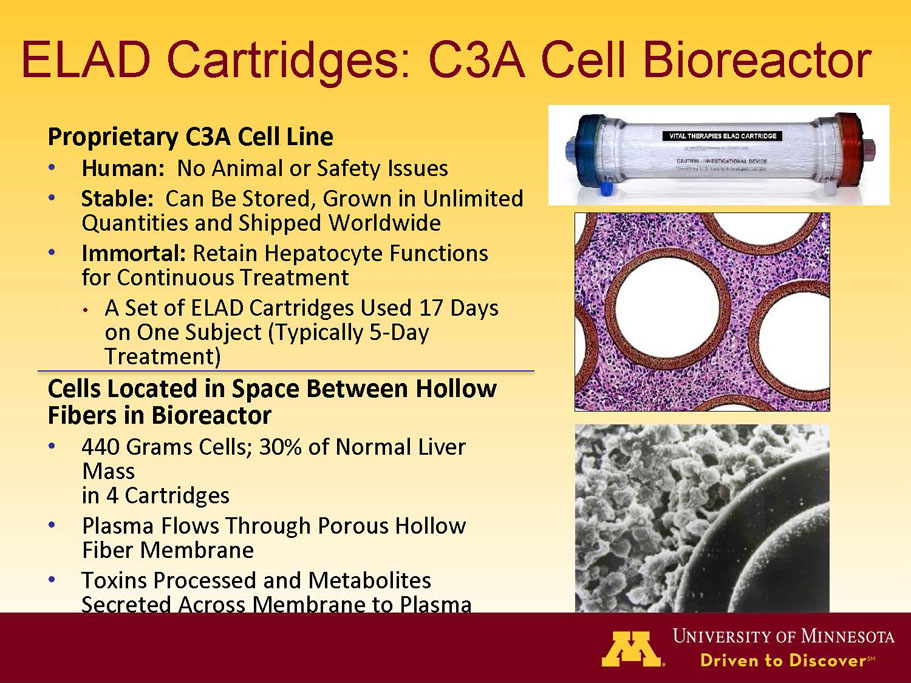
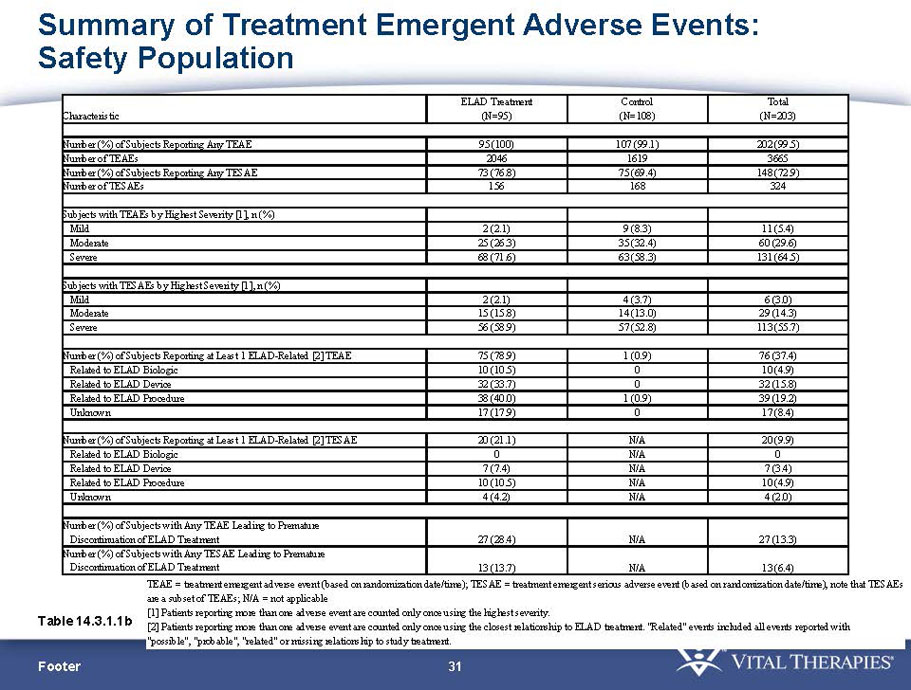
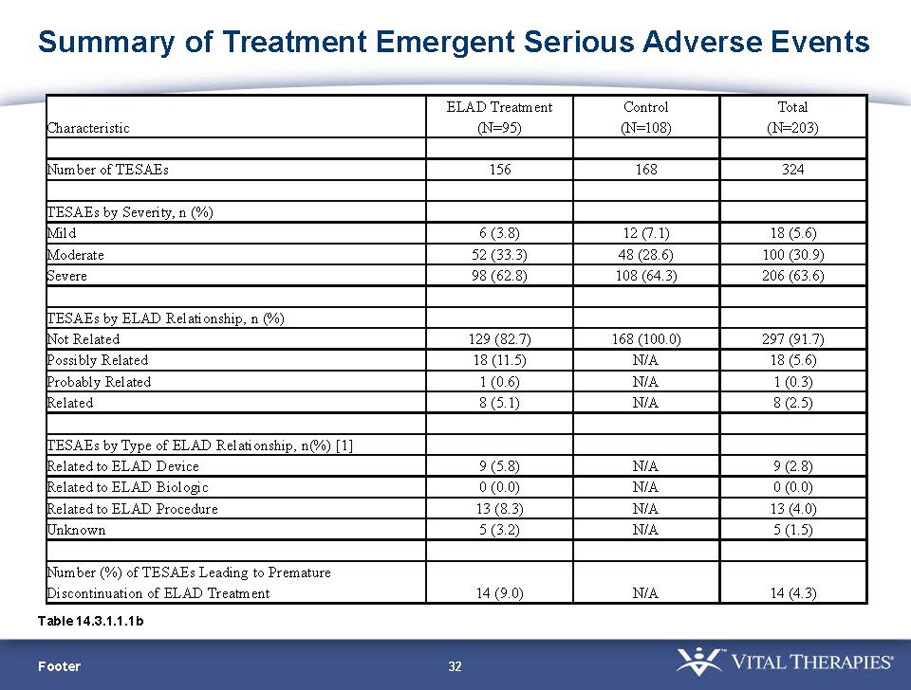
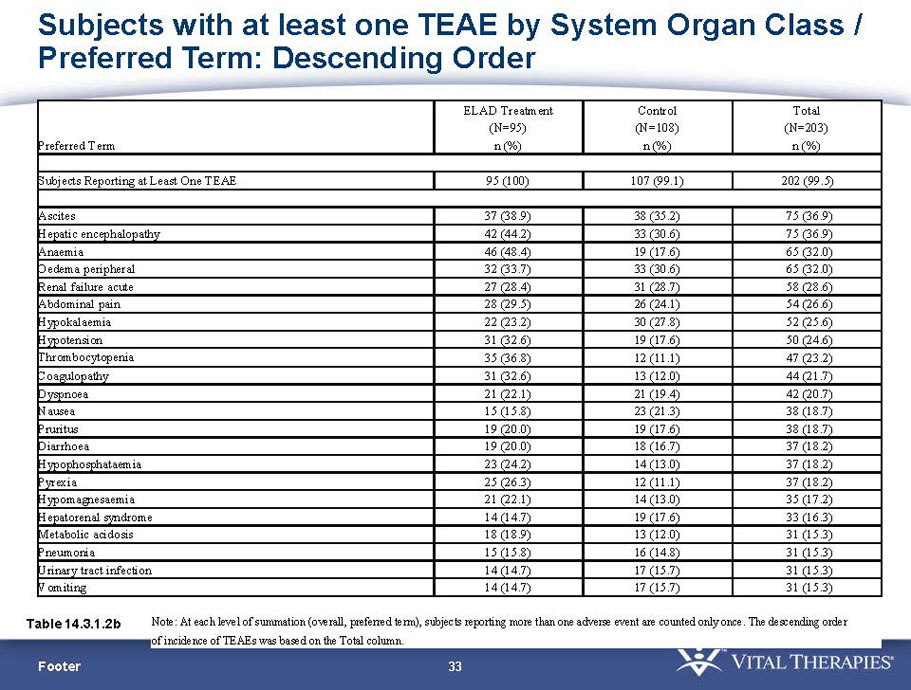
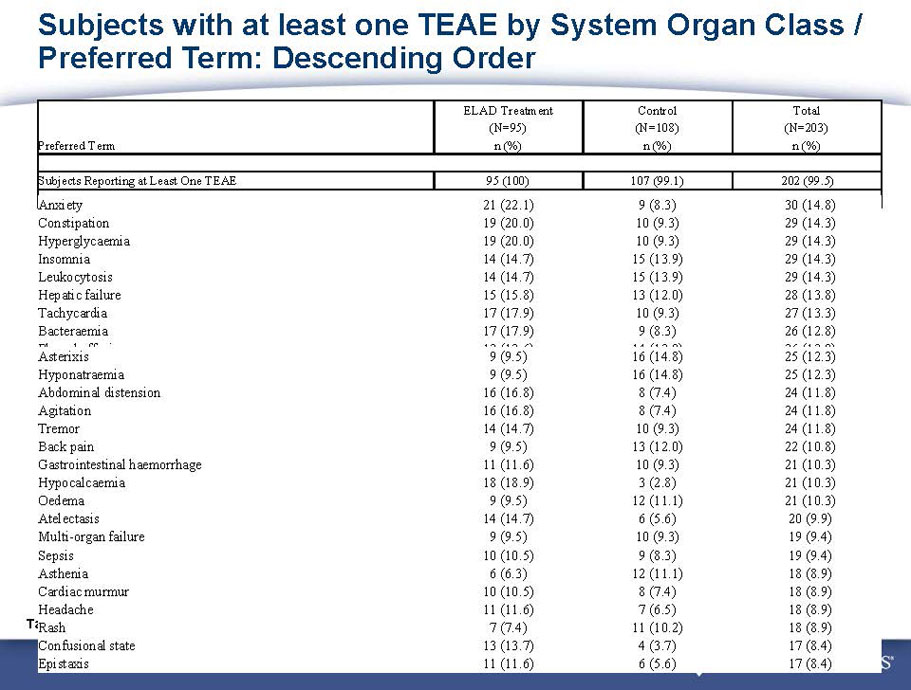
Exhibit 99.1

Vital Therapies Announces additional data from vti-208 presented at ROSTOCK CONFERENCE
SAN DIEGO, August 31, 2015 (GLOBE NEWSWIRE) -- Vital Therapies, Inc. (Nasdaq: VTL), a biotherapeutic company developing ELAD®, a cell-based therapy targeting the treatment of liver failure, today announced that additional data from its VTI-208 clinical trial were presented by two principal investigators of the study at the 16th International Symposium on Albumin Dialysis in Liver Disease on Saturday, August 29, 2015 in Rostock-Warnemunde, Germany. The conference’s focus this year is: “Artificial and