Exhibit 99.2

R&D
DAYSeptember 30, 20201
AgendaSectionSpeakersWelcome
& Kick-off Will Lewis Chair & Chief Executive OfficerTreprostinil Palmitil Inhalation
Powder OpportunityDr. Eugene Sullivan, M.D. Chief Product Strategy Officer Dr. Ronald
J. Oudiz, M.D., FACP, FACC, FCCP Professor of Medicine and Interim Chief Harbor-UCLA
Medical Center Division of Cardiology Lead Investigator for Planned Phase 2a TrialBrensocatib
Development Program UpdateDr. Kevin Mange, M.D., MSCE Head of Clinical Development Colleen
Silk General Manager, BrensocatibARIKAYCE Front Line and Global Expansion UpdateDr. Kevin
Mange, M.D., MSCE Head of Clinical Development Neil Hughes General Manager, Head of Europe,
Middle East and Africa (EMEA) Commercial Roger Adsett Chief Operating OfficerFinancial
Update Closing RemarksSara Bonstein Chief Financial OfficerQ&A Session Insmed Executive
Team & Participants2

•
• • Forward Looking StatementsThistation contains fotwatd.tooti,.$llltttnents
that involvt- sutlstartiall rilob andties.. "kkfwarG-tooking b." as tflat termidtfint!d
in tPrivate Stnritie Utigation Refonn Ad of 1995,. areu that art! not hGbOfical facts
and k'l o olvt' a nutllbt!r of rills and uneertairtie. Words I!Nf!in as "mlfo/; .....,.....
•should." •OCM.td; "would," •tllpt!(ts..• "plans; "an tidpa " •bt!lit!\aes,.•
•tstifn&te," "prc;ects,: "predicn,." •intMcts; "pootentia• •continuea;nd
sitnitw t? presions (awell as othef wordor t? preuions ting Mure ts.conditiOont sci'turnstances)
trOf identify for\IW'¥'d.tooti,.statements. .. $tlllttnents in this pr r&arft!
iobansed 1Jf1011 the Comf*'IYC'l WN!nt expectations and bt!liefs, and iJwol oe known
and unknown rliob,. un:trtaintie and othef factol'l,. may autht! eotr.,an'(s adoal rtstJtis,.
and athiew!mt!OU and Utmi itlg of ts todifftr materially frcm the l!s, perfonnanet',
acfli ts or titning di$c:usst'd, projectf!d,. anticipated 01 indicattd in any forwarcHoot"tf18
statemt!nts. $U('h risks, I.W'!Ot!rtainties and Otllt!r factots indude, a.nong otlltn.
tilt- foloM.:trisk that brt!nsocatib GoeJ not prefftocti oe or uie fot patitnts in the
STQP.CO\I1Dl9 bulint!ss or «<notnit: disruptionsto catastro¢\es Ot otlltr
NMU. ind udiog natonldiW stersor public htaJttl O"Ges; impact of the nowcor (COVII>19)
panGt!mk and efforts to utt' its sprtad 01'1 OUt busintu, ttnployets, indudingPt'f'SOMd.
patients, par1J'Iers and .w; faiiUI'f! to sucxts:Sfully commercialae or maintain U.S.
approyal for ARIKAYCt", tht' Company'l OrfV appro..td product; uncertllintits in U*.gret-
ol martttand m111keting inh'asb'ucture at to p¥ttter 110ith UW'd p¥ties that
offer sucfl 11:n ir.f rastruc:ture f or diltribution of AIUKAYCE or any of tht! Comp3tly's
candidates that is &ppro..td in tfle future;hirurt! toobt!i'l rtgulatorvapproy&l
to expand ARIKA't'CE's indication to a tw'oaGt!r patient population; failure to suo::ts:Sfully
confutdinical b'illlJ f or AIUKAYCE, brM$001tib and the Company'l odlt!r pr'Oduct caruldates,.
ini'AI!Iins dut! to the Cornpr;;wly'l limited expt!rien:t- in conpreclinical deW! nt
actillitit'$ and clinical tri&l$ llt!a'Ssar( f OI"u.gu Moy approval and tfle eotr.,&i?(s
inability to tnroll or l'f!lllin suHicit!nt patients to c:onduct&nd compleott! utrill:I$or
Sf!n«M:t! data nect':S$¥'( f or reg\.btorv appro11lll;riW tflal tht! eotr.,an'(s
dinic&l studit!s will be delayt!d at that strious side effects wil bt! hk!ntifted
during crrug de<.lelopment; failto obtain.or dt!ltrvs in obtainq, rtgulatorvapproy&l$fat
ARICAVCE outstihe u.s. at for tht! an'(s product eandidMe: in tilt- U.S., C..oJapan C/1'
otlltr m111keb,. i11C'hrding tilt- United Kir'lgdom as a result of its recent exit from
the lin Unia; failure ol third parties ontht! Company is dept!illk!nt to m&nufed
si.KII'ietlt quantities of ARIKAYCf at the Company'l candidates for C:OI'Mltl'ti&l
or dinical nt!eds, to conduct tht! Ccmpany's dinkall trill:I'-or to v,ijt}J lawand tf!8utMiclns
that impBd theCOf'r1)&n'(s busint':S$ at ments with t"""'lin'(; tflt! Companfs inalllflly
to at1n1rt lind retain kper50Mel at to elftttNt!ly m111191Company's grcwtfl; tfle Company'l
inability to &dapt to its hiSJIIy titNt! &nd t!IY.rironment; the Company'l liflly
to ade(Jiately prot«t its i uaJ property riBJlts or prf!Yt!ntof its tradeand othtr
poropor'i!otaty inf Ot"mation andexpt!(tatioM indicated by theCcmpany's forwarcHooking
ts tlecause, bV thdr nature.forward-too-killiJ ts invrim andb«ause they relate
to NMts and depend on drcumltanees that"*'!or fro( not ootul' in tfut n.fat addltion&l
information &bout tflt! riSks andties that fro( llff«t tfle eotr.,&n'(s
bucillt!U,. plt!atlfll!UitldiKUSStd in Item lA."Rilol llctol'l,." in tht! CompAnnual
Report 01'1 form lO.K fat tfle yea. M*d r 31. 2019, Q.uarttrfo( Rt!ports 01'1 form 1()-Q
for the quartttS M*d M!«h 31, 2020 and AJne lO, 2020.. &nd IIJ?fS UMt CompBny
filtlgJ roith the soc.Tht! Company eai.Cions readt!rs not to pl&ce unGue l'f!lillnte
on &ny such forward- l«tting .ltlltements, vA!ic::fl speat Oflly as ol
the date ol this presentatim. The Company disclaitns any obtiglltitw,as $f!t:ifica!ly
!Wed by 111w and the rol tfle SEC. to pub&ly update or reuai ny such .ltllttmMts
to l'f!li&t &ny cfla,.in expt :tations 01' in nts.conditions Cl' drcumltlltiCt':S
on vdiich &nV sucfl statements "*'! II!! bast!or that may alfect tht! litlelihood
that actual rtsults'Aildiffer from thcbt'set forth in tfle forwatd.looti"S statements.IIC!Oefltanee
of ARIKA't'CE by phv*iam. patitnts, tflird-p611y P'l'fOI"S and otflers in thecommunity;
tht' Comp&ny'l inability to otJtain full approwll of ARIKAYCE from tOil. including
trisk that tht' Company roill not timt!ly and SUCXts:Sfully oompl'the stucJ'( to validatt'
a PAO tool and oomtht' 0011firmatorv pos:tof'llark!ti,. .m.v ruif!d fCI' full appnMI
of ARIKAYCC; inability ol tht! Company, PARI or tht! Company'lotht'r third party manufat.burs
to compfy v,ijtfl regutatorv rtquirtmMts rto ARIKAYCf or tlllmira• Nt!buliltr stht!
eotr.,an'(s in&bility to obtain lldeq ut'St!me. from soutmmt!nt or third-f:larty
payors f or AIUKAYCE or atteptable prices f or ARIKAYCC; dt!uelopment of u'llllttv 01'
elf icatV OOIIC:ti'TI$ rto ARIKAYCE or brM b; ina0C:Ur11C:ies in tfle eotr.,al?(s e:tin'lflte
of tflt! soi 1 tpotetti&l markets f or MIKA't'CE at its product candidates or in
data the Company has u$t'd to i ntify pfly.licians; exrate of f*ient uptake, Guration
of treatmt!nt, at ex p : titnt adi!Nf!nee or dGo:lntinuation rattht! eotr.,&i?(s
in&bifity to CN!alt! 11n dh!ctiw directOC»tS aswith lltiption at othtr proa!tdif'lgJ
relai:M to sucfl ; rtstriclion$ 01' othef obigations on UCompany by its gr ts rto ARIKAYCf
or tCotnpai?(s product candidates. including its k:ense ll mt!nts v,(jtfl PARI lind As11a2Mtca
Aa, lind failureof tfle Company tocompiV t/Oith its ob&g;Mions under sucflgr ts;
tfle oe»t and potenti.W N!putational result ing frC/1'1'1iliglltion tollroflicfl
theCcmpany isor maya f*tV, i11C'Iuding pr0l11uct btlffity dllims; Ueotr.,&n'(s lmittd
eJCperietlte Opt!rllting irumation&ll)t; cflain Ia"" and r ns &pplicallle to
tfle Company'$ bu$iness, ir'doldany pricirlg i'f!IOt"m, and flliiWto oompr:v with .M.ICh
laws and rtgulations; i nallillty to repay tht! Company's exiW"S inddltedness and unoertainties
""ith repect to tilt- Cornpr;;wly'l abilty to &oe:eu futeapitat; and Gt!l:tyl in
tfle tcution d plans to build out &n addilim&l fOA.·&pprO\Itd third-
paf1V manufaorturi"S ladlity and u pedt!d expMse: asKidllttd 'Aith thoseTht! Compa.,.,.
mav not ac.tuaty the reults., plans, intMtions or••••••••

Will
Lewis Chair & CEOMission, Vision & Strategy4

Transformational
Growth StrategyWorld Class TeamResearch & Development ExpertiseGlobal Commercial
PresenceBusiness Development StrengthBrensocatib NCFBEPipeline TPIP Brensocatib expansionBrensocatib
NCFBEARIKAYCE EU/Japan Front LineARIKAYCE EU/Japan Front LineARIKAYCE EU/Japan Front
LineARIKAYCE Refractory MACARIKAYCE Refractory MACARIKAYCE Refractory MACARIKAYCE Refractory
MAC5

Dr.
Eugene Sullivan, M.D. Chief Product Strategy OfficerDr. Ronald J. Oudiz, M.D., FACP,
FACC, FCCP Professor of Medicine and Interim Chief Harbor-UCLA Medical Center Division
of Cardiology Lead Investigator for Planned Phase 2a TrialTreprostinil Palmitil Development
Update 7

Harnessing
the Full Potential of the Prostanoid Pathway in Pulmonary Arterial Hypertension (PAH)Prostanoids
have long been considered the cornerstone therapy for PAH but clinical use has faced
challenges related to rapid metabolism and tolerability issuesTreprostinil palmitil has
the potential to address the shortcomings of the existing prostanoid therapies by:•
Potentially minimizing side effects, both local and systemic • Potentially maximizing
the vasodilatory activity, by maintaining high local concentrations in the lung •
Allowing for histologic disease-modifying effects of prostanoid therapy8

Agenda1
Pulmonary Arterial Hypertension (PAH)2 Treprostinil Palmitil Inhalation Powder (TPIP)3
Highlights of Preclinical Data4 Product Differentiation5 Path Forward9

Dr.
Ronald J. Oudiz, M.D., FACP, FACC, FCCP Professor of Medicine and Interim Chief Harbor-UCLA
Medical Center Division of Cardiology Lead Investigator Planned Phase 2a TrialPulmonary
Arterial Hypertension (PAH) Treatment Landscape10
Types
of Pulmonary Hypertension11
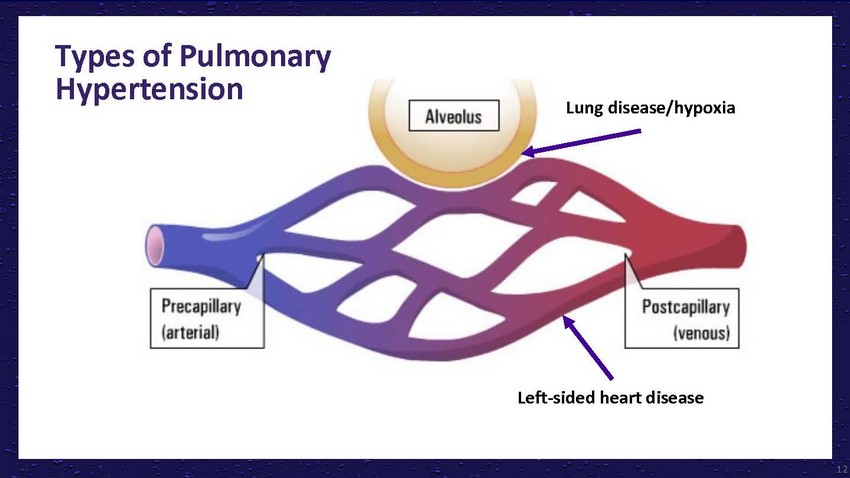
Types
of Pulmonary HypertensionLung disease/hypoxiaLeft-sided heart disease12
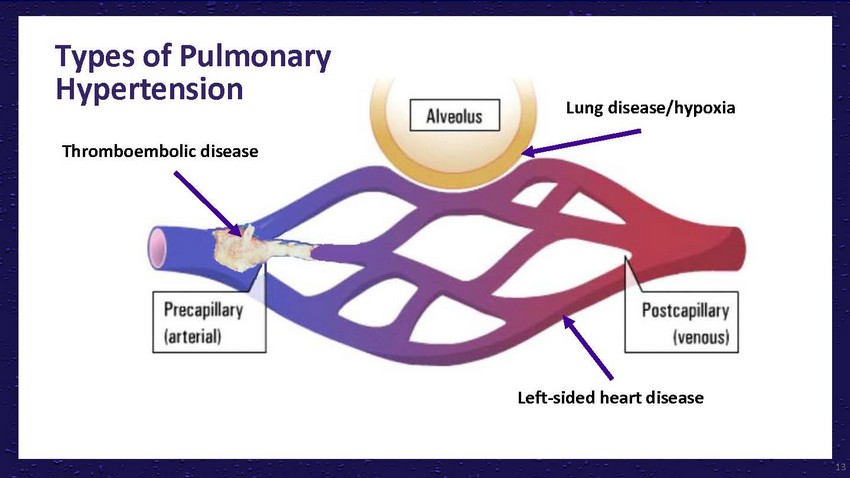
Types
of Pulmonary HypertensionLung disease/hypoxiaThromboembolic diseaseLeft-sided heart disease13
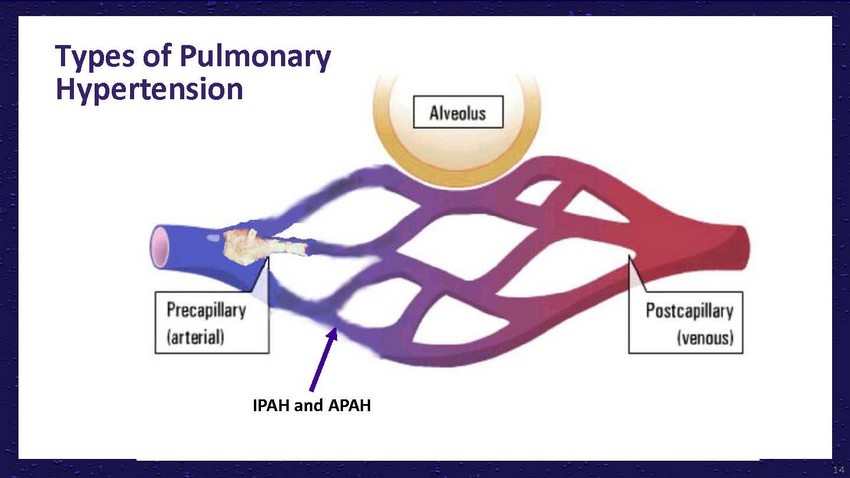
Types
of Pulmonary HypertensionThromboembolic diseaseIPAH and APAHLung disease/hypoxiaLeft-sided
heart disease14

