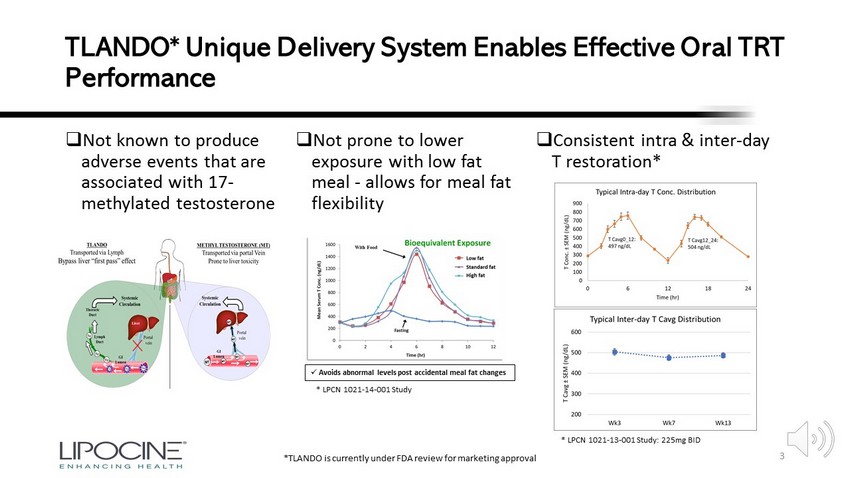
Is Oral Testosterone a Potential Treatment for COVID - 19 in Men? Benjamin J. Bruno, PharmD/PhD 1 ; Kongnara Papangkorn, PhD 1 , Anthony DelConte, MD 1,2 , Nachiappan Chidambaram, PhD 1 , Kilyoung Kim, PhD 1 , Mahesh V. Patel, PhD 1 1 1 Lipocine Inc., Salt Lake City, UT; 2 Saint Joseph’s University, Philadelphia, PA SMSNA 2020 * Drs. Bruno, Papangkorn, Chidambaram, Kim, and Patel are employees of Lipocine Inc. ** Dr. DelConte is a paid consultant to Lipocine Inc. Exhibit 99.2
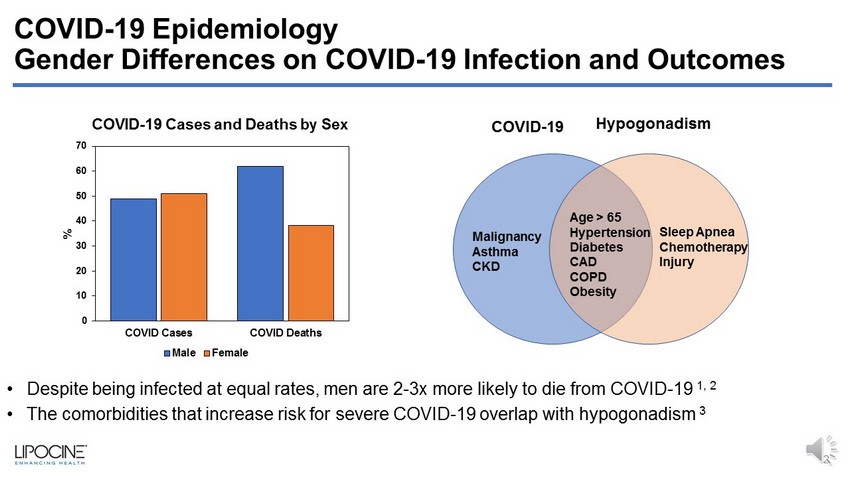
COVID - 19 Epidemiology Gender Differences on COVID - 19 Infection and Outcomes 2 Sleep Apnea Chemotherapy Injury Age > 65 Hypertension Diabetes CAD COPD Obesity Malignancy Asthma CKD COVID - 19 Hypogonadism 0 10 20 30 40 50 60 70 COVID Cases COVID Deaths % Male Female • Despite being infected at equal rates, men are 2 - 3x more likely to die from COVID - 19 1, 2 • The comorbidities that increase risk for severe COVID - 19 overlap with hypogonadism 3 COVID - 19 Cases and Deaths by Sex
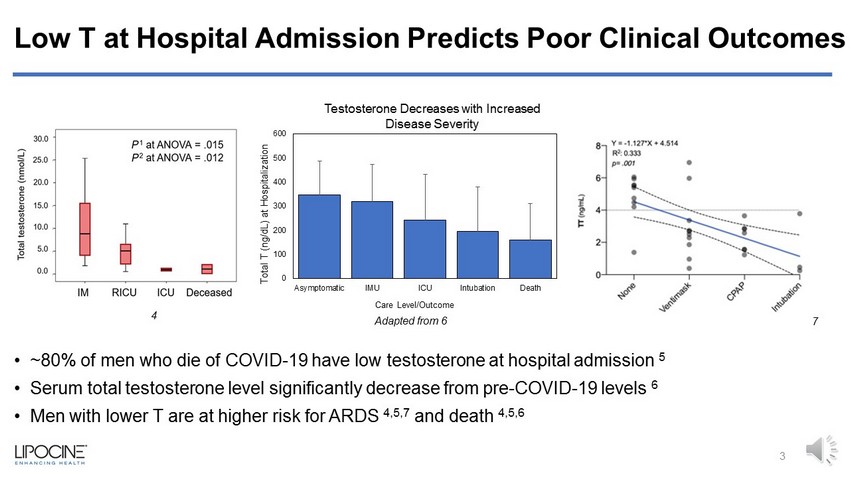
Low T at Hospital Admission Predicts Poor Clinical Outcomes • ~80% of men who die of COVID - 19 have low testosterone at hospital admission 5 • Serum total testosterone level significantly decrease from pre - COVID - 19 levels 6 • Men with lower T are at higher risk for ARDS 4,5,7 and death 4,5,6 0 100 200 300 400 500 600 Asymptomatic IMU ICU Intubation Death Total T (ng/dL) at Hospitalization Care Level/Outcome 3 Adapted from 6 4 7 Testosterone Decreases with Increased Disease Severity
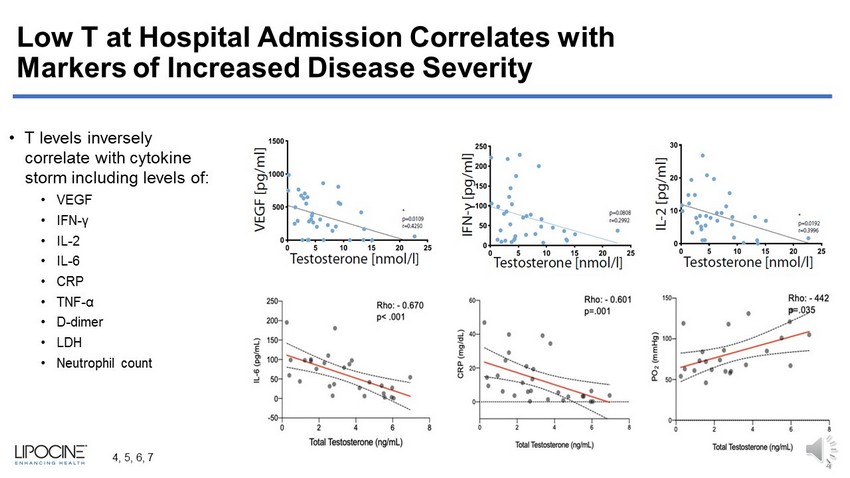
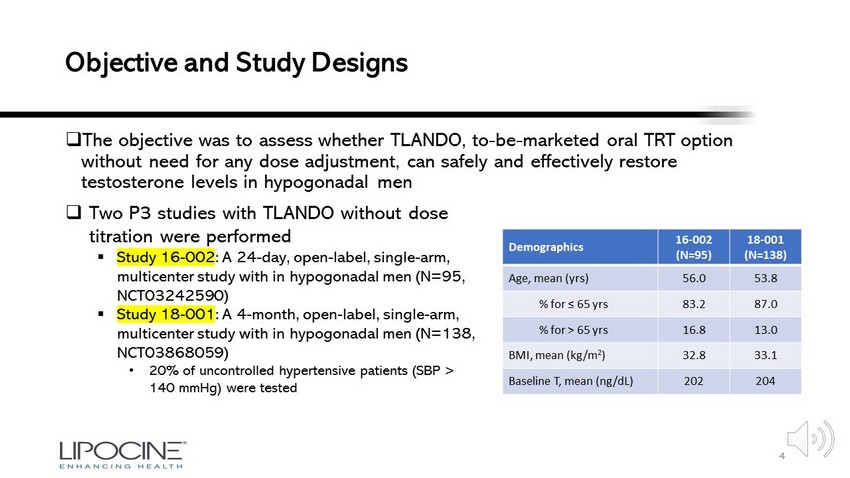
Low T at Hospital Admission Correlates with Markers of Increased Disease Severity • T levels inversely correlate with cytokine storm including levels of: • VEGF • IFN - γ • IL - 2 • IL - 6 • CRP • TNF - α • D - dimer • LDH • Neutrophil count 4 4, 5, 6, 7