Bio-artificial Liver Support-Mechanisms of Action and Clinical Effects.” Topline data from the VTI-208 clinical trial, which failed to reach its primary or secondary endpoints, was disclosed on August 21, 2015.
The presentations, titled “Acute Alcoholic Hepatitis and Liver Support,” and “Appropriate Patient Selection as a Key to Increase Benefit/Risk Ratio for ELAD”, were made by Julie Thompson, MD, M.P.H., Assistant Professor of Medicine, Gastroenterology Division, University of Minnesota, and Ram M. Subramanian, MD, Associate Professor of Medicine and Surgery, Emory University School of Medicine. Drs. Thompson and Subramanian were the two highest enrolling principal investigators in the VTI-208 clinical trial with 17 subjects each. Newly presented information includes:
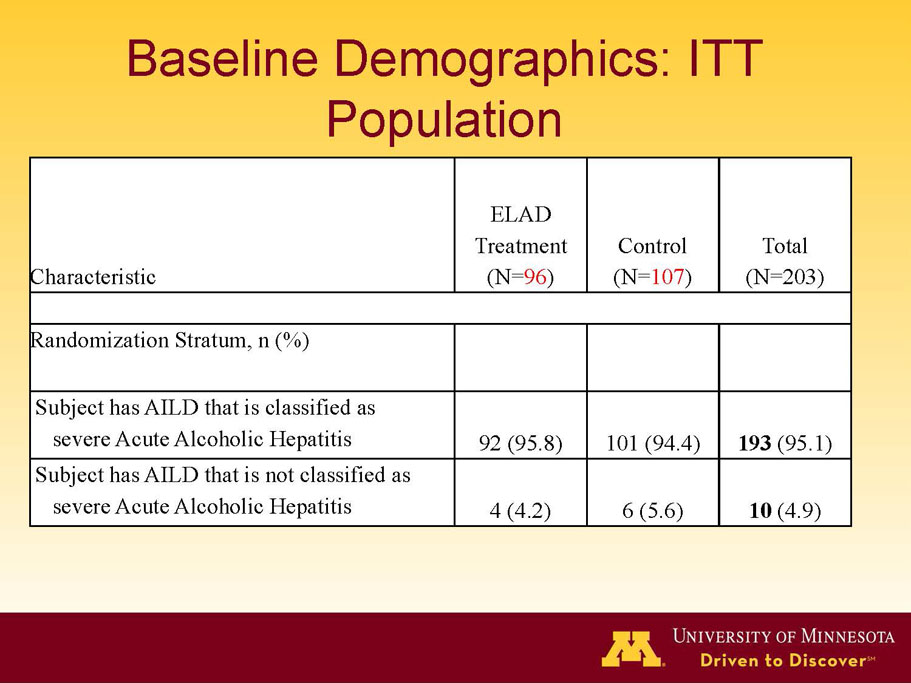
