UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the fiscal year ended December 31, 2019 | |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
For the transition period from __________to __________
Commission file number 001-35887
(Exact name of registrant as specified in its charter)
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |
(Address of principal executive offices)
(Zip Code)
(770 ) 651-9100
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act: None.
Title of each class | Trading Symbol | Name of each exchange on which registered |
N/A | N/A | N/A |
Securities registered pursuant to Section 12(g) of the Act:
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes o No þ
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. Yes o No þ
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes o No þ
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§223.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ¨ No þ
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer | ☐ | ☑ | |
Non-accelerated filer | ☐ | Smaller reporting company | |
Emerging growth company | |||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No þ
The aggregate market value of the registrant’s voting common equity held by non-affiliates of the registrant as of June 28, 2019 (the last business day of the registrant’s most recently completed second quarter) was approximately $425.1 million based upon the last sale price ($4.05) of the shares as reported on the OTC Pink Market on such date.
There were 110,328,875 shares of the registrant’s common stock, par value $0.001 per share, outstanding as of June 25, 2020.
Documents Incorporated By Reference
None.
Table of Contents
Item | Description | Page |
Part I | ||
Item 1. | Business | |
Item 1A. | Risk Factors | |
Item 1B. | Unresolved Staff Comments | |
Item 2. | Properties | |
Item 3. | Legal Proceedings | |
Item 4. | Mine Safety Disclosures | |
Part II | ||
Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities | |
Item 6. | Selected Financial Data | |
Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations | |
Item 7A. | Quantitative and Qualitative Disclosures About Market Risk | |
Item 8. | Financial Statements and Supplementary Data | |
Item 9. | Changes in Disagreements with Accountants on Accounting and Financial Disclosure | |
Item 9A. | Controls and Procedures | |
Item 9B. | Other Information | |
Part III | ||
Item 10. | Directors, Executive Officers and Corporate Governance | |
Item 11. | Executive Compensation | |
Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | |
Item 13. | Certain Relationships and Related Transactions, and Director Independence | |
Item 14. | Principal Accounting Fees and Services | |
Part IV | ||
Item 15. | Exhibits, Financial Statement Schedules | |
Item 16. | Form 10-K Summary | |
Signatures | ||
PART I
EXPLANATORY NOTE
As used herein, the terms “MiMedx,” “the Company,” “we,” “our” and “us” refer to MiMedx Group, Inc., a Florida corporation, and its consolidated subsidiaries as a combined entity, except where it is clear that the terms mean only MiMedx Group, Inc.
Prior Investigation and Restatement
In February 2018, the Audit Committee (the “Audit Committee”) of the Company’s Board of Directors (the “Board”) retained King & Spalding LLP (“King & Spalding”) as counsel to the Audit Committee to assist in conducting an independent investigation into current and prior-period matters relating to allegations regarding certain sales and distribution practices at the Company and certain other matters (the “Investigation” or the “Audit Committee Investigation”). The Investigation focused primarily on the following areas: (1) the Company’s revenue recognition practices; (2) revenue management activities; (3) actions taken against whistleblowers; (4) tone set by former senior management and (5) Anti-Kickback Statute and related allegations.
In a Form 8-K dated June 6, 2018, we disclosed that our Audit Committee, with the concurrence of management, concluded that the Company’s previously issued consolidated financial statements and financial information relating to each of the fiscal years ended December 31, 2016, 2015, 2014, 2013 and 2012 and each of the interim periods within such years, along with the unaudited condensed consolidated financial statements included in the Company’s Quarterly Reports on Form 10-Q for the quarters ended March 31, 2017, June 30, 2017 and September 30, 2017 (collectively, the “Non-Reliance Periods”), would need to be restated under United States generally accepted accounting principles (“GAAP”) and could no longer be relied upon.
Our annual report on Form 10-K for the year ended December 31, 2018 (the “2018 Form 10-K”), filed on March 17, 2020, included our audited consolidated balance sheets, consolidated statements of operations, stockholders’ equity and cash flows as of and for the years ended December 31, 2018 and 2017, which had not previously been filed, and for the year ended December 31, 2016, which were restated from the consolidated financial statements previously filed in our Annual Report on Form 10-K for the year ended December 31, 2016, as well as selected unaudited condensed consolidated financial data as of and for the years ended December 31, 2015 (Restated) and 2014 (Restated), which reflected adjustments to our previously filed consolidated financial statements as of and for the years ended December 31, 2015 and 2014 (collectively, the “Restatement”). Refer to Item 6, “Selected Financial Data” of our 2018 Form 10-K for information regarding the applicable adjustments or restatements of our financial results for 2016, 2015 and 2014.
Forward-Looking Statements
This Form 10-K contains forward-looking statements. All statements relating to events or results that may occur in the future are forward-looking statements, including, without limitation, statements regarding the following:
• | our strategic focus, as illustrated by our strategic priorities and our ability to implement these priorities; |
• | our ability to access capital sufficient to implement our strategic priorities; |
• | our expectations regarding our ability to fund our ongoing and future operating costs; |
• | our expectations regarding future income tax liability; |
• | the advantages of our products and development of new products; |
• | market opportunities for our products; |
• | the regulatory pathway for our products, including our existing and planned investigative new drug application and pre-market approval requirements, the design and success of our clinical trials and pursuit of Biological License Applications (“BLAs”) for certain products; |
• | our expectations regarding our ability to manufacture certain of our products in compliance with current Good Manufacturing Practices (“cGMP”); |
• | our expectations regarding costs relating to compliance with regulatory standards, including those arising from our clinical trials, pursuit of BLAs, and cGMP compliance; |
• | our ability to continue marketing our micronized products and certain other products during and following the end of the period of enforcement discretion announced by the United States Food and Drug Administration (“FDA”); |
4
• | expectations regarding government and other third-party coverage and reimbursement for our products; |
• | expectations regarding future revenue growth; |
• | our belief in the sufficiency of our intellectual property rights in our technology; |
• | our ability to procure sufficient supplies of human tissue to manufacture and process our products; |
• | the outcome of pending litigation and investigations; |
• | our ability to complete remedial actions to address all observations in the Forms FDA 483 issued to us by the FDA; |
• | our ability to regain and remain in compliance with Securities and Exchange Commission (the "SEC") reporting obligations; |
• | our ability to relist our common stock, par value $0.001 per share (the “Common Stock”) on The Nasdaq Capital Market; |
• | ongoing and future effects arising from the Audit Committee Investigation, the Restatement, and related litigation; |
• | ongoing and future effects arising from the COVID-19 pandemic, and our responses intended to mitigate such effects; |
• | demographic and market trends; |
• | our plans to remediate the identified material weaknesses in our internal control environment and to strengthen our internal control environment; |
• | our expectations regarding research and development costs, including those arising from filing additional investigative new drug applications and pursuing new BLAs; |
• | our expectations regarding the cost savings and effects resulting from reductions in employee salaries; and |
• | our ability to compete effectively. |
Forward-looking statements generally can be identified by words such as “expect,” “will,” “change,” “intend,” “seek,” “target,” “future,” “plan,” “continue,” “potential,” “possible,” “could,” “estimate,” “may,” “anticipate,” “to be” and similar expressions. These statements are based on numerous assumptions and involve known and unknown risks, uncertainties and other factors that could significantly affect the Company’s operations and may cause the Company’s actual actions, results, financial condition, performance or achievements to differ materially from any future actions, results, financial condition, performance or achievements expressed or implied by any such forward-looking statements. Factors that may cause such a difference include, without limitation, those discussed under the heading “Risk Factors” in this Form 10-K.
Unless required by law, the Company does not intend, and undertakes no obligation, to update or publicly release any revision to any forward-looking statements, whether as a result of the receipt of new information, the occurrence of subsequent events, a change in circumstances or otherwise. Each forward-looking statement contained in this Form 10-K is specifically qualified in its entirety by the aforementioned factors. Readers are advised to carefully read this Form 10-K in conjunction with the important disclaimers set forth above prior to reaching any conclusions or making any investment decisions and not to place undue reliance on forward-looking statements, which apply only as of the date of the filing of this Form 10-K with the SEC.
5
Item 1. Business
Overview
MiMedx is an industry leader in advanced wound care and an emerging therapeutic biologics company, developing and distributing placental tissue allografts with patent-protected processes for multiple sectors of healthcare. We derive our products from human placental tissues processed using our proprietary processing methodologies, including the PURION® process. We employ aseptic processing techniques in addition to terminal sterilization to produce our allografts. MiMedx provides products in the wound care, burn, surgical, orthopedic, spine, sports medicine, ophthalmic, and dental sectors of healthcare. Our mission is to offer products and tissues to help the body heal itself. All of our products are regulated by the FDA.
MiMedx is the leading supplier of human placental allografts, which are human tissues that are transplanted from one person (a donor) to another person (a recipient). MiMedx has supplied over 1.9 million allografts, through both direct and consignment shipments. Our biomaterial platform technologies include AmnioFix®, EpiFix®, EpiCord®, AmnioCord® and AmnioFill®. AmnioFix and EpiFix are our tissue allografts derived from the amnion and chorion layers of the human placental membrane. EpiCord and AmnioCord are tissue allografts derived from umbilical cord tissue. AmnioFill is a placental connective tissue matrix, derived from the placental disc and other placental tissue.
Our EpiFix and EpiCord product lines are promoted for external use, such as in advanced wound care applications, while our AmnioFix, AmnioCord and AmnioFill products are positioned for use in surgical applications, including lower extremity repair, plastic surgery, vascular surgery and multiple orthopedic repairs and reconstructions. We describe these in greater detail below under the heading “Our Product Portfolio.”
2017 FDA Guidance. The products we sell are regulated by the FDA. Historically, we marketed our products as Human Cells, Tissues and Cellular and Tissue – Based Products (“HCT/Ps”), which do not require pre-market clearance or approval by the FDA and are subject solely to Section 361 of the Public Health Service Act (“Section 361”) and related regulations. However, in November 2017 the FDA published a series of related guidances, including one entitled “Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue–Based Products: Minimal Manipulation and Homologous Use – Guidance for Industry and Food and Drug Administration Staff” (the “Guidance”), that established an updated framework for the FDA’s regulation of cellular and tissue-based products. Among other things, the guidances clarified the FDA’s views about the criteria that differentiate those products subject to regulation solely under Section 361 (“Section 361 HCT/Ps”) from those cellular and tissue-based products that are considered to be drugs and biological products (“Section 351 HCT/Ps”) subject to licensure under Section 351 of the Public Health Service Act (“Section 351”) and related regulations. As described below and elsewhere in this Form 10-K, the guidances clarified the FDA’s expectation that certain products such as those that MiMedx has long marketed as Section 361 HCT/Ps will be treated as Section 351 HCT/Ps moving forward. The Guidance also confirmed that amniotic membrane in sheet form generally can be characterized as “minimally manipulated” and therefore regulated solely under Section 361.
Effect on Our Products. Under the Guidance, we expect that the FDA will continue to regulate our amniotic membrane sheet products (AmnioFix, EpiFix, EpiBurn and EpiXL) and umbilical cord products (EpiCord and AmnioCord) as Section 361 HCT/Ps so long as the claims we make for them are consistent with the Section 361 framework. We expect, however, that the FDA will regulate certain of our other products, such as our micronized products (AmnioFix Injectable and EpiFix Micronized) under Section 351 as biological products. We also expect other products, like AmnioFill, will be regulated as biological products under the Section 351 regulations.
Enforcement Discretion. The Guidance stated that the FDA intends to exercise enforcement discretion under limited conditions with respect to the investigative new drug (“IND”) application and pre-market approval requirements for certain HCT/Ps through November 2020. This means that, through November 2020, the FDA does not intend to enforce certain provisions as they currently apply to certain entities or activities. The FDA intended this period of enforcement discretion to give sponsors time to evaluate their products, have a dialogue with the agency and, if necessary, begin clinical trials and file the appropriate pre-market applications to transition products that had been marketed as Section 361 HCT/Ps into compliance with Section 351. The FDA’s approach is risk-based, and the Guidance clarified that high-risk products and uses might be subject to immediate enforcement action.
During the Period of Enforcement Discretion. We have continued to market our micronized products under this policy of enforcement discretion, while at the same time pursuing a Biologics License Application (“BLA”) for certain of our micronized products.
We have already filed INDs for three indications for our micronized product, AmnioFix injectable: plantar fasciitis, osteoarthritis knee pain, and Achilles tendonitis and have been conducting clinical trials. We also intend to file additional INDs for both AmnioFill and for injectable micronized EpiFix in the second half of 2020, but have not yet initiated any clinical trials under an IND in furtherance of any regulatory approvals for these indications. Further, as we previously announced, we will need more time than
6
we originally anticipated to file our BLAs with the FDA, and clinical trial protocol amendments and enhancements, further resources, and additional capabilities and expertise will be required. See “Clinical Trials” below for information.
We have also begun investing in additional plant and equipment and compliance personnel to allow us to manufacture and market in accordance with Section 351 requirements at scale. Among other things, this required us to make capital expenditures in 2019 which have continued in 2020. See discussion below – “Risk Factors” under the heading “If any of the BLAs are approved, the Company would be subject to additional regulation which will increase costs and results in adverse sanctions for non-compliance.”
Efforts to Seek Extension of Enforcement Discretion Period. MiMedx has provided a recommendation to the FDA that the FDA extend its enforcement discretion period beyond November 2020 to allow for the continued marketing of the impacted products in accordance with an agreed upon transition plan. However, there is no guarantee that the FDA will grant an extension, and even if issued, such an extension may be limited to the products and indications that are subject to clinical trials. See discussion below - “Risk Factors” under the heading “To the extent our products do not qualify for regulation as human cells, tissues and cellular and tissue-based products solely under Section 361 of the Public Health Service Act, this could result in removal of the applicable products from the market, would make the introduction of new tissue products more expensive, and would significantly delay the expansion of our tissue product offerings and subject us to additional post-market regulatory requirements.”
Post-Enforcement Discretion. Following the period of enforcement discretion, we may need to cease selling our micronized products and other products regulated under Section 351 until the FDA approves a BLA, and then we will only be able to market such products for indications that have been approved in a BLA. The loss of our ability to market and sell our micronized products would have a material adverse impact on our revenues, earnings and financial position. In addition, we expect the cost to manufacture our products will increase due to the costs to comply with the requirements that apply to Section 351 biological products such as cGMPs and ongoing product testing costs. See discussion below – “Risk Factors” under the heading “To the extent our products do not qualify for regulation as human cells, tissues and cellular and tissue-based products solely under Section 361 of the Public Health Service Act, this could result in removal of the applicable products from the market, would make the introduction of new tissue products more expensive, and would significantly delay the expansion of our tissue product offerings and subject us to additional post-market regulatory requirements.”
The majority of our revenues are generated by wound care applications. We intend to sharpen our focus in advanced wound care, continue developing and expanding our product pipeline, and work toward continued operational excellence to support future growth and sustained productivity. This includes focusing on effective and efficient execution in our core advanced wound care business and maximizing clinical adoption. In the second half of 2020, we plan to continue executing our commercial strategy, bring our manufacturing and quality systems toward compliance with the requirements that apply to Section 351 biological products, and continue pursuing a dialogue with the FDA in advance of the end of the period of enforcement discretion. The Company is advancing its therapeutic biologics pipeline targeting specific FDA-approved clinical indications for the treatment of musculoskeletal degeneration and other areas of unmet clinical need. See the discussion below – “Clinical Trials” for more information.
Our History
Our current business began on February 8, 2008 when Alynx, Co., our predecessor company, acquired MiMedx, Inc., a development-stage medical device company, the assets of which included licenses to two development-stage medical device technology platforms, which are regulated under Section 361 and we do not currently market. On March 31, 2008, Alynx, Co. merged into MiMedx Group, Inc., a Florida corporation and wholly-owned subsidiary that had been formed for purposes of the merger, with MiMedx Group, Inc. (the “Company”) as the surviving corporation in the merger. In January 2011, the Company acquired all of the outstanding equity interests of Surgical Biologics, LLC (n/k/a MiMedx Tissue Services, LLC).
Recent Developments
Delisting of Common Stock and Related Matters
Due to our failure to remain current in our reporting obligations under SEC requirements, The Nasdaq Stock Market LLC (“Nasdaq”) suspended our common stock (“Common Stock”) from trading on The Nasdaq Capital Market on November 8, 2018, and subsequently delisted our Common Stock effective March 8, 2019. We are in the process of applying to relist our Common Stock after we become current with respect to our SEC reporting obligations, but we cannot guarantee when, or if, we will be able to relist our Common Stock.
7
Leadership Changes to Our Management and Board of Directors
Since June 2018, most of our executive leadership team has changed. The Board appointed Timothy R. Wright as Chief Executive Officer, effective as of May 13, 2019. In December 2019, William “Butch” Hulse joined the Company as General Counsel and Secretary. Effective March 18, 2020, the Board appointed Peter M. Carlson as Chief Financial Officer, and on May 1, 2020 the Company appointed William L. Phelan as Chief Accounting Officer.
Charles R. Evans, the Company’s lead director, was appointed Chairman of the Board on July 2, 2018. In June 2019, Dr. M. Kathleen Behrens succeeded him as Chair of the Board.
The Board is in the process of executing a plan to refresh the composition of the Board while providing important business oversight and leadership continuity. The Board is currently comprised of nine directors elected by holders of Company Common Stock, five of whom have joined the Board since June 2019. In addition, the Board agreed to nominate a mutually-agreed candidate with Prescience Partners, LP, a Delaware limited partnership (“Prescience Partners”), for election as a Class III director at the upcoming 2019 annual meeting of shareholders (the “2019 Annual Meeting”) to succeed one of our incumbent directors. Pursuant to the Preferred Stock Transaction described below, the Company increased the size of the Board of Directors, and Martin P. Sutter and William A. Hawkins III were appointed to serve as Preferred Directors effective July 2, 2020. As a result, following the 2019 Annual Meeting, eight of our eleven directors will be new to the Board since June 2019.
Financing Transactions
In June 2019, the Company secured $75 million of debt financing from Blue Torch Finance LLC (“Blue Torch”). Effective April 22, 2020, the Company amended its loan agreement with Blue Torch (the “BT Loan Agreement”) to (1) relax the Total Leverage Ratio (as defined in the BT Loan Agreement) covenant, which is a quarterly test, from a maximum Total Leverage Ratio of 3.00 to 1.00 to 5.00 to 1.00 for the quarterly periods ending on June 30, 2020, September 30, 2020, and December 31, 2020; and (2) to reduce the minimum Liquidity (as defined in the BT Loan Agreement) covenant, which is a monthly requirement, from $40 million to $20 million for April and May 2020 and from $30 million to $20 million for June through November 2020.
On July 2, 2020, the Company issued $100 million of the Company’s Series B Convertible Preferred Stock, par value $0.001 per share (the “Series B Preferred Stock”), to an affiliate of EW Healthcare Partners and to certain funds managed by Hayfin Capital Management LLP pursuant to the Securities Purchase Agreement, dated as of June 30, 2020 (the “Securities Purchase Agreement”), for an aggregate purchase price of $100,000,000 (the “Preferred Stock Transaction”). On July 2, 2020, the Company also borrowed an aggregate of $50 million pursuant to the loan agreement, dated as of June 30, 2020 (the “Hayfin Loan Agreement”), by and among the Company, certain of the Company’s subsidiaries, Hayfin Services LLP and other funds managed by Hayfin Capital Management LLP and has obtained an additional committed but undrawn $25 million facility pursuant to the Hayfin Loan Agreement (collectively, the “Hayfin Loan Transaction”). A portion of the proceeds from these transactions was used to repay the outstanding balance of principal and accrued but unpaid interest, and repayment premium, under the BT Loan Agreement. For further information regarding the Preferred Stock Transaction, the Hayfin Loan Transaction and the repayment and termination of the BT Loan Agreement, see Item 9B, “Other Information.”
Government Investigations Update
On November 26, 2019, the Company announced that it finalized a settlement with the SEC resolving a previously disclosed investigation into the Company's financial accounting practices. The Company agreed to settle with the SEC, without admitting or denying the SEC's allegations, by consenting to the entry of a final judgment that permanently restrains and enjoins the Company from violating certain provisions of the federal securities laws. As part of the settlement, the Company paid a $1.5 million civil penalty. The SEC recognized the Company's cooperation during the investigation, as well as its remedial efforts.
On April 6, 2020, the Company announced that it had finalized a settlement with the Department of Justice (the “DOJ”), resolving an investigation concerning the accuracy of commercial pricing disclosures to the United States Department of Veterans Affairs (the “VA”) for one of the Company’s products in connection with the Company’s Federal Supply Schedule contract, and a related qui tam action filed in Minnesota. The Company self-disclosed the matter to the VA Office of Inspector General (VA-OIG) in November 2018, prior to its knowledge of the qui tam suit or any underlying government investigation and, as the DOJ acknowledged in the settlement agreement, the Company cooperated with the government’s investigation into the matter. Without admitting the allegations, the Company agreed to pay $6.5 million to the DOJ to resolve the matter. The Company previously disclosed that it had accrued an amount to cover the settlement and anticipated related expenses in its annual report on Form 10-K for the year ended December 31, 2018.
8
Current Business Priorities
Advanced wound care includes products or procedures used in the treatment of acute and chronic wounds. These products or procedures are used when standard wound care has failed, or after 4 weeks of non-healing. The advanced wound care category is expected to continue growing due to certain demographic trends, including an aging population, increasing incidence of obesity and diabetes, and the associated higher susceptibility to non-healing chronic wounds. Furthermore, the increasing number of patients requiring advanced treatment represents a significant cost burden on the healthcare system. After evaluating the potential impact of this data on the Company’s wound care franchise, we incorporated a strategy not only to participate in this market growth but also to increase the Company’s market share by demonstrating the positive health economics of our products.
Our priorities include sharpening our focus in advanced wound care, developing and expanding our portfolio pipeline and driving continued operational excellence to support future growth and sustained productivity, with the following elements:
Focus on effective and efficient execution in our core advanced wound care business, maximizing clinical adoption and health economics value.
We have identified and are in the final process of aligning new sales territories to focus our sales force and drive efficiencies, enabling the MiMedx field personnel and sales infrastructure to enhance productivity and better serve our customers and patients. We are advancing additional health economics outcomes data to further support the use of EpiFix and have expanded efforts to best position EpiCord within the treatment paradigm, capitalizing on expanded product coverage throughout our leading technology portfolio.
Enhance business development efforts, driving growth throughout the Company’s existing product portfolio pipeline and strategic adjacencies to create a long-term competitive advantage.
Our long-range planning identified opportunities for innovative pipeline growth and international regulatory and product coverage expansion within targeted high growth geographies. Additionally, an ongoing assessment of the Company’s development programs has highlighted the need for greater cross-functional collaboration and increased investment. We continue to evaluate these opportunities in alignment with our focus on advanced wound care. We remain focused on advancing our BLA programs and are therefore aligning voice-of-customer input, industry expertise and additional resources toward seeking FDA approval for micronized dehydrated human amnion/chorion membrane (“dHACM”) for a potential indication to treat musculoskeletal degeneration across multiple indications.
Enable operational and organizational excellence to support future growth and sustained productivity.
In December 2018, we announced the launch of a broad-based organizational realignment, cost reduction and efficiency program to better ensure the Company’s cost structure was appropriate given its overall lower revenue expectations. This program included management changes, a realignment of the Company’s sales force, reductions in non-employee expenses and certain changes to our business practices in response to the Investigation. The program created business efficiencies supportive of sustained, achievable and independent growth. Since enactment through December 31, 2019, the Company has realized cost savings of approximately $37 million associated with the realignment program. Additionally, management has continued its efforts to position the business for long-term success. As part of our effort to continue to improve our sales force effectiveness, the Company has prioritized the alignment of various market access functions across the organization under one business functional area. This is aimed toward aligning with providers and patients where our payer coverage, reimbursement and Group Purchasing Organization (“GPO”) and Integrated Delivery Network (“IDN”) contract opportunities exist.
We have re-focused our priorities on refining our near-term approach for our business and our products following the end of the enforcement discretion period, bringing our manufacturing and quality systems toward compliance with the requirements that apply to Section 351 biological products, and advancing our commercial initiatives focused on building market awareness.
Our Product Portfolio
We sell our placenta-based allograft products under our own brands and, on a limited basis, through a private label or original equipment manufacturer (“OEM”) basis. We maintain strict controls on quality at each step of the manufacturing process beginning at the time of procurement. Our Quality Management System has long been focused on compliance with the American Association of Tissue Banks’ (“AATB”) standards and the FDA’s current Good Tissue Practices (“cGTP”), and we are strengthening our controls for future BLA products through development of our current Good Manufacturing Practices (“cGMP”) program.
9
EpiFix
Our EpiFix allograft is a semi-permeable protective barrier membrane product comprised of dehydrated human amnion/chorion membrane that may be used in the treatment of chronic wounds, including diabetic foot ulcers (“DFUs”), venous leg ulcers (“VLUs”), arterial ulcers, pressure ulcers and burns. EpiFix is available in a variety of sizes that can be used appropriately for wounds of varying sizes.
MiMedx also has a micronized version of this product. As further discussed below under the heading “Government Regulation -Recent FDA Guidance and Transition Policy for HCT/Ps,” the FDA clarified in its 2017 guidance that it regards micronized amniotic membrane products as being subject to FDA licensure as biological products under Section 351. We intend to file an IND for EpiFix micronized in the second half of 2020 for potential application in DFUs or other areas of advanced wound care, but have not yet initiated any clinical trials under an IND in furtherance of any regulatory approvals.
AmnioFix
Our AmnioFix allograft is a semi-permeable protective barrier membrane product comprised of dehydrated human amnion/chorion membrane that may be used in the treatment of wounds related to surgical procedures. AmnioFix is configured in a variety of sizes, appropriate for various applications for internal use. Currently, our AmnioFix product line consists of two main configurations, including AmnioFix sheet and AmnioFix Injectable:
• | AmnioFix sheet form is used in a variety of surgical wound repair and internal surgical procedures. It is primarily used in lower extremity repair, spine, orthopedic, sports medicine, gastrointestinal, urologic, and other general surgery applications. |
• | AmnioFix Injectable is supplied in micronized powder form and is reconstituted with 0.9% sterile saline for injection. This product is our lead BLA candidate. We are studying the product’s potential to address musculoskeletal degeneration across multiple indications. We have three clinical studies underway to support INDs: plantar fasciitis, Achilles tendonitis and knee osteoarthritis. We currently are in Phase 3 for plantar fasciitis and Achilles tendonitis and in Phase 2B for knee osteoarthritis. |
EpiCord and AmnioCord
EpiCord and AmnioCord are dehydrated human umbilical cord allografts intended for homologous use. EpiCord and AmnioCord provide a protective environment for the healing process and are used in the treatment of wounds or in surgical procedures. Our cord products are thicker than the EpiFix or AmnioFix allografts and have application in deeper wounds or in areas where suturing the allograft in place may be advantageous.
AmnioFill
AmnioFill is a connective tissue matrix derived from placental disc, umbilical cord, and amnion/chorion tissues. It is used to replace or supplement damaged integumental tissue. Its primary application is in larger and uneven wound surfaces, or deep/tunneling wounds including pressure ulcers. We intend to file an IND for AmnioFill in the second half of 2020. However, we have not yet initiated any clinical trials under an IND in furtherance of any regulatory approvals for AmnioFill.
OEM Products
We sell a selection of allografts for dental applications on an OEM basis pursuant to an agreement under which we have granted a third party an exclusive license to some of our technology for use in dental applications. Other than dental applications, we have a limited number of OEM relationships.
We continue to research new opportunities for amniotic and other placental tissue, and we have several additional offerings in various stages of conceptualization and development.
Placenta Donation Program
We partner with physicians and hospitals to recover donated placental tissue. Through our donor program, a mother who delivers a healthy baby via a Caesarean section can donate her placenta and umbilical cord tissue in lieu of having it discarded as medical waste. After consent for donation is obtained, a blood sample from each donor is tested for communicable diseases, and the donor is screened for risk factors in order to determine eligibility in compliance with federal regulations and AATB standards. We operate a licensed tissue bank that is registered as a tissue establishment with the FDA, and we are an accredited member of the AATB. All donor records and test results are reviewed by our Medical Director and staff prior to the release of the tissue for distribution.
10
We have developed a large network of hospitals that participate in our placenta donation program, and we employ a dedicated staff that work with these hospitals. We also utilize third-party providers of placenta donations to mitigate risks. We believe that we will be able to obtain an adequate supply of tissue to meet anticipated demand. However, see discussion below “Risk Factors” under the heading “Our products depend on the availability of tissue from human donors, and any disruption in supply could adversely affect our business.”
Processing (Manufacturing)
Over several years, we have developed and patented a unique and proprietary technique (PURION) for processing allografts from the donated placental tissue. This technique specifically focuses on preserving the tissue’s natural growth factor content and maintaining the structure and collagen matrix of the tissue. Our patented and proprietary processing method employs aseptic processing techniques in addition to terminal sterilization for increased patient safety. We believe that our process preserves more of the natural characteristics of the tissue than the processes used by many of our competitors.
The PURION process produces an allograft that retains the tissue’s inherent biological properties (cytokines, chemokines, growth factors, etc.) found in the placental tissue and produces an allograft that is easy for healthcare providers to use. The allograft can be stored at ambient temperature and has a five-year shelf life. Each sheet allograft incorporates specialized visual embossments that assist the health care practitioner with proper allograft placement and orientation.
To ensure the safety of human tissue products, the FDA enforces current Good Tissue Practice (“cGTP”) manufacturing regulations. We believe that MiMedx has developed mature systems to comply with, and is in compliance with, these regulations. As an important part of the Company’s product safety compliance, MiMedx products are terminally sterilized to an internationally recognized industry standard in addition to having been processed via the PURION process.
Our facilities are subject to periodic unannounced inspections by regulatory authorities and may undergo compliance inspections conducted by the FDA and corresponding state and foreign agencies. We are registered with the FDA as a tissue establishment and are subject to the FDA’s cGTP quality program regulations, state regulations and regulations promulgated by various regulatory authorities outside the United States. The Company’s most recent FDA inspection for compliance with GTP regulations, which took place in September 2018, resulted in no observations and a no action indicated (NAI) rating, which is the most favorable designation the FDA provides after an inspection.
In recent years, the FDA has clarified through inspection activity, letters to industry, and guidance documents its expectation that certain human tissue products, including product types manufactured by MiMedx, meet additional requirements that apply to traditional biological products, such as BLA approval and cGMP compliance beginning in November 2020. The guidance documents apply to products offered by many companies, not just MiMedx, and the guidance has implications for manufacturing processes, among other things. For example, the FDA generally requires products subject to Section 351 to be manufactured in compliance with cGMPs. After the end of the enforcement discretion period, these products will be subject to cGMP compliance. The Company is developing and enhancing systems to meet these requirements, and intends to complete those efforts by November 2020, although there is no guarantee that the Company will be able to meet the requirements by such date, or at all. In December 2019, the FDA conducted cGMP inspections at our Marietta, Georgia and Kennesaw, Georgia processing facilities. The FDA issued a Form FDA 483 (“483”), which is a list of inspectional observations, at the conclusion of each inspection. Specifically, the FDA issued a 483 consisting of 9 observations at our Marietta, Georgia processing facility, and a 483 consisting of 14 observations at our Kennesaw, Georgia processing facility. MiMedx timely responded to the FDA regarding each observation, providing substantive responses to all of the observations. The Company’s response included completed and planned actions to address each observation, and as of the date of this filing, all of these remedial actions are now complete.
Intellectual Property
Our intellectual property includes owned and licensed patents, owned and licensed patent applications and patents pending, proprietary manufacturing processes and trade secrets, and trademarks associated with our technology. We believe that our patents, proprietary manufacturing processes, trade secrets, trademarks, and technology licensing rights provide us with important competitive advantages.
11
Patents and Patent Applications
Due to the substantial expertise and investment of time, effort and financial resources required to bring new regenerative biomaterial products and implants to the market, the importance of obtaining and maintaining patent protection for significant new technologies, products and processes cannot be underestimated. As of the date of the filing of this Form 10-K, in addition to international patents and patent applications, we own 52 U.S. patents related to our amniotic tissue technology and products, and 32 additional patent applications covering aspects of this technology are pending at the United States Patent and Trademark Office. The vast majority of our domestic patents covering our core amniotic tissue technology and products will not begin to expire until August 2027. See discussion below – “Risk Factors” under the heading “Risks Related to Our Intellectual Property.”
Market Overview
Domestic sales currently account for substantially all of our revenue, and we are considering international expansion, primarily targeting Europe and Asia Pacific. In the United States, advanced wound care, including burns and lower extremity surgical applications, are our primary applications.
Wound Care
The broad wound care category includes traditional dressings such as bandages, gauzes and ointments, which are used to treat non-severe or non-chronic wounds, and advanced wound care products such as mechanical devices, advanced dressings, xenografts, biological products, and HCT/Ps, which are used to treat severe wounds or chronic wounds that have not appropriately closed after four weeks of treatment with traditional dressings.
In the United States in 2018, third-party estimates indicate that there were 8.2 million total reported wounds, with 2.9 million of these wounds classified as chronic wounds. Of these chronic wounds, we estimate that 35% are candidates for advanced skin substitute product treatment regimens, providing for a total addressable opportunity of approximately $3.3 billion. The overall cost of treating chronic wounds is rising sharply, and the current annual estimated cost in the United States exceeds $28 billion.
MiMedx is a leader in the advanced wound care category and the amniotic tissue allograft sub-category. Both of these categories are expected to continue growing due to certain demographic trends, including an aging population, increasing incidence of obesity and diabetes and the associated higher susceptibility to non-healing chronic wounds. Furthermore, the increasing number of patients requiring advanced treatment represents a significant cost burden on the healthcare system.
Traditional dressings such as bandages, gauzes and ointments, along with treatment of active infection and debridement, currently represent the “standard of care” for treating chronic wounds such as DFUs, VLUs, pressure ulcers and arterial ulcers. If after four weeks of use, the wound has not responded appropriately to “standard of care” therapy, clinical research has shown that advanced therapy such as a skin and dermal substitute can be beneficial as part of the patient’s treatment plan. However, often times advanced therapies are not employed - this represents a large target market for the Company and one of the drivers for the growth of the advanced therapy market. According to data provided by BioMedGPS, MiMedx’s EpiFix is the current product of choice for physicians choosing to use a skin and dermal substitute product as a barrier or cover. EpiFix stores at ambient conditions for up to five years compared to certain cultured skin substitutes currently on the market that require cryogenic freezer storage and expire within days to months from the time of processing. In addition, we market multiple sizes of EpiFix sheets for use as protective barriers which enables a healthcare provider to select an appropriate size graft based on the size of the wound to reduce product waste.
Our AmnioFix tissue allografts have been used in a variety of surgical applications including, but not limited to, plastic surgery, general surgery, gynecology, urology, orthopedics, spinal surgery, lower extremity repair and sports medicine. AmnioFix can be used as a barrier membrane in procedures where a second surgery may be required and scar tissue formation may be problematic.
Biologics License Application (BLA) Programs
The FDA clarified its expectations in late 2017 that certain cellular and tissue-based products, including types of products marketed by MiMedx, are considered drugs and biological products subject to Section 351 requirements under the federal Food, Drug and Cosmetic Act (the “FD&C Act”). In order to conform to this regulatory guidance, MiMedx is pursuing several indications under the BLA pathway, although there can be no assurance that we will obtain a BLA and may ultimately decide not to pursue a BLA for certain products or indications. See Risk Factors - “Obtaining and maintaining the necessary regulatory approvals for certain of our products will be expensive and time consuming and may impede our ability to fully exploit our technologies.” AmnioFix Injectable is our lead BLA product candidate, and we are studying its potential to address a number of degenerative musculoskeletal conditions. In this regard, we have three ongoing IND programs: plantar fasciitis, Achilles tendonitis and knee osteoarthritis. We are currently completing a Phase 3 plantar fasciitis study and are well advanced in the enrollment of subjects in a Phase 2B knee
12
osteoarthritis study. Results of double-blinded, randomized, interim analyses of these studies revealed separation between treatment and control groups, but indicated that the power to observe a result with statistical and clinical significance could be increased by increasing the sample size. We have since amended the protocols and have taken other steps to improve these trials. We also have completed subject enrollment in a Phase 3 IND study for Achilles tendonitis, and we plan to review our options for this program after we have assessed the results of this study. However, an interim analysis of this study indicated that the sample size needed to be significantly increased to provide sufficient statistical and clinical significance. We have decided not to increase the size of the study, but have chosen to continue it to completion with the original sample size as we evaluate the study endpoints for appropriateness, including appropriateness of the measures and the time required to measure differences between the treatment groups (e.g., three months, six months, etc.). In addition, we have begun efforts to file an IND for AmnioFill in the second half of 2020, although we have not yet initiated any clinical trials under an IND in furtherance of any regulatory approvals for this product. Similar activities have also been initiated toward the filing of an IND for injectable micronized EpiFix for the treatment of DFUs or other areas of advanced wound care in the second half of 2020. Clinical study initiation will depend on FDA feedback for both of these programs. Given the timelines of these proposed filings and anticipated delays at FDA in processing applications due to the COVID-19 pandemic, it is likely that studies will not begin enrollment in 2020.
We are studying AmnioFix Injectable for a variety of uses other than wound care, and the applications described above (plantar fasciitis, osteoarthritis knee pain, and Achilles tendonitis) address unmet needs outside of traditional wound care. After oral non-habit forming pain medication fails to adequately relieve a patient’s joint, ligament or tendon pain, market available injections such as corticosteroids are a commonly available treatment option. However, a number of patients still do not get adequate relief from corticosteroid injections, or do not want to use corticosteroids given their potential to damage human tissue. Additionally, in light of the current crisis with opioid abuse, non-surgical treatments and alternative approaches to musculoskeletal pain management are under consideration. Patients and physicians are searching for new products that are safe and effective for the management of chronic musculoskeletal conditions. According to data from the National Health Interview Survey 2007-2008, it was estimated that 14 million people in the U.S. have symptomatic knee osteoarthritis, with more than half under the age of 65. We are studying AmnioFix Injectable as a potential product candidate to address this unmet need, as well as in other degenerative musculoskeletal applications. As of the date of the filing of this Form 10-K, it has not been approved by the FDA for any such use.
Marketing and Sales
Our direct sales force focuses on the advanced wound care category through multiple sites of service. We also maintain a network of independent sales agents that focus on musculoskeletal applications leveraging the complementary products in their portfolios, access to certain customers, and to provide sales coverage for areas where we do not have a full time sales representative.
We also sell our products through distributors. Distributors purchase products from us at wholesale prices and resell products to end users. Sales through distributors comprised a smaller percentage of our total sales in 2019 than in prior years. See Note 17, “Revenue Data by Customer Type.” As discussed above, we sell allografts for dental applications on an OEM basis pursuant to an agreement under which we granted a third party an exclusive license to some of our technology for use in certain fields in a specified field of use. We also sell our amnion/chorion and umbilical tissue products through a variety of agents for use in additional musculoskeletal applications on a non-exclusive basis.
Coverage and Reimbursement
With the exception of government accounts, most purchasers of our products are physicians, hospitals or ambulatory surgery centers (“ASCs”) that rely on reimbursement by third-party payers. Accordingly, our growth substantially depends on adequate levels of third-party reimbursement for our products from these payers. Third-party payers are sensitive to the cost of products and services and are increasingly seeking to implement cost containment measures to control, restrict access to, or influence the purchase of health care products and services. In the U.S., such payers include U.S. federal healthcare programs (e.g., Medicare and Medicaid), private insurance plans, managed care programs and workers’ compensation plans. Federal healthcare programs have prescribed coverage criteria and reimbursement rates for medical products, services and procedures. Similarly, private, third-party payers have their own coverage criteria and negotiate reimbursement amounts for medical products, services and procedures with providers. In addition, in the U.S., an increasing percentage of insured individuals are receiving their medical care through managed care programs (including managed federal healthcare programs) which monitor and may require pre-approval of the products and services that a member receives. Ultimately, however, each third-party payer determines whether and on what conditions they will provide coverage for our products, and such decisions often include each payer’s assessment of the science and efficacy of the applicable product.
A significant portion of our products are purchased by U.S. government accounts (e.g., the VA, the Public Health Service (including the Indian Health Service)), which do not depend on reimbursement from third party payers. In order for a company to be eligible to have its products purchased by such federal agencies and paid for by the Medicaid program, federal law requires the Company
13
to participate in the VA Federal Supply Schedule (“FSS”) pricing program. To participate, we are required to enter into a Master Agreement with the VA for our products and agree to certain prices.
EpiFix Sheet Products and EpiCord
Medicare Coverage
By far, the largest third-party payer in the United States is the Medicare program, which is a federally-funded program that provides healthcare coverage for senior citizens and certain disabled individuals. The Medicare program is administered by the Centers for Medicare and Medicaid Services (“CMS”), an agency within the U.S. Department of Health and Human Services (“HHS”). Medicare Administrative Contractors (“MACs”) are private insurance companies that serve as agents of CMS in the administration of the Medicare program and are responsible for making coverage decisions and paying claims for the designated Medicare jurisdiction. There are seven Part A/B MACs in the U.S., each with its own geographical jurisdiction, and each has its own standards and process for determining coverage and reimbursement for a procedure or product. Private payers often follow the lead of governmental payers in making coverage and reimbursement determinations. Therefore, achieving favorable Medicare coverage and reimbursement is usually a significant gating factor for successful coverage and reimbursement for a new product by private payers.
The coverage and reimbursement framework for products under Medicare is determined in accordance with the Social Security Act and pursuant to regulations promulgated by CMS, as well as the agency’s regulatory coverage and reimbursement determinations. Ultimately, however, each of the MACs determines whether and on what conditions they will provide coverage for the product. Such decisions are based on each MAC’s assessments of the science and efficacy of the applicable product. As noted below under the heading “Research and Development,” we have devoted significant resources to clinical studies to provide data to the MACs, as well as other payers, in order to demonstrate the efficacy and clinical effectiveness of our tissue technologies. As of the date of this report, both EpiFix sheets and EpiCord allografts are eligible for coverage by all MACs. In January 2019, EpiFix and EpiCord received separate CMS HCPCS Codes, Q4186 and Q4187, distinguishing each product in coverage and reimbursement policies.
For Medicare reimbursement purposes, our EpiFix and EpiCord allografts are classified as “skin substitutes.” Current reimbursement methodology varies between the hospital outpatient department (“HOPD”) and ASCs setting versus the physician office. Currently, skin substitutes are reimbursed under a “packaged” or “bundled” methodology along with the related application procedure under a two-tier payment system. In the HOPD and ASCs setting, providers receive a single payment that reimburses for the application of the product as well as the product itself. CMS classifies skin substitutes into low cost or high cost groups, based on a geometric mean unit cost and per day cost. For 2019, the geometric mean unit cost threshold applicable to both our EpiFix and EpiCord allograft products was $49 per square centimeter, and the per day cost threshold is $790. The national HOPD average packaged (“bundled”) rate for our EpiFix and EpiCord allograft products was $1,427 in 2017, was $1,568 in 2018, was $1,549 in 2019, and is $1,623 in 2020. All skin substitute products administered in the HOPD and ASCs setting are bundled except for those that have been approved by CMS for pass-through status. EpiFix was approved by CMS for pass-through status but that status expired on December 31, 2014, and EpiCord has not been approved by CMS for pass-through status. This “bundled” payment structure applies only to the HOPD and ASCs settings.
Currently, providers that administer EpiFix or EpiCord allografts and other skin substitutes in the physician office setting are reimbursed based on the size of the graft, computed on a per square centimeter basis. The payment rate is calculated using the manufacturer’s reported average sales price (“ASP”) submitted quarterly to CMS. This payment methodology applies only to physician offices. The Medicare payment rates are updated quarterly based on this ASP information for many skin substitute products but not all. EpiFix is included on the Medicare national ASP Drug Pricing File, but EpiCord is not. The published skin substitute Medicare payment rate established by statute is ASP plus 6%. Reimbursement for products not included on the Medicare national ASP Drug Pricing File are at the discretion of each MAC, which typically is invoice cost or wholesale acquisition cost (“WAC”) plus 3%.
Medicare payments for all items and services, including EpiFix sheet products and EpiCord, since 2013 have been reduced by 2% under the sequestration required by the Budget Control Act of 2011, as amended by the American Taxpayer Relief Act of 2012. Subsequent legislation extended the 2% reduction, on average, to 2027 (although the sequestration was suspended for the remainder of 2020 due to COVID-19). This 2% reduction in Medicare payments affects all parts of the Medicare program. The law allows for additional sequestration orders, potentially resulting in up to a 4% reduction in Medicare payments under a statutory PAYGO sequestration order.
14
Private Payers
We have devoted considerable resources to clinical trials to support coverage and reimbursement of our products and have confirmed an increasing number of private payers that reimburse for EpiFix in the physician office, the HOPD and the ASCs settings. Coverage and reimbursement vary according to the patient’s health plan and related benefits. The majority of health plans currently provide coverage for EpiFix for the treatment of DFUs, and many include treatment of VLUs. In 2019, numerous health plans have added EpiCord coverage for the treatment of DFUs. MiMedx has secured payer coverage for over 286 million covered lives, allowing a significant number of patients access to our products.
We have established and continue to grow a reimbursement support group to educate providers and patients with regard to accurate coverage and reimbursement information regarding our products. See discussion below – “Risk Factors” under the heading “Our revenues depend on adequate reimbursement from public and private insurers and health systems.”
Hospital Use
Products administered in the hospital inpatient setting are bundled when submitted as part of the hospital’s claim under a diagnosis-related group (“DRG”). In these cases, we continue to educate the hospital that our products are cost-effective, and have the potential to improve patient outcomes and reduce the length of stay. We are working to develop additional health economic data to support this effort. As noted above, the ability to sell products in a hospital is dependent upon demonstrating to the hospital the product’s efficacy and cost effectiveness.
Micronized and Other Products
Currently, our micronized products are available for coverage by only a limited number of Medicare, commercial and state Medicaid plans. EpiFix Micronized is listed on the Medicare national ASP Drug Pricing File and, similar to most Medicare Part B drugs, is reimbursed at ASP plus 6%, effective July 2019. There is currently no specific third-party reimbursement available for AmnioCord or AmnioFill, except to the extent such products are bundled as part of a hospital’s claim under a DRG. See discussion below – “Risk Factors” under the heading “Our revenues depend on adequate reimbursement from public and private insurers and health systems.”
Customer Concentration
A significant portion of our products are purchased by U.S. government accounts (e.g., the VA, the Public Health Service (including the Indian Health Service). For the years ended December 31, 2019, 2018 and 2017, our net sales to all U.S. government accounts comprised approximately 6%, 15%, and 9%, respectively, of our net sales. We have contracted with a third party as our indefinite delivery/indefinite quantity channel partner into the VA and DoD markets. See discussion below – “Risk Factors” under the heading “A significant portion of our revenues and accounts receivable come from government accounts.”
Competition
Due to lower barriers of entry in the 361 HCT/P regulated market, competition in the placenta-based and allograft tissue field is intense and subject to new entrants and evolving market dynamics. Companies within the industry compete on the basis of price, ease of handling, logistics and efficacy. Another important factor is third-party reimbursement, which is difficult to obtain as it is a time-consuming and expensive process. We believe our success in obtaining third-party reimbursement, robust GPO position and established clinical evidence for our products are competitive advantages.
The Agency for Healthcare Research and Quality (“AHRQ”) recently published a technology assessment analyzing Skin Substitutes for Treating Chronic Wounds. AHRQ conducted a literature search yielding 164 studies and 81 Supplemental Evidence and Data for Systematic Reviews (“SEADs”) submissions. Only 22 randomized, controlled trials (“RCTs”) met the inclusion criteria to be reviewed in the AHRQ analysis, and out of the 22 RCTs MiMedx had 6 RCTs included in the final brief. Of the 22 studies reviewed, only 12 were assessed as low risk of bias (ROB) of which 5 were MiMedx RCTs. This important government assessment highlights our commitment to providing unbiased level 1 clinical evidence in advanced wound treatment. This dedication to elevating the standard of care is further underscored by the fact that the AHRQ points out that MiMedx was the only entity to provide two studies out of the 22 evaluated that performed a subgroup analysis of patients with diabetic foot ulcers that received adequate debridement. Both studies reported an increase in wounds healed with adequate debridement.
Advanced wound care therapies employ technologies to aid in wound healing in cases where the wound is chronic and healing progress has stalled or stopped. The primary competitive products in the skin and dermal substitutes category include, among others, amniotic membrane allografts, tissue-engineered living skin equivalents, porcine-, bovine- and fish skin-derived xenografts and collagen matrix products. Xenografts, or tissue transplants from non-human species, serve mainly as an extracellular matrix and have to undergo aggressive processing to remove immunogenic animal products from the tissue. In addition, challenges with
15
xenografts include limited clinical published data, and some products may require suturing or stapling to the wound bed, making handling more difficult.
Our main competitors in the skin substitute market are Integra LifeSciences Holdings Corporation, Organogenesis, Inc., and Smith & Nephew plc, which sell a variety of advanced wound care products including allografts.
The primary competitive products in the surgical, orthopedic or sports medicine categories are other amniotic membrane allografts and injectable solutions, such as platelet-rich plasma, evolving cellular alternatives, or steroids.
See discussion below – “Risk Factors” under the heading “We are in a highly competitive and evolving field and face competition from well-established tissue processors and medical device manufacturers, as well as new market entrants.”
Government Regulation
The products manufactured and processed by the Company are derived from human tissue. As discussed below, Section 361 HCT/Ps are tissue-based products that are regulated solely under Section 361 and do not require pre-market clearance or approval by the FDA. Section 351 HCT/Ps are also tissue products but are regulated as biological products, medical devices or drugs and, in order to be lawfully marketed in the United States, require FDA pre-market clearance or approval. See discussion below – “Risk Factors” under the heading “Risks Related to Regulatory Approval of Our Products and Other Government Regulations.”
Tissue Products
In 1997, the FDA proposed a new regulatory framework for cells and tissues. This framework was intended to provide adequate protection of public health while enabling the development of new therapies and products with as little regulatory burden as possible. A key innovation in the system is that covered HCT/Ps would be regulated solely under Section 361 and would not be subject to pre-market clearance. The registration and listing rules were finalized in January 2001 in 21 CFR Part 1271. Additional rules regarding donor eligibility and good tissue practices were soon adopted. Together, these rules form a comprehensive system intended to encourage significant innovation.
The FDA requires each HCT/P establishment to register and establish that its product meets the requirements to qualify for regulation solely under Section 361. To be a Section 361 HCT/P, a cellular or tissue-based product generally must meet all four of the following criteria (fully set forth in 21 CFR Part 1271):
• | it must be minimally manipulated; |
• | it must be intended for homologous use; |
• | its manufacture must not involve combination with another article, except for water, crystalloids or a sterilizing, preserving or storage agent; and |
• | it must not have a systemic effect and must not be dependent upon the metabolic activity of living cells for its primary function. |
Amniotic and other birth tissue are considered cellular and tissue-based articles and are therefore eligible for regulation solely as a Section 361 HCT/P depending on whether the specific product at issue and the claims made for it are consistent with the criteria set forth above. HCT/Ps that do not meet these criteria are subject to more extensive regulation as drugs, medical devices, biological products or combination products.
Products Regulated Solely as HCT/Ps
The FDA has specific regulations governing HCT/Ps, including some regulations specific to Section 361 HCT/Ps, which are set forth in 21 CFR Part 1271. All establishments that manufacture Section 361 HCT/Ps must register and list their HCT/Ps with the FDA’s Center for Biologics Evaluation and Research within five days after commencing operations. In addition, establishments are required to update their registration annually in December or within 30 days of certain changes and submit changes in HCT/P listing at the time of or within six months of such change.
The regulations in 21 CFR Part 1271 also require establishments to comply with donor screening, eligibility and testing requirements and cGTP to prevent the introduction, transmission and spread of communicable diseases. The cGTP govern, as may be applicable, the facilities, controls and methods used in the manufacture of all HCT/Ps, including processing, storage, recovery, labeling, packaging and distribution of Section 361 HCT/Ps. cGTP require us, among other things, to maintain a quality program, train personnel, control and monitor environmental conditions as appropriate, control and validate processes, properly store, handle and
16
test our products and raw materials, maintain our facilities and equipment, keep records and comply with standards regarding recovery, pre-distribution, distribution, tracking and labeling of our products and complaint handling. 21 CFR Part 1271 also mandates compliance with adverse reaction and cGTP deviation reporting and labeling requirements.
The FDA conducts periodic inspections of HCT/P manufacturing facilities, and contract manufacturers’ facilities, to assess compliance with cGTP. Such inspections can occur at any time with or without written notice at such frequency as determined by the FDA in its sole discretion. To determine compliance with the applicable provisions, the inspection may include, but is not limited to, an assessment of the establishment’s facilities, equipment, finished and unfinished materials, containers, processes, HCT/Ps, procedures, labeling, records, files, papers and controls required to be maintained under 21 CFR Part 1271. If the FDA were to find serious non-compliant manufacturing or processing practices during such an inspection, it could take regulatory actions that could adversely affect our business, results of operations, financial condition and cash flows.
FDA Letter Regarding AmnioFix Injectable and Other Micronized Products
In August 2013, the Company received an untitled letter from the Office of Compliance and Biologics Quality (“OCBQ”) within the FDA’s Center for Biologics Evaluation and Research concerning AmnioFix Injectable and other micronized products (the “Untitled Letter”). The Untitled Letter asserted that our micronized products, including AmnioFix Injectable, are not properly regulated solely under Section 361 because they are more than “minimally manipulated” as that term is defined in FDA regulations. Accordingly, the Untitled Letter asserted that the products at issue are drugs and biological products that require valid biologics licenses to be in effect in order to be lawfully marketed.
The Company disagreed at the time, taking the position that micronization was allowed for Section 361 HCT/Ps under the then applicable guidance. Because the Untitled Letter seemed to be contrary to existing guidance, the Company attempted to engage with OCBQ and ultimately pursued two levels of supervisory review. As part of that process, the Company agreed to pursue a biologics license for AmnioFix Injectable, and has since filed IND applications with the FDA covering clinical studies for AmnioFix Injectable that are discussed in greater detail below. In November 2016, following this supervisory review process, the Acting Chief Scientist of the FDA informed the Company that additional agency review of the Untitled Letter was not warranted.
Recent FDA Guidance and Transition Policy for HCT/Ps
In November 2017, the FDA released four guidance documents that, collectively, the agency described as a “comprehensive policy framework” for applying existing laws and regulations governing regenerative medicine products, including HCT/Ps. One guidance document in particular, “Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue – Based Products: Minimal Manipulation and Homologous Use – Guidance for Industry and Food and Drug Administration Staff,” offered important clarity on some of the issues that the Company raised on appeal to the Untitled Letter.
The guidance documents confirmed that sheet forms of amniotic tissue are appropriately regulated as solely Section 361 HCT/Ps when intended for use as a barrier or covering. We are in the process of evaluating our marketing materials for each of our products to align with the FDA’s guidance.
Second, the guidance documents confirmed the FDA’s stance that all micronized amniotic membrane products require a biologics license to be lawfully marketed in the United States. However, the guidance documents also stated that the FDA intends to exercise enforcement discretion under limited conditions with respect to the IND application and pre-market approval requirements for certain HCT/Ps through November 2020. This 36-month period of enforcement discretion was intended to give sponsors time to evaluate their products, have a dialogue with the agency and, if necessary, begin clinical trials and file the appropriate pre-market applications. The FDA’s approach is risk-based, and the guidance documents clarified that high-risk products and uses could be subject to immediate enforcement action.
This enforcement discretion applies across our industry, and the Company has continued to market its products under this policy of enforcement discretion. At the same time, we are pursuing the BLA pre-market approval process for certain uses of AmnioFix Injectable. There is no assurance that the FDA will grant these approvals on a timely basis, or at all, or that we will not discontinue our pursuit of a BLA for certain products or indications. We previously announced that we will need more time to file our BLAs with the FDA and that clinical trial protocol amendments and enhancements, further resources and additional capabilities and expertise will be required. See “Clinical Trials” below for information regarding the revised timelines.
During the remainder of the 36-month enforcement discretion period, the Company will also continue to explore possible options for extending this enforcement discretion period. To this end, the Company has initiated dialogue and efforts for a further transition plan with the FDA to allow for continued marketing of the impacted products while the Company transitions to compliance with Section 351, the applicable sections of the FD&C Act, the cGMP regulations in 21 CFR Part 210 and 211, and other applicable
17
FDA regulations. This would be an extension of the current policy, and there is no guarantee that the FDA will provide more time, either for MiMedx or the industry at large.
Products Regulated as Biologics – The BLA Pathway
The typical steps for obtaining FDA approval of a BLA to market a biological product in the United States include:
• | Completion of preclinical laboratory tests, animal studies and formulations studies under the FDA’s Good Laboratory Practice regulations; |
• | Submission to the FDA of an IND application for human clinical testing, which must become effective before human clinical trials may begin and which must include independent Institutional Review Board approval at each clinical site before the trials may be initiated; |
• | Performance of adequate and well-controlled clinical trials in accordance with Good Clinical Practices to establish the safety and efficacy of the product and its dosage (as applicable) for each indication; |
• | Development of purity, potency and identity tests to demonstrate consistency and reliability of the manufacturing process through a chemistry, manufacturing and control program; |
• | Submission to the FDA of a BLA for marketing the product, which includes, among other things, reports of the outcomes and full data sets of the clinical trials, and proposed labeling and packaging for the product; |
• | Satisfactory review of the contents of the BLA by the FDA, including the satisfactory resolution of any questions raised during the review; |
• | Satisfactory completion of an FDA Advisory Committee review, if applicable; |
• | Satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with FDA’s cGMP regulations, to assure that the facilities, methods and controls are adequate to ensure the product’s identity, strength, quality and purity; and |
• | FDA approval of the BLA, including agreement on post-marketing commitments, if applicable. |
Generally, clinical trials are conducted in three phases, though the phases may overlap or be combined. Phase 1 trials typically involve a small number of healthy volunteers and are designed to provide information about the product safety and to evaluate the pattern of drug distribution and metabolism within the body. Phase 2 trials are conducted in a larger but limited group of patients afflicted with a particular disease or condition in order to determine preliminary efficacy, dosage tolerance and optimal dosing, and to identify possible adverse effects and safety risks. Dosage studies are typically designated as Phase 2A, and efficacy studies are designated as Phase 2B. Phase 3 clinical trials are generally large-scale, multi-center, comparative trials conducted with patients who have a particular disease or condition in order to provide statistically valid proof of efficacy, as well as safety and potency. In some cases, the FDA will require Phase 4, or post-marketing trials, to collect additional data after a product is on the market. All phases of clinical trials are subject to extensive record keeping, monitoring, auditing and reporting requirements.
The FDA has broad regulatory compliance and enforcement powers. If the FDA determines that MiMedx has failed to comply with applicable regulatory requirements, it can take a variety of compliance or enforcement actions, such as issuing an FDA Form 483 notice of inspectional observations; sending a warning letter or untitled letter; issuing an order of retention, destruction, or cessation of marketing; imposing civil money penalties; suspending or delaying issuance of approvals; requiring product recalls; imposing a total or partial shutdown of production; withdrawing approvals or clearances already granted; pursuing product seizures, consent decrees or other injunctive relief; and criminal prosecution through the DOJ.
18
Clinical Trials
Trial Overview
The Company is currently conducting three IND programs investigating the use of AmnioFix Injectable to reduce pain and increase function in patients with plantar fasciitis, Achilles tendonitis, and osteoarthritis of the knee. Based on a review of the studies and interim results, the Company has instituted several actions with respect to its ongoing and anticipated clinical trials to address the resources, capabilities and expertise needed for commercial launch, including our strategy around an increased dialogue with the FDA regarding our BLA progress. The trials were developed and initially overseen by senior managers who are no longer with the Company and, as previously disclosed, we have concluded that the trials must be improved if they are to support BLA submissions and approvals. However, there can be no assurance that we will obtain BLA approval and we may ultimately decide not to pursue a BLA for certain products or indications. See Risk Factors - “Obtaining and maintaining the necessary regulatory approvals for certain of our products will be expensive and time consuming and may impede our ability to fully exploit our technologies.”
Plantar Fasciitis
In March 2015, we initiated a Phase 2B prospective, single-blinded, RCT investigating a single injection of 40 mg of AmnioFix Injectable as compared to a single intra-plantar injection of saline (placebo control) in the treatment of patients with recalcitrant plantar fasciitis pain and foot dysfunction. This trial enrolled 145 patients at 15 study sites. In September 2017, we announced the trial had met its efficacy endpoints, and the three-month endpoint data were published in 2018.
In April 2017, we met with the FDA and informally discussed preliminary data from the Phase 2 study, our progress toward achieving GMP compliance, and our proposed Phase 3 study design. We incorporated the FDA’s formal feedback into our development plans, and will plan further meetings as needed and required to achieve the goal of successful BLA submission. Based on the Phase 2B interim data, in January 2018 we initiated a Phase 3 prospective, double-blinded, RCT to assess the safety and efficacy of a single 40 mg intra-plantar injection of AmnioFix Injectable as compared to a single intra-plantar injection of saline (placebo control) to treat patients with recalcitrant plantar fasciitis pain. The trial plan was initially to enroll 164 patients. In July 2019, we conducted an interim analysis to assess adequacy of the sample size to assess differences between the two treatment groups. We analyzed the data received from this sample size analysis, conducted on subjects representing 50% of total enrollment that had reached the primary efficacy endpoint. This analysis indicated that a significant increase in sample size would be required to observe clinically and statistically significant improvement and separation between treatment and control groups. We determined that increasing the sample size to 276 patients would provide sufficient power to observe an efficacy result with statistical and clinical significance. We have instituted these changes and amendments and expect to complete enrollment by October 2020. The COVID-19 pandemic has had a major dampening effect on study enrollment. There can be no assurance that this effect will fully resolve and allow completion of the study in the anticipated timeframe, that a second wave of virus infections will not occur, that no further disruptions can be expected, or when completed, that the FDA will view such study as sufficient to support a BLA filing.
If the plantar fasciitis trials are successful, determined to be adequate proof of efficacy and safety, we expect to file a BLA for AmnioFix Injectable to treat patients with plantar fasciitis in the second half of 2021. We expect the outcome of this trial to help inform additional areas of unmet need for potential clinical study. However, we now expect that FDA approval to market AmnioFix Injectable for this indication will take longer than previously expected and may take several years, and there can be no assurance that we will receive FDA approval. Approval may be delayed due to a variety of factors, including failure of the studies to achieve their endpoints, the extra effort and cost required to improve our clinical trials as described above, the impact of the COVID-19 pandemic on study enrollment and FDA operations, the potential that we reevaluate our commercialization strategy, and the work required to achieve commercial and manufacturing readiness. See discussion below - “Risk Factors” under the heading “Obtaining and maintaining the necessary regulatory approvals for certain of our products will be expensive and time-consuming and may impede our ability to fully exploit our technologies.”
Knee Osteoarthritis
In March 2018, the FDA granted AmnioFix Injectable the Regenerative Medicine Advanced Therapy (“RMAT”) designation for use in the treatment of osteoarthritis of the knee. RMAT-designated products are eligible for increased and earlier interactions with the FDA, similar to those interactions available to fast track and breakthrough-designated therapies. In addition, these products may be eligible for rolling review and accelerated approval. The meetings with sponsors of RMAT-designated products may include discussions of whether accelerated approval would be appropriate based on surrogate or intermediate endpoints reasonably likely to predict long-term clinical benefit or reliance upon data obtained from a meaningful number of sites.
In March 2018, we initiated a Phase 2B prospective, double-blinded RCT investigating a single intra-articular injection of 40 mg of AmnioFix Injectable as compared to a single injection of saline (placebo control) in the treatment of pain and functional impairment in patients with osteoarthritis of the knee. This trial was planned to enroll 318 patients, with an interim analysis to
19
assess adequacy of this sample size built into the statistical plan. This blinded interim analysis was performed in August 2019 and revealed that while differences in the treatment groups were observed, the power to observe statistically and clinically significant results would be enhanced by increasing the sample size to 466 patients. Amendments to the protocol to allow this increase were subsequently approved and instituted and enrollment is progressing.
We also amended the protocol and established an open label extension to the trial, to allow patients to receive a second injection of the active treatment at six months, nine months, or 12 months subsequent to their completion of study visits, if their pain has not resolved or responded, regardless of treatment arm. The study will still be blinded to subjects, sites and MiMedx during this extension. However, we now expect that FDA approval to market AmnioFix Injectable for this indication will take longer than previously expected, and it may take several years. There can be no assurance that we will ultimately receive FDA approval. Approval may be delayed due to a variety of factors, including failure of the studies to achieve their endpoints, the extra effort and cost required to improve our clinical trials as described above, the impact of the COVID-19 pandemic on study enrollment and FDA operations, and the work required to achieve commercial and manufacturing readiness. See discussion below - “Risk Factors” under the heading “Obtaining and maintaining the necessary regulatory approvals for certain of our products will be expensive and time-consuming and may impede our ability to fully exploit our technologies.”
Achilles Tendonitis
In January 2018, we initiated a Phase 3 prospective, double-blinded RCT investigating a single intra-tendon injection of 40 mg of AmnioFix Injectable as compared to a single injection of saline (placebo control) in the treatment of Achilles tendonitis. The planned trial enrollment was 158 patients, with an interim analysis to assess adequacy of the sample size built into the statistical plan. We analyzed data received from this sample size analysis, conducted on patients representing 50% of total enrollment that had reached the primary efficacy endpoint. This indicated that a substantial increase in sample size would be required to observe clinically and statistically significant improvement and separation between treatment and control groups. With this in mind, we have concluded that the most reasonable approach was to continue the study to completion with the originally planned sample size, and analyze the final results to determine the adequacy of the measures employed and time points of observation to show meaningful clinical and statistical analyses. Enrollment for this study has completed and we anticipate that the last patient visit will occur in the first half of 2021.
Other
In addition, we intend to file an IND for AmnioFill in the second half of 2020, although we have not yet initiated any clinical trials under an IND in furtherance of any regulatory approvals for AmnioFill. We also intend to file an IND for injectable micronized EpiFix for the treatment of DFUs or other areas of advanced wound care in the second half of 2020. Clinical study initiation will depend on FDA feedback for both of these programs. Given the timelines of these proposed filings and anticipated delays at FDA in processing applications due to the COVID-19 pandemic, it is likely that studies will not begin enrollment in this calendar year.
BLA Process
If any of the study results support potential product approval and potential for commercialization, we intend to file BLAs as described above. The process of obtaining an approved BLA requires the expenditure of substantial time, effort and financial resources and may take years to complete. The fee for filing a BLA and the annual user fees payable with respect to any establishment that manufactures biologics and with respect to each approved product are substantial. While there can be no assurance that we will ultimately obtain regulatory approval for our micronized products, we have already completed substantial work towards multiple BLAs, including engineering our manufacturing processes to conform to cGMP guidances.