Pulmonary
Hypertension: Diagnostic classificationPulmonary arterial hypertension (PAH)1.1. Idiopathic
PAH (IPAH) 1.2. Heritable 1.2.1. BMPR2 1.2.2. ALK1, ENG, SMAD9, CAV1, KCNK3 1.2.3. Unknown
1.3. Drug- and toxin-induced 1.4. Associated with 1.4.1. Connective tissue diseases 1.4.2.
HIV infectionPH due to Lung Diseases and/or Hypoxemia3.1. Chronic obstructive pulmonary
disease 3.2. Interstitial lung disease 3.3. Other pulmonary diseases with mixed restrictive
and obstructive pattern 3.4. Sleep-disordered breathing 3.5. Alveolar hypoventilation
disorders 3.6. Chronic exposure to high altitude 3.7. Developmental abnormalities1.4.3.
Portal hypertension 1.4.4. Congenital heart diseases 1.4.5. Schistosomiasis 1’
Pulmonary veno-occlusive disease and / or pulmonary capillary hemangiomatosis 1’
Persistent pulmonary hypertension of the newbornPulmonary hypertension due to left heart
disease2.1. Left ventricular systolic dysfunction 2.2. Left ventricular diastolic dysfunction
2.3. Valvular disease 2.4. Congenital/acquired left heart inflow/ outflow tract obstruction
and congenital cardiomyopathiesChronic thromboembolic pulmonary hypertension (CTEPH)Pulmonary
hypertension with unclear multifactorial mechanisms5.1. Hematologic disorders: chronic
hemolytic anemia, myeloproliferative disorders, splenectomy 5.2. Systemic disorders:
sarcoidosis, pulmonary histiocytosis: lymphangioleiomyomatosis 5.3. Metabolic disorders:
glycogen storage disease, Gaucher disease, thyroid disorders 5.4. Others: tumoral obstruction,
fibrosing mediastinitis, chronic renal failure, segmental PH15

Pulmonary
Hypertension: Diagnostic classificationPulmonary arterial hypertension (PAH)1.1. Idiopathic
PAH (IPAH) 1.2. Heritable 1.2.1. BMPR2 1.2.2. ALK1, ENG, SMAD9, CAV1, KCNK3 1.2.3. Unknown
1.3. Drug- and toxin-induced 1.4. Associated with 1.4.1. Connective tissue diseases 1.4.2.
HIV infectionPH due to Lung Diseases and/or Hypoxemia3.1. Chronic obstructive pulmonary
disease 3.2. Interstitial lung disease 3.3. Other pulmonary diseases with mixed restrictive
and obstructive pattern 3.4. Sleep-disordered breathing 3.5. Alveolar hypoventilation
disorders 3.6. Chronic exposure to high altitude 3.7. Developmental abnormalities1.4.3.
Portal hypertension 1.4.4. Congenital heart diseases 1.4.5. Schistosomiasis 1’
Pulmonary veno-occlusive disease and / or pulmonary capillary hemangiomatosis 1’
Persistent pulmonary hypertension of the newbornPulmonary hypertension due to left heart
disease2.1. Left ventricular systolic dysfunction 2.2. Left ventricular diastolic dysfunction
2.3. Valvular disease 2.4. Congenital/acquired left heart inflow/ outflow tract obstruction
and congenital cardiomyopathiesChronic thromboembolic pulmonary hypertension (CTEPH)Pulmonary
hypertension with unclear multifactorial mechanisms5.1. Hematologic disorders: chronic
hemolytic anemia, myeloproliferative disorders, splenectomy 5.2. Systemic disorders:
sarcoidosis, pulmonary histiocytosis: lymphangioleiomyomatosis 5.3. Metabolic disorders:
glycogen storage disease, Gaucher disease, thyroid disorders 5.4. Others: tumoral obstruction,
fibrosing mediastinitis, chronic renal failure, segmental PH16
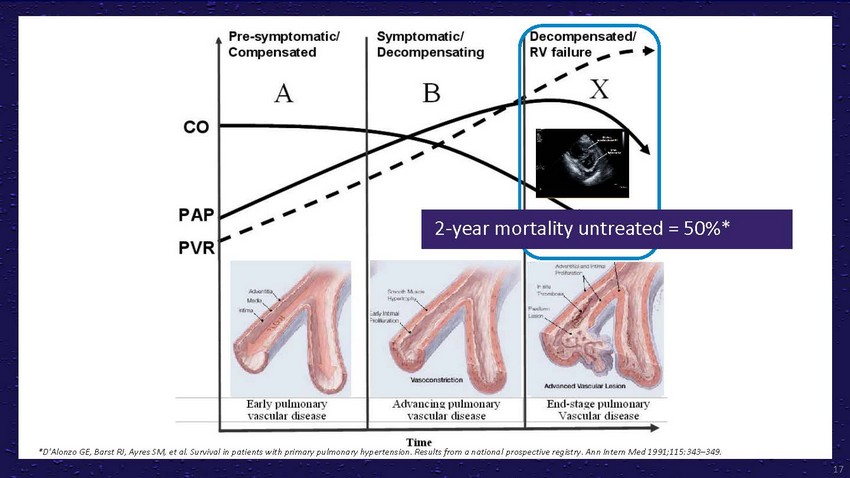
2-year
mortality untreated = 50%**D'Alonzo GE, Barst RJ, Ayres SM, et al. Survival in patients
with primary pulmonary hypertension. Results from a national prospective registry. Ann
Intern Med 1991;115:343–349.17
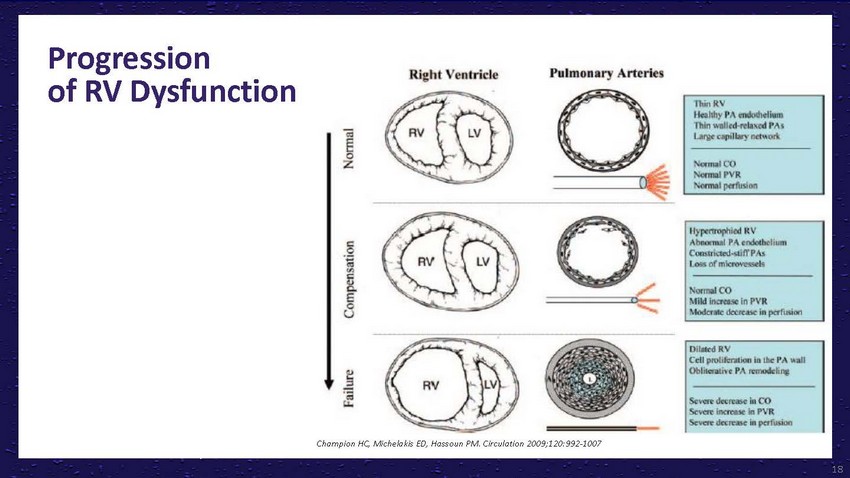
Progression
of RV DysfunctionChampion HC, Michelakis ED, Hassoun PM. Circulation 2009;120:992-100718

History,
symptoms,sogns and/or laboratory tests suggestove of PHAssess probaboloty of PHEchocardiographoc
probabolity of PH'Lowldentofy hogh-nsk patoentsFast-track referralof selected patientsHigh
or ontermedoateConsider V/Q scan to screen for CTEPH•Consider other causes and/or
follow-up•Doagnose common causes of PH V/0 scan abnormalDoagnose rare causes of
PHConsoder left heart dosease (assess pre-test probabolotyland lung doscase§No clonocally
sognifocant left heart dosease orRefer to PH expert centre!

History,
symptoms,sogns and/or laboratory tests suggestove of PHAssess probaboloty of PHEchocardiographoc
probabolity Low of PH'ldentofy hogh-nsk patoentsFast-track referralof selected patientsHigh
or ontermedoateConsider V/Q scan to screen for CTEPH•Consider other causes and/or
follow-up•Doagnose common causes of PH V/0 scan abnormalDoagnose rare causes of
PHConsoder left heart dosease (assess pre-test probabolotyland lung doscase§No clonocally
sognifocant left heart dosease orRefer to PH expert centre!

Algorithm
for the diagnosis of PH and its causes (cont)/ Management at PH expert centreICTEPH doagnostoe)+Any
m smatched -Review orNormalperfuSIOnalgonthm perlusoon defectperform f--.scan•PH
not PH not confirmed Consoder other confirmed! M ttomodabty d1.1gnoshc assessment l RHC
for PH doagno'' and haemodynam ,causes1 chara'trlf'IS..lhonPH conformedCTEPH not confirmed
Revoew by muttod scoptonary PH team• I Monitor andCTEPH confirmedl l \ss PH ctassolocatoonessI
RHC·Right Heart Catheterization IPH classoloedll Approproatenot certamConsoder
troat oltreatment treatment'

Algorithm
for the diagnosis of PH and its causes (cont)/ Management at PH expert centreICTEPH doagnostoe)+Any
m smatched -Review orNormalperfuSIOnalgonthm perlusoon defectperform f--.scan•!
M ttomodabty d1.1gnoshc assessment+PH notPHn RHC for PH doagno''confirmed Consoder other
confirm d and haemodynarn , causes1 ct)ara'trlf'IS..lhonPH conformedCTEPH not confirmed
Revoew by muttod scoptonary PH team•"""'I Monitor andCTEPH confirmedPH classoloedll\ss
PH ctassolocatoon not certamessI RHC·Right Heart Catheterization IApproproateConsoder
troat oltreatment treatment'
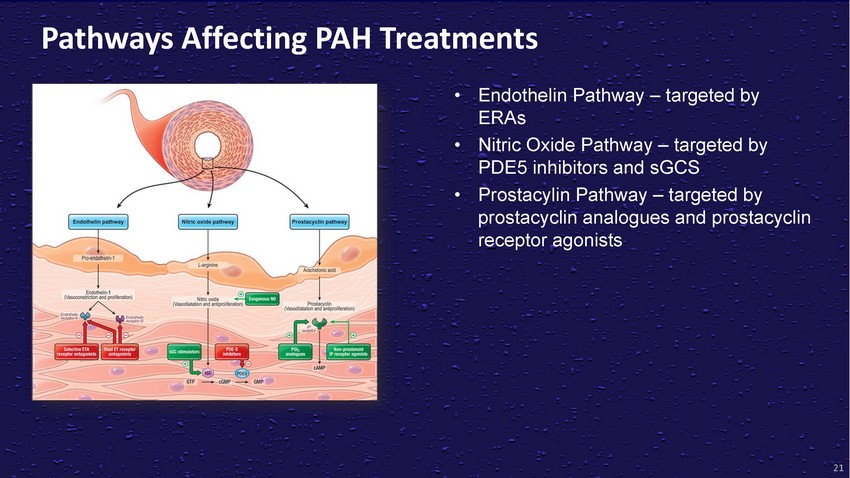
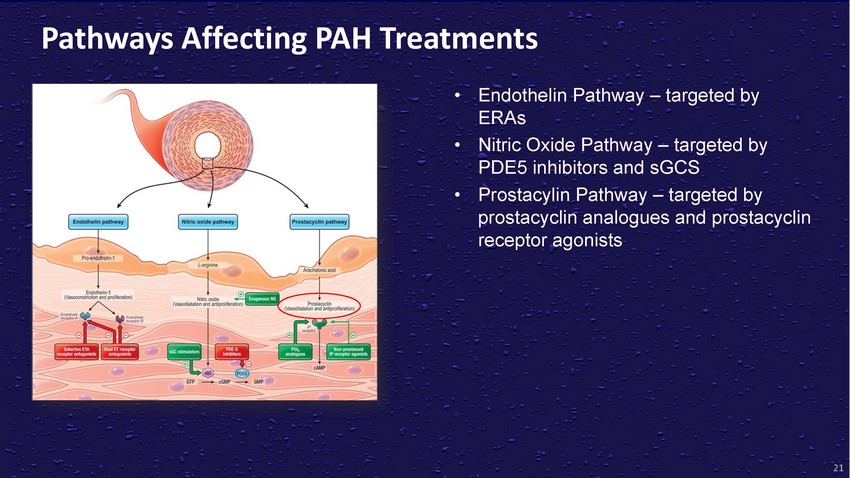
Pathways
Affecting PAH Treatments• Endothelin Pathway – targeted by ERAs• Nitric
Oxide Pathway – targeted by PDE5 inhibitors and sGCS• Prostacylin Pathway
– targeted by prostacyclin analogues and prostacyclin receptor agonists21
Pathways
Affecting PAH Treatments• Endothelin Pathway – targeted by ERAs• Nitric
Oxide Pathway – targeted by PDE5 inhibitors and sGCS• Prostacylin Pathway
– targeted by prostacyclin analogues and prostacyclin receptor agonists21

Pathways
Affecting PAH Treatments• Endothelin Pathway – targeted by ERAs• Nitric
Oxide Pathway – targeted by PDE5 inhibitors and sGCS• Prostacylin Pathway
– targeted by prostacyclin analogues and prostacyclin receptor agonistsParenteral
prostanoids are recommended as first- line therapy for FC IV patients and FC II and III
patients with rapid progression. Inhaled prostanoids are recommended for indicated patients
unwilling or unable to manage parenteral prostanoids, or as second-line alternatives
for patients who are uncontrolled on ERAs or PDE5s (alone or in combination).21

PAH
Treatments – Approved TherapiesSpecific PAH agentsProstacyclin Analogues•
epoprostenol (IV)• iloprost (inh)• treprostinil (IV, SQ, inh, po)• selexipag
(po)Endothelin antagonists• bosentan (po)• ambrisentan (po)• macitentan
(po)PDE-5 inhibitors• sildenafil (po)• tadalafil (po)Soluble gunalylate cyclase
agonist• riociguat (po)26

Prostacyclin
Advantages and Trade-OffsAdministration Advantage LimitationsOral Convenient • GI
side effects limitSystemically administered prostanoids (oral or parenteral) are limited
by the side effects:Parenteral EffectiveInhaled Targeteddosing• Systemic toxicities
limit dosing • Site pain, infection, inconvenience• Short treatment effect
requires multiple administrations per day • Sawtooth PK/PD profile• Hypotension
• Nausea/vomiting • Diarrhea • Headache• Flushing • Jaw pain27

