Use these links to rapidly review the document
Table of contents
Index to consolidated financial statements
As filed with the Securities and Exchange Commission on November 9, 2017.
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
QUANTERIX CORPORATION
(Exact name of registrant as specified in its charter)
| Delaware (State or other jurisdiction of incorporation or organization) |
3826 (Primary Standard Industrial Classification Code Number) |
20-8957988 (I.R.S. Employer Identification Number) |
113 Hartwell Avenue
Lexington, MA 02421
(617) 301-9400
(Address, including zip code, and telephone number, including area code, of registrant's principal executive offices)
E. Kevin Hrusovsky
Executive Chairman, President and Chief Executive Officer
Quanterix Corporation
113 Hartwell Avenue
Lexington, MA 02421
(617) 301-9400
(Name, address, including zip code, and telephone number, including area code, of agent for service)
| Copies to: | ||||
William T. Whelan, Esq. Megan N. Gates, Esq. John P. Condon, Esq. Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. One Financial Center Boston, MA 02111 (617) 542-6000 |
Brian P. Keane, Esq. General Counsel Quanterix Corporation 113 Hartwell Avenue Lexington, MA 02421 (617) 301-9400 |
Patrick O'Brien, Esq. Ropes & Gray LLP Prudential Tower 800 Boylston Street Boston, MA 02199 (617) 951-7000 |
||
Approximate date of commencement of proposed sale to the public:
As soon as practicable after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933 check the following box. o
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See the definitions of "large accelerated filer," "accelerated filer," "smaller reporting company" and "emerging growth company" in Rule 12b-2 of the Exchange Act.
| Large accelerated filer o | Accelerated filer o | Non-accelerated filer ý (Do not check if a smaller reporting company) |
Smaller reporting company o Emerging growth company ý |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided to Section 7(a)(2)(B) of the Securities Act. o
CALCULATION OF REGISTRATION FEE
|
||||
| Title of each class of securities to be registered |
Proposed maximum aggregate offering price(1) |
Amount of registration fee(2) |
||
|---|---|---|---|---|
Common stock, $0.001 par value per share |
$57,500,000 | $7,158.75 | ||
|
||||
(1) Includes initial public offering price of shares that the underwriters have the option to purchase to cover overallotments, if any. Estimated solely for the purpose of calculating the amount of the registration fee pursuant to Rule 457(o) under the Securities Act of 1933, as amended.
(2) Calculated pursuant to Rule 457(o) based on an estimate of the proposed maximum aggregate initial public offering price.
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until this registration statement shall become effective on such date as the Commission, acting pursuant to said Section 8(a), may determine.
Subject to completion, dated November 9, 2017
The information in this preliminary prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell nor does it seek an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
Prospectus
shares

Common stock
This is an initial public offering of common stock by Quanterix Corporation. We are offering shares of our common stock. The estimated initial public offering price is between $ and $ per share.
Prior to this offering, there has been no public market for our common stock. We have applied to list our common stock on The Nasdaq Global Market under the symbol "QTRX."
We are an "emerging growth company" under applicable Securities and Exchange Commission rules and will be subject to reduced public company reporting requirements.
| | | | | | | | |
| |
Per share |
Total |
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
Initial public offering price |
$ | $ | |||||
Underwriting discounts and commissions(1) |
$ |
$ |
|||||
Proceeds to Quanterix Corporation, before expenses |
$ |
$ |
|||||
| | | | | | | | |
(1) The underwriters will receive compensation in addition to the underwriting discount. See "Underwriting" beginning on page 155.
The underwriters have the option to purchase up to an additional shares from us at the public offering price, less the underwriting discounts and commissions, within 30 days from the date of this prospectus to cover overallotments, if any.
Investing in our common stock involves a high degree of risk. See "Risk factors" beginning on page 14.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
The underwriters expect to deliver the shares of common stock to investors on or about , 2017.
Joint book running managers
| J.P. Morgan | Leerink Partners | |||
Co-managers |
||||
BTIG |
Evercore ISI |
|||
, 2017
We have not authorized anyone to provide any information or to make any representations other than those contained in this prospectus or in any free writing prospectuses we have prepared. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. This prospectus is an offer to sell only the shares offered hereby, but only under circumstances and in jurisdictions where it is lawful to do so. The information contained in this prospectus is current only as of its date.
For investors outside of the United States: We have not, and the underwriters have not, done anything that would permit this offering or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than the United States. Persons outside of the United States who come into possession of this prospectus must inform themselves about, and observe any restrictions relating to, the offering of the shares of common stock and the distribution of this prospectus outside of the United States.
Market and other industry data
Unless otherwise indicated, market data and certain industry forecasts used throughout this prospectus were obtained from various sources, including internal surveys, market research, consultant surveys, publicly available information and industry publications and surveys. Industry surveys, publications, consultant surveys and forecasts generally state that the information contained therein has been obtained from sources believed to be reliable, but that the accuracy and completeness of such information is not guaranteed. We have not independently verified any of the data from third-party sources nor have we ascertained the underlying economic assumptions relied upon therein. Similarly, internal surveys, industry forecasts and market research, which we believe to be reliable based upon our management's knowledge of the industry, have not been independently verified. This prospectus also contains estimates and other statistical data from a custom market research report by an independent third-party research firm, which was commissioned by us and was issued in June 2017. Such data involves a number of assumptions and
i
limitations and contains projections and estimates of the future performance of the markets in which we operate and intend to operate that are subject to a high degree of uncertainty. We caution you not to give undue weight to such projections, assumptions and estimates. The future performance of the industry and markets in which we operate and intend to operate is necessarily subject to a high degree of uncertainty and risk due to a variety of factors, including those described in the sections entitled "Risk factors" and "Special note regarding forward-looking statements" and elsewhere in this prospectus. These and other factors could cause results to differ materially from those expressed in these publications and reports.
ii
This summary highlights selected information contained elsewhere in this prospectus and does not contain all of the information that you should consider in making your investment decision. Before investing in our common stock, you should carefully read this entire prospectus, including our financial statements and the related notes thereto and the information set forth under the "Risk factors" and "Management's discussion and analysis of financial condition and results of operations," sections of this prospectus. Unless the context otherwise requires, the terms "Quanterix," the "Company," "we," "us" and "our" in this prospectus refer to Quanterix Corporation.
We are a life sciences company that has developed a next generation, ultra-sensitive digital immunoassay platform that advances precision health for life sciences research and diagnostics. Our platform enables customers to reliably detect protein biomarkers in extremely low concentrations in blood, serum and other fluids that, in many cases, are undetectable using conventional, analog immunoassay technologies. These capabilities provide our customers with insight into the role of protein biomarkers in human health that has not been possible with other existing technologies and enable researchers to better characterize the continuum between health and disease. We believe this greater insight provided by our platform, in research applications today and in diagnostic and precision health settings in the future, will enable the development of novel therapies and diagnostics and facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention. In addition to enabling new applications and insights in protein analysis, we are also developing our Simoa technology to detect nucleic acids in biological samples.
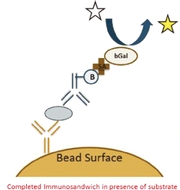
Our platform is based on our proprietary digital single molecule array, or Simoa, detection technology, which is the most sensitive commercially available protein detection technology. Simoa significantly advances ELISA technology, which has been the industry standard for protein detection for over forty years, through its ability to trap single molecules in tiny microwells that are 2.5 billion times smaller than traditional ELISA wells, allowing for an analysis and digital readout of each individual molecule, which is not possible with conventional ELISA technology. We believe Simoa's unprecedented sensitivity, combined with our target customers' familiarity with the core ELISA technology, provides us a significant competitive advantage in driving market adoption of our platform as well as integrating our products into our customers' workflows. We currently sell our Simoa products for research use only, but intend to expand into the diagnostic and precision health screening markets.
Researchers and clinicians rely extensively on protein biomarkers for use as research and clinical tools. However, normal physiological levels of many proteins are not detectable using conventional, analog immunoassay technologies, and many of these technologies can only detect proteins once they have reached levels that reflect more advanced disease or injury. We believe that the early detection of nascent disease or injury processes enabled by Simoa's sensitivity holds the key to intervention before disease or injury has advanced and more significant clinical signs and symptoms have appeared. Simoa's sensitivity also enables researchers to define and validate the function of novel protein biomarkers that are only present in very low concentrations and have been discovered using technologies such as mass spectrometry.
Protein expression reflects both genetic and environmental factors through a process whereby genetic information encoded in DNA is transcribed into RNA, which in turn is translated into proteins. Recently there have been significant advancements in understanding genetics due to the development of genomic
1
analytical technologies, such as polymerase chain reaction, or PCR, and next generation sequencing, which have significantly expanded the market for genetic analysis tools. While genomic analysis provides valuable information about the role of genes in health and disease, proteins are more prevalent than nucleic acids and, we believe, more relevant to understanding precisely the continuum between health and disease. Unlike the advancements in analytical tools for genomics, there has not been a corresponding advancement in tools for the analysis and detection of proteins. With our ultra-sensitive Simoa detection technology, researchers can assess the symptoms of disease or injury and compare them to the presence and levels of relevant proteins that are not detectable using conventional technologies, leading to a better understanding of how proteins individually and/or collectively impact and influence important biological processes and the health and well-being of individuals. We believe understanding the individual characteristics and functioning of proteins enabled by Simoa will be central to the development of novel therapies as well as to earlier disease detection, monitoring, prognosis and, ultimately, prevention, by providing researchers with the ability to assess the impact of low abundance and other proteins on the progress of disease and injury from the time of early onset of symptoms.
While our initial focus has been on the use of Simoa to detect protein biomarkers, Simoa is also able to directly detect nucleic acids in biological samples. In nucleic acid analysis, Simoa has the potential to provide the same sensitivity as traditional PCR-based assays without some of its inherent drawbacks, such as amplification bias. We believe the ability of our platform to provide our customers with both proteomic and genomic solutions will further drive adoption of our technology.
We intend to commercialize our Simoa technology in the life science research, diagnostics and precision health screening markets. Our initial target market has been the life science research market, and all of our product and service revenue to date has been in this market. While we have received revenue from upfront and milestone payments related to collaborations with diagnostic companies, neither we nor any of our diagnostic partners have sold Simoa products or services in the diagnostics or precision health screening markets. We have focused on areas of high growth and unmet need where existing platforms have significant shortcomings that our technology addresses. Specifically, our focus areas include: neurology, oncology, cardiology, infectious disease and inflammation. As our customers continue to gain experience with our proprietary Simoa technology, we believe the opportunity to access markets beyond research will be significant. According to estimates in a report commissioned by us from an independent third-party research firm, referred to herein as the Third-Party Research Report, we believe the current total life science research market addressable by Simoa, including both proteomics and genomics research, is currently $3 billion per year and has the potential to reach $8 billion per year. In addition, according to the Third-Party Research Report, we estimate that the future aggregate market opportunity for us or others using our Simoa technology has the potential to expand to approximately $38 billion, approximately $30 billion of which would be addressable by the Simoa technology upon receipt of the necessary regulatory approvals to market products using this technology in areas other than life science research, which neither we nor our partners have begun the process to obtain. To the extent any collaborators or licensees, such as bioMérieux SA, pursue a portion of this addressable market using Simoa technology licensed from us, our participation in this market size could be limited to receipt of royalties and milestone payments rather than through direct sales.
- •
- Life science research. We believe Simoa is well-positioned to capture a significant share of the large and growing life science research market because of its superior sensitivity. It has the ability to detect both proteins that are currently undetectable by other technologies, and nucleic acids directly. According to estimates in the Third-Party Research Report, we believe that the total life science research market addressable by Simoa is currently $3 billion per year and has the potential to reach $8 billion per year.
2
- •
- Diagnostics. We believe existing
diagnostics can be improved by Simoa's sensitivity to enable earlier detection of diseases and injuries, and that new diagnostics may be developed using protein biomarkers that are undetectable using
conventional, analog immunoassay technologies but are detectable using Simoa. We also believe that our platform can enable the development of new diagnostic tests based on blood, serum, saliva and
other fluids that have the potential to replace current more invasive, expensive and inconvenient diagnostic methods, such as spinal tap, diagnostic imaging and biopsy. Simoa also has significant
potential in the emerging field of companion diagnostics based on novel biomarkers.
- •
- Precision health screening. We believe that Simoa's ability to detect and quantify normal physiological levels of proteins in low abundance that are undetectable using conventional, analog immunoassay technologies may enable our technology to be used to monitor protein biomarker levels of seemingly healthy, asymptomatic people, and potentially to signal and provide earlier detection of the onset of disease.
According to estimates in the Third-Party Research Report, we believe that the total diagnostic and precision health screening markets addressable by us and others using Simoa have the potential to reach an aggregate of $30 billion per year, which would be addressable upon receipt of the necessary regulatory approvals to market our products in areas other than life science research, which we have not yet begun the process to obtain.
Products sold by us or collaborators in the diagnostics and precision health screening markets will be subject to regulation by the United States Food and Drug Administration, or FDA, or comparable international agencies, including requirements for regulatory clearance or approval of such products before they can be marketed. To date, neither we nor any of our diagnostic partners have received or applied for regulatory approvals for Simoa products. See "Risk factors—Risks related to governmental regulation and diagnostic product reimbursement" and "Business—Government regulation" for a more detailed discussion regarding the regulatory approvals that may be required.
Our Simoa HD-1 Analyzer, which was launched in January 2014, is the most sensitive protein detection platform commercially available and is currently capable of analyzing up to six biomarkers per test, with anticipated expansion capability to up to 35 biomarkers per test in 2018. Assays run on the HD-1 Analyzer are fully automated, which we believe provides us with an additional significant competitive advantage with biopharmaceutical customers. We have currently developed more than 80 Simoa digital biomarker assays and continue to expand our assay menu. We have sold more than 160 HD-1 Analyzers to over 100 customers around the world. We also have seven HD-1 Analyzers in our own Simoa Accelerator Laboratory.
We have developed a new instrument, the Quanterix SR-X, which we plan to introduce through an early adopter program and launch commercially in 2018. The Quanterix SR-X will utilize the same core Simoa technology and assay kits as the HD-1 Analyzer in a compact benchtop form with a lower price point, more flexible assay preparation, and a wider range of applications, including direct detection of nucleic acids.
We also provide contract research services for customers through our Simoa Accelerator Laboratory which provides customers with access to Simoa's technology, and supports multiple projects and services, including sample testing, homebrew assay development and custom assay development. To date, we have completed over 340 projects for more than 145 customers from all over the world using our Simoa platform.
In order to accelerate the use of our technology to develop applications in the diagnostics market, we have entered into a collaboration with bioMérieux SA, a leading diagnostic company, pursuant to which we have granted them an exclusive license to develop and sell in vitro diagnostic products used in clinical lab
3
applications and for food quality control testing, and pharmaceutical quality control testing based on our Simoa technology and a co-exclusive license for other in vitro diagnostic products, relating to point-of-care testing and laboratory developed testing. Given the exclusive nature of the license rights granted to bioMérieux in the areas of in vitro diagnostics used in clinical lab applications, food quality control testing, and pharmaceutical quality control testing, our ability to collaborate with others in these areas is limited, subject to our right to make and sell the current version of the Simoa HD-1 Analyzer for use in clinical lab applications, either directly or through a partner. Neither we nor bioMérieux have begun the process to secure regulatory approvals or clearances to market products using our Simoa technology in areas other than life science research. See "Business—Key agreements—License agreement with bioMérieux SA" for a more detailed description of this collaboration arrangement.
- •
- Proprietary ultra-sensitive digital immunoassay Simoa technology platform, enabling researchers and clinicians to
obtain information from less invasive procedures and smaller sample sizes. Simoa's sensitivity allows researchers and clinicians to measure critical protein biomarkers at
earlier stages in the progression of a disease or injury, which we believe will enable the development of novel therapies and diagnostics and facilitate a paradigm shift in healthcare from an emphasis
on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention. The sensitivity of our Simoa technology allows researchers to gather biomarker information from smaller
samples that can be collected less invasively than samples required by other assay technologies.
- •
- Technology platform that leverages and improves upon industry standard ELISA
technology. Simoa uses the basic principles of conventional bead-based ELISA. However, unlike ELISA, which runs the enzyme-substrate reactions on
all molecules in one well, Simoa reactions are run on individual molecules in tiny microwells, 40 trillionths of a milliliter, that are 2.5 billion times smaller than traditional ELISA
wells. We believe Simoa's unprecedented sensitivity, combined with our target customers' familiarity with the core ELISA technology, provides us a significant competitive advantage as well as the
ability to integrate into our customers' workflows.
- •
- Leader in large and growing market for detecting proteins in low
abundance. Simoa is the most sensitive commercially available protein detection technology. We believe our growing market acceptance is
establishing Simoa as the reference platform for detecting proteins in low abundance across sample types in our end markets.
- •
- Deep and expanding scientific
validation. Our Simoa technology has been cited in approximately 160 articles in peer reviewed publications covering over 150 biomarkers, and is
becoming a vital tool in cutting edge life sciences research. Our company has established relationships with key opinion leaders, and our growing base of over 200 customers includes some of the
world's leading academic and government research centers, as well as 17 of the 20 largest pharmaceutical and biotechnology companies.
- •
- Leading position in market solidified by robust customization capabilities, assay design flexibility and automation of our HD-1 Analyzer. Our technical capabilities and expertise allow our customers to design high-quality, customized assays utilizing our Simoa platform. The flexibility of the Simoa detection technology allows us to provide innovative, low cost solutions for customers in multiple markets across various applications. In addition, the Simoa HD-1 Analyzer provides fully automated analysis from sample introduction to analytical results. Furthermore, our proprietary array approach to ELISA digitization enables rapid digital data acquisition and assay results.
4
- •
- Highly attractive business model that leverages growing installed base of
instruments. We have sold more than 160 HD-1 Analyzers to over 100 customers around the world and plan to commercially launch our latest
instrument, the Quanterix SR-X, in 2018. As we continue to grow our installed base, optimize workflows and expand our assay menu, we expect to increase our revenues derived from consumables. The
integration of our technology in our customers' projects also provides ongoing sales of assays and consumables, resulting in a growing revenue stream.
- •
- Our highly experienced senior management team. We are led by a dedicated and highly experienced senior management team with significant industry experience and proven ability to develop novel solutions. Each of the members of our senior management has more than 20 years of relevant experience.
Proteins are versatile macromolecules and serve critical functions in nearly all biological processes. They are complex molecules that organisms require for the structure, function and regulation of the body's tissues and organs. For example, proteins provide immune protection, generate movement, transmit nerve impulses and control cell growth and differentiation. Understanding an organism's proteome, the complete set of proteins and their expression levels, can provide a powerful and unique window into its health, a window that other types of research, such as genomics, cannot provide.
The human body contains approximately 20,000 genes. One of the core functions of genes, which are comprised of DNA, is to regulate protein production—which ones are produced, the volume of each, and for how long—influenced by both biological and environmental factors. These 20,000 genes help govern the expression of over 100,000 proteins, approximately 10,500 of which are known to be secreted in blood, and fewer than 1,300 of which can be consistently detected in healthy individuals using conventional immunoassay technologies. Accordingly, the study of much of the proteome has not been practical given the limited level of sensitivity of existing technologies. To date, we have developed assays that address approximately 80 of the proteins secreted in blood. We estimate that the current sensitivity of our Simoa technology has the potential to detect and measure up to one-third of the approximately 9,200 proteins secreted in blood that are not consistently detectable using conventional immunoassay technologies.
While research on nucleic acids provides valuable information about the role of genes in health and disease, proteins are both more prevalent than nucleic acids and, we believe, more relevant to understand precisely the nuanced continuum between health and disease. Genes may indicate the risk of developing a certain disease later in life, but they are not able to account for the impact of environmental factors and lifestyle, such as diet and exercise, or provide insight into what is happening in a patient's body in real time.
Much like the sequencing of the human genome with the Human Genome Project and the development of both PCR and next generation sequencing technologies to detect nucleic acids, both of which accelerated biomedical genomic research, we believe the ability to study more of the proteome will be enabled by a more sensitive protein detection technology and will have a profound impact on proteomic research. Using our Simoa technology, researchers can gain insight into how these proteins are individually and/or collectively important contributors to health and well-being, as well as injury and disease.
5
Existing technologies and their limitations
Protein analysis
The enzyme-linked immunosorbent assay, or ELISA, has been the most widely used method of sensitive detection of proteins for over 40 years. In ELISA, an unknown amount of antigen (e.g., protein, peptide, antibody, hormone) is affixed to a solid surface, usually a polystyrene multiwall plate, either directly, or indirectly through use of a conjugated secondary or "capture" antibody (sandwich ELISA). A specific "detection" antibody is applied over the surface to bind to the antigen. This detection antibody is linked to an enzyme, and in the final step, a substance called an enzyme substrate is added, and the enzyme converts to colored or fluorescent product molecules, which are detected by a plate reader.
Aside from ELISA, there are other technologies available for protein analysis today, such as Western blotting, mass spectrometry, chromatography, surface plasmon resonance, Raman-enhanced signal detection, immune-PCR and biobarcode assay. However, the proteins detectable by these conventional, analog immunoassay technologies are fewer than 1,300 of what is estimated to be approximately 10,500 secreted proteins in circulation in human blood. While a number of techniques have been used to attempt to increase sensitivity of detection, we believe all of these approaches have limitations, including:
- •
- dilution of colored or fluorescent product molecules due to large volume of liquid in traditional-sized wells, limiting sensitivity;
- •
- narrow dynamic range (i.e., the range of concentration of proteins being detected), that may require sample dilution, diluting molecules
and increasing sample volume requiring additional enzymes to reach detection limit;
- •
- low detection limit of readers restrict sensitivity and ability to detect low-abundance proteins, particularly when proteins are at normal
physiological levels; and
- •
- limited success in increasing sensitivity of detection due to procedural complexity and length.
Genomic analysis
Over the past few decades, scientists have developed a variety of genomic analysis methods to measure an increasing number of genomic biomarkers aimed at more effectively detecting diseases. The most widely used method for genetic testing is PCR, which involves amplifying, or generating billions of copies of, the DNA sequence in question and then detecting the DNA with the use of fluorescent dyes. The expression of the nucleic acid is then inferred based on the number of amplification cycles required for the target to become detectable. The wide availability of PCR chemistry makes it a popular approach for measuring the expression of nucleic acids, but the use of enzymes in numerous cycles of amplification can introduce distortion and bias into the data, potentially compromising the reliability of results, particularly at low concentrations.
Our Simoa platform is highly flexible, and provides superior sensitivity, automated workflow capabilities, multiplexing and works with a broad range of sample types. We believe these characteristics will drive adoption of Simoa in life science research, diagnostics and precision health screening markets.
6
We believe our platform provides the following advantages over other technologies available for protein analysis today:
- •
- Simoa digital technology measurements are independent of sample concentration intensity and rely on a binary signal/no signal readout, enabling
detection sensitivity that was not previously possible;
- •
- Enables direct detection of low abundant and previously undetectable biomarkers;
- •
- Simoa multiplexing maintains single plex precision, while competitive platforms can lose sensitivity when multiplexing is used; and
- •
- Simoa's automation and speed provides customers high research and development productivity through greater throughput and lab efficiency.
Our initial focus has been on the use of Simoa to detect protein biomarkers. However, the role of genomic information in research and medicine is evolving rapidly, and our Simoa technology is also able to detect nucleic acids in biological samples. While methods for measuring nucleic acid molecules have advanced substantially, currently available techniques such as PCR still have drawbacks. In nucleic acid analysis, we believe that Simoa has the potential to provide the same sensitivity as traditional PCR-based assays with the following benefits:
- •
- No need for amplification of the targeted nucleic acid, which can result in amplification distortion and bias;
- •
- Reduced cross-contamination because of direct detection of single molecules as compared to the detection of a large number of copies of the
nucleic acid; and
- •
- The ability to detect nucleic acids directly from samples, such as from environmental water, without requiring purification.
Our goal is to enable new research into proteins and nucleic acids to allow greater insight into their role in human health in ways that have not been possible with any other current research and diagnostic technologies. We believe this greater insight will facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention.
Our strategy to achieve this includes:
- •
- Focus on the highly attractive, expanding market for protein detection and analysis;
- •
- Continue to drive adoption of the Simoa platform in the research, diagnostics and precision health screening markets;
- •
- Leverage the Simoa "ecosystem" to grow our customer base and further penetrate our existing customer base;
- •
- Utilize the flexibility of the Simoa platform to expand into complementary markets, including nucleic acid detection;
- •
- Leverage the data generated by Simoa to drive adoption of our technology; and
- •
- Grow into new markets organically with our customers and through strategic collaborations.
7
Investing in our common stock involves substantial risk. You should carefully consider all of the information in this prospectus prior to investing in our common stock. There are several risks related to our business that are described under "Risk factors" elsewhere in this prospectus. Among these important risks are the following:
- •
- We have incurred losses since we were formed and expect to incur losses in the future. We cannot be certain that we will achieve or sustain
profitability;
- •
- Our quarterly and annual operating results and cash flows have fluctuated in the past and might continue to fluctuate, causing the value of our
common stock to decline substantially;
- •
- We are an early, commercial-stage company and have a limited operating history, which may make it difficult to evaluate our current business
and predict our future performance;
- •
- If we are unable to maintain adequate revenue growth or do not successfully manage such growth, our business and growth prospects will be
harmed;
- •
- Our future capital needs are uncertain and we may need to raise additional funds in the future;
- •
- If our products fail to achieve and sustain sufficient market acceptance, our revenue will be adversely affected;
- •
- Our future success is dependent upon our ability to further penetrate our existing customer base and attract new customers;
- •
- Some of the reagents used in our products are labeled "research use only" and will have to undergo additional testing before we could use them
in a product intended for clinical use;
- •
- In the near term, our business will depend on levels of research and development spending by academic and governmental research institutions
and biopharmaceutical companies, a reduction in which could limit demand for our products and adversely affect our business and operating results;
- •
- If we do not successfully develop and introduce new assays for our technology, we may not generate new sources of revenue and may not be able
to successfully implement our growth strategy;
- •
- If the FDA determines that our products are medical devices or if we seek to market our products for clinical diagnostic or health screening
use, we will be required to obtain regulatory clearance(s) or approval(s), and any such regulatory process would be expensive, time-consuming, and uncertain both in timing and in outcome;
- •
- If we are unable to protect our intellectual property, it may reduce our ability to maintain any technological or competitive advantage over
our competitors and potential competitors, and our business may be harmed; and
- •
- Our principal stockholders and management own a significant percentage of our stock and will be able to exercise significant influence over matters subject to stockholder approval.
Implications of being an emerging growth company
We are an emerging growth company as defined in the Jumpstart Our Business Startups Act of 2012, as amended, or the JOBS Act. We will remain an emerging growth company until the earlier of (1) the last day of the fiscal year following the fifth anniversary of the completion of this offering, (2) the last day of the fiscal year in which we have total annual gross revenue of at least $1.07 billion, (3) the last day of the fiscal year in which we are deemed to be a "large accelerated filer" as defined in Rule 12b-2 under the Securities Exchange Act of 1934, as amended, or the Exchange Act, which would occur if the market value
8
of our common stock held by non-affiliates exceeded $700.0 million as of the last business day of the second fiscal quarter of such fiscal year or (4) the date on which we have issued more than $1.0 billion in non-convertible debt securities during the prior three-year period. An emerging growth company may take advantage of specified reduced reporting requirements and is relieved of certain other significant requirements that are otherwise generally applicable to public companies. As an emerging growth company,
- •
- we may present only two years of audited financial statements, plus unaudited condensed financial statements for any interim period, and
related management's discussion and analysis of financial condition and results of operations in our initial registration statement;
- •
- we may avail ourselves of the exemption from the requirement to obtain an attestation and report from our auditors on the assessment of our
internal control over financial reporting pursuant to the Sarbanes-Oxley Act of 2002, or Sarbanes-Oxley;
- •
- we may provide reduced disclosure about our executive compensation arrangements; and
- •
- we may not require stockholder non-binding advisory votes on executive compensation or golden parachute arrangements.
We have elected to take advantage of the extended transition period afforded by the JOBS Act for the implementation of new or revised accounting standards and, as a result, will comply with new or revised accounting standards not later than the relevant dates on which adoption of such standards is required for non-public companies. There are currently accounting standards that are expected to affect the financial reporting of many public companies as early as the first calendar quarter of 2018, including ASC 606, Revenue from contracts with customers. As a result of this election, the timeline to comply with these standards will in many cases be delayed as compared to other public companies that are not eligible to have made or have not made this election. For more information on the effect of this election, including the timing of when we currently plan to adopt certain accounting standards that could materially affect our financial statements, refer to Note 2 to our consolidated financial statements appearing at the end of this prospectus.
We were incorporated under the laws of the State of Delaware in April 2007 under the name "Digital Genomics, Inc." In August 2007, we changed our name to "Quanterix Corporation." Our principal executive offices are located at 113 Hartwell Avenue, Lexington, MA 02421, and our telephone number is (617) 301-9400. Our website address is www.quanterix.com. The information contained on, or that can be accessed through, our website is not part of this prospectus. We have included our website address in this prospectus solely as an inactive textual reference. Investors should not rely on any such information in deciding whether to purchase our common stock.
"Quanterix," "Simoa," "Simoa HD-1," "SR-X," "HD-1 Analyzer" and our logo are our trademarks. All other service marks, trademarks and trade names appearing in this prospectus are the property of their respective owners. We do not intend our use or display of other companies' trade names, trademarks or service marks to imply a relationship with, or endorsement or sponsorship of us by, these other companies.
9
| Common stock offered by us | shares | |
Common stock to be outstanding after this offering |
shares |
|
Option to purchase additional shares |
The underwriters have an option within 30 days of the date of this prospectus to purchase up to additional shares of our common stock to cover over-allotments, if any. |
|
Use of proceeds |
We estimate the net proceeds from this offering will be approximately $ million (or $ million if the underwriters exercise their option to purchase additional shares in full), assuming an initial public offering price of $ per share, which is the midpoint of the estimated price range set forth on the cover page of this prospectus, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
|
We intend to use the net proceeds from the offering: (1) to expand our life sciences commercial operations to grow and support the installed base of our products among life sciences research customers in the United States and internationally; (2) to improve and update our Simoa technology and instruments and to develop additional assays, including assays for nucleic acid detection; (3) to support the launch of our new Quanterix SR-X instrument, currently scheduled for launch in 2018; (4) to potentially move into a larger corporate headquarters in order to have the appropriate infrastructure to support the increase in our employee base in addition to an increase in our manufacturing footprint; (5) to pursue regulatory approvals or clearances to develop instruments, assay kits and consumables in areas outside of life science research, including potentially LDTs, IVD tests and other markets, and, subject to the receipt of such necessary regulatory approvals or clearances, to develop such instruments, assay kits and consumables; (6) to potentially pursue acquisitions or other business development opportunities; and (7) for working capital and other general corporate purposes. See "Use of proceeds" for additional information. |
||
Risk factors |
You should read the "Risk factors" section of this prospectus beginning on page 14 and other information included in this prospectus for a discussion of factors to consider carefully before deciding to invest in shares of our common stock. |
|
Proposed Nasdaq Global Market symbol |
"QTRX" |
10
The number of shares of our common stock to be outstanding after this offering is based on 54,330,740 shares of our common stock outstanding as of October 31, 2017, including 676,121 shares of unvested restricted common stock, after giving effect to the assumptions set forth below and excluding the following:
- •
- 7,327,673 shares of common stock issuable upon the exercise of outstanding stock options as of October 31, 2017, having a
weighted-average exercise price of $1.83 per share;
- •
- 387,811 shares of common stock issuable upon the exercise of outstanding warrants as of October 31, 2017, having a
weighted-average exercise price of $2.98 per share; and
- •
- shares of common stock reserved for issuance pursuant to future awards under our 2017 Equity Incentive Plan, as well as any automatic increases in the number of shares of our common stock reserved for future issuance under this plan, which will become effective upon the closing of this offering.
Except as otherwise indicated, all information contained in this prospectus assumes or gives effect to:
- •
- the automatic conversion of all of our outstanding shares of preferred stock into an aggregate of 45,561,745 shares of common stock
prior to the completion of this offering;
- •
- the automatic conversion of warrants to purchase (i) 64,441 shares of our Series A-2 preferred stock into warrants to
purchase 64,441 shares of common stock, (ii) 284,542 shares of our Series C preferred stock into warrants to purchase 284,542 shares of common stock and
(iii) 38,828 shares of our Series D preferred stock into warrants to purchase 38,828 shares of common stock prior to the completion of this offering;
- •
- no exercise by the underwriters of their option purchase up to an
additional shares of our common stock in this offering;
- •
- the adoption of our restated certificate of incorporation and restated by-laws prior to the closing of this offering; and
- •
- a one-for- reverse stock split of our common stock effected on , 2017.
11
You should read the following summary financial data together with our financial statements and the related notes appearing at the end of this prospectus and the "Selected financial data" and "Management's discussion and analysis of financial condition and results of operations" sections of this prospectus. We have derived the statement of operations data for the years ended December 31, 2015 and 2016 from our audited consolidated financial statements included elsewhere in this prospectus. The statement of operations data for the nine months ended September 30, 2016 and 2017 and the balance sheet data as of September 30, 2017 have been derived from our unaudited consolidated financial statements included elsewhere in this prospectus and which have been prepared on the same basis as the audited consolidated financial statements. In the opinion of management, the unaudited data reflects all adjustments, consisting only of normal recurring adjustments, necessary for a fair presentation of the financial information in those statements. Our historical results are not necessarily indicative of results that should be expected in the future.
Consolidated statement of operations data (in thousands, except per share data)
| | | | | | | | | | | | | | |
| |
Year ended December 31 |
Nine months ended September 30 |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Total revenue |
$ | 12,180 | $ | 17,585 | $ | 10,906 | $ | 16,285 | |||||
Cost of revenue |
6,465 |
9,837 |
6,746 |
9,179 |
|||||||||
Research and development |
10,083 | 16,993 | 10,192 | 12,377 | |||||||||
Selling, general and administrative |
10,155 | 12,466 | 8,866 | 13,641 | |||||||||
| | | | | | | | | | | | | | |
Total operating expenses |
26,703 | 39,296 | 25,804 | 35,197 | |||||||||
| | | | | | | | | | | | | | |
Loss from operations |
(14,523 |
) |
(21,711 |
) |
(14,898 |
) |
(18,912 |
) |
|||||
Interest expense, net |
(1,040 |
) |
(1,298 |
) |
(1,012 |
) |
(735 |
) |
|||||
Other income (expense), net |
(380 | ) | (164 | ) | 51 | 10 | |||||||
| | | | | | | | | | | | | | |
Net loss |
(15,943 | ) | (23,173 | ) | (15,859 | ) | (19,637 | ) | |||||
Accretion and accrued dividends on redeemable convertible preferred stock |
(4,355 |
) |
(4,445 |
) |
(3,325 |
) |
(3,349 |
) |
|||||
| | | | | | | | | | | | | | |
Net loss attributable to common stockholders |
$ | (20,298 | ) | $ | (27,618 | ) | $ | (19,184 | ) | $ | (22,986 | ) | |
| | | | | | | | | | | | | | |
Net loss per share attributable to common stockholders, basic and diluted |
$ | (3.48 | ) | $ | (4.01 | ) | $ | (2.83 | ) | $ | (2.96 | ) | |
| | | | | | | | | | | | | | |
Weighted-average common shares outstanding |
5,828 | 6,887 | 6,782 | 7,768 | |||||||||
| | | | | | | | | | | | | | |
12
Consolidated balance sheet data (in thousands)
| |
|
|
|
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | |
| |
As of September 30, 2017 | |||||||||
| |
Actual |
Pro forma(1) |
Pro forma as adjusted(2) |
|||||||
| | | | | | | | | | | |
| |
|
(unaudited) |
|
|||||||
Cash and cash equivalents |
$ | 18,690 | $ | 18,690 | $ | |||||
Total assets |
30,515 | 30,515 | ||||||||
Total long term debt |
9,328 | 9,328 | 9,328 | |||||||
Total redeemable convertible preferred stock |
142,387 | — | — | |||||||
Total stockholders' (deficit) equity |
(136,544 | ) | 6,624 | |||||||
| | | | | | | | | | | |
(1) The summary pro forma balance sheet data as of September 30, 2017 has been prepared to give effect to the conversion of all outstanding shares of preferred stock into an aggregate of 45,561,745 shares of our common stock prior to the completion of this offering and the conversion of warrants to purchase 387,811 shares of our preferred stock into warrants to purchase 387,811 shares of common prior to the completion of this offering. The summary pro forma balance sheet data is for informational purposes only and does not purport to indicate balance sheet data as of any future date.
(2) The summary pro forma as adjusted balance sheet data as of September 30, 2017 has been prepared to give effect to the pro forma adjustments and to further reflect the issuance and sale by us of shares of our common stock in this offering, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, at an assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus. The summary pro forma as adjusted balance sheet data is for informational purposes only and does not purport to indicate balance sheet data as of any future date.
13
Investing in our common stock involves a high degree of risk. Before making an investment decision, you should carefully consider the risks and uncertainties described below, which we believe are the material risks associated with our business and this offering. If any of the following risks were to materialize, our business, financial condition, results of operations, and future growth prospects could be materially and adversely affected. In that event, the trading price of our common stock could decline, and you may lose all or part of your investment. In assessing these risks, you should also refer to all of the other information contained in this prospectus, including our financial statements and related notes.
Risks related to our financial condition and need for additional capital
We have incurred losses since we were formed and expect to incur losses in the future. We cannot be certain that we will achieve or sustain profitability.
We incurred net losses of $15.9 million and $23.2 million for the years ended December 31, 2015 and 2016, respectively, and $19.6 million for the nine months ended September 30, 2017. As of September 30, 2017, we had an accumulated deficit of $136.6 million. We cannot predict if we will achieve sustained profitability in the near future or at all. We expect that our losses will continue at least through the next 24 months as we plan to invest significant additional funds toward expansion of our commercial organization and the development of our technology and related assays. In addition, as a public company, we will incur significant legal, accounting, and other expenses that we did not incur as a private company. These increased expenses will make it harder for us to achieve and sustain future profitability. We may incur significant losses in the future for a number of reasons, many of which are beyond our control, including the other risks described in this prospectus, the market acceptance of our products, future product development and our market penetration and margins.
Our quarterly and annual operating results and cash flows have fluctuated in the past and might continue to fluctuate, causing the value of our common stock to decline substantially.
Numerous factors, many of which are outside our control, may cause or contribute to significant fluctuations in our quarterly and annual operating results. These fluctuations may make financial planning and forecasting difficult. In addition, these fluctuations may result in unanticipated decreases in our available cash, which could negatively affect our business and prospects. In addition, one or more of such factors may cause our revenue or operating expenses in one period to be disproportionately higher or lower relative to the others. As a result, comparing our operating results on a period-to-period basis might not be meaningful. You should not rely on our past results as indicative of our future performance. Moreover, our stock price might be based on expectations of future performance that are unrealistic or that we might not meet and, if our revenue or operating results fall below the expectations of investors or securities analysts, the price of our common stock could decline substantially.
Our operating results have varied in the past. In addition to other risk factors listed in this section, some of the important factors that may cause fluctuations in our quarterly and annual operating results include:
- •
- adoption of our Simoa technology platform and products by customers;
- •
- the timing of customer orders to purchase our Simoa instruments;
- •
- the rate of utilization of consumables by our customers;
- •
- receipt and timing of revenue for services provided in our Simoa Accelerator Laboratory;
- •
- the timing of the introduction of new products, product enhancements and services; and
14
- •
- the receipt and timing of revenue from collaborations.
In addition, a significant portion of our operating expense is relatively fixed in nature, and planned expenditures are based in part on expectations regarding future revenue. Accordingly, unexpected revenue shortfalls might decrease our gross margins and could cause significant changes in our operating results from quarter to quarter. If this occurs, the trading price of our common stock could fall substantially.
We are an early, commercial-stage company and have a limited operating history, which may make it difficult to evaluate our current business and predict our future performance.
We are an early, commercial-stage company and have a limited commercial history. Our revenues are derived from sales of our instruments, consumables and services, which are all based on our Simoa technology, which we launched commercially in 2014. Our limited commercial history may make it difficult to evaluate our current business and makes predictions about our future success or viability subject to significant uncertainty. We will continue to encounter risks and difficulties frequently experienced by early, commercial-stage companies, including scaling up our infrastructure and headcount. If we do not address these risks successfully, our business will suffer.
If we are unable to maintain adequate revenue growth or do not successfully manage such growth, our business and growth prospects will be harmed.
We have experienced significant revenue growth in a short period of time. We may not achieve similar growth rates in future periods. Investors should not rely on our operating results for any prior periods as an indication of our future operating performance. To effectively manage our anticipated future growth, we must continue to maintain and enhance our financial, accounting, manufacturing, customer support and sales administration systems, processes and controls. Failure to effectively manage our anticipated growth could lead us to over-invest or under-invest in development, operational, and administrative infrastructure; result in weaknesses in our infrastructure, systems, or controls; give rise to operational mistakes, losses, loss of customers, productivity or business opportunities; and result in loss of employees and reduced productivity of remaining employees.
Our continued growth could require significant capital expenditures and might divert financial resources from other projects such as the development of new products and services. As additional products are commercialized, we may need to incorporate new equipment, implement new technology systems, or hire new personnel with different qualifications. Failure to manage this growth or transition could result in turnaround time delays, higher product costs, declining product quality, deteriorating customer service, and slower responses to competitive challenges. A failure in any one of these areas could make it difficult for us to meet market expectations for our products, and could damage our reputation and the prospects for our business.
If our management is unable to effectively manage our anticipated growth, our expenses may increase more than expected, our revenue could decline or grow more slowly than expected and we may be unable to implement our business strategy. The quality of our products and services may suffer, which could negatively affect our reputation and harm our ability to retain and attract customers.
Our future capital needs are uncertain and we may need to raise additional funds in the future.
We believe that the net proceeds from this offering, together with our cash generated from commercial sales and our existing cash and cash equivalents as of September 30, 2017, excluding any future available borrowings under our debt facility, will enable us to fund our operating expenses and capital expenditure
15
requirements for at least the next 24 months. However, we may need to raise substantial additional capital to:
- •
- expand our sales and marketing efforts to further commercialize our products;
- •
- expand our research and development efforts to improve our existing products and develop and launch new products, particularly if any of our
products are deemed by the United States Food and Drug Administration, or FDA, to be medical devices or otherwise subject to additional regulation by the FDA;
- •
- seek PMA approval or 510(k) clearance from the FDA for our existing products or new products if or when we decide to market products for use in
the prevention, diagnosis or treatment of a disease or other condition (see "Risk Factors—If the FDA determines that our products are medical devices or if we seek to market our products
for clinical diagnostic or health screening use, we will be required to obtain regulatory clearance(s) or approval(s), and may be required to cease or limit sales of our then marketed products, which
could materially and adversely affect our business, financial condition and results of operations. Any such regulatory process would be expensive, time-consuming and uncertain both in timing and in
outcome." and "Government regulation—501(k) clearance pathway;" "Government regulation—Premarket approval pathway" and "Government regulation—Clinical trials" for
further information about the FDA approvals that we may be required to seek and obtain in that circumstance);
- •
- lease a larger facility or build out our existing facility as we continue to grow our employee headcount;
- •
- hire additional personnel;
- •
- enter into collaboration arrangements, if any, or in-license other products and technologies;
- •
- add operational, financial and management information systems; and
- •
- incur increased costs as a result of operating as a public company.
Our future funding requirements will depend on many factors, including:
- •
- market acceptance of our products, including our Quanterix SR-X instrument that we expect to launch commercially in 2018;
- •
- the cost and timing of establishing additional sales, marketing and distribution capabilities;
- •
- the cost of our research and development activities;
- •
- the success of our existing collaborations and our ability to enter into additional collaborations in the future; and
- •
- the effect of competing technological and market developments.
We cannot assure you that we will be able to obtain additional funds on acceptable terms, or at all. If we raise additional funds by issuing equity or equity-linked securities, our stockholders may experience dilution. Future debt financing, if available, may involve covenants restricting our operations or our ability to incur additional debt. Any debt or equity financing may contain terms that are not favorable to us or our stockholders. If we raise additional funds through collaboration and licensing arrangements with third parties, it may be necessary to relinquish some rights to our technologies or our products, or grant licenses on terms that are not favorable to us. If we do not have, or are not able to obtain, sufficient funds, we may have to delay development or commercialization of our products. We also may have to reduce marketing, customer support or other resources devoted to our products or cease operations. Any
16
of these factors could have a material adverse effect on our financial condition, operating results and business.
Our ability to use net operating losses to offset future income may be subject to certain limitations.
As of December 31, 2016, we had federal net operating loss carry forwards, or NOLs, to offset future taxable income of approximately $87.9 million, which expire at various dates through 2035, if not utilized. A lack of future taxable income would adversely affect our ability to utilize these NOLs. In addition, under Section 382 of the Internal Revenue Code of 1986, as amended, or the Code, a corporation that undergoes an "ownership change" is subject to limitations on its ability to utilize its NOLs to offset future taxable income. We have already experienced one or more ownership changes as defined under Section 382 of the Code. Depending on the timing of any future utilization of our NOLs, we may be limited as to the amount that can be utilized each year as a result of such previous ownership changes. In addition, future changes in our stock ownership, including this or future offerings, as well as other changes that may be outside of our control, could result in additional ownership changes under Section 382 of the Code. Our NOLs may also be impaired under similar provisions of state law. We have recorded a full valuation allowance related to our NOLs and other deferred tax assets due to the uncertainty of the ultimate realization of the future benefits of those assets.
U.S. taxation of international business activities or the adoption of tax reform policies could materially impact our future financial position and results of operations.
Limitations on the ability of taxpayers to claim and utilize foreign tax credits and the deferral of certain tax deductions until earnings outside of the United States are repatriated to the United States, as well as changes to U.S. tax laws that may be enacted in the future, could impact the tax treatment of future foreign earnings. Should the scale of our international business activities expand, any changes in the U.S. taxation of such activities could increase our worldwide effective tax rate and harm our future financial position and results of operations.
Provisions of our secured term loan facility with Hercules Capital, Inc. may restrict our ability to pursue our business strategies. In addition, repayment of our outstanding debt and other obligations under our secured term loan facility with Hercules is subject to acceleration upon the occurrence of an event of default, which would have a material adverse effect on our business, financial condition and results of operations.
Our secured term loan facility with Hercules Capital, Inc., or Hercules, requires us, and any debt instruments we may enter into in the future may require us, to comply with various covenants that limit our ability to take on new indebtedness, to permit new liens, to pay dividends, to dispose of our property (including to license in certain situations), to engage in mergers or acquisitions and make certain other changes in our business. Debt instruments we may enter into in the future may also include financial covenants such as a requirement to maintain a specified minimum liquidity level or achieve a minimum annual revenue level. These restrictions could inhibit our ability to pursue our business strategies, including our ability to raise additional capital and make certain dispositions or investments without the consent of our lenders.
The obligations under our secured term loan facility with Hercules are subject to acceleration upon the occurrence of specified events of default, including our failure to make payments when due, our breach or default in the performance of our covenants and obligations under the facility following a cure period, bankruptcy and similar events, and the occurrence of a circumstance that would reasonably be expected to have a material adverse effect on (i) our business, operations, properties, assets or financial condition,
17
(ii) our ability to perform our obligations in accordance with the facility documents, (iii) the lender's ability to enforce any of its rights or remedies with respect to our obligations, or (iv) the collateral, the liens on the collateral or the first priority of the lender's liens. While we do not believe it is probable that the lender would accelerate the obligations under the facility, the definition of a material adverse effect is inherently subjective in nature, and we cannot assure that a material adverse effect will not occur or be deemed to have occurred by the lender.
If our products fail to achieve and sustain sufficient market acceptance, our revenue will be adversely affected.
Our success depends on our ability to develop and market products that are recognized and accepted as reliable, enabling and cost-effective. Most of the potential customers for our products already use expensive research systems in their laboratories that they have used for many years and may be reluctant to replace those systems with ours. Market acceptance of our Simoa technology will depend on many factors, including our ability to convince potential customers that our technology is an attractive alternative to existing technologies. Compared to some competing technologies, our Simoa technology is new and complex, and many potential customers have limited knowledge of, or experience with, our products. Prior to adopting our systems, some potential customers may need to devote time and effort to testing and validating our systems. Any failure of our systems to meet these customer benchmarks could result in potential customers choosing to retain their existing systems or to purchase systems other than ours. In addition, it is important that our Simoa technology be perceived as accurate and reliable by the scientific and medical research community as a whole. Historically, a significant part of our sales and marketing efforts has been directed at demonstrating the advantages of our technology to industry leaders and encouraging such leaders to publish or present the results of their evaluation of our system. If we are unable to continue to motivate leading researchers to use Simoa technology, or if such researchers are unable to achieve or unwilling to publish or present significant experimental results using our systems, acceptance and adoption of our systems will be slowed and our ability to increase our revenue would be adversely affected.
Our future success is dependent upon our ability to further penetrate our existing customer base and attract new customers.
Our current customer base is primarily composed of academic and governmental research institutions, as well as biopharmaceutical and contract research companies. Our success will depend upon our ability to respond to the evolving needs of, and increase our market share among, existing customers and additional potential customers, marketing new products as we develop them. Identifying, engaging and marketing to customers who are unfamiliar with our current products requires substantial time, expertise and expense and involves a number of risks, including:
- •
- our ability to attract, retain and manage the sales, marketing and service personnel necessary to expand market acceptance for our Simoa
technology;
- •
- the time and cost of maintaining and growing a specialized sales, marketing and service force; and
- •
- our sales, marketing and service force may be unable to execute successful commercial activities.
We have utilized third parties to assist with sales, distribution and customer support in certain regions of the world. There is no guarantee, when we enter into such arrangements, that we will be successful in attracting desirable sales and distribution partners. There is also no guarantee that we will be able to
18
enter into such arrangements on favorable terms. Any failure of our sales and marketing efforts, or those of any third-party sales and distribution partners, would adversely affect our business.
Some of the reagents used in our products are labeled for "research use only" and will have to undergo additional testing before we could use them in a product intended for clinical use.
Some of the materials that are used in our consumable products, including certain reagents, are purchased from suppliers with a restriction that they be used for research use only, or RUO. While we have focused initially on the life sciences research market, part of our business strategy is to expand our product line, either alone or in collaboration with third parties, to encompass systems and products that can be used for clinical purposes. Whether or not we continue to use the same RUO materials that we currently use, or obtain similar materials that are not labeled with the RUO restriction, we will be required to demonstrate that the use of our system and products as a clinical test complies with all applicable requirements. In addition, if we were to change the supplier of any material or component used in a clinical test, we would be required to confirm through additional testing that the change does not adversely affect the reliability of the test. Any such additional testing may be expensive and time-consuming and delay our introduction of new products and systems.
In the near term, our business will depend on levels of research and development spending by academic and governmental research institutions and biopharmaceutical companies, a reduction in which could limit demand for our products and adversely affect our business and operating results.
In the near term, we expect that our revenue will be derived primarily from sales of our instruments and consumables to academic and governmental research institutions, as well as biopharmaceutical and contract research companies worldwide for research applications. The demand for our products will depend in part upon the research and development budgets of these customers, which are impacted by factors beyond our control, such as:
- •
- changes in government programs that provide funding to research institutions and companies;
- •
- macroeconomic conditions and the political climate;
- •
- changes in the regulatory environment;
- •
- differences in budgetary cycles; and
- •
- market acceptance of relatively new technologies, such as ours.
For example, in March 2017, the federal government announced the intent to cut federal biomedical research funding by as much as 18%. While there has been significant opposition to these funding cuts, the uncertainty regarding the availability of research funding for potential customers may adversely affect our operating results. Our operating results may fluctuate substantially due to reductions and delays in research and development expenditures by these customers. Any decrease in customers' budgets or expenditures, or in the size, scope or frequency of capital or operating expenditures, could materially and adversely affect our business, operating results and financial condition.
The sales cycle for our Simoa instruments can be lengthy and variable, which makes it difficult for us to forecast revenue and other operating results.
The sales process for our Simoa instruments generally involves numerous interactions with multiple individuals within an organization, and often includes in-depth analysis by potential customers of our technology and products and a lengthy review process. Our customers' evaluation processes often involve a number of factors, many of which are beyond our control. As a result of these factors, the capital
19
investment required to purchase our systems and the budget cycles of our customers, the time from initial contact with a customer to our receipt of a purchase order can vary significantly. Given the length and uncertainty of our sales cycle, we have in the past experienced, and expect to in the future experience, fluctuations in our sales on a period-to-period basis. In addition, any failure to meet customer expectations could result in customers choosing to retain their existing systems, use existing assays not requiring capital equipment or purchase systems other than ours.
Our long-term results depend upon our ability to improve existing products and introduce and market new products successfully.
Our business is dependent on the continued improvement of our existing Simoa products and our development of new products utilizing our Simoa or other potential future technology. As we introduce new products or refine, improve or upgrade versions of existing products, we cannot predict the level of market acceptance or the amount of market share these products will achieve, if any. We cannot assure you that we will not experience material delays in the introduction of new products in the future. In addition, introducing new products could result in a decrease in revenues from our existing products. For example, introduction of the Quanterix SR-X may result in a decrease in revenue from our existing Simoa HD-1 Analyzer instrument. Consistent with our strategy of offering new products and product refinements, we expect to continue to use a substantial amount of capital for product development and refinement. We may need additional capital for product development and refinement than is available on terms favorable to us, if at all, which could adversely affect our business, financial condition or results of operations.
We generally sell our products in industries that are characterized by rapid technological changes, frequent new product introductions and changing industry standards. If we do not develop new products and product enhancements based on technological innovation on a timely basis, our products may become obsolete over time and our revenues, cash flow, profitability and competitive position will suffer. Our success will depend on several factors, including our ability to:
- •
- correctly identify customer needs and preferences and predict future needs and preferences;
- •
- allocate our research and development funding to products with higher growth prospects;
- •
- anticipate and respond to our competitors' development of new products and technological innovations;
- •
- innovate and develop new technologies and applications, and acquire or obtain rights to third-party technologies that may have valuable
applications in the markets we serve;
- •
- successfully commercialize new technologies in a timely manner, price them competitively and manufacture and deliver sufficient volumes of new
products of appropriate quality on time; and
- •
- convince customers to adopt new technologies.
In addition, if we fail to accurately predict future customer needs and preferences or fail to produce viable technologies, we may invest heavily in research and development of products that do not lead to significant revenue. Even if we successfully innovate and develop new products and product enhancements, we may incur substantial costs in doing so, and our profitability may suffer.
Our ability to develop new products based on innovation can affect our competitive position and often requires the investment of significant resources. Difficulties or delays in research, development or production of new products and services or failure to gain market acceptance of new products and technologies may reduce future revenues and adversely affect our competitive position.
20
If we do not successfully develop and introduce new assays for our technology, we may not generate new sources of revenue and may not be able to successfully implement our growth strategy.
Our business strategy includes the development of new assays for our Simoa instruments. New assays require significant research and development and a commitment of significant resources prior to their commercialization. Our technology is complex, and we cannot be sure that any assays we may intend to develop will be developed successfully, be proven to be effective, offer improvements over currently available tests, meet applicable standards, be produced in commercial quantities at acceptable costs or be successfully marketed. Moreover, development of particular assays may require licenses or access to third party intellectual property which may not be available on commercially reasonable terms, or at all. In addition, we believe that our future success will depend, in part, on our ability to develop and commercialize multiplex assays that can simultaneously measure multiple biomarkers. The most robust multiplex assay that we have commercially launched to date is a 4-plex assay. If we do not successfully develop new assays for our Simoa instruments, including multiplex assays with the ability to detect an increased number of biomarkers in a single sample, we could lose revenue opportunities with existing or future customers.
If we do not successfully manage the development and launch of new products, our financial results could be adversely affected.
We expect to launch our Quanterix SR-X instrument in 2018. We face risks associated with launching new products such as the Quanterix SR-X. If we encounter development or manufacturing challenges or discover errors during our product development cycle, the product launch dates of new products may be delayed. The expenses or losses associated with unsuccessful product development or launch activities or lack of market acceptance of our new products could adversely affect our business or financial condition.
Undetected errors or defects in our products could harm our reputation, decrease market acceptance of our products or expose us to product liability claims.
Our Simoa products may contain undetected errors or defects when first introduced or as new versions or new products are released. Disruptions affecting the introduction or release of, or other performance problems with, our products may damage our customers' businesses and could harm their and our reputation. If that occurs, we may incur significant costs, the attention of our key personnel could be diverted, or other significant customer relations problems may arise. We may also be subject to warranty and liability claims for damages related to errors or defects in our products. In addition, if we do not meet industry or quality standards, if applicable, our products may be subject to recall. A material liability claim, recall or other occurrence that harms our reputation or decreases market acceptance of our products could harm our business and operating results.
Although we do not, and cannot currently, promote the use of our products, or services based on our products, for diagnostic purposes, if our customers develop or use them for diagnostic purposes, someone could file a product liability claim alleging that one of our products contained a design or manufacturing defect that resulted in the failure to adequately perform, leading to death or injury. A product liability claim could result in substantial damages and be costly and time consuming to defend, either of which could materially harm our business or financial condition. We cannot assure investors that our product liability insurance would adequately protect our assets from the financial impact of defending a product liability claim. Any product liability claim brought against us, with or without merit, could increase our product liability insurance rates or prevent us from securing insurance coverage in the future.
21
We depend on strategic collaborations and licensing arrangements with third parties to develop in vitro diagnostic products. We may not be successful in establishing or maintaining additional collaborations or license agreements.
We have established strategic collaborations and licensing agreements with third parties to develop products based on our Simoa technology, such as for certain in vitro diagnostic purposes. For example, we have entered into a license agreement with bioMérieux SA, pursuant to which we have granted them an exclusive license to, among other things, develop and sell certain in vitro diagnostic products used in clinical lab applications based on our Simoa technology and a co-exclusive license for certain other in vitro diagnostic products. If bioMérieux or any other partners do not prioritize and commit sufficient resources to develop and sell products based on our Simoa technology, our ability to generate revenue from sales in respect of in vitro diagnostic products may be limited.
We may seek to enter into additional such arrangements; however, there is no assurance that we will be successful in doing so. Moreover, given the exclusive nature of a portion of the license rights granted to bioMérieux, our ability to collaborate with others in the areas of in vitro diagnostics used in clinical lab applications, food quality control testing, and pharmaceutical quality control testing will be limited, in that we may not establish collaborations with others covering these areas while the exclusive license to bioMérieux remains in effect, subject to our right to make and sell the current version of the Simoa HD-1 Analyzer for use in clinical lab applications, either directly or through a partner (but not both). Establishing collaborations and licensing arrangements is difficult and time-consuming. Discussions may not lead to collaborations or licenses on favorable terms, if at all. Even if we establish new relationships, they may never result in the successful development or commercialization of products based on our Simoa technology.
Our reliance on distributors for sales of our products outside of the United States could limit or prevent us from selling our products and could impact our revenue.
We have established exclusive distribution agreements for our Simoa HD-1 Analyzer and related consumable products within Australia, China, India, Japan, Lebanon, Singapore and South Korea. We intend to continue to grow our business internationally, and to do so we must attract additional distributors and retain existing distributors to maximize the commercial opportunity for our products. There is no guarantee that we will be successful in attracting or retaining desirable sales and distribution partners or that we will be able to enter into such arrangements on favorable terms. Distributors may not commit the necessary resources to market and sell our products to the level of our expectations or may choose to favor marketing the products of our competitors. If current or future distributors do not perform adequately, or we are unable to enter into effective arrangements with distributors in particular geographic areas, we may not realize long-term international revenue growth. In addition, if our distributors fail to comply with applicable laws and ethical standards, including anti-bribery laws, this could damage our reputation and could have a significant adverse effect on our business and our revenues.
We expect to generate a substantial portion of our revenue internationally in the future and can become further subject to various risks relating to our international activities, which could adversely affect our business, operating results and financial condition.
During 2016 and for the nine months ended September 30, 2017, approximately 36% and 46%, respectively, of our product revenue was generated from customers located outside of North America. We believe that a substantial percentage of our future revenue will come from international sources as we expand our overseas operations and develop opportunities in additional areas. We have limited experience
22
operating internationally and engaging in international business involves a number of difficulties and risks, including:
- •
- required compliance with existing and changing foreign regulatory requirements and laws;
- •
- difficulties and costs of staffing and managing foreign operations;
- •
- difficulties protecting or procuring intellectual property rights;
- •
- required compliance with anti-bribery laws, such as the U.S. Foreign Corrupt Practices Act, data privacy requirements, labor laws and
anti-competition regulations;
- •
- export or import restrictions;
- •
- laws and business practices favoring local companies;
- •
- longer payment cycles and difficulties in enforcing agreements and collecting receivables through certain foreign legal systems;
- •
- political and economic instability; and
- •
- potentially adverse tax consequences, tariffs, customs charges, bureaucratic requirements and other trade barriers.
Historically, most of our revenue has been denominated in U.S. dollars. In the future, we may sell our products and services in local currency outside of the United States. As our operations in countries outside of the United States grow, our results of operations and cash flows may be subject to fluctuations due to changes in foreign currency exchange rates, which could harm our business in the future. For example, if the value of the U.S. dollar increases relative to foreign currencies, in the absence of a corresponding change in local currency prices, our revenue could be adversely affected as we convert revenue from local currencies to U.S. dollars. If we dedicate significant resources to our international operations and are unable to manage these risks effectively, our business, operating results and financial condition will suffer.
We could be adversely affected by violations of the U.S. Foreign Corrupt Practices Act and other worldwide anti-bribery laws by us or our agents.
We are subject to the U.S. Foreign Corrupt Practices Act, or FCPA, which prohibits companies and their intermediaries from making payments in violation of law to non-U.S. government officials for the purpose of obtaining or retaining business or securing any other improper advantage. Our reliance on independent distributors to sell our products internationally demands a high degree of vigilance in maintaining our policy against participation in corrupt activity, because these distributors could be deemed to be our agents, and we could be held responsible for their actions. Other U.S. companies in the medical device and pharmaceutical fields have faced criminal penalties under the FCPA for allowing their agents to deviate from appropriate practices in doing business with these individuals. We are also subject to similar antibribery laws in the jurisdictions in which we operate, including the United Kingdom's Bribery Act of 2010, which also prohibits commercial bribery and makes it a crime for companies to fail to prevent bribery. We have limited experience in complying with these laws and in developing procedures to monitor compliance with these laws by our agents. These laws are complex and far-reaching in nature, and, as a result, we cannot assure you that we would not be required in the future to alter one or more of our practices to be in compliance with these laws or any changes in these laws or the interpretation thereof. Any violations of these laws, or allegations of such violations, could disrupt our operations, involve significant management distraction, involve significant costs and expenses, including legal fees, and could
23
result in a material adverse effect on our business, prospects, financial condition, or results of operations. We could also incur severe penalties, including criminal and civil penalties, disgorgement, and other remedial measures.
If we are unable to recruit, train, retain, motivate and integrate key personnel, we may not achieve our goals.
Our future success depends on our ability to recruit, train, retain, motivate and integrate key personnel, including our recently expanded senior management team, as well as our research and development, manufacturing and sales and marketing personnel. Competition for qualified personnel is intense. Our growth depends, in particular, on attracting and retaining highly-trained sales personnel with the necessary scientific background and ability to understand our systems at a technical level to effectively identify and sell to potential new customers and develop new products. Because of the complex and technical nature of our products and the dynamic market in which we compete, any failure to attract, train, retain, motivate and integrate qualified personnel could materially harm our operating results and growth prospects.
We have limited experience in marketing and selling our products, and if we are unable to successfully commercialize our products, our business and operating results will be adversely affected.
We have limited experience marketing and selling our products. We currently sell all our products for research use only, through our direct field sales and support organizations located in North America and Europe and through a combination of our own sales force and third-party distributors in additional major markets such as Australia, China, India, Japan, Lebanon, Singapore and South Korea.
The future sales of our products will depend in large part on our ability to effectively market and sell our products, successfully manage and expand our sales force, and increase the scope of our marketing efforts. We may also enter into additional distribution arrangements in the future. Because we have limited experience in marketing and selling our products, our ability to forecast demand, the infrastructure required to support such demand and the sales cycle to customers is unproven. If we do not build an efficient and effective sales force, our business and operating results will be adversely affected.
We rely on a single contract manufacturer to manufacture and supply our Simoa HD-1 Analyzer and will rely on a different single contract manufacturer to manufacture and supply our Quanterix SR-X. If either of these manufacturers should fail or not perform satisfactorily, our ability to supply these instruments would be negatively and adversely affected.
We currently rely on a single contract manufacturer, STRATEC Biomedical AG, or STRATEC, an analytical and diagnostic systems manufacturer located in Germany, to manufacture and supply all of our Simoa HD-1 Analyzer instruments. See "Business–Key agreements–Development agreement and supply agreement with STRATEC." In addition, we will rely on a single contract manufacturer, Paramit Corporation, or Paramit, a contract manufacturer located in California, to manufacture and supply all of our Quanterix SR-X instruments. Since our contract with STRATEC does not commit them to supply quantities beyond the amounts included in our forecasts and our contract with Paramit does not commit them to carry inventory or make available any particular quantities, these contract manufacturers may give other customers' needs higher priority than ours, and we may not be able to obtain adequate supplies in a timely manner or on commercially reasonable terms. If either of these manufacturers were to be unable to supply instruments, our business would be harmed.
Pursuant to our Supply Agreement with STRATEC, as amended, we are required to purchase a minimum number of commercial units of our Simoa HD-1 Analyzer over a seven-year period ending in May 2021. If we fail to purchase a required minimum number of commercial units, including as a result of the impact of
24
sales of the Quanterix SR-X going forward, we would be obligated to pay a fee based on the shortfall of commercial units purchased compared to the required number. If we fail to purchase a required minimum number of commercial instruments and terminate the arrangement in certain circumstances, we would be obligated to issue a warrant to purchase shares of our Series A-3 preferred stock. Any amount we may have to pay STRATEC for failing to purchase the minimum number of commercial units of our Simoa HD-1 Analyzer will cause our operating results to suffer.
In the event it becomes necessary to utilize a different contract manufacturer for either the Simoa HD-1 Analyzer or the Quanterix SR-X, we would experience additional costs, delays and difficulties in doing so as a result of identifying and entering into an agreement with a new supplier as well as preparing such new supplier to meet the logistical requirements associated with manufacturing our units, and our business would suffer. We may also experience additional costs and delays in the event we need access to or rights under any intellectual property of STRATEC.
In addition, certain of the components used in our instruments are sourced from limited or sole suppliers. If we were to lose such suppliers, there can be no assurance that we will be able to identify or enter into agreements with alternative suppliers on a timely basis on acceptable terms, if at all. An interruption in our ability to sell and deliver instruments to customers could occur if we encounter delays or difficulties in securing these components, or if the quality of the components supplied do not meet specifications, or if we cannot then obtain an acceptable substitute. If any of these events occur, our business and operating results could be harmed.
We may experience manufacturing problems or delays that could limit the growth of our revenue or increase our losses.
We may encounter unforeseen situations that would result in delays or shortfalls in our production as well as delays or shortfalls caused by our outsourced manufacturing suppliers and by other third-party suppliers who manufacture components for our products. If we are unable to keep up with demand for our products, our revenue could be impaired, market acceptance for our products could be adversely affected and our customers might instead purchase our competitors' products. Our inability to successfully manufacture our products would have a material adverse effect on our operating results.
We rely on a limited number of suppliers or, in some cases, one supplier, for some of our materials and components used in our consumable products, and may not be able to find replacements or immediately transition to alternative suppliers, which could have a material adverse effect on our business, financial condition, results of operations and reputation.
We rely on limited or sole suppliers for certain reagents and other materials and components that are used in our consumable products. While we periodically forecast our needs for such materials and enter into standard purchase orders with them, we do not have long-term contracts with many of these suppliers. If we were to lose such suppliers, there can be no assurance that we will be able to identify or enter into agreements with alternative suppliers on a timely basis on acceptable terms, if at all. An interruption in our operations could occur if we encounter delays or difficulties in securing these materials, or if the quality of the materials supplied do not meet our requirements, or if we cannot then obtain an acceptable substitute. The time and effort required to qualify a new supplier and ensure that the new materials provide the same or better quality results could result in significant additional costs. Any such interruption could significantly affect our business, financial condition, results of operations and reputation.
25
If we cannot provide quality technical and applications support, we could lose customers and our business and prospects will suffer.
The placement of our products at new customer sites, the introduction of our technology into our customers' existing laboratory workflows and ongoing customer support can be complex. Accordingly, we need highly trained technical support personnel. Hiring technical support personnel is very competitive in our industry due to the limited number of people available with the necessary scientific and technical backgrounds and ability to understand our Simoa technology at a technical level. To effectively support potential new customers and the expanding needs of current customers, we will need to substantially expand our technical support staff. If we are unable to attract, train or retain the number of highly qualified technical services personnel that our business needs, our business and prospects will suffer.
The life sciences research and diagnostic markets are highly competitive. If we fail to effectively compete, our business, financial condition and operating results will suffer.
We face significant competition in the life sciences research and diagnostic markets. We currently compete with both established and early stage companies that design, manufacture and market systems and consumable supplies. We believe our principal competitors in the life sciences research and diagnostic markets include Bio-Techne, Luminex Corporation, MesoScale Diagnostics, Singulex, Gyros Corporation and Nanostring Technologies, Inc. As we expand the applications for our products to include health screening, we expect to compete with companies such as Siemens, Abbott, Roche, Ortho Clinical Diagnostics and Thermo Fisher Scientific. In addition, there are a number of new market entrants in the process of developing novel technologies for the life sciences research, diagnostic and screening markets.
Many of our current competitors are either publicly traded, or are divisions of publicly-traded companies, and may enjoy a number of competitive advantages over us, including:
- •
- greater name and brand recognition;
- •
- substantially greater financial and human resources;
- •
- broader product lines;
- •
- larger sales forces and more established distributor networks;
- •
- substantial intellectual property portfolios;
- •
- larger and more established customer bases and relationships; and
- •
- better established, larger scale, and lower cost manufacturing capabilities.
We believe that the principal competitive factors in all of our target markets include:
- •
- cost of instruments and consumables;
- •
- accuracy, including sensitivity and specificity, and reproducibility of results;
- •
- reputation among customers;
- •
- innovation in product offerings;
- •
- flexibility and ease of use; and
- •
- compatibility with existing laboratory processes, tools and methods.