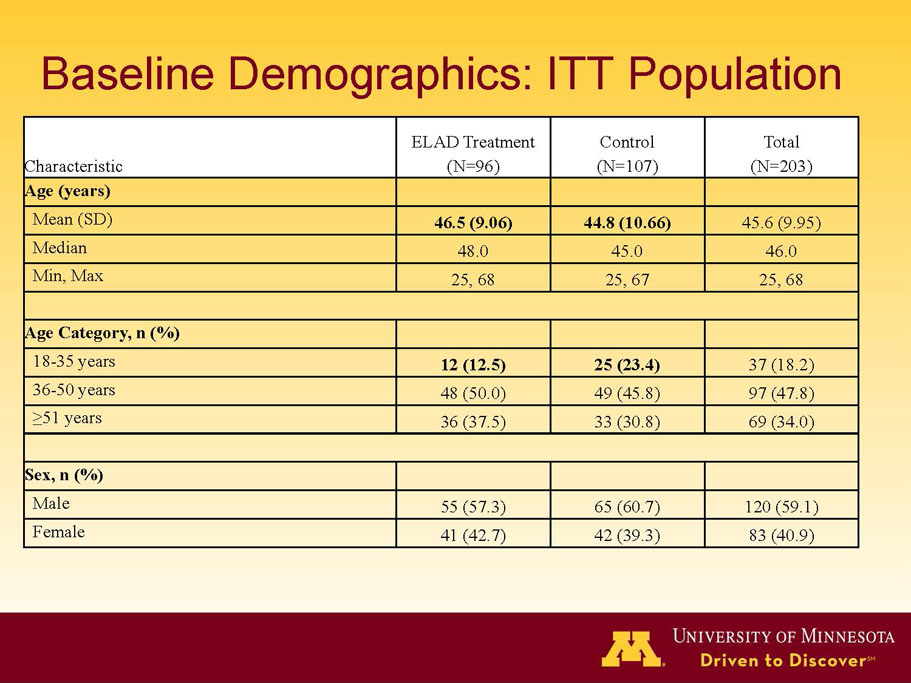
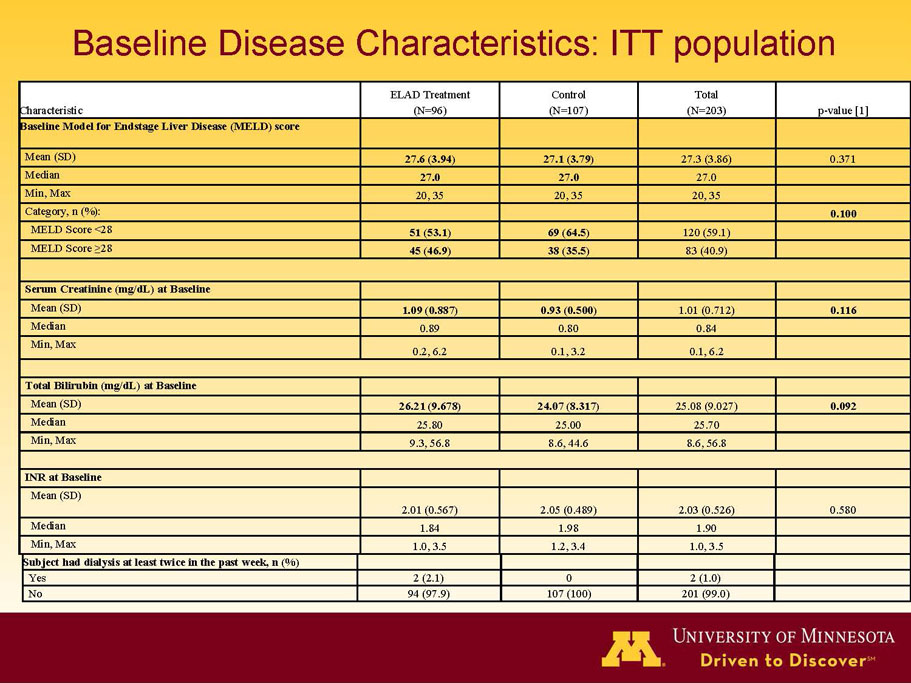
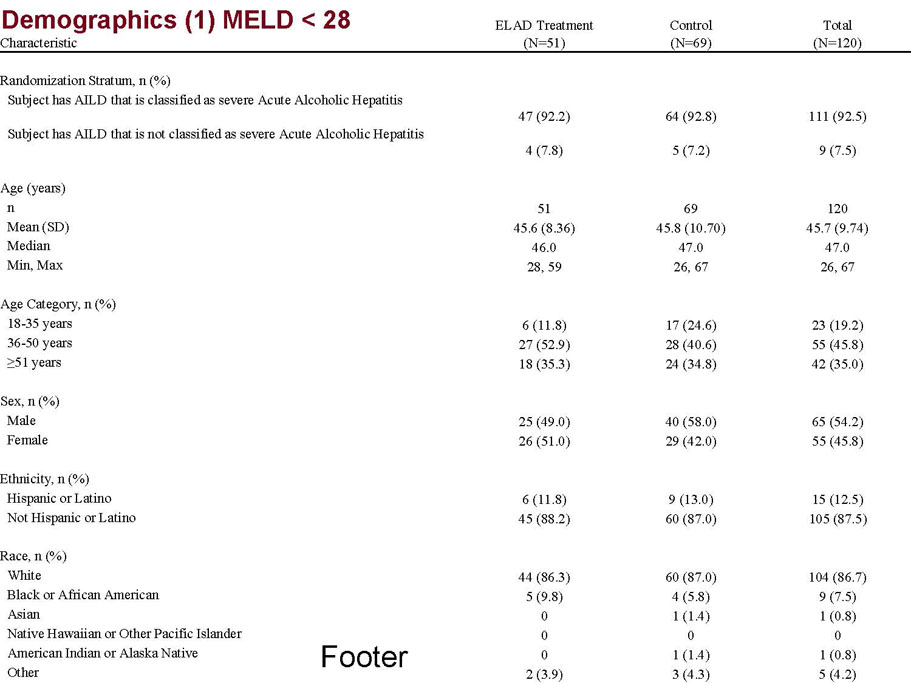
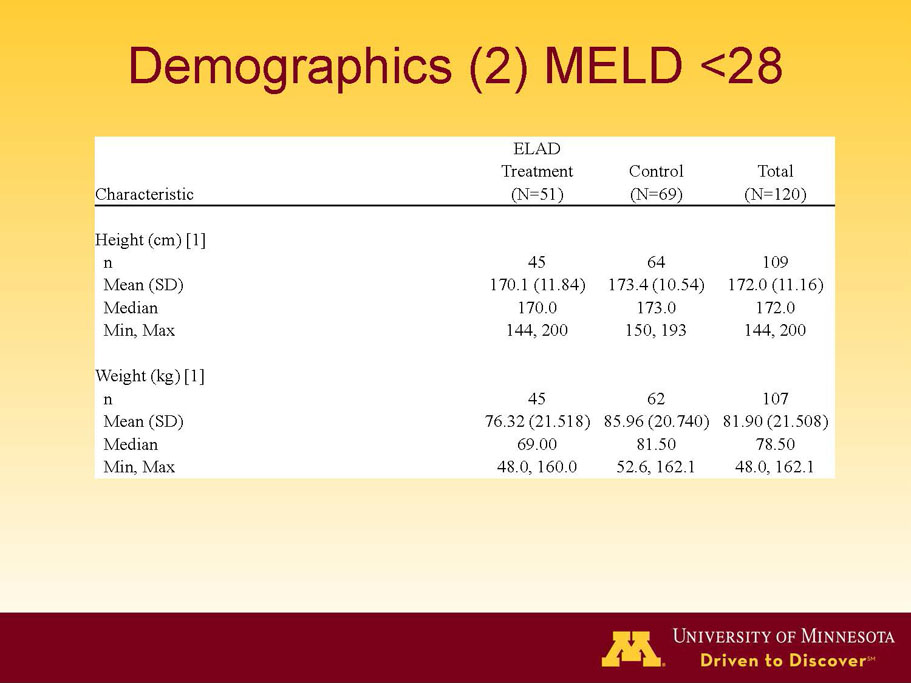
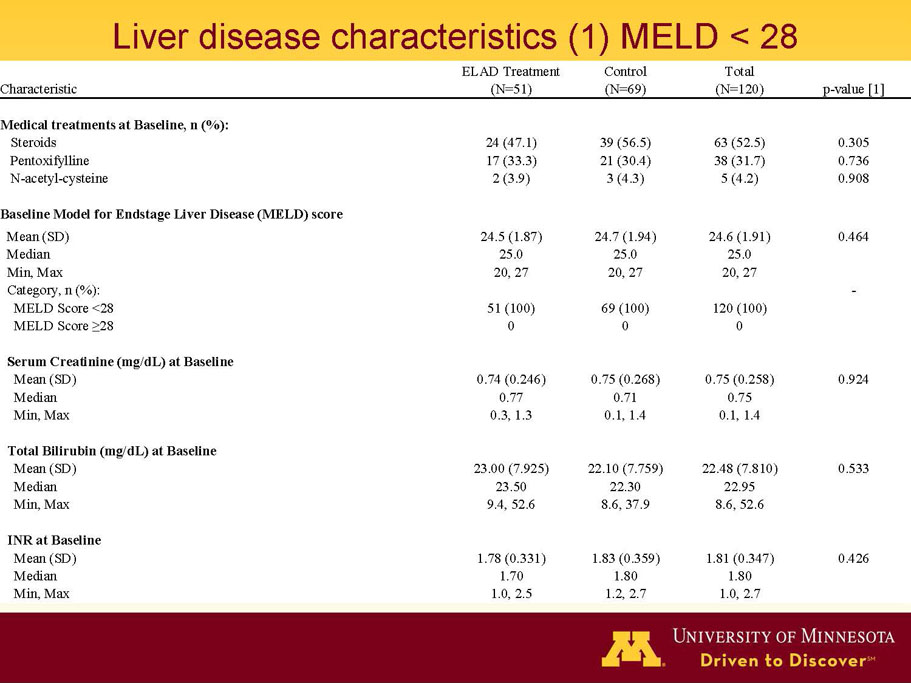
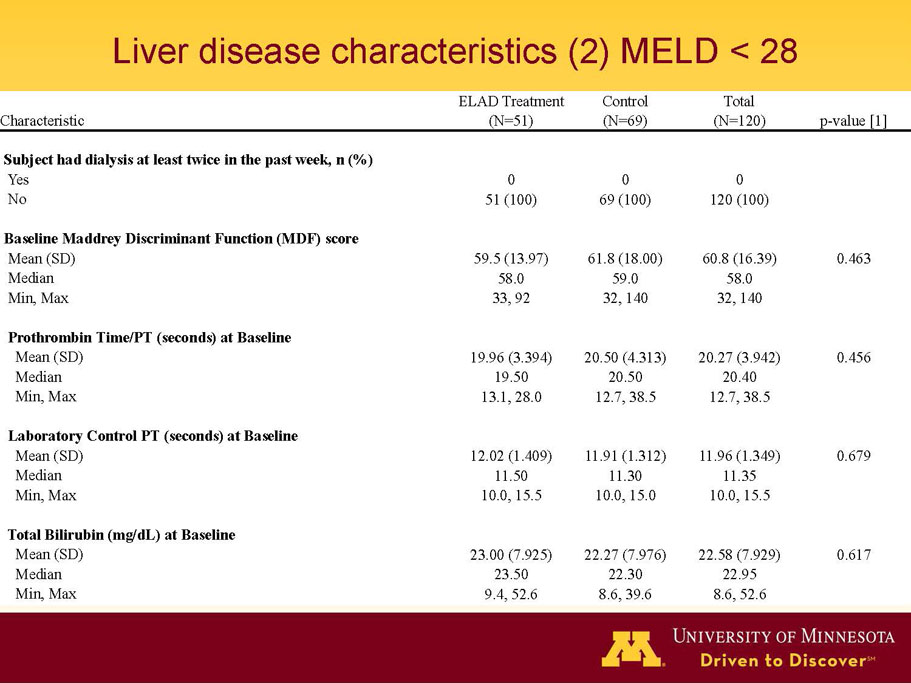
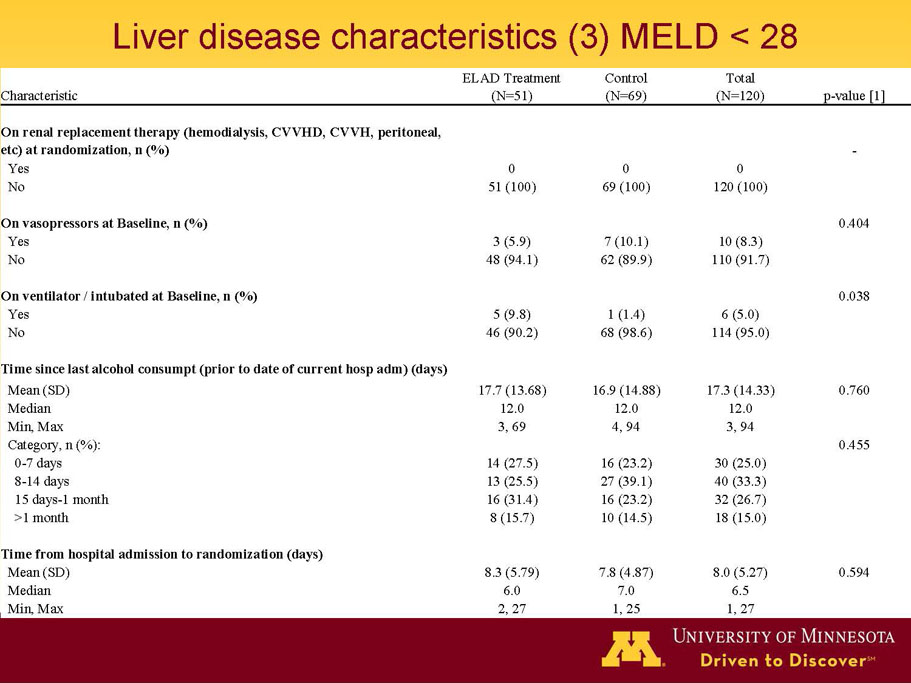
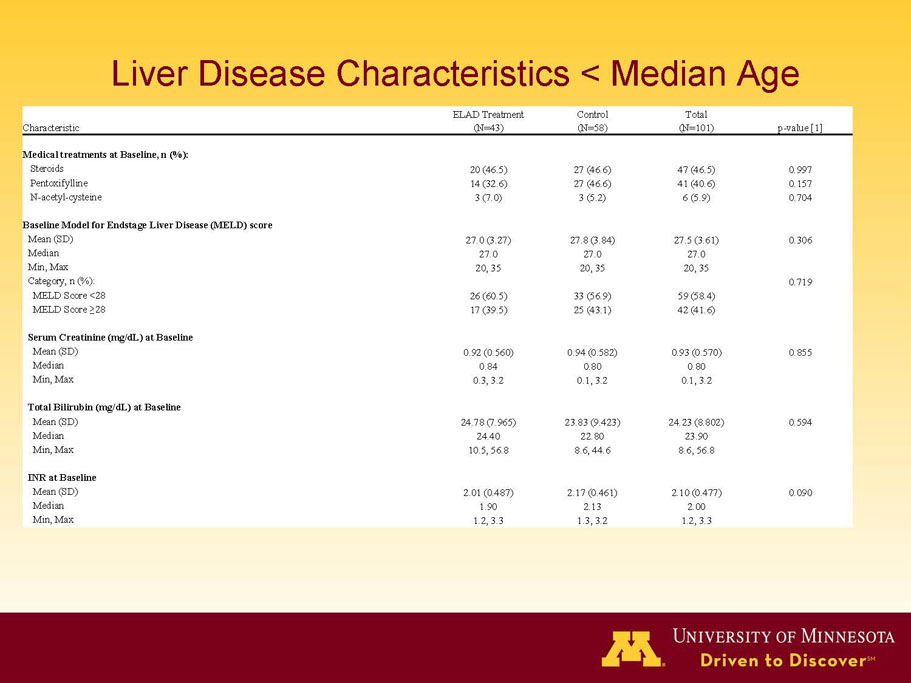
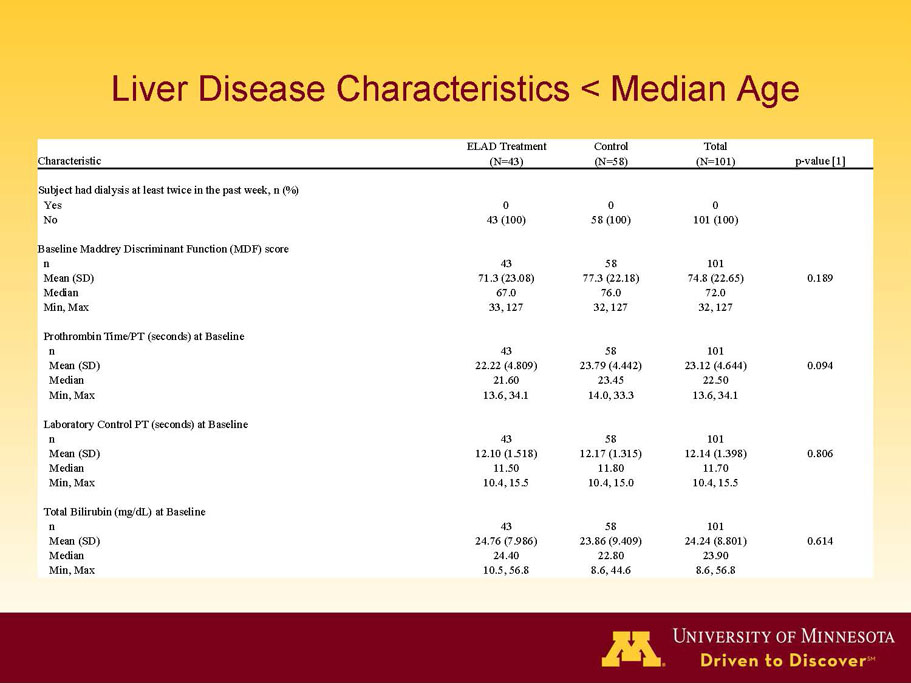
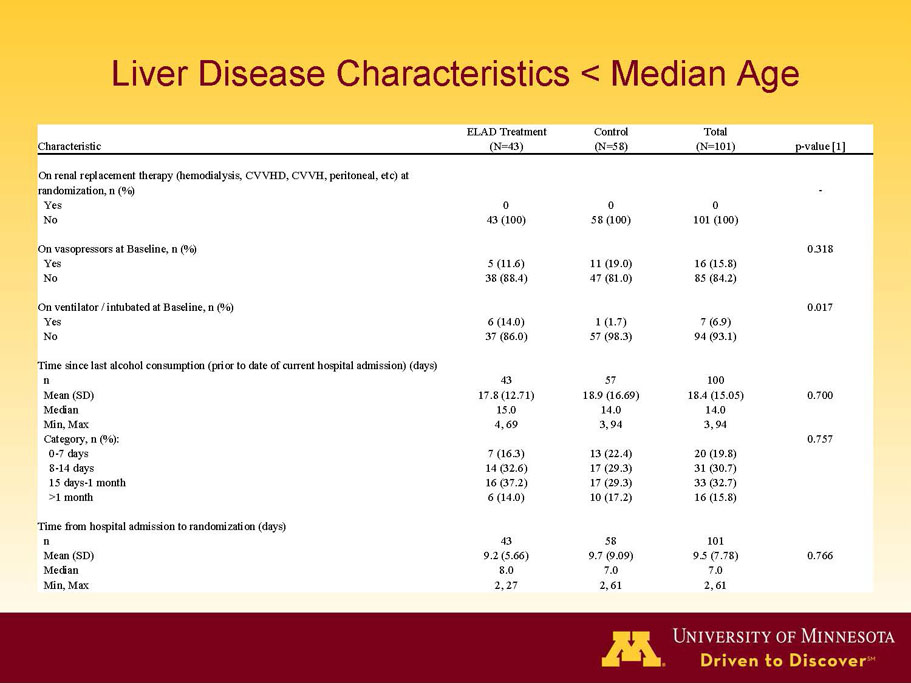
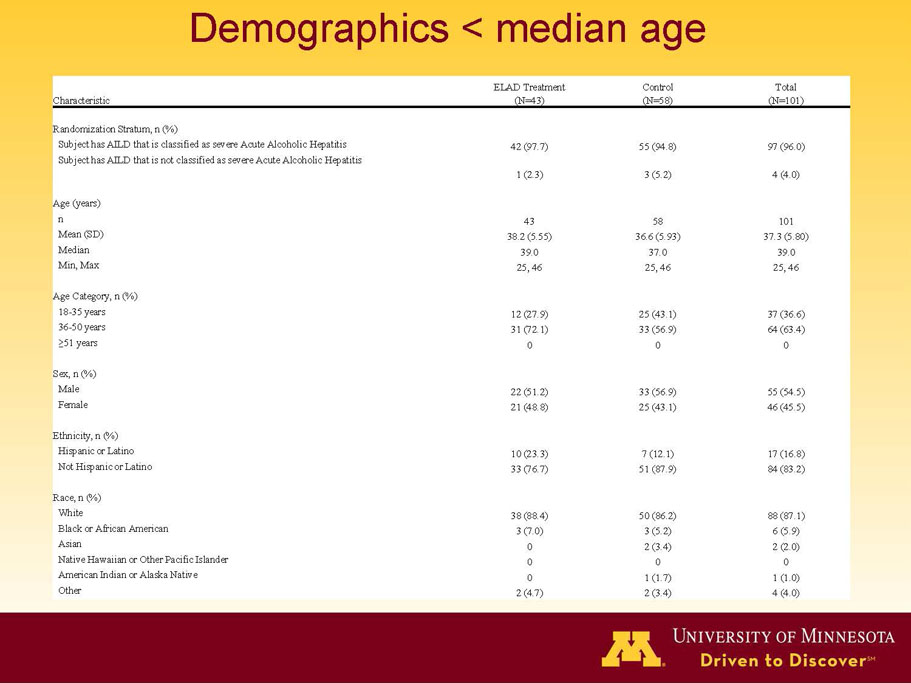
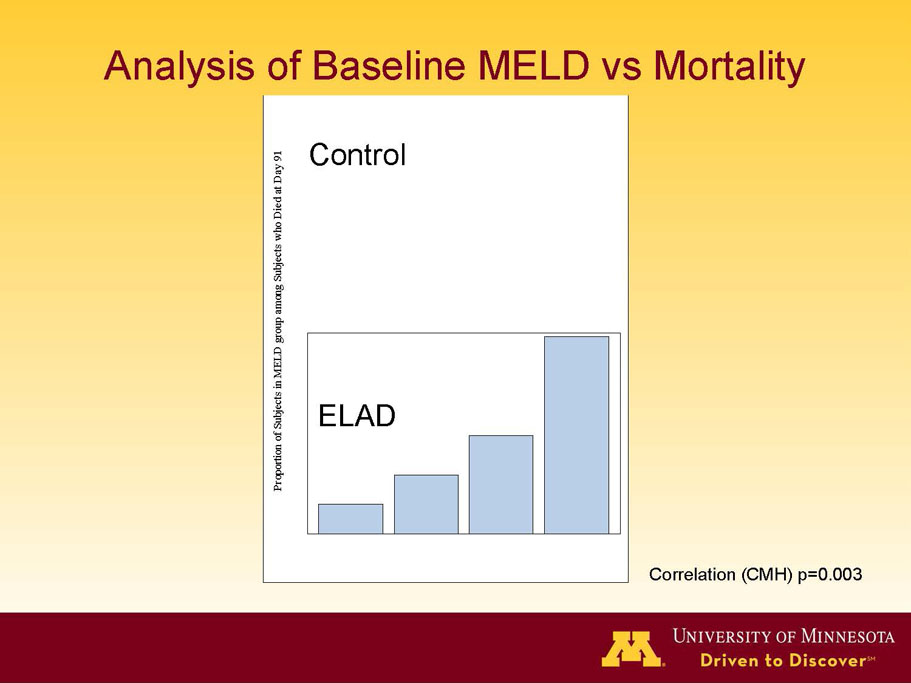
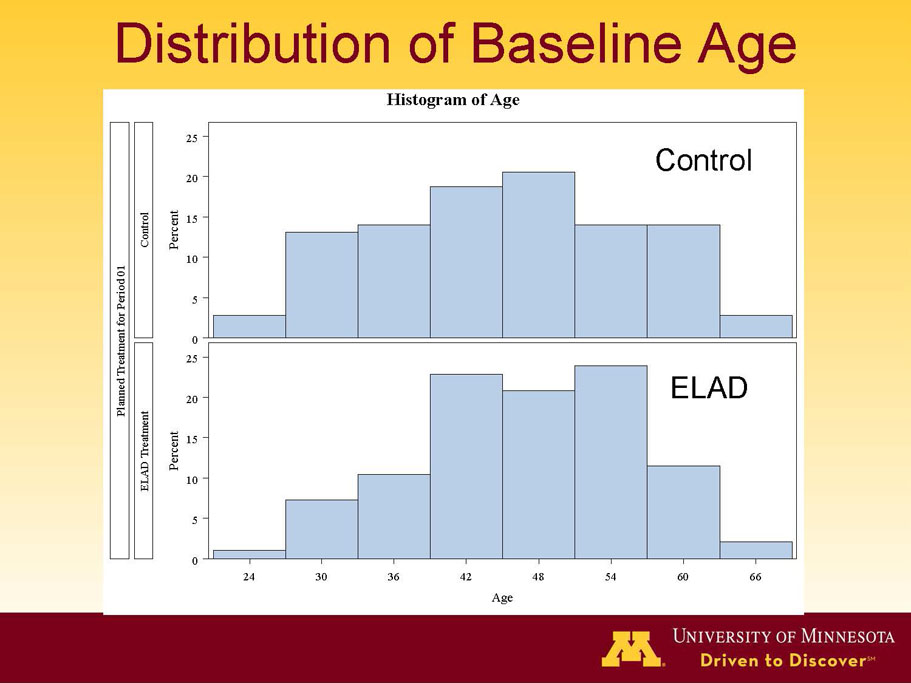
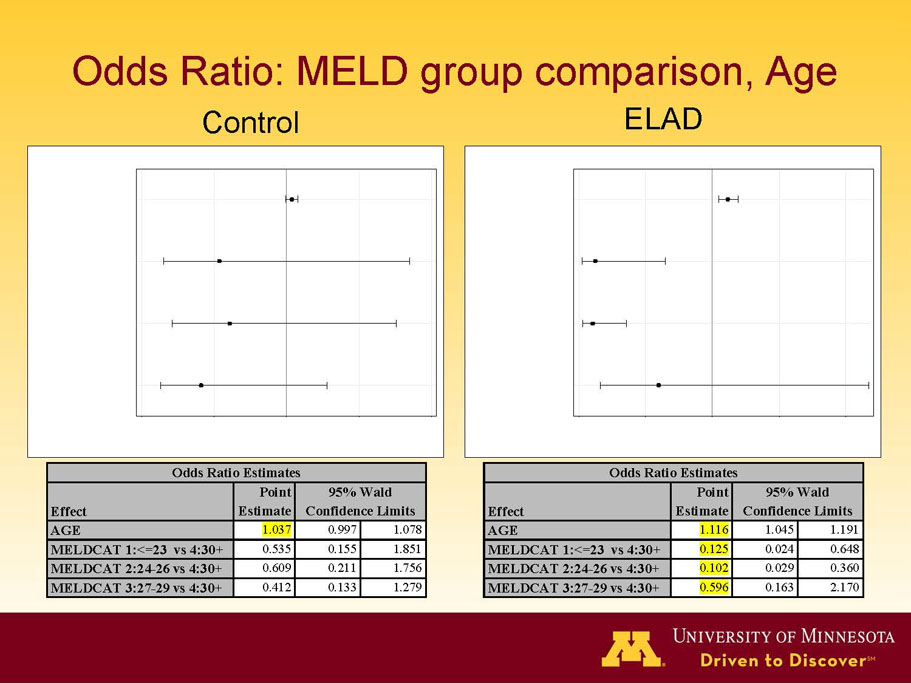
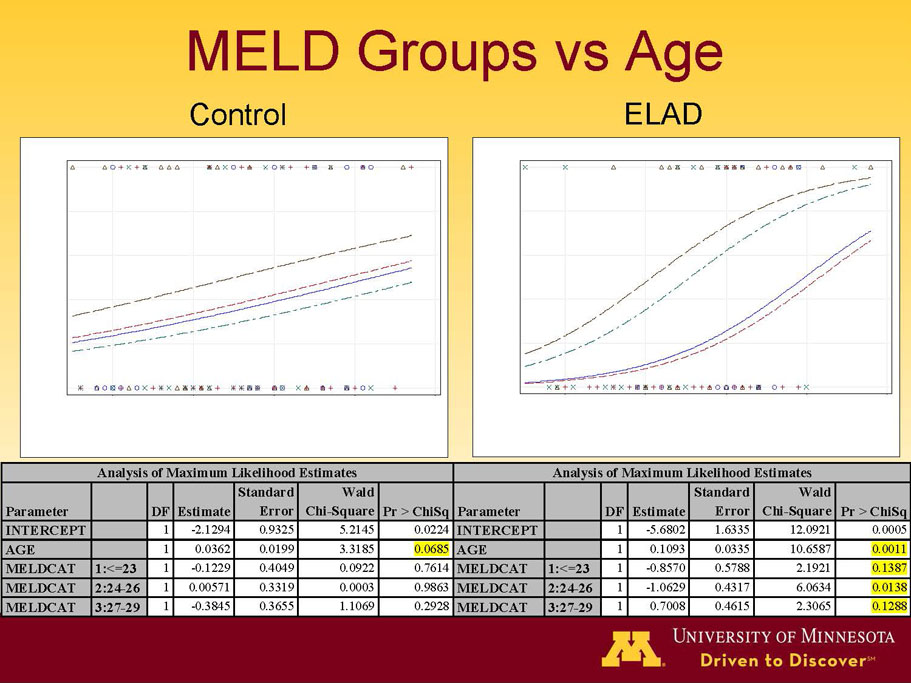
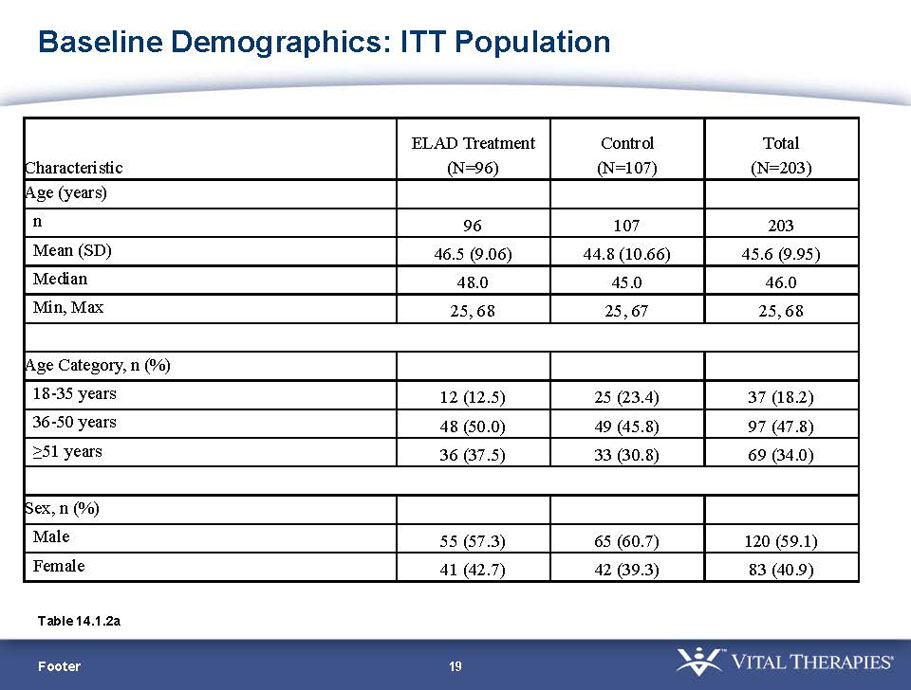
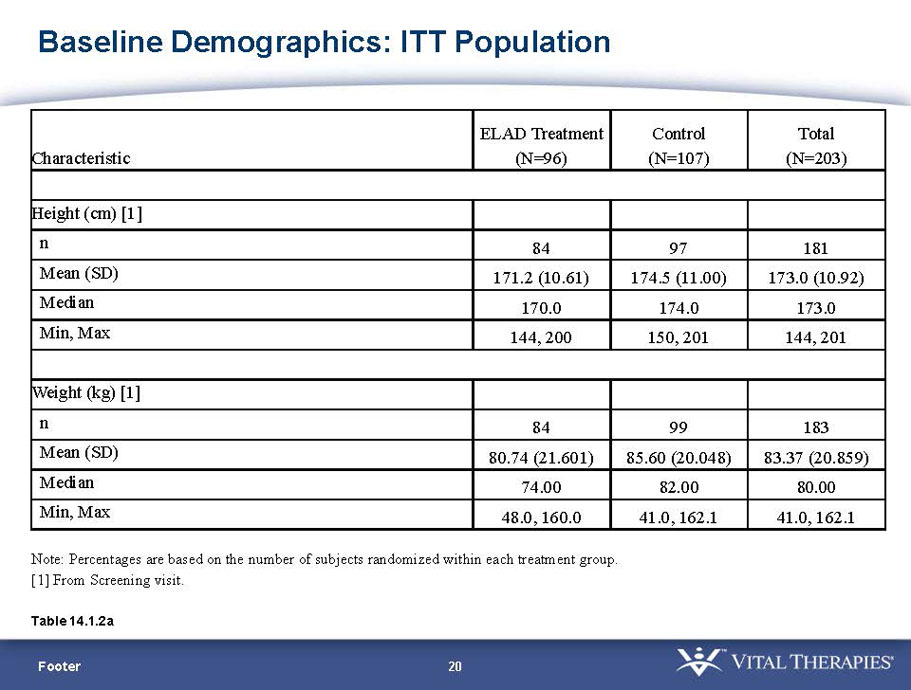
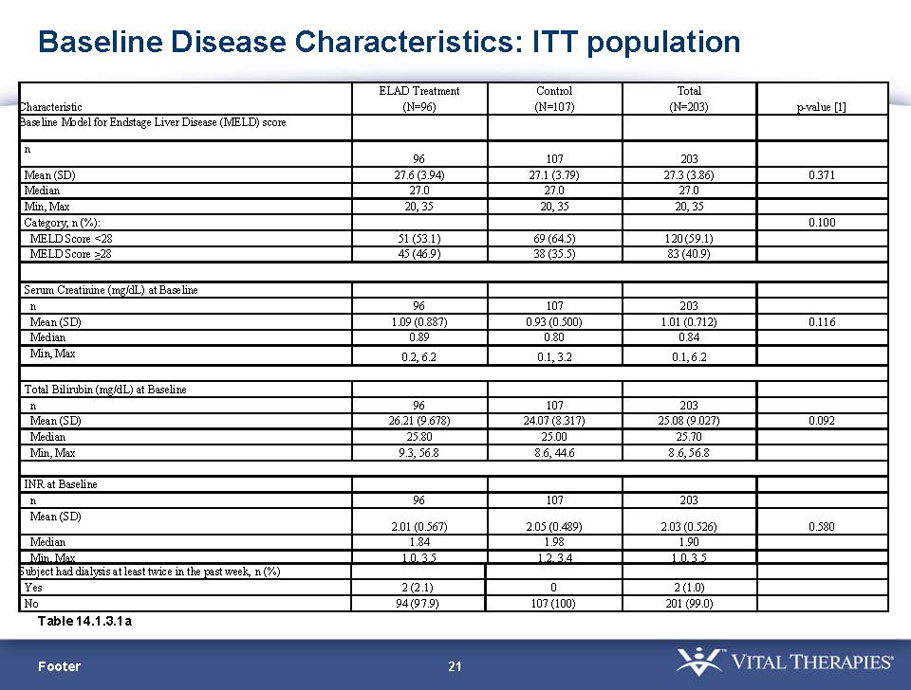
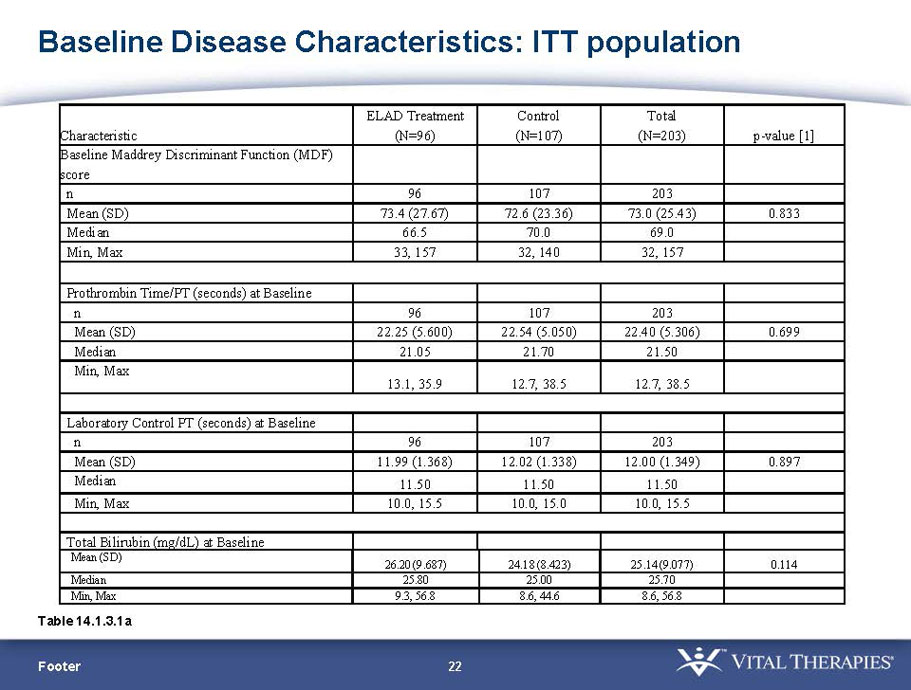
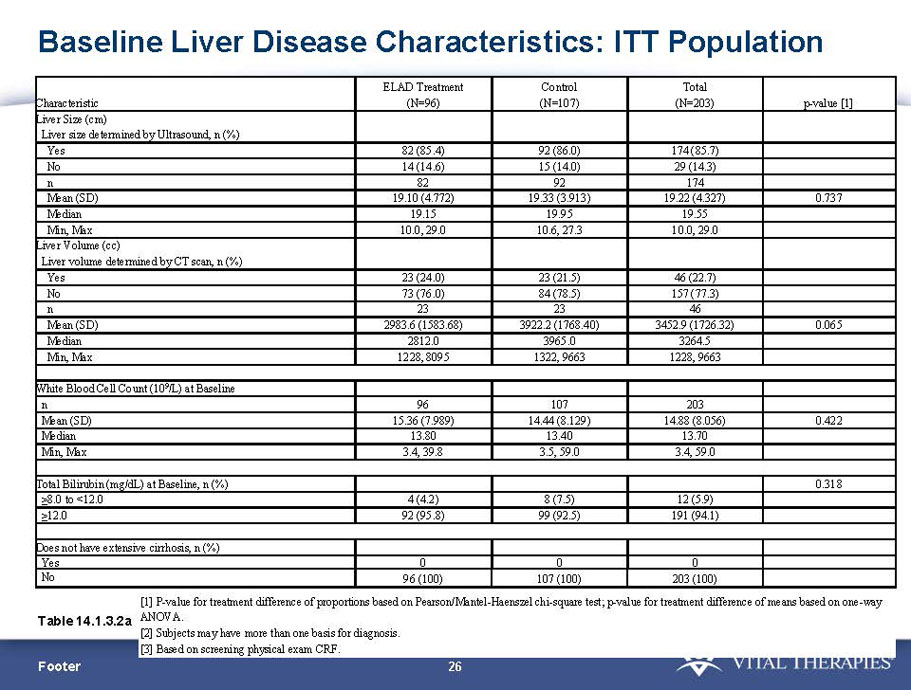