Prostacyclin
Advantages and Trade-OffsAdministration Advantage LimitationsOral Convenient • GI
side effects limitSystemically administered prostanoids (oral or parenteral) are limited
by the side effects:Parenteral EffectiveInhaled Targeteddosing• Systemic toxicities
limit dosing • Site pain, infection, inconvenience• Short treatment effect
requires multiple administrations per day • Sawtooth PK/PD profile• Hypotension
• Nausea/vomiting • Diarrhea • Headache• Flushing • Jaw painGoal
for a new prostanoid therapy: convenient administration; sustained, high lung concentration;
lower systemic exposures; potential for anti-proliferative, disease-modifying effects.28

A
Poten Differentiated Prostanoid 29
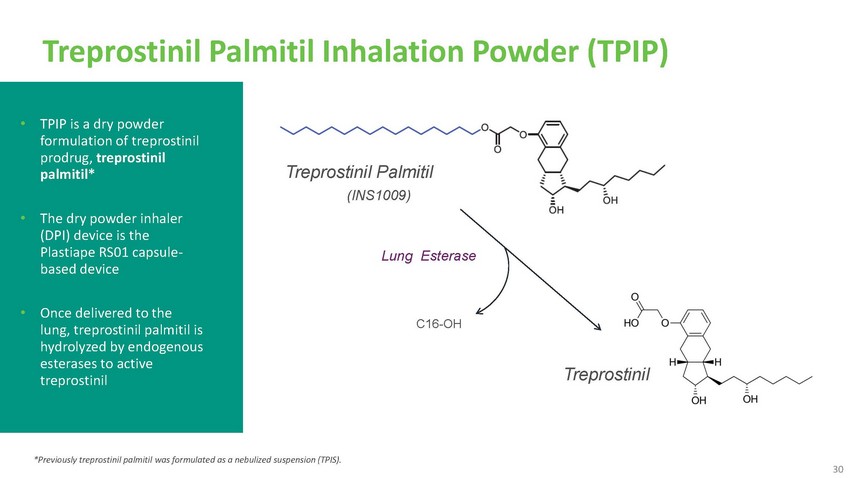
Treprostinil
Palmitil Inhalation Powder (TPIP)• TPIP is a dry powder formulation of treprostinil
prodrug, treprostinil palmitil*• The dry powder inhaler (DPI) device is the Plastiape
RS01 capsule- based deviceTreprostinil Palmitil(INS1009)Lung Esterase• Once delivered
to the lung, treprostinil palmitil is hydrolyzed by endogenous esterases to active treprostinil*Previously
treprostinil palmitil was formulated as a nebulized suspension (TPIS).C16-OHTreprostinil30

Highlights
of Preclinical Data 3

Highlights
of Preclinical Data Preclinical data demonstrate the following features of treprostinil
palmitil:Improved pharmacokinetics• Prolonged lung residence time • Lower systemic
Cmax and prolonged T1/2 compared to treprostinilProlonged duration of pharmacodynamic
activity compared to treprostinil“Local” pharmacodynamic effect• Achieves
the same pharmacodynamic (vasodilatory) effect at significantly lower systemic concentrations,
compared with IV treprostinilEffect on hallmark histopathologic features in the sugen/hypoxia
model• Remodeling effect, superior to other prostanoidsReduced incidence of cough
(guinea pig model)32

Highlights
of Preclinical DataPotential Implications of Preclinical Data Preclinical data demonstrate
the following features of treprostinil palmitil:Improved pharmacokinetics• Prolonged
lung residence time • Lower systemic Cmax and prolonged T1/2 compared to treprostinilProlonged
duration of pharmacodynamic activity compared to treprostinil“Local” pharmacodynamic
effect• Achieves the same pharmacodynamic (vasodilatory) effect at significantly
lower systemic concentrations, compared with IV treprostinilEffect on hallmark histopathologic
features in the sugen/hypoxia model• Remodeling effect, superior to other prostanoidsReduced
incidence of cough (guinea pig model)33

Potential
Implications of Preclinical Data Preclinical data demonstrate the following features
of treprostinil palmitil:Improved pharmacokinetics• Prolonged lung residence time
• Lower systemic Cmax and prolonged T1/2 compared to treprostinilProlonged duration
of pharmacodynamic activity compared to treprostinil“Local” pharmacodynamic
effect• Achieves the same pharmacodynamic (vasodilatory) effect at significantly
lower systemic concentrations, compared with IV treprostinilOnce-or-twice-daily administrationEffect
on hallmark histopathologic features in the sugen/hypoxia model• Remodeling effect,
superior to other prostanoidsReduced incidence of cough (guinea pig model)34

Potential
Implications of Preclinical Data Preclinical data demonstrate the following features
of treprostinil palmitil:Comparable effects at lower systemic exposuresImproved pharmacokinetics•
Prolonged lung residence time • Lower systemic Cmax and prolonged T1/2 compared
to treprostinilProlonged duration of pharmacodynamic activity compared to treprostinil“Local”
pharmacodynamic effect• Achieves the same pharmacodynamic (vasodilatory) effect
at significantly lower systemic concentrations, compared with IV treprostinilEffect on
hallmark histopathologic features in the sugen/hypoxia model• Remodeling effect,
superior to other prostanoidsReduced incidence of cough (guinea pig model)35

Potential
Implications of Preclinical Data Preclinical data demonstrate the following features
of treprostinil palmitil:Improved pharmacokinetics• Prolonged lung residence time
• Lower systemic Cmax and prolonged T1/2 compared to treprostinilProlonged duration
of pharmacodynamic activity compared to treprostinil“Local” pharmacodynamic
effect• Achieves the same pharmacodynamic (vasodilatory) effect at significantly
lower systemic concentrations, compared with IV treprostinilPotential disease modifying
activityEffect on hallmark histopathologic features in the sugen/hypoxia model•
Remodeling effect, superior to other prostanoidsReduced incidence of cough (guinea pig
model)36

Potential
Implications of Preclinical Data Preclinical data demonstrate the following features
of treprostinil palmitil:Improved pharmacokinetics• Prolonged lung residence time
• Lower systemic Cmax and prolonged T1/2 compared to treprostinilProlonged duration
of pharmacodynamic activity compared to treprostinil“Local” pharmacodynamic
effect• Achieves the same pharmacodynamic (vasodilatory) effect at significantly
lower systemic concentrations, compared with IV treprostinilPotential Improved TolerabilityEffect
on hallmark histopathologic features in the sugen/hypoxia model• Remodeling effect,
superior to other prostanoidsReduced incidence of cough (guinea pig model)37

Highlights
of Preclinical Data Preclinical data demonstrate the following features of treprostinil
palmitil:Improved pharmacokinetics• Prolonged lung residence time • Lower systemic
Cmax and prolonged T1/2 compared to treprostinilProlonged duration of pharmacodynamic
activity compared to treprostinil“Local” pharmacodynamic effect• Achieves
the same pharmacodynamic (vasodilatory) effect at significantly lower systemic concentrations,
compared with IV treprostinilEffect on hallmark histopathologic features in the sugen/hypoxia
model• Remodeling effect, superior to other prostanoidsReduced incidence of cough
(guinea pig model)38
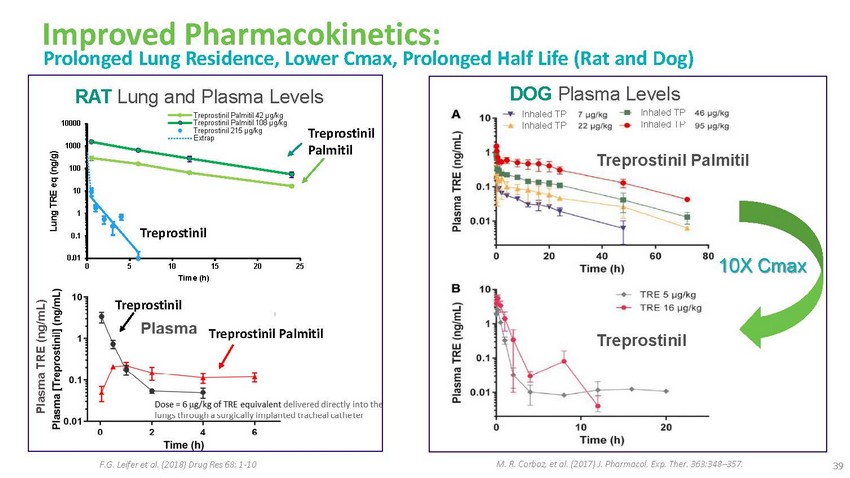
Improved
Pharmacokinetics: Prolonged Lung Residence, Lower Cmax, Prolonged Half Life (Rat and
Dog)RAT Lung and Plasma LevelsDOG Plasma Levels100001000100Treprostinil Palmitil 42 μg/kg
Treprostinil Palmitil 108 μg/kg Treprostinil 215 μg/kg ExtrapTreprostinil PalmitilInhaled
TP Inhaled TPInhaled TP Inhaled TPTreprostinil Palmitil1010.1Treprostinil0.010 5 10 15
20 25 Time (h)10X CmaxTreprostinilTreprostinil PalmitilTreprostinilF.G. Leifer et al.
(2018) Drug Res 68: 1-10M. R. Corboz, et al. (2017) J. Pharmacol. Exp. Ther. 363:348–357.
39

Highlights
of Preclinical Data Preclinical data demonstrate the following features of treprostinil
palmitil:Improved pharmacokinetics• Prolonged lung residence time • Lower systemic
Cmax and prolonged T1/2 compared to treprostinilProlonged duration of pharmacodynamic
activity compared to treprostinil“Local” pharmacodynamic effect• Achieves
the same pharmacodynamic (vasodilatory) effect at significantly lower systemic concentrations,
compared with IV treprostinilEffect on hallmark histopathologic features in the sugen/hypoxia
model• Remodeling effect, superior to other prostanoidsReduced incidence of cough
(guinea pig model)40
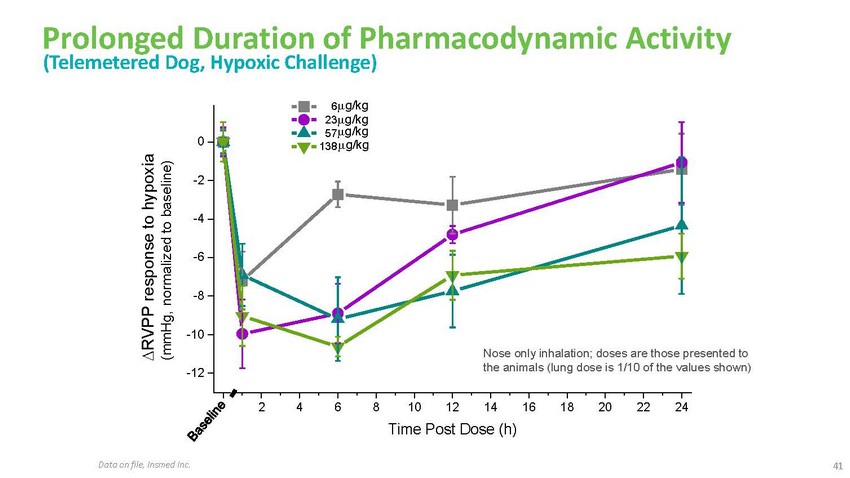
Prolonged
Duration of Pharmacodynamic Activity (Telemetered Dog, Hypoxic Challenge)6 g/kg 23g/kg
57g/kg 0 138 g/kg-2-4-6-8-10-12Nose only inhalation; doses are those presented to the
animals (lung dose is 1/10 of the values shown)0 2 4 6 8 10 12 14 16 18 20 22 24Time
Post Dose (h)Data on file, Insmed Inc.41
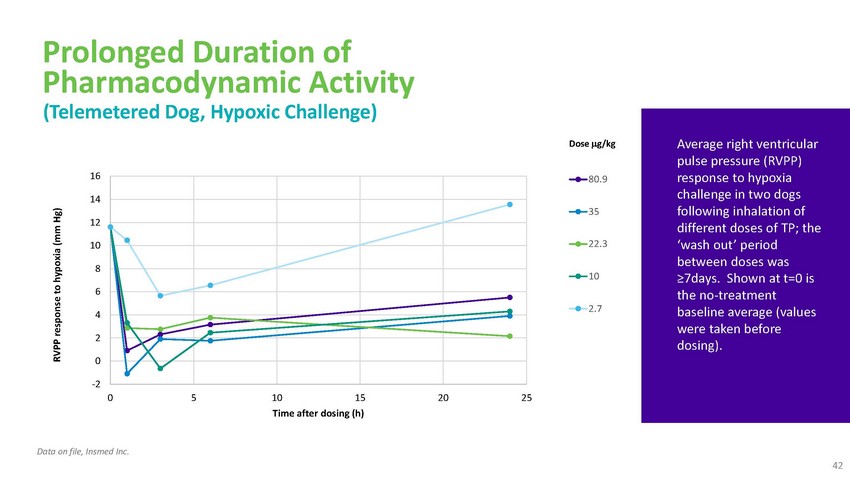
Prolonged
Duration of Pharmacodynamic Activity (Telemetered Dog, Hypoxic Challenge)1614121086420Dose
g/kg80.93522.3102.7Average right ventricular pulse pressure (RVPP) response to hypoxia
challenge in two dogs following inhalation of different doses of TP; the ‘wash
out’ period between doses was ≥7days. Shown at t=0 is the no-treatment baseline
average (values were taken before dosing).-2 0 5 10 15 20 25 Time after dosing (h)Data
on file, Insmed Inc. 42

