Exhibit 99.1

GW Pharmaceuticals Announces Physician Reports of Epidiolex® Treatment Effect in Children and Young Adults with Treatment-Resistant Epilepsy from Physician-Led Expanded Access Treatment Program
Data show promising signals of efficacy and safety
Conference Call Today at 8:30 a.m. ET, 1:30 p.m. (UK)
London, UK; 17 June 2014: GW Pharmaceuticals plc (Nasdaq: GWPH, AIM: GWP, “GW,” “the Company” or “the Group”), a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform, today announced physician reports of efficacy and safety data on 27 children and young adults with treatment-resistant epilepsy who have been treated with GW’s investigational cannabidiol (CBD) product candidate, Epidiolex®, for a period of 12 weeks. The treatment-resistant patients suffer from a range of epilepsies in which current anti-epileptic drugs have been unsuccessful in adequately controlling seizures, and included such severe forms of epilepsy as Dravet syndrome and Lennox-Gastaut syndrome. Uncontrolled data from two hospital sites in the United States that were generated under expanded access Investigational New Drug applications (INDs) authorized by the Food and Drug Administration (FDA) were made available to the Company.
“We are pleased to report preliminary data on Epidiolex which provides promising signals of efficacy in children with treatment-resistant epilepsy, including patients with Dravet syndrome. It is noteworthy that, of the patients who have responded to Epidiolex, a high proportion show a reduction in seizure frequency of greater than 50% and a portion of these patients were reported to be seizure-free at the end of 12 weeks of treatment,” stated Justin Gover, GW’s Chief Executive Officer. “We believe that these signals of efficacy, together with the side effect profile observed to date, support GW’s decision to advance Epidiolex into a formal development program and we look forward to commencing a Phase 2/3 clinical trial of Epidiolex in Dravet syndrome in the second half of 2014.”
“There remains an enormous unmet need in a range of pediatric and adult treatment-resistant epilepsy syndromes, which affect approximately 750,000 Americans. Some of the greatest needs are in children with severe epilepsy syndromes such as Dravet and Lennox-Gastaut, where frequent seizures often persist despite high doses of multiple anti-epileptic drugs,” stated Orrin Devinsky MD, Professor of Neurology, Neurosurgery, and Psychiatry at NYU School of Medicine, and Director, NYU Comprehensive Epilepsy Center. “The initial open-label study with Epidiolex has provided encouraging results. Some children have had marked reductions in their seizures and overall, the medication has been well tolerated.”
Available Data
Data were made available on 27 patients whom have been treated with Epidiolex for at least 12 weeks. Two patients commenced treatment in 2013 and the remaining 25 patients commenced treatment in the first quarter of 2014. All patients were treated at New York University Langone Medical Center (NYU) or the University of California at San Francisco (UCSF). Treatment effect data have been calculated in a manner consistent with the FDA’s recommended endpoint for evaluating efficacy, which compares percent change in the average 4 week seizure frequency throughout the 12 week treatment period to seizure frequency during a 4 week baseline period. The same approach of average 4 week seizure frequency throughout the 12 week treatment period has been used in a responder analysis.
In addition, safety data have been collected to date on a total of 62 patients (27 patients with 12 weeks treatment plus 35 additional patients who have commenced treatment under the expanded access treatment program but have yet to reach 12 weeks of treatment from NYU, UCSF and Massachusetts General Hospital in Boston).
Data were collected at hospital sites by the local medical teams and sent to GW for compiling into a database. It should be noted that expanded access studies, sometimes called “compassionate use”, are uncontrolled, carried out by individual investigators, not conducted in strict compliance with Good Clinical Practices and not intended to be analyzed together as study data. Therefore, the data reported from these programs may not be indicative of results from, or duplicated in, placebo-controlled company-sponsored clinical trials.
Epilepsy Types and Demographics
Of the 27 patients, the largest single type of epilepsy was Dravet syndrome (n=9). The remaining patients comprise a range of treatment-resistant epilepsies with convulsive and/or non-convulsive seizures.
The 27 patients were predominately children with an average age of 10.5 years (26 of the 27 patients were between 3 to 18 years of age, and 1 patient was 26 years of age). In all cases, Epidiolex was added to current anti-epileptic drugs (AEDs). On average, patients were taking 2.7 other AEDs.