26
We cannot assure investors that our products will compete favorably or that we will be successful in the face of increasing competition from new products and technologies introduced by our existing competitors or new companies entering our markets. In addition, we cannot assure investors that our competitors do not have or will not develop products or technologies that currently or in the future will enable them to produce competitive products with greater capabilities or at lower costs than ours. Any failure to compete effectively could materially and adversely affect our business, financial condition and operating results.
Acquisitions or joint ventures could disrupt our business, cause dilution to our stockholders and otherwise harm our business.
We may acquire other businesses, products or technologies as well as pursue strategic alliances, joint ventures, technology licenses or investments in complementary businesses. We have not made any acquisitions to date, and our ability to do so successfully is unproven. Any of these transactions could be material to our financial condition and operating results and expose us to many risks, including:
- •
- disruption in our relationships with customers, distributors or suppliers as a result of such a transaction;
- •
- unanticipated liabilities related to acquired companies;
- •
- difficulties integrating acquired personnel, technologies and operations into our existing business;
- •
- diversion of management time and focus from operating our business to acquisition integration challenges;
- •
- increases in our expenses and reductions in our cash available for operations and other uses; and
- •
- possible write-offs or impairment charges relating to acquired businesses.
Foreign acquisitions involve unique risks in addition to those mentioned above, including those related to integration of operations across different cultures and languages, currency risks and the particular economic, political and regulatory risks associated with specific countries.
Also, the anticipated benefit of any acquisition may not materialize. Future acquisitions or dispositions could result in potentially dilutive issuances of our equity securities, the incurrence of debt, contingent liabilities or amortization expenses or write-offs of goodwill, any of which could harm our financial condition. We cannot predict the number, timing or size of future joint ventures or acquisitions, or the effect that any such transactions might have on our operating results.
Risks related to government regulation and diagnostic product reimbursement
If the FDA determines that our products are medical devices or if we seek to market our products for clinical diagnostic or health screening use, we will be required to obtain regulatory clearance(s) or approval(s), and may be required to cease or limit sales of our then marketed products, which could materially and adversely affect our business, financial condition and results of operations. Any such regulatory process would be expensive, time-consuming and uncertain both in timing and in outcome.
We have focused initially on the life sciences research market. This includes laboratories associated with academic and governmental research institutions, as well as pharmaceutical, biotechnology and contract research companies. Accordingly, our products are labeled as "Research Use Only," or RUO, and are not intended for diagnostic use. While we have focused initially on the life sciences research market and RUO products only, our strategy is to expand our product line to encompass products that are intended to be used for the diagnosis of disease, either alone or in collaboration with third parties (such as our collaboration with bioMérieux). Such IVD products will be subject to regulation by the FDA as medical
27
devices, or comparable international agencies, including requirements for regulatory clearance or approval of such products before they can be marketed. If the FDA were to determine that our products are intended for clinical use or if we decided to market our products for such use, we would be required to obtain FDA 510(k) clearance or premarket approval in order to sell our products in a manner consistent with FDA laws and regulations. Such regulatory approval processes or clearances are expensive, time-consuming and uncertain; our efforts may never result in approved premarket approval application, or PMA, or 510(k) clearance for our products; and failure by us or a collaborator to obtain or comply with such approvals and clearances could have an adverse effect on our business, financial condition or operating results.
IVD products may be regulated as medical devices by the FDA and comparable international agencies and may require either clearance from the FDA following the 510(k) pre-market notification process or PMA from the FDA, in each case prior to marketing. If we or our collaborators are required to obtain a PMA or 510(k) clearance for products based on our technology, we or they would be subject to a substantial number of additional requirements for medical devices, including establishment registration, device listing, Quality Systems Regulations, or QSRs, which cover the design, testing, production, control, quality assurance, labeling, packaging, servicing, sterilization (if required), and storage and shipping of medical devices (among other activities), product labeling, advertising, recordkeeping, post-market surveillance, post-approval studies, adverse event reporting, and correction and removal (recall) regulations. One or more of the products we or a collaborator may develop using our technology may also require clinical trials in order to generate the data required for PMA approval. Complying with these requirements may be time-consuming and expensive. We or our collaborators may be required to expend significant resources to ensure ongoing compliance with the FDA regulations and/or take satisfactory corrective action in response to enforcement action, which may have a material adverse effect on the ability to design, develop, and commercialize products using our technology as planned. Failure to comply with these requirements may subject us or a collaborator to a range of enforcement actions, such as warning letters, injunctions, civil monetary penalties, criminal prosecution, recall and/or seizure of products, and revocation of marketing authorization, as well as significant adverse publicity. If we or our collaborators fail to obtain, or experience significant delays in obtaining, regulatory approvals for IVD products, such products may not be able to be launched or successfully commercialized in a timely manner, or at all.
Laboratory developed tests, or LDTs, are a subset of IVD tests that are designed, manufactured and used within a single laboratory. The FDA maintains that LDTs are medical devices and has for the most part exercised enforcement discretion for most LDTs. A significant change in the way that the FDA regulates any LDTs that we, our collaborators or our customers develop using our technology could affect our business. The FDA has considered the appropriate way to regulate such tests, but after publishing several draft guidances and holding a number of public hearings and workshops, no final guidance has been issued. However, if the FDA requires laboratories to undergo premarket review and comply with other applicable FDA requirements in the future, the cost and time required to commercialize an LDT will increase substantially, and may reduce the financial incentive for laboratories to develop LDTs, which could reduce demand for our instruments and our other products.
Failure to comply with applicable FDA requirements could subject us to misbranding or adulteration allegations under the Federal Food, Drug, and Cosmetic Act. We could be subject to a range of enforcement actions, including warning letters, injunctions, civil monetary penalties, criminal prosecution, and recall and/or seizure of products, as well as significant adverse publicity. In addition, changes to the current regulatory framework, including the imposition of additional or new regulations, could arise at any
28
time during the development or marketing of our products, which may negatively affect our ability to obtain or maintain FDA or comparable regulatory approval of our products, if required.
Foreign jurisdictions have laws and regulations similar to those described above, which may adversely affect our ability to market our products as planned in such countries. The number and scope of these requirements are increasing. As in the United States, the cost and time required to comply with regulatory requirements may be substantial, and there is no guarantee that we will obtain the necessary authorization(s) required to make our products commercially viable. As a result, the imposition of foreign requirements may also have a material adverse effect on the commercial viability of our operations.
We expect to rely on third parties in conducting any required future studies of diagnostic products that may be required by the FDA or other regulatory authorities, and those third parties may not perform satisfactorily.
We do not have the ability to independently conduct clinical trials or other studies that may be required to obtain FDA and other regulatory clearance or approval for future diagnostic products. Accordingly, we expect that we would rely on third parties, such as clinical investigators, consultants, and collaborators to conduct such studies if needed. Our reliance on these third parties for clinical and other development activities would reduce our control over these activities. If these third parties do not successfully carry out their contractual duties or regulatory obligations or meet expected deadlines, if the third parties need to be replaced or if the quality or accuracy of the data they obtain is compromised, we may not be able to obtain regulatory clearance or approval.
If diagnostic procedures that are enabled by our technology are subject to unfavorable pricing regulations or third-party coverage and reimbursement policies, our business could be harmed.
The ability of our customers to commercialize diagnostic tests based on our technology will depend in part on the extent to which coverage and reimbursement for these test will be available from government health programs, private health insurers and other third-party payors. In the United States, the principal decisions about reimbursement for new technologies are often made by the Centers for Medicare and Medicaid Services, or CMS. Private payors often follow CMS to a substantial degree. It is difficult to predict what CMS will decide with respect to reimbursement. A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and third-party payors have attempted to control costs by limiting coverage and the amount of payments for particular products and procedures. We cannot be sure that coverage will be available for any diagnostic tests based on our technology, and, if coverage is available, the level of payments. Reimbursement may impact the demand for those tests. If reimbursement is not available or is available only to limited levels, our customers may not be able to successfully commercialize any tests for which they receive marketing authorization.
Current and future legislation may increase the difficulty and cost to obtain marketing approval of and commercialize any products based on our technology and affect the prices that may be obtained.
In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, collectively, the ACA, became law. The ACA is a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for the healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. The current administration supports a repeal of the ACA and an Executive Order has been signed commanding federal agencies to try to waive or delay requirements of the ACA that impose economic or regulatory burdens on states, families, the health-care industry and others. The Executive
29
Order also declares that the administration will seek the "prompt repeal" of the law and that the government should prepare to "afford the States more flexibility and control to create a more free and open healthcare market." In addition, following the passage of the budget resolution for fiscal year 2017, the U.S. House of Representatives passed legislation known as the American Health Care Act, which, if enacted, would have amended or repealed significant portions of the ACA. The U.S. Senate could adopt the American Health Care Act as passed by the U.S. House of Representatives or other legislation to amend or replace elements of the ACA. It is uncertain whether the American Health Care Act will become law. At this time, the immediate impact of the Executive Order is not clear, and we cannot know how any legislation that may be passed to amend or replace the ACA will impact our business in the United States.
In addition, other legislative changes have been proposed and adopted since the ACA was enacted. We expect that the ACA, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we or our collaborators will receive for any cleared or approved product. Any reduction in payments from Medicare or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize any of our products for which we receive marketing approval.
In addition, sales of our tests outside of the United States will subject us to foreign regulatory requirements, which may also change over time.
We cannot predict whether future healthcare initiatives will be implemented at the federal or state level or in countries outside of the United States in which we may do business, or the effect any future legislation or regulation will have on us. The expansion in government's effect on the United States healthcare industry may result in decreased profits to us, lower reimbursements by payors for our products or reduced medical procedure volumes, all of which may adversely affect our business, financial condition and results of operations.
Risks related to our operations
We depend on our information technology systems, and any failure of these systems could harm our business.
We depend on information technology and telecommunications systems to operate our business. We have installed, and expect to expand, a number of enterprise software systems that affect a broad range of business processes and functional areas, including, for example, systems handling human resources, accounting, manufacturing, inventory control, financial controls and reporting, sales administration, and other infrastructure operations. In addition to the aforementioned business systems, we intend to extend the capabilities of both our preventative and detective security controls by augmenting the monitoring and alerting functions, network design, and automatic countermeasure operations of our technical systems. These information technology and telecommunications systems support a variety of functions, including manufacturing operations, quality control, customer service support, and general administrative activities.
30
Information technology and telecommunications systems are vulnerable to damage from a variety of sources, including telecommunications or network failures, malicious human acts and natural disasters. Moreover, despite network security and back-up measures, some of our servers are potentially vulnerable to physical or electronic break-ins, computer viruses, and similar disruptive problems. Despite the precautionary measures we have taken to prevent unanticipated problems that could affect our information technology and telecommunications systems, failures or significant downtime of our information technology or telecommunications systems or those used by our third-party suppliers could prevent us from operating our business and managing the administrative aspects of our business. Any disruption or loss of information technology or telecommunications systems on which critical aspects of our operations depend could have an adverse effect on our business.
Security breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation.
In the ordinary course of our business, we collect and store sensitive data, intellectual property and proprietary business information owned or controlled by ourselves or our customers. This data encompasses a wide variety of business-critical information including research and development information, commercial information, and business and financial information. We face four primary risks relative to protecting this critical information: loss of access; inappropriate disclosure; inappropriate modification; and inadequate monitoring of our controls over the first three risks.
The secure processing, storage, maintenance, and transmission of this critical information is vital to our operations and business strategy, and we devote significant resources to protecting such information. Although we take measures to protect sensitive information from unauthorized access or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers or viruses or breached due to employee error, malfeasance, or other disruptions. Any such breach or interruption could compromise our networks and the information stored there could be accessed by unauthorized parties, publicly disclosed, lost, or stolen. Any such access, disclosure or other loss of information could adversely affect our reputation and our business.
We face risks related to handling of hazardous materials and other regulations governing environmental safety.
Our operations are subject to complex and stringent environmental, health, safety and other governmental laws and regulations that both public officials and private individuals may seek to enforce. Our activities that are subject to these regulations include, among other things, our use of hazardous materials and the generation, transportation and storage of waste. Although we have secured clearance from the EPA historically, and currently are operating in compliance with applicable EPA rules and regulations, our business could be adversely affected if we discover that we or an acquired business is not in material compliance with these rules and regulations. In the future, we may pursue the use of other surfactant substances that will require clearance from the EPA, and we may fail to obtain such clearance. Existing laws and regulations may also be revised or reinterpreted, or new laws and regulations may become applicable to us, whether retroactively or prospectively, that may have a negative effect on our business and results of operations. It is also impossible to eliminate completely the risk of accidental environmental contamination or injury to individuals. In such an event, we could be liable for any damages that result, which could adversely affect our business.
31
Risks related to intellectual property
If we are unable to protect our intellectual property, it may reduce our ability to maintain any technological or competitive advantage over our competitors and potential competitors, and our business may be harmed.
We rely on patent protection as well as trademark, copyright, trade secret and other intellectual property rights protection and contractual restrictions to protect our proprietary technologies, all of which provide limited protection and may not adequately protect our rights or permit us to gain or keep any competitive advantage. As of September 30, 2017, we owned or exclusively licensed 16 granted U.S. patents and approximately 14 pending U.S. patent applications. We also owned or exclusively licensed approximately 31 pending patent applications and granted patents in particular jurisdictions outside of the United States. If we fail to protect our intellectual property, third parties may be able to compete more effectively against us, we may lose our technological or competitive advantage, or we may incur substantial litigation costs in our attempts to recover or restrict use of our intellectual property.
We cannot assure investors that any of our currently pending or future patent applications will result in granted patents, and we cannot predict how long it will take for such patents to be granted. It is possible that, for any of our patents that have granted or that may grant in the future, others will design around our patented technologies. Further, we cannot assure investors that other parties will not challenge any patents granted to us or that courts or regulatory agencies will hold our patents to be valid or enforceable. We cannot guarantee investors that we will be successful in defending challenges made against our patents and patent applications. Any successful third-party challenge to our patents could result in the unenforceability or invalidity of such patents, or to such patents being interpreted narrowly or otherwise in a manner adverse to our interests. Our ability to establish or maintain a technological or competitive advantage over our competitors may be diminished because of these uncertainties. For these and other reasons, our intellectual property may not provide us with any competitive advantage. For example:
- •
- We or our licensors might not have been the first to make the inventions covered by each of our pending patent applications or granted patents;
- •
- We or our licensors might not have been the first to file patent applications for these inventions. To determine the priority of these
inventions, we may have to participate in interference proceedings or derivation proceedings declared by the United States Patent and Trademark Office, or USPTO, that could result in substantial cost
to us. No assurance can be given that our patent applications or granted patents (or those of our licensors) will have priority over any other patent or patent application involved in such a
proceeding;
- •
- Others may independently develop similar or alternative products and technologies or duplicate any of our products and technologies;
- •
- It is possible that our owned or licensed pending patent applications will not result in granted patents, and even if such pending patent
applications grant as patents, they may not provide a basis for intellectual property protection of commercially viable products, may not provide us with any competitive advantages, or may be
challenged and invalidated by third parties;
- •
- We may not develop additional proprietary products and technologies that are patentable;
- •
- The patents of others may have an adverse effect on our business; and
32
- •
- We apply for patents covering our products and technologies and uses thereof, as we deem appropriate. However, we may fail to apply for patents on important products and technologies in a timely fashion or at all, or we may fail to apply for patents in potentially relevant jurisdictions.
To the extent our intellectual property offers inadequate protection, or is found to be invalid or unenforceable, we would be exposed to a greater risk of direct competition. If our intellectual property does not provide adequate coverage of our competitors' products, our competitive position could be adversely affected, as could our business.
Software is a critical component of our instruments. To the extent such software is not protected by our patents, we depend on trade secret protection and non-disclosure agreements with our employees, strategic partners and consultants, which may not provide adequate protection.
The measures that we use to protect the security of our intellectual property and other proprietary rights may not be adequate, which could result in the loss of legal protection for, and thereby diminish the value of, such intellectual property and other rights.
In addition to pursuing patents on our technology, we also rely upon trademarks, trade secrets, copyrights and unfair competition laws, as well as license agreements and other contractual provisions, to protect our intellectual property and other proprietary rights. Despite these measures, any of our intellectual property rights could be challenged, invalidated, circumvented or misappropriated. In addition, we take steps to protect our intellectual property and proprietary technology by entering into confidentiality agreements and intellectual property assignment agreements with our employees, consultants, corporate partners and, when needed, our advisors. Such agreements may not be enforceable or may not provide meaningful protection for our trade secrets or other proprietary information in the event of unauthorized use or disclosure or other breaches of the agreements, and we may not be able to prevent such unauthorized disclosure. Moreover, if a party having an agreement with us has an overlapping or conflicting obligation to a third party, our rights in and to certain intellectual property could be undermined. Monitoring unauthorized disclosure is difficult, and we do not know whether the steps we have taken to prevent such disclosure are, or will be, adequate. If we were to enforce a claim that a third party had illegally obtained and was using our trade secrets, it would be expensive and time consuming, the outcome would be unpredictable, and any remedy may be inadequate. In addition, courts outside the United States may be less willing to protect trade secrets.
In addition, competitors could purchase our products and attempt to replicate some or all of the competitive advantages we derive from our development efforts, willfully infringe our intellectual property rights, design around our protected technology or develop their own competitive technologies that fall outside of our intellectual property rights. If our intellectual property does not adequately protect our market share against competitors' products and methods, our competitive position could be adversely affected, as could our business.
Some of our owned and in-licensed intellectual property has been discovered through government funded programs and thus is subject to federal regulations such as "march-in" rights, certain reporting requirements, and a preference for U.S. industry. Compliance with such regulations may limit our exclusive rights, subject us to expenditure of resources with respect to reporting requirements, and limit our ability to contract with non-U.S. manufacturers.
Some of the intellectual property rights we own and have in-licensed have been generated through the use of U.S. government funding and are therefore subject to certain federal regulations. For example, all of the issued U.S. patents we own and all of the intellectual property rights licensed to us under our license
33
agreement with Tufts have been generated using U.S. government funds. As a result, the U.S. government has certain rights to intellectual property embodied in our current or future product candidates pursuant to the Bayh-Dole Act of 1980, or Bayh-Dole Act. These U.S. government rights in certain inventions developed under a government-funded program include a non-exclusive, non-transferable, irrevocable worldwide license to use inventions for any governmental purpose. In addition, the U.S. government has the right to require us to grant exclusive, partially exclusive, or non-exclusive licenses to any of these inventions to a third party if the government determines that: (i) adequate steps have not been taken to commercialize the invention; (ii) government action is necessary to meet public health or safety needs; or (iii) government action is necessary to meet requirements for public use under federal regulations (also referred to as "march-in rights"). The U.S. government also has the right to take title to these inventions if we fail, or the applicable licensor fails, to disclose the invention to the government, elect title, and file an application to register the intellectual property within specified time limits. In addition, the U.S. government may acquire title to these inventions in any country in which a patent application is not filed within specified time limits. Intellectual property generated under a government funded program is also subject to certain reporting requirements, compliance with which may require us, or the applicable licensor, to expend substantial resources. In addition, the U.S. government requires that any products embodying the subject invention or produced through the use of the subject invention be manufactured substantially in the U.S. The manufacturing preference requirement can be waived if the owner of the intellectual property can show that reasonable but unsuccessful efforts have been made to grant licenses on similar terms to potential licensees that would be likely to manufacture substantially in the U.S. or that under the circumstances domestic manufacture is not commercially feasible. This preference for U.S. manufacturing may limit our ability to license the applicable patent rights on an exclusive basis under certain circumstances.
If we enter into future arrangements involving government funding, and we make inventions as a result of such funding, intellectual property rights to such discoveries may be subject to the applicable provisions of the Bayh-Dole Act. To the extent any of our current or future intellectual property is generated through the use of U.S. government funding, the provisions of the Bayh-Dole Act may similarly apply. Any exercise by the government of certain of its rights could harm our competitive position, business, financial condition, results of operations and prospects.
We depend on technology that is licensed to us by Tufts University. Any loss of our rights to this technology could prevent us from selling our products.
Our core Simoa technology is licensed exclusively to us from Tufts University. We do not own the patents that underlie this license. Our rights to use this technology and employ the inventions claimed in the licensed patents are subject to the continuation of and compliance with the terms of the license. Our principal obligations under our license agreement with Tufts are as follows:
- •
- royalty payments;
- •
- milestone payments;
- •
- annual maintenance fees;
- •
- using commercially reasonable efforts to develop and sell a product using the licensed technology and developing a market for such product;
- •
- paying and/or reimbursing fees related to prosecution, maintenance and enforcement of patent rights; and
34
- •
- providing certain reports.
If we breach any of these obligations, Tufts may have the right to terminate the license, which could result in our being unable to develop, manufacture and sell our Simoa products or a competitor's gaining access to the Simoa technology. Termination of our license agreement with Tufts would have a material adverse effect on our business.
In addition, we are a party to a number of other agreements that include licenses to intellectual property, including non-exclusive licenses. We expect that we may need to enter into additional license agreements in the future. Our business could suffer, for example, if any current or future licenses terminate, if the licensors fail to abide by the terms of the license, if the licensed patents or other rights are found to be invalid or unenforceable, or if we are unable to enter into necessary licenses on acceptable terms.
As we have done previously, we may need or may choose to obtain licenses from third parties to advance our research or allow commercialization of our current or future products, and we cannot provide any assurances that third-party patents do not exist that might be enforced against our current or future products in the absence of such a license. We may fail to obtain any of these licenses on commercially reasonable terms, if at all. Even if we are able to obtain a license, it may be non-exclusive, thereby giving our competitors access to the same technologies licensed to us. In that event, we may be required to expend significant time and resources to develop or license replacement technology. If we are unable to do so, we may be unable to develop or commercialize the affected products, which could materially harm our business and the third parties owning such intellectual property rights could seek either an injunction prohibiting our sales, or, with respect to our sales, an obligation on our part to pay royalties and/or other forms of compensation.
Licensing of intellectual property is important to our business and involves complex legal, business and scientific issues. Disputes may arise between us and our licensors regarding intellectual property subject to a license agreement, including:
- •
- the scope of rights granted under the license agreement and other interpretation-related issues;
- •
- whether and the extent to which our technology and processes infringe on intellectual property of the licensor that is not subject to the
licensing agreement;
- •
- our right to sublicense patent and other rights to third parties under collaborative development relationships;
- •
- our diligence obligations with respect to the use of the licensed technology in relation to our development and commercialization of our
products, and what activities satisfy those diligence obligations; and
- •
- the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our partners.
If disputes over intellectual property that we have licensed prevent or impair our ability to maintain our current licensing arrangements on acceptable terms, we may be unable to successfully develop and commercialize the affected product, or the dispute may have an adverse affect on our results of operation.
In addition to agreements pursuant to which we in-license intellectual property, we have in the past and will continue in the future to grant licenses under our intellectual property. For example, we have granted certain exclusive and co-exclusive licenses in certain fields to bioMérieux and a non-exclusive license to a
35
diagnostic company in certain fields. Like our in-licenses, our out-licenses are complex and disputes may arise between us and our licensees, such as the types of disputes described above. Moreover, our licensees may breach their obligations, or we may be exposed to liability due to our failure or alleged failure to satisfy our obligations. Any such an occurrence could have an adverse affect on our business.
If we or any of our partners are sued for infringing intellectual property rights of third parties, it would be costly and time consuming, and an unfavorable outcome in that litigation could have a material adverse effect on our business.
Our success also depends on our ability to develop, manufacture, market and sell our products and perform our services without infringing upon the proprietary rights of third parties. Numerous U.S. and foreign-issued patents and pending patent applications owned by third parties exist in the fields in which we are developing products and services. As part of a business strategy to impede our successful commercialization and entry into new markets, competitors may claim that our products and/or services infringe their intellectual property rights.
We could incur substantial costs and divert the attention of our management and technical personnel in defending ourselves against claims of infringement made by third parties. Any adverse ruling by a court or administrative body, or perception of an adverse ruling, may have a material adverse impact on our ability to conduct our business and our finances. Moreover, third parties making claims against us may be able to obtain injunctive relief against us, which could block our ability to offer one or more products or services and could result in a substantial award of damages against us. In addition, since we sometimes indemnify customers, collaborators or licensees, we may have additional liability in connection with any infringement or alleged infringement of third party intellectual property.
Because patent applications can take many years to issue there may be pending applications, some of which are unknown to us, that may result in issued patents upon which our product candidates or proprietary technologies may infringe. Moreover, we may fail to identify issued patents of relevance or incorrectly conclude that an issued patent is invalid or not infringed by our technology or any of our products. There is a substantial amount of litigation involving patent and other intellectual property rights in our industry. If a third-party claims that we or any of our licensors, customers or collaboration partners infringe upon a third-party's intellectual property rights, we may have to:
- •
- seek to obtain licenses that may not be available on commercially reasonable terms, if at all;
- •
- abandon any infringing product or redesign our products or processes to avoid infringement;
- •
- pay substantial damages including, in an exceptional case, treble damages and attorneys' fees, which we may have to pay if a court decides that
the product candidate or proprietary technology at issue infringes upon or violates the third-party's rights;
- •
- pay substantial royalties or fees or grant cross-licenses to our technology; or
- •
- defend litigation or administrative proceedings that may be costly whether we win or lose, and which could result in a substantial diversion of our financial and management resources.
We may be involved in lawsuits to protect or enforce our patents or the patents of our licensors, which could be expensive, time consuming and unsuccessful.
Competitors may infringe our patents or the patents of our licensors. In the event of infringement or unauthorized use, we may file one or more infringement lawsuits, which can be expensive and time consuming. An adverse result in any such litigation proceedings could put one or more of our patents at
36
risk of being invalidated, being found to be unenforceable or being interpreted narrowly and could put our patent applications at risk of not issuing. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation.
Most of our competitors are larger than we are and have substantially greater resources. They are, therefore, likely to be able to sustain the costs of complex patent litigation longer than we could. In addition, the uncertainties associated with litigation could have a material adverse effect on our ability to raise the funds necessary to continue our operations, continue our internal research programs, in-license needed technology, or enter into development partnerships that would help us bring our products to market.
In addition, patent litigation can be very costly and time consuming. An adverse outcome in such litigation or proceedings may expose us or any of our future development partners to loss of our proprietary position, expose us to significant liabilities, or require us to seek licenses that may not be available on commercially acceptable terms, if at all.
Our issued patents could be found invalid or unenforceable if challenged in court, which could have a material adverse impact on our business.
If we or any of our partners were to initiate legal proceedings against a third-party to enforce a patent covering one of our products or services, the defendant in such litigation could counterclaim that our patent is invalid and/or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity and/or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness or non-enablement, or failure to claim patent eligible subject matter. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement, during prosecution. Third parties may also raise similar claims before the USPTO even outside the context of litigation. The outcome following legal assertions of invalidity and unenforceability is unpredictable. With respect to the validity question, for example, we cannot be certain that there is no invalidating prior art of which we and the patent examiner were unaware during prosecution. If a defendant were to prevail on a legal assertion of invalidity and/or unenforceability, we would lose at least part, and perhaps all, of the challenged patent. Such a loss of patent protection would have a material adverse impact on our business.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed alleged trade secrets of their other clients or former employers to us, which could subject us to costly litigation.
As is common in the life sciences industry, we engage the services of consultants and independent contractors to assist us in the development of our products. Many of these consultants and independent contractors were previously employed at, or may have previously or may be currently providing consulting or other services to, universities or other technology, biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may become subject to claims that our company, a consultant or an independent contractor inadvertently or otherwise used or disclosed trade secrets or other information proprietary to their former employers or their former or current clients. We may similarly be subject to claims stemming from similar actions of an employee, such as one who was previously employed by another company, including a competitor or potential competitor. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in
37
substantial costs and be a distraction to our management team. If we were not successful we could lose access or exclusive access to valuable intellectual property.
We may be subject to claims challenging the inventorship or ownership of our patents and other intellectual property.
We generally enter into confidentiality and intellectual property assignment agreements with our employees, consultants, and contractors. These agreements generally provide that inventions conceived by the party in the course of rendering services to us will be our exclusive property. However, those agreements may not be honored and may not effectively assign intellectual property rights to us. For example, even if we have a consulting agreement in place with an academic advisor pursuant to which such academic advisor is required to assign any inventions developed in connection with providing services to us, such academic advisor may not have the right to assign such inventions to us, as it may conflict with his or her obligations to assign all such intellectual property to his or her employing institution.
In addition, we sometimes enter into agreements where we provide services to third parties, such as customers. Under such circumstances, our agreements may provide that certain intellectual property that we conceive in the course of providing those services is assigned to the customer. In those cases, we would not be able to use that particular intellectual property in, for example, our work for other customers without a license.
We may not be able to protect our intellectual property rights throughout the world, which could materially, negatively affect our business.
Filing, prosecuting and defending patents on current and future products in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States can be less extensive than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States. Consequently, regardless of whether we are able to prevent third parties from practicing our inventions in the United States, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not pursued and obtained patent protection to develop their own products, and further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as it is in the United States. These products may compete with our products and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing. Even if we pursue and obtain issued patents in particular jurisdictions, our patent claims or other intellectual property rights may not be effective or sufficient to prevent third parties from so competing. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time-consuming process with uncertain outcomes. Accordingly, we may choose not to seek patent protection in certain countries, and we will not have the benefit of patent protection in such countries.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biotechnology, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or
38
interpreted narrowly and our patent applications at risk of not issuing and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license and may adversely impact our business.
In addition, we and our partners also face the risk that our products are imported or reimported into markets with relatively higher prices from markets with relatively lower prices, which would result in a decrease of sales and any payments we receive from the affected market. Recent developments in U.S. patent law have made it more difficult to stop these and related practices based on theories of patent infringement.
Changes in patent laws or patent jurisprudence could diminish the value of patents in general, thereby impairing our ability to protect our product candidates.
As is the case with other life science industry companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents involve both technological complexity and legal complexity. Therefore, obtaining and enforcing patents is costly, time-consuming and inherently uncertain. In addition, the America Invents Act, or the AIA, was signed into law on September 16, 2011, and many of the substantive changes became effective on March 16, 2013.
An important change introduced by the AIA is that, as of March 16, 2013, the United States transitioned to a "first-to-file" system for deciding which party should be granted a patent when two or more patent applications are filed by different parties claiming the same invention. A third party that files a patent application in the USPTO, after that date but before us could therefore be awarded a patent covering an invention of ours even if we had made the invention before it was made by the third party. This will require us to be cognizant going forward of the time from invention to filing of a patent application, but circumstances could prevent us from promptly filing patent applications on our inventions.
Among some of the other changes introduced by the AIA are changes that limit where a patent holder may file a patent infringement suit and providing additional opportunities for third parties to challenge any issued patent in the USPTO. This applies to all of our owned and in-licensed U.S. patents, even those issued before March 16, 2013. Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in U.S. federal courts necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action. Accordingly, a third party may attempt to use the USPTO procedures to invalidate our patent claims that would not have been invalidated if first challenged by the third party as a defendant in a district court action. The AIA and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
Additionally, the U.S. Supreme Court has ruled on several patent cases in recent years, such as Impression Products, Inc. v. Lexmark International, Inc., Association for Molecular Pathology v. Myriad Genetics, Inc,, Mayo Collaborative Services v. Prometheus Laboratories, Inc. and Alice Corporation Pty. Ltd. v. CLS Bank International, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable
39
ways that could weaken our ability to obtain new patents or to enforce our existing patents and patents that we might obtain in the future.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other provisions during the patent process. There are situations in which noncompliance can result in abandonment or lapse of a patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, competitors might be able to enter the market earlier than would otherwise have been the case. In some cases, our licensors may be responsible for, for example, these payments, thereby decreasing our control over compliance with these requirements.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
Our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented or declared generic or determined to be infringing on other marks. We may not be able to protect our rights to these trademarks and trade names, which we need to build name recognition by potential partners or customers in our markets of interest. At times, competitors may adopt trade names or trademarks similar to ours, thereby impeding our ability to build brand identity and possibly leading to market confusion. In addition, there could be potential trade name or trademark infringement claims brought by owners of other registered trademarks. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected.
We may use third-party open source software components in future products, and failure to comply with the terms of the underlying open source software licenses could restrict our ability to sell such products.
While our current products do not contain any software tools licensed by third-party authors under "open source" licenses, we may choose to use open source software in future products. Use and distribution of open source software may entail greater risks than use of third-party commercial software, as open source licensors generally do not provide warranties or other contractual protections regarding infringement claims or the quality of the code. Some open source licenses may contain requirements that we make available source code for modifications or derivative works we create based upon the type of open source software we use. If we combine our proprietary software with open source software in a certain manner, we could, under certain open source licenses, be required to release the source code of our proprietary software to the public. This would allow our competitors to create similar products with less development effort and time and ultimately could result in a loss of product sales.
Although we intend to monitor any use of open source software to avoid subjecting our products to conditions we do not intend, the terms of many open source licenses have not been interpreted by U.S. courts, and there is a risk that any such licenses could be construed in a way that could impose unanticipated conditions or restrictions on our ability to commercialize our products. Moreover, we cannot assure investors that our processes for controlling our use of open source software in our products will be effective. If we are held to have breached the terms of an open source software license, we could be required to seek licenses from third parties to continue offering our products on terms that are not
40
economically feasible, to re-engineer our products, to discontinue the sale of our products if re-engineering could not be accomplished on a timely basis, or to make generally available, in source code form, our proprietary code, any of which could adversely affect our business, operating results, and financial condition.
We use third-party software that may be difficult to replace or cause errors or failures of our products that could lead to lost customers or harm to our reputation.
We use software licensed from third parties in our products. In the future, this software may not be available to us on commercially reasonable terms, or at all. Any loss of the right to use any of this software could result in delays in the production of our products until equivalent technology is either developed by us, or, if available, is identified, obtained and integrated, which could harm our business. In addition, any errors or defects in third-party software or other third-party software failures could result in errors, defects or cause our products to fail, which could harm our business and be costly to correct. Many of these providers attempt to impose limitations on their liability for such errors, defects or failures, and if enforceable, we may have additional liability to our customers or third-party providers that could harm our reputation and increase our operating costs.
We will need to maintain our relationships with third-party software providers and to obtain software from such providers that does not contain any errors or defects. Any failure to do so could adversely impact our ability to deliver reliable products to our customers and could harm our results of operations.
Numerous factors may limit any potential competitive advantage provided by our intellectual property rights.
The degree of future protection afforded by our intellectual property rights is uncertain because intellectual property rights have limitations, and may not adequately protect our business, provide a barrier to entry against our competitors or potential competitors, or permit us to maintain our competitive advantage. Moreover, if a third party has intellectual property rights that cover the practice of our technology, we may not be able to fully exercise or extract value from our intellectual property rights. The following examples are illustrative:
- •
- others may be able to develop and/or practice technology that is similar to our technology or aspects of our technology but that is not covered
by the claims of any patents that have, or may, issue from our owned or in-licensed patent applications;
- •
- we might not have been the first to make the inventions covered by a pending patent application that we own or license;
- •
- we might not have been the first to file patent applications covering an invention;
- •
- others may independently develop similar or alternative technologies without infringing our intellectual property rights;
- •
- pending patent applications that we own or license may not lead to issued patents;
- •
- patents, if issued, that we own or license may not provide us with any competitive advantages, or may be held invalid or unenforceable, as a
result of legal challenges by our competitors;
- •
- third parties may compete with us in jurisdictions where we do not pursue and obtain patent protection;
41
- •
- we may not be able to obtain and/or maintain necessary or useful licenses on reasonable terms or at all;
- •
- third parties may assert an ownership interest in our intellectual property and, if successful, such disputes may preclude us from exercising
exclusive rights over that intellectual property;
- •
- we may not be able to maintain the confidentiality of our trade secrets or other proprietary information;
- •
- we may not develop or in-license additional proprietary technologies that are patentable; and
- •
- the patents of others may have an adverse effect on our business.
Should any of these events occur, they could significantly harm our business and results of operations.
Risks related to our common stock, this offering and being a public company
We expect that our stock price may fluctuate significantly and investors may not be able to resell their shares at or above the initial public offering price.
Prior to this offering, you could not buy or sell our common stock publicly. Although we have applied to have our common stock listed on The Nasdaq Global Market, an active trading market for our shares may never develop or be sustained following this offering. You may be unable to sell your shares of common stock at or above the initial offering price. The market price of shares of our common stock could be subject to wide fluctuations in response to many risk factors listed in this section, and others beyond our control, including:
- •
- actual or anticipated fluctuations in our financial condition and operating results;
- •
- announcements by us, our partners or our competitors of new products, significant contracts, strategic partnerships, joint ventures,
collaborations, commercial relationships or capital commitments;
- •
- competition from existing products or new products that may emerge;
- •
- failure to meet or exceed financial estimates and projections of the investment community or that we provide to the public;
- •
- issuance of new or updated research or reports by securities analysts or recommendations for our stock;
- •
- adverse regulatory announcements;
- •
- disputes or other developments related to proprietary rights, including patents, litigation matters, and our ability to obtain patent
protection for our technologies;
- •
- commencement of, or our involvement in, litigation;
- •
- fluctuations in the valuation of companies perceived by investors to be comparable to us;
- •
- market conditions in our markets;
- •
- manufacturing disputes or delays;
- •
- any future sales of our common stock or other securities;
- •
- any change to the composition of the board of directors or key personnel;
- •
- expiration of contractual lock-up agreements with our executive officers, directors and security holders;
42
- •
- general economic conditions and slow or negative growth of our markets;
- •
- share price and volume fluctuations attributable to inconsistent trading volume levels of our shares;
- •
- announcement or expectation of additional debt or equity financing efforts; and
- •
- other factors described in this section of the prospectus.
These and other market and industry factors may cause the market price and demand for our common stock to fluctuate substantially, regardless of our actual operating performance, which may limit or prevent investors from readily selling their shares of common stock and may otherwise negatively affect the liquidity of our common stock. In addition, the stock market in general, and life science companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. In the past, when the market price of a stock has been volatile, holders of that stock have on occasion instituted securities class action litigation against the company that issued the stock. If any of our stockholders were to bring a lawsuit against us, the defense and disposition of the lawsuit could be costly and divert the time and attention of our management and harm our operating results.
If securities or industry analysts do not publish research reports about our business, or if they issue an adverse opinion about our business, our stock price and trading volume could decline.
The trading market for our common stock will be influenced by the research and reports that industry or securities analysts publish about us or our business. If one or more of the analysts who cover us issues an adverse opinion about our company, our stock price would likely decline. If one or more of these analysts ceases coverage of us or fails to regularly publish reports on us, we could lose visibility in the public markets, which could cause our stock price or trading volume to decline.
Our principal stockholders and management own a significant percentage of our stock and will be able to exercise significant influence over matters subject to stockholder approval.
Our executive officers, directors and principal stockholders listed in the table in the section entitled "Principal stockholders" beneficially owned approximately 82% of our shares of common stock outstanding as of October 31, 2017, which reflects the assumed conversion of all outstanding shares of our preferred stock prior to the completion of this offering, and we expect that upon the closing of this offering, that same group will beneficially own at least % of our common stock. Accordingly, after this offering, our executive officers, directors and principal stockholders will continue to have significant influence over our operations. This concentration of ownership could have the effect of delaying or preventing a change in our control or otherwise discouraging a potential acquirer from attempting to obtain control of us, which in turn could have a material adverse effect on our stock price and may prevent attempts by our stockholders to replace or remove the board of directors or management.
Future sales of our common stock in the public market could cause our stock price to fall.
Our stock price could decline as a result of sales of a large number of shares of our common stock after this offering or the perception that these sales could occur. These sales, or the possibility that these sales may occur, also might make it more difficult for us to sell equity securities in the future at a time and at a price that we deem appropriate.
Upon the closing of this offering, shares of our common stock will be outstanding (or shares assuming full exercise of the underwriters' option to purchase additional shares), based on our shares outstanding as of October 31, 2017. All shares of common stock expected to be sold in this
43
offering will be freely tradable without restriction or further registration under the Securities Act of 1933, as amended, or the Securities Act, unless held by our "affiliates," as that term is defined in Rule 144 under the Securities Act. The resale of the remaining shares, or % of our outstanding shares after this offering, are currently prohibited or otherwise restricted as a result of securities law provisions, market standoff agreements entered into by our stockholders with us or lock-up agreements entered into by our stockholders with the underwriters; however, subject to applicable securities law restrictions, these shares will be able to be sold in the public market beginning 180 days after the date of this prospectus. In addition, the shares subject to outstanding options and warrants, of which options and warrants to purchase 2,742,669 shares and 387,811 shares, respectively, were exercisable as of October 31, 2017, and the shares reserved for future issuance under our stock option and equity incentive plans will become available for sale immediately upon the exercise of such options and the expiration of any applicable market stand-off or lock-up agreements. For more information, see the section of this prospectus captioned "Shares eligible for future sale."
Holders of approximately shares of our common stock issued or issuable upon conversion of preferred stock and exercise of warrants, or %, of our common stock, will have rights, subject to some conditions, to require us to file registration statements covering the sale of their shares or to include their shares in registration statements that we may file for ourselves or other stockholders. We also intend to register the offer and sale of all shares of common stock that we may issue under our equity compensation plans. Once we register the offer and sale of shares for the holders of registration rights and option holders, they can be freely sold in the public market upon issuance, subject to the lock-up agreements described in the section of this prospectus entitled "Underwriting."
In addition, in the future, we may issue additional shares of common stock or other equity or debt securities convertible into common stock in connection with a financing, acquisition, litigation settlement, employee arrangements or otherwise. Any such issuance could result in substantial dilution to our existing stockholders and could cause our stock price to decline.
Our management team has broad discretion to use the net proceeds from this offering and its investment of these proceeds may not yield a favorable return. They may invest the proceeds of this offering in ways with which investors disagree.
We have broad discretion as to how to spend and invest the proceeds from this offering, and we may spend or invest these proceeds in a way with which our stockholders disagree. Accordingly, investors will need to rely on our judgment with respect to the use of these proceeds. We currently intend to use the proceeds from this offering: (1) to expand our life sciences commercial operations to grow and support the installed base of our products among life sciences research customers in the United States and internationally; (2) to improve and update our Simoa technology and instruments and to develop additional assays, including assays for nucleic acid detection; (3) to support the launch of our new Quanterix SR-X instrument, currently scheduled for launch in 2018; (4) to potentially move into a larger corporate headquarters in order to have the appropriate infrastructure to support the increase in our employee base in addition to an increase in our manufacturing footprint; (5) to pursue regulatory approvals or clearances to develop instruments, assay kits and consumables in areas outside of life science research, including potentially LDTs, IVD tests and other markets, and, subject to the receipt of such necessary regulatory approvals or clearances, to develop such instruments, assay kits and consumables; (6) to potentially pursue acquisitions or other business development opportunities; and (7) for working capital and other general corporate purposes. We may also use a portion of the net proceeds to acquire, license and invest in complementary products, technologies or businesses; however, we currently have no agreements or
44
commitments to complete any such transaction. These uses may not yield a favorable return to our stockholders.
We cannot specify with certainty all of the particular uses for the net proceeds to be received upon the closing of this offering. In addition, the amount, allocation and timing of our actual expenditures will depend upon numerous factors, including the revenue generated from the sale of our products. Accordingly, we will have broad discretion in using these proceeds. In addition, until the net proceeds are used, they may be placed in investments that do not produce significant income or that may lose value.
We have never paid dividends on our capital stock and we do not anticipate paying any dividends in the foreseeable future. Consequently, any gains from an investment in our common stock will likely depend on whether the price of our common stock increases.
We have not paid dividends on any of our classes of capital stock to date and we currently intend to retain our future earnings, if any, to fund the development and growth of our business. In addition, the terms of our indebtedness with Hercules prohibit us from paying dividends. As a result, capital appreciation, if any, of our common stock will be your sole source of gain for the foreseeable future. Consequently, in the foreseeable future, you will likely only experience a gain from your investment in our common stock if the price of our common stock increases.
Investors in this offering will pay a higher price than the book value of our common stock.
The initial public offering price of our common stock is substantially higher than the net tangible book value per share of our common stock. If you purchase common stock in this offering, you will pay more for your shares than the amounts paid by existing stockholders for their shares. You will incur immediate and substantial dilution of $ per share, representing the difference between our pro forma as adjusted net tangible book value per share after giving effect to this offering and the assumed initial public offering price of $ per share, which is the midpoint of the estimated offering price range set forth on the cover page of this prospectus. In the past, we issued options and warrants to acquire capital stock at prices significantly below the initial public offering price. To the extent the underwriters exercise their option to purchase additional shares or any outstanding options or warrants are ultimately exercised, you will sustain further dilution. For a further description of the dilution that you will experience immediately after the offering, see the section of this prospectus captioned "Dilution."
Anti-takeover provisions contained in our restated certificate of incorporation and restated by-laws to be effective upon the closing of the offering, as well as provisions of Delaware law, could impair a takeover attempt.
Our restated certificate of incorporation, restated by-laws and Delaware law contain provisions which could have the effect of rendering more difficult, delaying or preventing an acquisition deemed undesirable by our board of directors. Our corporate governance documents include or will include provisions:
- •
- authorizing our board of directors to issue up to 5,000,000 shares of preferred stock without stockholder approval upon the terms and
conditions and with the rights, privileges and preferences as our board of directors may determine;
- •
- specifying that special meetings of our stockholders can be called only by our board of directors and that our stockholders may not act by written consent;
45
- •
- establishing an advance notice procedure for stockholder proposals to be brought before an annual meeting of our stockholders, including
proposed nominations of persons for election to our board of directors;
- •
- providing that directors may be removed only for cause;
- •
- providing that our board of directors may create new directorships and that vacancies on our board of directors may be filled only by a
majority of directors then in office, even though less than a quorum;
- •
- establishing that our board of directors is divided into three classes—Class I, Class II, and
Class III—with each class serving staggered three-year terms;
- •
- providing that our board of directors may amend our restated by-laws without stockholder approval; and
- •
- requiring a super-majority of votes to amend certain of the above-mentioned provisions.
These provisions, alone or together, could delay or prevent hostile takeovers and changes in control or changes in our management.
As a Delaware corporation, we are also subject to provisions of Delaware law, including Section 203 of the Delaware General Corporation law, which prevents some stockholders holding more than 15% of our outstanding common stock from engaging in certain business combinations without approval of the holders of substantially all of our outstanding common stock.
Any provision of our restated certificate of incorporation, restated by-laws or Delaware law that has the effect of delaying or deterring a change in control could limit the opportunity for our stockholders to receive a premium for their shares of our common stock, and could also affect the price that some investors are willing to pay for our common stock.
We will incur significant costs as a result of operating as a public company and our management expects to devote substantial time to public company compliance programs.
As a public company, we will incur significant legal, accounting and other expenses due to our compliance with regulations and disclosure obligations applicable to us, including compliance with the Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act, as well as rules implemented by the SEC and the Nasdaq Stock Market, or Nasdaq. The SEC and other regulators have continued to adopt new rules and regulations and make additional changes to existing regulations that require our compliance. In July 2010, the Dodd-Frank Wall Street Reform and Consumer Protection Act, or the Dodd-Frank Act, was enacted. There are significant corporate governance and executive compensation related provisions in the Dodd-Frank Act that have required the SEC to adopt additional rules and regulations in these areas. Stockholder activism, the current political environment, and the current high level of government intervention and regulatory reform may lead to substantial new regulations and disclosure obligations, which may lead to additional compliance costs and impact, in ways we cannot currently anticipate, the manner in which we operate our business. Our management and other personnel will devote a substantial amount of time to these compliance programs and monitoring of public company reporting obligations and as a result of the corporate governance and executive compensation related rules, regulations, and guidelines prompted by the Dodd-Frank Act and further regulations and disclosure obligations expected in the future, we will likely need to devote additional time and costs to comply with such compliance programs and rules. These rules and regulations will cause us to incur significant legal and financial compliance costs and will make some activities more time-consuming and costly.
46
To comply with the requirements of being a public company, we may need to undertake various actions, including implementing new internal controls and procedures and hiring new accounting or internal audit staff. The Sarbanes-Oxley Act requires that we maintain effective disclosure controls and procedures and internal control over financial reporting. We are continuing to develop and refine our disclosure controls and other procedures that are designed to ensure that information required to be disclosed by us in the reports that we file with the SEC is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms, and that information required to be disclosed in reports under the Exchange Act, is accumulated and communicated to our principal executive and financial officers. Our current controls and any new controls that we develop may become inadequate and weaknesses in our internal control over financial reporting may be discovered in the future. Any failure to develop or maintain effective controls when we become subject to this requirement could adversely affect the results of periodic management evaluations and annual independent registered public accounting firm attestation reports regarding the effectiveness of our internal control over financial reporting that we may be required to include in our periodic reports we will file with the SEC under Section 404 of the Sarbanes-Oxley Act, harm our operating results, cause us to fail to meet our reporting obligations, or result in a restatement of our prior period financial statements. Moreover, if we are not able to comply with the requirements of Section 404 in a timely manner, or if we or our independent registered public accounting firm identifies deficiencies in our internal control over financial reporting that are deemed to be material weaknesses, the market price of our stock could decline and we could be subject to sanctions or investigations by the SEC or other regulatory authorities, which would require additional financial and management resources. In the event that we are not able to demonstrate compliance with the Sarbanes-Oxley Act, that our internal control over financial reporting is perceived as inadequate or that we are unable to produce timely or accurate financial statements, investors may lose confidence in our operating results and the price of our common stock could decline. In addition, if we are unable to continue to meet these requirements, we may be unable to remain listed on Nasdaq.
We are not currently required to comply with the SEC rules that implement Section 404 of the Sarbanes-Oxley Act, and are therefore not yet required to make a formal assessment of the effectiveness of our internal control over financial reporting for that purpose. Upon becoming a public company, we will be required to comply with certain of these rules, which will require management to certify financial and other information in our quarterly and annual reports and provide an annual management report on the effectiveness of our internal control over financial reporting commencing with our second annual report. This assessment will need to include the disclosure of any material weaknesses in our internal control over financial reporting identified by our management or our independent registered public accounting firm. We are just beginning the costly and challenging process of compiling the system and processing documentation needed to comply with such requirements. We may not be able to complete our evaluation, testing and any required remediation in a timely fashion. During the evaluation and testing process, if we identify one or more material weaknesses in our internal control over financial reporting, we will be unable to assert that our internal control over financial reporting is effective.
Our independent registered public accounting firm may not be required to formally attest to the effectiveness of our internal control over financial reporting until the later of our second annual report or the first annual report required to be filed with the SEC following the date we are no longer an "emerging growth company" as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, depending on whether we choose to rely on certain exemptions set forth in the JOBS Act. We cannot assure you that there will not be material weaknesses or significant deficiencies in our internal controls in the future. We have in the past discovered, and may in the future discover, areas of our internal financial and accounting controls and procedures that need improvement. If we are unable to assert that our
47
internal control over financial reporting is effective, or if our independent registered public accounting firm is unable to express an opinion on the effectiveness of our internal control over financial reporting, we could lose investor confidence in the accuracy and completeness of our financial reports, which could have a material adverse effect on the price of our common stock.
We are an "emerging growth company" and will be able to avail ourselves of reduced disclosure requirements applicable to emerging growth companies, and we plan to avail ourselves of the ability to adopt new accounting standards on the timeline permitted for private companies, which could make our common stock less attractive to investors and our financial statements less comparable to other companies who are complying with new accounting standards on public company timelines.
We are an "emerging growth company," as defined in the JOBS Act, and we intend to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not "emerging growth companies," including not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. In addition, Section 107 of the JOBS Act also provides that an emerging growth company can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act, for complying with new or revised accounting standards. In other words, an emerging growth company can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected to take advantage of the extended transition period afforded by the JOBS Act for the implementation of new or revised accounting standards and, as a result, will comply with new or revised accounting standards not later than the relevant dates on which adoption of such standards is required for non-public companies. There are currently accounting standards that are expected to affect the financial reporting of many public companies as early as the first calendar quarter of 2018 including ASC 606 Revenue from contracts with customers. As a result of this election, the timeline to comply with these standards will in many cases be delayed as compared to other public companies who are not eligible to have made or have not made this election. For more information on the effect of this election, including the timing of when we currently plan to adopt certain accounting standards that could materially affect our financial statements, refer to Note 2 to the consolidated financial statements included elsewhere in this prospectus. As a result, investors may view our financial statements as not comparable to other public companies. We cannot predict if investors will find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile. We may take advantage of these reporting exemptions until we are no longer an emerging growth company. We will remain an emerging growth company until the earliest of (i) the last day of the fiscal year in which we have total annual gross revenue of $1.07 billion or more; (ii) the last day of our fiscal year following the fifth anniversary of the date of the closing of this offering; (iii) the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; or (iv) the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
48
Special note regarding forward-looking statements
This prospectus contains forward-looking statements that involve risks and uncertainties. All statements other than statements of historical facts contained in this prospectus are forward-looking statements. In some cases, you can identify forward-looking statements by words such as "anticipate," "believe," "contemplate," "continue," "could," "estimate," "expect," "intend," "may," "plan," "potential," "predict," "project," "seek," "should," "target," "will," "would," or the negative of these words or other comparable terminology. These forward-looking statements include, but are not limited to, statements about:
- •
- our use of the net proceeds from this offering;
- •
- the implementation of our business model and strategic plans for our business, products and services;
- •
- the size of the life science research, diagnostics, and health screening markets addressable by our Simoa technology;
- •
- the size and market opportunity for our Simoa technology in the fields of neurology, oncology, cardiology, infectious disease and inflammation;
- •
- the commercialization and adoption of our existing products and services and the success of our new product offerings, including Quanterix SR-X
and the detection of nucleic acids;
- •
- our ability to develop additional assays, including multiplexed assays;
- •
- the accuracy of our estimates regarding expenses, future revenues, capital requirements and our needs for additional financing;
- •
- the ability of our Simoa technology's sensitivity to improve existing diagnostics and to enable the development of new diagnostic tests and
tools;
- •
- the potential of our Simoa technology in the field of companion diagnostics and its adoption by healthcare professionals;
- •
- the impact of our Simoa technology on proteomic research;
- •
- the relevance of proteins versus nucleic acids in understanding the continuum between health and disease;
- •
- the usefulness of the data generated by our Simoa technology in the life science research, diagnostic and precision health screening fields;
and
- •
- our financial performance.
These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including those described in "Risk factors" and elsewhere in this prospectus. Moreover, we operate in a very competitive and rapidly changing environment, and new risks emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this prospectus may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements.
49
You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances reflected in the forward-looking statements will be achieved or occur. We undertake no obligation to update publicly any forward-looking statements for any reason after the date of this prospectus to conform these statements to new information, actual results or to changes in our expectations, except as required by law.
You should read this prospectus and the documents that we reference in this prospectus and have filed with the Securities and Exchange Commission, or SEC, as exhibits to the registration statement of which this prospectus is a part with the understanding that our actual future results, levels of activity, performance, and events and circumstances may be materially different from what we expect.
This prospectus includes statistical and other industry and market data that we obtained from industry publications and research, surveys and studies conducted by third parties. Industry publications and third-party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. This prospectus also contains estimates and other statistical data from a custom market research report by an independent third-party research firm, which was commissioned by us and was issued in June 2017, referred to herein as the Third-Party Research Report. Such data involves a number of assumptions and limitations and contains projections and estimates of the future performance of the markets in which we operate and intend to operate that are subject to a high degree of uncertainty. We caution you not to give undue weight to such projections, assumptions and estimates.
50
We estimate that we will receive net proceeds of approximately $ million from the sale of the shares of common stock offered in this offering, or approximately $ million if the underwriters exercise their option to purchase additional shares in full, based on an assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
Each $1.00 increase (decrease) in the assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus, would increase (decrease) the net proceeds to us from this offering, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, by approximately $ million, assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same. We may also increase or decrease the number of shares we are offering. An increase (decrease) of 1,000,000 in the number of shares we are offering would increase (decrease) the net proceeds to us from this offering, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, by approximately $ million, assuming the initial public offering price stays the same.
We currently expect to use the net proceeds from this offering: (1) to expand our life sciences commercial operations to grow and support the installed base of our products among life sciences research customers in the United States and internationally; (2) to improve and update our Simoa technology and instruments and to develop additional assays, including assays for nucleic acid detection; (3) to support the launch of our new Quanterix SR-X instrument, currently scheduled for launch in 2018; (4) to potentially move into a larger corporate headquarters in order to have the appropriate infrastructure to support the increase in our employee base in addition to an increase in our manufacturing footprint; (5) to pursue regulatory approvals or clearances to develop instruments, assay kits and consumables in areas outside of life science research, including potentially LDTs, IVD tests and other markets, and, subject to receipt of such necessary regulatory approvals or clearances, to develop such instruments, assay kits and consumables; (6) to potentially pursue acquisitions or other business development opportunities; and (7) for working capital and other general corporate purposes. We do not currently market our products outside of the research use only market in part because we do not have the regulatory clearances that would be necessary for us to do so, nor have we begun the process of obtaining any such regulatory clearances. Furthermore, we may require additional funds to pursue any required regulatory approvals or clearances.
We believe opportunities may exist from time to time to expand our current business through acquisitions or in-licenses of complementary companies or technologies. While we have no current agreements, commitments or understandings for any specific acquisitions or in-licenses at this time, we may use a portion of the net proceeds for these purposes.
Although we currently anticipate that we will use the net proceeds from this offering as described above, there may be circumstances where a reallocation of funds is necessary. Due to the uncertainties inherent in the product development process, it is difficult to estimate with certainty the exact amounts of the net proceeds from this offering that may be used for the above purposes. The amounts and timing of our actual expenditures will depend upon numerous factors, including our sales and marketing and commercialization efforts, demand for our technology, our operating costs and the other factors described under "Risk factors" in this prospectus. Accordingly, our management will have flexibility in applying the
51
net proceeds from this offering. An investor will not have the opportunity to evaluate the economic, financial or other information on which we base our decisions on how to use the proceeds.
Pending their use as described above, we plan to invest the net proceeds in short-term, interest-bearing obligations, investment-grade instruments, certificates of deposit or guaranteed obligations of the U.S. government.
We have never declared or paid cash dividends on our capital stock. We currently intend to retain all available funds and future earnings, if any, for use in the operation of our business and do not anticipate paying any cash dividends on our common stock in the foreseeable future. In addition, the terms of our indebtedness with Hercules Capital, Inc. prohibit us from paying dividends. Any future determination to declare and pay dividends will be made at the discretion of our board of directors and will depend on various factors, including applicable laws, our results of operations, our financial condition, our capital requirements, general business conditions, our future prospects and other factors that our board of directors may deem relevant. Investors should not purchase our common stock with the expectation of receiving cash dividends.
52
The following table sets forth our cash and cash equivalents and capitalization as of September 30, 2017:
- •
- on an actual basis;
- •
- on a pro forma basis to reflect (1) the conversion of all outstanding shares of our preferred stock into an aggregate of 45,561,745
shares of common stock prior to the completion of this offering and (2) the conversion of warrants to purchase 387,811 shares of our preferred stock into warrants to purchase 387,811 shares of
common stock prior to the completion of this offering; and
- •
- on a pro forma as adjusted basis to additionally reflect the issuance and sale by us of shares of our common stock in this offering, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, at an assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus.
Our capitalization following the closing of this offering will be adjusted based on the actual initial public offering price and other terms of the offering determined at pricing. You should read this information together with our audited financial statements and related notes appearing elsewhere in this prospectus
53
and the information set forth under the heading "Selected financial data" and "Management's discussion and analysis of financial condition and results of operations."
| | | | | | | | | | | |
| |
As of September 30, 2017 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (in thousands, except share and per share data) |
Actual |
Pro forma |
Pro forma as adjusted |
|||||||
| | | | | | | | | | | |
| |
(unaudited) |
|||||||||
Cash and cash equivalents |
$ | 18,690 | $ | 18,690 | $ | |||||
Long-term debt |
9,328 | 9,328 | 9,328 | |||||||
Preferred stock warrant liability |
781 | — | — | |||||||
Series A Preferred Stock, $0.001 par value: 16,464,442 shares authorized, actual, 16,400,001 shares issued and outstanding, actual; no shares authorized, issued or outstanding, pro forma and pro forma as adjusted |
31,925 | — | — | |||||||
Series B Preferred Stock, $0.001 par value: 6,186,594 shares authorized, actual, 6,021,636 shares issued and outstanding, actual; no shares authorized, issued or outstanding, pro forma and pro forma as adjusted |
18,134 | — | — | |||||||
Series C Preferred Stock, $0.001 par value: 9,791,421 shares authorized, actual, 8,605,944 shares issued and outstanding, actual; no shares authorized, issued or outstanding, pro forma and pro forma as adjusted |
38,397 | — | — | |||||||
Series D Preferred Stock, $0.001 par value: 14,572,992 shares authorized, actual, 14,534,164 shares issued and outstanding, actual; no shares authorized, issued or outstanding, pro forma and pro forma as adjusted |
53,931 | — | — | |||||||
Common stock, $0.001 par value: 72,113,902 shares authorized, actual, 8,021,559 shares issued and outstanding, actual; 72,113,902 shares authorized, pro forma; 53,583,304 shares issued and outstanding, pro forma; shares authorized, pro forma as adjusted; shares issued and outstanding, pro forma as adjusted |
8 | 54 | ||||||||
Additional paid-in capital |
— | 143,122 | ||||||||
Accumulated deficit |
(136,552 | ) | (136,552 | ) | (136,552 | ) | ||||
| | | | | | | | | | | |
Total stockholders' (deficit) equity |
(136,544 | ) | 6,624 | |||||||
Total capitalization |
$ | 15,952 | $ | 15,952 | $ | |||||
| | | | | | | | | | | |
The number of shares of our common stock to be outstanding after this offering excludes the following:
- •
- 7,364,345 shares of common stock issuable upon the exercise of outstanding stock options as of September 30, 2017, having a
weighted-average exercise price of $1.83 per share;
- •
- 387,811 shares of common stock issuable upon the exercise of outstanding warrants as of September 30, 2017, having a weighted-average
exercise price of $2.98 per share; and
- •
- shares of common stock reserved for issuance pursuant to future awards under our 2017 Equity Incentive Plan, as well as any automatic increases in the number of shares of our common stock reserved for future issuance under this plan, which will become effective upon the closing of this offering.
54
If you invest in our common stock, your interest will be diluted to the extent of the difference between the initial public offering price per share of our common stock in this offering and the pro forma as adjusted net tangible book value per share of our common stock after this offering.
As of September 30, 2017, our historical net tangible book value was $(138.8) million, or $(17.31) per share of common stock. Our historical net tangible book value per share is equal to our total tangible assets, less total liabilities and preferred stock, divided by the number of outstanding shares of our common stock. As of September 30, 2017, the pro forma net tangible book value of our common stock was $ million, or $ per share of common stock, taking into account (i) the automatic conversion of our outstanding preferred stock into an aggregate of 45,561,745 shares of common stock prior to the completion of this offering and (ii) the conversion of warrants to purchase 387,811 shares of our preferred stock into warrants to purchase 387,811 shares of common stock prior to the completion of this offering. After giving further effect to the sale of shares of common stock in this offering, after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us, at an assumed initial public offering price of $ per share, the midpoint of the price range set forth on the cover of this prospectus, our pro forma as adjusted net tangible book value as of September 30, 2017, would have been approximately $ million, or approximately $ per share of common stock. This represents an immediate increase in pro forma as adjusted net tangible book value of $ per share to our existing stockholders and an immediate dilution of $ per share to investors participating in this offering. The following table illustrates this per share dilution:
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
Assumed initial public offering price per share of our common stock |
$ | ||||||
Historical net tangible book value per share of our common stock as of September 30, 2017, before giving effect to this offering |
$ | (17.31 | ) | ||||
| | | | | | | | |
Increase attributable to the conversion of outstanding preferred stock |
|||||||
| | | | | | | | |
Pro forma net tangible book value per share as of September 30, 2017, before giving effect to this offering |
|||||||
Increase in net tangible book value per share attributable to new investors |
|||||||
| | | | | | | | |
Pro forma as adjusted net tangible book value per share of our common stock after giving effect to this offering |
|||||||
| | | | | | | | |
Dilution per share of common stock to new investors participating in this offering |
$ | ||||||
| | | | | | | | |
The information discussed above is illustrative only, and the dilution information following this offering will be adjusted based on the actual initial public offering price and other terms of this offering determined at pricing. A $1.00 increase (decrease) in the assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus, would increase (decrease) the pro forma as adjusted net tangible book value by $ per share and the dilution to new investors by $ per share, assuming the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same, and after deducting the estimated underwriting discounts and commissions and estimated expenses payable by us. We may also increase or decrease the number of shares we are offering. An increase of 1,000,000 shares offered by us would increase the pro forma as adjusted net tangible book value by $ per share and decrease the dilution to new investors by $ per share, assuming the assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus, remains the same and after
55
deducting the estimated underwriting discounts and commissions and estimated expenses payable by us. Similarly, a decrease of 1,000,000 shares offered by us would decrease the pro forma as adjusted net tangible book value by $ per share and increase the dilution to new investors by $ per share, assuming the assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus, remains the same and after deducting the estimated underwriting discounts and commissions and estimated expenses payable by us.
The table above assumes no exercise of the underwriters' option to purchase additional shares in this offering. If the underwriters exercise their option to purchase additional shares in full, the pro forma as adjusted net tangible book value as of September 30, 2017, will increase to $ million, or $ per share, representing an increase to existing stockholders of $ per share, and there will be an immediate dilution of $ per share to new investors.
The following table summarizes as of September 30, 2017, on the pro forma as adjusted basis as described above, the differences between the number of shares of common stock purchased from us, the total consideration and the average price per share paid by existing stockholders (giving effect to the conversion of all of our preferred stock into shares of common stock) and by investors participating in this offering, after deducting the estimated underwriting discounts and commissions and estimated offering expenses, at an assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus.
| | | | | | | | | | | | | | | | | |
| |
|
|
Total consideration |
|
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Shares purchased | Average price/ share |
||||||||||||||
| |
Number |
Percent |
Amount |
Percent |
||||||||||||
| | | | | | | | | | | | | | | | | |
Existing stockholders |
% | $ | % | $ | ||||||||||||
Investors participating in this offering |
% | $ | % | $ | ||||||||||||
| | | | | | | | | | | | | | | | | |
Total |
100 | % | $ | 100 | % | |||||||||||
| | | | | | | | | | | | | | | | | |
Each $1.00 increase (decrease) in the assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus, would increase (decrease) the total consideration paid by new investors by $ , and increase (decrease) the percentage of total consideration paid by new investors by approximately %, assuming that the number of shares offered by us, as listed on the cover page of this prospectus, remains the same. Similarly, each increase (decrease) of 1,000,000 shares in the number of shares of common stock offered by us would increase (decrease) the total consideration paid by new investors by $ million and increase (decrease) the percentage of total consideration paid by new investors by approximately % assuming that the assumed initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus, remains the same.
If all of the outstanding options and warrants noted below were exercised, (1) the number of shares of our common stock held by existing stockholders would be increased to shares, or % of the total number of shares of our common stock outstanding after this offering, and the percentage of shares of common stock held by new investors participating in the offering would be decreased to % of the total number of shares of our common stock outstanding after this offering, (2) the consideration paid by existing stockholders would be increased to $ , or % of the total consideration paid by stockholders after this offering, and the percentage of consideration paid by new investors participating in the offering would be decreased to % of the total consideration paid by stockholders after this
56
offering, and (3) the average price per share paid by existing stockholders would decrease to $ per share.
The table above assumes no exercise of the underwriters' option to purchase additional shares in this offering. If the underwriters' option to purchase additional shares is exercised in full, the number of shares of our common stock held by existing stockholders would be reduced to % of the total number of shares of our common stock outstanding after this offering, and the number of shares of common stock held by new investors participating in the offering would be increased to % of the total number of shares of our common stock outstanding after this offering.
The number of shares of common stock to be outstanding after this offering is based on 54,312,740 shares of common stock outstanding as of September 30, 2017, including 729,436 shares of unvested restricted common stock, and excludes the following:
- •
- 7,364,345 shares of common stock issuable upon the exercise of outstanding stock options as of September 30, 2017, having a
weighted-average exercise price of $1.83 per share;
- •
- 387,811 shares of common stock issuable upon the exercise of outstanding warrants as of September 30, 2017, having a weighted-average
exercise price of $2.98 per share; and
- •
- shares of common stock reserved for issuance pursuant to future awards under our 2017 Equity Incentive Plan, as well as any automatic increases in the number of shares of our common stock reserved for future issuance under this plan, which will become effective upon the closing of this offering.
To the extent that any options or warrants are exercised, new options or other securities are issued under our equity incentive plans, or we issue additional shares of common stock in the future, there will be further dilution to investors participating in this offering. In addition, we may choose to raise additional capital because of market conditions or strategic considerations, even if we believe that we have sufficient funds for our current or future operating plans. If we raise additional capital through the sale of equity or convertible debt securities, the issuance of such securities could result in further dilution to our stockholders.
57
You should read the following selected financial data together with our consolidated financial statements and the related notes appearing at the end of this prospectus and the "Management's discussion and analysis of financial condition and results of operations" section of this prospectus. We have derived the statement of operations data for the years ended December 31, 2015 and 2016, and the balance sheet data as of December 31, 2015 and 2016, from our audited consolidated financial statements included elsewhere in this prospectus. The statement of operations data for the nine months ended September 30, 2016 and 2017 and the balance sheet data as of September 30, 2017 have been derived from our unaudited consolidated financial statements included elsewhere in this prospectus and which have been prepared on the same basis as the audited consolidated financial statements. In the opinion of management, the unaudited data reflects all adjustments, consisting only of normal recurring adjustments, necessary for a fair presentation of the financial information in those statements. Our historical results are not necessarily indicative of the results that should be expected in the future.