Highlights
of Preclinical Data Preclinical data demonstrate the following features of treprostinil
palmitil:Improved pharmacokinetics• Prolonged lung residence time • Lower systemic
Cmax and prolonged T1/2 compared to treprostinilProlonged duration of pharmacodynamic
activity compared to treprostinil“Local” pharmacodynamic effect• Achieves
the same pharmacodynamic (vasodilatory) effect at significantly lower systemic concentrations,
compared with IV treprostinilEffect on hallmark histopathologic features in the sugen/hypoxia
model• Remodeling effect, superior to other prostanoidsReduced incidence of cough
(guinea pig model)43
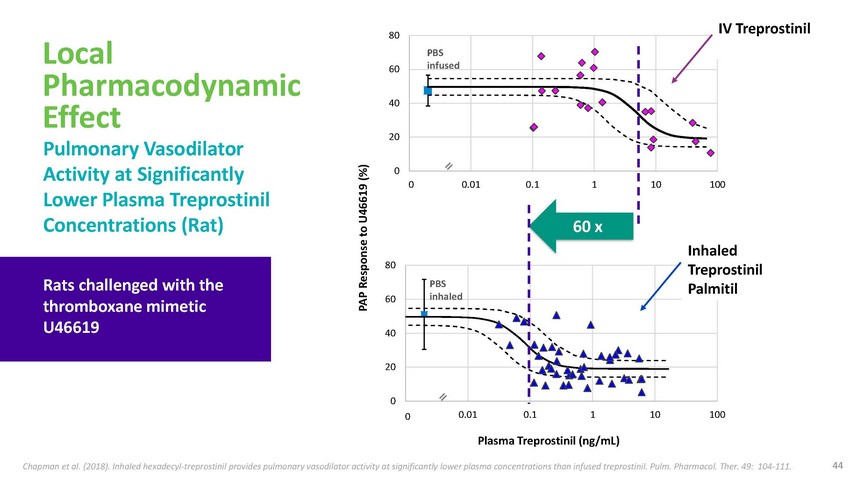
Local
Pharmacodynamic Effect Pulmonary Vasodilator Activity at Significantly80PBS 60 infused40200IV
TreprostinilLower Plasma Treprostinil Concentrations (Rat)0.0001 0.01 0.1 1 10 10060
xRats challenged with the thromboxane mimetic U4661980PBS 60 inhaled40Inhaled Treprostinil
Palmitil200 0.0001 0.01 0.1 1 10 100Plasma Treprostinil (ng/mL)Chapman et al. (2018).
Inhaled hexadecyl-treprostinil provides pulmonary vasodilator activity at significantly
lower plasma concentrations than infused treprostinil. Pulm. Pharmacol. Ther. 49: 104-111.
44
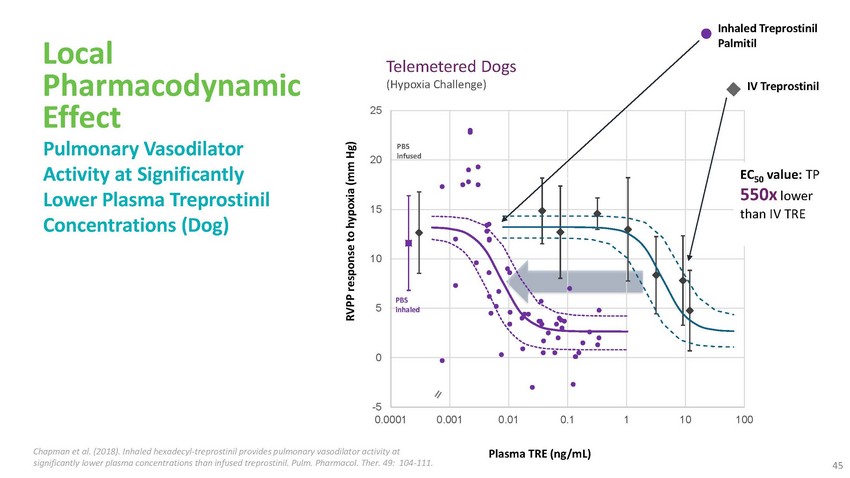
Local
Pharmacodynamic Effect Pulmonary Vasodilator Activity at Significantly Lower Plasma Treprostinil
Concentrations (Dog)Telemetered Dogs (Hypoxia Challenge)25PBS 20 infused15Inhaled Treprostinil
PalmitilIV TreprostinilEC50 value: TP 550x lower than IV TRE10PBS 5 inhaled0-5 0.0001
0.001 0.01 0.1 1 10 100Chapman et al. (2018). Inhaled hexadecyl-treprostinil provides
pulmonary vasodilator activity atPlasma TRE (ng/mL)significantly lower plasma concentrations
than infused treprostinil. Pulm. Pharmacol. Ther. 49: 104-111.45

Local
Pharmacodynamic Effect Continuously Inhaled vs. Continuously Infused (IV) Treprostinil
in Sheep…prostacyclin analogs are more potent pulmonary vasodilators when delivered
by aerosol than when given intravenously. Systemic hemodynamic effects are also minimized
by aerosol administration of the drug, making this approach especially appealing. Duration
of action of prostacyclin is short, requiring an unrealistic frequency of administration
for clinical use (6, 33), but development of analogs (e.g., treprostinil) or formulations
that are long acting could make this approach feasible.”“We speculate that
this may result from treprostinil stimulated production of vasodilatory mediators from
pulmonary epithelium.46

Highlights
of Preclinical Data Preclinical data demonstrate the following features of treprostinil
palmitil:Improved pharmacokinetics• Prolonged lung residence time • Lower systemic
Cmax and prolonged T1/2 compared to treprostinilProlonged duration of pharmacodynamic
activity compared to treprostinil“Local” pharmacodynamic effect• Achieves
the same pharmacodynamic (vasodilatory) effect at significantly lower systemic concentrations,
compared with IV treprostinilEffect on hallmark histopathologic features in the sugen/hypoxia
model• Remodeling effect, superior to other prostanoidsReduced incidence of cough
(guinea pig model)47

Treprostinil
Palmitil Inhalation Solution Improved Both Hemodynamics and Vascular Remodeling in the
Rat Sugen-HypoxiaModel of PAHImproves Pulmonary Arterial PressureReduces Wall Thickening*
significantly different from the naïve group# significantly different from the vehicle
group1Fulton Index is a commonlyTreprostinil Palmitil†NaïveVehicleTreprostinil
Palmitil†used quantitative measure of the degree of right ventricularImproves
Fulton Index1 Reduces Vessel ObliterationsMuscular Semi-muscular Non muscularhypertrophy
Am. J. Res. Crit. Care Med. 2020: 201, A2094. Poster was not presented due to COVID19.
Data on file, Insmed Inc.100 90 80 70 60 50 40 30 20 10 0*** # #Data on file, Insmed
Inc. †Pulmonary dosesTreprostinil Palmitil†NCaïtvr eVNeehgicClter
2ug/kg INS 1009 6ug/kg INS 1009 Treprostinil Palmitil† 48
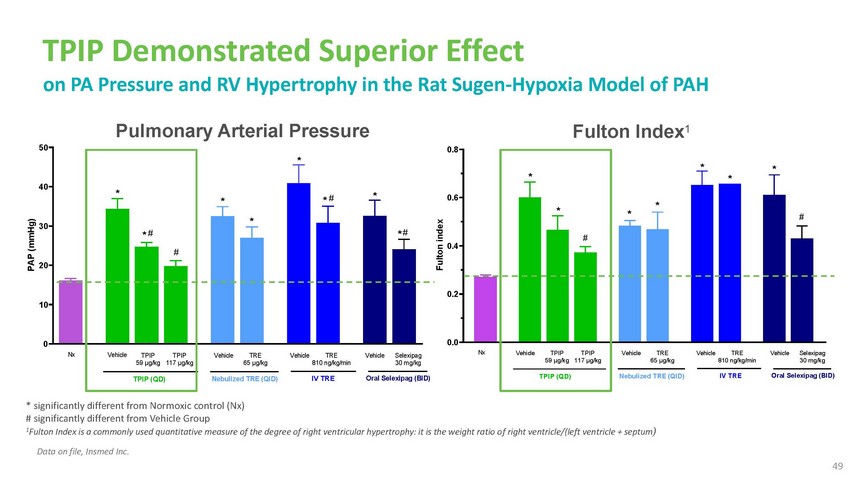
TPIP
Demonstrated Superior Effect on PA Pressure and RV Hypertrophy in the Rat Sugen-Hypoxia
Model of PAHPulmonary Arterial Pressure 50 *40 *0.8Fulton Index1* * * **30 * * ## 20*
# ** #0.60.4* * *#0.2 100 Nx VehicleTPIP 59 µg/kgTPIP (QD)TPIP 117 µg/kgVehicle
TRE 65 µg/kgNebulized TRE (QID)Vehicle TRE 810 ng/kg/minIV TREVehicle Selexipag
30 mg/kgOral Selexipag (BID)0.0Nx Vehicle TPIP 59 µg/kgTPIP (QD)TPIP 117 µg/kgVehicle
TRE 65 µg/kgNebulized TRE (QID)Vehicle TRE 810 ng/kg/minIV TREVehicle Selexipag
30 mg/kgOral Selexipag (BID)* significantly different from Normoxic control (Nx) # significantly
different from Vehicle Group 1Fulton Index is a commonly used quantitative measure of
the degree of right ventricular hypertrophy: it is the weight ratio o f right ventricle/(left
ventricle + septum)Data on file, Insmed Inc. 49

TPIP
Demonstrated Superior Effect on Vascular Remodeling in the Sugen-Hypoxia Rat Model of
PAHWall Thickness ������ ��������
������������
− ������� �����������������
�������� ������������
% = 40������ ��������
������������×
���***30 #***##******#******##1009080Non muscular Semi-muscular
Muscular*** *20100* 706050403020 ***10 *** 0##***### ***### ***### *********# ***#*********#
***# *********###***### ***NormoxicVehicle59 µg/kg117 µg/kgVehicle65 µg/kgVehicle
IV810Vehicle30 mg/kgNormoxicVehicle59 µg/kg117 µg/kg Vehicle Neb65 µg/kgVehicle
IV810Vehicle30 mg/kgcontrol (n = TPIP (n =TPIP (n =TPIP (n =Neb TRENeb TRE TRE (n = 7)
ng/kg/minSelexipagSelexipagcontrol (n = TPIP (n = 9)TPIP (n =TPIP (n =TRE (n = 6) Neb
TRE (nTREng/kg/minSelexipagSelexipag8) 9)11)10)(n = 6)(n = 10)IV TRE (n = 6)(n = 7)(n
= 9)8) 11)10)= 10)IV TRE(n = 7)(n = 9)Data on file, Insmed Inc.* significantly different
from Normoxic control group # significantly different from Vehicle group* and # p <0.05
** and ## p < 0.01 *** and ### p < 0.001 50
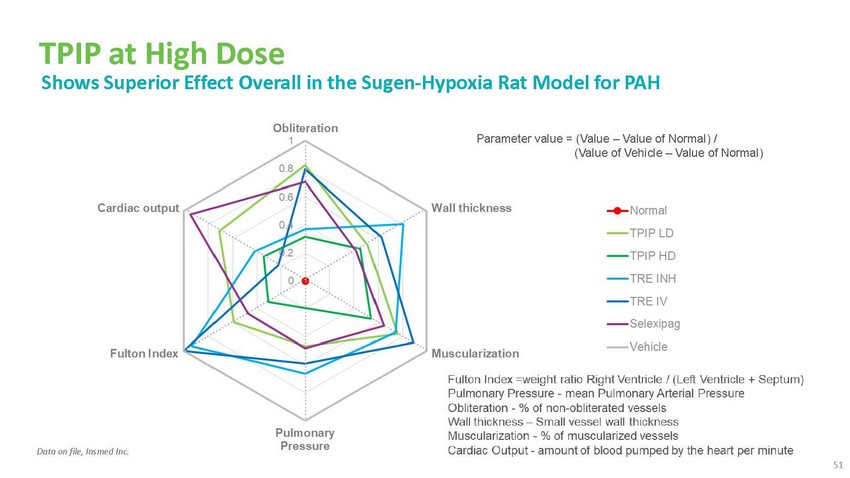
TPIP
at High Dose Shows Superior Effect Overall in the Sugen-Hypoxia Rat Model for PAHParameter
value = (Value – Value of Normal) / (Value of Vehicle – Value of Normal)Data
on file, Insmed Inc. 51

Highlights
of Preclinical Data Preclinical data demonstrate the following features of treprostinil
palmitil:Improved pharmacokinetics• Prolonged lung residence time • Lower systemic
Cmax and prolonged T1/2Prolonged duration of pharmacodynamic activity“Local”
pharmacodynamic effect• Achieves the same pharmacodynamic (vasodilatory) effect
at significantly lower systemic concentrations, compared with IV treprostinilEffect on
hallmark histopathologic features in the sugen/hypoxia model• Remodeling effect,
superior to other prostanoidsReduced incidence of cough (guinea pig model)52
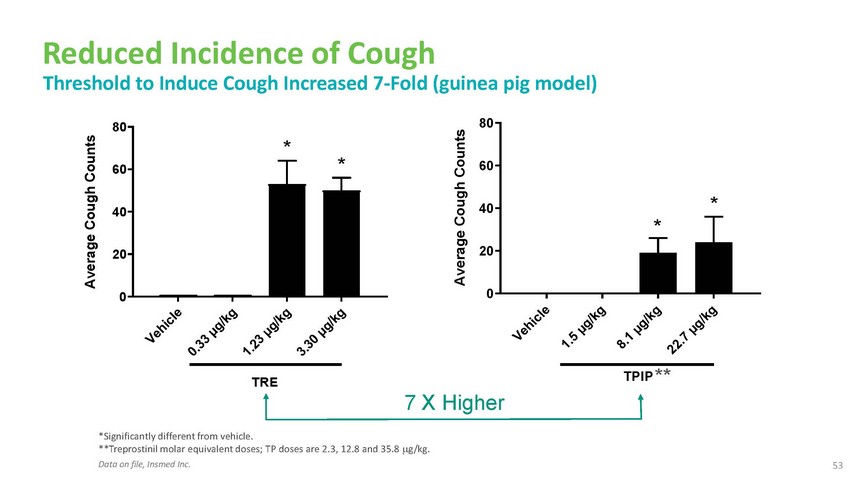
Reduced
Incidence of Cough Threshold to Induce Cough Increased 7-Fold (guinea pig model)80 80
* 60 * 6040 40 * * 20 200 0TRE7 X HigherTPIP ***Significantly different from vehicle.
**Treprostinil molar equivalent doses; TP doses are 2.3, 12.8 and 35.8 g/kg. Data on
file, Insmed Inc.53

Product
Differentiation 54
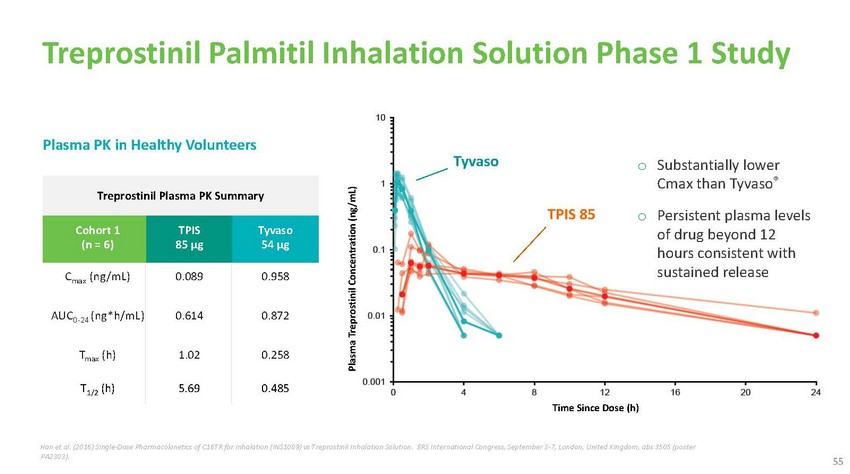
Treprostinil
Palmitil Inhalation Solution Phase 1 StudyPlasma PK in Healthy VolunteersTreprostinil
Plasma PK SummaryTyvasoTPIS 85o Substantially lower Cmax than Tyvaso®o Persistent
plasma levelsCohort 1 (n = 6)TPIS 85 µgTyvaso 54 µgof drug beyond 12 hours
consistent withCmax (ng/mL) 0.089 0.958AUC0-24 (ng*h/mL) 0.614 0.872Tmax (h) 1.02 0.258T1/2
(h) 5.69 0.485Time Since Dose (h)sustained releaseHan et al. (2016) Single-Dose Pharmacokinetics
of C16TR for Inhalation (INS1009) vs Treprostinil Inhalation Solution. ERS International
Congress, September 3-7, London, United Kingdom, abs 3505 (poster PA2303).55

