Table of Contents
SECURITIES AND EXCHANGE COMMISSION
| o | REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| þ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE
SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December 31, 2010 |
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| o | SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
(Jurisdiction of incorporation or organization)
(Address of principal executive offices)
Company Secretary
GlaxoSmithKline plc
980 Great West Road
England
+44 20 8047 5000
(Name, Telephone, E-mail and/or Facsimile number and Address of Company Contact Person)
| Title of Each Class | Name of Each Exchange On Which Registered | |
American Depositary Shares, each representing
2 Ordinary Shares, Par value 25 pence
|
New York Stock Exchange | |
4.850% Notes due 2013
|
New York Stock Exchange | |
|
5.650% Notes due 2018
|
New York Stock Exchange | |
6.375% Notes due 2038
|
New York Stock Exchange |
(Title of class)
(Title of class)
Ordinary Shares of Par value 25 pence each |
5,196,264,019 | |||
Large accelerated filer þ
|
Accelerated filer o | Non-accelerated filer o |
| U.S. GAAP o | International Financial Reporting Standards as issued by the International Accounting Standards Board | þ | Other o |
Table of Contents

Table of Contents
01
quality of human life by
enabling people to do more,
feel better and live longer.
| We work by respecting people, maintaining our focus on the patient and consumer whilst operating with both integrity and transparency. |
| We are looking to deliver shareholder value through growth of a diversified and global business, by delivering more products of value, simplifying our operating model and by running our business responsibly. |
| What follows is our report to shareholders for 2010. |
Forward-looking statements involve inherent risks and uncertainties. The Group cautions investors that a number of important factors, including those in this document, could cause actual results to differ materially from those contained in any forward-looking statement. Such factors include, but are not limited to, those discussed under ‘Risk factors’ on pages 53 to 57 of this Annual Report.
Table of Contents
02
research-based pharmaceutical and
healthcare companies. We are
committed to improving the quality
of human life by enabling people
to do more, feel better and live longer.
| • | Grow a diversified global business | |
| • | Deliver more products of value | |
| • | Simplify GSK’s operating model |
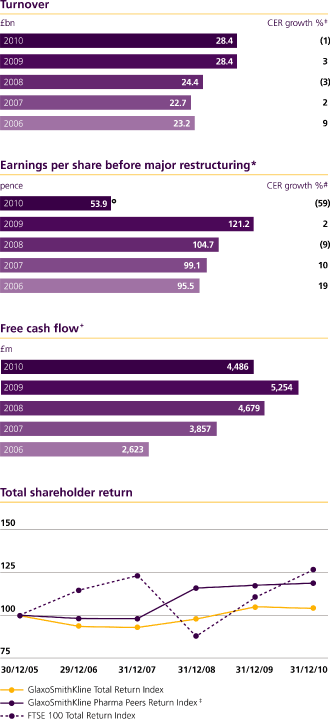
£28.4bn
|
32.1p | |
Turnover
|
Earnings per share | |
53.9p
|
65p | |
Earnings per share before major restructuring |
Dividend per share |
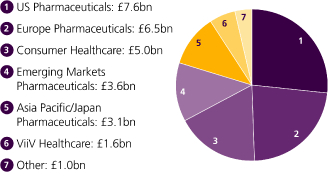
| Research & development | Consumer Healthcare | |||||
c.30 |
£3.96bn | 20% | No.1 | |||
A peer-leading pipeline |
In 2010, we spent £3.96bn | Growth of Horlicks | Sensodyne has been the | |||
with around 30 late-stage |
in R&D before major | in India in 2010. | world’s fastest growing | |||
assets. |
restructuring, or 14% | toothpaste brand over | ||||
| of our total sales. | the last 5 years. | |||||
| We are one of the world’s | ||||||
| biggest investors in R&D and are | ||||||
| the biggest private sector funder | ||||||
| of R&D in the UK. | ||||||
10 |
14% | c.1bn | 2 | |||
10 new compounds and |
We are committed to | Units of Lucozade, Ribena | New Consumer Healthcare | |||
vaccines starting phase III |
improving returns in R&D, | and Horlicks manufactured | Research and Innovation | |||
clinical trials since the start |
aiming to increase our | in the UK every year. | centres opened in China | |||
of 2010. |
estimated return | and India. | ||||
| on investment in | ||||||
| this area to 14%. | ||||||
| Vaccines | Emerging markets | |||||
1.4bn |
24% | |||||
Doses of our vaccines |
Of total GSK turnover from | |||||
supplied to 179 countries |
emerging markets, by the broader | |||||
around the world in 2010. |
definition (Pharmaceutical and | |||||
| Consumer Healthcare turnover | ||||||
| in all markets excluding USA, | ||||||
| Western Europe, Canada, Japan, | ||||||
| Australia and New Zealand). | ||||||
Table of Contents
03
96,500 |
5% | 3 | ||
Employees. |
Share of world | Leading presence in | ||
| pharmaceutical market. | Consumer Healthcare | |||
| (Source: IMS Health) | global categories: OTC, | |||
| Oral Care, Nutritionals. |
| |
Table of Contents
04

Table of Contents
05

|
 |
|
Sir Christopher Gent
|
Andrew Witty | |
Chairman
|
Chief Executive Officer |
| † | See page 21. |
Table of Contents
 |
| |
GSK Annual Report 2010
Table of Contents
07
| * | See page 21. |
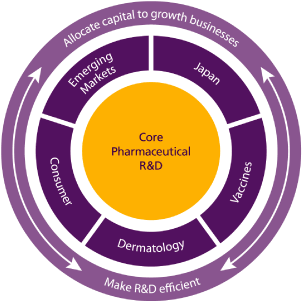
| • | Drive growth in the pharmaceutical business in our core markets | |
| • | Fulfil the potential of Emerging Markets | |
| • | Expand our business in Japan | |
| • | Build our leadership in dermatology | |
| • | Grow the Vaccines and Consumer Healthcare businesses |
| • | Focus on the best science | |
| • | Diversify through externalisation | |
| • | Re-personalise R&D | |
| • | Focus on return on investment |
| • | Evolve our commercial model | |
| • | Re-shape manufacturing | |
| • | Streamline our processes | |
| • | Reduce working capital |
Table of Contents
08
Our strategies
|
Our measures | Our progress in 2010 | ||
We have focused the business around the
delivery of three strategic priorities.
|
We use a number of measures to
track our progress against the strategic priorities
over the medium to long term. These include the
following: |
We made good progress during the year, with a number of notable successes: | ||
Grow a diversified global business Broadening and balancing our portfolio and moving away from a reliance on ‘white pills/western markets’.† |
• Performance of core Pharmaceuticals and vaccines businesses
|
• Excluding pandemic products, Avandia and Valtrex, underlying pharmaceutical (including vaccines) sales* were £21.1 billion and grew 4% in the year. |
||
• Diversification of sales
|
• Sales from ‘white pills/western markets’† fell from 40% of turnover in 2007 to 25% in 2010 |
|||
• Contribution of Emerging Markets to our overall sales and growth
|
• Sales in our Emerging Markets pharmaceutical business grew by 22% to more than £3.6 billion and now represent 15% of pharmaceutical turnover. |
|||
• Growth of Consumer Healthcare business
|
• Sales in our Consumer Healthcare business grew by 5% to £5.0 billion and now represent 17.6% of Group turnover. |
|||
• Build our leadership position in dermatology
|
• Dermatology sales grew on a pro-forma basis (excluding 2010 acquisitions) by approximately 6% to nearly £1.1 billion, representing nearly 4% of Group turnover. |
|||
• Expansion of Japanese business
|
• Sales in GSK Japan grew 14% to nearly £2.0 billion. |
|||
• We received approvals for four new compounds. |
||||
• Build biopharmaceutical portfolio
|
• Arzerra recorded sales of £26 million on its first full year on the US market and was launched in Europe. Benlysta filed for approval in both the USA and Europe. |
|||
Deliver more products of value Transforming R&D to ensure we not only deliver the current pipeline but are also able to sustain the flow of products for years to come. |
• Contribution to sales of new products
|
• New products launched since 2007 (excluding flu pandemic vaccines) grew 36% and contributed 7% of pharmaceutical sales in 2010. |
||
• Number of reimbursable product approvals and filings
|
• We received six product approvals in the USA and EU since the start of 2010 |
|||
• Seven assets are currently filed with regulators. |
||||
• Sustaining late-stage pipeline
|
• We maintained around 30 assets in phase III and registration, with ten new chemical entities and new vaccines entering phase III since the start of 2010. |
|||
• Enhanced R&D productivity and increased externalisation for Drug Discovery
|
• Our objective is to increase our estimated rate of return for R&D from around 11% to 14%. |
|||
• During 2010 we signed eight new collaborations to increase the external nature of our discovery, giving 54 external discovery engines to complement our 38 Discovery
Performance Units. |
||||
Simplifying the operating model Simplifying our operating model to ensure that it is fit for purpose and able to support our business in the most cost efficient way. |
• Delivery of major restructuring programme
|
• We have achieved annual cost savings of £1.7 billion and remain on track to reach £2.2 billion of annualised savings by 2012. |
||
• Reduce working capital. |
• Working capital reduced by £1.3 billion in 2010 (including £600 million of cash from lower pandemic receivables). |
| * | The calculation of underlying sales growth is described on page 21. |
| † | See page 21. |
Table of Contents
09
| ‡ | This index includes Abbott Labs, Amgen, AstraZeneca, Bristol Myers Squibb, Eli Lilly, Johnson & Johnson, Merck, Novartis, Pfizer, Roche Holdings and Sanofi-Aventis. | |
| o | Reflects £4bn legal charge. | |
| # | The calculation of CER growth is described on page 21. | |
| * | The calculation of results before major restructuring is described in Note 1 to the financial statements, ‘Presentation of the financial statements’. | |
| + | The calculation of free cash flow is described on page 44. |
Table of Contents
10
Table of Contents
11
Table of Contents
12
10
|
6 | 5 | ||
assets moved into
|
approvals in USA | assets terminated | ||
Phase III
|
or EU | from Phase III development | ||
IPX066†, for Parkinson’s
disease 1120212†, a MEK inhibitor, for metastatic melanoma 2118436, a BRaf inhibitor, for metastatic melanoma 573719 + vilanterol†, a combination drug for COPD 1605786†, for Crohn’s disease Zoster vaccine, for the prevention of shingles 2402968†, for Duchenne muscular dystrophy migalastat HCI†, for Fabry disease 1349572†, an integrase inhibitor, for HIV and as a fixed dose combination with Epzicom/Kivexa 2696273†, for adenosine deaminase severe combined immune deficiency |
Tyverb/Tykerb, for first line therapy for hormone receptor positive breast cancer (USA/EU) Arzerra, for refractory chronic lymphocytic leukaemia (EU) Revolade/Promacta, for idiopathic thrombocytopaenic purpura (EU) Duodart/Jalyn, a fixed dose combination drug for benign prostatic hyperplasia (USA/EU) Votrient, for renal cell cancer (EU) Prolia, for post- menopausal osteoporosis (EU) |
Avandamet XR, for type 2 diabetes Avandia + statin, for type 2 diabetes almorexant, for primary insomnia New generation flu, for influenza prophylaxis Simplirix, for genital herpes prophylaxis. |
Table of Contents
13
| Therapeutic area | Compound | Indication | Phase III | Filed | Approved | |||||
Biopharmaceuticals
|
albiglutide† | type 2 diabetes | l | |||||||
| Arzerra† | chronic lymphocytic leukaemia, first line therapy and use in relapsed patients | l | ||||||||
| Arzerra† | diffuse large B cell lymphoma (relapsed patients) | l | ||||||||
| Arzerra† | follicular lymphoma (refractory & relapsed patients) | l | ||||||||
| otelixizumab† | type 1 diabetes | l | ||||||||
| Benlysta† | systemic lupus erythematosus | l | ||||||||
| denosumab† | bone metastatic disease | l | ||||||||
| Arzerra† | chronic lymphocytic leukaemia (refractory patients) | l | ||||||||
| Prolia† | hormone ablative/chemotherapy bone loss in prostate cancer patients | l | ||||||||
| Prolia† | postmenopausal osteoporosis | l | ||||||||
Cardiovascular
& metabolic
|
darapladib† | atherosclerosis | l | |||||||
Infectious
diseases
|
Relenza† | treatment of influenza | l | |||||||
Neurosciences
|
IPX066† | Parkinson’s disease | l | |||||||
| Horizant† | restless legs syndrome | l | ||||||||
| Trobalt/Potiga (retigabine/ezogabine)† | epilepsy – partial seizures | l | ||||||||
Oncology
|
1120212† | metastatic melanoma | l | |||||||
| 2118436 | metastatic melanoma | l | ||||||||
| Votrient | ovarian cancer, maintenance therapy | l | ||||||||
| Revolade/Promacta† | chronic liver disease induced thrombocytopaenia | l | ||||||||
| Revolade/Promacta† | hepatitis C induced thrombocytopaenia | l | ||||||||
| Tyverb/Tykerb | breast cancer, adjuvant therapy | l | ||||||||
| Tyverb/Tykerb | gastric cancer | l | ||||||||
| Tyverb/Tykerb | head & neck squamous cell carcinoma (resectable disease) | l | ||||||||
| Votrient | renal cell cancer, adjuvant therapy | l | ||||||||
| Votrient | sarcoma | l | ||||||||
| Votrient + Tyverb/Tykerb | inflammatory breast cancer | l | ||||||||
| Avodart | reduction in the risk of prostate cancer | l | ||||||||
| Duodart/Jalyn | benign prostatic hyperplasia -fixed dose combination | l | ||||||||
| Revolade/Promacta† | idiopathic thrombocytopaenic purpura | l | ||||||||
| Tyverb/Tykerb | breast cancer, first line therapy | l | ||||||||
| Votrient | renal cell cancer | l | ||||||||
Respiratory
&
|
573719 | COPD | l | |||||||
immuno-inflammation
|
573719 + vilanterol† | COPD | l | |||||||
| vilanterol (642444)† | COPD | l | ||||||||
| 1605786 (CCX282)† | Crohn’s disease | l | ||||||||
| Relovair (vilanterol† + 685698) |
asthma | l | ||||||||
| Relovair (vilanterol + 685698) |
COPD | l | ||||||||
Paediatric vaccines
|
Mosquirix | malaria prophylaxis (plasmodium falciparum) | l | |||||||
| Nimenrix (MenACWY-TT) | neisseria meningitis groups A, C, W & Y disease prophylaxis | l | ||||||||
| MenHibrix (Hib-MenCY-TT) | neisseria meningitis groups C & Y & haemophilus influenzae type b disease prophylaxis | l | ||||||||
Other vaccines
|
Flu vaccine | seasonal influenza prophylaxis | l | |||||||
| Zoster | herpes zoster prevention | l | ||||||||
| Flu (pre-) pandemic | pre-pandemic & pandemic influenza prophylaxis | l | ||||||||
| Pumarix | pandemic influenza prophylaxis | l | ||||||||
Antigen Specific Cancer
|
MAGE-A3 | treatment of melanoma | l | |||||||
Immunotherapeutic (ASCI)
|
MAGE-A3 | treatment of non-small cell lung cancer | l | |||||||
Rare diseases
|
2402968† | Duchenne muscular dystrophy | l | |||||||
| 2696273† | adenosine deaminase severe combined immune deficiency |
l | ||||||||
| migalastat HCI† | Fabry disease | l | ||||||||
Dermatology
|
tazarotene foam | acne vulgaris | l | |||||||
| Duac low dose | acne vulgaris | l | ||||||||
| calcipotriene | mild to moderate plaque psoriasis | l | ||||||||
| itraconazole tablets | onychomycosis | l | ||||||||
| Veltin | acne vulgaris | l | ||||||||
HIV
|
1349572† | HIV infections | l | |||||||
| 1349572† + abacavir sulphate + lamivudine |
HIV infections | l | ||||||||
Table of Contents
14
| • | Abbott Laboratories | |
| • | Amgen | |
| • | AstraZeneca | |
| • | Bristol-Myers Squibb | |
| • | Eli Lilly | |
| • | Johnson & Johnson | |
| • | Merck | |
| • | Novartis | |
| • | Pfizer | |
| • | Roche Holdings | |
| • | Sanofi-Aventis |
Table of Contents
15
| USA | EU | |||||||||
| Products | Compounds | Indication(s) | Major | Patent expiry dates | ||||||
| competitor brands | USA | EU | ||||||||
| Respiratory | ||||||||||
Veramyst
|
fluticasone furoate | rhinitis | Nasacort | 2021 | 2023 | |||||
Flixotide/Flovent
|
fluticasone propionate | asthma/COPD | Qvar, Singulair | expired (compound) 2011-2016 (Diskus device) 2013-2025 (HFA-device/ formulation) |
expired (compound) expired (Diskus device) 2012-2017 (HFA-device/ formulation) |
|||||
Seretide/Advair*
|
salmeterol xinafoat/ fluticasone propionate | asthma/COPD | Singulair, Symbicort, Spiriva, Asmanex, Pulmicort, Foster |
expired (combination) 2011-2016 (Diskus device) 2013-2025 (HFA-device/ formulation) |
20131 (combination) expired (Diskus device) 2012-2017 (HFA-device/ formulation) |
|||||
Serevent
|
salmeterol xinafoate | asthma/COPD | Foradil, Spiriva | expired (compound) 2011-2016 (Diskus device) NA |
expired (compound) expired (Diskus device) 2012-2019 (HFA-device/ formulation) |
|||||
| Anti-virals | ||||||||||
Relenza
|
zanamivir | influenza | Tamiflu | 2013 | 2014 | |||||
Valtrex
|
valaciclovir | genital herpes, coldsores, shingles | Famvir | expired | expired | |||||
Zeffix/Epivir-HBV
|
lamivudine | chronic hepatitis B | Hepsera | 2013 (use) |
2012 (use) |
|||||
| Central nervous system | ||||||||||
Lamictal
|
lamotrigine | epilepsy, bipolar disorder | Keppra, Dilantin | expired | expired | |||||
Imigran/lmitrex
|
sumatriptan | migraine | Zomig, Maxalt, Relpax | expired | expired | |||||
Requip
|
ropinirole | Parkinson’s disease, restless legs syndrome | Mirapex | expired | expired | |||||
Requip XL
|
ropinirole | Parkinson’s disease | Mirapex | 2012† (formulation) |
2011 (use) |
|||||
Seroxat/Paxil
|
paroxetine | depression, various anxiety disorders |
Effexor, Cymbalta, Lexapro |
expired | expired | |||||
Treximet
|
sumatriptan and naproxen | migraine | Zomig, Maxalt, Relpax | 20171 (combination and use) |
NA | |||||
Wellbutrin SR
|
bupropion | depression | Effexor, Cymbalta, Lexapro | expired | expired | |||||
| Cardiovascular and urogenital | ||||||||||
Arixtra
|
fondaparinux | deep vein thrombosis, pulmonary embolism | Lovenox, Fragmin Innohep |
expired | expired | |||||
Avodart
|
dutasteride | benign prostatic hyperplasia | Proscar, Flomax, finasteride | 20151 | 2017 | |||||
Coreg CR
|
carvedilol phosphate | mild-to-severe heart failure, hypertension, left ventricular dysfunction post MI |
Toprol XL | 2016† (formulation) |
NA | |||||
Fraxiparine
|
nadroparin | deep vein thrombosis, pulmonary embolism |
Lovenox, Fragmin Innohep |
expired | expired | |||||
Lovaza
|
omega-3 acid ethyl esters | very high triglycerides | Tricor | 20171 (formulation) |
NA | |||||
| * | See Outlook on page 7 for details of uncertainty on the timing of follow-on competition. | |
| † | Generic competition possible in 2011. |
Table of Contents
16
| USA | EU | |||||||||
| Products | Compounds | Indication(s) | Major | Patent expiry dates | ||||||
| competitor brands | USA | EU | ||||||||
| Anti-bacterials | ||||||||||
Augmentin
|
amoxicillin/clavulanate | common bacterial potassium infections |
generic products | expired | expired | |||||
| Oncology | ||||||||||
Arzerra
|
ofatumumab | refractory chronic lymphocytic leukaemia | MabThera/Rituxan | pending | pending | |||||
Hycamtin
|
topotecan | ovarian cancer, small cell lung cancer, cervical cancer |
Doxil, Gemzar | expired | 2011 | |||||
Promacta/Revolade
|
eltrombopag | idiopathic thrombocytopenic purpura |
Nplate | 2021 | 2021 | |||||
Tykerb/Tyverb
|
lapatanib | advanced and metastatic breast cancer in HER2 positive patients |
Herceptin | 2020 | 2023 | |||||
Votrient
|
pazopanib | metastatic renal cell carcinoma | Sutent, Nexavar, Afinitor | 2021 | 2021 | |||||
| Vaccines | ||||||||||
Boostrix
|
diphtheria, tetanus, acellular pertussis |
booster vaccination | Adacel | 2017 | 2017 | |||||
Infanrix/Pediarix
|
diphtheria, tetanus, pertussis, polio, hepatitis B (HepB), inactivated antigens |
diphtheria, tetanus, pertussis, polio, hepatitis B (HepB), |
Pentacel, Pediacel, Pentaxim, Pentavac |
2017 | 2014 | |||||
Cervarix
|
HPV 16 & 18 virus like particles (VLPs), AS04 adjuvant (MPL + aluminium hydroxide) |
human papilloma virus type 16 & 18 |
Gardasil (Silgard) | 2020 | 2020 | |||||
Fluarix
|
split inactivated influenza virus subtypes A and type B antigens |
seasonal influenza | Vaxigrip, Mutagrip, Fluzone, Influvac, Aggripal, Fluad |
2022 | 2022 | |||||
FluLaval
|
split inactivated influenza virus subtypes A and type B antigens |
seasonal influenza | Vaxigrip, Mutagrip, Fluzone, Influvac, Aggripal, Fluad |
none | none | |||||
Pandemrix
|
derived split inactivated influenza virus antigen, A503 adjuvant |
A(H1N1)v2009 influenza prophylaxis |
Focetria, Celvapan, emerflu | 2014 | 2014 | |||||
Prepandrix
|
derived split inactivated influenza virus antigen, A503 adjuvant |
influenza prophylaxis | Aflunov | 2014 | 2014 | |||||
Synflorix
|
conjugated pneumococcal polysaccharide |
invasive pneumococcal disease |
Prevenar (Prevnar) | NA | 2021 | |||||
| HIV | ||||||||||
Combivir
|
lamivudine and zidovudine | HIV/AIDS | Truvada, Atripla | 20121 (combination) |
2013 (combination) |
|||||
Epivir
|
lamivudine | HIV/AIDS | Truvada, Atripla | expired | expired | |||||
Epzicom/Kivexa
|
lamivudine and abacavir | HIV/AIDS | Truvada, Atripla | 2016 (combination) |
2016 (combination) |
|||||
Lexiva
|
fosamprenavir | HIV/AIDS | Prezista, Kaletra, Reyataz | 2017 | 2019 | |||||
Selzentry
|
maraviroc | HIV/AIDS | Isentress, Intelence, Prezista | 2021 | 2021 | |||||
Trizivir
|
lamivudine, zidovudine and abacavir | HIV/AIDS | Truvada, Atripla | 2016 (combination) |
2016 (combination) |
|||||
| 1 | See Note 44 to the financial statements, ‘Legal proceedings’ |
Table of Contents
17
| Brand | Products | Application | Markets | Competition | ||||
| Oral healthcare | ||||||||
Aquafresh
|
toothpastes, toothbrushes, mouthwashes | prevention of caries, gum disease and bad breath |
global | Colgate-Palmolive’s Colgate, Procter & Gamble’s Crest | ||||
Sensodyne
|
toothpastes, toothbrushes | prevention of dental sensitivity |
global | Colgate-Palmolive sensitivity toothpastes | ||||
Biotene
|
mouthwash, gel | treat dry mouth | many markets | none | ||||
Polident Poligrip Corega |
denture adhesive, denture cleanser |
to improve comfort of fitted dentures and to clean dentures | global | Fixodent | ||||
| OTC medicines | ||||||||
Panadol
|
tablets, capulets, infant drops | paracetamol-based treatment of headache and joint pain, fever, cold symptoms |
global, except USA | Nurofen | ||||
NicoDerm, NiQuitin CQ, and Nicabate. Also Nicorette (USA only) |
gum, patch, mini lozenge, original lozenge |
treatment of nicotine withdrawal as an aid to quitting smoking |
global | Novartis’ Nicotinell, retailers’ own brands |
||||
| Nutritional healthcare | ||||||||
Lucozade
|
energy and sports drinks | energy and hydration | UK, Ireland, some other markets |
various sports drinks | ||||
Horlicks
|
malted, milk-based drinks and foods |
nutrition | UK, Ireland, India | Ovaltine, Milo | ||||
Ribena
|
blackcurrant juice-based drink | vitamin C-delivering health drink |
UK, Ireland, some other markets |
Robinsons | ||||
| • | consumers are demanding better quality, better value and improved performance | |
| • | retailers have consolidated and globalised which has strengthened their negotiation power | |
| • | cycle times for innovation have reduced. |
| • | in the USA: Metamucil (laxative), Pepcid (indigestion) and private label smoking control products | |
| • | in the UK: Lemsip (cold remedy), Nurofen and Anadin (analgesics), and Nicorette and Nicotinell (smoking control treatments). |
Table of Contents
18
Table of Contents
19

Table of Contents
20
| World market by | Value | % of | ||||||
| geographic region | £bn | total | ||||||
USA |
194 | 41 | ||||||
Europe |
129 | 27 | ||||||
Rest of World |
153 | 32 | ||||||
Emerging markets |
67 | 14 | ||||||
Asia Pacific |
20 | 4 | ||||||
Japan |
52 | 11 | ||||||
Canada |
13 | 3 | ||||||
Total |
476 | 100 | ||||||
| World market – | Value | % of | ||||||
| top six therapeutic classes | £bn | total | ||||||
Central nervous system |
76 | 16 | ||||||
Cardiovascular |
69 | 15 | ||||||
Antineoplastic/lmmunomodulatory |
63 | 13 | ||||||
Alimentary tract and metabolic |
57 | 12 | ||||||
Anti-infectives (bacterial, viral and fungal) excluding vaccines |
49 | 10 | ||||||
Respiratory |
33 | 7 | ||||||
Table of Contents
21
| 2010 | 2009 | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Group turnover |
28,392 | 28,368 | (1.2 | ) | ||||||||
Avandia, Valtrex and pandemic products |
(2,285 | ) | (3,668 | ) | ||||||||
Underlying Group turnover |
26,107 | 24,700 | 4.5 | |||||||||
| 2010 | 2009 | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Pharmaceutical turnover |
23,382 | 23,694 | (2 | ) | ||||||||
Avandia, Valtrex and pandemic products |
(2,285 | ) | (3,668 | ) | ||||||||
Underlying pharmaceutical turnover |
21,097 | 20,026 | 4 | |||||||||
| Emerging | Asia Pacific/ | Other trading | ||||||||||||||||||||||
| 2010 | USA | Europe | Markets | Japan | and unallocated | Total | ||||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||||
Pandemic products |
44 | 494 | 227 | 462 | 86 | 1,313 | ||||||||||||||||||
Avandia |
237 | 88 | 42 | 24 | 49 | 440 | ||||||||||||||||||
Valtrex |
252 | 68 | 28 | 176 | 8 | 532 | ||||||||||||||||||
| Emerging | Asia Pacific/ | Other trading | ||||||||||||||||||||||
| 2009 | USA | Europe | Markets | Japan | and unallocated | Total | ||||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||||
Pandemic products |
324 | 737 | 89 | 331 | 122 | 1,603 | ||||||||||||||||||
Avandia |
425 | 171 | 76 | 41 | 58 | 771 | ||||||||||||||||||
Valtrex |
942 | 160 | 26 | 152 | 14 | 1,294 | ||||||||||||||||||
Table of Contents
22
| 2009 | ||||||||||||
| 2010 | (restated) | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Turnover |
7,648 | 8,578 | (11 | ) | ||||||||
Operating profit |
5,043 | 5,933 | (16 | ) | ||||||||

Table of Contents
23
| 2009 | ||||||||||||
| 2010 | (restated) | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Turnover |
6,548 | 7,087 | (6 | ) | ||||||||
Operating profit |
3,744 | 3,993 | (4 | ) | ||||||||
Table of Contents
24
| 2009 | ||||||||||||
| 2010 | (restated) | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Turnover |
3,556 | 2,895 | 22 | |||||||||
Operating profit |
1,271 | 948 | 31 | |||||||||
Table of Contents
25
| 2009 | ||||||||||||
| 2010 | (restated) | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Turnover |
3,102 | 2,628 | 9 | |||||||||
Operating profit |
1,730 | 1,352 | 15 | |||||||||
| 1 | Includes 4 New Chemical Entities | |
| 2 | Includes New Chemical Entities, line extensions, new promotions or re-formulations |
Table of Contents
26
| 2009 | ||||||||||||
| 2010 | (restated) | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Turnover |
1,556 | 1,605 | (3 | ) | ||||||||
Operating profit |
851 | 1,071 | (21 | ) | ||||||||


Table of Contents
27
| 2009 | ||||||||||||
| 2010 | (restated) | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Turnover |
5,010 | 4,674 | 5 | |||||||||
Operating profit |
1,043 | 931 | 8 | |||||||||