Consolidated statement of operations data (in thousands, except per share data)
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Year ended December 31 |
Nine months ended September 30 |
|||||||||||
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Total revenue |
$ | 12,180 | $ | 17,585 | $ | 10,906 | $ | 16,285 | |||||
Cost of revenue |
6,465 |
9,837 |
6,746 |
9,179 |
|||||||||
Research and development |
10,083 | 16,993 | 10,192 | 12,377 | |||||||||
Selling, general and administrative |
10,155 | 12,466 | 8,866 | 13,641 | |||||||||
| | | | | | | | | | | | | | |
Total operating expenses |
26,703 | 39,296 | 25,804 | 35,197 | |||||||||
| | | | | | | | | | | | | | |
Loss from operations |
(14,523 | ) | (21,711 | ) | (14,898 | ) | (18,912 | ) | |||||
Interest expense, net |
(1,040 | ) | (1,298 | ) | (1,012 | ) | (735 | ) | |||||
Other income (expense), net |
(380 | ) | (164 | ) | 51 | 10 | |||||||
| | | | | | | | | | | | | | |
Net loss |
(15,943 | ) | (23,173 | ) | (15,859 | ) | (19,637 | ) | |||||
Accretion and accrued dividends on redeemable convertible preferred stock |
(4,355 | ) | (4,445 | ) | (3,325 | ) | (3,349 | ) | |||||
| | | | | | | | | | | | | | |
Net loss attributable to common stockholders |
$ | (20,298 | ) | $ | (27,618 | ) | $ | (19,184 | ) | $ | (22,986 | ) | |
| | | | | | | | | | | | | | |
Net loss per share attributable to common stockholders, basic and diluted |
$ | (3.48 | ) | $ | (4.01 | ) | $ | (2.83 | ) | $ | (2.96 | ) | |
| | | | | | | | | | | | | | |
Weighted-average common shares outstanding |
5,828 | 6,887 | 6,782 | 7,768 | |||||||||
| | | | | | | | | | | | | | |
Consolidated balance sheet data (in thousands)
| |
|
|
|
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | |
| |
As of December 31, 2015 |
As of December 31, 2016 |
As of September 30, 2017 |
|||||||
| | | | | | | | | | | |
| |
|
|
(unaudited) |
|||||||
Cash and cash equivalents |
$ | 2,323 | $ | 29,671 | $ | 18,690 | ||||
Total assets |
7,351 | 37,117 | 30,515 | |||||||
Total long term debt |
9,726 | 10,243 | 9,328 | |||||||
Total redeemable convertible preferred stock |
73,445 | 128,585 | 142,387 | |||||||
Total stockholders' deficit |
(88,640 | ) | (115,109 | ) | (136,544 | ) | ||||
| | | | | | | | | | | |
58
Management's discussion and analysis of financial condition and results of operations
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our consolidated financial statements and related notes included elsewhere in this prospectus. In addition to historical consolidated financial information, the following discussion contains forward-looking statements that reflect our plans, estimates and beliefs. Our actual results could differ materially from those discussed in the forward-looking statements. See "Special note regarding forward-looking statements." Factors that could cause or contribute to these differences include those discussed below and elsewhere in this prospectus, particularly in "Risk factors."
We are a life sciences company that has developed a next generation, ultra-sensitive digital immunoassay platform that advances precision health for life sciences research and diagnostics. Our platform enables customers to reliably detect protein biomarkers in extremely low concentrations in blood, serum and other fluids that, in many cases, are undetectable using conventional, analog immunoassay technologies. It also allows researchers to define and validate the function of novel protein biomarkers that are only present in very low concentrations and have been discovered using technologies such as mass spectrometry. These capabilities provide our customers with insight into the role of protein biomarkers in human health that has not been possible with other existing technologies and enable researchers to unlock unique insights into the continuum between health and disease. We believe this greater insight will enable the development of novel therapies and diagnostics and facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention. We are currently focusing our platform on protein detection, which we believe is an area of significant unmet need and where we have significant competitive advantages. In addition to enabling new applications and insights in protein analysis, we are also developing our Simoa technology to detect nucleic acids in biological samples.
We currently sell all of our products for life science research, primarily to laboratories associated with academic and governmental research institutions, as well as pharmaceutical, biotechnology and contract research companies, through a direct sales force and support organizations in North America and Europe, and through distributors or sales agents in other select markets, including Australia, China, Japan, India, Lebanon, Singapore and South Korea. As of September 30, 2017, we had sold 161 Simoa instruments to approximately 110 customers globally. We grew our revenue from $12.2 million in 2015 to $17.6 million in 2016, an increase of 44% year over year. During the nine months ended September 30, 2017 our revenue was $16.3 million, a 49% increase over the same period in 2016.
Our instruments are designed to be used either with assays fully developed by us, including all antibodies and supplies required to run the tests, or with "homebrew" kits where we supply some of the components required for testing, and the customer supplies the remaining required elements. Accordingly, our installed instruments generate a recurring revenue stream. We believe that our recurring consumable revenue is driven by our customers' ability to extract more valuable data using our platform and to process a large number of samples quickly with little hands-on preparation.
While we expect the Quanterix SR-X reader to generate lower consumables revenue per instrument than the Simoa HD-1 Analyzer due to its lower throughput, as the installed base of the Simoa instruments increases, total consumables revenue overall is expected to increase. We believe that consumables revenue
59
should be subject to less period-to-period fluctuation than our instrument sales revenue, and will become an increasingly important contributor to our overall revenue.
As of September 30, 2017, we had cash and cash equivalents of $18.7 million. To date, we have financed our operations principally through private placements of our convertible preferred stock, borrowings from credit facilities and revenue from our commercial operations. Since inception, we have incurred net losses. Our net loss was $15.9 million and $23.2 million for the years ended December 31, 2015 and December 31, 2016, respectively, and $15.9 million and $19.6 million for the nine months ended September 30, 2016 and September 30, 2017, respectively. As of September 30, 2017, we had an accumulated deficit of $136.6 million. We expect to continue to incur significant expenses and operating losses at least through the next 24 months. We expect our expenses will increase substantially in connection with our ongoing activities as we:
- •
- expand our sales and marketing efforts to further commercialize our products;
- •
- expand our research and development efforts to improve our existing products and develop and launch new products;
- •
- hire additional personnel;
- •
- enter into collaboration arrangements, if any, or in-license other products and technologies;
- •
- add operational, financial and management information systems; and
- •
- incur increased costs as a result of operating as a public company.
Revenue
We generate product revenue from sales of our Simoa instruments and related reagents and other consumables. We currently sell our products for research use only applications and our customers are primarily laboratories associated with academic and governmental research institutions, as well as pharmaceutical, biotechnology and contract research companies. Sales of our consumables have consistently increased due to an increasing number of Simoa instruments being installed in the field, all of which require certain of our consumables to run customers' specific tests. Consumable revenue consists of sales of complete assays which are developed internally by us, plus sales of "homebrew" kits which contain all the elements necessary to run tests with the exception of the specific antibodies utilized which are separately provided by the customer.
Service and other revenue consists of testing services provided by us in our Simoa Accelerator Laboratory on behalf of certain research customers, in addition to warranty and other service-based revenue. Services provided in our Simoa Accelerator Laboratory include sample testing, homebrew assay development and custom assay development.
Collaboration and license revenue consists of revenue associated with licensing our technology to third parties and for related services.
60
The following table presents our revenue for the periods indicated (in thousands):
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Year ended December 31, |
Nine months ended September 30, |
|||||||||||
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Product revenue |
$ | 9,477 | $ | 10,601 | $ | 7,435 | $ | 10,055 | |||||
Service and other revenue |
2,515 | 5,012 | 3,330 | 5,424 | |||||||||
Collaboration and license revenue |
188 | 1,972 | 141 | 806 | |||||||||
| | | | | | | | | | | | | | |
Total revenue |
$ | 12,180 | $ | 17,585 | $ | 10,906 | $ | 16,285 | |||||
| | | | | | | | | | | | | | |
The following table reflects product revenue (in thousands) by geography and as a percentage of total product revenue, based on the billing address of our customers. North America consists of the United States, Canada and Mexico; EMEA consists of Europe, Middle East, and Africa; and Asia Pacific includes Japan, China, South Korea, Singapore, Malaysia and Australia.
| |
|
|
|
|
|
|
|
|
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | | |
| |
Year ended December 31, | Nine months ended September 30, | ||||||||||||||||||
| |
2015 | 2016 | 2016 | 2017 | ||||||||||||||||
| |
$ |
% |
$ |
% |
$ |
% |
$ |
% |
||||||||||||
| | | | | | | | | | | | | | | | | | | | | |
| |
|
|
|
|
(unaudited) |
|||||||||||||||
North America |
$ | 7,131 | 75% | $ | 6,816 | 64% | $ | 4,994 | 67% | $ | 5,446 | 54% | ||||||||
EMEA |
$ | 1,708 | 18% | $ | 2,679 | 25% | $ | 1,620 | 22% | $ | 3,132 | 31% | ||||||||
Asia Pacific |
$ | 638 | 7% | $ | 1,106 | 11% | $ | 821 | 11% | $ | 1,477 | 15% | ||||||||
| | | | | | | | | | | | | | | | | | | | | |
Total |
$ | 9,477 | 100% | $ | 10,601 | 100% | $ | 7,435 | 100% | $ | 10,055 | 100% | ||||||||
| | | | | | | | | | | | | | | | | | | | | |
Our revenue is denominated primarily in U.S. dollars. Our expenses are generally denominated in the currencies in which our operations are located, which is primarily in the United States. Changes in foreign currency exchange rates have not materially affected us to date; however, they may become material to us in the future as our operations outside of the United States expand.
Cost of products, services and collaboration revenue
Cost of goods sold for products consists of Simoa instrument cost from the manufacturer, raw material parts costs and associated freight, shipping and handling costs, contract manufacturer costs, salaries and other personnel costs, stock-based compensation, overhead and other direct costs related to those sales recognized as product revenue in the period.
Cost of goods sold for services consists of salaries and other personnel costs, stock-based compensation and facility costs associated with operating the Simoa Accelerator Laboratory on behalf of customers, in addition to costs related to warranties and other costs of servicing equipment at customer sites.
Cost of collaboration revenue consists of royalty expense due to third parties from revenue generated by collaboration or license deals.
Research and development expenses
Research and development expenses consist of salaries and other personnel costs, stock-based compensation, research supplies, third-party development costs for new products, materials for prototypes,
61
and allocated overhead costs that include facility and other overhead costs. We have made substantial investments in research and development since our inception, and plan to continue to make substantial investments in the future. Our research and development efforts have focused primarily on the tasks required to support development and commercialization of new and existing products. We believe that our continued investment in research and development is essential to our long-term competitive position and expect these expenses to increase in future periods.
Selling, general and administrative expenses
Selling, general and administrative expenses consist primarily of salaries and other personnel costs, and stock-based compensation for our sales and marketing, finance, legal, human resources and general management, as well as professional services, such as legal and accounting services. We expect selling, general and administrative expenses to increase in future periods as the number of sales, technical support and marketing and administrative personnel grows and we continue to introduce new products, broaden our customer base and grow our business. We also expect to incur additional expenses as a public company, including expenses related to compliance with the rules and regulations of the Securities and Exchange Commission and the Nasdaq Stock Market, additional insurance expenses, and expenses related to investor relations activities and other administrative and professional services.
Critical accounting policies, significant judgments and estimates
Our consolidated financial statements and the related notes included elsewhere in this prospectus are prepared in accordance with accounting principles generally accepted in the United States. The preparation of these consolidated financial statements requires us to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenue, costs and expenses and related disclosures. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances. Changes in accounting estimates may occur from period to period. Accordingly, actual results could differ significantly from the estimates made by our management. We evaluate our estimates and assumptions on an ongoing basis. To the extent that there are material differences between these estimates and actual results, our future financial statement presentation, financial condition, results of operations and cash flows will be affected.
We believe that the following critical accounting policies involve a greater degree of judgment and complexity than our other significant accounting policies. Accordingly, these are the policies we believe are the most critical to understanding and evaluating our consolidated financial condition and results of operations. Our significant accounting policies are more fully described in Note 2 of the notes to our consolidated financial statements included elsewhere in this prospectus.
Revenue recognition
We recognize revenue when (1) persuasive evidence of an arrangement exists, (2) shipment and installation, if applicable, has occurred or services have been rendered, (3) the price to the customer is fixed or determinable and (4) collection of the related receivable is reasonably assured. We primarily generate revenue from the sale of products and delivery of services, as well as under license and collaboration agreements. Our product revenue includes the sale of instruments as well as assay kits and other consumables which are used to perform tests on the instrument. Our service revenue is generated from service contracts related to research services performed on behalf of customers and maintenance and support services.
62
Product revenue
Revenue for instrument sales is recognized upon installation at the customer's location or upon transfer of title to the customer when installation is not required, which is generally the case with sales to distributors. In sales to end-customers, we always provide the installation service and often payment is tied to the completion of the installation service. When installation is required, we account for the instrument and installation service as one unit of accounting and recognize revenue when installation is completed, assuming all other revenue recognition criteria are met. Instrument transactions often have multiple elements, as discussed below. Included with the purchase of an instrument is a one-year assurance type product warranty assuring that the instrument is free of material defects and will function according to specifications. In addition, the sale of an instrument includes an implied warranty which is promised to the customer during the pre-sales process, at the time that the sales quote is issued to the customer. The implied warranty is provided over the same one-year period as the standard warranty. The services included in the implied warranty are the same as those included in the extended service contracts, and include two bi-annual preventative maintenance service visits, minor hardware updates and software upgrades, additional training and troubleshooting, which is beyond the scope of the standard product warranty. The implied warranty has been identified by us as a separate deliverable and unit of accounting. Consideration allocated to the implied one-year warranty is recognized over the one year period of performance as service and other revenue as described below. Consideration allocated to any other elements is recognized as the goods are delivered or the services are performed.
Service and other revenue
Service revenue includes revenue from the implied one-year service type warranty obligation, revenue from extended service contracts, research services performed on behalf of customers in our Simoa Accelerator Laboratory, and other services that may be performed. Revenue for extended warranty contracts is recognized ratably over the service period. Revenue for the implied one-year service type warranty is initially deferred at the time of instrument revenue recognition and is recognized ratably over a 12-month period starting on the date of instrument installation. Revenue for research and development services and other services is generally recognized based on proportional performance of the contract when our ability to complete project requirements is reasonably assured. Most of these services are completed in a short period of time from the receipt of the customer's order. When significant risk exists in our ability to fulfill project requirements, revenue is recognized upon completion of the contract.
Collaboration and license revenue
Collaboration and license revenue relates to our agreements with bioMérieux and another diagnostic company. For a complete discussion of the accounting policies specific to these collaboration and license agreements, refer to Note 11 to the consolidated financial statements included elsewhere in this prospectus.
Multiple element arrangements
Many of our instrument sales involve the delivery of multiple products and services. The elements of an instrument sale typically include the instruments, installation (when required), an implied one-year service type warranty, and in some cases, assays, consumables and other services. Revenue recognition for contracts with multiple deliverables is based on the individual units of accounting determined to exist in the contract. A delivered item is considered a separate unit of accounting when the delivered item has value to the customer on a stand-alone basis. In determining the units of accounting, management evaluates certain criteria, including whether the deliverables have standalone value. Items are considered
63
to have stand-alone value when they are sold separately by any vendor or when the customer could resell the item on a stand-alone basis.
The consideration received is allocated among the separate units of accounting using the relative selling price method, and the applicable revenue recognition criteria are applied to each of the separate units. We determine the estimated selling price for deliverables within the arrangement using vendor-specific objective evidence (VSOE) of selling price, if available. If VSOE is not available, we consider whether third-party evidence is available. If third-party evidence of selling price or VSOE is not available, we use our best estimate of selling price for the deliverable.
In order to establish VSOE of selling price, we must regularly sell the product or service on a standalone basis with a substantial majority priced within a relatively narrow range. If there are not a sufficient number of standalone sales such that VSOE of selling price cannot be determined, then we consider whether third party evidence can be used to establish selling price. Due to the lack of similar products and services sold by other companies within the industry, we have not established selling price using third-party evidence.
For product and service sales, we determine our best estimate of selling price for instruments, consumables, services and assays using average selling prices over a rolling 12-month period coupled with an assessment of market conditions, as VSOE and third-party evidence cannot be established. We recognize revenue for delivered elements only when we determine there are no uncertainties regarding customer acceptance.
Distributor transactions
In certain markets, we sell products and provide services to customers through distributors that specialize in life science products. In cases where the product is delivered to a distributor, revenue recognition generally occurs when title transfers to the distributor. The terms of sales transactions through distributors are generally consistent with the terms of direct sales to customers, except the distributors do not require our services to install the instrument at the end customer and perform the services for the customer that are beyond our standard warranty in the first year following the sale. These transactions are accounted for in accordance with our revenue recognition policy described herein.
Stock-based compensation
We account for stock-based compensation awards in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification (ASC) Topic 718, Compensation—Stock Compensation, or ASC 718. ASC 718 requires all stock-based payments to employees, including grants of employee stock options, to be recognized in the statement of operations based on their fair values. Our stock-based compensation awards have historically consisted of stock options.
Prior to adoption of ASU 2016-09 on January 1, 2017, we recognized compensation costs related to stock options granted to employees based on the estimated fair value of the awards on the date of grant, net of estimated forfeitures. Effective January 1, 2017, we ceased utilizing an estimated forfeiture rate and began recognizing forfeitures as they occur. We estimate the grant date fair value, and the resulting stock-based compensation expense, using the Black-Scholes option-pricing model. The grant date fair value of the stock-based awards is generally recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the respective awards.
We recognized compensation costs related to stock options granted to non-employees based on the estimated fair value of the awards on the date of grant in the same manner as we do for options for
64
employees; however, the fair value of the stock options granted to non-employees is re-measured each reporting period until the service is complete, and the resulting increase or decrease in value, if any, is recognized as expense or income, respectively, during the period the related services are rendered. There were no material non-employee awards outstanding during the years ended December 31, 2015 and 2016 or the nine months ended September 30, 2016 and 2017.
The Black-Scholes option-pricing model requires the use of subjective assumptions, including the expected volatility of our common stock, the assumed dividend yield, the expected term of our stock options, the risk-free interest rate for a period that approximates the expected term of our stock options, and the fair value of the underlying common stock on the date of grant. In applying these assumptions, we considered the following factors:
- •
- we do not have sufficient history to estimate the volatility of our common stock;
- •
- we calculate expected volatility based on reported data for selected similar publicly traded companies for which the historical information is
available;
- •
- we plan to continue to use the guideline peer group volatility information until the historical volatility of our common stock is sufficient to
measure expected volatility for future option grants;
- •
- the assumed dividend yield of zero is based on our expectation of not paying dividends for the foreseeable future;
- •
- we use the simplified method for determining the expected term of stock options due to the lack of historical exercise data and the plain
nature of the stock options; and
- •
- we determine the risk-free interest rate by reference to implied yields available from U.S. Treasury securities with a remaining term equal to the expected life assumed at the date of grant.
The following summarizes the assumptions we used to estimate the fair value of stock options that we granted for the periods indicated:
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Year ended December 31, | Nine months ended September 30, | |||||||||||
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Weighted-average expected volatility |
41% | 46% | 46% | 52% | |||||||||
Weighted-average risk-free rate |
1.7% | 1.2% | 1.2% | 2.0% | |||||||||
Dividend yield |
0% | 0% | 0% | 0% | |||||||||
Expected term (in years) |
6.0 | 6.0 | 6.0 | 6.0 | |||||||||
| | | | | | | | | | | | | | |
For the years ended December 31, 2015 and 2016, stock-based compensation expense was $1.1 million and $0.9 million, respectively.
For the nine months ended September 30, 2016 and 2017, stock-based compensation expense was $0.7 million and $1.4 million, respectively. As of September 30, 2017, we had $6.0 million of total unrecognized stock-based compensation costs which we expect to recognize over a weighted-average period of 3.0 years.
The following table summarizes by grant date the number of shares of our common stock subject to stock options granted from January 1, 2016 through September 30, 2017, as well as the associated per-share exercise price of the award and the estimated fair value per share of our common stock on the grant date.
65
Options granted from January 1, 2016 to September 30, 2017, substantially all of which were granted to our employees and non-employee directors:
| |
|
|
|
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | |
| Grant date |
Number of shares underlying option granted |
Exercise price per share |
Estimated fair value per share of common stock at grant date |
|||||||
| | | | | | | | | | | |
August 31, 2017 (unaudited) |
275,088 | $ | 2.93 | $ | 2.93 | |||||
June 2, 2017 (unaudited) |
969,737 | $ | 2.70 | $ | 2.70 | |||||
May 25, 2017 (unaudited) |
165,500 | $ | 2.70 | $ | 2.70 | |||||
March 31, 2017 (unaudited) |
2,562,444 | $ | 2.54 | $ | 2.70 | |||||
August 25, 2016 |
215,000 | $ | 1.69 | $ | 1.69 | |||||
June 24, 2016 |
670,500 | $ | 1.58 | $ | 1.58 | |||||
| | | | | | | | | | | |
Prior to becoming publicly traded, the fair value of our common stock underlying our stock options was estimated on each grant date by our board of directors. We have performed valuations on a quarterly basis since September 30, 2015. Awards on March 31, 2017 were issued at an exercise price equal to the most recent available valuation, as of December 31, 2016, and the grant date fair value of the awards was determined once the valuation as of March 31, 2017 was finalized. In order to determine the fair value of our common stock underlying granted stock options, our board of directors considered, among other things, the most recent valuations of our common shares prepared by an unrelated third-party valuation firm in accordance with the guidance provided by the American Institute of Certified Public Accountants Practice Guide, Valuation of Privately-Held-Company Equity Securities Issued as Compensation.
Given the absence of a public trading market for our common stock, our board of directors exercised reasonable judgment and considered a number of objective and subjective factors to determine the best estimate of the fair value of our common stock, including (1) our business, financial condition and results of operations, including related industry trends affecting our operations; (2) our forecasted operating performance and projected future cash flows discounted to present value using our estimated weighted average cost of capital; (3) the illiquid nature of our common stock; (4) liquidation preferences and other rights and privileges of our preferred stock over our common stock; (5) likeliness and estimated timing of the potential option to have our stock become publicly traded; (6) market multiples of our most comparable public peers; (7) recently completed equity financing transactions; and (8) market conditions affecting our industry.
After the closing of the offering contemplated hereby, our board of directors will determine the fair value of each common share underlying share-based awards based on the closing price of our common shares as reported by Nasdaq on the date of grant.
Based on an assumed initial public offering price of $ per share, the midpoint of the price range set forth on the cover page of this prospectus, the intrinsic value of stock options outstanding at September 30, 2017 was $ million, with $ million related to vested options and $ million related to unvested options.
66
The table below summarizes the stock-based compensation expense recognized in our statements of operation by classification (in thousands):
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Year ended December 31, |
Nine months ended September 30, |
|||||||||||
| Stock-based compensation expense |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Cost of product revenue |
$ | 6 | $ | 6 | $ | 6 | $ | 18 | |||||
Cost of service revenue |
1 | 12 | 10 | 31 | |||||||||
Research and development |
112 | 59 | 51 | 126 | |||||||||
Selling, general and administrative |
985 | 851 | 670 | 1,256 | |||||||||
| | | | | | | | | | | | | | |
Total |
$ | 1,104 | $ | 928 | $ | 737 | $ | 1,431 | |||||
| | | | | | | | | | | | | | |
Preferred stock warrant liability
As of January 1, 2015, we had outstanding warrants to purchase 64,441 shares of Series A-2 redeemable convertible preferred stock, or Series A-2 Preferred Stock, 1,300,000 shares of Series A-3 convertible preferred stock, or Series A-3 Preferred Stock, 562,488 shares of Series B redeemable convertible preferred stock, or Series B Preferred Stock, and 226,733 shares of Series C redeemable convertible preferred stock, or Series C Preferred Stock. On March 4, 2015, we issued a warrant to purchase 46,248 shares of Series C Preferred Stock to a lender related to an amendment to a debt facility. The fair value of the warrant was initially accounted for as a debt discount. On January 29, 2016, we issued a warrant to purchase 57,810 shares of Series C Preferred Stock to a lender related to a second amendment to a debt facility. The fair value of the warrant was initially accounted for as a debt discount. On November 18, 2016, we issued a warrant to purchase 700,000 shares of Series A-3 Preferred Stock to a vendor. The fair value of the warrant was recorded as research and development expense. On March 31, 2017, we issued a warrant to purchase 38,828 shares of Series D redeemable convertible preferred stock (Series D Preferred Stock) to a lender as part of a third amendment to a debt facility. The fair value of the warrant was initially accounted for as a debt discount. All of the warrants were initially recorded at fair value and marked to market on each reporting and exercise date with changes in the fair value recorded in other expense (income) on the statement of operations and comprehensive loss. Holders of warrants to purchase shares of Series A-3 and B Preferred Stock exercised the warrants during the year ended December 31, 2016 and holders of warrants to purchase shares of Series A-3 Preferred Stock exercised the warrants during the three months ended March 31, 2017. Upon exercise, the fair value of the warrants was reclassified to redeemable convertible preferred stock along with any proceeds received.
67
The changes in preferred stock warrant liability measured at fair value for which we have used Level 3 inputs to determine fair value are as follows (in thousands):
| |
|
|||
|---|---|---|---|---|
| | | | | |
| |
Warrant liability |
|||
| | | | | |
Balance at January 1, 2015 |
$ | 4,862 | ||
Issuance of warrants related to debt facility |
87 | |||
Changes in fair value of warrants |
598 | |||
| | | | | |
Balance at December 31, 2015 |
5,547 | |||
Issuance of warrants related to debt facility |
128 | |||
Issuance of warrants related to a vendor |
2,078 | |||
Changes in fair value of warrants |
307 | |||
Warrant exercises |
(5,258 | ) | ||
| | | | | |
Balance at December 31, 2016 |
2,802 | |||
Issuance of warrants related to debt facility (unaudited) |
119 | |||
Changes in fair value of warrants (unaudited) |
(62 | ) | ||
Warrant exercises (unaudited) |
(2,078 | ) | ||
| | | | | |
Balance at September 30, 2017 (unaudited) |
$ | 781 | ||
| | | | | |
The warrants are classified as liabilities because they are exercisable into shares of redeemable convertible preferred stock. On each measurement date, we utilized a Black-Scholes option pricing model to determine the fair value of the warrants and utilized various valuation assumptions based on available market data and other relevant but observable factors. Expected volatility for our redeemable convertible preferred stock was determined based on an analysis of the historical volatility of a representative group of guideline public companies, because there is currently no market for our stock and, therefore, a lack of market-based company-specific historical and implied volatility information. The expected term reflects the remaining contractual term of the warrants. The assumed dividend yield is based upon our expectation of not paying dividends in the foreseeable future. The risk-free rate is based upon the U.S. Treasury yield curve in effect at the valuation date, commensurate with the remaining contractual life of the warrants. The fair value of the underlying preferred shares was determined by management, with the assistance of a third-party valuation specialist, using a hybrid valuation method, which includes a weighted analysis of two scenarios. The first scenario is based on the completion of an initial public offering utilizing a market approach and the second scenario is based on remaining privately held utilizing either an income approach or a weighted-average of an income approach and a backsolve to a recent financing event, depending on the proximity of the financing event to the measurement date. The assumption regarding our probability of completing an initial public offering is the primary contributing factor to the changes in fair value of the common stock. See Note 2 to our consolidated financial statements appearing elsewhere in this prospectus for further details on the changes of the probability of completing an initial public offering.
68
In order to determine the fair value of each warrant to purchase preferred stock at issuance at each reporting period, the following assumptions were utilized:
| |
|
|
|
|
|
|
|
|||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | | | | |
| Balance sheet date |
Value of underlying Series D preferred stock |
Value of underlying Series C preferred stock |
Value of underlying Series B preferred stock |
Value of underlying Series A-3 preferred stock |
Value of underlying Series A-2 preferred stock |
Volatility |
Probability of an initial public offering |
|||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | |
September 30, 2017 (unaudited) |
$ | 4.30 | $ | 4.28 | N/A | N/A | $ | 3.47 | 46% | 65% | ||||||||||||
December 31, 2016 |
N/A | $ | 4.16 | N/A | $ | 2.97 | $ | 2.95 | 52% | 40% | ||||||||||||
December 31, 2015 |
N/A | $ | 3.92 | $ | 3.00 | $ | 3.00 | $ | 1.90 | 41% | 25% | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | |
Comparison of the nine months ended September 30, 2016 and September 30, 2017 (dollars in thousands):
| |
|
|
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | |
| |
Nine months ended September 30, 2016 |
% of revenue |
Nine months ended September 30, 2017 |
% of revenue |
$ change |
% change |
|||||||||
| | | | | | | | | | | | | | | | |
|
(unaudited) | ||||||||||||||
Product revenue |
$ | 7,435 | 68.2% | $ | 10,055 | 61.7% | $ | 2,620 | 35.2% | ||||||
Service and other revenue |
3,330 | 30.5% | 5,424 | 33.3% | 2,094 | 62.9% | |||||||||
Collaboration and license revenue |
141 | 1.3% | 806 | 5.0% | 665 | 471.6%* | |||||||||
| | | | | | | | | | | | | | | | |
Total revenue |
10,906 | 100.0% | 16,285 | 100.0% | 5,379 | 49.3% | |||||||||
| | | | | | | | | | | | | | | | |
Cost of product revenue |
4,501 | 41.3% | 5,573 | 34.2% | 1,072 | 23.8% | |||||||||
Cost of service revenue |
2,245 | 20.6% | 3,606 | 22.2% | 1,361 | 60.6% | |||||||||
Cost of license revenue |
0 | 0% | 0 | 0% | 0 | 0% | |||||||||
Research and development |
10,192 | 93.4% | 12,377 | 76.0% | 2,185 | 21.4% | |||||||||
Selling, general and administrative |
8,866 | 81.3% | 13,641 | 83.7% | 4,775 | 53.9% | |||||||||
| | | | | | | | | | | | | | | | |
Total operating expenses |
25,804 | 236.6% | 35,197 | 216.1% | 9,393 | 36.4% | |||||||||
| | | | | | | | | | | | | | | | |
Loss from operations |
(14,898 | ) | (136.6)% | (18,912 | ) | (116.1)% | (4,014 | ) | 26.9% | ||||||
Interest expense, net |
(1,012 |
) |
(9.3)% |
(735 |
) |
(4.5)% |
277 |
(27.4)% |
|||||||
Other income (expense), net |
51 |
0.5% |
10 |
0% |
(41 |
) |
(80.4)% |
||||||||
| | | | | | | | | | | | | | | | |
Net loss |
$ | (15,859 | ) | (145.4)% | $ | (19,637 | ) | (120.6)% | $ | (3,778 | ) | 23.8% | |||
Installed instrument base |
105 | 161 | 56 | 53.3% | |||||||||||
| | | | | | | | | | | | | | | | |
* Not meaningful.
Revenue
Revenue increased by $5.4 million, or 49%, to $16.3 million for the nine months ended September 30, 2017 as compared to $10.9 million for the same period in 2016. Product revenue consisted of sales of
69
34 instruments totaling $4.2 million and sales of consumables and other products of $3.2 million for the nine months ended September 30, 2016. Product revenue consisted of sales of 41 instruments totaling $4.9 million and sales of consumables and other products of $5.2 million for the nine months ended September 30, 2017. Average sales prices of instruments and consumables did not change materially in the nine months ended September 30, 2017 as compared with the nine months ended September 30, 2016. The increase in product revenue of $2.6 million was primarily due to the sale of seven more instruments in the nine months ended September 30, 2017 and increased sales of consumables. The installed base of Simoa instruments increased by 56 from September 30, 2016 to September 30, 2017, and as these additional instruments were used by customers, the consumable sales increased. The increase in service and other revenue of $2.1 million was due to increased services performed in our Simoa Accelerator Laboratory; more customers are using these services, and existing customers are using the Accelerator Laboratory more frequently. Collaboration and license revenue increased due to a modification to the collaboration arrangement with bioMérieux that was executed in the fourth quarter of 2016. As part of the modification, we received $2.0 million in additional consideration. This additional consideration along with the deferred revenue on the date of the modification is being recognized over our estimated period of performance, which has been initially determined to be 36 months.
Cost of product, service and license revenue
Cost of product revenue increased by $1.1 million, or 24%, to $5.6 million for the nine months ended September 30, 2017 as compared to $4.5 million for the same period in 2016. The increase was primarily due to increased sales of consumables. Cost of service revenue increased from $2.2 million in the nine months ended September 30, 2016 to $3.6 million for the nine months ended September 30, 2017. The increase was primarily due to higher utilization of the Simoa Accelerator Laboratory, plus increased personnel costs from the build out of our field service organization. Overall cost of goods sold as a percentage of revenue slightly decreased to 56.4% of total revenue for the nine months ended September 30, 2017 as compared to 61.9% in the comparable prior year period, primarily as a result of improved margins on consumables due to a reduction in costs.
Research and development expense
Research and development expense increased by $2.2 million, or 21%, to $12.4 million for the nine months ended September 30, 2017 as compared to $10.2 million for the same period in 2016. The increase was primarily due to headcount additions in research and development and the increased use of outside development firms as we increased our new product development efforts, primarily in relation to the benchtop reader that we are developing as well as other improvements we are making to our floor standing instrument.
Selling, general and administrative expense
Selling, general and administrative expense increased by $4.7 million, or 54%, to $13.6 million for the nine months ended September 30, 2017 as compared to $8.9 million for the same period in 2016. The increase was primarily due to headcount additions in various departments as we build out our organization to support future growth.
Interest and other expense, net
Interest and other expense, net decreased by $0.3 million, to $0.7 million for the nine months ended September 30, 2017 as compared to $1.0 million for the same period in 2016, primarily due to the amortization of debt discounts from warrants we have issued to a lender.
70
Comparison of the years ended December 31, 2015 and December 31, 2016 (dollars in thousands):
| |
|
|
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | |
| |
Year ended December 31, 2015 |
% of revenue |
Year ended December 31, 2016 |
% of revenue |
$ change |
% change |
|||||||||
| | | | | | | | | | | | | | | | |
Product revenue |
$ | 9,477 | 77.8% | $ | 10,601 | 60.3% | $ | 1,124 | 11.9% | ||||||
Service and other revenue |
2,515 | 20.7% | 5,012 | 28.5% | 2,497 | 99.3% | |||||||||
Collaboration and license revenue |
188 | 1.5% | 1,972 | 11.2% | 1,784 | 948.9%* | |||||||||
| | | | | | | | | | | | | | | | |
Total revenue |
12,180 | 100.0% | 17,585 | 100.0% | 5,405 | 44.4% | |||||||||
| | | | | | | | | | | | | | | | |
Cost of product revenue |
5,661 | 46.5% | 6,299 | 35.8% | 638 | 11.3% | |||||||||
Cost of service revenue |
804 | 6.6% | 3,163 | 18.0% | 2,359 | 293.4% | |||||||||
Cost of license revenue |
— | 0.0% | 375 | 2.1% | 375 | — | |||||||||
Research and development |
10,083 | 82.8% | 16,993 | 96.7% | 6,910 | 68.5% | |||||||||
Selling, general and administrative |
10,155 | 83.3% | 12,466 | 70.9% | 2,311 | 22.8% | |||||||||
| | | | | | | | | | | | | | | | |
Total operating expenses |
$ | 26,703 | 219.2% | $ | 39,296 | 223.5% | $ | 12,593 | 47.2% | ||||||
| | | | | | | | | | | | | | | | |
Loss from operations |
(14,523 | ) | (119.2)% | (21,711 | ) | (123.5)% | (7,188 | ) | 49.5% | ||||||
Interest expense, net |
(1,040 | ) | (8.6)% | (1,298 | ) | (7.4)% | (258 | ) | 24.8% | ||||||
Other income (expense), net |
(380 | ) | (3.1)% | (164 | ) | (0.9)% | 216 | (56.8)% | |||||||
| | | | | | | | | | | | | | | | |
Net loss |
$ | (15,943 | ) | (130.9)% | $ | (23,173 | ) | (131.8)% | $ | (7,230 | ) | 45.4% | |||
Installed instrument base |
71 | 120 | 49 | 69.0% | |||||||||||
| | | | | | | | | | | | | | | | |
* Not meaningful.
Revenue
Revenue increased by $5.4 million, or 44%, to $17.6 million for the year ended December 31, 2016 as compared to $12.2 million for the year ended December 31, 2015. Product revenue consisted of sales of 49 instruments totaling $6.2 million and consumable and assay revenue of $4.4 million for the year ended December 31, 2016. Product revenue consisted of sales of 50 instruments totaling $6.5 million and consumable and assay revenue of $3.0 million for the year ended December 31, 2015. Average sales price of instruments and consumables did not change materially in the year ended December 31, 2016 as compared with the year ended December 31, 2015. The increase in product revenue of $1.1 million was primarily due to increased sales of consumables of $1.5 million due to having an increased installed base of Simoa instruments as a result of the sale of 49 instruments during 2016. This was partially offset by decreased revenue related to the sale of one less instrument during the year ended December 31, 2016 compared to the year ended December 31, 2015 due to timing of customer orders. The increase in service and other revenue of $2.5 million was primarily due to increased utilization of our Simoa Accelerator Laboratory, plus increased warranty revenues. The increase in collaboration and license revenue was primarily due to the execution of a license with a diagnostic company in 2016 which resulted in the recognition of $1.8 million of revenue in 2016.
Cost of product, service and license revenue
Cost of product revenue increased by $0.6 million, or 11%, to $6.3 million for the year ended December 31, 2016 as compared to $5.7 million for the year ended December 31, 2015. The increase was primarily due to increased sales of instruments and consumables. Cost of service revenue increased from $0.8 million for
71
the year ended December 31, 2015 to $3.2 million for the year ended December 31, 2016. The increase was primarily due to higher utilization of our Simoa Accelerator Laboratory and a significant increase in the staffing of our field service team. Cost of license revenue was $0.4 million for the year ended December 31, 2016 versus $0 in the prior year due to a royalty payment that we are required to pay Tufts, a related party, as a result of the license with a diagnostic company and a modification to our bioMérieux collaboration agreement. Overall cost of goods sold as a percentage of revenue increased to 55.9% of revenue for the year ended December 31, 2016 as compared to 53.1% in the comparable prior year period, primarily as a result of lower gross margins on service and other revenue due to the increase in staffing as noted previously.
Research and development expense
Research and development expense increased by $6.9 million, or 69%, to $17.0 million for the year ended December 31, 2016 as compared to $10.1 million for the same period in 2015. The increase was primarily due to increases in salary and other compensation costs from increases in research and development headcount and increased use of outside development firms as we increased our new product development efforts, primarily in regards to our Quanterix SR-X instrument which is currently under development.
Selling, general and administrative expense
Selling, general and administrative expense increased by $2.3 million, or 23%, to $12.5 million for the year ended December 31, 2016 as compared to $10.2 million for the year ended December 31, 2015. The increase was primarily due to headcount additions in various departments as we build out our organization to support future growth.
Interest and other expense, net
Interest and other expense, net increased by $0.1 million to $1.5 million for the year ended December 31, 2016 as compared to $1.4 million for the year ended December 31, 2015, primarily due to higher interest expense related to the amortization of debt discounts and higher average borrowings.
Liquidity and capital resources
Since our inception, we have incurred net losses and negative cash flows from operations. We incurred net losses of $15.9 million, $23.2 million and $19.6 million, and used $12.5 million, $17.7 million and $17.7 million of cash from our operating activities for the years ended December 31, 2015 and 2016 and the nine months ended September 30, 2017, respectively. As of September 30, 2017, we had an accumulated deficit of $136.6 million.
As of September 30, 2017, we had cash and cash equivalents of $18.7 million. In addition, our debt facility was amended in March 2017 to increase the amount of the facility by $5 million.
Sources of liquidity
To date, we have financed our operations principally through private placements of our convertible preferred stock, borrowings from credit facilities and revenue from our commercial operations.
Preferred stock financings
As of December 31, 2016, we had raised approximately $99 million in gross proceeds through sales of our preferred stock, including the sale of 12,420,262 shares of our Series D redeemable convertible preferred
72
stock, or Series D Preferred Stock, in March 2016 at a purchase price of $3.67 per share for gross proceeds of $45.6 million.
In June 2017, we also issued 2,113,902 shares of Series D-1 Preferred Stock at a purchase price of $4.021 per share for gross proceeds of $8.5 million.
See Note 7 to our consolidated financial statements for a discussion of the terms and provisions of our Series D and Series D-1 Preferred Stock issued in 2016 and 2017.
Loan facility with Hercules
On April 14, 2014, we executed a Loan Agreement with Hercules Capital, Inc. (formerly known as Hercules Technology Growth Capital, Inc.). The Loan Agreement provided a total debt facility of $10.0 million, which is secured by substantially all of our assets. At closing, we borrowed $5.0 million in principal and had the ability to draw the additional $5.0 million over the period from November 1, 2014 to March 31, 2015. The interest rate on this term loan was variable based on a calculation of 8% plus the prime rate less 5.25%, with a minimum interest rate of 8%. Interest was to be paid monthly beginning the month following the borrowing date. Principal payments were scheduled to begin on September 1, 2015, unless we achieved certain milestones which would have extended this date to December 1, 2015 or March 1, 2016. In connection with the execution of the Loan Agreement, we issued Hercules a warrant to purchase up to 173,428 shares of our Series C Preferred Stock at an exercise price of $3.3299 per share.
On March 4, 2015, we executed Amendment 1 to the Loan Agreement and drew the additional $5.0 million available under the Loan Agreement at that time. The terms of the amendment deferred principal payments to start on December 1, 2015 or March 1, 2016 if we obtained at least $10.0 million in equity financing before December 1, 2015. This equity financing did not occur before December 1, 2015.
In January 2016, we executed Amendment 2 to the Loan Agreement, which increased the total facility available by $5.0 million to a total of $15.0 million and further delaying the start of principal payments to July 1, 2016. Following the Series D Preferred Stock financing in March 2016, we could have elected to further delay the start of principal payments until January 1, 2017, however we voluntarily began paying principal on July 1, 2016. Upon signing this amendment, we drew an additional $3.0 million under the debt facility. The remaining $2.0 million available for borrowing expired unused in 2016, decreasing the amounts available under the debt facility to $13.0 million.
In March 2017, we signed Amendment 3 to the Loan Agreement increasing the total facility available by $5.0 million to a total of $18.0 million. Additionally, the lender is providing an optional term loan, solely at the lender's discretion, for an incremental $5.0 million, increasing the total potential facility to $23.0 million. As of September 30, 2017 we have not drawn any of this additional facility. Principal payments are delayed to March 1, 2018 and the loan maturity date was extended to March 1, 2019. The start of principal payments may be further delayed until September 3, 2018 if a milestone event is achieved, whereby must achieve $15.0 million in revenue over the trailing nine-month period commencing in April 2017. The amendment did not affect the due date of the existing end of term fees (in aggregate $0.5 million) which remain due on February 1, 2018. Upon signing Amendment 3 to the Loan Agreement, we did not draw any of the additional amounts available under the amended debt facility and no amounts have been subsequently drawn under the facility. In connection with this amendment, we issued Hercules a warrant to purchase up to 38,828 shares of our Series D Preferred Stock at an exercise price of $3.67 per share.
The Loan Agreement and amendments contain end of term payments and are recorded in the debt accounts. $0.5 million of end of term payments are due in the first quarter of 2018.
73
The Loan Agreement contains negative covenants restricting our activities, including limitations on dispositions, mergers or acquisitions, incurring indebtedness or liens, paying dividends or making investments and certain other business transactions. There are no financial covenants associated with the Loan Agreement. The obligations under the Loan Agreement are subject to acceleration upon the occurrence of specified events of default, including a material adverse change in our business, operations or financial or other condition, which is subjective in nature. We have determined that the risk of subjective acceleration under the material adverse events clause is not probable and therefore have classified the outstanding principal in current and long-term liabilities based on scheduled principal payments.
Debt principal repayments, including the end of term fees, due as of December 31, 2016 (reflecting changes in the principal payment schedule resulting from the amendment in March 2017) are (in thousands):
| |
|
|||
|---|---|---|---|---|
| | | | | |
| Years ending December 31: |
|
|||
| | | | | |
2017 |
$ | 921 | ||
2018 |
5,133 | |||
2019 |
4,430 | |||
| | | | | |
|
$ | 10,484 | ||
| | | | | |
Cash flows
The following table presents our cash flows for each period presented (in thousands):
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Year ended December 31, | Nine months ended September 30, |
|||||||||||
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Net cash used in operating activities |
$ | (12,517 | ) | $ | (17,742 | ) | $ | (13,554 | ) | $ | (17,704 | ) | |
Net cash used in investing activities |
(554 | ) | (826 | ) | (390 | ) | (793 | ) | |||||
Net cash provided by financing activities |
11,704 | 45,916 | 47,120 | 7,516 | |||||||||
| | | | | | | | | | | | | | |
Net increase (decrease) in cash and cash equivalents |
$ | (1,367 | ) | $ | 27,348 | $ | 33,176 | $ | (10,981 | ) | |||
| | | | | | | | | | | | | | |
Net cash used in operating activities
We derive cash flows from operations primarily from the sale of our products and services. Our cash flows from operating activities are also significantly influenced by our use of cash for operating expenses to support the growth of our business. We have historically experienced negative cash flows from operating activities as we have developed our technology, expanded our business and built our infrastructure and this may continue in the future.
Net cash used in operating activities was $17.7 million during the nine months ended September 30, 2017. Net cash used in operating activities primarily consisted of net loss of $19.6 million, primarily offset by a $1.7 million increase in deferred revenue.
Net cash used in operating activities was $13.6 million during the nine months ended September 30, 2016. Net cash used in operating activities primarily consisted of net loss of $15.9 million, primarily offset by a $1.7 million increase in deferred revenue.
74
Net cash used in operating activities was $17.7 million during the year ended December 31, 2016. Net cash used in operating activities primarily consisted of a net loss of $23.2 million and an increase in accounts receivable of $1.7 million, primarily offset by non-cash charges related to issuance of warrants of $2.1 million, other non-cash items including depreciation and stock based compensation of $1.8 million and increases in current liabilities of $2.3 million and deferred revenue of $0.9 million.
Net cash used in operating activities was $12.5 million during the year ended December 31, 2015. Net cash used in operating activities primarily consisted of a net loss of $15.9 million and an increase in accounts receivable of $1.5 million, primarily offset by non-cash items including depreciation and stock based compensation expense of $1.8 million and increases in current liabilities of $1.8 million and deferred revenue of $0.8 million.
Net cash used in investing activities
Historically, our primary investing activities have consisted of capital expenditures for the purchase of capital equipment to support our expanding infrastructure and work force. We expect to continue to incur additional costs for capital expenditures related to these efforts in future periods.
We used $0.8 million of cash in investing activities during the nine months ended September 30, 2017 for purchases of capital equipment to support our infrastructure.
We used $0.4 million of cash in investing activities during the nine months ended September 30, 2016 for purchases of capital equipment to support our infrastructure.
We used $0.8 million of cash in investing activities during the year ended December 31, 2016 primarily for purchases of capital equipment to support our infrastructure, and for a $0.3 million equity investment in another company.
We used $0.6 million of cash in investing activities during the year ended December 31, 2015 for purchases of capital equipment to support our infrastructure.
Net cash provided by (used in) financing activities
Historically, we have financed our operations principally through private placements of our convertible preferred stock and borrowings from credit facilities, as well as revenues from our commercial operations.
We generated $7.5 million of cash in financing activities during the nine months ended September 30, 2017, which primarily was from the sale of 2,113,902 shares of our Series D-1 Preferred Stock in June 2017 for net proceeds of $8.4 million, which was partially offset by payments of outstanding debt.
We generated $47.1 million of cash from financing activities during the nine months ended September 30, 2016, which was from the sale of 12,420,262 shares of our Series D Preferred Stock in March 2016 for net proceeds of $45.4 million, plus $3.0 million in additional borrowings, partially offset by debt payments of $1.3 million.
We generated $45.9 million of cash from financing activities during the year ended December 31, 2016, which was primarily from the sale of our Series D Preferred Stock in March 2016 for net proceeds of $45.4 million.
We generated $11.7 million of cash from financing activities during the year ended December 31, 2015, which was primarily from the sale of preferred stock of $7.0 million plus an increase in long-term debt of $4.7 million, net of principal payments.
75
Capital resources
We have not achieved profitability on a quarterly or annual basis since our inception, and we expect to continue to incur net losses in the future. We also expect that our operating expenses will increase as we continue to increase our marketing efforts to drive adoption of our commercial products. Additionally, as a public company, we will incur significant audit, legal and other expenses that we did not incur as a private company. Our liquidity requirements have historically consisted, and we expect that they will continue to consist, of sales and marketing expenses, research and development expenses, working capital, debt service and general corporate expenses.
We believe the net proceeds from this offering, together with the cash generated from commercial sales, our current cash and cash equivalents, and interest income we earn on these balances will be sufficient to meet our anticipated operating cash requirements for at least the next 24 months. In the future, we expect our operating and capital expenditures to increase as we increase headcount, expand our sales and marketing activities and grow our customer base. Our estimates of the period of time through which our financial resources will be adequate to support our operations and the costs to support research and development and our sales and marketing activities are forward-looking statements and involve risks and uncertainties and actual results could vary materially and negatively as a result of a number of factors, including the factors discussed in the section "Risk factors" of this prospectus. We have based our estimates on assumptions that may prove to be wrong and we could utilize our available capital resources sooner than we currently expect. Our future funding requirements will depend on many factors, including:
- •
- market acceptance of our products, including our Quanterix SR-X benchtop reader that we expect to launch commercially in 2018;
- •
- the cost and timing of establishing additional sales, marketing and distribution capabilities;
- •
- the cost of our research and development activities;
- •
- the success of our existing collaborations and our ability to enter into additional collaborations in the future;
- •
- the cost and timing of potential regulatory clearances or approvals that may be required in the future for our products; and
- •
- the effect of competing technological and market developments.
We cannot assure you that we will be able to obtain additional funds on acceptable terms, or at all. If we raise additional funds by issuing equity or equity-linked securities, our stockholders may experience dilution. Future debt financing, if available, may involve covenants restricting our operations or our ability to incur additional debt. Any debt or equity financing that we raise may contain terms that are not favorable to us or our stockholders. If we raise additional funds through collaboration and licensing arrangements with third parties, it may be necessary to relinquish some rights to our technologies or our products, or grant licenses on terms that are not favorable to us. If we do not have or are not able to obtain sufficient funds, we may have to delay development or commercialization of our products. We also may have to reduce marketing, customer support or other resources devoted to our products or cease operations.
76
Off-balance sheet arrangements
We did not have, during the periods presented, and we do not currently have, any off-balance sheet arrangements, as defined under applicable SEC rules.
Contractual obligations, commitments and contingencies
The following table summarizes our contractual obligations as of December 31, 2016 (in thousands):
| |
|
|
|
|
|
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | |
| |
Payments due by period |
|||||||||||||||
| (in thousands) |
Less than 1 Year |
1 to 3 years |
3 to 5 years |
More than 5 years |
Total |
|||||||||||
| | | | | | | | | | | | | | | | | |
Contractual Obligations:(1) |
||||||||||||||||
Operating lease obligations |
$ | 1,124 | $ | 2,956 | $ | 0 | $ | 0 | $ | 4,080 | ||||||
Principal payments and end of term fees on the term loan |
$ | 921 | $ | 9,563 | $ | 0 | $ | 0 | $ | 10,484 | ||||||
Total |
$ |
2,045 |
$ |
12,519 |
$ |
0 |
$ |
0 |
$ |
14,564 |
||||||
| | | | | | | | | | | | | | | | | |
(1) See "—Development and supply agreement" for additional contractual obligations.
Our operating lease obligations primarily relate to leases for our current headquarters in Lexington, Massachusetts.
We also have ongoing obligations related to license agreements which contain immaterial minimum annual payments that are credited against the actual royalty expense.
Purchase orders or contracts for the purchase of supplies and other goods and services are not included in the table above. We are not able to determine the aggregate amount of such purchase orders that represent contractual obligations, as purchase orders may represent authorizations to purchase rather than binding agreements. Our purchase orders are based on our current procurement or development needs and are fulfilled by our vendors within short time horizons.
Development and supply agreement
We do not have significant agreements for the purchase of supplies or other goods specifying minimum quantities or set prices that exceed our expected requirements for the next three to six months, with the exception of the agreement with STRATEC, who manufactures our HD-1 system. In 2013, we entered into a supply agreement, or the Supply Agreement, with STRATEC which requires us to purchase a minimum number of commercial units over a seven-year period ending in May 2021. We could be obligated to pay a fee based on the shortfall of commercial units purchased compared to the required number. Based on the commercial units purchased as of December 31, 2016, assuming no additional commercial units were purchased thereafter but prior to May 2021, this fee would equal $12.9 million. The amount we could be obligated to pay under the minimum purchase commitment is reduced as each commercial unit is purchased. We believe that we will purchase sufficient units to meet the requirements of the minimum purchase commitment and, therefore, have not accrued for any of the minimum purchase commitment.
Also, if we terminate the Supply Agreement under certain circumstances and do not purchase up to a required number of commercial units, we would be required to issue warrants to purchase 300,000 shares of Series A-3 Preferred Stock, or the Supply Warrants, at $0.001 per share. We believe that we will not
77
issue such warrant and therefore have not recorded any amounts related to the potential equity consideration.
In August 2011, we entered into a Strategic Development Services and Equity Participation Agreement, or the Development Agreement, with STRATEC, pursuant to which STRATEC undertook the development of the Simoa HD-1 instrument for manufacture and sale to us or a partner whom we designate. During the year ended December 31, 2016, the Development Agreement was amended to modify the deliverables related to the final milestone, to agree on instrument design changes to be implemented, and to reduce the minimum purchase commitment in the Supply Agreement. The reduction in the minimum purchase commitment did not affect the fee that would be payable based on the units purchased as of December 31, 2016, assuming no additional units were purchased prior to May 2021.
Additionally, the parties agreed on additional development services for a total fee of $1.5 million, which is payable when development is completed. This amount includes the final milestone payment that was due under the terms of the original agreement.
We generally expect to ship all orders received in a given period and as a result our backlog at the end of any period is typically insignificant.
Quantitative and qualitative disclosures about market risk
Market risk represents the risk of loss that may impact our financial position due to adverse changes in financial market prices and rates. Our market risk exposure is primarily a result of fluctuations in foreign currency exchange rates and interest rates. We do not hold or issue financial instruments for trading purposes.
Foreign currency exchange risk
As we expand internationally our results of operations and cash flows will become increasingly subject to fluctuations due to changes in foreign currency exchange rates. Historically, the substantial majority of our revenue has been denominated in U.S. dollars. Our expenses are generally denominated in the currencies in which our operations are located, which is primarily in the United States, with a portion of expenses incurred in Canada, Europe, Japan and China. Our results of operations and cash flows are, therefore, subject to fluctuations due to changes in foreign currency exchange rates. Fluctuations in currency exchange rates could harm our business in the future. The effect of a 10% adverse change in exchange rates on foreign denominated cash, receivables and payables as of September 30, 2017 would not have been material.
To date, we have not entered into any material foreign currency hedging contracts although we may do so in the future.
Interest rate sensitivity
We had cash and cash equivalents of $18.7 million as of September 30, 2017. These amounts were held primarily in cash on deposit with banks. Due to the short-term nature of these investments, we believe that we do not have any material exposure to changes in the fair value of our investment portfolio as a result of changes in interest rates. Declines in interest rates, however, will reduce future investment income. If overall interest rates had decreased by 10% during the periods presented, our interest income would not have been materially affected.
78
As of September 30, 2017, the principal amount of our term debt outstanding with Hercules was $9.0 million. If overall interest rates had increased by 10% during the periods presented, our interest expense would have increased by approximately $0.8 million on an annualized basis.
JOBS Act: emerging growth company status
We are an "emerging growth company," as defined in the Jumpstart Our Business Startups Act, or JOBS Act. Under the JOBS Act, emerging growth companies can delay adopting new or revised accounting standards until such time as those standards apply to non-public companies. We have elected to take advantage of the extended transition period afforded by the JOBS Act for the implementation of new or revised accounting standards and, as a result, will comply with new or revised accounting standards not later than the relevant dates on which adoption of such standards is required for non-public companies.
For so long as we are an emerging growth company we expect that:
- •
- we will present only two years of audited financial statements, plus unaudited condensed financial statements for any interim period, and
related management's discussion and analysis of financial condition and results of operations in our initial registration statement;
- •
- we will avail ourselves of the exemption from the requirement to obtain an attestation and report from our auditors on the assessment of our
internal control over financial reporting pursuant to the Sarbanes-Oxley Act;
- •
- we will avail ourselves of the extended transition periods available to emerging growth companies under the JOBS Act for complying with new or
revised accounting standards; and
- •
- we will provide less extensive disclosure about our executive compensation arrangements.
We will remain an emerging growth company for up to five years, although we will cease to be an "emerging growth company" upon the earliest of: (1) the last day of the fiscal year following the fifth anniversary of this offering, (2) the last day of the first fiscal year in which our annual revenues are $1 billion or more, (3) the date on which we have, during the previous rolling three-year period, issued more than $1 billion in non-convertible debt securities, and (4) the date on which we are deemed to be a "large accelerated filer" as defined in the Exchange Act.
79
We are a life sciences company that has developed a next generation, ultra-sensitive digital immunoassay platform that advances precision health for life sciences research and diagnostics. Our platform enables customers to reliably detect protein biomarkers in extremely low concentrations in blood, serum and other fluids that, in many cases, are undetectable using conventional, analog immunoassay technologies. It also allows researchers to define and validate the function of novel protein biomarkers that are only present in very low concentrations and have been discovered using technologies such as mass spectrometry. These capabilities provide our customers with insight into the role of protein biomarkers in human health that has not been possible with other existing technologies and enable researchers to unlock unique insights into the continuum between health and disease. We believe this greater insight will enable the development of novel therapies and diagnostics and facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention. We are currently focusing our platform on protein detection, which we believe is an area of significant unmet need and where we have significant competitive advantages. In addition to enabling new applications and insights in protein analysis, we are also developing our Simoa technology to detect nucleic acids in biological samples.
Our platform is based on our proprietary digital single molecule array, or Simoa, detection technology, which is the most sensitive commercially available protein detection technology. Simoa significantly advances ELISA technology, which has been the industry standard for protein detection for over forty years. Proteins are complex molecules that are required for the structure, function and regulation of the body's tissues and organs, and are the functional units that carry out specific tasks in every cell. The human body contains approximately 20,000 genes, each of which can produce multiple proteins. It is estimated that these 20,000 genes can produce over 100,000 different proteins, approximately 10,500 of which are known to be secreted in blood. Accordingly, while research on nucleic acids provides valuable information about the role of genes in health and disease, proteins are more prevalent and, we believe, more relevant to a precise understanding of the nuanced continuum between health and disease. Protein measurement goes beyond genetic predisposition, reflecting the impact of a range of influences on health, including environmental factors and lifestyle, providing deeper and more relevant insight into what is happening in a person's body in real time.
Researchers and clinicians rely extensively on protein biomarkers for use as research and clinical tools. However, normal physiological levels of many proteins are not detectable using conventional, analog immunoassay technologies, and many of these technologies can only detect proteins once they have reached levels that reflect more advanced disease or injury. For many other low abundance proteins, these technologies cannot detect proteins even at disease- or injury-elevated levels. We believe that Simoa's sensitivity offers a new way to monitor healthy individuals and detect proteins associated with nascent disease or injury early in the disease cascade, which holds the key to intervention before disease or injury has advanced to the point where more significant clinical signs and symptoms have appeared.
Our Simoa platform has achieved significant scientific validation and commercial adoption. Simoa has been cited by published research in approximately 160 articles in peer-reviewed publications covering over 150 biomarkers in areas of high unmet medical need and research interest such as neurology, oncology, cardiology, infectious disease and inflammation. Our growing customer base is comprised of over 200 customers across our end markets, and includes 17 of the 20 largest biopharmaceutical companies.
80
Our Simoa technology has applications across the life science research, diagnostics and precision health screening markets. Our initial target market has been the life science research market, and all of our product and service revenue to date has been in this market. While we have received revenue from upfront and milestone payments related to collaborations with diagnostic companies, neither we nor any of our diagnostic partners have sold Simoa products or services in the diagnostics or precision health screening markets. As our customers continue to gain experience with our proprietary Simoa technology, we believe the opportunity to access markets beyond research will be significant. According to estimates in the Third-Party Research Report, we believe the future aggregate market opportunity for us and others using our Simoa technology has the potential to expand to approximately $38 billion, approximately $30 billion of which would be addressable by Simoa upon receipt of the necessary regulatory approvals to market our products in areas other than life science research, which we have not yet begun the process to obtain.
Life science research
Our initial target market is the large and growing life science research market. We believe Simoa is well-positioned to capture a significant share of this market because of its superior sensitivity, automated workflow capabilities, multiplexing and its ability to work with a broader range of sample types. By substantially lowering the limit of detection of protein biomarkers, we believe that Simoa is penetrating the existing market for protein analysis and holds potential to significantly grow the life science research market as researchers expand their research into the diseases associated with the thousands of proteins that were previously undetectable. Simoa also enables earlier detection of the proteins that are currently detectable by other technologies only after they have reached levels that reflect more advanced disease or injury. As an indication of the market's acceptance of our Simoa platform, biopharmaceutical researchers are also integrating our platform into drug development protocols to more efficiently and effectively develop drugs. In addition to enabling new applications and insights in protein analysis, our Simoa technology can be used to detect nucleic acids, which expands our market opportunity. We believe that Simoa has the potential to ultimately provide the same sensitivity as polymerase chain reaction, or PCR, which is the most commonly used technology for nucleic acid detection, without the distortion and bias issues associated with amplification used in PCR.
According to estimates in the Third-Party Research Report, we believe that the total life science research market addressable by Simoa, including both proteomics and genomics research, is $3 billion per year and has the potential to reach $8 billion per year.
Diagnostics
The diagnostic market represents a significant commercial opportunity for Simoa as well. We believe existing diagnostics can be improved by Simoa's sensitivity to enable earlier detection of diseases and injuries, and that new diagnostics may be developed using protein biomarkers that are not detectable using conventional, analog immunoassay technologies but are detectable using Simoa. We also believe that the ultra-sensitive protein detection provided by Simoa can enable the development of a new category of non-invasive diagnostic tests and tools based on blood, serum, saliva and other fluids that have the potential to replace current more invasive, expensive and inconvenient diagnostic methods, including spinal tap, diagnostic imaging and biopsy. In order to accelerate the use of our technology to develop applications in the diagnostic market, we have entered into a collaboration agreement with bioMérieux, a leading diagnostic company.
81
Simoa also has significant potential in the emerging field of companion diagnostics. Drug developers can use Simoa to stratify patients into categories, enabling selection of those patients for whom a drug is expected to be most effective and safe. Not only can Simoa be used to develop companion diagnostics to stratify patients in clinical trials and for treatment, but Simoa's sensitivity also enables the development of companion diagnostics based on protein biomarkers that can regularly monitor whether an approved drug is having the desired biological effect, enabling doctors to quickly and efficiently adjust the course of treatment as appropriate.
Precision health screening
Simoa's ability to detect and quantify normal physiological levels of proteins in low abundance that are undetectable using conventional, analog immunoassay technologies may enable our technology to be used to monitor protein biomarker levels of seemingly healthy, asymptomatic people, and potentially to signal and provide earlier detection of the onset of disease. We believe there is the potential for a number of neurological, cardiovascular, oncologic and other protein biomarkers associated with disease to be measured with a simple blood draw on a regular, ongoing basis as part of a patient's routine health screening, and for those results to be compared periodically with baseline measurements to predict or detect the early onset of disease, prior to the appearance of symptoms.
According to estimates in the Third-Party Research Report, we believe that the total diagnostic and precision health screening markets addressable by us and others using Simoa have the potential to reach an aggregate of $30 billion per year, which would be addressable upon receipt of the necessary regulatory approvals to market our products in areas other than life science research, which we have not yet begun the process to obtain.
Products sold by us or collaborators in the diagnostics and precision health screening markets will be subject to regulation by the FDA or comparable international agencies, including requirements for regulatory clearance or approval of such products before they can be marketed. To date, neither we nor any of our diagnostic partners have received or applied for regulatory approvals for Simoa products. See "Risk factors—Risks related to governmental regulation and diagnostic product reimbursement" and "—Government regulation" for a more detailed discussion regarding the regulatory approvals that may be required.
Our proprietary Simoa technology is based on traditional enzyme-linked immunosorbent assay, or ELISA, technology, which has been the most widely used method of detection of proteins for over 40 years. Given our target customers' familiarity with the core ELISA technology, we believe this offers us a significant competitive advantage. Simoa differs, however, from conventional ELISA in its ability to trap single molecules in tiny microwells, 40 trillionths of a milliliter, that are 2.5 billion times smaller than traditional ELISA wells, allowing for an analysis and digital readout of each individual molecule, which is not possible with conventional ELISA technology. This ability is the key to Simoa's unprecedented sensitivity.
We commercially launched our Simoa HD-1 Analyzer in January 2014. The HD-1 Analyzer is the most sensitive protein detection platform commercially available, and is currently capable of analyzing up to six biomarkers per test, with anticipated expansion capability to up to 35 biomarkers per test in 2018. Assays run on the HD-1 Analyzer are also fully automated. We believe that the increased multiplexing capability and the full automation of the HD-1 Analyzer provides us with an additional significant competitive advantage with biopharmaceutical customers. We have currently developed more than 80 Simoa digital biomarker assays. The Simoa platform also allows ease and flexibility in assay design, enabling our
82
customers to develop their own in-house assays, called "homebrew" assays. We intend to continue to increase the number of Simoa digital biomarker assays. We have sold more than 160 HD-1 Analyzers to over 100 customers around the world. We also have seven HD-1 Analyzers in our own Simoa Accelerator Laboratory.
We continually seek to expand our product offerings to meet the needs of our customers. To that end, we have developed a new instrument, the Quanterix SR-X, which we plan to introduce through an early adopter program. We plan to initiate our commercial launch of this new instrument in 2018. The Quanterix SR-X will utilize the same core Simoa technology and assay kits as the HD-1 Analyzer in a compact benchtop form with a lower price point, more flexible assay preparation, and a wider range of applications, including direct detection of nucleic acids. The SR-X will support detection capability of up to six biomarkers per test at launch, with anticipated expansion to capability of up to 35 biomarkers per test in 2018.
We also provide contract research services for customers through our Simoa Accelerator Laboratory. The Simoa Accelerator Laboratory provides customers with access to Simoa's technology, and supports multiple projects and services, including sample testing, homebrew assay development and custom assay development. To date, we have completed over 340 projects for more than 145 customers from all over the world using our Simoa platform. In addition to being an important source of revenue, we have also found the Simoa Accelerator Laboratory to be a significant catalyst for placing additional instruments, as over 35 customers for whom we have provided contract research services have subsequently purchased an instrument from us.
We sell our instruments, consumables and services to the life science, pharmaceutical and diagnostics industries through a direct sales force and support organizations in North America and Europe, and through distributors or sales agents in other select markets. We have an extensive base of customers in world class academic and governmental research institutions, as well as pharmaceutical, biotechnology and contract research companies, using our technology to gather information to better understand human health. Our Simoa platform has enabled us and our customers to publish research in approximately 160 articles in peer-reviewed publications, covering over 150 biomarkers in areas of high unmet medical need, including neurology, oncology, cardiology, infectious disease and inflammation.
We believe that our competitive strengths include the following:
- •
- Proprietary ultra-sensitive digital immunoassay Simoa technology platform, enabling researchers and clinicians to obtain information from less invasive procedures in smaller sample sizes
Simoa is the most sensitive commercially available protein detection technology, and can detect and quantify proteins of clinical interest that are undetectable using conventional, analog immunoassay technologies. This sensitivity allows researchers to measure critical protein biomarkers at earlier stages in the progression of a disease or injury, which we believe will enable the development of novel therapies and diagnostics and facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention. The sensitivity of our Simoa technology allows researchers to gather biomarker information from smaller samples that can be collected less invasively than samples required by other assay technologies.
83
- •
- Technology platform that leverages and improves upon industry standard ELISA technology
Simoa uses the basic principles of conventional bead-based ELISA. However, unlike ELISA, which runs the enzyme-substrate reactions on all molecules in one well, Simoa reactions are run on individual molecules in tiny microwells that are 2.5 billion times smaller than traditional ELISA wells. Adding digital capability to this industry standard platform has resulted in expanded capabilities and improved performance. Given our target customers' familiarity with the core ELISA technology, Simoa is easily integrated with existing customer workflows including data analysis.
- •
- Leader in large and growing market for detecting proteins in low abundance
Simoa is the most sensitive commercially available protein detection technology. We believe our growing market acceptance is establishing Simoa as the reference platform for detecting proteins in low abundance across sample types in our end markets.
- •
- Deep and expanding scientific validation
Our Simoa technology has been cited in approximately 160 articles in peer-reviewed publications, including JAMA Neurology and Nature, covering over 150 biomarkers, and is becoming a vital tool in cutting edge life sciences research. Our company has established relationships with key opinion leaders, and our growing base of over 200 customers includes some of the world's leading academic and government research institutions as well as 17 of the 20 largest pharmaceutical and biotechnology companies.
- •
- Leading position in market solidified by robust customization capabilities, assay design flexibility and automation of our HD-1 Analyzer
Our technical capabilities and expertise allow our customers to design high-quality, customized assays utilizing our Simoa platform. The needs of our customers vary widely, and the flexibility of the Simoa detection technology allows us to provide innovative, low cost solutions for customers in multiple markets across various applications. In addition, the Simoa HD-1 Analyzer provides fully automated analysis from sample introduction to analytical results. Furthermore, our proprietary array approach to ELISA digitization enables rapid digital data acquisition and assay results. This automation and speed provides customers high research and development productivity through greater throughput and lab efficiency.
- •
- Highly attractive business model that leverages growing installed base of instruments
We have sold more than 160 HD-1 Analyzers to over 100 customers around the world and plan to commercially launch our latest instrument, the Quanterix SR-X, in 2018. As we continue to grow our installed base, optimize workflows and expand our assay menu, we expect to increase our revenues derived from consumables. The integration of our technology in our customers' projects also provides ongoing sales of assays and consumables, resulting in a growing revenue stream.
- •
- Our highly experienced senior management team
We are led by a dedicated and highly experienced senior management team with significant industry experience and proven ability to develop novel solutions. Each of the members of our senior management has more than 20 years of relevant experience.
Our goal is to enable new research into protein and nucleic acids to allow greater insight into their role in human health in ways that have not been possible with any other current research and diagnostic technology. We believe this greater insight will facilitate a paradigm shift in healthcare from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention.
84
Our strategy to achieve this includes:
- •
- Focus on the highly attractive, expanding market for protein detection and analysis
Our focus on the detection of protein biomarkers is driven by a growing understanding of the essential role and impact of proteins on human health. While genomic research provides valuable information about the role of genes in health and disease, proteins are both more prevalent than nucleic acids and, we believe, more relevant to a precise understanding of the nuanced continuum between health and disease. Protein measurement goes beyond genetic predisposition, indicating the impact of a range of influences on health, including environmental factors and lifestyle, providing deeper and more relevant insight into what is happening in a person's body in real time. Our technology provides a unique bridge between understanding the human genotype and phenotype, which we believe addresses a large unmet need in life science research, translational medicine and drug development.
