Exhibit 99.1
|
|
|
FOR RELEASE ON JULY 24, 2014 at 4:01 PM ET
TESARO ANNOUNCES SECOND-QUARTER 2014 OPERATING RESULTS
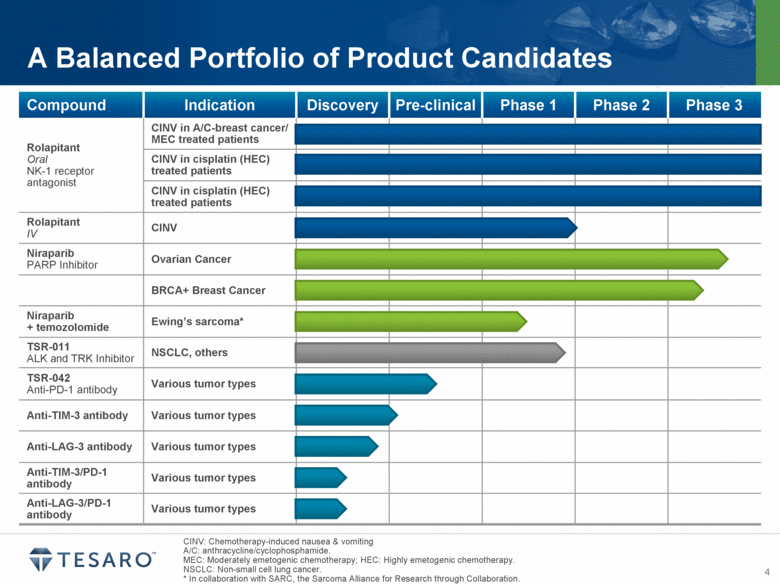
· Oral Rolapitant New Drug Application (NDA) on Track for Submission to U.S. FDA
· Enrollment in Niraparib NOVA Trial Non-gBRCA Cohort Expected to Be Complete at Year End
· Enrollment in Phase 1 Trial of Niraparib in Patients with Ewing’s Sarcoma Initiated
· Anti-TIM-3 Clinical Lead and Back Up Antibodies Identified
· Cash and Cash Equivalents Totaled Approximately $151 Million as of June 30, 2014
WALTHAM, MA, July 24, 2014 — TESARO, Inc. (NASDAQ: TSRO), an oncology-focused biopharmaceutical company, today reported financial results for the second quarter of 2014.
“We are very pleased with the feedback we received from the oncology community regarding the rolapitant pivotal data set presented at the recent ASCO and MASCC conferences,” said Lonnie Moulder, CEO of TESARO. “The rolapitant NDA remains on track for submission to the U.S. FDA in approximately six weeks, and pre-launch commercial activities are well underway. We are also pleased with the pace of enrollment in our Phase 3 niraparib NOVA trial for patients with ovarian cancer, and we expect the non-germline BRCA cohort to be fully enrolled at the end of this year.”
Recent Business Highlights
· Final results from three Phase 3 clinical trials of oral rolapitant were presented in June at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago and at the MASCC/ISOO International Symposium on Supportive Care in Cancer annual meeting in Miami. Each of the three Phase 3 trials successfully achieved the primary endpoint of complete response, defined as no emesis and no use of rescue medication, during the delayed phase following administration of chemotherapy.
· A multiple ascending dose (MAD) study of intravenous (IV) rolapitant has been completed and confirmed the safety and tolerability profile of repeated daily doses.
· Patient enrollment continues in the Phase 3 NOVA and BRAVO trials of niraparib, and planning is underway to support initiation of additional niraparib clinical trials in the small cell lung cancer and first line ovarian cancer maintenance settings.
· Patient enrollment has begun in the Phase 1 study of niraparib plus temozolomide in patients with Ewing’s sarcoma;
· The clinical activity of a fractionated dose of TSR-011 continues to be evaluated in ALK-positive and TRK-positive patients.
· Preclinical studies of TSR-042 (anti-PD-1 antibody candidate) continue in collaboration with AnaptysBio, and lead and back up clinical antibody candidates targeting TIM-3 have been identified.