Table of Contents
28
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Pharmaceuticals - direct project costs |
1,432 | 1,489 | 1,209 | |||||||||
(excl.
vaccines) - indirect costs |
959 | 1,056 | 844 | |||||||||
- unallocated costs |
563 | 474 | 490 | |||||||||
Pharmaceuticals R&D |
2,954 | 3,019 | 2,543 | |||||||||
In-market pharmaceutical development |
147 | 81 | 40 | |||||||||
Vaccines |
533 | 524 | 369 | |||||||||
Corporate and other costs |
172 | 177 | 304 | |||||||||
| 3,806 | 3,801 | 3,256 | ||||||||||
Consumer Healthcare |
158 | 150 | 114 | |||||||||
R&D before major restructuring |
3,964 | 3,951 | 3,370 | |||||||||
Major restructuring |
493 | 155 | 170 | |||||||||
Total R&D |
4,457 | 4,106 | 3,540 | |||||||||
| 2010 | 2009 | Growth | ||||||||||
| £m | £m | CER% | ||||||||||
Veramyst |
193 | 142 | 33 | |||||||||
Cervarix |
242 | 187 | 26 | |||||||||
Coreg CR |
157 | 161 | (3 | ) | ||||||||
Lamictal XR |
68 | 18 | >100 | |||||||||
Requip XL |
148 | 123 | 22 | |||||||||
Rotarix |
235 | 282 | (18 | ) | ||||||||
Synflorix |
221 | 73 | >100 | |||||||||
Treximet |
56 | 55 | 2 | |||||||||
Tykerb |
227 | 169 | 34 | |||||||||
Others |
180 | 52 | >100 | |||||||||
| 1,727 | 1,262 | 36 | ||||||||||
| • | In early 2010, we announced our intention to cease discovery research into certain areas of neurology, such as pain and depression, and instead concentrate activities in neurodegenerative and neuroinflammatory diseases where we feel the prospects for successful registration and launch of differentiated medicines are greater. This change led us to exit five R&D centres. In two of the largest of these – Verona, Italy and Zagreb, Croatia – the operations were transferred to external groups thereby preserving the majority of jobs. |
| • | We have successfully out-licensed and spun off some of the early stage neurology assets in the UK through deals with Convergence Pharmaceuticals and Proximagen Group. |
| • | Through these changes and other actions we have achieved a reduction in our footprint of 29% since 2006. |
| • | We continue to increase the external nature of our discovery activities. During 2010 we signed eight new collaborations to access novel discovery, giving us a total of 54 external discovery engines to complement our 38 DPUs. |
| • | We have streamlined the resourcing of our clinical trials contract research organisations, reducing this from over 100 to just two suppliers. While this provides savings in terms of economies of scale, it will also ensure consistency and rigour in clinical trials around the globe. |
| • | We combined our Molecular Discovery Research (MDR) and Preclinical Development (PCD) in 2010 to create an end-to-end scientific and technical platform supporting the discovery and development efforts. The remit of this group remains to create the materials and knowledge that enable our R&D to take ideas, generate hypotheses and test them in preclinical and clinical settings and ultimately launch new medicines. |
Table of Contents
29
| • | Commit to transparency |
| • | Show respect for people |
| • | Always demonstrate the highest integrity in our conduct |
| • | Be patient focused. |
Table of Contents
30
Table of Contents
31
Table of Contents
32
| • | carbon dioxide and other emissions that contribute to climate change |
| • | water use |
| • | environmental stewardship, which covers the use of materials, generation of waste and pollution. |
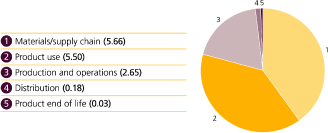
Table of Contents
33
| • | Commit to transparency |
| • | Show respect for people |
| • | Always demonstrate the highest integrity in your conduct |
| • | Be patient focused. |
Table of Contents
34
| 2010 | 2009 | Growth* | ||||||||||||||
| £m | £m | CER% | £% | |||||||||||||
USA |
7,648 | 8,578 | (11 | ) | (11 | ) | ||||||||||
Europe |
6,548 | 7,087 | (6 | ) | (8 | ) | ||||||||||
Emerging Markets |
3,556 | 2,895 | 22 | 23 | ||||||||||||
Asia Pacific/Japan |
3,102 | 2,628 | 9 | 18 | ||||||||||||
ViiV
Healthcare |
1,566 | 1,605 | (3 | ) | (2 | ) | ||||||||||
Other |
962 | 901 | (1 | ) | 7 | |||||||||||
| 23,382 | 23,694 | (2 | ) | (1 | ) | |||||||||||
| * | CER% represents growth at constant exchange rates. £% represents growth at actual exchange rates. Turnover by quarter is given on pages 188 to 192. |
Table of Contents
35
| Total | USA | Europe | Emerging Markets | Rest of World | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Therapeutic area/ | 2010 | 2009 | Growth | 2010 | Growth | 2010 | Growth | 2010 | Growth | 2010 | Growth | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| major products | £m | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | ||||||||||||||||||||||||||||||||||||||||||||||||
Respiratory |
7,238 | 6,977 | 3 | 4 | 3,394 | 1 | 2 | 2,149 | – | (2 | ) | 616 | 19 | 21 | 1,079 | 4 | 14 | |||||||||||||||||||||||||||||||||||||||||||||||
Avamys/Veramyst |
193 | 142 | 33 | 36 | 69 | – | 1 | 56 | 27 | 24 | 31 | >100 | >100 | 37 | 94 | >100 | ||||||||||||||||||||||||||||||||||||||||||||||||
Flixonase/Flonase |
164 | 171 | (5 | ) | (4 | ) | 37 | 37 | 37 | 40 | (7 | ) | (7 | ) | 39 | 11 | 11 | 48 | (30 | ) | (27 | ) | ||||||||||||||||||||||||||||||||||||||||||
Flixotide/Flovent |
804 | 775 | 2 | 4 | 431 | 8 | 9 | 159 | (9 | ) | (11 | ) | 48 | 38 | 41 | 166 | (10 | ) | (1 | ) | ||||||||||||||||||||||||||||||||||||||||||||
Seretide/Advair |
5,139 | 4,977 | 2 | 3 | 2,604 | – | – | 1,601 | 2 | – | 328 | 16 | 19 | 606 | 10 | 21 | ||||||||||||||||||||||||||||||||||||||||||||||||
Serevent |
201 | 236 | (16 | ) | (15 | ) | 64 | (12 | ) | (12 | ) | 98 | (16 | ) | (16 | ) | 2 | (33 | ) | (33 | ) | 37 | (23 | ) | (16 | ) | ||||||||||||||||||||||||||||||||||||||
Ventolin |
522 | 477 | 8 | 9 | 179 | 16 | 17 | 142 | (3 | ) | (5 | ) | 112 | 19 | 20 | 89 | (2 | ) | 10 | |||||||||||||||||||||||||||||||||||||||||||||
Zyrtec |
82 | 75 | 4 | 9 | – | – | – | – | – | – | 14 | – | – | 68 | 5 | 11 | ||||||||||||||||||||||||||||||||||||||||||||||||
Anti-virals |
1,086 | 2,416 | (56 | ) | (55 | ) | 370 | (68 | ) | (68 | ) | 109 | (73 | ) | (73 | ) | 223 | (3 | ) | (1 | ) | 384 | (44 | ) | (39 | ) | ||||||||||||||||||||||||||||||||||||||
Hepsera |
128 | 114 | 6 | 12 | – | – | – | 1 | – | – | 58 | 10 | 14 | 69 | 2 | 10 | ||||||||||||||||||||||||||||||||||||||||||||||||
Relenza |
121 | 720 | (84 | ) | (83 | ) | 43 | (69 | ) | (69 | ) | 6 | (97 | ) | (97 | ) | 1 | (97 | ) | (97 | ) | 71 | (80 | ) | (79 | ) | ||||||||||||||||||||||||||||||||||||||
Valtrex |
532 | 1,294 | (60 | ) | (59 | ) | 252 | (73 | ) | (73 | ) | 68 | (56 | ) | (58 | ) | 28 | 8 | 8 | 184 | 2 | 11 | ||||||||||||||||||||||||||||||||||||||||||
Zeffix |
233 | 217 | 4 | 7 | 13 | (24 | ) | (24 | ) | 26 | (10 | ) | (10 | ) | 136 | 17 | 18 | 58 | (5 | ) | 4 | |||||||||||||||||||||||||||||||||||||||||||
Central nervous |
1,753 | 1,870 | (8 | ) | (6 | ) | 505 | (23 | ) | (22 | ) | 540 | (4 | ) | (6 | ) | 223 | 17 | 17 | 485 | (2 | ) | 7 | |||||||||||||||||||||||||||||||||||||||||
system |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Imigran/Imitrex |
212 | 266 | (21 | ) | (20 | ) | 75 | (39 | ) | (39 | ) | 85 | (10 | ) | (11 | ) | 5 | – | – | 47 | 2 | 12 | ||||||||||||||||||||||||||||||||||||||||||
Lamictal |
504 | 500 | 1 | 1 | 257 | (4 | ) | (4 | ) | 143 | (6 | ) | (7 | ) | 57 | 23 | 19 | 47 | 42 | 52 | ||||||||||||||||||||||||||||||||||||||||||||
Requip |
233 | 209 | 11 | 11 | 44 | 69 | 69 | 137 | 2 | (1 | ) | 3 | 50 | 50 | 49 | 2 | 14 | |||||||||||||||||||||||||||||||||||||||||||||||
Seroxat/Paxil |
482 | 523 | (12 | ) | (8 | ) | 27 | (36 | ) | (36 | ) | 82 | (15 | ) | (17 | ) | 73 | (3 | ) | (4 | ) | 300 | (9 | ) | (2 | ) | ||||||||||||||||||||||||||||||||||||||
Treximet |
56 | 55 | 2 | 2 | 55 | 2 | – | – | – | – | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Wellbutrin |
81 | 132 | (39 | ) | (39 | ) | 24 | (73 | ) | (73 | ) | 39 | 33 | 30 | 13 | 30 | 30 | 5 | (25 | ) | 25 | |||||||||||||||||||||||||||||||||||||||||||
Cardiovascular |
2,570 | 2,298 | 11 | 12 | 1,571 | 10 | 11 | 610 | 7 | 5 | 134 | 25 | 24 | 255 | 23 | 33 | ||||||||||||||||||||||||||||||||||||||||||||||||
and urogenital |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Arixtra |
301 | 254 | 19 | 19 | 177 | 25 | 26 | 99 | 8 | 4 | 10 | 43 | 43 | 15 | 18 | 36 | ||||||||||||||||||||||||||||||||||||||||||||||||
Avodart |
629 | 530 | 18 | 19 | 337 | 5 | 6 | 175 | 22 | 18 | 33 | 50 | 50 | 84 | 90 | >100 | ||||||||||||||||||||||||||||||||||||||||||||||||
Coreg |
171 | 172 | (1 | ) | (1 | ) | 170 | (1 | ) | (1 | ) | – | – | – | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||||||||||
Fraxiparine |
222 | 229 | (2 | ) | (3 | ) | – | – | – | 154 | (9 | ) | (11 | ) | 55 | 29 | 31 | 13 | (7 | ) | (7 | ) | ||||||||||||||||||||||||||||||||||||||||||
Lovaza |
530 | 450 | 17 | 18 | 528 | 17 | 18 | – | – | – | – | – | – | 2 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Vesicare |
114 | 104 | 9 | 10 | 113 | 8 | 9 | – | – | – | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Volibris |
46 | 19 | >100 | >100 | – | – | – | 40 | >100 | >100 | 1 | – | – | 5 | >100 | >100 | ||||||||||||||||||||||||||||||||||||||||||||||||
Metabolic |
678 | 1,181 | (44 | ) | (43 | ) | 238 | (59 | ) | (59 | ) | 166 | (38 | ) | (40 | ) | 91 | (24 | ) | (24 | ) | 183 | (17 | ) | (11 | ) | ||||||||||||||||||||||||||||||||||||||
Avandia products |
440 | 771 | (44 | ) | (43 | ) | 237 | (45 | ) | (44 | ) | 88 | (48 | ) | (49 | ) | 42 | (43 | ) | (45 | ) | 73 | (32 | ) | (26 | ) | ||||||||||||||||||||||||||||||||||||||
Bonviva/Boniva |
78 | 255 | (69 | ) | (69 | ) | – | (100 | ) | (100 | ) | 64 | (26 | ) | (28 | ) | 2 | – | – | 12 | 22 | 33 | ||||||||||||||||||||||||||||||||||||||||||
Anti-bacterials |
1,396 | 1,457 | (4 | ) | (4 | ) | 75 | (28 | ) | (27 | ) | 536 | (14 | ) | (16 | ) | 609 | 10 | 10 | 176 | 1 | 7 | ||||||||||||||||||||||||||||||||||||||||||
Augmentin |
625 | 667 | (6 | ) | (6 | ) | 11 | (76 | ) | (76 | ) | 240 | (17 | ) | (19 | ) | 291 | 15 | 14 | 83 | 10 | 17 | ||||||||||||||||||||||||||||||||||||||||||
Oncology and |
688 | 629 | 9 | 9 | 350 | 13 | 14 | 201 | 1 | (1 | ) | 62 | 7 | 9 | 75 | 17 | 25 | |||||||||||||||||||||||||||||||||||||||||||||||
emesis |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Arzerra |
31 | 3 | >100 | >100 | 26 | >100 | >100 | 4 | – | – | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Hycamtin |
144 | 172 | (16 | ) | (16 | ) | 83 | (17 | ) | (17 | ) | 48 | (17 | ) | (19 | ) | 7 | 17 | 17 | 6 | (14 | ) | (14 | ) | ||||||||||||||||||||||||||||||||||||||||
Promacta |
31 | 13 | >100 | >100 | 25 | 92 | 92 | 5 | – | – | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Tyverb/Tykerb |
227 | 169 | 34 | 34 | 70 | 28 | 30 | 94 | 28 | 25 | 30 | 36 | 36 | 33 | 72 | 83 | ||||||||||||||||||||||||||||||||||||||||||||||||
Votrient |
38 | 1 | >100 | >100 | 33 | >100 | >100 | 4 | – | – | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Vaccines |
4,326 | 3,706 | 15 | 17 | 763 | (7 | ) | (6 | ) | 1,681 | (2 | ) | (4 | ) | 927 | 38 | 39 | 955 | 85 | 100 | ||||||||||||||||||||||||||||||||||||||||||||
Boostrix |
181 | 139 | 29 | 30 | 110 | 51 | 51 | 43 | 10 | 8 | 9 | 29 | 29 | 19 | (16 | ) | – | |||||||||||||||||||||||||||||||||||||||||||||||
Cervarix |
242 | 187 | 26 | 29 | 13 | >100 | >100 | 116 | (14 | ) | (16 | ) | 25 | 4 | 9 | 88 | >100 | >100 | ||||||||||||||||||||||||||||||||||||||||||||||
Fluarix, FluLaval |
241 | 211 | 14 | 14 | 110 | 51 | 51 | 63 | (8 | ) | (11 | ) | 40 | (5 | ) | (5 | ) | 28 | – | 12 | ||||||||||||||||||||||||||||||||||||||||||||
Flu Pandemic |
1,192 | 883 | 31 | 35 | 1 | (99 | ) | (99 | ) | 488 | (6 | ) | (7 | ) | 226 | >100 | >100 | 477 | >100 | >100 | ||||||||||||||||||||||||||||||||||||||||||||
Hepatitis |
720 | 665 | 7 | 8 | 307 | 19 | 19 | 242 | (6 | ) | (8 | ) | 88 | 8 | 10 | 83 | 15 | 26 | ||||||||||||||||||||||||||||||||||||||||||||||
Infanrix, Pediarix |
700 | 649 | 8 | 8 | 146 | 8 | 9 | 429 | 8 | 6 | 50 | 13 | 11 | 75 | 3 | 17 | ||||||||||||||||||||||||||||||||||||||||||||||||
Rotarix |
235 | 282 | (18 | ) | (17 | ) | 74 | (4 | ) | (3 | ) | 38 | (28 | ) | (28 | ) | 102 | (22 | ) | (21 | ) | 21 | (17 | ) | (13 | ) | ||||||||||||||||||||||||||||||||||||||
Synflorix |
221 | 73 | >100 | >100 | – | – | – | 43 | 38 | 34 | 149 | >100 | >100 | 29 | >100 | >100 | ||||||||||||||||||||||||||||||||||||||||||||||||
Dermatologicals |
1,087 | 707 | 51 | 54 | 358 | 70 | 70 | 246 | 48 | 45 | 286 | 52 | 56 | 197 | 26 | 37 | ||||||||||||||||||||||||||||||||||||||||||||||||
Bactroban |
119 | 123 | (3 | ) | (3 | ) | 51 | (14 | ) | (14 | ) | 27 | 8 | 4 | 28 | 7 | 4 | 13 | – | 18 | ||||||||||||||||||||||||||||||||||||||||||||
Dermovate |
74 | – | – | – | – | – | – | 19 | – | – | 30 | – | – | 25 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Duac |
116 | 46 | >100 | >100 | 67 | >100 | >100 | 23 | >100 | >100 | 11 | >100 | >100 | 15 | >100 | >100 | ||||||||||||||||||||||||||||||||||||||||||||||||
Soriatane |
71 | 28 | >100 | >100 | 71 | >100 | >100 | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Zovirax |
152 | 129 | 15 | 18 | 53 | >100 | >100 | 27 | (10 | ) | (10 | ) | 26 | 9 | 13 | 46 | (14 | ) | (8 | ) | ||||||||||||||||||||||||||||||||||||||||||||
Other |
994 | 848 | 16 | 17 | 24 | 53 | 41 | 310 | 9 | 6 | 385 | 37 | 38 | 275 | – | 6 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 21,816 | 22,089 | (2 | ) | (1 | ) | 7,648 | (11 | ) | (11 | ) | 6,548 | (6 | ) | (8 | ) | 3,556 | 22 | 23 | 4,064 | 6 | 15 | |||||||||||||||||||||||||||||||||||||||||||
ViiV Healthcare
(HIV) |
1,566 | 1,605 | (3 | ) | (2 | ) | 660 | (8 | ) | (8 | ) | 585 | (5 | ) | (8 | ) | 146 | 35 | 39 | 175 | 7 | 16 | ||||||||||||||||||||||||||||||||||||||||||
Combivir |
363 | 425 | (16 | ) | (15 | ) | 143 | (24 | ) | (24 | ) | 117 | (21 | ) | (23 | ) | 63 | 22 | 29 | 40 | (3 | ) | 5 | |||||||||||||||||||||||||||||||||||||||||
Epivir |
115 | 129 | (12 | ) | (11 | ) | 40 | (17 | ) | (17 | ) | 37 | (22 | ) | (24 | ) | 18 | 31 | 38 | 20 | – | 5 | ||||||||||||||||||||||||||||||||||||||||||
Epzicom/Kivexa |
555 | 546 | 1 | 2 | 210 | (7 | ) | (6 | ) | 245 | 3 | – | 29 | 38 | 38 | 71 | 14 | 22 | ||||||||||||||||||||||||||||||||||||||||||||||
Lexiva |
155 | 178 | (12 | ) | (13 | ) | 80 | (19 | ) | (19 | ) | 51 | (15 | ) | (18 | ) | 13 | 86 | 86 | 11 | – | 10 | ||||||||||||||||||||||||||||||||||||||||||
Selzentry |
80 | 1 | >100 | >100 | 34 | – | – | 41 | >100 | >100 | 2 | – | – | 3 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||
Trizivir |
144 | 201 | (28 | ) | (28 | ) | 73 | (30 | ) | (30 | ) | 60 | (26 | ) | (27 | ) | 4 | (43 | ) | (43 | ) | 7 | (13 | ) | (13 | ) | ||||||||||||||||||||||||||||||||||||||
| 23,382 | 23,694 | (2 | ) | (1 | ) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table of Contents
36
| % of | 2010 | 2009 | Growth* | |||||||||||||||||
| total | £m | £m | CER% | £% | ||||||||||||||||
Over-the-counter |
||||||||||||||||||||
medicines |
49 | 2,456 | 2,339 | 3 | 5 | |||||||||||||||
Oral healthcare |
32 | 1,602 | 1,484 | 6 | 8 | |||||||||||||||
Nutritional healthcare |
19 | 952 | 851 | 9 | 12 | |||||||||||||||
| 100 | 5,010 | 4,674 | 5 | 7 | ||||||||||||||||
| * | CER% represents growth at constant exchange rates. £% represents growth at actual exchange rates. Turnover by quarter is given on page 199. |
Table of Contents
37
| 2010 | 2009 | Growth | ||||||||||||||||||||||
| £m | % | £m | % | CER% | £% | |||||||||||||||||||
Turnover |
28,392 | 100 | 28,368 | 100 | (1 | ) | – | |||||||||||||||||
Cost of sales |
(7,592 | ) | (26.7 | ) | (7,380 | ) | (26.0 | ) | 3 | 3 | ||||||||||||||
Selling, general and administration |
(13,053 | ) | (46.0 | ) | (9,592 | ) | (33.8 | ) | 36 | 36 | ||||||||||||||
Research and
development |
(4,457 | ) | (15.7 | ) | (4,106 | ) | (14.4 | ) | 8 | 9 | ||||||||||||||
Other operating
income |
493 | 1.7 | 1,135 | 3.9 | ||||||||||||||||||||
Operating profit |
3,783 | 13.3 | 8,425 | 29.7 | (59 | ) | (55 | ) | ||||||||||||||||
Table of Contents
38
| 2010 | 2009 | |||||||
| Finance income | £m | £m | ||||||
Interest and other finance income |
102 | 67 | ||||||
Unwinding of discounts on assets |
1 | 2 | ||||||
Fair value adjustments and hedges |
13 | 1 | ||||||
| 116 | 70 | |||||||
Finance costs
|
||||||||
Interest costs |
(767 | ) | (770 | ) | ||||
Unwinding of discounts on liabilities |
(18 | ) | (11 | ) | ||||
Fair value adjustments and hedges |
(21 | ) | (2 | ) | ||||
Other finance expense |
(25 | ) | – | |||||
| (831 | ) | (783 | ) | |||||
| 2010 | 2009 | Growth | ||||||||||||||||||||||
| £m | % | £m | % | CER% | £% | |||||||||||||||||||
Turnover |
28,392 | 100 | 28,368 | 100 | (1 | ) | – | |||||||||||||||||
Cost of sales |
(7,405 | ) | (26.1 | ) | (7,095 | ) | (25.0 | ) | 4 | 4 | ||||||||||||||
Selling, general and administration |
(12,388 | ) | (43.6 | ) | (9,200 | ) | (32.4 | ) | 35 | 35 | ||||||||||||||
Research and
development |
(3,964 | ) | (14.0 | ) | (3,951 | ) | (13.9 | ) | – | – | ||||||||||||||
Other operating
income |
493 | 1.8 | 1,135 | 3.9 | ||||||||||||||||||||
Operating profit |
5,128 | 18.1 | 9,257 | 32.6 | (48 | ) | (45 | ) | ||||||||||||||||
Table of Contents
39
| Finance income | 2010 | 2009 | ||||||
| £m | £m | |||||||
Interest and other income |
102 | 67 | ||||||
Unwinding of discounts on assets |
1 | 2 | ||||||
Fair value adjustments and hedges |
13 | 1 | ||||||
| 116 | 70 | |||||||
Finance costs |
||||||||
Interest costs |
(767 | ) | (770 | ) | ||||
Unwinding of discounts on liabilities |
(15 | ) | (8 | ) | ||||
Fair value adjustments and hedges |
(21 | ) | (2 | ) | ||||
Other finance expense |
(25 | ) | – | |||||
| (828 | ) | (780 | ) | |||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
UK corporation tax at the UK statutory rate |
82 | 600 | ||||||
Less double taxation relief |
(156 | ) | (183 | ) | ||||
| (74 | ) | 417 | ||||||
Overseas taxation |
1,496 | 1,997 | ||||||
Current taxation |
1,422 | 2,414 | ||||||
Deferred taxation |
(118 | ) | (192 | ) | ||||
Taxation on total profits |
1,304 | 2,222 | ||||||
| 2010 | 2009 | Growth | ||||||||||||||
| £m | £m | CER% | £% | |||||||||||||
Total profit after taxation for the year |
1,853 | 5,669 | (71 | ) | (67 | ) | ||||||||||
Total profit attributable to shareholders |
1,634 | 5,531 | (75 | ) | (70 | ) | ||||||||||
Basic earnings per share (pence) |
32.1 | p | 109.1 | p | (75 | ) | (71 | ) | ||||||||
Basic earnings per ADS (US$) |
$1.00 | $3.40 | ||||||||||||||
Results before major restructuring profit after taxation for the year |
2,961 | 6,283 | (56 | ) | (53 | ) | ||||||||||
Results before major restructuring
profit attributable to shareholders |
2,742 | 6,145 | (59 | ) | (55 | ) | ||||||||||
Adjusted earnings per share (pence) |
53.9 | p | 121.2 | p | (59 | ) | (56 | ) | ||||||||
Adjusted earnings per ADS (US$) |
$1.67 | $3.78 | ||||||||||||||
Weighted average number of shares (millions) |
5,085 | 5,069 | ||||||||||||||
Diluted total earnings per share (pence) |
31.9 | p | 108.2 | p | ||||||||||||
Diluted total earnings per ADS (US$) |
$0.99 | $3.38 | ||||||||||||||
Diluted weighted average number of shares (millions) |
5,129 | 5,108 | ||||||||||||||
Table of Contents
40
| • | Turnover | |
| • | Taxation | |
| • | Legal and other disputes | |
| • | Property, plant & equipment | |
| • | Goodwill | |
| • | Other intangible assets | |
| • | Pensions and other post-employment benefits. |
| • | GSK has arrangements with certain indirect customers whereby the customer is able to buy products from wholesalers at reduced prices. A chargeback represents the difference between the invoice price to the wholesaler and the indirect customer’s contractual discounted price. Accruals for estimating chargebacks are calculated based on the terms of each agreement, historical experience and product growth rates | |
| • | Customer rebates are offered to key managed care and group purchasing organisations (GPO) and other direct and indirect customers. These arrangements require the customer to achieve certain performance targets relating to the value of product purchased, formulary status or pre-determined market shares relative to competitors. The accrual for customer rebates is estimated based on the specific terms in each agreement, historical experience and product growth rates | |
| • | The US Medicaid programme is a state-administered programme providing assistance to certain poor and vulnerable patients. In 1990, the Medicaid Drug Rebate Program was established to reduce state and federal expenditure on prescription drugs. In 2010, the Patient and Affordable Care Act became law. GSK participates by providing rebates to states. Accruals for Medicaid rebates are calculated based on the specific terms of individual state agreements using a combination of historical experience, product and population growth, anticipated price increases and the impact of contracting strategies | |
| • | Cash discounts are offered to customers to encourage prompt payment. These are accrued for at the time of invoicing and adjusted subsequently to reflect actual experience | |
| • | Where there is historical experience of customer returns, GSK records an accrual for estimated sales returns by applying historical experience of customer returns to the amounts invoiced, together with market related information such as stock levels at wholesalers, anticipated price increases and competitor activity. |
| 2009 | 2008 | |||||||||||||||||||||||
| 2010 | (restated) | (restated) | ||||||||||||||||||||||
| £m | % | £m | % | £m | % | |||||||||||||||||||
Gross turnover |
10,802 | 100 | 11,674 | 100 | 10,782 | 100 | ||||||||||||||||||
Chargebacks |
(993 | ) | 9 | (1,124 | ) | 10 | (836 | ) | 8 | |||||||||||||||
Managed care, Medicare
Part D and GPO rebates |
(894 | ) | 8 | (907 | ) | 8 | (756 | ) | 7 | |||||||||||||||
US government and
state programmes |
(742 | ) | 7 | (542 | ) | 5 | (470 | ) | 4 | |||||||||||||||
Cash discounts |
(193 | ) | 2 | (200 | ) | 2 | (191 | ) | 1 | |||||||||||||||
Customer returns |
(179 | ) | 1 | (172 | ) | 1 | (118 | ) | 1 | |||||||||||||||
Prior year adjustments |
38 | – | 24 | – | 35 | – | ||||||||||||||||||
Other items |
(191 | ) | 2 | (175 | ) | 1 | (192 | ) | 2 | |||||||||||||||
Total deductions |
(3,154 | ) | 29 | (3,096 | ) | 27 | (2,528 | ) | 23 | |||||||||||||||
Net turnover |
7,648 | 71 | 8,578 | 73 | 8,254 | 77 | ||||||||||||||||||
| At 31st | ||||||||
| At 31st | December | |||||||
| December | 2009 | |||||||
| 2010 | (restated) | |||||||
| £m | £m | |||||||
Chargebacks |
50 | 41 | ||||||
Managed care, Medicare Part D and GPO rebates |
422 | 426 | ||||||
US government and state programmes |
445 | 322 | ||||||
Cash discounts |
21 | 20 | ||||||
Customer returns |
254 | 192 | ||||||
Other |
28 | 26 | ||||||
Total |
1,220 | 1,027 | ||||||
Table of Contents
41
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Assets |
||||||||
Non-current assets |
||||||||
Property, plant and equipment |
9,045 | 9,374 | ||||||
Goodwill |
3,606 | 3,361 | ||||||
Other intangible assets |
8,532 | 8,183 | ||||||
Investments in associates and joint ventures |
1,081 | 895 | ||||||
Other investments |
711 | 454 | ||||||
Deferred tax assets |
2,566 | 2,374 | ||||||
Derivative financial instruments |
97 | 68 | ||||||
Other non-current assets |
556 | 583 | ||||||
Total non-current assets |
26,194 | 25,292 | ||||||
Current assets |
||||||||
Inventories |
3,837 | 4,064 | ||||||
Current tax recoverable |
56 | 58 | ||||||
Trade and other receivables |
5,793 | 6,492 | ||||||
Derivative financial instruments |
93 | 129 | ||||||
Liquid investments |
184 | 268 | ||||||
Cash and cash equivalents |
6,057 | 6,545 | ||||||
Assets held for sale |
16 | 14 | ||||||
Total current assets |
16,036 | 17,570 | ||||||
Total assets |
42,230 | 42,862 | ||||||
Liabilities |
||||||||
Current liabilities |
||||||||
Short-term borrowings |
(291 | ) | (1,471 | ) | ||||
Trade and other payables |
(6,888 | ) | (6,772 | ) | ||||
Derivative financial instruments |
(188 | ) | (168 | ) | ||||
Current tax payable |
(1,047 | ) | (1,451 | ) | ||||
Short-term provisions |
(4,380 | ) | (2,256 | ) | ||||
Total current liabilities |
(12,794 | ) | (12,118 | ) | ||||
Non-current liabilities |
||||||||
Long-term borrowings |
(14,809 | ) | (14,786 | ) | ||||
Deferred tax liabilities |
(707 | ) | (645 | ) | ||||
Pensions and other post-employment benefits |
(2,672 | ) | (2,981 | ) | ||||
Other provisions |
(904 | ) | (985 | ) | ||||
Derivative financial instruments |
(5 | ) | – | |||||
Other non-current liabilities |
(594 | ) | (605 | ) | ||||
Total non-current liabilities |
(19,691 | ) | (20,002 | ) | ||||
Total liabilities |
(32,485 | ) | (32,120 | ) | ||||
Net assets |
9,745 | 10,742 | ||||||
Equity |
||||||||
Share capital |
1,418 | 1,416 | ||||||
Share premium account |
1,428 | 1,368 | ||||||
Retained earnings |
4,779 | 6,321 | ||||||
Other reserves |
1,262 | 900 | ||||||
Shareholders’ equity |
8,887 | 10,005 | ||||||
Non-controlling interests |
858 | 737 | ||||||
Total equity |
9,745 | 10,742 | ||||||
Table of Contents
42
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Cash, cash equivalents and liquid investments |
6,241 | 6,813 | ||||||
Borrowings – repayable within one year |
(291 | ) | (1,471 | ) | ||||
Borrowings – repayable after one year |
(14,809 | ) | (14,786 | ) | ||||
Net debt |
(8,859 | ) | (9,444 | ) | ||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Net debt at beginning of year |
(9,444 | ) | (10,173 | ) | ||||
(Decrease)/increase in cash and bank overdrafts |
(642 | ) | 1,054 | |||||
Cash inflow from liquid investments |
(91 | ) | (87 | ) | ||||
Net increase in long-term loans |
– | (1,358 | ) | |||||
Net repayment of short-term loans |
1,290 | 102 | ||||||
Debt of subsidiary undertakings acquired |
(20 | ) | (9 | ) | ||||
Exchange movements |
61 | 1,041 | ||||||
Other movements |
(13 | ) | (14 | ) | ||||
Net debt at end of year |
(8,859 | ) | (9,444 | ) | ||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Total equity at beginning of year |
10,742 | 8,318 | ||||||
Total comprehensive income for the year |
2,086 | 4,996 | ||||||
Dividends to shareholders |
(3,205 | ) | (3,003 | ) | ||||
Ordinary Shares issued |
62 | 43 | ||||||
Changes in
non-controlling interests |
– | 338 | ||||||
Put option over non-controlling interest |
– | (2 | ) | |||||
Consideration received for shares transferred by ESOP Trusts |
17 | 13 | ||||||
Ordinary Shares acquired by ESOP Trusts |
(16 | ) | (57 | ) | ||||
Share-based incentive plans |
175 | 171 | ||||||
Tax on share-based incentive plans |
2 | 14 | ||||||
Distributions to non-controlling interests |
(118 | ) | (89 | ) | ||||
Total equity at end of year |
9,745 | 10,742 | ||||||
Table of Contents
43
| Total | Under 1 yr | 1-3 yrs | 3-5 yrs | 5 yrs+ | ||||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Loans |
14,997 | 259 | 4,158 | 2,407 | 8,173 | |||||||||||||||
Interest on loans |
10,312 | 755 | 1,394 | 1,097 | 7,066 | |||||||||||||||
Finance lease obligations |
103 | 32 | 45 | 18 | 8 | |||||||||||||||
Finance lease charges |
16 | 5 | 8 | 3 | – | |||||||||||||||
Operating lease commitments |
415 | 123 | 119 | 57 | 116 | |||||||||||||||
Intangible assets |
11,762 | 720 | 1,626 | 2,150 | 7,266 | |||||||||||||||
Property, plant & equipment |
380 | 278 | 95 | 7 | – | |||||||||||||||
Business combinations |
285 | 253 | 12 | 20 | – | |||||||||||||||
Investments |
37 | 16 | – | 21 | – | |||||||||||||||
Purchase commitments |
1,127 | 239 | 314 | 293 | 281 | |||||||||||||||
Pensions |
1,095 | 365 | 730 | – | – | |||||||||||||||
Other commitments |
242 | 110 | 78 | 49 | 5 | |||||||||||||||
Total |
40,771 | 3,155 | 8,579 | 6,122 | 22,915 | |||||||||||||||
| Total | Under 1 yr | 1-3 yrs | 3-5 yrs | 5 yrs+ | ||||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Guarantees |
110 | 64 | 1 | 1 | 44 | |||||||||||||||
Other contingent liabilities |
55 | 22 | 10 | 1 | 22 | |||||||||||||||
Total |
165 | 86 | 11 | 2 | 66 | |||||||||||||||
Table of Contents
44
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Net cash inflow from operating activities |
6,797 | 7,841 | ||||||
Net cash outflow from investing activities |
(1,868 | ) | (4,013 | ) | ||||
Net cash outflow from financing activities |
(5,571 | ) | (2,774 | ) | ||||
(Decrease)/increase in cash and bank overdrafts |
(642 | ) | 1,054 | |||||
Exchange adjustments |
81 | (158 | ) | |||||
Cash and bank overdrafts at beginning of year |
6,368 | 5,472 | ||||||
Cash and bank overdrafts at end of year |
5,807 | 6,368 | ||||||
Cash and bank overdrafts at end of year
comprise: |
||||||||
Cash and cash equivalents |
6,057 | 6,545 | ||||||
Overdrafts |
(250 | ) | (177 | ) | ||||
| 5,807 | 6,368 | |||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Net cash inflow from operating activities |
6,797 | 7,841 | ||||||
Purchase of property, plant and equipment |
(1,014 | ) | (1,418 | ) | ||||
Purchase of non-current intangible assets |
(621 | ) | (455 | ) | ||||
Disposal of property, plant and equipment |
92 | 48 | ||||||
Interest paid |
(775 | ) | (780 | ) | ||||
Interest received |
107 | 90 | ||||||
Dividends received from joint ventures and associated undertaking |
18 | 17 | ||||||
Distributions to
non-controlling interests |
(118 | ) | (89 | ) | ||||
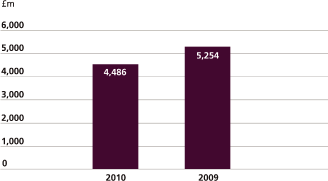
Free cash flow |
4,486 | 5,254 | ||||||
Table of Contents
45
| • | to settle terms of payment with suppliers when agreeing the terms of the transaction | |
| • | to ensure that suppliers are made aware of the agreed terms of payment | |
| • | to abide by the terms of payment. |
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Bank balances and deposits |
5,660 | 5,206 | ||||||
US Treasury and Treasury repo only money market funds |
360 | 1,305 | ||||||
Corporate debt instruments |
10 | 10 | ||||||
Government securities |
211 | 292 | ||||||
| 6,241 | 6,813 | |||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Cash and liquid investments |
6,241 | 6,813 | ||||||
Gross debt – fixed |
(13,740 | ) | (13,706 | ) | ||||
– floating |
(1,358 | ) | (2,550 | ) | ||||
– non-interest bearing |
(2 | ) | (1 | ) | ||||
Net debt |
(8,859 | ) | (9,444 | ) | ||||
Table of Contents
46
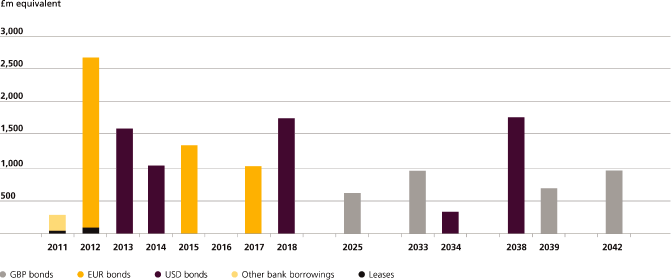
Table of Contents
47
Table of Contents
48
| Total | USA | Europe | Rest of World | |||||||||||||||||||||||||||||||||||||||||||||||||
| Therapeutic area/ | 2009 | 2008 | Growth | 2009 | Growth | 2009 | Growth | 2009 | Growth | |||||||||||||||||||||||||||||||||||||||||||
| major products | £m | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||||
Respiratory |
6,977 | 5,817 | 5 | 20 | 3,323 | 3 | 22 | 2,201 | 3 | 11 | 1,453 | 14 | 30 | |||||||||||||||||||||||||||||||||||||||
Avamys/Veramyst |
142 | 72 | 72 | 97 | 68 | 2 | 21 | 45 | >100 | >100 | 29 | >100 | >100 | |||||||||||||||||||||||||||||||||||||||
Flixonase/Flonase |
171 | 186 | (20 | ) | (8 | ) | 27 | (56 | ) | (48 | ) | 43 | (21 | ) | (17 | ) | 101 | 2 | 23 | |||||||||||||||||||||||||||||||||
Flixotide/Flovent |
775 | 677 | – | 14 | 396 | 5 | 25 | 178 | (4 | ) | 2 | 201 | (6 | ) | 9 | |||||||||||||||||||||||||||||||||||||
Seretide/Advair |
4,977 | 4,137 | 5 | 20 | 2,592 | 1 | 20 | 1,609 | 5 | 14 | 776 | 23 | 39 | |||||||||||||||||||||||||||||||||||||||
Serevent |
236 | 263 | (19 | ) | (10 | ) | 73 | (14 | ) | 1 | 116 | (18 | ) | (15 | ) | 47 | (31 | ) | (15 | ) | ||||||||||||||||||||||||||||||||
Ventolin |
477 | 339 | 26 | 41 | 153 | >100 | >100 | 150 | 1 | 9 | 174 | 2 | 12 | |||||||||||||||||||||||||||||||||||||||
Zyrtec |
75 | 38 | 58 | 97 | – | – | – | – | – | – | 75 | 58 | 97 | |||||||||||||||||||||||||||||||||||||||
Anti-virals |
4,150 | 3,206 | 12 | 29 | 1,897 | – | 19 | 1,074 | 16 | 26 | 1,179 | 32 | 56 | |||||||||||||||||||||||||||||||||||||||
HIV |
1,605 | 1,513 | (7 | ) | 6 | 716 | (6 | ) | 12 | 635 | (10 | ) | – | 254 | (3 | ) | 7 | |||||||||||||||||||||||||||||||||||
Agenerase, Lexiva |
178 | 160 | (4 | ) | 11 | 99 | 1 | 19 | 62 | (8 | ) | 2 | 17 | (13 | ) | 6 | ||||||||||||||||||||||||||||||||||||
Combivir |
425 | 433 | (13 | ) | (2 | ) | 187 | (12 | ) | 4 | 151 | (17 | ) | (9 | ) | 87 | (7 | ) | – | |||||||||||||||||||||||||||||||||
Epivir |
129 | 139 | (19 | ) | (7 | ) | 48 | (13 | ) | 2 | 49 | (24 | ) | (16 | ) | 32 | (18 | ) | (6 | ) | ||||||||||||||||||||||||||||||||
Epzicom/Kivexa |
546 | 442 | 8 | 24 | 223 | 6 | 25 | 244 | 6 | 17 | 79 | 25 | 44 | |||||||||||||||||||||||||||||||||||||||
Trizivir |
201 | 212 | (17 | ) | (5 | ) | 104 | (17 | ) | (2 | ) | 82 | (21 | ) | (11 | ) | 15 | – | 7 | |||||||||||||||||||||||||||||||||
Ziagen |
105 | 106 | (13 | ) | (1 | ) | 51 | (4 | ) | 13 | 35 | (14 | ) | (3 | ) | 19 | (28 | ) | (24 | ) | ||||||||||||||||||||||||||||||||
Valtrex |
1,294 | 1,195 | (8 | ) | 8 | 942 | (9 | ) | 8 | 160 | – | 11 | 192 | (13 | ) | 6 | ||||||||||||||||||||||||||||||||||||
Relenza |
720 | 57 | >100 | >100 | 137 | >100 | >100 | 212 | >100 | >100 | 371 | >100 | >100 | |||||||||||||||||||||||||||||||||||||||
Zeffix |
217 | 188 | (1 | ) | 15 | 17 | (7 | ) | 13 | 29 | (4 | ) | 7 | 171 | – | 17 | ||||||||||||||||||||||||||||||||||||
Central nervous system |
1,870 | 2,897 | (44 | ) | (35 | ) | 651 | (69 | ) | (64 | ) | 574 | (7 | ) | 2 | 645 | 4 | 25 | ||||||||||||||||||||||||||||||||||
Imigran/lmitrex |
266 | 687 | (65 | ) | (61 | ) | 123 | (79 | ) | (78 | ) | 96 | (8 | ) | – | 47 | (2 | ) | 15 | |||||||||||||||||||||||||||||||||
Lamictal |
500 | 926 | (53 | ) | (46 | ) | 267 | (68 | ) | (62 | ) | 154 | (4 | ) | 5 | 79 | 6 | 16 | ||||||||||||||||||||||||||||||||||
Requip |
209 | 266 | (30 | ) | (21 | ) | 26 | (78 | ) | (75 | ) | 138 | (5 | ) | 4 | 45 | 16 | 45 | ||||||||||||||||||||||||||||||||||
Requip XL |
123 | 43 | >100 | >100 | 32 | >100 | >100 | 89 | >100 | >100 | 2 | – | – | |||||||||||||||||||||||||||||||||||||||
Seroxat/Paxil |
523 | 514 | (15 | ) | 2 | 42 | (51 | ) | (47 | ) | 99 | (21 | ) | (14 | ) | 382 | (5 | ) | 19 | |||||||||||||||||||||||||||||||||
Treximet |
55 | 25 | 88 | >100 | 55 | 84 | >100 | – | – | – | – | – | – | |||||||||||||||||||||||||||||||||||||||
Wellbutrin, Wellbutrin XL |
132 | 342 | (67 | ) | (61 | ) | 88 | (76 | ) | (72 | ) | 30 | 50 | 67 | 14 | (7 | ) | – | ||||||||||||||||||||||||||||||||||
Cardiovascular and urogenital |
2,298 | 1,847 | 8 | 24 | 1,415 | 8 | 28 | 583 | 3 | 14 | 300 | 18 | 32 | |||||||||||||||||||||||||||||||||||||||
Arixtra |
254 | 170 | 29 | 49 | 141 | 35 | 60 | 95 | 18 | 34 | 18 | 55 | 64 | |||||||||||||||||||||||||||||||||||||||
Avodart |
530 | 399 | 16 | 33 | 319 | 11 | 32 | 148 | 13 | 25 | 63 | 51 | 62 | |||||||||||||||||||||||||||||||||||||||
Coreg |
172 | 203 | (29 | ) | (15 | ) | 171 | (28 | ) | (15 | ) | – | – | – | 1 | (67 | ) | (67 | ) | |||||||||||||||||||||||||||||||||
Fraxiparine |
229 | 226 | (7 | ) | 1 | – | – | – | 173 | (10 | ) | (3 | ) | 56 | 6 | 17 | ||||||||||||||||||||||||||||||||||||
Levitra |
75 | 60 | 7 | 25 | 70 | 4 | 23 | 4 | 33 | 33 | 1 | – | – | |||||||||||||||||||||||||||||||||||||||
Lovaza |
450 | 290 | 31 | 55 | 448 | 31 | 55 | – | – | – | 2 | 100 | 100 | |||||||||||||||||||||||||||||||||||||||
Vesicare |
104 | 71 | 24 | 46 | 104 | 24 | 46 | – | – | – | – | – | – | |||||||||||||||||||||||||||||||||||||||
Volibris |
19 | 2 | >100 | >100 | – | – | – | 18 | >100 | >100 | 1 | – | – | |||||||||||||||||||||||||||||||||||||||
Metabolic |
1,181 | 1,191 | (14 | ) | (1 | ) | 581 | (17 | ) | (2 | ) | 275 | (15 | ) | (6 | ) | 325 | (8 | ) | 6 | ||||||||||||||||||||||||||||||||
Avandia products |
771 | 805 | (16 | ) | (4 | ) | 425 | (17 | ) | (2 | ) | 171 | (21 | ) | (14 | ) | 175 | (9 | ) | 1 | ||||||||||||||||||||||||||||||||
Avandia |
462 | 512 | (21 | ) | (10 | ) | 276 | (22 | ) | (8 | ) | 67 | (24 | ) | (18 | ) | 119 | (18 | ) | (9 | ) | |||||||||||||||||||||||||||||||
Avandamet |
268 | 256 | (8 | ) | 5 | 122 | (6 | ) | 12 | 99 | (19 | ) | (11 | ) | 47 | 19 | 31 | |||||||||||||||||||||||||||||||||||
Bonviva/Boniva |
255 | 237 | (7 | ) | 8 | 155 | (16 | ) | (1 | ) | 89 | 7 | 20 | 11 | 57 | 57 | ||||||||||||||||||||||||||||||||||||
Anti-bacterials |
1,592 | 1,429 | 2 | 11 | 173 | (16 | ) | (1 | ) | 662 | (4 | ) | 4 | 757 | 13 | 22 | ||||||||||||||||||||||||||||||||||||
Augmentin |
667 | 587 | 4 | 14 | 45 | (22 | ) | (8 | ) | 295 | – | 8 | 327 | 14 | 23 | |||||||||||||||||||||||||||||||||||||
Oncology and emesis |
629 | 496 | 10 | 27 | 308 | 7 | 27 | 204 | 10 | 21 | 117 | 23 | 39 | |||||||||||||||||||||||||||||||||||||||
Hycamtin |
172 | 140 | 7 | 23 | 100 | 4 | 23 | 59 | 10 | 20 | 13 | 20 | 30 | |||||||||||||||||||||||||||||||||||||||
Promacta |
13 | – | – | – | 13 | – | – | – | – | – | – | – | – | |||||||||||||||||||||||||||||||||||||||
Tyverb/Tykerb |
169 | 102 | 45 | 66 | 54 | (4 | ) | 15 | 75 | 62 | 79 | 40 | >100 | >100 | ||||||||||||||||||||||||||||||||||||||
Zofran |
109 | 110 | (11 | ) | (1 | ) | 9 | >100 | >100 | 52 | (24 | ) | (17 | ) | 48 | (5 | ) | 9 | ||||||||||||||||||||||||||||||||||
Vaccines |
3,706 | 2,539 | 30 | 46 | 815 | 9 | 30 | 1,744 | 37 | 51 | 1,147 | 37 | 52 | |||||||||||||||||||||||||||||||||||||||
Boostrix |
139 | 70 | 73 | 99 | 73 | 77 | >100 | 40 | 38 | 54 | 26 | >100 | >100 | |||||||||||||||||||||||||||||||||||||||
Cervarix |
187 | 125 | 38 | 50 | 4 | – | – | 138 | 23 | 33 | 45 | 100 | >100 | |||||||||||||||||||||||||||||||||||||||
Fluarix, FluLaval |
211 | 215 | (13 | ) | (2 | ) | 73 | (27 | ) | (14 | ) | 71 | (18 | ) | (9 | ) | 67 | 17 | 29 | |||||||||||||||||||||||||||||||||
Flu pandemic |
883 | 66 | >100 | >100 | 187 | >100 | >100 | 525 | >100 | >100 | 171 | >100 | >100 | |||||||||||||||||||||||||||||||||||||||
Hepatitis (Engerix/ |
665 | 665 | (11 | ) | – | 257 | (21 | ) | (7 | ) | 262 | (8 | ) | – | 146 | 2 | 15 | |||||||||||||||||||||||||||||||||||
Fendrix, Havrix, Twinrix) |
||||||||||||||||||||||||||||||||||||||||||||||||||||
Infanrix, Pediarix |
649 | 682 | (15 | ) | (5 | ) | 134 | (47 | ) | (37 | ) | 406 | (3 | ) | 8 | 109 | 5 | 17 | ||||||||||||||||||||||||||||||||||
Rotarix |
282 | 167 | 50 | 69 | 76 | >100 | >100 | 53 | 14 | 23 | 153 | 33 | 49 | |||||||||||||||||||||||||||||||||||||||
Synflorix |
73 | – | – | – | – | – | – | 32 | – | – | 41 | – | – | |||||||||||||||||||||||||||||||||||||||
Other |
1,063 | 959 | 1 | 11 | 17 | – | 6 | 364 | 7 | 13 | 662 | (2 | ) | 10 | ||||||||||||||||||||||||||||||||||||||
| 23,446 | 20,381 | 1 | 15 | 9,180 | (13 | ) | 3 | 7,681 | 9 | 18 | 6,585 | 16 | 32 | |||||||||||||||||||||||||||||||||||||||
Stiefel products |
248 | – | – | |||||||||||||||||||||||||||||||||||||||||||||||||
| 23,714 | 2 | 16 | ||||||||||||||||||||||||||||||||||||||||||||||||||
Table of Contents
49
co-promotion income within each market.
| 2009 | 2008 | Growth* | ||||||||||||||
| £m | £m | CER% | £% | |||||||||||||
USA |
9,180 | 8,894 | (13 | ) | 3 | |||||||||||
Europe |
7,681 | 6,483 | 9 | 18 | ||||||||||||
Emerging Markets |
2,973 | 2,290 | 20 | 30 | ||||||||||||
Asia Pacific/Japan |
2,700 | 1,918 | 16 | 41 | ||||||||||||
Other
trading‡ |
1,180 | 796 | 29 | 46 | ||||||||||||
| 23,714 | 20,381 | 2 | 16 | |||||||||||||
| * | CER% represents growth at constant exchange rates. £% represents growth at actual exchange rates. | |
| ‡ | Including Stiefel |
| % of | 2009 | 2008 | Growth* | |||||||||||||||||
| total | £m | £m | CER% | £% | ||||||||||||||||
Over-the-counter
medicines |
50 | 2,319 | 1,935 | 8 | 21 | |||||||||||||||
alli |
203 | 75 | >100 | >100 | ||||||||||||||||
Breathe Right |
92 | 81 | (1 | ) | 14 | |||||||||||||||
Cold sore franchise |
96 | 89 | (3 | ) | 8 | |||||||||||||||
Nicotine replacement therapy |
339 | 299 | (1 | ) | 13 | |||||||||||||||
Panadol franchise |
393 | 324 | 10 | 21 | ||||||||||||||||
Tums |
106 | 91 | (1 | ) | 16 | |||||||||||||||
Oral healthcare |
32 | 1,484 | 1,240 | 7 | 20 | |||||||||||||||
Aquafresh franchise |
496 | 452 | (1 | ) | 10 | |||||||||||||||
Biotene |
26 | 1 | >100 | >100 | ||||||||||||||||
Denture care |
336 | 271 | 8 | 24 | ||||||||||||||||
Sensodyne franchise |
457 | 363 | 13 | 26 | ||||||||||||||||
Nutritional healthcare |
18 | 851 | 796 | 3 | 7 | |||||||||||||||
Lucozade |
376 | 382 | (3 | ) | (2 | ) | ||||||||||||||
Horlicks |
255 | 204 | 17 | 25 | ||||||||||||||||
Ribena |
160 | 161 | (4 | ) | (1 | ) | ||||||||||||||
| 100 | 4,654 | 3,971 | 7 | 18 | ||||||||||||||||
| * | CER% represents growth at constant exchange rates. £% represents growth at actual exchange rates. |
Table of Contents
50
| 2009 | 2008 | Growth | ||||||||||||||||||||||
| £m | % | £m | % | CER% | £% | |||||||||||||||||||
Turnover |
28,368 | 100 | 24,352 | 100 | 3 | 16 | ||||||||||||||||||
Cost of sales |
(7,380 | ) | (26.0 | ) | (6,415 | ) | (26.3 | ) | 6 | 15 | ||||||||||||||
Selling, general and administration |
(9,592 | ) | (33.8 | ) | (7,656 | ) | (31.4 | ) | 6 | 25 | ||||||||||||||
Research and development |
(4,106 | ) | (14.4 | ) | (3,681 | ) | (15.2 | ) | 1 | 12 | ||||||||||||||
Other operating income |
1,135 | 3.9 | 541 | 2.2 | ||||||||||||||||||||
Operating profit |
8,425 | 29.7 | 7,141 | 29.3 | 4 | 18 | ||||||||||||||||||
(2008 – £639 million).
Table of Contents
51
| Finance income | 2009 | 2008 | ||||||
| £m | £m | |||||||
Interest and other finance income |
67 | 321 | ||||||
Unwinding of discounts on assets |
2 | 1 | ||||||
Fair value adjustments and hedges |
1 | (9 | ) | |||||
| 70 | 313 | |||||||
Finance costs |
||||||||
Interest costs |
(770 | ) | (829 | ) | ||||
Unwinding of discounts on liabilities |
(11 | ) | (16 | ) | ||||
Fair value adjustments and hedges |
(2 | ) | 2 | |||||
| (783 | ) | (843 | ) | |||||
| 2009 | 2008 | Growth | ||||||||||||||||||||||
| £m | % | £m | % | CER% | £% | |||||||||||||||||||
Turnover |
28,368 | 100 | 24,352 | 100 | 3 | 16 | ||||||||||||||||||
Cost of sales |
(7,095 | ) | (25.0 | ) | (5,776 | ) | (23.7 | ) | 13 | 23 | ||||||||||||||
Selling, general and administration |
(9,200 | ) | (32.4 | ) | (7,352 | ) | (30.2 | ) | 6 | 25 | ||||||||||||||
Research and development |
(3,951 | ) | (13.9 | ) | (3,506 | ) | (14.4 | ) | 2 | 13 | ||||||||||||||
Other operating income |
1,135 | 3.9 | 541 | 2.2 | ||||||||||||||||||||
Operating profit |
9,257 | 32.6 | 8,259 | 33.9 | (1 | ) | 12 | |||||||||||||||||
Table of Contents
52
| Finance income | 2009 | 2008 | ||||||
| £m | £m | |||||||
Interest and other income |
67 | 321 | ||||||
Unwinding of discounts on assets |
2 | 1 | ||||||
Fair value adjustments and hedges |
1 | (9 | ) | |||||
| 70 | 313 | |||||||
Finance costs |
||||||||
Interest costs |
(770 | ) | (829 | ) | ||||
Unwinding of discounts on liabilities |
(8 | ) | (11 | ) | ||||
Fair value adjustments and hedges |
(2 | ) | 2 | |||||
| (780 | ) | (838 | ) | |||||
| 2009 | 2008 | |||||||
| £m | £m | |||||||
UK corporation tax |
417 | 289 | ||||||
Overseas taxation |
1,997 | 1,589 | ||||||
Current taxation |
2,414 | 1,878 | ||||||
Deferred taxation |
(192 | ) | 69 | |||||
Taxation on total profits |
2,222 | 1,947 | ||||||
| 2009 | 2008 | Growth | ||||||||||||||
| £m | £m | CER% | £% | |||||||||||||
Total profit after taxation for the year |
5,669 | 4,712 | 6 | 20 | ||||||||||||
Total profit attributable to shareholders |
5,531 | 4,602 | 6 | 20 | ||||||||||||
Basic earnings per share (pence) |
109.1 | p | 88.6 | p | ||||||||||||
Basic earnings per ADS (US$) |
$3.40 | $3.28 | ||||||||||||||
Results before major restructuring profit after taxation for the year |
6,283 | 5,551 | – | 13 | ||||||||||||
Results before major restructuring profit attributable to shareholders |
6,145 | 5,441 | – | 13 | ||||||||||||
Adjusted earnings per share (pence) |
121.2 | p | 104.7 | p | 2 | 16 | ||||||||||
Adjusted earnings per ADS (US$) |
$3.78 | $3.87 | ||||||||||||||
Weighted average number of shares (millions) |
5,069 | 5,195 | ||||||||||||||
Diluted total earnings per share (pence) |
108.2 | p | 88.1 | p | ||||||||||||
Diluted total earnings per ADS (US$) |
$3.38 | $3.26 | ||||||||||||||
Diluted weighted average number of shares (millions) |
5,108 | 5,226 | ||||||||||||||
Table of Contents
53
Table of Contents
54
Table of Contents
55
Table of Contents
56
The Group operates in a number of Middle Eastern and North African markets that subsequent to the year-end are experiencing political unrest. These events may lead to business disruption and liquidity problems that could adversely impact the Group’s results.
Table of Contents
57
Table of Contents
58

(Aged 62)
Chairman.

Chief Executive Officer.

(Aged 63)
Non-Executive Director.

(Aged 56)
Non-Executive Director.

Non-Executive Director.

Non-Executive Director.

Executive Director and Chief Financial Officer Designate.

Chief Financial Officer.
Table of Contents
59

(Aged 63)
Non-Executive Director.

Non-Executive Director.

(Aged 57)
Non-Executive Director.

Chairman, Research & Development.

Non-Executive Director.

(Aged 67)
Non-Executive Director & Senior Independent Director.
Table of Contents
60







Pharmaceuticals Europe



Table of Contents
61







Table of Contents
62

Table of Contents
63
Chairman
Table of Contents
64
| • | engaging at Board meetings with and challenging the CEO, the other Executive Directors and members of the CET as appropriate, on the financial and operating performance of GSK and external issues material to the Group’s prospects, | |
| • | evaluating progress towards the achievement of the Group’s financial and business objectives and annual plans and the Non- Executive Directors scrutinising the performance of management in meeting these objectives and plans, and | |
| • | monitoring, through reports received directly or from various committees, the significant risks facing the Group. |
| Number of meetings | ||||||||
| held whilst a Board | Number of | |||||||
| member | meetings attended | |||||||
Sir Christopher Gent |
6 | 6 | ||||||
Andrew Witty |
6 | 6 | ||||||
Julian Heslop |
6 | 6 | ||||||
Dr Moncef Slaoui |
6 | 6 | ||||||
Professor Sir Roy
Anderson |
6 | 6 | ||||||
Dr Stephanie Burns |
6 | 6 | ||||||
Larry Culp |
6 | 5 | ||||||
Sir Crispin Davis |
6 | 6 | ||||||
Sir Deryck Maughan* |
6 | 4 | ||||||
James Murdoch |
6 | 6 | ||||||
Dr Daniel Podolsky |
6 | 6 | ||||||
Tom de Swaan |
6 | 5 | ||||||
Sir Robert Wilson |
6 | 6 | ||||||
| * | Sir Deryck was unable to attend two meetings for personal reasons. He gave his comments to the Chairman on the matters to be discussed in advance of both meetings. |
Table of Contents
65
Table of Contents
66
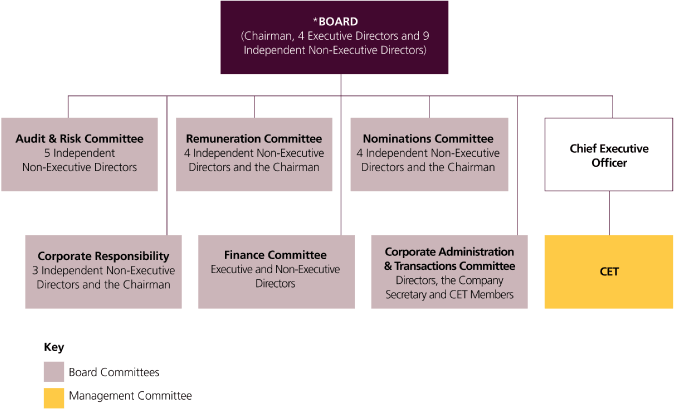
| * | There have been 4 Executive Directors since the appointment of the Chief Financial Officer
Designate on 4th January 2011 (there will be 3 Executive Directors following the retirement of the Chief Financial Officer on 31st March 2011). |
| Corporate | ||||||||||||||||
| Audit & Risk | Remuneration | Nominations | Responsibility | |||||||||||||
Sir Christopher Gent |
– | M | C | C | ||||||||||||
Professor Sir Roy Anderson |
M | – | – | – | ||||||||||||
Dr Stephanie Burns |
– | – | – | M | ||||||||||||
Larry Culp |
– | M | M | – | ||||||||||||
Sir Crispin Davis |
– | C | M | – | ||||||||||||
Sir Deryck Maughan |
M | – | M | – | ||||||||||||
James Murdoch |
– | M | – | M | ||||||||||||
Dr Daniel Podolsky |
M | – | – | M | ||||||||||||
Tom de Swaan |
C | M | – | – | ||||||||||||
Sir Robert Wilson |
M | – | M | – | ||||||||||||
Table of Contents
67
| No of meetings | Committee Report | |||||||||
| Committee | Role and Terms of Reference | Membership comprises | per year | on page | ||||||
Audit & Risk
|
Reviews the financial and internal reporting process, the external and internal audit processes, the system of internal controls, and the identification and management of risks. The Committee also proposes to shareholders the appointment, re-appointment and removal of the external auditors and is directly responsible for their remuneration and oversight of their work. | Independent Non-Executive Directors |
≥ 4 | 74-76 | ||||||
Remuneration
|
Determines the terms of service and remuneration of the Executive Directors and members of the CET and, with the assistance of external independent advisers, it evaluates and makes recommendations to the Board on overall executive remuneration policy that assists the long-term success of the Group. | Independent Non-Executive Directors and the Chairman | ≥ 4 | 81-101 | ||||||
| (The Chairman and the CEO are responsible for evaluating and making recommendations to the Board on the remuneration of Non-Executive Directors.) | ||||||||||
Nominations
|
Reviews the structure, size and composition of the Board (including the skills, knowledge, independence, experience and diversity) and appointment of members to the Board and the CET, and makes recommendations to the Board as appropriate. The Committee monitors the planning of succession to the Board and Senior Management. The Committee also considers and if appropriate authorises directors’ conflicts of interest. | Independent Non-Executive Directors and the Chairman | ≥ 2 | 77 | ||||||
Corporate Responsibility |
Provides a Board-level forum for the regular review of external issues that have the potential for serious impact upon the Group’s business and reputation. The Committee is also responsible for oversight of GSK’s worldwide donations and community support. | Independent Non-Executive Directors and the Chairman | ≥ 3 | 78-80 | ||||||
Finance
|
Reviews and approves, on behalf of the Board, the Annual Report and Form 20-F, and convening of the AGM, together with the preliminary and quarterly statements of trading results. It also approves certain major licensing and capital transactions and changes to the Group’s Investment Instrument and Counterparty Limits. | Executive and Non-Executive Directors | As necessary | – | ||||||
Corporate Administration & Transactions |
Reviews and approves matters in connection with the administration of the Group’s business and certain corporate transactions. | Executive and Non-Executive Directors, CET members and the Company Secretary | As necessary | – | ||||||
Table of Contents
68
| Method of evaluation | Actions and areas of focus | |||||
2008
|
Dr Long, Boardroom Review, facilitated this review |
• | Utilise Board and Committee time more effectively and facilitate further contribution by Non-Executive Directors | |||
| • | Enhance continuous education
process for Non-Executive Directors |
|||||
| • | Provide greater visibility to executive talent and management succession planning process | |||||
2009
|
Sir Robert Wilson, Senior Independent Director |
• | Increase Board time devoted to strategic discussion and the indicators of success in the delivery of the R&D pipeline | |||
| • | Devote more time to focused consideration of the company’s key risks on an ongoing basis | |||||
| • | Provide the Board with more regular updates and insights into the newly enhanced management succession planning process | |||||
| • | had increased its focus on R&D activities and successful delivery of the pipeline and was pleased with progress from R&D during the year. Separately, on behalf of the Board, the Remuneration Committee initiated discussions with management and shareholders over the introduction of more strategically aligned performance criteria for the company’s long term incentive plans. As a result of a successful conclusion to these discussions, the grant of LTI options made in February 2011 was made with two additional performance criteria, one of which focuses on R&D new product performance. For more details, please refer to the Remuneration Report on pages 81 to 101 |
| • | had sought assistance from the Audit & Risk Committee to more fully understand the Group’s key risks. This work is ongoing and the Board will continue to consider regular reports from the Audit & Risk Committee during 2011, in advance of the Board’s annual review of the effectiveness of the company’s risk management next year |
| • | was pleased with the operation by the Nominations Committee of the enhanced succession planning process. This had resulted in the appointment of the CFO Designate and positive progress was being made on the recruitment of new Board members to refresh the Board. |
| • | given the fundamental strategic challenges facing the pharmaceutical industry, the Board will seek to continue to allocate more time on a regular basis to focus on strategic issues and the significant challenges facing the industry, with the direct aim of further enhancing returns to shareholders |
| • | to further enhance information flows by providing Board members with a wider variety of external perspectives on the company and the industry |
| • | R&D will continue to be a major expense to the company and the Board will be seeking to assess the extent to which the new policies implemented in recent years have added value |
| • | to continue to support Executive Management on ethical leadership within the Group. |
Table of Contents
69
| No. of | Percentage of issued | |||||||
| shares | capital (%)* | |||||||
BlackRock, Inc. |
291,516,314 | 5.62 | ||||||
Legal & General Group Plc |
194,024,944 | 3.74 | ||||||
| * | Percentage of Ordinary Shares in issue, excluding Treasury shares. |
Table of Contents
70
| • | he/she resigns | |
| • | he/she offers to resign and the Board accept that offer | |
| • | all other Directors (being at least three in number) require him/ her to resign | |
| • | he/she is suffering from physical or mental ill health | |
| • | he/she has missed Directors’ meetings for a continuous period of six months without permission and the Board resolves that he/she shall cease to be a Director | |
| • | he/she becomes bankrupt or compounds with his/her creditors generally | |
| • | he/she ceases to be a Director by virtue of the Articles or the Companies Acts, or | |
| • | he/she is prohibited from being a Director by law. |
| Political donations to: | 2010 | 2009 | 2008 | |||||||||
| £ | £ | £ | ||||||||||
EU political organisations |
– | – | – | |||||||||
Non-EU political organisations comprising: |
||||||||||||
USA |
– | – | 319,000 | |||||||||
Canada |
– | – | 28,000 | |||||||||
| – | – | 347,000 | ||||||||||
Table of Contents
71
| • | Receiving and adopting GlaxoSmithKline’s 2010 Annual Report |
| • | Approving the 2010 Remuneration Report The Remuneration Report on pages 81 to 101 sets out the remuneration policies operated by GSK and disclosures on Directors’ remuneration, including those required by the Companies Act 2006 and The Large and Medium-sized Companies and Groups (Accounts and Reports) Regulations 2008. A resolution will be proposed to approve the Remuneration Report. |
| • | Retirement and re-election of Directors Simon Dingemans, who was appointed before the AGM will retire in accordance with the Articles and, being eligible, will offer himself for election at the AGM. All the continuing Directors will retire by rotation at the 2011 AGM and offer themselves for re-election. |
| • | Re-appointment and remuneration of auditors Resolutions will be proposed to authorise the Audit & Risk Committee to re-appoint PricewaterhouseCoopers LLP as auditors and to determine their remuneration. |
| • | Special business The company will seek authority to: |
| • | make donations to EU political organisations and incur EU political expenditure, capped at £100,000 | ||
| • | allot Ordinary Shares in the company | ||
| • | give the Directors authority to disapply pre-emption rights when allotting new shares in connection with rights issues or otherwise up to a maximum of 5% of the current issued share capital and to purchase its own Ordinary Shares up to a maximum of just under 10% of the current issued share capital | ||
| • | exempt the auditors from having to state the name of their senior statutory auditor for the company in GSK’s Annual Report | ||
| • | reduce the notice required to call a general meeting to not less than 14 clear days. |
Table of Contents
72
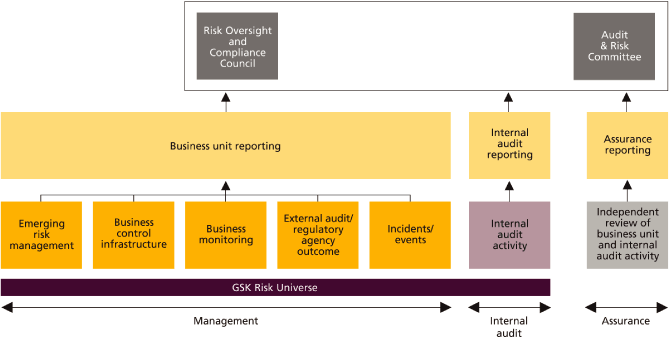
Table of Contents
73
| • | Commercial and Financial |
| • | Information Technology |
| • | Manufacturing (including Environmental Health, Safety and Sustainability) |
| • | Research and Development |
Table of Contents
74

Dear Shareholder
Audit & Risk Committee Chairman
| Attendance at | ||||||||
| full meetings | ||||||||
| Members | Committee member since | during 2010 | ||||||
Mr Tom de Swaan |
1st January 2006 | 6/6 | ||||||
(Chairman from |
||||||||
1st September 2006) |
||||||||
Professor Sir Roy |
20th May 2009 | 6/6 | ||||||
Anderson |
||||||||
Sir Deryck Maughan |
21st January 2005 | 5/6 | ||||||
Dr Daniel Podolsky |
1st January 2007 | 6/6 | ||||||
Sir Robert Wilson |
12th December 2003 | 6/6 | ||||||
| • | CEO | |
| • | CFO | |
| • | Chairman | |
| • | General Counsel | |
| • | Head of Audit & Assurance | |
| • | Company Secretary | |
| • | Chairman, Research & Development | |
| • | Chief Medical Officer | |
| • | Head of Governance, Ethics and Assurance | |
| • | External Auditors. |
| • | Reviewing the corporate accounting and financial reporting process | |
| • | Monitoring the integrity of the financial statements | |
| • | Evaluating the system of internal control and identifying and managing risks, including in relation to the financial reporting process and the preparation of consolidated accounts | |
| • | Overseeing activities of each of the Group’s compliance and audit functions and overseeing compliance with laws, regulations and ethical codes of practice. |
Table of Contents
75
| Financial & accounting experience | ||
Mr Tom de Swaan
|
• Chief Financial Officer of ABN AMRO until
31st December 2005 |
|
• Determined by the Board to be the Audit
Committee Financial Expert, as defined by the
Sarbanes Oxley Act of 2002 (Sarbanes-Oxley) |
||
• Non-Executive Director of KPMG’s Public
Interest Committee |
||
Sir Deryck Maughan
|
• A Partner of Kohlberg Kravis Roberts & Co.
(KKR) and Chairman of KKR Japan |
|
• Former Chairman & CEO of Citigroup
International and Vice Chairman of
Citigroup Inc. |
||
• Former Chairman and Co-Chief Executive
Officer of Salomon Smith Barney |
||
• Former Chairman and Chief Executive
Officer of Salomon Brothers Inc. |
||
Sir Robert Wilson
|
• Economist, and former Non-Executive
Chairman of The Economist Group |
|
• Chairman of BG Group plc |
||
• Retired from RioTinto in 2003 where he held
Senior Management positions culminating in
his appointment as Executive Chairman |
||
| Scientific expertise | ||
Professor Sir Roy Anderson |
• A world renowned medical scientist with
advanced knowledge of infectious disease
epidemiology |
|
• Professor of Infectious Disease Epidemiology
in the Faculty of Medicine, Imperial College,
London |
||
• Fellow of the Royal Society |
||
• Foreign Associate Member of the Institute
of Medicine at the US National Academy
of Sciences |
||
• Foreign Associate Member of the French
Academy of Sciences |
||
• Former Rector of Imperial College, London |
||
• Former Chief Scientific Adviser at the Ministry
of Defence in the UK |
||
Dr Daniel Podolsky
|
• A world renowned researcher with
advanced knowledge of underlying
mechanisms of disease and new therapies
for gastrointestinal disorders |
|
• President of the University of Texas
Southwestern Medical Center and Professor
of Internal Medicine |
||
• Member, Institute of Medicine of the US
National Academy of Sciences |
||
• Former Mallinckrodt Professor of Medicine,
Harvard Medical School |
||
• Former Chief Academic Officer, Partners
Healthcare |
||
Table of Contents
76
External auditors
|
reported on all critical accounting policies, significant judgements and practices used by the Group, alternative accounting treatments which had been discussed with management and their resultant conclusion, material written communications with management and any restrictions on access to information | |
CFO
|
reported on the financial performance of the company and on technical financial and accounting matters | |
General Counsel
|
reported on material litigation | |
Company Secretary & Corporate Compliance Officer
|
reported on corporate governance and on the activities undertaken by the ROCC | |
Heads of audit and
assurance and the
Group’s compliance
and audit groups
|
the majority of the Heads of these groups reported on their audit scope, annual coverage, audit resources and on the results of audits conducted throughout the year | |
Company Secretary,
as Chairman of
the Disclosure
Committee
|
reported on matters that affected the quality and timely disclosure of financial and other material information to the Board, to the public markets and to shareholders. This enabled the Audit & Risk Committee to review the clarity and completeness of the disclosures in the published annual financial statements, interim reports, quarterly and preliminary results announcements and other formal announcements relating to financial performance prior to approval by the Board. | |
Table of Contents
77

Sir Christopher Gent
Nominations Committee Chairman
| Attendance at | ||||||||
| full meetings | ||||||||
| Members | Committee member since | during 2010 | ||||||
Sir Christopher Gent |
9th December 2004 | 3/3 | ||||||
(Chairman from |
||||||||
1st January 2005) |
||||||||
Larry Culp |
28th March 2008 | 2/3 | ||||||
Sir Crispin Davis |
9th July 2009 | 3/3 | ||||||
Sir Deryck Maughan |
9th July 2009 | 2/3 | ||||||
Sir Robert Wilson |
28th March 2008 | 3/3 | ||||||
| • | CEO | |
| • | Head of Human Resources | |
| • | Company Secretary | |
| • | where relevant, appropriate external advisers. |
Table of Contents
78

Sir Christopher Gent
Corporate Responsibility Committee Chairman
| Attendance at | ||||||||
| full meetings | ||||||||
| Members | Committee member since | during 2010 | ||||||
Sir Christopher Gent (Chairman from 1st January 2005) |
9th December 2004 | 4/4 | ||||||
Dr Stephanie Burns |
6th December 2007 | 3/4 | ||||||
James Murdoch |
20th May 2009 | 4/4 | ||||||
Dr Daniel Podolsky |
1st July 2006 | 4/4 | ||||||
| • | CEO | |
| • | General Counsel | |
| • | Head of Governance, Ethics and Assurance | |
| • | Head of Global Communications | |
| • | Head of Corporate Responsibility | |
| • | Company Secretary | |
| • | Independent External Corporate Responsibility Adviser. |
| • | access to medicines | |
| • | standards of ethical conduct | |
| • | research and innovation | |
| • | employment practices; and | |
| • | community investment |
| CR Principles | Committee’s area of focus during 2010 | |
Access to medicines
|
Access to and pricing of medicines in middle income and least developed countries. | |
Standards of ethical conduct
|
Embedding ethical values in the organisation. | |
Research and innovation
|
Policy on use of animals in research and development. Research integrity and transparency. Governance of research conducted by external suppliers and collaborators. R&D on treatments for rare conditions and for diseases of the developing world. The potential of stem cell science for regenerative medicines. | |
Employment practices
|
Diversity and inclusion. Leading and developing employees. Employee relations including consultation arrangements. Realignment of the pay for performance strategy. Management of health and safety risks in manufacturing. | |
Community investment
|
Community partnerships and investment. Humanitarian donations. | |
Caring for the environment
|
Environmental sustainability strategy. Management of environment risks in manufacturing. | |
Products and customers
|
Disclosure of payments to healthcare professionals. | |
Table of Contents
79
| • | they have each reviewed the Annual Report and Form 20-F | |
| • | based on their knowledge, it contains no material misstatements or omissions | |
| • | based on their knowledge, the financial statements and other financial information fairly present, in all material respects, the financial condition, results of operations and cash flows as of the dates, and for the periods, presented in the Annual Report and Form 20-F | |
| • | they are responsible for establishing and maintaining disclosure controls and procedures that ensure that material information is made known to them, and have evaluated the effectiveness of these controls and procedures as at the year-end, the results of such evaluation being contained in the Annual Report and Form 20-F | |
| • | they are responsible for establishing and maintaining internal control over financial reporting that provides reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles | |
| • | they have disclosed in the Annual Report and Form 20-F any changes in internal controls over financial reporting during the period covered by the Annual Report and Form 20-F that have materially affected, or are reasonably likely to affect materially, the company’s internal control over financial reporting | |
| • | they have disclosed, based on their most recent evaluation of internal control over financial reporting, to the external auditors and the Audit & Risk Committee, all significant deficiencies and material weaknesses in the design or operation of internal controls over financial reporting which are reasonably likely to affect adversely the company’s ability to record, process, summarise and report financial information, and any fraud (regardless of materiality) involving persons that have a significant role in the company’s internal control over financial reporting. |
Table of Contents
80
| • | Management is responsible for establishing and maintaining adequate internal control over financial reporting for the Group. Internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with IFRS | |
| • | Management conducted an evaluation of the effectiveness of internal control over financial reporting based on the framework in Internal Control — Integrated Framework issued by the Committee of Sponsoring Organisations of the Treadway Commission | |
| • | Management has assessed the effectiveness of internal control over financial reporting, as at 31st December 2010 and has concluded that such internal control over financial reporting was effective. In addition, there have been no changes in the Group’s internal control over financial reporting during 2010 that have materially affected, or are reasonably likely to affect materially, the Group’s internal control over financial reporting | |
| • | PricewaterhouseCoopers LLP, which has audited the consolidated financial statements of the Group for the year ended 31st December 2010, has also assessed the effectiveness of the Group’s internal control over financial reporting under Auditing Standard No. 5 of the Public Company Accounting Oversight Board (United States). Their audit report may be found on page 103. |
Table of Contents
81