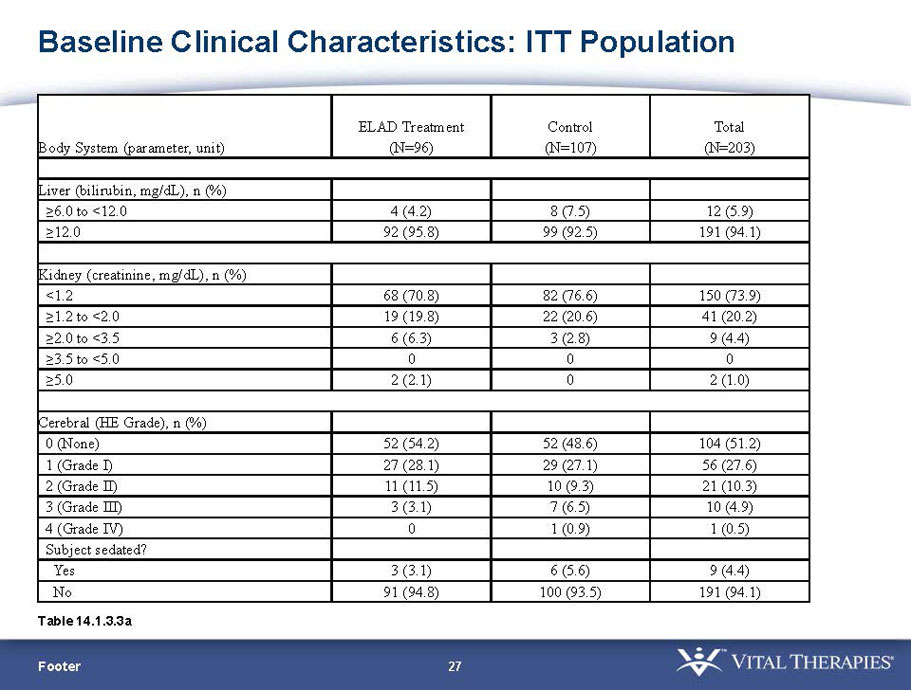
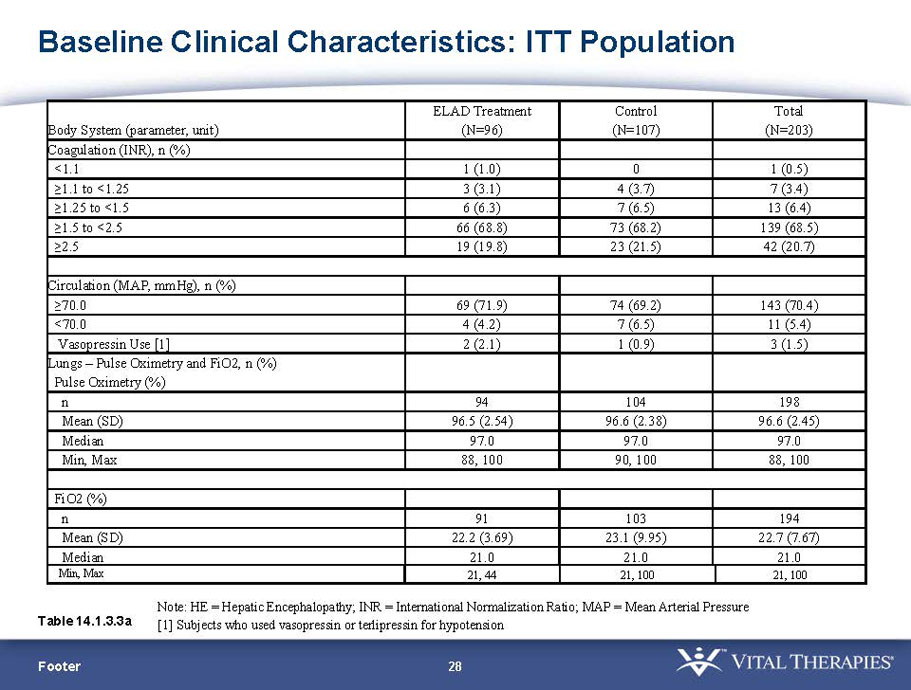
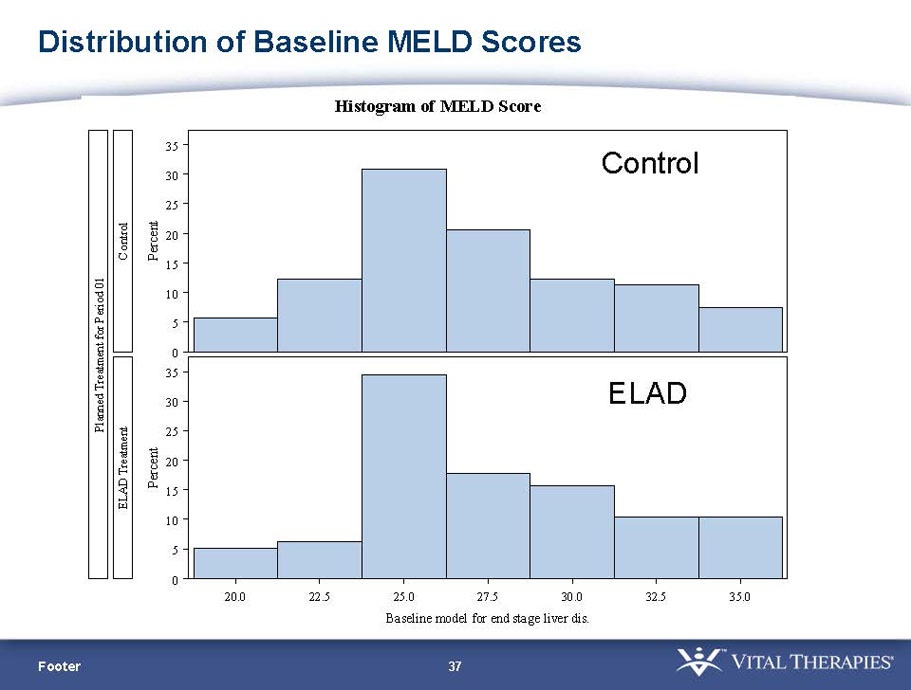
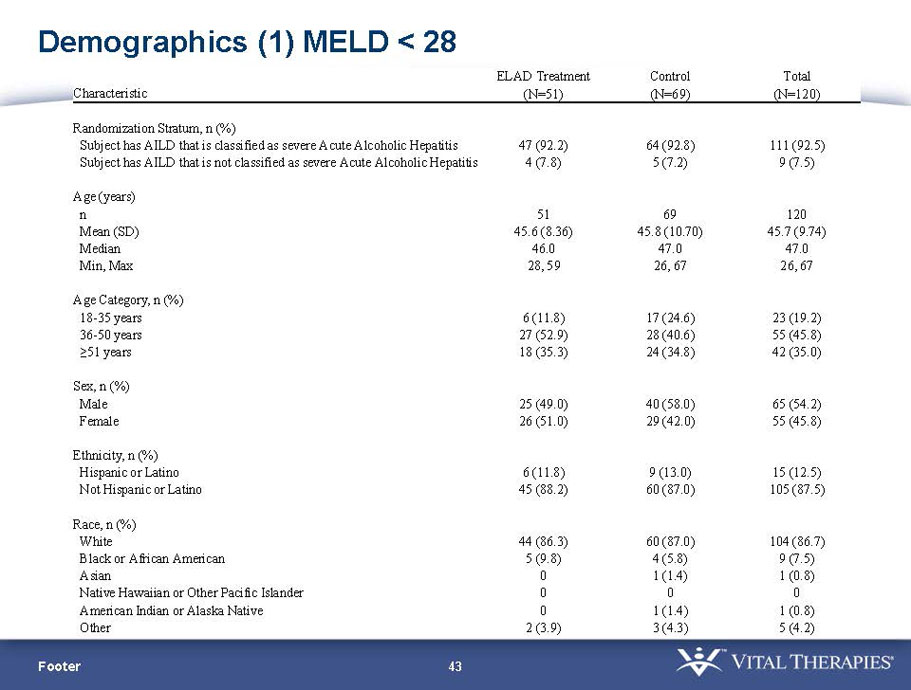
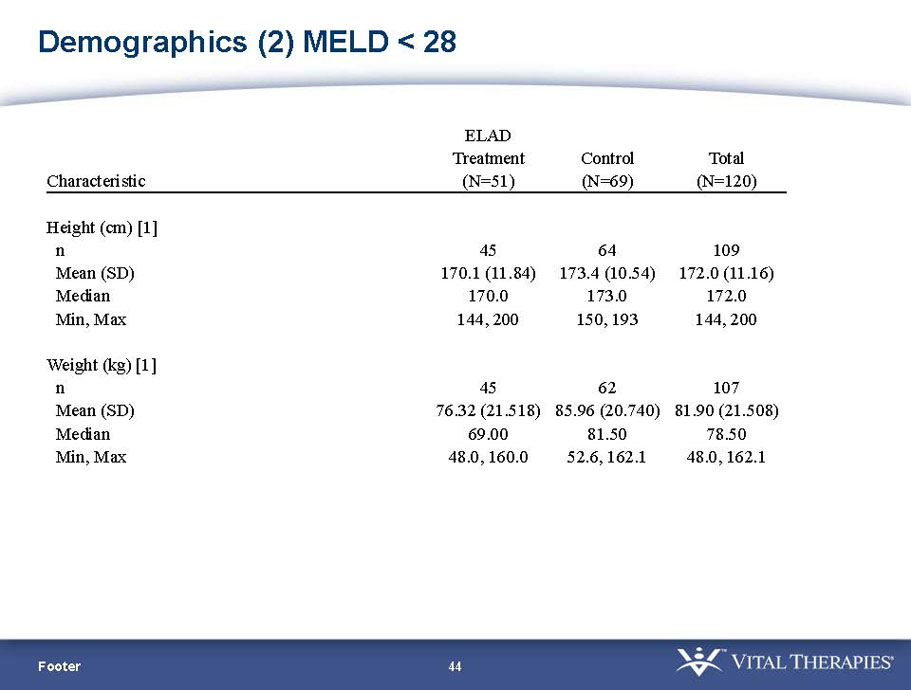
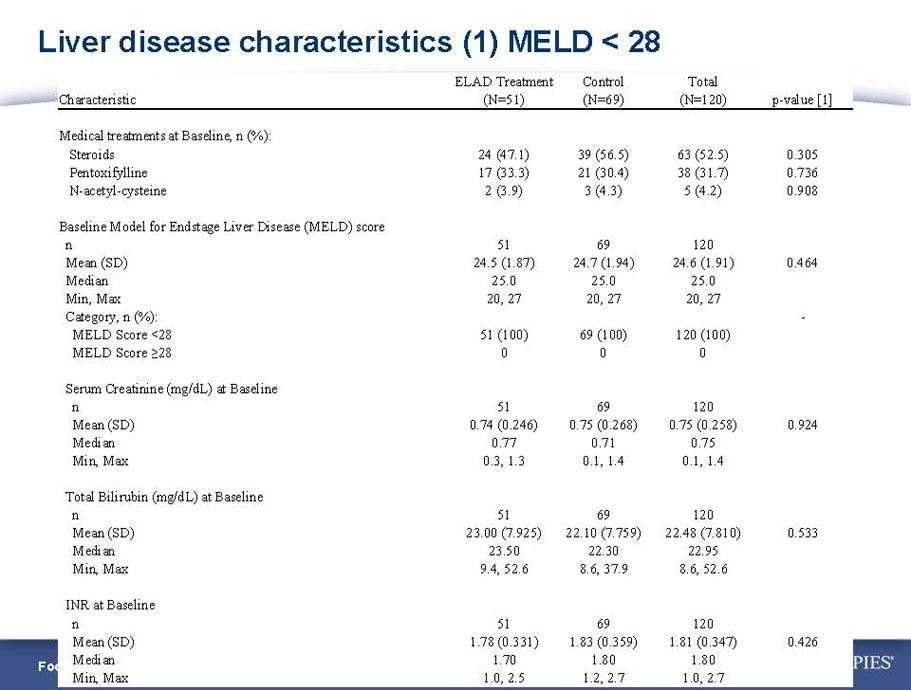
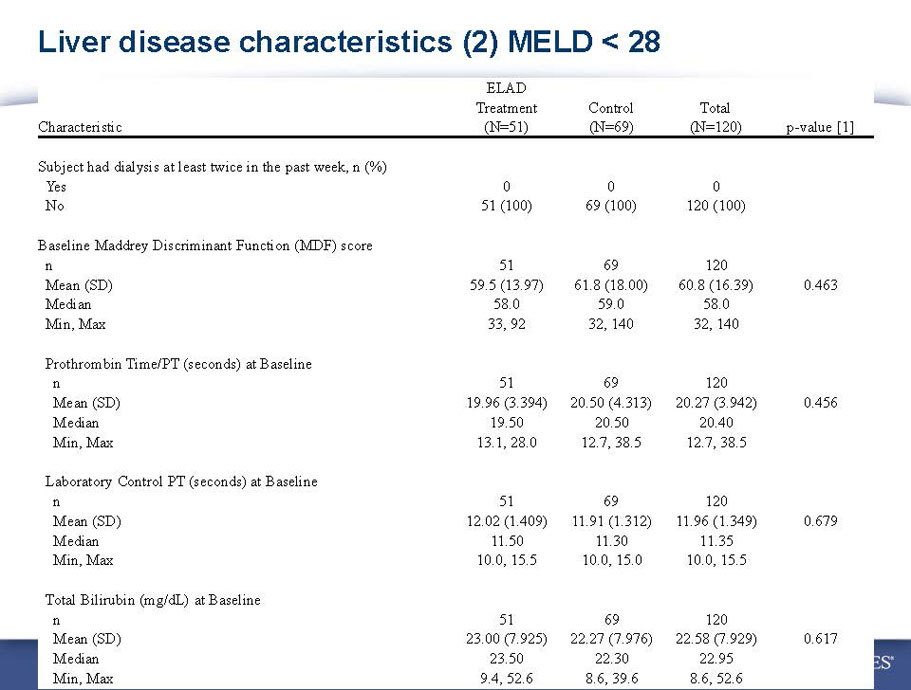
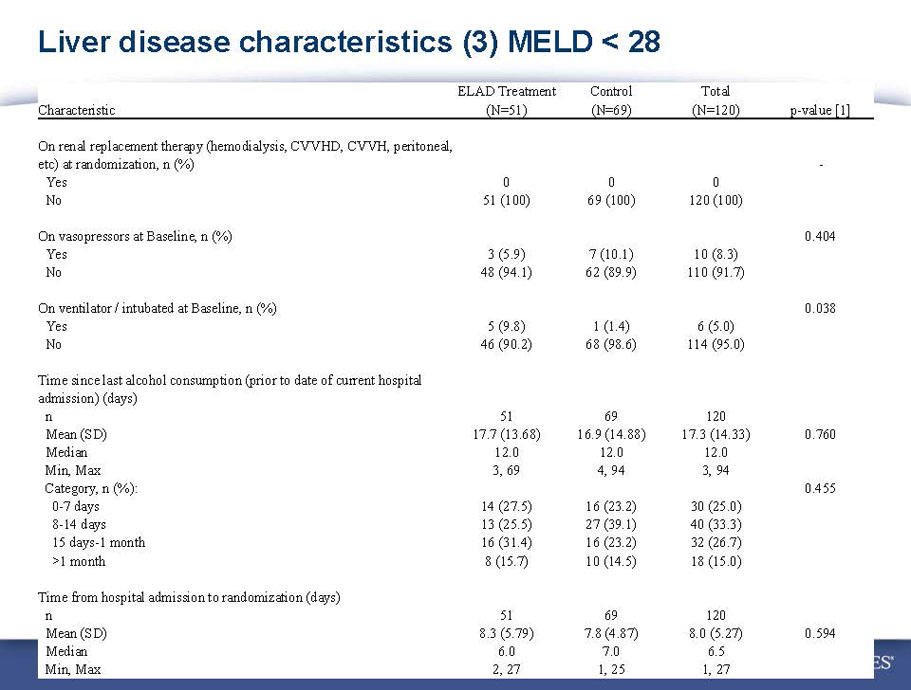
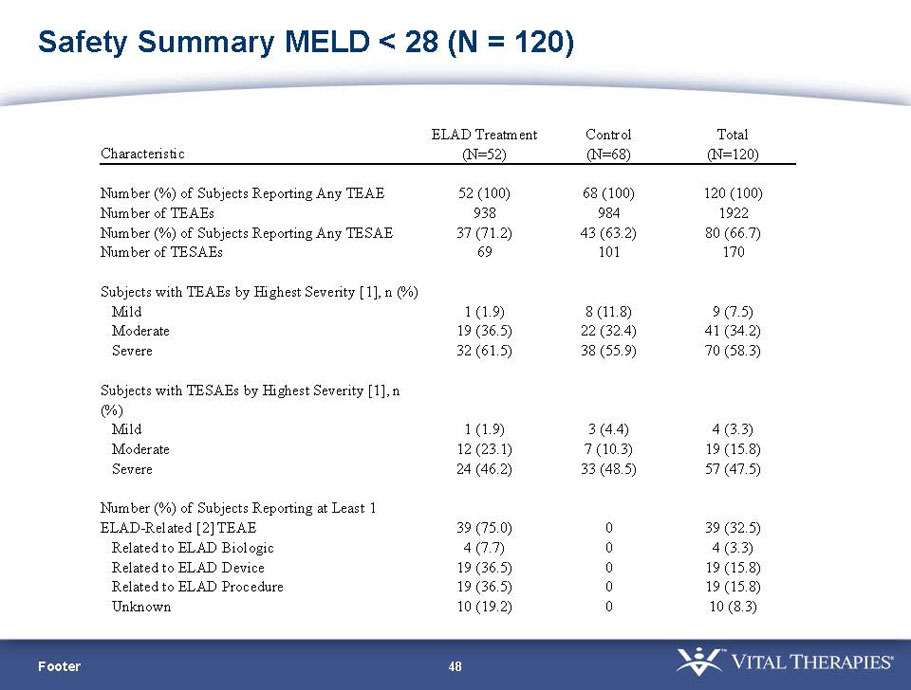
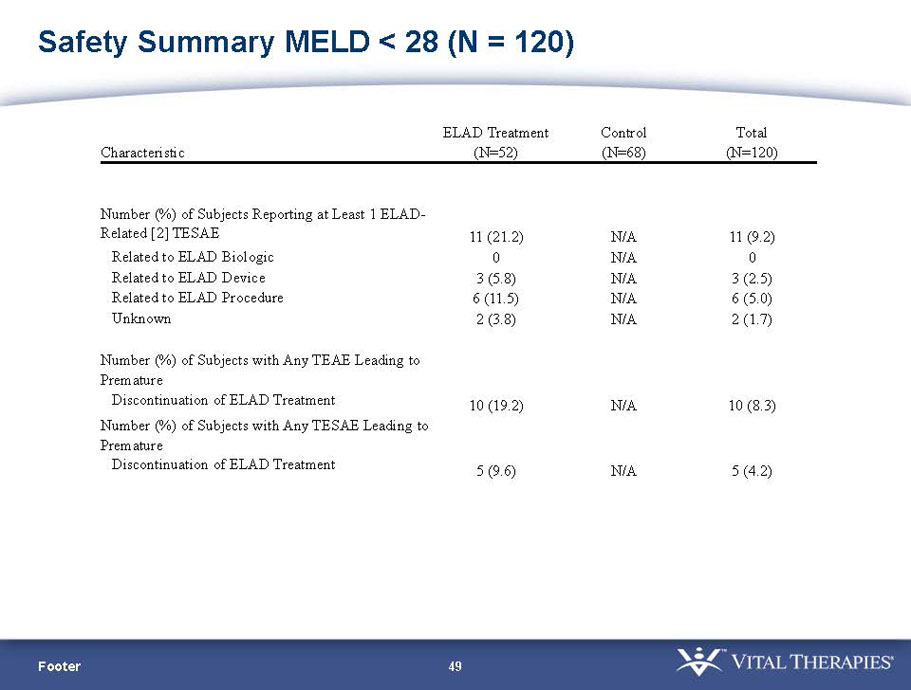
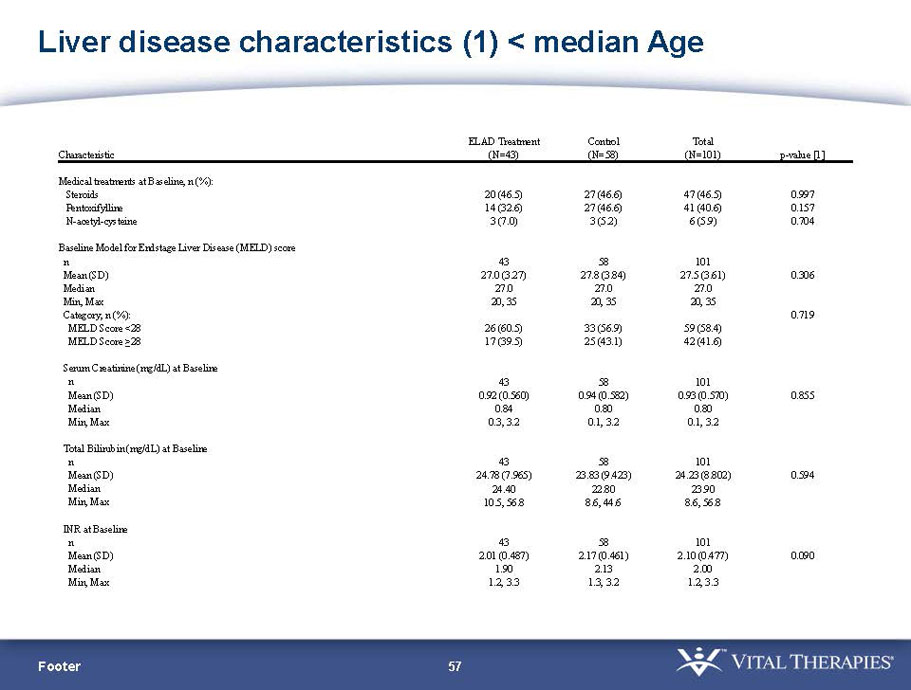
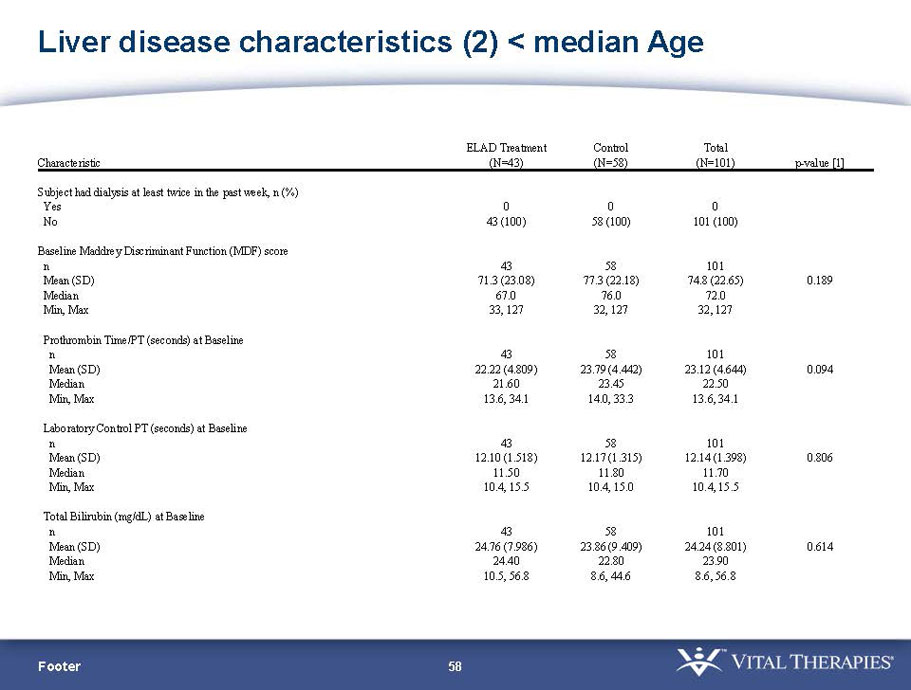
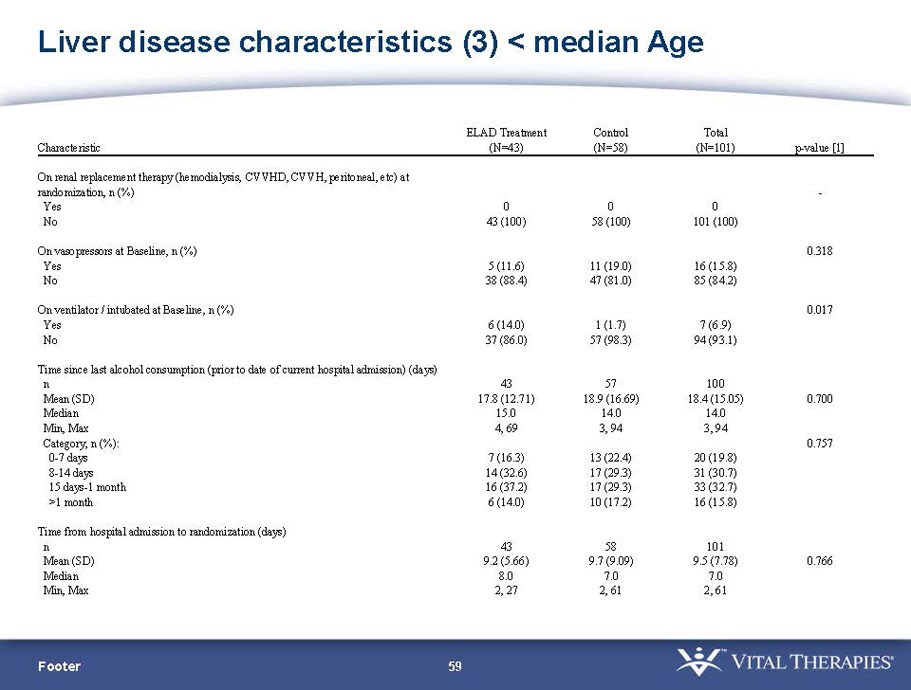
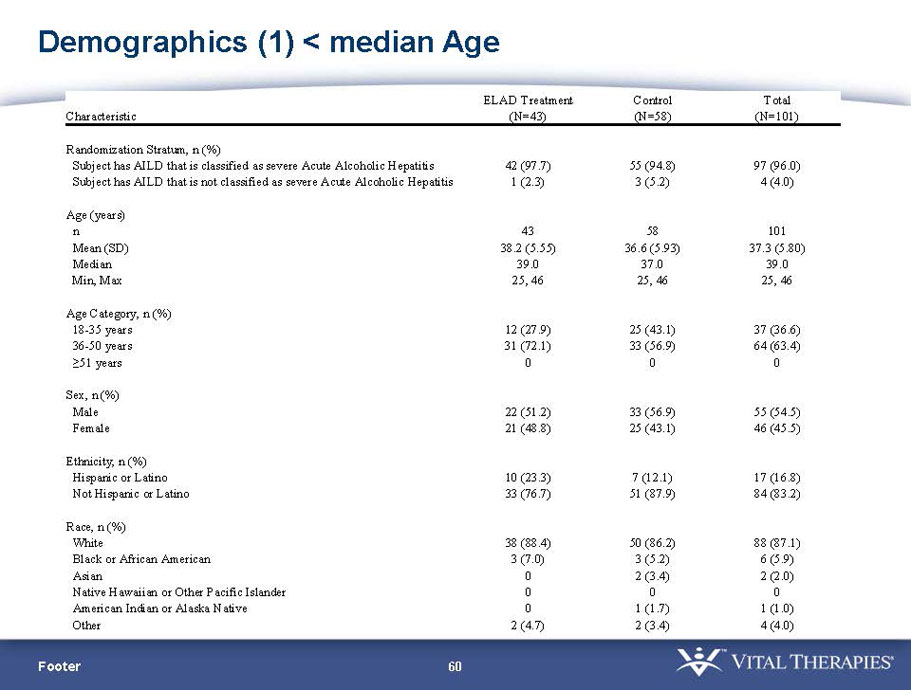
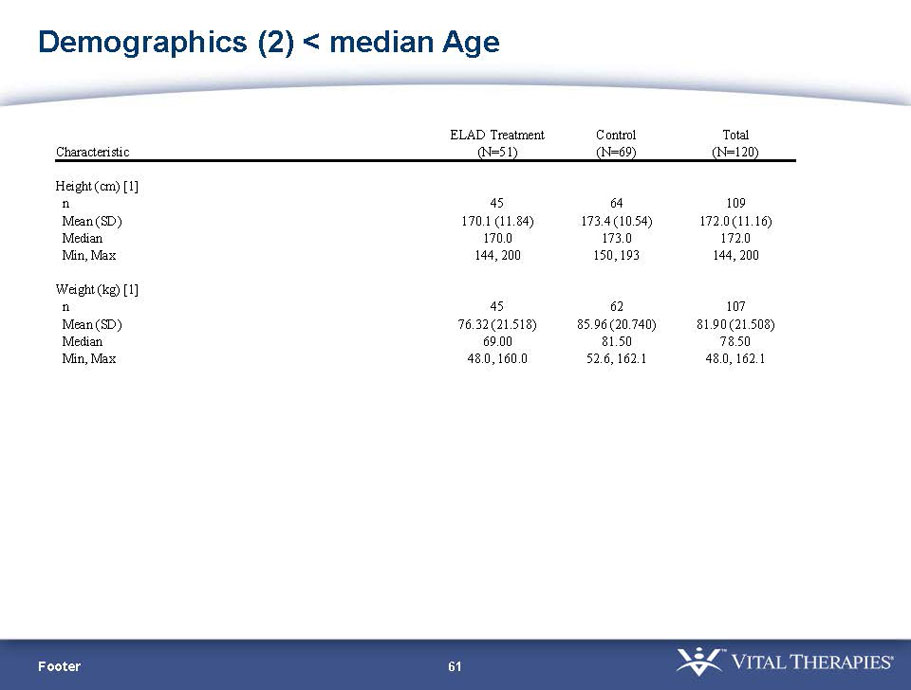
- A review of baseline characteristics and demographics of two of the pre-specified subsets: subjects with baseline Model for End-stage Liver Disease (MELD) score under 28, and age less than the median.