FDA Post – Market Regulation
Tissue processors regulated solely under Section 361 are still required to register as an establishment with the FDA. As a registered establishment, we are required to comply with regulations regarding labeling, record keeping, donor eligibility, screening and testing. We are also required to process the tissue in accordance with established cGTP, as well as report any adverse reactions caused by a possible transmission of an infectious disease attributed to our tissue. Our facilities are also subject to periodic inspections to assess our compliance with the regulations.
Products covered by a BLA, New Drug Application, 510(k) clearance or a pre-market approval are subject to numerous additional regulatory requirements, which include, among others, compliance with cGMP (or, in the case of devices, with FDA’s Quality System Regulation), which imposes certain procedural, substantive and record keeping requirements, and labeling regulations to ensure a product’s identity, strength, quality, and purity. These products are also subject to the FDA’s general prohibition against promoting products for unapproved or “off-label” uses, and additional adverse reaction reporting.
20
As part of our BLA development effort, we are updating our manufacturing establishments into compliance with cGMP for production for our injectable and other applicable Section 351 products. We are also pursuing opportunities to partner with a contract manufacturing organization. The transition process includes development and enhancement of production processes, procedures, test and assays, and it requires extensive validation work. It can also involve the procurement and installation of new production or lab equipment. These efforts require human capital, expertise and resources. We have made significant improvements in this transition over the last year. We have engaged industry experts to assess our state of compliance and to provide guidance on the additional activities needed to meet cGMPs. Our goal is to achieve compliance with cGMP for our injectable and other applicable Section 351 products by the time the FDA’s current period of enforcement discretion is complete in November 2020. See discussion below – “Risk Factors” under the heading “To the extent our products do not qualify for regulation as human cells, tissues and cellular and tissue-based products solely under Section 361 of the Public Health Service Act, this could result in removal of the applicable products from the market, would make the introduction of new tissue products more expensive and would significantly delay the expansion of our tissue product offerings and subject us to additional post-market regulatory requirements,” and “We may be subject to fines, penalties, injunctions and even criminal sanctions if we are deemed to have made a misstatement of compliance to a federal agency.”
Other Regulation Specific to Tissue Products
National Organ Transplant Act
Procurement of certain human organs and tissue for transplantation is subject to the restrictions of the National Organ Transplant Act (“NOTA”), which prohibits the transfer of certain human organs, including skin and related tissue, for valuable consideration, but permits the reasonable payment associated with the removal, transportation, implantation, processing, preservation, quality control and storage of human tissue and skin. Our wholly-owned subsidiary, MiMedx Tissue Services, LLC, is registered with the FDA as an establishment that manufactures human cells, tissues and cellular and tissue-based productions and is involved with the recovery and storage of donated human amniotic tissue. We reimburse tissue banks, hospitals and physicians for their services associated with the recovery and storage of donated human tissue.
Tissue Bank Laws, Regulations, and Related Accreditation
As discussed above, we are required to register with the FDA as an establishment that manufactures human cells, tissues and cellular and tissue-based products. We are licensed, registered, or permitted as a tissue bank in California, Georgia, New York, Delaware, Illinois, Oregon, and Maryland. Additionally, we received and actively maintain AATB accreditation. The AATB has issued operating standards for tissue banking. Compliance with these standards is required in order to become an AATB-accredited tissue establishment. AATB standards include specific requirements for recovery, screening, testing, labeling and processing of placenta tissue. We believe we are compliant in all material respects with AATB standards and our state licensure requirements.
To the extent we sell our products outside of the United States, we also are subject to laws and regulations of foreign countries.
Other Healthcare Laws and Compliance Requirements
In the United States, our activities are potentially subject to regulation by various federal, state and local authorities in addition to the FDA, including CMS, other divisions of the HHS (e.g., the Office of Inspector General), the DOJ and individual United States Attorney offices within the DOJ, and state and local governments. These regulations include those described below.
• | The federal Anti-Kickback Statute, which is a criminal law that prohibits, among other things, any person from knowingly and willfully offering, soliciting, receiving or providing any remuneration (including any kickback, bribe or rebate), directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward referrals, purchases or orders, or arranging for or recommending the purchase, order or referral of any item or service for which payment may be made in whole or in part by a federal healthcare program, such as the Medicare and Medicaid programs. The term “remuneration” has been broadly interpreted to include anything of value. The Patient Protection and Affordable Care Act amended the intent requirement of the federal Anti-Kickback Statute, so that a person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it. A conviction for violation of the Anti-Kickback Statute results in criminal fines and requires mandatory exclusion from participation in federal health care programs. Although there are a number of statutory exceptions and regulatory safe harbors to the federal Anti-Kickback Statute that protect certain common industry practices from prosecution, the exceptions and safe harbors are drawn narrowly, and arrangements may be subject to scrutiny or penalty if they do not fully satisfy all elements of an available exception or safe harbor. See discussion below under “Risk Factors–We and our sales representatives, whether employees or independent contractors, must comply with various federal and state anti-kickback, self-referral, false claims and similar laws, any breach of which could cause an adverse effect on our business, results of operations and financial condition.” |
21
• | The federal False Claims Act (“FCA”) imposes significant civil liability on any person or entity that knowingly presents, or causes to be presented, a claim for payment to the U.S. government, including the Medicare and Medicaid programs, that is false or fraudulent. The FCA also allows a private individual or entity as a whistleblower to sue on behalf of the government to recover civil penalties and treble damages. FCA liability is potentially significant in the healthcare industry because the statute provides for treble damages and mandatory penalties of between $11,181 and $22,363 per false claim or statement for penalties assessed after January 29, 2018, with respect to violations occurring after November 2, 2015. As a result of a modification made by the Fraud Enforcement and Recovery Act of 2009, a claim includes “any request or demand” for money or property presented to the U.S. government. |
• | The federal Health Insurance Portability and Accountability Act of 1996 (“HIPAA”) fraud and abuse provisions prohibit executing a scheme to defraud any healthcare benefit program, willfully obstructing a criminal investigation of a health care offense, or making false statements or concealing a material fact relating to payment for healthcare benefits, items or services. |
• | While manufacturers of human cell and tissue products regulated solely under Section 361 are not subject to the federal Physician Payments Sunshine Act and its implementing regulations (together with the Act, the “Sunshine Act”), in the future, if we receive a BLA approval, this law will require us (with certain exceptions) to report information to CMS related to certain payments or other transfers of value we make to U.S.-licensed physicians and teaching hospitals, and for reports submitted on or after January 1, 2022, physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists and certified nurse-midwives. If we receive a BLA approval, the Sunshine Act would also require us to report annually certain ownership and investment interests held by U.S.-licensed physicians and their immediate family members. Such information will subsequently be made publicly available by CMS on the Open Payments website. |
• | Federal conflicts of interest laws, the Standards of Ethical Conduct for Employees of the Executive Branch, and local site policies for each federal institution we call upon govern our interactions with federal employees at our various government accounts (e.g., Department of Defense (“DoD”), VA, etc.) and impose a number of limitations on such interactions. |
• | There are state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws, which may apply to items or services reimbursed by any third-party payer, including commercial insurers, many of which differ from each other in significant ways and often are not preempted by federal laws, thus complicating compliance efforts. |
In addition, we may be subject to data privacy and security regulation by both the federal government and the states in which we conduct our business. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act (“HITECH”) and its implementing regulations, imposes certain requirements relating to the privacy, security and transmission of protected health information. Among other things, HITECH made HIPAA’s privacy and security standards directly applicable to “business associates,” independent contractors or agents of covered entities that receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also created four new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates and possibly other persons and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions. In addition, state laws govern the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
Seasonality
We typically experience seasonality, with lower shipments in the first quarter of each year compared to the immediately preceding fourth quarter. This seasonal shipments pattern relates to U.S. annual insurance deductible resets and unfunded flexible spending accounts.
Research and Development
Our research and development group has extensive experience in developing products related to our field of interest, and works to design products that are intended to improve patient outcomes, simplify techniques, shorten procedures, reduce hospitalization and rehabilitation times and, as a result, reduce costs. Our research and development group also works to establish scientific evidence in support of the use of our products. Clinical trials that demonstrate the safety, efficacy and cost effectiveness of our products are key to obtaining broader third-party reimbursement for our products. In addition to our internal staff, we contract with outside labs and physicians who aid us in our research and development process. See Part II, Item 7, below, for information regarding expenditures for research and development in each of the last three fiscal years.
22
Environmental Matters
Our tissue preservation activities generate a small amount of chemical and biomedical waste, consisting primarily of diluted alcohols and acids and human biological waste, including human tissue and body fluids removed during laboratory procedures. The biomedical waste generated by our tissue processing operations are placed in appropriately constructed and labeled containers and are segregated from other waste. We contract with third parties for transport, treatment, and disposal of our biomedical waste.
Employees
As of December 31, 2019, we had 696 employees. We consider our relationships with our employees to be satisfactory. None of our employees are covered by a collective bargaining agreement.
Available Information
We are required to file proxy statements, annual reports on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K with the SEC. The SEC maintains an internet site, www.sec.gov, where these reports are available free of charge.
We also make these reports available free of charge on our website, www.mimedx.com, under the heading “Investors–SEC Filings.” In addition, our Audit Committee, Compensation Committee, Ethics and Compliance Committee, and Nominating and Corporate Governance Committee Charters as well as our Code of Business Conduct and Ethics, are on our website under the heading “Investors–Corporate Governance.” The reference to our website does not constitute incorporation by reference of any information contained on that site.
23
Item 1A. Risk Factors
An investment in our Common Stock involves a substantial risk of loss. Set forth below are descriptions of those risks and uncertainties that we currently believe to be material, but the risks and uncertainties described below are not the only risks and uncertainties that could materially adversely affect our business, financial condition and operating results. If any of these risks materialize, our business, financial condition or operating results could suffer. In this case, the trading price of our Common Stock could decline, and you may lose part or all of your investment.
Risks Related to Our Business and Industry
Our substantial indebtedness may adversely affect our financial health.
On July 2, 2020, the Company borrowed an aggregate of $50 million and obtained an additional committed but undrawn $25 million facility pursuant to the Hayfin Loan Agreement. See Item 9B, “Other Information.” Following the closing of the Preferred Stock Transaction and the Hayfin Loan Transaction, and the repayment of the BT Loan Agreement, as of July 2, 2020, the Company had approximately $110 million of cash and cash equivalents and approximately $50 million of long-term debt.
Our substantial outstanding debt may limit our ability to borrow additional funds or may adversely affect the terms on which such additional funds may be available. Additionally, a default under certain other indebtedness constitutes an event of default under the Hayfin Loan Agreement. Consequently, the effects of a default under other debt may be amplified by the lender exercising the remedies available to them in the Hayfin Loan Agreement for events of default, including foreclosure on the collateral securing our obligations and the declaration that all amounts outstanding under the Hayfin Loan Agreement are immediately due and payable. The limitations on our ability to access additional borrowing and the potential effects of a cross-default under the Hayfin Loan Agreement may limit our liquidity and have an adverse effect on our business, financial condition, and results of operations.
The restrictive covenants in the Hayfin Loan Agreement, and the Company’s obligation to make debt payments under the Hayfin Loan Agreement, limit our operating and financial flexibility and may adversely affect our business, results of operations and financial condition.
The Hayfin Loan Agreement imposes operating and financial restrictions and covenants. For example, the Hayfin Loan Agreement contains (a) certain covenants that impose certain reporting and/or performance obligations on the Company and its subsidiaries, including (i) a maximum Total Net Leverage Ratio (as defined in the Hayfin Loan Agreement) of 5.0x through December 31, 2020, stepping down to 4.5x through June 30, 2021 and to 4.0x thereafter until July 2, 2025, in each case tested quarterly; and (ii) Minimum Liquidity (as defined in the Hayfin Loan Agreement) of $10 million, an at-all-times covenant tested monthly and (b) certain negative covenants that generally limit, subject to various exceptions, the Company and its subsidiaries from taking certain actions, including, without limitation, incurring indebtedness (including with respect to drawdowns under the delayed draw term loan (the “DD TL”) if the Total Net Leverage Ratio (pro forma for such drawdowns) exceeds 3.5x), making investments, incurring liens, paying dividends and engaging in mergers and consolidations, sale and leaseback transactions and asset dispositions.
A breach of a financial covenant in the Hayfin Loan Agreement would result in an event of default that would trigger the lenders’ remedies, including the right to accelerate the entire principal balance of the loan under the Hayfin Loan Agreement (the “Hayfin Term Loan”). There can be no assurances that we will be able to repay all such amounts or be able to find alternative financing in case of such or other event of a default. Even if alternative financing is available in an event of a default under the Hayfin Loan Agreement, it may be on unfavorable terms, and the interest rate charged on any new borrowings could be substantially higher than the interest rate under the Hayfin Loan Agreement, thus adversely affecting our cash flows, liquidity, and results of operations. Acceleration of the repayment of the loan pursuant to the terms of the Hayfin Loan Agreement, in combination with the Company’s current commitments and contingent liabilities, could also cast doubt on the Company’s ability to continue as a going concern.
Our variable rate indebtedness under the Hayfin Loan Agreement subjects us to interest rate risk, which could result in higher expense in the event of increases in interest rates and adversely affect our business, financial condition, and results of operations.
Borrowings under the Hayfin Loan Agreement bear interest at a per annum rate equal to London Interbank Offered Rate (“LIBOR,” subject to a “floor” of 1.5%) plus a margin of 6.75% per annum. (Such margin is subject to step down after December 31, 2020 to 6.5% or 6.0% based on Total Net Leverage Ratio levels, as defined in the Hayfin Loan Agreement.) As a result, we are exposed to interest rate risk, which we do not hedge. If LIBOR rises, the interest rate on outstanding borrowings under the Hayfin Loan Agreement will increase. Therefore, an increase in LIBOR will increase our interest payment obligations under the Hayfin Loan Agreement and have a negative effect on our cash flows and liquidity, and could have a negative effect on our ability to make payments due under the Hayfin Loan Agreement.
24
If we do not successfully execute our priorities, our business, operating results and financial condition could be adversely affected.
Our priorities are to participate in the growth in the advanced wound care category, increase the Company’s market share by demonstrating the positive health economics of our products, and accelerate the timeline to achieve our long-range growth objectives, including our BLA pipeline. We have sought and may continue to seek capital to implement our priorities, which include advancing our BLA programs and seeking FDA approval for micronized dHACM to treat musculoskeletal degeneration across multiple indications.
In developing our priorities, we evaluated many factors including, without limitation, those related to developments in our industry, customer demand, competition, regulatory developments, and the ability of the Company to execute a capital raise and general economic conditions. Actual conditions may be different from our assumptions, and we may not be able to successfully execute our priorities. If we do not successfully execute our priorities, or if actual results vary significantly from our assumptions, our business, operating results and financial condition could be adversely impacted.
In addition, managing our growth may be more difficult than we expect. We anticipate that a period of significant expansion will be required to penetrate and service the market for our existing and anticipated future products and to continue to develop new products. This expansion will place a significant strain on management and operational and financial resources. To manage the expected growth of our operations and personnel, we must both modify our existing operational and financial systems, procedures and controls and implement new systems, procedures and controls. We must also expand our finance, administrative and operations staff. Management may be unable to hire, train, retain, motivate and manage necessary personnel or to identify, manage and exploit existing and potential relationships and market opportunities.
We are in a highly competitive and evolving field and face competition from well-established tissue processors and medical device manufacturers, as well as new market entrants.
Our business is in a very competitive and evolving field. Competition from other tissue processors, medical device companies, and biotherapeutic companies, and from research and academic institutions, is intense, expected to increase and subject to rapid change and could be significantly affected by new product introductions. Established competitors and newer market entrants are investing in additional clinical research that may allow them to gain further clinician usage, adoption and payer coverage of their products. In addition, consolidation and cost containment measures in the healthcare industry may cause hospitals to consolidate their purchases with suppliers that have a broad portfolio of products. This would continue to give rise to demands for price concessions, which could have an adverse effect on our business, results of operations and financial condition. Further, competitors may introduce amniotic membrane products in the future at lower prices, adding new features or gaining additional reimbursement coverage. Further, they may copy our products outside the United States. The presence of this competition may lead to pricing pressure, which could have an adverse effect on our business, results of operations and financial condition.
Rapid technological change could cause our products to become obsolete and, if we do not enhance our product offerings through our research and development efforts, we may be unable to compete effectively.
The technologies underlying our products are subject to rapid and profound technological change. Competition intensifies as technical advances in each field are made and become more widely known. Others may develop services, products or processes with significant advantages over the products, services and processes that we offer or are seeking to develop. Any such occurrence could have an adverse effect on our business, results of operations and financial condition.
We plan to enhance and broaden our product offerings in response to changing customer demands and competitive pressure and technologies. The success of any new product offering or enhancement to an existing product will depend on numerous factors, including our ability to:
• | properly identify and anticipate physician and patient needs; |
• | acquire, through licensing, co-development or outright purchase, new technology developed outside of MiMedx; |
• | develop and introduce new products or product enhancements in a timely manner; |
• | adequately protect our intellectual property and avoid infringing upon the intellectual property rights of third parties; |
• | demonstrate the safety and efficacy of new products; and |
• | obtain the necessary regulatory clearances or approvals for new products or product enhancements. |
25
If we do not develop and, when necessary, obtain regulatory clearance or approval for new products or product enhancements in time to meet market demand, or if there is insufficient demand for these products or enhancements, our results of operations and financial condition will suffer. Our research and development efforts may require a substantial investment of time and resources before we are adequately able to determine the commercial viability of a new product, technology, material or other innovation. In addition, even if we are able to successfully develop enhancements or new generations of our products, these enhancements or new generations of products may not produce sales in excess of the costs of development, or they may never receive required regulatory approval and they may be quickly rendered obsolete by changing customer preferences or the introduction by our competitors of products embodying new technologies or features.
Our products depend on the availability of tissue from human donors, and any disruption in supply could adversely affect our business.
The success of our human tissue products depends upon, among other factors, the availability of tissue from human donors. Any failure to obtain tissue from our sources will interfere with our ability to effectively meet demand for our products incorporating human tissue. The availability of donated tissue could also be adversely impacted by regulatory changes, public opinion of the donor process and our own reputation in the industry. Obtaining adequate supplies of human tissue involves several risks, including limited control over availability (for example, access to hospital accounts and the number of consenting mothers), quality and delivery schedules. In addition, any interruption in the supply of any human tissue component could harm our ability to manufacture our products until a new source of supply, if any, could be found. We also utilize third-party providers of placental donations to mitigate risks but there can be no assurance that these third parties will be able to provide donated tissues at all times. We may be unable to find a sufficient alternative supply channel in a reasonable time period or on commercially reasonable terms, if at all, which would have an adverse effect on our business, results of operations and financial condition.
The COVID-19 pandemic and governmental and societal responses thereto have adversely affected our business, results of operations and financial condition, and the continuation of COVID-19 or the outbreak of other health epidemics could harm our business, results of operations, and financial condition.
The COVID-19 pandemic and governmental and societal responses thereto have adversely affected our business, results of operations and financial condition, and will likely continue to do so. See Item 7, “Management’s Discussion and Analysis - Results of Operations.”
The continuation or additional waves of the outbreak of the COVID-19 pandemic has adversely affected, and may continue to adversely affect, our operations and increase our costs and expenses in numerous ways. Our clinical researchers and customers have experienced restrictions in their access to hospitals and ability to access other healthcare providers. If our leadership, employees, sales agents, suppliers, medical professionals, or users of our products are impacted by an epidemic, by illness, or through social distancing, quarantine or other precautionary measures, then our manufacturing operations, sales and demand for our product, and clinical trials may be adversely affected. This risk is particularly acute for our manufacturing operations, which take place in a confined area. Additionally, if we experience shortages of donated placentas because donors or our recovery specialists are excluded from hospitals, or because additional testing protocols are implemented for donated tissues based on guidance issued by the AATB, FDA, or other standards and are screened as ineligible, our results of operations may be adversely affected. In many areas, our sales force was excluded from hospitals and the offices of other health care providers from late March until mid-May 2020. This adversely affected our revenues beginning late in the first quarter of 2020 and continuing into April. While access to hospitals and healthcare providers by our sales force had been mostly restored by mid-May, future restrictions on access to hospitals by our sales force or patients may have an additional adverse effect on our revenues and results of operations. Disruptions to the health care system generally, such as if patients are unable or unwilling to visit health care providers, or if health care providers prioritize treatment of acute or communicable illnesses over wound care, have and may continue to adversely affect our revenues and results of operations. For example, from mid-March through mid-May 2020, many patients stayed away from hospitals and other medical facilities, which adversely impacted revenues and stalled enrollments in our clinical trials. Additionally, as of early July 2020, additional restrictions have been put in place in some areas of the country that again limit or postpone elective surgical procedures, and in particular, in areas of the country that contribute a larger portion of our sales. Also, the severity of the COVID-19 pandemic has been uneven across the country, and additional waves of the outbreak of COVID-19 may have a greater impact on us than did the first wave, depending on where infection rates are highest. To date, COVID-19 has had only a modest impact on our ability to source and manufacture our products. However, the negative consequences arising from the pandemic and governmental and societal responses thereto may be more severe the longer COVID-19 continues to circulate domestically or internationally. The ultimate impact of the COVID-19 pandemic is highly uncertain and subject to change. We do not yet know the full extent of delays or impacts on our business, our clinical trials, healthcare systems or the global economy as a whole, or how long such effects will endure. The effects of the COVID-19 pandemic or other health epidemics could have an adverse impact on our business, results of operations and financial condition.
26
We depend on our senior leadership team and may not be able to retain or replace these employees or recruit additional qualified personnel, which would harm our business, results of operations and financial condition.
Our business and success are materially dependent on attracting and retaining members of our senior leadership team to formulate and execute the Company’s business plans. Since June 2018, we have needed to add or replace a number of our senior leadership team members including our Chief Executive Officer, Chief Financial Officer, Chief Operating Officer, Chief Accounting Officer, and General Counsel and Secretary. We have experienced difficulties in recruiting due to legal and business uncertainties resulting from the issues which were the subject of the Audit Committee Investigation.
Leadership changes can be inherently difficult to manage and may cause material disruption to our business or management team. Changes in senior management could also lead to an environment that presents additional challenges in recruiting and retaining employees, which could have an adverse effect on our business, results of operations and financial condition. Our success will depend, in part, upon our ability to attract and retain skilled personnel, including sales, managerial and technical personnel. There can be no assurance that we will be able to find and attract additional qualified employees to support our expected growth or retain any such personnel. Beginning in June 2018 and continuing into 2019, we experienced higher than normal attrition in our general workforce. Our inability to hire and retain qualified personnel or the loss of services of our key personnel may have an adverse effect on our business, results of operations and financial condition.
A significant portion of our revenues and accounts receivable come from government accounts.
We have significant sales to the government (whether we are selling our products directly to government accounts or through a distributor). Any disruption of our products on the Federal Supply Schedule (“FSS”), or of the use of Indefinite Delivery, Indefinite Quantity contracts, or any change in the way the government purchases products like ours or the price it is willing to pay for our products, could adversely affect our business, results of operations and financial condition. Similarly, competitive pricing pressures and any non-compliance with applicable guidelines could cause the Company to lose existing or future contracts with the VA, which may result in an overall decline in revenue.
During 2018 and 2019, the Company conducted a comprehensive review of its pre- and post-award VA sales under its FSS contract and identified a potential issue that it self-disclosed to the VA concerning the eligibility of one of its products for inclusion in the Company’s FSS contract. The Company announced in April 2020 that it had resolved this matter for $6.5 million. See Note 16, “Commitments and Contingencies,” below. However, any resulting negative impact to our contractual relationship with the VA going forward may adversely affect our business, results of operations and financial condition.
Our revenues depend on adequate reimbursement from public and private insurers and health systems.
Our success depends on the extent to which our customers receive adequate reimbursement for the costs of our products and related treatments from third-party payers, including government healthcare programs, such as Medicare and Medicaid, as well as private insurers and health systems. Government and other third-party payers attempt to contain healthcare costs by limiting both coverage and the level of reimbursement of medical products, particularly new products. Therefore, significant uncertainty usually exists as to the reimbursement status of new healthcare products by third-party payers. Although EpiFix has coverage with the majority of payers, a significant number of public and private insurers and health systems currently do not cover or reimburse our other products. If we are not successful in obtaining adequate coverage and reimbursement for our products from these third-party payers, it could have an adverse effect on market acceptance of our products. Inadequate reimbursement levels would likely also create downward price pressure on our products. Even if we do succeed in obtaining widespread coverage and reimbursement rates or policies for our products, future changes in coverage or reimbursement rates or policies could have a negative impact on our business, financial condition and results of operations. For example, through its rule-making process, CMS has requested stakeholder comments on the reimbursement methodology under the Medicare Hospital Outpatient Prospective Payment System for an episode of wound care for future years. In other words, the Medicare reimbursement payment methodology may change after 2020 in the hospital outpatient setting from the current reimbursement methodology, which is based on a bundled payment amount per wound care application (i.e. per skin substitute application), to a fixed, global payment to treat the wound until it is healed (i.e. a lump sum payment that covers the entire wound care episode). We are unable to assess the potential effects of these reimbursement changes on our business at this time, as it is not clear if any changes will take effect and CMS has not disclosed specific reimbursement details for a wound episode model. We are and will continue to participate in discussions with CMS on potential solutions for future wound episode reimbursement models.
Further, we have experienced some reluctance by payers to cover products for applications other than those for which we have published clinical trials. For example, Noridian, the MAC for 13 states, published a Local Coverage Article effective November 8, 2018 that limits coverage for amniotic membrane derived skin substitute products to diabetic foot ulcers and venous stasis ulcers only. Prior to the published article, Noridian did not have a written policy on the matter, which provided a pathway for physicians to utilize amniotic membrane derived skin substitute products, such as ours, based on medical necessity in a wide variety of wounds.
27
Currently, there are three MACs that do not have a written medical policy in the form of a Local Coverage Determination (“LCD”) or article. If the three MACs created written medical policy criteria, this could limit providers to the use of products that have published clinical evidence for a specific wound type. As a result of the Noridian published article, our revenues for 2019 declined significantly compared to 2018. Our future revenues could experience additional declines if other MACs or other payers further limit their coverage of our products. This decline would adversely affect our business, financial condition and results of operations.
Our revenue, results of operations and cash flows may suffer upon the loss of a GPO or IDN.
As with many manufacturers in the healthcare space, the Company contracts with GPOs and IDNs to establish contracted pricing and terms and conditions for the members of GPOs and IDNs. Approximately three-quarters of our sales in the year ended December 31, 2019 came from customers that are members of our main GPOs or IDNs.
Our agreements with GPOs and IDNs allow us to sell our products efficiently to large groups of customers. Our agreements with GPOs and IDNs typically provide their members with favorable ordering terms and conditions and access to favorable product pricing. These customers purchase our product through GPO and IDN arrangements in part because of favorable pricing and terms and conditions. If our agreement with any GPO or IDN is terminated or expires without being extended, renewed or renegotiated this could adversely affect our revenue, results of operations and cash flows.
We contract with independent sales agents and distributors.
In 2019, approximately 17% of our sales through our relationships with independent agents and distributors. (Sales agents act directly on behalf of MiMedx to arrange sales, while distributors take title to product and may set their own prices.) See Note 17, “Revenue Date by Customer Type.”
Because our agents and distributors are not employees, there is a risk we will be unable to ensure that our sales processes, compliance safeguards, and related policies will be adhered to despite our communication and training of agents and distributors regarding these requirements. Further, if we fail to maintain relationships with our key independent agents, or fail to ensure that our independent agents adhere to our sales processes, compliance safeguards and related policies, there could be an adverse effect on our business, results of operations, and financial condition.
Also, if our relationships with our independent sales agents or distributors were terminated for any reason, it could materially and adversely affect our revenues and profits. Because the independent agent often controls the customer relationships within its territory, there is a risk that if our relationship with the agent ends, our relationship with the customer will be lost.
We may obtain the assistance of additional distributors and independent sales representatives to sell products in certain sales channels, particularly in territories and fields where agents are commonly used. Our success is partially dependent upon our ability to train, retain and motivate our independent sales agencies, distributors, and their representatives to appropriately and compliantly sell our products in certain territories or fields. They may not be successful in implementing our marketing plans or compliance safeguards. Some of our independent sales agencies and distributors do not sell our products exclusively and may offer similar products from other companies. Our independent sales agencies and distributors may terminate their contracts with us, may devote insufficient sales efforts to our products or may focus their sales efforts on other products that produce greater commissions for them, which could have an adverse effect on our business, results of operations and financial condition. We also may not be able to find additional independent sales agencies and distributors who will agree to appropriately and compliantly market or distribute our products on commercially reasonable terms, if at all. If we are unable to establish new independent sales representative and distribution relationships or renew current sales agency and distribution agreements on commercially acceptable terms, our business, financial condition, and results of operations could be materially and adversely affected.
Disruption of our processing could adversely affect our business, financial condition and results of operations.
Our business depends upon the continued operation of our processing facilities in Marietta, Georgia and Kennesaw, Georgia. Risks that could impact our ability to use these facilities include the occurrence of natural and other disasters, the outbreak of pandemics, and the need to comply with the requirements of directives from government agencies, including the FDA. See, for example, Item 1A, Risk Factors - The COVID-19 pandemic and governmental and societal responses thereto have adversely affected our business, results of operations and financial condition, and the continuation of COVID-19 or the outbreak of other health epidemics could harm our business, results of operations, and financial condition. Either of our processing facilities can serve as a redundant processing facility for our Section 361 products in the event the other facility experiences a disaster event. We have made efforts to transition manufacturing into compliance with cGMPs for commercial production for our Section 351 products. These efforts are concentrated at our Kennesaw, Georgia facility for tissue processing and at our Marietta, Georgia facility for upstream and downstream supply chain activities. However, the unavailability of our processing facilities could have a material adverse effect on our business, financial condition and results of operations during the period of such unavailability.
28
To be commercially successful, we must convince physicians, where appropriate, that our products are proper alternatives to existing treatments and that our products should be used in their procedures.
We believe physicians will only use our products if they determine, based on their independent medical judgment and experience, clinical data, and published peer reviewed journal articles, that the use of our products in a particular procedure is a favorable alternative to other treatments. Physicians may be hesitant to change their existing medical treatment practices for the following reasons, among others:
• | their lack of experience with advanced therapeutics, such as our placenta based allografts; |
• | lack of evidence supporting additional patient benefits of advanced therapeutics, such as our placenta-based allografts, over conventional methods in certain therapeutic applications; |
• | perceived liability risks generally associated with the use of new products and procedures; |
• | limited availability of reimbursement from third-party payers; and |
• | the time that must be dedicated to physician training in the use of our products. |
If we cannot successfully address quality issues that may arise with our products, our brand reputation could suffer, and our business, financial condition, and results of operations could be adversely impacted.
In the course of conducting our business, we must adequately address quality issues that may arise with our products, as well as defects in third-party components included in our products, as any quality issues or defects may negatively impact physician use of our products. Although we have established internal procedures to minimize risks that may arise from quality issues, we may not be able to eliminate or mitigate occurrences of these issues and associated liabilities. If the quality of our products does not meet the expectations of physicians or patients, then our brand reputation could suffer and our business could be adversely impacted. We must also ensure any promotional claims made for our products comport with government regulations.
The formation of physician-owned distributorships (“PODs”) could result in increased pricing pressure on our products or harm our ability to sell our products to physicians who own or are affiliated with those distributorships.
PODs are medical product distributors that are owned, directly or indirectly, by physicians. These physicians derive a proportion of their revenue from selling or arranging for the sale of medical products for use in procedures they perform on their own patients at hospitals that agree to purchase from or through the POD, or that otherwise furnish ordering physicians with income that is based directly or indirectly on those orders of medical products. The Office of Inspector General (“OIG”) of the Department of Health & Human Services has issued a Special Fraud Alert on PODs, indicating that they are inherently suspect under the federal Anti-Kickback Statute.
Our commercial strategy emphasizes selling directly to healthcare providers and, to a limited extent, through distributors. To our knowledge, we do not directly sell to or distribute any of our products through PODs. The number and strength of PODs in the industry may continue to grow as economic pressures increase throughout the industry and hospitals, insurers and physicians search for ways to reduce costs, and, in the case of the physicians, identify additional sources to increase their incomes. These companies and the physicians who own, or partially own, PODs have significant market knowledge, access to and influence on the physicians who use our products and the hospitals that purchase our products, and we may not be able to compete effectively for business from physicians who own PODs.
We face the risk of product liability claims and may not be able to obtain or maintain adequate product liability insurance.
Our business exposes us to the risk of product liability claims that are inherent in the manufacturing, processing and marketing of human tissue products. We may be subject to such claims if our products cause, or appear to have caused, an injury. Claims may be made by patients, healthcare providers or others selling our products. Defending a lawsuit, regardless of merit, could be costly, divert management attention and result in adverse publicity, which could result in the withdrawal of, or reduced acceptance of, our products in the market.
Although we have product liability insurance that we believe is adequate, this insurance is subject to deductibles and coverage limitations, and we may not be able to maintain this insurance. Also, it is possible that claims could exceed the limits of our coverage. If we are unable to maintain product liability insurance at an acceptable cost or on acceptable terms with adequate coverage or otherwise protect ourselves against potential product liability claims or we underestimate the amount of insurance we need, we could be exposed to significant liabilities, which may harm our business. A product liability claim or other claim with
29
respect to uninsured liabilities or for amounts in excess of insured liabilities could result in significant costs and significant harm to our business.
The products we manufacture and process are derived from human tissue and therefore have the potential for disease transmission.
The utilization of human tissue creates the potential for transmission of communicable disease, including, without limitation, human immunodeficiency virus, viral hepatitis, syphilis and other viral, fungal or bacterial pathogens. We are required to comply with federal and state regulations intended to prevent communicable disease transmission.
We maintain strict quality controls designed in accordance with cGTP to ensure the safe procurement and processing of our tissue, including terminal sterilization of our products. These controls are intended to prevent the transmission of communicable disease. However, risks exist with any human tissue implantation. We are also in the process of developing and enhancing cGMP systems to comply with the regulations that will apply to our Section 351 HCT/Ps following the end of the FDA’s enforcement discretion period under the Guidance. In addition, negative publicity concerning disease transmission from other companies’ improperly processed donated tissue could have a negative impact on the demand for our products and adversely affect our business, financial condition and results of operations.
We may implement a product recall or voluntary market withdrawal, which could significantly increase our costs, damage our reputation, disrupt our business and adversely affect our business, results of operations and financial condition.
The processing and marketing of our tissue products involves an inherent risk that our tissue products or processes do not meet applicable quality standards and requirements. In that event, we may voluntarily implement a recall or market withdrawal or may be required to do so by a regulatory authority.
For example, in March 2020, MiMedx submitted to the FDA a biological product deviation report (“BPDR”) regarding tissue recovered from four donors in Palm Beach County, Florida. These tissues were recovered by a third-party recovery partner. At the time of recovery, Palm Beach County had only just been designated as an area of active Zika transmission by the Center for Disease Control. Our recovery partner received an FDA 483 observation for recovering and providing this tissue to MiMedx in February 2020. MiMedx contacted each facility that received allografts containing the subject tissues. Following MiMedx’s submission of the BPDR to the FDA, the FDA notified MiMedx that this event meets the formal definition of a “recall” and will be classified as a Class II recall on the FDA’s recall website.
A recall or market withdrawal of one of our products could be costly and may divert management resources. A recall or withdrawal of one of our products, or a similar product processed by another entity, also could impair sales of our products as a result of confusion concerning the scope of the recall or withdrawal, or as a result of the damage to our reputation for quality and safety.
Significant disruptions of information technology systems or breaches of information security could adversely affect our business, results of operation and financial condition.
A breach of cybersecurity, a disruption in availability, or the unauthorized alteration of systems or data could adversely affect our business, results of operations and financial condition. We rely on technology for day-to-day operations as well as positioning to enhance our stance in the market. We generate intellectual property that is central to the future success of the business and transmit large amounts of confidential information. Additionally, we collect, store and transmit confidential information of customers, patients, employees and third parties. We also have outsourced significant elements of our operations to third parties, including significant elements of our information technology infrastructure, and, as a result, we are managing many independent vendor relationships with third parties who may or could have access to our confidential information. The continually changing threat landscape of cybersecurity today makes our systems potentially vulnerable to service interruptions or to security breaches from inadvertent or intentional actions by our employees, partners, and vendors, and from attacks by malicious third parties, including supply chain attacks originating at our third-party partners. Such attacks are of ever-increasing levels of sophistication. Attacks are made by individuals or groups that have varying levels of expertise, some of which are technologically advanced and well-funded including, without limitation, nation states, organized criminal groups and hacktivists organizations.
To ensure protection of our information, we have invested in cybersecurity and have implemented processes and procedural controls to maintain the confidentiality and integrity of such information. We measure these controls and their success through a cybersecurity framework that is based on industry standards. While we have invested in the protection of our data and technology, there can be no guarantees that our efforts will prevent all service interruptions or security breaches. Any such interruption or breach of our systems could adversely affect our business operations and result in the loss of critical or sensitive confidential information or intellectual property, and could result in financial, legal and reputational harm to our business, including legal claims and proceedings, liability under laws that protect the privacy of personal information, government enforcement actions and regulatory
30
penalties, as well as remediation costs. We maintain cyber liability insurance. However, this insurance may not be sufficient to cover the financial, legal or reputational losses that may result from an interruption or breach of our systems.
We may expand or contract our business through acquisitions, divestitures, licenses, investments, and other commercial arrangements in other companies or technologies, which may adversely affect our business, results of operations and financial condition.
We periodically evaluate opportunities to acquire or divest companies, divisions, technologies, products, and rights through licenses, distribution agreements, investments, and outright acquisitions to grow our business. In connection with one or more of those transactions, we may, subject to the requirements and limitations set forth in the Hayfin Loan Agreement:
• | issue additional equity securities that would dilute the value of equity currently held by our shareholders; |
• | divest or license existing products or technology; |
• | use cash that we may need in the future to operate our business; |
• | incur debt that could have terms unfavorable to us or that we might be unable to repay; |
• | structure the transaction in a manner that has unfavorable tax consequences, such as a stock purchase that does not permit a step-up in the tax basis for the assets acquired; |
• | be unable to realize the anticipated benefits, such as increased revenues, cost savings, or synergies from additional sales; |
• | be unable to secure the services of key employees related to the transaction(s); and |
• | be unable to succeed in the marketplace with the transaction(s). |
Any of these items could adversely affect our revenues, results of operations and financial condition. Business acquisitions also involve the risk of unknown liabilities associated with the acquired business, which could be material. Incurring unknown liabilities or the failure to realize the anticipated benefits of any transaction could adversely affect our business if we are unable to recover our initial investment. Inability to recover our investment, or any write off of such investment, associated goodwill or assets could have an adverse effect on our business, results of operations and financial condition.
If any of the BLAs are approved, the Company would be subject to additional regulation which will increase costs and could result in adverse sanctions for non-compliance.
Products subject to the FDA’s BLA requirements must comply with a range of pre- and post-market provisions. Pre-market compliance includes the conduct of clinical trials in support of BLA approval, the development and submission of a BLA, and the production of product for use in the clinical trials that meets FDA’s quality expectations. Post-approval requirements for BLA products include: compliance with cGMPs, which will require us to make enhancements in our fixed plant as well as incur regular costs and reduced product yields from testing products to ensure quality, identity, purity, and potency; compliance with promotional and labeling requirements, which limit our ability to make claims about regulated products; submission of annual reports in appropriate circumstances; compliance with the FDA’s “Biological Product Deviation Reporting System,” when applicable; “submission of adverse events;” reporting and correcting product problems within established timeframes; recalling or stopping the manufacture of a product if a significant problem is detected; complying with the appropriate laws and regulations relevant to the biologics license; and identifying any changes needed to help ensure product quality. In some instances, the FDA can also require that applicants conduct post-market studies or trials of the product. This additional compliance burden may increase costs, and failure to comply with such requirements may subject the Company to sanctions that would have an adverse impact on our business, results of operations and financial condition.
New lines of business or new products and services may subject us to additional risks.
From time to time, we may implement or may acquire new lines of business or offer new products and services within existing lines of business. There are risks and uncertainties associated with these efforts, particularly in instances where the markets are not fully developed or are evolving. In developing and marketing new lines of business and new products and services, we may invest significant time and resources. External factors, such as regulatory compliance obligations, competitive alternatives, and shifting market preferences, may also impact the successful implementation of a new line of business or a new product or service. Failure to successfully manage these risks in the development and implementation of new lines of business or new products or services could have an adverse effect on our business, results of operations and financial condition.
31
Our international expansion and operations outside the U.S. expose us to risks associated with international sales and operations.
We may consider further expansion outside the U.S. Managing a global organization is difficult, time consuming and expensive. Conducting international operations subjects us to risks that could be different than those faced by us in the United States. The sale and shipment of our products across international borders, as well as the purchase of components and products from international sources, subject us to extensive U.S. and foreign governmental trade, import and export and customs regulations and laws, including, without limitation, the Export Administration Regulations and trade sanctions against embargoed countries, which are administered by the Office of Foreign Assets Control within the Department of the Treasury, as well as the laws and regulations administered by the Department of Commerce. These regulations limit our ability to market, sell, distribute or otherwise transfer our products or technology to prohibited countries or persons.
International regulations on allowable promotional claims also make the promotion of our products more difficult.
Compliance with these regulations and laws is costly, and failure to comply with applicable legal and regulatory obligations could adversely affect us in a variety of ways that include, without limitation, significant criminal, civil and administrative penalties, including imprisonment of individuals, fines and penalties, denial of export privileges, seizure of shipments and restrictions on certain business activities. Also, the failure to comply with applicable legal and regulatory obligations could result in the disruption of our distribution and sales activities.
These risks may limit or disrupt our expansion, restrict the movement of funds or result in the deprivation of contractual rights or the taking of property by nationalization or expropriation without fair compensation. Operating outside of the U.S. also requires significant management attention and financial resources.
Risks Related to Regulatory Approval of Our Products and Other Government Regulations
To the extent our products do not qualify for regulation as human cells, tissues and cellular and tissue-based products solely under Section 361 of the Public Health Service Act, this could result in removal of the applicable products from the market, would make the introduction of new tissue products more expensive and would significantly delay the expansion of our tissue product offerings and subject us to additional post-market regulatory requirements.
The products we manufacture and process are derived from human tissue. Amniotic and other birth tissue is generally regulated as an HCT/P and is therefore eligible for regulation solely as a Section 361 HCT/P depending on whether the specific product at issue and the claims made for it are consistent with the applicable criteria. HCT/Ps that do not meet these criteria are subject to more extensive regulation as drugs, medical devices, biological products, or combination products. These HCT/Ps must comply with both the FDA’s requirements for HCT/Ps and the requirements applicable to biologics, devices or drugs, including pre-market clearance or approval from the FDA. Obtaining FDA pre-market clearance or approval involves significant time and investment by the Company.
In November 2017, the FDA released a guidance document entitled “Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue – Based Products: Minimal Manipulation and Homologous Use – Guidance for Industry and Food and Drug Administration Staff.” The document confirmed the FDA’s stance that all micronized amniotic products require a biologics license to be lawfully marketed in the United States. It also indicated that sheet forms of amniotic tissue are appropriately regulated as solely Section 361 HCT/Ps when manufactured in accordance with 21 CFR Part 1271 and intended for use as a barrier or covering. The final guidance also stated that the FDA intends to exercise enforcement discretion under limited conditions with respect to the IND application and pre-market approval requirements for certain HCT/Ps for a period of 36 months from the date of the guidance. The FDA’s approach is risk-based, and the guidance clarified that high-risk products and uses could be subject to immediate enforcement action. MiMedx continues to market AmnioFix Injectable and other micronized products under the policy of enforcement discretion as it works on the transition from Section 361 products to Section 351 products. Our sales of micronized products for all uses was $45.0 million, $68.4 million, and $42.4 million respectively, in 2017, 2018, and 2019. At the same time, we are pursuing the BLA pre-market approval process for certain of our micronized products, as more fully discussed under “Business – Government Regulation.”
Following the period of enforcement discretion under the Guidance, we may need to cease selling our micronized products and other products regulated under Section 351 until the FDA approves a BLA, and then we will only be able to market such products for indications that have been approved in a BLA. The loss of our ability to market and sell our micronized products would have an adverse impact on our revenues, business, financial condition and results of operations. In addition, we expect the cost to manufacture our products will increase due to the costs to comply with the requirements that apply to Section 351 biological products such as current cGMP and ongoing product testing costs. Increased costs relating to regulatory compliance could have an adverse impact on our business, financial condition and results of operations.
32
In addition, the FDA might, at some future point, modify the scope of its enforcement discretion or change its position on which current or future products qualify as Section 361 HCT/Ps, or determine that some or all of our micronized products may not be lawfully marketed under the FDA’s policy of enforcement discretion. Any regulatory changes could have adverse consequences for us and make it more difficult or expensive for us to conduct our business by requiring pre-market clearance or approval and compliance with additional post-market regulatory requirements with respect to those products. It is also possible that the FDA could decide it will not allow the Company to market any form of a micronized product during the rest of the 36-month enforcement discretion period without a biologics license, and it could even require the Company to recall its micronized products. Further, under the November 2017 guidance, the FDA expressed its expectation that following the expiration of its 36-month enforcement discretion period, sales of micronized amniotic tissue will be limited to those products and indications for which applicants have received a BLA. In April 2019, we announced that we will need more time to file and commercialize our BLAs with the FDA and that clinical trial protocol enhancements, further resources and additional capabilities and expertise will be required for commercial launch; see Item 1, “Business - Clinical Trials.” While we do not track all uses of our micronized products by physicians, we believe that our micronized product is being used by physicians for more indications than those for which we presently intend to pursue BLAs, as well as in additional sizes (e.g. 100 mg). If the FDA does allow the Company to continue to market a micronized form of its sheet allografts without a biologics license, the FDA may impose conditions, such as labeling restrictions and the requirement that the product be manufactured in compliance with cGMP. Although the Company is preparing for these requirements in connection with its pursuit of a BLA for certain of its products, earlier compliance with these conditions would require significant additional time and cost investments by the Company.
Moreover, increased regulatory scrutiny within the industry in which we operate could lead to increased regulation of HCT/Ps, including Section 361 HCT/Ps, which could ultimately increase our costs and adversely impact our business, results of operations and financial condition.
If the FDA approves the BLAs we seek, we will incur increased compliance costs on an ongoing basis. See “If any of the BLAs are approved, the Company would be subject to additional regulation which will increase costs and could result in adverse sanctions for non-compliance.”
Obtaining and maintaining the necessary regulatory approvals for certain of our products will be expensive and time consuming and may impede our ability to fully exploit our technologies.
The process of obtaining regulatory clearances or approvals to market a biological product or medical device from the FDA or similar regulatory authorities outside of the U.S. may be costly and time consuming, and such clearances or approvals may not be granted on a timely basis, or at all. We are pursuing approval of BLAs for certain of our micronized products, but have not yet submitted a BLA for review. Additionally, the FDA may take the position that some of the other products that we currently market require a BLA as well. Some of the future products and enhancements to our current products that we expect to develop and market may require marketing clearance or approval from the FDA. However, clearance or approval may not be granted with respect to any of our products or enhancements and FDA review will involve delays that may adversely affect our ability to market such products or enhancements.
The process of obtaining an approved BLA requires the expenditure of substantial time, effort and financial resources and may take years to complete. The fee for filing a BLA and program fees payable with respect to any establishment that manufactures biologics are substantial. Additionally, there are significant costs associated with clinical trials that can be difficult to accurately estimate until a BLA is approved. Clinical trials may not be successful or may return results that do not support approval. Moreover, data obtained from clinical activities are not always conclusive and may be susceptible to varying interpretations, which could delay, limit or prevent regulatory approval. The FDA may not grant approval on a timely basis, or at all, or we may decide not to pursue a BLA for certain products or indications. Additionally, the FDA may limit the indications for use or place other conditions on any approvals that could restrict the commercial application of the products. If we do receive approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further testing requirements and FDA review and approval. Our revenues will be adversely affected if we fail to obtain BLA approvals on a timely basis or at all, if the FDA requires us to stop marketing our products until a BLA is approved, or if the FDA limits the indications for use or places other conditions that restrict the commercial application of our products.
Further, in April 2019, we announced that we will need more time than we originally anticipated to file our BLAs with the FDA. Based on a review of the studies and interim results, the Company has instituted several actions with respect to its ongoing and planned clinical trials to address the resources, capabilities, and expertise needed for commercial launch including our strategy around an increased dialogue with the FDA regarding our BLA progress. For these reasons, we have increased enrollment in our current clinical trials, and will need to initiate additional clinical trials. This has added expense, time, and additional uncertainty to the overall BLA approval process. See Item 1, “Business - Clinical Trials.” If the BLAs we seek are approved, we will incur increased compliance costs on an ongoing basis. See “If any of the BLAs are approved, the Company would be subject to additional regulation which will increase costs and could result in adverse sanctions for non-compliance.”
33
Our business is subject to continuing regulatory compliance by the FDA and other authorities, which is costly, and our failure to comply could result in negative effects on our business, results of operations and financial condition.
As discussed above, the FDA has specific regulations governing our tissue-based products, or HCT/Ps. The FDA has broad post-market and regulatory and enforcement powers, even for Section 361 HCT/Ps. The FDA’s regulation of HCT/Ps includes requirements for registration and listing of products, donor screening and testing, processing and distribution, labeling, record keeping and adverse-reaction reporting, and inspection and enforcement.
HCT/Ps that are regulated as drugs, biological products or medical devices are subject to even more stringent regulation by the FDA. Even if pre-market clearance or approval is obtained, the approval or clearance may place substantial restrictions on the indications for which the product may be marketed or to whom it may be marketed, may require warnings to accompany the product or impose additional restrictions on the sale or use of the product. In addition, regulatory approval is subject to continuing compliance with regulatory standards, including the FDA’s quality system regulations.
If we fail to comply with the FDA regulations regarding our tissue products, the FDA could take enforcement action, including, without limitation, any of the following sanctions and the manufacture of our products or processing of our tissue could be delayed or terminated:
• | untitled letters, warning letters, cease and desist orders, fines, injunctions, and civil penalties; |
• | recall or seizure of our products; |
• | operating restrictions, partial suspension or total shutdown of production; |
• | refusing our requests for clearance or approval of new products; |
• | withdrawing or suspending current applications for approval or approvals already granted; |
• | refusal to grant export approval for our products; and |
• | criminal prosecution. |
The FDA’s regulation of HCT/Ps may continue to evolve. Complying with any such new regulatory requirements may entail significant time delays and expense, which could have an adverse effect on our business, results of operations and financial condition.
The American Association of Tissue Banks (“AATB”) has issued operating standards for tissue banking. Compliance with these standards is a requirement in order to become an accredited tissue bank. In addition, some states have their own tissue banking regulations.
In addition, procurement of certain human organs and tissue for transplantation is subject to the restrictions of the National Organ Transplant Act (“NOTA”), which prohibits the transfer of certain human organs, including skin and related tissue for valuable consideration, but permits the reasonable payment associated with the removal, transportation, implantation, processing, preservation, quality control and storage of human tissue and skin. We reimburse tissue banks, hospitals and physicians for their services associated with the recovery and storage of donated human tissue. Although we have independent third party appraisals that confirm the reasonableness of the service fees we pay, if we were to be found to have violated NOTA’s prohibition on the sale or transfer of human tissue for valuable consideration, we potentially would be subject to criminal enforcement sanctions, which could adversely affect our results of operations.
Finally, we and other manufacturers of skin substitutes are required to provide average selling price (“ASP”) information to CMS on a quarterly basis. The Medicare payment rates are updated quarterly based on this ASP information. If a manufacturer is found to have made a misrepresentation in the reporting of ASP, such manufacturer is subject to civil monetary penalties of up to $10,000 for each misrepresentation for each day in which the misrepresentation was applied, and potential False Claims Act liability. See We and our sales representatives, whether employees or independent contractors, must comply with various federal and state anti-kickback, self-referral, false claims and similar laws, any breach of which could cause an adverse effect on our business, results of operations and financial condition.
We may be subject to fines, penalties, injunctions and other sanctions if we are deemed to be promoting the use of our products for unapproved, or off-label, uses.
34
As a general rule, we can only market our 361 HCT/Ps for appropriate homologous uses and we can only promote pre-approved biological products or devices for FDA-approved indications. Generally, unless the products are approved by the FDA for alternative uses, the FDA contends that we may not make claims about the safety or effectiveness of our products, or promote them, for such uses. Such limitations present a risk that the FDA or other federal or state law enforcement authorities could determine that the nature and scope of our sales, marketing and support activities, though designed to comply with all FDA requirements, constitute the promotion of our products for an unapproved use in violation of the federal Food, Drug, and Cosmetic Act. We also face the risk that the FDA or other governmental authorities might pursue enforcement based on past activities that we have discontinued or changed, including sales activities, arrangements with institutions and doctors, educational and training programs and other activities.
Investigations concerning the promotion of unapproved product uses and related issues are typically expensive, disruptive and burdensome and generate negative publicity. If our promotional activities are found to be in violation of the law, we may face significant legal action, fines, penalties, and even criminal liability and may be required to substantially change our sales, promotion, grant and educational activities. There is also a possibility that we could be enjoined from selling some or all of our products for any unapproved use. In addition, as a result of an enforcement action against us or any of our executive officers, we could be excluded from participation in government healthcare programs such as Medicare and Medicaid.
However, the FDA’s Guidance stated that the FDA intends to exercise enforcement discretion under limited conditions with respect to IND application and pre-market approval requirements for certain HCT/Ps through November 2020. This means that, through November 2020, the FDA does not intend to enforce certain provisions as they currently apply to certain entities or activities. During the period of enforcement discretion, we have marketed, and intend to continue to market, our micronized products while at the same time pursuing a BLA for certain of our micronized products. We have already filed IND applications for three indications for our micronized product: plantar fasciitis, osteoarthritis knee pain, and Achilles tendonitis. We also intend to file additional INDs for both AmnioFill and for injectable micronized EpiFix for the treatment of DFUs or other areas of advanced wound care in the second half of 2020, but we have not yet initiated any clinical trials under an IND in furtherance of any regulatory approvals for these indications.
Nevertheless, while we believe we are in compliance with the FDA's Guidance on HCT/Ps and enforcement discretion regarding products that do not meet some or all of the HCT/P requirements, there can be no assurance that we are correct or that the FDA will not suspend its enforcement discretion and, in such cases, we may need to discontinue marketing a product and/or may be subject to fines, penalties, injunctions, and other sanctions if we are deemed to be promoting the use of our products for unapproved uses.
We and our sales representatives, whether employees or independent contractors, must comply with various federal and state anti-kickback, self-referral, false claims and similar laws, any breach of which could cause an adverse effect on our business, results of operations and financial condition.
Our relationships with physicians, hospitals and other healthcare providers are subject to various federal and state healthcare fraud and abuse laws. Healthcare fraud and abuse laws are complex and, in some instances, even minor or inadvertent violations can give rise to liability. Possible sanctions for violation of the healthcare fraud and abuse laws include, without limitation, monetary fines, civil and criminal penalties, exclusion from participating in the federal and state healthcare programs, including, without limitation, Medicare, Medicaid, the Department of Veterans Affairs (“VA”) health programs and TRICARE (the healthcare program administered by or on behalf of the U.S. Department of Defense for uniformed service members, including both those in active duty and retirees, as well as their dependents), and forfeiture of amounts collected in violation of such prohibitions. Many states have similar fraud and abuse laws, imposing substantial penalties for violations. A finding of a violation of one or more of these laws, or even a government investigation or inquiry into the same, would likely result in a material adverse effect on the market price of our Common Stock, as well as on our business, results of operations, and financial condition.
The federal Anti-Kickback Statute is a criminal law that prohibits, among other things, any person from knowingly and willfully offering, paying, soliciting or receiving remuneration, directly or indirectly, in cash or in kind, to induce or reward referrals, purchases or orders or arranging for or recommending the purchase, order or referral of any item or service for which payment may be made in whole or in part by a federal healthcare program, such as the Medicare and Medicaid programs. The term “remuneration” has been broadly interpreted to include anything of value. The Patient Protection and Affordable Care Act (the “PPACA”) amended the federal Anti-Kickback Statute to clarify the intent that is required to prove a violation. Under the federal Anti-Kickback Statute as amended, a person or entity need not have actual knowledge of this statute or specific intent to violate it. The PPACA also amended the federal Anti-Kickback Statute to provide that any claims for items or services resulting from a violation of the federal Anti-Kickback Statute are considered false or fraudulent for purposes of the federal False Claims Act (“FCA”). A conviction for violation of the Anti-Kickback Statute results in criminal fines and requires mandatory exclusion from participation in federal health care programs. Although there are a number of statutory exceptions and regulatory safe harbors to the federal Anti-Kickback Statute that protect certain common industry practices from prosecution, the exceptions and safe harbors
35
are drawn narrowly, and arrangements may be subject to scrutiny or penalty if they do not fully satisfy all elements of an available exception or safe harbor. We have entered into consulting agreements, speaker agreements, research agreements and product development agreements with physicians, including some who may order or recommend our products or make decisions to use them. In addition, some of these physicians own our stock, which they purchased in arm’s-length transactions on terms identical to those offered to non-physicians, or received stock awards from us in the past as consideration for services performed by them. While we believe these transactions generally meet the requirements of applicable laws, including the federal Anti-Kickback Statute and analogous state laws, it is possible that our arrangements with physicians and other providers may be questioned by regulatory or enforcement authorities under such laws, which could lead us to redesign the arrangements and subject us to significant civil or criminal penalties. We have designed our policies and procedures to comply with the federal Anti-Kickback Statute, FCA, and industry best practices. In addition, we have conducted training sessions on these principles. If, however, regulatory or enforcement authorities were to view these arrangements as non-compliant with applicable laws, there would be risk of government investigations/inquiries or penalties. There is also risk that one or more of our employees or agents will disregard the rules we have established. Because our strategy relies on the involvement of physicians who consult with us on the design of our products, perform clinical research on our behalf or educate other health care professionals about the efficacy and uses of our products, we could be materially impacted if regulatory or enforcement agencies or courts interpret our financial relationships with physicians who refer or order our products to be in violation of applicable laws. This could harm our reputation and the reputations of the physicians we engage to provide services on our behalf. In addition, the cost of noncompliance with these laws could be substantial since we could be subject to monetary fines and civil or criminal penalties, and we could also be excluded from federally-funded healthcare programs, including Medicare, Medicaid, VA and TRICARE.
The FCA imposes civil liability on any person or entity that knowingly submits, or causes the submission of, a false or fraudulent claim to the U.S. government. Damages under the FCA can be significant and consist of the imposition of fines and penalties. The FCA also allows a private individual or entity to sue on behalf of the government to recover civil penalties and treble damages as a whistleblower. FCA liability is potentially significant in the healthcare industry because the statute provides for treble damages and mandatory penalties of between $11,181 and $22,363 per false claim or statement for penalties assessed after January 29, 2018, with respect to violations occurring after November 2, 2015.
Manufacturers can be held liable under the FCA even when they do not submit claims directly to government payers if they are deemed to “cause” the submission of false or fraudulent claims. The PPACA provides that claims tainted by a violation of the federal Anti-Kickback Statute are false for purposes of the FCA. The Department of Justice (the “DOJ”) on behalf of the government has previously alleged that the marketing and promotional practices of pharmaceutical and medical device manufacturers, including the off-label promotion of products or the payment of prohibited kickbacks to doctors, violated the FCA, resulting in the submission of improper claims to federal and state healthcare programs such as Medicare and Medicaid. In certain cases, manufacturers have entered into criminal and civil settlements with the federal government under which they entered into plea agreements, paid substantial monetary amounts and entered into onerous corporate integrity agreements with the government that require, among other things, substantial reporting and remedial actions, as well as oversight and review by an outside entity, an Independent Review Organization (“IRO”), at substantial expense to the Company.
Under the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”) criminal federal healthcare fraud statute, it is a crime to knowingly and willfully execute, or attempt to execute, a scheme or artifice to defraud any health care benefit program or to obtain, by means of false or fraudulent pretenses, representations or promises, any of the money or property owned by, or under the custody or control of, any health care benefit program, in connection with the delivery of or payment for health care benefits, items or services.
There are federal and state laws requiring detailed reporting of manufacturer interactions with and payments to healthcare providers, such as the federal Physician Payments Sunshine Act (“Sunshine Act”). The Sunshine Act requires, among others, “applicable manufacturers” of drugs, devices, biological products, and medical supplies reimbursed under Medicare, Medicaid or the Children’s Health Insurance Program to annually report to CMS information related to payments and other transfers of value provided to “covered recipients.” The term covered recipients includes U.S.-licensed physicians and teaching hospitals, and, for reports submitted on or after January 1, 2022, physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, and certified nurse-midwives. While manufacturers of human cell and tissue products regulated solely under Section 361 are not subject to the Sunshine Act, in the future, if we receive a BLA, we will be subject to this law. There is also risk that CMS or another government agency may take the position that our products are not human cell and tissue products regulated solely under Section 361, and thereby assert that we are currently subject to the Sunshine Act, which could subject us to civil penalties and the administrative burden of having to comply with the law.
There are state law equivalents to the Anti-Kickback Statute and FCA. There are also so-called state “all-payer” anti-kickback laws which may apply to items or services reimbursed by any third-party payer, including commercial insurers, as well as when no insurer is involved (i.e. cash-pay patients).
36
The enforcement of all of these laws is uncertain and subject to rapid change. Federal or state regulatory or enforcement authorities may investigate or challenge our current or future activities under these laws. Any investigation or challenge could have a material adverse effect on our business, financial condition and results of operations. Any state or federal regulatory or enforcement review of us, regardless of the outcome, would be costly and time consuming. Additionally, we cannot predict the impact of any changes in these laws, whether these changes are retroactive or will have effect on a going-forward basis only.
We may be subject to fines, penalties, injunctions and even criminal sanctions if we are deemed to have made a misstatement of compliance to a federal agency.
Products that are subject to pre-approval as biologicals must also be manufactured in accord with cGMP. In August 2013, the FDA sent the Company an Untitled Letter asserting that its micronized amniotic allografts were unapproved biologics. The Company disputed the FDA’s position at the time and filed various appeals but ultimately agreed during the appeals process to pursue BLAs for certain products, but the transition to cGMP compliance for micronized products sold commercially was a larger task. In February 2016, the FDA inspected the Company’s Marietta facility against cGMP requirements for the commercially available product. The transition to cGMP compliance was underway, but the work was in its initial stages. At the close of the inspection, the FDA issued a Form 483 that included 13 observations. In response, the Company developed an action plan (the “Action Plan”). The Action Plan, which was shared with FDA, called for a systematic approach to the work and provided a vehicle to update the FDA on progress. Over the course of the next year, the site did substantial work to transition to cGMP for the commercially available, micronized product and filed several updates with the FDA.
In February 2017, the Company sent a close-out letter to the FDA that indicated the work under the Action Plan had been completed. That letter overstated our state of compliance in regard to the commercially available product. The goal of the letter was to communicate the substantial progress to the FDA and to indicate that the work under the Action Plan had been completed. The site continues to transition to cGMP compliance for its micronized products, and we expect to complete the work by November 2020 when the FDA’s industry-wide exercise of enforcement discretion for products like our micronized allografts expires. Exaggeration or misstatement of compliance to a federal agency creates regulatory risk. If the government were to take issue with the letter, it could take any number of actions adverse to the Company. These include issuing a warning letter, terminating the current exercise of enforcement discretion with respect to the sale of micronized products and initiating a civil judicial action against the Company and opening a criminal investigation. Each of these potential actions would be disruptive to the Company’s operations, consume considerable resources and potentially prohibit sales of certain products and adversely affect our business, financial condition and results of operations.
In July 2019, the Company formally notified the FDA that its February 2017 correspondence overstated the Company’s state of cGMP compliance.
In December 2019, the FDA conducted a cGMP audit of each of the Company’s two manufacturing facilities. At the close of the inspection the FDA issued two Form 483s (one for each facility). The Company timely responded to the Form 483s. See the discussion under “Item 1. Business - Processing (Manufacturing).”
Our results of operations may be adversely affected by current and potential future healthcare reforms.
In response to perceived increases in healthcare costs in recent years, there have been and continue to be proposals by the U.S. federal government, state governments, regulators and third-party payers to control these costs and, more generally, to reform the U.S. healthcare system. In the U.S., the PPACA was enacted in 2010 with a goal of reducing the cost of healthcare and substantially changing the way healthcare is financed by both government and private insurers.
In addition, other legislative changes have been proposed and adopted in the U.S. since the PPACA was enacted. The Budget Control Act of 2011 created measures for spending reductions by Congress. A Joint Select Committee on Deficit Reduction, tasked with recommending a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, was unable to reach required goals, thereby triggering the legislation’s automatic reduction to several government programs. This included aggregate reductions of Medicare payments to providers of 2% per fiscal year, which went into effect on April 1, 2013. In January 2013, the American Taxpayer Relief Act was signed into law, which, among other things, further reduced Medicare payments to several provider types, including hospitals.
The current U.S. Presidential Administration and certain members of the U.S. Congress have stated that they will seek to modify, repeal or otherwise invalidate all, or certain provisions of, the PPACA. In 2017, the U.S. President signed an executive order which stated that it is the policy of his Administration to seek the prompt repeal of the PPACA and directed executive departments and federal agencies to waive, defer, grant exemptions from or delay the implementation of the provisions of the PPACA to the maximum extent permitted by law. Additionally, the House and Senate attempted, but failed, to pass legislation to repeal all or portions of the PPACA, and these efforts may be resumed. In December 2017, the U.S. President signed the Tax Cuts and Jobs Act, which,
37
among numerous other actions, repealed the individual mandate of the PPACA, effective on January 1, 2019. In December 2018, a federal district court in Texas ruled the individual mandate was unconstitutional and could not be severed from the PPACA. As a result, the court ruled the remaining provisions of the PPACA were also invalid, though the court declined to issue a preliminary injunction with respect to the PPACA. The court’s ruling was appealed to the U.S. Court of Appeals for the Fifth Circuit. On March 25, 2019, the DOJ reversed its prior position and stated in a legal filing with the Fifth Circuit that the district court’s ruling that the PPACA was invalid should be upheld. In December 2019, the Fifth Circuit agreed that the individual mandate was unconstitutional, but remanded the case back to the district court to reassess how much of the PPACA would be damaged without the individual mandate provision, and if the individual mandate could indeed be severed. In January 2020, 21 state Attorneys General urged the Supreme Court of the United States to decide whether or not the PPACA should be struck down as unconstitutional, claiming that the Fifth Circuit erroneously remanded the case to the district court. The House of Representatives filed a similar petition and motion. The state Attorneys General and the House of Representatives also filed motions to expedite the Supreme Court’s decision to review the case, which the Supreme Court subsequently denied. This litigation is still ongoing, and places great uncertainty upon the longevity and nature of the PPACA moving forward. In addition, further legislative changes to and regulatory changes under PPACA remain possible.