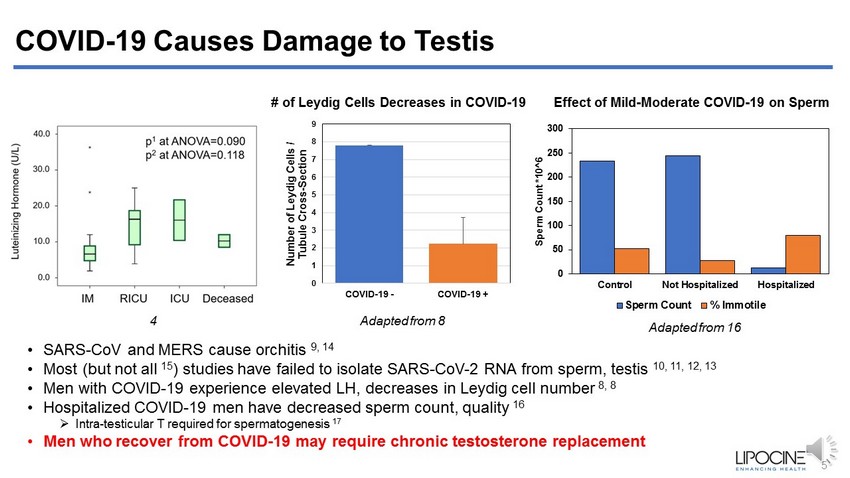
COVID - 19 Causes Damage to Testis • SARS - CoV and MERS cause orchitis 9, 14 • Most (but not all 15 ) studies have failed to isolate SARS - CoV - 2 RNA from sperm, testis 10, 11, 12, 13 • Men with COVID - 19 experience elevated LH, decreases in Leydig cell number 8, 8 • Hospitalized COVID - 19 men have decreased sperm count, quality 16 » Intra - testicular T required for spermatogenesis 17 • Men who recover from COVID - 19 may require chronic testosterone replacement 0 50 100 150 200 250 300 Control Not Hospitalized Hospitalized Sperm Count *10^6 Sperm Count % Immotile Adapted from 16 Adapted from 8 5 Effect of Mild - Moderate COVID - 19 on Sperm # of Leydig Cells Decreases in COVID - 19 0 1 2 3 4 5 6 7 8 9 COVID-19 - COVID-19 + Number of Leydig Cells / Tubule Cross - Section 4
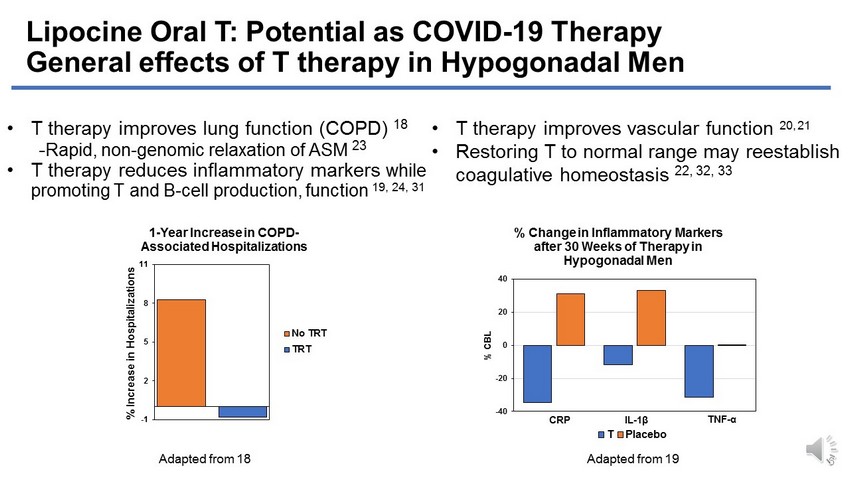
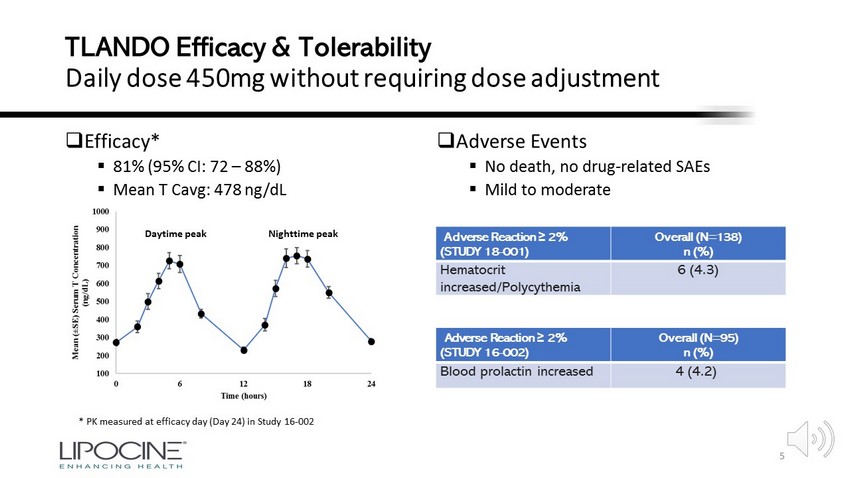
Lipocine Oral T: Potential as COVID - 19 Therapy General effects of T therapy in Hypogonadal Men • T therapy improves lung function (COPD) 18 - Rapid, non - genomic relaxation of ASM 23 • T therapy reduces inflammatory markers while promoting T and B - cell production, function 19, 24, 31 -40 -20 0 20 40 % CBL % Change in Inflammatory Markers after 30 Weeks of Therapy in Hypogonadal Men T Placebo CRP IL - 1 β TNF - α Adapted from 19 6 • T therapy improves vascular function 20, 21 • Restoring T to normal range may reestablish coagulative homeostasis 22, 32, 33 Adapted from 18 -1 2 5 8 11 1 % Increase in Hospitalizations 1 - Year Increase in COPD - Associated Hospitalizations No TRT TRT
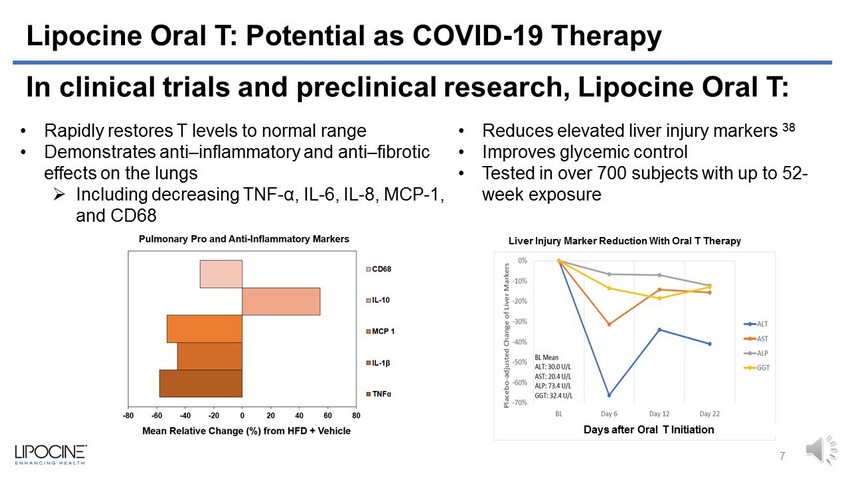
