox Therapeutic (a) | United States | $ | 2,255 | | | $ | 2,012 | | | $ | 1,155 | |
| International | 464 | | | 439 | | | 232 | |
| Total | $ | 2,719 | | | $ | 2,451 | | | $ | 1,387 | |
Vraylar (a) | United States | $ | 2,037 | | | $ | 1,728 | | | $ | 951 | |
| International | 1 | | | — | | | — | |
| Total | $ | 2,038 | | | $ | 1,728 | | | $ | 951 | |
| Duodopa | United States | $ | 95 | | | $ | 102 | | | $ | 103 | |
| International | 363 | | | 409 | | | 391 | |
| Total | $ | 458 | | | $ | 511 | | | $ | 494 | |
Ubrelvy (a) | United States | $ | 680 | | | $ | 552 | | | $ | 125 | |
| Qulipta | United States | $ | 158 | | | $ | — | | | $ | — | |
| | | | | | |
| | | | | | |
Other Neuroscience (a) | United States | $ | 456 | | | $ | 667 | | | $ | 528 | |
| International | 19 | | | 18 | | | 11 | |
| Total | $ | 475 | | | $ | 685 | | | $ | 539 | |
| | | | | | | | | | | |
95 |  | 2022 Form 10-K | 2022 Form 10-K | | |
| | | | | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| Eye Care | | | | | |
Lumigan/Ganfort (a) | United States | $ | 242 | | | $ | 273 | | | $ | 165 | |
| International | 272 | | | 306 | | | 213 | |
| Total | $ | 514 | | | $ | 579 | | | $ | 378 | |
Alphagan/Combigan (a) | United States | $ | 202 | | | $ | 373 | | | $ | 223 | |
| International | 144 | | | 156 | | | 103 | |
| Total | $ | 346 | | | $ | 529 | | | $ | 326 | |
Restasis (a) | United States | $ | 621 | | | $ | 1,234 | | | $ | 755 | |
| International | 45 | | | 56 | | | 32 | |
| Total | $ | 666 | | | $ | 1,290 | | | $ | 787 | |
Other Eye Care (a) | United States | $ | 538 | | | $ | 523 | | | $ | 305 | |
| International | 637 | | | 646 | | | 388 | |
| Total | $ | 1,175 | | | $ | 1,169 | | | $ | 693 | |
| Other Key Products | | | | | |
| Mavyret | United States | $ | 755 | | | $ | 754 | | | $ | 785 | |
| International | 786 | | | 956 | | | 1,045 | |
| Total | $ | 1,541 | | | $ | 1,710 | | | $ | 1,830 | |
| Creon | United States | $ | 1,278 | | | $ | 1,191 | | | $ | 1,114 | |
Linzess/Constella (a) | United States | $ | 1,003 | | | $ | 1,006 | | | $ | 649 | |
| International | 32 | | | 32 | | | 18 | |
| Total | $ | 1,035 | | | $ | 1,038 | | | $ | 667 | |
| All other | | $ | 4,137 | | | $ | 5,019 | | | $ | 5,119 | |
| Total net revenues | $ | 58,054 | | | $ | 56,197 | | | $ | 45,804 | |
(a)Net revenues include Allergan product revenues after the acquisition closing date of May 8, 2020.