- •
- Continue to drive adoption of the Simoa platform in the life science research, diagnostics and precision health screening markets
Simoa has the potential to significantly expand the life science research market because of its unrivaled sensitivity, in particular by enabling researchers to perform studies on protein biomarkers that they were previously unable to perform. We believe Simoa has the capability to enable the development of a new category of non-invasive diagnostic tests and tools based on blood, serum, saliva and other fluids that could replace current invasive, expensive and inconvenient diagnostic methods, including spinal tap, diagnostic imaging and biopsy. In the precision health screening market, we believe that Simoa can be used to monitor protein biomarker levels of seemingly healthy, asymptomatic people, and potentially to signal and provide earlier detection and monitoring of the onset of disease.
- •
- Leverage the Simoa "ecosystem" to grow our customer base and further penetrate our existing customer base
In an effort to enhance the productivity of our instrument base, we have launched an extensive customer outreach program that we call Catalyzing Customer Engagement, or CCE. Through CCE, we actively engage customers to optimize their workflow and better understand our instruments' and products' capabilities, resulting in increased utilization of our installed instrument base.
- •
- Utilize the flexibility of the Simoa platform to expand into complementary markets, including nucleic acid detection
We plan to utilize the flexibility of the Simoa platform to expand our product offering to include other testing capabilities, including detection of nucleic acids. We believe that our Simoa technology has the potential to provide the same sensitivity as PCR-based assays in detecting nucleic acids without the issues associated with amplification. The ability to integrate nucleic acid and protein testing capabilities into a full service instrument would hold significant value to our customers.
- •
- Leverage the data generated by Simoa to drive adoption of our technology
Technology being employed in the healthcare industry has become increasingly sophisticated, creating the need to aggregate and digitize the significant amount of data being created in order to better achieve the goals of higher quality and more efficient care. Simoa generates digitized data for highly relevant biomarkers that can provide a nuanced view into the continuum of health and disease. We plan to use the data generated by the Simoa technology to improve and create additional assays, with the goal of enabling more precise research today and contributing to precision health in the future.
85
- •
- Grow into new markets organically with our customers and through strategic collaborations
Our customers have access to a large breadth of diverse markets, spanning research and clinical settings. As these customers continue to gain experience with our proprietary Simoa technology and further appreciate its potential, we believe moving into diagnostics and ultimately precision health is a natural extension of some of the work that our customers are doing today in the research market. For example, Simoa's unprecedented sensitivity has the potential to uncover research insights that could identify novel biomarkers, which could help stratify patients in clinical trials potentially leading to a companion diagnostic, and ultimately a precision health test that could monitor and identify early disease. This progression with our customers will help us move into new markets organically in a cost effective manner, while also retaining significant upside. Additionally, we currently have partnerships in place with leading diagnostics companies and plan to continue evaluating strategic collaborations that could help us access these new markets.
We intend to pursue the application of our Simoa technology to the life science research, diagnostics and precision health screening markets. Our initial commercial strategy targets the large and growing life science research market and we believe that the diagnostic market and the precision health screening market represents a significant future commercial opportunities for Simoa. According to estimates in the Third-Party Research Report, we believe the aggregate market opportunity for us or others using Simoa has the potential to expand to $38 billion as researchers and healthcare practitioners develop new applications for our products that span the continuum from research through diagnosis and precision health.
Proteins are versatile macromolecules and serve critical functions in nearly all biological processes. They are complex molecules that organisms require for the structure, function and regulation of the body's tissues and organs. For example, proteins provide immune protection, generate movement, transmit nerve impulses and control cell growth and differentiation. Understanding an organism's proteome, the complete set of proteins and their expression levels, can provide a powerful and unique window into its health, a window that other types of research, such as genomics, cannot provide.
The human body contains approximately 20,000 genes. One of the core functions of genes, which are comprised of DNA, is to regulate protein production—which ones are produced, the volume of each, and for how long—influenced by both biological and environmental factors. These 20,000 genes help govern the expression of over 100,000 proteins, approximately 10,500 of which are known to be secreted in blood, and fewer than 1,300 of which can be consistently detected in healthy individuals using conventional immunoassay technologies. Accordingly, the study of much of the proteome has not been practical given the limited level of sensitivity of existing technologies. To date, we have developed assays that address approximately 80 of the proteins secreted in blood. We estimate that the current sensitivity of our Simoa technology has the potential to detect and measure up to one-third of the approximately 9,200 proteins secreted in blood that are not consistently detectable using conventional immunoassay technologies.
While genomic research provides valuable information about the role of genes in health and disease, proteins are both more prevalent than nucleic acids and, we believe, more relevant to understand precisely the nuanced continuum between health and disease. Genes may indicate the risk of developing a certain disease later in life, but they are not able to account for the impact of environmental factors and lifestyle, such as diet and exercise, or provide insight into what is happening in a patient's body in real time. For
86
example, identical twins have the same genotype, but may develop different diseases over the course of their lifetime, largely due to environmental factors.
Much like the sequencing of the human genome with the Human Genome Project and the development of both PCR and next generation sequencing technologies to detect nucleic acids, both of which accelerated biomedical genomic research, we believe the ability to study more of the proteome enabled by our more sensitive protein detection technology will have a profound impact on proteomic research. With our ultra-sensitive Simoa detection technology, researchers can assess the symptoms of disease or injury and compare them to the presence and levels of relevant proteins that are not detectable using conventional technologies, leading to a better understanding of how proteins individually and/or collectively impact and influence important biological processes and the health and well-being of individuals. We believe this research into understanding the individual characteristics and functioning of proteins will be central to earlier detection, monitoring, prognosis and, ultimately, prevention, by providing researchers with the ability to assess the impact of particular proteins on the progress of disease and injury from the time of early onset of symptoms.
Existing technologies and their limitations
Protein analysis
The enzyme-linked immunosorbent assay, or ELISA, has been the most widely used method of sensitive detection of proteins for over 40 years. In simple terms, in ELISA, an unknown amount of antigen (e.g., protein, peptide, antibody, hormone) is affixed to a solid surface, usually a polystyrene multiwell plate, either directly, or indirectly through use of a conjugated secondary or "capture" antibody (sandwich ELISA). A specific "detection" antibody is applied over the surface to bind to the antigen. This detection antibody is linked to an enzyme, and in the final step, a substance called an enzyme substrate is added, and the enzyme converts to colored or fluorescent product molecules, which are detected by a plate reader. Sandwich ELISA is depicted in the graphic below:
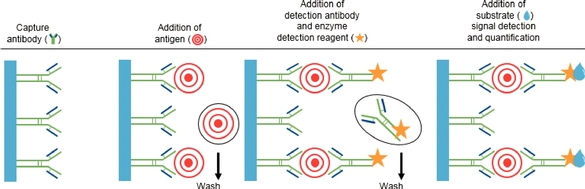
Aside from ELISA, there are other technologies available for protein analysis today, such as Western blotting, mass spectrometry, chromatography, surface plasmon resonance, Raman-enhanced signal detection, immune-PCR, and biobarcode assay. However, the proteins detectable by these conventional, analog immunoassay technologies represent a mere fraction of what is estimated to be approximately 10,500 secreted proteins in circulation in human blood. While a number of techniques have been used to attempt to increase sensitivity of detection, we believe all of these approaches have limitations, including:
- •
- dilution of colored or fluorescent product molecules due to large volume of liquid in traditional-sized wells, limiting sensitivity;
87
- •
- narrow dynamic range (i.e., the range of concentration of proteins being detected), that may require sample dilution, diluting molecules
and increasing sample volume requiring additional enzymes to reach detection limit;
- •
- low detection limit of readers restrict sensitivity and ability to detect low abundance proteins, particularly when proteins are at normal
physiological levels; and
- •
- limited success in increasing sensitivity of detection due to procedural complexity and length.
Genomic analysis
Over the past few decades, scientists have developed a variety of genomic analysis methods to measure an increasing number of genomic biomarkers aimed at more effectively detecting diseases. The most widely used method for genetic testing is PCR, which involves amplifying, or generating billions of copies of, the DNA sequence in question and then detecting the DNA with the use of fluorescent dyes. PCR is used to amplify the nucleic acid through the use of enzymes and repeated heating and cooling cycles, with fluorescent dyes incorporated during each amplification cycle. The expression of the nucleic acid is then inferred based on the number of amplification cycles required for the target to become detectable. PCR is sometimes referred to as an analog technology because the number of cycles of amplification, rather than a direct measure, is used to infer the level of gene expression. The wide availability of PCR chemistry makes it a popular approach for measuring the expression of nucleic acids, but the use of enzymes in numerous cycles of amplification can introduce distortion and bias into the data, potentially compromising the reliability of results, particularly at low concentrations.
Due to the complexity, susceptibility to contamination and significant costs related to PCR and other existing technologies, the genomic testing market generally remains limited to reference laboratories, research facilities and laboratories associated with large hospitals. A typical molecular diagnostics laboratory in a hospital or research laboratory setting is a dedicated facility that employs highly skilled technologists and is supervised by a technician with a Ph.D. or M.D./Ph.D. To guard against contamination, which is a common result of target amplification, a typical laboratory will require at least three separate rooms, or isolation areas, to perform PCR-based assay methods for genomic testing.
Our Simoa technology significantly advances conventional sandwich ELISA technology and is capable of unprecedented protein detection sensitivity. Simoa digital immunoassays utilize the basic principles of conventional bead-based sandwich ELISA and require two antibodies: one for capture, which is applied to the beads, and one for detection. Unlike ELISA, which runs the enzyme-substrate reaction on all molecules in one well, Simoa reactions are run on individual molecules in tiny microwells, 40 trillionths of a milliliter that are 2.5 billion times smaller than traditional ELISA wells. Traditional ELISA analog measurements increase in intensity only as the concentration of a sample increases. Simoa digital technology measurements, however, are independent of sample concentration intensity and rely on a binary signal/no signal readout, enabling detection sensitivity that was not previously possible.
Our Simoa platform is highly flexible, designed to enable practical high-sensitivity protein analysis for academic researchers looking at novel proteins all the way through to high throughput analysis performed by large biopharmaceutical organizations. The following chart describes the steps through which our Simoa technology detects proteins:
88
|
Sample Preparation of ELISA Sandwich
|
Simoa uses beads coated with capture antibodies that bind specifically to the protein being measured. After an enzyme-linked detection antibody binds to the protein, the enzyme substrate is added (as depicted by the white star in the graphic on the left). The enzyme associated with the enzyme-linked detection antibody then reacts with the enzyme substrate causing the enzyme substrate to become fluorescent (as depicted by the change in color of the star in the graphic). | |
Injection of Bead/Substrate Solution into Simoa Disk
|
This mixture of beads and enzyme substrate is then injected into our proprietary Simoa disk, which contains 24 arrays of microwells arranged radially. Each 3 × 4 millimeter array contains approximately 239,000 microwells, each of which is large enough to accommodate only a single bead. |
|
Bead/Substrate Solution Settles and Wells are Sealed
|
The bead/substrate solution is drawn across the array and the beads settle by gravity onto the surface of the array, and a fraction of them fall into the microwells. The remainder lie on the surface, and oil is introduced into the channel to displace the substrate solution and excess beads, and, most importantly, to seal the wells. |
|
Simoa Readout
|
The entire array is then imaged using ultrasensitive digital imaging, and the sealed wells that contain beads associated with captured and enzyme labeled protein molecules are identified. |
89
Our Simoa technology offers unprecedented protein detection sensitivity and enables detection of low abundance and previously undetectable biomarkers. The following chart shows examples of the levels of detection, or LoD, of certain Simoa assays and commercially available ELISA assay compared to the median lower normal plasma or clinical ranges of various protein biomarkers. As shown below, the LoD for most of the assays from a leading manufacturer of commercial ELISA assays is above the median lower normal plasma or clinical ranges, making these biomarkers undetectable at normal physiological levels with these assays.
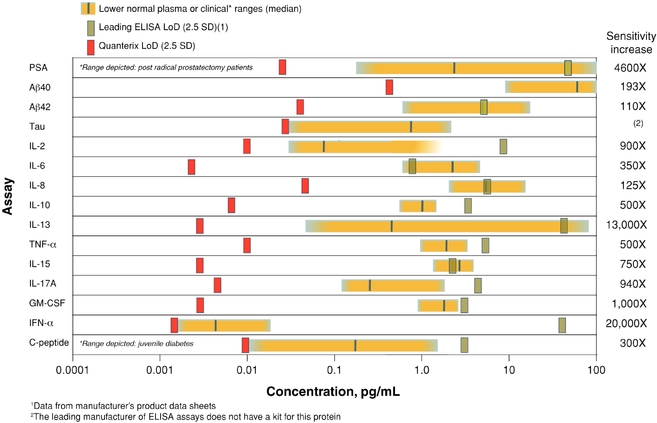
Each of the increments in the horizontal axis in the table above represents a 10-fold increase in sensitivity. Using the protein IL-2 as an example from the graphic above, the LoD for the leading commercially available IL-2 assay is approximately 9 pg/mL, whereas the LoD for our Simoa assay is approximately 0.01 pg/mL, representing a 900-fold increase in sensitivity.
Multiplexing capability
The ability to multiplex, or simultaneously measure multiple proteins (or other biomarkers) in a single assay, can be important to researchers to maximize the biological information from a sample, and to develop more specific diagnostic tests. Importantly, Simoa multiplexing maintains single plex precision, while competitive platforms lose sensitivity when multiplexing is used. Multiplexing is achieved with our Simoa technology by using beads labeled with different fluorescent dyes specific to the biomarker being analyzed. After the assay is run, the array of microwells is imaged across the wavelengths of the different labeled beads. The results are measured for each protein captured by each of the different beads.
90
We have demonstrated the ability to identify and differentiate up to 35 different bead subpopulations on the Simoa HD-1 Analyzer, which is a prerequisite to our ability to develop an assay with the capacity to detect an equivalent number of proteins in a single sample.
In 2017, we commercially launched a Simoa neurology 4-plex assay (Nf-L, tau, GFAP and UCH-L1) for the study of traumatic brain injury and other neurodegenerative conditions. Simoa is the only technology with the sensitivity to detect all four of these markers in blood, whereas other assay technologies require cerebrospinal fluid, or CSF, to detect all four of these markers due to sensitivity limitations. This is a significant advantage in terms of ease of use, patient comfort, speed and cost-effectiveness.
Nucleic acid testing
Our initial focus has been on the use of Simoa to detect protein biomarkers. However, we are also developing our Simoa technology to detect nucleic acids in biological samples. While methods for measuring nucleic acid molecules have advanced substantially, currently available techniques still have drawbacks. For example, polymerase chain reaction, or PCR, is a sensitive method that is widely used for measuring gene expression. However, PCR carries the potential for data distortion and bias from the repeated addition of enzymes, and heating and cooling cycles needed to amplify a copy of the nucleic acid being measured. In nucleic acid analysis, we believe that Simoa has the potential to provide the same sensitivity as traditional PCR-based assays with the following benefits:
- •
- no need for amplification of the targeted nucleic acid, which can result in amplification distortion and bias;
- •
- reduced cross-contamination because of direct detection of single molecules vs. the detection of a large number of copies of the nucleic acid;
and
- •
- the ability to detect some samples without requiring purification of the nucleic acid, such as in environmental water.
For detection of nucleic acids with Simoa, instead of coating the beads with capture antibodies as is done for detecting proteins, the beads are coated with nucleic acid capture probes. Samples with the target nucleic acid molecules are then added and are captured by the beads. Nucleic acid detection probes (instead of detection antibodies) are then added and attach to the target nucleic acid molecules which are then labeled using an enzyme substrate that is detected and counted using the Simoa disk and instrument. This assay is pictured below:
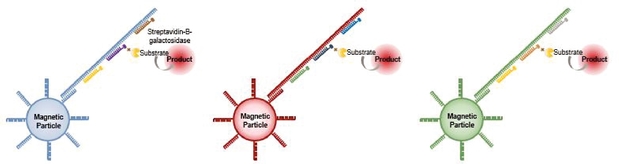
Simoa has been used to detect short sequences of RNA, known as microRNA, that are important in a number of biological systems, and are widely used in innovative therapeutic and gene editing technologies. The assay was used to detect microRNA-122, or miR-122, a marker of liver toxicity, from the serum of
91
patients who had overdosed with acetaminophen. As shown in the graph below, these patients had elevated miR-122 levels compared to healthy controls.
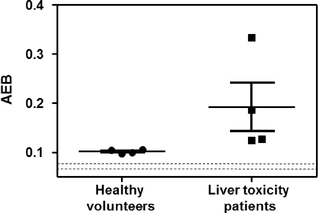
This approach suggests potential for applications for measuring drug-induced liver injury for both safety testing of drugs in development and for monitoring of approved drugs.
Our commercial strategy is to pursue the application of our Simoa technology to the life science research, diagnostics and precision health screening markets.
Life science research
Our initial target market is the large and growing life science research market, including both proteomics and genomics research. We believe Simoa is well-positioned to capture a significant share of this market because of its superior sensitivity, automated workflow capabilities, multiplexing and its ability to work with a broader range of sample types.
Proteomics, the study of the proteins produced by the body, is important to understanding disease, and researchers study proteins to understand the biological basis for disease and how to improve diagnosis and treatment. The proteins detectable by conventional, analog immunoassay technologies represent a mere fraction of the proteins that can be detected by Simoa, and we believe that Simoa can inspire a new level of research into these previously undetectable proteins and their role in disease. While it is estimated that there are approximately 10,500 secreted proteins in circulation in human blood, fewer than 1,300 of them can be detected in healthy individuals using conventional immunoassay technologies. In addition, many of the proteins which can be detected by other technologies are only detectable after they have reached levels that reflect more advanced disease or injury. By substantially lowering the limit of detection of protein biomarkers, Simoa holds significant potential to expand research into the diseases associated with the thousands of proteins that were previously undetectable, as well as into earlier detection of the proteins currently detectable by other technologies only after they have reached levels that reflect more advanced disease or injury. Simoa provides researchers the ability to see the nuanced continuum of health to disease more efficiently and effectively than any other technology commercially available today, offering the potential for the first time to better understand the onset of disease cascades and catalyzing a new era of medical and life science research, drug discovery and disease prevention.
92
As an indication of the market's acceptance of our Simoa platform, researchers at pharmaceutical and biotechnology companies are integrating our platform into drug development protocols to more efficiently and effectively develop drugs. Using Simoa's unprecedented sensitivity to measure previously undetectable levels of target biomarkers prior to and following administration of a drug, drug developers can non-invasively and objectively determine whether a drug candidate is having a desired impact on the target biomarker.
For example, a large pharmaceutical company used Simoa to measure decreases in Ab1-42, a neurological biomarker, after administration of an Alzheimer's disease drug candidate, and was able to see specific and dose dependent reductions in this biomarker in plasma.
In addition, researchers can also use Simoa to monitor a drug candidate's unwanted effect on "off-target" biomarkers and predict side effects, addressing the significant issue of drug toxicity, which is the fourth leading cause of death in the United States.
Beyond proteins, our Simoa technology has the potential to be used to detect nucleic acids, which expands our market opportunity. We believe that our Simoa technology has the potential to ultimately provide the same sensitivity as PCR-based assays without the issues associated with amplification used in PCR. We believe this represents another significant commercial opportunity for us.
According to estimates in the Third-Party Research Report, we believe that the total life science research market addressable by Simoa, including both proteomics and genomics research, is $3 billion per year and has the potential to reach $8 billion per year.
Diagnostics
The diagnostic market represents a significant future commercial opportunity for Simoa as well. We believe existing biomarker diagnostics can be improved by Simoa's sensitivity to enable earlier detection of diseases and injuries, and that new diagnostics may be developed using protein biomarkers that are not detectable using conventional, analog immunoassay technologies but are detectable using Simoa. We also believe that the ultra-sensitive protein detection provided by Simoa can enable the development of a new category of non-invasive diagnostic tests and tools based on blood, serum and other fluids that have the potential to replace current more invasive, expensive and inconvenient diagnostic methods, including spinal tap, diagnostic imaging and biopsy.
For example, researchers have conducted studies using Simoa that indicate that neurological biomarkers, including tau and Nf-L, may someday be able to replace diagnostic imaging to diagnose traumatic brain injury, or TBI. Our Simoa assays for tau and Nf-L are 3,500-fold and 840-fold more sensitive, respectively, than the leading assay platforms, and are the only assays that can reliably detect these critical protein biomarkers in blood. Almost 90% of patients who visit U.S. hospital emergency rooms and receive a CT scan show no structural brain injury. In addition, CT scans have approximately 100 times more radiation than a chest x-ray, and are suspected of causing cancer in up to 29,000 people per year, underscoring the need for development of a safe and accurate blood-based diagnostic test for TBI, which we believe may be enabled by our Simoa technology.
Simoa also has significant potential in the emerging field of companion diagnostics. A companion diagnostic test is a biomarker test that is specifically linked to a therapeutic drug that can help predict how a patient will respond to the drug. Drug developers can use companion diagnostics to stratify patients and select only those patients to study for whom a drug is expected to be most effective and safe. Companion diagnostics have demonstrated the ability to both improve the probability of approval and accelerate
93
approval of new drugs. Not only can Simoa be used to develop companion diagnostics to stratify patients in clinical trials and for treatment, but Simoa's sensitivity also enables the development of companion diagnostics based on protein biomarkers that can actively and regularly monitor whether an approved drug is having the desired biological effect. This can quickly and efficiently enable doctors to adjust the course of treatment as appropriate by increasing or decreasing dosages or even switching therapies.
There has been significant interest from third parties to use our technology to develop applications for the diagnostic market, which has resulted in collaborations with leading diagnostic companies, such as bioMérieux. In addition, we have had discussions with lab service companies that are interested in using our technology to develop laboratory developed tests that may be more sensitive than currently available commercial tests.
Precision health screening
We believe that Simoa's ability to detect and quantify normal physiological levels of low abundance proteins that are undetectable using conventional, analog immunoassay technologies will enable our technology to be used to monitor protein biomarker levels of seemingly healthy, asymptomatic people, and potentially to signal and provide earlier detection of the onset of disease. This may facilitate a paradigm shift in healthcare, from an emphasis on treatment to a focus on earlier detection, monitoring, prognosis and, ultimately, prevention, enabling a "precision health" revolution.
We believe there is the potential for a number of neurological, cardiovascular, oncologic and other protein biomarkers associated with disease to be measured with a simple blood draw on a regular, ongoing basis as part of a patient's routine health screening, and for those results to be compared periodically with baseline measurements to predict or detect the early onset of disease, prior to the appearance of symptoms.
According to estimates in the Third-Party Research Report, we believe that the total diagnostic and precision health screening markets addressable by us and others using Simoa have the potential to reach an aggregate of $30 billion per year upon receipt of the necessary regulatory approvals, which we have not yet begun the process to obtain.
We have focused the application of our Simoa technology on areas of high growth and high unmet need and where existing platforms have significant shortcomings that our technology addresses. In particular, we have focused on the following areas: neurology, oncology, cardiology, infectious disease and inflammation. According to estimates in the Third-Party Research Report, we believe that these are areas of high unmet need with a total addressable market for us and others using Simoa that has the potential to reach $38 billion across research, diagnostic and precision health screening indications.
Neurology
We believe that the ability of our Simoa technology to detect neurological biomarkers in blood at ultra-low levels, which have traditionally only been detectable in cerebrospinal fluid, or CSF, has the potential to rapidly advance neurology research and drug development, and transform the way brain injuries and diseases are diagnosed and treated. To our knowledge, the brain is the only organ in the body for which there is not currently a blood-based diagnostic test. The challenge with developing blood-based tests for the brain is that the blood-brain barrier, which is formed by endothelial cells lining the cerebral microvasculature, is very tight and severely restricts the movement of proteins and other substances
94
between these endothelial cells and into blood circulation. Accordingly, diagnosis of brain disease and injury has traditionally required either an MRI scan of the brain or a spinal tap to collect CSF, both of which are costly and highly invasive for the patient. The sensitivity of the Simoa platform has enabled researchers to discover that extremely small amounts of critical neural biomarkers diffuse through the blood-brain barrier, and are released into the blood during injury and in connection with many neurodegenerative brain diseases. However, the concentrations of these neural biomarkers in the blood are so low that they are undetectable by conventional, analog immunoassay technologies.
As one example, we have developed ultra-sensitive protein assays for the neural biomarkers Ab42 and tau that are approximately 2,000 to 3,500-fold more sensitive, respectively, than benchmark commercial assays. Our protein assays are the only currently available assays on the market capable of precise measurement of these neural biomarkers in blood in diseased and healthy individuals.
To date, there have been over 70 scientific publications on approximately 48 neural biomarkers using our Simoa technology, and we believe that ultra-sensitive digital detection of neural related biomarkers in the blood is becoming an essential research and development tool for an increasing range of neurological disorders, including CTE, Alzheimer's Disease, Dementia, Parkinson's Disease, Multiple Sclerosis and TBI. The goal of this research is to eventually develop accurate diagnostic tools, predictive health screens and, ultimately, more effective treatments.
Earlier this year, researchers using Simoa technology published a paper in JAMA Neurology demonstrating that a simple blood test for ther neurological biomarker Nf-L exhibited the same level of diagnostic accuracy for diagnosing Alzheimer's dementia as currently established CSF biomarkers. The study was a major study of almost 600 patients from the Alzheimer's Disease Neuroimaging Initiative. The graph below depicts the diagnostic accuracy of plasma Simoa Nf-L measurements compared with traditional CSF biomarkers. The diagnostic accuracy of the plasma Simoa Nf-L results approached 90%, in line with the CSF biomarkers on the same patients.
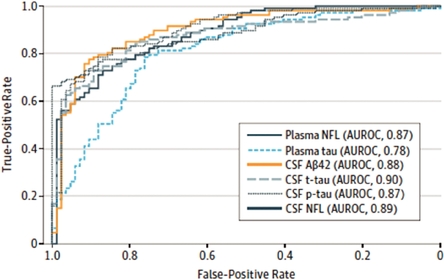
95
In addition, Simoa plasma Nf-L values were associated with cognitive deficits and neuroimaging hallmarks of Alzheimer's dementia at baseline and during follow-up. High plasma Nf-L correlated with poor cognition and Alzheimer's dementia -related brain atrophy and with brain hypometabolism (lower neural energy). These data suggest a simple Simoa blood test for NF-L may have clinical utility as a noninvasive biomarker in AD.
Traumatic brain injuries, or TBIs, lead to approximately 5 million individuals visiting emergency rooms per year in the United States alone, often with broad and inconclusive diagnosis. Current methods of TBI diagnosis involve computerized tomography, or CT, scans that fail to diagnose approximately 90% of mild TBI. Simoa has demonstrated the sensitivity to identify relevant neurological biomarkers, such as Nf-L, tau, GFAP and UCHL-1, to more adequately address diagnosis of TBIs and overall brain health.
Leading researchers in neurology have used Simoa to study biomarkers in the blood of athletes after concussion in many high-impact sports. Our platform measures critical neural biomarkers in blood that correlate repeated head trauma from both concussions and subconcussive events with poor patient outcomes, including the potential development of Chronic Traumatic Encephalopathy, or CTE, which currently can only be diagnosed after death via a brain autopsy. A recent publication by an NIH researcher indicates that measuring tau in the blood with Simoa may help identify concussed individuals requiring additional rest before they can safely return to play. Eventually, we believe it may be possible to develop a mobile screen enabling clinicians to quickly and accurately determine whether it is safe for concussed athletes to return to play.
In 2017, we commercially launched a Simoa neurology 4-plex assay (Nf-L, tau, GFAP and UCH-L1) for the study of traumatic brain injury and other neurodegenerative conditions. Whereas other assay technologies require cerebrospinal fluid, or CSF, to detect all four of these markers, due to its sensitivity, Simoa is the only assay that can detect all of these biomarkers directly from blood samples. This is a significant advantage in terms of ease of use, patient comfort, speed and cost-effectiveness.
In 2016, Fast Company named Quanterix one of the "World's Most Innovative Companies" for our work in concussion detection. We also were awarded two competitive grants from the NFL-GE Head Health Challenge to advance this work in the detection and quantification of mild traumatic brain injury, or TBI.
We estimate that the total addressable market for Simoa for neurology has the potential to reach $6 billion across research, diagnostic and precision health screening indications.
Oncology
Our ultra-sensitive Simoa technology has the potential to detect increased levels of oncology biomarkers during the very early stages in disease development. Biomarkers can be useful tools for diagnostics, prognostics and predictive cancer detection. However, many traditional assay technologies can only detect these biomarkers after the disease has progressed and the patient has become symptomatic. Simoa's highly sensitive detection capability may result in earlier detection, better monitoring and treatment and improved prognoses for patients. Additionally, Simoa has shown early promise as an alternative to more invasive diagnostic procedures. To date, there have been 16 scientific publications on approximately 39 cancer biomarkers using our Simoa technology.
Simoa was used in a recent unpublished scientific study that we understand indicates it may be possible to eventually replace routine mammograms with a very sensitive, more accurate, low cost, non-invasive blood test. In this retrospective study, researchers found that Simoa resulted in significantly fewer false positives and false negatives than mammography. Inaccurate mammography results in unnecessary stress, additional health care costs from follow up diagnostic mammograms, unnecessary biopsies and increased lifetime exposure to radiation. Researchers are also developing ultrasensitive assays for lung and pancreatic cancer biomarkers using Simoa, potentially replacing the need for imaging and biopsy. We believe our Simoa
96
technology has the potential to lead to rapid, cost effective, accurate blood-based health screens, further enabling the liquid biopsy market, which is estimated to grow to almost $3 billion by 2026.
Cancer immunotherapy is a promising new area that is significantly affecting cancer remission rates. One challenge of immunotherapy approaches is that the elicited immune responses are not always predictable and can vary from person to person and protocol to protocol. There exists a significant need to develop biomarker tools to monitor these drugs and their effects. Serum protein biomarkers have the potential to be used in the field of immuno-oncology to stratify patients, predict response, predict recurrence, reveal mechanism of action and predict side effects. One technical challenge to using these biomarkers has been the development of immunoassays with sufficient sensitivity to measure immune modulates directly in serum. We have developed a set of 38 ultrasensitive immune modulation assays (cytokines and chemokines) that can be used to directly monitor the immune response. In particular key immune regulatory cells (T-regs, dendritic cells, macrophages) secrete very low amounts of the protein Interferon gamma (IFN-gamma) and these levels cannot be detected in serum using conventional, analog immunoassay technology, however they can be tracked with our Simoa IFN-gamma assay. Additionally, we have developed an ultra-sensitive assay for PD-L1 which is one of the major immuno-oncology targeted antigens. Several studies have shown that our ultrasensitive assays can be valuable tools for monitoring immuno-oncology drugs and protocols.
Additionally, we believe residual cancer cell detection post-surgery or treatment may significantly improve outcomes for a variety of cancer types, by helping identify and segment patients at a greater risk of reoccurrence post-surgery due to residual cancer. We have developed an ultra-sensitive biomarker assay for Prostate Specific Antigen, or PSA, that is over 1,000-fold more sensitive than benchmark commercial PSA assays. This assay is the only currently available technology that can detect levels of PSA in blood samples of prostate cancer patients shortly following radical prostatectomy, and we and researchers from Johns Hopkins and NYU conducted a pilot study on the utility of this assay to predict recurrence of prostate cancer after this procedure. In this study, the blood of prostate cancer patients taken three to six months following a radical prostatectomy at least five years earlier was analyzed with Simoa. The majority of samples had PSA levels below the detectable limits of traditional PSA assays. Our Simoa technology, however, was able to detect and quantify PSA levels in all samples. As shown in the following graph, the study demonstrated that the PSA assay using our Simoa technology has the potential to be highly predictive of prostate cancer recurrence over a five-year period. This has the potential to be a powerful prognostic tool, and allowing adjuvant radiation treatment to be targeted only to the men who actually would benefit.
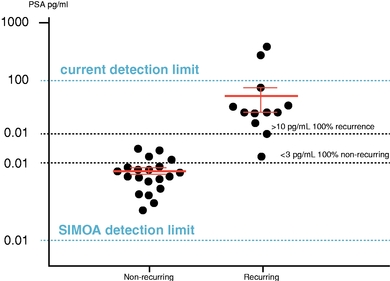
We estimate that the total addressable market for Simoa for oncology has the potential to reach $25 billion across research, diagnostic and precision health screening indications.
97
Cardiology
Heart disease and related cardiovascular ailments remain the leading cause of death in the United States, contributing to nearly 1 in 4 deaths in the United States, according to the CDC. A significant need remains for early prediction of heart attacks and other cardiac events. Simoa's highly sensitive digital measurement capabilities have the potential to be used to predict early cardiac disease.
To date, there have been six scientific publications on approximately 11 cardiology biomarkers using our Simoa technology.
Infectious disease
The ability to detect infectious disease biomarkers before the onset of an immune response, where a virus is most contagious and multiplying rapidly, is critical for controlling the spread of disease. We believe that our Simoa technology can have a significant impact in reducing the spread of infectious diseases by making early stage detection more specific and widely available.
Today, early detection of infectious disease is conducted using nucleic acid testing to detect the nucleic acid of the viral or bacterial organism because the levels of infectious disease specific antigens are too low in the early stage of disease to be detected by traditional immunoassay technology. However, the sensitivity of our single molecule detection capabilities enables the detection of extremely low levels of infectious disease specific antigens with sensitivity that rivals the use of nucleic acid testing in this application, without the potential biases inherent in amplification technologies, such as PCR.
We have developed a simple Simoa assay with more than 4,000-fold greater sensitivity than benchmark commercial protein assays capable of detecting the HIV-specific antigen, p24. This Simoa p24 sensitivity matches the sensitivity of more expensive and complex nucleic acid testing methods. The following graph shows a comparison that we conducted in 2011 of the Simoa p24 assay with a commercially available nucleic acid testing method, as well as two commercially available p24 immunoassay methods for early detection of HIV infection. The Simoa p24 assay detects infection as early as the nucleic acid testing method (11 days from initial blood draw), and a full week before the earliest signs of infection by the
98
conventional p24 immunoassay methods. This early detection of acute HIV infection can be critical for controlling the spread of HIV, as HIV is ten times more infectious in the acute phase.
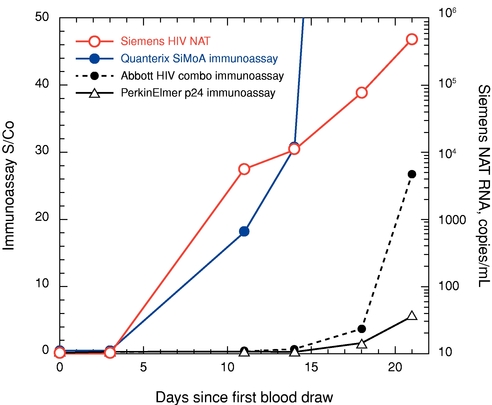
In addition, we believe the detection of a specific protein is more relevant to the determination of the pathogenic effect than detection of the organism itself because someone may carry a pathogenic organism with no pathogenic effect. Researchers have demonstrated that Simoa can detect Clostridium difficile (C. diff) toxins A and B with sensitivities similar to the PCR detection of the C. diff organism itself. Because the C. diff organism does not always produce toxins, PCR methods that detect the C. diff organism suffer from a very high false positive rates, which may result in incorrect diagnoses and the overuse of antibiotics. We believe that using Simoa to detect the toxins rather than the organism has the potential to provide a higher level of sensitivity and specificity, greatly reducing false positives.
We will continue to develop Simoa assays for pathogenic antigens that are competitive in sensitivity to PCR but more specific to the pathogenicity of the offending organism. We believe that these Simoa assays could also be invaluable tools for the development of anti-infective drugs and treatment monitoring of anti-viral and anti-bacterial drugs.
To date, there have been 26 scientific publications on approximately 12 infectious disease biomarkers using our Simoa technology.
Inflammation
Inflammation underlies the response of the body to injury in a variety of diseases. Simoa assays can measure inflammatory and anti-inflammatory molecules in serum and plasma with unprecedented sensitivity. This has the potential to enable new discoveries into the role of inflammation in the biology of health and disease. Our Simoa technology measures low levels of inflammatory proteins, including cytokines and chemokines, that characterize a range of inflammatory diseases, including Crohn's disease,
99
asthma, rheumatoid arthritis and neuro-inflammation. We believe the sensitivity of Simoa can provide a clearer picture of the underlying state of the immune response and disease progression.
Our Simoa technology also has the potential to be used by companies developing anti-inflammatory drugs to quantify the effect a drug has on a particular inflammatory cytokine and to monitor therapeutic efficacy. For example, we conducted a study in conjunction with the Mayo Clinic using our Simoa technology on patients with clinically active Crohn's disease undergoing anti-TNF-a therapy with Remicade, Humira or Enbrel. As shown in the graph below, researchers were able to detect and quantify the TNF-a levels of the patients before and after treatment. These levels were all below the LoD of traditional immunoassays.

We believe that a better understanding of the inflammatory response will be critical to future opportunities for wellness screening and disease response monitoring. Anti-inflammatory drugs are expensive and can have serious side effects, such as increased risk of infection. By monitoring biomarkers indicative of response, clinicians may be able to adjust dose to reduce side effects or increase efficacy.
To date, there have been 20 scientific publications on approximately 44 inflammatory biomarkers using our Simoa technology.
100
Our commercial portfolio includes instruments, assay kits and other consumables, and contract research services offered through our Simoa Accelerator Laboratory, as follows:
| |
|
|
|---|---|---|
| | | |
| Product |
Key attributes |
|
| | | |
| Simoa HD-1 Analyzer | ||
|
• commercially launched in January 2014
• most sensitive immunoassay platform on market
• fully automated, floor-standing instrument
• wide dynamic range
• multiplexing capability with small sample volume
• up to 400 samples per eight-hour shift
• homebrew capabilities |
|
| Quanterix SR-X | ||
|
• initiate early adopter program in 2017
• expected commercial launch in 2018
• reader only, benchtop instrument with lower price point
• same sensitivity, dynamic range and homebrew capabilities as HD-1
• multiplexing capability: SR-X to have 6-plex capability at launch with anticipated expansion to 35-plex capability
• sample prep and assay protocol flexibility |
101
| | | |
| Product |
Key attributes |
|
|---|---|---|
| | | |
| Assays and other consumables | ||
|
• over 80 assays developed for neurology, oncology, cardiology, infectious diseases and inflammation research
• homebrew kits containing beads and reagents required for customers to custom build assays
• proprietary Simoa disk with 24 arrays, each containing approximately 239,000 microwells |
|
| Services | ||
|
• contract research services provided through our Simoa Accelerator Laboratory
• over 320 projects completed to date
• extended warranty and service contracts |
Instruments and consumables
Simoa HD-1 Analyzer
We commercially launched the Simoa HD-1 Analyzer in January 2014. The HD-1 Analyzer is the most sensitive protein detection platform commercially available, and is currently capable of analyzing up to six biomarkers per test, with anticipated expansion capability to up to 35 biomarkers per test in 2018. Assays for the HD-1 Analyzer are fully automated (i.e. sample in to result out), and results for up to 66 samples are available in approximately one hour. We believe that this automation provides us an additional significant competitive advantage with pharmaceutical and biotechnology customers. Samples can be input into the instrument via 96-well microtiter plates or sample tubes where the system can multiplex and process tests in a variety of assay protocol configurations. Simoa digital immunoassays utilize the basic principles of conventional bead-based sandwich ELISA and require two antibodies: one for capture onto the beads, and one for detection (antigen 'sandwich').
Specialized software controls the Simoa instrumentation, analyzes the digital images produced, and provides customers with detailed analysis of their samples, such as the concentration of multiple biological molecules. The Simoa HD-1 Analyzer software automates the processes for running the instrument and
102
analyzing data from the user-defined protocols. Proprietary image analysis software is embedded in the system, which converts the raw images into signals for each biological molecule being analyzed within a sample. Data reduction software automatically converts those signals to concentrations for the different biological molecules.
We have sold more than 160 HD-1 Analyzers to over 100 customers around the world. We also have seven systems in our own Simoa Accelerator Laboratory.
Quanterix SR-X
We expect to initiate an early adopter program for our Quanterix SR-X instrument in the fourth quarter of 2017, with commercial launch in 2018. The Quanterix SR-X will utilize the same core Simoa technology and assay kits as the HD-1 Analyzer in a compact benchtop form with a lower price point designed to address the needs of researchers who value the ultra-sensitive detection capabilities enabled by Simoa.
In contrast to the fully automated workflow of the HD-1 Analyzer, the assay incubation and washing steps for the SR-X are performed outside of the instruments using conventional liquid handling methods. The offline sample prep provides additional flexibility to enable researchers to apply Simoa detection in an expanded range of applications including direct detection of nucleic acids. The SR-X system automates the steps loading Simoa beads onto Simoa disks with subsequent imaging, detection and data reduction. Processing time for imaging a 96 well plate is approximately 2.5 hours.
The SR-X system will support detection capability of up to six biomarkers per test at launch with expansion to capability of up to 35 biomarkers per test in 2018.
Assays and consumables
Recurring revenue is derived through the sale of consumables used to run assays on our instruments, and from our growing menu of Simoa digital biomarker assays, with more than 80 assays developed to date. In addition to these assays we have developed, the Simoa platform allows ease and flexibility in assay design, enabling our customers to develop their own proprietary in-house assays, called homebrew assays, using our Homebrew Assay Development Kit. Our goal is to continue to add to our assay kits to extend our application base.
We have developed a multi-tier product strategy dependent on customer requirements to accelerate the development and production of new assays. Discovery Kits for rapid commercialization are ideal for applications that need minimal validation and no multi-lot testing. In more regulated spaces, we offer Advantage Kits with additional testing. Our kits include all components required to run tests, such as beads, capture and detector reagents, enzyme reagents and enzyme substrate. Our consumables portfolio also includes our proprietary Simoa disks that are unique to our systems, as well as cuvettes, and disposable tips.
We have staffed our assay development and manufacturing teams to do the upfront work of antibody sourcing, assay development and optimization, sample testing and validation, transfer to manufacturing and final documentation. We outsource some of our assay development activities to other antibody and/or assay development providers and expect to continue to do so to achieve our aggressive menu expansion goals.
Services
Our Simoa Accelerator Laboratory provides customers a contract research option. Researchers, academics and principal investigators can work with our scientists to test specimens with existing Simoa assays, or
103
prototype, develop and optimize new assays. The Accelerator Laboratory supports multiple projects and services, including:
- •
- Sample testing. Utilizing commercially available Simoa kits, we have run
large studies for customers with thousands of specimens and small experiments with just a few samples. The sample protocol can be tailored precisely to the customer's needs and even large studies can
be run quickly. We have extensive experience testing many different sample types where biomarkers may be present at very low levels.
- •
- Homebrew assay development. Utilizing proprietary or commercially available
reagents in combination with the Quanterix Homebrew Assay Development Kit, we can rapidly develop a prototype assay exhibiting improved sensitivity compared to ELISA. The Accelerator Laboratory can
also be used to screen reagents to identify the optimal assay format or expand prototype efforts for further assay optimization or validation to ultimately deliver the highest level of performance.
- •
- Custom development. After identifying the optimal assay and conditions, the Accelerator Laboratory can be used to generate qualified bulk reagents or custom assay kits, providing customer access to validated kits for assays not yet commercially available on the Simoa platform.
To date, we have completed over 340 projects for over 145 customers from all over the world using our Simoa platform. In addition to being an important source of revenue, we have also found the Simoa Accelerator Laboratory to be a significant catalyst for placing additional instruments, as over 35 customers for whom we have provided contract research services have subsequently purchased an instrument from us.
We also generate revenues through extended-warranty and service contracts for our installed base of instruments.
Research and development
We continually seek to improve our platform and technology to enable more sensitive detection and measurement of biological molecules. This evaluation includes examining new assay formats and instrumentation improvements and upgrades to increase the performance of our Simoa assays and instruments. We are particularly focused on expanding our assay menu to extend the scope of applications for our platform and grow our customer base. Our assay menu expansion is driven by a number of factors, including input from key opinion leaders, customer feedback, homebrew projects, Accelerator Laboratory projects, new publications on biomarkers of industry interest, and feedback from our sales and marketing team. We also intend to continue to develop and market new instruments with different and/or improved capabilities in order to further broaden our market reach.
We distribute our instruments and reagents via direct field sales and support organizations located in North America and Europe and through a combination of our own sales force and third-party distributors in additional major markets such as Australia, China, India, Japan, South Korea, Lebanon and Singapore. Our domestic and international sales force informs our current and potential customers of current product offerings, new product and new assay introductions, and technological advances in Simoa systems, workflows, and notable research being performed by our customers or ourselves. As our primary point of contact in the marketplace, our sales force focuses on delivering a consistent marketing message and high level of customer service, while also attempting to help us better understand evolving market and customer needs.
104
As of September 30, 2017, we had approximately 40 people employed in sales, sales support and marketing, including 16 technical field application scientists. This staff is primarily located in North America and Europe. We intend to significantly expand our sales, support, and marketing efforts in the future by expanding our direct footprint in Europe as well as developing a comprehensive distribution and support network in China where significant new market opportunities exist. Additionally, we believe that there is significant opportunity in other Asia-Pacific region countries such as South Korea and Australia as well as in South America. We plan to expand into these regions via initial penetration with distributors and then subsequent support with Quanterix-employed sales and support personnel.
Our sales and marketing efforts are targeted at key opinion leaders, laboratory directors and principal investigators at leading biotechnology and pharmaceutical companies and governmental research institutions.
In addition to our selling activities, we align with key opinion leaders at leading institutions and clinical research laboratories to help increase scientific and commercial awareness of our technology, demonstrate its benefits relative to existing technologies and accelerate its adoption. We also seek to increase awareness of our products through participation at trade shows, academic conferences, online webinars and dedicated scientific events attended by prominent users and prospective customers.
To develop a thought leadership position in the precision health arena, we were a Platinum Sponsor of the inaugural Powering Precision Health Summit, or PPHS, in Cambridge, Massachusetts in September 2016 and of the second annual PPHS in Cambridge in October 2017. At PPHS in 2016, there were 22 cutting edge scientific talks covering neurology, cardiology, oncology and inflammation. There were over 200 registered attendees, including senior scientists, patient advocates, investors, and potential partners. At PPHS in 2017, there were 37 scientific talks and over 425 registered attendees.
Our systems are relatively new to the life science marketplace and require a capital investment by our customers. The sales process typically involves numerous interactions and demonstrations with multiple people within an organization. Some potential customers conduct in-depth evaluations of the system including running experiments in the Simoa Accelerator Laboratory and comparing results from competing systems. In addition, in most countries, sales to academic or governmental institutions require participation in a tender process involving preparation of extensive documentation and a lengthy review process. As a result of these factors and the budget cycles of our customers, our sales cycle, the time from initial contact with a customer to our receipt of a purchase order, can often be six to 12 months, or longer.
Our manufacturing strategy has two components: to outsource instrument development and manufacturing with industry leaders, and to internally develop and kit assays in our own facility.
Systems
The Simoa HD-1 Analyzer is manufactured by STRATEC Biomedical AG, based in Birkenfeld, Germany, and is manufactured and shipped from their Birkenfeld and Beringen, Switzerland facilities. See "—Key Agreements—Development agreement and supply agreement with STRATEC" for a description of this agreement. Simoa HD-1 Analyzers are shipped by STRATEC to our global customers' locations. Installation of, and training on, our products is provided by our employees in the markets where we conduct direct sales, and by distributors in those markets where we operate with distributors. The Quanterix SR-X will be manufactured by Paramit Corporation, based in Morgan Hill, California, and will be shipped to global customers by Paramit. We expect to commercially launch this instrument in 2018.
105
We believe this manufacturing strategy is efficient and conserves capital. However, in the event it becomes necessary to utilize a different contract manufacturer for either the Simoa HD-1 Analyzer or the Quanterix SR-X, we would experience additional costs, delays and difficulties in doing so, and our business would be harmed.
Consumables
We assemble our assay kits in our Lexington, Massachusetts facility. Our reagents are sourced from a limited number of suppliers, including certain single-source suppliers. Reagents include all components required to run an enzyme based immunoassay, such as beads, capture and detector reagents, enzyme reagents and enzyme substrate. Although we believe that alternatives would be available, it would take time to identify and validate replacement reagents for our assay kits, which could negatively affect our ability to supply assay kits on a timely basis.
Simoa disks are supplied through a single source supplier pursuant to a long-term supply agreement with STRATEC Consumables, a subsidiary of STRATEC Biomedical. This agreement provides for a sufficient notification period to allow for supply continuity and the identification and tech transfer to a new supplier in the event either party wishes to terminate the relationship. Our cuvettes are single sourced through STRATEC Biomedical, and the disposable tips used in our assays are commercially available.
License agreement with bioMérieux SA
In November 2012, we entered into a Joint Development and License Agreement, or JDLA, with bioMérieux SA. Under the terms of the JDLA, we granted bioMérieux an exclusive, royalty-bearing license to manufacture and sell instruments and assays using our Simoa technology for in vitro diagnostics used in clinical lab applications, food quality control testing, in vitro diagnostics and pharmaceutical quality control testing, and a co-exclusive, royalty-bearing license in certain other fields. Under the JDLA, bioMérieux was required to purchase instruments from us subject to certain minimum purchase requirements. We received a $10 million upfront payment and we were eligible to receive developmental and regulatory milestone payments, royalties on the sale of assays by bioMérieux and payments for the manufacture and delivery of instruments based on a contractual rate subject to future adjustments.
On December 22, 2016, we entered into an Amended and Restated License Agreement with bioMérieux, or the BMX Agreement, which modified the JDLA resulting in the termination of the ongoing joint development efforts between the parties and clarified and amended prospective rights and obligations of both parties. Under the BMX Agreement, bioMérieux retains an exclusive license to our Simoa technology for in vitro diagnostics used in clinical lab applications, food quality control testing, and pharmaceutical quality control testing, each as defined in the agreement, subject to a right we have retained to make and sell the current version of our HD-1 instrument for use in clinical lab applications, either directly or through a partner (but not both), if an affiliate of ours is manufacturing and selling in vitro diagnostics tests, or solely through a partner (subject to restrictions as to the particular parties with which we could elect to partner and the assays that can be developed) in the event we do not have an affiliate manufacturing and selling in vitro diagnostics tests. For sales by a partner, we would be required to pay to bioMérieux a mid-double-digit percentage of royalties received based on sales of assays by the partner. bioMérieux also retains a co-exclusive license to our Simoa technology for certain in vitro diagnostics, including point-of-care testing and laboratory developed testing. We retained rights to research use only applications and to nucleic acid assay applications. We also granted bioMérieux a non-exclusive, royalty-free license to the source and object code of our Level 1 Data Reduction, or L1DR, software
106
including rights to updates and upgrades in the future. The L1DR software is our proprietary image processing algorithms that convert images of microscopic beads associated with biomarker molecules in microwells. bioMérieux's minimum purchasing requirements were eliminated and it is permitted to independently develop and manufacture certain instruments.
bioMérieux has a three-year option to acquire distribution rights to the HD-1 instrument in the exclusive and co-exclusive fields. If the option to acquire distribution rights to the HD-1 is exercised, the BMX Agreement provides that we and bioMérieux shall negotiate, in good faith, a distribution agreement which will include a specified lump sum payment. If bioMérieux does not exercise this right prior to December 22, 2019, all rights and licenses granted to bioMérieux with respect to the HD-1 instrument (other than the license to the L1DR software) will terminate.
bioMérieux also has a period of not more than three years from the date of the BMX Agreement to evaluate its interest in developing a new, smaller in vitro diagnostic instrument using the Simoa technology for use in clinical lab applications, food quality control testing, and pharmaceutical quality control testing. If bioMérieux does not elect to pursue development of a new instrument within the three year period ending December 22, 2019, all rights and licenses granted to bioMérieux for instruments other than the HD-1 instrument will terminate. If bioMérieux does elect to pursue development of a new instrument, they will have a set number of years to obtain a CE mark for such instrument and a set period of time thereafter to obtain FDA approval. Subject to a cure period, if these regulatory milestones are not met, the BMX Agreement will terminate (subject to the continuing right of bioMérieux to distribute the HD-1 instrument if it had previously exercised its option and negotiated a distribution agreement).
We have been advised by bioMérieux that its current objective is to identify and develop an assay menu supporting the commercial launch of a new, benchtop in-vitro diagnostic instrument using the Simoa technology for use in clinical lab applications, food quality control testing, and pharmaceutical quality control testing. This will require identifying assays that support the commercial launch of such an instrument and developing or adapting technology to facilitate a benchtop platform. Pursuant to the exclusive license to the Simoa technology granted in the BMX Agreement, bioMérieux has the sole right to determine whether or not to develop such an instrument for use in clinical lab applications, food quality control testing, and pharmaceutical quality control testing, and we can not assure you that bioMérieux will decide to do so. If they were to do so, we would be restricted from selling a benchtop instrument for use in clinical lab applications, food quality control testing, and pharmaceutical quality control testing, but not for use in any other applications.
On execution of the BMX Agreement, we received an upfront payment of $2 million. We are also eligible to receive royalties on net sales of assays sold by bioMérieux in the mid to high single digits, and to receive low double digit royalties on sales of instruments by bioMérieux based on manufacturing cost. The future developmental and regulatory milestone payments under the JDLA were not achieved as the parties agreed to no longer pursue joint development, subject to the right for bioMérieux to develop its own instrument using the Simoa technology with a different form and size than our HD-1 instrument, prior to the achievement of these milestones. Accordingly, these milestones were no longer applicable and were removed in the BMX Agreement and are no longer eligible to be earned. The BMX Agreement has an indefinite term, but can be terminated by bioMérieux for any reason with six months notice to us. In addition, either party may terminate the agreement upon 60 days notice in the event of an uncured material breach by the other party.
107
Development agreement and supply agreement with STRATEC
In August 2011, we entered into a Strategic Development Services and Equity Participation Agreement, or the Development Agreement, with STRATEC Biomedical Systems AG, pursuant to which STRATEC undertook the development of the Simoa HD-1 instrument for manufacture and sale to us or a partner whom we designate. Under the Development Agreement, we were required to pay a fee and issue to STRATEC warrants to purchase 2,000,000 shares of our Series A-3 Preferred Stock at an exercise price of $0.001 per share, all of which have been exercised as of September 30, 2017. These fees and warrants were subject to a milestone based payment schedule. The Development Agreement was amended in November 2016. The Amendment reduced our obligation to satisfy a minimum purchase commitment under the Supply and Manufacturing Agreement described below. Additionally, the parties agreed on additional development services for an additional fee, which is payable when the additional development is completed. This fee includes the final milestone payment that was associated with the final milestone due under the terms of the Development Agreement. The services are expected to be completed during the year ending December 31, 2018.
The Development Agreement may be terminated on the insolvency of a party, for an uncured material breach, or, by us, on a change of control of our company (subject to certain obligations to compensate STRATEC on such termination) or if we and STRATEC are unable to agree on pricing of the instrument, within certain parameters.
In September 2011, we also entered into a Supply and Manufacturing Agreement with STRATEC, or the Supply Agreement, pursuant to which STRATEC agreed to supply HD-1 instruments to us, and we agreed to procure those instruments exclusively from STRATEC, subject to STRATEC's ability to supply the instruments. We are responsible for obtaining any regulatory approval necessary to sell the instruments. We agreed to purchase a certain number of instruments in the seven years following the acceptance of the first validation instrument. The Supply Agreement was amended in November 2016 to reduce the number of instruments we are committed to procure from STRATEC. The instrument price stipulated in the Supply Agreement was established based on certain specified assumptions and is subject to certain adjustments.
The Supply Agreement is terminable by either party on twelve months' notice to the other party, provided that neither party may terminate the Supply Agreement prior to the later of the seven year anniversary of the acceptance of the first prototype instrument and the purchase of the minimum number of instruments which we committed to procure. The Supply Agreement may also be terminated on the insolvency of a party or the uncured material breach of a party, or, by us, on a change of control of our company (subject to certain obligations to compensate STRATEC on such termination). On termination by us for STRATEC's insolvency or uncured material breach or termination by STRATEC for convenience, we are granted a nonexclusive royalty free license under STRATEC intellectual property to manufacture the instruments. In certain of these circumstances, we could be obligated to issue warrants to purchase preferred stock.
Paramit Manufacturing Services Agreement
In November 2016, we entered into a Manufacturing Services Agreement, or the Paramit Agreement, with Paramit Corporation, or Paramit. Under the terms of the Paramit Agreement, we engaged Paramit to produce and test our Quanterix SR-X instrument on an as-ordered basis. We also engaged Paramit to supply spare parts. Paramit has no obligation to manufacture our instrument without a purchase order and no obligation to maintain inventory in excess of any open purchase orders or materials in excess of the amount Paramit reasonably determines will be consumed within 90 days or within the lead time of manufacturing our instrument, whichever is greater. We have an obligation to purchase any material or instruments deemed in excess pursuant to the Paramit Agreement. The price is determined according to a
108
mutually agreed-upon pricing formula. The parties agreed to review the pricing methodology yearly or upon a material change in cost.
The Paramit Agreement has an initial three-year term with automatic one year extensions. It is terminable by either party for convenience with nine months' written notice to the other party given at least nine months prior to the end of the then-current term. The agreement may also be terminated by us with three months' notice to Paramit upon the occurrence of (i) a failure of Paramit to obtain any necessary governmental licenses, registrations or approvals required to manufacture our instrument or (ii) an assignment by Paramit of its rights or obligations under the agreement without our consent. The Paramit Agreement is terminable by Paramit with 30 days' notice to us in the event of a material breach after written notice and a 60-day opportunity to cure the breach.
We compete with both established and development-stage life science companies that design, manufacture and market instruments for protein detection, nucleic acid detection and additional applications. For example, companies such as Bio-Techne, Luminex Corporation, MesoScale Diagnostics, Singulex, Gyros Corporation, Nanostring Technologies, Inc., and others, have products for protein detection that compete in certain segments of the market in which we sell our products. As we or our partners expand the applications for our products to include diagnostics and precision health screening, we expect to compete with companies such as Siemens, Abbott, Roche, Ortho Clinical Diagnostics and Thermo Fisher Scientific. In addition, a number of other companies and academic groups are in the process of developing novel technologies for the life science research, diagnostic and precision health screening markets. Many of the companies with which we compete have substantially greater resources than we have.
The life science instrumentation industry is highly competitive and expected to grow more competitive with the increasing knowledge gained from ongoing research and development. We believe the principal competitive factors in our target markets include:
- •
- sensitivity;
- •
- cost of instruments and consumables;
- •
- assay menu;
- •
- reputation among customers and key opinion leaders;
- •
- innovation in product offerings;
- •
- accuracy and reproducibility of results; and
- •
- customer support infrastructure.
We believe that we are well positioned with respect to these competitive factors and expect to enhance our position through ongoing global expansion, innovative new product introductions and ongoing collaborations and partnerships with key opinion leaders.
Our core technology, directed to general methods and devices for single molecule detection, originated at Tufts University, in the laboratory of Professor David Walt, who is the founder of Quanterix and a current member of our Board of Directors. Prof. Walt and his students pioneered the single molecule array
109
technology, including technologies that enabled the detection of single enzyme labels in arrays of microwells, thereby facilitating the ultra-sensitive detection of proteins, nucleic acids, and cells. We have exclusively licensed from Tufts the relevant patent filings related to these technologies. (See "—License agreement with Tufts University" below). In addition to licensed patents, we have developed our own portfolio of issued patents and patent applications directed to commercial products and technologies for potential development. We believe our proprietary platform is a core strength of our business and our strategy includes the continued development of our patent portfolio.
Our patent strategy is multilayered, providing coverage of aspects of the core technology as well as specific uses and applications, some of the foregoing of which are reflected in our current products and some of which are not. The first layer is based on protecting the fundamental methods for detecting single molecules independent of the specific analyte to be detected. The second layer covers embodiments of the core technology directed to the detection of specific analytes. The third layer protects novel instrumentation, consumables, and manufacturing processes used in applying the invention to certain commercial products or future product opportunities. The fourth layer is concerned with specific uses of the core technology (e.g., biomarkers and diagnostics). Our patent strategy is both offensive and defensive in nature; seeking to protect not only technology we currently practice but also alternative, related embodiments.
As of September 30, 2017, we had exclusively licensed 16 patents and two patent applications from Tufts. These patents and patent applications include seven issued U.S. patents and two pending U.S patent applications, three granted European patents, three granted Japanese patents, two granted Canadian patents and one granted Australian patent.
A first patent family licensed from Tufts is directed to methods for detecting single molecules. This patent family includes four granted U.S. patents, two pending U.S. patent applications, three granted European patents, three granted Japanese patents, two granted Canadian patents and one granted Australian patent. The standard patent expiration date for U.S. patents in this family is February 16, 2027, and for the non-U.S. patents is February 20, 2027 or August 30, 2027.
A second patent family licensed from Tufts is directed to methods for detecting the presence of target analytes in multiple samples. This patent family includes one granted U.S. patent. The standard patent expiration date for the U.S. patent in this family is August 22, 2025.
A third patent family licensed from Tufts is directed to methods for analyzing analytes using a sensor system with cross-reactive elements. This patent family includes one granted U.S. patent. The standard patent expiration date for the U.S. patent in this family is March 14, 2021.
A fourth patent family licensed from Tufts is directed to electro-optical systems including an array and a plurality of electrodes. This patent family includes one granted U.S. patent. The standard patent expiration date for the U.S. patent in this family is February 14, 2023.
As of September 30, 2017, we owned nine issued U.S. patents and twelve pending U.S. patent applications, two granted European patents and three pending European patent applications, five granted Japanese patents and one pending Japanese patent applications, two granted Chinese patents and four pending Chinese patent applications, two granted Canadian patents and one pending Canadian patent application, and two registered Hong Kong patent applications.
A first patent family owned by us is directed to methods for determining a measure of the concentration of analyte molecules or particles in a fluid sample, and in particular to methods for analyte capture on beads,
110
including multiplexing. This patent family includes three granted U.S. patents and one pending U.S. patent application, one granted European patent (nationalized in eight countries) and one pending European application, two granted Japanese patents, one granted Chinese patent and one pending Chinese patent application, and one granted Canadian patent. The standard patent expiration date for the U.S. patents in this family is March 24, 2030, and for the non-U.S. patents is March 1, 2031.
A second patent family owned by us is directed to methods and systems for determining a measure of the concentration of analyte molecules or particles in a fluid sample, and in particular to methods or systems for determining concentration based on either counting or measured intensity (extending the dynamic range). This patent family includes three granted U.S. patents and one pending U.S. patent application, one granted European patent (nationalized in seven countries), two granted Japanese patents, one granted Chinese patent, and one granted Canadian patent. The standard patent expiration date for the U.S. patents in this family is March 24, 2030, and for the non-U.S. patents is March 1, 2031.
A third patent family owned by us is directed to methods for determining a measure of the concentration of analyte molecules or particles in a fluid sample, and in particular to methods for analyte capture on beads with or without dissociation. This patent family includes two granted U.S. patents. The standard patent expiration date for the U.S. patents in this family is September 28, 2028.
A fourth patent family owned by us is directed to methods for determining a measure of the concentration of analyte molecules or particles in a fluid sample, and in particular to methods for determining concentration using multiple binding ligands for the same analyte molecule. This patent family includes one granted U.S. patent. The standard patent expiration date for the U.S. patent in this family is March 24, 2030.
A fifth patent family owned by us is directed to instruments and consumables. This patent family includes one granted Japanese patent and one pending Japanese patent application, three pending Chinese patent applications, two registered Hong Kong patent applications and one pending patent application in each of the U.S., Europe, and Canada. The standard patent expiration date for any U.S. patents that may issue from this family is February 25, 2031, and for any non-U.S. patents is January 27, 2032.
A sixth patent family owned by us is directed to methods and materials for covalently associating a molecular species with a surface. This patent family includes one pending U.S. patent application. The standard patent expiration date for any U.S. patents that may issue from this family is May 9, 2034.
A seventh patent family owned by us is directed to methods for improving the accuracy of capture based assays. This patent family includes one pending U.S. patent application. The standard patent expiration date for any U.S. patents that may issue from this family is January 13, 2036.
An eighth patent family owned by us is directed to methods and systems for reducing and/or preventing signal decay. This patent family includes one pending U.S. provisional patent application. If we pursue protection by filing any non-provisional applications, the standard patent expiration date for any patents that may issue from this family will be in 2038.
We own or co-own six additional patent families directed to the measurement of particular types of analytes, including prostate specific antigen (PSA), b-amyloid peptide, tau protein, toxin B of C. difficile, and DNA or RNA molecules. Any patents that may issue from these patent applications would have standard expiration dates between 2032 and 2038.
We have licensed additional patents and patent applications from third parties.
111
In addition to pursuing patents on our technology, we have taken steps to protect our intellectual property and proprietary technology by entering into confidentiality agreements and intellectual property assignment agreements with our employees, consultants, corporate partners and, when needed, our advisors.
License agreement with Tufts University
In June 2007, as amended in April 2013, we entered into a license agreement with Tufts University, or Tufts, pursuant to which we obtained an exclusive, worldwide license to research, develop, commercialize, use, make, or have made, import or have imported, distribute or have distributed, offer or have offered, and sell or have sold products and services covered by patent rights to the Simoa technology owned by Tufts, as well as a non-exclusive license to related know-how. The rights licensed to us are for all fields of use and are sublicensable for a fee.
Under the terms of the agreement, as amended, we paid a one-time, non-refundable upfront fee and issued Tufts shares of our common stock. In addition, in connection with the April 2013 amendment, we issued Tufts shares of our Series C-1 Preferred Stock. We are required to pay Tufts low single-digit royalties on all net sales of products and services as well as a portion of any sublicensing revenues. We are also obligated to pay annual maintenance fees, which are fully creditable against any royalty payments made by us, and a milestone payment upon any sublicense by us. We were also required to reimburse Tufts for all patent prosecution cost incurred prior to the agreement and for all future patent prosecution costs.
The term of the license agreement will continue on a country-by-country basis so long as there is a valid claim of a licensed patent in such country. Tufts may terminate the agreement or convert to a non-exclusive license in the event (1) we fail to pay any undisputed amount when required and fail to cure such non-payment within 60 days after receipt of notice from Tufts, (2) we are in breach of any material provision of the agreement and fail to remedy such breach within 60 days after receipt of notice from Tufts, (3) we do not demonstrate diligent efforts to develop a product incorporating the licensed technology, (4) we are found on five separate audits to have underpaid pursuant to the terms of the agreement, (5) we cease to carry on the business related to the licensed technology either directly or indirectly, or (6) we are adjudged insolvent, make an assignment for the benefit of creditors or have a petition in bankruptcy filed for or against us that is not removed within 60 days. We may terminate the agreement at any time upon at least 60 days' written notice. Upon termination of the agreement, all rights revert to Tufts.
Our products are currently intended for research use only, or RUO, applications, although our customers may use our products to develop their own products that are subject to regulation by the FDA. Although most products intended for RUO are not currently subject to clearance or approval by the FDA, RUO products fall under the FDA's jurisdiction if they are used for clinical rather than research purposes. Consequently, our products are labeled "For Research Use Only."
On November 25, 2013, the FDA issued Final Guidance for Industry and Food and Drug Administration Staff on "Distribution of In Vitro Diagnostic Products Labeled for Research Use Only or Investigational Use Only," or, the RUO/IUO Guidance. The purpose of an FDA guidance document is to provide the FDA's current thinking on when IVD products are properly labeled for RUO or for IUO, but as with all FDA's guidance documents, this guidance does not establish legally enforceable responsibilities and should be viewed as recommendations unless specific regulatory or statutory requirements are cited. The RUO/IUO Guidance explains that the FDA will review the totality of the circumstances when evaluating whether equipment and
112
testing components are properly labeled as RUO. Merely including a labeling statement that a product is intended for research use only will not necessarily exempt the device exempt from the FDA's 510(k) clearance, premarket approval, or other requirements, if the circumstances surrounding the distribution of the product indicate that the manufacturer intends its product to be used for clinical diagnostic use. These circumstances may include written or verbal marketing claims or links to articles regarding a product's performance in clinical applications, a manufacturer's provision of technical support for clinical validation or clinical applications, or solicitation of business from clinical laboratories, all of which could be considered evidence of intended uses that conflict with RUO labeling. Although the RUO/IUO Guidance is a statement of the FDA's thinking with respect to certain RUOs and IUOs in 2013 and was not intended as a compliance requirement, we believe that our labeling and promotion of our products is consistent with the RUO/IUO Guidance because we have not promoted our products for clinical use in humans. When we develop products for clinical use, we will do so in accordance with FDA requirements at that time.
When our products are marketed for clinical diagnostic use, our products will be regulated by the FDA as medical devices. The FDA defines a medical device in part as an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article which is intended for the diagnosis of disease or other conditions or in the cure, mitigation, treatment, or prevention of disease in man. This means that the FDA will regulate the development, testing, manufacturing, marketing, post-market surveillance, distribution, advertising and labeling of our clinical products and we will be required to register as a medical device manufacturer and list our marketed products.
The FDA classifies medical devices into one of three classes on the basis of the intended use of the device, the risk associated with the use of the device for that indication, as determined by the FDA, and on the controls deemed by the FDA to be necessary to reasonably ensure their safety and effectiveness. Class I devices, which have the lowest level of risk associated with them, are subject to general controls. Class II devices are subject to general controls and special controls, including performance standards. Class III devices, which have the highest level of risk associated with them, are subject to general controls and premarket approval. Most Class I devices and some Class II devices are exempt from a requirement that the manufacturer submit a premarket notification, or 510(k), and receive clearance from the FDA which is otherwise a premarketing requirement for a Class II device. Class III devices may not be commercialized until a premarket approval application, or PMA, is submitted to and approved by the FDA.
510(k) clearance pathway
To obtain 510(k) clearance, a sponsor must submit to the FDA a premarket notification demonstrating that the device is substantially equivalent, or SE, to a device legally marketed in the U.S. for which a PMA was not required. The FDA is supposed to make a SE determination within 90 days of FDA's receipt of the 510(k), but it often takes longer if the FDA requests additional information. Most 510(k)s do not require supporting data from clinical trials, but the FDA may request such data.