Lipocine Oral T: Potential as COVID - 19 Therapy • Rapidly restores T levels to normal range • Demonstrates anti – inflammatory and anti – fibrotic effects on the lungs » Including decreasing TNF - α , IL - 6, IL - 8, MCP - 1, and CD68 Days after Oral T Initiation 7 • Reduces elevated liver injury markers 38 • Improves glycemic control • Tested in over 700 subjects with up to 52 - week exposure In clinical trials and preclinical research, Lipocine Oral T: Liver Injury Marker Reduction With Oral T Therapy

Conclusions • Men are more susceptible to severe COVID - 19 and death • Men with COVID - 19 have low T, elevated LH, damaged Leydig cells, and elevated cytokines • T therapy in hypogonadal men improves lung and vascular function, is immunomodulatory, reducing cytokines and promoting B and T cell response • Oral T may improve COVID - 19 outcomes in men by rapidly restoring testosterone to the eugonadal range Future steps • Longitudinally measure testosterone levels in men hospitalized with COVID - 19 • Follow results of trials using antiandrogens to treat COVID - 19: - NCT04446429, NCT04475601, NCT04509999, NCT04374279, NCT04397718 • Interventional trial of testosterone in COVID - 19 8

1. Ng, J et al., COVID - 19 Mortality Rates by Age and Gender: Why is the Disease killing more Men than Women? RGA White Paper 2. Metro.co.uk l Source: Global Health l 50/50 3. CDC.gov 4. Rastrelli, G et al., Low testosterone levels predict clinical adverse outcomes in SARS - CoV - 2 pneumonia patients. Andrology, 2020 . 5. Schroeder, M et al. The majority of male patients with COVID - 19 present low testosterone levels on admission to Intensive Care i n Hamburg, Germany: a retrospective cohort study. medRxiv preprint, 2020. 6. Cayan, S et al., Effect of serum total testosterone and its relationship with other laboratory parameters on the prognosis of coronavirus disease 2019 (COVID - 19) in SARS - CoV - 2 infected male patients: a cohort study. The Aging Male, 2020. 7. Salciccia, S et al., Interplay between Male Testosterone Levels and the Risk for Subsequent Invasive Respiratory Assistance among COVID - 19 Patients at Hospital Admission. Endocrine, 2020. 