Sir Crispin Davis
Remuneration Committee Chairman
| Key Strategic Priorities | Long-Term Incentive Performance Measure | |||
• Grow a diversified global business
|
→ | • Business diversification performance | ||
• Deliver more products of value
|
→ | • R&D new product performance | ||
• Simplify the operating model
|
→ | • Adjusted free cash flow | ||
• Deliver value to shareholders
|
→ | • Relative total shareholder return (TSR) |
Remuneration Committee Chairman
1st March 2011
Table of Contents
82
| Attendance | ||||||||
| at full | ||||||||
| meetings | ||||||||
| Committee member | during | |||||||
| Members | since | 2010 | ||||||
Sir Crispin Davis |
1st July 2003 | 6/6 | ||||||
(Chairman from 20th May 2009) |
||||||||
Sir Robert Wilson |
1st January 2004 | 3/3 | ||||||
(to 25th March 2010) |
||||||||
Larry Culp* |
1st January 2004 | 4/6 | ||||||
Sir Christopher Gent |
1st January 2007 | 6/6 | ||||||
James Murdoch |
1st October 2009 | 6/6 | ||||||
Tom de Swaan** |
20th May 2009 | 6/6 | ||||||
| * | Larry Culp was unable to attend two meetings for personal reasons. He reviewed the papers and provided his views to the Committee Chairman in advance of these meetings. | |
| ** | Tom de Swaan is also the Chairman of the Audit & Risk Committee. |
Table of Contents
83
Month |
Executives’ Remuneration |
Annual bonus |
Long-term incentive plans |
Governance and other matters |
||||||||
January
|
Approve CET 2010 remuneration, including salaries of CEO, CFO and Chairman, R&D | Set CEO 2010 bonus objectives Review bonus plan arrangements for Chairman, R&D |
Annual Committee evaluation results Review remuneration report Review 2010 remuneration budget Review voting policy guidelines on remuneration |
|||||||||
February
|
Review and approve Executive Directors’ and CET 2009 bonuses |
Set 2010 PSP cash flow target Approve 2007 PSP and Share Option Plan vesting Grant 2010 LTI awards to Executive Directors, CET and below Approve deferred annual bonus award elections |
Approve remuneration report |
|||||||||
March
|
Scope review of strategic alignment of LTI performance measures | Review voting policy guidelines on remuneration |
||||||||||
July
|
Agree CFO retirement and appointment packages | Review progress against 2010 bonus objectives |
Review of strategic alignment
of LTI performance measures
Grant interim 2010 LTI awards |
Review of general market developments (including pensions) | ||||||||
October
|
Agree CET 2011 salary review process |
Review CET 2010 bonus process |
Review of strategic alignment of LTI performance measures | Annual Committee assessment |
||||||||
November
|
Annual meetings with investors |
Annual meetings with investors | ||||||||||
December
|
Consider feedback from annual meetings with investors on remuneration policy Review remuneration benchmarking and competitiveness below CET level |
Consider new LTI performance targets framework |
Review tax change implications for pension arrangements |
Table of Contents
84
Salary
|
•
Salary levels reviewed annually and influenced by Executive’s role
and experience. Benchmarked against relevant comparator group(s) |
|
Pension
|
•
For UK Executives, defined contribution plan and legacy final salary plans
(closed to new entrants since 2001). UK Executives participating in defined contribution plan benefit from company contributions of 20% of base salary, plus matched contribution of 5% of base
salary |
|
•
Following changes to UK pension tax regime, changes
made to arrangements for UK Executives to continue to meet long-standing commitments within this new regime – see page 89 |
||
•
For US Executives, GSK operates a US Cash Balance
Plan (US Plan). US Executives participating in US Plan benefit from contributions of up to 38% of salary |
||
|
Annual bonus |
• Maximum bonus opportunity of 200% of salary |
• Majority of bonus based on achievement of financial targets (Group profit before
interest and tax and business unit operating profit) |
||||||||
|
•
Individual performance against pre-determined personal
objectives
|
||||||||||
|
•
R&D-specific key performance indicators for R&D
employees
|
||||||||||
|
•
‘Clawback’ – Committee reviews ongoing financial
impact of any prior year activities and Executive’s role in them
and may make appropriate adjustments to individual bonus awards to
reflect circumstances.
|
||||||||||
|
Deferred Annual Bonus Plan |
• Individuals may elect to defer up to 50% of any bonus earned |
•
Awards vest at end of three-year performance period based on four equally weighted performance measures:
|
||||||||
|
- Business diversification performance - R&D new product performance - Adjusted free cash flow - Relative TSR |
||||||||||
|
•
Deferred bonuses may be matched up to one-for-one subject to performance |
||||||||||
|
Performance Share Plan
(PSP)
|
For 2011, performance share awards are as follows: |
•
Relative TSR is calculated on twelve month averaging period, using
comparator group comprising 10 pharmaceutical companies. 30% vests at
median, with 100% vesting for upper quartile performance.
•
For business diversification, R&D new product measures and
adjusted free cash flow, 25% vests at threshold, rising to 100%
for stretching performance exceeding set threshold by a specified margin
|
||||||||
|
% of Salary |
||||||||||
• CEO
|
500 | |||||||||
|
• Chairman, R&D
|
500 | |||||||||
• CFO Designate
|
350 | |||||||||
Table of Contents
85
| UK cross-industry comparator group | Global pharmaceutical comparator group | ||||
AngloAmerican
|
France | Sanofi-Aventis | |||
AstraZeneca
|
Switzerland | Novartis | |||
Barclays
|
Roche Holdings | ||||
BG Group
|
UK | AstraZeneca | |||
BHP Billiton
|
USA | Abbott Laboratories | |||
BP
|
Amgen* | ||||
British American Tobacco
|
Bristol-Myers Squibb | ||||
Diageo
|
Eli Lilly | ||||
HSBC
|
Johnson & Johnson | ||||
Reckitt Benckiser
|
Merck | ||||
Royal Dutch Shell
|
Pfizer | ||||
Rio Tinto |
|||||
Standard Chartered |
|||||
Tesco |
|||||
Unilever |
|||||
Vodafone |
|||||
| * | Amgen is included for benchmarking but since 2009 has not been in the current TSR comparator group. |
| UK | Global | |||||||
| Primary Comparator Group | cross-industry | pharmaceutical | ||||||
Andrew Witty, CEO |
ü | |||||||
Julian Heslop, CFO* |
ü | |||||||
Simon Dingemans, CFO Designate** |
ü | |||||||
Dr Moncef Slaoui, Chairman, R&D |
ü | |||||||
| * | Julian Heslop will retire from the Board on 31st March 2011 | |
| ** | Simon Dingemans joined the Board on 4th January 2011 |
| 2010 base | Effective date for | 2011 base | Effective date for | % | ||||||||||||||||
| salary | 2010 salary | salary | 2011 salary | change | ||||||||||||||||
Andrew Witty, CEO |
£1,000,000 | 1st April 2010 | £1,000,000 | 1st April 2011 | 0 | |||||||||||||||
Julian Heslop, |
£525,000 | 1st April 2010 | – | – | – | |||||||||||||||
CFO* |
||||||||||||||||||||
Simon Dingemans, |
– | – | £660,000 | 4th January 2011 | – | |||||||||||||||
CFO Designate** |
||||||||||||||||||||
Dr Moncef Slaoui, |
$975,000 | 1st April 2010 | $1,125,000 | 1st April 2011 | 15 | |||||||||||||||
Chairman, R&D |
||||||||||||||||||||
| * | Julian Heslop will retire from the Board on 31st March 2011 | |
| ** | Simon Dingemans joined the Board on 4th January 2011 |
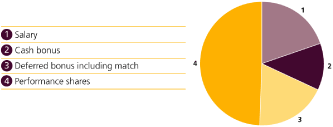
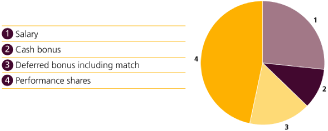
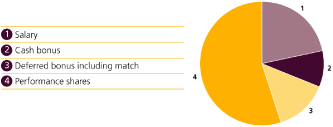
Table of Contents
86
| On-target bonus as a % of base salary | ||||
Andrew Witty, CEO |
125% | |||
Simon Dingemans, CFO Designate |
80% | |||
Dr Moncef Slaoui, Chairman, R&D |
85% | |||
Table of Contents
87
| 2011 | 2011 award level | |||||||
| award | % of base salary | |||||||
Andrew Witty, CEO |
424,448 shares | 500% | ||||||
Simon Dingemans, |
196,095 shares | 350% | ||||||
CFO Designate |
||||||||
Dr Moncef Slaoui, |
147,521 ADS | 500% | ||||||
Chairman, R&D |
||||||||
| Strategy | Remuneration | |||||||
Key strategic priorities:
|
LTI performance
measures
(over 3-year performance period) |
% of award |
||||||
Grow a diversified global business
|
|
Business
diversification
|
25 | % | ||||
|
Deliver more products of value
|
|
R&D
new product
|
25 | % | ||||
Simplify the operating model
|
|
Adjusted free cash
flow
|
25 | % | ||||
Deliver value to shareholders |
|
Relative TSR
|
25 | % | ||||
Table of Contents
88
Long- term incentive measures for 2011 awards
|
% of award |
Vesting schedule for 2011 awards | |
|||||||||||
| Business diversification performance | 25 | % | Due to commercial sensitivity, targets for business diversification and R&D new product measures will be disclosed along with outcomes in the 2013 Remuneration Report. | |||||||||||
| Incentivises growth of a global, diversified business | ||||||||||||||
| |
||||||||||||||
Designed to focus on our major growth areas: Vaccines,
Consumer Healthcare, Emerging Markets and Japan
(excluding Vaccines) and Dermatology businesses.
|
Proportion of threshold achieved |
Proportion of award available |
||||||||||||
| Below threshold | 0 | % | ||||||||||||
Aggregate revenue target for four business divisions over three-year performance period reflects strong growth against
previous periods and above market growth.
|
Threshold Maximum |
25 100 |
% % |
|||||||||||
|
|
||||||||||||||
R&D new product performance
|
25 | % |
The target for maximum performance (expressed as a percentage of the
threshold) for these two measures is shown below: |
|||||||||||
Recognises importance of R&D to future business growth
|
Measure |
Maximum expressed as % of threshold |
||||||||||||
Revenue target based on New Product Sales to incentivise better R&D performance.
|
Business | 114 | % | |||||||||||
| diversification | ||||||||||||||
New Products defined as products launched in performance period and two preceding years. Therefore, for 2011-13 performance period, products launched in years 2009-13 will be included in measurement. |
R&D new product |
122 |
% |
|||||||||||
Aggregate three-year revenue target for 2011 awards for New Product Sales reflects growth on historic performance. |
||||||||||||||
Adjusted free cash flow
|
25 | % |
Three year adjusted free cash flow targets |
% vesting |
||||||||||
Recognises importance of effective working capital and cash management
|
Below threshold Threshold |
£16.15 billion | 0 25 |
% % |
||||||||||
| £16.65 billion | 50 | % | ||||||||||||
| £18.32 billion | 75 | % | ||||||||||||
| Maximum | £19.15 billion | 100 | % | |||||||||||
|
|
||||||||||||||
Relative TSR
|
25 | % | ||||||||||||
Focuses on delivery of value to shareholders
Relative TSR using a comparator group comprising 10 global pharmaceutical companies. With move to four complementary measures, relative TSR now measured over three years in line with performance period for all other performance measures. To measure performance on a stable basis and better reflect long-term nature of pharmaceutical industry, twelve-month averaging period is used for relative TSR. |
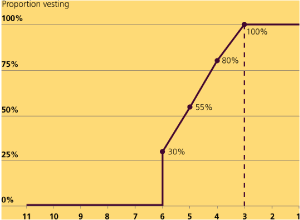 |
|
||||||||||||
|
TSR rank position |
Median performance |
Upper quartile performance |
||||||||||||
Table of Contents
89
Table of Contents
90
| Share Ownership Requirement | ||||
CEO |
4 x base salary | |||
Other Executive Directors |
3 x base salary | |||
CET members |
2 x base salary | |||
| Holding for | Holding for | |||||||||||
| Ordinary shares | Ordinary shares | |||||||||||
| for SOR purposes | for SOR purposes | % increase in | ||||||||||
| as at 31/12/09 | as at 25/02/11 | * | shareholding | |||||||||
Andrew Witty |
91,472 | 226,199 | 147 | |||||||||
Julian Heslop |
49,350 | 92,182 | 87 | |||||||||
Simon Dingemans** |
– | 40,000 | – | |||||||||
Dr Moncef Slaoui |
66,938 | 169,906 | 154 | |||||||||
| * | Shares to be sold for tax following the vesting of the 2008 PSP awards have been excluded. | |
| ** | The disclosure for Simon Dingemans is from the date he joined the Board on 4th January 2011. |
31st December 2010
achieved under the company’s annual bonus and long-term
incentive plans for performance periods that ended on
31st December 2010.
| • | delivering underlying sales growth (excluding pandemic products, Avandia and Valtrex) | |
| • | strong sales performance in investment areas of the business, particularly Emerging Markets and Consumer Healthcare | |
| • | increasing R&D pipeline potential and achieving key milestones in the transformation of R&D productivity, particularly in relation to the late stage R&D pipeline products | |
| • | simplification of GSK’s business model, improved cash generation, before legal settlements, and achievement of operational efficiencies. |
| Annual bonus | Annual bonus | |||||||||||
| 2010 000 |
2009 000 |
% change | ||||||||||
Andrew Witty |
£1,177 | £2,000 | (41 | ) | ||||||||
Julian Heslop |
£417 | £602 | (31 | ) | ||||||||
Dr Moncef Slaoui |
$1,434 | $1,439 | – | |||||||||
| Executive | % of total bonus | Deferred Award | Matching Award | |||||||||
Andrew Witty |
32% | 31,921 shares | 31,921 shares | |||||||||
Dr Moncef Slaoui |
50% | 18,756 ADS | 18,756 ADS | |||||||||
Table of Contents
91
| Performance Share | Share Option | |||||||||||
| Plan | Plan | |||||||||||
| Vesting | Vesting | |||||||||||
| under TSR | under EPS | |||||||||||
| Performance period | measure % | measure % | ||||||||||
2003 |
2004 - 2006 | 0 | 100 | |||||||||
2004 |
2005 - 2007 | 38.47 | 100 | |||||||||
2006 |
2006 - 2008 | 0 | 50.7 | |||||||||
2007 |
2007 - 2009 | 35 | 0 | |||||||||
2008 |
2008 - 2010 | 35 | 0 | |||||||||
| Average annual vesting | 21.69 | 50.14 | ||||||||||
Notice period on termination by employing company or executive |
12 calendar months | |
Termination payment
|
1 x annual salary payable on termination |
|
Vesting of LTIs
|
Rules of relevant incentive plan, as approved by shareholders | |
Pension
|
Based on existing arrangements and terms of relevant pension plan |
|
Non-compete clause
|
12 months from termination notice date* | |
| * | The ability to impose a 12-month non-compete period (and a non-solicitation restriction) on an Executive is considered important by the company in order to have the ability to protect the Group’s intellectual property and staff. In light of this, the Committee believes that it would not be appropriate to provide for mitigation in the contracts. |
Table of Contents
92
| Current Directors | Date of contract | Effective date | Expiry date | |||
Andrew Witty*
|
18th June 2008 | 22nd May 2008 | 31st August 2024 | |||
Julian Heslop
|
16th March 2006 | 1st April 2005 | 31st January 2014 | |||
Simon Dingemans
|
8th September 2010 | 4th January 2011 | 30th April 2028 | |||
Dr Moncef Slaoui**
|
21st December 2010 | 21st December 2010 | 1st August 2019 | |||
| * | Andrew Witty’s contract was renewed in June 2008 following his appointment as CEO, and was amended on 4th February 2010 to reflect the changes to his severance terms outlined above. | |
| ** | Dr Moncef Slaoui’s previous contract dated 16th May 2008 was replaced on 21st December 2010 to reflect the changes to his severance terms outlined above. |
| • | in the case of outstanding awards under the GlaxoSmithKline Annual Investment Plan (which was closed to new deferrals with effect from the first quarter of 2006) provided that their agreement is terminated other than for cause, the executive must exercise any Bonus Investment Rights within six months of termination to receive any deferred amounts, and any income and gains; and | |
| • | in line with the policy applicable to US senior executives, Dr Moncef Slaoui may become eligible, at a future date, to receive continuing medical and dental insurance after retirement. |
| Non-Executive Director | Date of letter of appointment | |||
Sir Christopher Gent |
26th May 2004 | |||
Professor Sir Roy Anderson |
28th September 2007 | |||
Dr Stephanie Burns |
12th February 2007 | |||
Larry Culp |
9th June 2003 | |||
Sir Crispin Davis |
9th June 2003 | |||
Sir Deryck Maughan |
26th May 2004 | |||
James Murdoch |
26th February 2009 | |||
Dr Daniel Podolsky |
3rd July 2008 | |||
Tom de Swaan |
21st December 2005 | |||
Sir Robert Wilson |
9th June 2003 | |||
| Per annum | ||||
Standard annual cash retainer fee |
£75,000 | |||
Supplemental
fees |
||||
Chairman of Audit & Risk Committee |
£80,000 | |||
Senior Independent Director and Scientific/Medical Experts |
£30,000 | |||
Chairmen of the Remuneration and Corporate Responsibility Committees* |
£20,000 | |||
Non-Executive Director undertaking intercontinental travel to meetings |
£7,500 per meeting |
|||
| * | Sir Christopher Gent is the current Chairman of the Corporate Responsibility Committee, but does not receive the additional fee listed above. |
Table of Contents
93
| Exchange rate | ||||||||
| Date of approval | Period rate applied | £1/US$ | ||||||
29th July 2004 |
01.10.04 – 31.03.08 | US$1.8162 | ||||||
28th March 2008 |
01.04.08 – 30.09.09 | US$1.9918 | ||||||
3rd December 2009* |
01.10.09 – 31.12.09 | US$1.6395 | ||||||
| 01.01.10 – 31.12.10 | US$1.6326 | |||||||
| 01.01.11 – 31.12.11 | US$1.5798 | |||||||
| * | Given fluctuations in the Sterling: US dollar exchange rate, it was agreed that with effect from 1st October 2009 the exchange rate would be set annually based on the average daily rate for the last quarter of the year prior to payment. The rate would be reviewed if exchange rates moved significantly during the year. |
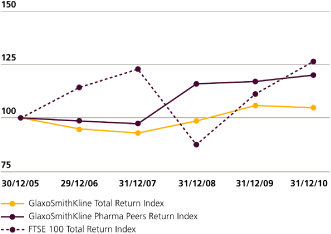
Table of Contents
94
| 2010 | 2009 | |||||||||||||||||||||||||||||||||||
| Total | Total | |||||||||||||||||||||||||||||||||||
| Fees and | Other | Annual | annual | Fees and | Other | Annual | annual | |||||||||||||||||||||||||||||
| salary | benefits | bonus | remuneration | salary | benefits | bonus | remuneration | |||||||||||||||||||||||||||||
| Footnote | 000 | 000 | 000 | 000 | 000 | 000 | 000 | 000 | ||||||||||||||||||||||||||||
Executive Directors |
||||||||||||||||||||||||||||||||||||
Andrew Witty |
a,b,c,e | £1,000 | £126 | £1,177 | £2,303 | £948 | £89 | £2,000 | £3,037 | |||||||||||||||||||||||||||
Julian Heslop |
a,b | £525 | £108 | £417 | £1,050 | £507 | £56 | £602 | £1,165 | |||||||||||||||||||||||||||
Dr Moncef Slaoui |
c,d,e | $953 | $405 | $1,434 | $2,792 | $865 | $355 | $1,439 | $2,659 | |||||||||||||||||||||||||||
Non-Executive Directors |
||||||||||||||||||||||||||||||||||||
Professor Sir Roy Anderson |
£128 | – | – | £128 | £120 | – | – | £120 | ||||||||||||||||||||||||||||
Sir Crispin Davis |
£118 | – | – | £118 | £102 | – | – | £102 | ||||||||||||||||||||||||||||
Sir Christopher Gent |
£675 | £2 | – | £677 | £675 | £5 | – | £680 | ||||||||||||||||||||||||||||
James Murdoch |
f | £98 | – | – | £98 | £54 | – | – | £54 | |||||||||||||||||||||||||||
Tom de Swaan |
£177 | £1 | – | £178 | £133 | – | – | £133 | ||||||||||||||||||||||||||||
Sir Robert Wilson |
£128 | – | – | £128 | £116 | – | – | £116 | ||||||||||||||||||||||||||||
Dr Stephanie Burns |
$146 | – | – | $146 | $188 | – | – | $188 | ||||||||||||||||||||||||||||
Larry Culp |
$135 | – | – | $135 | $188 | – | – | $188 | ||||||||||||||||||||||||||||
Sir Deryck Maughan |
$147 | – | – | $147 | $188 | – | – | $188 | ||||||||||||||||||||||||||||
Dr Daniel Podolsky |
$208 | – | – | $208 | $245 | – | – | $245 | ||||||||||||||||||||||||||||
Former Directors |
||||||||||||||||||||||||||||||||||||
Dr Michèle Barzach |
g | – | – | – | – | £80 | – | – | £80 | |||||||||||||||||||||||||||
Sir Ian Prosser |
h | – | – | – | – | £48 | £5 | – | £53 | |||||||||||||||||||||||||||
Dr Ronaldo Schmitz |
h | – | – | – | – | £37 | £5 | – | £42 | |||||||||||||||||||||||||||
Dr Jean-Pierre Garnier |
b | – | $118 | – | $118 | – | $5,885 | – | $5,885 | |||||||||||||||||||||||||||
Total remuneration |
£3,873 | £574 | £2,518 | £6,965 | £3,893 | £4,160 | £3,525 | £11,578 | ||||||||||||||||||||||||||||
Analysed as: |
||||||||||||||||||||||||||||||||||||
Executive Directors |
£2,140 | £495 | £2,518 | £5,153 | £2,009 | £373 | £3,525 | £5,907 | ||||||||||||||||||||||||||||
Non-Executive Directors |
£1,733 | £3 | – | £1,736 | £1,719 | £5 | – | £1,724 | ||||||||||||||||||||||||||||
Former Directors |
– | £76 | – | £76 | £165 | £3,782 | – | £3,947 | ||||||||||||||||||||||||||||
Total remuneration |
£3,873 | £574 | £2,518 | £6,965 | £3,893 | £4,160 | £3,525 | £11,578 | ||||||||||||||||||||||||||||
| a) | Andrew Witty and Julian Heslop both participate in Salary Sacrifice schemes. | |
| b) | Following the merger, and in order to encourage employees to convert their non-savings related options held over legacy shares or ADS for options over GlaxoSmithKline shares or ADS, employees were granted an additional cash benefit equal to 10% of the grant price of the original option. This additional benefit, known as the Exchange Offer Incentive, was only payable when the new option was exercised or lapsed underwater. To qualify for this additional cash benefit, participants had to retain these options until at least the second anniversary of the effective date of the merger. During the year, Andrew Witty received £93,002 (2009 – £49,499) and Julian Heslop received £89,936 (2009 – £32,000). Dr Jean-Pierre Garnier received $5,512,369 in 2009 as a result of options granted to him in 1999 lapsing. No further payments will be made. | |
| c) | Andrew Witty and Dr Moncef Slaoui have elected to participate in GSK’s Deferred Annual Bonus Plan in respect of their 2010 bonuses. Andrew Witty also participated in 2009 (Dr Moncef Slaoui was not eligible to participate in that year). | |
| d) | Dr Moncef Slaoui is a Non-Executive Director of the Agency for Science, Technology and Research (A*STAR) in respect of which he received $1,005 (2009 – $3,951) during 2010 which is not included above. | |
| e) | Other benefits in 2009 for Andrew Witty and Dr Moncef Slaoui have been restated to reflect certain elements of remuneration no longer being deemed a benefit. | |
| f) | James Murdoch was appointed to the Board with effect from 20th May 2009. | |
| g) | Dr Michele Barzach received fees of €nil (2009 – €89,700) from GSK France for healthcare consultancy provided. These are included within fees and salary above, | |
| h) | Sir Ian Prosser and Dr Ronaldo Schmitz retired as Non-Executive Directors of the company on 20th May 2009. On leaving the Board both Sir Ian Prosser and Dr Ronaldo Schmitz received the accumulated balance of shares previously awarded under the Non-Executive Directors’ share arrangements based on the share price at the payment date. A final payment in respect of the balance for Sir Ian Prosser was made during 2010. Further details are as set out in the table on page 96. These are not included within fees and salaries above. |
Table of Contents
95
| 2010 | 2009 | |||||||||||||||||||||||
| Total | Cash | Shares/ADS | Total | Cash | Shares/ADS | |||||||||||||||||||
| Fees | 000 | 000 | 000 | 000 | 000 | 000 | ||||||||||||||||||
Current Non-Executive Directors |
||||||||||||||||||||||||
Professor Sir Roy Anderson |
£128 | £96 | £32 | £120 | £90 | £30 | ||||||||||||||||||
Sir Crispin Davis |
£118 | – | £118 | £102 | – | £102 | ||||||||||||||||||
Sir Christopher Gent |
£675 | £540 | £135 | £675 | £540 | £135 | ||||||||||||||||||
James Murdoch |
£98 | – | £98 | £54 | £40 | £14 | ||||||||||||||||||
Tom de Swaan |
£177 | £133 | £44 | £133 | £99 | £34 | ||||||||||||||||||
Sir Robert Wilson |
£128 | £96 | £32 | £116 | £87 | £29 | ||||||||||||||||||
Dr Stephanie Burns |
$146 | $73 | $73 | $188 | $141 | $47 | ||||||||||||||||||
Larry Culp |
$135 | – | $135 | $188 | $141 | $47 | ||||||||||||||||||
Sir Deryck Maughan |
$147 | – | $147 | $188 | $141 | $47 | ||||||||||||||||||
Dr Daniel Podolsky |
$208 | $52 | $156 | $245 | $184 | $61 | ||||||||||||||||||
Former Non-Executive Directors |
||||||||||||||||||||||||
Sir Ian Prosser |
– | – | – | £48 | £31 | £17 | ||||||||||||||||||
Dr Ronaldo Schmitz |
– | – | – | £37 | £26 | £11 | ||||||||||||||||||
Total Remuneration |
£1,733 | £945 | £788 | £1,804 | £1,302 | £502 | ||||||||||||||||||
| Number of shares and ADS | ||||||||||||||||||||
| Non-Executive Directors’ share arrangements | 31st December 2009 | Allocated & elected |
Dividends reinvested |
Paid out | 31st December 2010 | |||||||||||||||
Current Non-Executive Directors |
||||||||||||||||||||
Shares |
||||||||||||||||||||
Professor Sir Roy Anderson |
5,730 | 2,585 | 305 | – | 8,620 | |||||||||||||||
Sir Crispin Davis |
42,909 | 9,527 | 2,206 | – | 54,642 | |||||||||||||||
Sir Christopher Gent |
53,025 | 11,006 | 2,709 | – | 66,740 | |||||||||||||||
James Murdoch |
1,077 | 7,896 | 132 | – | 9,105 | |||||||||||||||
Tom de Swaan |
8,952 | 3,605 | 471 | – | 13,028 | |||||||||||||||
Sir Robert Wilson |
12,133 | 2,585 | 623 | – | 15,341 | |||||||||||||||
ADS |
||||||||||||||||||||
Dr Stephanie Burns |
5,096 | 1,984 | 272 | – | 7,352 | |||||||||||||||
Larry Culp |
18,832 | 3,609 | 976 | – | 23,417 | |||||||||||||||
Sir Deryck Maughan |
16,678 | 3,917 | 867 | – | 21,462 | |||||||||||||||
Dr Daniel Podolsky |
8,017 | 4,182 | 436 | – | 12,635 | |||||||||||||||
Former Non-Executive Directors |
||||||||||||||||||||
Shares |
||||||||||||||||||||
Sir Ian Prosser |
1,240 | – | – | 1,240 | – | |||||||||||||||
Table of Contents
96
| Value of | Value of | |||||||||||||||||||||||
| Date of | awards on | awards on | Payments | Payments | ||||||||||||||||||||
| Footnote | leaving | allocation | leaving | in 2009 | in 2010 | |||||||||||||||||||
Sir Ian Prosser |
a,b | 20.05.09 | £382,142 | £356,644 | £343,525 | £15,767 | ||||||||||||||||||
| a) | The changes in value of awards between allocation, leaving and subsequent payment are attributable to dividends reinvested and the change in the share price for each award. | |
| b) | Awards to Sir Ian Prosser under the Non-Executive Directors’ share arrangements were partially settled in shares during 2009 with the balance of 1,240 shares settled in 2010. |
| Shares | ADS | |||||||||||||||||||||||||||
| 24th February | 31st December | 1st January | 24th February | 31st December | 1st January | |||||||||||||||||||||||
| Footnote | 2011 | 2010 | 2010 | 2011 | 2010 | 2010 | ||||||||||||||||||||||
Executive Directors |
||||||||||||||||||||||||||||
Andrew Witty |
a,d | 184,281 | 151,213 | 91,472 | – | – | – | |||||||||||||||||||||
Simon Dingemans |
b | 40,000 | – | – | – | – | – | |||||||||||||||||||||
Julian Heslop |
a,d | 76,900 | 76,254 | 49,350 | – | – | – | |||||||||||||||||||||
Dr Moncef Slaoui |
c,d | 59,622 | 59,133 | 60,948 | 37,883 | 18,459 | 592 | |||||||||||||||||||||
Non-Executive Directors |
||||||||||||||||||||||||||||
Professor Sir Roy Anderson |
e | 8,620 | 8,620 | 5,730 | – | – | – | |||||||||||||||||||||
Dr Stephanie Burns |
e | 44 | 44 | 44 | 7,418 | 7,418 | 5,161 | |||||||||||||||||||||
Larry Culp |
e | – | – | – | 23,417 | 23,417 | 18,832 | |||||||||||||||||||||
Sir Crispin Davis |
e | 61,402 | 61,402 | 49,669 | – | – | – | |||||||||||||||||||||
Sir Christopher Gent |
e | 66,741 | 66,741 | 53,025 | – | – | – | |||||||||||||||||||||
Sir Deryck Maughan |
e | – | – | – | 21,462 | 21,462 | 16,678 | |||||||||||||||||||||
James Murdoch |
e | 10,105 | 10,105 | 2,077 | – | – | – | |||||||||||||||||||||
Dr Daniel Podolsky |
e | – | – | – | 12,635 | 12,635 | 8,017 | |||||||||||||||||||||
Tom de Swaan |
e | 13,028 | 13,028 | 8,952 | – | – | – | |||||||||||||||||||||
Sir Robert Wilson |
e | 21,470 | 21,470 | 18,262 | – | – | – | |||||||||||||||||||||
| a) | Includes shares purchased through the GlaxoSmithKline ShareReward Plan for Andrew Witty totalling 2,577 at 31st December 2010 (31st December 2009 – 2,216) and 2,628 shares at 24th February 2011 and Julian Heslop totalling 2,577 at 31st December 2010 (31st December 2009 – 2,216) and 2,628 shares at 24th February 2011. | |
| b) | Simon Dingemans joined the Board on 4th January 2011. | |
| c) | Includes ADS purchased in the GlaxoSmithKline Stock Fund within the US Retirement Savings Plan and US Executive Supplemental Savings Plan. | |
| d) | The 2008 Performance Share Plan vesting conditions were approved on 24th February 2011, as detailed on page 91. However, the shares did not vest until 25th February 2011 and are not included in the totals above. | |
| e) | Includes shares and ADS received as part or all of their fees, as described under Non-Executive Directors’ share allocation plan on page 93. Dividends received on these shares and ADS were converted to shares and ADS as at 31st December 2010. |
Table of Contents
97
| Options – Shares | Granted | |||||||||||||||||||||||||||||||
| Footnote | 31st December 2009 |
Date of grant | Exercise period | Grant price | Number | Lapsed | 31st December 2010 |
|||||||||||||||||||||||||
Andrew Witty |
1,549,702 | – | – | – | – | 259,200 | 1,290,502 | |||||||||||||||||||||||||
Julian Heslop |
889,400 | – | – | – | – | 304,350 | 585,050 | |||||||||||||||||||||||||
Dr Moncef Slaoui |
155,190 | – | – | – | – | 14,870 | 140,320 | |||||||||||||||||||||||||
| Options – ADS | Granted | |||||||||||||||||||||||||||||||
| 31st December 2009 |
Date of grant | Exercise period | Grant price | Number | Lapsed | 31st December 2010 |
||||||||||||||||||||||||||
Dr Moncef Slaoui |
a | 489,330 | 22.02.10 | 22.02.13 – 22.02.20 | 37.32 | 1,100 | 168,695 | 321,735 | ||||||||||||||||||||||||
| a) | These details include a change to Dr Slaoui’s connected person, who is also an employee of GSK. |
| Weighted average | Vesting date | Lapse date | ||||||||||||||||||||||||
| Andrew Witty | grant price | Number | earliest | latest | earliest | latest | ||||||||||||||||||||
Options above market price at year-end: |
vested | 15.26 | 369,993 | 29.03.04 | 20.02.09 | 28.03.11 | 22.02.16 | |||||||||||||||||||
Options below market price at year-end: |
vested | 11.39 | 249,500 | 02.12.05 | 30.11.07 | 30.11.12 | 01.12.14 | |||||||||||||||||||
| unvested | 11.63 | 671,009 | 18.02.11 | 01.12.11 | 31.05.12 | 20.07.18 | ||||||||||||||||||||
Total share options as at 31st December 2010 |
12.62 | 1,290,502 | ||||||||||||||||||||||||
| Weighted average | Vesting date | Lapse date | ||||||||||||||||||||||||
| Julian Heslop | grant price | Number | earliest | latest | earliest | latest | ||||||||||||||||||||
Options above market price at year-end: |
vested | 15.59 | 279,117 | 29.03.04 | 20.02.09 | 28.03.11 | 22.02.16 | |||||||||||||||||||
Options below market price at year-end: |
vested | 11.23 | 62,250 | 03.12.07 | 03.12.07 | 01.12.14 | 01.12.14 | |||||||||||||||||||
| unvested | 11.46 | 243,683 | 18.02.11 | 30.11.12 | 31.05.13 | 16.02.18 | ||||||||||||||||||||
Total share options as at 31st December 2010 |
13.41 | 585,050 | ||||||||||||||||||||||||
| Weighted average | Vesting date | Lapse date | ||||||||||||||||||||||||
| Dr Moncef Slaoui | grant price | Number | earliest | latest | earliest | latest | ||||||||||||||||||||
Options above market price at year-end: |
vested | 14.68 | 68,520 | 20.02.09 | 20.02.09 | 23.02.16 | 20.02.16 | |||||||||||||||||||
Options below market price at year-end: |
vested | 11.58 | 71,800 | 02.12.05 | 03.12.07 | 03.12.12 | 01.12.14 | |||||||||||||||||||
Total share options as at 31st December 2010 |
13.09 | 140,320 | ||||||||||||||||||||||||
Options above market price at year-end: |
vested | 56.92 | 935 | 27.07.09 | 19.02.10 | 25.07.16 | 17.02.17 | |||||||||||||||||||
| unvested | 44.75 | 159,850 | 18.02.11 | 18.02.11 | 16.02.18 | 16.02.18 | ||||||||||||||||||||
Options below market price at year-end: |
unvested | 33.45 | 160,950 | 17.02.12 | 21.02.13 | 15.02.19 | 20.02.20 | |||||||||||||||||||
Total ADS options as at 31st December 2010 |
39.13 | 321,735 | ||||||||||||||||||||||||
Table of Contents
98
| Performance target | ||||||||||||||||||||
| Vesting status | Annualised growth | Percentage of | ||||||||||||||||||
| Grant | Footnote | Performance period | at 31st December 2010 | in EPS | award vesting | |||||||||||||||
February 2008 |
a | 2008 – 2010 | Unvested | > RPI + 6% | 100% | |||||||||||||||
| RPI + 5% | 83% | |||||||||||||||||||
| RPI + 4% | 67% | |||||||||||||||||||
| RPI + 3% | 50% | |||||||||||||||||||
| <RPI + 3% | 0% | |||||||||||||||||||
| a) | The performance targets for these share options were not met, and as a result they lapsed on the third anniversary of the date of grant. |
| Performance target | ||||||||||||||||||||
| Vesting status | Annualised growth | Percentage of | ||||||||||||||||||
| Grant | Performance period | at 31st December 2010 | in EPS | award vesting | ||||||||||||||||
February 2009 – 50% of award |
2009 – 2011 | Unvested | > RPI + 6% | 100% | ||||||||||||||||
February 2009 – 50% of award |
2009 – 2012 | Unvested | RPI + 5% | 85% | ||||||||||||||||
| RPI + 4% | 65% | |||||||||||||||||||
| RPI + 3% | 30% | |||||||||||||||||||
| <RPI + 3% | 0% | |||||||||||||||||||
| Andrew Witty – Shares | Vested | |||||||||||||||||||||||||||||||||||||||
| Unvested | Market price | Additional | Unvested | |||||||||||||||||||||||||||||||||||||
| at 31st | Number | price on | shares by | at 31st | Number | |||||||||||||||||||||||||||||||||||
| December | granted in | date of | Market | Vested | dividends | December | granted in | |||||||||||||||||||||||||||||||||
| Performance period | 2009 | 2010 | grant | Number | price | Gain | Lapsed | reinvested | 2010 | 2011 | ||||||||||||||||||||||||||||||
2007 – 2009 |
95,410 | – | £14.88 | 33,781 | £12.09 | 408,238 | 62,735 | 1,106 | – | – | ||||||||||||||||||||||||||||||
2008 – 2010 |
242,010 | – | £11.47 | – | – | – | – | 12,439 | 254,449 | – | ||||||||||||||||||||||||||||||
2008 – 2010 |
65,923 | – | £12.21 | – | – | – | – | 3,388 | 69,311 | – | ||||||||||||||||||||||||||||||
2009 – 2011 |
476,146 | – | £10.51 | – | – | – | – | 24,471 | 500,617 | – | ||||||||||||||||||||||||||||||
2010 – 2012 |
– | 415,454 | £12.04 | – | – | – | – | 10,327 | 425,781 | – | ||||||||||||||||||||||||||||||
2011 – 2013 |
– | – | – | – | – | – | – | – | – | 424,448 | ||||||||||||||||||||||||||||||
| Julian Heslop – Shares | Vested | |||||||||||||||||||||||||||||||||||||||
| Unvested | Market | Additional | Unvested | |||||||||||||||||||||||||||||||||||||
| at 31st | Number | price on | shares by | at 31st | ||||||||||||||||||||||||||||||||||||
| December | granted in | date of | Market | Vested | dividends | December | ||||||||||||||||||||||||||||||||||
| Performance period | 2009 | 2010 | grant | Number | price | Gain | Lapsed | reinvested | 2010 | |||||||||||||||||||||||||||||||
2007 – 2009 |
117,859 | – | £14.88 | 41,729 | £12.09 | 504,294 | 77,497 | 1,367 | – | |||||||||||||||||||||||||||||||
2008 – 2010 |
112,938 | – | £11.47 | – | – | – | – | 5,805 | 118,743 | |||||||||||||||||||||||||||||||
2009 – 2011 |
199,982 | – | £10.51 | – | – | – | – | 10,278 | 210,260 | |||||||||||||||||||||||||||||||
2010 – 2012 |
– | 174,491 | £12.04 | – | – | – | – | 4,337 | 178,828 | |||||||||||||||||||||||||||||||
Table of Contents
99
| Dr Moncef Slaoui – ADS | Vested | |||||||||||||||||||||||||||||||||||||||
| Unvested | Market | Additional | Unvested | |||||||||||||||||||||||||||||||||||||
| at 31st | Number | price on | shares by | at 31st | Number | |||||||||||||||||||||||||||||||||||
| December | granted in | date of | Market | Vested | dividends | December | granted in | |||||||||||||||||||||||||||||||||
| Performance period | 2009 | 2010 | grant | Number | price | Gain | Lapsed | reinvested | 2010 | 2011 | ||||||||||||||||||||||||||||||
2007 – 2009 |
79,286 | – | $58.00 | 27,454 | $37.42 | $1,027,342 | 52,748 | 916 | – | – | ||||||||||||||||||||||||||||||
2008 – 2010 |
75,991 | – | $44.75 | – | – | – | 1,778 | 3,939 | 78,152 | – | ||||||||||||||||||||||||||||||
2009 – 2011 |
2,686 | – | $33.42 | – | – | – | 2,825 | 139 | – | – | ||||||||||||||||||||||||||||||
2009 – 2011 |
69,804 | – | $33.50 | – | – | – | – | 3,618 | 73,422 | – | ||||||||||||||||||||||||||||||
2010 – 2012 |
– | 133,247 | – | – | – | – | 2,684 | 3,256 | 133,819 | – | ||||||||||||||||||||||||||||||
2011 – 2013 |
– | – | – | – | – | – | – | – | – | 147,521 | ||||||||||||||||||||||||||||||
| Simon Dingemans - Shares | Vested | |||||||||||||||||||||||||||||||||||||||
| Unvested | Market | Additional | Unvested | |||||||||||||||||||||||||||||||||||||
| at 31st | Number | price on | shares by | at 31st | Number | |||||||||||||||||||||||||||||||||||
| December | granted in | date of | Market | Vested | dividends | December | granted in | |||||||||||||||||||||||||||||||||
| Performance period | 2009 | 2010 | grant | Number | price | Gain | Lapsed | reinvested | 2010 | 2011 | ||||||||||||||||||||||||||||||
2011 – 2013 |
– | – | – | – | – | – | – | – | – | 196,095 | ||||||||||||||||||||||||||||||
| TSR vesting schedule | |||||||||||||||||
| Award | % of Award | Performance Period | TSR rank with 12 other companies | Percentage of award vesting | |||||||||||||
2008 |
100 | 2008 – 2010 | 1 | 100% | |||||||||||||
| 2 | 100% | ||||||||||||||||
| 3 | 87% | ||||||||||||||||
| 4 | 74% | ||||||||||||||||
| 5 | 61% | ||||||||||||||||
| 6 | 48% | ||||||||||||||||
| Median | 35% | ||||||||||||||||
| Below median | 0% | ||||||||||||||||
The following vesting schedules apply to PSP awards made in 2009 and 2010. |
|||||||||||||||||
| TSR vesting schedule | |||||||||||||||||
| Award | % of Award | Performance Period | TSR rank with 10 other companies | Percentage of award vesting | |||||||||||||
2009 |
30 | 2009 – 2011 | 1 | 100% | |||||||||||||
| 30 | 2009 – 2012 | 2 | 100% | ||||||||||||||
2010 |
30 | 2010 – 2012 | 3 | 100% | |||||||||||||
| 30 | 2010 – 2013 | 4 | 80% | ||||||||||||||
| 5 | 55% | ||||||||||||||||
| Median | 30% | ||||||||||||||||
| Below median | 0% | ||||||||||||||||
| Adjusted free cash flow vesting schedule | |||||||||||||||||
| Cash flow Targets | |||||||||||||||||
| Award | % of Award | Performance Period | £bn | Percentage of award vesting | |||||||||||||
2009 |
40 | 2009 – 2011 | 13.5 – 16.0 | 25% – 100% | |||||||||||||
2010 |
40 | 2010 – 2012 | 17.3 – 20.5 | 25% – 100% | |||||||||||||
Table of Contents
100
| Andrew Witty – Shares | Vested | |||||||||||||||||||||||||||||||||||||||
| Unvested | Market | Additional | Unvested | |||||||||||||||||||||||||||||||||||||
| at 31st | Number | price on | shares by | at 31st | Number | |||||||||||||||||||||||||||||||||||
| December | granted in | date of | Market | Vested | dividends | December | granted in | |||||||||||||||||||||||||||||||||
| Performance period | 2009 | 2010 | grant | Number | price | Gain | Lapsed | reinvested | 2010 | 2011 | ||||||||||||||||||||||||||||||
2010 – 2012 |
– | 24,291 | 12.35 | – | – | – | – | 616 | 24,907 | – | ||||||||||||||||||||||||||||||
2011 – 2013 |
– | – | – | – | – | – | – | – | – | 31,921 | ||||||||||||||||||||||||||||||
| Dr Moncef Slaoui - ADS | Vested | |||||||||||||||||||||||||||||||||||||||
| Unvested | Market | Additional | Unvested | |||||||||||||||||||||||||||||||||||||
| at 31st | Number | price on | shares by | at 31st | Number | |||||||||||||||||||||||||||||||||||
| December | granted in | date of | Market | Vested | dividends | December | granted in | |||||||||||||||||||||||||||||||||
| Performance period | 2009 | 2010 | grant | Number | price | Gain | Lapsed | reinvested | 2010 | 2011 | ||||||||||||||||||||||||||||||
2011 – 2013 |
– | – | – | – | – | – | – | – | – | 18,756 | ||||||||||||||||||||||||||||||
| Share Value Plan awards | ||||||||||||||||||||||||||||
| Dr Moncef Slaoui – Shares and ADS | Vested & deferred | |||||||||||||||||||||||||||
| Unvested | Market | Unvested | ||||||||||||||||||||||||||
| Plan year | at 31st | price on | at 31st | Number | ||||||||||||||||||||||||
| December | date of | Market | December | granted in | ||||||||||||||||||||||||
| 2009 | grant | Number | price | Gain | 2010 | 2011 | ||||||||||||||||||||||
2007 |
510 | $58.00 | 510 | $37.32 | 19,033 | – | – | |||||||||||||||||||||
2008 |
640 | $44.75 | – | – | – | 640 | – | |||||||||||||||||||||
2009 |
640 | $33.42 | – | – | – | 640 | – | |||||||||||||||||||||
2010 |
640 | $37.32 | – | – | – | 640 | – | |||||||||||||||||||||
2011 |
– | $38.13 | – | – | – | – | 2,450 | |||||||||||||||||||||
| Accrued | Change in | |||||||||||||||||||||||||||||||||||
| benefit at | Accrued | Change in | Personal | Transfer | Transfer | accrued | Transfer value | |||||||||||||||||||||||||||||
| 31st | benefit at | accrued | contributions | value at | value at | Change | benefit over | of change | ||||||||||||||||||||||||||||
| December | 31st December | benefit | made during | 31st December | 31st December | in transfer | year net | in accrued | ||||||||||||||||||||||||||||
| 2009 | 2010 | over year | the year | 2009 | 2010 | value | of inflation | benefit* | ||||||||||||||||||||||||||||
| Executive Directors | 000 | 000 | 000 | 000 | 000 | 000 | 000* | 000 | 000 | |||||||||||||||||||||||||||
Andrew Witty |
£446 | £497 | £51 | £30 | £6,272 | £9,651 | £3,349 | £51 | £987 | |||||||||||||||||||||||||||
Julian Heslop |
£201 | £222 | £21 | £16 | £3,787 | £5,308 | £1,505 | £21 | £563 | |||||||||||||||||||||||||||
Dr Moncef Slaoui |
$187 | $230 | $43 | – | $1,101 | $1,518 | $417 | $41 | – | |||||||||||||||||||||||||||
Dr Moncef Slaoui |
€59 | €65 | €6 | – | €647 | €689 | €42 | €5 | – | |||||||||||||||||||||||||||
| * | These are shown net of contributions made by the individual. |
Table of Contents
101
Remuneration Committee Chairman
1st March 2011
Table of Contents
102
to the Group financial statements
| • | select suitable accounting policies and then apply them consistently; | |
| • | make judgements and accounting estimates that are reasonable and prudent; | |
| • | state that the Group financial statements comply with IFRS as adopted by the European Union and IFRS as issued by the IASB, subject to any material departures disclosed and explained in the Group financial statements. |
| • | the Group financial statements, which have been prepared in accordance with IFRS as adopted by the EU and IFRS as issued by IASB, give a true and fair view of the assets, liabilities, financial position and profit of the Group; and |
| • | the Business review section contained in the Annual Report includes a fair review of the development and performance of the business and the position of the Group, together with a description of the principal risks and uncertainties that it faces. |
| • | so far as he or she is aware, there is no relevant audit information of which the company’s auditors are unaware; and | |
| • | he or she has taken all the steps that he or she ought to have taken as a Director to make himself or herself aware of any relevant audit information and to establish that the company’s auditors are aware of that information. |
Chairman
1st March 2011
Table of Contents
103
London
United Kingdom
4th March 2011
Table of Contents
104
| 2010 | ||||||||||||||||
| Results | ||||||||||||||||
| before major | Major | |||||||||||||||
| restructuring | restructuring | Total | ||||||||||||||
| Notes | £m | £m | £m | |||||||||||||
Turnover |
6 | 28,392 | – | 28,392 | ||||||||||||
Cost of sales |
(7,405 | ) | (187 | ) | (7,592 | ) | ||||||||||
Gross profit |
20,987 | (187 | ) | 20,800 | ||||||||||||
Selling, general and administration |
(12,388 | ) | (665 | ) | (13,053 | ) | ||||||||||
Research and development |
(3,964 | ) | (493 | ) | (4,457 | ) | ||||||||||
Other operating income |
8 | 493 | – | 493 | ||||||||||||
Operating profit |
9 | 5,128 | (1,345 | ) | 3,783 | |||||||||||
Finance income |
11 | 116 | – | 116 | ||||||||||||
Finance costs |
12 | (828 | ) | (3 | ) | (831 | ) | |||||||||
Profit on disposal of interest in associates |
8 | – | 8 | |||||||||||||
Share of after tax profits of associates and joint ventures |
13 | 81 | – | 81 | ||||||||||||
Profit before taxation |
4,505 | (1,348 | ) | 3,157 | ||||||||||||
Taxation |
14 | (1,544 | ) | 240 | (1,304 | ) | ||||||||||
Profit after taxation for the year |
2,961 | (1,108 | ) | 1,853 | ||||||||||||
Profit attributable to non-controlling interests |
219 | – | 219 | |||||||||||||
Profit attributable to shareholders |
2,742 | (1,108 | ) | 1,634 | ||||||||||||
| 2,961 | (1,108 | ) | 1,853 | |||||||||||||
Basic earnings per share (pence) |
15 | 32.1 | p | |||||||||||||
Diluted earnings per share (pence) |
15 | 31.9 | p | |||||||||||||
| 2010 | ||||
| £m | ||||
Profit for the year |
1,853 | |||
Exchange movements on overseas net assets and net investment hedges |
166 | |||
Reclassification of exchange on liquidation or disposal of overseas subsidiaries |
(2 | ) | ||
Tax on exchange movements |
– | |||
Fair value movements on available-for-sale investments |
94 | |||
Deferred tax on fair value movements on available-for-sale investments |
(25 | ) | ||
Reclassification of fair value movements on available-for-sale investments |
1 | |||
Deferred tax reversed on reclassification of available-for-sale investments |
(3 | ) | ||
Fair value movements on cash flow hedges |
(8 | ) | ||
Deferred tax on fair value movements on cash flow hedges |
1 | |||
Reclassification of cash flow hedges to income statement |
3 | |||
Fair value movement on subsidiary acquisition |
– | |||
Cash flow
hedge reclassified to goodwill |
6 | |||
Actuarial losses on defined benefit plans |
(1 | ) | ||
Deferred tax on actuarial movements in defined benefit plans |
1 | |||
Other comprehensive income/(expense) for the year |
233 | |||
Total comprehensive income for the year |
2,086 | |||
Total comprehensive income for the year attributable to: |
||||
Shareholders |
1,847 | |||
Non-controlling interests |
239 | |||
Total comprehensive income for the year |
2,086 | |||
Table of Contents
105
| 2009 | 2008 | |||||||||||||||||||||
| Results | Results | |||||||||||||||||||||
| before major | Major | before major | Major | |||||||||||||||||||
| restructuring | restructuring | Total | restructuring | restructuring | Total | |||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||
| 28,368 | – | 28,368 | 24,352 | – | 24,352 | |||||||||||||||||
| (7,095 | ) | (285 | ) | (7,380 | ) | (5,776 | ) | (639 | ) | (6,415 | ) | |||||||||||
| 21,273 | (285 | ) | 20,988 | 18,576 | (639 | ) | 17,937 | |||||||||||||||
| (9,200 | ) | (392 | ) | (9,592 | ) | (7,352 | ) | (304 | ) | (7,656 | ) | |||||||||||
| (3,951 | ) | (155 | ) | (4,106 | ) | (3,506 | ) | (175 | ) | (3,681 | ) | |||||||||||
| 1,135 | – | 1,135 | 541 | – | 541 | |||||||||||||||||
| 9,257 | (832 | ) | 8,425 | 8,259 | (1,118 | ) | 7,141 | |||||||||||||||
| 70 | – | 70 | 313 | – | 313 | |||||||||||||||||
| (780 | ) | (3 | ) | (783 | ) | (838 | ) | (5 | ) | (843 | ) | |||||||||||
| 115 | – | 115 | – | – | – | |||||||||||||||||
| 64 | – | 64 | 48 | – | 48 | |||||||||||||||||
| 8,726 | (835 | ) | 7,891 | 7,782 | (1,123 | ) | 6,659 | |||||||||||||||
| (2,443 | ) | 221 | (2,222 | ) | (2,231 | ) | 284 | (1,947 | ) | |||||||||||||
| 6,283 | (614 | ) | 5,669 | 5,551 | (839 | ) | 4,712 | |||||||||||||||
| 138 | – | 138 | 110 | – | 110 | |||||||||||||||||
| 6,145 | (614 | ) | 5,531 | 5,441 | (839 | ) | 4,602 | |||||||||||||||
| 6,283 | (614 | ) | 5,669 | 5,551 | (839 | ) | 4,712 | |||||||||||||||
| 109.1 | p | 88.6 | p | |||||||||||||||||||
| 108.2 | p | 88.1 | p | |||||||||||||||||||
| 2009 | 2008 | |||||||||||||||||||||
| £m | £m | |||||||||||||||||||||
| 5,669 | 4,712 | |||||||||||||||||||||
| (194 | ) | 1,017 | ||||||||||||||||||||
| (44 | ) | 84 | ||||||||||||||||||||
| 19 | 15 | |||||||||||||||||||||
| 42 | (47 | ) | ||||||||||||||||||||
| (24 | ) | 5 | ||||||||||||||||||||
| – | (34 | ) | ||||||||||||||||||||
| 13 | 3 | |||||||||||||||||||||
| (6 | ) | 6 | ||||||||||||||||||||
| 2 | (3 | ) | ||||||||||||||||||||
| 1 | – | |||||||||||||||||||||
| (6 | ) | – | ||||||||||||||||||||
| – | – | |||||||||||||||||||||
| (659 | ) | (1,370 | ) | |||||||||||||||||||
| 183 | 441 | |||||||||||||||||||||
| (673 | ) | 117 | ||||||||||||||||||||
| 4,996 | 4,829 | |||||||||||||||||||||
| 4,895 | 4,670 | |||||||||||||||||||||
| 101 | 159 | |||||||||||||||||||||
| 4,996 | 4,829 | |||||||||||||||||||||
Table of Contents
106
| 2010 | 2009 | |||||||||||
| Notes | £m | £m | ||||||||||
Non-current assets |
||||||||||||
Property, plant and equipment |
17 | 9,045 | 9,374 | |||||||||
Goodwill |
18 | 3,606 | 3,361 | |||||||||
