false2021FY0001016169P5YP2MP3M00010161692021-01-012021-12-3100010161692021-06-30iso4217:USD00010161692022-02-28xbrli:shares00010161692021-12-3100010161692020-12-31iso4217:USDxbrli:shares0001016169us-gaap:ProductMember2021-01-012021-12-310001016169us-gaap:ProductMember2020-01-012020-12-310001016169us-gaap:ProductMember2019-01-012019-12-310001016169atrs:LicensingAndDevelopmentRevenueMember2021-01-012021-12-310001016169atrs:LicensingAndDevelopmentRevenueMember2020-01-012020-12-310001016169atrs:LicensingAndDevelopmentRevenueMember2019-01-012019-12-310001016169us-gaap:RoyaltyMember2021-01-012021-12-310001016169us-gaap:RoyaltyMember2020-01-012020-12-310001016169us-gaap:RoyaltyMember2019-01-012019-12-3100010161692020-01-012020-12-3100010161692019-01-012019-12-310001016169us-gaap:CommonStockMember2018-12-310001016169us-gaap:AdditionalPaidInCapitalMember2018-12-310001016169us-gaap:RetainedEarningsMember2018-12-310001016169us-gaap:AccumulatedOtherComprehensiveIncomeMember2018-12-3100010161692018-12-310001016169us-gaap:CommonStockMember2019-01-012019-12-310001016169us-gaap:AdditionalPaidInCapitalMember2019-01-012019-12-310001016169us-gaap:RetainedEarningsMember2019-01-012019-12-310001016169us-gaap:AccumulatedOtherComprehensiveIncomeMember2019-01-012019-12-310001016169us-gaap:CommonStockMember2019-12-310001016169us-gaap:AdditionalPaidInCapitalMember2019-12-310001016169us-gaap:RetainedEarningsMember2019-12-310001016169us-gaap:AccumulatedOtherComprehensiveIncomeMember2019-12-3100010161692019-12-310001016169us-gaap:CommonStockMember2020-01-012020-12-310001016169us-gaap:AdditionalPaidInCapitalMember2020-01-012020-12-310001016169us-gaap:RetainedEarningsMember2020-01-012020-12-310001016169us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-01-012020-12-310001016169us-gaap:CommonStockMember2020-12-310001016169us-gaap:AdditionalPaidInCapitalMember2020-12-310001016169us-gaap:RetainedEarningsMember2020-12-310001016169us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-12-310001016169us-gaap:CommonStockMember2021-01-012021-12-310001016169us-gaap:AdditionalPaidInCapitalMember2021-01-012021-12-310001016169us-gaap:RetainedEarningsMember2021-01-012021-12-310001016169us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-01-012021-12-310001016169us-gaap:CommonStockMember2021-12-310001016169us-gaap:AdditionalPaidInCapitalMember2021-12-310001016169us-gaap:RetainedEarningsMember2021-12-310001016169us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-12-31atrs:subsidiary0001016169us-gaap:FairValueInputsLevel1Member2021-12-310001016169us-gaap:FairValueInputsLevel1Member2020-12-310001016169srt:MinimumMember2021-01-012021-12-310001016169srt:MaximumMember2021-01-012021-12-310001016169atrs:ComputerEquipmentAndSoftwareMembersrt:MinimumMember2021-01-012021-12-310001016169atrs:ComputerEquipmentAndSoftwareMembersrt:MaximumMember2021-01-012021-12-310001016169srt:MinimumMemberatrs:FurnitureFixturesAndOfficeEquipmentMember2021-01-012021-12-310001016169srt:MaximumMemberatrs:FurnitureFixturesAndOfficeEquipmentMember2021-01-012021-12-310001016169srt:MinimumMemberatrs:ProductionMoldsToolingAndEquipmentMember2021-01-012021-12-310001016169srt:MaximumMemberatrs:ProductionMoldsToolingAndEquipmentMember2021-01-012021-12-310001016169us-gaap:LeaseholdImprovementsMember2021-01-012021-12-31xbrli:pure0001016169srt:MinimumMemberatrs:InsuranceCustomersMember2021-01-012021-12-310001016169srt:MaximumMemberatrs:InsuranceCustomersMember2021-01-012021-12-310001016169atrs:GovernmentsMembersrt:MinimumMember2021-01-012021-12-310001016169atrs:GovernmentsMembersrt:MaximumMember2021-01-012021-12-310001016169atrs:RebatesAndChargebacksMember2019-12-310001016169atrs:PatientDiscountProgramsMember2019-12-310001016169atrs:ReturnsMember2019-12-310001016169atrs:WholesalerDistributionFeesMember2019-12-310001016169atrs:PromptPaymentDiscountsMember2019-12-310001016169atrs:RebatesAndChargebacksMember2020-01-012020-12-310001016169atrs:PatientDiscountProgramsMember2020-01-012020-12-310001016169atrs:ReturnsMember2020-01-012020-12-310001016169atrs:WholesalerDistributionFeesMember2020-01-012020-12-310001016169atrs:PromptPaymentDiscountsMember2020-01-012020-12-310001016169atrs:RebatesAndChargebacksMember2020-12-310001016169atrs:PatientDiscountProgramsMember2020-12-310001016169atrs:ReturnsMember2020-12-310001016169atrs:WholesalerDistributionFeesMember2020-12-310001016169atrs:PromptPaymentDiscountsMember2020-12-310001016169atrs:RebatesAndChargebacksMember2021-01-012021-12-310001016169atrs:PatientDiscountProgramsMember2021-01-012021-12-310001016169atrs:ReturnsMember2021-01-012021-12-310001016169atrs:WholesalerDistributionFeesMember2021-01-012021-12-310001016169atrs:PromptPaymentDiscountsMember2021-01-012021-12-310001016169atrs:RebatesAndChargebacksMember2021-12-310001016169atrs:PatientDiscountProgramsMember2021-12-310001016169atrs:ReturnsMember2021-12-310001016169atrs:WholesalerDistributionFeesMember2021-12-310001016169atrs:PromptPaymentDiscountsMember2021-12-3100010161692022-01-012021-12-310001016169atrs:ProductionMoldsToolingAndEquipmentMember2021-12-310001016169atrs:ProductionMoldsToolingAndEquipmentMember2020-12-310001016169us-gaap:LeaseholdImprovementsMember2021-12-310001016169us-gaap:LeaseholdImprovementsMember2020-12-310001016169atrs:FurnitureFixturesAndOfficeEquipmentMember2021-12-310001016169atrs:FurnitureFixturesAndOfficeEquipmentMember2020-12-310001016169atrs:ComputerEquipmentAndSoftwareMember2021-12-310001016169atrs:ComputerEquipmentAndSoftwareMember2020-12-310001016169atrs:ConstructionAndToolingInProcessMember2021-12-310001016169atrs:ConstructionAndToolingInProcessMember2020-12-310001016169atrs:MinneapolisMember2021-12-31atrs:renewal_option00010161692021-03-010001016169us-gaap:SubsequentEventMember2021-11-012022-03-01atrs:extension00010161692019-07-01utr:sqft00010161692019-07-012019-07-010001016169atrs:TLANDOProductRightsMember2021-01-012021-12-310001016169atrs:TLANDOProductRightsMember2021-12-310001016169atrs:TLANDOProductRightsMember2020-12-310001016169atrs:NocdurnaProductRightsMember2021-01-012021-12-310001016169atrs:NocdurnaProductRightsMember2021-12-310001016169atrs:NocdurnaProductRightsMember2020-12-310001016169us-gaap:PatentsMembersrt:MinimumMember2021-01-012021-12-310001016169us-gaap:PatentsMembersrt:MaximumMember2021-01-012021-12-310001016169us-gaap:PatentsMember2021-12-310001016169us-gaap:PatentsMember2020-12-310001016169atrs:LicenseAgreementMemberatrs:LipocineIncMember2021-10-012021-10-310001016169atrs:LicenseAgreementMemberatrs:LipocineIncMember2021-12-310001016169atrs:LicenseAgreementMemberatrs:LipocineIncMember2021-10-012021-12-310001016169atrs:LicenseAgreementMemberatrs:LipocineIncMember2021-01-012021-12-310001016169atrs:LicenseAgreementMemberatrs:FerringInternationalCenterSAAndItsAffiliatesMember2020-10-012020-10-310001016169atrs:LicenseAgreementMemberatrs:FerringInternationalCenterSAAndItsAffiliatesMember2021-10-012021-10-310001016169us-gaap:SellingGeneralAndAdministrativeExpensesMember2021-01-012021-12-310001016169us-gaap:SellingGeneralAndAdministrativeExpensesMember2020-01-012020-12-310001016169us-gaap:SellingGeneralAndAdministrativeExpensesMember2019-01-012019-12-310001016169atrs:TermLoanMembersrt:MaximumMemberatrs:HerculesCapitalIncMember2017-06-060001016169atrs:HerculesCapitalIncMemberatrs:TermLoanTrancheOneMember2017-06-060001016169atrs:TermLoanMemberatrs:HerculesCapitalIncMemberus-gaap:PrimeRateMember2017-06-162017-06-160001016169atrs:TermLoanMemberatrs:HerculesCapitalIncMember2020-12-310001016169atrs:TermLoanMemberatrs:HerculesCapitalIncMember2021-01-012021-12-310001016169atrs:TermLoanMemberatrs:FirstAmendmentMemberatrs:HerculesCapitalIncMember2019-06-250001016169atrs:TermLoanMemberatrs:FirstAmendmentMemberatrs:HerculesCapitalIncMember2019-06-260001016169atrs:FirstAmendmentMemberatrs:HerculesCapitalIncMemberatrs:TermLoanTrancheTwoMember2019-06-262019-06-260001016169atrs:FirstAmendmentMembersrt:MinimumMemberatrs:HerculesCapitalIncMemberatrs:TermLoanTrancheThreeMember2019-06-260001016169atrs:FirstAmendmentMembersrt:MaximumMemberatrs:HerculesCapitalIncMemberatrs:TermLoanTrancheThreeMember2019-06-260001016169atrs:HerculesCapitalIncMemberatrs:TermLoanTrancheOneMember2021-01-012021-12-310001016169atrs:HerculesCapitalIncMemberatrs:TermLoanTrancheTwoMember2021-01-012021-12-310001016169atrs:TermLoanMembersrt:MinimumMemberatrs:HerculesCapitalIncMember2021-01-012021-12-310001016169atrs:TermLoanMembersrt:MaximumMemberatrs:HerculesCapitalIncMember2021-01-012021-12-310001016169atrs:TermLoanMemberatrs:HerculesCapitalIncMember2021-11-010001016169atrs:TermLoanMemberatrs:HerculesCapitalIncMember2021-11-012021-11-010001016169atrs:CreditAgreementMember2021-11-010001016169atrs:CreditAgreementMemberus-gaap:SecuredDebtMember2021-11-010001016169atrs:CreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:LineOfCreditMember2021-11-010001016169us-gaap:LetterOfCreditMemberatrs:CreditAgreementMemberus-gaap:LineOfCreditMember2021-11-010001016169atrs:CreditAgreementMemberus-gaap:LineOfCreditMemberus-gaap:BridgeLoanMember2021-11-010001016169atrs:TermLoanMemberatrs:HerculesCapitalIncMemberus-gaap:SecuredDebtMember2021-11-012021-11-010001016169atrs:CreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:LineOfCreditMember2021-12-310001016169us-gaap:LetterOfCreditMemberatrs:CreditAgreementMemberus-gaap:LineOfCreditMember2021-12-310001016169atrs:CreditAgreementMemberus-gaap:LineOfCreditMemberus-gaap:BridgeLoanMember2021-12-310001016169srt:MinimumMemberatrs:CreditAgreementMemberus-gaap:RevolvingCreditFacilityMember2021-01-012021-12-310001016169srt:MaximumMemberatrs:CreditAgreementMemberus-gaap:RevolvingCreditFacilityMember2021-01-012021-12-310001016169atrs:CreditAgreementMember2021-01-012021-12-310001016169atrs:CreditAgreementMemberus-gaap:RevolvingCreditFacilityMember2021-01-012021-12-310001016169us-gaap:LetterOfCreditMembersrt:MinimumMemberatrs:CreditAgreementMemberus-gaap:FederalFundsEffectiveSwapRateMember2021-01-012021-12-310001016169srt:MinimumMemberatrs:CreditAgreementMemberus-gaap:LondonInterbankOfferedRateLIBORMember2021-01-012021-12-310001016169us-gaap:LetterOfCreditMembersrt:MinimumMemberatrs:CreditAgreementMemberus-gaap:LondonInterbankOfferedRateLIBORMember2021-12-310001016169atrs:CreditAgreementMemberus-gaap:BaseRateMember2021-01-012021-12-310001016169atrs:CreditAgreementMemberus-gaap:LondonInterbankOfferedRateLIBORMember2021-01-012021-12-310001016169atrs:CreditAgreementMember2021-12-310001016169atrs:SalesAgreementMembersrt:MaximumMemberatrs:CowenAndCompanyLimitedLiabilityCompanyMember2017-08-012017-08-310001016169atrs:SalesAgreementMemberatrs:CowenAndCompanyLimitedLiabilityCompanyMember2017-08-012017-08-310001016169atrs:ATMFacilityMember2019-01-012019-12-3100010161692021-06-012021-06-300001016169atrs:AmendedAndRestatedEquityCompensationPlanMember2021-12-310001016169atrs:AmendedAndRestatedEquityCompensationPlanMember2021-01-012021-12-310001016169atrs:AmendedAndRestatedEquityCompensationPlanMemberus-gaap:EmployeeStockOptionMember2021-01-012021-12-310001016169atrs:AmendedAndRestatedEquityCompensationPlanMembersrt:MinimumMemberus-gaap:EmployeeStockOptionMember2021-01-012021-12-310001016169atrs:AmendedAndRestatedEquityCompensationPlanMemberus-gaap:EmployeeStockOptionMember2021-12-310001016169us-gaap:EmployeeStockOptionMember2021-01-012021-12-310001016169us-gaap:EmployeeStockOptionMember2020-01-012020-12-310001016169us-gaap:EmployeeStockOptionMember2019-01-012019-12-310001016169atrs:LongTermIncentiveProgramMemberus-gaap:EmployeeStockOptionMember2021-01-012021-12-310001016169us-gaap:PerformanceSharesMemberatrs:LongTermIncentiveProgramMember2021-01-012021-12-31atrs:installment0001016169us-gaap:PerformanceSharesMember2018-12-310001016169us-gaap:RestrictedStockUnitsRSUMember2018-12-310001016169us-gaap:PerformanceSharesMember2019-01-012019-12-310001016169us-gaap:RestrictedStockUnitsRSUMember2019-01-012019-12-310001016169us-gaap:PerformanceSharesMember2019-12-310001016169us-gaap:RestrictedStockUnitsRSUMember2019-12-310001016169us-gaap:PerformanceSharesMember2020-01-012020-12-310001016169us-gaap:RestrictedStockUnitsRSUMember2020-01-012020-12-310001016169us-gaap:PerformanceSharesMember2020-12-310001016169us-gaap:RestrictedStockUnitsRSUMember2020-12-310001016169us-gaap:PerformanceSharesMember2021-01-012021-12-310001016169us-gaap:RestrictedStockUnitsRSUMember2021-01-012021-12-310001016169us-gaap:PerformanceSharesMember2021-12-310001016169us-gaap:RestrictedStockUnitsRSUMember2021-12-310001016169srt:MinimumMemberus-gaap:PerformanceSharesMemberatrs:LongTermIncentiveProgramMember2021-01-012021-12-310001016169srt:MaximumMemberus-gaap:PerformanceSharesMemberatrs:LongTermIncentiveProgramMember2021-01-012021-12-310001016169us-gaap:PerformanceSharesMemberatrs:AwardDatePeriodOneMemberatrs:LongTermIncentiveProgramMember2021-12-310001016169us-gaap:PerformanceSharesMemberus-gaap:SubsequentEventMemberatrs:LongTermIncentiveProgramMember2022-01-012022-03-310001016169us-gaap:RestrictedStockUnitsRSUMemberatrs:LongTermIncentiveProgramMember2021-01-012021-12-310001016169atrs:EmployeesTaxObligationsMember2021-01-012021-12-310001016169atrs:EmployeesTaxObligationsMember2020-01-012020-12-310001016169atrs:EmployeesTaxObligationsMember2019-01-012019-12-310001016169us-gaap:RestrictedStockUnitsRSUMembersrt:DirectorMember2021-01-012021-12-310001016169srt:DirectorMember2021-01-012021-12-310001016169srt:DirectorMember2020-01-012020-12-310001016169srt:DirectorMember2019-01-012019-12-310001016169us-gaap:EmployeeStockOptionMember2021-12-310001016169atrs:OTREXUPAssetsMemberus-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMember2021-12-310001016169atrs:OTREXUPAssetsMemberus-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMember2021-12-012021-12-310001016169us-gaap:DomesticCountryMember2021-12-310001016169srt:MinimumMemberus-gaap:DomesticCountryMember2021-01-012021-12-310001016169srt:MaximumMemberus-gaap:DomesticCountryMember2021-01-012021-12-310001016169us-gaap:ForeignCountryMember2021-12-310001016169us-gaap:ForeignCountryMember2021-01-012021-12-310001016169atrs:ProprietaryProductMemberus-gaap:SalesMember2021-01-012021-12-310001016169atrs:ProprietaryProductMemberus-gaap:SalesMember2020-01-012020-12-310001016169atrs:ProprietaryProductMemberus-gaap:SalesMember2019-01-012019-12-310001016169us-gaap:SalesMemberatrs:PartneredProductMember2021-01-012021-12-310001016169us-gaap:SalesMemberatrs:PartneredProductMember2020-01-012020-12-310001016169us-gaap:SalesMemberatrs:PartneredProductMember2019-01-012019-12-310001016169us-gaap:SalesMember2021-01-012021-12-310001016169us-gaap:SalesMember2020-01-012020-12-310001016169us-gaap:SalesMember2019-01-012019-12-310001016169atrs:LicensingAndDevelopmentRevenueMember2021-01-012021-12-310001016169atrs:LicensingAndDevelopmentRevenueMember2020-01-012020-12-310001016169atrs:LicensingAndDevelopmentRevenueMember2019-01-012019-12-310001016169atrs:RoyaltiesRevenueMember2021-01-012021-12-310001016169atrs:RoyaltiesRevenueMember2020-01-012020-12-310001016169atrs:RoyaltiesRevenueMember2019-01-012019-12-310001016169country:US2021-01-012021-12-310001016169country:US2020-01-012020-12-310001016169country:US2019-01-012019-12-310001016169srt:EuropeMember2021-01-012021-12-310001016169srt:EuropeMember2020-01-012020-12-310001016169srt:EuropeMember2019-01-012019-12-310001016169atrs:OtherGeographicalAreaMember2021-01-012021-12-310001016169atrs:OtherGeographicalAreaMember2020-01-012020-12-310001016169atrs:OtherGeographicalAreaMember2019-01-012019-12-310001016169us-gaap:RevenueFromContractWithCustomerMemberatrs:TevaMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001016169us-gaap:RevenueFromContractWithCustomerMemberatrs:TevaMemberus-gaap:CustomerConcentrationRiskMember2020-01-012020-12-310001016169us-gaap:RevenueFromContractWithCustomerMemberatrs:TevaMemberus-gaap:CustomerConcentrationRiskMember2019-01-012019-12-310001016169atrs:McKessonMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001016169atrs:McKessonMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2020-01-012020-12-310001016169atrs:McKessonMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2019-01-012019-12-310001016169us-gaap:RevenueFromContractWithCustomerMemberatrs:AmerisourceBergenMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001016169us-gaap:RevenueFromContractWithCustomerMemberatrs:AmerisourceBergenMemberus-gaap:CustomerConcentrationRiskMember2020-01-012020-12-310001016169atrs:CardinalHealthMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001016169atrs:CardinalHealthMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2020-01-012020-12-310001016169us-gaap:RevenueFromContractWithCustomerMemberatrs:CovisMemberus-gaap:CustomerConcentrationRiskMember2019-01-012019-12-310001016169atrs:LipocineIncMember2021-10-012021-10-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
______________________________________________________________________
FORM 10-K
(Mark One)
| | | | | |
| ☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15 (d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2021
| | | | | |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15 (d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For transition period from ______ to ______
Commission file number 001-32302
ANTARES PHARMA, INC.
(Exact name of registrant as specified in its charter)
Delaware
(State or other jurisdiction of incorporation)
41-1350192
(I.R.S. Employer Identification No.)
100 Princeton South, Suite 300, Ewing, NJ
(Address of principal executive offices)
08628
(Zip Code)
Registrant’s telephone number, including area code: (609) 359-3020
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | | | | | | | |
| Title of each class | | Trading
Symbol(s) | | Name of each exchange on which registered |
| Common Stock, par value $0.01 per share | | ATRS | | NASDAQ |
Securities registered pursuant to section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes x No ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | | | | | | | |
| Large accelerated filer | ¨ | Accelerated filer | x | Non-accelerated filer | ¨ |
| Smaller reporting company | ¨ | Emerging growth company | ¨ | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. Yes ☒ No ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No x
The aggregate market value of the voting and non-voting common stock held by non-affiliates of the registrant as of June 30, 2021, was $739.4 million based on the closing price of $4.36 per share on June 30, 2021 as reported by the NASDAQ Capital Market.
As of February 28, 2022, there were 170,106,346 shares of common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the definitive Proxy Statement to be filed within 120 days after the end of the period covered by this report for the registrant’s 2022 Annual Meeting of Stockholders are incorporated by reference into Part III of this Annual Report on Form 10-K.
ANTARES PHARMA, INC.
Annual Report on Form 10-K
For the Year Ended December 31, 2021
TABLE OF CONTENTS
Note Regarding Forward-Looking Statements
This annual report contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and the Private Securities Litigation Reform Act of 1995 that are subject to risks and uncertainties. Undue reliance should not be placed on those statements because they are subject to numerous uncertainties and factors relating to our operations and business environment, all of which are difficult to predict and many of which are beyond our control. These statements can be identified by the fact that they do not relate strictly to historical or current facts. Such statements may include words such as “anticipate,” “will,” “estimate,” “expect,” “project,” “intend,” “should,” “plan,” “believe,” “hope,” “may,” “continue,” or other words and terms of similar meaning in connection with any discussion of, among other things, future operating or financial performance, strategic initiatives and business strategies, regulatory or competitive environments, our intellectual property, our commercial operations and product development. In particular, these forward-looking statements include, among others, statements about:
•our expectations about the ongoing COVID-19 pandemic (the “Pandemic”) and any potential disruption or impact to our operations, financial position or cash flows;
•our expectations regarding the continued commercialization of XYOSTED® (testosterone enanthate) injection and the continued growth in prescriptions and revenues related thereto;
•our expectations regarding the commercialization of NOCDURNA® (desmopressin acetate) in the U.S. under a licensing agreement with Ferring International Center S.A. and its affiliates, (“Ferring”) and future sales and revenue from the same;
•our expectations regarding future FDA approval of TLANDO® in the U.S. under a licensing agreement with Lipocine Inc. (“Lipocine”), the manufacturing and commercialization of TLANDO® and future sales and revenue from the same;
•our expectations regarding whether we will exercise the option for LPCN 1111 (“TLANDO XR”) and, if exercised, the future timing and success of the clinical development program for TLANDO XR and future FDA approval, market acceptance and revenue from the same;
•our expectations regarding future sales of OTREXUP® (methotrexate) injection to Otter Pharmaceuticals, LLC (a wholly-owned subsidiary of Assertio Holdings, Inc., together with Assertio Holdings, Inc., as guarantor, individually and collectively referred to as “Otter”) under a newly entered into supply agreement, as well as the ability of Otter to pay remaining installment payments of the purchase price;
•our expectations regarding the ability of our partner, Teva Pharmaceutical Industries, Ltd. (“Teva”), to continue to commercialize Epinephrine Injection USP, the generic equivalent version of EpiPen® (“generic epinephrine injection”), and any future revenue related thereto;
•our expectations regarding the ability of the Covis Group S.a.r.l. (“CG”), who acquired AMAG Pharmaceuticals, Inc. (“AMAG”) (collectively CG and AMAG are herein after referred to as “Covis”) in November 2020, to continue to commercialize Makena® (hydroxyprogesterone caproate injection) and our continued future sales to Covis and royalty revenue from the same, in light of the U.S. Food and Drug Administration’s (“FDA”) proposal to withdraw approval of Makena® (hydroxyprogesterone caproate injection) and the timing and outcome of any hearings and future regulatory actions by the FDA;
•our expectations regarding continued sales of Sumatriptan Injection USP to our partner, Teva, and Teva’s ability to distribute and sell Sumatriptan Injection USP;
•our expectations regarding continued product development with Teva of the teriparatide disposable pen injector, Teva’s ability to obtain FDA approval and AB-rating for the products and if approved Teva’s ability to successfully commercialize the teriparatide disposable pen injector product outside the U.S.;
•our expectations about the development of a rescue pen for an undisclosed drug with our partner Pfizer Inc. (“Pfizer”) and potential future regulatory approval and future revenue from the same;
•our expectations about our development activities with Idorsia Pharmaceuticals Ltd (“Idorsia”) and the timing and results of the Phase 3 clinical trial of the drug device combination product for selatogrel, a new chemical entity being developed for the treatment of a suspected acute myocardial infarction (“AMI”) in adult patients with a history of AMI, and the potential future FDA and global regulatory approval of the same;
•our expectations about the development of ATRS-1902 for adrenal crisis rescue, including the timing and results of clinical trials and our anticipated 505(b)(2) NDA filing with the FDA;
•our expectations about our other internal and external research and development projects, including but not limited to ATRS-1901 and ATRS-1903, the timing and results of clinical trials, and our anticipated continued reliance on third parties in conducting studies, trials and other research and development activities;
•our expectations about the timing and outcome of pending or potential claims and litigation, including without limitation, the pending securities class action and derivative actions;
•our anticipated continued reliance on contract manufacturers to manufacture, assemble and package our products;
•our anticipated continued reliance on third parties to provide certain services for our products including logistics, warehousing, distribution, invoicing, contract administration and chargeback processing;
•our sales and marketing plans;
•our expectation about our future revenues, our cash flows and our ability to support our operations and maintain profitability;
•our estimates and expectations regarding the sufficiency of our cash resources, anticipated capital requirements and our ability to obtain additional financing, if needed;
•the potential impact of new accounting pronouncements and tax legislation; and
•other statements regarding matters that are not historical facts or statements of current condition.
These forward-looking statements are based on assumptions that we have made considering our industry experience as well as our perceptions of historical trends, current conditions, expected future developments and other factors we believe are appropriate under the circumstances. As you read and consider this annual report, you should understand that these statements are not guarantees of performance results. Forward-looking statements involve known and unknown risks, uncertainties and assumptions, and other factors that may cause our or our industry’s actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. While we believe that we have a reasonable basis for each forward-looking statement contained in this annual report, we caution you that these statements are based on a combination of facts and factors currently known by us and projections of the future about which we cannot be certain. Many factors may affect our ability to achieve our objectives, including:
•potential business interruptions and/or any financial or operational impact as a result of the Pandemic;
•delays in product introduction or unsuccessful marketing and commercialization efforts by us or our partners;
•interruptions in supply or an inability to adequately manage third party contract manufacturers to meet customer supply requirements;
•our inability to obtain or maintain adequate third-party payer coverage of marketed products;
•the timing and results of our or our partners’ research projects or clinical trials of product candidates in development;
•actions by the FDA or other regulatory agencies with respect to our products or product candidates of our partners;
•our inability to generate or sustain continued growth in product sales and royalties;
•the lack of market acceptance of our and our partners’ products and future revenues from these products;
•a decrease in business from our major customers and partners;
•our inability to compete successfully against new and existing competitors or to leverage our research and development capabilities or our marketing capabilities;
•our inability to establish and maintain our commercial capabilities, our inability to effectively market our services or obtain and maintain arrangements with our customers, partners and manufacturers;
•our inability to attract and retain key personnel;
•changes or delays in the regulatory review and approval process;
•our inability to effectively protect our intellectual property;
•costs associated with future litigation and the outcome of such litigation; and
•adverse economic and political conditions.
The performance of our business and our securities may be adversely affected by these factors and by other factors common to other businesses or to the general economy. Forward-looking statements speak only as of the date on which such statements are made. Actual results could differ materially from those currently anticipated as a result of a number of risk factors, including, but not limited to, the risks and uncertainties discussed in Item 1A of Part II of this Annual Report on Form 10-K. New risks and uncertainties emerge from time to time, and it is impossible for us to predict these events or how they may affect us. Forward-looking statements are qualified by some or all of these risk factors. Therefore, you should consider these forward-looking statements with caution and form your own critical and independent conclusions about the likely effect of these risk factors on our future performance. We undertake no obligation to update or revise any forward-looking statements included in this annual report to reflect events or circumstances after the date on which such statement is made, except as required by law. In light of these risks and significant uncertainties, you should not regard the forward-looking statements in this annual report as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified timeframe, if at all. You should carefully review the disclosures and the risk factors described in this Annual Report on Form 10-K and in other documents we file from time to time with the Securities and Exchange Commission (“SEC”), including our Quarterly Reports on Form 10-Q and Current Reports on Form 8-K.
PART I
ITEM 1. BUSINESS
Company Overview
Antares Pharma, Inc. (“Antares,” “we,” “our,” “us” or the “Company”) is a specialty pharmaceutical company focused primarily on the development and commercialization of pharmaceutical products and technologies that address patient needs in targeted therapeutic areas. We develop, manufacture and commercialize, for ourselves or with partners, novel therapeutic products using our advanced drug delivery systems that are designed to provide commercial or functional advantages such as improved safety and efficacy, convenience, improved tolerability, and enhanced patient comfort and adherence. We also seek product opportunities that complement and leverage our commercial platform. We have a portfolio of proprietary and partnered commercial products and ongoing product development programs in various stages of development. We have formed partnership arrangements with several different industry leading pharmaceutical companies.
Our FDA-approved products include XYOSTED® (testosterone enanthate) injection; OTREXUP® (methotrexate) injection, which was sold to Otter in December 2021; NOCDURNA® (desmopressin acetate), which is licensed from Ferring; and Sumatriptan Injection USP, which is distributed by Teva. We are also the exclusive supplier of devices to Teva for their Epinephrine Injection USP, the generic equivalent of EpiPen® and EpiPen® Jr.; the devices for Teva’s generic teriparatide; OTREXUP® (methotrexate) injection to Otter beginning in December 2021; and of the Makena® subcutaneous auto injectors to Covis.
2021 Highlights and Areas of Focus
We achieved several significant operating and financial milestones in 2021:
•Record Revenue and Financial Results – We generated record revenue of $184.0 million for the year ended December 31, 2021 as compared to $149.6 million for the year ended December 31, 2020, representing year-over-year growth of 23.0%. We generated record pre-tax income of $62.3 million resulting in net income and basic earnings per share of $46.3 million and $0.27, respectively, for the year ended December 31, 2021 as compared to net income and basic earnings per share of $56.2 million and $0.34, respectively, for the year ended December 31, 2020. Earnings per share on a fully diluted basis was $0.26 for the year ended December 31, 2021 compared to $0.33 for the year ended December 31, 2020.
•Significant Growth of XYOSTED® – Our proprietary product XYOSTED® (testosterone enanthate) injection, indicated for testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of endogenous testosterone, continued to grow significantly in 2021, generating annual net revenue of $62.2 million for the year ended December 31, 2021 compared to $46.5 million for the year ended December 31, 2020. We attribute this 33.7% increase to successful marketing strategies, achieving and maintaining targeted reimbursement coverage, and our ability to continue to leverage our virtual capabilities to support the growth in 2021 despite the challenges presented by the Pandemic. XYOSTED® is the only FDA approved subcutaneous testosterone enanthate product for once-weekly, at-home self-administration of testosterone replacement therapy.
•Expanded Product Portfolio with In-License of TLANDO® – We entered into an exclusive license agreement with Lipocine for the product TLANDO® (testosterone undecanoate) in the U.S., a twice-daily oral formulation of testosterone for testosterone replacement therapy indicated for conditions associated with a deficiency or absence of endogenous testosterone, or hypogonadism in adult males. TLANDO® was granted tentative approval from the FDA in December 2020 and will be eligible for final FDA approval and marketing in the U.S. upon expiration of the exclusivity period previously granted to Clarus Therapeutics, Inc. (“Clarus”) for JATENZO® on March 27, 2022. On February 3, 2022, we announced the FDA’s acceptance of our NDA resubmission for TLANDO® with a target action date set for March 28, 2022. We continue to prepare for the launch of TLANDO® in 2022 pending final FDA approval after the expiration of JATENZO®’s exclusivity period.
•Aligned Proprietary Portfolio with Divestiture of OTREXUP® – We divested and sold our proprietary product line OTREXUP® (methotrexate) injection for the treatment of rheumatoid arthritis to Otter in December 2021 for a total purchase price of $44.0 million (the “Asset Purchase Agreement”), subject to finalization of changes in closing inventory to be transferred. This divestiture allows us to further align our commercial detailing strategy to focus on the urology and endocrinology fields to enhance our growth strategy for XYOSTED®, NOCDURNA® and the anticipated launch of TLANDO® in 2022. With the asset sale, we will continue to manufacture and supply OTREXUP® to Otter as a partnered product.
•Significant Progress on Our Adrenal Crisis Rescue Pen (ATRS-1902) – We further advanced our ATRS-1902 development program with positive results from a Phase 1 clinical study for adrenal crisis and were granted Fast Track designation by the FDA. The results support the advancement of our ATRS-1902 development program to a pivotal clinical study for the treatment of acute adrenal insufficiency (“adrenal crisis”) in adults and adolescents, using our Vai™ novel proprietary rescue pen platform to deliver a liquid stable formulation of hydrocortisone. We anticipate starting this pivotal clinical study in the second quarter of 2022 and expect to submit a 505(b)(2) NDA with the FDA by the end of 2022 pending the success of the pivotal clinical study.
•Continued Navigation of the Global Pandemic – We have taken several measures to manage and minimize the impact of the Pandemic on our business. We have implemented safety measures and protocols to protect the health and safety of our employees and comply with governmental and public health guidelines while ensuring the sustainability of our business operations and continuity of product supply. We continue to monitor the situation, including the COVID-19 variants, and potential effects on our business, suppliers, partners and workforce. We have implemented a hybrid work environment with the ability to shift remote as necessary to limit the number of individuals in our facilities to those necessary for essential functions such as research, development, manufacturing and supply. While our field-based team has resumed in-person interaction with fewer restrictions, we believe we are also well-positioned with our virtual capabilities to continue to engage healthcare professionals and patients through the Pandemic and beyond.
In addition to these significant achievements and areas of focus in 2021, we continued to devote resources and advance our internal research and development programs to further expand our product pipeline. We also made significant progress on partnered development projects, made investments in capital improvements and infrastructure, and maintained a disciplined approach to growth and operating expenditures to support our continued and future growth.
Product Portfolio Overview
The following table provides an overview of our proprietary and partnered commercial products and product opportunities:
| | | | | | | | | | | | | | |
| Approved Products | Drug | Partner | Disease/Condition | Territory |
XYOSTED® (testosterone enanthate) injection | Testosterone | None | Testosterone Replacement Therapy (“TRT”) | U.S. |
OTREXUP® (methotrexate) injection 1 | Methotrexate | None 1 | Rheumatoid Arthritis; pJIA, Psoriasis | U.S. |
NOCDURNA® (desmopressin acetate) | Desmopressin | None 2 | Nocturnal Polyuria | U.S. |
Epinephrine Injection USP (generic equivalent to EpiPen® and EpiPen® Jr.) | Epinephrine | Teva | Anaphylaxis | U.S. |
Sumatriptan Injection USP (generic version of Imitrex® STATdose Pen®) | Sumatriptan succinate | Teva | Migraines | U.S. |
Makena® Subcutaneous Auto Injector | Hydroxy-progesterone caproate | Covis | Reduced Risk of Pre-term Birth | U.S. |
Teriparatide Injection (generic version of Forsteo®) | Teriparatide | Teva | Osteoporosis | Outside U.S. |
| | | | | | | | | | | | | | |
| Select Products in Development | Drug | Partner | Indication | Territory |
TLANDO® (testosterone undecanoate) 3 | Testosterone | None 3 | Testosterone Replacement Therapy (“TRT”) | U.S. |
Disposable Pen Injector 4 | Exenatide | Teva | Diabetes | U.S. |
| Disposable Pen Injector | Teriparatide | Teva | Osteoporosis | U.S. |
QuickShot® Auto Injector | Undisclosed | Pfizer | Undisclosed Rescue Pen | U.S. |
QuickShot® Auto Injector | Selatogrel | Idorsia | Acute Myocardial Infarction | Worldwide |
| Drug/Device Product | ATRS-1901 | None | Urologic Oncology | U.S. |
| Drug/Device Product | ATRS-1902 | None | Endocrinology | U.S. |
| Drug/Device Product | ATRS-1903 | None | Immunology | U.S. |
1 Certain worldwide assets used in the operation of the OTREXUP® product were sold to Otter in December 2021. We will continue to manufacture and supply OTREXUP® to Otter as a partnered product under a separate supply agreement.
2 Distributed and sold by us under an exclusive license agreement with Ferring.
3 TLANDO® was granted tentative approval from the FDA in December 2020 and will be eligible for final FDA approval and marketing in the U.S. upon expiration of the exclusivity period previously granted to Clarus for JATENZO® on March 27, 2022. TLANDO® is expected to be distributed and sold by us under an exclusive license agreement with Lipocine pending final approval from the FDA. We announced the FDA’s acceptance of our application for final approval on February 3, 2022 with a target action date set for March 28, 2022.
4 On February 25, 2022, Teva notified us that it was terminating the exenatide program and related agreement due to a lack of commercial viability. The termination is effective August 23, 2022.
For a detailed discussion of our proprietary and partnered approved and marketed products, and other products currently in development, see “Our Products” and “Research and Development” sections below included in Item 1 of Part I of this Annual Report on Form 10-K.
Corporate Information
Antares was incorporated under Delaware law in 2005 with principal executive offices located in Ewing, New Jersey. We have two wholly owned subsidiaries in Switzerland (Antares Pharma AG and Antares Pharma IPL AG) and operate under a single reportable operating segment, which encompasses all of our pharmaceutical products and technologies. Segment and geographic financial information are included in Note 2 and Note 14 to the Consolidated Financial Statements in Item 8 of Part II of this Annual Report on Form 10-K.
Our Strategy and Market Opportunity
Our strategy is to grow the business through targeted investments in internal and partnered product development and other corporate opportunities, as well as leverage our commercial infrastructure, primarily focused in certain therapeutic areas. We have built a robust commercial organization to market and sell our proprietary products and have significant experience in developing drug/device combination products and navigating the regulatory approval process.
Historically, our focus has been primarily the market for self-administered injectable drugs. We identify development and commercialization opportunities, both internally and through partnered or business development opportunities that apply patented drug delivery technologies to new or existing approved drug formulations. Through these opportunities, we seek to enhance the drug delivery methods and provide commercial and/or functional advantages, such as improved safety and efficacy, reduced side effects, convenience and enhanced patient comfort and adherence. In addition to self-administered injectable drugs, we explore opportunities beyond injectable drugs that may complement our strategy and leverage our capabilities. We pursue these opportunities both on our own or with partners. We believe this strategy offers a distinct value to patients, healthcare providers, pharmaceutical partners and our shareholders.
Injection is a common drug delivery pathway, and the delivery of pharmaceutical therapies through injection systems often improves the systemic bioavailability of those treatments by overcoming absorption barriers common with oral and, in some cases, transdermal delivery. Improved bioavailability is beneficial when evaluating the role of route of administration on pharmaceutical efficacy. We believe that our advanced injection technology platforms provide drug delivery solutions that can result in improved safety and efficacy, reduced side effects, and enhanced patient comfort and adherence. Many pharmaceutical companies focus on the development of important chronic care products and emergency rescue therapies that can be administered only by injection. We believe our advanced injection technologies uniquely address these market needs and are well suited for both the branded and generic marketplace.
We and our partners have historically sought, and are in the process of seeking, FDA approval for certain product candidates primarily using the 505(b)(2) NDA (New Drug Application) or ANDA (Abbreviated New Drug Application) approval pathways, as well as more recently the 505(b)(1) NDA pathway with certain partners, which are further described in the “Government Regulation” section below included in Item 1 of Part I of this Annual Report on Form 10-K. Our technology platforms allow for device customization, which can provide multiple opportunities in both the 505(b)(2) NDA and generic market spaces, as well as the 505(b)(1) NDA pathway.
According to a Market Research Engine report, it is estimated that the global injectable drug delivery market will grow to $1.3 billion by 2024, representing a compounded annual growth rate of 12.9% in the forecast period. This expected growth is attributable to several factors, including label expansion for approved products, increasing the patient pool for such products, a pipeline of injectable medications at various stages of clinical development, and the increasing incidence of certain diseases that will necessitate the utilization of injectable medications.
See also “Our Products” and “Research and Development” below, included in Item 1 of Part I of this Annual Report on Form 10-K for additional discussion of market size and opportunities relative to the current therapeutic areas associated with our existing portfolio of products and products in development.
Our Competitive Strengths
We believe our key competitive strengths are our commercial capabilities and infrastructure, proprietary injection technologies, and our ability to form significant strategic alliances with industry-leading pharmaceutical partners to develop and commercialize products. We also believe our management team has unique knowledge of, and experience in the drug/device combination product space along with navigating the related regulatory approval process, which creates opportunities for us and potential pharmaceutical partners. Our business model for developing and commercializing proprietary and partnered products has been validated, we believe, by the successive FDA approvals of our NDAs for XYOSTED® and OTREXUP® and our ANDA for Sumatriptan Injection USP, as well as the FDA approval of Teva’s AB-rated generic version of the EpiPen®.
Intellectual Property, Patents, Trade Secrets and Proprietary Information
We strive to protect and enhance the proprietary technologies that we believe are important to our business and rely on know-how and continuing technological innovation to develop, strengthen and maintain our competitive position. When appropriate, we seek protection for our products and proprietary information by means of U.S. and international patents and trademarks. We currently hold a portfolio of patents with expirations ranging from 2021 to 2038, and numerous patent applications pending in the U.S. and other countries. These patents consist primarily of design, formulation and method-of-use patents.
In addition to our patents and patent applications, our proprietary know-how and inventions play an important role in protecting our products and technologies, and provide protection beyond patents and regulatory exclusivity. We strive to preserve the confidentiality of our proprietary know-how and inventions by maintaining physical security of our sites and electronic security of our information technology systems. We also require all employees, contractors and third-party consultants with access to proprietary information to execute confidentiality agreements prohibiting the disclosure of confidential information to anyone outside the Company. These agreements also require disclosure and assignment to us of discoveries and inventions made by such individuals while devoted to Company-sponsored activities. Partners with which we have entered into development agreements have the right to certain technology developed in connection with such agreements.
Human Capital
We believe that our success is largely dependent upon our ability to attract and retain qualified employees. We currently have 201 full-time employees and 2 part-time employees, of which 40 employees are based in our New Jersey corporate facility, 65 employees in our Minnesota operations and 98 employees based in the field. Our workforce includes 108 employees directly involved in or supporting our commercial sales organization, 27 in research and development, 37 in manufacturing and quality and 31 in corporate and administrative functions. We are not party to any collective bargaining arrangements. Although we believe that the size of our current workforce is appropriate to achieve our objectives, we may hire additional employees with specialized expertise as we continue to grow our business. We believe that we have been successful to date in attracting skilled and experienced scientific and business professionals.
We continue to focus on building a high performing organization with an engaging work culture and have established initiatives to support this strategic priority. We perform periodic employee engagement surveys, set and monitor retention goals, and intend to invest in training and leadership development to cultivate our emerging leaders. Additionally, we are committed to diversity and inclusion as a core focus of our human capital strategy. We embrace differences, diversity and varying perspectives amongst our employee base, and are proud to be an equal opportunity employer. We do not discriminate based on race, religious creed, color, national origin, ancestry, physical disability, mental disability, medical condition, genetic information, marital status, sex, gender, gender identity, gender expression, age, military or veteran status, sexual orientation or any other protected characteristic established by federal, state or local laws. A diverse workforce, as well as an inclusive culture and work environment, are fundamentally important and strategic to us, beginning with our Board of Directors and extending to all levels of the organization. As of December 31, 2021, our total employee base was 56% diverse on the basis of gender and race. We also have implemented company-wide diversity and inclusion training.