8. Yang, M et al., Pathological Findings in the Testes of COVID - 19 Patients: Clinical Implications. Eur Uro Foc, 2020. 9. Xu, J et al. Orchitis: a complication of Severe Acute Respiratory Syndrome (SARS). Biol Reprod, 2006. 10. Song, C et al., Absence of 2019 novel coronavirus in semen and testes of COVID - 19 patients. Biol Reprod, 2020. 11. Pan, F et al., No evidence of severe acute respiratory syndrome – coronavirus 2 in semen of males recovering from coronavirus disease 2019. Fertil Steril, 2020. 12. Song, C et al., Detection of 2019 novel coronavirus in semen and testicular biopsy specimen of COVID - 19 patients. medRxiv preprint, 2020. 9 References

13. Guo, L et al., Absence of SARS - CoV - 2 in semen of a COVID - 19 patient cohort. Andrology, 2020. 14. Abobaker, A et al., Does COVID - 19 affect male fertility? World J Urol, 2020. 15. Li, D et al., Clinical Characteristics and Results of Semen Tests Among Men With Coronavirus Disease 2019. Infectious Disease s, 2020. 16. Holtmann, N et al., Assessment of SARS - CoV - 2 in human semen — a cohort study. Fertil Steril, 2020. 17. Walker, W, Testosterone signaling and the regulation of spermatogenesis. Spermatogenesis, 2011. 18. Baillargeon, J et al., Testosterone replacement therapy and hospitalization rates in men with COPD. Chron Resp Dis, 2018 19. Kalinchenko, S et al., Effects of testosterone supplementation on markers of the metabolic syndrome and inflammation in hypogonadal men with the metabolic syndrome: the double - blinded placebo - controlled Moscow study. Clin Endocrin, 2020. 20. Lopes, R et al., Testosterone and Vascular Function in Aging. Front Physiol, 2012. 21. Yaron, M et al. Effect of testosterone replacement therapy on arterial stiffness in older hypogonadal men. Eur J Endocrinol, 200 9. 