EXHIBIT 99.2
2018
HALF-YEAR MANAGEMENT REPORT
CONTENTS
| 2 HALF-YEAR MANAGEMENT REPORT |
51 | |||
| A/ Significant events of the first half of 2018 |
51 | |||
| B/ Events subsequent to June 30, 2018 |
55 | |||
| C/ Consolidated financial statements for the first half of 2018 |
56 | |||
| D/ Risk factors and related party transactions |
82 | |||
| E/ Outlook |
82 | |||
| F/ Appendix – Research and Development Pipeline |
84 | |||
2 HALF-YEAR MANAGEMENT REPORT
A/ SIGNIFICANT EVENTS OF THE FIRST HALF OF 2018
A.1. PHARMACEUTICALS
A.1.1. ACQUISITIONS AND ALLIANCES
On January 22, 2018, Sanofi and Bioverativ Inc., a biotechnology company focused on therapies for hemophilia and other rare blood disorders, entered into a definitive agreement under which Sanofi offered to acquire all of the outstanding shares of Bioverativ for $105 per share in cash, representing an equity value of approximately $11.6 billion (on a fully diluted basis). The transaction was unanimously approved by both the Sanofi and Bioverativ Boards of Directors. The acquisition is expected to be immediately accretive to Sanofi’s business earnings per share in 2018, and up to 5% accretive in 2019. The minimum tender condition and all of the other conditions having been satisfied, on March 8, 2018 Sanofi and its wholly-owned subsidiaries Sanofi-Aventis NA Holding, Inc. and Blink Acquisition Corp. accepted all the shares validly tendered into the offer. Bioverativ remains in existence as an indirect, wholly-owned subsidiary of Sanofi.
In January 2018, Sanofi and Ablynx, a biopharmaceutical company engaged in the discovery and development of Nanobodies®, entered into a definitive agreement under which Sanofi offered to acquire all of the outstanding ordinary shares, including shares represented by American Depositary Shares (ADSs), warrants and convertible bonds of Ablynx, at a price per Ablynx share of €45 in cash, valuing Ablynx at approximately €3.9 billion (on a fully diluted basis). The transaction was unanimously approved by both the Sanofi and Ablynx Boards of Directors. On May 14, 2018, Sanofi and Ablynx announced that at the end of the initial acceptance period of the tender offer, Sanofi held 95.60% of the outstanding shares of Ablynx, of which more than 90% had been acquired through the offers. On June 19, 2018, after the end of the squeeze-out tender period, Sanofi announced that it held all the outstanding shares of Ablynx.
In January 2018, Sanofi and Regeneron announced that they were accelerating and expanding investment in the clinical development of (i) the PD-1 (programmed cell death protein 1) antibody cemiplimab in oncology, (ii) dupilumab in Type 2 allergic diseases and (iii) the anti-IL33 monoclonal antibody (REGN 3500/SAR 440340). Both of these breakthrough therapies have the potential to benefit a number of different patient populations, and these strategic investments will enable the companies to evaluate cemiplimab and dupilumab in broad clinical development programs.
Also in January 2018, Sanofi and Alnylam announced a strategic restructuring of their RNAi therapeutics alliance to streamline and optimize development and commercialization of certain products for the treatment of rare genetic diseases. Specifically:
| • | Sanofi obtains global development and commercialization rights to fitusiran, an investigational RNAi therapeutic currently in development for the treatment of people with hemophilia A and B. Global commercialization of fitusiran, upon approval, will be done by Sanofi Genzyme, Sanofi’s Specialty Care Global Business Unit. Alnylam will receive royalties based on net sales of fitusiran products. |
| • | Alnylam obtains global development and commercialization rights to its investigational RNAi therapeutics programs for the treatment of ATTR amyloidosis, including patisiran and ALN-TTRsc02. Sanofi will receive royalties based on net sales of those products. |
| • | With respect to other products falling under the RNAi therapeutics alliance, the material terms agreed between Alnylam and Sanofi Genzyme in 2014 remain unchanged. |
On March 8, 2018, Evotec AG and Sanofi entered into exclusive negotiations to accelerate infectious disease research and development by creating a new open innovation platform near Lyon, France, to be managed by Evotec. In support of this platform, Sanofi will license most of its infectious disease research and early-stage development portfolio and transfer its infectious disease research unit to Evotec. The transaction excludes the vaccine R&D unit and related projects. This joint initiative will bring together more than 150 scientists within Evotec, all of them experts in this field. Sanofi is to pay Evotec an initial, one-time, upfront cash payment of €60 million on the closing date of the deal, and will provide significant further long-term funding. Sanofi retains certain option rights on the development, manufacturing and commercialization of anti-infective products. Under the agreement, Evotec will integrate Sanofi’s infectious disease research unit, which includes more than 100 employees, into its global drug discovery and development operations. The transfer is backed by specific undertakings from Evotec to safeguard jobs for a five-year period and to maintain its activities in the Lyon region, to take advantage of the local scientific and medical ecosystem. Evotec will expand its existing long-term initiatives focused on innovation to fight infectious diseases; those initiatives include maintaining a portfolio of projects aimed at diseases affecting the developing world. The focus of drug discovery will be on new mode-of-action antimicrobials.
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 51
In April 2018, Advent International (Advent) and Sanofi entered into exclusive negotiations under which Advent would acquire Zentiva, Sanofi’s European generics business for €1.9 billion1. Advent’s offer is firm, binding and fully financed. Advent is a global private equity firm, with over 25 years’ experience of investing in the healthcare sector. It has extensive experience of executing corporate carve-outs and will work collaboratively with Sanofi to form a new independent operation. Advent will support the Zentiva management team to invest in the company’s operations, production facilities and R&D pipeline. On June 28, 2018, Sanofi and Advent announced that they had finalized negotiations for the acquisition. The signing of this agreement marked a critical step towards the closing of the deal and the transfer of the Zentiva business to Advent, which is expected to take place in the fourth quarter of 2018. The transaction is still subject to clearance from the relevant regulatory authorities, and is being conducted in strict compliance with obligations to consult employee representative bodies within Sanofi. As of June 30, 2018, completion of the transaction being regarded as highly probable, and the initial tender offer acceptance period having expired, all assets of the European Generics business included in the sale, and all liabilities directly related to those assets, are presented in the line items Assets held for sale or exchange and Liabilities related to assets held for sale or exchange, respectively, in the consolidated balance sheet. The operations of the held-for-sale European Generics business will continue to be presented within the relevant line items in the income statement until the effective date of sale, given that the business is not an operating segment of Sanofi and does not qualify as a discontinued operation under IFRS 5 (Non-Current Assets Held for Sale and Discontinued Operations).
A.1.2. FILINGS FOR MARKETING AUTHORIZATION FOR NEW PRODUCTS
In January 2018, Sanofi and Regeneron announced that the Ministry of Health, Labor and Welfare in Japan had granted manufacturing and marketing authorization for Dupixent® in moderate-to-severe atopic dermatitis in adults inadequately controlled by existing treatments.
In March 2018, the European Medicines Agency (EMA) accepted for review Sanofi’s marketing authorization application for sotagliflozin (developed in partnership with Lexicon Pharmaceuticals, Inc.) in the treatment of adults with type 1 diabetes. A license application for sotagliflozin in the treatment of type 1 diabetes has also been filed in the United States, and was accepted for review by the US Food and Drug Administration (FDA) in early June 2018. A decision from the FDA is expected in March 2019.
In April 2018, the EMA accepted for review Sanofi’s marketing authorization application for Dupixent® as an add-on maintenance treatment for inadequately controlled moderate-to-severe asthma in certain adults and adolescents. The FDA has also accepted for review a supplemental Biologics License Application for Dupixent® in the same indication. In accordance with the Prescription Drug User Fee Act, the FDA is expected to announce its decision on October 20, 2018.
In April 2018, the EMA accepted for review Sanofi’s marketing authorization application for cemiplimab in the treatment of metastatic cutaneous squamous cell carcinoma (CSCC) or patients with locally advanced CSCC who are not candidates for surgery. Also in April 2018, the FDA accepted for priority review a Biologics License Application for cemiplimab in the same indication; a decision is expected in October 2018. Advanced CSCC is the deadliest non-melanoma skin cancer. Cemiplimab is an investigational human monoclonal antibody targeting the PD-1 (programmed cell death 1) immune checkpoint inhibitor. It was granted Breakthrough Therapy designation status by the FDA in September 2017. Cemiplimab is being jointly developed by Sanofi and Regeneron under a global collaboration agreement. Cemiplimab is also undergoing clinical trials in the treatment of non small cell lung cancer, basal cell carcinoma and cervical cancer, and investigational studies are ongoing in the treatment of head and neck squamous cell carcinoma, melanoma, colorectal cancer, prostate cancer, and Hodgkin’s and non-Hodgkin’s lymphoma.
In June 2018, the EMA’s Committee for Medicinal Products for Human Use (CHMP) recommended approval of CabliviTM (caplacizumab) in Europe for the treatment of adults experiencing an episode of acquired thrombotic thrombocytopenic purpura (aTTP), a rare blood-clotting disorder. The European Commission will review the CHMP recommendation, and a final decision on the marketing authorization application for CabliviTM in the European Union is expected in the coming months. Directed against von Willebrand Factor (vWF), CabliviTM is Ablynx’s first Nanobody®-based medicine to receive a positive CHMP opinion and, if approved, will be the first therapeutic specifically indicated for the treatment of aTTP.
| 1 | Enterprise value of €1,919 million. |
52 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
In March 2018, the results of the ODYSSEY OUTCOMES trial were presented to the American College of Cardiology. The trial met its primary endpoint, showing that Praluent® (alirocumab, developed in collaboration with Regeneron) significantly reduced the risk of major adverse cardiovascular events in patients who had suffered a recent acute coronary syndrome event. In June 2018, the results of this trial were submitted to the EMA and the FDA. To help ensure more affordable and timely access to Praluent® for patients most in need, Sanofi and Regeneron will offer a further reduced net price to US payers that agree to reduce access barriers for high-risk patients. That net price will be in alignment with a new cost-effectiveness analysis for high-risk patients from the US Institute for Clinical and Economic Review (ICER). In keeping with ICER’s established “in confidence” procedures, Sanofi and Regeneron provided early access to data from the ODYSSEY OUTCOMES trial to ICER – an independent organization that evaluates the value of prescription drugs and other healthcare innovations – to enable a revised assessment of the value of alirocumab incorporating the results from the trial. A Phase III study evaluating Praluent® in children with heterozygous familial hypercholesterolemia (HeFH) has also been initiated.
A.1.3. RESEARCH AND DEVELOPMENT
For an update on our research and development (R&D) pipeline, refer to the appendix in Section F of this half-year management report.
The principal R&D announcements during the first half of 2018 were as follows:
Phase III:
| • | On May 21, 2018, the New England Journal of Medicine (NEJM) published detailed results from two Phase III clinical trials for the investigational use of Dupixent® (dupilumab) in the treatment of moderate-to-severe asthma. The results showed that Dupixent® significantly reduced the risk of severe asthma attacks (exacerbations), improved lung function and reduced dependence on oral corticosteroids. The trials, known as QUEST and VENTURE, are part of the pivotal clinical trial program that evaluated Dupixent® in uncontrolled asthma patients. The data were simultaneously presented at the American Thoracic Society 2018 International Conference. |
| • | In May 2018, a pivotal Phase III trial evaluating Dupixent® (dupilumab) in the treatment of moderate-to-severe atopic dermatitis in adolescents aged 12 to 17 years met its primary endpoint, and a number of key secondary endpoints. In the trial, treatment with Dupixent® as monotherapy significantly improved measures of overall disease severity and also improved skin clearing, itching, and certain health-related quality of life measures. Dupixent® is the first and only biologic to show positive results in this patient population. |
| • | The ATLAS Phase III program evaluating fitusiran resumed in the first quarter of 2018. The ATLAS-INH study is evaluating fitusiran in adult and adolescent hemophilia A or B patients with inhibitors. |
| • | Mavacamten (SAR439152/MYK461, developed in collaboration with MyoKardia), a myosin inhibitor, entered Phase III in the treatment of obstructive hypertrophic cardiomyopathy (HCM). A Phase II trial in non-HCM patients has also begun. |
| • | A Phase III trial has begun to evaluate Cerdelga® in children with Gaucher disease type 1 as a substitute for enzyme replacement therapy (ERT). |
| • | Sotagliflozine (developed in collaboration with Lexicon) has moved into Phase III in diabetic patients with worsening heart failure. |
| • | A Phase II/III study evaluating venglustat, an orally administered GCS inhibitor, is under preparation in patients at risk of autosomal dominant polycystic kidney disease. |
| • | AMPLITUDE, a Phase III cardiovascular morbidity/mortality study on efpeglenatide, has been initiated. |
| • | In June 2018, positive non-inferiority results from the BRIGHT study, comparing Toujeo® to insulin degludec were presented to the American Diabetes Association (ADA). |
Phase II:
| • | ALX0171, an anti RSV Nanobody®, entered the pipeline in Phase II following our acquisition of Ablynx. |
| • | Phase I/II data for BIVV001, a long-acting Factor VIII, demonstrating a half-life of 37 hours, were presented to the World Federation of Hemophilia (WFH). |
| • | Development was halted on two programs: SAR566658 (a maytansine-loaded anti-CA6 monoclonal antibody) in triple negative breast cancer, and a recombinant tuberculosis vaccine. |
| • | ST400, a genome editing technology derived from a collaboration between Sangamo and Ablynx, entered Phase II in beta thalassemia. |
| • | SAR440340, an anti-IL33 monoclonal antibody (partnership with Regeneron), is in the process of being initiated in a Phase II study in chronic obstructive pulmonary disease (COPD). |
| • | A Phase II study evaluating dupilumab in grass immunotherapy has been initiated. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 53
A.2. CONSUMER HEALTHCARE
At the end of June 2008, we sold a portfolio of 12 Sanofi Consumer Healthcare brands to Cooper-Vemedia, the European over-the-counter drug manufacturing and distribution subsidiary of Charterhouse Capital Partners, for €158 million. The sale sharpens the focus of Sanofi Consumer Healthcare on its four strategic fields: Pain; Allergy, Cough & Cold; Digestive; and Nutritionals.
A.3. HUMAN VACCINES (VACCINES) SEGMENT
In April 2018, Sanofi announced an investment of €350 million (CAD 500 million) in the construction of a new state-of-the-art vaccine manufacturing facility at the Sanofi Pasteur Canadian headquarters in Toronto, Ontario. The new facility will allow Sanofi Pasteur, Sanofi’s vaccines global business unit, to meet the growing demand for five-component acellular pertussis (5-acP) antigen. Upon completion in 2021, the new building will also be equipped to produce the antigens used in the manufacture of diphtheria and tetanus vaccines.
A.4. OTHER SIGNIFICANT EVENTS OF THE FIRST HALF OF 2018
A.4.1. CORPORATE GOVERNANCE
On January 19, 2018, Sanofi announced the appointment of Dominique Carouge as Executive Vice President Head of Business Transformation, effective February 15, 2018. In his new role, Mr Carouge is tasked with accelerating the transformation of Sanofi, and joins the Executive Committee. Previously, he served as Deputy CFO and Head of Finance Operations and Group Controlling from January 1, 2016.
Dr Elias Zerhouni, Head of Global Research and Development, retired from Sanofi on June 30, 2018, after more than nine years of distinguished service with the company. Dr John C. Reed, M.D., Ph.D., succeeded him effective July 1, 2018.
The Annual General Meeting of Sanofi shareholders was held in Paris on May 2, 2018. All of the resolutions submitted to the vote were adopted by the shareholders. The meeting approved a cash dividend of €3.03 per share, payable on May 15, 2018. It also approved the reappointment of Olivier Brandicourt, Patrick Kron and Christian Mulliez as directors, and appointed Emmanuel Babeau as an independent director, to serve for a four-year term expiring at the Annual General Meeting called to approve the 2021 financial statements. Robert Castaigne, who had been a Board member since 2000 and chaired the Audit Committee, did not seek re-election. Fabienne Lecorvaisier succeeded him as chair of the Audit Committee, which now comprises Fabienne Lecorvaisier, Emmanuel Babeau, Christian Mulliez and Diane Souza. The Board of Directors also announced the formation of a new Scientific Committee, chaired by Thomas Südhof. Following the meeting, the new Board of Directors still has 16 members, including six women and two employee representative directors. A substantial majority of the directors are independent.
