Exhibit 99.1
| Universal Biosensors Inc ARBN 121 559 993
1 Corporate Avenue Rowville VIC 3178 Australia
Telephone +61 3 9213 9000 Facsimile +61 3 9213 9099 Email info@universalbiosensors.com www.universalbiosensors.com |

|
22 June 2017
Chairman’s Address
Annual General Meeting
Good morning ladies & gentlemen and welcome to Universal Biosensor’s 2017 Annual General Meeting.
Since our meeting last year, I am pleased to report that Universal Biosensors has demonstrated strong momentum and achievement of many milestones. We are now firmly established as a technology leader in the global point-of-care diagnostics sector and, importantly for our shareholders, have over the past 12 months transitioned to be a profitable company that is also generating positive cash flows.
I would like to highlight what I see as two very important value drivers for our Company.
Firstly, we have now proven our ability to execute technically. We have demonstrated that we can turn innovation, technology and ideas into products that not only work, but make an important difference to people’s lives.
Secondly, we have a successful track record of collaboration with leading global healthcare companies to bring products to market. Our relationships with LifeScan (owned by Johnson & Johnson) and Siemens Healthineers, are testament to the success of our partnership commercialisation strategy.
These two drivers have been and remain fundamental for the success of our Company, and we have demonstrated our employees and management team have the capabilities to execute and deliver in these areas.
Now briefly turning to our financial performance, in February 2017 we released our full year financial results for the year ending 31 December 2016. We reported strong revenue growth and net profit and positive operating cash flow. More recently, in April we released our results for the first quarter of 2017, which showed the continuation of these trends and was again, a very strong financial result.
The performance of the One Touch Verio® strips has been key to driving our strong financial performance. Our revenue growth has been underpinned by increasing Quarterly Service Fees paid to us by LifeScan, which continue to grow as the OneTouch Verio® blood glucose meter captures market share. Importantly, we have also been successful in lowering the cost base of our business – achieving reductions in both R&D spend and general operating expenditure.
I will talk to our financial performance in more detail during my Business Update presentation shortly.
As expected, the revenue from the supply of test strips for the Siemens Xprecia Stride™ Coagulation Analyser didn’t make a significant revenue contribution in 2016, but sales in 2017 have started to ramp. There have been some important developments in this area over the past year. The first was FDA approval to sell the Xprecia Stride™ Coagulation Analyser in the US which was received by our partner Siemens Healthcare in October 2016. Following receipt of this FDA approval, in May 2017 Siemens commenced sales activities in the US market. Additionally, full commercial launch has now occurred in other markets such as Europe.
The combination of these developments is beginning to translate to improving results from the supply of Xprecia Stride™ test strips for UBI. In the first quarter of 2017 our revenue was $872,000, which was almost 5 times the total revenue we received from Xprecia Stride™ for the full 2016 financial year. We expect to see Xprecia Stride™ test strips make an increasingly important financial contribution to UBI’s financial position in 2017.
Looking to the year ahead, I expect that UBI will continue the positive momentum it has demonstrated in 2016. We anticipate continued growth in sales of One Touch Verio® blood glucose test strips and Siemens Xprecia Stride™ Coagulation Analyser strips.
From a development perspective, our strong track record of innovation and partnerships in the point-of-care diagnostics space also lays the foundation for successful new products. We have clinical trials for new products scheduled to occur in 2017 and with our strong cash balance and cash inflows, we are well placed to fund attractive new opportunities as they arise.
On behalf of the Board, I would like to thank all our dedicated employees for their contribution to the success of the Company over the past year. I would also like to thank my fellow Board members for their important ongoing work and commitment to the success of the Company.
I would now like to present a Business Update which provides further detail on UBI’s achievements over the past 12 months as well as potential news flow and opportunities for the coming year.
Mr Andrew Denver
Executive Chairman and Chief Executive Officer

Annual General Meeting 22 June 2017 Business Update
This presentation is intended to provide a general outline only and is not intended to be a definitive statement on the subject matter. This presentation is not financial advice and has been prepared without taking into account the objectives, financial situation or needs of a particular person. Neither the Company, nor its officers or advisors or any other person warrants the accuracy of the analysis herein or guarantees the investment performance of the Company. Investors must make their own independent assessment of the Company and undertake such additional enquiries as they deem necessary or appropriate for their own investment purposes. The statements contained in this presentation that are not purely historical are forward-looking statements within the meaning of the United States Exchange Act. Forward-looking statements in this presentation include statements regarding our expectations, beliefs, hopes, intentions or strategies. All forward-looking statements included in this presentation are based upon information available to us as of the date hereof, and we assume no obligation to update any such forward-looking statement as a result of new information, future events or otherwise. Our actual results could differ materially from our current expectations. The Company is subject to a number of risks. For a summary of key risks, refer to the Company’s most recent Form 10-K filed with the United States Securities and Exchange Commission and the Australian Securities Exchange. Under applicable United States securities laws all of the shares of our common stock are “restricted securities” as that term is defined in Rule 144 under the Securities Act of 1933, as amended. Restricted securities may be resold in the public market to United States persons as defined in Regulation S only if registered for resale or if they qualify for an exemption from registration under the Securities Act. We have not agreed to register any of our common stock for resale by security holders. 2 Important Disclaimer