22. Erem, C et al., Blood coagulation and fibrinolysis in male patient with hypogonadotropic hypogonadism: plasma factor V and fa cto r X activities increase in hypogonadotropic hypogonadism. J Endocrin, 2008. 23. Kouloumenta, V et al., Non - genomic effect of testosterone on airway smooth muscle. Br J Pharmacol, 2006. 24. Vignozzi, L et al., Antiinflammatory effect of androgen receptor activation in human benign prostatic hyperplasia cells. J En doc rin, 2012. 25. Trumble , B et al., Associations between male testosterone and immune function in a pathogenically stressed forager - horticultural population. Am J Phys Anthropol , 2016 10 References

References 26. Mann, D et al., Neonatal treatment with luteinizing hormone - releasing hormone analogs alters peripheral lymphocyte subsets and cellular and humorally mediated immune responses in juvenile and adult male monkeys. J Clin Endocrinol Metab, 1994. 27. Cain, D and Cidlowski, J, After 62 years of regulating immunity, dexamethasone meets COVID - 19. Nat Rev Immun, 2020. 28. Giagulli, V et al., Worse progression of COVID - 19 in men: Is testosterone a key factor? Andrology, 2020. 29. RECOVERY Collaborative Group, Dexamethasone in Hospitalized Patients with Covid - 19 — Preliminary Report. NEJM, 2020. 30. National Institutes of Health, Coronavirus Disease 2019 (COVID - 19) Treatment Guidelines. NIH.gov. 31. Page, T et al., Effect of medical castration on CD4+CD25+ T cells, CD8+ T cell IFN - γ expression, and NK cells: a physiological role for testosterone and/or its metabolites. Am J Physiol Endocrinol Metab, 2006. 32. Middleton, E et al., Neutrophil extracellular traps contribute to immunothrombosis in COVID - 19 acute respiratory distress syndro me. Blood, 2020. 33. Markman, J et al., Loss of testosterone impairs anti - tumor neutrophil function. Nat Commun, 2020. 34. 2020 Li, F et al. Distinct mechanisms for TMPRSS2 expression explain organ - specific inhibition of SARS - CoV - 2 infection by enzalutamide. bioRxiv preprint, 2020. 