Net revenues to external customers by geographic area, based on product shipment destination, were as follows:
| | | | | | | | | | | | | | | | | |
| years ended December 31 (in millions) | 2022 | | 2021 | | 2020 |
| United States | $ | 45,713 | | | $ | 43,510 | | | $ | 34,879 | |
| Germany | 1,340 | | | 1,223 | | | 1,049 | |
| Canada | 1,159 | | | 1,397 | | | 1,159 | |
| Japan | 956 | | | 1,090 | | | 1,198 | |
| China | 912 | | | 857 | | | 471 | |
| France | 787 | | | 936 | | | 797 | |
| Australia | 508 | | | 533 | | | 527 | |
| Spain | 506 | | | 519 | | | 453 | |
| United Kingdom | 462 | | | 497 | | | 509 | |
| Italy | 444 | | | 506 | | | 379 | |
| Brazil | 430 | | | 368 | | | 406 | |
| All other countries | 4,837 | | | 4,761 | | | 3,977 | |
| Total net revenues | $ | 58,054 | | | $ | 56,197 | | | $ | 45,804 | |
Long-lived assets, primarily net property and equipment, by geographic area were as follows:
| | | | | | | | | | | |
| as of December 31 (in millions) | 2022 | | 2021 |
| United States and Puerto Rico | $ | 3,243 | | | $ | 3,369 | |
| Europe | 1,369 | | | 1,400 | |
| All other | 323 | | | 341 | |
| Total long-lived assets | $ | 4,935 | | | $ | 5,110 | |
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 96 |
Note 17 Fourth Quarter Financial Results (unaudited) | | | | | | | |
| quarter ended December 31 (in millions except per share data) | 2022 | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| Net revenues | $ | 15,121 | | | |
| Gross margin | 10,951 | | | |
Net earnings attributable to AbbVie Inc. | 2,473 | | | |
| Basic earnings per share attributable to AbbVie Inc. | $ | 1.39 | | | |
| Diluted earnings per share attributable to AbbVie Inc. | $ | 1.38 | | | |
| Cash dividends declared per common share | $ | 1.48 | | | |
| | | | | | | | | | | |
97 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of AbbVie Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of AbbVie Inc. and subsidiaries (the Company) as of December 31, 2022 and 2021, the related consolidated statements of earnings, comprehensive income, equity and cash flows for each of the three years in the period ended December 31, 2022, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2022 and 2021, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2022, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company's internal control over financial reporting as of December 31, 2022, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) and our report dated February 17, 2023 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 98 |
| | | | | | | | |
| | Sales rebate accruals for Medicaid, Medicare and managed care programs |
| Description of the Matter | | As discussed in Note 2 to the consolidated financial statements under the caption “Revenue Recognition,” the Company established provisions for sales rebates in the same period the related product is sold. At December 31, 2022, the Company had $10,717 million in sales rebate accruals, a large portion of which were for rebates provided to pharmacy benefit managers, state government Medicaid programs, insurance companies that administer Medicare drug plans and private entities for Medicaid, Medicare and managed care programs. In order to establish these sales rebate accruals, the Company estimated its rebates based upon the identification of the products subject to a rebate, the applicable price and rebate terms and the estimated lag time between the sale and payment of the rebate. Auditing the Medicaid, Medicare and managed care sales rebate accruals was complex and required significant auditor judgment because the accruals consider multiple subjective and complex estimates and assumptions. These estimates and assumptions included the estimated inventory in the distribution channel, which impacts the lag time between the sale to the customer and payment of the rebate, and the final payer related to product sales, which impacts the applicable price and rebate terms. In deriving these estimates and assumptions, the Company used both internal and external sources of information to estimate product in the distribution channels, payer mix, prescription volumes and historical experience. Management supplemented its historical data analysis with qualitative adjustments based upon changes in rebate trends, rebate programs and contract terms, legislative changes, or other significant events which indicate a change in the reserve is appropriate. |
| How We Addressed the Matter in Our Audit | | We obtained an understanding, evaluated the design and tested the operating effectiveness of controls over the Company’s sales rebate accruals for Medicaid, Medicare and managed care programs. This included testing controls over management’s review of the significant assumptions and other inputs used in the estimation of Medicaid, Medicare and managed care rebates, among others, including the significant assumptions discussed above. The testing was inclusive of management’s controls to evaluate the accuracy of its reserve judgments to actual rebates paid, rebate validation and processing, and controls to ensure that the data used to evaluate and support the significant assumptions was complete, accurate and, where applicable, verified to external data sources. To test the sales rebate accruals for Medicaid, Medicare and managed care programs, our audit procedures included, among others, understanding and evaluating the significant assumptions and underlying data used in management’s calculations. Our testing of significant assumptions included corroboration to external data sources. We evaluated the reasonableness of assumptions considering industry and economic trends, product profiles, and other regulatory factors. We assessed the historical accuracy of management’s estimates by comparing actual activity to previous estimates and performed analytical procedures, based on internal and external data sources, to evaluate the completeness of the reserves. For Medicaid, we involved a specialist with an understanding of statutory reimbursement requirements to assess the consistency of the Company’s calculation methodologies with applicable government regulations and policy. |
| | |
| | | | | | | | | | | |
99 |  | 2022 Form 10-K | 2022 Form 10-K | | |
| | | | | | | | |
| | Valuation of contingent consideration |