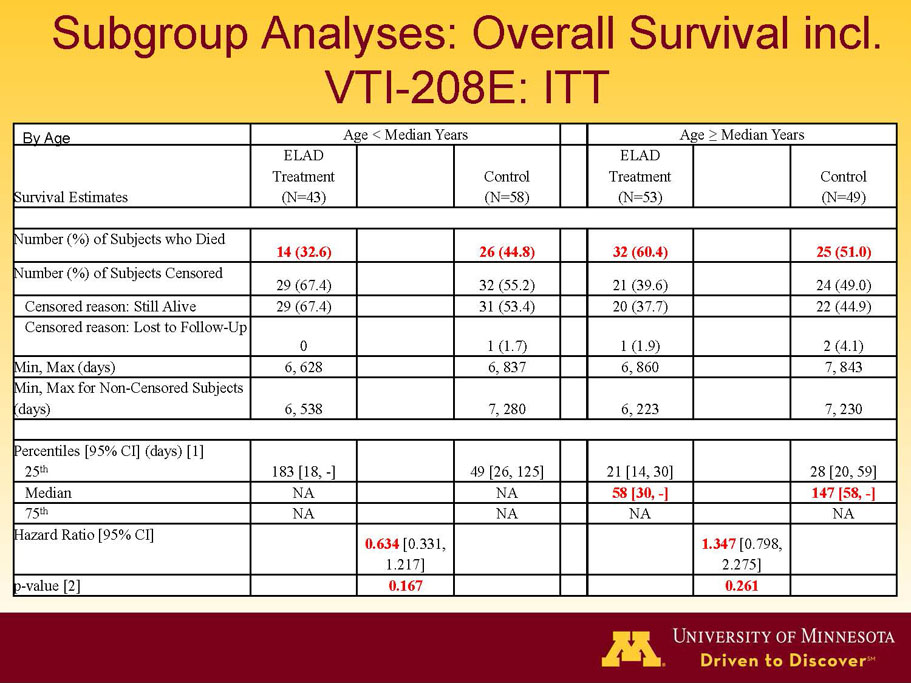
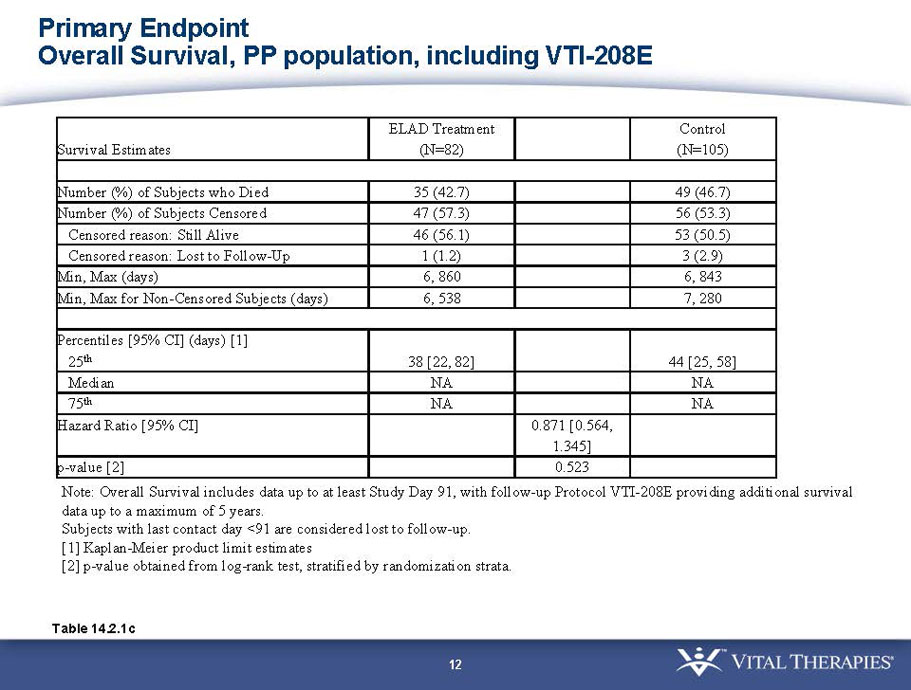
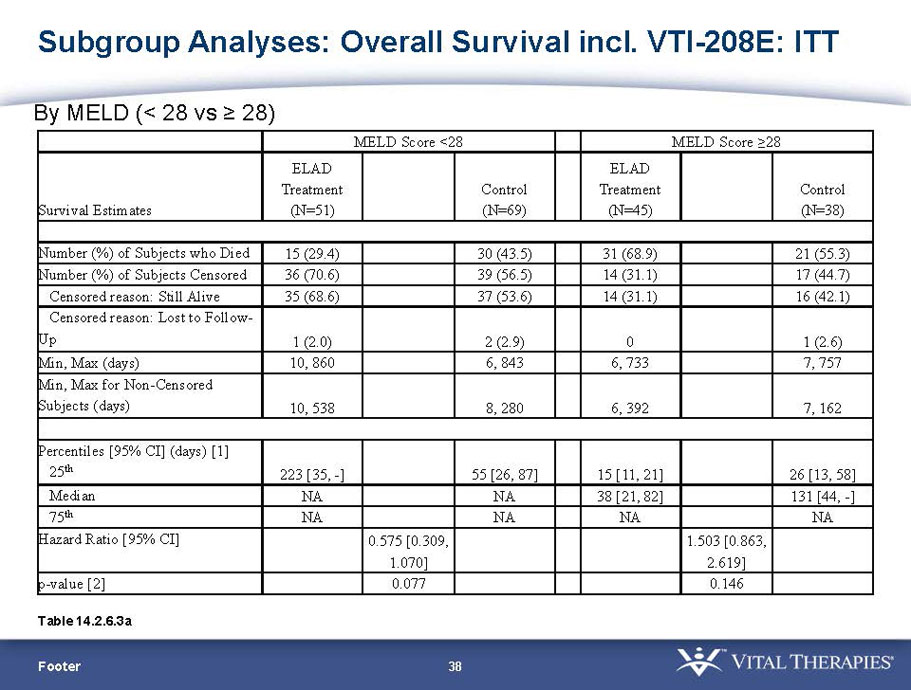
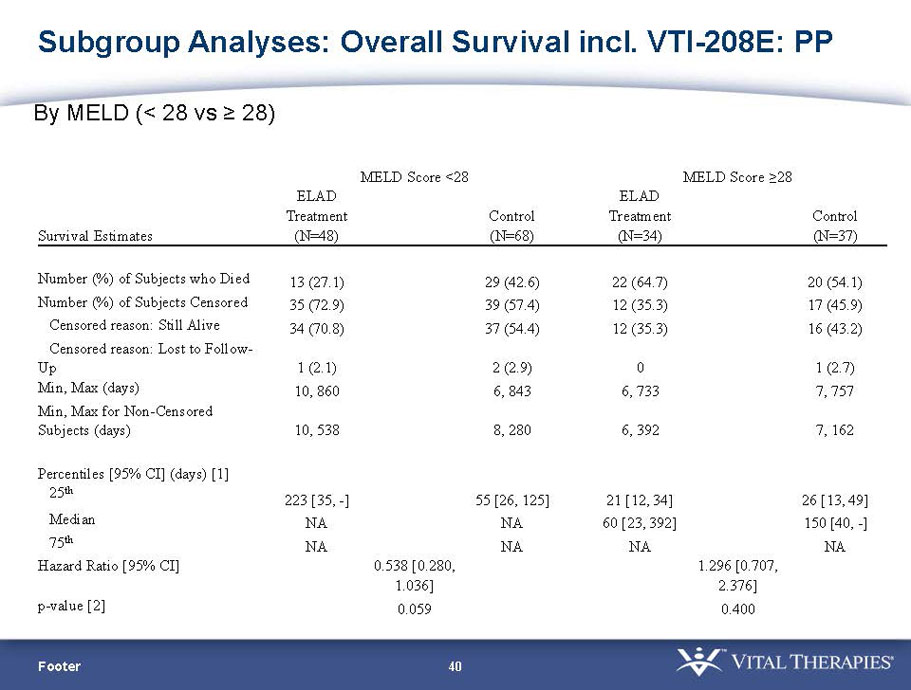
- The outcomes in these two pre-specified subsets of MELD under 28 and age less than median are in the following table:
| Subjects |
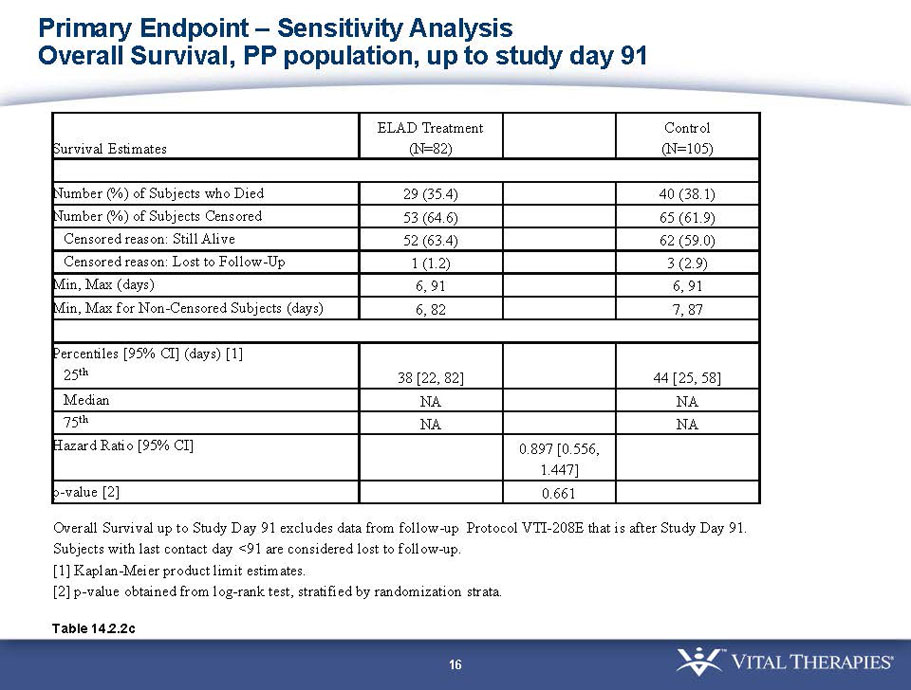
Pre-defined primary endpoint parameter: Kaplan Meier survivor analysis (log-rank) with a min. follow up of 91 days (incorporating VTI-208E data)
|
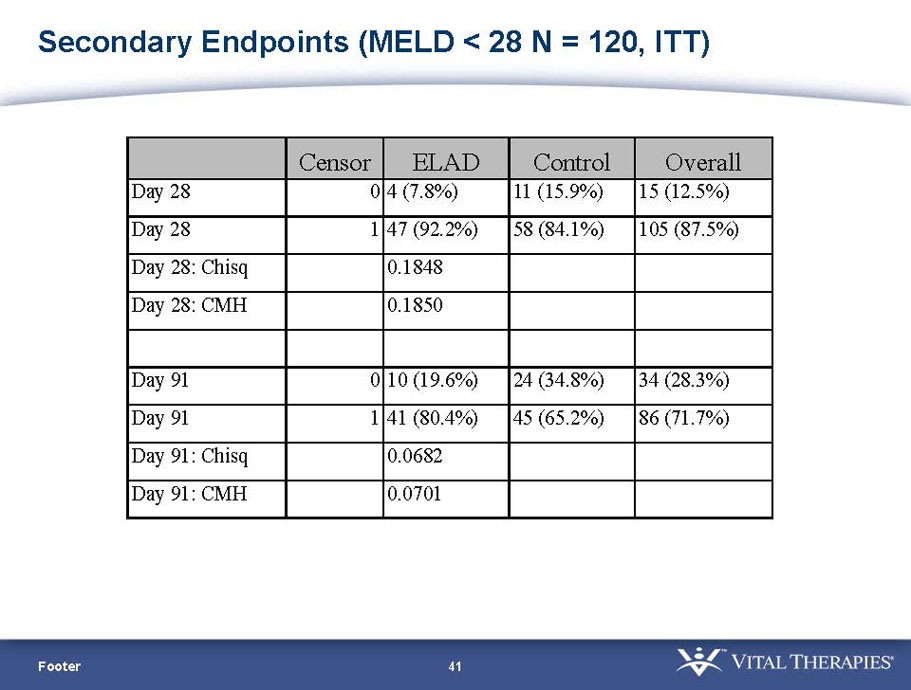
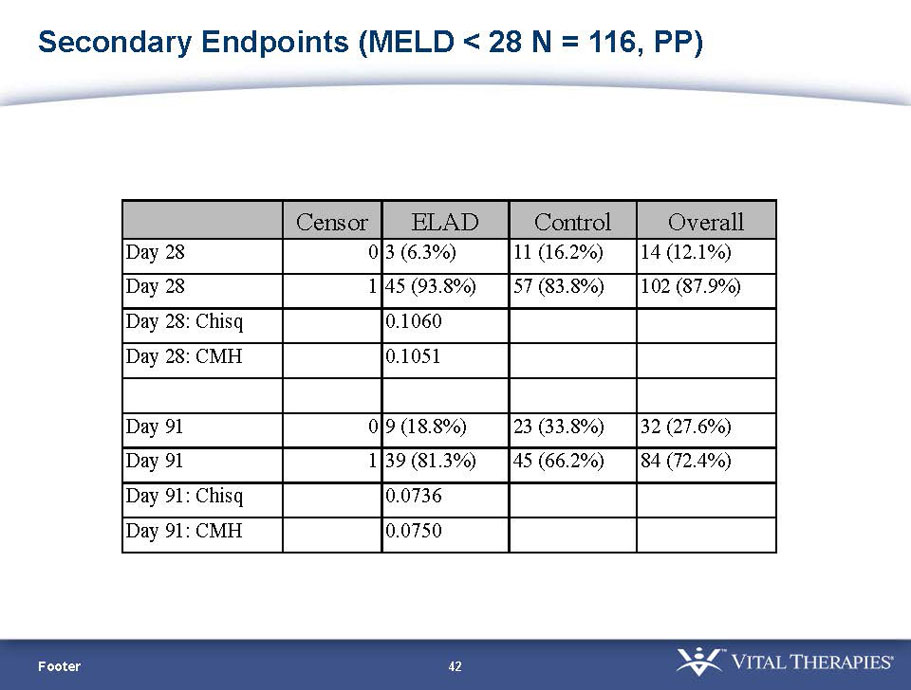
Pre-defined secondary endpoint parameter: Proportion survivor analysis (Pearson’s chi-squared) at day 91
| |||||||
| Total | ELAD | Control | Hazard Ratio | P-value | ELAD survival | Control Survival | P-value | ||
| MELD < 28 | ITT | 120 | 51 | 69 | 0.575 | 0.077 (NS) | 80.4% | 65.2% | 0.068 (NS) |
| PP | 116 | 48 | 68 | 0.538 | 0.059 (NS) | 81.3% | 66.2% | 0.074 (NS) | |
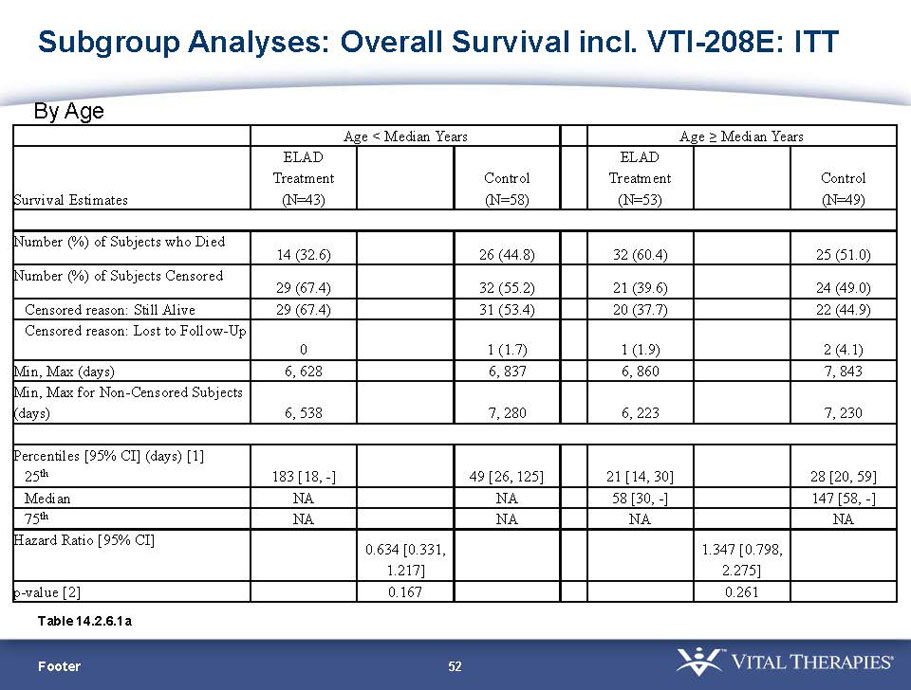
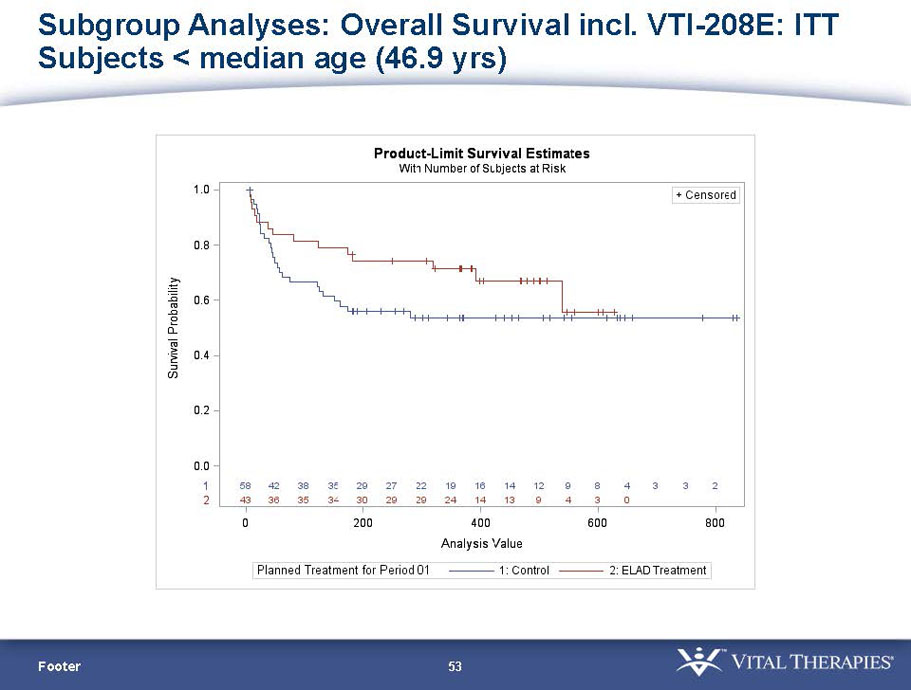
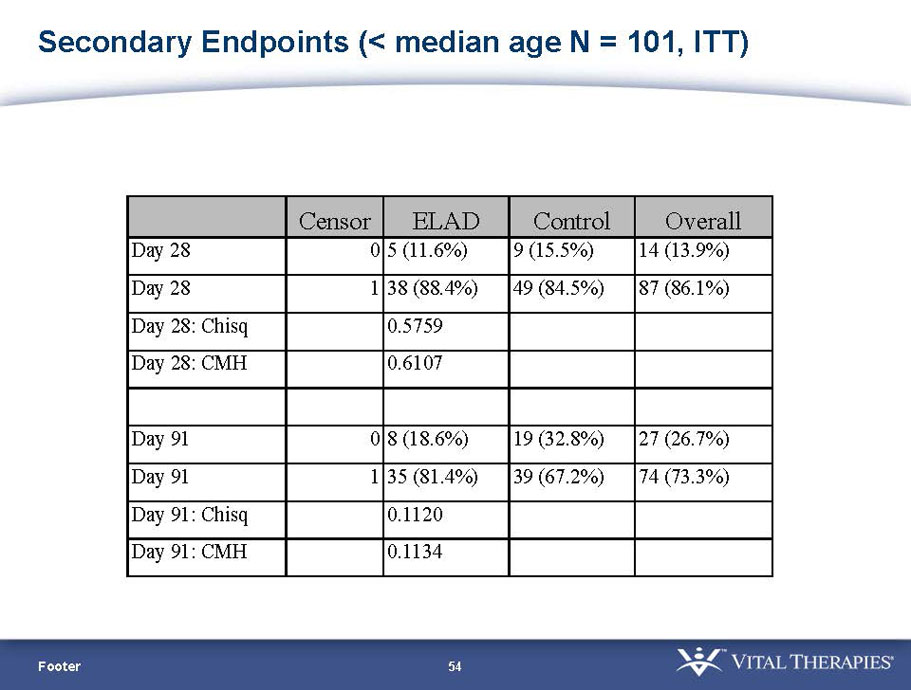
| Age < Median (46.9 yrs) | ITT | 101 | 43 | 58 | 0.634 | 0.167 (NS) | 81.4% | 67.2% | 0.112 (NS) |