There is uncertainty with respect to the impact the U.S. Administration, the executive order, and the attempted legislation may have, if any, and any changes will likely take time to unfold and could have an impact on coverage and reimbursement for healthcare items and services, including our products. We believe that substantial uncertainty remains regarding the net effect of the PPACA, or its repeal and potential replacement, on our business, including uncertainty over how benefit plans purchased on exchanges will cover our products, how the expansion or contraction of the Medicaid program will affect access to our products, the effect of risk-sharing payment models such as Accountable Care Organizations and other value-based purchasing programs on coverage for our product, and the effect of the general increase or decrease in federal oversight of healthcare payers. The taxes imposed and the expansion in government’s role in the U.S. healthcare industry under the PPACA, if unchanged, may result in decreased revenues, lower reimbursements by payers for our products and reduced medical procedure volumes, all of which could have a material adverse effect on our business, results of operations and financial condition.
We may fail to obtain or maintain foreign regulatory approvals to market our products in other countries.
We currently market our products internationally and intend to consider expansion of our international marketing. International jurisdictions require separate regulatory approvals and compliance with numerous and varying regulatory requirements. The approval procedures vary among countries and may involve requirements for additional testing. Certain of our products require clearance or approval by the FDA. However, such clearance or approval does not ensure approval or certification by regulatory authorities in other countries or jurisdictions, and approval or certification by one foreign regulatory authority does not ensure approval or certification by regulatory authorities in other foreign countries or by the FDA. The foreign regulatory approval or certification process may include all of the risks associated with obtaining FDA clearance or approval. We may not obtain foreign regulatory approvals on a timely basis, if at all. We may not be able to file for regulatory approvals or certifications and may not receive necessary approvals to commercialize our products in any foreign jurisdiction. Furthermore, many foreign jurisdictions operate under socialized medical care, and obtaining reimbursement for our products under that construct may also prove difficult. If we fail to receive necessary approvals, certifications, or reimbursements necessary to commercialize our products in foreign jurisdictions on a timely basis, or at all, our business, results of operations and financial condition could be adversely affected.
Federal and state laws that protect the privacy and security of personal information may increase our costs and limit our ability to collect and use that information and subject us to liability if we are unable to fully comply with such laws.
Numerous federal and state laws, rules and regulations govern the collection, dissemination, use, security and confidentiality of personal information, including protected health information and individually identifiable health information. These laws include:
• | provisions of HIPAA that limit how covered entities and business associates may use and disclose protected health information, provide certain rights to individuals with respect to that information and impose certain security requirements; |
• | HITECH, which strengthened and expanded the HIPAA Privacy Rule and Security Rules, imposed data breach notification obligations, created new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates and gave state attorneys general new authority to file civil actions for damages or injunctions in U.S. federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions; |
• | other federal and state laws restricting the use and protecting the privacy and security of personal information, including health information, many of which are not preempted by HIPAA; |
• | federal and state consumer protection laws; and |
38
• | federal and state laws regulating the conduct of research with human subjects. |
One relevant state law is the California Consumer Protection Act (“CCPA”), which became effective on January 1, 2020. The CCPA is a privacy bill that requires certain companies doing business in California to disclose information regarding the collection and use of a consumer’s personal data and to delete a consumer’s data upon request. The Act also permits the imposition of civil penalties and expands existing state security laws by providing a private right of action for consumers in certain circumstances where consumer data is subject to a breach. We are still evaluating whether and how this rule will impact our U.S. operations and /or limit the ways in which we can provide services or use personal data collected while providing services.
As part of our business operations, including our medical record keeping, third-party billing and reimbursement and research and development activities, we collect and maintain protected health information in paper and electronic format. Standards related to health information, whether implemented pursuant to HIPAA, HITECH, state laws, federal or state action or otherwise, could have a significant effect on the manner in which we handle personal information, including healthcare-related data, and communicate with payers, providers, patients, donors and others, and compliance with these standards could impose significant costs on us or limit our ability to offer services, thereby negatively impacting the business opportunities available to us.
If we are alleged not to comply with existing or new laws, rules and regulations related to personal information, we could be subject to litigation and to sanctions that include monetary fines, civil or administrative penalties, civil damage awards or criminal penalties.
Risks Related to Our Intellectual Property
Our ability to protect our intellectual property and proprietary technology through patents and other means is uncertain and may be inadequate, which could have an adverse effect on our business, results of operations and financial condition.
Our success depends significantly on our ability to protect our proprietary rights to the technologies used in our products. We rely on patent protection, as well as a combination of copyright, trade secret and trademark laws and nondisclosure, confidentiality and other contractual restrictions to protect our proprietary technology, including our licensed technology. These legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep any competitive advantage. In addition, our pending patent applications include claims to material aspects of our products and procedures that are not currently protected by issued patents. The patent application process can be time consuming and expensive. Our pending patent applications might not result in issued patents. Competitors may be able to design around our patents or develop products that provide outcomes that are comparable or even superior to ours. Although we have taken steps to protect our intellectual property and proprietary technology, including entering into confidentiality agreements and intellectual property assignment agreements with some of our officers, employees, consultants and advisors, such agreements may not be enforceable or may not provide meaningful protection for our trade secrets or other proprietary information in the event of unauthorized use or disclosure or other breaches of the agreements.
The failure to obtain and maintain patents or protect our intellectual property rights could have an adverse effect on our business, results of operations, and financial condition. Whether a patent claim is valid is a complex matter of science, facts and law, and therefore we cannot be certain that, if challenged, our patent claims would be upheld. If any of those patent claims are invalidated, our competitive advantage may be reduced or eliminated.
In the event a competitor infringes upon our licensed patents, issued patents, pending patent applications or other intellectual property rights, enforcing those rights may be costly, uncertain, difficult and time consuming. Even if successful, litigation to enforce or defend our intellectual property rights could be expensive and time consuming and could divert our management’s attention. Further, bringing litigation to enforce our patents subjects us to the potential for counterclaims. Other companies or entities also have commenced, and may again commence, actions seeking to establish the invalidity of our patents and certain related claims. In the event that any of our patent claims are challenged, a court, the United States Patent and Trademark Office (“USPTO”), or the Patent Trial and Appeal Board (“PTAB”) of the USPTO may invalidate one or more challenged patent claims or determine that the patent is unenforceable, which could harm our competitive position. If the USPTO or the PTAB ultimately cancels or narrows the claim scope of any of our patents through these proceedings, it could prevent or hinder us from being able to enforce them against competitors. Such adverse decisions could negatively impact our business, results of operations, and financial condition. See Item 3, “Legal Proceedings” for information regarding our ongoing patent infringement lawsuits and related inter partes review proceedings.
In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the United States. Many companies have encountered significant problems in enforcing and defending intellectual property rights in certain foreign jurisdictions. This could make it difficult for us to stop infringement of our foreign patents, if obtained, or the misappropriation of our other intellectual property rights. For example, some foreign countries have compulsory licensing laws
39
under which a patent owner must grant licenses to third parties. In addition, some countries limit the enforceability of patents against third parties, including government agencies or government contractors. In these countries, patents may provide limited or no benefit. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time-consuming process with uncertain outcomes. Accordingly, we may choose not to seek patent protection in certain countries, and we will not have the benefit of patent protection in such countries. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business. Accordingly, our efforts to protect our intellectual property rights in some countries may be inadequate.
We may become subject to claims of infringement of the intellectual property rights of others, which could prohibit us from developing our products, require us to obtain licenses from third parties or to develop non-infringing alternatives, and subject us to substantial monetary damages.
Third parties could assert that our products infringe their patents or other intellectual property rights. Whether a product infringes a patent claim or other intellectual property right involves a complex combination of legal and factual issues, the determination of which is often uncertain. Therefore, we cannot be certain that we have not infringed the intellectual property rights of others. Because patent applications may take years to issue, there also may be applications now pending of which we are unaware that may later result in issued patent claims that our products or processes infringe. There also may be existing patents or pending patent applications of which we are unaware that our products or processes may inadvertently infringe.
Any infringement claim could cause us to incur significant costs, place significant strain on our financial resources, divert management’s attention from our business and harm our reputation. If the relevant patent claims at issue in such a dispute were upheld as valid and enforceable and we were found to infringe, we could be prohibited from selling any product that is found to infringe those claims unless we could obtain licenses to use the technology covered by the asserted patent claims or other intellectual property, or are able to design around the patent claim or claims at issue or other intellectual property. We may be unable to obtain such a license on terms acceptable to us, if at all, and we may not be able to redesign our products to avoid infringement. A court could also order us to pay compensatory damages for such infringement, plus prejudgment interest and could, in addition, treble the compensatory damages and award attorney fees. These damages could be substantial and could harm our reputation, business, financial condition and operating results. A court also could enter orders that temporarily, preliminarily or permanently enjoin us and our customers from making, using, or selling products, and could enter an order mandating that we undertake certain remedial measures. Depending on the nature of the relief ordered by the court, we could become liable for additional damages to third parties.
We may be subject to damages resulting from claims that we, our employees, or our independent contractors have wrongfully used or disclosed alleged trade secrets, proprietary or confidential information of our competitors or are in breach of non-competition or non-solicitation agreements with our competitors.
Some of our employees were previously employed at other medical device, pharmaceutical or tissue companies. We may also hire additional employees who are currently employed at other medical device, pharmaceutical or tissue companies, including our competitors. Additionally, consultants or other independent agents with which we may contract may be or have been in a contractual arrangement with one or more of our competitors. Although no claims are currently pending, we may be subject to claims that we, our employees, or our independent contractors have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of these former employers or competitors. In addition, we have been and may in the future be subject to claims that we caused an employee to breach the terms of his or her non-competition or non-solicitation agreement. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management. If we fail to defend such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Any future litigation or the threat thereof may adversely affect our ability to hire additional direct sales representatives. A loss of key personnel or their work product could hamper or prevent our ability to market existing or new products, which could severely harm our business, financial condition and operating results.
Risks Related to the Audit Committee Investigation, Consolidated Financial Statements, Internal Controls and Related Matters
We have identified material weaknesses in our internal control over financial reporting, and we have concluded that our internal control over financial reporting and our disclosure controls and procedures were not effective as of December 31, 2019. If we fail to properly remediate these or any future material weaknesses or deficiencies, further material misstatements in our financial statements could occur and impair our ability to produce accurate and timely financial statements, preclude us from relisting our stock on a securities exchange, require significant expenditure of financial and other resources, give rise to litigation against us and otherwise affect our business, financial condition and operating results.
40
We have concluded that our internal control over financial reporting was not effective as of December 31, 2019 due to the existence of material weaknesses in such controls and we have also concluded that our disclosure controls and procedures were not effective as of December 31, 2019 due to material weaknesses in our control over financial reporting, all as described in Item 9A, “Controls and Procedures,” of this Form 10-K.While we continued meaningful remediation efforts during 2019 to address the identified weaknesses, we were not able to fully remediate our material weaknesses in internal controls as of December 31, 2019. In addition, one or more additional material weaknesses in our internal control over financial reporting might arise or be identified in the future. We intend to continue our control remediation activities and, in doing so, we will continue to incur expenses and expend management time on compliance-related issues.
If our remediation measures are insufficient to address the identified deficiencies, or if additional deficiencies in our internal control over financial reporting are discovered or occur in the future, our consolidated financial statements may contain material misstatements and we could be required to restate our financial results. Moreover, because of the inherent limitations of any control system, material misstatements due to error or fraud may not be prevented or detected on a timely basis, or at all. If we are unable to provide reliable and timely financial reports in the future, our business and reputation may be further harmed. Failures in internal controls may also cause us to fail to meet reporting obligations, negatively affect investor confidence in our management and the accuracy of our financial statements and disclosures, or result in adverse publicity and concerns from investors, any of which could have a negative effect on the price of our Common Stock, subject us to further regulatory investigations and penalties or shareholder litigation, and adversely impact our business, results of operations and financial condition.
Matters relating to and arising out of the Audit Committee Investigation, including the accounting review of our previously issued consolidated financial statements and the audits of fiscal years 2018, 2017 and 2016, have been time consuming and expensive, and may result in additional expense.
We incurred significant expenses in connection with the Investigation, and we are continuing to incur significant expenses, including audit, legal, consulting and other professional fees, in connection with the ongoing review of our accounting practices and systems, the audit of our financial statements and the remediation of deficiencies in our internal control over financial reporting. Specifically, in connection with the Audit Committee Investigation, audit and compliance efforts and related litigation, the Company incurred Investigation, Restatement and related expenses in the aggregate amount of approximately $60.5 million and $51.3 million for the years ended December 31, 2019 and 2018, respectively. We expect to incur expenses in 2020 despite the conclusion of the Investigation and completion of the Restatement, because litigation involving the Company and/or its former officers and directors remains unsettled, and we are obligated to advance the costs of defense to our current and former officers and directors in those matters. See Note 16, “Commitments and Contingencies.” To the extent our remediation efforts are unsuccessful or incomplete, or we identify additional problems requiring remediation, our management may be required to devote significant additional time to such efforts and we may be forced to incur significant additional expenses, including legal and accounting expenses. The incurrence of significant additional expense, or the requirement that management devote significant time that could reduce the time available to execute on our business strategies, could have an adverse effect on our business, results of operations and financial condition.
Matters relating to or arising from the Restatement and the Audit Committee Investigation have had and could continue to have an adverse effect on our business, results of operations and financial condition.
We have been and could continue to be the subject of negative publicity focusing on the Restatement and the results of the Investigation. As a result, our customers and others with whom we do business have voiced concerns regarding the effort required to address our accounting and control environment and our ability to be a long-term provider to our customers. Further negative publicity could adversely affect our business, financial condition and results of operations.
We are currently, and may in the future be, subject to substantial litigation and ongoing investigations that could cause us to incur significant legal expenses and result in harm to our business.
We are exposed to potential liabilities and reputational risk associated with litigation, regulatory proceedings and government enforcement actions. See Item 3, “Legal Proceedings” and Note 16, “Commitments and Contingencies” in the Consolidated Financial Statements for information regarding proceedings that we believe may be material to the Company as of the date of the filing of this Form 10-K. In addition, we are obligated to indemnify and advance expenses to certain individuals involved in certain of these proceedings. Further, volatility in our stock price may also make us vulnerable to future class action litigation.
Any adverse judgment in or settlement of any pending or any future litigation could result in payments, fines and penalties that could adversely affect our business, results of operations and financial condition. Regardless of the outcome, legal proceedings have resulted in, and may continue to result in, significant legal fees and expenses, diversion of management’s time and other resources, and adverse publicity. Such proceedings could also adversely affect our business, results of operations and financial condition.
41
Our Common Stock might not be relisted, or once relisted, it might not remain listed.
Because we are not current in filing our periodic reports with the SEC, we were unable to comply with the listing standards of Nasdaq, and our Common Stock was suspended from trading on The Nasdaq Capital Market effective November 8, 2018 and was subsequently delisted effective March 8, 2019. We have taken initial steps to relist our Common Stock. However, we may not be able to complete the requirements to relist in an expeditious manner or at all. Even if our Common Stock is relisted, an active trading market may not develop or, if one develops, may not continue. The lack of an active trading market may limit the liquidity of an investment in our Common Stock, meaning you may not be able to sell any shares of Common Stock you own at times, or at prices, attractive to you. Any of these factors may adversely affect the price of our Common Stock.
Risks Related to the Securities Markets and Ownership of Our Common Stock
EW Healthcare Partners may have influence over us, and its interests may conflict with those of our other shareholders.
On July 2, 2020, we issued 90,000 shares of Series B Preferred Stock to an affiliate of EW Healthcare Partners (“EW Healthcare Partners”) pursuant to the Securities Purchase Agreement. As of July 2, 2020, EW Healthcare Partners and their affiliates own 90% of the outstanding shares of Series B Preferred Stock which would result, upon conversion, in an ownership interest of approximately 17.2% of our Common Stock (calculated on the basis set forth under Item 12, “Security Ownership Of Certain Beneficial Owners And Management” below). Also, for as long as EW Healthcare Partners and its affiliates collectively hold at least (i) 10% of the outstanding shares of our Common Stock (calculated on an as converted basis), EW Healthcare Partners has the right to designate two directors to our board and (ii) (ii) 5% (but less than 10%) of the outstanding shares of our outstanding Common Stock (calculated on an as converted basis), EW Healthcare Partners has the right to designate one individual to serve on our Board. Such individuals will initially be Preferred Directors and therefore not subject to election by the holders of Common Stock. At the closing of the Preferred Stock Transaction, EW Healthcare Partners designated Martin P. Sutter and William A. Hawkins, III to serve on our board as preferred directors, and they were appointed to our Board on July 2, 2020. The interests of EW Healthcare Partners may conflict with those of our other shareholders, and EW Healthcare Partners may seek to influence, and may be able to influence, us through its director designation rights and its share ownership.
Holders of shares of Series B Preferred Stock have rights, preferences and privileges that are not held by, and are preferential to, the rights of, our common shareholders.
Holders of shares of Series B Preferred Stock are entitled to cumulative dividends at a rate of 4.0% per annum until July 2, 2021 and 6.0% per annum thereafter, in each case compounding quarterly in arrears. The dividends are payable quarterly in whole or in part, in cash. However, the Company may, at its option, elect to not pay any such dividend and to instead accrue the amount of such dividend. The payment of regular dividends in cash to the holders of Series B Preferred Stock could impact our liquidity and reduce the amount of cash available for working capital, capital expenditures, growth opportunities, acquisitions, and other general corporate purposes. If we elect to accrue the dividends in lieu of paying them in cash, holders of Common Stock could effectively be diluted because such accrual of dividends will increase the number of shares of Common Stock into which the Series B Preferred Stock would then be convertible. Our obligations to the holders of Series B Preferred Stock could also limit our ability to obtain additional equity or debt financing or increase our borrowing costs, which could have an adverse effect on our financial condition.
The Series B Preferred Stock ranks senior to our Common Stock with respect to dividends and distributions on liquidation, winding-up, and dissolution. Upon a liquidation, dissolution, or winding-up of the Company, each share of Series B Preferred Stock will be entitled to receive $1,000 per share (subject to adjustment), plus any accrued and unpaid dividends. This amount will be payable prior to any distribution of our available assets to the holders of our Common Stock.
Holders of Series B Preferred Stock generally are entitled to vote together as a single class with the holders of the shares of Common Stock, on an as converted basis, on all matters submitted for a vote of holders of our Common Stock subject to certain limitations on their voting rights contained in the related Articles of Amendment. Additionally, certain matters will require the approval of the holders of the majority of the outstanding shares of Series B Preferred Stock, voting as a separate class, including the following:
• | any changes to the rights, preferences, or privileges of the Series B Preferred Stock; |
• | amendments or restatements of any organizational document of the Company or its subsidiaries in a manner that materially, adversely, and disproportionately affects the rights, preferences, and privileges of the Series B Preferred Stock as compared to our Common Stock; |
• | the authorization or creation of any class or series of senior or parity equity securities; |
• | the declaration of any dividends or any other distributions, or the repurchase or redemption, of any equity securities of the Company ranking junior to or on parity with the Series B Preferred Stock (subject to certain exceptions); |
42
• | prior to January 2, 2023, the sale, transfer, or other disposition of any assets, business, or operations for $25 million or more (other than sales of inventory in the ordinary course of business), or the purchase or acquisition of any assets, business, or operations for $75 million or more; |
• | prior to January 2, 2023, the merger or consolidation of the Company unless either (x) the surviving company will have no class of equity securities ranking superior to or on parity with the Series B Preferred Stock or (y) the holders of shares of the Series B Preferred Stock will receive in connection therewith consideration per share of Series B Preferred Stock valued at 200% or more of the purchase price per share of $1,000; |
• | prior to January 2, 2023, commencing a voluntary case under any applicable bankruptcy, insolvency, or other similar law or consenting to the entry of an order for relief in an involuntary case under any such law, or effectuating any general assignment for the benefit of creditors; and |
• | prior to January 2, 2023, entering into any settlement agreement regarding the Company’s securities class action litigation. |
The interests of our holders of Series B Preferred Stock and our Common Stock may conflict in certain circumstances, and these provisions may constrain the Company from taking certain actions that may be in the best interest of its holders of Common Stock.
The conversion price of the Series B Preferred Stock is subject to anti-dilution adjustments in the event that the Company sells or issues Common Stock to any third-party investor at any time prior to July 2, 2022 at a price that is less than $3.85 per share of Common Stock (although such adjustments cannot result in a conversion price for the Series B Preferred Stock of less than $3.47). Additionally, as long as EW Healthcare Partners holds at least 10% of our outstanding Common Stock (calculated on an as converted basis), it has certain preemptive rights to participate in offerings of Common Stock to any person, subject to customary exceptions.
Furthermore, in the event that the Company undergoes a change of control, the holders of Series B Preferred Stock will have certain redemption rights, which, if exercised, could require us to repurchase all of the outstanding shares of Series B Preferred Stock for cash at the original purchase price of Series B Preferred Stock plus all accrued and unpaid dividends thereon. Any required repurchase of the outstanding Series B Preferred Stock could impact our liquidity and reduce the amount of cash available for working capital, capital expenditures, growth opportunities, acquisitions, and other general corporate purposes.
The preferential rights of the Series B Preferred Stock could also result in divergent interests between the holders of Series B Preferred Stock and our common shareholders.
See Item 9B - “Other Information” for more information regarding our Series B Preferred Stock.
Our Series B Preferred Stock is convertible into shares of our Common Stock, and any such conversion may dilute the value of our Common Stock.
Holders of shares of Series B Preferred Stock have the right, at their option, to convert each share of Series B Preferred Stock into shares of our Common Stock, except that no holder may convert its shares of Series B Preferred Stock into shares of Common Stock if such conversion would result in such holder and its affiliates holding more than 19.9% of the aggregate voting power of our Common Stock or beneficially owning in excess of 19.9% of our then-outstanding shares of Common Stock. Additionally, each share of Series B Preferred Stock (including any accrued and unpaid dividends) will automatically convert into shares of our Common Stock at any time after July 2, 2022, provided that our Common Stock has traded at 200% or more of the then conversion price for 20 out of 30 consecutive trading days preceding, and as of the close of trading on the date immediately prior to conversion. The conversion of Series B Preferred Stock may significantly dilute our common shareholders and adversely affect both our net income per share of Common Stock and the market price of our Common Stock.
Our Common Stock has been delisted from The Nasdaq Capital Market, which may negatively impact the trading price of our Common Stock and the levels of liquidity available to our shareholders.
The trading of our Common Stock was suspended from the Nasdaq Capital Market in November 2018 and delisted in March 2019. It is currently quoted on the “over the counter” market operated by the OTC Markets Group, Inc. under the symbol “MDXG,” which may negatively impact the trading price of our Common Stock and the liquidity available to our shareholders.
Our Common Stock is subject to SEC rules and regulations relating to the market for penny stocks. A penny stock is any equity security not traded on a national securities exchange that has a market price of less than $5.00 per share. On June 15, 2020, the last sale price per share of our Common Stock as reported on the OTC Markets was $3.65. If our Common Stock is or becomes subject to regulation as a penny stock, such regulations may severely affect the market liquidity for our Common Stock and could limit the ability of shareholders to sell securities in the secondary market. Accordingly, investors in our Common Stock may find it more difficult to dispose of or obtain accurate quotations as to the market value of our Common Stock, and there can be no
43
assurance that our Common Stock will continue to be eligible for trading or quotation on the over the counter market or any other alternative exchanges or markets.
Further, the delisting of our Common Stock from The Nasdaq Capital Market may adversely affect our ability to raise additional capital through public or private sales of equity securities, may significantly affect the ability of investors to trade our securities and may negatively affect the value and liquidity of our Common Stock. Such delisting may also have other negative effects, including the potential loss of confidence of employees, the loss of institutional investor interest, and fewer business development opportunities. Furthermore, because of the limited market and low volume of trading in our Common Stock that could occur, the share price of our Common Stock could be disproportionately affected by broad market fluctuations, general market conditions, fluctuations in our operating results, changes in the market’s perception of our business and announcements made by us, our competitors, parties with whom we have business relationships or third parties.
The price of our Common Stock has been, and will likely continue to be, volatile.
The market price of our Common Stock, like that of the securities of many other healthcare companies that are engaged in research, development, and commercialization, has fluctuated over a wide range, and it is likely that the price of our Common Stock will fluctuate in the future. The market price of our Common Stock could be impacted by a variety of factors, including:
• | Fluctuations in stock market prices and trading volumes of similar companies or of the markets generally; |
• | Our ability to successfully launch, market and earn significant revenue from our products; |
• | Our ability to obtain additional financing to support our continuing operations; |
• | Disclosure of the details and results of regulatory applications and proceedings; |
• | Developments in and disclosure or publicity regarding existing or new litigation or contingent liabilities; |
• | Changes in government regulations or our failure to comply with any such regulations; |
• | Additions or departures of key personnel; |
• | Our investments in research and development or other corporate resources; |
• | Announcements of technological innovations or new commercial products by us or our competitors; |
• | Developments in the patents or other proprietary rights owned or licensed by us or our competitors; |
• | The timing of new product introductions; |
• | Actual or anticipated fluctuations in our operating results, including any restatements of previously reported results; |
• | Our ability to effectively and consistently manufacture our products and avoid costs associated with the recall of defective or potentially defective products; |
• | Our ability and the ability of our distribution partners to market and sell our products; |
• | Changes in reimbursement for our products or the price for our products to our customers; |
• | Removal of our products from the FSS, or changes in how government accounts purchase products such as ours or in the price for our products to government accounts; |
• | Material amounts of short-selling of our Common Stock; and |
• | The other risks detailed in this Item 1A. |
Price volatility or a decrease in the market price of our Common Stock could have an adverse effect on our ability to raise capital, liquidity, business, financial condition and results of operations.
Fluctuations in revenue or results of operations could cause additional volatility in our stock price.
Any unanticipated shortfall in our revenue in any fiscal quarter could have an adverse effect on our results of operations in that quarter. The effect on our net income of such a shortfall could be exacerbated by the relatively fixed nature of most of our costs,
44
which primarily include personnel costs as well as facilities costs. These fluctuations could cause the trading price of our stock to be negatively affected. Our quarterly operating results have varied substantially in the past and may vary substantially in the future.
We do not intend to pay cash dividends on our Common Stock.
Holders of our Series B Preferred Stock are entitled to contractually-determined dividends before holders of our Common Stock. See Holders of shares of Series B Preferred Stock have rights, preferences and privileges that are not held by, and are preferential to, the rights of, our common shareholders.
We have never declared or paid cash dividends on our Common Stock. We currently expect to use available funds and any future earnings to pay dividends on the Series B Preferred Stock; in the development, operation and expansion of our business; to repay debt; and, to the extent authorized by our Board, repurchasing our Common Stock. We do not anticipate paying any cash dividends on our Common Stock in the foreseeable future. As a result, capital appreciation, if any, of our Common Stock will be an investor’s only source of potential gain from our Common Stock for the foreseeable future.
Certain provisions of Florida law and anti-takeover provisions in our organizational documents may discourage or prevent a change of control, even if an acquisition would be beneficial to shareholders, which could affect our share price adversely and prevent attempts by shareholders to remove current management.
The Florida Business Corporation Act (the “FBCA”) includes several provisions applicable to the Company that may discourage potential acquirors. Such provisions include provisions that:
• | allow directors to take other stakeholders into account in discharging their duties; |
• | a requirement that certain transactions with a shareholder of 10% or more ownership must be approved by the affirmative vote of two-thirds of the other shareholders unless approved by a majority of the disinterested directors or certain fair price requirements are met; and |
• | voting rights acquired by a shareholder at ownership levels at or above one-fifth, one-third and a majority of voting power are denied unless authorized by the Board prior to such acquisition or by a majority of the other shareholders (excluding interested shares (as defined in the FBCA)). |
Additionally, our organizational documents contain provisions: authorizing the issuance of blank check preferred stock; restricting persons who may call shareholder meetings; providing for a classified Board; permitting shareholders to remove directors only “for cause” and only by super-majority vote; and providing the Board with the exclusive right to fill vacancies and to fix the number of directors. These provisions of Florida law and our articles of incorporation and bylaws could negatively affect our share price, prevent attempts by shareholders to remove current management, prohibit or delay mergers or other takeovers or changes of control of the Company and discourage attempts by other companies to acquire us, even if such a transaction would be beneficial to our shareholders.
Item 1B. Unresolved Staff Comments
There are no unresolved SEC Staff comments with respect to our SEC filings.
Item 2. Properties
Our corporate headquarters are located in Marietta, Georgia, where we lease office, laboratory, tissue processing and warehouse space. We also lease a facility in Kennesaw, Georgia, which primarily consists of laboratory, tissue processing and warehouse space, and additional warehouse space in Marietta, Georgia. All of our properties are used by our one business segment, Regenerative Biomaterials, which includes the design, manufacture and marketing of products and tissue processing services for the wound care, burn, surgical, orthopedic, spine, sports medicine, ophthalmic and dental sectors of healthcare.
The Company’s properties are suitable and adequate for current business operations. We are making investments and exploring alternatives to increase our manufacturing capacity, especially in the context of enhancements to facilitate the processing of products required to be manufactured under cGMP.
45
Item 3. Legal Proceedings
Shareholder Derivative Suits
On December 6, 2018, the United States District Court for the Northern District of Georgia entered an order consolidating three shareholder derivative actions (Evans v. Petit, et al. filed September 25, 2018, Georgalas v. Petit, et al. filed September 27, 2018, and Roloson v. Petit, et al. filed October 22, 2018) that had been filed in the Northern District of Georgia. On January 22, 2019, plaintiffs filed a verified consolidated shareholder derivative complaint. The consolidated action sets forth claims of breach of fiduciary duty, corporate waste and unjust enrichment against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Joseph G. Bleser, J. Terry Dewberry, Charles R. Evans, Larry W. Papasan, Luis A. Aguilar, Bruce L. Hack, Charles E. Koob, Neil S. Yeston and Christopher M. Cashman. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Company filed a motion to stay on February 18, 2019, pending the completion of the investigation by the Company’s Special Litigation Committee. The Special Litigation Committee completed its investigation relating to this action and filed an executive summary of its findings with the Court on July 1, 2019. The parties (together with parties from the Hialeah derivative lawsuit, the Nix and Demaio derivative lawsuit, and the Murphy derivative lawsuit, each described below) held a mediation on February 11, 2020. Following continued discussions, on May 1, 2020, the parties notified the Court that plaintiffs and the Company had reached an agreement in principle to settle this consolidated derivative action, which settlement also encompasses all claims asserted in the Hialeah derivative lawsuit, the Nix and Demaio derivative lawsuit, and the Murphy derivative lawsuit. As of the date of the filing of this Form 10-K, the parties are drafting, and intend to file, a stipulation of settlement and motion seeking preliminary approval of the settlement.
On October 29, 2018, the City of Hialeah Employees Retirement System (“Hialeah”) filed a shareholder derivative complaint in the Circuit Court for the Second Judicial Circuit in and for Leon County, Florida (the “Florida Court”). The complaint alleges claims for breaches of fiduciary duty and unjust enrichment against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Joseph G. Bleser, J. Terry Dewberry, Charles R. Evans, Bruce L. Hack, Charles E. Koob, Larry W. Papasan, and Neil S. Yeston. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Company moved to stay the action on February 7, 2019, to allow the prior-filed consolidated derivative action in the Northern District of Georgia to be resolved first and to allow the Company’s Special Litigation Committee time to complete its investigation. The Company also filed a motion to dismiss on April 8, 2019. As discussed above, the plaintiff participated in the mediation that took place in connection with the prior-filed consolidated derivative action in the Northern District of Georgia and is a party to the agreement in principle to settle that consolidated derivative action. The agreement in principle provides that the plaintiff in this action will file a notice of dismissal to dismiss its action with prejudice within seven calendar days after the date that the judgment entered by the Northern District of Georgia becomes final.
On May 15, 2019, two individuals purporting to be shareholders of the Company filed a shareholder derivative complaint in the Superior Court for Cobb County, Georgia. (Nix and Demaio v. Evans, et al.) The complaint alleges claims for breaches of fiduciary duty, corporate waste and unjust enrichment against certain current and former directors and officers of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Chris Cashman, Lou Roselli, Mark Diaz, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Court ordered this matter stayed pending the resolution of the consolidated derivative suit pending in the Northern District of Georgia. As discussed above, the plaintiff participated in the mediation that took place in connection with the prior-filed consolidated derivative action in the Northern District of Georgia and is a party to the agreement in principle to settle that consolidated derivative action. The agreement in principle provides that the plaintiffs in this action will file a notice of dismissal to dismiss their action with prejudice within seven calendar days after the date that the judgment entered by the Northern District of Georgia becomes final.
46
On August 12, 2019, John Murphy filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida (Murphy v. Petit, et al.). The complaint alleged claims for breaches of fiduciary duty and unjust enrichment against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Company filed a motion to transfer this action to the Northern District of Georgia. Prior to resolution of that motion, the plaintiff voluntarily dismissed this action without prejudice. As discussed above, the plaintiff participated in the mediation that took place in connection with the prior-filed consolidated derivative action in the Northern District of Georgia and is a party to the agreement in principle to settle that consolidated derivative action. Under the agreement in principle, the plaintiff has agreed that this action shall not be reinstated and, after the judgment entered by the Northern District of Georgia becomes final, this action shall be deemed dismissed with prejudice.
On February 10, 2020, Charles Pike filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida (Pike v. Petit, et al.). The complaint alleges claims for breaches of fiduciary duty against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. Similar to the prior-filed actions discussed above, the allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. On May 12, 2020, prior to the Company’s time to respond to the complaint, the plaintiff filed a notice of voluntary dismissal of this action without prejudice.
On February 18, 2020, Bruce Cassamajor filed a shareholder derivative complaint in the United States District Court for the Northern District of Florida (Cassamajor v. Petit, et al.). The complaint alleges claims for breaches of fiduciary duty against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. Similar to the prior-filed actions discussed above, the allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. On May 22, 2020, prior to service of the complaint, the plaintiff filed a notice of voluntary dismissal of this action without prejudice. On May 26, 2020, the court ordered this case to be dismissed for failure to serve process.
Securities Class Action
On January 16, 2019, the United States District Court for the Northern District of Georgia entered an order consolidating two purported securities class actions (MacPhee v. MiMedx Group, Inc., et al. filed February 23, 2018 and Kline v. MiMedx Group, Inc., et al. filed February 26, 2018). The order also appointed Carpenters Pension Fund of Illinois as lead plaintiff. On May 1, 2019, the lead plaintiff filed a consolidated amended complaint, naming as defendants the Company, Michael J. Senken, Parker H. Petit, William C. Taylor, Christopher M. Cashman and Cherry Bekaert & Holland LLP. The amended complaint (the “Securities Class Action Complaint”) alleged violations of Section 10(b) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), Rule 10b-5 promulgated thereunder and Section 20(a) of the Exchange Act. It asserted a class period of March 7, 2013 through June 29, 2018. Following the filing of motions to dismiss by the various defendants, the lead plaintiff was granted leave to file an amended complaint. The lead plaintiff filed its amended complaint against the Company, Michael Senken, Pete Petit, William Taylor, and Cherry Bekaert & Holland (Christopher Cashman was dropped as a defendant) on March 30, 2020; defendants filed motions to dismiss on May 29, 2020.
47
Investigations
SEC Investigation
On April 4, 2017, the Company received a subpoena from the SEC requesting information related to, among other things, the Company’s recognition of revenue, practices with certain distributors and customers, its internal accounting controls and certain employment actions. The Company cooperated with the SEC in its investigation (the “SEC Investigation”). In November 2019, the SEC brought claims against the Company and the Company’s former officers Parker H. Petit, Michael J. Senken, and William C. Taylor. The SEC alleged that from 2013 to 2017, the Company prematurely recognized revenue from sales to its distributors and exaggerated its revenue growth. The SEC’s complaint also alleged that the Company improperly recognized revenue because its former CEO and COO entered into undisclosed side arrangements with certain distributors. These side arrangements allowed distributors to return product to the Company or conditioned distributors’ payment obligations on sales to end users. The SEC complaint further alleged that the Company’s former CEO, COO, and CFO allegedly covered up their scheme for years, including after the Company’s former controller raised concerns about the Company’s accounting for specific distributor transactions. The SEC also alleged that the Company’s former CEO, COO, and CFO all misled the Company’s outside auditors, members of the Company’s Audit Committee, and outside lawyers who inquired about these transactions. The SEC brought claims against the Company and its former CEO, COO, and CFO for violating the antifraud, reporting, books and records, and internal controls provisions of the federal securities laws. The SEC also brought claims against the Company’s former CEO, COO, and CFO for lying to the Company’s outside auditors. In November 2019, without admitting or denying the SEC’s allegations, the Company settled with the SEC by consenting to the entry of a final judgment that permanently restrains and enjoins the Company from violating certain provisions of the federal securities laws. As part of the resolution, the Company paid a civil penalty of $1.5 million. The settlement concluded, as to the Company, the matters alleged by the SEC in its complaint. The SEC’s litigation continues against the Company’s former officers.
United States Attorney’s Office for the Southern District of New York (“USAO-SDNY”) Investigation
The USAO-SDNY conducted an investigation into topics similar to those at issue in the SEC Investigation. The USAO-SDNY requested that the Company provide it with copies of all information the Company furnished to the SEC and made additional requests for information. The USAO-SDNY conducted interviews of various individuals, including employees and former employees of the Company. The USAO-SDNY issued indictments in November 2019 against former executives Messrs. Petit and Taylor for securities fraud and conspiracy to commit securities fraud, to make false filings with the SEC, and improperly influence the conduct of audits relating to alleged misconduct that resulted in inflated revenue figures for fiscal 2015. The Company is cooperating with the USAO-SDNY.
Department of Veterans’ Affairs Office of Inspector General (“VA-OIG”) and Civil Division of the Department of Justice (“DOJ-Civil”) Subpoenas and/or Investigations
VA-OIG has issued subpoenas to the Company seeking, among other things, information concerning the Company’s financial relationships with VA clinicians. DOJ-Civil has requested similar information. The Company has cooperated fully and produced responsive information to VA-OIG and DOJ-Civil. Periodically, VA-OIG has requested additional documents and information regarding payments to individual VA clinicians. Most recently, on June 3, 2020, the Company received a subpoena from the VA-OIG requesting information regarding the Company’s financial relationships and interactions with two healthcare providers at the VA Long Beach Healthcare System. The Company has continued to cooperate and respond to these requests.
As part of its cooperation, the Company provided documents in response to subpoenas concerning its relationship with three now former VA employees in South Carolina, who were ultimately indicted in May 2018. Among other things, the indictment referenced speaker fees paid by the Company to the former VA employees and other interactions between now former Company employees and the former VA employees. In January 2019, prosecution was deferred for 18 months to allow the three former VA employees to enter and complete a Pretrial Diversion Program, the completion of which would result in the dismissal of the indictment. As far as the Company is aware, two of the former VA employees have completed the program early and the indictment has been dismissed with respect to them. To date, no actions have been taken against the Company with respect to this matter.
United States Attorney’s Office for the Middle District of North Carolina (“USAO-MDNC”) Investigation
On January 9, 2020, the USAO-MDNC informed the Company that it is investigating the Company’s financial relationships with two former clinicians at the Durham VA Medical Center. The Company is cooperating with the investigation.
Qui Tam Actions
48
On January 19, 2017, a former employee of the Company filed a qui tam False Claims Act complaint in the United States District Court for the District of South Carolina (United States of America, ex rel. Jon Vitale v. MiMedx Group, Inc.) alleging that the Company’s donations to the patient assistance program, Patient Access Network Foundation, violated the Anti-Kickback Statute and resulted in submission of false claims to the government. The government declined to intervene and the complaint was unsealed on August 10, 2018. The Company filed a motion to dismiss on October 1, 2018. The Company’s motion to dismiss was granted in part and denied in part on May 15, 2019. The case is in discovery.
On January 20, 2017, two former employees of the Company, filed a qui tam False Claims Act complaint in the United States District Court for the District of Minnesota (Kruchoski et. al. v. MiMedx Group, Inc.). An amended complaint was filed on January 27, 2017. The operative complaint alleges that the Company failed to provide truthful, complete and accurate information about the pricing offered to commercial customers in connection with the Company’s Federal Supply Schedule contract. On May 7, 2019, the Department of Justice (“DOJ”) declined to intervene, and the case was unsealed. In April 2020, without admitting the allegations, the Company agreed to pay $6.5 million to the DOJ to resolve this matter.
Former Employee Litigation
On December 13, 2016, the Company filed a complaint in the Circuit Court for Palm Beach County, Florida (MiMedx Group, Inc. v. Academy Medical, LLC et. al.) alleging several claims against a former employee, primarily based on his alleged competitive activities while he was employed by the Company (breach of contract, breach of fiduciary duty and breach of duty of loyalty). The former employee countersued for monetary damages and injunctive relief, alleging whistleblower retaliation in violation of the Dodd-Frank Wall Street Reform and Consumer Protection Act (the “Dodd-Frank Act”), unlawful discharge and defamation. The Court dismissed the Dodd-Frank Act whistleblower counterclaim, and in response, the former employee filed an amended complaint on September 11, 2018, adding allegations of post-termination retaliation in violation of the Dodd-Frank Act. The court dismissed the former employee’s retaliation counterclaim on January 24, 2019. After this dismissal, only the former employee’s claims of unlawful discharge and defamation remained pending. The parties resolved this matter and the case was dismissed on September 5, 2019.
On December 29, 2016, the Company filed a complaint in the United States District Court for the Northern District of Illinois (MiMedx Group, Inc. v. Michael Fox) alleging several claims against a former employee of the Company, primarily based on his alleged competitive activities while he was employed by the Company (breach of contract, breach of fiduciary duty and breach of duty of loyalty). The former employee countersued the Company for monetary damages and injunctive relief, alleging improper wage rate adjustment, interference with the former employee’s job after his termination from the Company and retaliation. The parties resolved this matter and the case was dismissed on November 4, 2019.
On July 13, 2018, a former employee filed a complaint against the Company in the United States District Court for the Northern District of Texas (Jennifer R. Scott v. MiMedx Group, Inc.), alleging sex discrimination and retaliation. The parties resolved this matter, and the case was dismissed on November 6, 2019.
On November 19, 2018, the Company’s former Chief Financial Officer filed a complaint in the Superior Court for Cobb County, Georgia (Michael J. Senken v. MiMedx Group, Inc.) in which he claims that the Company has breached its obligations under the Company’s charter and bylaws to advance to him, and indemnify him for, his legal fees and costs that he incurred in connection with certain Company internal investigations and litigation. The Company filed its answer denying the plaintiff’s claims on April 19, 2019. To date, no deadlines have been established by the court.
On January 21, 2019, a former employee filed a complaint in the Fifth Judicial Circuit, Richland County, South Carolina (Jon Michael Vitale v. MiMedx Group, Inc. et. al.) against the Company alleging retaliation, defamation and unjust enrichment and seeking monetary damages. The former employee claims he was retaliated against after raising concerns related to insurance fraud and later defamed by comments concerning the indictments of three South Carolina VA employees. On February 19, 2019, the case was removed to the U.S. District Court for the District of South Carolina. The Company filed a motion to dismiss on April 8, 2019, which was denied by the Court. This case is in discovery.
In December 2019, MiMedx received notice of a complaint filed in July 2018 with the Occupational Safety and Health Administration (“OSHA”) section of the Department of Labor (“DOL”) by Thomas Tierney, a former Regional Sales Director, against MiMedx and the referenced individuals, Tierney v. MiMedx Group, Inc., Parker Petit, William Taylor, Christopher Cashman, Thornton Kuntz, Jr. and Alexandra Haden, DOL No. 4-5070-18-243. Mr. Tierney alleged that he was terminated from MiMedx in retaliation for reporting concerns about revenue recognition practices, compliance issues, and the corporate culture, in violation of the anti-retaliation provisions of the Sarbanes-Oxley Act. The parties settled this matter and OSHA dismissed the complaint on May 20, 2020.
Defamation Claims
49
On June 4, 2018, Sparrow Fund Management, LP (“Sparrow”) filed a complaint against the Company and Mr. Petit, including claims for defamation and civil conspiracy in the United States District Court for the Southern District of New York (Sparrow Fund Management, L.P. v. MiMedx Group, Inc. et. al.). The complaint seeks monetary damages and injunctive relief and alleges the defendants commenced a campaign to publicly discredit Sparrow by falsely claiming it was a short seller who engaged in illegal and criminal behavior by spreading false information in an attempt to manipulate the price of our Common Stock. On March 31, 2019, a judge granted defendants’ motions to dismiss in full, but allowed Sparrow the ability to file an amended complaint. The Magistrate has recommended Sparrow’s motion for leave to amend be granted in part and denied in part and the Judge adopted the Magistrate’s recommendation. Sparrow filed its amended complaint against MiMedx (Mr. Petit has been dropped from the lawsuit) on April 3, 2020 and the Company filed its answer. This case is in discovery.
On June 17, 2019, the principals of Viceroy Research (“Viceroy”), filed suit in the Circuit Court for the Seventeenth Judicial Circuit in Broward County, Florida (Fraser John Perring et. al. v. MiMedx Group, Inc. et. al.) against the Company and Mr. Petit, alleging defamation and malicious prosecution based on the defendants’ alleged campaign to publicly discredit Viceroy and the lawsuit the Company previously filed against the plaintiffs, but which the Company subsequently dismissed without prejudice. On November 1, 2019, the Court granted Mr. Petit’s motion to dismiss on jurisdictional grounds, denied the Company’s motion to dismiss, and granted plaintiffs leave to file an amended complaint to address the deficiencies in its claims against Mr. Petit, which they did on November 21, 2019. The Company filed its answer on December 20, 2019.
Intellectual Property Litigation
The Bone Bank Action
On May 16, 2014, the Company filed a patent infringement lawsuit against Transplant Technology, Inc. d/b/a Bone Bank Allografts (“Bone Bank”) and Texas Human Biologics, Ltd. (“Biologics”) in the United States District Court for the Western District of Texas (MiMedx Group, Inc. v. Tissue Transplant Technology, LTD. d/b/a/ Bone Bank Allografts et. al.). The Company has asserted that Bone Bank and Biologics infringed certain of the Company’s patents through the manufacturing and sale of their placental-derived tissue graft products, and the Company is seeking permanent injunctive relief and unspecified damages. On July 10, 2014, Bone Bank and Biologics filed an answer to the complaint, denying the allegations in the complaint, and filed counterclaims seeking declaratory judgments of non-infringement and invalidity. The matter settled in 2019 prior to trial, and the case was dismissed on April 4, 2019.
The NuTech Action
On March 2, 2015, the Company filed a patent infringement lawsuit against NuTech Medical, Inc. (“NuTech”) and DCI Donor Services, Inc. (“DCI”) in the United States District Court for the Northern District of Alabama (MiMedx Group, Inc. v. NuTech Medical, Inc. et. al.). The Company has alleged that NuTech and DCI infringed and continue to infringe the Company’s patents through the manufacture, use, sale and/or offering of their tissue graft product. The Company has also asserted that NuTech knowingly and willfully made false and misleading representations about its products to customers and prospective customers. The Company is seeking permanent injunctive relief and unspecified damages. The case was stayed pending the restatement of the Company’s financial statements. Since the Company has completed its restatement, the case has resumed and discovery has recommenced.
The Osiris Action
On February 20, 2019, Osiris Therapeutics, Inc. (“Osiris”) refiled its trade secret and breach of contract action against the Company (which had been dismissed in a different forum) in the United States District Court for the Northern District of Georgia (Osiris Therapeutics, Inc. v. MiMedx Group, Inc.). Osiris has alleged that the Company acquired Stability, a former distributor of Osiris, in order to illegally obtain trade secrets. On February 24, 2020, the Court issued an order granting in part and denying in party MiMedx’s motion to dismiss. The Court dismissed Osiris’s claims for tortious interference, conspiracy to breach contract, unfair competition, and conspiracy to commit unfair competition. The Court denied MiMedx’s motion to dismiss with respect to the claim for breach of the contract between Osiris and Stability, finding that there is a question as to whether Osiris can maintain such a claim by piercing the corporate veil between MiMedx and its former subsidiary. If Osiris cannot pierce the corporate veil, the claim against MiMedx fails; if Osiris can pierce the corporate veil, the breach of contract claim must be brought in an arbitration proceeding. MiMedx did not move to dismiss Osiris’s claims for misappropriation of trade secrets and conspiracy to misappropriate trade secrets. MiMedx plans to defend against all remaining claims.
50
Other Matters
In addition to the matters described above, the Company is a party to a variety of other legal matters that arise in the ordinary course of the Company’s business, none of which is deemed to be individually material at this time. Due to the inherent uncertainty of litigation, there can be no assurance that the resolution of any particular claim or proceeding would not have a material adverse effect on the Company’s business, results of operations, financial position or liquidity.
Item 4. Mine Safety Disclosures
Not applicable.
51
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market for Common Stock
Our Common Stock trades on the “over the counter” market operated by the OTC Markets Group Inc. (the “OTC Market”) under the symbol “MDXG.” The OTC Market quotations reflect inter-dealer prices, without retail markup, mark-down or commission and may not represent actual transactions. Previously, our Common Stock traded on Nasdaq under the symbol “MDXG.” Due to our inability to file periodic reports with the SEC, we were not able to comply with Nasdaq listing standards, and our Common Stock was suspended from trading on Nasdaq and subsequently delisted, effective on March 8, 2019.
Based upon information supplied from our transfer agent, there were approximately 1,179 shareholders of record of our Common Stock as of June 25, 2020.
We have not paid any cash dividends and do not anticipate paying any cash dividends on our Common Stock in the foreseeable future.
Information required by this Item regarding equity compensation plans is contained in our Proxy Statement under the caption “Equity Compensation Plan Information,” and is incorporated herein by reference.
Stock Performance Graph
The following graph compares the cumulative total stockholder return on our Common Stock with the cumulative total stockholder return of the Nasdaq Composite Index and the Nasdaq Biotechnology Index, assuming an investment of $100.00 on December 31, 2014, in each of our Common Stock, the stocks comprising the Nasdaq Composite Index, and the stocks comprising the Nasdaq Biotechnology Index.
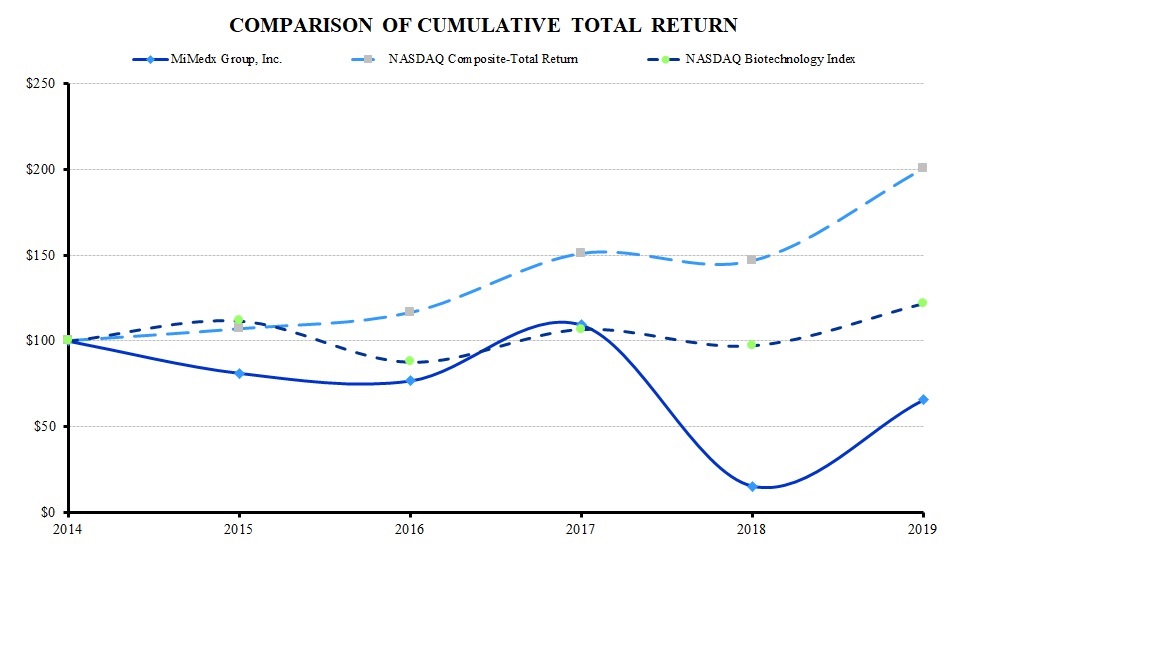
ASSUMES $100 INVESTED ON DEC. 31, 2014
ASSUMES NO DIVIDENDS
FISCAL YEAR ENDED DEC. 31, 2019
52
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
The following table sets forth information regarding the purchases of the Company’s equity securities made by or on behalf of the Company or any affiliated purchaser (as defined in Rule 10b-18 under the Exchange Act) during the three-month period ended December 31, 2019.
Period | Total Number of Shares Purchased | Average Price Paid per Share | Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs | Approximate Dollar Value of Shares that May Yet Be Purchased Under Plans or Programs | ||||||
October 1, 2019 - October 31, 2019 | 30,506 | $ | 5.05 | — | $ | — | ||||
November 1, 2019 - November 31, 2019 | 204 | $ | 5.59 | — | $ | — | ||||
December 1, 2019 - December 31, 2019 | 5,068 | $ | 6.70 | — | $ | — | ||||
Total for the quarter (1) | 35,778 | $ | 5.34 | — | $ | — | ||||
(1) Shares repurchased during the quarter include only shares surrendered by employees to satisfy tax withholding obligations upon vesting of restricted stock.
53
Item 6. Selected Financial Data
The selected consolidated financial data displayed below for the years ended December 31, 2019, 2018, and 2017 was derived from our audited consolidated financial statements for the three-year period ended December 31, 2019. As described below, the selected financial data as of and for the years ended December 31, 2016 and 2015 have been derived from our restated audited consolidated financial statements, which reflect the impact of adjustments to, or restatement of, our previously filed financial information, including a January 1, 2014 cumulative effect adjustment to stockholders’ equity to correct for accounting errors in periods prior to January 1, 2014. The selected financial data set forth below is not necessarily indicative of results of future operations, and should be read in conjunction with Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the Consolidated Financial Statements.
Year Ended December 31, in thousands | |||||||||||||||||||||
2019 (1) (3) | 2018 (3) | 2017 (2) (4) | 2016 (2) | 2015 | |||||||||||||||||
Statement of Operations Data: | |||||||||||||||||||||
Net sales | $ | 299,255 | $ | 359,111 | $ | 321,139 | $ | 221,712 | $ | 153,131 | |||||||||||
Gross profit | 256,174 | 322,725 | 285,920 | 190,774 | 137,579 | ||||||||||||||||
Operating (loss) income | (21,160 | ) | (3,924 | ) | 46,223 | 884 | (5,880 | ) | |||||||||||||
Net (loss) income | (25,580 | ) | (29,979 | ) | 64,727 | 390 | (16,354 | ) | |||||||||||||
Net (loss) income per common share - basic | (0.24 | ) | $ | (0.28 | ) | $ | 0.61 | $ | 0.00 | $ | (0.15 | ) | |||||||||
Net (loss) income per common share - diluted | $ | (0.24 | ) | $ | (0.28 | ) | $ | 0.56 | $ | 0.00 | $ | (0.14 | ) | ||||||||
(1) 2019 includes the adjustments discussed in Item 8, Note 3 “Revenue Recognition.”
(2) Includes the following:
• | Sales to external customers by Stability Biologics, LLC, our wholly-owned subsidiary acquired on January 13, 2016 and sold on September 30, 2017, were $7.0 million and $11.7 million during the years ended December 31, 2017 and 2016, respectively. |
(3) Includes legal fees, forensic audit fees, and consulting fees relating to the Restatement; and legal fees relating to the SEC Investigation, shareholder derivative lawsuits, and other litigation, as well as settlements made with former employees.
• | Investigation, restatement and related expenses were $66.5 million in 2019 as compared with $51.3 million in 2018; |
• | As a result of the December 2018 broad-based organizational realignment, cost reduction and efficiency program, the Company incurred pre-tax charges of $6.1 million during 2018. |
(4) Includes the following:
• | Loss on sale of Stability Biologics, LLC of $1.0 million recognized during the year ended December 31, 2017 and further discussed in Item 8, Note 4 “Stability Biologics, LLC.” |
For information regarding the comparability of the financial data presented in the tables above and factors that may impact comparability of future results, see Item 7. “Management’s Discussion and Analysis of Financial Condition and Results of Operations” as well as the Consolidated Financial Statements.
54
As of December 31, in thousands | |||||||||||||||||||||
2019 | 2018 | 2017 | 2016 | 2015 | |||||||||||||||||
Balance Sheet Data: | |||||||||||||||||||||
Cash and cash equivalents | $ | 69,069 | $ | 45,118 | $ | 27,476 | $ | 30,321 | $ | 26,301 | |||||||||||
Short term investments | — | — | — | — | 3,000 | ||||||||||||||||
Accounts receivable, net | 32,327 | — | — | 1,927 | — | ||||||||||||||||
Inventory, net | 9,104 | 15,986 | 9,467 | 15,872 | 7,460 | ||||||||||||||||
Prepaid expenses | 6,669 | 6,673 | 2,125 | 1,838 | 945 | ||||||||||||||||
Income tax receivable | 18 | 454 | 656 | — | — | ||||||||||||||||
Other current assets | 6,058 | 5,818 | 9,023 | 9,516 | 7,260 | ||||||||||||||||
Total current assets | 123,245 | 74,049 | 48,747 | 59,474 | 44,966 | ||||||||||||||||
Total assets | $ | 167,166 | $ | 122,844 | $ | 121,255 | $ | 117,274 | $ | 69,560 | |||||||||||
Current portion of long term debt | $ | 3,750 | $ | — | $ | — | $ | — | $ | — | |||||||||||
Accounts payable | 8,710 | 14,864 | $ | 8,454 | 12,412 | $ | 6,987 | ||||||||||||||
Accrued compensation | 21,302 | 23,024 | 20,941 | 12,691 | 15,276 | ||||||||||||||||
Accrued expenses | 32,161 | 31,842 | 15,768 | 19,207 | 9,679 | ||||||||||||||||
Current portion of earn out liability | — | — | — | 8,260 | — | ||||||||||||||||
Deferred tax liability | — | — | — | 1,129 | 803 | ||||||||||||||||
Income taxes | — | — | — | 5,611 | 410 | ||||||||||||||||
Other current liabilities | 1,399 | 1,817 | 647 | 1,482 | 533 | ||||||||||||||||
Total current liabilities | 67,322 | 71,547 | 45,810 | 60,792 | 33,688 | ||||||||||||||||
Long term liabilities | 65,446 | 1,642 | 1,648 | 8,415 | 1,148 | ||||||||||||||||
Additional paid in capital | 147,231 | 164,744 | 164,649 | 161,481 | 163,438 | ||||||||||||||||
Accumulated deficit | (102,140 | ) | (76,560 | ) | (46,581 | ) | (111,308 | ) | (111,698 | ) | |||||||||||
Total stockholders' equity | 34,398 | 49,655 | 73,797 | 48,067 | 34,724 | ||||||||||||||||
Total liabilities and stockholders' equity | $ | 167,166 | $ | 122,844 | $ | 121,255 | $ | 117,274 | $ | 69,560 | |||||||||||
Working capital | 55,923 | 2,502 | 2,937 | (1,318 | ) | 11,278 | |||||||||||||||
55
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
Overview
MiMedx is an industry leader in advanced wound care and an emerging therapeutic biologics company, developing and distributing placental tissue allografts with patent-protected processes for multiple sectors of healthcare. We derive our products from human placental tissues processed using our proprietary processing methodologies, including the PURION® process. We employ aseptic processing techniques in addition to terminal sterilization to produce our allografts. MiMedx provides products in the wound care, burn, surgical, orthopedic, spine, sports medicine, ophthalmic, and dental sectors of healthcare. Our mission is to offer products and tissues to help the body heal itself. All of our products are regulated by the FDA.
MiMedx is the leading supplier of human placental allografts, which are human tissues that are transplanted from one person (a donor) to another person (a recipient). MiMedx has supplied over 1.9 million allografts, through both direct sales and consignment shipments. Our biomaterial platform technologies include AmnioFix®, EpiFix®, EpiCord®, AmnioCord® and AmnioFill®. AmnioFix and EpiFix are our tissue allografts derived from the amnion and chorion layers of the human placental membrane. EpiCord and AmnioCord are tissue allografts derived from umbilical cord tissue. AmnioFill is a placental connective tissue matrix derived from the placental disc and other placental tissue.
Our EpiFix and EpiCord product lines are promoted for external use, such as in advanced wound care applications, while our AmnioFix, AmnioCord and AmnioFill products are positioned for use in surgical applications, including lower extremity repair, plastic surgery, vascular surgery and multiple orthopedic repairs and reconstructions.
MiMedx has two primary distribution channels: (1) direct to customers (healthcare professionals and/or facilities); and (2) sales through distributors.
Trends in Our Business
Demographic shifts are creating opportunities in the wound care space
The advanced wound care category is expected to continue growing due to certain demographic trends, including an aging population, increasing incidence of obesity and diabetes, and the associated higher susceptibility to non-healing chronic wounds. Furthermore, the increasing number of patients requiring advanced treatment represents a significant cost burden on the healthcare system. We expect that these shifts will benefit our business.
As we look for ways to achieve long-term competitive advantages, we plan to continue to invest in research & development
We continue to evaluate these opportunities in alignment with our focus on advanced wound care. We remain focused on advancing our BLA programs and are therefore aligning customer input, industry expertise, and additional resourcing toward seeking FDA approval for micronized dehydrated human amnion/chorion membrane (“dHACM”) for the potential indication to treat musculoskeletal degeneration across multiple indications. In addition, we expect to incur additional costs to achieve compliance with evolving regulatory standards.
Certain areas of our business suffered as a result of the issues identified in the Audit Committee Investigation
The Investigation has caused us to incur significant legal fees, fines, and penalties. Additionally, the Company has incurred significant costs in connection with the associated Restatement. Negative publicity in the marketplace has created challenges for the Company in selling product to customers and retaining talented employees. All of these matters have caused the Company to incur significant costs and have negatively impacted our financial performance. We have incurred additional related costs in 2020 and expect to continue incurring such costs throughout 2020, including the possibility of settlement costs for existing contingencies.
Expected Impact of COVID-19 Pandemic
On March 11, 2020, the World Health Organization designated the outbreak of a novel strain of coronavirus (“COVID-19”) as a global pandemic. Governments and businesses around the world have taken unprecedented actions to mitigate the spread of COVID-19, including imposing restrictions on movement and travel such as quarantines and shelter-in-place requirements, and restricting or prohibiting outright some or all commercial and business activity, including the manufacture and distribution of certain goods and the provision of nonessential services. As of the end of the first half of 2020, significant uncertainty exists surrounding the efficacy of these measures to mitigate the spread of the virus, in addition to uncertainty surrounding timing and availability of a vaccine. The evolution of the outbreak, combined with these uncertainties, could result in the imposition of similar or greater restrictions for indefinite periods of time.
56
COVID-19 did not affect our financial condition and results of operations for the year ended December 31, 2019. It began affecting us late in the first quarter of 2020.
Sourcing and Manufacturing
We source the raw materials for our product from donors in hospitals. We have a large, geographically-diverse network of donor hospitals. We experienced interruptions to our access to some hospitals in some geographic areas beginning in the second half of March, 2020. However, we were successful in mitigating this disruption to our supply by adding additional donor hospitals, using third-party providers of donated placentas, and by increasing efforts at hospitals that did not impose access limits. Additionally, in anticipation of expected disruptions, we ran manufacturing at levels greater than demand and have been successful in building our inventory of safety stock.
We process donated tissue in a sterile environment. However, the manufacturing space is a confined area where an affected employee might spread the virus to other employees despite the use of personal protective equipment required for this environment. We monitor our employees’ temperatures prior to entering our facilities as of June 30, 2020 only three manufacturing employees and two sales representatives have tested positive for the virus, each of whom was isolated from our workforce. Additionally, we required our non-manufacturing employees including our executives to work from home from March 13, 2020 until June 1, 2020, and we have continued to allow most employees flexibility in their work arrangements as a result of the pandemic. To date, and due to significant mitigation efforts, COVID-19 has had only a modest impact on our ability to source and manufacture our products.
Sales and Marketing
Our ability to sell our product has been hampered by the pandemic. Our sales force is spread across the country. In many areas, our sales force was excluded from hospitals and the offices of other health care providers. Additionally, many patients stayed away from hospitals and other medical facilities. This had an adverse effect on our revenues beginning late in the first quarter of 2020 and continuing into April. However, by mid-May, access to hospitals and healthcare providers by our sales force had been mostly restored, and we began to see significant numbers of patients return to hospitals and other healthcare providers, including for elective procedures. However, as of early July 2020, additional restrictions have been put in place in some areas of the country that again limit or postpone elective surgical procedures, and in particular, in areas of the country that contribute a larger portion of our sales. Future sales will depend on patients’ willingness and ability to visit healthcare providers for care, and our sales force’ access to healthcare providers. Also, the severity of the COVID-19 pandemic has been uneven across the country, and a second-wave outbreak of COVID-19 may have a greater impact on us than did the first wave depending on where infection rates are highest. We are not able to estimate COVID-19’s future effect on patient behavior and consequently future demand for our products. See Item 1A. - Risk Factors - The COVID-19 pandemic and governmental and societal responses thereto have adversely affected our business, results of operations and financial condition, and the continuation of COVID-19 or the outbreak of other health epidemics could harm our business, results of operations, and financial condition.
Selling and General Administrative Expenses
In response to these challenges, our management team initiated several actions. Most discretionary expenses such as travel were cancelled. We negotiated additional discounts with vendors. Merit salary increases scheduled for the second quarter of 2020 were deferred until the fourth quarter of 2020. Beginning on April 5, 2020, we reduced employees’ salaries, including those of senior executives, on a sliding scale with larger reductions applied to larger salaries; we intend for these reductions to last up to six months, and estimate that this initiative will save us approximately $18.6 million. This has allowed us to reduce our expense base and reduce cash outlays, although we expect our margins to be temporarily reduced until sales return to normal levels. Nevertheless, at the end of the first quarter of 2020 and continuing into April, we saw a reduction in the amount of cash generated by the business. At May 31, 2020, our cash balance, net of minimum balance covenants set forth in our BT Loan Agreement, was $27.4 million.