Treprostinil
Palmitil Inhalation Powder (TPIP) a Potentially Differentiated ProstanoidConvenience:
portable, dry powder inhalerIf the characteristics suggested by the preclinical studies
are borne out in the clinical development program, TPIP could represent a differentiated
productQD or BID dosing (prolonged lung residence time, prolonged half life, prolonged
duration of pharmacodynamic activity)Improved safety/tolerability • Systemic tolerability
(lower Cmax) • Local tolerability (reduced cough)Greater vasodilatory pharmacodynamic
effect at lower systemic exposures (sustained lung residence time takes advantage of
the local effect seen in multiple animal models)Potential disease modifier – may
be able to benefit the histopathologic process, whereas Tyvaso, due to transient local
exposure, cannot56

Treprostinil
Palmitil Inhalation Powder (TPIP) a Potentially Differentiated ProstanoidConvenience:
portable, dry powder inhalerIf the characteristics suggested by the preclinical studies
are borne out in the clinical development program, TPIP could represent a differentiated
productQD or BID dosing (prolonged lung residence time, prolonged half life, prolonged
duration of pharmacodynamic activity)Improved safety/tolerability • Systemic tolerability
(lower Cmax) • Local tolerability (reduced cough)Greater vasodilatory pharmacodynamic
effect at lower systemic exposures (sustained lung residence time takes advantage of
the local effect seen in multiple animal models)Potential disease modifier – may
be able to benefit the histopathologic process, whereas Tyvaso, due to transient local
exposure, cannot56
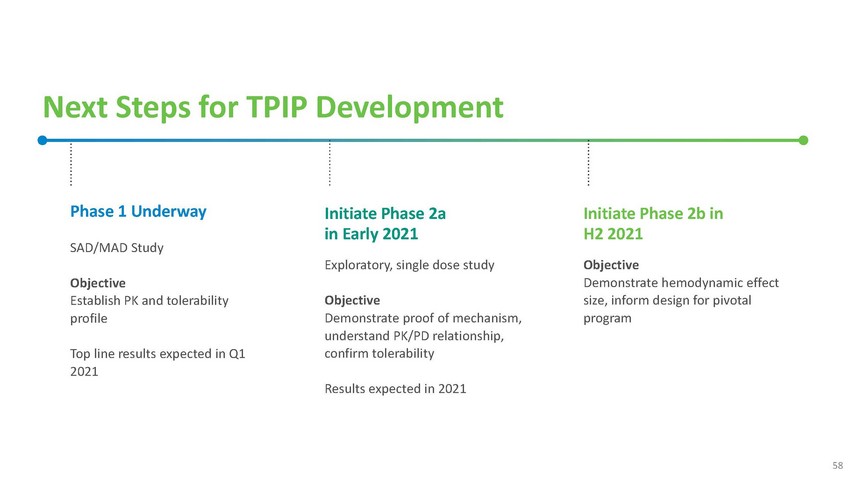
Next
Steps for TPIP DevelopmentPhase 1 UnderwaySAD/MAD StudyObjective Establish PK and tolerability
profileTop line results expected in Q1 2021Initiate Phase 2a in Early 2021Exploratory,
single dose studyObjective Demonstrate proof of mechanism, understand PK/PD relationship,
confirm tolerabilityResults expected in 2021Initiate Phase 2b in H2 2021Objective Demonstrate
hemodynamic effect size, inform design for pivotal program58

Next
Steps for TPIP DevelopmentPhase 1 UnderwaySAD/MAD StudyObjective Establish PK and tolerability
profileTop line results expected in Q1 2021Initiate Phase 2a in Early 2021Exploratory,
single dose studyObjective Demonstrate proof of mechanism, understand PK/PD relationship,
confirm tolerabilityResults expected in 2021Initiate Phase 2b in H2 2021Objective Demonstrate
hemodynamic effect size, inform design for pivotal program58

Brensocatib
NCFBE to Serve as ‘Launch Pad’ for Neutrophil-mediated PortfolioSerious Unmet
Need Conditions• Cystic Fibrosis • Granulomatosis with Polyangiitis •
Apha-1 Antitrypsin DeficiencyNCFBE Program WILLOW + ASPENLarger MarketsResearch Pipeline•
Inflammatory Bowel Disease* • Rheumatoid Arthritis* • Lupus Nephritis* •
COPD/Asthma† • Oncology• External collaborations • Insmed expertiseNCFBE,
non-CF bronchiectasis * Preclinical data generated †AstraZeneca has exercised
its option to develop brensocatib in COPD & asthma through Phase 2b. Further development
subject to agreement on commercial terms with Insmed. 60

Brensocatib
Clinical Development 61
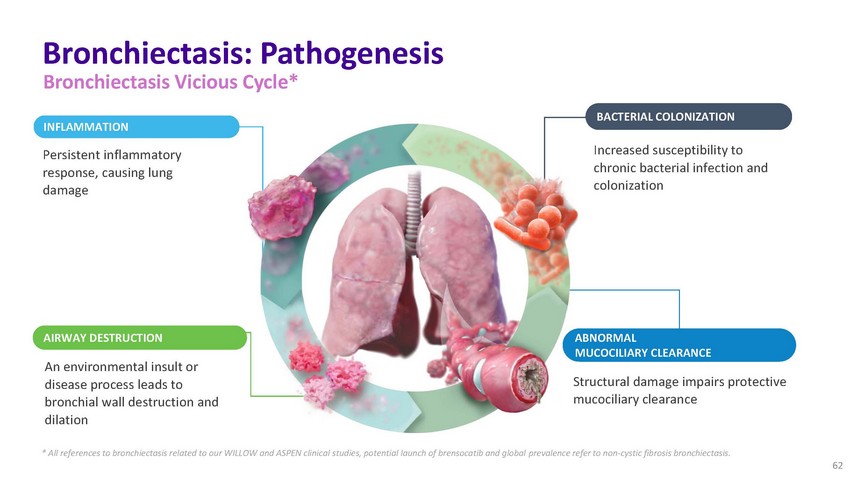
Bronchiectasis:
Pathogenesis Bronchiectasis Vicious Cycle*INFLAMMATIONPersistent inflammatory response,
causing lung damageBACTERIAL COLONIZATIONIncreased susceptibility to chronic bacterial
infection and colonizationAIRWAY DESTRUCTION ABNORMAL MUCOCILIARY CLEARANCEAn environmental
insult or disease process leads to bronchial wall destruction and dilationStructural
damage impairs protective mucociliary clearance* All references to bronchiectasis related
to our WILLOW and ASPEN clinical studies, potential launch of brensocatib and global
prevalence refer to non-cystic fibrosis bronchiectasis. 62

WILLOW
Study Results Underscore Potential of Brensocatib63
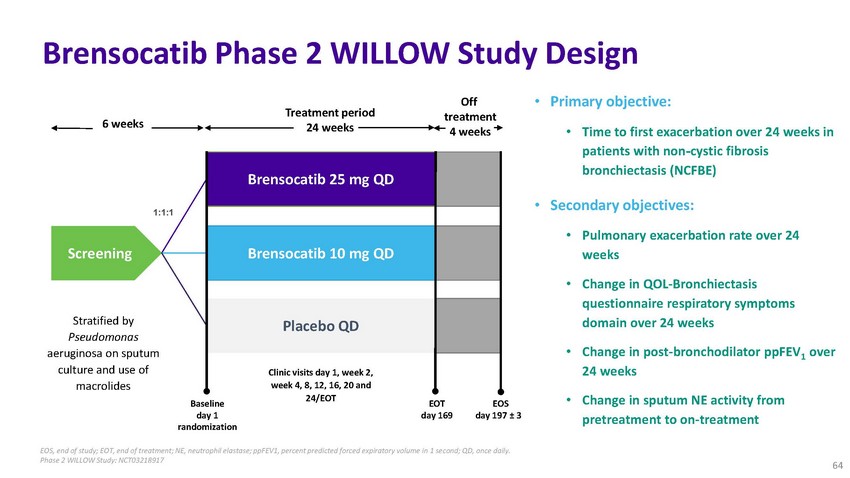
Brensocatib
Phase 2 WILLOW Study Design6 weeksScreeningStratified by Pseudomonas1:1:1Treatment period
24 weeksBrensocatib 25 mg QD Brensocatib 10 mg QD Placebo QDOff treatment 4 weeks•
Primary objective:• Time to first exacerbation over 24 weeks in patients with non-cystic
fibrosis bronchiectasis (NCFBE)• Secondary objectives:• Pulmonary exacerbation
rate over 24 weeks• Change in QOL-Bronchiectasis questionnaire respiratory symptoms
domain over 24 weeksaeruginosa on sputum culture and use of macrolidesBaseline day 1
randomizationClinic visits day 1, week 2, week 4, 8, 12, 16, 20 and 24/EOTEOT day 169EOS
day 197 ± 3• Change in post-bronchodilator ppFEV1 over 24 weeks• Change
in sputum NE activity from pretreatment to on-treatmentEOS, end of study; EOT, end of
treatment; NE, neutrophil elastase; ppFEV1, percent predicted forced expiratory volume
in 1 second; QD, once daily. Phase 2 WILLOW Study: NCT0321891764
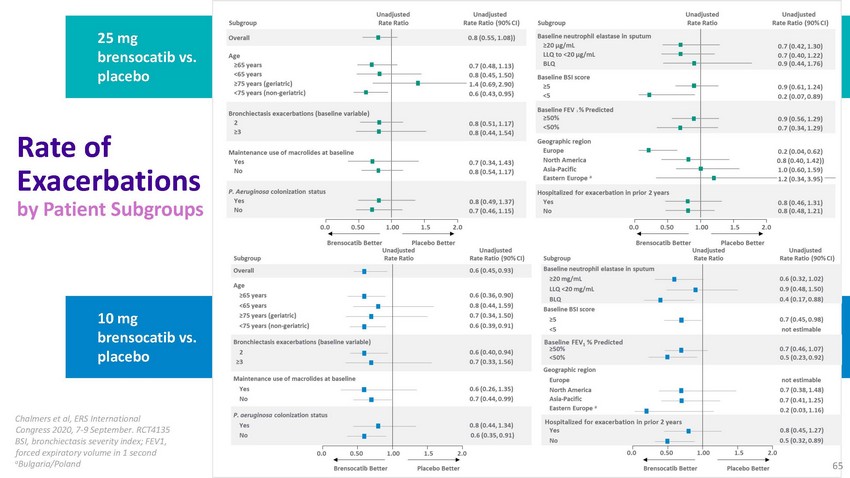
25
mg brensocatib vs. placeboRate of Exacerbations by Patient Subgroups10 mg brensocatib
vs. placeboChalmers et al, ERS International Congress 2020, 7-9 September. RCT4135 BSI,
bronchiectasis severity index; FEV1, forced expiratory volume in 1 second aBulgaria/Poland65
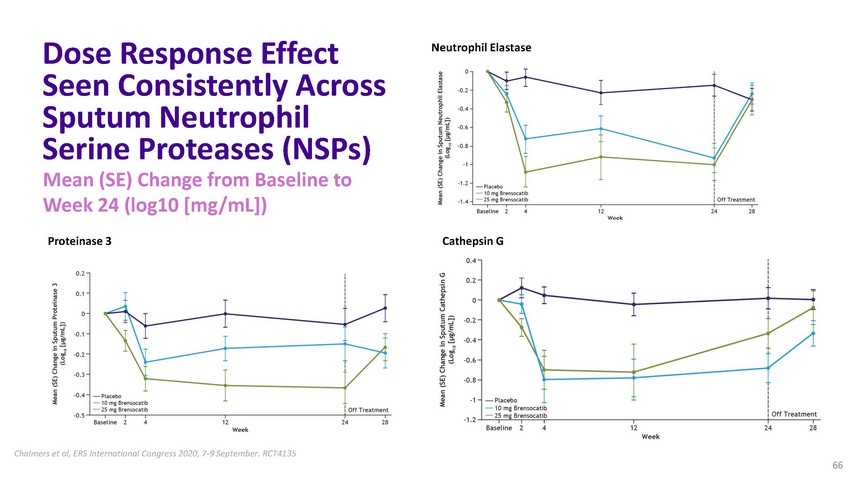
Dose
Response Effect Seen Consistently Across Sputum Neutrophil Serine Proteases (NSPs) Mean
(SE) Change from Baseline to Week 24 (log10 [mg/mL])Neutrophil ElastaseProteinase 3 Cathepsin
GChalmers et al, ERS International Congress 2020, 7-9 September. RCT4135 66

WILLOW
Safety Datano. (%) Placebo (n=85)Brensocatib 10 mg (n=81)Brensocatib 25 mg (n=89)TEAE
resulting in study discontinuation 3 (3.5) 3 (3.7) 4 (5.5) TEAE resulting in treatment
discontinuation 9 (10.6) 6 (7.4) 6 (6.7) Serious TEAE19 (22.4) 11 13.6) 10 (11.2) Serious
TEAEs in ≥ 3% of patients in any groupInfective exacerbation of bronchiectasis 9 (10.6)
5 (6.2) 4 (4.5) Pneumonia3 (3.5) 0 4 (4.5) Any TEAE 67 (78.8) 75 (92.6) 74 (83.1)TEAEs
in ≥ 10% of patients in any groupCough 10 (11.8) 15 (18.5) 12 (13.5) Headache3 (3.5)
8 (9.9) 12 (13.5) Sputum increased6 (7.1) 9 (11.1) 9 (10.1) Dyspnea2 (2.4) 3 (3.7) 9
(10.1) Infective exacerbation of bronchiectasis 9 (10.6) 5 (6.2) 4 (4.5)Diarrhea 9 (10.6)
5 (6.2) 3 (3.4) 67

Brensocatib
Phase 3 Development Program 68
ASPEN
Study: Single Trial Designed to Support Global FilingBreakthrough Therapy Designation
for brensocatib in non-cystic fibrosis bronchiectasis (NCFBE)Robust efficacy endpoints,
supported by WILLOW resultsIntegrates feedback from Health Authorities69
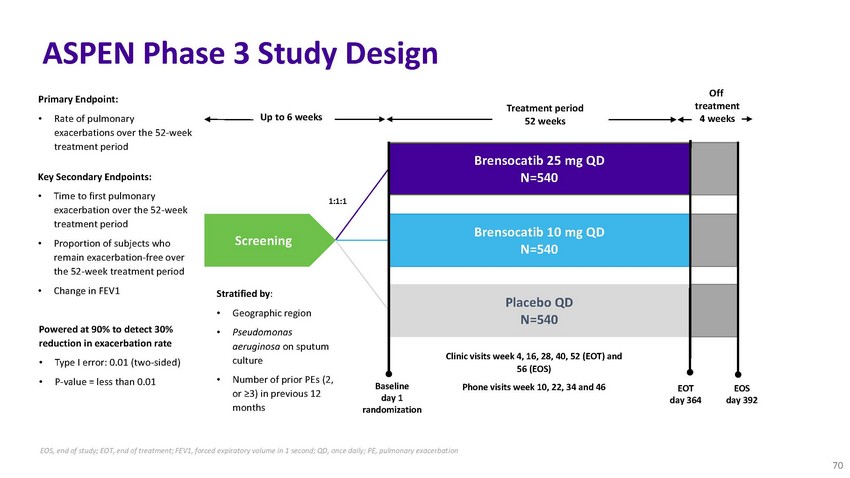
ASPEN
Phase 3 Study DesignPrimary Endpoint:Treatment periodOff treatment• Rate of pulmonary
exacerbations over the 52-week treatment periodKey Secondary Endpoints:• Time to
first pulmonary exacerbation over the 52-week treatment period• Proportion of subjects
who remain exacerbation-free over the 52-week treatment period• Change in FEV1Powered
at 90% to detect 30%Up to 6 weeksScreeningStratified by:• Geographic region•
Pseudomonas1:1:152 weeksBrensocatib 25 mg QD N=540Brensocatib 10 mg QD N=540Placebo QD
N=5404 weeksreduction in exacerbation rate• Type I error: 0.01 (two-sided)•
P-value = less than 0.01aeruginosa on sputum culture• Number of prior PEs (2, or
≥3) in previous 12 monthsBaseline day 1 randomizationClinic visits week 4, 16, 28,
40, 52 (EOT) and 56 (EOS)Phone visits week 10, 22, 34 and 46EOT day 364EOS day 392EOS,
end of study; EOT, end of treatment; FEV1, forced expiratory volume in 1 second; QD,
once daily; PE, pulmonary exacerbation 70