Other intangible assets |
19 | 8,532 | 8,183 | |||||||||
Investments in associates and joint ventures |
20 | 1,081 | 895 | |||||||||
Other investments |
21 | 711 | 454 | |||||||||
Deferred tax assets |
14 | 2,566 | 2,374 | |||||||||
Derivative financial instruments |
41 | 97 | 68 | |||||||||
Other non-current assets |
22 | 556 | 583 | |||||||||
Total non-current assets |
26,194 | 25,292 | ||||||||||
Current assets |
||||||||||||
Inventories |
23 | 3,837 | 4,064 | |||||||||
Current tax recoverable |
14 | 56 | 58 | |||||||||
Trade and other receivables |
24 | 5,793 | 6,492 | |||||||||
Derivative financial instruments |
41 | 93 | 129 | |||||||||
Liquid investments |
32 | 184 | 268 | |||||||||
Cash and cash equivalents |
25 | 6,057 | 6,545 | |||||||||
Assets held for sale |
26 | 16 | 14 | |||||||||
Total current assets |
16,036 | 17,570 | ||||||||||
Total assets |
42,230 | 42,862 | ||||||||||
Current liabilities |
||||||||||||
Short-term borrowings |
32 | (291 | ) | (1,471 | ) | |||||||
Trade and other payables |
27 | (6,888 | ) | (6,772 | ) | |||||||
Derivative financial instruments |
41 | (188 | ) | (168 | ) | |||||||
Current tax payable |
14 | (1,047 | ) | (1,451 | ) | |||||||
Short-term provisions |
29 | (4,380 | ) | (2,256 | ) | |||||||
Total current liabilities |
(12,794 | ) | (12,118 | ) | ||||||||
Non-current liabilities |
||||||||||||
Long-term borrowings |
32 | (14,809 | ) | (14,786 | ) | |||||||
Deferred tax liabilities |
14 | (707 | ) | (645 | ) | |||||||
Pensions and other post-employment benefits |
28 | (2,672 | ) | (2,981 | ) | |||||||
Other provisions |
29 | (904 | ) | (985 | ) | |||||||
Derivative financial instruments |
41 | (5 | ) | – | ||||||||
Other non-current liabilities |
30 | (594 | ) | (605 | ) | |||||||
Total non-current liabilities |
(19,691 | ) | (20,002 | ) | ||||||||
Total liabilities |
(32,485 | ) | (32,120 | ) | ||||||||
Net assets |
9,745 | 10,742 | ||||||||||
Equity |
||||||||||||
Share capital |
33 | 1,418 | 1,416 | |||||||||
Share premium account |
33 | 1,428 | 1,368 | |||||||||
Retained earnings |
34 | 4,779 | 6,321 | |||||||||
Other reserves |
34 | 1,262 | 900 | |||||||||
Shareholders’ equity |
8,887 | 10,005 | ||||||||||
Non-controlling interests |
858 | 737 | ||||||||||
Total equity |
9,745 | 10,742 | ||||||||||
Chairman
Table of Contents
107
| Shareholders’ equity | ||||||||||||||||||||||||||||
| Non- | ||||||||||||||||||||||||||||
| Share | Share | Retained | Other | controlling | Total | |||||||||||||||||||||||
| capital | premium | earnings | reserves | Total | interests | equity | ||||||||||||||||||||||
| £m | £m | £m | £m | £m | £m | £m | ||||||||||||||||||||||
At 1st January 2008 |
1,503 | 1,266 | 6,475 | 359 | 9,603 | 307 | 9,910 | |||||||||||||||||||||
Profit for the year |
– | – | 4,602 | – | 4,602 | 110 | 4,712 | |||||||||||||||||||||
Other comprehensive income/(expense) for the year |
– | – | 121 | (53 | ) | 68 | 49 | 117 | ||||||||||||||||||||
Distributions to non-controlling interests |
– | – | – | – | – | (79 | ) | (79 | ) | |||||||||||||||||||
Dividends to shareholders |
– | – | (2,929 | ) | – | (2,929 | ) | – | (2,929 | ) | ||||||||||||||||||
Ordinary shares issued |
2 | 60 | – | – | 62 | – | 62 | |||||||||||||||||||||
Ordinary shares purchased and cancelled |
(90 | ) | – | (3,706 | ) | 90 | (3,706 | ) | – | (3,706 | ) | |||||||||||||||||
Ordinary shares acquired by ESOP Trusts |
– | – | – | (19 | ) | (19 | ) | – | (19 | ) | ||||||||||||||||||
Ordinary shares transferred by ESOP Trusts |
– | – | – | 10 | 10 | – | 10 | |||||||||||||||||||||
Write-down of shares held by ESOP Trusts |
– | – | (181 | ) | 181 | – | – | – | ||||||||||||||||||||
Share-based incentive plans |
– | – | 241 | – | 241 | – | 241 | |||||||||||||||||||||
Tax on share-based incentive plans |
– | – | (1 | ) | – | (1 | ) | – | (1 | ) | ||||||||||||||||||
At 31st December 2008 |
1,415 | 1,326 | 4,622 | 568 | 7,931 | 387 | 8,318 | |||||||||||||||||||||
Profit for the year |
– | – | 5,531 | – | 5,531 | 138 | 5,669 | |||||||||||||||||||||
Other comprehensive (expense)/income for the year |
– | – | (663 | ) | 27 | (636 | ) | (37 | ) | (673 | ) | |||||||||||||||||
Distributions to non-controlling interests |
– | – | – | – | – | (89 | ) | (89 | ) | |||||||||||||||||||
Changes in non-controlling interests |
– | – | – | – | – | 338 | 338 | |||||||||||||||||||||
Put option over non-controlling interest |
– | – | – | (2 | ) | (2 | ) | – | (2 | ) | ||||||||||||||||||
Dividends to shareholders |
– | – | (3,003 | ) | – | (3,003 | ) | – | (3,003 | ) | ||||||||||||||||||
Ordinary shares issued |
1 | 42 | – | – | 43 | – | 43 | |||||||||||||||||||||
Ordinary shares acquired by ESOP Trusts |
– | – | – | (57 | ) | (57 | ) | – | (57 | ) | ||||||||||||||||||
Ordinary shares transferred by ESOP Trusts |
– | – | – | 13 | 13 | – | 13 | |||||||||||||||||||||
Write-down of shares held by ESOP Trusts |
– | – | (351 | ) | 351 | – | – | – | ||||||||||||||||||||
Share-based incentive plans |
– | – | 171 | – | 171 | – | 171 | |||||||||||||||||||||
Tax on share-based incentive plans |
– | – | 14 | – | 14 | – | 14 | |||||||||||||||||||||
At 31st December 2009 |
1,416 | 1,368 | 6,321 | 900 | 10,005 | 737 | 10,742 | |||||||||||||||||||||
Profit for the year |
– | – | 1,634 | – | 1,634 | 219 | 1,853 | |||||||||||||||||||||
Other comprehensive income for the year |
– | – | 144 | 69 | 213 | 20 | 233 | |||||||||||||||||||||
Distributions to non-controlling interests |
– | – | – | – | – | (118 | ) | (118 | ) | |||||||||||||||||||
Dividends to shareholders |
– | – | (3,205 | ) | – | (3,205 | ) | – | (3,205 | ) | ||||||||||||||||||
Ordinary shares issued |
2 | 60 | – | – | 62 | – | 62 | |||||||||||||||||||||
Ordinary shares acquired by ESOP Trusts |
– | – | – | (16 | ) | (16 | ) | – | (16 | ) | ||||||||||||||||||
Ordinary shares transferred by ESOP Trusts |
– | – | – | 17 | 17 | – | 17 | |||||||||||||||||||||
Write-down of shares held by ESOP Trusts |
– | – | (292 | ) | 292 | – | – | – | ||||||||||||||||||||
Share-based incentive plans |
– | – | 175 | – | 175 | – | 175 | |||||||||||||||||||||
Tax on share-based incentive plans |
– | – | 2 | – | 2 | – | 2 | |||||||||||||||||||||
At 31st December 2010 |
1,418 | 1,428 | 4,779 | 1,262 | 8,887 | 858 | 9,745 | |||||||||||||||||||||
Table of Contents
108
| 2010 | 2009 | 2008 | ||||||||||||||
| Notes | £m | £m | £m | |||||||||||||
Cash flow from operating activities |
||||||||||||||||
Profit after taxation for the year |
1,853 | 5,669 | 4,712 | |||||||||||||
Adjustments reconciling profit after tax to operating cash flows |
36 | 6,778 | 3,876 | 4,343 | ||||||||||||
Cash generated from operations |
8,631 | 9,545 | 9,055 | |||||||||||||
Taxation paid |
(1,834 | ) | (1,704 | ) | (1,850 | ) | ||||||||||
Net cash inflow from operating activities |
6,797 | 7,841 | 7,205 | |||||||||||||
Cash flow from investing activities |
||||||||||||||||
Purchase of property, plant and equipment |
(1,014 | ) | (1,418 | ) | (1,437 | ) | ||||||||||
Proceeds from sale of property, plant and equipment |
92 | 48 | 20 | |||||||||||||
Purchase of intangible assets |
(621 | ) | (455 | ) | (632 | ) | ||||||||||
Proceeds from sale of intangible assets |
126 | 356 | 171 | |||||||||||||
Purchase of equity investments |
(279 | ) | (154 | ) | (87 | ) | ||||||||||
Proceeds from sale of equity investments |
27 | 59 | 42 | |||||||||||||
Purchase of businesses, net of cash acquired |
38 | (354 | ) | (2,792 | ) | (454 | ) | |||||||||
Investments in associates and joint ventures |
38 | (61 | ) | (29 | ) | (9 | ) | |||||||||
Decrease in liquid investments |
91 | 87 | 905 | |||||||||||||
Interest received |
107 | 90 | 320 | |||||||||||||
Dividends from associates and joint ventures |
18 | 17 | 12 | |||||||||||||
Proceeds from disposal of associates |
– | 178 | – | |||||||||||||
Net cash outflow from investing activities |
(1,868 | ) | (4,013 | ) | (1,149 | ) | ||||||||||
Cash flow from financing activities |
||||||||||||||||
Proceeds from own shares for employee share options |
17 | 13 | 9 | |||||||||||||
Shares acquired by ESOP Trusts |
(16 | ) | (57 | ) | (19 | ) | ||||||||||
Issue of share capital |
33 | 62 | 43 | 62 | ||||||||||||
Purchase of own shares for cancellation |
– | – | (3,706 | ) | ||||||||||||
Increase in long-term loans |
– | 1,358 | 5,523 | |||||||||||||
Increase in short-term loans |
6 | 646 | 275 | |||||||||||||
Repayment of short-term loans |
(1,296 | ) | (748 | ) | (3,334 | ) | ||||||||||
Net repayment of obligations under finance leases |
(45 | ) | (48 | ) | (48 | ) | ||||||||||
Interest paid |
(775 | ) | (780 | ) | (730 | ) | ||||||||||
Dividends paid to shareholders |
(3,205 | ) | (3,003 | ) | (2,929 | ) | ||||||||||
Distributions to non-controlling interests |
(118 | ) | (89 | ) | (79 | ) | ||||||||||
Other financing cash flows |
(201 | ) | (109 | ) | 68 | |||||||||||
Net cash outflow from financing activities |
(5,571 | ) | (2,774 | ) | (4,908 | ) | ||||||||||
(Decrease)/increase in cash and bank overdrafts |
37 | (642 | ) | 1,054 | 1,148 | |||||||||||
Exchange adjustments |
81 | (158 | ) | 1,103 | ||||||||||||
Cash and bank overdrafts at beginning of year |
6,368 | 5,472 | 3,221 | |||||||||||||
Cash and bank overdrafts at end of year |
5,807 | 6,368 | 5,472 | |||||||||||||
Cash and bank overdrafts at end of year comprise: |
||||||||||||||||
Cash and cash equivalents |
6,057 | 6,545 | 5,623 | |||||||||||||
Overdrafts |
(250 | ) | (177 | ) | (151 | ) | ||||||||||
| 5,807 | 6,368 | 5,472 | ||||||||||||||
Table of Contents
109
| • | Consolidated income statement | |
| • | Consolidated statement of comprehensive income | |
| • | Consolidated balance sheet | |
| • | Consolidated statement of changes in equity | |
| • | Consolidated cash flow statement | |
| • | Notes to the financial statements. |
Table of Contents
110
| • | the assets and liabilities, and the results and cash flows, of the company and its subsidiaries, including ESOP Trusts |
| • | the Group’s share of the results and net assets of associates and joint ventures. |
Table of Contents
111
Table of Contents
112
Freehold buildings
|
20 to 50 years | |
Leasehold land and buildings
|
Lease term or 20 to 50 years | |
Plant and machinery
|
10 to 20 years | |
Fixtures and equipment
|
3 to 10 years | |
Table of Contents
113
Table of Contents
114
Table of Contents
115
Table of Contents
116
| 2010 | 2009 | 2008 | ||||||||||
Average rates: |
||||||||||||
£/US$ |
1.55 | 1.56 | 1.85 | |||||||||
£/Euro |
1.16 | 1.12 | 1.26 | |||||||||
£/Yen |
136 | 146 | 192 | |||||||||
Period end rates: |
||||||||||||
£/US$ |
1.56 | 1.61 | 1.44 | |||||||||
£/Euro |
1.17 | 1.13 | 1.04 | |||||||||
£/Yen |
127 | 150 | 131 | |||||||||
Table of Contents
117
| Turnover by segment | 2009 | 2008 | ||||||||||
| 2010 | (restated) | (restated) | ||||||||||
| £m | £m | £m | ||||||||||
US pharmaceuticals |
7,648 | 8,578 | 8,254 | |||||||||
Europe pharmaceuticals |
6,548 | 7,087 | 5,847 | |||||||||
Emerging Markets pharmaceuticals |
3,556 | 2,895 | 2,177 | |||||||||
Asia Pacific/Japan pharmaceuticals |
3,102 | 2,628 | 1,848 | |||||||||
ViiV Healthcare |
1,566 | 1,605 | 1,513 | |||||||||
Other trading and unallocated pharmaceuticals |
962 | 901 | 742 | |||||||||
Pharmaceuticals turnover |
23,382 | 23,694 | 20,381 | |||||||||
Consumer Healthcare turnover |
5,010 | 4,674 | 3,971 | |||||||||
| 28,392 | 28,368 | 24,352 | ||||||||||
| Pharmaceutical turnover by therapeutic area | 2009 | 2008 | ||||||||||
| 2010 | (restated) | (restated) | ||||||||||
| £m | £m | £m | ||||||||||
Respiratory |
7,238 | 6,977 | 5,817 | |||||||||
Anti-virals |
1,086 | 2,416 | 1,584 | |||||||||
Central nervous system |
1,753 | 1,870 | 2,897 | |||||||||
Cardiovascular and urogenital |
2,570 | 2,298 | 1,847 | |||||||||
Metabolic |
678 | 1,181 | 1,191 | |||||||||
Anti-bacterials |
1,396 | 1,457 | 1,301 | |||||||||
Oncology and emesis |
688 | 629 | 496 | |||||||||
Vaccines |
4,326 | 3,706 | 2,539 | |||||||||
Dermatologicals |
1,087 | 707 | 414 | |||||||||
ViiV Healthcare (HIV) |
1,566 | 1,605 | 1,513 | |||||||||
Other |
994 | 848 | 782 | |||||||||
| 23,382 | 23,694 | 20,381 | ||||||||||
| Consumer Healthcare turnover by category | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
OTC medicines |
2,456 | 2,339 | 1,935 | |||||||||
Oral healthcare |
1,602 | 1,484 | 1,240 | |||||||||
Nutritional healthcare |
952 | 851 | 796 | |||||||||
| 5,010 | 4,674 | 3,971 | ||||||||||
Table of Contents
118
| Segment profit | 2009 | 2008 | ||||||||||
| 2010 | (restated) | (restated) | ||||||||||
| £m | £m | £m | ||||||||||
US Pharmaceuticals |
5,043 | 5,933 | 5,461 | |||||||||
Europe Pharmaceuticals |
3,744 | 3,993 | 3,229 | |||||||||
Emerging Markets pharmaceuticals |
1,271 | 948 | 837 | |||||||||
Asia Pacific/Japan pharmaceuticals |
1,730 | 1,352 | 1,016 | |||||||||
ViiV Healthcare |
851 | 1,071 | 1,005 | |||||||||
Pharmaceuticals R&D |
(3,105 | ) | (3,082 | ) | (2,840 | ) | ||||||
Other trading and unallocated pharmaceuticals costs |
(783 | ) | (705 | ) | (110 | ) | ||||||
Pharmaceuticals operating profit |
8,751 | 9,510 | 8,598 | |||||||||
Consumer Healthcare operating profit |
1,043 | 931 | 881 | |||||||||
Segment profit |
9,794 | 10,441 | 9,479 | |||||||||
Corporate and other unallocated costs and disposal profits |
(4,666 | ) | (1,184 | ) | (1,220 | ) | ||||||
Operating profit before major restructuring |
5,128 | 9,257 | 8,259 | |||||||||
Major restructuring |
(1,345 | ) | (832 | ) | (1,118 | ) | ||||||
Total operating profit |
3,783 | 8,425 | 7,141 | |||||||||
Finance income |
116 | 70 | 313 | |||||||||
Finance costs |
(831 | ) | (783 | ) | (843 | ) | ||||||
Profit on disposal of interest in associate |
8 | 115 | – | |||||||||
Share of after tax profits of associates and joint ventures |
81 | 64 | 48 | |||||||||
Profit before taxation |
3,157 | 7,891 | 6,659 | |||||||||
Taxation |
(1,304 | ) | (2,222 | ) | (1,947 | ) | ||||||
Profit after taxation for the year |
1,853 | 5,669 | 4,712 | |||||||||
| Depreciation and amortisation by segment | 2009 | 2008 | ||||||||||
| 2010 | (restated) | (restated) | ||||||||||
| £m | £m | £m | ||||||||||
US pharmaceuticals |
101 | 116 | 110 | |||||||||
Europe pharmaceuticals |
31 | 24 | 36 | |||||||||
Emerging Markets pharmaceuticals |
51 | 29 | 22 | |||||||||
Asia Pacific/Japan pharmaceuticals |
25 | 15 | 11 | |||||||||
ViiV Healthcare |
29 | 5 | – | |||||||||
Pharmaceuticals R&D |
262 | 280 | 267 | |||||||||
Other trading and unallocated pharmaceuticals |
809 | 789 | 580 | |||||||||
Pharmaceuticals depreciation and amortisation |
1,308 | 1,258 | 1,026 | |||||||||
Consumer Healthcare depreciation and amortisation |
66 | 63 | 52 | |||||||||
Segment depreciation and amortisation |
1,374 | 1,321 | 1,078 | |||||||||
Corporate and other unallocated depreciation and amortisation |
85 | 80 | 76 | |||||||||
Depreciation and amortisation before major restructuring |
1,459 | 1,401 | 1,154 | |||||||||
Major restructuring |
220 | 161 | 77 | |||||||||
Total depreciation and amortisation |
1,679 | 1,562 | 1,231 | |||||||||
Table of Contents
119
| PP&E, intangible asset and goodwill impairment by segment | 2009 | 2008 | ||||||||||
| 2010 | (restated) | (restated) | ||||||||||
| £m | £m | £m | ||||||||||
US pharmaceuticals |
– | 1 | 1 | |||||||||
Europe pharmaceuticals |
1 | 7 | 2 | |||||||||
Emerging Markets pharmaceuticals |
1 | – | – | |||||||||
Asia Pacific/Japan pharmaceuticals |
2 | 1 | 2 | |||||||||
ViiV Healthcare |
– | – | – | |||||||||
Pharmaceuticals R&D |
134 | 118 | 107 | |||||||||
Other trading and unallocated pharmaceuticals |
129 | 124 | 30 | |||||||||
Pharmaceuticals impairment |
267 | 251 | 142 | |||||||||
Consumer Healthcare impairment |
5 | 1 | – | |||||||||
Segment impairment |
272 | 252 | 142 | |||||||||
Corporate and other unallocated impairment |
4 | 23 | 52 | |||||||||
Impairment before major restructuring |
276 | 275 | 194 | |||||||||
Major restructuring |
89 | 57 | 197 | |||||||||
Total impairment |
365 | 332 | 391 | |||||||||
| PP&E, intangible asset and goodwill impairment reversals by segment | 2009 | 2008 | ||||||||||
| 2010 | (restated) | (restated) | ||||||||||
| £m | £m | £m | ||||||||||
US pharmaceuticals |
– | (1 | ) | – | ||||||||
Europe pharmaceuticals |
– | – | – | |||||||||
Emerging Markets pharmaceuticals |
– | – | – | |||||||||
Asia Pacific/Japan pharmaceuticals |
– | – | – | |||||||||
ViiV Healthcare |
– | – | – | |||||||||
Pharmaceuticals R&D |
(1 | ) | (1 | ) | (10 | ) | ||||||
Other trading and unallocated pharmaceuticals |
(4 | ) | (9 | ) | – | |||||||
Pharmaceuticals impairment reversals |
(5 | ) | (11 | ) | (10 | ) | ||||||
Consumer Healthcare impairment reversals |
– | – | – | |||||||||
Segment impairment reversals |
(5 | ) | (11 | ) | (10 | ) | ||||||
Corporate and other unallocated impairment reversals |
– | – | (10 | ) | ||||||||
Impairment reversals before major restructuring |
(5 | ) | (11 | ) | (20 | ) | ||||||
Major restructuring |
(14 | ) | – | – | ||||||||
Total impairment reversals |
(19 | ) | (11 | ) | (20 | ) | ||||||
Table of Contents
120
| Net assets by segment | 2009 | |||||||||
| 2010 | (restated) | |||||||||
| £m | £m | |||||||||
US pharmaceuticals |
616 | 1,049 | ||||||||
Europe pharmaceuticals |
1,031 | 1,567 | ||||||||
Emerging Markets pharmaceuticals |
1,840 | 1,508 | ||||||||
Asia Pacific/Japan pharmaceuticals |
1,057 | 982 | ||||||||
ViiV Healthcare |
832 | 835 | ||||||||
Pharmaceuticals R&D |
1,656 | 2,278 | ||||||||
Other trading and unallocated pharmaceuticals |
13,320 | 13,037 | ||||||||
Pharmaceuticals net operating assets |
20,352 | 21,256 | ||||||||
Consumer Healthcare net operating assets |
2,972 | 2,990 | ||||||||
Segment net operating assets |
23,324 | 24,246 | ||||||||
Corporate and other unallocated operating net assets |
(6,682 | ) | (5,334 | ) | ||||||
Net operating assets |
16,642 | 18,912 | ||||||||
Net debt |
(8,859 | ) | (9,444 | ) | ||||||
Investments in associates and joint ventures |
1,081 | 895 | ||||||||
Derivative financial instruments |
(3 | ) | 29 | |||||||
Current and deferred taxation |
868 | 336 | ||||||||
Assets held for sale |
16 | 14 | ||||||||
Net assets |
9,745 | 10,742 | ||||||||
| Turnover by location of customer | 2009 | 2008 | ||||||||||
| 2010 | (restated) | (restated) | ||||||||||
| £m | £m | £m | ||||||||||
UK |
1,820 | 1,864 | 1,636 | |||||||||
USA |
9,345 | 10,315 | 9,746 | |||||||||
Rest of World |
17,227 | 16,189 | 12,970 | |||||||||
External turnover |
28,392 | 28,368 | 24,352 | |||||||||
| Turnover by location of subsidiary | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
UK |
4,965 | 4,469 | 3,096 | |||||||||
USA |
13,072 | 13,711 | 12,925 | |||||||||
Rest of World |
21,220 | 19,661 | 15,977 | |||||||||
Turnover including inter-segment turnover |
39,257 | 37,841 | 31,998 | |||||||||
UK |
2,032 | 1,556 | 1,042 | |||||||||
USA |
3,717 | 3,395 | 3,114 | |||||||||
Rest of World |
5,116 | 4,522 | 3,490 | |||||||||
Inter-segment turnover |
10,865 | 9,473 | 7,646 | |||||||||
UK |
2,933 | 2,913 | 2,054 | |||||||||
USA |
9,355 | 10,316 | 9,811 | |||||||||
Rest of World |
16,104 | 15,139 | 12,487 | |||||||||
External turnover |
28,392 | 28,368 | 24,352 | |||||||||
Table of Contents
121
| Operating profit by location | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
UK |
1,033 | 2,608 | 1,861 | |||||||||
USA |
420 | 2,337 | 1,951 | |||||||||
Rest of World |
2,330 | 3,480 | 3,329 | |||||||||
Total operating profit |
3,783 | 8,425 | 7,141 | |||||||||
| Net operating assets by location | 2009 | |||||||||||
| 2010 | (restated) | |||||||||||
| £m | £m | |||||||||||
UK |
3,177 | 4,540 | ||||||||||
USA |
4,235 | 3,168 | ||||||||||
Rest of World |
9,230 | 11,204 | ||||||||||
Net operating assets |
16,642 | 18,912 | ||||||||||
| Non-current assets by location | 2009 | |||||||||||
| 2010 | (restated) | |||||||||||
| £m | £m | |||||||||||
UK |
5,066 | 5,270 | ||||||||||
USA |
6,972 | 6,863 | ||||||||||
Rest of World |
10,372 | 9,847 | ||||||||||
Non-current assets |
22,410 | 21,980 | ||||||||||
| • | cost saving projects in R&D, focused primarily on the simplification and streamlining of support infrastructure, including some site rationalisations, principally Verona in Italy and Harlow and Tonbridge in the UK; | |
| • | the adoption of more customised sales approaches, leading to staff reductions in a number of sales forces, principally in the USA, France and Italy; | |
| • | the closure of a number of manufacturing sites, including Dartford and Crawley in the UK, giving rise to asset write-downs and staff reductions; and | |
| • | projects to simplify or eliminate processes, leading to staff reductions in administrative and support functions. |
Table of Contents
122
| 2010 | Asset | Staff | Other | |||||||||||||
| impairment | reductions | costs | Total | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Cost of sales |
(14 | ) | (58 | ) | (115 | ) | (187 | ) | ||||||||
Selling, general and administration |
(17 | ) | (503 | ) | (145 | ) | (665 | ) | ||||||||
Research and development |
(44 | ) | (117 | ) | (332 | ) | (493 | ) | ||||||||
Effect on operating profit |
(75 | ) | (678 | ) | (592 | ) | (1,345 | ) | ||||||||
Net finance expense |
(3 | ) | ||||||||||||||
Effect on profit before taxation |
(1,348 | ) | ||||||||||||||
Effect on taxation |
240 | |||||||||||||||
Effect on earnings |
(1,108 | ) | ||||||||||||||
| 2009 | Asset | Staff | Other | |||||||||||||
| impairment | reductions | costs | Total | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Cost of sales |
(41 | ) | (112 | ) | (132 | ) | (285 | ) | ||||||||
Selling, general and administration |
(1 | ) | (337 | ) | (54 | ) | (392 | ) | ||||||||
Research and development |
(15 | ) | (68 | ) | (72 | ) | (155 | ) | ||||||||
Effect on operating profit |
(57 | ) | (517 | ) | (258 | ) | (832 | ) | ||||||||
Net finance expense |
(3 | ) | ||||||||||||||
Effect on profit before taxation |
(835 | ) | ||||||||||||||
Effect on taxation |
221 | |||||||||||||||
Effect on earnings |
(614 | ) | ||||||||||||||
| 2008 | Asset | Staff | Other | |||||||||||||
| impairment | reductions | costs | Total | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Cost of sales |
(181 | ) | (370 | ) | (88 | ) | (639 | ) | ||||||||
Selling, general and administration |
(2 | ) | (177 | ) | (125 | ) | (304 | ) | ||||||||
Research and development |
(14 | ) | (143 | ) | (18 | ) | (175 | ) | ||||||||
Effect on operating profit |
(197 | ) | (690 | ) | (231 | ) | (1,118 | ) | ||||||||
Net finance expense |
(5 | ) | ||||||||||||||
Effect on profit before taxation |
(1,123 | ) | ||||||||||||||
Effect on taxation |
284 | |||||||||||||||
Effect on earnings |
(839 | ) | ||||||||||||||
Table of Contents
123
| The costs of the major restructuring programmes have arisen as follows: | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
Increase in provision for major restructuring programmes (see Note 29) |
(837 | ) | (487 | ) | (740 | ) | ||||||
Amount of provision reversed unused (see Note 29) |
40 | 15 | 7 | |||||||||
Impairments losses recognised |
(75 | ) | (57 | ) | (197 | ) | ||||||
Foreign exchange gain/(loss) recognised on liquidation of subsidiary |
– | 44 | (84 | ) | ||||||||
Other non-cash charges |
(240 | ) | (168 | ) | (53 | ) | ||||||
Other cash costs |
(233 | ) | (179 | ) | (51 | ) | ||||||
Net finance expense |
(3 | ) | (3 | ) | (5 | ) | ||||||
Effect on profit before taxation |
(1,348 | ) | (835 | ) | (1,123 | ) | ||||||
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Royalty income |
296 | 296 | 307 | |||||||||
Milestone income |
7 | 90 | 11 | |||||||||
Impairment of equity investments |
(65 | ) | (135 | ) | (63 | ) | ||||||
Disposal of equity investments |
17 | 40 | 33 | |||||||||
Disposal of
other assets, asset rights and legal settlements |
227 | 539 | 260 | |||||||||
Gain recognised on creation of ViiV Healthcare |
– | 296 | – | |||||||||
Fair value movements on derivative financial instruments |
(6 | ) | (5 | ) | (10 | ) | ||||||
Other income |
17 | 14 | 3 | |||||||||
| 493 | 1,135 | 541 | ||||||||||
Table of Contents
124
| The following items have been included in operating profit: | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
Employee costs (Note 10) |
6,994 | 7,167 | 6,524 | |||||||||
Advertising |
971 | 923 | 805 | |||||||||
Distribution costs |
413 | 363 | 310 | |||||||||
Depreciation of property, plant and equipment |
1,146 | 1,130 | 920 | |||||||||
Impairment of property, plant and equipment, net of reversals |
186 | 149 | 256 | |||||||||
Amortisation of intangible assets |
533 | 432 | 311 | |||||||||
Impairment of intangible assets and goodwill, net of reversals in 2008 |
160 | 172 | 115 | |||||||||
Net foreign exchange losses/(gains) |
60 | 163 | (145 | ) | ||||||||
Inventories: |
||||||||||||
Cost of inventories included in cost of sales |
7,014 | 6,743 | 5,734 | |||||||||
Write-down of inventories |
305 | 276 | 298 | |||||||||
Reversal of prior year write-down of inventories |
(66 | ) | (116 | ) | (118 | ) | ||||||
Operating lease rentals: |
||||||||||||
Minimum lease payments |
136 | 160 | 143 | |||||||||
Contingent rents |
14 | 13 | 15 | |||||||||
Sub-lease payments |
7 | 6 | 1 | |||||||||
Fees payable to the company’s auditor and its associates in relation to the Group (see below) |
22.2 | 24.1 | 19.2 | |||||||||
| Fees payable to the company's auditor and its associates | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
Audit of parent company and consolidated financial statements |
2.0 | 2.0 | 1.6 | |||||||||
Audit of accounts of the Group’s UK and overseas subsidiaries, pursuant to legislation |
11.2 | 10.2 | 9.3 | |||||||||
Other
assurance services, pursuant to legislation, including attestation under
s.404 of Sarbanes-Oxley Act 2002 |
3.3 | 3.0 | 2.9 | |||||||||
Audit and assurance services |
16.5 | 15.2 | 13.8 | |||||||||
Other tax services |
2.5 | 7.3 | 2.5 | |||||||||
All other services, including regulatory, compliance and treasury related services |
3.2 | 1.6 | 2.9 | |||||||||
| 22.2 | 24.1 | 19.2 | ||||||||||
| In addition to the above, fees paid in respect of the GSK pension schemes were: | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
Audit |
0.4 | 0.4 | 0.4 | |||||||||
Other services |
– | – | – | |||||||||
Table of Contents
125
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Wages and salaries |
5,079 | 5,387 | 4,640 | |||||||||
Social security costs |
600 | 661 | 653 | |||||||||
Pension and other post-employment costs, including augmentations (Note 28) |
554 | 491 | 505 | |||||||||
Cost of share-based incentive plans |
179 | 179 | 241 | |||||||||
Severance and other costs from integration and restructuring activities |
582 | 449 | 485 | |||||||||
| 6,994 | 7,167 | 6,524 | ||||||||||
| The average number of persons employed by the Group (including Directors) during the year: | 2010 | 2009 | 2008 | |||||||||
| Number | Number | Number | ||||||||||
Manufacturing |
30,883 | 31,467 | 33,372 | |||||||||
Selling, general and administration |
53,778 | 53,183 | 52,115 | |||||||||
Research and development |
13,824 | 14,204 | 15,646 | |||||||||
| 98,485 | 98,854 | 101,133 | ||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Wages and salaries |
20 | 23 | 17 | |||||||||
Social security costs |
2 | 1 | 1 | |||||||||
Pension and other post-employment costs |
3 | 3 | 3 | |||||||||
Cost of share-based incentive plans |
11 | 4 | 12 | |||||||||
| 36 | 31 | 33 | ||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Interest income arising from: |
||||||||||||
cash and cash equivalents |
58 | 46 | 107 | |||||||||
available-for-sale investments |
8 | 15 | 31 | |||||||||
derivatives at fair value through profit or loss |
24 | (5 | ) | 159 | ||||||||
loans and receivables |
12 | 11 | 22 | |||||||||
Realised gains on liquid investments |
– | – | 2 | |||||||||
Fair value movements on derivatives at fair value through profit or loss |
13 | (3 | ) | 4 | ||||||||
Net investment hedge ineffectiveness |
– | 4 | (13 | ) | ||||||||
Unwinding of discounts on assets |
1 | 2 | 1 | |||||||||
| 116 | 70 | 313 | ||||||||||
Table of Contents
126
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Interest expense arising on: |
||||||||||||
financial liabilities at amortised cost |
(767 | ) | (790 | ) | (664 | ) | ||||||
derivatives at fair value through profit or loss |
– | 20 | (165 | ) | ||||||||
Fair value hedges: |
||||||||||||
fair value movements on derivatives designated as hedging instruments |
26 | (37 | ) | 92 | ||||||||
fair value adjustments on hedged items |
(27 | ) | 38 | (90 | ) | |||||||
Fair value movements on other derivatives at fair value through profit or loss |
(16 | ) | (2 | ) | – | |||||||
Reclassification of cash flow hedge from other comprehensive income |
(3 | ) | (1 | ) | – | |||||||
Unwinding of discounts on provisions |
(18 | ) | (11 | ) | (16 | ) | ||||||
Net investment hedge ineffectiveness |
(1 | ) | – | – | ||||||||
Other finance expense |
(25 | ) | – | – | ||||||||
| (831 | ) | (783 | ) | (843 | ) | |||||||
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Associates: |
||||||||||||
Share of after tax profits of Quest Diagnostics Inc. |
79 | 73 | 47 | |||||||||
Share of after tax profits of Aspen Pharmacare Holdings Limited |
32 | 2 | – | |||||||||
Share of after tax losses of other associates |
(7 | ) | (3 | ) | (3 | ) | ||||||
| 104 | 72 | 44 | ||||||||||
Share of after tax (losses)/profits of joint ventures |
(23 | ) | (8 | ) | 4 | |||||||
| 81 | 64 | 48 | ||||||||||
Share of turnover of joint ventures |
18 | 13 | 13 | |||||||||
Sales to joint ventures and associates |
90 | 26 | 9 | |||||||||
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Total turnover: |
||||||||||||
Quest Diagnostics Inc. |
4,754 | 4,779 | 3,919 | |||||||||
Aspen Pharmacare Holdings Limited |
1,171 | 67 | – | |||||||||
Others |
65 | 7 | 3 | |||||||||
| 5,990 | 4,853 | 3,922 | ||||||||||
Total profit: |
||||||||||||
Quest Diagnostics Inc. |
465 | 467 | 314 | |||||||||
Aspen Pharmacare Holdings Limited |
233 | 12 | – | |||||||||
Others |
(23 | ) | (14 | ) | (7 | ) | ||||||
| 675 | 465 | 307 | ||||||||||
Table of Contents
127
| Taxation charge based on profits for the year | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
UK corporation tax at the UK statutory rate |
82 | 600 | 2,213 | |||||||||
Less double taxation relief |
(156 | ) | (183 | ) | (1,924 | ) | ||||||
| (74 | ) | 417 | 289 | |||||||||
Overseas taxation |
1,496 | 1,997 | 1,589 | |||||||||
Current taxation |
1,422 | 2,414 | 1,878 | |||||||||
Deferred taxation |
(118 | ) | (192 | ) | 69 | |||||||
| 1,304 | 2,222 | 1,947 | ||||||||||
| Reconciliation of the taxation rate on Group profits | 2010 | 2009 | 2008 | |||||||||
| % | % | % | ||||||||||
UK statutory rate of taxation |
28.0 | 28.0 | 28.5 | |||||||||
Differences in overseas taxation rates |
8.1 | 3.5 | 1.9 | |||||||||
Benefit of special tax status |
(2.6 | ) | (1.8 | ) | (2.4 | ) | ||||||
R&D credits |
(3.7 | ) | (1.9 | ) | (1.3 | ) | ||||||
Inter-company stock profit |
1.7 | 0.5 | 2.1 | |||||||||
Impact of share based payments |
1.4 | 0.1 | 0.7 | |||||||||
Tax on profit of associates |
(1.2 | ) | (0.2 | ) | (0.4 | ) | ||||||
Losses for which no benefit is recognised |
5.5 | 0.6 | 0.0 | |||||||||
Other permanent differences |
6.2 | (0.9 | ) | 1.2 | ||||||||
Prior year items |
(6.5 | ) | 0.1 | (1.6 | ) | |||||||
Restructuring |
4.4 | 0.2 | 0.5 | |||||||||
Tax rate |
41.3 | 28.2 | 29.2 | |||||||||
| Tax on items charged to equity and statement of comprehensive income | 2010 | 2009 | 2008 | |||||||||
| £m | £m | £m | ||||||||||
Current taxation |
||||||||||||
Share based payments |
– | 1 | 4 | |||||||||
Foreign exchange movements |
– | 19 | 15 | |||||||||
| – | 20 | 19 | ||||||||||
Deferred taxation |
||||||||||||
Share based payments |
2 | 13 | (5 | ) | ||||||||
Defined benefit plans |
1 | 183 | 441 | |||||||||
Fair value movements on cash flow hedges |
1 | 2 | (3 | ) | ||||||||
Fair value movements on available-for-sale investments |
(28 | ) | (11 | ) | 8 | |||||||
| (24 | ) | 187 | 441 | |||||||||
Total (charge)/credit to equity and statement of comprehensive income |
(24 | ) | 207 | 460 | ||||||||
Table of Contents
128
| Pensions & | Manu- | Share | Other | |||||||||||||||||||||||||||||||||||||||||||||
| Accelerated | Intra- | other post | Legal | facturing | Stock | option | net | Offset | ||||||||||||||||||||||||||||||||||||||||
| capital | group | employment | Tax | & other | restruct- | valuation | and award | temporary | within | |||||||||||||||||||||||||||||||||||||||
| allowances | Intangibles | profit | benefits | losses | disputes | uring | adjustments | schemes | differences | countries | Total | |||||||||||||||||||||||||||||||||||||
| £m | £m | £m | £m | £m | £m | £m | £m | £m | £m | £m | £m | |||||||||||||||||||||||||||||||||||||
Deferred tax assets at 1st January 2010 |
24 | 177 | 1,183 | 1,043 | 211 | 303 | 157 | 30 | 126 | 822 | (1,702 | ) | 2,374 | |||||||||||||||||||||||||||||||||||
Deferred tax liabilities at 1st January 2010 |
(628 | ) | (1,476 | ) | – | – | (28 | ) | – | – | (198 | ) | – | (17 | ) | 1,702 | (645 | ) | ||||||||||||||||||||||||||||||
At 1st January 2010 |
(604 | ) | (1,299 | ) | 1,183 | 1,043 | 183 | 303 | 157 | (168 | ) | 126 | 805 | – | 1,729 | |||||||||||||||||||||||||||||||||
Exchange adjustments |
(5 | ) | (10 | ) | 70 | 15 | 3 | 7 | 2 | (2 | ) | – | 31 | – | 111 | |||||||||||||||||||||||||||||||||
Credit/(charge) to income statement |
146 | 10 | (126 | ) | (36 | ) | (88 | ) | 115 | (42 | ) | 148 | (35 | ) | 26 | – | 118 | |||||||||||||||||||||||||||||||
Credit to equity |
– | – | – | – | – | – | – | – | 2 | – | – | 2 | ||||||||||||||||||||||||||||||||||||
Credit/(charge) to statement of comprehensive
income |
– | – | – | 1 | – | – | – | – | – | (27 | ) | – | (26 | ) | ||||||||||||||||||||||||||||||||||
Acquisitions |
– | (40 | ) | – | – | – | – | – | – | – | (35 | ) | – | (75 | ) | |||||||||||||||||||||||||||||||||
At 31st December 2010 |
(463 | ) | (1,339 | ) | 1,127 | 1,023 | 98 | 425 | 117 | (22 | ) | 93 | 800 | – | 1,859 | |||||||||||||||||||||||||||||||||
Deferred tax assets at 31st December 2010 |
49 | 224 | 1,127 | 1,023 | 98 | 425 | 117 | 29 | 93 | 914 | (1,533 | ) | 2,566 | |||||||||||||||||||||||||||||||||||
Deferred tax liabilities at 31st December 2010 |
(512 | ) | (1,563 | ) | – | – | – | – | – | (51 | ) | – | (114 | ) | 1,533 | (707 | ) | |||||||||||||||||||||||||||||||
| (463 | ) | (1,339 | ) | 1,127 | 1,023 | 98 | 425 | 117 | (22 | ) | 93 | 800 | – | 1,859 | ||||||||||||||||||||||||||||||||||
Table of Contents
129
| Tax losses | Recognised | Unrecognised | ||||||||||||||
| 2010 | 2009 | 2010 | 2009 | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Trading losses expiring: |
||||||||||||||||
Within 10 years |
163 | 76 | 14 | 34 | ||||||||||||
In more than 10 years |
329 | 445 | 529 | 159 | ||||||||||||
Available indefinitely |
1 | 96 | 5,302 | 4,204 | ||||||||||||
At 31st December |
493 | 617 | 5,845 | 4,397 | ||||||||||||
Deferred tax asset |
98 | 183 | – | – | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| pence | pence | pence | ||||||||||
Basic earnings per share |
32.1 | 109.1 | 88.6 | |||||||||
Adjustment for major restructuring |
21.8 | 12.1 | 16.1 | |||||||||
Basic earnings per share before major restructuring |
53.9 | 121.2 | 104.7 | |||||||||
Diluted earnings per share |
31.9 | 108.2 | 88.1 | |||||||||
Adjustment for major restructuring |
21.6 | 12.1 | 16.0 | |||||||||
Diluted earnings per share before major restructuring |
53.5 | 120.3 | 104.1 | |||||||||
| Weighted average number of shares in issue | 2010 | 2009 | 2008 | |||||||||
| millions | millions | millions | ||||||||||
Basic |
5,085 | 5,069 | 5,195 | |||||||||
Dilution for share options |
43 | 39 | 31 | |||||||||
Diluted |
5,128 | 5,108 | 5,226 | |||||||||
Table of Contents
130
| 2010 | First interim | Second interim | Third interim | Fourth interim | Total | |||||||||||||||
Total dividend (£m) |
764 | 759 | 816 | 967 | 3,306 | |||||||||||||||
Dividend per share (pence) |
15 | 15 | 16 | 19 | 65 | |||||||||||||||
Paid/payable |
8th July 2010 | 7th October 2010 | 6th January 2011 | 7th April 2011 | ||||||||||||||||
2009 |
||||||||||||||||||||
Total dividend (£m) |
701 | 713 | 763 | 919 | 3,096 | |||||||||||||||
Dividend per share (pence) |
14 | 14 | 15 | 18 | 61 | |||||||||||||||
Paid |
9th July 2009 | 8th October 2009 | 7th January 2010 | 8th April 2010 | ||||||||||||||||
2008 |
||||||||||||||||||||
Total dividend (£m) |
683 | 679 | 730 | 859 | 2,951 | |||||||||||||||
Dividend per share (pence) |
13 | 13 | 14 | 17 | 57 | |||||||||||||||
Paid |
10th July 2008 | 9th October 2008 | 8th January 2009 | 9th April 2009 | ||||||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Dividends to shareholders |
3,205 | 3,003 | 2,929 | |||||||||
| Plant, | ||||||||||||||||
| Land and | equipment | Assets in | ||||||||||||||
| buildings | and vehicles | construction | Total | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Cost at 1st January 2009 |
5,979 | 10,686 | 2,322 | 18,987 | ||||||||||||
Exchange adjustments |
(343 | ) | (493 | ) | (154 | ) | (990 | ) | ||||||||
Additions |
188 | 432 | 803 | 1,423 | ||||||||||||
Additions through business combinations |
67 | 76 | 8 | 151 | ||||||||||||
Capitalised borrowing costs |
– | – | 1 | 1 | ||||||||||||
Disposals and write-offs |
(184 | ) | (614 | ) | (5 | ) | (803 | ) | ||||||||
Reclassifications |
309 | 430 | (735 | ) | 4 | |||||||||||
Transfer to assets held for sale |
(14 | ) | (2 | ) | – | (16 | ) | |||||||||
Cost at 31st December 2009 |
6,002 | 10,515 | 2,240 | 18,757 | ||||||||||||
Exchange adjustments |
80 | 60 | (7 | ) | 133 | |||||||||||
Additions |
75 | 293 | 670 | 1,038 | ||||||||||||
Additions through business combinations |
20 | 7 | – | 27 | ||||||||||||
Capitalised borrowing costs |
– | – | 6 | 6 | ||||||||||||
Disposals and write-offs |
(111 | ) | (661 | ) | (2 | ) | (774 | ) | ||||||||
Reclassifications |
223 | 432 | (671 | ) | (16 | ) | ||||||||||
Transfer to assets held for sale |
(171 | ) | (105 | ) | – | (276 | ) | |||||||||
Cost at 31st December 2010 |
6,118 | 10,541 | 2,236 | 18,895 | ||||||||||||
Table of Contents
131
| Plant, | ||||||||||||||||
| Land and | equipment | Assets in | ||||||||||||||
| buildings | and vehicles | construction | Total | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Depreciation at 1st January 2009 |
(2,062 | ) | (6,630 | ) | – | (8,692 | ) | |||||||||
Exchange adjustments |
128 | 312 | – | 440 | ||||||||||||
Charge for the year |
(283 | ) | (847 | ) | – | (1,130 | ) | |||||||||
Disposals and write-offs |
129 | 478 | – | 607 | ||||||||||||
Transfer to assets held for sale |
1 | 1 | – | 2 | ||||||||||||
Depreciation at 31st December 2009 |
(2,087 | ) | (6,686 | ) | – | (8,773 | ) | |||||||||
Exchange adjustments |
(39 | ) | (51 | ) | – | (90 | ) | |||||||||
Charge for the year |
(321 | ) | (825 | ) | – | (1,146 | ) | |||||||||
Disposals and write-offs |
11 | 508 | – | 519 | ||||||||||||
Transfer to assets held for sale |
147 | 95 | – | 242 | ||||||||||||
Depreciation at 31st December 2010 |
(2,289 | ) | (6,959 | ) | – | (9,248 | ) | |||||||||
Impairment at 1st January 2009 |
(161 | ) | (412 | ) | (44 | ) | (617 | ) | ||||||||
Exchange adjustments |
6 | 10 | 4 | 20 | ||||||||||||
Disposals and write-offs |
28 | 104 | 4 | 136 | ||||||||||||
Impairment losses |
(27 | ) | (108 | ) | (25 | ) | (160 | ) | ||||||||
Reversal of impairments |
1 | 10 | – | 11 | ||||||||||||
Impairment at 31st December 2009 |
(153 | ) | (396 | ) | (61 | ) | (610 | ) | ||||||||
Exchange adjustments |
– | 2 | (1 | ) | 1 | |||||||||||
Disposals and write-offs |
64 | 111 | – | 175 | ||||||||||||
Impairment losses |
(43 | ) | (160 | ) | (2 | ) | (205 | ) | ||||||||
Reversal of impairments |
14 | 5 | – | 19 | ||||||||||||
Transfer to assets held for sale |
18 | – | – | 18 | ||||||||||||
Impairment at 31st December 2010 |
(100 | ) | (438 | ) | (64 | ) | (602 | ) | ||||||||
Total depreciation and impairment at 31st December 2009 |
(2,240 | ) | (7,082 | ) | (61 | ) | (9,383 | ) | ||||||||
Total depreciation and impairment at 31st December 2010 |
(2,389 | ) | (7,397 | ) | (64 | ) | (9,850 | ) | ||||||||
Net book value at 1st January 2009 |
3,756 | 3,644 | 2,278 | 9,678 | ||||||||||||
Net book value at 31st December 2009 |
3,762 | 3,433 | 2,179 | 9,374 | ||||||||||||
Net book value at 31st December 2010 |
3,729 | 3,144 | 2,172 | 9,045 | ||||||||||||
Table of Contents
132
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Cost at 1st January |
3,361 | 2,101 | ||||||
Exchange adjustments |
95 | (116 | ) | |||||
Additions through business combinations |
160 | 1,376 | ||||||
Impairment losses |
(10 | ) | – | |||||
Cost at 31st December |
3,606 | 3,361 | ||||||
Net book value at 1st January |
3,361 | 2,101 | ||||||
Net book value at 31st December |
3,606 | 3,361 | ||||||
| 2010 | 2009 | |||||||||||
| Cash generating unit | £m | £m | ||||||||||
Stiefel Laboratories, Inc. |
US, Europe, Emerging Markets, Asia Pacific, Other pharmaceuticals | 894 | 901 | |||||||||
ID Biomedical Corporation |
US, Europe, Emerging Markets, Asia Pacific, Japan, Other pharmaceuticals | 464 | 426 | |||||||||
Reliant Pharmaceuticals, Inc. |
US pharmaceuticals | 448 | 434 | |||||||||
Sirtris Pharmaceuticals, Inc. |
US, Europe, Emerging Markets, Asia Pacific, Japan, Other pharmaceuticals | 304 | 294 | |||||||||
Pfizer HIV business |
ViiV Healthcare | 255 | 255 | |||||||||
GlaxoSmithKline K.K. |
Japan pharmaceuticals | 246 | 208 | |||||||||
Domantis Limited |
US, Europe, Emerging Markets, Asia Pacific, Japan, Other pharmaceuticals | 181 | 181 | |||||||||
CNS, Inc. |
Consumer Healthcare | 142 | 137 | |||||||||
Polfa Poznan S.A. |
Europe pharmaceuticals | 118 | 118 | |||||||||
Certain businesses from UCB S.A. |
Emerging Markets and Asia Pacific pharmaceuticals | 89 | 87 | |||||||||
Laboratorios Phoenix S.A.I.C.yF. |
Emerging Markets pharmaceuticals | 66 | – | |||||||||
NovaMin Technology Inc. |
Consumer Healthcare | 52 | 50 | |||||||||
Others |
347 | 270 | ||||||||||
| 3,606 | 3,361 | |||||||||||
Table of Contents
133
| Europe, Emerging Markets, Asia Pacific, | ||||||
| Other pharmaceuticals CGUs | ViiV Healthcare CGU | |||||
| Valuation basis | Value in use | Fair value less costs to sell | ||||
| Key assumptions | Sales growth rates Profit margins Discount rate |
Sales growth rates Profit margins Discount rate |
||||
| Determination of assumptions | Growth rates are internal forecasts based on both
internal and external market information. Margins reflect past experience, adjusted for expected changes. Discount rate based on Group WACC. |
Growth rates are internal forecasts based on both internal and external market information. Margins reflect past experience, adjusted for expected changes. Discount rate based on Group WACC. | ||||
Period of specific projected cash flows |
5 years | 5 years | ||||
Discount rate |
8% | 8% | ||||
Terminal growth rate |
Europe | 6% p.a. decline | 2.5% p.a. | |||
| Emerging markets | 1% p.a. | |||||
| Asia Pacific | 0% p.a. | |||||
| Other | 0% p.a. | |||||
Table of Contents
134
| Japan pharmaceuticals CGU | Consumer Healthcare CGU | |||
Valuation basis |
Fair value less costs to sell | Fair value less costs to sell | ||
Key assumptions |
Sales growth rates Profit margins Discount rate |
Sales growth rates Advertising and promotion investment Terminal growth rate Discount rate |
||
Determination of assumptions |
Growth rates are internal forecasts based on both internal and external market
information. Margins reflect past experience, adjusted for expected changes. Discount rate based on Group WACC. |
Growth rates are internal forecasts based on both internal and external market
information. Advertising and promotion investment based on historical levels adjusted for management’s view of support needed for innovation and expansion. Terminal growth rate based on management’s estimate of future long-term average growth rates. Discount rate based on Group WACC. |
||
Period of specific projected cash flows |
5 years | 5 years | ||
Discount rate |
8% | 8% | ||
Terminal growth rate |
2% p.a. | 3% p.a. | ||
Table of Contents
135
| Computer | Licences, | Amortised | Indefinite life | |||||||||||||||||
| software | patents, etc. | brands | brands | Total | ||||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Cost at 1st January 2009 |
1,003 | 4,794 | 331 | 1,823 | 7,951 | |||||||||||||||
Exchange adjustments |
(36 | ) | (193 | ) | (23 | ) | (99 | ) | (351 | ) | ||||||||||
Capitalised internal development costs |
13 | – | – | – | 13 | |||||||||||||||
Additions through business combinations |
30 | 1,883 | 51 | 758 | 2,722 | |||||||||||||||
Other additions |
41 | 391 | – | – | 432 | |||||||||||||||
Disposals and asset write-offs |
(17 | ) | (26 | ) | – | – | (43 | ) | ||||||||||||
Reclassifications |
(4 | ) | – | – | – | (4 | ) | |||||||||||||
Cost at 31st December 2009 |
1,030 | 6,849 | 359 | 2,482 | 10,720 | |||||||||||||||
Exchange adjustments |
14 | 150 | 7 | 55 | 226 | |||||||||||||||
Capitalised internal development costs |
81 | – | – | – | 81 | |||||||||||||||
Additions through business combinations |
– | 214 | 11 | 27 | 252 | |||||||||||||||
Capitalised borrowing costs |
– | 2 | – | – | 2 | |||||||||||||||
Other additions |
58 | 469 | – | – | 527 | |||||||||||||||
Disposals and asset write-offs |
(25 | ) | (13 | ) | – | – | (38 | ) | ||||||||||||
Reclassifications |
16 | – | – | – | 16 | |||||||||||||||
Cost at 31st December 2010 |
1,174 | 7,671 | 377 | 2,564 | 11,786 | |||||||||||||||
Amortisation at 1st January 2009 |
(698 | ) | (995 | ) | (24 | ) | – | (1,717 | ) | |||||||||||
Exchange adjustments |
27 | 58 | – | – | 85 | |||||||||||||||
Charge for the year |
(113 | ) | (306 | ) | (13 | ) | – | (432 | ) | |||||||||||
Disposals and asset write-offs |
16 | 1 | – | – | 17 | |||||||||||||||
Amortisation at 31st December 2009 |
(768 | ) | (1,242 | ) | (37 | ) | – | (2,047 | ) | |||||||||||
Exchange adjustments |
(8 | ) | (37 | ) | – | – | (45 | ) | ||||||||||||
Charge for the year |
(106 | ) | (411 | ) | (16 | ) | – | (533 | ) | |||||||||||
Disposals and asset write-offs |
20 | 1 | – | – | 21 | |||||||||||||||
Amortisation at 31st December 2010 |
(862 | ) | (1,689 | ) | (53 | ) | – | (2,604 | ) | |||||||||||
Impairment at 1st January 2009 |
(32 | ) | (304 | ) | – | (29 | ) | (365 | ) | |||||||||||
Exchange adjustments |
1 | 19 | – | 3 | 23 | |||||||||||||||
Impairment losses |
(4 | ) | (168 | ) | – | – | (172 | ) | ||||||||||||
Disposals and asset write-offs |
2 | 22 | – | – | 24 | |||||||||||||||
Impairment at 31st December 2009 |
(33 | ) | (431 | ) | – | (26 | ) | (490 | ) | |||||||||||
Exchange adjustments |
(1 | ) | (13 | ) | – | (1 | ) | (15 | ) | |||||||||||
Impairment losses |
(5 | ) | (143 | ) | (2 | ) | – | (150 | ) | |||||||||||
Disposals and asset write-offs |
3 | – | 2 | – | 5 | |||||||||||||||
Impairment at 31st December 2010 |
(36 | ) | (587 | ) | – | (27 | ) | (650 | ) | |||||||||||
Total amortisation and impairment at 31st December 2009 |
(801 | ) | (1,673 | ) | (37 | ) | (26 | ) | (2,537 | ) | ||||||||||
Total amortisation and impairment at 31st December 2010 |
(898 | ) | (2,276 | ) | (53 | ) | (27 | ) | (3,254 | ) | ||||||||||
Net book value at 1st January 2009 |
273 | 3,495 | 307 | 1,794 | 5,869 | |||||||||||||||
Net book value at 31st December 2009 |
229 | 5,176 | 322 | 2,456 | 8,183 | |||||||||||||||
Net book value at 31st December 2010 |
276 | 5,395 | 324 | 2,537 | 8,532 | |||||||||||||||
Table of Contents
136
| Amortisation | Net impairment losses | |||||||||||||||
| 2010 | 2009 | 2010 | 2009 | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Cost of sales |
26 | 29 | – | 1 | ||||||||||||
Selling, general and administration |
353 | 270 | 13 | 1 | ||||||||||||
Research and development |
154 | 133 | 137 | 170 | ||||||||||||
| 533 | 432 | 150 | 172 | |||||||||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Fluviral |
663 | 648 | ||||||
Lovaza |
593 | 637 | ||||||
Selzentry |
299 | 337 | ||||||
Arzerra |
294 | 191 | ||||||
Duac |
