Exhibit 99.1

TESARO Announces Fourth Quarter and Year-end 2012 Operating Results
· Niraparib to be Advanced in a Phase 3 Pivotal Trial for Ovarian Cancer
· Enrollment Continues in the Global Registration Program for Rolapitant
· Achieved Third Dose-level in a Phase 1/2 Clinical Trial of TSR-011
Waltham, Massachusetts — February 14, 2013 — TESARO, Inc. (Nasdaq: TSRO) an oncology-focused biopharmaceutical company today reported financial results for the fourth quarter and full year 2012 and provided an update on the Company’s progress.
“We are very pleased to be advancing niraparib into a registration program in the maintenance setting for platinum sensitive ovarian cancer and intend to initiate this Phase 3 clinical trial by mid-year,” said Lonnie Moulder, Chief Executive Officer. “In addition, we are on track to report topline results from the rolapitant global registration program in the second half of the year, and have achieved the third dose-level in a Phase 1/2 clinical trial for TSR-011. We look forward to an exciting and productive 2013 for TESARO.”
Recent Accomplishments
TESARO continues to advance the development of its pipeline of oncology supportive care and anticancer product candidates.
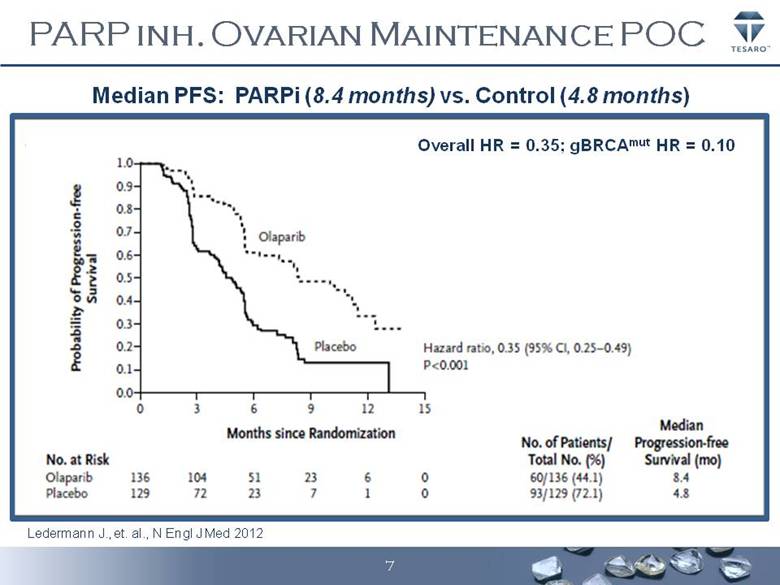
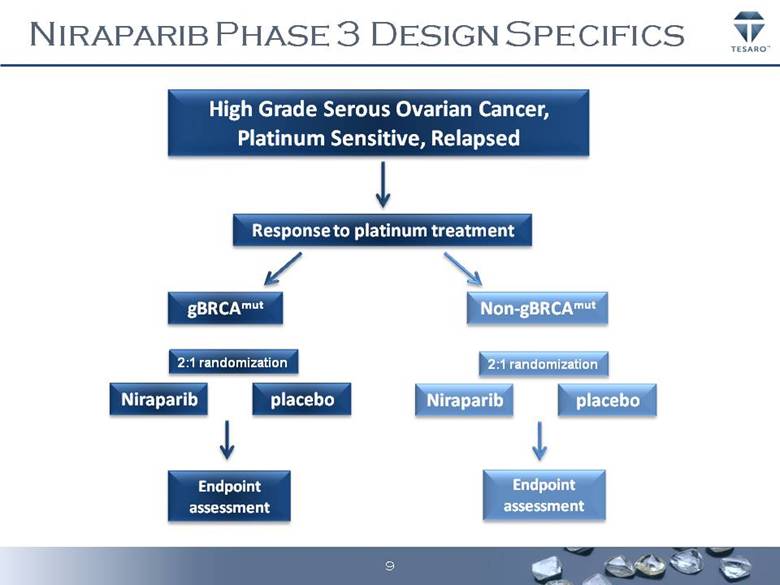
· The Company has met with the FDA and finalized its clinical development program for niraparib, an orally active and potent PARP (poly (ADP-ribose) polymerase) inhibitor, in the ovarian cancer setting. TESARO will initiate a pivotal Phase 3 randomized double-blind, international trial to evaluate the potential benefit of niraparib compared to a placebo on the maintenance of response following completion of a platinum containing chemotherapy regimen. The study will separately evaluate the benefit of niraparib in two groups of ovarian cancer patients, those with and without certain deficiencies in DNA repair. The rationale for this study includes robust Phase 1 and 2 data showing a high response rate in patients with ovarian cancer who are treated with PARP inhibitors, compelling Phase 1 data from the dose escalation and expansion clinical study of niraparib, as well as the results of a study with a comparable design in the ovarian cancer maintenance setting that showed a strong progression free survival, or PFS, benefit for patients receiving another investigational PARP inhibitor. The Company expects to initiate this Phase 3 clinical trial by mid-year.
