Exhibit 99.1
|
|
|
|
FOR RELEASE ON MARCH 13, 2014 at 7:01 AM ET
TESARO AND ANAPTYSBIO ANNOUNCE COLLABORATION AND
EXCLUSIVE WORLDWIDE LICENSE AGREEMENT FOR
MULTIPLE IMMUNO-ONCOLOGY PROGRAMS
· Immuno-oncology platform provides a portfolio of candidates focused on high value targets, potentially augments the value of other pipeline candidates and enhances prospects for future collaborations to advance TESARO’s overall oncology strategy
· Potential first-in-class anti-TIM-3 antibody clinical candidate expected to be selected second-quarter 2014
· Anti-LAG-3 antibody clinical candidate expected to be selected by third-quarter 2014
· Dual reactive antibody clinical candidates targeting PD-1/TIM-3 and PD-1/LAG-3 expected to be selected by first-quarter 2015
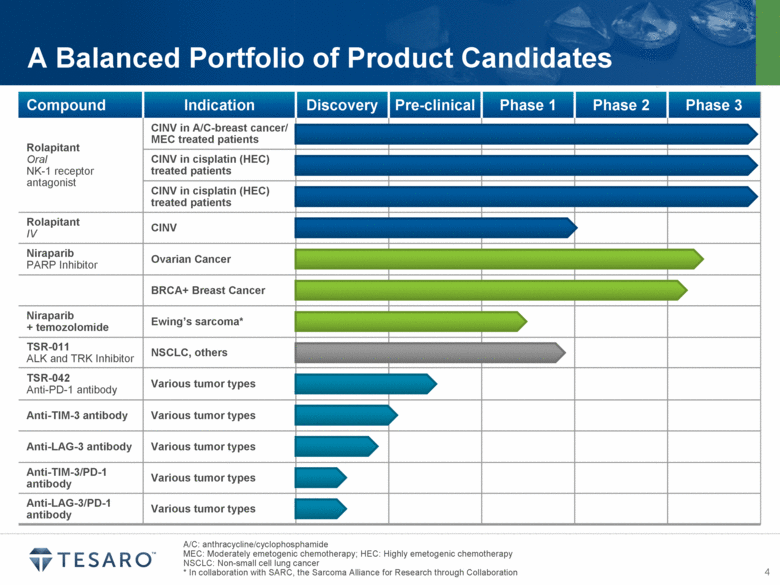
· Investigation of preclinical combinations of TSR-042 (anti-PD-1 antibody) with each of TSR-011 (ALK/TRK inhibitor) and niraparib (PARP inhibitor) planned
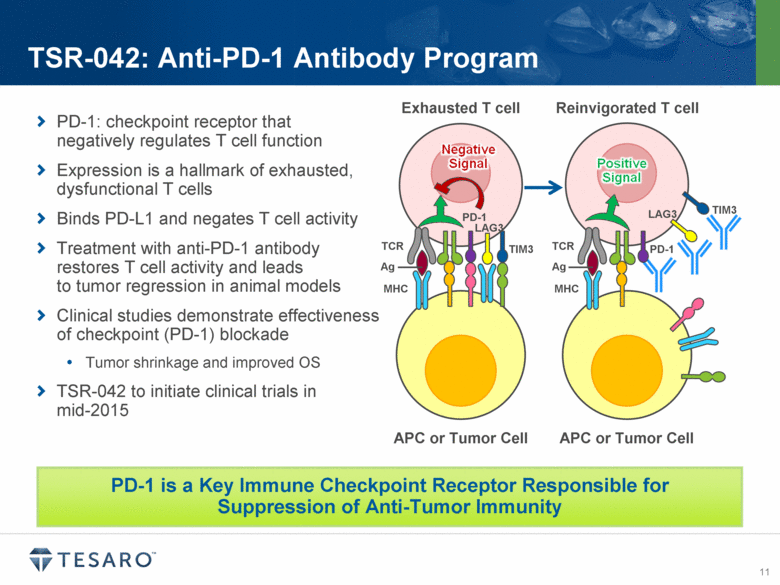
· First clinical trial of TSR-042 projected to begin in mid-2015
· Conference call and webcast scheduled for 8:30 AM ET today
WALTHAM, MA, and SAN DIEGO, CA, March 13, 2014 — TESARO, Inc. (NASDAQ: TSRO), an oncology-focused biopharmaceutical company, and AnaptysBio, Inc., a privately held therapeutic antibody company, today announced an exclusive, worldwide license agreement and immuno-oncology antibody collaboration. Under the terms of the agreement, TESARO receives rights to monospecific antibody drug candidates targeting TIM-3, LAG-3 and PD-1 and dual reactive antibody drug candidates targeting PD-1/TIM-3 and PD-1/LAG-3. Therapeutic antibodies selected from these programs will form the basis of a strategic platform that will enable TESARO to develop novel monotherapy and combination-based approaches with immuno-oncology and other anti-cancer agents in a variety of indications. Antibody candidates from these programs are expected to enter clinical trials over the next 18 to 24 months.