157 | 165 | ||||||
Fraxiparine |
135 | 158 | ||||||
Others |
3,254 | 3,040 | ||||||
| 5,395 | 5,176 | |||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Panadol |
426 | 399 | ||||||
Sensodyne |
270 | 271 | ||||||
Stiefel trade name |
216 | 209 | ||||||
Breathe Right |
199 | 193 | ||||||
Physiogel |
182 | 176 | ||||||
Polident |
114 | 115 | ||||||
Corega |
102 | 102 | ||||||
Biotene |
111 | 108 | ||||||
Poligrip |
70 | 71 | ||||||
Solpadeine |
59 | 59 | ||||||
Others |
788 | 753 | ||||||
| 2,537 | 2,456 | |||||||
Table of Contents
137
| Joint | Associated | 2010 | Joint | Associated | 2009 | |||||||||||||||||||
| ventures | undertakings | Total | ventures | undertakings | Total | |||||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||||
At 1st January |
46 | 849 | 895 | 28 | 524 | 552 | ||||||||||||||||||
Exchange adjustments |
4 | 8 | 12 | (3 | ) | (44 | ) | (47 | ) | |||||||||||||||
Additions |
30 | 35 | 65 | 36 | 312 | 348 | ||||||||||||||||||
Disposals |
– | (2 | ) | (2 | ) | – | (69 | ) | (69 | ) | ||||||||||||||
Transfer from other investments |
– | 40 | 40 | – | 56 | 56 | ||||||||||||||||||
Distributions received |
(3 | ) | (18 | ) | (21 | ) | (7 | ) | (10 | ) | (17 | ) | ||||||||||||
Other movements |
– | 11 | 11 | – | 8 | 8 | ||||||||||||||||||
(Loss)/profit after tax recognised in the consolidated
income statement |
(23 | ) | 104 | 81 | (8 | ) | 72 | 64 | ||||||||||||||||
At 31st December |
54 | 1,027 | 1,081 | 46 | 849 | 895 | ||||||||||||||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Total assets: |
||||||||
Quest Diagnostics Inc. |
5,466 | 5,319 | ||||||
Aspen Pharmacare Holdings Limited |
1,913 | 1,318 | ||||||
Others |
360 | 121 | ||||||
| 7,739 | 6,758 | |||||||
Total liabilities: |
||||||||
Quest Diagnostics Inc. |
(2,868 | ) | (2,828 | ) | ||||
Aspen Pharmacare Holdings Limited |
(786 | ) | (689 | ) | ||||
Others |
(73 | ) | (19 | ) | ||||
| (3,727 | ) | (3,536 | ) | |||||
Net assets |
4,012 | 3,222 | ||||||
Table of Contents
138
| 2010 | 2009 | |||||||
| £m | £m | |||||||
At 1st January |
454 | 478 | ||||||
Exchange adjustments |
7 | (48 | ) | |||||
Additions |
281 | 175 | ||||||
Net fair value movements |
96 | 57 | ||||||
Impairment losses |
(60 | ) | (95 | ) | ||||
Transfer to investments in associates and joint ventures |
(40 | ) | (56 | ) | ||||
Disposals |
(27 | ) | (57 | ) | ||||
At 31st December |
711 | 454 | ||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Original cost |
429 | 401 | ||||||
Impairments recognised in profit and loss |
(340 | ) | (292 | ) | ||||
Subsequent fair value increases |
45 | 43 | ||||||
Carrying value at 31st December |
134 | 152 | ||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Amounts receivable under insurance contracts |
343 | 299 | ||||||
Pension schemes in surplus |
23 | 23 | ||||||
Other receivables |
190 | 261 | ||||||
| 556 | 583 | |||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Raw materials and consumables |
1,466 | 1,153 | ||||||
Work in progress |
751 | 1,437 | ||||||
Finished goods |
1,620 | 1,474 | ||||||
| 3,837 | 4,064 | |||||||
Table of Contents
139
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Trade receivables |
4,727 | 5,486 | ||||||
Prepaid pension contributions |
2 | 1 | ||||||
Other prepayments and accrued income |
256 | 301 | ||||||
Interest receivable |
16 | 20 | ||||||
Employee loans and advances |
50 | 48 | ||||||
Other receivables |
742 | 636 | ||||||
| 5,793 | 6,492 | |||||||
| 2010 | 2009 | |||||||
Bad and doubtful debt provision |
£m | £m | ||||||
At 1st January |
116 | 129 | ||||||
Exchange adjustments |
– | (10 | ) | |||||
Charge for the year |
54 | 21 | ||||||
Subsequent recoveries of amounts provided for |
(19 | ) | (18 | ) | ||||
Utilised |
(1 | ) | (6 | ) | ||||
At 31st December |
150 | 116 | ||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Cash at bank and in hand |
1,027 | 856 | ||||||
Short-term deposits |
5,030 | 5,689 | ||||||
| 6,057 | 6,545 | |||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Land and buildings |
6 | 13 | ||||||
Plant, equipment and vehicles |
10 | 1 | ||||||
| 16 | 14 | |||||||
Table of Contents
140
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Trade payables |
2,141 | 1,855 | ||||||
Wages and salaries |
931 | 1,089 | ||||||
Social security |
115 | 125 | ||||||
Other payables |
296 | 280 | ||||||
Deferred income |
70 | 156 | ||||||
Customer return and rebate accruals |
1,632 | 1,379 | ||||||
Other accruals |
1,703 | 1,888 | ||||||
| 6,888 | 6,772 | |||||||
| 2010 | 2009 | 2008 | ||||||||||
Pension and other post-employment costs |
£m | £m | £m | |||||||||
UK pension schemes |
158 | 206 | 236 | |||||||||
US pension schemes |
115 | 94 | 60 | |||||||||
Other overseas pensions schemes |
125 | 101 | 87 | |||||||||
Unfunded post-retirement healthcare schemes |
156 | 90 | 118 | |||||||||
Other post-employment costs |
– | – | 4 | |||||||||
| 554 | 491 | 505 | ||||||||||
Analysed as: |
||||||||||||
Funded defined benefit/hybrid pension schemes |
325 | 338 | 318 | |||||||||
Unfunded defined benefit pension schemes |
28 | 25 | 23 | |||||||||
Unfunded post-retirement healthcare schemes |
156 | 90 | 118 | |||||||||
Defined benefit schemes |
509 | 453 | 459 | |||||||||
Defined contribution pension schemes |
45 | 38 | 42 | |||||||||
Other post-employment costs |
– | – | 4 | |||||||||
| 554 | 491 | 505 | ||||||||||
| The costs of the defined benefit pension and post-retirement healthcare schemes are charged in the income statement as follows: | ||||||||||||
Cost of sales |
117 | 121 | 179 | |||||||||
Selling, general and administration |
254 | 195 | 160 | |||||||||
Research and development |
138 | 137 | 120 | |||||||||
| 509 | 453 | 459 | ||||||||||
Table of Contents
141
| UK | USA | |||||||||||||||
| Male | Female | Male | Female | |||||||||||||
| Years | Years | Years | Years | |||||||||||||
Current |
27.4 | 28.7 | 24.6 | 26.3 | ||||||||||||
Projected for 2030 |
29.7 | 30.5 | 26.5 | 27.4 | ||||||||||||
| UK | USA | Rest of World | ||||||||||||||||||||||||||||||||||
| 2010 | 2009 | 2008 | 2010 | 2009 | 2008 | 2010 | 2009 | 2008 | ||||||||||||||||||||||||||||
| % pa | % pa | % pa | % pa | % pa | % pa | % pa | % pa | % pa | ||||||||||||||||||||||||||||
Rate of increase of future earnings |
4.50 | 4.60 | 3.90 | 4.50 | 4.50 | 4.50 | 3.50 | 3.00 | 3.10 | |||||||||||||||||||||||||||
Discount rate |
5.50 | 5.70 | 6.20 | 5.20 | 5.75 | 6.00 | 4.50 | 4.70 | 5.00 | |||||||||||||||||||||||||||
Expected pension increases |
3.50 | 3.60 | 2.90 | n/a | n/a | n/a | 2.20 | 2.20 | 2.10 | |||||||||||||||||||||||||||
Cash balance credit/conversion rate |
n/a | n/a | n/a | 4.20 | 4.75 | 4.50 | 1.30 | 1.60 | 1.20 | |||||||||||||||||||||||||||
Inflation rate |
3.50 | 3.60 | 2.70 | 2.25 | 2.50 | 2.50 | 1.70 | 1.70 | 1.70 | |||||||||||||||||||||||||||
Table of Contents
142
| Post-retirement | ||||||||||||||||||||
| Pensions | benefits | |||||||||||||||||||
| UK | USA | Rest of World | Group | Group | ||||||||||||||||
2010 |
£m | £m | £m | £m | £m | |||||||||||||||
Amounts charged to operating profit Current service cost |
130 | 68 | 70 | 268 | 31 | |||||||||||||||
Past service cost |
– | – | – | – | 5 | |||||||||||||||
Expected return on pension scheme assets |
(427 | ) | (134 | ) | (51 | ) | (612 | ) | – | |||||||||||
Interest on scheme liabilities |
425 | 151 | 64 | 640 | 73 | |||||||||||||||
Settlements and curtailments |
30 | 30 | (3 | ) | 57 | 47 | ||||||||||||||
| 158 | 115 | 80 | 353 | 156 | ||||||||||||||||
Actuarial gains/(losses) recorded in the statement of
comprehensive income |
73 | 43 | (37 | ) | 79 | (80 | ) | |||||||||||||
| Post-retirement | ||||||||||||||||||||
| Pensions | benefits | |||||||||||||||||||
| UK | USA | Rest of World | Group | Group | ||||||||||||||||
2009 |
£m | £m | £m | £m | £m | |||||||||||||||
Amounts charged to operating profit Current service cost |
121 | 66 | 64 | 251 | 35 | |||||||||||||||
Past service cost |
– | (6 | ) | – | (6 | ) | (27 | ) | ||||||||||||
Expected return on pension scheme assets |
(347 | ) | (121 | ) | (46 | ) | (514 | ) | – | |||||||||||
Interest on scheme liabilities |
378 | 148 | 62 | 588 | 74 | |||||||||||||||
Settlements and curtailments |
54 | 7 | (17 | ) | 44 | 8 | ||||||||||||||
| 206 | 94 | 63 | 363 | 90 | ||||||||||||||||
Actuarial (losses)/gains recorded in the statement of
comprehensive income |
(578 | ) | (5 | ) | (77 | ) | (660 | ) | 1 | |||||||||||
| Post-retirement | ||||||||||||||||||||
| Pensions | benefits | |||||||||||||||||||
| UK | USA | Rest of World | Group | Group | ||||||||||||||||
2008 |
£m | £m | £m | £m | £m | |||||||||||||||
Amounts charged to operating profit |
||||||||||||||||||||
Current service cost |
126 | 61 | 59 | 246 | 30 | |||||||||||||||
Past service cost |
– | 10 | 2 | 12 | 4 | |||||||||||||||
Expected return on pension scheme assets |
(442 | ) | (144 | ) | (47 | ) | (633 | ) | – | |||||||||||
Interest on scheme liabilities |
377 | 121 | 53 | 551 | 62 | |||||||||||||||
Settlements and curtailments |
175 | 12 | (22 | ) | 165 | 22 | ||||||||||||||
| 236 | 60 | 45 | 341 | 118 | ||||||||||||||||
Actuarial (losses)/gains recorded in the statement of
comprehensive income |
(776 | ) | (576 | ) | (82 | ) | (1,434 | ) | 64 | |||||||||||
Table of Contents
143
| UK | USA | Rest of World | Group | |||||||||||||||||||||||||
| Average | ||||||||||||||||||||||||||||
| At 31st December 2010 | Expected rate | Fair | Expected rate | Fair | expected rate | Fair | Fair | |||||||||||||||||||||
| of return | value | of return | value | of return | value | value | ||||||||||||||||||||||
| % | £m | % | £m | % | £m | £m | ||||||||||||||||||||||
Equities |
8.00 | 4,698 | 8.25 | 1,092 | 7.40 | 251 | 6,041 | |||||||||||||||||||||
Property |
7.00 | 272 | 7.25 | 147 | 7.00 | 6 | 425 | |||||||||||||||||||||
Bonds |
4.50 | 2,460 | 4.75 | 1,012 | 3.10 | 572 | 4,044 | |||||||||||||||||||||
Other assets |
3.50 | 1,188 | 0.25 | 59 | 3.80 | 399 | 1,646 | |||||||||||||||||||||
Fair value of assets |
8,618 | 2,310 | 1,228 | 12,156 | ||||||||||||||||||||||||
Present value of scheme obligations |
(9,119 | ) | (2,781 | ) | (1,479 | ) | (13,379 | ) | ||||||||||||||||||||
| (501 | ) | (471 | ) | (251 | ) | (1,223 | ) | |||||||||||||||||||||
Unrecognised past service cost |
– | (2 | ) | 1 | (1 | ) | ||||||||||||||||||||||
Recognised on the balance sheet |
(501 | ) | (473 | ) | (250 | ) | (1,224 | ) | ||||||||||||||||||||
Included in other non-current assets |
– | – | 23 | 23 | ||||||||||||||||||||||||
Included in pensions and other post-employment
benefits |
(501 | ) | (473 | ) | (273 | ) | (1,247 | ) | ||||||||||||||||||||
| (501 | ) | (473 | ) | (250 | ) | (1,224 | ) | |||||||||||||||||||||
Actual return on plan assets |
881 | 240 | 43 | 1,164 | ||||||||||||||||||||||||
| UK | USA | Rest of World | Group | |||||||||||||||||||||||||
| Average | ||||||||||||||||||||||||||||
| At 31st December 2009 | Expected rate | Fair | Expected rate | Fair | expected rate | Fair | Fair | |||||||||||||||||||||
| of return | value | of return | value | of return | value | value | ||||||||||||||||||||||
| % | £m | % | £m | % | £m | £m | ||||||||||||||||||||||
Equities |
8.00 | 4,209 | 8.25 | 914 | 7.50 | 232 | 5,355 | |||||||||||||||||||||
Property |
7.00 | 291 | 7.25 | 159 | 7.00 | 20 | 470 | |||||||||||||||||||||
Bonds |
4.90 | 2,632 | 5.00 | 907 | 3.50 | 562 | 4,101 | |||||||||||||||||||||
Other assets |
0.50 | 367 | 0.25 | 92 | 3.80 | 309 | 768 | |||||||||||||||||||||
Fair value of assets |
7,499 | 2,072 | 1,123 | 10,694 | ||||||||||||||||||||||||
Present value of scheme obligations |
(8,446 | ) | (2,628 | ) | (1,364 | ) | (12,438 | ) | ||||||||||||||||||||
| (947 | ) | (556 | ) | (241 | ) | (1,744 | ) | |||||||||||||||||||||
Unrecognised past service cost |
– | (2 | ) | 1 | (1 | ) | ||||||||||||||||||||||
Recognised on the balance sheet |
(947 | ) | (558 | ) | (240 | ) | (1,745 | ) | ||||||||||||||||||||
Included in other non-current assets |
– | – | 23 | 23 | ||||||||||||||||||||||||
Included in pensions and other post-employment benefits |
(947 | ) | (558 | ) | (263 | ) | (1,768 | ) | ||||||||||||||||||||
| (947 | ) | (558 | ) | (240 | ) | (1,745 | ) | |||||||||||||||||||||
Actual return on plan assets |
1,076 | 243 | 65 | 1,384 | ||||||||||||||||||||||||
Table of Contents
144
| UK | USA | Rest of World | Group | |||||||||||||||||||||||||
| Average | ||||||||||||||||||||||||||||
| At 31st December 2008 | Expected rate | Fair | Expected rate | Fair | expected rate | Fair | Fair | |||||||||||||||||||||
| of return | value | of return | value | of return | value | value | ||||||||||||||||||||||
| % | £m | % | £m | % | £m | £m | ||||||||||||||||||||||
Equities |
7.75 | 3,334 | 8.25 | 838 | 7.00 | 211 | 4,383 | |||||||||||||||||||||
Property |
6.75 | 331 | 7.25 | 259 | 6.75 | 22 | 612 | |||||||||||||||||||||
Bonds |
4.75 | 2,430 | 5.25 | 893 | 3.25 | 598 | 3,921 | |||||||||||||||||||||
Other assets |
2.75 | 40 | 1.50 | 26 | 4.25 | 306 | 372 | |||||||||||||||||||||
Fair value of assets |
6,135 | 2,016 | 1,137 | 9,288 | ||||||||||||||||||||||||
Present value of scheme obligations |
(6,885 | ) | (2,738 | ) | (1,357 | ) | (10,980 | ) | ||||||||||||||||||||
| (750 | ) | (722 | ) | (220 | ) | (1,692 | ) | |||||||||||||||||||||
Unrecognised past service cost |
– | – | 1 | 1 | ||||||||||||||||||||||||
Restriction on surplus |
– | – | (6 | ) | (6 | ) | ||||||||||||||||||||||
Recognised on the balance sheet |
(750 | ) | (722 | ) | (225 | ) | (1,697 | ) | ||||||||||||||||||||
Included in other non-current assets |
– | – | 39 | 39 | ||||||||||||||||||||||||
Included in pensions and other post-employment benefits |
(750 | ) | (722 | ) | (264 | ) | (1,736 | ) | ||||||||||||||||||||
| (750 | ) | (722 | ) | (225 | ) | (1,697 | ) | |||||||||||||||||||||
Actual return on plan assets |
(1,249 | ) | (470 | ) | (87 | ) | (1,806 | ) | ||||||||||||||||||||
Table of Contents
145
| Post-retirement | ||||||||||||||||||||
| Pensions | benefits | |||||||||||||||||||
| Movements in defined benefit obligations | UK | USA | Rest of World | Group | Group | |||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Obligations at 1st January 2008 |
(7,371 | ) | (1,945 | ) | (1,022 | ) | (10,338 | ) | (1,019 | ) | ||||||||||
Exchange adjustments |
– | (753 | ) | (353 | ) | (1,106 | ) | (351 | ) | |||||||||||
Service cost |
(126 | ) | (71 | ) | (61 | ) | (258 | ) | (28 | ) | ||||||||||
Interest cost |
(377 | ) | (121 | ) | (53 | ) | (551 | ) | (62 | ) | ||||||||||
Settlements and curtailments |
(175 | ) | (12 | ) | 19 | (168 | ) | (16 | ) | |||||||||||
Actuarial gains |
915 | 38 | 58 | 1,011 | 64 | |||||||||||||||
Scheme participants’ contributions |
(33 | ) | – | (5 | ) | (38 | ) | (9 | ) | |||||||||||
Benefits paid |
282 | 126 | 60 | 468 | 53 | |||||||||||||||
Transfers to other provisions |
– | – | – | – | 14 | |||||||||||||||
Obligations at 31st December 2008 |
(6,885 | ) | (2,738 | ) | (1,357 | ) | (10,980 | ) | (1,354 | ) | ||||||||||
Exchange adjustments |
– | 294 | 109 | 403 | 133 | |||||||||||||||
Service cost |
(121 | ) | (58 | ) | (64 | ) | (243 | ) | (5 | ) | ||||||||||
Interest cost |
(378 | ) | (148 | ) | (62 | ) | (588 | ) | (74 | ) | ||||||||||
Settlements and curtailments |
(54 | ) | (7 | ) | 68 | 7 | (8 | ) | ||||||||||||
Actuarial (losses)/gains |
(1,307 | ) | (127 | ) | (102 | ) | (1,536 | ) | 1 | |||||||||||
Scheme participants’ contributions |
(17 | ) | – | (8 | ) | (25 | ) | (11 | ) | |||||||||||
Benefits paid |
345 | 156 | 71 | 572 | 69 | |||||||||||||||
Acquisitions |
(29 | ) | – | (19 | ) | (48 | ) | (4 | ) | |||||||||||
Obligations at 31st December 2009 |
(8,446 | ) | (2,628 | ) | (1,364 | ) | (12,438 | ) | (1,253 | ) | ||||||||||
Exchange adjustments |
– | (84 | ) | (27 | ) | (111 | ) | (38 | ) | |||||||||||
Service cost |
(130 | ) | (68 | ) | (70 | ) | (268 | ) | (31 | ) | ||||||||||
Interest cost |
(425 | ) | (151 | ) | (64 | ) | (640 | ) | (73 | ) | ||||||||||
Settlements and curtailments |
(30 | ) | (30 | ) | 3 | (57 | ) | (44 | ) | |||||||||||
Actuarial losses |
(381 | ) | (63 | ) | (29 | ) | (473 | ) | (80 | ) | ||||||||||
Scheme participants’ contributions |
(20 | ) | – | (8 | ) | (28 | ) | (13 | ) | |||||||||||
Benefits paid |
313 | 243 | 80 | 636 | 73 | |||||||||||||||
Obligations at 31st December 2010 |
(9,119 | ) | (2,781 | ) | (1,479 | ) | (13,379 | ) | (1,459 | ) | ||||||||||
Unrecognised past service cost |
– | (2 | ) | 1 | (1 | ) | 34 | |||||||||||||
Recognised on the balance sheet at 31st December 2010 |
(9,119 | ) | (2,783 | ) | (1,478 | ) | (13,380 | ) | (1,425 | ) | ||||||||||
Table of Contents
146
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Funded |
(13,033 | ) | (12,126 | ) | (10,662 | ) | ||||||
Unfunded |
(346 | ) | (312 | ) | (318 | ) | ||||||
| (13,379 | ) | (12,438 | ) | (10,980 | ) | |||||||
| Post-retirement | ||||||||||||||||||||
| Pensions | benefits | |||||||||||||||||||
| UK | USA | Rest of World | Group | Group | ||||||||||||||||
| Movements in fair values of assets | £m | £m | £m | £m | £m | |||||||||||||||
Assets at 1st January 2008 |
7,293 | 2,004 | 885 | 10,182 | – | |||||||||||||||
Exchange adjustments |
– | 598 | 298 | 896 | – | |||||||||||||||
Expected return on assets |
442 | 144 | 47 | 633 | – | |||||||||||||||
Settlements and curtailments |
– | – | 3 | 3 | – | |||||||||||||||
Actuarial gains/(losses) |
(1,691 | ) | (614 | ) | (134 | ) | (2,439 | ) | – | |||||||||||
Employer contributions |
340 | 10 | 93 | 443 | 44 | |||||||||||||||
Scheme participants’ contributions |
33 | – | 5 | 38 | 9 | |||||||||||||||
Benefits paid |
(282 | ) | (126 | ) | (60 | ) | (468 | ) | (53 | ) | ||||||||||
Assets at 31st December 2008 |
6,135 | 2,016 | 1,137 | 9,288 | – | |||||||||||||||
Exchange adjustments |
– | (221 | ) | (93 | ) | (314 | ) | – | ||||||||||||
Expected return on assets |
347 | 121 | 46 | 514 | – | |||||||||||||||
Settlements and curtailments |
– | – | (51 | ) | (51 | ) | – | |||||||||||||
Actuarial losses |
729 | 122 | 19 | 870 | – | |||||||||||||||
Employer contributions |
594 | 190 | 110 | 894 | 58 | |||||||||||||||
Scheme participants’ contributions |
17 | – | 8 | 25 | 11 | |||||||||||||||
Benefits paid |
(345 | ) | (156 | ) | (71 | ) | (572 | ) | (69 | ) | ||||||||||
Acquisitions |
22 | – | 18 | 40 | – | |||||||||||||||
Assets at 31st December 2009 |
7,499 | 2,072 | 1,123 | 10,694 | – | |||||||||||||||
Exchange adjustments |
– | 66 | 26 | 92 | – | |||||||||||||||
Expected return on assets |
427 | 134 | 51 | 612 | – | |||||||||||||||
Actuarial gains |
454 | 106 | (8 | ) | 552 | – | ||||||||||||||
Employer contributions |
531 | 175 | 108 | 814 | 60 | |||||||||||||||
Scheme participants’ contributions |
20 | – | 8 | 28 | 13 | |||||||||||||||
Benefits paid |
(313 | ) | (243 | ) | (80 | ) | (636 | ) | (73 | ) | ||||||||||
Assets at 31st December 2010 |
8,618 | 2,310 | 1,228 | 12,156 | – | |||||||||||||||
Table of Contents
147
| Post-retirement | ||||||||||||||||||||
| Pensions | benefits | |||||||||||||||||||
| UK | USA | Rest of World | Group | Group | ||||||||||||||||
| History of experience gains and losses | £m | £m | £m | £m | £m | |||||||||||||||
2010 |
||||||||||||||||||||
Experience gains/(losses) of scheme assets |
454 | 106 | (8 | ) | 552 | |||||||||||||||
Percentage of scheme assets at 31st December 2010 |
5 | % | 5 | % | 1 | % | 5 | % | ||||||||||||
Experience (losses)/gains of scheme liabilities |
(45 | ) | 5 | (3 | ) | (43 | ) | (14 | ) | |||||||||||
Percentage of scheme obligations at 31st December 2010 |
– | – | – | – | 1 | % | ||||||||||||||
Fair value of assets |
8,618 | 2,310 | 1,228 | 12,156 | – | |||||||||||||||
Present value of scheme obligations |
(9,119 | ) | (2,781 | ) | (1,479 | ) | (13,379 | ) | (1,459 | ) | ||||||||||
Deficits in the schemes |
(501 | ) | (471 | ) | (251 | ) | (1,223 | ) | (1,459 | ) | ||||||||||
2009 |
||||||||||||||||||||
Experience gains of scheme assets |
729 | 122 | 19 | 870 | ||||||||||||||||
Percentage of scheme assets at 31st December 2009 |
10 | % | 6 | % | 2 | % | 8 | % | ||||||||||||
Experience gains/(losses) of scheme liabilities |
162 | (27 | ) | (15 | ) | 120 | 6 | |||||||||||||
Percentage of scheme obligations at 31st December 2009 |
2 | % | 1 | % | 1 | % | 1 | % | – | |||||||||||
Fair value of assets |
7,499 | 2,072 | 1,123 | 10,694 | _ | |||||||||||||||
Present value of scheme obligations |
(8,446 | ) | (2,628 | ) | (1,364 | ) | (12,438 | ) | (1,253 | ) | ||||||||||
Deficits in the schemes |
(947 | ) | (556 | ) | (241 | ) | (1,744 | ) | (1,253 | ) | ||||||||||
2008 |
||||||||||||||||||||
Experience losses of scheme assets |
(1,691 | ) | (614 | ) | (134 | ) | (2,439 | ) | ||||||||||||
Percentage of scheme assets at 31st December 2008 |
28 | % | 30 | % | 12 | % | 26 | % | ||||||||||||
Experience (losses)/gains of scheme liabilities |
(148 | ) | 2 | 1 | (145 | ) | (14 | ) | ||||||||||||
Percentage of scheme obligations at 31st December 2008 |
2 | % | – | – | 1 | % | 1 | % | ||||||||||||
Fair value of assets |
6,135 | 2,016 | 1,137 | 9,288 | – | |||||||||||||||
Present value of scheme obligations |
(6,885 | ) | (2,738 | ) | (1,357 | ) | (10,980 | ) | (1,354 | ) | ||||||||||
Deficits in the schemes |
(750 | ) | (722 | ) | (220 | ) | (1,692 | ) | (1,354 | ) | ||||||||||
2007 |
||||||||||||||||||||
Experience gains/(losses) of scheme assets |
168 | 46 | (18 | ) | 196 | |||||||||||||||
Percentage of scheme assets at 31st December 2007 |
2 | % | 2 | % | 2 | % | 2 | % | ||||||||||||
Experience gains/(losses) of scheme liabilities |
33 | (30 | ) | 6 | 9 | – | ||||||||||||||
Percentage of scheme obligations at 31st December 2007 |
– | 2 | % | 1 | % | – | – | |||||||||||||
Fair value of assets |
7,293 | 2,004 | 885 | 10,182 | – | |||||||||||||||
Present value of scheme obligations |
(7,371 | ) | (1,945 | ) | (1,022 | ) | (10,338 | ) | (1,019 | ) | ||||||||||
(Deficits)/surpluses in the schemes |
(78 | ) | 59 | (137 | ) | (156 | ) | (1,019 | ) | |||||||||||
2006 |
||||||||||||||||||||
Experience gains of scheme assets |
227 | 168 | 26 | 421 | ||||||||||||||||
Percentage of scheme assets at 31st December 2006 |
3 | % | 9 | % | 4 | % | 5 | % | ||||||||||||
Experience (losses)/gains of scheme liabilities |
(37 | ) | (16 | ) | (42 | ) | (95 | ) | 17 | |||||||||||
Percentage of scheme obligations at 31st December 2006 |
– | 1 | % | 4 | % | 1 | % | 2 | % | |||||||||||
Fair value of assets |
6,554 | 1,953 | 741 | 9,248 | – | |||||||||||||||
Present value of scheme obligations |
(7,444 | ) | (1,949 | ) | (952 | ) | (10,345 | ) | (1,063 | ) | ||||||||||
(Deficits)/surpluses in the schemes |
(890 | ) | 4 | (211 | ) | (1,097 | ) | (1,063 | ) | |||||||||||
Table of Contents
148
| £m | ||||
A 0.25% decrease in discount rate would have the following approximate effect: |
||||
Increase in annual pension cost |
4 | |||
Increase in annual post-retirement benefits cost |
– | |||
Increase in pension obligation |
472 | |||
Increase in post-retirement benefits obligation |
41 | |||
A one year increase in life expectancy would have the following approximate effect: |
||||
Increase in annual pension cost |
20 | |||
Increase in annual post-retirement benefits cost |
4 | |||
Increase in pension obligation |
305 | |||
Increase in post-retirement benefits obligation |
60 | |||
A 0.25% decrease in expected rates of return on assets would have the following approximate effect: |
||||
Increase in annual pension cost |
28 | |||
A 1% increase in the rate of future healthcare inflation would have the following approximate effect: |
||||
Increase in annual post-retirement benefits cost |
1 | |||
Increase in post-retirement benefits obligation |
25 | |||
A 0.25% increase in inflation would have the following approximate effect: |
||||
Increase in annual pension cost |
25 | |||
Increase in pension obligation |
339 | |||
| Integration | ||||||||||||||||||||||||
| Legal | Major | Employee | and | |||||||||||||||||||||
| and other | restructuring | related | manufacturing | Other | ||||||||||||||||||||
| disputes | programmes | provisions | reorganisation | provisions | Total | |||||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||||
At 1st January 2010 |
2,020 | 574 | 241 | 55 | 351 | 3,241 | ||||||||||||||||||
Exchange adjustments |
12 | (4 | ) | 6 | 1 | 4 | 19 | |||||||||||||||||
Charge for the year |
4,111 | 837 | 39 | – | 15 | 5,002 | ||||||||||||||||||
Reversed unused |
(103 | ) | (40 | ) | (5 | ) | (4 | ) | (16 | ) | (168 | ) | ||||||||||||
Unwinding of discount |
7 | 3 | – | – | 8 | 18 | ||||||||||||||||||
Utilised |
(2,047 | ) | (610 | ) | (35 | ) | (17 | ) | (19 | ) | (2,728 | ) | ||||||||||||
Transfer to pensions obligations |
– | (110 | ) | – | – | – | (110 | ) | ||||||||||||||||
Reclassifications and other movements |
– | 24 | 5 | (8 | ) | (11 | ) | 10 | ||||||||||||||||
At 31st December 2010 |
4,000 | 674 | 251 | 27 | 332 | 5,284 | ||||||||||||||||||
To be settled within one year |
3,654 | 512 | 34 | 4 | 176 | 4,380 | ||||||||||||||||||
To be settled after one year |
346 | 162 | 217 | 23 | 156 | 904 | ||||||||||||||||||
At 31st December 2010 |
4,000 | 674 | 251 | 27 | 332 | 5,284 | ||||||||||||||||||
Table of Contents
149
Table of Contents
150
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Accruals and deferred income |
103 | 124 | ||||||
Other payables |
491 | 481 | ||||||
| 594 | 605 | |||||||
| 2010 | 2009 | |||||||||
| Listing exchange | £m | £m | ||||||||
Current assets: |
||||||||||
Liquid investments |
184 | 268 | ||||||||
Cash and cash equivalents |
6,057 | 6,545 | ||||||||
| 6,241 | 6,813 | |||||||||
Short-term borrowings: |
||||||||||
US$ Floating Rate Note 2010 |
New York Stock Exchange | – | (621 | ) | ||||||
Commercial paper |
– | (621 | ) | |||||||
Bank loans and overdrafts |
(259 | ) | (182 | ) | ||||||
Loan stock |
– | (7 | ) | |||||||
Obligations under finance leases |
(32 | ) | (40 | ) | ||||||
| (291 | ) | (1,471 | ) | |||||||
Long-term borrowings: |
||||||||||
3.00% € European Medium Term Note 2012 |
London Stock Exchange | (640 | ) | (662 | ) | |||||
5.125% € European Medium Term Note 2012 |
London Stock Exchange | (1,919 | ) | (1,985 | ) | |||||
4.85% US$ US Medium Term Note 2013 |
New York Stock Exchange | (1,599 | ) | (1,548 | ) | |||||
4.375% US$ US Medium Term Note 2014 |
London Stock Exchange | (1,049 | ) | (990 | ) | |||||
3.875% € European Medium Term Note 2015 |
London Stock Exchange | (1,358 | ) | (1,404 | ) | |||||
5.625% € European Medium Term Note 2017 |
London Stock Exchange | (1,062 | ) | (1,100 | ) | |||||
5.65% US$ US Medium Term Note 2018 |
New York Stock Exchange | (1,756 | ) | (1,701 | ) | |||||
4.00% € European Medium Term Note 2025 |
London Stock Exchange | (632 | ) | (653 | ) | |||||
5.25% £ European Medium Term Note 2033 |
London Stock Exchange | (981 | ) | (979 | ) | |||||
5.375% US$ US Medium Term Note 2034 |
London Stock Exchange | (318 | ) | (308 | ) | |||||
6.375% US$ US Medium Term Note 2038 |
New York Stock Exchange | (1,744 | ) | (1,689 | ) | |||||
6.375% £ European Medium Term Note 2039 |
London Stock Exchange | (694 | ) | (693 | ) | |||||
5.25% £ European Medium Term Note 2042 |
London Stock Exchange | (985 | ) | (984 | ) | |||||
Bank loans |
(1 | ) | – | |||||||
Obligations under finance leases |
(71 | ) | (90 | ) | ||||||
| (14,809 | ) | (14,786 | ) | |||||||
Net debt |
(8,859 | ) | (9,444 | ) | ||||||
Table of Contents
151
| 2010 | 2009 | |||||||
| Finance lease obligations | £m | £m | ||||||
Rental payments due within one year |
37 | 44 | ||||||
Rental payments due between one and two years |
32 | 38 | ||||||
Rental payments due between two and three years |
21 | 26 | ||||||
Rental payments due between three and four years |
13 | 16 | ||||||
Rental payments due between four and five years |
8 | 6 | ||||||
Rental payments due after five years |
8 | 16 | ||||||
Total future rental payments |
119 | 146 | ||||||
Future finance charges |
(16 | ) | (16 | ) | ||||
Total finance lease obligations |
103 | 130 | ||||||
Table of Contents
152
| Share | ||||||||||||
| Ordinary Shares of 25p each | premium | |||||||||||
| Number | £m | £m | ||||||||||
Share capital authorised |
||||||||||||
At 31st December 2008 |
10,000,000,000 | 2,500 | ||||||||||
At 31st December 2009 |
10,000,000,000 | 2,500 | ||||||||||
At 31st December 2010 |
10,000,000,000 | 2,500 | ||||||||||
Share capital issued and fully paid |
||||||||||||
At 1st January 2008 |
6,012,587,026 | 1,503 | 1,266 | |||||||||
Issued under share option schemes |
5,640,119 | 2 | 60 | |||||||||
Share capital purchased and cancelled |
(356,910,908 | ) | (90 | ) | – | |||||||
At 31st December 2008 |
5,661,316,237 | 1,415 | 1,326 | |||||||||
Issued under share option schemes |
3,812,482 | 1 | 42 | |||||||||
At 31st December 2009 |
5,665,128,719 | 1,416 | 1,368 | |||||||||
Issued under share option schemes |
5,329,458 | 2 | 60 | |||||||||
At 31st December 2010 |
5,670,458,177 | 1,418 | 1,428 | |||||||||
| 31st December | 31st December | |||||||
| 2010 | 2009 | |||||||
Number (‘000) of shares issuable under outstanding options (Note 42) |
207,132 | 213,110 | ||||||
Number (‘000) of unissued shares not under option |
4,122,410 | 4,121,761 | ||||||
Table of Contents
153
| Net translation exchange included in: | ||||||||||||||||
| Non- | Total | |||||||||||||||
| Fair value | Retained | controlling | translation | |||||||||||||
| reserve | earnings | interests | exchange | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
At 1st January 2008 |
9 | 335 | (75 | ) | 269 | |||||||||||
Exchange movements on overseas net assets |
1 | 952 | 64 | 1,017 | ||||||||||||
Reclassification of exchange on liquidation of overseas subsidiary |
– | 84 | – | 84 | ||||||||||||
At 31st December 2008 |
10 | 1,371 | (11 | ) | 1,370 | |||||||||||
Exchange movements on overseas net assets |
1 | (161 | ) | (34 | ) | (194 | ) | |||||||||
Reclassification of exchange on liquidation of overseas subsidiary |
– | (44 | ) | – | (44 | ) | ||||||||||
At 31st December 2009 |
11 | 1,166 | (45 | ) | 1,132 | |||||||||||
Exchange movements on overseas net assets |
– | 145 | 21 | 166 | ||||||||||||
Reclassification of exchange on liquidation or disposal of overseas subsidiaries |
– | (2 | ) | – | (2 | ) | ||||||||||
At 31st December 2010 |
11 | 1,309 | (24 | ) | 1,296 | |||||||||||
| ESOP Trust | Fair value | Cash flow | Other | |||||||||||||||||
| shares | reserve | hedge reserve | reserves | Total | ||||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
At 1st January 2008 |
(1,617 | ) | 49 | (7 | ) | 1,934 | 359 | |||||||||||||
Transferred to income and expense in the year on disposals |
– | (32 | ) | – | – | (32 | ) | |||||||||||||
Transferred to income and expense in the year on impairment |
– | (2 | ) | – | – | (2 | ) | |||||||||||||
Net fair value movement in the year |
– | (23 | ) | 4 | – | (19 | ) | |||||||||||||
Ordinary Shares purchased and cancelled |
– | – | – | 90 | 90 | |||||||||||||||
Ordinary Shares acquired by ESOP Trusts |
(19 | ) | – | – | – | (19 | ) | |||||||||||||
Ordinary Shares transferred by ESOP Trusts |
10 | – | – | – | 10 | |||||||||||||||
Write-down of shares held by ESOP Trusts |
181 | – | – | – | 181 | |||||||||||||||
At 31st December 2008 |
(1,445 | ) | (8 | ) | (3 | ) | 2,024 | 568 | ||||||||||||
Transferred to income and expense in the year on disposals |
– | (40 | ) | 1 | – | (39 | ) | |||||||||||||
Transferred to income and expense in the year on impairment |
– | 40 | – | – | 40 | |||||||||||||||
Net fair value movement in the year |
– | 30 | (4 | ) | – | 26 | ||||||||||||||
Ordinary Shares acquired by ESOP Trusts |
(57 | ) | – | – | – | (57 | ) | |||||||||||||
Ordinary Shares transferred by ESOP Trusts |
13 | – | – | – | 13 | |||||||||||||||
Write-down of shares held by ESOP Trusts |
351 | – | – | – | 351 | |||||||||||||||
Put option over non-controlling interest |
– | – | – | (2 | ) | (2 | ) | |||||||||||||
At 31st December 2009 |
(1,138 | ) | 22 | (6 | ) | 2,022 | 900 | |||||||||||||
Transferred to income and expense in the year on disposals |
– | (5 | ) | 3 | – | (2 | ) | |||||||||||||
Transferred to income and expense in the year on impairment |
– | 5 | – | – | 5 | |||||||||||||||
Net fair value movement in the year |
– | 67 | (1 | ) | – | 66 | ||||||||||||||
Ordinary Shares acquired by ESOP Trusts |
(16 | ) | – | – | – | (16 | ) | |||||||||||||
Ordinary Shares transferred by ESOP Trusts |
17 | – | – | – | 17 | |||||||||||||||
Write-down of shares held by ESOP Trusts |
292 | – | – | – | 292 | |||||||||||||||
At 31st December 2010 |
(845 | ) | 89 | (4 | ) | 2,022 | 1,262 | |||||||||||||
Table of Contents
154
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Profit after tax |
1,853 | 5,669 | 4,712 | |||||||||
Tax on profits |
1,304 | 2,222 | 1,947 | |||||||||
Share of after tax profits of associates and joint ventures |
(81 | ) | (64 | ) | (48 | ) | ||||||
Finance income net of finance costs |
715 | 713 | 530 | |||||||||
Depreciation |
1,146 | 1,130 | 920 | |||||||||
Amortisation of intangible assets |
533 | 432 | 311 | |||||||||
Impairment and assets written off |
411 | 445 | 436 | |||||||||
Profit on sale of intangible assets |
(118 | ) | (835 | ) | (170 | ) | ||||||
Profit on sale of investments in associates |
(8 | ) | (115 | ) | – | |||||||
Profit on sale of equity investments |
(17 | ) | (40 | ) | (33 | ) | ||||||
Changes in working capital: |
||||||||||||
Decrease/(increase) in inventories |
238 | (132 | ) | (411 | ) | |||||||
Decrease/(increase) in trade receivables |
905 | (473 | ) | 519 | ||||||||
Decrease/(increase) in other receivables |
6 | (134 | ) | 22 | ||||||||
Increase/(decrease) in trade payables |
154 | 499 | (39 | ) | ||||||||
(Decrease)/increase in other payables |
(179 | ) | 409 | (162 | ) | |||||||
Increase/(decrease) in pension and other provisions |
1,653 | (320 | ) | 548 | ||||||||
Share-based incentive plans |
179 | 179 | 241 | |||||||||
Other |
(63 | ) | (40 | ) | (268 | ) | ||||||
| 6,778 | 3,876 | 4,343 | ||||||||||
Cash generated from operations |
8,631 | 9,545 | 9,055 | |||||||||
Table of Contents
155
| 2010 | 2009 | 2008 | ||||||||||
| £m | £m | £m | ||||||||||
Net debt at beginning of year |
(9,444 | ) | (10,173 | ) | (6,039 | ) | ||||||
(Decrease)/increase in cash and bank overdrafts |
(642 | ) | 1,054 | 1,148 | ||||||||
Cash inflow from liquid investments |
(91 | ) | (87 | ) | (905 | ) | ||||||
Net increase in long-term loans |
– | (1,358 | ) | (5,523 | ) | |||||||
Net repayment of short-term loans |
1,290 | 102 | 3,059 | |||||||||
Net repayment of obligations under finance leases |
45 | 48 | 48 | |||||||||
Debt of subsidiary undertakings acquired |
(20 | ) | (9 | ) | – | |||||||
Exchange adjustments |
61 | 1,041 | (1,918 | ) | ||||||||
Other non-cash movements |
(58 | ) | (62 | ) | (43 | ) | ||||||
Movement in net debt |
585 | 729 | (4,134 | ) | ||||||||
Net debt at end of year |
(8,859 | ) | (9,444 | ) | (10,173 | ) | ||||||
| Analysis of changes in net debt | At 31.12.09 | Exchange | Other | Reclassifications | Acquisitions | Cash flow | At 31.12.10 | |||||||||||||||||||||
| £m | £m | £m | £m | £m | £m | £m | ||||||||||||||||||||||
Liquid investments |
268 | 7 | – | – | – | (91 | ) | 184 | ||||||||||||||||||||
Cash and cash equivalents |
6,545 | 77 | – | – | 12 | (577 | ) | 6,057 | ||||||||||||||||||||
Overdrafts |
(177 | ) | 4 | – | – | – | (77 | ) | (250 | ) | ||||||||||||||||||
| 6,368 | 81 | – | – | 12 | (654 | ) | 5,807 | |||||||||||||||||||||
Debt due within one year: |
||||||||||||||||||||||||||||
Commercial paper |
(621 | ) | – | – | – | – | 621 | – | ||||||||||||||||||||
Eurobonds and Medium-Term Notes |
(621 | ) | (24 | ) | – | – | – | 645 | – | |||||||||||||||||||
Other |
(52 | ) | (1 | ) | (18 | ) | (15 | ) | (20 | ) | 65 | (41 | ) | |||||||||||||||
| (1,294 | ) | (25 | ) | (18 | ) | (15 | ) | (20 | ) | 1,331 | (41 | ) | ||||||||||||||||
Debt due after one year: |
||||||||||||||||||||||||||||
Eurobonds, Medium-Term Notes and
private financing |
(14,696 | ) | (1 | ) | (40 | ) | – | – | – | (14,737 | ) | |||||||||||||||||
Other |
(90 | ) | (1 | ) | – | 15 | – | 4 | (72 | ) | ||||||||||||||||||
| (14,786 | ) | (2 | ) | (40 | ) | 15 | – | 4 | (14,809 | ) | ||||||||||||||||||
Net debt |
(9,444 | ) | 61 | (58 | ) | – | (8 | ) | 590 | (8,859 | ) | |||||||||||||||||
Table of Contents
156
| Fair value | ||||||||||||
| Book value | adjustments | Fair value | ||||||||||
| £m | £m | £m | ||||||||||
Net assets acquired |
||||||||||||
Intangible assets |
– | 121 | 121 | |||||||||
Property, plant and equipment |
6 | 10 | 16 | |||||||||
Other assets including cash and cash equivalents |
39 | 7 | 46 | |||||||||
Deferred tax provision |
(1 | ) | (41 | ) | (42 | ) | ||||||
Other liabilities |
(27 | ) | (12 | ) | (39 | ) | ||||||
| 17 | 85 | 102 | ||||||||||
Goodwill |
– | 72 | 72 | |||||||||
Total consideration |
17 | 157 | 174 | |||||||||
| Fair value | ||||||||||||
| Book value | adjustments | Fair value | ||||||||||
| £m | £m | £m | ||||||||||
Net assets acquired |
||||||||||||
Intangible assets |
3 | 128 | 131 | |||||||||
Property, plant and equipment |
9 | 2 | 11 | |||||||||
Other assets including cash and cash equivalents |
20 | 12 | 32 | |||||||||
Deferred tax provision |
– | (33 | ) | (33 | ) | |||||||
Other liabilities |
(10 | ) | – | (10 | ) | |||||||
| 22 | 109 | 131 | ||||||||||
Goodwill |
– | 75 | 75 | |||||||||
Fair value gain recognised on conversion of associate to subsidiary |
– | (8 | ) | (8 | ) | |||||||
Total consideration |
22 | 176 | 198 | |||||||||
Table of Contents
157
| Associates | ||||||||||||||||
| Other | and joint | |||||||||||||||
| Cash flows | Phoenix | acquisitions | ventures | Total | ||||||||||||
| £m | £m | £m | £m | |||||||||||||
Total cash consideration |
174 | 198 | 61 | 433 | ||||||||||||
Cash and cash equivalents acquired |
(11 | ) | (1 | ) | – | (12 | ) | |||||||||
Cash consideration, net of cash acquired |
163 | 197 | 61 | 421 | ||||||||||||
Net cash consideration paid |
163 | 191 | 61 | 415 | ||||||||||||
Deferred consideration |
– | 6 | – | 6 | ||||||||||||
Cash consideration, net of cash acquired |
163 | 197 | 61 | 421 | ||||||||||||
| Book | Fair value | Fair | ||||||||||
| value | adjustment | value | ||||||||||
| £m | £m | £m | ||||||||||
Net assets acquired |
||||||||||||
Intangible assets |
417 | 28 | 445 | |||||||||
Property, plant and equipment |
1 | – | 1 | |||||||||
Cash and cash equivalents |
5 | – | 5 | |||||||||
Deferred tax provision |
– | (56 | ) | (56 | ) | |||||||
Other liabilities |
(5 | ) | – | (5 | ) | |||||||
| 418 | (28 | ) | 390 | |||||||||
Goodwill |
– | 87 | 87 | |||||||||
Total consideration |
418 | 59 | 477 | |||||||||
Table of Contents
158
| Book | Fair value | Fair | ||||||||||
| value | adjustment | value | ||||||||||
| £m | £m | £m | ||||||||||
Net assets acquired |
||||||||||||
Intangible assets |
274 | 1,239 | 1,513 | |||||||||
Property, plant and equipment |
111 | – | 111 | |||||||||
Other assets including cash and cash equivalents |
210 | 47 | 257 | |||||||||
Deferred tax provision |
35 | (331 | ) | (296 | ) | |||||||
Other liabilities |
(251 | ) | – | (251 | ) | |||||||
| 379 | 955 | 1,334 | ||||||||||
Goodwill |
– | 885 | 885 | |||||||||
Total consideration |
379 | 1,840 | 2,219 | |||||||||
Table of Contents
159
| Book | Fair value | Fair | ||||||||||
| value | adjustment | value | ||||||||||
| £m | £m | £m | ||||||||||
Net assets acquired |
||||||||||||
Intangible assets |
13 | 582 | 595 | |||||||||
Other assets including cash and cash equivalents |
10 | 11 | 21 | |||||||||
Deferred tax provision |
– | (172 | ) | (172 | ) | |||||||
| 23 | 421 | 444 | ||||||||||
Non-controlling interests |
– | (316 | ) | (316 | ) | |||||||
Goodwill |
– | 255 | 255 | |||||||||
Total consideration |
23 | 360 | 383 | |||||||||
Consideration |
||||||||||||
Fair value of assets contributed by GSK |
328 | |||||||||||
Fair value of contingent equity contributed by GSK |
37 | |||||||||||
Direct costs |
18 | |||||||||||
Total consideration |
383 | |||||||||||
| Certain | Stiefel | |||||||||||||||
| businesses | Laboratories, | |||||||||||||||
| Cash flows | of UCB S.A. | Inc. | Other | Total | ||||||||||||
| £m | £m | £m | £m | |||||||||||||
Cash consideration |
477 | 2,067 | 371 | 2,915 | ||||||||||||
Cash and cash equivalents acquired |
(5 | ) | (74 | ) | (15 | ) | (94 | ) | ||||||||
Net cash consideration |
472 | 1,993 | 356 | 2,821 | ||||||||||||
Contingent consideration |
– | 152 | 2 | 154 | ||||||||||||
Net purchase consideration |
472 | 2,145 | 358 | 2,975 | ||||||||||||
Table of Contents
160
| Book | Fair value | Fair | ||||||||||
| value | adjustment | value | ||||||||||
| £m | £m | £m | ||||||||||
Net assets acquired |
||||||||||||
Intangible assets |
– | 106 | 106 | |||||||||
Property, plant and equipment |
2 | – | 2 | |||||||||
Other assets including cash and cash equivalents |
86 | – | 86 | |||||||||
Deferred tax provision |
– | (21 | ) | (21 | ) | |||||||
Other liabilities |
(39 | ) | – | (39 | ) | |||||||
| 49 | 85 | 134 | ||||||||||
Goodwill |
– | 242 | 242 | |||||||||
Total consideration |
49 | 327 | 376 | |||||||||
| Cash flows | Sirtris | Other | Total | |||||||||
| £m | £m | £m | ||||||||||
Cash consideration |
376 | 139 | 515 | |||||||||
Cash and cash equivalents acquired |
(52 | ) | – | (52 | ) | |||||||
Net cash payment on acquisitions |
324 | 139 | 463 | |||||||||
Table of Contents
161
| Contractual obligations and commitments | 2010 | 2009 | ||||||
| £m | £m | |||||||
Contracted for but not provided in the financial statements: |
||||||||
Intangible assets |
11,762 | 12,280 | ||||||
Property, plant and equipment |
380 | 416 | ||||||
Investments |
37 | 86 | ||||||
Purchase commitments |
1,127 | 82 | ||||||
Business combinations |
285 | – | ||||||
Pensions |
1,095 | 1,460 | ||||||
Other commitments |
242 | 52 | ||||||
Interest on loans |
10,312 | 10,733 | ||||||
Finance lease charges |
16 | 16 | ||||||
| 25,256 | 25,125 | |||||||
| Commitments under non-cancellable operating leases | 2010 | 2009 | ||||||
| £m | £m | |||||||
Rental payments due within one year |
123 | 111 | ||||||
Rental payments due between one and two years |
73 | 72 | ||||||
Rental payments due between two and three years |
46 | 50 | ||||||
Rental payments due between three and four years |
32 | 21 | ||||||
Rental payments due between four and five years |
25 | 14 | ||||||
Rental payments due after five years |
116 | 69 | ||||||
Total commitments under non-cancellable operating leases |
415 | 337 | ||||||
Table of Contents
162
Table of Contents
163
| Credit rating of counterparty | ||||||||||||||||||||||||||||||||||||||||
| 2010 | Aaa/AAA | Aa2/AA | Aa3/AA- | A1/A+ | A2/A | Baa1/BBB+ | Baa2/BBB | Baa3/BBB- | Ba1/BB+ | Total | ||||||||||||||||||||||||||||||
| £m | £m | £m | £m | £m | £m | £m | £m | £m | £m | |||||||||||||||||||||||||||||||
Bank balances and deposits |
– | 1,772 | 1,226 | 2,494 | 67 | 1 | – | 84 | 16 | 5,660 | ||||||||||||||||||||||||||||||
US Treasury and Treasury repo only
money market funds |
360 | – | – | – | – | – | – | – | – | 360 | ||||||||||||||||||||||||||||||
Corporate debt instruments |
– | – | 10 | – | – | – | – | – | – | 10 | ||||||||||||||||||||||||||||||
Government securities |
192 | – | – | – | – | – | – | 8 | 11 | 211 | ||||||||||||||||||||||||||||||
3rd party financial derivatives |
– | 23 | 49 | 100 | – | – | – | – | – | 172 | ||||||||||||||||||||||||||||||
Total |
552 | 1,795 | 1,285 | 2,594 | 67 | 1 | – | 92 | 27 | 6,413 | ||||||||||||||||||||||||||||||
| Credit rating of counterparty | ||||||||||||||||||||||||||||||||||||||||
| 2009 | Aaa/AAA | Aa2/AA | Aa3/AA- | A1/A+ | A2/A | Baa1/BBB+ | Baa2/BBB | Baa3/BBB- | Ba1/BB+ | Total | ||||||||||||||||||||||||||||||
| £m | £m | £m | £m | £m | £m | £m | £m | £m | £m | |||||||||||||||||||||||||||||||
Bank balances and deposits |
793 | 1,385 | 1,359 | 1,467 | 102 | – | 27 | 63 | 10 | 5,206 | ||||||||||||||||||||||||||||||
US Treasury and Treasury repo only
money market funds |
1,305 | – | – | – | – | – | – | – | – | 1,305 | ||||||||||||||||||||||||||||||
Corporate debt instruments |
– | – | 10 | – | – | – | – | – | – | 10 | ||||||||||||||||||||||||||||||
Government securities |
237 | – | – | 43 | – | – | – | 11 | 1 | 292 | ||||||||||||||||||||||||||||||
3rd party financial derivatives |
– | 48 | 32 | 106 | – | – | – | – | – | 186 | ||||||||||||||||||||||||||||||
Total |
2,335 | 1,433 | 1,401 | 1,616 | 102 | – | 27 | 74 | 11 | 6,999 | ||||||||||||||||||||||||||||||
Table of Contents
164
continued
| • | Cash and cash equivalents – approximates to the carrying amount | |
| • | Liquid investments – based on quoted market prices or calculated based on observable inputs in the case of marketable securities; based on principal amounts in the case of non-marketable securities because of their short repricing periods | |
| • | Other investments – investments traded in an active market determined by reference to the relevant stock exchange quoted bid price; other investments determined by reference to the current market value of similar instruments or by reference to the discounted cash flows of the underlying net assets | |
| • | Short-term loans and overdrafts – approximates to the carrying amount because of the short maturity of these instruments | |
| • | Long-term loans – based on quoted market prices in the case of the Eurobonds and other fixed rate borrowings; approximates to the carrying amount in the case of floating rate bank loans and other loans | |
| • | Forward exchange contracts – based on market data and exchange rates at the balance sheet date | |
| • | Currency swaps – based on market data at the balance sheet date | |
| • | Interest rate swaps – based on the net present value of discounted cash flows | |
| • | Receivables and payables – approximates to the carrying amount | |
| • | Company-owned life insurance policies – based on cash surrender value | |
| • | Lease obligations – approximates to the carrying amount. |
(2009 – $3.9 billion (£2.4 billion)) of 364 days duration, renewable annually. At 31st December 2010 these were undrawn.
Table of Contents
165
| 2010 | 2009 | |||||||||||||||
| Carrying | Fair | Carrying | Fair | |||||||||||||
| value | value | value | value | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Cash and cash equivalents |
6,057 | 6,057 | 6,545 | 6,545 | ||||||||||||
Available-for-sale investments: |
||||||||||||||||
Liquid investments: |
||||||||||||||||
– Government bonds |
172 | 172 | 254 | 254 | ||||||||||||
– other |
12 | 12 | 14 | 14 | ||||||||||||
Total liquid investments |
184 | 184 | 268 | 268 | ||||||||||||
Other investments |
711 | 711 | 454 | 454 | ||||||||||||
Loans and receivables: |
||||||||||||||||
Trade and other receivables and certain Other non-current assets in scope of IAS 39 |
5,667 | 5,667 | 6,271 | 6,271 | ||||||||||||
Financial assets at fair value through profit or loss: |
||||||||||||||||
Other non-current assets |
187 | 187 | 153 | 153 | ||||||||||||
Held-for-trading financial assets: |
||||||||||||||||
Derivatives designated as accounting hedges |
97 | 97 | 104 | 104 | ||||||||||||
Other derivatives |
93 | 93 | 93 | 93 | ||||||||||||
Total financial assets |
12,996 | 12,996 | 13,888 | 13,888 | ||||||||||||
Financial liabilities measured at amortised cost: |
||||||||||||||||
Borrowings: |
||||||||||||||||
– bonds in a designated hedging relationship |
(6,029 | ) | (6,401 | ) | (6,139 | ) | (6,499 | ) | ||||||||
– other bonds |
(8,708 | ) | (9,653 | ) | (9,178 | ) | (9,864 | ) | ||||||||
– commercial paper |
– | – | (621 | ) | (621 | ) | ||||||||||
– bank loans and overdrafts |
(260 | ) | (260 | ) | (182 | ) | (182 | ) | ||||||||
– other loans and private financing |
– | – | (7 | ) | (7 | ) | ||||||||||
– obligations under finance leases |
(103 | ) | (103 | ) | (130 | ) | (130 | ) | ||||||||
Total borrowings |
(15,100 | ) | (16,417 | ) | (16,257 | ) | (17,303 | ) | ||||||||
Trade and
other payables, Other provisions and Other non-current
liabilities in scope of IAS 39 |
(6,590 | ) | (6,590 | ) | (6,051 | ) | (6,051 | ) | ||||||||
Held-for-trading financial liabilities: |
||||||||||||||||