UBI: Investment Highlights 3 Targeting the large and growing Point-of-Care Diagnostics market Fast growth in revenues based on the manufacture and sale of disposable test strips Unique patent-protected technology with low cost manufacturing Validating the technology through partnering deals with LifeScan (Johnson & Johnson) and Siemens Driving growth beyond the existing product platform via new product development

Transition to profitability and cash flow generation Disciplined R&D spend and operating cost reduction Strong revenue growth underpinned by Quarterly Service Fee’s as OneTouch® Verio continued to achieve market share gains Xprecia Stride™ US sales activities commenced by Siemens Strong growth momentum continues supported by focused development pipeline 4 Highlights : last 12 months
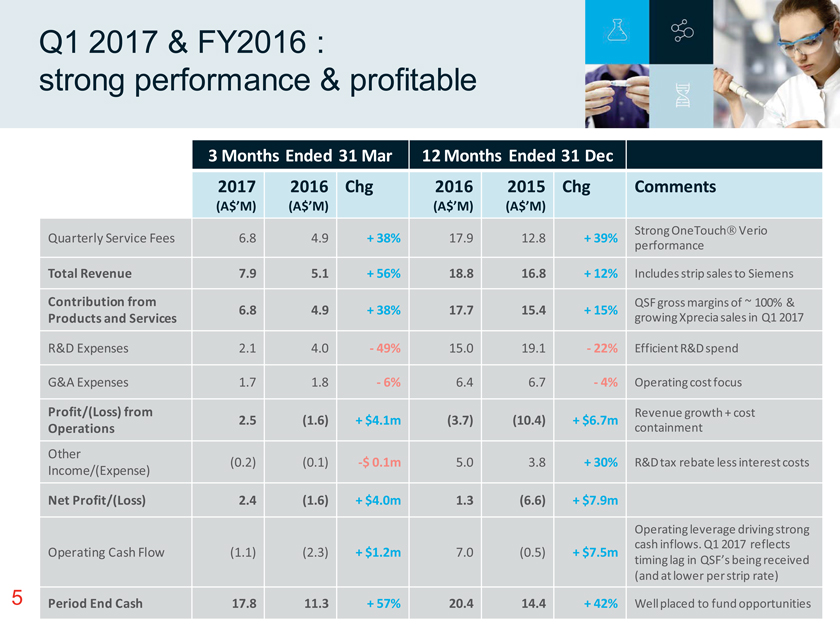
Q1 2017 & FY2016 : strong performance & profitable 3 Months Ended 31 Mar 12 Months Ended 31 Dec 2017 (A$’M) 2016 (A$’M) Chg 2016 (A$’M) 2015 (A$’M) Chg Comments Quarterly Service Fees 6.8 4.9 + 38% 17.9 12.8 + 39% Strong OneTouch Verio performance Total Revenue 7.9 5.1 + 56% 18.8 16.8 + 12% Includes strip sales to Siemens Contribution from Products and Services 6.8 4.9 + 38% 17.7 15.4 + 15% QSF gross margins of ~ 100% & growing Xprecia sales in Q1 2017 R&D Expenses 2.1 4.0—49% 15.0 19.1—22% Efficient R&D spend G&A Expenses 1.7 1.8—6% 6.4 6.7—4% Operating cost focus Profit/(Loss) from Operations 2.5 (1.6) + $4.1m (3.7) (10.4) + $6.7m Revenue growth + cost containment Other Income/(Expense) (0.2) (0.1) -$ 0.1m 5.0 3.8 + 30% R&D tax rebate less interest costs Net Profit/(Loss) 2.4 (1.6) + $4.0m 1.3 (6.6) + $7.9m Operating Cash Flow (1.1) (2.3) + $1.2m 7.0 (0.5) + $7.5m Operating leverage driving strong cash inflows. Q1 2017 reflects timing lag in QSF’s being received (and at lower per strip rate) Period End Cash 17.8 11.3 + 57% 20.4 14.4 + 42% Well placed to fund opportunities 5