“This agreement provides TESARO with a basis from which to develop novel therapeutics, including combination-based approaches, against a variety of tumor targets and may uniquely position TESARO for collaborations with new partners,” said Mary Lynne Hedley, Ph.D., president of TESARO. “These antibody candidates are being developed to address some of the most validated and promising targets in immuno-oncology. We are also interested in evaluating combinations of these antibodies with TSR-011, our ALK/TRK inhibitor, and niraparib, our PARP inhibitor, in addition to other anti-tumor agents with complementary mechanisms, such as immune modulating agents. The first clinical trial from this collaboration is projected to begin in mid-2015, and we expect to advance an additional candidate into clinical trials every one to two quarters thereafter.”
“We view immuno-oncology as a platform that can potentially transform the way in which numerous cancers are treated, and we expect immunotherapy-based approaches to become the
foundation of many future cancer therapy regimens,” stated Lonnie Moulder, CEO of TESARO. “Our team looks forward to collaborating with AnaptysBio’s scientists to rapidly move multiple novel antibody candidates into clinical trials and potentially explore additional immuno-oncology targets.”
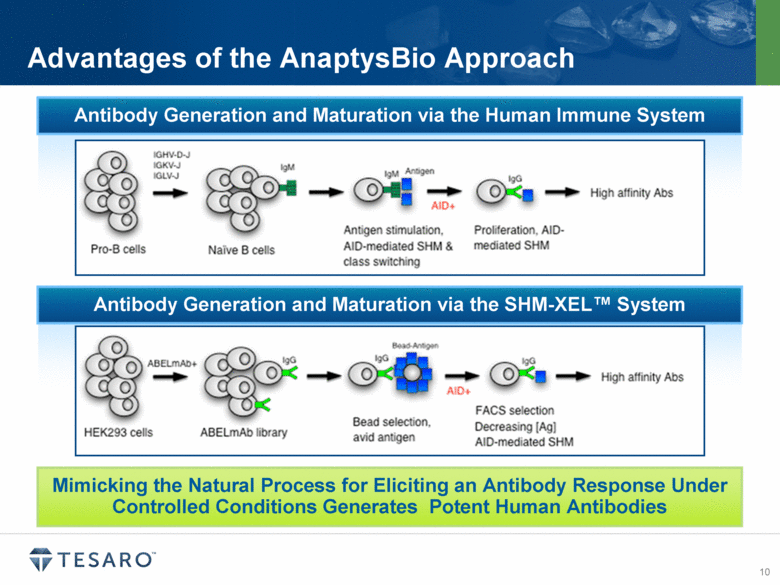
“AnaptysBio’s SHM-XEL platform has successfully generated highly potent therapeutic antibodies against multiple immuno-oncology checkpoint receptors. In addition to selecting for antibodies with optimal checkpoint antagonist function, our mammalian cell display system permits simultaneous selection for high antibody expression, stability and robust biophysical features required for successful development and ideal pharmacokinetic properties,” said Hamza Suria, president and CEO of AnaptysBio. “Our programs attracted partnership proposals from several companies interested in developing novel immuno-oncology antibody combinations. We specifically chose to collaborate with TESARO because of its strategic focus in oncology and the proven expertise of TESARO’s development team. We look forward to working with TESARO to advance these therapies to the clinic and deliver new treatment options for cancer patients.”