Second-Quarter 2014 Financial Results
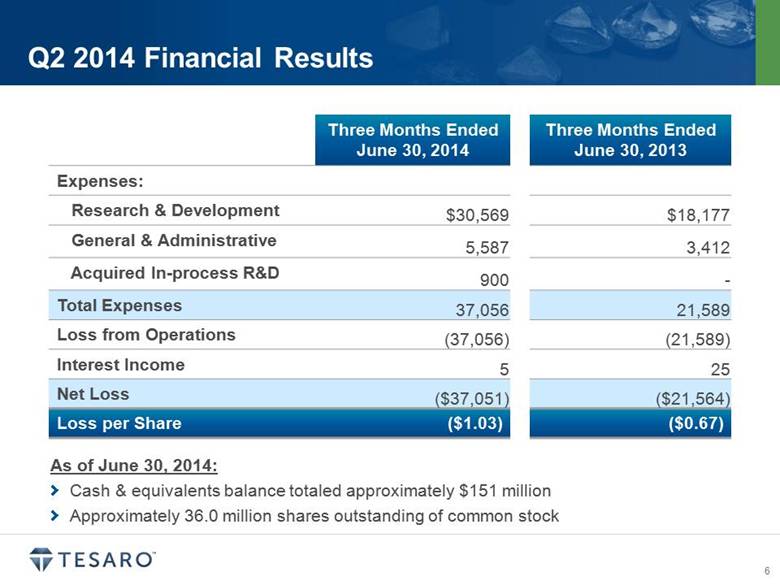
· TESARO reported a net loss of $37.1 million, or $1.03 per share, for the second quarter of 2014, compared to a net loss of $21.6 million, or $0.67 per share, for the second quarter of 2013.
· Research and development expenses increased to $30.6 million for the second quarter of 2014, compared to $18.2 million for the second quarter of 2013, driven primarily by higher costs related to expanded development activities and increased headcount. The Company also recorded $0.9 million of in-process research and development expense as a result of making a milestone payment related to niraparib clinical development during the second quarter of 2014.
· General and administrative expenses increased to $5.6 million for the second quarter of 2014, compared to $3.4 million for the second quarter of 2013, primarily related to increased headcount, higher non-cash stock-based compensation expense and higher professional service fees.
· Operating expenses as described above include total non-cash stock-based compensation expense of $3.1 million for the second quarter of 2014, compared to $1.6 million for the second quarter of 2013.
· As of June 30, 2014, TESARO had approximately $151.1 million in cash and cash equivalents, no debt and approximately 36.0 million outstanding shares of common stock.
2014 Corporate Objectives
TESARO anticipates achieving the following key objectives during the second half of 2014:
· Submit the oral rolapitant New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) in approximately six weeks;
· Initiate a clinical trial of IV rolapitant to support the planned future NDA submission for registration following regulatory approval of oral rolapitant;
· Continue to advance the Phase 3 NOVA trial of niraparib as a maintenance therapy for patients with ovarian cancer and the Phase 3 BRAVO trial of niraparib in breast cancer patients with germline BRCA mutations;
· Advance the TSR-011 controlled release formulation work and further evaluate the clinical activity of a fractionated dose of TSR-011 in ALK-positive and TRK-positive patients;
· Advance the development of TSR-042 (anti-PD-1 antibody) to support submission of an Investigational New Drug (IND) Application to the U.S. FDA in late 2015; and
· Initiate IND enabling studies for the anti-TIM-3 clinical candidate and select an anti-LAG-3 antibody for clinical development during the third quarter of 2014.
Today’s Conference Call and Webcast
TESARO will host a conference call to discuss the Company’s second quarter operating results today at 4:15 p.m. Eastern time. The accompanying slide presentation and live webcast of the conference call can be accessed by visiting the TESARO website at www.tesarobio.com. The call can be accessed by dialing (877) 853-5334 (U.S. and Canada) or (970) 315-0307 (international). A replay of the webcast will be archived on the Company’s website for 30 days following the call.
About TESARO
TESARO is an oncology-focused biopharmaceutical company dedicated to improving the lives of cancer patients by acquiring, developing and commercializing safer and more effective therapeutics. For more information, visit www.tesarobio.com.
Investor/Media Contact:
Jennifer Davis
Sr. Director, Corporate Development & Investor Relations
+1.781.325.1116 or jdavis@tesarobio.com
To the extent that statements contained in this press release are not descriptions of historical facts regarding TESARO, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Words such as “may,” “will,” “expect,” “anticipate,” “estimate,” “intend,” and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) are intended to identify forward-looking statements. Examples of forward looking statements contained in this press release include, among others, statements regarding our expectations regarding our development programs for our product candidates and statements under the heading 2014 Corporate Objectives. Forward-looking statements in this release involve substantial risks and uncertainties that could cause our clinical development programs, future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, the uncertainties inherent in the initiation and conduct of clinical trials, availability of data from ongoing clinical trials, the challenges in selecting product candidates, expectations with respect to regulatory submissions and approvals, challenges associated with expanding operations to include commercial activities, and other matters that could affect the availability or commercial potential of our drug candidates. TESARO undertakes no obligation to update or revise any forward-looking statements. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to the business of the Company in general, see TESARO’s Annual Report on Form 10-K for the year ended December 31, 2013 and other filings TESARO makes with the Securities and Exchange Commission.