We strongly believe that the success of Antares depends, in part, on open and regular communication with employees to help foster a high performing and engaged workforce. To help ensure that employees fully understand the Company’s long-term strategy and annual goals, along with how their work contributes to the Company’s success, we use a variety of channels to facilitate open and direct communication, including: (i) quarterly CEO Town Hall meetings; (ii) regular ongoing update communications; and (iii) employee engagement surveys.
We believe our success depends upon our ability to attract and retain highly qualified employees. Talent management and leadership development is critical to our ability to execute on our long-term growth strategy. We strive to provide pay, benefits, and services that are competitive to market and create incentives to attract and retain employees. Our compensation package includes market-competitive pay, discretionary broad-based stock grants and bonuses, health care and retirement benefits, paid time off and family leave. We also continue to advance transparency in our pay and representation data by complying with all applicable statutory filing requirements. To help support the development and advancement of our high performing employees, we offer training and development programs encouraging advancement from within and continue to fill our team with strong and experienced management talent. We leverage both formal and informal programs to identify, foster, and retain top talent throughout the organization.
Our compensation philosophy is to pay for performance, support the Company’s business strategies, and offer competitive compensation arrangements to attract and retain key individuals and therefore, have established a Compensation Committee of the Board of Directors. Consistent with this philosophy, the Compensation Committee considers the impact of our corporate performance in determining compensation for named executive officers, as well as each named executive officer’s individual performance, macroeconomic conditions, and data from peer group companies.
Our Technology and Product Platforms
Pressure assisted auto injection is a form of parenteral drug delivery that continues to gain acceptance and demand among the medical and patient community. Encompassing a wide variety of sizes and designs, our technology operates by using pressure to force the drug, in solution or suspension, through the skin and deposits the drug into the subcutaneous or intramuscular tissue. We have designed disposable, pressure assisted auto injector devices to address acute and chronic medical needs, such as rheumatoid arthritis and psoriasis, allergic reactions, migraine headaches, testosterone deficiency and maternal health. Our current platforms include the VIBEX® and the VIBEX® QuickShot® disposable pressure assisted auto injection systems, the Vai™ auto injector and disposable pen injection systems.
VIBEX® Auto Injectors
Our proprietary VIBEX® disposable auto injector systems combine a spring-based power source with a shielded needle, which delivers the needed drug solution subcutaneously or intramuscularly. To minimize the anxiety and perceived pain associated with injection-based technologies, the VIBEX® system features a triggering collar that shields the needle from view. The patented retracting collar springs back and locks in place as a protective needle guard after the injection, making the device safe for general disposal. We believe the key competitive advantages of the VIBEX® system include: reliable subcutaneous or intramuscular injection, designed to work with conventional pre-filled syringes, rapid injection with ability to deliver viscous solutions, ease of use in emergencies, and reduced pain.
The primary goal of the VIBEX® disposable pressure assisted auto injector is to provide a fast, safe, and time-efficient method of self-injection. This device is designed around conventional single dose pre-filled syringes, which is a primary drug container, offering ease of transition for potential pharmaceutical partners. Our proprietary product OTREXUP® (product line, excluding the NDA, sold to Otter in December 2021) uses the VIBEX® auto injector system for delivery of methotrexate. We also have two license agreements with Teva for our VIBEX® system, one for Teva’s generic epinephrine auto injector and the other for our Sumatriptan Injection USP.
VIBEX® QuickShot® Auto Injectors
An advancement of our proprietary line of VIBEX® auto injectors is the VIBEX® QuickShot® auto injector system, which offers a dose capacity of 1 mL or greater in a compact design. VIBEX® QuickShot® is designed to enhance performance on the attributes we believe most critical to patient acceptance, which are speed, comfort and discretion. VIBEX® QuickShot® achieves these advancements by incorporating a novel triggering mechanism and space-saving spring configuration. The unique design also accommodates fast injection of highly viscous drug products that less-powerful conventional auto injectors are typically unable to deliver. Many self-injectable drugs that are currently marketed or in clinical development are of higher viscosity and are formulated to be administered in a 1 mL dose volume. Our proprietary product XYOSTED®, and the Makena® subcutaneous auto injector that we developed with our partner Covis, were developed using the VIBEX® QuickShot® auto injector platform. We also have a development agreement with Pfizer to develop a rescue pen utilizing our VIBEX® QuickShot® auto injector system with an undisclosed Pfizer drug. In addition, we have a development agreement with Idorsia for a drug device combination product using a variation of our VIBEX® QuickShot® auto injector device with a new chemical entity selatogrel which is being developed for the treatment of acute myocardial infarction.
Vai™ Auto Injector
We developed the versatile Vai™ auto injector platform to meet evolving market needs. The Vai™ auto injector builds off the capabilities of our VIBEX® and QuickShot® platforms adding automatic needle insertion, subcutaneous or intramuscular injections of up to 1 inch and delivered volumes of up to 2 mL. The Vai™ auto injector can accommodate 1 mL standard, 1 mL long, and 2.25 mL syringes. The auto-insert technology is intended to improve compliance for drug products requiring deep intramuscular injections. This innovative device is easily convertible to different fill volumes and needle lengths and was designed specifically to meet the reliability requirements for emergency use applications while maintaining the simple and intuitive two-step process of administration of the QuickShot® device.
Pen Injector System
Our multi-dose, disposable pen injector technology complements our portfolio of single-use pressure assisted auto injector devices. The disposable pen injector devices are designed to deliver drugs by injection through needles from multi-dose cartridges. Our disposable pen injectors are designed for chronic conditions such as diabetes, which require daily injection of a product. Depending on dosage, our pens can deliver up to thirty days of drug. We have licensed our pen injector device technology to Teva for two products in late-stage development: (i) a multi-dose pen with teriparatide for the treatment of osteoporosis (a generic form of Forteo®); and (ii) a multi-dose pen with exenatide for the treatment of diabetes (a generic version of BYETTA®). On February 25, 2022, Teva notified us that it was terminating the exenatide program and related agreement due to a lack of commercial viability. The termination is effective August 23, 2022.
Our Products
The following is a discussion of our approved and marketed commercial products, including proprietary and partnered products. For a discussion of other product candidates currently with tentative approval or in development, see the “Products with Tentative Approval” and “Research and Development” sections below included in Item 1 of Part I of this Annual Report on Form 10-K.
XYOSTED® (testosterone enanthate) Injection
We market and sell in the U.S. our proprietary product XYOSTED® (testosterone enanthate) injection for subcutaneous administration of testosterone replacement therapy (“TRT”) in adult males for conditions associated with a deficiency or absence of endogenous testosterone. XYOSTED® is the only FDA approved subcutaneous testosterone enanthate product for once-weekly, at-home self-administration and is approved and marketed in three dosage strengths, 50 mg, 75 mg and 100 mg. XYOSTED® provides an easy and virtually pain-free administration, low risk of transference and the ability to achieve and maintain steady levels of testosterone.
In the U.S., there are several different formulations for TRT including intramuscular injection, transdermal patches and gels, oral formulations and nasal gels. According to IQVIA National Sales Perspectives® (“NSP”) reporting of nationally projected sales activities, the overall U.S. TRT market was approximately $1.4 billion in 2021 based on wholesale acquisition costs (“WAC”). Total prescriptions in the U.S. TRT market grew by 5.1% to 8.0 million prescriptions in 2021 as compared to 2020, entirely driven by an increase in prescriptions of injectables, which increased by 5.4%. The injectable TRT market grew from $484.4 million in 2020 to $545.0 million in 2021, an increase of 12.5% based on WAC. As of December 2021, XYOSTED® commanded approximately 65% and 52% of the branded TRT market among Urologists and Endocrinologists, respectively.
Competition in the U.S. testosterone replacement market includes transdermal solutions such as AbbVie’s Androgel® 1% and 1.62%, Perrigo’s generic Androgel® Topical Gel, 1.62%, Eli Lilly’s Axiron®, Endo’s Testim® and Fortesta® (and the authorized generic) and Allergan plc’s (“Allergan”) Androderm®. Other forms of TRT include injectables such as Endo’s Aveed®, Pfizer’s Depo®-Testosterone, and several generic oil testosterone products sold by Actavis, Sandoz, Viatris Inc., Teva and others, as well as Testopel® pellets by Endo and JATENZO®, an oral formulation, by Clarus. In addition, Marius Pharmaceuticals has submitted an NDA with the FDA for Kyzatrex, an oral formulation of testosterone, that is pending FDA approval.
OTREXUP® (methotrexate) Injection
Prior to December 2021, we marketed and sold in the U.S. our proprietary product OTREXUP® (methotrexate) injection. OTREXUP® is a subcutaneous methotrexate injection for once weekly self-administration with an easy-to-use, single dose, disposable auto injector, indicated for adults with severe active rheumatoid arthritis (“RA”), children with active polyarticular juvenile idiopathic arthritis and adults with severe recalcitrant psoriasis.
In December 2021, we sold certain assets used in the operation of the OTREXUP® product to Otter under an Asset Purchase Agreement for a total purchase price of $44.0 million (the “Asset Purchase Agreement”), subject to finalization of changes in closing inventory to be transferred. Pursuant to a separate supply agreement, Otter is responsible for supplying the pre-filled syringe of methotrexate and we will continue to manufacture and oversee the assembly and packaging of the final product which is sold to Otter as a partnered product. Further, we entered into a license agreement with Otter in which we granted Otter a worldwide, exclusive, fully paid-up license to certain patents relating to OTREXUP® that may also relate to our other products for Otter to commercialize and otherwise exploit OTREXUP® in the field as defined in the license agreement.
The primary competitor in the RA market is Medexus Pharma which markets and sells Rasuvo®, a once-weekly, subcutaneous, single-dose auto-injector of methotrexate indicated for the treatment of rheumatoid arthritis, psoriasis and juvenile idiopathic arthritis (JIA), which is a direct competitor to OTREXUP®. Cumberland Pharmaceuticals, Inc. also recently received FDA approval and launched RediTrex®, a methotrexate injection in the U.S. Competition in the methotrexate market also includes tablets and parenteral dosage forms that are distributed in the U.S. by several generic manufacturers, including Teva, Pfizer, Viatris Inc. (“Viatris”), Hospira and Accord Healthcare. Beyond disease modifying anti-rheumatic drugs (“DMARDs”), other commonly used pharmaceutical treatments for rheumatoid arthritis include analgesics, non-steroidal anti-inflammatory drugs (NSAIDs), corticosteroids and biologic response modifiers. In addition to methotrexate, the other DMARDs include azathioprine (Imuran®), cyclosporine (Neoral®), hydroxychloroquine (Plaquenil®), auranofin (Ridaura®), leflunomide (Arava®) and sulfasalazine (Azulfidine®). The biologic response modifiers include etanercept (Enbrel®), adalimumab (Humira®), golimumab (Simponi®), tocilizumab (Actemra®), certolizumab (Cimzia®), infliximab (Remicade®), abatacept (Orencia®), and rituximab (Rituxan®).
NOCDURNA® (desmopressin acetate) Sublingual Tablets
We market and sell NOCDURNA® (desmopressin acetate) in the U.S., which is the first and only sublingual tablet indicated for the treatment of nocturia due to nocturnal polyuria (“NP”) in adults who awaken at least two times per night to urinate. NOCDURNA® is a sublingual tablet, marketed in two dosage strengths, that dissolves quickly under the tongue without water and has been shown in clinical studies to reduce nighttime urination by nearly half (in patients who average three nighttime bathroom visits.) We license NOCDURNA® from Ferring. We began detailing NOCDURNA® with a soft launch in the fourth quarter of 2020 and reintroduced the product through a re-launch strategy to increase awareness and demand in the first quarter of 2021.
It is estimated that more than 50 million people in the U.S. are affected by nocturia, or frequent waking at night to urinate. Of the approximately 10 million patients diagnosed with nocturia, only about 1.5 million are treated for the condition. One of the leading causes of nocturia is nocturnal polyuria, which is present in up to 88% of nocturia patients. In patients diagnosed with nocturnal polyuria, the kidneys produce too much urine at night. Patients may already be taking medication for overactive bladder (“OAB”) or benign prostatic hyperplasia (“BPH”); however, these medications may not reduce night-time urination because they do not treat NP.
Pharmacological therapy is most useful in treating nocturia due to nocturnal polyuria, including desmopressin, an anti-diuretic hormone therapy. The anti-diuretic effects of desmopressin are mediated by stimulation of vasopressin 2 (“V2”) receptors, thereby increasing water re-absorption in the kidney, and hence reducing urine production. Desmopressin is available as both an oral tablet and a nasal spray. Noctiva™, an FDA-approved nasal formulation of desmopressin acetate, although not currently marketed in the U.S., is the only other FDA-approved branded desmopressin acetate indicated for the treatment of nocturia.
Epinephrine Injection USP
We are the exclusive supplier of the device, which we developed, for Teva’s generic Epinephrine Injection USP products, indicated for emergency treatment of severe allergic reactions including those that are life threatening (anaphylaxis) in adults and certain pediatric patients. Teva’s Epinephrine Injection, utilizing our patented VIBEX® injection technology, was approved by the FDA as a generic drug product with an AB rating, meaning that it is therapeutically equivalent to the branded products EpiPen® and EpiPen Jr® and therefore, subject to state law, substitutable at the pharmacy. We supply the device and Teva is responsible for the drug, assembly and packaging, distribution and commercialization of the finished product, for which we also receive royalties on Teva’s net sales.
Epinephrine is used for the treatment of severe allergic reactions (anaphylaxis) to insect venom, foods, drugs and other allergens as well as anaphylaxis to unknown substances or exercise-induced anaphylaxis. Viatris’s EpiPen®, along with its own authorized generic of the product, continues to be the global market leader in the epinephrine auto injector market. In the U.S., sales of generic epinephrine injection products were approximately $1.66 billion in 2021 based on WAC, according to the IQVIA NSP report. There are other companies and alternative products competing in the U.S. market, including the authorized generic for Adrenaclick® marketed by Amneal Pharmaceuticals, Inc. and Kaléo’s AUVI-Q® (Epinephrine Injection, USP). Auto-Injector in the U.S. beginning in February 2017, Adamis Pharmaceuticals also received FDA approval of SYMJEPI®, an epinephrine injection, which is marketed and distributed in the U.S. by US WorldMeds LLC.
Sumatriptan Injection USP
We, through our partner Teva, sell Sumatriptan Injection USP, indicated in the U.S. for the acute treatment of migraine headaches and cluster headache in adults. Sumatriptan Injection USP is a generic equivalent to Imitrex® STATdose Pen®, and available in the 4 mg/0.5 mL and 6 mg/0.5 mL single-dose pre-filled syringe auto-injectors. We have a license, supply and distribution agreement with Teva, under which Teva is responsible for the manufacture and supply of the drug, and we manufacture the device and complete assembly and packaging of the finished product. Teva is responsible for commercialization and distribution.
The total U.S. retail anti-migraine triptan market was $4.9 billion in 2021 according to IQVIA’s National Prescription Audit® (“NPA”) report based on TRx Pharmacy Dollars. The majority of patients who use triptans take oral tablets. Oral drugs accounted for $4.4 billion of the total, and injectable products accounted for approximately $257 million of the total market, measured in terms of TRx Pharmacy Dollars. Sumatriptan is currently available in an oral formulation, nasal spray and injectable. There is extensive competition in the anti-migraine marketplace and several manufacturers offer versions of injectable drugs with a delivery device, including GSK (Imitrex STATdose Pen®), Teva (AJOVY®), Pfizer (Alsuma), Endo International plc (Sumavel® DosePro®), Sun Pharma (generic sumatriptan autoinjector) and Upsher-Smith (Zembrace® SymTouch®). Sandoz, Inc. (“Sandoz”) also markets an authorized generic version of GSK’s Imitrex STATdose Pen®.
Makena® (hydroxyprogesterone caproate injection) Subcutaneous Auto Injector
We are the exclusive supplier of the device, a variation of our VIBEX® QuickShot® subcutaneous auto injector developed by us, for the progestin hormone drug Makena® (hydroxyprogesterone caproate injection). The Makena® subcutaneous auto injector drug-device combination product is a ready-to-administer treatment indicated to reduce the risk of preterm birth in women pregnant with one baby and who spontaneously delivered one preterm baby in the past. The product was approved by the FDA under the accelerated approval pathway. We are the exclusive supplier of the devices and the final assembled and packaged commercial product.
Makena® is a progestin that belongs to a class of drugs called progestogens. Progestogens have been studied to reduce preterm birth and have shown varying results depending upon the subjects enrolled. The active ingredient in Makena®, 17α hydroxyprogesterone caproate (often referred to as 17P), is the only FDA-approved treatment for pregnant women who have had a prior spontaneous preterm birth (which is a substantial risk factor for recurrent preterm birth) and has been and used for more than a decade by healthcare providers to treat patients with a history of spontaneous preterm birth. The approval of Makena® was based on the landmark Meis trial, conducted by the National Institute of Child Health and Human Development and the Maternal-Fetal Medicine Units Network and published in the New England Journal of Medicine in 2003. The Society for Maternal Fetal Medicine Publications Committee published clinical guidelines for the use of progestogens to reduce the risk of preterm birth in the American Journal of Obstetrics and Gynecology in May 2012, which were affirmed in 2014. Preterm birth is defined as a birth prior to 37 weeks of pregnancy being completed. According to the Centers for Disease Control and Prevention National Center for Health Statistics, the percentage of preterm births affected approximately 10% of births in the U.S. in 2020.
In October 2019, Covis announced that the FDA’s Bone, Reproductive and Urologic Drugs Advisory Committee met to better understand and interpret the PROLONG (Progestin’s Role in Optimizing Neonatal Gestation) confirmatory clinical trial for Makena® (hydroxyprogesterone caproate) injection. Nine advisory committee members voted to recommend that the FDA pursue withdrawal of approval for Makena® and seven committee members voted to leave the product on the market under accelerated approval and require a new confirmatory trial. In October 2020, Covis received notice that the FDA is proposing to withdraw approval of Makena® (hydroxyprogesterone caproate injection). Covis then formally requested a public hearing in response to the FDA’s proposal to withdraw its approval and has stated that it remains committed to working with the FDA to maintain patient access to Makena® as a treatment option to reduce pre-term birth. In August 2021, Covis announced the FDA had granted the request for a public hearing. A date for such meeting has not been set or announced by either the FDA or Covis.
Teriparatide Injection
We are the exclusive supplier of the multi-dose pen, which we developed, used in Teva’s generic teriparatide injection product. In 2020, our partner Teva launched Teriparatide Injection, the generic version of Eli Lilly’s branded product Forsteo® featuring the Antares multi-dose pen platform, for commercial sale in several countries outside of the U.S. Under an exclusive development, license and supply agreement with Teva, Antares is responsible for the manufacturing and supply of the multi-dose pen used in Teva’s generic teriparatide product and Teva is responsible for the sale and distribution of the product. Antares is compensated for devices sold to Teva and is entitled to receive royalties on net product sales by Teva in the territories.
Teriparatide is used for the treatment of osteoporosis in postmenopausal women and men at increased risk of fracture and for glucocorticoid induced osteoporosis in men and women. According to Eli Lilly’s annual report, 2021 global sales of Forteo® were $801.9 million, of which $441.6 million was generated in the U.S.
Products with Tentative Approval
The following is a discussion of our products with tentative approval from the FDA. For a discussion of other approved and marketed commercial products, including proprietary and partnered products and product candidates currently in development, see the “Our Products” and “Research and Development” sections above and below, respectively, included in Item 1 of Part I of this Annual Report on Form 10-K.
TLANDO® (testosterone undecanoate) Oral Formulation
TLANDO® (testosterone undecanoate) is a twice daily oral formulation of testosterone for TRT indicated for conditions associated with a deficiency or absence of endogenous testosterone, or hypogonadism in adult males. TLANDO® was granted tentative approval from the FDA in December 2020 and will be eligible for final approval and marketing in the U.S. upon expiration of the exclusivity period previously granted to Clarus for JATENZO® on March 27, 2022. On February 3, 2022, we announced the FDA’s acceptance of our NDA resubmission for TLANDO® in which the FDA designated the NDA as a Class 1 resubmission with a two-month review goal period and set a target action date of March 28, 2022. We continue to prepare for the launch of TLANDO® in 2022 pending final FDA approval after the expiration of JATENZO®’s exclusivity period.
In the U.S., there are several different formulations for TRT including intramuscular injection, transdermal patches and gels, oral formulations and nasal gel. According to IQVIA National Sales Perspectives® (“NSP”) reporting of nationally projected sales activities, the overall U.S. TRT market was approximately $1.4 billion in 2021 based on wholesale acquisition costs (“WAC”). Total prescriptions in the U.S. TRT market grew by 5.1% to 8.0 million prescriptions in 2021 as compared to 2020, entirely driven by an increase in prescriptions of injectables, which increased by 5.4%. The injectable TRT market grew from $484.4 million in 2020 to $545.0 million in 2021, an increase of 12.5% based on WAC.
Competition in the U.S. testosterone replacement market includes transdermal formulations such as AbbVie’s Androgel® 1% and 1.62%, Perrigo’s generic Androgel® Topical Gel, 1.62%, Eli Lilly’s Axiron®, Endo’s Testim® and Fortesta® and Allergan plc’s (“Allergan”) Androderm®. Other forms of TRT include injectables such as Endo’s Aveed®, Pfizer’s Depo®-Testosterone, and several generic testosterone products sold by Actavis, Sandoz, Viatris Inc., Teva and others, as well as Testopel® pellets by Endo and JATENZO®, an oral formulation, by Clarus. In addition, Marius Pharmaceuticals has submitted an NDA with the FDA for Kyzatrex, an oral formulation of testosterone that is pending final approval.
Research and Development
We are committed to a strong research and development program, recognizing that the development of new product offerings is important to our future success. An important part of our growth strategy is our continued investment in our evolving research and development activities and new product pipeline. While we are focused on opportunities within urology and endocrinology therapeutic areas, we are also exploring new product opportunities beyond these therapeutic areas that could further grow and diversify our portfolio. Our research and development efforts are focused primarily on leveraging our existing product and technology platforms by broadening their applications for use in other drug/device combination products, as well as exploring new pharmaceutical products, technologies and drug delivery methods. We also have a corporate development team that seeks and evaluates new business and product opportunities to further expand our pipeline.
Our research and development programs consist primarily of clinical, regulatory, formulation development, engineering and device development activities for our current products, next generation versions of current products, product extensions, and new proprietary and partnered products and technologies in development. Our internal research and development team works with external consultants, industry experts, physicians and other medical personnel in an effort to drive our product development pipeline. The following is a discussion of our significant research and development activities.
ATRS - 1901
We have initiated development of a proprietary drug device combination product for the urology oncology market, identified as ATRS-1901, and conducted formulation development work and non-clinical studies to help advance this program. In 2020, we received a response from the FDA regarding our pre-IND (Investigational New Drug) submission and believe we have determined our clinical and regulatory pathway forward. Our pre-clinical safety studies are ongoing.
ATRS - 1902
We have identified a program to develop a proprietary drug device combination product for the endocrinology market, an adrenal crisis pen, identified as ATRS-1902. The development program supports a proposed indication for the treatment of acute adrenal insufficiency, known as adrenal crisis, in adults and adolescents, using a novel proprietary auto-injector platform to deliver a liquid stable formulation of hydrocortisone. We have conducted initial formulation work and developed a working prototype of a new device to support this program.
In June 2021, we submitted an IND application with the FDA for the initiation of a Phase 1 clinical study of ATRS-1902 for adrenal crisis rescue. The IND application includes the protocol for an initial clinical study to compare the pharmacokinetic profile of our novel formulation of hydrocortisone versus Solu-Cortef®, which is an anti-inflammatory glucocorticoid and is the current standard of care for the management of acute adrenal crises.
In July 2021, the FDA accepted our IND for ATRS-1902 enabling us to initiate our Phase 1 clinical study. The Phase 1 clinical study designed to evaluate the safety, tolerability and pharmacokinetics (“PK”) of a liquid stable formulation of hydrocortisone was initiated in September 2021. The study is a cross-over design to establish the PK profile of ATRS-1902 (100 mg) compared to Solu-Cortef® (100 mg), the reference-listed drug, in 32 healthy adults. After this study is completed, we expect to conduct a bioequivalence study and second human factor study utilizing our proprietary auto-injector technology.
In January 2022, we announced the positive results from the Phase 1 clinical study and were granted Fast Track designation by the FDA. The positive results support the advancement of our ATRS-1902 development program to a pivotal study for the treatment of acute adrenal insufficiency, known as adrenal crisis, using our Vai™ novel proprietary rescue pen platform to deliver a liquid stable formulation of hydrocortisone. We anticipate starting this pivotal study in adults in the second quarter of 2022 and expect to submit a 505(b)(2) NDA with the FDA by the end of 2022 pending the success of the study, a further human factors study and confirmation of the product stability from our ongoing stability program.
ATRS-1903
We have initiated development of a proprietary drug device combination product utilizing our rescue pen technology for a rare immunology disorder, identified as ATRS-1903. Formulation development work has been conducted and we anticipate progressing this towards initial clinical testing to evaluate PK and tolerability in human subjects.
Additional Development Programs
We continue to pursue and evaluate other potential new products and product extensions that address patient needs primarily in targeted therapeutic areas. We explore new development opportunities including innovative delivery technologies and improved formulations of existing therapeutics.
Partnered Development Projects
We, in collaboration with our pharmaceutical partners, are engaged in research and development activities utilizing our auto injectors and disposable pen injectors. The development programs typically consist of determination of the device design, development of prototype tooling, production of prototype devices for testing and clinical studies, and development of commercial tooling and assembly. We expect development related to these products to continue; however, the development timelines are generally controlled by our partners and the extent of near-term and future development will be dependent on decisions made by our partners. The following is a summary of the development stages for select partnered products in development:
Pen Injector with Teriparatide
We are developing with Teva, under a license, development and supply agreement, a multi-dose disposable pen injector device with teriparatide for the treatment of osteoporosis. Teva is working toward a regulatory approval with the FDA for a generic version of Forteo® (teriparatide rDNA origin injection) using the ANDA pathway. See also the “Teriparatide Injection” section above for more information about the product and Teva’s commercialization activities outside the U.S.
Pen Injector with Exenatide
We were developing with Teva a multi-dose pen injector device for use with a generic form of BYETTA® (exenatide injection) for the treatment of diabetes. Teva was working through the U.S. regulatory approval process for its exenatide pen using the ANDA pathway. On February 25, 2022, Teva notified us that it was terminating the exenatide program and related agreement due to a lack of commercial viability. The termination is effective August 23, 2022.
Rescue Pen (drug undisclosed)
In August 2018, we entered into a development agreement with Pfizer and began developing a combination drug device rescue pen. This rescue pen will use the Antares QuickShot® auto injector and an undisclosed Pfizer drug. In 2021, we continued to work on this development program, and we expect to continue development of this product candidate.
Rescue Device with Selatogrel
In November 2019, we entered into a new global agreement with Idorsia to develop a novel, drug-device product containing selatogrel. A new chemical entity, selatogrel, is being developed for the treatment of a suspected acute myocardial infarction (“AMI”) in adult patients with a history of AMI. Idorsia will pay for the development of the combination product and will be responsible for applying for and obtaining global regulatory approvals for the product. The parties intend to enter into a separate commercial license and supply agreement pursuant to which we will provide fully assembled and labelled product to Idorsia at cost plus mark-up. Idorsia will then be responsible for global commercialization of the product, pending FDA or foreign approval. We will be entitled to receive royalties on net sales of the commercial product.
In 2020, we completed the initial design phase of the selatogrel device and Idorsia conducted a clinical bridging study utilizing these devices. We completed the usability and reliability studies for the device which has been tailored for emergency use ahead of the Phase 3 study.
According to publications by Idorsia, two Phase 2 studies in patients with stable coronary artery disease and acute myocardial infarction, respectively, have met their pharmacodynamic objectives of significantly inhibiting platelet aggregation. Subcutaneous administration of selatogrel 8 mg and 16 mg has demonstrated a rapid onset of action, within 15 minutes, with the height of its effect extending over 4-8 hours, depending on the dose. Selatogrel was safe and well tolerated in both studies and there were no treatment-emergent serious bleeds.
In December 2020, the FDA designated Idorsia’s investigation of selatogrel for the treatment of a suspected AMI in adult patients with a history of AMI as a “fast-track” development program. This designation is intended to promote communication and collaboration between the FDA and pharmaceutical companies for drugs that treat serious conditions and fill an unmet medical need.
In June 2021, Idorsia announced they initiated its Phase 3 registration study to evaluate the efficacy and safety of self-administered subcutaneous selatogrel, Idorsia’s P2Y12 receptor antagonist, in suspected AMI using Antares’ QuickShot® auto-injector. The study is an international, multi-center, double-blind, randomized, placebo-controlled, parallel-group, Phase 3 study to assess the clinical efficacy and safety of 16 mg of selatogrel when self-administered (on top of standard-of-care) upon occurrence of symptoms suggestive of an acute myocardial infarction. The primary efficacy endpoint is the occurrence of death from any cause, or non-fatal AMI after any study treatment self-administration. The study will enroll approximately 14,000 patients who are at high risk of recurrent AMI, at approximately 250 sites in approximately 30 countries. A Special Protocol Assessment has been agreed with the FDA for Idorsia’s selatogrel, which indicates the FDA is in agreement with the adequacy and acceptability of specific critical elements of overall protocol design (e.g., entry criteria, dose selection, endpoints and planned analyses) for a study intended to support a future marketing application.
According to the American Heart Association, the overall prevalence for myocardial infarction in the U.S. is about 7.9 million adults. There are also approximately 805,000 heart attacks in the U.S. annually, of which 605,000 are first heart attacks and 200,000 happen to people who have already had a heart attack according to the Centers for Disease Control and Prevention.
Manufacturing
We use third parties to manufacture our products and product candidates, including the products and related components we supply to our partners. For our products and product candidates, we must ensure they are manufactured in accordance with FDA’s current Good Manufacturing Practices (“cGMPs”) for drug products and FDA’s current Quality System Regulations (“QSRs”) for medical devices and equivalent provisions in the European Union (“EU”) and elsewhere, which are required as part of the overall obligations necessary. We believe that our third-party manufacturers are currently in compliance with cGMPs, QSRs and foreign equivalents, to the extent applicable. Assembly and packaging of our products and product candidates is performed by third-party service providers under our direction. All manufacturers and suppliers are monitored and evaluated by our quality department to assess compliance with regulatory requirements and our internal quality standards and benchmarks. We perform quality reviews of manufacturing for all of our product candidates and products, and quality releases for all of our product candidates and products that we sponsor or commercialize.
We use a range of third-party manufacturers to manufacture and supply certain components, drugs, final assembly and finished product. Below is a summary of our key production, manufacturing, assembly and packaging arrangements with third-party manufacturers for products commercialized by us and our partners:
•We have contracted with Phillips-Medisize Corporation (“Phillips”), an international outsource provider of design and manufacturing services, to produce commercial quantities of our QuickShot® auto injector device for XYOSTED®, our QuickShot® auto injector device for the Makena® product with Covis, and our VIBEX® epinephrine auto injector product with Teva.
•We use ComDel Innovation, Inc. (“ComDel”), a domestic provider of integrated solutions for product development, tooling, and manufacturing, to provide manufacturing services for the VIBEX® sumatriptan auto injector product and for the teriparatide pen product with Teva.
•We have contracted with Jabil Healthcare, an international manufacturing development company to supply commercial quantities of our VIBEX® auto injector device for the OTREXUP® product for Otter and the VIBEX® epinephrine auto injector product with Teva.
•We have contracted with Fresenius Kabi to supply commercial quantities of pre-filled syringes of testosterone for XYOSTED®.
•We have contracted with Ferring for the commercial supply of NOCDURNA®.
•We use Sharp Corporation (“Sharp”), an international contract packaging company, to assemble and package XYOSTED®, Sumatriptan Injection USP and the Makena® auto injector products, and the OTREXUP® auto injector product for Otter®.
Below is a summary of our key production, manufacturing, assembly and packaging arrangements with third-party manufacturers for TLANDO®:
•We use Pfizer to supply the active pharmaceutical ingredient (“API”).
•We have contracted with NextPharma, an international pharmaceutical manufacturing company, to supply the bulk capsule product.
•We use PCI Pharma Services (“PCI”), an international contract packaging company, to assemble and package TLANDO®.
We have an experienced quality group that works with and regularly inspects or meets with our manufacturers and suppliers to review the manufacturing process for our products and product candidates, and to provide input on quality matters.
In addition to the above manufacturing capabilities, on July 1, 2019, we entered into a lease for approximately 75,000 square feet of office, laboratory, manufacturing and warehousing space in Minnetonka, Minnesota. We completed the build-out of the facility and began occupying the space in 2020. The new facility supports our administrative functions, product development and quality operations and is intended to provide additional manufacturing and warehousing capabilities in the future.
Commercial Operations
We have built a robust internal commercial organization, consisting of specialty sales representatives, management and support staff, to market and sell our proprietary products XYOSTED®, OTREXUP® and NOCDURNA® in the U.S. As of December 2021, commercialization and distribution of OTREXUP® is the responsibility of Otter in accordance with the Asset Purchase Agreement. In anticipation of final FDA approval, we are preparing to launch TLANDO® in 2022. We have entered into agreements with vendors for certain commercialization services such as third-party logistics, distribution, data analytics and claims processing. We have and may continue to enter into licensing and or additional distribution arrangements for commercialization of our products outside the U.S.
Distribution – We have contracted with a third-party logistics provider, Cardinal Health 105, Inc., also known as Specialty Pharmaceutical Services (“Cardinal”), for key services related to logistics, warehousing and inventory management, distribution, contract administration and chargeback processing, accounts receivable management and call center management. We also use a division of Cardinal for sample administration. In addition, we use third parties to perform various other services for us relating to regulatory monitoring, including adverse event reporting, safety database management and other product maintenance services.
Trade – We contract with numerous wholesale distributors, including Cardinal, McKesson Corporation (“McKesson”) and AmerisourceBergen Corporation to distribute our proprietary products to retail pharmacies as well as the Veterans Administration and other governmental agencies. In addition to shipping our product, these distributors provide inventory and sales reports as well as other services. In exchange for these services, we pay fees to certain distributors based on a percentage of wholesale acquisition cost. We have also contracted with several specialty pharmacies to support fulfillment of certain prescriptions.
Third Party Reimbursement and Pricing – In the U.S., sales of pharmaceutical products to consumers depend to a significant degree on the availability of coverage and reimbursement by third-party payers, such as government and private insurance plans. Third-party payers are increasingly challenging the pricing of products and services and implementing other cost containment mechanisms, including demanding more aggressive pricing and rebates for favorable formulary placement. This is especially true in markets where generic options exist. Third-party payers often use a tiered reimbursement system and may require step edits or prior authorization. It is time consuming and expensive for us to seek and maintain coverage for our products and to process reimbursements from Medicaid, Medicare and private payers.
Participation in the Medicaid program requires payment of statutory rebates on unit dispenses. Some states have also created Medicaid preferred drug lists and include drugs on those lists only when the manufacturers agree to pay a supplemental rebate. Some States have implemented statutes imposing other consequences for a manufacturer’s failure in certain circumstances to negotiate supplemental rebates, including but not limited to, ordering managed care plans to limit or reduce reimbursement for a drug provided by a medical practitioner. If our products are not included on these preferred drug lists, they may be subject to prior authorization.
Similarly, in order to ensure coverage by Medicare Part D and commercial pharmacy benefit plans, we participate in certain rebate programs, which provide discounted prescriptions to qualified insured patients. Under these rebate programs, we pay a rebate to the third-party administrator of the program. We also provide discounts to authorized users of the Federal Supply Schedule (“FSS”) of the General Services Administration under an FSS contract negotiated by the Department of Veterans Affairs, including discounts mandated by the Veterans Health Care Act, discounted prescriptions to Department of Defense’s (“DoD”) Tricare retail pharmacy program, and statutory discounts to federal grantees and safety net providers referred to as covered entities pursuant to our pharmaceutical pricing agreement with the Department of Health and Human Services and the 340B drug discount program, which is required as a condition of Medicaid coverage. Government agencies ordering under the FSS and covered 340B entities purchase products from the wholesale distributors at the discounted price, and the wholesale distributors then charge back the difference between the current wholesale acquisition cost and the price the entity paid for the product.
We also offer co-pay assistance programs to patients for our proprietary products under which patients covered by commercial pharmacy benefit plans receive discounts on their prescriptions. Our XYOSTED® STEADYCare Co-pay Assistance Program provides financial support to most commercially insured patients to assist with out-of-pocket costs of XYOSTED®. In addition, certain commercially insured patients are eligible for our “first fill free” program for XYOSTED® to assist the patient during the initial claims adjudication process. Similar to XYOSTED®, we offer a co-pay assistance program for NOCDURNA®, which also provides financial support to most commercially insured patients to assist with out-of-pocket costs. We use contract service providers to process and pay claims to patients for actual usage. We also offer the ability for patients who do not have insurance, or whose insurance does not cover our proprietary products, the ability to purchase either XYOSTED® or NOCDURNA® with a valid prescription at a cash price via a specialty pharmacy.
International Distribution – We are contracting with a third-party logistics provider, Cardinal, for key services related to logistics, warehousing and inventory management, international shipping, export and customs administration to support our international distributor, Lunatus. We entered into an exclusive distribution agreement with Lunatus in August 2020 to distribute and promote the sale of XYOSTED® in Saudi Arabia and the United Arab Emirates. Lunatus is responsible for obtaining regulatory approval and, assuming approval, for the promotion and commercialization of the product in the territories.
Sales, Marketing & Distribution of Partnered Products
Our partnered products may encounter some of the same reimbursement issues described above, and although we do not control the reimbursement rate or discounts contracted with third-party payers by our partners, it ultimately affects our royalty payments we receive on net sales. The industry has experienced an increasingly widening gap between gross sales and net sales after discounts.
Epinephrine Injection USP – We are the exclusive supplier of the device used in Teva’s epinephrine injection product. We receive payment for each device sold to Teva and royalties on Teva’s commercial sales of the product. Teva’s epinephrine injection was approved as a generic drug product with an AB rating, meaning that it is therapeutically equivalent to Viatris Inc.’s branded products EpiPen® and EpiPen Jr® and therefore, subject to state law, is substitutable at the pharmacy. Teva is solely responsible for commercialization and distribution of the finished product.
Makena® Subcutaneous Auto Injector – We are the exclusive supplier of the device used in the Makena® subcutaneous auto injector. We receive payment for each device sold to Covis and royalties on Covis’ commercial sales of the product. Covis primarily sells Makena® to specialty pharmacies, specialty distributors, home infusion companies and pharmacies which, in turn, sell Makena® to healthcare providers, hospitals, government agencies and integrated delivery systems. Covis is solely responsible for commercialization and distribution of the finished product.
Sumatriptan Injection USP – We are the exclusive supplier of the product containing sumatriptan which is commercialized by Teva. We are compensated at cost for shipments of product to Teva. In addition, net profits from sales of the product, after deduction of product sales allowances such as discounts, rebates and chargebacks, are split 50/50 between us and Teva. Teva is solely responsible for commercialization and distribution of the finished product.
Teriparatide Injection – We are the exclusive supplier of the pen injection device used in Teva’s Teriparatide Injection product outside the U.S. Teva launched its generic version of Forsteo® in certain territories outside the U.S. in 2020. We receive payment for each device sold to Teva and royalties on Teva’s commercial sales of the product in the territories. Teva is solely responsible for commercialization and distribution of the finished product.
OTREXUP® (methotrexate) Injection – Pursuant to the Asset Purchase Agreement, license agreement and supply agreement, we are the exclusive supplier of the device used in OTREXUP® beginning in December 2021. We receive payment for each device sold to Otter. Otter is solely responsible for commercialization and distribution of the finished product.
Information about Revenues and Customer Concentrations
For information about revenues and customer concentrations, see Item 7 of Part II of this Annual Report on Form 10-K. Significant customers from which we derive 10% or more of our total revenue in each or any of the years in the three-year period ended December 31, 2021 include: Teva, McKesson, AmerisourceBergen Corporation, Cardinal Health and Covis. For more detailed information, see Note 14 – Revenues, Significant Customers and Concentrations of Risk in the Notes to Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K.
Collaborative Arrangements and License Agreements
We have entered into significant partnering arrangements and licensing agreements with Teva, Covis, Pfizer, Idorsia and other pharmaceutical partners. The following is a summary of certain agreements.
Teva License, Development and Supply Agreements
In July 2006, we entered into an exclusive license, development and supply Agreement with Teva for an epinephrine auto injector product to be marketed in the U.S. and Canada. Pursuant to the agreement, Teva is obligated to purchase all of its delivery device requirements from us. We received an upfront cash payment and a milestone payment upon FDA product approval. We also receive a negotiated purchase price for each device sold, as well as royalties on Teva’s commercial sales of the product. This agreement has been amended to provide for payment of capital equipment and other ongoing development work that was outside the scope of the original agreement. The agreement will continue until the expiration of the last to expire patent that is filed no later than 12 months after FDA approval. We have multiple patents that have been granted by the United States Patent and Trademark Officer (“USPTO”) that cover this product, the latest of which will expire in 2033. We have and plan to continue to file patent applications covering this product.
In December 2007, we entered into a license, development and supply agreement with Teva under which we developed and will supply a disposable pen injector for two therapeutic products: exenatide and teriparatide. Under the agreement, we received an upfront payment and development milestones, and may receive royalties on future product sales. This agreement has been amended numerous times and provides for payment of capital equipment and other development work that was outside the scope of the original agreement. This agreement will continue until the expiration date of the last to expire patent covering the device or product that is filed no later than 12 months after FDA approval and will be automatically renewed for successive periods of two years each. Currently, the expiration date of the last to expire patent is 2035. On February 25, 2022, Teva notified us that it was terminating the exenatide program and related agreement due to a lack of commercial viability. The termination is effective August 23, 2022.
In November 2012, we entered into a license, supply and distribution agreement with Teva for an auto injector product containing sumatriptan for the treatment of migraines. Under the agreement, we received an upfront payment and a milestone payment upon commercial launch. Teva is responsible for the manufacture and supply of the drug, and we are responsible for the manufacture and supply of the device and assembly and packaging of the finished product. We are compensated at cost for product shipment to Teva and Teva distributes the product in the U.S. Teva also received an option for distribution rights in other territories. In addition, net profits are split 50/50 between us and Teva. The term of the agreement continues seven years from commercial launch, which was in June 2016, with automatic one-year renewals unless terminated sooner by either party in accordance with the terms of the agreement.
Covis Agreements
In September 2014, we entered into a development and license agreement with Lumara Health, Inc., which was subsequently acquired by AMAG, which was subsequently acquired by Covis, to develop and supply an auto injector system for use with Makena®, a progestin drug (hydroxyprogesterone caproate) indicated to reduce the risk of preterm birth. Under the agreement, we granted an exclusive, worldwide, royalty-bearing license, with the right to sublicense, to certain intellectual property rights, including know-how, patents and trademarks, and received an upfront payment for our license and development activities. We are also entitled to milestone payments upon the achievement of pre-determined amounts of net sales of the product.
Covis was responsible for the clinical development and preparation, submission and maintenance of all regulatory applications, and is responsible for the manufacture and supply of the drug to be used in the product, and to market, distribute and sell the product. We are the exclusive supplier of the auto injection system devices for the product and are responsible for the manufacture and supply of the devices and final assembly and packaging of the finished product. Under the arrangement, we receive payment for each device, and royalties based on net sales of products commencing on product launch in a particular country until the product is no longer developed, marketed, sold or offered for sale in such country. The royalty rates range from high single digit to low double digits and are tiered based on levels of net sales of products and decrease after the expiration of licensed patents or where there are generic equivalents to the auto injector product being sold in a particular country.
In March 2018, we entered into a manufacturing agreement with Covis for the exclusive supply of the devices and fully assembled and packaged final finished product of the Makena® subcutaneous auto injector. The term of the agreement is concurrent with the term of the development and license agreement and will continue until such time as commercialization of the product is halted. We receive a contracted price per unit on each product manufactured.