ASPEN
Timeline Considerations36 Countries~480 sites6 Fast Track CountriesTargeting WILLOW study
sites71

Brensocatib
Commercial Outlook 72

Brensocatib
Strategy Builds on Successful First Launch in NCFBEN Phase 3 trial for non-cystic fibrosis
onchiectasis (NCFBE)Prepare for successful global launch in NCFBEAdvance clinical programs
for cystic fibrosisaluate other neutrophil-modulated eases for future research &
label xpansion73

An
Approved NCFBE Treatment to Reduce ExacerbationsCould Address Unmet Needs Across StakeholdersPatients
HealthcarePayers“It’s like waiting for• Symptoms of cough, dyspnea,
fatigue interfere with daily life• Fear of exacerbations negatively impacts emotional
and social well-being• Frustrated with time- consuming therapies that are not specific
to BEProviders• Lack of efficacious anti- inflammatory option• Treatments that
improve patient QOL and prevent exacerbations seen as largest unmet need• Recognize
current treatments lack BE-specific clinical trial data• Treatment option with BE
indication, greater efficacy, and reduction in exacerbations seen as unmet needsa hurricane
– there’s a constant level of fear and anxiety of the unknown. When will
an exacerbation hit? - BRONCHIECTASIS PATIE”NT74
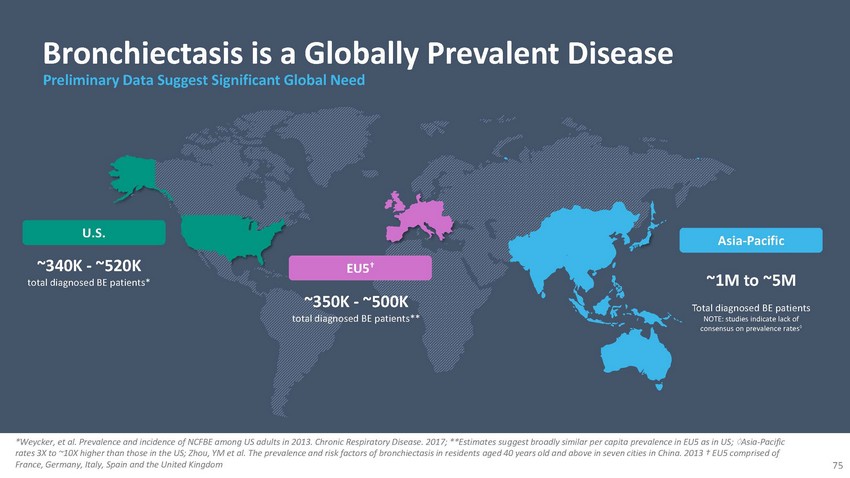
Bronchiectasis
is a Globally Prevalent Disease
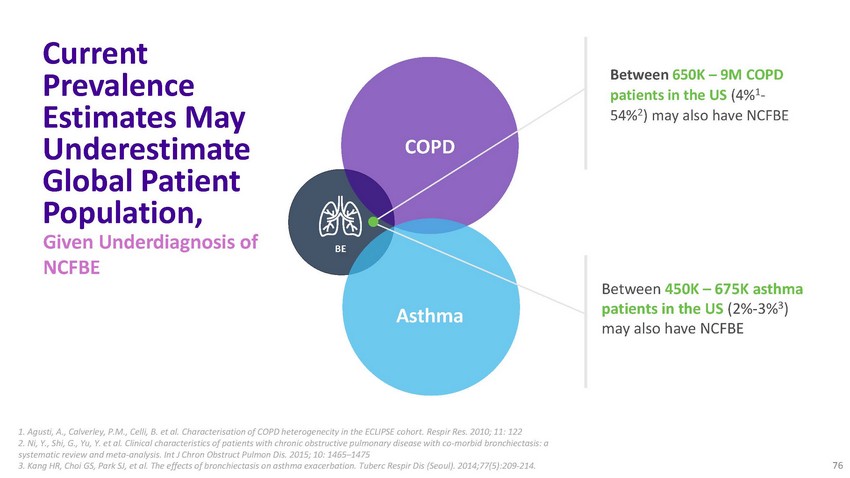
Current
Prevalence Estimates May Underestimate Global Patient Population, Given Underdiagnosis
of NCFBECOPDBEAsthmaBetween 650K – 9M COPD patients in the US (4%1- 54%2) may also
have NCFBEBetween 450K – 675K asthma patients in the US (2%-3%3) may also have
NCFBE1. Agusti, A., Calverley, P.M., Celli, B. et al. Characterisation of COPD heterogenecity
in the ECLIPSE cohort. Respir Res. 2010; 11: 122 2. Ni, Y., Shi, G., Yu, Y. et al. Clinical
characteristics of patients with chronic obstructive pulmonary disease with co-morbid
bronchiectasis: a systematic review and meta-analysis. Int J Chron Obstruct Pulmon Dis.
2015; 10: 1465–1475 3. Kang HR, Choi GS, Park SJ, et al. The effects of bronchiectasis
on asthma exacerbation. Tuberc Respir Dis (Seoul). 2014;77(5):209‐214. 76
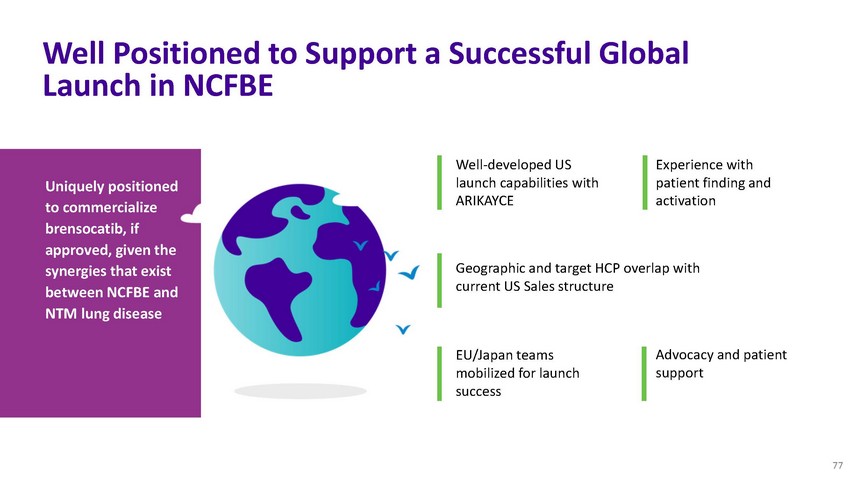
Well
Positioned to Support a Successful Global Launch in NCFBEUniquely positioned to commercialize
brensocatib, if approved, given theWell-developed US launch capabilities with ARIKAYCEExperience
with patient finding and activationsynergies that exist between NCFBE and NTM lung diseaseGeographic
and target HCP overlap with current US Sales structureEU/Japan teams mobilized for launch
successAdvocacy and patient support77

Brensocatib
Additional Opportunities 78
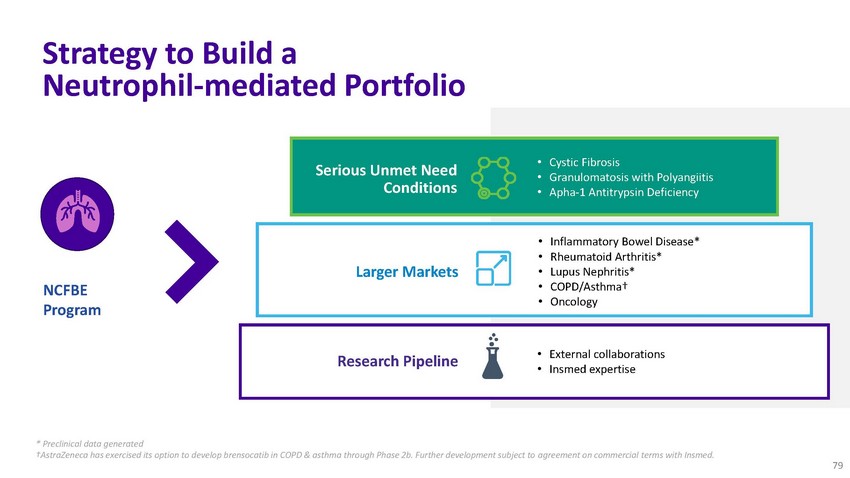
Strategy
to Build a Neutrophil-mediated PortfolioSerious Unmet Need Conditions• Cystic Fibrosis
• Granulomatosis with Polyangiitis • Apha-1 Antitrypsin DeficiencyNCFBE ProgramLarger
MarketsResearch Pipeline• Inflammatory Bowel Disease* • Rheumatoid Arthritis*
• Lupus Nephritis* • COPD/Asthma† • Oncology• External collaborations
• Insmed expertise* Preclinical data generated †AstraZeneca has exercised
its option to develop brensocatib in COPD & asthma through Phase 2b. Further development
subject to agreement on commercial terms with Insmed. 79
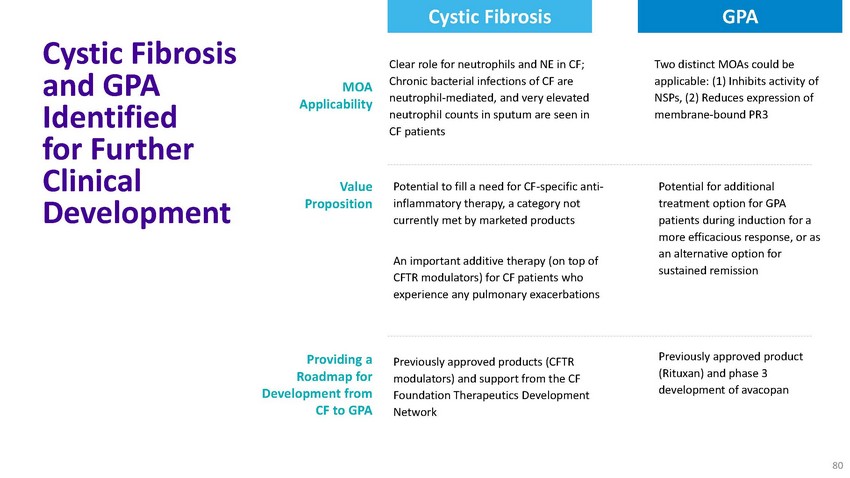
Cystic
Fibrosis GPACystic Fibrosis and GPA Identified for Further Clinical DevelopmentMOA ApplicabilityValue
PropositionProviding a Roadmap for Development from CF to GPAClear role for neutrophils
and NE in CF; Chronic bacterial infections of CF are neutrophil-mediated, and very elevated
neutrophil counts in sputum are seen in CF patientsPotential to fill a need for CF-specific
anti- inflammatory therapy, a category not currently met by marketed productsAn important
additive therapy (on top of CFTR modulators) for CF patients who experience any pulmonary
exacerbationsPreviously approved products (CFTR modulators) and support from the CF Foundation
Therapeutics Development NetworkTwo distinct MOAs could be applicable: (1) Inhibits activity
of NSPs, (2) Reduces expression of membrane-bound PR3Potential for additional treatment
option for GPA patients during induction for a more efficacious response, or as an alternative
option for sustained remissionPreviously approved product (Rituxan) and phase 3 development
of avacopan80

Ongoing
Investigator-Sponsored Clinical Study for Treatment of Severe COVID-19 InfectionsCould
brensocatib be used to treat COVID-19?Currently under investigation in multi-site UK
investigator-initiated research (IIR) studyDouble-blind, prospective, placebo-controlled,
randomized trialUp to 300 patients; 10 sitesLed by University of Dundee’s Professor
James Chalmers, MBChB, Ph.D.Prioritized by UK’s National Institute for Health ResearchPotential
Data read-out: H1 202181

Kevin
C. Mange, M.D., MSCE SVP, Clinical DevelopmentNeil Hughes General Manager, Head of EMEARoger
Adsett Chief Operating OfficerARIKAYCENot for promotional use 82

Kevin
C. Mange, M.D., MSCE SVP, Clinical DevelopmentARIKAYCEFront Line Program 83 Not for promotional
use 83