Premarket approval pathway
A PMA must be submitted if a new device cannot be cleared through the 510(k) process. The PMA process is generally more complex, costly and time consuming than the 510(k) process. A PMA must be supported by extensive data including, but not limited to, technical, preclinical, clinical trials, manufacturing and labeling to demonstrate to the FDA's satisfaction the safety and effectiveness of the device for its intended use. After a PMA is sufficiently complete, the FDA will accept the application for filing and begin an in-depth review of the submitted information. By statute, the FDA has 180 days to review the accepted application, although, review of the application generally can take between one and three years. During this review period, the FDA may request additional information or clarification of information already
113
provided. Also during the review period, an advisory panel of experts from outside the FDA may be convened to review and evaluate the application and provide recommendations to the FDA as to the approvability of the device. In addition, the FDA will conduct a preapproval inspection of the manufacturing facility to ensure compliance with its quality system regulations, or QSRs. New premarket approval applications or premarket approval application supplements are also required for product modifications that affect the safety and efficacy of the device.
Clinical trials
Clinical trials are usually required to support a PMA and are sometimes required for a 510(k). In the U.S., if the device is determined to present a "significant risk," the manufacturer may not begin a clinical trial until it submits an investigational device exemption application, or IDE, and obtains approval of the IDE from the FDA. These clinical trials are also subject to the review, approval and oversight of an institutional review board, or IRB, at each clinical trial site. The clinical trials must be conducted in accordance with the FDA's IDE regulations and good clinical practices. A clinical trial may be suspended by FDA, the sponsor or an IRB at its institution at any time for various reasons, including a belief that the risks to the study participants outweigh the benefits of participation in the trial. Even if a clinical trial is completed, the results may not demonstrate the safety and efficacy of a device to the satisfaction of the FDA, or may be equivocal or otherwise not be sufficient to obtain approval of a device.
After a medical device is placed on the market, numerous regulatory requirements apply. These include among other things:
- •
- compliance with QSRs, which require manufacturers to follow stringent design, testing, control, documentation, record maintenance, including
maintenance of complaint and related investigation files, and other quality assurance controls during the manufacturing process;
- •
- reporting of device malfunctions, serious injuries or deaths;
- •
- registration of the establishments where the devices are produced;
- •
- labeling regulations, which prohibit the promotion of products for uncleared or unapproved uses; and
- •
- medical device reporting obligations, which require that manufacturers investigate and report to the FDA adverse events, including deaths, or serious injuries that may have been or were caused by a medical device and malfunctions in the device that would likely cause or contribute to a death or serious injury if it were to recur.
Failure to comply with applicable regulatory requirements can result in enforcement action by the FDA, which may include sanctions, including but not limited to, warning letters; fines, injunctions, and civil penalties; recall or seizure of the device; operating restrictions, partial suspension or total shutdown of production; refusal to grant 510(k) clearance or PMA approvals of new devices; withdrawal of 510(k) clearance or PMA approvals; and civil or criminal prosecution. To ensure compliance with regulatory requirements, medical device manufacturers are subject to market surveillance and periodic, pre-scheduled and unannounced inspections by the FDA.
Laboratories that purchase certain of our products and perform clinical diagnostic testing are also subject to extensive regulation under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), requiring clinical laboratories to meet specified standards in areas such as personnel qualifications, administration, participation in proficiency testing, patient test management, quality control, quality assurance and inspections. Adverse interpretations of current CLIA regulations or future changes in CLIA regulations could
114
have an adverse effect on sales of any affected products. Moreover, if we decide to operate our own clinical testing laboratory, we will be required to comply with CLIA. If, in the future, we operate our own clinical laboratory to perform clinical diagnostic testing, we would become subject to the Health Insurance Portability and Accountability Act of 1996, or HIPAA, as well as additional federal and state laws that impose a variety of fraud and abuse prohibitions on healthcare providers, including clinical laboratories.
Europe/rest of world government regulation
Whether or not we obtain FDA approval for a product, we must obtain the requisite approvals from regulatory authorities in non-U.S. countries prior to the commencement of clinical trials or marketing of our product for clinical diagnostic use in those countries. The regulations in other jurisdictions vary from those in the U.S. and may be easier or more difficult to satisfy and are subject to change. For example, the European Union, or EU, recently published new regulations that will result in greater regulation of medical devices and IVDs. The IVD Regulation is significantly different from the IVD Directive that it replaces in that it will ensure that the new requirements apply uniformly and on the same schedule across the member states, include a risk-based classification system and increase the requirements for conformity assessment. The conformity assessment process results in the receipt of a CE designation which has been sufficient to begin marketing many types of IVDs. That process will become more difficult and costly to complete.
Other governmental regulation
We are subject to laws and regulations related to the protection of the environment, the health and safety of employees and the handling, transportation and disposal of medical specimens, infectious and hazardous waste and radioactive materials. For example, the U.S. Occupational Safety and Health Administration (OSHA), has established extensive requirements relating specifically to workplace safety for healthcare employers in the U.S. This includes requirements to develop and implement multi-faceted programs to protect workers from exposure to blood-borne pathogens, including preventing or minimizing any exposure through needle stick injuries. For purposes of transportation, some biological materials and laboratory supplies are classified as hazardous materials and are subject to regulation by one or more of the following agencies: the U.S. Department of Transportation, the U.S. Public Health Service, the United States Postal Service and the International Air Transport Association. We generally use third-party vendors to dispose of regulated medical waste, hazardous waste and radioactive materials that we may use during our research.
As of September 30, 2017, we had 122 employees, of which 40 work in sales, sales support and marketing, 44 work in engineering and research and development, 21 work in manufacturing and operations and 17 work in general and administrative. As of September 30, 2017, of our 122 employees, 111 were located in the United States and 11 were employed outside the United States. None of our employees is represented by a labor union or is subject to a collective bargaining agreement.
We lease approximately 30,655 square feet of office, laboratory, and manufacturing space at our headquarters in Lexington, Massachusetts, under a lease that expires June 30, 2020. We believe that we will need additional space as we grow our operations, but believe that suitable additional or substitute space will be available to accommodate future growth of our business. We believe that our existing office, laboratory and manufacturing space will be sufficient to meet our needs in the interim.
115
Executive officers and directors
The following table provides information regarding our executive officers and directors as of November 7, 2017:
| |
|
|
|||
|---|---|---|---|---|---|
| | | | | | |
| Name |
Age |
Position |
|||
| | | | | | |
Executive Officers: |
|||||
E. Kevin Hrusovsky(1) |
56 | Executive Chairman, President and Chief Executive Officer | |||
Joseph Driscoll |
52 | Chief Financial Officer | |||
Ernest Orticerio |
54 | Senior Vice President, Finance and Corporate Development | |||
Bruce Bal |
59 | Vice President of Operations, Service and Quality | |||
David C. Duffy, Ph.D. |
46 | Senior Vice President, Research & Development and Chief Technology Officer | |||
Mark T. Roskey, Ph.D. |
58 | Senior Vice President and General Manager of Applications and Reagents | |||
Non-Employee Directors(1): |
|
||||
Douglas G. Cole, M.D.(1) |
57 | Director | |||
John M. Connolly(1) |
65 | Director | |||
Keith L. Crandell(1) |
57 | Director | |||
Marijn Dekkers, Ph.D.(1) |
60 | Director | |||
Martin D. Madaus, Ph.D.(1) |
58 | Director | |||
Paul M. Meister(1) |
65 | Director | |||
David R. Walt, Ph.D.(1) |
64 | Director | |||
| | | | | | |
(1) See "Certain relationships and related party transactions—Agreements with stockholders" for a discussion of arrangements among our stockholders pursuant to which this director was selected.
Executive officers
E. Kevin Hrusovsky has been our Executive Chairman and a member of our board of directors since June 2014 and has been our President and Chief Executive Officer since January 2015. Prior to joining us, Mr. Hrusovsky served as Senior Vice President of PerkinElmer, Inc., a publicly traded company that produces analytical instruments, genetic testing and diagnostic tools, medical imaging components, software, instruments and consumables for multiple end markets from February 2012 to May 2013 and served as President of the Life Sciences and Technology business unit of PerkinElmer, Inc. from November 2011 to May 2013. From May 2013 through September 2013, he served as a consultant to PerkinElmer. Previously, Mr. Hrusovsky served as Chief Executive Officer and President of Caliper Life Sciences, Inc., a life sciences company that developed and sold lab automation equipment, from July 2003 to November 2011 when it was acquired by PerkinElmer, Inc. Prior to that, he served as Chief Executive Officer and President of Zymark, a company that provided laboratory automation, robotics, and liquid handling solutions, and Director of International Business, Agricultural Chemical Division, and President of the Pharmaceutical Division for FMC Corporation, a publicly traded chemical manufacturing company. He also held several management positions at E.I. DuPont de Nemours, a publicly traded chemical manufacturing company. Mr. Hrusovsky serves on the boards of directors of several private companies, including BioreclamationIVT, LLC, Cellaria, Cell Signaling Technology, Inc., 908 Devices Inc., and Solect Energy Development, LLC., and is the founder of the Powering Precision Health Summit, an executive think tank of
116
researchers, scientists, physicians and innovators focused on the latest research on new biomarkers that have the potential to advance personalized medicine. Mr. Hrusovsky also serves on the Educational Board of the Massachusetts Biotech Council, the Advisory Committee for the Center for Biomedical Engineering at Brown University, the Association for Laboratory Automation, the JALA Editorial Board and the Strategy Committee of Children's Hospital Boston. He formerly served on the boards of SynapDx Corp., SeraCare, Caliper Life Sciences, Xenogen—XGEN and Alliant Medical Technology. He was selected as the 2013 Entrepreneur of the Year from Ohio State University and holds an Honorary Doctorate degree from Framingham State University for contributions in life sciences and personal medicine. Mr. Hrusovsky has a B.S. in mechanical engineering from Ohio State University and an M.B.A. from Ohio University. Our board of directors has concluded that Mr. Hrusovsky possesses specific attributes that qualify him to serve as a member of our board of directors, including the perspective and experience he brings as our President and Chief Executive Officer, which provides our board with historic knowledge, operational expertise and continuity.
Joseph Driscoll has been our Chief Financial Officer since April 2017. Prior to that, Mr. Driscoll served as Chief Financial Officer of Verscend Technologies, Inc., a healthcare data analytics company, from October 2016 to April 2017. From March 2012 to October 2016, he served as the Chief Financial Officer, Senior Vice President and Treasurer of PC Connection, Inc., an IT solutions provider, where he also served as the company's Principal Financial and Accounting Officer. From September 2006 to March 2012, Mr. Driscoll served as the Chief Financial Officer of Summer Infant, Inc., a consumer products company, where he also served as the company's Treasurer and Principal Accounting Officer. From May 2001 to September 2006, Mr. Driscoll served as a Vice President of Finance and Chief Financial Officer of ACT Electronics, Inc., an electronics contract manufacturer. From May 2000 to May 2001, Mr. Driscoll served as Vice President of Finance at PCI, Inc., a marketing software company. From April 1997 to May 2000, he served as a Vice President of Finance and Chief Financial Officer of Safety 1st, Inc. He joined Safety 1st Inc. in April 1997 as Controller and served as the Chief Financial Officer from September 1998 to May 2000. From September 1993 to April 1997, Mr. Driscoll served as an Assistant Corporate Controller and Director of Financial Reporting at Staples, Inc., an office supply retailer. From July 1986 to February 1992, Mr. Driscoll served as an Audit Manager of KPMG Peat Marwick, a national accounting firm. From February 1992 to September 1993, Mr. Driscoll served as Corporate Controller of E-II Holdings, Inc., a diversified holding company. He is a licensed Certified Public Accountant, and holds a B.S. in Accounting from Boston College.
Ernest Orticerio has been our Senior Vice President, Finance and Business Development since April 2017. Prior to that he served as Vice President, Chief Financial Officer from May 2016 to March 2017, Vice President, Chief Financial Officer and Vice President of Operations from January 2013 to April 2016, and our Chief Financial Officer from January 2012 to December 2012. Prior to joining us, he served in a number of accounting, finance and operations capacities with Millipore Corporation, a publicly traded global life science tools and services company, including as Vice President, Finance—International and Corporate Operations from July 2009 to July 2011, Vice President Finance—Corporate Operations from January 2008 to June 2009, Vice President, Finance—Global Operations from July 2005 to December 2007, and Vice President, Global Customer Service from January 2004 to June 2005. He started his career with McDonald's Corporation, a publicly traded leading global food service retailer, holding a number of progressive accounting and finance positions supporting operations, marketing and real estate functions. He has a B.S. in business administration from Bryant College.
Bruce Bal has been our Vice President of Operations, Service and Quality since May 2016. Prior to joining Quanterix, Mr. Bal served as Vice President of Operations at PerkinElmer, a leading provider of instruments and reagents to the human and environmental health markets, following the acquisition of Caliper Life
117
Sciences where he held the role of Senior Vice President of Operations from November 2011 to May 2016. Mr. Bal joined Caliper Life Sciences, a leading provider of drug discovery and life sciences research solutions for the pharmaceutical and biotechnology industries, following the acquisition of Zymark, where he served as Vice President of R&D and Operations from April 1997 to July 2003. He worked as Director of Operations in the Biotechnology Division of FMC Corporation, a diversified chemical company with leading positions in agricultural, industrial and consumer markets, from 1995 to 1997. Between 1981 and 1993, Mr. Bal held a range of management positions at DuPont, a global leader in providing a wide range of innovative products and services for multiple global markets, and was general manager of United States Pollution Control, Inc. Mr. Bal holds a B.S. in Chemical Engineering from the University of Wisconsin and an M.B.A. from Loyola University, Louisiana.
David C. Duffy, Ph.D. is Senior Vice President, Research & Development, and has been our Chief Technology Officer since March 2013. Prior to this role, Dr. Duffy served as our Vice President, Research from November 2011 to February 2013, and Senior Director, Platform Research from July 2007 to October 2011. Prior to joining us, Dr. Duffy served as Director, Pharmacomer Technology at Surface Logix, Inc., a biomedical development company. Prior to that, Dr. Duffy was a Senior Scientist at Gamera Bioscience Corporation, a developer of automated biotechnology test equipment, from December 1998 to January 2000. Previously, Dr. Duffy was a postdoctoral research fellow in the Department of Chemistry and Chemical Biology at Harvard University. He was the first Sir Alan Wilson Research Fellow of Emmanuel College, University of Cambridge. Dr. Duffy has a Ph.D. in physical chemistry from the Department of Chemistry at the University of Cambridge, and B.A. and M.A. degrees in Natural Sciences from the University of Cambridge.
Mark T. Roskey, Ph.D., is Senior Vice President and General Manager of Applications and Reagents since September 2014. Prior to joining us, Dr. Roskey served as Vice President and General Manager of the America's Sales and Service organization in PerkinElmer's Life Science and Technologies Division from December 2011 to September 2014. PerkinElmer is a global life science company. Prior to PerkinElmer, Dr. Roskey served as Senior Vice President of Biology Research and Development at Caliper Life Sciences, Inc. from March 2009 to November 2011 when it was acquired by PerkinElmer. At Caliper Life Sciences, Inc., Dr. Roskey served as Senior Vice President of Applied Biology Research from 2008 to 2011 and Vice President of Worldwide Marketing from July 2003 to 2008. Prior to that, Dr. Roskey served as Vice President of Worldwide Marketing for Zymark Corporation, a laboratory automation company, from December 2001 to August 2003 when it was acquired by Caliper Life Sciences, Inc. Prior to that, Dr. Roskey served as Director of Marketing at Applied Biosystems, Inc. Dr. Roskey completed a postdoctoral fellowship in molecular immunobiology at the Harvard Medical School and has a Ph.D. in microbiology from the University of Notre Dame and a B.S. in biology from Framingham State University.
Non-employee directors
Douglas G. Cole, M.D. has served as a member of our board of directors since June 2007. Dr. Cole is a managing partner of venture firm Flagship Pioneering, where he has focused on life science investments since 2001. He currently serves on the board of directors of Editas Medicine, Inc., a public biopharmaceutical company. He also serves on the boards of directors of several private biopharmaceutical and diagnostics companies, including Denali Therapeutics, Inc., Ensemble Therapeutics Corporation, KSQ Therapeutics, Inc. and Torque Therapeutics, Inc. In the past five years, Dr. Cole has served on the boards of the following public biopharmaceutical companies: Agios Pharmaceuticals, Inc., Concert Pharmaceuticals, Inc., Receptos, Inc., which was acquired by Celgene, Inc., Tetraphase Pharmaceuticals, Inc. and of the following private biopharmaceutical companies: Avedro, Inc., Moderna Therapeutics, Resolvyx Pharmaceuticals, Inc., Selecta Biosciences, Inc., Seventh Sense Biosystems, Inc. and Syros
118
Pharmaceuticals Inc. Dr. Cole has a B.A. in English from Dartmouth College and an M.D. from the University of Pennsylvania School of Medicine. Our board of directors has concluded that Dr. Cole possesses specific attributes that qualify him to serve as a member of our board of directors, including his substantial experience as an investor in emerging biopharmaceutical and life sciences companies as well as his experience serving on the board of directors for several biopharmaceutical companies.
John M. Connolly has served as a member of our board of directors since December 2012. Since December 2015, Mr. Connolly has served as a senior advisor for Bain Capital Ventures, a venture capital firm he joined in 2009. Mr. Connolly was previously a managing director at Bain Capital Ventures from September 2009 to January 2015. Mr. Connolly also served as Interim Chief Executive Officer of Education Holdings 1, Inc. from May 2012 to September 2012, Interim President and Chief Executive Officer of The Princeton Review from March 2011 to April 2012, Chairman of the board of directors of EDGAR Online, Inc. from March 2011 to August 2012 and Interim President and Chief Executive Officer of EDGAR Online, Inc. from September 2010 to March 2011. Prior to that, Mr. Connolly served as President and Chief Executive Officer of M|C Communications, Inc., a leading provider of continuing medical education in the United States, from March 2007 to July 2009, and as Chairman of the board of directors of M|C Communications, Inc. from August 2009 to August 2010. Prior to that, from March 2004 to March 2007, Mr. Connolly served as President and Chief Executive Officer of Institutional Shareholder Services Inc., which provides proxy voting, corporate governance, compliance, and risk management solutions. Education Holdings 1, Inc. filed a voluntary chapter 11 bankruptcy petition in January 2013. Mr. Connolly has a B.A. from St. Norbert College and an Executive Education Degree from the Executive Education Program at INSEAD which is located in Fontainebleau, France. Our board of directors has concluded that Mr. Connolly possesses specific attributes that qualify him to serve as a member of our board of directors, including his substantial experience as an investor in emerging companies as well as his significant managerial and corporate governance experience.
Keith L. Crandell has served as a member of our board of directors since June 2007. Since July 1994, Mr. Crandell has served as a managing director of ARCH Venture Partners, a venture capital firm focused on early-stage technology companies. He serves on the board of directors of Adesto Technologies Corporation, a publicly-traded provider of application-specific and ultra-low power non-volatile memory products. He is also a director of several private companies and he also serves as a director of the Illinois Venture Capital Association. Mr. Crandell has a B.S. degree in chemistry and mathematics from St. Lawrence University, an M.S. degree in chemistry from the University of Texas at Arlington and an M.B.A. from the University of Chicago. Our board of directors has concluded that Mr. Crandell possesses specific attributes that qualify him to serve as a member of our board of directors, including his financial expertise and his substantial experience as an investor in emerging companies.
Marijn Dekkers, Ph.D. has served as a member of our board of directors since March 2017. Since April 2016, he has served as Chairman of the board of directors of Unilever. From 2010 to 2016, Dr. Dekkers was Chief Executive Officer of Bayer AG in Leverkusen, Germany. From 2000 to 2002, he was the Chief Operating Officer at Thermo Electron Corporation (later renamed Thermo Fisher Scientific Inc.). In 2002, he became Thermo Fisher's President and Chief Executive Officer, where he served until 2009. Dr. Dekkers began his career in 1985 as a research scientist at General Electric, gaining experience in various units of the company before joining AlliedSignal (subsequently Honeywell International) in 1995. Dr. Dekkers currently serves on the board of directors of General Electric and Unilever N.V. He holds a degree in chemistry from Radboud University in Nijmegen, the Netherlands, and a Ph.D. in chemical engineering from the University of Eindhoven. Our board of directors has concluded that Dr. Dekkers possesses specific attributes that qualify him to serve as a member of our board of directors, including his substantial
119
experience leading commercial stage healthcare companies and his significant corporate governance experience.
Martin D. Madaus, Ph.D. has served as a member of our board of directors since November 2010. Dr. Madaus previously served as our Executive Chairman from November 2010 to June 2014, as our Chief Executive Officer from October 2011 to July 2012 and as our President from June 2011 to July 2012. Since June 2014, Dr. Madaus is serving as Chairman and Chief Executive Officer at Ortho-Clinical Diagnostics, Inc., a diagnostics company that makes products and diagnostic equipment for blood testing. Previously, Dr. Madaus was the Chairman, President and Chief Executive Officer of Millipore Corporation (MIL), a life sciences company serving the bioscience research and biopharmaceutical manufacturing industry, from January 2005 to July 2010, when Millipore was acquired by Merck KGaA. From July 2009 to May 2015, Dr. Madaus served as a member of the board of directors of Mettler Toledo International, a manufacturer of scales and analytical instruments. Dr. Madaus received a Doctor of Veterinary Medicine from the University of Munich in Germany and a Ph.D. in Veterinary Medicine from the Veterinary School of Hanover in Germany. Dr. Madaus has extensive public and private company board experience and our board of directors has concluded that Dr. Madaus possesses specific attributes that qualify him to serve as a member of our board of directors, including his substantial knowledge of and managerial experience in the diagnostics industry.
Paul M. Meister has served as a member of our board of directors since September 2013. Mr. Meister is President of MacAndrews & Forbes Holdings Inc., a holding company with interests in a diversified portfolio of public and private companies. He is also Co-Founder of Liberty Lane Partners, LLC, a private investment company with investments in healthcare, technology, and distribution-related industries, and Perspecta Trust, a trust company that provides trust and investment services. From August 2010 to September 2014, Mr. Meister served as Chief Executive Officer of iVentiv Health, a leading provider of commercial, consulting and clinical research services to the pharmaceutical and biotech industries, and as its Chairman from August 2010 to February 2014. From November 2006 to April 2007, he was Chairman of the board of directors of Thermo Fisher Scientific Inc., a provider of products and services to businesses and institutions in the field of science, which was formed by the merger of Fisher Scientific International Inc. and Thermo Electron Corporation in November 2006. Mr. Meister was Vice Chairman of Fisher Scientific International Inc. from 2001 to 2006, and served as its chief financial officer from 1991 to 2001. Fisher Scientific International provided products and services to research, healthcare, industrial, educational and government markets. Mr. Meister is a member of the board of directors of Scientific Games Corporation, which provides customized, end-to-end solutions to the gaming industry, LKQ Corporation, a global distributor of vehicle products, and inVentiv Health. Mr. Meister is Co-Chair of the University of Michigan's Life Sciences Institute External Advisory Board and serves on the Executive Advisory Board of the Chemistry of Life Processes Institute at Northwestern University. Mr. Meister has an M.B.A. from Northwestern University and a B.A. from the University of Michigan. Our board of directors has concluded that Mr. Meister possesses specific attributes that qualify him to serve as a member of our board of directors, including his financial and investment expertise and his extensive knowledge of the life sciences industry.
David R. Walt, Ph.D. has served as a member of our board of directors since April 2007. Dr. Walt was our founding scientist and serves as chairman of our scientific advisory board. Dr. Walt currently serves as a faculty member at Harvard Medical School in the Department of Pathology at the Brigham and Women's Hospital and is a core faculty member of the Wyss Institute for Biologically Inspired Engineering. He is also a Howard Hughes Medical Institute Professor. He previously served as University Professor, Professor of Biomedical Engineering, Professor of Genetics, Professor of Neuroscience, Professor of Cell and Molecular
120
Biology, and Professor of Oral Medicine at Tufts University, from 1981-2017. Dr. Walt was also the founding scientist of Illumina, Inc. and served as a member of the board of its directors from 1998-2016 and now serves as chairman of its scientific advisory board. He was also a founder and currently serves as a member of the board of directors of Ultivue, Inc., and Arbor, Inc., and serves as a member of the board of directors of Exicure, Inc. He has received numerous national and international awards and honors for his fundamental and applied work in the field of optical sensors, microwell arrays and single molecule detection. He is a member of the U.S. National Academy of Engineering, U.S. National Academy of Medicine, American Academy of Arts and Sciences, a fellow of the American Institute for Medical and Biological Engineering, a fellow of the National Academy of Inventors, and a fellow of the American Association for the Advancement of Science. Dr. Walt has a B.S. in Chemistry from the University of Michigan and a Ph.D. in Chemical Biology from Stony Brook University. Our board of directors has concluded that Dr. Walt possesses specific attributes that qualify him to serve as a member of our board of directors, including his experience in developing life sciences companies and his expertise in chemistry, diagnostics technologies and biomedical engineering.
As of November 7, 2017, our board of directors consisted of eight members, all of whom are members pursuant to the board composition provisions of our Fourth Amended and Restated Stockholders Agreement, which is described under "Certain relationships and related party transactions—Agreements with stockholders" in this prospectus. These board composition provisions will terminate upon the completion of this offering. Upon the termination of these provisions, there will be no further contractual obligations regarding the election of our directors. Our nominating and governance committee and our board of directors may therefore consider a broad range of factors relating to the qualifications and background of nominees, which may include diversity, which is not only limited to race, gender or national origin. We have no formal policy regarding board diversity. Our nominating and governance committee's and our board of directors' priority in selecting board members is the identification of persons who will further the interests of our stockholders through his or her established record of professional accomplishment, the ability to contribute positively to the collaborative culture among board members, knowledge of our business, understanding of the competitive landscape and professional and personal experiences and expertise relevant to our growth strategy. Our directors hold office until their successors have been elected and qualified or until the earlier of their death, resignation or removal. Our restated certificate of incorporation and restated by-laws that will become effective upon the completion of this offering also provide that our directors may be removed only for cause by the affirmative vote of the holders of at least 75% of the votes that all our stockholders would be entitled to cast in an annual election of directors, and that any vacancy on our board of directors, including a vacancy resulting from an enlargement of our board of directors, may be filled only by vote of a majority of our directors then in office.
Director independence
Our board of directors has determined that all members of our board of directors, except E. Kevin Hrusovsky, are independent directors, including for purposes of the rules of The Nasdaq Stock Market and relevant federal securities laws and regulations. There are no family relationships among any of our directors or executive officers.
121
Staggered board
In accordance with the terms of our restated certificate of incorporation and restated by-laws that will become effective upon the completion of this offering, our board of directors will be divided into three staggered classes of directors of the same or nearly the same number and each will be assigned to one of the three classes. At each annual meeting of the stockholders, a class of directors will be elected for a three-year term to succeed the directors of the same class whose terms are then expiring. The terms of the directors will expire upon the election and qualification of successor directors at the annual meeting of stockholders to be held during the years 2018 for Class I directors, 2019 for Class II directors and 2020 for Class III directors:
- •
- our Class I directors will be , and ;
- •
- our Class II directors will be , and ; and
- •
- our Class III directors will be , and .
Our restated certificate of incorporation and restated by-laws provide that the number of our directors shall be fixed from time to time by a resolution of the majority of our board of directors. Any additional directorships resulting from an increase in the number of directors will be distributed among the three classes so that, as nearly as possible, each class shall consist of one third of the board of directors.
The division of our board of directors into three classes with staggered three-year terms may delay or prevent stockholder efforts to effect a change of our management or a change in control.
Committees of the board of directors
Our board of directors has an audit committee, a compensation committee and a nominating and governance committee, each of which will have the composition and responsibilities described below upon completion of this offering. Each of the below committees will have a written charter approved by our board of directors, effective upon completion of the offering. Each of the committees will report to our board of directors as such committee deems appropriate and as our board of directors may request. Upon completion of this offering, copies of each charter will be posted on the investor relations section of our website. Members serve on these committees until their resignation or until otherwise determined by our board of directors.
Audit Committee
Effective upon completion of this offering, our audit committee will be comprised of , and , with serving as chairman of the committee. Our board of directors has determined that each member of the audit committee meets the independence requirements of Rule 10A-3 under the Exchange Act and the applicable Nasdaq Stock Market rules, and has sufficient knowledge in financial and auditing matters to serve on the audit committee. Our board of directors has determined that is an "audit committee financial expert" within the meaning of the SEC regulations and the applicable rules of The Nasdaq Stock Market. Our board of directors will adopt a written charter for the audit committee, which will be available on our website at www.quanterix.com substantially concurrently with the completion of this offering. The information on our website is deemed not to be incorporated in this prospectus or to be part of this prospectus. The audit committee's responsibilities upon completion of this offering will include:
- •
- selecting a firm to serve as the independent registered public accounting firm to audit our financial statements;
122
- •
- ensuring the independence of the independent registered public accounting firm;
- •
- discussing the scope and results of the audit with the independent registered public accounting firm, and reviewing, with management and that
firm, our interim and year-end operating results;
- •
- establishing procedures for employees to anonymously submit concerns about questionable accounting or audit matters;
- •
- considering the effectiveness of our internal controls and, if any, our internal audit function;
- •
- reviewing material related-party transactions or those that require disclosure; and
- •
- approving or, as permitted, pre-approving all audit and non-audit services to be performed by the independent registered public accounting firm.
Compensation committee
Effective upon completion of this offering, our compensation committee will be comprised of , and , with serving as chairman of the committee. Each member of this committee is a non-employee director, as defined by Rule 16b-3 promulgated under the Exchange Act, and an outside director, as defined pursuant to Section 162(m) of the Internal Revenue Code of 1986, as amended. Our board of directors has determined that each member of the compensation committee is "independent" as defined in the rules of The Nasdaq Stock Market. The composition of our compensation committee meets the requirements for independence under the listing standards of The Nasdaq Stock Market, including the applicable transition rules. Our board of directors will adopt a written charter for the compensation committee, which will be available on our website at www.quanterix.com substantially concurrently with the completion of this offering. The information on our website is deemed not to be incorporated in this prospectus or to be part of this prospectus. The compensation committee's responsibilities upon completion of this offering will include:
- •
- reviewing and approving, or recommending that our board of directors approve, the compensation of our executive officers;
- •
- reviewing and recommending to our board of directors the compensation of our directors;
- •
- reviewing and recommending to our board of directors the terms of any compensatory agreements with our executive officers;
- •
- administering our stock and equity incentive plans;
- •
- reviewing and approving, or making recommendations to our board of directors with respect to, incentive compensation and equity plans; and
- •
- reviewing all overall compensation policies and practices.
Nominating and governance committee
Effective upon completion of this offering, our nominating and governance committee will be comprised of , and , with as the chairman of the committee. Our board of directors has determined that each member of the nominating and governance committee is "independent" as defined in the applicable rules of The Nasdaq Stock Market. Our board of directors will adopt a written charter for the nominating and governance committee, which will be available on our website at www.quanterix.com substantially concurrently with the completion of this offering. The information on our
123
website is deemed not to be incorporated in this prospectus or to be part of this prospectus. The nominating and governance committee's responsibilities upon completion of this offering will include:
- •
- identifying and recommending candidates for membership on our board of directors;
- •
- recommending directors to serve on board committees;
- •
- reviewing and recommending our corporate governance guidelines and policies;
- •
- reviewing proposed waivers of the code of conduct for directors and executive officers;
- •
- evaluating, and overseeing the process of evaluating, the performance of our board of directors and individual directors; and
- •
- assisting our board of directors on corporate governance matters.
Board leadership structure and role in risk oversight
Our board of directors does not have a policy regarding the separation of the roles of Chief Executive Officer and Chairman of the board of directors, as our board of directors believes it is in the best interest of the Company to make that determination based on the position and direction of the Company and the membership of the board of directors. Our board of directors has determined that having an employee director serve as Chairman is in the best interest of our stockholders at this time because of the efficiencies achieved in having the role of Chief Executive Officer and Chairman combined, and because the detailed knowledge of our day-to-day operations and business that the Chief Executive Officer possesses greatly enhances the decision-making processes of our board of directors as a whole. We have a strong governance structure in place, including independent directors, to ensure the powers and duties of the dual role are handled responsibly. We do not have a lead independent director.
The Executive Chairman of the board of directors and the other members of the board of directors work in concert to provide oversight of our management and affairs. Our board of directors encourages communication among its members and between management and the board of directors to facilitate productive working relationships. Working with the other members of the board of directors, our Executive Chairman also strives to ensure that there is an appropriate balance and focus among key board responsibilities such as strategic development, review of operations and risk oversight.
Compensation committee interlocks and insider participation
None of the members of our compensation committee has at any time during the prior three years been one of our officers or employees. None of our executive officers currently serves, or in the past fiscal year has served, as a member of the board of directors or compensation committee of any entity that has one or more executive officers serving on our board of directors or compensation committee. For a description of transactions between us and members of our compensation committee and affiliates of such members, please see "Certain relationships and related party transactions."
Code of business conduct and ethics
We plan to adopt a code of business conduct and ethics that applies to all of our employees, officers and directors, including those officers responsible for financial reporting, which will be effective upon completion of this offering. Upon the completion of this offering, our code of business conduct and ethics will be available on our website at www.quanterix.com. The information on our website is deemed not to be incorporated in this prospectus or to be part of this prospectus. We intend to disclose any amendments to the code, or any waivers of its requirements, on our website or in a Current Report on Form 8-K.
124
Executive and director compensation
The following table shows the total compensation paid or accrued during the fiscal year ended December 31, 2016 to our Executive Chairman, President and Chief Executive Officer and our two next most highly compensated executive officers who earned more than $100,000 during the fiscal year ended December 31, 2016 and were serving as executive officers as of such date.
| |
|
|
|
|
|
|
|||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | |
Name and principal position |
Year | Salary ($) |
Bonus ($)(1) |
Option awards ($)(2) |
All other compensation ($) |
Total ($) |
|||||||||||||
| | | | | | | | | | | | | | | | | | | | |
E. Kevin Hrusovsky |
2016 | 411,000 | 137,500 | — | — | 548,500 | |||||||||||||
Executive Chairman, President and Chief Executive Officer |
|||||||||||||||||||
Bruce Bal(3) |
2016 |
142,442 |
34,111 |
240,724 |
— |
417,277 |
|||||||||||||
Vice President of Operations, Service and Quality |
|||||||||||||||||||
Mark T. Roskey, Ph.D. |
2016 |
236,812 |
82,884 |
— |
— |
319,696 |
|||||||||||||
Senior Vice President and General Manager of Applications and Reagents |
|||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | |
(1) Amounts represent cash bonuses earned for the 12-month period from January 1, 2016 to December 31, 2016 and paid in 2017, and exclude payments made in 2016 for cash bonuses earned in 2015.
(2) These amounts represent the aggregate grant date fair value for option awards granted during our fiscal year ended December 31, 2016, computed in accordance with FASB ASC Topic 718. A discussion of the assumptions used in determining grant date fair value may be found in Note 8 to our consolidated financial statements for the year ended December 31, 2016, included elsewhere in this prospectus.
(3) Mr. Bal began his employment with us on May 8, 2016. The amount under "Salary" for Mr. Bal includes $6,000 for consulting services provided prior to his employment.
Narrative disclosure to summary compensation table
We have entered into agreements with each of our named executive officers in connection with their employment with us, the material terms of which are described below. Except as noted below, these agreements provide for "at will" employment and obligate each named executive officer to refrain from disclosing any of our proprietary information received during the course of employment.
E. Kevin Hrusovsky
We entered into an agreement with Mr. Hrusovsky with respect to his service as President and Chief Executive Officer on January 1, 2015. Under the terms of the agreement, Mr. Hrusovsky received a one-time signing bonus of $250,000 and was entitled to an initial annual base salary of $400,000. His current base salary is $429,520. Under the agreement, Mr. Hrusovsky is eligible to receive an annual bonus of up to $125,000 based on the compensation committee's assessment of his and the Company's performance against goals established by the committee. For 2016, our compensation committee awarded Mr. Hrusovsky a discretionary bonus of $137,500. In connection with his employment, in February 2015 Mr. Hrusovsky was granted 2,510,330 shares of restricted stock. The shares underlying this grant vest, subject to continued service, as follows: (1) with respect to 540,743 of the shares, 25% of the shares vested on July 1, 2015, with the remainder vesting over the next three years in equal monthly installments on the first day of each succeeding calendar month thereafter, (2) with respect to 1,764,850 of the shares, 25% of the shares
125
vested on September 1, 2015, with the remainder vesting over the next three years in equal monthly installments on the first day of each succeeding calendar month thereafter and (3) 204,737 shares vest based upon the achievement of certain milestones. Pursuant to the agreement, Mr. Hrusovsky is entitled to continuation of his then-current base salary and health insurance benefits for six months in the event we terminate his employment without Cause, as defined in the agreement, or Mr. Hrusovsky terminates his employment with us for Good Reason, as defined in the agreement, subject to Mr. Hrusovsky's execution of a release satisfactory to us following such termination. If such termination occurs within 12 months of the sale of the Company, he is entitled to continuation of his then-current base salary and health insurance benefits for 12 months. If we terminate his employment without Cause, his employment terminates due to his death or disability or he terminates his employment with us for Good Reason within three months prior to the end of a year, he is entitled to a pro rata portion of his bonus. In addition, Mr. Hrusovsky has entered into an employee non-competition, non-solicitation, confidentiality and assignment agreement pursuant to which he has agreed (1) not to engage in any competitive business for six months following his termination of employment with us (12 months if the termination is within 12 months of the sale of the Company), (2) not to solicit our employees, customers or suppliers for six months following his termination of employment with us (12 months if the termination is within 12 months of the sale of the Company) and (3) to assign any inventions conceived or developed during the course of his employment with us.
Bruce Bal
We entered into an agreement with Mr. Bal with respect to his service as Vice President of Operations, Service and Quality on April 6, 2016. Under the terms of the agreement, Mr. Bal was entitled to an initial annual base salary of $215,000. His current base salary is $222,525. Under the agreement, Mr. Bal is eligible to receive an annual bonus of up to 25% of his then-current base salary based on his performance and our business conditions in the sole discretion of our board of directors. Pursuant to the agreement, Mr. Bal was granted an option to purchase up to 325,000 shares of our common stock at an exercise price of $1.58 per share, which was the fair market value on the date of grant. This option vested as to 25% of the shares on May 9, 2017, with the remainder vesting over the subsequent three years in equal monthly installments. In addition, Mr. Bal has entered into an employee non-solicitation, confidentiality and assignment agreement pursuant to which he has agreed (1) not to solicit our employees, customers or suppliers for one year following termination of his employment and (2) to assign any inventions conceived or developed during the course of his employment with us.
Mark T. Roskey, Ph.D.
We entered into an agreement with Dr. Roskey with respect to his service as Senior Vice President and General Manager of Applications and Reagents on August 8, 2014. Under the terms of the agreement, Dr. Roskey was entitled to an initial annual base salary of $230,000. His current base salary is $246,382. Under the agreement, Dr. Roskey is eligible to receive an annual bonus of up to 35% of his then-current base salary based on his performance and our business conditions in the sole discretion of our board of directors. Pursuant to the agreement, Dr. Roskey was granted an option to purchase up to 400,000 shares of our common stock at an exercise price of $0.92 per share, which was the fair market value on the date of grant. This option vested as to 25% of the shares on September 2, 2015, with the remainder vesting over the subsequent three years in equal monthly installments. In addition, Dr. Roskey has entered into an employee non-competition, non-solicitation, confidentiality and assignment agreement pursuant to which he has agreed (1) not to engage in any business with certain competitors for one year following his voluntary termination of employment with us, (2) not to solicit our employees, customers or suppliers for one year following voluntary termination of his employment and (3) to assign any inventions conceived or developed during the course of his employment with us.
126
Outstanding equity awards at 2016 fiscal year end
The following table shows grants of stock options and grants of unvested stock awards outstanding on the last day of the fiscal year ended December 31, 2016, to each of the executive officers named in the Summary Compensation Table.
| |
|
|
|
|
|
|
|
|
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | | | | | | | |
|
Option awards(1) | Stock awards(1) | |||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | |
Name |
Number of securities underlying unexercised options (#) exercisable |
Number of securities underlying unexercised options (#) unexercisable |
Option exercise price ($) |
Option expiration date |
Number of shares or units of stock that have not vested (#) |
Market value of shares or units of stock that have not vested ($)(2) |
Equity incentive plan awards: number of unearned shares, units or other rights that have not vested (#) |
Equity incentive plan awards: market or payout value of unearned shares, units or other rights that have not vested ($)(2) |
|||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | |
E. Kevin Hrusovsky |
— | — | — | — | 95,094 | (3) | — | — | |||||||||||||||||
|
— | — | — | — | 986,186 | (4) | 127,961 | (4) | |||||||||||||||||
Bruce Bal |
— |
325,000 |
(5) |
1.58 |
6/24/2026 |
— |
— |
— |
— |
||||||||||||||||
Mark T. Roskey, Ph.D. |
233,328 |
(6) |
166,672 |
(6) |
0.92 |
9/23/2024 |
— |
— |
— |
— |
|||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | |
(1) Each of the outstanding equity awards in the table above was granted pursuant to our 2007 Stock Option and Grant Plan, as amended.
(2) There was no public market for our common stock at December 31, 2016. We have estimated the market value of the unvested stock awards assuming an initial public offering price of $ per share, the midpoint of the estimated price range set forth on the cover page of this prospectus.
(3) On December 2, 2014, Mr. Hrusovsky was granted 253,623 shares of restricted stock in connection with his service as our Executive Chairman. The shares underlying this grant vest, subject to continued service, as follows: 25% of the shares vested on July 1, 2015, with the remainder vesting over the next three years in equal monthly installments on the first day of each succeeding calendar month thereafter. Upon the sale of the Company, the vesting is accelerated such that no more than 25% of the shares shall remain unvested. If Mr. Hrusovsky is terminated without Cause (as defined in his employment agreement) or he terminates his service for Good Reason (as defined in his employment agreement) within 12 months following a sale of the Company, all of the unvested shares will become vested.
(4) On February 13, 2015, Mr. Hrusovsky was granted 2,510,330 shares of restricted stock in connection with his appointment as our President and Chief Executive Officer. The shares underlying this grant vest, subject to continued service, as follows: (i) with respect to 540,743 of the shares, 25% of the shares vested on July 1, 2015, with the remainder vesting over the next three years in equal monthly installments on the first day of each succeeding calendar month thereafter, (ii) with respect to 1,764,850 of the shares, 25% of the shares vested on September 1, 2015, with the remainder vesting over the next three years in equal monthly installments on the first day of each succeeding calendar month thereafter and (iii) 204,737 shares vest based upon the achievement of certain milestones. Upon the sale of the Company, the vesting is accelerated such that no more than 25% of the shares shall remain unvested. If Mr. Hrusovsky is terminated without Cause (as defined in his employment agreement) or he terminates his service for Good Reason (as defined in his employment agreement) within 12 months following a sale of the Company, all of the unvested shares will become vested.
(5) Represents an option to purchase shares of our common stock granted on June 24, 2016. The shares underlying this option vest, subject to continued service, as follows: 25% of the shares vested on May 9, 2017, with the remainder vesting over the next three years in equal monthly installments on the last day of each succeeding calendar month thereafter.
(6) Represents an option to purchase shares of our common stock granted on September 23, 2014. The shares underlying this option vest, subject to continued service, as follows: 25% of the shares vested on September 2, 2015, with the remainder vesting over the next three years in equal monthly installments on the last day of each succeeding calendar month thereafter. Upon the sale of the Company, the vesting is accelerated by an additional 18 months.
127
The following table shows the total compensation paid or accrued during the fiscal year ended December 31, 2016, to each of our non-employee directors. Directors who are employed by us are not compensated for their service on our board of directors.
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
Name |
Fees earned or paid in cash ($) |
Option awards ($)(1) |
All other compensation ($) |
Total ($) |
|||||||||
| | | | | | | | | | | | | | |
Douglas G. Cole, M.D. |
— | — | — | — | |||||||||
John M. Connolly |
— | — | — | — | |||||||||
Keith L. Crandell |
— | — | — | — | |||||||||
Marijn Dekkers, Ph.D. |
— | (2) | — | — | — | ||||||||
Martin D. Madaus, Ph.D. |
25,000 | (2) | — | — | 25,000 | ||||||||
Paul M. Meister |
25,000 | (2) | — | — | 25,000 | ||||||||
Dennis Sandstedt(3) |
— | — | — | — | |||||||||
David R. Walt, Ph.D. |
25,000 | (2) | — | — | 25,000 | ||||||||
| | | | | | | | | | | | | | |
(1) There were no options or other equity awards granted to directors in 2016. Except as noted below, none of our directors held options to purchase our common stock or any other stock awards as of December 31, 2016. In connection with his appointment as a director, on March 31, 2017, Dr. Dekkers was granted an option to purchase 100,000 shares of our common stock at an exercise price of $2.54 per share, which was the fair market value on the date of grant. This option vests as to 25% of the shares on March 31, 2018, with the remainder vesting over the subsequent three years in equal monthly installments.
| |
|
|||
|---|---|---|---|---|
| | | | | |
Name |
Aggregate number of shares subject to stock options |
|||
| | | | | |
Martin D. Madaus, Ph.D. |
276,959 | |||
Paul M. Meister |
75,000 | |||
| | | | | |
(2) Our practice in recent years has been to pay each non-employee director who is not affiliated with our major stockholders an annual retainer of $25,000. Dr. Dekkers joined the Board on March 31, 2017 and therefore did not earn any fee for service as a director in 2016.
(3) Mr. Sandstedt was a director in 2016, but resigned as of November 6, 2017.
Director indemnification
Pursuant to the terms of our Fourth Amended and Restated Stockholders Agreement dated June 2, 2017, for so long as any nominee of our major investors (Douglas G. Cole, M.D., John M. Connolly and Keith L. Crandell) continue to serve on our Board of Directors, we have agreed to promptly reimburse in full each non-employee director for all of his or her reasonable out-of-pocket expenses incurred in attending each meeting of our Board of Directors or its committees. Directors may be reimbursed for travel, food, lodging and other expenses directly related to their service as directors. Directors are also entitled to the protection provided by their indemnification agreements, the indemnification provisions of our Fourth Amended and Restated Stockholders Agreement dated June 2, 2017, and the indemnification provisions in our current certificate of incorporation and by-laws, as well as the restated certificate of incorporation and restated by-laws that will become effective upon the completion of this offering. In connection with this offering, we will also enter into indemnification agreements with each of our directors.
128
Equity compensation plans and other benefit plans
2017 Equity incentive plan
We plan to adopt a 2017 Equity Incentive Plan, or the 2017 Plan, which will become effective upon the closing of this offering. The 2017 Plan will expire in 2027. Under the 2017 Plan, we may grant incentive stock options, non-qualified stock options, restricted and unrestricted stock awards and other stock-based awards. Each of the share numbers that follows in this description of the 2017 Plan are fixed and are not subject to change based on our reverse stock split. There will be (1) shares of our common stock authorized for issuance under the 2017 Plan plus up to (2) shares of our common stock represented by awards granted under our 2007 Stock Option and Grant Plan, as amended, or the 2007 Plan, that are forfeited, expire or are cancelled without delivery of shares or which result in the forfeiture of shares of our common stock back to us on or after the date that the 2017 Plan becomes effective.
In addition, the 2017 Plan contains an "evergreen" provision, which allows for an annual increase in the number of shares of our common stock available for issuance under the 2017 Plan on the first day of each fiscal year during the period beginning in fiscal year 2018 and ending in fiscal year 2027. The annual increase in the number of shares shall be equal to the lowest of:
- •
- 4% of the number of shares of our common stock outstanding as of such date; and
- •
- an amount determined by our board of directors or compensation committee.
Our board of directors has authorized our compensation committee to administer the 2017 Plan. In accordance with the provisions of the plan, the compensation committee will determine the terms of options and other awards, including the following:
- •
- which employees, directors and consultants shall be granted awards;
- •
- the number of shares of our common stock subject to options and other awards;
- •
- the exercise price of each option, which generally shall not be less than fair market value on the date of grant;
- •
- the termination or cancellation provisions applicable to options;
- •
- the terms and conditions of other awards, including conditions for repurchase, termination or cancellation, issue price and repurchase price;
and
- •
- all other terms and conditions upon which each award may be granted in accordance with our plan.
No participant may receive awards for more than s hares of our common stock in any fiscal year. In addition, our board of directors or any committee to which the board of directors delegates authority may, with the consent of the affected plan participants, re-price or otherwise amend outstanding awards consistent with the terms of our plan.
Upon a merger, consolidation or sale of all or substantially all of our assets, our board of directors or any committee to which the board of directors delegates authority, or the board of directors of any corporation assuming our obligations, may, in its sole discretion, take any one or more of the following actions pursuant to our 2017 Plan, as to some or all outstanding awards, to the extent not otherwise agreed under any individual optionholder's option or employment agreement:
- •
- provide that outstanding options will be assumed or substituted for options of the successor corporation;
129
- •
- provide that the outstanding options must be exercised within a certain number of days, either to the extent the options are then exercisable,
or at our board of directors' discretion, any such options being made partially or fully exercisable;
- •
- terminate outstanding options in exchange for a cash payment of an amount equal to the difference between (a) the consideration payable
upon consummation of the corporate transaction to a holder of the number of shares into which such option would have been exercisable to the extent then exercisable, or in our board of directors'
discretion, any such options being made partially or fully exercisable, and (b) the aggregate exercise price of those options;
- •
- provide that outstanding stock grants will be substituted for shares of the successor corporation or consideration payable with respect to our
outstanding stock in connection with the corporate transaction; and
- •
- terminate outstanding stock grants in exchange for payment of an amount equal to the consideration payable upon consummation of the corporate transaction to a holder of the same number of shares comprising the stock grant, to the extent the stock grant is no longer subject to any forfeiture or repurchase rights, or at our board of directors' discretion, all forfeiture and repurchase rights being waived upon the corporation transaction.
2007 Stock option and grant plan, as amended
The 2007 Stock Option and Grant Plan, as amended, or the 2007 Plan, was adopted in June 2007 and was last amended in March 2017. As of September 30, 2017, a maximum of 13,981,013 shares of our common stock was authorized for issuance under the 2007 Plan. The 2007 Plan allows us to grant options and restricted and unrestricted stock awards to our employees, officers and directors as well as outside consultants we retain from time to time. As of October 31, 2017, under the 2007 Plan, options to purchase 7,327,673 shares of our common stock were outstanding, 1,665,491 shares of our common stock had been issued and were outstanding pursuant to the exercise of options, 3,628,532 shares of our common stock had been issued and were outstanding pursuant to restricted or unrestricted stock awards, and 1,359,317 shares of our common stock were available for future awards. We anticipate that in connection with the completion of this offering, we will terminate the 2007 Plan.
Under the 2007 Plan, in the event of our dissolution or liquidation, the sale of all or substantially all of our assets to an unrelated person or entity, or a merger, reorganization or consolidation in which our outstanding shares of common stock are converted into or exchanged for securities of the successor entity and the holders of our outstanding voting power immediately prior to such transaction do not own at least a majority of the outstanding voting power of the successor entity immediately upon completion of such transaction (taking into account only ownership interests resulting from pre-transaction interests in the Company), then outstanding options under the 2007 Plan will be terminated upon such event unless we and the other party to the transaction provide that the outstanding options will be assumed and continued by the successor entity or that new options of the successor entity or its parent will be substituted for the outstanding options under the 2007 Plan, with appropriate adjustment as to the number and kind of shares and, if appropriate, the per share exercise prices, as we and the other party may agree. If all of the outstanding options are terminated in connection with such event, we will provide option holders with an opportunity to exercise their outstanding vested options within a specific number of days, after which the options will terminate. If the holders of our common stock receive a cash payment for shares of common stock surrendered in such transaction, we may provide to each option holder in exchange for the cancellation of such holder's outstanding vested options a payment in cash in an amount equal to the amount by which (a) the product of (i) the value of the consideration per share of common stock payable
130
in the transaction and (ii) the number of outstanding vested options held by such holder exceeds (b) the aggregate exercise price of the outstanding vested options held by such holder.
We currently maintain broad-based benefits that are provided to all employees, including health insurance, life and long-term disability insurance and dental insurance.
131
Certain relationships and related party transactions
Since January 1, 2014, we have engaged in the following transactions with our directors, executive officers and holders of more than 5% of our voting securities, which we refer to as our principal stockholders, and affiliates or immediate family members of our directors, executive officers and principal stockholders. We believe that all of these transactions were on terms as favorable as could have been obtained from unrelated third parties.
Some of our directors are or were recently affiliated with our principal stockholders as indicated in the table below:
| |
|
|
|---|---|---|
| | | |
| Director |
Affiliation with principal stockholder |
|
| | | |
| Keith L. Crandell | Managing Director of ARCH Venture Partners VI, LLC, the ultimate general partner of ARCH Venture Fund VI, L.P., and Managing Director of ARCH Venture Partners VIII, LLC, the general partner of ARCH Venture Fund VIII Overage, L.P. | |
Douglas G. Cole, M.D. |
Managing Partner of Flagship Pioneering and a member of Flagship Ventures General Partner LLC, the sole general partner of Flagship Ventures Fund 2004, L.P. |
|
John M. Connolly |
Senior Advisor to Bain Capital Ventures, the ultimate general partner of Bain Capital Venture Fund 2005, L.P., BCIP Associates III, LLC and BCIP Associates III-B, LLC. |
|
Dennis Sandstedt(1) |
Senior Vice President, Corporate Business Development of bioMérieux S.A. |
|
| | | |
(1) Mr. Sandstedt was a member of our board of directors from November 14, 2012 until November 6, 2017.
See "—Agreements with stockholders" for a discussion of arrangements among our stockholders pursuant to which our directors were selected.
Series D-1 preferred stock financing
In June 2017, we issued an aggregate of 2,113,902 shares of our Series D-1 preferred stock at $4.021 per share for an aggregate purchase price of approximately $8.5 million, which we refer to as the Series D-1 preferred stock financing. Each share of our Series D-1 preferred stock will convert automatically into shares of our common stock immediately prior to the completion of this offering.
The following table summarizes the Series D-1 preferred stock purchased by our directors, executive officers and principal stockholders.
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
Name of purchaser |
Number of shares of Series D-1 preferred stock purchased |
Aggregate purchase price |
|||||
| | | | | | | | |
Marijn Dekkers, Ph.D.(1) |
248,694 | $ | 999,999 | ||||
| | | | | | | | |
(1) Marijn Dekkers, Ph.D. is a director of the Company.
132
Series D preferred stock financing
In March 2016, we issued an aggregate of 12,420,262 shares of our Series D preferred stock at $3.67 per share for an aggregate purchase price of approximately $45.6 million, which we refer to as the Series D preferred stock financing. Each share of our Series D preferred stock will convert automatically into shares of our common stock immediately prior to the completion of this offering.
The following table summarizes the Series D preferred stock purchased by our directors, executive officers and principal stockholders.
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
Name of purchaser |
Number of shares of Series D preferred stock purchased |
Aggregate purchase price |
|||||
| | | | | | | | |
Entities affiliated with ARCH Venture Funds(1) |
5,313,351 | $ | 19,499,998 | ||||
Entities affiliated with Bain Capital(2) |
404,632 | $ | 1,484,999 | ||||
Entities affiliated with Cormorant(3) |
2,724,794 | $ | 9,999,994 | ||||
David R. Walt, Ph.D.(4) |
435,967 | $ | 1,599,999 | ||||
Trinitas Innovation-Q Investment Co., Ltd. |
2,997,275 | $ | 10,999,999 | ||||
Woburn Abbey March 2009 Trust, U.D.T. March 31, 2009(5) |
85,484 | $ | 313,726 | ||||
| | | | | | | | |
(1) Consists of 1,226,158 shares issued to ARCH Venture Fund VI, L.P. and 4,087,193 shares issued to ARCH Venture Fund VIII Overage, L.P.
(2) Consists of 353,283 shares issued to Bain Capital Venture Fund 2005, L.P.; 50,336 shares issued to BCIP Associates III, LLC; and 1,013 shares issued to BCIP Associates III-B, LLC.
(3) Consists of 2,070,844 shares issued to Cormorant Private Healthcare Fund I, L.P.; 544,959 shares issued to Cormorant Global Healthcare Master Fund, L.P. and 108,991 shares issued to CRMA SPV, L.P.
(4) David R. Walt, Ph.D. is a director of the Company.
(5) Paul M. Meister is a director of the Company and is related to the Woburn Abbey March 2009 Trust, U.D.T. March 31, 2009.
Joint development agreement with bioMérieux SA
We have entered into a joint development agreement with bioMérieux SA See "Business—Key agreements—License agreement with bioMérieux SA" for a discussion of this agreement. On January 16, 2015 and May 28, 2015, we issued 1,501,546 shares of Series C preferred stock and 600,618 shares of Series C preferred stock, respectively, to bioMérieux S.A. at a purchase price of $3.3299 per share for an aggregate of $5.0 million and $2.0 million, respectively, upon the achievement of equity milestones under the joint development agreement. Each share of our Series C preferred stock will convert automatically into shares of our common stock immediately prior to the completion of this offering.
License Agreement with Tufts University
We entered into a license agreement with Tufts University. See "Business—Intellectual property—License agreement with Tufts University." One of our directors and our founding scientist, David R. Walt, Ph.D. previously served as University Professor, Professor of Biomedical Engineering, Professor of Genetics, Professor of Neuroscience, Professor of Cell and Molecular Biology, and Professor of Oral Medicine at Tufts University. Under that agreement, we are required to pay Tufts University royalties on all net sales of products and services as well as a portion of any sublicensing revenues. Tufts University pays a portion of the royalties received from us to Dr. Walt, the amount of which is controlled solely by Tufts University.
133
In connection with the Series D-1 preferred stock financing described above under "—Series D-1 preferred stock financing," we entered into various stockholder agreements with our security holders relating to voting rights, information rights and registration rights, among other things. Our Fourth Amended and Restated Stockholders Agreement dated June 2, 2017, or the Stockholders Agreement, requires the stockholders party thereto to vote to elect to our board of directors one individual designated by ARCH Venture Fund VI, L.P., currently Keith L. Crandell; one individual designed by Flagship Ventures Fund 2004, L.P., currently Douglas G. Cole, M.D.; one individual designated by Bain Capital Venture Fund 2005, L.P., BCIP Associates III, LLC, BCIP Associates III-B, LLC, RGIP, LLC and their affiliated funds, currently John M. Connolly; our Chief Executive Officer, currently E. Kevin Hrusovsky; one individual designated by a majority-in-interest of the holders of shares of common stock outstanding, currently David R. Walt, Ph.D.; one individual designated by bioMérieux S.A., currently vacant; two individuals who are industry representatives, currently Martin D. Madaus, Ph.D. and Marijn Dekkers, Ph.D.; and one individual who is a financial expert designated by the majority-in-interest of the holders of preferred stock and approved by a majority-in-interest of the holders of common stock, currently Paul M. Meister. We refer to Mr. Crandell, Dr. Cole, Mr. Connolly and any individual elected as a director to fill the vacancy that may be designated by bioMérieux S.A. as the Investor Directors. The Stockholders Agreement also requires us to maintain a compensation committee, an audit committee and such other committees as our board of directors deems necessary or convenient from time to time, and that each such committee consist of three directors, at least two of whom shall be Investor Directors. The voting and information rights under this stockholder agreement will terminate upon the completion of this offering.
Following the expiration of the lock-up period described below in "Shares eligible for future sale—Lock-up agreements," pursuant to our registration rights agreement, the holders of 46,240,115 shares of common stock, which includes 2,255,000 shares of common stock outstanding as of September 30, 2017, 43,561,745 shares of common stock issuable upon conversion of our outstanding preferred stock, 323,370 shares of common stock issuable upon the exercise of warrants outstanding as of September 30, 2017 and 100,000 shares of our common stock issuable upon the exercise of options outstanding as of September 30, 2017, are entitled to registration rights with respect to the shares of common stock held by them. These shares include all of the shares held (and shares issuable upon the exercise of warrants held) following this offering by our principal stockholders and their affiliates; our directors Marijn Dekkers, Ph.D. and David R. Walt, Ph.D.; and an affiliate of our director Paul M. Meister. See "Description of capital stock—Registration rights" for a more detailed description of these registration rights.
On March 20, 2017, we entered into a letter agreement with our director Marijn Dekkers, Ph.D. pursuant to which Dr. Dekkers agreed to serve as a member of our board of directors. Pursuant to the letter agreement, we granted Dr. Dekkers an option to purchase 100,000 shares of our common stock vesting over four years. In addition, we agreed to provide Dr. Dekkers with the opportunity to participate in any private placement financing transaction we entered into prior to completion of this offering on the same terms as other investors in such transaction. We also agreed to pay Dr. Dekkers compensation of $25,000 per year for service on our board of directors and to reimburse Dr. Dekkers for his reasonable out-of-pocket expenses in attending board and committee meetings. We also agreed to provide
134
indemnification of Dr. Dekkers pursuant to our certificate of incorporation, an indemnification agreement and our director and officer insurance.
On August 7, 2013, we entered into a letter agreement with our director Paul M. Meister pursuant to which Mr. Meister agreed to serve as a member of our board of directors. Pursuant to the letter agreement, we granted Mr. Meister an option to purchase 75,000 shares of our common stock vesting over four years. We also agreed to pay Mr. Meister compensation of $25,000 per year for service on our board of directors and to reimburse Mr. Meister for his reasonable out-of-pocket expenses in attending board and committee meetings. We also agreed to provide indemnification of Mr. Meister pursuant to our certificate of incorporation, an indemnification agreement and our director and officer insurance.
On January 1, 2014, we entered into a letter agreement with our director David Walt, Ph.D. pursuant to which we agreed to pay Dr. Walt compensation of $25,000 per year for service on our board of directors, commencing as of January 1, 2014 and for so long as he continues to serve as a non-employee member of our board of directors, and to reimburse Dr. Walt for his reasonable out-of-pocket expenses in attending board and committee meetings.
Indemnification agreements with officers and directors and directors' and officers' liability insurance
In connection with this offering, we will enter into indemnification agreements with each of our executive officers and directors. The indemnification agreements, our restated certificate of incorporation and our restated by-laws to be in effect upon completion of this offering will require us to indemnify our directors to the fullest extent not prohibited by Delaware law. Subject to certain limitations, our restated by-laws also require us to advance expenses incurred by our directors and officers.
In addition, pursuant to the terms of our Fourth Amended and Restated Stockholders Agreement dated June 2, 2017, we have also agreed to indemnify the parties to the stockholders agreement, as well as their respective affiliates, direct and indirect partners (including partners of partners and stockholders and members of partners), members, stockholders, directors, officers, employees, investment advisers and agents and each person who controls any of them from any losses (including reasonable costs and expenses) based upon, arising out of, or by reason of any claim relating to the indemnified party's status as a security holder, creditor, director or controlling person of the Company or otherwise to the extent that such claims relate to such indemnified party's involvement with the Company, except for losses arising from or based on (i) an untrue statement or omission or an alleged untrue statement or omission in a registration statement or prospectus made in reliance upon and in conformity with written information furnished to the Company by or on behalf of such indemnified party, or (ii) conduct by an indemnified party which constitutes fraud or willful misconduct.
Our 2007 Stock Option and Grant Plan, as amended, also provides that our directors will not be liability for any act, omission, interpretation, construction or determination made in good faith in connection with the plan, and the members of our board of directors and any committee administering the plan will be entitled to indemnification and reimbursement by us in respect of any claim, loss, damage or expense (including reasonable attorneys' fees) arising or resulting therefrom to the fullest extent permitted by law and/or under any directors' and officers' liability insurance coverage which may be in effect from time to time.
We also maintain a general liability insurance policy which covers certain liabilities of directors and officers of our company arising out of claims based on acts or omissions in their capacities as directors or officers. Under our Fourth Amended and Restated Stockholders Agreement dated June 2, 2017, we also agreed to
135
use our reasonable best efforts prior to this offering to increase our directors' and officers' liability insurance to at least $10 million per occurrence, including coverage of claims under the Securities Act and the Exchange Act.
Policies and procedures for related party transactions
In connection with this offering, we plan to adopt a written policy, effective upon completion of this offering, that requires all future transactions between us and any director, executive officer, holder of 5% or more of any class of our capital stock or any member of the immediate family of, or entities affiliated with, any of them, or any other related persons, as defined in Item 404 of Regulation S-K, or their affiliates, in which the amount involved is equal to or greater than $120,000, be approved in advance by our audit committee. Any request for such a transaction must first be presented to our audit committee for review, consideration and approval. In approving or rejecting any such proposal, our audit committee is to consider the relevant facts and circumstances available and deemed relevant to the audit committee, including, but not limited to, the extent of the related party's interest in the transaction, and whether the transaction is on terms no less favorable to us than terms we could have generally obtained from an unaffiliated third party under the same or similar circumstances.
136
The following table sets forth certain information with respect to the beneficial ownership of our common stock at October 31, 2017, and as adjusted to reflect the sale of our common stock in this offering, for:
- •
- each of our directors;
- •
- each of our named executive officers;
- •
- all of our current directors and executive officers as a group; and
- •
- each person, or group of affiliated persons, who beneficially owned more than 5% of our common stock.
The number of shares of our common stock beneficially owned by each entity, person, director or executive officer is determined in accordance with the rules of the SEC, and the information is not necessarily indicative of beneficial ownership for any other purpose. Under such rules, beneficial ownership includes any shares over which the individual has sole or shared voting power or investment power as well as any shares that the individual has the right to acquire within 60 days of September 30, 2017 through the exercise of any stock option, warrants or other rights. Except as otherwise indicated, and subject to applicable community property laws, the persons named in the table have sole voting and investment power with respect to all shares of common stock held by that person.
The percentage of shares beneficially owned is computed on the basis of 54,330,740 shares of our common stock outstanding as of October 31, 2017, including 676,121 shares of unvested restricted common stock, which reflects the assumed conversion of all outstanding shares of our preferred stock into an aggregate of 45,561,745 shares of our common stock. Shares of our common stock that a person has the right to acquire within 60 days of October 31, 2017, are deemed outstanding for purposes of computing the percentage ownership of the person holding such rights, but are not deemed outstanding for purposes of
137
computing the percentage ownership of any other person, except with respect to the percentage ownership of all directors and executive officers as a group.
| |
|
|
|
|
|
|
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | |
|
After this offering | After this offering |
|||||||||||||||
| | | | | | | | | | | | | | | | | | |
|
Prior to this offering | Assuming the underwriters' option is not exercised |
Assuming the underwriters' option is exercised in full |
||||||||||||||
| | | | | | | | | | | | | | | | | | |
Name of Beneficial Owner |
Shares of common stock |
Percentage of common stock |
Shares of common stock |
Percentage of common stock |
Shares of common stock |
Percentage of common stock |
|||||||||||
| | | | | | | | | | | | | | | | | | |
Principal Stockholders: |
|||||||||||||||||
ARCH Venture Partners and affiliated funds(1) |
11,865,196 | 21.8% | |||||||||||||||
Bain Capital Venture Entities(2) |
6,890,956 | 12.7% | |||||||||||||||
bioMérieux S.A.(3) |
6,606,804 | 12.2% | |||||||||||||||
Cormorant Global Healthcare Fund, LP and affiliated entities(4) |
2,724,794 | 5.0% | |||||||||||||||
Flagship Ventures Fund 2004, L.P.(5) |
6,551,845 | 12.1% | |||||||||||||||
Trinitas Innovation-Q Investment Co., Ltd.(6) |
2,997,275 | 5.5% | |||||||||||||||
Directors and Named Executive Officers: |
|||||||||||||||||
E. Kevin Hrusovsky(7) |
2,763,953 | 5.1% | |||||||||||||||
Douglas G. Cole, M.D.(8) |
0 | * | |||||||||||||||
John M. Connolly(9) |
0 | * | |||||||||||||||
Keith L. Crandell(10) |
11,865,196 | 21.8% | |||||||||||||||
Marjin Dekkers, Ph.D.(11) |
248,694 | * | |||||||||||||||
Martin Madaus, Ph.D.(12) |
1,000,000 | 1.8% | |||||||||||||||
Paul M. Meister(13) |
75,000 | * | |||||||||||||||
David Walt, Ph.D.(14) |
3,263,823 | 6.0% | |||||||||||||||
Bruce Bal(15) |
128,640 | * | |||||||||||||||
Mark T. Roskey, Ph.D.(16) |
324,991 | * | |||||||||||||||
All executive officers and directors as a group (13 persons)(17) |
20,493,731 | 36.7% | |||||||||||||||
| | | | | | | | | | | | | | | | | | |
* Indicates beneficial ownership of less than 1%.
(1) Consists of (i) 7,744,791 shares of common stock issuable upon the conversion of 1,280,000 shares of Series A-1 preferred stock, 3,360,000 shares of Series A-2 preferred stock, 1,533,214 shares of Series B preferred stock, 345,419 shares of Series C preferred stock and 1,226,158 shares of Series D preferred stock held by ARCH Venture Fund VI, L.P. ("ARCH Fund VI"), (ii) 33,212 shares of common stock issuable upon the exercise of warrants which, prior to this offering, were exercisable for 33,212 shares of Series C preferred stock held by ARCH Fund VI, and (iii) 4,087,193 shares of common stock issuable upon the conversion of 4,087,193 shares of Series D preferred stock held by ARCH Venture Fund VIII Overage, L.P. ("ARCH Fund Overage"). The sole general partner of ARCH Fund VI is ARCH Venture Partners VI, L.P. ("ARCH Partners VI"), which may be deemed to beneficially own the shares held by ARCH Fund VI. The sole general partner of ARCH Partners VI is ARCH Venture Partners VI, LLC ("ARCH VI LLC"), which may be deemed to beneficially own the shares held by ARCH Fund VI. ARCH Partners VI and ARCH VI LLC disclaim beneficial ownership of such shares, except to the extent of any pecuniary interest therein. The managing directors of ARCH VI LLC are Keith L. Crandell, Clinton Bybee and Robert Nelsen, and they may be deemed to beneficially own the shares held by ARCH Fund VI. Messrs. Crandell, Bybee and Nelsen disclaim beneficial ownership of such shares, except to the extent of any pecuniary interest therein. The sole general partner of ARCH Fund Overage is ARCH Venture Partners VIII, LLC ("ARCH VIII LLC"), which may be deemed to beneficially own the shares held by ARCH Fund Overage. ARCH VIII LLC disclaim beneficial ownership of such shares, except to the extent of any pecuniary interest therein. The managing directors of ARCH VIII LLC are Keith L. Crandell, Clinton Bybee and Robert Nelsen, and they may be deemed to beneficially own the shares held by ARCH Fund Overage. Messrs. Crandell, Bybee and Nelsen disclaim beneficial ownership of
138
such shares, except to the extent of any pecuniary interest therein. The address of ARCH Fund VI and ARCH Fund Overage is 8755 West Higgins Road, Suite 1025, Chicago, Illinois 60631.