Clinical Effect Data — All Patients
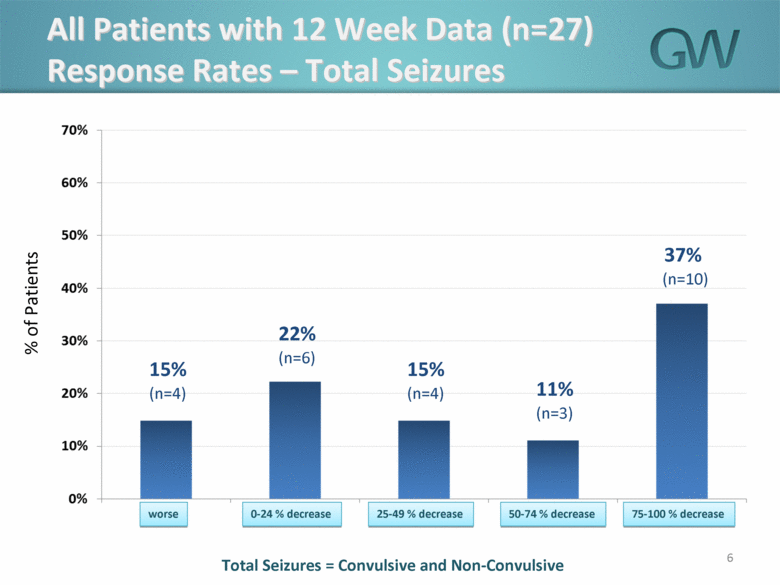
Data were made available on all 27 patients and includes information collected on all seizures (convulsive and non-convulsive) reported for each patient.
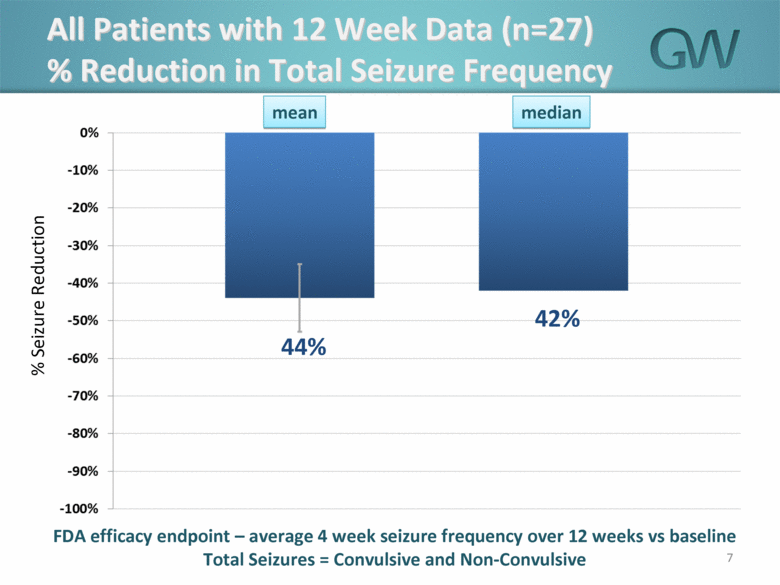
· The mean overall reduction in seizure frequency as compared to baseline seizure frequency was 44% and median overall reduction in seizure frequency as compared to baseline seizure frequency was 42%
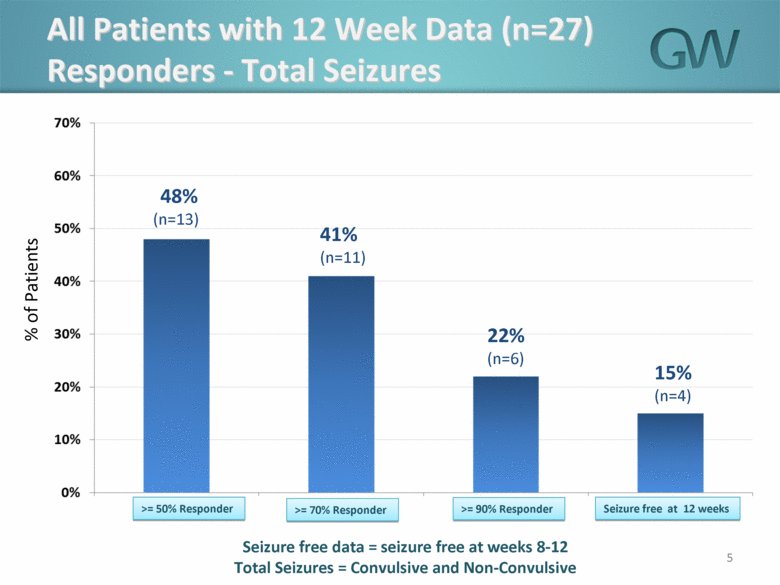
· 48% of all patients obtained at least a 50% reduction in seizure frequency as compared to baseline seizure frequency
· 41% of all patients obtained at least a 70% reduction in seizure frequency as compared to baseline seizure frequency
· 22% of all patients obtained at least a 90% reduction in seizure frequency as compared to baseline seizure frequency
· At the end of 12 weeks, 15% of all patients were seizure-free
Clinical Effect Data — Dravet syndrome patients only
With respect to the 9 patients with Dravet syndrome, the data presented below include only convulsive seizures reported for each patient, the only types of seizures considered by FDA in assessing primary efficacy for Dravet syndrome trials. The 9 patients with Dravet syndrome ranged from 3 years to 16 years of age with an average of age of 8.3 years.