Liquidity and Capital Resources
On April 22, 2020, we executed the First Amendment (the “Amendment”) to our BT Loan Agreement with Blue Torch. The amendment provided for an increase in the maximum Total Leverage Ratio (as defined in the BT Loan Agreement), which is a quarterly test, for the remainder of 2020, and also provided for a reduction in the minimum Liquidity (as defined in the BT Loan Agreement) requirement from April 2020 through and including November 2020. Specifically, the maximum Total Leverage Ratio increased from 3 to 1 to 5 to 1 through December 31, 2020. The minimum Liquidity requirement was reduced from $40.0 million to $20.0 million for April and May 2020, and from $30.0 million to $20.0 million for June through November 2020. In connection with the Amendment, we agreed to pay a one-time fee (the “Amendment Fee”) of approximately $0.7 million, added
57
to the principal balance, and a one percentage point increase in the interest rate to LIBOR plus 9%. See “Recent Events” section below for discussion on the financing transactions.
In addition, the Amendment loosened restrictions on our ability to borrow additional funds; enabling us to borrow up to $10 million under the Paycheck Protection Program (the “PPP Loan”) offered by the U.S. Small Business Administration under the Coronavirus Aid, Relief, and Economic Security Act. We applied for the PPP Loan prior to obtaining the aforementioned amendment to the BT Loan Agreement, and received the proceeds of the PPP Loan on April 24, 2020.
On May 8, 2020, we received a letter from the U.S. House of Representatives’ Committee on Oversight and Reform’s Select Subcommittee on the Coronavirus Crisis requesting that we return the proceeds of the PPP Loan so that the funds earmarked under the program could be used by smaller companies with more limited access to the capital markets. We repaid the PPP Loan in full on May 11, 2020.
Reserves and Financial Estimates
We do not expect that there be significant changes in judgments in determining the fair value of other assets measured in accordance with U.S. GAAP. As a result of the pandemic, we do not expect to incur any material impairments (e.g., with respect to goodwill, intangible assets, long-lived assets, right of use assets, investment securities), increases in allowances for credit losses, restructuring charges, other expenses, or changes in accounting judgments that have had or are reasonably likely to have a material impact on your financial statements, although we expect our days sales outstanding, post revenue recognition transition discussed in the “Critical Accounting Policies” below, to increase modestly as a result of patient behavior.
The uncertain future impacts of COVID-19 make it difficult for us to forecast future results. This is not helpful when seeking capital, but we are not otherwise able to quantify the effects of COVID-19 on our ability to obtain additional capital.
Financial Reporting Systems and Internal Controls
We have invested in technology to allow our office staff to work remotely. As a result, we do not expect the pandemic to have a material adverse effect on our financial reporting systems, internal controls over financial reporting and disclosure controls and procedures, although we have experienced delays when working with third parties who do not have remote access to our systems or whose procedures require them to review certain physical records.
Recent Events
FDA Guidance and Enforcement Discretion
In November 2017, the FDA published a series of related guidances, including one entitled “Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products: Minimal Manipulation and Homologous Use–Guidance for Industry and Food and Drug Administration Staff” that established an updated framework for the FDA’s regulation of cellular and tissue-based products. Among other things, the guidances clarified the FDA’s views about the criteria that differentiate Section 361 HCT/Ps from Section 351 HCT/Ps. As described elsewhere in this Form 10-K, the guidances clarified the FDA’s expectation that certain products, such as micronized products that MiMedx has long marketed as Section 361 HCT/Ps, will be treated as Section 351 HCT/Ps moving forward. The guidances also confirmed that amniotic membrane in sheet form generally can be characterized as “minimally manipulated” and therefore regulated solely under Section 361.
The guidances stated that the FDA intends to exercise enforcement discretion under limited conditions with respect to the IND application and pre-market approval requirements for certain HCT/Ps through November 2020. This means that, through November 2020, the FDA does not intend to enforce certain provisions as they currently apply to certain entities or activities but provided no assurances. The FDA intended this period of enforcement discretion to give sponsors time to evaluate their products, have a dialogue with the agency and, if necessary, begin clinical trials and file the appropriate pre-market applications to transition products that had been marketed as Section 361 HCT/Ps into compliance with Section 351. The FDA’s approach is risk-based, and the guidances clarified that high-risk products and uses might be subject to immediate enforcement action. We have continued to market our micronized products under this policy of enforcement discretion while at the same time pursuing BLAs for certain of our micronized products. For more information, refer to Item 1, “Business–Overview” and “Government Regulation,” and Item 1A, “Risk Factors,” under the heading “To the extent our products do not qualify for regulation as human cells, tissues and cellular and tissue-based products under Section 361 of the Public Health Service Act, this could result in removal of the applicable products from the market, would make the introduction of new tissue products more expensive and significantly delay the expansion of our tissue product offerings and subject us to additional post-market regulatory requirements.”
58
BLA Development Effort
As part of our BLA development effort, we have also made efforts to transition our manufacturing establishments into compliance with cGMP. During the enforcement discretion period, the FDA is permitting products that will become Section 351 HCT/Ps to be manufactured in compliance with GTP regulation. However, after the end of the enforcement discretion period, these products will be subject to cGMP compliance. The transition from GTP to cGMP compliance includes development and enhancement of production processes, procedures, test and assays, and it requires extensive validation work. It can also involve the procurement and installation of new production or lab equipment. These efforts require human capital, expertise and resources. The Company is developing and enhancing systems to meet these requirements, and expects to complete those efforts by November 2020, though there is no guarantee that the Company will be able to meet the requirements on that timeline or at all. For more information on our clinical trials, BLA development effort, and FDA enforcement discretion, see the section entitled “Business–Government Regulation.”
Coronavirus Aid, Relief and Economic Security (CARES) Act
On March 27, 2020, the “Coronavirus Aid, Relief and Economic Security (CARES) Act” (the “CARES Act”) was signed into law. The CARES Act includes provisions relating to refundable payroll tax credits, deferment of the employer portion of certain payroll taxes, loans and grants to certain business, net operating loss carryback periods, alternative minimum tax credit refunds, modifications to the net interest deduction limitations and technical corrections to tax depreciation methods for qualified improvement property. We applied for and received a $10.0 million loan under the PPP loan. On May 11, 2020 we completed the repayment of the PPP Loan.
In addition, modifications to the tax rules for carryback of net operating losses are expected to result in an estimated federal tax refund of $11.3 million and a resulting income tax benefit.
Financing Transactions
On July 2, 2020, the Company issued $100 million of Series B Preferred Stock to an affiliate of EW Healthcare Partners and to certain funds managed by Hayfin Capital Management LLP pursuant to the Securities Purchase Agreement for an aggregate purchase price of $100,000,000. Also on July 2, 2020, the Company borrowed an aggregate of $50 million and obtained an additional committed but undrawn $25 million facility pursuant to the Hayfin Loan Agreement. See Item 9B, “Other Information.” A portion of the proceeds of the Preferred Stock Transaction and the Hayfin Loan Transaction was used to repay the outstanding principal balance of $72.0 million, accrued interest and fees of $0.1 million, and prepayment penalty of $1.4 million under the BT Loan Agreement. The Company also terminated the BT Loan Agreement on July 2, 2020. For further information regarding the Preferred Stock Transaction, the Hayfin Loan Transaction and the termination of the BT Loan Agreement, see Item 9B, “Other Information.”
Critical Accounting Policies
We believe that of our significant accounting policies, which are described in Note 3 “Significant Accounting Policies” to our consolidated financial statements appearing elsewhere in this report, the following accounting policies involve a greater degree of judgment and complexity.
Revenue Recognition
We sell our products primarily to individual customers and independent distributors (collectively referred to as “customers”). In 2017, 2018, and into part of 2019 our control environment was such that it created uncertainty surrounding all of our customer arrangements which required consideration related to the proper revenue recognition under the applicable literature. The control environment allowed for the existence of extra-contractual or undocumented terms or arrangements initiated by or agreed to by us and former members of Company management at the outset of the transactions (side agreements). Concessions were also agreed to subsequent to the initial sale (e.g. sales above established customer credit limits extended and unusually long payment terms, return or exchange rights, and contingent payment obligations) that called into question the ability to recognize revenue at the time that product was shipped to a customer. The applicable revenue recognition guidance also changed beginning January 1, 2018 which further impacted our revenue recognition methodology.
As a result, our application of the applicable revenue recognition guidance varies for of the years ended December 31, 2019, 2018 and 2017. Additionally, we changed our pattern of revenue recognition effective October 1, 2019. The application of the relevant revenue recognition guidance and the revenue recognition policy are further discussed below for each period presented.
Fiscal Year Ended December 31, 2017
59
For the year ended December 31, 2017, we applied the revenue recognition guidance in ASC Topic 605, Revenue Recognition (“ASC 605”). Under ASC 605, revenue should not be recognized until it is realized or realizable and earned. SEC Staff Accounting Bulletin (“SAB”) Topic 13.A.1 (as codified in ASC 605-10-S99-1) outlines four criteria that generally indicate when revenue is realized or realizable and earned. If any of these criteria are not met, revenue recognition should be deferred until all criteria have been met.
Based on our evaluation, we determined that the revenue recognition criteria stipulated under ASC 605 were met only when both of the following events had occurred: (1) we fulfilled the customer's purchase order by delivering product ordered, and (2) we collected payment for the product delivered. Furthermore, we determined that the amount of revenue to be recognized should be limited to the amount of payment received in a given period less the amount expected to be refunded or credited to customers for sales returns made after payment.
An exception to the above revenue recognition under ASC 605 during the year ended December 31, 2017 related to the sales generated by our wholly-owned subsidiary, Stability. For sales of our products through Stability, we recognized revenue under ASC 605 only when both of the following events had occurred: (1) we fulfilled the customer’s purchase order by delivering all product ordered; and (2) the product has been delivered to the customer. Total sales from Stability were $7.0 million for the year ended December 31, 2017. We divested Stability on September 30, 2017.
Fiscal Year Ended December 31, 2018
We adopted ASC Topic 606, Revenue from Contracts with Customers (“ASC 606”), on January 1, 2018 by using the modified retrospective method. ASC 606 establishes principles for reporting information about the nature, amount, timing and uncertainty of revenue and cash flows arising from the entity's contracts to provide goods or services to customers. The core principle requires an entity to recognize revenue to depict the transfer of goods or services to customers in an amount that reflects the consideration that it expects to be entitled to receive in exchange for those goods or services recognized as performance obligations under the relevant criteria. We assessed the impact of the ASC 606 guidance by reviewing customer contracts and accounting policies and practices to identify differences, including identification of the contract and the evaluation of our performance obligations, transaction price, customer payments, transfer of control and principal versus agent considerations.
ASC 606 establishes a five-step model for revenue recognition. The first of these steps requires the identification of the contract as described in ASC 606-10-25-1. The specific criteria (the “Step 1 Criteria”) to this determination are as follows:
• | The parties to the contract have approved the contract (in writing, orally, or in accordance with other customary business practices) and are committed to perform their respective obligations; |
• | The entity can identify each party’s rights regarding the goods or services to be transferred; and |
• | The entity can identify the payment terms for the goods or services to be transferred. |
• | The contract has commercial substance. |
• | It is probable that the entity will collect substantially all of the consideration to which it will be entitled in exchange for the goods or services that will be transferred to the customer. |
We concluded that the first three of the above criteria were not met upon shipment of product to the customer, the fourth criteria had been met and we acknowledge that there is a degree of uncertainty as to whether last criteria above had been met. Although the parties to the contract may have approved the contract and purchase orders in writing, we concluded that upon shipment of products to the customer there is not sufficient evidence that our customers were committed to perform their obligations defined in the contract due to the existence of extra-contractual or undocumented terms or arrangements (e.g., regarding payment terms, right of return, etc.). We could not reliably identify each party’s rights regarding the products to be transferred upon shipment of those products to customers.
We determined the transaction price of our contracts to equal the amount of consideration received from customers less the amount expected to be refunded or credited to customers, which is recognized as a refund liability that is updated at the end of each reporting period for changes in circumstances. The refund liability is included within accrued expenses in our consolidated balance sheet.
60
Fiscal Year Ended December 31, 2019
We continued to assess new and existing contracts throughout 2019 to determine if the Step 1 Criteria noted above for the determination of a contract under ASC 606 were met for new contracts at the outset of a sales transaction (i.e., upon shipment of product) or for existing contracts at some point within 2019 when all the terms of the arrangement would have been known. Until it was determined if the Step 1 Criteria had been met, revenue recognition continued to be deferred consistent with the assessment for the year ended December 31, 2018.
As further discussed above, the primary factors contributing to the determination in prior periods that the Step 1 Criteria had not been met were the inappropriate tone at the top and the existence of pervasive extra-contractual or undocumented terms or arrangements. These prior business practices and the lack of transparency surrounding them created a systemically implied right for customers to demand future and unknown performance by us. Although some of the former executives were employed by us only through June 2018, we determined that based on the impact of the prior tone at the top, the continued internal sales force strategy and the existing customer base’s continued expectations (based on past practice), there would be flexibility with respect to arrangement terms even after delivery of the product so pervasive that all customer arrangements continued to be subject to uncertain modification of terms into 2019.
After identifying the primary factors contributing to the lack of knowledge regarding our customer contractual terms, we began implementing changes in mid-2018 to remediate the pervasive weaknesses in the control environment, followed by gradually implementing measures to empower our compliance, legal, and accounting departments; educate our sales force on appropriate business practices; and communicate our revised terms of sale to customers. We assessed our efforts throughout 2019 to determine when, if at any point, the factors contributing to the inability to satisfy the Step 1 Criteria were sufficiently addressed such that the Step 1 Criteria were met at the time of physical delivery to the customer. Determining when these conditions were effectively satisfied was a matter of judgment; however, we determined that adequate knowledge of the contractual arrangements with our customers did exist in 2019 for new and certain existing arrangements. We did note that there is no single determinative change that overcame the pervasive challenges noted above, but rather an accumulation of efforts that taken together, resulted in sufficient knowledge of contractual relationships both internally and externally with our customers.
To address the tone at the top issues, we noted that proper remediation involved not only the removal of members of management who were setting an inappropriate tone but also the establishment of new management throughout the organization that emphasized a commitment to integrity, ethical values and transparency and have that reinforcement for a sustained period of time. The changes made to management positions throughout the organization and the resulting organization behavior changes were assessed to have been sufficiently addressed by the end of the second quarter of 2019.
To determine when we had either eliminated or had sufficient knowledge to identify any extra-contractual arrangements, we noted that a key factor contributing to our historical lack of visibility into the arrangements with our customers was the failure to adhere to credit limits, payment terms and return policies. The establishment of additional controls and the emphasis on adherence to our existing policies and controls was an iterative process that continued through the first two quarters of 2019. Additional factors contributing to the increased visibility into our contractual arrangements involved further education and training of the sales personnel regarding our terms and conditions as well as monitoring of the sales personnel and customers for compliance with the contractual arrangements. We implemented a disciplined approach to educating the sales personnel regarding the prior practices that were considered unacceptable, ensuring they were knowledgeable regarding current terms and conditions and implementing an open dialogue with the credit and collections department. Monitoring of the customer base was accomplished through a variety of measures including, but not limited to, analysis of payments made within the original terms, levels of returns post-shipment, and various continued communication with the customer account representatives by members of our credit and collections department. During the third quarter of 2019, management determined that these efforts with the sales personnel and the external customers had been in place for a sufficient period of time to provide us an understanding of its contractual arrangements with customers.
Therefore, beginning October 1, 2019 for all new customer arrangements, we determined adequate measures were in place to understand the terms of our contracts with customers such that the Step 1 Criteria would be met prior to shipment of product to the customer or implantation (or surgical insertion) of the products on consignment.
We also reassessed whether the Step 1 Criteria had been met for all shipments of product where payment had not been received as of September 30, 2019. While the measures summarized above provided significant evidence necessary to understand the terms of our contractual arrangements with our customers, certain of these customers continued to exhibit behaviors that resulted in extended periods until cash collection. Such delays in collection suggested that uncertainty regarding extra-contractual arrangements may continue, particularly as it relates to payment terms. As a result, we concluded the following for any existing arrangements, which remained unpaid at September 30, 2019.
61
• | For customer arrangements where collection was considered probable within 90 days from the date of the original shipment or implantation of the products, we concluded the Step 1 Criteria were met (the “Transition Adjustment”). |
• | For the remaining customer arrangements (the “Remaining Contracts”), we concluded that, due to the uncertainty that extracontractual arrangements may continue, the Step 1 Criteria would not be satisfied until we receive payment from the customer. At that point, we determined that an accounting contract would exist and our performance obligations to deliver product to the customer to pay for the product would be satisfied. As of December 31, 2019, upon reassessment, we concluded that the Step 1 Criteria continued to not be met due to the same circumstances described above for these contracts. |
We continued to record the deferred cost of sales on the arrangements that failed the Step 1 Criteria where collectibility was reasonably assured and will recognize the costs when the related revenue is recognized. We also continued to offset deferred revenue with the associated accounts receivable obligations for these arrangements that continued to fail the Step 1 Criteria.
For all customer transactions concluded to meet the Step 1 Criteria, we then assessed the remaining criteria of ASC 606 to determine the proper timing of revenue recognition.
Under ASC 606, we recognize revenue following the five-step model: (i) identify the contract with a customer (the Step 1 Criteria); (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the entity satisfies a performance obligation. As noted above, beginning October 1, 2019, we determined that we had met the Step 1 Criteria for new and certain existing arrangements. We also determined that the performance obligation was met upon delivery of the product to the customer, or at the time the product is implanted for products on consignment, at which point we determined we will collect the consideration we are entitled to in exchange for the product transferred to the customer. As a result, we recognize as revenue the amount of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied, generally upon shipment of the product to the customer. The nature of our contracts gives rise to certain types of variable consideration, including rebates and other discounts. We include estimated amounts of variable consideration in the transaction price to the extent that it is probable there will not be a significant reversal of revenue. Estimates are based on historical or anticipated performance and represent our best judgment at the time of sale. We have consignment agreements with several customers and distributors which allow us to better market our products by moving them closer to the end user. We determined that we have fulfilled our performance obligation once control of the product has been delivered to the customer, which occurs simultaneously with the product being implanted.
We act as the principal in all of our customer arrangements and therefore record revenue on a gross basis. Shipping is considered immaterial in the context of the overall customer arrangement, and damages or loss of goods in transit are rare. Therefore, shipping is not deemed a separately recognized performance obligation and we have elected to treat shipping costs as activities to fulfill the promise to transfer the product. We maintain a returns policy that allows our customers to return product that is consigned, damaged or non-conforming, ordered in error, or due to a recall. The estimate of the provision for returns is based upon historical experience with actual returns. Our payment terms for customers are typically 30 to 60 days from receipt of title of the goods.
62
Based on the assessment noted above, we concluded that through the first two quarters of 2019, the pattern of revenue recognition under ASC 606 remained the same as the application for the year ended December 31, 2018; that is, revenue was deferred until the product was paid for or returned. In order to account for the determination that the Step 1 Criteria had been met during the third quarter of 2019, for existing customer arrangements, we recorded the following (in millions):
Amounts Invoiced and Not Collected | Deferred Cost of Sales | ||||||
Amounts as of September 30, 2019 | $ | 48,883 | $ | 6,415 | |||
Revenue recognized related to amounts invoiced and not collected at September 30, 2019: | |||||||
Transition Adjustment during the three months ended September 30, 2019 | (21,385 | ) | (2,565 | ) | |||
Cash collected during the three months ended December 31, 2019 related to the Remaining Contracts | (8,219 | ) | (1,151 | ) | |||
(29,604 | ) | (3,716 | ) | ||||
Write-off of customer contracts where collection is no longer reasonably assured (a) | (10,273 | ) | (1,438 | ) | |||
Amounts as of December 31, 2019 | $ | 9,006 | $ | 1,261 | |||
(a) The Company determined that for approximately $10.3 million of existing contracts where payment had not been received, collection was no longer reasonably assured. As a result, $1.4 million of deferred cost of sales relating to these customers was written off. Any future collections relating to these customer contracts will be recorded as revenue at the time payment is received. | |||||||
Goodwill and Impairment of Long-Lived Assets
Goodwill represents the excess of purchase price over the fair value of net assets of acquired businesses. Goodwill is tested for impairment annually on September 30, or whenever an event occurs or circumstances change that would indicate that the carrying amount may be impaired. When testing for goodwill impairment, we first assess qualitative factors to determine whether events or circumstances lead to a determination that it is more likely than not that the fair value of a reporting unit is less than its carrying amount. If we conclude it is more likely than not that the fair value of a reporting unit is less than its carrying amount, we perform a quantitative fair value test to determine any potential impairment loss. We may also choose to bypass the qualitative assessment and proceed directly to the quantitative analysis. As of the date of the filing of this Form 10-K, we have only one reporting unit.
Acquired indefinite lived intangible assets are tested for impairment annually or whenever events or changes in circumstances indicate that the carrying amount of an intangible asset may not be recoverable. Refer to Note 8 to the Consolidated Financial Statements for additional information. Our impairment reviews are based on an estimated future cash flow approach that requires significant judgment with respect to future revenue and expense growth estimates. We use estimates consistent with business plans and a market participant view of the assets being evaluated. Actual results may differ from the estimates used in these analyses.
There were no recorded impairment losses related to goodwill in 2019, 2018, or 2017. We recorded impairment losses of $1.3 million, $0.0 million, and $0.6 million, related to the abandonment of patents in process and write-off of certain customer relationships during 2019, 2018, and 2017, respectively.
Share-based Compensation
Our share-based compensation cost for equity awards granted to employees, members of the Board, and non-employee consultants is measured at the grant date based on the fair value of the award, is adjusted by the estimated forfeitures and is recognized as an expense over the requisite service or vesting period in accordance with Financial Accounting Standards Board (“FASB”) ASC Topic 718 “Compensation–Stock Compensation,” and under the recently issued guidance following FASB’s pronouncement, ASU 2018-07, “Compensation-Stock Compensation (Topic 718): Improvements to Nonemployee Share-Based Payment Accounting”, which we adopted on January 1, 2019, the effective date of the new guidance.
Determining the appropriate fair value model and calculating the fair value of employee and non-employee stock option and restricted common stock awards requires estimates and judgments. Our share‐based compensation is a “critical accounting estimate” because changes in the assumptions used to develop estimates of fair value, the requisite service period, probability of achieving performance vesting conditions, or estimated forfeitures could materially affect key financial measures, including results of operations.
The fair value of restricted common stock is a value of common stock on a grant date. The fair value of stock option grants is estimated using the Black-Scholes option pricing model. Use of the valuation model requires management to make certain
63
assumptions with respect to selected model inputs. We use the simplified method for share-based compensation to estimate the expected term. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant for the estimated option expected term. We estimate volatility using a blend of our own historical stock price volatility as well as that of market comparable publicly traded peer companies, since historically, we did not have enough history to establish volatility based upon our own stock trading. We routinely review our calculation of volatility for potential changes in future volatility, our life cycle, our peer group, and other factors. In addition, an expected dividend yield of zero is used in the option valuation model because we do not pay cash dividends and do not expect to pay any cash dividends in the foreseeable future.
For awards with service conditions only, we recognize stock-based compensation expense on a straight-line basis over the requisite service or vesting period. For awards with service and performance-based vesting conditions, we recognize stock-based compensation expense using the graded vesting expense attribution method over the requisite service period beginning in the period in which the awards are deemed probable to vest. Vesting probability for an award with performance vesting conditions is assessed based upon our expectations to become compliant with applicable securities law regulatory requirements and reporting obligations, as well as other performance vesting conditions specified in the restricted share unit award agreements. We recognize the cumulative effect of changes in the probability outcomes in the period in which the changes occur.
We recognize the fixed dollar amount known on a grant date with respect to the restricted stock unit awards that will be settled by issuing shares on the vesting date, with the number of shares to be determined based on our stock price on the settlement date over the vesting period, with an offsetting liability. Once the number of shares has been fixed and the shares are issued, we reclassify the liability related to the restricted share unit awards to equity.
Income Taxes
Our income tax expense, deferred tax assets and liabilities, and liabilities for unrecognized tax benefits reflect management’s best assessment of estimated current and future taxes to be paid. We are subject to income taxes in the United States, including numerous state jurisdictions.
Deferred income taxes arise from temporary differences between the tax basis of assets and liabilities and their reported amounts in the financial statements, which will result in taxable or deductible amounts in the future. In evaluating our ability to recover our deferred tax assets within the jurisdiction from which they arise, we consider all available positive and negative evidence, including scheduled reversals of deferred tax liabilities, projected future taxable income, tax-planning strategies, and results of recent operations. In projecting future taxable income, we begin with historical results adjusted for the results of discontinued operations and incorporate assumptions about the amount of future state, federal, and foreign pretax operating income adjusted for items that do not have tax consequences. The assumptions about future taxable income require significant judgment and are consistent with the plans and estimates we are using to manage the underlying businesses. In evaluating the objective evidence that historical results provide, we consider three years of cumulative operating income (loss). We account for income taxes under the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements. Under this method, deferred tax assets and liabilities are determined on the basis of the differences between the financial statement and tax bases of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. The effect of a change in tax rates on deferred tax assets and liabilities is recognized in income in the period that includes the enactment date.
We recognize deferred tax assets to the extent that we believe these assets are more likely than not to be realized. If we determine that we would be able to realize our deferred tax assets in the future in excess of their net recorded amount, we would make an adjustment to the deferred tax asset valuation allowance, which would reduce the provision for income taxes.
The calculation of our tax liabilities involves dealing with uncertainties in the application of complex tax laws and regulations both for U.S. federal income tax purposes and across numerous state jurisdictions. ASC Topic 740 (“ASC 740”) states that a tax benefit from an uncertain tax position may be recognized when it is more likely than not that the position will be sustained upon examination, including resolutions of any related appeals or litigation processes, on the basis of the technical merits. We (1) record unrecognized tax benefits as liabilities in accordance with ASC 740, and (2) adjust these liabilities when our judgment changes as a result of the evaluation of new information not previously available. Because of the complexity of some of these uncertainties, the ultimate resolution may result in a payment that is materially different from our current estimate of the unrecognized tax benefit liabilities. These differences will be reflected as increases or decreases to income tax expense in the period in which new information is available.
We record uncertain tax positions in accordance with ASC 740 on the basis of a two-step process whereby (1) we determine whether it is more likely than not that the tax positions will be sustained on the basis of the technical merits of the position, and (2) for those tax positions that meet the more-likely-than-not recognition threshold, we recognize the largest amount of tax benefit that is more than 50 percent likely to be realized upon ultimate settlement with the related tax authority.
64
We recognize interest and penalties related to unrecognized tax benefits within the income tax expense line in the accompanying Consolidated Statement of Operations. Accrued interest and penalties, if any, are included within the related tax liability line in the consolidated balance sheet.
As of December 31, 2019, 2018, and 2017, we had a valuation allowance recorded of $30.6 million, $27.3 million, and $0.6 million, respectively, against our net deferred tax assets. The decrease in the valuation allowance during 2017 is primarily related to the weight of available evidence which resulted in a determination to release our valuation allowance and recognize an income tax benefit as of September 30, 2017. The increase in valuation allowance during 2018 is primarily related to the weight of available evidence which resulted in the determination to increase our valuation allowance and recognize income tax expense as of December 31, 2018.
To the extent we determine that, based on the weight of available evidence, all or a portion of our valuation allowance is no longer necessary, we will recognize an income tax benefit in the period such determination is made for the reversal of the valuation allowance. If management determines that, based on the weight of available evidence, it is more-likely-than-not that all or a portion of the net deferred tax assets will not be realized, we may recognize income tax expense in the period such determination is made to increase the valuation allowance.
U.S. Tax Reform
On December 22, 2017, the United States enacted into law the Tax Cuts and Jobs Act (“Tax Act”). The Tax Act made broad and complex changes to the U.S. tax code, including a permanent corporate rate reduction to 21%. The Tax Act includes provisions that affected 2017, including: (1) requiring a remeasurement of all U.S. deferred tax assets and liabilities to the newly enacted corporate tax rate of 21%; (2) providing for additional first-year depreciation that allows full expensing of qualified property placed into service after September 27, 2017; (3) repealing the domestic production activities deduction and (4) modifying the deductibility of certain meals & entertainment expenses incurred.
In late December 2017, the SEC staff issued Staff Accounting Bulletin 118 (“SAB 118”), which provided guidance on accounting for the tax effects of the Tax Act. SAB 118 provides a measurement period that should not extend beyond one year from the Tax Act enactment date for companies to complete the related accounting under GAAP. The most significant impact of the Tax Act was a one-time reduction in net deferred income tax assets of approximately $12 million, due primarily to the re-measurement of deferred tax assets at the lower 21% U.S. federal corporate income tax rate. Our accounting for the tax implications of the Tax Cuts and Jobs Act was complete as of December 31, 2018.
Contingencies
We are subject to various patent challenges, product liability claims, government investigations and other legal proceedings in the ordinary course of business. Material legal proceedings are discussed in Note 16, “Commitments and Contingencies” in the Consolidated Financial Statements. Contingent accruals and legal settlements are recorded in the consolidated statements of operations as litigation-related and other contingencies when we determine that a loss is both probable and reasonably estimable. Legal fees and other expenses related to litigation are expensed as incurred and included in selling, general and administrative expenses in the consolidated statements of operations.
Due to the fact that legal proceedings and other contingencies are inherently unpredictable, our estimates of the probability and amount of any such liabilities involve significant judgment regarding future events. The factors we consider in developing our liabilities for legal proceedings include the merits and jurisdiction of the proceeding, the nature and the number of other similar current and past proceedings, the nature of the product and the current assessment of the science subject to the proceeding, if applicable, and the likelihood of the conditions of settlement being met.
In order to evaluate whether a claim is probable of loss, we may rely on certain information about the claim. Without access to and review of such information, we may not be in a position to determine whether a loss is probable. Further, the timing and extent to which we obtain any such information, and our evaluation thereof, is often impacted by items outside of our control including, without limitation, the normal cadence of the litigation process and the provision of claim information to us by opposing counsel. The amount of our liabilities for legal proceedings may change as we receive additional information and/or become aware of additional asserted or unasserted claims. Additionally, there is a possibility that we will suffer adverse decisions or verdicts of substantial amounts or that we will enter into additional monetary settlements, either of which could be in excess of amounts previously accrued for. Any changes to our liabilities for legal proceedings could have a material adverse effect on our business, financial condition, results of operations and cash flows.
As of December 31, 2019, our reserve for loss contingencies totaled $12.8 million related to the legal proceedings discussed in Note 16, “Commitments and Contingencies”. Although we believe there is a reasonable possibility that a loss in excess of the
65
amount recognized exists, we are unable to estimate the possible loss or range of loss in excess of the amount recognized at this time.
Recently Adopted Accounting Pronouncements
See Note 3, “Significant Accounting Policies,” in the Consolidated Financial Statements for recently adopted accounting pronouncements.
Components of and Key Factors Influencing Our Results of Operations
In assessing the performance of our business, we consider a variety of performance and financial measures. We believe the items discussed below provide insight into the factors that affect these key measures.
Revenue
The majority of our revenues are generated by wound care applications. We have two distribution channels: (1) direct to customers and (2) sales through distributors. Each distribution channel can be further disaggregated between sales to federal customers and non-federal customers.
Several factors affect our reported revenue in any period, including product, payer and geographic sales mix, operational effectiveness, pricing realization, marketing and promotional efforts, timing of orders and shipments, regulatory actions including healthcare reimbursement scenarios, competition, and business acquisitions that involve our customers or competitors.
Cost of goods sold and gross profit
Cost of goods sold includes product testing costs, quality assurance costs, personnel costs, manufacturing costs, raw materials and product costs, and facility costs associated with our manufacturing and warehouse facilities. Fluctuations in our cost of goods sold correspond with the fluctuations in sales units driven by the changes in our sales force and sales territories, product portfolio offerings and the number of facilities that offer our products.
Gross profit is calculated as net revenue less cost of goods sold. Our gross profit is affected by product and geographic sales mix, realized pricing of our products, the efficiency of our manufacturing operations and the costs of materials used to make our products. Regulatory actions, including with respect to reimbursement for our products, may require costly expenditures or result in pricing pressure, and may decrease our gross profit and gross profit margin.
Selling, general and administrative expenses
Selling, general and administrative expenses include personnel costs, commissions, incentive compensation, customer support, administrative and labor costs, insurance, professional fees, depreciation and bad debt expense. We expect our selling, general and administrative expenses to fluctuate based on revenue fluctuations, geographic changes and any changes to the size of our sales and marketing forces.
Research and development expenses
Research and development expenses relate to our investments in improvements to our manufacturing processes (including additional costs to transition our manufacturing establishments into compliance with cGMP for commercial production), product enhancements, and additional investments in our product pipeline and platforms. Our research and development costs also include expenses such as clinical trial and regulatory costs.
We expense research and development costs as incurred. Our research and development expenses fluctuate from period to period primarily based on the ongoing improvement to our manufacturing processes and product enhancements. We expect that these costs will increase in the near term as we continue to transition our manufacturing facilities into compliance with cGMP, advance our IND applications, and pursue BLAs for certain of our micronized products.
Important Cautionary Statement
We caution the reader that actual results may differ materially from our expectations, including those described under the subheadings “Expected Impact of COVID-19 Pandemic” above and “Results of Operations - Recent Developments and Outlook” below. Among the factors that could cause actual results to differ are: variances from our expectations or assumptions; changes in reimbursement policy from public and private insurers and health systems; the loss of a GPO or IDN; changes in purchasing behavior by government accounts; the loss of independent sales agents or distributors; the removal of any of our products from
66
the market as a result of regulatory actions; the success of our marketing efforts; the fact that obtaining and maintaining the necessary regulatory approvals for certain of our products will be expensive and time consuming and may impede our ability to fully exploit our technologies; rapid technological change could cause our products to become obsolete and, if we do not enhance our product offerings through our research and development efforts, we may be unable to compete effectively; our ability to transition our manufacturing facilities into compliance with cGMP, advance our IND applications, complete our clinical trials and pursue BLAs for certain of our micronized products; the fact that our business is subject to continuing regulatory compliance by the FDA and other authorities, which is costly, and our failure to comply could result in adverse effects on our business, results of operations and financial condition; the fact that litigation and other matters relating to and arising out of the Investigation, including the accounting review of our previously issued consolidated financial statements and the audits of fiscal years 2018, 2017 and 2016, have been time consuming and expensive, and may result in additional expense; and the fact that our variable rate indebtedness under the Hayfin Term Loan subjects us to interest rate risk, which could result in higher expense in the event of increases in interest rates and adversely affect our business, financial condition, and results of operations. See Item 1A, “Risk Factors,” for more information.
Recent Developments and Outlook
Revenue for the quarter ended March 31, 2020 of $61.7 million, a $4.8 million or 7.2% decrease over the quarter ended March 31, 2019 revenue of $66.6 million. The decrease primarily resulted from reduced sales of approximately $7.0 million in the Epi Wound product line and, to a lesser extent, due to the COVID-19 Pandemic in March 2020.
Gross profit margin in the first quarter of 2020 was 84% as compared to 89% in the first quarter of 2019. Gross profit decreased due to an increase in standard cost per unit caused by higher overhead/burden cost within production and quality assurance related to more stringent standards/regulations for cGMP compliance and BLA. This was partially offset by lower volume due to reduction in sales.
Investigation, restatement and related expenses were $15.6 million in the quarter ended March 31, 2020.
As of March 31, 2020, the Company had $53.5 million of cash and cash equivalents and $72.2 million of long-term debt.
We expect net sales during the quarter ended June 30, 2020 to decline between 23-27% from $67.4 million of net sales in the quarter ended June 30, 2019. We attribute most of this decline to the impact of COVID-19 as well as the discontinuation of OrthoFlo and AmnioFix Sports Medicine products. The pandemic caused elective procedures to be delayed and/or canceled, as well as increased access restrictions within patient settings making it difficult to retain and generate new business. Gross margin for the quarter ended June 30, 2020, is expected to be between 84-86%. As noted above, the cost-containment actions continued in the second quarter and we expect SG&A expenses in the quarter ended June 30, 2020, to have decreased 16-20%, compared to the quarter ended June 30, 2019. We expect Investigation, restatement and related expenses to be approximately $11.0 million in the quarter ended June 30, 2020.
Our expectations for 2020 results are based on our results to date. However, given the uncertainty regarding the impact on the economy from the COVID-19 virus, it is particularly difficult to reliably forecast 2020 financial metrics. See Item 1A. Risk Factors - “The COVID-19 pandemic and governmental and societal responses thereto have adversely affected our business, results of operations and financial condition, and the continuation of COVID-19 or the outbreak of other health epidemics could harm our business, results of operations, and financial condition.”
67
Results of Operations for 2019 Compared to 2018
Year Ended December 31, | ||||||||||||||
(in thousands) | ||||||||||||||
2019 | 2018 | $ Change | % Change | |||||||||||
Net Sales | $ | 299,255 | $ | 359,111 | $ | (59,856 | ) | (16.7 | )% | |||||
Gross profit | 256,174 | 322,725 | (66,551 | ) | (20.6 | )% | ||||||||
Selling, general and administrative | 198,205 | 258,528 | (60,323 | ) | (23.3 | )% | ||||||||
Investigation, restatement and related | 66,504 | 51,322 | 15,182 | 29.6 | % | |||||||||
Research and development | 11,140 | 15,765 | (4,625 | ) | (29.3 | )% | ||||||||
Amortization of intangible assets | 1,039 | 1,034 | 5 | 0.5 | % | |||||||||
Impairment of intangible assets | 446 | — | 446 | 100.0 | % | |||||||||
Interest (expense) income, net | (4,708 | ) | 527 | (5,235 | ) | (993.4 | )% | |||||||
Other income, net | 283 | — | 283 | 100.0 | % | |||||||||
Income tax provision benefit (expense) | 5 | (26,582 | ) | 26,587 | 100.0 | % | ||||||||
Net loss | $ | (25,580 | ) | $ | (29,979 | ) | $ | 4,399 | (14.7 | )% | ||||
Net Sales
We recorded revenue for the year ended December 31, 2019 of $299.3 million, a decrease of $59.9 million or 16.7% over 2018 revenue of $359.1 million. As discussed in the “Critical Accounting Policies” section above, the Company assessed its sales arrangements with customers during 2019 beginning with the definition of a contract under ASC 606 at the time of shipment of goods to the customer or upon the delivery of such goods if so stipulated by the terms of sale. Based on this assessment, the Company recognized revenue from a revenue benefit in the third quarter of 2019 related to the Transition and collections from the Remaining Customers during the fourth quarter of 2019 totaling approximately $29.6 million. Excluding this benefit related to the method in which the Company recognizes revenue, the decrease primarily resulted from unfavorable insurance coverage developments, which resulted in a decrease in the number of units sold. Additionally, approximately one-half of the reduction of the Company’s workforce announced in December 2018 and completed through 2019 were sales personnel that resulted in fewer visits to customers. Further, both the negative publicity resulting from the Audit Committee Investigation and discontinuing the OrthoFlo and AmnioFix Sports Medicine product lines adversely affected revenues.
Gross Profit Margin
Gross profit margin in 2019 was 85.6%, as compared to 89.9% in 2018. The gross profit margin decrease reflects fixed overhead costs being spread over lower production levels, increased costs of production related to the higher quality standards of cGMP, and higher scrap levels in the second half of the year. We implemented an electronic batch record system late in 2019, and are enhancing that system in 2020, and expect to reduce the incidence of scrap going forward.
Research and Development Expenses
Our research and development expenses decreased approximately $4.6 million, or 29.3%, to $11.1 million in 2019, compared to $15.8 million in the prior year. The decrease is primarily due to year-over-year decreases in clinical trial activities, the reductions in personnel due to the Company’s reduction in workforce as well as the decision to significantly reduce animal studies. We anticipate increasing our research and development spend in 2020 as we file additional INDs and work towards the BLAs.
Selling, General and Administrative Expenses
Selling, General and Administrative (“SG&A”) expense for 2019 decreased approximately $60.3 million, or 23.3%, to $198.2 million (or 66.2% of revenues), compared to $258.5 million (or 72.0% of revenues) for 2018.
Sales and Marketing expense included in SG&A decreased by $33.1 million, or 19.8%, to $134.2 million for 2019 compared to $167.3 million for 2018. The decrease was primarily due to the reduction in the workforce discussed below and lower commissions from the reduction in net sales discussed above.
General and administrative (“G&A”) expense included in SG&A decreased by $27.2 million, or 29.9%, to $64.0 million for 2019 compared to $91.2 million for 2018. The decrease was largely due to the completion of the Investigation in May 2019 and was
68
partially offset by costs in 2019 related to the two proxy contests in connection with the 2018 annual meeting of shareholders. The decrease in total G&A was also due to the reduction of our workforce announced in December 2018 by approximately 240 full-time employees, or 24% of our total workforce, of which about half were sales force personnel.
Share-based compensation included in SG&A for the years ended December 31, 2019 and 2018, was approximately $11.3 million and $13.5 million, respectively, a decrease of approximately $2.2 million, or 16.0%. The decrease was primarily due to the reduction in the workforce discussed above.
Investigation, Restatement and Related Expenses
Investigation, restatement, and related expenses increased by $15.2 million, or 29.6% to $66.5 million for 2019 compared to $51.3 million for 2018. The increase in 2019 as compared to 2018 primarily resulted from an increase in restatement, litigation, consulting fees and settlements of $21.7 million partially offset by a decrease in investigation fees of $6.5 million.
• | The Investigation was completed in 2019 and we do not expect to incur these costs going forward. |
• | Restatement costs are third-party service costs related to compiling, completing and auditing the financial statements included in the 2018 Form 10-K and in this filing, and thus we expect to incur these costs in the first half of 2020. |
• | Litigation fees increased by $11.6 million year over year from $14.6 million for 2018 compared to $26.2 million for 2019 due to the increase in settlement disputes and near-term contingencies related to the internal investigation; we expect to continue incurring these costs in the future as we address our contingent liabilities. |
• | Consulting costs in 2019 related to staff augmentation for restatement activities and advisory services related to financial reporting, internal controls, and the 2019 proxy contests. We continued to incur such costs in 2020 to assist with our effort to become current with our SEC financial reporting requirements. |
• | We may incur settlement costs in the future as we resolve contingent liabilities; see Note 16, “Commitments and Contingencies.” |
Through March 31, 2020, we have incurred $17.6 million of legal and other fees under indemnification agreements for current and former officers and directors. We expect to incur additional indemnification costs in 2020 as these cases continue.
Amortization of Intangible Assets
Amortization expense related to intangible assets remained relatively consistent for 2019 as compared 2018.
Impairment of Intangible Assets
Impairment of intangible assets of $0.4 million for 2019 related to the impairment of customer relationships that were part of the divestiture of Stability in 2017.
Interest (Expense) Income, net
Interest (expense) income, net increased by $5.2 million to $(4.7) million during the year ended December 31, 2019 from $0.5 million during the year ended December 31, 2018. This increase was due to the interest on our borrowings under the BT Loan Agreement entered into on June 10, 2019.
Other Income, Net
Other income, net of $0.3 million during the year ended December 31, 2019 reflects a settlement payment received for patent infringement case.
Income Taxes
The effective tax rate for 2019 was 0.0% on pre-tax book loss of $25.6 million, reflecting the lack of current tax expense due to our net loss position and the offset of deferred tax benefits by the corresponding adjustment to the valuation allowance. During 2019, a valuation allowance was recorded against current year losses resulting in effectively no tax expense or benefit. The effective tax rate in 2018 of (782.6)%, based on pre-tax book loss of $3.4 million, reflects the increase in the valuation allowance.
69
Results of Operations for 2018 Compared to 2017
Year Ended December 31, | ||||||||||||||
(in thousands) | ||||||||||||||
2018 | 2017 | $ Change | % Change | |||||||||||
Net Sales | $ | 359,111 | $ | 321,139 | $ | 37,972 | 11.8 | % | ||||||
Gross profit | 322,725 | 285,920 | 36,805 | 12.9 | % | |||||||||
Selling, general and administrative | 258,528 | 220,119 | 38,409 | 17.4 | % | |||||||||
Investigation, restatement and related | 51,322 | — | 51,322 | 100.0 | % | |||||||||
Research and development | 15,765 | 17,900 | (2,135 | ) | (11.9 | )% | ||||||||
Amortization of intangible assets | 1,034 | 1,678 | (644 | ) | (38.4 | )% | ||||||||
Loss on divestiture | — | (1,048 | ) | 1,048 | n/a | |||||||||
Interest income (expense), net | 527 | (87 | ) | 614 | (705.7 | )% | ||||||||
Income tax provision (expense) benefit | (26,582 | ) | 19,639 | (46,221 | ) | 235.4 | % | |||||||
Net (loss) income | $ | (29,979 | ) | $ | 64,727 | $ | (94,706 | ) | (146.3 | )% | ||||
Revenue
We recorded revenue for the year ended December 31, 2018 of $359.1 million, an increase of $38.0 million or 11.8% over 2017 revenue of $321.1 million. The increase primarily resulted from favorable insurance coverage developments, which resulted in an increase in the number of units sold. Additionally, we increased our direct sales force through the first three quarters of 2018 which resulted in the addition of new customers. Further, revenues benefited from sales made in prior periods and collected during 2018. These effects were partially offset by unfavorable insurance coverage developments, unfavorable publicity resulting from the Investigation, increased turnover of experienced sales personnel and related events in the fourth quarter of 2018.
Gross Profit Margin
Gross profit margin in 2018 was 89.9%, as compared to 89.0% in 2017. Gross profit margin increased due to the mix of products sold in 2018, including as a result of the divestiture of Stability on September 30, 2017 (whose products were relatively lower-margin). During 2018, we also saw an improvement in yield as a result of improved manufacturing efficiency in our wound care line.
Research and Development Expenses
Our research and development expenses decreased approximately $2.1 million, or 11.9%, to $15.8 million in 2018, compared to approximately $17.9 million in the prior year. The decrease is primarily due to year-over-year decreases in clinical trial activities as well as the decision to significantly reduce animal studies in 2018. In 2017, research and development expenses were driven by several multiple-site clinical trials related to our EpiCord and EpiFix products. These projects were completed during 2018.
Selling, General and Administrative Expenses
Selling, General and Administrative (“SG&A”) expense for 2018 increased approximately $38.4 million, or 17.4%, to $258.5 million (or 72.0% of revenues), compared to $220.1 million (or 68.5% of revenues) for 2017.
Sales and Marketing expense included in SG&A increased by $13.4 million, or 8.7%, to $167.3 million for 2019 compared to $153.9 million for 2017. The increase was primarily due to an increase in compensation related to the additional headcount of 72 employees from December 2017 through August 2018, as well as an increase in sales commissions based on shipments during the year.
General and administrative expense included in SG&A increased by $25.0 million, or 37.7%, to $91.2 million for 2019 compared to $66.2 million for 2017. Legal fees increased by $4.8 million, or 35.7%, to $18.4 million compared to $13.6 million for 2017. Consulting fees included in SG&A were $1.6 million for 2018 as compared with $0.3 million for 2017. The increase was primarily due to an increase in compensation related to the additional headcount of 34 employees from December 2017 through August 2018, prior to the reduction in force in December 2018, as well as an increase in accounting fees due to the change in audit firms in mid-2018.
70
In December 2018, we announced a reduction of our workforce by approximately 240 full-time employees, or 24% of our total workforce, of which about half were sales force personnel as part of previously announced plans to implement a broad-based organizational realignment, cost reduction and efficiency program to better ensure our cost structure is appropriate given our revenue expectations. As a result of the December 2018 broad-based organizational realignment, cost reduction and efficiency program, we incurred pre-tax charges of $6.1 million during the year ended December 31, 2018.
Investigation, restatement, and related expenses was $51.3 million in 2018 compared to $0 for 2017. The increase in legal, accounting, and other professional consulting fees incurred in 2018 as compared to 2017 primarily resulted from the Investigation, including legal fees, forensic audit fees, and consulting fees relating to the Restatement; and legal fees relating to the SEC Investigation, shareholder derivative lawsuits, and other litigation, as well as settlements made with former employees.
Share-based compensation included in SG&A for the years ended December 31, 2018 and 2017, was approximately $13.5 million and $20.1 million, respectively, a decrease of approximately $6.6 million, or 32.8%. The decrease was primarily due to our reduction in workforce in 2018, forfeitures of outstanding equity awards in connection with the reduction in workforce, and a reduction in the size of new equity-based awards to employees in 2018.
Amortization of Intangible Assets
Amortization expense related to intangible assets decreased approximately $0.7 million, or 38.4%, to $1.0 million for the year ended December 31, 2018, compared to $1.7 million in the prior year. Amortization decreased primarily due to the divestiture of Stability during the third quarter of 2017. We amortize our intangible assets over a period of 4 to 20 years, which we believe represents the remaining useful lives of the patents underlying the licensing rights and intellectual property. We do not amortize goodwill, but we test our goodwill at least annually for impairment and periodically evaluate other intangibles for impairment based on events or changes in circumstances as they occur.
Interest Income (Expense), net
Interest income (expense), net increased to $0.5 million during the year ended December 31, 2018 from $(0.1) million during the year ended December 31, 2017. This increase was due to a decision in late 2017 to begin charging interest on past due customer balances, partially offset by the amortization of deferred financing costs incurred during 2018 related to our $50 million revolving credit facility and commitments and undrawn fees connected to our line of credit. See Note 10, “Long-Term Debt,” in the Consolidated Financial Statements for further details.
Income Taxes
The effective tax rate for 2018 was (782.6)% on pre-tax book loss of $3.4 million. This compares to an effective tax rate of (43.6)% based on pre-tax book income of $45.1 million in 2017.
Our 2018 effective tax rate was driven largely by increases in our valuation allowance, causing $26.8 million of incremental income tax expense, an effective tax rate impact of (788.3)%. Other offsetting tax adjustments yielded an effective tax rate of 5.7%. The decrease in the valuation allowance during 2017 is primarily related to the weight of available evidence which resulted in a determination to release our valuation allowance and recognize an income tax benefit as of September 30, 2017.
In 2017, the Company derived significant tax benefits associated with the disposition of the Company’s interest in Stability (total benefit of approximately $5.3 million; effective tax rate impact of (8.9)% along with significant tax benefits associated with stock compensation-related deductions (total benefit of approximately $4.8 million; effective tax rate benefit of (9.9)%.
As a result of the Restatement, we re-evaluated the valuation allowance determinations made in prior years. Our analysis was updated to consider the changes to our historical operating results following the Investigation and subsequent review by management. In that process, we evaluated the weight of all evidence, including the ability or inability to project future income to utilize our deferred tax assets, and we concluded that as of December 31, 2015, our U.S. federal and state net deferred tax assets were no longer more-likely-than-not to be realized and that a valuation allowance was required. The decrease in the valuation allowance during 2017 is primarily related to the weight of available evidence which resulted in a determination to release the Company’s valuation allowance and recognize an income tax benefit as of September 30, 2017. The increase in valuation allowance during 2018 is primarily related to the weight of available evidence which resulted in the determination to increase the Company’s valuation allowance and recognize income tax expense as of December 31, 2018.
71
Contractual Obligations
Contractual obligations associated with ongoing business activities are expected to result in cash payments in future periods. The table below summarizes the amounts and estimated timing of these future cash payments as of December 31, 2019 (in thousands):
Less than | |||||||||||||||||||
Contractual Obligations | Total | 1 year | 1-3 years | 3-5 years | Thereafter | ||||||||||||||
BT Term Loan principal | $ | 73,125 | $ | 3,750 | $ | 69,375 | $ | — | $ | — | |||||||||
BT Term Loan interest | 17,965 | 7,515 | 10,450 | — | — | ||||||||||||||
Operating lease obligations | 4,713 | 1,463 | 3,250 | — | — | ||||||||||||||
Meeting space commitments | 1,988 | 684 | 1,304 | — | — | ||||||||||||||
Other | 370 | $ | 116 | $ | 254 | $ | — | $ | — | ||||||||||
$ | 98,161 | $ | 13,528 | $ | 84,633 | $ | — | $ | — | ||||||||||
Off-Balance Sheet Arrangements
We have no off-balance sheet arrangements.
Liquidity and Capital Resources
Our business requires capital for its operating activities, including costs associated with the sale of product through direct and indirect sales channels, the conduct of research and development activities, compliance costs, and legal and consulting fees in connection with ongoing litigation and other matters. We generally fund our operating capital requirements through our operating activities, cash reserves and, since June 2019, proceeds from term loans. We expect to use capital in the near and medium term to implement our priorities, including for capital investments, steps to complete achievement of cGMP compliance, advancement of our IND applications, pursuit of BLAs for certain of our micronized products, and settlements of certain legal matters.
As of December 31, 2019, the Company had approximately $69.1 million of cash and cash equivalents.
Our net working capital at December 31, 2019, increased $53.4 million to $55.9 million from $2.5 million at December 31, 2018. The increase in working capital was primarily due to the change in our revenue recognition methodology and the establishment of accounts receivable along with an increase in cash collections. Our current ratio (current assets divided by current liabilities) was 1.8 to 1 as of December 31, 2019 and 1.0 to 1 as of December 31, 2018.
We have funded our cash requirements, including for our operating activities and for the Investigation and Restatement, through existing cash reserves and from operating activities and the term loans described below under “Term Loans.” In addition, on July 2, 2020, we issued $100 million of Series B Preferred Stock to an affiliate of EW Healthcare Partners and to certain funds managed by Hayfin Capital Management LLP pursuant to the Securities Purchase Agreement for an aggregate purchase price of $100 million.
The Company is currently paying its obligations in the normal course of business. We believe that, due to the Preferred Stock Transaction and the Hayfin Loan Agreement (which provides for significantly less restrictive financial covenants than the BT Loan Agreement), our anticipated cash from operating activities, existing cash, and cash equivalents will enable us to meet our operational liquidity needs. See Note 19, “Restructuring,” in the Consolidated Financial Statements.
We expect to incur additional costs in connection with efforts to enhance our cGMP compliant manufacturing capabilities and toward the completion of the BLA process. This includes development and enhancement of production processes, procedures, tests and assays, and it requires extensive validation work. It can also involve the procurement and installation of new production or lab equipment. These efforts also require human capital, expertise and resources.
Additionally, as discussed in Note 16, “Commitments and Contingencies,” and further addressed in Note 21, “Subsequent Events,” of the Consolidated Financial Statements, we anticipate cash requirements related to the following items within one year from the date of the filing of this Form 10-K:
• | lawsuits or potential settlements for which we are not able to estimate a loss; |
72
• | private securities lawsuits, for which we are currently not able to estimate a loss and for which it is unclear whether we would be indemnified under various insurance policies; and |
• | investments and other expenditures required in order to bring the Company’s facilities into compliance with current Good Manufacturing Practices (“cGMPs”). |
Following the Preferred Stock Transaction, the Hayfin Loan Transaction, and the repayment and termination of the BT Loan Agreement, we analyzed our ability to address the aforementioned commitments and potential liabilities while remaining compliant with the financial covenants set forth in the Hayfin Loan Agreement discussed below for the 12 months extending from the date of the filing of this 2019 Form 10-K. Based on this analysis, and in combination with existing cash on hand, we believe it is probable that we will meet all obligations as they come due without violating the financial covenants set forth in the Hayfin Loan Agreement, as discussed below. Therefore, we concluded that there is no substantial doubt surrounding our ability to continue as a going concern.
Term Loans
BT Term Loan. On June 10, 2019, we entered into the BT Loan Agreement to borrow funds with a face value of $75.0 million (the “BT Term Loan”), the full amount of which was borrowed and funded. The proceeds of the BT Term Loan were used (i) for working capital and general corporate purposes and (ii) to pay transaction fees, costs and expenses incurred in connection with the BT Term Loan and the related transactions. The BT Term Loan was issued net of the original issue discount of $2.3 million. The Company also incurred $6.7 million of deferred financing costs.
The interest rate applicable to any borrowings under the BT Term Loan was equal to LIBOR plus a margin of 8.00% per annum. The BT Term Loan had an interest rate equal to 10.46% at the time the BT Loan Agreement was executed and an interest rate equal to 9.95% at March 31, 2020.
The BT Loan Agreement contained financial covenants requiring us to maintain the following:
• | Maximum total leverage ratio, defined as funded debt divided by consolidated adjusted EBITDA, of not more than 3.0 to 1.0 as of the last day of the four consecutive fiscal quarters. Our total leverage ratio was 1.7 times at December 31, 2019, a cushion of 1.3 times. |
• | Minimum liquidity of the Company, defined as unrestricted cash and cash equivalents, is not permitted, as of the last business day of each fiscal month following the BT Term Loan closing date through and including the fiscal month ending May 31, 2020, to be less than $40 million, and (b) beginning with the fiscal month ending June 30, 2020 and for each month ending thereafter, to be less than $30 million; provided that, beginning with fiscal month ending December 31, 2020, the total leverage ratio is less than 2.50 to 1.00 as of the last business day of any fiscal month, the liquidity of the Company shall not be less than $20 million. Our cash on hand was $69.1 million at December 31, 2019, exceeding the $40 million minimum liquidity by $29.1 million, and our cash on hand was $53.5 million as of March 31, 2020. |
Effective April 22, 2020, the BT Loan Agreement was amended to provide for an increase in the maximum Total Leverage Ratio, which was a quarterly test, from a Total Leverage Ratio of 3.00 to 1.00 to a new Total Leverage Ratio of 5.00 to 1.00 for the quarterly periods ending on June 30, 2020, September 30, 2020, and December 31, 2020, and also provided for a reduction in the minimum Liquidity covenant, which was a monthly requirement, from $40 million to $20 million for April and May 2020 and form $30 million to $20 million for June through November 2020. In connection with the amendment, we agreed to pay a one-time fee of approximately $0.7 million, added to the principal balance, and a 1 percentage point increase in the interest rate to LIBOR plus 9%.
The BT Loan Agreement provided that any prepayment of the loan subjected MiMedx to a prepayment penalty as of the date of the prepayment with respect to the BT Term Loan of:
• | During the period from June 10, 2019 through June 10, 2020, an amount equal to 3% of the principal amount of the BT Term Loan prepaid on such date; and |
• | During the period from June 11, 2020 through June 10, 2021, an amount equal to 2% of the principal amount of the BT Term Loan prepaid on such date. |
Principal prepayments after June 10, 2021 were not subject to a prepayment penalty.
73
Hayfin Term Loan; Preferred Stock Transaction; Termination of BT Loan Agreement. On July 2, 2020, the Company borrowed an aggregate of $50 million and obtained an additional committed but undrawn $25 million facility pursuant to the Hayfin Loan Agreement. A portion of the proceeds of the Preferred Stock Transaction and the Hayfin Loan Transaction was used to repay the outstanding principal balance of $72.0 million, accrued interest and fees of $0.1 million, and prepayment penalty of $1.4 million under the BT Loan Agreement. The Company also terminated the BT Loan Agreement on July 2, 2020. For further information regarding the Preferred Stock Transaction, the Hayfin Loan Transaction (including the terms of the Hayfin Loan Agreement) and the termination of the BT Loan Agreement, see Item 9B, “Other Information.”
Liquidity Considerations
Prior to the Preferred Stock Transaction and the Hayfin Loan Transaction, at June 26, 2020, the Company had approximately $48 million of cash and cash equivalents and approximately $73 million of long-term debt. Following the closing of the Preferred Stock Transaction and the Hayfin Loan Transaction, and the repayment of the BT Loan Agreement, as of July 2, 2020, the Company had approximately $110 million of cash and cash equivalents and approximately $50 million of long-term debt. See Note 21 “Subsequent Events” for additional discussion.
Since receiving the proceeds from our term loan borrowing in June 2019, our cash and cash equivalents were $96.9 million on June 30, 2019, $94.1 million on September 30, 2019, $69.1 million on December 31, 2019, and $53.5 million on March 31, 2020. The decline in cash and cash equivalents during this time is primarily the result of investigation, restatement and legal expenses we have incurred. While investigation activities are primarily completed and restatement activities are nearing completion, costs to remediate our internal controls and resolve outstanding legal matters are expected to continue.
Our net sales declined 14% in 2019 compared to 2018. In the first quarter of 2020, our net sales also declined 14% compared to the first quarter of 2019. Because of the COVID-19 pandemic, we anticipate a steeper reduction to our reported second quarter of 2020 net sales of 23% - 27% compared to the second quarter of 2019.
In addition, all of our revenues from micronized products, which accounted for $45.0 million, $68.4 million, and $42.4 million respectively, in 2017, 2018, and 2019, are at risk upon the expiration of the FDA’s enforcement discretion, which is scheduled to expire on November 20, 2020. See Item 1A - Risk Factors - “To the extent our products do not qualify for regulation as human cells, tissues and cellular and tissue-based products solely under Section 361 of the Public Health Service Act, this could result in removal of the applicable products from the market, would make the introduction of new tissue products more expensive and would significantly delay the expansion of our tissue product offerings and subject us to additional post-market regulatory requirements.”
Further, our liquidity is challenged by expected costs, required investments to bring our manufacturing facilities up to cGMP compliance, required investments in clinical trials to support BLAs, and contingent liabilities:
• | We expect to continue to incur costs to indemnify former officers and directors in legal proceedings against them. The Company has already borne substantial costs to satisfy these indemnification and expense advancement obligations and expects to continue to do so in the future. We may need to increase these expenditures if we experience further delays in trial dates or other proceedings and our ability to recovery any of these advanced costs in the future is uncertain. See Note 16, “Commitments and Contingencies.” |
• | We need to continue to invest in our manufacturing establishments to bring them into compliance with cGMP for production for our micronized products. We are also pursuing opportunities to partner with a contract manufacturing organization. The transition process includes development and enhancement of production processes, procedures, test and assays, and it requires extensive validation work. It can also involve the procurement and installation of new production or lab equipment. These efforts require human capital, expertise and resources. See Item 1A. – “Risk Factors” under the heading “To the extent our products do not qualify for regulation as human cells, tissues and cellular and tissue-based products solely under Section 361 of the Public Health Service Act, this could result in removal of the applicable products from the market, would make the introduction of new tissue products more expensive and would significantly delay the expansion of our tissue product offerings and subject us to additional post-market regulatory requirements.” |
• | The clinical program to support our BLAs will involve substantial cost. Products subject to the FDA’s BLA requirements must comply with a range of pre- and post-market provisions. Pre-market compliance includes the conduct of clinical trials in support of BLA approval, the development and submission of a BLA, and the production of product for use in the clinical trials that meets FDA’s quality expectations. See Item 1A - Risk Factors - “If any of the BLAs are approved, the Company would be subject to additional regulation which will increase costs and could result in adverse sanctions for non-compliance.” |
74
• | We may become obligated to make payment in respect of certain contingent obligations in addition to our indemnity obligations to former officers and directors. |
• | We are exposed to potential liabilities and reputational risk associated with litigation, regulatory proceedings, and government enforcement actions. See Item 3, “Legal Proceedings” and Note 16, “Commitments and Contingencies” and Item 1A. - Risk Factors - “We are currently, and may in the future be, subject to substantial litigation and ongoing investigations that could cause us to incur significant legal expenses and result in harm to our business.” |
Even after the amendment of the BT Loan Agreement in April 2020, we remained subject to financial convents in the BT Loan Agreement that would have become more restrictive after November 30, 2020. A breach of a financial covenant in the BT Loan Agreement, if uncured or unable to be cured, would likely have resulted in an event of default that would have triggered the lender’s remedies, including acceleration of the entire principal balance of the loan. The BT Loan Agreement covenants are summarized below:
Financial Covenant | Original BT Loan Agreement | Amended BT Loan Agreement | Amended BT Loan Agreement (after December 31, 2020)* |
Total Leverage Ratio (tested quarterly) | 3:1 | 5:1 | 3:1 |
minimum Liquidity Ratio (tested monthly) | $40 million; $30 million 4/2020 and 5/2020 | $20 million | $30 million* |
Applicable Margin (i.e. interest rate) | 3-month LIBOR + 8% | 3-month LIBOR + 9% | 3-month LIBOR + 9% |
*requirement would increase to $30 million effective November 30, 2020 (i.e. for the month ending December 31, 2020); requirement would decrease to $20 million so long as the Total Leverage Ratio is less than 2.5 to 1.
The relief provided by the April 2020 amendment generally would have expired at the end of 2020. Further, we would have remained at risk of breaching these covenants during 2020 if we had another setback in our business, for example, if we experienced a second wave of the COVID-19 pandemic. Also, if the Company’s net revenue decreased by more than 20% in 2020, the Company might have exceeded the applicable maximum leverage ratio permitted by the BT Loan Agreement during the second half of 2020.
A breach of a financial covenant in the BT Loan Agreement, if uncured or unable to be cured, would likely have resulted in an Event of Default that would have triggered the lender’s remedies, including acceleration of the entire principal balance of the BT Loan. We might not have been able to repay all such amounts or be able to find alternative financing in an event of a default. Even if alternative financing were available in an event of a default under the BT Loan Agreement, it might have been on unfavorable terms, and the interest rate charged on any new borrowings could have been substantially higher than the interest rate under the BT Loan Agreement, thus adversely affecting our cash flows, liquidity, and results of operations. Acceleration of the repayment of the loan pursuant to the terms of the BT Loan Agreement, in combination with the Company’s current commitments and contingent liabilities, also could have cast doubt on the Company’s ability to continue as a going concern.
Moreover, as noted above, our revenues for 2020 were down as compared to revenues for 2019. The COVID-19 pandemic makes it difficult to predict future revenues and there is no assurance that the COVID-19 pandemic will not continue to adversely affect revenue in 2020 or 2021. More specifically:
• | Our customers have experienced, and may continue to experience, restrictions in their access to hospitals and ability to access other healthcare providers, particularly for elective procedures. |
• | Our manufacturing operations, sales and demand for our products, and clinical trials may be adversely affected if our leadership, employees, sales agents, suppliers, medical professionals, or users of our products are impacted by illness or through actions taken to stop or slow the spread of the COVID-19 pandemic. |
• | Our results of operations may be adversely affected if we experience shortages of donated placentas because donors or our recovery specialists are excluded from hospitals, or because additional testing protocols are implemented for donated tissues based on guidance issued by the AATB, FDA, or other standards and are screened as ineligible. |
75
• | Because our sales are not evenly spread across the United States, to the extent that areas most impacted by COVID-19 are those where we have more of our sales, the pandemic will have a greater adverse impact on our revenues. |
• | The ultimate impact of the COVID-19 pandemic is highly uncertain and subject to change, and we do not yet know the full extent of delays or impacts on our business, or how long such effects will endure. |
As a result of these factors, there were doubts about our ability to continue as a going concern in the absence of the Preferred Stock Transaction and the Hayfin Loan Agreement.
Due to these factors and concern regarding the Company’s ability to continue to comply with the financial covenants contained in the BT Loan Agreement, in December 2019, we engaged a nationally recognized investment bank to act as sole placement agent for the purpose of managing any possible capital sourcing process.