Pfizer Agreement
In August 2018, we entered into a development agreement with Pfizer to jointly develop a combination drug device rescue pen. This rescue pen will use the Antares QuickShot® auto injector and an undisclosed Pfizer drug. In 2021, we continued to work on this development program, and we expect to continue development of this product candidate.
Idorsia Agreement
In November 2019, we entered into a new global agreement with Idorsia to develop a novel, drug-device product containing selatogrel. A new chemical entity selatogrel is being developed for the treatment of a suspected AMI in adult patients with a history of AMI. Idorsia will pay for the development of the combination product and will be responsible for applying for and obtaining global regulatory approvals for the product. The parties intend to enter into a separate commercial license and supply agreement pursuant to which Antares will provide fully assembled and labelled product to Idorsia at cost plus mark-up. Idorsia will then be responsible for global commercialization of the product, pending FDA or foreign approval. Antares will be entitled to receive royalties on net sales of the commercial product.
Ferring Agreement
In October 2020, we entered into an exclusive license and commercial supply agreement with Ferring for the marketed product NOCDURNA® (desmopressin acetate) in the U.S., which is indicated for the treatment of nocturia due to nocturnal polyuria (NP) in adults who awaken at least two times per night to urinate. Under the terms of the license agreement, the Company paid Ferring an upfront payment of $5.0 million upon execution and paid an additional $2.5 million on October 1, 2021. Ferring is eligible for tiered royalties and additional commercial milestone payments potentially totaling up to $17.5 million based on our net sales of NOCDURNA® in the U.S.
Lipocine Agreement
In October 2021, we entered into an exclusive license agreement with Lipocine for the product TLANDO® (testosterone undecanoate) in the U.S., a twice-daily oral formulation of testosterone for testosterone replacement therapy indicated for conditions associated with a deficiency or absence of endogenous testosterone, or hypogonadism in adult males. TLANDO® was granted tentative approval from the FDA in December 2020 and will be eligible for final approval and marketing in the U.S. upon expiration of the exclusivity period previously granted to Clarus for JATENZO® on March 27, 2022. On February 3, 2022, we announced the FDA’s acceptance of our NDA resubmission for TLANDO® with a target action date set for March 28, 2022. We continue to prepare for the launch of TLANDO® in 2022 pending final FDA approval after the expiration of JATENZO®’s exclusivity period. Under the terms of the license agreement, we paid Lipocine an upfront payment of $11.0 million. Lipocine is eligible for additional milestone payments up to $10.0 million and tiered royalty and commercial milestones based on net sales of TLANDO® in the U.S. We will be responsible for the manufacturing and commercialization of TLANDO®.
The license agreement also grants us the option to license and develop LPCN 1111 (TLANDO XR) in the U.S., a potential once daily oral testosterone product containing testosterone tridecanoate in development for the treatment of hypogonadism in adult males. Results of the Phase 2b study for TLANDO XR met its primary endpoints, including identifying the dose expected to be tested in a Phase 3 study. TLANDO XR was well tolerated with no drug-related severe or serious adverse events reported and the target Phase 3 dose also met its primary and secondary endpoints in the Phase 2b study. TLANDO XR is an investigational drug containing tridecanoate and has not been approved by the FDA, nor has the name been approved. If elected, upon exercise of the option, we will be required to pay an additional $4.0 million in license fees in two installments and will be responsible for additional development and commercial milestone payments as well as tiered royalties on net sales of TLANDO XR in the U.S. In addition, we will be responsible for completing the development program including the conduct of a Phase 3 clinical trial and applying for regulatory approval in the U.S.
Otter Agreement
In December 2021, we entered into an Asset Purchase Agreement with Otter to sell certain worldwide assets used in the operation of the OTREXUP® product line for $44.0 million, subject to finalization of changes in closing inventory to be transferred, and a license agreement for rights to commercialize OTREXUP®. Simultaneously, we entered into a supply agreement with Otter to manufacture the VIBEX® auto-injection system device, designed and developed to incorporate a pre-filled syringe for delivery of methotrexate, assemble, package, label and supply the final OTREXUP® product and related samples to Otter at cost plus mark-up. Otter is responsible for manufacturing, formulation and testing of methotrexate and the corresponding pre-filled syringe for assembly with the device manufactured by us, along with the commercialization and distribution of OTREXUP®.
Seasonality of Business
Certain parts of our business may be affected by seasonality. Customer purchases have historically been lower in the first quarter of the year due to the resetting of high-deductible health insurance plans. Seasonality affects quarterly comparisons within any fiscal year; however, we believe this impact is generally not material to our annual consolidated results. Our revenues may be influenced by many factors, including regulatory and reimbursement approvals, timing of product launches, acquisitions or divestitures, holiday schedules, and other macro-economic conditions.
Competition
The pharmaceutical and medical device industries are intensely competitive and subject to rapid and significant technological change. We have a wide range of competitors depending upon the branded or generic marketplace, the therapeutic product category, and the product type, including dosage strengths and route of administration. Our competitors include established specialty pharmaceutical companies, major brand name and generic manufacturers of pharmaceuticals such as Teva, Viatris, Eli Lilly and Endo, as well as a wide range of medical device companies that sell a single or limited number of competitive products or participate in only a specific market segment. Our competitors also include third party contract medical device design and development companies such as Scandinavian Health Ltd., Ypsomed AG, West Pharmaceutical and Owen Mumford Ltd. Many of our competitors have greater financial and other resources than we have, such as more commercial resources, larger research and development staffs and more extensive marketing and manufacturing organizations. Smaller or early stage emerging companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies.
If competitors introduce new products, delivery systems or processes with therapeutic or cost advantages, our products can be subject to progressive price reductions or decreased volume of sales, or both. Branded products not only face competition from other brands, but also from generic versions. Generic versions are generally significantly less expensive than branded versions, and, where available, may be required in preference to the branded version under third-party reimbursement programs, or substituted by pharmacies. Most new products that we introduce must compete with other products already on the market or products that are later developed by competitors. Manufacturers of generic pharmaceuticals typically invest far less in research and development than research-based pharmaceutical companies and therefore can price their products significantly lower than branded products. Accordingly, when a branded product loses its market exclusivity, it normally faces intense price competition from generic forms of the product. To successfully compete for business with managed care and pharmacy benefit management organizations, we must often demonstrate that our products offer not only medical benefits but also cost advantages as compared with other forms of care.
Newly introduced generic products with limited or no other generic competition typically command higher prices initially. At the expiration of the exclusivity period, other generic distributors may enter the market, resulting in a significant price decline for the drug. As a result, the maintenance of profitable operations in generic pharmaceuticals depends, in part, on our ability to select, develop and launch new generic products in a timely and cost-efficient manner and to maintain efficient, high quality manufacturing capabilities.
Industry Trends
Based upon our experience, we believe the following significant trends have important implications for the growth of our business. Recent trends in the pharmaceutical industry include merger and acquisition activity leading to further market consolidation. In many cases, the resulting combined pharmaceutical companies are bigger and have more financial, technical and market strength and greater resources, which increases competitive pressure in the industry.
There is ongoing effort by public and private payers to reduce the cost of drugs and reduce the overall cost of health care. There continues to be greater pressure on drug manufacturers to provide greater discounts and rebates on their products. The drug distribution channels are complex and involve many different parties. Recently, such channels have undergone and continue to undergo consolidation. Drug wholesalers and retail drug chains have merged or consolidated resulting in significantly larger organizations with greater resources and bargaining power controlling multiple levels of the drug distribution network. Consequently, pharmaceutical companies are facing increasing pressure to reduce prices. Additionally, the emergence of large buying groups representing independent retail pharmacies and other drug distributors, and the prevalence and influence of managed care organizations and similar institutions, enable those groups to demand larger price discounts on our products. Large wholesalers and retailer customers have continued to form partnerships. As a result of this consolidation among wholesale distributors as well as the growth of large retail drug store chains, a small number of large wholesale distributors control a significant share of the market.
Government Regulation
Any potential products discovered, developed and manufactured by us or our collaborative partners must comply with comprehensive regulation by the FDA in the U.S. and by comparable authorities in other countries. These national agencies and other federal, state, and local entities regulate, among other things, the pre-clinical and clinical testing, safety, effectiveness, approval, manufacturing operations, quality, labeling, distribution, controlled substance security, export, import, storage, record keeping, safety and other reporting, sampling, advertising, marketing, and promotion of pharmaceutical products and medical devices. Facilities and certain company records are also subject to inspections by the FDA and comparable authorities or their representatives.
The FDA has broad discretion in enforcing the Federal Food, Drug and Cosmetic Act (“FFDCA”) and the regulations thereunder, and noncompliance can result in a variety of regulatory enforcement actions ranging from warning letters, product detentions, device alerts or field corrections to recalls, seizures, manufacturing shut downs, quarantines, refusal of the government to approve NDAs or ANDAs, or supplements to the same, clinical holds, injunctive actions, withdrawal of approvals, civil or criminal actions or penalties, disgorgement, adverse publicity, labeling revisions, dear healthcare provider letters, FDA debarment, exclusion from Federal healthcare programs, contract debarment or refusal of future orders under existing government contracts, consent decrees, and corporate integrity agreements. Furthermore, new government requirements may be established that could delay or prevent regulatory approval of our products under development.
Drug Approval Process
FDA approval of our own and our partners’ products is required before the products may be commercialized in the U.S. Section 505 of the FFDCA describes three regulatory pathways for marketing authorization for a new drug:
•A 505(b)(1) NDA is an application that is used for the approval of a new drug that contains full reports of investigations of safety and effectiveness.
•A 505(b)(2) NDA is an application where at least some of the information required for approval comes from studies not conducted by or for the applicant and for which the applicant has not obtained a right of reference or use from the person by or for whom the investigations were conducted. This alternate route for regulatory approval permits the applicant to rely in part upon the FDA’s findings of safety and effectiveness for previously approved products and/or published scientific literature.
•Section 505(j) establishes an abbreviated approval process for generic versions of approved drug products through the submission of an ANDA. An ANDA provides for marketing of a drug product that has the same active ingredients in the same strengths, route of administration, and dosage form as the listed drug, which has the same labeling, performance, characteristics, and intended use as the listed drug, and has been shown to be bioequivalent to the listed drug. ANDA applicants are generally required to conduct bioequivalence testing to confirm pharmaceutical and therapeutic equivalence to the branded reference drug. Generic versions of drugs can often be substituted by pharmacists under prescriptions written for the branded reference drug, pursuant to state laws.
For both NDAs and ANDAs, the FDA reviews applications to determine, among other things, whether a product is safe and effective for its intended use and whether the manufacturing methods and controls are adequate to assure and preserve the product’s identity, strength, quality, safety, potency, and purity. The following table provides a summary description of the various regulatory pathways:
| | | | | | | | | | | | | | | | | |
| ANDA | | 505(b)(2) NDA | | 505(b)(1) NDA |
Clinical Trials/Testing Required | Generally, bioequivalence. | | Yes, to address potential differences between the branded reference product and the 505(b)(2) product, as well as bridging studies. | | Yes, full reports of safety and efficacy. |
Results in Orange Book Listed Patents | No | | Yes, for novel formulations, other enhancements and new indications. | | Yes |
| Exclusivity | Potential for 180 days against other generic filers if first generic to file a substantially complete application containing a paragraph IV certification that is lawfully maintained. | | Potential for 30-month stay if ANDA or 505(b)(2) applicant citing our or our partners’ product as a reference listed drug includes a paragraph IV certification. Also, potential for three- or five-year exclusivity, like 505(b)(1) NDAs. | | Potential of five years for a new chemical entity, or three years for new clinical investigations (other than bioavailability and bioequivalence studies) that are essential to approval of the application. Potential for 30-month stay if ANDA or 505(b)(2) applicant citing our or our partners’ product as a reference listed drug includes a paragraph IV certification. |
| Patent Certification Required | Yes | | Yes | | No |
Potential Orphan Drug Designation Drug Status | No | | Yes | | Yes |
NDA Submission
The process required by the FDA before a new drug pharmaceutical product or a change to an already approved pharmaceutical product, may be approved for marketing in the U.S. generally involves:
•pre-clinical laboratory and animal tests;
•submission to the FDA of an Investigational New Drug (“IND”) application, which must be in effect before clinical trials may begin;
•adequate and well controlled human clinical trials to establish the safety and efficacy of the drug for its intended indication(s);
•development of manufacturing processes to ensure the drug’s identity, strength, quality and purity;
•submission to the FDA of an NDA;
•FDA compliance inspections and/or clearance of all manufacturers and facilities, as well as select clinical trial sites; and
•FDA review of the NDA in order to determine, among other things, whether the drug is safe and effective for its intended uses.
The preclinical and clinical testing and approval process takes many years and the actual time required to obtain approval, if any, may vary substantially based upon the type, complexity and novelty of the product or disease. Preclinical tests include laboratory evaluation of product chemistry, formulation and toxicity, as well as animal studies to assess the characteristics and potential safety and efficacy of the product. The conduct of the preclinical tests must comply with federal regulations and requirements, including cGLPs. The results of preclinical testing are submitted to the FDA as part of an IND, to support human clinical trials along with other information, including information about product chemistry, manufacturing and controls, available scientific literature, and a proposed clinical trial protocol. Some preclinical testing may continue even after the IND is submitted. In the case of drug product candidates for which the sponsor will seek marketing approval via a 505(b)(2) NDA application, some of the above information may be abbreviated or omitted.
IND applications automatically become effective 30 days after receipt by the FDA, unless the FDA places the clinical trial on a clinical hold. If the FDA places a trial on clinical hold, the sponsor must address the issue to the FDA’s satisfaction before the trial may begin. In addition, an independent Institutional Review Board (“IRB”) must review, approve and monitor the plan for any clinical trial, subject communications, and informed consent information for subjects before the trial commences. The FDA, the IRB or the sponsor may suspend a clinical trial, place a trial on hold or discontinue a trial at any time on various grounds.
Once an IND is in effect, each new clinical protocol and any amendments to the protocols must be submitted to the IND for FDA review, and to the IRB for approval. Progress reports detailing the results of the clinical trials must also be submitted at least annually to the FDA and the IRB and more frequently if serious adverse events or other significant safety information is found.
Sponsors of clinical trials generally must register and report, at the NIH-maintained website ClinicalTrials.gov, key parameters of certain clinical trials, including clinical trial results within set timeframes, with the exception of PK studies. Failure to submit the required information to ClinicalTrials.gov can result in monetary penalties. Investigators must also provide certain information to the clinical trial sponsors to enable sponsors to make certain financial disclosures to the FDA. Moreover, under the 21st Century Cures Act, manufacturers or distributors of investigational drugs for the diagnosis, monitoring or treatment of one or more serious diseases or conditions must have a publicly available policy concerning expanded access to investigational drugs.
Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients in accordance with the applicable protocol and all applicable laws, rules and regulations. Clinical trials are typically conducted in sequential phases, which may overlap, though in the case of a 505(b)(2) NDA, some study requirements may be abbreviated. Studies, in addition to the below, such as pediatric studies, may also be required by the FDA:
•Phase I – During Phase I, when the drug is initially given to human subjects, the product is tested for safety, dosage tolerance, absorption, distribution, metabolism and excretion. Phase I studies are often conducted with healthy volunteers depending on the drug being tested. If possible, Phase I trials may also be used to gain an initial indication of product effectiveness.
•Phase II – Phase II involves controlled studies in a limited patient population, typically patients with the conditions needing treatment, to evaluate preliminarily the efficacy of the product for specific, targeted indications; determine dosage tolerance and optimal dosage; and identify possible adverse effects and safety risks.
•Pivotal or Phase III – Adequate and well-controlled trials are undertaken in Phase III in order to evaluate efficacy and safety in a comprehensive fashion within an expanded patient population for seeking approval of the new drug. Typically, two Phase III trials are required by FDA for product approval.
In the case of 505(b)(2) NDAs, the above studies may be abbreviated. Following marketing approval, sponsors may also voluntarily or be required to conduct additional studies, called Phase IV studies.
In some cases, FDA programs may be available to expedite or simplify the process of drug development and FDA marketing application review. For instance, drug products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive accelerated approval, which means the FDA may approve the product based upon a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity or prevalence of the condition and the availability or lack of alternative treatments. A drug candidate approved on this basis is subject to the FDA’s prior review of promotional materials. Accelerated approval products are also required to conduct rigorous post-marketing compliance requirements, including the completion of Phase IV or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, will allow the FDA to withdraw the drug or biologic from the market on an expedited basis. If the FDA proposes withdrawing an accelerated approval, the agency provides the applicant with an opportunity for a hearing. If the applicant files a timely request for a hearing, the applicant must submit any data and information to the FDA upon which it plans to rely. At the hearing, an advisory committee is asked to review the applicable issues and provide advice and recommendations to the FDA.
Sponsors may also request that a product be designated under the FDA’s fast track program. Under this program, products that are intended to treat a serious or life-threatening disease or condition and demonstrates the potential to address an unmet medical need, if so designated by the FDA, are eligible for more frequent development meetings and correspondence with the FDA. In addition, the FDA may initiate review of sections of an application before the application is complete. This “rolling review” is available if the applicant provides and the FDA approves a schedule for the remaining information. In some cases, a fast-track product may be eligible for other FDA programs intended to expedite product development and approval for serious and life-threatening diseases. Notably, however, such designations and programs may be withdrawn by the FDA and such designations and programs do not guarantee that a product will ultimately be successfully developed or approved for marketing.
Another program that is intended to facilitate development is the special protocol assessment (“SPA”) program. Under this program, a sponsor may be able to request a special protocol assessment, or SPA to reach agreement with the FDA on certain studies. If a written agreement is reached regarding the applicable study protocol, the agreement will be binding on the FDA and the protocol may not be changed by the sponsor or the FDA after the trial begins except with the written agreement of the sponsor and the FDA or if the FDA determines that a substantial scientific issue essential to determining the safety or efficacy of the product candidate was identified after the testing began. An SPA is not binding if new circumstances arise, and there is no guarantee that a study will support an approval even if the study is subject to an SPA.
In addition, under the Pediatric Research Equity Act, or PREA, an NDA or supplement to an NDA for a new active ingredient, indication, dosage form, dosage regimen, or route of administration must contain data that is adequate to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults, or full or partial waivers from the pediatric data requirements.
The results of drug development, pre-clinical studies and clinical trials are submitted to the FDA as part of an NDA. NDAs also must contain extensive chemistry, manufacturing and control information. In most cases, the submission of an NDA is subject to a substantial application user fee. Fee waivers or reductions are available in certain circumstances.
Once the FDA receives an application, it has 60 days to review the NDA to determine if it is substantially complete to permit a substantive review and will be accepted for filing. The FDA may request additional information rather than accept an NDA for filing. Once the submission is accepted for filing, the FDA’s goal is to review 90% of all applications for non-New Molecular Entities (“NMEs”) within ten months from the submission date. For NMEs, the FDA has the goal of completing its review of 90% of applications within ten months of the 60-day filing date. The FDA, however, may give a priority review designation to drugs that are intended to treat serious conditions and, if approved, would provide significant improvements in the safety or effectiveness of the treatment, diagnosis, or prevention of the serious conditions. A priority review means that the goal for the FDA is to review an application within six months of the submission date for non-NMEs and within six months of the 60-day filing date for NMEs. These timeframes, however, are only goals, which the FDA may not meet. Moreover, the review process may also be extended if the FDA requests or the NDA sponsor otherwise provides substantial additional information or clarification regarding the submission.
The FDA may also choose or be required to refer drugs to advisory committees when it is determined that an advisory committee’s expertise would be beneficial to the regulatory decision-making process, including the evaluation of new technology. An advisory committee is a panel that includes clinicians and other experts, which review, evaluate, and make a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
After evaluating the NDA and all related information, and inspection reports regarding the manufacturing facilities and clinical trial sites, the FDA may issue an approval letter authorizing marketing for specific conditions, or, in some cases, a Complete Response Letter (“CRL”) describing the application deficiencies. If a CRL is issued, the applicant may either resubmit the NDA addressing all of the deficiencies identified in the letter; withdraw the application or request an opportunity for a hearing. The FDA has the goal of reviewing 90% of application resubmissions in either two or six months of the resubmission date, depending on the kind of resubmission.
If a product meets the FDA’s approval standards but the approval of the product cannot be made effective before the expiration of patents or exclusivities held by the reference listed drug, the FDA may issue a tentative approval letter to the applicant. Under these circumstances, the FDA delays the final approval of the product until all patent or exclusivity issues have been resolved. Products with tentative approvals may not be marketed until the FDA issues a final approval. Following a tentative approval, applicants may amend their application; however, amendments may delay the FDA’s ability to finalize the approval as the FDA must review and approve the amendment. In order to obtain final approval, the applicant must submit a request to the FDA for final approval (which we did for TLANDO® on January 28, 2022 with acceptance by the FDA announced on February 3, 2022). The FDA’s review of requests for final approval may take a number of months and depends on the content of the request and the kind of application. Even if a product has received tentative approval, there is no guarantee that the product will receive final approval, as new information may emerge that changes the FDA’s prior determination.
ANDA Submissions
A pharmaceutical company seeking to market a generic version of a branded drug must file an ANDA with the FDA. For ANDAs, applicants are not required to conduct complete clinical studies. Such applications, though, normally require bioavailability and/or bioequivalence studies conducted in accordance with Good Clinical Practices (“GCPs”) and under the supervision of an IRB.
Like NDAs, ANDAs must be accompanied by user fees. For generic drugs, other fees, such as fees for drug master files, program fees and fees for manufacturing facilities, may also be required to be paid by the applicant, manufacturer, and/or drug master file holder.
Following submission of an ANDA, the FDA has 60 days to evaluate the application to determine if it is substantially complete. If the agency finds that the application is substantially complete, it will receive the application and begin its substantive review. As part of this substantive review, the FDA will determine whether or not the generic version submitted by the company meets the necessary approval standards, including bioequivalence to the reference listed drug, adequate chemistry, manufacturing and controls, and manufacturing facilities and clinical study sites passing pre-approval inspections. Under the FDA’s Generic Drug User Fee Act performance goals, the FDA has the goal of reviewing and acting on 90% of standard original ANDAs within ten months of submission; however, certain factors can lengthen or shorten this review timeline.
Following its completion of the review of an ANDA, the FDA will either issue an approval letter authorizing marketing for specific conditions, or a CRL. If a CRL is issued, the applicant may either respond to the FDA addressing all of the deficiencies identified in the letter, withdraw the application or request an opportunity for a hearing. The FDA may also tentatively approve an ANDA application, as described above.
Upon approval, the FDA will rate generic drug products in the Orange Book. Products meeting bioequivalence standards will typically receive an AB rating. Under state law, such generic drug products may be able to be substituted at the pharmacy for the brand-name drug without the intervention of the prescribing physician, unless otherwise specified by the patient or physician. Many third-party payers of prescription drugs (e.g., health insurance plans, Medicare and Medicaid programs) have adopted policies to encourage the substitution of the lower-priced AB-rated generic drugs for the higher-priced branded drugs when an AB-rated generic drug is available as generic drugs are sold generally at prices below those of the corresponding branded products. Generic drugs may provide a cost-effective alternative for consumers while maintaining the same active ingredient(s), dosage form, strength, route of administration, and conditions of use as the branded product.
Generally Applicable Requirements
Clinical trials for all product candidates must be conducted in accordance with GCPs, which include the requirements that all research subjects provide their informed consent in writing for their participation in any clinical trial as well as review and approval of the study by an IRB. Before approving an application, the FDA may inspect one or more clinical trial sites to assure compliance with GCPs.
Further, during development, the manufacture of investigational drugs for the conduct of human clinical trials is subject to cGMP requirements. Investigational drugs and active pharmaceutical ingredients imported into the U.S. are also subject to regulation by the FDA relating to their labeling and distribution. Further, the export of investigational drug products outside of the U.S. is subject to regulatory requirements of the receiving country as well as U.S. export requirements under the FDCA.
For both NDAs and ANDAs, the FDA also may require submission of a risk evaluation and mitigation strategy (“REMS”) to ensure that the benefits of the drug outweigh the risks of the drug. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further testing requirements and FDA review and approval. Further, should new safety information arise, additional testing, product labeling or FDA notification may be required.
The Hatch-Waxman Amendments, Regulatory Exclusivity, and Patent Term Extension
Orange Book Patent Listing
When an NDA is submitted to the FDA seeking approval of a drug, including a 505(b)(2) NDA, the applicant is required to list certain patents whose claims cover the applicant’s product or method of use with the FDA. Upon approval of an NDA, each of the patents listed in the application for the drug is then published in the Orange Book. In an effort to clarify which patents must be listed in the Orange Book, in January 2021, Congress passed the Orange Book Transparency Act of 2020, which largely codifies the FDA’s existing practices into the FDCA.
The Orange Book listed NDA products may be cited by potential competitors in support of approval of an ANDA or 505(b)(2) NDA. Any applicant who files an ANDA seeking approval of a generic equivalent version of a drug listed in the Orange Book or a 505(b)(2) NDA referencing a drug listed in the Orange Book must certify to the FDA that: (1) no patent information on the drug product that is the subject of the application has been submitted to the FDA; (2) such patent has expired; (3) the date on which such patent expires and approval will not be sought until after the patent expiration; or (4) such patent is invalid or will not be infringed upon by the manufacture, use or sale of the drug product for which the application is submitted. This last certification is known as a paragraph IV patent certification. The applicant may also elect to submit a “section viii” statement certifying that its proposed label does not contain (or carves out) any language regarding the patented method-of-use rather than certify to a listed method-of-use patent. Generally, the ANDA or 505(b)(2) NDA approval cannot be made effective by FDA until all listed patents have expired, except where the ANDA or 505(b)(2) NDA applicant challenges a listed patent through a paragraph IV certification or if the applicant is not seeking approval of a patented method of use.
If the ANDA or 505(b)(2) applicant makes a paragraph IV certification challenging an Orange Book-listed patent, a notice of the paragraph IV certification must be provided to each owner of the patent that is the subject of the certification and to the holder of the approved NDA to which the ANDA or 505(b)(2) application refers. If the NDA holder or patent owners of the listed drug asserts infringement of the patent in court within 45 days of the receipt of the paragraph IV certification notice, the FDA is prohibited from making the approval of the ANDA or 505(b)(2) application effective until the earlier of 30 months from the receipt of the paragraph IV certification, the expiration of the patent, the settlement of the lawsuit, a decision in the infringement case that is favorable to the applicant, or such shorter or longer period as may be ordered by a court. The ANDA or 505(b)(2) application approval also will not be made effective until any applicable non-patent exclusivity listed in the Orange Book has expired as described in further detail below.
Recently, Congress, the Administration and administrative agencies have introduced and/or taken certain measures to increase drug competition and thus, decrease drug prices, including with respect to drug importation, making reference product available to facilitate the development and testing of generic and 505(b)(2) products, and shared and individual REMS. New legislative and regulatory efforts could ultimately have an adverse impact on our business and results of operation.
Non-Patent Exclusivity
The holder of an NDA may be entitled to a period of non-patent exclusivity, during which the FDA cannot make the approval of an ANDA or 505(b)(2) application that relies on the listed drug effective. For example, a pharmaceutical manufacturer may obtain five years of non-patent exclusivity upon NDA approval of a new chemical entity (“NCE”), during which the FDA cannot accept any ANDA or 505(b)(2) application for filing for the same active moiety except that the FDA may accept an application for filing after four years if the follow-on applicant makes a paragraph IV certification.
The holder of an NDA, including one approved under Section 505(b)(2), may obtain a three-year period of exclusivity for a particular condition of approval, or change to a marketed product, such as a new formulation for a previously approved product or a new dosage form or route of administration, if one or more new clinical studies (other than bioavailability or bioequivalence studies) was essential to the approval of the application and was conducted or sponsored by the applicant. Should this occur, the FDA would be precluded from making the approval of any ANDA or 505(b)(2) application effective for the protected modification until after that three-year exclusivity period has ended. However, unlike NCE exclusivity, the FDA can accept an application and begin the review process during the exclusivity period. Five-year and three-year exclusivity will also not delay the submission or approval of a full NDA.
In addition, an applicant submitting an ANDA to the FDA may be entitled to a 180-day market exclusivity period with respect to subsequently filed generic applications if such applicant is the first to submit a substantially complete application to the FDA and whose filing includes a paragraph IV certification that the applicable patent(s) are invalid, unenforceable and/or not infringed, obtains approval, and launches the product in the marketplace without triggering any statutory forfeiture provisions. An ANDA for a product designated as competitive generic therapy that does not otherwise have patent or exclusivity protections listed in the Orange Book and that is the first approved applicant, is also eligible for a period of 180 days of regulatory exclusivity with respect to other ANDAs. These ANDA exclusivity periods, however, can be lost under certain circumstances. Competitive generic therapies are products for which there is not more than one approved drug included in the Orange Book.
Pediatric exclusivity is another type of non‑patent marketing exclusivity in the U.S. and, if granted, provides for the attachment of an additional six months of marketing protection to the term of any existing regulatory and statutory exclusivity for NDA products, including the non‑patent exclusivity period described above and patent protections. This six‑month exclusivity may be granted if an NDA sponsor submits pediatric data that fairly respond to a written request from the FDA for such data. The data does not need to show the product to be effective in the pediatric population studied; rather, if the clinical trial is deemed to fairly respond to the FDA’s request, the additional protection is granted. If reports of requested pediatric studies are submitted to and accepted by the FDA within the required time frames, whatever statutory or regulatory periods of exclusivity or Orange Book listed patent protection cover the drug are extended by six months. This is not a patent term extension, but it effectively extends the regulatory period during which the FDA cannot approve an ANDA or 505(b)(2) application owing to regulatory exclusivity or listed patents. Moreover, pediatric exclusivity attaches to all formulations, dosage forms, and indications for products with existing marketing exclusivity or patent life that contain the same active moiety as that which was studied.
If approved, drug products may also be eligible for periods of U.S. patent term restoration if the approval is the first permitted commercial marketing for the product. If granted, patent term restoration extends the patent life of a single unexpired patent, that has not previously been extended, for a maximum of five years. The total patent life of the product with the extension also cannot exceed fourteen years from the product’s approval date. Subject to the prior limitations, the period of the extension is calculated by adding half of the time from the effective date of an IND to the initial submission of a marketing application, and all of the time between the submission of the marketing application and its approval. This period may be reduced by any time that the applicant did not act with due diligence. Whether any of our product candidates will be eligible for patent term restoration is currently unknown. Later, the applicable regulatory authorities may determine that we are not eligible for such restoration periods.
Depending on the drug product, other periods of regulatory exclusivity, such as orphan drug product exclusivity, may also block subsequent applicants.
Orphan Drug Designation
Some jurisdictions, including the U.S., may designate drugs for relatively small patient populations as orphan drugs. Pursuant to the Orphan Drug Act, the FDA grants orphan drug designation to drugs intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the U.S. or affects more than 200,000 in the U.S. and for which there is no reasonable expectation that the cost of developing and making the product available in the U.S. will be recovered from U.S. sales. Additionally, sponsors must present a plausible hypothesis for clinical superiority to obtain orphan designation if there is a product already approved by the FDA that is intended for the same indication and that is considered by the FDA to be the same as the already approved product. This hypothesis must be demonstrated to obtain orphan exclusivity. Orphan drug designation provides certain benefits, such as the opportunity for grants, tax credits, application user fee waivers, and exemption from program user fees under certain circumstances. The tax advantages, however, were limited in the 2017 Tax Cuts and Jobs Act. If approved for the orphan designation, orphan designated drugs may receive seven years of exclusivity, which, subject to certain exceptions, protects the drug from FDA approval of another drug with the same principal molecular features for the same orphan indication. The FDA may, however, approve a product with the same principal molecular features for the same orphan indication during this time period if such product is able to demonstrate clinical superiority. The FDA may further approve a product with the same principal molecular features for a different indication, or a different product for the same indication during the orphan exclusivity period. Orphan exclusivity can also be lost under certain circumstances, such as the inability of the application holder to ensure sufficient quantities of the product. Orphan drugs are also exempt from the above discussed PREA requirements. Orphan drugs are not, however, exempt from pediatric testing for molecularly targeted oncology drugs.
Combination Drug/Device Regulation
Our products and our products marketed by our partners, as well as our products being developed by our partners are most often categorized as “drug-device combination products” because they contain both a drug and a device to administer the drug. To date, our and our partners’ combination products have been regulated as drug, and are therefore subject to the NDA, ANDA, sNDA, sANDA and 505(b)(2) drug approval process and regulations. Combination drug/device products raise unique scientific, technical and regulatory issues. The FDA has established an Office of Combination Products (“OCP”) to address the challenges associated with the review and regulation of combination products. The OCP assists in determining strategies for the approval of drug/delivery device combinations and assuring agreement within the FDA on review responsibilities. The device specific information is filed with the FDA as part of the drug approval submission or it may be filed separately in the form of a device master file, also known as the master access file (“MAF”). A MAF is not an FDA approval submission but is a filing that can be used to provide supporting data for our partners’ drug approval submissions. A MAF will be reviewed by the FDA only when referenced in an approval submission. By filing a MAF, we are able to provide information directly to the FDA, which can then be referenced by our partners in their drug approval submissions without having to share our proprietary information directly with our partners.
Where common data elements may be part of several submissions for regulatory approval, as in the case of information supporting an injection system, a MAF filing with the FDA may be the preferred route. A delivery device that is applicable to a variety of drug/device combination products, represents another opportunity for such a filing. Another option would be to obtain a 510(k) premarket clearance, de novo authorization, or premarket approval (“PMA”) from the FDA for our delivery device as a stand-alone product. The type of premarket submission required is based on the FDA device classification for the delivery device (Class I, II, III, or not yet classified). We intend to pursue such strategies as permitted by the law and as directed by the FDA either through guidance documents or discussions.
Development of a device with a specific drug will likely be handled as part of the marketing application for the drug product, which may be an NDA, ANDA, or supplemental application. Under these circumstances, the device component is only approved if the drug component is approved.
To the extent that our injectors are packaged with the drug as part of a drug delivery system, the entire package will be subject to the requirements for drug/device combination products. These include drug manufacturing requirements, drug adverse reaction and other reporting requirements, and all of the restrictions that apply to drug labeling and advertising. Additionally, such products will also be subject to certain device requirements, including QSRs and certain reporting requirements, such as medical device reports. Sponsors of clinical studies using investigational devices are also required to comply with the FDA’s investigational device exemption regulations. These requirements necessitate additional expenditures of time and resources, which could have a substantial adverse impact on our ability to commercialize our products and our operations.
Other Post-Approval Requirements and Promotional Activities
Any product manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements related to manufacturing, recordkeeping, reporting, including adverse experience reporting, drug shortage reporting, periodic reporting, product sampling and distribution, advertising, marketing, promotion, and post‑approval obligations imposed as a condition of approval, such as Phase IV clinical trials, REMS and surveillance to assess safety and effectiveness after commercialization.
There also are continuing annual user fee requirements. In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register and, in the case of generic drug products, self-identify their establishments with the FDA and certain state agencies and list their drug products. Device manufacturers must also register their facilities and list the devices that they design, develop, manufacture or import, except those subject to a drug approval. These facilities must also pay annual registration fees. The distribution of prescription pharmaceutical product samples is also subject to the Prescription Drug Marketing Act (“PDMA”).
FDA post-approval requirements are continually evolving. For example, in March 2020, the U.S. Congress passed the Coronavirus Aid, Relief, and Economic Security Act (the “CARES Act”) which includes various provisions regarding FDA drug shortage and manufacturing volume reporting requirements, as well as provisions regarding supply chain security, such as risk management plan requirements, and the promotion of supply chain redundancy and domestic manufacturing. As part of the CARES Act implementation, the FDA recently issued guidance on the reporting of the volume of drugs produced, which reporting will require additional administrative efforts by drug manufacturers.
The FDA closely regulates the post-approval marketing and promotion of drugs and devices, including standards and regulations for direct-to-consumer advertising, dissemination of off-label information, industry-sponsored scientific and educational activities and promotional activities involving the Internet. Drugs may be marketed only for the approved indications and in accordance with the provisions of the approved label. Further, if there are any modifications to the drug, including changes in indications, labeling or manufacturing processes or facilities, we may be required to submit and obtain FDA approval of a new or supplemental NDA, which may require us to develop additional data or conduct additional preclinical studies and clinical trials. Failure to comply with these requirements can result in adverse publicity, Warning Letters, Untitled Letters, corrective advertising and potential civil and criminal penalties, as well as liability under the civil False Claims Act, exclusion from participation in federal healthcare programs, mandatory compliance programs under corporate integrity agreements, debarment, and refusal of government contracts among other consequences.
Physicians may prescribe legally available products for uses that are not described in the product’s labeling and that differ from those tested by us and approved by the FDA. Physicians may believe that such off-label uses are the best treatment for many patients in varied circumstances. The FDA does not regulate the behavior of physicians in their choice of treatments. The FDA does, however, impose stringent restrictions on manufacturers’ communications regarding off-label use. Specifically, manufacturers and product sponsors may not promote a product for off-label uses and must also comply with the FDA’s other promotional requirements.
Manufacturing and Quality Regulations
The FDA established regulations to require that the methods used in, and the facilities and controls used for, the manufacture, processing, packing and holding of drugs and medical devices conform to cGMPs and QSRs. The cGMP regulations the FDA enforces are comprehensive, cover all aspects of manufacturing operations and require the conduct of investigations and FDA reporting under certain circumstances. The cGMP regulations for devices, called the Quality System Regulation, are also comprehensive and cover all aspects of device design, quality and manufacturing, including, for example, pre-production design requirements and validation; production and process controls; complaint handling and investigations; corrective and preventative actions; and distribution, installation and servicing. Compliance with the regulations requires a continuous commitment of time, money and effort in all operational areas. The FDA conducts periodic inspections of drug and device facilities to assess the cGMP/QSR status of marketed products. Before approval of an application, the FDA will typically also conduct facility inspections to ensure cGMP/QSR compliance.
Controlled Substances Regulation
Certain of our drug products are considered “controlled substances” as defined in the Controlled Substances Act (“CSA”) and implementing regulations, which, depending on the controlled substance schedule, establish certain registration, security, monitoring, reporting, storage, distribution, importation, inventory, quota, record keeping, prescribing, dispensing, and other requirements administered by the Drug Enforcement Agency (“DEA”). The DEA regulates controlled substances as Schedule I, II, III, IV or V substances, with Schedule I and II substances considered to present the highest risk of substance abuse and Schedule V substances the lowest risk. These requirements are directly applicable to us and also applicable to our contract manufacturers and to distributors, prescribers and dispensers of our products.
The DEA regulates the handling of controlled substances through a closed chain of distribution. This control extends to the equipment and raw materials used in their manufacture and packaging, in order to prevent loss and diversion into illicit channels of commerce. Annual registration is required for any facility that manufactures, distributes, imports or exports any controlled substance. The registration is specific to the particular location, activity and controlled substance schedule. The DEA typically inspects a facility to review its security measures prior to issuing a registration and on a periodic basis. Certain reports must also be made for controlled substances, such as reports for thefts or significant losses of any controlled substance and reports of suspicious orders. Failure to maintain compliance with applicable requirements, particularly as manifested in loss or diversion, can result in administrative, civil or criminal enforcement action. Individual states may also regulate controlled substances.
Foreign Approval Process
In addition to regulations in the U.S., we (and, where appropriate, our partners marketing medicinal products incorporating our devices) are subject to various foreign regulations governing clinical trials, manufacturing, and the commercial sales and distribution of our medicinal products. We and/or our partners must obtain approval of a medicinal product by the comparable regulatory authorities of foreign countries before we can commence clinical trials or marketing of the product in those countries. The requirements governing the conduct of clinical trials, manufacturing, product licensing, pricing and reimbursement and the regulatory approval process all vary greatly from country to country. Additionally, the time it takes to complete the approval process in foreign countries may be longer or shorter than that required for FDA approval. Foreign regulatory approvals of our products are necessary whether or not we obtain FDA approval for such products. Finally, before a new drug may be exported from the U.S., it must either be approved for marketing in the U.S. or meet the requirements of exportation of an unapproved drug under Section 802 of the Export Reform and Enhancement Act or comply with FDA regulations pertaining to INDs.
In the EU, marketing authorizations for medicinal products can be obtained through several different procedures, principally the centralized procedure, the decentralized procedure and the mutual recognition procedure. The centralized procedure allows a company to submit a single application to the European Medicines Agency (“EMA”), which may provide a positive opinion regarding the application to the effect that it meets certain safety, quality and efficacy requirements. A centralized marketing authorization will be granted based on a positive opinion of the EMA as approved by the European Commission. It is valid in all EU member states and three of the four European Free Trade Association countries (Iceland, Liechtenstein and Norway). The centralized procedure is mandatory for certain medicinal products, including orphan medicinal products and biologic products, and optional for certain other high technology products. The decentralized procedure allows companies to file identical applications for authorization to several EU member states simultaneously for medicinal products that have not yet been authorized in any EU member state. The competent authority of one EU member state, selected by the applicant (the Reference Member State), assesses the application for marketing authorization. The competent authorities of the other EU member states for which marketing authorizations are sought (concerned member states) are subsequently required to grant marketing authorization for their territories on the basis of this assessment except where grounds of potential serious risk to public health require an authorization to be refused. This procedure allows companies that have a medicinal product already authorized in one EU member state to apply for this authorization to be recognized by the competent authorities in other EU member states. The mutual recognition procedure applies in the case where a marketing authorization for the same medicinal product has already been granted by an EU/European Economic Area (“EEA”) member state, whereas the decentralized procedure is applicable if no marketing authorization exists. Since January 1, 2021, the United Kingdom has separate approval processes to the EU as a consequence of Brexit.
In so far as our products may be placed on the market as medical devices outside of the U.S. (as opposed to a delivery system of a medicinal product), we are also subject to foreign legal and regulatory requirements. Legal restrictions on the sale of imported medical devices and products vary from country to country. The time required to obtain approval by a foreign country may be longer or shorter than that required for FDA approval, and the requirements may differ. We primarily rely upon the companies’ marketing our injectors in foreign countries to obtain the necessary regulatory approvals for sales of our products in those countries.
Our Minneapolis Quality Management System has ISO 13485: 2003 certification, the medical device industry standard for our quality systems. This certification shows that our device development and manufacturing comply with standards for quality assurance, design capability and manufacturing process control. Such certification, along with compliance with the EU Medical Devices Regulation 2017/745 (which replaced the European Medical Device Directive 93/42/EC on May 27, 2020), enables us to affix the CE Mark (a certification indicating that a product has met EU consumer safety, health or environmental requirements) to current products and supply the device with a Declaration of Conformity. Regular surveillance audits by our notified body, British Standards Institute, are required to demonstrate continued compliance.
Other Healthcare Laws and Compliance Requirements
In the U.S., the research, manufacturing, distribution, marketing, sale and promotion of drug products and medical devices are subject to numerous regulations by various federal, state and local authorities.
We are subject to various U.S. federal and state laws restricting certain marketing practices in the pharmaceutical industry, including anti-kickback laws and false claims laws. The federal healthcare program Anti-Kickback Statute is a criminal statute that prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce, reward or in return for purchasing, leasing, ordering or arranging for or recommending the purchase, lease or order of any healthcare item or service reimbursable under Medicare, Medicaid or other federally financed healthcare programs, except for activities protected by narrowly-drawn statutory and regulatory safe harbors. HHS recently promulgated a regulation that is effective in two phases. First, the regulation excludes from the definition of “remuneration” limited categories of (a) PBM rebates or other reductions in price to a plan sponsor under Medicare Part D or a Medicaid Managed Care Organization plan reflected in point-of sale reductions in price and (b) PBM service fees. Second, effective January 1, 2023, the regulation expressly provides that rebates to plan sponsors under Medicare Part D either directly to the plan sponsor under Medicare Part D, or indirectly through a pharmacy benefit manager will not be protected under the anti-kickback discount safe harbor. Liability under the federal Anti-Kickback Statute may be established without a person or entity having actual knowledge of the statute or specific intent to violate it, and a violation of the Anti-Kickback Statute may be grounds for a government or whistleblower claim under the federal False Claims Act. Violations of the federal Anti-Kickback Statute may be punished by civil and criminal fines, imprisonment, and/or exclusion from participation in federal healthcare programs. Separately, the Beneficiary Inducement Civil Monetary Penalties Law imposes similar restrictions on interactions between the biopharmaceutical industry and federal healthcare program beneficiaries.