In June 2018, Sanofi appointed Jean-Baptiste Chasseloup de Chatillon as Executive Vice President, Chief Financial Officer, and as a member of the Executive Committee. His appointment takes effect on October 1, 2018. He will join Sanofi on September 1 in order to ensure a smooth handover from Jérôme Contamine, who will retire on September 30 after more than nine years of distinguished service with the company.
A.4.2. LEGAL AND ARBITRATION PROCEEDINGS
For a description of the most significant developments in legal and arbitration proceedings since publication of the financial statements for the year ended December 31, 2017, refer to Note B.14. to the condensed half-year consolidated financial statements.
The following events have occurred in respect of litigation, arbitration and other legal proceedings in which Sanofi and its affiliates are involved:
PATENTS
| • | Co-Aprovel® Patent Infringement Actions (Europe) |
In Europe, pending damages claims in front of courts have been settled.
54 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
| • | Lantus® Merck Patent Litigation in the US |
In the patent infringement suit related to Lantus® filed by Sanofi against Merck Sharp and Dohme (“Merck”) in the United States District Court for the District of Delaware following Merck’s NDA filed for an insulin glargine drug pen product, a bench trial took place in the second quarter of 2018. A trial ruling is currently expected.
GOVERNMENT INVESTIGATIONS AND RELATED LITIGATION
In March 2018, Sanofi filed a motion to dismiss plaintiffs’ second amended complaint in the putative class actions filed against Sanofi US and Sanofi GmbH in Federal Court in Massachusetts on behalf of direct-purchasers of Lantus® alleging certain antitrust violations.
In May 2018, Sanofi US filed a joint motion to dismiss the consolidated amended complaint filed on behalf of a putative class of diabetes patients alleging violations of the Racketeer Influenced and Corrupt Organizations Act and various state unfair/deceptive trade practices statutes in connection with the pricing of Lantus®.
A.4.3. OTHER EVENTS
On March 15, 2018, Sanofi announced that it had successfully placed an €8 billion bond issue in six tranches under the Euro Medium Term Note program. This issue reduces the average cost and extends the average maturity of Sanofi’s debt.
In June 2018, Sanofi successfully placed a $2 billion bond issue in two tranches under the public bond issue program (shelf registration statement) registered with the US Securities and Exchange Commission (SEC) on March 15, 2016.
Sanofi intends to use the net proceeds of those bond issues for general corporate purposes, including the financing of the Bioverativ and Ablynx acquisitions.
B/ EVENTS SUBSEQUENT TO JUNE 30, 2018
| • | On July 26, 2018, Sanofi announced that Roberto Pucci, Head of Human Resources, will retire from Sanofi after more than 9 years of service with the company. He will be succeeded in the post by Caroline Luscombe who will join Sanofi on October 1, 2018. |
| • | On July 27, 2018, 2,401,184 shares (representing approximately 0.19% of our share capital) were issued in connection with the Action 2018 Sanofi worldwide employee share ownership plan, intended to give our employees a greater stake in the future development and results of Sanofi. From June 11 through June 29, 2018, 27,680 employees signed up for the plan, subscribing for Sanofi shares at a price of €52.66 per share. For each tranche of five shares applied for, the applicant received one additional new share by way of employer’s contribution; and for each application for 20 or more shares, the applicant received an additional four new shares by way of employer’s contribution. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 55
C/ CONSOLIDATED FINANCIAL STATEMENTS FOR THE FIRST HALF OF 2018
Unless otherwise indicated, all financial data in this report are presented in accordance with international financial reporting standards (IFRS), including international accounting standards and interpretations (see Note A.1. to the condensed half-year consolidated financial statements). Comparatives for 2017 have been restated in accordance with the new standard on revenue recognition, IFRS 15, which is applicable from January 1, 2018 onwards. The impacts of those restatements are described in detail in Note A.1.2. to the condensed half-year consolidated financial statements.
Consolidated income statements for the six months ended June 30, 2017 and June 30, 2018
| (€ million) | June 30, 2018 (6 months) |
as % of net sales |
June 30, 2017 (6 months)(a) |
as % of net sales |
||||||||||||
| Net sales |
16,074 | 100.0 | % | 17,324 | 100.0 | % | ||||||||||
| Other revenues |
533 | 3.3 | % | 519 | 3.0 | % | ||||||||||
| Cost of sales |
(5,265 | ) | (32.8 | %) | (5,671 | ) | (32.7 | %) | ||||||||
| Gross profit |
11,342 | 70.6 | % | 12,172 | 70.3 | % | ||||||||||
| Research and development expenses |
(2,755 | ) | (17.1 | %) | (2,667 | ) | (15.4 | %) | ||||||||
| Selling and general expenses |
(4,819 | ) | (30.0 | %) | (5,054 | ) | (29.2 | %) | ||||||||
| Other operating income |
323 | 173 | ||||||||||||||
| Other operating expenses |
(165 | ) | (71 | ) | ||||||||||||
| Amortization of intangible assets |
(999 | ) | (990 | ) | ||||||||||||
| Impairment of intangible assets |
(101 | ) | (12 | ) | ||||||||||||
| Fair value remeasurement of contingent consideration |
10 | (100 | ) | |||||||||||||
| Restructuring costs and similar items |
(607 | ) | (364 | ) | ||||||||||||
| Other gains and losses, and litigation |
(67 | ) | (7 | ) | ||||||||||||
| Operating income |
2,162 | 13.5 | % | 3,080 | 17.8 | % | ||||||||||
| Financial expenses |
(202 | ) | (218 | ) | ||||||||||||
| Financial income |
97 | 95 | ||||||||||||||
| Income before tax and investments accounted for using the equity method |
2,057 | 12.8 | % | 2,957 | 17.1 | % | ||||||||||
| Income tax expense |
(297 | ) | (612 | ) | ||||||||||||
| Share of profit/(loss) of investments accounted for using the equity method |
75 | 27 | ||||||||||||||
| Net income excluding the exchanged/held-for-exchange Animal Health business |
1,835 | 11.4 | % | 2,372 | 13.7 | % | ||||||||||
| Net income/(loss) of the exchanged/held-for-exchange Animal Health business(b) |
— | 4,421 | ||||||||||||||
| Net income |
1,835 | 6,793 | ||||||||||||||
| Net income attributable to non-controlling interests |
57 | 64 | ||||||||||||||
| Net income |
||||||||||||||||
| Attributable to equity holders of Sanofi |
1,778 | 11.1 | % | 6,729 | 38.8 | % | ||||||||||
| Average number of shares outstanding (million) |
1,247.8 | 1,260.3 | ||||||||||||||
| Average number of shares after dilution (million) |
1,254.9 | 1,270.6 | ||||||||||||||
| – Basic earnings per share (in euros) |
1.42 | 5.34 | ||||||||||||||
| – Basic earnings per share excluding the exchanged/held-for-exchange Animal Health business (in euros) |
1.42 | 1.83 | ||||||||||||||
| – Diluted earnings per share (in euros) |
1.42 | 5.30 | ||||||||||||||
| – Diluted earnings per share excluding the exchanged/held-for-exchange Animal Health business (in euros) |
1.42 | 1.82 | ||||||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
| (b) | For 2017, the gain on the divestment of the Animal Health business is presented separately in accordance with IFRS 5 (Non-Current Assets Held for Sale and Discontinued Operations); see Note D.36 to the consolidated financial statements for the year ended December 31, 2017. |
56 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.1. SEGMENT INFORMATION
C.1.1. OPERATING SEGMENTS
In accordance with IFRS 8 (Operating Segments), the segment information reported by Sanofi is prepared on the basis of internal management data provided to the Chief Executive Officer, who is the chief operating decision maker. The performance of those segments is monitored individually using internal reports and common indicators. The operating segment disclosures required under IFRS 8 are also provided in Note B.20. to the condensed half-year consolidated financial statements.
With effect from December 31, 2017 Sanofi has three operating segments: Pharmaceuticals, Consumer Healthcare and Human Vaccines (Vaccines).
The Pharmaceuticals segment comprises the commercial operations of the following global franchises: Specialty Care (Rare Diseases, Multiple Sclerosis, Oncology, Immunology), Diabetes & Cardiovascular, Established Prescription Products and Generics, together with research, development and production activities dedicated to our Pharmaceuticals segment. This segment also includes all associates whose activities are related to pharmaceuticals, in particular the share of profits from Regeneron.
The Consumer Healthcare segment comprises, for all geographical territories, the commercial operations for our Consumer Healthcare products, together with research, development and production activities dedicated to those products.
The Vaccines segment comprises, for all geographical territories (including from January 1, 2017 certain European territories previously included in the Sanofi Pasteur MSD joint venture), the commercial operations of Sanofi Pasteur, together with research, development and production activities dedicated to vaccines.
Inter-segment transactions are not material.
The costs of Sanofi’s global functions (Medical Affairs, External Affairs, Finance, Human Resources, Legal Affairs, Information Solutions & Technologies, Sanofi Business Services, etc.) are managed centrally at group-wide level. For the year ended December 31, 2017 and subsequent years, the costs of those functions are presented within the “Other” category. That category also includes other reconciling items such as retained commitments in respect of divested activities.
Consequently, the presentation of 2017 comparatives shown below, and the associated performance analysis, have been adjusted to reflect the new segmental reporting model.
C.1.2. BUSINESS OPERATING INCOME
Sanofi reports segment results on the basis of “business operating income”. This indicator is used internally by Sanofi’s chief operating decision maker to measure the performance of each operating segment and to allocate resources. For a definition of “business operating income”, and a reconciliation between that indicator and Income before tax and investments accounted for using the equity method, refer to Note B.20.1 to our condensed half-year consolidated financial statements.
C.2. BUSINESS NET INCOME
We believe that understanding of our operational performance by our management and our investors is enhanced by reporting “business net income”. This non-GAAP financial measure represents business operating income, less net financial expenses and the relevant income tax effects.
Business net income for the first half of 2018 was €3,156 million, 9.4% lower than in the first half of 2017 (€3,482 million1). That represents 19.6% of net sales, compared with 20.1% in the first half of 2017.
We also report “business earnings per share”, a non-GAAP financial measure which we define as business net income divided by the weighted average number of shares outstanding.
Business earnings per share was €2.53 for the first half of 2018, 8.3% lower than the 2017 first-half figure of €2.76, based on an average number of shares outstanding of 1,247.8 million for the first half of 2018 and 1,260.3 million for the first half of 2017.
| 1 | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 57
The table below reconciles our business operating income to our business net income:
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
December 31, 2017 (12 months)(a) |
|||||||||
| Business operating income |
4,126 | 4,734 | 9,323 | |||||||||
| Net financial expenses |
(105 | ) | (123 | ) | (273 | ) | ||||||
| Income tax expense |
(865 | ) | (1,129 | ) | (2,107 | ) | ||||||
| Business net income |
3,156 | 3,482 | 6,943 | |||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
We define business net income as Net income attributable to equity holders of Sanofi determined under IFRS, excluding the following items:
| • | amortization and impairment losses charged against intangible assets (other than software and other rights of an industrial or operational nature); |
| • | fair value remeasurements of contingent consideration relating to business combinations or divestments; |
| • | other impacts associated with acquisitions (including impacts of acquisitions on investments accounted for using the equity method); |
| • | restructuring costs and similar items1; |
| • | other gains and losses (including gains and losses on major disposals of non-current assets2); |
| • | other costs and provisions related to litigation2; |
| • | the tax effects of the items listed above; |
| • | the effects of major tax disputes; |
| • | the 3% tax levied on the distribution of dividends to equity holders of Sanofi in 2017; |
| • | the direct and indirect effects of the US tax reform enacted on December 22, 2017, and the consequences of the French Constitutional Council ruling of October 6, 2017 on the additional 3% levy on dividends paid out in cash; |
| • | those Animal Health items that are not included in business net income3; |
| • | the portion attributable to non-controlling interests of the items listed above. |
| 1 | Presented in the line item Restructuring costs and similar expenses in the consolidated income statement. |
| 2 | Presented in the line item Other gains and losses, and litigation in the consolidated income statement. |
| 3 | Comprises (i) impact of the discontinuation of depreciation and impairment of property, plant & equipment with effect from the start date of application of IFRS 5 (Discontinued and Held-for-Sale Operations) included in business net income; (ii) impact of the amortization and impairment of intangible assets until the start date of IFRS 5 application; (iii) costs directly incurred as a result of the divestment; and (iv) tax effects of those items. |
58 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
The table below reconciles our business net income to Net income attributable to equity holders of Sanofi:
| (€ million) | June 30, (6 months) |
June 30,(a) (6 months) |
December 31, (a) (12 months) |
|||||||||
| Net income attributable to equity holders of Sanofi |
1,778 | 6,729 | 8,416 | |||||||||
| Amortization of intangible assets (b) |
999 | 990 | 1,866 | |||||||||
| Impairment of intangible assets |
101 | 12 | 293 | |||||||||
| Fair value remeasurement of contingent consideration |
(10 | ) | 100 | 159 | ||||||||
| Expenses arising from the impact of acquisitions on inventories |
99 | 176 | 166 | |||||||||
| Other expenses related to business combinations |
10 | — | — | |||||||||
| Restructuring costs and similar items |
607 | 364 | 731 | |||||||||
| Other gains and losses, and litigation(c) |
67 | 7 | 215 | |||||||||
| Tax effects of the items listed above: |
(475 | ) | (628 | ) | (1,126 | ) | ||||||
| - tax effects of amortization and impairment of intangible assets |
(275 | ) | (349 | ) | (719 | ) | ||||||
| - tax effects of fair value remeasurement of contingent consideration |
11 | (31 | ) | 4 | ||||||||
| - tax effects of expenses arising from the impact of acquisitions on inventories |
(23 | ) | (56 | ) | (52 | ) | ||||||
| - tax effects of other expenses related to business combinations |
— | — | — | |||||||||
| - tax effects of restructuring costs and similar items |
(183 | ) | (126 | ) | (134 | ) | ||||||
| - other tax effects |
(5 | ) | (66 | ) | (225 | ) | ||||||
| Other tax items (d) |
(93 | ) | 111 | 741 | ||||||||
| Share of items listed above attributable to non-controlling interests |
(1 | ) | (1 | ) | (4 | ) | ||||||
| Investments accounted for using the equity method: restructuring costs and expenses arising from the impact of acquisitions |
74 | 43 | 129 | |||||||||
| Items relating to the Animal Health business (e) |
— | (4,421 | ) | (4,643 | ) | |||||||
| Business net income |
3,156 | 3,482 | 6,943 | |||||||||
| Average number of shares outstanding (million) |
1,247.8 | 1260.3 | 1,256.9 | |||||||||
| Basic earnings per share (in euros) |
1.42 | 5.34 | 6.70 | |||||||||
| Reconciling items per share (in euros) |
1.11 | (2.58 | ) | (1.18 | ) | |||||||
| Business earnings per share (in euros) |
2.53 | 2.76 | 5.52 | |||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
| (b) | Includes amortization expense generated by the remeasurement of intangible assets in connection with business combinations: €934 million in the six months ended June 30, 2018; €919 million in the six months ended June 30, 2017, and €1,726 million in the year ended December 31, 2017. |
| (c) | For 2018, this line consists mainly of separation costs associated with the process of disinvesting from the Generics business in Europe, before any tax effects. For the year ended December 31, 2017, this line item also includes an additional charge to a provision for a vendor’s liability guarantee on a past divestment. |
| (d) | In 2018, this line includes adjustments to our preliminary analysis of the direct and indirect impacts of US tax reform. In 2017, it shows the impact of the 3% tax on dividends in France. |
| (e) | This line includes the gain on the divestment of the Animal Health business, which is presented separately in accordance with IFRS 5 (Non-Current Assets Held for Sale and Discontinued Operations), along with the residual impacts of that divestment. |
The most significant reconciling items between our business net income and Net income attributable to equity holders of Sanofi relate to (i) the purchase accounting effects of our acquisitions and business combinations, particularly the amortization and impairment of intangible assets (other than software and other rights of an industrial or operational nature) and (ii)) the impacts of events regarded as non-recurring, where the amounts involved are particularly significant. . We believe that excluding those non-cash or non-recurring charges enhances an investor’s understanding of our underlying economic performance, because we do not consider that the excluded charges reflect the combined entity’s ongoing operating performance. Rather, we believe that each of the excluded charges reflects the decision to acquire the businesses concerned.