After considering the Company’s uncertain ability to estimate cash from operations, the near and mid-term risks to the Company’s ability to generate cash, and anticipated future expenses and required investments, the Board, in consultation with management and the Company’s outside advisors, considered various financing alternatives. The Company ultimately held discussions with more than 20 potential financing sources. Throughout the process, the Board evaluated the risks associated with deferring any capital raising process, such as until after the filing of this Form 10-K and/or the relisting of our Common Stock. After careful consideration, the Board unanimously approved the Preferred Stock Transaction and the Hayfin Loan Transaction.
Prior to the Preferred Stock Transaction and the Hayfin Loan Transaction, at June 26, 2020, the Company had approximately $48 million of cash and cash equivalents and approximately $73 million of long-term debt. Following the closing of the Preferred Stock Transaction and the Hayfin Loan Transaction, and the repayment of the BT Loan Agreement, as of July 2, 2020, the Company had approximately $110 million of cash and cash equivalents and approximately $50 million of long-term debt.
Discussion of Cash Flows
During the year ended December 31, 2019, net cash (used in) provided by operations decreased approximately $75.2 million to $(39.4) million, compared to $35.8 million for the year ended December 31, 2018. This decrease was primarily attributable to the effect of the change in revenue recognition policy of $17.4 million, an increase in accounts receivable of $10.9 million, as well as the $20.1 million decrease in cash related to the change in other balance sheet accounts in 2019.
During the year ended December 31, 2018, net cash provided by operations decreased approximately $27.1 million to $35.8 million, compared to $62.9 million for the year ended December 31, 2017. This decrease was primarily attributable to decrease in net income (as a result of higher selling, general and administrative expenses primarily as a result of the Investigation and Restatement), partially offset by a $6.6 million increase in accounts payable due to working capital management efforts.
During the year ended December 31, 2019, net cash provided by (used for) investing activities increased approximately $9.7 million to $0.5 million provided by investing activities compared to $9.2 million of cash used in investing activities for the year ended December 31, 2018 due to the repayment of the note receivable from Stability, partially offset by a significant reduction in the equipment purchased during 2019.
During the year ended December 31, 2018, net cash used for investing activities increased approximately $3.8 million to $9.2 million compared to $5.4 million for the year ended December 31, 2017 due to the higher equipment purchases in 2018 as compared with 2017.
During the year ended December 31, 2019, net cash flows provided by (used for) financing activities was approximately $62.9 million compared to $(8.9) million during the year ended December 31, 2018. The increase was primarily due to the BT Term Loan borrowing of $75.0 million in June 2019 partially offset by the deferred financing costs on the BT Term Loan and shares repurchased for tax withholdings on restricted shares. During 2019, the Company repurchased 429,918 shares of Common Stock surrendered by employees to satisfy tax withholding obligations upon vesting of restricted stock. The Company did not otherwise repurchase any shares of our Common Stock during 2019.
During the year ended December 31, 2018, net cash flows used for financing activities was approximately $8.9 million compared to $60.4 million during the year ended December 31, 2017. The decrease was primarily due to lower share repurchases and lower proceeds from stock option exercises.
76
Non-GAAP Financial Measures
In addition to our GAAP results, we provide certain Non-GAAP metrics including Earnings Before Interest, Taxes, Depreciation and Amortization (“EBITDA”) and Adjusted EBITDA. We believe that the presentation of these measures provides important supplemental information to management and investors regarding our performance. These measurements are not a substitute for GAAP measurements. Company management uses these Non-GAAP measurements as aids in monitoring our on-going financial performance from quarter-to-quarter and year-to-year on a regular basis and for benchmarking against comparable companies.
EBITDA is intended to provide a measure of the Company’s operating performance as it eliminates the effects of financing and capital expenditures. EBITDA consists of GAAP net income (loss) excluding: (i) depreciation, (ii) amortization of intangibles, (iii) interest (income) expense and (iv) income tax provision.
Adjusted EBITDA is intended to provide an enduring, normalized view of EBITDA and our broader business operations that we expect to experience on an ongoing basis by removing items which may be irregular, one-time, or non-recurring from EBITDA; most significantly those expenses related to the Audit Committee Investigation and Restatement. This enables us to identify underlying trends in our business that could otherwise be masked by such items.
Adjusted EBITDA consists of GAAP net income (loss) excluding: (i) depreciation, (ii) amortization of intangibles, (iii) interest expense (income), (iv) income tax provision, (v) costs incurred in connection with Audit Committee Investigation and Restatement, (vi) the effect of the change in revenue recognition on net income, (vii) share-based compensation, (viii) impairment of intangibles, (ix) loss on divestiture, and (x) one-time inventory fair value adjustments.
A reconciliation of GAAP Net Income (Loss) to EBITDA and Adjusted EBITDA appears in the table below (in thousands):
Years Ended December 31 | |||||||||||
2019 | 2018 | 2017 | |||||||||
Net (loss) income | $ | (25,580 | ) | $ | (29,979 | ) | $ | 64,727 | |||
Non-GAAP Adjustments: | |||||||||||
Depreciation expense | 6,546 | 5,882 | 4,087 | ||||||||
Amortization of intangible assets | 1,039 | 1,034 | 1,678 | ||||||||
Interest expense (income), net | 4,708 | (527 | ) | 87 | |||||||
Income tax provision (benefit) expense | (5 | ) | 26,582 | (19,639 | ) | ||||||
EBITDA | (13,292 | ) | 2,992 | 50,940 | |||||||
Additional Non-GAAP Adjustments: | |||||||||||
Costs incurred in connection with Audit Committee Investigation and Restatement | 66,504 | 51,322 | — | ||||||||
Effect of change in revenue recognition | (24,450 | ) | — | — | |||||||
Share-based compensation | 12,064 | 14,768 | 21,195 | ||||||||
Impairment of intangible assets | 1,258 | — | 590 | ||||||||
Loss on divestiture | — | — | 1,048 | ||||||||
One-time inventory fair value adjustments in connection with acquisition | — | — | 203 | ||||||||
Adjusted EBITDA | $ | 42,084 | $ | 69,082 | $ | 73,976 | |||||
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Based on our lack of market risk sensitive instruments outstanding at December 31, 2019, we have determined that we had no material market risk exposure as of such date.
77
Item 8. Financial Statements and Supplementary Data
Index to Financial Statements | |
Consolidated Balance Sheets – As of December 31, 2019 and 2018 | |
Schedule II - Valuation and Qualifying Accounts | |
F- 1
Report of Independent Registered Public Accounting Firm
Stockholders and Board of Directors
MiMedx Group, Inc.
Marietta, Georgia
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of MiMedx Group, Inc. (the “Company”) as of December 31, 2019 and 2018, the related consolidated statements of operations, stockholders’ equity, and cash flows for each of the three years in the period ended December 31, 2019, and the related notes and schedule (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2019 and 2018, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2019, in conformity with accounting principles generally accepted in the United States of America.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (“PCAOB”), the Company's internal control over financial reporting as of December 31, 2019, based on criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”) and our report dated July 6, 2020 expressed an adverse opinion thereon.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ BDO USA, LLP
We have served as the Company's auditor since 2019.
Atlanta, Georgia
July 6, 2020
F- 2
Report of Independent Registered Public Accounting Firm
Stockholders and Board of Directors
MiMedx Group, Inc.
Marietta, Georgia
Opinion on Internal Control over Financial Reporting
We have audited MiMedx Group, Inc.’s (the “Company’s”) internal control over financial reporting as of December 31, 2019, based on criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (the “COSO criteria”). In our opinion, the Company did not maintain, in all material respects, effective internal control over financial reporting as of December 31, 2019, based on the COSO criteria.
We do not express an opinion or any other form of assurance on management’s statements referring to any corrective actions taken by the Company after the date of management’s assessment.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (“PCAOB”), the consolidated balance sheets of the Company as of December 31, 2019 and 2018, the related consolidated statements of operations, stockholders’ equity, and cash flows for each of the three years in the period ended December 31, 2019, and the related notes and schedule (collectively referred to as “the financial statements”)” and our report dated July 6, 2020 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Item 9A, Management’s Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit of internal control over financial reporting in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audit also included performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis. The following material weaknesses have been identified and described in management’s assessment:
• | Failure to maintain an effective control environment based on the criteria in the COSO framework; |
• | Failure to design, implement and maintain controls over certain information technology systems, financial reporting, income tax, revenue, inventory, and procure-to-pay processes, including certain accruals for expenses such as stock-based compensation expense. |
These material weaknesses were considered in determining the nature, timing, and extent of audit tests applied in our audit of the 2019 financial statements, and this report does not affect our report dated July 6, 2020 on those financial statements.
Definition and Limitations of Internal Control over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are
F- 3
being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ BDO USA, LLP
Atlanta, Georgia
July 6, 2020
F- 4
MIMEDX GROUP, INC. AND SUBSIDIARIES CONSOLIDATED BALANCE SHEETS (in thousands, except share data) | |||||||
December 31, | |||||||
2019 | 2018 | ||||||
ASSETS | |||||||
Current assets: | |||||||
Cash and cash equivalents | $ | $ | |||||
Accounts receivable | |||||||
Inventory, net | |||||||
Prepaid expenses | |||||||
Income tax receivable | |||||||
Other current assets | |||||||
Total current assets | |||||||
Property and equipment, net | |||||||
Right of use asset | — | ||||||
Goodwill | |||||||
Intangible assets, net | |||||||
Other assets | |||||||
Total assets | $ | $ | |||||
LIABILITIES AND STOCKHOLDERS’ EQUITY | |||||||
Current liabilities: | |||||||
Accounts payable | $ | $ | |||||
Accrued compensation | |||||||
Accrued expenses | |||||||
Current portion of long term debt | |||||||
Other current liabilities | |||||||
Total current liabilities | |||||||
Long term debt, net | |||||||
Other liabilities | |||||||
Total liabilities | |||||||
Commitments and contingencies (Note 16) | |||||||
Stockholders’ equity: | |||||||
Preferred stock; $0.001 par value; 5,000,000 shares authorized and 0 shares issued and outstanding | |||||||
Common stock; $0.001 par value; 150,000,000 shares authorized; 112,703,926 issued and 110,818,649 outstanding at December 31, 2019 and 112,703,926 issued and 109,098,663 outstanding at December 31, 2018 | |||||||
Additional paid-in capital | |||||||
Treasury stock at cost: 1,885,277 shares at December 31, 2019 and 3,605,263 shares at December 31, 2018 | ( | ) | ( | ) | |||
Accumulated deficit | ( | ) | ( | ) | |||
Total stockholders’ equity | |||||||
Total liabilities and stockholders’ equity | $ | $ | |||||
See notes to the consolidated financial statements.
F- 5
MIMEDX GROUP, INC. AND SUBSIDIARIES CONSOLIDATED STATEMENTS OF OPERATIONS (in thousands, except share and per share data) | |||||||||||
Years Ended December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Net sales | $ | $ | $ | ||||||||
Cost of sales | |||||||||||
Gross profit | |||||||||||
Operating expenses: | |||||||||||
Selling, general and administrative | |||||||||||
Investigation, restatement and related | |||||||||||
Research and development | |||||||||||
Amortization of intangible assets | |||||||||||
Impairment of intangible assets | |||||||||||
Operating (loss) income | ( | ) | ( | ) | |||||||
Other income (expense) | |||||||||||
Loss on divestiture of Stability | ( | ) | |||||||||
Interest (expense) income, net | ( | ) | ( | ) | |||||||
Other income, net | |||||||||||
(Loss) income before income tax provision | ( | ) | ( | ) | |||||||
Income tax provision benefit (expense) | ( | ) | |||||||||
Net (loss) income | $ | ( | ) | $ | ( | ) | $ | ||||
Net (loss) income per common share - basic | $ | ( | ) | $ | ( | ) | $ | ||||
Net (loss) income per common share - diluted | $ | ( | ) | $ | ( | ) | $ | ||||
Weighted average shares outstanding - basic | |||||||||||
Weighted average shares outstanding - diluted | |||||||||||
See notes to the consolidated financial statements.
F- 6
MIMEDX GROUP, INC. AND SUBSIDIARIES CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY (in thousands, except share data) | |||||||||||||||||||||||||
Common Stock | Additional Paid-in | Treasury Stock | Accumulated | ||||||||||||||||||||||
Shares | Amount | Capital | Shares | Amount | Deficit | Total | |||||||||||||||||||
Balance at December 31, 2016 | $ | $ | $ | ( | ) | $ | ( | ) | $ | ||||||||||||||||
Share-based compensation expense | — | — | — | — | — | ||||||||||||||||||||
Exercise of stock options | ( | ) | ( | ) | — | ||||||||||||||||||||
Issuance of restricted stock | ( | ) | ( | ) | — | ||||||||||||||||||||
Restricted stock cancellation / forfeited | — | — | ( | ) | — | ||||||||||||||||||||
Shares issued for services performed | — | — | ( | ) | — | ||||||||||||||||||||
Shares repurchased | — | — | — | ( | ) | — | ( | ) | |||||||||||||||||
Shares repurchased for tax withholding on vesting of restricted stock units | — | — | — | ( | ) | — | ( | ) | |||||||||||||||||
Net income | — | — | — | — | — | ||||||||||||||||||||
Balance at December 31, 2017 | $ | $ | $ | ( | ) | $ | ( | ) | $ | ||||||||||||||||
Share-based compensation expense | — | — | — | — | — | ||||||||||||||||||||
Exercise of stock options | — | — | ( | ) | ( | ) | — | ||||||||||||||||||
Issuance of restricted stock | — | — | ( | ) | ( | ) | — | ||||||||||||||||||
Restricted stock cancellation / forfeited | — | — | ( | ) | — | ||||||||||||||||||||
Shares repurchased | — | — | — | ( | ) | — | ( | ) | |||||||||||||||||
Shares repurchased for tax withholding on vesting of restricted stock units | — | — | — | ( | ) | — | ( | ) | |||||||||||||||||
Net loss | — | — | — | — | — | ( | ) | ( | ) | ||||||||||||||||
Balance at December 31, 2018 | $ | $ | $ | ( | ) | $ | ( | ) | $ | ||||||||||||||||
Share-based compensation expense | — | — | — | — | — | ||||||||||||||||||||
Exercise of stock options | — | — | ( | ) | ( | ) | — | ||||||||||||||||||
Issuance of restricted stock | — | — | ( | ) | ( | ) | — | ||||||||||||||||||
Restricted stock cancellation / forfeited | — | — | ( | ) | — | ||||||||||||||||||||
Shares repurchased for tax withholding on vesting of restricted stock units | — | — | — | ( | ) | — | ( | ) | |||||||||||||||||
Net loss | — | — | — | — | — | ( | ) | ( | ) | ||||||||||||||||
Balance at December 31, 2019 | $ | $ | $ | ( | ) | $ | ( | ) | $ | ||||||||||||||||
See notes to the consolidated financial statements.
F- 7
MIMEDX GROUP, INC. AND SUBSIDIARIES CONSOLIDATED STATEMENTS OF CASH FLOWS (in thousands) | |||||||||||
Years Ended December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Cash flows from operating activities: | |||||||||||
Net (loss) income | $ | ( | ) | $ | ( | ) | $ | ||||
Adjustments to reconcile net (loss) income to net cash (used in) provided by operating activities: | |||||||||||
Effect of change in revenue recognition | ( | ) | |||||||||
Share-based compensation | |||||||||||
Depreciation | |||||||||||
Amortization of intangible assets | |||||||||||
Amortization of inventory fair value step-up | |||||||||||
Amortization of deferred financing costs and debt discount | |||||||||||
Amortization of discount on notes receivable | ( | ) | ( | ) | |||||||
Non cash lease expenses | |||||||||||
Change in fair value of earn-out consideration | ( | ) | |||||||||
Loss on fixed asset disposal | |||||||||||
Intangible asset impairment | |||||||||||
Change in deferred income taxes | ( | ) | |||||||||
Loss on divestiture of Stability | |||||||||||
Increase (decrease) in cash, net of effects of divestiture, resulting from changes in: | |||||||||||
Accounts receivable | ( | ) | ( | ) | |||||||
Inventory | ( | ) | |||||||||
Prepaid expenses | ( | ) | ( | ) | |||||||
Income tax receivable | ( | ) | |||||||||
Other assets | ( | ) | |||||||||
Accounts payable | ( | ) | ( | ) | |||||||
Accrued compensation | ( | ) | |||||||||
Accrued expenses | ( | ) | ( | ) | |||||||
Income taxes | ( | ) | |||||||||
Other liabilities | ( | ) | |||||||||
Net cash flows (used in) provided by operating activities | ( | ) | |||||||||
Cash flows from investing activities: | |||||||||||
Purchases of property and equipment | ( | ) | ( | ) | ( | ) | |||||
Proceeds from property and equipment sale | |||||||||||
Principal payments from note receivable | |||||||||||
Patent application costs | ( | ) | ( | ) | ( | ) | |||||
Net cash flows provided by (used in) investing activities | ( | ) | ( | ) | |||||||
Cash flows from financing activities: | |||||||||||
Proceeds from term loan | |||||||||||
Repayment of term loan | ( | ) | |||||||||
Deferred financing costs | ( | ) | |||||||||
Shares repurchased for tax withholdings on vesting of restricted stock | ( | ) | ( | ) | ( | ) | |||||
F- 8
MIMEDX GROUP, INC. AND SUBSIDIARIES CONSOLIDATED STATEMENTS OF CASH FLOWS (in thousands) | |||||||||||
Proceeds from exercise of stock options | |||||||||||
Shares repurchased under repurchase plan | ( | ) | ( | ) | |||||||
Payments under capital lease obligations | — | ( | ) | ( | ) | ||||||
Net cash flows provided by (used in) financing activities | ( | ) | ( | ) | |||||||
Net change in cash | ( | ) | |||||||||
Cash and cash equivalents, beginning of year | |||||||||||
Cash and cash equivalents, end of year | $ | $ | $ | ||||||||
See notes to the consolidated financial statements.
F- 9
MIMEDX GROUP, INC. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
1. |
MiMedx Group, Inc. (together with its subsidiaries except where the context otherwise requires “MiMedx,” or the “Company”) is an advanced wound care and emerging therapeutic biologics company, developing and distributing human placental tissue allografts with patent-protected processes for multiple sectors of healthcare. The Company derives its products from human placental tissues processed using proprietary processing methodologies. The Company’s mission is to offer physicians products and tissues to help the body heal itself. MiMedx provides products in the wound care, burn, surgical, orthopedic, spine, sports medicine, ophthalmic and dental sectors of healthcare. All of the Company’s products are regulated by the United States Food and Drug Administration (“FDA”).
MiMedx is the leading supplier of human placental allografts, which are human tissues that are transplanted from one person (a donor) to another person (a recipient). The Company operates in one business segment, Regenerative Biomaterials, which includes the design, manufacture, and marketing of products and tissue processing services for the wound care, burn, surgical, sports medicine, ophthalmic and dental sectors of healthcare. The Company’s allograft product families include: dHACM family with AmnioFix® and EpiFix® brands; Umbilical family with EpiCord® and AmnioCord® brands; and Placental Collagen family with AmnioFill™ brands. AmnioFix and EpiFix are tissue allografts derived from amnion and chorion layers of human placental membrane; EpiCord and AmnioCord are tissue allografts derived from umbilical cord tissue. AmnioFill is a placental connective tissue matrix, derived from the placental disc and other placental tissue.
2. | Liquidity and Capital Resources |
Net Working Capital
As of December 31, 2019, the Company had $69.1 million of cash and cash equivalents. The Company reported total current assets of $123.2 million and current liabilities of $67.3 million and had net working capital of $55.9 million as of December 31, 2019.
Overall Liquidity and Capital Resources
The Company’s largest cash requirement for the twelve months ended December 31, 2019 was cash for general working capital needs; investigation, restatement, and related expenses; and other cash requirements included capital expenditures. The Company funded its cash requirements for 2019 through its existing cash reserves, the Term Loan (as defined below) received in 2019 and its operating activities during the period. The Company believes that its anticipated cash from operating and financing activities and existing cash and cash equivalents will enable the Company to meet its operational liquidity needs and fund its planned investing activities for the twelve months from issuance of the consolidated financial statements.
As discussed in Note 16, “Commitments and Contingencies,” of the consolidated financial statements, the Company anticipates additional cash requirements related to the following items within one year from the date of filing this Form 10-K:
• | private securities lawsuits, for which the Company is currently not able to estimate a loss and for which it is unclear whether the Company would be indemnified under various insurance policies; and |
• | investments and other expenditures required in order to bring the Company’s facilities into compliance with current Good Manufacturing Practices (“cGMPs”). |
As discussed further in Note 21, “Subsequent Events,” the Company consummated the Preferred Stock Transaction (as defined below), entered into a new loan facility with Hayfin Services, LLP, repaid the BT Term Loan (as defined below), and terminated the BT Loan Agreement.
The Company has analyzed its ability to address the aforementioned commitments and potential liabilities while remaining compliant with the financial covenants set forth in the Hayfin Loan Agreement (as defined below) for the 12 months from the date of the issuance of the consolidated financial statements, consistent with the guidance prescribed by Financial Accounting Standards Board (“FASB”) Accounting Standards Update (“ASU”) 2014-05, “Going Concern: Disclosure of Uncertainties about an Entity’s Ability to Continue as a Going Concern.”
F- 10
Based on this analysis, and in combination with existing cash on hand, it is probable that the Company will be able to meet all obligations as they become due while remaining in compliance with the financial covenants set forth by the Hayfin Loan Agreement. Therefore, the Company concluded that there is no substantial doubt surrounding its ability to continue as a going concern.
3. Significant Accounting Policies
Use of Estimates
The consolidated financial statements have been prepared in accordance with generally accepted accounting principles (“GAAP”) in the United States of America (“U.S.”). Conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported consolidated statements of operations during the reporting period. Actual results could differ from those estimates. Significant estimates include estimated useful lives and potential impairment of property and equipment, goodwill and intangible assets, estimate for contingent liabilities, estimate of allowance for doubtful accounts, management’s assessment of the Company’s ability to continue as a going concern, estimate of fair value of share-based payments and valuation of deferred tax assets.
Principles of Consolidation
The consolidated financial statements include the accounts of MiMedx Group, Inc. and its wholly-owned subsidiaries, including, for the periods prior to its divestiture further discussed in Note 4, Stability Biologics, LLC (“Stability”) formerly known as Stability, Inc. All intercompany balances and transactions have been eliminated upon consolidation.
Segment Reporting
Accounting Standards Codification (“ASC”) 280, “Segment Reporting” requires use of the “management approach” model for segment reporting. The management approach model is based on the way a company’s chief operating decision-maker organizes segments within the Company for which separate discrete financial information is available regarding resource allocation and assessing performance. The Company has determined it has one operating segment.
Market Concentrations and Credit Risk
Cash and Cash Equivalents
Cash and cash equivalents include cash and FDIC insured certificates of deposit held at various banks with an original maturity of three months or less.
Accounts Receivable
Accounts receivable represent amounts due from customers for which revenue has been recognized. Generally, the Company does not require collateral or any other security to support its receivables.
Bad debt expense and the allowance for doubtful accounts are based on historical trends. The Company’s policy to reserve for potential bad debts is based on the aging of the individual receivables. The Company manages credit risk by not selling to customers who are delinquent, generally after sixty days of delinquency. The individual receivables are written-off after all reasonable efforts to collect the funds have been made. Actual write-offs may differ from the amounts reserved.
Notes Receivable
Notes receivable represent formal payment agreements with customers which generally arise in situations where amounts shipped and billed have aged significantly as well as the promissory note issued by Stability as part of the divestiture discussed in Note 4 which was repaid in full in the third quarter of 2019. The Company’s notes receivable are included in other current and long-term assets in the consolidated balance sheets and were valued taking into consideration cost of the market participant inputs, market conditions, liquidity, operating results and other qualitative factors.
F- 11
Inventories
Inventories are valued at the lower of cost or net realizable value, using the first–in, first-out (“FIFO”) method. Inventory is tracked through raw material, work-in-progress, and finished goods stages as the product progresses through various production steps and stocking locations. Labor and overhead costs are absorbed through the various production processes up to when the work order closes. Historical yields and normal capacities are utilized in the calculation of production overhead rates. Reserves for inventory obsolescence are utilized to account for slow-moving inventory as well as inventory no longer needed due to diminished demand.
Property and equipment are recorded at cost and depreciated on a straight-line method over their estimated useful lives, principally three years to seven years . Leasehold improvements are depreciated on a straight-line method over the shorter of the estimated useful lives or the lease term. The Company is party to various lease arrangements for its facility space and equipment. These arrangements include interest, scheduled rent increases and rent holidays which are included in the determination of minimum lease payments when assessing lease classification, and are included in rent expense on a straight line basis over the lease term. See “Lease Obligations” below and Note 7 “Leases,” for further information regarding capital leases, operating leases and rent expense.
Impairment of Long-lived Assets
The Company evaluates the recoverability of its long-lived assets (property and equipment) whenever adverse events or changes in business climate indicate that the expected undiscounted future cash flows from the related assets may be less than previously anticipated. If the net book value of the related assets exceeds the expected undiscounted future cash flows of the assets, the carrying amount would be reduced to the present value of their expected future cash flows and an impairment loss would be recognized.
Goodwill and Indefinite-lived Intangible Assets
Goodwill represents the excess of purchase price over the fair value of net assets of acquired businesses. The Company assesses the recoverability of its goodwill at least annually on September 30, or more frequently whenever events or substantive changes in circumstances indicate that the asset may be impaired. The Company may first choose to assess qualitative factors to determine whether it is more likely than not that the fair value of a reporting unit is less than its carrying amount. If the Company determines that it is more likely than not that the fair value of a reporting unit is less than its carrying amount, then the Company performs a quantitative analysis. The Company may also choose to bypass the qualitative assessment and proceed directly to the quantitative analysis.
When testing for goodwill impairment, the Company first assesses qualitative factors to determine whether the existence of events or circumstances lead to a determination that it is more likely than not that the fair value of a reporting unit is less than its carrying amount. If the Company concludes it is more likely than not that the fair value of a reporting unit is less than its carrying amount, a quantitative fair value test is performed. At present, the Company has only one reporting unit.
Under the quantitative test, if the carrying value exceeds the fair value of the reporting unit, goodwill impairment is recorded for the amount that the reporting unit’s carrying value exceeds its fair value, not to exceed the total amount of goodwill allocated to the reporting unit. The Company determines the fair value utilizing the income and market approaches. Under the income approach, the fair value of the Company is the present value of its future economic benefits. These benefits can include revenue, cost savings, tax deductions, and proceeds from its disposition. Value indications are developed by discounting expected cash flows to their present value at a rate of return that incorporates the risk-free rate for the use of funds, trends within the industry, and risks associated with particular investments of similar type and quality as of the goodwill impairment testing date. Under the market approach, the Company uses its market capitalization which is calculated by taking the Company’s share price times the number of outstanding shares. The Company’s estimates associated with the goodwill impairment test are considered critical due to the amount of goodwill recorded on its consolidated balance sheets and the judgment required in determining fair value, including projected future cash flows.
Acquired indefinite live intangible assets are tested for impairment annually on September 30 or whenever events or changes in circumstances indicate that the carrying amount of an intangible asset may not be recoverable. The Company’s impairment reviews are based on an estimated future cash flow approach that requires significant judgment with respect to future revenue and expense growth estimates. The Company uses estimates consistent with business plans and a market participant view of the assets being evaluated. Actual results may differ from the estimates used in these analyses.
F- 12
Impairment of Intangible Assets with Finite Lives
The Company reviews purchased intangible assets with finite lives for impairment whenever events or changes in circumstances indicate the carrying value of an asset may not be recoverable using a two-step impairment test. In step one, the Company determines the sum of the undiscounted future cash flows of the assets based on management’s estimates and compare it to the carrying value of the assets. If the carrying amount is greater than the sum of the undiscounted cash flows, then the asset is impaired and step two is required. In step two, the impairment loss is calculated as the difference between the fair value of the assets and the carrying value of the assets.
The Company recorded an impairment loss of $0.5 million during 2019. There were no impairment losses recognized with respect to intangible assets with finite lives in 2018 or 2017.
Patent Costs
Lease Obligations
Effective January 1, 2019, the Company accounts for its leases under ASC 842, “Leases”. The Company determines if an arrangement is, or contains, a lease at inception. Right-of-use assets and the related liabilities resulting from operating leases were included in Right of use asset, Other current liabilities and Other liabilities, respectively, in the consolidated balance sheet as of December 31, 2019.
Operating lease assets represent the Company's right to use an underlying asset for the lease term and lease liabilities represent the Company's obligation to make lease payments arising from the lease. Operating lease assets and liabilities are recognized at the lease commencement date based on the estimated present value of lease payments over the lease term. Since most of the Company’s leases do not have a readily determinable implicit discount rate, the Company uses its incremental borrowing rate to calculate the present value of lease payments determined using the rate of interest that the Company would have to pay on collaterialized or secured borrowing over a similar term. Variable components of the lease payments such as fair market value adjustments, utilities, and maintenance costs are expensed as incurred and not included in determining the present value of lease liabilities, which will include options to extend or terminate the lease when it is reasonably certain that the Company will exercise that option. As an accounting policy election, the Company excludes short-term leases having initial terms of 12 months or fewer. Lease expense is recognized on a straight-line basis over the lease term. The Company continues to account for leases in the prior period financial statements under ASC 840. See Note 7, “Leases” for further information regarding lease obligations.
Lease expense for operating lease payments is recognized on a straight-line basis over the term of the lease. Operating lease assets and liabilities are recognized based on the present value of lease payments over the lease term. Since most of the Company’s leases do not have a readily determinable implicit discount rate, the Company uses its incremental borrowing rate to calculate the present value of lease payments determined using the rate of interest that the Company would have to pay on collaterialized or secured borrowing over a similar term. As a practical expedient, the Company has made an accounting policy election not to separate lease components from non-lease components in the event that the agreement contains both. The Company includes both the lease and non-lease components for purposes of calculating the right-of-use asset and related lease liability.
F- 13
Contingencies
The Company is subject to various patent challenges, product liability claims, government investigations, shareholder derivative suits, former employee matters and other legal proceedings, see Note 16 “Commitments and Contingencies.” Legal fees and other expenses related to litigation are expensed as incurred and included in selling, general and administrative expenses in the consolidated statements of operations. The Company records an accrual for legal settlements and other contingencies in the consolidated financial statements when the Company determines that a loss is both probable and reasonably estimable. The Company discloses all ongoing legal matters for which a loss is probable, regardless of whether an estimate can be reasonably determined.
Due to the fact that legal proceedings and other contingencies are inherently unpredictable, the Company’s estimates of the probability and amount of any such liabilities involve significant judgment regarding future events. The actual costs of resolving a claim may be substantially different from the amount of reserve the Company recorded. The Company records a receivable from its product liability insurance carriers only when the resolution of any dispute has been reached and realization of the amounts equal to the potential claim for recovery is considered probable. Any recovery of an amount in excess of the related recorded contingent loss will be recognized only when all contingencies relating to recovery have been resolved.
Revenue Recognition
The Company sells its products primarily to individual customers and independent distributors (collectively referred to as “customers”). In 2017, 2018, and into part of 2019, the Company’s control environment was such that it created uncertainty surrounding all of its customer arrangements, which required consideration related to the proper revenue recognition under the applicable literature. The control environment allowed for the existence of extra-contractual or undocumented terms or arrangements initiated by or agreed to by the Company and former members of Company management at the outset of the transactions (side agreements). Concessions were also agreed to subsequent to the initial sale (e.g. sales above established customer credit limits extended and unusually long payment terms, return or exchange rights, and contingent payment obligations) that called into question the ability to recognize revenue at the time that product was shipped to a customer. The applicable revenue recognition guidance also changed beginning January 1, 2018, which further impacted the Company’s revenue recognition methodology.
As a result, the Company’s application of the applicable revenue recognition guidance varies for of the years ended December 31, 2019, 2018 and 2017. Additionally, the Company changed its pattern of revenue recognition effective October 1, 2019. The application of the relevant revenue recognition guidance and the pattern of revenue recognition are further discussed below for each period presented.
Fiscal Year Ended December 31, 2017
For the year ended December 31, 2017, the Company applied the revenue recognition guidance in ASC Topic 605, Revenue Recognition (“ASC 605”). Under ASC 605, revenue should not be recognized until it is realized or realizable and earned. SEC Staff Accounting Bulletin (“SAB”) Topic 13.A.1 (as codified in ASC 605-10-S99-1) outlines four criteria that generally indicate when revenue is realized or realizable and earned. If any of these criteria are not met, revenue recognition should be deferred until all criteria have been met. Therefore, the Company assessed these four criteria as follows:
1. | Persuasive evidence of an arrangement exists - The Company’s sales are driven either by contracts or purchase orders. These types of documents are typically used to establish persuasive evidence of an arrangement. The Company’s customary business practices, however, must be taken into account as a contract can be written, oral, or modified based on customary business practices. Throughout 2017, although the Company may have created a legal contract upon the execution of a contract and/or fulfillment of a purchase order, the lack of clarity around the final terms of the arrangement due to the pervasive side agreements with customers precluded the Company’s sales transactions from meeting this criterion upon shipment of product. Therefore, even though there may have been a legal contract governing the arrangement (which typically would indicate persuasive evidence of an arrangement), the Company’s selling and collection practices amended the stated contract terms. After considering these factors, the Company concluded that persuasive evidence of an arrangement did not exist upon shipment of product. |
2. | Delivery has occurred or services have been rendered - For sales to customers, physical possession and title transferred upon shipment to the customer. However, the Company concluded that it did not pass the risks of ownership to the customer upon shipment because customers were allowed to return product for multiple reasons, which included being unable to sell the product, damages which may have occurred subsequent to delivery, and dropped product. See below |
F- 14
for additional discussion of the Company’s rationale for concluding that delivery had not yet occurred upon shipment to the customer.
3. | The seller’s price to the buyer is fixed or determinable - At certain quarter-ends, the Company was significantly increasing sales to customers without having visibility into the level of product remaining unsold at the customer’s location. This practice made it difficult to develop an appropriate estimate of future credits to be issued to customers at the time of sale, which, in turn, impacted whether the price at the time of transfer of physical possession to the customer was fixed or determinable. This previous practice in combination with the following actions of the Company precluded the price of the Company’s sales transactions from being fixed or determinable upon shipment of product: |
• | Offering customers an unconditional right of return, |
• | Offering extended payment terms to customers, and |
• | A history of exceeding established credit limits for customers. |
4. | Collectibility is reasonably assured - At the time of transfer of physical possession to the customer, collectibility of the sales was questionable. The Company determined that the customers’ intention to pay amounts when due was uncertain in light of the conflicting messages customers received with respect to the payment terms, rights of return and lack of adherence to credit limits. Although the Company did have processes in place to establish credit limits, evidence indicated that those credit limits were overridden by certain sales personnel and members of management. The Company recovered the majority of its billings made in 2017 with insignificant write-offs recorded; however, a significant amount of these billings were collected well after payment was due under the contractual terms. Furthermore, the quantitative and qualitative evidence gathered by the Company raised considerable doubt as to the collectibility of its billings at the time of shipment, but this evidence was not persuasive enough for the Company to reach a conclusion as to whether collectibility was reasonably assured. |
In the Company’s evaluation of the point at which delivery occurred (the second criterion discussed above), the Company further considered the fact that there are instances under ASC 605 where the transfer of title of the product did not coincide with revenue recognition. Based on its review of all facts and circumstances, the Company determined that it did not meet all of the criteria to recognize revenue at the time of shipment of product to the customer. Specifically, the Company determined that they did not transfer the risks of ownership upon the transfer of physical possession because the Company’s customers were routinely granted an extended return period with very limited restrictions on the right of return and extended payment terms which raised doubt as to the intent or ability of customers to use and pay for the product delivered. Customers were allowed to return product for multiple reasons which included being unable to sell the product, damages which may have occurred subsequent to delivery, and dropped product (i.e., product that becomes contaminated and unusable). In other words, only upon use of the product in a surgical application (whether by the customer or by the ultimate end user in the case of distributors) would the customer no longer have the ability to return the product.
Accordingly, the Company determined that the aforementioned revenue recognition criteria were met only when both of the following events had occurred: (1) the Company fulfilled the customer's purchase order by delivering product ordered, and (2) the Company collected payment for the product delivered. Furthermore, the Company determined that the amount of revenue to be recognized should be limited to the amount of payment received in a given period less the amount expected to be refunded or credited to customers for sales returns made after payment.
An exception to the above revenue recognition under ASC 605 during the year ended December 31, 2017 related to the sales generated by the Company’s wholly owned subsidiary, Stability. On January 13, 2016, the Company completed the acquisition of Stability, a provider of human tissue products to surgeons, facilities, and distributors serving the surgical, spine, and orthopedic sectors of the healthcare industry. For sales of the Company’s products through Stability, the Company recognized revenue under ASC 605 only when both of the following events had occurred: (1) the Company has fulfilled the customer’s purchase order by delivering all product ordered; and (2) the product has been delivered to the customer. Total sales from Stability were $7.0 million for the year ended December 31, 2017. Stability was divested on September 30, 2017.
Prior to 2015, substantially all federal healthcare providers, including the Department of Veterans Affairs, purchased Company product from one distributor customer of the Company, AvKARE Inc. (“AvKARE”), which is a veteran-owned General Services Administration Federal Supply Schedule contractor. In 2015, the Company began selling product directly to federal customers rather than exclusively allowing federal healthcare providers to purchase Company product from AvKARE. Upon expiration of the Company’s agreement with AvKARE on June 30, 2017, the Company had an obligation to repurchase AvKARE’s remaining inventory within 90 days in accordance with the terms of the agreement. As of September 30, 2017, the Company had satisfied the repurchase obligation.
F- 15
Additionally, the Company considered how to account for costs associated with the delivered products of the contract for which revenue has been deferred, which is whether to match the related cost of sales expense with revenue or to recognize expense upon shipment. In making this assessment, the Company considered the financial viability of its distributors and customers based on their creditworthiness to determine if collectibility of amounts sufficient to realize the costs of the products shipped was reasonably assured at the time of shipment. As the Company determined that there was a probable future economic benefit associated with the sales transactions, the Company deferred the costs of sales until the revenue was recognized.
The Company offset deferred revenue with the associated accounts receivable obligations in connection with the sales of products to its customers. The Company believes that because the conditions for revenue recognition have not yet been met and payment has not been received, neither party has fulfilled its obligations under the contract. The amount shipped and billed but not recorded as revenue was $64.8 million for the year ended December 31, 2017.
Fiscal Year Ended December 31, 2018
The Company adopted ASC Topic 606, Revenue from Contracts with Customers (“ASC 606”), on January 1, 2018 by using the modified retrospective method. ASC 606 establishes principles for reporting information about the nature, amount, timing and uncertainty of revenue and cash flows arising from the entity's contracts to provide goods or services to customers. The core principle requires an entity to recognize revenue to depict the transfer of goods or services to customers in an amount that reflects the consideration that it expects to be entitled to receive in exchange for those goods or services recognized as performance obligations are satisfied. The Company assessed the impact of the ASC 606 guidance by reviewing customer contracts and accounting policies and practices to identify differences, including identification of the contract and the evaluation of the Company’s performance obligations, transaction price, customer payments, transfer of control and principal versus agent considerations.
ASC 606 establishes a five-step model for revenue recognition. The first of these steps requires the identification of the contract as described in ASC 606-10-25-1. The specific criteria (the “Step 1 Criteria”) to this determination are as follows:
• | The parties to the contract have approved the contract (in writing, orally, or in accordance with other customary business practices) and are committed to perform their respective obligations; |
• | The entity can identify each party’s rights regarding the goods or services to be transferred; and |
• | The entity can identify the payment terms for the goods or services to be transferred. |
• | The contract has commercial substance. |
• | It is probable that the entity will collect substantially all of the consideration to which it will be entitled in exchange for the goods or services that will be transferred to the customer. |
The Company concluded that the first three of the above criteria were not met upon shipment of product to the customer, the fourth criteria had been met and the Company acknowledges that there is a degree of uncertainty as to whether last criteria above had been met. Although the parties to the contract may have approved the contract and purchase orders in writing, the Company concluded that upon shipment of products to the customer there is not sufficient evidence that its customers were committed to perform their obligations defined in the contract due to the existence of extra-contractual or undocumented terms or arrangements (e.g., regarding payment terms, right of return, etc.). The Company could not reliably identify each party’s rights regarding the products to be transferred upon shipment of those products to customers. The Company’s sales personnel continued to make side agreements with customers which directly conflicted with the explicitly stated terms of sale. These side agreements created significant ambiguity around the rights and obligations of both parties involved in the transaction. This practice continued to result in extended payment terms and returns occurring long after the original sale was made. The Company’s business practices created an implied right for the customer to demand future, unknown, performance by the Company. As a result, each party (and in particular the Company) could not at the time of product shipment adequately determine its rights regarding the good transferred as required by ASC 606-10-25-1. Upon shipment of product to the customer, the Company could not reliably identify the payment terms for the products it sold to customers. Although the written payment terms were known to both parties, the Company’s pervasive business practices (e.g., informal and undocumented side agreements) overrode the written payment terms and often resulted in extensions of the terms for payment. The Company’s contracts did appear to have commercial substance (i.e., the risk, timing, or amount of the Company’s future cash flows was expected to change as a result of the contract) upon fulfillment of a purchase order, as most fulfillments have eventually resulted in the Company receiving cash. Therefore, the Company concluded that this criterion appears to be met upon shipment of product to customers (i.e., fulfillment of the purchase order).
F- 16
The probability that the Company would collect the consideration to which it was entitled in exchange for products shipped to the customer was questionable. In evaluating whether the collectibility of an amount of consideration was probable, the Company considered the customer’s ability and intention to pay that amount of consideration when it was due. Historically, the customers’ intention to pay amounts when due was uncertain in light of the conflicting messages customers received with respect to the payment terms and rights of return and lack of adherence to credit limits. The assessment in ASC 606 is based on whether the customer has the ability and intention to pay for the product being delivered by the Company. Assessment of a customer’s ability to pay is typically done through a credit check process and the establishment of a credit limit for each customer by the Company’s accounts receivable team. Although the Company did have a process in place to establish credit limits, the evidence previously mentioned indicates that those credit limits were routinely overridden by certain sales personnel and members of management. Despite these overrides, the Company recovered the majority of its billings made in 2018. Furthermore, the quantitative and qualitative evidence gathered by the Company raised considerable doubt as to the collectibility of its billings at the time of shipment, but this evidence was not persuasive enough for the Company to conclude that collectibility was not probable. As a result of the considerations outlined above, the Company determined that it did not meet the criteria necessary for its revenue arrangements to qualify as “contracts” under the requirements of ASC 606 (i.e., these arrangements did not pass the Step 1 Criteria of the revenue recognition model).
The Company’s inability to fulfill these criteria was due to uncertainties of contractual adjustments with customers created by a combination of an inappropriate tone at the top and extra-contractual arrangements. Consequently, as of the date of the Company’s adoption of ASC 606 effective January 1, 2018 and for the remainder of the year ended December 31, 2018, the Company concluded that it did not meet the Step 1 Criteria upon physical delivery of the product. Subsequent to the delivery of product, uncertainties surrounding contractual adjustment were not resolved until either: (1) the customer returned the product prior to payment; or (2) the Company received payment from the customer. At that point, the Company determined that an accounting contract existed and the performance obligations of the Company to deliver product and the customer to pay for the product were satisfied. The Company determined the transaction price of its contracts to equal the amount of consideration received from customers less the amount expected to be refunded or credited to customers, which is recognized as a refund liability that is updated at the end of each reporting period for changes in circumstances. The refund liability is included within accrued expenses in the consolidated balance sheet.
The Company continued to defer the costs of sales consistent with the assessment noted above for the year ended December 31, 2017. The Company also continued to offset deferred revenue with the associated accounts receivable obligations in connection with the sales of products to its customers. The amount shipped and billed but not recorded as revenue was $51.0 million for the year ended December 31, 2018.
Fiscal Year Ended December 31, 2019
The Company continued to assess contracts, new and existing, throughout 2019 to determine if the Step 1 Criteria noted above for the determination of a contract under ASC 606 were met for new contracts at the outset of a sales transaction (i.e., upon shipment of product) or for existing contracts at some point within 2019 when all the terms of the arrangement would have been known. Until it was determined if the Step 1 Criteria had been met, revenue recognition continued to be deferred consistent with the assessment for the year ended December 31, 2018.
As further discussed above, the primary factors contributing to the determination in prior periods that the Step 1 Criteria had not been met were the inappropriate tone at the top and the existence of pervasive extra-contractual or undocumented terms or arrangements. These prior business practices and the lack of transparency surrounding them created a systemically implied right for customers to demand future, unknown, performance by the Company. Although some of the former executives were employed by the Company only through June 2018, the Company determined that based on the impact of the prior tone at the top, the continued internal sales force strategy and the existing customer base’s continued expectations (based on past practice), there would be flexibility with respect to arrangement terms even after delivery of the product so pervasive that all customer arrangements continued to be subject to uncertain modification of terms into 2019.
After identifying the primary factors contributing to the lack of knowledge regarding its customer contractual terms, the Company began implementing changes in mid-2018 to remediate the pervasive weaknesses in the control environment, followed by gradually implementing measures to empower its compliance, legal, and accounting departments, educating its sales force on appropriate business practices, and communicating its revised terms of sale to customers. The Company assessed its efforts throughout 2019 to determine when, if at any point, the factors contributing to the inability to satisfy the Step 1 Criteria were sufficiently addressed such that the Step 1 Criteria were met at the time of physical delivery to the customer. Determining when these conditions were effectively satisfied was a matter of judgment; however, the Company determined that adequate knowledge of the contractual arrangements with its customers did exist in 2019 for new and certain existing arrangements. Management did note that there is no single determinative change that overcame the pervasive challenges noted above, but rather an accumulation of efforts that
F- 17
taken together, resulted in sufficient knowledge of contractual relationships both internally within the Company and externally with its customers.
To address the tone at the top issues, the Company noted that proper remediation involved not only the removal of members of management who were setting an inappropriate tone but also the establishment of new management throughout the organization who emphasized a commitment to integrity, ethical values and transparency and have that reinforcement for a sustained period of time. The changes made to management positions throughout the organization and the resulting organization behavior changes were assessed to have been sufficiently addressed by mid-2019.
To determine when the Company had either eliminated or had sufficient knowledge to identify any extra-contractual arrangements, the Company noted that a key factor contributing to its historical lack of visibility into the arrangements with its customers was the failure to adhere to credit limits, payment terms and return policies. The establishment of additional controls and the emphasis on adherence to the Company’s existing policies and controls was an iterative process that continued through the first two quarters of 2019. Additional factors contributing to the increased visibility into its contractual arrangements involved further education and training of the sales personnel regarding the Company’s terms and conditions as well as monitoring of the sales personnel and customers for compliance with the contractual arrangements. The Company implemented a disciplined approach to educating the sales personnel regarding the prior practices that were considered unacceptable, ensuring they were knowledgeable regarding current terms and conditions and implementing an open dialogue with the credit and collections department. Monitoring of the customer base was accomplished through a variety of measures including, but not limited to, analysis of payments made within the original terms, levels of returns post-shipment, and various continued communication with the customer account representatives by members of the Company’s credit and collections department. During the third quarter of 2019, management determined that these efforts with the sales personnel and the external customers had been in place for a sufficient period of time to provide the customers an understanding of the Company’s contractual arrangements with them.
Therefore, beginning October 1, 2019, for all new customer arrangements, the Company determined adequate measures were in place to understand the terms of its contracts with customers. As such, beginning October 1, 2019, the Company concluded that the Step 1 Criteria would be met prior to shipment of product to the customer or implantation of the products on consignment.
The Company also reassessed whether the Step 1 Criteria had been met for all shipments of products where payment had not been received as of September 30, 2019. While the measures summarized above provided significant evidence necessary to understand the terms of the Company’s contractual arrangements with its customers, certain of these customers continued to exhibit behaviors that resulted in extended periods until cash collection. Such delays in collection suggested that uncertainty regarding extracontractual arrangements may continue, particularly as it relates to payment terms. As a result, the Company concluded the following for any existing arrangements, which remained unpaid at September 30, 2019:
• | For customer arrangements where collection was considered probable within 90 days from the date of original shipment or implantation of the products, the Company concluded the Step 1 Criteria were met (the “Transition Adjustment”). |
• | For the remaining customer arrangements (the “Remaining Contracts”), the Company concluded that due to the uncertainty that extracontractual arrangements may continue the Step 1 Criteria would not be satisfied until the Company receives payment from the customer. At that point, the Company determined that an accounting contract would exist and the performance obligations of the Company to deliver product and the customer to pay for the product would be satisfied. As of December 31, 2019, upon reassessment, the Company concluded that the Step 1 Criteria continued to not be met due to the same circumstances described above. |
The Company continued to record the deferred costs of sales on the arrangements that failed the Step 1 Criteria where collectibility was reasonably assured and will recognize the costs when the related revenue is recognized. The Company also continued to offset deferred revenue with the associated accounts receivable obligations for these arrangements that continued to fail the Step 1 Criteria.
For all customer transactions concluded to meet the Step 1 Criteria, the Company then assessed the remaining criteria of ASC 606 to determine the proper timing of revenue recognition.
Under ASC 606, the Company recognizes revenue following the five-step model: (i) identify the contracts with a customer (the Step 1 Criteria); (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the entity satisfies a performance obligation. As noted above, beginning October 1, 2019, the Company determined that they had met the Step 1 Criteria for new customer arrangements. The Company has determined that the performance obligation was met upon delivery of the product to the customer, or at the time the product is implanted for products on consignment, at which point the Company determined it will collect the consideration it is entitled to in exchange for the product transferred to the customer. As a result, the Company
F- 18
recognizes as revenue the amount of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied, generally upon shipment of the product to the customer. The nature of the Company’s contracts gives rise to certain types of variable consideration, including rebates and other discounts. The Company includes estimated amounts of variable consideration in the transaction price to the extent that it is probable there will not be a significant reversal of revenue. Estimates are based on historical or anticipated performance. The Company does have consignment agreements with several customers and distributors which allow the Company to better market its products by moving them closer to the end user. In these cases, the Company determined that it has fulfilled its performance obligation once control of the product has been delivered to the customer, which occurs simultaneously with the product being implanted.
The Company acts as the principal in all of its customer arrangements and therefore records revenue on a gross basis. Shipping is considered immaterial in the context of the overall customer arrangement, and damages or loss of goods in transit are rare. Therefore, shipping is not deemed a separately recognized performance obligation and the Company has elected to treat shipping costs as activities to fulfill the promise to transfer the product. The Company maintains a returns policy that allows its customers to return product that is consigned, damaged or non-conforming, ordered in error, or due to a recall. The estimate of the provision for returns is based upon historical experience with actual returns given consideration to any changes in historical periods presented. The Company’s payment terms for customers are typically 30 to 60 days from receipt of title of the goods.
Based on the assessment noted above, the Company concluded that through the first two quarters of 2019, the pattern of revenue recognition under ASC 606 remained the same as the application for the year ended December 31, 2018; that is, revenue was deferred until the product was paid for or returned. In order to account for the determination that the Step 1 Criteria had been met during the third quarter of 2019, for certain existing customer arrangements, the Company recorded the following (in thousands):
Amounts Invoiced and Not Collected | Deferred Cost of Sales | ||||||
Amounts as of September 30, 2019 | $ | $ | |||||
Revenue recognized related to amounts invoiced and not collected at September 30, 2019: | |||||||
Transition Adjustment during the three months ended September 30, 2019 | ( | ) | ( | ) | |||
Cash collected during the three months ended December 31, 2019 related to the Remaining Contracts | ( | ) | ( | ) | |||
( | ) | ( | ) | ||||
Write-off of customer contracts where collection is no longer reasonably assured (a) | ( | ) | ( | ) | |||
Amounts as of December 31, 2019 | $ | $ | |||||
(a) The Company determined that for approximately $10.3 million of existing contracts where payment had not been received, collection was no longer reasonably assured. As a result, $1.4 million of deferred cost of sales relating to these customers was written off. Any future collections relating to these customer contracts will be recorded as revenue at the time payment is received. | |||||||
GPO Fees
The Company sells to Group Purchasing Organization (“GPO”) members who transact directly with the Company at GPO-agreed pricing. GPOs are funded by administrative fees that are paid by the Company. These fees are set as a percentage of the purchase volume, which is typically 3 % of sales made to the GPO members. Prior to adoption of ASC 606, for all periods presented prior to January 1, 2018, the Company presented the administrative fees paid to GPOs as a reduction of revenues as the benefit received by the Company in exchange for the GPO fees was not sufficiently separable from the GPO member’s purchase of the Company’s products. Upon adoption of ASC 606, the Company concluded that although it benefited from the access that a GPO provides to its members, this benefit was neither distinct from other promises in the Company’s contracts with GPOs nor was the benefit separable from the sale of goods by the Company to the end customer. Therefore, the Company continued presenting fees paid to GPOs as a reduction of product revenues.
Cost of Sales
Cost of sales includes all costs directly related to bringing the Company’s products to their final selling destination. Amounts include direct and indirect costs to manufacture products including raw materials, personnel costs and direct overhead expenses necessary to convert collected tissues into finished goods, product testing costs, quality assurance costs, facility costs associated with the Company’s manufacturing and warehouse facilities, including depreciation, freight charges, costs to operate equipment and other shipping and handling costs for products shipped to customers.
F- 19
Prior to the Transition, the Company deferred the cost of sales resulted from transactions where title to inventory transferred from the Company to the customer, but for which all revenue recognition criteria have not yet been met. Once all revenue recognition criteria are met, the revenue and associated cost of sales was recognized. These amounts were recorded within other current assets on the consolidated balance sheet in the amount of $4.3 million as of December 31, 2018.
Research and Development Costs
Research and development costs consist of direct and indirect costs associated with the development of the Company’s technologies. These costs are expensed as incurred.
Advertising expense
Income Taxes
Income tax expense (benefit), deferred tax assets and liabilities, and liabilities for unrecognized tax benefits reflect management’s best assessment of estimated current and future taxes to be paid. The Company is subject to income taxes in the United States, including numerous state jurisdictions.
Deferred income taxes arise from temporary differences between the tax basis of assets and liabilities and their reported amounts in the financial statements, which will result in taxable or deductible amounts in the future. The Company recognizes deferred tax assets to the extent that it believes these assets are more likely than not to be realized. If the Company determines that it would be able to realize its deferred tax assets in the future in excess of their net recorded amount, the Company would make an adjustment to the deferred tax asset valuation allowance.
In evaluating the Company’s ability to recover its deferred tax assets within the jurisdiction from which they arise, management considers all available positive and negative evidence, including scheduled reversals of deferred tax liabilities, projected future taxable income, tax-planning strategies, and results of recent operations. In projecting future taxable income, the Company begins with historical results and incorporates assumptions about the amount of future state and federal pretax operating income adjusted for items that do not have tax consequences. The assumptions about future taxable income require significant judgment and are consistent with the plans and estimates the Company uses to manage the underlying businesses. In evaluating the objective evidence that historical results provide, management considers three years of cumulative income (loss). The Company accounts for income taxes under the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements. Under this method, deferred tax assets and liabilities are determined on the basis of the differences between the financial statement and tax bases of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. The effect of a change in tax rates on deferred tax assets and liabilities is recognized in the tax provision (benefit) in the period that includes the enactment date.
The calculation of income tax liabilities involves dealing with uncertainties in the application of complex tax laws and regulations both for U.S. federal income tax purposes and across numerous state jurisdictions. ASC Topic 740 (“ASC 740”) states that a tax benefit from an uncertain tax position may be recognized when it is more likely than not that the position will be sustained upon examination, including resolutions of any related appeals or litigation processes, on the basis of the technical merits. The Company (1) records unrecognized tax benefits as liabilities in accordance with ASC 740 included within other liabilities on the consolidated balance sheets, and (2) adjusts these liabilities when management’s judgment changes as a result of the evaluation of new information not previously available. Because of the complexity of some of these uncertainties, the ultimate resolution may result in a payment that is materially different from management’s current estimate of the unrecognized tax benefit liabilities. These differences will be reflected as increases or decreases to the deferred tax asset or income tax expense in the period in which new information is available.
The Company records uncertain tax positions in accordance with ASC 740 on the basis of a two-step process whereby (1) it determines whether it is more likely than not that the tax positions will be sustained on the basis of the technical merits of the position, and (2) for those tax positions that meet the more-likely-than-not recognition threshold, it recognizes the largest amount of tax benefit that is more than 50 percent likely to be realized upon ultimate settlement with the related tax authority.
F- 20
The Company recognizes interest and penalties related to unrecognized tax benefits within the income tax expense line in the consolidated statements of operations. Accrued interest and penalties, if any, are included within the related deferred tax liability line in the consolidated balance sheet and recorded as a component of income tax expense.
The Company grants share-based awards to employees and members of the Company’s Board of Directors (the “Board”) and non-employee consultants. Such awards are recognized as share-based payment expense over the requisite service or vesting period, to the extent such awards are expected to vest in accordance with FASB ASC Topic 718 “Compensation—Stock Compensation,” and under the issued guidance following FASB’s pronouncement, ASU 2018-07, “Compensation-Stock Compensation (Topic 718): Improvements to Nonemployee Share-Based Payment Accounting”, which the Company adopted on January 1, 2019, the effective date of the new guidance. The amount of expense to be recognized is determined by the fair value of the award using inputs available as of the grant date.
The fair value of restricted common stock is the value of common stock on the grant date. The fair value of stock option grants is estimated using the Black-Scholes option pricing model. Use of the valuation model requires management to make certain assumptions with respect to selected model inputs. The Company uses the simplified method for share-based compensation to estimate the expected term. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant for the estimated option expected term. The Company estimates volatility using a blend of its own historical stock price volatility as well as that of market comparable publicly traded peer companies, since historically, the Company did not have enough history to establish volatility based upon its own stock trading. The Company routinely reviews its calculation of volatility for potential changes in future volatility, the Company’s life cycle, its peer group, and other factors. In addition, an expected dividend yield of zero is used in the option valuation model because the Company does not pay cash dividends and does not expect to pay any cash dividends in the foreseeable future.
For awards with service conditions only, the Company recognizes share-based compensation expense on a straight-line basis over the requisite service or vesting period. For awards with service and performance-based vesting conditions, the Company recognizes stock-based compensation expense using the graded vesting method over the requisite service period beginning in the period in which the awards are deemed probable to vest. Vesting probability for an award with performance vesting conditions is assessed based upon the Company’s expectations to become compliant with applicable securities law regulatory requirements and reporting obligations, as well as other performance vesting conditions specified in the restricted share unit award agreements. The Company recognizes the cumulative effect of changes in the probability outcomes in the period in which the changes occur.
The Company recognizes the fixed dollar amount known on a grant date with respect to the restricted stock unit awards that will be settled by issuing shares on the vesting date, with the number of shares to be determined based on the Company’s stock price on the settlement date over the vesting period, with an offsetting liability. Once the number of shares has been fixed and the shares are issued, the Company reclassifies the liability related to the restricted share unit awards to equity.
Basic net (loss) income per share is determined by dividing net (loss) income by the weighted average ordinary shares outstanding during the period. Diluted net income per ordinary share is based on the weighted average number of ordinary shares outstanding and potentially dilutive ordinary shares outstanding determined by using the treasury stock method. For all periods presented with a net loss, the shares underlying the common share options, warrants and restricted stock have been excluded from the calculation because their effect would have been anti‑dilutive. Therefore, the weighted average shares outstanding used to calculate both basic and diluted loss per share are the same for periods with a net loss.
Fair Value of Financial Instruments and Fair Value Measurements
The respective carrying value of certain on-balance sheet financial instruments approximated their fair values due to the short-term nature and type of these instruments. These financial instruments include cash and cash equivalents, accounts receivable, notes receivable, and certain current financial liabilities.
F- 21
Fair value financial instruments are recorded in accordance with the fair value measurement framework. The fair value measurement framework includes a fair value hierarchy that prioritizes observable and unobservable inputs used to measure fair values in their broad levels. These levels from highest to lowest priority are as follows:
• | Level 1: Quoted prices (unadjusted) in active markets that are accessible at the measurement date for identical assets or liabilities; |
• | Level 2: Quoted prices in active markets for similar assets or liabilities or observable prices that are based on inputs not quoted on active markets, but corroborated by market data. |
• | Level 3: Unobservable inputs or valuation techniques that are used when little or no market data is available. |
The determination of fair value and the assessment of a measurement’s placement within the hierarchy require judgment. Level 3 valuations often involve a higher degree of judgment and complexity. Level 3 valuations may require the use of various cost, market, or income valuation methodologies applied to unobservable management estimates and assumptions. Management’s assumptions could vary depending on the asset or liability valued and the valuation method used. Such assumptions could include: estimates of prices, earnings, costs, actions of market participants, market factors, or the weighting of various valuation methods. The Company may also engage external advisors to assist it in determining fair value, as appropriate.
Although the Company believes that the recorded fair value of its financial instruments is appropriate, these fair values may not be indicative of net realizable value or reflective of future fair values.
Recently Adopted Accounting Pronouncements
In February 2016, FASB issued ASU No. 2016-02, Leases (Topic 842), (“ASU 2016-02”) which amended the guidance on accounting for leases. The FASB issued this update to increase transparency and comparability among organizations. This update requires the recognition of lease assets and lease liabilities on the balance sheet and the disclosure of key information about leasing arrangements. The Company adopted ASU 2016-02 effective January 1, 2019 using the additional (optional) approach, in accordance with ASU 2018-11 Leases (Topic 842): Targeted Improvements. The Company initially recorded a right of use asset and lease liability of $4.3 million, net of the $0.9 million rent credit, and $5.2 million, in Right of use asset, Other current liabilities and Other liabilities for the non-current portion, respectively. There was no effect on opening retained earnings, and the Company continues to account for leases in the prior period financial statements under ASC Topic 840.
In adopting the new lease standard, the Company elected the permitted package of practical expedients permitted, which allowed the Company to account for existing leases under their current classification, as well as omit any new costs classified as initial direct costs, under the new standard. See Note 7 for additional information on leases.
In February 2018, the FASB issued ASU No. 2018-02, Income Statement - Reporting Comprehensive Income (Topic 220) (“ASU 2018-02”), to address certain income tax effects in Accumulated Other Comprehensive Income (“AOCI”) resulting from the tax reform enacted in 2017. The amended guidance provides an option to reclassify tax effects within AOCI to retained earnings in the period in which the effect of the tax reform is recorded. The amendments were effective for fiscal years beginning after December 15, 2018, including interim periods. The Company has adopted ASU 2018-02 as of January 1, 2019, which did not have any impact on the Company's results of operations or financial condition as there were no balances in AOCI that are tax effected.
In June 2018, the FASB issued ASU 2018-07, “Compensation-Stock Compensation (Topic 718): Improvements to Non-employee Share-Based Payment Accounting” (“ASU 2018-07”), which simplifies the accounting for share-based payments to non-employees by aligning it with the accounting for share-based payments to employees, with certain exceptions. Under the new guidance, the measurement of equity-classified nonemployee awards will be fixed at the grant date. ASU 2018-07 is effective for interim and annual reporting periods beginning after December 15, 2018 and early adoption was permitted. The Company adopted the new standard on January 1, 2019. The adoption of ASU 2018-07 did not have a material impact on the Company’s consolidated financial statements and related disclosures.
Recently Issued Accounting Pronouncements Not Yet Adopted
In June 2016, the FASB issued ASU 2016-13, “Financial Instruments - Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments,” that introduces a new model for recognizing credit losses on financial instruments based on an estimate of current expected credit losses. This includes accounts receivable, trade receivables, loans, held-to-maturity debt securities, net investments in leases and certain off-balance sheet credit exposures. The guidance also modifies the impairment model for available-for-sale debt securities. This ASU is effective for the Company and all public filers which do not qualify as smaller reporting companies for fiscal years beginning after December 15, 2019. The Company does not expect adoption to materially affect the consolidated financial statements.
F- 22
All other ASUs issued and not yet effective for the twelve months ended December 31, 2019, and through the date of this report, were assessed and determined to be either not applicable or are expected to have minimal impact on the Company’s current or future financial position or results of operations.
4. | Stability Biologics, LLC |
On January 13, 2016, the Company completed the acquisition of Stability Inc., a provider of human tissue products to surgeons, facilities, and distributors serving the surgical, spine, and orthopedic sectors of the healthcare industry. As a result of this transaction, the Company acquired all of the outstanding shares of Stability, Inc. in exchange for $6.0 million cash, $3.3 million (or 441,009 shares) of the Company’s common stock, par value $0.001 per share (“Common Stock”), and assumed debt of $1.8 million. Additional one-time costs incurred in connection with the transaction totaled $1.1 million and were included within selling, general and administrative expenses on the consolidated statements of operations. Contingent consideration might have been payable based on a formula determined by sales less certain expenses for the years 2016 and 2017. The contingent consideration was valued at $17.5 million as of January 13, 2016 and is shown in the schedule below as fair value of earn-out. The contingent consideration was classified as a liability.
On September 30, 2017, the Company completed its divestiture of Stability pursuant to the Membership Interest Purchase Agreement by and among the Company, Stability, each person that, as of January 13, 2016, was a stockholder of Stability Inc., a Florida corporation and a predecessor-in-interest to Stability, and Brian Martin, as stockholder representative.
A summary of the assets divested and consideration received follows (in thousands):
Year ended | ||||
December 31, 2017 | ||||
Assets divested | ||||
Trade receivables | $ | |||
Inventories | ||||
Prepaid expenses and other assets | ||||
Goodwill (a) | ||||
Intangible assets | ||||
Property and equipment, net | ||||
Total assets divested | ||||
Liabilities divested | ||||
Accounts payable and accrued liabilities | ||||
Total liabilities divested | ||||
Total net assets divested | $ | |||
Transaction costs | ||||
Consideration received | ||||
Non-trade receivable (b) | ||||
Note receivable (c) | ||||
Intangible assets (d) | ||||
Extinguishment of earn out liability (e) | ||||
Total consideration received | $ | |||
Loss on sale | $ | ( | ) | |
F- 23
(a) In accordance with ASC 350-20-35-52 when a portion of a reporting unit is disposed of, goodwill associated with that business shall be included in the carrying amount of the business in determining the gain on disposal. In accordance with ASC 350-20-35-53, the amount of goodwill to be included in that carrying amount shall be based on the relative fair values of the business to be disposed of and the portion of the reporting unit that will be retained. Based on an estimated fair value of Stability of $16.2 million representing a consideration received for the business compared to the fair value of business retained determined based on the market approach, approximately $0.2 million of the total goodwill of $20.2 million residing in the reporting unit was included in the carrying amount of the business sold.
(b) non-trade receivable represents a cash payment due within 60 days of closing.
(c) a promissory note issued by Stability in the principal amount of $3.5 million in favor of the Company recognized at a discounted value of $3.2 million.
(d) a fair value of $0.5 million for the distributor agreements with Stability and a fair value of $0.1 million for the non-compete agreements with the former stockholders of Stability Inc.