Derivatives designated as accounting hedges |
(23 | ) | (23 | ) | (55 | ) | (55 | ) | ||||||||
Other derivatives |
(170 | ) | (170 | ) | (113 | ) | (113 | ) | ||||||||
Total financial liabilities |
(21,883 | ) | (23,200 | ) | (22,476 | ) | (23,522 | ) | ||||||||
Net financial assets and financial liabilities |
(8,887 | ) | (10,204 | ) | (8,588 | ) | (9,634 | ) | ||||||||
Table of Contents
166
| The following tables categorise the Group’s financial assets and liabilities held at fair value by the valuation methodology applied in determining their fair value. Where possible, quoted prices in active markets are used (Level 1). Where such prices are not available, the asset or liability is classified as Level 2, provided all significant inputs to the valuation model used are based on observable market data. If one or more of the significant inputs to the valuation model is not based on observable market data, the instrument is classified as Level 3. |
| At 31st December 2010 | Level 1 | Level 2 | Level 3 | Total | ||||||||||||
| £m | £m | £m | £m | |||||||||||||
Financial assets at fair value |
||||||||||||||||
Available–for–sale financial assets: |
||||||||||||||||
Liquid investments |
159 | 25 | – | 184 | ||||||||||||
Other investments |
491 | – | 220 | 711 | ||||||||||||
Financial assets at fair value through profit or loss: |
||||||||||||||||
Other non-current assets |
– | 187 | – | 187 | ||||||||||||
Held–for–trading financial assets: |
||||||||||||||||
Derivatives designated as accounting hedges |
– | 97 | – | 97 | ||||||||||||
Other derivatives |
– | 92 | 1 | 93 | ||||||||||||
| 650 | 401 | 221 | 1,272 | |||||||||||||
Financial liabilities at fair value |
||||||||||||||||
Held–for–trading financial liabilities: |
||||||||||||||||
Derivatives designated as accounting hedges |
– | (23 | ) | – | (23 | ) | ||||||||||
Other derivatives |
– | (169 | ) | (1 | ) | (170 | ) | |||||||||
| – | (192 | ) | (1 | ) | (193 | ) | ||||||||||
| At 31st December 2009 | Level 1 | Level 2 | Level 3 | Total | ||||||||||||
| £m | £m | £m | £m | |||||||||||||
Financial assets at fair value |
||||||||||||||||
Available–for–sale financial assets: |
||||||||||||||||
Liquid investments |
249 | 19 | – | 268 | ||||||||||||
Other investments |
245 | – | 209 | 454 | ||||||||||||
Financial assets at fair value through profit or loss: |
||||||||||||||||
Other non-current assets |
– | 153 | – | 153 | ||||||||||||
Held–for–trading financial assets: |
||||||||||||||||
Derivatives designated as accounting hedges |
– | 104 | – | 104 | ||||||||||||
Other derivatives |
– | 93 | – | 93 | ||||||||||||
| 494 | 369 | 209 | 1,072 | |||||||||||||
Financial liabilities at fair value |
||||||||||||||||
Held–for–trading financial liabilities: |
||||||||||||||||
Derivatives designated as accounting hedges |
– | (55 | ) | – | (55 | ) | ||||||||||
Other derivatives |
– | (113 | ) | – | (113 | ) | ||||||||||
| – | (168 | ) | – | (168 | ) | |||||||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
At 1st January |
209 | 159 | ||||||
Losses recognised in the income statement |
(13 | ) | (11 | ) | ||||
(Losses)/gains recognised in other comprehensive income |
(1 | ) | 1 | |||||
Additions |
51 | 81 | ||||||
Disposals |
(3 | ) | (4 | ) | ||||
Transfers from Level 3 |
(26 | ) | – | |||||
Exchange |
3 | (17 | ) | |||||
At 31st December |
220 | 209 | ||||||
Table of Contents
167
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Trade and other receivables (Note 24) |
5,793 | 6,492 | ||||||
Other non-current assets (Note 22) |
556 | 583 | ||||||
| 6,349 | 7,075 | |||||||
Analysed as: |
||||||||
Financial assets in scope of IAS 39 |
5,854 | 6,424 | ||||||
Other assets |
495 | 651 | ||||||
| 6,349 | 7,075 | |||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Past due by 1–30 days |
134 | 262 | ||||||
Past due by 31–90 days |
138 | 105 | ||||||
Past due by 91–180 days |
61 | 60 | ||||||
Past due by 181–365 days |
66 | 54 | ||||||
Past due by more than 365 days |
67 | 78 | ||||||
| 466 | 559 | |||||||
| 2010 | 2009 | |||||||
| £m | £m | |||||||
Trade and other payables (Note 27) |
(6,888 | ) | (6,772 | ) | ||||
Other provisions (Note 29) |
(5,284 | ) | (3,241 | ) | ||||
Other non-current liabilities (Note 30) |
(594 | ) | (605 | ) | ||||
| (12,766 | ) | (10,618 | ) | |||||
Analysed as: |
||||||||
Financial liabilities in scope of IAS 39 |
(6,590 | ) | (6,051 | ) | ||||
Other liabilities |
(6,176 | ) | (4,567 | ) | ||||
| (12,766 | ) | (10,618 | ) | |||||
Table of Contents
168
| 2010 | 2009 | |||||||||||||||||||||||
| Effect of | Effect of | |||||||||||||||||||||||
| interest | interest | |||||||||||||||||||||||
| Debt | rate swaps | Total | Debt | rate swaps | Total | |||||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||||
Floating and fixed rate debt less than one year |
(259 | ) | (1,049 | ) | (1,308 | ) | (1,431 | ) | (990 | ) | (2,421 | ) | ||||||||||||
Between one and two years |
(2,559 | ) | – | (2,559 | ) | – | – | – | ||||||||||||||||
Between two and three years |
(1,599 | ) | – | (1,599 | ) | (2,647 | ) | – | (2,647 | ) | ||||||||||||||
Between three and four years |
(1,049 | ) | 1,049 | – | (1,548 | ) | – | (1,548 | ) | |||||||||||||||
Between four and five years |
(1,358 | ) | – | (1,358 | ) | (990 | ) | 990 | – | |||||||||||||||
Between five and ten years |
(2,819 | ) | – | (2,819 | ) | (4,205 | ) | – | (4,205 | ) | ||||||||||||||
Greater than ten years |
(5,354 | ) | – | (5,354 | ) | (5,306 | ) | – | (5,306 | ) | ||||||||||||||
Total |
(14,997 | ) | – | (14,997 | ) | (16,127 | ) | – | (16,127 | ) | ||||||||||||||
Original issuance profile: |
||||||||||||||||||||||||
Fixed rate interest |
(14,757 | ) | 1,049 | (13,708 | ) | (14,696 | ) | 990 | (13,706 | ) | ||||||||||||||
Floating rate interest |
(239 | ) | (1,049 | ) | (1,288 | ) | (1,430 | ) | (990 | ) | (2,420 | ) | ||||||||||||
Total interest bearing |
(14,996 | ) | – | (14,996 | ) | (16,126 | ) | – | (16,126 | ) | ||||||||||||||
Non-interest bearing |
(1 | ) | – | (1 | ) | (1 | ) | – | (1 | ) | ||||||||||||||
| (14,997 | ) | – | (14,997 | ) | (16,127 | ) | – | (16,127 | ) | |||||||||||||||
| 2010 | 2009 | |||||||||||||||
| Increase | Reduction | Increase | Reduction | |||||||||||||
| Non-functional currency foreign exchange exposure | in income | in equity | in income | in equity | ||||||||||||
| £m | £m | £m | £m | |||||||||||||
20% appreciation of the US dollar |
386 | – | 214 | 755 | ||||||||||||
20% appreciation of the Euro |
35 | 1,697 | 72 | 1,779 | ||||||||||||
20% appreciation of the Yen |
– | – | 5 | 45 | ||||||||||||
Table of Contents
169
| 2010 | 2009 | |||||||
| Increase/(decrease) | Increase/(decrease) | |||||||
| Impact of foreign exchange movements on net debt | in net debt | in net debt | ||||||
| £m | £m | |||||||
20% appreciation of the US dollar |
851 | 523 | ||||||
20% appreciation of the Euro |
606 | 686 | ||||||
20% appreciation of the Yen |
(13 | ) | 89 | |||||
| 2010 | 2009 | |||||||
| Increase/(decrease) | Increase/(decrease) | |||||||
| in income | in income | |||||||
| £m | £m | |||||||
2% (200 basis points) increase in Sterling interest rates |
29 | (2 | ) | |||||
2% (200 basis points) increase in US dollar interest rates |
(18 | ) | 38 | |||||
2% (200 basis points) increase in Euro interest rates |
37 | 18 | ||||||
| Finance charge | Trade payables and | |||||||||||||||||||||||
| Obligations | on obligations | other | ||||||||||||||||||||||
| Interest on | under finance | under finance | liabilities not | |||||||||||||||||||||
| At 31st December 2010 | Debt | debt | leases | leases | in net debt | Total | ||||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||||
Due in less than one year |
(259 | ) | (755 | ) | (32 | ) | (5 | ) | (6,280 | ) | (7,331 | ) | ||||||||||||
Between one and two years |
(2,564 | ) | (756 | ) | (27 | ) | (5 | ) | (178 | ) | (3,530 | ) | ||||||||||||
Between two and three years |
(1,603 | ) | (638 | ) | (18 | ) | (3 | ) | (35 | ) | (2,297 | ) | ||||||||||||
Between three and four years |
(962 | ) | (559 | ) | (11 | ) | (2 | ) | (57 | ) | (1,591 | ) | ||||||||||||
Between four and five years |
(1,368 | ) | (538 | ) | (7 | ) | (1 | ) | (7 | ) | (1,921 | ) | ||||||||||||
Between five and ten years |
(2,831 | ) | (2,053 | ) | (8 | ) | – | (21 | ) | (4,913 | ) | |||||||||||||
Greater than ten years |
(5,425 | ) | (5,013 | ) | – | – | (12 | ) | (10,450 | ) | ||||||||||||||
Gross contractual cash flows |
(15,012 | ) | (10,312 | ) | (103 | ) | (16 | ) | (6,590 | ) | (32,033 | ) | ||||||||||||
| Finance charge | Trade payables and | |||||||||||||||||||||||
| Obligations | on obligations | other | ||||||||||||||||||||||
| Interest on | under finance | under finance | liabilities not | |||||||||||||||||||||
| At 31st December 2009 | Debt | debt | leases | leases | in net debt | Total | ||||||||||||||||||
| £m | £m | £m | £m | £m | £m | |||||||||||||||||||
Due in less than one year |
(1,431 | ) | (757 | ) | (40 | ) | (4 | ) | (5,828 | ) | (8,060 | ) | ||||||||||||
Between one and two years |
– | (753 | ) | (32 | ) | (6 | ) | (161 | ) | (952 | ) | |||||||||||||
Between two and three years |
(2,655 | ) | (754 | ) | (24 | ) | (2 | ) | (28 | ) | (3,463 | ) | ||||||||||||
Between three and four years |
(1,553 | ) | (594 | ) | (14 | ) | (2 | ) | (14 | ) | (2,177 | ) | ||||||||||||
Between four and five years |
(932 | ) | (536 | ) | (5 | ) | (1 | ) | (5 | ) | (1,479 | ) | ||||||||||||
Between five and ten years |
(4,230 | ) | (2,088 | ) | (15 | ) | (1 | ) | (15 | ) | (6,349 | ) | ||||||||||||
Greater than ten years |
(5,382 | ) | (5,251 | ) | – | – | – | (10,633 | ) | |||||||||||||||
Gross contractual cash flows |
(16,183 | ) | (10,733 | ) | (130 | ) | (16 | ) | (6,051 | ) | (33,113 | ) | ||||||||||||
Table of Contents
170
| 2010 | 2009 | |||||||||||||||
| Receivables | Payables | Receivables | Payables | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Less than one year |
13,555 | (13,511 | ) | 21,341 | (21,318 | ) | ||||||||||
Between one and two years |
288 | (365 | ) | 72 | (51 | ) | ||||||||||
Between two and three years |
31 | (10 | ) | 285 | (321 | ) | ||||||||||
Between three and four years |
14 | (7 | ) | 21 | (19 | ) | ||||||||||
Between four and five years |
– | – | 10 | (11 | ) | |||||||||||
Greater than five years |
– | – | – | – | ||||||||||||
Gross contractual cash flows |
13,888 | (13,893 | ) | 21,729 | (21,720 | ) | ||||||||||
| 2010 | 2009 | |||||||||||||||
| Fair value | Fair value | |||||||||||||||
| Assets | Liabilities | Assets | Liabilities | |||||||||||||
| £m | £m | £m | £m | |||||||||||||
Fair value hedges – Interest rate swaps (principal amount – £962 million (2009 – £932 million)) |
97 | – | 68 | – | ||||||||||||
Net investment hedges – Foreign exchange contracts (principal amount – £3,506 million (2009 – £7,756 million)) |
– | (23 | ) | 36 | (55 | ) | ||||||||||
Derivatives designated as accounting hedges |
97 | (23 | ) | 104 | (55 | ) | ||||||||||
Foreign exchange contracts (principal amount – £6,474 million (2009 – £8,568 million)) |
88 | (160 | ) | 89 | (108 | ) | ||||||||||
Embedded and other derivatives |
5 | (10 | ) | 4 | (5 | ) | ||||||||||
Derivatives not designated as accounting hedges |
93 | (170 | ) | 93 | (113 | ) | ||||||||||
Total derivative instruments |
190 | (193 | ) | 197 | (168 | ) | ||||||||||
Analysed as: |
||||||||||||||||
Current |
93 | (188 | ) | 129 | (168 | ) | ||||||||||
Non-current |
97 | (5 | ) | 68 | – | |||||||||||
Total |
190 | (193 | ) | 197 | (168 | ) | ||||||||||
Table of Contents
171
| 2010 | 2009 | 2008 | ||||||||||
Risk-free interest rate |
0.8% – 1.9 | % | 1.4% – 2.9 | % | 1.3% – 4.8 | % | ||||||
Dividend yield |
5.3 | % | 5.2 | % | 4.8 | % | ||||||
Volatility |
26% – 29 | % | 23% – 29 | % | 19% – 24 | % | ||||||
Expected lives of options granted under: |
||||||||||||
Share option schemes |
5 years | 5 years | 5 years | |||||||||
Savings-related share option and share award schemes |
3-4 years | 3-4 years | 3 years | |||||||||
Weighted average share price for grants in the year: |
||||||||||||
Ordinary Shares |
£12.04 | £11.72 | £11.59 | |||||||||
ADS |
$37.29 | $33.73 | $45.02 | |||||||||
Table of Contents
172
| Options outstanding | Share option | Share option | Savings-related | |||||||||||||||||||||||||
| schemes – shares | schemes – ADS | share option schemes | ||||||||||||||||||||||||||
| Weighted | Weighted | Weighted | Weighted | Weighted | Weighted | |||||||||||||||||||||||
| Number | exercise | fair | Number | exercise | fair | Number | exercise | fair | ||||||||||||||||||||
| 000 | price | value | 000 | price | value | 000 | price | value | ||||||||||||||||||||
At 1st January 2008 |
149,041 | £15.38 | 77,274 | $49.91 | 8,538 | £11.02 | ||||||||||||||||||||||
Options granted |
11,314 | £11.50 | £1.32 | 7,690 | $44.89 | $3.84 | 5,570 | £9.51 | £2.56 | |||||||||||||||||||
Options exercised |
(2,198 | ) | £11.84 | (1,989 | ) | $42.18 | (453 | ) | £10.26 | |||||||||||||||||||
Options lapsed |
(21,602 | ) | £16.52 | (7,497 | ) | $53.13 | (2,401 | ) | £10.67 | |||||||||||||||||||
At 31st December 2008 |
136,555 | £14.93 | 75,478 | $49.29 | 11,254 | £10.38 | ||||||||||||||||||||||
Options granted |
11,393 | £11.76 | £1.16 | 7,741 | $33.68 | $3.41 | 1,648 | £9.72 | £2.22 | |||||||||||||||||||
Options exercised |
(2,660 | ) | £11.80 | (353 | ) | $37.03 | (1,460 | ) | £11.34 | |||||||||||||||||||
Options lapsed |
(21,269 | ) | £17.18 | (9,447 | ) | $55.64 | (3,377 | ) | £11.09 | |||||||||||||||||||
At 31st December 2009 |
124,019 | £14.32 | 73,419 | $46.88 | 8,065 | £9.77 | ||||||||||||||||||||||
Options granted |
11,257 | £12.04 | £1.19 | 7,384 | $37.29 | $3.95 | – | – | – | |||||||||||||||||||
Options exercised |
(3,625 | ) | £11.86 | (916 | ) | $36.59 | (1,310 | ) | £10.45 | |||||||||||||||||||
Options lapsed |
(21,551 | ) | £15.10 | (7,776 | ) | $49.62 | (800 | ) | £10.02 | |||||||||||||||||||
At 31st December 2010 |
110,100 | £14.02 | 72,111 | $45.73 | 5,955 | £9.59 | ||||||||||||||||||||||
Range of exercise prices |
£10.76 | – | £19.40 | $33.42 | – | $58.00 | £9.51 | – | £10.50 | |||||||||||||||||||
Weighted average market
price on exercise |
£12.39 | $38.71 | £12.46 | |||||||||||||||||||||||||
Weighted average remaining
contractual life |
4.19 years | 4.39 years | 1.52 years | |||||||||||||||||||||||||
| Options
outstanding at 31st December 2010 |
Share option | Share option | Savings-related | |||||||||||||||||||||||||||||||||
| schemes – shares | schemes – ADS | share option schemes | ||||||||||||||||||||||||||||||||||
| Weighted | Latest | Weighted | Latest | Weighted | Latest | |||||||||||||||||||||||||||||||
| Number | exercise | exercise | Number | exercise | exercise | Number | Exercise | exercise | ||||||||||||||||||||||||||||
| Year of grant | 000 | price | date | 000 | price | date | 000 | price | date | |||||||||||||||||||||||||||
2001 |
29,047 | £18.13 | 29.11.11 | 18,244 | $51.85 | 28.11.11 | – | – | – | |||||||||||||||||||||||||||
2002 |
11,414 | £11.98 | 03.12.12 | 4,727 | $37.68 | 03.12.12 | – | – | – | |||||||||||||||||||||||||||
2003 |
16,256 | £12.67 | 16.12.13 | 9,186 | $43.54 | 16.12.13 | – | – | – | |||||||||||||||||||||||||||
2004 |
5,074 | £11.23 | 03.12.14 | 5,516 | $43.17 | 02.12.14 | – | – | – | |||||||||||||||||||||||||||
2005 |
155 | £13.07 | 02.11.15 | 374 | $47.31 | 02.11.15 | – | – | – | |||||||||||||||||||||||||||
2006 |
6,979 | £14.69 | 28.11.16 | 5,427 | $51.32 | 28.07.16 | – | – | – | |||||||||||||||||||||||||||
2007 |
9,041 | £14.81 | 25.07.17 | 7,061 | $57.54 | 25.07.17 | 175 | £10.50 | 25.04.11 | |||||||||||||||||||||||||||
2008 |
10,306 | £11.50 | 27.07.18 | 7,196 | $44.90 | 05.11.18 | 4,351 | £9.51 | 23.04.12 | |||||||||||||||||||||||||||
2009 |
10,827 | £11.76 | 22.07.19 | 7,228 | $33.68 | 22.07.19 | 1,429 | £9.72 | 22.04.13 | |||||||||||||||||||||||||||
2010 |
11,001 | £12.04 | 21.07.20 | 7,152 | $37.29 | 21.07.20 | – | – | – | |||||||||||||||||||||||||||
Total |
110,100 | £14.02 | 72,111 | $45.73 | 5,995 | £9.59 | ||||||||||||||||||||||||||||||
Options normally become exercisable from three years from the date of grant but may, under certain
circumstances, vest earlier as set out within the various scheme rules.
| ||||||||||||||||||||||||||||||||||||
There has been no change in the effective exercise price of any outstanding options during the
year.
| ||||||||||||||||||||||||||||||||||||
| Options exercisable | Share option | Share option | Savings-related | |||||||||||||||||||||||||||||||||
| schemes – shares | schemes – ADS | share option schemes | ||||||||||||||||||||||||||||||||||
| Weighted | Weighted | Weighted | ||||||||||||||||||||||||||||||||||
| Number | exercise | Number | exercise | Number | exercise | |||||||||||||||||||||||||||||||
| 000 | price | 000 | price | 000 | price | |||||||||||||||||||||||||||||||
At 31st December 2008 |
109,207 | £15.29 | 55,384 | $48.57 | 3,248 | £11.45 | ||||||||||||||||||||||||||||||
At 31st December 2009 |
94,967 | £14.86 | 53,493 | $47.63 | 254 | £11.40 | ||||||||||||||||||||||||||||||
At 31st December 2010 |
81,362 | £14.80 | 53,831 | $48.26 | 175 | £10.50 | ||||||||||||||||||||||||||||||
Table of Contents
173
Performance Share Plan
| Number of shares and ADS issuable | Shares | Weighted | ADS | Weighted | ||||||||||||
| Number (000) | fair value | Number (000) | fair value | |||||||||||||
At 1st January 2008 |
5,731 | 4,327 | ||||||||||||||
Awards granted |
2,834 | £7.77 | 1,467 | $27.99 | ||||||||||||
Awards exercised |
(1,519 | ) | (1,516 | ) | ||||||||||||
Awards cancelled |
(511 | ) | (420 | ) | ||||||||||||
At 31st December 2008 |
6,535 | 3,858 | ||||||||||||||
Awards granted |
3,365 | £8.80 | 1,392 | $29.45 | ||||||||||||
Awards exercised |
(1,270 | ) | (21 | ) | ||||||||||||
Awards cancelled |
(1,024 | ) | (1,497 | ) | ||||||||||||
At 31st December 2009 |
7,606 | 3,732 | ||||||||||||||
Awards granted |
3,812 | £9.13 | 1,624 | $29.91 | ||||||||||||
Awards exercised |
(440 | ) | (386 | ) | ||||||||||||
Awards cancelled |
(2,085 | ) | (1,357 | ) | ||||||||||||
At 31st December 2010 |
8,893 | 3,613 | ||||||||||||||
| Shares | Weighted | ADS | Weighted | |||||||||||||
| Number (000) | fair value | Number (000) | fair value | |||||||||||||
At 1st January 2008 |
9,634 | 8,283 | ||||||||||||||
Awards granted |
5,572 | £9.85 | 4,640 | $36.46 | ||||||||||||
Awards exercised |
(926 | ) | (931 | ) | ||||||||||||
Awards cancelled |
(592 | ) | (630 | ) | ||||||||||||
At 31st December 2008 |
13,688 | 11,362 | ||||||||||||||
Awards granted |
5,572 | £9.86 | 4,291 | $30.53 | ||||||||||||
Awards exercised |
(4,345 | ) | (3,783 | ) | ||||||||||||
Awards cancelled |
(680 | ) | (561 | ) | ||||||||||||
At 31st December 2009 |
14,235 | 11,309 | ||||||||||||||
Awards granted |
5,844 | £10.04 | 4,355 | $31.30 | ||||||||||||
Awards exercised |
(4,993 | ) | (3,939 | ) | ||||||||||||
Awards cancelled |
(834 | ) | (747 | ) | ||||||||||||
At 31st December 2010 |
14,252 | 10,978 | ||||||||||||||
Table of Contents
174
| Number of shares and ADS issuable | Shares | Weighted | ADS | Weighted | ||||||||||||
| Number (000) | fair value | Number (000) | fair value | |||||||||||||
At 1st January 2008 |
224 | 96 | ||||||||||||||
Awards granted |
334 | £11.70 | 70 | $43.80 | ||||||||||||
Awards exercised |
(20 | ) | (20 | ) | ||||||||||||
Awards cancelled |
– | (27 | ) | |||||||||||||
At 31st December 2008 |
538 | 119 | ||||||||||||||
Awards granted |
46 | £12.04 | 132 | $31.94 | ||||||||||||
Awards exercised |
(15 | ) | (32 | ) | ||||||||||||
Awards cancelled |
(20 | ) | (10 | ) | ||||||||||||
At 31st December 2009 |
549 | 209 | ||||||||||||||
Awards granted |
290 | £12.20 | 96 | $36.85 | ||||||||||||
Awards exercised |
(72 | ) | (9 | ) | ||||||||||||
Awards cancelled |
(23 | ) | (16 | ) | ||||||||||||
At 31st December 2010 |
744 | 280 | ||||||||||||||
| Shares held for share award schemes | 2010 | 2009 | ||||||
Number of shares (‘000) |
51,125 | 57,197 | ||||||
| £m | £m | |||||||
Nominal value |
13 | 14 | ||||||
Carrying value |
208 | 217 | ||||||
Market value |
634 | 755 | ||||||
| Shares held for share option schemes | 2010 | 2009 | ||||||
Number of shares (‘000) |
54,347 | 60,538 | ||||||
| £m | £m | |||||||
Nominal value |
14 | 15 | ||||||
Carrying value |
637 | 921 | ||||||
Market value |
674 | 799 | ||||||
Table of Contents
175
| Europe | Location | Subsidiary | Sector | Activity | % | |||||||
England
|
Brentford | +GlaxoSmithKline Holdings Limited | Ph,CH | h | ||||||||
| Brentford | +GlaxoSmithKline Holdings (One) Limited | Ph,CH | h | |||||||||
| Brentford | +GlaxoSmithKline Services Unlimited | Ph,CH | s | |||||||||
| Brentford | +GlaxoSmithKline Mercury Limited | Ph | h | |||||||||
| Brentford | GlaxoSmithKline Finance plc | Ph,CH | f | |||||||||
| Brentford | GlaxoSmithKline Capital plc | Ph,CH | f | |||||||||
| Brentford | SmithKline Beecham Limited | Ph,CH | d e h m p r | |||||||||
| Brentford | Wellcome Limited | Ph,CH | h | |||||||||
| Brentford | Glaxo Group Limited | Ph | h | |||||||||
| Brentford | Glaxo Operations UK Limited | Ph | p | |||||||||
| Brentford | GlaxoSmithKline Export Limited | Ph | e | |||||||||
| Brentford | GlaxoSmithKline Research & Development Limited | Ph | d r | |||||||||
| Brentford | GlaxoSmithKline UK Limited | Ph | m p | |||||||||
| Brentford | Glaxochem Pte Ltd (i) | Ph | h | |||||||||
| Brentford | Setfirst Limited | Ph,CH | h | |||||||||
| Brentford | The Wellcome Foundation Limited | Ph | p | |||||||||
| Cambridge | Domantis Limited | Ph | d r | |||||||||
| Brentford | ViiV Healthcare Limited | Ph | h | 85 | ||||||||
| Brentford | ViiV Healthcare UK Limited | Ph | m s | 85 | ||||||||
| Brentford | ViiV Healthcare Trading Services Limited | Ph | e f | 85 | ||||||||
Austria
|
Vienna | GlaxoSmithKline Pharma GmbH | Ph | m | ||||||||
Belgium
|
Genval | GlaxoSmithKline S.A. | Ph | m | ||||||||
| Rixensart | GlaxoSmithKline Biologicals S.A. | Ph | d e m p r | |||||||||
Czech Republic
|
Prague | GlaxoSmithKline s.r.o. | Ph,CH | m | ||||||||
Denmark
|
Orestadt | GlaxoSmithKline Consumer Healthcare A/S | CH | m | ||||||||
| Brøndby | GlaxoSmithKline Pharma A/S | Ph | m | |||||||||
Finland
|
Espoo | GlaxoSmithKline Oy | Ph | m | ||||||||
France
|
Marly le Roi | Groupe GlaxoSmithKline S.A.S. | Ph | h | ||||||||
| Marly le Roi | Laboratoire GlaxoSmithKline S.A.S. | Ph | m r d | |||||||||
| Marly le Roi | Glaxo Wellcome Production S.A.S. | Ph | p | |||||||||
| Marly le Roi | GlaxoSmithKline Sante Grand Public S.A.S. | CH | m | |||||||||
| Marly le Roi | ViiV Healthcare S.A.S. | Ph | m | 85 | ||||||||
| St. Amand Les Eaux | GlaxoSmithKline Biologicals S.A.S. | Ph | p | |||||||||
Germany
|
Buehl | GlaxoSmithKline Consumer Healthcare GmbH & Co. KG | CH | d h m p r s | ||||||||
| Munich | GlaxoSmithKline GmbH & Co. KG | Ph | d h m s | |||||||||
Greece
|
Athens | GlaxoSmithKline A.E.B.E | Ph,CH | m | ||||||||
Hungary
|
Budapest | GlaxoSmithKline Medicine and Healthcare Products Limited | Ph,CH | e m | ||||||||
Italy
|
Verona | GlaxoSmithKline S.p.A. | Ph | d h m | ||||||||
| Milan | GlaxoSmithKline Consumer Healthcare S.p.A. | CH | m | |||||||||
| Verona | GlaxoSmithKline Manufacturing S.p.A. | Ph | p | |||||||||
Table of Contents
176
| Europe | Location | Subsidiary | Sector | Activity | % | |||||||
Luxembourg
|
Mamer | GlaxoSmithKline International (Luxembourg) S.A.R.L | Ph,CH | f h | ||||||||
Netherlands
|
Zeist | GlaxoSmithKline B.V. | Ph | m | ||||||||
| Zeist | GlaxoSmithKline Consumer Healthcare B.V. | CH | m | |||||||||
Norway
|
Oslo | GlaxoSmithKline AS | Ph | m | ||||||||
Poland
|
Poznan | GlaxoSmithKline Pharmaceuticals S.A. | Ph | p | ||||||||
| Poznan | GSK Services Sp.z o.o. | Ph | m | |||||||||
| Warsaw | GlaxoSmithKline Consumer Healthcare Sp.z o.o. | CH | m e | |||||||||
Portugal
|
Alges | GlaxoSmithKline-Produtos Farmaceuticos, Limitada | Ph | m | ||||||||
Republic of
|
Carrigaline | SmithKline Beecham (Cork) Limited (ii) | Ph | d p r | ||||||||
Ireland
|
Cork | GlaxoSmithKline Trading Services Limited (ii) | Ph | e | ||||||||
| Dublin | GlaxoSmithKline Consumer Healthcare (Ireland) Limited (ii) | CH | m | |||||||||
| Dublin | GlaxoSmithKline (Ireland) Limited | Ph | m | |||||||||
| Dungarvan | Stafford Miller (Ireland) Limited (ii) | CH | p | |||||||||
| Dungarvan | GlaxoSmithKline Dungarvan Limited (ii) | CH | p | |||||||||
Romania
|
Brasov | Europharm Holding S.A. | Ph,CH | s | ||||||||
| Bucharest | GlaxoSmithKline (GSK) S.R.L. | Ph | m r s | |||||||||
Russian
|
Moscow | GlaxoSmithKline Trading ZAO | Ph | m | ||||||||
Federation
|
Moscow | GlaxoSmithKline Healthcare ZAO | CH | m | ||||||||
Spain
|
Madrid | GlaxoSmithKline S.A. | Ph | m | ||||||||
| Madrid | GlaxoSmithKline Consumer Healthcare S.A. | CH | m | |||||||||
| Aranda de Duero | Glaxo Wellcome, S.A. | Ph | p | |||||||||
Sweden
|
Solna | GlaxoSmithKline AB | Ph | m | ||||||||
Switzerland
|
Muenchenbuchsee | GlaxoSmithKline AG | Ph | m | ||||||||
| USA | ||||||||||||
USA
|
Research Triangle Park | Stiefel Laboratories, Inc. | Ph | h m p | ||||||||
| Marietta | Corixa Corporation | Ph | m p | |||||||||
| Philadelphia | GlaxoSmithKline LLC | Ph,CH | d e h m p r s | |||||||||
| Pittsburgh | GlaxoSmithKline Consumer Healthcare, L.P. | CH | m p | 88 | ||||||||
| Pittsburgh | Block Drug Company, Inc. | CH | h m | |||||||||
| Wilmington | GlaxoSmithKline Holdings (Americas) Inc. | Ph,CH | h | |||||||||
| Wilmington | GlaxoSmithKline Capital Inc. | Ph | f | |||||||||
| Cambridge | Sirtris Pharmaceuticals Inc. | Ph | r | |||||||||
| Research Triangle Park | ViiV Healthcare Company | Ph | m | 85 | ||||||||
| Americas | ||||||||||||
Bermuda
|
Hamilton | GlaxoSmithKline Insurance Ltd | Ph,CH | i | ||||||||
Canada
|
Mississauga | GlaxoSmithKline Inc. | Ph | m p r | ||||||||
| Mississauga | GlaxoSmithKline Consumer Healthcare Inc. | CH | m | |||||||||
| Laval | ID Biomedical Corporation | Ph | h | |||||||||
| Quebec City | ID Biomedical Corporation of Quebec | Ph | d m p r | |||||||||
Mexico
|
Delegacion Tlalpan | GlaxoSmithKline Mexico S.A. de C.V. | Ph,CH | e m p s | ||||||||
Puerto Rico
|
Guaynabo | GlaxoSmithKline Puerto Rico Inc. | Ph | m | ||||||||
| Asia Pacific | ||||||||||||
Australia
|
Boronia | GlaxoSmithKline Australia Pty Ltd | Ph,CH | d e m p r | ||||||||
China
|
Beijing | GlaxoSmithKline (China) Investment Co. Ltd | Ph,CH | d h m s | ||||||||
| Hong Kong | GlaxoSmithKline Limited | Ph,CH | m | |||||||||
| Shanghai | GlaxoSmithKline Biologicals (Shanghai) Ltd | Ph | m p | |||||||||
| Tianjin | Sino-American Tianjin Smith Kline & French Laboratories Ltd | CH | d m p r | 55 | ||||||||
Table of Contents
177
| Asia Pacific | Location | Subsidiary | Sector | Activity | % | |||||||
India
|
Mumbai | GlaxoSmithKline Pharmaceuticals Limited | Ph | m p | 51 | |||||||
| Nabha | GlaxoSmithKline Consumer Healthcare Limited (iii) | CH | d e m p r | 43 | ||||||||
Malaysia
|
Petaling Jaya | GlaxoSmithKline Pharmaceutical Sdn Bhd | Ph | m | ||||||||
| Selangor | GlaxoSmithKline Consumer Healthcare Sdn Bhd | CH | m | |||||||||
New Zealand
|
Auckland | GlaxoSmithKline NZ Limited | Ph,CH | m | ||||||||
Pakistan
|
Karachi | GlaxoSmithKline Pakistan Limited | Ph,CH | m p e | 82 | |||||||
Philippines
|
Makati | GlaxoSmithKline Philippines Inc | Ph,CH | m | ||||||||
Singapore
|
Singapore | Glaxo Wellcome Manufacturing Pte Ltd | Ph | d h p r | ||||||||
| Singapore | GlaxoSmithKline Pte Ltd | Ph,CH | m | |||||||||
South Korea
|
Seoul | GlaxoSmithKline Korea Limited | Ph,CH | m | ||||||||
Thailand
|
Bangkok | GlaxoSmithKline (Thailand) Limited | Ph,CH | m | ||||||||
| Japan | ||||||||||||
Japan
|
Tokyo | GlaxoSmithKline K.K. | Ph,CH | d m p | ||||||||
| Latin America | ||||||||||||
Argentina
|
Buenos Aires | GlaxoSmithKline Argentina S.A. | Ph,CH | d e m p r | ||||||||
| Buenos Aires | Laboratorios Phoenix Sociedad Anonima Industrial Comercial y Financiera | Ph | d e m p | |||||||||
Brazil
|
Rio de Janeiro | GlaxoSmithKline Brasil Limitada | Ph,CH | e m p | ||||||||
Colombia
|
Bogota | GlaxoSmithKline Colombia S.A. | Ph,CH | m | ||||||||
Venezuela
|
Caracas | GlaxoSmithKline Venezuela, C.A. | Ph,CH | m | ||||||||
| Middle East & Africa | ||||||||||||
Egypt
|
Cairo | GlaxoSmithKline S.A.E | Ph | m p | 91 | |||||||
South Africa
|
Johannesburg | GlaxoSmithKline South Africa (Pty) Limited | Ph,CH | e m p | ||||||||
Turkey
|
Istanbul | GlaxoSmithKline llaclari Sanayi ve Ticaret A.S. | Ph,CH | m | ||||||||
| USA | Location | Associate | Sector | Activity | % | |||||||
USA |
Madison | Quest Diagnostics Incorporated (iv) | Clinical testing | 18 | ||||||||
| Middle East & Africa | ||||||||||||
South Africa
|
Johannesburg | Aspen Pharmacare Holdings Limited (iv) | Ph,CH | m p r | 19 | |||||||
| (i) | Incorporated in Singapore. | |
| (ii) | Exempt from the provisions of Section 7 of the Companies (Amendment) Act 1986 (Ireland). | |
| (iii) | Consolidated as a subsidiary undertaking in accordance with Section 1162 (4)(a) of the Companies Act 2006 on the grounds of dominant influence. | |
| (iv) | Equity accounted on the grounds of significant influence. Subsequent to the year-end GSK sold its entire shareholding in Quest Diagnostics Inc. See Note 20 for further details. | |
| + | Directly held wholly owned subsidiary of GlaxoSmithKline plc. |
Business sector:
|
Ph Pharmaceuticals, CH Consumer Healthcare | |
Business activity:
|
d development, e exporting, f finance, h holding company, i insurance, m marketing, p production, r research, s service |
Table of Contents
178
Table of Contents
179
Table of Contents
180
Table of Contents
181
Table of Contents
182
| A number of states, through their respective attorneys general, and most of the counties in New York State have filed civil lawsuits in state and federal courts against the Group and many other drug companies claiming damages and restitution due to AWP and/or WAC price reporting for pharmaceutical products covered by the states’ Medicaid programmes. The states seek recovery on behalf of the states as payers and, in some cases, on behalf of in-state patients as consumers. | ||
| The Group has separately resolved AWP claims by state Medicaid programmes in more than two-thirds of the states through the DOJ Settlement or separate negotiations. Litigation concerning AWP issues is continuing with six states. | ||
| In November 2009, a Kentucky state court jury returned a $661,860 compensatory damages only verdict against the Group in another such case filed by the State of Kentucky. The jury found the Group liable for violating the state’s consumer protection laws, but not liable under the state’s Medicaid fraud and false advertising statutes. In January 2010, the judge in the case awarded the State of Kentucky an additional $5,828,000 in statutory penalties. The Group has settled the case with Kentucky. The judgment was vacated, and the Group denied liability as part of the settlement. |
Table of Contents
183
Table of Contents
184
Table of Contents
185
Table of Contents
186
| Income statement – total | 12 months 2010 | Q4 2010 | ||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||
Turnover – Pharmaceuticals |
23,382 | (2 | ) | (1 | ) | 5,930 | (16 | ) | (14 | ) | ||||||||||||||
– Consumer Healthcare |
5,010 | 5 | 7 | 1,267 | 4 | 7 | ||||||||||||||||||
Total turnover |
28,392 | (1 | ) | – | 7,197 | (13 | ) | (11 | ) | |||||||||||||||
Cost of sales |
(7,592 | ) | 3 | 3 | (2,077 | ) | (2 | ) | (2 | ) | ||||||||||||||
Selling,
general and administration |
(13,053 | ) | 36 | 36 | (4,461 | ) | 48 | 51 | ||||||||||||||||
Research and development |
(4,457 | ) | 8 | 9 | (1,097 | ) | (4 | ) | (3 | ) | ||||||||||||||
Other operating income |
493 | 118 | ||||||||||||||||||||||
Operating profit |
3,783 | (59 | ) | (55 | ) | (320 | ) | (>100 | ) | (>100 | ) | |||||||||||||
Finance income |
116 | 58 | ||||||||||||||||||||||
Finance costs |
(831 | ) | (240 | ) | ||||||||||||||||||||
Profit on disposal of interest in associate |
8 | 8 | ||||||||||||||||||||||
Share of after tax profits of associates and joint ventures |
81 | 18 | ||||||||||||||||||||||
Profit before taxation |
3,157 | (64 | ) | (60 | ) | (476 | ) | (>100 | ) | (>100 | ) | |||||||||||||
Taxation |
(1,304 | ) | (157 | ) | ||||||||||||||||||||
Tax rate % |
41.3 | % | 33.0 | % | ||||||||||||||||||||
Profit after taxation for the period |
1,853 | (71 | ) | (67 | ) | (633 | ) | (>100 | ) | (>100 | ) | |||||||||||||
Profit
attributable to non-controlling interests |
219 | 57 | ||||||||||||||||||||||
Profit attributable to shareholders |
1,634 | (690 | ) | |||||||||||||||||||||
Basic earnings per share (pence) |
32.1 | p | (75 | ) | (71 | ) | (13.6 | )p | (>100 | ) | (>100 | ) | ||||||||||||
Diluted earnings per share (pence) |
31.9 | p | (13.4 | )p | ||||||||||||||||||||
| Income statement – results before major restructuring | ||||||||||||||||||||||||
Total turnover |
28,392 | (1 | ) | – | 7,197 | (13 | ) | (11 | ) | |||||||||||||||
Cost of sales |
(7,405 | ) | 4 | 4 | (1,980 | ) | (6 | ) | (6 | ) | ||||||||||||||
Selling,
general and administration |
(12,388 | ) | 35 | 35 | (4,289 | ) | 51 | 54 | ||||||||||||||||
Research and development |
(3,964 | ) | – | – | (1,083 | ) | (3 | ) | (1 | ) | ||||||||||||||
Other operating income |
493 | 118 | ||||||||||||||||||||||
Operating profit |
5,128 | (48 | ) | (45 | ) | (37 | ) | (>100 | ) | (>100 | ) | |||||||||||||
Finance income |
116 | 58 | ||||||||||||||||||||||
Finance costs |
(828 | ) | (240 | ) | ||||||||||||||||||||
Profit on disposal of interest in associate |
8 | 8 | ||||||||||||||||||||||
Share of after tax profits of associates and joint ventures |
81 | 18 | ||||||||||||||||||||||
Profit before taxation |
4,505 | (52 | ) | (48 | ) | (193 | ) | (>100 | ) | (>100 | ) | |||||||||||||
Taxation |
(1,544 | ) | (134 | ) | ||||||||||||||||||||
Tax rate % |
34.3 | % | 69.4 | % | ||||||||||||||||||||
Profit after taxation for the period |
2,961 | (56 | ) | (53 | ) | (327 | ) | (>100 | ) | (>100 | ) | |||||||||||||
Profit
attributable to non-controlling interests |
219 | 57 | ||||||||||||||||||||||
Profit attributable to shareholders |
2,742 | (384 | ) | |||||||||||||||||||||
Adjusted earnings per share (pence) |
53.9 | p | (59 | ) | (56 | ) | (7.5 | )p | (>100 | ) | (>100 | ) | ||||||||||||
Diluted earnings per share (pence) |
53.5 | p | (7.5 | )p | ||||||||||||||||||||
Table of Contents
187
| Q3 2010 | Q2 2010 | Q1 2010 | ||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | ||||||||||||||||||||||||||
| 5,553 | (3 | ) | (1 | ) | 5,773 | – | 3 | 6,126 | 14 | 9 | ||||||||||||||||||||||||
| 1,260 | 4 | 8 | 1,252 | 3 | 7 | 1,231 | 9 | 7 | ||||||||||||||||||||||||||
| 6,813 | (2 | ) | 1 | 7,025 | – | 4 | 7,357 | 13 | 9 | |||||||||||||||||||||||||
| (1,906 | ) | 7 | 7 | (1,657 | ) | 4 | 2 | (1,952 | ) | 11 | 9 | |||||||||||||||||||||||
| (2,040 | ) | (9 | ) | (5 | ) | (4,202 | ) | 82 | (83 | ) | (2,350 | ) | 17 | 7 | ||||||||||||||||||||
| (1,004 | ) | 11 | 14 | (1,196 | ) | 21 | (23 | ) | (1,160 | ) | 7 | 3 | ||||||||||||||||||||||
| 95 | 81 | 199 | ||||||||||||||||||||||||||||||||
| 1,958 | (10 | ) | (5 | ) | 51 | (106 | ) | (98 | ) | 2,094 | 22 | 22 | ||||||||||||||||||||||
| 22 | 19 | 17 | ||||||||||||||||||||||||||||||||
| (197 | ) | (189 | ) | (205 | ) | |||||||||||||||||||||||||||||
| – | – | – | ||||||||||||||||||||||||||||||||
| 16 | 22 | 25 | ||||||||||||||||||||||||||||||||
| 1,799 | (11 | ) | (6 | ) | (97 | ) | 1,931 | 15 | 16 | |||||||||||||||||||||||||
| (456 | ) | (155 | ) | (536 | ) | |||||||||||||||||||||||||||||
| 25.3 | % | 159.8 | % | 27.8 | % | |||||||||||||||||||||||||||||
| 1,343 | (8 | ) | (2 | ) | (252 | ) | 1,395 | 19 | 19 | |||||||||||||||||||||||||
| 55 | 52 | 55 | ||||||||||||||||||||||||||||||||
| 1,288 | (304 | ) | 1,340 | |||||||||||||||||||||||||||||||
| 25.3 | p | (10 | ) | (4 | ) | (6.0 | )p | (129 | ) | (121 | ) | 26.4 | p | 18 | 18 | |||||||||||||||||||
| 25.1 | p | (5.9 | )p | 26.1 | p | |||||||||||||||||||||||||||||
| 6,813 | (2 | ) | 1 | 7,025 | – | 4 | 7,357 | 13 | 9 | |||||||||||||||||||||||||
| (1,875 | ) | 9 | 8 | (1,626 | ) | (2 | ) | – | (1,924 | ) | 19 | 17 | ||||||||||||||||||||||
| (1,956 | ) | (9 | ) | (5 | ) | (3,845 | ) | 71 | 73 | (2,298 | ) | 18 | 8 | |||||||||||||||||||||
| (948 | ) | 8 | 10 | (994 | ) | 5 | 8 | (939 | ) | (9 | ) | (13 | ) | |||||||||||||||||||||
| 95 | 81 | 199 | ||||||||||||||||||||||||||||||||
| 2,129 | (9 | ) | (4 | ) | 641 | (80 | ) | (73 | ) | 2,395 | 21 | 21 | ||||||||||||||||||||||
| 22 | 19 | 17 | ||||||||||||||||||||||||||||||||
| (196 | ) | (188 | ) | (204 | ) | |||||||||||||||||||||||||||||
| – | – | – | ||||||||||||||||||||||||||||||||
| 16 | 22 | 25 | ||||||||||||||||||||||||||||||||
| 1,971 | (10 | ) | (5 | ) | 494 | (86 | ) | (78 | ) | 2,233 | 16 | 16 | ||||||||||||||||||||||
| (480 | ) | (312 | ) | (618 | ) | |||||||||||||||||||||||||||||
| 24.4 | % | 63.2 | % | 27.7 | % | |||||||||||||||||||||||||||||
| 1,491 | (5 | ) | 1 | 182 | (96 | ) | (89 | ) | 1,615 | 18 | 18 | |||||||||||||||||||||||
| 55 | 52 | 55 | ||||||||||||||||||||||||||||||||
| 1,436 | 130 | 1,560 | ||||||||||||||||||||||||||||||||
| 28.2 | p | (6 | ) | (1 | ) | 2.6 | p | (99 | ) | (92 | ) | 30.7 | p | 16 | 17 | |||||||||||||||||||
| 28.0 | p | 2.5 | p | 30.4 | p | |||||||||||||||||||||||||||||
Table of Contents
188
| Q4 2010 | Q3 2010 | Q2 2010 | Q1 2010 | |||||||||||||||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||
Respiratory |
1,917 | (2 | ) | – | 1,726 | 5 | 8 | 1,829 | 2 | 5 | 1,766 | 6 | 2 | |||||||||||||||||||||||||||||||||||
Avamys/Veramyst |
50 | 45 | 52 | 40 | 23 | 29 | 57 | 19 | 21 | 46 | 52 | 48 | ||||||||||||||||||||||||||||||||||||
Flixonase/Flonase |
37 | 3 | 6 | 32 | 11 | 14 | 50 | 21 | 28 | 45 | (30 | ) | (35 | ) | ||||||||||||||||||||||||||||||||||
Flixotide/Flovent |
220 | (5 | ) | (1 | ) | 187 | 7 | 11 | 201 | 1 | 6 | 196 | 5 | 1 | ||||||||||||||||||||||||||||||||||
Seretide/Advair |
1,346 | (4 | ) | (1 | ) | 1,243 | 5 | 8 | 1,286 | – | 3 | 1,264 | 9 | 4 | ||||||||||||||||||||||||||||||||||
Serevent |
50 | (21 | ) | (18 | ) | 48 | (13 | ) | (11 | ) | 52 | (14 | ) | (12 | ) | 51 | (16 | ) | (18 | ) | ||||||||||||||||||||||||||||
Ventolin |
142 | (1 | ) | 2 | 130 | 15 | 18 | 134 | 16 | 20 | 116 | 3 | – | |||||||||||||||||||||||||||||||||||
Zyrtec |
23 | (5 | ) | 5 | 19 | – | 6 | 20 | 6 | 18 | 20 | 17 | 11 | |||||||||||||||||||||||||||||||||||
Anti-virals |
224 | (64 | ) | (62 | ) | 218 | (68 | ) | (65 | ) | 286 | (50 | ) | (47 | ) | 358 | (44 | ) | (46 | ) | ||||||||||||||||||||||||||||
Hepsera |
33 | – | 10 | 32 | 7 | 14 | 34 | 7 | 17 | 29 | 11 | 7 | ||||||||||||||||||||||||||||||||||||
Relenza |
11 | (96 | ) | (96 | ) | 18 | (91 | ) | (90 | ) | 8 | (97 | ) | (87 | ) | 84 | (60 | ) | (62 | ) | ||||||||||||||||||||||||||||
Valtrex |
96 | (60 | ) | (57 | ) | 95 | (75 | ) | (73 | ) | 165 | (59 | ) | (56 | ) | 176 | (46 | ) | (49 | ) | ||||||||||||||||||||||||||||
Zeffix |
64 | 9 | 16 | 55 | (4 | ) | 2 | 62 | 7 | 13 | 52 | 4 | (2 | ) | ||||||||||||||||||||||||||||||||||
Central nervous system |
450 | (14 | ) | (1 | ) | 436 | 1 | 4 | 450 | (4 | ) | – | 417 | (13 | ) | (16 | ) | |||||||||||||||||||||||||||||||
Imigran/Imitrex |
50 | (40 | ) | (38 | ) | 53 | (4 | ) | – | 52 | (25 | ) | (24 | ) | 57 | (9 | ) | (11 | ) | |||||||||||||||||||||||||||||
Lamictal |
130 | (3 | ) | (2 | ) | 131 | 7 | 8 | 123 | 15 | 19 | 120 | (11 | ) | (17 | ) | ||||||||||||||||||||||||||||||||
Requip |
60 | (9 | ) | (8 | ) | 58 | 33 | 35 | 60 | 16 | 18 | 55 | 14 | 10 | ||||||||||||||||||||||||||||||||||
Seroxat/Paxil |
128 | (15 | ) | (8 | ) | 115 | (11 | ) | (4 | ) | 133 | (9 | ) | (4 | ) | 106 | (12 | ) | (16 | ) | ||||||||||||||||||||||||||||
Treximet |
14 | – | – | 13 | (13 | ) | (13 | ) | 16 | 17 | 33 | 13 | 7 | (7 | ) | |||||||||||||||||||||||||||||||||
Wellbutrin |
22 | (5 | ) | – | 18 | 13 | 13 | 21 | (33 | ) | (30 | ) | 20 | (67 | ) | (69 | ) | |||||||||||||||||||||||||||||||
Cardiovascular and urogenital |
696 | 11 | 13 | 650 | 15 | 18 | 654 | 10 | 13 | 570 | 9 | 3 | ||||||||||||||||||||||||||||||||||||
Arixtra |
80 | 8 | 8 | 72 | 17 | 20 | 79 | 28 | 30 | 70 | 25 | 19 | ||||||||||||||||||||||||||||||||||||
Avodart |
177 | 22 | 24 | 156 | 16 | 19 | 157 | 14 | 17 | 139 | 20 | 14 | ||||||||||||||||||||||||||||||||||||
Coreg |
41 | 29 | 32 | 44 | 8 | 13 | 44 | (16 | ) | (14 | ) | 42 | (12 | ) | (18 | ) | ||||||||||||||||||||||||||||||||
Fraxiparine |
55 | (8 | ) | (8 | ) | 54 | – | (4 | ) | 57 | (3 | ) | (2 | ) | 56 | 4 | 2 | |||||||||||||||||||||||||||||||
Lovaza |
147 | 11 | 14 | 138 | 20 | 24 | 138 | 29 | 33 | 107 | 9 | 1 | ||||||||||||||||||||||||||||||||||||
Vesicare |
31 | 3 | 7 | 28 | 8 | 12 | 30 | 12 | 15 | 25 | 13 | 4 | ||||||||||||||||||||||||||||||||||||
Volibris |
16 | >100 | >100 | 11 | 100 | 83 | 10 | >100 | >100 | 9 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Metabolic |
110 | (65 | ) | (63 | ) | 125 | (58 | ) | (56 | ) | 213 | (33 | ) | (30 | ) | 230 | (18 | ) | (22 | ) | ||||||||||||||||||||||||||||
Avandia products |
49 | (76 | ) | (74 | ) | 70 | (65 | ) | (62 | ) | 152 | (26 | ) | (23 | ) | 169 | (10 | ) | (14 | ) | ||||||||||||||||||||||||||||
Bonviva/Boniva |
18 | (73 | ) | (73 | ) | 17 | (70 | ) | (72 | ) | 20 | (70 | ) | (70 | ) | 23 | (63 | ) | (63 | ) | ||||||||||||||||||||||||||||
Anti-bacterials |
370 | (2 | ) | (2 | ) | 333 | (4 | ) | (2 | ) | 337 | (4 | ) | (3 | ) | 356 | (6 | ) | (9 | ) | ||||||||||||||||||||||||||||
Augmentin |
168 | (2 | ) | (3 | ) | 153 | (7 | ) | (6 | ) | 144 | (3 | ) | (1 | ) | 160 | (10 | ) | (14 | ) | ||||||||||||||||||||||||||||
Oncology and emesis |
172 | – | 1 | 172 | 13 | 15 | 175 | 2 | 5 | 169 | 23 | 17 | ||||||||||||||||||||||||||||||||||||
Arzerra |
9 | >100 | >100 | 9 | – | – | 8 | – | – | 5 | – | – | ||||||||||||||||||||||||||||||||||||
Hycamtin |
29 | (36 | ) | (36 | ) | 35 | (15 | ) | (15 | ) | 40 | (12 | ) | (7 | ) | 40 | – | (7 | ) | |||||||||||||||||||||||||||||
Promacta |
10 | 80 | 100 | 7 | >100 | >100 | 8 | >100 | >100 | 6 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Tyverb/Tykerb |
60 | 23 | 25 | 58 | 26 | 26 | 56 | 32 | 37 | 53 | 62 | 56 | ||||||||||||||||||||||||||||||||||||
Votrient |
14 | >100 | >100 | 11 | – | – | 8 | – | – | 5 | – | – | ||||||||||||||||||||||||||||||||||||
Vaccines |
994 | (36 | ) | (35 | ) | 982 | 19 | 22 | 939 | 17 | 24 | 1,411 | >100 | >100 | ||||||||||||||||||||||||||||||||||
Boostrix |
49 | 37 | 40 | 59 | 49 | 51 | 43 | 8 | 10 | 30 | 19 | 15 | ||||||||||||||||||||||||||||||||||||
Cervarix |
67 | 68 | 76 | 48 | 64 | 71 | 50 | (33 | ) | (32 | ) | 77 | 60 | 60 | ||||||||||||||||||||||||||||||||||
Fluarix, FluLaval |
69 | 64 | 64 | 167 | 14 | 14 | – | – | – | 5 | (43 | ) | (29 | ) | ||||||||||||||||||||||||||||||||||
Flu pandemic |
161 | (82 | ) | (81 | ) | 58 | >100 | >100 | 275 | >100 | >100 | 698 | >100 | >100 | ||||||||||||||||||||||||||||||||||
Hepatitis |
164 | 5 | 9 | 189 | 9 | 11 | 170 | (16 | ) | (13 | ) | 197 | 38 | 32 | ||||||||||||||||||||||||||||||||||
Infanrix, Pediarix |
190 | 24 | 24 | 168 | 1 | 1 | 176 | 14 | 14 | 166 | (3 | ) | (5 | ) | ||||||||||||||||||||||||||||||||||
Rotarix |
79 | 11 | 13 | 52 | (40 | ) | (38 | ) | 39 | (49 | ) | (45 | ) | 65 | 19 | 14 | ||||||||||||||||||||||||||||||||
Synflorix |
48 | (2 | ) | – | 90 | >100 | >100 | 38 | >100 | >100 | 45 | – | – | |||||||||||||||||||||||||||||||||||
Dermatologicals |
288 | 10 | 13 | 272 | 20 | 24 | 262 | >100 | >100 | 265 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Bactroban |
29 | 4 | 4 | 33 | (3 | ) | – | 30 | (6 | ) | (6 | ) | 27 | (7 | ) | (10 | ) | |||||||||||||||||||||||||||||||
Dermovate |
22 | – | – | 18 | – | – | 19 | – | – | 15 | – | – | ||||||||||||||||||||||||||||||||||||
Duac |
27 | 13 | 17 | 33 | 39 | 43 | 29 | – | – | 27 | – | – | ||||||||||||||||||||||||||||||||||||
Soriatane |
17 | – | 6 | 19 | 58 | 58 | 17 | – | – | 18 | – | – | ||||||||||||||||||||||||||||||||||||
Zovirax |
39 | (3 | ) | 3 | 31 | 11 | 15 | 33 | (6 | ) | – | 49 | 61 | 58 | ||||||||||||||||||||||||||||||||||
Other |
306 | 19 | 21 | 238 | 14 | 15 | 239 | 12 | 15 | 211 | 20 | 17 | ||||||||||||||||||||||||||||||||||||
| 5,527 | (17 | ) | (15 | ) | 5,152 | (4 | ) | (1 | ) | 5,384 | – | 4 | 5,753 | 15 | 11 | |||||||||||||||||||||||||||||||||
VIIV Healthcare (HIV) |
403 | (4 | ) | (3 | ) | 401 | (1 | ) | 2 | 389 | 1 | 3 | 373 | (7 | ) | (11 | ) | |||||||||||||||||||||||||||||||
Combivir |
99 | (11 | ) | (9 | ) | 96 | (10 | ) | (6 | ) | 86 | (18 | ) | (16 | ) | 82 | (23 | ) | (27 | ) | ||||||||||||||||||||||||||||
Epivir |
29 | (3 | ) | (3 | ) | 31 | (15 | ) | (9 | ) | 27 | (13 | ) | (13 | ) | 28 | (15 | ) | (18 | ) | ||||||||||||||||||||||||||||
Epzicom/Kivexa |
146 | (3 | ) | (2 | ) | 138 | 3 | 5 | 140 | 8 | 9 | 131 | (1 | ) | (4 | ) | ||||||||||||||||||||||||||||||||
Lexiva |
36 | (18 | ) | (18 | ) | 39 | (12 | ) | (9 | ) | 39 | (12 | ) | (9 | ) | 41 | (8 | ) | (15 | ) | ||||||||||||||||||||||||||||
Selzentry |
22 | >100 | >100 | 20 | – | – | 19 | – | – | 19 | – | – | ||||||||||||||||||||||||||||||||||||
Trizivir |
32 | (35 | ) | (35 | ) | 38 | (21 | ) | (19 | ) | 36 | (29 | ) | (27 | ) | 38 | (27 | ) | (32 | ) | ||||||||||||||||||||||||||||
| 5,930 | (16 | ) | (14 | ) | 5,553 | (3 | ) | (1 | ) | 5,773 | – | 3 | 6,126 | 14 | 9 | |||||||||||||||||||||||||||||||||
Table of Contents
189
| Q4 2010 | Q3 2010 | Q2 2010 | Q1 2010 | |||||||||||||||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||
Respiratory |
875 | (6 | ) | (4 | ) | 846 | 9 | 14 | 868 | 1 | 5 | 805 | 3 | (5 | ) | |||||||||||||||||||||||||||||||||
Avamys/Veramyst |
17 | 7 | 13 | 15 | (7 | ) | – | 20 | 11 | 11 | 17 | (10 | ) | (15 | ) | |||||||||||||||||||||||||||||||||
Flixonase/Flonase |
5 | (17 | ) | (17 | ) | 8 | >100 | >100 | 18 | >100 | >100 | 6 | (40 | ) | (40 | ) | ||||||||||||||||||||||||||||||||
Flixotide/Flovent |
118 | – | 3 | 105 | 19 | 24 | 109 | 8 | 12 | 99 | 8 | – | ||||||||||||||||||||||||||||||||||||
Seretide/Advair |
670 | (7 | ) | (5 | ) | 649 | 6 | 11 | 655 | (3 | ) | 1 | 630 | 4 | (4 | ) | ||||||||||||||||||||||||||||||||
Serevent |
15 | (25 | ) | (25 | ) | 16 | (6 | ) | – | 17 | (6 | ) | (6 | ) | 16 | (11 | ) | (16 | ) | |||||||||||||||||||||||||||||
Ventolin |
49 | – | 2 | 50 | 37 | 43 | 45 | 38 | 41 | 35 | – | (8 | ) | |||||||||||||||||||||||||||||||||||
Zyrtec |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Anti-virals |
41 | (82 | ) | (81 | ) | 57 | (84 | ) | (83 | ) | 118 | (66 | ) | (64 | ) | 154 | (42 | ) | (47 | ) | ||||||||||||||||||||||||||||
Hepsera |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Relenza |
(5 | ) | – | – | 13 | (73 | ) | (71 | ) | 5 | (84 | ) | (74 | ) | 30 | >100 | >100 | |||||||||||||||||||||||||||||||
Valtrex |
24 | (83 | ) | (81 | ) | 27 | (91 | ) | (90 | ) | 94 | (69 | ) | (68 | ) | 107 | (55 | ) | (58 | ) | ||||||||||||||||||||||||||||
Zeffix |
3 | (25 | ) | (25 | ) | 3 | (25 | ) | (25 | ) | 4 | (40 | ) | (20 | ) | 3 | – | (25 | ) | |||||||||||||||||||||||||||||
Central nervous system |
114 | (37 | ) | (36 | ) | 124 | 3 | 8 | 131 | (11 | ) | (8 | ) | 136 | (32 | ) | (37 | ) | ||||||||||||||||||||||||||||||
Imigran/Imitrex |
15 | (65 | ) | (65 | ) | 18 | (11 | ) | (5 | ) | 18 | (45 | ) | (45 | ) | 24 | (11 | ) | (14 | ) | ||||||||||||||||||||||||||||
Lamictal |
66 | (11 | ) | (8 | ) | 70 | 5 | 9 | 60 | 29 | 33 | 61 | (23 | ) | (29 | ) | ||||||||||||||||||||||||||||||||
Requip |
10 | (38 | ) | (38 | ) | 13 | >100 | >100 | 11 | 83 | 83 | 10 | 38 | 25 | ||||||||||||||||||||||||||||||||||
Seroxat/Paxil |
– | (100 | ) | (100 | ) | 5 | – | – | 12 | (8 | ) | (8 | ) | 10 | (29 | ) | (29 | ) | ||||||||||||||||||||||||||||||
Treximet |
14 | – | – | 13 | (13 | ) | (13 | ) | 15 | 25 | 25 | 13 | – | (7 | ) | |||||||||||||||||||||||||||||||||
Wellbutrin |
7 | (30 | ) | (30 | ) | 4 | (25 | ) | – | 5 | (70 | ) | (75 | ) | 8 | (85 | ) | (85 | ) | |||||||||||||||||||||||||||||
Cardiovascular and urogenital |
422 | 10 | 13 | 409 | 17 | 22 | 403 | 8 | 12 | 337 | 6 | (2 | ) | |||||||||||||||||||||||||||||||||||
Arixtra |
49 | 12 | 14 | 43 | 28 | 34 | 46 | 36 | 39 | 39 | 27 | 18 | ||||||||||||||||||||||||||||||||||||