The federal civil False Claims Act prohibits, among other things, any person from knowingly presenting or causing to be presented a false or fraudulent claim for payment of federal funds; knowingly making or causing to be made, a false statement to get a false claim paid; knowingly making, using or causing to be made or used, a false record or statement to get a false or fraudulent claim paid or approved by the government; conspiring to defraud the government by getting a false or fraudulent claim paid or approved by the government; or knowingly making, using or causing to be made or used a false record or statement to avoid, decrease or conceal an obligation to pay money to the federal government. Claims may be pursued by whistleblowers through qui tam actions, even if the government declines to intervene. Intent to deceive is not necessary to establish civil liability, which may be predicated on reckless disregard for or deliberate ignorance of the truth. The civil False Claims Act authorizes imposition of treble damages and a civil penalty for each false claim, such as an invoice, submitted for payment and may result in significant financial penalties and damages. The criminal federal False Claims Act imposes criminal fines or imprisonment against individuals or entities who make or present a claim to the government knowing such claim to be false fictitious or fraudulent. Conviction or civil judgment for violation of the False Claims Act can also result in debarment from government procurement programs and exclusion from participation in federal healthcare programs.
The Civil Monetary Penalties Statute further imposes penalties against any person or entity who, among other things, is determined to have presented or caused to be presented a claim to a federal healthcare program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent. Federal consumer protection and unfair competition laws also broadly regulate marketplace activities and activities that potentially harm consumers.
The federal Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), also created federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payer (e.g., public or private), knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters. The ACA amended the intent requirement of certain of these criminal statutes so that a person or entity no longer needs to have actual knowledge of the statute, or the specific intent to violate it.
Various federal and state healthcare programs obligate us to report drug pricing information that is used as the basis for their reimbursement rates for pharmacies and other healthcare providers including under the Medicaid and Medicare programs, prices charged certain federal agencies and non-federal purchasers, and required manufacturer rebates on prescriptions paid by Medicaid and other plans. Payment for a manufacturer’s drugs by these programs is conditioned on submission of this pricing information. States, such as California, have also enacted transparency laws that require manufacturers to report price increases and related information. Some government healthcare programs impose penalties if drug price increases exceed specified percentages or inflation rates, and these penalties can result in mandatory penny prices for certain federal and 340B program customers or rebates equal to 100% of average price. Failure to comply with the rules for calculating and submitting pricing information or otherwise overcharging the government or its beneficiaries may result in criminal, civil, or administrative sanctions or enforcement actions, including False Claims Act liability.
In addition, the Physician Payment Sunshine Act provisions of the Healthcare Reform Act require extensive tracking of certain payments and other transfers of value to physicians (as defined under the Social Security Act), certain other types of healthcare professionals (physician assistants, nurse practitioners, clinical nurse specialists, certified registered nurse anesthetists and certified nurse midwives licensed in the U.S.), and U.S. teaching hospitals, as well as ownership and investment interest held by physicians and their immediate family members. Payments made to principal investigators and research institutions at teaching hospitals for clinical trials are also included within this law for reporting purposes. These payments and other transfers of value are required to be reported to the Centers for Medicare and Medicaid Services (“CMS”), which publishes the data publicly on the CMS Open Payments website. Government agencies and private entities may inquire about our marketing practices, and government entities may pursue enforcement activities based on the disclosures in those public reports. Similar state laws also impose reporting requirements for various types of payments and other transfers of value to healthcare providers and organizations, including marketing and promotional expenses, and impose various types of gift bans and other compliance requirements on manufacturers. Failure to comply with required reporting requirements, gift bans and other compliance requirements under these laws could subject manufacturers and others to substantial civil money penalties.
The majority of states also have statutes or regulations similar to the federal consumer protection and unfair competition laws, Anti-Kickback Statute and civil False Claims Act, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply to reimbursement for healthcare items and services regardless of the payer type, including private payers. A number of states now have implemented transparency laws requiring manufacturers to report pricing information and require pharmaceutical companies to report expenses relating to the marketing and promotion of pharmaceutical products and to report gifts and payments to individual physicians in the states. Other states restrict when and where pharmaceutical companies may provide meals to prescribers or engage in other marketing related activities. In addition, some states require pharmaceutical companies to abide by the pharmaceutical industry’s voluntary compliance guidelines and implement compliance programs or marketing codes of conduct. Failure to comply with state laws could result in regulatory enforcement actions, including the assessment of significant administrative penalties.
Although we may not provide financial assistance to Medicare patients taking drugs sold by us, the Office of Inspector General (“OIG”) has established guidelines that permit pharmaceutical manufacturers to make donations to charitable organizations that provide co-pay assistance to Medicare patients, provided that such organizations, among other things, are bona fide charities, are entirely independent of and not controlled by the manufacturer, provide aid to applicants on a first-come basis according to consistent objective financial criteria, and do not link aid to use of a donor’s product. If we, our vendors or donation recipients are deemed to fail to comply with relevant laws, regulations or evolving government guidance in the operation of these programs, we could be subject to damages, fines, penalties or other criminal, civil or administrative sanctions or enforcement actions.
The Veterans Health Care Act of 1992 requires, as a condition of payment by certain federal agencies and the Medicaid program, that manufacturers of “covered drugs” enter into a Master Agreement, Pharmaceutical Pricing Agreement, and Federal Supply Schedule (“FSS”) contract with the Department of Veterans Affairs through which their covered drugs must be offered for sale at a mandatory ceiling price to certain federal agencies, including the VA and Department of Defense. FSS contracts require compliance with applicable federal procurement laws and regulations, including disclosure of commercial prices during contract negotiations and maintenance of price relationships during the term of the contract, and subject manufacturers to contractual remedies as well as administrative, civil and criminal sanctions. The Veterans Health Care Act also requires manufacturers to enter into pricing agreements with the Department of Health and Human Services to charge no more than a different ceiling price (derived from the Medicaid rebate percentage) to covered entities participating in the 340B drug discount program. Failure to provide the mandatory discount may subject the manufacturer to specific civil monetary penalties, including when subsequent ceiling price recalculations due to pricing data submitted to CMS or new drug price estimations result in a covered entity having paid more that the revised ceiling price and the manufacturer has failed or refused to refund or credit a covered entity. Termination of either of these agreements under the Veterans Health Care Act also jeopardizes payment by Medicaid for the manufacturer’s drugs.
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010, which we refer to together as the Healthcare Reform Act, expanded healthcare coverage within the U.S., primarily through establishment of state insurance exchanges and expansion of the Medicaid program. This law substantially changed the way healthcare is financed by both governmental and private insurers and significantly impacts the pharmaceutical industry. Changes that may affect our business include those governing enrollment in federal healthcare programs, reimbursement changes, payment of an annual fee by manufacturers of branded drugs and biological products based on their share of the federal market, benefits for patients within a coverage gap in the Medicare Part D prescription drug program (commonly known as the “donut hole”), rules regarding prescription drug benefits under the health insurance exchanges, changes to the Medicaid Drug Rebate program, expansion of the Public Health Service’s 340B drug pricing discount program, and fraud and abuse enforcement. These changes impact existing government healthcare programs and are resulting in the development of new programs, including Medicare payment for performance initiatives and improvements to the physician quality reporting system and feedback program. The Affordable Care Act has since been amended to repeal the individual health insurance mandate, change price reporting rules for authorized generics, and increase manufacturers’ share of Medicare Part D prescription costs in the donut hole, and other provisions of the law may be repealed and replaced by Congress, which may greatly affect these government and third-party programs and their effect on our business.
In addition, we may be subject to, or our marketing activities may be limited by, data privacy and security regulation by both the federal government and the states in which we conduct our business. One such statute is the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”) and its implementing regulations, as amended by the Health Information Technology for Economic and Clinical Health Act (“HITECH”). HIPAA established uniform federal standards for “covered entities,” which are certain healthcare providers, health plans and healthcare clearinghouses, as well as their business associates, governing the conduct of specified electronic healthcare transactions, protecting the security and privacy of protected health information, and mandating security breach notification standards. In addition, other federal and state privacy laws, such as the California Consumer Privacy Act, may govern the privacy and security of personal information in certain circumstances, many of which differ from each other in significant ways, thus complicating compliance efforts.
The Foreign Corrupt Practices Act (“FCPA”) further prohibits any U.S. individual or business from paying, offering, or authorizing payment or offering of anything of value, directly or indirectly, to any foreign official, political party or candidate for the purpose of influencing any act or decision of the foreign entity in order to assist the individual or business in obtaining or retaining business. The FCPA also obligates companies whose securities are listed in the U.S. to comply with accounting provisions requiring the company to maintain books and records that accurately and fairly reflect all transactions of the corporation, including international subsidiaries, and to devise and maintain an adequate system of internal accounting controls for international operations. Activities that violate the FCPA, even if they occur wholly outside the U.S., can result in criminal and civil fines, imprisonment, disgorgement, oversight, and debarment from government contracts.
If our operations are found to be in violation of any of the healthcare regulatory laws described above or any other laws that apply to us, we may be subject to penalties, including criminal and significant civil monetary penalties, damages, fines, imprisonment, disgorgement, contractual damages, reputational harm, exclusion from participation in government healthcare programs, debarment prohibiting participation in government procurement and non-procurement covered transactions, injunctions, recall or seizure of products, total or partial suspension of production, denial or withdrawal of pre-marketing product approvals, private qui tam actions brought by individual whistleblowers in the name of the government or refusal to allow us to enter into supply contracts, including government contracts and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations. To the extent that any of our products are sold in a foreign country, we may be subject to similar foreign laws and regulations. In particular, in the EU the data privacy regime (the General Data Protection Regulation which came into effect on May 25, 2018) is regarded as stricter than the U.S. data protection laws. EU laws restrict the export of personal data outside the EU, for instance to the U.S., unless certain safeguards are in place. EU laws and industry codes also restrict certain marketing practices, including inappropriate inducements.
Third-Party Payer Coverage and Reimbursement
The commercial success of the approved products in our portfolio depends, in part, upon the availability of coverage and adequate reimbursement from third-party payers at the federal, state and private levels. Patients who are prescribed treatments for their conditions and providers performing the prescribed services generally rely on third-party payers to reimburse all or part of the associated healthcare costs. Sales of our product portfolio will therefore depend substantially, both domestically and abroad, on the extent to which the costs of our product portfolio will be paid by health maintenance, managed care, pharmacy benefit and/or similar healthcare management organizations, or are reimbursed by government health administration authorities, such as Medicare and Medicaid, private healthcare coverage insurers and other third-party payers. The market for our product portfolio will depend significantly on access to third-party payers’ formularies or lists of treatments for which third-party payers provide coverage and reimbursement.
Also, third-party payers are developing increasingly sophisticated methods of controlling healthcare costs. For example, for high-cost specialty drugs, third party payers have begun demanding value-based pricing in which price is linked to performance metrics. Recent state enactments establish significant negative incentives requiring negotiation of supplemental rebates, and a recent CMS regulation, implemented a most-favored nations pricing model seeking to lower prices under Medicare Part B by tying the costs of certain medicines to cheaper prices in other developed countries. This regulation was rescinded by a final rule published December 29, 2021; however, legislative and executive branch proposals also seek to establish this type of pricing for certain federal healthcare programs. Further, coverage and reimbursement for therapeutic products can differ significantly from payer to payer. As a result, the coverage determination process will require us to provide scientific and clinical support for the use of our products to each payer separately, with no assurance that adequate coverage and reimbursement will be obtained. The cost of pharmaceuticals and medical devices continues to generate substantial governmental and third-party payer scrutiny. We expect that the pharmaceutical industry will experience continued pricing pressures due to the trend toward managed healthcare, the increasing influence of managed care organizations and additional legislative and administrative proposals. Our results of operations and business could be adversely affected by current and future third-party payer policies as well as healthcare legislative and administrative reforms.
Some third-party payers also require pre-approval of coverage for new or innovative devices or drug therapies before they will reimburse healthcare providers who use such therapies. While we cannot predict whether any proposed cost-containment measures will be adopted or otherwise implemented in the future, these requirements or any announcement or adoption of such proposals could have a material adverse effect on our ability to obtain adequate prices for our product portfolio and to operate profitably.
In international markets, there are health technology assessment regimes with price ceilings and supply and demand side restraints on specific products and therapies and profit controls in most EU member states as well as the United Kingdom. There can be no assurance that our products will be considered medically reasonable and necessary for a specific indication, that our products will be considered cost-effective by third-party payers, that an adequate level of reimbursement will be available or that the third-party payers’ reimbursement policies will not adversely affect our ability to sell our products profitably.
Healthcare Reform
In the U.S. and foreign jurisdictions, the legislative landscape continues to evolve. There have been a number of legislative and regulatory changes to the healthcare system that will likely affect our future operations. In particular, there have been and continue to be a number of initiatives at the U.S. federal and state levels that seek to reduce healthcare costs, improve access, and improve quality. The Affordable Care Act (“ACA”), passed in 2010, provided more Americans with healthcare coverage while attempting to curb the growth in healthcare spending in the U.S. The legislation included reforms to patient rights and protections, rules for insurance companies, taxes, tax breaks, funding and spending, and amended other laws including the Food, Drug and Cosmetics Act. Since enactment of the ACA, some of its provisions have been repealed or amended, and other provisions may be repealed and replaced by Congress. Some of the main provisions of the ACA that affected the pharmaceutical and biotechnology industry include, among others, the following:
•an annual, nondeductible fee on any entity that manufactures or imports certain branded prescription drugs and biologic agents, apportioned among these entities according to their market share in certain government healthcare programs;
•an increase in the statutory minimum rebates a manufacturer must pay under the Medicaid Drug Rebate Program to 23.1% and 13% of the average manufacturer price for most branded and generic drugs, respectively, and inclusion of Medicaid managed care plan utilization in manufacturers’ rebate obligations;
•new methodologies by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated;
•a new Medicare Part D coverage gap discount program;
•expansion of eligibility criteria for Medicaid programs thereby potentially increasing manufacturers’ Medicaid rebate liability;
•expansion of the entities eligible for discounts under the Public Health Service pharmaceutical pricing program;
•expansion of healthcare fraud and abuse laws, including the federal civil False Claims Act and the federal Anti-Kickback Statute, new government investigative powers, and enhanced penalties for noncompliance.
The Drug Supply Chain Security Act imposes on manufacturers of certain pharmaceutical products obligations related to product tracking and tracing, among others. Among the requirements of this legislation, manufacturers are required to provide certain information regarding the drug product to individuals and entities to which product ownership is transferred, label the drug product with a product identifier, and keep certain records regarding the drug product. The transfer of information to subsequent product owners by manufacturers is required to be done electronically. Manufacturers are also required to verify that purchasers of the manufacturers’ products are appropriately licensed. Further, manufacturers have drug product investigation, quarantine, disposition, and FDA and trading partner notification responsibilities related to counterfeit, diverted, stolen, and intentionally adulterated products that would result in serious adverse health consequences or death to humans, as well as products that are the subject of fraudulent transactions or which are otherwise unfit for distribution such that they would be reasonably likely to result in serious health consequences or death. Similar requirements are also imposed on other trading partners in the supply chain.
We expect that additional state and federal healthcare reform measures will be adopted in the future. Legislators and regulators at both the federal and state level are increasingly focused on containing the cost of drugs, and there has been increasing legislative and enforcement interest in the U.S. with respect to specialty drug pricing practices. Specifically, there have been recent U.S. congressional inquiries and proposed bills designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient support programs, and reform government program reimbursement methodologies for drugs. For example, California enacted a transparency law requiring manufacturers to report drug price increases and related information, including the reasons for the price increases. Congress also amended the Medicaid statute to alter price reporting of branded products sold as authorized generics, which effectively increases the rebates paid on the brand. Recent executive orders focusing on domestic sourcing also have required government agencies, to the maximum extent practicable, to limit procurement of essential medicines, including epinephrine, to products that are manufactured in the U.S. from U.S. API and other critical inputs. The list of essential medicines is established by the FDA and subject to change. These and any additional healthcare reform and procurement measures could further constrain our business or limit the amounts that federal and state governments will pay for healthcare products and services, which could result in additional pricing pressures.
Other Regulatory Requirements and Considerations
In addition to regulations enforced by the FDA, we must also comply with regulations under the Occupational Safety and Health Act, the Environmental Protection Act, the Toxic Substances Control Act, the Resource Conservation and Recovery Act and other federal, state and local regulations. We are also subject to various laws and regulations regarding laboratory practices, the experimental use of animals, and the use and disposal of hazardous or potentially hazardous substances in connection with our research. In each of these areas, as above, the FDA and other government agencies have broad regulatory and enforcement powers, including, among other things, the ability to levy fines and civil penalties, suspend or delay issuance of approvals, seize or recall products, and withdraw approvals, any one or more of which could have a material adverse effect on us. The effects of potential future changes in regulations or new legislation, if any, as a result of the new administration are also unknown.
Available Information
We file with the U.S. Securities and Exchange Commission (“SEC”) annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy statements and other documents as required by applicable law and regulations. The SEC maintains a website (http://www.sec.gov) that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC. We make available free of charge on or through our website (http://www.antarespharma.com) our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and if applicable, amendments to those reports filed or furnished pursuant to the Exchange Act, as soon as reasonably practicable after electronically filing those documents with or furnishing them to the SEC. The information on our website is not incorporated into and is not a part of this Annual report on Form 10-K.
ITEM 1A. RISK FACTORS
The following “risk factors” contain important information about us and our business and should be read in their entirety. Additional risks and uncertainties not known to us or that we now believe to be not material could also impair our business. If any of the following risks actually occur, our business, results of operations and financial condition could suffer significantly. As a result, the market price of our common stock could decline, and you could lose all of your investment. In this Section, the terms the “Company,” “we”, “our” and “us” refer to Antares Pharma, Inc.
Risks Related to Our Operations
We have significant outstanding indebtedness under a loan and security agreement. If we do not have sufficient cash available to repay the outstanding indebtedness as it becomes due, or if an event of default were to occur that provides Wells Fargo Bank, National Association the right to accelerate the outstanding balance of the loan and to take possession of some or all of our collateral securing the loan, either situation could have a material adverse effect on our business.
On November 1, 2021, we entered into a Credit Agreement (the “Credit Agreement”) with Wells Fargo Bank, National Association, as administrative agent for the lenders, for credit facilities in an aggregate principal amount of up to $40.0 million with a maturity date of November 1, 2024. The Credit Agreement consists of a $20.0 million term loan facility (the “Term Loan Facility”) and a $20.0 million revolving credit facility, $5.0 million of which is available for the issuance of letters of credit and $1.0 million of which is available for swingline loans (the “Revolving Credit Facility”), (collectively the “Credit Facilities”), which are secured by substantially all of our assets. The Term Loan Facility was funded upon execution of the Credit Agreement with the proceeds used to repay our $20.0 million Term Loan with Hercules Capital and to pay fees and expenses incurred in connection with the early repayment. The Revolving Credit Facility remains available for future use and is expected to be used for ongoing working capital requirements and other general corporate purposes as needed. Payments under the Term Loan Facility are due in consecutive quarterly installments on the last business day of each of March, June, September and December, commencing on March 31, 2022. Interest accrues at either the base rate or LIBOR plus the applicable margin, which varies based on our consolidated total leverage ratio and will initially be 1.50% for base rate loans and 2.50% for LIBOR loans. The transaction is expected to provide approximately $1.2 million in annual interest expense savings based on an interest rate of approximately 2.60% (one-month LIBOR rate plus the applicable margin of 2.50%) as of December 31, 2021.
There is no guarantee that healthcare providers and patients will adopt our or our partners’ products or continue to use or prescribe our or our partners’ products, or that we and our partners will be able to receive and maintain adequate payer coverage and reimbursement.
Successful sales of our products depend on the continued prescription by healthcare providers, adoption by patients, and the availability of adequate coverage and reimbursement from third-party payers. There is no guarantee that healthcare and patients’ providers will adopt any newly approved products or continue to prescribe and use products, or that insurers and
governmental healthcare programs, such as Medicare and Medicaid, will provide adequate coverage and reimbursement, or will not disadvantage our products through imposition of prior authorization, step therapy, high co-payments, or similar formulary management techniques. For instance, coverage decisions may depend upon clinical and economic standards that disfavor new drug products when more established or lower cost therapeutic alternatives are already available or subsequently become available. Additionally, certain third-party payers restrict or block access to patients for new products until a clinical review has occurred or clinical evidence is provided to support the benefits for covered patients. Many states also use formularies and preferred drug lists to obtain supplemental Medicaid rebates in excess of those required for Medicaid coverage. The industry competition to be included in such formularies and not disadvantaged often leads to downward pricing pressures on pharmaceutical companies. Any labeled limitations on the use of a product or warnings could discourage adoption of the product by patients, healthcare providers, and insurers.
To ensure sales, manufacturers often must provide multiple discounts on the same drug in the chain of distribution to the healthcare provider and the payer. Further, manufacturers are required to assume responsibility for a percentage of Medicare Part D prescription costs for innovator drugs and biologics while the beneficiary is in the coverage gap. Increasingly, payers are looking for metrics and performance-based pricing to justify increased costs of therapeutic advancements. Even if coverage is obtained, the net realization from price concessions may negatively impact our profitability. Government health programs also impose inflation penalties that may have adverse consequences if we increase prices in the future. Moreover, we and our partners may experience a delay in receiving coverage and reimbursement for any new products or may not receive adequate levels of coverage or reimbursement at all. New competitive products may be approved, and payers may disadvantage our products in favor of the newly approved products and technologies. If the time to obtain coverage is lengthy, if we are unable to obtain or maintain adequate coverage, or if the rebates we negotiate are higher than anticipated, it may negatively impact our revenue from product sales.
Additionally, if healthcare providers and patients do not adopt any new product, or if insurers restrict patient access or disadvantage our or our partners’ products in their formularies or otherwise do not provide adequate coverage and reimbursement, we and our partners may not be able to generate sustainable revenue growth from product sales and royalties which will have a material adverse effect on our business and future product opportunities. We and our partners, accordingly, may need to take steps to assist patients in their ability to afford our products, such as offering bridge programs, free-trials, discounts, rebates and co-pay coupon programs.
New information concerning our or our partners’ products learned through required post-approval studies and product use may also result in changes to our or our partners’ products. Should any of these events occur, they could have a material and adverse effect on our operations and business.
Any post-approval requirements, including Phase IV studies may also require the dedication of substantial time and resources. By example, as a post-marketing requirement for XYOSTED®, we must conduct a pediatric study. The FDA has also asked us to conduct a separate label comprehension study that assesses patients’ understanding of key risk messages in the Medication Guide for XYOSTED® and a study of testosterone replacement therapy in pediatric males ages 14 years and older for conditions associated with a deficiency or absence of endogenous testosterone. The label comprehension study findings may result in revisions to the Medication Guide to optimize patients’ understanding of important risks of XYOSTED® and potentially other label restrictions or changes. The FDA found that our first label comprehension study did not fulfill the post approval requirement and, thus, we are preparing to conduct a new label comprehension study, which must be completed by the middle of 2023 and will require dedication of funds and resources. Additionally, the outcome of any post-approval studies, including the pediatric study, is uncertain and may not result in an expanded label indication or could result in additional labeling requirements or other post-approval restrictions or regulatory actions.
Additionally, use of our or our partners’ products by patients and in Phase IV and post-marketing studies may result in the discovery of new information concerning the products. This may result in regulatory or other actions, including, product liability actions, enforcement actions, distribution and manufacturing restrictions, changes to product labeling and promotional materials, the imposition of post-market requirements, such as REMS or additional Phase IV studies, withdrawal of marketing application approvals, withdrawal of the product from the market, refusal to approve new marketing applications or supplements, product recalls, clinical holds and suspension of clinical studies, safety alerts, dear healthcare provider letters, adverse publicity, and reimbursement and insurance coverage consequences, among others. Should any of these events occur, they could have a material and adverse effect on our operations and business. By way of example, in October 2020 the FDA proposed that Makena® be withdrawn from the market following a review by the FDA’s Bone, Reproductive and Urologic Drugs Advisory Committee. Covis subsequently requested an FDA public hearing, and the FDA has granted the request. The FDA has not yet set a date for the public hearing. While, at this time, Makena® may still be marketed, we do not yet know what the FDA’s ultimate decision will be and whether resources that it dedicated to Makena® will be reduced. The uncertainty with
regard to Makena® has negatively impacted our product revenue and royalties from Covis and may adversely impact the business.
Our employees, independent contractors, consultants, commercial partners, manufacturers, principal investigators, or contract research organizations (“CROs”) may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have a material adverse effect on our business.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees, independent contractors, consultants, commercial partners, manufacturers, investigators or CROs could include intentional, reckless, negligent, or unintentional failures to comply with FDA regulations, comply with applicable fraud and abuse laws, provide accurate information to the FDA, properly calculate pricing information required by federal programs, comply with federal procurement rules or contract terms, report financial information or data accurately or disclose unauthorized activities to us. This misconduct could also involve the improper use or misrepresentation of information obtained during clinical trials, which could result in regulatory sanctions and serious harm to our reputation. It is not always possible to identify and deter this type of misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with such laws or regulations. Moreover, it is possible for a whistleblower to pursue a False Claims Act (“FCA”) case against us based on the actions or inactions of these third parties even if the government considers the claim unmeritorious and declines to intervene, which could require us to incur costs defending against such a claim. Further, due to the risk that a judgment in an FCA case could result in exclusion from federal health programs or debarment from procurement programs, whistleblower cases often result in large settlements. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, financial condition, and results of operations, including the imposition of significant fines or other sanctions.
We rely on third parties to perform many necessary services for our products including services related to the distribution, invoicing, rebates and contract administration, co-pay program administration, sample distribution and administration, storage and transportation of our products.
Depending on the product, we have retained third-party service providers to perform a variety of functions related to the distribution, invoicing, rebates and contract administration, co-pay program administration, sample distribution and administration, storage and transportation of our products, key aspects of which are out of our direct control. We place substantial reliance on these providers as well as other third-party providers that perform services for us, including, depending on the product, entrusting our inventories of products to their care and handling. We also may rely on third parties to administer our drug price reporting and rebate payments and contracting obligations under federal programs. Despite our reliance on third parties, we are responsible for compliance with the applicable legal and program requirements. By example, in certain states, we are required to hold licenses to distribute our products in these states and must comply with the associated state laws. Moreover, if these third-party service providers fail to meet expected deadlines, or otherwise do not carry out their contractual duties to us or encounter physical damage or a natural disaster at their facilities, our ability to deliver products to meet commercial demand would be significantly impaired. In addition, we may use third parties to perform various other services for us relating to regulatory monitoring, including adverse event reporting, safety database management and other product maintenance services. If our employees or any third-party service providers fail to comply with applicable laws and regulations, we and/or they may face regulatory or FCA enforcement actions. Moreover, if the quality or accuracy of the data maintained by these service providers is insufficient, our ability to continue to market our products could be jeopardized or we and/or they could be subject to regulatory sanctions. We do not currently have the internal capacity to perform these important commercial functions, and we may not be able to maintain commercial arrangements for these services on reasonable terms.
We are dependent on numerous third parties in our supply chain for the supply and manufacture of our products and our partners’ products. If we do not develop and maintain relationships with suppliers, manufacturers, assemblers and/or licensees of our and our partners’ drug/device products or product candidates, or if such third parties are unable to supply or manufacture products or assemble and package the final products, we may be unable to successfully manufacture, assemble, package and sell our and our partners’ products, which could have a material adverse effect on our business.
The availability of our products and product candidates depends upon our ability to procure the raw materials, components, packaging materials and finished products that we need from third parties. We have entered into supply agreements with numerous third-party suppliers, many of which are currently our single source for the materials necessary for certain of our products. If any of these or other third parties are unable to supply their respective components for any reason, including due to violations of the FDA’s QSR or cGMP requirements, our or our partners’ ability to manufacture the finished product will be adversely affected and our ability to meet the supply and demand for any sales of such products and the resulting revenue therefrom will be negatively affected. Additionally, as many of our components are manufactured by sole third-party suppliers, in the event of a failure to supply, we may not be able to find alternative third suppliers in a timely or cost-effective manner.
Moreover, any failure to comply with the applicable regulatory requirements could subject us, our suppliers, or our collaborators to regulatory enforcement actions or recalls. In the case of product candidates, our and our partners’ ability to conduct the necessary studies would also be adversely impacted.
We do not currently use our own facilities to manufacture commercial quantities of our or our partners’ drug/device combination products or components. We currently must contract with third parties and/or our partners to produce products, components, and product candidates and to assemble and package finished products and related components according to specifications and that must comply with all applicable manufacturing requirements, including cGMPs for drug products and QSRs for medical devices. The future development and delivery of our and our partners’ products and product candidates depend on the capability, as well as the timely, profitable and competitive performance of these third parties and/or our partners, in addition to their initial and continued FDA approval following regulatory authority facility inspections and compliance. There is also no assurance that such third parties and/or our partners will be willing to manufacture, assemble or sell the drug/device products or components or that they will not encounter manufacturing delays, problems, or difficulties. A limited number of manufacturers exist that can manufacture our and our partners’ products, components, and product candidates.
In addition, contract manufacturers may use their own technology, technology developed by us, technology developed by our partners, or technology acquired or licensed from third parties. When contract manufacturers develop proprietary process technology, our reliance on such contract manufacturers is increased. A technology transfer from the original contract manufacturer may be required. Any such technology transfer may also require the transfer of requisite data for regulatory purposes, including information contained in a proprietary drug or device master file held by a contract manufacturer. We and/or our partners would be dependent on the contract manufacturer for the maintenance and right of reference to the drug or device master file. If the contract manufacturer fails to maintain a drug or device master file or withdraws our or our partners’ right of reference, we and/or our partners may no longer be able to manufacture, develop, market, and sell our or our partners’ products or product candidates.
We rely on multiple commercial supply arrangements with third-party manufacturers. Our third-party manufacturers may fail to pass the audits by our or our partners’ internal quality and regulatory group. Any of these actions could delay or prevent our development of products, delay or prevent the submission of these products for regulatory approval, delay or prevent marketing approval, or result in insufficient product or product candidate quantity to support commercial demand or development. We may also be required to replace manufacturers, which would be time consuming and expensive, and we may not be able to reach favorable agreements with or FDA approval for alternative manufacturers. As a result, our business, financial condition and results of operations could be seriously harmed.
In addition to the above manufacturing capabilities provided by third party manufacturers, on July 1, 2019, we entered into a lease for office, laboratory, manufacturing and warehousing space in Minnetonka, Minnesota. We completed the build-out of the facility and began occupying the space in 2020. The new facility supports our administrative functions, product development and quality operations, and is intended to provide additional manufacturing and warehousing capabilities in the future. If we begin manufacturing or producing commercial products in the future, we will be subject to the same risks as our third-party manufacturers. We may also not be able to begin product manufacturing and production in a timely manner due to a number of different reasons, including, but not limited, an inability to obtain the necessary supplies, labor and expertise. We may also be delayed if we are not able to obtain necessary licensing and regulatory authority inspections in a timely manner, such as due to inspection restrictions during the Pandemic. Any failure to commence operations at our own facilities could have a material impact on our business.
Some of our suppliers may experience disruption to their respective supply chains due to the effects of the COVID-19 pandemic (“Pandemic”), which could delay, prevent or impair our development or commercialization efforts.
We obtain certain critical materials and components used in manufacturing our products from third-party suppliers whose operations may be directly or indirectly affected by the Pandemic. If we are unable to obtain these critical materials and components in sufficient quantities and in a timely manner, our supply of product for commercialization may be disrupted or delayed which could have a material impact on our revenue and the development, testing and clinical study of our products and product candidates might be delayed or infeasible, and regulatory approval or commercialization of our products and product candidates might be delayed, not obtained or hindered, which could significantly harm our business.
See additional risk factors associated with manufacturing in the section “Risks Related to Regulatory Matters.”
We rely on many of our partners to manufacture and supply the drug for their product and to distribute and commercialize their products.
We have entered into license and supply agreements with several different partners. Under many of the arrangements, our partners are responsible for supplying the drug product and we are responsible for manufacturing the auto injector and for final assembly and packaging of the final product. Our partners are responsible for the distribution and commercialization of the products in the U.S.
There is no guarantee that our arrangements with any of our partners will be successful. Our partners typically control the manufacture and supply of the drug for their auto-injector product. If, at any time, our partner is unable to source their drug or ceases to manufacture and supply us with their drug or fails to produce sufficient supplies of the drug, we may be unable to produce a finished product or sell our auto injectors designed for their product. In addition, if any of our partners are not able to produce sufficient supplies of the drug in accordance with cGMPs, we also will be unable to produce a finished product and we and/or our partner may be subject to regulatory enforcement action. We also rely on our partners to commercialize and distribute their product within the U.S. and if they are unsuccessful in commercializing the product, the resulting revenue may be lower than expected. In many instances, our partners hold the marketing authorization for their products and should our partner fail to comply with the applicable regulatory requirements, our partner or we may be subject to regulatory enforcement action. There may be instances in which we hold the marketing authorization of the product, and therefore, we may face greater risk of regulatory enforcement should any of our partners fail to comply with the applicable regulatory requirements. Any enforcement action could impact the ability to produce, market, commercialize, sell, and distribute the finished drug product, and in turn, our revenue. Additionally, our partners’ control the distribution and commercialization strategies for their products and we may disagree with their decision or business strategies. Such decisions may impact the success of their product commercialization, and we may receive less revenue than desired or expected. Also, as our partners are subject to the same product development and commercialization risks as us, any adverse impact on our partners could have an adverse impact on us.
We may incur significant liability if it is determined that we are promoting or have in the past promoted the “off-label” use of drugs or medical devices, or otherwise promoted or marketed approved products in a manner inconsistent with the FDA’s requirements.
In the U.S. and certain other jurisdictions, companies may not promote drugs or medical devices for “off-label” uses, that is, uses that are not described in the product’s labeling and that differ from those that were approved or cleared by the FDA or other foreign regulatory agencies. Under what is known in the C’s as the “practice of medicine,” physicians and other healthcare practitioners may prescribe drug products and use medical devices for off-label or unapproved uses, and such uses are common across some medical specialties. Although the FDA does not regulate a physician’s choice of medications, treatments or product uses, the Federal Food, Drug and Cosmetic Act and FDA regulations significantly restrict permissible communications on the subject of off-label uses of drug products and medical devices by pharmaceutical and medical device companies. As the sponsors of FDA approved products, we and our partners will not only be responsible for the actions of the companies but also can be held liable for the actions of employees and contractors, requiring that all employees and contractors engaging in regulated functions, such as product promotion, be adequately trained and monitored, which requires time and monetary expenditures.
If the FDA determines that a company has improperly promoted a product “off label” or otherwise not in accordance with the agency’s promotional requirements, the FDA may issue a warning letter or seek other enforcement action to limit or restrict certain promotional activities or materials or seek to have product withdrawn from the market or seize product, among other enforcement requirements. In addition, a company that is found to have improperly promoted off-label uses may be subject to significant liability, including civil fines, criminal fines and penalties, civil damages and exclusion from federal funded healthcare programs such as Medicare and Medicaid and/or government contracting, consent decrees and corporate integrity agreements, as well as potential liability under the federal FCA and applicable state false claims acts. Conduct giving rise to such liability could also form the basis for private civil litigation by third-party payers or other persons allegedly harmed by such conduct.
Moreover, in addition to the regulatory restrictions on off-label promotion, there are other FDA restrictions on and requirements concerning product promotion and advertising, such as requirements that such communications be truthful and non-misleading and adequately supported. The FDA also has requirements concerning the distribution of drug samples. The FDA and other authorities may take the position that we are not in compliance with promotional, advertising, and marketing requirements, and, if such non-compliance is proven, we may be subject to significant liability, including but not limited to administrative, civil and criminal penalties and fines, in addition to regulatory enforcement actions.
We currently depend on a limited number of customers for the majority of our revenue, and the loss of any one of these customers could substantially reduce our revenue and impact our liquidity.
For the year ended December 31, 2021, we derived approximately 42% of our revenue from Teva. In addition, we derived a significant portion of our product sales revenue from wholesale distributors including McKesson, AmerisourceBergen and Cardinal Health, which each accounted for approximately 13%, 12% and 11% of total revenues in the year ended December 31, 2021, respectively. The loss of any of these significant customers or partners or reduction in our business activities could cause our revenues to decrease significantly and impact our income from operations. If we do not broaden our customer base, we will continue to depend on these few customers for the majority of our revenues. Additionally, if we are unable to negotiate favorable business terms with these customers in the future, our revenues and gross profits may be insufficient to allow us to achieve and/or sustain profitability or continue operations.
The failure of our licensees to perform under any of our existing licensing agreements or the failure of our licensees/partners to develop and obtain regulatory approval for their product candidates or the failure to enter into new licensing agreements could substantially affect our revenue.
One of our business strategies is to enter into license agreements with pharmaceutical companies covering the development, manufacture, use and marketing of our drug delivery devices with specific drug therapies. Under these arrangements, the partners typically assist us in the development of the product and sponsor the collection of the appropriate data for submission for regulatory approval of the use of the drug delivery device with the licensed drug therapy. Our licensees may also be responsible for distribution and marketing of the product or technologies for these therapies either worldwide or in specific territories. We are currently a party to a number of such agreements, all of which are currently in varying stages of development. We may not be able to meet future milestones established in our agreements (such milestones generally being structured around satisfactory completion of certain phases of clinical development, regulatory approvals and commercialization of our product), and thus would not receive the fees expected from such arrangements, related future royalties or product sales. Moreover, there can be no assurance that we will be successful in executing additional collaborative agreements or that existing or future agreements will result in increased sales of our drug delivery technologies or products. In such event, our business, results of operations and financial condition could be adversely affected, and our revenues and gross profits may be insufficient to allow us to achieve and/or sustain profitability.
As a result of our collaborative agreements, we are dependent upon the development, data collection and marketing efforts of our licensees. The amount and timing of resources such licensees devote to these efforts are not within our control, and such licensees could make material decisions regarding these efforts that could adversely affect our future financial condition and results of operations. If one or more of these pharmaceutical company partners fail to pursue the development or marketing of, or are unable to receive marketing approval for our and our partners’ products as planned, or fail to perform their contractual obligations in accordance with all regulatory requirements, our revenues and profits may not reach expectations or may decline. In addition, factors that adversely impact the introduction and level of sales of any drug or drug device covered by such licensing arrangements, including competition within the pharmaceutical and medical device industries, the timing of regulatory or other approvals and intellectual property litigation, may also negatively affect sales of our drug delivery technology. For instance, competition in this market could also force us or our partners to reduce the prices of our technologies below currently planned levels, which could adversely affect our revenues and future profitability. Moreover, our partners and licensees will be subject to many of the same regulatory risks as we are. To the extent that they are not able to comply with the applicable regulatory requirements or are not able to obtain or maintain regulatory product approvals, we and they may be subject to regulatory enforcement action, the performance of their obligations under their contracts with us may be inhibited, and we may not be able to realize the benefit of the relationship.
We are relying on partners such as Teva, Covis, Pfizer and Idorsia for future milestone, sales and royalty revenue. Our partners may fail to obtain FDA or foreign approvals of a product with our technologies or may be unsuccessful in commercializing a product. There is no assurance that development of our partners’ products will continue or that they will ultimately receive FDA approval in a timely manner or at all, or if FDA approved, they will be a significant revenue source for us. Significant delays in anticipated launches of these products in development may occur. While we assist our partners in some cases in obtaining regulatory approvals and advancing new products, we depend on these partners and cannot control their decision-making or progress in achieving such goals. Any potential loss of anticipated future revenue could have an adverse effect on our business and the value of your investment.
An increase in the number of competitors targeting generic and 505(b)(2) ANDA opportunities and seeking U.S. market exclusivity may adversely affect our revenues and profits.
Our ability to achieve continued growth and profitability through sales of pharmaceuticals is dependent on our and our partners’ continued success in challenging patents, developing non-infringing products or developing products with improved efficacy,
safety or usability to provide opportunities with U.S. market exclusivity or limited competition. There is substantial competition in the pharmaceutical industry. We and our partners will face competition from generic drug products, drug products that are similar to our or our partners’ products, drug products containing the same active ingredient as our or our partners’ products, and drug products for the same indication as our or our partners’ products.
Our or our partners’ products may be eligible for periods of regulatory exclusivity, as described elsewhere in this annual report. This exclusivity, however, may not adequately protect our or our partners’ products from competition. If any periods of exclusivity that we or our partners may have not adequately protected the applicable product or if we or they do not receive or lose anticipated periods of regulatory exclusivity, we or they may be subject to abbreviated new drug application (“ANDA”) and/or 505(b)(2) competition sooner than we anticipate. We or our partners may also be subject to increased generic competition sooner than anticipated as the FDA, Congress, and the Administration have taken steps to facilitate the approval of generic products and increase competition in the prescription drug market. New legislative and regulatory efforts could ultimately have an adverse impact on our business and results of operation.
Further, regardless of any granted exclusivities, we or our partners may face competition from products or product uses that are not otherwise blocked by our or our partners’ patents or exclusivities. For example, exclusivity does not prevent physicians from prescribing a similar product even if it is not approved for the same indication. By further example, in 2019, the FDA approved Clarus’s product, JATENZO®, an oral testosterone undecanoate capsule for testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of endogenous testosterone. The introduction of JATENZO®, which was launched for commercial sale in February 2020, and other oral or competing testosterone products may materially impact our sales of XYOSTED®. Moreover, we or our partners may face competition from other products intended for the same use and/or that otherwise contain the same active ingredients, which may be less expensive than our or our partners’ products. Any increase or changes in the competitive landscape for our or our partners’ products may impact product sales and the amount that can be charged for a given product.
Additionally, the number of generic manufacturers targeting significant new generic opportunities with Hatch-Waxman exclusivity, or which are complex to develop, continues to increase. Other companies may also be developing drugs using the 505(b)(2) pathway that are substantially similar to our products and/or product candidates. The failure to successfully develop and commercialize highly complex products could adversely affect our sales and profitability. For instance, to the extent that another company receives a period of regulatory exclusivity, the FDA may not accept or make our application effective during that company’s exclusivity period. This would delay our and our partners’ marketing of products and may prevent us or them from establishing a sufficient market share for our product. Similarly, should another company obtain FDA approval for a pharmaceutically equivalent product to one of our product candidates, we may no longer be able to use the 505(b)(2) pathway. In that case, it is the FDA’s policy that the appropriate submission would be an ANDA. We may, however, not be able to immediately submit an ANDA or have an ANDA approval made effective, as we could be blocked by others’ periods of patent and regulatory exclusivity protection.
Although we have applied for and/or have received several patents and trademarks, we may be unable to protect our intellectual property, which would negatively affect our ability to compete.
Our success depends, in part, on our ability to obtain and enforce patents for our products and device technologies and to preserve our trade secrets and other proprietary information. If we cannot do so, our competitors may exploit our innovations and deprive us of the ability to realize revenues and profits from our developments.
We currently hold numerous patents and have numerous patent applications pending in the U.S. and other countries. Our current patents may not be valid or enforceable and may not protect us against competitors that challenge our patents, obtain their own patents that may have an adverse effect on our ability to conduct business, or are able to otherwise circumvent our patents. Additionally, our products and technologies are complex, and one patent may not be sufficient to protect our products where a series of patents may be needed. Further, we may not have the necessary financial resources to enforce or defend our patents or patent applications. Even issued patents may later be modified or declared invalid by the U.S. Patent and Trademark Office by analogous foreign offices or in legal proceedings. In addition, any patent applications we may have made or may make relating to inventions for our actual or potential products and technologies may not result in patents being issued or may result in patents that provide insufficient or incomplete coverage for our inventions.
We may seek to protect our patent rights by asserting an allegation of infringement against third parties. For instance, for any products approved via the NDA pathway, we will be required to submit certain patent information for inclusion in the FDA’s Orange Book. There is no guarantee, however, that we will be able to obtain patents that may be included in the Orange Book. To the extent that we do not include a patent in the Orange Book, we would not be able to avail ourselves of the
protections provided in the Hatch Waxman Act, including the possibility of a 30-month stay. To the extent that we include a patent in the Orange Book that should not be included, we could also face legal action.