· The mean reduction in seizure frequency as compared to baseline seizure frequency was 52% and median reduction in seizure frequency as compared to baseline seizure frequency was 63%
· 56% of Dravet patients obtained at least a 50% reduction in seizure frequency as compared to baseline seizure frequency
· 44% of Dravet patients obtained at least a 70% reduction in seizure frequency as compared to baseline seizure frequency
· 33% of Dravet patients obtained at least a 90% reduction in seizure frequency as compared to baseline seizure frequency
· At the end of 12 weeks, 33% of Dravet patients were seizure-free
Safety Data
Safety data were made available on 62 patients and represents approximately 120 patient-months of treatment with Epidiolex.
· At least one adverse event was reported in 81% (50) of patients
· The most common adverse events (occurring in 10% or more patients and resulting from all causes) were:
|
|
· |
Somnolence |
40% of patients |
|
|
· |
Fatigue |
26% of patients |
|
|
· |
Diarrhea |
16% of patients |
|
|
· |
Decreased appetite |
11% of patients |
|
|
· |
Increased appetite |
10% of patients |
· 80% of reported adverse events were mild or moderate
· There were no withdrawals from treatment due to adverse events
· There was 1 withdrawal from treatment due to lack of clinical effect. An additional 2 patients are currently being gradually withdrawn from treatment after 3 months due to lack of clinical effect
· Serious adverse events were reported in 7 patients, including 1 death of a patient from SUDEP (sudden unexpected death in epilepsy). None of these serious adverse events, including the 1 reported death, were deemed related to Epidiolex by the independent investigators
· In some instances, the addition of Epidiolex may be associated with changes in serum concentrations of concomitant anti-epileptic drugs
Emergency INDs
In addition to the expanded access IND patients, 5 patients have commenced treatment with Epidiolex under emergency access INDs. GW has received information from the treating physicians that 3 of these patients reported perceived benefits after treatment with Epidiolex and all 5 patients remain on treatment.
GW Development Program
GW is developing Epidiolex in the treatment of Dravet syndrome, for which the Company has received both orphan drug designation and Fast Track designation from the FDA. A company-sponsored IND is open with the FDA and a Phase 2/3 trial is expected to commence in the second half of 2014. GW anticipates commencing an additional Phase 3 trial in Dravet syndrome in the first quarter of 2015. In addition to Dravet syndrome, GW plans to conduct a clinical development program for Epidiolex in the treatment of Lennox-Gastaut syndrome (LGS). Following receipt earlier in 2014 of orphan drug designation by the FDA, GW expects to hold a pre-IND meeting with the FDA for Epidiolex in the treatment of LGS in mid-2014, and expects to conduct two Phase 3 trials in LGS during 2015.
Conference Call and Presentation
The Company will hold a conference call and broadcast a slide show today at 8:30 a.m. ET (13:30 BST) to discuss these data. The audio conference call may be accessed by dialing 877-407-8133 for domestic callers, 201-689-8040 for international callers, and toll free from the United Kingdom, 0 800 756 3429. The passcode for the call is 13584906. To view the slide show while using the audio dial-in please go to the presentation and events section of GW’s website at www.gwpharm.com. The call and slide show will also be webcast live under the events section and will be archived there following the call for 90 days. Please connect to GW’s website several minutes prior to the start of the broadcast to ensure adequate time for any software download that may be necessary. An audio replay will be available through 17 July, 2014 by calling 1-877-660-6853 (toll free) or 1-201-612-7415 (international) and entering access code 13584906.