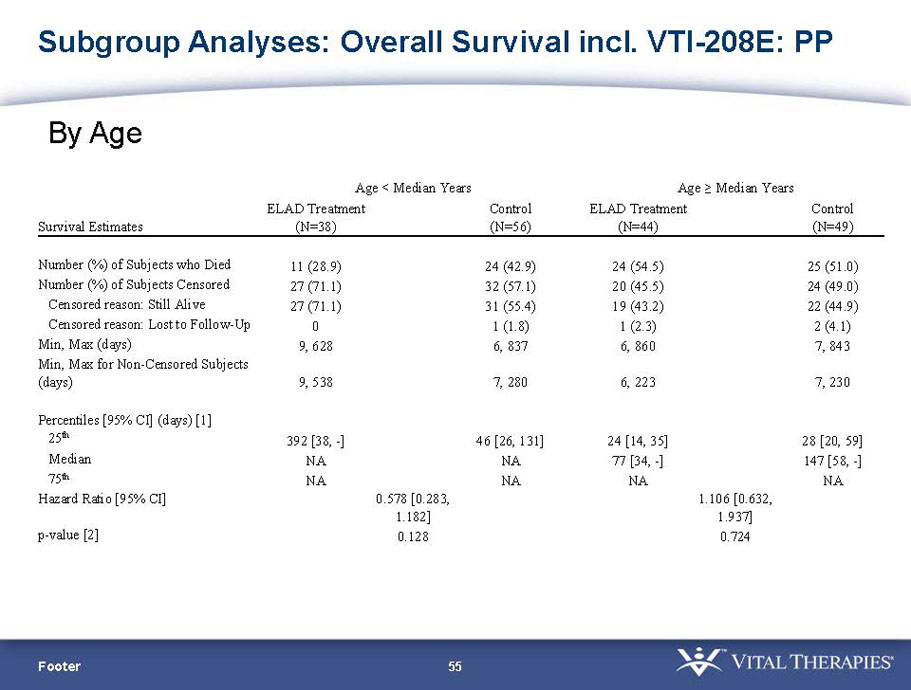
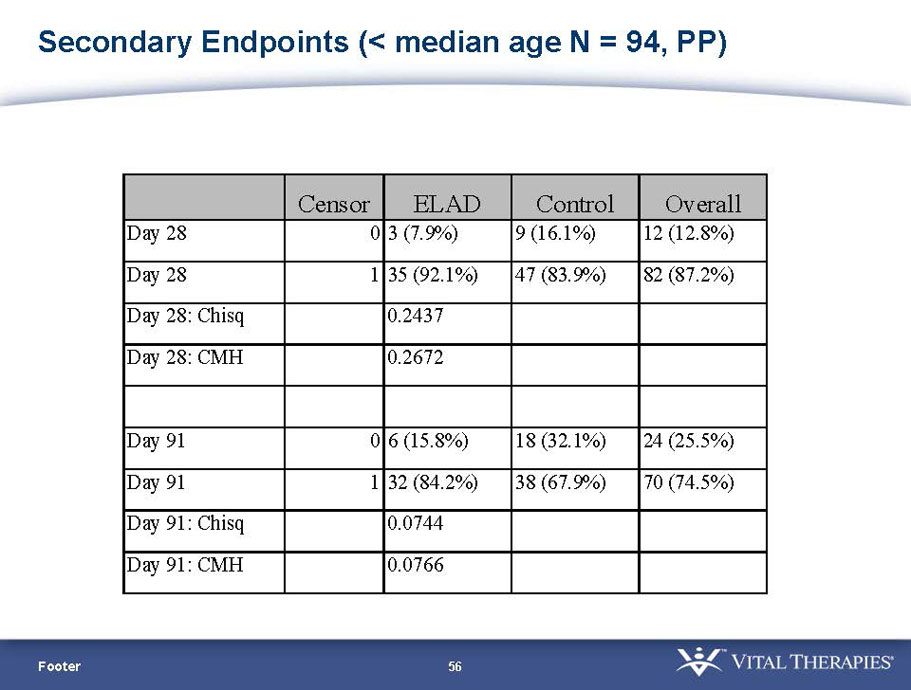
| PP | 94 | 38 | 56 | 0.578 | 0.128 (NS) | 84.2% | 67.9% | 0.074 (NS) | |
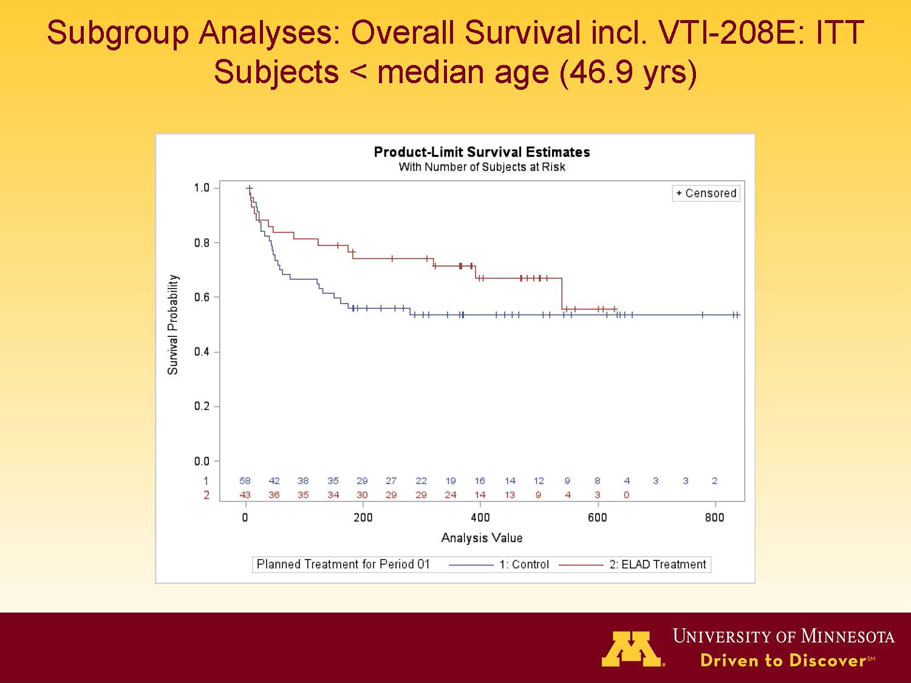
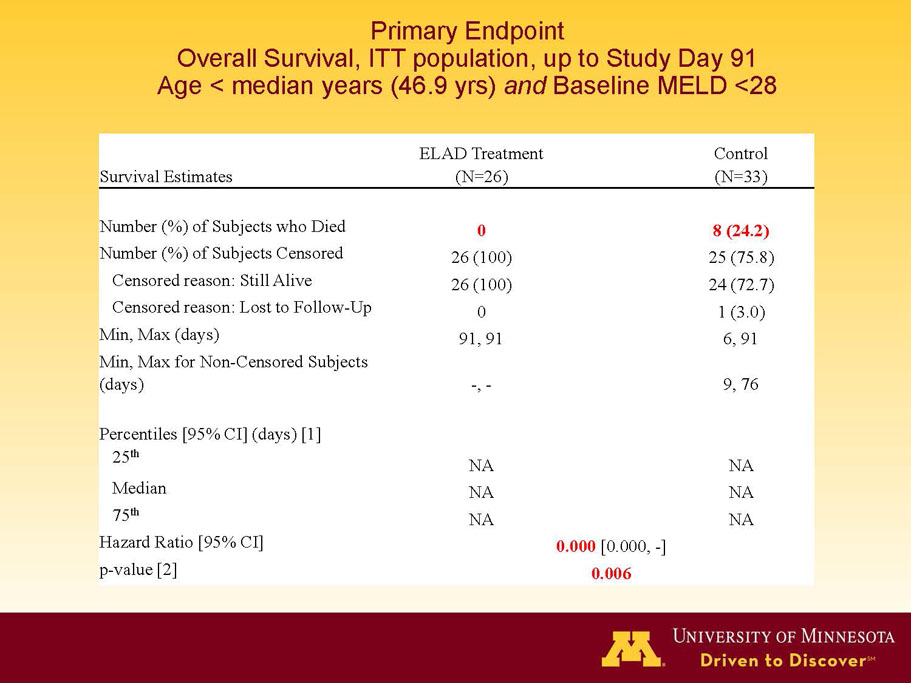
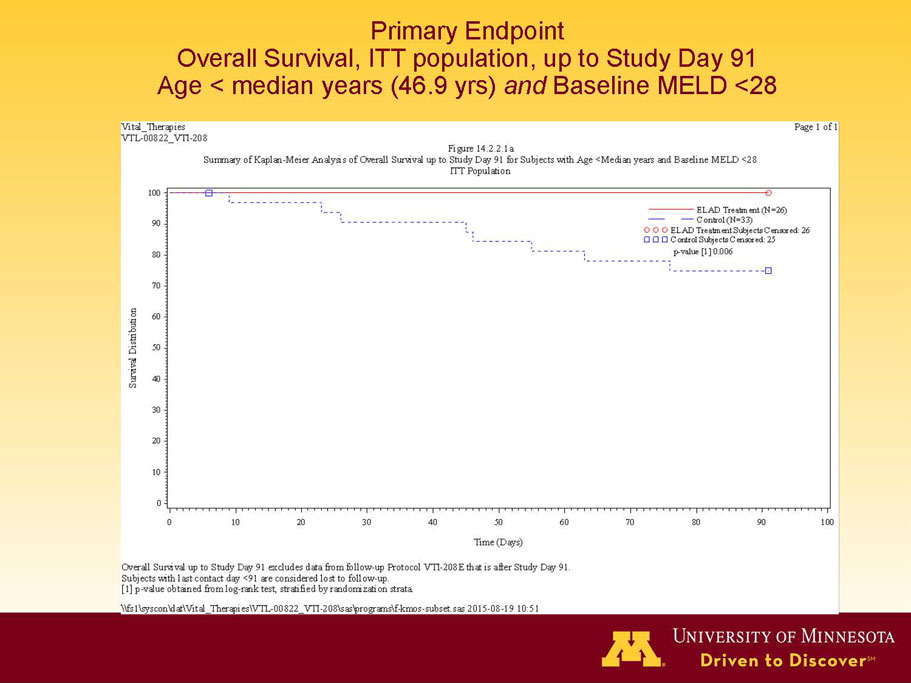
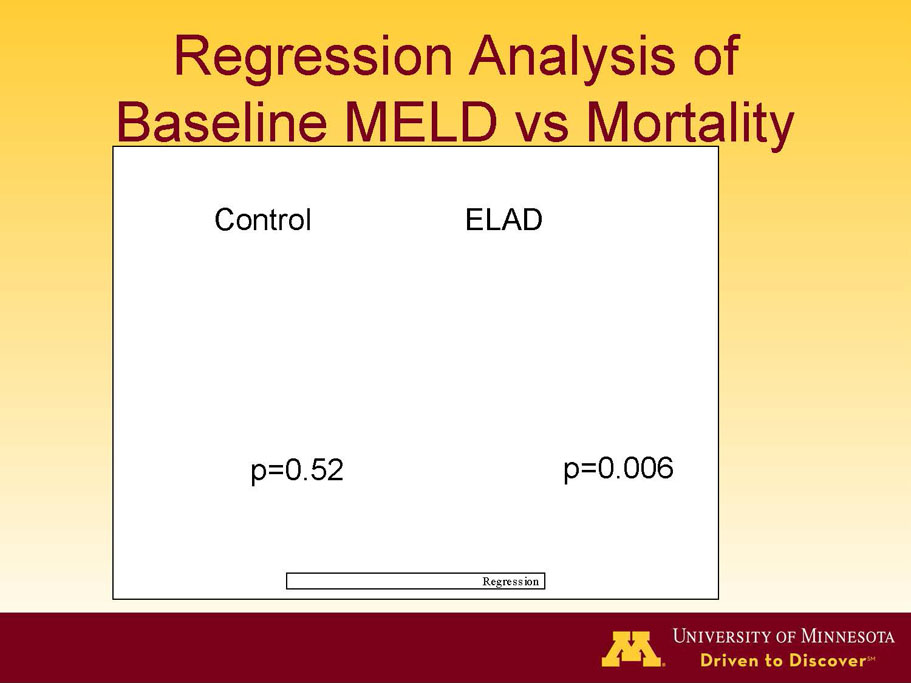
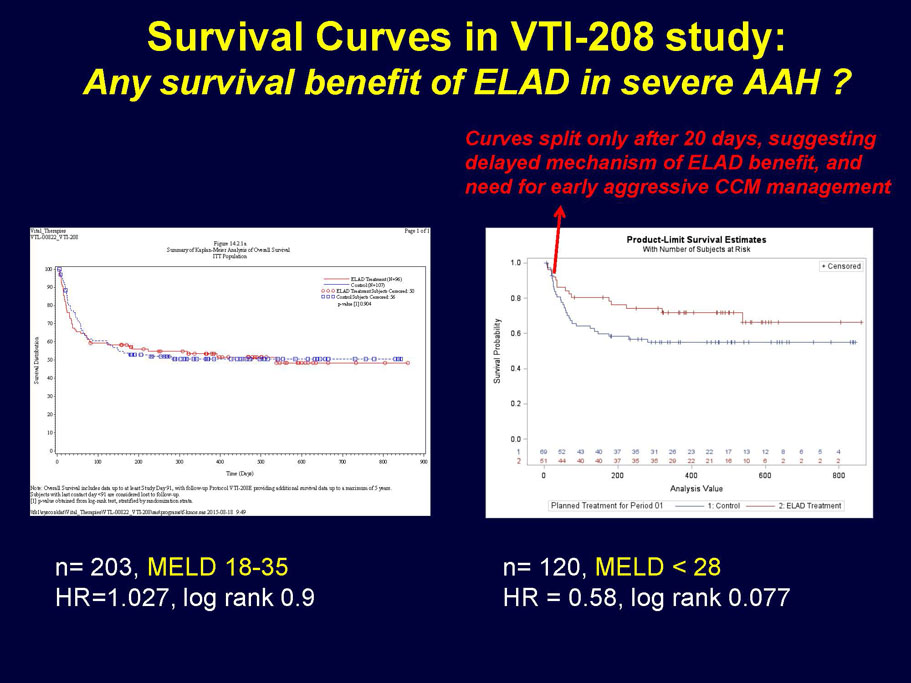
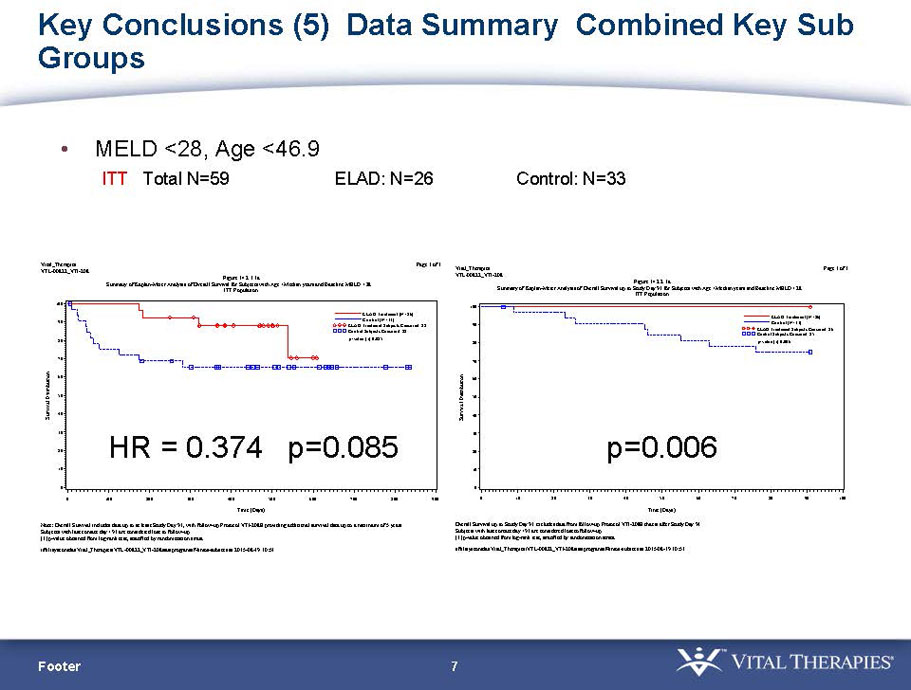
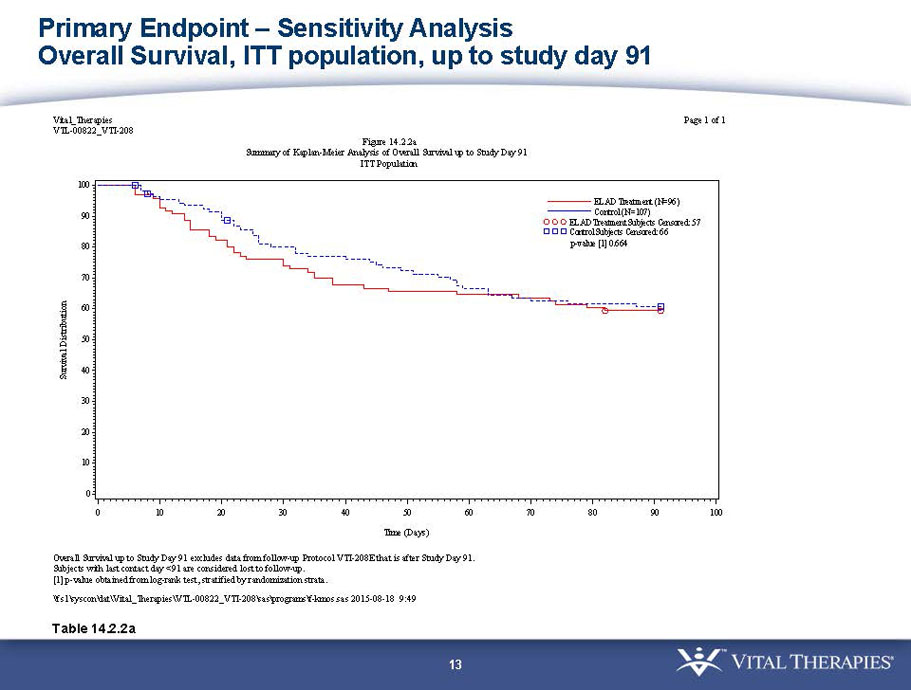
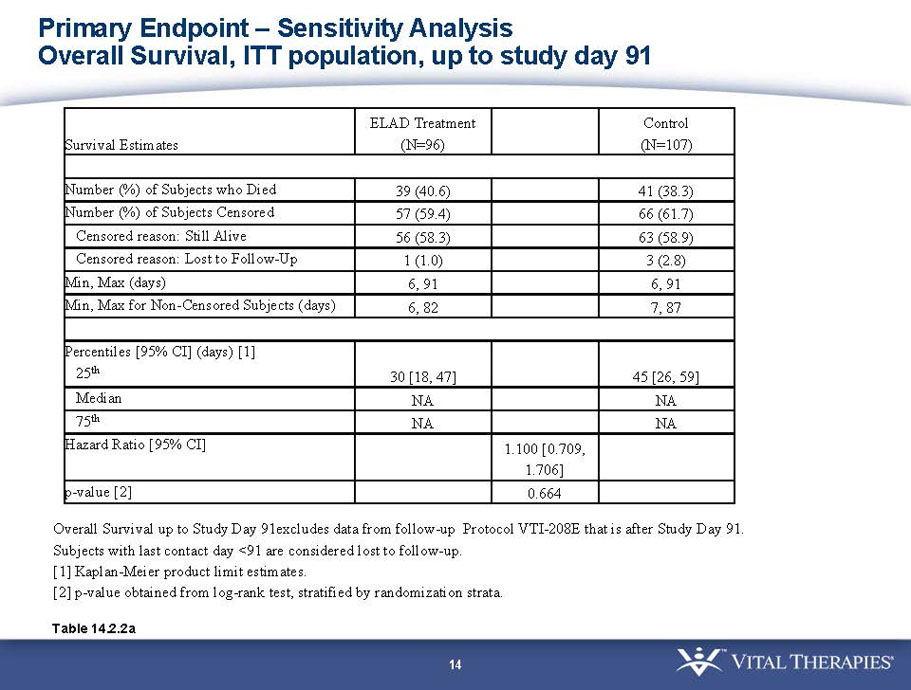
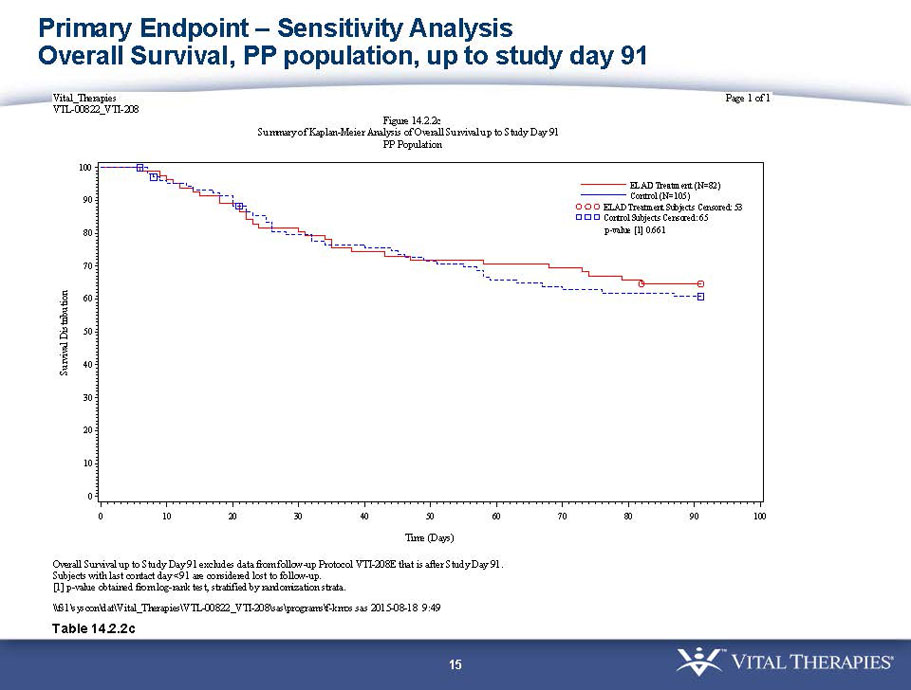
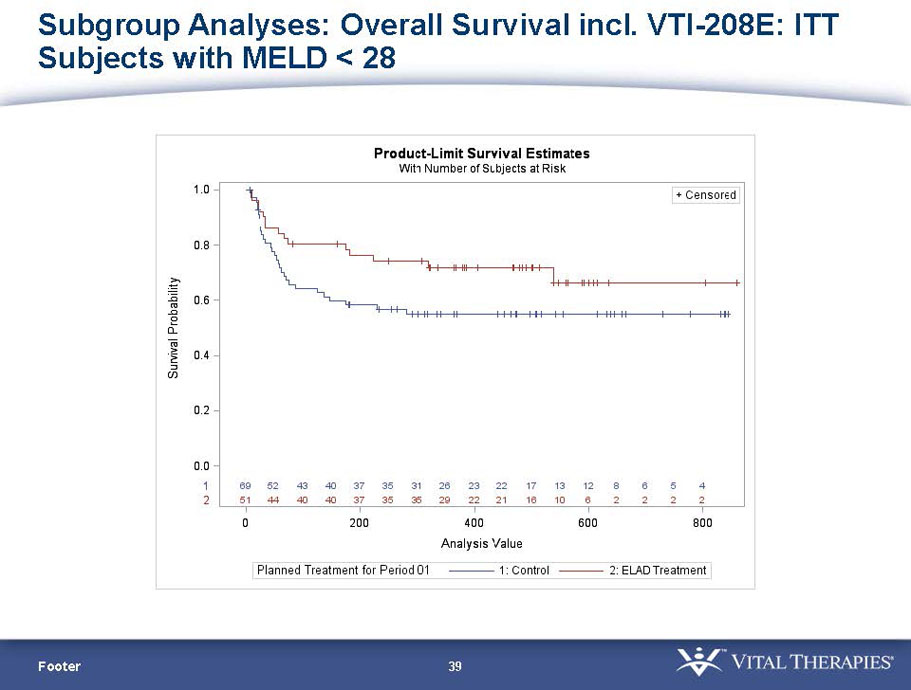
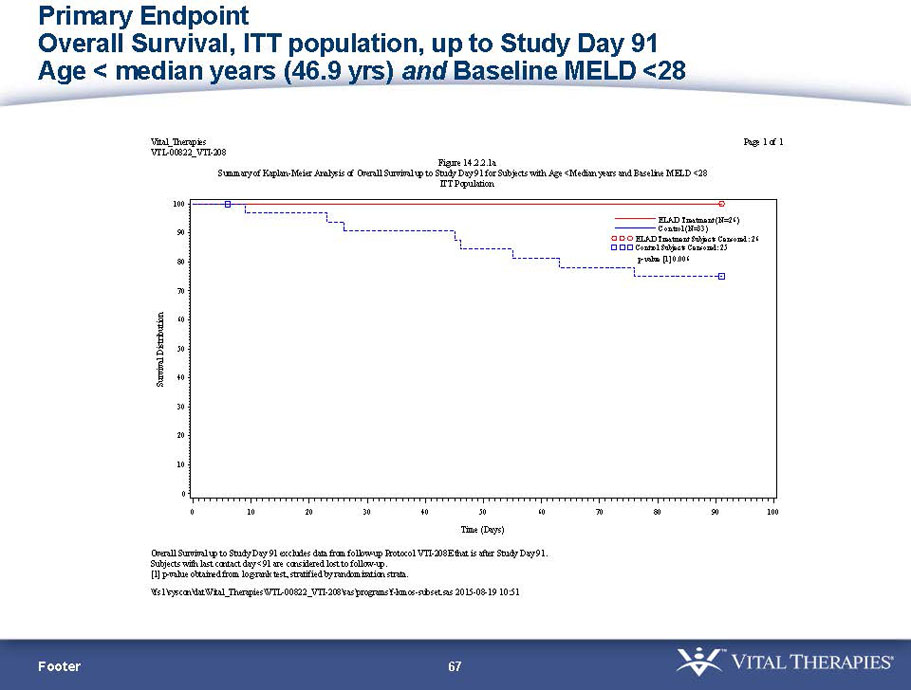
- A post-hoc analysis using log-rank statistics of the Kaplan Meier survival curves up to study day 91 (which was a pre-specified sensitivity analysis in the intention-to-treat, or ITT, and per protocol, or PP, populations), applied to the 59 subject (ELAD N = 26, Control N = 33) combination of these two pre-defined subsets (MELD less than 28 and age less than the median of 46.9 years), revealed no deaths in the ELAD group and eight deaths in the Control group. Had this combination been pre-defined, the p-value would have been <0.01.