If third parties identify our products as reference listed drugs in any ANDAs or 505(b)(2) applications, they will be required to provide patent certifications in their applications for our listed patents, and notifications to us. In the event such third parties make paragraph IV certifications, we would be entitled to file a patent infringement lawsuit, and if that is accomplished within 45 days after receiving the notification, it would trigger a 30-month stay against the FDA making the approval of the third party’s application effective. Patent litigation is costly and time consuming and the outcome is uncertain. There is no assurance of success with any patent litigation. Depending on the ultimate outcome of the litigation it may have an adverse effect on results of operations and our market penetration. We may also determine that it is not in our business interest to file a patent infringement lawsuit in response to a paragraph IV certification.
To protect our trade secrets and proprietary technologies and processes, we rely, in part, on confidentiality agreements with employees, consultants and advisors. These agreements may not provide adequate protection for our trade secrets and other proprietary information in the event of any unauthorized use or disclosure, or if others lawfully and independently develop the same or similar information. In addition, we may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties. We employ individuals who were previously employed at other biotechnology or pharmaceutical companies. We may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise used or disclosed confidential information of our employees’ former employers or other third parties. We may also be subject to claims that former employers or other third parties have an ownership interest in our patents. Litigation may be necessary to defend against these claims. There is no guarantee of success in defending these claims, and if we do not prevail, we could be required to pay substantial damages and could lose rights to important intellectual property. Even if we are successful, litigation could result in substantial cost and be a distraction to our management and other employees.
Others may bring infringement claims against us, which could be time-consuming and expensive to defend and the outcomes could be uncertain.
Third parties may claim that the manufacture, use or sale of our drug delivery technologies infringes their patent rights. As with any litigation where claims may be asserted, we may have to seek licenses, defend infringement actions or challenge the validity of those patents in the U.S. Patent and Trademark Office or the courts. If these claims are not resolved favorably, we may not be able to continue to develop and commercialize our product candidates. Even if we were able to obtain rights to a third party’s intellectual property, these rights may be non-exclusive, thereby giving our competitors potential access to the same intellectual property. Moreover, because we are developing and may develop products using the ANDA and/or 505(b)(2) pathways, we may face a greater risk of patent infringement lawsuits and associated 30-month stay in the event that we or our partners make a paragraph IV certification as part of our FDA marketing application. If we are found liable for infringement or are not able to have these patents declared invalid or unenforceable, we may be liable for significant monetary damages, encounter significant delays in bringing products to market or be precluded from participating in the manufacture, use or sale of products or methods of drug delivery covered by others’ patents. Any litigation could be costly and time-consuming and could divert the attention of our management and key personnel from our business operations. In addition, there is risk that a court will order us or our partners to pay the other party damages for having violated the other party's patents. We may not have identified, or be able to identify in the future, U.S. or foreign patents that pose a risk of potential infringement claims. In addition, a 505(b)(2) application or ANDA approval will not be made effective until any existing nonpatent exclusivities have expired or, if possible, is carved out from the label. Accordingly, we may invest a significant amount of time and expense in the development of one or more product candidates only to be subject to significant delay and patent litigation. Ultimately, we may be unable to commercialize some of our product candidates as a result of patent infringement claims, which could potentially harm our business.
Product liability, product recalls and related claims could harm our business.
The development, manufacture, testing, marketing and sale of pharmaceutical products and medical devices are associated with significant risks of product liability claims or recalls. Side effects or adverse events known or reported to be associated with, or manufacturing defects in, the products sold by us could exacerbate a patient’s condition or could result in serious injury or impairments or even death. This could result in product liability claims and/or recalls of one or more of our products. Product liability claims may be brought by individuals seeking relief for themselves, or by groups seeking to represent a class of injured patients. Further, third party payers, either individually or as a putative class, may bring actions seeking to recover monies spent on one of our products. While we have not had to defend against any product liability claims to date, as sales of our products increase, we may have product liability claims made against us. The risk of product liability claims may also increase if a company receives a warning letter from a regulatory or other enforcement agency. We cannot predict the frequency, outcome or
cost to defend any such claims. Product liability insurance coverage is expensive, can be difficult to obtain and may not be available in the future on acceptable terms, or at all. Our product liability insurance may not cover all of the future liabilities we might incur in connection with the development, manufacture or sale of our products. In addition, we may not be able to continue to obtain insurance on satisfactory terms or in adequate amounts.
While we maintain product and clinical trial liability insurance and evaluate our insurance requirements on an ongoing basis, a successful claim or claims brought against us in excess of available insurance coverage could subject us to significant liabilities and could have a material adverse effect on our business, financial condition, results of operations and growth prospects. Such claims could also harm our reputation and the reputation of our products, adversely affecting our ability to market our products successfully. Product liability claims can also result in regulatory consequences, including, but not limited to investigations and regulatory enforcement actions, as well as recalls, revocation of approvals, or labeling, marketing or promotional restrictions or changes. In addition, defending a product liability lawsuit is expensive and can divert the attention of key employees from operating our business. Such claims can also impact our ability to initiate or complete clinical trials.
To the extent that a product fails to conform to its specifications or comply with the applicable laws or regulations, we or our partners may be required to or may decide to voluntarily recall the product or regulatory authorities may request or require that we recall a product even if there is no immediate potential harm to a patient. Any recall of our products or products or their components that we supply to our partners could materially adversely affect our business by rendering us unable to sell those products or components for some time and by adversely affecting our reputation. Recalls are costly and take time and effort to administer. Even if a recall only initially relates to a single product, product batch, or a portion of a batch, recalls may later be expanded to additional products or batches or we or our partners may incur additional costs and need to dedicate additional efforts to investigate and rule out the potential for additional impacted products or batches. Moreover, if any of our partners recall a product due to an issue with a product or component that we supplied, they may claim that we are responsible for such issue and may seek to recover the costs related to such recall or be entitled to certain contractual remedies from us, Recalls may further result in decreased demand for our or our partners’ products, could cause our partners or distributors to return products to us for which we may be required to provide refunds or replacement products, or could result in product shortages. Recalls may also require regulatory reporting and prompt regulators to conduct additional inspections of our or our partners’ or contractors’ facilities, which could result in findings of noncompliance and regulatory enforcement actions. A recall could also result in product liability claims by individuals and third-party payers. In addition, product liability claims or other safety issues could result in an investigation of the safety or efficacy of our products, our manufacturing processes and facilities, or our marketing programs conducted by the FDA or the authorities of the EU member states. Such investigations could also potentially lead to a recall of our products or more serious enforcement actions, limitations on the indications for which they may be used, or suspension, variation, or withdrawal of approval. Any such regulatory action by the FDA, the European Medicines Agency (“EMA”) or the competent authorities of the EU member states could lead to product liability lawsuits as well.
We depend on information technology and computer systems to operate our business, and any failures or interruptions in our internal computer systems, including a data breach or cybersecurity incident, could have a negative impact on our business.
Despite the implementation of security measures, our internal computer systems and those of our current and any future partners, contractors and consultants are vulnerable to cybersecurity attacks including damage from computer viruses, unauthorized access, attacks by computer hackers and ransomware, natural disasters, terrorism, war and telecommunication and electrical failures. While we have not experienced any such material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our manufacturing activities, development programs and business operations. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential, protected health or proprietary information, we could incur liability or damage to our reputation, and the further commercialization and development of our products and product candidates could be delayed. Likewise, data privacy or security breaches by employees or others may pose a risk that sensitive data, including our intellectual property, or trade secrets or the personal information of our employees, patients or other business partners may be exposed to unauthorized persons or to the public. There can be no assurance that our efforts, or the efforts of our partners and vendors, will prevent service interruptions, or identify breaches in our systems, that could adversely affect our business and operations and/or result in the loss of critical or sensitive information, which could result in financial, legal, business or reputational harm to us. In addition, our liability insurance may not be sufficient in type or amount to cover us against claims related to security breaches, cyberattacks and other related breaches.
Our corporate compliance program cannot guarantee that we are in compliance with all potentially applicable laws and regulations, and we have incurred and will continue to incur costs relating to compliance with applicable laws and regulations.
As a pharmaceutical technology company, we are subject to a large body of legal and regulatory requirements, guidance, and recommendations from a variety of regulatory authorities, such as the FDA, the EMA, and HHS OIG. In addition, as a publicly traded company we are subject to significant regulations, including the Sarbanes-Oxley Act of 2002. While we have developed and instituted a corporate compliance program based on what we believe are the current best practices and continue to update the program in response to newly implemented regulatory requirements and guidance, we cannot ensure that we are or will be in compliance with all potentially applicable regulations. Failure to comply with all potentially applicable laws and regulations could lead to the imposition of fines, cause the value of our common stock to decline, and impede our ability to raise capital or list our securities on certain securities exchanges.
We face uncertainty and risks related to the outbreak of the novel coronavirus disease, COVID-19, which could significantly disrupt our operations and may materially and adversely impact our business and financial conditions.
The Pandemic continues to evolve, including the spread of new more contagious virus strains, and the related risks and uncertainty could materially and adversely affect our business, operating results and financial conditions.
Our sales force has been subject to varying limitations on its ability to visit physicians, and we are utilizing virtual meeting platforms and other forms of social media to connect with our existing and potential customers and healthcare professionals. The restrictions and closures imposed as a result of the Pandemic have also limited patient access to physicians, and we have experienced, and may continue to experience, a decrease in new prescriptions for our proprietary products. Our partners may also experience a decrease in demand for our partnered products due to the Pandemic or the related restrictions. While we have taken measures to help minimize the potential impact of the various government orders, the effects of these restrictions may negatively impact productivity and demand for our products, disrupt our business and delay development programs and timelines, the magnitude of which will depend, in part, on the length and severity of the restrictions and other limitations on our ability to conduct our business in the ordinary course. These restrictions and others in the future, as well as the continued uncertainty on the duration, scope and severity of the Pandemic, could negatively impact our business, operating results and financial condition.
We currently rely on many third parties to source active pharmaceutical ingredient and drug products, manufacture and assemble our devices, distribute finished products and provide various logistics activities for our business. If any of these third parties are adversely impacted by the Pandemic or the restrictions resulting from the outbreak, for example, one of our third party manufacturers’ facility had a temporary shutdown as a result of a positive COVID-19 diagnosis by an employee, if they are not able to obtain necessary supplies, or if they need to prioritize other products or customers over us or our partners, we may experience delays or disruptions in our product supply chain which could have a material and adverse impact on our business. Additionally, if we or any of these third parties require a regulatory authority inspection, we or they may be delayed in obtaining such inspection as a result of the Pandemic. These third parties may also need to deviate from their standard manufacturing procedures as a result of the Pandemic, which could adversely impact our or our partners’ products. These third parties may also need to deviate from their standard manufacturing procedures as a result of the Pandemic, which could adversely impact our or our partners’ products.
In addition, to the extent that we or our partners are conducting clinical trials, the Pandemic could cause delays or disruptions in these or future development programs. The foregoing may require that we consult with relevant review and ethics committees, Institutional Review Boards (“IRBs”) and the FDA, and could negatively impact our business. We may also need to make filings to the applicable regulatory authorities to account for changes that are necessary to continue to adapt to the Pandemic.
As the Pandemic continues, it may impact our and our partner’s business operations in any number of ways. This has been recognized by the FDA, which has promulgated a number of guidance specific on operations during the Pandemic. As the Pandemic develops, the regulatory guidance may continue to change and evolve, requiring that we and our partners continue to adapt to new requirements.
The full extent to which the Pandemic may impact our business or the economy as a whole is unknown and will depend on future developments, which are highly uncertain and cannot be predicted, such as the ultimate spread and rate of infection, the duration of the Pandemic, travel restrictions and social distancing requirements, business closures or business disruptions and the effectiveness of actions taken in the U.S. and in other countries to contain and treat the disease and to address its impact, including on financial markets or otherwise. These effects could have a material adverse impact on our business and operations. To the extent the Pandemic adversely affects our business and financial results, it may also have the effect of heightening many of the other risks described in this section and in the “Risk Factor” section in our other filings with the Securities and Exchange Commission. Additional risks and uncertainties not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, financial condition and/or operating results.
We have historically incurred significant losses, and there is no guarantee that we can sustain or grow our profitability.
Although we generated net income of $46.3 million and $56.2 million for the years ended December 31, 2021 and 2020, respectively, we incurred net losses of $2.0 million for the year ended December 31, 2019. In addition, we had an accumulated deficit as of December 31, 2021 of $176.3 million. The costs for research and development of our products, product candidates and drug delivery technologies, along with marketing and selling expenses and general and administrative expenses, have been the principal causes of our historical losses. Although we have reported net earnings and earnings per share in recent two most recent annual periods, there is no guarantee we will continue to post profitable results of operations or maintain profitability on an annual or quarterly basis.
Risks Related to Regulatory Matters
Our and our partners’ product candidates are subject to the inherent risk of product development and clinical trials, in that product development and clinical trials may not be successful, and they may not receive regulatory marketing approval on a timely basis or at all. If we or our partners fail to obtain, or have delays in obtaining, regulatory approvals for any product candidates, our business, financial condition and results of operations may be materially adversely affected.
The design, development, testing, manufacturing and marketing of pharmaceutical compounds and medical devices are subject to regulation by governmental authorities, including the FDA and comparable regulatory authorities in other countries and is an inherently risky and uncertain process. To conduct our and our partners’ clinical and preclinical studies, we and they rely on third parties, including CROs and clinical trial sites to carry out the studies in accordance with the written protocol, the instructions, our and our partners’ agreements with them, and the applicable regulatory requirements. There is no guarantee that we or our partners will be able to negotiate agreements with these third parties on acceptable terms, on a timely basis, or at all. To the extent that these third parties do not carry out their responsibilities, as is required, or to the extent that we, our partners, or such third parties terminate the applicable agreements, we or our partners may need to replace them, which may take significant time, effort, and expense. Additionally, we or our partners may be subject to regulatory enforcement action for such third parties’ and our or our partners’ actions, and the FDA or foreign regulatory authorities may find that the study data that is generated cannot form the basis for approval of a marketing application, requiring that we or our partners conduct additional preclinical and clinical studies. Moreover, investigators and CROs may be subject to conflicts of interest that compromise or appear to compromise the resulting data. Such third parties may also have relationships with other entities that they may prioritize over the conduct of our or our partners’ studies.
Despite the time and expense associated with preclinical and clinical studies, failure can occur at any stage, and we or our partners could encounter problems that cause us or they to repeat or perform additional preclinical studies, chemistry, manufacturing and controls (“CMC”) studies or clinical trials. We may experience numerous unforeseen events during, or as a result of, clinical trials that could delay or prevent our ability to receive marketing approval or commercialize our product candidates or that may increase the cost of development, including, failure to receive FDA or IRB authorization to begin or continue a trial, negative or inconclusive results, slow or insufficient subject enrollment, failure to obtain adequate clinical supply of product candidates that meet the applicable regulatory quality requirements, and failure by us, our partners, CROs, and clinical trial sites to follow the applicable regulatory requirements, including GCPs. We or our partners may also not have sufficient funding to conduct or complete a clinical trial or pay the substantial FDA application user fees.
The FDA and similar foreign authorities could also delay, limit or deny approval of a product candidate for many reasons, including because they: may not deem a product candidate to be adequately safe and effective or, in the case of a generic drug product, bioequivalent to the reference listed drug; may not find that we have adequately bridged to the reference listed drug, in the case of a 505(b)(2) application; may not find the data, including foreign data, from preclinical studies, CMC studies and clinical trials to be sufficient to support a claim of safety and efficacy; may interpret data from preclinical studies, CMC studies and clinical trials significantly differently than we or our partners do; may not approve the manufacturing processes or facilities associated with our product candidates; may not agree with the pathway that we or our partners have chosen for our product candidates, requiring us to pursue more difficult approval pathways, including submitting full NDAs, or may not agree with our or our partners’ intended indications; may find that our or our partners reliance on a reference listed drug for an ANDA or 505(b)(2) application or literature for a 505(b)(2) application is not appropriate; may not agree with the design and/or implementation of our clinical and/or pre-clinical studies; may require us to conduct additional clinical and/or pre-clinical studies or gather additional information or data; may find safety or efficacy issues with respect to a reference listed drug, either before or after a product candidate’s approval; may change approval policies (including with respect to our product candidates’ class of drugs), or adopt new regulations; may not meet their review goal dates; or may not accept a submission due to, among other reasons, the content or formatting of the submission. Significant delays also could shorten any periods during which we or our partners may have the exclusive right to commercialize our product candidates, which would allow competitors to bring products to market before we do.
Undesirable side effects caused by any product candidate that we or our partners develop, a lack of bioequivalence for ANDA product candidates, and/or an inability to demonstrate product candidate efficacy could result in the denial of regulatory approval by the FDA or other regulatory authorities for any or all targeted indications or cause us or our partners to evaluate the future of our development programs. Undesirable side effects could also interrupt, delay, or halt clinical trials. The regulatory review and approval process is lengthy, expensive and inherently uncertain.
Our and our partners’ reliance on FDA’s 505(b)(2) and ANDA pathways may also impact the risk of development that we would not be subject to under a full NDA. These pathways are continually evolving. Based on evolving regulatory policies, we or our partners may not be able to use the 505(b)(2) or ANDA pathway in the future, requiring that we or they pursue the costlier and time consuming 505(b)(1) full NDA pathway. We or our partners may also face delays or impediments to the approval of any product candidates if a competitor files a citizen petition with the FDA. Moreover, any FDA intervening approvals of drug products that are the same or similar to our or our partners’ product candidates could impact our potential market position and prospects, as well as impact the approval of our or our partners’ product candidates. By example, should the FDA approve a product that is pharmaceutically equivalent to one of our or our partners’ 505(b)(2) NDA product candidates before we or they submit a marketing application, we or they would be required to change the marketing application to an ANDA application. Similarly, should FDA approve a product that is more similar to any of our or our partners’ ANDA product candidates than the current reference listed drug, we or our partners may be required to change the reference listed drug for the ANDA. Either of these scenarios could require additional development work, and clinical or preclinical studies. FDA intervening approvals could also delay the timeframe within which we or our partners may submit product applications to the FDA or within which the FDA may make approvals of such applications effective, due to periods of patent protections and regulatory exclusivities for the newly approved product. Because the FDA cannot disclose whether such predicate product is under development or has been submitted at any time during another company’s review cycle, we would not know whether there are any intervening products or applications until such product or application is approved.
Should the FDA or another regulatory authority refuse to approve any of our or our partners’ product candidates, we or they will be delayed in marketing, may need to conduct additional studies and collect additional data and information, and may need to make changes to the product candidates or their manufacturing processes, any of which could materially harm our business and results of operation. Moreover, if granted, any regulatory approvals may be subject to certain limits or other costly and burdensome requirements, such as labeled warnings, including box warnings, limitations on the indicated use, and post-approval requirements. The FDA also can withdraw product clearances and approvals for failure to comply with regulatory requirements or if unforeseen problems follow initial marketing. Any limitation on use imposed by the FDA or foreign regulatory agencies would adversely affect the marketing of these products and our ability to generate product revenue, which would adversely affect our financial condition and results of operations.
With respect to any new products, we may also face increased risk with respect to regulatory approval, compliance and commercialization. By example, while TLANDO® has received tentative FDA approval, it may never receive final approval. Moreover, to the extent that we do not have prior experience with a specific kind of product, such as the TLANDO® gel capsules, we will need to acquire the necessary experience and expertise to successfully manufacture and commercialize the product, which we may never be able to do.
In other jurisdictions, we, and the pharmaceutical companies with which we are developing technologies (both drugs and devices), must obtain required regulatory approvals from regulatory agencies and comply with extensive regulations regarding safety and quality. If approvals to market the products are delayed, if we fail to receive these approvals, or if we lose previously received approvals, our revenues may not materialize or may decline. We may not be able to obtain all necessary regulatory approvals. Additionally, clinical data that we generate or obtain from partners from FDA regulatory filings may not be sufficient for regulatory filings in other jurisdictions and we may be required to incur significant costs in obtaining those regulatory approvals.
Because our and our partners’ products and product candidates are considered to be drug/device combination products, the approval and post-approval requirements that we and they are required to comply with will be more complex.
Most of our and our partners’ products and product candidates are considered to be drug/device combination products by the FDA, consisting of a drug product and a drug delivery device. If marketed individually, each component would be subject to different regulatory pathways and reviewed by different centers within the FDA. A combination product, however, is assigned to a center that will have primary jurisdiction over its pre-market review and regulation based on a determination of the product’s primary mode of action, which is the single mode of action that provides the most important therapeutic action. In the case of our and our partners’ products and product candidates, the primary mode of action is attributable to the drug component of the products, which means that the Center of Drug Evaluation and Research has primary jurisdiction over the products’ premarket development and review. These products and product candidates will be and have been subject to the FDA drug
approval process and will not require a separate FDA clearance or approval for the device component. Even though these products and product candidates will not require a separate FDA clearance or approval, both the drug and device centers within the FDA will review the marketing application for safety, the efficacy of both the drug and device component, including the design and reliability of the injector, and a number of other different areas, such as to ensure that the drug labeling adequately discloses all relevant information and risks, and to confirm that the instructions for use are accurate and easy to use. These reviews could increase the time needed for review completion of a successful application and may require additional studies, such as usage studies, to establish the validity of the instructions. Such reviews and requirements may extend the time necessary for the approval of drug-device combinations. In the case of combination product candidates for which we or our partners are seeking approval via the ANDA pathway, it is also possible that the agency may decide that the unique nature of combination products leads it to question the claims of bioequivalence and/or same labeling, resulting in the need to refile the application under Section 505(b)(2). This may result in delays in product approval and may cause us or our partners to incur additional costs associated with testing, including clinical trials. Approval via the 505(b)(2) pathway may also result in additional selling expenses and a decrease in market acceptance due to the lack of substitutability by pharmacies or formularies. In addition, approval under the 505(b)(2) or ANDA regulatory pathway is not a guarantee of an exclusive position for the approved product in the marketplace.
Further, although precedent and guidance exist for the approval of such combination products, there is no assurance that the FDA will not change what it requires or how it reviews submissions. Changes in review processes or the requirement for the study of combination products could delay anticipated launch dates or be cost prohibitive. Such delay or failure to launch these products or devices could adversely affect our revenues and future profitability. If our or our partners’ combination product candidates are approved, we, our partners, and any of our respective contractors will be required to comply with FDA regulatory requirements related to both drugs and devices. For instance, drug/device combination products must comply with both the drug cGMPs and device QSRs. Depending on whether the drug and device components are at the same facility, however, the FDA regulations provide a streamlined method to comply with both sets of requirements. The FDA has specifically promulgated guidance on injectors, which discuss FDA’s requirements with respect to marketing application and post-market injector design controls and reliability analyses. Additionally, drug/device combination products will be subject to additional FDA and constituent part reporting requirements. These requirements will require additional effort and monetary expenditure to ensure that our and our partners’ products and product candidates.
We and our partners are subject to ongoing obligations and continued regulatory review, which may result in significant additional expense for our and their approved and unapproved products. Failure to comply with these obligations could result in regulatory and/or legal consequences.
Our and our partners’ products and product candidates are subject to extensive and rigorous government regulation by the FDA and foreign regulatory agencies, including requirements related to research, development, pre-clinical and clinical testing before and after product approval, manufacture, safety, effectiveness, recordkeeping, reporting, labeling, packaging, storage, distribution, deviation, approval, facility registration and product listing, the payment of user fees, advertising, marketing, promotion, sale, distribution, sampling, and import and export of pharmaceutical and medical device products. Because our and our partners’ products and product candidates are drug/device combination products, we and they will have to comply with more regulatory requirements that would otherwise be required for products that are not combination products. Failure to comply with FDA and other applicable regulatory requirements may, either before or after product approval, subject us to administrative or judicially imposed sanctions. Moreover, were we or our partners to seek regulatory approval for additional indications or uses of any products that we or they may have already received marketing approval for, we or they would be subject to the risks of product development, including the failure to obtain regulatory approval. The applicable FDA, HHS and other governmental policies, laws, and regulations may also change, and additional laws, policies, and regulations may be enacted that could prevent, limit, or delay regulatory approval of our product candidates or products, or that could impose additional regulatory obligations on us.
The FDA and foreign regulatory agencies will continue to monitor products after approval for continued safety, efficacy, and compliance. We, our partners, and our independent contractors will also be subject to periodic unannounced inspections by the FDA to monitor and ensure compliance with regulatory requirements. Later discovery of previously unknown adverse events or that the drug is less effective than previously thought or other problems with our products, manufacturers, or manufacturing processes, or failure to comply with regulatory requirements both before and after approval, may yield various results, including warning letters, untitled letters, cyber letters, manufacturing and distribution restrictions, changes to product labeling, post-marketing study or other requirements such as REMS, refusal to approve marketing applications or supplements, withdrawal of marketing application approvals, removal of the product from the market, labeling or promotional material modifications, product recalls, market withdrawals, field corrections, clinical holds and suspensions of clinical studies, fines, penalties, disgorgement, corporate integrity agreements, consent decrees, seizure, injunctions, prohibition on importing and exporting, dear healthcare provider letters, adverse publicity, FDA debarment, debarment from government procurement programs or
refusal of orders under existing contracts, and exclusion from federal healthcare programs, among other consequences. Any of these events could have other material adverse effects on our operations and business.
For certain of our products, we and our independent contractors, distributors, prescribers, and dispensers are required to comply with regulatory requirements related to controlled substances, which will require the expenditure of additional time and will incur additional expenses to maintain compliance.
Certain of our products are controlled substances and accordingly, we, and our contractors, distributors, prescribers, and dispensers must comply with Federal controlled substances laws and regulations, enforced by the U.S. Drug Enforcement Administration (“DEA”), as well as state-controlled substances laws and regulations enforced by state authorities. These requirements include, but are not limited to, registration, security, recordkeeping, reporting, storage, distribution, importation, exportation, inventory, and other requirements. These requirements are enforced by the DEA through periodic inspections. Not only must continuous controlled substance registration be maintained, but compliance with the applicable controlled substance requirements will require significant efforts and expenditures, which could also inhibit the successful commercialization. If we and our contractors, distributors, prescribers, and dispensers do not comply with the applicable controlled substance requirements, we or they may be subject to administrative, civil or criminal enforcement, including civil penalties, refusals to renew necessary registrations, revocation of registrations, criminal proceedings, or consent decrees.
Any relationships with healthcare professionals, principal investigators, consultants, customers (actual and potential) and third-party payers in connection with our current and future business activities are and will continue to be subject, directly or indirectly, to federal and state healthcare fraud and abuse laws, false claims laws, marketing expenditure tracking and disclosure (or “sunshine”) laws, government price reporting, and health information privacy and security laws. If we are unable to comply, or have not fully complied, with such laws, we could face penalties, contractual damages, reputational harm, diminished profits and future earnings.
Our business operations and activities may be directly, or indirectly, subject to various federal, state and local fraud and abuse laws, including, without limitation, the federal Anti-Kickback Statute and the federal FCA and similar laws in some state and foreign markets. These laws may impact, among other things, our current activities with principal investigators and research subjects, as well as proposed and future sales, marketing and education programs. In addition, we may be subject to patient privacy regulation by the federal government, state governments and foreign jurisdictions in which we conduct our business. The laws in the U.S. that may affect our ability to operate are further discussed above in the “Government Regulation” section of this Form 10-K.
Changes in healthcare law and implementing regulations, as well as changes in healthcare policy, may impact our business in ways that we cannot currently predict, and these changes could have a material adverse effect on our business and financial condition.
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010 (together “the Healthcare Reform Act”) substantially changes the way healthcare is financed by both governmental and private insurers, and significantly impacts the pharmaceutical industry. Recently, the law’s individual health insurance mandate was repealed and manufacturers’ responsibility for the cost of prescriptions in the Medicare Part D donut hole has increased. We expect that the Healthcare Reform Act, as currently enacted or as it may be amended in the future, and other healthcare reform measures at the federal and state level that may be adopted in the future, could have a material adverse effect on our industry generally and on our ability to maintain or increase sales of our existing products or to successfully commercialize our product candidates, if approved. For example, CMS recently finalized (and subsequently rescinded) a rule establishing a pricing model for Medicare Part B drugs based on the average price among other industrialized countries. This type of regulatory development, including if extended to other federal healthcare programs, could have a significant impact on our business.
To help patients afford certain of our products, we offer discount, rebate, and co-pay coupon programs. Co-pay coupon programs have received some negative publicity related to their use to promote branded pharmaceutical products over other less costly alternatives. CMS recently has issued a regulation imposing additional obligations on manufacturers in order to continue excluding such programs from government pricing calculations to avoid payment of increased Medicaid rebates. In recent years, other pharmaceutical manufacturers have been named in class action lawsuits challenging the legality of their co-pay programs under a variety of federal and state laws. In addition, at least one insurer has directed its network pharmacies to no longer accept co-pay coupons for certain specialty drugs the insurers identified. Our co-pay coupon programs could become the target of similar lawsuits or insurer actions. It is possible that the outcome of litigation against other manufacturers, changes in insurer policies regarding co-pay coupons, and/or the introduction and enactment of new legislation or regulatory action could restrict or otherwise negatively affect these programs.
We are dependent on third parties to decide to use our and our partners’ products and to make them readily available at the point of care throughout their networks of pharmacies.
In addition to extensive internal efforts, the successful commercialization of our and our partners’ products require many third parties, over which we have no control, to decide to use them, and to make them readily available at the point of care throughout their networks of pharmacies. These third parties include HMOs, long term care facilities, and pharmacy benefit managers, or PBMs, which use pharmacy and therapeutics committees, commonly referred to as P&T committees, to make purchasing and reimbursement decisions. We cannot guarantee that we and/or our partners will be successful in getting the approvals we need from sufficient P&T committees quickly enough to maintain and grow sales of our or our partners’ products.
Risks Related to our Common Stock
The market price of our common stock has been, and may continue to be volatile and fluctuate significantly, which could result in substantial losses for investors.
The trading price for our common stock has been, and we expect it to continue to be, volatile. The price at which our common stock trades depends upon a number of factors, including our historical and anticipated operating results, our financial situation, clinical trial results, announcements of technological innovations or new products by us, our partners or our competitors, our ability or inability to raise the additional capital we may need and the terms on which we raise it, and general market and economic conditions. Some of these factors are beyond our control. Broad market fluctuations may lower the market price of our common stock and affect the volume of trading in our stock, regardless of our financial condition, results of operations, business or prospects.
We are at risk of securities class action and similar litigation.
In the past, securities class action litigation has often been brought against a company following a decline in the market price of its securities. This risk is especially relevant for us because pharmaceutical companies have experienced significant stock price volatility in recent years. For example, on October 23, 2017, Randy Smith filed a complaint in the District of New Jersey, on behalf of a putative class of persons who purchased or otherwise acquired Antares securities against us, Robert F. Apple and Fred M. Powell. In addition, in January 2018, three stockholders filed separate derivative actions, one in the District of New Jersey and two in the Superior Court of New Jersey Chancery Division, Mercer County, purportedly on our behalf, against certain directors and officers, as well as the Company as a nominal defendant. There can be no assurance that we will ultimately prevail in these legal proceedings. Even if we are successful and ultimately prevail, litigation could be costly to defend and divert management’s attention and resources, which could further materially harm our financial condition and results of operations.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
If we do not generate sufficient net taxable income in the future, our ability to use our net operating loss carryforwards to offset U.S. federal taxable income may be subject to limitations, which could potentially result in increased future tax liability to us.
Anti-takeover effects of certain certificate of incorporation and bylaw provisions could discourage, delay or prevent a change in control.
Our certificate of incorporation authorizes our board of directors, without action of our stockholders, to designate and issue preferred stock in one or more series, with such rights, preferences and privileges as the board of directors shall determine. In addition, our bylaws grant our board of directors the authority to adopt, amend or repeal all or any of our bylaws, subject to the power of the stockholders to change or repeal the bylaws. In addition, our bylaws limit who may call meetings of our stockholders.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 2. PROPERTIES
Our properties consist of leased office, laboratory, warehouse and manufacturing facilities. We lease our corporate headquarters located in Ewing, New Jersey, primarily consisting of office space. We also lease a building in Minnetonka, Minnesota consisting of office, laboratory, manufacturing and warehousing space. We believe our current facilities are sufficient for our existing needs and to support future anticipated business growth.
Additional information related to lease obligations is included in Item 7 of Part II of this Annual Report on Form 10-K.
ITEM 3. LEGAL PROCEEDINGS
The information set forth under “Note 16. Commitments and Contingencies – Pending Litigation” to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K is incorporated herein by reference.
Although the results of actual, pending or threatened legal proceedings and litigation cannot be predicted with certainty, we do not believe that there is a reasonable possibility that the final outcome of these matters will have a material adverse effect on our business or financial results. Regardless of the final outcome, litigation could have an adverse impact on us because of defense or settlement costs, diversion of management resources, harm to our reputation and brand, and other factors.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our common stock trades on the NASDAQ Capital Market under the trading symbol “ATRS”.
Common Shareholders
As of February 28, 2022, we had 69 shareholders of record of our common stock and approximately 21,825 shareholders in street name. Information on securities authorized for issuance under our equity compensation plans can be found in Item 12 of Part III of this Annual Report on Form 10-K.
Dividends
We have not paid or declared cash dividends on our common stock during the past ten years and have no intention of paying cash dividends on our common stock in the foreseeable future.
Performance Graph
The graph below provides an indication of cumulative total stockholder returns (“Total Return”) for our common stock as compared with the NASDAQ Composite Index and the NASDAQ Biotechnology Stock Index. The graph covers the period beginning December 31, 2016 through December 31, 2021. The graph assumes $100 was invested at market close on December 31, 2016 in our common stock, the NASDAQ Composite Index and the NASDAQ Biotechnology Stock Index and that all dividends were reinvested. The graph is not necessarily indicative of future investment performance.
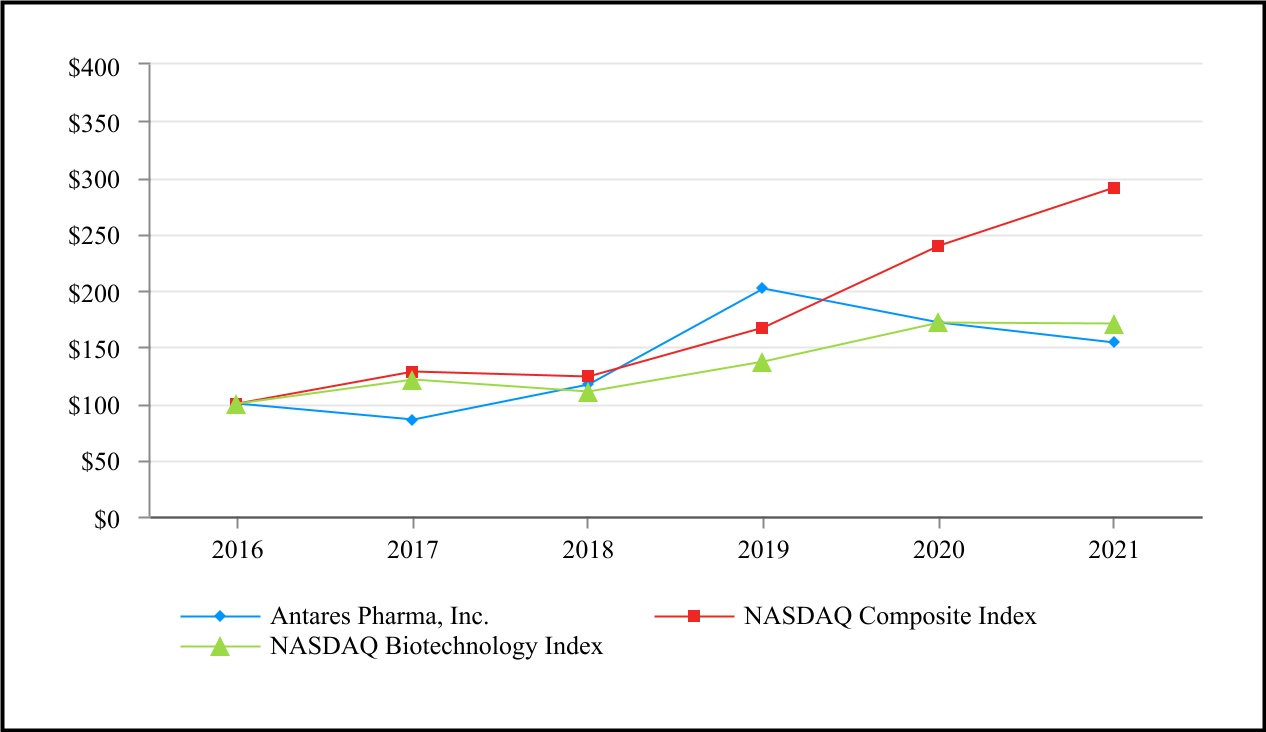
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, |
| (in actual dollars) | 2016 | | 2017 | | 2018 | | 2019 | | 2020 | | 2021 |
| Antares Pharma, Inc. | $ | 100.00 | | | $85.41 | | $116.74 | | $201.72 | | $171.24 | | $153.22 |
| NASDAQ Composite Index | 100.00 | | | 128.24 | | 123.26 | | 166.68 | | 239.42 | | 290.63 |
| NASDAQ Biotechnology Index | 100.00 | | | 121.06 | | 109.77 | | 136.56 | | 171.64 | | 170.55 |
Recent Sales of Unregistered Securities
None.
Issuer Purchases of Equity Securities
None.
ITEM 6. [RESERVED]
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Management’s Discussion and Analysis of Financial Condition and Results of Operations (“MD&A”) is designed to provide a reader of our financial statements with a narrative from the perspective of management on our financial condition, results of operations, liquidity and certain other factors that may affect our future results. Our MD&A is presented in five sections.
•Company Overview
•Results of Operations
•Liquidity and Capital Resources
•Critical Accounting Policies and Use of Estimates
•Off-Balance Sheet Arrangements
Our MD&A should be read in conjunction with the consolidated financial statements and related footnotes included in Item 8 of Part II of this Annual Report on Form 10-K and risk factors identified in Item 1A of Part I of this Annual Report on Form 10-K. Some of the statements included below are considered forward-looking statements. See the discussion regarding forward-looking statements preceding Item 1 of Part I of this Annual Report on Form 10-K.
The terms “Antares,” “we,” “our,” “us” or the “Company” in this Annual Report on Form 10-K, unless the context otherwise requires, refer to Antares Pharma, Inc. and its wholly owned subsidiaries.
Company Overview
Antares Pharma, Inc. is a specialty pharmaceutical company focused primarily on the development and commercialization of pharmaceutical products and technologies that address patient needs in targeted therapeutic areas. We develop, manufacture and commercialize, for ourselves or with partners, novel therapeutic products using our advanced drug delivery systems that are designed to provide commercial or functional advantages such as improved safety and efficacy, convenience, improved tolerability, and enhanced patient comfort and adherence. We also seek product opportunities that complement and leverage our commercial platform. We have a portfolio of proprietary and partnered commercial products and ongoing product development programs in various stages of development. We have formed partnership arrangements with several different industry leading pharmaceutical companies.
We market and sell in the U.S. our proprietary product XYOSTED® (testosterone enanthate) injection indicated for testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of endogenous testosterone. XYOSTED® is the only FDA approved subcutaneous testosterone enanthate product for once-weekly, at-home self-administration.
In December 2021, we sold certain assets used in the operation of the OTREXUP® product under an asset purchase agreement with Otter for $44.0 million, subject to finalization of changes in closing inventory to be transferred, with $18.0 million received at closing and an additional $26.0 million to be paid in installments over a one-year period. Prior to the asset sale, we marketed and sold OTREXUP® (methotrexate) injection, a subcutaneous methotrexate injection for once weekly self-administration with an easy-to-use, single dose, disposable auto injector, indicated for adults with severe active rheumatoid arthritis, children with active polyarticular juvenile idiopathic arthritis and adults with severe recalcitrant psoriasis, as a proprietary product in the U.S. In conjunction with the asset sale, we entered into a supply agreement with Otter to manufacture the VIBEX® auto-injection system device at cost plus mark-up. Otter is responsible for manufacturing, formulation and testing of methotrexate and the corresponding prefilled syringe for assembly with the device manufactured by us, along with the commercialization and distribution of OTREXUP® going forward. We also entered into a license agreement with Otter granting them a worldwide, exclusive, royalty-free, fully paid-up, irrevocable, transferable license with the right to sublicense to certain patents relating to the OTREXUP® product that may relate to other products we produce.
In October 2020, we entered into an exclusive license agreement (the “NOCDURNA® License Agreement”) with Ferring for the marketed product NOCDURNA® (desmopressin acetate) in the United States, which is indicated for the treatment of nocturia due to nocturnal polyuria (“NP”) in adults who awaken at least two times per night to urinate. We began detailing NOCDURNA® with a soft launch in the fourth quarter of 2020 and are currently executing a reintroduction of the product through a comprehensive re-launch strategy to increase awareness and demand.
In October 2021, we entered into an exclusive license agreement (the “TLANDO® License Agreement”) with Lipocine for the product TLANDO® (testosterone undecanoate) in the U.S., a twice-daily oral formulation of testosterone for testosterone replacement therapy indicated for conditions associated with a deficiency or absence of endogenous testosterone, or hypogonadism in adult males. TLANDO® was granted tentative approval from the FDA in December 2020 and will be eligible for final approval and marketing in the U.S. upon expiration of the exclusivity period previously granted to Clarus for JATENZO® on March 27, 2022. On February 3, 2022, we announced the FDA’s acceptance of our NDA resubmission for TLANDO® with a target action date set for March 28, 2022. We continue to prepare for the launch of TLANDO® in 2022 pending final FDA approval after the expiration of JATENZO®’s exclusivity period. Under the terms of the TLANDO® License Agreement, we paid Lipocine an upfront payment of $11.0 million. Lipocine is eligible for additional milestone payments up to $10.0 million and tiered royalty and commercial milestones based on net sales of TLANDO® in the U.S. We will be responsible for the manufacturing and commercialization of TLANDO®.
The TLANDO® License Agreement also grants us the option to license and develop LPCN 1111 (TLANDO XR) in the U.S., a potential once daily oral testosterone product containing testosterone tridecanoate in development for the treatment of hypogonadism in adult males. If elected, upon exercise of the option, we will be required to pay an additional $4.0 million in license fees in two installments and will be responsible for additional development and commercial milestone payments as well as tiered royalties on net sales of TLANDO XR in the U.S. In addition, we will be responsible for completing the development program including the conduct of a Phase 3 clinical trial and applying for regulatory approval in the U.S.
In collaboration with Teva, we developed a version of our VIBEX® auto injector for use in a generic epinephrine auto injector product that was approved by the FDA. Teva’s Epinephrine Injection USP is indicated for emergency treatment of severe allergic reactions including those that are life threatening (anaphylaxis) in adults and certain pediatric patients and was approved as a generic drug product with an AB rating, meaning that it is therapeutically equivalent to the branded products EpiPen® and EpiPen Jr® and therefore, subject to state law, substitutable at the pharmacy. We are the exclusive supplier of the device and Teva is responsible for commercialization and distribution of the finished product, for which we also receive royalties on Teva’s net sales.
Through our commercialization partner Teva, we sell Sumatriptan Injection USP indicated in the U.S. for the acute treatment of migraine and cluster headache in adults.
We are the exclusive supplier of the device, a variation of our VIBEX® QuickShot® subcutaneous auto injector developed by us, for the progestin hormone drug Makena® (hydroxyprogesterone caproate injection), indicated to help reduce the risk of preterm birth in women pregnant with one baby and who spontaneously delivered one preterm baby in the past. As the exclusive supplier, we perform final assembly and packaging of the commercial product and receive royalties on Covis’ net sales of the product. In October 2020, following an FDA advisory committee meeting, Covis in November 2020, received notice that the FDA is proposing to withdraw approval of Makena® (hydroxyprogesterone caproate injection). Covis formally requested a public hearing in response to the FDA's proposal to withdraw its approval and has stated that it remains committed to working with the FDA to maintain patient access to Makena® as a treatment option to reduce pre-term birth.