The principal purchase accounting effects of acquisitions and business combinations on net income are:
| • | amortization and net impairment losses charged against intangible assets (other than software and other rights of an industrial or operational nature), net of taxes and non-controlling interests; and |
| • | the incremental cost of sales incurred on the workdown of acquired inventories remeasured at fair value, net of taxes. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 59
We believe (subject to the limitations described below) that disclosing our business net income enhances the comparability of our operating performance, for the following reasons:
| • | the elimination of charges related to the purchase accounting effect of our acquisitions and business combinations (particularly amortization and impairment of finite-lived intangible assets, other than software and other rights of an industrial or operational nature) enhances the comparability of our ongoing operating performance relative to our peers in the pharmaceutical industry that carry those intangible assets (principally patents and trademarks) at low book values either because they are the result of in-house research and development that has already been expensed in prior periods or because they were acquired through business combinations that were accounted for as poolings-of-interest; |
| • | the elimination of selected items – such as the incremental cost of sales arising from the workdown of acquired inventories remeasured at fair value in business combinations, major gains and losses on disposals, and costs and provisions associated with major litigation and any other major non-recurring items, improves comparability from one period to the next; and |
| • | the elimination of restructuring costs and similar items enhances comparability because those costs are incurred in connection with reorganization and transformation processes intended to optimize our operations. |
We remind investors, however, that business net income should not be considered in isolation from, or as a substitute for, Net income attributable to equity holders of Sanofi reported in accordance with IFRS. In addition, we strongly encourage investors and potential investors not to rely on any single financial measure but to review our financial statements, including the notes thereto, carefully and in their entirety.
We compensate for the material limitations described above by using business net income only to supplement our IFRS financial reporting and by ensuring that our disclosures provide sufficient information for a full understanding of all adjustments included in business net income.
Because our business net income is not a standardized measure, it may not be directly comparable with the non-GAAP financial measures of other companies using the same or a similar non-GAAP financial measure.
60 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.3. NET SALES
Comparatives for 2017 have been restated in accordance with the new standard on revenue recognition, IFRS 15, which is applicable from January 1, 2018 onwards. The impacts of those restatements are described in detail in Note A.1.2. to the condensed half-year consolidated financial statements.
Consolidated net sales for the first half of 2018 amounted to €16,074 million, 7.2% lower than in the first half of 2017. Exchange rate fluctuations had a negative effect of 7.1 percentage points overall, due mainly to unfavorable trends in the exchange rate for the euro against the US dollar, Brazilian real and Argentinean peso. At constant exchange rates (CER, see definition below), net sales were virtually unchanged (-0.1% year-on-year, with lower sales for the Diabetes franchise and Established Prescription Products offset by a good performance from the Immunology franchise and by sales of Bioverativ products for rare blood disorders, consolidated from March 8, 2018 onwards. At constant exchange rates and on a constant structure basis (CER/CS, see definition below), net sales were down 1.8%.
Reconciliation of net sales to net sales at constant exchange rates and on a constant structure basis
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
Change | |||||||||
| Net sales |
16,074 | 17,324 | -7.2 | % | ||||||||
| Effect of exchange rates |
1,227 | |||||||||||
| Net sales at constant exchange rates |
17,301 | 17,324 | -0.1 | % | ||||||||
| Impact of change in structure (Bioverativ)(b) |
303 | |||||||||||
| Net sales at constant exchange rates and on a constant structure basis |
17,301 | 17,627 | -1.8 | % | ||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
| (b) | Net sales of Bioverativ products (consolidated from March 8, 2018) for the period from March 9, 2017 through June 30, 2017. |
When we refer to changes in our net sales at constant exchange rates (CER), that means that we have excluded the effect of exchange rates by recalculating net sales for the relevant period using the exchange rates that were used for the previous period.
When we refer to changes in our net sales on a constant structure basis, that means that we eliminate the effect of changes in structure by restating the net sales for the previous period as follows:
| • | by including sales generated by entities or product rights acquired in the current period for a portion of the previous period equal to the portion of the current period during which we owned them, based on sales information we receive from the party from whom we make the acquisition; |
| • | similarly, by excluding sales for a portion of the previous period when we have sold an entity or rights to a product in the current period; and |
| • | for a change in consolidation method, by recalculating the previous period on the basis of the method used for the current period. |
To facilitate analysis and comparisons with prior periods, some figures are given at constant exchange rates and on a constant structure basis (CER/CS).
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 61
C.3.1. NET SALES BY OPERATING SEGMENT
Our net sales comprise the net sales generated by our Pharmaceuticals, Consumer Healthcare and Human Vaccines (Vaccines) segments. Following the gradual integration of BI’s Consumer Healthcare business, acquired on January 1, 2017, our Consumer Healthcare business represents a separate operating segment of Sanofi in accordance with IFRS 8. Consequently, we present our Consumer Healthcare net sales separately for the periods ended June 30, 2017 and June 30, 2018.
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
Change | |||||||||
| Pharmaceuticals |
12,199 | 13,038 | -6.4 | % | ||||||||
| Consumer Healthcare |
2,353 | 2,486 | -5.3 | % | ||||||||
| Vaccines |
1,522 | 1,800 | -15.4 | % | ||||||||
| Net sales |
16,074 | 17,324 | -7.2 | % | ||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
C.3.2. NET SALES BY GLOBAL BUSINESS UNIT (GBU)
The table below presents net sales for our Global Business Units (GBUs). Note that Emerging Markets sales of Diabetes & Cardiovascular and Specialty Care products are included in the General Medicines & Emerging Markets GBU.
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
Change on a reported basis |
Change at constant exchange rates |
||||||||||||
| Sanofi Genzyme GBU (Specialty Care)(b)(c) |
3,268 | 2,817 | +16.0 | % | +24.8 | % | ||||||||||
| Diabetes & Cardiovascular GBU(b) |
2,195 | 2,801 | -21.6 | % | -15.6 | % | ||||||||||
| General Medicines & Emerging Markets GBU(d)(e) |
6,736 | 7,420 | -9.2 | % | -2.6 | % | ||||||||||
| Total Pharmaceuticals |
12,199 | 13,038 | -6.4 | % | +0.5 | % | ||||||||||
| Consumer Healthcare GBU |
2,353 | 2,486 | -5.3 | % | +3.0 | % | ||||||||||
| Sanofi Pasteur (Vaccines) GBU |
1,522 | 1,800 | -15.4 | % | -9.3 | % | ||||||||||
| Total net sales |
16,074 | 17,324 | -7.2 | % | -0.1 | % | ||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
| (b) | Does not include Emerging Markets net sales. |
| (c) | Rare Diseases, Multiple Sclerosis, Oncology and Immunology, and Rare Blood Disorder. |
| (d) | Includes net sales in Emerging Markets of Specialty Care and Diabetes & Cardiovascular products. |
| (e) | Emerging Markets: World excluding United States, Canada, Western and Eastern Europe (apart from Russia, Ukraine, Georgia, Belarus, Armenia and Turkey), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
62 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.3.3. NET SALES BY FRANCHISE
The table below sets forth our 2018 first-half net sales by franchise in order to facilitate direct comparisons with our peers. For a detailed reconciliation of net sales by franchise and net sales by GBU for our Pharmaceuticals segment, refer to the table later in this report showing Pharmaceuticals segment net sales by geographical region.
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
Change on a reported basis |
Change at exchange rates |
||||||||||||
| Rare Diseases |
1,438 | 1,466 | -1.9 | % | +6.5 | % | ||||||||||
| Multiple Sclerosis |
982 | 1,045 | -6.0 | % | +2.0 | % | ||||||||||
| Oncology |
727 | 794 | -8.4 | % | -1.8 | % | ||||||||||
| Immunology |
313 | 27 | +1,059.3 | % | +1,170.4 | % | ||||||||||
| Rare Blood Disorder |
321 | — | — | — | ||||||||||||
| Total Specialty Care |
3,781 | 3,332 | +13.5 | % | +23.1 | % | ||||||||||
| - of which Developed Markets (Sanofi Genzyme GBU) |
3,268 | 2,817 | +16.0 | % | +24.8 | % | ||||||||||
| - of which Emerging Markets (b)(c) |
513 | 515 | -0.4 | % | +13.4 | % | ||||||||||
| Diabetes |
2,722 | 3,313 | -17.8 | % | -10.9 | % | ||||||||||
| Cardiovascular |
273 | 257 | +6.2 | % | +15.2 | % | ||||||||||
| Total Diabetes & Cardiovascular |
2,995 | 3,570 | -16.1 | % | -9.0 | % | ||||||||||
| - of which Developed Markets (Diabetes & Cardiovascular GBU) |
2,195 | 2,801 | -21.6 | % | -15.6 | % | ||||||||||
| - of which Emerging Markets (b)(c) |
800 | 769 | +4.0 | % | +15.0 | % | ||||||||||
| Established Prescription Products(b) |
4,586 | 5,230 | -12.3 | % | -7.1 | % | ||||||||||
| Generics(b) |
837 | 906 | -7.6 | % | -0.3 | % | ||||||||||
| Total Pharmaceuticals |
12,199 | 13,038 | -6.4 | % | +0.5 | % | ||||||||||
| Consumer Healthcare (Consumer Healthcare GBU) |
2,353 | 2,486 | -5.3 | % | +3.0 | % | ||||||||||
| Vaccines (Sanofi Pasteur GBU) |
1,522 | 1,800 | -15.4 | % | -9.3 | % | ||||||||||
| Total net sales |
16,074 | 17,324 | -7.2 | % | -0.1 | % | ||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
| (b) | These lines are aggregated to form the net sales of the General Medicines and Emerging Markets GBU. |
| (c) | Emerging Markets: World excluding United States, Canada, Western and Eastern Europe (apart from Russia, Ukraine, Georgia, Belarus, Armenia and Turkey), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
C.3.4. PHARMACEUTICALS SEGMENT
Net sales of the Pharmaceuticals segment (excluding Consumer Healthcare) were €12,199 million in the first half of 2018, down 6.4% on a reported basis but up 0.5% at constant exchange rates. At constant exchange rates and on a constant structure basis, net sales were down 1.7%.
The €839 million decrease in net sales compared with the first half of 2017 was mainly due to the €909 million negative effect of exchange rates. At constant exchange rates, the year-on-year change in net sales reflects the following impacts:
| • | positive performances from the Rare Blood Disorder franchise (+€351 million) reflecting the inclusion of Bioverativ products in consolidated net sales from March 8, 2018, the Immunology franchise (+€316 million), the Rare Diseases franchise (+€95 million); the Cardiovascular franchise (+€39 million), and the Multiple Sclerosis franchise (+€21 million); |
| • | lower net sales for the Established Prescription Products franchise (-€373 million), the Diabetes franchise (-€362 million), the Oncology franchise (-€14 million) and the Generics franchise (-€3 million). |
Comments on the performances of our major Pharmaceuticals segment products are provided below.
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 63
Net sales by product and franchise
| (€ million) | Indication | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
Change a reported basis |
Change at constant exchange |
|||||||||||||
| Cerezyme® |
Gaucher disease | 356 | 370 | -3.8 | % | +6.2 | % | |||||||||||
| Cerdelga® |
Gaucher disease | 74 | 62 | +19.4 | % | +29.0 | % | |||||||||||
| Myozyme®/Lumizyme® |
Pompe disease | 405 | 394 | +2.8 | % | +9.4 | % | |||||||||||
| Fabrazyme® |
Fabry disease | 358 | 367 | -2.5 | % | +6.5 | % | |||||||||||
| Aldurazyme® |
Mucopolysaccharidosis | 103 | 110 | -6.4 | % | +1.8 | % | |||||||||||
| Other |
142 | 163 | -12.9 | % | -5.5 | % | ||||||||||||
| Total Rare Diseases |
1,438 | 1,466 | -1.9 | % | +6.5 | % | ||||||||||||
| Aubagio® |
Multiple Sclerosis | 775 | 796 | -2.6 | % | +6.0 | % | |||||||||||
| Lemtrada® |
Multiple Sclerosis | 207 | 249 | -16.9 | % | -10.8 | % | |||||||||||
| Total Multiple Sclerosis |
982 | 1,045 | -6.0 | % | +2.0 | % | ||||||||||||
| Jevtana® |
Prostate cancer | 202 | 197 | +2.5 | % | +10.2 | % | |||||||||||
| Thymoglobulin® |
Organ rejection | 144 | 148 | -2.7 | % | +6.1 | % | |||||||||||
| Eloxatin® |
Colorectal cancer | 90 | 90 | +0.0 | % | +5.6 | % | |||||||||||
| Taxotere® |
Breast, lung, prostate, stomach, and head & neck cancers | 84 | 90 | -6.7 | % | -1.1 | % | |||||||||||
| Mozobil® |
Hematologic malignancies | 82 | 80 | +2.5 | % | +10.0 | % | |||||||||||
| Zaltrap® |
Colorectal cancer | 46 | 34 | +35.3 | % | +41.2 | % | |||||||||||
| Other |
79 | 155 | -49.0 | % | -44.5 | % | ||||||||||||
| Total Oncology |
727 | 794 | -8.4 | % | -1.8 | % | ||||||||||||
| Eloctate® |
Hemophilia A | 219 | — | — | — | |||||||||||||
| Alprolix® |
Hemophilia B | 102 | — | — | — | |||||||||||||
| Total Rare Blood Disorder |
321 | — | — | — | ||||||||||||||
| Dupixent® |
Atopic dermatitis | 283 | 26 | +988.5 | % | +1,092.3 | % | |||||||||||
| Kevzara® |
Rheumatoid arthritis | 30 | 1 | +2,900.0 | % | +3,200.0 | % | |||||||||||
| Total Immunology |
313 | 27 | +1,059.3 | % | +1,170.4 | % | ||||||||||||
| Total Specialty Care |
3,781 | 3,332 | +13.5 | % | +23.1 | % | ||||||||||||
| Lantus® |
Diabetes | 1,802 | 2,425 | -25.7 | % | -19.1 | % | |||||||||||
| Toujeo® |
Diabetes | 414 | 403 | +2.7 | % | +10.7 | % | |||||||||||
| Apidra® |
Diabetes | 183 | 191 | -4.2 | % | +3.1 | % | |||||||||||
| Amaryl® |
Diabetes | 170 | 173 | -1.7 | % | +5.8 | % | |||||||||||
| Insuman® |
Diabetes | 47 | 56 | -16.1 | % | -12.5 | % | |||||||||||
| Lyxumia® |
Diabetes | 11 | 14 | -21.4 | % | -14.3 | % | |||||||||||
| Soliqua® 100/33 / SuliquaTM |
Diabetes | 26 | 9 | +188.9 | % | +222.2 | % | |||||||||||
| Other |
Diabetes | 69 | 42 | +64.3 | % | +73.8 | % | |||||||||||
| Total Diabetes |
2,722 | 3,313 | -17.8 | % | -10.9 | % | ||||||||||||
| Multaq® |
Atrial fibrillation | 162 | 181 | -10.5 | % | -1.7 | % | |||||||||||
| Praluent® |
Hypercholesterolemia | 111 | 76 | +46.1 | % | +55.3 | % | |||||||||||
| Total Cardiovascular |
273 | 257 | +6.2 | % | +15.2 | % | ||||||||||||
| Total Diabetes & Cardiovascular |
2,995 | 3,570 | -16.1 | % | -9.0 | % | ||||||||||||
| Lovenox® |
Thrombosis | 768 | 817 | -6.0 | % | -1.5 | % | |||||||||||
| Plavix® |
Atherothrombosis | 761 | 764 | -0.4 | % | +4.6 | % | |||||||||||
| Aprovel® / Avapro® |
Hypertension | 343 | 382 | -10.2 | % | -4.7 | % | |||||||||||
| Depakine® |
Epilepsy | 230 | 224 | +2.7 | % | +7.6 | % | |||||||||||
| Renagel®/Renvela® |
Hyperphosphatemia | 201 | 494 | -59.3 | % | -55.7 | % | |||||||||||
| Synvisc® / Synvisc-One® |
Arthritis | 160 | 206 | -22.3 | % | -14.6 | % | |||||||||||
| Allegra® |
Allergic rhinitis, urticaria | 80 | 102 | -21.6 | % | -14.7 | % | |||||||||||
| Stilnox®/Ambien®/Myslee® |
Sleep disorders | 116 | 137 | -15.3 | % | -8.0 | % | |||||||||||
| Tritace® |
Hypertension | 115 | 124 | -7.3 | % | -2.4 | % | |||||||||||
| Other |
1,812 | 1,980 | -8.5 | % | -3.1 | % | ||||||||||||
| Total Established Prescription Products |
4,586 | 5,230 | -12.3 | % | -7.1 | % | ||||||||||||
| Generics |
837 | 906 | -7.6 | % | -0.3 | % | ||||||||||||
| Total Pharmaceuticals |
12,199 | 13,038 | -6.4 | % | +0.5 | % | ||||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
64 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
RARE DISEASES FRANCHISE
In the first half of 2018, net sales for the Rare Diseases franchise were €1,438 million, down 1.9% on a reported basis but up 6.5% at constant exchange rates, reflecting stronger sales in all geographies but especially in Emerging Markets1 (+13.3% CER at €268 million), Europe2 (+6.8% CER at €502 million) and the United States (+3.5% CER at €509 million).