35. Baracthian, M et al. No evidence that androgen regulation of pulmonary TMPRSS2 explain sex - discordant COVID - 19 outcomes. bioRxiv preprint. 36. Wang, K et al., Angiotensin Converting Enzyme 2: A Double - Edged Sword. Circulation, 2020. 37. Ma, L et al., Effect of SARS - CoV - 2 infection upon male gonadal function: A single center - based study. medRxiv preprint, 2020. 38. Albhaisi, S et al., LPCN 1144 Resolves NAFLD in Hypogonadal Males. Hepatology Comm, 2020. 11

Appendix 12

Testosterone Normalizes, Dexamethasone Suppresses: Immunomodulation vs Immunosuppression • Dexamethasone suppresses innate and adaptive immune system » Including B cells and CD4+, CD8+ T cells 27 » Greatest benefit for patients with late disease stage 29, 30 » Need to avoid immunosuppression during viral phase? • T is required for proper immune response » Male hypogonadism is associated with impaired T - cell, 31 B - cell, antibody response 26 » T therapy in hypogonadal men curbed innate immune response, cytokines, while promoting T and B - cell production, function 24, 31 » T may inhibit production of clots, hypercoagulation 28, 32, 33 13 The Panel recommends against using dexamethasone for the treatment of COVID - 19 in patients who do not require supplemental oxygen (AI)
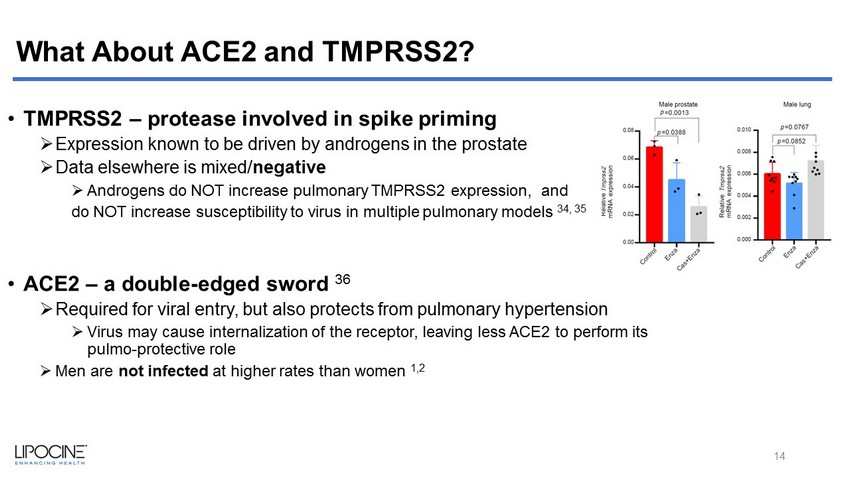
What About ACE2 and TMPRSS2? • TMPRSS2 – protease involved in spike priming » Expression known to be driven by androgens in the prostate » Data elsewhere is mixed/ negative » Androgens do NOT increase pulmonary TMPRSS2 expression, and do NOT increase susceptibility to virus in multiple pulmonary models 34, 35 • ACE2 – a double - edged sword 36 » Required for viral entry, but also protects from pulmonary hypertension » Virus may cause internalization of the receptor, leaving less ACE2 to perform its pulmo - protective role » Men are not infected at higher rates than women 1,2 14