ARIKAYCE®
(amikacin liposomal inhalation suspension) Indication and Use• LIMITED POPULATION:
ARIKAYCE is indicated in adults, who have limited or no alternative treatment options,
for the treatment of Mycobacterium avium complex (MAC) lung disease as part of a combination
antibacterial drug regimen in patients who do not achieve negative sputum cultures after
a minimum of 6 consecutive months of a multidrug background regimen therapy. As only
limited clinical safety and effectiveness data for ARIKAYCE are currently available,
reserve ARIKAYCE for use in adults who have limited or no alternative treatment options.
This drug is indicated for use in a limited and specific population of patients.•
This indication is approved under accelerated approval based on achieving sputum culture
conversion (defined as 3 consecutive negative monthly sputum cultures) by Month 6. Clinical
benefit has not yet been established. Continued approval for this indication may be contingent
upon verification and description of clinical benefit in confirmatory trials.• Limitation
of Use: ARIKAYCE has only been studied in patients with refractory MAC lung disease defined
as patients who did not achieve negative sputum cultures after a minimum of 6 consecutive
months of a multidrug background regimen therapy. The use of ARIKAYCE is not recommended
for patients with non-refractory MAC lung disease.WARNING: RISK OF INCREASED RESPIRATORY
ADVERSE REACTIONS ARIKAYCE has been associated with an increased risk of respiratory
adverse reactions, including hypersensitivity pneumonitis, hemoptysis, bronchospasm,
and exacerbation of underlying pulmonary disease that have led to hospitalizations in
some cases.Not for promotional use 84
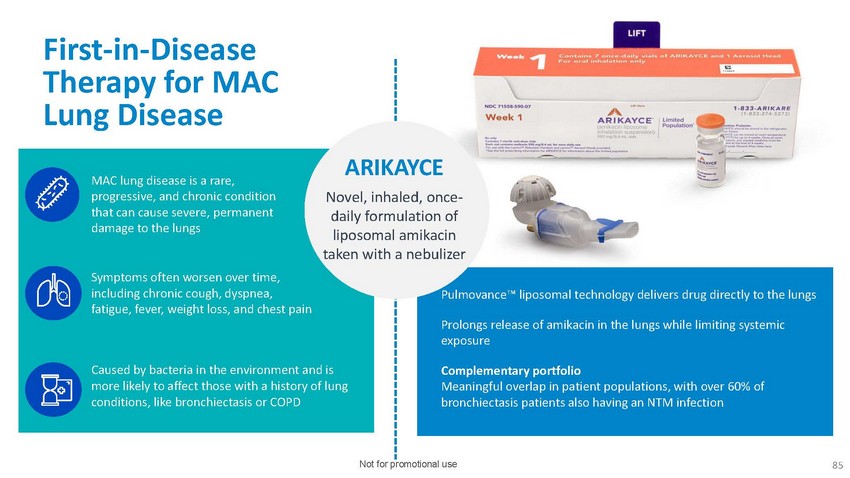
First-in-Disease
Therapy for MAC Lung DiseaseMAC lung disease is a rare, progressive, and chronic condition
that can cause severe, permanent damage to the lungsSymptoms often worsen over time,
including chronic cough, dyspnea, fatigue, fever, weight loss, and chest painARIKAYCENovel,
inhaled, once- daily formulation of liposomal amikacin taken with a nebulizerce™
liposomal technology delivers drug directly to the lungsProlongs release of amikacin
in the lungs while limiting systemic exposureCaused by bacteria in the environment and
is more likely to affect those with a history of lung conditions, like bronchiectasis
or COPDComplementary portfolio Meaningful overlap in patient populations, with over 60%
of bronchiectasis patients also having an NTM infectionNot for promotional use 85
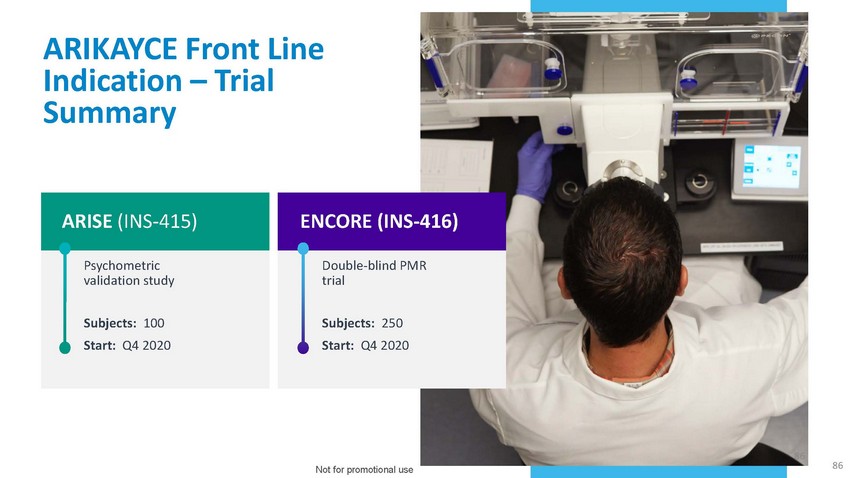
ARIKAYCE
Front Line Indication – Trial SummaryARISE (INS-415)Psychometric validation studySubjects:
100Start: Q4 2020ENCORE (INS-416)Double-blind PMR trialSubjects: 250Start: Q4 202086
Not for promotional use 86
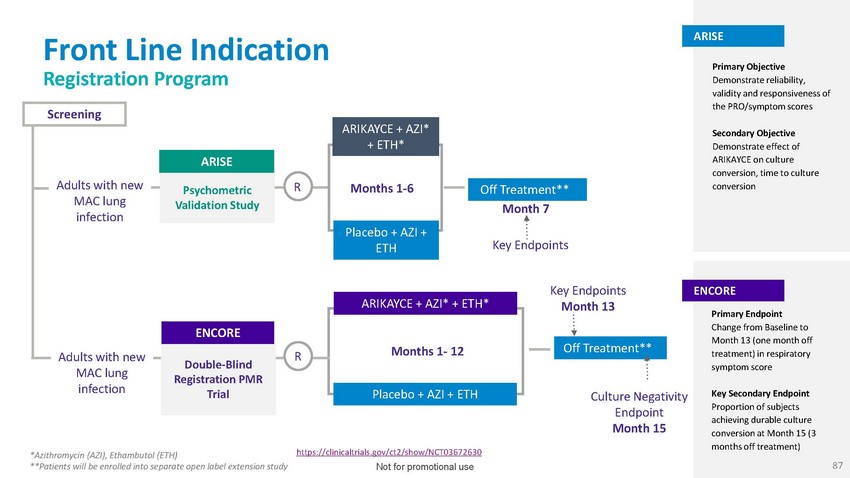
Front
Line Indication Registration ProgramScreeningARISEARIKAYCE + AZI* + ETH*ARISEPrimary
Objective Demonstrate reliability, validity and responsiveness of the PRO/symptom scoresSecondary
Objective Demonstrate effect of ARIKAYCE on culture conversion, time to cultureAdults
with newPsychometricR Months 1-6Off Treatment**conversionMAC lung infectionValidation
Study Month 7Placebo + AZI +ETHKey EndpointsAdults with new MAC lung infectionENCOREDouble-Blind
R Registration PMR TrialARIKAYCE + AZI* + ETH*Months 1- 12Placebo + AZI + ETHKey Endpoints
Month 13Off Treatment**Culture Negativity Endpoint Month 15ENCOREPrimary Endpoint Change
from Baseline to Month 13 (one month off treatment) in respiratory symptom scoreKey Secondary
Endpoint Proportion of subjects achieving durable culture conversion at Month 15 (3 months
off treatment)*Azithromycin (AZI), Ethambutol (ETH) **Patients will be enrolled into
separate open label extension studyhttps://clinicaltrials.gov/ct2/show/NCT03672630 Not
for promotional use 87
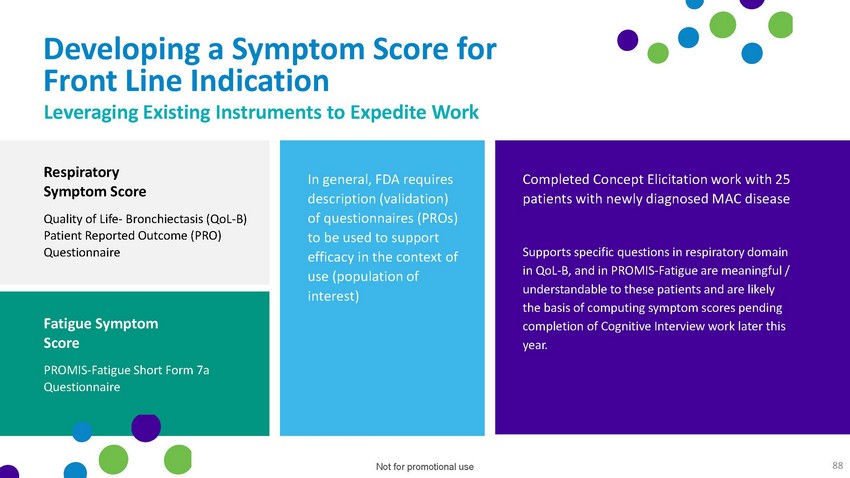
Developing
a Symptom Score for Front Line Indication Leveraging Existing Instruments to Expedite
WorkRespiratory Symptom ScoreQuality of Life- Bronchiectasis (QoL-B) Patient Reported
Outcome (PRO) QuestionnaireFatigue Symptom ScorePROMIS-Fatigue Short Form 7a QuestionnaireIn
general, FDA requires description (validation) of questionnaires (PROs) to be used to
support efficacy in the context of use (population of interest)Completed Concept Elicitation
work with 25 patients with newly diagnosed MAC diseaseSupports specific questions in
respiratory domain in QoL-B, and in PROMIS-Fatigue are meaningful / understandable to
these patients and are likely the basis of computing symptom scores pending completion
of Cognitive Interview work later this year.Not for promotional use 88
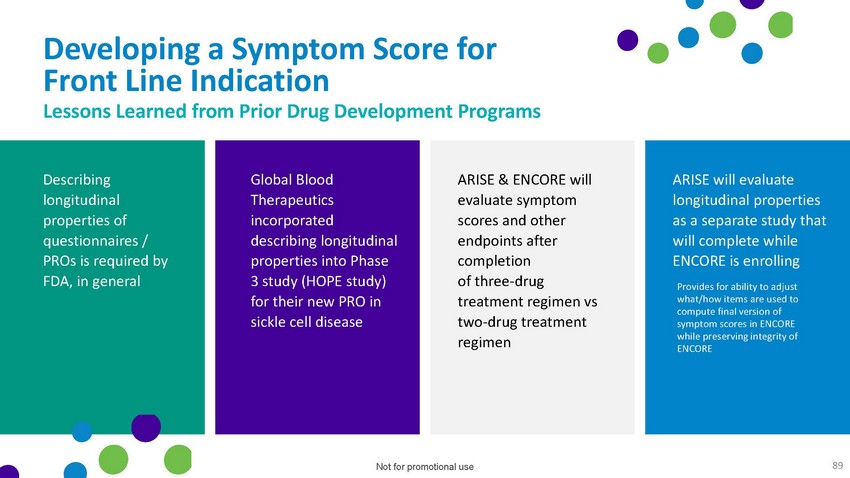
Developing
a Symptom Score for Front Line Indication Lessons Learned from Prior Drug Development
ProgramsDescribing longitudinal properties of questionnaires / PROs is required by FDA,
in generalGlobal Blood Therapeutics incorporated describing longitudinal properties into
Phase 3 study (HOPE study) for their new PRO in sickle cell diseaseARISE & ENCORE
will evaluate symptom scores and other endpoints after completion of three-drug treatment
regimen vs two-drug treatment regimenARISE will evaluate longitudinal properties as a
separate study that will complete while ENCORE is enrolling• Provides for ability
to adjust what/how items are used to compute final version of symptom scores in ENCORE
while preserving integrity of ENCORENot for promotional use 89

Neil
Hughes General Manager, Head of EMEAARIKAYCEEU Launch UpdateNot for promotional use 90

ARIKAYCE
a Blueprint for Future Success2019 $136M net salesQ2 2020 $42.5MEngaged HCP & patient
populationPositive reimbursement trends• Engaged healthcare providers• Educated
patient population• Focus on payers to ensure reimbursementNot for promotional useARIKAYCE
was one of the Top 10 most successful non-oncology rare disease launchesWe plan to repeat
history with our growing pipeline91

ARIKAYCE
Added to International Treatment Guidelines for Treatment of MAC Lung DiseaseGuidelines
are a globally recognized standard for the prevention, diagnosis and treatment of NTM
lung diseaseAddition of ARIKAYCE to new standard treatment regimen for adult patients
with limited or no treatment options who failed to convert to a negative sputum culture
after at least six months of treatmentTreatment should be continued for 12 months after
culture conversionNot for promotional use 92

ARIKAYCE
Added to International Treatment Guidelines for Treatment of MAC Lung DiseaseGuidelines
are a globally recognized standard for the prevention, diagnosis and treatment of NTM
lung diseaseAddition of ARIKAYCE to new standard treatment regimen for adult patients
with limited or no treatment options who failed to convert to a negative sputum culture
after at least six months of treatmentTreatment should be continued for 12 months after
culture conversionNot for promotional use 92
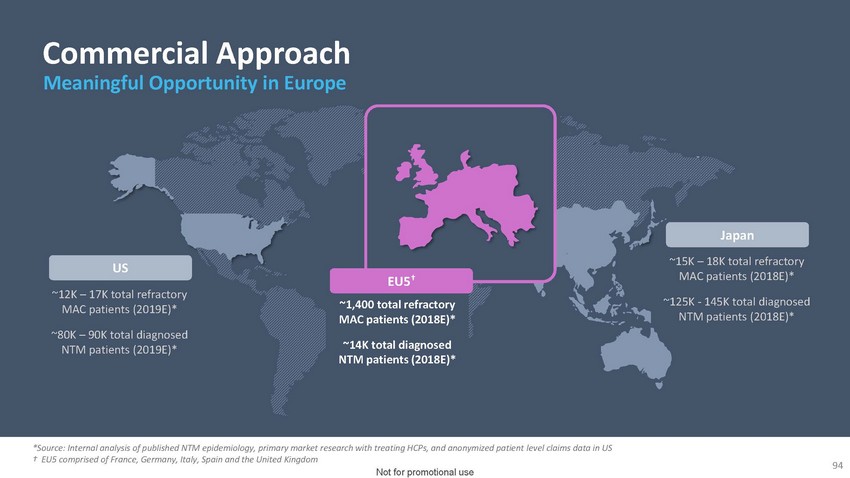
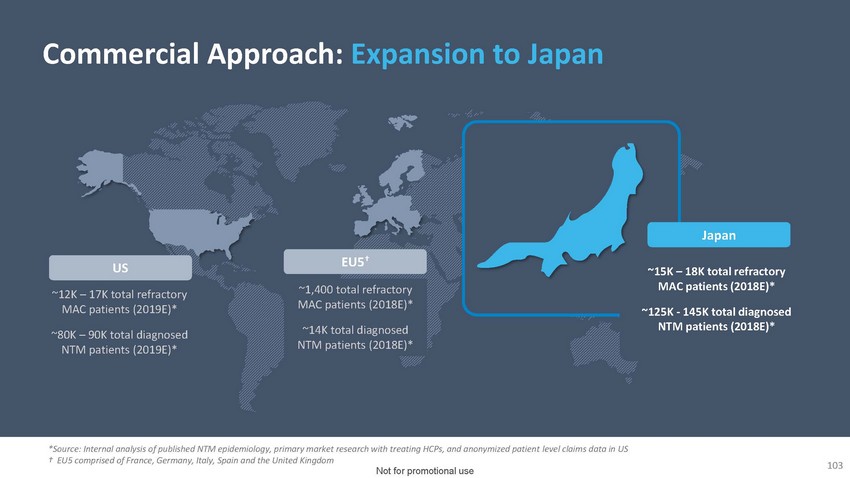
Commercial
Approach Meaningful Opportunity in EuropeUS~12K – 17K total refractory MAC patients
(2019E)*~80K – 90K total diagnosed NTM patients (2019E)*EU5†~1,400 total
refractory MAC patients (2018E)*~14K total diagnosed NTM patients (2018E)*Japan~15K –
18K total refractory MAC patients (2018E)*~125K - 145K total diagnosed NTM patients (2018E)**Source:
Internal analysis of published NTM epidemiology, primary market research with treating
HCPs, and anonymized patient level claims data in US † EU5 comprised of France,
Germany, Italy, Spain and the United Kingdom94 Not for promotional use

ARIKAYCE
Received Positive CHMP Opinion July 24, 2020Committee for Medicinal Products for Human
Use (CHMP)of the European Medicines Agency has adopted a positive opinion recommending
ARIKAYCE for the treatment of NTM lung infections caused by MAC in non- CF Patients with
limited treatment optionsNot for promotional use 95