Avodart |
86 | 1 | 4 | 87 | 5 | 9 | 88 | 2 | 6 | 76 | 12 | 4 | ||||||||||||||||||||||||||||||||||||
Coreg |
41 | 29 | 32 | 43 | 8 | 10 | 44 | (16 | ) | (12 | ) | 42 | (12 | ) | (18 | ) | ||||||||||||||||||||||||||||||||
Fraxiparine |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Lovaza |
146 | 11 | 14 | 137 | 20 | 25 | 138 | 29 | 33 | 107 | 10 | 2 | ||||||||||||||||||||||||||||||||||||
Vesicare |
31 | 3 | 7 | 27 | 4 | 8 | 30 | 12 | 15 | 25 | 13 | 4 | ||||||||||||||||||||||||||||||||||||
Volibris |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Metabolic |
40 | (75 | ) | (73 | ) | 32 | (78 | ) | (76 | ) | 77 | (51 | ) | (48 | ) | 89 | (36 | ) | (41 | ) | ||||||||||||||||||||||||||||
Avandia products |
40 | (65 | ) | (63 | ) | 33 | (70 | ) | (66 | ) | 75 | (33 | ) | (30 | ) | 89 | (14 | ) | (21 | ) | ||||||||||||||||||||||||||||
Bonviva/Boniva |
– | (100 | ) | (100 | ) | – | (100 | ) | (100 | ) | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||
Anti-bacterials |
16 | (38 | ) | (33 | ) | 14 | (41 | ) | (36 | ) | 21 | (29 | ) | (25 | ) | 24 | (10 | ) | (17 | ) | ||||||||||||||||||||||||||||
Augmentin |
– | (100 | ) | (100 | ) | 1 | (89 | ) | (89 | ) | 2 | (91 | ) | (82 | ) | 8 | (44 | ) | (50 | ) | ||||||||||||||||||||||||||||
Oncology and emesis |
75 | (16 | ) | (13 | ) | 89 | 34 | 39 | 94 | 2 | 7 | 92 | 41 | 31 | ||||||||||||||||||||||||||||||||||
Arzerra |
6 | >100 | 100 | 8 | – | – | 7 | – | – | 5 | – | – | ||||||||||||||||||||||||||||||||||||
Hycamtin |
14 | (46 | ) | (46 | ) | 21 | (17 | ) | (13 | ) | 24 | (4 | ) | – | 24 | – | (8 | ) | ||||||||||||||||||||||||||||||
Promacta |
6 | 20 | 20 | 6 | 100 | 100 | 7 | >100 | >100 | 6 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Tyverb/Tykerb |
17 | 14 | 21 | 18 | 42 | 50 | 18 | – | 6 | 17 | 73 | 55 | ||||||||||||||||||||||||||||||||||||
Votrient |
11 | >100 | >100 | 9 | – | – | 8 | – | – | 5 | – | – | ||||||||||||||||||||||||||||||||||||
Vaccines |
171 | (44 | ) | (42 | ) | 278 | 32 | 35 | 143 | (31 | ) | (27 | ) | 171 | 55 | 44 | ||||||||||||||||||||||||||||||||
Boostrix |
28 | 65 | 65 | 41 | 67 | 71 | 26 | 19 | 24 | 15 | 55 | 36 | ||||||||||||||||||||||||||||||||||||
Cervarix |
1 | (75 | ) | (75 | ) | 4 | – | – | 6 | – | – | 2 | – | – | ||||||||||||||||||||||||||||||||||
Fluarix, FluLaval |
28 | >100 | >100 | 81 | 29 | 29 | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||
Flu pandemic |
– | (100 | ) | (100 | ) | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||
Hepatitis |
56 | 6 | 10 | 97 | 39 | 45 | 62 | (33 | ) | (29 | ) | 92 | 92 | 77 | ||||||||||||||||||||||||||||||||||
Infanrix, Pediarix |
36 | 30 | 33 | 38 | 23 | 27 | 40 | 3 | 5 | 32 | (13 | ) | (18 | ) | ||||||||||||||||||||||||||||||||||
Rotarix |
21 | 18 | 24 | 17 | (27 | ) | (23 | ) | 9 | (64 | ) | (59 | ) | 27 | 93 | 80 | ||||||||||||||||||||||||||||||||
Synflorix |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Dermatologicals |
94 | 1 | 3 | 93 | 22 | 26 | 74 | >100 | >100 | 97 | 100 | >100 | ||||||||||||||||||||||||||||||||||||
Bactroban |
12 | (14 | ) | (14 | ) | 14 | (7 | ) | (7 | ) | 14 | (13 | ) | (7 | ) | 11 | (20 | ) | (27 | ) | ||||||||||||||||||||||||||||
Dermovate |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Duac |
15 | (7 | ) | – | 20 | 27 | 33 | 15 | – | – | 17 | – | – | |||||||||||||||||||||||||||||||||||
Soriatane |
17 | – | 6 | 19 | 58 | 58 | 17 | – | – | 18 | – | – | ||||||||||||||||||||||||||||||||||||
Zovirax |
14 | 8 | 8 | 8 | 75 | 100 | 5 | 25 | 25 | 26 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Other |
6 | >100 | 100 | 8 | 14 | 14 | 6 | >100 | >100 | 4 | (20 | ) | (20 | ) | ||||||||||||||||||||||||||||||||||
| 1,854 | (22 | ) | (20 | ) | 1,950 | (8 | ) | (4 | ) | 1,935 | (13 | ) | (10 | ) | 1,909 | (1 | ) | (9 | ) | |||||||||||||||||||||||||||||
VIIV Healthcare (HIV) |
163 | (16 | ) | (14 | ) | 162 | (8 | ) | (4 | ) | 176 | 3 | 7 | 159 | (11 | ) | (18 | ) | ||||||||||||||||||||||||||||||
Combivir |
34 | (30 | ) | (28 | ) | 36 | (19 | ) | (16 | ) | 39 | (16 | ) | (11 | ) | 34 | (30 | ) | (36 | ) | ||||||||||||||||||||||||||||
Epivir |
10 | (17 | ) | (17 | ) | 10 | (25 | ) | (17 | ) | 10 | (9 | ) | (9 | ) | 10 | (15 | ) | (23 | ) | ||||||||||||||||||||||||||||
Epzicom/Kivexa |
55 | (16 | ) | (13 | ) | 50 | (8 | ) | (4 | ) | 57 | 10 | 14 | 48 | (10 | ) | (17 | ) | ||||||||||||||||||||||||||||||
Lexiva |
20 | (20 | ) | (20 | ) | 19 | (25 | ) | (21 | ) | 20 | (17 | ) | (13 | ) | 21 | (15 | ) | (22 | ) | ||||||||||||||||||||||||||||
Selzentry |
9 | – | – | 8 | – | – | 9 | – | – | 8 | – | – | ||||||||||||||||||||||||||||||||||||
Trizivir |
16 | (38 | ) | (38 | ) | 19 | (22 | ) | (17 | ) | 19 | (24 | ) | (24 | ) | 19 | (33 | ) | (37 | ) | ||||||||||||||||||||||||||||
Table of Contents
190
| Q4 2010 | Q3 2010 | Q2 2010 | Q1 2010 | |||||||||||||||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||
Respiratory |
557 | (4 | ) | (6 | ) | 488 | (3 | ) | (5 | ) | 535 | (1 | ) | (3 | ) | 569 | 6 | 4 | ||||||||||||||||||||||||||||||
Avamys/Veramyst |
12 | 9 | 9 | 10 | 11 | 11 | 21 | 31 | 31 | 13 | 56 | 44 | ||||||||||||||||||||||||||||||||||||
Flixonase/Flonase |
10 | (10 | ) | – | 8 | – | (11 | ) | 12 | – | – | 10 | (17 | ) | (17 | ) | ||||||||||||||||||||||||||||||||
Flixotide/Flovent |
42 | (10 | ) | (14 | ) | 33 | (11 | ) | (13 | ) | 39 | (9 | ) | (9 | ) | 45 | (6 | ) | (6 | ) | ||||||||||||||||||||||||||||
Seretide/Advair |
416 | (3 | ) | (5 | ) | 370 | – | (2 | ) | 392 | – | (2 | ) | 423 | 10 | 7 | ||||||||||||||||||||||||||||||||
Serevent |
24 | (17 | ) | (17 | ) | 24 | (11 | ) | (11 | ) | 24 | (17 | ) | (17 | ) | 26 | (16 | ) | (16 | ) | ||||||||||||||||||||||||||||
Ventolin |
38 | (7 | ) | (10 | ) | 32 | (9 | ) | (9 | ) | 35 | 3 | (3 | ) | 37 | – | – | |||||||||||||||||||||||||||||||
Zyrtec |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Anti-virals |
24 | (73 | ) | (73 | ) | 24 | (71 | ) | (72 | ) | 26 | (64 | ) | (64 | ) | 35 | (78 | ) | (79 | ) | ||||||||||||||||||||||||||||
Hepsera |
1 | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Relenza |
– | – | – | 2 | (95 | ) | (95 | ) | 2 | (96 | ) | (92 | ) | 2 | (97 | ) | (98 | ) | ||||||||||||||||||||||||||||||
Valtrex |
15 | (61 | ) | (63 | ) | 14 | (63 | ) | (63 | ) | 16 | (59 | ) | (59 | ) | 23 | (43 | ) | (45 | ) | ||||||||||||||||||||||||||||
Zeffix |
6 | (14 | ) | (14 | ) | 7 | (14 | ) | – | 6 | (13 | ) | (25 | ) | 7 | – | – | |||||||||||||||||||||||||||||||
Central nervous system |
132 | (8 | ) | (10 | ) | 131 | (4 | ) | (6 | ) | 137 | (3 | ) | (5 | ) | 140 | (2 | ) | (3 | ) | ||||||||||||||||||||||||||||
Imigran/Imitrex |
20 | (20 | ) | (20 | ) | 22 | – | (4 | ) | 21 | (9 | ) | (9 | ) | 22 | (12 | ) | (12 | ) | |||||||||||||||||||||||||||||
Lamictal |
34 | (10 | ) | (13 | ) | 35 | (8 | ) | (8 | ) | 37 | (3 | ) | (3 | ) | 37 | (3 | ) | (5 | ) | ||||||||||||||||||||||||||||
Requip |
33 | (8 | ) | (11 | ) | 32 | – | (6 | ) | 36 | 6 | 3 | 36 | 13 | 13 | |||||||||||||||||||||||||||||||||
Seroxat/Paxil |
19 | (9 | ) | (14 | ) | 19 | (14 | ) | (14 | ) | 22 | (15 | ) | (19 | ) | 22 | (21 | ) | (21 | ) | ||||||||||||||||||||||||||||
Treximet |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Wellbutrin |
11 | 22 | 22 | 10 | 25 | 25 | 9 | 43 | 29 | 9 | 50 | 50 | ||||||||||||||||||||||||||||||||||||
Cardiovascular and urogenital |
159 | 6 | 3 | 144 | 4 | 1 | 154 | 8 | 6 | 153 | 11 | 9 | ||||||||||||||||||||||||||||||||||||
Arixtra |
24 | – | (8 | ) | 22 | (8 | ) | (8 | ) | 27 | 22 | 17 | 26 | 23 | 18 | |||||||||||||||||||||||||||||||||
Avodart |
50 | 31 | 28 | 41 | 19 | 14 | 44 | 19 | 19 | 40 | 17 | 11 | ||||||||||||||||||||||||||||||||||||
Coreg |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Fraxiparine |
38 | (13 | ) | (16 | ) | 34 | (17 | ) | (19 | ) | 39 | (7 | ) | (9 | ) | 43 | – | – | ||||||||||||||||||||||||||||||
Lovaza |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Vesicare |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Volibris |
13 | 86 | 86 | 10 | 100 | 100 | 9 | >100 | >100 | 8 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Metabolic |
13 | (80 | ) | (81 | ) | 37 | (42 | ) | (45 | ) | 55 | (23 | ) | (23 | ) | 61 | (7 | ) | (10 | ) | ||||||||||||||||||||||||||||
Avandia products |
(4 | ) | – | – | 20 | (52 | ) | (52 | ) | 34 | (22 | ) | (26 | ) | 38 | (12 | ) | (12 | ) | |||||||||||||||||||||||||||||
Bonviva/Boniva |
14 | (35 | ) | (39 | ) | 13 | (41 | ) | (41 | ) | 17 | (22 | ) | (26 | ) | 20 | (5 | ) | (5 | ) | ||||||||||||||||||||||||||||
Anti-bacterials |
153 | (11 | ) | (13 | ) | 121 | (9 | ) | (12 | ) | 120 | (12 | ) | (14 | ) | 142 | (21 | ) | (22 | ) | ||||||||||||||||||||||||||||
Augmentin |
69 | (13 | ) | (16 | ) | 55 | (16 | ) | (19 | ) | 53 | (13 | ) | (13 | ) | 63 | (23 | ) | (25 | ) | ||||||||||||||||||||||||||||
Oncology and emesis |
55 | 10 | 6 | 49 | (2 | ) | (4 | ) | 47 | (4 | ) | (6 | ) | 50 | – | (2 | ) | |||||||||||||||||||||||||||||||
Arzerra |
3 | – | – | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Hycamtin |
12 | (20 | ) | (20 | ) | 11 | (21 | ) | (21 | ) | 12 | (20 | ) | (20 | ) | 13 | (7 | ) | (13 | ) | ||||||||||||||||||||||||||||
Promacta |
3 | – | – | 1 | – | – | 1 | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Tyverb/Tykerb |
25 | 24 | 19 | 23 | 21 | 21 | 22 | 28 | 22 | 24 | 41 | 41 | ||||||||||||||||||||||||||||||||||||
Votrient |
3 | – | – | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Vaccines |
393 | (49 | ) | (51 | ) | 310 | (8 | ) | (10 | ) | 365 | 14 | 14 | 613 | >100 | >100 | ||||||||||||||||||||||||||||||||
Boostrix |
11 | – | – | 12 | 9 | 9 | 11 | 10 | 10 | 9 | 25 | 13 | ||||||||||||||||||||||||||||||||||||
Cervarix |
25 | 37 | 32 | 11 | (35 | ) | (35 | ) | 21 | (65 | ) | (67 | ) | 59 | 51 | 51 | ||||||||||||||||||||||||||||||||
Fluarix, FluLaval |
13 | 18 | 18 | 50 | (13 | ) | (17 | ) | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||
Flu pandemic |
90 | (82 | ) | (82 | ) | 2 | (50 | ) | (50 | ) | 92 | >100 | >100 | 304 | >100 | >100 | ||||||||||||||||||||||||||||||||
Hepatitis |
63 | – | (2 | ) | 55 | (14 | ) | (15 | ) | 63 | (13 | ) | (13 | ) | 61 | 2 | – | |||||||||||||||||||||||||||||||
Infanrix, Pediarix |
120 | 22 | 19 | 96 | (6 | ) | (9 | ) | 109 | 22 | 20 | 104 | (2 | ) | (5 | ) | ||||||||||||||||||||||||||||||||
Rotarix |
9 | (36 | ) | (36 | ) | 8 | (43 | ) | (43 | ) | 8 | (42 | ) | (33 | ) | 13 | 8 | – | ||||||||||||||||||||||||||||||
Synflorix |
8 | (27 | ) | (27 | ) | 9 | (9 | ) | (18 | ) | 14 | 40 | 40 | 12 | – | – | ||||||||||||||||||||||||||||||||
Dermatologicals |
62 | 14 | 11 | 59 | 15 | 13 | 63 | >100 | >100 | 62 | >100 | 100 | ||||||||||||||||||||||||||||||||||||
Bactroban |
7 | 33 | 17 | 7 | – | – | 7 | – | – | 6 | – | – | ||||||||||||||||||||||||||||||||||||
Dermovate |
5 | – | – | 5 | – | – | 5 | – | – | 4 | – | – | ||||||||||||||||||||||||||||||||||||
Duac |
6 | – | – | 5 | 50 | 25 | 6 | – | – | 6 | – | – | ||||||||||||||||||||||||||||||||||||
Soriatane |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Zovirax |
7 | (14 | ) | – | 6 | – | (14 | ) | 7 | (13 | ) | (13 | ) | 7 | (13 | ) | (13 | ) | ||||||||||||||||||||||||||||||
Other |
99 | – | (3 | ) | 65 | (6 | ) | (7 | ) | 78 | 26 | 18 | 68 | 24 | 24 | |||||||||||||||||||||||||||||||||
| 1,647 | (24 | ) | (26 | ) | 1,428 | (9 | ) | (11 | ) | 1,580 | 1 | (1 | ) | 1,893 | 16 | 13 | ||||||||||||||||||||||||||||||||
VIIV Healthcare (HIV) |
145 | (5 | ) | (6 | ) | 136 | (10 | ) | (12 | ) | 145 | (4 | ) | (6 | ) | 159 | (3 | ) | (5 | ) | ||||||||||||||||||||||||||||
Combivir |
28 | (22 | ) | (24 | ) | 26 | (25 | ) | (28 | ) | 30 | (19 | ) | (19 | ) | 33 | (17 | ) | (20 | ) | ||||||||||||||||||||||||||||
Epivir |
8 | (27 | ) | (27 | ) | 9 | (25 | ) | (25 | ) | 10 | (17 | ) | (17 | ) | 10 | (21 | ) | (29 | ) | ||||||||||||||||||||||||||||
Epzicom/Kivexa |
63 | 2 | – | 57 | (2 | ) | (5 | ) | 61 | 5 | 3 | 64 | 6 | 3 | ||||||||||||||||||||||||||||||||||
Lexiva |
10 | (21 | ) | (29 | ) | 13 | (13 | ) | (13 | ) | 13 | (13 | ) | (19 | ) | 15 | (12 | ) | (12 | ) | ||||||||||||||||||||||||||||
Selzentry |
11 | >100 | >100 | 10 | – | – | 9 | – | – | 11 | – | – | ||||||||||||||||||||||||||||||||||||
Trizivir |
14 | (32 | ) | (26 | ) | 15 | (16 | ) | (21 | ) | 14 | (25 | ) | (30 | ) | 17 | (29 | ) | (11 | ) | ||||||||||||||||||||||||||||
Table of Contents
191
| Q4 2010 | Q3 2010 | Q2 2010 | Q1 2010 | |||||||||||||||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||
Respiratory |
162 | 17 | 20 | 145 | 11 | 14 | 166 | 19 | 25 | 143 | 30 | 24 | ||||||||||||||||||||||||||||||||||||
Avamys/Veramyst |
9 | 100 | >100 | 8 | >100 | >100 | 9 | >100 | >100 | 5 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Flixonase/Flonase |
11 | 38 | 38 | 9 | (11 | ) | – | 10 | 10 | – | 9 | 13 | 13 | |||||||||||||||||||||||||||||||||||
Flixotide/Flovent |
12 | 33 | 33 | 10 | – | 11 | 12 | 50 | 50 | 14 | 75 | 75 | ||||||||||||||||||||||||||||||||||||
Seretide/Advair |
85 | 14 | 18 | 77 | 11 | 17 | 86 | 12 | 18 | 80 | 28 | 23 | ||||||||||||||||||||||||||||||||||||
Serevent |
– | (100 | ) | (100 | ) | 1 | – | – | 1 | – | – | – | – | (100 | ) | |||||||||||||||||||||||||||||||||
Ventolin |
30 | 20 | 20 | 27 | 18 | 23 | 31 | 25 | 29 | 24 | 14 | 9 | ||||||||||||||||||||||||||||||||||||
Zyrtec |
4 | – | – | 4 | (33 | ) | (33 | ) | 4 | 33 | 33 | 2 | 100 | 100 | ||||||||||||||||||||||||||||||||||
Anti-virals |
61 | (13 | ) | (9 | ) | 56 | (10 | ) | (7 | ) | 59 | 10 | 13 | 47 | 9 | – | ||||||||||||||||||||||||||||||||
Hepsera |
15 | 8 | 15 | 16 | 25 | 33 | 15 | 8 | 15 | 12 | – | (8 | ) | |||||||||||||||||||||||||||||||||||
Relenza |
(1 | ) | – | – | 1 | (92 | ) | (92 | ) | – | – | (100 | ) | 1 | – | – | ||||||||||||||||||||||||||||||||
Valtrex |
8 | – | – | 7 | – | 17 | 8 | 33 | 33 | 5 | – | (17 | ) | |||||||||||||||||||||||||||||||||||
Zeffix |
39 | 28 | 34 | 32 | 7 | 10 | 36 | 21 | 24 | 29 | 11 | 4 | ||||||||||||||||||||||||||||||||||||
Central nervous system |
62 | 22 | 24 | 63 | 22 | 24 | 53 | 11 | 13 | 45 | 14 | 7 | ||||||||||||||||||||||||||||||||||||
Imigran/Imitrex |
1 | (50 | ) | (50 | ) | 1 | – | – | 2 | 100 | 100 | 1 | – | – | ||||||||||||||||||||||||||||||||||
Lamictal |
16 | 42 | 33 | 15 | 27 | 36 | 14 | 17 | 17 | 12 | 8 | (8 | ) | |||||||||||||||||||||||||||||||||||
Requip |
1 | – | – | 1 | – | – | 1 | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Seroxat/Paxil |
19 | 5 | – | 19 | – | – | 20 | (14 | ) | (5 | ) | 15 | – | (12 | ) | |||||||||||||||||||||||||||||||||
Treximet |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Wellbutrin |
4 | 33 | 33 | 3 | 33 | – | 3 | – | 50 | 3 | 50 | 50 | ||||||||||||||||||||||||||||||||||||
Cardiovascular and urogenital |
36 | 28 | 24 | 34 | 21 | 21 | 36 | 26 | 33 | 28 | 25 | 17 | ||||||||||||||||||||||||||||||||||||
Arixtra |
3 | 50 | 50 | 3 | 50 | 50 | 2 | – | – | 2 | 100 | 100 | ||||||||||||||||||||||||||||||||||||
Avodart |
9 | 50 | 50 | 8 | 33 | 33 | 9 | 60 | 80 | 7 | 60 | 40 | ||||||||||||||||||||||||||||||||||||
Coreg |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Fraxiparine |
15 | 25 | 25 | 15 | 27 | 36 | 14 | 40 | 40 | 11 | 22 | 22 | ||||||||||||||||||||||||||||||||||||
Lovaza |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Vesicare |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Volibris |
– | – | – | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Metabolic |
17 | (43 | ) | (39 | ) | 12 | (61 | ) | (61 | ) | 31 | (9 | ) | (3 | ) | 31 | 18 | 11 | ||||||||||||||||||||||||||||||
Avandia products |
3 | (76 | ) | (82 | ) | 2 | (90 | ) | (90 | ) | 18 | (14 | ) | (14 | ) | 19 | 6 | 6 | ||||||||||||||||||||||||||||||
Bonviva/Boniva |
– | – | (100 | ) | 1 | – | – | 1 | – | – | – | – | – | |||||||||||||||||||||||||||||||||||
Anti-bacterials |
159 | 14 | 15 | 151 | 7 | 9 | 153 | 8 | 12 | 146 | 11 | 4 | ||||||||||||||||||||||||||||||||||||
Augmentin |
78 | 29 | 26 | 74 | 6 | 9 | 69 | 15 | 17 | 70 | 12 | 4 | ||||||||||||||||||||||||||||||||||||
Oncology and emesis |
17 | 13 | 13 | 17 | 6 | 6 | 16 | – | 14 | 12 | 8 | – | ||||||||||||||||||||||||||||||||||||
Arzerra |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Hycamtin |
1 | – | – | 2 | – | – | 2 | – | – | 2 | 100 | 100 | ||||||||||||||||||||||||||||||||||||
Promacta |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Tyverb/Tykerb |
9 | 50 | 50 | 9 | 29 | 29 | 7 | 17 | 17 | 5 | 67 | 67 | ||||||||||||||||||||||||||||||||||||
Votrient |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Vaccines |
260 | 11 | 14 | 216 | 29 | 32 | 179 | 18 | 23 | 272 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Boostrix |
4 | >100 | >100 | 2 | – | – | 1 | (80 | ) | (80 | ) | 2 | 100 | 100 | ||||||||||||||||||||||||||||||||||
Cervarix |
7 | – | – | 8 | 17 | 33 | 6 | 50 | 50 | 4 | (33 | ) | (33 | ) | ||||||||||||||||||||||||||||||||||
Fluarix, FluLaval |
20 | 43 | 43 | 19 | 36 | 36 | 1 | (89 | ) | (89 | ) | – | (100 | ) | (100 | ) | ||||||||||||||||||||||||||||||||
Flu pandemic |
25 | (60 | ) | (55 | ) | 6 | – | – | 43 | – | – | 152 | – | – | ||||||||||||||||||||||||||||||||||
Hepatitis |
23 | 17 | 28 | 20 | (9 | ) | (9 | ) | 25 | 9 | 14 | 20 | 17 | 11 | ||||||||||||||||||||||||||||||||||
Infanrix, Pediarix |
13 | 56 | 44 | 16 | 7 | 7 | 11 | 11 | 22 | 10 | (8 | ) | (17 | ) | ||||||||||||||||||||||||||||||||||
Rotarix |
45 | 36 | 36 | 20 | (56 | ) | (53 | ) | 20 | (34 | ) | (31 | ) | 17 | (25 | ) | (29 | ) | ||||||||||||||||||||||||||||||
Synflorix |
35 | 13 | 13 | 74 | >100 | >100 | 14 | – | – | 26 | – | – | ||||||||||||||||||||||||||||||||||||
Dermatologicals |
82 | 39 | 44 | 73 | 35 | 40 | 69 | 74 | 82 | 62 | 75 | 72 | ||||||||||||||||||||||||||||||||||||
Bactroban |
7 | 17 | 17 | 8 | 14 | 14 | 7 | 14 | – | 6 | (14 | ) | (14 | ) | ||||||||||||||||||||||||||||||||||
Dermovate |
11 | – | – | 7 | – | – | 7 | – | – | 5 | – | – | ||||||||||||||||||||||||||||||||||||
Duac |
3 | >100 | >100 | 3 | 50 | 50 | 2 | – | – | 3 | – | – | ||||||||||||||||||||||||||||||||||||
Soriatane |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Zovirax |
6 | 20 | 20 | 6 | 20 | 20 | 8 | – | 14 | 6 | – | – | ||||||||||||||||||||||||||||||||||||
Other |
113 | 62 | 64 | 106 | 30 | 34 | 86 | 24 | 26 | 80 | 30 | 27 | ||||||||||||||||||||||||||||||||||||
| 969 | 16 | 19 | 873 | 14 | 17 | 848 | 17 | 22 | 866 | 43 | 36 | |||||||||||||||||||||||||||||||||||||
VIIV Healthcare (HIV) |
50 | 57 | 67 | 59 | 73 | 79 | 19 | (17 | ) | (17 | ) | 18 | – | (5 | ) | |||||||||||||||||||||||||||||||||
Combivir |
26 | 67 | 73 | 24 | 69 | 85 | 7 | (36 | ) | (36 | ) | 6 | (33 | ) | (40 | ) | ||||||||||||||||||||||||||||||||
Epivir |
6 | 25 | 50 | 7 | 75 | 75 | 2 | (33 | ) | (33 | ) | 3 | 50 | 50 | ||||||||||||||||||||||||||||||||||
Epzicom/Kivexa |
9 | 14 | 29 | 12 | 86 | 71 | 6 | 50 | 50 | 2 | – | (33 | ) | |||||||||||||||||||||||||||||||||||
Lexiva |
2 | – | (33 | ) | 7 | >100 | >100 | 2 | 50 | – | 2 | >100 | 100 | |||||||||||||||||||||||||||||||||||
Selzentry |
1 | – | – | – | – | – | 1 | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Trizivir |
– | (50 | ) | (100 | ) | 3 | (75 | ) | (25 | ) | – | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||
Table of Contents
192
| Q4 2010 | Q3 2010 | Q2 2010 | Q1 2010 | |||||||||||||||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||
Respiratory |
323 | 5 | 17 | 247 | 5 | 17 | 260 | 2 | 15 | 249 | 4 | 8 | ||||||||||||||||||||||||||||||||||||
Avamys/Veramyst |
12 | >100 | >100 | 7 | 50 | 75 | 7 | (30 | ) | (30 | ) | 11 | >100 | >100 | ||||||||||||||||||||||||||||||||||
Flixonase/Flonase |
11 | – | – | 7 | (14 | ) | – | 10 | (33 | ) | 11 | 20 | (41 | ) | (49 | ) | ||||||||||||||||||||||||||||||||
Flixotide/Flovent |
48 | (16 | ) | (2 | ) | 39 | (3 | ) | 5 | 41 | (15 | ) | – | 38 | (5 | ) | (5 | ) | ||||||||||||||||||||||||||||||
Seretide/Advair |
175 | 2 | 14 | 147 | 11 | 21 | 153 | 10 | 24 | 131 | 23 | 28 | ||||||||||||||||||||||||||||||||||||
Serevent |
11 | (18 | ) | – | 7 | (36 | ) | (36 | ) | 10 | (18 | ) | (9 | ) | 9 | (18 | ) | (18 | ) | |||||||||||||||||||||||||||||
Ventolin |
25 | (13 | ) | 4 | 21 | 11 | 17 | 23 | (5 | ) | 15 | 20 | – | 5 | ||||||||||||||||||||||||||||||||||
Zyrtec |
19 | (6 | ) | 6 | 15 | 17 | 25 | 16 | – | 14 | 18 | 12 | 6 | |||||||||||||||||||||||||||||||||||
Anti-virals |
98 | (59 | ) | (55 | ) | 81 | (55 | ) | (48 | ) | 83 | (14 | ) | (2 | ) | 122 | (27 | ) | (27 | ) | ||||||||||||||||||||||||||||
Hepsera |
17 | (12 | ) | – | 16 | (6 | ) | – | 19 | 6 | 19 | 17 | 21 | 21 | ||||||||||||||||||||||||||||||||||
Relenza |
17 | (89 | ) | (88 | ) | 2 | (98 | ) | (98 | ) | 1 | – | (92 | ) | 51 | (49 | ) | (50 | ) | |||||||||||||||||||||||||||||
Valtrex |
49 | (2 | ) | 11 | 47 | 8 | 18 | 47 | 2 | 9 | 41 | – | 5 | |||||||||||||||||||||||||||||||||||
Zeffix |
16 | (7 | ) | 7 | 13 | (14 | ) | (7 | ) | 16 | 8 | 23 | 13 | (7 | ) | (7 | ) | |||||||||||||||||||||||||||||||
Central nervous system |
142 | (4 | ) | 9 | 118 | (4 | ) | 4 | 129 | (1 | ) | 11 | 96 | 3 | – | |||||||||||||||||||||||||||||||||
Imigran/Imitrex |
14 | 18 | 27 | 12 | – | 20 | 11 | (9 | ) | – | 10 | – | – | |||||||||||||||||||||||||||||||||||
Lamictal |
14 | 33 | 56 | 11 | 63 | 38 | 12 | 13 | 50 | 10 | 67 | 67 | ||||||||||||||||||||||||||||||||||||
Requip |
16 | 27 | 45 | 12 | (23 | ) | (8 | ) | 12 | 11 | 33 | 9 | – | (10 | ) | |||||||||||||||||||||||||||||||||
Seroxat/Paxil |
90 | (11 | ) | 2 | 72 | (14 | ) | (3 | ) | 79 | (5 | ) | 3 | 59 | (7 | ) | (12 | ) | ||||||||||||||||||||||||||||||
Treximet |
– | – | – | – | – | – | 1 | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Wellbutrin |
– | – | – | 1 | – | – | 4 | 100 | >100 | – | (50 | ) | – | |||||||||||||||||||||||||||||||||||
Cardiovascular and urogenital |
79 | 23 | 41 | 63 | 28 | 37 | 61 | 19 | 27 | 52 | 21 | 24 | ||||||||||||||||||||||||||||||||||||
Arixtra |
4 | – | 33 | 4 | 100 | 100 | 4 | – | 33 | 3 | – | – | ||||||||||||||||||||||||||||||||||||
Avodart |
32 | 100 | >100 | 20 | 89 | >100 | 16 | 78 | 78 | 16 | 88 | 100 | ||||||||||||||||||||||||||||||||||||
Coreg |
– | – | – | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Fraxiparine |
2 | (67 | ) | (33 | ) | 5 | >100 | 67 | 4 | (60 | ) | (20 | ) | 2 | – | (33 | ) | |||||||||||||||||||||||||||||||
Lovaza |
1 | – | – | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Vesicare |
– | – | – | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Volibris |
3 | – | – | – | – | (100 | ) | 1 | – | – | 1 | – | – | |||||||||||||||||||||||||||||||||||
Metabolic |
40 | (32 | ) | (25 | ) | 44 | (28 | ) | (19 | ) | 50 | (8 | ) | (2 | ) | 49 | 2 | 2 | ||||||||||||||||||||||||||||||
Avandia products |
10 | (64 | ) | (60 | ) | 15 | (50 | ) | (42 | ) | 25 | (13 | ) | 4 | 23 | 4 | (4 | ) | ||||||||||||||||||||||||||||||
Bonviva/Boniva |
4 | – | 100 | 3 | 67 | – | 2 | – | 100 | 3 | – | – | ||||||||||||||||||||||||||||||||||||
Anti-bacterials |
42 | (2 | ) | 2 | 47 | – | 12 | 43 | (2 | ) | 2 | 44 | 5 | 10 | ||||||||||||||||||||||||||||||||||
Augmentin |
21 | (10 | ) | 5 | 23 | 24 | 35 | 20 | 33 | 33 | 19 | – | – | |||||||||||||||||||||||||||||||||||
Oncology and emesis |
25 | 41 | 47 | 17 | (17 | ) | (6 | ) | 18 | 21 | 29 | 15 | 27 | 36 | ||||||||||||||||||||||||||||||||||
Arzerra |
– | – | – | – | – | – | 1 | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Hycamtin |
2 | (33 | ) | (33 | ) | 1 | 100 | – | 2 | (50 | ) | – | 1 | – | – | |||||||||||||||||||||||||||||||||
Promacta |
1 | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Tyverb/Tykerb |
9 | 14 | 29 | 8 | 13 | – | 9 | – | – | 7 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Votrient |
– | – | – | 1 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Vaccines |
170 | (28 | ) | (17 | ) | 178 | 81 | >100 | 252 | >100 | >100 | 355 | >100 | >100 | ||||||||||||||||||||||||||||||||||
Boostrix |
6 | (17 | ) | – | 4 | – | – | 5 | 67 | 67 | 4 | (67 | ) | (33 | ) | |||||||||||||||||||||||||||||||||
Cervarix |
34 | >100 | >100 | 25 | >100 | >100 | 17 | >100 | >100 | 12 | >100 | >100 | ||||||||||||||||||||||||||||||||||||
Fluarix, FluLaval |
8 | (33 | ) | (33 | ) | 17 | 50 | 70 | (1 | ) | – | – | 4 | >100 | >100 | |||||||||||||||||||||||||||||||||
Flu pandemic |
46 | (67 | ) | (57 | ) | 49 | >100 | >100 | 140 | – | – | 242 | >100 | >100 | ||||||||||||||||||||||||||||||||||
Hepatitis |
22 | 11 | 22 | 17 | – | 6 | 20 | 29 | 43 | 24 | 22 | 33 | ||||||||||||||||||||||||||||||||||||
Infanrix, Pediarix |
21 | 13 | 31 | 18 | (6 | ) | 6 | 16 | (6 | ) | – | 20 | 13 | 33 | ||||||||||||||||||||||||||||||||||
Rotarix |
4 | (33 | ) | (33 | ) | 7 | 40 | 40 | 2 | (75 | ) | (75 | ) | 8 | 40 | 60 | ||||||||||||||||||||||||||||||||
Synflorix |
5 | (33 | ) | (17 | ) | 7 | >100 | >100 | 10 | >100 | >100 | 7 | – | – | ||||||||||||||||||||||||||||||||||
Dermatologicals |
50 | (12 | ) | (2 | ) | 47 | 2 | 12 | 56 | 76 | 93 | 44 | 95 | 100 | ||||||||||||||||||||||||||||||||||
Bactroban |
3 | – | 50 | 4 | (25 | ) | – | 2 | (33 | ) | (33 | ) | 4 | 100 | 100 | |||||||||||||||||||||||||||||||||
Dermovate |
6 | – | – | 6 | – | – | 7 | – | – | 6 | – | – | ||||||||||||||||||||||||||||||||||||
Duac |
3 | >100 | >100 | 5 | 100 | >100 | 6 | – | – | 1 | – | – | ||||||||||||||||||||||||||||||||||||
Soriatane |
– | – | – | – | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Zovirax |
12 | (15 | ) | (8 | ) | 11 | (9 | ) | – | 13 | (14 | ) | (7 | ) | 10 | (17 | ) | (17 | ) | |||||||||||||||||||||||||||||
Other |
88 | – | 11 | 59 | 14 | 16 | 69 | (17 | ) | (4 | ) | 59 | 11 | 4 | ||||||||||||||||||||||||||||||||||
| 1,057 | (16 | ) | (6 | ) | 901 | (1 | ) | 9 | 1,021 | 16 | 31 | 1,085 | 35 | 35 | ||||||||||||||||||||||||||||||||||
VIIV Healthcare (HIV) |
45 | 5 | 10 | 44 | 6 | 22 | 49 | 24 | 32 | 37 | (5 | ) | – | |||||||||||||||||||||||||||||||||||
Combivir |
11 | – | 10 | 10 | (20 | ) | – | 10 | – | – | 9 | – | 13 | |||||||||||||||||||||||||||||||||||
Epivir |
5 | 100 | 67 | 5 | (33 | ) | (17 | ) | 5 | – | – | 5 | (20 | ) | – | |||||||||||||||||||||||||||||||||
Epzicom/Kivexa |
19 | 19 | 19 | 19 | 23 | 46 | 16 | – | 7 | 17 | 7 | 21 | ||||||||||||||||||||||||||||||||||||
Lexiva |
4 | – | 100 | – | (33 | ) | (100 | ) | 4 | – | 100 | 3 | – | – | ||||||||||||||||||||||||||||||||||
Selzentry |
1 | – | – | 2 | – | – | – | – | – | – | – | – | ||||||||||||||||||||||||||||||||||||
Trizivir |
2 | – | – | 1 | 100 | – | 3 | (100 | ) | (25 | ) | 1 | >100 | – | ||||||||||||||||||||||||||||||||||
Table of Contents
193
| Q4 2010 | Q3 2010 | Q2 2010 | Q1 2010 | |||||||||||||||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||
Over-the-counter medicines |
645 | 1 | 4 | 601 | 1 | 5 | 593 | (2 | ) | 3 | 617 | 11 | 8 | |||||||||||||||||||||||||||||||||||
Oral healthcare |
411 | 7 | 10 | 400 | 4 | 7 | 410 | 9 | 12 | 381 | 5 | 4 | ||||||||||||||||||||||||||||||||||||
Nutritional healthcare |
211 | 7 | 10 | 259 | 12 | 16 | 249 | 6 | 10 | 233 | 12 | 10 | ||||||||||||||||||||||||||||||||||||
| 1,267 | 4 | 7 | 1,260 | 4 | 8 | 1,252 | 3 | 7 | 1,231 | 9 | 7 | |||||||||||||||||||||||||||||||||||||
| Q4 2010 | Q3 2010 | Q2 2010 | Q1 2010 | |||||||||||||||||||||||||||||||||||||||||||||
| £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | £m | CER% | £% | |||||||||||||||||||||||||||||||||||||
USA |
279 | 5 | 7 | 249 | (4 | ) | – | 263 | – | 4 | 246 | 3 | (5 | ) | ||||||||||||||||||||||||||||||||||
Europe |
503 | (3 | ) | (5 | ) | 491 | (2 | ) | (3 | ) | 493 | (2 | ) | (3 | ) | 471 | 9 | 8 | ||||||||||||||||||||||||||||||
Rest of World |
485 | 13 | 22 | 520 | 15 | (25 | ) | 496 | 11 | 22 | 514 | 13 | 13 | |||||||||||||||||||||||||||||||||||
| 1,267 | 4 | 7 | 1,260 | 4 | 8 | 1,252 | 3 | 7 | 1,231 | 9 | 7 | |||||||||||||||||||||||||||||||||||||
Table of Contents
194
| Turnover by business segment | 2010 | 2009 | 2008 | 2007 | 2006 | |||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Pharmaceutical |
23,382 | 23,694 | 20,381 | 19,163 | 20,013 | |||||||||||||||
Consumer Healthcare |
5,010 | 4,674 | 3,971 | 3,553 | 3,212 | |||||||||||||||
| 28,392 | 28,368 | 24,352 | 22,716 | 23,225 | ||||||||||||||||
| Pharmaceutical turnover by therapeutic area | 2010 | 2009 | 2008 | 2007 | 2006 | |||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Respiratory |
7,238 | 6,977 | 5,817 | 5,032 | 4,991 | |||||||||||||||
Anti-virals |
1,086 | 2,416 | 1,584 | 1,478 | 1,191 | |||||||||||||||
HIV |
1,566 | 1,605 | 1,513 | 1,442 | 1,515 | |||||||||||||||
Central nervous system |
1,753 | 1,870 | 2,897 | 3,348 | 3,642 | |||||||||||||||
Cardiovascular and urogenital |
2,570 | 2,298 | 1,847 | 1,554 | 1,636 | |||||||||||||||
Metabolic |
678 | 1,181 | 1,191 | 1,508 | 1,870 | |||||||||||||||
Anti-bacterials |
1,396 | 1,457 | 1,301 | 1,213 | 1,271 | |||||||||||||||
Oncology and emesis |
688 | 629 | 496 | 477 | 1,069 | |||||||||||||||
Vaccines |
4,326 | 3,706 | 2,539 | 1,993 | 1,692 | |||||||||||||||
Dermatologicals |
1,087 | 707 | 414 | 375 | 367 | |||||||||||||||
Other |
994 | 848 | 782 | 743 | 769 | |||||||||||||||
| 23,382 | 23,694 | 20,381 | 19,163 | 20,013 | ||||||||||||||||
| Pharmaceutical turnover by geographic area | 2010 | 2009 | 2008 | 2007 | 2006 | |||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
USA |
8,308 | 9,294 | 8,894 | 9,273 | 10,353 | |||||||||||||||
Europe |
7,133 | 7,720 | 6,483 | 5,560 | 5,437 | |||||||||||||||
Emerging Markets |
3,702 | 3,000 | 2,282 | 1,883 | 1,769 | |||||||||||||||
Asia Pacific/Japan |
3,204 | 2,715 | 1,918 | 1,701 | 1,666 | |||||||||||||||
Other |
1,035 | 965 | 804 | 746 | 788 | |||||||||||||||
| 23,382 | 23,694 | 20,381 | 19,163 | 20,013 | ||||||||||||||||
| Consumer Healthcare turnover | 2010 | 2009 | 2008 | 2007 | 2006 | |||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
OTC medicines |
2,456 | 2,339 | 1,935 | 1,788 | 1,561 | |||||||||||||||
Oral healthcare |
1,602 | 1,484 | 1,240 | 1,049 | 993 | |||||||||||||||
Nutritional healthcare |
952 | 851 | 796 | 716 | 658 | |||||||||||||||
| 5,010 | 4,674 | 3,971 | 3,553 | 3,212 | ||||||||||||||||
Table of Contents
195
| Financial results – total | 2010 | 2009 | 2008 | 2007 | 2006 | |||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Turnover |
28,392 | 28,368 | 24,352 | 22,716 | 23,225 | |||||||||||||||
Operating profit |
3,783 | 8,425 | 7,141 | 7,593 | 7,808 | |||||||||||||||
Profit before taxation |
3,157 | 7,891 | 6,659 | 7,452 | 7,799 | |||||||||||||||
Profit after taxation |
1,853 | 5,669 | 4,712 | 5,310 | 5,498 | |||||||||||||||
| pence | pence | pence | pence | pence | ||||||||||||||||
Basic earnings per share |
32.1 | 109.1 | 88.6 | 94.4 | 95.5 | |||||||||||||||
Diluted earnings per share |
31.9 | 108.2 | 88.1 | 93.7 | 94.5 | |||||||||||||||
| Financial results – before major restructuring | 2010 | 2009 | 2008 | |||||||||||||||||
| £m | £m | £m | ||||||||||||||||||
Turnover |
28,392 | 28,368 | 24,352 | |||||||||||||||||
Operating profit |
5,128 | 9,257 | 8,259 | |||||||||||||||||
Profit before taxation |
4,505 | 8,726 | 7,782 | |||||||||||||||||
Profit after taxation |
2,961 | 6,283 | 5,551 | |||||||||||||||||
| pence | pence | pence | ||||||||||||||||||
Adjusted earnings per share |
53.9 | 121.2 | 104.7 | |||||||||||||||||
Adjusted diluted earnings per share |
53.5 | 120.3 | 104.1 | |||||||||||||||||
| 2010 | 2009 | 2008 | 2007 | 2006 | ||||||||||||||||
| millions | millions | millions | millions | millions | ||||||||||||||||
Weighted average number of shares in issue: |
||||||||||||||||||||
Basic |
5,085 | 5,069 | 5,195 | 5,524 | 5,643 | |||||||||||||||
Diluted |
5,128 | 5,108 | 5,226 | 5,567 | 5,700 | |||||||||||||||
| % | % | % | % | % | ||||||||||||||||
Return on capital employed |
30.8 | 82.8 | 73.1 | 76.2 | 90.6 | |||||||||||||||
| Balance sheet | 2010 | 2009 | 2008 | 2007 | 2006 | |||||||||||||||
| £m | £m | £m | £m | £m | ||||||||||||||||
Non-current assets |
26,194 | 25,292 | 22,124 | 17,377 | 14,561 | |||||||||||||||
Current assets |
16,036 | 17,570 | 17,269 | 13,626 | 10,992 | |||||||||||||||
Total assets |
42,230 | 42,862 | 39,393 | 31,003 | 25,553 | |||||||||||||||
Current liabilities |
(12,794 | ) | (12,118 | ) | (10,017 | ) | (10,345 | ) | (7,265 | ) | ||||||||||
Non-current liabilities |
(19,691 | ) | (20,002 | ) | (21,058 | ) | (10,748 | ) | (8,640 | ) | ||||||||||
Total liabilities |
(32,485 | ) | (32,120 | ) | (31,075 | ) | (21,093 | ) | (15,905 | ) | ||||||||||
Net assets |
9,745 | 10,742 | 8,318 | 9,910 | 9,648 | |||||||||||||||
Shareholders’ equity |
8,887 | 10,005 | 7,931 | 9,603 | 9,386 | |||||||||||||||
Non-controlling interests |
858 | 737 | 387 | 307 | 262 | |||||||||||||||
Total equity |
9,745 | 10,742 | 8,318 | 9,910 | 9,648 | |||||||||||||||
Table of Contents
196
| 2010 | 2009 | 2008 | 2007 | 2006 | ||||||||||||||||
USA |
17,555 | 22,594 | 21,176 | 24,838 | 24,726 | |||||||||||||||
Europe |
39,910 | 42,048 | 44,677 | 46,869 | 45,758 | |||||||||||||||
Rest of World: |
||||||||||||||||||||
Asia Pacific, including China |
23,388 | 21,011 | 18,983 | 17,525 | 17,570 | |||||||||||||||
Japan |
3,461 | 3,264 | 3,174 | 3,284 | 3,195 | |||||||||||||||
Middle East, Africa |
3,609 | 3,619 | 3,403 | 3,156 | 3,204 | |||||||||||||||
Latin America |
6,432 | 5,169 | 5,228 | 5,249 | 5,856 | |||||||||||||||
Canada |
2,106 | 2,208 | 2,362 | 2,562 | 2,386 | |||||||||||||||
Rest of World |
38,996 | 35,271 | 33,150 | 31,776 | 32,211 | |||||||||||||||
| 96,461 | 99,913 | 99,003 | 103,483 | 102,695 | ||||||||||||||||
Manufacturing |
30,611 | 31,162 | 32,622 | 33,995 | 33,235 | |||||||||||||||
Selling |
43,918 | 44,621 | 42,430 | 44,499 | 44,484 | |||||||||||||||
Administration |
8,850 | 9,405 | 8,787 | 8,960 | 9,024 | |||||||||||||||
Research and development |
13,082 | 14,725 | 15,164 | 16,029 | 15,952 | |||||||||||||||
| 96,461 | 99,913 | 99,003 | 103,483 | 102,695 | ||||||||||||||||
| 2010 | 2009 | 2008 | 2007 | 2006 | ||||||||||||||||
Average |
1.55 | 1.56 | 1.85 | 2.00 | 1.85 | |||||||||||||||
| Feb | Jan | Dec | Nov | Oct | Sept | |||||||||||||||||||
| 2011 | 2011 | 2010 | 2010 | 2010 | 2010 | |||||||||||||||||||
High |
1.62 | 1.60 | 1.59 | 1.63 | 1.60 | 1.58 | ||||||||||||||||||
Low |
1.60 | 1.55 | 1.54 | 1.55 | 1.57 | 1.53 | ||||||||||||||||||
| * | On 31st December 2008, the Federal Reserve Bank of New York ceased publishing noon buying rates. The Bank of England 4pm buying rates have been used for subsequent calculations. |
Table of Contents
197
| Key | ||||||
†
|
In-license or other alliance relationship with third party | BLA | Biological License Application | |||
S
|
Month of first submission | MAA | Marketing Authorisation Application (Europe) | |||
A
|
Month of first regulatory approval (for MAA, this is the first EU | NDA | New Drug Application (USA) | |||
| approval letter) | Phase I | Evaluation of clinical pharmacology, usually conducted in volunteers | ||||
AL/CR
|
Month Approvable or Complete Response Letter received – indicates | Phase II | Determination of dose and initial evaluation of efficacy, conducted in a | |||
| that ultimately approval may be given subject to resolution of | small number of patients | |||||
| outstanding queries | Phase III | Large comparative study (compound versus placebo and/or established | ||||
PO
|
Month of EU Positive Opinion | treatment) in patients to establish clinical benefit and safety | ||||
TA
|
FDA Tentative Approval | |||||
| Achieved Regulatory | ||||||||||
| review milestones | ||||||||||
| Compound | Type | Indication | Phase | MAA | NDA/BLA | |||||
| Biopharmaceuticals | ||||||||||
933776
|
beta amyloid monoclonal antibody | Alzheimer’s disorders | I | |||||||
1070806
|
IL18 monoclonal antibody | metabolic disease | I | |||||||
1223249
|
NOGO-A monoclonal antibody | amyotrophic lateral sclerosis & multiple sclerosis | I | |||||||
2401502†
|
domain antibody targetted multi-component vaccine
|
malignant melanoma | I | |||||||
2586881 (APN01)†
|
recombinant human angiotensin converting anzyme 2
|
acute respiratory distress syndrome | I | |||||||
iboctadekin† (+ Doxil)
|
IL18 immunomodulator (+ topoisomerase II inhibitor)
|
ovarian cancer | I | |||||||
otelixizumab†
|
anti-CD3 monoclonal antibody (i.v.) | Graves eye disease | I | |||||||
otelixizumab†
|
anti-CD3 monoclonal antibody (s.c. & i.v.) | rheumatoid arthritis | I | |||||||
otelixizumab†
|
anti-CD3 monoclonal antibody (s.c.) | type 1 diabetes | I | |||||||
249320
|
myelin-associated glycoprotein monoclonal antibody
|
stroke | II | |||||||
315234
|
oncostatin M monoclonal antibody | rheumatoid arthritis | II | |||||||
768974†
|
parathyroid hormone | osteoporosis | II | |||||||
albiglutide†
|
glucagon-like peptide 1 (GLP 1) agonist | heart failure | II | |||||||
Benlysta†
|
anti-B lymphocyte stimulator monoclonal antibody (s.c.)
|
systemic lupus erythematosus | II | |||||||
mepolizumab
|
anti-IL5 monoclonal antibody | severe asthma & nasal polyposis | II | |||||||
ofatumumab†
|
anti-CD20 human monoclonal antibody (s.c.) | multiple sclerosis | II | |||||||
ofatumumab†
|
anti-CD20 human monoclonal antibody (s.c.) | rheumatoid arthritis | II | |||||||
otelixizumab†
|
anti-CD3 monoclonal antibody (i.v.) | myaesthenia gravis | II | |||||||
albiglutide†
|
GLP 1 agonist | type 2 diabetes | III | |||||||
Arzerra†
|
anti-CD20 human monoclonal antibody | chronic lymphocytic leukaemia, first line therapy & use in relapsed patients
|
III | |||||||
Arzerra†
|
anti-CD20 human monoclonal antibody | diffuse large B cell lymphoma (relapsed patients) | III | |||||||
Arzerra†
|
anti-CD20 human monoclonal antibody | follicular lymphoma (refractory & relapsed patients) | III | |||||||
otelixizumab†
|
anti-CD3 monoclonal antibody (i.v.) | type 1 diabetes | III | |||||||
Benlysta†
|
anti-B lymphocyte stimulator monoclonal
antibody (i.v.)
|
systemic lupus erythematosus | Submitted | S: Jun10 | S: Jun10 | |||||
denosumab†
|
anti-receptor activator for nuclear kappa (RANK) ligand human monoclonal antibody
|
bone metastatic disease | Submitted | N/A | N/A | |||||
Arzerra†
|
anti-CD20 human monoclonal antibody | chronic lymphocytic leukaemia (refractory patients) | Approved | A: Apr10 | A: Oct09 | |||||
Prolia†
|
anti-RANK ligand human monoclonal antibody | hormone ablative/chemotherapy bone loss in prostate cancer patients
|
Approved | N/A | N/A | |||||
Prolia†
|
anti-RANK ligand human monoclonal antibody | postmenopausal osteoporosis | Approved | A: May10 | N/A | |||||
| Cardiovascular & Metabolic | ||||||||||
1614235†
|
sodium dependent glucose transport (SGLT1) inhibitor
|
type 2 diabetes | I | |||||||
2245840
|
SIRT1 activator | sarcopaenia (also COPD & psoriasis) | I | |||||||
256073
|
high affinity nicotinic acid receptor (HM74A) agonist
|
metabolic disorders | II | |||||||
557296
|
oxytocin antagonist | premature ejaculation | II | |||||||
1278863
|
prolyl hydroxylase inhibitor | anaemia associated with chronic renal disease | II | |||||||
1278863
|
prolyl hydroxylase inhibitor | peripheral arterial disease | II | |||||||
1292263
|
G-protein coupled receptor 119 (GRP119) agonist
|
metabolic disorders | II | |||||||
1521498
|
mu-opioid receptor inverse agonist | compulsive eating disorders | II | |||||||
2245840
|
SIRT1 activator | type 2 diabetes (also COPD & psoriasis) | II | |||||||
losmapimod
|
p38 kinase inhibitor | cardiovascular disease (also COPD & pain) | II | |||||||
retosiban
|
oxytocin antagonist | threatened pre-term labour | II | |||||||
rilapladib†
|
Lp-PLA2 inhibitor | atherosclerosis | II | |||||||
darapladib†
|
Lp-PLA2 inhibitor | atherosclerosis | III | |||||||
Arixtra
|
synthetic factor Xa inhibitor | treatment of acute coronary syndrome | Approved | A: Aug07 | AL: Feb07 & Sep07 |
|||||
Table of Contents
198
| Achieved Regulatory | ||||||||||
| review milestones | ||||||||||
| Compound | Type | Indication | Phase | MAA | NDA/BLA | |||||
Infectious Diseases |
||||||||||
2251052†
|
leucyl t-RNA synthetase inhibitor (oral & i.v.) | bacterial infections | I | |||||||
2336805
|
hepatitis C virus inhibitor | hepatitis C | I | |||||||
2485852
|
hepatitis C virus inhibitor | hepatitis C | I | |||||||
1322322
|
polypeptide deformylase inhibitor | bacterial infections | II | |||||||
tafenoquine†
|
8-aminoquinoline | Plasmodium vivax malaria | II | |||||||
Relenza†
|
neuraminidase inhibitor (i.v.) | influenza | III | |||||||
Neurosciences |
||||||||||
2018682
|
sphingosine-1-phosphate receptor 1 (S1P1) agonist |
multiple sclerosis | I | |||||||
239512
|
histamine H3 antagonist | dementia & schizophrenia | II | |||||||
649868†
|
orexin antagonist | sleep disorders | II | |||||||
742457
|
5HT6 antagonist | dementia | II | |||||||
firategrast†
|
dual alpha4 integrin antagonist (VLA4) | multiple sclerosis | II | |||||||
Horizant†
|
voltage-gated calcium channel modulator | post-herpetic neuralgia | II | N/A | ||||||
losmapimod
|
p38 kinase inhibitor | pain (also cardiovascular disease & COPD) | II | |||||||
orvepitant
|
NK1 antagonist | depression & anxiety | II | |||||||
IPX066†
|
dopamine precursor + DOPA decarboxylase
inhibitor |
Parkinson’s disease | III | N/A | ||||||
Horizant†
|
voltage-gated calcium channel modulator | restless legs syndrome | Submitted | S: Sep08, Jan09 & Oct10 |
||||||
Trobalt/Potiga (retigabine/ezogabine)† |
neuronal potassium channel opener | epilepsy - partial seizures | Submitted | PO: Jan11 | CR: Nov10 | |||||
Oncology |
||||||||||
2110183
|
AKT protein kinase inhibitor | cancer | I | |||||||
2126458
|
Pi3 kinase inhibitor | cancer | I | |||||||
2141795
|
AKT protein kinase inhibitor | cancer | I | |||||||
2256098
|
focal adhesion kinase inhibitor | cancer | I | |||||||
vestipitant
|
NK1 antagonist (i.v.) | post operative nausea & vomiting | I | |||||||
1120212† + BKM120
|
mitogen-activated protein kinase inhibitor
(MEK1/2) + Pi3 alpha kinase inhibitor |
cancer | I | |||||||
1120212†
|
MEK1/2 inhibitor | pancreatic cancer | II | |||||||
1120212† +2118436
|
MEK1/2 inhibitor + BRaf protein kinase inhibitor | metastatic melanoma | II | |||||||
2285921†
|
thrombopoietin receptor agonist | thrombocytopaenia | II | |||||||
foretinib†
|
mesenchymal-epithelial transition factor
(C-met) kinase inhibitor |
papillary renal cell carcinoma and other cancers | II | |||||||
Revolade/Promacta†
|
thrombopoietin receptor agonist | oncology-related thrombocytopaenia | II | |||||||
1120212†
|
MEK1/2 inhibitor | metastatic melanoma | III | |||||||
2118436
|
BRaf protein kinase inhibitor | metastatic melanoma | III | |||||||
Votrient
|
multi-kinase angiogenesis inhibitor | ovarian cancer, maintenance therapy | III | |||||||
Revolade/Promacta†
|
thrombopoietin receptor agonist | chronic liver disease induced thrombocytopaenia | III | |||||||
Revolade/Promacta†
|
thrombopoietin receptor agonist | hepatitis C induced thrombocytopaenia | III | |||||||
Tyverb/Tykerb
|
Her2 and EGFR dual kinase inhibitor | breast cancer, adjuvant therapy | III | |||||||
Tyverb/Tykerb
|
Her2 and EGFR dual kinase inhibitor | gastric cancer | III | |||||||
Tyverb/Tykerb
|
Her2 and EGFR dual kinase inhibitor | head & neck squamous cell carcinoma (resectable disease)
|
III | |||||||
Votrient
|
multi-kinase angiogenesis inhibitor | renal cell cancer, adjuvant therapy | III | |||||||
Votrient
|
multi-kinase angiogenesis inhibitor | sarcoma | III | |||||||
Votrient + Tyverb/Tykerb
|
multi-kinase angiogenesis inhibitor +
Her2 and EGFR dual kinase inhibitor |
inflammatory breast cancer | III | |||||||
Avodart
|
5-alpha reductase inhibitor | reduction in the risk of prostate cancer | Submitted | S: Sep09 & Mar10 | CR: Jan11 | |||||
Duodart/Jalyn
|
5-alpha reductase inhibitor + alpha blocker | benign prostatic hyperplasia - fixed dose combination | Approved | A: Mar10 | A: Jun10 | |||||
Revolade/Promacta†
|
thrombopoietin receptor agonist | idiopathic thrombocytopaenic purpura | Approved | A: Mar10 | A: Nov08 | |||||
Tyverb/Tykerb
|
Her2 and EGFR dual kinase inhibitor | breast cancer, first line therapy | Approved | A: Jun10 | A: Jan10 | |||||
Votrient
|
multi-kinase angiogenesis inhibitor | renal cell cancer | Approved | A: Jun10 | A:Oct09 | |||||
Ophthalmology |
||||||||||
pazopanib
|
multi-kinase angiogenesis inhibitor (oral) | age-related macular degeneration (also cancer indications) | I | |||||||
pazopanib
|
multi-kinase angiogenesis inhibitor (eye drops) | age-related macular degeneration | II | |||||||
Table of Contents
199
| Achieved Regulatory | ||||||||||
| review milestones | ||||||||||
| Compound | Type | Indication | Phase | MAA | NDA/BLA | |||||
| Respiratory & Immuno-inflammation | ||||||||||
610677
|
p38 kinase inhibitor (inhaled) | COPD | I | |||||||
705498
|
transient receptor potential vanilloid
(TRPV1) antagonist (topical) |
pruritis | I | |||||||
1322888
|
motilin receptor agonist | delayed gastric emptying | I | |||||||
1325756
|
chemokine receptor (CXCR2) antagonist | COPD | I | |||||||
1440115
|
urotensin antagonist | asthma | I | |||||||
2245840
|
SIRT1 activator | COPD (also type 2 diabetes & sarcopaenia) | I | |||||||
2245840
|
SIRT1 activator | psoriasis (also type 2 diabetes & sarcopaenia) | II | |||||||
256066
|
PDE4 inhibitor (inhaled) | COPD | II | |||||||
656933
|
chemokine receptor (CXCR2) antagonist | cystic fibrosis | II | |||||||
685698
|
glucocorticoid agonist | asthma | II | |||||||
681323
|
p38 kinase inhibitor (i.v.) | acute lung injury & acute respiratory distress syndrome | II | |||||||
705498
|
transient receptor potential vanilloid
(TRPV1) antagonist (intranasal) |
non-allergic rhinitis | II | |||||||
870086
|
novel glucocorticoid agonist (inhaled) | asthma | II | |||||||
870086
|
novel glucocorticoid agonist (topical) | atopic dermatitis | II | |||||||
961081†
|
muscarinic antagonist, beta2 agonist | COPD | II | |||||||
962040
|
motilin receptor agonist | delayed gastric emptying | II | |||||||
1399686
|
anti-inflammatory macrolide conjugate (oral) | inflammatory bowel disease | II | |||||||
2190915†
|
5-lipoxygenase-activating protein (FLAP)
inhibitor |
asthma | II | |||||||
losmapimod
|
p38 kinase inhibitor (oral) | COPD (also cardiovascular disease & pain) | II | |||||||
573719
|
muscarinic acetylcholine antagonist | COPD | III | |||||||
573719 + vilanterol
|
muscarinic acetylcholine antagonist + | COPD | III | |||||||
(642444)†
|
long-acting beta2 agonist | |||||||||
vilanterol (642444)†
|
long-acting beta2 agonist | COPD | III | |||||||
1605786 (CCX282)†
|
CCR9 antagonist | Crohn’s disease | III | |||||||
Relovair |
||||||||||
(vilanterol† + 685698)
|
long-acting beta2 agonist +
glucocorticoid agonist |
asthma | III | |||||||
Relovair |
||||||||||
(vilanterol† + 685698)
|
long-acting beta2 agonist +
glucocorticoid agonist |
COPD | III | |||||||
| Paediatric Vaccines | ||||||||||
Heptavalent combination vaccine |
conjugated | Neisseria meningitis C, Haemophilus influenzae type b,
diphtheria, Hepatitis B, tetanus, pertussis and poliomyelitis
disease prophylaxis
|
II | |||||||
MMR
|
live attenuated | measles, mumps, rubella prophylaxis | II (USA) | A: Oct03 | ||||||
S. pneumoniae paediatric
next generation
|
recombinant – conjugated | Streptococcus pneumoniae disease prophylaxis | II | |||||||
Mosquirix
|
recombinant | malaria prophylaxis (Plasmodium falciparum) | III | N/A | ||||||
Nimenrix (MenACWY-TT)
|
conjugated | Neisseria meningitis groups A, C, W & Y disease prophylaxis
|
Submitted | |||||||
MenHibrix (Hib-MenCY-TT) |
conjugated | Neisseria meningitis groups C & Y & Haemophilus influenzae
type b disease prophylaxis |
Submitted | N/A | CR: Jun10 | |||||
| Other Vaccines | ||||||||||
Flu pandemic†
|
cell-culture based H5N1 vaccine | pandemic influenza prophylaxis | I | |||||||
HIV
|
recombinant | HIV disease prophylaxis | I | |||||||
HIV
|
recombinant | HIV disease immunotherapy | II | |||||||
Tuberculosis
|
recombinant | tuberculosis prophylaxis | II | |||||||
Flu vaccine
|
inactivated split – quadrivalent | seasonal influenza prophylaxis | III | |||||||
Zoster
|
recombinant | Herpes Zoster prevention | III | |||||||
Flu (pre-) pandemic
|
H5N1 inactivated split – monovalent (Quebec) | pre-pandemic & pandemic influenza prophylaxis | Submitted | N/A | S:Jun09 (Canada) |
|||||
Pumarix
|
H5N1 inactivated split – monovalent (Quebec) | pandemic influenza prophylaxis | Submitted | PO: Nov10 | N/A | |||||
Table of Contents