| Description of the Matter | | As discussed in Note 2 to the consolidated financial statements under the caption “Business Combinations” and in Note 11 under the caption “Fair Value Measures,” the Company recognized contingent consideration liabilities at the estimated fair value on the acquisition date in connection with applying the acquisition method of accounting for business combinations. Subsequent changes to the fair value of the contingent consideration liabilities were recorded within the consolidated statement of earnings in the period of change. At December 31, 2022, the Company had $16,384 million in contingent consideration liabilities, which represented a ‘Level 3’ fair value measurement in the fair value hierarchy due to the significant unobservable inputs used in determining the fair value and the use of management judgment about the assumptions market participants would use in pricing the liabilities. Auditing the valuation of contingent consideration liabilities was complex and required significant auditor judgment due to the use of a Monte Carlo simulation model and the high degree of subjectivity in evaluating certain assumptions required to estimate the fair value of contingent royalty payments. In particular, the fair value measurement was sensitive to the significant assumptions underlying the estimated amount of future sales of the acquired products. Management utilized its expertise within the industry, including commercial dynamics, trends and utilization, as well as knowledge of clinical development and regulatory approval processes to determine certain of these assumptions. |
| How We Addressed the Matter in Our Audit | | We obtained an understanding, evaluated the design and tested the operating effectiveness of controls over the Company’s contingent consideration liabilities process including, among others, management’s process to establish the significant assumptions and measure the liability. This included testing controls over management’s review of the significant assumptions and other inputs used in the determination of fair value. The testing was inclusive of key management review controls to monitor and evaluate clinical development of the acquired products and estimated future sales, and controls to ensure that the data used to evaluate and support the significant assumptions was complete, accurate and, where applicable, verified to external data sources. To test the estimated fair value of contingent consideration liabilities, our audit procedures included, among others, inspecting the terms of the executed agreement, assessing the Monte Carlo simulation model used and testing the key contractual inputs and significant assumptions discussed above. We evaluated the assumptions and judgments considering observable industry and economic trends and standards, external data sources and regulatory factors. Estimated amounts of future sales were evaluated for reasonableness in relation to internal and external analyses, clinical development progress and timelines, probability of success benchmarks, and regulatory notices. Our procedures included evaluating the data sources used by management in determining its assumptions and, where necessary, included an evaluation of available information that either corroborated or contradicted management’s conclusions. We involved a valuation specialist to assess the Company’s Monte Carlo simulation model and to perform corroborative fair value calculations. |
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 2013.
Chicago, Illinois
February 17, 2023
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 100 |
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE None.
ITEM 9A. CONTROLS AND PROCEDURES Disclosure Controls and Procedures; Internal Control Over Financial Reporting
Evaluation of disclosure controls and procedures. The Chief Executive Officer, Richard A. Gonzalez, and the Chief Financial Officer, Scott T. Reents, evaluated the effectiveness of AbbVie's disclosure controls and procedures as of the end of the period covered by this report, and concluded that AbbVie's disclosure controls and procedures were effective to ensure that information AbbVie is required to disclose in the reports that it files or submits with the Securities and Exchange Commission under the Securities Exchange Act of 1934 is recorded, processed, summarized and reported, within the time periods specified in the Commission's rules and forms, and to ensure that information required to be disclosed by AbbVie in the reports that it files or submits under the Securities Exchange Act of 1934 is accumulated and communicated to AbbVie's management, including its principal executive officer and principal financial officer, as appropriate to allow timely decisions regarding required disclosure.
Changes in internal control over financial reporting. There were no changes in AbbVie's internal control over financial reporting (as defined in Rule 13a-15(f) under the Securities Exchange Act of 1934) that have materially affected, or are reasonably likely to materially affect, AbbVie's internal control over financial reporting during the quarter ended December 31, 2022.
Inherent limitations on effectiveness of controls. AbbVie's management, including its Chief Executive Officer and its Chief Financial Officer, do not expect that AbbVie's disclosure controls or internal control over financial reporting will prevent or detect all errors and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system's objectives will be met. The design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Further, because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that misstatements due to error or fraud will not occur or that all control issues and instances of fraud, if any, have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty and that breakdowns can occur because of simple error or mistake. Controls can also be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the controls.
The design of any system of controls is based in part on certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. Projections of any evaluation of controls effectiveness to future periods are subject to risks. Over time, controls may become inadequate because of changes in conditions or deterioration in the degree of compliance with policies or procedures.