We are also developing with Teva a multi-dose pen for a generic form of Forteo® (teriparatide rDNA origin injection) for the treatment of osteoporosis, and were developing another multi-dose pen for a generic form of BYETTA® (exenatide injection) for the treatment of type 2 diabetes. On February 25, 2022, Teva notified us that it was terminating the exenatide program and related agreement due to a lack of commercial viability. The termination is effective August 23, 2022. Teva continues to work through the U.S. regulatory process with the FDA for teriparatide using the ANDA pathway. In 2020, Teva launched Teriparatide Injection (“teriparatide”), the generic version of Eli Lilly’s branded product Forsteo® featuring the Antares multi-dose pen used platform in several countries outside the U.S. We are responsible for the manufacturing and supply of the multi-dose pen utilized in Teva’s generic teriparatide product under an exclusive development, license and supply agreement with Teva, the scope of which is worldwide.
In August 2018, we entered into a development agreement with Pfizer to develop a combination drug device rescue pen. This rescue pen will utilize the Antares QuickShot® auto injector and an undisclosed Pfizer drug. In 2021, we continued to work on this development program, and we expect to continue development of this product candidate.
In November 2019, we entered into a global agreement with Idorsia to develop a novel, drug-device product containing selatogrel. The new chemical entity selatogrel is being developed for the treatment of a suspected AMI in adult patients with a history of AMI. Idorsia will pay for the development of the combination product and will be responsible for applying for and obtaining global regulatory approvals for the product. The parties intend to enter into a separate commercial license and supply agreement pursuant to which we will provide fully assembled and labelled product to Idorsia at cost plus mark-up. Idorsia will then be responsible for global commercialization of the product, pending FDA or foreign approval. We will be entitled to receive royalties on net sales of the commercial product.
In June 2021, Idorsia announced it had initiated its Phase 3 registration study to evaluate the efficacy and safety of self-administered subcutaneous selatogrel. The study will enroll approximately 14,000 patients who are at high risk of recurrent AMI, at approximately 250 sites in approximately 30 countries.
We are also committed to advancing our internal research and development programs and continue to invest in the development of our proprietary product pipeline. Our research and development efforts are focused primarily on leveraging our existing product and technology platforms by broadening their applications for use in other drug device combination products, as well as exploring new pharmaceutical products, technologies and drug delivery methods.
We have initiated development of a proprietary drug device combination product for the urology oncology market, identified as ATRS-1901, and have conducted formulation development work and non-clinical studies to help advance this program. In 2020, we received a response from the FDA regarding our pre-IND (Investigational New Drug) submission.
We have identified a program to develop a proprietary drug device combination product for the endocrinology market, an adrenal crisis pen, identified as ATRS-1902. The development program supports a proposed indication for the treatment of acute adrenal insufficiency, known as adrenal crisis, in adults and adolescents, using a novel proprietary auto-injector platform to deliver a liquid stable formulation of hydrocortisone. We conducted initial formulation work and developed a working prototype of a new device to support this program. We received a response from the FDA regarding our pre-IND submission and believe we have determined the regulatory and clinical path forward.
In July 2021, the FDA accepted our IND for ATRS-1902 enabling us to initiate our Phase 1 clinical study. In January 2022, we announced the positive results from the Phase 1 clinical study and were granted Fast Track designation by the FDA. The positive results support the advancement of our ATRS-1902 development program to a pivotal clinical study for the treatment of acute adrenal insufficiency, known as adrenal crisis, in adults and adolescents, using our Vai™ novel proprietary rescue pen platform to deliver a liquid stable formulation of hydrocortisone. We anticipate starting this pivotal clinical study in the second quarter of 2022 and expect to submit a 505(b)(2) NDA with the FDA by the end of 2022 pending the success of the pivotal clinical study.
We have initiated development of a proprietary drug device combination product utilizing our rescue pen technology for a rare immunology disorder, identified as ATRS-1903. Formulation development work has been conducted and we anticipate progressing this towards initial clinical testing to evaluate PK and tolerability in human subjects.
COVID-19
In December 2019, a novel strain of coronavirus (COVID-19) emerged in China, and has since spread worldwide, including every state in the U.S. On March 11, 2020, the World Health Organization declared the outbreak a Pandemic and on March 13, 2020, the U.S. declared a national emergency with respect to the outbreak. The Pandemic has impacted global economic activity and lead to disruptions in supply chain, labor shortages, business closures, travel restrictions and other health, safety and social distancing requirements.
We have taken several measures to actively manage and help minimize the impact of the ongoing Pandemic on our business. We have implemented safety measures and protocols to protect the health and safety of our employees and comply with governmental and public health guidelines while working to ensure the sustainability of our business operations and continuity of product supply. We continue to monitor the situation, including COVID-19 variants, and potential effects on our business, suppliers, partners and workforce.
We have implemented a hybrid work environment with the ability to shift remote as necessary to limit the number of individuals in our facilities to those necessary for essential functions such as research, development, manufacturing and supply. While our field-based team has resumed in-person interaction with fewer restrictions, we believe we are also well-positioned with our virtual capabilities to continue to engage healthcare professionals and patients through the ongoing Pandemic and beyond. Although, we have not experienced significant delays or disruption in our development programs or significant demand reductions for our partnered products due to the Pandemic, we continue monitor the situation, including COVID-19 variants, for potential effects on our or our partners’ clinical trials or delays or disruptions in activities with the FDA.
Although, we have taken measures to help minimize the potential impact of the Pandemic on our business, given the fluidity of the Pandemic and other macroeconomic factors, we are unable to estimate the impact the Pandemic has had on our operations or cash flows as of the date of this filing. We also believe there continues to be uncertainty around the timing and duration of any potential future disruptions due to the COVID-19 variants and the magnitude of any potential impact. As a result, we are unable to estimate the potential impact on future operations or cash flows as of the date of this filing. For more information on these risks see “Part I — Item 1A. Risk Factors — We face uncertainty and risks related to the outbreak of the novel coronavirus disease, COVID-19, which could significantly disrupt our operations and may materially and adversely impact our business and financial conditions.”
Results of Operations
The following is a discussion and analysis of our financial results, cash flows, and liquidity and capital resources for the years ended December 31, 2021 and 2020. A discussion of changes in our financial results, cash flow comparison and liquidity and capital resources for the years ended December 31, 2020 and 2019 has been omitted from this Form 10-K but may be found in Item 7 of Part II of our Annual Report on Form 10-K for the year ended December 31, 2020, filed with the SEC on March 2, 2021.
Revenue, Net
We generate revenue from proprietary and partnered product sales, license and development activities and royalty arrangements. The following table provides details about the components and drivers of our overall revenue growth:
| | | | | | | | | | | | | | | | | | | | | | | |
| Years Ended December 31, | | Increased / (Decreased) |
| (in thousands) | 2021 | | 2020 | | $ | | % |
| Proprietary product sales, net | $ | 80,016 | | | $ | 62,878 | | | $ | 17,138 | | | 27.3 | % |
| Partnered product sales | 46,651 | | | 50,956 | | | (4,305) | | | (8.4) | % |
| Total product revenue, net | 126,667 | | | 113,834 | | | 12,833 | | | 11.3 | % |
| Licensing and development revenue | 19,623 | | | 14,466 | | | 5,157 | | | 35.6 | % |
| Royalties | 37,692 | | | 21,299 | | | 16,393 | | | 77.0 | % |
| Total revenue, net | $ | 183,982 | | | $ | 149,599 | | | $ | 34,383 | | | 23.0 | % |
Product Revenue, Net
Net revenue from product sales increased 11.3% primarily due to increased sales of our proprietary products XYOSTED® and NOCDURNA® and partnered sales of OTREXUP® to Otter subsequent to sale of the product line, partially offset by a reduction in sumatriptan sales to Teva and sales of Makena® subcutaneous auto-injectors to Covis.
Sales of our proprietary products are presented net of estimated product returns and sales allowances. The OTREXUP® product line was sold to Otter in December 2021 with a supply agreement executed simultaneously; therefore, all revenue generated prior to the date of sale is included in proprietary product sales and all revenue generated subsequent to the date of sale is included in partnered products sales. The increase in propriety product sales of 27.3% was primarily attributable to continued growth in prescriptions and sales of XYOSTED®, which we launched for commercial sale in 2018, and sales of NOCDURNA®, which we in-licensed and began detailing in the fourth quarter of 2020, partially offset by a reduction in sales of OTREXUP® due to the sale of the product line to Otter in December 2021. We attribute this growth to successful marketing and launch strategies, achieving and maintaining targeted reimbursement coverage, and our ability to leverage our virtual sales capabilities to support the continued growth despite any potential softening or impact due to the global Pandemic in 2021 and 2020.
We also manufacture and sell devices, components and fully assembled packaged product to our partners Teva, Covis and Otter. Partnered product sales decreased by 8.4% due to a decrease in sumatriptan sales to Teva, lower production and sales volumes of Makena® to Covis and a decrease in shipments of epinephrine auto injectors to Teva, partially offset by revenue generated from production of OTREXUP® for Otter and higher teriparatide sales to Teva.
Licensing and Development Revenue
Licensing and development revenues include license fees received from partners for the right to use our intellectual property and amounts earned in joint development arrangements with partners under which we perform development activities or develop new products on their behalf. Fluctuations in our licensing and development revenue are generally attributable to the development timelines of our various partnered development projects, the timing of which is often controlled by our partner, and the timing of achievement of certain milestones.
Licensing and development revenue increased 35.6% primarily as a result of incremental development and maintenance activities with Teva to support replacement of molds and tooling related to the commercial production of the epinephrine auto injector and continuing development activities under other ongoing partnered development projects, partially offset by a decline in development activities with Pfizer.
Royalties
Royalties are earned in connection with licenses granted to our partners under license and development arrangements. Royalties are generally based upon a percentage of our partners’ net sales of the partnered product. Royalty revenue increased 77.0% primarily due to an increase in royalties from Teva on its net sales of generic EpiPens®.
Cost of Revenue
The following table summarizes our cost of product sales and development revenue:
| | | | | | | | | | | | | | | | | | | | | | | |
| Years Ended December 31, | | Increased / (Decreased) |
| (in thousands) | 2021 | | 2020 | | $ | | % |
| Cost of product sales | $ | 54,418 | | | $ | 53,960 | | | $ | 458 | | | 0.8 | % |
| Cost of development revenue | 13,863 | | | 9,140 | | | 4,723 | | | 51.7 | % |
| Total cost of revenue | $ | 68,281 | | | $ | 63,100 | | | $ | 5,181 | | | 8.2 | % |
| % of revenue | 37.1 | % | | 42.2 | % | | | | |
Fluctuations in cost of product sales is generally a function of the product revenue mix. Proprietary products generally have a lower cost of sales as a percentage of revenue than partnered product sales. The year-over-year increase in cost of development revenue is attributable to and relatively consistent with the growth in development revenue from partnered development activities.
Research and Development Expenses
Research and development (“R&D”) expenses consist of external costs for clinical studies and analysis activities, formulation development, engineering design work and prototype development, FDA application fees, personnel costs and other general operating expenses associated with our research and development activities.
| | | | | | | | | | | | | | | | | | | | | | | |
| Years Ended December 31, | | Increased / (Decreased) |
| (in thousands) | 2021 | | 2020 | | $ | | % |
| Research and development | $ | 14,502 | | | $ | 10,121 | | | $ | 4,381 | | | 43.3 | % |
R&D expenses increased 43.3% primarily due to our ongoing internal development programs including ATRS-1902 and ATRS-1901, along with higher employee compensation expense. Overall, R&D expense fluctuates based on phases of development and timing of clinical studies, including internal and external development costs incurred. As discussed above, we further advanced our ATRS-1902 development program with positive result from a Phase 1 clinical study for adrenal crisis rescue in January 2022 and were granted Fast Track designation by the FDA. The results support the advancement of our ATRS-1902 development program to a pivotal clinical study which we anticipate starting in the second quarter of 2022.
Selling, General and Administrative
| | | | | | | | | | | | | | | | | | | | | | | |
| Years Ended December 31, | | Increased / (Decreased) |
| (in thousands) | 2021 | | 2020 | | $ | | % |
| Selling, general and administrative | $ | 73,857 | | | $ | 62,759 | | | $ | 11,098 | | | 17.7 | % |
Selling, general and administrative expenses increased 17.7% primarily due to higher sales and marketing costs associated with the relaunch of NOCDURNA®, which we in-licensed and began detailing in the fourth quarter of 2020. The increase is also attributable to higher sales and marketing costs associated with XYOSTED®, which were down in 2020 due to the Pandemic as the various restrictions and limitations imposed during the Pandemic led to decreased spending that has returned to pre-Pandemic levels, along with higher employee compensation. General and administrative expenses also increased primarily driven by higher professional service fees, facility costs, insurance expense and employee compensation costs to support the continued growth of the business.
Gain on Sale
In December 2021, we sold certain assets used in the operation of the OTREXUP® product to Otter for $44.0 million, subject to finalization of changes in closing inventory to be transferred, with $18.0 million received at closing and an additional $26.0 million to be paid in installments over a one-year period. The gain on sale of assets of $38.6 million represents the purchase price adjusted for estimated changes in closing inventory to be transferred less net book value of the assets sold and direct transaction costs.
Income Tax Expense (Benefit)
| | | | | | | | | | | | | | | | | | | | | | | |
| Years Ended December 31, | | Increased / (Decreased) |
| (in thousands) | 2021 | | 2020 | | $ | | % |
| Income tax provision (benefit) | $ | 15,982 | | | $ | (46,280) | | | $ | 62,262 | | | 134.5 | % |
| Effective tax rate | 25.7 | % | | (466.5) | % | | | | |
Income tax expense recorded for the year ended December 31, 2021 was driven by the generation of income before income taxes of $62.3 million, resulting in an effective tax rate of 25.7%. The effective tax rate is primarily driven by the federal and state tax rates, along with discrete income tax items for compensation expense. For the year ended December 31, 2020, we recorded an income tax benefit of $46.3 million on pre-tax income of $9.9 million primarily due to the release of our valuation allowance on our deferred tax assets which favorably impacted our effective tax rate. As of December 31, 2020, we concluded that, as a result of generating pre-tax earnings, utilization of net operating loss carryovers and future projected pre-tax earnings, it is more likely than not that its deferred taxes are realizable and may be utilized to offset future tax liabilities. Excluding the release of our valuation allowance on our deferred tax assets, the effective tax rate for the year ended December 31, 2020 would have been higher than the effective tax rate for the year ended December 31, 2021 primarily due to the impact of permanent tax items on a lower income before income taxes.
Net Income and Earnings Per Common Share
| | | | | | | | | | | | | | | | | | | | | | | |
| Years Ended December 31, | | Increased / (Decreased) |
| (in thousands, except per share amounts) | 2021 | | 2020 | | $ | | % |
| Net income | $ | 46,289 | | | $ | 56,201 | | | $ | (9,912) | | | (17.6) | % |
| | | | | | | |
| Earnings per common share | | | | | | | |
| Basic | $ | 0.27 | | | $ | 0.34 | | | $ | (0.07) | | | (20.6) | % |
| Diluted | $ | 0.26 | | | $ | 0.33 | | | $ | (0.07) | | | (21.2) | % |
Liquidity and Capital Resources
As of December 31, 2021 we had cash and cash equivalents of $65.9 million. We believe that the combination of our current cash and cash equivalents, along with our projected product sales, development revenue and royalties will provide us with sufficient funds to meet our obligations, including debt maturities, and support operations through at least the first quarter of 2023. We reported net income and positive cash flows from operations for the years ended December 31, 2021 and 2020. We had an accumulated deficit as of December 31, 2021 of $176.3 million. Prior to 2020, we had not historically generated enough operating cash flow to support our operations and funded our operations through equity offerings and debt issuance.
If additional capital is needed to support our operations in the future, we may need to raise additional funds through financing, such as drawing our current credit facility, issuance of additional debt, equity or notes convertible into our common stock. However, we may be unable to obtain such financing, or obtain it on favorable terms, in which case we may be required to curtail development of new products, limit expansion of operations or accept financing terms that are not as attractive as we may desire.
Long-term Debt Financing
As of December 31, 2020, we were party to a loan and security agreement, as amended, with Hercules Capital, Inc. (the “Term Loan”). The amortizing Term Loan was secured by substantially all of our assets, excluding intellectual property, and accrued interest at a prime-based variable rate with a maximum of 9.5%, which was 8.5% in 2021. In 2021, we made principal prepayments of $20.0 million and paid a 1.0% prepayment fee. On November 1, 2021, we extinguished the Loan Agreement with Hercules Capital, Inc. and repaid the outstanding $20.0 million principal on the Term Loan, along with a 1.0% prepayment fee and the end of term charge of $1.7 million. All remaining unamortized debt issuance costs associated with the Term Loan were immediately amortized to interest expense.
On November 1, 2021, we entered into a Credit Agreement (the “Credit Agreement”) with Wells Fargo Bank, National Association, as administrative agent for the lenders, for credit facilities in an aggregate principal amount of up to $40.0 million with a maturity date of November 1, 2024. The Credit Agreement consists of a $20.0 million term loan facility (the “Term Loan Facility”) and a $20.0 million revolving credit facility (the “Revolving Credit Facility”), (collectively the “Credit Facilities”), which are secured by substantially all of our assets. The Term Loan Facility was funded upon execution of the Credit Agreement with the proceeds used to repay our $20.0 million Term Loan with Hercules Capital, Inc. and to pay fees and expenses incurred in connection with the early repayment.
Total interest-bearing debt as of December 31, 2021 was $20.0 million and we had $20.0 million of unused borrowing capacity on our Revolving Credit Facility, which is expected to be used for ongoing working capital requirements and other general corporate purposes as needed. Commitment fees are payable on the unused portion of the Revolving Credit Facility at rates between 0.30% and 0.45% based on our Consolidated Total Leverage Ratio, as defined in the Credit Agreement, remeasured quarterly. Payments under the Term Loan Facility are due in consecutive quarterly installments on the last business day of each of March, June, September and December, commencing on March 31, 2022. At our election, interest accrues at LIBOR plus the applicable margin ranging from 2.25% to 3.00%, which varies based on our Consolidated Total Leverage Ratio. The new Credit Facilities, which replaced the previous Term Loan, are expected to provide approximately $1.2 million in annual interest expense savings based on an interest rate of approximately 2.60% (one-month LIBOR rate plus the applicable margin of 2.50%) as of December 31, 2021.
Under the Credit Agreement, we are subject to customary affirmative and negative covenants, including, among others, restrictions on our ability to incur debt; create liens; make investments; merge, consolidate or dispose of assets or subsidiaries; enter into transactions with affiliates; modify accounting practices, our year end and organizational documents; pledge assets; revise nature of business; perform sale leasebacks; and enter into any restrictive agreements and customary events of default (including payment defaults, covenant defaults, change of control defaults and bankruptcy defaults). The Credit Agreement also contains financial covenants, including the ratio of consolidated total indebtedness to consolidated earnings before income, taxes, depreciation and amortization (“Consolidated EBITDA”) (“Consolidated Total Leverage Ratio”), as defined in the Credit Agreement” and the ratio of consolidated senior secured indebtedness to Consolidated EBITDA (“Consolidated Senior Secured Leverage Ratio”), as well as the ratio of Adjusted EBITDA to consolidated fixed charges (“Consolidated Fixed Charge Coverage Ratio”), as defined in the Credit Agreement. These covenants restrict our ability to purchase outstanding shares of our common stock. As of December 31, 2021, we were in compliance with all affirmative, negative and financial covenants.
See Note 8 to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K for additional information on our financing arrangements.
Cash Flow Comparisons
The following table summarizes our cash flows:
| | | | | | | | | | | |
| Years Ended December 31, |
| (in thousands) | 2021 | | 2020 |
| Total cash provided by (used in): | | | |
| Operating activities | $ | 36,619 | | | $ | 21,320 | |
| Investing activities | (3,852) | | | 8,167 | |
| Financing activities | (19,990) | | | 447 | |
| Effect of exchange rate changes on cash | (1) | | | 2 | |
| Increase (decrease) in cash and cash equivalents | 12,776 | | | 29,936 | |
| Cash and cash equivalents, beginning of period | 53,137 | | | 23,201 | |
| Cash and cash equivalents, end of period | $ | 65,913 | | | $ | 53,137 | |
Operating Activities
Operating cash inflows are generated primarily from net product sales, license and development fees and royalties. Operating cash outflows consist principally of expenditures for manufacturing costs, personnel costs, general and administrative expenses, research and development activities, and sales and marketing costs. Fluctuations in cash from operating activities are primarily a result of the timing of cash receipts and disbursements.
The change in the net cash from operating activities was primarily a result of the increase in our net income, excluding non-cash activity, and changes in operating assets and liabilities due to timing of cash receipts and cash disbursements, principally driven by depletion of inventory and an increase in accrued liabilities, partially offset by an increase in accounts receivable and deferred revenue.
Investing Activities
Net cash used in investing activities for the year ended December 31, 2021 was attributable to the purchase of TLANDO® intangible product rights for $11.3 million, capital expenditures of $6.7 million, an additional NOCDURNA® intangible product rights contractual payment of $2.5 million and investment security purchases of $1.2 million, partially offset by net proceeds of $17.8 from the sale of the OTREXUP® product line. Net cash provided by investing activities for the year ended December 31, 2020 was attributable to $22.5 million proceeds from maturities of short-term investments, partially offset by capital expenditures of $9.6 million primarily for our manufacturing facility and the purchase of NOCDURNA® intangible product rights for $5.0 million.
Financing Activities
Net cash used in financing activities for the year ended December 31, 2021 consisted of $40.0 million in principal payments on the extinguishment of our Term Loan with Hercules Capital, Inc., $2.8 million paid to taxing authorities in connection with net-share settled share-based awards for which we withheld shares equivalent to the value of the employee’s tax obligation for the applicable income and other employment taxes, $2.1 million in prepayment fees and an end of term charge on our Term Loan and $0.3 million in debt issuance costs, partially offset by $20.0 million in proceeds from the issuance of our new Term Loan Facility with Wells Fargo and $5.2 million in proceeds received from exercises of stock options. Net cash provided by financing activities for the year ended December 31, 2020 included $1.8 million in proceeds from the exercise of stock options, partially offset by $1.4 million paid to taxing authorities in connection with net-share settled stock-based awards for which we withheld shares equivalent to the value of the employees’ tax obligation for the applicable income and other employment taxes.
Contractual Obligations
As of December 31, 2021, our contractual obligations are as follows:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Payments Due by Period |
| (in thousands) | Total | | Less than
1 year | | 1 - 3
years | | 3 - 5
years | | More than
5 years |
Long-term debt obligations 1 | $ | 20,000 | | | $ | 1,500 | | | $ | 18,500 | | | $ | — | | | $ | — | |
Interest payable on long-term debt 2 | 1,350 | | | 512 | | | 838 | | | — | | | — | |
Unused revolving line of credit fee 3 | 201 | | | 71 | | | 130 | | | — | | | — | |
Operating lease obligations 4 | 8,012 | | | 1,334 | | | 1,731 | | | 1,354 | | | 3,593 | |
Purchase commitments 5 | 31,312 | | | 31,312 | | | — | | | — | | | — | |
| Total | $ | 60,875 | | | $ | 34,729 | | | $ | 21,199 | | | $ | 1,354 | | | $ | 3,593 | |
1 Long-term debt includes principal installment payments on our Term Loan. Refer to Note 8 to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K for additional information regarding our financing arrangements.
2 Calculated using the variable interest rate as of December 31, 2021 based on LIBOR plus required spread on our Term Loan. Refer to Note 8 to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K for additional information regarding our financing arrangements.
3 Calculated using the commitment fee rate as of December 31, 2021 based on our consolidated total leverage ratio assuming the entire revolving line of credit remains undrawn for the duration of the agreement. Refer to Note 8 to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K for additional information regarding our financing arrangements.
4 Operating leases are primarily for office space, as well as vehicles and equipment. Refer to Note 5 to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K for additional lease information.
5 Purchase commitments include open purchase orders with suppliers and inventory to be purchased in accordance with the TLANDO® exclusive license agreement entered into with Lipocine in October 2021. Refer to Note 6 to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K for additional information on the agreement.
Off Balance Sheet Arrangements
As of December 31, 2021, we did not have any off-balance sheet arrangements, including any arrangements with any structured finance, special purpose or variable interest entities.
Critical Accounting Policies and Use of Estimates
The preceding discussion and analysis of our results of operations and financial condition is based upon our Consolidated Financial Statements. Our Consolidated Financial Statements have been prepared in accordance with U.S. generally accepted accounting principles (“GAAP”), which require us to make estimates and assumptions in certain circumstances that, giving due consideration to materiality, affect the reported amounts of assets and liabilities, revenues and expenses and related disclosures as of the date of the financial statements. We regularly review our estimates and assumptions, which are based on historical experience and various other factors that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from our estimates, and significant variances could materially impact our financial condition and results of operations under different assumptions or conditions.
We believe that of our significant accounting policies, the following are particularly important to the portrayal of our results of operations and financial position and is subject to an inherent degree of uncertainty as it may require the application of a higher level of subjectivity and judgment by us. Our significant accounting policies are fully described in Note 2 to the Consolidated Financial Statements included in Item 8 of Part II of this Annual Report on Form 10-K.
Revenue Recognition (Variable Consideration)
We generate revenue from proprietary and partnered product sales, license and development activities and royalty arrangements. Revenue is recognized when or as we transfer control of the promised goods or services to our customers in an amount that reflects the consideration to which we expect to be entitled to in exchange for those goods or services.
We enter into contracts with partners that often contain multiple elements such as licensing, development, manufacturing and commercialization components. These arrangements are often complex, and we may receive various types of consideration over the life of the arrangement, including: up-front fees, reimbursements for research and development services, milestone payments, payments on product shipments, margin sharing arrangements, license fees and royalties.
In assessing our revenue arrangements, we must identify the contract, determine the transaction price including an estimation of any variable consideration we expect to receive in connection with the contract, identify the promises of goods or services to the customer and each distinct performance obligation, allocate the transaction price to each of the performance obligations, and recognize revenue when or as the performance obligations are satisfied. Each of these steps in the revenue recognition process requires management to make judgements and/or estimates. The most significant judgements and estimates involve the determination of variable consideration to be included in the transaction price, such as the estimation of product returns and sales allowances in connection with the sale of our proprietary products.
Variable consideration is recognized at an amount we believe is not subject to significant reversal and is adjusted at each reporting period if the most likely amount of expected consideration changes or becomes fixed. For example, we must estimate future product returns and sales allowances at the time of sales to distributors. The expected value is determined based on unit sales data, customer purchasing patterns, product expiration dates and levels of inventory in the distribution channel, contractual terms with customers and third-party payers, historical and expected utilization rates, and any new or anticipated changes in programs or regulations. We believe this provides a reasonable basis for recognizing revenue, however, actual results could differ from estimates and significant changes in estimates could impact our results of operations in future periods.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Foreign Currency Exchange Risk
We are exposed to foreign exchange rate fluctuations of the Swiss Franc to the U.S. dollar as the financial position and operating results of our subsidiaries in Switzerland are translated into U.S. dollars for consolidation. Our exposure to foreign exchange rate fluctuations also arises from transferring funds to our Swiss subsidiaries in Swiss Francs. In addition, we have exposure to exchange rate fluctuations between the Euro and the U.S. dollar for some of our transactions. We do not currently use derivative financial instruments to hedge against exchange rate risk. The effect of foreign exchange rate fluctuations on our financial results for the year ended December 31, 2021 was not material. In addition, a hypothetical 10% appreciation or depreciation in foreign currencies against the U.S. dollar, assuming all other variables are held constant, would not have a material impact on our financial position or operating results for the year ended December 31, 2021.
Interest Rate Risk
We are directly exposed to changes in market interest rates on our long-term debt as our secured floating rate credit facility requires interest payments to be calculated at a floating rate tied in part to LIBOR or, if LIBOR is no longer available, at a replacement rate as defined within the Credit Facility Agreement. As a result, changes in the floating interest rate can affect our operating results and liquidity. As of December 31, 2021, we had floating interest rate debt of $20.0 million outstanding carrying a floating interest rate of approximately 2.60% (one-month LIBOR rate plus the applicable margin of 2.50%). A hypothetical increase of 1 percentage point in floating interest rate, assuming principal payments in accordance with the Credit Facility Agreement and all other variables held constant, would result in $0.2 million increase in future annual interest expense.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
ANTARES PHARMA, INC.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors
Antares Pharma, Inc.:
Opinions on the Consolidated Financial Statements and Internal Control Over Financial Reporting
We have audited the accompanying consolidated balance sheets of Antares Pharma, Inc. and subsidiaries (the Company) as of December 31, 2021 and 2020, the related consolidated statements of operations, comprehensive income (loss), stockholders’ equity, and cash flows for each of the years in the three-year period ended December 31, 2021, and the related notes (collectively, the consolidated financial statements). We also have audited the Company’s internal control over financial reporting as of December 31, 2021, based on criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2021 and 2020, and the results of its operations and its cash flows for each of the years in the three-year period ended December 31, 2021, in conformity with U.S. generally accepted accounting principles. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2021 based on criteria established in Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission.
Basis for Opinions
The Company’s management is responsible for these consolidated financial statements, for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Management's Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company’s consolidated financial statements and an opinion on the Company’s internal control over financial reporting based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud, and whether effective internal control over financial reporting was maintained in all material respects.
Our audits of the consolidated financial statements included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the consolidated financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of a critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
Government and insurance plan rebate reserves
As discussed in Note 2 to the consolidated financial statements, the Company records estimated reserves for rebates and chargebacks, which includes government and insurance plan rebate reserves. As of December 31, 2021, the reserve for rebates and chargebacks was $13,301 thousand. The estimated reserves for government and insurance plan rebates are recorded as a reduction to product revenue in the same period that the related revenue is recognized and the reserves are included within current liabilities in the consolidated balance sheets. The government and insurance plan rebate reserves are estimated based on unit sales data, contractual terms with third-party payers, historical and estimated future percentages of rebates incurred on sales, historical and estimated future insurance plan billings, any new or anticipated changes in programs or regulations that would impact the amount of the actual rebates to be paid, and levels of inventory in the distribution channel.
We identified the evaluation of certain government and insurance plan rebate reserves as a critical audit matter. Subjective and challenging auditor judgment was required to evaluate the estimated future percentages of rebates incurred on sales for government plan rebates and the estimated future insurance plan billings for insurance plan rebates due to the unpredictability of those future amounts and the length of time between when the sale occurred and when the rebates are paid to the administrator of the programs.
The following are the primary procedures we performed to address this critical audit matter. We evaluated the design and tested the operating effectiveness of certain internal controls related to the Company’s government and insurance plan rebate reserves processes. This included controls related to management’s process to develop the estimated future percentages of rebates incurred on sales and the estimated future insurance plan billings. We tested the historical rebates incurred on sales and insurance plan billings, which are used in the determination of estimated future percentages of rebates incurred on sales and insurance plan billings respectively, by comparing samples of the historical rebates incurred on sales and insurance plan billings to underlying invoices and evidence of the cash disbursement. For both government and insurance plan rebate reserves, we assessed management’s estimate by evaluating the consistency of the inputs with the trend of actual historical percentages of rebates incurred on sales and insurance plan billings. For the insurance plan rebate reserve, we evaluated management’s estimate by comparing insurance plan billings received after period end to the estimated insurance plan billings recorded at year-end. For the government plan rebate reserve, we performed sensitivity analyses over the estimated future percentages of rebates incurred on sales using historical information to assess the impact of changes in those assumptions on management’s estimate. We evaluated the Company’s ability to estimate government and insurance plan rebate reserves accurately by comparing actual amounts paid for the related rebates to historical estimates.
/s/ KPMG LLP
We have served as the Company’s auditor since 1995.
Minneapolis, Minnesota
March 3, 2022
ANTARES PHARMA, INC.
CONSOLIDATED BALANCE SHEETS
(in thousands, except per share amounts)
| | | | | | | | | | | |
| December 31, 2021 | | December 31, 2020 |
| Assets | | | |
| Current assets | | | |
| Cash and cash equivalents | $ | 65,913 | | | $ | 53,137 | |
| Short-term investments | 1,245 | | | — | |
| Accounts receivable, net | 56,697 | | | 42,221 | |
| Other receivables | 26,311 | | | — | |
| Inventories, net | 11,544 | | | 18,216 | |
| Contract assets | 8,030 | | | 8,140 | |
| Prepaid expenses and other current assets | 4,532 | | | 4,877 | |
| Total current assets | 174,272 | | | 126,591 | |
| Deferred tax assets, net | 33,043 | | | 46,982 | |
| Property and equipment, net | 26,015 | | | 24,020 | |
| Operating lease right-of-use assets | 3,774 | | | 4,621 | |
| Intangibles, net | 17,879 | | | 7,693 | |
| Goodwill | 1,095 | | | 1,095 | |
| Other long-term assets | 1,427 | | | 1,529 | |
| Total assets | $ | 257,505 | | | $ | 212,531 | |
| Liabilities and Stockholders’ Equity | | | |
| Current liabilities | | | |
| Accounts payable | $ | 17,056 | | | $ | 16,194 | |
| Accrued expenses and other liabilities | 35,043 | | | 25,635 | |
| Current maturities of long-term debt, net | 1,500 | | | 16,230 | |
| Operating lease liabilities, current | 904 | | | 1,203 | |
| Deferred revenue | 4,427 | | | 3,943 | |
| Total current liabilities | 58,930 | | | 63,205 | |
| Long-term debt, less current maturities | 18,241 | | | 24,669 | |
| Operating lease liabilities, long-term | 4,576 | | | 4,816 | |
| Other long-term liabilities | — | | | 726 | |
| Total liabilities | 81,747 | | | 93,416 | |
| Commitments and contingencies (Note 16) | | | |
| Stockholders’ Equity | | | |
Preferred Stock: $0.01 par; 3,000 shares authorized, none outstanding | — | | | — | |
Common Stock: $0.01 par; 300,000 shares authorized; 170,072 and 166,836 issued and outstanding as of December 31, 2021 and December 31, 2020, respectively | 1,701 | | | 1,668 | |
| Additional paid-in capital | 351,079 | | | 340,756 | |
| Accumulated deficit | (176,337) | | | (222,626) | |
| Accumulated other comprehensive loss | (685) | | | (683) | |
Total stockholders’ equity | 175,758 | | | 119,115 | |
| Total liabilities and stockholders’ equity | $ | 257,505 | | | $ | 212,531 | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these consolidated financial statements.
ANTARES PHARMA, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(in thousands, except per share amounts)
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Revenue | | | | | |
| Product sales, net | $ | 126,667 | | | $ | 113,834 | | | $ | 92,103 | |
| Licensing and development revenue | 19,623 | | | 14,466 | | | 7,529 | |
| Royalties | 37,692 | | | 21,299 | | | 24,232 | |
| Total revenue, net | 183,982 | | | 149,599 | | | 123,864 | |
| Operating expenses | | | | | |
| Cost of product sales | 54,418 | | | 53,960 | | | 46,267 | |
| Cost of development revenue | 13,863 | | | 9,140 | | | 4,208 | |
| Research and development | 14,502 | | | 10,121 | | | 10,624 | |
| Selling, general and administrative | 73,857 | | | 62,759 | | | 61,773 | |
| Total operating expenses | 156,640 | | | 135,980 | | | 122,872 | |
| Gain on sale of assets | 38,591 | | | — | | | — | |
| Operating income | 65,933 | | | 13,619 | | | 992 | |
| Other income (expense) | | | | | |
| Interest expense | (3,611) | | | (3,787) | | | (3,549) | |
| Other income (expense), net | (51) | | | 89 | | | 530 | |
| Total other expense, net | (3,662) | | | (3,698) | | | (3,019) | |
| Income (loss) before income taxes | 62,271 | | | 9,921 | | | (2,027) | |
| Income tax provision (benefit) | 15,982 | | | (46,280) | | | — | |
| Net income (loss) | $ | 46,289 | | | $ | 56,201 | | | $ | (2,027) | |
| | | | | |
| Earnings (loss) per common share | | | | | |
| Basic | $ | 0.27 | | | $ | 0.34 | | | $ | (0.01) | |
| Diluted | $ | 0.26 | | | $ | 0.33 | | | $ | (0.01) | |
| | | | | |
| Weighted average common shares outstanding | | | | | |
| Basic | 169,226 | | | 166,066 | | | 162,574 | |
| Diluted | 174,733 | | | 170,155 | | | 162,574 | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these Consolidated Financial Statements.
ANTARES PHARMA, INC.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE INCOME (LOSS)
(in thousands)
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Net income (loss) | $ | 46,289 | | | $ | 56,201 | | | $ | (2,027) | |
| Foreign currency translation adjustment | (2) | | | 19 | | | 1 | |
| Comprehensive income (loss) | $ | 46,287 | | | $ | 56,220 | | | $ | (2,026) | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these Consolidated Financial Statements.
ANTARES PHARMA, INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(in thousands)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Common Stock | | | | | | | | |
| Number
of
Shares | | Amount | | Additional
Paid-In
Capital | | Accumulated
Deficit | | Accumulated
Other
Comprehensive
Income (Loss) | | Total
Stockholders’
Equity |
| Balance, December 31, 2018 | 159,721 | | | $ | 1,597 | | | $ | 314,907 | | | $ | (276,800) | | | $ | (703) | | | $ | 39,001 | |
| Issuance of common stock | 2,307 | | | 23 | | | 7,758 | | | — | | | — | | | 7,781 | |
| Common stock issued under equity compensation plan, net of shares withheld for taxes | 664 | | | 7 | | | (1,138) | | | — | | | — | | | (1,131) | |
| Exercise of options | 2,529 | | | 25 | | | 4,380 | | | — | | | — | | | 4,405 | |
| Share-based compensation | — | | | — | | | 6,470 | | | — | | | — | | | 6,470 | |
| Net loss | — | | | — | | | — | | | (2,027) | | | — | | | (2,027) | |
| Other comprehensive income | — | | | — | | | — | | | — | | 1 | | | 1 | |
| Balance, December 31, 2019 | 165,221 | | | 1,652 | | | 332,377 | | | (278,827) | | | (702) | | | 54,500 | |
| Common stock issued under equity compensation plan, net of shares withheld for taxes | 676 | | | 7 | | | (1,374) | | | — | | | — | | | (1,367) | |
| Exercise of options | 939 | | | 9 | | | 1,805 | | | — | | | — | | | 1,814 | |
| Share-based compensation | — | | | — | | | 7,948 | | | — | | | — | | | 7,948 | |
| Net income | — | | | — | | | — | | | 56,201 | | | — | | | 56,201 | |
| Other comprehensive income | — | | | — | | | — | | | — | | | 19 | | | 19 | |
| Balance, December 31, 2020 | 166,836 | | | 1,668 | | | 340,756 | | | (222,626) | | | (683) | | | 119,115 | |
| Common stock issued under equity compensation plan, net of shares withheld for taxes | 942 | | | 10 | | | (2,851) | | | — | | | — | | | (2,841) | |
| Exercise of options | 2,294 | | | 23 | | | 5,159 | | | — | | | — | | | 5,182 | |
| Share-based compensation | — | | | — | | | 8,015 | | | — | | | — | | | 8,015 | |
| Net income | — | | | — | | | — | | | 46,289 | | | — | | | 46,289 | |
| Other comprehensive loss | — | | | — | | | — | | | — | | | (2) | | | (2) | |
| Balance, December 31, 2021 | 170,072 | | | $ | 1,701 | | | $ | 351,079 | | | $ | (176,337) | | | $ | (685) | | | $ | 175,758 | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these Consolidated Financial Statements.
ANTARES PHARMA, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Cash Flows from Operating Activities | | | | | |
| Net income (loss) | $ | 46,289 | | | $ | 56,201 | | | $ | (2,027) | |
| Adjustments to reconcile net income (loss) to net cash provided by (used in) operating activities: | | | | | |
| Deferred taxes | 13,939 | | | (47,203) | | | (371) | |
| Stock-based compensation | 8,015 | | | 7,948 | | | 6,470 | |
| Depreciation and amortization | 3,901 | | | 2,627 | | | 2,557 | |
| Non-cash interest expense | 648 | | | 504 | | | 405 | |
| Increase in inventory reserve | 152 | | | 511 | | | 325 | |
| Gain on sale of assets | (38,591) | | | — | | | — | |
| Other | 671 | | | 42 | | | 12 | |
| Changes in operating assets and liabilities: | | | | | |
| Accounts receivable | (14,476) | | | (7,128) | | | (16,095) | |
| Inventories, net | 2,748 | | | (2,728) | | | (4,975) | |
| Contract assets | 110 | | | 53 | | | (2,793) | |
| Prepaid expenses and other current assets | (1,056) | | | (1,460) | | | (655) | |
| Accounts payable | 2,189 | | | 2,594 | | | 926 | |
| Accrued expenses and other liabilities | 11,596 | | | 7,157 | | | 4,888 | |
| Deferred revenue | 484 | | | 2,202 | | | 718 | |
| Net cash provided by (used in) operating activities | 36,619 | | | 21,320 | | | (10,615) | |
| Cash Flows from Investing Activities | | | | | |
| Purchases of property and equipment | (6,617) | | | (9,615) | | | (2,350) | |
| Proceeds from sale of assets, net of transaction costs | 17,825 | | | 282 | | | 5,000 | |
| Purchase of intangibles, including transaction costs | (13,815) | | | (5,000) | | | — | |
| Purchases of investment securities | (1,245) | | | — | | | (22,645) | |
| Proceeds from maturities of investment securities | — | | | 22,500 | | | — | |
| Net cash provided by (used in) investing activities | (3,852) | | | 8,167 | | | (19,995) | |
| Cash Flows from Financing Activities | | | | | |
| Proceeds from issuance of long-term debt | 20,000 | | | — | | | 15,000 | |
| Principal payments of long-term debt | (40,000) | | | — | | | — | |
| Prepayment fees and end of term charge on long-term debt | (2,055) | | | — | | | — | |
| Payment of debt issuance costs for long-term debt | (276) | | | — | | | (136) | |
| Proceeds from issuance of common stock, net | — | | | — | | | 7,781 | |
| Proceeds from exercise of stock options | 5,182 | | | 1,814 | | | 4,405 | |
| Taxes paid related to net share settlement of equity awards | (2,841) | | | (1,367) | | | (1,131) | |
| Net cash provided by (used in) financing activities | (19,990) | | | 447 | | | 25,919 | |
| Effect of exchange rate changes on cash and cash equivalents | (1) | | | 2 | | | — | |
| Cash and cash equivalents | | | | | |
| Net increase (decrease) during the period | 12,776 | | | 29,936 | | | (4,691) | |
| Beginning of period | 53,137 | | | 23,201 | | | 27,892 | |
| End of period | $ | 65,913 | | | $ | 53,137 | | | $ | 23,201 | |
| | | | | |
| Supplemental disclosure of cash flow information | | | | | |
| Cash paid for interest | $ | 2,736 | | | $ | 3,538 | | | $ | 3,025 | |
| Cash paid for income taxes | $ | 1,783 | | | $ | 90 | | | $ | — | |
| Supplemental disclosure of non-cash investing activities | | | | | |
Purchases of property and equipment recorded in accounts payable and accrued expenses | $ | 259 | | | $ | 2,017 | | | $ | 970 | |
| Purchase of intangible assets included in accrued liabilities | $ | — | | | $ | 2,500 | | | $ | — | |
| Gain on sale of assets recognized in excess of cash received | $ | 20,766 | | | $ | — | | | $ | — | |
The accompanying Notes to Consolidated Financial Statements are an integral part of these Consolidated Financial Statements.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Note 1. Description of Business
Antares Pharma, Inc. (“Antares,” “we,” “our,” “us” or the “Company”) is a specialty pharmaceutical company focused primarily on the development and commercialization of pharmaceutical products and technologies in targeted therapeutic areas. We develop, manufacture and commercialize, for ourselves or with partners, novel therapeutic products using our advanced drug delivery systems that are designed to provide commercial or functional advantages, such as improved safety and efficacy, convenience, improved tolerability, and enhanced patient comfort and adherence. We also seek product opportunities that complement and leverage our commercial platform. We have a portfolio of proprietary and partnered commercial products and ongoing product development programs in various stages of development. We have formed partnership arrangements with several different industry leading pharmaceutical companies.