Prevalence
of NTM in EU May be Greater Than Previously Reported…while anticipated EU indication
slightly wider than only refractory MACNot for promotional use 96
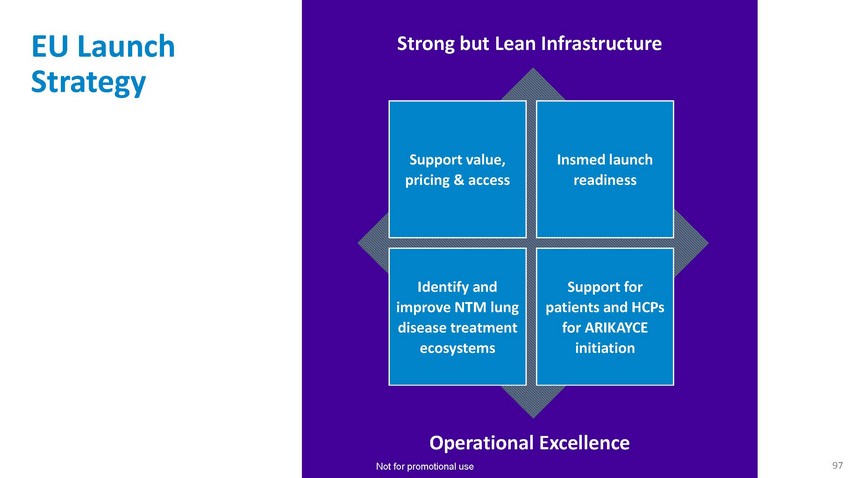
EU
Launch StrategyStrong but Lean InfrastructureSupport value, pricing & accessInsmed
launch readinessIdentify and improve NTM lung disease treatment ecosystemsSupport for
patients and HCPs for ARIKAYCE initiationOperational Excellence Not for promotional use
97

Insmed
Projected EMEA Country Footprint Specific Field Force Structures Reflect Country- Specific
Patient Pathways and NTM Management ProtocolsCountry KeyRegional HQ – No Local
Commercial OrgInsmed Commercial OrganizationUnder Review - Insmed vs DistributorCustomer
facing FTEs grow as country reimbursement comes online*FTEs 2020 2021 2022Field Based
Medical 14 18 21Field Based Commercial 10 21 29*ProjectionsNot for promotional use 98

Insmed
Projected EMEA Country Footprint Specific Field Force Structures Reflect Country- Specific
Patient Pathways and NTM Management ProtocolsCountry KeyRegional HQ – No Local
Commercial OrgInsmed Commercial OrganizationUnder Review - Insmed vs DistributorCustomer
facing FTEs grow as country reimbursement comes online*FTEs 2020 2021 2022Field Based
Medical 14 18 21Field Based Commercial 10 21 29*ProjectionsNot for promotional use 98
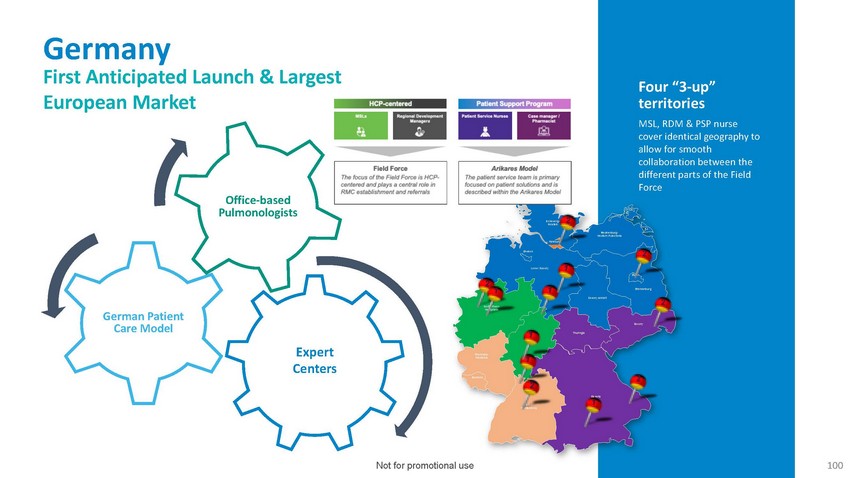
Germany
First Anticipated Launch & Largest European MarketFour “3-up” territories
MSL, RDM & PSP nurse cover identical geography to allow for smooth collaboration
between the different parts of the FieldOffice-based PulmonologistsSchleswig- 2 HolsteinMecklenburg-
Western PomeraniaBremenHamburg2Lower Saxony 12 1 1Saxony-AnhaltBerlinBrandenburg2German
Patient Care ModelExpert CentersNorth Rhein- WestphaliaRheinland- PalatinateSaarland1
Hesse12ThuringiaSaxony2Baden- WürttembergBavaria 1Not for promotional use100
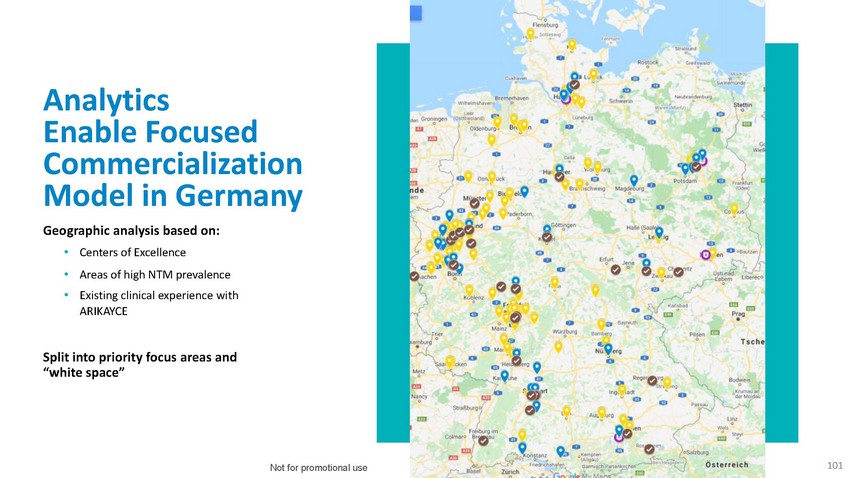
Analytics
Enable Focused Commercialization Model in GermanyGeographic analysis based on:•
Centers of Excellence• Areas of high NTM prevalence• Existing clinical experience
with ARIKAYCESplit into priority focus areas and “white space”Not for promotional
use101

Roger
Adsett Chief Operating OfficerARIKAYCEJapan Launch UpdateNot for promotional use10210
Commercial
Approach: Expansion to Japan
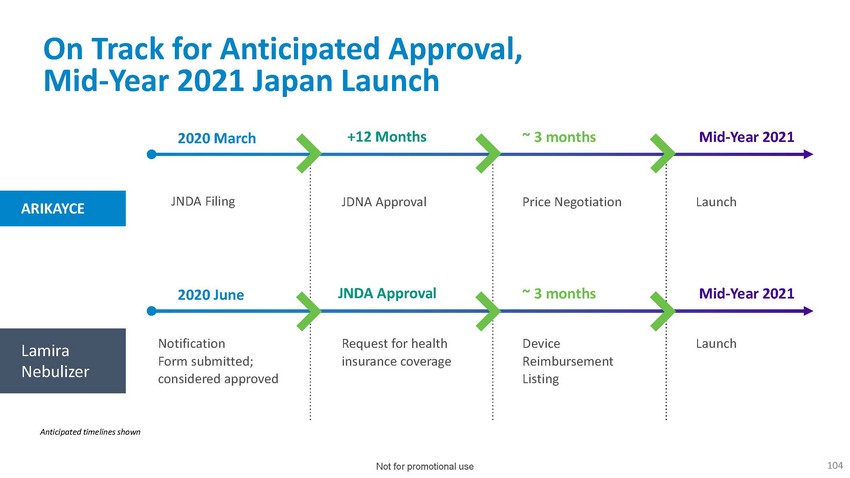
On
Track for Anticipated Approval, Mid-Year 2021 Japan Launch2020 March +12 Months ~ 3 months
Mid-Year 2021ARIKAYCEJNDA FilingJDNA Approval Price Negotiation Launch2020 June JNDA
Approval ~ 3 months Mid-Year 2021Lamira NebulizerNotification Form submitted; considered
approvedRequest for health insurance coverageDevice Reimbursement ListingLaunchAnticipated
timelines shownNot for promotional use104
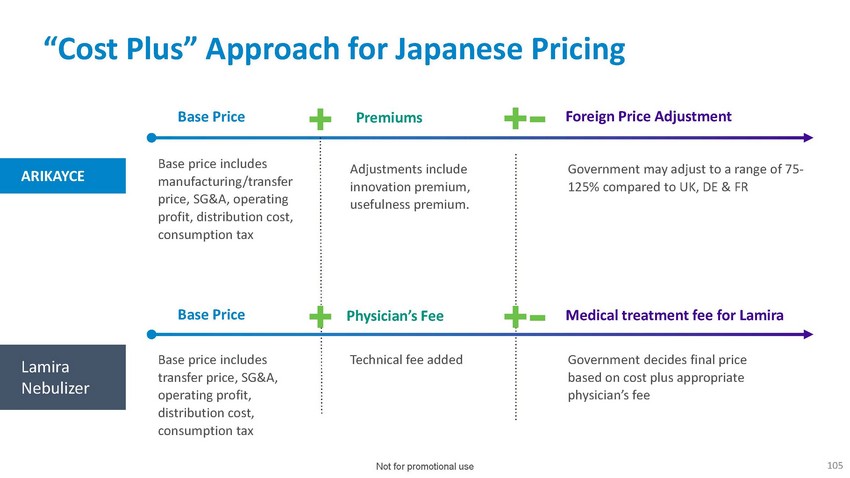
“Cost
Plus” Approach for Japanese PricingBase Price Premiums Foreign Price AdjustmentARIKAYCEBase
price includes manufacturing/transfer price, SG&A, operating profit, distribution
cost, consumption taxAdjustments include innovation premium, usefulness premium.Government
may adjust to a range of 75- 125% compared to UK, DE & FRBase Price Physician’s
Fee Medical treatment fee for LamiraLamira NebulizerBase price includes transfer price,
SG&A, operating profit, distribution cost, consumption taxTechnical fee added Government
decides final price based on cost plus appropriate physician’s feeNot for promotional
use105

Nuances
of Japanese MarketPrescriptions are limited to two-week supply for the first year drug
is on the marketCo-pay market: approved drugs reimbursed at fixed ratesPhysician offices
would exclusively train patients on use of ARIKAYCE with Lamira deviceNot for promotional
use106

Building
a Strong Presence in Japan 28 People with Japanese AffiliateNot for promotional use107

Engaging
Major Medical AssociationsNot for promotional use108
Sales
Territories in Japan*Greater OsakaGreater TokyoWest;;;. 7 Reps EAST;;;;. 8 Reps*Jf approvedNot
for promotional use 109

Sara
Bonstein Chief Financial OfficerFinancial Update & Closing Remarks110
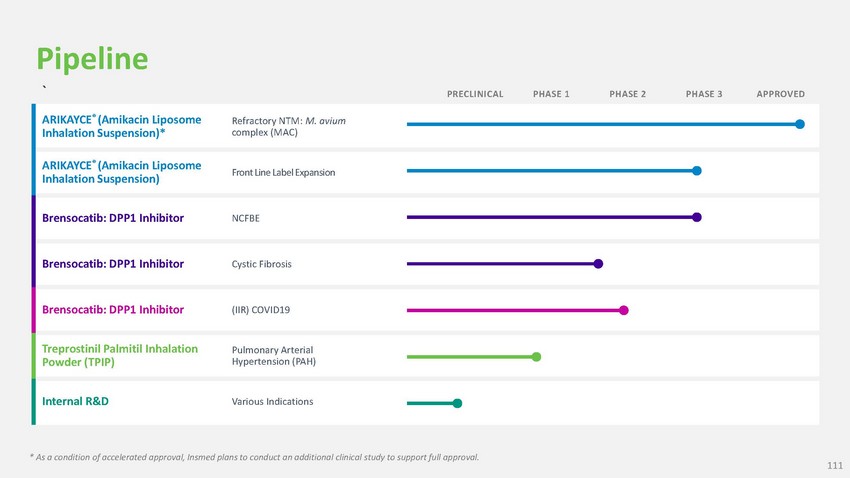
Pipeline
` PRECLINICAL PHASE 1 PHASE 2 PHASE 3 APPROVEDARIKAYCE® (Amikacin Liposome Inhalation
Suspension)*Refractory NTM: M. avium complex (MAC)ARIKAYCE® (Amikacin Liposome Inhalation
Suspension) Front Line Label ExpansionBrensocatib: DPP1 Inhibitor NCFBE Brensocatib:
DPP1 Inhibitor Cystic Fibrosis Brensocatib: DPP1 Inhibitor (IIR) COVID19Treprostinil
Palmitil Inhalation Powder (TPIP)Pulmonary Arterial Hypertension (PAH)Internal R&D
Various Indications* As a condition of accelerated approval, Insmed plans to conduct
an additional clinical study to support full approval.111

Financial
Strength to Achieve Our Objectives$641.9M as of 6/30/20Cash and Cash Equivalents>3
Years as of 9/30/20101M* as of 6/30/20Estimated Cash RunwayCommon Shares Outstanding*Excludes
stock options, unvested RSUs and shares underlying outstanding convertible notes112
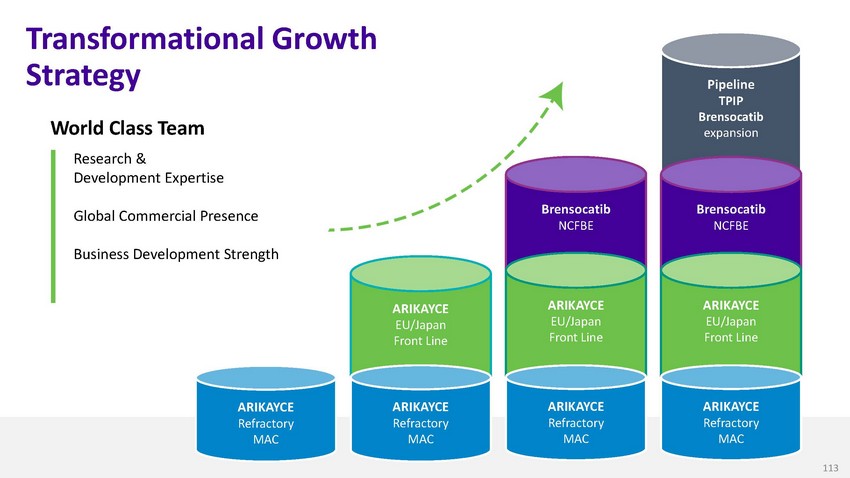
Transformational
Growth StrategyWorld Class TeamResearch & Development ExpertiseGlobal Commercial
PresenceBusiness Development StrengthBrensocatib NCFBEPipeline TPIP Brensocatib expansionBrensocatib
NCFBEARIKAYCE EU/Japan Front LineARIKAYCE EU/Japan Front LineARIKAYCE EU/Japan Front
LineARIKAYCE Refractory MACARIKAYCE Refractory MACARIKAYCE Refractory MACARIKAYCE Refractory
MAC113

Questions
& Answers114114

We
are committed to bringing forth technologies and medicines in therapeutic areas with
the greatest potential to make a difference in patients’ livesThank YouInvestor
Relations investor.relations@insmed.com 646-351-0954115





















































































