200
| Achieved Regulatory | ||||||||||
| review milestones | ||||||||||
| Compound | Type | Indication | Phase | MAA | NDA/BLA | |||||
| Antigen Specific Cancer Immunotherapeutic (ASCI) | ||||||||||
PRAME
|
recombinant | treatment of metastatic melanoma | I | |||||||
PRAME
|
recombinant | treatment of resectable non-small cell lung cancer | I | |||||||
NY-ESO-1
|
recombinant | treatment of metastatic melanoma | I | |||||||
WT1
|
recombinant | treatment of acute myelogenous leukaemia | II | |||||||
MAGE-A3
|
recombinant | treatment of melanoma | III | |||||||
MAGE-A3
|
recombinant | treatment of non-small cell lung cancer | III | |||||||
| Rare Diseases | ||||||||||
2402968†
|
antisense oligonucleotide | Duchenne muscular dystrophy | III | |||||||
2696273†
|
ex-vivo stem cell gene therapy | adenosine deaminase severe combined immune deficiency
(ADA-SCID)
|
III | |||||||
migalastat HCl†
|
pharmacological chaperone | Fabry disease | III | |||||||
| Stiefel (late stage assets only) | ||||||||||
tazarotene foam
|
retinoid foam | acne vulgaris | III | |||||||
Duac low dose
|
clindamycin/benzoyl peroxide gel | acne vulgaris | Submitted | S: Nov10 | ||||||
Sorilux
(calcipotriene foam)
|
vitamin D3 analog | mild to moderate plaque psoriasis | Approved | A: Oct10 | ||||||
itraconazole tablets
|
oral anti-fungal | onychomycosis | Approved | A: Apr10 | ||||||
Veltin
|
antibiotic/retinoid gel | acne vulgaris | Approved | A: Jul10 | ||||||
| HIV (ViiV Healthcare) | ||||||||||
1265744†
|
HIV integrase inhibitor (long-acting formulation) | HIV infections | I | |||||||
2248761†
|
non-nucleoside reverse transcriptase inhibitor | HIV infections | II | |||||||
PF-232798
|
CCR5 antagonist | HIV infections | II | |||||||
UK-453061
|
non-nucleoside reverse transcriptase inhibitor | HIV infections | II | |||||||
1349572†
|
HIV integrase inhibitor | HIV infections | III | |||||||
1349572† + abacavir sulphate + lamivudine |
HIV integrase inhibitor + reverse transcriptase inhibitors (fixed dose combination) |
HIV infections | III | |||||||
Company
|
Disease Area | Phase | ||
Cancer Research UK
|
cancer | I | ||
ChemoCentryx
|
inflammatory disease | I* & II | ||
Galapagos
|
autoimmune disease | I* | ||
OncoMed Pharmaceuticals
|
oncology | I* | ||
Prosensa Therapeutics
|
neuroscience | I | ||
Ranbaxy Laboratories
|
respiratory | I | ||
Theravance
|
pain | I | ||
Telethon Institute for Gene Therapy
|
stem cell gene therapy | I & II | ||
Affiris
|
Alzheimer’s disease treatment vaccine | II | ||
Nabi
|
nicotine vaccine | III | ||
| * Two assets | ||
Table of Contents
201
| Share price | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| £ | £ | £ | ||||||||||
At 1st January |
13.20 | 12.85 | 12.79 | |||||||||
High during the year |
13.40 | 13.34 | 13.85 | |||||||||
Low during the year |
10.95 | 9.87 | 9.95 | |||||||||
At 31st December |
12.40 | 13.20 | 12.85 | |||||||||
Increase/(decrease) |
(6.1% | ) | 2.7% | 0.5% | ||||||||
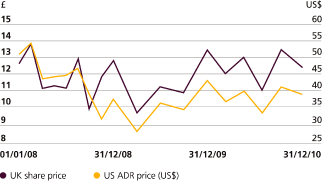
| Year | pence | |||
2010 |
65 | |||
2009 |
61 | |||
2008 |
57 | |||
2007 |
53 | |||
2006 |
48 | |||
| Year | US$ | |||
2010 |
2.04 | |||
2009 |
1.93 | |||
2008 |
2.01 | |||
2007 |
2.14 | |||
2006 |
1.80 | |||
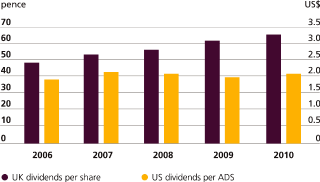
| Quarter | Ex-dividend date | Record date | Payment date | |||||||||
Q4 2010 |
9th February 2011 | 11th February 2011 | 7th April 2011 | |||||||||
Q1 2011 |
4th May 2011 | 6th May 2011 | 7th July 2011 | |||||||||
Q2 2011 |
3rd August 2011 | 5th August 2011 | 6th October 2011 | |||||||||
Q3 2011 |
2nd November 2011 | 4th November 2011 | 5th January 2012 | |||||||||
| Publication | Date | |||
Results announcements |
||||
Quarter 1 |
April 2011 | |||
Quarter 2 |
July 2011 | |||
Quarter 3 |
October 2011 | |||
Preliminary/Quarter 4 |
February 2012 | |||
Annual Report/Summary |
February/March 2012 | |||
Table of Contents
202
| Pence per share | ||||||||
| High | Low | |||||||
Quarter ended 31st March 2011* |
1270 | 1128 | ||||||
February 2011* |
1200 | 1128 | ||||||
January 2011 |
1270 | 1129 | ||||||
December 2010 |
1277 | 1231 | ||||||
November 2010 |
1262 | 1212 | ||||||
October 2010 |
1319 | 1221 | ||||||
September 2010 |
1290 | 1249 | ||||||
Quarter ended 31st December 2010 |
1319 | 1212 | ||||||
Quarter ended 30th September 2010 |
1290 | 1095 | ||||||
Quarter ended 30th June 2010 |
1281 | 1119 | ||||||
Quarter ended 31st March 2010 |
1340 | 1196 | ||||||
Quarter ended 31st December 2009 |
1334 | 1219 | ||||||
Quarter ended 30th September 2009 |
1252 | 1063 | ||||||
Quarter ended 30th June 2009 |
1117 | 987 | ||||||
Quarter ended 31st March 2009 |
1305 | 1003 | ||||||
Year ended 31st December 2008 |
1385 | 995 | ||||||
Year ended 31st December 2007 |
1493 | 1160 | ||||||
Year ended 31st December 2006 |
1577 | 1326 | ||||||
| US dollars per ADS | ||||||||
| High | Low | |||||||
Quarter ended 31st March 2011* |
39.86 | 36.33 | ||||||
February 2011* |
39.15 | 36.98 | ||||||
January 2011 |
39.86 | 36.33 | ||||||
December 2010 |
40.04 | 38.66 | ||||||
November 2010 |
40.85 | 38.28 | ||||||
October 2010 |
41.86 | 39.04 | ||||||
September 2010 |
40.47 | 38.78 | ||||||
Quarter ended 31st December 2010 |
41.86 | 38.28 | ||||||
Quarter ended 30th September 2010 |
40.47 | 33.78 | ||||||
Quarter ended 30th June 2010 |
39.57 | 32.34 | ||||||
Quarter ended 31st March 2010 |
42.97 | 37.03 | ||||||
Quarter ended 31st December 2009 |
42.91 | 38.72 | ||||||
Quarter ended 30th September 2009 |
40.03 | 34.36 | ||||||
Quarter ended 30th June 2009 |
36.56 | 29.11 | ||||||
Quarter ended 31st March 2009 |
39.24 | 27.27 | ||||||
Year ended 31st December 2008 |
54.36 | 32.02 | ||||||
Year ended 31st December 2007 |
59.35 | 47.87 | ||||||
Year ended 31st December 2006 |
58.38 | 50.15 | ||||||
| * | to 24th February 2011 |
The Queen Elizabeth II Conference Centre, 5th May 2011 |
||||
Broad Sanctuary, Westminster, |
||||
London SW1P 3EE |
||||
Tel: +44 (0)20 8047 5000
Tel: 1 888 825 5249 (US toll free)
Tel: +1 215 751 4000 (outside the USA)
Table of Contents
203
Aspect House, Spencer Road, Lancing, West Sussex BN99 6DA
www.shareview.co.uk
Tel: 0871 384 2991 (inside the UK)
Tel: +44 (0)121 415 7067 (outside the UK)
| • | Nominee dealing account and Individual Savings Account (SA) | |
| • | GlaxoSmithKline Corporate Sponsored Nominee | |
| • | Shareview service | |
| • | Share dealing service | |
| • | Dividend Reinvestment Plan. |
Oxford House, Oxford Road, Aylesbury, Bucks HP21 8SZ
Tel: +44 (0)1296 414141
BNY Mellon Shareowner Services
PO Box 358516
Pittsburgh, PA 15252-8516
www.bnymellon.com/shareowner
Tel: 1 877 353 1154 (US toll free)
Tel: +1 201 680 6825 (outside the USA)
email: shrrelations@bnymellon.com
| Number of | % of total | % of total | Number of | |||||||||||||
| accounts | accounts | shares | shares | |||||||||||||
Holding of shares |
||||||||||||||||
Up to 1,000 |
112,292 | 71 | 1 | 41,170,410 | ||||||||||||
1,001 to 5,000 |
35,996 | 23 | 1 | 77,156,810 | ||||||||||||
5,001 to 100,000 |
8,144 | 5 | 2 | 117,679,956 | ||||||||||||
100,001 to 1,000,000 |
827 | 1 | 5 | 302,361,401 | ||||||||||||
Over 1,000,000 |
411 | – | 91 | 5,132,089,600 | ||||||||||||
| 157,670 | 100 | 100 | 5,670,458,177 | |||||||||||||
Held by |
||||||||||||||||
Nominee companies |
28,100 | 18 | 74 | 4,195,447,676 | ||||||||||||
Investment and trust companies |
39 | – | – | 1,626,510 | ||||||||||||
Insurance companies |
8 | – | – | 4,834 | ||||||||||||
Individuals and other corporate bodies |
129,521 | 82 | 5 | 252,304,068 | ||||||||||||
BNY (Nominees) Limited |
1 | – | 13 | 746,880,931 | ||||||||||||
Held as Treasury shares by GlaxoSmithKline |
1 | – | 8 | 474,194,158 | ||||||||||||
| 157,670 | 100 | 100 | 5,670,458,177 | |||||||||||||
Table of Contents
204
Table of Contents
205
| Terms used in the Annual Report | US equivalent or brief description | |
Accelerated capital allowances
|
Tax allowance in excess of depreciation arising from the purchase of fixed assets that delay the charging and payment of tax. The US equivalent of tax depreciation. | |
American Depositary Receipt (ADR)
|
Receipt evidencing title to an ADS. Each GlaxoSmithKline ADR represents two Ordinary Shares. | |
American Depositary Shares (ADS)
|
Listed on the New York Stock Exchange; represents two Ordinary Shares. | |
Basic earnings per share
|
Basic income per share. | |
Called-up share capital
|
Ordinary Shares, issued and fully paid. | |
CER growth
|
Growth at constant exchange rates. | |
Combined Code
|
Guidelines required by the Listing Rules of the Financial Services Authority to address the principal aspects of Corporate Governance. | |
The company
|
GlaxoSmithKline plc. | |
Currency swap
|
An exchange of two currencies, coupled with a subsequent re-exchange of those currencies, at agreed exchange rates and dates. | |
Defined benefit plan
|
Pension plan with specific employee benefits, often called ‘final salary scheme’. | |
Defined contribution plan
|
Pension plan with specific contributions and a level of pension dependent upon the growth of the pension fund. | |
Derivative financial instrument
|
A financial instrument that derives its value from the price or rate of some underlying item. | |
Diluted earnings per share
|
Diluted income per share. | |
Employee Share Ownership Plan Trusts
|
Trusts established by the Group to satisfy share-based employee incentive plans. | |
Finance lease
|
Capital lease. | |
Freehold
|
Ownership with absolute rights in perpetuity. | |
Gearing ratio
|
Net debt as a percentage of total equity. | |
The Group
|
GlaxoSmithKline plc and its subsidiary undertakings. | |
Hedging
|
The reduction of risk, normally in relation to foreign currency or interest rate movements, by making off-setting commitments. | |
Intangible fixed assets
|
Assets without physical substance, such as computer software, brands, licences, patents, know-how and marketing rights purchased from outside parties. | |
Profit
|
Income. | |
Profit attributable to shareholders
|
Net income. | |
Share capital
|
Ordinary Shares, capital stock or common stock issued and fully paid. | |
Shareholders’ funds
|
Shareholders’ equity. | |
Share option
|
Stock option. | |
Share premium account
|
Additional paid-up capital or paid-in surplus (not distributable). | |
Shares in issue
|
The number of shares outstanding. | |
Subsidiary
|
An entity in which GlaxoSmithKline holds a majority shareholding and/or exercises control. | |
Treasury share
|
Treasury stock. | |
Turnover
|
Revenue. | |
Table of Contents
206
| (i) | the holding of Ordinary Shares in uncertificated form; | |
| (ii) | the transfer of title to Ordinary Shares by means of a system such as CREST; and | |
| (iii) | any provisions of the relevant regulations. |
Table of Contents
207
| (i) | be party to, or otherwise interested in, any contract with the company, or in which the company has a direct or indirect interest, | |
| (ii) | hold any other office or place of profit with the company (except that of auditor) in conjunction with his office of director for such period and upon such terms, including remuneration, as the Directors may decide; | |
| (iii) | act by himself or through a firm with which he is associated in a professional capacity for the company or any other company in which the company may be interested (otherwise than as auditor); | |
| (iv) | be or become a director of, or employed by, or otherwise be interested in any holding company or subsidiary company of the company or any other company in which the company may be interested; and | |
| (v) | be or become a director of any other company in which the company does not have an interest and which cannot reasonably be regarded as giving rise to a conflict of interest at the time of his appointment as director of that other company. |
Table of Contents
208
| (i) | he resigns or offers to resign and the board resolves to accept such offer; | |
| (ii) | his resignation is requested by all of the other directors and all of the other directors are not less than three in number; | |
| (iii) | he is or has been suffering from mental or physical ill health and the board resolves that his office be vacated; | |
| (iv) | he is absent without permission of the board from meetings of the board (whether or not an alternate director appointed by him attends) for six consecutive months and the board resolves that his office is vacated; | |
| (v) | he becomes bankrupt or compounds with his creditors generally; | |
| (vi) | he is prohibited by law from being a director; | |
| (vii) | he is removed from office pursuant to the Articles or the Companies Acts. |
Table of Contents
209
| 1. | the Company must satisfy the audit committee requirements of the Securities and Exchange Commission (the “SEC”); | |
| 2. | the Chief Executive Officer (the “CEO”) must promptly notify the NYSE in writing
after any executive officer of the Company becomes aware of any non-compliance with any applicable provisions of the NYSE’s corporate governance standards; |
|
| 3. | the Company must submit an annual
affirmation to the NYSE affirming GlaxoSmithKline’s compliance with applicable NYSE corporate
governance standards, and submit interim affirmations to the NYSE notifying it of specified changes
to the audit committee or a change to the status of the Company as a foreign private issuer; and |
|
| 4. | the Company must provide a brief description of any significant differences between its corporate governance practices and those followed by US companies under the NYSE listing standards. |
| NYSE | Description of differences between GlaxoSmithKline’s governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
Director Independence |
||
1. Listed companies must have a majority of
independent directors.
|
GlaxoSmithKline complies with the equivalent domestic requirements contained in the UK Corporate Governance Code (formerly the Combined Code). A new edition of the UK Corporate Governance Code, for reporting years beginning on or after 29th June 2010 was issued in June 2010. | |
| The UK Corporate Governance Code provides that the board of directors of GlaxoSmithKline (the “Board”) and its committees should have the appropriate balance of skills, experience, independence and knowledge of the company to enable them to discharge their respective duties and responsibilities effectively (B.1). The Board should include an appropriate combination of Executive and Non-Executive Directors (and, in particular, independent Non-Executive Directors) such that no individual or small group of individuals can dominate the Board’s decision taking (B.1). At least half the Board, excluding the Chairman, should comprise Non-Executive Directors determined by the Board to be independent (B.1.2). The roles of Chairman and Chief Executive should not be exercised by the same individual. The division of responsibilities between the Chairman and Chief Executive should be clearly established, set down in writing and agreed by the Board (A.2.1). | ||
| The Board considers that Professor Sir Roy Anderson, Dr Burns, Mr Culp, Sir Crispin Davis, Sir Deryck Maughan, Mr James Murdoch, Dr Podolsky, Mr de Swaan and Sir Robert Wilson are “independent” under the UK Corporate Governance Code. | ||
| A majority of the Board members are “independent” Non-Executive Directors and, in accordance with the recommendations of the UK Corporate Governance Code, the Board has appointed one of the “independent” Non-Executive Directors as Senior Independent Director to provide a sounding board for the Chairman and act as an intermediary for other Non-Executive Directors where necessary (A.4.1). | ||
Table of Contents
210
| NYSE | Description of differences between GlaxoSmithKline’s governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
2. In order to tighten the definition of “independent director” for
purposes of these standards: (a) No director qualifies as “independent” unless the board of
directors affirmatively determines that the director has no material
relationship with the listed company (either directly or as a partner,
shareholder or officer of an organization that has a relationship with
the company). (b) In addition, a director is not independent if: (i) The director is, or has been within the last three years, an
employee of the listed company, or an immediate family
member is, or has been within the last three years, an executive
officer, of the listed company. (ii) The director has received, or has an immediate family member
who has received, during any twelve-month period within the
last three years, more than $120,000 in direct compensation
from the listed company, other than director and committee
fees and pension or other forms of deferred compensation for
prior service (provided such compensation is not contingent in
any way on continued service). (iii) (A) The director is a current partner or employee of a firm that is
the listed company’s internal or external auditor; (B) the director
has an immediate family member who is a current partner of
such a firm; (C) the director has an immediate family member
who is a current employee of such a firm and who personally
works on the listed company’s audit; or (D) the director or an
immediate family member was within the last three years a
partner or employee of such a firm and personally worked on
the listed company’s audit within that time. (iv) The director or an immediate family member is, or has been
within the last three years, employed as an executive officer of
another company where any of the listed company’s present
executive officers at the same time serves or served on that
company’s compensation committee. (v) The director is a current employee, or an immediate family
member is a current executive officer, of a company that has
made payments to, or received payments from, the listed
company for property or services in an amount which, in any of
the last three fiscal years, exceeds the greater of $1 million, or
2% of such other company’s consolidated gross revenues. (For the purposes of these standards “executive officer” is defined to have the meaning specified for the term “officer” in Rule 16a-1(f) under the Securities Exchange Act of 1934, as amended the “Exchange Act”). |
GlaxoSmithKline complies with the corresponding domestic
requirements contained in the UK Corporate Governance Code,
which sets out the principles for the Company to determine
whether a director is “independent”. The Board is required to determine and state its reasons for the determination of whether directors are independent in character and judgment and whether there are relationships or circumstances which are likely to affect, or could affect, the directors’ judgment. In undertaking this process, the Board is required, amongst other factors, to consider if the director: (a) has been an employee of GlaxoSmithKline within the last five
years; (b) has, or has had within the last three years, a material business
relationship with the Company either directly or as a partner,
shareholder, director or senior employee of a body that has such
a relationship with the Company; (c) has received or receives additional remuneration from the
Company apart from a director’s fee, participates in the
Company’s share option or a performance-related pay scheme,
or is a member of the Company’s pension scheme; (d) has close family ties with any of the Company’s advisers,
directors or senior employees; (e) holds cross-directorships or has significant links with other
directors through involvement in other companies or bodies; (f) represents a significant shareholder; or (g) has served on the Board for more than nine years from the date
of his or her first election (B.1.1). The Board considers all its Non-Executive Directors to be independent in character and judgment and has concluded that all its Non-Executive Directors are independent in accordance with the UK Corporate Governance Code. A new requirement introduced by the UK Corporate Governance Code is that all Directors should be subject to annual election by shareholders. GlaxoSmithKline intends to comply with this requirement at its 2011 Annual General Meeting. The UK Corporate Governance Code also provides that the Board should undertake a formal and rigorous annual evaluation of its own performance and that of its committees and individual Directors (B.6). In addition, the evaluation of the Board should be externally facilitated at least every three years and a statement should be made available of whether an external facilitator has any other connection with the Company (B.6.2). All Directors should receive an induction on joining the Board (B.4). The Chairman should regularly review and agree with each Director their training and development needs (B.4.2). |
|
Table of Contents
211
| NYSE | Description of differences between GlaxoSmithKline’s governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
3. To empower non-management directors to serve as a more
effective check on management, the non-management
directors of each listed company must meet at regularly
scheduled executive sessions without management.
|
GlaxoSmithKline complies with the equivalent domestic requirements set out in the UK Corporate Governance Code, which requires that the Chairman of GlaxoSmithKline should hold meetings with the Non-Executive Directors without executives present. The Non-Executive Directors, led by the senior independent director, also meet without the Chairman present to appraise the Chairman’s performance (A.4.2). | |
| The Chairman promotes a culture of openness and debate by facilitating the effective contribution of Non-Executive Directors (A.3) and, in particular, ensuring constructive relations between Executive and Non-Executive Directors (A.3). In addition, the Chairman is responsible for ensuring that all Directors are made aware of shareholders’ concerns (E.1). | ||
| Nominating/corporate governance committee | ||
4.
(a) Listed companies must have a nominating/corporate
governance committee composed entirely of independent
directors. (b) The nominating/corporate governance committee must have
a written charter that addresses: (i) the committee’s purpose and responsibilities
– which, at minimum, must be to: identify individuals
qualified to become board members, consistent with criteria
approved by the board, and to select, or to recommend that the
board select, the director nominees for the next annual
meeting of shareholders; develop and recommend to the
board a set of corporate governance guidelines applicable
to the corporation; and oversee the evaluation of the
board and management; and (ii) an annual performance evaluation of
the committee. |
GlaxoSmithKline complies with the corresponding domestic
requirements set out in the UK Corporate Governance Code,
which requires that GlaxoSmithKline should have a Nominations
Committee that is comprised of a majority of independent Non-
Executive Directors (B.2.1). GlaxoSmithKline’s Nominations Committee has written terms of reference in accordance with the UK Corporate Governance Code. The terms of reference are available and explain the Nominations Committee’s role and the authority delegated to it by the Board (B.2.1). The Nominations Committee reviews the structure, size, diversity (including gender diversity), and composition of the Board and appointment of members to the Board and the Corporate Executive Team (the “CET”), and makes recommendations to the Board as appropriate. The Committee also monitors the planning of succession for the Board and Senior Management. In compliance with the UK Corporate Governance Code, the terms and conditions of appointment of Non-Executive Directors are available for inspection (B.3.2). The UK Corporate Governance Code requires that a separate section in the Company’s Annual Report describe the work of the Nominations Committee in discharging its duties, including the process it has used in relation to Board appointments (B.2.4). The Board is responsible for regularly reviewing its corporate governance standards and practices. The Company Secretary oversees corporate governance matters for the Group. The Company Secretary is responsible for advising the Board through the Chairman on all corporate governance matters. Domestic requirements do not mandate that GlaxoSmithKline establish a corporate governance committee |
|
Table of Contents
212
| NYSE | Description of differences between GlaxoSmithKline’s governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
Management resources and compensation committee |
||
5.
(a) Listed companies must have a compensation committee
composed entirely of independent directors. (b) The compensation committee must have a written charter
that addresses: (i) the committee’s purpose and responsibilities – which, at
minimum, must be to have direct responsibility to: (A) review and approve corporate goals and objectives
relevant to CEO compensation, evaluate the CEO’s
performance in light of those goals and objectives,
and, either as a committee or together with the other
independent directors (as directed by the board),
determine and approve the CEO’s compensation level
based on this evaluation; (B) make recommendations to the board with respect
to non-CEO executive officer compensation, and
incentive-compensation and equity-based plans that
are subject to board approval; and (C) prepare the disclosure required by item 407(e)(5)
or Regulation S-K under the Exchange Act; (ii) an annual performance evaluation of the compensation
committee. |
GlaxoSmithKline complies with the equivalent domestic requirements
set out in the UK Corporate Governance Code, which requires that
GlaxoSmithKline should have a Remuneration Committee that is
comprised of at least three “independent” Non-Executive Directors in
addition to the Chairman (D.2.1). GlaxoSmithKline’s Remuneration Committee has written terms of reference in accordance with the UK Corporate Governance Code (D.2.1). The Remuneration Committee determines the terms of service and remuneration of the Executive Directors and members of the CET and, with the assistance of external independent advisers, it evaluates and makes recommendations to the Board on overall executive remuneration policy (the Chairman and the CEO are responsible for evaluating and making recommendations to the Board on the remuneration of Non-Executive Directors). The UK Corporate Governance Code provides that the Remuneration Committee: (a) should consult with the Chairman and/or CEO about their
proposals relating to the remuneration of other Executive Directors
(D.2) and should delegate responsibility for setting remuneration
for all Executive Directors and the Chairman, including pension
rights and any compensation payments (D.2.2); (b) should recommend and monitor the level and structure of
remuneration for senior management (D.2.2); (c) should consider what compensation commitments (including pension contributions and all other elements) the directors’ terms
of appointment would entail in the event of early termination
(D.1.4.); (d) should invite shareholders specifically to approve all new long-term
incentive schemes and significant changes to existing schemes
(D.2.4.); (e) should judge where to position the Company relative to other
companies and should be sensitive to pay and employment
conditions elsewhere in the group, especially when determining
annual salary increases (D.1); and (f) should consider whether the Directors should be eligible for
annual bonuses and benefits under long-term incentive schemes
(Schedule A), bearing in mind that performance-related elements
of Executive Directors’ remuneration should be designed to
promote the long-term success of the Company (D.1 and D.1.1). The UK Corporate Governance Code requires that payouts under incentive schemes should be subject to non-financial performance criteria where appropriate and compatible with the Company’s risk policies and systems (Schedule A). In addition, remuneration of Non-Executive Directors should not include performance-related elements (D.1.3). |
|
Table of Contents
213
plc’s corporate governance practice
| NYSE | Description of differences between GlaxoSmithKline’s governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
Audit & Risk committee |
||
6. Listed companies must have an audit committee that
satisfies the requirements of Rule 10A-3 under the Exchange
Act.
|
GlaxoSmithKline complies with equivalent domestic requirements set out in the UK Corporate Governance Code, which require that GlaxoSmithKline have an Audit Committee that is comprised entirely of “independent” Non-Executive Directors (C.3.1). The Board should satisfy itself that at least one member of the Audit Committee has recent and relevant financial experience. The Board of Directors approved and adopted new terms of reference and accepted proposals to change the name of the Committee to the Audit & Risk Committee with effect from 10th December 2009; | |
| The UK Corporate Governance Code provides that the Audit Committee: |
||
(a) monitors the integrity of the financial statements of the Company
and any formal announcements relating to the Company’s financial
performance, reviewing significant financial reporting judgments
contained in them (C.3.2); |
||
(b) review the Company’s internal financial controls and internal
control and risk management systems; |
||
(c) monitor and review the effectiveness of the Company’s internal
audit function (C.3.2); |
||
(d) make recommendations to the Board, for it to put to the
shareholders for their approval in general meeting, in relation
to the appointment, re-appointment and removal of the
external auditor and to approve the remuneration and terms of
engagement of the external auditor (C.3.2); |
||
(e) review and monitor the external auditor’s independence and
objectivity and the effectiveness of the audit process, taking into
consideration relevant UK professional and regulatory requirements
(C.3.2); |
||
(f) develop and implement policy on the engagement of external
auditors to supply non-audit services, taking into account relevant
ethical guidance regarding the provision of non-audit services by the
external audit firm, and to report to the Board, identifying any matters
in respect of which it considers that action or improvement is needed
and making recommendations as to the steps to be taken (C.3.2); |
||
(g) review arrangements by which the staff of the company may, in
confidence, raise concerns about possible improprieties in matters of
financial reporting or other matters (C.3.4) |
||
| GlaxoSmithKline’s Audit & Risk Committee meets the requirements of the Sarbanes-Oxley Act of 2002 in that: | ||
• each member of the Audit & Risk Committee is deemed to be
“independent” in accordance with the Securities Exchange Act of
1934, as amended, and applicable NYSE and UK requirements; |
||
• the Audit & Risk Committee, amongst other things, is responsible
for recommending the appointment, compensation, maintenance
of independence and oversight of the work of any registered public
accounting firm engaged for the purpose of preparing or issuing
an audit report or performing other audit, review or attest services
for the Company, and each such accounting firm must report
directly to the Audit & Risk Committee; |
||
Table of Contents
214
plc’s corporate governance practice
| NYSE | Description of differences between GlaxoSmithKline's governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
6. continued
|
• the Audit & Risk Committee has established a procedure for
the receipt, retention and treatment of complaints regarding
accounting, internal accounting controls or auditing matters, and
for the confidential, anonymous submission by employees of
concerns regarding questionable accounting or auditing matters; |
|
• the Audit & Risk Committee has the authority to engage
independent counsel and other advisors as it determines necessary
to carry out its duties; and |
||
• GlaxoSmithKline must provide appropriate funding for the Audit
& Risk Committee. |
||
| The Board has determined that Mr de Swaan has the appropriate qualifications and background to be an “Audit Committee Financial Expert” as defined in rules promulgated by the SEC under the Sarbanes-Oxley Act of 2002. | ||
7. |
||
(a) The audit committee must have a minimum of three members.
All audit committee members must satisfy the requirements
for independence set out in Section 303 A.02 and, in the
absence of an applicable exemption, Rule 10A-3(b)(1) under
the Exchange Act. (b) The audit committee must have a written charter that
addresses: (i) the committee’s purpose – which, at minimum, must be
to: (A) assist board oversight of (1) the integrity of the
listed company’s financial statements, (2) the listed
company’s compliance with legal and regulatory
requirements, (3) the independent auditor’s
qualifications and independence, and (4) the
performance of the listed company’s internal audit
function and independent auditors; and (B) prepare the disclosure required by Item 407(d)(3)(i) of
Regulation S-K under the Exchange Act; (ii) an annual performance evaluation of the audit committee;
and (iii) the duties and responsibilities of the audit committee –
which, at a minimum, must include those set out in Rule
10A-3(b)(2), (3), (4) and (5) of the Exchange Act as well
as to: (A) at least annually, obtain and review a report by the
independent auditor describing: the firm’s internal
quality-control procedures; any material issues
raised by the most recent internal quality-control
review, or peer review, of the firm, or by any inquiry
or investigation by governmental or professional authorities, within the preceding five years, respecting
one or more independent audits carried out by the
firm, and any steps taken to deal with any such
issues; and (to assess the auditor’s independence) all
relationships between the independent auditor and the
listed company;
|
GlaxoSmithKline complies with the equivalent domestic requirements
set out in the UK Corporate Governance Code, which require that
the Audit Committee should be comprised of a minimum of three
“independent” Non-Executive Directors. GlaxoSmithKline’s Audit & Risk Committee has written terms of reference in accordance with the UK Corporate Governance Code (C.3.3). The Committee’s main responsibilities include reviewing the financial reporting process, the system of internal control and overseeing the identification and management of risks, the external and internal process and for monitoring compliance with laws, regulations and ethical codes of practice, including review throughout the year of integrated assurance reports comprising business unit and associated consolidated internal audit reports. The UK Corporate Governance Code requires that a separate section in the Company’s Annual Report describe the work of the Committee in discharging its duties (C.3.3). Please see section 6 above for a description of the main role and responsibilities of the Audit & Risk Committee. |
|
Table of Contents
215
| NYSE | Description of differences between GlaxoSmithKline’s governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
7. continued |
||
(B) meet to review and
discuss the listed company’s
annual audited financial statements and quarterly
financial statements with management and the
independent auditor, including reviewing the listed
company’s specific disclosures under “Management’s
Discussion and Analysis of Financial Condition and
Results of Operations”; |
||
(C) discuss the listed
company’s earnings press releases,
as well as financial information and earnings guidance
provided to analysts and rating agencies; |
||
(D) discuss policies
with respect to risk assessment and
risk
management; |
||
(E) meet separately,
periodically, with management, with
internal auditors (or other personnel responsible for
the internal audit function) and with independent
auditors; |
||
(F) review with the
independent auditor any audit
problems or difficulties and management’s response; |
||
(G) set clear hiring
policies for employees or former
employees of the independent auditors; and |
||
(H) report regularly to
the board of directors. |
||
(c) Each listed company must have an internal audit
function. |
||
8. Shareholders must be given the opportunity to vote on all
equity-compensation plans and material revisions thereto,
except for employment inducement awards, certain grants,
plans and amendments in the context of mergers and
acquisitions, and certain specific types of plans. |
GlaxoSmithKline complies with corresponding domestic requirements in the Listing Rules of the UK Listing Authority, which mandate that the Company must seek shareholder approval for employee share schemes (D.2.4 and Listing Rule 9.4). Please see section 5(d) above. | |
Corporate governance guidelines |
||
9. Listed companies must adopt and disclose corporate
governance guidelines.
|
GlaxoSmithKline complies with corresponding domestic requirements in the Listing Rules of the UK Listing Authority and the UK Corporate Governance Code, which require that GlaxoSmithKline include an explanation in its Annual Report of how it complies with the principles of the UK Corporate Governance Code and that it confirm that it complies with the Code’s provisions or, where it does not, provide an explanation of why it does not comply (Listing Rule 9.8.6). In addition, for accounting periods beginning on or after 29th June 2008, issuers are required to make certain mandatory corporate governance statements in the Directors’ Report in accordance with new UK Disclosure and Transparency Rules, DTR 7, which was issued by the UK Financial Services Authority to implement the eighth Company Law Directive; GlaxoSmithKline complies with these requirements in its 2010 Annual Report. | |
Table of Contents
216
| NYSE | Description of differences between GlaxoSmithKline’s governance | |
| Corporate Governance Standards | practice and the NYSE Corporate Governance Standards | |
10. Listed companies must adopt and disclose a code of business
conduct and ethics for directors, officers and employees, and
promptly disclose any waivers of the code for directors or
executive officers.
|
GlaxoSmithKline’s Code of Conduct for all employees, including the CEO, CFO and other senior financial officers, is available. | |
Description of significant differences |
||
11. Listed foreign private issuers must disclose any significant
ways in which their corporate governance practices differ
from those followed by domestic companies under NYSE
listing standards.
|
GlaxoSmithKline fulfills this requirement by publishing this document. GlaxoSmithKline fulfils this requirement by including this disclosure on its annual report on Form 20-F. | |
Listed foreign private issuers are required to provide this
disclosure in the English language and in their annual reports
filed on Form 20-F. |
||
Table of Contents
217
| Received during 2010 | Received during 2010 and 2011 | Claimed in respect of 2010 | ||||||||||
| Direct and indirect payments by the Depositary | in respect of 2009 | in respect of 2010 | but not yet received | |||||||||
Reimbursement of NYSE listing fees |
– | $350,483 | – | |||||||||
Reimbursement of legal fees claimed in US dollars |
$333,735 | $225,000 | $200,000 | |||||||||
Reimbursement of legal fees claimed in Sterling |
£23,268 | £91,198.25 | – | |||||||||
Reimbursement of PCAOB fees |
$161,700 | $177,500 | – | |||||||||
Reimbursement of Annual Report production costs1 |
£300,437 | £216,208.94 | – | |||||||||
Reimbursement of Investor Relations expenses2 |
$294,084 | $321,306.19 | $103,477.16 | |||||||||
Distribution of annual general meeting materials |
– | $427,480.94 | – | |||||||||
Tabulation of voting instructions cards |
– | $32,533.90 | – | |||||||||
Reimbursement of other programme-related expenditures
claimed in US dollars |
$16,050 | $5,852.20 | $3,030.90 | |||||||||
Reimbursement of
other programme-related expenditures claimed in Sterling |
£32,280 | – | £7,099.35 | |||||||||
| 1 | Annual report production costs include SEC filing fees. | |
| 2 | Investor relations expenses include travel expenses, fees of investor relations consultants, expenses involved in arranging investor relations meetings and telephone expenses. |
Table of Contents
218
| Exhibit No. | Description | |
1.1
|
Memorandum and Articles of Association of the Registrant as in effect on the date hereof. | |
2.1
|
Deposit Agreement among the Registrant and The Bank of New York, as Depositary, and the holders from time to time of the American Depositary Receipts issued thereunder, including the form of American Depositary Receipt, is incorporated by reference to the Registration Statement on Form F-6 (No. 333-148017) filed with the Commission on December 12, 2007. | |
4.1
|
UK Service Agreement between GlaxoSmithKline Services Unlimited and Julian Heslop is incorporated by reference to Exhibit 4.3 to the Registrant’s Annual Report on Form 20-F filed with the Commission on March 3, 2006. | |
4.2
|
Service Agreement between SmithKline Beecham Corporation and Moncef Slaoui is incorporated by reference to Exhibit 4.4 to the Registrant’s Annual Report on Form 20-F filed with the Commission on February 29, 2008. | |
4.3
|
Amendment to Service Agreement between GlaxoSmithKline LLC (formerly known as SmithKline Beecham Corporation) and Moncef Slaoui dated December 21, 2010. | |
4.4
|
UK Service Agreement between GlaxoSmithKline Services Unlimited and Andrew Witty is incorporated by reference to Exhibit 4.5 to the Registrant’s Annual Report on Form 20-F filed with the Commission on February 29, 2008. | |
4.5
|
Amendment to UK Service Agreement between GlaxoSmithKline Services Unlimited and Andrew Witty dated June 18, 2008 is incorporated by reference to Exhibit 4.4 to the Registrant’s Annual Report on Form 20-F filed with the Commission on March 4, 2009. | |
4.6
|
Amendment to UK Service Agreement between GlaxoSmithKline Services Unlimited and Andrew Witty dated February 4, 2010 is incorporated by reference to Exhibit 4.5 to the Registrant’s Annual Report on Form 20-F filed with the Commission on March 1,2010. | |
4.7
|
UK Service Agreement between GlaxoSmithKline Services Unlimited and Simon Dingemans dated September 8, 2010 | |
8.1
|
A list of the Registrant’s principal subsidiaries is incorporated by reference to pages 175 to 177 of this Annual Report on Form 20-F. | |
12.1
|
Certification Required by Rule 13a-14(a) or 15d-14(a) under the Securities Exchange Act of 1934 – Andrew Witty. | |
12.2
|
Certification Required by Rule 13a-14(a) or 15d-14(a) under the Securities Exchange Act of 1934 – Julian Heslop. | |
13.1
|
Certification Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (Subsections (a) and (b) of Section 1350, Chapter 63 of Title 18, United States Code). | |
15.1
|
Consent of PricewaterhouseCoopers LLP. | |
Table of Contents
219
Table of Contents
220
| Item | Page | |||
1
|
Identity of directors, senior management and advisers | n/a | ||
2
|
Offer statistics and expected timetable | n/a | ||
3
|
Key information | |||
A
|
Selected financial data | 194-196 | ||
D
|
Risk factors | 53-57 | ||
4
|
Information on the company | |||
A
|
History and development of the company | 155-157,219 | ||
B
|
Business overview | |||
| Products | 14-17 | |||
| Economy and market | 20,22-27,34-36 | |||
| Manufacture and supply | 19 | |||
| Marketing and distribution | 19,40,164 | |||
| Intellectual property | 14-16 | |||
| Competition | 14-17 | |||
| Regulation | 18 | |||
| Research and development | 10-13,28 | |||
| Environmental sustainability | 32 | |||
| Responsible business and community investment | 29-31 | |||
C
|
Organisational structure | 175-177 | ||
D
|
Property, plant and equipment | 41 | ||
| Note 6 – Segment information | 117-121 | |||
| Note 17 – Property, plant and equipment | 130-131 | |||
4A
|
Unresolved staff comments | n/a | ||
5
|
Operating and financial review and prospects | |||
A
|
Operating results | |||
| 2010 and 2009 | 22-28,34-40 | |||
| 2009 and 2008 | 47-52 | |||
B
|
Liquidity and capital resources | 41-46 | ||
C
|
Research and development, patents and licenses, etc. | 10-17 | ||
D
|
Trend information | 34-40 | ||
E
|
Off-balance sheet arrangements | n/a | ||
F
|
Tabular disclosure of contractual obligations | 43 | ||
6
|
Directors, senior management and employees | |||
A
|
Directors and senior management | 58-63 | ||
B
|
Compensation | |||
| Remuneration Report | 81-101 | |||
C
|
Board practices | |||
| Corporate governance | 62-80 | |||
D
|
Employees | 33 | ||
| Note 10 – Employee costs | 125 | |||
| Note 28 – Pensions and post-employment benefits | 140-148 | |||
| Financial record | 196 | |||
E
|
Share ownership | |||
| Note 42 – Employee share schemes | 171-174 | |||
| Share options | 91 | |||
| Incentive plans | 87-88 | |||
| Directors’ interests | 96 | |||
7
|
Major shareholders and related party transactions | |||
A
|
Major shareholders | 65,69,201 | ||
B
|
Related party transactions | |||
| Note 35 – Related party transactions | 154 | |||
| Item | Page | |||
8
|
Financial information | |||
A
|
Consolidated statements and other financial information Financial statements | See item 18 | ||
| Dividend policy | 4 | |||
| Note 44 – Legal proceedings | 178-185 | |||
B
|
Significant changes | |||
| Note 40 – Post balance sheet events | 161 | |||
9
|
The offer and listing | |||
A
|
Offer and listing details | |||
| Share price listing | 201,202 | |||
C
|
Markets | 202 | ||
10
|
Additional information | |||
B
|
Articles of Association | 206-208 | ||
C
|
Material contracts | n/a | ||
D
|
Exchange controls | 202 | ||
E
|
Taxation | 204 | ||
H
|
Documents on display | 202 | ||
11
|
Quantitative and qualitative disclosures about market risk | |||
| Treasury policies | 45-46 | |||
| Note 41 – Financial instruments and related disclosures | 162-171 | |||
12
|
Description of securities other than equity securities | |||
D
|
American Depositary Shares | 217 | ||
13
|
Defaults, dividend arrearages and delinquencies | n/a | ||
14
|
Material modifications to the rights of security holders and use of proceeds | n/a | ||
15
|
Controls and procedures | 71-73,79 | ||
16
|
[Reserved] | |||
16A
|
Audit Committee financial expert | 75 | ||
16B
|
Code of ethics | 77 | ||
16C
|
Principal accountant fees and services | |||
| Note 9 – Operating profit | 124 | |||
16D
|
Exemptions from the listing standard for audit committees | |||
| n/a | ||||
16E
|
Purchases of equity securities by the issuer and affiliated purchasers | |||
| Note 33 – Share capital and share premium account | 152 | |||
16F
|
Change in registrants’ certifying accountant | n/a | ||
16G
|
Corporate Governance | 209-216 | ||
17
|
Financial statements | n/a | ||
18
|
Financial statements | |||
| Report of Independent Regulated Public Accounting Firm | 103 | |||
| Consolidated income statement | 104-105 | |||
| Consolidated statement of comprehensive income | 104-105 | |||
| Consolidated balance sheet | 106 | |||
| Consolidated statement of changes in equity | 107 | |||
| Consolidated cash flow statement | 108 | |||
| Notes to the financial statements | 109-185 | |||
19
|
Exhibits | Footnote (i), 218 |
||
| Footnote (i) – see the company’s Form 20-F filing with the Securities and Exchange Commission. | ||||
Table of Contents
|
|
||||||
|
|
||||||

|
||||||
www.gsk.com Head Office and Registered Office GlaxoSmithKline plc |
||||||
980 Great West Road |
||||||
Brentford, Middlesex TW8 9GS |
||||||
United Kingdom |
||||||
Tel: +44 (0)20 8047 5000 |
||||||
Registered number: 3888792 |
||||||
Printed in the UK. The paper used |
||||||
in the production of this document |
||||||
is made from 100% post consumer |
||||||
waste. The pulp is bleached using |
||||||
a totally chlorine free process. |
||||||
Table of Contents
| GlaxoSmithKline plc |
||||
| March 4, 2011 | By: | /s/ Julian Heslop | ||
| Julian Heslop | ||||
| Chief Financial Officer | ||||