Figure 1. Overall Survival, ITT population, up to Study Day 91 Age < median years (46.9 yrs) and Baseline MELD <28
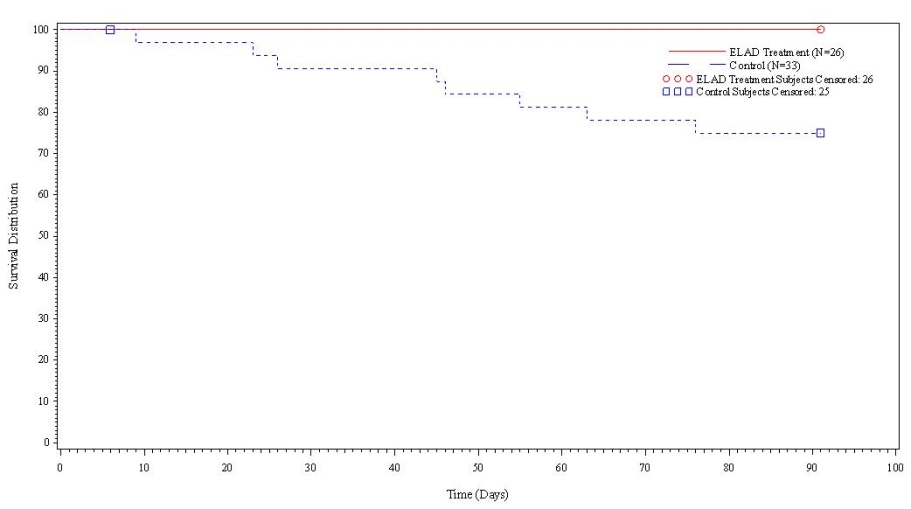
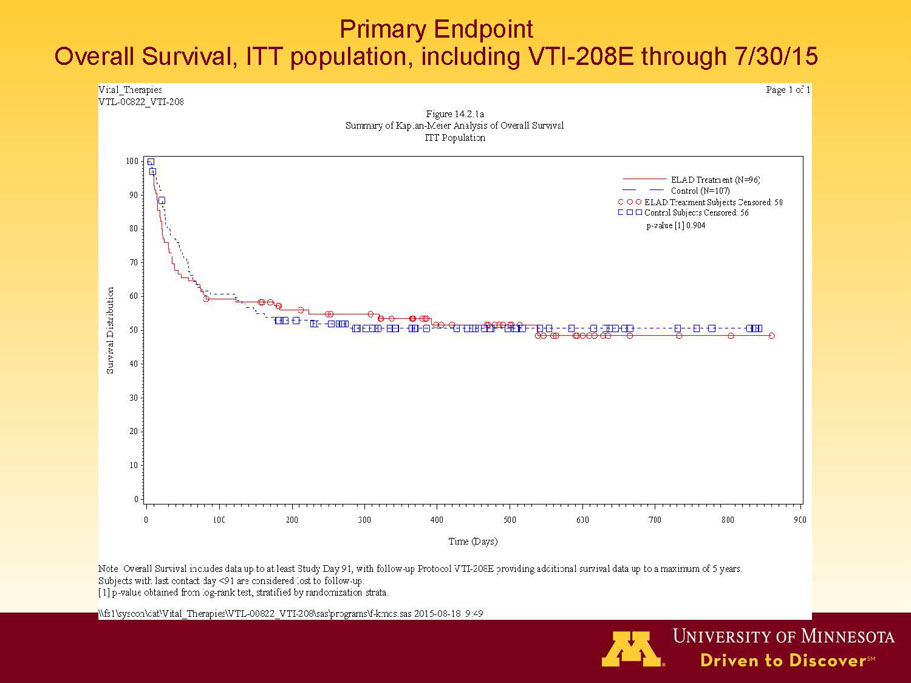
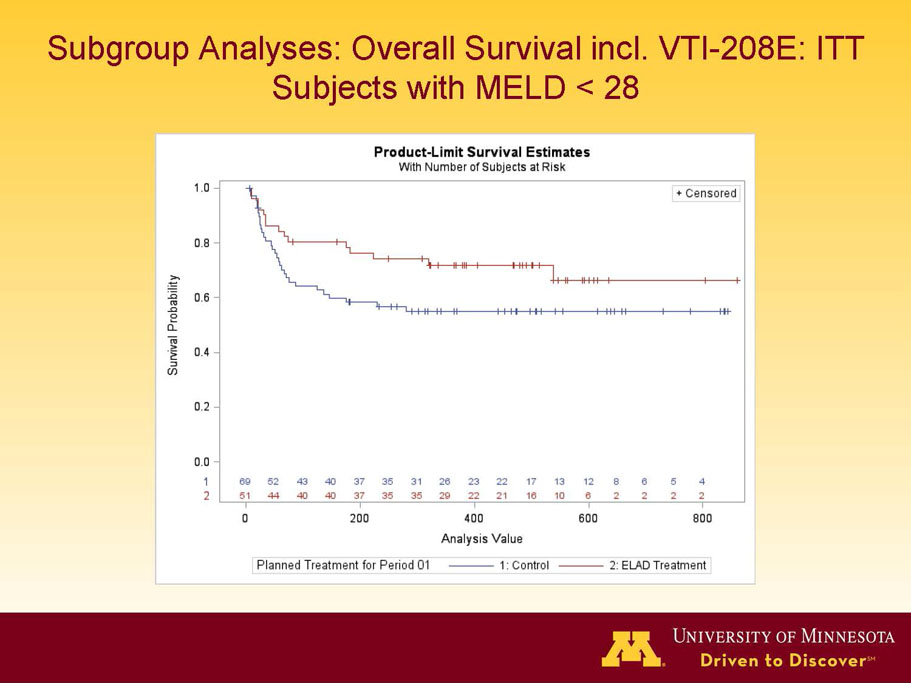
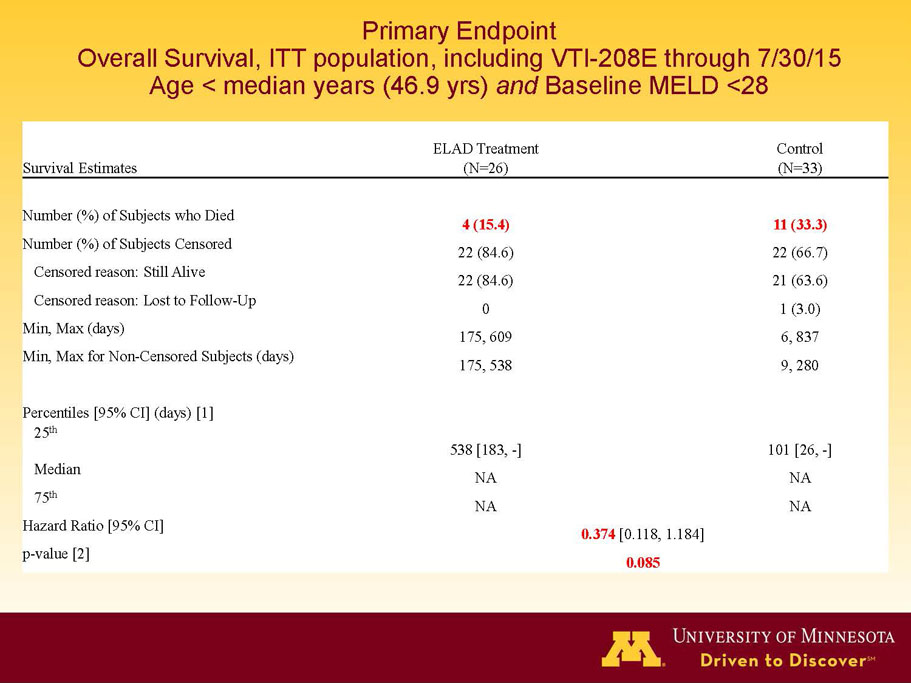
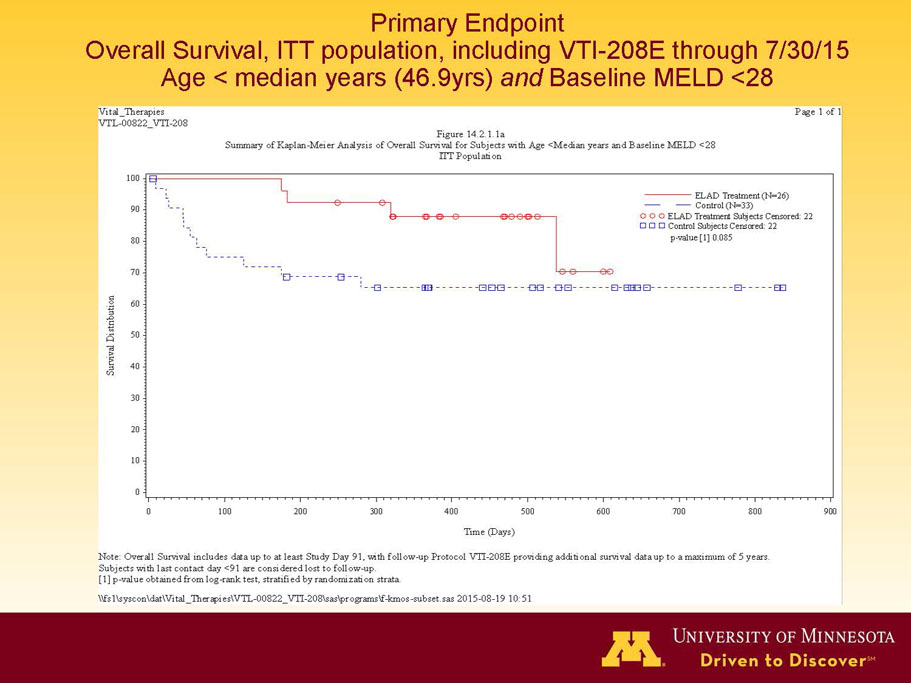
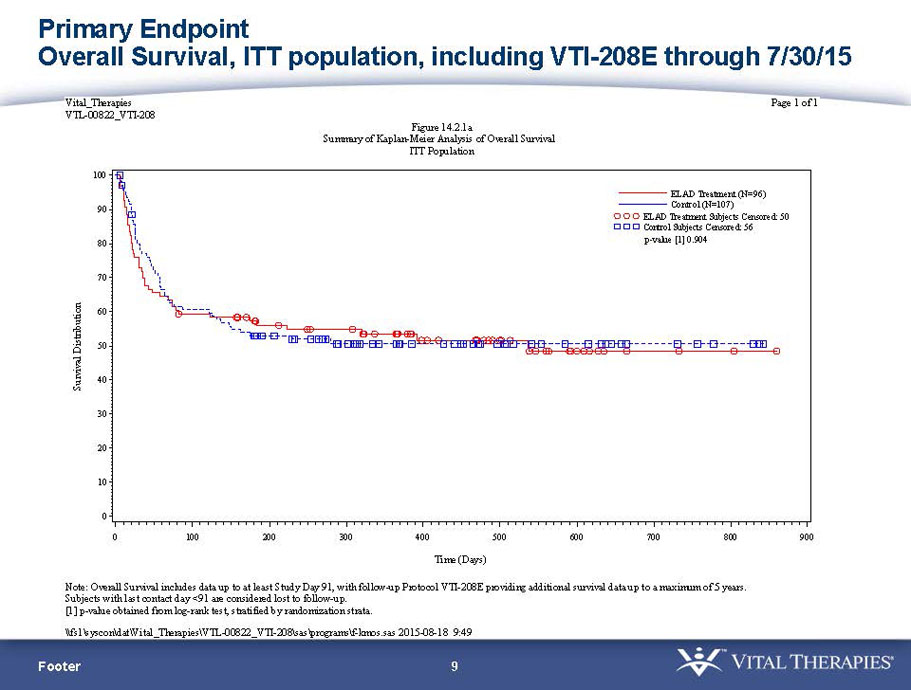
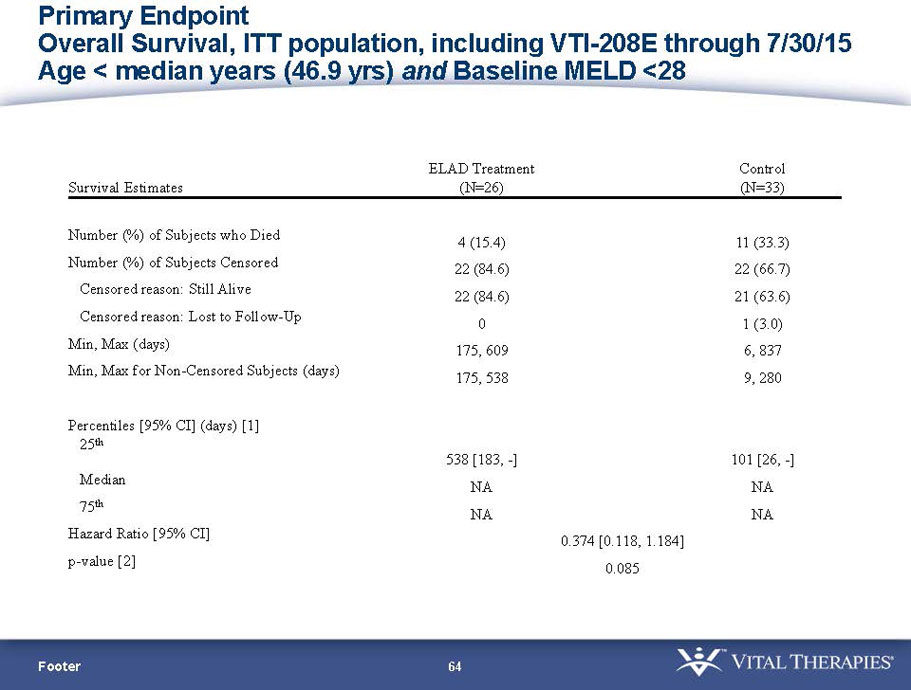
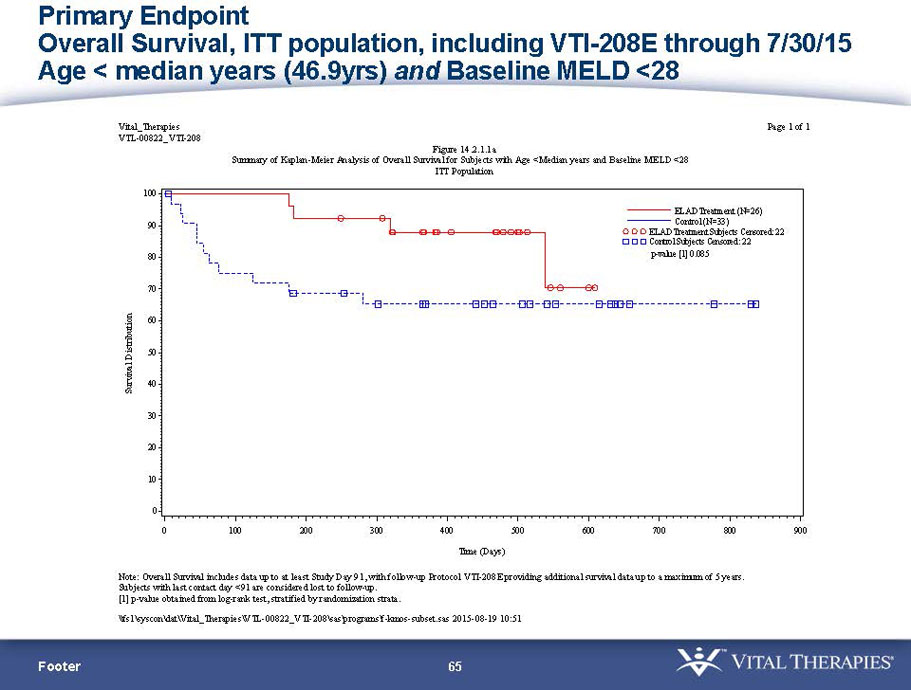
- A further post-hoc analysis of Kaplan Meier survival curves of this population which extends the analysis to incorporate data from VTI-208E, for which some subjects have now been followed for over 800 days, showed a hazard ratio of 0.375 in favor of ELAD, which, if pre-defined, would have shown a p-value of <0.10.