Management's annual report on internal control over financial reporting. Management of AbbVie is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Rule 13a-15(f) under the Securities Exchange Act of 1934. AbbVie's internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles in the United States. However, all internal control systems, no matter how well designed, have inherent limitations. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and reporting.
Management assessed the effectiveness of AbbVie's internal control over financial reporting as of December 31, 2022. In making this assessment, management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework (2013 framework). Based on that assessment, management concluded that AbbVie maintained effective internal control over financial reporting as of December 31, 2022, based on the COSO criteria.
The effectiveness of AbbVie's internal control over financial reporting as of December 31, 2022 has been audited by Ernst & Young LLP, an independent registered public accounting firm, as stated in their attestation report below, which expresses an unqualified opinion on the effectiveness of AbbVie's internal control over financial reporting as of December 31, 2022.
Report of independent registered public accounting firm. The report of AbbVie's independent registered public accounting firm related to its assessment of the effectiveness of internal control over financial reporting is included below.
| | | | | | | | | | | |
101 |  | 2022 Form 10-K | 2022 Form 10-K | | |
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of AbbVie Inc.
Opinion on Internal Control over Financial Reporting
We have audited AbbVie Inc. and subsidiaries' internal control over financial reporting as of December 31, 2022, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). In our opinion, AbbVie Inc. and subsidiaries (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2022, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of AbbVie Inc. and subsidiaries as of December 31, 2022 and 2021, the related consolidated statements of earnings, comprehensive income, equity and cash flows for each of the three years in the period ended December 31, 2022, and the related notes and our report dated February 17, 2023 expressed an unqualified opinion thereon.
Basis for Opinion
The Company's management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management's Annual Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company's internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations on Internal Control Over Financial Reporting
A company's internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company's internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company's assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
Chicago, Illinois
February 17, 2023
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 102 |
ITEM 9B. OTHER INFORMATION None.
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS Not Applicable.
| | | | | | | | | | | |
103 |  | 2022 Form 10-K | 2022 Form 10-K | | |
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE Incorporated herein by reference are "Information Concerning Director Nominees," "The Board of Directors and its Committees—Committees of the Board of Directors," "Communicating with the Board of Directors," and "Deadlines for Notice of Stockholder Actions to be Considered at the 2024 Annual Meeting of Stockholders" to be included in the 2023 AbbVie Inc. Proxy Statement. The 2023 Definitive Proxy Statement will be filed on or about March 20, 2023. Also incorporated herein by reference is the text found in this Form 10-K under the caption, "Information about Our Executive Officers."
AbbVie's code of business conduct requires all its business activities to be conducted in compliance with all applicable laws, regulations and ethical principles and values. All directors, officers and employees of AbbVie are required to read, understand and abide by the requirements of the code of business conduct applicable to them. AbbVie's code of business conduct is available in the corporate governance section of AbbVie's investor relations website at www.abbvieinvestor.com.
Any waiver of the code of business conduct for directors or executive officers may be made only by AbbVie's audit committee. AbbVie will disclose any amendment to, or waiver from, a provision of the code of conduct for the principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions, on its website within four business days following the date of the amendment or waiver. In addition, AbbVie will disclose any waiver from the code of business conduct for the other executive officers and for directors on the website.
AbbVie has a chief ethics and compliance officer who reports to the Executive Vice President, General Counsel and Secretary and to the public policy committee. The chief ethics and compliance officer is responsible for overseeing, administering and monitoring AbbVie's compliance program.
ITEM 11. EXECUTIVE COMPENSATION The material to be included in the 2023 AbbVie Inc. Proxy Statement under the headings "Director Compensation," "Executive Compensation," and "Compensation Committee Report" is incorporated herein by reference. The 2023 Definitive Proxy Statement will be filed on or about March 20, 2023.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 104 |
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS (a) Equity Compensation Plan Information.
The following table presents information as of December 31, 2022 about AbbVie's equity compensation plans under which AbbVie common stock has been authorized for issuance: | | | | | | | | | | | | | | | | | |
| Plan Category | (a)
Number of
securities to be
issued upon
exercise of
outstanding
options,
warrants and
rights (1) | | (b)
Weighted-
average exercise price of
outstanding options,
warrants and
rights (2) | | (c)
Number of securities remaining available for
future issuance under equity compensation plans (excluding securities
reflected in
column (a)) (3) |
| Equity compensation plans approved by security holders | 22,351,655 | | | $ | 91.84 | | | 67,365,289 | |
| Equity compensation plans not approved by security holders | — | | | — | | | — | |
| Total | 22,351,655 | | | $ | 91.84 | | | 67,365,289 | |
(1)Includes 34,879 shares issuable under AbbVie's Incentive Stock Program pursuant to awards granted by Abbott and adjusted into AbbVie awards in connection with AbbVie's separation from Abbott.