Universal Biosensors
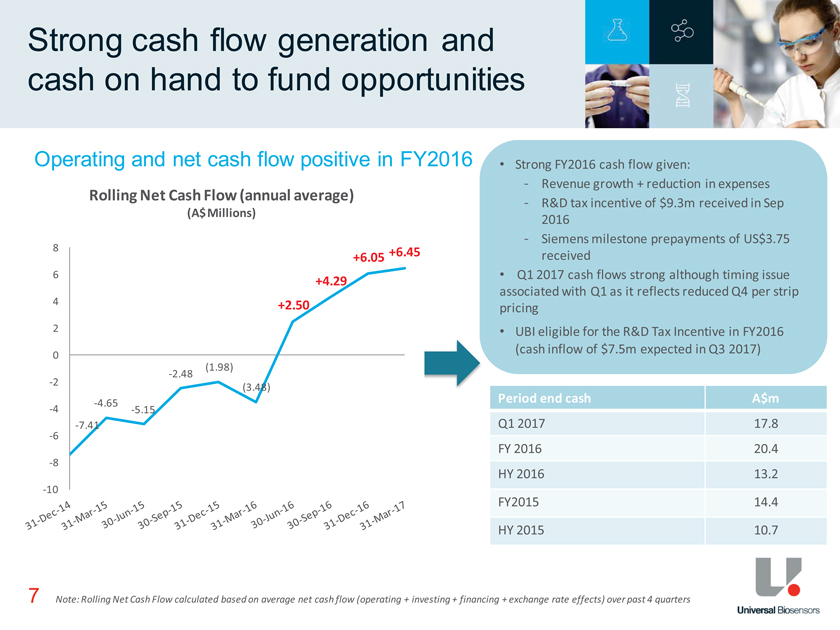
Strong cash flow generation and cash on hand to fund opportunities 7 Note: Rolling Net Cash Flow calculated based on average net cash flow (operating + investing + financing + exchange rate effects) over past 4 quarters • Strong FY2016 cash flow given:—Revenue growth + reduction in expenses—R&D tax incentive of $9.3m received in Sep 2016—Siemens milestone prepayments of US$3.75 received • Q1 2017 cash flows strong although timing issue associated with Q1 as it reflects reduced Q4 per strip pricing • UBI eligible for the R&D Tax Incentive in FY2016 (cash inflow of $7.5m expected in Q3 2017) Operating and net cash flow positive in FY2016 Period end cash A$m Q1 2017 17.8 FY 2016 20.4 HY 2016 13.2 FY2015 14.4 HY 2015 10.7 -7.41 -4.65 -5.15 -2.48 (1.98) (3.48) +2.50 +4.29 +6.05 +6.45 -10 -8 -6 -4 -2 0 2 4 6 8 Rolling Net Cash Flow (annual average) (A$ Millions)
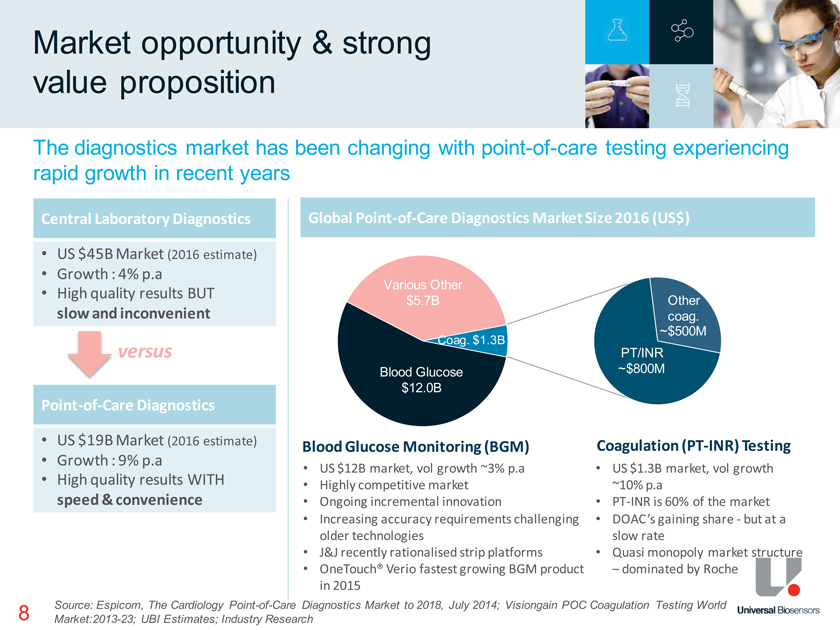
8 Market opportunity & strong value proposition The diagnostics market has been changing with point-of-care testing experiencing rapid growth in recent years Source: Espicom, The Cardiology Point-of-Care Diagnostics Market to 2018, July 2014; Visiongain POC Coagulation Testing World Market:2013-23; UBI Estimates; Industry Research Various Other $5.7B Blood Glucose $12.0B Coag. $1.3B PT/INR ~$800M Other coag. ~$500M Central Laboratory Diagnostics • US $45B Market (2016 estimate) • Growth : 4% p.a • High quality results BUT slow and inconvenient Point-of-Care Diagnostics • US $19B Market (2016 estimate) • Growth : 9% p.a • High quality results WITH speed & convenience versus Global Point-of-Care Diagnostics Market Size 2016 (US$) • US $12B market, vol growth ~3% p.a • Highly competitive market • Ongoing incremental innovation • Increasing accuracy requirements challenging older technologies • J&J recently rationalised strip platforms • OneTouch® Verio fastest growing BGM product in 2015 Blood Glucose Monitoring (BGM) Coagulation (PT-INR) Testing • US $1.3B market, vol growth ~10% p.a • PT-INR is 60% of the market • DOAC’s gaining share—but at a slow rate • Quasi monopoly market structure – dominated by Roche