(e) a waiver by the former stockholders of Stability Inc. of all claims and rights to earn-out consideration, which was recorded as a liability at a fair value of $12.2 million immediately prior to the divestiture. The fair value of the earn-out liability was determined based on the income approach and includes the actual realized results of operations and expected future performance over the remaining earn-out period.
The total loss on the Stability Divestiture of $0.5 million is comprised of a pretax book loss of $1.0 million and an associated tax benefit of $0.5 million.
The earn-out arrangement was classified as a liability on the Stability acquisition date of January 13, 2016 and remeasured at fair value each reporting period until the Stability was divested on September 30, 2017. A decrease in fair value of $3.6 million for the year ended December 31, 2017 was included in Selling, general and administration expenses on the consolidated statements of operations.
5. Inventory
Inventory consists of the following (in thousands):
December 31, | |||||||
2019 | 2018 | ||||||
Raw materials | $ | $ | |||||
Work in process | |||||||
Finished goods | |||||||
Inventory, gross | |||||||
Reserve for obsolescence | ( | ) | ( | ) | |||
Inventory, net | $ | $ | |||||
6. Property and Equipment
Property and equipment consist of the following (in thousands):
December 31, | |||||||
2019 | 2018 | ||||||
Leasehold improvements | $ | $ | |||||
Laboratory and clean room equipment | |||||||
Furniture and equipment | |||||||
Construction in progress | |||||||
Property and equipment, gross | |||||||
Less accumulated depreciation and amortization | ( | ) | ( | ) | |||
Property and equipment, net | $ | $ | |||||
Depreciation expense for each of the years ended December 31, 2019, 2018, and 2017 was recorded in certain captions of the consolidated statements of operations for those periods in the amounts shown in the table below (in thousands):
F- 24
For the year ended December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Cost of sales | $ | $ | $ | ||||||||
Selling, general and administrative expenses | |||||||||||
Research and development expenses | |||||||||||
Total | $ | $ | $ | ||||||||
7. | Leases |
As discussed in Note 3, on January 1, 2019, the Company adopted new guidance for the accounting and reporting of leases. The Company has operating leases primarily for corporate offices, vehicles, and certain equipment. Such leases do not require any contingent rental payments, impose any financial restrictions, or contain any residual value guarantees. The Company determines if an arrangement is or contains a lease at inception.
Under ASC 842 transition guidance, the Company has not elected the hindsight practical expedient to determine the lease term for existing leases, which permits companies to consider available information prior to the effective date of the new guidance as to the actual or likely exercise of options to extend or terminate the lease. Certain of the Company’s leases include renewal options and escalation clauses; renewal options have not been included in the calculation of the lease liabilities and right of use assets as the Company is not reasonably certain to exercise the options.
Lease expense for operating lease payments is recognized on a straight-line basis over the term of the lease. Operating lease assets and liabilities are recognized based on the present value of lease payments over the lease term. Since most of the Company’s leases do not have a readily determinable implicit discount rate, the Company uses its incremental borrowing rate to calculate the present value of lease payments determined using the rate of interest that the Company would have to pay on collaterialized or secured borrowing over a similar term. As a practical expedient, the Company has made an accounting policy election not to separate lease components from non-lease components in the event that the agreement contains both. The Company includes both the lease and non-lease components for purposes of calculating the right-of-use asset and related lease liability.
The Company does not act as a lessor or have any leases classified as financing leases.
Operating lease cost was $1.5 million for the year ended December 31, 2019 and was recorded in Selling, general, and administrative expenses. Interest on lease obligations was $0.5 million for the year ended December 31, 2019 and was recorded in Selling, general, and administrative expenses. Cash paid for amounts included in the measurement of operating lease liabilities was $1.7 million at December 31, 2019. The amortization of leased assets for the year ended December 31, 2019 was $0.9 million.
Supplemental balance sheet information related to operating leases is as follows (amounts in thousands, except lease term and discount rate):
December 31, 2019 | ||||
Assets | ||||
Right of use asset | $ | |||
Liabilities | ||||
Short term lease liability | $ | |||
Long term lease liability | $ | |||
Weighted-average remaining lease term (years) | ||||
Weighted-average discount rate | % | |||
Maturities of operating leases liabilities are as follows (amounts in thousands):
F- 25
Year ending December 31, | Maturities | |||
2020 | $ | |||
2021 | ||||
2022 | ||||
2023 | ||||
2024 | ||||
Thereafter | ||||
Total lease payments | ||||
Less: imputed interest | ( | ) | ||
$ | ||||
Future minimum lease payments under operating leases at December 31, 2018 and thereafter were as follows (amounts in thousands):
Year ending December 31, | ||||
2019 | $ | |||
2020 | ||||
2021 | ||||
2022 | ||||
2023 | ||||
Thereafter | ||||
Total lease payments | $ | |||
8. Goodwill and Intangible Assets
Intangible assets are summarized as follows (in thousands):
December 31, 2019 | December 31, 2018 | |||||||||||||||||||
Gross Carrying Amount | Accumulated Amortization | Net Carrying Amount | Gross Carrying Amount | Accumulated Amortization | Net Carrying Amount | |||||||||||||||
Amortized intangible assets | ||||||||||||||||||||
Licenses | $ | $ | ( | ) | $ | $ | $ | ( | ) | $ | ||||||||||
Patents and know how | ( | ) | ( | ) | ||||||||||||||||
Customer and supplier relationships | ( | ) | ( | ) | ||||||||||||||||
Non-compete agreements | ( | ) | ( | ) | ||||||||||||||||
Total amortized intangible assets | $ | $ | ( | ) | $ | $ | $ | ( | ) | $ | ||||||||||
Unamortized intangible assets | ||||||||||||||||||||
Trade names and trademarks | $ | $ | $ | $ | ||||||||||||||||
Patents in process | ||||||||||||||||||||
Total intangible assets | $ | $ | $ | $ | ||||||||||||||||
F- 26
Amortization expense for the years ended December 31, 2019, 2018, and 2017, was $1.0 million. $1.0 million, and $1.7 million, respectively. Patents and patents in process related write-downs due to abandonment were $1.3 million, $0.0 million, and $0.6 million during the years ended December 31, 2019, 2018, and 2017, respectively and are recorded in Selling, general and administrative expenses.
Expected future amortization of intangible assets as of December 31, 2019, is as follows (in thousands):
Estimated | |||
Amortization | |||
Year ending December 31, | Expense | ||
2020 | $ | ||
2021 | |||
2022 | |||
2023 | |||
2024 | |||
Thereafter | |||
$ | |||
Goodwill is evaluated for impairment on an annual basis on September 30 and in interim periods when events or changes indicate the carrying value may not be recoverable. The Company operates under one reporting unit.
For the year ended December 31, 2019, the Company elected to perform a qualitative analysis to determine whether it was more likely than not that the fair value of its reporting unit was less than their carrying value. As a result of this assessment, the Company determined that it was not necessary to perform a quantitative impairment test and concluded that goodwill was not impaired at December 31, 2019. For the year ended December 31, 2018, the Company performed a quantitative analysis to determine if there was any impairment. As a result of this assessment, the Company determined that there was no impairment for the year ended December 31, 2018.
The following represents the changes in the carrying amount of goodwill for 2019 and 2018 (in thousands):
Goodwill | |||
Balance as of January 1, 2018 | $ | ||
Activity | |||
Balance as of December 31, 2018 | |||
Activity | |||
Balance as of December 31, 2019 | $ | ||
9. | Accrued Expenses |
Accrued expenses consist of the following (in thousands):
December 31, | |||||||
2019 | 2018 | ||||||
Legal costs | $ | $ | |||||
Settlement costs | |||||||
Pricing adjustment settlement with Veterans Affairs | |||||||
Estimated returns | |||||||
External commissions | |||||||
Accrued clinical trials | |||||||
Other | |||||||
Total | $ | $ | |||||
F- 27
10. | Long Term Debt |
Credit Facility
On October 12, 2015, the Company and its subsidiaries entered into a Credit Agreement (the “Credit Agreement”) with certain lenders and Bank of America, N.A., as administrative agent. The Credit Agreement established a senior secured revolving credit facility in favor of the Company with a maturity date of October 12, 2018 and an aggregate lender commitment of up to $50 million. In September 2017, the expiration date of the Credit Agreement was extended to October 12, 2019. The Credit Agreement also provided for an uncommitted incremental facility of up to $35 million, which could be exercised as one or more revolving commitment increases or new term loans, all subject to certain customary terms and conditions set forth in the Credit Agreement. The obligations of the Company under the Credit Agreement were guaranteed by the Company’s subsidiaries. The obligations of the loan parties under the Credit Agreement and the other credit documents were secured by liens on and security interests in substantially all of the assets of each of the loan parties and a pledge of the equity interests of each subsidiary owned by a loan party, subject to certain customary exclusions. Borrowings under the facility had an interest at LIBOR plus 1.5 % to 2.25 %. Fees paid in connection with the initiation of the credit facility totaled approximately $0.5 million. These deferred financing costs were being amortized to interest expense over the three-year life of the facility. The Credit Agreement contained customary representations, warranties, covenants, and events of default, including restrictions on certain payments of dividends by the Company.
On August 31, 2018, the lending parties’ terminated their commitments to make loans and issue letters of credit under the Credit Agreement due to the Company’s failure to timely file its periodic reports with the SEC. Accordingly, since then, the Company has not had the ability to borrow under the Credit Agreement. There were no outstanding borrowings or letters of credit issued under the Credit Agreement at the time of termination, and the Company never drew down any amounts under the credit facility during the entire term of the Credit Agreement. No termination penalties were paid as a result of the termination.
BT Term Loan
On June 10, 2019, the Company entered into a loan agreement (the “BT Loan Agreement”) with Blue Torch Finance LLC (“Blue Torch”), as administrative agent and collateral agent, to borrow funds with a face value of $75.0 million (the “BT Term Loan”), of which the full amount was borrowed and funded. The proceeds from the BT Term Loan were used (i) for working capital and general corporate purposes and (ii) to pay transaction fees, costs and expenses incurred in connection with the BT Term Loan and the related transactions. The BT Loan Agreement provided that the BT Term Loan would mature on June 20, 2022 and was repayable in quarterly installments of $0.9 million, with the balance due on June 20, 2022. Blue Torch maintained a first-priority security interest in substantially all the Company’s assets. The BT Term Loan was issued net of the original issue discount of $2.3 million. The Company also incurred $6.7 million of deferred financing costs. The BT Term Loan was amended on April 22, 2020 and was repaid on July 2, 2020, each of which is addressed in Note 21, “Subsequent Events,” of the consolidated financial statements.
The interest rate applicable to any borrowings under the BT Term Loan accrued at a rate equal to LIBOR plus a margin of 8.00 % per annum. The BT Term Loan had an interest rate equal to 10.46 % at the time the BT Loan Agreement was executed. The interest as of December 31, 2019 was 10.11 %.
The BT Loan Agreement originally contained financial covenants requiring the Company, on a consolidated basis, to maintain the following:
• | Maximum Total Leverage Ratio, defined as funded debt divided by consolidated adjusted EBITDA, of not more than |
• | Minimum Liquidity, defined as unrestricted cash and cash equivalents, of less than $ |
The BT Loan Agreement also provided that any prepayment of the loan, voluntary or mandatory, as defined in the BT Loan Agreement, would subject MiMedx to a prepayment penalty as of the date of the prepayment with respect to the Term Loan of:
• | During the period from June 10, 2019 through June 10, 2020, an amount equal to |
• | During the period from June 11, 2020 through June 10, 2021, an amount equal to |
F- 28
Principal prepayments after June 10, 2021 were not subject to a prepayment penalty.
The BT Loan Agreement also included events of default customary for facilities of this type, and the BT Loan Agreement provided that upon the occurrence of such events of default, subject to customary cure rights, all outstanding loans under the BT Loan Agreement may be accelerated and/or the lenders’ commitments terminated.
The balances of the BT Term Loan as of December 31, 2019 was as follows (amounts in thousands):
December 31, 2019 | |||||||
Current portion | Long-term | ||||||
Liability component - principal | $ | $ | |||||
Original issue discount | ( | ) | |||||
Deferred financing cost | ( | ) | |||||
Liability component - net carrying value | $ | $ | |||||
Interest expense related to the BT Term Loan, included in Interest income (expense), net in the consolidated statements of operations, was as follows (amounts in thousands):
For the Year Ended | |||
December 31, 2019 | |||
Interest expense - stated interest rate | $ | ||
Interest expense - amortization of original issue discount | |||
Interest expense - amortization of deferred financing costs | |||
Total term loan interest expense | $ | ||
The future principal payments for the Company’s BT Term Loan as of December 31, 2019 were as follows (in thousands):
Year ending December 31, | Principal | ||
2020 | $ | ||
2021 | |||
2022 | |||
2023 | |||
2024 | |||
Thereafter | |||
Total Long Term Debt | $ | ||
As of December 31, 2019, the fair value of the Company’s BT Term Loan was $70.6 million. This valuation was calculated based on a series of Level 2 and Level 3 inputs by calculating a discount rate based on the credit risk spread of debt instruments of a similar risk character in reference to U.S. Treasury instruments with identical securities, with an incremental risk premium for Company-specific risk factors. The remaining cash flows associated with the BT Term Loan were discounted to December 31, 2019 with this calculated discount rate to derive the fair value as of that date.
As described below in Note 21, “Subsequent Events,” on July 2, 2020, a portion of the proceeds from the Preferred Stock Transaction (as defined below) and the Hayfin Loan Transaction (as defined below) was used to repay the outstanding balance of principal, accrued but unpaid interest, and prepayment premium under the BT Loan Agreement. In connection with the repayment of the BT Term Loan, the Company terminated the BT Loan Agreement.
11. Net (Loss) Income Per Share
Basic net (loss) income per common share is computed using the weighted-average number of common shares outstanding during the period. Diluted net income per common share is computed using the weighted-average number of common and dilutive common equivalent shares from stock options and restricted stock using the treasury stock method.
F- 29
The following table sets forth the computation of basic and diluted net income per share (in thousands except for share and per share data):
Year Ended December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Net (loss) income | $ | ( | ) | $ | ( | ) | $ | ||||
Denominator for basic earnings (loss) per share - weighted average shares | |||||||||||
Effect of dilutive securities: Stock options and restricted stock (a) | |||||||||||
Denominator for diluted (loss) earnings per share - weighted average shares adjusted for dilutive securities | |||||||||||
(Loss) income per common share - basic | $ | ( | ) | $ | ( | ) | $ | ||||
(Loss) income per common share - diluted | $ | ( | ) | $ | ( | ) | $ | ||||
(a)Securities that are included in the computation of the denominator above, utilizing the treasury stock method for the years ended December 31, 2019, 2018 and 2017 are as follows:
Effect of dilutive securities: | 2019 | 2018 | 2017 | |||||
Stock options | ||||||||
Restricted stock awards | ||||||||
12. Equity
Stock Incentive Plans
The Company has two share-based compensation plans which provide for the granting of equity awards, including qualified incentive and non-qualified stock options, stock appreciation awards and restricted Common Stock awards: the MiMedx Group, Inc. 2016 Equity and Cash Incentive Plan (the “2016 Plan”), which was approved by shareholders on May 18, 2016 and the MiMedx Group, Inc. Assumed 2006 Stock Incentive Plan (the “Prior Incentive Plan”). During the years ended December 31, 2019, 2018 and 2017 the Company used only the 2016 Plan to make grants.
The 2016 Plan permits the grant of equity awards to the Company’s employees, directors, consultants and advisors for up to 5,000,000 shares of the Company’s Common Stock plus (i) the number of shares of the Company’s Common Stock that remain available for issuance under the Prior Incentive Plan, and (ii) the number of shares that are represented by outstanding awards that later become available because of the expiration or forfeiture of the award without the issuance of the underlying shares. The awards are subject to a vesting schedule as set forth in each individual agreement. Option awards are generally granted with an exercise price equal to the market price of the Company’s stock at the date of grant, and those option awards generally vest based on three years of continuous service and have 10 -year contractual terms. Restricted Common Stock awards generally vest over three years . Certain option and restricted stock awards provide for accelerated vesting if there is a change in control and upon death or disability.
F- 30
A summary of stock option activity as of December 31, 2019, and changes during the year then ended are presented below:
Number of Shares | Weighted- Average Exercise Price | Weighted- Average Remaining Contractual Term (in years) | Aggregate Intrinsic Value | |||||||||
Outstanding at January 1, 2019 | $ | |||||||||||
Granted | ||||||||||||
Exercised | ( | ) | ||||||||||
Unvested options forfeited | ||||||||||||
Vested options expired | ( | ) | ||||||||||
Outstanding at December 31, 2019 | $ | |||||||||||
Exercisable at December 31, 2019 | $ | $ | ||||||||||
The fair value of options vested during the years ended December 31, 2019, 2018 and 2017 were $1.4 million, $0.1 million, and $3.7 million, respectively. There were no options granted during the years ended December 31, 2019, 2018 and 2017 and no unrecognized compensation expense at December 31, 2019.
During the year ended December 31, 2019, the Company extended the contractual life of 612,000 fully vested share options held by 7 members of the Board and 278,916 fully vested share options held by a former employee. As a result of that modification, the Company recognized incremental share-based compensation expense of $0.4 million for the year ended December 31, 2019.
The incremental fair value of the modified options in 2019 was estimated on the modification date using the Black-Scholes-Merton option-pricing model that uses assumptions for expected volatility, expected dividends, expected term, and the risk-free interest rate. Expected volatilities were the blend of the Company’s historical stock price volatility as well as that of market comparable publicly traded peer companies and other factors estimated over the expected term of the options. The term of the modified options was the remaining time until the end of the contractual maturity of ten years . The risk-free rate was based on the U.S. Treasury yield curve in effect at the time of modification for the period of the expected term.
2019 Option Modification | |
Expected volatility | 65% - 95% |
Expected life (in years) | 0.28 - 5.12 |
Expected dividend yield | |
Risk-free interest rate | 1.56% - 2.02% |
Restricted Stock Awards
Following is summary information for restricted stock awards for the year ended December 31, 2019. Shares vest over a one to three year period in equal annual increments and require continuous service.
F- 31
As of December 31, 2019, there was approximately $11.4 million of total unrecognized stock-based compensation related to non-vested restricted stock. That expense is expected to be recognized over a weighted-average period of 1.8 years, which approximates the remaining vesting period of these grants. All shares noted below as unvested are considered issued and outstanding at December 31, 2019.
Number of Shares | Weighted-Average Grant Date Fair Value | ||||||
Unvested at January 1, 2019 | $ | ||||||
Granted | |||||||
Vested | ( | ) | |||||
Forfeited | ( | ) | |||||
Unvested at December 31, 2019 | $ | ||||||
The total fair value of restricted stock awards vested during the years ended December 31, 2019, 2018, and 2017, was $5.2 million, $17.9 million, and $17.3 million, respectively.
During the year ended December 31, 2019, the Company granted a fixed dollar value restricted share unit award to the members of its Board in the amount of $1.6 million. The restricted share unit awards vest upon the earlier of one year or the date of the 2019 Annual Meeting and will be settled in Common Stock with the number of shares of Common Stock to be determined based on the Company’s closing share price on the future settlement date. During the year ended December 31, 2019, the Company recognized $0.4 million of share-based compensation expense, with an offsetting liability recorded in Accrued compensation in the consolidated balance sheets.
For the years ended December 31, 2019, 2018, and 2017 the Company recognized share-based compensation as follows (in thousands):
Years Ended December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Cost of sales | $ | $ | $ | ||||||||
Research and development | |||||||||||
Selling, general and administrative | |||||||||||
Total share-based compensation | $ | $ | $ | ||||||||
Income tax benefit | ( | ) | ( | ) | ( | ) | |||||
Total share-based compensation, net of tax benefit | $ | $ | $ | ||||||||
Treasury Stock
On May 8, 2014, the Board authorized the repurchase of up to $10 million of shares of Common Stock from time to time through December 31, 2014. The Board increased the authorization during the year ended December 31, 2015 to $60 million, during the year ended December 31, 2016 to $66 million, and during the year ended December 31, 2017 to $130 million. In January 2018 the Board announced that it had increased the total authorization to $140 million. The share repurchase program subsequently expired during the year ended December 31, 2018.
For the years ended December 31, 2018 and 2017, the Company purchased 507,600 , and 5,635,077 shares of its Common Stock, respectively, for an aggregate purchase price of approximately $7.6 million, and $68.3 million, respectively, exclusive of commissions of approximately $0.0 million, and $0.2 million, respectively.
F- 32
13. Income Taxes
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes.
Significant components of the Company’s deferred tax assets and liabilities are as follows (in thousands):
December 31, | ||||||||
2019 | 2018 | |||||||
Deferred Tax Assets: | ||||||||
Accrued expenses | $ | $ | ||||||
Deferred revenue | ||||||||
Bad debts | ||||||||
Sales return and allowances | ||||||||
Accrued settlement costs | ||||||||
Research and development and other tax credits | ||||||||
Net operating loss | ||||||||
Share-based compensation | ||||||||
Lease obligation | ||||||||
Other | ||||||||
Deferred Tax Liabilities: | ||||||||
Prepaid expenses | ( | ) | ( | ) | ||||
Right of use asset | ( | ) | ||||||
Property and equipment | ( | ) | ( | ) | ||||
Unearned insurance refund | ( | ) | ||||||
Deferred costs of goods sold | ( | ) | ||||||
Intangible assets | ( | ) | ( | ) | ||||
Net Deferred Tax Assets | ||||||||
Less: Valuation allowance | ( | ) | ( | ) | ||||
Net Deferred Tax Assets after Valuation Allowance | $ | $ | ||||||
F- 33
The reconciliation of the federal statutory income tax rate of 21 % for 2019 and 2018, and 35 % for 2017 to the effective rate is as follows:
December 31, | ||||||||
2019 | 2018 | 2017 | ||||||
Federal statutory rate | % | % | % | |||||
State taxes, net of federal benefit | ( | )% | % | % | ||||
Nondeductible compensation | ( | )% | ( | )% | % | |||
Meals and entertainment | ( | )% | ( | )% | % | |||
Keyman life insurance | ( | )% | ( | )% | % | |||
Inventory contribution deduction | % | % | ( | )% | ||||
Domestic production activities deduction | % | % | ( | )% | ||||
Fair value adjustment | % | % | ( | )% | ||||
Share-based compensation | ( | )% | % | ( | )% | |||
Tax credits | % | % | ( | )% | ||||
Uncertain tax position | % | ( | )% | % | ||||
Write-off of net operating losses | % | ( | )% | % | ||||
Payable true-up | % | ( | )% | % | ||||
Sale of Stability | % | % | ( | )% | ||||
Fixed asset true-up | % | % | % | |||||
Federal provision to return | % | % | % | |||||
Impact of federal rate change | % | % | % | |||||
Other | ( | )% | ( | )% | ( | )% | ||
Valuation allowance | ( | )% | ( | )% | ( | )% | ||
% | ( | )% | ( | )% | ||||
Share-based Compensation had a significant impact on the Company's effective tax rate as of December 31, 2019. Additionally, state taxes, Meals and Entertainment, and Nondeductible Compensation had a significant impact on the Company's effective tax rate.
Meals and Entertainment had a significant impact on the Company's effective tax rate as of December 31, 2018 due to the impact of the Act on the Company's method of calculating this permanent adjustment. Additionally, Federal and state tax credits, mostly related to the Company's Research and Development activities, had a significant impact on the Company's effective rate.
Stock based compensation had a significant impact on the Company’s effective tax rate as of December 31, 2017 due to the Company’s adoption of ASU 2016-09. Additionally, on September 30, 2017, the Company completed the Stability Divestiture, which resulted in a significant reduction in the Company’s effective tax rate. See Note 4 for details regarding the transaction.
F- 34
Current and deferred income tax expense (benefit) is as follows (in thousands):
December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Current: | |||||||||||
Federal | $ | ( | ) | $ | $ | ||||||
State | |||||||||||
Total current | ( | ) | |||||||||
Deferred: | |||||||||||
Federal | ( | ) | |||||||||
State | ( | ) | |||||||||
Total deferred | ( | ) | |||||||||
Total expense (benefit) | $ | ( | ) | $ | $ | ( | ) | ||||
A valuation allowance of $30.6 million and $27.3 million was recorded against the deferred tax asset balance as of December 31, 2019 and December 31, 2018, respectively. To the extent the Company determines that, based on the weight of available evidence, all or a portion of its valuation allowance is no longer necessary, the Company will recognize an income tax benefit in the period such determination is made for the reversal of the valuation allowance. If management determines that, based on the weight of available evidence, it is more-likely-than-not that all or a portion of the net deferred tax assets will not be realized, the Company may recognize income tax expense in the period such determination is made to increase the valuation allowance.
At December 31, 2019 and 2018, the Company had income tax net operating loss (“NOL”) carryforwards for federal and state purposes of $56.8 million and $49.3 million and $11.4 million and $15.6 million, respectively. A portion of the Company’s NOLs and tax credits are subject to annual limitations due to ownership change limitations provided by Internal Revenue Code Section 382. If not utilized, the federal and state tax NOL carryforwards will expire between 2027 and 2037. As of December 31, 2019, the Company has recorded a deferred tax asset for both federal and state NOL carryforwards of approximately $11.9 million and $3.1 million, respectively. As of December 31, 2018, the Company has recorded a deferred tax asset for federal and state NOL carryforwards of $2.4 million and approximately $0.9 million, respectively.
The following is a tabular reconciliation of the total amounts of unrecognized tax benefits (in thousands) included in other liabilities in the consolidated balance sheets:
2019 | 2018 | 2017 | |||||||||
Unrecognized tax benefits - January 1 | $ | $ | $ | ||||||||
Gross increases - tax positions in current period | |||||||||||
Gross increases - tax positions in prior period | |||||||||||
Gross decreases - lapse of statute of limitations | ( | ) | |||||||||
Unrecognized tax benefits - December 31 | $ | $ | $ | ||||||||
F- 35
The Company recognizes accrued interest related to unrecognized tax benefits and penalties as income tax expense. Related to the unrecognized tax benefits noted above, the Company accrued $0.1 million of interest during 2019, and, in total, as of December 31, 2019 has recognized $0.1 million of interest. The Company accrued $0.1 million of interest during 2018, and, in total, as of December 31, 2018 had recognized $0.1 million of interest. The Company accrued $0.1 million of interest during 2017, and, in total, as of December 31, 2017 had recognized $0.1 million of interest.
Certain positions included in the tabular reconciliation above will be reduced as a result of the expiration of statutes of limitations within the next 12 months. The reserve would be reduced by approximately $0.2 million.
14. Supplemental Disclosure of Cash Flow and Non-Cash Investing and Financing Activities
Selected cash payments, receipts, and noncash activities are as follows (in thousands):
Years Ended December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Cash paid for interest | $ | $ | $ | ||||||||
Income taxes paid | |||||||||||
Non-cash activities: | |||||||||||
Purchases of equipment included in accounts payable | |||||||||||
Deferred financing costs | |||||||||||
Stock issuance in exchange for services performed | |||||||||||
15. 401(k) Plan
The Company has a 401(k) plan (the “401(k) Plan”) covering all employees who have completed one month of service. Under the 401(k) Plan, participants could defer up to 90 % of their eligible wages to a maximum of $19,000 per year (annual limit for 2019). Employees age 50 or over in 2018 could make additional pre-tax contributions up to $6,000 . Annually, the Company could elect to match employee contributions up to 5 % of the employee’s eligible compensation. Additionally, the Company could elect to make a discretionary contribution to the 401(k) Plan. The Company did not provide matching contributions for the year ended December 31, 2017. The matching contribution for the year ended December 31, 2019 and 2018 was $1.5 million and $1.9 million, respectively.
16. Commitments and Contingencies
Contractual Commitments
In addition to the leases noted under Note 7 “Leases,” the Company has commitments for meeting space. These leases expire over 3 to 3.5 years following December 31, 2019, and generally contain renewal options. The Company anticipates that most of these leases will be renewed or replaced upon expiration.
F- 36
The estimated annual lease payment and meeting space commitments are as follows (in thousands):
Years Ended December 31, | |||
2020 | $ | ||
2021 | |||
2022 | |||
2023 | |||
2024 | |||
Thereafter | |||
$ | |||
Rent expense for the years ended December 31, 2019, 2018 and 2017, was approximately $1.4 million, $1.5 million, and $1.6 million, respectively, and is allocated among cost of sales, research and development, and selling, general and administrative expenses.
Letters of Credit
Previously, as a condition of the leases for the Company’s facilities, the Company was obligated under standby letters of credit in the amount of approximately $0.1 million. The Company amended its lease during 2018 to eliminate this obligation.
Separation and Transition Services Agreement of Edward J. Borkowski
On November 18, 2019, the Company entered into a Separation and Transition Services Agreement (“Separation Agreement”) with Edward J. Borkowski, under which Mr. Borkowski resigned as Executive Vice President and Interim Chief Financial Officer of the Company, as well as from any and all officer, director or other positions that he held with the Company and its affiliates, effective November 15, 2019. Pursuant to the Separation Agreement, Mr. Borkowski agreed to perform the duties of the Interim Chief Financial Officer with respect to the Company’s 2018 Form 10-K and assist with the transition of his duties as described in the Separation Agreement from November 15, 2019 through the earlier of the first business day following the Company’s filing of its 2018 Form 10-K with the SEC or December 31, 2019 (the “Transition Period”). From the end of the Transition Period until March 31, 2020, Mr. Borkowski agreed to provide services as may be requested by the Company with respect to matters related to the 2018 Form 10-K and the Company’s Annual Report on Form 10-K for the year ended December 31, 2019. The Company has paid Mr. Borkowski the full amount of $4.0 million as of the date of the issuance of these consolidated financial statements payable under the Separation Agreement.
Litigation and Regulatory Matters
In the ordinary course of business, the Company and its subsidiaries are parties to numerous civil claims and lawsuits and subject to regulatory examinations, investigations, and requests for information. Some of these matters involve claims for substantial amounts. The Company’s experience has shown that the damages alleged by plaintiffs or claimants are often overstated, based on unsubstantiated legal theories, unsupported by facts, and/or bear no relation to the ultimate award that a court might grant. Additionally, the outcome of litigation and regulatory matters and the timing of ultimate resolution are inherently difficult to predict. These factors make it difficult for the Company to provide a meaningful estimate of the range of reasonably possible outcomes of claims in the aggregate or by individual claim. However, on a case-by-case basis, reserves are established for those legal claims in which it is probable that a loss will be incurred and the amount of such loss can be reasonably estimated. The Company's financial statements at December 31, 2019 reflect the Company's current best estimate of probable losses associated with these matters, including costs to comply with various settlement agreements, where applicable. The actual costs of resolving these claims may be substantially higher or lower than the amounts reserved.
The following is a description of certain litigation and regulatory matters:
F- 37
Shareholder Derivative Suits
On December 6, 2018, the United States District Court for the Northern District of Georgia entered an order consolidating three shareholder derivative actions (Evans v. Petit, et al. filed September 25, 2018, Georgalas v. Petit, et al. filed September 27, 2018, and Roloson v. Petit, et al. filed October 22, 2018) that had been filed in the Northern District of Georgia. On January 22, 2019, plaintiffs filed a verified consolidated shareholder derivative complaint. The consolidated action sets forth claims of breach of fiduciary duty, corporate waste and unjust enrichment against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Joseph G. Bleser, J. Terry Dewberry, Charles R. Evans, Larry W. Papasan, Luis A. Aguilar, Bruce L. Hack, Charles E. Koob, Neil S. Yeston and Christopher M. Cashman. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Company filed a motion to stay on February 18, 2019, pending the completion of the investigation by the Company’s Special Litigation Committee. The Special Litigation Committee completed its investigation relating to this action and filed an executive summary of its findings with the Court on July 1, 2019. The parties (together with parties from the Hialeah derivative lawsuit, the Nix and Demaio derivative lawsuit, and the Murphy derivative lawsuit, each described below) held a mediation on February 11, 2020. Following continued discussions, on May 1, 2020, the parties notified the Court that plaintiffs and the Company had reached an agreement in principle to settle this consolidated derivative action, which settlement also encompasses all claims asserted in the Hialeah derivative lawsuit, the Nix and Demaio derivative lawsuit, and the Murphy derivative lawsuit. As of the date of the filing of this Form 10-K, the parties are drafting, and intend to file, a stipulation of settlement and motion seeking preliminary approval of the settlement.
On October 29, 2018, the City of Hialeah Employees Retirement System (“Hialeah”) filed a shareholder derivative complaint in the Circuit Court for the Second Judicial Circuit in and for Leon County, Florida (the “Florida Court”). The complaint alleges claims for breaches of fiduciary duty and unjust enrichment against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Joseph G. Bleser, J. Terry Dewberry, Charles R. Evans, Bruce L. Hack, Charles E. Koob, Larry W. Papasan, and Neil S. Yeston. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Company moved to stay the action on February 7, 2019, to allow the prior-filed consolidated derivative action in the Northern District of Georgia to be resolved first and to allow the Company’s Special Litigation Committee time to complete its investigation. The Company also filed a motion to dismiss on April 8, 2019. As discussed above, the plaintiff participated in the mediation that took place in connection with the prior-filed consolidated derivative action in the Northern District of Georgia and is a party to the agreement in principle to settle that consolidated derivative action. The agreement in principle provides that the plaintiff in this action will file a notice of dismissal to dismiss its action with prejudice within seven calendar days after the date that the judgment entered by the Northern District of Georgia becomes final.
On May 15, 2019, two individuals purporting to be shareholders of the Company filed a shareholder derivative complaint in the Superior Court for Cobb County, Georgia. (Nix and Demaio v. Evans, et al.) The complaint alleges claims for breaches of fiduciary duty, corporate waste and unjust enrichment against certain current and former directors and officers of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Chris Cashman, Lou Roselli, Mark Diaz, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Court ordered this matter stayed pending the resolution of the consolidated derivative suit pending in the Northern District of Georgia. As discussed above, the plaintiff participated in the mediation that took place in connection with the prior-filed consolidated derivative action in the Northern District of Georgia and is a party to the agreement in principle to settle that consolidated derivative action. The agreement in principle provides that the plaintiffs in this action will file a notice of dismissal to dismiss their action with prejudice within seven calendar days after the date that the judgment entered by the Northern District of Georgia becomes final.
On August 12, 2019, John Murphy filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida (Murphy v. Petit, et al.). The complaint alleged claims for breaches of fiduciary duty and unjust enrichment against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Alexandra O. Haden, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. The allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. The Company filed a motion to transfer this action to the Northern District of Georgia. Prior to resolution of that motion, the plaintiff voluntarily dismissed this action without prejudice. As discussed above, the plaintiff participated in the mediation that took place in connection with the prior-filed consolidated derivative action in the Northern District of Georgia and is a party to the agreement in principle to settle that consolidated derivative action. Under the agreement in principle, the plaintiff has agreed that this action shall not be reinstated and, after the judgment entered by the Northern District of Georgia becomes final, this action shall be deemed dismissed with prejudice.
F- 38
On February 10, 2020, Charles Pike filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida (Pike v. Petit, et al.). The complaint alleges claims for breaches of fiduciary duty against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. Similar to the prior-filed actions discussed above, the allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. On May 12, 2020, prior to the Company’s time to respond to the complaint, the plaintiff filed a notice of voluntary dismissal of this action without prejudice.
On February 18, 2020, Bruce Cassamajor filed a shareholder derivative complaint in the United States District Court for the Northern District of Florida (Cassamajor v. Petit, et al.). The complaint alleges claims for breaches of fiduciary duty against certain former officers, and certain current and former directors, of the Company: Parker H. Petit, William C. Taylor, Michael J. Senken, John E. Cranston, Charles R. Evans, Luis A. Aguilar, Joseph G. Bleser, J. Terry Dewberry, Bruce L. Hack, Charles E. Koob, Larry W. Papasan and Neil S. Yeston. Similar to the prior-filed actions discussed above, the allegations generally involve claims that the defendants breached their fiduciary duties by causing or allowing the Company to misrepresent its financial statements as a result of improper revenue recognition. On May 22, 2020, prior to service of the complaint, the plaintiff filed a notice of voluntary dismissal of this action without prejudice. On May 26, 2020, the court ordered this case to be dismissed for failure to serve process.
Securities Class Action
On January 16, 2019, the United States District Court for the Northern District of Georgia entered an order consolidating two purported securities class actions (MacPhee v. MiMedx Group, Inc., et al. filed February 23, 2018 and Kline v. MiMedx Group, Inc., et al. filed February 26, 2018). The order also appointed Carpenters Pension Fund of Illinois as lead plaintiff. On May 1, 2019, the lead plaintiff filed a consolidated amended complaint, naming as defendants the Company, Michael J. Senken, Parker H. Petit, William C. Taylor, Christopher M. Cashman and Cherry Bekaert & Holland LLP. The amended complaint (the “Securities Class Action Complaint”) alleged violations of Section 10(b) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), Rule 10b-5 promulgated thereunder and Section 20(a) of the Exchange Act. It asserted a class period of March 7, 2013 through June 29, 2018. Following the filing of motions to dismiss by the various defendants, the lead plaintiff was granted leave to file an amended complaint. The lead plaintiff filed its amended complaint against the Company, Michael Senken, Pete Petit, William Taylor, and Cherry Bekaert & Holland (Christopher Cashman was dropped as a defendant) on March 30, 2020; defendants filed motions to dismiss on May 29, 2020.
Investigations
SEC Investigation
On April 4, 2017, the Company received a subpoena from the SEC requesting information related to, among other things, the Company’s recognition of revenue, practices with certain distributors and customers, its internal accounting controls and certain employment actions. The Company cooperated with the SEC in its investigation (the “SEC Investigation”). In November 2019, the SEC brought claims against the Company and the Company’s former officers Parker H. Petit, Michael J. Senken, and William C. Taylor. The SEC alleged that from 2013 to 2017, the Company prematurely recognized revenue from sales to its distributors and exaggerated its revenue growth. The SEC’s complaint also alleged that the Company improperly recognized revenue because its former CEO and COO entered into undisclosed side arrangements with certain distributors. These side arrangements allowed distributors to return product to the Company or conditioned distributors’ payment obligations on sales to end users. The SEC complaint further alleged that the Company’s former CEO, COO, and CFO allegedly covered up their scheme for years, including after the Company’s former controller raised concerns about the Company’s accounting for specific distributor transactions. The SEC also alleged that the Company’s former CEO, COO, and CFO all misled the Company’s outside auditors, members of the Company’s Audit Committee, and outside lawyers who inquired about these transactions. The SEC brought claims against the Company and its former CEO, COO, and CFO for violating the antifraud, reporting, books and records, and internal controls provisions of the federal securities laws. The SEC also brought claims against the Company’s former CEO, COO, and CFO for lying to the Company’s outside auditors. In November 2019, without admitting or denying the SEC’s allegations, the Company settled with the SEC by consenting to the entry of a final judgment that permanently restrains and enjoins the Company from violating certain provisions of the federal securities laws. As part of the resolution, the Company paid a civil penalty of $1.5 million. The settlement concluded, as to the Company, the matters alleged by the SEC in its complaint. The SEC’s litigation continues against the Company’s former officers.
F- 39
United States Attorney’s Office for the Southern District of New York (“USAO-SDNY”) Investigation
The USAO-SDNY conducted an investigation into topics similar to those at issue in the SEC Investigation. The USAO-SDNY requested that the Company provide it with copies of all information the Company furnished to the SEC and made additional requests for information. The USAO-SDNY conducted interviews of various individuals, including employees and former employees of the Company. The USAO-SDNY issued indictments in November 2019 against former executives Messrs. Petit and Taylor for securities fraud and conspiracy to commit securities fraud, to make false filings with the SEC, and improperly influence the conduct of audits relating to alleged misconduct that resulted in inflated revenue figures for fiscal 2015. The Company is cooperating with the USAO-SDNY.
Department of Veterans’ Affairs Office of Inspector General (“VA-OIG”) and Civil Division of the Department of Justice (“DOJ-Civil”) Subpoenas and/or Investigations
VA-OIG has issued subpoenas to the Company seeking, among other things, information concerning the Company’s financial relationships with VA clinicians. DOJ-Civil has requested similar information. The Company has cooperated fully and produced responsive information to VA-OIG and DOJ-Civil. Periodically, VA-OIG has requested additional documents and information regarding payments to individual VA clinicians. Most recently, on June 3, 2020, the Company received a subpoena from the VA-OIG requesting information regarding the Company’s financial relationships and interactions with two healthcare providers at the VA Long Beach Healthcare System. The Company has continued to cooperate and respond to these requests.
As part of its cooperation, the Company provided documents in response to subpoenas concerning its relationship with three now former VA employees in South Carolina, who were ultimately indicted in May 2018. Among other things, the indictment referenced speaker fees paid by the Company to the former VA employees and other interactions between now former Company employees and the former VA employees. In January 2019, prosecution was deferred for 18 months to allow the three former VA employees to enter and complete a Pretrial Diversion Program, the completion of which would result in the dismissal of the indictment. As far as the Company is aware, two of the former VA employees have completed the program early and the indictment has been dismissed with respect to them. To date, no actions have been taken against the Company with respect to this matter.
United States Attorney’s Office for the Middle District of North Carolina (“USAO-MDNC”) Investigation
On January 9, 2020, the USAO-MDNC informed the Company that it is investigating the Company’s financial relationships with two former clinicians at the Durham VA Medical Center. The Company is cooperating with the investigation.
Qui Tam Actions
On January 19, 2017, a former employee of the Company filed a qui tam False Claims Act complaint in the United States District Court for the District of South Carolina (United States of America, ex rel. Jon Vitale v. MiMedx Group, Inc.) alleging that the Company’s donations to the patient assistance program, Patient Access Network Foundation, violated the Anti-Kickback Statute and resulted in submission of false claims to the government. The government declined to intervene and the complaint was unsealed on August 10, 2018. The Company filed a motion to dismiss on October 1, 2018. The Company’s motion to dismiss was granted in part and denied in part on May 15, 2019. The case is in discovery.
On January 20, 2017, two former employees of the Company, filed a qui tam False Claims Act complaint in the United States District Court for the District of Minnesota (Kruchoski et. al. v. MiMedx Group, Inc.). An amended complaint was filed on January 27, 2017. The operative complaint alleges that the Company failed to provide truthful, complete and accurate information about the pricing offered to commercial customers in connection with the Company’s FSS contract. On May 7, 2019, the Department of Justice (“DOJ”) declined to intervene, and the case was unsealed. In April 2020, without admitting the allegations, the Company agreed to pay $6.5 million to the DOJ to resolve this matter.
Former Employee Litigation
On December 13, 2016, the Company filed a complaint in the Circuit Court for Palm Beach County, Florida (MiMedx Group, Inc. v. Academy Medical, LLC et. al.) alleging several claims against a former employee, primarily based on his alleged competitive activities while he was employed by the Company (breach of contract, breach of fiduciary duty and breach of duty of loyalty). The former employee countersued for monetary damages and injunctive relief, alleging whistleblower retaliation in violation of the Dodd-Frank Wall Street Reform and Consumer Protection Act (the “Dodd-Frank Act”), unlawful discharge and defamation. The Court dismissed the Dodd-Frank Act whistleblower counterclaim, and in response, the former employee filed an amended complaint on September 11, 2018, adding allegations of post-termination retaliation in violation of the Dodd-Frank Act. The court dismissed the former employee’s retaliation counterclaim on January 24, 2019. After this dismissal, only the former employee’s
F- 40
claims of unlawful discharge and defamation remained pending. The parties resolved this matter and the case was dismissed on September 5, 2019.
On December 29, 2016, the Company filed a complaint in the United States District Court for the Northern District of Illinois (MiMedx Group, Inc. v. Michael Fox) alleging several claims against a former employee of the Company, primarily based on his alleged competitive activities while he was employed by the Company (breach of contract, breach of fiduciary duty and breach of duty of loyalty). The former employee countersued the Company for monetary damages and injunctive relief, alleging improper wage rate adjustment, interference with the former employee’s job after his termination from the Company and retaliation. The parties resolved this matter and the case was dismissed on November 4, 2019.
On July 13, 2018, a former employee filed a complaint against the Company in the United States District Court for the Northern District of Texas (Jennifer R. Scott v. MiMedx Group, Inc.), alleging sex discrimination and retaliation. The parties resolved this matter, and the case was dismissed on November 6, 2019.
On November 19, 2018, the Company’s former Chief Financial Officer filed a complaint in the Superior Court for Cobb County, Georgia (Michael J. Senken v. MiMedx Group, Inc.) in which he claims that the Company has breached its obligations under the Company’s charter and bylaws to advance to him, and indemnify him for, his legal fees and costs that he incurred in connection with certain Company internal investigations and litigation. The Company filed its answer denying the plaintiff’s claims on April 19, 2019. To date, no deadlines have been established by the court.
On January 21, 2019, a former employee filed a complaint in the Fifth Judicial Circuit, Richland County, South Carolina (Jon Michael Vitale v. MiMedx Group, Inc. et. al.) against the Company alleging retaliation, defamation and unjust enrichment and seeking monetary damages. The former employee claims he was retaliated against after raising concerns related to insurance fraud and later defamed by comments concerning the indictments of three South Carolina VA employees. On February 19, 2019, the case was removed to the U.S. District Court for the District of South Carolina. The Company filed a motion to dismiss on April 8, 2019, which was denied by the Court. This case is in discovery.
In December 2019, MiMedx received notice of a complaint filed in July 2018 with the Occupational Safety and Health Administration (“OSHA”) section of the Department of Labor (“DOL”) by Thomas Tierney, a former Regional Sales Director, against MiMedx and the referenced individuals, Tierney v. MiMedx Group, Inc., Parker Petit, William Taylor, Christopher Cashman, Thornton Kuntz, Jr. and Alexandra Haden, DOL No. 4-5070-18-243. Mr. Tierney alleged that he was terminated from MiMedx in retaliation for reporting concerns about revenue recognition practices, compliance issues, and the corporate culture, in violation of the anti-retaliation provisions of the Sarbanes-Oxley Act. The parties settled this matter and OSHA dismissed the complaint on May 20, 2020.
Defamation Claims
On June 4, 2018, Sparrow Fund Management, LP (“Sparrow”) filed a complaint against the Company and Mr. Petit, including claims for defamation and civil conspiracy in the United States District Court for the Southern District of New York (Sparrow Fund Management, L.P. v. MiMedx Group, Inc. et. al.). The complaint seeks monetary damages and injunctive relief and alleges the defendants commenced a campaign to publicly discredit Sparrow by falsely claiming it was a short seller who engaged in illegal and criminal behavior by spreading false information in an attempt to manipulate the price of our Common Stock. On March 31, 2019, a judge granted defendants’ motions to dismiss in full, but allowed Sparrow the ability to file an amended complaint. The Magistrate has recommended Sparrow’s motion for leave to amend be granted in part and denied in part and the Judge adopted the Magistrate’s recommendation. Sparrow filed its amended complaint against MiMedx (Mr. Petit has been dropped from the lawsuit) on April 3, 2020 and the Company filed its answer. This case is in discovery.
On June 17, 2019, the principals of Viceroy Research (“Viceroy”), filed suit in the Circuit Court for the Seventeenth Judicial Circuit in Broward County, Florida (Fraser John Perring et. al. v. MiMedx Group, Inc. et. al.) against the Company and Mr. Petit, alleging defamation and malicious prosecution based on the defendants’ alleged campaign to publicly discredit Viceroy and the lawsuit the Company previously filed against the plaintiffs, but which the Company subsequently dismissed without prejudice. On November 1, 2019, the Court granted Mr. Petit’s motion to dismiss on jurisdictional grounds, denied the Company’s motion to dismiss, and granted plaintiffs leave to file an amended complaint to address the deficiencies in its claims against Mr. Petit, which they did on November 21, 2019. The Company filed its answer on December 20, 2019.
F- 41
Intellectual Property Litigation
The Bone Bank Action
On May 16, 2014, the Company filed a patent infringement lawsuit against Transplant Technology, Inc. d/b/a Bone Bank Allografts (“Bone Bank”) and Texas Human Biologics, Ltd. (“Biologics”) in the United States District Court for the Western District of Texas (MiMedx Group, Inc. v. Tissue Transplant Technology, LTD. d/b/a/ Bone Bank Allografts et. al.). The Company has asserted that Bone Bank and Biologics infringed certain of the Company’s patents through the manufacturing and sale of their placental-derived tissue graft products, and the Company is seeking permanent injunctive relief and unspecified damages. On July 10, 2014, Bone Bank and Biologics filed an answer to the complaint, denying the allegations in the complaint, and filed counterclaims seeking declaratory judgments of non-infringement and invalidity. The matter settled in 2019 prior to trial, and the case was dismissed on April 4, 2019.
The NuTech Action
On March 2, 2015, the Company filed a patent infringement lawsuit against NuTech Medical, Inc. (“NuTech”) and DCI Donor Services, Inc. (“DCI”) in the United States District Court for the Northern District of Alabama (MiMedx Group, Inc. v. NuTech Medical, Inc. et. al.). The Company has alleged that NuTech and DCI infringed and continue to infringe the Company’s patents through the manufacture, use, sale and/or offering of their tissue graft product. The Company has also asserted that NuTech knowingly and willfully made false and misleading representations about its products to customers and prospective customers. The Company is seeking permanent injunctive relief and unspecified damages. The case was stayed pending the restatement of the Company’s financial statements. Since the Company has completed its restatement, the case has resumed and discovery has recommenced.
The Osiris Action
On February 20, 2019, Osiris Therapeutics, Inc. (“Osiris”) refiled its trade secret and breach of contract action against the Company (which had been dismissed in a different forum) in the United States District Court for the Northern District of Georgia (Osiris Therapeutics, Inc. v. MiMedx Group, Inc.). Osiris has alleged that the Company acquired Stability, a former distributor of Osiris, in order to illegally obtain trade secrets. On February 24, 2020, the Court issued an order granting in part and denying in party MiMedx’s motion to dismiss. The Court dismissed Osiris’s claims for tortious interference, conspiracy to breach contract, unfair competition, and conspiracy to commit unfair competition. The Court denied MiMedx’s motion to dismiss with respect to the claim for breach of the contract between Osiris and Stability, finding that there is a question as to whether Osiris can maintain such a claim by piercing the corporate veil between MiMedx and its former subsidiary. If Osiris cannot pierce the corporate veil, the claim against MiMedx fails; if Osiris can pierce the corporate veil, the breach of contract claim must be brought in an arbitration proceeding. MiMedx did not move to dismiss Osiris’s claims for misappropriation of trade secrets and conspiracy to misappropriate trade secrets. MiMedx plans to defend against all remaining claims.
As of December 31, 2019, the Company has accrued approximately $12.8 million related to the legal proceedings discussed above. The Company has paid approximately $9.2 million to settle certain cases noted above.
As of December 31, 2018, the Company accrued $15.6 million related to legal proceedings and other matters of litigation.
Other Matters
Under the Florida Business Corporation Act and agreements with its current and former officers and directors, the Company is obligated to indemnify its current and former officers and directors who are made party to a proceeding, including a proceeding brought by or in the right of the corporation, with certain exceptions, and to advance expenses to defend such matters. The Company has already borne substantial costs to satisfy these indemnification and expense advance obligations and expects to continue to do so in the future.
In addition to the matters described above, the Company is a party to a variety of other legal matters that arise in the ordinary course of the Company’s business, none of which is deemed to be individually material at this time. Due to the inherent uncertainty of litigation, there can be no assurance that the resolution of any particular claim or proceeding would not have a material adverse effect on the Company’s business, results of operations, financial position or liquidity.
F- 42
17. Revenue Data by Customer Type
MiMedx has two primary distribution channels: (1) direct to customers (healthcare professionals and/or facilities) (“Direct Customers”); and (2) sales through distributors (“Distributors”). For purposes of the required disclosure under ASC 606-10-50-5, the Company groups its customers into these two groups. This grouping by customer types does not constitute a basis for resource allocation but is information intended to provide the reader with ability to better understand how the nature, amount, timing, and uncertainty of revenue and cash flows are affected by economic factors applicable to each customer type. These groupings also do not meet the criteria under ASC 280-10-50-1 to qualify as separate operating segments. The Company did not have significant foreign operations or a single external customer from which 10% or more of revenues were derived during the years ended December 31, 2019, 2018 and 2017.
Below is a summary of net sales by each customer type (in thousands):
Years Ended December 31, | |||||||||||
2019 | 2018 | 2017 | |||||||||
Direct Customers | $ | $ | $ | ||||||||
Distributors | |||||||||||
Other(1) | |||||||||||
Total | $ | $ | $ | ||||||||
18. | Related Party Transactions |
The Company employs Simon Ryan, the brother-in-law of the Company’s former General Counsel, Alexandra O. Haden (who resigned from the Company effective August 12, 2019), as a sales representative. In 2018, the Company paid Mr. Ryan total compensation of $0.2 million, consisting of a salary of $0.1 million and sales commissions, equity and other compensation of $0.1 million. In 2019, the Company paid Mr. Ryan total compensation of $0.2 million, consisting of a salary of $0.1 million and sales commissions, equity and other compensation of $0.1 million.
The Company has employed Thomas Koob as its Chief Scientific Officer (a non-executive officer) since 2006. Thomas Koob is the brother of a director, Charles Koob. Subsequent to the Company’s employment of Thomas Koob, Charles Koob was appointed as a director of the Company in March 2008. In 2018, the Company paid Thomas Koob a salary of $0.2 million and provided equity, incentive compensation and other compensation of $0.3 million. In 2019, the Company paid Thomas Koob an annual salary of $0.2 million and provided equity, incentive compensation and other compensation of $0.2 million.
19. | Restructuring |
Set forth below are disclosures relating to restructuring initiatives that resulted in material expenses or cash expenditures during the year ended December 31, 2019, and resulted in material restructuring liabilities at December 31, 2019. Employee retention and certain other employee benefit-related costs related to the Company’s restructuring are expensed ratably over an agreed-upon service period. One-time employee separation and related employee benefit costs are generally expensed as incurred.
In December 2018, the Company announced a reduction of the Company’s workforce by approximately 240 full-time employees, or 24 % of its total workforce, of which approximately half were sales personnel as part of the plans to implement a broad-based organizational realignment, cost reduction and efficiency program to better ensure the Company’s cost structure was appropriate given its revenue expectations.
As a result of the December 2018 broad-based organizational realignment, cost reduction and efficiency program, the Company incurred pre-tax charges of $8.5 million and $6.1 million during the years ended December 31, 2019 and 2018, respectively. The charges related to employee retention and other one-time employee separation benefit-related costs. These charges are included in the cost of sales, research and development, and selling, general and administrative expenses in the consolidated statements of operations.
F- 43
The liability related to restructuring activities during 2019 are included in accrued compensation in the consolidated balance sheets. Changes to this liability during the year ended December 31, 2019 were as follows (in thousands):
Liability balance as of January 1, 2018 | $ | ||
Expenses | |||
Cash distributions | ( | ) | |
Liability balance as of December 31, 2018 | |||
Expenses | |||
Cash distributions | ( | ) | |
Liability balance as of December 31, 2019 | $ | ||
20. | Quarterly Financial Data (Unaudited) (in thousands except per share data) |
The following table sets forth selected quarterly financial data for 2019 and 2018:
First Quarter | Second Quarter | Third Quarter (1) | Fourth Quarter | |||||||||||||
Net sales | 2019 | $ | $ | $ | $ | |||||||||||
2018 | $ | |||||||||||||||
Gross profit | 2019 | $ | $ | $ | $ | |||||||||||
2018 | $ | $ | ||||||||||||||
Income tax (provision) benefit | 2019 | $ | ( | ) | $ | ( | ) | $ | $ | ( | ) | |||||
2018 | ( | ) | ( | ) | ||||||||||||
Net income (loss) | 2019 | $ | ( | ) | $ | ( | ) | $ | $ | ( | ) | |||||
2018 | ( | ) | ( | ) | ||||||||||||
Net income (loss) per common share - basic | 2019 | $ | ( | ) | $ | ( | ) | $ | $ | ( | ) | |||||
2018 | ( | ) | ||||||||||||||
Net income (loss) per common share - diluted | 2019 | $ | ( | ) | $ | ( | ) | $ | $ | ( | ) | |||||
2018 | ( | ) | ||||||||||||||
(1) - Third quarter amounts include the transition adjustment discussed in Note 3.
21. | Subsequent Events |
Coronavirus Aid, Relief and Economic Security (CARES) Act
On March 27, 2020, the “Coronavirus Aid, Relief and Economic Security (CARES) Act” was signed into law. The Act includes provisions relating to refundable payroll tax credits, deferment of the employer portion of certain payroll taxes, loans and grants to certain business, net operating loss carryback periods, alternative minimum tax credit refunds, modifications to the net interest deduction limitations and technical corrections to tax depreciation methods for qualified improvement property. The Company applied for and received a $10.0 million loan under the Paycheck Protection Program. On May 11, 2020 the Company repaid the PPP loan.
F- 44
In addition, modifications to the tax rules for carryback of net operating losses are expected to result in an estimated federal tax refund of $11.3 million and a resulting income tax benefit.
The COVID-19 pandemic and governmental and societal responses thereto have affected the Company’s business, results of operations and financial condition. The continuation or a second-wave outbreak of COVID-19 or the outbreak of other health epidemics could harm the Company’s operations and increase the Company’s costs and expenses in numerous ways. The ultimate impact of the COVID-19 pandemic is highly uncertain and subject to change. The Company does not yet know the full extent of delays or impacts on the business, clinical trials, healthcare systems or the global economy as a whole, or how long such effects will endure. The effects of the COVID-19 pandemic or other health epidemics could have an adverse impact on the Company’s business, results of operations and financial condition.
Sublease
On April 1, 2020 the Company successfully subleased its industrial warehouse space that expires on May 31, 2023. The Company performed an asset recovery test comparing the sum of estimated undiscounted future cash flows attributable to the sublease to its carrying amount. The total undiscounted cash flows for the remaining lease term exceed the carrying amount of the asset, therefore there is no impairment.
BT Term Loan Amendment
On April 22, 2020, the Company amended its BT Loan Agreement with Blue Torch. The amendment provided for an increase in the maximum Total Leverage Ratio (as defined in the BT Loan Agreement), which was a quarterly test, for the remainder of 2020, and also provided for a reduction in the minimum Liquidity (as defined in the BT Loan Agreement) requirement from April 2020 through and including November 2020. Specifically, the maximum Total Leverage Ratio increased from 3.0 to 1.0 to 5.0 to 1.0 through December 31, 2020. The minimum Liquidity requirement was reduced from $40.0 million to $20.0 million for April and May 2020, and from $30.0 million to $20.0 million for June through November 2020. In connection with the amendment, the Company agreed to pay a one-time fee of approximately $0.7 million, added to the principal balance, and a 1 percentage point increase in the interest rate to LIBOR plus 9 %.
Repayment and Termination of BT Loan Agreement
On July 2, 2020, the Company terminated the BT Loan Agreement and repaid the $72.0 million outstanding balance of principal and accrued but unpaid interest under the BT Loan Agreement. As a result of the early repayment of the loans under the BT Loan Agreement, the Company also paid a prepayment premium in the amount of $1.4 million. The Company paid the outstanding balance, accrued but unpaid interest, and prepayment premium using a portion of the proceeds from the Preferred Stock Transaction and the Hayfin Loan Transaction.
Issuance of $100 Million of Series B Convertible Preferred Stock
On July 2, 2020, the Company issued $100 million of the Company’s Series B Convertible Preferred Stock, par value $0.001 per share (the “Series B Preferred Stock”) to an affiliate of EW Healthcare Partners and certain funds managed by Hayfin Capital Management LLP pursuant to a Securities Purchase Agreement with Falcon Fund 2 Holding Company, L.P., an affiliate of EW Healthcare Partners, and certain funds managed by Hayfin Capital Management LLP, dated as of June 30, 2020, for an aggregate purchase price of $100,000,000 (the “Preferred Stock Transaction”). See Item 9B, “Other Information.”
$75 Million Loan Facility with Hayfin
On June 30, 2020, the Company entered into a Loan Agreement with, among others, Hayfin Services, LLP, an affiliate of Hayfin Capital Management LLP (the “Hayfin Loan Agreement”), which was funded on July 2, 2020 (the “Hayfin Loan Transaction”) and provided the Company with a senior secured term loan in an aggregate amount of $50 million (the “Term Loan”) and an additional $25 million delayed draw term loan (the “DD TL”) in the form of a committed but undrawn facility. The Term Loan and the DD TL mature on July 2, 2025 (the “Maturity Date”). The Term Loan and the DD TL have no fixed amortization (i.e. interest only through the Maturity Date).
Borrowings under the Hayfin Loan Agreement bear interest at a rate equal to LIBOR (subject to a floor of 1.5 %) plus a margin of 6.75 %. The margin will be eligible to step down to 6.5 % or 6.0 % based on future Total Net Leverage levels, as defined in the Hayfin Loan Agreement. The Company paid an upfront commitment fee of 2 % of the aggregate of the Hayfin Term Loan and the DD TL. The DD TL is subject to an additional commitment fee of 1 % of the amount undrawn.
The Hayfin Loan Agreement contains certain affirmative covenants that impose certain reporting and/or performance obligations on the Company and its subsidiaries, including (i) Maximum Total Net Leverage of 5.0 x through December 31, 2020, stepping
F- 45
Schedule II Valuation and Qualifying Accounts
MIMEDX GROUP, INC. AND SUBSIDIARIES SCHEDULE II VALUATION AND QUALIFYING ACCOUNTS | ||||||||||||||||
Years ended December 31, 2019, 2018 and 2017 (in thousands) | ||||||||||||||||
Balance at Beginning of Year | Additions charged to Expense or Revenue | Deductions and write-offs | Balance at End of Year | |||||||||||||
For the Year ended December 31, 2019 | ||||||||||||||||
Allowance for product returns | ( | ) | ||||||||||||||
Allowance for obsolescence | ( | ) | ||||||||||||||
For the Year ended December 31, 2018 | ||||||||||||||||
Allowance for product returns | $ | $ | $ | $ | ||||||||||||
Allowance for obsolescence | ( | ) | ||||||||||||||
For the Year ended December 31, 2017 | ||||||||||||||||
Allowance for product returns | $ | $ | $ | ( | ) | $ | ||||||||||
Allowance for obsolescence | ( | ) | ||||||||||||||
F- 46
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Management maintains a set of disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), designed to ensure that information required to be disclosed by us in reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC's rules and forms, and that such information is accumulated and communicated to management, including our CEO and CFO, to allow for timely decisions regarding required disclosure.
An evaluation of the effectiveness of the design and operation of our disclosure controls and procedures was performed under the supervision and with the participation of our management, including our CEO and CFO. As a result of this evaluation, our CEO and CFO concluded that our disclosure controls and procedures were not effective as of December 31, 2019 because of certain material weaknesses in internal control over financial reporting, as further described below.
Management's Report on Internal Control Over Financial Reporting
Management, including our CEO and CFO, is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act and based upon the criteria established in Internal Control-Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (the "COSO framework" ). The Company's internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of our financial reporting and the preparation of our financial statements for external purposes in accordance with United States generally accepted accounting principles (“GAAP”).
An effective internal control system, no matter how well designed, has inherent limitations, including the possibility of human error or overriding of controls, and therefore can provide only reasonable assurance with respect to reliable financial reporting. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. In addition, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may demonstrate.
Under the supervision and with the participation of our management, including our CEO and CFO, we have conducted an evaluation of the effectiveness of our internal control over financial reporting based on the COSO framework. Based on evaluation under these criteria, management determined, based upon the existence of the material weaknesses described below, that we did not maintain effective internal control over financial reporting as of December 31, 2019.
A material weakness (as defined in Rule 12b-2 under the Exchange Act) is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that a reasonable possibility exists that a material misstatement of our annual or interim financial statements will not be prevented or detected on a timely basis.
The Company previously disclosed material weaknesses in internal control over financial reporting as of December 31, 2018 in our Annual Report on Form 10-K for the year ended December 31, 2018. The previously disclosed material weaknesses related to our control environment, risk assessment, control activities, information and communication, and monitoring activities. The material weaknesses led to the delayed filing of our annual consolidated financial statements for the years ended December 31, 2018 and 2017 and the restatement of our financial statements for the year ended December 31, 2016. As described below, while management has developed and implemented certain remediation actions to address the material weaknesses, further actions are still ongoing or have not been implemented for a sufficient amount of time to test and conclude on the effectiveness of the remediation actions as of December 31, 2019. As a result, the material weaknesses continue to be present as of December 31, 2019. Management has reported to the Audit Committee the status of these remediation actions. These control deficiencies could have resulted in other misstatements in financial statement accounts and disclosures that would result in a material misstatement to the annual or interim consolidated financial statements that might not have been prevented or detected.
The Company identified material weaknesses corresponding to the control environment and control activities components of internal control as defined by COSO as described below:
Control Environment
79
The Company did not maintain an effective control environment based on the criteria established in the COSO framework. Specifically, the Company identified control deficiencies that constitute material weaknesses, either individually or in the aggregate, relating to: (i) appropriate organizational structure, reporting lines, and authority and responsibilities in pursuit of objectives, and (ii) holding individuals accountable for their internal control related responsibilities.
Control Activities
The control deficiencies identified specific to the Control Activities COSO element constitute material weaknesses, either individually or in the aggregate, relating to: (i) the operation of control activities and general information technology controls that contribute to the mitigation of risks and support achievement of objectives for a sufficient period of time during the year ended December 31, 2019 and (ii) deploying control activities through policies that establish what is expected and procedures that put policies into action. Deficiencies in control activities contributed to the potential for there to have been material accounting errors in substantially all financial statements account balances and disclosures, specifically:
• | Information Technology General Controls (ITGC’s) for certain information technology systems and other ITGCs did not operate effectively for a sufficient period of time for the Company to rely on the accuracy and completeness of information on which certain business process controls (automated and manual) are dependent. As a result, it is possible that the Company’s business process controls that depend on the accuracy and completeness of data or financial reports generated by the information technology system could be adversely affected due to the lack of operating effectiveness of ITGC’s. |
• | There was a lack of robust, established and documented accounting policies and insufficiently detailed Company procedures to ensure controls operated as designed and policies were applied effectively to ensure material transactions were recorded in the financial statements. |
• | The Company did not have adequate management documentation around the completeness and accuracy of data material to financial reporting of certain transactions including revenue recognition and completeness of inventory. |
• | The Company, for certain processes, did not maintain adequate controls around segregation of duties within the revenue process. Other application controls related to revenue were not evaluated because of ineffective ITGCs or failed due to inadequate evidentiary matter or controls did not operate in a consistent manner during the year ended December 31, 2019. |
• | The Company did not design and maintain adequate controls to ensure that accounting for income tax provisions were appropriately recorded in accordance with GAAP. |
• | The Company did not have adequate staffing resources to properly perform review of certain accounting determinations, including (but not limited to) the following: review of significant assumptions for stock-based compensation expense, timely review of consignment inventory, the review of significant assumptions used to estimate accrued expenses. |
• | The Company did not design and maintain adequate controls over the inventory process and related accounting assumptions, to ensure that accounting determinations related to inventory appropriately considered and recorded in accordance with GAAP, that the inventory balance was complete and accurate and that disclosures related to the inventory balance were appropriately reflected within the financial statements. |
• | The Company’s controls over financial close and reporting did not operate effectively for a sufficient period of time to meet a variety of its financial reporting objectives which exposed the financial statements to potential for disclosure that did not meet the requirements of GAAP. |
BDO USA, LLP, our independent registered public accounting firm, has audited the effectiveness of our internal control over financial reporting as of December 31, 2019. BDO USA, LLP has expressed an adverse report on internal control over financial reporting which appears on page F-2 of this Form 10-K.