Note Regarding Expanded Access Studies
Expanded access studies are uncontrolled, carried out by individual investigators, and not typically conducted in strict compliance with Good Clinical Practices, all of which can lead to a treatment effect which may differ from that in placebo-controlled trials. Data from these studies provide only anecdotal evidence of efficacy for regulatory review, contain no control or comparator group for reference and are not designed to be aggregated or reported as study results. Moreover, data from such small numbers of patients may be highly variable. Such information may not reliably predict data collected via systematic evaluation of the efficacy in company-sponsored clinical trials. Reliance on such information may lead to Phase 2 and 3 clinical trials that are not adequately designed to demonstrate efficacy and could delay or prevent GW’s ability to seek approval of Epidiolex. Expanded access programs may provide supportive safety information for regulatory review. Physicians conducting these studies may use Epidiolex in a manner inconsistent with the protocol, including in children with conditions different from those being studied in GW-sponsored trials. Any adverse events or reactions experienced by subjects in the expanded access program may be attributed to Epidiolex and may limit GW’s ability to obtain regulatory approval with labeling that GW considers desirable, or at all.
About GW Pharmaceuticals plc
Founded in 1998, GW is a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform in a broad range of disease areas. GW commercialized the world’s first plant-derived cannabinoid prescription drug, Sativex®, which is approved for the treatment of spasticity due to multiple sclerosis in 25 countries. Sativex is also in Phase 3 clinical development as a potential treatment of pain in people with advanced cancer. This Phase 3 program is intended to support the submission of a New Drug Application for the use of Sativex to treat cancer pain with the U.S. Food and Drug Administration and in other markets around the world. GW has a deep pipeline of additional cannabinoid product candidates, including Epidiolex which has received Orphan Drug Designation from the FDA for the treatment of Dravet and Lennox-Gastaut syndromes, severe, drug-resistant epilepsy syndromes. GW’s product pipeline also includes compounds in Phase 1 and 2 clinical development for glioma, ulcerative colitis, type-2 diabetes, and schizophrenia. For further information, please visit www.gwpharm.com.
Forward-looking statements
This news release may contain forward-looking statements that reflect GWs current expectations regarding future events, including statements regarding the therapeutic benefit, safety profile and commercial value of the company’s investigational drug Epidiolex®, the development and commercialization of Epidiolex, plans and objectives for product development, plans and objectives for present and future clinical trials and results of such trials, plans and objectives for regulatory approval. Forward-looking statements involve risks and uncertainties. Actual events could differ materially from those projected herein and depend on a number of factors, including (inter alia), the success of the GW’s research strategies, the applicability of the discoveries made therein, the successful and timely completion of uncertainties related to the regulatory process, and the acceptance of Sativex®, Epidiolex®,
and other products by consumer and medical professionals. A further list and description of risks, uncertainties and other risks associated with an investment in GW can be found in GW’s filings with the U.S. Securities and Exchange Commission. Existing and prospective investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof. GW undertakes no obligation to update or revise the information contained in this press release, whether as a result of new information, future events or circumstances or otherwise.
Enquiries:
|
GW Pharmaceuticals plc |
(Today) +44 20 3727 1000 |
|
|
|
|
Justin Gover, Chief Executive Officer |
(Thereafter) + 44 1980 557000 |
|
|
|
|
Stephen Schultz, VP Investor Relations (US) |
917 280 2424 / 401 500 6570 |
|
|
|
|
|
|
|
FTI Consulting (Media Enquiries) |
|
|
|
|
|
Ben Atwell / Simon Conway / John Dineen (UK) |
+ 44 20 3727 1000 |
|
|
|
|
Robert Stanislaro (US) |
212 850 5657 |
|
|
|
|
|
|
|
Trout Group, LLC (US investor relations) |
|
|
|
|
|
Todd James / Chad Rubin |
646 378 2900 |
|
|
|
|
|
|
|
Peel Hunt LLP (UK NOMAD) |
|
|
|
|
|
James Steel / Clare Terlouw |
+44 20 7418 8900 |