Our marketed proprietary products include:
•XYOSTED® (testosterone enanthate) injection, indicated for testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of endogenous testosterone, and is the first and only subcutaneous testosterone enanthate product for once-weekly, at-home self-administration to be approved by the U.S. Food and Drug Administration (“FDA”);
•OTREXUP® (methotrexate) injection, indicated for adults with severe active rheumatoid arthritis, children with active polyarticular juvenile idiopathic arthritis and adults with severe recalcitrant psoriasis, which was sold to Otter Pharmaceuticals, LLC (a subsidiary of Assertio Holdings, Inc., together with Assertio Holdings, Inc., as guarantor, individually and collectively referred to as “Otter”) in December 2021 as discussed in Note 12; and
•NOCDURNA® (desmopressin acetate), marketed in the U.S. for the treatment of nocturia due to nocturnal polyuria (“NP”) in adults who awaken at least two times per night to urinate.
We are also party to various partnered product development and supply arrangements:
•We developed and are the exclusive supplier of devices for Teva Pharmaceutical Industries, Ltd.’ (“Teva”) Epinephrine Injection USP products, the generic equivalent of EpiPen® and EpiPen® Jr., indicated for emergency treatment of severe allergic reactions including those that are life threatening (anaphylaxis) in adults and certain pediatric patients;
•Through our commercialization partner Teva, we sell Sumatriptan Injection USP, a generic equivalent to the Imitrex® STATdose Pen®, in the U.S. indicated for the acute treatment of migraine headaches and cluster headache in adults;
•In collaboration with AMAG Pharmaceuticals, Inc. (“AMAG”), acquired by Covis Group S.a.r.l. (“CG”) (collectively CG and AMAG are herein after referred to as “Covis”) in November 2020, we developed a subcutaneous auto injector and are the exclusive supplier of devices and the final assembled and packaged commercial product of Makena® (hydroxyprogesterone caproate injection) subcutaneous auto injector, which is a ready-to-administer treatment indicated to reduce the risk of preterm birth in women pregnant with one baby and who spontaneously delivered at least one preterm baby in the past.
•We developed and are the exclusive supplier of devices for Teva’s generic equivalent of Forsteo® (Teriparatide Injection) which is approved and currently sold by Teva in various countries outside the U.S.
Additionally, we are developing other devices in collaboration with various pharmaceutical partners and advancing other internal research and development programs.
We also have a proprietary product, TLANDO® (testosterone undecanoate) is a twice-day oral formulation of testosterone for TRT indicated for conditions associated with a deficiency or absence of endogenous testosterone, or hypogonadism in adult males, with tentative FDA approval.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Note 2. Summary of Significant Accounting Policies
Basis of Presentation
The accompanying consolidated financial statements include the accounts of Antares Pharma, Inc. and its two wholly-owned foreign subsidiaries. All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements in accordance with U.S. generally accepted accounting principles (“GAAP”) requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities, disclosure of contingent assets and liabilities as of the date of the financial statements and reported amounts of revenues and expenses during the reporting period. Our most significant accounting estimates relate to revenue recognition and variable consideration, inventory valuation, the carrying value of deferred tax assets and the valuation of equity instruments used in the computation of share-based compensation. Actual results could differ from these estimates, and significant variances could materially impact our financial condition and results of operations.
Reclassifications
Certain reclassifications have been made to prior year amounts to conform with the current year presentation. As of and for the year ended December 31, 2020, the cost of product sales and the cost of development revenue were classified under the heading Operating expenses in the Consolidated Statements of Operations, and the corresponding prior period amount was reclassified to conform to this presentation. The reclassifications had no impact on our operating income (loss), net income (loss) or cash flows as previously reported.
Accounting Pronouncements Recently Adopted
We adopted FASB ASU No. 2018-15, Customers’ Accounting for Implementation Costs Incurred in Cloud Computing Arrangement that is a Service Contract, effective January 1, 2020, which provides new guidance on a customer’s accounting for implementation, set-up, and other upfront costs incurred in a cloud computing arrangement that is hosted by the vendor (i.e., a service contract). Under the new guidance, entities apply the same criteria for capitalizing implementation costs as they would for an arrangement that has a software license. Adoption of this standard did not have a material impact on our financial condition, results of operations or disclosures.
We adopted FASB ASU No. 2018-18, Clarifying the Interaction Between Topic 808 and 606, effective January 1, 2020, which clarifies that certain transactions between collaborative arrangement participants should be accounted for under the revenue guidance, adds unit of account guidance to the collaborative arrangement guidance to align with the revenue standard, and clarifies presentation guidance for transactions with a collaborative arrangement participant that is not accounted for under the revenue standard. Adoption of this standard did not have a material impact on our financial condition, results of operations or disclosures.
Recently Issued Accounting Pronouncements Not Yet Adopted as December 31, 2021
In June 2016, the FASB issued ASU 2016-13, Financial Instruments—Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments, followed by related amendments, which changes the accounting for credit losses on instruments measured at amortized cost by adding an impairment model that is based on expected losses rather than incurred losses. Any entity will recognize as an allowance its estimate of expected credit losses, which is believed to result in more timely recognition of such losses as the standard eliminates the probable initial recognition threshold. The new guidance is required to be adopted using a modified retrospective approach with a cumulative-effect adjustment to retained earnings as of the beginning of the first reporting period of adoption. Adoption of the new guidance was originally required for annual periods beginning after December 15, 2019, including interim periods within the annual period.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
In October 2019, the FASB issued ASU 2019-10, Financial Instruments—Credit Losses (Topic 326), Derivatives and Hedging (Topic 815), and Leases (Topic 842): Effective Dates, which deferred the effective date of ASU 2016-13 for certain entities, including those that are eligible for smaller reporting company classification. Determination of eligibility for deferral was a one-time assessment as of November 15, 2019 based on the entity’s most recent smaller reporting company eligibility determination as of the last business day of its most recently completed second quarter. Based on this determination, we qualified as a smaller reporting entity and was therefore eligible for the adoption deferral resulting in a new effective date of January 1, 2023. The impact on our financial condition, results of operations and disclosures is being evaluated but is not expected to be significant as we have historically had minimal credit losses on financial instruments.
In April 2020, the FASB issued ASU No. 2020-04, Facilitation of the Effects of Reference Rate Reform on Financial Reporting, which provides relief for companies preparing for discontinuation of interest rates such as LIBOR. The standard can be applied immediately through December 31, 2022. We have not yet evaluated the impact the adoption of this guidance may have on our financial condition, results of operations or disclosures.
Foreign Currency Translation
The majority of our foreign subsidiaries’ revenues are denominated in U.S. dollars, and any required funding of the subsidiaries is provided by the U.S. parent. Nearly all operating expenses of our foreign subsidiaries are denominated in Swiss Francs. Additionally, bank accounts held by foreign subsidiaries are denominated in Swiss Francs, there is a low volume of intercompany transactions and there is not an extensive interrelationship between the operations of the subsidiaries and the parent company. As such, we have determined that the Swiss Franc is the functional currency for our foreign subsidiaries. Our reporting currency is the United States Dollar (“USD”). The financial statements of our foreign subsidiaries are translated into USD for consolidation purposes. All assets and liabilities are translated using period-end exchange rates. Statements of operations items are translated using average exchange rates for the period. The resulting translation adjustments are recorded as a separate component of stockholders’ equity, comprising all of the accumulated other comprehensive income (loss). Sales to certain customers and purchases from certain vendors by the U.S. parent are in currencies other than USD and are subject to foreign currency exchange rate fluctuations. Foreign currency transaction gains and losses are included in other income (expense) in the Consolidated Statements of Operations.
Cash and Cash Equivalents
Cash and cash equivalents represent demand deposits at commercial banks and highly liquid investments with an original maturity of three months or less. Cash equivalents, consisting of investments in money market funds and bank certificate of deposits, are remeasured and reported at fair value each reporting period based on quoted market prices, which is a Level 1 input within the three-level valuation hierarchy for disclosure of fair value measurements, and totaled $26,889 and $36,133 as of December 31, 2021 and 2020, respectively.
Investments
From time to time, we also invest in bank certificates of deposit that are classified as held-to-maturity because of our intent and ability to hold securities to maturity. Investments with original maturities greater than three months but less than one year are classified as short-term investments on the Consolidated Balance Sheets. The investment securities are carried at their amortized cost and fair value is determined by quoted market prices for identical or similar securities. The carrying value of our short-term investments as of December 31, 2021 approximate fair value.
Fair Value Measurements
Financial assets and liabilities are required to be measured and reported at fair value each reporting period. Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value must maximize the use of observable inputs and minimize the use of unobservable inputs. When considering market participant assumptions in fair value measurement, the following fair value hierarchy distinguishes between observable and unobservable inputs, which are categorized in one of the following levels.
•Level 1: Unadjusted quoted prices which are available in active markets for identical assets or liabilities accessible to us at the measurement date.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
•Level 2: Inputs other than Level 1 that are observable, either directly or indirectly, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities.
•Level 3: Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
The hierarchy gives the highest priority to Level 1, as this level provides the most reliable measure of fair value, while given the lowest priority to Level 3.
Financial assets and liabilities that are not measured at fair value on a recurring basis include held-to-maturity investments and long-term debt as the carrying values of which approximate fair value. The estimated fair value of debt is based on Level 2 inputs, including our understanding of current market rates we could obtain for similar loans. The fair value of our cash and cash equivalents, accounts receivable, other receivables, contract assets, accounts payable and accrued liabilities approximate fair value due to their short-term nature.
We measure certain financial instruments at fair value on a nonrecurring basis. These assets primarily include goodwill and intangible assets, as well as property and equipment and right-of-use lease assets. These assets were initially measured and recognized at amounts equal to the fair value determined as of the date of acquisition or purchase. Periodically, these assets are tested for impairment, by comparing their respective carrying values to the estimated fair value of the reporting unit or asset group in which they reside. In the event any of these assets were to become impaired, we would recognize an impairment loss equal to the amount by which the carrying value of the reporting unit, impaired asset or asset group exceeds its estimated fair value. Fair value measurement of the reporting unit associated with our goodwill balance is estimated at least annually in the fourth quarter of each calendar year for purposes of impairment testing if a quantitative analysis is performed. Fair value measurements associated with our intangible assets, other long-lived assets and property and equipment are estimated when events or changes in circumstances such as market value, asset utilization, physical change, legal factors or other matters indicate that the carrying value may not be recoverable.
Accounts Receivable
Trade accounts receivable represents amounts billed to customers and are stated at the amount we expect to collect. Customer creditworthiness, past transaction history with the customer and changes in customer payment terms are factors considered when determining collectability of specific customer accounts. As of December 31, 2021, our trade accounts receivable balance was due primarily from Teva and major wholesale distributors. Each of these customers have historically paid in a timely manner and demonstrated creditworthiness. Accordingly, we believe the risk of accounts being uncollectible is minimal and no significant allowances for doubtful accounts was established as of December 31, 2021 or 2020. If the financial condition of our customers were to deteriorate, adversely affecting their ability to make payments, additional allowances may be required. We had no material write-offs to bad debt expense in the years ended December 31, 2021, 2020 or 2019.
Royalties receivable from partners are included in accounts receivable and are typically payable to us within 45 to 60 days after the end of each quarter in which they were earned.
Inventories
Inventories are stated at the lower of cost or net realizable value with cost determined on a first-in, first-out basis. Certain components of our products are provided by a limited number of vendors, and our production, assembly, warehousing and distribution operations are outsourced to third-party suppliers where substantially all of our inventory is located. Disruption of supply from key vendors or third-party suppliers may have a material adverse impact on our operations and financial results.
We record reserves for potentially excess, dated or obsolete inventories based on forecasted product demand estimates and the likelihood of consumption in the normal course of business, considering the expiration dates of the inventories on hand, planned production volumes and lead times required for restocking of customer inventories. Although every effort is made to ensure that forecasts and assessments are reasonable, changes to these assumptions are possible. In such cases, estimates may prove inaccurate and result in an understatement or overstatement of the reserves required to fairly state such inventories.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Contract Assets
Contract assets are recognized when control of goods or services has transferred to the customer, and corresponding revenue is recognized on an over time basis but is not yet billable to the customer in accordance with the terms of the contract. Costs that have been incurred in connection with development services provided to partners for which the associated revenue has not yet been recognized are also recorded as contract assets and totaled $564 and $1,685 as of December 31, 2021 and 2020, respectively.
Property and Equipment
Property and equipment are stated at cost less accumulated depreciation. Depreciation is computed using the straight-line method over an asset’s estimated useful life as follows:
| | | | | |
| Useful Life |
| Computer equipment and software | 3-5 years |
| Furniture, fixtures and office equipment | 5-7 years |
| Production molds, tooling and equipment | 3-10 years |
| Leasehold improvements | Lesser of useful life or lease term |
Expenditures, including interest costs, for assets under construction and internal-use software that are not yet ready for their intended use are capitalized and will be depreciated based on the above guidelines when placed in service. Costs associated with repairs and maintenance activities are expensed as incurred.
Leases
We recognize right-of-use (“ROU”) assets and lease liabilities when we obtain the right to control the asset under a leasing arrangement with an initial term greater than twelve months. We evaluate the nature of each lease at the inception of an arrangement to determine whether it is an operating or financing lease and recognize the ROU asset and lease liability based on the present value of future minimum lease payments over the expected lease term. Our leases do not generally contain an implicit interest rate; therefore, we use the incremental borrowing rate we would expect to pay to borrow on a similar collateralized basis over a similar term in order to determine the present value of our lease payments. The incremental borrowing rate is used in determining the present value of lease payments, unless an implicit rate is specified. Certain lease arrangements contain renewal options that have not been included in the determination of the lease term, as they are not reasonably certain of exercise. For contracts that contain lease and non-lease components, we account for both components as a single lease component. Variable lease payments are expensed as incurred.
Intangible Assets
We capitalize and include the costs of acquired product licenses and trademark rights as intangible assets. These intangible assets with finite useful lives are presented net of accumulated amortization. Amortization is computed on a straight-line basis over the shorter of the contractual or estimated economic life of the underlying contract, which generally ranges from five to ten years.
Impairment of Long-Lived Assets and Intangible Assets
Long-lived assets and intangible assets are reviewed for impairment whenever events or changes in circumstances such as market value, asset utilization, physical change, legal factors or other matters indicate that the carrying value of an asset or asset group may not be recoverable. The impairment test is based on a comparison of the undiscounted cash flows expected to be generated from the use of the asset or asset group and its eventual disposition to the carrying value of the asset. If impairment is indicated, the asset is written down by the amount by which the carrying value of the asset exceeds the related fair value of the asset with the related impairment charge recognized within the Consolidated Statement of Operations. The determination of an asset’s fair value requires management to make certain estimates and judgements.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Goodwill
Goodwill is evaluated for impairment annually as of December 31, or more frequently if an event occurs or circumstances change such as market value, asset utilization, legal factors or other matters that indicate the carrying value may not be recoverable. Evaluating goodwill for impairment involves the determination of the fair value of each reporting unit in which goodwill is recorded using a qualitative or quantitative analysis. A reporting unit is an operating segment or a component of an operating segment for which discrete financial information is available and reviewed by management on a regular basis.
As of December 31, 2021 and 2020, we have goodwill with a carrying value of $1,095, attributable to our single reporting unit. Based on the results of our qualitative analysis, we determined that goodwill was not impaired, and no impairment loss was recognized in the years ended December 31, 2021, 2020, and 2019, respectively.
Revenue Recognition
We generate revenue from proprietary and partnered product sales, license and development activities and royalty arrangements. Revenue is recognized when or as we transfer control of the promised goods or services to the customer at the transaction price, which is the amount that reflects the consideration to which we expect to be entitled to in exchange for those goods or services.
At inception of each contract, we identify the goods and services that have been promised to the customer and each of those that represent a distinct performance obligation, determine the transaction price including any variable consideration, allocate the transaction price to the distinct performance obligations and determine whether control transfers to the customer at a point in time or over time. Variable consideration is included in the transaction price to the extent that it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is subsequently resolved. We reassess our reserves for variable consideration at each reporting date and make adjustments, if necessary, which may affect revenue and earnings in periods in which any such changes become known.
We have elected to recognize the cost for freight and shipping activities as a fulfillment cost. Amounts billed to customers for shipping and handling are included as part of the transaction price and recognized as revenue when control of underlying goods are transferred to the customer. The related shipping and freight charges incurred are included in cost of product sales in the Consolidated Statements of Operations.
Proprietary Product Sales
We sell our proprietary commercial products primarily to wholesale and specialty distributors. Revenue is recognized when control has transferred to the customer, which is typically upon delivery, at the net selling price, which reflects the variable consideration for which reserves and sales allowances are established for estimated returns, wholesale distribution fees, prompt payment discounts, government rebates and chargebacks, plan rebate arrangements and patient discount and support programs.
The determination of certain reserves and sales allowances requires us to make a number of judgements and estimates to reflect our best estimate of the transaction price and the amount of consideration to which we believe we would be ultimately entitled to receive. The expected value is determined based on unit sales data, contractual terms with customers and third-party payers, historical and estimated future percentage of rebates incurred on sales, historical and future insurance plan billings, any new or anticipated changes in programs or regulations that would impact the amount of the actual rebates, customer purchasing patterns, product expiration dates and levels of inventory in the distribution channel. Reserves for prompt payment discounts are recorded as a reduction in accounts receivable in the Consolidated Balance Sheets. Reserves for returns, distributor fees, rebates and customer co-pay support programs are included within current liabilities in the Consolidated Balance Sheets.
Wholesaler Distribution Fees – Distribution fees are paid to certain wholesalers based on contractually determined rates and units purchased. Since the fee paid to the customer is not for a distinct good or service, the consideration is recognized as a reduction of the transaction price of the goods delivered. We accrue the estimated fee due at the time of sale based on the contracted price and adjust the accrual at each reporting period, if necessary, to reflect actual experience.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Prompt Pay Discounts – We offer cash discounts to our customers, generally 2% of the sales price, as an incentive for prompt payment. Based on historical experience, customers take advantage of this discount and accordingly we accrue 100% of the cash discounts offered by reducing accounts receivable and recognizing the discount as a reduction of revenue in the same period the related sales are made. The accrual is reviewed at each reporting period and adjusted if actual experience differs from estimates.
Chargebacks – We provide discounts primarily to authorized users of the Federal Supply Schedule (“FSS”) of the General Services Administration under an FSS contract negotiated by the Department of Veterans Affairs and various organizations under Medicaid contracts and regulations. These entities purchase products from the wholesale distributors at a discounted price, and the wholesale distributors then charge us back the difference between the current wholesale acquisition cost and the price the entity paid for the product. We estimate and accrue chargebacks based on estimated wholesaler inventory levels, current contract prices and historical chargeback activity. Chargebacks are recognized as a reduction of revenue in the same period the related revenue is recognized.
Rebates – We participate in certain government and insurance plan rebate programs, which provide discounted prescriptions to qualified insured patients. Under these rebate programs, we pay a rebate to the third-party administrators of the programs. The rebate payments are generally made in periods subsequent to the quarter in which prescriptions subject to the rebate are filled, generally on a two- to three-month lag for insurance plan rebates and three- to six-month lag for government plan rebates. We estimate and accrue for these rebates based on unit sales data, contractual terms with third-party payers, historical and estimated future percentage of rebates incurred on sales, historical and future insurance plan billings, any new or anticipated changes in programs or regulations that would impact the amount of the actual rebates to be paid, and levels of inventory in the distribution channel. Rebates are recognized as a reduction of revenue in the same period the related revenue is recognized.
Patient Discount Programs – We offer discount cards, co-pay coupons and free trial programs to off-set the cost of prescriptions to patients. We estimate the total amount that will be redeemed or used based on historical redemption experience and on levels of inventory in the distribution and retail channels, and recognize the discount as a reduction of revenue in the same period the related revenue is recognized.
Product Returns – Consistent with industry practice, we generally offer wholesalers and specialty distributors a limited right to return products, generally within six months prior to and 12 months following the product’s expiration date. Our proprietary products generally have expiration dates ranging from 24 to 33 months. Product returns are estimated and recorded at the time of sale based on historical return patterns. Actual returns are tracked by individual production lots and charged against reserves. Returns reserves may be adjusted, if necessary, if actual returns differ from historical estimates. We also monitor and take into consideration the amount of estimated product inventory in the distribution channel, product dating and any known or expected changes in the marketplace when establishing the estimated rate of returns.
Changes in reserves for product returns and sales allowances are as follows:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Rebates and
Chargebacks | | Patient
Discount
Programs | | Returns | | Wholesaler
Distribution
Fees | | Prompt
Payment
Discounts |
| Balance as of December 31, 2019 | $ | 6,308 | | | $ | 845 | | | $ | 370 | | | $ | 1,683 | | | $ | 320 | |
| Accruals and adjustments | 34,947 | | | 12,422 | | | 2,657 | | | 11,619 | | | 2,494 | |
| Payments and other reserve reductions | (34,068) | | | (11,975) | | | (2,569) | | | (10,804) | | | (2,378) | |
| Balance as of December 31, 2020 | 7,187 | | | 1,292 | | | 458 | | | 2,498 | | | 436 | |
| Accruals and adjustments | 52,243 | | | 15,629 | | | 4,163 | | | 15,683 | | | 3,423 | |
| Payments and other reserve reductions | (46,129) | | | (13,971) | | | (3,992) | | | (14,498) | | | (3,222) | |
| Balance as of December 31, 2021 | $ | 13,301 | | | $ | 2,950 | | | $ | 629 | | | $ | 3,683 | | | $ | 637 | |
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Partnered Product Sales
We are party to several license, development, supply and distribution arrangements with pharmaceutical partners, under which we produce and are the exclusive supplier of certain products, devices and/or components. Revenue is recognized when or as control of the goods transfers to the customer as discussed below.
We are the exclusive supplier of the Makena® subcutaneous auto injector product to Covis and beginning in December 2021, OTREXUP® to Otter. Because these products are custom manufactured for each customer with no alternative use and we have a contractual right to payment for performance completed to date, control is continuously transferred to the customer as the product is produced pursuant to firm purchase orders. Revenue is recognized over time using the output method based on the contractual selling price and number of units produced. The amount of revenue recognized in excess of the amount shipped/billed to the customer, if any, is recorded as contract assets in the Consolidated Balance Sheets due to the short-term nature in which the amount is ultimately expected to be billed and collected from the customer.
All other partnered product sales are recognized at the point in time in which control is transferred to the customer, which is typically upon shipment. Sales terms and pricing are governed by the respective supply and distribution agreements, and there is generally no right of return. Revenue is recognized at the transaction price, which includes the contractual per unit selling price and estimated variable consideration, such as volume-based pricing arrangements or profit-sharing arrangements, if any. We recognize revenue, including the estimated variable consideration we expect to receive for contract margin on future commercial sales, upon shipment of the goods to our partner. The estimated variable consideration is recognized at an amount we believe is not subject to significant reversal based on historical experience and is adjusted at each reporting period if the most likely amount of expected consideration changes or becomes fixed.
Licensing and Development Revenue
We have entered into several license, development and supply arrangements with pharmaceutical partners under which we grant a license to our device technology and know-how and provide research and development services that often involve multiple performance obligations and highly customized deliverables. For such arrangements, we identify each of the promised goods and services within the contract and the distinct performance obligations at inception and allocate consideration to each performance obligation based on relative standalone selling price, which is generally determined based on the expected cost plus mark-up.
If the contract includes an enforceable right to payment for performance completed to date and performance obligations are satisfied over time, we recognize revenue over the development period using either the input or output method depending on which is most appropriate given the nature of the distinct deliverable. For other contracts that do not contain an enforceable right to payment for performance completed to date, revenue is recognized when control is transferred to the customer. Factors that may indicate that the transfer of control has occurred include the transfer of legal title, transfer of physical possession, the customer has obtained the significant risks and rewards of ownership of the assets and we have a present right to payment.
Our typical payment terms for development contracts may include an upfront payment equal to a percentage of the total contract value with the remaining portion to be billed upon completion and transfer of the individual deliverables or satisfaction of the individual performance obligations. We record a liability for cash received in advance of performance, which is presented within deferred revenue in the Consolidated Balance Sheets and recognized as revenue when the associated performance obligations have been satisfied. We recognized $3,889 in licensing and development revenue in connection with contract liabilities that were outstanding as of December 31, 2020 and satisfied during the year ended December 31, 2021.
License fees and milestones received in exchange for the grant of a license to our functional intellectual property (“IP”) such as patented technology and know-how in connection with a partnered development arrangement are generally recognized at inception of the arrangement, or over the development period depending on the facts and circumstances, as the license is generally not distinct from the non-licensed goods or services to be provided under the contract. Milestone payments that are contingent upon the occurrence of future events, are evaluated and recorded at the most likely amount, and to the extent that it is probable that a significant reversal will not occur when the associated uncertainty is resolved.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Royalties
We earn royalties in connection with licenses granted under license and development arrangements with partners. Royalties are based upon a percentage of commercial sales of partnered products with rates ranging from mid-single digits to low double digits and are tiered based on levels of net sales. These sales-based royalties, for which the license was deemed the predominant element to which the royalties relate, are estimated and recognized in the period in which the partners’ commercial sales occur. The royalties are generally reported and payable to us within 45 to 60 days of the end of the period in which the commercial sales are made. We base our estimates of royalties earned on actual sales information from our partners when available or estimated prescription sales from external sources and estimated net selling price. If actual royalties received are different than amounts estimated, we would adjust the royalty revenue in the period in which the adjustment becomes known.
Remaining Performance Obligations
Remaining performance obligations represent the transaction price of firm orders and development contract deliverables for which work has not been completed or orders fulfilled, and excludes potential purchase orders under ordering-type supply contracts with indefinite delivery or quantity. As of December 31, 2021, the aggregate value of remaining performance obligations, excluding contracts with an original expected length of one year or less, was $14,879. We expect to recognize revenue on the remaining performance obligations over the next three years, with the majority being recognized in the next twelve months.
Share-Based Compensation
We use share-based compensation in the form of stock options, restricted stock units (“RSUs”) and performance-based restricted stock units (“PSUs”). We record compensation expense associated with share-based awards granted to employees at the fair value of the award on the date of grant. The Black-Scholes option valuation model is used to determine the fair value of stock options. The fair values of RSU and PSU grants containing service or performance conditions are based on the market value of our common stock on the date of grant. The fair value of PSUs containing a market condition are estimated using a Monte Carlo simulation. The value of the portion of the award that is ultimately expected to vest is expensed ratably over the requisite service period as compensation expense in the Consolidated Statements of Operations. Forfeitures are recorded as incurred. Assumptions concerning our stock price volatility and projected employee exercise behavior over the contractual life of the award impact the estimated fair value of the stock option awards.
Research and Development
Research and development expenses include costs directly attributable to the conduct of research and development programs including personnel costs, materials and supplies associated with design work and prototype development, FDA filing fees and the cost of services provided by outside contractors such as expenses related to clinical trials. All costs associated with research and development activities are expensed as incurred.
Income Taxes
Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. Valuation allowances are established when necessary to reduce deferred tax assets to the amount expected to be realized.
We account for uncertain tax positions in accordance with ASC 740, Income Taxes (“ASC 740”), which applies to all tax positions related to income taxes. Tax benefits are recognized when it is more-likely-than-not that a tax position will be sustained upon examination by the authorities. Interest and penalties accrued related to uncertain tax benefits are recognized as a component of income tax expense in the Consolidated Statements of Operations.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Earnings (Loss) Per Common Share
Basic earnings (loss) per common share is computed by dividing the net income (loss) applicable to common stockholders by the weighted-average number of common shares outstanding for the period. Diluted earnings (loss) per common share is computed in a similar manner, except that the weighted average number of shares outstanding is increased to reflect the potential dilution from the exercise or conversion of securities into common stock. Diluted earnings (loss) per common share contemplate a complete conversion to common shares of all convertible instruments only if such instruments are dilutive in nature with respect to earnings (loss) per common share.
Segments
Operating segments are components of an enterprise for which separate financial information is available and is evaluated regularly by the Chief Operating Decision Maker (“CODM”), our Chief Executive Officer, in deciding how to allocate resources and assess performance. Our CODM currently evaluates our operations as a whole from a number of different operational perspectives, including but not limited to, on a product-by-product, customer and partner basis. We derive all significant revenues from pharmaceutical products and development services, and have a single reportable, operating segment of business.
Going Concern
We are responsible for evaluating, and providing disclosure of uncertainties about, our ability to continue as a going concern. As of December 31, 2021, we had cash and cash equivalents of $65,913. Based on our evaluation, we concluded there is no substantial doubt or uncertainty about our ability to meet our obligations within one year from the date the Consolidated Financial Statements were issued.
Note 3. Inventories
Inventories consisted of the following:
| | | | | | | | | | | |
| December 31, 2021 | | December 31, 2020 |
| Raw materials | $ | 325 | | | $ | 325 | |
| Work in process | 6,784 | | | 7,120 | |
| Finished goods | 4,435 | | | 10,771 | |
| Total inventories, net | $ | 11,544 | | | $ | 18,216 | |
A reserve is recorded for potentially excess, dated or obsolete inventory based on an analysis of inventory on hand compared to forecasted future sales, which was $214 and $619 as of December 31, 2021 and 2020, respectively. In 2021, we wrote off $359 of inventory and reduced the reserve for excess, dated or obsolete inventory by $46. In 2020, we wrote off $356 of inventory and increased the reserve for excess, dated or obsolete inventory by $511.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Note 4. Property and Equipment
Property and equipment, net consisted of the following:
| | | | | | | | | | | |
| December 31, 2021 | | December 31, 2020 |
| Production molds, tooling and equipment | $ | 22,069 | | | $ | 20,260 | |
| Leasehold improvements | 7,559 | | | 6,298 | |
| Furniture, fixtures and office equipment | 907 | | | 865 | |
| Computer equipment and software | 1,717 | | | 756 | |
| Construction and tooling in process | 6,942 | | | 6,214 | |
| Total property and equipment | 39,194 | | | 34,393 | |
| Less: Accumulated depreciation | (13,179) | | | (10,373) | |
| Total property and equipment, net | $ | 26,015 | | | $ | 24,020 | |
Depreciation expense was $2,864, $2,341 and $2,205 for the years ended December 31, 2021, 2020 and 2019, respectively. In 2021 and 2020, we disposed of certain property and equipment that was fully depreciated and no longer used. We capitalized $52 and $231 of interest costs associated with construction of property and equipment during the years ended December 31, 2021 and 2020, respectively.
Note 5. Leases
We are party to non-cancellable operating leases for our corporate headquarters facilities in Ewing, New Jersey, and two facilities in the suburbs of Minneapolis, Minnesota used for research and development, manufacturing and administrative functions. We have also entered into a master operating lease arrangement for a fleet of vehicles for use by our sales force and other operating leases for various office and warehouse equipment. Our lease agreements do not contain any material residual value guarantees, material bargain purchase options or material restrictive covenants. We have no material sublease arrangements with third parties or lease transactions with related parties.
On November 1, 2021, January 1, 2022 and March 1, 2022, we entered into two-month lease extensions on our operating lease for our corporate headquarters in Ewing, New Jersey. The three extensions set new lease expiration dates of December 31, 2021, February 28, 2022 and April 30, 2022, respectively, and maintained the same conditions as the original lease.
On July 1, 2019, we entered into an operating lease for approximately 75,000 square feet of office, laboratory, manufacturing and warehousing space in Minnetonka, Minnesota. The initial lease term is 12.5 years with an option to renew the lease for one additional renewal period of three years. The landlord delivered possession of the premises to us on July 1, 2019 and payment of rent commenced on January 1, 2020. The lease provides for the payment of fixed base rent and additional rent for operating expenses, insurance premiums and taxes. We are completing the build-out of the premises at our cost with an allowance for tenant improvement to be reimbursed by the landlord up to $1,200.
The operating leases require payment of all executory costs such as maintenance and property taxes. Operating lease costs were $2,176, $2,174 and $1,391 for the years ended December 31, 2021, 2020 and 2019 respectively. Cash paid for amounts included in the measurement of operating lease liabilities was $2,005, $1,884 and $1,401 and non-cash operating lease ROU assets obtained in exchange for operating lease obligations were $850, $778 and $6,511 for the years ended December 31, 2021, 2020 and 2019 respectively. As of and for the years ended December 31, 2021, 2020 and 2019 the weighted average discount rate was approximately 8.9%, 8.6% and 8.3% respectively, and the weighted average remaining lease term was 8.3 years, 8.3 years and 8.4 years respectively.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Future lease payments under non-cancelable leases for the next five years and thereafter as of December 31, 2021 are as follows:
| | | | | |
| Future Lease Payments |
| 2022 | $ | 1,334 | |
| 2023 | 969 | |
| 2024 | 762 | |
| 2025 | 676 | |
| 2026 | 678 | |
| Thereafter | 3,593 | |
Total remaining lease payments | 8,012 | |
| Less: Imputed interest | (2,532) | |
Present value of lease liabilities | $ | 5,480 | |
As of December 31, 2021, we have no material additional operating leases that have not yet commenced.
Note 6. Intangible Assets
Intangible assets are as follows:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | December 31, 2021 | | December 31, 2020 |
| Useful Life
(in Years) | | Gross Carrying Amount | | Accumulated Amortization | | Net Book Value | | Gross Carrying Amount | | Accumulated Amortization | | Net Book Value |
TLANDO® product rights | 10 | | $ | 11,315 | | | $ | — | | | $ | 11,315 | | | $ | — | | | $ | — | | | $ | — | |
NOCDURNA® product rights | 10 | | 7,500 | | | (937) | | | 6,563 | | | 7,500 | | | (188) | | | 7,312 | |
Patents 1 | 5 - 10 | | 1,048 | | | (1,047) | | | 1 | | | 3,995 | | | (3,614) | | | 381 | |
Total intangibles, net | | | $ | 19,863 | | | $ | (1,984) | | | $ | 17,879 | | | $ | 11,495 | | | $ | (3,802) | | | $ | 7,693 | |
1 Patents related to OTREXUP® were sold as part of the Asset Purchase Agreement entered into with Otter in December 2021. See Note 12 for further discussion regarding the sale of assets.
In October 2021, we entered into an exclusive license agreement (the “TLANDO® License Agreement”) with Lipocine, Inc. (“Lipocine”) for the right to commercialize the product TLANDO® (testosterone undecanoate) in the U.S., a twice-daily oral formulation of testosterone for testosterone replacement therapy indicated for conditions associated with a deficiency or absence of endogenous testosterone, or hypogonadism in adult males. TLANDO® was granted tentative approval from the FDA in December 2020 and will be eligible for final approval and marketing in the U.S. upon expiration of the exclusivity period previously granted to Clarus for JATENZO® on March 27, 2022. Under the terms of the TLANDO® License Agreement, we paid Lipocine an upfront payment of $11,000 upon execution of agreement. Lipocine is eligible for additional milestone payments up to $10,000, minimum tiered royalty payments of $4,500 over the first three years after commercialization and commercial milestones potentially totaling up to $160,000 based on net sales of TLANDO® in the U.S. We are also obligated to purchase $2,002 of TLANDO® inventory from Lipocine of which $1,056 was purchased as of December 31, 2021. We accounted for the transaction as an asset purchase. Amortization of the product rights intangible asset, including direct transaction costs of $315, will commence and be included in selling, general and administrative expenses upon commercialization of TLANDO® once final approval is obtained from the FDA after the exclusivity period previously granted to Clarus Therapeutics, Inc. (“Clarus”) for JATENZO® expires on March 27, 2022. The additional milestone and commercial milestone payments associated with TLANDO® are contingent on future events and will be accrued when they are both probable and estimable. Royalty payments will be accrued and included in costs of product sales as incurred.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
In connection with the NOCDURNA® license and commercial supply agreement entered into with Ferring International Center S.A. and its affiliates (“Ferring”) in October 2020, we paid Ferring an upfront payment of $5,000 upon execution and an additional $2,500 in October 2021. Ferring is eligible for tiered royalties and additional commercial milestone payments potentially totaling up to $17,500 based on net sales of NOCDURNA® in the U.S. We accounted for the transaction as an asset purchase. Amortization of the product rights intangible asset is included in selling, general and administrative expenses. The royalty payments are accrued and included in the cost of product sales as incurred. The commercial milestones were determined to be contingent liabilities and will be accrued when they are both probable and estimable.
Amortization expense for the years ended December 31, 2021, 2020 and 2019 was $1,037, $286 and $352, respectively, and is recorded in selling, general and administrative expenses in the Consolidated Statements of Operations. Estimated future aggregate amortization expense is as follows:
| | | | | |
| Estimated
Amortization
Expense |
| 2022 | $ | 1,600 | |
| 2023 | 1,882 | |
| 2024 | 1,882 | |
| 2025 | 1,882 | |
| 2026 | 1,882 | |
| Thereafter | 8,751 | |
| Total future amortization expense | $ | 17,879 | |
Future amortization amounts presented above are estimates. Actual future amortization expense may be different due to future acquisitions, impairments, changes in amortization periods or other factors.
Note 7. Accrued Expenses and Other Current Liabilities
Accrued expenses and other liabilities consisted of the following:
| | | | | | | | | | | |
| December 31, 2021 | | December 31, 2020 |
| Product returns and sales allowances | $ | 20,563 | | | $ | 11,435 | |
| Accrued employee compensation and benefits | 5,648 | | | 4,555 | |
| License fees payable | — | | | 2,500 | |
| Other accrued expenses and liabilities | 8,832 | | | 7,145 | |
| Total accrued expense and other liabilities | $ | 35,043 | | | $ | 25,635 | |
Note 8. Long-Term Debt
Term Loan
On June 6, 2017, we entered into a loan and security agreement (the “Loan Agreement”) with Hercules Capital, Inc., for a term loan of up to $35,000 (the “Term Loan”), under which we initially borrowed $25,000 (“Tranche I”), the proceeds of which are being used for working capital and general corporate purposes. The Term Loan was secured by substantially all of our assets, excluding intellectual property and accrued interest at a calculated prime-based variable rate with a maximum interest rate of 9.50%. The interest rate in effect as of December 31, 2020 was 8.50%. Payments under the loan were interest-only until the first principal payment was due.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
On June 26, 2019, we entered into a First Amendment (the “Amendment”) to the Loan Agreement, which increased the aggregate principal amount available under the Term Loan from $35,000 to $50,000 and extended the interest-only payment period of the Term Loan to August 1, 2021. The interest only period could be further extended to August 1, 2022 if we achieved a certain loan extension milestone, requested such extension, and paid an extension fee equal to one half of one percent of the principal amount outstanding. Upon signing of the Amendment, an additional $15,000 (“Tranche II”) was funded to us. The Term Loan maturity date remained July 1, 2022; however, the Term Loan maturity date could be extended to July 1, 2024 contingent upon satisfaction of a certain loan extension milestone. We were eligible, but not obligated, to request one or more additional advances of at least $5,000, not to exceed $10,000 in the aggregate (“Tranche III”). Our option to request additional advances expired on October 31, 2020.
We were required to pay an end of term fee (“End of Term Charge”) equal to 4.25% of Tranche I and 3.95% of the borrowings under Tranche II, payable upon the earlier of July 1, 2022 or repayment of the Term Loan. The Loan Agreement also imposed a prepayment fee of 1.0% to 3.0% if any or all of the balance were prepaid prior to the maturity date.
As of December 31, 2020, the carrying value of the Term Loan was $40,899, which consisted of the principal balance outstanding and the End of Term Charge accrual, less unamortized debt issuance costs that are being amortized/accrued to interest expense over the term of the Term Loan using the effective interest method. The fair value of our debt was estimated to approximate the carrying value based on our understanding of current market conditions and rates we could obtain for similar loans at that time.
In July 2021, having previously met the loan extension milestone, we requested that the interest-only period be extended to August 1, 2022 and the maturity date be extended to July 1, 2024 in accordance with the terms of the Amendment. The Lender granted the extension of the interest-only period and maturity date and waived the extension fee. In 2021, we made principal prepayments of $20,000 and paid a 1.0% prepayment fee.
On November 1, 2021, we extinguished the Loan Agreement with Hercules Capital, Inc. and repaid the outstanding $20,000 principal on the Term Loan, along with the 1.0% prepayment fee of $200 and the End of Term Charge of $1,655. All remaining unamortized debt issuance costs associated with the Term Loan were immediately amortized to interest expense.
Credit Facilities
On November 1, 2021, we entered into a Credit Agreement (the “Credit Agreement”) with Wells Fargo Bank, National Association, as administrative agent for the lenders, (“Administrative Agent”) for credit facilities in an aggregate principal amount of up to $40,000 with a maturity date of November 1, 2024. The Credit Agreement provides for a $20,000 term loan facility (the “Term Loan Facility”) and a $20,000 revolving credit facility, $5,000 of which is available for the issuance of letters of credit and $1,000 of which is available for Swingline loans (the “Revolving Credit Facility”), (collectively the “Credit Facilities”), which are secured by substantially all of our assets. The Term Loan Facility was funded upon execution of the Credit Agreement with the proceeds used to repay our $20,000 Term Loan with Hercules Capital, Inc. and to pay fees and expenses incurred in connection with the early repayment. The Revolving Credit Facility remains available for future use and can be drawn upon for ongoing working capital requirements and other general corporate purposes as needed.
As of December 31, 2021, we had $20,000 outstanding under our Term Loan Facility with a carrying value of $19,741 which consisted of the principal balance outstanding, less unamortized debt issuance costs that are being amortized/accrued to interest expense over the term of the Term Loan Facility using the effective interest rate method. The fair value of our debt is estimated to approximate the carrying value based on our understanding of current market conditions and rates we could obtain for similar loans.
As of December 31, 2021, there were no outstanding borrowings under the Revolving Credit Facility, including no outstanding letters of credit drawn from the Revolving Credit Facility or Swingline loans. Commitment fees are payable on the unused portion of the Revolving Credit Facility at rates between 0.30% and 0.45% based on our Consolidated Total Leverage Ratio, as defined in the Credit Agreement and below, remeasured quarterly. For the year ended December 31, 2021, commitment fees incurred totaled $12.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
As defined in the Credit Agreement governing the Term Loan Facility, principal payments of the outstanding term loans are due in consecutive quarterly installments on the last business day of each of March, June, September and December, commencing on March 31, 2022. The Credit Agreement also requires prepayment of the outstanding loans under the Term Loan Facility, subject to certain exceptions, with (a) 100% of the net cash proceeds of (i) any incurrence or issuance of certain debt, other than debt permitted under the Credit Agreement; (ii) issuance of equity other than that associated with employee compensation; and (iii) certain asset sales and casualty and condemnation events, subject to reinvestment rights and certain other exceptions. We may voluntarily prepay outstanding loans under the Term Facility at any time without premium or penalty. All obligations under the Term Facility are secured, subject to certain exceptions, by substantially all of our assets and the assets of our subsidiaries.
Borrowings made under the Credit Agreement bear interest at a rate per annum equal to either the Base Rate or LIBOR plus the Applicable Margin, as defined in the Credit Agreement. Swingline loans bear interest at a rate per annual equal to the Base Rate plus the Applicable Margin. The Applicable Margin is based on the Consolidated Total Leverage Ratio, as defined in the Credit Agreement and below, remeasured quarterly. Base Rate is defined as the highest of (a) the Prime Rate, (b) the Federal Funds Rate plus 0.50% and (c) LIBOR for an interest period of one month plus 1%. In the event of default, we no longer have the option to request LIBOR rate loans, Swingline Loans or Letters of Credit and all outstanding financial instruments will bear interest at a rate per annum of 2% in excess of the calculated interest rate.
We have the option to select either the Base Rate or LIBOR as the rate of interest for the Term Loan and Revolving Credit Facilities, along with an interest period of either 1-month, 3-months or 6-months. Upon cessation of LIBOR on June 30, 2023, an appropriate benchmark replacement will be determined pursuant to the terms of the Credit Agreement. We have not yet evaluated the impact the cessation of LIBOR will have on our financial condition and results of operations. As of December 31, 2021, the Applicable Margin was 1.50% for Base Rate loans and 2.50% for LIBOR loans with a 1-month LIBOR selected as the rate of interest for the Term Loan Facility. The weighted average interest rate on the Term Loan Facility outstanding balance during the year ended December 31, 2021 was approximately 2.59%.