(2)The weighted-average exercise price does not include outstanding restricted stock units, restricted stock awards and performance shares that have no exercise price.
(3)Excludes shares issuable upon the exercise of stock options and pursuant to other rights granted under the Stemcentrx 2011 Equity Incentive Plan, which was assumed by AbbVie upon the consummation of its acquisition of Stemcentrx, Inc. As of December 31, 2022, 41,212 options remained outstanding under this plan. The options have a weighted-average exercise price of $18.02. No further awards will be granted under this plan.
(b)Information Concerning Security Ownership. Incorporated herein by reference is the material under the heading "Securities Ownership—Securities Ownership of Executive Officers and Directors" in the 2023 AbbVie Inc. Proxy Statement. The 2023 Definitive Proxy Statement will be filed on or about March 20, 2023.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE The material to be included in the 2023 AbbVie Inc. Proxy Statement under the headings "The Board of Directors and its Committees," "Corporate Governance Materials," and "Procedures for Approval of Related Person Transactions" is incorporated herein by reference. The 2023 Definitive Proxy Statement will be filed on or about March 20, 2023.
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES The material to be included in the 2023 AbbVie Inc. Proxy Statement under the headings "Audit Fees and Non-Audit Fees" and "Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of the Independent Registered Public Accounting Firm" is incorporated herein by reference. The 2023 Definitive Proxy Statement will be filed on or about March 20, 2023.
| | | | | | | | | | | |
105 |  | 2022 Form 10-K | 2022 Form 10-K | | |
PART IV
ITEM 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES
(a)Documents filed as part of this Form 10-K.
(1)Financial Statements: See Item 8, "Financial Statements and Supplementary Data" for a list of financial statements.
(2)Financial Statement Schedules: All schedules omitted are inapplicable or the information required is shown in the consolidated financial statements or notes thereto.
(3)Exhibits Required by Item 601 of Regulation S-K: The information called for by this paragraph is set forth in Item 15(b) below.
(b) Exhibits:
| | | | | | | | | | | |
Exhibit Number | | Exhibit Description | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | *Supplemental Indenture No. 4, dated as of November 17, 2016, among AbbVie Inc., U.S. Bank National Association, as trustee, Elavon Financial Services DAC, U.K. Branch, as paying agent and Elavon Financial Services DAC, as transfer agent and registrar, including forms of notes (incorporated by reference to Exhibit 4.1 of the company's Current Report on Form 8-K filed on November 17, 2016). | |
| | | |
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 106 |
| | | | | | | | | | | |
Exhibit Number | | Exhibit Description | |
| | *Supplemental Indenture No. 6, dated September 26, 2019, among AbbVie Inc., U.S. Bank National Association, as trustee, transfer agent and registrar, and Elavon Financial Services DAC, UK Branch, as paying agent, including forms of notes (incorporated by reference to Exhibit 4.2 of the company’s Current Report on Form 8-K filed on September 26, 2019). | |
| | | |
| | | |
| | *Supplemental Indenture No. 9, dated May 14, 2020, among AbbVie Inc., U.S. Bank and National Association, as trustee, transfer agent and registrar, and Elavon Financial Services DAC, U.K. Branch, as paying agent (incorporated by reference to Exhibit 4.15 of the company's Current Report on Form 8-K filed on May 14, 2020). | |
| | *Agency Agreement, dated as of November 17, 2016, among AbbVie Inc., U.S. Bank National Association, as trustee, Elavon Financial Services DAC, U.K. Branch, as paying agent and Elavon Financial Services DAC, as transfer agent and registrar (incorporated by reference to Exhibit 4.2 of the company's Current Report on Form 8-K filed on November 17, 2016). | |