(2) Consists of (i) 5,987,764 shares of common stock issuable upon the conversion of 1,106,387 shares of Series A-1 preferred stock, 2,904,268 shares of Series A-2 preferred stock, 1,325,258 shares of Series B preferred stock, 298,568 shares of Series C preferred stock and 353,283 shares of Series D preferred stock held by Bain Capital Venture Fund 2005, L.P. ("Fund 2005"), (ii) 28,707 shares of common stock issuable upon the exercise of warrants which, prior to this offering, were exercisable for 28,707 shares of Series C preferred stock held by Fund 2005, (iii) 853,140 shares of common stock issuable upon the conversion of 157,639 shares of Series A-1 preferred stock, 413,802 shares of Series A-2 preferred stock, 188,823 shares of Series B preferred stock, 42,540 shares of Series C preferred stock and 50,336 shares of Series D preferred stock held by BCIP Associates III, LLC, ("BCIP III") (iv) 4,090 shares of common stock issuable upon the exercise of warrants which, prior to this offering, were exercisable for 4,090 shares of Series C preferred stock held by BCIP III, (v) 17,173 shares of common stock issuable upon the conversion of 3,174 shares of Series A-1 preferred stock, 8,331 shares of Series A-2 preferred stock, 3,799 shares of Series B preferred stock, 856 shares of Series C preferred stock and 1,013 shares of Series D preferred stock held by BCIP Associates III-B, LLC, ("BCIP III-B," collectively with Fund 2005 and BCIP III, the "Bain Capital Entities") and (vi) 82 shares of common stock issuable upon the exercise of warrants which, prior to this offering, were exercisable for 82 shares of Series C preferred stock held by BCIP III-B. Bain Capital Venture Investors, LLC ("BCVI") is the general partner of Bain Capital Venture Partners 2005, L.P., which is the general partner of Fund 2005. Boylston Coinvestors, LLC is the managing partner of BCIP Associates III and BCIP Associates III-B. BCIP Associates III is the manager of BCIP III. BCIP Associates III-B is the manager of BCIP III-B. The governance, investment strategy and decision-making process with respect to the investments held by the Bain Capital Entities is directed by the Executive Committee of BCVI, which consists of Michael A. Krupka and Ajay Agarwal. The address of the Bain Capital Entities is c/o Bain Capital Venture Investors, LLC, 200 Clarendon Street, Boston, Massachusetts 02116.
(3) Consists of 6,606,804 shares of common stock issuable upon the conversion of 6,606,804 shares of Series C preferred stock held by bioMérieux, S.A. ("bioMérieux"). The board of directors of bioMérieux consists of Jean-Luc Belingard, Alexandre Mérieux, Alain Mérieux, Marie-Hélène Habert, Michele Palladino, Philippe Gillet, Agnès Lemarchand, Philippe Archinard, and Harold Boël, who have shared investment and voting control with respect to the shares held by bioMérieux and may exercise such control only with the support of the majority of the members of the board of directors of bioMérieux. No individual member of the board of directors of bioMérieux is deemed to hold any beneficial ownership or reportable pecuniary interest in the shares held by bioMérieux. Dennis Sandstedt, one of our former directors, is Senior Vice President, Corporate Business Development at bioMérieux but has no voting or investment power over the securities held by bioMérieux. The address for bioMérieux is Chemin de l'Orme, 69280 Marcy-l'Etoile, France.
(4) Consists of (i) 544,959 shares of common stock issuable upon the conversion of 544,959 shares of Series D preferred stock held by Cormorant Global Healthcare Master Fund, L.P. ("CGHMF"); (ii) 2,070,844 shares of common stock issuable upon the conversion of 2,070,844 shares of Series D preferred stock held by Cormorant Private Healthcare Fund I, L.P. ("CPHFI") and (iii) 108,991 shares of common stock issuable upon the conversion of 108,991 shares of Series D preferred stock held by CRMA SPV, L.P. ("CRMA"). Cormorant Global Healthcare GP, LLC ("CGH") is the general partner of CGHMF. Cormorant Private Healthcare GP, LLC ("CPH") is the general partner of CPHFI. Cormorant Asset Management, LLC ("CAM") is the investment manager of CGHMF, CGHMF and CRMA. Bihua Chen is the sole managing member of the CAM and may be deemed to have sole voting and investment power of the securities held by CGHMF, CGHMF and CRMA. Bihua Chen disclaims beneficial ownership of such securities except to the extent of her pecuniary interest therein. The address of CGHMF, CGHMF and CRMA is c/o Cormorant Asset Management, LLC, 200 Clarendon Street, 52nd Floor, Boston, Massachusetts 02116.
(5) Consists of (i) 6,518,633 shares of common stock issuable upon the conversion of 1,280,000 shares of Series A-1 preferred stock, 3,360,000 shares of Series A-2 preferred stock, 1,533,214 shares of Series B preferred stock and 345,419 shares of Series C preferred stock held by Flagship Ventures Fund 2004, L.P. and (ii) 33,212 shares of common stock issuable upon the exercise of warrants which, prior to this offering, were exercisable for 33,212 shares of Series C preferred stock held by Flagship Ventures Fund 2004, L.P. Flagship Ventures General Partner LLC is the General Partner of Flagship Ventures Fund 2004, L.P. Flagship Ventures General Partner LLC is referred to as the "GP." Douglas G. Cole M.D., a member of our board of directors, is a managing partner of Flagship Pioneering and a member of the GP, the sole general partner of Flagship Ventures Fund 2004, L.P. Dr. Cole does not have either voting or investment control over the Fund's shares and he disclaims beneficial ownership of the Fund's shares, except to the extent of his pecuniary interest therein. Dr. Cole does not own shares in his individual capacity. Noubar B. Afeyan, Ph.D. and Edwin M. Kania, Jr. are the Managers of the GP. As a result, the GP, Dr. Afeyan and Mr. Kania may be deemed to possess voting and investment control over all shares held by Flagship Ventures Fund 2004, L.P. Neither the GP, Dr. Afeyan, nor Mr. Kania own directly any of the Company's securities. The address of the GP, Dr. Afeyan, Mr. Kania, and Flagship 2004 is 55 Cambridge Parkway, Suite 800E, Cambridge, Massachusetts 02142.
(6) Consists of 2,997,275 shares of common stock issuable upon the conversion of 2,997,275 shares of Series D preferred stock held by Trinitas Innovation-Q Investment Co., Ltd. ("Trinitas Innovation"). Trinitas Innovation is wholly owned by Shanghai Trinitas Capital Centre (Limited Partnership) ("Shanghai Trinitas"). Lhasa Trinitas Investment Management Co., Ltd. ("Lhasa Trinitas") is the general partner of Yantai Trinitas Equity Investment Management Centre (Limited Partnership) ("Yantai Trinitas"), which is the general partner of Shanghai Trinitas. Lhasa Economic and Technological Development Zone Yixing Financial Holding Investment Co., Ltd. ("Lhasa Economic") and Beijing Jundaocheng Investment Consulting Co., Ltd. ("Beijing Jundaocheng") are the owners of Lhasa Trinitas and share voting and investment power over the securities held by Trinitas Innovation. Bing Han is Director of Trinitas Innovation and Executive Director of Lhasa Economic, of which he owns 100% of the shares. Cheng Zhou is Executive Director of Beijing Jundaocheng, of which he owns 100% of the shares. The address of Trinitas Innovation is 401, 4/F Building 2, No. 39, Dongzhimenwai Street, Dongcheng District, Beijing, China.
(7) Consists of 2,763,953 shares of common stock held by Mr. Hrusovsky.
(8) Dr. Cole is a managing partner of Flagship Pioneering but has no voting or investment power with respect to the securities described in footnote 5.
(9) Mr. Connolly is a senior advisor for Bain Capital Ventures but has no voting or investment power with respect to the securities described in footnote 2.
139
(10) Consists of securities held by ARCH Fund VI and ARCH Fund Overage as set forth in footnote 1. Mr. Crandell disclaims beneficial ownership of such shares except to the extent of his pecuniary interest therein, if any.
(11) Consists of 248,694 shares of common stock issuable upon the conversion of 248,694 shares of Series D-1 preferred stock held by Dr. Dekkers.
(12) Consists of 276,959 shares of common stock issuable upon the exercise of stock options exercisable within 60 days of October 31, 2017 held by Mr. Madaus and 723,041 shares of common stock held by a trust for which Mr. Madaus is a trust advisor who shares voting and investment power over the shares held by the trust.
(13) Consists of 75,000 shares of common stock issuable upon the exercise of stock options exercisable within 60 days of October 31, 2017 held by Mr. Meister. Does not include 385,793 shares of common stock issuable upon the conversion of 300,309 shares of Series C preferred stock and 85,484 shares of Series D preferred stock held by Woburn Abbey March 2009 Trust, U.D.T. March 31, 2009 as Mr. Meister does not have voting or investment power over the shares held by the trust.
(14) Consists of (i) 1,275,000 shares held by Dr. Walt, (ii) 1,980,729 shares of common stock issuable upon the conversion of 105,931 shares of Series A-1 preferred stock, 278,068 shares of Series A-2 preferred stock, 1,076,642 shares of Series B preferred stock, 84,121 shares of Series C preferred stock and 435,967 shares of Series D preferred stock held by Dr. Walt, and (iii) 8,094 shares of common stock issuable upon the exercise of warrants which, prior to this offering, were exercisable for 8,094 shares of Series C preferred stock held by Dr. Walt. Does not include 645,000 shares of common stock held by The David R. Walt 2008 Irrevocable Family Trust (the "Walt Trust"), of which his spouse is trustee and has sole voting and investment power over the shares held by the Walt Trust.
(15) Consists of 128,640 shares of common stock issuable upon the exercise of stock options exercisable within 60 days of October 31, 2017 held by Mr. Bal.
(16) Consists of 324,991 shares of common stock issuable upon the exercise of stock options exercisable within 60 days of October 31, 2017 held by Dr. Roskey.
(17) See footnotes 7 through 16. Also includes 461,717 shares of common stock issuable upon the exercise of stock options exercisable within 60 days of October 31, 2017 held by Ernest Orticerio, our Senior Vice President, Finance and Corporate Development, 100,000 shares of common stock held by David C. Duffy, Ph.D., our Senior Vice President, Research & Development and Chief Technology Officer, and 261,717 shares of common stock issuable upon the exercise of stock options exercisable within 60 days of October 31, 2017 held by Dr. Duffy. None of the stock options held by Joseph Driscoll, our Chief Financial Officer, are exercisable within 60 days of October 31, 2017.
140
Upon the completion of this offering, our authorized capital stock will consist of shares of common stock, par value $0.001 per share, and 5,000,000 shares of preferred stock, par value $0.001 per share, all of which will be undesignated, and there will be shares of common stock outstanding and no shares of preferred stock outstanding. As of September 30, 2017, we had approximately 70 record holders of our capital stock. All of our outstanding shares of preferred stock will automatically convert into shares of our common stock upon the completion of this offering.
The following description of our capital stock and provisions of our restated certificate of incorporation and restated by-laws are summaries of material terms and provisions and are qualified by reference to our restated certificate of incorporation and restated by-laws, copies of which have been filed with the SEC as exhibits to the registration statement of which this prospectus is a part. The descriptions of our common stock and preferred stock reflect the content of the restated certificate of incorporation and restated by-laws that will become effective immediately prior to the completion of this offering.
Upon the completion of this offering, we will be authorized to issue one class of common stock. Holders of our common stock are entitled to one vote for each share of common stock held of record for the election of directors and on all matters submitted to a vote of stockholders. Holders of our common stock are entitled to receive dividends ratably, if any, as may be declared by our board of directors out of legally available funds, subject to any preferential dividend rights of any preferred stock then outstanding. Upon our dissolution, liquidation or winding up, holders of our common stock are entitled to share ratably in our net assets legally available after the payment of all our debts and other liabilities, subject to the preferential rights of any preferred stock then outstanding. Holders of our common stock have no preemptive, subscription, redemption or conversion rights. The rights, preferences and privileges of holders of common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of preferred stock that we may designate and issue in the future. Except as described under "—Anti-takeover effects of Delaware law, our restated certificate of incorporation and our restated by-laws" below, a majority vote of the holders of common stock is generally required to take action under our restated certificate of incorporation and restated by-laws.
Upon the completion of this offering, our board of directors will be authorized, without action by the stockholders, to designate and issue up to an aggregate of 5,000,000 shares of preferred stock in one or more series. Our board of directors can designate the rights, preferences and privileges of the shares of each series and any of its qualifications, limitations or restrictions. Our board of directors may authorize the issuance of preferred stock with voting or conversion rights that could adversely affect the voting power or other rights of the holders of common stock. The issuance of preferred stock, while providing flexibility in connection with possible future financings and acquisitions and other corporate purposes could, under certain circumstances, have the effect of restricting dividends on our common stock, diluting the voting power of our common stock, impairing the liquidation rights of our common stock, or delaying, deferring or preventing a change in control of our company, which might harm the market price of our
141
common stock. See also "—Anti-takeover effects of Delaware law, our restated certificate of incorporation and our restated by-laws—Blank check preferred stock" below.
Our board of directors will make any determination to issue such shares based on its judgment as to our company's best interests and the best interests of our stockholders. Upon the completion of this offering, we will have no shares of preferred stock outstanding and we have no current plans to issue any shares of preferred stock following completion of this offering.
As of October 31, 2017, we had warrants outstanding for the number of shares of our preferred stock at the exercise prices and expiration dates set forth below. These warrants entitle the holder to purchase shares of our preferred stock at the specified exercise price at any time prior to the expiration date. Following the conversion of all outstanding shares of preferred stock into common stock and the completion of this offering, these warrants will become exercisable for the number of shares of common stock and the specified prices set forth below.
| |
|
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | |
| Warrants |
Number of shares of preferred stock for which the warrants are exercisable |
Exercise price per share of preferred stock prior to the completion of this offering |
Number of shares of common stock for which the warrants will be exercise following the completion of this offering |
Exercise price per share of common stock following the completion of this offering |
Expiration date |
|||||||||
| | | | | | | | | | | | | | | |
Warrants to purchase Series A-2 preferred stock(1)(2) |
64,441 | $ | 1.0416667 | 64,441 | $ | 1.0416667 | (3) | |||||||
Warrants to purchase Series C preferred stock(1) |
284,542 | $ | 3.3299 | 284,542 | $ | 3.3299 | (4) | |||||||
Warrants to purchase Series D preferred stock(1)(2) |
38,828 | $ | 3.67 | 38,828 | $ | 3.67 | March 31, 2027 | |||||||
| | | | | | | | | | | | | | | |
(1) Each of these warrants has net exercise provisions under which the holder may, in lieu of payment of the exercise price in cash, surrender the warrant and receive a net amount of shares of our common based on the fair market value of the underlying shares of our common stock at the time of exercise of the warrant, after deduction of the aggregate exercise price.
(2) Each of these warrants provide that immediately before its expiration or termination, if the fair market value of one share of our common stock is greater than the exercise price, the warrant will be automatically exercised pursuant to its net exercise provision.
(3) Warrants to purchase 38,400 shares of Series A-2 preferred stock expire on January 8, 2019 and warrants to purchase 26,041 shares of Series A-2 preferred stock expire on August 7, 2019.
(4) Warrants to purchase 57,810 shares of Series C preferred stock expire on the later of January 29, 2026 or five years from the effective date of the registration statement of which this prospectus is a part, warrants to purchase 115,618 shares of Series C preferred stock expire on the later of April 14, 2024 or five years from the effective date of the registration statement of which this prospectus is a part, and warrants to purchase 111,114 shares of Series C preferred stock expire on November 14, 2017.
142
We entered into a Fourth Amended and Restated Registration Rights Agreement, dated as of June 2, 2017, or the Registration Rights Agreement, with certain holders of our capital stock. These shares will represent approximately % of our outstanding common stock after this offering, or % if the underwriters exercise their option to purchase additional shares in full. These shares also may be sold under Rule 144 under the Securities Act, depending on their holding period and subject to restrictions in the case of shares held by persons deemed to be our affiliates.
Under the Registration Rights Agreement, holders of registrable shares can demand that we file a registration statement or request that their shares be included on a registration statement that we are otherwise filing, in either case, registering the resale of their shares of common stock. These registration rights are subject to conditions and limitations, including the right, in certain circumstances, of the underwriters of an offering to limit the number of shares included in such registration and our right, in certain circumstances, not to effect a registration upon demand of the holders of registrable shares within 90 days following the effective date of any registration statement that we file covering a firm commitment underwritten public offering in which the holders of registrable shares were entitled to join and in which we effectively registered all registrable shares that were requested to be registered.
Demand registration rights
Following the date that is 180 days after the date of this offering, the holders of a majority of the registrable shares may require us to file a registration statement under the Securities Act at our expense, subject to certain exceptions, with respect to the resale of their registrable shares having an aggregate offering price (net of underwriting discounts and commissions, if any) of at least $5 million, and we are required to use our reasonable best efforts to effect the registration. The holders of a majority of the registrable shares may require us to effect up to two such demand registrations for the holders of registrable shares as a group. In the event we are required to effect such a demand registration, we may not effect any other registration of securities for sale for our own account (other than a registration effected solely to implement an employee benefit plan or in certain business combination transactions) within 120 days following the effective date of the demand registration.
After the completion of this offering, we are required to use our reasonable best efforts to qualify and remain qualified to register securities pursuant to a registration statement on Form S-3 under the Securities Act. Any holder or holders of registrable shares anticipated to have an aggregate sale price (net of underwriting discounts and commissions, if any) in excess of $1 million will have the right to require us to file, at our expense, an unlimited number of registration statements on Form S-3 for the registrable shares held by such requesting holder or holders, and we are required to use our reasonable best efforts to effect such registrations.
Piggyback registration rights
If we propose to register any of our securities under the Securities Act for sale to the public (except with respect to registration statements on Form S-4, Form S-8 or another form not available for registering the registrable shares for sale to the public), the holders of registrable shares are entitled to notice of such registration and to request that we include registrable shares for resale on such registration statement, subject to the right of any underwriter to limit the number of shares included in such registration.
We will pay all registration expenses, other than underwriting fees, commissions or discounts, related to any demand or piggyback registration, including up to $50,000 of fees and disbursements of counsel for
143
the holders of registrable shares. The Registration Rights Agreement contains customary cross-indemnification provisions, pursuant to which we are obligated to indemnify the selling stockholders in the event of misstatements or omissions in the registration statement attributable to us and they are obligated to indemnify us for misstatements or omissions attributable to them.
Anti-takeover effects of Delaware law, our restated certificate of incorporation and our restated by-laws
Our restated certificate of incorporation and restated by-laws that will take effect in connection with the closing of this offering include a number of provisions that may have the effect of encouraging persons considering unsolicited tender offers or other unilateral takeover proposals to negotiate with our board of directors rather than pursue non-negotiated takeover attempts. These provisions include the items described below.
Board composition and filling vacancies
In accordance with our restated certificate of incorporation, our board is divided into three classes serving three-year terms, with one class being elected each year. Our restated certificate of incorporation also provides that directors may be removed only for cause and then only by the affirmative vote of the holders of 75% or more of the shares then entitled to vote at an election of directors. Furthermore, any vacancy on our board of directors, however occurring, including a vacancy resulting from an increase in the size of our board, may only be filled by the affirmative vote of a majority of our directors then in office, even if less than a quorum.
No written consent of stockholders
Our restated certificate of incorporation provides that all stockholder actions are required to be taken by a vote of the stockholders at an annual or special meeting, and that stockholders may not take any action by written consent in lieu of a meeting.
Meetings of stockholders
Our restated by-laws provide that only a majority of the members of our board of directors then in office may call special meetings of stockholders and only those matters set forth in the notice of the special meeting may be considered or acted upon at a special meeting of stockholders. Our restated by-laws limit the business that may be conducted at an annual meeting of stockholders to those matters properly brought before the meeting.
Advance notice requirements
Our restated by-laws establish advance notice procedures with regard to stockholder proposals relating to the nomination of candidates for election as directors or new business to be brought before meetings of our stockholders. These procedures provide that notice of stockholder proposals must be timely given in writing to our corporate secretary prior to the meeting at which the action is to be taken. Generally, to be timely, notice must be received at our principal executive offices not less than 90 days or more than 120 days prior to the first anniversary date of the annual meeting for the preceding year. The notice must contain certain information specified in the restated by-laws. These provisions may have the effect of precluding the conduct of certain business at a meeting if the proper procedures are not followed. These provisions may also discourage or deter a potential acquirer from conducting a solicitation of proxies to elect the acquirer's own slate of directors or otherwise attempting to obtain control of our company.
144
Amendment to by-laws and certificate of incorporation
As required by the Delaware General Corporation Law, any amendment of our restated certificate of incorporation must first be approved by a majority of our board of directors and, if required by law or our restated certificate of incorporation, thereafter be approved by a majority of the outstanding shares entitled to vote on the amendment, and a majority of the outstanding shares of each class entitled to vote thereon as a class, except that the amendment of the provisions relating to stockholder action, directors, limitation of liability, exclusive jurisdiction of Delaware courts and the amendment of our restated by-laws and restated certificate of incorporation must be approved by not less than 75% of the outstanding shares entitled to vote on the amendment, and not less than 75% of the outstanding shares of each class entitled to vote thereon as a class. Our restated by-laws may be amended by the affirmative vote of a majority of the directors then in office, subject to any limitations set forth in the restated by-laws; and may also be amended by the affirmative vote of at least 75% of the outstanding shares entitled to vote on the amendment, or, if the board of directors recommends that the stockholders approve the amendment, by the affirmative vote of the majority of the outstanding shares entitled to vote on the amendment, in each case voting together as a single class.
Blank check preferred stock
Our restated certificate of incorporation provides for 5,000,000 authorized shares of preferred stock. The existence of authorized but unissued shares of preferred stock may enable our board of directors to render more difficult or to discourage an attempt to obtain control of us by means of a merger, tender offer, proxy contest or otherwise. For example, if in the due exercise of its fiduciary obligations, our board of directors were to determine that a takeover proposal is not in the best interests of us or our stockholders, our board of directors could cause shares of preferred stock to be issued without stockholder approval in one or more private offerings or other transactions that might dilute the voting or other rights of the proposed acquirer or insurgent stockholder or stockholder group. In this regard, our restated certificate of incorporation grants our board of directors broad power to establish the rights and preferences of authorized and unissued shares of preferred stock. The issuance of shares of preferred stock could decrease the amount of earnings and assets available for distribution to holders of shares of common stock. The issuance may also adversely affect the rights and powers, including voting rights, of the holders of shares of common stock and may have the effect of delaying, deterring or preventing a change in control of us.
Section 203 of the Delaware General Corporation Law
Upon completion of this offering, we will be subject to the provisions of Section 203 of the Delaware General Corporation Law. In general, Section 203 prohibits a publicly-held Delaware corporation from engaging in a "business combination" with an "interested stockholder" for a three-year period following the time that this stockholder becomes an interested stockholder, unless the business combination is approved in a prescribed manner. A "business combination" includes, among other things, a merger, asset or stock sale or other transaction resulting in a financial benefit to the interested stockholder. An "interested stockholder" is a person who, together with affiliates and associates, owns, or did own within three years prior to the determination of interested stockholder status, 15% or more of the corporation's voting stock.
Under Section 203, a business combination between a corporation and an interested stockholder is prohibited unless it satisfies one of the following conditions:
- •
- before the stockholder became interested, the board of directors approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder;
145
- •
- upon consummation of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at
least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding, shares owned by persons who are
directors and also officers, and employee stock plans, in some instances; or
- •
- at or after the time the stockholder became interested, the business combination was approved by the board of directors of the corporation and authorized at an annual or special meeting of the stockholders by the affirmative vote of at least two-thirds of the outstanding voting stock that is not owned by the interested stockholder.
A Delaware corporation may "opt out" of these provisions with an express provision in its original certificate of incorporation or an express provision in its restated certificate of incorporation or by-laws resulting from a stockholders' amendment approved by at least a majority of the outstanding voting shares. We have not opted out of these provisions. As a result, mergers or other takeover or change in control attempts of us may be discouraged or prevented.
Exclusive jurisdiction of certain actions
Our restated certificate of incorporation requires, to the fullest extent permitted by law, that derivative actions brought in our name, actions against our directors, officers and employees for breach of fiduciary duty and other similar actions may be brought only in the Court of Chancery in the State of Delaware, unless we otherwise consent. Although we believe this provision benefits us by providing increased consistency in the application of Delaware law in the types of lawsuits to which it applies, the provision may have the effect of discouraging lawsuits against our directors and officers.
We have applied to list our common stock on The Nasdaq Global Market under the trading symbol "QTRX."
The transfer agent and registrar for our common stock will be Computershare Trust Company, N.A. The transfer agent and registrar's address is 250 Royall Street, Canton, MA 02021.
146
Shares eligible for future sale
Prior to this offering, there has been no public market for our common stock. Future sales of our common stock, including shares issued upon the exercise of outstanding options or warrants, in the public market after this offering, or the perception that those sales may occur, could cause the prevailing market price for our common stock to fall or impair our ability to raise equity capital in the future. As described below, only a limited number of shares of our common stock will be available for sale in the public market for a period of several months after completion of this offering due to contractual and legal restrictions on resale described below. Future sales of our common stock in the public market either before (to the extent permitted) or after restrictions lapse, or the perception that those sales may occur, could adversely affect the prevailing market price of our common stock at such time and our ability to raise equity capital at a time and price we deem appropriate.
Upon the closing of this offering, based on the number of shares of our common stock outstanding as of October 31, 2017, and assuming (1) the conversion of our outstanding preferred stock into common stock, (2) no exercise of the underwriters' option to purchase additional shares of common stock and (3) no exercise of outstanding options or warrants, we will have outstanding an aggregate of approximately shares of common stock. Of these shares, all of the shares of common stock to be sold in this offering, and any shares sold upon exercise of the underwriters' option to purchase additional shares will be freely tradable in the public market without restriction or further registration under the Securities Act, unless the shares are held by any of our "affiliates" as such term is defined in Rule 144 of the Securities Act. All remaining shares of common stock held by existing stockholders immediately prior to the completion of this offering will be "restricted securities" as such term is defined in Rule 144. These restricted securities were issued and sold by us, or will be issued and sold by us, in private transactions and are eligible for public sale only if registered under the Securities Act or if they qualify for an exemption from registration under the Securities Act, including the exemptions provided by Rule 144 or Rule 701, which rules are summarized below.
As a result of the lock-up agreements referred to below and the provisions of Rule 144 and Rule 701 under the Securities Act, the shares of our common stock (excluding the shares sold in this offering) that will be available for sale in the public market are as follows:
| | | |
| Number of shares and % of total outstanding |
Date available for sale into public market |
|
|---|---|---|
| | | |
| shares, or % | On the date of this prospectus | |
shares, or % |
90 days after the date of this prospectus |
|
shares, or % |
180 days after the date of this prospectus, due to lock-up agreements between the holders of these shares and the underwriters. However, the representatives of the underwriters, acting together, can waive the provisions of these lock-up agreements and allow these stockholders to sell their shares at any time. |
|
| | | |
Additionally, of the shares of common stock issuable upon exercise of options outstanding as of October 31, 2017, approximately shares will be vested and eligible for sale 180 days after the
147
date of this prospectus, and shares of common stock issuable upon exercise of warrants outstanding as of October 31, 2017 may become eligible for sale beginning 180 days after the date of this prospectus.
In connection with this offering, we, our directors, our executive officers and holders of substantially all of our shares of common stock, warrants and stock options outstanding as of October 31, 2017 have agreed, subject to certain exceptions, with the underwriters not to dispose of or hedge any shares of our common stock or securities convertible into or exchangeable for shares of common stock during the period from the date of the lock-up agreement continuing through the date 180 days after the date of this prospectus, except with the prior written consent of (i) in the case of the Company, J.P. Morgan Securities LLC and Leerink Partners LLC, as the representatives of the underwriters, and (ii) in the case of our directors, our executive officers and holders of substantially all of our shares of common stock, J.P. Morgan Securities LLC, Leerink Partners LLC and Evercore Group L.L.C., and certain other exceptions. The representatives of the underwriters have advised us that they have no current intent or arrangement to release any of the shares subject to the lock-up agreements prior to the expiration of the lock-up period.
Following the lock-up periods set forth in the agreements described above, and assuming that the representatives of the underwriters do not release any parties from these agreements, all of the shares of our common stock that are restricted securities or are held by our affiliates as of the date of this prospectus will be eligible for sale in the public market in compliance with Rule 144 under the Securities Act.
In addition, pursuant to our Fourth Amended and Restated Stockholders Agreement and our Fourth Amended and Restated Registration Rights Agreement, the stockholders that are parties thereto have agreed, that if requested by us and our underwriters in connection with this offering, that they will not, directly or indirectly, offer, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant for the sale of or otherwise dispose of or transfer any shares of our stock during the same 180-day restricted period referred to above. We also agreed to obtain lock-up agreements from each holder of our stock or options in connection with this offering.
In general, under Rule 144, as currently in effect, once we have been subject to the public company reporting requirements of the Exchange Act for at least 90 days, a person (or persons whose shares are required to be aggregated) who is not deemed to have been one of our "affiliates" for purposes of Rule 144 at any time during the three months preceding a sale, and who has beneficially owned restricted securities within the meaning of Rule 144 for at least six months, including the holding period of any prior owner other than one of our "affiliates," is entitled to sell those shares in the public market (subject to the lock-up agreement referred to above, if applicable) without complying with the manner of sale, volume limitations or notice provisions of Rule 144, but subject to compliance with the public information requirements of Rule 144. If such a person has beneficially owned the sales proposed to be sold for at least one year, including the holding period of any prior owner other than "affiliates," then such person is entitled to sell such shares in the public market without complying with any of the requirements of Rule 144 (subject to the lock-up agreement referred to above, if applicable). In general, under Rule 144, as currently in effect, once we have been subject to the public company reporting requirements of the Exchange Act for at least 90 days, our "affiliates," as defined in Rule 144, who have beneficially owned the
148
shares proposed to be sold for at least six months are entitled to sell in the public market, upon expiration of any applicable lock-up agreements and within any three-month period, a number of those shares of our common stock that does not exceed the greater of:
- •
- 1% of the number of common shares then outstanding, which will equal approximately shares of common
stock immediately after
this offering (calculated on the basis of the number of shares of our common stock outstanding as of October 31, 2017, the assumptions described above and assuming no exercise of the
underwriters' option to purchase additional shares and no exercise of outstanding options or warrants); or
- •
- the average weekly trading volume of our common stock on The Nasdaq Global Market during the four calendar weeks preceding the filing of a notice on Form 144 with respect to such sale.
Such sales under Rule 144 by our "affiliates" or persons selling shares on behalf of our "affiliates" are also subject to certain manner of sale provisions, notice requirements and to the availability of current public information about us. Notwithstanding the availability of Rule 144, the holders of substantially all of our restricted securities have entered into lock-up agreements as referenced above and their restricted securities will become eligible for sale (subject to the above limitations under Rule 144) upon the expiration of the restrictions set forth in those agreements.
In general, under Rule 701 as currently in effect, any of our employees, directors, officers, consultants or advisors who acquired common stock from us in connection with a written compensatory stock or option plan or other written agreement in compliance with Rule 701 under the Securities Act before the effective date of the registration statement of which this prospectus is a part (to the extent such common stock is not subject to a lock-up agreement) is entitled to rely on Rule 701 to resell such shares beginning 90 days after we become subject to the public company reporting requirements of the Exchange Act in reliance on Rule 144, but without compliance with the holding period requirements contained in Rule 144. Accordingly, subject to any applicable lock-up agreements, beginning 90 days after we become subject to the public company reporting requirements of the Exchange Act, under Rule 701 persons who are not our "affiliates," as defined in Rule 144, may resell those shares without complying with the minimum holding period or public information requirements of Rule 144, and persons who are our "affiliates" may resell those shares without compliance with Rule 144's minimum holding period requirements (subject to the terms of the lock-up agreement referred to below, if applicable).
We intend to file with the SEC a registration statement under the Securities Act covering the shares of common stock that we may issue upon exercise of outstanding options under the 2007 Plan or that are reserved for issuance under the 2017 Plan. Such registration statement is expected to be filed and become effective as soon as practicable after the completion of this offering. Accordingly, shares registered under such registration statement will be available for sale in the open market following its effective date, subject to Rule 144 volume limitations and the lock-up agreements described above, if applicable.
149
Material U.S. federal income and estate tax consequences to non-U.S. holders
The following is a summary of the material U.S. federal income tax consequences of the ownership and disposition of our common stock to Non-U.S. Holders (defined below), but does not purport to be a complete analysis of all the potential tax considerations relating thereto. This summary is based upon the provisions of the Internal Revenue Code of 1986, as amended, or the Code, Treasury regulations promulgated thereunder, administrative rulings and judicial decisions, all as of the date of this prospectus. These authorities may be changed or subject to differing interpretations, possibly with retroactive effect, so as to result in U.S. federal income tax consequences different from those set forth below. We have not sought and will not seek any ruling from the Internal Revenue Service, or the IRS, with respect to the statements made and the conclusions reached in the following summary, and there can be no assurance that the IRS or a court will agree with such statements and conclusions.
This summary also does not address the tax considerations arising under the laws of any U.S. state or local or any non-U.S. jurisdiction, the 3.8% Medicare tax on net investment income or any alternative minimum tax consequences. In addition, this discussion does not address tax considerations applicable to a Non-U.S. Holder's particular circumstances or to a Non-U.S. Holder that may be subject to special tax rules, including, without limitation:
- •
- banks, insurance companies or other financial institutions;
- •
- tax-exempt or government organizations;
- •
- brokers of or dealers in securities or currencies;
- •
- traders in securities that elect to use a mark-to-market method of accounting for their securities holdings;
- •
- persons that own, or are deemed to own, more than five percent of our capital stock;
- •
- certain U.S. expatriates, citizens or former long-term residents of the United States;
- •
- persons who hold our common stock as a position in a hedging transaction, "straddle," "conversion transaction," synthetic security, other
integrated investment, or other risk reduction transaction;
- •
- persons who do not hold our common stock as a capital asset within the meaning of Section 1221 of the Code (generally, for investment
purposes);
- •
- persons deemed to sell our common stock under the constructive sale provisions of the Code;
- •
- real estate investment trusts or regulated investment companies;
- •
- pension plans;
- •
- partnerships, or other entities or arrangements treated as partnerships for U.S. federal income tax purposes, or investors in any such
entities);
- •
- persons for whom our stock constitutes "qualified small business stock" within the meaning of Section 1202 of the Code;
- •
- integral parts or controlled entities of foreign sovereigns;
150
- •
- tax-qualified retirement plans;
- •
- controlled foreign corporations;
- •
- passive foreign investment companies and corporations that accumulate earnings to avoid U.S. federal income tax; or
- •
- persons that acquire our common stock as compensation for services.
In addition, if a partnership, including any entity or arrangement classified as a partnership for U.S. federal income tax purposes, holds our common stock, the tax treatment of a partner generally will depend on the status of the partner, the activities of the partnership, and certain determinations made at the partner level. Accordingly, partnerships that hold our common stock, and partners in such partnerships, should consult their tax advisors regarding the U.S. federal income tax consequences to them of the purchase, ownership, and disposition of our common stock.
You are urged to consult your tax advisor with respect to the application of the U.S. federal income tax laws to your particular situation, as well as any tax consequences of the purchase, ownership and disposition of our common stock arising under the U.S. federal estate or gift tax rules or under the laws of any U.S. state or local or any non-U.S. or other taxing jurisdiction or under any applicable tax treaty.
Definition of a non-U.S. holder
For purposes of this summary, a "Non-U.S. Holder" is any beneficial owner of our common stock that is, for U.S. federal income tax purposes, not a "U.S. person," not a partnership, and not an entity disregarded from its owner. A U.S. person is any person that, for U.S. federal income tax purposes, is or is treated as any of the following:
- •
- an individual who is a citizen or resident of the United States;
- •
- a corporation (or other entity classified for U.S. federal income tax purposes as a corporation) created or organized under the laws of the
United States, any state thereof, or the District of Columbia;
- •
- an estate, the income of which is subject to U.S. federal income tax regardless of its source; or
- •
- a trust that (1) is subject to the primary supervision of a U.S. court and the control of one or more U.S. persons (within the meaning of Section 7701(a)(30) of the Code), or (2) has a valid election in effect to be treated as a U.S. person for U.S. federal income tax purposes.
As discussed under "Dividend policy," above, we do not anticipate paying any dividends on our capital stock in the foreseeable future. If we make distributions on our common stock, those payments will constitute dividends for U.S. income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. To the extent those distributions exceed both our current and our accumulated earnings and profits, they will constitute a return of capital and will first reduce a Non-U.S. Holder's basis in our common stock, but not below zero. Any excess will be treated as capital gain and will be treated as described below under "Gain on sale or other disposition of common stock." Any such distributions would be subject to the discussions below regarding back-up withholding and FATCA.
151
Subject to the discussion below on effectively connected income, any dividend paid to a Non-U.S. Holder generally will be subject to U.S. withholding tax either at a rate of 30% of the gross amount of the dividend or such lower rate as may be specified by an applicable income tax treaty. In order to receive a reduced treaty rate, a Non-U.S. Holder must provide us or our agent with an IRS Form W-8BEN (generally including a U.S. taxpayer identification number), IRS Form W-8-BEN-E or another appropriate version of IRS Form W-8 (or a successor form), which must be updated periodically, and which, in each case, must certify qualification for the reduced rate. Non-U.S. Holders should consult their tax advisors regarding their entitlement to benefits under any applicable income tax treaty.
Dividends paid to a Non-U.S. Holder that are effectively connected with the Non-U.S. Holder's conduct of a U.S. trade or business within the United States (and, if required by an applicable income tax treaty, are attributable to a permanent establishment maintained by the Non-U.S. Holder in the United States) generally are exempt from the withholding tax described above. In order to obtain this exemption, the Non-U.S. Holder must provide the applicable withholding agent with an IRS Form W-8ECI or successor form or other applicable IRS Form W-8 certifying that the dividends are effectively connected with the Non-U.S. Holder's conduct of a trade or business within the United States. Such effectively connected dividends, although not subject to withholding tax, are taxed at the same graduated rates applicable to U.S. persons, net of certain deductions and credits, subject to an applicable income tax treaty providing otherwise. In addition, if you are a Non-U.S. Holder that is a corporation, dividends you receive that are effectively connected with your conduct of a U.S. trade or business (and, if an income tax treaty applies, are attributable to a permanent establishment maintained by the you in the United States) may also be subject to a branch profits tax at a rate of 30% (or such lower rate as may be specified by an applicable income tax treaty) on such effectively connected dividends, as adjusted for certain items.
If you are eligible for a reduced rate of withholding tax pursuant to a tax treaty, you may be able to obtain a refund of any excess amounts currently withheld if you timely file an appropriate claim for refund with the IRS.
Gain on sale or other disposition of common stock
Subject to the discussion below regarding backup withholding and FATCA, a Non-U.S. Holder generally will not be required to pay U.S. federal income tax on any gain realized upon the sale or other disposition of our common stock unless:
- •
- the gain is effectively connected with the Non-U.S. Holder's conduct of a trade or business within the United States (and, if an income tax
treaty applies, the gain is attributable to a permanent establishment maintained by the Non-U.S. Holder in the U.S.), in which case the Non-U.S. Holder will be required to pay tax on the net gain
derived from the sale under regular graduated U.S. federal income tax rates, and for a Non-U.S. Holder that is a corporation, such Non-U.S. Holder may be subject to the branch profits tax at a 30%
rate (or such lower rate as may be specified by an applicable income tax treaty) on such effectively connected dividends, as adjusted for certain items;
- •
- the Non-U.S. Holder is an individual who is present in the United States for a period or periods aggregating 183 days or more during the calendar year in which the sale or disposition occurs and certain other conditions are met, in which case the Non-U.S. Holder will be required to pay a flat 30% tax on the gain derived from the sale, which tax may be offset by U.S. source capital losses (even though the Non-U.S. Holder is not considered a resident of the United States) (subject to applicable income tax or other treaties); or
152
- •
- our common stock constitutes a U.S. real property interest by reason of our status as a "U.S. real property holding corporation" for U.S. federal income tax purposes, a USRPHC, at any time within the shorter of the five-year period preceding the disposition or the Non-U.S. Holder's holding period for our common stock. We believe we are not currently and do not anticipate becoming a USRPHC. However, because the determination of whether we are a USRPHC depends on the fair market value of our U.S. real property interests relative to the fair market value of our other business assets, there can be no assurance that we will not become a USRPHC in the future. Even if we become a USRPHC, however, gain arising from the sale or other taxable disposition by a Non-U.S. Holder of our common stock will not be subject to U.S. federal income tax as long as our common stock is regularly traded on an established securities market and such Non-U.S. Holder does not, actually or constructively, hold more than five percent of our common stock at any time during the applicable period that is specified in the Code. If the foregoing exception does not apply, then if we are or were to become a USRPHC a purchaser may be required to withhold 15% of the proceeds payable to a Non-U.S. Holder from a sale of our common stock and such Non-U.S. Holder generally will be taxed on its net gain derived from the disposition at the graduated U.S. federal income tax rates applicable to United States persons (as defined in the Code).
Backup withholding and information reporting
Generally, we must file information returns annually to the IRS in connection with any dividends on our common stock paid to a Non-U.S. Holder, regardless of whether any tax was withheld. A similar report will be sent to the Non-U.S. Holder. Pursuant to applicable income tax treaties or other agreements, the IRS may make these reports available to tax authorities in the Non-U.S. Holder's country of residence.
Payments of dividends or of proceeds on the disposition of stock made to a Non-U.S. Holder may be subject to additional information reporting and backup withholding at a current rate of 28% unless such Non-U.S. Holder establishes an exemption, for example by properly certifying its non-U.S. status on an IRS Form W-8BEN, IRS Form W-8BEN-E, IRS Form W-8ECI, or another appropriate version of IRS Form W-8 (or a successor form). Notwithstanding the foregoing, backup withholding and information reporting may apply if either we or our paying agent has actual knowledge, or reason to know, that a holder is a U.S. person.
Backup withholding is not an additional tax; rather, the U.S. income tax liability of persons subject to backup withholding will be reduced by the amount of tax withheld. If withholding results in an overpayment of taxes, a refund or credit may generally be obtained from the IRS, provided that the required information is furnished to the IRS in a timely manner.
Foreign account tax compliance act
The Foreign Account Tax Compliance Act, or FATCA, imposes withholding tax on certain types of payments made to foreign financial institutions and certain other non-U.S. entities. The law imposes a 30% withholding tax on dividends from, and beginning January 1, 2019, gross proceeds from the sale or other disposition of, our common stock paid to a "foreign financial institution" or to certain "non-financial foreign entities" (each as defined in the Code), unless (i) the foreign financial institution undertakes certain diligence and reporting obligations, (ii) the non-financial foreign entity either certifies it does not have any "substantial United States owners" (as defined in the Code) or furnishes identifying information regarding each substantial United States owner, or (iii) the foreign financial institution or non-financial foreign entity otherwise qualifies for an exemption from these rules. If the payee is a foreign financial institution and is subject to the diligence and reporting requirements in (i) above, it must enter into an agreement with the U.S. Treasury requiring, among other things, that it undertake to identify accounts held by "specified United States persons" or "United States-owned foreign entities" (each as defined in the Code), annually
153
report certain information about such accounts, and withhold 30% on payments to account holders whose actions prevent it from complying with these reporting and other requirements. If the country in which a payee is resident has entered into an "intergovernmental agreement" with the United States regarding FATCA, that agreement may permit the payee to report to that country rather than to the U.S. Department of the Treasury. Prospective investors should consult their own tax advisors regarding the possible impact of these rules on their investment in our common stock, and the possible impact of these rules on the entities through which they hold our common stock, including, without limitation, the process and deadlines for meeting the applicable requirements to prevent the imposition of this 30% withholding tax under FATCA.
Common stock we have issued that is owned (or treated as owned) by an individual who is not a citizen or a resident of the United States (as defined for U.S. federal estate tax purposes) at the time of death will be included in the individual's gross estate for U.S. federal estate tax purposes unless an applicable estate or other tax treaty provides otherwise, and therefore may be subject to U.S. federal estate tax.
The preceding discussion of U.S. federal tax considerations is for general information only. It is not tax advice. Each prospective investor should consult its tax advisor regarding the particular U.S. federal, state and local and non-U.S. tax consequences of purchasing, holding and disposing of our common stock, including the consequences of any proposed change in applicable laws.
154
We are offering the shares of common stock described in this prospectus through a number of underwriters. J.P. Morgan Securities LLC and Leerink Partners LLC are acting as joint book-running managers of the offering and as representatives of the underwriters. Subject to the terms and conditions of the underwriting agreement, we have agreed to sell to the underwriters, and each underwriter has severally agreed to purchase, at the public offering price less the underwriting discounts and commissions set forth on the cover page of this prospectus, the number of shares of common stock listed next to its name in the following table:
| |
|
|||
|---|---|---|---|---|
| | | | | |
| Name |
Number of shares |
|||
| | | | | |
J.P. Morgan Securities LLC |
||||
Leerink Partners LLC |
||||
BTIG, LLC |
||||
Evercore Group L.L.C. |
||||
| | | | | |
Total |
||||
| | | | | |
The underwriters are committed to purchase all the shares of common stock offered by us if they purchase any shares. The underwriting agreement also provides that if an underwriter defaults, the purchase commitments of non-defaulting underwriters may also be increased or the offering may be terminated.
The underwriters propose to offer the shares of common stock directly to the public at the initial public offering price set forth on the cover page of this prospectus and to certain dealers at that price less a concession not in excess of $ per share. After the initial offering of the shares to the public, the offering price and other selling terms may be changed by the underwriters. Sales of shares made outside of the United States may be made by affiliates of the underwriters.
The underwriters have an option to buy up to additional shares of common stock from us to cover sales of shares by the underwriters which exceed the number of shares specified in the table above. The underwriters have 30 days from the date of this prospectus to exercise this option to purchase additional shares. If any shares are purchased with this option to purchase additional shares, the underwriters will purchase shares in approximately the same proportion as shown in the table above. If any additional shares of common stock are purchased, the underwriters will offer the additional shares on the same terms as those on which the shares are being offered.
The underwriting fee is equal to the public offering price per share of common stock less the amount paid by the underwriters to us per share of common stock. The underwriting fee is $ per share. The following table shows the per share and total underwriting discounts and commissions to be paid to the
155
underwriters assuming both no exercise and full exercise of the underwriters' option to purchase additional shares.
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
| |
Without option to purchase additional shares exercise |
With full option to purchase additional shares exercise |
|||||
| | | | | | | | |
Per Share |
$ | $ | |||||
Total |
$ | $ | |||||
| | | | | | | | |
We estimate that the total expenses of this offering, including registration, filing and listing fees, printing fees and legal and accounting expenses, but excluding the underwriting discounts and commissions, will be approximately $ . We have agreed to reimburse the underwriters for expenses relating to the clearance of this offering with the Financial Industry Regulatory Authority, Inc. in an amount up to $ .
A prospectus in electronic format may be made available on the web sites maintained by one or more underwriters, or selling group members, if any, participating in the offering. The underwriters may agree to allocate a number of shares to underwriters and selling group members for sale to their online brokerage account holders. Internet distributions will be allocated by the representatives to underwriters and selling group members that may make Internet distributions on the same basis as other allocations.
We have agreed that we will not (i) offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase or otherwise transfer or dispose of, directly or indirectly, or file with the SEC a registration statement under the Securities Act relating to, any shares of our common stock or securities convertible into or exchangeable or exercisable for any shares of our common stock, or publicly disclose the intention to make any offer, sale, pledge, disposition or filing, or (ii) enter into any swap or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of any shares of common stock or any such other securities (regardless of whether any of these transactions are to be settled by the delivery of shares of common stock or such other securities, in cash or otherwise), in each case without the prior written consent of J.P. Morgan Securities LLC and Leerink Partners LLC for a period of 180 days after the date of the final prospectus for this offering, other than: the shares of our common stock to be sold in this offering; any shares of our common stock issued upon the exercise of options granted under our existing stock-based compensation plans; shares of our common stock issued upon exercise of any warrant or conversion of our preferred stock outstanding as of the date of the underwriting agreement; up to 5% of the total number of outstanding shares of our common stock immediately following the issuance of the shares in this offering, issued by us in connection with mergers, acquisitions or commercial or strategic transactions (including, without limitation, joint ventures, marketing or distribution arrangements, collaboration agreements or intellectual property licenses); and the filing by us of any registration statement on Form S-8 or a successor form thereto relating to a stock-based compensation plan described in this prospectus.
All of our directors and executive officers, and substantially all of our stockholders, warrantholders and optionholders have entered into lock-up agreements with the underwriters prior to the commencement of this offering, which we refer to as the locked-up parties, pursuant to which each of these persons or entities, with limited exceptions, for a period of 180 days after the date of the final prospectus for this
156
offering, may not, without the prior written consent of J.P. Morgan Securities LLC, Leerink Partners LLC and Evercore Group L.L.C., (1) offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, or otherwise transfer or dispose of, directly or indirectly, any shares of our common stock or any securities convertible into or exercisable or exchangeable for our common stock (including, without limitation, common stock or such other securities which may be deemed to be beneficially owned by such locked-up parties in accordance with the rules and regulations of the SEC and securities which may be issued upon exercise of a stock option or warrant), or publicly disclose the intention to make any offer, sale, pledge or disposition, (2) enter into any swap or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of the common stock or such other securities, whether any such transaction described in clause (1) or (2) above is to be settled by delivery of common stock or such other securities, in cash or otherwise, or (3) make any demand for or exercise any right with respect to the registration of any shares of our common stock or any security convertible into or exercisable or exchangeable for our common stock.
Each such locked-up party has agreed that, subject to certain conditions, the foregoing restrictions shall not apply to certain transactions, including:
(i) transfers of shares as a bona fide gift or gifts or to a trust the beneficiaries of which are exclusively such locked-up party or members of their immediate family, or by will or intestate succession upon the death of such locked-up party;
(ii) if the locked-up party is a corporation, partnership, limited liability company or other business entity, distributions of shares to members or stockholders of such locked-up party;
(iii) if the locked-up party is a corporation, partnership, limited liability company or other business entity, any transfer made by such locked-up party to another corporation, partnership, limited liability company or other business entity so long as the transferee controls, is controlled by or is under common control with such locked-up party and such transfer is not for value;
(iv) transactions relating to common stock or other securities convertible into or exercisable or exchangeable for common stock acquired by such locked-up party in this offering or in open market transactions after completion of this offering;
(v) the entry into any trading plan providing for the sale of common stock by such locked-up party, which trading plan meets the requirements of Rule 10b5-1(c) under the Exchange Act, provided, however, that such plan does not provide for, or permit, the sale of any common stock during the lock-up period and no filing under Section 16(a) of the Exchange Act or other public announcement is voluntarily made or required regarding such plan during the lock-up period;
(vi) any transfers made by such locked-up party to the Company solely to satisfy tax withholding obligations pursuant to our equity incentive plans or arrangements disclosed in this prospectus, provided that no filing under Section 16(a) of the Exchange Act or other public announcement is voluntarily made regarding such transfers during the lock-up period, and provided, further, that any filing required under Section 16(a) of the Exchange Act shall clearly indicate in the codes and footnotes thereto that any such disposition of shares was made solely to satisfy such locked-up party's tax withholding obligations;
(vii) any transfers made by such locked-up party by operation of law, such as pursuant to a qualified domestic order or in connection with a divorce settlement;
157
(viii) to the Company pursuant to agreements under which we have the option to repurchase shares or shares are forfeited upon termination of service of such locked-up party;
(ix) conversion of preferred stock into shares of common stock in connection with the completion of this offering (any such shares of common stock received by such locked-up party upon such conversion shall be subject to the lock-up restrictions); or
(x) dispositions solely in connection with the "cashless" exercise of stock options or warrants to acquire shares of common stock described in this prospectus or issued pursuant to an equity plan or arrangement described in this prospectus for the purpose of exercising such stock options or warrants, in any event, solely if such stock options or warrants would otherwise expire (any such shares of common stock received upon such exercise shall be subject to all of the lock-up restrictions), provided that any filing required under Section 16(a) of the Exchange Act shall clearly indicate in the codes and footnotes thereto that any disposition of shares in connection with a "cashless" exercise was made solely to the Company.
We have agreed to indemnify the underwriters against certain liabilities, including liabilities under the Securities Act.
We have applied to have our common stock approved for listing on The Nasdaq Global Market under the symbol "QTRX."
In connection with this offering, the underwriters may engage in stabilizing transactions, which involves making bids for, purchasing and selling shares of common stock in the open market for the purpose of preventing or retarding a decline in the market price of the common stock while this offering is in progress. These stabilizing transactions may include making short sales of the common stock, which involves the sale by the underwriters of a greater number of shares of common stock than they are required to purchase in this offering, and purchasing shares of common stock on the open market to cover positions created by short sales. Short sales may be "covered" shorts, which are short positions in an amount not greater than the underwriters' option to purchase additional shares referred to above, or may be "naked" shorts, which are short positions in excess of that amount. The underwriters may close out any covered short position either by exercising their option to purchase additional shares, in whole or in part, or by purchasing shares in the open market. In making this determination, the underwriters will consider, among other things, the price of shares available for purchase in the open market compared to the price at which the underwriters may purchase shares through the option to purchase additional shares. A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of the common stock in the open market that could adversely affect investors who purchase in this offering. To the extent that the underwriters create a naked short position, they will purchase shares in the open market to cover the position.
The underwriters have advised us that, pursuant to Regulation M of the Securities Act, they may also engage in other activities that stabilize, maintain or otherwise affect the price of the common stock, including the imposition of penalty bids. This means that if the representatives of the underwriters purchase common stock in the open market in stabilizing transactions or to cover short sales, the representatives can require the underwriters that sold those shares as part of this offering to repay the underwriting discount received by them.
These activities may have the effect of raising or maintaining the market price of the common stock or preventing or retarding a decline in the market price of the common stock and, as a result, the price of the common stock may be higher than the price that otherwise might exist in the open market. If the
158
underwriters commence these activities, they may discontinue them at any time. The underwriters may carry out these transactions on The Nasdaq Global Market, in the over-the-counter market or otherwise.
Prior to this offering, there has been no public market for our common stock. The initial public offering price will be determined by negotiations between us and the representatives of the underwriters. In determining the initial public offering price, we and the representatives of the underwriters expect to consider a number of factors including:
- •
- the information set forth in this prospectus and otherwise available to the representatives;
- •
- our prospects and the history and prospects for the industry in which we compete;
- •
- an assessment of our management;
- •
- our prospects for future earnings;
- •
- the general condition of the securities markets at the time of this offering;
- •
- the recent market prices of, and demand for, publicly traded common stock of generally comparable companies; and
- •
- other factors deemed relevant by the underwriters and us.
Neither we nor the underwriters can assure investors that an active trading market will develop for our common stock, or that the shares will trade in the public market at or above the initial public offering price.
Other than in the United States, no action has been taken by us or the underwriters that would permit a public offering of the securities offered by this prospectus in any jurisdiction where action for that purpose is required. The securities offered by this prospectus may not be offered or sold, directly or indirectly, nor may this prospectus or any other offering material or advertisements in connection with the offer and sale of any such securities be distributed or published in any jurisdiction, except under circumstances that will result in compliance with the applicable rules and regulations of that jurisdiction. Persons into whose possession this prospectus comes are advised to inform themselves about and to observe any restrictions relating to the offering and the distribution of this prospectus. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities offered by this prospectus in any jurisdiction in which such an offer or a solicitation is unlawful.
Certain of the underwriters and their affiliates may provide from time to time in the future certain commercial banking, financial advisory, investment banking and other services for us and our affiliates in the ordinary course of their business, for which they may receive customary fees and commissions. In addition, from time to time, certain of the underwriters and their affiliates may effect transactions for their own account or the account of customers, and hold on behalf of themselves or their customers, long or short positions in our debt or equity securities or loans, and may do so in the future.
In relation to each Member State of the European Economic Area which has implemented the Prospectus Directive (each, a "Relevant Member State"), with effect from and including the date on which the
159
Prospectus Directive is implemented in that Relevant Member State, no offer of shares may be made to the public in that Relevant Member State other than:
A. to any legal entity which is a qualified investor as defined in the Prospectus Directive;
B. to fewer than 150 natural or legal persons (other than qualified investors as defined in the Prospectus Directive), subject to obtaining the prior consent of the underwriters; or
C. in any other circumstances falling within Article 3(2) of the Prospectus Directive;
provided that no such offer of shares shall require the Company or any underwriter to publish a prospectus pursuant to Article 3 of the Prospectus Directive or supplement a prospectus pursuant to Article 16 of the Prospectus Directive and each person who initially acquires any shares or to whom any offer is made will be deemed to have represented, acknowledged and agreed to and with each of the underwriters and the Company that it is a "qualified investor" within the meaning of the law in that Relevant Member State implementing Article 2(1)(e) of the Prospectus Directive.
In the case of any shares being offered to a financial intermediary as that term is used in Article 3(2) of the Prospectus Directive, each such financial intermediary will be deemed to have represented, acknowledged and agreed that the shares acquired by it in the offer have not been acquired on a non-discretionary basis on behalf of, nor have they been acquired with a view to their offer or resale to, persons in circumstances which may give rise to an offer of any shares to the public other than their offer or resale in a Relevant Member State to qualified investors as so defined or in circumstances in which the prior consent of the representatives has been obtained to each such proposed offer or resale.
For the purposes of this provision, the expression an "offer of shares to the public" in relation to any shares in any Relevant Member State means the communication in any form and by means of sufficient information on the terms of the offer and the shares to be offered so as to enable an investor to decide to purchase shares, as the same may be varied in that Member State by any measure implementing the Prospectus Directive in that Member State, the expression "Prospectus Directive" means Directive 2003/71/EC (as amended, including by Directive 2010/73/EU), and includes any relevant implementing measure in the Relevant Member State.
In addition, in the United Kingdom, this document is being distributed only to, and is directed only at, and any offer subsequently made may only be directed at persons who are "qualified investors" (as defined in the Prospectus Directive) (i) who have professional experience in matters relating to investments falling within Article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005, as amended (the "Order") and/or (ii) who are high net worth companies (or persons to whom it may otherwise be lawfully communicated) falling within Article 49(2)(a) to (d) of the Order (all such persons together being referred to as "relevant persons") or otherwise in circumstances which have not resulted and will not result in an offer to the public of the shares in the United Kingdom within the meaning of the Financial Services and Markets Act 2000.
Any person in the United Kingdom that is not a relevant person should not act or rely on the information included in this document or use it as basis for taking any action. In the United Kingdom, any investment or investment activity that this document relates to may be made or taken exclusively by relevant persons.
160
The shares may be sold only to purchasers purchasing, or deemed to be purchasing, as principal that are accredited investors, as defined in National Instrument 45-106 Prospectus Exemptions or subsection 73.3(1) of the Securities Act (Ontario), and are permitted clients, as defined in National Instrument 31-103 Registration Requirements, Exemptions and Ongoing Registrant Obligations. Any resale of the shares must be made in accordance with an exemption from, or in a transaction not subject to, the prospectus requirements of applicable securities laws.
Securities legislation in certain provinces or territories of Canada may provide a purchaser with remedies for rescission or damages if this prospectus (including any amendment thereto) contains a misrepresentation, provided that the remedies for rescission or damages are exercised by the purchaser within the time limit prescribed by the securities legislation of the purchaser's province or territory. The purchaser should refer to any applicable provisions of the securities legislation of the purchaser's province or territory for particulars of these rights or consult with a legal advisor.
Pursuant to section 3A.3 of National Instrument 33-105 Underwriting Conflicts (NI 33-105), the underwriters are not required to comply with the disclosure requirements of NI 33-105 regarding underwriter conflicts of interest in connection with this offering.
The shares may not be publicly offered in Switzerland and will not be listed on the SIX Swiss Exchange ("SIX") or on any other stock exchange or regulated trading facility in Switzerland. This document does not constitute a prospectus within the meaning of, and has been prepared without regard to the disclosure standards for issuance prospectuses under art. 652a or art. 1156 of the Swiss Code of Obligations or the disclosure standards for listing prospectuses under art. 27 ff. of the SIX Listing Rules or the listing rules of any other stock exchange or regulated trading facility in Switzerland. Neither this document nor any other offering or marketing material relating to the shares or the offering may be publicly distributed or otherwise made publicly available in Switzerland.
Neither this document nor any other offering or marketing material relating to the offering, the Company or the shares has been or will be filed with or approved by any Swiss regulatory authority. In particular, this document will not be filed with, and the offer of shares will not be supervised by, the Swiss Financial Market Supervisory Authority FINMA (FINMA), and the offer of shares has not been and will not be authorized under the Swiss Federal Act on Collective Investment Schemes ("CISA"). The investor protection afforded to acquirers of interests in collective investment schemes under the CISA does not extend to acquirers of shares.
Dubai International Financial Centre ("DIFC")
This document relates to an Exempt Offer in accordance with the Markets Rules 2012 of the Dubai Financial Services Authority ("DFSA"). This document is intended for distribution only to persons of a type specified in the Markets Rules 2012 of the DFSA. It must not be delivered to, or relied on by, any other person. The DFSA has no responsibility for reviewing or verifying any documents in connection with Exempt Offers. The DFSA has not approved this prospectus nor taken steps to verify the information set forth herein and has no responsibility for this document. The securities to which this document relates may be illiquid and/or subject to restrictions on their resale. Prospective purchasers of the securities offered
161
should conduct their own due diligence on the securities. If you do not understand the contents of this document you should consult an authorized financial advisor.
In relation to its use in the DIFC, this document is strictly private and confidential and is being distributed to a limited number of investors and must not be provided to any person other than the original recipient, and may not be reproduced or used for any other purpose. The interests in the securities may not be offered or sold directly or indirectly to the public in the DIFC.
The shares have not been, and are not being, publicly offered, sold, promoted or advertised in the United Arab Emirates (including the DIFC) other than in compliance with the laws of the United Arab Emirates (and the DIFC) governing the issue, offering and sale of securities. Further, this prospectus does not constitute a public offer of securities in the United Arab Emirates (including the DIFC) and is not intended to be a public offer. This prospectus has not been approved by or filed with the Central Bank of the United Arab Emirates, the Securities and Commodities Authority or the DFSA.
This prospectus:
- •
- does not constitute a product disclosure document or a prospectus under Chapter 6D.2 of the Corporations Act 2001 (Cth) (the
"Corporations Act");
- •
- has not been, and will not be, lodged with the Australian Securities and Investments Commission ("ASIC"), as a disclosure document for the
purposes of the Corporations Act and does not purport to include the information required of a disclosure document under Chapter 6D.2 of the Corporations Act;
- •
- does not constitute or involve a recommendation to acquire, an offer or invitation for issue or sale, an offer or invitation to arrange the
issue or sale, or an issue or sale, of interests to a "retail client" (as defined in section 761G of the Corporations Act and applicable regulations) in Australia; and
- •
- may only be provided in Australia to select investors who are able to demonstrate that they fall within one or more of the categories of investors, or Exempt Investors, available under section 708 of the Corporations Act.
The shares may not be directly or indirectly offered for subscription or purchased or sold, and no invitations to subscribe for or buy the shares may be issued, and no draft or definitive offering memorandum, advertisement or other offering material relating to any shares may be distributed in Australia, except where disclosure to investors is not required under Chapter 6D of the Corporations Act or is otherwise in compliance with all applicable Australian laws and regulations. By submitting an application for the shares, you represent and warrant to us that you are an Exempt Investor.
As any offer of shares under this document will be made without disclosure in Australia under Chapter 6D.2 of the Corporations Act, the offer of those securities for resale in Australia within 12 months may, under section 707 of the Corporations Act, require disclosure to investors under Chapter 6D.2 if none of the exemptions in section 708 applies to that resale. By applying for the shares you undertake to us that you will not, for a period of 12 months from the date of issue of the shares, offer, transfer, assign or otherwise alienate those securities to investors in Australia except in circumstances where disclosure to investors is not required under Chapter 6D.2 of the Corporations Act or where a compliant disclosure document is prepared and lodged with ASIC.
162
The shares have not been and will not be registered pursuant to Article 4, Paragraph 1 of the Financial Instruments and Exchange Act. Accordingly, none of the shares nor any interest therein may be offered or sold, directly or indirectly, in Japan or to, or for the benefit of, any "resident" of Japan (which term as used herein means any person resident in Japan, including any corporation or other entity organized under the laws of Japan), or to others for re-offering or resale, directly or indirectly, in Japan or to or for the benefit of a resident of Japan, except pursuant to an exemption from the registration requirements of, and otherwise in compliance with, the Financial Instruments and Exchange Act and any other applicable laws, regulations and ministerial guidelines of Japan in effect at the relevant time.
The shares have not been offered or sold and will not be offered or sold in Hong Kong, by means of any document, other than (a) to "professional investors" as defined in the Securities and Futures Ordinance (Cap. 571) of Hong Kong and any rules made under that Ordinance; or (b) in other circumstances which do not result in the document being a "prospectus" as defined in the Companies (Winding Up and Miscellaneous Provisions) Ordinance (Cap. 32) of Hong Kong or which do not constitute an offer to the public within the meaning of that Ordinance. No advertisement, invitation or document relating to the shares has been or may be issued or has been or may be in the possession of any person for the purposes of issue, whether in Hong Kong or elsewhere, which is directed at, or the contents of which are likely to be accessed or read by, the public of Hong Kong (except if permitted to do so under the securities laws of Hong Kong) other than with respect to shares which are or are intended to be disposed of only to persons outside Hong Kong or only to "professional investors" as defined in the Securities and Futures Ordinance and any rules made under that Ordinance.
This prospectus has not been registered as a prospectus with the Monetary Authority of Singapore. Accordingly, this prospectus and any other document or material in connection with the offer or sale, or invitation for subscription or purchase, of shares may not be circulated or distributed, nor may the shares be offered or sold, or be made the subject of an invitation for subscription or purchase, whether directly or indirectly, to persons in Singapore other than (i) to an institutional investor under Section 274 of the Securities and Futures Act, Chapter 289 of Singapore (the "SFA"), (ii) to a relevant person pursuant to Section 275(1), or any person pursuant to Section 275(1A), and in accordance with the conditions specified in Section 275 of the SFA, or (iii) otherwise pursuant to, and in accordance with the conditions of, any other applicable provision of the SFA.
Where the shares are subscribed or purchased under Section 275 of the SFA by a relevant person which is:
a) a corporation (which is not an accredited investor (as defined in Section 4A of the SFA)) the sole business of which is to hold investments and the entire share capital of which is owned by one or more individuals, each of whom is an accredited investor; or
b) a trust (where the trustee is not an accredited investor) whose sole purpose is to hold investments and each beneficiary of the trust is an individual who is an accredited investor;
securities (as defined in Section 239(1) of the SFA) of that corporation or the beneficiaries' rights and interest (howsoever described) in that trust shall not be transferred within six months after that
163
corporation or that trust has acquired the shares pursuant to an offer made under Section 275 of the SFA except:
a) to an institutional investor or to a relevant person defined in Section 275(2) of the SFA, or to any person arising from an offer referred to in Section 275(1A) or Section 276(4)(i)(B) of the SFA;
b) where no consideration is or will be given for the transfer;
c) where the transfer is by operation of law;
d) as specified in Section 276(7) of the SFA; or
e) as specified in Regulation 32 of the Securities and Futures (Offers of Investments) (Shares and Debentures) Regulations 2005 of Singapore.
164
The validity of the shares of common stock offered by this prospectus will be passed upon for us by Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C., Boston, Massachusetts. Certain legal matters in connection with this offering will be passed upon for the underwriters by Ropes & Gray LLP, Boston, Massachusetts.
Ernst & Young LLP, independent registered accounting firm, has audited our consolidated financial statements at December 31, 2016 and 2015, and for each of the years then ended, as set forth in their report. We have included our consolidated financial statements in the prospectus and elsewhere in the registration statement in reliance on Ernst & Young LLP's report, given on their authority as experts in accounting and auditing.
Where you can find more information
We have filed with the SEC a registration statement on Form S-1, including exhibits and schedules, under the Securities Act that registers the shares of our common stock to be sold in this offering. This prospectus does not contain all the information contained in the registration statement and the exhibits and schedules filed as part of the registration statement. For further information with respect to us and our common stock, we refer you to the registration statement and the exhibits and schedules filed as part of the registration statement. Statements contained in this prospectus as to the contents of any contract or other document are not necessarily complete. If a contract or document has been filed as an exhibit to the registration statement, we refer you to the copies of the contract or document that has been filed. Each statement in this prospectus relating to a contract or document filed as an exhibit is qualified in all respects by the filed exhibit.
Upon the completion of this offering, we will file annual, quarterly and current reports, proxy statements and other information with the SEC under the Exchange Act. You can read our SEC filings, including the registration statement, at the SEC's website at www.sec.gov.
You may read and copy this information at the SEC's Public Reference Room at 100 F Street, N.E., Washington D.C. 20549, at prescribed rates. You may obtain information regarding the operation of the public reference room by calling the SEC at 1-800-SEC-0330. The SEC also maintains a website (http://www.sec.gov) that contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC.
Our website address is www.quanterix.com. The information contained in, and that can be accessed through, our website is not incorporated into and is not part of this prospectus.