Under the Credit Agreement, we are subject to customary affirmative and negative covenants, including, among others, restrictions on our ability to incur debt; create liens; make investments; merge, consolidate or dispose of assets or subsidiaries; enter into transactions with affiliates; modify accounting practices, our year end and organizational documents; pledge assets; revise nature of business; perform sale leasebacks; and enter into any restrictive agreements and customary events of default (including payment defaults, covenant defaults, change of control defaults and bankruptcy defaults). The Credit Agreement also contains financial covenants, including the ratio of consolidated total indebtedness to consolidated earnings before income, taxes, depreciation and amortization (“Consolidated EBITDA”) (“Consolidated Total Leverage Ratio”), as defined in the Credit Agreement” and the ratio of consolidated senior secured indebtedness to Consolidated EBITDA (“Consolidated Senior Secured Leverage Ratio”), as well as the ratio of Adjusted EBITDA to consolidated fixed charges (“Consolidated Fixed Charge Coverage Ratio”), as defined in the Credit Agreement. These covenants restrict our ability to purchase outstanding shares of our common stock. As of December 31, 2021, we were in compliance with all affirmative, negative and financial covenants.
Future principal payments under the Term Loan Facility are as follows:
| | | | | |
| Future Principal Payments |
| 2022 | $ | 1,500 | |
| 2023 | 1,500 | |
| 2024 | 17,000 | |
| Total future principal payments | $ | 20,000 | |
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Note 9. Stockholders’ Equity
In August 2017, we entered into a sales agreement (the “Sales Agreement”) with Cowen and Company, LLC (“Cowen”) under which we could offer and sell, from time to time and at our sole discretion, shares of our common stock having an aggregate offering price of up to $30,000 through Cowen as our sales agent and/or as principal. Cowen could sell the common stock by any method permitted by law deemed to be an “at the market offering” as defined in Rule 415 of the Securities Act of 1933, as amended (the “ATM Facility”). The Sales Agreement requires us to pay a commission of 3.0% of the gross sales proceeds of any common stock sold through Cowen.
During year ended December 31, 2019, we sold 2,307 shares of common stock under the ATM Facility, resulting in net offering proceeds of $7,781. On June 26, 2019, the Company delivered written notice to Cowen that it was terminating the Sales Agreement effective July 6, 2019, and accordingly the ATM Facility is no longer available for use.
Note 10. Share-Based Compensation
We have an Equity Compensation Plan (the “Plan”), which allows for grants in the form of incentive stock options, non-qualified stock options, stock units, stock awards, stock appreciation rights, and other stock-based awards. The Plan was amended and restated in June 2021 to increase the total number of shares available for grant under the Plan by 10,000 shares. The cumulative number of shares that have been authorized for issuance under the Plan to date is 50,200 shares and the maximum number of shares of stock that may be granted to any one participant during a calendar year is 4,000 shares. Options to purchase shares of common stock are granted at exercise prices not less than 100% of fair market value on the date of grant. The term of each option is ten years, and the options typically vest over a three-year period with a minimum vesting period of one year. As of December 31, 2021, the Plan had approximately 366 shares available for grant. Stock option exercises, and the vesting of restricted stock and performance stock awards, are satisfied through the issuance of new shares.
Stock Options
Stock option activity under the Plan is as follows:
| | | | | | | | | | | | | | | | | | | | | | | |
| Number of
Shares | | Weighted
Average
Exercise
Price | | Weighted
Average
Remaining
Contractual
Term (Years) | | Aggregate Intrinsic
Value |
| Outstanding as of December 31, 2018 | 14,079 | | $ | 2.19 | | | | | |
Granted | 2,489 | | 3.01 | | | | | |
Exercised | (2,572) | | 1.76 | | | | | $ | 6,477 | |
Cancelled / Forfeited | (135) | | 2.81 | | | | | |
| Outstanding as of December 31, 2019 | 13,861 | | 2.41 | | | 6.7 | | 31,713 | |
Granted | 3,335 | | 2.73 | | | | | |
Exercised | (939) | | 1.93 | | | | | 1,072 | |
Cancelled / Forfeited | (736) | | 2.83 | | | | | |
| Outstanding as of December 31, 2020 | 15,521 | | 2.49 | | | 6.6 | | 23,407 | |
Granted | 2,660 | | 4.37 | | | | | |
Exercised | (2,307) | | 2.27 | | | | | 5,052 | |
Cancelled / Forfeited | (297) | | 3.02 | | | | | |
| Outstanding as of December 31, 2021 | 15,577 | | | 2.83 | | | 6.5 | | 13,839 | |
| Exercisable as of December 31, 2021 | 10,644 | | | $ | 2.46 | | | 5.4 | | $ | 12,028 | |
The per share weighted average fair value of options granted during 2021, 2020 and 2019 was estimated as $2.29, $1.42 and $1.54, respectively, on the date of grant using the Black-Scholes option pricing model based on the assumptions noted in the table below. Expected volatilities are based on the historical volatility of our stock. The weighted average expected life is based on both historical and anticipated employee behavior.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Risk-free interest rate | 0.8 | % | | 0.4 | % | | 1.9 | % |
| Annualized volatility | 59.3 | % | | 59.4 | % | | 55.7 | % |
| Weighted average expected life (in years) | 5.4 | | 5.5 | | 5.5 |
| Expected dividend yield | 0.0 | % | | 0.0 | % | | 0.0 | % |
Option exercises during 2021, 2020 and 2019 resulted in proceeds of $5,182, $1,814 and $4,405, respectively, and the issuance of shares of common stock of 2,294 in 2021, 939 in 2020 and 2,529 in 2019. In 2021 and 2019, certain options were net exercised, whereby we withheld 13 and 43 shares, respectively, the fair value of which was equivalent to the aggregate exercise price and tax withholding on the date of exercise.
Long Term Incentive Program
Our Board of Directors has approved a long-term incentive program (“LTIP”) for the benefit of our senior executives. Pursuant to the LTIP, our senior executives are awarded stock options, restricted stock units (“RSU”) and performance stock units (“PSU”) with targeted values based on similar award structures granted by our peer group. The stock options have a ten-year term, an exercise price equal to the closing price of our common stock on the date of grant, vest in quarterly installments over three years, were otherwise granted on the same standard terms and conditions as other stock options granted pursuant to the Plan and are included in the stock options table above. The RSUs generally vest in three equal annual installments, and the PSUs vest and convert into shares of our common stock based on our attainment of certain performance goals over a performance period, which is typically three years.
PSUs and RSUs granted under the LTIP are summarized as follows:
| | | | | | | | | | | | | | | | | | | | | | | |
| Performance Stock Units | | Restricted Stock Units |
| Number of Shares | | Weighted Average Grant Date Fair Value | | Number of Shares | | Weighted Average Grant Date Fair Value |
| Outstanding as of December 31, 2018 | 1,842 | | | $ | 2.41 | | | 1,226 | | | $ | 2.44 | |
Granted | 593 | | | 2.99 | | | 789 | | 2.92 | |
Incremental shares earned | 59 | | | 1.25 | | | — | | | — |
Vested / Settled | (415) | | 1.18 | | | (614) | | 2.19 | |
Forfeited / Expired | (238) | | 1.12 | | | — | | — |
| Outstanding as of December 31, 2019 | 1,841 | | | 3.00 | | | 1,401 | | | 2.82 | |
Granted | 605 | | | 2.00 | | | 1,078 | | | 2.73 | |
Incremental shares earned | 77 | | | 3.10 | | | — | | | — |
Vested / Settled | (388) | | 3.11 | | | (785) | | 2.80 | |
Forfeited / Expired | (494) | | 3.02 | | | (127) | | 2.83 | |
| Outstanding as of December 31, 2020 | 1,641 | | | 2.61 | | | 1,567 | | | 2.77 | |
Granted | 243 | | | 5.55 | | | 769 | | | 4.42 | |
Incremental shares earned | 210 | | | 3.18 | | | — | | | — | |
Vested / Settled | (766) | | 2.86 | | | (832) | | 2.76 | |
| Outstanding as of December 31, 2021 | 1,328 | | | $ | 3.04 | | | 1,504 | | | $ | 3.62 | |
The outstanding balance of PSUs is stated at the target number of shares to be awarded upon attainment of certain performance goals. Depending on the outcome of the related performance goals, a recipient may ultimately earn a number of shares that is greater or less than the target number of units granted, ranging from 0% to 150%. The balance of PSUs outstanding as of December 31, 2021 included 308 units granted in 2019 with a performance period ended December 31, 2021 that were subsequently deemed to be achieved and approved for settlement in the first quarter of 2022 for a total of 304 shares.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
In each of the years in the three-year period ended December 31, 2021, the LTIP awards include PSUs that will be earned based on our total shareholder return (“TSR”) as compared to the Nasdaq Biotechnology Index (“NBI”) at the end of the respective annual performance periods. The fair values of the TSR PSUs granted were determined using a Monte Carlo simulation and used the following inputs and assumptions:
| | | | | | | | | | | | | | | | | |
| 2021 Award | | 2020 Award | | 2019 Award |
| Closing stock price on grant date | $ | 4.42 | | | $ | 2.73 | | | $ | 2.92 | |
| Performance period starting price | $ | 3.70 | | | $ | 4.78 | | | $ | 3.01 | |
| Term of award (in years) | 2.56 | | 2.55 | | 2.55 |
| Volatility | 54.4 | % | | 57.5 | % | | 63.7 | % |
| Risk-free interest rate | 0.23 | % | | 0.21 | % | | 1.79 | % |
| Expected dividend yield | 0.00 | % | | 0.00 | % | | 0.00 | % |
| Fair value per TSR PSU | $ | 5.55 | | | $ | 2.00 | | | $ | 3.18 | |
The performance period starting price is measured as the average closing price over the last 20 trading days prior to the performance period start. The Monte Carlo simulation model also assumed correlations of returns of the prices of our common stock and the common stocks of the NBI companies and stock price volatilities of the NBI companies. The fair value of the target number of shares that can be earned under the TSR PSUs is being recognized as compensation expense over the term of the award. Other PSUs that are not market-based awards are expensed using the grant date fair value of shares expected to vest over the remaining performance period when it becomes probable that the related performance goal will be achieved.
LTIP awards are generally net-share settled such that we withhold shares with value equivalent to the employees’ minimum statutory obligation for the applicable income and other employment taxes, and remit cash to the appropriate taxing authorities. Total shares withheld for net-settled awards were 626, 425 and 409 in 2021, 2020 and 2019, respectively, and were based on the value of the shares on their vesting date as determined by our closing stock price. Total payments for the employees’ tax obligations to the taxing authorities were $2,841, $1,367 and $1,131 in 2021, 2020 and 2019, respectively, and are reflected as a financing activity within the Consolidated Statements of Cash Flows. These net-share settlements reduced the number of shares that would have otherwise been issued as a result of the vesting.
Members of our Board of Directors also receive grants of RSUs that vest one year from the date of grant. Certain Directors have elected to defer receipt of vested shares until retirement or separation from the Board of Directors, for which 30, 72 and no shares vested with deferral as of and for the years ended December 31, 2021, 2020 and 2019, respectively.
Share-based Compensation Expense
Compensation costs incurred in connection with share-based awards are as follows:
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Stock options | $ | 4,102 | | | $ | 3,709 | | | $ | 3,436 | |
| Restricted stock units | $ | 2,620 | | | 2,239 | | | 1,830 | |
| Performance stock units | $ | 1,293 | | | 2,000 | | | 1,204 | |
| Total share-based compensation expense | $ | 8,015 | | | $ | 7,948 | | | $ | 6,470 | |
As of December 31, 2021, there was $6,838 of total unrecognized compensation costs related to non-vested stock option awards that are expected to be recognized over a weighted average period of approximately 1.95 years.
Note 11. Employee 401(k) Savings Plan
We sponsor a 401(k) defined contribution retirement savings plan that covers all U.S. employees who have met minimum age and service requirements subject to the provisions of the Employee Retirement Income Security Act. Under the plan, eligible employees may contribute a portion of their annual compensation into the plan up to the IRS annual limits on a pre-tax or after-
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
tax basis. We have elected to make matching contributions to the plan based on a percentage of employee contributions. The total amount contributed by us is determined by plan provisions for matching contributions, as well as at our discretion. Employer matching and discretionary contributions were $1,151, $1,097 and $993 for the years ended December 31, 2021, 2020 and 2019, respectively.
Note 12. Sale of Assets
In December 2021, we entered into an asset purchase agreement (the “Asset Purchase Agreement”) with Otter Pharmaceuticals, LLC (a subsidiary of Assertio Holdings, Inc., together with Assertio Holdings, Inc., as guarantor, individually and collectively referred to as “Otter”) to sell certain worldwide assets used in the operation of the OTREXUP® product line for $44,021 of which we received a $18,000 at closing and will receive the remaining $26,021 in installments over a one-year period. As of December 31, 2021, we recorded an increase to the purchase price for estimated changes in closing inventory to be transferred. The Asset Purchase Agreement included the transfer of OTREXUP® patents, trademark and intellectual property, product finished goods and sample inventory, and certain other contracts associated with the OTREXUP® product line. Subject to the terms of the OTREXUP® Asset Purchase Agreement, we generally retained ownership (and related liabilities) of assets not solely related to the OTREXUP® product line. We also agreed via the execution of a separate supply agreement to continue to manufacture and supply OTREXUP® and sample products to Otter at cost plus mark-up. Further, we entered into a license agreement with Otter pursuant to which we granted Otter a worldwide, exclusive, fully paid-up license to certain patents relating to OTREXUP® that may relate to our other products.
We recorded the entire $38,591 gain on sale of assets in the Consolidated Statements of Operations for the year ended December 31, 2021 as all requirements of the agreement were determined to have been met and it was probable that a significant reversal of the gain would not occur. The gain includes the purchase price of $44,021 adjusted for estimated changes in closing inventory to be transferred less the net book value of the assets sold and direct transaction costs. The remaining $26,311 purchase price to be received is classified as other receivables in the Consolidated Balance Sheets as of December 31, 2021, and we recognized $17,825 of net proceeds from the sale of assets in the Statements of Cash Flows for the year ended December 31, 2021.
Note 13. Income Taxes
We were subject to taxes in both the U.S. and Switzerland in each of the years ended December 31, 2021, 2020 and 2019. Income (loss) before income taxes was derived from the following jurisdictions:
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| U.S. | $ | 62,626 | | | $ | 10,284 | | | $ | (1,734) | |
| Switzerland | (355) | | | (363) | | | (293) | |
| Total income (loss) before income taxes | $ | 62,271 | | | $ | 9,921 | | | $ | (2,027) | |
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
The income tax provision (benefit) was comprised of:
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Current | | | | | |
Federal | $ | — | | | $ | — | | | $ | — | |
State | 2,041 | | | 700 | | | — |
Foreign | 2 | | | 2 | | | — |
Total current income tax provision (benefit) | 2,043 | | | 702 | | | — | |
| Deferred | | | | | |
Federal | 11,918 | | | (39,542) | | | — |
State | 2,021 | | | (7,440) | | | — |
Foreign | — | | — | | — | |
Total deferred income tax provision (benefit) | 13,939 | | | (46,982) | | | — | |
| Total income tax provision (benefit) | $ | 15,982 | | | $ | (46,280) | | | $ | — | |
Effective tax rates differ from statutory income tax rates as follows:
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Statutory income tax rate | 21.0 | % | | 21.0 | % | | 21.0 | % |
| State income taxes | 5.5 | | | 7.1 | | | 14.4 | |
| Effect of foreign operations | 0.1 | | | 0.2 | | | (1.0) | |
| Changes in valuation allowance | (0.2) | | | (516.5) | | | (59.9) | |
| Change in unused net operating loss and credit carryforwards | — | | | — | | | 24.7 | |
| Change in uncertain tax positions | (0.1) | | | 21.4 | | | — | |
| Research and development credit | (0.7) | | | (6.0) | | | — | |
| Stock-based compensation | (2.0) | | | 3.7 | | | 22.3 | |
| 162(m) limitation | 2.1 | | | 1.9 | | | (18.2) | |
| Nondeductible items | — | | | 1.6 | | | (1.8) | |
| Impact of Tax Cuts and Jobs Act | — | | | — | | | (1.5) | |
| Other | — | | | (0.9) | | | — | |
Effective income tax rate | 25.7 | % | | (466.5) | % | | — | % |
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Deferred tax assets (liabilities) consist of the following:
| | | | | | | | | | | |
| December 31, 2021 | | December 31, 2020 |
| Gross deferred tax assets | | | |
Net operating loss carryforward – U.S. | $ | 24,738 | | | $ | 36,071 | |
Net operating loss carryforward – Switzerland | 162 | | | 106 | |
Research and development tax credit carryforward | 5,836 | | | 5,418 | |
Deferred revenue | 14 | | | 219 | |
Stock-based compensation | 3,423 | | | 2,954 | |
Inventory reserve | 56 | | 159 | |
Compensation accruals | 1,426 | | | 1,304 | |
Product reserves | 5,235 | | | 2,820 | |
Operating lease liabilities | 1,436 | | | 1,546 | |
Amortization | 64 | | | 607 | |
Other | 188 | | | 145 | |
Total deferred tax assets | 42,578 | | | 51,349 | |
| Deferred tax liabilities | | | |
Depreciation | (1,753) | | | (1,838) | |
Operating lease right-of-use asset | (1,048) | | | (1,303) | |
| Installment sale | (5,580) | | | — | |
Total deferred tax liabilities | (8,381) | | | (3,141) | |
| Net deferred tax asset before valuation allowance | 34,197 | | | 48,208 | |
| Less: Valuation allowance | (1,154) | | | (1,226) | |
Net deferred tax asset | $ | 33,043 | | | $ | 46,982 | |
In assessing the realizability of deferred tax assets, we consider whether it is more likely than not that some portion or all of the deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which temporary differences become deductible or in which net operating loss or tax credit carryforwards can be used. As of each reporting date, we consider new evidence, both positive and negative, that could affect our view of the future realization of deferred tax assets.
As of December 31, 2021 and 2020, there is sufficient positive evidence to conclude that it is more likely than not that our net U.S. deferred tax assets of $33,043 and $46,982, respectively, are realizable as a result of generating pretax earnings, utilizing net operating loss carryovers and projecting pre-tax earnings. For the year ended December 31, 2020, we recorded a net valuation allowance release of $53,383 based on our reassessment of the amount of our deferred tax assets that are more likely than not to be realized. The valuation allowances of $1,154 and $1,226 as of December 31, 2021 and 2020, respectively, relate to certain state and foreign carryovers for which projected income cannot support utilization.
We have a U.S. federal net operating loss carryforward as of December 31, 2021 of $99,939, which, subject to limitations of Internal Revenue Code (“IRC”) Section 382, is available to reduce income taxes payable in future years. As of December 31, 2021, we have performed a full analysis of IRC Section 382 and concluded that net operating losses and credits will be able to be used without limitation. If not used, the portion of the carryforward generated before 2018 will expire in the years 2033 through 2037, and the net operating loss carryforward generated in 2018 and any future years will carry forward indefinitely. Additionally, we have U.S. Research Credit carryforwards of $7,328 which will expire in years 2022 through 2041 if unused.
We also have a Swiss net operating loss carryforward as of December 31, 2021, of $1,130, which is available to reduce income taxes payable in future years. If not used, this carryforward will begin to expire in 2023.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
A summary of changes to our liability for unrecognized tax benefits is as follows:
| | | | | | | | | | | |
| December 31, 2021 | | December 31, 2020 |
| Beginning liability for unrecognized tax benefits | $ | 2,127 | | | $ | — | |
| Increase (decrease) due to tax positions related to prior years | (70) | | | 2,067 | |
| Increase due to tax positions related to the current year | — | | | 60 | |
| Ending liability for unrecognized tax benefits | $ | 2,057 | | | $ | 2,127 | |
Included in the balance of unrecognized tax benefits as of December 31, 2021 and 2020, are $2,057 and $2,127, respectively, that if recognized would impact the effective tax rate. There is no interest or penalties charged or accrued in relation to unrecognized tax benefits. We will classify any future interest and penalties as a component of income tax expense. We do not anticipate that the total amount of unrecognized tax benefits will change significantly in the next twelve months. We are subject to federal and state examinations for the years 2017 and thereafter.
Note 14. Revenues, Significant Customers and Concentrations of Risk
We disaggregate our revenue by type of goods and services and customer location.
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Types of Goods and Services | | | | | |
| Proprietary product sales, net | $ | 80,016 | | | $ | 62,878 | | | $ | 39,215 | |
| Partnered product sales | 46,651 | | | 50,956 | | | 52,888 | |
| Total product revenue, net | 126,667 | | | 113,834 | | | 92,103 | |
| Licensing and development revenue | 19,623 | | | 14,466 | | | 7,529 | |
| Royalties | 37,692 | | | 21,299 | | | 24,232 | |
| Total revenue, net | $ | 183,982 | | | $ | 149,599 | | | $ | 123,864 | |
| | | | | |
| Customer Location | | | | | |
| U.S. | $ | 178,290 | | | $ | 145,789 | | | $ | 120,231 | |
| Europe | 5,692 | | | 3,810 | | | 3,463 | |
| Other | — | | | — | | | 170 | |
| Total revenue, net | $ | 183,982 | | | $ | 149,599 | | | $ | 123,864 | |
Customers from which we derive 10% or more of our total revenue are as follows:
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
| Teva | 42% | | 40% | | 41% |
McKesson 1 | 13% | | 12% | | 10% |
AmerisourceBergen Corporation 1 | 12% | | 12% | | <10% |
Cardinal Health 1 | 11% | | 11% | | <10% |
| Covis | <10% | | <10% | | 20% |
1 Revenue from sales to distributors, net of estimated sales returns and allowances based on shipments.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
Note 15. Earnings (Loss) per Share
Basic earnings (loss) per common share is computed by dividing net income applicable to common stockholders by the daily weighted-average number of common shares outstanding for the applicable period. Diluted earnings (loss) per common share is computed in a similar manner, except that the weighted average number of shares outstanding is increased to reflect the potential dilution from the exercise or conversion of securities into common stock. Diluted earnings (loss) per share contemplates a complete conversion to common shares of all convertible instruments only if such instruments are dilutive in nature with respect to earnings per common share. The following table sets forth the computation for basic and diluted earnings (loss) per common share:
| | | | | | | | | | | | | | | | | |
| Years Ended December 31, |
| 2021 | | 2020 | | 2019 |
Net income (loss) | $ | 46,289 | | | $ | 56,201 | | | $ | (2,027) | |
| | | | | |
Weighted average common shares outstanding | 169,226 | | | 166,066 | | | 162,574 | |
Dilutive effects of stock options and share-based awards issuable under equity compensation plans | 5,507 | | | 4,089 | | | — |
Weighted average dilutive common shares outstanding | 174,733 | | | 170,155 | | | 162,574 | |
| | | | | |
| Earnings (loss) per common share | | | | | |
Basic | $ | 0.27 | | | $ | 0.34 | | | $ | (0.01) | |
Diluted | $ | 0.26 | | | $ | 0.33 | | | $ | (0.01) | |
| | | | | |
Anti-dilutive common stock equivalents 1 | 2,224 | | | 7,092 | | | 17,103 | |
1 These common stock equivalents were outstanding for the period but were not included in the computation of diluted earnings (loss) per common share for those periods as their inclusion would have had an anti-dilutive effect.
Note 16. Commitments and Contingencies
Contingent Considerations
In connection with the TLANDO® exclusive license agreement and asset purchase entered into with Lipocine in October 2021, we paid Lipocine and upfront payment of $11,000 upon execution of agreement. Lipocine is eligible for additional milestone payments up to $10,000, minimum tiered royalty payments of $4,500 over the first three years after commercialization has occurred and commercial milestones up to $160,000 based on net sales of TLANDO® in the U.S. The additional milestone and commercial milestone payments are contingent on future events and will be accrued when they are both probable and estimable. We also have the option to license and develop LPCN 1111 (TLANDO XR) for an additional $4,000 in license fees to be paid in two installments upon exercise of the option, if exercised. The option to license and develop LPCN 111 (TLANDO XR) will be accrued and expensed to research and development when and only if we decide to exercise our option. No decision had been made as of December 31, 2021 to exercise the option; therefore, no accrual was recorded.
In connection with the NOCDURNA® license agreement and asset purchase entered into with Ferring in October 2020, we paid Ferring an upfront payment of $5,000 upon execution and paid an additional $2,500 in October 2021. Ferring is eligible for additional commercial milestone payments potentially totaling up to $17,500 based on our net sales of NOCDURNA® in the U.S.
Pending Litigation
From time to time, we may be involved in various legal matters generally incidental to our business. Although the results of litigation and claims cannot be predicted with certainty, after discussion with legal counsel, we are not aware of any matters for which the likelihood of a loss is probable and reasonably estimable and which could have a material impact on our consolidated financial condition, liquidity, or results of operations.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
On October 23, 2017, Randy Smith filed a complaint in the District of New Jersey, captioned Randy Smith, Individually and on Behalf of All Others Similarly Situated v. Antares Pharma, Inc., Robert F. Apple and Fred M. Powell (“Smith”), Case No. 3:17-cv-8945-MAS-DEA, on behalf of a putative class of persons who purchased or otherwise acquired Antares securities between December 21, 2016 and October 12, 2017, inclusive, asserting claims for purported violations of Sections 10(b) and 20(a) of the Securities Exchange Act of 1934, as amended, against Antares, Robert F. Apple and Fred M. Powell. The Smith complaint contends that defendants made false and/or misleading statements and/or failed to disclose that: (i) Antares had provided insufficient data to the FDA in connection with the NDA for XYOSTED®; and (ii) accordingly, Antares had overstated the approval prospects for XYOSTED®. On July 27, 2018, the court entered an order appointing Serghei Lungu as lead plaintiff, Pomerantz LLP as lead counsel, and Lite DePalma Greenberg, LLC as liaison counsel for plaintiff. On August 3, 2018, the parties submitted a stipulation and proposed order, setting forth an agreed-upon schedule for responding to the complaint, which the court granted. Pursuant to that order, plaintiff filed a Consolidated Amended Class Action Complaint on October 9, 2018. On November 26, 2018, defendants filed a motion to dismiss. Plaintiff filed an opposition to the motion on January 10, 2019 and defendants filed a reply in support of their motion on February 25, 2019. On July 2, 2019, the court dismissed the complaint in its entirety without prejudice. On July 29, 2019, plaintiff filed a Consolidated Second Amended Class Action Complaint against the same parties alleging substantially similar claims. On September 12, 2019, defendants filed a motion to dismiss the Consolidated Second Amended Class Action Complaint. Plaintiffs’ opposition was filed on October 28, 2019 and defendants’ reply in support of their motion was filed on November 27, 2019. On April 28, 2020, the court dismissed the Consolidated Second Amended Class Action Complaint in its entirety. The court further ordered that plaintiff may file an amended complaint by May 29, 2020 and provide the court with a form of the amended complaint that indicates in what respect(s) it differs from the complaint which it proposes to amend. On May 29, 2020, plaintiff filed a Consolidated Third Amended Class Action Complaint and defendants filed a motion to dismiss on July 10, 2020. Briefing on defendants’ motion was complete on August 25, 2020. On February 26, 2021, the court granted defendants’ motion to dismiss with prejudice, and on March 29, 2021 the plaintiff filed a notice of appeal. On June 21, 2021, plaintiff-appellant filed his opening brief. Defendants-appellees’ response brief was filed on August 4, 2021 and plaintiff-appellant’s reply was filed on September 8, 2021. On January 25, 2022, the Third Circuit ruled in defendants’ favor affirming dismissal. If plaintiffs choose to appeal, they have ninety days to file a petition for writ of certiorari to the U.S. Supreme Court. We believe the claims in the Smith action lack merit and intend to continue to defend them vigorously.
On January 12, 2018, a stockholder of the Company filed a derivative civil action, captioned Chiru Mackert, derivatively on behalf of Antares Pharma, Inc., v. Robert F. Apple, et al., in the Superior Court of New Jersey Chancery Division, Mercer County (Case No. C-11-18). On January 17, 2018, another stockholder filed a derivative action in the same court, captioned Vikram Rao, Derivatively on Behalf of Antares Pharma, Inc. v. Robert F. Apple, et al. (Case No. C-4-18). Both complaints name Robert F. Apple, Fred M. Powell, Thomas J. Garrity, Jacques Gonella, Anton Gueth, Leonard S. Jacob, Marvin Samson and Robert P. Roche, Jr. as defendants, and the Company as nominal defendant, and they assert claims for breach of fiduciary duties, unjust enrichment, abuse of control, gross mismanagement, and waste of corporate assets arising from the same facts underlying the Smith securities class action. The plaintiffs seek damages, corporate governance and internal procedure reforms and improvements, restitution, reasonable attorneys’ fees, experts’ fees, costs, and expenses. The parties have filed a stipulation and order consolidating the two actions and staying the proceedings pending the court’s decision on defendants’ motion to dismiss the Smith action; the motion to dismiss in Smith was granted on February 26, 2021 and notice of appeal was filed on March 29, 2021. On January 25, 2022, the Third Circuit ruled in defendants’ favor affirming dismissal of the securities fraud class action. If plaintiffs in the securities action choose to appeal, they have ninety days to file a petition for writ of certiorari to the U.S. Supreme Court.
ANTARES PHARMA, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except per share amounts)
On January 17, 2018, a stockholder of the Company filed a derivative civil action, captioned Robert Clark, Derivatively on Behalf of Antares Pharma, Inc. v. Robert F. Apple, et al. (“Clark”) (Case No. 3:18-cv-703-MAS-DEA), against Robert F. Apple, Thomas J. Garrity, Jacques Gonella, Leonard S. Jacob, Marvin Samson, Anton G. Gueth and Robert P. Roche, Jr. as defendants, and Company as a nominal defendant. The action was filed in the U.S. District Court for the District of New Jersey and asserts claims for breach of fiduciary duties, unjust enrichment, abuse of control, waste of corporate assets, and a violation of Section 14(a) of the Securities Exchange Act of 1934. This complaint relates to the same facts underlying the Smith securities class action and the other derivative actions. The plaintiff in Clark seeks damages, corporate governance and internal procedure reforms and improvements, reasonable attorneys’ fees, accountants’ and experts’ fees, costs, and expenses. The parties have filed a stipulation and order staying the action pending the court’s decision on defendants’ motion to dismiss the Smith action; the motion to dismiss in Smith was granted on February 26, 2021 and notice of appeal was filed on March 29, 2021. After the expiration of all appeals related to the Smith dismissal, the parties shall submit a proposed order regarding the derivative action. On January 25, 2022, the Third Circuit ruled in defendants’ favor affirming dismissal of the securities fraud class action. If plaintiffs in the securities action choose to appeal, they have ninety days to file a petition for writ of certiorari to the U.S. Supreme Court.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
Our management evaluated, under the supervision and with the participation of our Chief Executive Officer and Chief Financial Officer, the effectiveness of disclosure controls and procedures (as defined in Rule 13a-15(e) under the Securities Exchange Act of 1934, as amended) as of the end of the period covered by this report. Based upon this evaluation, our Chief Executive Officer and Chief Financial Officer concluded that, as of December 31, 2021, our disclosure controls and procedures were effective.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting (as defined in Rule 13a-15(f) under the Securities Exchange Act of 1934, as amended). Under the supervision and with the participation of our Chief Executive Officer and the Chief Financial Officer, management conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 2021. This assessment was based on the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission, or COSO, in Internal Control-Integrated Framework (2013).
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that:
(i)pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect transactions and dispositions of the company’s assets;
(ii)provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that the company’s receipts and expenditures are being made only in accordance with authorizations of the company’s management and board of directors; and
(iii)provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Based on management’s assessment using the COSO Internal Control-Integrated Framework (2013) criteria, management has concluded that its internal control over financial reporting was effective as of December 31, 2021 to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements in accordance with U.S. generally accepted accounting principles.
KPMG LLP, an independent registered public accounting firm, has audited the effectiveness of our internal control over financial reporting as of December 31, 2021, and has issued an audit report on our internal control over financial reporting, which appears in Item 8 of this Annual Report on Form 10-K.
Changes in internal control over financial reporting.
There was no change in our internal control over financial reporting that occurred during the quarter ended December 31, 2021 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. OTHER INFORMATION
None.
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
Information required by this item concerning our directors will be set forth under the caption “Election of Directors” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference.
Information required by this item concerning our executive officers will be set forth under the caption “Executive Officers of the Company” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference.
Information required by this item concerning compliance with Section 16(a) of the Exchange Act, as amended, will be set forth under the caption “Section 16(a) Beneficial Ownership Reporting Compliance” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference.
Information required by this item concerning our audit committee, the audit committee as our financial expert and any material changes to the way in which security holders may recommend nominees to our Board of Directors will be set forth under the caption “Corporate Governance” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference.
The Board of Directors adopted a Code of Business Conduct and Ethics applicable to all employees and directors, which is posted on our website at www.antarespharma.com. We will provide copies of our Code of Business Conduct and Ethics without charge upon request. To obtain a copy, please visit our website or send your written request to Antares Pharma, Inc., 100 Princeton South, Suite 300, Ewing, NJ 08628, Attn: Corporate Secretary. With respect to any amendments or waivers of this Code of Business Conduct and Ethics (to the extent applicable to our chief executive officer, principal accounting officer or controller, or persons performing similar functions) we intend to either post such amendments or waivers on our website or disclose such amendments or waivers pursuant to a Current Report on Form 8-K.
ITEM 11. EXECUTIVE COMPENSATION
Information required by this item will be set forth under the caption “Executive Compensation” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
Information required by this item concerning ownership will be set forth under the caption “Security Ownership of Certain Beneficial Owners” and “Security Ownership of Directors and Executive Officers” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference. The following table provides information about our equity compensation plans as of December 31, 2021 (in thousands, except exercise price):
| | | | | | | | | | | | | | | | | | | | |
| Plan Category | | Number of securities
to be issued upon
exercise of
outstanding options,
warrants and rights | | Weighted-
average
exercise price of
outstanding
options,
warrants and
rights | | Number of securities
remaining available
for future issuance
under equity
compensation plans
(excluding shares
reflected in the first
column) |
Equity compensation plans approved by security holders | | 15,577 | | | $2.83 | | 366 | |
Equity compensation plans not approved by security holders | | None | | None | | None |
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
Information required by this item will be set forth under the captions “Certain Relationships and Related Transactions” and “Corporate Governance” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference.
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES
The information required by this item will be set forth under the caption “Ratification of Selection of Independent Registered Public Accountants” in our Definitive Proxy Statement to be filed pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this Annual Report on Form 10-K for our 2022 Annual Meeting of Stockholders and is incorporated herein by reference.
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
(a)The following documents are filed as part of this Annual Report on Form 10-K:
(1)Financial Statements - see Item 8 of Part II in this Annual Report on Form 10-K.
(2)Financial Statement Schedules
All schedules have been omitted because they are not applicable, are immaterial or are not required because the information is included in the consolidated financial statements or the notes thereto.
(3)Item 601 Exhibits - see list of Exhibits below.
(b) Exhibits
The following is a list of exhibits filed as part of, or incorporated by reference into, this Annual Report on Form 10-K.
| | | | | | | | |
Exhibit
No. | | Description |
| | |
| 3.1 | | Certificate of Incorporation of Antares Pharma, Inc. (filed as Exhibit 4.1 to Form S-3 on April 12, 2006 and incorporated herein by reference). |
| 3.2 | | Certificate of Amendment to Certificate of Incorporation of Antares Pharma, Inc. (filed as Exhibit 3.1 to Form 8-K on May 19, 2008 and incorporated herein by reference). |
| 3.3 | | Amended and Restated By-laws of Antares Pharma, Inc. (filed as Exhibit 3.1 to Form 8-K on May 15, 2007 and incorporated herein by reference). |
| 3.4 | | Certificate of Amendment to Certificate of Incorporation of Antares Pharma, Inc. (filed as Exhibit 3.1 to Form 8-K on May 28, 2013 and incorporated herein by reference). |
| 3.5 | | Certificate of Amendment to Certificate of Incorporation of Antares Pharma, Inc. (filed as Exhibit 10.3 to Form 10-Q on August 9, 2016 and incorporated herein by reference). |
| 4.1 | | Form of Certificate of Common Stock (filed as Exhibit 4.1 to Form S-1/A on August 15, 1996 and incorporated herein by reference). |
| 4.2 | | Registration Rights Agreement with Permatec Holding AG dated January 31, 2001 (filed as Exhibit 10.2 to Form 10-K on April 16, 2001 and incorporated herein by reference). |
| 4.3 | | Stock Purchase Agreement with Sicor Pharmaceuticals, Inc., dated November 23, 2005 (filed as Exhibit 10.55 to Form 10-K on March 20, 2006 and incorporated herein by reference). |
| 4.4* | | Antares Pharma, Inc. Equity Compensation Plan, as amended and restated, and approved by stockholders (filed as Exhibit 4.1 to the Company’s Form S-8 filed July 23, 2019 and incorporated herein by reference). |
| 4.5 | | Description of Company’s Securities Registered Pursuant to Section 12 of the Securities Exchange Act of 1934 (filed as Exhibit 4.5 to Form 10-K on March 3, 2020 and incorporated herein by reference). |
| 10.0 | | Stock Purchase Agreement with Permatec Holding AG, Permatec Pharma AG, Permatec Technologie AG and Permatec NV with First and Second Amendments dated July 14, 2000 (filed as an Exhibit to Schedule 14A on December 28, 2000 and incorporated herein by reference). |
| 10.1 | | Third Amendment of Stock Purchase Agreement, dated January 31, 2001 (filed as Exhibit 10.1 to Form 10-K on April 16, 2001 and incorporated herein by reference). |
| | | | | | | | |
Exhibit
No. | | Description |
| | |
| 10.2 | | Lease Agreement between Princeton South Investors, LLC and Antares Pharma, Inc., dated February 3, 2012 (filed as Exhibit 10.21 to Form 10-K for the year ended December 31, 2011 and incorporated herein by reference). |
| 10.3 | | First Amendment to Lease between Princeton South Investors, LLC and Antares Pharma, Inc., dated January 28, 2013 (filed as Exhibit 10.22 to Form 10-K for the year ended December 31, 2012 and incorporated herein by reference). |
| 10.4 | | Second Amendment to Lease between Princeton South Investors, LLC and Antares Pharma, Inc., dated December 4, 2013 (filed as Exhibit 10.22 to Form 10-K for the year ended December 31, 2013 and incorporated herein by reference) |
| 10.5 | | Third Amendment to Lease between Princeton Office Center, LLC and Antares Pharma, Inc., dated May 7, 2019 (filed as Exhibit 10.1 to Form 10-Q on August 6, 2019 and incorporated herein by reference). |
| 10.6 | | Lease Agreement between St. Paul Fire and Marine Insurance Company and Antares Pharma, Inc., dated December 20, 2013 (filed as Exhibit 10.23 to Form 10-K for the year ended December 31, 2013 and incorporated herein by reference). |
| 10.7 | | Lease Agreement by and between Antares Pharma, Inc. and Whitewater Properties I, LLC dated July 1, 2019 (filed as Exhibit 10.1 to Form 8-K on July 5, 2019 and incorporated herein by reference). |
| 10.8* | | Antares Pharma, Inc. Severance Plan, dated May 29, 2014 (filed as Exhibit 10.4 to Form 10-Q on August 7, 2014 and incorporated herein by reference). |
| 10.9 | | Form of Indemnification Agreement between Antares Pharma, Inc. and each of its directors and executive officers (filed as Exhibit 10.9 to Form 10-K on March 12, 2019 and incorporated herein by reference). |
| 10.10* | | Antares Pharma, Inc. Annual Incentive Plan, effective December 2, 2015 (filed as Exhibit 99.1 to Form 8-K on December 8, 2015 and incorporated herein by reference). |
| 10.11* | | Employment Agreement dated March 4, 2016 between Antares Pharma, Inc. and Robert F. Apple (filed as Exhibit 10.1 to Form 10-Q on May 9, 2016 and incorporated herein by reference). |
| 10.12* | | Amended and Restated Employment Agreement dated June 30, 2016 between Antares Pharma, Inc. and Peter J. Graham (filed as Exhibit 10.2 to Form 10-Q on August 9, 2016 and incorporated herein by reference). |
| 10.13* | | Employment Agreement effective October 31, 2016 between Antares Pharma, Inc. and Fred M. Powell (filed as Exhibit 10.1 to Form 10-Q on November 9, 2016 and incorporated herein by reference). |
| 10.14* | | |
| 10.15* | | Form of Nonqualified Stock Option Grant Agreement (filed as exhibit 10.4 to Form 10-Q on August 6, 2019 and incorporated herein by reference). |
| 10.16* | | Form of Restricted Stock Unit Grant Agreement (filed as Exhibit 10.5 to Form 10-Q on August 6, 2019 and incorporated herein by reference). |
| 10.17* | | Form of Restricted Stock Grant Agreement (filed as Exhibit 10.6 to Form 10-Q on August 9, 2016 and incorporated herein by reference). |
| 10.18* | | Form of Performance Stock Unit Grant Agreement (filed as Exhibit 10.6 to Form 10-Q on August 6, 2019 and incorporated herein by reference). |
| 10.19* | | Form of Nonqualified Stock Option Grant Agreement (Non-Employee Director) (filed as Exhibit 10.7 to Form 10-Q on August 6, 2019 and incorporated herein by reference). |
| | | | | | | | |
Exhibit
No. | | Description |
| | |
| 10.20* | | Form of Restricted Stock Unit Grant Agreement (Non-Employee Director) (filed as Exhibit 10.8 to Form 10-Q on August 6, 2019 and incorporated herein by reference). |
| 10.21* | | Antares Pharma, Inc. Equity Compensation Plan, as amended and restated, and approved by stockholders (filed as Exhibit 4.1 to Form S-8 filed June 24, 2021 and incorporated herein by reference). |
| 10.22 | | Credit Agreement, dated November 1, 2021, by and among Antares Pharma, Inc., Wells Fargo Bank, National Association, and the lenders from time to time party thereto (filed as Exhibit 10.1 to Form 8-K filed November 2, 2021 and incorporated herein by reference). |
| 10.23 | | Asset Purchase Agreement, dated December 15, 2021, by and between Antares Pharma, Inc., Otter Pharmaceuticals, LLC, a subsidiary of Assertio Holdings, Inc., and Assertio Holdings, Inc., as guarantor (filed herewith). |
| 23.1 | | |
| 31.1 | | |
| 31.2 | | |
| 32.1 | | |
| 32.2 | | |
| 101.INS | | XBRL Instance Document (the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document) (filed herewith). |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema Document (filed herewith). |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase Document (filed herewith). |
| 101.DEF | | Inline XBRL Taxonomy Extension Definition Linkbase Document (filed herewith). |
| 101.LAB | | Inline XBRL Taxonomy Extension Label Linkbase Document (filed herewith). |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase Document (filed herewith). |
| 104 | | Cover Page Interactive Data File (the cover page interactive data does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document) (filed herewith). |
| | |
| * | | Denotes a management contract or compensatory plan or arrangement required to be filed as an exhibit pursuant to Item 15 of Part IV of this Annual Report on Form 10-K. |
ITEM 16. FORM 10-K SUMMARY
None.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| | | | | | | | | | | |
| | | ANTARES PHARMA, INC. |
| | | (Registrant) |
| | | |
| Date: | March 3, 2022 | | /s/ Robert F. Apple |
| | | Robert F. Apple |
| | | President and Chief Executive Officer |
| | | (Principal Executive Officer) |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| | | | | | | | | | | |
| Signature | | Capacity | Date |
| /s/ Robert F. Apple | | President and Chief Executive Officer, Director | March 3, 2022 |
| Robert F. Apple | | (Principal Executive Officer) | |
| | | |
| /s/ Fred M. Powell | | Executive Vice President and Chief Financial Officer | March 3, 2022 |
| Fred M. Powell | | (Principal Financial and Accounting Officer) | |
| | | |
| /s/ Leonard S. Jacob | | Director, Chairman of the Board | March 3, 2022 |
| Dr. Leonard S. Jacob | | | |
| | | |
| /s/ Thomas J. Garrity | | Director | March 3, 2022 |
| Thomas J. Garrity | | | |
| | | |
| /s/ Peter S. Greenleaf | | Director | March 3, 2022 |
| Peter S. Greenleaf | | | |
| | | |
| /s/ Anton G. Gueth | | Director | March 3, 2022 |
| Anton G. Gueth | | | |
| | | |
| /s/ Robert P. Roche, Jr. | | Director | March 3, 2022 |
| Robert P. Roche, Jr. | | | |
| | | |
| /s/ Karen L. Smith | | Director | March 3, 2022 |
| Dr. Karen L. Smith | | | |
| | | |
| /s/ Carmen B. Volkart | | Director | March 3, 2022 |
| Carmen B. Volkart | | | |