| | *Agency Agreement, dated September 26, 2019, among AbbVie Inc., U.S. Bank National Association, as trustee, transfer agent and registrar, and Elavon Financial Services DAC, U.K. Branch, as paying agent (incorporated by reference to Exhibit 4.3 of the company’s Current Report on Form 8-K filed on September 26, 2019). | |
| | | |
| | *Agency Agreement, dated May 14, 2020, among AbbVie Inc., U.S. Bank National Association, as trustee, transfer agent and registrar, and Elavon Financial Services DAC, U.K. Branch, as paying agent and calculation agent (incorporated by reference to Exhibit 4.16 of the company’s Current Report on Form 8-K filed on May 14, 2020). | |
| | *Registration Rights Agreement, dated May 14, 2020, among AbbVie Inc. and Morgan Stanley & Co. LLC, BofA Securities, Inc., Citigroup Global Markets Inc., BNP Paribas Securities Corp., HSBC Securities (USA) Inc., Mizuho Securities USA LLC and Wells Fargo Securities, LLC (incorporated by reference to Exhibit 4.23 of the company’s Current Report on Form 8-K filed on May 14, 2020). | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | | | | | | | | | |
107 |  | 2022 Form 10-K | 2022 Form 10-K | | |
| | | | | | | | | | | |
Exhibit Number | | Exhibit Description | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
|
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 108 |
| | | | | | | | | | | |
Exhibit Number | | Exhibit Description | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| 101 | | The following financial statements and notes from the AbbVie Inc. Annual Report on Form 10-K for the year ended December 31, 2022 filed on February 17, 2023, formatted in Inline XBRL (eXtensible Business Reporting Language): (i) Consolidated Statements of Earnings; (ii) Consolidated Statements of Comprehensive Income; (iii) Consolidated Balance Sheets; (iv) Consolidated Statements of Equity; (v) Consolidated Statements of Cash Flows; and (vi) the Notes to Consolidated Financial Statements. | |
| 104 | | Cover Page Interactive Data File (the cover page from the AbbVie Inc. Annual Report on Form 10-K formatted as Inline XBRL and contained in Exhibit 101). | |
| | | The AbbVie Inc. 2023 Definitive Proxy Statement will be filed with the Securities and Exchange Commission under separate cover on or about March 20, 2023. | |
_______________________________________________________________________________
* Incorporated herein by reference. Commission file number 001-35565.
** Denotes management contract or compensatory plan or arrangement required to be filed as an exhibit hereto.
Exhibits 32.1 and 32.2, above, are furnished herewith and should not be deemed to be "filed" under the Securities Exchange Act of 1934. AbbVie will furnish copies of any of the above exhibits to a stockholder upon written request to the Secretary, AbbVie Inc., 1 North Waukegan Road, North Chicago, Illinois 60064.
| | | | | | | | | | | |
109 |  | 2022 Form 10-K | 2022 Form 10-K | | |
ITEM 16. FORM 10-K SUMMARY None.
| | | | | | | | | | | |
| | 2022 Form 10-K |  | 110 |
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, AbbVie Inc. has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | | | | | | | |
| AbbVie Inc. |
| By: | | /s/ RICHARD A. GONZALEZ |
| | | Name: | | Richard A. Gonzalez |
| | | Title: | | Chairman of the Board and
Chief Executive Officer |
| Date: | | February 17, 2023 |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of AbbVie Inc. on February 17, 2023 in the capacities indicated below.
| | | | | | | | |
| /s/ RICHARD A. GONZALEZ | | /s/ SCOTT T. REENTS |
Richard A. Gonzalez
Chairman of the Board and
Chief Executive Officer
(Principal Executive Officer) | | Scott T. Reents
Executive Vice President, Chief Financial Officer
(Principal Financial Officer)
|
| | |
| /s/ BRIAN L. DURKIN | | |
Brian L. Durkin
Vice President, Controller
(Principal Accounting Officer) | | |
| | |
| /s/ ROBERT J. ALPERN, M.D. | | /s/ ROXANNE S. AUSTIN |
Robert J. Alpern, M.D.