In the first half of 2018, net sales for the Gaucher disease franchise (Cerezyme® and Cerdelga®) reached €430 million, up 9.5% CER, on stronger sales of Cerezyme® in Emerging Markets (+25.0% CER at €120 million) and growing penetration of Cerdelga® in the United States (+10.4% CER at €47 million) and Europe (+100.0% CER at €22 million). In the first half of 2018, Cerezyme® posted net sales of €356 million (+6.2% CER), while net sales of Cerdelga® reached €74 million, up 29.0%.
Net sales of Myozyme® / Lumizyme® in Pompe disease rose by 9.4% CER in the first half of 2018 to €405 million, driven by sales growth in Europe (+11.8% CER, at €188 million) and in the United States (+9.6% CER, at €133 million). Sales also grew in Emerging Markets (+5.0% CER, at €56 million) and in the Rest of the World region3 (+3.3% CER at €28 million). This growth reflects the rising number of patients diagnosed with, and treated for, Pompe disease.
Fabrazyme® posted net sales growth of 6.5% CER to €358 million. Sales are advancing in many countries due to the rising number of patients diagnosed with, and treated for, Fabry disease. Net sales of the product advanced by 5.8% CER (to €179 million) in the United States and by 7.4% CER (to €87 million) in Europe.
MULTIPLE SCLEROSIS FRANCHISE
In the first half of 2018, the Multiple Sclerosis franchise generated net sales of €982 million, down 6.0% on a reported basis but up 2.0% CER, driven by growth for Aubagio®.
Aubagio® achieved net sales of €775 million in the first half of 2018, up 6.0% CER, supported by growth in the United States (+11.0% CER, at €541 million) and Emerging Markets (+52.6% CER, at €25 million). That more than offset lower sales in Europe (-9.8% CER, at €184 million), reflecting a tough comparative given that in the first half of 2017 Aubagio® was boosted by an order of nearly €30 million for use in clinical trials. In 2017, Sanofi reached settlement with all 20 generic Aubagio® Abbreviated New Drug Application (ANDA) first filers granting each a royalty-free license to enter the United States market on March 12, 2023.
In the first half of 2018, net sales of Lemtrada® were €207 million, down 10.8% CER on lower sales in the United States (-19.2% CER, at €93 million) due to increased competition and the need to rebuild the patient cohort in light of the product’s unique dosage profile and long-lasting effects. In Europe and Emerging Markets, net sales of Lemtrada® rose by 1.1% CER to €92 million and by 36.4% to €12 million, respectively.
ONCOLOGY FRANCHISE
In the first half of 2018, net sales for the Oncology franchise amounted to €727 million, down 8.4% on a reported basis and 1.8% CER. We divested Leukine® on January 31, 2018, as part of our portfolio refocusing strategy. Excluding Leukine®, Oncology franchise net sales increased by 4.3% CER in the first half, on strong performances from Jevtana® in the United States and from Jevtana® and Zaltrap® in the Rest of the World region.
Jevtana® reported net sales of €202 million in the first half of 2018, up 10.2% CER, mainly on sales growth in the United States (+16.0% CER, at €84 million), though sales were also stronger in Europe (+4.0% CER, at €78 million) and Japan (+21.7% CER, at €26 million).
In the first half of 2018, sales of Thymoglobulin® and Eloxatin® were boosted by China, where sales of the two products rose by 6.1% CER (to €144 million) and 5.6% CER (to €90 million), respectively.
Sales of Zaltrap® totaled €46 million in the first half of 2018, up 41.2% CER, driven by Japan (+900.0% CER, at €9 million).
| 1 | World excluding United States, Canada, Western and Eastern Europe (apart from Russia, Ukraine, Georgia, Belarus, Armenia and Turkey), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
| 2 | Western Europe and Eastern Europe excluding Eurasia (Russia, Ukraine, Georgia, Belarus, Armenia and Turkey). |
| 3 | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 65
IMMUNOLOGY FRANCHISE
Dupixent® (developed in collaboration with Regeneron) was launched in the United States in April 2017 for moderate-to-severe atopic dermatitis in adults; further launches followed in Germany (December 2017) and in the Netherlands, Canada, Denmark, Sweden and Japan during the first half of 2018. Net sales of Dupixent® reached €283 million in the first half of 2018, of which €246 million was generated in the United States.
Kevzara® (developed in collaboration with Regeneron) was launched as a rheumatoid arthritis treatment in the United States in June 2017; in Germany, the United Kingdom and the Netherlands in the second half of 2017; and in Japan, Belgium, Sweden and Finland in the first half of 2018. First-half sales of Kevzara® amounted to €30 million, of which €23 million was generated in the United States.
RARE BLOOD DISORDER FRANCHISE
We have completed the acquisition of Bioverativ, which is consolidated with effect from March 9, 2018. Consolidated net sales for the Rare Blood Disorder franchise in the period from that date to June 30, 2018 amounted to €321 million, including €51 million of non-US sales (mainly in Japan). At constant exchange rates and on a constant structure basis, the franchise grew sales by 15.8%. After Colombia in the first quarter of 2018, further launches in Emerging Markets countries are anticipated later this year.
Consolidated sales of Eloctate® (a recombinant fusion protein that fuses human coagulation factor VIII to the Fc portion of IgG1, indicated in the treatment of hemophilia A) amounted to €219 million. At constant exchange rates and on a constant structure basis, sales of Eloctate® rose by 20.6%.
Consolidated sales of Alprolix® (a recombinant fusion protein that fuses human coagulation factor IX to the Fc portion of IgG1, indicated in the treatment of hemophilia B) reached €102 million. At constant exchange rates and on a constant structure basis, sales of Alprolix® rose by 6.7%.
DIABETES FRANCHISE
Net sales for the Diabetes franchise amounted to €2,722 million in the first half of 2018, down 17.8% on a reported basis and 10.9% at constant exchange rates. The main factor was a decrease in sales of Lantus® in the United States, where Diabetes franchise sales were down 28.3% CER at €1,059 million. That decrease reflects the exclusion of a number of diabetes treatments from the reimbursement list of some leading private healthcare providers; changes to the Medicare Part D welfare program; and the ongoing decline in average net prices for insulin glargines in the United States. Elsewhere in the world, net sales for the Diabetes franchise rose in Emerging Markets (+14.7% CER, at €793 million); remained stable in Europe (-0.3% CER, at €648 million), where a good performance by Toujeo® partly offset lower sales of Lantus®; and decreased in the Rest of the World region (-1.6% CER, at €222 million).
In the first half of 2018, net sales of our insulin glargines (Lantus® and Toujeo®) were down 21.6% on a reported basis and down 14.9% CER, at €2,216 million.
Net sales of Lantus® decreased by 19.1% CER in the first half to €1,802 million. In the United States, sales were down 32.4% CER, at €816 million, for the reasons explained above. Net sales in Europe decreased by 9.4% CER to €355 million, due largely to the launch of a biosimilar of Lantus® and the switching of patients to Toujeo®. In Emerging Markets, sales of Lantus® advanced by 4.8% CER to €492 million.
In the first half of 2018, the new-generation basal insulin Toujeo® posted net sales of €414 million, up 10.7% CER, driven by strong performances in Europe (+42.0%, at €142 million) and Emerging Markets (+100.0%, at €65 million). However sales were lower in in the United States (-19.0% CER, at €171 million), mainly as a result of a decrease in the net average selling price.
In the first half of 2018, net sales of Apidra® rose by 3.1% CER to €183 million. Lower sales in the United States (-19.6% CER, at €40 million) were compensated for by sales growth in Emerging Markets (+29.2% CER, at €54 million) and Europe (+4.5% CER, at €70 million).
In the first half of 2018, net sales of Amaryl® were up 5.8% CER at €170 million, of which €146 million was generated in Emerging Markets (+10.6% CER).
Soliqua® 100/33 and Suliqua™ (insulin glargine 100 units/ml and lixisenatide 33 mcg/ml injectable) were launched in the United States in January 2017, and various European and Emerging Markets countries during the rest of 2017. Sales of Soliqua® 100/33 / Suliqua™ amounted to €26 million in the first half of 2018.
66 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
CARDIOVASCULAR FRANCHISE
In the first half of 2018, sales of Praluent® (developed in collaboration with Regeneron) rose by 55.3% CER to €111 million (including €61 million in the United States, reflecting increased market access for the product, and €41 million in Europe).
As mentioned in Section A.1.3. of this half-year management report, Sanofi and Regeneron presented positive results from the ODYSSEY OUTCOMES trial in March 2018. Those results showed that Praluent® significantly reduced the risk of major adverse cardiovascular events in patients who had suffered a recent acute coronary syndrome event. Sanofi and Regeneron also announced measures to help ensure more affordable and timely access to Praluent® for high-risk patients with significant unmet needs. Discussions are ongoing with US payers to streamline the reimbursement criteria for Praluent®.
In the first half of 2018, net sales of Multaq® were €162 million, down 1.7% CER. Sales were generated primarily in the United States (net sales of €135 million, down 1.9% CER) and in Europe (€21 million, down 4.5% CER).
ESTABLISHED PRESCRIPTION PRODUCTS
Net sales of Established Prescription Products in the first half of 2018 amounted to €4,586 million, down 12.3% on a reported basis and 7.1% at constant exchange rates. Stronger sales in Emerging Markets (+8.7% CER, at €1,958 million) failed to offset lower net sales in the United States due to competition from generics of Renvela®/Renagel®; the impact of generic competition in Japan, especially for Plavix® and Aprovel®; and a decrease in net sales in Europe (-3.9% CER, at €1,719 million). In the United States and the Rest of the World region, net sales of Established Prescription Products were down 44.1% CER (at €379 million) and 20.0% CER (at €530 million), respectively.
In the first half of 2018, net sales of Lovenox® totaled €768 million, down 1.5% CER; this reflects competition in Europe (-5.4% CER, at €471 million) with the arrival of biosimilars in the United Kingdom, Germany and Italy. This more than offset a good performance in Emerging Markets (+9.9% CER, at €236 million).
Net sales of Plavix® in the first half of 2018 were €761 million, a rise of 4.6% CER; this reflected competition from generics in Japan (-32.8% CER, at €79 million), although the effect was more than offset by another strong performance in Emerging Markets (+14.6% CER, at €575 million), especially China (+17.2%, at €441 million). Sales of Plavix® in the United States and Puerto Rico are handled by BMS under the terms of the Sanofi-BMS alliance1.
In the first half of 2018, net sales of Aprovel® /Avapro® amounted to €343 million, down 4.7% CER, reflecting competition from generics in Japan (-66.7% CER, at €22 million) and Europe (-8.3% CER, at €55 million). The effect was partly offset by stronger sales in Emerging Markets (+15.1% CER, at €242 million), especially China (+20.7% CER, at €157 million).
In the first half of 2018, net sales of Renvela® /Renagel® came to €201 million, down 55.7% CER, due largely to competition from generics in the United States (-67.5% CER, at €121 million) where the first generics of the product in powder form and pill form were authorized in June and July 2017, respectively. In October 2017, Sanofi launched its own authorized generic version of Renvela®/Renagel® in the United States. In Europe, sales of Renvela®/Renagel® were down 13.5% CER at €32 million, also due to generic competition.
GENERICS
Net sales of Generics for the first half of 2018 totaled €837 million, down 7.6% on a reported basis and 0.3% at constant exchange rates. Higher sales in Emerging Markets (+4.1% CER, at €351 million) and the Rest of the World region (+21.9% CER, at €71 million) failed to offset lower sales in Europe (-4.7% CER, at €367 million) and the United States (-21.7% CER, at €48 million).
In June 2018, Advent International and Sanofi announced that they had finalized negotiations with a view to Advent acquiring Zentiva, Sanofi’s European generics business, for an enterprise value of €1.9 billion. The transaction is expected to close in the fourth quarter of 2018, subject to clearance from the regulatory authorities (see Section A.1.1. of this Half-Year Management Report).