Agreement Terms
Under the terms of this agreement with AnaptysBio, TESARO will pay an upfront license fee of $17 million, as well as provide funding of costs incurred by AnaptysBio related to the development programs. For each development program, AnaptysBio is eligible to receive milestone payments of $18 million if certain research and development events are achieved and an additional $90 million associated with certain U.S. and ex-U.S. regulatory submissions and approvals in multiple indications. AnaptysBio will also be eligible to receive tiered single-digit royalties related to worldwide net sales of products developed under the collaboration and certain commercial milestone payments if specified levels of annual worldwide net sales are attained. AnaptysBio and TESARO will together complete preclinical development of the antibody candidates, with TESARO being solely responsible for all clinical development, manufacturing, regulatory and commercial activities.
Immuno-Oncology Platform
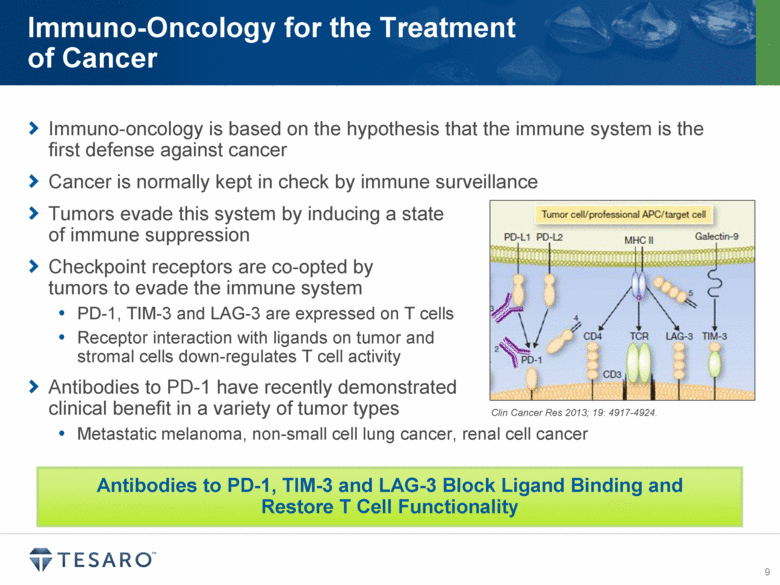
Antibodies to immune checkpoint receptors have recently demonstrated promise in the treatment of certain solid tumors, including metastatic melanoma, renal cell carcinoma and non-small cell lung cancer. Although the normal function of immune checkpoint receptors is to maintain immune homeostasis, they are co-opted by certain tumors to evade immune surveillance. PD-1, TIM-3 and LAG-3 are each checkpoint regulators that modulate the function of the immune system via different mechanisms, and may limit the ability of the immune system to respond effectively to tumors. By blocking the interaction of PD-1, TIM-3 and LAG-3 with their respective ligands, the antibodies exclusively licensed under this collaboration aim to restore immune anti-cancer function in patients across a variety of tumor types.
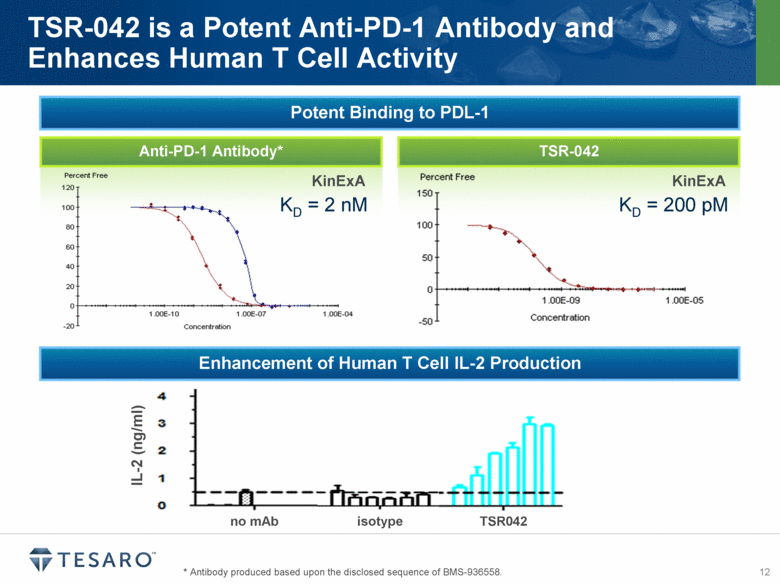
PD-1, or programmed death-1, is a key immune checkpoint molecule that can limit T-cell-mediated immune responses. The presence of the PD-1 ligand, PD-L1 has been identified on many
tumor types, and expression of PD-L1 has been linked to poor clinical outcomes in a variety of cancers. Anti-PD-1 antibodies have demonstrated in vivo efficacy in tumor models and have shown promising results in several clinical studies. TSR-042 is anticipated to begin clinical trials in mid-2015, and combination preclinical pharmacology studies with TSR-011, niraparib and other anti-tumor agents are planned to initiate during 2014.