Director of AbbVie Inc. | | Roxanne S. Austin
Director of AbbVie Inc. |
| | |
| /s/ WILLIAM H.L. BURNSIDE | | /s/ THOMAS C. FREYMAN |
William H.L. Burnside
Director of AbbVie Inc. | | Thomas C. Freyman
Director of AbbVie Inc. |
| | |
| /s/ BRETT J. HART | | |
Brett J. Hart
Director of AbbVie Inc. | | |
| | |
| /s/ MELODY B. MEYER | | /s/ EDWARD J. RAPP |
Melody B. Meyer
Director of AbbVie Inc. | | Edward J. Rapp
Director of AbbVie Inc. |
| | |
| /s/ REBECCA B. ROBERTS | | /s/ GLENN F. TILTON |
Rebecca B. Roberts
Director of AbbVie Inc. | | Glenn F. Tilton
Director of AbbVie Inc. |
| | |
| /s/ FREDERICK H. WADDELL | | |
Frederick H. Waddell
Director of AbbVie Inc. | | |
| | | | | | | | | | | |
111 |  | 2022 Form 10-K | 2022 Form 10-K | | |


 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
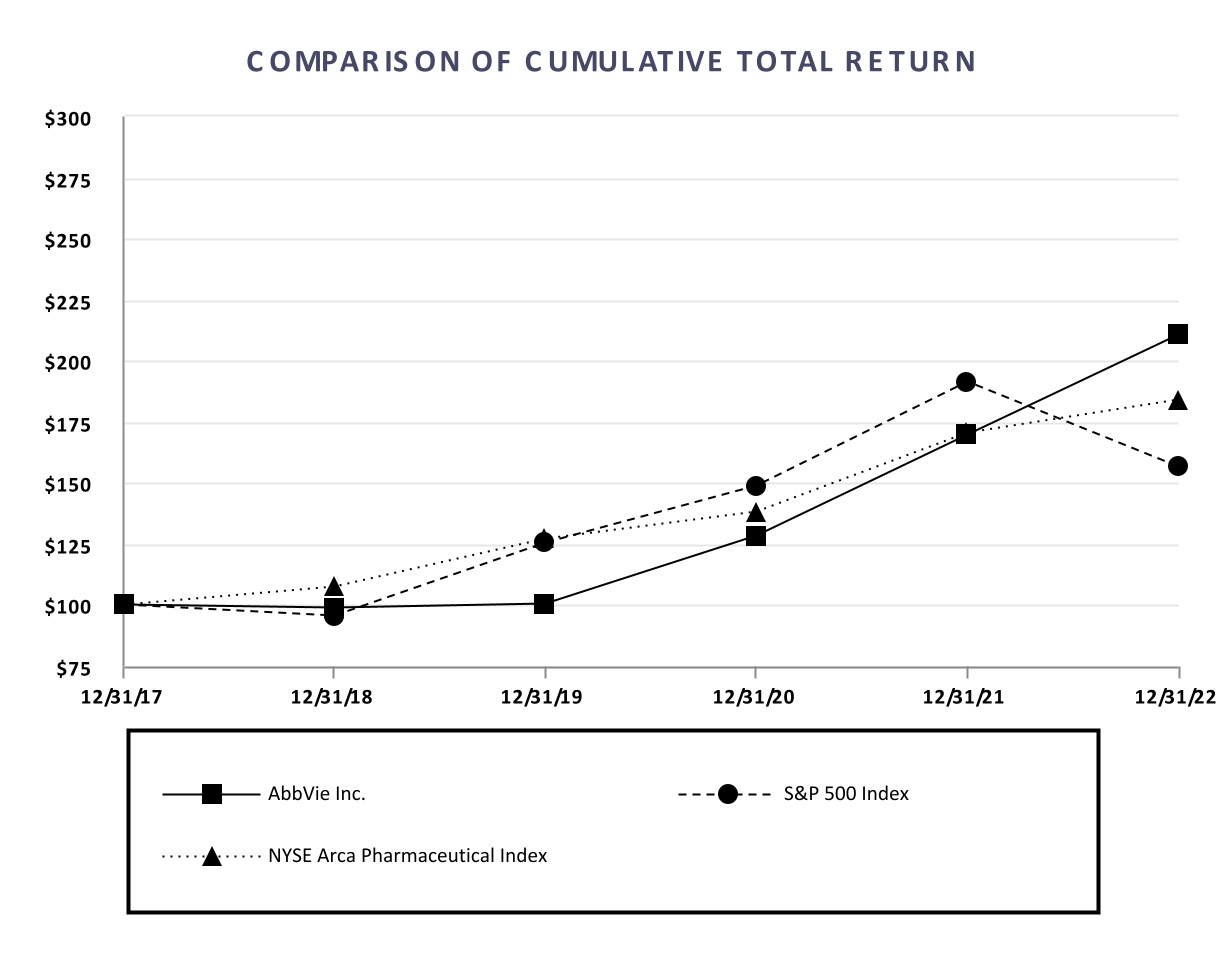
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K
 | 2022 Form 10-K
| 2022 Form 10-K