| 1 | See Note C.2 to our consolidated financial statements for the year ended December 31, 2017, on page F-33 of our Annual Report on Form 20-F; this document is available on our corporate website, www.sanofi.com. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 67
2018 first-half Pharmaceuticals net sales by geographical region
| (€ million) |
Total GBU |
Europe(a) | Change CER |
United States |
Change CER |
Rest of the world(b) |
Change CER |
Emerging Markets(c) |
Change CER |
Total Franchise |
Change CER |
|||||||||||||||||||||||||||||||||
| Cerezyme® |
236 | 134 | -2.9 | % | 83 | +1.1 | % | 19 | -13.0 | % | 120 | +25.0 | % | 356 | +6.2 | % | ||||||||||||||||||||||||||||
| Cerdelga® |
73 | 22 | +100.0 | % | 47 | +10.4 | % | 4 | +33.3 | % | 1 | — | 74 | +29.0 | % | |||||||||||||||||||||||||||||
| Myozyme®/Lumizyme® |
349 | 188 | +11.8 | % | 133 | +9.6 | % | 28 | +3.3 | % | 56 | +5.0 | % | 405 | +9.4 | % | ||||||||||||||||||||||||||||
| Fabrazyme® |
320 | 87 | +7.4 | % | 179 | +5.8 | % | 54 | +9.3 | % | 38 | +4.7 | % | 358 | +6.5 | % | ||||||||||||||||||||||||||||
| Aldurazyme® |
71 | 38 | +0.0 | % | 21 | +9.1 | % | 12 | +0.0 | % | 32 | +0.0 | % | 103 | +1.8 | % | ||||||||||||||||||||||||||||
| Other |
121 | 33 | -2.9 | % | 46 | -21.3 | % | 42 | +6.7 | % | 21 | +9.1 | % | 142 | -5.5 | % | ||||||||||||||||||||||||||||
| Total Rare Diseases |
1,170 | 502 | +6.8 | % | 509 | +3.5 | % | 159 | +4.2 | % | 268 | +13.3 | % | 1,438 | +6.5 | % | ||||||||||||||||||||||||||||
| Aubagio® |
750 | 184 | -9.8 | % | 541 | +11.0 | % | 25 | -7.1 | % | 25 | +52.6 | % | 775 | +6.0 | % | ||||||||||||||||||||||||||||
| Lemtrada® |
195 | 92 | +1.1 | % | 93 | -19.2 | % | 10 | -43.8 | % | 12 | +36.4 | % | 207 | -10.8 | % | ||||||||||||||||||||||||||||
| Total Multiple Sclerosis |
945 | 276 | -6.4 | % | 634 | +5.2 | % | 35 | -20.5 | % | 37 | +46.7 | % | 982 | +2.0 | % | ||||||||||||||||||||||||||||
| Jevtana® |
191 | 78 | +4.0 | % | 84 | +16.0 | % | 29 | +23.1 | % | 11 | -13.3 | % | 202 | +10.2 | % | ||||||||||||||||||||||||||||
| Thymoglobulin® |
107 | 19 | -5.0 | % | 78 | +2.4 | % | 10 | +0.0 | % | 37 | +25.0 | % | 144 | +6.1 | % | ||||||||||||||||||||||||||||
| Eloxatin® |
16 | 1 | -50.0 | % | — | — | 15 | +6.7 | % | 74 | +6.8 | % | 90 | +5.6 | % | |||||||||||||||||||||||||||||
| Taxotere® |
17 | 2 | +0.0 | % | 1 | +0.0 | % | 14 | -17.6 | % | 67 | +2.9 | % | 84 | -1.1 | % | ||||||||||||||||||||||||||||
| Mozobil® |
77 | 24 | +9.1 | % | 45 | +2.0 | % | 8 | +50.0 | % | 5 | +66.7 | % | 82 | +10.0 | % | ||||||||||||||||||||||||||||
| Zaltrap® |
41 | 26 | +0.0 | % | 4 | +25.0 | % | 11 | — | 5 | +50.0 | % | 46 | +41.2 | % | |||||||||||||||||||||||||||||
| Other |
71 | 26 | -3.7 | % | 36 | -61.5 | % | 9 | -10.0 | % | 8 | +0.0 | % | 79 | -44.5 | % | ||||||||||||||||||||||||||||
| Total Oncology |
520 | 176 | +1.1 | % | 248 | -15.2 | % | 96 | +20.0 | % | 207 | +8.3 | % | 727 | -1.8 | % | ||||||||||||||||||||||||||||
| Eloctate® |
219 | — | — | 187 | — | 32 | — | — | — | 219 | — | |||||||||||||||||||||||||||||||||
| Alprolix® |
102 | — | — | 83 | — | 19 | — | — | — | 102 | — | |||||||||||||||||||||||||||||||||
| Total Rare Blood Disorder |
321 | — | — | 270 | — | 51 | — | — | — | 321 | — | |||||||||||||||||||||||||||||||||
| Dupixent® |
282 | 26 | — | 246 | +950.0 | % | 10 | — | 1 | — | 283 | +1,092.3 | % | |||||||||||||||||||||||||||||||
| Kevzara® |
30 | 5 | — | 23 | +2,500.0 | % | 2 | — | — | — | 30 | +3,200.0 | % | |||||||||||||||||||||||||||||||
| Total Immunology |
312 | 31 | — | 269 | +1,007.4 | % | 12 | — | 1 | — | 313 | +1,170.4 | % | |||||||||||||||||||||||||||||||
| Sanofi Genzyme (Specialty Care) |
3,268 | 985 | +4.9 | % | 1,930 | +36.3 | % | 353 | +27.7 | % | 513 | +13.4 | % | 3,781 | +23.1 | % | ||||||||||||||||||||||||||||
| Lantus® |
1,310 | 355 | -9.4 | % | 816 | -32.4 | % | 139 | -7.9 | % | 492 | +4.8 | % | 1,802 | -19.1 | % | ||||||||||||||||||||||||||||
| Toujeo® |
349 | 142 | +42.0 | % | 171 | -19.0 | % | 36 | +31.0 | % | 65 | +100.0 | % | 414 | +10.7 | % | ||||||||||||||||||||||||||||
| Apidra® |
129 | 70 | +4.5 | % | 40 | -19.6 | % | 19 | +0.0 | % | 54 | +29.2 | % | 183 | +3.1 | % | ||||||||||||||||||||||||||||
| Amaryl® |
24 | 8 | -27.3 | % | 1 | +0.0 | % | 15 | -10.5 | % | 146 | +10.6 | % | 170 | +5.8 | % | ||||||||||||||||||||||||||||
| Insuman® |
37 | 36 | -10.0 | % | 1 | +0.0 | % | — | +0.0 | % | 10 | -20.0 | % | 47 | -12.5 | % | ||||||||||||||||||||||||||||
| Lyxumia® |
10 | 7 | -12.5 | % | — | — | 3 | -20.0 | % | 1 | +0.0 | % | 11 | -14.3 | % | |||||||||||||||||||||||||||||
| Soliqua® 100/33 / SuliquaTM |
25 | 2 | — | 23 | +188.9 | % | — | — | 1 | — | 26 | +222.2 | % | |||||||||||||||||||||||||||||||
| Other |
45 | 28 | -12.5 | % | 7 | — | 10 | +22.2 | % | 24 | +2,400 | % | 69 | +69.8 | % | |||||||||||||||||||||||||||||
| Total Diabetes |
1,929 | 648 | -0.3 | % | 1,059 | -28.3 | % | 222 | -1.6 | % | 793 | +14.7 | % | 2,722 | -10.9 | % | ||||||||||||||||||||||||||||
| Multaq® |
159 | 21 | -4.5 | % | 135 | -1.9 | % | 3 | +100.0 | % | 3 | +0.0 | % | 162 | -1.7 | % | ||||||||||||||||||||||||||||
| Praluent® |
107 | 41 | +115.8 | % | 61 | +26.4 | % | 5 | +150.0 | % | 4 | +150.0 | % | 111 | +55.3 | % | ||||||||||||||||||||||||||||
| Total Cardiovascular |
266 | 62 | +51.2 | % | 196 | +5.3 | % | 8 | +133.3 | % | 7 | +50.0 | % | 273 | +15.2 | % | ||||||||||||||||||||||||||||
| Diabetes & Cardiovascular |
2,195 | 710 | +2.7 | % | 1,255 | -24.6 | % | 230 | +0.0 | % | 800 | +15.0 | % | 2,995 | -9.0 | % | ||||||||||||||||||||||||||||
| Lovenox® |
768 | 471 | -5.4 | % | 20 | -24.1 | % | 41 | -4.3 | % | 236 | +9.9 | % | 768 | -1.5 | % | ||||||||||||||||||||||||||||
| Plavix® |
761 | 76 | -2.6 | % | — | — | 110 | -25.3 | % | 575 | +14.6 | % | 761 | +4.6 | % | |||||||||||||||||||||||||||||
| Aprovel® / CoAprovel® |
343 | 55 | -8.3 | % | 5 | -16.7 | % | 41 | -50.5 | % | 242 | +15.1 | % | 343 | -4.7 | % | ||||||||||||||||||||||||||||
| Depakine® |
230 | 84 | +2.4 | % | — | — | 7 | -12.5 | % | 139 | +12.0 | % | 230 | +7.6 | % | |||||||||||||||||||||||||||||
| Renagel®/Renvela® |
201 | 32 | -13.5 | % | 121 | -67.5 | % | 15 | -10.5 | % | 33 | +59.1 | % | 201 | -55.7 | % | ||||||||||||||||||||||||||||
| Synvisc® / Synvisc-One® |
160 | 13 | -23.5 | % | 111 | -22.2 | % | 7 | -12.5 | % | 29 | +43.5 | % | 160 | -14.6 | % | ||||||||||||||||||||||||||||
| Allegra® |
80 | 5 | -16.7 | % | — | — | 75 | -14.6 | % | — | — | 80 | -14.7 | % | ||||||||||||||||||||||||||||||
| Stilnox®/Ambien®/Myslee® |
116 | 20 | +0.0 | % | 22 | -14.3 | % | 42 | -17.5 | % | 32 | +9.4 | % | 116 | -8.0 | % | ||||||||||||||||||||||||||||
| Tritace® |
115 | 73 | -6.4 | % | — | — | 2 | +50.0 | % | 40 | +2.3 | % | 115 | -2.4 | % | |||||||||||||||||||||||||||||
| Other |
1,812 | 890 | -2.6 | % | 100 | -2.7 | % | 190 | -12.2 | % | 632 | -0.8 | % | 1,812 | -3.1 | % | ||||||||||||||||||||||||||||
| Total Established Prescription Products |
4,586 | 1,719 | -3.9 | % | 379 | -44.1 | % | 530 | -20.0 | % | 1,958 | +8.7 | % | 4,586 | -7.1 | % | ||||||||||||||||||||||||||||
| Generics |
837 | 367 | -4.7 | % | 48 | -21.7 | % | 71 | +21.9 | % | 351 | +4.1 | % | 837 | -0.3 | % | ||||||||||||||||||||||||||||
| Total Emerging Markets—Specialty Care |
513 | 513 | +13.4 | % | ||||||||||||||||||||||||||||||||||||||||
| Total Emerging Markets—Diabetes & Cardiovascular |
800 | 800 | +15.0 | % | ||||||||||||||||||||||||||||||||||||||||
| General Medicines & Emerging Markets |
6,736 | 2,086 | -4.1 | % | 427 | -42.2 | % | 601 | -16.6 | % | 3,622 | +10.2 | % | |||||||||||||||||||||||||||||||
| Total Pharmaceuticals |
12,199 | 3,781 | -0.6 | % | 3,612 | -5.4 | % | 1,184 | -3.5 | % | 3,622 | +10.2 | % | 12,199 | +0.5 | % | ||||||||||||||||||||||||||||
| (a) | Western Europe and Eastern Europe excluding Eurasia (Russia, Ukraine, Georgia, Belarus, Armenia and Turkey). |
| (b) | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
| (c) | World excluding United States, Canada, Western and Eastern Europe (apart from Eurasia), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
68 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.3.5. CONSUMER HEALTHCARE
During 2017, we gradually integrated the Consumer Healthcare operations of Boehringer Ingelheim (BI) into our Consumer Healthcare Global Business Unit (GBU). Following completion of the integration process and with effect from December 31, 2017, we have identified our Consumer Healthcare business as an operating segment. Consequently, the net sales of the Consumer Healthcare business are presented separately below.
Net sales of Consumer Healthcare products for the first half of 2018 were €2,353 million, down 5.3% on a reported basis but up 3.0% at constant exchange rates, driven by Emerging Markets (+12.3% CER, at €801 million) – especially Latin America – and by the Pain (+9.6% CER, at €628 million) and Digestive (+11.7% CER ,at €496 million) categories.
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
Change on a reported basis |
Change at constant exchange |
||||||||||||
| Allegra® |
229 | 250 | -8.4 | % | +2.0 | % | ||||||||||
| Mucosolvan® |
49 | 46 | +6.5 | % | +13.0 | % | ||||||||||
| Other |
302 | 363 | -16.8 | % | -10.7 | % | ||||||||||
| Allergy, Cough & Cold |
580 | 659 | -12.0 | % | -4.2 | % | ||||||||||
| Doliprane® |
161 | 156 | +3.2 | % | +4.5 | % | ||||||||||
| Buscopan® |
104 | 83 | +25.3 | % | +42.2 | % | ||||||||||
| Other |
363 | 385 | -5.7 | % | +4.7 | % | ||||||||||
| Pain |
628 | 624 | +0.6 | % | +9.6 | % | ||||||||||
| Dulcolax® |
109 | 103 | +5.8 | % | +12.6 | % | ||||||||||
| Enterogermina® |
94 | 89 | +5.6 | % | +14.6 | % | ||||||||||
| Essentiale® |
89 | 85 | +4.7 | % | +11.8 | % | ||||||||||
| Zantac® |
62 | 57 | +8.8 | % | +21.1 | % | ||||||||||
| Other |
142 | 144 | -1.4 | % | +5.6 | % | ||||||||||
| Digestive |
496 | 478 | +3.8 | % | +11.7 | % | ||||||||||
| Pharmaton® |
44 | 48 | -8.3 | % | +0.0 | % | ||||||||||
| Other |
286 | 304 | -5.9 | % | +2.3 | % | ||||||||||
| Nutritionals |
330 | 352 | -6.3 | % | +2.0 | % | ||||||||||
| Gold Bond® |
97 | 100 | -3.0 | % | +9.0 | % | ||||||||||
| Other |
222 | 273 | -18.7 | % | -11.0 | % | ||||||||||
| Other products |
319 | 373 | -14.5 | % | -5.6 | % | ||||||||||
| Total Consumer Healthcare |
2,353 | 2,486 | -5.3 | % | +3.0 | % | ||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
In Emerging Markets, Consumer Healthcare net sales reached €801 million in the first half, up 12.3% CER, boosted by Pain (+24.9% CER, at €239 million) and Digestive (+20.5% CER, at €211 million), and by strong performances in Brazil and Argentina.
In Europe, 2018 first-half Consumer Healthcare net sales remained stable at €706 million. A decline in sales of products in the “Other” category (-11.6% CER, at €60 million) was offset by growth for all our other Consumer Healthcare franchises.
Sales of Consumer Healthcare products in the United States totaled €541 million, down 5.3% CER versus the comparable period of 2017. The main franchise affected was Allergy, Cough & Cold (-19.4% CER, at €173 million), reflecting a lower incidence of seasonal allergies and competition from retailer own brands, especially in anti-allergy nasal sprays.
In the Rest of the World region, first-half net sales for the Consumer Healthcare business reached €305 million, up 2.5% CER, driven largely by sales in Japan (+5.6% CER, at €157 million).
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 69
| (€ million) | Total | Europe (a) | Change CER |
United States |
Change CER |
Rest of the world (b) |
Change CER |
Emerging Markets (c) |
Change CER | |||||||||||||||||||||
| Allegra® |
229 | 15 | +50.0 | % | 122 | -4.8% | 29 | -3.2% | 63 | +12.5% | ||||||||||||||||||||
| Mucosolvan® |
49 | 26 | +4.0 | % | — | +0.0% | 1 | +0.0% | 22 | +19.0% | ||||||||||||||||||||
| Other |
302 | 126 | -3.1 | % | 51 | -41.2% | 48 | +7.8% | 77 | +1.2% | ||||||||||||||||||||
| Allergy, Cough & Cold |
580 | 167 | +1.2 | % | 173 | -19.4% | 78 | +4.9% | 162 | +7.6% | ||||||||||||||||||||
| Doliprane® |
161 | 134 | +1.5 | % | — | +0.0% | — | +0.0% | 27 | +20.8% | ||||||||||||||||||||
| Buscopan® |
104 | 39 | +14.7 | % | — | +0.0% | 5 | +0.0% | 60 | +69.8% | ||||||||||||||||||||
| Other |
363 | 81 | -4.7 | % | 78 | -2.2% | 52 | +5.8% | 152 | +13.3% | ||||||||||||||||||||
| Pain |
628 | 254 | +1.2 | % | 78 | -2.2% | 57 | +5.2% | 239 | +24.9% | ||||||||||||||||||||
| Dulcolax® |
109 | 50 | +8.7 | % | 31 | +9.7% | 11 | +9.1% | 17 | +33.3% | ||||||||||||||||||||
| Enterogermina® |
94 | 35 | -2.8 | % | — | +0.0% | — | +0.0% | 59 | +24.5% | ||||||||||||||||||||
| Essentiale® |
89 | 19 | +0.0 | % | — | +0.0% | — | -100.0% | 70 | +13.4% | ||||||||||||||||||||
| Zantac® |
62 | — | +0.0 | % | 55 | +19.2% | 7 | +40.0% | — | +0.0% | ||||||||||||||||||||
| Other |
142 | 59 | -1.7 | % | 9 | -9.1% | 9 | -23.1% | 65 | +21.7% | ||||||||||||||||||||
| Digestive |
496 | 163 | +1.2 | % | 95 | +12.8% | 27 | +7.1% | 211 | +20.5% | ||||||||||||||||||||
| Pharmaton® |
44 | 8 | -11.1 | % | — | +0.0% | 1 | -100.0% | 35 | +5.3% | ||||||||||||||||||||
| Other |
286 | 54 | +3.8 | % | 18 | -9.1% | 122 | +3.1% | 92 | +3.0% | ||||||||||||||||||||
| Nutritionals |
330 | 62 | +1.6 | % | 18 | -9.1% | 123 | +2.3% | 127 | +3.6% | ||||||||||||||||||||
| Gold Bond® |
97 | — | +0.0 | % | 95 | +9.2% | 2 | +0.0% | — | +0.0% | ||||||||||||||||||||
| Other |
222 | 60 | -11.6 | % | 82 | -4.2% | 18 | -18.2% | 62 | -16.1% | ||||||||||||||||||||
| Other products |
319 | 60 | -11.6 | % | 177 | +2.6% | 20 | -16.7% | 62 | -16.1% | ||||||||||||||||||||
| Total Consumer Healthcare |
2,353 | 706 | +0.0 | % | 541 | -5.3% | 305 | +2.5% | 801 | +12.3% | ||||||||||||||||||||
| (a) | Western Europe and Eastern Europe excluding Eurasia (Russia, Ukraine, Georgia, Belarus, Armenia and Turkey). |
| (b) | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
| (c) | World excluding United States, Canada, Western and Eastern Europe (apart from Eurasia), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
C.3.6. HUMAN VACCINES (VACCINES) SEGMENT
In the first half of 2018, the Vaccines segment reported net sales of €1,522 million, down 15.4% on a reported basis and 9.3% at constant exchange rates. This mainly reflects the expected effect of the constraints on supplies of Pentaxim® in China, plus lower sales of Dengvaxia® (€1 million, versus €18 million for the first half of 2017) following the announcement of product label updates in November 2017. Net sales for the Vaccines segment in Emerging Markets were down 16.7% CER at €569 million. In the United States, Vaccines sales were down 12.6% CER, at €524 million. In Europe, sales reached €271 million (+16.2% CER), driven by the Adult Booster Vaccines franchise.