165
Index to consolidated financial statements
F-1
Report of independent registered public accounting firm
The
Board of Directors and Stockholders
Quanterix Corporation
We have audited the accompanying consolidated balance sheets of Quanterix Corporation as of December 31, 2015 and 2016, and the related consolidated statements of operations and comprehensive loss, redeemable convertible preferred stock and stockholders' (deficit) equity and cash flows for the years then ended. These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. We were not engaged to perform an audit of the Company's internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the consolidated financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of Quanterix Corporation at December 31, 2015 and 2016, and the consolidated results of its operations and its cash flows for the years then ended in conformity with U.S. generally accepted accounting principles.
| /s/ Ernst & Young LLP |
Boston,
Massachusetts
July 20, 2017, except for note 14(a),
as to which the date is August 31, 2017
F-2
Quanterix Corporation
Consolidated balance sheets
(amounts in thousands, except share and per share data)
| | | | | | | | | | | | | | |
| |
December 31, | September 30, | Pro forma September 30, |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2015 |
2016 |
2017 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
(unaudited) |
|||||||||
Assets |
|||||||||||||
Current assets: |
|||||||||||||
Cash and cash equivalents |
$ | 2,323 | $ | 29,671 | 18,690 | 18,690 | |||||||
Accounts receivable (including $63, $124, and $13 from related parties as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively) |
2,262 | 3,917 | 4,204 | 4,204 | |||||||||
Inventory |
1,002 | 1,528 | 2,563 | 2,563 | |||||||||
Prepaid expenses and other current assets |
133 | 127 | 486 | 486 | |||||||||
| | | | | | | | | | | | | | |
Total current assets |
5,720 | 35,243 | 25,943 | 25,943 | |||||||||
Property and equipment, net |
1,080 | 1,223 | 1,882 | 1,882 | |||||||||
Other non-current assets |
551 | 651 | 2,690 | 2,690 | |||||||||
| | | | | | | | | | | | | | |
Total assets |
$ | 7,351 | $ | 37,117 | $ | 30,515 | $ | 30,515 | |||||
| | | | | | | | | | | | | | |
Liabilities, redeemable convertible preferred stock and stockholders' (deficit) equity |
|||||||||||||
Current liabilities: |
|||||||||||||
Accounts payable (including $13, $8, and $0 to related parties as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively) |
$ | 1,346 | $ | 2,549 | $ | 3,450 | $ | 3,450 | |||||
Accrued compensation and benefits |
1,546 | 1,693 | 2,118 | 2,118 | |||||||||
Other accrued expenses (including $94, $516, and $120 to related parties as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively) |
1,344 | 2,386 | 3,359 | 3,359 | |||||||||
Deferred revenue (including $394, $1,204, and $1,125 with related parties as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively) |
1,474 | 3,428 | 3,826 | 3,826 | |||||||||
Current portion of long term debt |
2,078 | 899 | 3,626 | 3,626 | |||||||||
| | | | | | | | | | | | | | |
Total current liabilities |
7,788 | 10,955 | 16,379 | 16,379 | |||||||||
Preferred stock warrant liability |
5,547 |
2,802 |
781 |
— |
|||||||||
Deferred revenue, net of current portion (including $1,226, $149, and $1,343 with related parties as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively) |
1,363 | 328 | 1,652 | 1,652 | |||||||||
Long term debt, net of current portion |
7,648 | 9,344 | 5,702 | 5,702 | |||||||||
Other non-current liabilities |
200 | 212 | 158 | 158 | |||||||||
| | | | | | | | | | | | | | |
Total liabilities |
$ | 22,546 | $ | 23,641 | $ | 24,672 | $ | 23,891 | |||||
Commitments and contingencies (Note 9) |
|||||||||||||
Redeemable convertible preferred stock: |
|||||||||||||
Series A redeemable convertible preferred stock, $0.001 par value: authorized—16,464,442 shares; issued and outstanding—14,400,001, 15,700,001 and 16,400,001 shares as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively (liquidation preference of $27,698 and $29,960 as of December 31, 2016 and September 30, 2017 (unaudited)); no shares issued and outstanding at September 30, 2017, pro forma (unaudited) |
23,898 | 28,979 | 31,925 | — | |||||||||
Series B redeemable convertible preferred stock, $0.001 par value: authorized—6,186,594 shares; issued and outstanding—5,624,106, 6,021,636 and 6,021,636 shares as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively (liquidation preference of $16,890 and $17,614 as of December 31, 2016 and September 30, 2017 (unaudited)); no shares issued and outstanding at September 30, 2017, pro forma (unaudited) |
15,178 |
17,459 |
18,134 |
— |
|||||||||
Series C redeemable convertible preferred stock, $0.001 par value: authorized—9,791,421 shares; issued and outstanding—8,605,944, as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively (liquidation preference of $36,685 and $38,400 as of December 31, 2016 and September 30, 2017 (unaudited)); no shares issued and outstanding at September 30, 2017 pro forma (unaudited) |
34,369 |
36,678 |
38,397 |
— |
|||||||||
Series D redeemable convertible preferred stock, $0.001 par value: authorized—0, 12,420,262 and 14,572,992 shares; issued and outstanding—0, 12,420,262 and 14,534,164 shares as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively (liquidation preference of $45,582 and $54,082 as of December 31, 2016 and September 30, 2017 (unaudited)); no shares issued and outstanding at September 30, 2017, pro forma (unaudited) |
— |
45,469 |
53,931 |
— |
|||||||||
| | | | | | | | | | | | | | |
Total redeemable convertible preferred stock |
73,445 | 128,585 | 142,387 | — | |||||||||
Stockholders' (deficit) equity: |
|||||||||||||
Common stock, $0.001 par value: |
|||||||||||||
Authorized—72,113,902 shares; issued and outstanding—6,354,062, 7,442,052 and 8,021,559 shares as of December 31, 2015 and 2016, and September 30, 2017 (unaudited), respectively; 53,583,304 shares issued and outstanding at September 30, 2017, pro forma (unaudited) |
6 | 7 | 8 | 54 | |||||||||
Additional paid-in capital |
— | — | — | 143,122 | |||||||||
Accumulated deficit |
(88,646 | ) | (115,116 | ) | (136,552 | ) | (136,552 | ) | |||||
| | | | | | | | | | | | | | |
Total stockholders' (deficit) equity |
(88,640 | ) | (115,109 | ) | (136,544 | ) | 6,624 | ||||||
| | | | | | | | | | | | | | |
Total liabilities, redeemable convertible preferred stock and stockholders' (deficit) equity |
$ | 7,351 | $ | 37,117 | $ | 30,515 | $ | 30,515 | |||||
| | | | | | | | | | | | | | |
See accompanying notes.
F-3
Quanterix Corporation
Consolidated statements of operations and comprehensive loss
(amounts in thousands, except share and per share data)
| | | | | | | | | | | | | | |
| |
Year ended December 31, | Nine Months Ended September 30, |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Product revenue (including related party activity of $527 and $509 for the years ended December 31, 2015 and 2016, respectively, and $401 and $237 for the nine months ended September 30, 2016 and 2017 (unaudited), respectively) |
$ | 9,477 | $ | 10,601 | $ | 7,435 | $ | 10,055 | |||||
Service and other revenue (including related party activity of $93 and $107 for the years ended December 31, 2015 and 2016, respectively, and $68 and $129 for the nine months ended September 30, 2016 and 2017 (unaudited), respectively) |
2,515 | 5,012 | 3,330 | 5,424 | |||||||||
Collaboration and license revenue (including related party activity of $188 and $172 for the years ended December 31, 2015 and 2016, respectively, and $141 and $806 for the nine months ended September 30, 2016 and 2017 (unaudited), respectively) |
188 | 1,972 | 141 | 806 | |||||||||
| | | | | | | | | | | | | | |
Total revenue |
12,180 | 17,585 | 10,906 | 16,285 | |||||||||
Operating expenses: |
|||||||||||||
Cost of product revenue (including related party activity of $415, and $322 for the years ended December 31, 2015 and 2016, respectively, and $238 and $164 for the nine months ended September 30, 2016 and 2017 (unaudited), respectively) |
5,661 | 6,299 | 4,501 | 5,573 | |||||||||
Cost of services and other revenue |
804 | 3,163 | 2,245 | 3,606 | |||||||||
Cost of license revenue, related party |
— | 375 | — | — | |||||||||
Research and development |
10,083 | 16,993 | 10,192 | 12,377 | |||||||||
Selling, general and administrative |
10,155 | 12,466 | 8,866 | 13,641 | |||||||||
| | | | | | | | | | | | | | |
Total operating expenses |
26,703 | 39,296 | 25,804 | 35,197 | |||||||||
| | | | | | | | | | | | | | |
Loss from operations |
(14,523 | ) | (21,711 | ) | (14,898 | ) | (18,912 | ) | |||||
Interest expense, net |
(1,040 | ) | (1,298 | ) | (1,012 | ) | (735 | ) | |||||
Other (expense) income, net |
(380 | ) | (164 | ) | 51 | 10 | |||||||
| | | | | | | | | | | | | | |
Net loss |
$ | (15,943 | ) | $ | (23,173 | ) | $ | (15,859 | ) | $ | (19,637 | ) | |
| | | | | | | | | | | | | | |
Reconciliation of net loss to net loss attributable to common stockholders: |
|||||||||||||
Net loss |
$ | (15,943 | ) | $ | (23,173 | ) | $ | (15,859 | ) | $ | (19,637 | ) | |
Accretion of preferred stock to redemption value |
(4,355 | ) | (4,437 | ) | (3,324 | ) | (3,301 | ) | |||||
Accrued dividends on preferred stock |
— | (8 | ) | (1 | ) | (48 | ) | ||||||
| | | | | | | | | | | | | | |
Net loss attributable to common stockholders |
$ | (20,298 | ) | $ | (27,618 | ) | $ | (19,184 | ) | $ | (22,986 | ) | |
| | | | | | | | | | | | | | |
Net loss per share attributable to common stockholders, basic and diluted |
$ | (3.48 | ) | $ | (4.01 | ) | $ | (2.83 | ) | $ | (2.96 | ) | |
| | | | | | | | | | | | | | |
Weighted-average common shares outstanding, basic and diluted |
5,827,667 | 6,887,118 | 6,782,404 | 7,768,248 | |||||||||
| | | | | | | | | | | | | | |
Pro forma net loss per share attributable to common stockholders, basic and diluted (unaudited) |
$ | (0.49 | ) | $ | (0.38 | ) | |||||||
| | | | | | | | | | | | | | |
Pro forma weighted-average common shares outstanding, basic and diluted (unaudited) |
46,512,622 | 52,088,919 | |||||||||||
| | | | | | | | | | | | | | |
See accompanying notes.
F-4
Quanterix Corporation
Period and year ended December 31, 2015 and 2016 and
nine months ended September 30, 2017
Statements of redeemable convertible preferred stock and stockholders' (deficit) equity
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Series A redeemable convertible preferred stock |
Series B redeemable convertible preferred stock |
Series C redeemable convertible preferred stock |
Series D redeemable convertible preferred stock |
|
|
|
|
|
|
||||||||||||||||||||||||||||||
| |
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||
| |
|
Common stock | Additional paid-in capital |
|
Total stockholders' (deficit) equity |
|||||||||||||||||||||||||||||||||||
| |
|
Accumulated (deficit) |
||||||||||||||||||||||||||||||||||||||
| |
Shares |
Value |
Shares |
Value |
Shares |
Value |
Shares |
Value |
|
Shares |
Value |
|||||||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Balance at December 31, 2014 |
14,400,001 | $ | 22,692 | 5,624,106 | $ | 14,266 | 6,503,780 | $ | 25,132 | — | $ | — | 5,481,637 | $ 5 | — | $ | (69,494 | ) | $ | (69,489 | ) | |||||||||||||||||||
Issuance of Series C preferred stock, net of issuance costs |
— | — | — | — | 2,102,164 | 7,000 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
Exercise of common stock options |
— | — | — | — | — | — | — | — | 113,621 | — | 43 | — | 43 | |||||||||||||||||||||||||||
Vesting of restricted stock |
— | — | — | — | — | — | — | — | 758,804 | 1 | (1) | — | — | |||||||||||||||||||||||||||
Accretion of preferred stock to redemption value |
— | 1,206 | — | 912 | — | 2,237 | — | — | — | — | (1,146) | (3,209 | ) | (4,355 | ) | |||||||||||||||||||||||||
Stock-based compensation expense |
— | — | — | — | — | — | — | — | — | — | 1,104 | — | 1,104 | |||||||||||||||||||||||||||
Net loss |
— | — | — | — | — | — | — | — | — | — | — | (15,943 | ) | (15,943 | ) | |||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Balance at December 31, 2015 |
14,400,001 | $ | 23,898 | 5,624,106 | $ | 15,178 | 8,605,944 | $ | 34,369 | — | $ | — | 6,354,062 | $ 6 | $ — | $ | (88,646 | ) | $ | (88,640 | ) | |||||||||||||||||||
Issuance of Series D preferred stock, net of issuance costs |
— | — | — | — | — | — | 12,420,262 | 45,428 | — | — | — | — | — | |||||||||||||||||||||||||||
Exercise of preferred stock warrants |
1,300,000 | 3,901 | 397,530 | 1,374 | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
Exercise of common stock options |
— | — | — | — | — | — | — | — | 292,112 | — | 213 | — | 213 | |||||||||||||||||||||||||||
Vesting of restricted stock |
— | — | — | — | — | — | — | — | 795,878 | 1 | (1) | — | — | |||||||||||||||||||||||||||
Accretion of preferred stock to redemption value |
— | 1,180 | — | 907 | — | 2,309 | — | 41 | — | — | (1,140) | (3,297 | ) | (4,437 | ) | |||||||||||||||||||||||||
Stock-based compensation expense |
— | — | — | — | — | — | — | — | — | — | 928 | — | 928 | |||||||||||||||||||||||||||
Net loss |
— | — | — | — | — | — | — | — | — | — | — | (23,173 | ) | (23,173 | ) | |||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Balance at December 31, 2016 |
15,700,001 | $ | 28,979 | 6,021,636 | $ | 17,459 | 8,605,944 | $ | 36,678 | 12,420,262 | $ | 45,469 | 7,442,052 | $ 7 | $ — | $ | (115,116 | ) | $ | (115,109 | ) | |||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Issuance of Series D-1 preferred stock, net of issuance costs |
— | — | — | — | — | — | 2,113,902 | 8,423 | — | — | — | — | — | |||||||||||||||||||||||||||
Exercise of preferred stock warrants |
700,000 | 2,078 | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||
Exercise of common stock options and vesting of restricted stock |
— | — | — | — | — | — | — | — | 579,507 | 1 | 71 | — | 72 | |||||||||||||||||||||||||||
Cumulative effect of adoption of ASU No. 2016-09 |
— | — | — | — | — | — | — | — | — | — | 141 | (141 | ) | — | ||||||||||||||||||||||||||
Accretion of preferred stock to redemption value |
— | 868 | — | 675 | — | 1,719 | — | 39 | — | — | (1,643) | (1,658 | ) | (3,301 | ) | |||||||||||||||||||||||||
Stock-based compensation expense |
— | — | — | — | — | — | — | — | — | — | 1,431 | — | 1,431 | |||||||||||||||||||||||||||
Net loss |
— | — | — | — | — | — | — | — | — | — | — | (19,637 | ) | (19,637 | ) | |||||||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Balance at September 30, 2017, (unaudited) |
16,400,001 | $ | 31,925 | 6,021,636 | $ | 18,134 | 8,605,944 | $ | 38,397 | 14,534,164 | $ | 53,931 | 8,021,559 | $ 8 | — | $ | (136,552 | ) | $ | (136,544 | ) | |||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Conversion of preferred stock into common stock (unaudited) |
(16,400,001 | ) | (31,925 | ) | (6,021,636 | ) | (18,134 | ) | (8,605,944 | ) | (38,397 | ) | (14,534,164 | ) | (53,931 | ) | 45,561,745 | 46 | 143,122 | — | 143,168 | |||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Pro forma balance at September 30, 2017 (unaudited) |
— | $ | — | — | $ | — | — | $ | — | — | $ | — | 53,583,304 | $54 | $143,122 | $ | (136,552 | ) | $ | 6,624 | ||||||||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
See accompanying notes.
F-5
Quanterix Corporation
Consolidated statements of cash flows
(amounts in thousands)
| | | | | | | | | | | | | | |
| |
Year ended December 31, |
Nine months ended September 30, |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Operating activities |
|||||||||||||
Net loss |
$ | (15,943 | ) | $ | (23,173 | ) | $ | (15,859 | ) | $ | (19,637 | ) | |
Adjustments to reconcile net loss to net cash used in operating activities: |
|||||||||||||
Depreciation expense |
425 | 444 | 331 | 337 | |||||||||
Stock-based compensation expense |
1,104 | 928 | 737 | 1,431 | |||||||||
Non-cash interest expense |
306 | 388 | 300 | 183 | |||||||||
Gain on disposal of fixed assets |
— | 11 | 11 | — | |||||||||
Non-cash research and development expense for issuance of warrants to a vendor |
— | 2,078 | — | — | |||||||||
Change in fair value of preferred stock warrants |
598 | 307 | (66 | ) | (62 | ) | |||||||
Changes in operating assets and liabilities: |
— | ||||||||||||
Accounts receivable |
(1,467 | ) | (1,655 | ) | 222 | (287 | ) | ||||||
Restricted cash and deposits |
(1 | ) | 200 | (300 | ) | — | |||||||
Prepaid expenses and other assets |
65 | 6 | (28 | ) | (358 | ) | |||||||
Inventory |
(254 | ) | (526 | ) | (461 | ) | (1,035 | ) | |||||
Other non-current assets |
— | — | — | (2,039 | ) | ||||||||
Accounts payable |
542 | 1,131 | (255 | ) | 697 | ||||||||
Accrued compensation and benefits, other accrued expenses and other liabilities |
1,302 | 1,200 | 147 | 1,344 | |||||||||
Deferred revenue |
806 | 919 | 1,667 | 1,722 | |||||||||
| | | | | | | | | | | | | | |
Net cash used in operating activities |
(12,517 | ) | (17,742 | ) | (13,554 | ) | (17,704 | ) | |||||
| | | | | | | | | | | | | | |
Investing activities |
|||||||||||||
Purchases of property and equipment |
(597 | ) | (526 | ) | (390 | ) | (793 | ) | |||||
Investment in equity securities |
— | (300 | ) | — | — | ||||||||
Proceeds from sale of property and equipment |
43 | — | — | — | |||||||||
| | | | | | | | | | | | | | |
Net cash used in investing activities |
(554 | ) | (826 | ) | (390 | ) | (793 | ) | |||||
| | | | | | | | | | | | | | |
Financing activities |
|||||||||||||
Proceeds from sale of preferred stock, net of issuance costs |
7,000 | 45,428 | 45,428 | 8,423 | |||||||||
Proceeds from exercise of stock warrants |
— | 18 | 18 | 1 | |||||||||
Proceeds from stock options exercised |
43 | 213 | 53 | 72 | |||||||||
Proceeds from the issuance of notes payable and warrants, net of issuance costs |
5,000 | 2,954 | 2,954 | (59 | ) | ||||||||
Payments on notes payable |
(339 | ) | (2,697 | ) | (1,333 | ) | (921 | ) | |||||
| | | | | | | | | | | | | | |
Net cash provided by financing activities |
11,704 | 45,916 | 47,120 | 7,516 | |||||||||
| | | | | | | | | | | | | | |
Net increase (decrease) in cash and cash equivalents |
(1,367 | ) | 27,348 | 33,176 | (10,981 | ) | |||||||
Cash and cash equivalents at beginning of year |
3,690 | 2,323 | 2,323 | 29,671 | |||||||||
| | | | | | | | | | | | | | |
Cash and cash equivalents at end of year |
$ | 2,323 | $ | 29,671 | $ | 35,499 | $ | 18,690 | |||||
| | | | | | | | | | | | | | |
Supplemental cash flow information |
|||||||||||||
Accretion of redeemable convertible preferred stock to redemption value |
$ | 4,355 | $ | 4,437 | $ | 3,324 | $ | 3,301 | |||||
Cash paid for interest |
$ | 702 | $ | 945 | $ | 725 | $ | 560 | |||||
Warrants issued to lenders |
$ | 87 | $ | 128 | $ | 128 | $ | 119 | |||||
Purchases of property and equipment included in accounts payable |
$ | — | $ | 72 | $ | 144 | $ | 277 | |||||
Fair value of preferred stock warrants exercised and reclassified as shares of preferred stock |
$ | — | $ | 5,257 | $ | 4,137 | $ | 2,078 | |||||
| | | | | | | | | | | | | | |
See accompanying notes.
F-6
Quanterix Corporation
Notes to consolidated financial statements
(Information as of September 30, 2017 and for the nine months
ended September 30, 2016 and 2017 is unaudited)
1. Organization and operations
Quanterix Corporation (the Company) is a life sciences company that has developed a next generation, ultra-sensitive digital immunoassay platform that advances precision health for life sciences research and diagnostics. The Company's platform enables customers to reliably detect protein biomarkers in extremely low concentrations in blood, serum and other fluids that, in many cases, are undetectable using conventional, analog immunoassay technologies. It also allows researchers to define and validate the function of novel protein biomarkers that are only present in very low concentrations and have been discovered using technologies such as mass spectrometry. These capabilities provide the Company's customers with insight into the role of protein biomarkers in human health that has not been possible with other existing technologies and enable researchers to unlock unique insights into the continuum between health and disease. The Company is currently focusing its platform on protein detection and is also developing its Simoa technology to detect nucleic acids in biological samples.
The Company currently markets the Simoa HD-1 Analyzer, a fully automated immunoassay platform with multiplexing and custom assay capability, and related assay test kits and consumable materials. The Company also performs research services on behalf of customers to apply the Simoa technology to specific customer needs. The Company's primary customers are in the research use only market which includes academic and governmental research institutions, the research and development laboratories of pharmaceutical manufacturers, contract research organizations, and specialty research laboratories performing lab developed tests.
The Company has had recurring losses from operations since inception and has an accumulated deficit of $136.6 million at September 30, 2017 and the Company incurred a net loss of $15.9 million, $23.2 million and $19.6 million for the years ended December 31, 2015 and 2016 and the nine months ended September 30, 2017, respectively. The Company has funded its operations principally from issuances of preferred stock, debt financings, grants, product and service sales and development and license agreements. At September 30, 2017, the Company had $18.7 million of unrestricted cash and cash equivalents. The Company expects the current cash balance along with preferred stock proceeds received in the second quarter of 2017 and available capital under a debt facility will be sufficient to fund operations for a period of at least one year from the date the consolidated financial statements are issued. Prior to achieving profitability, the Company projects that it will need additional funding and intends to pursue an initial public offering of its common stock to fund future operations. However, if the Company is unable to complete a sufficient initial public offering in a timely manner it would need to pursue other financing alternatives, such as private financing of debt or equity or collaboration and license agreements. There can be no assurances, however, that additional funding will be available on terms acceptable to the Company, or at all.
2. Significant accounting policies
The following is a summary of significant accounting policies followed in the preparation of these financial statements.
F-7
Principles of consolidation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (U.S. GAAP) and include the accounts of Quanterix Corporation and its wholly-owned subsidiary. All material intercompany transactions and balances have been eliminated in consolidation.
Reclassifications
Certain amounts presented in the prior year financial statements have been reclassified to conform to current year presentation. Cost of revenue has been separated into cost of product revenue, cost of service and other revenue and cost of license revenue, related party, on the statement of operations and comprehensive loss. Also, in 2015, the Company had recorded $0.4 million in end of term fees on the Loan Agreement (Note 11) as other non-current liabilities. This amount has been reclassified to long-term debt, net of current portion, in the accompanying consolidated balance sheet as of December 31, 2015 to be consistent with the presentation as of December 31, 2016.
Use of estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the amounts reported in the consolidated financial statements and accompanying notes. In making those estimates and assumptions, the Company bases its estimates on historical experience and on various other assumptions believed to be reasonable. The Company's significant estimates included in the preparation of the consolidated financial statements are related to revenue recognition, fair value of equity instruments, valuation allowances recorded against deferred tax assets, and stock-based compensation. Actual results could differ from those estimates.
Unaudited interim financial information
The accompanying consolidated balance sheet as of September 30, 2017, the related consolidated statements of operations and comprehensive loss and consolidated statements of cash flows for the nine months ended September 30, 2016 and 2017 and the consolidated statement of redeemable convertible preferred stock, and stockholders' (deficit) equity for the nine months ended September 30, 2017 are unaudited. The interim unaudited consolidated financial statements have been prepared on the same basis as the annual audited consolidated financial statements and in the opinion of management, reflect all adjustments, which include only normal recurring adjustments necessary for the fair statement of the Company's financial position as of September 30, 2017 and the results of its operations and its cash flows for the nine months ended September 30, 2016 and 2017. The financial data and other information disclosed in these notes related to the nine months ended September 30, 2016 and 2017 are unaudited. The results for the nine months ended September 30, 2017 are not necessarily indicative of results to be expected for the year ending December 31, 2017, any other interim periods or any future year or period.
Revenue recognition
The Company recognizes revenue when (1) persuasive evidence of an arrangement exists, (2) shipment and installation, if applicable, has occurred or services have been rendered, (3) the price to the customer is fixed or determinable and (4) collection of the related receivable is reasonably assured. The Company primarily generates revenue from the sale of products and delivery of services, as well as under license and collaboration agreements. The Company's product revenue includes the sale of instruments as well as assay kits and consumables which are used to perform tests on the instrument. The Company's service revenue is generated from services performed in the Company's Simoa Accelerator Lab under contracts to perform research services on behalf of customers and maintenance and support services.
F-8
Product revenue
Revenue for instrument sales is recognized upon installation at the customer's location or upon transfer of title to the customer when installation is not required, which is generally the case with sales to distributors. In sales to end-customers, the Company provides the installation service and often payment is tied to the completion of the installation service. When installation is required, the Company accounts for the instrument and installation service as one unit of accounting and recognizes revenue when installation is completed, assuming all other revenue recognition criteria are met. Instrument transactions often have multiple elements, as discussed below. Included with the purchase of an instrument is a one-year assurance type product warranty assuring that the instrument is free of material defects and will function according to specifications. In addition, the sale of an instrument includes an implied warranty which is promised to the customer during the pre-sales process, at the time that the sales quote is issued to the customer. The imlied warranty is provided over the same one-year period as the standard warranty. The services included in the implied warranty are the same as those included in the extended service contracts, and include two bi-annual preventative maintenance service vists, minor hardware updates and software upgrades, additional training and troubleshooting which is beyond the scope of the standard product warranty. The implied warranty has been identified by the Company as a separate deliverable and unit of accounting. Consideration allocated to the implied one year service type warranty is recognized over the one year period of performance as service and other revenue as described below. Consideration allocated to any other elements is recognized as the goods are delivered or the services are performed.
Service and other revenue
Service revenue includes revenue from the implied one-year service type warranty obligation, revenue from extended service contracts, research services performed on behalf of a customer in the Company's Simoa Accelerator Lab, and other services that may be performed. Revenue for the implied one-year service type warranty is initially deferred at the time of instrument revenue recognition and is recognized ratably over a 12-month period starting on the date of instrument installation. Revenue for extended warranty contracts is recognized ratably over the service period. Revenue for research and development services and other services is generally recognized based on proportional performance of the contract, when the Company's ability to complete project requirements is reasonably assured. Most of these services are completed in a short period of time from the receipt of the customer's order. When significant risk exists in the Company's ability to fulfill project requirements, revenue is recognized upon completion of the contract.
Collaboration and license revenue
Collaboration and license revenue relates to the Joint Development and License Agreement (JLDA) with bioMérieux SA (bioMérieux) as amended and restated in December 2016 by the Amended and Restated License Agreement (the 2016 Amendment) and the agreements with a diagnostics company. Refer to Note 11 for a description of these arrangements and the Company's revenue recognition policies for these agreements.
Multiple element arrangements
Many of our instrument sales involve the delivery of multiple products and services. The elements of an instrument sale typically include the instrument installation (when required), an implied one year service type warranty, and in some cases the Company may also sell assays, consumables, or other services. Revenue recognition for contracts with multiple deliverables is based on the individual units of accounting determined to exist in the contract. A delivered item is considered a separate unit of accounting when the delivered item has value to the customer on a stand-alone basis. Items are considered to have stand-alone
F-9
value when they are sold separately by any vendor or when the customer could resell the item on a stand-alone basis.
The consideration received is allocated among the separate units of accounting using the relative selling price method, and the applicable revenue recognition criteria are applied to each of the separate units. The Company determines the estimated selling price for deliverables within the arrangement using vendor-specific objective evidence (VSOE) of selling price, if available. If VSOE is not available, the Company considers if third-party evidence is available. If third-party evidence of selling price or VSOE is not available, the Company uses its best estimate of selling price for the deliverable.
In order to establish VSOE of selling price, the Company must regularly sell the product or service on a standalone basis with a substantial majority priced within a relatively narrow range. If there are not a sufficient number of standalone sales such that VSOE of selling price cannot be determined, then the Company considers whether third party evidence can be used to establish selling price. Due to the lack of similar products and services sold by other companies within the industry, the Company has not established selling price using third-party evidence.
For product and service sales, the Company determines its best estimate of selling price for instruments, consumables, services and assays using average selling prices over a rolling 12-month period coupled with an assessment of market conditions, as VSOE and third-party evidence cannot be established. The Company recognizes revenue for delivered elements only when it determines there are no uncertainties regarding customer acceptance.
Distributor transactions
In certain markets, the Company sells products and provides services to customers through distributors that specialize in life sciences products. In cases where the product is delivered to a distributor, revenue recognition generally occurs when title transfers to the distributor. The terms of sales transactions through distributors are generally consistent with the terms of direct sales to customers, except the distributors do not require the Company's services to install the instrument at the end customer and perform the services for the customer that are beyond our standard warranty in the first year following the sale. These transactions are accounted for in accordance with the Company's revenue recognition policy described herein.
Cost of revenue
Cost of product revenue consists of raw materials, part costs and associated freight, shipping and handling costs, contract manufacturer costs, personnel costs, yield loss, in-license payments and royalties, stock-based compensation, other direct costs and overhead.
Cost of service and other revenue consists of personnel, facility costs associated with operating the Simoa Accelerator Lab on behalf of customers, costs related to instrument maintenance and servicing equipment at customer sites, other direct costs and overhead.
Cost of license revenue, related party consists of license fees that are the direct result of cash payments received related to license agreements.
Research and development expenses
Research and development expenses, including personnel costs, allocated facility costs, lab supplies, outside services, and contract laboratory costs are charged to research and development expense as incurred. The Company accounts for nonrefundable advance payments for goods and services that will be used in future research and development activities as expenses when the service has been performed or when the goods have been received rather than when the payment is made.
F-10
Selling, general, and administrative expenses
Selling, general, and administrative expenses are primarily composed of compensation and benefits associated with sales and marketing, finance, human resources, and other administrative personnel, outside marketing, advertising, allocated facilities costs, legal expenses, and other general and administrative costs.
Comprehensive loss
Comprehensive loss is defined as the change in equity of a business enterprise during a period from transactions and other events and circumstances from non-owner sources. For the years ended December 31, 2015 and 2016 and the nine months ended September 30, 2016 and 2017, comprehensive loss was equal to net loss.
Net loss per share
Basic net loss per common share attributable to common stockholders is calculated by dividing the net loss attributable to common stockholders by the weighted-average number of common shares outstanding during the period, without consideration for potentially dilutive securities. Diluted net loss per share is computed by dividing the net loss attributable to common stockholders by the weighted-average number of common shares and potentially dilutive securities outstanding for the period determined using the treasury-stock and if-converted methods. For purposes of the diluted net loss per share calculation, preferred stock, unvested restricted common stock and stock options are considered to be potentially dilutive securities, but are excluded from the calculation of diluted net loss per share because their effect would be anti-dilutive and therefore basic and diluted net loss per share were the same for all periods presented.
The following table sets forth the outstanding potentially dilutive securities that have been excluded in the calculation of diluted net loss per share because to do so would be anti-dilutive (in common stock equivalent shares):
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Year ended December 31, | Nine months ended September 30, |
|||||||||||
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Series A redeemable convertible preferred stock |
14,400,001 | 15,700,001 | 15,700,001 | 16,400,001 | |||||||||
Series B redeemable convertible preferred stock |
5,624,106 | 6,021,636 | 5,709,136 | 6,021,636 | |||||||||
Series C redeemable convertible preferred stock |
8,605,944 | 8,605,944 | 8,605,944 | 8,605,944 | |||||||||
Series D redeemable convertible preferred stock |
— | 12,420,262 | 12,420,262 | 14,534,164 | |||||||||
Unvested restricted common stock |
2,005,149 | 1,209,271 | 1,394,808 | 729,436 | |||||||||
Outstanding stock options |
3,352,547 | 3,598,918 | 3,955,074 | 7,364,345 | |||||||||
Outstanding preferred warrants |
2,153,662 | 1,048,983 | 661,484 | 387,811 | |||||||||
| | | | | | | | | | | | | | |
Total |
36,141,409 | 48,605,015 | 48,446,709 | 54,043,337 | |||||||||
| | | | | | | | | | | | | | |
As of December 31, 2015 and 2016 and September 30, 2016 and 2017, the Company had an obligation to issue warrants to purchase an additional 300,000 shares of Series A-3 Preferred Stock to a vendor if a contract is terminated prior to a minimum purchase commitment being met. No amounts are presented in
F-11
the table above for this obligation to issue a warrant as the issuance of the warrant is not considered probable.
The Company's redeemable convertible preferred stock is entitled to receive dividends based on dividends declared to common stockholders, thereby giving the preferred stockholders the right to participate in undistributed earnings of the Company above the stated dividend rate. However, preferred stockholders do not have a contractual obligation to share in the net losses of the Company. The Company operated in a net loss position for the years ended December 31, 2015 and 2016 and for the nine months ended September 30, 2016 and 2017, therefore the Company's accounting for basic and diluted earnings per share was unaffected by the participation rights of the preferred stockholders.
Unaudited pro forma information
The accompanying unaudited pro forma consolidated balance sheet as of September 30, 2017 has been prepared to give effect to the automatic conversion of all shares of preferred stock outstanding as of September 30, 2017 into 45,561,745 shares of common stock as if the proposed initial public offering had occurred on September 30, 2017. In addition, the pro forma balance sheet information assumes the reclassification of the preferred stock warrant liability to additional paid-in capital upon completion of an initial public offering, as the warrants to purchase convertible preferred stock will be converted into warrants to purchase common stock. The unaudited pro forma balance sheet information does not assume any proceeds from the proposed initial public offering or from the potential exercise of the outstanding warrants.
The unaudited pro forma net loss attributable to common stockholders used in the calculation of unaudited basic and diluted pro forma net loss per share attributable to common stockholders for the year ended December 31, 2016 and the nine months ended September 30, 2017 does not include the effects of the accretion of issuance costs, discounts, and accruing dividends on preferred stock because it assumes that the conversion of the preferred stock into common stock occurred on the later of the beginning of the period or the issuance date of the preferred stock. Also, the numerator in the pro forma basic and diluted net loss per common share calculation has been adjusted to remove gains or losses resulting from the remeasurement of the warrant liability for redeemable convertible preferred stock as it will be reclassified to additional paid-in capital upon the completion of an initial public offering. The unaudited pro forma net loss per common share does not include the shares to be sold and related proceeds to be received from the initial public offering or any proceeds from the exercise of warrants. The following table summarizes
F-12
the Company's unaudited pro forma net loss per share attributable to common stockholders (in thousands, except share and per share data):
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
| |
Year ended December 31, 2016 |
Nine months ended September 30, 2017 |
|||||
| | | | | | | | |
| |
(unaudited) |
||||||
Net loss attributable to common stockholders |
$ | (27,618 | ) | $ | (22,986 | ) | |
Add: |
|||||||
Changes in fair value of preferred stock warrant liability |
307 | (62 | ) | ||||
Accretion of preferred stock to redemption value |
4,437 | 3,301 | |||||
Accrued dividends on Series B preferred stock |
8 | 48 | |||||
| | | | | | | | |
Pro forma net loss attributable to common stockholders |
$ | (22,866 | ) | $ | (19,699 | ) | |
| | | | | | | | |
Weighted-average number of common shares outstanding, basic and diluted |
6,887,118 | 7,768,248 | |||||
Add: |
|||||||
Pro forma adjustments to reflect assumed conversion of preferred stock |
39,625,504 | 44,320,671 | |||||
| | | | | | | | |
Shares used to compute pro forma net loss per share attributable to common stockholders, basic and diluted |
46,512,622 | 52,088,919 | |||||
| | | | | | | | |
Pro forma basic and diluted net loss per share attributable to common stockholders |
$ | (0.49 | ) | $ | (0.38 | ) | |
| | | | | | | | |
Cash and cash equivalents
Cash and cash equivalents consists of cash deposits and short-term, highly liquid investments that are readily convertible into cash, with original maturities of three months or less. Cash equivalents are carried at fair value based on third-party pricing services. Cash and cash equivalents consist of the following (in thousands):
| | | | | | | | | | | |
| |
As of December 31, | As of September 30, 2017 |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| |
2015 |
2016 |
||||||||
| | | | | | | | | | | |
| |
|
|
(unaudited) |
|||||||
Cash and cash equivalents: |
||||||||||
Cash |
$ | 1,992 | $ | 29,671 | $ | 18,690 | ||||
Money market funds invested in U.S. Treasury obligations |
331 | — | — | |||||||
| | | | | | | | | | | |
Total cash and cash equivalents |
$ | 2,323 | $ | 29,671 | $ | 18,690 | ||||
| | | | | | | | | | | |
Restricted cash and deposits
As of December 31, 2015 and 2016 and September 30, 2017, the Company has $0.6 million, $0.4 million and $0.4 million, respectively, in restricted cash and deposits related to amounts held as a security deposit for the Company's facility lease obligation and a business registration application which are recorded in other non-current assets on the consolidated balance sheets.
Accounts receivable and allowance for doubtful accounts
The Company provides credit, in the normal course of business, to customers and does not require collateral. Accounts receivable consist of amounts due to the Company for sales to customers and are
F-13
recorded net of an allowance for doubtful accounts. The Company reviews accounts receivable on a regular basis to determine if any receivable will potentially be uncollectable and to estimate the amount of allowance for doubtful accounts necessary. Once a receivable is deemed uncollectible, such balance is written off and charged against the allowance for doubtful accounts. The Company has not incurred material write offs in any of the periods presented. As of December 31, 2015 and 2016 and September 30, 2017, no allowance for doubtful accounts has been recorded.
Inventory
Inventory is stated at the lower of cost or market on a first-in, first-out (FIFO) basis. The Company analyzes its inventory levels on each reporting date and writes down inventory that is expected to expire prior to being sold and inventory in excess of expected sales requirements. In the event that the Company identifies these conditions exist in its inventory, the carrying value is reduced to its estimated net realizable value.
Property and equipment
Property and equipment, including leasehold improvements, are stated at cost and are depreciated, or amortized in the case of leasehold improvements, over their estimated useful lives using the straight-line method. Expenditures for maintenance and repairs are charged to expense as incurred, whereas major betterments are capitalized as additions to property and equipment. The Company reviews its property and equipment whenever events or changes in circumstances indicate that the carrying value of certain assets might not be recoverable and recognizes an impairment loss when it is probable that an asset's realizable value is less than the carrying value. To date, no such impairment losses have been recorded. Depreciation is calculated based upon the following estimated useful lives of the assets:
| | | |
| Laboratory and manufacturing equipment | Five years | |
| Computers and software | Three years | |
| Office furniture and equipment | Seven years | |
| Leasehold improvements | Shorter of the useful life of the asset or the remaining term of the lease | |
| | | |
Software development costs
The Company develops and modifies software related to the operation of the instrument. Software development costs are expensed as incurred until the point the Company establishes technological feasibility. Based on the Company's product development process, technological feasibility is established upon the completion of a working model. The Company does not incur material costs between the completion of the working model and the point at which the product is ready for release. Therefore, software development costs are charged to the statement of operations as incurred as research and development expense.
Investments
During the third quarter of 2016, the Company purchased a minority interest in preferred stock in a privately held company for $0.3 million. The investment is recorded on a cost basis in other non-current assets on the accompanying consolidated balance sheets as the Company does not have a controlling investment, does not have the ability to exercise significant influence over the privately held company and the fair value of this equity investment is not readily determinable. The Company performs an impairment analysis at each reporting period to determine if the carrying value must be reduced due to a decrease in the value of the investment, which includes consideration of whether an event or change in circumstances has occurred that may have a significant adverse effect on the fair value of the investment. The Company
F-14
determined there was no impairment during the year ended December 31, 2016 and nine months ended September 30, 2017.
Deferred initial public offering costs
The Company capitalizes initial public offering costs, which primarily consist of direct, incremental legal and accounting fees relating to the Company's initial public offering within other non-current assets. The deferred public offering costs will be offset against proceeds from the initial public offering upon the consummation of the transaction. In the event the transaction is terminated or is significantly delayed, deferred initial public offerings costs will be expensed. No amounts were capitalized as of December 31, 2015 or 2016. At September 30, 2017, $2.0 million of offering costs were capitalized to other non-current assets in the consolidated financial statements.
Fair value of financial instruments
ASC Topic 820, Fair Value Measurement ("ASC 820"), establishes a fair value hierarchy for instruments measured at fair value that distinguishes between assumptions based on market data (observable inputs) and the Company's own assumptions (unobservable inputs). Observable inputs are inputs that market participants would use in pricing the asset or liability based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company's assumptions about the inputs that market participants would use in pricing the asset or liability, and are developed based on the best information available in the circumstances.
ASC 820 identifies fair value as the exchange price, or exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As a basis for considering market participant assumptions in fair value measurements, ASC 820 establishes a three-tier fair value hierarchy that distinguishes between the following:
Level 1 inputs are quoted prices (unadjusted) in active markets for identical assets or liabilities.
Level 2 inputs are inputs other than quoted prices that are observable for the asset or liability, either directly or indirectly; and
Level 3 inputs are unobservable inputs that reflect the Company's own assumptions about the assumptions market participants would use in pricing the asset or liability.
To the extent that the valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument's level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
The carrying amount reflected on the balance sheets for cash and cash equivalents, accounts receivable, prepaid expenses and other current assets, accounts payable and accrued liabilities approximated their fair values, due to the short-term nature of these instruments. The carrying value of the long-term debt approximates its fair value as the debt arrangement is based on interest rates the Company believes it could obtain for borrowings with similar terms. The Company has an investment in the preferred stock of a privately held company which is recorded within other non-current assets on a cost basis. This cost method investment's fair value has not been estimated as there are no identified events or changes in circumstances that would indicate a significant adverse effect on the fair value of the investment and to do so would be impractical.
F-15
Fair value measurements as of September 30, 2017 are as follows (in thousands):
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| Description |
Total |
Quoted prices in active markets (Level 1) |
Significant other observable inputs (Level 2) |
Significant unobservable inputs (Level 3) |
|||||||||
| | | | | | | | | | | | | | |
| |
(unaudited) |
||||||||||||
Financial liabilities |
|||||||||||||
Preferred stock warrant liability |
$ | 781 | $ | — | $ | — | $ | 781 | |||||
| | | | | | | | | | | | | | |
Total |
$ | 781 | $ | — | $ | — | $ | 781 | |||||
| | | | | | | | | | | | | | |
Fair value measurements as of December 31, 2016 are as follows (in thousands):
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| Description |
Total |
Quoted prices in active markets (Level 1) |
Significant other observable inputs (Level 2) |
Significant unobservable inputs (Level 3) |
|||||||||
| | | | | | | | | | | | | | |
Financial liabilities |
|||||||||||||
Preferred stock warrant liability |
$ | 2,802 | $ | — | $ | — | $ | 2,802 | |||||
| | | | | | | | | | | | | | |
Total |
$ | 2,802 | $ | — | $ | — | $ | 2,802 | |||||
| | | | | | | | | | | | | | |
Fair value measurements as of December 31, 2015 are as follows (in thousands):
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| Description |
Total |
Quoted prices in active markets (Level 1) |
Significant other observable inputs (Level 2) |
Significant unobservable inputs (Level 3) |
|||||||||
| | | | | | | | | | | | | | |
Financial assets |
|||||||||||||
Cash equivalents |
$ | 331 | $ | 331 | $ | — | $ | — | |||||
| | | | | | | | | | | | | | |
Total |
$ | 331 | $ | 331 | $ | — | $ | — | |||||
| | | | | | | | | | | | | | |
Financial liabilities |
|||||||||||||
Preferred stock warrant liability |
$ | 5,547 | $ | — | $ | — | $ | 5,547 | |||||
| | | | | | | | | | | | | | |
Total |
$ | 5,547 | $ | — | $ | — | $ | 5,547 | |||||
| | | | | | | | | | | | | | |
As of January 1, 2015, the Company had outstanding warrants to purchase 64,441 shares of Series A-2 redeemable convertible preferred stock (Series A-2 Preferred Stock), 1,300,000 shares of Series A-3 convertible preferred stock (Series A-3 Preferred Stock), 562,488 shares of Series B redeemable convertible preferred stock (Series B Preferred Stock), and 226,733 shares of Series C redeemable convertible preferred stock (Series C Preferred Stock). During the years ended December 31, 2015, and 2016 and the nine months ended September 30, 2017, the Company issued the following warrants:
- •
- On March 4, 2015, the Company issued a warrant to purchase 46,248 shares of Series C Preferred Stock to a lender related to an amendment to a debt facility (Note 10).
F-16
- •
- On January 29, 2016, the Company issued a warrant to purchase 57,810 shares of Series C Preferred Stock to a lender related to a
second amendment to a debt facility (Note 10)
- •
- On November 18, 2016, the Company issued a warrant to purchase 700,000 shares of Series A-3 Preferred Stock to a vendor
(Note 9).
- •
- On March 31, 2017, the Company issued a warrant to purchase 38,828 shares of Series D redeemable convertible preferred stock (Series D Preferred Stock) to a lender as part of a third amendment to a debt facility (Note 10).
All of the warrants were initially recorded as a preferred stock warrant liability on the accompanying consolidated balance sheets at fair value. Warrants issued for goods or services are initially accounted for under ASC 505-50 and are recognized over the required performance period in the consolidated statements of operations or consolidated balance sheets at the vesting date or reporting date fair value based on the nature of the underlying arrangement. Warrants issued in connection with a product development contract were recorded to research and development expense. Warrants issued in connection with a revenue arrangement were recorded as a reduction in revenue. Warrants issued in connection with debt arrangements were recorded as a reduction in the carrying value of debt. Once the counterparty's performance is complete and the warrants have become fully vested, they are marked to market on each reporting and exercise date with changes in the fair value recorded in other expense (income) on the statement of operations and comprehensive loss. Holders of warrants to purchase 1,300,000 shares of Series A-3 Preferred Stock and 562,488 Series B Preferred Stock exercised the warrants during the year ended December 31, 2016 and holders of warrants to purchase 700,000 shares of Series A-3 Preferred stock exercised the warrants during the six months ended June 30, 2017. Upon exercise, the fair value of the warrants was reclassified to redeemable convertible preferred stock along with any proceeds received.
The changes in preferred stock warrant liability measured at fair value for which the Company has used Level 3 inputs to determine fair value are as follows (in thousands):
| |
|
|||
|---|---|---|---|---|
| | | | | |
| |
Warrant liability |
|||
| | | | | |
Balance at January 1, 2015 |
$ | 4,862 | ||
Issuance of warrants related to debt facility |
87 | |||
Changes in fair value of warrants |
598 | |||
| | | | | |
Balance at December 31, 2015 |
5,547 | |||
Issuance of warrants related to debt facility |
128 | |||
Issuance of warrants related to a vendor |
2,078 | |||
Changes in fair value of warrants |
307 | |||
Warrant exercises |
(5,258 | ) | ||
| | | | | |
Balance at December 31, 2016 |
2,802 | |||
Issuance of warrants related to debt facility (unaudited) |
119 | |||
Changes in fair value of warrants (unaudited) |
(62 | ) | ||
Warrant exercises (unaudited) |
(2,078 | ) | ||
| | | | | |
Balance at September 30, 2017 (unaudited) |
$ | 781 | ||
| | | | | |
The warrants are classified as liabilities because they are exercisable into shares of redeemable convertible preferred stock. On each measurement date, the Company has utilized a black-scholes option pricing model to determine the fair value of the warrants and has utilized various valuation assumptions based on available market data and other relevant but unobservable factors. Expected volatility for the Company's redeemable convertible preferred stock was determined based on an analysis of the historical volatility of
F-17
a representative group of guideline public companies because there is currently no market for the Company's stock and, therefore, a lack of market-based company-specific historical and implied volatility information. The expected term reflects the remaining contractual term of the warrants. The assumed dividend yield is based upon the Company's expectation of not paying dividends in the foreseeable future. The risk-free rate is based upon the U.S. Treasury yield curve in effect at the valuation date, commensurate with the remaining contractual life of the warrants. The fair value of the underlying preferred shares was determined by management, with the assistance of a third party valuation specialist, using a hybrid valuation method, which includes a probability weighted analysis of two scenarios. The first scenario is based on the completion of an initial public offering utilizing a market approach and the second scenario is based on the Company remaining privately held utilizing either an income approach or a weighted-average of an income approach and a backsolve to a recent financing event, depending on the proximity of the financing event to the measurement date. The assumption regarding the Company's probability of completing an initial public offering is the primary contributing factor to the changes in fair value of the underlying preferred stock. See "Stock Based Compensation" section of this Note 2 for discussion on the changes of the probability of completing an initial public offering.
In order to determine the fair value of each warrant to purchase preferred stock at issuance at each reporting period, the following assumptions were utilized:
| |
|
|
|
|
|
|
|
|||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | | | | |
| Balance sheet date |
Value of underlying Series D preferred stock |
Value of underlying Series C preferred stock |
Value of underlying Series B preferred stock |
Value of underlying Series A-3 preferred stock |
Value of underlying Series A-2 preferred stock |
Volatility |
Probability of an initial public offering |
|||||||||||||||
| | | | | | | | | | | | | | | | | | | | | | | |
September 30, 2017 (unaudited) |
$ | 4.30 | $ | 4.28 | N/A | N/A | $ | 3.47 | 46% | 65% | ||||||||||||
December 31, 2016 |
N/A | $ | 4.16 | N/A | $ | 2.97 | $ | 2.95 | 52% | 40% | ||||||||||||
December 31, 2015 |
N/A | $ | 3.92 | $ | 3.00 | $ | 3.00 | $ | 1.90 | 41% | 25% | |||||||||||
| | | | | | | | | | | | | | | | | | | | | | | |
Warranties
The Company provides a one-year warranty and maintenance service related to its instruments and sells extended warranty contracts for additional periods. The Company defers revenue associated with these services and recognizes them on a pro-rata basis over the period of service. If expected costs are in excess of deferred revenue, a warranty accrual is recorded. As of December 31, 2015 and 2016 and September 30, 2017, no warranty accruals are recorded.
Income taxes
The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of events that have been recognized in the Company's consolidated financial statements or tax returns. Under this method, deferred tax assets and liabilities are determined based on differences between the consolidated financial statement carrying amounts and the tax bases of the assets and liabilities using the enacted tax rates in effect in the years in which the differences are expected to reverse. A valuation allowance against deferred tax assets is recorded if, based on the weight of the available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized.
The Company accounts for uncertain tax positions in accordance with the provisions of ASC 740 Income Taxes ("ASC 740"). When uncertain tax positions exist, the Company recognizes the tax benefit of tax positions to the extent that the benefit will more likely than not be realized. The determination as to whether the tax benefit will more likely than not be realized is based upon the technical merits of the tax position as well as consideration of the available facts and circumstances. As of December 31, 2015 and 2016, the Company does not have any significant uncertain tax positions.
F-18
Credit, product and supplier concentrations and off-balance-sheet risk
The Company has no significant off-balance-sheet risk, such as foreign exchange contracts, option contracts, or other hedging arrangements. Financial instruments that potentially expose the Company to concentrations of credit risk primarily consist of cash and cash equivalents and a cost method investment. The Company places its cash and cash equivalents principally in depository accounts with a bank.
The Company is also subject to supply chain risks related to the outsourcing of the manufacturing of its instruments. Although there are a limited number of manufacturers for instruments of this type, the Company believes that other suppliers could provide similar products on comparable terms. A change in suppliers, however, could cause a delay in manufacturing and a possible loss of sales, which would adversely affect operating results. In addition to outsourcing the manufacturing of its instruments, the Company also purchases antibodies through a number of different suppliers. Although a disruption in service from any one of its antibody suppliers is possible, the Company believes that it would be able to find an adequate supply from alternative suppliers.
Customers outside the United States represented 28%, 21% and 45% of the Company's gross trade accounts receivable balance as of December 31, 2015 and 2016 and September 30, 2017, respectively.
At December 31, 2016, one customer's accounts receivable balance was 26% of the Company's aggregate accounts receivable and represented 11% of the Company's revenue for the year ended December 31, 2016. At September 30, 2017, two customer's accounts receivable balances were a combined 29% of the Company's aggregate accounts receivable balance. During the year ended December 31, 2015 and nine month period ended September 30, 2017, no customer accounted for greater than 10% of revenue.
Segment information
Operating segments are defined as components of an enterprise about which separate discrete information is available for evaluation by the chief operating decision-maker in deciding how to allocate resources and assess performance. The Company and the Company's chief operating decision-maker reviews the Company's operations and manages its business as a single operating segment.
Net revenue by product and service line are as follows (in thousands):
| | | | | | | | | | | | | | |
| |
Year ended December 31, |
Nine months ended September 30, |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Product revenue |
|||||||||||||
Instrument |
$ | 6,542 | $ | 6,167 | $ | 4,236 | $ | 4,881 | |||||
Consumable and other product |
2,935 | 4,434 | 3,199 | 5,174 | |||||||||
| | | | | | | | | | | | | | |
Total |
$ | 9,477 | $ | 10,601 | $ | 7,435 | $ | 10,055 | |||||
| | | | | | | | | | | | | | |
Service and other revenue |
|||||||||||||
Simoa Accelerator Lab services |
$ | 1,625 | $ | 3,092 | $ | 1,990 | $ | 3,442 | |||||
Other services |
890 | 1,920 | 1,340 | 1,982 | |||||||||
| | | | | | | | | | | | | | |
Total |
$ | 2,515 | $ | 5,012 | $ | 3,330 | $ | 5,424 | |||||
| | | | | | | | | | | | | | |
The following table reflects total revenue (in thousands) by geography and as a percentage of total revenue, based on the billing address of our customers. North America consists of the United States,
F-19
Canada and Mexico; EMEA consists of Europe, Middle East, and Africa; and Asia Pacific includes Japan, China, South Korea, Singapore, Malaysia and Australia.
| |
|
|
|
|
|
|
|
|
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | | | | | | | | |
| |
Year ended December 31, | Nine months ended September 30, | ||||||||||||||||||
| |
2015 | 2016 | 2016 | 2017 | ||||||||||||||||
| |
$ |
% |
$ |
% |
$ |
% |
$ |
% |
||||||||||||
| | | | | | | | | | | | | | | | | | | | | |
| |
|
|
|
|
(unaudited) |
|||||||||||||||
North America |
$ | 9,417 | 77% | $ | 13,018 | 74% | $ | 8,032 | 74% | $ | 9,629 | 59% | ||||||||
EMEA |
$ | 2,081 | 17% | $ | 3,416 | 19% | $ | 2,021 | 18% | $ | 5,093 | 31% | ||||||||
Asia Pacific |
$ | 682 | 6% | $ | 1,151 | 7% | $ | 853 | 8% | $ | 1,563 | 10% | ||||||||
| | | | | | | | | | | | | | | | | | | | | |
Total |
$ | 12,180 | 100% | $ | 17,585 | 100% | $ | 10,906 | 100% | $ | 16,285 | 100% | ||||||||
| | | | | | | | | | | | | | | | | | | | | |
Stock-based compensation
The Company accounts for stock-based compensation awards in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification (ASC) Topic 718, Compensation—Stock Compensation, or ASC 718. ASC 718 requires all stock-based payments to employees, including grants of employee stock options, to be recognized in the statement of operations based on their fair values. Stock-based compensation awards have historically consisted of stock options and restricted stock.
Prior to adoption of ASU 2016-09 on January 1, 2017, the Company recognized compensation costs related to stock options granted to employees based on the estimated fair value of the awards on the date of grant, net of estimated forfeitures. Effective January 1, 2017, the Company ceased utilizing an estimated forfeiture rate and began recognizing forfeitures as they occur. The Company estimates the grant date fair value, and the resulting stock-based compensation expense, using the Black-Scholes option-pricing model. The grant date fair value of the stock-based awards is generally recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the respective awards.
The Company recognizes compensation costs related to share-based payments granted to non-employees based on the estimated fair value of the awards on the date of grant in the same manner as options for employees; however, the fair value of the stock options granted to non-employees is re-measured each reporting period until the service is complete, and the resulting increase or decrease in value, if any, is recognized as expense or income, respectively, during the period the related services are rendered to the same financial statement line item as any cash consideration would be recognized. There were no material non-employee awards outstanding during the years ended December 31, 2015 and 2016 or the nine months ended September 30, 2016 and 2017.
F-20
The fair value of stock options granted to employees and directors for their services on the Company's Board of Directors is estimated on the grant date using the Black-Scholes option-pricing model, based on the assumptions noted in the following table:
| | | | | | | | | | | | | | |
| |
Year ended December 31, |
Nine months ended September 30, |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Risk-free interest rate |
1.7% | 1.2% | 1.2% | 2.0% | |||||||||
Expected dividend yield |
None | None | None | None | |||||||||
Expected term (in years) |
6.0 | 6.0 | 6.0 | 6.0 | |||||||||
Expected volatility |
41.3% | 46.0% | 46% | 51.7% | |||||||||
| | | | | | | | | | | | | | |
Using the Black-Scholes option-pricing model, the weighted-average grant date fair value of options granted for the years ended December 31, 2015 and 2016 and the nine months ended September 30, 2016 and 2017, was $0.64, $0.75, $0.75 and $1.40 per share, respectively. Expected volatility was calculated based on reported volatility data for a representative group of guideline publicly traded companies for which historical information was available. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant, commensurate with the expected life assumption. The Company estimates the expected life of options granted to employees utilizing the simplified method which calculates the expected life of an option as the average of the time to vesting and contractual life of the options. The expected life is applied to the stock option grant group as a whole, as the Company does not expect substantially different exercise or post-vesting termination behavior among its employee population. The Company uses the simplified method due to the lack of historical exercise data and the plain nature of the stock options. The Company uses the remaining contractual term for the expected life of non-employee awards. The expected dividend yield is assumed to be zero as the Company has never paid dividends and has no current plans to pay any dividends on common stock. The fair value of the underlying common shares was determined by management, with the assistance of a third party valuation specialist, using a hybrid valuation method, which includes a weighted analysis of two scenarios. The first scenario is based on the completion of an initial public offering utilizing a market approach and the second scenario is based on the Company remaining privately held utilizing either an income approach or a weighted-average of an income approach and a backsolve to a recent financing event approach, depending on the proximity of the financing event to the measurement date. The initial public offering scenario reflected data gathered from relevant comparable initial public offering transactions and the current value method of equity allocation was used in determining the value of common stock. For the privately held scenario, traditional income methods of business valuation were employed, where the total equity value was then allocated using the option pricing model (OPM). The assumption regarding the Company's probability of completing an initial public offering is the primary contributing factor to the changes in fair value of the common stock. At December, 31, 2014 the valuation of the common stock assumed a probability of an initial public offering of 10%. The probability of initial public offering was 25%, 40%, and 65% at December 31, 2015, December 31, 2016 and September 30, 2017, respectively. Since December 31, 2015, the Company has performed the common stock valuations on a quarterly basis.
The probability of completing an initial public offering was based on the facts and circumstances as of each measurement date. During the three months ended December 31, 2016, the Company began initial preparations for completing an initial public offering; including assessing quarterly financial information and holding initial discussions with prospective investment bankers, which resulted in an increase in the
F-21
probability of completing an initial public offering. Subsequent to March 31, 2017, the Company obtained approval from the Board of Directors to pursue the transaction, selected investment bankers, held an organizational meeting, and performed other procedures necessary to complete an initial public offering. As a result, the probability of completing an initial public offering increased subsequent to March 31, 2017.
The Company is using the straight-line attribution method to recognize stock-based compensation expense for service based awards for employees and non-employees. However, cumulative compensation expense recognized through the end of any period must at least equal the value of vested awards through that period, with compensation expense adjusted accordingly. For the years ended December 31, 2015 and 2016, the amount of stock-based compensation expense recognized during a period is based on the value of the portion of the awards that are ultimately expected to vest. Prior to January 1, 2017, forfeitures are estimated at the time of grant, and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates. During the year ended December 31, 2015 and December 31, 2016, the Company applied an estimate of forfeitures which did not have a material effect on the consolidated financial statements. Effective January 1, 2017, the Company adopted Accounting Standards Update (ASU) 2016-09 Stock Compensation, and has elected to account for forfeitures as incurred and therefore no forfeiture estimate is utilized in the nine months ended September 30, 2017. The effect of this adoption has been recorded as a $0.1 million cumulative effect adjustment to accumulated deficit as of January 1, 2017.
The Company applies an accelerated attribution method to recognize stock-based compensation expense when accounting for performance-based stock awards. The Company records the expense for stock-based compensation awards subject to performance-based milestone vesting over the remaining service period when management determines that achievement of the milestone is probable. Management evaluates when the achievement of a performance-based milestone is probable based on the expected satisfaction of the performance conditions as of the reporting date. Compensation expense for performance-based stock awards is included in total stock-based compensation expense. There are no material performance-based stock awards outstanding as of December 31, 2015, 2016 and September 30, 2017.
Recent accounting pronouncements
The Company is considered to be an "emerging growth company" as defined in the Jumpstart Our Business Startups Act of 2012, as amended ("JOBS Act"). The JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. Thus, an emerging growth company can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected to avail ourselves of this extended transition period and, as a result, we will not be required to adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies.
In May 2014, the FASB issued ASU No. 2014-09, Revenue from Contracts with Customers (Topic 606) ("ASU 2014-09"). The FASB has issued several updates to the standard which i) clarify the application of the principal versus agent guidance; ii) clarify the guidance relating to performance obligations and licensing; iii) clarify assessment of the collectability criterion, presentation of sales taxes, measurement date for non-cash consideration and completed contracts at transaction; and iv) clarify narrow aspects of ASC 606 or corrects unintended application of the guidance (collectively, the "Revenue ASUs"). The Revenue ASUs provide an accounting standard for a single comprehensive model for use in accounting for revenue arising from contracts with customers and supersedes most current revenue recognition guidance. Under Topic 606, an entity recognizes revenue when its customer obtains control of promised goods or services, in an amount that reflects the consideration which the entity expects to receive in exchange for
F-22
those goods or services. To determine revenue recognition for arrangements that an entity determines are within the scope of Topic 606, the entity performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the entity satisfies a performance obligation. Topic 606 also impacts certain other areas, such as the accounting for costs to obtain or fulfill a contract. The standard also requires disclosure of the nature, amount, timing, and uncertainty of revenue and cash flows arising from contracts with customers.
The accounting standard is effective for the Company for the year ended December 31, 2019 and for interim periods within this year. Early adoption is permitted. The guidance permits two methods of adoption: retrospectively to each prior reporting period presented (the full retrospective method), or retrospectively with the cumulative effect of initially applying the guidance recognized at the date of initial application (the modified retrospective method). We do not currently intend to early adopt the provisions of this accounting standard and currently intend to adopt the standard effective January 1, 2019. The Company is in the process of determining which adoption method will be utilized. The Company is in the process of assessing the effect of this accounting standard with regards to the arrangements with bioMérieux and a diagnostic company (see Note 11 for the Company's revenue recognition under current guidance for these agreements). The Company's performance under the bioMérieux arrangement is not expected to be completed prior to the anticipated date of adoption on January 1, 2019, and the revenue recognition for this contract may be affected by Topic 606. The Company cannot predict at this time whether performance obligations under the arrangement with a diagnostic company will remain open at January 1, 2019. The Company is also assessing the other significant revenue streams, including instrument revenue, consumable revenue, research services revenue, and services contract revenue, to determine the effect of the adoption of this standard on those arrangements.
In July 2015, the FASB issued ASU No. 2015-11, Inventory (Topic 330): Simplifying the Measurement of Inventory ("ASU 2015-11"). ASU 2015-11 simplifies the subsequent measurement of inventory by requiring inventory to be measured at the lower of cost and net realizable value. Net realizable value is the estimated selling price in the ordinary course of business, less reasonably predictable costs of completion, disposal and transportation. ASU 2015-11, which is applied prospectively, is effective for the Company for the year ended December 31, 2017 and for interim periods beginning in the three months ended March 31, 2018 with early application permitted. The Company is currently evaluating the requirements of ASU 2015-11 and has not yet determined whether the adoption of the standard will have a material impact on its consolidated financial statements.
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842): Recognition and Measurement of Financial Assets and Financial Liabilities ("ASU 2016-02"). Under ASU 2016-02, lessees will be required to recognize a lease liability and a right-of-use asset for all leases (with the exception of short term leases) at the commencement date. Lessor accounting under ASU 2016-02 is largely unchanged. ASU 2016-02 is effective for the Company for the year ended December 31, 2020. Early adoption is permitted. Under ASU 2016-02, lessees (for capital and operating leases) and lessors (for sales-type, direct financing, and operating leases) must apply a modified retrospective transition approach for leases existing at, or entered into after, the beginning of the earliest comparative period presented in the financial statements. Lessees and lessors may not apply a full retrospective transition approach. The Company is currently evaluating the requirements of ASU 2016-02 and has not yet determined whether the adoption of the standard will have a material impact on its consolidated financial statements.
In March 2016, the FASB issued ASU No. 2016-09, Compensation—Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting ("ASU 2016-09"). ASU 2016-09 simplifies the
F-23
accounting for share-based payment award transactions including the financial statement presentation of excess tax benefits and deficiencies, classification of awards as either equity or liabilities, accounting for forfeitures and classification on the statement of cash flows. The Company early adopted ASU 2016-09 on January 1, 2017 and elected to account for forfeitures as they occur. The effect of this change in accounting policy has been recorded as a $0.1 million cumulative effect adjustment to accumulated deficit, as of January 1, 2017. ASU 2016-09 also provides that companies no longer record excess tax benefits or certain tax deficiencies in additional paid-in capital. Instead, all excess tax benefits and tax deficiencies are recorded as income tax expense or benefit in the statement of operations and comprehensive loss. There was no financial statement impact of adopting this provision of ASU 2016-09 as the Company is currently in a net operating loss position and the excess tax benefits that existed from options previously exercised had a full valuation allowance. The effects of adopting the remaining provisions in ASU 2016-09 affecting the classification of awards as either equity or liabilities when an entity partially settles the award in cash in excess of the employer's minimum statutory withholding requirements and classification in the statement of cash flows did not have a significant impact on the Company's financial position, results of operations or cash flows.
In January 2016, the FASB issued ASU No. 2016-01, Financial Instruments—Overall (Subtopic 825-10): Recognition and Measurement of Financial Assets and Financial Liabilities ("ASU 2016-01"). This guidance changes how entities measure equity investments that do not result in consolidation and are not accounted for under the equity method. Entities will be required to measure these investments at fair value at the end of each reporting period and recognize changes in fair value in net income. A practicability exception will be available for equity investments that do not have readily determinable fair values, however; the exception requires the Company to consider relevant transactions that can be reasonably known to identify any observable price changes that would impact the fair value. This guidance also changes certain disclosure requirements and other aspects of current U.S. GAAP. This guidance is effective for the Company for the year ended December 31, 2019 and for interim periods effective the three months ended March 31, 2020. Early adoption is permitted. The Company is currently evaluating the requirements of ASU 2016-01 and has not yet determined whether the adoption of the standard will have a material impact on its consolidated financial statements.
In August 2014, the FASB issued ASU No. 2014-15, Disclosure of Uncertainties about an Entity's Ability to Continue as a Going Concern ("ASU 2014-15"). ASU 2014-15 requires management to evaluate, at each annual or interim reporting period, whether there are conditions or events that exist that raise substantial doubt about an entity's ability to continue as a going concern within one year after the date the financial statements are issued and provide related disclosures. ASU 2014-15 is effective for the Company for the year ended December 31, 2016 and interim periods thereafter. The adoption of ASU 2014-15 did not have a material effect on the Company's consolidated financial statements but the standard requires enhanced disclosures in certain circumstances based on the Company's assessment of whether any such conditions or events exist that raise substantial doubt regarding the Company's ability to continue as a going concern within the one-year period.
In August 2016, the FASB issued ASU No. 2016-15, Statement of Cash Flow (Topic 230) ("ASU 2016-15"). The guidance reduces diversity in how certain cash receipts and cash payments are presented and classified in the Statements of Cash Flows. Certain requirements of ASU 2016-15 are as follows: i) cash payments for debt prepayment or debt extinguishment costs should be classified as cash outflows for financing activities, ii) contingent consideration payments made soon after a business combination should be classified as cash outflows for investing activities and cash payment made thereafter should be classified as cash outflows for financing up to the amount of the contingent consideration liability
F-24
recognized at the acquisition date with any excess classified as operating activities, iii) cash proceeds from the settlement of insurance claims should be classified on the basis of the nature of the loss, iv) cash proceeds from the settlement of Corporate-Owned Life Insurance (COLI) Policies should be classified as cash inflows from investing activities and cash payments for premiums on COLI policies may be classified as cash outflows for investing activities, operating activities, or a combination of investing and operating activities, and v) cash paid to a tax authority by an employer when withholding shares from an employee's award for tax-withholding purposes should be classified as cash outflows for financing activities. The guidance is effective for the Company for the year ended December 31, 2019 and for interim periods for the three months ended March 31, 2020. Early adoption is permitted. The adoption of ASU 2016-15 is not expected to have a material effect on the Company's consolidated financial statements.
In November 2016, the FASB issued ASU No. 2016-18, Statement of Cash Flows (Topic 230), Restricted Cash ("ASU 2016-18"). The amendments of ASU 2016-18 were issued to address the diversity in classification and presentation of changes in restricted cash and restricted cash equivalents on the statement of cash flows which is currently not addressed under Topic 230. The ASU would require an entity to include amounts generally described as restricted cash and restricted cash equivalents with cash and cash equivalents when reconciling the beginning of period and end of period total amounts on the statement of cash flows. The ASU is effective for annual reporting periods beginning after December 15, 2018, and interim periods within those annual periods. Early adoption is permitted and the adoption of the ASU should be applied retrospectively. The Company does not believe the accounting standard will have a material effect on the consolidated financial statements upon adoption, but would affect the presentation of restricted cash in the statement of cash flows. The amount of restricted cash held as of December 31, 2016 and September 30, 2017 is $50 thousand.
There have been no other changes in accounting standards issued by the FASB which have not yet been adopted that are expected to have a material impact on the Company's financial position, results of operations or cash flows.
3. Inventory
Inventory consists of the following (in thousands):
| |
|
|
|
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | |
| |
As of December 31, |
|
||||||||
| |
As of September 30, 2017 |
|||||||||
| |
2015 |
2016 |
||||||||
| | | | | | | | | | | |
| |
|
|
(unaudited) |
|||||||
Raw materials |
$ | 330 | $ | 563 | $ | 1,141 | ||||
Work in process |
198 | 304 | 402 | |||||||
Finished goods |
474 | 661 | 1,020 | |||||||
| | | | | | | | | | | |
Total |
$ | 1,002 | $ | 1,528 | $ | 2,563 | ||||
| | | | | | | | | | | |
Inventory comprises commercial instruments, assays, and the materials required to manufacture assays.