###
TESARO, Inc.
Unaudited Condensed Consolidated Statements of Operations
(in thousands, except per share amounts)
|
|
|
Three Months Ended |
|
Six Months Ended |
| |||||||||
|
|
|
June 30, |
|
June 30, |
| |||||||||
|
|
|
2013 |
|
2014 |
|
2013 |
|
2014 |
| |||||
|
Expenses: |
|
|
|
|
|
|
|
|
| |||||
|
Research and development (1) |
|
$ |
18,177 |
|
$ |
30,569 |
|
$ |
34,680 |
|
$ |
58,686 |
| |
|
General and administrative (1) |
|
3,412 |
|
5,587 |
|
5,812 |
|
10,275 |
| |||||
|
Acquired in-process research and development |
|
— |
|
900 |
|
— |
|
17,900 |
| |||||
|
Total expenses |
|
21,589 |
|
37,056 |
|
40,492 |
|
86,861 |
| |||||
|
Loss from operations |
|
(21,589 |
) |
(37,056 |
) |
(40,492 |
) |
(86,861 |
) | |||||
|
Interest income |
|
25 |
|
5 |
|
59 |
|
10 |
| |||||
|
Net loss |
|
$ |
(21,564 |
) |
$ |
(37,051 |
) |
$ |
(40,433 |
) |
$ |
(86,851 |
) | |
|
|
|
|
|
|
|
|
|
|
| |||||
|
Net loss per share applicable to common stockholders - basic and diluted |
|
$ |
(0.67 |
) |
$ |
(1.03 |
) |
$ |
(1.32 |
) |
$ |
(2.45 |
) | |
|
|
|
|
|
|
|
|
|
|
| |||||
|
Weighted-average number of common shares used in net loss per share applicable to common stockholders - basic and diluted |
|
32,336 |
|
35,982 |
|
30,577 |
|
35,422 |
| |||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
| | ||||||||||||||
|
(1) Expenses include the following amounts of non-cash stock-based compensation expense: | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
Research and development |
|
$ |
462 |
|
$ |
1,454 |
|
$ |
794 |
|
$ |
2,364 |
|
|
|
General and administrative |
|
1,122 |
|
1,648 |
|
1,560 |
|
3,276 |
| ||||
TESARO, Inc.
Unaudited Condensed Consolidated Balance Sheets
(in thousands)
|
|
|
December 31, |
|
June 30, |
| ||
|
|
|
2013 |
|
2014 |
| ||
|
Assets |
|
|
|
|
| ||
|
Current assets: |
|
|
|
|
| ||
|
Cash and cash equivalents |
|
$ |
130,310 |
|
$ |
151,088 |
|
|
Other current assets |
|
4,029 |
|
2,997 |
| ||
|
Total current assets |
|
134,339 |
|
154,085 |
| ||
|
|
|
|
|
|
| ||
|
Property and equipment, net |
|
440 |
|
1,147 |
| ||
|
Other assets |
|
799 |
|
908 |
| ||
|
Total assets |
|
$ |
135,578 |
|
$ |
156,140 |
|
|
|
|
|
|
|
| ||
|
Liabilities and stockholders’ equity |
|
|
|
|
| ||
|
Current liabilities: |
|
|
|
|
| ||
|
Accounts payable |
|
$ |
1,869 |
|
$ |
2,849 |
|
|
Accrued expenses |
|
10,541 |
|
16,163 |
| ||
|
Other current liabilities |
|
13 |
|
— |
| ||
|
Total current liabilities |
|
12,423 |
|
19,012 |
| ||
|
|
|
|
|
|
| ||
|
Other non-current liabilities |
|
3 |
|
— |
| ||
|
|
|
|
|
|
| ||
|
Commitments and contingencies |
|
|
|
|
| ||
|
|
|
|
|
|
| ||
|
Total stockholders’ equity |
|
123,152 |
|
137,128 |
| ||
|
Total liabilities and stockholders’ equity |
|
$ |
135,578 |
|
$ |
156,140 |
|