Figure 2. Overall Survival, ITT population, including VTI-208E through 7/30/15
Age < median years (46.9yrs) and Baseline MELD <28
- An analysis of co-morbidities typically found in subjects with liver failure, with recommendations on how to potentially improve ELAD’s benefit/risk profile.
The presentations are available at http://ir.vitaltherapies.com in the "Clinical Publications and Presentations" section.
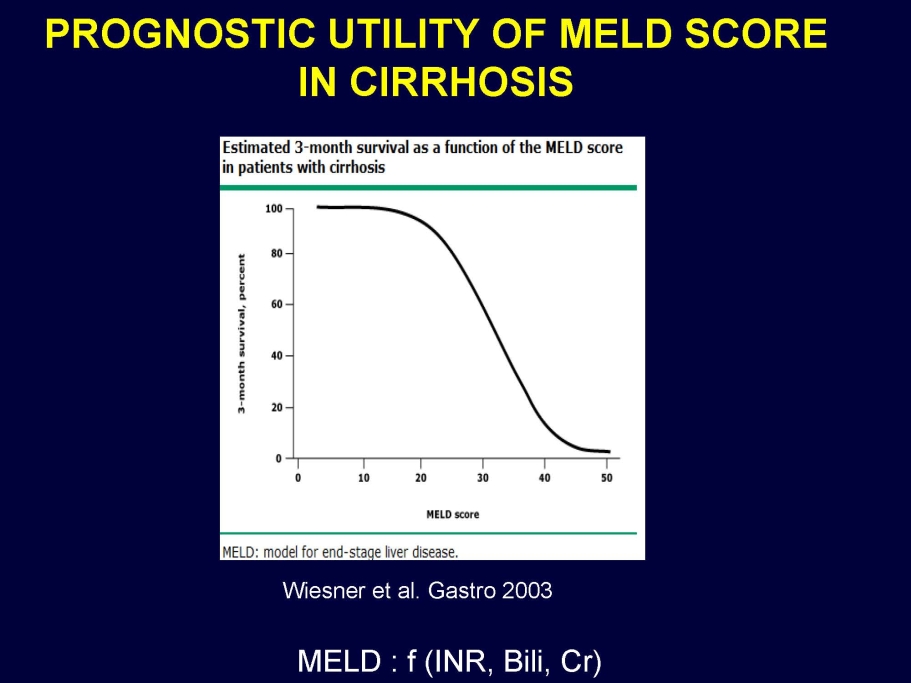
The Company continues analyzing the entire VTI-208 data set in order to determine the potential next steps in clinical development. In particular, the company is exploring the design of a new phase 3 clinical trial, which limits patients based on both MELD and age. The Company will provide periodic updates on material developments.
About Vital Therapies, Inc.
Vital Therapies, Inc. is a biotherapeutic company developing a cell-based therapy targeting the treatment of liver failure. The Company’s ELAD System, is an extracorporeal human allogeneic cellular liver therapy. Vital Therapies, Inc. is based in San Diego, California. Vital Therapies® and ELAD® are trademarks of Vital Therapies, Inc.
About VTI-208 and VTI-208E
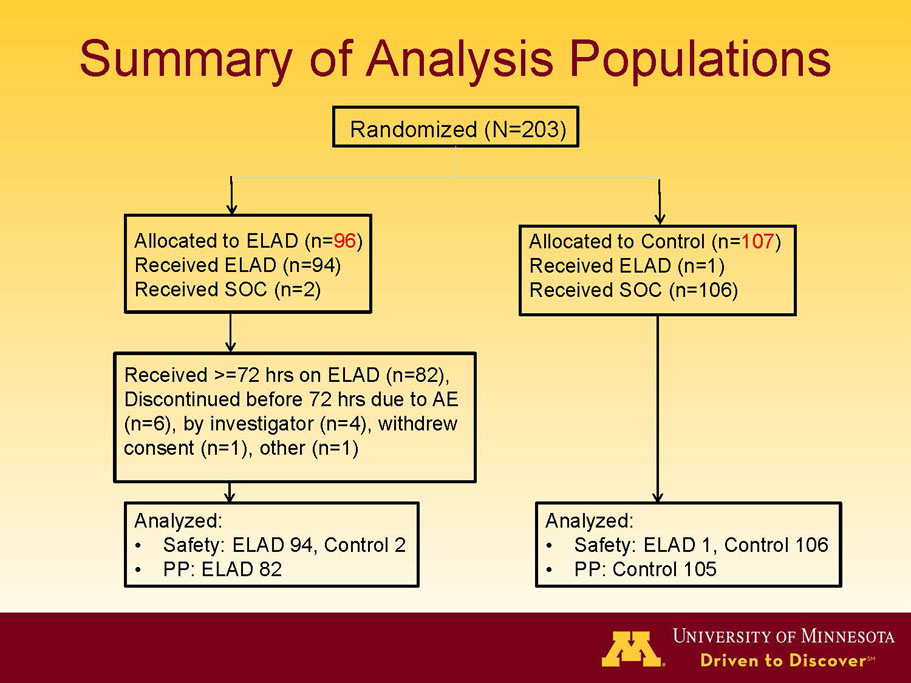
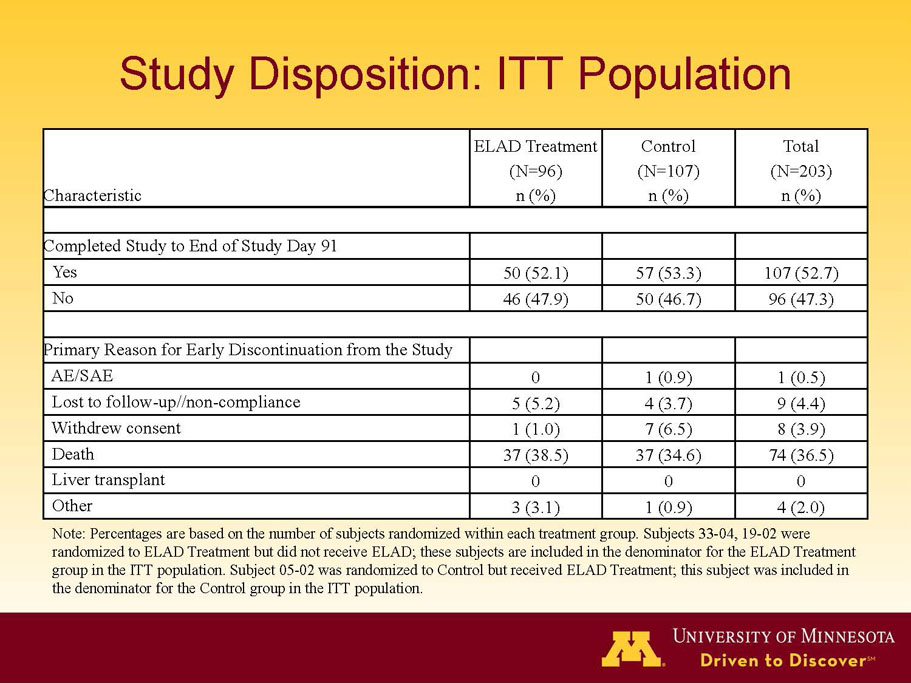
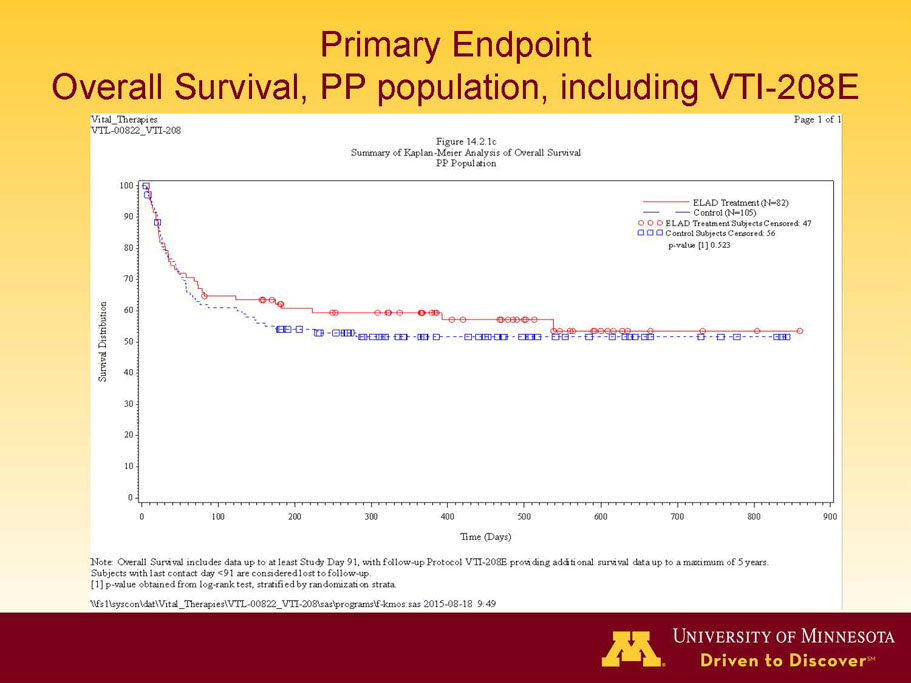
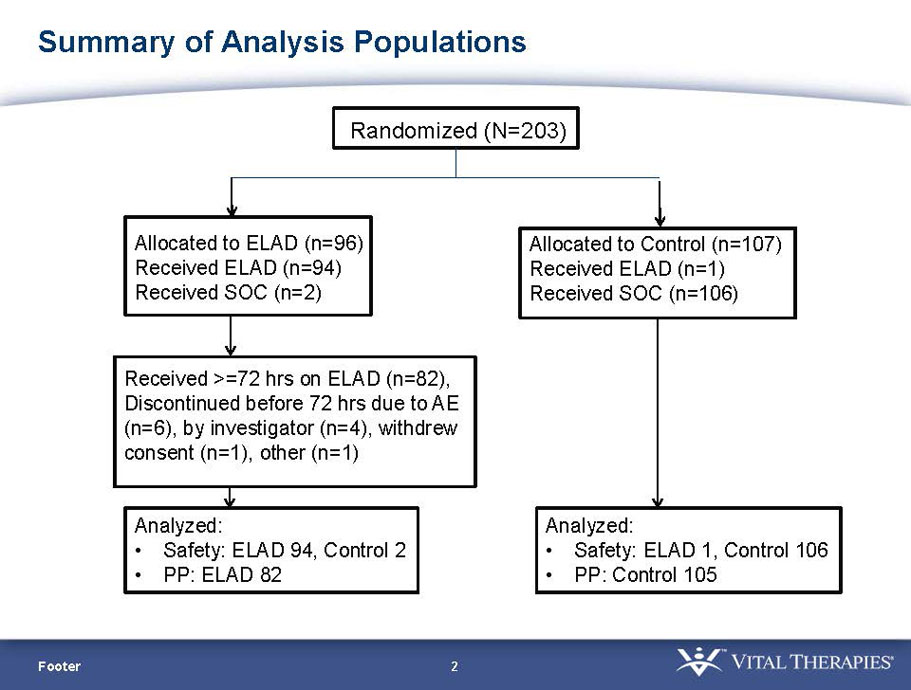
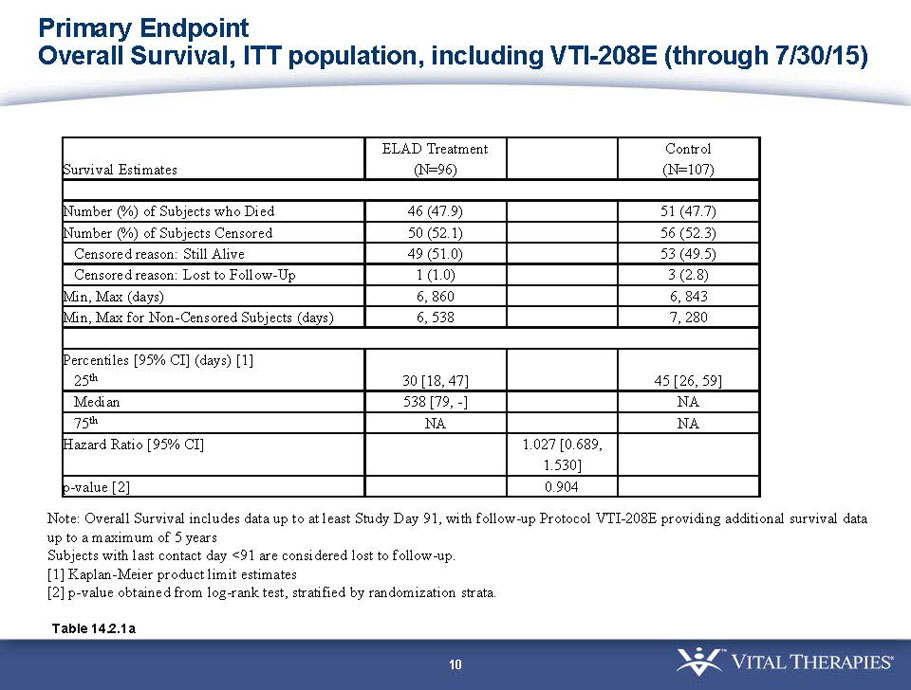
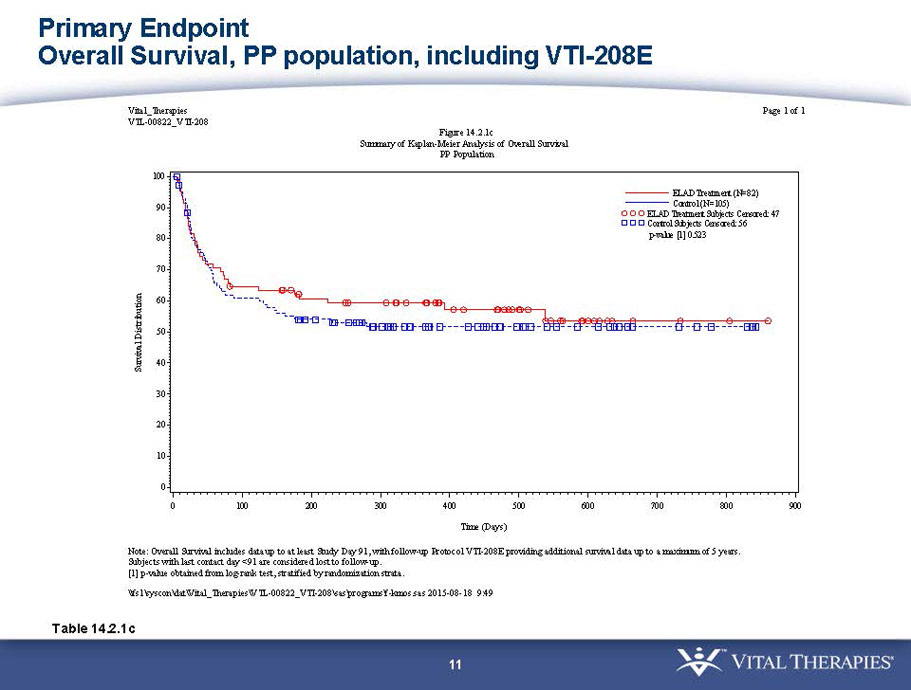
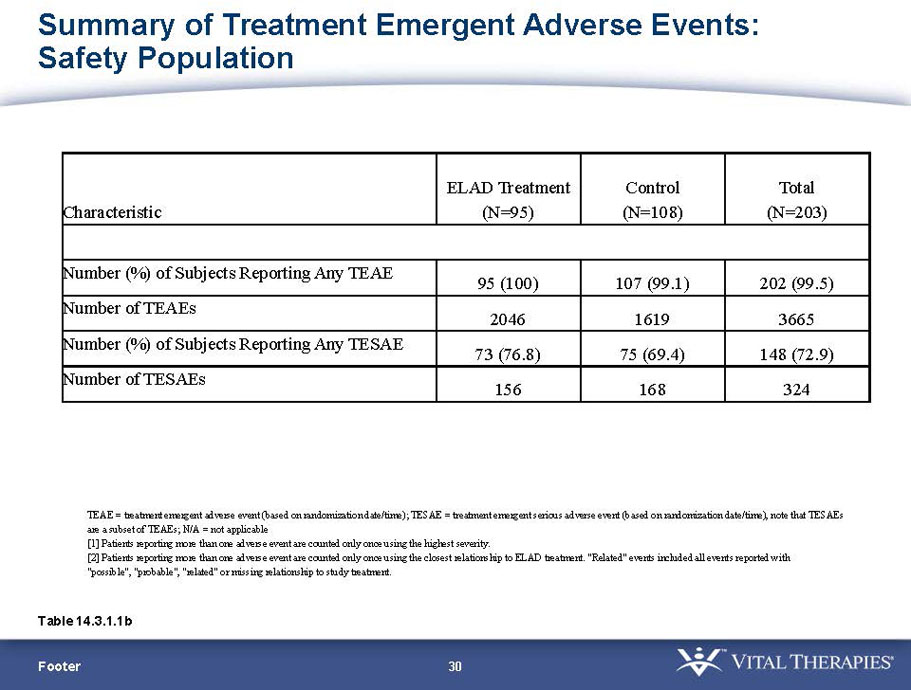
VTI-208 is a phase 3 randomized, controlled, open-label trial, evaluating the ELAD System in subjects with alcohol-induced liver decompensation (AILD). The primary endpoint is overall survival through at least 91 days assessed using the Kaplan Meier statistical method, and the secondary endpoint is proportion of survivors at study days 28 and 91. The trial enrolled 203 subjects over 22 months with 96 subjects randomized to the treated group and 107 randomized to the control group. VTI-208E is an extension study which assesses subject outcomes for 5 years after enrollment in VTI-208. Topline efficacy and safety data from the VTI-208 clinical trial, incorporating VTI-208E survival data through July 30, 2015, was disclosed on August 21, 2015: the trial failed to reach its primary or secondary endpoints.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include, among others, statements relating to our continuing analyses of the data from the VTI-208 clinical trial, including exploratory subset analyses, our plans to restructure our clinical development program, including a potential new trial and potential inclusion and exclusion criteria and study design, and related statements. Forward-looking statements are based on management's current expectations and are subject to various risks and uncertainties that could cause actual results to differ materially and adversely from those expressed or implied by such forward-looking statements. Accordingly, these forward-looking statements do not constitute guarantees of future performance and you are cautioned not to place undue reliance on these forward-looking statements. Risks and uncertainties include, but are not limited to, the risk that further analysis of the VTI-208 data does not support and/or contradicts our preliminary findings; the risk that the VTI-208 data will not support a proposed new clinical trial; the risk that the FDA does not approve a new clinical trial plan; delays in meeting with FDA or completing those meetings, the success or failure of a new clinical trial, if any; and the uncertainties inherent in Vital Therapies’ clinical and development programs, including, without limitation, Vital Therapies’ ability to adequately demonstrate the safety and efficacy of the ELAD System, future clinical results, which may not support further development of the ELAD System, and challenges related to conducting pivotal clinical trials, including, but not limited to, the impact of VTI-208, failure to achieve favorable results in clinical trials, the successful opening and the continued participation of clinical sites and their ongoing adherence to protocols, assumptions regarding enrollment rates, timing and availability of subjects meeting inclusion and exclusion criteria, changes to protocols or regulatory requirements, the ability to comply with and meet applicable laws and regulations, and unexpected adverse events or safety issues. There can be no assurance that data from any of our clinical trials will be sufficient to support an application for marketing in any country or that any such application will ever be approved. These and other risks regarding our business are described in detail in our Securities and Exchange Commission filings, including in our Quarterly Report on Form 10-Q for the quarter ended June 30, 2015. These forward-looking statements speak only as of the date hereof and Vital Therapies, Inc. disclaims any obligation to update these statements except as may be required by law.
Contact:
Vital Therapies, Inc.
Al Kildani
Vice President, Investor Relations and Business Development
858-673-6840
akildani@vitaltherapies.com