Remediation Plan and Status
Remediation of the identified material weaknesses and strengthening our internal control environment was an identified priority for us throughout 2019 and will continue to be a priority in 2020. We will test the design and ongoing operating effectiveness of the new and existing controls in future periods. The material weaknesses cannot be considered completely remediated until the applicable controls have operated for a sufficient period of time and management has concluded, through testing, that these controls are operating effectively. With continued oversight from the Audit Committee, the Company's management has designed and begun implementing changes in processes and controls to remediate the material weaknesses described above and has
80
improved the Company's internal control over financial reporting as follows:
Control Environment
The Board created an Ethics and Compliance Committee consisting solely of independent directors which is responsible for reviewing the status of the Company's ethics and compliance program, reviewing and advising the Board regarding any open cases and trends that may impact the business, and recommending future initiatives to improve compliance performance and effectiveness.
Management reinforced the importance of integrity, accountability, and adherence to the redesigned internal controls, policies, and procedures through the adoption of a revised Code of Business Conduct Policy. All Board members and employees, including executives, newly hired employees and agents are required to certify that they read and understood the policy upon hire and all said individuals then re-certify their reading and understanding of the policy annually thereafter.
The Company enhanced the onboarding training provided to newly hired salespeople to emphasize the importance of compliance with the various regulations specific to the Life Sciences industry to which the Company is subject. The Company is implementing a policy to ensure required trainings are completed by relevant personnel.
Management began, and will continue, to schedule training sessions with the Company's Sales Department to ensure that they are familiar with the Company's current sales related policies and procedures, including those which are significant to the Company's financial reporting objectives. Portions of these training sessions are facilitated by the Chief Accounting Officer (“CAO”), who presents on topics such as the Company's current sales return policy, acceptable credit terms for customers, events that would trigger commission claw-backs, customer credit limit modification approval protocol, and the importance of proper revenue recognition.
The Ethics and Compliance Committee of the Board, and the full Board, have received regular updates of the ethics and compliance efforts of the Company from the SVP and Chief Compliance Officer. This includes an explanation of all the policies and procedures that have either been revised and reissued or newly created. Both the Ethics and Compliance Committee and the Board have also both helped in reviewing, approving and training on the new Code of Business Conduct and Ethics.
The purpose of the whistleblower hotline and the mechanics of its use were formally communicated by the Chief Compliance Officer during numerous meetings with all levels of the sales department during 2019, with an emphasis on the following: (a) each employee's responsibility to report any actual or apparent violations of law or ethical standards and any questionable accounting or auditing matters so that they may be investigated and dealt with appropriately, (b) management's commitment to ensuring that any employees communicating such an issue via the hotline will not be subject to retaliation, and (c) the Board of Directors’ oversight of complaints raised through the hotline to ensure appropriate actions are taken.
In addition to enhancing processes and controls over adoption of new accounting standards and the proper application of existing accounting standards, the Company enhanced the technical capabilities of its accounting department by leveraging third party consultants with expertise in GAAP. As of the date of the filing of this Form 10-K, the Company has hired a new Chief Financial Officer and a Chief Accounting Officer. Furthermore, the Company intends to lessen its reliance on third-party consultants for its technical accounting needs during 2020 by transitioning roles currently assigned to outside consultants to full-time employees with similar technical accounting competencies.
Management plans to develop and implement a contract management policy that defines who is required to review new, extended, or amended contracts (including those with distributors and agents). This policy includes the implementation of a checklist for standard and non-standard contracts to ensure that the revenue recognition criteria are properly considered for each of the standard and non-standard contracts.
Control Activities
We previously disclosed that in 2018, management concluded there to be a material weakness in the application of the Company’s revenue recognition methodology which was not aligned with the Company's customary business practices, resulting in certain revenue events being recorded prior to the time at which all of the sales recognition criteria were met. To remediate the material weakness specific to the revenue recognition methodology, the Company implemented controls in which the customary business practices were aligned to GAAP criteria to determine the point at which revenue recognition is appropriate. See Transition in Revenue Recognition footnote disclosures.
Management collaborated with outside consultants possessing significant financial reporting and internal control expertise to perform an extensive review of the design of the Company's internal controls over financial reporting. This review included the identification of internal control deficiencies and the development of remediation plans for each identified deficiency. These
81
internal control deficiencies identified included (but were not limited to) the following: improvements to the financial close and reporting process, accounting for satisfaction of performance obligations related to revenue recognition, calculation of inventory costing and related accuracy of inventory, accounting for income taxes, accurate calculation of stock-based compensation expense, timely review of consignment inventory and the development of quality estimates related to accrued expenses.
The Company is enhancing its financial close process by formalizing its accounting policies, introducing additional layers of independent reviews by appropriately qualified individuals, improving the precision and timeliness of reviews applied to various financial result analyses, and providing required education and training to the members of the finance department.
The Company is enhancing the design and adherence to controls addressing the accuracy and completeness of the accounting for income taxes, including retention of evidence of review and review of significant judgements to ensure proper application of GAAP.
Management is enhancing its oversight of the completeness and accuracy of data material to financial reporting by establishing criteria in the performance of controls to evaluate the accuracy and completeness of data. Management is implementing required training for control owners specific to the evaluation of the accuracy and completeness of data used in control activities.
The Company continues to conduct required training and education for control owners in critical financial reporting roles.
The Company has enhanced its review of salesperson activity which may indicate noncompliance with the Company's sales policies, such as a quarterly review of data by the CFO, CAO, and SVP of Sales and other key metrics both by region and at the individual salesperson level and has added controls to monitor potential instances of noncompliance.
Management has gained a better understanding of system functionality through a comprehensive review of permissions and profiles within each IT application that is significant to the Company's financial reporting objectives, and subsequently reconfigured profiles with appropriate permissions to better align with job responsibilities and enforce segregation of duties. Once user profiles and their associated permissions were reconfigured, management employed procedures to ensure the continued appropriateness of all applicable system and network access. This objective was achieved through the performance of periodic user access reviews and the enhancement of procedures related to the granting and removing of system and network access, however, these controls have not operated for a sufficient period of time for management to evaluate the effectiveness of these remediated controls.
Management modified its policy regarding the periodic review of sales to involve the Finance Department in an effort to enhance the Finance Department's awareness and oversight of sales activities in order to verify the validity and proper accounting treatment of sales transactions.
Risk Assessment
We previously disclosed that in 2018, management concluded there to be a material weakness in the Risk Assessment specific to the lack of an appropriate risk assessment, the lack of processes for communicating changes to risks throughout the organization and lack of policy to ensure the accounting department was aware of sales practices. Through the completion of the following activities, the previously disclosed material weaknesses related to risk assessment have been remediated:
• | The Compliance function, led by the SVP and Chief Compliance Officer, has conducted several enterprise-wide risk assessments since 2018. The results of those audits have been shared with the Ethics and Compliance Committee initially and regular updates have been provided on the Company’s risk assessment program. |
• | Management developed a process to prepare and did prepare an annual comprehensive fraud risk assessment designed to evaluate risks related to fraudulent financial reporting, management override, potential loss of assets, and corruption. The methodology adopted within this assessment is designed to evaluate the impact and likelihood of various fraud risks susceptible to the Company. Such risks, if relevant, are then mapped to controls within the current risk environment. |
• | Management developed a set of controls to identify and define its population of related parties, identify transactions with those related parties, and analyze such transactions to determine whether additional approval or financial statement disclosure is required. |
• | The Company established a Disclosure Committee comprised of senior management representatives from all relevant departments within the organization. Members of this committee were and are responsible for reviewing quarterly and annual SEC filings to ensure that the disclosures within the filings are reflective of the knowledge of the Company and the Company’s operations that each member of the committee brings to the review process. The members of the committee met prior to each filing to discuss the completeness and accuracy of the document being filed, if applicable, suggest |
82
additional disclosure, and once the Committee believed the disclosure to be appropriate, approved the draft filing for audit committee consideration.
• | The Company designed and implemented a variety of new controls, including monthly operational meetings amongst senior management that are attended by members of the accounting department, to ensure that the accounting department is aware of operational changes that may affect the Company's accounting policies. |
• | On an annual basis (or more frequently, should a significant triggering event occur), the Company now performs a risk assessment designed to ensure that the scope of its Sarbanes-Oxley compliance program adequately reflects changes to the business and its operations. If a significant triggering event occurs, the Company evaluates the key control activities related to the transaction or activity and determines that the related controls are within the scope of the Sarbanes-Oxley compliance program. |
Information and Communication
We previously disclosed that in 2018, management concluded there to be a material weakness in the Company’s Information and Communications activities specific to the generation and provision of quality information as established under the requirements of COSO. Through the completion of the following activities, the previously disclosed material weaknesses related to information and Communication have been remediated:
• | Management conducted required internal training courses over Sarbanes-Oxley regulations, the Company's internal control over financial reporting program and documentation evidencing the operation of controls for Company personnel and management involved in the execution of controls. |
• | Management developed a regular cadence for reporting the results of control testing to the board of directors of the Company. |
• | Management implemented quarterly required communications amongst relevant members of senior management in the form of certification surveys. A control certification survey is distributed to obtain information regarding any internal control related issues or concerns that control owners may have, and additional certification surveys are distributed to Disclosure Committee members and key members of the sales department which address (to the best of their knowledge) whether the period's financial statements are free from either material misstatements, material misclassifications, or material omissions. |
Monitoring Activities
We previously disclosed that in 2018, management concluded there to be a material weakness in the Company’s monitoring activities specific to the assessment of controls, the competency of those monitoring the control environment and the lack of adequate procedures to monitor when controls are overridden. Through the completion of the following activities, the previously disclosed material weaknesses related to monitoring activities have been remediated:
• | Established its Internal Audit Department, led by a VP of Internal Audit under the direction of the audit committee. The Internal Audit Department identified and hired internal resources with the appropriate level of competency, who are tasked with continually evaluating and monitoring the effectiveness of the Company's internal controls over financial reporting. |
• | Management established a framework for identifying, communicating and remediating internal control deficiencies, which includes appropriate escalation of issues to appropriate stakeholders in the internal control framework and communication of remediation status, as relevant, to the board of directors on a regular basis. |
• | Management led required training sessions with the Company's Sales Department to ensure that they are familiar with the Company's current sales related policies and procedures, including those which are significant to the Company's financial reporting objectives. Portions of these training sessions were facilitated by the CAO, who presents on topics such as the Company's current sales return policy, acceptable credit terms for customers, events that would trigger commission claw-backs, customer credit limit modification approval protocol, and the importance of proper revenue recognition. |
Changes in Internal Control Over Financial Reporting
83
Other than the changes described above in “Remediation Plan and Status,” there were no changes during the quarter ended December 31, 2019 in our internal control over financial reporting (as such term is defined in the Exchange Act) that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
Item 1.01 Entry into a Material Definitive Agreement.
Item 3.02 Unregistered Sales of Equity Securities.
Item 3.03 Material Modification to Rights of Security Holders.
Item 5.03 Amendments to Articles of Incorporation or Bylaws; Change in Fiscal Year.
Issuance of $100 Million of Series B Convertible Preferred Stock
On June 30, 2020, the Company entered into a Securities Purchase Agreement (the “Purchase Agreement” or “Securities Purchase Agreement”) with Falcon Fund 2 Holding Company, L.P. (the “EW Purchaser”), an affiliate of EW Healthcare Partners, and certain funds managed by Hayfin Capital Management LLP (the “Hayfin Purchasers” and together with the EW Purchaser, the “Purchasers”), in connection with the offering, issuance, and sale of (1) 90,000 shares of the Company’s Series B Convertible Preferred Stock, par value $0.001 per share (the “Series B Preferred Stock”) to the EW Purchaser for an aggregate purchase price of $90,000,000, and (2) 10,000 shares of Series B Preferred Stock in the aggregate to the Hayfin Purchasers for an aggregate purchase price of $10,000,000, in each case on the terms and subject to the conditions of the Purchase Agreement (such shares, the “Purchased Shares” and such transaction, the “Preferred Stock Transaction”).
Pursuant to the Purchase Agreement, the Company has filed an amendment to its articles of incorporation, as amended, setting forth the terms of the Series B Preferred Stock (the “Preferred Stock Amendment”). The Company completed the closing of the sale and purchase of the Purchased Shares (the “Preferred Stock Closing”) on July 2, 2020. The offering and sale of the Purchased Shares was exempt from registration under the Securities Act of 1933, as amended (the “Securities Act”), pursuant to Section 4(a)(2) of the Securities Act and certain rules and regulations thereunder. The shares of Common Stock issuable upon conversion of the shares of Series B Preferred Stock will be issued in reliance upon the exemption from registration in Section 3(a)(9) of the Securities Act.
The proceeds from the sale of the Purchased Shares have been or will be used to repay outstanding debt, as further described below, for working capital and general corporate purposes and to pay transaction fees, costs and expenses incurred in connection with the transactions contemplated by the Purchase Agreement.
Terms of the Purchase Agreement
The Purchase Agreement contains representations, warranties and covenants of the Company and the Purchasers customary for transactions of this type. In addition, certain specific terms and conditions of the Purchase Agreement are described below.
Purchaser Director and Nominees
On the terms and subject to the conditions of the Purchase Agreement and the Preferred Stock Amendment, for so long as the EW Purchaser and its affiliates have beneficial ownership of (i) at least 10.0% of the total number of outstanding shares of common stock of the Company, par value $0.001 per share (“Common Stock”) (calculated on a fully-diluted, as converted basis) (a “10% Holder”), the EW Purchaser will be entitled to designate two individuals to serve on the Board, and (ii) at least 5.0% but less than 10% of the total number of outstanding shares of Common Stock (calculated on a fully-diluted, as converted basis) (a “5% Holder”), the EW Purchaser will be entitled to designate one individual to serve on the Board (such appointed directors, the “Preferred Directors”). The Preferred Directors will not be members of any class of directors that is elected by the holders of shares of Common Stock (a “Common Director”). However, the Board may, by notice to the EW Investor, either appoint such Preferred Director as a Common Director or nominate such director for election as a Common Director, provided that (i) no such appointment or nomination takes place such that such director would be up for election as a Common Director prior to the 2022 annual meeting of shareholders of the Company, and (ii) if anyone the EW Purchaser has designated to serve on the Board has been appointed or nominated as a Common Director prior to July 2, 2022, then no other person designated by the EW Purchaser to serve on the Board may be appointed or nominated as a Common Director prior to July 2, 2022. From and after the time that no Series B Preferred Stock remains outstanding, the EW Purchaser’s right to designate directors in accordance with the preceding sentence will convert into a right, subject to the same ownership thresholds described above, to designate up to two individuals to be nominated by the Company to serve on the Board. The initial Preferred Directors are Martin P. Sutter and William A. Hawkins, III, who were appointed to the Board of Directors effective as of July 2, 2020.
Lock-Up Period
84
The Purchasers may not transfer any of the Purchased Shares (or any Common Stock into which the Purchased Shares are convertible) for a period of two years after the Preferred Stock Closing (the “Lock-Up Period”), subject to certain customary exceptions. After the Lock-Up Period, the Purchasers may transfer the Purchased Shares (or shares of Common Stock into which the Purchased Shares are convertible) to any person subject to certain limited restrictions designed to prevent transfers to competitors of the Company.
Standstill
Subject to certain customary exceptions, the Purchasers are subject to a standstill provision which restricts them and their affiliates from taking certain actions without the consent of the board of directors (acting upon a majority vote of the directors other than the designees of the EW Purchaser to the board) including (i) acquiring any securities or material assets or businesses of the Company or its subsidiaries, (ii) proposing any merger, business combination, recapitalization, restructuring or other extraordinary transaction with the Company or its subsidiaries, (iii) initiating shareholder proposals or convening a shareholder’s meeting of the Company or its subsidiaries, (iv) soliciting proxies or otherwise seeking to influence, advise or direct the voting of capital stock of the Company, (v) influencing, advising, changing or controlling the management, board of directors, governing instruments, affairs or policies of the Company or any of its subsidiaries, and (vi) forming, joining or participating in any “group” (within the meaning of Section 13(d)(3) of the Exchange Act), until (1) in the case of the EW Purchaser, the later of (x) July 2, 2023, and (y) the date on which the EW Purchaser is no longer a 10% Holder nor a 5% Holder, and (2) in the case of the Hayfin Purchaser, July 2, 2023.
Preemptive Rights
Subject to customary exceptions, so long as the EW Purchaser is a 10% Holder, if the Company intends to issue and sell equity securities to any person, then the EW Purchaser has the right to participate in such equity offering up to its pro-rata percentage of such equity securities (calculated on a fully-diluted, as converted basis).
Terms of the Series B Preferred Stock
Ranking and Liquidation Preference: | The Series B Preferred Stock ranks senior to Common Stock with respect to dividends and distributions on liquidation, winding-up, and dissolution. Upon a liquidation, dissolution, or winding-up of the Company, each share of Series B Preferred Stock will be entitled to receive $1,000 per share (the “Purchase Price Per Share”), plus any accrued and unpaid dividends (the “Liquidation Preference”). |
Conversion at Purchaser’s Option: | Each holder of Series B Preferred Stock (each a “Holder” and collectively, the “Holders”) will have the right, at its option, to convert its Series B Preferred Stock, in whole or in part, into a number of fully paid and non-assessable shares of Common Stock equal to the Purchase Price Per Share, plus any accrued and unpaid dividends, divided by $3.85 (the “Conversion Price”). No Holder may convert its shares of Series B Preferred Stock into shares of Common Stock if such conversion would result in the Holder, together with its affiliates, holding more than 19.9% of the votes entitled to be cast at any stockholders meeting or beneficially owning in excess of 19.9% of the then-outstanding shares of Common Stock (the “Beneficial Ownership Cap”). |
Mandatory Conversion: | The Series B Preferred Stock (including any accrued and unpaid dividends) will, subject to the Beneficial Ownership Cap, automatically convert into Common Stock at any time after July 2, 2023, provided that the Common Stock has traded at 200% or more of the Conversion Price for 20 out of 30 consecutive trading days and as of the close of trading on the trading day immediately prior to the date of conversion, the Common Stock has traded at 200% or more of the Conversion Price. To the extent any Series B Preferred Stock cannot be converted due to operation of the Beneficial Ownership Cap, it shall remain outstanding and automatically convert at such time as such conversion would be permitted under the Beneficial Ownership Cap. |
Dividends: | The Holders will be entitled to cumulative dividends at a rate of 4.0% per annum for the period ending June 30, 2021 and 6.0% per annum thereafter, in each case compounding quarterly in arrears. The dividends are payable quarterly in whole or in part, in cash. However, the Company may, at its option, elect to not pay such dividend and to instead accrue the amount of such dividend. Accrued and unpaid dividends will be paid in cash or included in the conversion of the Series B Preferred Stock upon the occurrence of the Mandatory Conversion or the Company’s redemption of the Series B Preferred Stock. |
Voting: | Subject to certain exceptions, each share of Series B Preferred Stock is entitled to be voted on by the Holders and will vote on an as-converted basis as a single class with the Common Stock, subject to certain limitations on voting set forth in the related Articles of Amendment. |
85
Consent Rights: | The following matters will require the approval of the majority of the outstanding Series B Preferred Stock, voting as a separate class: - any changes to the rights, preferences, or privileges of the Series B Preferred Stock; - amendments or restatements of any organizational document of the Company or its subsidiaries in a manner that materially, adversely and disproportionately affects the rights, preferences and privileges of the Series B Preferred Stock as compared to Common Stock; - the authorization or creation of any class or series of senior or parity equity securities; - the declaration of any dividends or any other distributions, or the repurchase or redemption, of any equity securities of the Company ranking junior to or on parity with the Series B Preferred Stock (subject to certain exceptions); - prior to January 2, 2023 the sale, transfer, or other disposition of any assets, business, or operations for $25 million or more (other than sales of inventory in the ordinary course of business), or the purchase or acquisition of any assets, business, or operations for $75 million or more; - prior to January 2, 2023, the merger or consolidation of the Company unless either (x) the surviving company will have no class of equity securities ranking superior in parity with the Series B Preferred Stock or (y) the Holders of the Series B Preferred Stock will receive in connection therewith consideration per share of Series B Preferred Stock valued at 200% or more of the Purchase Price Per Share; - prior to January 2, 2023, commencing a voluntary case under any applicable bankruptcy, insolvency, or other similar law or consenting to the entry of an order for relief in an involuntary case under any such law, or effectuating any general assignment for the benefit of creditors; and - prior to the January 2, 2023, enter into any settlement agreement regarding the Company’s securities class action litigation. |
Change of Control: | If the Company undergoes a Change of Control (as defined in the Preferred Stock Amendment), the Company will have the option to repurchase any or all of a Holder’s then-outstanding shares of Series B Preferred Stock for cash in an amount equal to the Liquidation Preference, plus all accrued and unpaid dividends, subject to the right of each Holder to convert its Series B Preferred Stock into Common Stock. If the Company does not exercise such repurchase right, the Holder will have the option to (i) require the Company to repurchase any or all of its then-outstanding shares of Series B Preferred Stock for cash in an amount equal to the Liquidation Preference or (ii) convert its Series B Preferred Stock (including accrued and unpaid dividends) into Common Stock and receive its pro rata consideration thereunder. |
Anti-dilution | The Conversion Price is subject to certain customary anti-dilution adjustments if the Company issues shares of Common Stock as a dividends or distribution on the Common Stock or effects a stock split or stock combination of the Common Stock. The Conversion Price is also subject to a weighted average anti-dilution adjustment if the Company issues Common Stock (or securities convertible into Common Stock) at a price per share less than the Conversion Price within two years after Preferred Stock Closing but such adjustment may not result in a Conversion Price of less than $3.47. |
Registration Rights Agreement
On July 2, 2020, the Company and the EW Purchaser also entered into a registration rights agreement obligating the Company to register for resale shares of Common Stock issued upon conversion of its Series B Preferred Stock, which rights may be exercised from and after the date 90 days prior to the expiration of the Lock-Up Period. In general, the Registration Rights Agreement provides the EW Purchaser with the right to request a shelf registration in respect of such resales (including up to two underwritten shelf takedowns (but no more than one in any twelve month period)), up to two demand registrations (but only if no shelf registration is then in effect covering the resale of all securities held by the EW Purchaser) and unlimited piggyback registration rights.
The foregoing descriptions of the Purchase Agreement, the terms of the Series B Preferred Stock, and the Registration Rights Agreement are not complete and are qualified in their entirety by reference to the full text of the Purchase Agreement and the Preferred Stock Amendment, and the Registration Rights Agreement, copies of which are filed as Exhibits 10.38, 3.3, and 10.39 to this Annual Report on Form 10-K and are incorporated herein by reference.
The Purchase Agreement contains representations and warranties by each of the parties to the Purchase Agreement, which were made only for purposes of that agreement and as of specified dates. The representations, warranties, and covenants in the Purchase Agreement were made solely for the benefit of the parties to the Purchase Agreement; are subject to limitations agreed upon by the contracting parties, including being qualified by confidential disclosure schedules; may have been made for the purposes of allocating contractual risk between the parties to the Purchase Agreement instead of establishing these matters as facts; and are
86
subject to standards of materiality applicable to the contracting parties that may differ from those applicable to investors. Investors should not rely on the representations, warranties, and covenants or any descriptions thereof as characterizations of the actual state of facts or condition of the Company or any of its subsidiaries or affiliates. Moreover, information concerning the subject matter of the representations, warranties, and covenants may change after the date of the Purchase Agreement, which subsequent information may or may not be fully reflected in the Company’s public disclosures.
$75 Million Loan Facility with Hayfin
The Company entered into a Loan Agreement, dated as of June 30, 2020, (the “Hayfin Loan Agreement”), by and among the Company, certain of the Company’s subsidiaries, Hayfin Services LLP, as administrative agent and collateral agent, and other funds managed by Hayfin Capital Management LLP, that provides the Company with a senior secured term loan in an aggregate principal amount of $50 million (the “Hayfin Term Loan”), which was funded on July 2, 2020 (the “Hayfin Loan Closing Date”), and an additional $25 million delayed draw term loan (the “DD TL” and together with the Hayfin Term Loan, the “Loans”) in the form of a committed facility that is available for drawdown from the Hayfin Loan Closing Date (as defined below) to the first anniversary thereof (the “Hayfin Loan Transaction”).
The Hayfin Loan Agreement does not provide for the issuance of warrants or other equity interests in the Company.
The proceeds of the Loans are permitted to be used for (i) working capital and general corporate purposes (including, without limitation, the funding of forecasted growth, compliance and capital expenditures initiatives), (ii) to consummate the refinancing of the BT Loan Agreement as defined and described below, and (iii) to pay transaction fees, costs and expenses incurred in connection with the Loan Agreement and related transactions.
Repayment; Maturity. The Loans, including any DD TL, if borrowed, mature on July 2, 2025 (the “Maturity Date”). The Hayfin Term Loan and the DD TL have no fixed amortization (i.e. accrued interest only is payable through the Maturity Date).
Interest Rate; Fees. The Loans will bear interest at a per annum rate equal to LIBOR (subject to a “floor” of 1.5%) plus a margin of 6.75%. Such margin is subject to step down after December 31, 2020 to 6.5% or 6.0% based on Total Net Leverage Ratio levels, as defined in the Hayfin Loan Agreement. The Company paid an upfront fee of 2% of the aggregate amount of the Loans on the Hayfin Loan Closing Date. The DD TL is subject to a commitment fee of 1% of the undrawn DD TL commitments, payable quarterly in arrears on the first day of each fiscal quarter.
Mandatory Prepayments. A mandatory prepayment of the Loans is required upon (i) the incurrence of any indebtedness in breach of the Hayfin Loan Agreement, in an amount equal to 100% of the proceeds of such indebtedness, (ii) the occurrence of an event of default under the Hayfin Loan Agreement that results in an acceleration of the Loans, in an amount equal to the portion of the Loans accelerated together with all related outstanding amounts under the Hayfin Loan Agreement, (iii) a change of control, in an amount equal to the aggregate Loans together with all related outstanding amounts under the Hayfin Loan Agreement, and (iv) the receipt of proceeds for certain asset dispositions or insurance events, in an amount equal to the net proceeds thereof. In addition, 50% of Excess Cash Flow, as defined in the Hayfin Loan Agreement, for any year is required to be applied to prepay the Loans, with step-downs to (i) 25% based on Total Net Leverage Ratio levels of less than 1.00:1.00, but greater than or equal to 0.50:1.00, and (ii) 0% based on Total Net Leverage Ratio levels of less than 0.50:1.00 as set out and defined in the Hayfin Loan Agreement.
Prepayment Penalties. The Hayfin Loan Agreement imposes the following penalties with respect to any voluntary prepayment and any mandatory prepayment (other than pursuant to the Excess Cash Flow sweep or resulting from insurance events and, with respect to disposal actions, only to the extent they relate to a sale of all or substantially all of the assets and properties of the Company and its subsidiaries): (i) make-whole during the first year after the Hayfin Loan Closing Date; (ii) 102% after the first year but on or before the end of the second year after the Hayfin Loan Closing Date; (iii) 101% after the second year but on or before the end of the third year after the Hayfin Loan Closing Date and (iii) par thereafter.
Representations; Covenants; Events of Default.
The Hayfin Loan Agreement contains customary representations and warranties by the Company and its subsidiaries, subject, in certain instances, to customary materiality, material adverse effect or knowledge qualifiers. The Hayfin Loan Agreement also contains (a) certain affirmative and financial covenants that impose certain reporting and/or performance obligations on the Company and its subsidiaries, including (i) maximum Total Net Leverage Ratio (as defined in the Hayfin Loan Agreement) of 5.0x through December 31, 2020, stepping down to 4.5x through June 30, 2021, stepping down to 4.0x through the Maturity Date, in each case tested quarterly; and (ii) minimum Liquidity (as defined in the Hayfin Loan Agreement) of $10 million, an at-all-times covenant tested monthly; (b) certain negative covenants that generally limit, subject to various exceptions, the Company and its subsidiaries from taking certain actions, including, without limitation, incurring indebtedness (including with respect to the incurrence of DD TL if the Total Net Leverage Ratio (pro forma for such DD TL) exceed 3.5x), making investments, incurring
87
liens, paying dividends and engaging in mergers and consolidations, sale and leaseback transactions and asset dispositions, and (c) customary events of default for financings of this type. The Loans and other obligations under the Hayfin Loan Agreement may be declared due and payable upon the occurrence and during the continuance of any event of default and become automatically due and payable upon the occurrence of customary bankruptcy or insolvency events of default.
The summary set forth above is not intended to be complete and is qualified in its entirety by reference to the full text of the Hayfin Loan Agreement, which is filed as Exhibit 10.36 to this Annual Report.
The Hayfin Loan Agreement contains representations and warranties by each of the Company and its subsidiaries that are parties to the Hayfin Loan Agreement, which were made only for purposes of that agreement and as of specified dates. The representations, warranties and covenants in the Hayfin Loan Agreement were made solely for the benefit of the lenders and agents parties to the Hayfin Loan Agreement; are subject to limitations agreed upon by the contracting parties, including being qualified by confidential disclosure schedules; may have been made for the purposes of allocating contractual risk between the parties to the Hayfin Loan Agreement instead of establishing these matters as facts; and are subject to standards of materiality applicable to the applicable contracting parties that may differ from those applicable to investors. Investors should not rely on the representations, warranties and covenants or any descriptions thereof as characterizations of the actual state of facts or condition of the Company or any of its subsidiaries or affiliates. Moreover, information concerning the subject matter of the representations, warranties and covenants may change after the date of the Hayfin Loan Agreement, which subsequent information may or may not be fully reflected in the Company’s public disclosures.
Repayment and Termination of the Blue Torch Loan Agreement
Item 1.02 Termination of a Material Definitive Agreement.
Item 2.03 Creation of a Direct Financial Obligation or an Obligation under an Off-Balance Sheet Arrangement of a Registrant.
Item 2.04 Trigger Events That Accelerate or Increase a Direct Financial Obligation or an Obligation under an Off-Balance Sheet Arrangement.
On July 2, 2020, the Company terminated the BT Loan Agreement and repaid the $72,013,859 outstanding balance of principal and accrued but unpaid interest under the BT Loan Agreement. As a result of the early repayment of the loans under the BT Loan Agreement, the Company also paid a prepayment premium in the amount of $1,439,438. The Company paid the outstanding balance, accrued but unpaid interest, and prepayment premium using a portion of the proceeds from the Preferred Stock Transaction and the Hayfin Loan Transaction.
Appointment of Two Directors
Item 5.02 Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers.
Effective July 2, 2020, pursuant to the terms of the Purchase Agreement and the Preferred Stock Amendment, the Company increased the size of the Board of Directors and appointed Martin P. Sutter and William A. Hawkins III to serve as Preferred Directors. It is expected that each will serve on at least one committee of the Board of Directors, which have yet to be determined. Each will have the same compensation arrangement as the Company’s other non-employee directors.
88
PART III
Item 10. Directors, Executive Officers and Corporate Governance
Board of Directors
Set forth below is certain information regarding our current directors. There are no family relationships among any of our directors or executive officers.
Name | Age | Since | Tenure | Independent | Committees | |||||
Richard J. Barry | 61 | 2019 | 1 | ü | CC* | |||||
M. Kathleen Behrens | 67 | 2019 | 1 | ü | COB, EC | |||||
James L. Bierman | 67 | 2019 | 1 | ü | AC, CC | |||||
J. Terry Dewberry | 76 | 2009 | 11 | ü | AC, NCG | |||||
Charles R. Evans | 73 | 2012 | 8 | ü | AC, NCG* | |||||
William A. Hawkins III | 66 | 2020 | NA | TBD | TBD | |||||
Charles E. Koob | 75 | 2008 | 12 | — | ||||||
K. Todd Newton | 57 | 2019 | 1 | ü | AC*, EC | |||||
Martin P. Sutter | 65 | 2020 | N/A | TBD | TBD | |||||
Timothy R. Wright | 62 | 2019 | 1 | — | ||||||
Neil S. Yeston | 77 | 2012 | 8 | ü | CC, EC*, SL | |||||
* = Chair; AC = Audit Committee; CC = Compensation Committee; COB = Chairperson of the Board; EC = Ethics & Compliance Committee; NCG = Nominating and Corporate Governance Committee; SL = Science and Research Liaison; TBD = to be determined.
Pursuant to the terms of the Purchase Agreement and the Preferred Stock Amendment, the Company increased the size of the Board of Directors and appointed Martin P. Sutter and William A. Hawkins III to serve as Preferred Directors effective July 2, 2020. It is expected that each will serve on at least one committee of the Board of Directors, which have yet to be determined. The Board has not yet made a determination regarding the independence under the Nasdaq listing standards for director independence with respect to Mr. Hawkins or Mr. Sutter.
Richard J. Barry, age 61. Mr. Barry has served as a director of Sarepta Therapeutics, Inc. (SRPT), a genetic medicine company, since June 2015, and he has been a Partner and Advisory Board member of the San Diego Padres since 2009. Earlier in his career, he was a founding member of Eastbourne Capital Management LLC, a large equity hedge fund investing in a variety of industries, including health care, and served as the Managing General Partner and Portfolio Manager from 1999 to its close in 2010. Prior to that, he was a Portfolio Manager and Managing Director of Robertson Stephens Investment Management, an investment company, from 1995 until 1999. Before that, Mr. Barry spent over 13 years in various roles in institutional equity and investment management firms, including Lazard Frères, Legg Mason and Merrill Lynch. Mr. Barry has served as a Managing Member of GSM Fund, LLC, a fund established for the sole purpose of investing in Elcelyx Therapeutics, and previously served as a director of Elcelyx Therapeutics, Inc., a private pharmaceutical company, from 2013 until 2019. Mr. Barry previously served as a director of Cluster Wireless, LLC, a software company, from 2011 until 2014, and of BlackLight Power, Inc. (n/k/a Brilliant Light Power, Inc.), an energy research company, from 2009 until 2010. Mr. Barry holds a B.A. from Pennsylvania State University and is a member of its Shreyer’s Honors College Advisory Board. Mr. Barry has served on the Board since June 2019 and was nominated as a director because of his substantial experience, including in the healthcare and biotechnology sectors.
M. Kathleen Behrens, Ph.D., age 67. Dr. Behrens has worked as an independent life sciences consultant and investor since December 2009. Dr. Behrens served as the Co-Founder, President and Chief Executive Officer, and as a director, of the KEW Group Inc., a private oncology services company, from January 2012 until June 2014. Earlier in her career, Dr. Behrens served as a general partner for selected venture funds for RS Investments, a mutual fund firm, from 1996 until December 2009. While Dr. Behrens worked at RS Investments, from 1996 to 2002, she served as a managing director at the firm and, from 2003 to December 2009, she served as a consultant to the firm. During that time, Dr. Behrens also served as a member of the President’s Council of Advisors on Science and Technology (PCAST) from 2001 to 2009 and as chairwoman of PCAST’s Subcommittee on Personalized Medicine, as well as the President, director and chairwoman of the National Venture Capital Association, an organization that advocates for public policy that supports the American entrepreneurial ecosystem, from 1993 until 2000. Prior to that, she served as a general partner and managing director for Robertson Stephens & Co., an investment company, from 1983 through 1996. Dr. Behrens has served as a member of the board of directors of each of Sarepta Therapeutics, Inc. (NASDAQ:
89
SRPT), a medical research and drug development company, since March 2009 (Chairwoman of the Board since April 2015) and IGM Biosciences, Inc. (NASDAQ: IGMS), a clinical stage biotechnology company focused on creating and developing IgM antibodies, since January 2019. She served as a director of Amylin Pharmaceuticals, Inc. (formerly NASDAQ: AMLN), a biopharmaceutical company, from 2009 until its sale in 2012 to Bristol-Myers Squibb Co. Prior to that, she served on the board of directors of Abgenix, Inc. (formerly NASDAQ: ABGX), a biopharmaceutical company, from 2001 until the company was sold to Amgen, Inc. in 2006. From 1997 to 2005, Dr. Behrens was a director of Science, Technology and Economic Policy for the National Research Council. Dr. Behrens was also a Co-Founder of the Coalition for 21st Century Medicine, a trade association for new generation diagnostics companies. Dr. Behrens holds a B.S. in biology and a Ph.D. in microbiology from the University of California, Davis. Dr. Behrens has served on the Board since June 2019 and was nominated as a director because of her substantial experience in the financial services and biotechnology sectors, as well as in healthcare policy.
James L. Bierman, age 67. Mr. Bierman served as President and Chief Executive Officer and as a member of the board of directors of Owens & Minor, Inc. (NYSE: OMI), a Fortune 500 company and a leading distributor of medical and surgical supplies, from September 2014 to June 2015. Previously, he served in various other senior roles at Owens & Minor, including President and Chief Operating Officer from August 2013 to September 2014, Executive Vice President and Chief Operating Officer from March 2012 to August 2013, Executive Vice President and Chief Financial Officer from April 2011 to March 2012, and Senior Vice President and Chief Financial Officer from June 2007 to April 2011. Earlier in his career Mr. Bierman served as Executive Vice President and Chief Financial Officer at Quintiles Transnational Corp. (formerly NASDAQ: QTRN). Quintiles was a market leader in providing product development and commercialization solutions to the pharmaceutical, biotech, and medical device industries. As a member of management, he helped lead the successful privatization of the company in 2004. Before joining Quintiles, Mr. Bierman was a partner with Arthur Andersen LLP from 1988 to 1998. Mr. Bierman currently serves on the board of directors of Tenet Healthcare Corporation (NYSE: THC), a Fortune 100 company and a diversified healthcare services company operating more than 500 facilities, acute care hospitals and outpatient centers, throughout the United States. He previously served on the board of directors of Team Health Holdings, Inc. (formerly NYSE: TMH) where as Independent Lead Director, he helped lead the successful privatization of the company in 2017. Team Health is one of the largest suppliers of outsourced healthcare professional staffing and administrative services to hospitals and other healthcare providers in the United States. Mr. Bierman earned his B.A. from Dickinson College and his M.B.A. at Cornell University’s Johnson Graduate School of Management. Mr. Bierman has served on the Board since June 2019 and was nominated as a director because of his substantial operational and financial experience in the healthcare sector.
J. Terry Dewberry, age 76. Mr. Dewberry is a private investor with significant experience at both the management and board levels in the healthcare industry. He has extensive experience in corporate mergers and takeovers on both the buy and sell sides for consideration up to $5 billion. Mr. Dewberry has served on the boards of directors of several publicly traded healthcare products and services companies, including Respironics, Inc. (Nasdaq: RESP) (1998-2008), Matria Healthcare, Inc. (Nasdaq: MATR) (2006-2008), Healthdyne Information Enterprises, Inc. (1996-2002), Healthdyne Technologies, Inc. (1993-1997), Home Nutritional Services, Inc. (1989-1994) and Healthdyne, Inc. (1981-1996). From March 1992 until March 1996, Mr. Dewberry was Vice Chairman of Healthdyne, Inc. From 1984 to 1992, he served as President and Chief Operating Officer, and Executive Vice President of Healthdyne, Inc. Mr. Dewberry received a Bachelor of Electrical Engineering from Georgia Institute of Technology in 1967 and a Master of Professional Accountancy from Georgia State University in 1972. Mr. Dewberry has served on the Board since 2009 and was nominated as a director due to his extensive business and financial background and experience as a member of the boards of directors of other publicly traded companies and a member of the audit committee of at least one other public company.
Charles R. Evans, age 73. The Board named Mr. Evans Lead Director on March 9, 2018, and he served as Chairman from July 2, 2018 through June 2019. Mr. Evans has over 40 years of experience in the healthcare industry. He is currently President of the International Health Services Group, an organization he founded to support health services development in underserved areas of the world. Since 2009, he has served as a senior adviser with Jackson Healthcare, a consortium of companies that provide physician and clinical staffing, anesthesia management and information technology solutions for hospitals, health systems and physician groups. In addition, Mr. Evans is a Fellow in the American College of Healthcare Executives having previously served as Governor of the College from 2004 to 2007 and as Chairman Officer from 2008 to 2011. In 2012, he attained the Board Leadership Fellow credential of the National Association of Corporate Directors. Previously, Mr. Evans was a senior officer with Hospital Corporation of America (HCA), having managed various HCA divisions and completing his service with the responsibility for operations in the Eastern half of the country. Mr. Evans currently serves on the board of directors of Jackson Healthcare and WellStreet Urgent Care. Mr. Evans also serves on the boards of nonprofit organizations including American International Health Alliance and FaithBridge Foster Care. Mr. Evans has served on the Board since 2012 and was nominated as a director due to his healthcare management expertise.
William A. Hawkins III, age 66. Mr. Hawkins serves as a Senior Advisor to EW Healthcare Partners, a life sciences private equity firm. Mr. Hawkins is the former Chairman and CEO of Medtronic, Inc., a global leader in medical technology. He was at
90
Medtronic from 2002 until 2011. After retiring from Medtronic, he served as President and Chief Executive Officer of Immucor, Inc., a private equity backed global leader in transfusion and transplant medicine from October 2011 to July 2015. From 1998 to 2001 Mr. Hawkins served as President and Chief Executive Officer of Novoste Corporation (NASDAQ:NOVST), an interventional cardiology company. Prior to that, Mr. Hawkins served in a variety of senior roles at American Home Products, a consumer, pharma and medical device company, Johnson & Johnson, a healthcare company, Guidant Corporation, a medical products company, and Eli Lilly and Company, a global pharmaceutical company. Mr. Hawkins also serves as a director of Biogen Inc. (NASDAQ: BIIB), a biopharmaceutical company; Avanos Medical, Inc. (NYSE:AVNS), a medical technology company; as Chairman of Bioventus, LLC; as Chairman of 4 Tech; and as a director of AskBio, Cirtec, Virtue Labs, Immucor, Inc., Cereius, Inc. and Baebies, Inc., all of which are life science companies. He previously served on the board of Thoratec Corporation. Mr. Hawkins is Vice Chair of the Duke University Board of Trustees and is Chair of the Duke University Health System. Mr. Hawkins was elected as a member of the AIMBE College of Fellows and the National Academy of Engineering. He has a dual B.S.E.E. degree in Electrical and Biomedical Engineering from Duke University and a M.B.A. from the University of Virginia’s Darden School of Business. Mr. Hawkins has significant leadership experience as a chief executive officer, significant knowledge of, and experience in, the healthcare industry and significant international experience. He also has extensive governance and public company board experience.
Charles E. (“Chuck”) Koob, age 75. In 2007, Mr. Koob retired as a partner in the law firm of Simpson Thacher & Bartlett, LLP. While at that firm, Mr. Koob was the co-head of the Litigation Department and served on the firm’s Executive Committee. Mr. Koob specialized in competition, trade regulation and antitrust issues. Throughout his 37-year tenure, he represented clients before the Federal Trade Commission, the Antitrust Division of the Department of Justice, and numerous state and foreign competition authorities. He received his B.A. from Rockhurst College in 1966 and his J.D. from Stanford Law School in 1969. Mr. Koob serves on the board of Stanford Hospital and Clinics. He previously served on the board of a private drug development company and MRI Interventions (OTCBB: MRIC), a publicly traded medical device company. Mr. Koob has served on the Board since 2008 and was nominated as a director due to his extensive legal expertise in representing both publicly traded and privately held businesses.
K. Todd Newton, age 57. Mr. Newton has served as Chief Executive Officer and as a member of the board of directors of Apollo Endosurgery, Inc. (NASDAQ: APEN), a medical device company, since July 2014. Earlier in his career, Mr. Newton served as Executive Vice President, Chief Financial Officer and Chief Operating Officer at ArthroCare Corporation (formerly NASDAQ: ARTC), a medical device company, from 2009 to June 2014. Prior to that, Mr. Newton served in a number of executive officer roles, including President and Chief Executive Officer and as a director, at Synenco Energy, Inc., a Canadian oil sands company, from 2004 until 2008. Mr. Newton was a Partner at Deloitte & Touche LLP, a professional services network and accounting organization, from 1994 to 2004. Mr. Newton holds a B.B.A. in accounting from The University of Texas at San Antonio. Mr. Newton has served on the Board since June 2019 and was nominated as a director because of his significant experience in the medical device sector as well as strong executive leadership experience.
Martin P. Sutter, age 65. Since 1985, Mr. Sutter has been the Co-Founder and a Managing Director of EW Healthcare Partners, previously known as Essex Woodlands Health Ventures, a healthcare-focused growth equity firm. Mr. Sutter has been directly involved with more than 30 of EW Healthcare Partners’ portfolio company investments. Educated in chemical engineering and finance, Mr. Sutter has more than 35 years of management experience in operations, marketing, finance and venture capital. Mr. Sutter holds a Bachelor of Science degree from Louisiana State University and a Master of Business Administration from the University of Houston. He currently serves on the Boards of Abiomed, Inc. (NASDAQ: ABMD), Bioventus LLC and Prolacta Bioscience. He previously served on the boards of directors of the following EW Healthcare Partners’ portfolio investments: ATS Medical (later acquired by Medtronic, Inc.); BioForm Medical (later acquired by Merz GmbH & Co KGaA); LifeCell (later acquired by Kinetic Concepts); St. Francis Medical (later acquired by Kyphon, Inc./Medtronic, Inc.); Confluent Surgical (later acquired by Tyco International/Covidien); and Rinat Neurosciences (later acquired by Pfizer, Inc.). We believe that Mr. Sutter’s in-depth knowledge of the medical device industry, his skills as an investor in developing medical device companies, his extensive board experience and his position as a representative of a large stockholder in our Company qualify him to serve as a member of our Board of Directors.
Timothy R. Wright, age 62, joined the Company as its Chief Executive Officer on May 13, 2019. Mr. Wright has more than 30 years of experience in the pharmaceutical, biotech and medical devices industries. Most recently, Mr. Wright served as a Partner at Signal Hill Advisors, LLC, a consulting practice, since February 2011. Mr. Wright served as President and Chief Executive Officer of M2Gen Corp., a privately held cancer and health informatics company, between July 2017 and September 2018. Prior to M2Gen Corp., Mr. Wright served as Executive Vice President, Mergers and Acquisitions, Strategy and Innovation for Teva Pharmaceutical Industries Ltd. (“Teva”), a pharmaceutical company specializing in generic medicines, from April 2015 until August 2017. Before Teva, Mr. Wright was the founding partner of The Ohio State University Comprehensive Cancer Drug Development Institute. Mr. Wright also served as Chairman, Interim Chief Executive Officer and a director of Curaxis Pharmaceutical Corporation (“Curaxis”), a pharmaceutical company specializing in the development of drugs for the treatment
91
of Alzheimer’s disease and various cancers, from July 2011 to July 2012. Curaxis had been experiencing financial difficulties prior to Mr. Wright’s tenure and, as a result, the company filed for Chapter 11 bankruptcy in July 2012. Mr. Wright has been a director of Agenus, Inc. (NASDAQ: AGEN), an immune oncology company, since 2006 and its lead director since 2009. Mr. Wright also serves as Chairperson of The Ohio State University Comprehensive Cancer Center Drug Development Institute, serves as director of The Ohio State Innovation Foundation and sits on The Ohio State University College of Pharmacy Dean’s Corporate Council. Over his career, Mr. Wright has served on boards of directors in North America, Europe and Asia. Mr. Wright earned a Bachelor’s of Science in Marketing from The Ohio State University. Mr. Wright has served on the Board since June 2019 and was nominated as a director to bring the perspective of the Chief Executive Officer on the Board and also for the benefit of his many years of experience in the healthcare and pharmaceutical industry.
Neil S. Yeston, M.D., age 77 Dr. Yeston is the Past President of the New England Surgical Society and currently serves as Active Senior Staff, Department of Surgery at Hartford Hospital. During his association with Hartford Hospital, Dr. Yeston previously served in various roles including Vice President of Academic Affairs, Director of Corporate Compliance, Vice President of Quality Management and Director of the Section on Critical Care Medicine, Department of Surgery. In addition, Dr. Yeston was responsible for the enterprise wide acquisition of all biomedical engineering technology. Dr. Yeston has formerly served as Professor of Surgery at the University of Connecticut and the Assistant Dean, Medical Education at the University of Connecticut School of Medicine. Prior to his associations with Hartford Hospital and the University of Connecticut, Dr. Yeston served in various positions with the Boston University Medical Center including the Vice Chairman of the Department of Surgery, Associate Professor of Anesthesiology, Director Progressive Care Unit, and Associate Professor of Surgery. Dr. Yeston has served on the Board since 2012 and was nominated as a director because of his in-depth understanding of healthcare issues from the perspective of the practitioner, academician, administrator and executive.
Audit Committee and Audit Committee Financial Expert
The following directors serve on the Audit Committee: K. Todd Newton (Chair), James L. Bierman, J. Terry Dewberry, and Charles R. Evans, each of whom satisfies NASDAQ’s independence standards for audit committee members. The Board has determined that each of Messrs. Bierman, Dewberry, and Newton is an “audit committee financial expert” as that term is defined by the SEC in Item 407(d)(5)(ii) of Regulation S-K.
Code of Business Conduct and Ethics
We have adopted a Code of Business Conduct and Ethics that applies to all of our employees, officers and directors, a copy of which is on our website at https://mimedx.gcs-web.com/corporate-governance/highlights. Any amendments to or waivers of the Code of Business Conduct and Ethics that require disclosure under applicable law or listing standards will be disclosed on our website at www.mimedx.com. We undertake to provide a copy to any person, without charge, upon written request to Secretary, MiMedx Group, Inc., 1775 West Oak Commons Court, NE Marietta, Georgia 30062.
Procedures by which Security Holders May Nominate Individuals for Election to the Board
To nominate a person for election as a director at an annual meeting of shareholders, the Company’s Amended and Restated Bylaws require that timely notice of the nomination in proper written form, including all required information as specified in the Amended and Restated Bylaws, be mailed to the Secretary, at 1775 West Oak Commons Court, NE, Marietta, Georgia 30062. The Nominating and Corporate Governance Committee will consider for nomination candidates recommended by shareholders on the same basis as candidates recommended by members of the Board or other sources. Any proposed director candidate shall satisfy the criteria for Board membership set forth in the charter of the Nominating and Corporate Governance Committee or otherwise approved by the Nominating and Corporate Governance Committee and the Board from time to time.
Cooperation Agreement
The Company entered into a Cooperation Agreement, dated as of May 29, 2019 (the “Cooperation Agreement”), with M. Kathleen Behrens, K. Todd Newton, Richard J. Barry, Prescience Partners, LP, a Delaware limited partnership (“Prescience Partners”), its affiliates and Eiad Asbahi (Prescience Partners, together with Prescience Point Special Opportunity LP, Prescience Capital, LLC, Prescience Investment Group, LLC d/b/a Prescience Point Capital Management LLC and Mr. Asbahi, “Prescience Point”; Prescience Point, Dr. Behrens, Mr. Barry and Mr. Newton collectively, the “Investor Group”). With certain exceptions relating to breaches of the Cooperation Agreement, the Cooperation Agreement terminates at least five business days after the Company or the Investor Group delivers notice of termination (the “Termination Date”) following the date of the 2020 Annual Meeting. Pursuant to the Cooperation Agreement, the Company nominated Dr. Behrens, Mr. Newton and Mr. Wright as three Class II director candidates for election to the Board at the 2018 Annual Meeting. The 2018 Annual Meeting was duly held on June 17, 2019, and Dr. Behrens, Mr. Newton, and Mr. Wright were elected to the Board. The Board also appointed Mr. Barry and
92
Mr. Bierman as Class III directors pursuant to the Cooperation Agreement. The Cooperation Agreement further provides for the Company and Prescience Point to identify and mutually agree upon an individual (the “Mutual Designee”) to stand for election as a Class III director at the 2019 Annual Meeting. As of the date of this Form 10-K, the Board and Prescience Point have yet to identify the Mutual Designee for election as Class III directors at the 2019 Annual Meeting (which will be held in 2020).
The Cooperation Agreement provides Prescience Point with certain other rights with respect to designating replacement Board nominees and with respect to the designated directors’ service on certain Board committees, as long as Prescience Point holds more than 5.0% of the outstanding shares of Common Stock. The Cooperation Agreement contains customary standstill restrictions, and through the Termination Date and subject to certain exceptions, Prescience Point is required to vote all of its shares of Common Stock at any annual or special meeting, and any consent solicitation of the Company’s shareholders, in accordance with the recommendations of the Board. Pursuant to the Cooperation Agreement, the Company reimbursed Prescience Point for $500,000 of its reasonable, documented out-of-pocket fees and expenses incurred in connection with the matters related to the 2018 Annual Meeting.
Executive Officers
The following persons currently serve as our executive officers:
Timothy R. Wright, 62, became the Company’s Chief Executive Officer in May 2019. The biography for Mr. Wright can be found under the heading “Board of Directors” above.
Peter M. Carlson, age 56, was appointed Chief Financial Officer in March 2020. He joined the Company as Executive Vice President - Finance in December 2019. From 2017 to 2018, Mr. Carlson served as Chief Operating Officer at Brighthouse Financial, Inc., where he helped establish the $200 billion (assets) U.S. life and annuity insurance company as a separate entity following its August 2017 spin-off from MetLife, Inc., one of the world’s leading financial services companies. He was the Chief Accounting Officer at MetLife, Inc. from 2009 to 2017 where his global responsibilities included accounting, financial planning, tax, and investment finance. Prior to joining MetLife in 2009, Carlson was the Corporate Controller at Wachovia Corporation. He currently serves as a director of White Mountains Insurance Company (NYSE: WTM). Mr. Carlson holds a Bachelor of Science from Wake Forest University and is a trustee of the university. He is licensed as a certified public accountant in North Carolina and New York.
Mark P. Graves, age 55, was appointed Chief Compliance Officer in July 2018. Prior to joining the Company, he served as the U.S. leader for the global Patient Experience & Value function in the neurology division of UCB, Inc., a biopharmaceutical company. From 2011 to 2015, he was UCB’s Deputy Compliance Officer involved in all aspects of compliance including the implementation and management of the company’s corporate integrity agreement. Prior to that, Graves was Senior Director in the Office of Ethics and Compliance for the Pharmaceutical Products Division of Abbott Laboratories, as well as Deputy Ethics & Compliance Officer for Takeda Pharmaceuticals North America, Inc. and TAP Pharmaceutical Products, Inc. Prior to his pharmaceutical and biotech career, he practiced labor and employment law. Mr. Graves holds a B.A. in Criminology and Law, and a J.D., from the University of Florida as well as an MBA from the University of Chicago Booth School of Business.
William F. “Butch” Hulse IV, age 47, has served as General Counsel since December 2019. Prior to joining the Company, Mr. Hulse was a member of Dykema Gossett, PLLC, a national law firm since 2017. Prior thereto, he was with Acelity, LP, Inc. (formerly Kinetic Concepts, Inc.), a global medical technology company with leadership positions in advanced wound care, surgical solutions and regenerative medicine, from 2008 to 2017 in a variety of roles of increasing responsibility. From 2013 to 2017, he served as Acelity’s Chief Compliance Officer and Senior Vice President for Enterprise Risk Management, Quality, and Regulatory. Prior to that, he served as Division General Counsel for Acelity’s advanced wound care business unit and as Associate General Counsel for litigation matters. Mr. Hulse holds a Bachelor of Arts from Angelo State University and a J.D. from the Baylor University School of Law.
Scott M. Turner, age 55, has served as Senior Vice President, Operations and Procurement since April 2017. Mr. Turner oversees supply chain including donor recovery services, procurement, processing, and facilities. Mr. Turner joined the Company in April 2016 as Vice President, Procurement. Prior to joining the Company, Mr. Turner served as a director with Alvarez & Marsal North America, LLC in their Corporate Performance Improvement group from October 2015 until March 2016. Prior thereto, Mr. Turner served as Vice President, Supply Chain, with Larson-Juhl, a Berkshire Hathaway company, from June 2013 until September 2015. Additionally, Mr. Turner has more than 20 years of Supply Chain and Procurement leadership in life sciences at Shionogi and Johnson & Johnson, spanning the consumer, medical device, and pharmaceutical sectors domestically and overseas. Mr. Turner holds a Bachelor of Science in Commerce & Engineering from Drexel University and a President / Key Executives MBA from Pepperdine University.
93
Item 11. Executive Compensation
COMPENSATION DISCUSSION AND ANALYSIS
The Compensation Committee is responsible for evaluating and determining the compensation paid to the executive officers who are listed in the Summary Compensation Table (the “NEOs”). All components of compensation for the NEOs are then recommended by the Compensation Committee for approval by the Board. This Compensation Discussion and Analysis (“CD&A”) pertains to 2019 compensation.
For 2019, the Company’s NEOs were:
• | Timothy R. Wright. Mr. Wright joined MiMedx as Chief Executive Officer on May 13, 2019. |
• | David Coles. Mr. Coles served as Interim Chief Executive Officer from July 2, 2018 until May 13, 2019. He was an employee of Alvarez & Marsal North America, LLC. We paid Alvarez & Marsal for Mr. Cole’s services, as described below under “Agreements with Our Executive Officers-Agreement with Alvarez & Marsal to Employ Mr. Coles.” |
• | Edward Borkowski. Mr. Borkowski served as Executive Vice President and Interim Chief Financial Officer from June 7, 2018 until his resignation on November 15, 2019. Subsequently, he served as Acting Chief Financial Officer through March 17, 2020 pursuant to a Separation and Transition Services Agreement, as described below under “Agreements with Our Executive Officers - Agreement with Mr. Borkowski.” |
• | Peter M. Carlson. Mr. Carlson joined the Company as Executive Vice President - Finance in December 2019. He became Chief Financial Officer in March 2020. |
• | I. Mark Landy. Mr. Landy served as Executive Vice President and Chief Strategy Officer from December 5, 2018 until the Company eliminated this role effective September 16, 2019 (which terminated his employment). |
• | Scott M. Turner. Mr. Turner has served as Senior Vice President—Operations & Procurement since December 5, 2018 and continues to serve in such role. |
Prior Say-on-Pay Proposal and Shareholder Support
The Company conducted an advisory say-on-pay vote at the 2016 annual meeting of shareholders, where approximately 95% of the votes cast were in favor of the proposal. The Board and Compensation Committee reviewed these final vote results together with the other factors and data discussed in this Compensation Discussion and Analysis and determined that, given the significant level of support of the Company’s approach to compensation by its shareholders, no changes to its executive compensation policies and related decisions were necessary at such time.
The next shareholder vote with respect to say-on-pay and the frequency of the say-on-pay vote will occur at the 2019 Annual Meeting, which will be held in 2020. The Board intends to recommend annual say-on-pay votes to allow for more timely shareholder feedback.
Compensation Philosophy
MiMedx’s executive compensation philosophy is based on the belief that competitive compensation is essential to attract and retain highly-qualified executives and incentivize them to achieve the Company’s operational and financial goals. In line with this philosophy, the Company’s practice is to provide total compensation that is competitive with comparable positions at peer organizations. The compensation program is based on individual and organizational performance and includes components that reinforce the Company’s incentive and retention-related compensation objectives.
The principal components of compensation for MiMedx’s NEOs are base salary, annual cash incentives and long-term equity incentives. Cash incentives are included to encourage and reward effective performance relative to the Company’s near-term plans and objectives. Equity incentives are included to promote longer-term focus, to help retain key contributors and to align the interests of the Company’s executives and shareholders.
Pay-Setting Process
94
Compensation Consultant
Beginning in mid-2018, the Compensation Committee engaged an independent executive compensation consulting firm, Meridian Compensation Partners, LLC (“Meridian”), to provide compensation consulting services relating to (1) NEO compensation, (2) peer group composition and practices, (3) incentives design, (4) compensation governance, (5) amount and form of director compensation and (6) alternatives to equity compensation. Meridian’s services were provided only to the Compensation Committee, and the Compensation Committee determined that Meridian’s work did not raise any conflict of interest.
In October, 2019, following changes in the membership of the Compensation Committee and the Board, the Compensation Committee engaged a new, independent executive compensation consulting firm, Aon Consulting, Inc. through its Radford subdivision (“Radford”), to replace Meridian and to provide compensation consulting services relating to (1) NEO compensation, (2) peer group composition and practices, (3) incentives design, (4) compensation governance, (5) amount and form of director compensation and (6) alternatives to equity compensation. Radford’s services were provided only to the Compensation Committee, and the Compensation Committee determined that Radford’s work did not raise any conflict of interest.
Use of a Peer Group
In making compensation decisions, the Compensation Committee has considered the recommendations of the CEO and of a senior HR executive, which, in turn, have been informed by a compensation analysis of the practices of peer group companies, which are publicly-traded companies in the medical device, pharmaceuticals, biotechnology and life sciences sectors of the healthcare industry. The peer group was determined primarily using organizational criteria, revenue, market capitalization, and industry sector. Organizational criteria include number of employees as well as qualitative factors such as industry, markets, and development stage. The data from the peer group companies for the NEOs provides the Compensation Committee with a benchmark that it views as a point of reference, but not as a determining factor, for the compensation of the NEOs. In 2019, the Company’s peer group was as follows:
Abiomed, Inc. | Geron Corporation | LivaNova PLC | ||
Acorda Therapeutics, Inc. | Halozyme Therapeutics, Inc. | Momenta Pharmaceuticals, Inc. | ||
AMAG Pharmaceuticals, Inc. | ImmunoGen, Inc. | Newlink Genetics Corp. | ||
Array BioPharma, Inc. | Infinity Pharmaceuticals, Inc. | OPKO Health, Inc. | ||
CryoLife, Inc. | Insulet Corporation | Osiris Therapeutics, Inc. | ||
DexCom, Inc. | Insys Therapeutics, Inc. | Seattle Genetics, Inc. | ||
Exelixis, Inc | Ionis Pharmaceuticals, Inc. | Spectrum Pharmaceuticals, Inc. | ||
Genomic Health, Inc. | Ironwood Pharmaceuticals, Inc. | Vanda Pharmaceuticals, Inc. | ||
Wright Medical Group, Inc. | ||||
In order to compete effectively for top executive-level talent, the Compensation Committee generally targets cash compensation for the NEOs between the 50th and 60th percentile and long-term equity compensation between the 60th and 75th percentile of compensation paid to similarly-situated executives of the companies comprising the peer group. However, in practice and in the case of 2019, total compensation actually awarded by the Compensation Committee has generally lagged these targets primarily due to the award of below-median long-term incentives. Although peer data and compensation survey data are useful guides for comparative purposes, the Compensation Committee believes that a successful compensation program also requires the application of judgment and subjective determinations of individual performance. In that regard, the Compensation Committee applies its judgment in reconciling the program’s objectives with the realities of attracting and retaining key employees.
2019 Compensation Components
Base Salaries
MiMedx employees, including its NEOs, are paid a base salary commensurate with the responsibilities of their positions, the skills and experience required for the position, their individual performance, business performance, labor market conditions, and with reference to peer company salary levels. Base salaries may be increased depending on the compensation of comparable positions within the peer group companies and published compensation surveys, the executive’s responsibilities, skills, expertise, experience and performance, the executive’s contributions to the Company’s results, and the overall performance of the Company compared to its peer group and other participants within the industry. In determining the increases, the Compensation Committee relies on judgment about each individual, as well as on recommendations from its compensation consultant and senior management, rather than applying a stated formula. Base salaries to the NEOs in 2019 were as follows:
95
NEO | Base Salary | Revised Base Salary(1) |
Mr. Wright | $750,000 | n/a |
Mr. Borkowski | $550,000 | $600,000 |
Mr. Carlson | $525,000 | n/a |
Mr. Landy | $425,000 | $455,000 |
Mr. Turner | $340,000 | $355,000 |
(1) | During 2019, in recognition of Mr. Borkowski’s assumption of the duties of Interim Chief Financial Officer, the Board increased Mr. Borkowski’s base salary to $600,000. The Board also increased Mr. Landy’s base salary to $455,000 and Mr. Turner’s to $355,000 during 2019 following their assumption of increased responsibilities. |
Annual Non-Equity Incentive Awards
Historically, the Company has adopted an annual non-equity incentive plan in which the NEOs participate. Through this plan, the Company delivers a target bonus opportunity expressed as a percentage of each executive’s base salary as shown below. During 2019, following Mr. Borkowski’s assumption of the duties of Interim Chief Financial Officer, the Board increased Mr. Borkowski’s target annual incentive from 60% to 65% of his base salary.
NEO | Base Salary | Target Annual Incentive as a Percent of Base Salary | Target Annual Incentive | Bonus Paid in 2019 |
Mr. Wright | $750,000 | 100% | $750,000 | $720,000 |
Mr. Borkowski | $600,000 | 65% | $390,000 | n/a(1) |
Mr. Carlson | $525,000 | 55% | $288,750 | n/a(2) |
Mr. Landy | $455,000 | 50% | $227,500 | n/a(3) |
Mr. Turner | $355,000 | 40% | $142,000 | $136,320 |
(1) | The Company made payments to Mr. Borkowski pursuant to Separation and Transition Services Agreement in lieu of, among other things, his annual incentive. |
(2) | Mr. Carlson joined the Company effective December 16, 2019 and therefore was not eligible for an annual incentive for 2019. |
(3) | The Company eliminated Mr. Landy’s role during 2019 and made payments to him in 2020 equal to one times his base salary and target annual incentive. |
However, 2019 was a year of rapid and significant change for the Company. Ultimately, the Board did not approve an annual incentive plan for 2019 due to the following factors:
• | a rapidly changing financial forecast following adverse insurance coverage decisions relating to the Company’s products in late 2018 and the reduction in force in December 2018; |
• | the failure to complete the audit of the financial statements for the year ended December 31, 2018, which also affected the Company’s ability to establish meaningful quantitative goals for 2019; |
• | the adoption of a new strategic plan which addressed the changing regulatory landscape for several of the Company’s products and investments related to future BLA products; and |
•changes in several of the Company’s key officers, including its CEO and CFO.
Nevertheless, the Board determined that it was important to grant bonuses for 2019 in recognition of extraordinary efforts during the year, to retain key leaders during a period of significant change and risk, and for internal pay equity. For 2019, the Board authorized the Company to pay discretionary bonuses to each of the NEOs who was still employed by the Company at the end of the year equal to 96% of each NEO’s target annual incentive as follows: Mr. Wright - $720,000; and Mr. Turner - $136,320. Mr. Carlson joined the Company on December 15, 2019 and was not eligible for an annual incentive award in 2019.