· The Company initiated the rolapitant global registration program in early 2012 and is continuing to enroll cancer patients in each of three phase 3 clinical trials. This global registration program evaluating rolapitant for the prevention of chemotherapy induced nausea and vomiting (CINV) is currently being conducted at more than 200 clinical trial sites located in 25 countries. TESARO intends to announce topline results for the three Phase 3 pivotal trials in the second half of 2013.
· Following clearance in late September of the Investigational New Drug (IND) application for TSR-011, an orally available anaplastic lymphoma kinase (ALK) inhibitor (targeted anti-cancer agent) by the FDA, the first cancer patient was enrolled in a Phase 1/2 clinical study. The third dosing cohort in the dose escalation phase of the study was initiated in January of 2013. Once the maximum tolerated dose of TSR-011 is identified, the Company plans to evaluate TSR-011 in three parallel cohorts of patients: those with ALK positive non-small cell lung cancer (NSCLC) who have not been previously treated with an ALK inhibitor, those with ALK positive NSCLC who have progressed during treatment with an ALK inhibitor and those patients with other tumor types that express ALK.
Fourth Quarter and Year-end 2012 Financial Results
· TESARO reported a net loss of $18.7 million for the fourth quarter of 2012 and $61.8 million for the year ended December 31, 2012. This compares to a net loss of $9.1 million and $16.4 million for the fourth quarter and year ended December 31, 2011, respectively. Net loss attributable to common stockholders for the fourth quarter of 2012 was $0.70 per share, compared to $15.41 per share for the fourth quarter of 2011. Net loss attributable to common stockholders for the year ended December 31, 2012 was $4.51 per share, compared to $31.90 per share for the year ended December 31, 2011.
· Research and development expenses totaled $15.6 million for the fourth quarter of 2012 and $47.2 million for the full year 2012, compared to $8.0 million for the fourth quarter of 2011 and $11.8 million for the full year 2011. Research and development expenses increased from 2011 primarily due to the expanded development activities for rolapitant and TSR-011 as well as the in-licensing of niraparib in mid-2012.
· The Company recorded $8.0 million of acquired in-process research and development expense in 2012, $7.0 million of which related to the in-licensing of niraparib and $1.0 million related to a fourth quarter development milestone payment with respect to TSR-011.
· General and administrative expenses totaled $2.1 million for the fourth quarter of 2012 and $6.7 million for the full year 2012, compared to $1.1 million and $3.2 million for the comparable periods in the previous year. The increase in general and administrative expenses was primarily due to increased personnel and professional fees.
· Operating expenses as described above include stock-based compensation expense of $1.8 million for the full year 2012, compared to $0.3 million for the full year 2011.
· As of December 31, 2012, TESARO had $125.4 million in cash and cash equivalents, no debt and 27.1 million outstanding shares of common stock.
2013 Key Objectives
During 2013, TESARO anticipates achieving the following key objectives:
· Announce topline results for the three Phase 3 pivotal trials evaluating oral rolapitant for the prevention of CINV.
· Advance the clinical development of rolapitant intravenous (IV) formulation in order to support a future submission for registration concurrent with the approval of the oral formulation.
· Begin enrollment of the Phase 3 pivotal trial of niraparib as a potential maintenance therapy for ovarian cancer patients.
· Advance the TSR-011 development program and define a strategy for the next phase of clinical development.
Today’s Conference Call and Webcast Reminder
TESARO will host a conference call to discuss the Company’s fourth quarter and year-end 2012 accomplishments and financial results today at 8:00 a.m. (ET). The call can be accessed by dialing 1.877.853.5334 (US and Canada) or 1.970.315.0307 (international). A live webcast of the conference call can also be accessed by visiting the TESARO website at www.tesarobio.com. A replay of the webcast will be archived on the Company’s website for 30 days following the call.
About TESARO
TESARO is an oncology-focused biopharmaceutical company dedicated to improving the lives of cancer patients by acquiring, developing and commercializing safer and more effective therapeutics. TESARO is headquartered in Waltham, Massachusetts. For more information, visit www.tesarobio.com.
To the extent that statements contained in this press release are not descriptions of historical facts regarding TESARO, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Words such as “may,” “will,” “expect,” “anticipate,” “estimate,” “intend,” and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) are intended to identify forward-looking statements. Examples of forward looking statements contained in this press release include, among others, statements regarding our expectations regarding our development plans for our
product candidates and statements under the heading 2013 Key Objectives. Forward-looking statements in this release involve substantial risks and uncertainties that could cause our clinical development programs, future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, the uncertainties inherent in the initiation of future clinical trials, availability of data from ongoing clinical trials, expectations for regulatory approvals, and other matters that could affect the availability or commercial potential of our drug candidates. TESARO undertakes no obligation to update or revise any forward-looking statements. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to the business of the Company in general, see TESARO’s Prospectus filed with the Securities and Exchange Commission on June 29, 2012, and future filings with the SEC, including TESARO’s Form 10-K for the year ended December 31, 2012.