Universal Biosensors
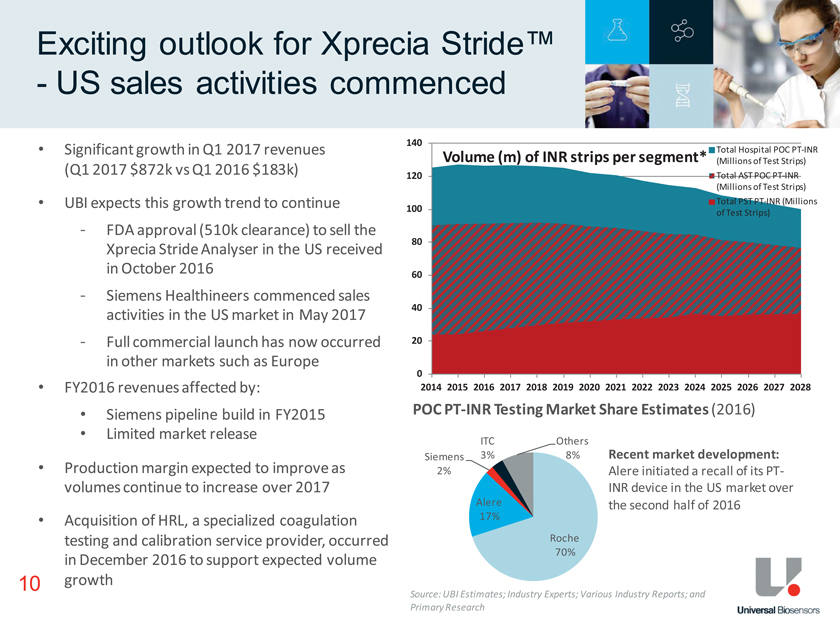
Exciting outlook for Xprecia Stride™—US sales activities commenced Source: UBI Estimates; Industry Experts; Various Industry Reports; and Primary Research POC PT-INR Testing Market Share Estimates (2016) ITC Roche 10 • Significant growth in Q1 2017 revenues (Q1 2017 $872k vs Q1 2016 $183k) • UBI expects this growth trend to continue—FDA approval (510k clearance) to sell the Xprecia Stride Analyser in the US received in October 2016—Siemens Healthineers commenced sales activities in the US market in May 2017—Full commercial launch has now occurred in other markets such as Europe • FY2016 revenues affected by: • Siemens pipeline build in FY2015 • Limited market release • Production margin expected to improve as volumes continue to increase over 2017 • Acquisition of HRL, a specialized coagulation testing and calibration service provider, occurred in December 2016 to support expected volume growth Recent market development: Alere initiated a recall of its PT-INR device in the US market over the second half of 2016 Roche 70% Alere 17% Siemens 2% ITC 3% Others 8% 0 20 40 60 80 100 120 140 2014 2015 2016 2017 2018 2019 2020 2021 2022 2023 2024 2025 2026 2027 2028 Volume (m) of INR strips per segment* Total Hospital POC PT-INR (Millions of Test Strips) Total AST POC PT-INR (Millions of Test Strips) Total PST PT-INR (Millions of Test Strips)

Focussed development pipeline to drive growth • Rigorous product development criteria: – Deliver ROI for shareholders – Ability to sustain positive cash flow • Exciting new products are in the development pipeline with Siemens • Clinical trials targeted to commence in 2017. Regulatory approval will be sought after completion of these trials • Proven track record of collaboration with two of the world’s leading healthcare companies to bring products to market 11 Strong track record of innovation & partnerships in the Point-of-Care diagnostics space Feasibility Development In Market Coagulation PT-INR Test (Siemens) Coagulation Test 3 (Siemens) Molecular Blood Glucose Test (LifeScan) Concept Coagulation Test 2 (Siemens)

Anticipated news flow: next 12 months 12 • Quarterly Service Fees from OneTouch® Verio announced every quarter • Growth in Xprecia Stride strip sales • Announcement of commencement of clinical trials in potential new product areas • Feasibility completed for internal Molecular platform Upcoming market communications: • H1 2017 results : July 2017 • Appendix 4C Q2 2017 : July 2017