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017(a) (6 months) |
Change on a reported basis |
Change exchange |
||||||||||||
| Polio/Pertussis/Hib Vaccines (including Pentacel®, Pentaxim® & Imovax®) |
734 | 901 | -18.5 | % | -12.8 | % | ||||||||||
| Meningitis/Pneumonia Vaccines (including Menactra®) |
205 | 290 | -29.3 | % | -22.1 | % | ||||||||||
| Adult Booster Vaccines (including Adacel®) |
186 | 194 | -4.1 | % | +2.1 | % | ||||||||||
| Travel and Other Endemics Vaccines |
228 | 219 | +4.1 | % | +9.6 | % | ||||||||||
| Influenza Vaccines (including Vaxigrip® & Fluzone®) |
127 | 136 | -6.6 | % | +0.7 | % | ||||||||||
| Dengvaxia® |
1 | 18 | -94.4 | % | -94.4 | % | ||||||||||
| Other vaccines |
41 | 42 | -2.4 | % | +7.1 | % | ||||||||||
| Total Vaccines |
1,522 | 1,800 | -15.4 | % | -9.3 | % | ||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
Polio/Pertussis/Hib vaccines posted 2018 first-half net sales of €734 million, down 12.8% CER, reflecting constraints on supplies of Pentaxim® in China and tough comparatives in the first half of 2017. Those factors meant that sales for the franchise in Emerging Markets decreased by 21.4% CER to €338 million. Net sales of Polio/Pertussis/Hib vaccines were also lower in the United States (-10.0% CER, at €176 million), due to fluctuations in inventories of Pentacel® and Daptacel®.
Net sales of Meningitis/Pneumonia vaccines totaled €205 million, down 22.1% CER, due mainly to lower sales of Menactra® on a tough 2017 first-half comparative that reflected trends in orders from the CDC in the United States and high sales in Australia following an outbreak of meningitis.
70 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
Travel and Other Endemics vaccines posted a 9.6% rise in net sales to €228 million in the first half of 2018, driven by growth in sales of the yellow fever vaccine and Typhim®.
First-half 2018 net sales of Adult Booster vaccines were €186 million, up 2.1% CER, reflecting increased sales in Europe (+43.5% CER, at €66 million) as limitations on supplies of Repevax® ended.
Sales of Influenza vaccines were stable at €127 million. Lower sales in Emerging Markets (-7.9% CER, at €98 million) due to delays in deliveries for the vaccination campaign in the southern hemisphere were partly offset by sales growth in other geographies.
2018 first-half Vaccines net sales by geographical region
| (€ million) | Total | Europe (a) | Change CER |
United States |
Change CER |
Rest of World (b) |
Change CER |
Emerging Markets (c) |
Change CER |
|||||||||||||||||||||||||||
| Polio/Pertussis/Hib Vaccines (including Pentacel®, Pentaxim® & Imovax®) |
734 | 139 | +0.7 | % | 176 | -10.0 | % | 81 | +4.8 | % | 338 | -21.4 | % | |||||||||||||||||||||||
| Meningitis/Pneumonia Vaccines (including Menactra®) |
205 | — | -100.0 | % | 157 | -21.0 | % | 7 | -63.6 | % | 41 | -6.3 | % | |||||||||||||||||||||||
| Adult Booster Vaccines (including Adacel®) |
186 | 66 | +43.5 | % | 97 | -11.6 | % | 13 | +7.7 | % | 10 | -21.4 | % | |||||||||||||||||||||||
| Travel and Other Endemics Vaccines |
228 | 59 | +37.2 | % | 62 | -5.6 | % | 28 | +7.1 | % | 79 | +9.1 | % | |||||||||||||||||||||||
| Influenza Vaccines (including Vaxigrip® & Fluzone®) |
127 | 1 | +0.0 | % | 4 | +66.7 | % | 24 | +36.8 | % | 98 | -7.9 | % | |||||||||||||||||||||||
| Dengvaxia® |
1 | — | — | — | — | — | — | 1 | -94.4 | % | ||||||||||||||||||||||||||
| Other vaccines |
41 | 6 | +20.0 | % | 28 | — | 5 | +20.0 | % | 2 | — | |||||||||||||||||||||||||
| Total Vaccines |
1,522 | 271 | +16.2 | % | 524 | -12.6 | % | 158 | +0.6 | % | 569 | -16.7 | % | |||||||||||||||||||||||
| (a) | Western Europe and Eastern Europe excluding Eurasia (Russia, Ukraine, Georgia, Belarus, Armenia and Turkey). |
| (b) | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
| (c) | World excluding United States, Canada, Western and Eastern Europe (apart from Eurasia), Japan, South Korea, Australia, New Zealand and Puerto Rico. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 71
C.3.7. NET SALES BY GEOGRAPHICAL REGION
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017(a) (6 months) |
Change on a reported basis |
Change at constant exchange |
||||||||||||
| United States |
4,677 | 5,562 | -15.9 | % | -6.3 | % | ||||||||||
| Emerging Markets (b) |
4,992 | 5,185 | -3.7 | % | +6.8 | % | ||||||||||
| - of which Asia (including South Asia(c)) |
1,993 | 1,929 | +3.3 | % | +9.2 | % | ||||||||||
| - of which Latin America |
1,298 | 1,406 | -7.7 | % | +8.9 | % | ||||||||||
| - of which Africa and Middle East |
1,030 | 1,167 | -11.7 | % | -4.3 | % | ||||||||||
| - of which Eurasia (c) |
597 | 617 | -3.2 | % | +14.3 | % | ||||||||||
| Europe(e) |
4,758 | 4,761 | -0.1 | % | +0.3 | % | ||||||||||
| Rest of the World (f) |
1,647 | 1,816 | -9.3 | % | -2.1 | % | ||||||||||
| - of which Japan |
875 | 1,001 | -12.6 | % | -5.4 | % | ||||||||||
| - of which South Korea |
206 | 211 | -2.4 | % | +2.8 | % | ||||||||||
| Total net sales |
16,074 | 17,324 | -7.2 | % | -0.1 | % | ||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
| (b) | World excluding United States, Canada, Europe, Japan, South Korea, Australia, New Zealand and Puerto Rico. |
| (c) | India, Bangladesh and Sri Lanka. |
| (d) | Russia, Ukraine, Georgia, Belarus, Armenia and Turkey. |
| (e) | Western Europe and Eastern Europe (excluding Eurasia). |
| (f) | Japan, South Korea, Canada, Australia, New Zealand and Puerto Rico. |
2018 first-half net sales in the United States were €4,677 million, down 15.9% on a reported basis and down 6.3% on a constant structure basis and at constant exchange rates. The main factors were lower sales for the Diabetes franchise(-28.3% CER at €1,059 million) and the Established Prescription Products franchise (-44.1% CER, at €379 million, reflecting competition from generics of Renvela®/Renagel®). Those effects were partly offset by the performance of the Rare Blood Disorder franchise (€270 million) following the inclusion of Bioverativ in the consolidation with effect from March 9, 2018, and by the Immunology franchise which posted net sales of €269 million with the recent launches of Dupixent® and Kevzara®.
Emerging Markets net sales for the first half of 2018 reached €4,992 million, down 3.7% on a reported basis but up 6.8% CER. All our franchises achieved sales growth in Emerging Markets except for Vaccines (-16.7 CER, at €569 million). The strongest-growing franchises were Multiple Sclerosis (+46.7% CER, at €37 million), Diabetes (+14.7% CER, at €793 million), Rare Diseases (+13.3% CER, at €268 million), Consumer Healthcare (+12.3% CER, at €801 million) and Established Prescription Products (+8.7% CER, at €1,958 million). In Asia, 2018 first-half net sales rose by 9.2% CER to €1,993 million, driven by a solid performance in China (+12.3%, at €1,254 million), despite local constraints affecting supplies of Pentaxim®. In Latin America, net sales for the first half were up 8.9% CER at €1,298 million, driven by Brazil (+7.0% CER, at €541 million), Argentina and Mexico. The best performers in this zone were Consumer Healthcare (+23.6% CER, at €334 million) and Established Prescription Products (+8.5% CER, at €347 million). In the Africa and Middle East region, 2018 first-half net sales amounted to €1,030 million, down 4.3% CER, due largely to the divestment of our majority equity interest in the Maphar site (Morocco) at the end of the second quarter of 2017, in line with our simplification strategy; sales from that business are no longer consolidated by Sanofi. Solid performances in Saudi Arabia (+34.6% CER) and Algeria (+9.0% CER) were dented by lower sales in Morocco (-41.6% CER) and South Africa (-35.6% CER). Net sales in the Eurasia region advanced by 14.3% CER to €597 million in the first half of 2018, the strongest performers being Turkey (+20.0% CER, at €237 million) and Russia (+11.4% CER, at €307 million).
In Europe, first-half net sales were stable year-on-year at €4,758 million. Higher sales for Vaccines (+16,2% CER, at €271 million), the Rare Diseases franchise (+6.8% CER, at €502 million), the Immunology franchise (€31 million) and the Cardiovascular franchise (+51.2% CER, at €62 million) were offset by lower sales for Established Prescription Products (-3.9% CER, at €1,719 million) and Multiple Sclerosis (-6.4% CER, at €276 million).
In the Rest of the World region, net sales decreased by 2.1% CER to €1,647 million. In Japan, 2018 first-half net sales were €875 million (-5.4% CER) due to competition from generics of Plavix® and lower sales of Aprovel® and Allegra®, although the effect was partly offset by the performances of the Rare Blood Disorder, Oncology and Immunology franchises.
72 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.4. OTHER INCOME STATEMENT ITEMS
Comparatives for the six months ended June 30, 2017 and the year ended December 31, 2017 have been restated in accordance with the new standard on revenue recognition, IFRS 15, which is applicable from January 1, 2018 onwards. The impacts of those restatements are described in detail in Note A.1.2. to the condensed half-year consolidated financial statements; as well as affecting net sales, they also impact certain other items as discussed below.
The 2017 comparatives also reflect our new segmental reporting model. During 2017, we gradually integrated the Consumer Healthcare activities of Boehringer Ingelheim, which we acquired on January 1, 2017. The integration process having been completed on December 31, 2017, our Consumer Healthcare activities now constitute a separate operating segment. We have also realigned our management reporting structure to reflect changes to our organizational structure that took place in 2017. Consequently, the costs of our global functions (Medical Affairs, External Affairs, Finance, Human Resources, Legal Affairs, Information Solutions & Technologies, Sanofi Business Services, etc.) are now included as reconciling items within the “Other” segment.
C.4.1. OTHER REVENUES
Other revenues advanced by 2.7% to €533 million in the first half of 2018 (versus €519 million in the first half of 2017). This line item mainly comprises VaxServe sales of non-Sanofi products (€397 million, versus €368 million for the first half of 2017, within the Vaccines segment), and revenues under our agreements with Swedish Orphan Biovitrum AB.
C.4.2. GROSS PROFIT
Gross profit amounted to €11,342 million in the first half of 2018, versus €12,172 million a year earlier, a decrease of 6.8%. Gross margin increased year-on-year, representing 70.6% of net sales in the first half of 2018 (versus 70.3% in the first half of 2017).
For the Pharmaceuticals segment, gross margin for the first half of 2018 was 0.3 of a percentage point lower at 74.6%. Good performances from the Immunology, Rare Diseases and Multiple Sclerosis franchises, plus the inclusion of Bioverativ products in the consolidation, failed to offset lower average net prices for insulin glargines in the United States, competition from generics of Renagel®/Renvela®, and unfavorable foreign exchange effects.
Gross margin for the Consumer Healthcare segment rose by 0.5 of a percentage point in the first half of 2018 to 67.6%, thanks largely to a good performance in Emerging Markets.
The Vaccines segment saw gross margin decrease by 2.2 percentage points in the first half of 2018 to 56.0%, largely as a result of restrictions on supplies of Pentaxim® in China.
C.4.3. RESEARCH AND DEVELOPMENT EXPENSES
Research and development expenses (R&D expenses) amounted to €2,755 million in the first half of 2018 (versus €2,667 million in the first half of 2017) and represented 17.1% of net sales (versus 15.4% in the first half of 2017). Overall, R&D expenses increased by 3.3%, mainly due to the acquisitions of Bioverativ and Ablynx and to spending on immuno-oncology and diabetes programs in the Pharmaceuticals segment.
C.4.4. SELLING AND GENERAL EXPENSES
Selling and general expenses were €4,819 million in the first half of 2018 (30.0% of net sales), compared with €5,054 million for the first half of 2017 (29.2% of net sales). The year-on-year decrease of 4.6% was mainly due to exchange rate effects. At constant exchange rates, selling and general expenses increased year-on-year, reflecting the first-time consolidation of Bioverativ and Ablynx and investment in immunology, partly offset by lower spending on Diabetes in the United States, within the Pharmaceuticals segment.
For the Consumer Healthcare segment, 2018 first-half selling and general expenses were 1.9 percentage points lower at 33.5% of net sales, versus 35.4% in the first half of 2017. This was mainly due to synergies realized following the integration of Boehringer Ingelheim’s Consumer Healthcare business, as well as the reduction in marketing expenses linked to the launch of Xyzal® in the US in March 2017.
C.4.5. OTHER OPERATING INCOME AND EXPENSES
Other operating income amounted to €323 million in the first half of 2018 (versus €173 million in the first half of 2017), and Other operating expenses to €165 million (versus €71 million in the first half of 2017).
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 73
Overall, other operating income and expenses represented a net gain of €158 million in the first half of 2018, compared with a net gain of €102 million a year earlier, a net year-on-year improvement of €56 million.
| (€ million) | June 30, 2018 |
June 30, 2017 |
Change | |||||||||
| Other operating income |
323 | 173 | +150 | |||||||||
| Other operating expenses |
(165 | ) | (71 | ) | -94 | |||||||
| Other operating income/(expenses), net |
158 | 102 | +56 | |||||||||
The overall year-on-year improvement of €56 million mainly reflected gains on disposals relating to ongoing operations (€226 million in the first half of 2018, versus €57 million in the first half of 2017), arising on the divestment of some mature products in Latin America and certain Consumer Healthcare products in Europe. The effect was partly offset by a decrease in income from our pharmaceutical alliance partners (-€57 million in the first half of 2018, +€27 million in the first half of 2017), mainly relating to Regeneron following the launch of Dupixent® and Kevzara®, and by costs relating to the acquisitions of Bioverativ and Ablynx (recorded in “Other” for segmental reporting purposes).
C.4.6. AMORTIZATION OF INTANGIBLE ASSETS
Amortization charged against intangible assets in the first half of 2018 was €999 million, versus €990 million in the comparable period of 2017. This €9 million increase was due to (i) an increase in amortization expense generated by the intangible assets recognized in connection with the acquisitions of Bioverativ and Protein Sciences (€161 million and €29 million, respectively), partly offset by reductions in amortization expense due to life cycle effects on assets recognized on the acquisitions of Genzyme (€385 million in the first half of 2018, versus €458 million in the first half of 2017) and Aventis (€145 million in the first half of 2018, versus €204 million in the first half of 2017).
C.4.7. IMPAIRMENT OF INTANGIBLE ASSETS
The results of impairment tests on other intangible assets led to the recognition of an impairment loss of €101 million in the first half of 2018, mainly on Lemtrada®, a product marketed in the United States.
In the first half of 2017 this line item showed an impairment loss of €12 million, mainly comprising a write-down of rights relating to the marketed product Leukine®.
C.4.8. FAIR VALUE REMEASUREMENT OF CONTINGENT CONSIDERATION
Fair value remeasurements of contingent consideration relating to acquisitions (in accordance with the revised IFRS 3) represented a net gain of €10 million in the first half of 2018 versus a net expense of €100 million in the first half of 2017. These remeasurements mainly relate to the contingent value rights (CVRs) issued in connection with the acquisition of Genzyme (€23 million expense) and to contingent consideration payable to Bayer as a result of an acquisition made by Genzyme prior to the latter’s acquisition by Sanofi (gain of €33 million in the first half of 2018, versus expense of €84 million in the first half of 2017). See Note B.11. to our condensed half-year consolidated financial statements.