TESARO, Inc.
Unaudited Condensed Consolidated Statements of Operations
(in thousands, except per share amounts)
|
|
|
Three Months Ended |
|
Years Ended |
| ||||||||
|
|
|
2011 |
|
2012 |
|
2011 |
|
2012 |
| ||||
|
|
|
|
|
|
|
|
|
|
| ||||
|
Expenses: |
|
|
|
|
|
|
|
|
| ||||
|
Research and development |
|
$ |
8,001 |
|
$ |
15,642 |
|
$ |
11,768 |
|
$ |
47,200 |
|
|
General and administrative |
|
1,090 |
|
2,095 |
|
3,158 |
|
6,715 |
| ||||
|
Acquired in-process research and development |
|
— |
|
1,000 |
|
500 |
|
8,000 |
| ||||
|
Total expenses |
|
9,091 |
|
18,737 |
|
15,426 |
|
61,915 |
| ||||
|
Loss from operations |
|
(9,091 |
) |
(18,737 |
) |
(15,426 |
) |
(61,915 |
) | ||||
|
Interest income |
|
13 |
|
40 |
|
38 |
|
152 |
| ||||
|
Other income(expense) |
|
— |
|
— |
|
(1,010 |
) |
— |
| ||||
|
Net loss |
|
$ |
(9,078 |
) |
$ |
(18,697 |
) |
$ |
(16,398 |
) |
$ |
(61,763 |
) |
|
|
|
|
|
|
|
|
|
|
| ||||
|
Net loss per share applicable to common stockholders - basic and diluted |
|
$ |
(15.41 |
) |
$ |
(0.70 |
) |
$ |
(31.90 |
) |
$ |
(4.51 |
) |
|
|
|
|
|
|
|
|
|
|
| ||||
|
Weighted-average number of common shares used in net loss per share applicable to common stockholders - basic and diluted |
|
589 |
|
26,740 |
|
514 |
|
13,696 |
| ||||
TESARO, Inc.
Unaudited Condensed Consolidated Balance Sheets
(in thousands)
|
|
|
December 31, |
|
December 31, |
| ||
|
|
|
|
|
|
| ||
|
Assets |
|
|
|
|
| ||
|
Current assets: |
|
|
|
|
| ||
|
Cash and cash equivalents |
|
$ |
39,825 |
|
$ |
125,445 |
|
|
Other current assets |
|
2,606 |
|
1,175 |
| ||
|
Total current assets |
|
42,431 |
|
126,620 |
| ||
|
|
|
|
|
|
| ||
|
Property and equipment, net |
|
118 |
|
219 |
| ||
|
Restricted cash |
|
200 |
|
— |
| ||
|
Other assets |
|
130 |
|
541 |
| ||
|
|
|
|
|
|
| ||
|
Total assets |
|
$ |
42,879 |
|
$ |
127,380 |
|
|
|
|
|
|
|
| ||
|
Liabilities, convertible preferred stock and stockholders’ (deficit) equity |
|
|
|
|
| ||
|
Current liabilities: |
|
|
|
|
| ||
|
Accounts payable |
|
$ |
605 |
|
$ |
3,170 |
|
|
Accrued expenses |
|
2,980 |
|
8,545 |
| ||
|
Other current liabilities |
|
11 |
|
3 |
| ||
|
|
|
|
|
|
| ||
|
Total current liabilities |
|
3,596 |
|
11,718 |
| ||
|
|
|
|
|
|
| ||
|
Other non-current liabilities |
|
3 |
|
— |
| ||
|
|
|
|
|
|
| ||
|
Commitments and contingencies |
|
|
|
|
| ||
|
|
|
|
|
|
| ||
|
Convertible preferred stock |
|
64,348 |
|
— |
| ||
|
|
|
|
|
|
| ||
|
Total stockholders’ (deficit) equity |
|
(25,068 |
) |
115,662 |
| ||
|
Total liabilities, convertible preferred stock and stockholders’ (deficit) equity |
|
$ |
42,879 |
|
$ |
127,380 |
|
This selected financial information should be read in conjunction with the consolidated financial statements and notes thereto included in TESARO’s prospectus filed with the Securities and Exchange Commission on June 29, 2012, which includes the audited financial statements for the year ended December 31, 2011, and the financial statements for the year ended December 31, 2012, which will be filed with TESARO’s Form 10-K for the year ended December 31, 2012.
For Further Information Contact:
Richard Rodgers
Executive Vice President & CFO
+1.339.970.0903
rrodgers@tesarobio.com