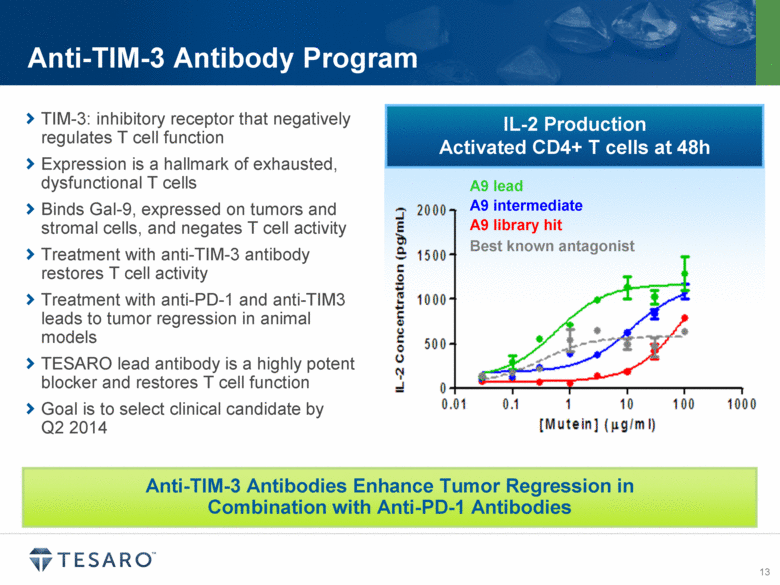
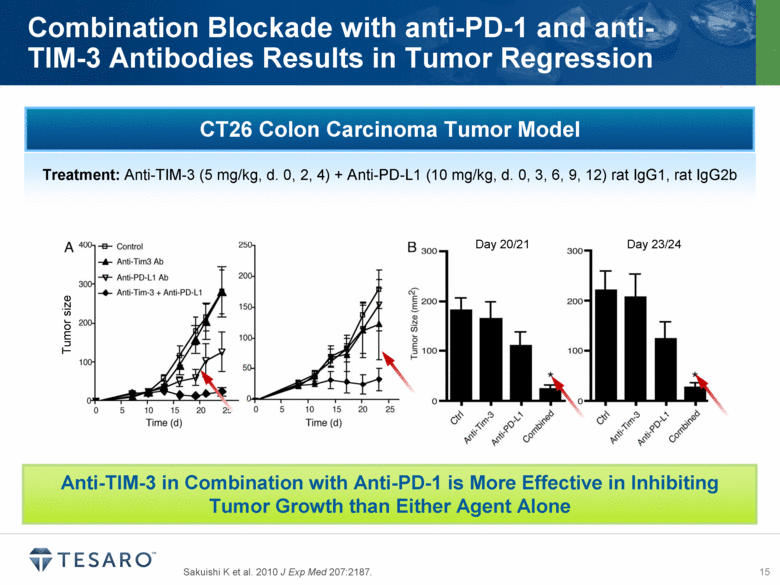
TIM-3, or T-cell immunoglobulin and mucin domain-3, functions as a pattern recognition receptor that dampens the anti-tumor immune response. Anti-TIM-3 antibodies have shown preclinical anti-tumor activity and may enhance anti-tumor immunity in combination with an anti-PD-1 agent or other immune modulating molecules. In collaboration with AnaptysBio, TESARO expects to select a TIM-3 antibody for clinical development during the second quarter of 2014.