F-25
4. Property and equipment
Property and equipment consists of the following (in thousands):
| |
|
|
|
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | |
| |
As of December 31, |
|
||||||||
| |
As of September 30, 2017 |
|||||||||
| |
2015 |
2016 |
||||||||
| | | | | | | | | | | |
| |
|
|
(unaudited) |
|||||||
Laboratory and manufacturing equipment |
$ | 1,680 | $ | 1,937 | $ | 2,853 | ||||
Office furniture and equipment |
563 | 657 | 689 | |||||||
Computers and software |
283 | 451 | 451 | |||||||
Leasehold improvements |
133 | 133 | 180 | |||||||
| | | | | | | | | | | |
|
2,659 | 3,178 | 4,173 | |||||||
Less: accumulated depreciation |
(1,579 | ) | (1,955 | ) | (2,291 | ) | ||||
| | | | | | | | | | | |
Property and equipment, net |
$ | 1,080 | $ | 1,223 | $ | 1,882 | ||||
| | | | | | | | | | | |
The Company incurred depreciation expense of $0.4 million, $0.4 million, $0.3 million and $0.3 million for the years ended December 31, 2015 and 2016, and for the nine months ended September 30, 2016 and 2017, respectively.
5. Other accrued expenses
Other accrued expenses consists of the following (in thousands):
| |
|
|
|
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | |
| |
As of December 31, |
|
||||||||
| |
As of September 30, 2017 |
|||||||||
| |
2015 |
2016 |
||||||||
| | | | | | | | | | | |
| |
|
|
(unaudited) |
|||||||
Accrued inventory |
$ | 184 | $ | 70 | $ | 516 | ||||
Accrued royalties |
154 | 544 | 194 | |||||||
Accrued professional services |
372 | 396 | 469 | |||||||
Accrued development costs |
218 | 843 | 1,090 | |||||||
Accrued other |
416 | 533 | 1,090 | |||||||
| | | | | | | | | | | |
Total accrued expenses |
$ | 1,344 | $ | 2,386 | $ | 3,359 | ||||
| | | | | | | | | | | |
F-26
6. Income taxes
A reconciliation of the expected income tax provision computed using the federal statutory income tax rate to the Company's effective income tax rate is as follows:
| |
|
|
||
|---|---|---|---|---|
| | | | | |
| |
Year ended December 31, 2015 |
Year ended December 31, 2016 |
||
| | | | | |
Tax due at statutory rates |
34.0% | 34.0% | ||
State taxes, net of federal benefit |
4.7% | 4.3% | ||
Permanent differences |
(2.7)% | (0.8)% | ||
Tax credits |
2.1% | 3.0% | ||
Change in valuation allowance |
(38.3)% | (42.4)% | ||
Other, net |
0.2% | 1.9% | ||
| | | | | |
|
0.0% | 0.0% | ||
| | | | | |
No provision for income taxes has been recorded as the Company has incurred losses since inception. As of December 31, 2016, the Company had federal and state net operating loss (NOL) carryforwards of approximately $87.9 million and $67.0 million, respectively, which may be used to offset future taxable income. The Company also had federal and state credits of $2.4 million and $0.6 million, respectively, to offset future tax liabilities as of December 31, 2016. The NOL and tax credit carryforwards will expire at various dates through 2035, and are subject to review and possible adjustment by federal and state tax authorities. The Internal Revenue Code of 1986, as amended (the Code) contains provisions that may limit the NOL and tax credit carryforwards available to be used in any given year in the event of certain changes in the ownership interests of significant stockholders under Section 382 and 383 of the Code. The Company has not determined whether such a change has occurred.
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amount of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. Significant components of the Company's net deferred income taxes are as follows (in thousands):
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
| |
Year ended December 31, 2015 |
Year ended December 31, 2016 |
|||||
| | | | | | | | |
Deferred tax assets: |
|||||||
Net operating loss carryforwards |
$ | 25,529 | $ | 33,461 | |||
Research and development credit carryforwards |
2,119 | 2,801 | |||||
Deferred revenue |
606 | 1,443 | |||||
Depreciation and amortization |
866 | 879 | |||||
Stock compensation |
65 | 73 | |||||
Other deferred tax assets |
189 | 206 | |||||
| | | | | | | | |
Total deferred tax assets |
29,374 | 38,863 | |||||
Less valuation allowance |
(28,641 | ) | (38,457 | ) | |||
| | | | | | | | |
Net deferred tax assets |
733 | 406 | |||||
Deferred tax liabilities—stock compensation |
(733 | ) | (406 | ) | |||
| | | | | | | | |
Net deferred tax assets |
$ | — | $ | — | |||
| | | | | | | | |
F-27
A full valuation allowance is required to reduce the deferred tax assets reported if, based on the weight of the evidence, it is more likely than not that some portion or all of the deferred tax assets will not be realized. After consideration of all the evidence, both positive and negative, management has determined that a full valuation allowance is necessary to reduce the deferred tax assets to the amount that will more likely than not be realized. The change in the valuation allowance for the years ended December 31, 2015 and 2016 is $6.1 million and $9.8 million, respectively, primarily related to operating losses generated during the year for which the Company did not provide tax benefit.
At December 31, 2015 and 2016, the Company had no unrecognized tax benefits. The Company has not, as yet, conducted a study of its research and development credit carryforwards. This study may result in an adjustment to the Company's research and development credit carryforwards; however, until a study is completed and any adjustment is known, no amounts are being presented as an uncertain tax position. A full valuation allowance has been provided against the Company's research and development credits and, if an adjustment is required, this adjustment would be offset by an adjustment to the valuation allowance. Thus, there would be no impact to the consolidated balance sheets or consolidated statements of operations and comprehensive loss if an adjustment were required.
Interest and penalty charges, if any, related to unrecognized tax benefits would be classified as income tax expense in the accompanying consolidated statements of operations and comprehensive loss. Since the Company is in a loss carryforward position, the Company is generally subject to examination by the U.S. federal, state, and local income tax authorities for all tax years in which a loss carryforward is available. There are no current examinations pending.
7. Redeemable convertible preferred stock
As of September 30, 2017, the Company had authorized 47,015,449 shares of preferred stock, $0.001 par value per share, of which 3,972,415 shares are designated Series A-1 redeemable convertible preferred stock (Series A-1 Preferred Stock), 10,492,027 shares are designated Series A-2 Preferred Stock, 2,000,000 shares are designated Series A-3 Preferred Stock, 6,186,594 shares are designated Series B Preferred Stock, 9,247,089 shares are designated as Series C Preferred Stock, 544,332 shares are designated Series C-1 redeemable convertible preferred stock (Series C-1 Preferred Stock), 12,459,090 shares are designated Series D Preferred Stock and 2,113,902 are designated Series D-1 redeemable convertible preferred stock (Series D-1 Preferred Stock).
In February 2016, the Company issued 1,300,000 shares of Series A-3 Preferred Stock to a vendor (Note 9) upon the exercise of Series A-3 Preferred Stock warrants at a purchase price of $0.001 per share. The fair value of the settled warrant was $3.9 million at the time of exercise which was reclassified from Preferred Stock Warrant Liability to Series A Preferred Stock.
In March 2016, the Company issued 12,420,262 shares of Series D Preferred Stock at a purchase price of $3.67 per share. The issuance resulted in cash proceeds of $45.4 million, net of issuance costs.
In June and July 2016, the Company issued 397,530 shares of Series B Preferred Stock upon exercise of Series B Preferred Stock warrants, which included 312,500 shares of Series B Preferred Stock at a purchase price of $0.001 per share, 8,330 shares of Series B Preferred Stock at purchase price of $2.00 per share, and 76,700 shares of Series B Preferred Stock upon a cashless exercise of a warrant. The fair value of the settled warrants was $1.4 million at the time of exercise which was reclassified from Preferred Stock Warrant Liability to Series B Preferred Stock.
F-28
In January 2017, the Company issued 700,000 shares of Series A-3 Preferred Stock to a vendor (Note 9) upon the exercise of Series A-3 Preferred Stock warrants at a purchase price of $0.001 per share. The fair value of the settled warrant was $2.1 million at the time of exercise which was reclassified from Preferred Stock Warrant Liability to Series A Preferred Stock.
In June 2017, the Company issued 2,113,902 shares of Series D-1 Preferred Stock at a purchase price of $4.021 per share. The issuance resulted in cash proceeds of $8.4 million, net of issuance costs.
The Company had a Stock Purchase Agreement (SPA) with bioMérieux, a related party, which required the Company to issue additional shares of Series C Preferred Stock if certain milestones were met in exchange for $10.0 million in gross proceeds. The milestones were related to activities under a Joint Development and License Agreement (JDLA) (Note 11). bioMérieux also purchased Series C Preferred Stock when the JDLA was entered into in 2012. When the SPA was entered into, the Company evaluated whether the requirement to issue additional shares ('Tranche Feature") required separate accounting. The Company determined that the Tranche Feature was not legally detachable and therefore was an embedded feature in the Series C Preferred Stock that bioMérieux purchased.
During the year ended December 31, 2015, the Company amended the terms of the SPA which restructured the equity milestone from one payment of $10.0 million to three separate payments ($5.0 million; $3.0 million and $2.0 million) based on components of the initial technical milestones. No other terms of the Series C Preferred Stock changed. The Company achieved the first milestone in January 2015 at which time bioMérieux purchased 1,501,546 shares of Series C Preferred Stock at a price of $3.3299 for total proceeds of $5.0 million. The Company also achieved the second milestone in May 2015 at which time bioMérieux purchased 600,618 shares of Series C Preferred Stock at a price of $3.3299 for total proceeds of $2.0 million. In December 2016, the Company further amended the JDLA and SPA which cancelled the third and final milestone (Note 11).
The rights, preferences, and privileges of Series A-1, A-2, A-3, B, C, C-1, D, and D-1 Preferred Stock are as follows:
Conversion
Shares of Series A-1, A-2, A-3, B, C, C-1, D, and D-1 Preferred Stock are convertible into common stock on a one-for-one basis, adjustable for certain dilutive events. Conversion is at the option of the preferred stockholders, although conversion is automatic upon the earlier of the consummation of an initial public offering, resulting in gross proceeds to the Company of at least $40.0 million and for a minimum per-share amount of $5.00 per share, or the approval of a Preferred Majority, defined as 60% of the outstanding shares of Series A-1, A-2, B, C, D, and D-1 Preferred Stock voting as a single class.
Dividends
Holders of the Series A-1, A-2, B, C, and C-1 Preferred Stock are entitled to receive, before any cash is paid out or set aside for any common stock, cumulative dividends in arrears at the annual rate of $0.08, $0.08, $0.16, $0.2664, and $0.2664 per share, respectively, subject to adjustment for stock splits, stock dividends, combinations and reorganizations. Holders of Series D, and D-1 Preferred Stock are entitled to receive non-cumulative dividends at the rate of $0.2936, and $0.3217 per share, respectively, subject to adjustment for stock splits, when and if declared by the Board of Directors of the Company. The cumulative accrued dividends as of September 30, 2017, are $3.3 million, $7.7 million, $5.6 million, $9.1 million, and $0.6 million for Series A-1, A-2, B, C, and C-1 Preferred Stock, respectively. Holders of Series A-3 Preferred Stock are not entitled to receive any preferred stock dividends. Upon full payment of preferred dividends,
F-29
additional dividends shall be shared among all preferred stock holders and common stock holders on a pro rata basis.
Liquidation preference
Holders of the Series A-1, A-2, A-3, B, C, C-1, D, and D-1 Preferred Stock have preference in the event of a liquidation or dissolution of the Company equal to $1.0416667, $1.0416667, $2.00, $2.00, $3.3299, $3.3299, $3.67, and $4.021 per share, respectively, plus any accrued but unpaid dividends. In any liquidation event, Series D and D-1 Preferred Stock holders will receive first priority in liquidation payments. In the event that the amounts available for distribution are insufficient to pay the full amounts, the assets shall be distributed ratably among Series D, and D-1 Preferred Stock holders in proportion to their aggregate liquidation preference amounts until such amounts are paid full. Series B, C, and C-1 Preferred Stock holders will receive next priority in liquidation payments after Series D Preferred Stock holders. In the event that the amounts available for distribution are insufficient to pay the full amounts, the assets shall be distributed ratably among B, C, and C-1 Preferred Stock holders in proportion to their aggregate liquidation preference amounts. Series A-1, A-2, and A-3 Preferred Stock holders will receive next priority in liquidation preference after Series B, C, and C-1 Preferred Stock holders. In the event that the amounts available for distribution after payment are insufficient to pay the full amounts, the assets shall be distributed ratably among A-1, A-2, and A-3 Preferred Stock holders in proportion to their aggregate liquidation preference amounts. Any remaining amounts are distributed to holders of common stock on a pro rata basis. However, if the holders of any series of preferred stock would receive a greater liquidation preference if they were converted into shares of common stock immediately prior to the liquidation event, then these shares will receive consideration equal to the amount that would be received if the shares had been converted in common stock in lieu of the applicable liquidation preference.
Voting rights
Holders of the Series A-1, A-2, A-3, B, C, D, and D-1 Preferred Stock (Voting Preferred) are entitled to vote as a single class with the holders of common stock, and shall have one vote for each equivalent common share into which the preferred stock is convertible. A Preferred Majority vote is required in order to amend the Certificate of Incorporation or By-Laws, reclassify common stock or establish another class of stock, create or authorize additional shares of preferred stock, effect a sale, liquidation, or merger of the Company, repurchase or redeem any capital stock, or engage in any action which would adversely affect the holders of the preferred stock.
The holders of the Series A-1, A-2, and B Preferred Stock can elect three members to the Board of Directors, voting as a single class. The holders of the Series C Preferred Stock can elect one member to the Board of Directors. The holders of the Voting Preferred can elect one member to the Board of Directors, voting as a single class.
Holders of Series C-1 Preferred Stock have no voting rights.
Redemption rights
Prior to the issuance of Series D Preferred Stock in March 2016, a majority vote of the Series B, C, and C-1 Preferred Stock holders could elect to redeem all of the outstanding shares of Series B, C, and C-1 Preferred Stock at any time on or after November 14, 2016. The Series A-1 and A-2 Preferred Stockholders had the right to elect to redeem all of the outstanding shares at any time after the redemption of the Series B, C, and C-1 Preferred Stock shares is made. The preferred stockholders were entitled to the redemption in three equal annual installments.
F-30
Upon issuance of the Series D Preferred Stock in March 2016, the redemption rights were adjusted. A majority vote of the Series D Preferred Stockholders could elect to redeem all of the outstanding shares of Series D on or after March 18, 2019. Upon issuance of the Series D-1 Preferred Stock in June 2017 the redemption rights were adjusted. A majority vote of the Series D and D-1 Preferred Stockholders, voting as separate classes, could elect to redeem all of the outstanding shares of Series D and D-1 on or after June 2, 2020. Holders of the Series C-1, C, and B Preferred Stock can only redeem their shares following the redemption in full of all shares of Series D and D-1 Preferred Stock and upon a Preferred Majority Vote. Holders of the Series A-1 and A-2 Preferred Stock can only redeem their shares following the redemption in full of the Series D-1, D, C-1, C and B Preferred Stock, and upon a Preferred Majority Vote. Series A-3 Preferred Stock does not have redemption rights other than in certain deemed liquidation scenarios.
The redemption value of the Series A-1, A-2, B, C, and C-1 Preferred Stock is equal to the original issuance price of the preferred stock plus any accrued or declared but unpaid cumulative dividends. The redemption price of the Series D, and D-1 Preferred Stock is the greater of i) the fair market value of the common stock which it is convertible into or ii) the original issuance price plus all declared but unpaid dividends, which are non-cumulative. As of December 31, 2016, the fair market value of the Company's common stock was less than the original issuance price of the Series D Preferred Stock.
The redemption amount by class of Preferred Stock as of September 30, 2017 is as follows (in thousands):
| |
|
|||
|---|---|---|---|---|
| | | | | |
| Preferred Stock |
Redemption amount at September 30, 2017 |
|||
| | | | | |
A-1 |
$ | 7,416 | ||
A-2 |
$ | 18,544 | ||
A-3 |
$ | 4,000 | ||
B |
$ | 17,614 | ||
C |
$ | 35,959 | ||
C-1 |
$ | 2,441 | ||
D |
$ | 45,582 | ||
D-1 |
$ | 8,500 | ||
| | | | | |
Total |
$ | 140,056 | ||
| | | | | |
The redemption values listed above reflect the values as of September 30, 2017. The redemption amount may increase between September 30, 2017 and the first redemption date of June 2, 2020 if additional dividends are accrued or declared and if the value of the common stock at that time exceeds the issue price of the series D and Series D-1 Preferred Stock or could decrease if any dividends previously accrued are paid prior to the redemption date. Upon a redemption event, the redemption amount is payable over three equal annual installments. The table below reflects the potential cash payment that would be
F-31
required to be paid by the Company in the event of a redemption election using the redemption amounts as of September 30, 2017 (in thousands):
| | | | | |
Year ending December 31: |
||||
2017 |
$ | — | ||
2018 |
$ | — | ||
2019 |
$ | — | ||
2020 |
$ | 46,685 | ||
2021 |
$ | 46,685 | ||
2022 |
$ | 46,686 | ||
| | | | | |
Preferred stock is presented in mezzanine equity. The Series A-1, A-2, B, C, C-1 D, and D-1 Preferred Stock are redeemable at the option of the holder at a fixed date and therefore the Company is accreting the preferred stock to its redemption value through the earliest possible redemption date for all issuances where the carrying value is less than the redemption value. The Series A-3 Preferred Stock is redeemable only upon certain deemed liquidation scenarios which are outside of the Company's control. The accretion includes the accretion of issuance costs and cumulative preferred stock dividends. Series A-3 preferred stock is not entitled to dividends. The Company assessed all terms and features of the preferred stock in order to identify any potential embedded features that would require bifurcation or any beneficial conversion features. As part of this analysis, the Company assessed the economic characteristics and risks of its preferred stock, including conversion and liquidation features, as well as dividend and voting rights. The Company determined that all features of the preferred stock are clearly and closely associated with an equity host, and although the preferred stock includes conversion features, such conversion features do not require bifurcation as a derivative liability. On the date of issuance, the fair value of common stock into which the Series A-1, A-2, A-3, B, C, C-1, D, and D-1 Preferred Stock was convertible was less than the effective conversion price of the Series A-1, A-2, A-3, B, C, C-1, D, and D-1 Preferred Stock and as such, there was no intrinsic value of the conversion option at the commitment date.
8. Common stock, restricted stock, stock options and warrants
Common stock reserved
The Company has reserved the following shares of common stock, on a common stock equivalent basis, for the potential conversion of outstanding preferred stock, the exercise of warrants and common stock options and vesting of restricted common stock:
| |
|
|
|
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | |
| |
Year ended December 31, | Nine months ended September 30, 2017 |
||||||||
| |
2015 |
2016 |
||||||||
| | | | | | | | | | | |
| |
|
|
(unaudited) |
|||||||
Series A Preferred Stock |
14,400,001 | 15,700,001 | 16,400,001 | |||||||
Series B Preferred Stock |
5,624,106 | 6,021,636 | 6,021,636 | |||||||
Series C Preferred Stock |
8,605,944 | 8,605,944 | 8,605,944 | |||||||
Series D Preferred Stock |
— | 12,420,262 | 14,534,164 | |||||||
Preferred stock warrants |
2,153,662 | 1,048,983 | 387,811 | |||||||
Common stock options and unvested restricted common stock |
5,357,696 | 4,808,189 | 8,093,781 | |||||||
| | | | | | | | | | | |
|
36,141,409 | 48,605,015 | 54,043,337 | |||||||
| | | | | | | | | | | |
F-32
Warrants
The following tables summarize the Company's outstanding warrants as of December 31, 2015, 2016 and September 30, 2017:
As of December 31, 2015:
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
| Series |
Issued and exercisable |
Exercise price |
|||||
| | | | | | | | |
Series A-3 preferred stock |
1,300,000 | $ | 0.0010 | ||||
Series A-2 preferred stock |
64,441 | $ | 1.0417 | ||||
Series B preferred stock |
312,500 | $ | 0.0010 | ||||
Series B preferred stock |
249,988 | $ | 2.0000 | ||||
Series C preferred stock |
111,114 | $ | 3.3299 | ||||
Series D preferred stock |
115,619 | $ | 3.3299 | ||||
| | | | | | | | |
|
2,153,662 | ||||||
| | | | | | | | |
As of December 31, 2016:
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
| Series |
Issued and exercisable |
Exercise price |
|||||
| | | | | | | | |
Series A-3 preferred stock |
700,000 | $ | 0.0010 | ||||
Series A-2 preferred stock |
64,441 | $ | 1.0417 | ||||
Series C preferred stock |
111,114 | $ | 3.3299 | ||||
Series C preferred stock |
173,428 | $ | 3.3299 | ||||
| | | | | | | | |
|
1,048,983 | ||||||
| | | | | | | | |
As of September 30, 2017:
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
| |
Issued and exercisable |
Exercise price |
|||||
| | | | | | | | |
| |
(unaudited) |
||||||
Series A-2 preferred stock |
64,441 | $ | 1.0417 | ||||
Series C preferred stock |
284,542 | $ | 3.3299 | ||||
Series D preferred stock |
38,828 | $ | 3.6700 | ||||
| | | | | | | | |
|
387,811 | ||||||
| | | | | | | | |
The Company has an agreement with a vendor (Note 9) where the Company could be obligated to issue warrants to purchase an additional 300,000 shares of Series A-3 Preferred Stock to the vendor if the contract with the vendor is terminated prior to a minimum purchase commitment being met. No shares have been reserved related to these potential obligations to issue warrants in the future.
F-33
Stock options and restricted stock
Share-based compensation expense for all stock awards consists of the following:
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Year ended December 31, | Nine months ended September 30, |
|||||||||||
| |
2015 |
2016 |
2016 |
2017 |
|||||||||
| | | | | | | | | | | | | | |
| |
|
|
(unaudited) |
||||||||||
Cost of product revenue |
$ | 6 | $ | 6 | $ | 6 | $ | 18 | |||||
Cost of service and other revenue |
1 | 12 | 10 | 31 | |||||||||
Research and development |
112 | 59 | 51 | 126 | |||||||||
General and administrative |
985 | 851 | 670 | 1,256 | |||||||||
| | | | | | | | | | | | | | |
Total |
$ | 1,104 | $ | 928 | $ | 737 | $ | 1,431 | |||||
| | | | | | | | | | | | | | |
In June 2007, the Company adopted the 2007 Stock Option and Grant Plan (the Plan), under which it may grant incentive stock options, non-qualified stock options, restricted stock, and stock grants. At December 31, 2016 the Plan allowed for the issuance of up to 10,381,013 shares of common stock. During the three months ended March 31, 2017, the Plan was amended to allow for the issuance of an additional 2,000,000 shares of common stock for total issuance of up to 12,381,013 shares of common stock at June 30, 2017. During the three months ended September 30, 2017 the plan was further amended to allow for the issuance of an additional 1,600,000 shares of common stock for total issuance of upto 13,981,013 shares of common stock at September 30, 2017. As of September 30, 2017, 1,340,645 shares were available for future grant under the Plan.
Under the Plan, stock options may not be granted with exercise prices of less than fair market value on the date of the grant. Options generally vest ratably over a four-year period with 25% vesting on the first
F-34
anniversary and the remaining 75% vesting ratably on a monthly basis over the remaining three years. These options expire ten years after the grant date. Activity under the Plan is as follows:
| |
|
|
|
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | | | | | | | | | | | | | |
| |
Options |
Weighted- average exercise price |
Remaining contractual life (in years) |
Aggregate intrinsic value (in thousands) |
|||||||||
| | | | | | | | | | | | | | |
Outstanding at December 31, 2014 |
2,957,086 | $ | 0.72 | 7.5 | $ | 752 | |||||||
Granted |
606,658 | $ | 0.97 | ||||||||||
Exercised |
(113,621 | ) | $ | 0.38 | |||||||||
Cancelled or forfeited |
(97,576 | ) | $ | 0.82 | |||||||||
| | | | | | | | | | | | | | |
Outstanding at December 31, 2015 |
3,352,547 | $ | 0.77 | 7.1 | $ | 2,313 | |||||||
Granted |
885,500 | $ | 1.61 | ||||||||||
Exercised |
(292,112 | ) | $ | 0.73 | |||||||||
Cancelled or forfeited |
(347,017 | ) | $ | 1.29 | |||||||||
| | | | | | | | | | | | | | |
Outstanding at December 31, 2016 |
3,598,918 | $ | 0.93 | 6.8 | $ | 5,796 | |||||||
Granted (unaudited) |
3,972,769 | $ | 2.61 | ||||||||||
Exercised (unaudited) |
(99,672 | ) | $ | 0.73 | |||||||||
Cancelled or forfeited (unaudited) |
(107,670 | ) | $ | 1.60 | |||||||||
| | | | | | | | | | | | | | |
Outstanding at September 30, 2017 (unaudited) |
7,364,345 | $ | 1.83 | 7.9 | $ | 10,012 | |||||||
| | | | | | | | | | | | | | |
Vested and expected to vest at December 31, 2016 |
3,598,918 | $ | 0.93 | 6.8 | $ | 5,796 | |||||||
Exercisable at December 31, 2016 |
2,328,480 | $ | 0.72 | 5.6 | $ | 4,228 | |||||||
Vested and expected to vest at September 30, 2017 (unaudited) |
7,364,345 | $ | 1.83 | 7.9 | $ | 10,012 | |||||||
Exercisable at September 30, 2017 (unaudited) |
2,718,288 | $ | 0.83 | 5.4 | $ | 6,365 | |||||||
| | | | | | | | | | | | | | |
Using the Black-Scholes option pricing model, the weighted-average fair value of options granted to employees and directors during the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017 was $0.64, $0.75, $0.75 and $1.40 per share, respectively. The expense related to awards granted to employees was $0.3 million, $0.2 million, $0.2 million and $0.9 million for the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017, respectively. The intrinsic value of stock options exercised was $0.1 million, $0.4 million, $0.1 million and $0.2 million for the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017, respectively. Activity related to non-employee awards was not material to the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017.
At September 30, 2017, there was $5.4 million of total unrecognized compensation cost related to unvested stock options, which is expected to be recognized over the remaining weighted-average vesting period of 3.3 years.
In December 2014, the Company issued 253,623 shares of restricted common stock to a director of the Company under the Plan. Under the terms of the agreement, shares of common stock issued are subject to a four year vesting schedule. Vesting occurs periodically at specified time intervals and specified percentages. In January 2015, the Company issued 2,510,330 shares of restricted common stock to an executive of the Company under the Plan. The majority of these shares were issued subject to a four year
F-35
vesting schedule with 25% vesting on the first anniversary and the remaining vesting 75% ratably on a monthly basis over the remaining three years, while another portion was issued subject to performance based vesting. The vesting of performance based awards is dependent upon achievement of specified financial targets of the Company. The majority of the performance criteria were achieved during the years ended December 31, 2015 and 2016 and the remaining unvested awards with performance conditions are not material. No restricted stock awards were granted during the year ended December 31, 2016 or the nine months ended September 30, 2017. A summary of restricted stock activity is as follows:
| |
|
|
|||||
|---|---|---|---|---|---|---|---|
| | | | | | | | |
| |
Shares |
Weighted- average grant date fair value per share |
|||||
| | | | | | | | |
Unvested restricted common stock as of December 31, 2014 |
253,623 | $ | 0.92 | ||||
Granted |
2,510,330 | $ | 0.97 | ||||
Vested |
(758,804 | ) | $ | 0.97 | |||
| | | | | | | | |
Unvested restricted common stock as of December 31, 2015 |
2,005,149 | $ | 0.97 | ||||
Vested |
(795,878 | ) | $ | 0.96 | |||
| | | | | | | | |
Unvested restricted common stock as of December 31, 2016 |
1,209,271 | $ | 0.97 | ||||
Vested (unaudited) |
(479,835 | ) | $ | 0.97 | |||
| | | | | | | | |
Unvested restricted common stock as of September 30, 2017 (unaudited) |
729,436 | $ | 0.97 | ||||
| | | | | | | | |
The expense related to awards granted to employees and directors was $0.8 million, $0.7 million, $0.6 million, and $0.5 million for the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017, respectively.
At September 30, 2017, there was $0.6 million of total unrecognized compensation cost related to unvested restricted stock, which is expected to be recognized over the remaining weighted-average vesting period of 0.9 years.
The aggregate fair value of restricted stock awards that vested during the years ended December 31, 2015 and 2016 and the nine months ended September 30, 2017, based on estimated fair values of the stock underlying the restricted stock awards on the day of vesting, was $1.1 million, $1.1 million, and $1.4 million, respectively.
9. Commitments and contingencies
Tufts University
In June 2007, the Company entered into a license agreement (the License Agreement) for certain intellectual property with Tufts University (Tufts). Tufts is a related party to the Company due to Tuft's equity ownership in the Company and because a board member of the Company's Board of Directors is affiliated with Tufts. The License Agreement, which was subsequently amended, is exclusive and sub licensable, and will continue in effect on a country by country basis as long as there is a valid claim of a licensed patent in a country. The Company is committed to pay license and maintenance fees, prior to commercialization, in addition to low single digit royalties on direct sales and services and a royalty on sublicense income. During the year ended December 31, 2016 the Company executed a license agreement
F-36
with a diagnostic company and also amended the bioMérieux agreement (Note 11). The Company accrued $0.4 million for license fees related to these arrangements during the year ended December 31, 2016 which were recorded to cost of collaboration and license revenue on the consolidated statements of operations and comprehensive loss. During the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017 the Company recorded royalty expense of $0.2 million, $0.3 million, $0.2 million and $0.3 million, respectively, in cost of product revenue on the consolidated statements of operations and comprehensive loss.
Other licenses
During the year ended December 31, 2012, the Company entered into a license agreement for certain intellectual property with a third party. The non-exclusive, non-sublicenseable third party's license provides the Company access to certain patents specifically for protein detection, and shall be in effect until the expiration of the last licensed patent. In consideration for these rights, the Company committed to certain license fees, milestone payments, minimum annual royalties and a mid-single digit royalty. The license also extends to bioMérieux as a partner of the Company, in addition to containing restrictions on a change of control of the Company and identification of excluded parties, without the third party's prior consent. The Company is required to make mid-single digit royalty payments on net sales of products and services which utilize the licensed technology. The Company must pay the greater of calculated royalties on net sales or an annual minimum royalty of $50 thousand. During the year ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017, the Company recorded royalty expense of $0.2 million, $0.3 million, $0.3 million and $0.2 million, respectively, in cost of product revenue on the consolidated statements of operations.
During the year ended December 31, 2014, the Company entered into a lease agreement for the Company's current corporate headquarters with a lease term that expires in June 2020 which can be extended to June 2023. The lease agreement contains a period of free rent and annual increases to rental amounts. Rent expense is recognized straight-line over the course of the lease term. As of December 31, 2016, $0.2 million of deferred rent expense was recorded in other non-current liabilities. As of September 30, 2017, $0.2 million of deferred rent expense was recorded in other non-current liabilities, and less than $0.1 million was recorded in other accrued expenses.
As of December 31, 2016, the minimum future rent payments under the lease agreement and amendment are as follows (in thousands):
| | | | | |
Years ending December 31: |
||||
2017 |
$ | 1,124 | ||
2018 |
1,155 | |||
2019 |
1,196 | |||
2020 |
605 | |||
| | | | | |
|
$ | 4,080 | ||
| | | | | |
The Company recorded $0.9 million, $1.1 million, $0.8 million and $0.8 million in rent expense for the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017, respectively.
F-37
Development and supply agreement
On August 15, 2011, the Company signed a Development Services and Equity Participation Agreement (Development Agreement) with a strategic manufacturing partner, STRATEC Biomedical Systems (STRATEC Biomedical), for the development/customization and manufacture of an instrument based on STRATEC technology using the Company's single-molecule assay (Simoa) technology. Under the terms of the Development Agreement, the Company was originally required to pay a fee of $1.5 million (the Development Fee) and issue warrants (the Development Warrants) for the purchase of 2,000,000 shares of Series A-3 Preferred Stock at an exercise price of $0.001 per share. The Development Warrants become exercisable upon the achievement of certain developmental milestones.
The Company also had entered into a supply agreement with STRATEC Biomedical which requires the Company to purchase a minimum number of commercial units over a seven-year period ending in May 2021. If the Company were to fail to purchase a required number of commercial units, the Company would be obligated to pay termination costs and in addition a fee based on the shortfall of commercial units purchased compared to the required minimum amount. Based on the number of commercial instruments purchased as of December 31, 2016, assuming no additional commercial units were purchased, this fee would equal $12.9 million. The amount the Company could be obligated to pay under the minimum purchase commitment is reduced as each commercial unit is purchased. Also, if the Company terminates the Supply Agreement under certain circumstances and has not purchased a required number of commercial units, it would be obligated to issue warrants to purchase 300,000 shares of Series A-3 Preferred Stock (the Supply Warrants) at $0.001 per share. The Company believes that it will purchase sufficient units to meet the requirements of the minimum purchase commitment and, therefore, has not accrued for any of the potential cash consideration. The Supply Warrants are accounted for at fair value; however, the fair value of the Supply Warrants as of December 31, 2015 and 2016 and September 30, 2017 was insignificant as there was a low probability of the warrants being issued.
During the year ended December 31, 2016 the Development Agreement was amended (the Amendment) to modify the deliverables related to the final milestone, to agree on instrument design changes to be implemented, and to reduce the minimum purchase commitment. Prior to the Amendment, the Company had paid $0.9 million of the $1.5 million Development Fee and issued 1.3 million of the 2.0 million Development Warrants to purchase shares of Series A-3 Preferred Stock, which were exercised during the year ended December 31, 2016. The final milestone in the Development Agreement included the release of an in vitro diagnostic (IVD) instrument for manufacturing and was determined to not be probable of completion and, as a result, no expense had been recorded related to this milestone in the year ended December 31, 2016 or prior years.
Upon signing the Amendment, the Company agreed to issue the remaining 700,000 Development Warrants immediately, in consideration for reducing the required number of commercial units to be purchased and certain development activites, and those warrants were fully vested upon issuance. The reduction in the minimum purchase commitment did not affect the fee that would be payable based on the units purchased as of December 31, 2016 assuming no additional units were purchased. The Company recognized a total of $2.1 million in research and development expense for the year ended December 31, 2016 for the issuance of the additional Development Warrants representing the fair value of the 700,000 warrants at the time of issuance. These Development Warrants were exercised during the three months ended March 31, 2017.
Additionally, the parties agreed on additional development services for an additional fee, which is payable when the additional development is completed. The fee includes the final Development Fee of $0.6 million that was due under the terms of the Development Agreement. These amounts are being recorded to
F-38
research and development expense and accrued expenses as the services are performed. The services are expected to be completed during the year ended December 31, 2018. Substantive efforts related to these additional development activities started in the first quarter of 2017.
The Company is subject to claims in the ordinary course of business, however, the Company is not currently a party to any pending or threatened litigation, the outcome of which would be expected to have a material adverse effect on its financial condition or the results of its operations. The Company accrues for contingent liabilities to the extent that the liability is probable and estimable.
10. Notes payable
Loan agreement
On April 14, 2014, the Company executed a Loan Agreement with a lender. The Loan Agreement provided a total debt facility of $10.0 million which is secured by substantially all of the assets of the Company. At closing, the Company borrowed $5.0 million in principal and had the ability to draw the additional $5.0 million over the period from November 1, 2014 to March 31, 2015. The interest rate on this term loan is variable based on a calculation of the prime rate less 5.25% with a minimum interest rate of 8%. Interest is paid monthly beginning the month following the borrowing date. Principal payments were scheduled to begin on September 1, 2015, unless the Company achieved certain milestones which would extend this date to either December 1, 2015 or March 1, 2016. The Loan Agreement also contains prepayment penalties and an end of term charge. The end of term charge of $0.2 million is being accreted over the life of the loan.
In connection with the Loan Agreement, the Company granted the lender warrants to purchase shares of either Series C Preferred Stock or shares of preferred stock in the next financing round. The number of warrants eligible to be issued increases as the Company draws on the facility. Therefore, additional warrants will be issued if the Company draws on any of the remaining debt facility. The warrants issued in connection with the initial borrowing were initially recorded at fair value of $0.1 million as a preferred stock warrant liability in the accompanying consolidated balance sheets and a corresponding debt discount was recorded.
The Company also incurred debt issuance costs of $0.1 million. As a result of the debt discounts recorded related to the warrants and the debt issuance costs, the debt was initially recorded at less than its face value. The debt, including the end of term charge, is being accreted over the life of the loan using the effective interest method.
The Loan Agreement also provided the lender with a right to invest up to $1.0 million or, subject to Company approval and consent, to convert up to $1.0 million of outstanding principal into shares of preferred stock in the next financing round at the same price as all other investors. The lender invested $1.0 million in March 2016 as part of the Series D Preferred Stock financing.
Amendment 1 to loan agreement
On March 4, 2015 the Company executed Amendment 1 to the Loan Agreement (Amendment 1) and borrowed the remaining $5.0 million that was available under the loan facility. The terms of Amendment 1 allowed the Company to defer the commencement of principal payments to December 1, 2015 and extended the loan maturity date to February 1, 2018. If the Company obtained at least $10.0 million in equity financing before December 1, 2015, the commencement of principal payments could be further
F-39
deferred until March 1, 2016 and the loan maturity date could be extended to May 1, 2018. As the financing milestone was not achieved, the Company made the first principal payment of $0.3 million on December 1, 2015 and the loan maturity date was February 1, 2018 under Amendment 1.
The additional $5.0 million borrowed included an additional $0.2 million end of term charge. The end of term charge on this borrowing is being accreted over the life of the loan as additional interest expense. The additional borrowing also resulted in the issuance of additional warrants with a grant date fair value of $0.1 million. The fair value of the additional warrants were initially recorded at fair value as a preferred stock warrant liability in the accompanying consolidated balance sheets and a corresponding debt discount was recorded. The debt, including the end of term charge, is being accreted over the remaining life of the loan using the effective interest method.
Amendment 2 to loan agreement
In January 2016, the Company executed Amendment 2 to the Loan Agreement (Amendment 2). Amendment 2 increased the total facility available by $5.0 million to a total of $15.0 million and further delayed the commencement of principal payments to July 1, 2016. Under Amendment 2, following the Series D Preferred Stock financing (Note 6), the Company could have elected to further delay the commencement of principal payments until January 1, 2017, however the Company voluntarily began paying principal on July 1, 2016. Upon signing Amendment 2, the Company drew an additional $3.0 million under the debt facility. The remaining $2.0 million available under the facility expired unexercised in April 2016, which reduced the amounts available under the facility to $13.0 million.
The additional $3.0 million borrowed included an additional $0.1 million end of term charge. The end of term charge on this borrowing is being accreted over the life of the loan. The additional borrowing also resulted in the issuance of additional warrants with a grant date fair value of $0.1 million. The fair value of the additional warrants were initially recorded at fair value as a preferred stock warrant liability in the accompanying consolidated balance sheets and a corresponding debt discount was recorded. The debt, including the end of term charge, is being accreted to over the remaining life of the loan using the effective interest method.
Amendment 3 to loan agreement
In March 2017, the Company signed Amendment 3 to the Loan Agreement (Amendment 3). Amendment 3 increased the total facility available by $5.0 million to a total of $18.0 million. Additionally, the lender may provide an additional optional term loan, solely at the lender's discretion, for an incremental $5.0 million, increasing the total potential facility to $23.0 million. As of September 30, 2017 we have not drawn any of this additional facility. The terms of Amendment 3 allowed the Company to defer the commencement of principal payments to March 1, 2018 and extended the loan maturity date to March 1, 2019. If the Company obtains certain a specified revenue target of $15.0 million in revenue over a trailing 9 month period commencing in April 2017, the commencement of principal payments could be further deferred until September 3, 2018. Amendment 3 did not change the due date of the existing end of term fees of $0.5 million which remain due on February 1, 2018. Upon signing Amendment 3, the Company did not draw any of the additional amounts available under the amended debt facility and no amounts have been subsequently drawn under the facility. The Company has until March 31, 2018 to draw the additional amounts unless the aforementioned revenue target is met, in which case the Company has until September 3, 2018.
In connection with Amendment 3, the Company issued the lender additional warrants with a grant date fair value of $0.1 million. The fair value of the additional warrants were initially recorded at fair value as a
F-40
preferred stock warrant liability in the accompanying consolidated balance sheets and a corresponding debt discount was recorded. The debt is being accreted to its face value over the remaining life of the loan using the effective interest method.
Debt payment obligations and end of term fees due based on principal payments commencing in March 1, 2018, are as follows (in thousands):
| | | | | |
Years ending December 31: |
||||
2017* |
$ | 921 | ||
2018 |
5,133 | |||
2019 |
4,430 | |||
| | | | | |
|
10,484 | |||
| | | | | |
* reflects principal payments made in Q1 2017 prior to the execution of Amendment 3.
Non-cash interest expense related to debt discount amortization and accretion of end of term fees was $0.3 million, $0.4 million, $0.3 million and $0.2 million for the year ended December 31, 2015 and 2016 and the nine months ended September 30, 2016 and 2017, respectively.
The Company assessed all terms and features of the Loan Agreement and the subsequent amendments in order to identify any potential embedded features that would require bifurcation. As part of this analysis, the Company assessed the economic characteristics and risks of the debt. The Company determined that all features of the Loan Agreement and the subsequent amendments are either clearly and closely associated with a debt host or have a de minimis fair value and, as such, do not require separate accounting as a derivative liability. The Company assessed each amendment under ASC 470-50 and concluded that all of the amendments constituted modifications. In this analysis, consideration was given to the fact that Amendments 1 and 2 were executed within one year of each other. The Company also assessed whether the amendments represented a troubled debt restructuring and concluded they did not. The Company accounted for each of the amendments to the Loan Agreement as a modification of its debt and the unamortized discount and issuance costs related to the prior debt are amortized over the modified term of the new debt.
The Loan Agreement and the subsequent amendments contain negative covenants restricting the Company's activities, including limitations on dispositions, mergers or acquisitions, incurring indebtedness or liens, paying dividends or making investments and certain other business transactions. There are no financial covenants associated with the Loan Agreement and the subsequent amendments. The obligations under the Loan Agreement and subsequent amendments are subject to acceleration upon the occurrence of specified events of default, including a material adverse change in the Company's business, operations or financial or other condition. The Company has determined that the risk of subjective acceleration under the material adverse events clause is not probable and therefore has classified the outstanding principal in current and long-term liabilities based on scheduled principal payments.
11. Collaboration and license arrangements
Joint development and license agreement (JDLA)
In November 2012, the Company entered into the JDLA with bioMérieux, a related party. As discussed below, the JDLA has been subsequently amended. Under the terms the JDLA, the Company granted bioMérieux an exclusive, royalty-bearing license, without right to sublicense, to manufacture and sell instruments and assays using our Simoa technology exclusively for in vitro diagnoses used in clinical lab applications, food quality control testing, and pharma quality control testing, and co-exclusively in certain
F-41
related fields, as defined in the contract. As part of the JDLA, the Company was to also develop and manufacture instruments to bioMérieux's specifications for bioMérieux's use or for sale by bioMérieux. The Company retained rights to sell the instrument in the co-exclusive fields and any other fields not licensed exclusively to bioMérieux. bioMérieux was to develop and sell diagnostic assays to be used in conjunction with the Company's instruments.
Upon execution of the JDLA, the Company received $10.0 million in consideration and was entitled to receive two additional payments of $5.0 million each upon the achievement of certain developmental criteria. Neither of these criteria have been achieved. The Company was also entitled to receive royalty payments on the sale of assays and payments for the manufacture and delivery of instruments based on a contractual rate subject to future adjustments.
At the inception of the JDLA, the Company determined that the deliverables were as follows: 1) licenses to the Company's technology and trademarks, training, completion and delivery of a prototype instrument per contractual specifications ("License and Protoype"), 2) various activities to assist bioMérieux in the development of the initial assay and an instrument that is in vitro diagnostics ("IVD") compliant ("Initial Assay Assistance"), 3) various activities to assist bioMérieux in the development of a benchtop instrument ("Benchtop Assistance"), and 4) joint steering committee participation ("JSC"). Each of these deliverables were considered separate units of accounting, and the License and Prototype unit of accounting was determined to have standalone value as the License and Prototype unit of accounting could be utilized by bioMérieux without the related services included in the other units of accounting.
The Company allocated the allocable arrangement consideration based on the relative selling price of each unit of accounting. For all units of accounting, the Company determined the selling price using the best estimate of selling price, or BESP. Management's best estimate of the selling price of the License and Prototype unit of accounting was based on a discounted cash flow analysis to support the estimated selling price of the license. The Company determined the BESP of the other units of accounting based on internal estimates of the costs to perform the services, adjusted to reflect a reasonable profit margin as well as based on market prices for similar instruments and services.
Revenue related to the License and Prototype unit of accounting of $8.3 million was recognized in 2013 upon delivery of both the license which was delivered at inception and the first prototype instrument, which was required for bioMérieux to make use of the license. Prior to the effect of the 2016 Amendment described below, revenue for the other units of accounting were recognized over an estimated period of performance.
Amendments to the JDLA
In May 2014 and January 2015, the parties executed a First and Second Amendment to the JLDA, respectively. These amendments addressed revised timelines related to completing the development activities under the JDLA and enacted additional governance protocols to monitor those activities. These amendments did not change the deliverables under the JDLA or the total arrangement consideration. The Company revised its estimates of the remaining period of performance for the remaining undelivered units of accounting and these revisions did not have a material effect on revenue recognition.
F-42
On December 22, 2016, the Company entered into the 2016 Amendment which ended the ongoing joint development efforts between the parties, and modified the rights and obligations of both parties accordingly, as follows:
- •
- For a period of not more than three years from the date of the 2016 Amendment bioMérieux has the ability to evaluate
independently whether it will develop a new, smaller in vitro diagnostic instrument using the Simoa technology for use in clinical lab applications, food quality control testing, and pharmaceutical
quality control testing benchtop (the "Feasibility Period") and has the sole right to determine whether or not to develop such a new instrument during the Feasibility Period. If
bioMérieux does elect to pursue development of such a new instrument, they will have a set number of years to complete development within a specified period, which contains various
development milestones which must be accomplished.
- •
- bioMérieux received a license to the source and object code of the Company's Level 1 Data Reduction (L1DR) software. The
L1DR software the Company's proprietary image processing algorithms that convert images of microscopic beads associated with biomarker molecules in microwells. Also, the Company must provide to
bioMérieux access to any know how and intellectual property associated with the L1DR software, including any updates and upgrades to the L1DR software during the Feasibility Period. If
bioMéreiux exercises its right to develop an instrument independently, this right will continue throughout the development period to the end of the term of the agreement related to
independently developed instruments.
- •
- It was clarified that the Company can engage a collaboration partner (IVD Partner), subject to restrictions as to the particular parties with
which the Company could elect to partner and the assays that can be developed, in the field of in vitro diagnostics used in Clinical Lab Applications. The Company shall pay bioMérieux a
mid-double-digit percentage of royalties received from the IVD Partner based on assays sales by the IVD Partner.
- •
- bioMérieux's licenses include all patents and know-how owned or controlled by the Company related to the Company's Simoa
technology and upgrades thereto that are necessary for the development, manufacture, use or sale of instruments and assays or consumables on such instruments over the Feasibility Period. If
bioMérieux exercises its right to develop an instrument independently, this right will continue throughout the development period to the end of the term of the 2016 Amendment related to
independently developed instruments.
- •
- bioMérieux retains an option (the Option) to obtain worldwide distribution rights to the HD-1 floor standing instrument in the applicable fields. The Option is exercisable over a three year period and upon exercise, the Company and bioMérieux are required to negotiate, in good faith, a distribution agreement that would include a specified upfront payment.
The 2016 Amendment included a cash payment of $2.0 million from bioMérieux which was paid in January 2017.
Accounting assessment
Prior to the execution of the 2016 Amendment, the Company was recognizing revenue over the estimated period of performance of the ongoing units of accounting (Initial Assay Assistance, Benchtop Assistance, and JSC). As a result, the Company recognized $0.2 million and $0.2 million in revenue for the years ended December 31, 2015 and 2016, respectively. At the date of the execution of the 2016 Amendment, the Company had $1.2 million in deferred revenue related to the JDLA. Upon the execution of the 2016
F-43
Amendment all undelivered elements and contingent consideration of the JDLA were cancelled. The Company determined the 2016 Amendment should be accounted for as a modification to the JDLA and the balance of deferred revenue prior to the 2016 Amendment should be included as allocable consideration under the 2016 Amendment resulting in total allocable consideration of $3.2 million. The Company recorded an increase to deferred revenue upon receipt of the $2.0 million during the three months ended March 31, 2017.
The Company has determined that the deliverables included under the 2016 Amendment are rights to the L1DR software, training and rights to future technology improvements for L1DR Software, rights to all future technological improvements related to the Simoa technology, and participation on joint committees.
The Company determined that the L1DR and rights to unspecified technology improvements (the "L1DR Unit of Accounting") includes the sale of software and software related elements and therefore should be accounted for under ASC 985-605—Software Revenue Recognition. The Company cannot demonstrate Vendor Specific Objective Evidence (VSOE) of fair value for the ongoing obligation to provide unspecified technology improvements. Therefore, the deliverables in the L1DR Unit of Accounting cannot be separated. The Company has applied the combined service approach and the consideration allocated to this unit of accounting is being recognized ratably over the estimated period of performance, which has initially been determined to be estimated to be the three year Feasibility Period. This will be reevaluated each period to determine if there are any changes to the estimated period of performance.
The Company concluded that the rights to future technology improvements for the Simoa technology and the participation on joint committees represented a second unit of accounting (the "Instrument Know How Unit of Accounting"). The deliverables in the Instrument Know How Unit of Accounting are considered non-software deliverables that are subject to ASC 605-25 and will be delivered over time on a when and if available basis. Revenue is being recognized on a straight line basis over the estimated period of performance, which has initially been determined to be the three year Feasibility Period. This period will be reevaluated each period to determine if there are any changes to the period of performance.
The Option is considered substantive as the Company is at risk with regard to whether bioMérieux will exercise the Option. In addition, the Option exercise payment payable by bioMérieux upon exercise is not priced at a significant and incremental discount. Accordingly, the Option is not considered a deliverable at the inception of the arrangement and the associated Option exercise payment is not included in allocable arrangement consideration.
Revenue recognized for the year ended December 31, 2016 following the 2016 Amendment was not material. The Company recognized revenue of $0.8 million for the nine months ended September 30, 2017 as collaboration revenue and as of September 30, 2017, $2.4 million of arrangement consideration remains in deferred revenue.
Under the 2016 Agreement the Company is eligible to receive royalties on net sales of assays sold by bioMérieux in the mid to high single digits, and to receive low double digit royalties on sales of instruments by bioMérieux based on manufactured cost. No royalties have been recognized through September 30, 2017.
Evaluation and option agreements and license agreement
In 2015, the Company entered into three agreements, for three separate fields, with a diagnostic company for the evaluation of the Company's Simoa technology. These agreements each allowed for the option to negotiate a license agreement. In return, the Company received non-refundable payments totaling $2.0 million. In December 2016, the diagnostic company exercised one of its options and the parties
F-44
entered into a license agreement in one of the fields. This agreement has a one-time non-refundable license fee of $1.0 million and the right to receive running low single digit royalties on licensed products. The negotiation periods for the other two agreements were extended and the negotiations remain ongoing.
For each of the three fields, the right to evaluate the technology, negotiate a license to the technology, and the undelivered license to the technology represents a combined unit of accounting, and the licenses to each of the three fields each have standalone value. The Company has allocated the allocable arrangement consideration based on the relative selling price of each unit of accounting. The BESP of each of the three options was determined to be representative of the contractual amount paid for each option. The Company defers the amounts allocated to each of the three options until the corresponding license is delivered or, if no license agreement is executed and delivered, when the negotiations for each option terminates.
Upon execution of the license in one of the fields in December 2016, the $1.0 million license fee, in addition to the $0.8 million allocated to the option for this field, resulted in a total of $1.8 million of consideration being recognized as revenue as there were no remaining undelivered performance obligations. Because the negotiations remain ongoing with respect to the other two fields, the consideration allocated to these options of $1.2 million has been deferred and is recorded as deferred revenue as of December 31, 2016 and September 30, 2017.
12. Employee benefit-plans
The Company sponsors a 401(k) savings plan for our employees The Company may make discretionary contributions for each 401k Plan year. During the year years ended December 31, 2015 and 2016 and the nine months ended September 30, 2016 and 2017, the Company did not make contributions to the plan.
13. Related party transactions
As described in Notes 11 and 7, bioMérieux is a customer through its Joint Development and License Agreement and also a holder of the Company's Series C Preferred Stock. bioMérieux also has a seat on the Company's board of directors. The Company recognized revenue related to the JDLA with bioMérieux of $0.2 million, $0.2 million and $0.8 million in the years ended December 31, 2015 and 2016 and the nine months ended September 30, 2017, respectively, from bioMérieux. The Company also had deferred revenue of $1.6 million and $1.3 million, and $2.5 million at December 31, 2015 and 2016, and September 30, 2017, respectively. As described in Note 7, bioMérieux purchased shares of our Series C Preferred Stock totaling $7.0 million in the year ended December 31, 2015.
As described in Note 7, in March 2016, the Company issued an aggregate of 12,420,262 shares of Series D Preferred Stock for an aggregate purchase price of $45.6 million. Of the amount issued, $22.9 million was purchased by the Company's existing principal stockholders, officers and directors.
As described in Note 7, in June 2017, the Company issued an aggregate of 2,113,902 share of Series D-1 Preferred Stock for an aggregate purchase price of $8.5 million. Of the amount issued, $1.0 million was purchased by a director of the Company.
As described in Note 9, in June 2007, the Company entered into a license agreement (the License Agreement) for certain intellectual property with Tufts University (Tufts). Tufts is a related party to the Company due to Tuft's equity ownership in the Company and because a board member of the Company's Board of Directors is affiliated with Tufts. During the years ended December 31, 2015 and 2016, and the nine months ended September 30, 2016 and 2017 the Company recorded royalty expense of $0.2 million,
F-45
$0.3 million, $0.2 million and $0.3 million, respectively, in cost of product revenue on the consolidated statements of operations and comprehensive loss. During the year ended December 31, 2016, the Company recognized $0.4 million as cost of license revenue associated with a payment made to Tufts.
14. Subsequent events
The Company has evaluated, for potential recognition and disclosure, events that occurred prior to July 20, 2017, the date at which the consolidated financial statements were available to be issued and August 31, 2017, the date the revised financial statements were available to be issued. All material subsequent events are disclosed in the preceding notes and in the following paragraph.
- (a)
- On August 24, 2017, the Company increased the number of shares available under the Plan by an additional 1.6 million shares, increasing the total number of shares available under the Plan to 13,981,013 shares. The increase in the shares available under the Plan was approved by the Company's shareholders on August 31, 2017.
F-46
shares

Common stock
Prospectus
Joint book running managers
| J.P. Morgan | Leerink Partners | |||
Co-managers |
||||
| BTIG | Evercore ISI |
|||
, 2017
Until , 2017 (the 25th day after the date of this prospectus), all dealers that effect transactions in these securities, whether or not participating in this offering, may be required to deliver a prospectus. This requirement is in addition to the dealers' obligation to deliver a prospectus when acting as underwriters and with respect to their unsold allotments or subscriptions.
Part II
Information not required in prospectus
Item 13. Other expenses of issuance and distribution.
The following table sets forth all costs and expenses, other than underwriting discounts and commissions, paid or payable by the Registrant in connection with the sale of the common stock being registered. All amounts shown are estimates except for the SEC registration fee and the FINRA filing fee:
| |
|
|||
|---|---|---|---|---|
| | | | | |
| | | | | |
| |
Amount paid or to be paid |
|||
| | | | | |
SEC registration fee |
$ | 7,159 | ||
FINRA filing fee |
9,125 | |||
Initial Nasdaq Global Market listing fee |
125,000 | |||
Blue sky qualification fees and expenses |
||||
Printing and engraving expenses |
||||
Legal fees and expenses |
||||
Accounting fees and expenses |
||||
Transfer agent and registrar fees and expenses |
||||
Miscellaneous expenses |
||||
| | | | | |
Total |
$ | |||
| | | | | |
Item 14. Indemnification of directors and officers.
Section 145(a) of the Delaware General Corporation Law provides, in general, that a corporation may indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative (other than an action by or in the right of the corporation), because he or she is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against expenses (including attorneys' fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by the person in connection with such action, suit or proceeding, if he or she acted in good faith and in a manner he or she reasonably believed to be in or not opposed to the best interests of the corporation and, with respect to any criminal action or proceeding, had no reasonable cause to believe his or her conduct was unlawful.
Section 145(b) of the Delaware General Corporation Law provides, in general, that a corporation may indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action or suit by or in the right of the corporation to procure a judgment in its favor because the person is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against expenses (including attorneys' fees) actually and reasonably incurred by the person in connection with the defense or settlement of such action or suit if he or she acted in good faith and in a manner he or she reasonably believed to be in or not opposed to the best interests of the corporation, except that no indemnification shall be made with respect to any claim, issue or matter as to which he or she shall have been adjudged to be liable to the corporation unless and only to the extent that the Court of Chancery or other adjudicating court determines that,
II-1
despite the adjudication of liability but in view of all of the circumstances of the case, he or she is fairly and reasonably entitled to indemnity for such expenses that the Court of Chancery or other adjudicating court shall deem proper.
Section 145(g) of the Delaware General Corporation Law provides, in general, that a corporation may purchase and maintain insurance on behalf of any person who is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against any liability asserted against such person and incurred by such person in any such capacity, or arising out of his or her status as such, whether or not the corporation would have the power to indemnify the person against such liability under Section 145 of the Delaware General Corporation Law.
Our restated certificate of incorporation, or the Charter, which will become effective upon completion of the offering, provides that no director of our company shall be personally liable to us or our stockholders for monetary damages for any breach of fiduciary duty as a director, except for liability (1) for any breach of the director's duty of loyalty to us or our stockholders, (2) for acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law, (3) in respect of unlawful dividend payments or stock redemptions or repurchases, or (4) for any transaction from which the director derived an improper personal benefit. In addition, our Charter provides that if the Delaware General Corporation Law is amended to authorize the further elimination or limitation of the liability of directors, then the liability of a director of our company shall be eliminated or limited to the fullest extent permitted by the Delaware General Corporation Law, as so amended.
The Charter further provides that any repeal or modification of such article by our stockholders or amendment to the Delaware General Corporation Law will not adversely affect any right or protection existing at the time of such repeal or modification with respect to any acts or omissions occurring before such repeal or modification of a director serving at the time of such repeal or modification.
Our restated by-laws, or the By-Laws, which will become effective upon completion of the offering, provide that we will indemnify each of our directors and officers and, in the discretion of our board of directors, certain employees, to the fullest extent permitted by the Delaware General Corporation Law as the same may be amended (except that in the case of amendment, only to the extent that the amendment permits us to provide broader indemnification rights than the Delaware General Corporation Law permitted us to provide prior to such the amendment) against any and all expenses, judgments, penalties, fines and amounts reasonably paid in settlement that are incurred by the director, officer or such employee or on the director's, officer's or employee's behalf in connection with any threatened, pending or completed proceeding or any claim, issue or matter therein, to which he or she is or is threatened to be made a party because he or she is or was serving as a director, officer or employee of our company, or at our request as a director, partner, trustee, officer, employee or agent of another corporation, partnership, joint venture, trust, employee benefit plan or other enterprise, if he or she acted in good faith and in a manner he or she reasonably believed to be in or not opposed to the best interests of our company and, with respect to any criminal proceeding, had no reasonable cause to believe his or her conduct was unlawful. Article of the By-Laws further provides for the advancement of expenses to each of our directors and, in the discretion of the board of directors, to certain officers and employees.
In addition, the By-Laws provide that the right of each of our directors and officers to indemnification and advancement of expenses shall be a contract right and shall not be exclusive of any other right now possessed or hereafter acquired under any statute, provision of the Charter or By-Laws, agreement, vote of stockholders or otherwise. Furthermore, Article of the By-Laws authorizes us to provide insurance for
II-2
our directors, officers and employees, against any liability, whether or not we would have the power to indemnify such person against such liability under the Delaware General Corporation Law or the provisions of Article of the By-Laws.
In connection with the sale of common stock being registered hereby, we will enter into indemnification agreements with each of our directors and our executive officers. These agreements will provide that we will indemnify each of our directors and such officers to the fullest extent permitted by law and the Charter and By-Laws.
We also maintain a general liability insurance policy, which covers certain liabilities of directors and officers of our company arising out of claims based on acts or omissions in their capacities as directors or officers.
In any underwriting agreement we enter into in connection with the sale of common stock being registered hereby, the underwriters will agree to indemnify, under certain conditions, us, our directors, our officers and persons who control us within the meaning of the Securities Act of 1933, as amended, against certain liabilities.
Item 15. Recent sales of unregistered securities.
Set forth below is information regarding shares of preferred stock, common stock and warrants issued, and options granted, by us within the past three years that were not registered under the Securities Act. Also included is the consideration, if any, received by us for such shares, warrants and options, and information relating to the section of the Securities Act, or rule of the Securities and Exchange Commission, under which exemption from registration was claimed.
Issuances of stock and warrants
A. On January 16, 2015 and May 28, 2015, we issued 1,501,546 shares of Series C preferred stock and 600,618 shares of Series C preferred stock, respectively, to a collaborator at a purchase price of $3.3299 per share for an aggregate of $5.0 million and $2.0 million, respectively, upon the achievement of equity milestones under our joint development agreement with the collaborator. The 8,061,612 shares of Series C preferred stock outstanding, including the shares of Series C preferred stock described in this paragraph, will convert into 8,061,612 shares of common stock upon the closing of this offering.
B. On January 29, 2016, we issued a warrant to purchase 57,810 shares of Series C preferred stock to our lender in connection with an amendment to our loan facility.
C. On February 4, 2016, we issued 1,300,000 shares of Series A-3 preferred stock to one accredited investor upon the exercise of warrants to purchase 1,300,000 shares of Series A-3 preferred stock at an exercise price of $0.001 per share for an aggregate of $1,300. On January 26, 2017, we issued 700,000 shares of Series A-3 preferred stock to the same accredited investor upon the exercise of warrants to purchase 700,000 shares of Series A-3 preferred stock at an exercise price of $0.001 per share for an aggregate of $700. The 2,000,000 shares of Series A-3 preferred stock outstanding, including the shares of Series A-3 preferred stock described in this paragraph, will convert into 2,000,000 shares of common stock upon the closing of this offering.
D. On March 18, 2016, we issued an aggregate of 12,420,262 shares of Series D preferred stock to 15 accredited investors at a purchase price of $3.67 per share for an aggregate of $45.6 million. The 12,420,262 shares of Series D preferred stock outstanding will convert into 12,420,262 shares of common stock upon the closing of this offering.
II-3
E. From June 27, 2016 through December 6, 2016, we issued an aggregate of 397,530 shares of Series B preferred stock to 10 accredited investors upon the exercise of warrants to purchase an aggregate of 397,530 shares of Series B preferred stock. Warrants to purchase 8,330 shares of Series B preferred stock were exercised at an exercise price of $2.00 per share for an aggregate of $16,600, warrants to purchase Series B preferred stock were exchanged for 76,700 shares of Series B preferred stock in a cashless transaction, and warrants to purchase 312,500 shares of Series B preferred stock were exercised at a price of $0.001 per share for an aggregate of $313. The 6,021,636 shares of Series B preferred stock outstanding, including the shares of Series B preferred stock described in this paragraph, will convert into 6,021,636 shares of common stock upon the closing of this offering.
F. On March 31, 2017, we issued a warrant to purchase 38,828 shares of Series D preferred stock to our lender in connection with an amendment to our loan facility.
G. On June 2, 2017, we issued an aggregate of 2,113,902 shares of Series D-1 preferred stock to five accredited investors at a purchase price of $4.021 per share for an aggregate of $8.5 million. The 2,113,902 shares of Series D preferred stock outstanding will convert into 2,113,902 shares of common stock upon the closing of this offering.
H. From November 1, 2014 through October 31, 2017, we issued an aggregate of 1,249,152 shares of common stock upon the exercise of options and an aggregate of 2,763,953 shares of common stock representing stock awards to certain of our employees, directors and consultants under the 2007 Stock Option and Grant Plan, as amended.
Stock option and restricted stock grants
From November 1, 2014 through October 31, 2017, we granted (i) stock options under the 2007 Stock Option and Grant Plan, as amended, to purchase an aggregate of 5,093,767 shares of common stock, net of forfeitures, at a weighted-average exercise price of $2.32 per share, to certain of our employees, consultants and directors, and (ii) 2,763,953 shares of restricted common stock to one of our executive officers.
Securities act exemptions
The offers, sales and issuances of the securities described above were exempt from registration under the Securities Act in reliance on Section 4(a)(2) of the Securities Act and Rule 506 of Regulation D.
The grants of stock options described above under "—Stock Option Grants" were exempt from registration under the Securities Act in reliance on Rule 701 promulgated under the Securities Act as offers and sales of securities under compensatory benefit plans and contracts relating to compensation in compliance with Rule 701. Each of the recipients of securities in any transaction exempt from registration either received or had adequate access, through employment, business or other relationships, to information about us.
All certificates representing the securities issued in the transactions described in this Item 15 included appropriate legends setting forth that the securities had not been offered or sold pursuant to a registration statement and describing the applicable restrictions on transfer of the securities. There were no underwriters employed in connection with any of the transactions set forth in this Item 15.
Item 16. Exhibits and financial statement schedules.
(a) Exhibits.
See the Exhibit Index attached to this registration statement, which is incorporated by reference herein.
II-4
(b) Financial Statement Schedules.
No financial statement schedules are provided because the information called for is not required or is shown either in the financial statements or notes.
The undersigned Registrant hereby undertakes to provide to the underwriters at the closing specified in the underwriting agreement certificates in such denominations and registered in such names as required by the underwriters to permit prompt delivery to each purchaser.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
The undersigned Registrant hereby undertakes that:
(a) For purposes of determining any liability under the Securities Act, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective.
(b) For the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
II-5
II-6
II-7
| | | | |
| Exhibit number |
Description of exhibit | ||
| | | | |
| 10.17.3 | Amendment No. 2 to Loan and Security Agreement, dated January 29, 2016, by and between the Registrant and Hercules Capital, Inc. (formerly known as Hercules Technology Growth Capital, Inc.). | ||
| 10.17.4 | Amendment No. 3 to Loan and Security Agreement, dated March 31, 2017, by and between the Registrant and Hercules Capital, Inc. (formerly known as Hercules Technology Growth Capital, Inc.). | ||
| 10.18 | *@ | Form of Indemnification Agreement. | |
| 10.19 | @ | Letter Agreement, dated January 1, 2014, by and between the Registrant and David R. Walt, Ph.D. | |
| 21.1 | Subsidiaries of Registrant. | ||
| 23.1 | Consent of Ernst & Young LLP. | ||
| 23.2 | * | Consent of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. (included in Exhibit 5.1). | |
| 24.1 | Power of Attorney (included on signature page). | ||
| 99.1 | Consent of Health Advances LLC. | ||
| | | | |
- *
- To
be filed by amendment.
- #
- Confidential
treatment has been requested for portions of this exhibit. These portions have been omitted from the registration statement and have been filed separately
with the U.S. Securities and Exchange Commission.
- @
- Denotes management compensation plan or contract.
II-8
Pursuant to the requirements of the Securities Act of 1933, as amended, the Registrant has duly caused this registration statement on Form S-1 to be signed on its behalf by the undersigned, thereunto duly authorized, in Lexington, Massachusetts, on the 9th day of November, 2017.
| QUANTERIX CORPORATION | ||
/s/ E. KEVIN HRUSOVSKY E. Kevin Hrusovsky Executive Chairman, President and Chief Executive Officer |
Signatures and power of attorney
We, the undersigned directors and officers of Quanterix Corporation (the "Company"), hereby severally constitute and appoint E. Kevin Hrusovsky and Joseph Driscoll, and each of them singly, our true and lawful attorneys, with full power to them, and to each of them singly, to sign for us and in our names in the capacities indicated below, the registration statement on Form S-1 filed herewith, and any and all pre-effective and post-effective amendments to said registration statement, and any registration statement filed pursuant to Rule 462(b) under the Securities Act of 1933, as amended, in connection with the registration under the Securities Act of 1933, as amended, of equity securities of the Company, and to file or cause to be filed the same, with all exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as each of us might or could do in person, and hereby ratifying and confirming all that said attorneys, and each of them, or their substitute or substitutes, shall do or cause to be done by virtue of this Power of Attorney.
Pursuant to the requirements of the Securities Act of 1933, as amended, this registration statement on Form S-1 has been signed by the following persons in the capacities and on the dates indicated.
Signature
|
Title
|
Date
|
||
|---|---|---|---|---|
| /s/ E. KEVIN HRUSOVSKY E. Kevin Hrusovsky |
Executive Chairman, President and Chief Executive Officer and Director (principal executive officer) | November 9, 2017 | ||
/s/ JOSEPH DRISCOLL Joseph Driscoll |
Chief Financial Officer (principal financial officer and principal accounting officer) |
November 9, 2017 |
||
/s/ DOUGLAS G. COLE, M.D. Douglas G. Cole, M.D. |
Director |
November 9, 2017 |
II-9
Signature
|
Title
|
Date
|
||
|---|---|---|---|---|
| /s/ JOHN M. CONNOLLY John M. Connolly |
Director | November 9, 2017 | ||
/s/ KEITH L. CRANDELL Keith L. Crandell |
Director |
November 9, 2017 |
||
/s/ MARIJN DEKKERS, PH.D. Marijn Dekkers, Ph.D. |
Director |
November 9, 2017 |
||
/s/ MARTIN D. MADAUS, PH. D. Martin D. Madaus, Ph. D. |
Director |
November 9, 2017 |
||
/s/ PAUL M. MEISTER Paul M. Meister |
Director |
November 9, 2017 |
||
/s/ DAVID R. WALT, PH.D. David R. Walt, Ph.D. |
Director |
November 9, 2017 |
II-10