C.4.9. RESTRUCTURING COSTS AND SIMILAR ITEMS
Restructuring costs and similar items amounted to a charge of €607 million in the first half of 2018, compared with a charge of €364 million in the first half of 2017. Restructuring costs in the first half of 2018 mainly reflect (i) write-downs of industrial assets in the United States; (ii) employee-related expenses associated with headcount adjustment plans in Japan and Europe; and (iii) the costs of transferring the infectious diseases early stage R&D pipeline and research unit. Those transfer costs amount to €253 million and primarily consist of payments to Evotec over a five-year period, including an upfront payment of €60 million on finalization of the agreement in early July 2018.
C.4.10. OTHER GAINS AND LOSSES, AND LITIGATION
In the first half of 2018, the line item Other gains and losses, and litigation shows an expense of €67 million for separation costs associated with the process of disinvesting from the Generics business in Europe, before tax effects.
74 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.4.11. OPERATING INCOME
Operating income for the first half of 2018 was €2,162 million, 29.8% lower than the 2017 first-half figure of €3,080 million. This year-on-year change mainly reflects unfavorable foreign exchange effects on gross profit, including unfavorable foreign exchange effects on net sales.
C.4.12. FINANCIAL INCOME AND EXPENSES
Net financial expenses were €105 million for the first half of 2018, €18 million lower than the 2017 first-half figure of €123 million.
Financial expenses directly related to our debt, net of cash and cash equivalents (see the definition in Section C.5. below) increased slightly to €114 million, versus €110 million in the first half of 2017.
Other factors underlying the year-on-year change in net financial expense were:
| • | foreign exchange losses of €20 million, compared with foreign exchange gains of €1 million in the first half of 2017; |
| • | a higher level of gains on disposals of non-current financial assets (€63 million, versus €52 million for the first half of 2017); and |
| • | a reduction in the net interest cost on pension plans (€36 million, versus €47 million for the first half of 2017). |
C.4.13. INCOME BEFORE TAX AND INVESTMENTS ACCOUNTED FOR USING THE EQUITY METHOD
Income before tax and investments accounted for using the equity method for the first half of 2018 was €2,057 million, compared with €2,957 million for the first half of 2017, a decrease of 30.4%.
C.4.14. INCOME TAX EXPENSE
Income tax expense represented €297 million in the first half of 2018, versus €612 million in the first half of 2017, giving an effective tax rate (based on consolidated net income) of 14.4%, compared with 20.7% in the first half of 2017. The decrease in the effective tax rate can be attributed to Sanofi’s revised estimates in 2018 of direct and indirect impacts of the US tax reform (the Tax Cuts and Jobs Act of 2017). The effects of the US tax reform were based on a preliminary analysis of the Tax Cuts and Jobs Act of 2017. As more detailed information has become available adjustments have been made accordingly to reflect the progress of our analysis. In addition, 2017 included the additional 3% levy on dividends paid out in cash, which was annulled following the French Constitutional Council ruling of October 6, 2017.
Changes in the level of income tax expense are also significantly impacted by the tax effects of the amortization and impairment of intangible assets (€275 million in the first half of 2018, versus €349 million in the first half of 2017) and of restructuring costs (€183 million in the first half of 2018, versus €126 million in the first half of 2017).
The effective tax rate on our business net income is a non-GAAP financial measure. It is calculated on the basis of business operating income, minus net financial expenses and before (i) the share of profit/loss from investments accounted for using the equity method and (ii) net income attributable to non-controlling interests. We believe the presentation of this measure, used by our management, is also useful for investors as it provides a means to analyze the effective tax cost of our current business activities. It should not be seen as a substitute for the effective tax rate based on consolidated net income.
When calculated on business net income, our effective tax rate was 22% in the first half of 2018, compared with 24.5% in the first half of 2017 and 23.5 % for 2017 as a whole. The main impacts on this tax rate are the US tax reform (as explained above in our analysis of the change in the effective tax rate based on consolidated net income), as well as the geographical mix of the profits of Sanofi entities and the tax effects of the elimination of intragroup margin on inventory.
C.4.15. SHARE OF PROFIT/(LOSS) OF INVESTMENTS ACCOUNTED FOR USING THE EQUITY METHOD
Investments accounted for using the equity method contributed net income of €75 million in the first half of 2018, versus net income of €27 million in the comparable period of 2017. This line item mainly comprises our share of the profits and losses of Regeneron, which represented net income of €68 million in the first half of 2018 and €33 million in the first half of 2017.
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 75
C.4.16. NET INCOME EXCLUDING THE EXCHANGED/HELD-FOR-EXCHANGE ANIMAL HEALTH BUSINESS
Net income excluding the exchanged/held-for-exchange Animal Health business amounted to €1,835 million in the first half of 2018, versus €2,372 million in the first half of 2017.
C.4.17. NET INCOME/(LOSS) OF THE EXCHANGED/HELD-FOR-EXCHANGE ANIMAL HEALTH BUSINESS
In accordance with IFRS 5, the net income or loss of the Animal Health business is presented within a separate line item, Net income/(loss) of the exchanged/held-for-exchange Animal Health business, for 2017. On January 2, 2017, Sanofi and Boehringer Ingelheim (BI) confirmed that they had finalized the strategic transaction agreed in June 2016, involving the exchange of Sanofi’s Animal Health business (Merial) for BI’s Consumer Healthcare business. Consequently, for the first half of 2017 this line item shows the gain of €4,421 million on the divestment of the Animal Health business, net of taxes and before the impact of price adjustments and delayed business transfers.
C.4.18. NET INCOME
Net income amounted to €1,835 million in the first half of 2018, versus €6,793 million in the first half of 2017.
C.4.19. NET INCOME ATTRIBUTABLE TO NON-CONTROLLING INTERESTS
Net income attributable to non-controlling interests for the first half of 2018 was €57 million, against €64 million for the first half of 2017. This line item mainly comprises the share of pre-tax profits paid to BMS from territories managed by Sanofi (€42 million, versus €43 million in the first half of 2017); the year-on-year decrease was directly related to competition from generics of clopidogrel (the active ingredient of Plavix®) and of irbesartan (the active ingredient of Aprovel®) in Europe.
C.4.20. NET INCOME ATTRIBUTABLE TO EQUITY HOLDERS OF SANOFI
Net income attributable to equity holders of Sanofi amounted to €1,778 million in the first half of 2018, compared with €6,729 million in the first half of 2017.
Basic earnings per share (EPS) was €1.42, compared with €5.34 for the first half of 2017, based on an average number of shares outstanding of 1,247.8 million for the first half of 2018 and 1,260.3 million for the first half of 2017. Diluted EPS was also €1.42, compared with €5.30 for the first half of 2017, based on an average number of shares after dilution of 1,254.9 million for the first half of 2018 and 1,270.6 million for the first half of 2017.
76 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.5. SEGMENT RESULTS
Business operating income (as defined in Note B.20.1. to the condensed half-year consolidated financial statements) amounted to €4,126 million in the first half of 2018 versus €4,734 million in the first half of 2017, a decrease of 12.8%. It represented 25.7% of net sales, compared with 27.3% in the first half of 2017.
The comparatives for the year ended December 31, 2017 and the six months ended June 30, 2017 as presented below reflect (i) the impact of the first-time application of IFRS 15 (see Note A.1.2. to our condensed half-year consolidated financial statements) and our new segmental reporting model (see Section C.1.1. of this Half-Year Management Report).
The table below shows business operating income for the six-month periods ended June 30, 2018 and 2017:
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months)(a) |
Change | |||||||||
| Pharmaceuticals segment |
4,572 | 5,019 | -8.9 | % | ||||||||
| Consumer Healthcare segment |
820 | 782 | +4.9 | % | ||||||||
| Vaccines segment |
258 | 424 | -39.2 | % | ||||||||
| Other |
(1,524 | ) | (1,491 | ) | -2.2 | % | ||||||
| Business operating income |
4,126 | 4,734 | -12.8 | % | ||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to our condensed half-year consolidated financial statements), and of the presentation of segment data using Sanofi’s new segmental reporting model. |
The following tables present our segment results for the first half of 2018, the first half of 2017, and the year ended December 31, 2017:
First half of 2018
| June 30, 2018 (6 months) | ||||||||||||||||||||
| (€ million) | Pharmaceuticals | Consumer Healthcare |
Vaccines | Other | Total | |||||||||||||||
| Net sales |
12,199 | 2,353 | 1,522 | — | 16,074 | |||||||||||||||
| Other revenues |
134 | — | 399 | — | 533 | |||||||||||||||
| Cost of sales |
(3,230 | ) | (763 | ) | (1,068 | ) | (105 | ) | (5,166 | ) | ||||||||||
| Research and development expenses |
(2,113 | ) | (58 | ) | (268 | ) | (316 | ) | (2,755 | ) | ||||||||||
| Selling and general expenses |
(2,648 | ) | (788 | ) | (326 | ) | (1,047 | ) | (4,809 | ) | ||||||||||
| Other operating income and expenses |
132 | 82 | — | (56 | ) | 158 | ||||||||||||||
| Share of profit/(loss) of investments accounted for using the equity method |
150 | — | (1 | ) | — | 149 | ||||||||||||||
| Net income attributable to non-controlling interests |
(52 | ) | (6 | ) | — | — | (58 | ) | ||||||||||||
| Business operating income |
4,572 | 820 | 258 | (1,524 | ) | 4,126 | ||||||||||||||
First half of 2017(a)
| June 30, 2017 (6 months) | ||||||||||||||||||||
| (€ million) | Pharmaceuticals | Consumer Healthcare |
Vaccines | Other | Total | |||||||||||||||
| Net sales |
13,038 | 2,486 | 1,800 | — | 17,324 | |||||||||||||||
| Other revenues |
148 | — | 370 | 1 | 519 | |||||||||||||||
| Cost of sales |
(3,419 | ) | (818 | ) | (1,123 | ) | (135 | ) | (5,495 | ) | ||||||||||
| Research and development expenses |
(1,999 | ) | (52 | ) | (260 | ) | (356 | ) | (2,667 | ) | ||||||||||
| Selling and general expenses |
(2,807 | ) | (880 | ) | (363 | ) | (1,004 | ) | (5,054 | ) | ||||||||||
| Other operating income and expenses |
41 | 57 | 1 | 3 | 102 | |||||||||||||||
| Share of profit/(loss) of investments accounted for using the equity method |
71 | — | (1 | ) | — | 70 | ||||||||||||||
| Net income attributable to non-controlling interests |
(54 | ) | (11 | ) | — | — | (65 | ) | ||||||||||||
| Business operating income |
5,019 | 782 | 424 | (1,491 | ) | 4,734 | ||||||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to our condensed half-year consolidated financial statements), and of the presentation of segment data using Sanofi’s new segmental reporting model. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 77
Full year 2017(a)
| December 31, 2017 (12 months) | ||||||||||||||||||||
| (€ million) | Pharmaceuticals | Consumer Healthcare |
Vaccines | Other | Total | |||||||||||||||
| Net sales |
25,173 | 4,798 | 5,101 | — | 35,072 | |||||||||||||||
| Other revenues |
287 | — | 862 | — | 1,149 | |||||||||||||||
| Cost of sales |
(6,766 | ) | (1,612 | ) | (2,798 | ) | (271 | ) | (11,447 | ) | ||||||||||
| Research and development expenses |
(4,056 | ) | (123 | ) | (557 | ) | (736 | ) | (5,472 | ) | ||||||||||
| Selling and general expenses |
(5,649 | ) | (1,645 | ) | (728 | ) | (2,050 | ) | (10,072 | ) | ||||||||||
| Other operating income and expenses |
34 | 94 | (107 | ) | (17 | ) | 4 | |||||||||||||
| Share of profit/(loss) of investments accounted for using the equity method |
212 | 1 | 1 | — | 214 | |||||||||||||||
| Net income attributable to non-controlling interests |
(110 | ) | (15 | ) | — | — | (125 | ) | ||||||||||||
| Business operating income |
9,125 | 1,498 | 1,774 | (3,074 | ) | 9,323 | ||||||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to our condensed half-year consolidated financial statements), and of the presentation of segment data using Sanofi’s new segmental reporting model. |
Pharmaceuticals segment first-half business operating income, 2018 and 2017 (a)
| (€ million) | June 30, (6 months) |
as % of net sales |
June 30, (6 months)(a) |
as % of net sales |
Change | |||||||||||||||
| Net sales |
12,199 | 100.0 | % | 13,038 | 100.0 | % | -6.4 | % | ||||||||||||
| Other revenues |
134 | 1.1 | % | 148 | 1.1 | % | -9.5 | % | ||||||||||||
| Cost of sales |
(3,230 | ) | (26.5 | )% | (3,419 | ) | (26.2 | )% | -5.5 | % | ||||||||||
| Gross profit |
9,103 | 74.6 | % | 9,767 | 74.9 | % | -6.8 | % | ||||||||||||
| Research and development expenses |
(2,113 | ) | (17.3 | )% | (1,999 | ) | (15.3 | )% | +5.7 | % | ||||||||||
| Selling and general expenses |
(2,648 | ) | (21.7 | )% | (2,807 | ) | (21.5 | )% | -5.7 | % | ||||||||||
| Other operating income and expenses |
132 | 41 | ||||||||||||||||||
| Share of profit/(loss) of investments accounted for using the equity method |
150 | 71 | ||||||||||||||||||
| Net income attributable to non-controlling interests |
(52 | ) | (54 | ) | ||||||||||||||||
| Business operating income |
4,572 | 37.5 | % | 5,019 | 38.5 | % | -8.9 | % | ||||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to our condensed half-year consolidated financial statements), and of the presentation of segment data using Sanofi’s new segmental reporting model. |
Consumer Healthcare segment first-half business operating income, 2018 and 2017 (a)
| (€ million) | June 30, (6 months) |
as % of net sales |
June 30, (6 months)(a) |
as % of net sales |
Change | |||||||||||||||
| Net sales |
2,353 | 100.0 | % | 2,486 | 100.0 | % | -5.3 | % | ||||||||||||
| Other revenues |
— | — | — | — | — | |||||||||||||||
| Cost of sales |
(763 | ) | (32.4 | )% | (818 | ) | (32.9 | )% | -6.7 | % | ||||||||||
| Gross profit |
1,590 | 67.6 | % | 1,668 | 67.1 | % | -4.7 | % | ||||||||||||
| Research and development expenses |
(58 | ) | (2.5 | )% | (52 | ) | (2.1 | )% | +11.5 | % | ||||||||||
| Selling and general expenses |
(788 | ) | (33.5 | )% | (880 | ) | (35.4 | )% | -10.5 | % | ||||||||||
| Other operating income and expenses |
82 | 57 | ||||||||||||||||||
| Share of profit/(loss) of investments accounted for using the equity method |
— | — | ||||||||||||||||||
| Net income attributable to non-controlling interests |
(6 | ) | (11 | ) | ||||||||||||||||
| Business operating income |
820 | 34.8 | % | 782 | 31.5 | % | +4.9 | % | ||||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to our condensed half-year consolidated financial statements), and of the presentation of segment data using Sanofi’s new segmental reporting model. |
78 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
Vaccines segment first-half business operating income, 2018 and 2017 (a)
| (€ million) | June 30, (6 months) |
as % of net sales |
June 30, (6 months)(a) |
as % of net sales |
Change | |||||||||||||||
| Net sales |
1,522 | 100 | % | 1,800 | 100.0 | % | -15.4 | % | ||||||||||||
| Other revenues |
399 | 26.2 | % | 370 | 20.6 | % | +7.8 | % | ||||||||||||
| Cost of sales |
(1,068 | ) | (70.2 | )% | (1,123 | ) | (62.4 | )% | -4.9 | % | ||||||||||
| Gross profit |
853 | 56.0 | % | 1,047 | 58.2 | % | -18.5 | % | ||||||||||||
| Research and development expenses |
(268 | ) | (17.6 | )% | (260 | ) | (14.4 | )% | +3.1 | % | ||||||||||
| Selling and general expenses |
(326 | ) | (21.4 | )% | (363 | ) | (20.2 | )% | -10.2 | % | ||||||||||
| Other operating income and expenses |
— | 1 | ||||||||||||||||||
| Share of profit/(loss) of investments accounted for using the equity method |
(1 | ) | (1 | ) | ||||||||||||||||
| Net income attributable to non-controlling interests |
— | — | ||||||||||||||||||
| Business operating income |
258 | 17.0 | % | 424 | 23.6 | % | -39.2 | % | ||||||||||||
| (a) | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to our condensed half-year consolidated financial statements), and of the presentation of segment data using Sanofi’s new segmental reporting model. |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 79
C.6. CONSOLIDATED STATEMENTS OF CASH FLOWS
Summarized consolidated statements of cash flows
| (€ million) | June 30, 2018 (6 months) |
June 30, 2017 (6 months) |
December 31, 2017 (12 months) |
|||||||||
| Net cash provided by/(used in) operating activities |
1,773 | 2,556 | 7,379 | |||||||||
| Net cash provided by/(used in) investing activities |
(13,085 | ) | (1,062 | ) | (2,896 | ) | ||||||
| Net cash inflow from the exchange of the Animal Health business for BI’s Consumer Healthcare business |
5 | 4,349 | 3,535 | |||||||||
| Net cash provided by/(used in) financing activities |
8,494 | (5,192 | ) | (7,902 | ) | |||||||
| Impact of exchange rates on cash and cash equivalents |
(9 | ) | (47 | ) | (74 | ) | ||||||
| Net change in cash and cash equivalents |
(2,822 | ) | 604 | 42 | ||||||||
Net cash provided by operating activities came to €1,773 million in the first half of 2018, against €2,556 million in the first half of 2017.