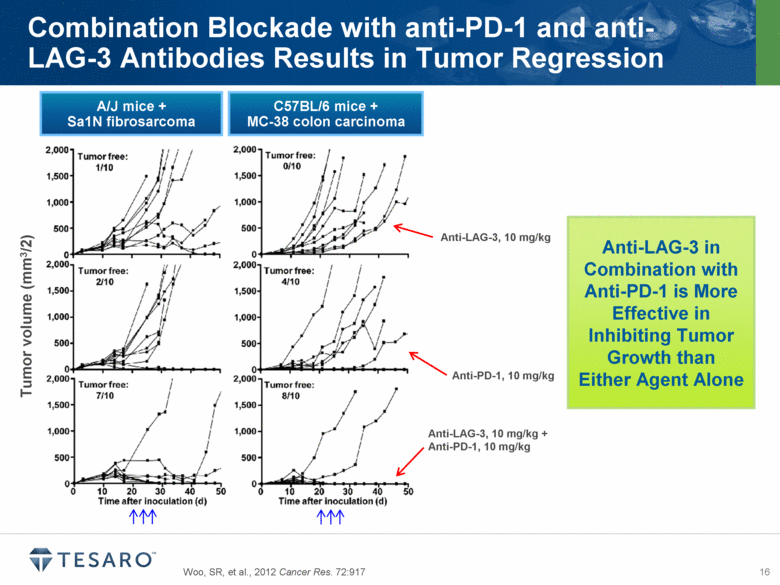
LAG-3, or lymphocyte activation gene-3, is a negative regulator of T-cell activity. Preclinical studies have demonstrated anti-tumor activity by blocking LAG-3 and PD-1 in tumor models, and LAG-3 IgG fusion protein has demonstrated promising results in clinical trials for various solid tumors. In collaboration with AnaptysBio, TESARO expects to select a LAG-3 antibody for clinical development in the third quarter of 2014.
TESARO Conference Call, Webcast and Slide Presentation
TESARO will host a conference call to discuss this license agreement today at 8:30 a.m. Eastern time. The live webcast of the conference call and the accompanying slide presentation can be accessed by visiting the Investors section of the TESARO website at www.tesarobio.com. The conference call can be accessed by dialing (877) 853-5334 (U.S. and Canada) or (970) 315-0307 (international). A replay of the webcast will be archived on the Company’s website for 30 days following the call.
About AnaptysBio
AnaptysBio is a privately-held company focused on the generation of antibody therapeutics and is the leader in the use of somatic hypermutation (SHM) for antibody discovery and optimization. AnaptysBio is developing a pipeline of novel therapeutic antibody candidates, including differentiated programs in immuno-oncology, inflammation, fibrosis, muscle wasting disorders and antibody-drug conjugate applications. AnaptysBio’s proprietary SHM-XEL™ platform, which couples fully human antibody libraries with in vitro somatic hypermutation in mammalian cells to generate high affinity antibodies, replicates key features of the human immune system and overcomes limitations of prior antibody technologies. For more information, visit www.AnaptysBio.com.
About TESARO
TESARO is an oncology-focused biopharmaceutical company dedicated to improving the lives of cancer patients by acquiring, developing and commercializing safer and more effective therapeutics. For more information, visit www.tesarobio.com.
TESARO Contact:
Jennifer Davis
Sr. Director, Corporate Development & Investor Relations
+1.781.325.1116 or jdavis@tesarobio.com
AnaptysBio Contact:
Julie Rathbun
+1.206.769.9219 or julie@rathbuncomm.com
To the extent that statements contained in this press release are not descriptions of historical facts regarding TESARO, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Words such as “may,” “will,” “expect,” “anticipate,” “estimate,” “intend,” and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) are intended to identify forward-looking statements. Examples of forward looking statements contained in this press release include, among others, statements regarding our expectations regarding the timing of both the selection of clinical candidates from the programs and the commencement of clinical testing, our development plans for any antibody therapeutic candidates individually and in combination other products and product candidates, and our ability to form partnerships in the future in support of our overall oncology strategy. Forward-looking statements in this release involve substantial risks and uncertainties that could cause our research and pre-clinical development programs, clinical development programs, future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, the uncertainties inherent in the research and development of therapeutic antibodies, including the selection, pre-clinical testing and manufacturing of antibodies, initiation of future clinical trials, availability of data from ongoing clinical trials, expectations for regulatory approvals, and other matters that could affect the availability or commercial potential of our drug candidates. TESARO undertakes no obligation to update or revise any forward-looking statements. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to the business of the Company in general, see TESARO’s Annual Report on Form 10-K for the year ended December 31, 2012, and Quarterly Report on Form 10-Q for the quarter ended September 30, 2013.
###