For 2020, the Committee and the Board have adopted a managing incentive plan (“MIP”), which is an annual cash incentive plan designed to incentivize and reward achievement of the current year’s financial and operational goals with three equally-weighted performance criteria - revenue, Adjusted EBITDA, and individual performance goals. Potential payouts under the 2020 MIP are capped at 1.5 times an executive’s target bonus.
Long-Term Equity Incentives
96
All equity incentive awards are granted under the Company’s 2016 Equity and Cash Incentive Plan (the “2016 Plan”), which was approved by shareholders in 2016. The 2016 Plan is designed to align the interests of the Company’s Named Executive Officers and other MiMedx officers, members of management and key employees with the interests of the Company’s shareholders, and serve as a key retention tool. Restricted stock vests over a period of time, generally pro rata annually over three years. The Company generally makes its an annual equity grant to a broad group of its management employees, including the NEOs in February or March of each year. The Company also typically grants restricted stock to certain newly-hired executive officers in connection with the commencement of their employment by the Company.
The Committee believes that equity grants are a positive motivator for the Company’s officers, management and key employees to focus their strategy and efforts on the Company’s long-term goals. Working toward the long-term growth of the price of the Company’s stock produces the ultimate financial gain for the executives’ equity awards and increase in value for the Company’s shareholders.
In recent years, the Compensation Committee has granted only restricted stock awards, rather than a mix of stock and stock options to conserve the number of shares available under the 2016 Plan. The Compensation Committee believes that restricted stock awards are an effective form of equity compensation because the vesting period is a strong retention tool for NEOs and other key executives. Restricted stock awards increase in value as the Company’s stock price increases over time, but they also continue to have value in the event of a stock price decline. Thus, unlike stock options, restricted stock does not lose its retention value in the event of a decline in stock price.
All awards of restricted stock granted to Named Executive Officers in 2019 were approved by the Compensation Committee for recommendation to the full Board for approval. All awards of restricted stock granted to all other eligible participants in the 2016 Plan were determined and approved by the Compensation Committee.
In determining the approved level of equity grants, the Compensation Committee considers the individual’s target annual long-term incentive value, the Company’s overall option “overhang,” the employee’s level of responsibility and performance, prior equity awards, comparative compensation information, and the anticipated expense to the Company. For 2019, all awards of restricted stock were dated and priced as follows:
• | All awards of restricted stock to current employees were granted and priced as of the close of the business day on which the Committee approved the grant. |
• | All awards of restricted stock granted to newly-hired employees were granted and priced as of the later of the business day on which the Board approved such grants or the date of employment. |
The Committee establishes vesting schedules for awards under the 2016 Plan at the time of the grant. To optimize the retention value of the awards and to orient recipients to the achievement of longer-term goals, objectives and success, awards typically vest in three equal installments on the first, second and third anniversaries of the Grant Date. The Company generally makes its an annual equity grant to a broad group of its management employees, including the Named Executive Officers, in February or March of each year. In 2019, all equity-based awards were issued under plans previously approved by the Company’s shareholders.
2019 Restricted Stock Grants to Named Executive Officers
The Compensation Committee’s philosophy with respect to annual grants is to benchmark long-term equity incentive awards at the 60th to 75th percentile of awards to similarly-situated executives of companies in the peer group. However, the actual amount of equity awards granted to the NEOs in 2019 was less than the benchmark target grant value in order to conserve the number of shares available for awards under the 2016 Plan. In general, in determining the level of equity grants, the Compensation Committee considers the individual’s target annual long-term incentive value, the Company’s unexercised and unvested grants, the employee’s level of responsibility and performance, prior equity awards, comparative compensation information, and the anticipated expense to the Company.
Grants to Current Officers
On February 21, 2019, the Company granted Mr. Borkowski 203,305 shares of restricted stock that were required to be granted to him pursuant to the Company’s agreement with him. On April 26, 2019, the Company granted each of Messrs. Landy and Turner 279,271 and 52,067 shares of restricted stock, respectively. It made no grant to its then-CEO, Mr. Coles, because he was an employee of Alvarez & Marsal. The grant to Mr. Turner vest pro rata annually over three years.
Grants to Newly-Hired Officers
97
In addition to the annual grants described above, the Company made certain grants of restricted stock to newly-hired employees during 2019. On May 6, 2019, the Company granted Mr. Wright 681,818 shares of restricted stock upon his appointment as Chief Executive Officer, scheduled to vest pro rata annually over three years; refer to the discussion “Agreement with Mr. Wright” below. In addition, in connection with the commencement of Mr. Carlson’s employment with the Company, the Company made a $350,000 restricted stock grant to Mr. Carlson on December 16, 2019 that vests pro rata annually over three years, and a $1 million restricted stock grant that vests upon the achievement of each of four discrete performance goals; refer to the discussion “Agreement with Mr. Carlson” below. Each of these awards will be settled in a number of shares of common stock based on our stock 30 days after the Company first becomes current with its SEC reporting obligations.
Agreements with our Executive Officers
Agreement with Alvarez & Marsal to employ Mr. Coles
The Board appointed Mr. Coles as Interim Chief Executive Officer of the Company, effective as of July 2, 2018. In connection with his appointment, the Company entered into an engagement letter with Alvarez & Marsal North America, LLC (“A&M”), where Mr. Coles had been employed since 1997, providing for Mr. Coles’ services and the services of additional A&M employees as needed to assist Mr. Coles in the execution of his duties. Under the terms of the engagement letter, during his service at the Company, Mr. Coles continued to be employed by A&M and was not entitled to receive any compensation directly from the Company or participate in any of the Company’s employee benefit plans. The Company instead paid A&M an hourly rate of $975 per hour for Mr. Coles’ services. In 2019, the Company paid A&M $908,663 for Mr. Coles’ services. Mr. Coles resigned on May 13, 2019 upon the hiring of Mr. Wright as our permanent Chief Executive Officer.
Agreement with Mr. Wright
In connection with his appointment as Chief Executive Officer in May 2019, Mr. Wright entered into a letter agreement (the “Letter Agreement”) with the Company that provides for an annual base salary of $750,000. The Company agreed in the Letter Agreement that he will be eligible to participate in the MIP with an annual target cash bonus amount equal to one hundred percent (100%) of his base salary. The Letter Agreement also provided for a special one-time signing bonus of $500,000, which was subject to repayment in full in the event that Mr. Wright resigned without “good reason” or had his employment terminated by the Company for “cause,” in each case within 12 months following the commencement of his employment with the Company. The Letter Agreement also provides that Mr. Wright’s MIP bonus would not be prorated for 2019, and that the Compensation Committee of the Board had approved and recommended to the Board for approval a minimum payout of not less than fifty percent (50%) of what his target bonus would have been if the Board had adopted the 2019 MIP. For 2019, the Company paid Mr. Wright a discretionary bonus in lieu of his target MIP bonus, as discussed above under “Annual Non-Equity Incentive Awards.”
In addition, pursuant to the Letter Agreement, the Company granted Mr. Wright a restricted stock award with a value of $3,375,000 as of the date that Mr. Wright commenced employment with the Company, which vests pro rata annually over three years and is subject to the terms and conditions of the 2016 Plan. In addition, the Letter Agreement provides that, following 2019, Mr. Wright will have a target long-term incentive award in an amount equal to four hundred and fifty percent (450%) of his then-current annual base salary.
The Letter Agreement further provided that in the event of the termination of Mr. Wright’s employment by the Company other than for “cause” or by Mr. Wright for “good reason,” Mr. Wright will be eligible to receive the following, subject to the execution and non-revocation of a release of claims (and continued compliance with any applicable restrictive covenant obligations): (i) a severance payment equal to 24 months of his then-current annual base salary plus two times his then-current annual target bonus amount and (ii) provided that Mr. Wright timely elects continued coverage under COBRA, continued participation in applicable Company benefit plans for him and his eligible dependents at active employee rates for 24 months following the termination of Mr. Wright’s employment. Notwithstanding the foregoing, in the event that Mr. Wright’s employment with the Company is terminated following a “change in control” for reasons other than death, disability, retirement, termination by the Company for “cause” or termination by Mr. Wright without “good reason,” Mr. Wright will be eligible to receive the following, subject to the execution and non-revocation of a release of claims (and continued compliance with any applicable restrictive covenant obligations): (i) a severance payment equal to 30 months of his then-current annual base salary plus 2.5 times his then-current annual target bonus amount, (ii) provided that Mr. Wright timely elects continued coverage under COBRA, continued participation in applicable Company benefit plans for him and his eligible dependents at active employee rates for 30 months following the termination of Mr. Wright’s employment and (iii) continued participation in life or other similar insurance or death benefit plans (excluding short-term or long-term disability insurance) for 30 months following the termination of Mr. Wright’s employment and at the Company’s expense.
98
The Letter Agreement also entitles Mr. Wright to certain relocation and commuting benefits.
Agreements with Mr. Borkowski
The Board appointed Mr. Borkowski, an Executive Vice President of the Company, as interim Chief Financial Officer effective June 6, 2018. In 2018, Mr. Borkowski received an annual salary of $550,000 and a target annual performance bonus of 60% of his base salary. The Board increased his salary and target bonus to $600,000 and 65%, respectively, during 2019.
The Company awarded Mr. Borkowski two restricted stock grants on February 21, 2019: one for 100,000 shares, one-third of which vested immediately and the other two-thirds were to vest ratably over a two-year period from the date of grant; and the other for 103,305 shares was to vest ratably over a two-year period from the date of grant. These awards were contemplated, but not granted, at the time Mr. Borkowski joined the Company. The Company made these grants with an abbreviated vesting schedule to approximate the result as if they had been granted as originally agreed because the grants were made nearly a year later than agreed.
In addition, the Company agreed to provide Mr. Borkowski severance, both in connection with a change in control and other than in connection with a change in control. The Company entered into a double-trigger Change in Control Severance Agreement with Mr. Borkowski, which provided for severance payments equal to 1.75 times his base salary and target bonus; and continuation of benefits for the period for which the severance is computed. The Company also entered into a severance agreement with Mr. Borkowski that was not conditioned upon a change in control and which provided for severance payments equal to 1.0 times his annual base salary plus target bonus, plus continuation of benefits for the period for which the severance is computed, if his employment was terminated for qualifying reasons. Mr. Borkowski was also eligible for relocation benefits.
On November 18, 2019, the Company entered into a Separation and Transition Services Agreement (the “Transition Agreement”) with Mr. Borkowski pursuant to which (i) he resigned as Executive Vice President and Interim Chief Financial Officer of the Company, as well as from any and all officer, director or other positions that he held with the Company and its affiliates, effective November 15, 2019, (ii) he agreed to perform the duties of the Acting Chief Financial Officer with respect to filing the 2018 Form 10-K and assist with the transition of his duties, and (iii) until March 31, 2020, he agreed to provide services as may be requested by the Company with respect to matters related to the 2018 Form 10-K and the Company’s Annual Report on Form 10-K for the fiscal year ending December 31, 2019. The Agreement provided for the Company to make special payments to Mr. Borkowski in installments as follows: (i) $1,700,000, which was paid within seven business days following the Transition Agreement, (ii) $1,750,000 which was paid following the filing of the 2018 Form 10-K with the SEC; and (iii) after March 31, 2020, $500,000 which was paid following the execution and delivery of a supplemental release by Mr. Borkowski. These payments were, among other things, in lieu of his equity grant and annual incentive for 2019. Mr. Borkowski forfeited all restricted stock owned by him which had not already vested, and all other claims to stock and other benefits. The Agreement also includes terms and conditions governing the Company’s and Mr. Borkowski’s general release of claims and other customary provisions.
Agreement with Mr. Carlson
The Company entered into an agreement with Mr. Carlson effective December 16, 2019 to employ him as Executive Vice President - Finance. The Company later named Mr. Carlson Chief Financial Officer effective March 18, 2020. Pursuant to the Company’s agreement with Mr. Carlson, he receives an annual base salary of $525,000 and will be eligible for a target annual incentive of fifty-five percent (55%) of his base salary and a target long-term incentive equal to two hundred percent (200%) of his base salary. In addition, he received (i) a special one-time signing bonus of $50,000 (which is subject to repayment in full in the event that he resigns or has his employment terminated by the Company within 12 months following the commencement of his employment with the Company), (ii) a restricted stock grant with a value of $350,000 which vests pro rata annually over three years, and (iii) a restricted stock grant with a value of $1,000,000, which vests upon the achievement of each of four discrete performance goals.
Agreement with Mr. Landy
On September 16, 2019, the Company eliminated the position of Chief Strategy Officer and terminated the employment of Mr. Landy without cause. Effective April 23, 2020, the Company entered into a Termination Agreement with Mr. Landy pursuant to which the Company will pay Mr. Landy twelve (12) months of his salary ($425,000) and target bonus (50%) that was in effect on the day his position was eliminated.
Additional Compensation Practices and Policies
Perquisites
99
The Company generally does not provide executive officers with perquisites and other personal benefits beyond the Company benefits offered to similarly situated employees, with the following exception: when the Company hosts performance incentive trips for its best-performing sales people, it requires certain executives to attend and assumes the incremental cost if the executive’s spouse attends, and when this occurs the Company reports the aggregate incremental travel expenses of the spouse as a perquisite. Also, during the Company’s transition, when its ability to attract and retain executives was reduced, the Company agreed to reimburse certain executives (Messrs. Wright and Borkowski) for commuting and transportation expenses between their respective homes and our corporate headquarters, temporary lodging, relocation and rental car expenses, and paid a tax-gross up on these amounts.
Stock Ownership Guidelines
The Board has adopted stock ownership guidelines that apply to the NEOs. Under the guidelines, covered persons are required to own stock, including unvested time-based restricted stock, equal to certain multiples of their annual cash compensation:
Person Subject to Policy | Requirement | ||||
CEO | 3.0X | ||||
CFO | 2.0X | ||||
General Counsel | 1.5X | ||||
Until such time as the NEO reaches his or her applicable threshold and subject to certain exceptions, the NEOs are required to hold 100% of the shares of Common Stock awarded to him/her from the Company or received upon vesting of restricted stock and upon exercise of stock options (net of any shares utilized to pay for tax withholding and any exercise price).
However, the Board has suspended the stock ownership guidelines until the Company becomes current in its SEC reporting obligations since subject persons may be prohibited by applicable insider trading laws from buying or selling Company securities. We expect to implement similar requirements once the Company’s officers are permitted to buy Company stock.
Recoupment of Compensation
The Board adopted a recoupment (clawback) policy, effective April 1, 2016, covering executive officers of the Company. The policy provides that if the Company is required to restate its financial results due to material noncompliance with financial reporting requirements under the securities laws, the Compensation Committee may seek reimbursement of any cash or equity-based bonus or other incentive compensation paid or awarded to the officer or effect cancellation of previously granted equity awards to the extent the bonus or incentive compensation was based on erroneous financial data and was in excess of what would have been paid to the officer under the restatement.
With the completion of the restatement of Company’s previously issued consolidated financial statements and financial information, the Compensation Committee has reviewed the annual non-equity incentive awards paid to executive officers based on financial performance for the years 2015 and 2016, and the amounts that would have been paid to such officers under the restated financial statements. In addition, the Compensation Committee has reviewed the annual non-equity incentive awards paid to executive officers for 2017 and 2018 (which had never been published and therefore technically not restated), and the amounts that would have been paid to such officers under the corrected financial statements. This review determined that the Company paid annual non-equity incentive awards between 2015 and 2018 to the following persons in excess of what would have been paid to such executive officers under the restated or revised financial metrics, by the following, aggregate amounts: our former Chief Executive Officer, Mr. Petit - $468,504; our former Chief Financial Officer, Mr. Senken - $215,550; our former President, Mr. Taylor - $356,555; our former General Counsel, Ms. Haden - $183,725; our former Interim Chief Financial Officer, Mr. Borkowski - $88,000; our former Chief Strategy Officer, Mr. Landy - $31,267; and Mr. Turner - $28,933. (The Company did not grant any equity awards based on incorrect financial metrics.)
The Compensation Committee notes that the Company effectively recovered $26.3 million of vested, unexercised options and unvested restricted stock as a result of the Board’s determination in September 2018 that the terminations of employment of Messrs. Petit, Senken and Taylor were “for cause,” which resulted in the forfeiture of those awards. Under the Plans, all unvested restricted stock awards and vested and unvested stock option awards were forfeited, as follows:
100
Former NEO | Options Forfeited | Value on 9/20/2018 at $6.20 per share | Unvested Restricted Stock Forfeited | Value on 9/20/2018 at $6.20 per share | Total Value of Equity Forfeited | |||||||
Petit | 2,867,820 | $12.1 million | 361,667 | $2.2 million | $14.3 million | |||||||
Senken | 887,107 | $3.7 million | 120,368 | $0.7 million | $4.4 million | |||||||
Taylor | 1,558,221 | $6.2 million | 229,234 | $1.4 million | $7.6 million | |||||||
TOTAL | 5,313,148 | $22.0 million | 711,269 | $4.3 million | $26.3 million | |||||||
(The value of forfeited options is based on the closing price of Common Stock on the date of forfeiture, which was $6.20 per share on September 20, 2018, less the exercise price. The value of forfeited restricted stock is based on the closing price of Common Stock on the date of forfeiture.)
The Compensation Committee also notes that on November 26, 2019, the SEC filed suit against Messrs. Petit, Senken and Taylor in the U.S. District Court for the Southern District of New York, including claims for relief as to Messrs. Petit and Senken for the disgorgement of all bonuses and all incentive-based and equity-based compensation pursuant to Section 304 of the Sarbanes-Oxley Act of 2002, among other claims for relief. The Committee further notes that Messrs. Landy and Turner only became executive officers in December 2018 and therefore were subject to the policy for less than one month.
In view of the pending criminal trials against Messrs. Petit and Taylor, and the SEC’s civil claims against Messrs. Petit, Taylor, and Senken, the Compensation Committee has not yet reached a final determination as to whether or how to recoup the amounts previously paid to these executives or to the other executives.
Anti-Hedging and Anti-Pledging Policies
Hedging transactions may permit the ownership of Company securities without the full risks and rewards of ownership. If a director, officer or employee engages in hedging transactions with respect to Company securities, he or she may no longer have the same objectives as the Company’s other shareholders. For this reason, the Company prohibits directors, officers and employees from engaging in hedging transactions in Company securities, subject to exceptions that may be granted in the sole discretion of the Company’s General Counsel in limited circumstances.
Securities held in a margin account as collateral for a margin loan may be sold by the broker without the customer’s consent if the customer fails to meet a margin call. Similarly, securities pledged as collateral for a loan may be sold if the borrower defaults on the loan, including at a time when the pledgor is aware of material nonpublic information or otherwise is not permitted to trade in Company securities. For these reasons, the Company prohibits directors, officers and other employees from holding Company securities in a margin account or otherwise pledging Company securities as collateral for a loan.
Compensation Risk Assessment
On an ongoing basis, the Compensation Committee considers the risks inherent in the Company’s compensation programs. With the change in the structure of the annual non-equity incentive compensation awards in late 2018, which de-emphasized revenue, the Compensation Committee believes that our compensation policies and practices do not encourage excessive and unnecessary risk-taking, and that the level of risk that they do encourage is not reasonably likely to have a material adverse effect on the Company. The Compensation Committee believes that the design of our compensation policies and practices encourages our employees to remain focused on both our short- and long-term goals.
101
COMPENSATION COMMITTEE REPORT
The Compensation Committee has reviewed the Compensation Discussion and Analysis in this Annual Report and discussed it with management. Based on its review and discussions with management, the Compensation Committee recommended to the Board that the Compensation Discussion and Analysis be included in this Annual Report and in the proxy statement for the Company’s 2019 annual meeting of shareholders. This report is provided by the following independent directors, who comprise the Compensation Committee:
Richard J. Barry, Chair (member of the Committee since June 2019) |
James L. Bierman (member of the Committee since July 2019) |
Neil S. Yeston (member of the Committee since September 2012) |
July 6, 2020
CEO Pay Ratio
In 2019, we paid total annual compensation to our median employee of $62,995. The annual total compensation of our CEO in 2019, as reported in the Summary Compensation Table, was $5,069,353. Based on this information, for 2019 the ratio of the annual total compensation of our CEO to the median annual total compensation of all employees was 80 to 1. (We note that the compensation paid to our CEO for 2019 was for a partial year, and we estimate that he would have received approximately$5,372,238 over the course of a full year, which equates to a ratio of 85 to 1.) We determined our median employee using all income as shown in Form W-2 box 1 for all employees other than our CEO, based on information as of December 31, 2019. As permitted by SEC rules, we excluded all non-U.S. employees in determining the median employee, which consisted of a single employee in Canada. The total number of U.S. and non-U.S. employees as of December 31, 2019 was 696.
102
2019 SUMMARY COMPENSATION TABLE
Name and Principal Position | Period | Salary | Bonus(6) | Stock(7) Awards | Option Awards | Non-Equity Incentive Plan Compensation Awards | All Other(8) Compensation | Total | ||||||||
Timothy R. Wright,(1) Chief Executive Officer | 2019 | $447,115 | $1,220,000 | $3,375,000 | $0 | $0 | $27,239 | $5,069,354 | ||||||||
David Coles,(2) Former Interim Chief Executive Officer | 2019 | $0 | $0 | $0 | $0 | $0 | $0 | $0 | ||||||||
2018 | $0 | $0 | $0 | $0 | $0 | $0 | $0 | |||||||||
Edward Borkowski,(3) EVP and Interim Chief Financial Officer | 2019 | $597,885 | $0 | $610,014 | $0 | $0 | $4,095,931 | $5,303,830 | ||||||||
2018 | $363,846 | $150,000 | $0 | $0 | $330,000 | $47,294 | $891,140 | |||||||||
Peter M. Carlson,(4) EVP - Finance | 2019 | $24,231 | $0 | $1,349,994 | $0 | $0 | $0 | $1,374,225 | ||||||||
I. Mark Landy,(5) EVP and Chief Strategy Officer | 2019 | $367,001 | $0 | $673,043 | $0 | $0 | $690,924 | $1,730,968 | ||||||||
2018 | $327,788 | $100,000 | $199,824 | $0 | $117,250 | $0 | $744,862 | |||||||||
Scott M. Turner, SVP, Operations & Procurement | 2019 | $351,596 | $136,320 | $125,481 | $0 | $0 | $6,059 | $619,456 | ||||||||
2018 | $302,788 | $0 | $156,592 | $0 | $108,500 | $9,978 | $577,858 | |||||||||
(1) | The Board appointed Mr. Wright as Chief Executive Officer effective May 13, 2019. |
(2) | Mr. Coles served as Interim Chief Executive Officer from July 2, 2018 until May 12, 2019. The Company paid his employer, A&M, $908,663 and $1,147,751 for Mr. Coles’ services in 2019 and 2018, respectively. |
(3) | Mr. Borkowski served as Interim Chief Financial Officer from June 6, 2018 until his resignation effective November 15, 2019. Subsequently, he served as Acting Chief Financial Officer. |
(4) | The Board appointed Mr. Carlson EVP - Finance effective December 16, 2019. The Company later named Mr. Carlson Chief Financial Officer effective March 18, 2020. |
(5) | Mr. Landy served as Executive Vice President and Chief Strategy Officer from December 5, 2018 until the Company eliminated this position on September 16, 2019. |
(6) | Reflects a one-time $500,000 cash signing bonus. Messrs. Wright and Turner also received discretionary bonuses in 2020 in lieu of their 2019 annual incentive in the amounts of $720,000 and $136,320, respectively. |
(7) | Represents the aggregate grant date fair value of awards of restricted stock made to the executive officer in accordance with FASB ASC Topic 718. The restricted stock awards vest pro rata annually over a three-year period. |
(8) | Represents the following amounts: (a) commuting expenses: Wright - $18,135; Borkowski - $43,733; (b) reimbursement for travel expenses for their spouses to attend certain work-related events: Borkowski - $5,098; Landy - $3,924; (c) severance: Borkowski - $4,000,000 (including $2,250,000 to be paid to him or on his behalf in 2020); Landy - $687,750 (paid in 2020); (c) 401(k) match: Wright - $1,442; Borkowski - $4,659; Turner - $6,059; and (d) tax gross-up on commuting expenses: Wright $7,662; Borkowski $42,441. Does not include $2,250,000 paid to Mr. Borkowski in 2020 pursuant to the Separation and Transition Services Agreement between the Company and him. See “Compensation, Discussion & Analysis - Agreements with Mr. Borkowski, above.” |
103
GRANTS OF PLAN-BASED AWARDS FOR 2019
The following table provides information regarding grants of plan-based awards to the Company’s NEOs during 2019.
Estimated Future Payouts Under Non-Equity Incentive Plan Awards(1) | All Other Stock Awards: Number of Shares of Stock or Units(2) | All Other Option Awards: Number of Securities Underlying Options | Exercise or Base Price of Option Awards | Grant Date Fair Value of Stock and Option Awards(3) | ||||||||||||||||
Name | Grant Date | Threshold | Target | Maximum | ||||||||||||||||
Wright | 6/7/2019 | $0 | $0 | $0 | 681,818 | $3,374,999 | ||||||||||||||
Coles | — | $0 | $0 | $0 | — | — | — | $0 | ||||||||||||
Borkowski | 2/21/2019 | $0 | $0 | $0 | 203,305 | (4) | $616,014 | |||||||||||||
Carlson | 12/16/2019 | $0 | $0 | $0 | 49,295 | $349,995 | ||||||||||||||
Carlson | 12/16/2019 | $0 | $0 | $0 | 140,845 | (5) | $1,000,000 | |||||||||||||
Landy | 4/26/2019 | $0 | $0 | $0 | 279,271 | (4) | $673,043 | |||||||||||||
Turner | 4/26/2019 | $0 | $0 | $0 | 52,067 | $125,481 | ||||||||||||||
(1) | The Board never formally approved the annual incentive plan in 2019. Refer to discussion of “annual incentive plan” in the Compensation Discussion & Analysis, above. |
(2) | Represents restricted stock awards granted under the 2016 Plan. The shares of restricted stock generally vest ratably over three years from the grant date. |
(3) | The amounts shown reflect the grant date fair market values of the awards computed in accordance with FASB ASC Topic 718—“Compensation-Stock compensation.” |
(4) | As discussed under “Compensation Discussion and Analysis,” Messrs. Landy and Borkowski forfeited all unvested restricted stock held by them upon the termination of their employment during 2019. |
(5) | Represents performance-based restricted stock units. |
104
OUTSTANDING EQUITY AWARDS ON DECEMBER 31, 2019
The following table shows the number of shares covered by exercisable and un-exercisable options and unvested restricted stock awards held by the Company’s NEOs on December 31, 2019. As discussed in the CD&A, Messrs. Landy and Borkowski forfeited all unvested restricted stock held by them upon the termination of their employment during 2019.
Option Awards | Stock Awards | ||||||||||||||||||
Name | Number of Securities Underlying Unexercised Options (#) Exercisable | Number of Securities Underlying Unexercised Options (#) Unexercisable | Option Exercise Price | Option Expiration Date | Number of Securities Unvested | Market Value of Unvested Securities(1) | |||||||||||||
Wright | — | — | 681,818 | (2) | $ | 5,168,180 | |||||||||||||
Coles | — | — | — | $0 | |||||||||||||||
Borkowski | — | — | — | $0 | |||||||||||||||
Carlson | — | — | 190,140 | (3) | $ | 1,441,261 | |||||||||||||
Landy | — | — | — | $0 | |||||||||||||||
Turner | — | — | 10,000 | (4) | $75,800 | ||||||||||||||
10,600 | (5) | $80,348 | |||||||||||||||||
6,667 | (6) | $50,536 | |||||||||||||||||
52,067 | (7) | $394,668 | |||||||||||||||||
(1) | Calculated based on a closing stock price of $7.58 per share on December 31, 2019. |
(2) | A portion vested on June 7, 2020, and the remaining balance is scheduled to vest on June 7, 2021 and 2022. |
(3) | Reflects (a) a time-vested restricted stock grant with a value of $350,000 which vests pro rata annually over three years on December 16, 2020, 2021, and 2022; and (b) a performance-vested restricted stock unit grant with a value of $1,000,000, which vests upon the achievement of each of four discrete performance goals. |
(4) | The remaining balance vested on February 22, 2020. |
(5) | The remaining balance is scheduled to vest on February 22, 2021. |
(6) | The remaining balance is scheduled to vest in two installments on December 11, 2020 and 2021. |
(7) | A portion vested on April 26, 2020, and the remaining balance is scheduled to vest in on April 26, 2021 and 2022. |
2019 OPTION EXERCISES AND STOCK VESTED TABLE
The following table provides information concerning each exercise of stock options and each vesting of restricted stock during 2019, on an aggregated basis with respect to each of the Company’s NEOs.
Option Awards | Stock Awards | ||||||||||
Name | Number of Securities Acquired on Exercise | Value Realized on Exercise | Number of Securities Acquired on Vesting | Value Realized on Vesting(1) | |||||||
Wright | — | $0 | — | $0 | |||||||
Coles | — | $0 | — | $0 | |||||||
Borkowski | — | $0 | 33,333 | $100,999 | |||||||
Carlson | — | $0 | — | $0 | |||||||
Landy | — | $0 | 25,433 | $103,193 | |||||||
Turner | — | $0 | 25,800 | $71,411 | |||||||
(1) | Represents the number of shares acquired on vesting multiplied by the closing price of Common Stock on the vesting date. |
105
2019 POTENTIAL PAYMENTS UPON TERMINATION OR CHANGE IN CONTROL
This section describes additional payments that the Company would make to the NEOs assuming a hypothetical termination of employment occurred on December 31, 2019 under various scenarios. We did not include Messrs. Coles or Borkowski in the table below because they voluntarily resigned their employment before December 31, 2019. See “Compensation Discussion and Analysis - Agreements with Our Executive Officers” for a discussion of certain severance payments to and arrangements made with Messrs. Borkowski and Landy.
The Company’s agreement with Mr. Wright provides for, and its agreement with Mr. Landy provided for, compensation to the executive in the event the executive’s employment with the Company is terminated involuntarily without “Cause” (as defined in the agreement), or if the executive voluntarily terminates employment for “Good Reason” (as defined in the agreement). The compensation payable under the agreements is a lump sum severance payment equal to a multiple of two times in the case of Mr. Wright, or one time in the case of Mr. Landy, the executive’s annual base salary and targeted base bonus as of the date of termination. In addition, following termination of employment, he is entitled to receive life, health insurance coverage (subject to a COBRA election), and certain other fringe benefits equivalent to those in effect at the date of termination for period of 24 months in the case of Mr. Wright, or 12 months in the case of Mr. Landy.
The Company’s agreements with Messrs. Wright and Turner provide for compensation to the executive in the event the executive’s employment with the Company is terminated following the consummation of a “change-in-control” for reasons other than the executive’s death, disability or for “Cause” (as defined in the respective agreements), or if the executive voluntarily terminates employment for “Good Reason” (as defined in the respective agreements). The compensation payable under the agreements is a lump sum severance payment equal to a multiple of the executive’s annual base salary and targeted base bonus as of the date of the change-in-control. The multiples are 2.5 and 0.5 Messrs. Wright and Turner, respectively. In addition, following termination of employment, these executives are entitled to receive life, health insurance coverage (subject to a COBRA election), and certain other fringe benefits equivalent to those in effect at the date of termination for periods of 30 months and 6 months for Messrs. Wright and Turner, respectively. The agreements require the executive to comply with certain covenants that preclude the executive from competing with the Company or soliciting customers or employees of the Company for a period following termination of employment equal to the period for which fringe benefits are continued under the applicable agreement. The agreements expire three years after a change in control of the Company or any successor to the Company.
Upon a “change in control,” as defined in the 2006 Plan and subject to any requirements of Section 409A of the Internal Revenue Code of 1986, as amended, all outstanding awards vest and become exercisable. The Compensation Committee has discretion whether to provide that awards granted under the 2016 Plan will vest upon a “change in control.” Thus far, the Committee has exercised such discretion and provided for full vesting upon a change in control for all awards granted under the 2016 Plan to NEOs to date.
106
2019 POTENTIAL PAYMENTS UPON TERMINATION OR CHANGE IN CONTROL
Executive | Involuntary Without Cause or for Good Reason | Involuntary or for Good Reason with Change in Control | Death or Disability | ||||||||||
Wright | |||||||||||||
cash severance | 3,000,000 | (1) | $3,750,000 | (1)(2) | $0 | ||||||||
estimated benefits | $24,881 | (3) | $31,101 | (2)(3) | $0 | ||||||||
estimated value of accelerated equity awards | — | $5,168,180 | (4) | $5,168,180 | (5) | ||||||||
Carlson | |||||||||||||
cash severance | $0 | $0 | $0 | ||||||||||
estimated benefits | $0 | $0 | $0 | ||||||||||
estimated value of accelerated equity awards | $0 | $0 | $1,441,261 | (5) | |||||||||
Turner | |||||||||||||
cash severance | $0 | $248,500 | (1)(2) | $0 | |||||||||
estimated benefits | $0 | $8,644 | (2)(3) | $0 | |||||||||
estimated value of accelerated equity awards | $0 | $601,352 | (4) | $601,352 | (5) | ||||||||
Landy | |||||||||||||
cash severance | $687,750 | n/a | (6) | n/a | (6) | ||||||||
estimated benefits | $55 | n/a | (6) | n/a | (6) | ||||||||
estimated value of accelerated equity awards | $0 | n/a | (6) | n/a | (6) | ||||||||
(1) | Includes (a) annual base salary as of December 31, 2019, plus (b) annual targeted bonus for the year ended December 31, 2019, times the multiple applicable to the NEO. |
(2) | Payable only in the event the executive’s employment is terminated without cause or for “good reason” within three years following a change in control. |
(3) | Includes (a) the estimated value of medical, dental, vision and life insurance, plus (b) the employer’s cost of FICA for the duration of the severance period. |
(4) | Includes the value of unvested restricted stock based on the December 31, 2019 stock price, the vesting of which is deemed accelerated to December 31, 2019. |
(5) | If the Participant’s employment with the Company terminated on account of the Participant’s death or disability, the shares shall become vested and non-forfeitable on termination of the Participant’s employment with the Company on account of the Participant’s death or disability. |
(6) | Mr. Landy’s employment actually terminated on September 16, 2009 when the Company eliminated his position. |
107
2019 DIRECTOR COMPENSATION
The Company compensates non-employee directors with a mix of equity and cash. Directors who are full-time Company employees do not receive any compensation for their service as directors or as members of Board committees. The Company compensates non-employee directors at approximately the median of peer practices. The 2016 Plan imposes limits on awards to directors for their service as directors of (i) 125,000 shares granted during any calendar year and (ii) a maximum of $300,000 for any consecutive 12-month period for awards stated with reference to a specific dollar amount.
Equity Compensation
Upon initial election or appointment to the Board, each non-employee director receives a one-time grant of restricted shares of Common Stock valued at $50,000, plus a prorated portion of the prior year’s annual grant (based on the number of months between the date of appointment to the Board and targeted date for the next annual meeting of shareholders). This grant vests on the first anniversary of the grant date. In addition, each non-employee director receives an annual grant of restricted shares of Common Stock valued at $175,000. The Board usually makes this grant on the date of the annual meeting of shareholders, and it vests on the earlier of the next annual meeting or the first anniversary of the grant date. Because, in 2019, the Restatement was incomplete and there was incomplete information publicly available about the Company, the Board made its annual grants in the form of restricted stock units, initially denominated in cash but which will be converted to a number of shares of common stock based on the stock price on the date thirty (30) days following the date the Company first becomes current with its SEC reporting obligations. The Board altered its grant practices in an attempt to ensure that the grants are based on a reliable price for the Company’s stock and which reflects all available information and current financial statements, to prevent the possibility of a windfall, and to ensure alignment with shareholders.
Due to the pending Audit Committee investigation in early 2018 and the expectation that the Company’s financial statements might need to be restated, the Board did not make the expected $175,000 equity grant to directors in 2018. Instead, on June 13, 2019 (prior to the election or appointment of Dr. Behrens and Messrs. Barry, Bierman and Newton to the Board), the Board, in its capacity as Administrator of the 2006 Plan, modified all options then outstanding held by non-employee directors under the Company’s Assumed 2006 Stock Incentive Plan, as amended and restated as of February 25, 2014 (the “2006 Plan”), such that all options held by incumbent directors who served on the Board prior to the Company’s 2018 annual meeting of shareholders would expire on the original expiration date of such options, rather than on the first to occur of (i) three months following the date of termination of a director’s service on the Board for any reason and (ii) the expiration date of the option. The modification resulted in an incremental expense charge under GAAP, which varied by director based upon the number of outstanding options then held by the director as well as other factors. The incremental fair value of such modified options has been included in the table below in the column, “Options.”
The Nominating and Corporate Governance Committee has adopted stock ownership guidelines for the Company’s non-employee directors to better align the interests of non-employee directors with shareholders. The guidelines require non-employee directors to own shares of Common Stock with a value equal to or greater than three times their annual gross cash compensation. Newly elected directors have three years from the date of election to the Board to comply with the ownership guidelines. Shares must be owned directly by the director or the director’s immediate family members residing in the same household, held in trust for the benefit of the non-employee director or the director’s immediate family or owned by a partnership, limited liability company or other entity to the extent of the director’s interest therein (including the interests of the director’s immediate family members residing in the same household) provided that the individual has the power to vote or dispose of the shares. Unvested shares of restricted stock and unexercised stock options (vested or unvested) do not count toward satisfaction of the guidelines. The Board has suspended application of these stock ownership guidelines because the Company is not current in its SEC reporting obligations and the Company’s insider trading policy prevents the non-employee directors from buying or selling shares of Common Stock at this time.
108
Cash Compensation
In 2019, the Company also paid the following cash amounts to non-employee directors:
Chairman | Non-Chair Member | |||
Board | $71,000 | $42,000 | ||
Audit Committee | $21,000 | $11,000 | ||
Compensation Committee | $16,000 | $8,500 | ||
Nominating and Corporate Governance | $11,000 | $6,000 | ||
Science and Research Liaison | $15,000 | n/a | ||
Ethics and Compliance Committee | $12,500 | $6,500 | ||
Special Litigation Committee (ad hoc) | $15,000 | $7,500 | ||
In addition, for 2019, the Board paid excess meeting fees, subject to a cap, once the number of meetings for a particular body exceeded a threshold, as follows:
Supplemental Meeting Fees | Threshold # of Meetings | Per Meeting Fee | Supplemental Meeting Fee Cap |
Board Meetings | 12 meetings | $2,500 Chair $1,250 Member | $30,000 Chair $15,000 Member |
Audit Committee | 15 meetings | $2,000 Chair $1,000 Member | $24,000 Chair $12,000 Member |
Compensation; Science & Research liaison; Special Litigation (ad hoc) | 12 meetings | ||
Nominating & Governance; Ethics & Compliance | 10 meetings | ||
The following table provides information concerning compensation of the Company’s non-employee directors who served in 2019.
Name | Fees Earned or Paid in Cash | Stock Awards(1) | Options | Total | ||||
Luis A. Aguilar(2) | $113,250 | $0 | $0 | $113,250 | ||||
Richard J. Barry | $14,500 | $225,000 | (6)(7) | $0 | $239,500 | |||
M. Kathleen Behrens | $34,471 | $225,000 | (6)(7) | $0 | $259,471 | |||
James L. Bierman | $18,038 | $225,000 | (6)(7) | $0 | $243,038 | |||
Joseph G. Bleser(3) | $78,750 | $0 | $89,437 | (5) | $168,187 | |||
J. Terry Dewberry | $108,000 | $175,000 | (6) | $82,019 | (5) | $365,019 | ||
Charles R. Evans | $152,925 | $175,000 | (6) | $0 | (5) | $327,925 | ||
Bruce L. Hack(3) | $51,000 | $0 | $89,437 | (5) | $140,437 | |||
Charles E. Koob | $57,000 | $175,000 | (6) | $0 | $232,000 | |||
K. Todd Newton | $15,913 | $225,000 | (6)(7) | $0 | $240,913 | |||
Larry W. Papasan(4) | $61,125 | $0 | $105,073 | (5) | $166,198 | |||
Neil S. Yeston(8) | $107,125 | $175,000 | (6) | $0 | (5) | $282,119 | ||
1. | The following directors had stock options outstanding as of December 31, 2019: Papasan - 87,000; Koob - 75,000; and Bleser, Dewberry, Evans, Hack, Koob, and Yeston—each with 60,000. In addition, on December 31, 2019 each of Messrs. Barry, Bierman, and Newton, and Ms. Behrens, had restricted stock units with a value of $225,000, and each of Messrs. Dewberry, Evans, Koob and Yeston had restricted stock units with a value of $175,000. |
2. | Mr. Aguilar resigned from the Board on September 19, 2019. |
3. | The terms of Mr. Bleser and Mr. Hack expired on June 17, 2019 following the 2018 Annual Meeting. |
4. | Mr. Papasan resigned from the Board on June 17, 2019 following the 2018 Annual Meeting. |
5. | Reflects incremental fair value of options as a result of modifications effective on June 13, 2019: Bleser - $89,437; Dewberry - $82,019; Hack $89,437; Papasan $105,073; Evans, Koob and Yeston - $0. |
6. | Reflects grant of $175,000 restricted stock unit award to all directors serving after June 17, 2019. |
7. | Reflects grant of $50,000 restricted stock unit award to new directors. |
8. | Mr. Yeston serves as the Science and Research liaison to the Board and as the Chairman of the ad hoc special litigation committee. |
109
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
EQUITY COMPENSATION PLAN INFORMATION
The following table provides information about the Company’s equity compensation plans as of December 31, 2019.
Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights | Weighted average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans | ||||||
Equity compensation plans approved by security holders | 2,885,334 | $4.42 | 6,334,170 | ||||||
Equity compensation plans not approved by security holders | — | — | — | ||||||
Total | 2,885,334 | $4.42 | 6,334,170 | ||||||
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following tables sets forth certain information regarding the Company’s capital stock, beneficially owned by each person known to the Company to beneficially own more than 5% of the outstanding shares of Common Stock, each NEO, each director, and all directors and executive officers as a group. Unless otherwise indicated below the address of those identified in the table is c/o MiMedx Group, Inc., 1775 West Oak Commons Court, NE, Marietta, Georgia 30062.
SERIES B CONVERTIBLE PREFERRED STOCK | |||||||
Name of Beneficial Owner | Number of Shares of Series B Convertible Preferred Stock | Number of Shares of Common Stock Into Which They May Convert(b) | Voting Percentage(c) | ||||
EW Healthcare Partners(a) | 90,000 | 23,376,623 | 16.9% | ||||
(a) | Represents shares of Common Stock issuable upon conversion of 90,000 shares of Series B Preferred Stock owned by Falcon Fund 2 Holding Company, L.P., a partnership controlled by EW Healthcare Partners. EW Healthcare Partners Fund 2-UGP, LLC, the general partner of Falcon Fund 2 Holding Company, L.P., may also be deemed to have sole voting and investment power with respect to such shares of Common Stock. EW Healthcare Partners Fund 2-UGP, LLC disclaims beneficial ownership of such shares of Common Stock except to the extent of its pecuniary interest therein. Martin P. Sutter, Scott Barry, Ronald W. Eastman, Petri Vainio and Steve Wiggins are each a manager and collectively the managers of EW Healthcare Partners Fund 2-UGP, LLC. Each of the managers may be deemed to exercise shared voting and investment power with respect to such shares. Each manager disclaims beneficial ownership of such shares of Common Stock except to the extent of his pecuniary interest therein. Martin P. Sutter is a member of the Company’s Board of Directors. The principal address of the EW Healthcare Partners entities and each of the managers is 21 Waterway Avenue, Suite 225, The Woodlands, Texas 77380. |
(b) | Each holder of Series B Preferred Stock (each a “Holder” and collectively, the “Holders”) will have the right, at its option, to convert its Series B Preferred Stock, in whole or in part, into a number of fully paid and non-assessable shares of Common Stock equal to the Purchase Price Per Share, plus any accrued and unpaid dividends, at the conversion price. For purposes of this table the conversion price is presumed to be $3.85. No Holder may convert its shares of Series B Preferred Stock into shares of Common Stock if such conversion would result in the Holder, together with its affiliates, holding more than 19.9% of the votes entitled to be cast at any stockholders meeting or beneficially owning in excess of 19.9% of then-outstanding shares of Common Stock. |
(c) | Subject to certain exceptions, each share of Series B Preferred Stock is entitled to be voted on by the Holders and will vote on an as-converted basis as a single class with the Common Stock, subject to certain limitations on voting set forth in the related Articles of Amendment. Percentage ownership set forth in the table is based on 110,328,875 shares of Common Stock outstanding on June 25, 2020, plus 2,359,043 shares deemed outstanding pursuant to Rule 13d-3 under the Exchange Act, which includes 25,022,299 shares of Common Stock to be issued upon conversion of the Series B Stock. |
110
Name of Beneficial Owner | Number of Shares | Percentage Ownership(1) | |||||
Prescience Investment Group, LLC(2) | 7,618,335 | 5.5% | |||||
Group One Trading, LP(3) | 6,379,103 | 4.6% | |||||
NEOs, Executive Officers, and Directors | Number of Shares | Percentage Ownership(1) | |||||
Richard J. Barry(4)(5) | 3,300,000 | 2.4% | |||||
M. Kathleen Behrens, Ph.D.(4) | — | * | |||||
James L. Bierman(5) | — | * | |||||
Edward J. Borkowski(6) | 21,194 | * | |||||
Peter M. Carlson(7) | 49,295 | * | |||||
David Coles(8) | — | * | |||||
J. Terry Dewberry(9)(10) | 187,126 | * | |||||
Charles R. Evans(10)(11) | 125,460 | * | |||||
William A. Hawkins(17) | — | * | |||||
Charles E. Koob(10)(12) | 1,520,628 | 1.1% | |||||
I. Mark Landy(13) | 33,529 | * | |||||
K. Todd Newton(4) | — | * | |||||
Martin P. Sutter(17) | 23,376,623 | 16.9% | |||||
Scott M. Turner(14) | 104,843 | * | |||||
Timothy R. Wright(15) | 592,227 | * | |||||
Neil S. Yeston(10)(16) | 130,460 | * | |||||
Total Directors and Executive Officers(18) (15 persons) | 29,232,524 | 21.1% | |||||
* | Less than 1% |
(1) | The beneficial ownership set forth in the table is determined in accordance with SEC rules. The percentage of beneficial ownership is based on 110,328,875 shares of Common Stock outstanding on June 25, 2020, plus 2,359,043 shares deemed outstanding pursuant to Rule 13d-3 under the Exchange Act and 25,974,026 shares deemed outstanding upon conversion of the Company’s Series B Preferred Stock at $3.85 per share. See notes (b) and (c), above. |
(2) | On May 30, 2019, Prescience Investment Group, LLC filed an amendment to its Schedule 13D indicating shared voting power and dispositive power over 7,618,335 shares, shared voting power and dispositive power over 4,888,652 shares by Prescience Partners, LP, shared voting power and dispositive power over 1,845,539 shares by Prescience Point Special Opportunity LP, and shared voting power and dispositive power over 6,734,191 shares by Prescience Capital, LLC. The address for Prescience Investment Group, LLC is 1670 Lobdell Avenue, Suite 200, Baton Rouge, LA 70806. |
(3) | According to the most recent Schedule 13G filed with the SEC on January 31, 2019, Group One Trading, LP had sole voting and dispositive power with respect to 6,379,103 shares. The address for Group One Trading, LP is 440 South LaSalle St, Ste. 3232, Chicago, IL 60605 |
(4) | Does not include restricted stock units granted on October 22, 2019 with a value of $225,000 which will be settled in Common Stock based on a stock price determined after the 2019 annual meeting of shareholders and after the Company becomes current in its reporting obligations. |
(5) | Reflects beneficial ownership of shares held by the Richard and Susan Barry Family Trust. |
(6) | Mr. Borkowski resigned as Executive Vice President and Interim Chief Financial Officer effective November 15, 2019. |
(7) | Mr. Carlson joined the Company as Executive Vice President, Finance, on December 16, 2019. Does not include 140,844 restricted stock units granted on December 16, 2019 that will vest based upon the achievement of certain performance criteria. |
(8) | Mr. Coles served as Interim Chief Executive Officer until May 13, 2019. |
111
(9) | Includes 60,000 shares issuable upon the exercise of options. |
(10) | Does not include restricted stock units granted on October 22, 2019 with a value of $175,000 which will be settled in Common Stock based on a stock price determined after the 2019 annual meeting of shareholders and after the Company becomes current in its reporting obligations. |
(11) | Includes 60,000 shares issuable upon the exercise of options. |
(12) | Includes 1,375,627 shares held by a trust and 60,000 shares issuable upon the exercise of options. |
(13) | The Company eliminated Mr. Landy's position of Chief Strategy Officer effective September 16, 2019. |
(14) | Does not include restricted stock units granted on February 18, 2020 with a value of $284,000 which will be settled in Common Stock based on a stock price determined after the Company becomes current in its reporting obligations. |
(15) | Does not include restricted stock units granted on February 18, 2020 with a value of $3,375,000 which will be settled in Common Stock based on a stock price determined after the Company becomes current in its reporting obligations. |
(16) | Includes 60,000 shares issuable upon the exercise of options. |
(17) | For purposes of this table all shares of Series B Preferred Stock are deemed to have converted to Common Stock at $3.85 per share. Effective July 2, 2020, pursuant to the terms of the Purchase Agreement and the Preferred Stock Amendment, the Company increased the size of the Board of Directors and appointed Martin P. Sutter and William A. Hawkins III to serve as Preferred Directors. Mr. Sutter is deemed to own beneficially shares controlled by EW Healthcare Partners. See notes (b), (c) and (1), above. No Holder may convert its shares of Series B Preferred Stock into shares of Common Stock if such conversion would result in the Holder, together with its affiliates, holding more than 19.9% of the votes entitled to be cast at any stockholders meeting or beneficially owning in excess of 19.9% of then-outstanding shares of Common Stock. |
(18) | Represents the ownership of only those persons currently serving as a director or executive officer of the Company. |
112
Item 13. Certain Relationships and Related Transactions, and Director Independence
Policies and Procedures for Approval of Related Party Transactions
Under its charter, the Audit Committee is responsible for reviewing and approving all transactions or arrangements between the Company and Section 16 reporting persons and any of their respective affiliates, associates or related parties. In determining whether to approve or ratify a related party transaction, the Audit Committee considers all relevant facts and circumstances available to it, such as:
•Whether the terms of the transaction are fair to the Company and at least as favorable to the Company as would apply if the transaction did not involve a related party;
•Whether there are demonstrable business reasons for the Company to enter into the transaction;
•Whether the transaction would impair the independence of an outside director; and
• | Whether the transaction would present an improper conflict of interest for any director or executive officer, taking into account the size of the transaction, the direct or indirect nature of the related party’s interest in the transaction and the ongoing nature of any proposed relationship, and any other factors the Audit Committee deems relevant. |
Related Party Transactions
The Company has employed Thomas Koob as its Chief Scientific Officer (a non-executive officer) since 2006. Thomas Koob is the brother of a director, Charles Koob. Subsequent to the Company’s employment of Thomas Koob, Charles Koob was appointed as a director of the Company in March 2008. In 2019, the Company paid Thomas Koob an annual salary of $235,210 and provided equity, incentive compensation and other compensation of $155,957.
The Company employs Simon Ryan, the brother-in-law of its former General Counsel, Alexandra O. Haden (who resigned from the Company effective August 12, 2019), as a sales representative. In 2019, the Company paid Mr. Ryan total compensation of $152,126, consisting of a salary of $95,000 and sales commissions, equity and other compensation of $57,126.
Director Independence
Although the Common Stock is no longer listed on NASDAQ due to the Company’s failure to timely file periodic reports, the Board continues to comply with NASDAQ’s listing standards with respect to Board independence. NASDAQ listing standards require that a majority of the members of the Board be independent, which means that they are not officers or employees of the Company and are free of any relationship that would interfere with the exercise of their independent judgment. The Board has determined that Dr. Behrens and Messrs. Barry, Bierman, Dewberry, Evans, Newton, and Yeston are “independent” under NASDAQ listing standards.
Compensation Committee Interlocks and Insider Participation
During 2019, the following persons served on the Compensation Committee: Richard J. Barry, James L. Bierman, Joseph G. Bleser, Larry W. Papasan, and Neil S. Yeston. No member of the Compensation Committee is or has been an officer or employee of the Company. During 2019, none of the Company’s executive officers served on the board of directors or compensation committee of any other entity that had an executive officer that serves on the Company’s Board or Compensation Committee.
113
Item 14. Principal Accounting Fees and Services
Audit Firm Fees
The Audit Committee’s duties include pre-approving audit and non-audit services provided to the Company by the Company’s independent registered public accounting firm, BDO USA, LLP (“BDO”). All of the services in respect of 2019 and 2018 under the Audit Fees, Audit-Related Fees, Tax Fees and All Other Fees categories below were pre-approved by the Audit Committee.
Type of Fee | Year Ended December 31, 2019 | Year Ended(1) December 31, 2018 | |
Audit Fees(2) | $3,875,000 | $2,433,333 | |
Audit-Related Fees(3) | $19,000 | $21,400 | |
Tax Fees | $0 | $0 | |
All Other Fees | $0 | $0 | |
(1) | The Company engaged BDO in May 2019 to audit its financial statements for the years ended December 31, 2018, 2017, and 2016. Total fees incurred by BDO were $7.3 million and were apportioned equally to each of the three years for the purposes of this tabular presentation. The Company paid or incurred these fees in 2019. |
(2) | This category includes fees for the audit of the Company’s annual financial statements and review of financial statements included in its quarterly reports on Form 10-Q. |
(3) | This relates to BDO’s audit of the Company’s 401(k) plan. |
114
PART IV
Item 15. Exhibits, Financial Statement Schedules
(a) | Documents filed as part of this report: |
(1) | Financial Statements |
(2) | Financial Statement Schedule: |
The following Financial Statement Schedule is filed as part of this Report:
Schedule II Valuation and Qualifying Accounts for the years ended December 31, 2019, 2018 and 2017
(3) | Exhibits |
See Item 15(b) below. Each management contract or compensation plan has been identified with an asterisk.
(b) | Exhibits |
Notes
* | Indicates a management contract or compensatory plan or arrangement |
# | Filed herewith |
## | Certain exhibits and schedules have been omitted pursuant to Item 601(b)(2) of Regulation S-K, but a copy will be furnished supplementally to the Securities and Exchange Commission upon request. |
Exhibit Number | Description | |
3.1 | Articles of Incorporation, together with Articles of Amendment effective each of May 14, 2010; August 8, 2012, November 8, 2012; and May 15, 2015 (incorporated by reference to Exhibit 3.1 to the Registrant’s Form 10-K filed on March 1, 2017). | |
3.2 | Articles of Amendment to the Articles of Incorporation effective November 6, 2018 (incorporated by reference to Exhibit 3.1 to the Registrant’s Form 8-A filed on November 7, 2018). | |
3.3# | ||
3.4 | Bylaws of MiMedx Group, Inc., as amended and restated as of October 3, 2018 (incorporated by reference to Exhibit 3.1 to the Registrant’s Form 8-K filed on October 4, 2018). | |
4.1# | ||
10.1 | Technology License Agreement dated January 29, 2007 between MiMedx, Inc., Shriners Hospitals for Children and University of South Florida Research Foundation (incorporated by reference to Exhibit 10.32 to the Registrant’s Form 8-K filed on February 8, 2008). | |
10.2 | Lease effective May 1, 2013 between Hub Properties of GA, LLC and MiMedx Group, Inc. (incorporated by reference to Exhibit 10.1 to the Registrant’s Form 10-Q filed on May 10, 2013). | |
10.3 | First Amendment to Lease dated March 7, 2017 between CPVF II West Oak LLC (as successor in interest to HUB Properties of GA, LLC) and MiMedx Group, Inc. (incorporated by reference to Exhibit 10.1 to the Registrant’s Form 8-K filed on March 13, 2017). | |
10.4 | Second Amendment to Lease made as of August 29, 2018 for real property and improvements located at 1775 West Oak Commons Court, Marietta, Georgia between RE Fields, LLC, successor in interest to HUB Properties GA, LLC, and CPVF II West Oak LLC, and MiMedx Group, Inc., dated January 25, 2013, as amended March 7, 2017 (incorporated by reference to Exhibit 10.4 to Annual Report on Form 10-K filed March 17, 2020). | |
10.5* | MiMedx Group, Inc. Assumed 2006 Stock Incentive Plan, as amended and restated effective February 25, 2014 (incorporated by reference to Exhibit 10.2 to the Registrant’s Form 8-K filed on March 3, 2014). | |
10.6* | Form of Incentive Stock Option Agreement under the MiMedx Group, Inc. Assumed 2006 Stock Incentive Plan (incorporated by reference to Exhibit 10.4 to the Registrant’s Form 10-K filed on March 4, 2014). | |
10.7* | Form of Nonqualified Stock Option Agreement under the MiMedx Group, Inc. Assumed 2006 Stock Incentive Plan (incorporated by reference to Exhibit 10.5 to the Registrant’s Form 10-K filed on March 4, 2014). | |
10.8* | Form of Restricted Stock Agreement for Non-Employee Directors under the MiMedx Group, Inc. 2006 Assumed Stock Incentive Plan (incorporated by reference to Exhibit 10.66 to the Registrant’s Form 10-Q filed on August 8, 2013). | |
115
Exhibit Number | Description | |
10.9* | Form of Restricted Stock Agreement under the MiMedx Group, Inc. 2006 Assumed Stock Incentive Plan (incorporated by reference to Exhibit 10.3 to the Registrant’s Form 10-K filed on March 4, 2014). | |
10.10* | 2016 Equity and Cash Incentive Plan (incorporated by reference to Appendix A to the Registrant’s Definitive Proxy Statement on Schedule 14A filed on April 12, 2016). | |
10.11* | Form of Incentive Stock Option Agreement under the MiMedx Group, Inc. 2016 Equity and Cash Incentive Plan (incorporated by reference to Exhibit 10.2 to the Registrant’s Form 10-Q filed on August 2, 2016). | |
10.12* | Form of Restricted Stock Agreement under the MiMedx Group, Inc 2016 Equity and Cash Incentive Plan (for shares not registered under the Securities Act of 1933) (incorporated by reference to Exhibit 10.9 to the Registrant’s Form 8-K filed on May 30, 2019). | |
10.13* | Form of Restricted Stock Agreement under the MiMedx Group, Inc. 2016 Equity and Cash Incentive Plan (incorporated by reference to Exhibit 10.3 to the Registrant’s Form 10-Q filed on August 2, 2016). | |
10.14* | Form of Restricted Stock Agreement for Non-Employee Directors under the MiMedx Group, Inc. 2016 Equity and Cash Incentive Plan (incorporated by reference to Exhibit 10.11 to the Registrant’s Form 8-K filed on May 30, 2019). | |
10.15* | Form of Nonqualified Stock Option Agreement under the MiMedx Group, Inc. 2016 Equity and Cash Incentive Plan (incorporated by reference to Exhibit 10.4 to the Registrant’s Form 10-Q filed on August 2, 2016). | |
10.16* | Form of Director Restricted Stock Unit Award Agreement (incorporated by reference to Exhibit 10.16 to the Registrant’s Annual Report on Form 10-K filed March 17, 2020). | |
10.17* | Consulting Agreement with Alexandra O. Haden dated August 27, 2019 (incorporated by reference to Exhibit 10.26 to the Registrant’s Annual Report on Form 10-K filed March 17, 2020). | |
10.18* | Cooperation Agreement dated as of May 29, 2019 among MiMedx Group, Inc., M. Kathleen Behrens Wilsey, K. Todd Newton, Richard J. Barry, Prescience Partners, LP, Prescience Point Special Opportunity LP, Prescience Capital LLC, Prescience Investment Group, LLC d/b/a Prescience Point Capital Management LLC and Eiad Asbahi (incorporated by reference to Exhibit 10.32 to the Registrant’s Form 8-K filed on May 30, 2019). | |
10.19## | Loan Agreement, dated June 10, 2019, by and between MiMedx Group, Inc., the other guarantors party thereto, the lenders party thereto and Blue Torch Finance LLC, as administrative agent and collateral agent (incorporated by reference to Exhibit 10.1 to the Registrant’s Form 8-K Filed June 11, 2019). | |
10.20 | First Amendment, dated as of April 22, 2020, to Loan Agreement, dated June 10, 2019, by and between MiMedx Group, Inc., the other guarantors party thereto, the lenders party thereto and Blue Torch Finance LLC, as administrative agent and collateral agent (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed April 27, 2020). | |
10.21* | Form of Change in Control Severance Compensation and Restrictive Covenant Agreement (incorporated by reference to Exhibit 10.24 to the Registrant’s Form 8-K filed on May 30, 2019). | |
10.22* | Form of (Non-change in Control) Executive Severance Agreement (incorporated by reference to Exhibit 10.25 to the Registrant’s Form 8-K filed on May 30, 2019). | |
10.23* | Form of Indemnification Agreement (incorporated by reference to Exhibit 10.65 to the Registrant’s Form 8-K filed July 15, 2008). | |
10.24* | Form of Employee Inventions and Assignment Agreement (incorporated by reference to Exhibit 10.4 to the Registrant’s Form 8-K/A filed on June 12, 2018). | |
10.25* | Form of Confidentiality and Non-Solicitation Agreement (incorporated by reference to Exhibit 10.2 to the Registrant’s Form 8-K/A filed on June 12, 2018). | |
10.26* | Form of Non-Competition Agreement (incorporated by reference to Exhibit 10.3 to the Registrant’s Form 8-K/A filed on June 12, 2018). | |
10.27* | Letter Agreement dated April 10, 2019 between MiMedx Group, Inc. and Timothy R. Wright (incorporated by reference to Exhibit 10.1 to the Registrant’s Form 8-K filed on May 9, 2019). | |
10.28* | Engagement Letter dated July 2, 2018 between MiMedx Group, Inc. and Alvarez & Marsal North America, LLC (incorporated by reference to Exhibit 10.1 to the Registrant’s Form 8-K/A filed on July 11, 2018). | |
10.29*# | Employment Offer Letter between the Company and Peter M. Carlson, as amended and restated on April 29, 2020. | |
10.30*# | Employment Offer Letter between the Company and William F. Hulse IV as of November 4, 2019. | |
10.31* | Employment Offer Letter dated April 3, 2018 between MiMedx Group, Inc. and Edward Borkowski (incorporated by reference to Exhibit 10.22 to the Registrant’s Form 8-K filed on May 30, 2019). | |
10.32* | Separation and Transition Services Agreement, dated as of November 15, 2019, between MiMedx Group, Inc. and Edward J. Borkowski (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed November 20, 2019). | |
10.33*# | ||
10.34*# | ||
10.35*# | ||
10.36# ## | Loan Agreement dated as of June 30, 2020 by and among MiMedx Group, Inc., certain subsidiaries of MiMedx Group, Inc. parties thereto, the Lenders from time to time party hereto, Hayfin Services LLP, as administrative agent for the Lenders and as collateral agent for the Secured Parties. | |
116
Exhibit Number | Description | |
10.37*# | Employment Offer Letter between the Company and William L. Phelan dated as of April 30, 2020. | |
10.38# ## | Securities Purchase Agreement, dated as of June 30, 2020, by and between MiMedx Group, Inc., Falcon Fund 2 Holding Company, L.P. and certain other investors. | |
10.39# | Registration Rights Agreement dated as of July 2, 2020, by and between MiMedx Group, Inc. and Falcon Fund 2 Holding Company, L.P. | |
16.1 | Letter from Cherry Bekaert LLP dated August 9, 2017 (incorporated by reference to Exhibit 16.1 to Current Report on Form 8-K filed August 10, 2017). | |
16.2 | Letter from Ernst & Young LLP dated December 7, 2018 (incorporated by reference to Exhibit 16.1 to Current Report on Form 8-K filed December 7, 2018). | |
21.1# | ||
23.1# | ||
24.1# | Power of Attorney (included on the signature page to this Report). | |
31.1# | Certification of Chief Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |
31.2# | Certification of Chief Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |
32.1# | Certification of Chief Executive Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
32.2# | Certification of Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
101.INS# | XBRL Instance Document | |
101.SCH# | XBRL Taxonomy Extension Schema Document | |
101.CAL# | XBRL Taxonomy Extension Calculation Linkbase Document | |
101.DEF# | XBRL Taxonomy Extension Definition Linkbase Document | |
101.LAB# | XBRL Taxonomy Extension Label Linkbase Document | |
101.PRE# | XBRL Taxonomy Extension Presentation Linkbase Document | |
Item 16. Form 10-K Summary
Not applicable.
117
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
July 6, 2020 | MIMEDX GROUP, INC. | |
By: | /s/ Peter M. Carlson | |
Peter M. Carlson | ||
Chief Financial Officer and Principal Financial Officer | ||
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below hereby constitutes and appoints William F. Hulse IV and David A. Wisniewski and each of them acting individually, as his or her true and lawful attorneys-in-fact and agents, each with full power of substitution and resubstitution, for him or her in any and all capacities, to sign any and all amendments to this Annual Report on Form 10-K for the year ended December 31, 2019, and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, hereby ratifying and confirming our signatures as they may be signed by our said attorney to any and all amendments to said Annual Report on Form 10-K.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
118
Signature / Name | Title | Date | ||
/s/ Timothy R. Wright | Chief Executive Officer and Director | July 6, 2020 | ||
Timothy R. Wright | (Principal Executive Officer) | |||
/s/ Peter M. Carlson | Chief Financial Officer | July 6, 2020 | ||
Peter M. Carlson | (Principal Financial Officer) | |||
/s/ William L. Phelan | Senior Vice President and Chief Accounting Officer | July 6, 2020 | ||
William L. Phelan | (Principal Accounting Officer) | |||
/s/ M. Kathleen Behrens | Chair of the Board (Director) | July 6, 2020 | ||
M. Kathleen Behrens | ||||
/s/ Richard J. Barry | Director | July 6, 2020 | ||
Richard J. Barry | ||||
/s/ James L. Bierman | Director | July 6, 2020 | ||
James L. Bierman | ||||
/s/ J. Terry Dewberry | Director | July 6, 2020 | ||
J. Terry Dewberry | ||||
/s/ Charles R. Evans | Director | July 6, 2020 | ||
Charles R. Evans | ||||
Director | ||||
William A. Hawkins III | ||||
/s/ Charles E. Koob | Director | July 6, 2020 | ||
Charles E. Koob | ||||
/s/ K. Todd Newton | Director | July 6, 2020 | ||
K. Todd Newton | ||||
Director | ||||
Martin P. Sutter | ||||
/s/ Neil S. Yeston | Director | July 6, 2020 | ||
Neil S. Yeston | ||||
119