Operating cash flow before changes in working capital for the first half of 2018 was €3,281 million, versus €3,835 million in the first half of 2017. Working capital requirements rose by €1,507 million in the first half of 2018, as opposed to an increase of €1,279 million in the first half of 2017, mainly as a result of movements in other current assets, current financial assets and other current liabilities (€1,232 million).
Net cash used in investing activities totaled €13,085 million in the first half of 2018, compared with €1,062 million in the first half of 2017.
Acquisitions of property, plant and equipment and intangible assets totaled €823 million, versus €998 million in the first half of 2017. There were €641 million of acquisitions of property, plant and equipment, most of which (€426 million) were in the Pharmaceuticals segment, primarily in industrial facilities . The Vaccines segment accounted for €153 million of acquisitions of property, plant and equipment during the period. Acquisitions of intangible assets (€182 million, versus €360 million in the first half of 2017) mainly comprised contractual payments for intangible rights under license and collaboration agreements.
Acquisitions of investments during the first half of 2018 totaled €12,816 million, net of the cash of acquired entities and after including assumed liabilities and commitments; this compares with €426 million in the first half of 2017. The main acquisitions in the first half of 2018 were Bioverativ (€8,932 million) and Ablynx (€3,642 million).
After-tax proceeds from disposals amounted to €486 million in the first half of 2018, and arose mainly from the sale of some Consumer Healthcare products to Cooper-Vemedia (€139 million), the divestment of equity interests in Impact Therapeutics (€94 million) and the divestment of some mature products in Latin America (€44 million). In the first half of 2017, after-tax proceeds from disposals amounted to €440 million, and arose mainly from the sale of some Consumer Healthcare products to Ipsen (€83 million) and the divestment of the equity interest in SPMSD (€127 million).
Net cash inflow from the exchange of the Animal Health business for BI’s Consumer Healthcare business comprises the following items for 2017: (i) the receipt by Sanofi of a balancing cash payment of €4,207 million; (ii) reimbursements of intragroup accounts with Merial entities totaling €967 million; (iii) the €1,784 million payment of the tax due on the gain arising on the divestment; and (iv) the cash held by the BI subsidiaries acquired by Sanofi. After taking account of final enterprise value adjustments, the total consideration for the businesses effectively transferred in 2017 was €10,557 million for the sale of the Animal Health business to BI, and €6,239 million for the acquisition of BI’s Consumer Healthcare business (see Note D.1. to the consolidated financial statements for the year ended December 31, 2017).
Net cash provided by/used in financing activities represented a net cash inflow of €8,494 million in the first half of 2018, compared with a net outflow of €5,192 million in the first half of 2017. The 2018 first-half figure includes the dividend payout to our shareholders of €3,773 million (versus €3,710 million in the first half of 2017), and the effect of changes in our share capital (repurchases of own shares, net of capital increases), amounting to €711 million (versus €1,601 million in the first half of 2017).
The net change in cash and cash equivalents in the first half of 2018 was a decrease of €2,822 million, compared with an increase of €604 million in the first half of 2017.
80 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
C.7. CONSOLIDATED BALANCE SHEET
Total assets were €112,778 million as of June 30, 2018, compared with €99,813 million as of December 31, 2017, an increase of €12,965 million.
Our debt, net of cash and cash equivalents was €21,278 million as of June 30, 2018, compared with €5,229 million as of December 31, 2017. We believe the presentation of this non-GAAP financial indicator, which is reviewed by our management, provides useful information to measure our overall liquidity and capital resources. We define “debt, net of cash and cash equivalents” as (i) the sum total of short term debt, long term debt, and interest rate derivatives and currency derivatives used to hedge debt, minus (ii) the sum total of cash and cash equivalents and interest rate derivatives and currency derivatives used to hedge cash and cash equivalents.
| (€ million) | June 30, 2018 |
December 31, 2017 |
||||||
| Long-term debt |
22,788 | 14,326 | ||||||
| Short-term debt and current portion of long-term debt |
6,153 | 1,275 | ||||||
| Interest rate and currency derivatives used to hedge debt |
(170 | ) | (57 | ) | ||||
| Cash and cash equivalents |
(7,493 | ) | (10,315 | ) | ||||
| Debt, net of cash and cash equivalents |
21,278 | 5,229 | ||||||
| Total equity |
56,361 | 58,239 | ||||||
| Gearing ratio |
37.8 | % | 9.0 | % | ||||
To assess our financing risk, we use the “gearing ratio”, another non-GAAP financial measure. This ratio (which we define as the ratio of debt, net of cash and cash equivalents, to total equity) increased from 9.0% as of December 31, 2017 to 37.8% as of June 30, 2018. Analyses of our debt as of June 30, 2018 and December 31, 2017 are provided in Note B.9. to the condensed half-year consolidated financial statements. We expect that the future cash flows generated by our operating activities will be sufficient to repay our debt. The financing arrangements in place as of June 30, 2018 at the Sanofi parent company level are not subject to covenants regarding financial ratios and do not contain any clauses linking credit spreads or fees to Sanofi’s credit rating.
Other key movements in the balance sheet are described below.
Total equity was €56,361 million as of June 30, 2018, versus €58,239 million as of December 31, 2017. The year-on-year change reflects the following principal factors:
| • | increases: our net income for the first half of 2018 (€1,835 million) and movements in currency translation differences (€804 million, mainly on the US dollar); and |
| • | decreases: the dividend payout to our shareholders (€3,773 million), and repurchases of our own shares (€729 million). |
As of June 30, 2018 we held 2.0 million of our own shares, recorded as a deduction from equity and representing 0.16% of our share capital.
Goodwill and Other intangible assets (€67,264 million in total) rose by €13,920 million year-on-year, the main factors being:
| • | increases: movements related to the acquisitions of Bioverativ (€2,640 million of goodwill and €8,154 million of other intangible assets) and Ablynx (€2,301 million of goodwill and €1,689 million of other intangible assets); and |
| • | decreases: amortization and impairment charged during the period (€1,163 million). |
Investments accounted for using the equity method (€2,964 million) increased by €117 million.
Other non-current assets were €231 million lower at €3,133 million. The main movement during the year was a decrease in the market value of the equity investment in Alnylam (€225 million).
Net deferred tax assets were €694 million as of June 30, 2018 versus €2,686 million as of December 31, 2017, a decrease of €1,992 million. This mainly reflects deferred taxes arising on the remeasurement of acquired intangible assets (€2,235 million).
Non-current provisions and other non-current liabilities (€8,949 million) decreased by €205 million, mainly due to a reduction in provisions for pensions and other post-employment benefits.
Liabilities related to business combinations and to non-controlling interests increased by €99 million to €1,468 million. The main reason for changes in this item is the impact of new transactions (€228 million), partly offset by contingent consideration payable to Bayer as a result of an acquisition made by Genzyme prior to the latter’s acquisition by Sanofi.
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 81
D/ RISK FACTORS AND RELATED PARTY TRANSACTIONS
D.1. RISK FACTORS
The risk factors to which Sanofi is exposed are described in our Annual Report on Form 20-F for the year ended December 31, 2017, filed with the US Securities and Exchange Commission on March 7, 2018. The nature of those risks has not significantly changed during the first half of 2018. With respect to data protection laws referred to under the “Claims and investigations relating to compliance, competition law, marketing practices, pricing, data privacy and other legal matters could adversely affect our business, results of operations and financial condition” risk factor in our Annual Report, the General Data Protection Regulation (GDPR) has created a range of new compliance obligations since it came into force within the European Union in May 2018. Violations of the GDPR carry financial risks due to penalties for data breach or improper processing of personal data (including a possible fine of up to 4% of total worldwide annual turnover for the preceding financial year for the most serious infringements) and may also harm our reputation. Also some uncertainty remains around the legal and regulatory environment for these evolving privacy and data protection laws. All those risk factors may materialize during the second half of 2018 or during subsequent periods.
D.2. RELATED-PARTY TRANSACTIONS
Our principal related parties are defined in Note D.33. to the consolidated financial statements included at item 18 of our Annual Report on Form 20-F for the year ended December 31, 2017 (page F-101)1.
Note B.5. to the condensed half-year consolidated financial statements provides a description of the principal transactions and balances for the six months ended June 30, 2018 with equity-accounted entities that qualify as related parties.
Sanofi did not enter into any transactions with key management personnel during the first half of 2018.
Financial relations with the Group’s principal shareholders fall within the ordinary course of business and were immaterial in the first half of 2018.
E/ OUTLOOK
At constant exchange rates, we expect growth in 2018 full-year business earnings per share2 (EPS) to be in a range from +3% to +5%, including the contribution from the Bioverativ and Ablynx acquisitions and barring major unforeseen adverse events. The impact of exchange rates on 2018 business EPS is estimated to be approximately -6%, based on July 2018 average exchange rates applied over the rest of the year.
Full-year business net income2 for 2017 was €6,943 million, giving business earnings per share of €5.523.
This guidance has been prepared using accounting methods consistent with those used in the preparation of our historical financial information. It draws upon assumptions defined by Sanofi and its subsidiaries, in particular regarding the following factors:
| • | growth in the national markets in which we operate; |
| • | healthcare reimbursement policies, pricing reforms, and other governmental measures affecting the pharmaceutical industry; |
| • | developments in the competitive environment, in terms of innovative products and the introduction of generics; |
| • | respect by others for our intellectual property rights; |
| • | progress on our research and development programs; |
| • | the impact of our operating cost control policy, and trends in our operating costs; |
| • | trends in exchange rates and interest rates; |
| • | the integration of contributions from our acquisitions; and |
| • | the average number of shares outstanding. |
Some of the information, assumptions and estimates concerned are derived from or based, in whole or in part, on judgments and decisions made by Sanofi management that may be liable to change or adjustment in future.
| 1 | This report is available on our corporate website: www.sanofi.com. |
| 2 | For a definition, see Section C.2., “Business net income” above. |
| 3 | Includes the effects of first-time application of IFRS 15 on revenue recognition (see Note A.1.2. to the condensed half-year consolidated financial statements). |
82 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
FORWARD-LOOKING STATEMENTS
This document contains forward-looking statements as defined in the US Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These statements include projections and estimates and their underlying assumptions, statements regarding plans, objectives, intentions and expectations with respect to future financial results, events, operations, services, product development and potential, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects,” “anticipates,” “believes,” “intends,” “estimates,” “plans” and similar expressions. Although Sanofi’s management believes that the expectations reflected in such forward-looking statements are reasonable, investors are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Sanofi, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements.
These risks and uncertainties include among other things, the uncertainties inherent in research and development, future clinical data and analysis including post marketing, decisions by regulatory authorities such as the FDA or the EMA regarding whether and when to approve any drug, device or biological application that may be filed for any such product candidates as well as their decisions regarding labeling and other matters that could affect the availability or commercial potential of such product candidates, the absence of guarantee that the product candidates if approved will be commercially successful, the future approval and commercial success of therapeutic alternatives, Sanofi’s ability to benefit from external growth opportunities and/or obtain regulatory clearances, risks associated with intellectual property and current and future intellectual property litigation and the outcome thereof, trends in exchange rates and prevailing interest rates, the instability of economic conditions, the impact of cost containment initiatives and subsequent changes thereto, and the average number of shares outstanding, as well as those discussed or identified in the public filings with the Securities and Exchange Commission (SEC) and the Autorité des marchés financiers (AMF) made by Sanofi, including those listed under “Risk Factors”1 and “Cautionary Statement Regarding Forward-Looking Statements” in Sanofi’s annual report on Form 20-F for the year ended December 31, 2017. For an update on litigation, refer to Note B.14. “Legal and arbitration proceedings” to our condensed half-year consolidated financial statements for the six months ended June 30, 2018, and to section “A.4.2. Legal and arbitration proceedings” and section “D/ Risk factors and related party transactions” on pages 54 and 82 respectively of our half-year management report.
Other than as required by applicable law, Sanofi does not undertake any obligation to update or revise any forward-looking information or statements.
| 1 | See pages 4 to 18 of our 2017 Annual Report on Form 20-F, available on our corporate website: www.sanofi.com |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 83
F/ APPENDIX – RESEARCH AND DEVELOPMENT PIPELINE
O : Opt-in rights products for which rights have not been exercised yet
R : Registrational Study (other than Phase 3)
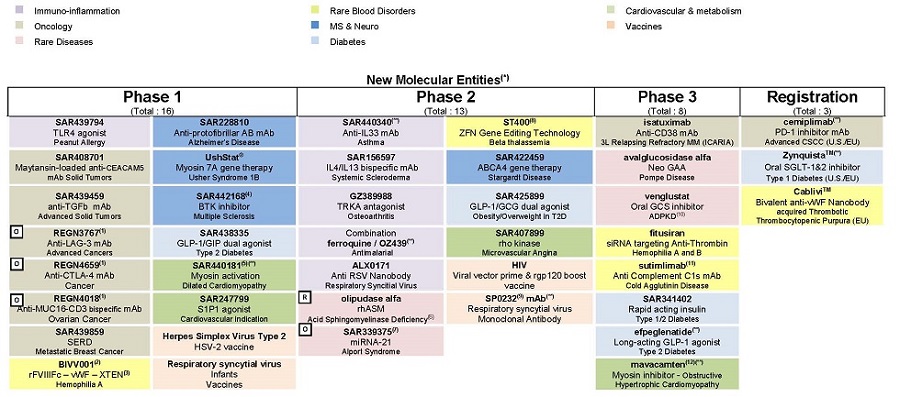
84 | Sanofi – 2018 HALF-YEAR FINANCIAL REPORT
Additional Indications(*)
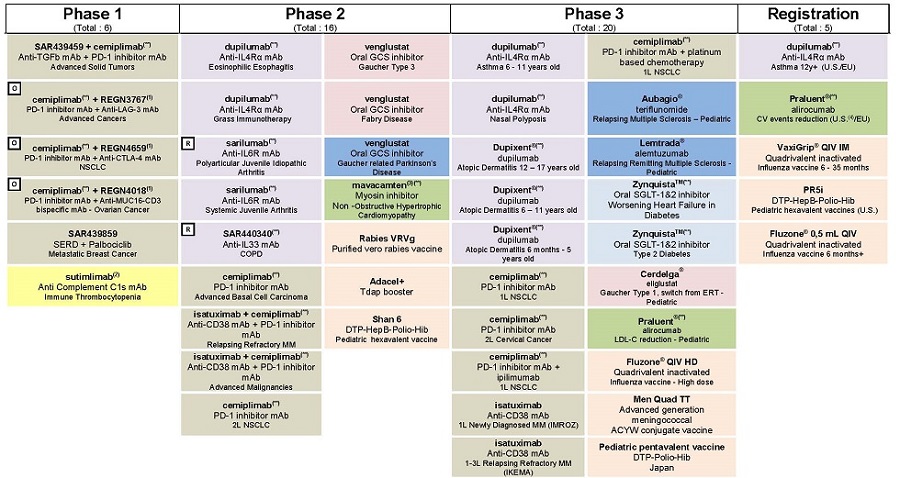
| (1) | Regeneron product for which Sanofi has opt-in rights |
| (2) | Also known as BIVV009 |
| (3) | Also known as SAR439152 and MYK461 |
| (4) | U.S. filing pending acceptance by FDA |
| (*) | Data related to all studies published on clinicaltrials.gov |
| (**) | Partnered and/or in collaboration - Sanofi may have limited or shared rights on some of these products |
2018 HALF-YEAR FINANCIAL REPORT – Sanofi | 85